User login
Support for medical marijuana transcends political affiliation
There is not much common ground between Republicans and Democrats these days, but both sides strongly supported the use of medical marijuana in a recent survey by the American Society of Clinical Oncology.
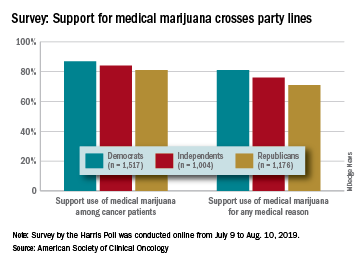
Overall support of medical marijuana among all 4,001 respondents was higher (84%) for use among cancer patients, but 76% also supported its use for any medical reason, according to data from the survey conducted for ASCO by the Harris Poll.
The differences in support between Republicans and Democrats were significant, but both parties were over 80% for marijuana use by cancer patients and over 70% for use for any medical reason. In both cases, the independents in between mirrored the overall population, with support at 84% and 76%, respectively, ASCO said.
Support for medical marijuana also was consistent based on the respondents’ cancer experience. For use by cancer patients, those who were current or previous patients were at 84%, caregivers (those providing unpaid care to an immediate family member or loved one with cancer) and other family members/loved ones were both at 87%, and those with no cancer experiences were at 82%, the survey results showed.
Use of marijuana for any medical reason was supported by 72% of current/previous patients, 79% of family members and loved ones, 80% of caregivers, and 74% of those with no cancer experience, ASCO reported.
In a question asked only of current or previous patients, 62% said that they are/were open to use of marijuana to alleviate cancer-related pain, nausea, or other symptoms, and 60% said that they wished they had more information about the benefits of medical marijuana use, according to the results of the survey, which was conducted online from July 9 to Aug. 10, 2019.
There is not much common ground between Republicans and Democrats these days, but both sides strongly supported the use of medical marijuana in a recent survey by the American Society of Clinical Oncology.

Overall support of medical marijuana among all 4,001 respondents was higher (84%) for use among cancer patients, but 76% also supported its use for any medical reason, according to data from the survey conducted for ASCO by the Harris Poll.
The differences in support between Republicans and Democrats were significant, but both parties were over 80% for marijuana use by cancer patients and over 70% for use for any medical reason. In both cases, the independents in between mirrored the overall population, with support at 84% and 76%, respectively, ASCO said.
Support for medical marijuana also was consistent based on the respondents’ cancer experience. For use by cancer patients, those who were current or previous patients were at 84%, caregivers (those providing unpaid care to an immediate family member or loved one with cancer) and other family members/loved ones were both at 87%, and those with no cancer experiences were at 82%, the survey results showed.
Use of marijuana for any medical reason was supported by 72% of current/previous patients, 79% of family members and loved ones, 80% of caregivers, and 74% of those with no cancer experience, ASCO reported.
In a question asked only of current or previous patients, 62% said that they are/were open to use of marijuana to alleviate cancer-related pain, nausea, or other symptoms, and 60% said that they wished they had more information about the benefits of medical marijuana use, according to the results of the survey, which was conducted online from July 9 to Aug. 10, 2019.
There is not much common ground between Republicans and Democrats these days, but both sides strongly supported the use of medical marijuana in a recent survey by the American Society of Clinical Oncology.

Overall support of medical marijuana among all 4,001 respondents was higher (84%) for use among cancer patients, but 76% also supported its use for any medical reason, according to data from the survey conducted for ASCO by the Harris Poll.
The differences in support between Republicans and Democrats were significant, but both parties were over 80% for marijuana use by cancer patients and over 70% for use for any medical reason. In both cases, the independents in between mirrored the overall population, with support at 84% and 76%, respectively, ASCO said.
Support for medical marijuana also was consistent based on the respondents’ cancer experience. For use by cancer patients, those who were current or previous patients were at 84%, caregivers (those providing unpaid care to an immediate family member or loved one with cancer) and other family members/loved ones were both at 87%, and those with no cancer experiences were at 82%, the survey results showed.
Use of marijuana for any medical reason was supported by 72% of current/previous patients, 79% of family members and loved ones, 80% of caregivers, and 74% of those with no cancer experience, ASCO reported.
In a question asked only of current or previous patients, 62% said that they are/were open to use of marijuana to alleviate cancer-related pain, nausea, or other symptoms, and 60% said that they wished they had more information about the benefits of medical marijuana use, according to the results of the survey, which was conducted online from July 9 to Aug. 10, 2019.
Researchers seek a way to predict cognitive deficits in children treated for ALL
Researchers are attempting to determine, early in the treatment process, which children with acute lymphoblastic leukemia (ALL) have an increased risk of neurocognitive deficits after chemotherapy.
The goal of the researchers’ project (5R01CA220568-02) is to determine if gene variants and biomarkers associated with oxidative stress, neuroinflammation, and folate physiology correlate with cognitive decline during and after chemotherapy. Ideally, certain variants and biomarkers will reveal patients who might benefit from interventions to prevent or even reverse cognitive deficits.
Peter D. Cole, MD, of Rutgers Cancer Institute, New Brunswick, N.J., and colleagues are conducting this research in patients from the DFCI-16-001 trial (NCT03020030). This multicenter, phase 3 study is enrolling patients (aged 1-21 years) with B- or T-cell ALL who then receive a multidrug chemotherapy regimen.
Dr. Cole and colleagues are analyzing a subset of patients from the trial, looking for relationships between chemotherapy-induced neurocognitive changes, gene variants, and changes in biomarkers detected in cerebrospinal fluid (CSF).
“We’re looking at a broad panel of target gene variants that are associated with either drug metabolism, defenses against oxidative stress, neuroinflammation, or folate physiology,” Dr. Cole said in an interview.
This includes variants Dr. Cole and colleagues identified in a previous, retrospective study of ALL survivors. The researchers found that survivors who were homozygous for NOS3 894T, had a variant SLCO2A1 G allele, or had at least one GSTP1 T allele were more likely to exhibit cognitive deficits (J Clin Oncol. 2015 Jul 1;33[19]:2205-11).
The researchers are also analyzing CSF samples, looking for changes in tau protein, homocysteine, homocysteic acid, the adenosylmethionine to adenosylhomocysteine ratio, and other biomarkers of oxidative stress, neuroinflammation, and folate physiology. The CSF is collected at five time points: the start of chemotherapy, day 18, the start of first consolidation, the end of first consolidation, and 7 weeks later in second consolidation.
Cognitive testing
While Dr. Cole is leading the genetic and biomarker analyses, Stephen A. Sands, PsyD, of Memorial Sloan Kettering Cancer Center in New York, is leading the cognitive testing.
The researchers are evaluating patients for cognitive decline using computerized tests from a company called Cogstate. The tests are designed to assess functions such as processing speed, attention, visual learning, and working memory. The tests are administered on an iPad and involve tasks like identifying features of playing cards and finding the correct way through a maze.
The patients – aged 3 years and older – undergo cognitive testing at six time points: baseline, which is any time between days 8 and 32 of induction (except within 72 hours after sedation or anesthesia); at first consolidation; the end of central nervous system therapy; 1 year into chemotherapy; the end of chemotherapy; and 1 year after chemotherapy ends.
In a prior study, Cogstate testing proved reliable for detecting neurocognitive changes in patients undergoing treatment for ALL (Support Care Cancer. 2017;25[2]:449-57). In the current study, the researchers are supplementing Cogstate test results with Wechsler IQ tests administered 1 year after patients complete chemotherapy.
Dr. Sands noted that Cogstate tests provide benefits over the Wechsler “paper-and-pencil” tests. One benefit is that Cogstate tests can be given more often without inducing practice effects (J Clin Exp Neuropsychol. 2006 Oct;28[7]:1095-112). Another is that Cogstate tests can be administered by anyone with a bachelor’s degree who has undergone the appropriate training, while Wechsler IQ tests must be given by psychologists.
Preliminary results
This research is ongoing, so it’s too early to announce any discoveries, but the study is moving along as planned.
“The preliminary data we have so far are demonstrating the validity of the study,” Dr. Cole said. “Things are going well. We’re able to do the cognitive testing and collect the samples that we need and ship them without losing the integrity of the samples.”
Dr. Sands noted that enrollment has been encouraging. As this is a substudy of DFCI-16-001, the researchers must obtain consent separately from the main study. Dr. Sands said about 89% of parents involved in the main study have agreed to enroll their children in the substudy.
Dr. Sands also said that early results from Cogstate testing have revealed patients who are experiencing cognitive decline during treatment. The researchers still have to determine if these results correlate with any biomarkers or gene variants.
Potential interventions
If the researchers can pinpoint patients at risk for cognitive deficits, the next step will be to investigate pharmacologic and behavioral interventions.
Dr. Cole said he is particularly interested in treatments that reduce oxidative stress, such as dextromethorphan and memantine. Dextromethorphan has been shown to resolve symptoms of methotrexate-induced neurotoxicity in patients (Pediatr Hematol Oncol. 2002 Jul-Aug;19[5]:319-27), and memantine reduced memory deficits in animals treated with methotrexate (Clin Cancer Res. 2013 Aug 15;19[16]:4446-54).
“Memantine hasn’t been used in kids with leukemia yet, but it’s something that I’d like to see brought to a clinical trial,” Dr. Cole said.
Dr. Sands pointed to other potential pharmacologic interventions, including the stimulants methylphenidate and modafinil. Both drugs have been shown to improve cognitive deficits in cancer survivors (J Clin Oncol. 2001 Mar 15;19[6]:1802-8; Cancer. 2009 Jun 15; 115[12]: 2605-16).
Computer-based cognitive training tools may be another option. One such tool, Lumosity, improved executive functions in a study of breast cancer survivors (Clin Breast Cancer. 2013 Aug;13[4]:299-306). Another tool, CogMed, improved working memory in survivors of brain tumors and ALL (Psychooncology. 2013 Aug; 22[8]: 1856-65).
Other behavioral interventions might include sleep hygiene and exercise. Sleep hygiene has been shown to improve cognitive function in childhood cancer survivors (Cancer. 2011 Jun 1;117[11]:2559-68), and a recent study revealed an association between exercise intolerance and negative neurocognitive outcomes in ALL survivors (Cancer. 2019 Oct 21. doi: 10.1002/cncr.32510).
“What we need to figure out is which children will respond to which interventions,” Dr. Sands said, adding that interventions will likely need to be combined.
“It’s not going to be one thing that will work for everybody,” he said. “It’s going to be: What packages of things will work for different people?”
Dr. Sands and Dr. Cole reported having no relevant financial disclosures.
Researchers are attempting to determine, early in the treatment process, which children with acute lymphoblastic leukemia (ALL) have an increased risk of neurocognitive deficits after chemotherapy.
The goal of the researchers’ project (5R01CA220568-02) is to determine if gene variants and biomarkers associated with oxidative stress, neuroinflammation, and folate physiology correlate with cognitive decline during and after chemotherapy. Ideally, certain variants and biomarkers will reveal patients who might benefit from interventions to prevent or even reverse cognitive deficits.
Peter D. Cole, MD, of Rutgers Cancer Institute, New Brunswick, N.J., and colleagues are conducting this research in patients from the DFCI-16-001 trial (NCT03020030). This multicenter, phase 3 study is enrolling patients (aged 1-21 years) with B- or T-cell ALL who then receive a multidrug chemotherapy regimen.
Dr. Cole and colleagues are analyzing a subset of patients from the trial, looking for relationships between chemotherapy-induced neurocognitive changes, gene variants, and changes in biomarkers detected in cerebrospinal fluid (CSF).
“We’re looking at a broad panel of target gene variants that are associated with either drug metabolism, defenses against oxidative stress, neuroinflammation, or folate physiology,” Dr. Cole said in an interview.
This includes variants Dr. Cole and colleagues identified in a previous, retrospective study of ALL survivors. The researchers found that survivors who were homozygous for NOS3 894T, had a variant SLCO2A1 G allele, or had at least one GSTP1 T allele were more likely to exhibit cognitive deficits (J Clin Oncol. 2015 Jul 1;33[19]:2205-11).
The researchers are also analyzing CSF samples, looking for changes in tau protein, homocysteine, homocysteic acid, the adenosylmethionine to adenosylhomocysteine ratio, and other biomarkers of oxidative stress, neuroinflammation, and folate physiology. The CSF is collected at five time points: the start of chemotherapy, day 18, the start of first consolidation, the end of first consolidation, and 7 weeks later in second consolidation.
Cognitive testing
While Dr. Cole is leading the genetic and biomarker analyses, Stephen A. Sands, PsyD, of Memorial Sloan Kettering Cancer Center in New York, is leading the cognitive testing.
The researchers are evaluating patients for cognitive decline using computerized tests from a company called Cogstate. The tests are designed to assess functions such as processing speed, attention, visual learning, and working memory. The tests are administered on an iPad and involve tasks like identifying features of playing cards and finding the correct way through a maze.
The patients – aged 3 years and older – undergo cognitive testing at six time points: baseline, which is any time between days 8 and 32 of induction (except within 72 hours after sedation or anesthesia); at first consolidation; the end of central nervous system therapy; 1 year into chemotherapy; the end of chemotherapy; and 1 year after chemotherapy ends.
In a prior study, Cogstate testing proved reliable for detecting neurocognitive changes in patients undergoing treatment for ALL (Support Care Cancer. 2017;25[2]:449-57). In the current study, the researchers are supplementing Cogstate test results with Wechsler IQ tests administered 1 year after patients complete chemotherapy.
Dr. Sands noted that Cogstate tests provide benefits over the Wechsler “paper-and-pencil” tests. One benefit is that Cogstate tests can be given more often without inducing practice effects (J Clin Exp Neuropsychol. 2006 Oct;28[7]:1095-112). Another is that Cogstate tests can be administered by anyone with a bachelor’s degree who has undergone the appropriate training, while Wechsler IQ tests must be given by psychologists.
Preliminary results
This research is ongoing, so it’s too early to announce any discoveries, but the study is moving along as planned.
“The preliminary data we have so far are demonstrating the validity of the study,” Dr. Cole said. “Things are going well. We’re able to do the cognitive testing and collect the samples that we need and ship them without losing the integrity of the samples.”
Dr. Sands noted that enrollment has been encouraging. As this is a substudy of DFCI-16-001, the researchers must obtain consent separately from the main study. Dr. Sands said about 89% of parents involved in the main study have agreed to enroll their children in the substudy.
Dr. Sands also said that early results from Cogstate testing have revealed patients who are experiencing cognitive decline during treatment. The researchers still have to determine if these results correlate with any biomarkers or gene variants.
Potential interventions
If the researchers can pinpoint patients at risk for cognitive deficits, the next step will be to investigate pharmacologic and behavioral interventions.
Dr. Cole said he is particularly interested in treatments that reduce oxidative stress, such as dextromethorphan and memantine. Dextromethorphan has been shown to resolve symptoms of methotrexate-induced neurotoxicity in patients (Pediatr Hematol Oncol. 2002 Jul-Aug;19[5]:319-27), and memantine reduced memory deficits in animals treated with methotrexate (Clin Cancer Res. 2013 Aug 15;19[16]:4446-54).
“Memantine hasn’t been used in kids with leukemia yet, but it’s something that I’d like to see brought to a clinical trial,” Dr. Cole said.
Dr. Sands pointed to other potential pharmacologic interventions, including the stimulants methylphenidate and modafinil. Both drugs have been shown to improve cognitive deficits in cancer survivors (J Clin Oncol. 2001 Mar 15;19[6]:1802-8; Cancer. 2009 Jun 15; 115[12]: 2605-16).
Computer-based cognitive training tools may be another option. One such tool, Lumosity, improved executive functions in a study of breast cancer survivors (Clin Breast Cancer. 2013 Aug;13[4]:299-306). Another tool, CogMed, improved working memory in survivors of brain tumors and ALL (Psychooncology. 2013 Aug; 22[8]: 1856-65).
Other behavioral interventions might include sleep hygiene and exercise. Sleep hygiene has been shown to improve cognitive function in childhood cancer survivors (Cancer. 2011 Jun 1;117[11]:2559-68), and a recent study revealed an association between exercise intolerance and negative neurocognitive outcomes in ALL survivors (Cancer. 2019 Oct 21. doi: 10.1002/cncr.32510).
“What we need to figure out is which children will respond to which interventions,” Dr. Sands said, adding that interventions will likely need to be combined.
“It’s not going to be one thing that will work for everybody,” he said. “It’s going to be: What packages of things will work for different people?”
Dr. Sands and Dr. Cole reported having no relevant financial disclosures.
Researchers are attempting to determine, early in the treatment process, which children with acute lymphoblastic leukemia (ALL) have an increased risk of neurocognitive deficits after chemotherapy.
The goal of the researchers’ project (5R01CA220568-02) is to determine if gene variants and biomarkers associated with oxidative stress, neuroinflammation, and folate physiology correlate with cognitive decline during and after chemotherapy. Ideally, certain variants and biomarkers will reveal patients who might benefit from interventions to prevent or even reverse cognitive deficits.
Peter D. Cole, MD, of Rutgers Cancer Institute, New Brunswick, N.J., and colleagues are conducting this research in patients from the DFCI-16-001 trial (NCT03020030). This multicenter, phase 3 study is enrolling patients (aged 1-21 years) with B- or T-cell ALL who then receive a multidrug chemotherapy regimen.
Dr. Cole and colleagues are analyzing a subset of patients from the trial, looking for relationships between chemotherapy-induced neurocognitive changes, gene variants, and changes in biomarkers detected in cerebrospinal fluid (CSF).
“We’re looking at a broad panel of target gene variants that are associated with either drug metabolism, defenses against oxidative stress, neuroinflammation, or folate physiology,” Dr. Cole said in an interview.
This includes variants Dr. Cole and colleagues identified in a previous, retrospective study of ALL survivors. The researchers found that survivors who were homozygous for NOS3 894T, had a variant SLCO2A1 G allele, or had at least one GSTP1 T allele were more likely to exhibit cognitive deficits (J Clin Oncol. 2015 Jul 1;33[19]:2205-11).
The researchers are also analyzing CSF samples, looking for changes in tau protein, homocysteine, homocysteic acid, the adenosylmethionine to adenosylhomocysteine ratio, and other biomarkers of oxidative stress, neuroinflammation, and folate physiology. The CSF is collected at five time points: the start of chemotherapy, day 18, the start of first consolidation, the end of first consolidation, and 7 weeks later in second consolidation.
Cognitive testing
While Dr. Cole is leading the genetic and biomarker analyses, Stephen A. Sands, PsyD, of Memorial Sloan Kettering Cancer Center in New York, is leading the cognitive testing.
The researchers are evaluating patients for cognitive decline using computerized tests from a company called Cogstate. The tests are designed to assess functions such as processing speed, attention, visual learning, and working memory. The tests are administered on an iPad and involve tasks like identifying features of playing cards and finding the correct way through a maze.
The patients – aged 3 years and older – undergo cognitive testing at six time points: baseline, which is any time between days 8 and 32 of induction (except within 72 hours after sedation or anesthesia); at first consolidation; the end of central nervous system therapy; 1 year into chemotherapy; the end of chemotherapy; and 1 year after chemotherapy ends.
In a prior study, Cogstate testing proved reliable for detecting neurocognitive changes in patients undergoing treatment for ALL (Support Care Cancer. 2017;25[2]:449-57). In the current study, the researchers are supplementing Cogstate test results with Wechsler IQ tests administered 1 year after patients complete chemotherapy.
Dr. Sands noted that Cogstate tests provide benefits over the Wechsler “paper-and-pencil” tests. One benefit is that Cogstate tests can be given more often without inducing practice effects (J Clin Exp Neuropsychol. 2006 Oct;28[7]:1095-112). Another is that Cogstate tests can be administered by anyone with a bachelor’s degree who has undergone the appropriate training, while Wechsler IQ tests must be given by psychologists.
Preliminary results
This research is ongoing, so it’s too early to announce any discoveries, but the study is moving along as planned.
“The preliminary data we have so far are demonstrating the validity of the study,” Dr. Cole said. “Things are going well. We’re able to do the cognitive testing and collect the samples that we need and ship them without losing the integrity of the samples.”
Dr. Sands noted that enrollment has been encouraging. As this is a substudy of DFCI-16-001, the researchers must obtain consent separately from the main study. Dr. Sands said about 89% of parents involved in the main study have agreed to enroll their children in the substudy.
Dr. Sands also said that early results from Cogstate testing have revealed patients who are experiencing cognitive decline during treatment. The researchers still have to determine if these results correlate with any biomarkers or gene variants.
Potential interventions
If the researchers can pinpoint patients at risk for cognitive deficits, the next step will be to investigate pharmacologic and behavioral interventions.
Dr. Cole said he is particularly interested in treatments that reduce oxidative stress, such as dextromethorphan and memantine. Dextromethorphan has been shown to resolve symptoms of methotrexate-induced neurotoxicity in patients (Pediatr Hematol Oncol. 2002 Jul-Aug;19[5]:319-27), and memantine reduced memory deficits in animals treated with methotrexate (Clin Cancer Res. 2013 Aug 15;19[16]:4446-54).
“Memantine hasn’t been used in kids with leukemia yet, but it’s something that I’d like to see brought to a clinical trial,” Dr. Cole said.
Dr. Sands pointed to other potential pharmacologic interventions, including the stimulants methylphenidate and modafinil. Both drugs have been shown to improve cognitive deficits in cancer survivors (J Clin Oncol. 2001 Mar 15;19[6]:1802-8; Cancer. 2009 Jun 15; 115[12]: 2605-16).
Computer-based cognitive training tools may be another option. One such tool, Lumosity, improved executive functions in a study of breast cancer survivors (Clin Breast Cancer. 2013 Aug;13[4]:299-306). Another tool, CogMed, improved working memory in survivors of brain tumors and ALL (Psychooncology. 2013 Aug; 22[8]: 1856-65).
Other behavioral interventions might include sleep hygiene and exercise. Sleep hygiene has been shown to improve cognitive function in childhood cancer survivors (Cancer. 2011 Jun 1;117[11]:2559-68), and a recent study revealed an association between exercise intolerance and negative neurocognitive outcomes in ALL survivors (Cancer. 2019 Oct 21. doi: 10.1002/cncr.32510).
“What we need to figure out is which children will respond to which interventions,” Dr. Sands said, adding that interventions will likely need to be combined.
“It’s not going to be one thing that will work for everybody,” he said. “It’s going to be: What packages of things will work for different people?”
Dr. Sands and Dr. Cole reported having no relevant financial disclosures.
Study detects lower CV risk in breast cancer survivors
Breast cancer survivors may have a lower prevalence of cardiac risk factors at the time of first myocardial infarction and better outcomes compared with those having a first MI from the general population, according to findings from a retrospective study.
In addition, women without breast cancer were younger at the time of first MI compared with survivors, wrote Srikanth Yandrapalli, MD, of New York Medical College, Valhalla, and colleagues. Their report is in the American Journal of Medicine.
The researchers identified 1,644,032 women with a first MI, 56,842 of whom were breast cancer survivors. The team evaluated differences in the prevalence of cardiac risk factors and related outcomes in breast cancer survivors in comparison to the general population.
At baseline, the mean age of subjects with a history of breast cancer was 77 years (range, 11 years), while the mean age of women without breast cancer was 71 years (range, 15 years).
Clinical data were collected from the United States National Inpatient Sample for January 2005 to September 2015. Other outcomes assessed were differences in baseline characteristics and the rate of in-hospital mortality in both groups.
After analysis, the researchers found that breast cancer survivors had a lower prevalence of diabetes mellitus (30.1% vs. 33.1%), obesity (9.4% vs. 13.0%), and smoking (24.1% vs. 27.0%), but higher rates of dyslipidemia (52.7% vs. 48.4%) and hypertension (73.6% vs. 68.1%), compared with women without breast cancer (All P less than .001).
With respect to age, women without breast cancer were 6 years younger than breast cancer survivors at the time of first acute MI (mean age, 71 vs. 77 years; P less than .001).
In addition, the rate of in-hospital mortality was higher in women without breast cancer (7.9%) compared with survivors (7.1%) (P less than .001). After risk adjustment, these results remained unchanged (odds ratio, 0.89; 95% confidence interval, 0.82-0.94).
“Breast cancer survivors in the U.S. are at least 6 years older than the general population of women without breast cancer, and they had a favorable cardiac risk factor profile at the time of first myocardial infarction,” Dr. Yandrapalli and colleagues explained. “The reason for these findings are unclear and hypothesis generating,” they added.
The researchers acknowledged that a key limitation of the study was the retrospective design. As a result, the potential effects of residual confounding should be considered when interpreting the results.
“The favorable impact of health education and participation in cancer survivorship programs on these observed differences in breast cancer survivors should be further explored,” they concluded.
No funding sources were reported. The authors reported having no conflicts of interest.
SOURCE: Yandrapalli S et al. Am J Med. 2019 Nov 9. doi: 10.1016/j.amjmed.2019.10.018.
Breast cancer survivors may have a lower prevalence of cardiac risk factors at the time of first myocardial infarction and better outcomes compared with those having a first MI from the general population, according to findings from a retrospective study.
In addition, women without breast cancer were younger at the time of first MI compared with survivors, wrote Srikanth Yandrapalli, MD, of New York Medical College, Valhalla, and colleagues. Their report is in the American Journal of Medicine.
The researchers identified 1,644,032 women with a first MI, 56,842 of whom were breast cancer survivors. The team evaluated differences in the prevalence of cardiac risk factors and related outcomes in breast cancer survivors in comparison to the general population.
At baseline, the mean age of subjects with a history of breast cancer was 77 years (range, 11 years), while the mean age of women without breast cancer was 71 years (range, 15 years).
Clinical data were collected from the United States National Inpatient Sample for January 2005 to September 2015. Other outcomes assessed were differences in baseline characteristics and the rate of in-hospital mortality in both groups.
After analysis, the researchers found that breast cancer survivors had a lower prevalence of diabetes mellitus (30.1% vs. 33.1%), obesity (9.4% vs. 13.0%), and smoking (24.1% vs. 27.0%), but higher rates of dyslipidemia (52.7% vs. 48.4%) and hypertension (73.6% vs. 68.1%), compared with women without breast cancer (All P less than .001).
With respect to age, women without breast cancer were 6 years younger than breast cancer survivors at the time of first acute MI (mean age, 71 vs. 77 years; P less than .001).
In addition, the rate of in-hospital mortality was higher in women without breast cancer (7.9%) compared with survivors (7.1%) (P less than .001). After risk adjustment, these results remained unchanged (odds ratio, 0.89; 95% confidence interval, 0.82-0.94).
“Breast cancer survivors in the U.S. are at least 6 years older than the general population of women without breast cancer, and they had a favorable cardiac risk factor profile at the time of first myocardial infarction,” Dr. Yandrapalli and colleagues explained. “The reason for these findings are unclear and hypothesis generating,” they added.
The researchers acknowledged that a key limitation of the study was the retrospective design. As a result, the potential effects of residual confounding should be considered when interpreting the results.
“The favorable impact of health education and participation in cancer survivorship programs on these observed differences in breast cancer survivors should be further explored,” they concluded.
No funding sources were reported. The authors reported having no conflicts of interest.
SOURCE: Yandrapalli S et al. Am J Med. 2019 Nov 9. doi: 10.1016/j.amjmed.2019.10.018.
Breast cancer survivors may have a lower prevalence of cardiac risk factors at the time of first myocardial infarction and better outcomes compared with those having a first MI from the general population, according to findings from a retrospective study.
In addition, women without breast cancer were younger at the time of first MI compared with survivors, wrote Srikanth Yandrapalli, MD, of New York Medical College, Valhalla, and colleagues. Their report is in the American Journal of Medicine.
The researchers identified 1,644,032 women with a first MI, 56,842 of whom were breast cancer survivors. The team evaluated differences in the prevalence of cardiac risk factors and related outcomes in breast cancer survivors in comparison to the general population.
At baseline, the mean age of subjects with a history of breast cancer was 77 years (range, 11 years), while the mean age of women without breast cancer was 71 years (range, 15 years).
Clinical data were collected from the United States National Inpatient Sample for January 2005 to September 2015. Other outcomes assessed were differences in baseline characteristics and the rate of in-hospital mortality in both groups.
After analysis, the researchers found that breast cancer survivors had a lower prevalence of diabetes mellitus (30.1% vs. 33.1%), obesity (9.4% vs. 13.0%), and smoking (24.1% vs. 27.0%), but higher rates of dyslipidemia (52.7% vs. 48.4%) and hypertension (73.6% vs. 68.1%), compared with women without breast cancer (All P less than .001).
With respect to age, women without breast cancer were 6 years younger than breast cancer survivors at the time of first acute MI (mean age, 71 vs. 77 years; P less than .001).
In addition, the rate of in-hospital mortality was higher in women without breast cancer (7.9%) compared with survivors (7.1%) (P less than .001). After risk adjustment, these results remained unchanged (odds ratio, 0.89; 95% confidence interval, 0.82-0.94).
“Breast cancer survivors in the U.S. are at least 6 years older than the general population of women without breast cancer, and they had a favorable cardiac risk factor profile at the time of first myocardial infarction,” Dr. Yandrapalli and colleagues explained. “The reason for these findings are unclear and hypothesis generating,” they added.
The researchers acknowledged that a key limitation of the study was the retrospective design. As a result, the potential effects of residual confounding should be considered when interpreting the results.
“The favorable impact of health education and participation in cancer survivorship programs on these observed differences in breast cancer survivors should be further explored,” they concluded.
No funding sources were reported. The authors reported having no conflicts of interest.
SOURCE: Yandrapalli S et al. Am J Med. 2019 Nov 9. doi: 10.1016/j.amjmed.2019.10.018.
FROM THE AMERICAN JOURNAL OF MEDICINE
Foundation launches direct-to-patient registry in multiple myeloma
The Multiple Myeloma Research Foundation (MMRF) recently launched its Direct-to-Patient registry, in what the organization’s leaders are describing as a “disruptive” step toward improving outcomes for patients with multiple myeloma.
The new registry is intended to build upon CoMMpass, a program started 8 years ago that now represents the largest genomic database of any type of cancer. Although CoMMpass includes data from about 1,150 patients with myeloma, it’s not enough information, according to the chief marketing and development officer at the MMRF, Anne Quinn Young.
“For a disease as heterogenous as myeloma is, we need more, particularly because we don’t have all the samples for later-stage disease,” Ms. Quinn Young said in an interview. “And even with the clinical data, given the patient population, both [in terms of] demographics and the nature of the disease, the numbers of patients still living after multiple relapses is rather small.”
In an earlier effort to gather more data, the MMRF first turned to other organizations for help, but this approach fell short because of scarcity of data, and in some cases, unwillingness to share. Steven Labkoff, MD, chief data officer at the MMRF, described this experience in an interview.
“When the MMRF was looking around for different data sources for myeloma data, it was always the claim that, ‘Sure, we have plenty of patients, we have plenty of data, and it’s rich and really complete.’ However, as we approached an array of organizations – big organizations – as we dug into the details and reviewed patient counts or data completeness, they either didn’t have a sufficient number of patients, they didn’t have sufficiently complete data for our needs, and in the case where some did have sufficient numbers and complete data sets, they simply weren’t in a position to share that data outside their institution,” Dr. Labkoff said.
Undeterred, the MMRF switched tactics to the current, patient-centric approach.
“We’re leveraging one aspect of the HIPAA legislation,” Dr. Labkoff said, referring to patients’ rights to request their own medical records and an institution’s legal obligation to provide those records.
In the short-term, the registry will collect three types of data: patient donated data (answers from a patient survey), electronic medical records abstracted from all relevant past providers, and genomic test results. Participating patients will have blood drawn at home by a phlebotomist for the genomic assay. Additional tubes of blood will be concurrently collected and biobanked. This will eventually allow for immune profiling, Dr. Labkoff said.
Future goals include a patient-reported outcomes module and the ability to link data with medical claims.
So far, 79 patients have participated in the pilot program, according to the MMRF. As the database builds, Ms. Quinn Young and Dr. Labkoff anticipate that it will yield answers to a variety of real-world questions.
Dr. Labkoff offered two examples. “Of all the patients who have been exposed to ‘name your drug,’ what were the costs of their therapy, and what were the outcomes?” he said. In addition, researchers will be able to query clinical trial inclusion criteria to search for data on a specific patient profile, such as patients with a 4:14 translocation, who have had a bone marrow transplant in the last 2 years, and have been exposed to a certain drug regimen.
Ms. Quinn Young noted that doctors may be able to use the database to reliably identify high-risk patients and guide agent selection. Common patient questions also will be addressed, she said, including best treatment regimens for certain types of patients.
“For patients who may have run out of all commercially available options, or for patients who are perhaps seen at a community center, where certainly this type of profiling is not standard, it’s opening up a whole new set of options for them,” Ms. Quinn Young said. “And if their physician doesn’t pursue those options, they have the report that they can use to seek a second opinion.”
The Direct-to-Patient registry is unique because it aims to empower patients in a way that hasn’t been done before, Ms. Quinn Young said. “We are committed ... ever since we conceived of this project, to giving results back to patients. That is disruptive because right now that doesn’t exist.”
But the cost of implementing the registry, which has an approximate budget of $20 million, stands in the way of a completely free flow of anonymized data. MMRF leaders are exploring different strategies to sustain funding for the program.
Another MMRF program, CoMMpass, uses a precompetitive consortium model, in which several pharmaceutical companies pay for a preview of the data 6 months in advance of nonprofit researchers. A similar model may be used with the Direct-to-Patient registry, but this has yet to be determined, according to Dr. Labkoff and Ms. Quinn Young.
For now, Ms. Quinn Young said she hopes that physicians will be receptive to the program. “[The short term goal is that] when patients come to their doctors asking about this, that there is support and open-mindedness,” she said.
Looking to the future, Dr. Labkoff described how the registry could accelerate myeloma research, ultimately toward a cure.
“It is generally accepted that it can take 17 years to get something – a therapy, a new drug, or a guideline – from the bench to the bedside,” he said. “It’s my hope that we can take next generation sequencing and the results of this registry and bend that curve, maybe ... to 10 [years], or very aggressively, to 7 or 5 [years], where doctors are able to use the information in these reports for the patients that have literally given themselves, and use this to help guide the choices of their therapy or the trials they apply for, to help them get a better outcome in general.”
The Direct-to-Patient registry is a collaborative effort between the MMRF and multiple organizations, including the health care technology company COTA, the Broad Institute of Harvard and MIT, Prometheus Research, Tempus, and the Dana Farber Cancer Institute.
The Multiple Myeloma Research Foundation (MMRF) recently launched its Direct-to-Patient registry, in what the organization’s leaders are describing as a “disruptive” step toward improving outcomes for patients with multiple myeloma.
The new registry is intended to build upon CoMMpass, a program started 8 years ago that now represents the largest genomic database of any type of cancer. Although CoMMpass includes data from about 1,150 patients with myeloma, it’s not enough information, according to the chief marketing and development officer at the MMRF, Anne Quinn Young.
“For a disease as heterogenous as myeloma is, we need more, particularly because we don’t have all the samples for later-stage disease,” Ms. Quinn Young said in an interview. “And even with the clinical data, given the patient population, both [in terms of] demographics and the nature of the disease, the numbers of patients still living after multiple relapses is rather small.”
In an earlier effort to gather more data, the MMRF first turned to other organizations for help, but this approach fell short because of scarcity of data, and in some cases, unwillingness to share. Steven Labkoff, MD, chief data officer at the MMRF, described this experience in an interview.
“When the MMRF was looking around for different data sources for myeloma data, it was always the claim that, ‘Sure, we have plenty of patients, we have plenty of data, and it’s rich and really complete.’ However, as we approached an array of organizations – big organizations – as we dug into the details and reviewed patient counts or data completeness, they either didn’t have a sufficient number of patients, they didn’t have sufficiently complete data for our needs, and in the case where some did have sufficient numbers and complete data sets, they simply weren’t in a position to share that data outside their institution,” Dr. Labkoff said.
Undeterred, the MMRF switched tactics to the current, patient-centric approach.
“We’re leveraging one aspect of the HIPAA legislation,” Dr. Labkoff said, referring to patients’ rights to request their own medical records and an institution’s legal obligation to provide those records.
In the short-term, the registry will collect three types of data: patient donated data (answers from a patient survey), electronic medical records abstracted from all relevant past providers, and genomic test results. Participating patients will have blood drawn at home by a phlebotomist for the genomic assay. Additional tubes of blood will be concurrently collected and biobanked. This will eventually allow for immune profiling, Dr. Labkoff said.
Future goals include a patient-reported outcomes module and the ability to link data with medical claims.
So far, 79 patients have participated in the pilot program, according to the MMRF. As the database builds, Ms. Quinn Young and Dr. Labkoff anticipate that it will yield answers to a variety of real-world questions.
Dr. Labkoff offered two examples. “Of all the patients who have been exposed to ‘name your drug,’ what were the costs of their therapy, and what were the outcomes?” he said. In addition, researchers will be able to query clinical trial inclusion criteria to search for data on a specific patient profile, such as patients with a 4:14 translocation, who have had a bone marrow transplant in the last 2 years, and have been exposed to a certain drug regimen.
Ms. Quinn Young noted that doctors may be able to use the database to reliably identify high-risk patients and guide agent selection. Common patient questions also will be addressed, she said, including best treatment regimens for certain types of patients.
“For patients who may have run out of all commercially available options, or for patients who are perhaps seen at a community center, where certainly this type of profiling is not standard, it’s opening up a whole new set of options for them,” Ms. Quinn Young said. “And if their physician doesn’t pursue those options, they have the report that they can use to seek a second opinion.”
The Direct-to-Patient registry is unique because it aims to empower patients in a way that hasn’t been done before, Ms. Quinn Young said. “We are committed ... ever since we conceived of this project, to giving results back to patients. That is disruptive because right now that doesn’t exist.”
But the cost of implementing the registry, which has an approximate budget of $20 million, stands in the way of a completely free flow of anonymized data. MMRF leaders are exploring different strategies to sustain funding for the program.
Another MMRF program, CoMMpass, uses a precompetitive consortium model, in which several pharmaceutical companies pay for a preview of the data 6 months in advance of nonprofit researchers. A similar model may be used with the Direct-to-Patient registry, but this has yet to be determined, according to Dr. Labkoff and Ms. Quinn Young.
For now, Ms. Quinn Young said she hopes that physicians will be receptive to the program. “[The short term goal is that] when patients come to their doctors asking about this, that there is support and open-mindedness,” she said.
Looking to the future, Dr. Labkoff described how the registry could accelerate myeloma research, ultimately toward a cure.
“It is generally accepted that it can take 17 years to get something – a therapy, a new drug, or a guideline – from the bench to the bedside,” he said. “It’s my hope that we can take next generation sequencing and the results of this registry and bend that curve, maybe ... to 10 [years], or very aggressively, to 7 or 5 [years], where doctors are able to use the information in these reports for the patients that have literally given themselves, and use this to help guide the choices of their therapy or the trials they apply for, to help them get a better outcome in general.”
The Direct-to-Patient registry is a collaborative effort between the MMRF and multiple organizations, including the health care technology company COTA, the Broad Institute of Harvard and MIT, Prometheus Research, Tempus, and the Dana Farber Cancer Institute.
The Multiple Myeloma Research Foundation (MMRF) recently launched its Direct-to-Patient registry, in what the organization’s leaders are describing as a “disruptive” step toward improving outcomes for patients with multiple myeloma.
The new registry is intended to build upon CoMMpass, a program started 8 years ago that now represents the largest genomic database of any type of cancer. Although CoMMpass includes data from about 1,150 patients with myeloma, it’s not enough information, according to the chief marketing and development officer at the MMRF, Anne Quinn Young.
“For a disease as heterogenous as myeloma is, we need more, particularly because we don’t have all the samples for later-stage disease,” Ms. Quinn Young said in an interview. “And even with the clinical data, given the patient population, both [in terms of] demographics and the nature of the disease, the numbers of patients still living after multiple relapses is rather small.”
In an earlier effort to gather more data, the MMRF first turned to other organizations for help, but this approach fell short because of scarcity of data, and in some cases, unwillingness to share. Steven Labkoff, MD, chief data officer at the MMRF, described this experience in an interview.
“When the MMRF was looking around for different data sources for myeloma data, it was always the claim that, ‘Sure, we have plenty of patients, we have plenty of data, and it’s rich and really complete.’ However, as we approached an array of organizations – big organizations – as we dug into the details and reviewed patient counts or data completeness, they either didn’t have a sufficient number of patients, they didn’t have sufficiently complete data for our needs, and in the case where some did have sufficient numbers and complete data sets, they simply weren’t in a position to share that data outside their institution,” Dr. Labkoff said.
Undeterred, the MMRF switched tactics to the current, patient-centric approach.
“We’re leveraging one aspect of the HIPAA legislation,” Dr. Labkoff said, referring to patients’ rights to request their own medical records and an institution’s legal obligation to provide those records.
In the short-term, the registry will collect three types of data: patient donated data (answers from a patient survey), electronic medical records abstracted from all relevant past providers, and genomic test results. Participating patients will have blood drawn at home by a phlebotomist for the genomic assay. Additional tubes of blood will be concurrently collected and biobanked. This will eventually allow for immune profiling, Dr. Labkoff said.
Future goals include a patient-reported outcomes module and the ability to link data with medical claims.
So far, 79 patients have participated in the pilot program, according to the MMRF. As the database builds, Ms. Quinn Young and Dr. Labkoff anticipate that it will yield answers to a variety of real-world questions.
Dr. Labkoff offered two examples. “Of all the patients who have been exposed to ‘name your drug,’ what were the costs of their therapy, and what were the outcomes?” he said. In addition, researchers will be able to query clinical trial inclusion criteria to search for data on a specific patient profile, such as patients with a 4:14 translocation, who have had a bone marrow transplant in the last 2 years, and have been exposed to a certain drug regimen.
Ms. Quinn Young noted that doctors may be able to use the database to reliably identify high-risk patients and guide agent selection. Common patient questions also will be addressed, she said, including best treatment regimens for certain types of patients.
“For patients who may have run out of all commercially available options, or for patients who are perhaps seen at a community center, where certainly this type of profiling is not standard, it’s opening up a whole new set of options for them,” Ms. Quinn Young said. “And if their physician doesn’t pursue those options, they have the report that they can use to seek a second opinion.”
The Direct-to-Patient registry is unique because it aims to empower patients in a way that hasn’t been done before, Ms. Quinn Young said. “We are committed ... ever since we conceived of this project, to giving results back to patients. That is disruptive because right now that doesn’t exist.”
But the cost of implementing the registry, which has an approximate budget of $20 million, stands in the way of a completely free flow of anonymized data. MMRF leaders are exploring different strategies to sustain funding for the program.
Another MMRF program, CoMMpass, uses a precompetitive consortium model, in which several pharmaceutical companies pay for a preview of the data 6 months in advance of nonprofit researchers. A similar model may be used with the Direct-to-Patient registry, but this has yet to be determined, according to Dr. Labkoff and Ms. Quinn Young.
For now, Ms. Quinn Young said she hopes that physicians will be receptive to the program. “[The short term goal is that] when patients come to their doctors asking about this, that there is support and open-mindedness,” she said.
Looking to the future, Dr. Labkoff described how the registry could accelerate myeloma research, ultimately toward a cure.
“It is generally accepted that it can take 17 years to get something – a therapy, a new drug, or a guideline – from the bench to the bedside,” he said. “It’s my hope that we can take next generation sequencing and the results of this registry and bend that curve, maybe ... to 10 [years], or very aggressively, to 7 or 5 [years], where doctors are able to use the information in these reports for the patients that have literally given themselves, and use this to help guide the choices of their therapy or the trials they apply for, to help them get a better outcome in general.”
The Direct-to-Patient registry is a collaborative effort between the MMRF and multiple organizations, including the health care technology company COTA, the Broad Institute of Harvard and MIT, Prometheus Research, Tempus, and the Dana Farber Cancer Institute.
Cancer pain management inadequate in opioid-saturated areas
Patients with cancer who live in regions with high levels of opioid misuse may be undertreated for pain, according to investigators who studied opioid prescription patterns and cancer incidence in rural southwest Virginia.
Among 4,324 patients with cancer, only 22.16% were prescribed a Controlled Schedule II (C-II) prescription opioid medication at least 3 times in 1 year, from prescribers likely to be treating cancer pain. More than 60% of patients never received a C-II opioid prescription, reported Virginia T. LeBaron, PhD, of the University of Virginia School of Nursing in Charlottesville, and colleagues.
“A clearer view of geographic patterns and predictors of both POM [prescription opioid medication] prescribing and potential harms can inform targeted interventions and policy initiatives that achieve a balanced approach to POMs – ensuring access for patients in need while reducing risk to both patients and communities. Our research makes an important contribution by exploring how the current ‘opioid epidemic’ relates to rural patients with cancer,” they wrote. Their report is in Journal of Oncology Practice.
The investigators studied the confluence of disproportionately high cancer mortality rates and opioid fatality rates in rural southwest Virginia, in the heart of Appalachia.
They conducted a longitudinal, exploratory secondary analysis of data from the Commonwealth of Virginia All Payer Claims database to look at opioid prescribing patterns and explore whether concerns about opioid misuse could result in undertreatment of pain in cancer patients.
They looked at prescribing patterns at the patient, provider, and insurance claim levels, predictors of opioid prescription frequency, opioid-related harms and patterns related to opioid prescribing, cancer incidence, and fatalities.
They identified 4,324 patients with cancer, 958 of whom (22.16%) received a C-II opioid at least three times in any study year. The majority of patients were in the 45-64 age range, and approximately 88% were diagnosed with solid malignancies, with breast cancer and lung cancer being the most frequent diagnoses.
As noted, more than 60% of patients never received a C-II prescription.
“The large percentages of cancer patients never prescribed a C-II are concerning for a number of reasons, especially when we consider the results per year,” the investigators wrote. “First, the ‘no C-II’ patients remain over 80% of the total sample, each year, even after accounting for the upscheduling (from C-III to C-II) of commonly-prescribed hydrocodone products in 2014. Second, anecdotal data and emerging empirical evidence demonstrate that patients with legitimate pain needs, including patients with cancer, experience significant difficulty accessing POMs.”
They noted that regulations regarding opioid prescriptions have become increasingly strict since the end date of their analysis in 2015, suggesting that the number of patients with cancer who are not receiving C-II opioids today may be even higher.
They also pointed to evidence of prescription practices suggesting suboptimal pain management or potential patient harm, such as frequent prescription of opioid-acetaminophen combinations that are dose-limited due to acetaminophen toxicity; coprescription of opioids and benzodiazepines, which is not recommended under current prescribing guidelines; and infrequent use of deterrent formulations of C-II opioids such as crush-resistant tablets.
The study was supported by the University of Virginia Cancer Center, Cancer Control & Population Health Division and the Virginia Tobacco Region Revitalization Commission. The authors reported having no disclaimers or conflicts of interest.
SOURCE: LeBaron VT et al. J Oncol Pract. 2019 Nov. 4. doi: 10.1200/JOP.19.00149.
Patients with cancer who live in regions with high levels of opioid misuse may be undertreated for pain, according to investigators who studied opioid prescription patterns and cancer incidence in rural southwest Virginia.
Among 4,324 patients with cancer, only 22.16% were prescribed a Controlled Schedule II (C-II) prescription opioid medication at least 3 times in 1 year, from prescribers likely to be treating cancer pain. More than 60% of patients never received a C-II opioid prescription, reported Virginia T. LeBaron, PhD, of the University of Virginia School of Nursing in Charlottesville, and colleagues.
“A clearer view of geographic patterns and predictors of both POM [prescription opioid medication] prescribing and potential harms can inform targeted interventions and policy initiatives that achieve a balanced approach to POMs – ensuring access for patients in need while reducing risk to both patients and communities. Our research makes an important contribution by exploring how the current ‘opioid epidemic’ relates to rural patients with cancer,” they wrote. Their report is in Journal of Oncology Practice.
The investigators studied the confluence of disproportionately high cancer mortality rates and opioid fatality rates in rural southwest Virginia, in the heart of Appalachia.
They conducted a longitudinal, exploratory secondary analysis of data from the Commonwealth of Virginia All Payer Claims database to look at opioid prescribing patterns and explore whether concerns about opioid misuse could result in undertreatment of pain in cancer patients.
They looked at prescribing patterns at the patient, provider, and insurance claim levels, predictors of opioid prescription frequency, opioid-related harms and patterns related to opioid prescribing, cancer incidence, and fatalities.
They identified 4,324 patients with cancer, 958 of whom (22.16%) received a C-II opioid at least three times in any study year. The majority of patients were in the 45-64 age range, and approximately 88% were diagnosed with solid malignancies, with breast cancer and lung cancer being the most frequent diagnoses.
As noted, more than 60% of patients never received a C-II prescription.
“The large percentages of cancer patients never prescribed a C-II are concerning for a number of reasons, especially when we consider the results per year,” the investigators wrote. “First, the ‘no C-II’ patients remain over 80% of the total sample, each year, even after accounting for the upscheduling (from C-III to C-II) of commonly-prescribed hydrocodone products in 2014. Second, anecdotal data and emerging empirical evidence demonstrate that patients with legitimate pain needs, including patients with cancer, experience significant difficulty accessing POMs.”
They noted that regulations regarding opioid prescriptions have become increasingly strict since the end date of their analysis in 2015, suggesting that the number of patients with cancer who are not receiving C-II opioids today may be even higher.
They also pointed to evidence of prescription practices suggesting suboptimal pain management or potential patient harm, such as frequent prescription of opioid-acetaminophen combinations that are dose-limited due to acetaminophen toxicity; coprescription of opioids and benzodiazepines, which is not recommended under current prescribing guidelines; and infrequent use of deterrent formulations of C-II opioids such as crush-resistant tablets.
The study was supported by the University of Virginia Cancer Center, Cancer Control & Population Health Division and the Virginia Tobacco Region Revitalization Commission. The authors reported having no disclaimers or conflicts of interest.
SOURCE: LeBaron VT et al. J Oncol Pract. 2019 Nov. 4. doi: 10.1200/JOP.19.00149.
Patients with cancer who live in regions with high levels of opioid misuse may be undertreated for pain, according to investigators who studied opioid prescription patterns and cancer incidence in rural southwest Virginia.
Among 4,324 patients with cancer, only 22.16% were prescribed a Controlled Schedule II (C-II) prescription opioid medication at least 3 times in 1 year, from prescribers likely to be treating cancer pain. More than 60% of patients never received a C-II opioid prescription, reported Virginia T. LeBaron, PhD, of the University of Virginia School of Nursing in Charlottesville, and colleagues.
“A clearer view of geographic patterns and predictors of both POM [prescription opioid medication] prescribing and potential harms can inform targeted interventions and policy initiatives that achieve a balanced approach to POMs – ensuring access for patients in need while reducing risk to both patients and communities. Our research makes an important contribution by exploring how the current ‘opioid epidemic’ relates to rural patients with cancer,” they wrote. Their report is in Journal of Oncology Practice.
The investigators studied the confluence of disproportionately high cancer mortality rates and opioid fatality rates in rural southwest Virginia, in the heart of Appalachia.
They conducted a longitudinal, exploratory secondary analysis of data from the Commonwealth of Virginia All Payer Claims database to look at opioid prescribing patterns and explore whether concerns about opioid misuse could result in undertreatment of pain in cancer patients.
They looked at prescribing patterns at the patient, provider, and insurance claim levels, predictors of opioid prescription frequency, opioid-related harms and patterns related to opioid prescribing, cancer incidence, and fatalities.
They identified 4,324 patients with cancer, 958 of whom (22.16%) received a C-II opioid at least three times in any study year. The majority of patients were in the 45-64 age range, and approximately 88% were diagnosed with solid malignancies, with breast cancer and lung cancer being the most frequent diagnoses.
As noted, more than 60% of patients never received a C-II prescription.
“The large percentages of cancer patients never prescribed a C-II are concerning for a number of reasons, especially when we consider the results per year,” the investigators wrote. “First, the ‘no C-II’ patients remain over 80% of the total sample, each year, even after accounting for the upscheduling (from C-III to C-II) of commonly-prescribed hydrocodone products in 2014. Second, anecdotal data and emerging empirical evidence demonstrate that patients with legitimate pain needs, including patients with cancer, experience significant difficulty accessing POMs.”
They noted that regulations regarding opioid prescriptions have become increasingly strict since the end date of their analysis in 2015, suggesting that the number of patients with cancer who are not receiving C-II opioids today may be even higher.
They also pointed to evidence of prescription practices suggesting suboptimal pain management or potential patient harm, such as frequent prescription of opioid-acetaminophen combinations that are dose-limited due to acetaminophen toxicity; coprescription of opioids and benzodiazepines, which is not recommended under current prescribing guidelines; and infrequent use of deterrent formulations of C-II opioids such as crush-resistant tablets.
The study was supported by the University of Virginia Cancer Center, Cancer Control & Population Health Division and the Virginia Tobacco Region Revitalization Commission. The authors reported having no disclaimers or conflicts of interest.
SOURCE: LeBaron VT et al. J Oncol Pract. 2019 Nov. 4. doi: 10.1200/JOP.19.00149.
FROM JOURNAL OF ONCOLOGY PRACTICE
Exercise improved QoL, functioning in breast cancer survivors
BARCELONA – A supervised and adapted exercise program improved quality of life, physical functioning, and strength in breast cancer survivors participating in the MAMA MOVE Gaia study.
Of 19 women who initiated participation in the program, which included a 16-week control phase followed by a 16-week exercise training intervention phase, 15 completed the program, and, after the training intervention, they experienced a significant increase in handgrip strength and sit-to-stand repetitions, Ana Joaquim, MD, of Centro Hospitalar de Vila Nova de Gaia/Espinho, Portugal, and colleagues reported in a poster at the European Society for Medical Oncology Congress.
During the control phase of the prospective nonrandomized study, participants experienced no significant changes over time in any domain of quality of life as measured by the EORTC QLQ-C30 questionnaire, although a trend toward improved physical functioning was noted at an evaluation performed 8 weeks after the control phase, compared with one performed just prior to the intervention phase (77.3 to 85.3 points, P = .051), the investigators said.
After the intervention phase, however, handgrip strength improved significantly at both the limb where surgery was performed and at the nonoperated limb (from 22.2 to 25.6 kg.f and from 22.6 to 26.9 kg.f). Similar results were observed for a sit-to-stand test (improvement from 12 to 17 repetitions).
Participants in the single-arm clinical trial were assessed after 8 weeks of the control phase, immediately prior to the intervention period, 8 weeks after the control phase, and 16 weeks into the invention phase.
The intervention phase consisted of 3 60-minute sessions per week of combined moderate to vigorous aerobic and strength exercise, defined as exercise at 65%-85% of maximum heart rate or at 6-8 points on the OMNI scale. Mean compliance among the participants was 63.6%.
The participants had a median age of 59 and 15 of the 19 were diagnosed with invasive carcinoma. Following surgery, 13 underwent radiotherapy, 15 received chemotherapy, and 18 received hormone therapy.
“Treatments for early breast cancer have side effects that affect quality of life and cause deconditioning,” the investigators wrote, adding that “physical exercise might have a supportive and coadjuvant role in the rehabilitation of breast cancer survivors.”
The MAMA MOVE trial aimed to assess the potential benefits of a community-based supervised exercise training program, and the findings suggest such programs could help improve quality of life, particularly with respect to physical functioning, they concluded.
The MAMA MOVE Gaia study was funded by Liga Portuguesa Contra o Cancro. The investigators reported having no disclosures.
SOURCE: Joaquim A et al. ESMO 2019, Abstract 234P.
BARCELONA – A supervised and adapted exercise program improved quality of life, physical functioning, and strength in breast cancer survivors participating in the MAMA MOVE Gaia study.
Of 19 women who initiated participation in the program, which included a 16-week control phase followed by a 16-week exercise training intervention phase, 15 completed the program, and, after the training intervention, they experienced a significant increase in handgrip strength and sit-to-stand repetitions, Ana Joaquim, MD, of Centro Hospitalar de Vila Nova de Gaia/Espinho, Portugal, and colleagues reported in a poster at the European Society for Medical Oncology Congress.
During the control phase of the prospective nonrandomized study, participants experienced no significant changes over time in any domain of quality of life as measured by the EORTC QLQ-C30 questionnaire, although a trend toward improved physical functioning was noted at an evaluation performed 8 weeks after the control phase, compared with one performed just prior to the intervention phase (77.3 to 85.3 points, P = .051), the investigators said.
After the intervention phase, however, handgrip strength improved significantly at both the limb where surgery was performed and at the nonoperated limb (from 22.2 to 25.6 kg.f and from 22.6 to 26.9 kg.f). Similar results were observed for a sit-to-stand test (improvement from 12 to 17 repetitions).
Participants in the single-arm clinical trial were assessed after 8 weeks of the control phase, immediately prior to the intervention period, 8 weeks after the control phase, and 16 weeks into the invention phase.
The intervention phase consisted of 3 60-minute sessions per week of combined moderate to vigorous aerobic and strength exercise, defined as exercise at 65%-85% of maximum heart rate or at 6-8 points on the OMNI scale. Mean compliance among the participants was 63.6%.
The participants had a median age of 59 and 15 of the 19 were diagnosed with invasive carcinoma. Following surgery, 13 underwent radiotherapy, 15 received chemotherapy, and 18 received hormone therapy.
“Treatments for early breast cancer have side effects that affect quality of life and cause deconditioning,” the investigators wrote, adding that “physical exercise might have a supportive and coadjuvant role in the rehabilitation of breast cancer survivors.”
The MAMA MOVE trial aimed to assess the potential benefits of a community-based supervised exercise training program, and the findings suggest such programs could help improve quality of life, particularly with respect to physical functioning, they concluded.
The MAMA MOVE Gaia study was funded by Liga Portuguesa Contra o Cancro. The investigators reported having no disclosures.
SOURCE: Joaquim A et al. ESMO 2019, Abstract 234P.
BARCELONA – A supervised and adapted exercise program improved quality of life, physical functioning, and strength in breast cancer survivors participating in the MAMA MOVE Gaia study.
Of 19 women who initiated participation in the program, which included a 16-week control phase followed by a 16-week exercise training intervention phase, 15 completed the program, and, after the training intervention, they experienced a significant increase in handgrip strength and sit-to-stand repetitions, Ana Joaquim, MD, of Centro Hospitalar de Vila Nova de Gaia/Espinho, Portugal, and colleagues reported in a poster at the European Society for Medical Oncology Congress.
During the control phase of the prospective nonrandomized study, participants experienced no significant changes over time in any domain of quality of life as measured by the EORTC QLQ-C30 questionnaire, although a trend toward improved physical functioning was noted at an evaluation performed 8 weeks after the control phase, compared with one performed just prior to the intervention phase (77.3 to 85.3 points, P = .051), the investigators said.
After the intervention phase, however, handgrip strength improved significantly at both the limb where surgery was performed and at the nonoperated limb (from 22.2 to 25.6 kg.f and from 22.6 to 26.9 kg.f). Similar results were observed for a sit-to-stand test (improvement from 12 to 17 repetitions).
Participants in the single-arm clinical trial were assessed after 8 weeks of the control phase, immediately prior to the intervention period, 8 weeks after the control phase, and 16 weeks into the invention phase.
The intervention phase consisted of 3 60-minute sessions per week of combined moderate to vigorous aerobic and strength exercise, defined as exercise at 65%-85% of maximum heart rate or at 6-8 points on the OMNI scale. Mean compliance among the participants was 63.6%.
The participants had a median age of 59 and 15 of the 19 were diagnosed with invasive carcinoma. Following surgery, 13 underwent radiotherapy, 15 received chemotherapy, and 18 received hormone therapy.
“Treatments for early breast cancer have side effects that affect quality of life and cause deconditioning,” the investigators wrote, adding that “physical exercise might have a supportive and coadjuvant role in the rehabilitation of breast cancer survivors.”
The MAMA MOVE trial aimed to assess the potential benefits of a community-based supervised exercise training program, and the findings suggest such programs could help improve quality of life, particularly with respect to physical functioning, they concluded.
The MAMA MOVE Gaia study was funded by Liga Portuguesa Contra o Cancro. The investigators reported having no disclosures.
SOURCE: Joaquim A et al. ESMO 2019, Abstract 234P.
REPORTING FROM ESMO 2019
Case-control study IDs several novel risk factors of post-HCT melanoma
(HCT), according to findings from a nested case-control study.
The study included 140 cases of melanoma and 557 controls matched by age at HCT, sex, primary disease, and survival time. The results showed a significantly increased melanoma risk in HCT survivors who received total body irradiation–based myeloablative conditioning, reduced-intensity conditioning with melphalan, or reduced-intensity conditioning with fludarabine, compared with those who received busulfan-based myeloablative conditioning (odds ratios, 1.77, 2.60, and 2.72, respectively), Megan M. Herr, PhD, of the division of cancer epidemiology and genetics at the National Cancer Institute, and the Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and colleagues reported in the Journal of the American Academy of Dermatology.
Melanoma risk also was increased in patients who experienced acute graft-versus-host disease (GVHD) with stage 2 or greater skin involvement (OR, 1.92 vs. those with no acute GVHD), chronic GVHD without skin involvement (OR, 1.91 vs. those with no chronic GVHD), or keratinocytic carcinoma (OR, 2.37), and in those who resided in areas with higher ambient ultraviolet radiation (OR for the highest vs. lowest tertile, 1.64).
The UV radiation finding was more pronounced for melanomas occurring 6 or more years after transplant (OR, 3.04 for highest vs. lowest tertile), whereas ambient UV radiation was not associated with melanomas occurring earlier (ORs, 1.37 for less than 3 years and 0.98 at 3-6 years), the investigators noted.
The findings, based on large-scale and detailed clinical data from the Center for International Blood and Marrow Transplant Research for HCT performed during 1985-2012, show that melanoma after HCT has a multifactorial etiology that includes patient-, transplant-, and posttransplant-related factors, they said, noting that the findings also underscore the importance of “prioritization of high-risk survivors for adherence to prevention and screening recommendations.”
Those recommendations call for routine skin examination and photoprotective precautions – particularly in HCT survivors at the highest risk – but studies of screening behaviors suggest that fewer than two-thirds of HCT survivors adhere to these recommendations, they said, concluding that further research on the cost-effectiveness of melanoma screening is warranted, as is investigation into whether current approaches are associated with melanoma risk.
This work was supported by the intramural research program of the National Cancer Institute, the National Institutes of Health, and the Department of Health & Human Services. The authors reported having no conflicts of interest.
SOURCE: Herr MM et al. J Am Acad Dermatol. 2019 Oct 22. doi: 10.1016/j.jaad.2019.10.034.
(HCT), according to findings from a nested case-control study.
The study included 140 cases of melanoma and 557 controls matched by age at HCT, sex, primary disease, and survival time. The results showed a significantly increased melanoma risk in HCT survivors who received total body irradiation–based myeloablative conditioning, reduced-intensity conditioning with melphalan, or reduced-intensity conditioning with fludarabine, compared with those who received busulfan-based myeloablative conditioning (odds ratios, 1.77, 2.60, and 2.72, respectively), Megan M. Herr, PhD, of the division of cancer epidemiology and genetics at the National Cancer Institute, and the Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and colleagues reported in the Journal of the American Academy of Dermatology.
Melanoma risk also was increased in patients who experienced acute graft-versus-host disease (GVHD) with stage 2 or greater skin involvement (OR, 1.92 vs. those with no acute GVHD), chronic GVHD without skin involvement (OR, 1.91 vs. those with no chronic GVHD), or keratinocytic carcinoma (OR, 2.37), and in those who resided in areas with higher ambient ultraviolet radiation (OR for the highest vs. lowest tertile, 1.64).
The UV radiation finding was more pronounced for melanomas occurring 6 or more years after transplant (OR, 3.04 for highest vs. lowest tertile), whereas ambient UV radiation was not associated with melanomas occurring earlier (ORs, 1.37 for less than 3 years and 0.98 at 3-6 years), the investigators noted.
The findings, based on large-scale and detailed clinical data from the Center for International Blood and Marrow Transplant Research for HCT performed during 1985-2012, show that melanoma after HCT has a multifactorial etiology that includes patient-, transplant-, and posttransplant-related factors, they said, noting that the findings also underscore the importance of “prioritization of high-risk survivors for adherence to prevention and screening recommendations.”
Those recommendations call for routine skin examination and photoprotective precautions – particularly in HCT survivors at the highest risk – but studies of screening behaviors suggest that fewer than two-thirds of HCT survivors adhere to these recommendations, they said, concluding that further research on the cost-effectiveness of melanoma screening is warranted, as is investigation into whether current approaches are associated with melanoma risk.
This work was supported by the intramural research program of the National Cancer Institute, the National Institutes of Health, and the Department of Health & Human Services. The authors reported having no conflicts of interest.
SOURCE: Herr MM et al. J Am Acad Dermatol. 2019 Oct 22. doi: 10.1016/j.jaad.2019.10.034.
(HCT), according to findings from a nested case-control study.
The study included 140 cases of melanoma and 557 controls matched by age at HCT, sex, primary disease, and survival time. The results showed a significantly increased melanoma risk in HCT survivors who received total body irradiation–based myeloablative conditioning, reduced-intensity conditioning with melphalan, or reduced-intensity conditioning with fludarabine, compared with those who received busulfan-based myeloablative conditioning (odds ratios, 1.77, 2.60, and 2.72, respectively), Megan M. Herr, PhD, of the division of cancer epidemiology and genetics at the National Cancer Institute, and the Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and colleagues reported in the Journal of the American Academy of Dermatology.
Melanoma risk also was increased in patients who experienced acute graft-versus-host disease (GVHD) with stage 2 or greater skin involvement (OR, 1.92 vs. those with no acute GVHD), chronic GVHD without skin involvement (OR, 1.91 vs. those with no chronic GVHD), or keratinocytic carcinoma (OR, 2.37), and in those who resided in areas with higher ambient ultraviolet radiation (OR for the highest vs. lowest tertile, 1.64).
The UV radiation finding was more pronounced for melanomas occurring 6 or more years after transplant (OR, 3.04 for highest vs. lowest tertile), whereas ambient UV radiation was not associated with melanomas occurring earlier (ORs, 1.37 for less than 3 years and 0.98 at 3-6 years), the investigators noted.
The findings, based on large-scale and detailed clinical data from the Center for International Blood and Marrow Transplant Research for HCT performed during 1985-2012, show that melanoma after HCT has a multifactorial etiology that includes patient-, transplant-, and posttransplant-related factors, they said, noting that the findings also underscore the importance of “prioritization of high-risk survivors for adherence to prevention and screening recommendations.”
Those recommendations call for routine skin examination and photoprotective precautions – particularly in HCT survivors at the highest risk – but studies of screening behaviors suggest that fewer than two-thirds of HCT survivors adhere to these recommendations, they said, concluding that further research on the cost-effectiveness of melanoma screening is warranted, as is investigation into whether current approaches are associated with melanoma risk.
This work was supported by the intramural research program of the National Cancer Institute, the National Institutes of Health, and the Department of Health & Human Services. The authors reported having no conflicts of interest.
SOURCE: Herr MM et al. J Am Acad Dermatol. 2019 Oct 22. doi: 10.1016/j.jaad.2019.10.034.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
FDA approves Ziextenzo for neutropenia-related infection reduction
The Food and Drug Administration has approved the biosimilar Ziextenzo (pegfilgrastim-bmez) to reduce the incidence of infection in patients with nonmyeloid cancer receiving suppressive anticancer drugs that are associated with febrile neutropenia.
More than 60,000 cancer patients are hospitalized in the United States each year with evidence of neutropenia, resulting in more than 4,000 deaths, according to Ziextenzo maker Sandoz, a Novartis division.
The FDA approval was based on analytical, preclinical, and clinical research, including data from a three-way pharmacokinetics and pharmacodynamics study that compared pegfilgrastim-bmez with the reference drug pegfilgrastim (Neulasta) from the United States and the European Union. Pharmacokinetic and pharmacodynamic similarity were shown between pegfilgrastim-bmez with the reference drugs, and there were no clinically significant differences in safety or immunogenicity.
The most common adverse events associated with pegfilgrastim-bmez are bone pain and pain in the extremities, according to the label.
The Food and Drug Administration has approved the biosimilar Ziextenzo (pegfilgrastim-bmez) to reduce the incidence of infection in patients with nonmyeloid cancer receiving suppressive anticancer drugs that are associated with febrile neutropenia.
More than 60,000 cancer patients are hospitalized in the United States each year with evidence of neutropenia, resulting in more than 4,000 deaths, according to Ziextenzo maker Sandoz, a Novartis division.
The FDA approval was based on analytical, preclinical, and clinical research, including data from a three-way pharmacokinetics and pharmacodynamics study that compared pegfilgrastim-bmez with the reference drug pegfilgrastim (Neulasta) from the United States and the European Union. Pharmacokinetic and pharmacodynamic similarity were shown between pegfilgrastim-bmez with the reference drugs, and there were no clinically significant differences in safety or immunogenicity.
The most common adverse events associated with pegfilgrastim-bmez are bone pain and pain in the extremities, according to the label.
The Food and Drug Administration has approved the biosimilar Ziextenzo (pegfilgrastim-bmez) to reduce the incidence of infection in patients with nonmyeloid cancer receiving suppressive anticancer drugs that are associated with febrile neutropenia.
More than 60,000 cancer patients are hospitalized in the United States each year with evidence of neutropenia, resulting in more than 4,000 deaths, according to Ziextenzo maker Sandoz, a Novartis division.
The FDA approval was based on analytical, preclinical, and clinical research, including data from a three-way pharmacokinetics and pharmacodynamics study that compared pegfilgrastim-bmez with the reference drug pegfilgrastim (Neulasta) from the United States and the European Union. Pharmacokinetic and pharmacodynamic similarity were shown between pegfilgrastim-bmez with the reference drugs, and there were no clinically significant differences in safety or immunogenicity.
The most common adverse events associated with pegfilgrastim-bmez are bone pain and pain in the extremities, according to the label.
First NCCN guideline on hematopoietic cell transplantation focuses on GVHD
Recommendations for the diagnosis and management of acute and chronic graft-versus-host disease (GVHD) are the central focus of the first National Comprehensive Cancer Network (NCCN) guideline on hematopoietic cell transplantation.
The guideline presents detailed recommendations for the evaluation of hematopoietic cell transplant (HCT) recipients, and an extensive section on the diagnosis and workup of GVHD, including information on staging and grading of acute GVHD, grading of chronic GVHD, treatment response criteria, and suggested systemic therapies for steroid-refractory disease.
“We wanted to both build on the commonly used approach to stage and treat graft-versus-host disease, and make sure that this information is readily available for physicians-in-training and young physicians who are learning about transplants,” said guideline committee chair Ayman Saad, MB BCh, of The Ohio State University Comprehensive Cancer Center, James Cancer Hospital and Solove Research Institute in Columbus, Ohio.
In an interview, Dr. Saad emphasized that an important goal of the guidelines is to encourage general oncologists to recognize early signs of GVHD and refer potential candidates to transplant centers for further evaluation.
“We also urge oncologists who may be caring for patients after HCT to familiarize themselves with the varied manifestations of GVHD – a very common and significant posttransplant complication – and to consult with transplant providers to optimize their ongoing care. The guidelines explain how to diagnose and treat this condition in order to achieve the best possible outcomes,” guideline panel member Alison W. Loren, MD, director of blood and marrow transplantation at Abraham Cancer Center, University of Pennsylvania, Philadelphia, said in a statement.
The guideline includes links to other NCCN guidelines for diseases where HCT is a common therapeutic option, including leukemias, myeloid malignancies, lymphomas, central nervous system cancers, and testicular cancer.
The HCT guideline includes:
- Pretransplant recipient evaluation with recommendations for clinical assessment and imaging.
- Diagnosis and workup of GVHD, with separate algorithms for suspected acute or chronic GVHD.
- Specific interventions for management of acute GVHD with corticosteroids or other systemic agents.
- Chronic GVHD diagnosis by organ site and symptoms, with a severity scoring system.
- Chronic GVHD steroid response definitions and criteria.
- Suggested systemic agents for steroid-refractory GVHD.
One feature that is unusual for an NCCN guideline document is a page of photographs to assist clinicians in diagnosing range-of-motion abnormalities in the shoulder, elbow, hand, and ankle of patients with suspected or confirmed GVHD. Dr. Saad said that future iterations of the guideline will include additional photos to help clinicians develop a visual repertoire of potential GVHD signs.
Future versions will also include a discussion section and more comprehensive information on other common complications following HCT transplant, as well as management of posttransplant relapse.
Ideally, the guideline will help clinicians document and justify clinical decisions surrounding HCT and GVHD management in discussions with third-party payers, Dr. Saad said.
“Sometimes we struggle with payers when we want to use a certain modality to treat GVHD, and they respond ‘that’s not approved,’ or ‘that’s not a common indication,’ et cetera,” he said. “What we’re trying to put here are the commonly used therapies that most, but not all, experts agree on, and we can use this to negotiate with payers.”
He also emphasized that the guideline is meant to be instructive rather than prescriptive and is not meant to hinder innovations that may emerge from clinical trials.
“We would appreciate any feedback from non-NCCN centers as well as experts at NCCN centers, and we’ll be more than happy to address any concerns or criticisms they have,” he said.
Dr. Saad reported financial relationships with Actinium and Incysus Biomedical. Dr. Loren has previously reported having no disclosures.
Recommendations for the diagnosis and management of acute and chronic graft-versus-host disease (GVHD) are the central focus of the first National Comprehensive Cancer Network (NCCN) guideline on hematopoietic cell transplantation.
The guideline presents detailed recommendations for the evaluation of hematopoietic cell transplant (HCT) recipients, and an extensive section on the diagnosis and workup of GVHD, including information on staging and grading of acute GVHD, grading of chronic GVHD, treatment response criteria, and suggested systemic therapies for steroid-refractory disease.
“We wanted to both build on the commonly used approach to stage and treat graft-versus-host disease, and make sure that this information is readily available for physicians-in-training and young physicians who are learning about transplants,” said guideline committee chair Ayman Saad, MB BCh, of The Ohio State University Comprehensive Cancer Center, James Cancer Hospital and Solove Research Institute in Columbus, Ohio.
In an interview, Dr. Saad emphasized that an important goal of the guidelines is to encourage general oncologists to recognize early signs of GVHD and refer potential candidates to transplant centers for further evaluation.
“We also urge oncologists who may be caring for patients after HCT to familiarize themselves with the varied manifestations of GVHD – a very common and significant posttransplant complication – and to consult with transplant providers to optimize their ongoing care. The guidelines explain how to diagnose and treat this condition in order to achieve the best possible outcomes,” guideline panel member Alison W. Loren, MD, director of blood and marrow transplantation at Abraham Cancer Center, University of Pennsylvania, Philadelphia, said in a statement.
The guideline includes links to other NCCN guidelines for diseases where HCT is a common therapeutic option, including leukemias, myeloid malignancies, lymphomas, central nervous system cancers, and testicular cancer.
The HCT guideline includes:
- Pretransplant recipient evaluation with recommendations for clinical assessment and imaging.
- Diagnosis and workup of GVHD, with separate algorithms for suspected acute or chronic GVHD.
- Specific interventions for management of acute GVHD with corticosteroids or other systemic agents.
- Chronic GVHD diagnosis by organ site and symptoms, with a severity scoring system.
- Chronic GVHD steroid response definitions and criteria.
- Suggested systemic agents for steroid-refractory GVHD.
One feature that is unusual for an NCCN guideline document is a page of photographs to assist clinicians in diagnosing range-of-motion abnormalities in the shoulder, elbow, hand, and ankle of patients with suspected or confirmed GVHD. Dr. Saad said that future iterations of the guideline will include additional photos to help clinicians develop a visual repertoire of potential GVHD signs.
Future versions will also include a discussion section and more comprehensive information on other common complications following HCT transplant, as well as management of posttransplant relapse.
Ideally, the guideline will help clinicians document and justify clinical decisions surrounding HCT and GVHD management in discussions with third-party payers, Dr. Saad said.
“Sometimes we struggle with payers when we want to use a certain modality to treat GVHD, and they respond ‘that’s not approved,’ or ‘that’s not a common indication,’ et cetera,” he said. “What we’re trying to put here are the commonly used therapies that most, but not all, experts agree on, and we can use this to negotiate with payers.”
He also emphasized that the guideline is meant to be instructive rather than prescriptive and is not meant to hinder innovations that may emerge from clinical trials.
“We would appreciate any feedback from non-NCCN centers as well as experts at NCCN centers, and we’ll be more than happy to address any concerns or criticisms they have,” he said.
Dr. Saad reported financial relationships with Actinium and Incysus Biomedical. Dr. Loren has previously reported having no disclosures.
Recommendations for the diagnosis and management of acute and chronic graft-versus-host disease (GVHD) are the central focus of the first National Comprehensive Cancer Network (NCCN) guideline on hematopoietic cell transplantation.
The guideline presents detailed recommendations for the evaluation of hematopoietic cell transplant (HCT) recipients, and an extensive section on the diagnosis and workup of GVHD, including information on staging and grading of acute GVHD, grading of chronic GVHD, treatment response criteria, and suggested systemic therapies for steroid-refractory disease.
“We wanted to both build on the commonly used approach to stage and treat graft-versus-host disease, and make sure that this information is readily available for physicians-in-training and young physicians who are learning about transplants,” said guideline committee chair Ayman Saad, MB BCh, of The Ohio State University Comprehensive Cancer Center, James Cancer Hospital and Solove Research Institute in Columbus, Ohio.
In an interview, Dr. Saad emphasized that an important goal of the guidelines is to encourage general oncologists to recognize early signs of GVHD and refer potential candidates to transplant centers for further evaluation.
“We also urge oncologists who may be caring for patients after HCT to familiarize themselves with the varied manifestations of GVHD – a very common and significant posttransplant complication – and to consult with transplant providers to optimize their ongoing care. The guidelines explain how to diagnose and treat this condition in order to achieve the best possible outcomes,” guideline panel member Alison W. Loren, MD, director of blood and marrow transplantation at Abraham Cancer Center, University of Pennsylvania, Philadelphia, said in a statement.
The guideline includes links to other NCCN guidelines for diseases where HCT is a common therapeutic option, including leukemias, myeloid malignancies, lymphomas, central nervous system cancers, and testicular cancer.
The HCT guideline includes:
- Pretransplant recipient evaluation with recommendations for clinical assessment and imaging.
- Diagnosis and workup of GVHD, with separate algorithms for suspected acute or chronic GVHD.
- Specific interventions for management of acute GVHD with corticosteroids or other systemic agents.
- Chronic GVHD diagnosis by organ site and symptoms, with a severity scoring system.
- Chronic GVHD steroid response definitions and criteria.
- Suggested systemic agents for steroid-refractory GVHD.
One feature that is unusual for an NCCN guideline document is a page of photographs to assist clinicians in diagnosing range-of-motion abnormalities in the shoulder, elbow, hand, and ankle of patients with suspected or confirmed GVHD. Dr. Saad said that future iterations of the guideline will include additional photos to help clinicians develop a visual repertoire of potential GVHD signs.
Future versions will also include a discussion section and more comprehensive information on other common complications following HCT transplant, as well as management of posttransplant relapse.
Ideally, the guideline will help clinicians document and justify clinical decisions surrounding HCT and GVHD management in discussions with third-party payers, Dr. Saad said.
“Sometimes we struggle with payers when we want to use a certain modality to treat GVHD, and they respond ‘that’s not approved,’ or ‘that’s not a common indication,’ et cetera,” he said. “What we’re trying to put here are the commonly used therapies that most, but not all, experts agree on, and we can use this to negotiate with payers.”
He also emphasized that the guideline is meant to be instructive rather than prescriptive and is not meant to hinder innovations that may emerge from clinical trials.
“We would appreciate any feedback from non-NCCN centers as well as experts at NCCN centers, and we’ll be more than happy to address any concerns or criticisms they have,” he said.
Dr. Saad reported financial relationships with Actinium and Incysus Biomedical. Dr. Loren has previously reported having no disclosures.
Levothyroxine dose for checkpoint inhibitor toxicity may be too high
CHICAGO – both for patients with preexisting and de novo hypothyroidism.
The real-world data, presented by Megan Kristan, MD, at the annual meeting of the American Thyroid Association, refine recommendations for dosing by body weight for levothyroxine in patients receiving checkpoint inhibitor therapy.
Immune checkpoint inhibitors stand a good chance of turning the tide against melanoma, some lung cancers, and other malignancies that have long been considered lethal. However, as more patients are exposed to the therapies, endocrinologists are seeing a wave of thyroid abnormalities, and must decide when, and at what doses, to treat hypothyroidism, said Dr. Kristan, a diabetes, endocrinology, and nutrition fellow at the University of Maryland, Baltimore.
Six checkpoint inhibitors are currently approved to hit a variety of molecular targets, and the prevalence of thyroid toxicity and hypothyroidism across the drug class ranges from a reported 9% to 40%, said Dr. Kristan.
The acknowledged thyroid toxicity of these drugs led the American Society for Clinical Oncology (ASCO) to issue guidelines advising that oncologists obtain baseline thyroid function tests before initiating checkpoint inhibitors, and that values be rechecked frequently – every 4-6 weeks – during therapy.
The guidelines advise dosing levothyroxine at approximately 1.6 mcg/kg per day, based on ideal patient body weight. The recommendation is limited to patients without risk factors, and approximates full levothyroxine replacement.
However, some patients enter cancer treatment with hypothyroidism, and some develop it de novo after beginning checkpoint inhibitor therapy. It is not known how best to treat each group, said Dr. Kristan.
To help answer that question, she and her collaborators at Georgetown University Hospital, McLean, Va., made use of a database drawn from five hospitals to perform a retrospective chart review. They looked at 822 patients who had received checkpoint inhibitor therapy, and from those patients, they selected 118 who had a diagnosis of hypothyroidism, or who received a prescription for levothyroxine during the 8-year study period.
The investigators assembled all available relevant data for each patient, including thyroid function tests, levothyroxine dosing, type of cancer, and type of therapy. They sorted participants into those who had received a diagnosis of hypothyroidism before or after receiving the first dose of checkpoint inhibitor therapy.
At baseline, 81 patients had preexisting hypothyroidism and were receiving a mean levothyroxine dose of 88.2 mcg. After treatment, the mean dose was 94.3 mcg, a nonsignificant difference. The median dose for this group remained at 88 mcg through treatment.
For the 37 patients who developed hypothyroidism de novo during checkpoint inhibitor therapy, the final observed levothyroxine dose was 71.2 mcg.
The mean age of the patients at baseline was 69 years. About half were women, and 91% were white. Either nivolumab or pembrolizumab was used in 72% of patients, making them the most commonly used checkpoint inhibitors, though 90% of patients received combination therapy. Taken together, melanoma and lung cancer accounted for about two-thirds of the cancers seen.
For both groups, the on-treatment levothyroxine dose was considerably lower than the ASCO-recommended, weight-based dosing, which would have been 122.9 mcg for those with preexisting hypothyroidism and 115.7 mcg for those who developed hypothyroidism on treatment (P less than .001 for both).
Dr. Kristan noted that thyroid stimulating hormone (TSH) values for patients with pretreatment hypothyroidism peaked between weeks 12 and 20, though there was no preemptive adjustment of levothyroxine dosing.
For those who developed on-treatment hypothyroidism, TSH values peaked at a series of times, at about weeks 8, 16, and 32. These waves of TSH elevation, she said, support the 4- to 6-week follow-up interval recommended in the ASCO guidelines.
However, she said, patients with de novo hypothyroidism “should not be started on the 1.6-mcg/kg-a-day weight-based dosing.” The cohort with de novo hypothyroidism in Dr. Kristan’s analysis required a daily dose of about 1 mcg/kg, she said. These real-world results support the idea that many patients on checkpoint inhibitors retain some thyroid reserve.
Dr. Kristan said that based on these findings, she and her collaborators recommend monitoring thyroid function every 4-6 weeks for patients taking immune checkpoint inhibitors. Patients with preexisting thyroid disease should not have an empiric adjustment of levothyroxine dose on checkpoint inhibitor initiation. For patients who develop thyroiditis after starting therapy, initiating a dose at 1 mcg/kg per day of ideal body weight is a good place to start, and treatment response should be monitored.
The study was limited by its retrospective nature and the small sample size, acknowledged Dr. Kristan. In addition, there were confounding variables and different frequencies of testing across institutions, and antibody status was not available and may have affected the results. Testing was performable for all participants.
Dr. Kristan said that the analysis opens up areas for further study, such as which patient populations are at risk for developing thyroid toxicity, what baseline characteristics can help predict which patients develop toxicity, and whether particular checkpoint inhibitors are more likely to cause toxicity. In addition, she said, a subset of patients will develop hyperthyroidism on checkpoint inhibitor therapy, and little is known about how to treat that complication.
Dr. Kristan reported no conflicts of interest. The research she presented was completed during her residency at Georgetown University.
SOURCE: Kristan M et al. ATA 2019, Oral Abstract 25.
CHICAGO – both for patients with preexisting and de novo hypothyroidism.
The real-world data, presented by Megan Kristan, MD, at the annual meeting of the American Thyroid Association, refine recommendations for dosing by body weight for levothyroxine in patients receiving checkpoint inhibitor therapy.
Immune checkpoint inhibitors stand a good chance of turning the tide against melanoma, some lung cancers, and other malignancies that have long been considered lethal. However, as more patients are exposed to the therapies, endocrinologists are seeing a wave of thyroid abnormalities, and must decide when, and at what doses, to treat hypothyroidism, said Dr. Kristan, a diabetes, endocrinology, and nutrition fellow at the University of Maryland, Baltimore.
Six checkpoint inhibitors are currently approved to hit a variety of molecular targets, and the prevalence of thyroid toxicity and hypothyroidism across the drug class ranges from a reported 9% to 40%, said Dr. Kristan.
The acknowledged thyroid toxicity of these drugs led the American Society for Clinical Oncology (ASCO) to issue guidelines advising that oncologists obtain baseline thyroid function tests before initiating checkpoint inhibitors, and that values be rechecked frequently – every 4-6 weeks – during therapy.
The guidelines advise dosing levothyroxine at approximately 1.6 mcg/kg per day, based on ideal patient body weight. The recommendation is limited to patients without risk factors, and approximates full levothyroxine replacement.
However, some patients enter cancer treatment with hypothyroidism, and some develop it de novo after beginning checkpoint inhibitor therapy. It is not known how best to treat each group, said Dr. Kristan.
To help answer that question, she and her collaborators at Georgetown University Hospital, McLean, Va., made use of a database drawn from five hospitals to perform a retrospective chart review. They looked at 822 patients who had received checkpoint inhibitor therapy, and from those patients, they selected 118 who had a diagnosis of hypothyroidism, or who received a prescription for levothyroxine during the 8-year study period.
The investigators assembled all available relevant data for each patient, including thyroid function tests, levothyroxine dosing, type of cancer, and type of therapy. They sorted participants into those who had received a diagnosis of hypothyroidism before or after receiving the first dose of checkpoint inhibitor therapy.
At baseline, 81 patients had preexisting hypothyroidism and were receiving a mean levothyroxine dose of 88.2 mcg. After treatment, the mean dose was 94.3 mcg, a nonsignificant difference. The median dose for this group remained at 88 mcg through treatment.
For the 37 patients who developed hypothyroidism de novo during checkpoint inhibitor therapy, the final observed levothyroxine dose was 71.2 mcg.
The mean age of the patients at baseline was 69 years. About half were women, and 91% were white. Either nivolumab or pembrolizumab was used in 72% of patients, making them the most commonly used checkpoint inhibitors, though 90% of patients received combination therapy. Taken together, melanoma and lung cancer accounted for about two-thirds of the cancers seen.
For both groups, the on-treatment levothyroxine dose was considerably lower than the ASCO-recommended, weight-based dosing, which would have been 122.9 mcg for those with preexisting hypothyroidism and 115.7 mcg for those who developed hypothyroidism on treatment (P less than .001 for both).
Dr. Kristan noted that thyroid stimulating hormone (TSH) values for patients with pretreatment hypothyroidism peaked between weeks 12 and 20, though there was no preemptive adjustment of levothyroxine dosing.
For those who developed on-treatment hypothyroidism, TSH values peaked at a series of times, at about weeks 8, 16, and 32. These waves of TSH elevation, she said, support the 4- to 6-week follow-up interval recommended in the ASCO guidelines.
However, she said, patients with de novo hypothyroidism “should not be started on the 1.6-mcg/kg-a-day weight-based dosing.” The cohort with de novo hypothyroidism in Dr. Kristan’s analysis required a daily dose of about 1 mcg/kg, she said. These real-world results support the idea that many patients on checkpoint inhibitors retain some thyroid reserve.
Dr. Kristan said that based on these findings, she and her collaborators recommend monitoring thyroid function every 4-6 weeks for patients taking immune checkpoint inhibitors. Patients with preexisting thyroid disease should not have an empiric adjustment of levothyroxine dose on checkpoint inhibitor initiation. For patients who develop thyroiditis after starting therapy, initiating a dose at 1 mcg/kg per day of ideal body weight is a good place to start, and treatment response should be monitored.
The study was limited by its retrospective nature and the small sample size, acknowledged Dr. Kristan. In addition, there were confounding variables and different frequencies of testing across institutions, and antibody status was not available and may have affected the results. Testing was performable for all participants.
Dr. Kristan said that the analysis opens up areas for further study, such as which patient populations are at risk for developing thyroid toxicity, what baseline characteristics can help predict which patients develop toxicity, and whether particular checkpoint inhibitors are more likely to cause toxicity. In addition, she said, a subset of patients will develop hyperthyroidism on checkpoint inhibitor therapy, and little is known about how to treat that complication.
Dr. Kristan reported no conflicts of interest. The research she presented was completed during her residency at Georgetown University.
SOURCE: Kristan M et al. ATA 2019, Oral Abstract 25.
CHICAGO – both for patients with preexisting and de novo hypothyroidism.
The real-world data, presented by Megan Kristan, MD, at the annual meeting of the American Thyroid Association, refine recommendations for dosing by body weight for levothyroxine in patients receiving checkpoint inhibitor therapy.
Immune checkpoint inhibitors stand a good chance of turning the tide against melanoma, some lung cancers, and other malignancies that have long been considered lethal. However, as more patients are exposed to the therapies, endocrinologists are seeing a wave of thyroid abnormalities, and must decide when, and at what doses, to treat hypothyroidism, said Dr. Kristan, a diabetes, endocrinology, and nutrition fellow at the University of Maryland, Baltimore.
Six checkpoint inhibitors are currently approved to hit a variety of molecular targets, and the prevalence of thyroid toxicity and hypothyroidism across the drug class ranges from a reported 9% to 40%, said Dr. Kristan.
The acknowledged thyroid toxicity of these drugs led the American Society for Clinical Oncology (ASCO) to issue guidelines advising that oncologists obtain baseline thyroid function tests before initiating checkpoint inhibitors, and that values be rechecked frequently – every 4-6 weeks – during therapy.
The guidelines advise dosing levothyroxine at approximately 1.6 mcg/kg per day, based on ideal patient body weight. The recommendation is limited to patients without risk factors, and approximates full levothyroxine replacement.
However, some patients enter cancer treatment with hypothyroidism, and some develop it de novo after beginning checkpoint inhibitor therapy. It is not known how best to treat each group, said Dr. Kristan.
To help answer that question, she and her collaborators at Georgetown University Hospital, McLean, Va., made use of a database drawn from five hospitals to perform a retrospective chart review. They looked at 822 patients who had received checkpoint inhibitor therapy, and from those patients, they selected 118 who had a diagnosis of hypothyroidism, or who received a prescription for levothyroxine during the 8-year study period.
The investigators assembled all available relevant data for each patient, including thyroid function tests, levothyroxine dosing, type of cancer, and type of therapy. They sorted participants into those who had received a diagnosis of hypothyroidism before or after receiving the first dose of checkpoint inhibitor therapy.
At baseline, 81 patients had preexisting hypothyroidism and were receiving a mean levothyroxine dose of 88.2 mcg. After treatment, the mean dose was 94.3 mcg, a nonsignificant difference. The median dose for this group remained at 88 mcg through treatment.
For the 37 patients who developed hypothyroidism de novo during checkpoint inhibitor therapy, the final observed levothyroxine dose was 71.2 mcg.
The mean age of the patients at baseline was 69 years. About half were women, and 91% were white. Either nivolumab or pembrolizumab was used in 72% of patients, making them the most commonly used checkpoint inhibitors, though 90% of patients received combination therapy. Taken together, melanoma and lung cancer accounted for about two-thirds of the cancers seen.
For both groups, the on-treatment levothyroxine dose was considerably lower than the ASCO-recommended, weight-based dosing, which would have been 122.9 mcg for those with preexisting hypothyroidism and 115.7 mcg for those who developed hypothyroidism on treatment (P less than .001 for both).
Dr. Kristan noted that thyroid stimulating hormone (TSH) values for patients with pretreatment hypothyroidism peaked between weeks 12 and 20, though there was no preemptive adjustment of levothyroxine dosing.
For those who developed on-treatment hypothyroidism, TSH values peaked at a series of times, at about weeks 8, 16, and 32. These waves of TSH elevation, she said, support the 4- to 6-week follow-up interval recommended in the ASCO guidelines.
However, she said, patients with de novo hypothyroidism “should not be started on the 1.6-mcg/kg-a-day weight-based dosing.” The cohort with de novo hypothyroidism in Dr. Kristan’s analysis required a daily dose of about 1 mcg/kg, she said. These real-world results support the idea that many patients on checkpoint inhibitors retain some thyroid reserve.
Dr. Kristan said that based on these findings, she and her collaborators recommend monitoring thyroid function every 4-6 weeks for patients taking immune checkpoint inhibitors. Patients with preexisting thyroid disease should not have an empiric adjustment of levothyroxine dose on checkpoint inhibitor initiation. For patients who develop thyroiditis after starting therapy, initiating a dose at 1 mcg/kg per day of ideal body weight is a good place to start, and treatment response should be monitored.
The study was limited by its retrospective nature and the small sample size, acknowledged Dr. Kristan. In addition, there were confounding variables and different frequencies of testing across institutions, and antibody status was not available and may have affected the results. Testing was performable for all participants.
Dr. Kristan said that the analysis opens up areas for further study, such as which patient populations are at risk for developing thyroid toxicity, what baseline characteristics can help predict which patients develop toxicity, and whether particular checkpoint inhibitors are more likely to cause toxicity. In addition, she said, a subset of patients will develop hyperthyroidism on checkpoint inhibitor therapy, and little is known about how to treat that complication.
Dr. Kristan reported no conflicts of interest. The research she presented was completed during her residency at Georgetown University.
SOURCE: Kristan M et al. ATA 2019, Oral Abstract 25.
REPORTING FROM ATA 2019