User login
Program Curbs Shoulder Morbidity Post Breast Cancer
SAN ANTONIO – Long-term upper-extremity pain and functional impairment following breast cancer surgery are reduced by an innovative physical therapy–centered surveillance program, according to a 5-year prospective study.
The program was developed at the breast care center of Walter Reed National Military Medical Center, Bethesda, Md. It begins with a preoperative patient interview and structured evaluation by a physical therapist. The assessment includes what physical therapists call an upper-quarter screen, which evaluates shoulder range of motion, strength, and arm volume. Self-reported pain, fatigue, function, and activity level are recorded.
At that time, the patient is also instructed in a home exercise program she is to start 2 weeks post surgery. These are simple range-of-motion exercises focused on forward flexion overhead; abduction; internal and external rotation of the glenohumeral joint; and scapular retraction, Nicole L. Stout explained at the San Antonio Breast Cancer Symposium.
The patient is subsequently seen by the physical therapist (in conjunction with her regular follow-up visits with an oncologist or surgeon) at 1, 3, 6, 9, 12, and 60 months post surgery. If upper-extremity dysfunction worsens, more intensive interventions are introduced.
The rationale for this prospective surveillance program lies in an understanding that the shoulder and arm impairments caused by breast cancer surgery and radiation therapy are typically subtle initially, but if left unidentified and uncorrected, they often progress to serious rotator cuff damage, "frozen" shoulder, and debilitating lymphedema, according to Ms. Stout, a civilian research physical therapist at the military center.
She had previously presented her 1-year prospective outcomes. This time around, she presented prospective 5-year follow-up data on 86 breast cancer patients who participated in the program. This is the first prospective cohort study in the United States to track breast cancer–related upper-extremity morbidity and functional outcomes for this long a time period.
The 12-month and 5-year visits included formal assessment of upper-extremity outcomes using the Harvard Alumni Activity Survey, the Upper Limb Disability Questionnaire, and the Short Form-36.
At 5 years, 11% of the women had stage I/II lymphedema and 38% had subclinical lymphedema (defined as less than 3% arm volume swelling as measured by a Perometer). These results compare favorably with those of published studies on lymphedema, which have reported long-term rates of stage I-III arm swelling in the 40%-60% range, she noted.
In all, 18% of patients reported arm numbness at 5 years, 17% noted shoulder pain, 3% had neck pain, and 1% had chest wall pain.
One-third of subjects reported fatigue of level 3 or more on a 10-point visual analog scale. Again, other investigators have documented long-term rates of clinically significant fatigue in the 50%-60% range.
At 5 years post surgery, patients demonstrated less impairment of physical activity than at preoperative baseline. At baseline, 33% had significant limitations in terms of carrying heavy objects, performing household chores, and other physical activities. After 5 years of prospective surveillance and tailored physical therapy, only 20% of patients had physical activity limitations.
However, 68% of patients reported experiencing some restrictions on their social activity at 5 years, and 52% experienced limitations on their recreational activities.
The study was funded by the medical center. Ms. Stout declared having no financial conflicts of interest.
SAN ANTONIO – Long-term upper-extremity pain and functional impairment following breast cancer surgery are reduced by an innovative physical therapy–centered surveillance program, according to a 5-year prospective study.
The program was developed at the breast care center of Walter Reed National Military Medical Center, Bethesda, Md. It begins with a preoperative patient interview and structured evaluation by a physical therapist. The assessment includes what physical therapists call an upper-quarter screen, which evaluates shoulder range of motion, strength, and arm volume. Self-reported pain, fatigue, function, and activity level are recorded.
At that time, the patient is also instructed in a home exercise program she is to start 2 weeks post surgery. These are simple range-of-motion exercises focused on forward flexion overhead; abduction; internal and external rotation of the glenohumeral joint; and scapular retraction, Nicole L. Stout explained at the San Antonio Breast Cancer Symposium.
The patient is subsequently seen by the physical therapist (in conjunction with her regular follow-up visits with an oncologist or surgeon) at 1, 3, 6, 9, 12, and 60 months post surgery. If upper-extremity dysfunction worsens, more intensive interventions are introduced.
The rationale for this prospective surveillance program lies in an understanding that the shoulder and arm impairments caused by breast cancer surgery and radiation therapy are typically subtle initially, but if left unidentified and uncorrected, they often progress to serious rotator cuff damage, "frozen" shoulder, and debilitating lymphedema, according to Ms. Stout, a civilian research physical therapist at the military center.
She had previously presented her 1-year prospective outcomes. This time around, she presented prospective 5-year follow-up data on 86 breast cancer patients who participated in the program. This is the first prospective cohort study in the United States to track breast cancer–related upper-extremity morbidity and functional outcomes for this long a time period.
The 12-month and 5-year visits included formal assessment of upper-extremity outcomes using the Harvard Alumni Activity Survey, the Upper Limb Disability Questionnaire, and the Short Form-36.
At 5 years, 11% of the women had stage I/II lymphedema and 38% had subclinical lymphedema (defined as less than 3% arm volume swelling as measured by a Perometer). These results compare favorably with those of published studies on lymphedema, which have reported long-term rates of stage I-III arm swelling in the 40%-60% range, she noted.
In all, 18% of patients reported arm numbness at 5 years, 17% noted shoulder pain, 3% had neck pain, and 1% had chest wall pain.
One-third of subjects reported fatigue of level 3 or more on a 10-point visual analog scale. Again, other investigators have documented long-term rates of clinically significant fatigue in the 50%-60% range.
At 5 years post surgery, patients demonstrated less impairment of physical activity than at preoperative baseline. At baseline, 33% had significant limitations in terms of carrying heavy objects, performing household chores, and other physical activities. After 5 years of prospective surveillance and tailored physical therapy, only 20% of patients had physical activity limitations.
However, 68% of patients reported experiencing some restrictions on their social activity at 5 years, and 52% experienced limitations on their recreational activities.
The study was funded by the medical center. Ms. Stout declared having no financial conflicts of interest.
SAN ANTONIO – Long-term upper-extremity pain and functional impairment following breast cancer surgery are reduced by an innovative physical therapy–centered surveillance program, according to a 5-year prospective study.
The program was developed at the breast care center of Walter Reed National Military Medical Center, Bethesda, Md. It begins with a preoperative patient interview and structured evaluation by a physical therapist. The assessment includes what physical therapists call an upper-quarter screen, which evaluates shoulder range of motion, strength, and arm volume. Self-reported pain, fatigue, function, and activity level are recorded.
At that time, the patient is also instructed in a home exercise program she is to start 2 weeks post surgery. These are simple range-of-motion exercises focused on forward flexion overhead; abduction; internal and external rotation of the glenohumeral joint; and scapular retraction, Nicole L. Stout explained at the San Antonio Breast Cancer Symposium.
The patient is subsequently seen by the physical therapist (in conjunction with her regular follow-up visits with an oncologist or surgeon) at 1, 3, 6, 9, 12, and 60 months post surgery. If upper-extremity dysfunction worsens, more intensive interventions are introduced.
The rationale for this prospective surveillance program lies in an understanding that the shoulder and arm impairments caused by breast cancer surgery and radiation therapy are typically subtle initially, but if left unidentified and uncorrected, they often progress to serious rotator cuff damage, "frozen" shoulder, and debilitating lymphedema, according to Ms. Stout, a civilian research physical therapist at the military center.
She had previously presented her 1-year prospective outcomes. This time around, she presented prospective 5-year follow-up data on 86 breast cancer patients who participated in the program. This is the first prospective cohort study in the United States to track breast cancer–related upper-extremity morbidity and functional outcomes for this long a time period.
The 12-month and 5-year visits included formal assessment of upper-extremity outcomes using the Harvard Alumni Activity Survey, the Upper Limb Disability Questionnaire, and the Short Form-36.
At 5 years, 11% of the women had stage I/II lymphedema and 38% had subclinical lymphedema (defined as less than 3% arm volume swelling as measured by a Perometer). These results compare favorably with those of published studies on lymphedema, which have reported long-term rates of stage I-III arm swelling in the 40%-60% range, she noted.
In all, 18% of patients reported arm numbness at 5 years, 17% noted shoulder pain, 3% had neck pain, and 1% had chest wall pain.
One-third of subjects reported fatigue of level 3 or more on a 10-point visual analog scale. Again, other investigators have documented long-term rates of clinically significant fatigue in the 50%-60% range.
At 5 years post surgery, patients demonstrated less impairment of physical activity than at preoperative baseline. At baseline, 33% had significant limitations in terms of carrying heavy objects, performing household chores, and other physical activities. After 5 years of prospective surveillance and tailored physical therapy, only 20% of patients had physical activity limitations.
However, 68% of patients reported experiencing some restrictions on their social activity at 5 years, and 52% experienced limitations on their recreational activities.
The study was funded by the medical center. Ms. Stout declared having no financial conflicts of interest.
FROM THE SAN ANTONIO BREAST CANCER SYMPOSIUM
Major Finding: After 5 years of participation in a surveillance/early intervention program for upper-extremity morbidity, only 11% of a group of breast cancer patients had stage I/II lymphedema, sharply lower than the 40%-60% long-term rates reported by other investigators.
Data Source: A 5-year prospective, uncontrolled, observational study to date involving 86 breast cancer patients.
Disclosures: The study was funded by the Walter Reed National Military Medical Center. Ms. Stout declared having no financial conflicts.
Family and Caregiver Needs Over the Course of the Cancer Trajectory
When a patient is diagnosed with cancer, family members often assume responsibility for providing care. They are typically involved not only with the diagnostic and treatment phases of care but also across the care trajectory and into survivorship. These caregivers are a primary source of support to individuals with cancer. The purpose of this article is to present an overview of the challenges, needs, and roles of family caregivers over the course of the cancer treatment trajectory and to discuss what support the professionals can provide.
Click of the PDF icon at the top of this introduction to read the full article.
When a patient is diagnosed with cancer, family members often assume responsibility for providing care. They are typically involved not only with the diagnostic and treatment phases of care but also across the care trajectory and into survivorship. These caregivers are a primary source of support to individuals with cancer. The purpose of this article is to present an overview of the challenges, needs, and roles of family caregivers over the course of the cancer treatment trajectory and to discuss what support the professionals can provide.
Click of the PDF icon at the top of this introduction to read the full article.
When a patient is diagnosed with cancer, family members often assume responsibility for providing care. They are typically involved not only with the diagnostic and treatment phases of care but also across the care trajectory and into survivorship. These caregivers are a primary source of support to individuals with cancer. The purpose of this article is to present an overview of the challenges, needs, and roles of family caregivers over the course of the cancer treatment trajectory and to discuss what support the professionals can provide.
Click of the PDF icon at the top of this introduction to read the full article.
SURVIVORSHIP Embracing the ‘new normal’
Since 1971, when President Richard M. Nixon announced the “war on cancer” with the signing of the National Cancer Act, we have seen an increase of 300% in the number of survivors, which is now reaching more than 12 million in the United States, according to the Centers for Disease Control. By 2020, that number will likely approach 20 million. Investment in research, early detection, and prevention has contributed to making these numbers a reality, and community-based oncology centers have played a critical role in delivering quality care and improved survival numbers based on the findings of that research. Therefore, it is logical that these same networks of community-based providers that have helped create survivors now help take the next step in addressing the needs of cancer patients on their journey to a life beyond cancer.
*For a PDF of the full article, click in the link to the left of this introduction.
Since 1971, when President Richard M. Nixon announced the “war on cancer” with the signing of the National Cancer Act, we have seen an increase of 300% in the number of survivors, which is now reaching more than 12 million in the United States, according to the Centers for Disease Control. By 2020, that number will likely approach 20 million. Investment in research, early detection, and prevention has contributed to making these numbers a reality, and community-based oncology centers have played a critical role in delivering quality care and improved survival numbers based on the findings of that research. Therefore, it is logical that these same networks of community-based providers that have helped create survivors now help take the next step in addressing the needs of cancer patients on their journey to a life beyond cancer.
*For a PDF of the full article, click in the link to the left of this introduction.
Since 1971, when President Richard M. Nixon announced the “war on cancer” with the signing of the National Cancer Act, we have seen an increase of 300% in the number of survivors, which is now reaching more than 12 million in the United States, according to the Centers for Disease Control. By 2020, that number will likely approach 20 million. Investment in research, early detection, and prevention has contributed to making these numbers a reality, and community-based oncology centers have played a critical role in delivering quality care and improved survival numbers based on the findings of that research. Therefore, it is logical that these same networks of community-based providers that have helped create survivors now help take the next step in addressing the needs of cancer patients on their journey to a life beyond cancer.
*For a PDF of the full article, click in the link to the left of this introduction.
FDA Issues One REMS for All Transmucosal Fentanyl
The Food and Drug Administration on Dec. 28 approved a class-wide risk management program for all transmucosal immediate-release fentanyl formulations.
The TIRF REMS [transmucosal immediate-release fentanyl Risk Evaluation and Mitigation Strategy] Access program is the agency’s first class-wide REMS for opioids. It replaces the individual REMS that exists for each product in the class now.
It will likely be the model for a REMS that the agency is developing for long-acting and extended-release opioids.
The REMS program is designed to reduce the risk of misuse, abuse, addiction, and overdose. REMS only applies to outpatient prescriptions. Hospitals, hospices and long-term care providers do not have to participate.
The TIRF REMS covers Abstral (fentanyl) sublingual tablet, Actiq (fentanyl citrate) oral transmucosal lozenge and its generic equivalents, Fentora (fentanyl citrate) buccal tablet, Lazanda (fentanyl) nasal spray, and Onsolis (fentanyl) buccal soluble film.
The FDA said that it developed a single risk management program for all the drugs to make management of these druigs easier for prescribers, pharmacists, distributors and outpatients. They all need to enroll only once to prescribe, dispense, or receive any of the products.
The program will start in March 2012. At that time, physicians will be able to enroll, after having completed an education program and assessment. Patients and health providers will also have to complete and sign an agreement form with the first prescription. And prescribers will have to give patients a Medication Guide that explains the risks, benefits, and proper use of the drug.
Health care providers who are already enrolled in an individual REMS program for at least one transmucosal immediate-release fentanyl medicine will not need to re-enroll because they will be automatically transitioned to the shared TIRF REMS Access program. Prescribers will be required to re-enroll in the TIRF REMS program every 2 years from the date of enrollment into either the individual REMS or TIRF REMS program, whichever was earlier.
The Food and Drug Administration on Dec. 28 approved a class-wide risk management program for all transmucosal immediate-release fentanyl formulations.
The TIRF REMS [transmucosal immediate-release fentanyl Risk Evaluation and Mitigation Strategy] Access program is the agency’s first class-wide REMS for opioids. It replaces the individual REMS that exists for each product in the class now.
It will likely be the model for a REMS that the agency is developing for long-acting and extended-release opioids.
The REMS program is designed to reduce the risk of misuse, abuse, addiction, and overdose. REMS only applies to outpatient prescriptions. Hospitals, hospices and long-term care providers do not have to participate.
The TIRF REMS covers Abstral (fentanyl) sublingual tablet, Actiq (fentanyl citrate) oral transmucosal lozenge and its generic equivalents, Fentora (fentanyl citrate) buccal tablet, Lazanda (fentanyl) nasal spray, and Onsolis (fentanyl) buccal soluble film.
The FDA said that it developed a single risk management program for all the drugs to make management of these druigs easier for prescribers, pharmacists, distributors and outpatients. They all need to enroll only once to prescribe, dispense, or receive any of the products.
The program will start in March 2012. At that time, physicians will be able to enroll, after having completed an education program and assessment. Patients and health providers will also have to complete and sign an agreement form with the first prescription. And prescribers will have to give patients a Medication Guide that explains the risks, benefits, and proper use of the drug.
Health care providers who are already enrolled in an individual REMS program for at least one transmucosal immediate-release fentanyl medicine will not need to re-enroll because they will be automatically transitioned to the shared TIRF REMS Access program. Prescribers will be required to re-enroll in the TIRF REMS program every 2 years from the date of enrollment into either the individual REMS or TIRF REMS program, whichever was earlier.
The Food and Drug Administration on Dec. 28 approved a class-wide risk management program for all transmucosal immediate-release fentanyl formulations.
The TIRF REMS [transmucosal immediate-release fentanyl Risk Evaluation and Mitigation Strategy] Access program is the agency’s first class-wide REMS for opioids. It replaces the individual REMS that exists for each product in the class now.
It will likely be the model for a REMS that the agency is developing for long-acting and extended-release opioids.
The REMS program is designed to reduce the risk of misuse, abuse, addiction, and overdose. REMS only applies to outpatient prescriptions. Hospitals, hospices and long-term care providers do not have to participate.
The TIRF REMS covers Abstral (fentanyl) sublingual tablet, Actiq (fentanyl citrate) oral transmucosal lozenge and its generic equivalents, Fentora (fentanyl citrate) buccal tablet, Lazanda (fentanyl) nasal spray, and Onsolis (fentanyl) buccal soluble film.
The FDA said that it developed a single risk management program for all the drugs to make management of these druigs easier for prescribers, pharmacists, distributors and outpatients. They all need to enroll only once to prescribe, dispense, or receive any of the products.
The program will start in March 2012. At that time, physicians will be able to enroll, after having completed an education program and assessment. Patients and health providers will also have to complete and sign an agreement form with the first prescription. And prescribers will have to give patients a Medication Guide that explains the risks, benefits, and proper use of the drug.
Health care providers who are already enrolled in an individual REMS program for at least one transmucosal immediate-release fentanyl medicine will not need to re-enroll because they will be automatically transitioned to the shared TIRF REMS Access program. Prescribers will be required to re-enroll in the TIRF REMS program every 2 years from the date of enrollment into either the individual REMS or TIRF REMS program, whichever was earlier.
We Asked, They Answered: What Is Palliative Care?
At the 7th Annual Chicago Supportive Oncology Conference, we asked leaders and experts in supportive oncology to define "palliative care." This is what they had to say:
At the 7th Annual Chicago Supportive Oncology Conference, we asked leaders and experts in supportive oncology to define "palliative care." This is what they had to say:
At the 7th Annual Chicago Supportive Oncology Conference, we asked leaders and experts in supportive oncology to define "palliative care." This is what they had to say:
Pre-Anthracycline-Based Chemo Cardiac Imaging Questioned
SAN ANTONIO – The guideline-recommended practice of routinely measuring left ventricular ejection fraction before anthracycline-based chemotherapy to screen out patients at increased risk for treatment-induced heart failure has come under fire as unproductive and financially wasteful.
It’s a practice endorsed by the American Heart Association and American College of Cardiology, enshrined in Food and Drug Administration labeling, required as part of most U.S. clinical trials, and common in community-based oncology practice.
Yet there are no data to support the utility of this practice as a screening tool aimed at minimizing heart failure induced by anthracycline-based chemotherapy, according to a report at the San Antonio Breast Cancer Symposium.
Dr. Seema M. Policepatil of the Gundersen Lutheran Medical Foundation, La Crosse, Wis., and colleagues presented a retrospective study that suggested routine cardiac ejection fraction screening under these circumstances is without merit. The study included 466 patients with early-stage, HER2-negative invasive breast cancer who were under consideration for anthracycline-based chemotherapy as part of their initial therapy. None had prior heart failure.
Left ventricular ejection fraction (LVEF) was measured by echocardiography, nuclear imaging, or MRI prior to chemotherapy in 241 of the patients. This reflects institutional practice: at Gundersen, pretreatment assessment of cardiac pump function is common but not uniform.
One of the 241 patients was found to have asymptomatic left ventricular dysfunction, with a screening ejection fraction of 48%, and she therefore didn’t receive anthracycline-based chemotherapy. Thus, modification of the treatment strategy in response to screening of ejection fraction occurred only rarely.
In addition, nine patients – six who had pretreatment cardiac imaging and three who did not – skipped the chemotherapy, either because of physician or patient preference or participation in clinical trials.
During a mean 5 years of follow-up, 3 of the remaining 456 women were diagnosed with heart failure: 2 among those with a pretreatment LVEF measurement, and 1 among those without it. That’s an acceptably low 0.7% event rate, she declared.
Current practice guidelines recommending pretreatment LVEF measurement are based upon expert consensus. It’s time to incorporate the available evidence, which in the case of the Gundersen experience doesn’t support the practice, Dr. Policepatil continued.
Assuming that nationally half of all patients with early-stage HER2-negative breast cancer undergo measurement of their LV ejection fraction before getting chemotherapy, eliminating this routine practice would save $7 million to $17 million annually based upon Medicare and Medicaid reimbursement rates, the physician added.
This study was funded by the Center for Cancer and Blood Disorders at the Gundersen Lutheran Medical Foundation. Dr. Policepatil declared having no financial conflicts.
SAN ANTONIO – The guideline-recommended practice of routinely measuring left ventricular ejection fraction before anthracycline-based chemotherapy to screen out patients at increased risk for treatment-induced heart failure has come under fire as unproductive and financially wasteful.
It’s a practice endorsed by the American Heart Association and American College of Cardiology, enshrined in Food and Drug Administration labeling, required as part of most U.S. clinical trials, and common in community-based oncology practice.
Yet there are no data to support the utility of this practice as a screening tool aimed at minimizing heart failure induced by anthracycline-based chemotherapy, according to a report at the San Antonio Breast Cancer Symposium.
Dr. Seema M. Policepatil of the Gundersen Lutheran Medical Foundation, La Crosse, Wis., and colleagues presented a retrospective study that suggested routine cardiac ejection fraction screening under these circumstances is without merit. The study included 466 patients with early-stage, HER2-negative invasive breast cancer who were under consideration for anthracycline-based chemotherapy as part of their initial therapy. None had prior heart failure.
Left ventricular ejection fraction (LVEF) was measured by echocardiography, nuclear imaging, or MRI prior to chemotherapy in 241 of the patients. This reflects institutional practice: at Gundersen, pretreatment assessment of cardiac pump function is common but not uniform.
One of the 241 patients was found to have asymptomatic left ventricular dysfunction, with a screening ejection fraction of 48%, and she therefore didn’t receive anthracycline-based chemotherapy. Thus, modification of the treatment strategy in response to screening of ejection fraction occurred only rarely.
In addition, nine patients – six who had pretreatment cardiac imaging and three who did not – skipped the chemotherapy, either because of physician or patient preference or participation in clinical trials.
During a mean 5 years of follow-up, 3 of the remaining 456 women were diagnosed with heart failure: 2 among those with a pretreatment LVEF measurement, and 1 among those without it. That’s an acceptably low 0.7% event rate, she declared.
Current practice guidelines recommending pretreatment LVEF measurement are based upon expert consensus. It’s time to incorporate the available evidence, which in the case of the Gundersen experience doesn’t support the practice, Dr. Policepatil continued.
Assuming that nationally half of all patients with early-stage HER2-negative breast cancer undergo measurement of their LV ejection fraction before getting chemotherapy, eliminating this routine practice would save $7 million to $17 million annually based upon Medicare and Medicaid reimbursement rates, the physician added.
This study was funded by the Center for Cancer and Blood Disorders at the Gundersen Lutheran Medical Foundation. Dr. Policepatil declared having no financial conflicts.
SAN ANTONIO – The guideline-recommended practice of routinely measuring left ventricular ejection fraction before anthracycline-based chemotherapy to screen out patients at increased risk for treatment-induced heart failure has come under fire as unproductive and financially wasteful.
It’s a practice endorsed by the American Heart Association and American College of Cardiology, enshrined in Food and Drug Administration labeling, required as part of most U.S. clinical trials, and common in community-based oncology practice.
Yet there are no data to support the utility of this practice as a screening tool aimed at minimizing heart failure induced by anthracycline-based chemotherapy, according to a report at the San Antonio Breast Cancer Symposium.
Dr. Seema M. Policepatil of the Gundersen Lutheran Medical Foundation, La Crosse, Wis., and colleagues presented a retrospective study that suggested routine cardiac ejection fraction screening under these circumstances is without merit. The study included 466 patients with early-stage, HER2-negative invasive breast cancer who were under consideration for anthracycline-based chemotherapy as part of their initial therapy. None had prior heart failure.
Left ventricular ejection fraction (LVEF) was measured by echocardiography, nuclear imaging, or MRI prior to chemotherapy in 241 of the patients. This reflects institutional practice: at Gundersen, pretreatment assessment of cardiac pump function is common but not uniform.
One of the 241 patients was found to have asymptomatic left ventricular dysfunction, with a screening ejection fraction of 48%, and she therefore didn’t receive anthracycline-based chemotherapy. Thus, modification of the treatment strategy in response to screening of ejection fraction occurred only rarely.
In addition, nine patients – six who had pretreatment cardiac imaging and three who did not – skipped the chemotherapy, either because of physician or patient preference or participation in clinical trials.
During a mean 5 years of follow-up, 3 of the remaining 456 women were diagnosed with heart failure: 2 among those with a pretreatment LVEF measurement, and 1 among those without it. That’s an acceptably low 0.7% event rate, she declared.
Current practice guidelines recommending pretreatment LVEF measurement are based upon expert consensus. It’s time to incorporate the available evidence, which in the case of the Gundersen experience doesn’t support the practice, Dr. Policepatil continued.
Assuming that nationally half of all patients with early-stage HER2-negative breast cancer undergo measurement of their LV ejection fraction before getting chemotherapy, eliminating this routine practice would save $7 million to $17 million annually based upon Medicare and Medicaid reimbursement rates, the physician added.
This study was funded by the Center for Cancer and Blood Disorders at the Gundersen Lutheran Medical Foundation. Dr. Policepatil declared having no financial conflicts.
FROM THE SAN ANTONIO BREAST CANCER SYMPOSIUM
Major Finding: Heart failure was diagnosed in three women within 5 years of anthracycline-based therapy – for an event rate of 0.7%.
Data Source: A single-center retrospective study of 466 breast cancer patients under consideration for anthracycline-based chemotherapy.
Disclosures: This study was funded by the Center for Cancer and Blood Disorders at the Gundersen Lutheran Medical Foundation. Dr. Policepatil declared having no financial conflicts.
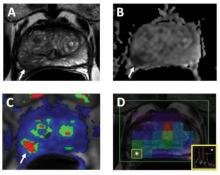
Multiparametric Magnetic Resonance Imaging Identifies Candidates for Prostate Cancer Surveillance
CHICAGO – Multiparametric magnetic resonance imaging trumped national guidelines in correctly classifying patients with prostate cancer as candidates for active surveillance in a retrospective study of 126 men.
National Comprehensive Cancer Network (NCCN) guidelines misclassified 22 of the 126 patients, compared with 12 using multiparametric magnetic resonance imaging (MP-MRI).
When MP-MRI was added to the NCCN criteria, only 5 patients were misclassified, Dr. Baris Turkbey reported in an award-winning paper at the annual meeting of the Radiological Society of North America.
"Presently, MRI is not in any urology guideline, but we want to change this," he said. "Our future goal is to create an NCI [National Cancer Institute] prostate cancer nomogram that includes multiparametric MRI, and our scientists are close to finishing it."
Dr. Turkbey, a fellow in the division of cancer treatment and diagnosis at the National Institutes of Health in Bethesda, Md., and his colleagues evaluated 126 men with biopsy-proven prostate cancer who underwent 3T MP-MRI of the prostate and subsequent radical prostatectomy at a median of 48 days. Their mean age was 59 years and mean prostate-specific antigen (PSA) level 6.67 ng/mL.
MP-MRI images were obtained of the largest and most aggressive lesion using T2-weighted MRI, diffusion-weighted MRI, MR spectroscopy, and dynamic contrast-enhanced MRI. Each dominant lesion was then assigned an MP-MRI score of low (at least two positive sequences), moderate (three positive sequences), or high (four positive sequences).
Patients were eligible for active surveillance on MP-MRI if they had a dominant tumor of less than 0.5 cm3 without extracapsular extension or seminal vesicle invasion and a low imaging score. The NCCN criteria for active surveillance are T1c disease, Gleason score of 6 or less, fewer than three positive biopsy cores, PSA less than 10 ng/mL, and PSA density less than 0.15 ng/mL/g.
Based on histopathological findings, 14 of 126 patents were eligible for active surveillance, with the remaining 112 candidates for active whole gland treatment.
NCCN guidelines wrongly classified 5 of 14 active surveillance patients and 17 of the 112 active treatment patients, whereas MP-MRI wrongly classified 1 active surveillance and 11 active treatment patients, Dr. Turkbey said.
The sensitivity, specificity, and overall accuracy of the NCCN guidelines were 64%, 35%, and 83%, respectively (P = .00002), compared with 93%, 54%, and 91% with MP-MRI (P less than .000001).
The study was limited by using a relatively simple, nonweighted MP-MRI scoring system and comparing MP-MRI with NCCN guidelines only, he acknowledged. Dr. Turkbey said the researchers are currently evaluating a system in which the various parameters are weighted to obtain better predictions.
When asked whether MP-MRI would be cost effective in routine clinical practice, Dr. Turkbey said that "compared to the costs of doing the wrong thing to a patient, an annual or semiannual MRI is well worth it."
Dr. Turkbey reported having no conflicts of interest. A coauthor reported serving as a researcher for Koninklijke Philips Electronics, General Electric, Siemens, Hoffman-La Roche, and iCAD.
National Comprehensive Cancer Network, NCCN, guidelines, misclassified, MP-MRI, Dr. Baris Turkbey, the Radiological Society of North America, NCI, National Cancer Institute, prostate cancer nomogram, multiparametric MRI, prostatectomy, MP-MRI images, MR spectroscopy, dynamic contrast-enhanced MRI,
CHICAGO – Multiparametric magnetic resonance imaging trumped national guidelines in correctly classifying patients with prostate cancer as candidates for active surveillance in a retrospective study of 126 men.
National Comprehensive Cancer Network (NCCN) guidelines misclassified 22 of the 126 patients, compared with 12 using multiparametric magnetic resonance imaging (MP-MRI).
When MP-MRI was added to the NCCN criteria, only 5 patients were misclassified, Dr. Baris Turkbey reported in an award-winning paper at the annual meeting of the Radiological Society of North America.
"Presently, MRI is not in any urology guideline, but we want to change this," he said. "Our future goal is to create an NCI [National Cancer Institute] prostate cancer nomogram that includes multiparametric MRI, and our scientists are close to finishing it."
Dr. Turkbey, a fellow in the division of cancer treatment and diagnosis at the National Institutes of Health in Bethesda, Md., and his colleagues evaluated 126 men with biopsy-proven prostate cancer who underwent 3T MP-MRI of the prostate and subsequent radical prostatectomy at a median of 48 days. Their mean age was 59 years and mean prostate-specific antigen (PSA) level 6.67 ng/mL.
MP-MRI images were obtained of the largest and most aggressive lesion using T2-weighted MRI, diffusion-weighted MRI, MR spectroscopy, and dynamic contrast-enhanced MRI. Each dominant lesion was then assigned an MP-MRI score of low (at least two positive sequences), moderate (three positive sequences), or high (four positive sequences).
Patients were eligible for active surveillance on MP-MRI if they had a dominant tumor of less than 0.5 cm3 without extracapsular extension or seminal vesicle invasion and a low imaging score. The NCCN criteria for active surveillance are T1c disease, Gleason score of 6 or less, fewer than three positive biopsy cores, PSA less than 10 ng/mL, and PSA density less than 0.15 ng/mL/g.
Based on histopathological findings, 14 of 126 patents were eligible for active surveillance, with the remaining 112 candidates for active whole gland treatment.
NCCN guidelines wrongly classified 5 of 14 active surveillance patients and 17 of the 112 active treatment patients, whereas MP-MRI wrongly classified 1 active surveillance and 11 active treatment patients, Dr. Turkbey said.
The sensitivity, specificity, and overall accuracy of the NCCN guidelines were 64%, 35%, and 83%, respectively (P = .00002), compared with 93%, 54%, and 91% with MP-MRI (P less than .000001).
The study was limited by using a relatively simple, nonweighted MP-MRI scoring system and comparing MP-MRI with NCCN guidelines only, he acknowledged. Dr. Turkbey said the researchers are currently evaluating a system in which the various parameters are weighted to obtain better predictions.
When asked whether MP-MRI would be cost effective in routine clinical practice, Dr. Turkbey said that "compared to the costs of doing the wrong thing to a patient, an annual or semiannual MRI is well worth it."
Dr. Turkbey reported having no conflicts of interest. A coauthor reported serving as a researcher for Koninklijke Philips Electronics, General Electric, Siemens, Hoffman-La Roche, and iCAD.
CHICAGO – Multiparametric magnetic resonance imaging trumped national guidelines in correctly classifying patients with prostate cancer as candidates for active surveillance in a retrospective study of 126 men.
National Comprehensive Cancer Network (NCCN) guidelines misclassified 22 of the 126 patients, compared with 12 using multiparametric magnetic resonance imaging (MP-MRI).
When MP-MRI was added to the NCCN criteria, only 5 patients were misclassified, Dr. Baris Turkbey reported in an award-winning paper at the annual meeting of the Radiological Society of North America.
"Presently, MRI is not in any urology guideline, but we want to change this," he said. "Our future goal is to create an NCI [National Cancer Institute] prostate cancer nomogram that includes multiparametric MRI, and our scientists are close to finishing it."
Dr. Turkbey, a fellow in the division of cancer treatment and diagnosis at the National Institutes of Health in Bethesda, Md., and his colleagues evaluated 126 men with biopsy-proven prostate cancer who underwent 3T MP-MRI of the prostate and subsequent radical prostatectomy at a median of 48 days. Their mean age was 59 years and mean prostate-specific antigen (PSA) level 6.67 ng/mL.
MP-MRI images were obtained of the largest and most aggressive lesion using T2-weighted MRI, diffusion-weighted MRI, MR spectroscopy, and dynamic contrast-enhanced MRI. Each dominant lesion was then assigned an MP-MRI score of low (at least two positive sequences), moderate (three positive sequences), or high (four positive sequences).
Patients were eligible for active surveillance on MP-MRI if they had a dominant tumor of less than 0.5 cm3 without extracapsular extension or seminal vesicle invasion and a low imaging score. The NCCN criteria for active surveillance are T1c disease, Gleason score of 6 or less, fewer than three positive biopsy cores, PSA less than 10 ng/mL, and PSA density less than 0.15 ng/mL/g.
Based on histopathological findings, 14 of 126 patents were eligible for active surveillance, with the remaining 112 candidates for active whole gland treatment.
NCCN guidelines wrongly classified 5 of 14 active surveillance patients and 17 of the 112 active treatment patients, whereas MP-MRI wrongly classified 1 active surveillance and 11 active treatment patients, Dr. Turkbey said.
The sensitivity, specificity, and overall accuracy of the NCCN guidelines were 64%, 35%, and 83%, respectively (P = .00002), compared with 93%, 54%, and 91% with MP-MRI (P less than .000001).
The study was limited by using a relatively simple, nonweighted MP-MRI scoring system and comparing MP-MRI with NCCN guidelines only, he acknowledged. Dr. Turkbey said the researchers are currently evaluating a system in which the various parameters are weighted to obtain better predictions.
When asked whether MP-MRI would be cost effective in routine clinical practice, Dr. Turkbey said that "compared to the costs of doing the wrong thing to a patient, an annual or semiannual MRI is well worth it."
Dr. Turkbey reported having no conflicts of interest. A coauthor reported serving as a researcher for Koninklijke Philips Electronics, General Electric, Siemens, Hoffman-La Roche, and iCAD.
National Comprehensive Cancer Network, NCCN, guidelines, misclassified, MP-MRI, Dr. Baris Turkbey, the Radiological Society of North America, NCI, National Cancer Institute, prostate cancer nomogram, multiparametric MRI, prostatectomy, MP-MRI images, MR spectroscopy, dynamic contrast-enhanced MRI,
National Comprehensive Cancer Network, NCCN, guidelines, misclassified, MP-MRI, Dr. Baris Turkbey, the Radiological Society of North America, NCI, National Cancer Institute, prostate cancer nomogram, multiparametric MRI, prostatectomy, MP-MRI images, MR spectroscopy, dynamic contrast-enhanced MRI,
FROM THE ANNUAL MEETING OF THE RADIOLOGICAL SOCIETY OF NORTH AMERICA
Major Finding: The sensitivity, specificity and overall accuracy of the NCCN guidelines were 64%, 35%, and 83% (P = .00002), compared with 93%, 54%, and 91% with MP-MRI (P less than .000001).
Data Source: Retrospective analysis of 126 patients with prostate cancer.
Disclosures: Dr. Turkbey reported no conflicts of interest. A coauthor reported serving as a researcher for Koninklijke Philips Electronics, General Electric, Siemens, Hoffman-La Roche, and iCAD.
Panel Endorses Surveillance for Low-Risk Prostate Cancer
An independent panel convened by the U.S. National Institutes of Health has endorsed the use of active surveillance and delay of treatment for men with localized, low-risk prostate cancer.
"Our panel found that many men with localized, low-risk prostate cancer should be closely monitored, permitting their treatment to be delayed until disease progression warrants it. Some of the men affected by prostate cancer will benefit from immediate treatment and others will benefit from observation," panel and conference chairperson Dr. Patricia A. Ganz said in a telebriefing.
However, monitoring or observational strategies, sometimes referred to as "watchful waiting" or "active surveillance," have not been uniformly studied, and available data do not yet point to clear follow-up protocols, said Dr. Ganz, director of the division of cancer prevention and control research at the Jonsson Comprehensive Cancer Center, University of California, Los Angeles.
The panel identified the combination of a prostate-specific antigen (PSA) level less than 10 ng/dL and a Gleason score of 6 or lower as the emerging consensus definition of "low-risk" disease. "Using this definition, we estimate that more than 100,000 men diagnosed with prostate cancer each year in the United States would be candidates for active monitoring rather than immediate treatment," she said.
Additional research will be needed to determine the point at which treatment might ultimately be needed for men who are being closely monitored. The panel recommended that such studies receiving federal funding should not be done at a single site, but rather should be multicenter studies with large patient populations.
The consensus about active surveillance came from increasing evidence that outcomes for men with low-risk disease are not better for those who undergo surgery or radiation therapy. Among the evidence considered was the newly reported findings of the Prostate Cancer Intervention Versus Observation Trial (PIVOT), the only randomized controlled trial conducted on men identified via PSA screening that compared watchful waiting with radical prostatectomy.
With a median follow-up of 10 years, there were no statistically significant differences in prostate cancer mortality or all-cause mortality. "Supporting data from additional cohort studies give us confidence that the risk of death is minimal in a low risk population," the panel wrote.
According to the document, only about 10% of men eligible for observational strategies choose this approach. The reasons for this are probably due both to physician communication that favors treatment and to patient expectations. "When men are given a diagnosis of cancer, it’s very difficult to decline the standard therapy for this disease, which is either surgery or radiation therapy," Dr. Ganz noted.
For this reason, the panel also endorsed consideration of a name change to remove the anxiety-provoking term cancer to describe this low-risk condition, much as was done with cervical intraepithelial neoplasia for early-stage cervical neoplasms and ductal carcinoma in situ for lower-risk breast lesions.
"Some of the men affected by prostate cancer will benefit from immediate treatment and others will benefit from observation."
They also discussed ways in which urologists and primary care physicians ordering PSA tests could be educated to counsel patients about the potential benefits of active surveillance and delaying treatment, Dr. Ganz said.
"Anything we can do to bring this into the consultation room so that the patient feels comfortable raising this issue with his physician ... [Stakeholders] now have an NIH-vetted document that describes this as a reasonable approach to the management of prostate cancer and for those reasons it can be very powerful," she said.
The 14-member state-of-the-science panel included experts in the fields of cancer prevention and control, urology, pathology, epidemiology, genetics, transplantation, bioethics, economics, health services research, shared decision-making, health communication, and community engagement.
Panel members were compensated for travel to the conference, but were not otherwise paid and have no additional conflicts of interest. The statement does not constitute federal policy.
The draft statement is posted online, and the final version will be posted by mid to late January 2012.
An independent panel convened by the U.S. National Institutes of Health has endorsed the use of active surveillance and delay of treatment for men with localized, low-risk prostate cancer.
"Our panel found that many men with localized, low-risk prostate cancer should be closely monitored, permitting their treatment to be delayed until disease progression warrants it. Some of the men affected by prostate cancer will benefit from immediate treatment and others will benefit from observation," panel and conference chairperson Dr. Patricia A. Ganz said in a telebriefing.
However, monitoring or observational strategies, sometimes referred to as "watchful waiting" or "active surveillance," have not been uniformly studied, and available data do not yet point to clear follow-up protocols, said Dr. Ganz, director of the division of cancer prevention and control research at the Jonsson Comprehensive Cancer Center, University of California, Los Angeles.
The panel identified the combination of a prostate-specific antigen (PSA) level less than 10 ng/dL and a Gleason score of 6 or lower as the emerging consensus definition of "low-risk" disease. "Using this definition, we estimate that more than 100,000 men diagnosed with prostate cancer each year in the United States would be candidates for active monitoring rather than immediate treatment," she said.
Additional research will be needed to determine the point at which treatment might ultimately be needed for men who are being closely monitored. The panel recommended that such studies receiving federal funding should not be done at a single site, but rather should be multicenter studies with large patient populations.
The consensus about active surveillance came from increasing evidence that outcomes for men with low-risk disease are not better for those who undergo surgery or radiation therapy. Among the evidence considered was the newly reported findings of the Prostate Cancer Intervention Versus Observation Trial (PIVOT), the only randomized controlled trial conducted on men identified via PSA screening that compared watchful waiting with radical prostatectomy.
With a median follow-up of 10 years, there were no statistically significant differences in prostate cancer mortality or all-cause mortality. "Supporting data from additional cohort studies give us confidence that the risk of death is minimal in a low risk population," the panel wrote.
According to the document, only about 10% of men eligible for observational strategies choose this approach. The reasons for this are probably due both to physician communication that favors treatment and to patient expectations. "When men are given a diagnosis of cancer, it’s very difficult to decline the standard therapy for this disease, which is either surgery or radiation therapy," Dr. Ganz noted.
For this reason, the panel also endorsed consideration of a name change to remove the anxiety-provoking term cancer to describe this low-risk condition, much as was done with cervical intraepithelial neoplasia for early-stage cervical neoplasms and ductal carcinoma in situ for lower-risk breast lesions.
"Some of the men affected by prostate cancer will benefit from immediate treatment and others will benefit from observation."
They also discussed ways in which urologists and primary care physicians ordering PSA tests could be educated to counsel patients about the potential benefits of active surveillance and delaying treatment, Dr. Ganz said.
"Anything we can do to bring this into the consultation room so that the patient feels comfortable raising this issue with his physician ... [Stakeholders] now have an NIH-vetted document that describes this as a reasonable approach to the management of prostate cancer and for those reasons it can be very powerful," she said.
The 14-member state-of-the-science panel included experts in the fields of cancer prevention and control, urology, pathology, epidemiology, genetics, transplantation, bioethics, economics, health services research, shared decision-making, health communication, and community engagement.
Panel members were compensated for travel to the conference, but were not otherwise paid and have no additional conflicts of interest. The statement does not constitute federal policy.
The draft statement is posted online, and the final version will be posted by mid to late January 2012.
An independent panel convened by the U.S. National Institutes of Health has endorsed the use of active surveillance and delay of treatment for men with localized, low-risk prostate cancer.
"Our panel found that many men with localized, low-risk prostate cancer should be closely monitored, permitting their treatment to be delayed until disease progression warrants it. Some of the men affected by prostate cancer will benefit from immediate treatment and others will benefit from observation," panel and conference chairperson Dr. Patricia A. Ganz said in a telebriefing.
However, monitoring or observational strategies, sometimes referred to as "watchful waiting" or "active surveillance," have not been uniformly studied, and available data do not yet point to clear follow-up protocols, said Dr. Ganz, director of the division of cancer prevention and control research at the Jonsson Comprehensive Cancer Center, University of California, Los Angeles.
The panel identified the combination of a prostate-specific antigen (PSA) level less than 10 ng/dL and a Gleason score of 6 or lower as the emerging consensus definition of "low-risk" disease. "Using this definition, we estimate that more than 100,000 men diagnosed with prostate cancer each year in the United States would be candidates for active monitoring rather than immediate treatment," she said.
Additional research will be needed to determine the point at which treatment might ultimately be needed for men who are being closely monitored. The panel recommended that such studies receiving federal funding should not be done at a single site, but rather should be multicenter studies with large patient populations.
The consensus about active surveillance came from increasing evidence that outcomes for men with low-risk disease are not better for those who undergo surgery or radiation therapy. Among the evidence considered was the newly reported findings of the Prostate Cancer Intervention Versus Observation Trial (PIVOT), the only randomized controlled trial conducted on men identified via PSA screening that compared watchful waiting with radical prostatectomy.
With a median follow-up of 10 years, there were no statistically significant differences in prostate cancer mortality or all-cause mortality. "Supporting data from additional cohort studies give us confidence that the risk of death is minimal in a low risk population," the panel wrote.
According to the document, only about 10% of men eligible for observational strategies choose this approach. The reasons for this are probably due both to physician communication that favors treatment and to patient expectations. "When men are given a diagnosis of cancer, it’s very difficult to decline the standard therapy for this disease, which is either surgery or radiation therapy," Dr. Ganz noted.
For this reason, the panel also endorsed consideration of a name change to remove the anxiety-provoking term cancer to describe this low-risk condition, much as was done with cervical intraepithelial neoplasia for early-stage cervical neoplasms and ductal carcinoma in situ for lower-risk breast lesions.
"Some of the men affected by prostate cancer will benefit from immediate treatment and others will benefit from observation."
They also discussed ways in which urologists and primary care physicians ordering PSA tests could be educated to counsel patients about the potential benefits of active surveillance and delaying treatment, Dr. Ganz said.
"Anything we can do to bring this into the consultation room so that the patient feels comfortable raising this issue with his physician ... [Stakeholders] now have an NIH-vetted document that describes this as a reasonable approach to the management of prostate cancer and for those reasons it can be very powerful," she said.
The 14-member state-of-the-science panel included experts in the fields of cancer prevention and control, urology, pathology, epidemiology, genetics, transplantation, bioethics, economics, health services research, shared decision-making, health communication, and community engagement.
Panel members were compensated for travel to the conference, but were not otherwise paid and have no additional conflicts of interest. The statement does not constitute federal policy.
The draft statement is posted online, and the final version will be posted by mid to late January 2012.
Major Finding: Many men with localized, low-risk prostate cancer should be closely monitored, permitting their treatment to be delayed until disease progression warrants it.
Data Source: Newly reported findings of the Prostate Cancer Intervention Versus Observation Trial (PIVOT).
Disclosures: Panel members were compensated for travel to the conference, but were not otherwise paid and have no additional conflicts of interest. The statement does not constitute federal policy.
Practice Changers Expected at San Antonio Breast Cancer Symposium
The 2011 San Antonio Breast Cancer Symposium that opened Dec. 6 features a hefty number of studies that could change clinical practice in the treatment of breast cancer.
Oncologists have been eagerly awaiting data from the phase III BOLERO-2 and CLEOPATRA trials, which are to be presented Dec. 8 and 9, respectively.
– BOLERO-2 investigators reported that pairing everolimus (Afinitor) with exemestane (Aromasin) increased median progression-free survival by 4.1 months in an interim analysis presented this fall at the European Multidisciplinary Cancer Congress in Stockholm. Women in this study had estrogen receptor-positive disease that was resistant to hormone therapy.
– Genentech announced last summer that dual HER2 blockade with pertuzumab* and trastuzumab (Herceptin) improved progression-free survival for women with HER2-positive disease who also received docetaxel (Taxotere) in CLEOPATRA. The pairing has also produced good results in the NeoSphere trial, and early reports from studies pairing lapatinib (Tykerb) with trastuzumab have been encouraging.
On Dec. 7, an early-morning press briefing has been scheduled for investigators to outline findings of four noteworthy studies.
– Swedish researchers will report that diabetes and obesity after age 60 are risk factors for breast cancer. Low lipids also increased risk, but high lipids did not in their study comparing medical records of more than 23,000 women. Similarly, risk went up with use of the diabetes drug glargine but down with metformin.
– Gene expression patterns at diagnosis of hormone receptor–positive breast cancer can predict which women will have recurrences late, early, or not at all, according to researchers from the United States.
– Women treated with brachytherapy were about twice as likely to undergo subsequent mastectomy when compared with those treated by whole breast irradiation in a study of more than 150,000 Medicare claims.
– A ductal carcinoma in situ (DCIS) risk score based on the multigene Oncotype DX assay has been validated for prediction of breast cancer recurrence in DCIS patients.
Other Dec. 7 highlights will include presentations from four studies of bisphosphonate use in breast cancer patients and an Institute of Medicine report on "Breast Cancer and the Environment."
For ongoing coverage of these and other presentations at the San Antonio Breast Cancer Symposium, visit http://www.oncologyreport.com.
*Correction, Dec. 8, 2011: An earlier version of this story referred to pertuzumab by the trade name Omnitarg. That trade name was discontinued by Genentech in 2007 and currently the drug goes by pertuzumab.
The 2011 San Antonio Breast Cancer Symposium that opened Dec. 6 features a hefty number of studies that could change clinical practice in the treatment of breast cancer.
Oncologists have been eagerly awaiting data from the phase III BOLERO-2 and CLEOPATRA trials, which are to be presented Dec. 8 and 9, respectively.
– BOLERO-2 investigators reported that pairing everolimus (Afinitor) with exemestane (Aromasin) increased median progression-free survival by 4.1 months in an interim analysis presented this fall at the European Multidisciplinary Cancer Congress in Stockholm. Women in this study had estrogen receptor-positive disease that was resistant to hormone therapy.
– Genentech announced last summer that dual HER2 blockade with pertuzumab* and trastuzumab (Herceptin) improved progression-free survival for women with HER2-positive disease who also received docetaxel (Taxotere) in CLEOPATRA. The pairing has also produced good results in the NeoSphere trial, and early reports from studies pairing lapatinib (Tykerb) with trastuzumab have been encouraging.
On Dec. 7, an early-morning press briefing has been scheduled for investigators to outline findings of four noteworthy studies.
– Swedish researchers will report that diabetes and obesity after age 60 are risk factors for breast cancer. Low lipids also increased risk, but high lipids did not in their study comparing medical records of more than 23,000 women. Similarly, risk went up with use of the diabetes drug glargine but down with metformin.
– Gene expression patterns at diagnosis of hormone receptor–positive breast cancer can predict which women will have recurrences late, early, or not at all, according to researchers from the United States.
– Women treated with brachytherapy were about twice as likely to undergo subsequent mastectomy when compared with those treated by whole breast irradiation in a study of more than 150,000 Medicare claims.
– A ductal carcinoma in situ (DCIS) risk score based on the multigene Oncotype DX assay has been validated for prediction of breast cancer recurrence in DCIS patients.
Other Dec. 7 highlights will include presentations from four studies of bisphosphonate use in breast cancer patients and an Institute of Medicine report on "Breast Cancer and the Environment."
For ongoing coverage of these and other presentations at the San Antonio Breast Cancer Symposium, visit http://www.oncologyreport.com.
*Correction, Dec. 8, 2011: An earlier version of this story referred to pertuzumab by the trade name Omnitarg. That trade name was discontinued by Genentech in 2007 and currently the drug goes by pertuzumab.
The 2011 San Antonio Breast Cancer Symposium that opened Dec. 6 features a hefty number of studies that could change clinical practice in the treatment of breast cancer.
Oncologists have been eagerly awaiting data from the phase III BOLERO-2 and CLEOPATRA trials, which are to be presented Dec. 8 and 9, respectively.
– BOLERO-2 investigators reported that pairing everolimus (Afinitor) with exemestane (Aromasin) increased median progression-free survival by 4.1 months in an interim analysis presented this fall at the European Multidisciplinary Cancer Congress in Stockholm. Women in this study had estrogen receptor-positive disease that was resistant to hormone therapy.
– Genentech announced last summer that dual HER2 blockade with pertuzumab* and trastuzumab (Herceptin) improved progression-free survival for women with HER2-positive disease who also received docetaxel (Taxotere) in CLEOPATRA. The pairing has also produced good results in the NeoSphere trial, and early reports from studies pairing lapatinib (Tykerb) with trastuzumab have been encouraging.
On Dec. 7, an early-morning press briefing has been scheduled for investigators to outline findings of four noteworthy studies.
– Swedish researchers will report that diabetes and obesity after age 60 are risk factors for breast cancer. Low lipids also increased risk, but high lipids did not in their study comparing medical records of more than 23,000 women. Similarly, risk went up with use of the diabetes drug glargine but down with metformin.
– Gene expression patterns at diagnosis of hormone receptor–positive breast cancer can predict which women will have recurrences late, early, or not at all, according to researchers from the United States.
– Women treated with brachytherapy were about twice as likely to undergo subsequent mastectomy when compared with those treated by whole breast irradiation in a study of more than 150,000 Medicare claims.
– A ductal carcinoma in situ (DCIS) risk score based on the multigene Oncotype DX assay has been validated for prediction of breast cancer recurrence in DCIS patients.
Other Dec. 7 highlights will include presentations from four studies of bisphosphonate use in breast cancer patients and an Institute of Medicine report on "Breast Cancer and the Environment."
For ongoing coverage of these and other presentations at the San Antonio Breast Cancer Symposium, visit http://www.oncologyreport.com.
*Correction, Dec. 8, 2011: An earlier version of this story referred to pertuzumab by the trade name Omnitarg. That trade name was discontinued by Genentech in 2007 and currently the drug goes by pertuzumab.
Stratify Patients for Opioid Misuse and Abuse Risk
NEW ORLEANS – Do you know which of your patients is likely to misuse or abuse opioid medications?
They can be stratified according to risk, but many physicians don’t know how or don’t take the time to do so, according to Dr. Lynn R. Webster, medical director of Lifetree Clinical Research in Salt Lake City and director at large for the American Academy of Pain Medicine.
It is important to try to prevent misuse or abuse, given that overdoses and deaths from prescription drugs are on the rise, said Dr. Webster at the American Medical Association House of Delegates Interim meeting.
According to the National Institute on Drug Abuse, emergency department visits involving nonmedical use of pharmaceuticals (either alone or in combination with another drug) increased 98% between 2004 and 2009, from 627,291 visits to 1.2 million visits. There was an 83% rise in emergency department visits involving adverse reactions to pharmaceuticals between 2005 and 2009, from 1.3 million to 2.3 million visits.
Opioids were the most frequently cited in those ED visits, accounting for about 50% of nonmedical use. Psychotherapeutic agents accounted for a third of the nonmedical use. According to the National Institute on Drug Abuse, the three most frequently cited drugs in those visits were hydrocodone (alone or in combination), accounting for 104,490 visits; oxycodone (alone or in combination), for 175,949 visits; and methadone, for 70,637 visits.
To properly assess and treat patients, physicians should understand and agree upon terminology, said Dr. Webster. Patients may "misuse" prescriptions if they are not taking them as directed. They aren’t seeking a high, they may just have undertreated pain, he said.
Abuse is the willful self-administration of a drug to get high. Addiction is a medical condition with complex genetic, psychosocial, and environmental factors that influence its development and manifestations. Addicts may have impaired control over drug use, compulsive use, or continued use despite harm.
"You don’t just put someone on an opioid and think you’ve done your job."
Pseudoaddiction results from undertreatment of pain, and may resolve with proper therapy. Patients in this state may manifest behavior that is misidentified as drug-seeking, according to Dr. Webster.
Even with chronic pain, "people prescribed opioids over a long period of time are going to have aberrant behavior," said Dr. Webster, noting that "it’s not a small problem." In research he conducted, up to 40% of patients in his chronic pain practice had aberrant behavior within a year of starting treatment.
But, he said, "only a small percent have the disease of addiction."
To prevent misuse and abuse, it’s important to understand patients’ motives, he said. Nonmedical users may have been prescribed pain medications, but they move on to recreational use and then, often, abuse.
Medical users generally are pain patients who are seeking more pain relief and end up misusing the drugs. They also may become abusers. In all, 40%-60% have other chronic conditions for which opioids provide relief, such as anxiety, said Dr. Webster. That use is obviously not appropriate, but it can only be prevented if physicians fully assess pain patients.
There is a constellation of risk factors that help determine potential for misuse and abuse, according to Dr. Webster.
The potency of a pharmacologic substance and how fast it works are important, as those factors help determine how much dopamine is released, and thus, the amount of reward the patient feels, said Dr. Webster. If the drug is readily available, that also determines its attractiveness.
One-fifth of the overall population is at risk for addiction. There is no on/off switch, but a continuum of risk, he explained. Biological, psychiatric, and social factors all play a role in where patients fall on that continuum. Age, gender, family history, cigarette smoking, pain severity and duration, and sleep disorders are all risk factors. Patients with a history of a psychiatric disorder, being sexual abused, or substance use disorder also are at higher risk. "We must assess mental health," said Dr. Webster, noting that patients with anxiety, depression, traumatic brain injury, and attention-deficit/hyperactivity disorder are at risk for self-medicating and overdose.
A history of legal problems and motor vehicle accidents, or poor family support and unemployment also are predictors, Dr. Webster said.
Aberrant behaviors are directly related to the duration and the intensity of pain. Patients who cannot completely eliminate their pain may resort to taking more medication, which they will see as less of a wrong than the pain itself, he said.
Physicians generally have not done a good job at evaluating these risks, and selecting and managing patients according to these factors. But how patients are managed can help determine whether they end up in the emergency room, "or going to a substance abuse treatment center or the morgue," he said.
There are tools that can be used to screen patients for risk. Among those are the Screener and Opioid Assessment for Patients With Pain (SOAPP); the Opioid Risk Tool (ORT); the Diagnosis, Intractability, Risk Efficacy (DIRE) test; and the Current Opioid Misuse Measure (COMM), which is used for patients already taking opioids.
These tests all are simple and take only 1 to 10 minutes to administer. They can be given by any physician, including primary care physicians, he said.
Once all the factors are taken into account, patients can be stratified according to risk. Lower-risk patients – those with moderate pain and no personal or family history of substance use disorders – can be managed by primary care physicians. Those at moderate risk may need the addition of a pain specialist. The higher-risk patients will need consultations with mental health professionals and pain physicians.
Monitoring higher-risk patients may require urine drug testing and prescription monitoring programs. Pharmacists should be kept in the loop to see whether patients are shopping around. Family and friends can be invaluable sources to help identify risky behaviors.
Physicians need to do a better job of titrating opioids and determining proper dosing. Many patients have died at initiation of opioid therapy, or from rotating from one drug to another, he said. Patients should not be allowed to determine how much of a long-acting medication they can take.
So how do doctors mitigate risk? The first order of business is to treat the pain. "Uncontrolled pain, untreated pain, I believe is the number one reason for aberrant behavior," said Dr. Webster. Physicians cannot eliminate all pain, however, so education also is crucial.
Physicians also have a duty to monitor pain patients. "You don’t just put someone on an opioid and think you’ve done your job," said Dr. Webster. A doctor would not stop managing a diabetic; pain patients need long-term management, he said.
Dr. Webster reported having no relevant conflicts of interest.
NEW ORLEANS – Do you know which of your patients is likely to misuse or abuse opioid medications?
They can be stratified according to risk, but many physicians don’t know how or don’t take the time to do so, according to Dr. Lynn R. Webster, medical director of Lifetree Clinical Research in Salt Lake City and director at large for the American Academy of Pain Medicine.
It is important to try to prevent misuse or abuse, given that overdoses and deaths from prescription drugs are on the rise, said Dr. Webster at the American Medical Association House of Delegates Interim meeting.
According to the National Institute on Drug Abuse, emergency department visits involving nonmedical use of pharmaceuticals (either alone or in combination with another drug) increased 98% between 2004 and 2009, from 627,291 visits to 1.2 million visits. There was an 83% rise in emergency department visits involving adverse reactions to pharmaceuticals between 2005 and 2009, from 1.3 million to 2.3 million visits.
Opioids were the most frequently cited in those ED visits, accounting for about 50% of nonmedical use. Psychotherapeutic agents accounted for a third of the nonmedical use. According to the National Institute on Drug Abuse, the three most frequently cited drugs in those visits were hydrocodone (alone or in combination), accounting for 104,490 visits; oxycodone (alone or in combination), for 175,949 visits; and methadone, for 70,637 visits.
To properly assess and treat patients, physicians should understand and agree upon terminology, said Dr. Webster. Patients may "misuse" prescriptions if they are not taking them as directed. They aren’t seeking a high, they may just have undertreated pain, he said.
Abuse is the willful self-administration of a drug to get high. Addiction is a medical condition with complex genetic, psychosocial, and environmental factors that influence its development and manifestations. Addicts may have impaired control over drug use, compulsive use, or continued use despite harm.
"You don’t just put someone on an opioid and think you’ve done your job."
Pseudoaddiction results from undertreatment of pain, and may resolve with proper therapy. Patients in this state may manifest behavior that is misidentified as drug-seeking, according to Dr. Webster.
Even with chronic pain, "people prescribed opioids over a long period of time are going to have aberrant behavior," said Dr. Webster, noting that "it’s not a small problem." In research he conducted, up to 40% of patients in his chronic pain practice had aberrant behavior within a year of starting treatment.
But, he said, "only a small percent have the disease of addiction."
To prevent misuse and abuse, it’s important to understand patients’ motives, he said. Nonmedical users may have been prescribed pain medications, but they move on to recreational use and then, often, abuse.
Medical users generally are pain patients who are seeking more pain relief and end up misusing the drugs. They also may become abusers. In all, 40%-60% have other chronic conditions for which opioids provide relief, such as anxiety, said Dr. Webster. That use is obviously not appropriate, but it can only be prevented if physicians fully assess pain patients.
There is a constellation of risk factors that help determine potential for misuse and abuse, according to Dr. Webster.
The potency of a pharmacologic substance and how fast it works are important, as those factors help determine how much dopamine is released, and thus, the amount of reward the patient feels, said Dr. Webster. If the drug is readily available, that also determines its attractiveness.
One-fifth of the overall population is at risk for addiction. There is no on/off switch, but a continuum of risk, he explained. Biological, psychiatric, and social factors all play a role in where patients fall on that continuum. Age, gender, family history, cigarette smoking, pain severity and duration, and sleep disorders are all risk factors. Patients with a history of a psychiatric disorder, being sexual abused, or substance use disorder also are at higher risk. "We must assess mental health," said Dr. Webster, noting that patients with anxiety, depression, traumatic brain injury, and attention-deficit/hyperactivity disorder are at risk for self-medicating and overdose.
A history of legal problems and motor vehicle accidents, or poor family support and unemployment also are predictors, Dr. Webster said.
Aberrant behaviors are directly related to the duration and the intensity of pain. Patients who cannot completely eliminate their pain may resort to taking more medication, which they will see as less of a wrong than the pain itself, he said.
Physicians generally have not done a good job at evaluating these risks, and selecting and managing patients according to these factors. But how patients are managed can help determine whether they end up in the emergency room, "or going to a substance abuse treatment center or the morgue," he said.
There are tools that can be used to screen patients for risk. Among those are the Screener and Opioid Assessment for Patients With Pain (SOAPP); the Opioid Risk Tool (ORT); the Diagnosis, Intractability, Risk Efficacy (DIRE) test; and the Current Opioid Misuse Measure (COMM), which is used for patients already taking opioids.
These tests all are simple and take only 1 to 10 minutes to administer. They can be given by any physician, including primary care physicians, he said.
Once all the factors are taken into account, patients can be stratified according to risk. Lower-risk patients – those with moderate pain and no personal or family history of substance use disorders – can be managed by primary care physicians. Those at moderate risk may need the addition of a pain specialist. The higher-risk patients will need consultations with mental health professionals and pain physicians.
Monitoring higher-risk patients may require urine drug testing and prescription monitoring programs. Pharmacists should be kept in the loop to see whether patients are shopping around. Family and friends can be invaluable sources to help identify risky behaviors.
Physicians need to do a better job of titrating opioids and determining proper dosing. Many patients have died at initiation of opioid therapy, or from rotating from one drug to another, he said. Patients should not be allowed to determine how much of a long-acting medication they can take.
So how do doctors mitigate risk? The first order of business is to treat the pain. "Uncontrolled pain, untreated pain, I believe is the number one reason for aberrant behavior," said Dr. Webster. Physicians cannot eliminate all pain, however, so education also is crucial.
Physicians also have a duty to monitor pain patients. "You don’t just put someone on an opioid and think you’ve done your job," said Dr. Webster. A doctor would not stop managing a diabetic; pain patients need long-term management, he said.
Dr. Webster reported having no relevant conflicts of interest.
NEW ORLEANS – Do you know which of your patients is likely to misuse or abuse opioid medications?
They can be stratified according to risk, but many physicians don’t know how or don’t take the time to do so, according to Dr. Lynn R. Webster, medical director of Lifetree Clinical Research in Salt Lake City and director at large for the American Academy of Pain Medicine.
It is important to try to prevent misuse or abuse, given that overdoses and deaths from prescription drugs are on the rise, said Dr. Webster at the American Medical Association House of Delegates Interim meeting.
According to the National Institute on Drug Abuse, emergency department visits involving nonmedical use of pharmaceuticals (either alone or in combination with another drug) increased 98% between 2004 and 2009, from 627,291 visits to 1.2 million visits. There was an 83% rise in emergency department visits involving adverse reactions to pharmaceuticals between 2005 and 2009, from 1.3 million to 2.3 million visits.
Opioids were the most frequently cited in those ED visits, accounting for about 50% of nonmedical use. Psychotherapeutic agents accounted for a third of the nonmedical use. According to the National Institute on Drug Abuse, the three most frequently cited drugs in those visits were hydrocodone (alone or in combination), accounting for 104,490 visits; oxycodone (alone or in combination), for 175,949 visits; and methadone, for 70,637 visits.
To properly assess and treat patients, physicians should understand and agree upon terminology, said Dr. Webster. Patients may "misuse" prescriptions if they are not taking them as directed. They aren’t seeking a high, they may just have undertreated pain, he said.
Abuse is the willful self-administration of a drug to get high. Addiction is a medical condition with complex genetic, psychosocial, and environmental factors that influence its development and manifestations. Addicts may have impaired control over drug use, compulsive use, or continued use despite harm.
"You don’t just put someone on an opioid and think you’ve done your job."
Pseudoaddiction results from undertreatment of pain, and may resolve with proper therapy. Patients in this state may manifest behavior that is misidentified as drug-seeking, according to Dr. Webster.
Even with chronic pain, "people prescribed opioids over a long period of time are going to have aberrant behavior," said Dr. Webster, noting that "it’s not a small problem." In research he conducted, up to 40% of patients in his chronic pain practice had aberrant behavior within a year of starting treatment.
But, he said, "only a small percent have the disease of addiction."
To prevent misuse and abuse, it’s important to understand patients’ motives, he said. Nonmedical users may have been prescribed pain medications, but they move on to recreational use and then, often, abuse.
Medical users generally are pain patients who are seeking more pain relief and end up misusing the drugs. They also may become abusers. In all, 40%-60% have other chronic conditions for which opioids provide relief, such as anxiety, said Dr. Webster. That use is obviously not appropriate, but it can only be prevented if physicians fully assess pain patients.
There is a constellation of risk factors that help determine potential for misuse and abuse, according to Dr. Webster.
The potency of a pharmacologic substance and how fast it works are important, as those factors help determine how much dopamine is released, and thus, the amount of reward the patient feels, said Dr. Webster. If the drug is readily available, that also determines its attractiveness.
One-fifth of the overall population is at risk for addiction. There is no on/off switch, but a continuum of risk, he explained. Biological, psychiatric, and social factors all play a role in where patients fall on that continuum. Age, gender, family history, cigarette smoking, pain severity and duration, and sleep disorders are all risk factors. Patients with a history of a psychiatric disorder, being sexual abused, or substance use disorder also are at higher risk. "We must assess mental health," said Dr. Webster, noting that patients with anxiety, depression, traumatic brain injury, and attention-deficit/hyperactivity disorder are at risk for self-medicating and overdose.
A history of legal problems and motor vehicle accidents, or poor family support and unemployment also are predictors, Dr. Webster said.
Aberrant behaviors are directly related to the duration and the intensity of pain. Patients who cannot completely eliminate their pain may resort to taking more medication, which they will see as less of a wrong than the pain itself, he said.
Physicians generally have not done a good job at evaluating these risks, and selecting and managing patients according to these factors. But how patients are managed can help determine whether they end up in the emergency room, "or going to a substance abuse treatment center or the morgue," he said.
There are tools that can be used to screen patients for risk. Among those are the Screener and Opioid Assessment for Patients With Pain (SOAPP); the Opioid Risk Tool (ORT); the Diagnosis, Intractability, Risk Efficacy (DIRE) test; and the Current Opioid Misuse Measure (COMM), which is used for patients already taking opioids.
These tests all are simple and take only 1 to 10 minutes to administer. They can be given by any physician, including primary care physicians, he said.
Once all the factors are taken into account, patients can be stratified according to risk. Lower-risk patients – those with moderate pain and no personal or family history of substance use disorders – can be managed by primary care physicians. Those at moderate risk may need the addition of a pain specialist. The higher-risk patients will need consultations with mental health professionals and pain physicians.
Monitoring higher-risk patients may require urine drug testing and prescription monitoring programs. Pharmacists should be kept in the loop to see whether patients are shopping around. Family and friends can be invaluable sources to help identify risky behaviors.
Physicians need to do a better job of titrating opioids and determining proper dosing. Many patients have died at initiation of opioid therapy, or from rotating from one drug to another, he said. Patients should not be allowed to determine how much of a long-acting medication they can take.
So how do doctors mitigate risk? The first order of business is to treat the pain. "Uncontrolled pain, untreated pain, I believe is the number one reason for aberrant behavior," said Dr. Webster. Physicians cannot eliminate all pain, however, so education also is crucial.
Physicians also have a duty to monitor pain patients. "You don’t just put someone on an opioid and think you’ve done your job," said Dr. Webster. A doctor would not stop managing a diabetic; pain patients need long-term management, he said.
Dr. Webster reported having no relevant conflicts of interest.
FROM THE AMERICAN MEDICAL ASSOCIATION HOUSE OF DELEGATES INTERIM MEETING