User login
Family environment important in early psychosis outcomes
Family environment may influence subsequent functional outcomes in patients with first-episode psychosis, new research suggests.
A study of more than 300 patients with first-episode psychosis (FEP) showed that although family environment was not associated with functioning at initial presentation, an interaction developed over time that could have “important implications for early interventions for both patients and caregivers,” investigators reported.
study coinvestigator Norma Verdolini, MD, PhD, bipolar and depressive disorders unit, hospital Clinic Barcelona, University of Barcelona, said in an interview.
The findings were scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
FAST measures
Previous research has shown that family environment influences the development of psychotic symptoms, with negative family environmental factors associated with poor prognoses.
Conversely, one study indicated that a positive family environment is linked to greater improvement in negative and disorganized symptoms in adolescents at imminent risk for psychosis onset.
However, the current investigators noted that the impact of family environment on longitudinal functioning in individuals presenting with FEP is unclear.
To investigate further, they conducted an analysis as part of the PEPs study, which included 335 patients with FEP and 253 healthy controls. Functioning was measured using the Functional Assessment Short Test (FAST), and family environmental styles were evaluated using the Family Environment Scale (FES), which assesses “emotional climate” of a family across 10 domains.
At baseline, the mean total FAST score was 27.8 in patients with FEP versus 3.5 in the healthy controls, indicating substantially worse functioning among the patients. Linear regression analysis indicated that at baseline there was no significant association between aspects of family environment on the FES and functional scores.
Patients were assessed again at 2 years, by which point 283 had been diagnosed with psychotic disorders and 52 with bipolar disorder. The mean total FAST scores were 20.98 among patients with psychotic disorders and 13.8 in those with bipolar disorder.
Family conflict
Results showed that, among those with bipolar disorder, worse functioning on FAST at 2 years was significantly associated with higher rates of open expression of conflict in the family (P = .004).
In patients with psychotic disorders, worse functioning was significantly associated with lower rates of participation in social activities (P = .006) and an achievement-oriented family environment (P = .039). Worse functioning in patients with psychotic disorders was also significantly associated with higher rates of religious practice and values (P = .003).
Dr. Verdolini noted the reason family environment does not appear to have an impact at initial FEP presentation may be that the “first kick” is given by an individual’s genetic liability for psychiatric disorders in combination with the family environment. In reality, the two are intertwined, especially when considering what it means to a family to have one member with a psychiatric disorder, which “will have an impact on the family environment.”
Dr. Verdolini added: “This is not actually the objective family environment,” but the perceived family environment.
“So maybe in the following 2 years the patient who experiences a first episode of psychosis may change their idea of the family environment itself,” she noted. She added that at her institution psychoeducation is offered to FEP patients’ families.
‘Interesting’ findings
Commenting on the study, Nicole Kozloff, MD, from the child, youth, and emerging adult program at the Centre for Addiction and Mental Health in Toronto, said one limitation of the study is that it’s not clear what care patients received – or who in the family completed the FES.
It is also important to note that “measures of association do not necessarily imply that one factor caused the other factor,” said Dr. Kozloff, who was not involved in the research. “For example, it may be that, among people with bipolar disorder, open expression of conflict in the family can lead to worse functioning, or that worse functioning can lead to more conflict in the family.”
Nevertheless, Dr. Kozloff described the finding of an emerging association between the family environment and functioning over time as “interesting.”
When young people with FEP enter treatment, “they have reached a crisis point and are functioning poorly,” she noted.
“It could be that there is less to differentiate among levels of functioning at treatment entry but, after 2 years, the individuals have separated into those who have been responsive to treatment and are functioning well, and those who continue to have functional challenges. And this is where we start to see a relationship with family environment emerge,” Dr. Kozloff said.
She also agreed with Dr. Verdolini’s take on the findings, and that family psychoeducation “can reduce relapse rates in schizophrenia and the emotional burden on the family.”
“We also know that having family involvement in care is one of the most robust predictors that young people with psychosis will remain engaged in mental health services,” she said.
Teaching families about psychosis and its treatment, about problem-solving and communication skills, and providing support to ensure that family members know how to get help in a crisis, “is a key part of comprehensive early psychosis intervention,” Dr. Kozloff said. “It is good for the patient and good for the family, and allows the clinicians to provide better care.”
Articulates clinical practice findings
Also commenting on the results, Brian O’Donoghue, MD, PhD, senior clinical research fellow at Orygen, the National Centre of Excellence in Youth Mental Health in Melbourne, described the research as important, adding that the study highlights the need for sufficient follow-up.
“It makes sense that the involvement of family over time has a strong impact upon outcome and functioning,” he said in an interview.
“These research findings articulate what we see in clinical practice, so it is good to see that it is captured,” added Dr. O’Donoghue, who was not associated with the study.
He noted that it is common for family involvement to influence outcome, especially if the family is positively involved. “It is invaluable toward their recovery. However, conversely, if there are ongoing family stressors, then this can be a trigger for relapse or lack of improvement.”
Overall, the results “really emphasize that the family needs to be involved in care.”
The Early Psychosis Prevention and Intervention Centre where Dr. O’Donoghue is a consultant psychiatrist offers a psychoeducational course “to inform families about psychosis, treatment, and how they can support their family members.”
“We also have family peer support workers and family therapists, which are essential to the service and for the young person’s recovery,” Dr. O’Donoghue said.
The investigators and Dr. O’Donoghue disclosed no relevant financial relationships. Dr. Kozloff reported receiving research funding from the CAMH Foundation, Brain & Behavior Research Foundation, Canadian Institutes of Health Research, and AFP Innovation Fund; honoraria from Humber River Hospital, the University of Calgary (Alta.), and the Canadian Consortium for Early Intervention in Psychosis; and salary support from Inner City Health Associates.
A version of this article originally appeared on Medscape.com.
Family environment may influence subsequent functional outcomes in patients with first-episode psychosis, new research suggests.
A study of more than 300 patients with first-episode psychosis (FEP) showed that although family environment was not associated with functioning at initial presentation, an interaction developed over time that could have “important implications for early interventions for both patients and caregivers,” investigators reported.
study coinvestigator Norma Verdolini, MD, PhD, bipolar and depressive disorders unit, hospital Clinic Barcelona, University of Barcelona, said in an interview.
The findings were scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
FAST measures
Previous research has shown that family environment influences the development of psychotic symptoms, with negative family environmental factors associated with poor prognoses.
Conversely, one study indicated that a positive family environment is linked to greater improvement in negative and disorganized symptoms in adolescents at imminent risk for psychosis onset.
However, the current investigators noted that the impact of family environment on longitudinal functioning in individuals presenting with FEP is unclear.
To investigate further, they conducted an analysis as part of the PEPs study, which included 335 patients with FEP and 253 healthy controls. Functioning was measured using the Functional Assessment Short Test (FAST), and family environmental styles were evaluated using the Family Environment Scale (FES), which assesses “emotional climate” of a family across 10 domains.
At baseline, the mean total FAST score was 27.8 in patients with FEP versus 3.5 in the healthy controls, indicating substantially worse functioning among the patients. Linear regression analysis indicated that at baseline there was no significant association between aspects of family environment on the FES and functional scores.
Patients were assessed again at 2 years, by which point 283 had been diagnosed with psychotic disorders and 52 with bipolar disorder. The mean total FAST scores were 20.98 among patients with psychotic disorders and 13.8 in those with bipolar disorder.
Family conflict
Results showed that, among those with bipolar disorder, worse functioning on FAST at 2 years was significantly associated with higher rates of open expression of conflict in the family (P = .004).
In patients with psychotic disorders, worse functioning was significantly associated with lower rates of participation in social activities (P = .006) and an achievement-oriented family environment (P = .039). Worse functioning in patients with psychotic disorders was also significantly associated with higher rates of religious practice and values (P = .003).
Dr. Verdolini noted the reason family environment does not appear to have an impact at initial FEP presentation may be that the “first kick” is given by an individual’s genetic liability for psychiatric disorders in combination with the family environment. In reality, the two are intertwined, especially when considering what it means to a family to have one member with a psychiatric disorder, which “will have an impact on the family environment.”
Dr. Verdolini added: “This is not actually the objective family environment,” but the perceived family environment.
“So maybe in the following 2 years the patient who experiences a first episode of psychosis may change their idea of the family environment itself,” she noted. She added that at her institution psychoeducation is offered to FEP patients’ families.
‘Interesting’ findings
Commenting on the study, Nicole Kozloff, MD, from the child, youth, and emerging adult program at the Centre for Addiction and Mental Health in Toronto, said one limitation of the study is that it’s not clear what care patients received – or who in the family completed the FES.
It is also important to note that “measures of association do not necessarily imply that one factor caused the other factor,” said Dr. Kozloff, who was not involved in the research. “For example, it may be that, among people with bipolar disorder, open expression of conflict in the family can lead to worse functioning, or that worse functioning can lead to more conflict in the family.”
Nevertheless, Dr. Kozloff described the finding of an emerging association between the family environment and functioning over time as “interesting.”
When young people with FEP enter treatment, “they have reached a crisis point and are functioning poorly,” she noted.
“It could be that there is less to differentiate among levels of functioning at treatment entry but, after 2 years, the individuals have separated into those who have been responsive to treatment and are functioning well, and those who continue to have functional challenges. And this is where we start to see a relationship with family environment emerge,” Dr. Kozloff said.
She also agreed with Dr. Verdolini’s take on the findings, and that family psychoeducation “can reduce relapse rates in schizophrenia and the emotional burden on the family.”
“We also know that having family involvement in care is one of the most robust predictors that young people with psychosis will remain engaged in mental health services,” she said.
Teaching families about psychosis and its treatment, about problem-solving and communication skills, and providing support to ensure that family members know how to get help in a crisis, “is a key part of comprehensive early psychosis intervention,” Dr. Kozloff said. “It is good for the patient and good for the family, and allows the clinicians to provide better care.”
Articulates clinical practice findings
Also commenting on the results, Brian O’Donoghue, MD, PhD, senior clinical research fellow at Orygen, the National Centre of Excellence in Youth Mental Health in Melbourne, described the research as important, adding that the study highlights the need for sufficient follow-up.
“It makes sense that the involvement of family over time has a strong impact upon outcome and functioning,” he said in an interview.
“These research findings articulate what we see in clinical practice, so it is good to see that it is captured,” added Dr. O’Donoghue, who was not associated with the study.
He noted that it is common for family involvement to influence outcome, especially if the family is positively involved. “It is invaluable toward their recovery. However, conversely, if there are ongoing family stressors, then this can be a trigger for relapse or lack of improvement.”
Overall, the results “really emphasize that the family needs to be involved in care.”
The Early Psychosis Prevention and Intervention Centre where Dr. O’Donoghue is a consultant psychiatrist offers a psychoeducational course “to inform families about psychosis, treatment, and how they can support their family members.”
“We also have family peer support workers and family therapists, which are essential to the service and for the young person’s recovery,” Dr. O’Donoghue said.
The investigators and Dr. O’Donoghue disclosed no relevant financial relationships. Dr. Kozloff reported receiving research funding from the CAMH Foundation, Brain & Behavior Research Foundation, Canadian Institutes of Health Research, and AFP Innovation Fund; honoraria from Humber River Hospital, the University of Calgary (Alta.), and the Canadian Consortium for Early Intervention in Psychosis; and salary support from Inner City Health Associates.
A version of this article originally appeared on Medscape.com.
Family environment may influence subsequent functional outcomes in patients with first-episode psychosis, new research suggests.
A study of more than 300 patients with first-episode psychosis (FEP) showed that although family environment was not associated with functioning at initial presentation, an interaction developed over time that could have “important implications for early interventions for both patients and caregivers,” investigators reported.
study coinvestigator Norma Verdolini, MD, PhD, bipolar and depressive disorders unit, hospital Clinic Barcelona, University of Barcelona, said in an interview.
The findings were scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
FAST measures
Previous research has shown that family environment influences the development of psychotic symptoms, with negative family environmental factors associated with poor prognoses.
Conversely, one study indicated that a positive family environment is linked to greater improvement in negative and disorganized symptoms in adolescents at imminent risk for psychosis onset.
However, the current investigators noted that the impact of family environment on longitudinal functioning in individuals presenting with FEP is unclear.
To investigate further, they conducted an analysis as part of the PEPs study, which included 335 patients with FEP and 253 healthy controls. Functioning was measured using the Functional Assessment Short Test (FAST), and family environmental styles were evaluated using the Family Environment Scale (FES), which assesses “emotional climate” of a family across 10 domains.
At baseline, the mean total FAST score was 27.8 in patients with FEP versus 3.5 in the healthy controls, indicating substantially worse functioning among the patients. Linear regression analysis indicated that at baseline there was no significant association between aspects of family environment on the FES and functional scores.
Patients were assessed again at 2 years, by which point 283 had been diagnosed with psychotic disorders and 52 with bipolar disorder. The mean total FAST scores were 20.98 among patients with psychotic disorders and 13.8 in those with bipolar disorder.
Family conflict
Results showed that, among those with bipolar disorder, worse functioning on FAST at 2 years was significantly associated with higher rates of open expression of conflict in the family (P = .004).
In patients with psychotic disorders, worse functioning was significantly associated with lower rates of participation in social activities (P = .006) and an achievement-oriented family environment (P = .039). Worse functioning in patients with psychotic disorders was also significantly associated with higher rates of religious practice and values (P = .003).
Dr. Verdolini noted the reason family environment does not appear to have an impact at initial FEP presentation may be that the “first kick” is given by an individual’s genetic liability for psychiatric disorders in combination with the family environment. In reality, the two are intertwined, especially when considering what it means to a family to have one member with a psychiatric disorder, which “will have an impact on the family environment.”
Dr. Verdolini added: “This is not actually the objective family environment,” but the perceived family environment.
“So maybe in the following 2 years the patient who experiences a first episode of psychosis may change their idea of the family environment itself,” she noted. She added that at her institution psychoeducation is offered to FEP patients’ families.
‘Interesting’ findings
Commenting on the study, Nicole Kozloff, MD, from the child, youth, and emerging adult program at the Centre for Addiction and Mental Health in Toronto, said one limitation of the study is that it’s not clear what care patients received – or who in the family completed the FES.
It is also important to note that “measures of association do not necessarily imply that one factor caused the other factor,” said Dr. Kozloff, who was not involved in the research. “For example, it may be that, among people with bipolar disorder, open expression of conflict in the family can lead to worse functioning, or that worse functioning can lead to more conflict in the family.”
Nevertheless, Dr. Kozloff described the finding of an emerging association between the family environment and functioning over time as “interesting.”
When young people with FEP enter treatment, “they have reached a crisis point and are functioning poorly,” she noted.
“It could be that there is less to differentiate among levels of functioning at treatment entry but, after 2 years, the individuals have separated into those who have been responsive to treatment and are functioning well, and those who continue to have functional challenges. And this is where we start to see a relationship with family environment emerge,” Dr. Kozloff said.
She also agreed with Dr. Verdolini’s take on the findings, and that family psychoeducation “can reduce relapse rates in schizophrenia and the emotional burden on the family.”
“We also know that having family involvement in care is one of the most robust predictors that young people with psychosis will remain engaged in mental health services,” she said.
Teaching families about psychosis and its treatment, about problem-solving and communication skills, and providing support to ensure that family members know how to get help in a crisis, “is a key part of comprehensive early psychosis intervention,” Dr. Kozloff said. “It is good for the patient and good for the family, and allows the clinicians to provide better care.”
Articulates clinical practice findings
Also commenting on the results, Brian O’Donoghue, MD, PhD, senior clinical research fellow at Orygen, the National Centre of Excellence in Youth Mental Health in Melbourne, described the research as important, adding that the study highlights the need for sufficient follow-up.
“It makes sense that the involvement of family over time has a strong impact upon outcome and functioning,” he said in an interview.
“These research findings articulate what we see in clinical practice, so it is good to see that it is captured,” added Dr. O’Donoghue, who was not associated with the study.
He noted that it is common for family involvement to influence outcome, especially if the family is positively involved. “It is invaluable toward their recovery. However, conversely, if there are ongoing family stressors, then this can be a trigger for relapse or lack of improvement.”
Overall, the results “really emphasize that the family needs to be involved in care.”
The Early Psychosis Prevention and Intervention Centre where Dr. O’Donoghue is a consultant psychiatrist offers a psychoeducational course “to inform families about psychosis, treatment, and how they can support their family members.”
“We also have family peer support workers and family therapists, which are essential to the service and for the young person’s recovery,” Dr. O’Donoghue said.
The investigators and Dr. O’Donoghue disclosed no relevant financial relationships. Dr. Kozloff reported receiving research funding from the CAMH Foundation, Brain & Behavior Research Foundation, Canadian Institutes of Health Research, and AFP Innovation Fund; honoraria from Humber River Hospital, the University of Calgary (Alta.), and the Canadian Consortium for Early Intervention in Psychosis; and salary support from Inner City Health Associates.
A version of this article originally appeared on Medscape.com.
FROM SIRS 2020
Community programs improve psychosis outcomes
Community-based services that tap into local environments not only reduce the duration of untreated psychosis (DUP) but also provide improved long-term outcomes for patients with first-episode psychosis (FEP), results of two new studies show.
In the first study, investigators led by Vinod Srihari, MD, director of specialized treatment early in psychosis at Yale University, New Haven, Conn., developed a program to reduce DUP to complement their first-episode service (FES).
Through a combination of mass media and social media campaigns, outreach events with local professionals, and rapid triage, the team was able to nearly halve the time from diagnosis to initiation of antipsychotic treatment.
In the second study, a team led by Delbert G. Robinson, MD, of Hofstra University, Hempstead, N.Y., conducted a 5-year follow-up of RAISE-ETP, the first U.S. randomized trial to compare a 2-year comprehensive early intervention service (EIS) with usual care.
These trial results showed that, among more than 400 FEP patients, the EIS significantly improved both symptoms and quality of life and reduced inpatient days in comparison with standard care.
The research was scheduled to be presented at the Congress of the Schizophrenia International Research Society (SIRS) 2020, but the meeting was canceled because of the coronavirus pandemic.
Norwegian model
Dr. Srihari and colleagues note that a specialized treatment early in psychosis (STEP), which delivers a specialty team–based FES, was established at their institution in 2006.
However, in a bid to reduce DUP, in 2015 they launched MindMap, a 4-year early-detection campaign based on the Scandinavian TIPS Early Detection in Psychosis Study.
Dr. Srihari said in an interview that they visited the team that developed the TIPS program in Norway “to try to understand what elements of their approach had resulted in a successful reduction of DUP.”
He pointed out that the health care system in Norway has “more reliable pathways to care, with an ability to route people in more predictable ways from primary care to secondary care, and so on.”
In the United States, “there’s no expectation that people will go through a primary care provider,” he said. He noted that patients “make their way to specialty care in many different ways.”
Dr. Srihari said, “The other change we realized we’d have to make was that, since TIPS had been completed many years back, the media environment had changed substantially.
“At the time that TIPS was done, there was no such thing as social media, whereas when we began to think about designing our campaign, we thought social media would be a very efficient and also cost-effective way to target young people.”
MindMap, which covered a 10-town catchment area that has a population of 400,000, targeted both demand- and supply-side aspects of DUP. The former focused on delays in identifying illness and help-seeking, and the latter concentrated on referral and treatment access delays.
The researchers used a combination of mass media and social media messaging, professional detailing, and the rapid triage of referrals.
The media campaign included Facebook, Twitter, and other social media platforms that allowed the team to “target individuals in a somewhat more fine-grained way than mass media allows,” Dr. Srihari said. They focused on individuals in a particular age range and geographic location.
The professional detailing encompassed mental health agencies, emergency departments, and inpatient units, as well as colleges, college counseling centers, high schools, and police departments. The team hosted meetings, “often at local restaurants, where we provided a meal and provided some general education about what our mission was,” said Dr. Srihari. The researchers followed up these meetings with more in-person visits.
“The third arm was finding ways to basically eliminate any kind of a waiting time at our front door, such as ensuring that transportation issues were circumvented, in addition to cutting through any ambivalence they might have about finally making the leap to come to the center.”
More rapid treatment
Over the course of the baseline year and the 4 years of the MindMap program, almost 1,500 individuals were assessed. Of these, approximately 200 were eligible, and almost all were enrolled.
The researchers measured DUP at two time points from the onset of psychosis – the initiation of antipsychotic treatment (DUP1) and the initiation of FES care. Across the study period, they found that DUP1 fell significantly between the pre- and postprogram assessments, from 329 days to 185 days (P = .03). By contrast, there was no change over the same period for the Prevention and Recovery in Early Psychosis FES program in Boston, which served as a comparator.
There was also a cumulative effect on DUP, with each year of the 4-year program associated with a 46-day reduction in DUP1. However, the significant reduction was restricted to the third quintile of DUP1 and was not found in the other quintiles of DUP1 or for DUP2, despite all measures showing a consistent trend for reduction over time.
Dr. Srihari acknowledges that the team was “disappointed” that DUP2 did not fall significantly in their study. He suggested, “It might take longer for agencies to change their workloads and refer patients to STEP, which is what ended up resulting in the DUP2 not dropping as quickly.”
To see whether there was indeed a time lag in changes to practice, the team conducted an analysis in which they cut out the first year from the results and analyzed only the last 3 years. Then “we do see a decline in DUP2,” he said.
The study’s full results are currently being prepared for publication, and the investigators are considering relaunching the initiative.
“The question we are having now is how to resource the campaign without the research funds and which parts of it we think we can launch sustainably so we can continue the reduction of DUP,” Dr. Srihari said.
Plans may include developing partnerships with local businesses to help fund the media costs and working with the state government to build a learning health care network, which would make it easier for mental health agencies to consult with the team on problematic cases.
“We’re trying to reduce DUP referrals on the supply side by providing this kind of learning health collaborative ... that we also think might be fiscally a more sustainable way to do this vs. what we did in MindMap,” which would be “very expensive” to implement on a statewide basis, Dr. Srihari added.
Long-term benefit
In the second study, Dr. Robinson and colleagues highlight that EISs have been implemented worldwide for FEP patients and have been associated with improved outcomes.
However, these services typically provide care for a limited period, and cross-sectional follow-up studies have identified few advantages in comparison with standard care.
To provide a more robust longitudinal assessment of the ongoing effects of an EIS, the team conducted a 5-year follow-up of the first U.S.-based, multicenter, randomized clinical trial comparing an EIS, NAVIGATE, with usual clinical care in FEP.
RAISE-ETP was conducted at 34 sites across the United States. Seventeen sites provided NAVIGATE to 223 individuals with FEP, and the remaining 17 sites provided usual care to 181 patients.
NAVIGATE, which continued for 2 years, consisted of treatments and services delivered by a coordinated team of providers. Those services included the following:
- Education on schizophrenia and its treatment for patients and their families.
- Symptom and relapse prevention medication, using a computerized decision support system.
- Strategies for illness management building personal resilience.
- A supported employment/education model.
Patients were assessed every 6 months for up to 60 months via a video link using the Heinrichs-Carpenter Quality of Life Scale (QLS) and the Positive and Negative Syndrome Scale (PANSS).
The average age of the participants was 23 years; 78% of those who received NAVIGATE and 66% of those who received usual care were male. The opportunity for each participant to engage in NAVIGATE treatment lasted an average of 33.8 months. The longest was 44.4 months.
Over 5 years, NAVIGATE was associated with a significant improvement over usual care in QLS scores by an average of 13.14 units (P < .001). PANSS scores improved by an average of 7.73 units (P < .002). QLS scores were not affected either by the length of opportunity to participate in NAVIGATE or by DUP, the team reports. Patients who received NAVIGATE also had an average of 2.5 fewer inpatient days, compared with those on usual care (P = .02).
The investigators note that the study “provides compelling evidence of a substantial long-term benefit for FEP treatment with the NAVIGATE EIS, compared with standard care.”
A ‘great message’
Commenting on the findings in an interview, Ragy R. Girgis, MD, associate professor of clinical psychiatry at Columbia University, New York, and the New York State Psychiatric Institute, said
Dr. Girgis, who was not involved in either study, said that the research is “a great message.” He noted that it is “really important for people to know and it’s really important that we’re still doing research in those areas.”
However, he noted that psychosocial interventions such as these “sometimes take a lot of work.” Dr. Girgis said that it is “so easy to just give people a medication” but that approach has its own disadvantages, including adverse effects and sometimes a lack of efficacy.
“Psychosocial interventions, on the other hand, are very well tolerated by people. They are very effective, but they may require a lot more manpower, and in some ways they can also be more expensive.
“So this is a dialectic that we oftentimes have to deal with when we figure out the right balance between psychosocial vs. medication types of treatments,” he said.
STEP has received research funding from the National Institutes of Health and the Patrick and Catherine Weldon Donaghue Medical Research Foundation. RAISE-ETP was funded by the National Institute of Mental Health as part of the Recovery After an Initial Schizophrenia Episode (RAISE) Project. Dr. Gopal is an employee of Janssen Research & Development, and owns stock/equity in Johnson & Johnson. Dr. Girgis has received research support from Genentech, BioAdvantex, Allegran/Forest, and Otsuka, and royalties from Wipf and Stock and Routledge/Taylor and Francis.
A version of this article originally appeared on Medscape.com.
Community-based services that tap into local environments not only reduce the duration of untreated psychosis (DUP) but also provide improved long-term outcomes for patients with first-episode psychosis (FEP), results of two new studies show.
In the first study, investigators led by Vinod Srihari, MD, director of specialized treatment early in psychosis at Yale University, New Haven, Conn., developed a program to reduce DUP to complement their first-episode service (FES).
Through a combination of mass media and social media campaigns, outreach events with local professionals, and rapid triage, the team was able to nearly halve the time from diagnosis to initiation of antipsychotic treatment.
In the second study, a team led by Delbert G. Robinson, MD, of Hofstra University, Hempstead, N.Y., conducted a 5-year follow-up of RAISE-ETP, the first U.S. randomized trial to compare a 2-year comprehensive early intervention service (EIS) with usual care.
These trial results showed that, among more than 400 FEP patients, the EIS significantly improved both symptoms and quality of life and reduced inpatient days in comparison with standard care.
The research was scheduled to be presented at the Congress of the Schizophrenia International Research Society (SIRS) 2020, but the meeting was canceled because of the coronavirus pandemic.
Norwegian model
Dr. Srihari and colleagues note that a specialized treatment early in psychosis (STEP), which delivers a specialty team–based FES, was established at their institution in 2006.
However, in a bid to reduce DUP, in 2015 they launched MindMap, a 4-year early-detection campaign based on the Scandinavian TIPS Early Detection in Psychosis Study.
Dr. Srihari said in an interview that they visited the team that developed the TIPS program in Norway “to try to understand what elements of their approach had resulted in a successful reduction of DUP.”
He pointed out that the health care system in Norway has “more reliable pathways to care, with an ability to route people in more predictable ways from primary care to secondary care, and so on.”
In the United States, “there’s no expectation that people will go through a primary care provider,” he said. He noted that patients “make their way to specialty care in many different ways.”
Dr. Srihari said, “The other change we realized we’d have to make was that, since TIPS had been completed many years back, the media environment had changed substantially.
“At the time that TIPS was done, there was no such thing as social media, whereas when we began to think about designing our campaign, we thought social media would be a very efficient and also cost-effective way to target young people.”
MindMap, which covered a 10-town catchment area that has a population of 400,000, targeted both demand- and supply-side aspects of DUP. The former focused on delays in identifying illness and help-seeking, and the latter concentrated on referral and treatment access delays.
The researchers used a combination of mass media and social media messaging, professional detailing, and the rapid triage of referrals.
The media campaign included Facebook, Twitter, and other social media platforms that allowed the team to “target individuals in a somewhat more fine-grained way than mass media allows,” Dr. Srihari said. They focused on individuals in a particular age range and geographic location.
The professional detailing encompassed mental health agencies, emergency departments, and inpatient units, as well as colleges, college counseling centers, high schools, and police departments. The team hosted meetings, “often at local restaurants, where we provided a meal and provided some general education about what our mission was,” said Dr. Srihari. The researchers followed up these meetings with more in-person visits.
“The third arm was finding ways to basically eliminate any kind of a waiting time at our front door, such as ensuring that transportation issues were circumvented, in addition to cutting through any ambivalence they might have about finally making the leap to come to the center.”
More rapid treatment
Over the course of the baseline year and the 4 years of the MindMap program, almost 1,500 individuals were assessed. Of these, approximately 200 were eligible, and almost all were enrolled.
The researchers measured DUP at two time points from the onset of psychosis – the initiation of antipsychotic treatment (DUP1) and the initiation of FES care. Across the study period, they found that DUP1 fell significantly between the pre- and postprogram assessments, from 329 days to 185 days (P = .03). By contrast, there was no change over the same period for the Prevention and Recovery in Early Psychosis FES program in Boston, which served as a comparator.
There was also a cumulative effect on DUP, with each year of the 4-year program associated with a 46-day reduction in DUP1. However, the significant reduction was restricted to the third quintile of DUP1 and was not found in the other quintiles of DUP1 or for DUP2, despite all measures showing a consistent trend for reduction over time.
Dr. Srihari acknowledges that the team was “disappointed” that DUP2 did not fall significantly in their study. He suggested, “It might take longer for agencies to change their workloads and refer patients to STEP, which is what ended up resulting in the DUP2 not dropping as quickly.”
To see whether there was indeed a time lag in changes to practice, the team conducted an analysis in which they cut out the first year from the results and analyzed only the last 3 years. Then “we do see a decline in DUP2,” he said.
The study’s full results are currently being prepared for publication, and the investigators are considering relaunching the initiative.
“The question we are having now is how to resource the campaign without the research funds and which parts of it we think we can launch sustainably so we can continue the reduction of DUP,” Dr. Srihari said.
Plans may include developing partnerships with local businesses to help fund the media costs and working with the state government to build a learning health care network, which would make it easier for mental health agencies to consult with the team on problematic cases.
“We’re trying to reduce DUP referrals on the supply side by providing this kind of learning health collaborative ... that we also think might be fiscally a more sustainable way to do this vs. what we did in MindMap,” which would be “very expensive” to implement on a statewide basis, Dr. Srihari added.
Long-term benefit
In the second study, Dr. Robinson and colleagues highlight that EISs have been implemented worldwide for FEP patients and have been associated with improved outcomes.
However, these services typically provide care for a limited period, and cross-sectional follow-up studies have identified few advantages in comparison with standard care.
To provide a more robust longitudinal assessment of the ongoing effects of an EIS, the team conducted a 5-year follow-up of the first U.S.-based, multicenter, randomized clinical trial comparing an EIS, NAVIGATE, with usual clinical care in FEP.
RAISE-ETP was conducted at 34 sites across the United States. Seventeen sites provided NAVIGATE to 223 individuals with FEP, and the remaining 17 sites provided usual care to 181 patients.
NAVIGATE, which continued for 2 years, consisted of treatments and services delivered by a coordinated team of providers. Those services included the following:
- Education on schizophrenia and its treatment for patients and their families.
- Symptom and relapse prevention medication, using a computerized decision support system.
- Strategies for illness management building personal resilience.
- A supported employment/education model.
Patients were assessed every 6 months for up to 60 months via a video link using the Heinrichs-Carpenter Quality of Life Scale (QLS) and the Positive and Negative Syndrome Scale (PANSS).
The average age of the participants was 23 years; 78% of those who received NAVIGATE and 66% of those who received usual care were male. The opportunity for each participant to engage in NAVIGATE treatment lasted an average of 33.8 months. The longest was 44.4 months.
Over 5 years, NAVIGATE was associated with a significant improvement over usual care in QLS scores by an average of 13.14 units (P < .001). PANSS scores improved by an average of 7.73 units (P < .002). QLS scores were not affected either by the length of opportunity to participate in NAVIGATE or by DUP, the team reports. Patients who received NAVIGATE also had an average of 2.5 fewer inpatient days, compared with those on usual care (P = .02).
The investigators note that the study “provides compelling evidence of a substantial long-term benefit for FEP treatment with the NAVIGATE EIS, compared with standard care.”
A ‘great message’
Commenting on the findings in an interview, Ragy R. Girgis, MD, associate professor of clinical psychiatry at Columbia University, New York, and the New York State Psychiatric Institute, said
Dr. Girgis, who was not involved in either study, said that the research is “a great message.” He noted that it is “really important for people to know and it’s really important that we’re still doing research in those areas.”
However, he noted that psychosocial interventions such as these “sometimes take a lot of work.” Dr. Girgis said that it is “so easy to just give people a medication” but that approach has its own disadvantages, including adverse effects and sometimes a lack of efficacy.
“Psychosocial interventions, on the other hand, are very well tolerated by people. They are very effective, but they may require a lot more manpower, and in some ways they can also be more expensive.
“So this is a dialectic that we oftentimes have to deal with when we figure out the right balance between psychosocial vs. medication types of treatments,” he said.
STEP has received research funding from the National Institutes of Health and the Patrick and Catherine Weldon Donaghue Medical Research Foundation. RAISE-ETP was funded by the National Institute of Mental Health as part of the Recovery After an Initial Schizophrenia Episode (RAISE) Project. Dr. Gopal is an employee of Janssen Research & Development, and owns stock/equity in Johnson & Johnson. Dr. Girgis has received research support from Genentech, BioAdvantex, Allegran/Forest, and Otsuka, and royalties from Wipf and Stock and Routledge/Taylor and Francis.
A version of this article originally appeared on Medscape.com.
Community-based services that tap into local environments not only reduce the duration of untreated psychosis (DUP) but also provide improved long-term outcomes for patients with first-episode psychosis (FEP), results of two new studies show.
In the first study, investigators led by Vinod Srihari, MD, director of specialized treatment early in psychosis at Yale University, New Haven, Conn., developed a program to reduce DUP to complement their first-episode service (FES).
Through a combination of mass media and social media campaigns, outreach events with local professionals, and rapid triage, the team was able to nearly halve the time from diagnosis to initiation of antipsychotic treatment.
In the second study, a team led by Delbert G. Robinson, MD, of Hofstra University, Hempstead, N.Y., conducted a 5-year follow-up of RAISE-ETP, the first U.S. randomized trial to compare a 2-year comprehensive early intervention service (EIS) with usual care.
These trial results showed that, among more than 400 FEP patients, the EIS significantly improved both symptoms and quality of life and reduced inpatient days in comparison with standard care.
The research was scheduled to be presented at the Congress of the Schizophrenia International Research Society (SIRS) 2020, but the meeting was canceled because of the coronavirus pandemic.
Norwegian model
Dr. Srihari and colleagues note that a specialized treatment early in psychosis (STEP), which delivers a specialty team–based FES, was established at their institution in 2006.
However, in a bid to reduce DUP, in 2015 they launched MindMap, a 4-year early-detection campaign based on the Scandinavian TIPS Early Detection in Psychosis Study.
Dr. Srihari said in an interview that they visited the team that developed the TIPS program in Norway “to try to understand what elements of their approach had resulted in a successful reduction of DUP.”
He pointed out that the health care system in Norway has “more reliable pathways to care, with an ability to route people in more predictable ways from primary care to secondary care, and so on.”
In the United States, “there’s no expectation that people will go through a primary care provider,” he said. He noted that patients “make their way to specialty care in many different ways.”
Dr. Srihari said, “The other change we realized we’d have to make was that, since TIPS had been completed many years back, the media environment had changed substantially.
“At the time that TIPS was done, there was no such thing as social media, whereas when we began to think about designing our campaign, we thought social media would be a very efficient and also cost-effective way to target young people.”
MindMap, which covered a 10-town catchment area that has a population of 400,000, targeted both demand- and supply-side aspects of DUP. The former focused on delays in identifying illness and help-seeking, and the latter concentrated on referral and treatment access delays.
The researchers used a combination of mass media and social media messaging, professional detailing, and the rapid triage of referrals.
The media campaign included Facebook, Twitter, and other social media platforms that allowed the team to “target individuals in a somewhat more fine-grained way than mass media allows,” Dr. Srihari said. They focused on individuals in a particular age range and geographic location.
The professional detailing encompassed mental health agencies, emergency departments, and inpatient units, as well as colleges, college counseling centers, high schools, and police departments. The team hosted meetings, “often at local restaurants, where we provided a meal and provided some general education about what our mission was,” said Dr. Srihari. The researchers followed up these meetings with more in-person visits.
“The third arm was finding ways to basically eliminate any kind of a waiting time at our front door, such as ensuring that transportation issues were circumvented, in addition to cutting through any ambivalence they might have about finally making the leap to come to the center.”
More rapid treatment
Over the course of the baseline year and the 4 years of the MindMap program, almost 1,500 individuals were assessed. Of these, approximately 200 were eligible, and almost all were enrolled.
The researchers measured DUP at two time points from the onset of psychosis – the initiation of antipsychotic treatment (DUP1) and the initiation of FES care. Across the study period, they found that DUP1 fell significantly between the pre- and postprogram assessments, from 329 days to 185 days (P = .03). By contrast, there was no change over the same period for the Prevention and Recovery in Early Psychosis FES program in Boston, which served as a comparator.
There was also a cumulative effect on DUP, with each year of the 4-year program associated with a 46-day reduction in DUP1. However, the significant reduction was restricted to the third quintile of DUP1 and was not found in the other quintiles of DUP1 or for DUP2, despite all measures showing a consistent trend for reduction over time.
Dr. Srihari acknowledges that the team was “disappointed” that DUP2 did not fall significantly in their study. He suggested, “It might take longer for agencies to change their workloads and refer patients to STEP, which is what ended up resulting in the DUP2 not dropping as quickly.”
To see whether there was indeed a time lag in changes to practice, the team conducted an analysis in which they cut out the first year from the results and analyzed only the last 3 years. Then “we do see a decline in DUP2,” he said.
The study’s full results are currently being prepared for publication, and the investigators are considering relaunching the initiative.
“The question we are having now is how to resource the campaign without the research funds and which parts of it we think we can launch sustainably so we can continue the reduction of DUP,” Dr. Srihari said.
Plans may include developing partnerships with local businesses to help fund the media costs and working with the state government to build a learning health care network, which would make it easier for mental health agencies to consult with the team on problematic cases.
“We’re trying to reduce DUP referrals on the supply side by providing this kind of learning health collaborative ... that we also think might be fiscally a more sustainable way to do this vs. what we did in MindMap,” which would be “very expensive” to implement on a statewide basis, Dr. Srihari added.
Long-term benefit
In the second study, Dr. Robinson and colleagues highlight that EISs have been implemented worldwide for FEP patients and have been associated with improved outcomes.
However, these services typically provide care for a limited period, and cross-sectional follow-up studies have identified few advantages in comparison with standard care.
To provide a more robust longitudinal assessment of the ongoing effects of an EIS, the team conducted a 5-year follow-up of the first U.S.-based, multicenter, randomized clinical trial comparing an EIS, NAVIGATE, with usual clinical care in FEP.
RAISE-ETP was conducted at 34 sites across the United States. Seventeen sites provided NAVIGATE to 223 individuals with FEP, and the remaining 17 sites provided usual care to 181 patients.
NAVIGATE, which continued for 2 years, consisted of treatments and services delivered by a coordinated team of providers. Those services included the following:
- Education on schizophrenia and its treatment for patients and their families.
- Symptom and relapse prevention medication, using a computerized decision support system.
- Strategies for illness management building personal resilience.
- A supported employment/education model.
Patients were assessed every 6 months for up to 60 months via a video link using the Heinrichs-Carpenter Quality of Life Scale (QLS) and the Positive and Negative Syndrome Scale (PANSS).
The average age of the participants was 23 years; 78% of those who received NAVIGATE and 66% of those who received usual care were male. The opportunity for each participant to engage in NAVIGATE treatment lasted an average of 33.8 months. The longest was 44.4 months.
Over 5 years, NAVIGATE was associated with a significant improvement over usual care in QLS scores by an average of 13.14 units (P < .001). PANSS scores improved by an average of 7.73 units (P < .002). QLS scores were not affected either by the length of opportunity to participate in NAVIGATE or by DUP, the team reports. Patients who received NAVIGATE also had an average of 2.5 fewer inpatient days, compared with those on usual care (P = .02).
The investigators note that the study “provides compelling evidence of a substantial long-term benefit for FEP treatment with the NAVIGATE EIS, compared with standard care.”
A ‘great message’
Commenting on the findings in an interview, Ragy R. Girgis, MD, associate professor of clinical psychiatry at Columbia University, New York, and the New York State Psychiatric Institute, said
Dr. Girgis, who was not involved in either study, said that the research is “a great message.” He noted that it is “really important for people to know and it’s really important that we’re still doing research in those areas.”
However, he noted that psychosocial interventions such as these “sometimes take a lot of work.” Dr. Girgis said that it is “so easy to just give people a medication” but that approach has its own disadvantages, including adverse effects and sometimes a lack of efficacy.
“Psychosocial interventions, on the other hand, are very well tolerated by people. They are very effective, but they may require a lot more manpower, and in some ways they can also be more expensive.
“So this is a dialectic that we oftentimes have to deal with when we figure out the right balance between psychosocial vs. medication types of treatments,” he said.
STEP has received research funding from the National Institutes of Health and the Patrick and Catherine Weldon Donaghue Medical Research Foundation. RAISE-ETP was funded by the National Institute of Mental Health as part of the Recovery After an Initial Schizophrenia Episode (RAISE) Project. Dr. Gopal is an employee of Janssen Research & Development, and owns stock/equity in Johnson & Johnson. Dr. Girgis has received research support from Genentech, BioAdvantex, Allegran/Forest, and Otsuka, and royalties from Wipf and Stock and Routledge/Taylor and Francis.
A version of this article originally appeared on Medscape.com.
The hidden dangers of supplements: A case of substance-induced psychosis
“You are what you eat,” my mother always said, and structured our dinner plates according to the USDA food pyramid. We dutifully consumed leafy greens, and prior to medical school I invested time and money into healthy diet choices. I drank green smoothies, pureed baby food for my children, read up on the pH balancing diet, grew sprouts on windowsills, bought organic.
With the stressors and time constraints of managing medical school and a family, nutrition tumbled down the ladder of priorities until eventually my family was subsisting on chicken nuggets, pizza, and peanut butter. Intern year has only added the occasional candy bar from the doctors’ lounge. I experienced a vague sense of loss for something I had once valued, but simultaneously felt dismissive of trendy topics such as omega-3 fatty acids and antioxidants in the face of myocardial infarctions and liver failure. A biochemistry professor once scoffed at “the laypeople’s obsession with toxins,” and nutrition received zero attention in our medical school curriculum or board exams.
However, a clinical experience on the inpatient psychiatric unit made me reevaluate the importance nutrition should have in both our personal lives and the practice of medicine. This is the case of an otherwise healthy young man with no psychiatric history who suffered a psychotic break after ingesting an excess of a supplement he purchased online with the purpose of improving his performance at a high-stress job.
CASE REPORT
Mr. K, a 28-year-old computer programmer, was voluntarily admitted to the inpatient psychiatry unit for paranoia and persecutory delusions along with auditory hallucinations. His father reported that Mr. K had been behaving erratically for several days prior to admission and was subsequently found wandering in the street.
On admission, Mr. K was not oriented to place or situation. He was unkempt and guarded, and claimed people were following him. His urine toxicology screen and blood alcohol levels were negative.
While hospitalized, Mr. K was hyperverbal and delusional. He related that at work he had been developing programs to make slaves in the computer, “algorithms for orchestration,” and that he was uncomfortable with the ethical implications. He eventually endorsed having purchased the supplement phenylethylamine (PEA) to improve his focus, and ingesting “two substantial scoops of the crystalline substance.”
We did not initiate any psychiatric medications. On the third day of his hospitalization, Mr. K was alert, oriented, euthymic, relaxed, and had a full range of affect; upon discharge we advised him to discard the PEA and avoid stimulants. He complied, quit his high-stress job, and had no subsequent psychotic symptoms in the 7 months since discharge.
Continue to: Dietary supplements carry risks
Dietary supplements carry risks
According to the FDA, dietary supplements are regulated as food, but many have strong biologic effects or may even contain drugs.1 More than 18% of Americans use herbal or nutritional therapies as part of their health regimen.2 However, many over-the-counter remedies have been found to exhibit psychotropic effects,3 and many more are purported to impact mental and physical health with little to no scientific research into these claims or potential adverse effects.
Phenylethylamine is sold as a nutritional supplement and marketed for its purported beneficial effects on weight loss, mood, and focus.4 However, PEA is known to act as a natural amphetamine and to play a role in the development of neuropsychiatric disorders.5 It is an endogenous psychotogenic molecule that has been previously theorized as a cause for primary psychosis.6 Phenylethylamine interacts with the same receptor ligand that responds to amphetamine and related compounds (such as methamphetamine and 3,4-methylenedioxy-methamphetamine [MDMA]), the genetic coding for which is located in an area of DNA associated with schizophrenia: chromosome 6q23.2.7 While the mechanisms and details of these interactions remain poorly understood, this case of PEA-induced psychosis represents a glimpse into the potential psychoactive properties of this readily available nutritional supplement.
This patient’s cautionary tale has given me pause regarding both my family’s nutrition and the oft-neglected dietary portion of the social history. Also, several subsequent patient experiences hearken back to my mother’s words regarding the importance of healthy eating. A patient with phenylketonuria presented with psychosis after running out of her formula and consuming junk food. Another patient with severely elevated blood glucose levels presented with confusion. I have come to realize that ingestion impacts presentation, or, in other words, you are what you eat.
1. US Food and Drug Administration. Dietary supplements. https://www.fda.gov/consumers/consumer-updates/dietary-supplements. Accessed December 11, 2019.
2. Tindle H, Davis R, Philips R, et al. Trends in use of complementary and alternative medicine by US adults: 1997-2002. Altern Ther Health Med. 2005;11(1):42-49.
3. Sarris J. Herbal medicines in the treatment of psychiatric disorders: 10-year updated review. Phytotherapy Research. 2018;32(7):1147-1162.
4. Irsfeld M, Spadafore M, Prüß BM. β-phenylethylamine, a small molecule with a large impact. WebmedCentral. 2013;4(9):4409.
5. Wolf M, Mosnaim A. Phenylethylamine in neuropsychiatric disorders. Gen Pharmacol. 1983;14(4):385-390.
6. Janssen P, Leysen J, Megens A, et al. Does phenylethylamine act as an endogenous amphetamine in some patients? In J Neuropsychopharmacol. 1999;2(3):229-240.
7. Zucchi R, Chiellini G, Scanlan TS, et al. Trace amine-associated receptors and their ligands. Br J Pharmacol. 2006;149(8):967-978.
“You are what you eat,” my mother always said, and structured our dinner plates according to the USDA food pyramid. We dutifully consumed leafy greens, and prior to medical school I invested time and money into healthy diet choices. I drank green smoothies, pureed baby food for my children, read up on the pH balancing diet, grew sprouts on windowsills, bought organic.
With the stressors and time constraints of managing medical school and a family, nutrition tumbled down the ladder of priorities until eventually my family was subsisting on chicken nuggets, pizza, and peanut butter. Intern year has only added the occasional candy bar from the doctors’ lounge. I experienced a vague sense of loss for something I had once valued, but simultaneously felt dismissive of trendy topics such as omega-3 fatty acids and antioxidants in the face of myocardial infarctions and liver failure. A biochemistry professor once scoffed at “the laypeople’s obsession with toxins,” and nutrition received zero attention in our medical school curriculum or board exams.
However, a clinical experience on the inpatient psychiatric unit made me reevaluate the importance nutrition should have in both our personal lives and the practice of medicine. This is the case of an otherwise healthy young man with no psychiatric history who suffered a psychotic break after ingesting an excess of a supplement he purchased online with the purpose of improving his performance at a high-stress job.
CASE REPORT
Mr. K, a 28-year-old computer programmer, was voluntarily admitted to the inpatient psychiatry unit for paranoia and persecutory delusions along with auditory hallucinations. His father reported that Mr. K had been behaving erratically for several days prior to admission and was subsequently found wandering in the street.
On admission, Mr. K was not oriented to place or situation. He was unkempt and guarded, and claimed people were following him. His urine toxicology screen and blood alcohol levels were negative.
While hospitalized, Mr. K was hyperverbal and delusional. He related that at work he had been developing programs to make slaves in the computer, “algorithms for orchestration,” and that he was uncomfortable with the ethical implications. He eventually endorsed having purchased the supplement phenylethylamine (PEA) to improve his focus, and ingesting “two substantial scoops of the crystalline substance.”
We did not initiate any psychiatric medications. On the third day of his hospitalization, Mr. K was alert, oriented, euthymic, relaxed, and had a full range of affect; upon discharge we advised him to discard the PEA and avoid stimulants. He complied, quit his high-stress job, and had no subsequent psychotic symptoms in the 7 months since discharge.
Continue to: Dietary supplements carry risks
Dietary supplements carry risks
According to the FDA, dietary supplements are regulated as food, but many have strong biologic effects or may even contain drugs.1 More than 18% of Americans use herbal or nutritional therapies as part of their health regimen.2 However, many over-the-counter remedies have been found to exhibit psychotropic effects,3 and many more are purported to impact mental and physical health with little to no scientific research into these claims or potential adverse effects.
Phenylethylamine is sold as a nutritional supplement and marketed for its purported beneficial effects on weight loss, mood, and focus.4 However, PEA is known to act as a natural amphetamine and to play a role in the development of neuropsychiatric disorders.5 It is an endogenous psychotogenic molecule that has been previously theorized as a cause for primary psychosis.6 Phenylethylamine interacts with the same receptor ligand that responds to amphetamine and related compounds (such as methamphetamine and 3,4-methylenedioxy-methamphetamine [MDMA]), the genetic coding for which is located in an area of DNA associated with schizophrenia: chromosome 6q23.2.7 While the mechanisms and details of these interactions remain poorly understood, this case of PEA-induced psychosis represents a glimpse into the potential psychoactive properties of this readily available nutritional supplement.
This patient’s cautionary tale has given me pause regarding both my family’s nutrition and the oft-neglected dietary portion of the social history. Also, several subsequent patient experiences hearken back to my mother’s words regarding the importance of healthy eating. A patient with phenylketonuria presented with psychosis after running out of her formula and consuming junk food. Another patient with severely elevated blood glucose levels presented with confusion. I have come to realize that ingestion impacts presentation, or, in other words, you are what you eat.
“You are what you eat,” my mother always said, and structured our dinner plates according to the USDA food pyramid. We dutifully consumed leafy greens, and prior to medical school I invested time and money into healthy diet choices. I drank green smoothies, pureed baby food for my children, read up on the pH balancing diet, grew sprouts on windowsills, bought organic.
With the stressors and time constraints of managing medical school and a family, nutrition tumbled down the ladder of priorities until eventually my family was subsisting on chicken nuggets, pizza, and peanut butter. Intern year has only added the occasional candy bar from the doctors’ lounge. I experienced a vague sense of loss for something I had once valued, but simultaneously felt dismissive of trendy topics such as omega-3 fatty acids and antioxidants in the face of myocardial infarctions and liver failure. A biochemistry professor once scoffed at “the laypeople’s obsession with toxins,” and nutrition received zero attention in our medical school curriculum or board exams.
However, a clinical experience on the inpatient psychiatric unit made me reevaluate the importance nutrition should have in both our personal lives and the practice of medicine. This is the case of an otherwise healthy young man with no psychiatric history who suffered a psychotic break after ingesting an excess of a supplement he purchased online with the purpose of improving his performance at a high-stress job.
CASE REPORT
Mr. K, a 28-year-old computer programmer, was voluntarily admitted to the inpatient psychiatry unit for paranoia and persecutory delusions along with auditory hallucinations. His father reported that Mr. K had been behaving erratically for several days prior to admission and was subsequently found wandering in the street.
On admission, Mr. K was not oriented to place or situation. He was unkempt and guarded, and claimed people were following him. His urine toxicology screen and blood alcohol levels were negative.
While hospitalized, Mr. K was hyperverbal and delusional. He related that at work he had been developing programs to make slaves in the computer, “algorithms for orchestration,” and that he was uncomfortable with the ethical implications. He eventually endorsed having purchased the supplement phenylethylamine (PEA) to improve his focus, and ingesting “two substantial scoops of the crystalline substance.”
We did not initiate any psychiatric medications. On the third day of his hospitalization, Mr. K was alert, oriented, euthymic, relaxed, and had a full range of affect; upon discharge we advised him to discard the PEA and avoid stimulants. He complied, quit his high-stress job, and had no subsequent psychotic symptoms in the 7 months since discharge.
Continue to: Dietary supplements carry risks
Dietary supplements carry risks
According to the FDA, dietary supplements are regulated as food, but many have strong biologic effects or may even contain drugs.1 More than 18% of Americans use herbal or nutritional therapies as part of their health regimen.2 However, many over-the-counter remedies have been found to exhibit psychotropic effects,3 and many more are purported to impact mental and physical health with little to no scientific research into these claims or potential adverse effects.
Phenylethylamine is sold as a nutritional supplement and marketed for its purported beneficial effects on weight loss, mood, and focus.4 However, PEA is known to act as a natural amphetamine and to play a role in the development of neuropsychiatric disorders.5 It is an endogenous psychotogenic molecule that has been previously theorized as a cause for primary psychosis.6 Phenylethylamine interacts with the same receptor ligand that responds to amphetamine and related compounds (such as methamphetamine and 3,4-methylenedioxy-methamphetamine [MDMA]), the genetic coding for which is located in an area of DNA associated with schizophrenia: chromosome 6q23.2.7 While the mechanisms and details of these interactions remain poorly understood, this case of PEA-induced psychosis represents a glimpse into the potential psychoactive properties of this readily available nutritional supplement.
This patient’s cautionary tale has given me pause regarding both my family’s nutrition and the oft-neglected dietary portion of the social history. Also, several subsequent patient experiences hearken back to my mother’s words regarding the importance of healthy eating. A patient with phenylketonuria presented with psychosis after running out of her formula and consuming junk food. Another patient with severely elevated blood glucose levels presented with confusion. I have come to realize that ingestion impacts presentation, or, in other words, you are what you eat.
1. US Food and Drug Administration. Dietary supplements. https://www.fda.gov/consumers/consumer-updates/dietary-supplements. Accessed December 11, 2019.
2. Tindle H, Davis R, Philips R, et al. Trends in use of complementary and alternative medicine by US adults: 1997-2002. Altern Ther Health Med. 2005;11(1):42-49.
3. Sarris J. Herbal medicines in the treatment of psychiatric disorders: 10-year updated review. Phytotherapy Research. 2018;32(7):1147-1162.
4. Irsfeld M, Spadafore M, Prüß BM. β-phenylethylamine, a small molecule with a large impact. WebmedCentral. 2013;4(9):4409.
5. Wolf M, Mosnaim A. Phenylethylamine in neuropsychiatric disorders. Gen Pharmacol. 1983;14(4):385-390.
6. Janssen P, Leysen J, Megens A, et al. Does phenylethylamine act as an endogenous amphetamine in some patients? In J Neuropsychopharmacol. 1999;2(3):229-240.
7. Zucchi R, Chiellini G, Scanlan TS, et al. Trace amine-associated receptors and their ligands. Br J Pharmacol. 2006;149(8):967-978.
1. US Food and Drug Administration. Dietary supplements. https://www.fda.gov/consumers/consumer-updates/dietary-supplements. Accessed December 11, 2019.
2. Tindle H, Davis R, Philips R, et al. Trends in use of complementary and alternative medicine by US adults: 1997-2002. Altern Ther Health Med. 2005;11(1):42-49.
3. Sarris J. Herbal medicines in the treatment of psychiatric disorders: 10-year updated review. Phytotherapy Research. 2018;32(7):1147-1162.
4. Irsfeld M, Spadafore M, Prüß BM. β-phenylethylamine, a small molecule with a large impact. WebmedCentral. 2013;4(9):4409.
5. Wolf M, Mosnaim A. Phenylethylamine in neuropsychiatric disorders. Gen Pharmacol. 1983;14(4):385-390.
6. Janssen P, Leysen J, Megens A, et al. Does phenylethylamine act as an endogenous amphetamine in some patients? In J Neuropsychopharmacol. 1999;2(3):229-240.
7. Zucchi R, Chiellini G, Scanlan TS, et al. Trace amine-associated receptors and their ligands. Br J Pharmacol. 2006;149(8):967-978.
New-onset psychosis while being treated for coronavirus
CASE Agitated, psychotic, and COVID-19–positive
Mr. G, age 56, is brought to the emergency department (ED) by emergency medical services (EMS) after his girlfriend reports that he was trying to climb into the “fiery furnace” to “burn the devil within him.” Mr. G had recently tested positive for coronavirus disease 2019 (COVID-19) via polymerase chain reaction and had been receiving treatment for it. In the ED, he is distressed and repeatedly exclaims, “The devil is alive!” He insists on covering himself with blankets, despite diaphoresis and soaking through his clothing within minutes. Because he does not respond to attempted redirection, the ED clinicians administer a single dose of IM haloperidol, 2 mg, for agitation.
HISTORY Multiple ED visits and hospitalizations
Mr. G, who has no known psychiatric history, lives with his girlfriend of 10 years. His medical history includes chronic obstructive pulmonary disease and prostate cancer. In 2015, he had a radical prostatectomy, without chemotherapy. His social history includes childhood neglect, which prompted him to leave home when he was a teenager. Mr. G had earned his general education development certificate and worked at a small retail store.
Mr. G had no previous history of mental health treatment per self-report, collateral information from his girlfriend, and chart review. He reported no known family psychiatric history. He did not endorse past psychiatric admissions or suicide attempts, nor previous periods of mania, depression, or psychosis. He said he used illicit substances as a teen, but denied using alcohol, tobacco products, or illicit substances in the past 20 years.
Mr. G recently had multiple ED visits and hospitalizations due to ongoing signs and symptoms associated with his COVID-19 diagnosis, primarily worsening shortness of breath and cough. Eleven days before EMS brought him to the ED at his girlfriend’s request, Mr. G had presented to the ED with chief complaints of shortness of breath and dry cough (Day 0). He reported that he had been “running a fever” for 2 days. In the ED, his initial vital signs were notable only for a temperature of 100.9°F (38.28°C). He was diagnosed with “acute viral syndrome” and received 1 dose of IV ceftriaxone, 2 g, and IV azithromycin, 500 mg. On Day 2, the ED clinicians prescribed a 4-day course of oral azithromycin, 250 mg/d, and discharged him home.
On Day 3, Mr. G returned to the ED with similar complaints—congestion and productive cough. He tested positive for COVID-19, and the ED discharged him home with quarantine instructions. Hours later, he returned to the ED via EMS with chief complaints of chest pain, diarrhea, and myalgias. He was prescribed a 5-day course ofoseltamivir, 75 mg twice daily, and azithromycin, 250 mg/d. The ED again discharged him home.
On Day 4, Mr. G returned to the ED for a fourth time. His chief complaint was worsening shortness of breath. His oxygen saturation was 94% on room air; it improved to 96% on 2 L of oxygen. His chest X-ray showed diffuse reticulonodular opacities throughout his bilateral lung fields and increased airspace opacification in the bilateral lower lobes. The ED admitted Mr. G to an internal medicine unit, where the primary treatment team enrolled him in a clinical trial. As part of the trial, Mr. G received hydroxychloroquine, 400 mg, on Day 4 and Day 5. The placebo-controlled component of the trial involved Mr. G receiving daily infusions of either remdesivir or placebo on Day 6 through Day 8. On Day 8, Mr. G was discharged home.
On Day 9, Mr. G returned to the ED with a chief complaint that his “thermometer wasn’t working” at home. The ED readmitted him to the internal medicine unit. On Day 9 through Day 11, Mr. G received daily doses of
Continue to: During the second hospitalization...
During the second hospitalization, nursing staff reported that Mr. G seemed religiously preoccupied and once reported seeing angels and demons. He was observed sitting in a chair praying to Allah that he would “come in on a horse to chop all the workers’ heads off.”
On Day 11, Mr. G was discharged home. Later that evening, the EMS brought him back in the ED due to his girlfriend’s concerns about his mental state.
EVALUATION Talks to God
On Day 12, psychiatry is consulted to evaluate Mr. G’s new-onset psychosis. Mr. G is alert and oriented to person, place, and time. His speech is loud, though the amount and rate are unremarkable. He displays no psychomotor agitation. His thought process is tangential and focuses on religious themes, specifically referring to Islam. He reports auditory hallucinations of God speaking directly to him. Mr. G states, “I am here because of a miraculous transformation from death back to life. Do you believe in God? Which God do you believe in? There are 2 Gods and only one of them is the true God. He is the God of all the 7 heavens and His true name is Allah, only one God, one faith. Allah is a ball of energy.”
Mr. G’s girlfriend provides collateral information that Mr. G had been raised Christian but was not religious as an adult. She says that he had never spoken about being Muslim. She adds that she had never known him to speak much about religion.
[polldaddy:10572249]
The authors’ observations
The etiology of new-onset psychosis can be related to several factors, including primary psychiatric illnesses, use of illicit substances, sequelae of general medical conditions, or adverse effects of prescribed medications. We considered each of these in the differential diagnosis for Mr. G.
Continue to: Psychiatric illness or illicit substance use
Psychiatric illness or illicit substance use. Because Mr. G was 56 years old and had no known psychiatric history or family psychiatric history, a primary psychiatric illness seemed less likely. Substance-induced psychosis related to illicit substance use also seemed unlikely because he denied using illicit substances, and an expanded urine drug screen was negative.
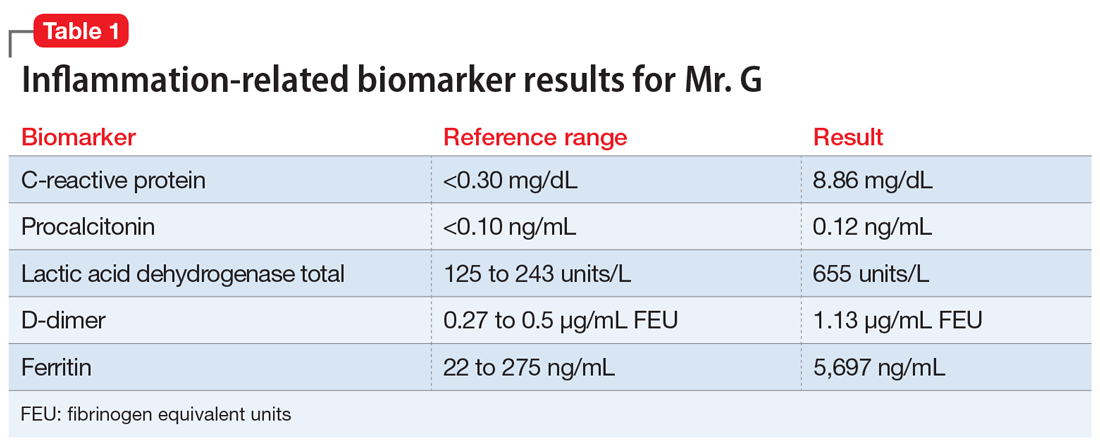
Psychosis due to a general medical condition. Results from Mr. G’s laboratory workup show marked elevation in multiple inflammation-related biomarkers (Table 1), consistent with the inflammatory profile seen with COVID-19 infection. However, results from several laboratory tests for potential etiologies of new-onset psychosis due to a general medical condition were negative (Table 2). Based on Mr. G’s history of prostate cancer, we considered the possibility of metastatic space-occupying lesions of the brain; however, Mr. G’s head CT showed no acute intracranial abnormalities. Another possible etiology we considered was COVID-19–induced encephalitis; however, Mr. G’s brain MRI with and without contrast showed no evidence of acute or chronic intracranial changes.
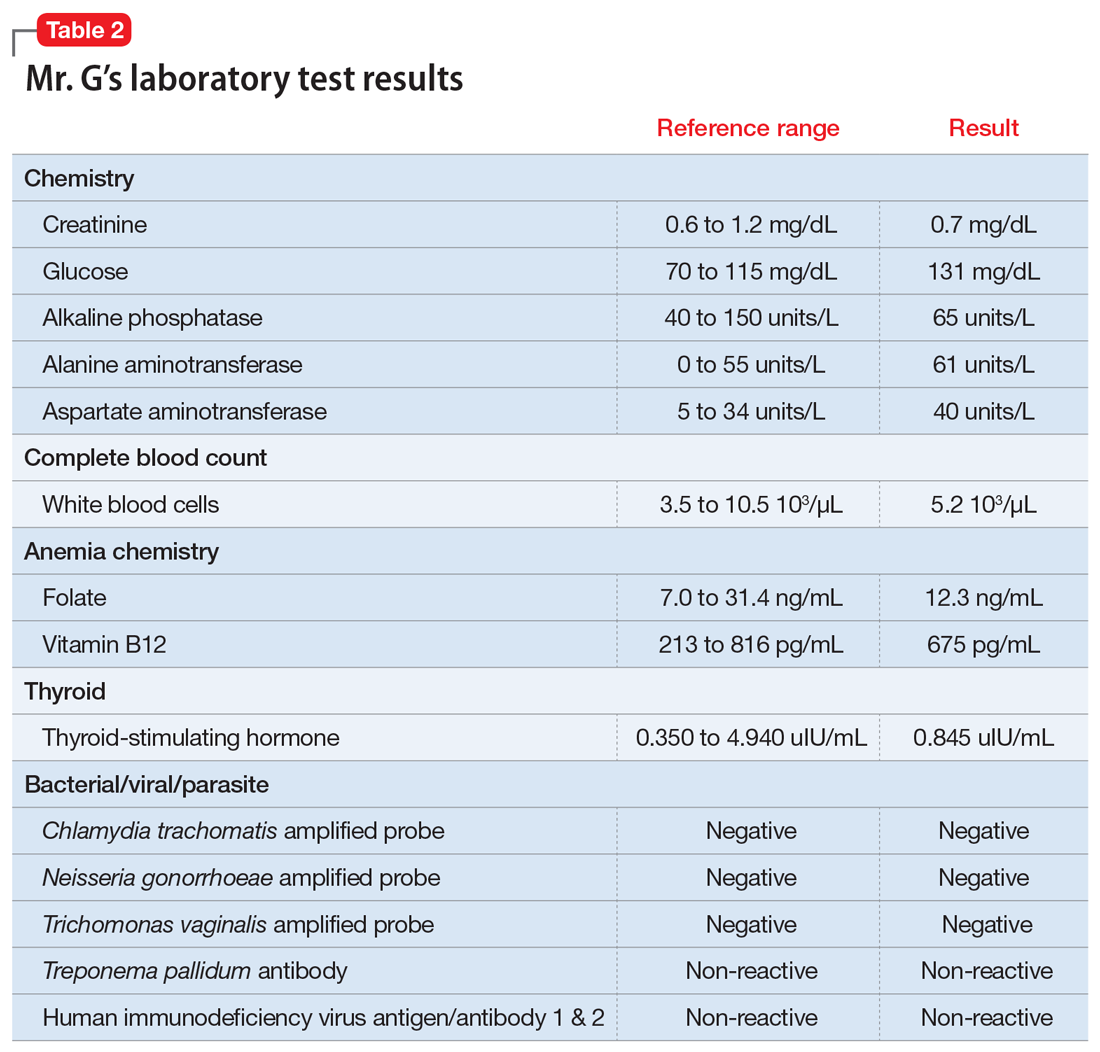
Medication-induced psychosis. After largely ruling out primary psychiatric illnesses, illicit substances, and sequelae of general medical conditions, we turned our attention to prescribed medications as a potential etiology of Mr. G’s new-onset psychosis. During his initial hospitalization, Mr. G had been prescribed 2 doses of hydroxychloroquine, 400 mg, to treat his diagnosis of COVID-19. Because none of the other medications he received were reported to have neuropsychiatric adverse effects, including psychosis, hydroxychloroquine-induced psychosis was therefore the primary team’s working diagnosis.
EVALUATION Request to leave AMA
On Day 13, Mr. G requests to leave the hospital against medical advice (AMA). Until this point, he had voluntarily remained in the hospital, which he repeatedly referred to as “Heaven.” When asked to describe his medical condition, Mr. G replies, “God told me my condition is far beyond man’s understanding.” He denies that he is positive for COVID-19. He states, “I am cured, and the real fight has just begun.”
At the recommendation of the psychiatry consultation-liaison (C-L) service, the primary treatment team determines that Mr. G does not have capacity to leave AMA. The team is concerned that because of his psychotic symptoms, Mr. G would be unable to understand and follow his quarantine instructions. He remains hospitalized on a medical hold.
Continue to: The authors' observations
The authors’ observations
One important consideration this case highlighted was potential third-party responsibility clinicians and hospital systems may face if they discharge a patient with a communicable illness who is unable to follow precautions based on a psychiatric condition.1 That concern was based on Mr. G’s reported desire to pursue missions “beyond man’s understanding,” which he felt compelled to complete, and which could unnecessarily place the public at risk. The psychiatry C-L service consulted the local health department and conferred with the hospital’s legal representatives, who agreed with the plan to keep Mr. G in the hospital for his safety as well as for the public’s safety.
TREATMENT Oral haloperidol
The psychiatry C-L service recommends initiating an antipsychotic. On Day 13, Mr. G starts oral haloperidol, 2.5 mg twice a day, to address his ongoing psychotic symptoms. On Day 14, the treatment team increases the dosage to 5 mg twice a day. Mr. G tolerates the haloperidol and gradually begins to improve. He demonstrates improved sleep, normal speech volume, less religious preoccupation, and a considerably improved understanding of his medical condition.
The authors’ observations
Mr. G’s initial psychiatric evaluation demonstrated an acute onset of psychotic symptoms, without evidence of delirium. Psychosis secondary to a general medical condition (such as COVID-19) and hydroxychloroquine-induced psychotic disorder topped our initial considerations in the differential diagnosis of this case. While the exact neuropsychiatric sequelae of COVID-19 are not yet clear, previous experiences with viral pandemics and case studies from the current pandemic demonstrate a wide variety of possible neuropsychiatric manifestations. Mood symptoms, psychosis, and encephalopathy represent some of the neuropsychiatric complications observed with past viral pandemics.2 Neuropsychiatric symptoms may be triggered by the virus itself, or from the host’s immune response to the infection.3 To further complicate matters, neuropsychiatric symptoms may manifest during the acute viral infection, or may surface later, as subacute or chronic neuropsychiatric illness.
Neuropsychiatric adverse events
Mr. G developed psychotic symptoms within the first few days of receiving hydroxychloroquine, which is consistent with the scant literature on this topic.8 Based on the available information, hydroxychloroquine remains the most likely etiology of his new-onset psychotic symptoms. Mr. G’s case is one example of the possible neuropsychiatric presentations clinicians may face while treating a novel viral illness.
Continue to: OUTCOME Homeward-bound
OUTCOME Homeward-bound
By Day 18, Mr. G’s psychotic symptoms have significantly improved. He is able to rationally process information about his COVID-19 diagnosis and the recommended quarantine instructions he needs to follow after discharge. He is cleared by infection control and discharged home to return to living with his girlfriend.
Mr. G attends his follow-up psychiatric appointment remotely 2 weeks after discharge. He reports that since discharge, he has continued taking his prescribed haloperidol, 5 mg twice a day. He demonstrates improved insight into his medical condition, acknowledging his COVID-19–positive status, and confirms that he has been following quarantine instructions. He does not report ongoing auditory or visual hallucinations, and is no longer religiously preoccupied. He says he is looking forward to being medically cleared to return to work.
The authors’ observations
This case highlights the need for prospective, longitudinal screening and monitoring of neuropsychiatric symptoms as part of the public health response to COVID-19. The case also highlights the importance of careful monitoring for adverse events, including neuropsychiatric symptoms, during clinical trials that involve experimental treatments. The long-term prognosis for individuals such as Mr. G who develop neuropsychiatric symptoms during acute COVID-19 infection remains unknown. Similarly, subacute and chronic neuropsychiatric manifestations that may develop after resolution of acute COVID-19 infection are unknown at this time. However, we can learn from past viral pandemics and anticipate that neuropsychiatric sequelae are likely to occur and should be part of the public health response to the pandemic.
Bottom Line
The coronavirus disease 2019 pandemic provides multiple clinical challenges pertinent to psychiatry. Neuropsychiatric symptoms may manifest from delirium, viral infection, host immune response, or adverse reactions to experimental treatments. These potential neuropsychiatric symptoms may complicate medical treatment. They can also raise important ethical and legal considerations, such as weighing patient autonomy vs third-party responsibility to the public at large.
Related Resources
- Ferrando SJ, Klepacz L, Lynch S, et al. COVID-19 psychosis: a potential new neuropsychiatric condition triggered by novel coronavirus infection and the inflammatory response? [published online May 19, 2020]. Psychosomatics. 2020. doi: 10.1016/j.psym.2020.05.012.
- Vlessides M. COVID-19 and psychosis: is there a link? Medscape Medical News. https://www.medscape.com/viewarticle/930224. Published May 8, 2020.
Drug Brand Names
Azithromycin • Zithromax
Ceftriaxone • Rocephin
Chloroquine • Aralen
Haloperidol • Haldol
Hydroxychloroquine • Plaquenil
Levofloxacin • Levaquin
Oseltamivir • Tamiflu
1. Ghossoub E, Newman WJ. COVID-19 and the duty to protect from communicable diseases. [published online ahead of print, May 8, 2020]. J Am Acad Psychiatry Law.
2. Menninger Ka. Psychoses associated with influenza: I. general data: statistical analysis. JAMA. 1919;72(4):235-241.
3. Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain, Behavior, and Immunity. 2020. doi:10.1016/j.bbi.2020.04.027.
4. Alkadi HO. Antimalarial drug toxicity: a review. Chemotherapy. 2007;53(6):385-391.
5. Bogaczewicz A, Sobów T. Psychiatric adverse effects of chloroquine. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-114.
6. Sato K, Mano T, Iwata A, et al. Neuropsychiatric adverse events of chloroquine: a real-world pharmacovigilance study using the FDA Adverse Event Reporting System (FAERS) database. Biosci Trends. 2020;14(2):139-143.
7. Cortegiani A, Ingoglia G, Ippolito M, et al. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care. 2020;57:279-283.
8. Das P, Rai A, Chopra A, et al. Psychosis likely induced by hydroxychloroquine in a patient with chronic Q fever: a case report and clinically relevant review of pharmacology. Psychosomatics. 2014;55(4):409-413.
CASE Agitated, psychotic, and COVID-19–positive
Mr. G, age 56, is brought to the emergency department (ED) by emergency medical services (EMS) after his girlfriend reports that he was trying to climb into the “fiery furnace” to “burn the devil within him.” Mr. G had recently tested positive for coronavirus disease 2019 (COVID-19) via polymerase chain reaction and had been receiving treatment for it. In the ED, he is distressed and repeatedly exclaims, “The devil is alive!” He insists on covering himself with blankets, despite diaphoresis and soaking through his clothing within minutes. Because he does not respond to attempted redirection, the ED clinicians administer a single dose of IM haloperidol, 2 mg, for agitation.
HISTORY Multiple ED visits and hospitalizations
Mr. G, who has no known psychiatric history, lives with his girlfriend of 10 years. His medical history includes chronic obstructive pulmonary disease and prostate cancer. In 2015, he had a radical prostatectomy, without chemotherapy. His social history includes childhood neglect, which prompted him to leave home when he was a teenager. Mr. G had earned his general education development certificate and worked at a small retail store.
Mr. G had no previous history of mental health treatment per self-report, collateral information from his girlfriend, and chart review. He reported no known family psychiatric history. He did not endorse past psychiatric admissions or suicide attempts, nor previous periods of mania, depression, or psychosis. He said he used illicit substances as a teen, but denied using alcohol, tobacco products, or illicit substances in the past 20 years.
Mr. G recently had multiple ED visits and hospitalizations due to ongoing signs and symptoms associated with his COVID-19 diagnosis, primarily worsening shortness of breath and cough. Eleven days before EMS brought him to the ED at his girlfriend’s request, Mr. G had presented to the ED with chief complaints of shortness of breath and dry cough (Day 0). He reported that he had been “running a fever” for 2 days. In the ED, his initial vital signs were notable only for a temperature of 100.9°F (38.28°C). He was diagnosed with “acute viral syndrome” and received 1 dose of IV ceftriaxone, 2 g, and IV azithromycin, 500 mg. On Day 2, the ED clinicians prescribed a 4-day course of oral azithromycin, 250 mg/d, and discharged him home.
On Day 3, Mr. G returned to the ED with similar complaints—congestion and productive cough. He tested positive for COVID-19, and the ED discharged him home with quarantine instructions. Hours later, he returned to the ED via EMS with chief complaints of chest pain, diarrhea, and myalgias. He was prescribed a 5-day course ofoseltamivir, 75 mg twice daily, and azithromycin, 250 mg/d. The ED again discharged him home.
On Day 4, Mr. G returned to the ED for a fourth time. His chief complaint was worsening shortness of breath. His oxygen saturation was 94% on room air; it improved to 96% on 2 L of oxygen. His chest X-ray showed diffuse reticulonodular opacities throughout his bilateral lung fields and increased airspace opacification in the bilateral lower lobes. The ED admitted Mr. G to an internal medicine unit, where the primary treatment team enrolled him in a clinical trial. As part of the trial, Mr. G received hydroxychloroquine, 400 mg, on Day 4 and Day 5. The placebo-controlled component of the trial involved Mr. G receiving daily infusions of either remdesivir or placebo on Day 6 through Day 8. On Day 8, Mr. G was discharged home.
On Day 9, Mr. G returned to the ED with a chief complaint that his “thermometer wasn’t working” at home. The ED readmitted him to the internal medicine unit. On Day 9 through Day 11, Mr. G received daily doses of
Continue to: During the second hospitalization...
During the second hospitalization, nursing staff reported that Mr. G seemed religiously preoccupied and once reported seeing angels and demons. He was observed sitting in a chair praying to Allah that he would “come in on a horse to chop all the workers’ heads off.”
On Day 11, Mr. G was discharged home. Later that evening, the EMS brought him back in the ED due to his girlfriend’s concerns about his mental state.
EVALUATION Talks to God
On Day 12, psychiatry is consulted to evaluate Mr. G’s new-onset psychosis. Mr. G is alert and oriented to person, place, and time. His speech is loud, though the amount and rate are unremarkable. He displays no psychomotor agitation. His thought process is tangential and focuses on religious themes, specifically referring to Islam. He reports auditory hallucinations of God speaking directly to him. Mr. G states, “I am here because of a miraculous transformation from death back to life. Do you believe in God? Which God do you believe in? There are 2 Gods and only one of them is the true God. He is the God of all the 7 heavens and His true name is Allah, only one God, one faith. Allah is a ball of energy.”
Mr. G’s girlfriend provides collateral information that Mr. G had been raised Christian but was not religious as an adult. She says that he had never spoken about being Muslim. She adds that she had never known him to speak much about religion.
[polldaddy:10572249]
The authors’ observations
The etiology of new-onset psychosis can be related to several factors, including primary psychiatric illnesses, use of illicit substances, sequelae of general medical conditions, or adverse effects of prescribed medications. We considered each of these in the differential diagnosis for Mr. G.
Continue to: Psychiatric illness or illicit substance use
Psychiatric illness or illicit substance use. Because Mr. G was 56 years old and had no known psychiatric history or family psychiatric history, a primary psychiatric illness seemed less likely. Substance-induced psychosis related to illicit substance use also seemed unlikely because he denied using illicit substances, and an expanded urine drug screen was negative.

Psychosis due to a general medical condition. Results from Mr. G’s laboratory workup show marked elevation in multiple inflammation-related biomarkers (Table 1), consistent with the inflammatory profile seen with COVID-19 infection. However, results from several laboratory tests for potential etiologies of new-onset psychosis due to a general medical condition were negative (Table 2). Based on Mr. G’s history of prostate cancer, we considered the possibility of metastatic space-occupying lesions of the brain; however, Mr. G’s head CT showed no acute intracranial abnormalities. Another possible etiology we considered was COVID-19–induced encephalitis; however, Mr. G’s brain MRI with and without contrast showed no evidence of acute or chronic intracranial changes.

Medication-induced psychosis. After largely ruling out primary psychiatric illnesses, illicit substances, and sequelae of general medical conditions, we turned our attention to prescribed medications as a potential etiology of Mr. G’s new-onset psychosis. During his initial hospitalization, Mr. G had been prescribed 2 doses of hydroxychloroquine, 400 mg, to treat his diagnosis of COVID-19. Because none of the other medications he received were reported to have neuropsychiatric adverse effects, including psychosis, hydroxychloroquine-induced psychosis was therefore the primary team’s working diagnosis.
EVALUATION Request to leave AMA
On Day 13, Mr. G requests to leave the hospital against medical advice (AMA). Until this point, he had voluntarily remained in the hospital, which he repeatedly referred to as “Heaven.” When asked to describe his medical condition, Mr. G replies, “God told me my condition is far beyond man’s understanding.” He denies that he is positive for COVID-19. He states, “I am cured, and the real fight has just begun.”
At the recommendation of the psychiatry consultation-liaison (C-L) service, the primary treatment team determines that Mr. G does not have capacity to leave AMA. The team is concerned that because of his psychotic symptoms, Mr. G would be unable to understand and follow his quarantine instructions. He remains hospitalized on a medical hold.
Continue to: The authors' observations
The authors’ observations
One important consideration this case highlighted was potential third-party responsibility clinicians and hospital systems may face if they discharge a patient with a communicable illness who is unable to follow precautions based on a psychiatric condition.1 That concern was based on Mr. G’s reported desire to pursue missions “beyond man’s understanding,” which he felt compelled to complete, and which could unnecessarily place the public at risk. The psychiatry C-L service consulted the local health department and conferred with the hospital’s legal representatives, who agreed with the plan to keep Mr. G in the hospital for his safety as well as for the public’s safety.
TREATMENT Oral haloperidol
The psychiatry C-L service recommends initiating an antipsychotic. On Day 13, Mr. G starts oral haloperidol, 2.5 mg twice a day, to address his ongoing psychotic symptoms. On Day 14, the treatment team increases the dosage to 5 mg twice a day. Mr. G tolerates the haloperidol and gradually begins to improve. He demonstrates improved sleep, normal speech volume, less religious preoccupation, and a considerably improved understanding of his medical condition.
The authors’ observations
Mr. G’s initial psychiatric evaluation demonstrated an acute onset of psychotic symptoms, without evidence of delirium. Psychosis secondary to a general medical condition (such as COVID-19) and hydroxychloroquine-induced psychotic disorder topped our initial considerations in the differential diagnosis of this case. While the exact neuropsychiatric sequelae of COVID-19 are not yet clear, previous experiences with viral pandemics and case studies from the current pandemic demonstrate a wide variety of possible neuropsychiatric manifestations. Mood symptoms, psychosis, and encephalopathy represent some of the neuropsychiatric complications observed with past viral pandemics.2 Neuropsychiatric symptoms may be triggered by the virus itself, or from the host’s immune response to the infection.3 To further complicate matters, neuropsychiatric symptoms may manifest during the acute viral infection, or may surface later, as subacute or chronic neuropsychiatric illness.
Neuropsychiatric adverse events
Mr. G developed psychotic symptoms within the first few days of receiving hydroxychloroquine, which is consistent with the scant literature on this topic.8 Based on the available information, hydroxychloroquine remains the most likely etiology of his new-onset psychotic symptoms. Mr. G’s case is one example of the possible neuropsychiatric presentations clinicians may face while treating a novel viral illness.
Continue to: OUTCOME Homeward-bound
OUTCOME Homeward-bound
By Day 18, Mr. G’s psychotic symptoms have significantly improved. He is able to rationally process information about his COVID-19 diagnosis and the recommended quarantine instructions he needs to follow after discharge. He is cleared by infection control and discharged home to return to living with his girlfriend.
Mr. G attends his follow-up psychiatric appointment remotely 2 weeks after discharge. He reports that since discharge, he has continued taking his prescribed haloperidol, 5 mg twice a day. He demonstrates improved insight into his medical condition, acknowledging his COVID-19–positive status, and confirms that he has been following quarantine instructions. He does not report ongoing auditory or visual hallucinations, and is no longer religiously preoccupied. He says he is looking forward to being medically cleared to return to work.
The authors’ observations
This case highlights the need for prospective, longitudinal screening and monitoring of neuropsychiatric symptoms as part of the public health response to COVID-19. The case also highlights the importance of careful monitoring for adverse events, including neuropsychiatric symptoms, during clinical trials that involve experimental treatments. The long-term prognosis for individuals such as Mr. G who develop neuropsychiatric symptoms during acute COVID-19 infection remains unknown. Similarly, subacute and chronic neuropsychiatric manifestations that may develop after resolution of acute COVID-19 infection are unknown at this time. However, we can learn from past viral pandemics and anticipate that neuropsychiatric sequelae are likely to occur and should be part of the public health response to the pandemic.
Bottom Line
The coronavirus disease 2019 pandemic provides multiple clinical challenges pertinent to psychiatry. Neuropsychiatric symptoms may manifest from delirium, viral infection, host immune response, or adverse reactions to experimental treatments. These potential neuropsychiatric symptoms may complicate medical treatment. They can also raise important ethical and legal considerations, such as weighing patient autonomy vs third-party responsibility to the public at large.
Related Resources
- Ferrando SJ, Klepacz L, Lynch S, et al. COVID-19 psychosis: a potential new neuropsychiatric condition triggered by novel coronavirus infection and the inflammatory response? [published online May 19, 2020]. Psychosomatics. 2020. doi: 10.1016/j.psym.2020.05.012.
- Vlessides M. COVID-19 and psychosis: is there a link? Medscape Medical News. https://www.medscape.com/viewarticle/930224. Published May 8, 2020.
Drug Brand Names
Azithromycin • Zithromax
Ceftriaxone • Rocephin
Chloroquine • Aralen
Haloperidol • Haldol
Hydroxychloroquine • Plaquenil
Levofloxacin • Levaquin
Oseltamivir • Tamiflu
CASE Agitated, psychotic, and COVID-19–positive
Mr. G, age 56, is brought to the emergency department (ED) by emergency medical services (EMS) after his girlfriend reports that he was trying to climb into the “fiery furnace” to “burn the devil within him.” Mr. G had recently tested positive for coronavirus disease 2019 (COVID-19) via polymerase chain reaction and had been receiving treatment for it. In the ED, he is distressed and repeatedly exclaims, “The devil is alive!” He insists on covering himself with blankets, despite diaphoresis and soaking through his clothing within minutes. Because he does not respond to attempted redirection, the ED clinicians administer a single dose of IM haloperidol, 2 mg, for agitation.
HISTORY Multiple ED visits and hospitalizations
Mr. G, who has no known psychiatric history, lives with his girlfriend of 10 years. His medical history includes chronic obstructive pulmonary disease and prostate cancer. In 2015, he had a radical prostatectomy, without chemotherapy. His social history includes childhood neglect, which prompted him to leave home when he was a teenager. Mr. G had earned his general education development certificate and worked at a small retail store.
Mr. G had no previous history of mental health treatment per self-report, collateral information from his girlfriend, and chart review. He reported no known family psychiatric history. He did not endorse past psychiatric admissions or suicide attempts, nor previous periods of mania, depression, or psychosis. He said he used illicit substances as a teen, but denied using alcohol, tobacco products, or illicit substances in the past 20 years.
Mr. G recently had multiple ED visits and hospitalizations due to ongoing signs and symptoms associated with his COVID-19 diagnosis, primarily worsening shortness of breath and cough. Eleven days before EMS brought him to the ED at his girlfriend’s request, Mr. G had presented to the ED with chief complaints of shortness of breath and dry cough (Day 0). He reported that he had been “running a fever” for 2 days. In the ED, his initial vital signs were notable only for a temperature of 100.9°F (38.28°C). He was diagnosed with “acute viral syndrome” and received 1 dose of IV ceftriaxone, 2 g, and IV azithromycin, 500 mg. On Day 2, the ED clinicians prescribed a 4-day course of oral azithromycin, 250 mg/d, and discharged him home.
On Day 3, Mr. G returned to the ED with similar complaints—congestion and productive cough. He tested positive for COVID-19, and the ED discharged him home with quarantine instructions. Hours later, he returned to the ED via EMS with chief complaints of chest pain, diarrhea, and myalgias. He was prescribed a 5-day course ofoseltamivir, 75 mg twice daily, and azithromycin, 250 mg/d. The ED again discharged him home.
On Day 4, Mr. G returned to the ED for a fourth time. His chief complaint was worsening shortness of breath. His oxygen saturation was 94% on room air; it improved to 96% on 2 L of oxygen. His chest X-ray showed diffuse reticulonodular opacities throughout his bilateral lung fields and increased airspace opacification in the bilateral lower lobes. The ED admitted Mr. G to an internal medicine unit, where the primary treatment team enrolled him in a clinical trial. As part of the trial, Mr. G received hydroxychloroquine, 400 mg, on Day 4 and Day 5. The placebo-controlled component of the trial involved Mr. G receiving daily infusions of either remdesivir or placebo on Day 6 through Day 8. On Day 8, Mr. G was discharged home.
On Day 9, Mr. G returned to the ED with a chief complaint that his “thermometer wasn’t working” at home. The ED readmitted him to the internal medicine unit. On Day 9 through Day 11, Mr. G received daily doses of
Continue to: During the second hospitalization...
During the second hospitalization, nursing staff reported that Mr. G seemed religiously preoccupied and once reported seeing angels and demons. He was observed sitting in a chair praying to Allah that he would “come in on a horse to chop all the workers’ heads off.”
On Day 11, Mr. G was discharged home. Later that evening, the EMS brought him back in the ED due to his girlfriend’s concerns about his mental state.
EVALUATION Talks to God
On Day 12, psychiatry is consulted to evaluate Mr. G’s new-onset psychosis. Mr. G is alert and oriented to person, place, and time. His speech is loud, though the amount and rate are unremarkable. He displays no psychomotor agitation. His thought process is tangential and focuses on religious themes, specifically referring to Islam. He reports auditory hallucinations of God speaking directly to him. Mr. G states, “I am here because of a miraculous transformation from death back to life. Do you believe in God? Which God do you believe in? There are 2 Gods and only one of them is the true God. He is the God of all the 7 heavens and His true name is Allah, only one God, one faith. Allah is a ball of energy.”
Mr. G’s girlfriend provides collateral information that Mr. G had been raised Christian but was not religious as an adult. She says that he had never spoken about being Muslim. She adds that she had never known him to speak much about religion.
[polldaddy:10572249]
The authors’ observations
The etiology of new-onset psychosis can be related to several factors, including primary psychiatric illnesses, use of illicit substances, sequelae of general medical conditions, or adverse effects of prescribed medications. We considered each of these in the differential diagnosis for Mr. G.
Continue to: Psychiatric illness or illicit substance use
Psychiatric illness or illicit substance use. Because Mr. G was 56 years old and had no known psychiatric history or family psychiatric history, a primary psychiatric illness seemed less likely. Substance-induced psychosis related to illicit substance use also seemed unlikely because he denied using illicit substances, and an expanded urine drug screen was negative.

Psychosis due to a general medical condition. Results from Mr. G’s laboratory workup show marked elevation in multiple inflammation-related biomarkers (Table 1), consistent with the inflammatory profile seen with COVID-19 infection. However, results from several laboratory tests for potential etiologies of new-onset psychosis due to a general medical condition were negative (Table 2). Based on Mr. G’s history of prostate cancer, we considered the possibility of metastatic space-occupying lesions of the brain; however, Mr. G’s head CT showed no acute intracranial abnormalities. Another possible etiology we considered was COVID-19–induced encephalitis; however, Mr. G’s brain MRI with and without contrast showed no evidence of acute or chronic intracranial changes.

Medication-induced psychosis. After largely ruling out primary psychiatric illnesses, illicit substances, and sequelae of general medical conditions, we turned our attention to prescribed medications as a potential etiology of Mr. G’s new-onset psychosis. During his initial hospitalization, Mr. G had been prescribed 2 doses of hydroxychloroquine, 400 mg, to treat his diagnosis of COVID-19. Because none of the other medications he received were reported to have neuropsychiatric adverse effects, including psychosis, hydroxychloroquine-induced psychosis was therefore the primary team’s working diagnosis.
EVALUATION Request to leave AMA
On Day 13, Mr. G requests to leave the hospital against medical advice (AMA). Until this point, he had voluntarily remained in the hospital, which he repeatedly referred to as “Heaven.” When asked to describe his medical condition, Mr. G replies, “God told me my condition is far beyond man’s understanding.” He denies that he is positive for COVID-19. He states, “I am cured, and the real fight has just begun.”
At the recommendation of the psychiatry consultation-liaison (C-L) service, the primary treatment team determines that Mr. G does not have capacity to leave AMA. The team is concerned that because of his psychotic symptoms, Mr. G would be unable to understand and follow his quarantine instructions. He remains hospitalized on a medical hold.
Continue to: The authors' observations
The authors’ observations
One important consideration this case highlighted was potential third-party responsibility clinicians and hospital systems may face if they discharge a patient with a communicable illness who is unable to follow precautions based on a psychiatric condition.1 That concern was based on Mr. G’s reported desire to pursue missions “beyond man’s understanding,” which he felt compelled to complete, and which could unnecessarily place the public at risk. The psychiatry C-L service consulted the local health department and conferred with the hospital’s legal representatives, who agreed with the plan to keep Mr. G in the hospital for his safety as well as for the public’s safety.
TREATMENT Oral haloperidol
The psychiatry C-L service recommends initiating an antipsychotic. On Day 13, Mr. G starts oral haloperidol, 2.5 mg twice a day, to address his ongoing psychotic symptoms. On Day 14, the treatment team increases the dosage to 5 mg twice a day. Mr. G tolerates the haloperidol and gradually begins to improve. He demonstrates improved sleep, normal speech volume, less religious preoccupation, and a considerably improved understanding of his medical condition.
The authors’ observations
Mr. G’s initial psychiatric evaluation demonstrated an acute onset of psychotic symptoms, without evidence of delirium. Psychosis secondary to a general medical condition (such as COVID-19) and hydroxychloroquine-induced psychotic disorder topped our initial considerations in the differential diagnosis of this case. While the exact neuropsychiatric sequelae of COVID-19 are not yet clear, previous experiences with viral pandemics and case studies from the current pandemic demonstrate a wide variety of possible neuropsychiatric manifestations. Mood symptoms, psychosis, and encephalopathy represent some of the neuropsychiatric complications observed with past viral pandemics.2 Neuropsychiatric symptoms may be triggered by the virus itself, or from the host’s immune response to the infection.3 To further complicate matters, neuropsychiatric symptoms may manifest during the acute viral infection, or may surface later, as subacute or chronic neuropsychiatric illness.
Neuropsychiatric adverse events
Mr. G developed psychotic symptoms within the first few days of receiving hydroxychloroquine, which is consistent with the scant literature on this topic.8 Based on the available information, hydroxychloroquine remains the most likely etiology of his new-onset psychotic symptoms. Mr. G’s case is one example of the possible neuropsychiatric presentations clinicians may face while treating a novel viral illness.
Continue to: OUTCOME Homeward-bound
OUTCOME Homeward-bound
By Day 18, Mr. G’s psychotic symptoms have significantly improved. He is able to rationally process information about his COVID-19 diagnosis and the recommended quarantine instructions he needs to follow after discharge. He is cleared by infection control and discharged home to return to living with his girlfriend.
Mr. G attends his follow-up psychiatric appointment remotely 2 weeks after discharge. He reports that since discharge, he has continued taking his prescribed haloperidol, 5 mg twice a day. He demonstrates improved insight into his medical condition, acknowledging his COVID-19–positive status, and confirms that he has been following quarantine instructions. He does not report ongoing auditory or visual hallucinations, and is no longer religiously preoccupied. He says he is looking forward to being medically cleared to return to work.
The authors’ observations
This case highlights the need for prospective, longitudinal screening and monitoring of neuropsychiatric symptoms as part of the public health response to COVID-19. The case also highlights the importance of careful monitoring for adverse events, including neuropsychiatric symptoms, during clinical trials that involve experimental treatments. The long-term prognosis for individuals such as Mr. G who develop neuropsychiatric symptoms during acute COVID-19 infection remains unknown. Similarly, subacute and chronic neuropsychiatric manifestations that may develop after resolution of acute COVID-19 infection are unknown at this time. However, we can learn from past viral pandemics and anticipate that neuropsychiatric sequelae are likely to occur and should be part of the public health response to the pandemic.
Bottom Line
The coronavirus disease 2019 pandemic provides multiple clinical challenges pertinent to psychiatry. Neuropsychiatric symptoms may manifest from delirium, viral infection, host immune response, or adverse reactions to experimental treatments. These potential neuropsychiatric symptoms may complicate medical treatment. They can also raise important ethical and legal considerations, such as weighing patient autonomy vs third-party responsibility to the public at large.
Related Resources
- Ferrando SJ, Klepacz L, Lynch S, et al. COVID-19 psychosis: a potential new neuropsychiatric condition triggered by novel coronavirus infection and the inflammatory response? [published online May 19, 2020]. Psychosomatics. 2020. doi: 10.1016/j.psym.2020.05.012.
- Vlessides M. COVID-19 and psychosis: is there a link? Medscape Medical News. https://www.medscape.com/viewarticle/930224. Published May 8, 2020.
Drug Brand Names
Azithromycin • Zithromax
Ceftriaxone • Rocephin
Chloroquine • Aralen
Haloperidol • Haldol
Hydroxychloroquine • Plaquenil
Levofloxacin • Levaquin
Oseltamivir • Tamiflu
1. Ghossoub E, Newman WJ. COVID-19 and the duty to protect from communicable diseases. [published online ahead of print, May 8, 2020]. J Am Acad Psychiatry Law.
2. Menninger Ka. Psychoses associated with influenza: I. general data: statistical analysis. JAMA. 1919;72(4):235-241.
3. Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain, Behavior, and Immunity. 2020. doi:10.1016/j.bbi.2020.04.027.
4. Alkadi HO. Antimalarial drug toxicity: a review. Chemotherapy. 2007;53(6):385-391.
5. Bogaczewicz A, Sobów T. Psychiatric adverse effects of chloroquine. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-114.
6. Sato K, Mano T, Iwata A, et al. Neuropsychiatric adverse events of chloroquine: a real-world pharmacovigilance study using the FDA Adverse Event Reporting System (FAERS) database. Biosci Trends. 2020;14(2):139-143.
7. Cortegiani A, Ingoglia G, Ippolito M, et al. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care. 2020;57:279-283.
8. Das P, Rai A, Chopra A, et al. Psychosis likely induced by hydroxychloroquine in a patient with chronic Q fever: a case report and clinically relevant review of pharmacology. Psychosomatics. 2014;55(4):409-413.
1. Ghossoub E, Newman WJ. COVID-19 and the duty to protect from communicable diseases. [published online ahead of print, May 8, 2020]. J Am Acad Psychiatry Law.
2. Menninger Ka. Psychoses associated with influenza: I. general data: statistical analysis. JAMA. 1919;72(4):235-241.
3. Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain, Behavior, and Immunity. 2020. doi:10.1016/j.bbi.2020.04.027.
4. Alkadi HO. Antimalarial drug toxicity: a review. Chemotherapy. 2007;53(6):385-391.
5. Bogaczewicz A, Sobów T. Psychiatric adverse effects of chloroquine. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-114.
6. Sato K, Mano T, Iwata A, et al. Neuropsychiatric adverse events of chloroquine: a real-world pharmacovigilance study using the FDA Adverse Event Reporting System (FAERS) database. Biosci Trends. 2020;14(2):139-143.
7. Cortegiani A, Ingoglia G, Ippolito M, et al. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care. 2020;57:279-283.
8. Das P, Rai A, Chopra A, et al. Psychosis likely induced by hydroxychloroquine in a patient with chronic Q fever: a case report and clinically relevant review of pharmacology. Psychosomatics. 2014;55(4):409-413.
Potential new biomarker for psychosis severity
ACE levels are lower in individuals with first episode psychosis (FEP) and even lower in those with resistant disease, suggesting the enzyme may be a biomarker of disease severity.
In a longitudinal cohort study, investigators found patients with FEP had significantly reduced ACE levels compared with their healthy peers.
With blood concentrations of the enzyme significantly reduced in those who were treatment-resistant, and results suggest “a possible relationship with disease severity,” noted the researchers, led by investigator Luisa Longo, MD, a resident in psychiatry, University of Bari Aldo Moro in Italy.
Moreover, the finding that lower ACE levels were associated with greater cognitive impairment on neuropsychological tests indicates the enzyme plays a role in “the alteration of neurocognitive abilities” in patients with FEP.
Taken together, the results “highlight ACE as a promising peripheral biomarker to identify patients at risk of treatment resistance to antipsychotics,” the investigators reported.
The findings were presented at the annual congress of the Schizophrenia International Research Society.
Mechanisms “poorly understood”
Previous studies suggest ACE may play a role in neurologic and psychiatric conditions, including schizophrenia, through alterations in function or blood concentrations.
However, the molecular mechanisms underlying disease onset and response to antipsychotics in patients with FEP “remain poorly understood,” the researchers noted. In addition, “despite adequate antipsychotic treatment, 20% of patients have persistent symptoms.”
To determine whether ACE levels are already altered in FEP patients, the investigators examined data on 138 patients with FEP and 115 healthy controls.
After measuring blood concentrations in 122 of the patients and 78 controls, they found that ACE levels were significantly lower in FEP patients (P = 4.1x10-13) after controlling for age, sex, ethnicity, duration of illness, smoking status, and chlorpromazine equivalents.
Cerebrospinal fluid (CSF) levels, which were measured in 19 patients and 18 controls, were also significantly lower in individuals with FEP (P = .01), with a strong correlation observed between blood and CSF levels (P = .0005).
Next, the team used Treatment Response and Resistance in Psychosis criteria to compare ACE levels in 32 treatment-resistant patients and 106 non–treatment-resistant patients. Results showed that ACE blood levels were significantly lower in the treatment-resistant patients (P = .03).
Finally, the association between ACE blood levels and range of clinical and neurocognitive variables were examined across all FEP patients.
The Scale for the Assessment of Negative and Positive Symptoms was administered, along with a battery of tests looking at processing speed, working memory, verbal learning and memory, visual learning and memory, ideational fluency, and executive function. All were combined into a composite score.
While there was no association between ACE blood levels and symptom severity in the patients, there was a significant association between levels and verbal memory (P = .007) and composite cognitive score (P = .04).
Notable finding
In an interview, Thomas W. Sedlak, MD, PhD, assistant professor of psychiatry and behavioral health, Johns Hopkins University, Baltimore, said the finding that ACE levels were associated with lower cognition scores is “notable in that we don’t really have any treatments for cognition.
“Antipsychotic drugs treat more of the famous symptoms of hallucinations and delusions and disordered speech, but they don›t really help cognition a whole lot — and too much medicine might even make that worse,” said Dr. Sedlak, who was not involved with the research.
“So it’s interesting to have another biomarker that might relate a symptom of schizophrenia that we don’t really have a good grip on addressing,” he said.
However, he noted that this study is “preliminary,” has not looked at longitudinal changes in ACE in the same patients, and is one of “many” correlation studies.
“I like to say you can pretty much Google any chemical in the body and somebody has a claim about it and schizophrenia,” Dr. Sedlak said.
Nevertheless, the current analysis is “convergent” with past studies on ACE, including those that have found associations between polymorphisms in the enzyme and schizophrenia, he noted.
Moreover, research has shown that ACE facilitates glutamate transmission, specifically via N-methyl-D-aspartate (NMDA) receptors in the prefrontal cortex, “which is probably the brain region most tied to schizophrenia abnormalities,” Dr. Sedlak said.
No tie to inflammation
The findings do contrast, however, with the increasing body of evidence linking schizophrenia to inflammation.
Dr. Sedlak said that is not surprising, as the more that is learned about schizophrenia, “the less it is likely to be a single homogeneous entity but something more akin to intellectual disability.
“We’re finding that there are different subtypes of schizophrenia, some of which might have more of an inflammatory tie and others which don’t.”
He added that it might be possible over the longer term to cluster patients by ACE levels and other biomarkers and then follow them long term to “see if we can predict outcomes better than purely clinical terms.”
In this way, Dr. Sedlak said he believes that psychiatry may become more like rheumatology, “which I’d say is the area of medicine, at least in terms of making diagnoses, most similar.”
The etiology of conditions such as lupus and rheumatoid arthritis is “not absolutely known” and there is “probably no single cause,” with the diagnosis relying on clinical findings alongside biomarker tests.
“I think psychiatry is, long term, hoping to discover some biomarkers ... that would be used together with the clinical findings to help come to more clinical agreement on how to define cases – and would be predictive long term in terms of the course of illness,” Dr. Sedlak concluded.
The study had no specific funding. The study investigators and Dr. Sedlak have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
ACE levels are lower in individuals with first episode psychosis (FEP) and even lower in those with resistant disease, suggesting the enzyme may be a biomarker of disease severity.
In a longitudinal cohort study, investigators found patients with FEP had significantly reduced ACE levels compared with their healthy peers.
With blood concentrations of the enzyme significantly reduced in those who were treatment-resistant, and results suggest “a possible relationship with disease severity,” noted the researchers, led by investigator Luisa Longo, MD, a resident in psychiatry, University of Bari Aldo Moro in Italy.
Moreover, the finding that lower ACE levels were associated with greater cognitive impairment on neuropsychological tests indicates the enzyme plays a role in “the alteration of neurocognitive abilities” in patients with FEP.
Taken together, the results “highlight ACE as a promising peripheral biomarker to identify patients at risk of treatment resistance to antipsychotics,” the investigators reported.
The findings were presented at the annual congress of the Schizophrenia International Research Society.
Mechanisms “poorly understood”
Previous studies suggest ACE may play a role in neurologic and psychiatric conditions, including schizophrenia, through alterations in function or blood concentrations.
However, the molecular mechanisms underlying disease onset and response to antipsychotics in patients with FEP “remain poorly understood,” the researchers noted. In addition, “despite adequate antipsychotic treatment, 20% of patients have persistent symptoms.”
To determine whether ACE levels are already altered in FEP patients, the investigators examined data on 138 patients with FEP and 115 healthy controls.
After measuring blood concentrations in 122 of the patients and 78 controls, they found that ACE levels were significantly lower in FEP patients (P = 4.1x10-13) after controlling for age, sex, ethnicity, duration of illness, smoking status, and chlorpromazine equivalents.
Cerebrospinal fluid (CSF) levels, which were measured in 19 patients and 18 controls, were also significantly lower in individuals with FEP (P = .01), with a strong correlation observed between blood and CSF levels (P = .0005).
Next, the team used Treatment Response and Resistance in Psychosis criteria to compare ACE levels in 32 treatment-resistant patients and 106 non–treatment-resistant patients. Results showed that ACE blood levels were significantly lower in the treatment-resistant patients (P = .03).
Finally, the association between ACE blood levels and range of clinical and neurocognitive variables were examined across all FEP patients.
The Scale for the Assessment of Negative and Positive Symptoms was administered, along with a battery of tests looking at processing speed, working memory, verbal learning and memory, visual learning and memory, ideational fluency, and executive function. All were combined into a composite score.
While there was no association between ACE blood levels and symptom severity in the patients, there was a significant association between levels and verbal memory (P = .007) and composite cognitive score (P = .04).
Notable finding
In an interview, Thomas W. Sedlak, MD, PhD, assistant professor of psychiatry and behavioral health, Johns Hopkins University, Baltimore, said the finding that ACE levels were associated with lower cognition scores is “notable in that we don’t really have any treatments for cognition.
“Antipsychotic drugs treat more of the famous symptoms of hallucinations and delusions and disordered speech, but they don›t really help cognition a whole lot — and too much medicine might even make that worse,” said Dr. Sedlak, who was not involved with the research.
“So it’s interesting to have another biomarker that might relate a symptom of schizophrenia that we don’t really have a good grip on addressing,” he said.
However, he noted that this study is “preliminary,” has not looked at longitudinal changes in ACE in the same patients, and is one of “many” correlation studies.
“I like to say you can pretty much Google any chemical in the body and somebody has a claim about it and schizophrenia,” Dr. Sedlak said.
Nevertheless, the current analysis is “convergent” with past studies on ACE, including those that have found associations between polymorphisms in the enzyme and schizophrenia, he noted.
Moreover, research has shown that ACE facilitates glutamate transmission, specifically via N-methyl-D-aspartate (NMDA) receptors in the prefrontal cortex, “which is probably the brain region most tied to schizophrenia abnormalities,” Dr. Sedlak said.
No tie to inflammation
The findings do contrast, however, with the increasing body of evidence linking schizophrenia to inflammation.
Dr. Sedlak said that is not surprising, as the more that is learned about schizophrenia, “the less it is likely to be a single homogeneous entity but something more akin to intellectual disability.
“We’re finding that there are different subtypes of schizophrenia, some of which might have more of an inflammatory tie and others which don’t.”
He added that it might be possible over the longer term to cluster patients by ACE levels and other biomarkers and then follow them long term to “see if we can predict outcomes better than purely clinical terms.”
In this way, Dr. Sedlak said he believes that psychiatry may become more like rheumatology, “which I’d say is the area of medicine, at least in terms of making diagnoses, most similar.”
The etiology of conditions such as lupus and rheumatoid arthritis is “not absolutely known” and there is “probably no single cause,” with the diagnosis relying on clinical findings alongside biomarker tests.
“I think psychiatry is, long term, hoping to discover some biomarkers ... that would be used together with the clinical findings to help come to more clinical agreement on how to define cases – and would be predictive long term in terms of the course of illness,” Dr. Sedlak concluded.
The study had no specific funding. The study investigators and Dr. Sedlak have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
ACE levels are lower in individuals with first episode psychosis (FEP) and even lower in those with resistant disease, suggesting the enzyme may be a biomarker of disease severity.
In a longitudinal cohort study, investigators found patients with FEP had significantly reduced ACE levels compared with their healthy peers.
With blood concentrations of the enzyme significantly reduced in those who were treatment-resistant, and results suggest “a possible relationship with disease severity,” noted the researchers, led by investigator Luisa Longo, MD, a resident in psychiatry, University of Bari Aldo Moro in Italy.
Moreover, the finding that lower ACE levels were associated with greater cognitive impairment on neuropsychological tests indicates the enzyme plays a role in “the alteration of neurocognitive abilities” in patients with FEP.
Taken together, the results “highlight ACE as a promising peripheral biomarker to identify patients at risk of treatment resistance to antipsychotics,” the investigators reported.
The findings were presented at the annual congress of the Schizophrenia International Research Society.
Mechanisms “poorly understood”
Previous studies suggest ACE may play a role in neurologic and psychiatric conditions, including schizophrenia, through alterations in function or blood concentrations.
However, the molecular mechanisms underlying disease onset and response to antipsychotics in patients with FEP “remain poorly understood,” the researchers noted. In addition, “despite adequate antipsychotic treatment, 20% of patients have persistent symptoms.”
To determine whether ACE levels are already altered in FEP patients, the investigators examined data on 138 patients with FEP and 115 healthy controls.
After measuring blood concentrations in 122 of the patients and 78 controls, they found that ACE levels were significantly lower in FEP patients (P = 4.1x10-13) after controlling for age, sex, ethnicity, duration of illness, smoking status, and chlorpromazine equivalents.
Cerebrospinal fluid (CSF) levels, which were measured in 19 patients and 18 controls, were also significantly lower in individuals with FEP (P = .01), with a strong correlation observed between blood and CSF levels (P = .0005).
Next, the team used Treatment Response and Resistance in Psychosis criteria to compare ACE levels in 32 treatment-resistant patients and 106 non–treatment-resistant patients. Results showed that ACE blood levels were significantly lower in the treatment-resistant patients (P = .03).
Finally, the association between ACE blood levels and range of clinical and neurocognitive variables were examined across all FEP patients.
The Scale for the Assessment of Negative and Positive Symptoms was administered, along with a battery of tests looking at processing speed, working memory, verbal learning and memory, visual learning and memory, ideational fluency, and executive function. All were combined into a composite score.
While there was no association between ACE blood levels and symptom severity in the patients, there was a significant association between levels and verbal memory (P = .007) and composite cognitive score (P = .04).
Notable finding
In an interview, Thomas W. Sedlak, MD, PhD, assistant professor of psychiatry and behavioral health, Johns Hopkins University, Baltimore, said the finding that ACE levels were associated with lower cognition scores is “notable in that we don’t really have any treatments for cognition.
“Antipsychotic drugs treat more of the famous symptoms of hallucinations and delusions and disordered speech, but they don›t really help cognition a whole lot — and too much medicine might even make that worse,” said Dr. Sedlak, who was not involved with the research.
“So it’s interesting to have another biomarker that might relate a symptom of schizophrenia that we don’t really have a good grip on addressing,” he said.
However, he noted that this study is “preliminary,” has not looked at longitudinal changes in ACE in the same patients, and is one of “many” correlation studies.
“I like to say you can pretty much Google any chemical in the body and somebody has a claim about it and schizophrenia,” Dr. Sedlak said.
Nevertheless, the current analysis is “convergent” with past studies on ACE, including those that have found associations between polymorphisms in the enzyme and schizophrenia, he noted.
Moreover, research has shown that ACE facilitates glutamate transmission, specifically via N-methyl-D-aspartate (NMDA) receptors in the prefrontal cortex, “which is probably the brain region most tied to schizophrenia abnormalities,” Dr. Sedlak said.
No tie to inflammation
The findings do contrast, however, with the increasing body of evidence linking schizophrenia to inflammation.
Dr. Sedlak said that is not surprising, as the more that is learned about schizophrenia, “the less it is likely to be a single homogeneous entity but something more akin to intellectual disability.
“We’re finding that there are different subtypes of schizophrenia, some of which might have more of an inflammatory tie and others which don’t.”
He added that it might be possible over the longer term to cluster patients by ACE levels and other biomarkers and then follow them long term to “see if we can predict outcomes better than purely clinical terms.”
In this way, Dr. Sedlak said he believes that psychiatry may become more like rheumatology, “which I’d say is the area of medicine, at least in terms of making diagnoses, most similar.”
The etiology of conditions such as lupus and rheumatoid arthritis is “not absolutely known” and there is “probably no single cause,” with the diagnosis relying on clinical findings alongside biomarker tests.
“I think psychiatry is, long term, hoping to discover some biomarkers ... that would be used together with the clinical findings to help come to more clinical agreement on how to define cases – and would be predictive long term in terms of the course of illness,” Dr. Sedlak concluded.
The study had no specific funding. The study investigators and Dr. Sedlak have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM SIRS 2020
Injectable vs. oral antipsychotics: Which do patients prefer?
Patients with schizophrenia appear to prefer long-acting injectable (LAI) antipsychotics, compared with oral versions of these medications, primarily because injectables are more convenient and give individuals more control over their lives, new research shows.
Patients also prefer injections once every 3 months to monthly injections, citing the need for fewer doctor visits and less pain as key reasons. They also reported a preference for deltoid versus gluteal injections, as they were faster and easier to administer, and less embarrassing.
Study investigator Srihari Gopal, MD, senior director at Janssen Research and Development in Titusville, N.J., said in an interview that stigma, which is a “is a really powerful force in mental health treatment,” underlies these findings in terms of the disease itself and its management.
“It’s one of the [key] reasons that schizophrenia patients decide to abandon their drugs and not go to the doctor,” he added.
The study was scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
Outdated perceptions
The investigators noted that there is limited information on patient preference with regard to LAI versus oral antipsychotics in the management of schizophrenia.
They also noted that LAIs have been shown to reduce the risk of relapse and rehospitalization because of treatment discontinuation and may help to improve to medication adherence.
However, these medications are still underutilized in clinical practice. Dr. Gopal estimated that only around 1 in 10 patients with schizophrenia in the United States take an LAI, although that figure varies considerably at a global level and is as high as 1 in 2 in Spain.
This is the result of a number of factors that act as potential barriers to LAI use, not the least of which is misconceptions among caregiver and health care professionals about the drugs.
“When I first was in medical school, this was in the 1990s ... there were really only first-generation antipsychotics available in a depot or a long-acting form, and those had very severe side effects,” said Dr. Gopal.
“They would tend to cause all sorts of movement disorders and would make patients feel really drowsy throughout the day, so they really hated taking them,” he said, noting that these depot medications were oil based, which was painful on injection and caused reactions.
While the newer generations of LAIs are water based and have a much-improved adverse effect profile, doctors “on my end of the age spectrum have all those negative connotations and memories in their minds about what these older LAIs were like, ” Dr. Gopal said
“It’s only the newer generation of doctors who were not around at the time that have a more forward-thinking attitude about the newer long-actings.”
Differences by country
To assess factors that determine patients’ medication preferences in order to better understand expectations and reduce potential barriers to treatment, the researchers analyzed data on 1,429 patients with schizophrenia who were participants in a double-blind, randomized, noninferiority study of paliperidone palmitate taken monthly versus once every 3 months.
Participants had a mean age of 38.4 years, and 55% were men. The majority (54%) were white, 8% were black or African American, and 38% were from other races. About one-eighth (12%) of the patients were from the United States.
The highest preference for LAIs was in Europe, at 88%, vs 59.1% in the United States and 70.7% in the rest of the world.
Interestingly, the preference for LAIs in the United States was comparable across different races, at 59.6% among black patients, 58.8% among whites, and 57.1% for other races.
All study participants had a confirmed diagnosis of schizophrenia and a Positive and Negative Syndrome Scale total score of between 70 and 120 at baseline, with worsening symptoms.
They completed the Medication Preference Questionnaire on day 1, day 120, and at the end of the study, with the current analysis focusing on day 1 responses, as that was the only time when patients would not have received any study medication.
Patient empowerment key
The most common reason patients cited for preferring LAIs over oral antipsychotics were that they felt healthier (57%), could get back to their favorite activities (56%), and didn’t have to think about taking their medication (54%).
In terms of their personal experiences, patients preferred LAIs to pills because they “are easier for me” (67% vs. 18%) and offered a greater sense of control and relieved them from having to think about taking medication (64% vs. 14%).
Finally, 50% of patients preferred LAI injections once every 3 months versus 38% for monthly and 3% for daily injections. Main reason cited were fewer injections (96%), less pain (84%), and fewer doctor visits (80%).
The preferred site for LAI injection was deltoid muscle over gluteal muscle, at 59%, with faster administration (63%), easier use (51%), and the location being less embarrassing (44%) cited as the primary reasons.
“Overall, patient empowerment and quality of life–related goals were important for patients who preferred LAI antipsychotics,” the investigators noted.
Logistic regression analysis indicated that only race and country were significantly associated with medication preferences, with white patients significantly more likely than others to prefer LAIs versus oral medications (adjusted odds ratio, 2.39; P < .001). U.S. patients were significantly less likely to prefer the drugs than those from other countries (aOR, 0.41; P < .001).
Dr. Gopal added that significant differences in patient preference for LAIs likely have a lot to do with the prevailing attitudes of doctors from different countries, with low LAI use corresponding to “more negative attitudes.”
“Better understanding of patients’ treatment priorities and perspective could help overcome barriers to LAI use and inform best course of personalized schizophrenia treatment for improved patient satisfaction and medication adherence,” the investigators noted.
Approached for comment, Matej Markota, MD, a psychiatrist at the Mayo Clinic in Rochester, Minn., who was not involved with the research, said that he agreed with the findings of the study.
He said in an interview that, in his clinical experience, the convenience of not having to take medications daily is an important factor that drives patient preference for LAI use over oral medications.
The study was funded by Janssen Research & Development. Dr. Gopal reports he is an employee of Janssen Research & Developmentand owns stock/equity in Johnson & Johnson. Dr. Markota has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with schizophrenia appear to prefer long-acting injectable (LAI) antipsychotics, compared with oral versions of these medications, primarily because injectables are more convenient and give individuals more control over their lives, new research shows.
Patients also prefer injections once every 3 months to monthly injections, citing the need for fewer doctor visits and less pain as key reasons. They also reported a preference for deltoid versus gluteal injections, as they were faster and easier to administer, and less embarrassing.
Study investigator Srihari Gopal, MD, senior director at Janssen Research and Development in Titusville, N.J., said in an interview that stigma, which is a “is a really powerful force in mental health treatment,” underlies these findings in terms of the disease itself and its management.
“It’s one of the [key] reasons that schizophrenia patients decide to abandon their drugs and not go to the doctor,” he added.
The study was scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
Outdated perceptions
The investigators noted that there is limited information on patient preference with regard to LAI versus oral antipsychotics in the management of schizophrenia.
They also noted that LAIs have been shown to reduce the risk of relapse and rehospitalization because of treatment discontinuation and may help to improve to medication adherence.
However, these medications are still underutilized in clinical practice. Dr. Gopal estimated that only around 1 in 10 patients with schizophrenia in the United States take an LAI, although that figure varies considerably at a global level and is as high as 1 in 2 in Spain.
This is the result of a number of factors that act as potential barriers to LAI use, not the least of which is misconceptions among caregiver and health care professionals about the drugs.
“When I first was in medical school, this was in the 1990s ... there were really only first-generation antipsychotics available in a depot or a long-acting form, and those had very severe side effects,” said Dr. Gopal.
“They would tend to cause all sorts of movement disorders and would make patients feel really drowsy throughout the day, so they really hated taking them,” he said, noting that these depot medications were oil based, which was painful on injection and caused reactions.
While the newer generations of LAIs are water based and have a much-improved adverse effect profile, doctors “on my end of the age spectrum have all those negative connotations and memories in their minds about what these older LAIs were like, ” Dr. Gopal said
“It’s only the newer generation of doctors who were not around at the time that have a more forward-thinking attitude about the newer long-actings.”
Differences by country
To assess factors that determine patients’ medication preferences in order to better understand expectations and reduce potential barriers to treatment, the researchers analyzed data on 1,429 patients with schizophrenia who were participants in a double-blind, randomized, noninferiority study of paliperidone palmitate taken monthly versus once every 3 months.
Participants had a mean age of 38.4 years, and 55% were men. The majority (54%) were white, 8% were black or African American, and 38% were from other races. About one-eighth (12%) of the patients were from the United States.
The highest preference for LAIs was in Europe, at 88%, vs 59.1% in the United States and 70.7% in the rest of the world.
Interestingly, the preference for LAIs in the United States was comparable across different races, at 59.6% among black patients, 58.8% among whites, and 57.1% for other races.
All study participants had a confirmed diagnosis of schizophrenia and a Positive and Negative Syndrome Scale total score of between 70 and 120 at baseline, with worsening symptoms.
They completed the Medication Preference Questionnaire on day 1, day 120, and at the end of the study, with the current analysis focusing on day 1 responses, as that was the only time when patients would not have received any study medication.
Patient empowerment key
The most common reason patients cited for preferring LAIs over oral antipsychotics were that they felt healthier (57%), could get back to their favorite activities (56%), and didn’t have to think about taking their medication (54%).
In terms of their personal experiences, patients preferred LAIs to pills because they “are easier for me” (67% vs. 18%) and offered a greater sense of control and relieved them from having to think about taking medication (64% vs. 14%).
Finally, 50% of patients preferred LAI injections once every 3 months versus 38% for monthly and 3% for daily injections. Main reason cited were fewer injections (96%), less pain (84%), and fewer doctor visits (80%).
The preferred site for LAI injection was deltoid muscle over gluteal muscle, at 59%, with faster administration (63%), easier use (51%), and the location being less embarrassing (44%) cited as the primary reasons.
“Overall, patient empowerment and quality of life–related goals were important for patients who preferred LAI antipsychotics,” the investigators noted.
Logistic regression analysis indicated that only race and country were significantly associated with medication preferences, with white patients significantly more likely than others to prefer LAIs versus oral medications (adjusted odds ratio, 2.39; P < .001). U.S. patients were significantly less likely to prefer the drugs than those from other countries (aOR, 0.41; P < .001).
Dr. Gopal added that significant differences in patient preference for LAIs likely have a lot to do with the prevailing attitudes of doctors from different countries, with low LAI use corresponding to “more negative attitudes.”
“Better understanding of patients’ treatment priorities and perspective could help overcome barriers to LAI use and inform best course of personalized schizophrenia treatment for improved patient satisfaction and medication adherence,” the investigators noted.
Approached for comment, Matej Markota, MD, a psychiatrist at the Mayo Clinic in Rochester, Minn., who was not involved with the research, said that he agreed with the findings of the study.
He said in an interview that, in his clinical experience, the convenience of not having to take medications daily is an important factor that drives patient preference for LAI use over oral medications.
The study was funded by Janssen Research & Development. Dr. Gopal reports he is an employee of Janssen Research & Developmentand owns stock/equity in Johnson & Johnson. Dr. Markota has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with schizophrenia appear to prefer long-acting injectable (LAI) antipsychotics, compared with oral versions of these medications, primarily because injectables are more convenient and give individuals more control over their lives, new research shows.
Patients also prefer injections once every 3 months to monthly injections, citing the need for fewer doctor visits and less pain as key reasons. They also reported a preference for deltoid versus gluteal injections, as they were faster and easier to administer, and less embarrassing.
Study investigator Srihari Gopal, MD, senior director at Janssen Research and Development in Titusville, N.J., said in an interview that stigma, which is a “is a really powerful force in mental health treatment,” underlies these findings in terms of the disease itself and its management.
“It’s one of the [key] reasons that schizophrenia patients decide to abandon their drugs and not go to the doctor,” he added.
The study was scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
Outdated perceptions
The investigators noted that there is limited information on patient preference with regard to LAI versus oral antipsychotics in the management of schizophrenia.
They also noted that LAIs have been shown to reduce the risk of relapse and rehospitalization because of treatment discontinuation and may help to improve to medication adherence.
However, these medications are still underutilized in clinical practice. Dr. Gopal estimated that only around 1 in 10 patients with schizophrenia in the United States take an LAI, although that figure varies considerably at a global level and is as high as 1 in 2 in Spain.
This is the result of a number of factors that act as potential barriers to LAI use, not the least of which is misconceptions among caregiver and health care professionals about the drugs.
“When I first was in medical school, this was in the 1990s ... there were really only first-generation antipsychotics available in a depot or a long-acting form, and those had very severe side effects,” said Dr. Gopal.
“They would tend to cause all sorts of movement disorders and would make patients feel really drowsy throughout the day, so they really hated taking them,” he said, noting that these depot medications were oil based, which was painful on injection and caused reactions.
While the newer generations of LAIs are water based and have a much-improved adverse effect profile, doctors “on my end of the age spectrum have all those negative connotations and memories in their minds about what these older LAIs were like, ” Dr. Gopal said
“It’s only the newer generation of doctors who were not around at the time that have a more forward-thinking attitude about the newer long-actings.”
Differences by country
To assess factors that determine patients’ medication preferences in order to better understand expectations and reduce potential barriers to treatment, the researchers analyzed data on 1,429 patients with schizophrenia who were participants in a double-blind, randomized, noninferiority study of paliperidone palmitate taken monthly versus once every 3 months.
Participants had a mean age of 38.4 years, and 55% were men. The majority (54%) were white, 8% were black or African American, and 38% were from other races. About one-eighth (12%) of the patients were from the United States.
The highest preference for LAIs was in Europe, at 88%, vs 59.1% in the United States and 70.7% in the rest of the world.
Interestingly, the preference for LAIs in the United States was comparable across different races, at 59.6% among black patients, 58.8% among whites, and 57.1% for other races.
All study participants had a confirmed diagnosis of schizophrenia and a Positive and Negative Syndrome Scale total score of between 70 and 120 at baseline, with worsening symptoms.
They completed the Medication Preference Questionnaire on day 1, day 120, and at the end of the study, with the current analysis focusing on day 1 responses, as that was the only time when patients would not have received any study medication.
Patient empowerment key
The most common reason patients cited for preferring LAIs over oral antipsychotics were that they felt healthier (57%), could get back to their favorite activities (56%), and didn’t have to think about taking their medication (54%).
In terms of their personal experiences, patients preferred LAIs to pills because they “are easier for me” (67% vs. 18%) and offered a greater sense of control and relieved them from having to think about taking medication (64% vs. 14%).
Finally, 50% of patients preferred LAI injections once every 3 months versus 38% for monthly and 3% for daily injections. Main reason cited were fewer injections (96%), less pain (84%), and fewer doctor visits (80%).
The preferred site for LAI injection was deltoid muscle over gluteal muscle, at 59%, with faster administration (63%), easier use (51%), and the location being less embarrassing (44%) cited as the primary reasons.
“Overall, patient empowerment and quality of life–related goals were important for patients who preferred LAI antipsychotics,” the investigators noted.
Logistic regression analysis indicated that only race and country were significantly associated with medication preferences, with white patients significantly more likely than others to prefer LAIs versus oral medications (adjusted odds ratio, 2.39; P < .001). U.S. patients were significantly less likely to prefer the drugs than those from other countries (aOR, 0.41; P < .001).
Dr. Gopal added that significant differences in patient preference for LAIs likely have a lot to do with the prevailing attitudes of doctors from different countries, with low LAI use corresponding to “more negative attitudes.”
“Better understanding of patients’ treatment priorities and perspective could help overcome barriers to LAI use and inform best course of personalized schizophrenia treatment for improved patient satisfaction and medication adherence,” the investigators noted.
Approached for comment, Matej Markota, MD, a psychiatrist at the Mayo Clinic in Rochester, Minn., who was not involved with the research, said that he agreed with the findings of the study.
He said in an interview that, in his clinical experience, the convenience of not having to take medications daily is an important factor that drives patient preference for LAI use over oral medications.
The study was funded by Janssen Research & Development. Dr. Gopal reports he is an employee of Janssen Research & Developmentand owns stock/equity in Johnson & Johnson. Dr. Markota has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM SIRS 2020
What’s pushing cannabis use in first-episode psychosis?
The desire to feel better is a major driver for patients with first-episode psychosis (FEP) to turn to cannabis, new research shows.
An analysis of more than 1,300 individuals from six European countries showed patients with FEP were four times more likely than their healthy peers to start smoking cannabis in order to make themselves feel better.
The results also revealed that initiating cannabis use to feel better was associated with a more than tripled risk of being a daily user.
as well as offer an opportunity for psychoeducation – particularly as the reasons for starting cannabis appear to influence frequency of use, study investigator Edoardo Spinazzola, MD, Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, said in an interview.
Patients who start smoking cannabis because their friends or family partakes may benefit from therapies that encourage more “assertiveness” and being “socially comfortable without the substance,” Dr. Spinazzola said, noting that it might also be beneficial to identify the specific cause of the psychological discomfort driving cannabis use, such as depression, and specifically treat that issue.
The results were scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
Answering the skeptics
Previous studies suggest that cannabis use can increase risk for psychosis up to 290%, with both frequency of use and potency playing a role, the researchers noted.
However, they added that “skeptics” argue the association could be caused by individuals with psychosis using cannabis as a form of self-medication, the comorbid effect of other psychogenic drugs, or a common genetic vulnerability between cannabis use and psychosis.
The reasons for starting cannabis use remain “largely unexplored,” so the researchers examined records from the European network of national schizophrenia networks studying Gene-Environment Interactions (EU-GEI) database, which includes patients with FEP and healthy individuals acting as controls from France, Italy, the Netherlands, Spain, United Kingdom, and Brazil.
The analysis included 1,347 individuals, of whom 446 had a diagnosis of nonaffective psychosis, 89 had bipolar disorder, and 58 had psychotic depression.
Reasons to start smoking cannabis and patterns of use were determined using the modified version of the Cannabis Experiences Questionnaire.
Results showed that participants who started cannabis to feel better were significantly more likely to be younger, have fewer years of education, to be black or of mixed ethnicity, to be single, or to not be living independently than those who started it because their friends or family were using it (P < .001 for all comparisons).
In addition, 68% of the patients with FEP and 85% of the healthy controls started using cannabis because friends or family were using it. In contrast, 18% of those with FEP versus 5% of controls starting using cannabis to feel better; 13% versus 10%, respectively, started using for “other reasons.”
After taking into account gender, age, ethnicity, and study site, the patients with FEP were significantly more likely than their healthy peers to have started using cannabis to feel better (relative risk ratio, 4.67; P < .001).
Starting to smoke cannabis to feel better versus any other reason was associated with an increased frequency of use in both those with and without FEP, with an RRR of 2.9 for using the drug more than once a week (P = .001) and an RRR of 3.13 for daily use (P < .001). However, the association was stronger in the healthy controls than in those with FEP, with an RRR for daily use of 4.45 versus 3.11, respectively.
The investigators also examined whether there was a link between reasons to start smoking and an individual’s polygenic risk score (PRS) for developing schizophrenia.
Multinomial regression indicated that PRS was not associated with starting cannabis to feel better or because friends were using it. However, there was an association between PRS score and starting the drug because family members were using it (RRR, 0.68; P < .05).
Complex association
Gabriella Gobbi, MD, PhD, professor in the neurobiological psychiatry unit, department of psychiatry, at McGill University, Montreal, said the data confirm “what we already know about cannabis.”
She noted that one of the “major causes” of young people starting cannabis is the social environment, while the desire to use the drug to feel better is linked to “the fact that cannabis, in a lot of cases, is used as a self-medication” in order to be calmer and as a relief from anxiety.
There is a “very complex” association between using cannabis to feel better and the self-medication seen with cigarette smoking and alcohol in patients with schizophrenia, said Dr. Gobbi, who was not involved with the research.
“When we talk about [patients using] cannabis, alcohol, and cigarettes, actually we’re talking about the same group of people,” she said.
Although “it is true they say that people look to cigarettes, tobacco, and alcohol to feel happier because they are depressed, the risk of psychosis is only for cannabis,” she added. “It is very low for alcohol and tobacco.”
As a result, Dr. Gobbi said she and her colleagues are “very worried” about the consequences for mental health of the legalization of cannabis consumption in Canada in October 2018 with the passing of the Cannabis Act.
Although there are no firm statistics yet, she has observed that since the law was passed, cannabis use has stabilized at a lower level among adolescents. “But now we have another population of people aged 34 and older that consume cannabis,” she said.
Particularly when considering the impact of higher strength cannabis on psychosis risk, Dr. Gobbi believes the increase in consumption in this age group will result in a “more elevated” risk for mental health issues.
Dr. Spinazzola and Dr. Gobbi have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The desire to feel better is a major driver for patients with first-episode psychosis (FEP) to turn to cannabis, new research shows.
An analysis of more than 1,300 individuals from six European countries showed patients with FEP were four times more likely than their healthy peers to start smoking cannabis in order to make themselves feel better.
The results also revealed that initiating cannabis use to feel better was associated with a more than tripled risk of being a daily user.
as well as offer an opportunity for psychoeducation – particularly as the reasons for starting cannabis appear to influence frequency of use, study investigator Edoardo Spinazzola, MD, Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, said in an interview.
Patients who start smoking cannabis because their friends or family partakes may benefit from therapies that encourage more “assertiveness” and being “socially comfortable without the substance,” Dr. Spinazzola said, noting that it might also be beneficial to identify the specific cause of the psychological discomfort driving cannabis use, such as depression, and specifically treat that issue.
The results were scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
Answering the skeptics
Previous studies suggest that cannabis use can increase risk for psychosis up to 290%, with both frequency of use and potency playing a role, the researchers noted.
However, they added that “skeptics” argue the association could be caused by individuals with psychosis using cannabis as a form of self-medication, the comorbid effect of other psychogenic drugs, or a common genetic vulnerability between cannabis use and psychosis.
The reasons for starting cannabis use remain “largely unexplored,” so the researchers examined records from the European network of national schizophrenia networks studying Gene-Environment Interactions (EU-GEI) database, which includes patients with FEP and healthy individuals acting as controls from France, Italy, the Netherlands, Spain, United Kingdom, and Brazil.
The analysis included 1,347 individuals, of whom 446 had a diagnosis of nonaffective psychosis, 89 had bipolar disorder, and 58 had psychotic depression.
Reasons to start smoking cannabis and patterns of use were determined using the modified version of the Cannabis Experiences Questionnaire.
Results showed that participants who started cannabis to feel better were significantly more likely to be younger, have fewer years of education, to be black or of mixed ethnicity, to be single, or to not be living independently than those who started it because their friends or family were using it (P < .001 for all comparisons).
In addition, 68% of the patients with FEP and 85% of the healthy controls started using cannabis because friends or family were using it. In contrast, 18% of those with FEP versus 5% of controls starting using cannabis to feel better; 13% versus 10%, respectively, started using for “other reasons.”
After taking into account gender, age, ethnicity, and study site, the patients with FEP were significantly more likely than their healthy peers to have started using cannabis to feel better (relative risk ratio, 4.67; P < .001).
Starting to smoke cannabis to feel better versus any other reason was associated with an increased frequency of use in both those with and without FEP, with an RRR of 2.9 for using the drug more than once a week (P = .001) and an RRR of 3.13 for daily use (P < .001). However, the association was stronger in the healthy controls than in those with FEP, with an RRR for daily use of 4.45 versus 3.11, respectively.
The investigators also examined whether there was a link between reasons to start smoking and an individual’s polygenic risk score (PRS) for developing schizophrenia.
Multinomial regression indicated that PRS was not associated with starting cannabis to feel better or because friends were using it. However, there was an association between PRS score and starting the drug because family members were using it (RRR, 0.68; P < .05).
Complex association
Gabriella Gobbi, MD, PhD, professor in the neurobiological psychiatry unit, department of psychiatry, at McGill University, Montreal, said the data confirm “what we already know about cannabis.”
She noted that one of the “major causes” of young people starting cannabis is the social environment, while the desire to use the drug to feel better is linked to “the fact that cannabis, in a lot of cases, is used as a self-medication” in order to be calmer and as a relief from anxiety.
There is a “very complex” association between using cannabis to feel better and the self-medication seen with cigarette smoking and alcohol in patients with schizophrenia, said Dr. Gobbi, who was not involved with the research.
“When we talk about [patients using] cannabis, alcohol, and cigarettes, actually we’re talking about the same group of people,” she said.
Although “it is true they say that people look to cigarettes, tobacco, and alcohol to feel happier because they are depressed, the risk of psychosis is only for cannabis,” she added. “It is very low for alcohol and tobacco.”
As a result, Dr. Gobbi said she and her colleagues are “very worried” about the consequences for mental health of the legalization of cannabis consumption in Canada in October 2018 with the passing of the Cannabis Act.
Although there are no firm statistics yet, she has observed that since the law was passed, cannabis use has stabilized at a lower level among adolescents. “But now we have another population of people aged 34 and older that consume cannabis,” she said.
Particularly when considering the impact of higher strength cannabis on psychosis risk, Dr. Gobbi believes the increase in consumption in this age group will result in a “more elevated” risk for mental health issues.
Dr. Spinazzola and Dr. Gobbi have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The desire to feel better is a major driver for patients with first-episode psychosis (FEP) to turn to cannabis, new research shows.
An analysis of more than 1,300 individuals from six European countries showed patients with FEP were four times more likely than their healthy peers to start smoking cannabis in order to make themselves feel better.
The results also revealed that initiating cannabis use to feel better was associated with a more than tripled risk of being a daily user.
as well as offer an opportunity for psychoeducation – particularly as the reasons for starting cannabis appear to influence frequency of use, study investigator Edoardo Spinazzola, MD, Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, said in an interview.
Patients who start smoking cannabis because their friends or family partakes may benefit from therapies that encourage more “assertiveness” and being “socially comfortable without the substance,” Dr. Spinazzola said, noting that it might also be beneficial to identify the specific cause of the psychological discomfort driving cannabis use, such as depression, and specifically treat that issue.
The results were scheduled to be presented at the Congress of the Schizophrenia International Research Society 2020, but the meeting was canceled because of the coronavirus pandemic.
Answering the skeptics
Previous studies suggest that cannabis use can increase risk for psychosis up to 290%, with both frequency of use and potency playing a role, the researchers noted.
However, they added that “skeptics” argue the association could be caused by individuals with psychosis using cannabis as a form of self-medication, the comorbid effect of other psychogenic drugs, or a common genetic vulnerability between cannabis use and psychosis.
The reasons for starting cannabis use remain “largely unexplored,” so the researchers examined records from the European network of national schizophrenia networks studying Gene-Environment Interactions (EU-GEI) database, which includes patients with FEP and healthy individuals acting as controls from France, Italy, the Netherlands, Spain, United Kingdom, and Brazil.
The analysis included 1,347 individuals, of whom 446 had a diagnosis of nonaffective psychosis, 89 had bipolar disorder, and 58 had psychotic depression.
Reasons to start smoking cannabis and patterns of use were determined using the modified version of the Cannabis Experiences Questionnaire.
Results showed that participants who started cannabis to feel better were significantly more likely to be younger, have fewer years of education, to be black or of mixed ethnicity, to be single, or to not be living independently than those who started it because their friends or family were using it (P < .001 for all comparisons).
In addition, 68% of the patients with FEP and 85% of the healthy controls started using cannabis because friends or family were using it. In contrast, 18% of those with FEP versus 5% of controls starting using cannabis to feel better; 13% versus 10%, respectively, started using for “other reasons.”
After taking into account gender, age, ethnicity, and study site, the patients with FEP were significantly more likely than their healthy peers to have started using cannabis to feel better (relative risk ratio, 4.67; P < .001).
Starting to smoke cannabis to feel better versus any other reason was associated with an increased frequency of use in both those with and without FEP, with an RRR of 2.9 for using the drug more than once a week (P = .001) and an RRR of 3.13 for daily use (P < .001). However, the association was stronger in the healthy controls than in those with FEP, with an RRR for daily use of 4.45 versus 3.11, respectively.
The investigators also examined whether there was a link between reasons to start smoking and an individual’s polygenic risk score (PRS) for developing schizophrenia.
Multinomial regression indicated that PRS was not associated with starting cannabis to feel better or because friends were using it. However, there was an association between PRS score and starting the drug because family members were using it (RRR, 0.68; P < .05).
Complex association
Gabriella Gobbi, MD, PhD, professor in the neurobiological psychiatry unit, department of psychiatry, at McGill University, Montreal, said the data confirm “what we already know about cannabis.”
She noted that one of the “major causes” of young people starting cannabis is the social environment, while the desire to use the drug to feel better is linked to “the fact that cannabis, in a lot of cases, is used as a self-medication” in order to be calmer and as a relief from anxiety.
There is a “very complex” association between using cannabis to feel better and the self-medication seen with cigarette smoking and alcohol in patients with schizophrenia, said Dr. Gobbi, who was not involved with the research.
“When we talk about [patients using] cannabis, alcohol, and cigarettes, actually we’re talking about the same group of people,” she said.
Although “it is true they say that people look to cigarettes, tobacco, and alcohol to feel happier because they are depressed, the risk of psychosis is only for cannabis,” she added. “It is very low for alcohol and tobacco.”
As a result, Dr. Gobbi said she and her colleagues are “very worried” about the consequences for mental health of the legalization of cannabis consumption in Canada in October 2018 with the passing of the Cannabis Act.
Although there are no firm statistics yet, she has observed that since the law was passed, cannabis use has stabilized at a lower level among adolescents. “But now we have another population of people aged 34 and older that consume cannabis,” she said.
Particularly when considering the impact of higher strength cannabis on psychosis risk, Dr. Gobbi believes the increase in consumption in this age group will result in a “more elevated” risk for mental health issues.
Dr. Spinazzola and Dr. Gobbi have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM SIRS 2020
Amid pandemic, prison psychiatrists adjust and persist
Maryland psychiatrist Annette Hanson, MD, hasn’t changed her morning routine much since the coronavirus pandemic began. She still avoids putting on a necklace or earrings, which could be torn away or used as a ligature, and heads to work.
The only difference is that Dr. Hanson wears easy-to-clean scrubs instead of business attire. “That way I can strip down and shower as soon as I get home. I’m not sure that’s necessary, but I’m being cautious,” said Dr. Hanson, a forensic psychiatrist who is an assistant professor of psychiatry at the University of Maryland and at Johns Hopkins University, both in Baltimore.
As many of her colleagues shelter in place and work from home with the help of telemedicine, prison psychiatrists such as Dr. Hanson continue to evaluate and treat patients in person – behind bars. That hasn’t changed. But so much else has, from the elimination of family visits to the suspension of many court hearings, leaving already vulnerable inmates in limbo.
“Prisons continue to be a poor place to receive mental health care. The setting is destructive to physical and mental health, and the pandemic has made it worse,” said Bandy X. Lee, MD, MDiv, of Yale University, New Haven, Conn., who treats inmates in several states.
Like the inmates they treat, “The most challenging part is to continue care in a system that has essentially been frozen in place,” Dr. Hanson said.
As of June 9, nearly 44,000 inmates in federal and state prisons had tested positive for coronavirus, according to the Associated Press and the Marshall Project. At least 500 people have died. Those numbers do not include inmates and staff members in local jails or juvenile detention centers.
Statistics about COVID-19 in prison staff members are incomplete since only 20 states reported them, and it’s not clear where they contracted the virus. Even so, at least 9,180 cases in staff members were reported, along with 38 deaths, the AP/Marshall Project report.
Using telemedicine is impossible at many jails and prisons, forcing many psychiatrists to protect themselves and their patients as best they can. At the Los Angeles County Jail, which does not use telemedicine, group sessions have been greatly reduced. Instead, psychiatrists are spending more time talking to inmates at the doors to cells or modules, said supervising psychiatrist Joseph R. Simpson, MD, PhD.
The risk of transmission still exists, he said. “Our health system has a comprehensive testing, monitoring, and isolation system in place now to slow the spread and flatten the curve,” Dr. Simpson said. “However, once COVID enters any correctional facility, preventing it from spreading entirely is difficult or impossible given the nature of the living arrangements.”
In interviews, psychiatrists said inmates are more stressed by the limitations spawned by the pandemic than the risk of infection. Many facilities have banned in-person visits, and telephone calls are an expensive alternative, said Nicolas Badre, MD, who treats inmates at jails in the San Diego region.
“That one lifeline you had is no longer there. The second lifeline is that your public defender will get you a plea deal, but they’ve postponed hearings,” he said. “I’ve seen cases of folks who are more anxious and more depressed because COVID is delaying their case or because they’re unable to speak with their families and friends.”
Restrictions on contact with people on the outside are especially difficult for inmates at risk of psychosis, Dr. Badre said. “You add those two [limitations], and how does that not sound to someone with schizophrenia like the government is out to get you? And when someone asks you to wear a mask, how do you trust them?”
According to Dr. Lee, some patients with severe mental illness are unable to comprehend the risk of the pandemic, and they fail to protect themselves. While she’s begun to rely on telemedicine, “it’s a very blunt instrument. Many of my patients are very sick and less able to interact with a screen. And sometimes you’re exhausted at the end of the day because you’ve been yelling at the screen and trying different ways to gain the attention of individuals who are responding to external stimuli and can’t engage.”
The pandemic has improved conditions in prisons and jails on one front: Many are releasing inmates to lower the risk of spreading infection. And Dr. Badre said, “a lot of people are doing just fine, finding themselves to be completely resilient and finding meaning at this time.”
Other than anxiety, the psychiatrists did not report seeing higher percentages of any specific conditions. And they said they are not prescribing any more medications than before COVID-19. But many of the perennial treatments for anxiety – improving the diet, getting out and exercising, developing a hobby, reaching out to others – can be difficult at the best of times behind bars. Those treatments might be impossible now.
At the juvenile justice system in the Chicago area, for example, the pandemic has forced the cancellation of activities such as writing, taking art classes, and barber training. In-person visits are banned, too. “For a lot of them, seeing their family relieves stress, makes them feel more hopeful. It gives them a sense of normalcy to hug their mom,” said Yana Oskin, MD.
But it’s still possible to urge the young people to read, write, work with puzzles, and exercise daily even if it’s just in their rooms, she said. “While their movements have been limited, they do still get to go outside. If they can’t go to the gym, the recreation specialist comes to their pod.”
And while some psychiatrists and older inmates might not be thrilled to have to adjust to therapy via screen during the pandemic, young people are a different story. Dr. Oskin is working with them via telemedicine, which allowed at least one inmate to gain a kind of victory.
“We have an assistant who sets up Skype visits, and the camera was not angled properly,” she recalled. “She couldn’t figure out. The kid sat down and fixed it in 2 seconds.”
Dr. Hanson is the coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has no other disclosures. Dr. Lee is the author of “Violence: An Interdisciplinary Approach to Causes, Consequences and Cures” (Wiley Blackwell, 2019). She has no other disclosures. Dr. Simpson is coauthor of “Neuroimaging in Forensic Psychiatry: From the Clinic to the Courtroom” (Wiley Blackwell, 2012). He has no other disclosures. Dr. Badre and Dr. Oskin reported no disclosures.
Maryland psychiatrist Annette Hanson, MD, hasn’t changed her morning routine much since the coronavirus pandemic began. She still avoids putting on a necklace or earrings, which could be torn away or used as a ligature, and heads to work.
The only difference is that Dr. Hanson wears easy-to-clean scrubs instead of business attire. “That way I can strip down and shower as soon as I get home. I’m not sure that’s necessary, but I’m being cautious,” said Dr. Hanson, a forensic psychiatrist who is an assistant professor of psychiatry at the University of Maryland and at Johns Hopkins University, both in Baltimore.
As many of her colleagues shelter in place and work from home with the help of telemedicine, prison psychiatrists such as Dr. Hanson continue to evaluate and treat patients in person – behind bars. That hasn’t changed. But so much else has, from the elimination of family visits to the suspension of many court hearings, leaving already vulnerable inmates in limbo.
“Prisons continue to be a poor place to receive mental health care. The setting is destructive to physical and mental health, and the pandemic has made it worse,” said Bandy X. Lee, MD, MDiv, of Yale University, New Haven, Conn., who treats inmates in several states.
Like the inmates they treat, “The most challenging part is to continue care in a system that has essentially been frozen in place,” Dr. Hanson said.
As of June 9, nearly 44,000 inmates in federal and state prisons had tested positive for coronavirus, according to the Associated Press and the Marshall Project. At least 500 people have died. Those numbers do not include inmates and staff members in local jails or juvenile detention centers.
Statistics about COVID-19 in prison staff members are incomplete since only 20 states reported them, and it’s not clear where they contracted the virus. Even so, at least 9,180 cases in staff members were reported, along with 38 deaths, the AP/Marshall Project report.
Using telemedicine is impossible at many jails and prisons, forcing many psychiatrists to protect themselves and their patients as best they can. At the Los Angeles County Jail, which does not use telemedicine, group sessions have been greatly reduced. Instead, psychiatrists are spending more time talking to inmates at the doors to cells or modules, said supervising psychiatrist Joseph R. Simpson, MD, PhD.
The risk of transmission still exists, he said. “Our health system has a comprehensive testing, monitoring, and isolation system in place now to slow the spread and flatten the curve,” Dr. Simpson said. “However, once COVID enters any correctional facility, preventing it from spreading entirely is difficult or impossible given the nature of the living arrangements.”
In interviews, psychiatrists said inmates are more stressed by the limitations spawned by the pandemic than the risk of infection. Many facilities have banned in-person visits, and telephone calls are an expensive alternative, said Nicolas Badre, MD, who treats inmates at jails in the San Diego region.
“That one lifeline you had is no longer there. The second lifeline is that your public defender will get you a plea deal, but they’ve postponed hearings,” he said. “I’ve seen cases of folks who are more anxious and more depressed because COVID is delaying their case or because they’re unable to speak with their families and friends.”
Restrictions on contact with people on the outside are especially difficult for inmates at risk of psychosis, Dr. Badre said. “You add those two [limitations], and how does that not sound to someone with schizophrenia like the government is out to get you? And when someone asks you to wear a mask, how do you trust them?”
According to Dr. Lee, some patients with severe mental illness are unable to comprehend the risk of the pandemic, and they fail to protect themselves. While she’s begun to rely on telemedicine, “it’s a very blunt instrument. Many of my patients are very sick and less able to interact with a screen. And sometimes you’re exhausted at the end of the day because you’ve been yelling at the screen and trying different ways to gain the attention of individuals who are responding to external stimuli and can’t engage.”
The pandemic has improved conditions in prisons and jails on one front: Many are releasing inmates to lower the risk of spreading infection. And Dr. Badre said, “a lot of people are doing just fine, finding themselves to be completely resilient and finding meaning at this time.”
Other than anxiety, the psychiatrists did not report seeing higher percentages of any specific conditions. And they said they are not prescribing any more medications than before COVID-19. But many of the perennial treatments for anxiety – improving the diet, getting out and exercising, developing a hobby, reaching out to others – can be difficult at the best of times behind bars. Those treatments might be impossible now.
At the juvenile justice system in the Chicago area, for example, the pandemic has forced the cancellation of activities such as writing, taking art classes, and barber training. In-person visits are banned, too. “For a lot of them, seeing their family relieves stress, makes them feel more hopeful. It gives them a sense of normalcy to hug their mom,” said Yana Oskin, MD.
But it’s still possible to urge the young people to read, write, work with puzzles, and exercise daily even if it’s just in their rooms, she said. “While their movements have been limited, they do still get to go outside. If they can’t go to the gym, the recreation specialist comes to their pod.”
And while some psychiatrists and older inmates might not be thrilled to have to adjust to therapy via screen during the pandemic, young people are a different story. Dr. Oskin is working with them via telemedicine, which allowed at least one inmate to gain a kind of victory.
“We have an assistant who sets up Skype visits, and the camera was not angled properly,” she recalled. “She couldn’t figure out. The kid sat down and fixed it in 2 seconds.”
Dr. Hanson is the coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has no other disclosures. Dr. Lee is the author of “Violence: An Interdisciplinary Approach to Causes, Consequences and Cures” (Wiley Blackwell, 2019). She has no other disclosures. Dr. Simpson is coauthor of “Neuroimaging in Forensic Psychiatry: From the Clinic to the Courtroom” (Wiley Blackwell, 2012). He has no other disclosures. Dr. Badre and Dr. Oskin reported no disclosures.
Maryland psychiatrist Annette Hanson, MD, hasn’t changed her morning routine much since the coronavirus pandemic began. She still avoids putting on a necklace or earrings, which could be torn away or used as a ligature, and heads to work.
The only difference is that Dr. Hanson wears easy-to-clean scrubs instead of business attire. “That way I can strip down and shower as soon as I get home. I’m not sure that’s necessary, but I’m being cautious,” said Dr. Hanson, a forensic psychiatrist who is an assistant professor of psychiatry at the University of Maryland and at Johns Hopkins University, both in Baltimore.
As many of her colleagues shelter in place and work from home with the help of telemedicine, prison psychiatrists such as Dr. Hanson continue to evaluate and treat patients in person – behind bars. That hasn’t changed. But so much else has, from the elimination of family visits to the suspension of many court hearings, leaving already vulnerable inmates in limbo.
“Prisons continue to be a poor place to receive mental health care. The setting is destructive to physical and mental health, and the pandemic has made it worse,” said Bandy X. Lee, MD, MDiv, of Yale University, New Haven, Conn., who treats inmates in several states.
Like the inmates they treat, “The most challenging part is to continue care in a system that has essentially been frozen in place,” Dr. Hanson said.
As of June 9, nearly 44,000 inmates in federal and state prisons had tested positive for coronavirus, according to the Associated Press and the Marshall Project. At least 500 people have died. Those numbers do not include inmates and staff members in local jails or juvenile detention centers.
Statistics about COVID-19 in prison staff members are incomplete since only 20 states reported them, and it’s not clear where they contracted the virus. Even so, at least 9,180 cases in staff members were reported, along with 38 deaths, the AP/Marshall Project report.
Using telemedicine is impossible at many jails and prisons, forcing many psychiatrists to protect themselves and their patients as best they can. At the Los Angeles County Jail, which does not use telemedicine, group sessions have been greatly reduced. Instead, psychiatrists are spending more time talking to inmates at the doors to cells or modules, said supervising psychiatrist Joseph R. Simpson, MD, PhD.
The risk of transmission still exists, he said. “Our health system has a comprehensive testing, monitoring, and isolation system in place now to slow the spread and flatten the curve,” Dr. Simpson said. “However, once COVID enters any correctional facility, preventing it from spreading entirely is difficult or impossible given the nature of the living arrangements.”
In interviews, psychiatrists said inmates are more stressed by the limitations spawned by the pandemic than the risk of infection. Many facilities have banned in-person visits, and telephone calls are an expensive alternative, said Nicolas Badre, MD, who treats inmates at jails in the San Diego region.
“That one lifeline you had is no longer there. The second lifeline is that your public defender will get you a plea deal, but they’ve postponed hearings,” he said. “I’ve seen cases of folks who are more anxious and more depressed because COVID is delaying their case or because they’re unable to speak with their families and friends.”
Restrictions on contact with people on the outside are especially difficult for inmates at risk of psychosis, Dr. Badre said. “You add those two [limitations], and how does that not sound to someone with schizophrenia like the government is out to get you? And when someone asks you to wear a mask, how do you trust them?”
According to Dr. Lee, some patients with severe mental illness are unable to comprehend the risk of the pandemic, and they fail to protect themselves. While she’s begun to rely on telemedicine, “it’s a very blunt instrument. Many of my patients are very sick and less able to interact with a screen. And sometimes you’re exhausted at the end of the day because you’ve been yelling at the screen and trying different ways to gain the attention of individuals who are responding to external stimuli and can’t engage.”
The pandemic has improved conditions in prisons and jails on one front: Many are releasing inmates to lower the risk of spreading infection. And Dr. Badre said, “a lot of people are doing just fine, finding themselves to be completely resilient and finding meaning at this time.”
Other than anxiety, the psychiatrists did not report seeing higher percentages of any specific conditions. And they said they are not prescribing any more medications than before COVID-19. But many of the perennial treatments for anxiety – improving the diet, getting out and exercising, developing a hobby, reaching out to others – can be difficult at the best of times behind bars. Those treatments might be impossible now.
At the juvenile justice system in the Chicago area, for example, the pandemic has forced the cancellation of activities such as writing, taking art classes, and barber training. In-person visits are banned, too. “For a lot of them, seeing their family relieves stress, makes them feel more hopeful. It gives them a sense of normalcy to hug their mom,” said Yana Oskin, MD.
But it’s still possible to urge the young people to read, write, work with puzzles, and exercise daily even if it’s just in their rooms, she said. “While their movements have been limited, they do still get to go outside. If they can’t go to the gym, the recreation specialist comes to their pod.”
And while some psychiatrists and older inmates might not be thrilled to have to adjust to therapy via screen during the pandemic, young people are a different story. Dr. Oskin is working with them via telemedicine, which allowed at least one inmate to gain a kind of victory.
“We have an assistant who sets up Skype visits, and the camera was not angled properly,” she recalled. “She couldn’t figure out. The kid sat down and fixed it in 2 seconds.”
Dr. Hanson is the coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has no other disclosures. Dr. Lee is the author of “Violence: An Interdisciplinary Approach to Causes, Consequences and Cures” (Wiley Blackwell, 2019). She has no other disclosures. Dr. Simpson is coauthor of “Neuroimaging in Forensic Psychiatry: From the Clinic to the Courtroom” (Wiley Blackwell, 2012). He has no other disclosures. Dr. Badre and Dr. Oskin reported no disclosures.
First-episode psychosis in the time of COVID-19
Patients may need more than weekly teletherapy
In response to COVID-19, we have seen a rapid transformation to virtually delivered mental health care, essential for the prevention and treatment of various mental health conditions during an isolating and stress-inducing pandemic. Yet teletherapy and virtual medication management alone may not adequately address the needs of some of the populations we serve.
Take Jackson, whose name and details have been changed for privacy. A year ago, Jackson, in his last year of high school, began hearing voices that others could not hear. After becoming increasingly withdrawn, his father sought out treatment for him and learned that Jackson was experiencing his first episode of psychosis.
Psychosis involves disruptions in the way one processes thoughts and feelings or behaves, and includes delusions – or unusual beliefs – and hallucinations, meaning seeing and hearing things that others cannot. “First-episode psychosis” (FEP) simply refers to the first time an individual experiences this. It typically occurs between one’s teenage years and their 20s. Whereas some individuals recover from their first episode and may not experience another, others go on to experience recurrence, and sometimes a waxing and waning illness course.
Jackson enrolled in a comprehensive mental health program that not only includes a psychiatrist, but also therapists who provide case management services, as well as a peer specialist; this is someone with lived experience navigating mental illness. The program also includes an employment and education specialist and family and group therapy sessions. His team helped him identify and work toward his personal recovery goals: graduating from high school, obtaining a job, and maintaining a strong relationship with his father.
One hundred thousand adolescents and young adults like Jackson experience FEP each year, and now, in the wake of COVID-19, they probably have more limited access to the kind of support that can be vital to recovery.
Studies have shown that untreated psychosis can detrimentally affect quality of life in several ways, including by negatively affecting interpersonal relationships, interfering with obtaining or maintaining employment, and increasing the risk for problematic substance use. The psychosocial effects of COVID-19 could compound problems that individuals navigating psychosis already face, such as stigmatization, social isolation, and unemployment. On top of this, individuals who experience additional marginalization and downstream effects of systematic discriminatory practices by virtue of their race or ethnicity, immigration status, or language bear the brunt of some of this pandemic’s worst health inequities.
Early and efficacious treatment is critically important for individuals experiencing psychosis. Evidence shows that engagement in coordinated specialty care (CSC) specifically can improve outcomes, including the likelihood of being engaged in school or work and lower rates of hospitalization. CSC is a team-based approach that utilizes the unique skills of every team member to support an individual in reaching their recovery goals, whether it’s starting or finishing college or building a new relationship.
Unlike traditional treatment goals, which often focus on “symptom reduction,” recovery-oriented care is about supporting an individual in obtaining a sense of satisfaction, meaning, and purpose in life. It also supports navigating such experiences as a job interview or a date. These key, multifaceted components must be made accessible and adapted during these times.
For individuals like Jackson, it is crucial to be able to continue accessing quality CSC, even during our current pandemic. Lisa Dixon, MD, a professor of psychiatry at Columbia University, leads ONTrackNY, a statewide FEP program. She states that “effective, recovery-oriented treatment can make such a huge difference in the lives of these young people who are at a potential inflection point in their lives. Creative, collaborative clinicians can maintain connection and support.”
So how can we adapt CSC during this time? In addition to virtualized medication management and individual therapy, other components of CSC can be creatively adapted for online platforms. Group sessions can be completed virtually, from family to peer-led. Though the unemployment rate continues to rise, we can still help participants with a desire to work find employers that are offering remote work or navigate the risks of potential COVID-19 work exposures if remote options aren’t available. We can also support their developing skills to be used once other employers that pose less risk reopen.
For those in school, virtual education support can provide study skills, ways to cope with transition to an online classroom, or help with obtaining tutoring. Nutritionists can work remotely to provide support and creatively use online platforms for real-time feedback in a participant’s kitchen. Virtual case management is even more essential in the wake of COVID-19, from assistance with applying for unemployment insurance and financial aid to obtaining health insurance or determining eligibility.
For those without access to virtual platforms, individual and group telephone sessions and text check-ins can provide meaningful opportunities for continued engagement. For those who are unstably housed or have limited privacy in housing, teams must generate ideas of where to have remote sessions, such as a nearby park.
In a world now dominated by virtual care, it is critically important that individuals needing to see a clinician in person still be able to do so. Whether it is due to an acute crisis or to administer a long-acting injection medication, it is our responsibility to thoughtfully and judiciously remain available to patients, using appropriate personal protective equipment and precautions.
Jackson is one of many young people in recovery from psychosis. He is not defined by or limited by his experiences, but rather is navigating the possibilities that lie ahead of him, defining for himself who he wants to be in this world as it evolves. In the midst of COVID-19, as we seek to innovate – from how we exercise to how we throw birthday parties – let’s also be innovative in how we provide care and support for individuals experiencing psychosis.
This article first appeared on Medscape.com.
Patients may need more than weekly teletherapy
Patients may need more than weekly teletherapy
In response to COVID-19, we have seen a rapid transformation to virtually delivered mental health care, essential for the prevention and treatment of various mental health conditions during an isolating and stress-inducing pandemic. Yet teletherapy and virtual medication management alone may not adequately address the needs of some of the populations we serve.
Take Jackson, whose name and details have been changed for privacy. A year ago, Jackson, in his last year of high school, began hearing voices that others could not hear. After becoming increasingly withdrawn, his father sought out treatment for him and learned that Jackson was experiencing his first episode of psychosis.
Psychosis involves disruptions in the way one processes thoughts and feelings or behaves, and includes delusions – or unusual beliefs – and hallucinations, meaning seeing and hearing things that others cannot. “First-episode psychosis” (FEP) simply refers to the first time an individual experiences this. It typically occurs between one’s teenage years and their 20s. Whereas some individuals recover from their first episode and may not experience another, others go on to experience recurrence, and sometimes a waxing and waning illness course.
Jackson enrolled in a comprehensive mental health program that not only includes a psychiatrist, but also therapists who provide case management services, as well as a peer specialist; this is someone with lived experience navigating mental illness. The program also includes an employment and education specialist and family and group therapy sessions. His team helped him identify and work toward his personal recovery goals: graduating from high school, obtaining a job, and maintaining a strong relationship with his father.
One hundred thousand adolescents and young adults like Jackson experience FEP each year, and now, in the wake of COVID-19, they probably have more limited access to the kind of support that can be vital to recovery.
Studies have shown that untreated psychosis can detrimentally affect quality of life in several ways, including by negatively affecting interpersonal relationships, interfering with obtaining or maintaining employment, and increasing the risk for problematic substance use. The psychosocial effects of COVID-19 could compound problems that individuals navigating psychosis already face, such as stigmatization, social isolation, and unemployment. On top of this, individuals who experience additional marginalization and downstream effects of systematic discriminatory practices by virtue of their race or ethnicity, immigration status, or language bear the brunt of some of this pandemic’s worst health inequities.
Early and efficacious treatment is critically important for individuals experiencing psychosis. Evidence shows that engagement in coordinated specialty care (CSC) specifically can improve outcomes, including the likelihood of being engaged in school or work and lower rates of hospitalization. CSC is a team-based approach that utilizes the unique skills of every team member to support an individual in reaching their recovery goals, whether it’s starting or finishing college or building a new relationship.
Unlike traditional treatment goals, which often focus on “symptom reduction,” recovery-oriented care is about supporting an individual in obtaining a sense of satisfaction, meaning, and purpose in life. It also supports navigating such experiences as a job interview or a date. These key, multifaceted components must be made accessible and adapted during these times.
For individuals like Jackson, it is crucial to be able to continue accessing quality CSC, even during our current pandemic. Lisa Dixon, MD, a professor of psychiatry at Columbia University, leads ONTrackNY, a statewide FEP program. She states that “effective, recovery-oriented treatment can make such a huge difference in the lives of these young people who are at a potential inflection point in their lives. Creative, collaborative clinicians can maintain connection and support.”
So how can we adapt CSC during this time? In addition to virtualized medication management and individual therapy, other components of CSC can be creatively adapted for online platforms. Group sessions can be completed virtually, from family to peer-led. Though the unemployment rate continues to rise, we can still help participants with a desire to work find employers that are offering remote work or navigate the risks of potential COVID-19 work exposures if remote options aren’t available. We can also support their developing skills to be used once other employers that pose less risk reopen.
For those in school, virtual education support can provide study skills, ways to cope with transition to an online classroom, or help with obtaining tutoring. Nutritionists can work remotely to provide support and creatively use online platforms for real-time feedback in a participant’s kitchen. Virtual case management is even more essential in the wake of COVID-19, from assistance with applying for unemployment insurance and financial aid to obtaining health insurance or determining eligibility.
For those without access to virtual platforms, individual and group telephone sessions and text check-ins can provide meaningful opportunities for continued engagement. For those who are unstably housed or have limited privacy in housing, teams must generate ideas of where to have remote sessions, such as a nearby park.
In a world now dominated by virtual care, it is critically important that individuals needing to see a clinician in person still be able to do so. Whether it is due to an acute crisis or to administer a long-acting injection medication, it is our responsibility to thoughtfully and judiciously remain available to patients, using appropriate personal protective equipment and precautions.
Jackson is one of many young people in recovery from psychosis. He is not defined by or limited by his experiences, but rather is navigating the possibilities that lie ahead of him, defining for himself who he wants to be in this world as it evolves. In the midst of COVID-19, as we seek to innovate – from how we exercise to how we throw birthday parties – let’s also be innovative in how we provide care and support for individuals experiencing psychosis.
This article first appeared on Medscape.com.
In response to COVID-19, we have seen a rapid transformation to virtually delivered mental health care, essential for the prevention and treatment of various mental health conditions during an isolating and stress-inducing pandemic. Yet teletherapy and virtual medication management alone may not adequately address the needs of some of the populations we serve.
Take Jackson, whose name and details have been changed for privacy. A year ago, Jackson, in his last year of high school, began hearing voices that others could not hear. After becoming increasingly withdrawn, his father sought out treatment for him and learned that Jackson was experiencing his first episode of psychosis.
Psychosis involves disruptions in the way one processes thoughts and feelings or behaves, and includes delusions – or unusual beliefs – and hallucinations, meaning seeing and hearing things that others cannot. “First-episode psychosis” (FEP) simply refers to the first time an individual experiences this. It typically occurs between one’s teenage years and their 20s. Whereas some individuals recover from their first episode and may not experience another, others go on to experience recurrence, and sometimes a waxing and waning illness course.
Jackson enrolled in a comprehensive mental health program that not only includes a psychiatrist, but also therapists who provide case management services, as well as a peer specialist; this is someone with lived experience navigating mental illness. The program also includes an employment and education specialist and family and group therapy sessions. His team helped him identify and work toward his personal recovery goals: graduating from high school, obtaining a job, and maintaining a strong relationship with his father.
One hundred thousand adolescents and young adults like Jackson experience FEP each year, and now, in the wake of COVID-19, they probably have more limited access to the kind of support that can be vital to recovery.
Studies have shown that untreated psychosis can detrimentally affect quality of life in several ways, including by negatively affecting interpersonal relationships, interfering with obtaining or maintaining employment, and increasing the risk for problematic substance use. The psychosocial effects of COVID-19 could compound problems that individuals navigating psychosis already face, such as stigmatization, social isolation, and unemployment. On top of this, individuals who experience additional marginalization and downstream effects of systematic discriminatory practices by virtue of their race or ethnicity, immigration status, or language bear the brunt of some of this pandemic’s worst health inequities.
Early and efficacious treatment is critically important for individuals experiencing psychosis. Evidence shows that engagement in coordinated specialty care (CSC) specifically can improve outcomes, including the likelihood of being engaged in school or work and lower rates of hospitalization. CSC is a team-based approach that utilizes the unique skills of every team member to support an individual in reaching their recovery goals, whether it’s starting or finishing college or building a new relationship.
Unlike traditional treatment goals, which often focus on “symptom reduction,” recovery-oriented care is about supporting an individual in obtaining a sense of satisfaction, meaning, and purpose in life. It also supports navigating such experiences as a job interview or a date. These key, multifaceted components must be made accessible and adapted during these times.
For individuals like Jackson, it is crucial to be able to continue accessing quality CSC, even during our current pandemic. Lisa Dixon, MD, a professor of psychiatry at Columbia University, leads ONTrackNY, a statewide FEP program. She states that “effective, recovery-oriented treatment can make such a huge difference in the lives of these young people who are at a potential inflection point in their lives. Creative, collaborative clinicians can maintain connection and support.”
So how can we adapt CSC during this time? In addition to virtualized medication management and individual therapy, other components of CSC can be creatively adapted for online platforms. Group sessions can be completed virtually, from family to peer-led. Though the unemployment rate continues to rise, we can still help participants with a desire to work find employers that are offering remote work or navigate the risks of potential COVID-19 work exposures if remote options aren’t available. We can also support their developing skills to be used once other employers that pose less risk reopen.
For those in school, virtual education support can provide study skills, ways to cope with transition to an online classroom, or help with obtaining tutoring. Nutritionists can work remotely to provide support and creatively use online platforms for real-time feedback in a participant’s kitchen. Virtual case management is even more essential in the wake of COVID-19, from assistance with applying for unemployment insurance and financial aid to obtaining health insurance or determining eligibility.
For those without access to virtual platforms, individual and group telephone sessions and text check-ins can provide meaningful opportunities for continued engagement. For those who are unstably housed or have limited privacy in housing, teams must generate ideas of where to have remote sessions, such as a nearby park.
In a world now dominated by virtual care, it is critically important that individuals needing to see a clinician in person still be able to do so. Whether it is due to an acute crisis or to administer a long-acting injection medication, it is our responsibility to thoughtfully and judiciously remain available to patients, using appropriate personal protective equipment and precautions.
Jackson is one of many young people in recovery from psychosis. He is not defined by or limited by his experiences, but rather is navigating the possibilities that lie ahead of him, defining for himself who he wants to be in this world as it evolves. In the midst of COVID-19, as we seek to innovate – from how we exercise to how we throw birthday parties – let’s also be innovative in how we provide care and support for individuals experiencing psychosis.
This article first appeared on Medscape.com.
Preventing arrhythmias and QTc prolongation in COVID-19 patients on psychotropics
Over the last few weeks, several conflicting reports about the efficacy of SARS-CoV-2 treatments have emerged, including high-profile papers that were placed in the limelight and groundbreaking retractions that were issued by the Lancet and New England Journal of Medicine, involving the potential dangers of COVID therapy with findings derived from the Surgisphere database. Hydroxychloroquine has garnered considerable media attention and was touted earlier by President Trump for its therapeutic effects.1 Naturally, there are political connotations associated with the agent, and it is unlikely that hydroxychloroquine will be supplanted in the near future as ongoing clinical trials have demonstrated mixed results amid the controversy.
As clinicians navigating unchartered territory within the hospital setting, we have to come to terms with these new challenges, tailoring treatment protocols accordingly with the best clinical practices in mind. Patients with preexisting mental health conditions and who are being treated for COVID-19 are particularly susceptible to clinical deterioration. Recent studies have indicated that psychiatric patients are more prone to feelings of isolation and/or estrangement as well as exacerbation of symptoms such as paranoia.2 Even more concerning is the medication regimen, namely, the novel combination therapies that arise when agents such as hydroxychloroquine are used in tandem with certain antipsychotics or antidepressants.
What’s at stake for COVID-19–positive mental health care patients?
Although the efficacy of hydroxychloroquine is currently being investigated,3 the antimalarial is usually prescribed in tandem with azithromycin for people with COVID-19. The National Institute of Allergy and Infectious Diseases has advised against that particular combination therapy because of ongoing concerns about toxicities.3,4
In another study, azithromycin was effectively substituted with doxycycline to help minimize systemic effects for patients with cardiac and/or pulmonary issues.5 Azithromycin is notorious in the literature for influencing the electrical activity of the heart with the potential for fatal arrhythmia and sudden cardiac death in individuals at risk for cardiovascular disease.5,6,7 It should be noted that both of these commonly prescribed COVID-19 medications (for example, hydroxychloroquine and azithromycin) could lead to QT interval prolongation especially within the context of combination therapy. This is largely concerning for psychiatrists and various other mental health practitioners for the following reasons: (1) higher rates of metabolic syndrome and cardiovascular diseases among psychiatric patients8 and/or (2) effects of certain antipsychotics (for example, IV haloperidol, thioridazine, and ziprasidone) and antidepressants (for example, citalopram and escitalopram) on the QT interval.9
SARS-CoV-2 and clinical judgment: Evaluating patients at higher risk
Although COVID-19 medication guidelines are still being actively developed, hydroxychloroquine appears to be commonly prescribed by physicians. The medication is known myriad untoward effects, including potential behavioral dysfunction (for example, irritability, agitation, suicidal ideation)10 as well as the aforementioned issues concerning arrhythmia (for example, torsades de pointes). Health care professionals might not have much control over the choice of COVID-19 agents because of a lack of available resources or limited options, but they can exercise clinical judgment with respect to selecting the appropriate psychotropic medications.
Treatment recommendations
1. Establish a baseline EKG
A baseline 12-lead EKG is the standard of care for patients currently being screened for COVID-19. It is necessary to rule out the presence of an underlying cardiovascular disease or a rhythm irregularity. A prolonged QTc interval is generally regarded as being around greater than 450-470 msecs with variations attributable to gender;11 numerous studies have affirmed that the risk of acquiring torsades de pointes is substantial when the QTc interval exceeds 500 msecs.12
2. Medical management and risk assessment
Commonly prescribed antipsychotics such as IV haloperidol and ziprasidone are known for exerting a negative effect on the interval and should readily be substituted with other agents in patients who are being treated for COVID-19; the combination of these antipsychotics alongside some COVID-19 medication regimens (for example, hydroxychloroquine/azithromycin) might prove to be fatal. The same logic applies to COVID-19 patients previously on antidepressant therapeutics such as citalopram and escitalopram.
3. Embrace an individually tailored approach to therapeutics
While American Psychiatric Association guidelines historically supported a cessation or reduction in the offending agent under normal circumstances,12 our team is recommending that the psychotropics associated with QTc interval prolongation are discontinued altogether (or substituted with a low-risk agent) in the event that a patient presents with suspected COVID-19. However, after the patients tests negative with COVID-19, they may resume therapy as indicated under the discretion of the mental health practitioner.
References
1. Offard C. “Lancet, NEJM Retract Surgisphere Studies on COVID-19 Patients.” The Scientist Magazine. 2020 Jun 4.
2. Shigemura J et al. Psychiatry Clin Neurosci. 2020 Apr;74(4):281-2.
3. Keshtkar-Jahromi M and Bavari S. Am J Trop Med Hyg. 2020 May;102(5):932-3.
4. Palca J. “NIH panel recommends against drug combination promoted by Trump for COVID-19.” NPR. 2020 Apr 21.
5. Mongelli L. “Long Island doctor tries new twist on hydroxychloroquine for elderly COVID-19 patients.” New York Post. 2020 Apr 4.
6. Hancox JC et al. Ther Adv Infect Dis. 2013 Oct;(5):155-65.
7. Giudicessi JR and Ackerman MJ. Cleve Clin J Med. 2013 Sep;80(9):539-44.
8. Casey DE. Am J Med. 2005 Apr 1;118(Suppl 2):15S-22S.
9. Beach SR et al. Psychosomatics. 2013 Jan 1;54(1):1-3.
10. Bogaczewicz A and Sobów T. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-4.
11. Chohan PS et al. Pak J Med Sci. 2015 Sep-Oct;31(5):1269-71.
12. Lieberman JA et al. APA guidance on the use of antipsychotic drugs and cardiac sudden death. NYS Office of Mental Health. 2012.
Dr. Faisal A. Islam is medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Faisal Islam disclosed no relevant financial relationships.
Dr. Mohammed Islam is affiliated with the department of psychiatry at the Interfaith Medical Center, New York. He disclosed no relevant financial relationships.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He disclosed no relevant financial relationships.
Over the last few weeks, several conflicting reports about the efficacy of SARS-CoV-2 treatments have emerged, including high-profile papers that were placed in the limelight and groundbreaking retractions that were issued by the Lancet and New England Journal of Medicine, involving the potential dangers of COVID therapy with findings derived from the Surgisphere database. Hydroxychloroquine has garnered considerable media attention and was touted earlier by President Trump for its therapeutic effects.1 Naturally, there are political connotations associated with the agent, and it is unlikely that hydroxychloroquine will be supplanted in the near future as ongoing clinical trials have demonstrated mixed results amid the controversy.
As clinicians navigating unchartered territory within the hospital setting, we have to come to terms with these new challenges, tailoring treatment protocols accordingly with the best clinical practices in mind. Patients with preexisting mental health conditions and who are being treated for COVID-19 are particularly susceptible to clinical deterioration. Recent studies have indicated that psychiatric patients are more prone to feelings of isolation and/or estrangement as well as exacerbation of symptoms such as paranoia.2 Even more concerning is the medication regimen, namely, the novel combination therapies that arise when agents such as hydroxychloroquine are used in tandem with certain antipsychotics or antidepressants.
What’s at stake for COVID-19–positive mental health care patients?
Although the efficacy of hydroxychloroquine is currently being investigated,3 the antimalarial is usually prescribed in tandem with azithromycin for people with COVID-19. The National Institute of Allergy and Infectious Diseases has advised against that particular combination therapy because of ongoing concerns about toxicities.3,4
In another study, azithromycin was effectively substituted with doxycycline to help minimize systemic effects for patients with cardiac and/or pulmonary issues.5 Azithromycin is notorious in the literature for influencing the electrical activity of the heart with the potential for fatal arrhythmia and sudden cardiac death in individuals at risk for cardiovascular disease.5,6,7 It should be noted that both of these commonly prescribed COVID-19 medications (for example, hydroxychloroquine and azithromycin) could lead to QT interval prolongation especially within the context of combination therapy. This is largely concerning for psychiatrists and various other mental health practitioners for the following reasons: (1) higher rates of metabolic syndrome and cardiovascular diseases among psychiatric patients8 and/or (2) effects of certain antipsychotics (for example, IV haloperidol, thioridazine, and ziprasidone) and antidepressants (for example, citalopram and escitalopram) on the QT interval.9
SARS-CoV-2 and clinical judgment: Evaluating patients at higher risk
Although COVID-19 medication guidelines are still being actively developed, hydroxychloroquine appears to be commonly prescribed by physicians. The medication is known myriad untoward effects, including potential behavioral dysfunction (for example, irritability, agitation, suicidal ideation)10 as well as the aforementioned issues concerning arrhythmia (for example, torsades de pointes). Health care professionals might not have much control over the choice of COVID-19 agents because of a lack of available resources or limited options, but they can exercise clinical judgment with respect to selecting the appropriate psychotropic medications.
Treatment recommendations
1. Establish a baseline EKG
A baseline 12-lead EKG is the standard of care for patients currently being screened for COVID-19. It is necessary to rule out the presence of an underlying cardiovascular disease or a rhythm irregularity. A prolonged QTc interval is generally regarded as being around greater than 450-470 msecs with variations attributable to gender;11 numerous studies have affirmed that the risk of acquiring torsades de pointes is substantial when the QTc interval exceeds 500 msecs.12
2. Medical management and risk assessment
Commonly prescribed antipsychotics such as IV haloperidol and ziprasidone are known for exerting a negative effect on the interval and should readily be substituted with other agents in patients who are being treated for COVID-19; the combination of these antipsychotics alongside some COVID-19 medication regimens (for example, hydroxychloroquine/azithromycin) might prove to be fatal. The same logic applies to COVID-19 patients previously on antidepressant therapeutics such as citalopram and escitalopram.
3. Embrace an individually tailored approach to therapeutics
While American Psychiatric Association guidelines historically supported a cessation or reduction in the offending agent under normal circumstances,12 our team is recommending that the psychotropics associated with QTc interval prolongation are discontinued altogether (or substituted with a low-risk agent) in the event that a patient presents with suspected COVID-19. However, after the patients tests negative with COVID-19, they may resume therapy as indicated under the discretion of the mental health practitioner.
References
1. Offard C. “Lancet, NEJM Retract Surgisphere Studies on COVID-19 Patients.” The Scientist Magazine. 2020 Jun 4.
2. Shigemura J et al. Psychiatry Clin Neurosci. 2020 Apr;74(4):281-2.
3. Keshtkar-Jahromi M and Bavari S. Am J Trop Med Hyg. 2020 May;102(5):932-3.
4. Palca J. “NIH panel recommends against drug combination promoted by Trump for COVID-19.” NPR. 2020 Apr 21.
5. Mongelli L. “Long Island doctor tries new twist on hydroxychloroquine for elderly COVID-19 patients.” New York Post. 2020 Apr 4.
6. Hancox JC et al. Ther Adv Infect Dis. 2013 Oct;(5):155-65.
7. Giudicessi JR and Ackerman MJ. Cleve Clin J Med. 2013 Sep;80(9):539-44.
8. Casey DE. Am J Med. 2005 Apr 1;118(Suppl 2):15S-22S.
9. Beach SR et al. Psychosomatics. 2013 Jan 1;54(1):1-3.
10. Bogaczewicz A and Sobów T. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-4.
11. Chohan PS et al. Pak J Med Sci. 2015 Sep-Oct;31(5):1269-71.
12. Lieberman JA et al. APA guidance on the use of antipsychotic drugs and cardiac sudden death. NYS Office of Mental Health. 2012.
Dr. Faisal A. Islam is medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Faisal Islam disclosed no relevant financial relationships.
Dr. Mohammed Islam is affiliated with the department of psychiatry at the Interfaith Medical Center, New York. He disclosed no relevant financial relationships.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He disclosed no relevant financial relationships.
Over the last few weeks, several conflicting reports about the efficacy of SARS-CoV-2 treatments have emerged, including high-profile papers that were placed in the limelight and groundbreaking retractions that were issued by the Lancet and New England Journal of Medicine, involving the potential dangers of COVID therapy with findings derived from the Surgisphere database. Hydroxychloroquine has garnered considerable media attention and was touted earlier by President Trump for its therapeutic effects.1 Naturally, there are political connotations associated with the agent, and it is unlikely that hydroxychloroquine will be supplanted in the near future as ongoing clinical trials have demonstrated mixed results amid the controversy.
As clinicians navigating unchartered territory within the hospital setting, we have to come to terms with these new challenges, tailoring treatment protocols accordingly with the best clinical practices in mind. Patients with preexisting mental health conditions and who are being treated for COVID-19 are particularly susceptible to clinical deterioration. Recent studies have indicated that psychiatric patients are more prone to feelings of isolation and/or estrangement as well as exacerbation of symptoms such as paranoia.2 Even more concerning is the medication regimen, namely, the novel combination therapies that arise when agents such as hydroxychloroquine are used in tandem with certain antipsychotics or antidepressants.
What’s at stake for COVID-19–positive mental health care patients?
Although the efficacy of hydroxychloroquine is currently being investigated,3 the antimalarial is usually prescribed in tandem with azithromycin for people with COVID-19. The National Institute of Allergy and Infectious Diseases has advised against that particular combination therapy because of ongoing concerns about toxicities.3,4
In another study, azithromycin was effectively substituted with doxycycline to help minimize systemic effects for patients with cardiac and/or pulmonary issues.5 Azithromycin is notorious in the literature for influencing the electrical activity of the heart with the potential for fatal arrhythmia and sudden cardiac death in individuals at risk for cardiovascular disease.5,6,7 It should be noted that both of these commonly prescribed COVID-19 medications (for example, hydroxychloroquine and azithromycin) could lead to QT interval prolongation especially within the context of combination therapy. This is largely concerning for psychiatrists and various other mental health practitioners for the following reasons: (1) higher rates of metabolic syndrome and cardiovascular diseases among psychiatric patients8 and/or (2) effects of certain antipsychotics (for example, IV haloperidol, thioridazine, and ziprasidone) and antidepressants (for example, citalopram and escitalopram) on the QT interval.9
SARS-CoV-2 and clinical judgment: Evaluating patients at higher risk
Although COVID-19 medication guidelines are still being actively developed, hydroxychloroquine appears to be commonly prescribed by physicians. The medication is known myriad untoward effects, including potential behavioral dysfunction (for example, irritability, agitation, suicidal ideation)10 as well as the aforementioned issues concerning arrhythmia (for example, torsades de pointes). Health care professionals might not have much control over the choice of COVID-19 agents because of a lack of available resources or limited options, but they can exercise clinical judgment with respect to selecting the appropriate psychotropic medications.
Treatment recommendations
1. Establish a baseline EKG
A baseline 12-lead EKG is the standard of care for patients currently being screened for COVID-19. It is necessary to rule out the presence of an underlying cardiovascular disease or a rhythm irregularity. A prolonged QTc interval is generally regarded as being around greater than 450-470 msecs with variations attributable to gender;11 numerous studies have affirmed that the risk of acquiring torsades de pointes is substantial when the QTc interval exceeds 500 msecs.12
2. Medical management and risk assessment
Commonly prescribed antipsychotics such as IV haloperidol and ziprasidone are known for exerting a negative effect on the interval and should readily be substituted with other agents in patients who are being treated for COVID-19; the combination of these antipsychotics alongside some COVID-19 medication regimens (for example, hydroxychloroquine/azithromycin) might prove to be fatal. The same logic applies to COVID-19 patients previously on antidepressant therapeutics such as citalopram and escitalopram.
3. Embrace an individually tailored approach to therapeutics
While American Psychiatric Association guidelines historically supported a cessation or reduction in the offending agent under normal circumstances,12 our team is recommending that the psychotropics associated with QTc interval prolongation are discontinued altogether (or substituted with a low-risk agent) in the event that a patient presents with suspected COVID-19. However, after the patients tests negative with COVID-19, they may resume therapy as indicated under the discretion of the mental health practitioner.
References
1. Offard C. “Lancet, NEJM Retract Surgisphere Studies on COVID-19 Patients.” The Scientist Magazine. 2020 Jun 4.
2. Shigemura J et al. Psychiatry Clin Neurosci. 2020 Apr;74(4):281-2.
3. Keshtkar-Jahromi M and Bavari S. Am J Trop Med Hyg. 2020 May;102(5):932-3.
4. Palca J. “NIH panel recommends against drug combination promoted by Trump for COVID-19.” NPR. 2020 Apr 21.
5. Mongelli L. “Long Island doctor tries new twist on hydroxychloroquine for elderly COVID-19 patients.” New York Post. 2020 Apr 4.
6. Hancox JC et al. Ther Adv Infect Dis. 2013 Oct;(5):155-65.
7. Giudicessi JR and Ackerman MJ. Cleve Clin J Med. 2013 Sep;80(9):539-44.
8. Casey DE. Am J Med. 2005 Apr 1;118(Suppl 2):15S-22S.
9. Beach SR et al. Psychosomatics. 2013 Jan 1;54(1):1-3.
10. Bogaczewicz A and Sobów T. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-4.
11. Chohan PS et al. Pak J Med Sci. 2015 Sep-Oct;31(5):1269-71.
12. Lieberman JA et al. APA guidance on the use of antipsychotic drugs and cardiac sudden death. NYS Office of Mental Health. 2012.
Dr. Faisal A. Islam is medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Faisal Islam disclosed no relevant financial relationships.
Dr. Mohammed Islam is affiliated with the department of psychiatry at the Interfaith Medical Center, New York. He disclosed no relevant financial relationships.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He disclosed no relevant financial relationships.