User login
Combo Tx Proves Best For Obese Binge Eaters
LAS VEGAS – Adding a weight-loss medication to cognitive-behavioral therapy for binge-eating disorder produced a higher remission rate and greater weight loss than cognitive-behavioral therapy alone in a randomized, double-blind, placebo-controlled trial.
More than a third (36%) of 25 obese binge eaters had a 5% weight loss with the combination of cognitive-behavioral therapy (CBT) and orlistat (Xenical), Carlos M. Grilo, Ph.D., reported at the annual meeting of North American Association for the Study of Obesity.
Fewer patients in the control group met the 5% standard for weight loss: only 8%, compared with the 36% of patients on combined therapy. The control group of 25 patients received CBT and a placebo.
Remission was achieved by nearly two-thirds (64%) of the combination therapy group during the 12-week study, and 52% were still in remission 3 months after the end of treatment, according to Dr. Grilo, director of the eating disorder program in the department of psychiatry at Yale University, New Haven.
In the control group, only 36% achieved remission, which was defined as no binge eating for at least 28 consecutive days.
The dosage of orlistat used in the combined therapy group was 120 mg, three times a day.
The average weight loss of 4.4 kg in the combined therapy group was small, but it was encouraging because helping binge eaters to achieve any degree of weight loss has been a major challenge. “This may appear modest, but with this patient group, it is a promising first step,” Dr. Grilo said at the meeting, which was cosponsored by the American Diabetes Association. The control group lost less weight on average–only 1.9 kg.
The Eating Disorder Examination interview was used to assess outcomes. After patients finished the program, they were encouraged to stay on a three-meal, three-snack-a-day regimen.
The trial enrolled 50 consecutive obese patients, mean age 47, who met strict criteria for binge eating. Predominantly white and female, the population averaged 13.5 binge-eating episodes per month and had an average body mass index of 36 kg/m2. Sixty percent had at least one additional psychiatric disorder, the most common of which was major depression.
The approach needs to be extended to other groups, especially diabetic binge eaters who were excluded from the study, he said.
LAS VEGAS – Adding a weight-loss medication to cognitive-behavioral therapy for binge-eating disorder produced a higher remission rate and greater weight loss than cognitive-behavioral therapy alone in a randomized, double-blind, placebo-controlled trial.
More than a third (36%) of 25 obese binge eaters had a 5% weight loss with the combination of cognitive-behavioral therapy (CBT) and orlistat (Xenical), Carlos M. Grilo, Ph.D., reported at the annual meeting of North American Association for the Study of Obesity.
Fewer patients in the control group met the 5% standard for weight loss: only 8%, compared with the 36% of patients on combined therapy. The control group of 25 patients received CBT and a placebo.
Remission was achieved by nearly two-thirds (64%) of the combination therapy group during the 12-week study, and 52% were still in remission 3 months after the end of treatment, according to Dr. Grilo, director of the eating disorder program in the department of psychiatry at Yale University, New Haven.
In the control group, only 36% achieved remission, which was defined as no binge eating for at least 28 consecutive days.
The dosage of orlistat used in the combined therapy group was 120 mg, three times a day.
The average weight loss of 4.4 kg in the combined therapy group was small, but it was encouraging because helping binge eaters to achieve any degree of weight loss has been a major challenge. “This may appear modest, but with this patient group, it is a promising first step,” Dr. Grilo said at the meeting, which was cosponsored by the American Diabetes Association. The control group lost less weight on average–only 1.9 kg.
The Eating Disorder Examination interview was used to assess outcomes. After patients finished the program, they were encouraged to stay on a three-meal, three-snack-a-day regimen.
The trial enrolled 50 consecutive obese patients, mean age 47, who met strict criteria for binge eating. Predominantly white and female, the population averaged 13.5 binge-eating episodes per month and had an average body mass index of 36 kg/m2. Sixty percent had at least one additional psychiatric disorder, the most common of which was major depression.
The approach needs to be extended to other groups, especially diabetic binge eaters who were excluded from the study, he said.
LAS VEGAS – Adding a weight-loss medication to cognitive-behavioral therapy for binge-eating disorder produced a higher remission rate and greater weight loss than cognitive-behavioral therapy alone in a randomized, double-blind, placebo-controlled trial.
More than a third (36%) of 25 obese binge eaters had a 5% weight loss with the combination of cognitive-behavioral therapy (CBT) and orlistat (Xenical), Carlos M. Grilo, Ph.D., reported at the annual meeting of North American Association for the Study of Obesity.
Fewer patients in the control group met the 5% standard for weight loss: only 8%, compared with the 36% of patients on combined therapy. The control group of 25 patients received CBT and a placebo.
Remission was achieved by nearly two-thirds (64%) of the combination therapy group during the 12-week study, and 52% were still in remission 3 months after the end of treatment, according to Dr. Grilo, director of the eating disorder program in the department of psychiatry at Yale University, New Haven.
In the control group, only 36% achieved remission, which was defined as no binge eating for at least 28 consecutive days.
The dosage of orlistat used in the combined therapy group was 120 mg, three times a day.
The average weight loss of 4.4 kg in the combined therapy group was small, but it was encouraging because helping binge eaters to achieve any degree of weight loss has been a major challenge. “This may appear modest, but with this patient group, it is a promising first step,” Dr. Grilo said at the meeting, which was cosponsored by the American Diabetes Association. The control group lost less weight on average–only 1.9 kg.
The Eating Disorder Examination interview was used to assess outcomes. After patients finished the program, they were encouraged to stay on a three-meal, three-snack-a-day regimen.
The trial enrolled 50 consecutive obese patients, mean age 47, who met strict criteria for binge eating. Predominantly white and female, the population averaged 13.5 binge-eating episodes per month and had an average body mass index of 36 kg/m2. Sixty percent had at least one additional psychiatric disorder, the most common of which was major depression.
The approach needs to be extended to other groups, especially diabetic binge eaters who were excluded from the study, he said.
PTSD Rate Highest in First Months After Brain Injury
MARCO ISLAND, FLA.–Posttraumatic stress disorder is not uncommon after moderate to severe traumatic brain injury, Jesse R. Fann, M.D., said at the annual meeting of the Academy of Psychosomatic Medicine.
Many people experience anxiety after moderate-to-severe traumatic brain injury. Because both brain injury and dissociation from posttraumatic stress disorder (PTSD) can impair declarative memory, the true occurrence of PTSD remains controversial, noted Dr. Fann, director of the psychiatry and psychology consultation service at the Seattle Cancer Care Alliance.
In a 6-month prospective follow-up study, Dr. Fann and his colleagues assessed 124 patients admitted to Harborview Medical Center in Seattle following traumatic brain injury to determine the incidence of PTSD, the risk factors, and how PTSD symptoms manifest in this population.
Researchers performed monthly assessments with the PTSD Checklist-Civilian Version, the Patient Health Questionnaire, and the Self Reported Health Status (SF-1) instruments. The first month had the highest incidence of PTSD, about 13%. “A lot of the PTSD may not be prolonged, lasting 1–3 months,” Dr. Fann said.
Patients with lower levels of education and those injured in an assault were significantly more likely to meet criteria for the disorder. Participants who met PTSD criteria most commonly reported feeling sad when recalling aspects of the event and feeling cut off from others, jumpy, hypervigilant, and irritable. Sleep disturbances were also common, he said.
The investigators looked at PTSD symptom clusters and found arousal symptoms in 23% of assessments over the 6 months. They also found intrusive symptoms in 20% and avoidance and numbing in 8%.
“There is a significant overlap of other comorbid psychiatric disorders, such as anxiety and depression, that can present a diagnostic challenge,” Dr. Fann said. “There is also overlap of PTSD and traumatic brain injury symptoms.”
The researchers also assessed patients for major depressive disorder, panic disorder, and other anxiety disorders. PTSD was significantly associated with current major depression, any other anxiety disorder, a blood alcohol level greater than 0.08, and a psychiatric history, according to a univariate analysis. A logistic regression analysis showed that people with a history of PTSD reported significantly increased functional impairment compared with those without PTSD.
The study was funded by the National Institutes of Health's National Center for Medical Rehabilitation Research.
MARCO ISLAND, FLA.–Posttraumatic stress disorder is not uncommon after moderate to severe traumatic brain injury, Jesse R. Fann, M.D., said at the annual meeting of the Academy of Psychosomatic Medicine.
Many people experience anxiety after moderate-to-severe traumatic brain injury. Because both brain injury and dissociation from posttraumatic stress disorder (PTSD) can impair declarative memory, the true occurrence of PTSD remains controversial, noted Dr. Fann, director of the psychiatry and psychology consultation service at the Seattle Cancer Care Alliance.
In a 6-month prospective follow-up study, Dr. Fann and his colleagues assessed 124 patients admitted to Harborview Medical Center in Seattle following traumatic brain injury to determine the incidence of PTSD, the risk factors, and how PTSD symptoms manifest in this population.
Researchers performed monthly assessments with the PTSD Checklist-Civilian Version, the Patient Health Questionnaire, and the Self Reported Health Status (SF-1) instruments. The first month had the highest incidence of PTSD, about 13%. “A lot of the PTSD may not be prolonged, lasting 1–3 months,” Dr. Fann said.
Patients with lower levels of education and those injured in an assault were significantly more likely to meet criteria for the disorder. Participants who met PTSD criteria most commonly reported feeling sad when recalling aspects of the event and feeling cut off from others, jumpy, hypervigilant, and irritable. Sleep disturbances were also common, he said.
The investigators looked at PTSD symptom clusters and found arousal symptoms in 23% of assessments over the 6 months. They also found intrusive symptoms in 20% and avoidance and numbing in 8%.
“There is a significant overlap of other comorbid psychiatric disorders, such as anxiety and depression, that can present a diagnostic challenge,” Dr. Fann said. “There is also overlap of PTSD and traumatic brain injury symptoms.”
The researchers also assessed patients for major depressive disorder, panic disorder, and other anxiety disorders. PTSD was significantly associated with current major depression, any other anxiety disorder, a blood alcohol level greater than 0.08, and a psychiatric history, according to a univariate analysis. A logistic regression analysis showed that people with a history of PTSD reported significantly increased functional impairment compared with those without PTSD.
The study was funded by the National Institutes of Health's National Center for Medical Rehabilitation Research.
MARCO ISLAND, FLA.–Posttraumatic stress disorder is not uncommon after moderate to severe traumatic brain injury, Jesse R. Fann, M.D., said at the annual meeting of the Academy of Psychosomatic Medicine.
Many people experience anxiety after moderate-to-severe traumatic brain injury. Because both brain injury and dissociation from posttraumatic stress disorder (PTSD) can impair declarative memory, the true occurrence of PTSD remains controversial, noted Dr. Fann, director of the psychiatry and psychology consultation service at the Seattle Cancer Care Alliance.
In a 6-month prospective follow-up study, Dr. Fann and his colleagues assessed 124 patients admitted to Harborview Medical Center in Seattle following traumatic brain injury to determine the incidence of PTSD, the risk factors, and how PTSD symptoms manifest in this population.
Researchers performed monthly assessments with the PTSD Checklist-Civilian Version, the Patient Health Questionnaire, and the Self Reported Health Status (SF-1) instruments. The first month had the highest incidence of PTSD, about 13%. “A lot of the PTSD may not be prolonged, lasting 1–3 months,” Dr. Fann said.
Patients with lower levels of education and those injured in an assault were significantly more likely to meet criteria for the disorder. Participants who met PTSD criteria most commonly reported feeling sad when recalling aspects of the event and feeling cut off from others, jumpy, hypervigilant, and irritable. Sleep disturbances were also common, he said.
The investigators looked at PTSD symptom clusters and found arousal symptoms in 23% of assessments over the 6 months. They also found intrusive symptoms in 20% and avoidance and numbing in 8%.
“There is a significant overlap of other comorbid psychiatric disorders, such as anxiety and depression, that can present a diagnostic challenge,” Dr. Fann said. “There is also overlap of PTSD and traumatic brain injury symptoms.”
The researchers also assessed patients for major depressive disorder, panic disorder, and other anxiety disorders. PTSD was significantly associated with current major depression, any other anxiety disorder, a blood alcohol level greater than 0.08, and a psychiatric history, according to a univariate analysis. A logistic regression analysis showed that people with a history of PTSD reported significantly increased functional impairment compared with those without PTSD.
The study was funded by the National Institutes of Health's National Center for Medical Rehabilitation Research.
Group Therapy Can Improve Viral Load in HIV
MARCO ISLAND, FLA.–Semistructured group therapy improves mood state in HIV-positive men, which improves cortisol levels and immune function and thereby reduces viral load, Karl Goodkin, M.D., said at the annual meeting of the Academy of Psychosomatic Medicine.
Bereavement outside of HIV has long been known to be associated with immunosuppression. Bereavement is also associated with increased mortality risk for surviving partners. The risk increases 40% for the first 6 months and 10-fold in the first year, the same time frame as observed decrements in the immune system, said Dr. Goodkin, professor of psychiatry and behavioral sciences, neurology, and psychology at the University of Miami.
In a randomized controlled trial, Dr. Goodkin and his colleagues compared the intervention with usual care in HIV-positive and HIV-negative gay men who had experienced a loss in the previous 6 months. Although effects of grief were similar, the two-tier group intervention decreased overall psychological stress–which improved mood and immune measures, including CD4 counts and viral loads–and reduced physician health visits, compared with the usual-care control group.
Participants attended a 90-minute session once weekly for 10 weeks. The groups consisted of 6–10 attendees and two professional coleaders. Enrollment was ongoing throughout the study. The first tier of the intervention fostered grief resolution; the second tier fostered stressor management, including identification of stressor impact and maladaptive behaviors. In addition to specific grief-related topics for each session, including past experiences of personal loss, reactions to surviving and implications for one's spirituality and mortality, non-bereavement-related stressor management was planned as a major focus of this unique group intervention, he said.
The usual-care group received any medical and psychosocial care that they had begun prior to initial assessment (if used consistently for 1 month or longer). Furthermore, they received four telephone calls during the 10-week intervention period to assess their clinical status. The total time for these calls was limited to 90 minutes over the 10-week period. Study staff avoided any therapeutic interactions during these calls and maintained a log documenting call content.
In the study, 166 participants (97 HIV-positive and 69 HIV-negative) completed the intervention or community usual-care group conditions. Participants were primarily in their late 30s, employed, and college educated. More than one-third were members of an ethnic minority.
The Stressor-Support-Coping model appears to have utility “with or without bereavement. We found an increase in positive life events in HIV-positive men after the intervention. Social support increased in the intervention group and declined in controls,” Dr. Goodkin noted.
Evidence from the study suggests that increased serum cortisol from stress is associated with decreases in lymphocyte proliferation in response to the artificial stimulant phytohemagglutinin in HIV-positive men and women. This is a functional measure of immunity that tends to decrease before CD4 count, he said.
The intervention decreased overall psychological distress in HIV-negative men, compared with controls, according to scores on the Distress-Grief Composite Measure. However, the decreases in grief, specifically, were less prominent than those for distress or the composite of the two measures for both the HIV-positive and -negative men.
In terms of immune effects, HIV-positive people had a true increase in their lymphocyte proliferation response up to 2 years, and the intervention provided HIV-positive participants with a buffer against decreases in CD4 levels seen in controls. The decrement among HIV-positive participants was smaller, compared with HIV-negative groups, where there was a larger spread, Dr. Goodkin explained.
All participants were asked to self-report physician health care visits in the 6 months prior to assessment. Among HIV-positive participants, there was an increase in the control group that was not as great in the intervention group. Researchers found that the same pattern held true among HIV-negative individuals. There was increased health care utilization among control participants and a decrease among the intervention group, he said.
Researchers were not able to analyze whether all health care visits were HIV or symptom related, an important caveat of the study. Another potential limitation was the difference in atmosphere between HIV-positive and HIV-negative group sessions. “HIV-positive groups talked more about concerns around their own mortality, but nonetheless it is important to note the consistency in findings across multiple domains, especially the physical domains,” Dr. Goodkin said.
“That suggests that if you improve mood state, you will improve cortisol, and you will improve immune function, which relates to improvements in viral load,” he said.
MARCO ISLAND, FLA.–Semistructured group therapy improves mood state in HIV-positive men, which improves cortisol levels and immune function and thereby reduces viral load, Karl Goodkin, M.D., said at the annual meeting of the Academy of Psychosomatic Medicine.
Bereavement outside of HIV has long been known to be associated with immunosuppression. Bereavement is also associated with increased mortality risk for surviving partners. The risk increases 40% for the first 6 months and 10-fold in the first year, the same time frame as observed decrements in the immune system, said Dr. Goodkin, professor of psychiatry and behavioral sciences, neurology, and psychology at the University of Miami.
In a randomized controlled trial, Dr. Goodkin and his colleagues compared the intervention with usual care in HIV-positive and HIV-negative gay men who had experienced a loss in the previous 6 months. Although effects of grief were similar, the two-tier group intervention decreased overall psychological stress–which improved mood and immune measures, including CD4 counts and viral loads–and reduced physician health visits, compared with the usual-care control group.
Participants attended a 90-minute session once weekly for 10 weeks. The groups consisted of 6–10 attendees and two professional coleaders. Enrollment was ongoing throughout the study. The first tier of the intervention fostered grief resolution; the second tier fostered stressor management, including identification of stressor impact and maladaptive behaviors. In addition to specific grief-related topics for each session, including past experiences of personal loss, reactions to surviving and implications for one's spirituality and mortality, non-bereavement-related stressor management was planned as a major focus of this unique group intervention, he said.
The usual-care group received any medical and psychosocial care that they had begun prior to initial assessment (if used consistently for 1 month or longer). Furthermore, they received four telephone calls during the 10-week intervention period to assess their clinical status. The total time for these calls was limited to 90 minutes over the 10-week period. Study staff avoided any therapeutic interactions during these calls and maintained a log documenting call content.
In the study, 166 participants (97 HIV-positive and 69 HIV-negative) completed the intervention or community usual-care group conditions. Participants were primarily in their late 30s, employed, and college educated. More than one-third were members of an ethnic minority.
The Stressor-Support-Coping model appears to have utility “with or without bereavement. We found an increase in positive life events in HIV-positive men after the intervention. Social support increased in the intervention group and declined in controls,” Dr. Goodkin noted.
Evidence from the study suggests that increased serum cortisol from stress is associated with decreases in lymphocyte proliferation in response to the artificial stimulant phytohemagglutinin in HIV-positive men and women. This is a functional measure of immunity that tends to decrease before CD4 count, he said.
The intervention decreased overall psychological distress in HIV-negative men, compared with controls, according to scores on the Distress-Grief Composite Measure. However, the decreases in grief, specifically, were less prominent than those for distress or the composite of the two measures for both the HIV-positive and -negative men.
In terms of immune effects, HIV-positive people had a true increase in their lymphocyte proliferation response up to 2 years, and the intervention provided HIV-positive participants with a buffer against decreases in CD4 levels seen in controls. The decrement among HIV-positive participants was smaller, compared with HIV-negative groups, where there was a larger spread, Dr. Goodkin explained.
All participants were asked to self-report physician health care visits in the 6 months prior to assessment. Among HIV-positive participants, there was an increase in the control group that was not as great in the intervention group. Researchers found that the same pattern held true among HIV-negative individuals. There was increased health care utilization among control participants and a decrease among the intervention group, he said.
Researchers were not able to analyze whether all health care visits were HIV or symptom related, an important caveat of the study. Another potential limitation was the difference in atmosphere between HIV-positive and HIV-negative group sessions. “HIV-positive groups talked more about concerns around their own mortality, but nonetheless it is important to note the consistency in findings across multiple domains, especially the physical domains,” Dr. Goodkin said.
“That suggests that if you improve mood state, you will improve cortisol, and you will improve immune function, which relates to improvements in viral load,” he said.
MARCO ISLAND, FLA.–Semistructured group therapy improves mood state in HIV-positive men, which improves cortisol levels and immune function and thereby reduces viral load, Karl Goodkin, M.D., said at the annual meeting of the Academy of Psychosomatic Medicine.
Bereavement outside of HIV has long been known to be associated with immunosuppression. Bereavement is also associated with increased mortality risk for surviving partners. The risk increases 40% for the first 6 months and 10-fold in the first year, the same time frame as observed decrements in the immune system, said Dr. Goodkin, professor of psychiatry and behavioral sciences, neurology, and psychology at the University of Miami.
In a randomized controlled trial, Dr. Goodkin and his colleagues compared the intervention with usual care in HIV-positive and HIV-negative gay men who had experienced a loss in the previous 6 months. Although effects of grief were similar, the two-tier group intervention decreased overall psychological stress–which improved mood and immune measures, including CD4 counts and viral loads–and reduced physician health visits, compared with the usual-care control group.
Participants attended a 90-minute session once weekly for 10 weeks. The groups consisted of 6–10 attendees and two professional coleaders. Enrollment was ongoing throughout the study. The first tier of the intervention fostered grief resolution; the second tier fostered stressor management, including identification of stressor impact and maladaptive behaviors. In addition to specific grief-related topics for each session, including past experiences of personal loss, reactions to surviving and implications for one's spirituality and mortality, non-bereavement-related stressor management was planned as a major focus of this unique group intervention, he said.
The usual-care group received any medical and psychosocial care that they had begun prior to initial assessment (if used consistently for 1 month or longer). Furthermore, they received four telephone calls during the 10-week intervention period to assess their clinical status. The total time for these calls was limited to 90 minutes over the 10-week period. Study staff avoided any therapeutic interactions during these calls and maintained a log documenting call content.
In the study, 166 participants (97 HIV-positive and 69 HIV-negative) completed the intervention or community usual-care group conditions. Participants were primarily in their late 30s, employed, and college educated. More than one-third were members of an ethnic minority.
The Stressor-Support-Coping model appears to have utility “with or without bereavement. We found an increase in positive life events in HIV-positive men after the intervention. Social support increased in the intervention group and declined in controls,” Dr. Goodkin noted.
Evidence from the study suggests that increased serum cortisol from stress is associated with decreases in lymphocyte proliferation in response to the artificial stimulant phytohemagglutinin in HIV-positive men and women. This is a functional measure of immunity that tends to decrease before CD4 count, he said.
The intervention decreased overall psychological distress in HIV-negative men, compared with controls, according to scores on the Distress-Grief Composite Measure. However, the decreases in grief, specifically, were less prominent than those for distress or the composite of the two measures for both the HIV-positive and -negative men.
In terms of immune effects, HIV-positive people had a true increase in their lymphocyte proliferation response up to 2 years, and the intervention provided HIV-positive participants with a buffer against decreases in CD4 levels seen in controls. The decrement among HIV-positive participants was smaller, compared with HIV-negative groups, where there was a larger spread, Dr. Goodkin explained.
All participants were asked to self-report physician health care visits in the 6 months prior to assessment. Among HIV-positive participants, there was an increase in the control group that was not as great in the intervention group. Researchers found that the same pattern held true among HIV-negative individuals. There was increased health care utilization among control participants and a decrease among the intervention group, he said.
Researchers were not able to analyze whether all health care visits were HIV or symptom related, an important caveat of the study. Another potential limitation was the difference in atmosphere between HIV-positive and HIV-negative group sessions. “HIV-positive groups talked more about concerns around their own mortality, but nonetheless it is important to note the consistency in findings across multiple domains, especially the physical domains,” Dr. Goodkin said.
“That suggests that if you improve mood state, you will improve cortisol, and you will improve immune function, which relates to improvements in viral load,” he said.
Psych Consult Delays Extend Hospital Stays
More timely psychiatric consultations could improve patient outcomes and cut costs by reducing hospital stays, reported Yasuhiro Kishi, M.D., and associates at the University of Minnesota, Minneapolis.
They reviewed the medical records of 541 consecutive patients referred for psychiatric consultation at the Fairview-University Medical Center at the university between Jan. 1, 2001, and Dec. 31, 2001 (Psychosomatics 2004;45:470–6).
The mean length of a hospital stay during the study period was 5.7 days for all patients and 18 days for patients who received psychiatric consultation. Referral time was the most predictive factor; a longer time to referral was significantly associated with a longer length of stay.
The two recommendations from a psychiatric consultation that predicted shorter stays were transfer to inpatient psychiatry and follow-up in outpatient psychiatry.
A pharmacotherapy recommendation was significantly associated with a longer stay. Although 191 patients (35%) were referred for depression, 23% of these patients were not diagnosed with depression by a psychiatrist during their referral consultations.
Overall, patients with blatant psychiatric problems were not at risk of delayed treatment, but women and patients with nonpsychiatric disorders who were in greater need of psychosocial intervention rather than psychiatric interventions were at risk for postponed care, the researchers noted.
More timely psychiatric consultations could improve patient outcomes and cut costs by reducing hospital stays, reported Yasuhiro Kishi, M.D., and associates at the University of Minnesota, Minneapolis.
They reviewed the medical records of 541 consecutive patients referred for psychiatric consultation at the Fairview-University Medical Center at the university between Jan. 1, 2001, and Dec. 31, 2001 (Psychosomatics 2004;45:470–6).
The mean length of a hospital stay during the study period was 5.7 days for all patients and 18 days for patients who received psychiatric consultation. Referral time was the most predictive factor; a longer time to referral was significantly associated with a longer length of stay.
The two recommendations from a psychiatric consultation that predicted shorter stays were transfer to inpatient psychiatry and follow-up in outpatient psychiatry.
A pharmacotherapy recommendation was significantly associated with a longer stay. Although 191 patients (35%) were referred for depression, 23% of these patients were not diagnosed with depression by a psychiatrist during their referral consultations.
Overall, patients with blatant psychiatric problems were not at risk of delayed treatment, but women and patients with nonpsychiatric disorders who were in greater need of psychosocial intervention rather than psychiatric interventions were at risk for postponed care, the researchers noted.
More timely psychiatric consultations could improve patient outcomes and cut costs by reducing hospital stays, reported Yasuhiro Kishi, M.D., and associates at the University of Minnesota, Minneapolis.
They reviewed the medical records of 541 consecutive patients referred for psychiatric consultation at the Fairview-University Medical Center at the university between Jan. 1, 2001, and Dec. 31, 2001 (Psychosomatics 2004;45:470–6).
The mean length of a hospital stay during the study period was 5.7 days for all patients and 18 days for patients who received psychiatric consultation. Referral time was the most predictive factor; a longer time to referral was significantly associated with a longer length of stay.
The two recommendations from a psychiatric consultation that predicted shorter stays were transfer to inpatient psychiatry and follow-up in outpatient psychiatry.
A pharmacotherapy recommendation was significantly associated with a longer stay. Although 191 patients (35%) were referred for depression, 23% of these patients were not diagnosed with depression by a psychiatrist during their referral consultations.
Overall, patients with blatant psychiatric problems were not at risk of delayed treatment, but women and patients with nonpsychiatric disorders who were in greater need of psychosocial intervention rather than psychiatric interventions were at risk for postponed care, the researchers noted.
Link Between Depression, Mortality Weakened
Depressive symptoms are not independent predictors of mortality, according to data from a national sample of 3,617 adults.
The findings of previous studies of associations between depressive symptoms and mortality have been inconsistent, and few of these studies have used population-based samples, said Susan A. Everson-Rose, Ph.D., of Rush University Medical Center, Chicago, and her colleagues (Psychosom. Med. 2004;66:823–30).
The investigators selected noninstitutionalized adults aged 25 years and older who were participating in an ongoing, longitudinal study called Americans' Changing Lives.
A total of 542 deaths occurred during 7.5 years of follow-up. Each increase of 1 standard unit on the Center for Epidemiological Studies Depression scale (CES-D) predicted a 21% increase in death from any cause after age, race, and gender were adjusted for. However, no excess risk of mortality was associated with CES-D scores in a fully adjusted model that included demographics, education, income, behavioral risk factors, and three indicators of health status (hypertension, functional impairment, and life-threatening conditions).
The physical complaints of patients with depression often resemble symptoms of other health problems, and distinguishing between clinical depression and poor physical health can be difficult, the investigators said.
Patients with scores in the highest quintile on the CES-D had an 85% greater risk of death from any cause, compared with participants with the lowest CES-D scores, but no other quintiles showed an increased mortality risk, Dr. Everson-Rose and her associates reported.
Depressive symptoms were not significantly associated with mortality risk in a healthy subgroup of 2,833 adults (with 306 deaths) who reported good or excellent health at baseline. In addition, depressive symptoms were not associated with increased mortality risk in patients without functional impairments at baseline.
Although depressive symptoms were associated with greater physical impairment over time after demographics, socioeconomic status, and baseline physical impairment were controlled for, the CES-D does not measure clinical depression, which has been studied as a possible link to mortality and cardiovascular health, the investigators noted.
Depressive symptoms are not independent predictors of mortality, according to data from a national sample of 3,617 adults.
The findings of previous studies of associations between depressive symptoms and mortality have been inconsistent, and few of these studies have used population-based samples, said Susan A. Everson-Rose, Ph.D., of Rush University Medical Center, Chicago, and her colleagues (Psychosom. Med. 2004;66:823–30).
The investigators selected noninstitutionalized adults aged 25 years and older who were participating in an ongoing, longitudinal study called Americans' Changing Lives.
A total of 542 deaths occurred during 7.5 years of follow-up. Each increase of 1 standard unit on the Center for Epidemiological Studies Depression scale (CES-D) predicted a 21% increase in death from any cause after age, race, and gender were adjusted for. However, no excess risk of mortality was associated with CES-D scores in a fully adjusted model that included demographics, education, income, behavioral risk factors, and three indicators of health status (hypertension, functional impairment, and life-threatening conditions).
The physical complaints of patients with depression often resemble symptoms of other health problems, and distinguishing between clinical depression and poor physical health can be difficult, the investigators said.
Patients with scores in the highest quintile on the CES-D had an 85% greater risk of death from any cause, compared with participants with the lowest CES-D scores, but no other quintiles showed an increased mortality risk, Dr. Everson-Rose and her associates reported.
Depressive symptoms were not significantly associated with mortality risk in a healthy subgroup of 2,833 adults (with 306 deaths) who reported good or excellent health at baseline. In addition, depressive symptoms were not associated with increased mortality risk in patients without functional impairments at baseline.
Although depressive symptoms were associated with greater physical impairment over time after demographics, socioeconomic status, and baseline physical impairment were controlled for, the CES-D does not measure clinical depression, which has been studied as a possible link to mortality and cardiovascular health, the investigators noted.
Depressive symptoms are not independent predictors of mortality, according to data from a national sample of 3,617 adults.
The findings of previous studies of associations between depressive symptoms and mortality have been inconsistent, and few of these studies have used population-based samples, said Susan A. Everson-Rose, Ph.D., of Rush University Medical Center, Chicago, and her colleagues (Psychosom. Med. 2004;66:823–30).
The investigators selected noninstitutionalized adults aged 25 years and older who were participating in an ongoing, longitudinal study called Americans' Changing Lives.
A total of 542 deaths occurred during 7.5 years of follow-up. Each increase of 1 standard unit on the Center for Epidemiological Studies Depression scale (CES-D) predicted a 21% increase in death from any cause after age, race, and gender were adjusted for. However, no excess risk of mortality was associated with CES-D scores in a fully adjusted model that included demographics, education, income, behavioral risk factors, and three indicators of health status (hypertension, functional impairment, and life-threatening conditions).
The physical complaints of patients with depression often resemble symptoms of other health problems, and distinguishing between clinical depression and poor physical health can be difficult, the investigators said.
Patients with scores in the highest quintile on the CES-D had an 85% greater risk of death from any cause, compared with participants with the lowest CES-D scores, but no other quintiles showed an increased mortality risk, Dr. Everson-Rose and her associates reported.
Depressive symptoms were not significantly associated with mortality risk in a healthy subgroup of 2,833 adults (with 306 deaths) who reported good or excellent health at baseline. In addition, depressive symptoms were not associated with increased mortality risk in patients without functional impairments at baseline.
Although depressive symptoms were associated with greater physical impairment over time after demographics, socioeconomic status, and baseline physical impairment were controlled for, the CES-D does not measure clinical depression, which has been studied as a possible link to mortality and cardiovascular health, the investigators noted.
Many Cardiac Patients Going Untreated
WASHINGTON – Depression is underdiagnosed in patients with acute coronary syndrome, Alpesh A. Amin, M.D., said in a poster at a scientific forum sponsored by the American Heart Association.
In a prospective study of 1,199 patients, scores on the Patient Health Questionnaire-9 consistent with depression were found in 183 (15%) at the time of hospital admission for ACS. Of those, 57 of 153 patients younger than 70 years (37%) and 4 of 30 patients aged 70 years and older (13.3%) had been clinically recognized as depressed. Only 32% of those under 70 years and 13.3% of those aged 70 years and older were taking medication to treat their depressive symptoms, said Dr. Amin of St. Luke's Hospital, Kansas City, Mo.
Recognition and treatment of depression were low across all age groups but significantly worse among patients aged 70 years and older, compared with those younger than 70 years. That gap highlights the need for improved psychiatric assessment of coronary patients, he noted.
WASHINGTON – Depression is underdiagnosed in patients with acute coronary syndrome, Alpesh A. Amin, M.D., said in a poster at a scientific forum sponsored by the American Heart Association.
In a prospective study of 1,199 patients, scores on the Patient Health Questionnaire-9 consistent with depression were found in 183 (15%) at the time of hospital admission for ACS. Of those, 57 of 153 patients younger than 70 years (37%) and 4 of 30 patients aged 70 years and older (13.3%) had been clinically recognized as depressed. Only 32% of those under 70 years and 13.3% of those aged 70 years and older were taking medication to treat their depressive symptoms, said Dr. Amin of St. Luke's Hospital, Kansas City, Mo.
Recognition and treatment of depression were low across all age groups but significantly worse among patients aged 70 years and older, compared with those younger than 70 years. That gap highlights the need for improved psychiatric assessment of coronary patients, he noted.
WASHINGTON – Depression is underdiagnosed in patients with acute coronary syndrome, Alpesh A. Amin, M.D., said in a poster at a scientific forum sponsored by the American Heart Association.
In a prospective study of 1,199 patients, scores on the Patient Health Questionnaire-9 consistent with depression were found in 183 (15%) at the time of hospital admission for ACS. Of those, 57 of 153 patients younger than 70 years (37%) and 4 of 30 patients aged 70 years and older (13.3%) had been clinically recognized as depressed. Only 32% of those under 70 years and 13.3% of those aged 70 years and older were taking medication to treat their depressive symptoms, said Dr. Amin of St. Luke's Hospital, Kansas City, Mo.
Recognition and treatment of depression were low across all age groups but significantly worse among patients aged 70 years and older, compared with those younger than 70 years. That gap highlights the need for improved psychiatric assessment of coronary patients, he noted.
Coitus May Be the Best Sexual Stress Reliever
SANTA FE, N.M. – It's been said that sex reduces stress, but all sexual activity is apparently not created equal.
In a laboratory test intended to induce extreme stress, healthy subjects who reported engaging in penile-vaginal intercourse during the previous 2 weeks experienced significantly smaller increases in blood pressure than those who reported masturbating or engaging in noncoital sexual activity, Stuart Brody, Ph.D., said at the annual meeting of the Society for Psychophysiological Research.
The subjects included 24 women and 22 men aged 19-37 years who were in the placebo arm of a larger study of the effects of vitamin C, said Dr. Brody of the University of Tübingen (Germany). They completed daily diaries about their sexual activities for a period of 2 weeks, and then their systolic and diastolic blood pressures were measured before, during, and after participating in the Trier Social Stress Test.
In this test, subjects are given 10 minutes to prepare an oral presentation on their job qualifications. After this preparatory period, they are ushered into a room where two examiners listen critically to the presentation. Subjects are then asked to perform mental calculations.
People who reported engaging only in penile-vaginal intercourse during the previous 14 days experienced significantly lower systolic blood pressure during the most stressful part of this test than did those who engaged in no sexual activity, masturbation, or noncoital intercourse.
The peak systolic blood pressure for the intercourse-only group averaged 130 mm Hg, while the other groups' averages ranged from 143 mm Hg to 165 mm Hg, which Dr. Brody described as “an enormous difference.” The effects on diastolic blood pressure were not as dramatic.
The magnitude of this effect was much greater than that reported in other studies. The beneficial effect of penile-vaginal intercourse on systolic blood pressure in the Trier Social Stress Test is apparently more pronounced than any other intervention, including whether the subjects smoke or have a family history of hypertension; whether they're using ACE inhibitors, β-blockers, or oral contraceptives; or whether they exercise, are depressed, or are in marital distress, he said.
The beneficial effect of penile-vaginal intercourse on blood pressure seemed to disappear in people who also engaged in masturbation or noncoital intercourse during the 14-day period.
The statistical significance of the results was not affected if participants had ever engaged in homosexual activity, and the groups did not differ on measures of neuroticism, extraversion, or anxiety.
Dr. Brody described his results as politically incorrect: “The politically correct thing is to parrot the ideology first espoused by Kinsey and also by Herbert Marcuse, which is that all forms of sex are equivalent, except that intercourse is worse because it's part of the patriarchal power structure.”
SANTA FE, N.M. – It's been said that sex reduces stress, but all sexual activity is apparently not created equal.
In a laboratory test intended to induce extreme stress, healthy subjects who reported engaging in penile-vaginal intercourse during the previous 2 weeks experienced significantly smaller increases in blood pressure than those who reported masturbating or engaging in noncoital sexual activity, Stuart Brody, Ph.D., said at the annual meeting of the Society for Psychophysiological Research.
The subjects included 24 women and 22 men aged 19-37 years who were in the placebo arm of a larger study of the effects of vitamin C, said Dr. Brody of the University of Tübingen (Germany). They completed daily diaries about their sexual activities for a period of 2 weeks, and then their systolic and diastolic blood pressures were measured before, during, and after participating in the Trier Social Stress Test.
In this test, subjects are given 10 minutes to prepare an oral presentation on their job qualifications. After this preparatory period, they are ushered into a room where two examiners listen critically to the presentation. Subjects are then asked to perform mental calculations.
People who reported engaging only in penile-vaginal intercourse during the previous 14 days experienced significantly lower systolic blood pressure during the most stressful part of this test than did those who engaged in no sexual activity, masturbation, or noncoital intercourse.
The peak systolic blood pressure for the intercourse-only group averaged 130 mm Hg, while the other groups' averages ranged from 143 mm Hg to 165 mm Hg, which Dr. Brody described as “an enormous difference.” The effects on diastolic blood pressure were not as dramatic.
The magnitude of this effect was much greater than that reported in other studies. The beneficial effect of penile-vaginal intercourse on systolic blood pressure in the Trier Social Stress Test is apparently more pronounced than any other intervention, including whether the subjects smoke or have a family history of hypertension; whether they're using ACE inhibitors, β-blockers, or oral contraceptives; or whether they exercise, are depressed, or are in marital distress, he said.
The beneficial effect of penile-vaginal intercourse on blood pressure seemed to disappear in people who also engaged in masturbation or noncoital intercourse during the 14-day period.
The statistical significance of the results was not affected if participants had ever engaged in homosexual activity, and the groups did not differ on measures of neuroticism, extraversion, or anxiety.
Dr. Brody described his results as politically incorrect: “The politically correct thing is to parrot the ideology first espoused by Kinsey and also by Herbert Marcuse, which is that all forms of sex are equivalent, except that intercourse is worse because it's part of the patriarchal power structure.”
SANTA FE, N.M. – It's been said that sex reduces stress, but all sexual activity is apparently not created equal.
In a laboratory test intended to induce extreme stress, healthy subjects who reported engaging in penile-vaginal intercourse during the previous 2 weeks experienced significantly smaller increases in blood pressure than those who reported masturbating or engaging in noncoital sexual activity, Stuart Brody, Ph.D., said at the annual meeting of the Society for Psychophysiological Research.
The subjects included 24 women and 22 men aged 19-37 years who were in the placebo arm of a larger study of the effects of vitamin C, said Dr. Brody of the University of Tübingen (Germany). They completed daily diaries about their sexual activities for a period of 2 weeks, and then their systolic and diastolic blood pressures were measured before, during, and after participating in the Trier Social Stress Test.
In this test, subjects are given 10 minutes to prepare an oral presentation on their job qualifications. After this preparatory period, they are ushered into a room where two examiners listen critically to the presentation. Subjects are then asked to perform mental calculations.
People who reported engaging only in penile-vaginal intercourse during the previous 14 days experienced significantly lower systolic blood pressure during the most stressful part of this test than did those who engaged in no sexual activity, masturbation, or noncoital intercourse.
The peak systolic blood pressure for the intercourse-only group averaged 130 mm Hg, while the other groups' averages ranged from 143 mm Hg to 165 mm Hg, which Dr. Brody described as “an enormous difference.” The effects on diastolic blood pressure were not as dramatic.
The magnitude of this effect was much greater than that reported in other studies. The beneficial effect of penile-vaginal intercourse on systolic blood pressure in the Trier Social Stress Test is apparently more pronounced than any other intervention, including whether the subjects smoke or have a family history of hypertension; whether they're using ACE inhibitors, β-blockers, or oral contraceptives; or whether they exercise, are depressed, or are in marital distress, he said.
The beneficial effect of penile-vaginal intercourse on blood pressure seemed to disappear in people who also engaged in masturbation or noncoital intercourse during the 14-day period.
The statistical significance of the results was not affected if participants had ever engaged in homosexual activity, and the groups did not differ on measures of neuroticism, extraversion, or anxiety.
Dr. Brody described his results as politically incorrect: “The politically correct thing is to parrot the ideology first espoused by Kinsey and also by Herbert Marcuse, which is that all forms of sex are equivalent, except that intercourse is worse because it's part of the patriarchal power structure.”
Secondary amenorrhea: Don’t dismiss it as ‘normal’
A young or middle-aged patient who stops menstruating may be pregnant or have an underlying medical problem that, left undiagnosed, could cause obesity, sexual dysfunction, infertility, osteoporosis, endometrial hyperplasia, or endometrial cancer.
Yet clinicians too often dismiss secondary amenorrhea as a “normal” result of a mental disorder or psychotropic. Psychiatrists need to:
- identify when a psychiatric disorder or drug disrupts menses
- diagnose medical causes, including thyroid dysfunction, pituitary adenomas, and polycystic ovary syndrome (PCOS).
This article outlines the most common and serious causes of secondary amenorrhea among psychiatric patients, and offers an algorithm for ruling out medical problems in nonpregnant women of child-bearing age who have stopped menstruating for 3 months. The diagnostic approach described here does not apply to women with primary amenorrhea (have never menstruated).
Table 1
Psychotropics that may cause amenorrhea
| Effect | Drug/class |
|---|---|
| Prolactin elevation | Antipsychotics (chlorpromazine, haloperidol, risperidone) SSRIs (citalopram, escitalopram, fluoxetine) |
| Sex hormone-binding globulin elevation | Carbamazepine |
| Association with PCOS unknown mechanism | Valproic acid |
| SSRIs: Selective serotonin reuptake inhibitors | |
| PCOS: Polycystic ovary syndrome | |
CASE REPORT: NO PREGNANCY, NO PERIOD
Two years ago Ms. J, age 28, was diagnosed with depression. Her psychiatrist prescribed fluoxetine, 20 mg/d titrated across 4 weeks to 40 mg/d. About 4 months later, she experienced her first manic episode. The psychiatrist changed the diagnosis to bipolar I disorder and added risperidone, 2 mg/d, to manage her mania.
Ms. J’s bipolar disorder has been under control for 1 year, but she reports that her menstruation stopped 6 months ago. She is sexually active; she and her partner use spermicide-coated condoms. She does not want to be pregnant now but might want to bear a child within the next year. Several home pregnancy tests across 6 months were negative.
The patient is obese (5 feet, 5 inches, 186 lbs, body mass index 31) and has gained about 30 pounds during the past year. Vital signs are normal; psychiatric examination indicates normal mood and affect. Skin exam reveals mild papular acne on her face and back and increased hair growth on her chin. Other physical findings—including cardiac, lung, and neurologic examinations—are normal.
Laboratory evaluation reveals a prolactin level of 105 ng/mL, a negative serum ß-Hcg reading, and normal TSH, FSH, DHEA-S and testosterone levels.
Discussion. Ms. J’s history, physical examination, and laboratory tests suggest several possible causes of secondary amenorrhea:
- Are psychotropics or a prolactin-secreting tumor elevating her prolactin level?
- Does she have PCOS, as her weight gain, hirsutism, and acne might indicate?
- Is her bipolar disorder a factor? Consider psychiatric illness, medication side effects, and medical causes when evaluating secondary amenorrhea.
PSYCHIATRIC ILLNESS
Patients with high emotional stress may have amenorrhea or menstrual irregularities related to hypothalamic dysfunction.1 Also:
Anorexia nervosa has been shown to cause hypothalamic dysfunction, leading to amenorrhea.2 A correlation exists between weight loss and menses cessation, and between regain of weight and menses resumption.2
Depression. Estradiol levels are lower in depressed women than in euthymic women, probably because of altered hypothalamic-pituitary axis (HPA) function. Also, physical distress is correlated with menses disruption.3
In a 3-year study of women ages 36 to 45,4 those with a history of depression exhibited 1.2 times the rate of perimenopause as nondepressed women. Subjects with Hamilton Rating Scale for Depression scores >8 at enrollment had twice the rate of perimenopause after 3 years compared with nondepressed women. The findings suggest that depression might increase a woman’s risk of ceasing ovarian function in her 30s or 40s. Natural menopause on average begins at age 51.5
In another study,6 16 of 32 women with PCOS had Center for Epidemiological Studies-Depression Rating Scale scores indicating depression (≥16). The study suggests a high prevalence of depression among women with PCOS, but was limited by possible selection bias, no further diagnostic evaluation for depression, small sample size, and lack of an age-matched control group.
Bipolar disorder. High rates of menstrual disturbances have been reported among women with bipolar disorder.7 Although the mechanism has not been ascertained, disruption of HPA function similar to that seen in depression is likely.7
MEDICATIONS AND AMENORRHEA
Medications can cause amenorrhea, primarily through hyperprolactinemia—although other mechanisms may be involved (Table 1). Prolactin suppresses hypothalamic luteinizing hormone-releasing hormone (LHRH) production, leading to decreased follicle-stimulating hormone (FSH) and luteinizing hormone (LH), thus reducing circulating estrogen. Prolactin-secreting pituitary tumors and drug side effects mostly commonly cause hyperprolactinemia.
Antipsychotics. Phenothiazines such as chlorpromazine, butyrophenones such as haloperidol, and the atypical antipsychotic risperidone raise prolactin levels via dopamine-receptor antagonism.
Other atypical antipsychotics—including aripiprazole, clozapine, olanzapine, quetiapine, and ziprasidone—are associated with lower serum prolactin levels than risperidone.8,9 Preliminary studies suggest, for example, that switching patients from risperidone to quetiapine may help resume menstruation without worsening psychotic symptoms,10 and that amenorrhea often resolves after the patient is switched to another atypical antipsychotic.11
SSRIs. All selective serotonin reuptake inhibitors except sertraline are associated with hyperprolactinemia and can lead to amenorrhea in some patients.12
Table 2
Differential diagnosis of secondary amenorrhea
Ovarian causes
|
Hypothalamic causes
|
Hyperprolactinemia
|
Uterine causes
|
| * Turner’s syndrome: A rare chromosomal disorder characterized by short stature, lack of sexual development at puberty. |
| † Asherman’s syndrome: Endometrial adhesions, scar tissue that develop after uterine curettage or infections. |
Anticonvulsants used as mood stabilizers to treat bipolar disorder may cause menstrual irregularities, although most data relate to women with seizure disorders.
Valproic acid has been associated with PCOS in patients with epilepsy,13 although it is unknown whether the agent’s androgenizing effects vary with age. Carbamazepine, which increases sex hormone-binding globulin, may also lead to menstrual disorders by decreasing bioavailability of circulating estrogen.14 Consider switching a patient with disrupted menses to lithium, lamotrigine, or oxcarbazepine, which have not been associated with menstrual dysfunction.
MEDICAL CAUSES
Pregnancy is the most common cause of menses cessation, followed by ovarian, hypothalamic, pituitary, or uterine dysfunction (Table 2). Hypothalamic and pituitary dysfunction often cause amenorrhea in psychiatric patients, whereas ovarian causes are common among all patients with secondary amenorrhea.15
Ovarian. In PCOS, the ovaries and sometimes the adrenal glands produce excess androgens, leading to infrequent or light periods (oligomenorrhea) or amenorrhea.
Patients with depression are prone to ovarian failure in their 30s or 40s, possibly because of chronic HPA disruption.4 Premature ovarian failure also is common among patients with Turner’s syndrome, a rare chromosomal disorder characterized by short stature and lack of sexual development at puberty. Ovarian failure also can occur spontaneously.
Hypothalamic. Functional hypothalamic amenorrhea occurs in mood and eating disorders. Emotional stress, excessive physical exercise, and nutritional deficiencies reduce LHRH secretion by the hypothalamus, which interrupts the reproductive cycle. Cardiovascular disease, respiratory disease, cancer, and other acute and chronic medical illnesses can cause significant physiologic stress, thus leading to HPA dysfunction. Hypothalamic amenorrhea is treated by targeting the underlying psychiatric or medical condition.
Pituitary. Prolactin-secreting pituitary tumors, such as a pituitary adenoma, must be ruled out in patients whose prolactin levels remain high after a medication change.15 Hypothyroidism also can trigger hyperprolactinemia by causing pituitary gland hyperplasia.
Uterine. Women who have had uterine curettage or infections can develop adhesions and scar tissue that ablate the endometrial lining. This condition, called Asherman’s syndrome, is the most common uterine cause of menstrual disruption.
EVALUATING SECONDARY AMENORRHEA
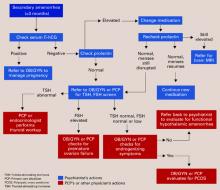
When a patient presents with secondary amenorrhea, immediately rule out pregnancy because psychiatric disorders often are managed differently in pregnant than in nonpregnant women.16
Next, take a thorough patient history to determine whether referral is necessary. Ask about weight loss (intentional or unintentional), increased stressors, or a medical illness that may point to functional hypothalamic amenorrhea. Galactorrhea or vision changes—particularly loss of peripheral vision—could suggest a pituitary tumor. Skin changes, cold intolerance, fatigue, or constipation could indicate hypothyroidism.
Menopausal symptoms such as hot flashes and vaginal dryness could point to premature ovarian failure. Galactorrhea may indicate high prolactin levels. Obesity, hirsutism, or acne could point to PCOS. Consider Asherman’s syndrome in patients with endometritis or who have had a uterine dilation and curettage.
Laboratory testing. Once pregnancy is ruled out, measure prolactin. If it exceeds 25 ng/mL by 15 ng/mL or more, do a confirmative second prolactin test. If a patient is taking a prolactin-raising medication and her prolactin was not gauged before treatment, change to a prolactin-sparing agent, then measure her prolactin 2 weeks later.17
When to refer. If prolactin persistently exceeds 50 ng/mL even after changing medications, refer the patient for brain MRI to rule out a pituitary tumor.
Tests for other underlying medical causes of secondary amenorrhea—and when to perform them—are shown in the algorithm. Psychiatrists can give these tests or refer the patient to her primary care physician.
Algorithm Laboratory evaluation of secondary amenorrhea
Communication between care team members is key to determining treatment. If a medical problem arises during psychiatric treatment, call the patient’s primary care physician or send a letter describing the problem. Also send the referring physician available lab reports.
CASE CONTINUED: TREATMENT CHANGE
Ms. J’s psychiatrist tapered risperidone to 1 mg/d for 2 weeks, then switched to olanzapine, 5 mg/d. Three weeks later, her prolactin decreased to 25 ng/mL. She continued fluoxetine, 40 mg/d, and tolerated the change in antipsychotics.
Ms. J’s bipolar disorder remains well-controlled, but menses had not resumed for another 2 months, so the psychiatrist referred Ms. J back to her primary care physician. Androgenizing and pituitary tumors were ruled out based on normal TSH, prolactin, and testosterone levels. Ms. J was diagnosed as having PCOS based on her constellation of signs and symptoms. She was started on metformin, an insulin sensitizer used to treat PCOS, and was referred to a dietitian to help her lose weight.
One year later, Ms. J still struggles with weight control, but menstruation is back to normal.
Related resources
- Lean M, De Smedt G. Schizophrenia and osteoporosis. Int Clin Psychopharmacol 2004;19:31-5.
- Berga SL, Marcus MD, Loucks TL, et al. Recovery of ovarian activity in women with functional hypothalamic amenorrhea who were treated with cognitive behavior therapy. Fertil Steril 2003;80:976-81.
- Carr BR, Bradshaw KD. Disturbances of menstruation and other common gynecologic complaints in women. In: Braunwald E, Hauser SL, Fauci AS, et al (eds). Harrison’s principles of internal medicine(15th ed). New York: McGraw-Hill, 2001:Chapter 52.
Drug brand names
- Aripiprazole • Abilify
- Carbamazepine • Tegretol
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Escitalopram • Lexapro
- Fluoxetine • Prozac
- Haloperidol • Haldol
- Lamotrigine • Lamictal
- Lithium • Eskalith, others
- Metformin • Glucovance, others
- Olanzapine • Zyprexa
- Oxcarbazepine • Trileptal
- Paroxetine • Paxil
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Sertraline • Zoloft
- Valproic acid • Depakene
- Ziprasidone • Geodon
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Kaplan JR, Manuck SB. Ovarian dysfunction, stress, and disease: a primate continuum. ILAR J 2004;45(2):89-115.
2. Mitan LA. Menstrual dysfunction in anorexia nervosa. J Pediatr Adolesc Gynecol 2004;17:81-85.
3. Young EA, Korzun A. The hypothalamic-pituitary-gonadal axis in mood disorders. Endocrinol Metab Clin North Am 2002;31(1):63-78.
4. Harlow BL, Wise LA, Otto MW, et al. Depression and its influence on reproductive endocrine and menstrual cycle markers associated with perimenopause. The Harvard Study of Moods and Cycles. Arch Gen Psychiatry 2003;60:29-36.
5. Brizendine L. Minding menopause. Psychotropics vs. estrogen? What you need to know now. Current Psychiatry 2003;2(10):12-31.
6. Rasgon NL, Rao RC, Hwang S, et al. Depression in women with polycystic ovary syndrome: clinical and biochemical correlates. J Affect Disord 2003;74(3):299-304.
7. Rasgon NL, Altshuler LL, Gundeman D, et al. Medication status and polycystic ovary syndrome in women with bipolar disorder: a preliminary report. J Clin Psychiatry 2000;61(3):173-8.
8. Kim KS, Pae CU, Chae JH, et al. Effects of olanzapine on prolactin levels of female patients with schizophrenia treated with risperidone. J Clin Psychiatry 2002;63(5):408-13.
9. Goodnick PJ, Rodriguez L, Santana O. Antipsychotics: impact on prolactin levels. Expert Opin Pharmacother 2002;3(10):1381-91.
10. Takahashi H, Higuchi H, Kamata M, et al. Effectiveness of switching to quetiapine for neuroleptic-induced amenorrhea. J Neuropsychiatry Clin Neurosci 2003;15:375-7.
11. Knegtering H, van der Moolen AE, Castelien S, et al. What are the effects of antipsychotics on sexual dysfunctions and endocrine functioning? Psychoneuroendocrinology 2003;28(suppl 2):109-23.
12. Goodnick PJ, Chaudry T, Artadi J, Arcey S. Women’s issues in mood disorders. Expert Opin Pharmacother 2000;1(5):903-16.
13. Isojarvi JI, Laatikainen TJ, Pakarinen AJ, et al. Polycystic ovaries and hyperandrogenism in women taking valproate for epilepsy. N Engl J Med 1993;329(19):1383-8.
14. Isojarvi JI. Reproductive dysfunction in women with epilepsy. Neurology 2003;61(6 Suppl 2):S27-S34.
15. Reindollar RH, Novak M, Tho SP, McDonough PG. Adult-onset amenorrhea: a study of 262 patients. Am J Obstet Gynecol 1986;155(3):531-43.
16. Altshuler L, Richards M, Yonkers K. Treating bipolar disorder during pregnancy. Current Psychiatry 2003;2(7):14-26.
17. Barbieri RL. Etiology, diagnosis and treatment of secondary amenorrhea. UpToDate 2003;12:1.-
A young or middle-aged patient who stops menstruating may be pregnant or have an underlying medical problem that, left undiagnosed, could cause obesity, sexual dysfunction, infertility, osteoporosis, endometrial hyperplasia, or endometrial cancer.
Yet clinicians too often dismiss secondary amenorrhea as a “normal” result of a mental disorder or psychotropic. Psychiatrists need to:
- identify when a psychiatric disorder or drug disrupts menses
- diagnose medical causes, including thyroid dysfunction, pituitary adenomas, and polycystic ovary syndrome (PCOS).
This article outlines the most common and serious causes of secondary amenorrhea among psychiatric patients, and offers an algorithm for ruling out medical problems in nonpregnant women of child-bearing age who have stopped menstruating for 3 months. The diagnostic approach described here does not apply to women with primary amenorrhea (have never menstruated).
Table 1
Psychotropics that may cause amenorrhea
| Effect | Drug/class |
|---|---|
| Prolactin elevation | Antipsychotics (chlorpromazine, haloperidol, risperidone) SSRIs (citalopram, escitalopram, fluoxetine) |
| Sex hormone-binding globulin elevation | Carbamazepine |
| Association with PCOS unknown mechanism | Valproic acid |
| SSRIs: Selective serotonin reuptake inhibitors | |
| PCOS: Polycystic ovary syndrome | |
CASE REPORT: NO PREGNANCY, NO PERIOD
Two years ago Ms. J, age 28, was diagnosed with depression. Her psychiatrist prescribed fluoxetine, 20 mg/d titrated across 4 weeks to 40 mg/d. About 4 months later, she experienced her first manic episode. The psychiatrist changed the diagnosis to bipolar I disorder and added risperidone, 2 mg/d, to manage her mania.
Ms. J’s bipolar disorder has been under control for 1 year, but she reports that her menstruation stopped 6 months ago. She is sexually active; she and her partner use spermicide-coated condoms. She does not want to be pregnant now but might want to bear a child within the next year. Several home pregnancy tests across 6 months were negative.
The patient is obese (5 feet, 5 inches, 186 lbs, body mass index 31) and has gained about 30 pounds during the past year. Vital signs are normal; psychiatric examination indicates normal mood and affect. Skin exam reveals mild papular acne on her face and back and increased hair growth on her chin. Other physical findings—including cardiac, lung, and neurologic examinations—are normal.
Laboratory evaluation reveals a prolactin level of 105 ng/mL, a negative serum ß-Hcg reading, and normal TSH, FSH, DHEA-S and testosterone levels.
Discussion. Ms. J’s history, physical examination, and laboratory tests suggest several possible causes of secondary amenorrhea:
- Are psychotropics or a prolactin-secreting tumor elevating her prolactin level?
- Does she have PCOS, as her weight gain, hirsutism, and acne might indicate?
- Is her bipolar disorder a factor? Consider psychiatric illness, medication side effects, and medical causes when evaluating secondary amenorrhea.
PSYCHIATRIC ILLNESS
Patients with high emotional stress may have amenorrhea or menstrual irregularities related to hypothalamic dysfunction.1 Also:
Anorexia nervosa has been shown to cause hypothalamic dysfunction, leading to amenorrhea.2 A correlation exists between weight loss and menses cessation, and between regain of weight and menses resumption.2
Depression. Estradiol levels are lower in depressed women than in euthymic women, probably because of altered hypothalamic-pituitary axis (HPA) function. Also, physical distress is correlated with menses disruption.3
In a 3-year study of women ages 36 to 45,4 those with a history of depression exhibited 1.2 times the rate of perimenopause as nondepressed women. Subjects with Hamilton Rating Scale for Depression scores >8 at enrollment had twice the rate of perimenopause after 3 years compared with nondepressed women. The findings suggest that depression might increase a woman’s risk of ceasing ovarian function in her 30s or 40s. Natural menopause on average begins at age 51.5
In another study,6 16 of 32 women with PCOS had Center for Epidemiological Studies-Depression Rating Scale scores indicating depression (≥16). The study suggests a high prevalence of depression among women with PCOS, but was limited by possible selection bias, no further diagnostic evaluation for depression, small sample size, and lack of an age-matched control group.
Bipolar disorder. High rates of menstrual disturbances have been reported among women with bipolar disorder.7 Although the mechanism has not been ascertained, disruption of HPA function similar to that seen in depression is likely.7
MEDICATIONS AND AMENORRHEA
Medications can cause amenorrhea, primarily through hyperprolactinemia—although other mechanisms may be involved (Table 1). Prolactin suppresses hypothalamic luteinizing hormone-releasing hormone (LHRH) production, leading to decreased follicle-stimulating hormone (FSH) and luteinizing hormone (LH), thus reducing circulating estrogen. Prolactin-secreting pituitary tumors and drug side effects mostly commonly cause hyperprolactinemia.
Antipsychotics. Phenothiazines such as chlorpromazine, butyrophenones such as haloperidol, and the atypical antipsychotic risperidone raise prolactin levels via dopamine-receptor antagonism.
Other atypical antipsychotics—including aripiprazole, clozapine, olanzapine, quetiapine, and ziprasidone—are associated with lower serum prolactin levels than risperidone.8,9 Preliminary studies suggest, for example, that switching patients from risperidone to quetiapine may help resume menstruation without worsening psychotic symptoms,10 and that amenorrhea often resolves after the patient is switched to another atypical antipsychotic.11
SSRIs. All selective serotonin reuptake inhibitors except sertraline are associated with hyperprolactinemia and can lead to amenorrhea in some patients.12
Table 2
Differential diagnosis of secondary amenorrhea
Ovarian causes
|
Hypothalamic causes
|
Hyperprolactinemia
|
Uterine causes
|
| * Turner’s syndrome: A rare chromosomal disorder characterized by short stature, lack of sexual development at puberty. |
| † Asherman’s syndrome: Endometrial adhesions, scar tissue that develop after uterine curettage or infections. |
Anticonvulsants used as mood stabilizers to treat bipolar disorder may cause menstrual irregularities, although most data relate to women with seizure disorders.
Valproic acid has been associated with PCOS in patients with epilepsy,13 although it is unknown whether the agent’s androgenizing effects vary with age. Carbamazepine, which increases sex hormone-binding globulin, may also lead to menstrual disorders by decreasing bioavailability of circulating estrogen.14 Consider switching a patient with disrupted menses to lithium, lamotrigine, or oxcarbazepine, which have not been associated with menstrual dysfunction.
MEDICAL CAUSES
Pregnancy is the most common cause of menses cessation, followed by ovarian, hypothalamic, pituitary, or uterine dysfunction (Table 2). Hypothalamic and pituitary dysfunction often cause amenorrhea in psychiatric patients, whereas ovarian causes are common among all patients with secondary amenorrhea.15
Ovarian. In PCOS, the ovaries and sometimes the adrenal glands produce excess androgens, leading to infrequent or light periods (oligomenorrhea) or amenorrhea.
Patients with depression are prone to ovarian failure in their 30s or 40s, possibly because of chronic HPA disruption.4 Premature ovarian failure also is common among patients with Turner’s syndrome, a rare chromosomal disorder characterized by short stature and lack of sexual development at puberty. Ovarian failure also can occur spontaneously.
Hypothalamic. Functional hypothalamic amenorrhea occurs in mood and eating disorders. Emotional stress, excessive physical exercise, and nutritional deficiencies reduce LHRH secretion by the hypothalamus, which interrupts the reproductive cycle. Cardiovascular disease, respiratory disease, cancer, and other acute and chronic medical illnesses can cause significant physiologic stress, thus leading to HPA dysfunction. Hypothalamic amenorrhea is treated by targeting the underlying psychiatric or medical condition.
Pituitary. Prolactin-secreting pituitary tumors, such as a pituitary adenoma, must be ruled out in patients whose prolactin levels remain high after a medication change.15 Hypothyroidism also can trigger hyperprolactinemia by causing pituitary gland hyperplasia.
Uterine. Women who have had uterine curettage or infections can develop adhesions and scar tissue that ablate the endometrial lining. This condition, called Asherman’s syndrome, is the most common uterine cause of menstrual disruption.
EVALUATING SECONDARY AMENORRHEA
When a patient presents with secondary amenorrhea, immediately rule out pregnancy because psychiatric disorders often are managed differently in pregnant than in nonpregnant women.16
Next, take a thorough patient history to determine whether referral is necessary. Ask about weight loss (intentional or unintentional), increased stressors, or a medical illness that may point to functional hypothalamic amenorrhea. Galactorrhea or vision changes—particularly loss of peripheral vision—could suggest a pituitary tumor. Skin changes, cold intolerance, fatigue, or constipation could indicate hypothyroidism.
Menopausal symptoms such as hot flashes and vaginal dryness could point to premature ovarian failure. Galactorrhea may indicate high prolactin levels. Obesity, hirsutism, or acne could point to PCOS. Consider Asherman’s syndrome in patients with endometritis or who have had a uterine dilation and curettage.
Laboratory testing. Once pregnancy is ruled out, measure prolactin. If it exceeds 25 ng/mL by 15 ng/mL or more, do a confirmative second prolactin test. If a patient is taking a prolactin-raising medication and her prolactin was not gauged before treatment, change to a prolactin-sparing agent, then measure her prolactin 2 weeks later.17
When to refer. If prolactin persistently exceeds 50 ng/mL even after changing medications, refer the patient for brain MRI to rule out a pituitary tumor.
Tests for other underlying medical causes of secondary amenorrhea—and when to perform them—are shown in the algorithm. Psychiatrists can give these tests or refer the patient to her primary care physician.
Algorithm Laboratory evaluation of secondary amenorrhea
Communication between care team members is key to determining treatment. If a medical problem arises during psychiatric treatment, call the patient’s primary care physician or send a letter describing the problem. Also send the referring physician available lab reports.
CASE CONTINUED: TREATMENT CHANGE
Ms. J’s psychiatrist tapered risperidone to 1 mg/d for 2 weeks, then switched to olanzapine, 5 mg/d. Three weeks later, her prolactin decreased to 25 ng/mL. She continued fluoxetine, 40 mg/d, and tolerated the change in antipsychotics.
Ms. J’s bipolar disorder remains well-controlled, but menses had not resumed for another 2 months, so the psychiatrist referred Ms. J back to her primary care physician. Androgenizing and pituitary tumors were ruled out based on normal TSH, prolactin, and testosterone levels. Ms. J was diagnosed as having PCOS based on her constellation of signs and symptoms. She was started on metformin, an insulin sensitizer used to treat PCOS, and was referred to a dietitian to help her lose weight.
One year later, Ms. J still struggles with weight control, but menstruation is back to normal.
Related resources
- Lean M, De Smedt G. Schizophrenia and osteoporosis. Int Clin Psychopharmacol 2004;19:31-5.
- Berga SL, Marcus MD, Loucks TL, et al. Recovery of ovarian activity in women with functional hypothalamic amenorrhea who were treated with cognitive behavior therapy. Fertil Steril 2003;80:976-81.
- Carr BR, Bradshaw KD. Disturbances of menstruation and other common gynecologic complaints in women. In: Braunwald E, Hauser SL, Fauci AS, et al (eds). Harrison’s principles of internal medicine(15th ed). New York: McGraw-Hill, 2001:Chapter 52.
Drug brand names
- Aripiprazole • Abilify
- Carbamazepine • Tegretol
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Escitalopram • Lexapro
- Fluoxetine • Prozac
- Haloperidol • Haldol
- Lamotrigine • Lamictal
- Lithium • Eskalith, others
- Metformin • Glucovance, others
- Olanzapine • Zyprexa
- Oxcarbazepine • Trileptal
- Paroxetine • Paxil
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Sertraline • Zoloft
- Valproic acid • Depakene
- Ziprasidone • Geodon
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
A young or middle-aged patient who stops menstruating may be pregnant or have an underlying medical problem that, left undiagnosed, could cause obesity, sexual dysfunction, infertility, osteoporosis, endometrial hyperplasia, or endometrial cancer.
Yet clinicians too often dismiss secondary amenorrhea as a “normal” result of a mental disorder or psychotropic. Psychiatrists need to:
- identify when a psychiatric disorder or drug disrupts menses
- diagnose medical causes, including thyroid dysfunction, pituitary adenomas, and polycystic ovary syndrome (PCOS).
This article outlines the most common and serious causes of secondary amenorrhea among psychiatric patients, and offers an algorithm for ruling out medical problems in nonpregnant women of child-bearing age who have stopped menstruating for 3 months. The diagnostic approach described here does not apply to women with primary amenorrhea (have never menstruated).
Table 1
Psychotropics that may cause amenorrhea
| Effect | Drug/class |
|---|---|
| Prolactin elevation | Antipsychotics (chlorpromazine, haloperidol, risperidone) SSRIs (citalopram, escitalopram, fluoxetine) |
| Sex hormone-binding globulin elevation | Carbamazepine |
| Association with PCOS unknown mechanism | Valproic acid |
| SSRIs: Selective serotonin reuptake inhibitors | |
| PCOS: Polycystic ovary syndrome | |
CASE REPORT: NO PREGNANCY, NO PERIOD
Two years ago Ms. J, age 28, was diagnosed with depression. Her psychiatrist prescribed fluoxetine, 20 mg/d titrated across 4 weeks to 40 mg/d. About 4 months later, she experienced her first manic episode. The psychiatrist changed the diagnosis to bipolar I disorder and added risperidone, 2 mg/d, to manage her mania.
Ms. J’s bipolar disorder has been under control for 1 year, but she reports that her menstruation stopped 6 months ago. She is sexually active; she and her partner use spermicide-coated condoms. She does not want to be pregnant now but might want to bear a child within the next year. Several home pregnancy tests across 6 months were negative.
The patient is obese (5 feet, 5 inches, 186 lbs, body mass index 31) and has gained about 30 pounds during the past year. Vital signs are normal; psychiatric examination indicates normal mood and affect. Skin exam reveals mild papular acne on her face and back and increased hair growth on her chin. Other physical findings—including cardiac, lung, and neurologic examinations—are normal.
Laboratory evaluation reveals a prolactin level of 105 ng/mL, a negative serum ß-Hcg reading, and normal TSH, FSH, DHEA-S and testosterone levels.
Discussion. Ms. J’s history, physical examination, and laboratory tests suggest several possible causes of secondary amenorrhea:
- Are psychotropics or a prolactin-secreting tumor elevating her prolactin level?
- Does she have PCOS, as her weight gain, hirsutism, and acne might indicate?
- Is her bipolar disorder a factor? Consider psychiatric illness, medication side effects, and medical causes when evaluating secondary amenorrhea.
PSYCHIATRIC ILLNESS
Patients with high emotional stress may have amenorrhea or menstrual irregularities related to hypothalamic dysfunction.1 Also:
Anorexia nervosa has been shown to cause hypothalamic dysfunction, leading to amenorrhea.2 A correlation exists between weight loss and menses cessation, and between regain of weight and menses resumption.2
Depression. Estradiol levels are lower in depressed women than in euthymic women, probably because of altered hypothalamic-pituitary axis (HPA) function. Also, physical distress is correlated with menses disruption.3
In a 3-year study of women ages 36 to 45,4 those with a history of depression exhibited 1.2 times the rate of perimenopause as nondepressed women. Subjects with Hamilton Rating Scale for Depression scores >8 at enrollment had twice the rate of perimenopause after 3 years compared with nondepressed women. The findings suggest that depression might increase a woman’s risk of ceasing ovarian function in her 30s or 40s. Natural menopause on average begins at age 51.5
In another study,6 16 of 32 women with PCOS had Center for Epidemiological Studies-Depression Rating Scale scores indicating depression (≥16). The study suggests a high prevalence of depression among women with PCOS, but was limited by possible selection bias, no further diagnostic evaluation for depression, small sample size, and lack of an age-matched control group.
Bipolar disorder. High rates of menstrual disturbances have been reported among women with bipolar disorder.7 Although the mechanism has not been ascertained, disruption of HPA function similar to that seen in depression is likely.7
MEDICATIONS AND AMENORRHEA
Medications can cause amenorrhea, primarily through hyperprolactinemia—although other mechanisms may be involved (Table 1). Prolactin suppresses hypothalamic luteinizing hormone-releasing hormone (LHRH) production, leading to decreased follicle-stimulating hormone (FSH) and luteinizing hormone (LH), thus reducing circulating estrogen. Prolactin-secreting pituitary tumors and drug side effects mostly commonly cause hyperprolactinemia.
Antipsychotics. Phenothiazines such as chlorpromazine, butyrophenones such as haloperidol, and the atypical antipsychotic risperidone raise prolactin levels via dopamine-receptor antagonism.
Other atypical antipsychotics—including aripiprazole, clozapine, olanzapine, quetiapine, and ziprasidone—are associated with lower serum prolactin levels than risperidone.8,9 Preliminary studies suggest, for example, that switching patients from risperidone to quetiapine may help resume menstruation without worsening psychotic symptoms,10 and that amenorrhea often resolves after the patient is switched to another atypical antipsychotic.11
SSRIs. All selective serotonin reuptake inhibitors except sertraline are associated with hyperprolactinemia and can lead to amenorrhea in some patients.12
Table 2
Differential diagnosis of secondary amenorrhea
Ovarian causes
|
Hypothalamic causes
|
Hyperprolactinemia
|
Uterine causes
|
| * Turner’s syndrome: A rare chromosomal disorder characterized by short stature, lack of sexual development at puberty. |
| † Asherman’s syndrome: Endometrial adhesions, scar tissue that develop after uterine curettage or infections. |
Anticonvulsants used as mood stabilizers to treat bipolar disorder may cause menstrual irregularities, although most data relate to women with seizure disorders.
Valproic acid has been associated with PCOS in patients with epilepsy,13 although it is unknown whether the agent’s androgenizing effects vary with age. Carbamazepine, which increases sex hormone-binding globulin, may also lead to menstrual disorders by decreasing bioavailability of circulating estrogen.14 Consider switching a patient with disrupted menses to lithium, lamotrigine, or oxcarbazepine, which have not been associated with menstrual dysfunction.
MEDICAL CAUSES
Pregnancy is the most common cause of menses cessation, followed by ovarian, hypothalamic, pituitary, or uterine dysfunction (Table 2). Hypothalamic and pituitary dysfunction often cause amenorrhea in psychiatric patients, whereas ovarian causes are common among all patients with secondary amenorrhea.15
Ovarian. In PCOS, the ovaries and sometimes the adrenal glands produce excess androgens, leading to infrequent or light periods (oligomenorrhea) or amenorrhea.
Patients with depression are prone to ovarian failure in their 30s or 40s, possibly because of chronic HPA disruption.4 Premature ovarian failure also is common among patients with Turner’s syndrome, a rare chromosomal disorder characterized by short stature and lack of sexual development at puberty. Ovarian failure also can occur spontaneously.
Hypothalamic. Functional hypothalamic amenorrhea occurs in mood and eating disorders. Emotional stress, excessive physical exercise, and nutritional deficiencies reduce LHRH secretion by the hypothalamus, which interrupts the reproductive cycle. Cardiovascular disease, respiratory disease, cancer, and other acute and chronic medical illnesses can cause significant physiologic stress, thus leading to HPA dysfunction. Hypothalamic amenorrhea is treated by targeting the underlying psychiatric or medical condition.
Pituitary. Prolactin-secreting pituitary tumors, such as a pituitary adenoma, must be ruled out in patients whose prolactin levels remain high after a medication change.15 Hypothyroidism also can trigger hyperprolactinemia by causing pituitary gland hyperplasia.
Uterine. Women who have had uterine curettage or infections can develop adhesions and scar tissue that ablate the endometrial lining. This condition, called Asherman’s syndrome, is the most common uterine cause of menstrual disruption.
EVALUATING SECONDARY AMENORRHEA
When a patient presents with secondary amenorrhea, immediately rule out pregnancy because psychiatric disorders often are managed differently in pregnant than in nonpregnant women.16
Next, take a thorough patient history to determine whether referral is necessary. Ask about weight loss (intentional or unintentional), increased stressors, or a medical illness that may point to functional hypothalamic amenorrhea. Galactorrhea or vision changes—particularly loss of peripheral vision—could suggest a pituitary tumor. Skin changes, cold intolerance, fatigue, or constipation could indicate hypothyroidism.
Menopausal symptoms such as hot flashes and vaginal dryness could point to premature ovarian failure. Galactorrhea may indicate high prolactin levels. Obesity, hirsutism, or acne could point to PCOS. Consider Asherman’s syndrome in patients with endometritis or who have had a uterine dilation and curettage.
Laboratory testing. Once pregnancy is ruled out, measure prolactin. If it exceeds 25 ng/mL by 15 ng/mL or more, do a confirmative second prolactin test. If a patient is taking a prolactin-raising medication and her prolactin was not gauged before treatment, change to a prolactin-sparing agent, then measure her prolactin 2 weeks later.17
When to refer. If prolactin persistently exceeds 50 ng/mL even after changing medications, refer the patient for brain MRI to rule out a pituitary tumor.
Tests for other underlying medical causes of secondary amenorrhea—and when to perform them—are shown in the algorithm. Psychiatrists can give these tests or refer the patient to her primary care physician.
Algorithm Laboratory evaluation of secondary amenorrhea
Communication between care team members is key to determining treatment. If a medical problem arises during psychiatric treatment, call the patient’s primary care physician or send a letter describing the problem. Also send the referring physician available lab reports.
CASE CONTINUED: TREATMENT CHANGE
Ms. J’s psychiatrist tapered risperidone to 1 mg/d for 2 weeks, then switched to olanzapine, 5 mg/d. Three weeks later, her prolactin decreased to 25 ng/mL. She continued fluoxetine, 40 mg/d, and tolerated the change in antipsychotics.
Ms. J’s bipolar disorder remains well-controlled, but menses had not resumed for another 2 months, so the psychiatrist referred Ms. J back to her primary care physician. Androgenizing and pituitary tumors were ruled out based on normal TSH, prolactin, and testosterone levels. Ms. J was diagnosed as having PCOS based on her constellation of signs and symptoms. She was started on metformin, an insulin sensitizer used to treat PCOS, and was referred to a dietitian to help her lose weight.
One year later, Ms. J still struggles with weight control, but menstruation is back to normal.
Related resources
- Lean M, De Smedt G. Schizophrenia and osteoporosis. Int Clin Psychopharmacol 2004;19:31-5.
- Berga SL, Marcus MD, Loucks TL, et al. Recovery of ovarian activity in women with functional hypothalamic amenorrhea who were treated with cognitive behavior therapy. Fertil Steril 2003;80:976-81.
- Carr BR, Bradshaw KD. Disturbances of menstruation and other common gynecologic complaints in women. In: Braunwald E, Hauser SL, Fauci AS, et al (eds). Harrison’s principles of internal medicine(15th ed). New York: McGraw-Hill, 2001:Chapter 52.
Drug brand names
- Aripiprazole • Abilify
- Carbamazepine • Tegretol
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Escitalopram • Lexapro
- Fluoxetine • Prozac
- Haloperidol • Haldol
- Lamotrigine • Lamictal
- Lithium • Eskalith, others
- Metformin • Glucovance, others
- Olanzapine • Zyprexa
- Oxcarbazepine • Trileptal
- Paroxetine • Paxil
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Sertraline • Zoloft
- Valproic acid • Depakene
- Ziprasidone • Geodon
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Kaplan JR, Manuck SB. Ovarian dysfunction, stress, and disease: a primate continuum. ILAR J 2004;45(2):89-115.
2. Mitan LA. Menstrual dysfunction in anorexia nervosa. J Pediatr Adolesc Gynecol 2004;17:81-85.
3. Young EA, Korzun A. The hypothalamic-pituitary-gonadal axis in mood disorders. Endocrinol Metab Clin North Am 2002;31(1):63-78.
4. Harlow BL, Wise LA, Otto MW, et al. Depression and its influence on reproductive endocrine and menstrual cycle markers associated with perimenopause. The Harvard Study of Moods and Cycles. Arch Gen Psychiatry 2003;60:29-36.
5. Brizendine L. Minding menopause. Psychotropics vs. estrogen? What you need to know now. Current Psychiatry 2003;2(10):12-31.
6. Rasgon NL, Rao RC, Hwang S, et al. Depression in women with polycystic ovary syndrome: clinical and biochemical correlates. J Affect Disord 2003;74(3):299-304.
7. Rasgon NL, Altshuler LL, Gundeman D, et al. Medication status and polycystic ovary syndrome in women with bipolar disorder: a preliminary report. J Clin Psychiatry 2000;61(3):173-8.
8. Kim KS, Pae CU, Chae JH, et al. Effects of olanzapine on prolactin levels of female patients with schizophrenia treated with risperidone. J Clin Psychiatry 2002;63(5):408-13.
9. Goodnick PJ, Rodriguez L, Santana O. Antipsychotics: impact on prolactin levels. Expert Opin Pharmacother 2002;3(10):1381-91.
10. Takahashi H, Higuchi H, Kamata M, et al. Effectiveness of switching to quetiapine for neuroleptic-induced amenorrhea. J Neuropsychiatry Clin Neurosci 2003;15:375-7.
11. Knegtering H, van der Moolen AE, Castelien S, et al. What are the effects of antipsychotics on sexual dysfunctions and endocrine functioning? Psychoneuroendocrinology 2003;28(suppl 2):109-23.
12. Goodnick PJ, Chaudry T, Artadi J, Arcey S. Women’s issues in mood disorders. Expert Opin Pharmacother 2000;1(5):903-16.
13. Isojarvi JI, Laatikainen TJ, Pakarinen AJ, et al. Polycystic ovaries and hyperandrogenism in women taking valproate for epilepsy. N Engl J Med 1993;329(19):1383-8.
14. Isojarvi JI. Reproductive dysfunction in women with epilepsy. Neurology 2003;61(6 Suppl 2):S27-S34.
15. Reindollar RH, Novak M, Tho SP, McDonough PG. Adult-onset amenorrhea: a study of 262 patients. Am J Obstet Gynecol 1986;155(3):531-43.
16. Altshuler L, Richards M, Yonkers K. Treating bipolar disorder during pregnancy. Current Psychiatry 2003;2(7):14-26.
17. Barbieri RL. Etiology, diagnosis and treatment of secondary amenorrhea. UpToDate 2003;12:1.-
1. Kaplan JR, Manuck SB. Ovarian dysfunction, stress, and disease: a primate continuum. ILAR J 2004;45(2):89-115.
2. Mitan LA. Menstrual dysfunction in anorexia nervosa. J Pediatr Adolesc Gynecol 2004;17:81-85.
3. Young EA, Korzun A. The hypothalamic-pituitary-gonadal axis in mood disorders. Endocrinol Metab Clin North Am 2002;31(1):63-78.
4. Harlow BL, Wise LA, Otto MW, et al. Depression and its influence on reproductive endocrine and menstrual cycle markers associated with perimenopause. The Harvard Study of Moods and Cycles. Arch Gen Psychiatry 2003;60:29-36.
5. Brizendine L. Minding menopause. Psychotropics vs. estrogen? What you need to know now. Current Psychiatry 2003;2(10):12-31.
6. Rasgon NL, Rao RC, Hwang S, et al. Depression in women with polycystic ovary syndrome: clinical and biochemical correlates. J Affect Disord 2003;74(3):299-304.
7. Rasgon NL, Altshuler LL, Gundeman D, et al. Medication status and polycystic ovary syndrome in women with bipolar disorder: a preliminary report. J Clin Psychiatry 2000;61(3):173-8.
8. Kim KS, Pae CU, Chae JH, et al. Effects of olanzapine on prolactin levels of female patients with schizophrenia treated with risperidone. J Clin Psychiatry 2002;63(5):408-13.
9. Goodnick PJ, Rodriguez L, Santana O. Antipsychotics: impact on prolactin levels. Expert Opin Pharmacother 2002;3(10):1381-91.
10. Takahashi H, Higuchi H, Kamata M, et al. Effectiveness of switching to quetiapine for neuroleptic-induced amenorrhea. J Neuropsychiatry Clin Neurosci 2003;15:375-7.
11. Knegtering H, van der Moolen AE, Castelien S, et al. What are the effects of antipsychotics on sexual dysfunctions and endocrine functioning? Psychoneuroendocrinology 2003;28(suppl 2):109-23.
12. Goodnick PJ, Chaudry T, Artadi J, Arcey S. Women’s issues in mood disorders. Expert Opin Pharmacother 2000;1(5):903-16.
13. Isojarvi JI, Laatikainen TJ, Pakarinen AJ, et al. Polycystic ovaries and hyperandrogenism in women taking valproate for epilepsy. N Engl J Med 1993;329(19):1383-8.
14. Isojarvi JI. Reproductive dysfunction in women with epilepsy. Neurology 2003;61(6 Suppl 2):S27-S34.
15. Reindollar RH, Novak M, Tho SP, McDonough PG. Adult-onset amenorrhea: a study of 262 patients. Am J Obstet Gynecol 1986;155(3):531-43.
16. Altshuler L, Richards M, Yonkers K. Treating bipolar disorder during pregnancy. Current Psychiatry 2003;2(7):14-26.
17. Barbieri RL. Etiology, diagnosis and treatment of secondary amenorrhea. UpToDate 2003;12:1.-
Spotting a silent killer
CASE 1: BEWARE ‘OLD MAN KIPLING’
Mrs. A, age 87, has Alzheimer’s disease. About 1 month before presentation, she entered a nursing home because of increasing agitation, paranoia, auditory and visual hallucinations, and decreased ability to care for herself. Her doctor started risperidone, 0.5 mg bid, to treat her agitation and psychosis.
Two days later, Mrs. A barricaded herself in her room. She told staff that “Old Man Kipling” was trying to break in, steal her money, and kill her and her son. She was sent to the emergency room; psychiatric consultation was ordered.
Mrs. A also has hypertension, renal cell carcinoma, anemia, and chronic renal failure. She had seen a psychiatrist for worsening cognitive function but has no other psychiatric history. Brain CT without contrast revealed generalized atrophy with no acute cerebral events. Workup showed decreased potassium (3.1 mEq/L), which returned to normal after Mrs. A was given potassium chloride, 20 mEq/d for 5 days. Other lab results were normal. Hydrochlorothiazide, 25 mg/d for hypertension, was stopped to prevent potassium depletion. No neurologic deficits were found.
Upon admission to the geriatric psychiatry unit, Mrs. A was paranoid and agitated. She talked to an imaginary person, continued to fear “Old Man Kipling,” and again tried to barricade herself.
ECG at admission—done because of Mrs. A’s age, cardiac history, and hydrochlorothiazide use—showed a corrected QT (QTc) interval of 494 msec, nearly 50 msec above the high-normal range for women. The interval was 460 msec at baseline (before risperidone treatment). Mrs. A was switched to olanzapine, 5 mg at bedtime, but her QTc intervals stayed between 494 and 495 msec, and her psychotic symptoms continued unabated.
Table 1
Mean antipsychotic-induced QTc interval change from baseline to steady state
| Antipsychotic | Mean QTc interval change |
|---|---|
| Haloperidol | 4.7 msec |
| Olanzapine | 6.4 msec |
| Risperidone | 10.0 msec |
| Quetiapine | 14.5 msec |
| Ziprasidone | 20.6 msec |
| Thioridazine | 35.8 msec |
| Source: reference 2. | |
The authors’ observations
Antipsychotics, used to treat behavioral disturbances in older patients, can prolong QTc intervals. Although often asymptomatic, a prolonged interval can lead to torsade de pointes, a polymorphic ventricular arrhythmia that can progress to ventricular fibrillation and cause sudden death.
Reilly et al1 suggest that antipsychotic-induced QTc prolongation may be dose-dependent. Age >65 is also a risk factor.
Start low and go slow when prescribing antipsychotics to patients with QTc intervals 450 msec. If prolonged intervals persist, switch antipsychotics and consult a cardiologist to help manage the patient’s care.
Switching agents will not entirely eliminate the risk, however. Mrs. A’s QTc interval remained elevated despite the switch to olanzapine, which is less likely than most antipsychotics to increase the interval.
Among mostly healthy men, haloperidol was shown to cause a lower mean QTc interval increase than other antipsychotics (Table 1), although QTc prolongations >60 msec were reported in 4% of those who took haloperidol.2 The agent also may cause tardive dyskinesia, and that risk is multiplied in patients >age 65.3 For Mrs. A, however, persistent psychosis and declining function outweighed the risks.
With haloperidol, start low and titrate slowly to reduce the risk of extrapyramidal symptoms (EPS). Decrease the dosage if involuntary movements develop. If a haloperidol decrease would lead to decompensation, add an anticholinergic agent such as benztropine, but be careful because anticholinergics can worsen cognitive function.
Test for involuntary movements before starting an antipsychotic. Retest every 4 to 6 months, when changing dosages or switching antipsychotics, and when patients complain of EPS.
CASE 1 CONTINUED: GOODBYE MR. KIPLING
Mrs. A was switched to haloperidol, 0.5 mg bid titrated over 3 weeks to 2 mg every morning and 3 mg nightly. Daily ECGs across 10 days showed QTc intervals 467 msec. Abnormal Involuntary Movement Scale testing showed no EPS. Her blood pressure was stable, ranging from 110 to 130 mm Hg (systolic) and 70 to 80 mm Hg (diastolic).
The patient became calmer and her paranoid delusions and hallucinations disappeared. Her Folstein Mini-Mental Status Examination score during her third and final week of hospitalization was 16, indicating moderate dementia. She was discharged to her son’s care; outpatient psychiatric care was also arranged. The psychiatrist started donepezil, 5 mg/d titrated to 10 mg/d after 6 weeks, to treat her memory impairments.
More than 1 year later, Mrs. A lives at home with her son. She has not needed psychiatric hospitalization. Her primary care physician monitors her cardiac health.
CASE 2: SUICIDALITY AND SEXUAL BEHAVIOR
Mr. B, age 50, has battled schizoaffective disorder for more than 30 years. Upon presenting to the ER, he told clinicians he planned to jump from his seventh-floor apartment after arguing with his neighbor.
The patient had been taking gabapentin, 300 mg bid; olanzapine, 10 mg at bedtime; citalopram, 20 mg/d; clonazepam, 1 mg at bedtime for panic symptoms; atorvastatin, 10 mg/d for hyperlipidemia; and esomeprazole, 40 mg/d, for ongoing GI problems. He also has bradycardia.
Electrolyte and magnesium levels, thyroid function, and liver function tests were normal. Potassium was 3.9 mEq/L, indicating possible deficiency. Toxicity screen was negative, ruling out substance abuse or medication overdose. Baseline ECG—ordered because of Mr. B’s bradycardia—showed a QTc interval of 519 msec (almost 80 msec above high-normal for men) and a heart rate of 50 bpm.
The cardiology team found that 1 year before, while being examined for suspected syncope, Mr. B had a prolonged QTc interval that resolved after olanzapine was stopped. Acting on cardiology’s advice, the psychiatrist stopped olanzapine and clonazepam, continued gabapentin, 300 mg/d, and added lorazepam, 1 mg as needed for agitation.
Within 48 hours, Mr. B’s QTc interval decreased to 400 msec. Gabapentin and lorazepam were continued. He received potassium chloride, 40 mEq qid for 4 days, and within 2 days potassium was normal (4.4 to 4.8 mEq/L). Magnesium also was monitored.
Over the next few days, Mr. B decompensated. He exposed himself, requested sexual favors from staff, and became agitated. Staff reported that he was responding to internal stimuli and had pressured speech and flight of ideas.
After consulting cardiology, the psychiatrist restarted olanzapine, 10 mg/d, and lorazepam, 1 mg bid. Daily ECGs were ordered. After two olanzapine doses, Mr. B’s QTc interval rose to 550 msec. The psychiatrist stopped all psychotropics except lorazepam, which was increased to 2 mg bid. When Mr. B became more agitated, throwing himself to the floor and hitting himself, he was isolated for his safety.
The authors’ observations
For years, olanzapine abated Mr. B’s mood and psychotic symptoms, and until the previous year significant QTc prolongation had not been detected. Other risk factors—such as electrolyte imbalance and change in olanzapine metabolism—were ruled out.
Mr. B’s chart indicated that he had responded well to haloperidol during a prior hospitalization. Divalproex, which has little effect on QTc interval, was also considered to control his mood.
CASE 2 CONTINUED: DRUG TRIALS
Eight days after Mr. B was hospitalized, the psychiatrist added divalproex, 250 mg tid titrated over 4 days to 1,000 mg/d. Mr. B became less manic but remained psychotic and disorganized. Lorazepam was increased to 2 mg tid and 3 mg at bedtime. His QTc interval now averaged 400 msec.
Loxapine, 10 mg tid, was added but then quickly discontinued after Mr. B’s QTc interval approached 500 msec.
Table 2
QTc interval ranges in men and women
| Range | Men (msec) | Women (msec) |
|---|---|---|
| Normal | <430 | <450 |
| Borderline | 431-450 | 451-470 |
| Source: reference 8. | ||
The following week, after consulting cardiology, the psychiatrist started haloperidol, 2 mg tid, and added benztropine, 1 mg for dystonia as needed. The next day, Mr. B’s QTc interval was 402 msec.
Medications were readjusted gradually. Gabapentin was restarted and increased to 600 mg tid, lorazepam was decreased to 1 mg tid, and divalproex was increased to 500 mg tid with no major QTc change.
Haloperidol was titrated to 5 mg bid, but the interval increased to 549 msec, then fell below 500 msec after haloperidol was readjusted to 2 mg bid.
Over the next 2 weeks, Mr. B’s mood and psychotic symptoms gradually improved. He was discharged after 27 days, at which point his QTc interval ranged between 360 and 409 msec. He was told to continue his medications.
The authors’ observations
Many factors other than antipsychotic use can lengthen QTc interval. Patients with major psychiatric disorders tend to have more risk factors compared with the general population.4
Serial or signal-averaged ECGs are the most accurate ways to monitor QTc intervals.5 Obtain a baseline ECG before starting an antipsychotic for patients with one or more risk factors:
Age >65. Older persons without coronary artery disease (CAD) have longer QTc intervals than do younger patients in similar health.6
Drug-drug interactions—common among the elderly—can further prolong the interval. Decreased drug metabolism also raises drug plasma levels and increases QTc prolongation risk.
Cardiac diseases. CAD, cardiac arrhythmias, and congestive heart failure are serious risk factors, particularly for older patients. Watch for pre-existing heart disease—which heightens risk of conduction defects—and family history of cardiac disease, syncope, or sudden death.
CNS diseases. Stroke, tumors, and brain infections can cause autonomic dysfunction and electrolyte imbalances.
Electrolyte imbalance. Hypokalemia and hypomagnesemia can prolong the interval.7 Take complaints of diarrhea or frequent vomiting seriously, and refer patients with renal disease or who are using diuretics for an ECG. Regularly test for electrolytes, especially potassium and magnesium.
Endocrine diseases. Diabetes, hypothyroidism, and pituitary insufficiency can cause electrolyte abnormalities.
Female sex. QTc intervals are on average 20 msec longer in women <age>Table 2)8 and are prolonged during the first half of the menstrual cycle. Androgen may shorten intervals in men. Women account for about 70% of drug-induced torsade de pointes cases.9
</age>
Medications. Antipsychotics, tricyclics, and antihistamines can prolong the interval alone or when combined with drugs that inhibit their metabolism. Concomitant use of agents that inhibit cytochrome P-450 enzyme systems may elevate serum concentrations of the interval-prolonging medication,4 as can decreased CYP 2D6 activity.10 Check plasma drug levels in patients who exhibit side effects.
Also check for congenital long QTc interval, autonomic CNS abnormalities, and overdose of a prescribed psychotropic.
Check ECGs every 2 days for inpatients and at every visit for outpatients taking antipsychotics. Frequent testing is crucial for elderly patients with multiple cardiac risk factors who are taking medications likely to increase the interval. Repeat ECGs if the patient reports lightheadedness or palpitations.
QTc interval prolongation is minimal in healthy young adults taking antipsychotics, so order ECGs only when symptoms arise. A baseline ECG is advisable but not necessary.
Order a cardiology consult and immediate ECG when the QTc interval exceeds 500 msec11 or if the patient exhibits arrhythmia symptoms (palpitation chest pain, dizziness, presyncope, syncope). Work with the cardiologist to manage medication.
Related resources
- University of Arizona Center for Education and Research on Therapeutics. Drugs that prolong the QT interval. http://www.qtdrugs.org/medical-pros/drug-lists/drug-lists.htm
- Glassman AH, Bigger JT Jr. Antipsychotic drugs: prolonged QTc interval, torsade de pointes, and sudden death. Am J Psychiatry 2001;158:1774-82.
Drug brand names
- Atorvastatin • Lipitor
- Benztropine • Cogentin
- Citalopram • Celexa
- Clonazepam • Klonopin
- Divalproex • Depakote
- Donepezil • Aricept
- Esomeprazole • Nexium
- Gabapentin • Neurontin
- Haloperidol • Haldol
- Hydrochlorothiazide • Atacand, others
- Lorazepam • Ativan
- Loxapine • Loxitane
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Thioridazine • Mellaril
- Ziprasidone •Geodon
Disclosure
Dr. Tampi receives research support from the division of state, community, and public health, bureau of health professions, Health Resources and Services Administration, Department of Health and Human Services.
Dr. Ruedrich receives grants from Pfizer Inc. and Eisai Inc., and is a consultant to Abbott Laboratories.
The other authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Reilly JG, Ayis SA, Ferrier IN, et al. QTc-interval abnormalities and psychotropic drug therapy in psychiatric patients. Lancet. 2000;355:1048-52.
2. U.S. Food and Drug Administration. Center for Drug Evaluation and Research, Psychopharmacological Drugs Advisory Committee. Meeting transcript for approval of Zeldox (ziprasidone), July 19, 2000. Available at: http://www.fda.gov/ohrms/docket/ac/00/transcripts/3619tla.pdf, 3619tlb.pdf. and 3619tlc.pdf.
3. Jeste DV, Caligiuri MP, Paulsen JS, et al. Risk of tardive dyskinesia in older patients. A prospective longitudinal study of 266 outpatients. Arch Gen Psychiatry. 1995;52:756-65.
4. Fayek M, Kingsbury SJ, Zada J, Simpson GM. Psychopharmacology: cardiac effects of antipsychotic medication. Psychiatr Serv. 2001;52:607-9.
5. Baker B, Dorian P, Sandor C, et al. Electrocardiographic effects of fluoxetine and doxepine in patients with major depression. J Clin Psychopharmacol. 1997;17:15-21.
6. Khan SP, Dhalvani S, Vieweg WVR, et al. Electrocardiographic QT interval in geropsychiatric inpatient population: a preliminary study. Med Psychiatr. 1998;1:71-4.
7. Crompton SJ, Lux RL, Ramsey MR, et al. Genetically defined therapy of inherited long-QT syndrome: correction of abnormal repolarization by potassium. Circulation. 1996;94:1018-22.
8. Piepho RW. Cardiovascular effects of antipsychotics used in bipolar illness. J Clin Psychiatry. 2002;63[suppl 4]:20-3.
9. Drici MD, Clement N. Is gender a risk factor for adverse drug reaction? The example of drug-induced long QT syndrome. Drug Saf. 2001;24(8):575-85
10. Francis PD. Effects of psychotropic medications on the pediatric electrocardiogram and recommendations for monitoring. Curr Opin Ped. 2002;14:224-30.
11. Bednar MM, Harrigan EP, Anziano RJ, et al. The QT interval. Prog Cardiovasc Dis. 2001;43:1-45.
CASE 1: BEWARE ‘OLD MAN KIPLING’
Mrs. A, age 87, has Alzheimer’s disease. About 1 month before presentation, she entered a nursing home because of increasing agitation, paranoia, auditory and visual hallucinations, and decreased ability to care for herself. Her doctor started risperidone, 0.5 mg bid, to treat her agitation and psychosis.
Two days later, Mrs. A barricaded herself in her room. She told staff that “Old Man Kipling” was trying to break in, steal her money, and kill her and her son. She was sent to the emergency room; psychiatric consultation was ordered.
Mrs. A also has hypertension, renal cell carcinoma, anemia, and chronic renal failure. She had seen a psychiatrist for worsening cognitive function but has no other psychiatric history. Brain CT without contrast revealed generalized atrophy with no acute cerebral events. Workup showed decreased potassium (3.1 mEq/L), which returned to normal after Mrs. A was given potassium chloride, 20 mEq/d for 5 days. Other lab results were normal. Hydrochlorothiazide, 25 mg/d for hypertension, was stopped to prevent potassium depletion. No neurologic deficits were found.
Upon admission to the geriatric psychiatry unit, Mrs. A was paranoid and agitated. She talked to an imaginary person, continued to fear “Old Man Kipling,” and again tried to barricade herself.
ECG at admission—done because of Mrs. A’s age, cardiac history, and hydrochlorothiazide use—showed a corrected QT (QTc) interval of 494 msec, nearly 50 msec above the high-normal range for women. The interval was 460 msec at baseline (before risperidone treatment). Mrs. A was switched to olanzapine, 5 mg at bedtime, but her QTc intervals stayed between 494 and 495 msec, and her psychotic symptoms continued unabated.
Table 1
Mean antipsychotic-induced QTc interval change from baseline to steady state
| Antipsychotic | Mean QTc interval change |
|---|---|
| Haloperidol | 4.7 msec |
| Olanzapine | 6.4 msec |
| Risperidone | 10.0 msec |
| Quetiapine | 14.5 msec |
| Ziprasidone | 20.6 msec |
| Thioridazine | 35.8 msec |
| Source: reference 2. | |
The authors’ observations
Antipsychotics, used to treat behavioral disturbances in older patients, can prolong QTc intervals. Although often asymptomatic, a prolonged interval can lead to torsade de pointes, a polymorphic ventricular arrhythmia that can progress to ventricular fibrillation and cause sudden death.
Reilly et al1 suggest that antipsychotic-induced QTc prolongation may be dose-dependent. Age >65 is also a risk factor.
Start low and go slow when prescribing antipsychotics to patients with QTc intervals 450 msec. If prolonged intervals persist, switch antipsychotics and consult a cardiologist to help manage the patient’s care.
Switching agents will not entirely eliminate the risk, however. Mrs. A’s QTc interval remained elevated despite the switch to olanzapine, which is less likely than most antipsychotics to increase the interval.
Among mostly healthy men, haloperidol was shown to cause a lower mean QTc interval increase than other antipsychotics (Table 1), although QTc prolongations >60 msec were reported in 4% of those who took haloperidol.2 The agent also may cause tardive dyskinesia, and that risk is multiplied in patients >age 65.3 For Mrs. A, however, persistent psychosis and declining function outweighed the risks.
With haloperidol, start low and titrate slowly to reduce the risk of extrapyramidal symptoms (EPS). Decrease the dosage if involuntary movements develop. If a haloperidol decrease would lead to decompensation, add an anticholinergic agent such as benztropine, but be careful because anticholinergics can worsen cognitive function.
Test for involuntary movements before starting an antipsychotic. Retest every 4 to 6 months, when changing dosages or switching antipsychotics, and when patients complain of EPS.
CASE 1 CONTINUED: GOODBYE MR. KIPLING
Mrs. A was switched to haloperidol, 0.5 mg bid titrated over 3 weeks to 2 mg every morning and 3 mg nightly. Daily ECGs across 10 days showed QTc intervals 467 msec. Abnormal Involuntary Movement Scale testing showed no EPS. Her blood pressure was stable, ranging from 110 to 130 mm Hg (systolic) and 70 to 80 mm Hg (diastolic).
The patient became calmer and her paranoid delusions and hallucinations disappeared. Her Folstein Mini-Mental Status Examination score during her third and final week of hospitalization was 16, indicating moderate dementia. She was discharged to her son’s care; outpatient psychiatric care was also arranged. The psychiatrist started donepezil, 5 mg/d titrated to 10 mg/d after 6 weeks, to treat her memory impairments.
More than 1 year later, Mrs. A lives at home with her son. She has not needed psychiatric hospitalization. Her primary care physician monitors her cardiac health.
CASE 2: SUICIDALITY AND SEXUAL BEHAVIOR
Mr. B, age 50, has battled schizoaffective disorder for more than 30 years. Upon presenting to the ER, he told clinicians he planned to jump from his seventh-floor apartment after arguing with his neighbor.
The patient had been taking gabapentin, 300 mg bid; olanzapine, 10 mg at bedtime; citalopram, 20 mg/d; clonazepam, 1 mg at bedtime for panic symptoms; atorvastatin, 10 mg/d for hyperlipidemia; and esomeprazole, 40 mg/d, for ongoing GI problems. He also has bradycardia.
Electrolyte and magnesium levels, thyroid function, and liver function tests were normal. Potassium was 3.9 mEq/L, indicating possible deficiency. Toxicity screen was negative, ruling out substance abuse or medication overdose. Baseline ECG—ordered because of Mr. B’s bradycardia—showed a QTc interval of 519 msec (almost 80 msec above high-normal for men) and a heart rate of 50 bpm.
The cardiology team found that 1 year before, while being examined for suspected syncope, Mr. B had a prolonged QTc interval that resolved after olanzapine was stopped. Acting on cardiology’s advice, the psychiatrist stopped olanzapine and clonazepam, continued gabapentin, 300 mg/d, and added lorazepam, 1 mg as needed for agitation.
Within 48 hours, Mr. B’s QTc interval decreased to 400 msec. Gabapentin and lorazepam were continued. He received potassium chloride, 40 mEq qid for 4 days, and within 2 days potassium was normal (4.4 to 4.8 mEq/L). Magnesium also was monitored.
Over the next few days, Mr. B decompensated. He exposed himself, requested sexual favors from staff, and became agitated. Staff reported that he was responding to internal stimuli and had pressured speech and flight of ideas.
After consulting cardiology, the psychiatrist restarted olanzapine, 10 mg/d, and lorazepam, 1 mg bid. Daily ECGs were ordered. After two olanzapine doses, Mr. B’s QTc interval rose to 550 msec. The psychiatrist stopped all psychotropics except lorazepam, which was increased to 2 mg bid. When Mr. B became more agitated, throwing himself to the floor and hitting himself, he was isolated for his safety.
The authors’ observations
For years, olanzapine abated Mr. B’s mood and psychotic symptoms, and until the previous year significant QTc prolongation had not been detected. Other risk factors—such as electrolyte imbalance and change in olanzapine metabolism—were ruled out.
Mr. B’s chart indicated that he had responded well to haloperidol during a prior hospitalization. Divalproex, which has little effect on QTc interval, was also considered to control his mood.
CASE 2 CONTINUED: DRUG TRIALS
Eight days after Mr. B was hospitalized, the psychiatrist added divalproex, 250 mg tid titrated over 4 days to 1,000 mg/d. Mr. B became less manic but remained psychotic and disorganized. Lorazepam was increased to 2 mg tid and 3 mg at bedtime. His QTc interval now averaged 400 msec.
Loxapine, 10 mg tid, was added but then quickly discontinued after Mr. B’s QTc interval approached 500 msec.
Table 2
QTc interval ranges in men and women
| Range | Men (msec) | Women (msec) |
|---|---|---|
| Normal | <430 | <450 |
| Borderline | 431-450 | 451-470 |
| Source: reference 8. | ||
The following week, after consulting cardiology, the psychiatrist started haloperidol, 2 mg tid, and added benztropine, 1 mg for dystonia as needed. The next day, Mr. B’s QTc interval was 402 msec.
Medications were readjusted gradually. Gabapentin was restarted and increased to 600 mg tid, lorazepam was decreased to 1 mg tid, and divalproex was increased to 500 mg tid with no major QTc change.
Haloperidol was titrated to 5 mg bid, but the interval increased to 549 msec, then fell below 500 msec after haloperidol was readjusted to 2 mg bid.
Over the next 2 weeks, Mr. B’s mood and psychotic symptoms gradually improved. He was discharged after 27 days, at which point his QTc interval ranged between 360 and 409 msec. He was told to continue his medications.
The authors’ observations
Many factors other than antipsychotic use can lengthen QTc interval. Patients with major psychiatric disorders tend to have more risk factors compared with the general population.4
Serial or signal-averaged ECGs are the most accurate ways to monitor QTc intervals.5 Obtain a baseline ECG before starting an antipsychotic for patients with one or more risk factors:
Age >65. Older persons without coronary artery disease (CAD) have longer QTc intervals than do younger patients in similar health.6
Drug-drug interactions—common among the elderly—can further prolong the interval. Decreased drug metabolism also raises drug plasma levels and increases QTc prolongation risk.
Cardiac diseases. CAD, cardiac arrhythmias, and congestive heart failure are serious risk factors, particularly for older patients. Watch for pre-existing heart disease—which heightens risk of conduction defects—and family history of cardiac disease, syncope, or sudden death.
CNS diseases. Stroke, tumors, and brain infections can cause autonomic dysfunction and electrolyte imbalances.
Electrolyte imbalance. Hypokalemia and hypomagnesemia can prolong the interval.7 Take complaints of diarrhea or frequent vomiting seriously, and refer patients with renal disease or who are using diuretics for an ECG. Regularly test for electrolytes, especially potassium and magnesium.
Endocrine diseases. Diabetes, hypothyroidism, and pituitary insufficiency can cause electrolyte abnormalities.
Female sex. QTc intervals are on average 20 msec longer in women <age>Table 2)8 and are prolonged during the first half of the menstrual cycle. Androgen may shorten intervals in men. Women account for about 70% of drug-induced torsade de pointes cases.9
</age>
Medications. Antipsychotics, tricyclics, and antihistamines can prolong the interval alone or when combined with drugs that inhibit their metabolism. Concomitant use of agents that inhibit cytochrome P-450 enzyme systems may elevate serum concentrations of the interval-prolonging medication,4 as can decreased CYP 2D6 activity.10 Check plasma drug levels in patients who exhibit side effects.
Also check for congenital long QTc interval, autonomic CNS abnormalities, and overdose of a prescribed psychotropic.
Check ECGs every 2 days for inpatients and at every visit for outpatients taking antipsychotics. Frequent testing is crucial for elderly patients with multiple cardiac risk factors who are taking medications likely to increase the interval. Repeat ECGs if the patient reports lightheadedness or palpitations.
QTc interval prolongation is minimal in healthy young adults taking antipsychotics, so order ECGs only when symptoms arise. A baseline ECG is advisable but not necessary.
Order a cardiology consult and immediate ECG when the QTc interval exceeds 500 msec11 or if the patient exhibits arrhythmia symptoms (palpitation chest pain, dizziness, presyncope, syncope). Work with the cardiologist to manage medication.
Related resources
- University of Arizona Center for Education and Research on Therapeutics. Drugs that prolong the QT interval. http://www.qtdrugs.org/medical-pros/drug-lists/drug-lists.htm
- Glassman AH, Bigger JT Jr. Antipsychotic drugs: prolonged QTc interval, torsade de pointes, and sudden death. Am J Psychiatry 2001;158:1774-82.
Drug brand names
- Atorvastatin • Lipitor
- Benztropine • Cogentin
- Citalopram • Celexa
- Clonazepam • Klonopin
- Divalproex • Depakote
- Donepezil • Aricept
- Esomeprazole • Nexium
- Gabapentin • Neurontin
- Haloperidol • Haldol
- Hydrochlorothiazide • Atacand, others
- Lorazepam • Ativan
- Loxapine • Loxitane
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Thioridazine • Mellaril
- Ziprasidone •Geodon
Disclosure
Dr. Tampi receives research support from the division of state, community, and public health, bureau of health professions, Health Resources and Services Administration, Department of Health and Human Services.
Dr. Ruedrich receives grants from Pfizer Inc. and Eisai Inc., and is a consultant to Abbott Laboratories.
The other authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
CASE 1: BEWARE ‘OLD MAN KIPLING’
Mrs. A, age 87, has Alzheimer’s disease. About 1 month before presentation, she entered a nursing home because of increasing agitation, paranoia, auditory and visual hallucinations, and decreased ability to care for herself. Her doctor started risperidone, 0.5 mg bid, to treat her agitation and psychosis.
Two days later, Mrs. A barricaded herself in her room. She told staff that “Old Man Kipling” was trying to break in, steal her money, and kill her and her son. She was sent to the emergency room; psychiatric consultation was ordered.
Mrs. A also has hypertension, renal cell carcinoma, anemia, and chronic renal failure. She had seen a psychiatrist for worsening cognitive function but has no other psychiatric history. Brain CT without contrast revealed generalized atrophy with no acute cerebral events. Workup showed decreased potassium (3.1 mEq/L), which returned to normal after Mrs. A was given potassium chloride, 20 mEq/d for 5 days. Other lab results were normal. Hydrochlorothiazide, 25 mg/d for hypertension, was stopped to prevent potassium depletion. No neurologic deficits were found.
Upon admission to the geriatric psychiatry unit, Mrs. A was paranoid and agitated. She talked to an imaginary person, continued to fear “Old Man Kipling,” and again tried to barricade herself.
ECG at admission—done because of Mrs. A’s age, cardiac history, and hydrochlorothiazide use—showed a corrected QT (QTc) interval of 494 msec, nearly 50 msec above the high-normal range for women. The interval was 460 msec at baseline (before risperidone treatment). Mrs. A was switched to olanzapine, 5 mg at bedtime, but her QTc intervals stayed between 494 and 495 msec, and her psychotic symptoms continued unabated.
Table 1
Mean antipsychotic-induced QTc interval change from baseline to steady state
| Antipsychotic | Mean QTc interval change |
|---|---|
| Haloperidol | 4.7 msec |
| Olanzapine | 6.4 msec |
| Risperidone | 10.0 msec |
| Quetiapine | 14.5 msec |
| Ziprasidone | 20.6 msec |
| Thioridazine | 35.8 msec |
| Source: reference 2. | |
The authors’ observations
Antipsychotics, used to treat behavioral disturbances in older patients, can prolong QTc intervals. Although often asymptomatic, a prolonged interval can lead to torsade de pointes, a polymorphic ventricular arrhythmia that can progress to ventricular fibrillation and cause sudden death.
Reilly et al1 suggest that antipsychotic-induced QTc prolongation may be dose-dependent. Age >65 is also a risk factor.
Start low and go slow when prescribing antipsychotics to patients with QTc intervals 450 msec. If prolonged intervals persist, switch antipsychotics and consult a cardiologist to help manage the patient’s care.
Switching agents will not entirely eliminate the risk, however. Mrs. A’s QTc interval remained elevated despite the switch to olanzapine, which is less likely than most antipsychotics to increase the interval.
Among mostly healthy men, haloperidol was shown to cause a lower mean QTc interval increase than other antipsychotics (Table 1), although QTc prolongations >60 msec were reported in 4% of those who took haloperidol.2 The agent also may cause tardive dyskinesia, and that risk is multiplied in patients >age 65.3 For Mrs. A, however, persistent psychosis and declining function outweighed the risks.
With haloperidol, start low and titrate slowly to reduce the risk of extrapyramidal symptoms (EPS). Decrease the dosage if involuntary movements develop. If a haloperidol decrease would lead to decompensation, add an anticholinergic agent such as benztropine, but be careful because anticholinergics can worsen cognitive function.
Test for involuntary movements before starting an antipsychotic. Retest every 4 to 6 months, when changing dosages or switching antipsychotics, and when patients complain of EPS.
CASE 1 CONTINUED: GOODBYE MR. KIPLING
Mrs. A was switched to haloperidol, 0.5 mg bid titrated over 3 weeks to 2 mg every morning and 3 mg nightly. Daily ECGs across 10 days showed QTc intervals 467 msec. Abnormal Involuntary Movement Scale testing showed no EPS. Her blood pressure was stable, ranging from 110 to 130 mm Hg (systolic) and 70 to 80 mm Hg (diastolic).
The patient became calmer and her paranoid delusions and hallucinations disappeared. Her Folstein Mini-Mental Status Examination score during her third and final week of hospitalization was 16, indicating moderate dementia. She was discharged to her son’s care; outpatient psychiatric care was also arranged. The psychiatrist started donepezil, 5 mg/d titrated to 10 mg/d after 6 weeks, to treat her memory impairments.
More than 1 year later, Mrs. A lives at home with her son. She has not needed psychiatric hospitalization. Her primary care physician monitors her cardiac health.
CASE 2: SUICIDALITY AND SEXUAL BEHAVIOR
Mr. B, age 50, has battled schizoaffective disorder for more than 30 years. Upon presenting to the ER, he told clinicians he planned to jump from his seventh-floor apartment after arguing with his neighbor.
The patient had been taking gabapentin, 300 mg bid; olanzapine, 10 mg at bedtime; citalopram, 20 mg/d; clonazepam, 1 mg at bedtime for panic symptoms; atorvastatin, 10 mg/d for hyperlipidemia; and esomeprazole, 40 mg/d, for ongoing GI problems. He also has bradycardia.
Electrolyte and magnesium levels, thyroid function, and liver function tests were normal. Potassium was 3.9 mEq/L, indicating possible deficiency. Toxicity screen was negative, ruling out substance abuse or medication overdose. Baseline ECG—ordered because of Mr. B’s bradycardia—showed a QTc interval of 519 msec (almost 80 msec above high-normal for men) and a heart rate of 50 bpm.
The cardiology team found that 1 year before, while being examined for suspected syncope, Mr. B had a prolonged QTc interval that resolved after olanzapine was stopped. Acting on cardiology’s advice, the psychiatrist stopped olanzapine and clonazepam, continued gabapentin, 300 mg/d, and added lorazepam, 1 mg as needed for agitation.
Within 48 hours, Mr. B’s QTc interval decreased to 400 msec. Gabapentin and lorazepam were continued. He received potassium chloride, 40 mEq qid for 4 days, and within 2 days potassium was normal (4.4 to 4.8 mEq/L). Magnesium also was monitored.
Over the next few days, Mr. B decompensated. He exposed himself, requested sexual favors from staff, and became agitated. Staff reported that he was responding to internal stimuli and had pressured speech and flight of ideas.
After consulting cardiology, the psychiatrist restarted olanzapine, 10 mg/d, and lorazepam, 1 mg bid. Daily ECGs were ordered. After two olanzapine doses, Mr. B’s QTc interval rose to 550 msec. The psychiatrist stopped all psychotropics except lorazepam, which was increased to 2 mg bid. When Mr. B became more agitated, throwing himself to the floor and hitting himself, he was isolated for his safety.
The authors’ observations
For years, olanzapine abated Mr. B’s mood and psychotic symptoms, and until the previous year significant QTc prolongation had not been detected. Other risk factors—such as electrolyte imbalance and change in olanzapine metabolism—were ruled out.
Mr. B’s chart indicated that he had responded well to haloperidol during a prior hospitalization. Divalproex, which has little effect on QTc interval, was also considered to control his mood.
CASE 2 CONTINUED: DRUG TRIALS
Eight days after Mr. B was hospitalized, the psychiatrist added divalproex, 250 mg tid titrated over 4 days to 1,000 mg/d. Mr. B became less manic but remained psychotic and disorganized. Lorazepam was increased to 2 mg tid and 3 mg at bedtime. His QTc interval now averaged 400 msec.
Loxapine, 10 mg tid, was added but then quickly discontinued after Mr. B’s QTc interval approached 500 msec.
Table 2
QTc interval ranges in men and women
| Range | Men (msec) | Women (msec) |
|---|---|---|
| Normal | <430 | <450 |
| Borderline | 431-450 | 451-470 |
| Source: reference 8. | ||
The following week, after consulting cardiology, the psychiatrist started haloperidol, 2 mg tid, and added benztropine, 1 mg for dystonia as needed. The next day, Mr. B’s QTc interval was 402 msec.
Medications were readjusted gradually. Gabapentin was restarted and increased to 600 mg tid, lorazepam was decreased to 1 mg tid, and divalproex was increased to 500 mg tid with no major QTc change.
Haloperidol was titrated to 5 mg bid, but the interval increased to 549 msec, then fell below 500 msec after haloperidol was readjusted to 2 mg bid.
Over the next 2 weeks, Mr. B’s mood and psychotic symptoms gradually improved. He was discharged after 27 days, at which point his QTc interval ranged between 360 and 409 msec. He was told to continue his medications.
The authors’ observations
Many factors other than antipsychotic use can lengthen QTc interval. Patients with major psychiatric disorders tend to have more risk factors compared with the general population.4
Serial or signal-averaged ECGs are the most accurate ways to monitor QTc intervals.5 Obtain a baseline ECG before starting an antipsychotic for patients with one or more risk factors:
Age >65. Older persons without coronary artery disease (CAD) have longer QTc intervals than do younger patients in similar health.6
Drug-drug interactions—common among the elderly—can further prolong the interval. Decreased drug metabolism also raises drug plasma levels and increases QTc prolongation risk.
Cardiac diseases. CAD, cardiac arrhythmias, and congestive heart failure are serious risk factors, particularly for older patients. Watch for pre-existing heart disease—which heightens risk of conduction defects—and family history of cardiac disease, syncope, or sudden death.
CNS diseases. Stroke, tumors, and brain infections can cause autonomic dysfunction and electrolyte imbalances.
Electrolyte imbalance. Hypokalemia and hypomagnesemia can prolong the interval.7 Take complaints of diarrhea or frequent vomiting seriously, and refer patients with renal disease or who are using diuretics for an ECG. Regularly test for electrolytes, especially potassium and magnesium.
Endocrine diseases. Diabetes, hypothyroidism, and pituitary insufficiency can cause electrolyte abnormalities.
Female sex. QTc intervals are on average 20 msec longer in women <age>Table 2)8 and are prolonged during the first half of the menstrual cycle. Androgen may shorten intervals in men. Women account for about 70% of drug-induced torsade de pointes cases.9
</age>
Medications. Antipsychotics, tricyclics, and antihistamines can prolong the interval alone or when combined with drugs that inhibit their metabolism. Concomitant use of agents that inhibit cytochrome P-450 enzyme systems may elevate serum concentrations of the interval-prolonging medication,4 as can decreased CYP 2D6 activity.10 Check plasma drug levels in patients who exhibit side effects.
Also check for congenital long QTc interval, autonomic CNS abnormalities, and overdose of a prescribed psychotropic.
Check ECGs every 2 days for inpatients and at every visit for outpatients taking antipsychotics. Frequent testing is crucial for elderly patients with multiple cardiac risk factors who are taking medications likely to increase the interval. Repeat ECGs if the patient reports lightheadedness or palpitations.
QTc interval prolongation is minimal in healthy young adults taking antipsychotics, so order ECGs only when symptoms arise. A baseline ECG is advisable but not necessary.
Order a cardiology consult and immediate ECG when the QTc interval exceeds 500 msec11 or if the patient exhibits arrhythmia symptoms (palpitation chest pain, dizziness, presyncope, syncope). Work with the cardiologist to manage medication.
Related resources
- University of Arizona Center for Education and Research on Therapeutics. Drugs that prolong the QT interval. http://www.qtdrugs.org/medical-pros/drug-lists/drug-lists.htm
- Glassman AH, Bigger JT Jr. Antipsychotic drugs: prolonged QTc interval, torsade de pointes, and sudden death. Am J Psychiatry 2001;158:1774-82.
Drug brand names
- Atorvastatin • Lipitor
- Benztropine • Cogentin
- Citalopram • Celexa
- Clonazepam • Klonopin
- Divalproex • Depakote
- Donepezil • Aricept
- Esomeprazole • Nexium
- Gabapentin • Neurontin
- Haloperidol • Haldol
- Hydrochlorothiazide • Atacand, others
- Lorazepam • Ativan
- Loxapine • Loxitane
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Thioridazine • Mellaril
- Ziprasidone •Geodon
Disclosure
Dr. Tampi receives research support from the division of state, community, and public health, bureau of health professions, Health Resources and Services Administration, Department of Health and Human Services.
Dr. Ruedrich receives grants from Pfizer Inc. and Eisai Inc., and is a consultant to Abbott Laboratories.
The other authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Reilly JG, Ayis SA, Ferrier IN, et al. QTc-interval abnormalities and psychotropic drug therapy in psychiatric patients. Lancet. 2000;355:1048-52.
2. U.S. Food and Drug Administration. Center for Drug Evaluation and Research, Psychopharmacological Drugs Advisory Committee. Meeting transcript for approval of Zeldox (ziprasidone), July 19, 2000. Available at: http://www.fda.gov/ohrms/docket/ac/00/transcripts/3619tla.pdf, 3619tlb.pdf. and 3619tlc.pdf.
3. Jeste DV, Caligiuri MP, Paulsen JS, et al. Risk of tardive dyskinesia in older patients. A prospective longitudinal study of 266 outpatients. Arch Gen Psychiatry. 1995;52:756-65.
4. Fayek M, Kingsbury SJ, Zada J, Simpson GM. Psychopharmacology: cardiac effects of antipsychotic medication. Psychiatr Serv. 2001;52:607-9.
5. Baker B, Dorian P, Sandor C, et al. Electrocardiographic effects of fluoxetine and doxepine in patients with major depression. J Clin Psychopharmacol. 1997;17:15-21.
6. Khan SP, Dhalvani S, Vieweg WVR, et al. Electrocardiographic QT interval in geropsychiatric inpatient population: a preliminary study. Med Psychiatr. 1998;1:71-4.
7. Crompton SJ, Lux RL, Ramsey MR, et al. Genetically defined therapy of inherited long-QT syndrome: correction of abnormal repolarization by potassium. Circulation. 1996;94:1018-22.
8. Piepho RW. Cardiovascular effects of antipsychotics used in bipolar illness. J Clin Psychiatry. 2002;63[suppl 4]:20-3.
9. Drici MD, Clement N. Is gender a risk factor for adverse drug reaction? The example of drug-induced long QT syndrome. Drug Saf. 2001;24(8):575-85
10. Francis PD. Effects of psychotropic medications on the pediatric electrocardiogram and recommendations for monitoring. Curr Opin Ped. 2002;14:224-30.
11. Bednar MM, Harrigan EP, Anziano RJ, et al. The QT interval. Prog Cardiovasc Dis. 2001;43:1-45.
1. Reilly JG, Ayis SA, Ferrier IN, et al. QTc-interval abnormalities and psychotropic drug therapy in psychiatric patients. Lancet. 2000;355:1048-52.
2. U.S. Food and Drug Administration. Center for Drug Evaluation and Research, Psychopharmacological Drugs Advisory Committee. Meeting transcript for approval of Zeldox (ziprasidone), July 19, 2000. Available at: http://www.fda.gov/ohrms/docket/ac/00/transcripts/3619tla.pdf, 3619tlb.pdf. and 3619tlc.pdf.
3. Jeste DV, Caligiuri MP, Paulsen JS, et al. Risk of tardive dyskinesia in older patients. A prospective longitudinal study of 266 outpatients. Arch Gen Psychiatry. 1995;52:756-65.
4. Fayek M, Kingsbury SJ, Zada J, Simpson GM. Psychopharmacology: cardiac effects of antipsychotic medication. Psychiatr Serv. 2001;52:607-9.
5. Baker B, Dorian P, Sandor C, et al. Electrocardiographic effects of fluoxetine and doxepine in patients with major depression. J Clin Psychopharmacol. 1997;17:15-21.
6. Khan SP, Dhalvani S, Vieweg WVR, et al. Electrocardiographic QT interval in geropsychiatric inpatient population: a preliminary study. Med Psychiatr. 1998;1:71-4.
7. Crompton SJ, Lux RL, Ramsey MR, et al. Genetically defined therapy of inherited long-QT syndrome: correction of abnormal repolarization by potassium. Circulation. 1996;94:1018-22.
8. Piepho RW. Cardiovascular effects of antipsychotics used in bipolar illness. J Clin Psychiatry. 2002;63[suppl 4]:20-3.
9. Drici MD, Clement N. Is gender a risk factor for adverse drug reaction? The example of drug-induced long QT syndrome. Drug Saf. 2001;24(8):575-85
10. Francis PD. Effects of psychotropic medications on the pediatric electrocardiogram and recommendations for monitoring. Curr Opin Ped. 2002;14:224-30.
11. Bednar MM, Harrigan EP, Anziano RJ, et al. The QT interval. Prog Cardiovasc Dis. 2001;43:1-45.
Treating depression, chronic pain
We read with interest Dr. Nelson’s and Dr. Krahn’s article on treating chronic pain and comorbid major depression (Current Psychiatry, May 2004).
We treat many patients who present with depression and chronic pain—often as a partial cause of their depression. The article’s recommendations will be most useful.
We have found that two agents—lamotrigine and mirtazapine—have been particularly helpful. The authors, however, did not mention these agents or only briefly referred to them.
Lamotrigine, although not FDA-approved for these uses, has demonstrated efficacy in unipolar depression1 and chronic pain.2 Although the medication has not been studied for treating comorbid depression and chronic pain, we can attest to its usefulness for such patients.
Mirtazapine is FDA-approved for depression and has been compared favorably with selective serotonin reuptake inhibitors3-4 or venlafaxine.5 Fewer data support using mirtazapine for chronic pain, but its sedating effects make it an option for treating any syndrome associated with sleep disturbance—ie, major depression and chronic pain.
Vida Robertson, MD
Michael S. Wilson, II, MD
Department of psychiatry
Louisiana State University Health Sciences Center
New Orleans
- Barbosa L, Berk M, Vorster M. A double-blind, randomized, placebo-controlled trial of augmentation with lamotrigine or placebo in patients concomitantly treated with fluoxetine for resistant major depressive episodes. J Clin Psychiatry 2003;64(4):403–7.
- Eisenberg E, Lurie Y, Braker C, et al. Lamotrigine reduces painful diabetic neuropathy: a randomized, controlled study. Neurology 2001;57(3):505–9.
- Winokur A. DeMartinis NA 3rd, McNally DP, et al. Comparative effects of mirtazapine and fluoxetine on sleep physiology measures in patients with major depression and insomnia. J Clin Psychiatry 2003;64(10):1224–9.
- Wade A, Crawford GM, Angus M, et al. A randomized, double-blind, 24-week study comparing the efficacy and tolerability of mirtazapine and paroxetine in depressed patients in primary care. Int Clin Psychopharmacol 2003;18(3):133–41.
- Guelfi JD, Ansseau M, Timmerman L, Korsgaard S; Mirtazapine-Venlafaxine Study Group. Mirtazapine versus venlafaxine in hospitalized severely depressed patients with melancholic features. J Clin Psychopharmacol 2001;21(4):425–31.
We read with interest Dr. Nelson’s and Dr. Krahn’s article on treating chronic pain and comorbid major depression (Current Psychiatry, May 2004).
We treat many patients who present with depression and chronic pain—often as a partial cause of their depression. The article’s recommendations will be most useful.
We have found that two agents—lamotrigine and mirtazapine—have been particularly helpful. The authors, however, did not mention these agents or only briefly referred to them.
Lamotrigine, although not FDA-approved for these uses, has demonstrated efficacy in unipolar depression1 and chronic pain.2 Although the medication has not been studied for treating comorbid depression and chronic pain, we can attest to its usefulness for such patients.
Mirtazapine is FDA-approved for depression and has been compared favorably with selective serotonin reuptake inhibitors3-4 or venlafaxine.5 Fewer data support using mirtazapine for chronic pain, but its sedating effects make it an option for treating any syndrome associated with sleep disturbance—ie, major depression and chronic pain.
Vida Robertson, MD
Michael S. Wilson, II, MD
Department of psychiatry
Louisiana State University Health Sciences Center
New Orleans
- Barbosa L, Berk M, Vorster M. A double-blind, randomized, placebo-controlled trial of augmentation with lamotrigine or placebo in patients concomitantly treated with fluoxetine for resistant major depressive episodes. J Clin Psychiatry 2003;64(4):403–7.
- Eisenberg E, Lurie Y, Braker C, et al. Lamotrigine reduces painful diabetic neuropathy: a randomized, controlled study. Neurology 2001;57(3):505–9.
- Winokur A. DeMartinis NA 3rd, McNally DP, et al. Comparative effects of mirtazapine and fluoxetine on sleep physiology measures in patients with major depression and insomnia. J Clin Psychiatry 2003;64(10):1224–9.
- Wade A, Crawford GM, Angus M, et al. A randomized, double-blind, 24-week study comparing the efficacy and tolerability of mirtazapine and paroxetine in depressed patients in primary care. Int Clin Psychopharmacol 2003;18(3):133–41.
- Guelfi JD, Ansseau M, Timmerman L, Korsgaard S; Mirtazapine-Venlafaxine Study Group. Mirtazapine versus venlafaxine in hospitalized severely depressed patients with melancholic features. J Clin Psychopharmacol 2001;21(4):425–31.
We read with interest Dr. Nelson’s and Dr. Krahn’s article on treating chronic pain and comorbid major depression (Current Psychiatry, May 2004).
We treat many patients who present with depression and chronic pain—often as a partial cause of their depression. The article’s recommendations will be most useful.
We have found that two agents—lamotrigine and mirtazapine—have been particularly helpful. The authors, however, did not mention these agents or only briefly referred to them.
Lamotrigine, although not FDA-approved for these uses, has demonstrated efficacy in unipolar depression1 and chronic pain.2 Although the medication has not been studied for treating comorbid depression and chronic pain, we can attest to its usefulness for such patients.
Mirtazapine is FDA-approved for depression and has been compared favorably with selective serotonin reuptake inhibitors3-4 or venlafaxine.5 Fewer data support using mirtazapine for chronic pain, but its sedating effects make it an option for treating any syndrome associated with sleep disturbance—ie, major depression and chronic pain.
Vida Robertson, MD
Michael S. Wilson, II, MD
Department of psychiatry
Louisiana State University Health Sciences Center
New Orleans
- Barbosa L, Berk M, Vorster M. A double-blind, randomized, placebo-controlled trial of augmentation with lamotrigine or placebo in patients concomitantly treated with fluoxetine for resistant major depressive episodes. J Clin Psychiatry 2003;64(4):403–7.
- Eisenberg E, Lurie Y, Braker C, et al. Lamotrigine reduces painful diabetic neuropathy: a randomized, controlled study. Neurology 2001;57(3):505–9.
- Winokur A. DeMartinis NA 3rd, McNally DP, et al. Comparative effects of mirtazapine and fluoxetine on sleep physiology measures in patients with major depression and insomnia. J Clin Psychiatry 2003;64(10):1224–9.
- Wade A, Crawford GM, Angus M, et al. A randomized, double-blind, 24-week study comparing the efficacy and tolerability of mirtazapine and paroxetine in depressed patients in primary care. Int Clin Psychopharmacol 2003;18(3):133–41.
- Guelfi JD, Ansseau M, Timmerman L, Korsgaard S; Mirtazapine-Venlafaxine Study Group. Mirtazapine versus venlafaxine in hospitalized severely depressed patients with melancholic features. J Clin Psychopharmacol 2001;21(4):425–31.