User login
Perinatal depression screening improves screening, treatment for postpartum depression
A policy of universal screening of perinatal depression for women receiving prenatal care at an academic medical center led to more regular screening of depression, and made it more likely that women with postpartum depression would be referred for treatment, according to recent research published in Obstetrics & Gynecology.
Emily S. Miller, MD, MPH, at Northwestern University, Chicago, and colleagues performed a retrospective study of 5,127 women receiving prenatal care at the center between 2008 and 2015. They divided the group into those who were at the center before (n = 1,122) and after (n = 4,005) initiation of a policy on universal perinatal depression screening, which consisted of two antenatal screenings at the first prenatal visit and third trimester, and one postpartum screening.
After initiation of the policy, screening increased during the first trimester (0.1% vs. 66%; P less than .001), the third trimester (0% vs. 43%; P less than .001), and at the postpartum visit (70% vs. 90%; P less than .001). Screening continued to increase at both prenatal visits, while screening prevalence remained the same for the postpartum visit. in the post-policy group (30% vs. 65%).
Katrina S. Mark, MD, associate professor of the department of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, said in an interview that the study “brings attention to an incredibly important topic.
“The researchers in this study found that, after implementation of a new policy regarding antenatal and postpartum depression screening, there was a significant increase in women who were screened during and after pregnancy as well as an increase in those who were appropriately treated,” she said. “Importantly, however, their intervention was not only a policy, but also provided education and resources to providers to increase awareness and knowledge surrounding the subject of depression and how to screen and treat this common condition.”
Dr. Miller and colleagues noted their study was limited because they were unable to determine whether prescriptions were filled or if referrals led to actual provider visits. Other obstacles to mental health care in the perinatal period also exist in the form of logistic barriers to appointments and stigma about mental health treatment.
“Depression is common, and screening and treatment during pregnancy and the postpartum period are extremely important to improve maternal and child health. As the authors point out, there has historically been a hesitation among obstetric providers to screen for depression,” Dr. Mark said. “My suspicion is that this hesitation is not because of a lack of awareness, but rather due to a lack of knowledge of what to do when a woman has a positive screen. In my opinion, the take-home message from this study is that implementation of a policy is possible and can lead to real change if it is accompanied by the appropriate resources and education.”
This study was funded by the Maternal-Fetal Medicine/Lumara Health Policy Award, and grants from the Eunice Kennedy Shriver National Institute of Child and Human Development and from the National Institutes of Health’s National Center for Advancing Translational Sciences. The authors reported no conflicts of interest.
SOURCE: Miller ES et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003369.
A policy of universal screening of perinatal depression for women receiving prenatal care at an academic medical center led to more regular screening of depression, and made it more likely that women with postpartum depression would be referred for treatment, according to recent research published in Obstetrics & Gynecology.
Emily S. Miller, MD, MPH, at Northwestern University, Chicago, and colleagues performed a retrospective study of 5,127 women receiving prenatal care at the center between 2008 and 2015. They divided the group into those who were at the center before (n = 1,122) and after (n = 4,005) initiation of a policy on universal perinatal depression screening, which consisted of two antenatal screenings at the first prenatal visit and third trimester, and one postpartum screening.
After initiation of the policy, screening increased during the first trimester (0.1% vs. 66%; P less than .001), the third trimester (0% vs. 43%; P less than .001), and at the postpartum visit (70% vs. 90%; P less than .001). Screening continued to increase at both prenatal visits, while screening prevalence remained the same for the postpartum visit. in the post-policy group (30% vs. 65%).
Katrina S. Mark, MD, associate professor of the department of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, said in an interview that the study “brings attention to an incredibly important topic.
“The researchers in this study found that, after implementation of a new policy regarding antenatal and postpartum depression screening, there was a significant increase in women who were screened during and after pregnancy as well as an increase in those who were appropriately treated,” she said. “Importantly, however, their intervention was not only a policy, but also provided education and resources to providers to increase awareness and knowledge surrounding the subject of depression and how to screen and treat this common condition.”
Dr. Miller and colleagues noted their study was limited because they were unable to determine whether prescriptions were filled or if referrals led to actual provider visits. Other obstacles to mental health care in the perinatal period also exist in the form of logistic barriers to appointments and stigma about mental health treatment.
“Depression is common, and screening and treatment during pregnancy and the postpartum period are extremely important to improve maternal and child health. As the authors point out, there has historically been a hesitation among obstetric providers to screen for depression,” Dr. Mark said. “My suspicion is that this hesitation is not because of a lack of awareness, but rather due to a lack of knowledge of what to do when a woman has a positive screen. In my opinion, the take-home message from this study is that implementation of a policy is possible and can lead to real change if it is accompanied by the appropriate resources and education.”
This study was funded by the Maternal-Fetal Medicine/Lumara Health Policy Award, and grants from the Eunice Kennedy Shriver National Institute of Child and Human Development and from the National Institutes of Health’s National Center for Advancing Translational Sciences. The authors reported no conflicts of interest.
SOURCE: Miller ES et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003369.
A policy of universal screening of perinatal depression for women receiving prenatal care at an academic medical center led to more regular screening of depression, and made it more likely that women with postpartum depression would be referred for treatment, according to recent research published in Obstetrics & Gynecology.
Emily S. Miller, MD, MPH, at Northwestern University, Chicago, and colleagues performed a retrospective study of 5,127 women receiving prenatal care at the center between 2008 and 2015. They divided the group into those who were at the center before (n = 1,122) and after (n = 4,005) initiation of a policy on universal perinatal depression screening, which consisted of two antenatal screenings at the first prenatal visit and third trimester, and one postpartum screening.
After initiation of the policy, screening increased during the first trimester (0.1% vs. 66%; P less than .001), the third trimester (0% vs. 43%; P less than .001), and at the postpartum visit (70% vs. 90%; P less than .001). Screening continued to increase at both prenatal visits, while screening prevalence remained the same for the postpartum visit. in the post-policy group (30% vs. 65%).
Katrina S. Mark, MD, associate professor of the department of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, said in an interview that the study “brings attention to an incredibly important topic.
“The researchers in this study found that, after implementation of a new policy regarding antenatal and postpartum depression screening, there was a significant increase in women who were screened during and after pregnancy as well as an increase in those who were appropriately treated,” she said. “Importantly, however, their intervention was not only a policy, but also provided education and resources to providers to increase awareness and knowledge surrounding the subject of depression and how to screen and treat this common condition.”
Dr. Miller and colleagues noted their study was limited because they were unable to determine whether prescriptions were filled or if referrals led to actual provider visits. Other obstacles to mental health care in the perinatal period also exist in the form of logistic barriers to appointments and stigma about mental health treatment.
“Depression is common, and screening and treatment during pregnancy and the postpartum period are extremely important to improve maternal and child health. As the authors point out, there has historically been a hesitation among obstetric providers to screen for depression,” Dr. Mark said. “My suspicion is that this hesitation is not because of a lack of awareness, but rather due to a lack of knowledge of what to do when a woman has a positive screen. In my opinion, the take-home message from this study is that implementation of a policy is possible and can lead to real change if it is accompanied by the appropriate resources and education.”
This study was funded by the Maternal-Fetal Medicine/Lumara Health Policy Award, and grants from the Eunice Kennedy Shriver National Institute of Child and Human Development and from the National Institutes of Health’s National Center for Advancing Translational Sciences. The authors reported no conflicts of interest.
SOURCE: Miller ES et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003369.
FROM OBSTETRICS & GYNECOLOGY
Key clinical point: A policy of universal perinatal screening improved adherence to screening and treatment for women with postpartum depression.
Major finding: After initiation of the policy, screening increased during the first prenatal visit (0.1% vs. 66%), the third trimester (0% vs. 43%), and at a postpartum visit (70% vs. 90%). Women who had a positive result after postpartum depression screening were more than twice as likely to receive treatment or a referral for their depression in the post-policy group (30% vs. 65%).
Study details: A retrospective cohort study of 5,127 women at a single academic center undergoing perinatal care before and after an institutional policy for perinatal depression screening between 2008 and 2015.
Disclosures: This study was funded by the Maternal-Fetal Medicine/Lumara Health Policy Award, and grants from the Eunice Kennedy Shriver National Institute of Child and Human Development and from the National Institutes of Health’s National Center for Advancing Translational Sciences. The authors reported no conflicts of interest.
Source: Miller ES et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003369.
Opioid exposure leads to poor perinatal and postnatal outcomes
according to data from more than 8,000 children.
Previous studies have shown the increased risk of a range of health problems associated with maternal opioid use, including neonatal abstinence syndrome (NAS), but data on the long-term consequences of in utero opioid exposure are limited, wrote Romuladus E. Azuine, DrPH, MPH, of the U.S. Department of Health and Human Services, Rockville, Md., and colleagues.
In a study published in JAMA Network Open, the researchers reviewed data from 8,509 mother/newborn pairs in the Boston Birth Cohort, a database that included a large urban, low-income, multiethnic population of women who had singleton births at the Boston Medical Center starting in 1998.
A total of 454 infants (5%) experienced prenatal opioid exposure. Mothers were interviewed 48-72 hours after delivery about sociodemographic factors, drug use, smoking, and alcohol use.
The risk of small for gestational age and preterm birth were significantly higher in babies exposed to opioids (OR 1.87 and OR 1.49, respectively), compared with unexposed newborns.
Children’s developmental outcomes were collected starting in 2003 based on electronic medical records. A total of 3,153 mother-newborn pairs were enrolled in a postnatal follow-up study. For preschoolers, prenatal opioid exposure was associated with increased risk of lack of expected physiological development and conduct disorder/emotional disturbance (OR 1.80 and OR 2.13, respectively), compared with unexposed children. School-aged children with prenatal opioid exposure had an increased risk of ADHD (OR 2.55).
The incidence of NAS in the study population was at least 24 per 1,000 hospital births starting in 2004, and peaked at 61 per 1,000 hospital births in 2008, but remained higher than 32 per 1,000 through 2016.
The study findings were limited by several factors including potential misclassification of opioid exposure, confounding from other pregnancy exposures, loss of many participants to follow-up, and a lack of generalizability, but the results support the need for additional research, and show that the prevalence of NAS was approximately 10 times the national average in a subset of low-income, urban, minority women, the researchers said.
“However, the effect of opioids is still difficult to disentangle from effects of other childhood exposures. Policy and programmatic efforts to prevent NAS and mitigate its health consequences require more comprehensive longitudinal and intergenerational research,” they concluded.
The study findings contribute to and support the evidence of poor neurodevelopmental and emotional/behavioral outcomes for children with prenatal exposure to opioids or a history of NAS, Susan Brogly, PhD, MSc, noted in an accompanying editorial. Other studies have shown increased risks for visual impairments including strabismus, reduced visual acuity, and delayed visual maturation.
Dr. Brogly, of Queen’s University, Kingston Health Science Center, Ontario, nonetheless noted that a child’s home environment may modify the impact of prenatal opioid exposure or NAS, as evidence has shown that children with in utero heroin exposure have improved outcomes in healthy home environments.
Although the mechanism for how opioid exposure affects development remains uncertain, she suggested that future research should address “interventions to improve health outcomes in this rapidly growing population of children, regardless of the causal mechanism of impairment.”
Dr. Brogly noted that most of the opioid-using mothers in the study by Azuine et al. were unmarried, non-Hispanic white, and multiparous, and had histories of other substance abuse. She emphasized the need for supportive communities for women at risk of opioid use, who also are more likely to have unstable housing situations and histories of sexual and physical abuse.
“The risks of poor pregnancy and child outcomes in cases of maternal opioid exposure are not because of prenatal opioid exposure alone; ongoing difficult social and environmental circumstances have an important role,” and future interventions should address these circumstances to improve long-term health of high-risk women and their children, she emphasized.
The Boston Birth Cohort study is supported in part by grants from the National Institutes of Health and the U.S. Department of Health and Human Services. None of the authors had financial conflicts to disclose.
Dr. Brogly disclosed grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development outside the submitted work.
SOURCE: Azuine RE et al. JAMA Network Open. 2019 Jun 28. doi: 10.1001/jamanetworkopen.2019.6405; Brogly S. JAMA Network Open. 2019 Jun 28. doi:10.1001/jamanetworkopen.2019.6428.
according to data from more than 8,000 children.
Previous studies have shown the increased risk of a range of health problems associated with maternal opioid use, including neonatal abstinence syndrome (NAS), but data on the long-term consequences of in utero opioid exposure are limited, wrote Romuladus E. Azuine, DrPH, MPH, of the U.S. Department of Health and Human Services, Rockville, Md., and colleagues.
In a study published in JAMA Network Open, the researchers reviewed data from 8,509 mother/newborn pairs in the Boston Birth Cohort, a database that included a large urban, low-income, multiethnic population of women who had singleton births at the Boston Medical Center starting in 1998.
A total of 454 infants (5%) experienced prenatal opioid exposure. Mothers were interviewed 48-72 hours after delivery about sociodemographic factors, drug use, smoking, and alcohol use.
The risk of small for gestational age and preterm birth were significantly higher in babies exposed to opioids (OR 1.87 and OR 1.49, respectively), compared with unexposed newborns.
Children’s developmental outcomes were collected starting in 2003 based on electronic medical records. A total of 3,153 mother-newborn pairs were enrolled in a postnatal follow-up study. For preschoolers, prenatal opioid exposure was associated with increased risk of lack of expected physiological development and conduct disorder/emotional disturbance (OR 1.80 and OR 2.13, respectively), compared with unexposed children. School-aged children with prenatal opioid exposure had an increased risk of ADHD (OR 2.55).
The incidence of NAS in the study population was at least 24 per 1,000 hospital births starting in 2004, and peaked at 61 per 1,000 hospital births in 2008, but remained higher than 32 per 1,000 through 2016.
The study findings were limited by several factors including potential misclassification of opioid exposure, confounding from other pregnancy exposures, loss of many participants to follow-up, and a lack of generalizability, but the results support the need for additional research, and show that the prevalence of NAS was approximately 10 times the national average in a subset of low-income, urban, minority women, the researchers said.
“However, the effect of opioids is still difficult to disentangle from effects of other childhood exposures. Policy and programmatic efforts to prevent NAS and mitigate its health consequences require more comprehensive longitudinal and intergenerational research,” they concluded.
The study findings contribute to and support the evidence of poor neurodevelopmental and emotional/behavioral outcomes for children with prenatal exposure to opioids or a history of NAS, Susan Brogly, PhD, MSc, noted in an accompanying editorial. Other studies have shown increased risks for visual impairments including strabismus, reduced visual acuity, and delayed visual maturation.
Dr. Brogly, of Queen’s University, Kingston Health Science Center, Ontario, nonetheless noted that a child’s home environment may modify the impact of prenatal opioid exposure or NAS, as evidence has shown that children with in utero heroin exposure have improved outcomes in healthy home environments.
Although the mechanism for how opioid exposure affects development remains uncertain, she suggested that future research should address “interventions to improve health outcomes in this rapidly growing population of children, regardless of the causal mechanism of impairment.”
Dr. Brogly noted that most of the opioid-using mothers in the study by Azuine et al. were unmarried, non-Hispanic white, and multiparous, and had histories of other substance abuse. She emphasized the need for supportive communities for women at risk of opioid use, who also are more likely to have unstable housing situations and histories of sexual and physical abuse.
“The risks of poor pregnancy and child outcomes in cases of maternal opioid exposure are not because of prenatal opioid exposure alone; ongoing difficult social and environmental circumstances have an important role,” and future interventions should address these circumstances to improve long-term health of high-risk women and their children, she emphasized.
The Boston Birth Cohort study is supported in part by grants from the National Institutes of Health and the U.S. Department of Health and Human Services. None of the authors had financial conflicts to disclose.
Dr. Brogly disclosed grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development outside the submitted work.
SOURCE: Azuine RE et al. JAMA Network Open. 2019 Jun 28. doi: 10.1001/jamanetworkopen.2019.6405; Brogly S. JAMA Network Open. 2019 Jun 28. doi:10.1001/jamanetworkopen.2019.6428.
according to data from more than 8,000 children.
Previous studies have shown the increased risk of a range of health problems associated with maternal opioid use, including neonatal abstinence syndrome (NAS), but data on the long-term consequences of in utero opioid exposure are limited, wrote Romuladus E. Azuine, DrPH, MPH, of the U.S. Department of Health and Human Services, Rockville, Md., and colleagues.
In a study published in JAMA Network Open, the researchers reviewed data from 8,509 mother/newborn pairs in the Boston Birth Cohort, a database that included a large urban, low-income, multiethnic population of women who had singleton births at the Boston Medical Center starting in 1998.
A total of 454 infants (5%) experienced prenatal opioid exposure. Mothers were interviewed 48-72 hours after delivery about sociodemographic factors, drug use, smoking, and alcohol use.
The risk of small for gestational age and preterm birth were significantly higher in babies exposed to opioids (OR 1.87 and OR 1.49, respectively), compared with unexposed newborns.
Children’s developmental outcomes were collected starting in 2003 based on electronic medical records. A total of 3,153 mother-newborn pairs were enrolled in a postnatal follow-up study. For preschoolers, prenatal opioid exposure was associated with increased risk of lack of expected physiological development and conduct disorder/emotional disturbance (OR 1.80 and OR 2.13, respectively), compared with unexposed children. School-aged children with prenatal opioid exposure had an increased risk of ADHD (OR 2.55).
The incidence of NAS in the study population was at least 24 per 1,000 hospital births starting in 2004, and peaked at 61 per 1,000 hospital births in 2008, but remained higher than 32 per 1,000 through 2016.
The study findings were limited by several factors including potential misclassification of opioid exposure, confounding from other pregnancy exposures, loss of many participants to follow-up, and a lack of generalizability, but the results support the need for additional research, and show that the prevalence of NAS was approximately 10 times the national average in a subset of low-income, urban, minority women, the researchers said.
“However, the effect of opioids is still difficult to disentangle from effects of other childhood exposures. Policy and programmatic efforts to prevent NAS and mitigate its health consequences require more comprehensive longitudinal and intergenerational research,” they concluded.
The study findings contribute to and support the evidence of poor neurodevelopmental and emotional/behavioral outcomes for children with prenatal exposure to opioids or a history of NAS, Susan Brogly, PhD, MSc, noted in an accompanying editorial. Other studies have shown increased risks for visual impairments including strabismus, reduced visual acuity, and delayed visual maturation.
Dr. Brogly, of Queen’s University, Kingston Health Science Center, Ontario, nonetheless noted that a child’s home environment may modify the impact of prenatal opioid exposure or NAS, as evidence has shown that children with in utero heroin exposure have improved outcomes in healthy home environments.
Although the mechanism for how opioid exposure affects development remains uncertain, she suggested that future research should address “interventions to improve health outcomes in this rapidly growing population of children, regardless of the causal mechanism of impairment.”
Dr. Brogly noted that most of the opioid-using mothers in the study by Azuine et al. were unmarried, non-Hispanic white, and multiparous, and had histories of other substance abuse. She emphasized the need for supportive communities for women at risk of opioid use, who also are more likely to have unstable housing situations and histories of sexual and physical abuse.
“The risks of poor pregnancy and child outcomes in cases of maternal opioid exposure are not because of prenatal opioid exposure alone; ongoing difficult social and environmental circumstances have an important role,” and future interventions should address these circumstances to improve long-term health of high-risk women and their children, she emphasized.
The Boston Birth Cohort study is supported in part by grants from the National Institutes of Health and the U.S. Department of Health and Human Services. None of the authors had financial conflicts to disclose.
Dr. Brogly disclosed grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development outside the submitted work.
SOURCE: Azuine RE et al. JAMA Network Open. 2019 Jun 28. doi: 10.1001/jamanetworkopen.2019.6405; Brogly S. JAMA Network Open. 2019 Jun 28. doi:10.1001/jamanetworkopen.2019.6428.
FROM JAMA NETWORK OPEN
Diverse vaginal microbiome may signal risk for preterm birth
in an analysis of approximately 12,000 samples, according to a study published in Nature Medicine.
Preterm births, defined as less than 37 weeks’ gestation, remain the second most common cause of neonatal death worldwide, but few strategies exist to prevent and predict preterm birth (PTB) wrote Jennifer M. Fettweis, MD, of Virginia Commonwealth University, Richmond, and her colleagues. In the United States, women of African ancestry are at significantly greater risk for PTB.
A highly diverse vaginal microbiome is thought to be associated with an increased risk of inflammation, infection, and PTB, “however, many asymptomatic healthy women have diverse vaginal microbiota,” the researchers said.
To identify vaginal microbiota distinct to women who experienced PTB, the researchers analyzed data from the Multi-Omic Microbiome Study: Pregnancy Initiative (MOMS-PI), part of the National Institutes of Health–sponsored Integrative Human Microbiome Project. The MOMS-PI study included 12,039 samples of vaginal flora from 597 pregnancies; the analysis included 45 singleton pregnancies that met the criteria for spontaneous PTB (23-36 weeks, 6 days of gestation) and 90 case-matched full-term singleton pregnancies (greater than or equal to 39 weeks). Approximately 78% of the women were of African descent in both groups, and their average age was 26 years in both groups.
Overall, the diversity of the vaginal microbiome was greater among women who experienced PTB, compared with term birth (TB). Women who experienced PTB had less Lactobacillus crispatus, but more bacterial vaginosis–associated bacterium-1 (BVAB1), Prevotella cluster 2, and Sneathia amnii, compared with TB women.
Of note, vaginal cytokine data showed that proinflammatory cytokines, which may be associated with the induction of labor, may be prompted by inflammation in the vaginal microbiome, Dr. Fettweis and her associates said. “We observed that vaginal IP-10/CXCL10 levels were inversely correlated with BVAB1 in PTB, inversely correlated with L. crispatus in TB, and positively correlated with L. iners in TB, suggesting complex host-microbiome interactions in pregnancy,” they said.
“Further studies are needed to determine whether the signatures of PTB reported in the present study replicate in other cohorts of women of African ancestry, to examine whether the observed differences in vaginal microbiome composition between women of different ancestries has a direct causal link to the ethnic and racial disparities in PTB rates, and to establish whether population-specific microbial markers can be ultimately integrated into a generalizable spectrum of vaginal microbiome states linked to the risk for PTB,” Dr. Fettweis and her associates said.
In a companion study also published in Nature Medicine, Myrna G. Serrano, MD, also of Virginia Commonwealth University, and her colleagues as part of the MOMS-PI initially determined that vaginal microbiome profiles varied between 613 pregnant and 1,969 nonpregnant women in that “pregnant women had significantly higher prevalence of the four most common Lactobacillus vagitypes (L. crispatus, L. iners, L. gasseri, and L. jensenii) and a commensurately lower prevalence of vagitypes dominated by other taxa.” The primary driver of the differences was L. iners.
They then compared vaginal microbiome data from 300 pregnant and 300 nonpregnant case-matched women of African, Hispanic, or European ancestry, as well as 90 pregnant women (49 of African ancestry and 41 of European) ancestry.
In the subset of 300 pregnant and 300 nonpregnant women, the vaginal microbiome of the pregnant women overall became more dominated by Lactobacillus early in pregnancy. Further stratification by race showed that pregnant women of African and Hispanic ancestry had significantly higher levels of four types of Lactobacillus than their nonpregnant counterparts, but no significant difference was seen between pregnant and nonpregnant women of European ancestry.
“It appears that changes occurring during pregnancy may render the reproductive tracts of women of all racial backgrounds more hospitable to taxa of Lactobacillus and less favorable for Gardnerella vaginalis and other taxa associated with BV [bacterial vaginosis] and dysbiosis,” the researchers said.
“Interestingly, BVAB1, which has been associated with dysbiotic vaginal conditions and risk of PTB, and which is present as a major vagitype largely in women of African ancestry, is not noticeably decreased in prevalence in pregnancy,” Dr. Serrano and her associates said. “Thus, BVAB1, for reasons yet to be determined, is apparently resistant to factors sculpting the microbiome in pregnant women, possibly explaining in part the enhanced risk for PTB experienced by women of African ancestry.”
In a look at the 49 pregnant women of African ancestry and 41 of European ancestry, those of African ancestry had “significantly lower representation of the L. crispatus, L. gasseri and L. jensenii vagitypes, and higher representation of L. iners and BVAB1 vagitypes. Variability in women of African ancestry was driven by BVAB1 and L. iners, whereas variability in women of non-African ancestry was driven by L. crispatus and L. iners. Again, pregnancy had no significant effect on prevalence of the BVAB1 vagitype. Prevalence of Lactobacillus-dominated profiles in women of African ancestry was lower in the first than in later trimesters, whereas women of European ancestry had a higher prevalence of Lactobacillus vagitypes throughout pregnancy.”
The presence of vaginal microbiome profiles associated with adverse pregnancy outcomes highlights the need for further studies that take advantage of this information, Dr. Serrano and her associates said. “That the vaginal microbiomes known to confer higher risk of poor health and adverse outcomes of pregnancy are more highly associated with women of African and Hispanic ancestry, but that pregnancy tends to drive these microbiomes toward more favorable microbiota, suggests that an external intervention that favors this trend might be beneficial for these populations,” they concluded. “What remains is to verify the most favorable microbiome and the most effective strategy for intervention.”
Dr. Fettweis had no financial conflicts to disclose; two coauthors are full-time employees at Pacific Biosciences. Dr. Serrano and her coauthors had no relevant financial disclosures. Dr. Serrano’s study received grants from the National Institutes of Health and other sources, as well as support from the Common Fund, the National Center for Complementary and Integrative Health, the Office of Research on Women’s Health, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, and the National Institute of Allergy and Infectious Diseases.
SOURCES: Fettweis J et al. Nature Medicine 2019 May 29. doi: 10.1038/s41591-019-0450-2; Serrano M et al. Nature Medicine. 2019 May 29. doi: 10.1038/s41591-019-0465-8.
in an analysis of approximately 12,000 samples, according to a study published in Nature Medicine.
Preterm births, defined as less than 37 weeks’ gestation, remain the second most common cause of neonatal death worldwide, but few strategies exist to prevent and predict preterm birth (PTB) wrote Jennifer M. Fettweis, MD, of Virginia Commonwealth University, Richmond, and her colleagues. In the United States, women of African ancestry are at significantly greater risk for PTB.
A highly diverse vaginal microbiome is thought to be associated with an increased risk of inflammation, infection, and PTB, “however, many asymptomatic healthy women have diverse vaginal microbiota,” the researchers said.
To identify vaginal microbiota distinct to women who experienced PTB, the researchers analyzed data from the Multi-Omic Microbiome Study: Pregnancy Initiative (MOMS-PI), part of the National Institutes of Health–sponsored Integrative Human Microbiome Project. The MOMS-PI study included 12,039 samples of vaginal flora from 597 pregnancies; the analysis included 45 singleton pregnancies that met the criteria for spontaneous PTB (23-36 weeks, 6 days of gestation) and 90 case-matched full-term singleton pregnancies (greater than or equal to 39 weeks). Approximately 78% of the women were of African descent in both groups, and their average age was 26 years in both groups.
Overall, the diversity of the vaginal microbiome was greater among women who experienced PTB, compared with term birth (TB). Women who experienced PTB had less Lactobacillus crispatus, but more bacterial vaginosis–associated bacterium-1 (BVAB1), Prevotella cluster 2, and Sneathia amnii, compared with TB women.
Of note, vaginal cytokine data showed that proinflammatory cytokines, which may be associated with the induction of labor, may be prompted by inflammation in the vaginal microbiome, Dr. Fettweis and her associates said. “We observed that vaginal IP-10/CXCL10 levels were inversely correlated with BVAB1 in PTB, inversely correlated with L. crispatus in TB, and positively correlated with L. iners in TB, suggesting complex host-microbiome interactions in pregnancy,” they said.
“Further studies are needed to determine whether the signatures of PTB reported in the present study replicate in other cohorts of women of African ancestry, to examine whether the observed differences in vaginal microbiome composition between women of different ancestries has a direct causal link to the ethnic and racial disparities in PTB rates, and to establish whether population-specific microbial markers can be ultimately integrated into a generalizable spectrum of vaginal microbiome states linked to the risk for PTB,” Dr. Fettweis and her associates said.
In a companion study also published in Nature Medicine, Myrna G. Serrano, MD, also of Virginia Commonwealth University, and her colleagues as part of the MOMS-PI initially determined that vaginal microbiome profiles varied between 613 pregnant and 1,969 nonpregnant women in that “pregnant women had significantly higher prevalence of the four most common Lactobacillus vagitypes (L. crispatus, L. iners, L. gasseri, and L. jensenii) and a commensurately lower prevalence of vagitypes dominated by other taxa.” The primary driver of the differences was L. iners.
They then compared vaginal microbiome data from 300 pregnant and 300 nonpregnant case-matched women of African, Hispanic, or European ancestry, as well as 90 pregnant women (49 of African ancestry and 41 of European) ancestry.
In the subset of 300 pregnant and 300 nonpregnant women, the vaginal microbiome of the pregnant women overall became more dominated by Lactobacillus early in pregnancy. Further stratification by race showed that pregnant women of African and Hispanic ancestry had significantly higher levels of four types of Lactobacillus than their nonpregnant counterparts, but no significant difference was seen between pregnant and nonpregnant women of European ancestry.
“It appears that changes occurring during pregnancy may render the reproductive tracts of women of all racial backgrounds more hospitable to taxa of Lactobacillus and less favorable for Gardnerella vaginalis and other taxa associated with BV [bacterial vaginosis] and dysbiosis,” the researchers said.
“Interestingly, BVAB1, which has been associated with dysbiotic vaginal conditions and risk of PTB, and which is present as a major vagitype largely in women of African ancestry, is not noticeably decreased in prevalence in pregnancy,” Dr. Serrano and her associates said. “Thus, BVAB1, for reasons yet to be determined, is apparently resistant to factors sculpting the microbiome in pregnant women, possibly explaining in part the enhanced risk for PTB experienced by women of African ancestry.”
In a look at the 49 pregnant women of African ancestry and 41 of European ancestry, those of African ancestry had “significantly lower representation of the L. crispatus, L. gasseri and L. jensenii vagitypes, and higher representation of L. iners and BVAB1 vagitypes. Variability in women of African ancestry was driven by BVAB1 and L. iners, whereas variability in women of non-African ancestry was driven by L. crispatus and L. iners. Again, pregnancy had no significant effect on prevalence of the BVAB1 vagitype. Prevalence of Lactobacillus-dominated profiles in women of African ancestry was lower in the first than in later trimesters, whereas women of European ancestry had a higher prevalence of Lactobacillus vagitypes throughout pregnancy.”
The presence of vaginal microbiome profiles associated with adverse pregnancy outcomes highlights the need for further studies that take advantage of this information, Dr. Serrano and her associates said. “That the vaginal microbiomes known to confer higher risk of poor health and adverse outcomes of pregnancy are more highly associated with women of African and Hispanic ancestry, but that pregnancy tends to drive these microbiomes toward more favorable microbiota, suggests that an external intervention that favors this trend might be beneficial for these populations,” they concluded. “What remains is to verify the most favorable microbiome and the most effective strategy for intervention.”
Dr. Fettweis had no financial conflicts to disclose; two coauthors are full-time employees at Pacific Biosciences. Dr. Serrano and her coauthors had no relevant financial disclosures. Dr. Serrano’s study received grants from the National Institutes of Health and other sources, as well as support from the Common Fund, the National Center for Complementary and Integrative Health, the Office of Research on Women’s Health, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, and the National Institute of Allergy and Infectious Diseases.
SOURCES: Fettweis J et al. Nature Medicine 2019 May 29. doi: 10.1038/s41591-019-0450-2; Serrano M et al. Nature Medicine. 2019 May 29. doi: 10.1038/s41591-019-0465-8.
in an analysis of approximately 12,000 samples, according to a study published in Nature Medicine.
Preterm births, defined as less than 37 weeks’ gestation, remain the second most common cause of neonatal death worldwide, but few strategies exist to prevent and predict preterm birth (PTB) wrote Jennifer M. Fettweis, MD, of Virginia Commonwealth University, Richmond, and her colleagues. In the United States, women of African ancestry are at significantly greater risk for PTB.
A highly diverse vaginal microbiome is thought to be associated with an increased risk of inflammation, infection, and PTB, “however, many asymptomatic healthy women have diverse vaginal microbiota,” the researchers said.
To identify vaginal microbiota distinct to women who experienced PTB, the researchers analyzed data from the Multi-Omic Microbiome Study: Pregnancy Initiative (MOMS-PI), part of the National Institutes of Health–sponsored Integrative Human Microbiome Project. The MOMS-PI study included 12,039 samples of vaginal flora from 597 pregnancies; the analysis included 45 singleton pregnancies that met the criteria for spontaneous PTB (23-36 weeks, 6 days of gestation) and 90 case-matched full-term singleton pregnancies (greater than or equal to 39 weeks). Approximately 78% of the women were of African descent in both groups, and their average age was 26 years in both groups.
Overall, the diversity of the vaginal microbiome was greater among women who experienced PTB, compared with term birth (TB). Women who experienced PTB had less Lactobacillus crispatus, but more bacterial vaginosis–associated bacterium-1 (BVAB1), Prevotella cluster 2, and Sneathia amnii, compared with TB women.
Of note, vaginal cytokine data showed that proinflammatory cytokines, which may be associated with the induction of labor, may be prompted by inflammation in the vaginal microbiome, Dr. Fettweis and her associates said. “We observed that vaginal IP-10/CXCL10 levels were inversely correlated with BVAB1 in PTB, inversely correlated with L. crispatus in TB, and positively correlated with L. iners in TB, suggesting complex host-microbiome interactions in pregnancy,” they said.
“Further studies are needed to determine whether the signatures of PTB reported in the present study replicate in other cohorts of women of African ancestry, to examine whether the observed differences in vaginal microbiome composition between women of different ancestries has a direct causal link to the ethnic and racial disparities in PTB rates, and to establish whether population-specific microbial markers can be ultimately integrated into a generalizable spectrum of vaginal microbiome states linked to the risk for PTB,” Dr. Fettweis and her associates said.
In a companion study also published in Nature Medicine, Myrna G. Serrano, MD, also of Virginia Commonwealth University, and her colleagues as part of the MOMS-PI initially determined that vaginal microbiome profiles varied between 613 pregnant and 1,969 nonpregnant women in that “pregnant women had significantly higher prevalence of the four most common Lactobacillus vagitypes (L. crispatus, L. iners, L. gasseri, and L. jensenii) and a commensurately lower prevalence of vagitypes dominated by other taxa.” The primary driver of the differences was L. iners.
They then compared vaginal microbiome data from 300 pregnant and 300 nonpregnant case-matched women of African, Hispanic, or European ancestry, as well as 90 pregnant women (49 of African ancestry and 41 of European) ancestry.
In the subset of 300 pregnant and 300 nonpregnant women, the vaginal microbiome of the pregnant women overall became more dominated by Lactobacillus early in pregnancy. Further stratification by race showed that pregnant women of African and Hispanic ancestry had significantly higher levels of four types of Lactobacillus than their nonpregnant counterparts, but no significant difference was seen between pregnant and nonpregnant women of European ancestry.
“It appears that changes occurring during pregnancy may render the reproductive tracts of women of all racial backgrounds more hospitable to taxa of Lactobacillus and less favorable for Gardnerella vaginalis and other taxa associated with BV [bacterial vaginosis] and dysbiosis,” the researchers said.
“Interestingly, BVAB1, which has been associated with dysbiotic vaginal conditions and risk of PTB, and which is present as a major vagitype largely in women of African ancestry, is not noticeably decreased in prevalence in pregnancy,” Dr. Serrano and her associates said. “Thus, BVAB1, for reasons yet to be determined, is apparently resistant to factors sculpting the microbiome in pregnant women, possibly explaining in part the enhanced risk for PTB experienced by women of African ancestry.”
In a look at the 49 pregnant women of African ancestry and 41 of European ancestry, those of African ancestry had “significantly lower representation of the L. crispatus, L. gasseri and L. jensenii vagitypes, and higher representation of L. iners and BVAB1 vagitypes. Variability in women of African ancestry was driven by BVAB1 and L. iners, whereas variability in women of non-African ancestry was driven by L. crispatus and L. iners. Again, pregnancy had no significant effect on prevalence of the BVAB1 vagitype. Prevalence of Lactobacillus-dominated profiles in women of African ancestry was lower in the first than in later trimesters, whereas women of European ancestry had a higher prevalence of Lactobacillus vagitypes throughout pregnancy.”
The presence of vaginal microbiome profiles associated with adverse pregnancy outcomes highlights the need for further studies that take advantage of this information, Dr. Serrano and her associates said. “That the vaginal microbiomes known to confer higher risk of poor health and adverse outcomes of pregnancy are more highly associated with women of African and Hispanic ancestry, but that pregnancy tends to drive these microbiomes toward more favorable microbiota, suggests that an external intervention that favors this trend might be beneficial for these populations,” they concluded. “What remains is to verify the most favorable microbiome and the most effective strategy for intervention.”
Dr. Fettweis had no financial conflicts to disclose; two coauthors are full-time employees at Pacific Biosciences. Dr. Serrano and her coauthors had no relevant financial disclosures. Dr. Serrano’s study received grants from the National Institutes of Health and other sources, as well as support from the Common Fund, the National Center for Complementary and Integrative Health, the Office of Research on Women’s Health, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, and the National Institute of Allergy and Infectious Diseases.
SOURCES: Fettweis J et al. Nature Medicine 2019 May 29. doi: 10.1038/s41591-019-0450-2; Serrano M et al. Nature Medicine. 2019 May 29. doi: 10.1038/s41591-019-0465-8.
FROM NATURE MEDICINE
Some Brits snuff out TORCH screen to raise awareness of congenital syphilis
LJUBLJANA, SLOVENIA – Pediatricians in the south of England are so concerned about the recent national increase in the diagnosis of syphilis in adults and its ramifications for neonates that they’ve ditched the traditional TORCH newborn screen because the acronym doesn’t specifically remind clinicians to think about congenital syphilis, Mildred A. Iro, MD, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“ explained Dr. Iro of the University of Southampton (England).
She highlighted salient features of three recent cases of congenital syphilis managed at Southampton Children’s Hospital.
“The key message that we’d like to share is that we just need to be more aware about congenital syphilis. Retest mothers if their risk factor status changes, and test suspected infants and children,” Dr. Iro said.
As a practical matter, however, even though current guidelines recommend retesting mothers whose risk factor status becomes heightened following an initial negative syphilis serology result early in pregnancy, clinicians often are unaware that a mother’s risk status has changed. And retesting all mothers during pregnancy isn’t attractive from a cost-benefit standpoint. This makes scrupulous screening of newborns all the more important. And yet TORCH, which stands for Toxoplasmosis, Other, Rubella, Cytomegalovirus, and Herpes infections, isn’t an acronym that promotes awareness of congenital syphilis, a disease which occupies an obscure position in TORCH under the “O” for “Other” heading. That’s why the term “congenital infection screen” has become the new norm in the south of England, she explained.
However, one pediatrician who didn’t consider congenital infection screen to be an improvement in terminology over TORCH had an alternative suggestion, which struck a favorable chord with his fellow audience members: Simply change the acronym to TORCHS, with the S standing for syphilis.
Dr. Iro noted that two of the three affected children were diagnosed at age 7-8 weeks. The third wasn’t diagnosed until age 15 months, when the mother tested positive for syphilis in a subsequent pregnancy. As is typical of the disease known as “the great masquerader,” while all three of the affected children were unwell early in infancy, they presented with a wide range of symptoms. Among the more prominent features were prolonged irritability, respiratory distress, odd rashes, anemia, hepatomegaly, and tachypnea. One infant had reduced movement and pain in one arm.
All three children underwent extensive testing. None had neurosyphilis. All achieved good outcomes on standard guideline-directed therapy.
As for the mothers, they were aged 19, 21, and 23 years when diagnosed with syphilis. All were Caucasian, and antenatal blood testing was negative in all three. None were retested during pregnancy, even though two of them had a male partner or former partner who was positive for syphilis, and the partner of the third disclosed to her that he had sex with men.
At diagnosis, all three women had a strongly positive Treponema pallidum particle agglutination assay, a high rapid plasma reagin, and a positive syphilis IgM assay.
Dr. Iro reported having no financial conflicts regarding her presentation.
LJUBLJANA, SLOVENIA – Pediatricians in the south of England are so concerned about the recent national increase in the diagnosis of syphilis in adults and its ramifications for neonates that they’ve ditched the traditional TORCH newborn screen because the acronym doesn’t specifically remind clinicians to think about congenital syphilis, Mildred A. Iro, MD, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“ explained Dr. Iro of the University of Southampton (England).
She highlighted salient features of three recent cases of congenital syphilis managed at Southampton Children’s Hospital.
“The key message that we’d like to share is that we just need to be more aware about congenital syphilis. Retest mothers if their risk factor status changes, and test suspected infants and children,” Dr. Iro said.
As a practical matter, however, even though current guidelines recommend retesting mothers whose risk factor status becomes heightened following an initial negative syphilis serology result early in pregnancy, clinicians often are unaware that a mother’s risk status has changed. And retesting all mothers during pregnancy isn’t attractive from a cost-benefit standpoint. This makes scrupulous screening of newborns all the more important. And yet TORCH, which stands for Toxoplasmosis, Other, Rubella, Cytomegalovirus, and Herpes infections, isn’t an acronym that promotes awareness of congenital syphilis, a disease which occupies an obscure position in TORCH under the “O” for “Other” heading. That’s why the term “congenital infection screen” has become the new norm in the south of England, she explained.
However, one pediatrician who didn’t consider congenital infection screen to be an improvement in terminology over TORCH had an alternative suggestion, which struck a favorable chord with his fellow audience members: Simply change the acronym to TORCHS, with the S standing for syphilis.
Dr. Iro noted that two of the three affected children were diagnosed at age 7-8 weeks. The third wasn’t diagnosed until age 15 months, when the mother tested positive for syphilis in a subsequent pregnancy. As is typical of the disease known as “the great masquerader,” while all three of the affected children were unwell early in infancy, they presented with a wide range of symptoms. Among the more prominent features were prolonged irritability, respiratory distress, odd rashes, anemia, hepatomegaly, and tachypnea. One infant had reduced movement and pain in one arm.
All three children underwent extensive testing. None had neurosyphilis. All achieved good outcomes on standard guideline-directed therapy.
As for the mothers, they were aged 19, 21, and 23 years when diagnosed with syphilis. All were Caucasian, and antenatal blood testing was negative in all three. None were retested during pregnancy, even though two of them had a male partner or former partner who was positive for syphilis, and the partner of the third disclosed to her that he had sex with men.
At diagnosis, all three women had a strongly positive Treponema pallidum particle agglutination assay, a high rapid plasma reagin, and a positive syphilis IgM assay.
Dr. Iro reported having no financial conflicts regarding her presentation.
LJUBLJANA, SLOVENIA – Pediatricians in the south of England are so concerned about the recent national increase in the diagnosis of syphilis in adults and its ramifications for neonates that they’ve ditched the traditional TORCH newborn screen because the acronym doesn’t specifically remind clinicians to think about congenital syphilis, Mildred A. Iro, MD, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“ explained Dr. Iro of the University of Southampton (England).
She highlighted salient features of three recent cases of congenital syphilis managed at Southampton Children’s Hospital.
“The key message that we’d like to share is that we just need to be more aware about congenital syphilis. Retest mothers if their risk factor status changes, and test suspected infants and children,” Dr. Iro said.
As a practical matter, however, even though current guidelines recommend retesting mothers whose risk factor status becomes heightened following an initial negative syphilis serology result early in pregnancy, clinicians often are unaware that a mother’s risk status has changed. And retesting all mothers during pregnancy isn’t attractive from a cost-benefit standpoint. This makes scrupulous screening of newborns all the more important. And yet TORCH, which stands for Toxoplasmosis, Other, Rubella, Cytomegalovirus, and Herpes infections, isn’t an acronym that promotes awareness of congenital syphilis, a disease which occupies an obscure position in TORCH under the “O” for “Other” heading. That’s why the term “congenital infection screen” has become the new norm in the south of England, she explained.
However, one pediatrician who didn’t consider congenital infection screen to be an improvement in terminology over TORCH had an alternative suggestion, which struck a favorable chord with his fellow audience members: Simply change the acronym to TORCHS, with the S standing for syphilis.
Dr. Iro noted that two of the three affected children were diagnosed at age 7-8 weeks. The third wasn’t diagnosed until age 15 months, when the mother tested positive for syphilis in a subsequent pregnancy. As is typical of the disease known as “the great masquerader,” while all three of the affected children were unwell early in infancy, they presented with a wide range of symptoms. Among the more prominent features were prolonged irritability, respiratory distress, odd rashes, anemia, hepatomegaly, and tachypnea. One infant had reduced movement and pain in one arm.
All three children underwent extensive testing. None had neurosyphilis. All achieved good outcomes on standard guideline-directed therapy.
As for the mothers, they were aged 19, 21, and 23 years when diagnosed with syphilis. All were Caucasian, and antenatal blood testing was negative in all three. None were retested during pregnancy, even though two of them had a male partner or former partner who was positive for syphilis, and the partner of the third disclosed to her that he had sex with men.
At diagnosis, all three women had a strongly positive Treponema pallidum particle agglutination assay, a high rapid plasma reagin, and a positive syphilis IgM assay.
Dr. Iro reported having no financial conflicts regarding her presentation.
EXPERT ANALYSIS FROM ESPID 2019
Minimize iatrogenic neonatal abstinence syndrome
BALTIMORE – Some infants, especially among those with persistent pulmonary hypertension, are at risk for developing iatrogenic neonatal abstinence syndrome, according to Amber Dave, MD, a neonatal-perinatal medicine fellow at Georgetown University Hospital in Washington.
Of 70 infants administered morphine or fentanyl for longer than a day in the neonatal ICU, almost a third (22, or 31%) developed iatrogenic neonatal abstinence syndrome (INAS). As a result, they needed prolonged respiratory support, more time to reach full feeds, and extended lengths of stay. Children exposed to opioids before birth were excluded from the analysis.
The greatest risk was in infants with persistent pulmonary hypertension; INAS was diagnosed in 13 of 22 (57%).
Opioid dosing also was all over the map for a given Neonatal Pain, Agitation, and Sedation Scale (N-PASS) score, Dr. Dave said. Some infants with an N-PASS pain score of 2, for instance, received no opioids, while others received up to 1,500 mg/kg morphine equivalents.
N-PASS is used in NICUs nationwide to guide dosing, but the variability seen in the study suggests that there’s need for a more objective measure of neonatal distress and for neonatologists to establish ground rules for NICU opioid use, she added.
The use of opioids has been increasing in NICUs for years (J Opioid Manag. 2015 Jul-Aug;11[4]:305-12), and at least one institution (J Perinatol. 2017 Sep;37[9]:1038-42) already has established guidelines to curb overuse. Dr. Dave said that several neonatologists, after viewing her poster at the Pediatric Academic Societies annual meeting, told her that they probably had the same problem at their NICUs but had not examined their data.
“We are using” these medications more in the NICU, “but how much is too much? We need to find that balance. We need to improve our practice.”
“The overarching question is if there are better alternatives for treating pain and stress in critically ill neonates.” Dexmedetomidine, an opioid-sparing alpha-2 agonist adrenoreceptor sedative, analgesic, and anxiolytic, is one of several options “being looked at closely in this population. We also need to think of nonpharmacologic measures,” Dr. Dave said.
In addition to infants with persistent pulmonary hypertension, the 22 INAS cases at the study site included, among others, three children on extracorporeal membrane oxygenation, one with meconium aspiration syndrome, and one surgical case, out of the 15 included in the study. The common denominator was the need to keep infants calm and comfortable during prolonged intubation, which was a mean of 10.5 days among INAS infants versus 5 among children who didn’t go into opioid withdrawal.
INAS infants had a daily mean morphine-equivalent dose of 106.6 mg/kg, with a mean exposure of 17 days and mean cumulative dose of 1,515 mg/kg. The daily mean morphine-equivalent dose among infants who didn’t develop INAS was 42.4 mg/kg, with a mean exposure of 4 days and mean cumulative dose of 246 mg/kg.
INAS infants spent a mean of 27 days in the hospital, and it took them a mean of almost 6 days to reach full feeds, versus 15 days for the other infants full feeds by day 4. Over half of the INAS infants (12) also were on midazolam, and they had higher cumulative doses of the sedative than infants who didn’t develop INAS (mean, 2.64 mg/kg vs. 0.19 mg/kg). The findings all were statistically significant.
Dr. Dave said the most surprising finding was the variability in opioid dosing. In another example, some infants received up to 1,400 mg/kg morphine equivalents even when their fraction of inspired oxygen requirement fell below 60%, which meant that they were getting better. Other infants by that point were off opioids altogether.
“This has definitely brought awareness to my practice. Before I would say, ‘Okay, let’s just go up,’ ” when a nurse requested an opioid increase based on N-PASS scores. Now, “I try to really figure out why they think the baby needs an increase, and I may say ‘Actually, we are turning a corner now, and maybe the baby can be a little bit more awake. How do you feel about that?’ ” she said.
“My long-term goal for this project is putting some guidelines in place,” she said.
There was no industry funding for the work, and Dr. Dave didn’t have any disclosures.
BALTIMORE – Some infants, especially among those with persistent pulmonary hypertension, are at risk for developing iatrogenic neonatal abstinence syndrome, according to Amber Dave, MD, a neonatal-perinatal medicine fellow at Georgetown University Hospital in Washington.
Of 70 infants administered morphine or fentanyl for longer than a day in the neonatal ICU, almost a third (22, or 31%) developed iatrogenic neonatal abstinence syndrome (INAS). As a result, they needed prolonged respiratory support, more time to reach full feeds, and extended lengths of stay. Children exposed to opioids before birth were excluded from the analysis.
The greatest risk was in infants with persistent pulmonary hypertension; INAS was diagnosed in 13 of 22 (57%).
Opioid dosing also was all over the map for a given Neonatal Pain, Agitation, and Sedation Scale (N-PASS) score, Dr. Dave said. Some infants with an N-PASS pain score of 2, for instance, received no opioids, while others received up to 1,500 mg/kg morphine equivalents.
N-PASS is used in NICUs nationwide to guide dosing, but the variability seen in the study suggests that there’s need for a more objective measure of neonatal distress and for neonatologists to establish ground rules for NICU opioid use, she added.
The use of opioids has been increasing in NICUs for years (J Opioid Manag. 2015 Jul-Aug;11[4]:305-12), and at least one institution (J Perinatol. 2017 Sep;37[9]:1038-42) already has established guidelines to curb overuse. Dr. Dave said that several neonatologists, after viewing her poster at the Pediatric Academic Societies annual meeting, told her that they probably had the same problem at their NICUs but had not examined their data.
“We are using” these medications more in the NICU, “but how much is too much? We need to find that balance. We need to improve our practice.”
“The overarching question is if there are better alternatives for treating pain and stress in critically ill neonates.” Dexmedetomidine, an opioid-sparing alpha-2 agonist adrenoreceptor sedative, analgesic, and anxiolytic, is one of several options “being looked at closely in this population. We also need to think of nonpharmacologic measures,” Dr. Dave said.
In addition to infants with persistent pulmonary hypertension, the 22 INAS cases at the study site included, among others, three children on extracorporeal membrane oxygenation, one with meconium aspiration syndrome, and one surgical case, out of the 15 included in the study. The common denominator was the need to keep infants calm and comfortable during prolonged intubation, which was a mean of 10.5 days among INAS infants versus 5 among children who didn’t go into opioid withdrawal.
INAS infants had a daily mean morphine-equivalent dose of 106.6 mg/kg, with a mean exposure of 17 days and mean cumulative dose of 1,515 mg/kg. The daily mean morphine-equivalent dose among infants who didn’t develop INAS was 42.4 mg/kg, with a mean exposure of 4 days and mean cumulative dose of 246 mg/kg.
INAS infants spent a mean of 27 days in the hospital, and it took them a mean of almost 6 days to reach full feeds, versus 15 days for the other infants full feeds by day 4. Over half of the INAS infants (12) also were on midazolam, and they had higher cumulative doses of the sedative than infants who didn’t develop INAS (mean, 2.64 mg/kg vs. 0.19 mg/kg). The findings all were statistically significant.
Dr. Dave said the most surprising finding was the variability in opioid dosing. In another example, some infants received up to 1,400 mg/kg morphine equivalents even when their fraction of inspired oxygen requirement fell below 60%, which meant that they were getting better. Other infants by that point were off opioids altogether.
“This has definitely brought awareness to my practice. Before I would say, ‘Okay, let’s just go up,’ ” when a nurse requested an opioid increase based on N-PASS scores. Now, “I try to really figure out why they think the baby needs an increase, and I may say ‘Actually, we are turning a corner now, and maybe the baby can be a little bit more awake. How do you feel about that?’ ” she said.
“My long-term goal for this project is putting some guidelines in place,” she said.
There was no industry funding for the work, and Dr. Dave didn’t have any disclosures.
BALTIMORE – Some infants, especially among those with persistent pulmonary hypertension, are at risk for developing iatrogenic neonatal abstinence syndrome, according to Amber Dave, MD, a neonatal-perinatal medicine fellow at Georgetown University Hospital in Washington.
Of 70 infants administered morphine or fentanyl for longer than a day in the neonatal ICU, almost a third (22, or 31%) developed iatrogenic neonatal abstinence syndrome (INAS). As a result, they needed prolonged respiratory support, more time to reach full feeds, and extended lengths of stay. Children exposed to opioids before birth were excluded from the analysis.
The greatest risk was in infants with persistent pulmonary hypertension; INAS was diagnosed in 13 of 22 (57%).
Opioid dosing also was all over the map for a given Neonatal Pain, Agitation, and Sedation Scale (N-PASS) score, Dr. Dave said. Some infants with an N-PASS pain score of 2, for instance, received no opioids, while others received up to 1,500 mg/kg morphine equivalents.
N-PASS is used in NICUs nationwide to guide dosing, but the variability seen in the study suggests that there’s need for a more objective measure of neonatal distress and for neonatologists to establish ground rules for NICU opioid use, she added.
The use of opioids has been increasing in NICUs for years (J Opioid Manag. 2015 Jul-Aug;11[4]:305-12), and at least one institution (J Perinatol. 2017 Sep;37[9]:1038-42) already has established guidelines to curb overuse. Dr. Dave said that several neonatologists, after viewing her poster at the Pediatric Academic Societies annual meeting, told her that they probably had the same problem at their NICUs but had not examined their data.
“We are using” these medications more in the NICU, “but how much is too much? We need to find that balance. We need to improve our practice.”
“The overarching question is if there are better alternatives for treating pain and stress in critically ill neonates.” Dexmedetomidine, an opioid-sparing alpha-2 agonist adrenoreceptor sedative, analgesic, and anxiolytic, is one of several options “being looked at closely in this population. We also need to think of nonpharmacologic measures,” Dr. Dave said.
In addition to infants with persistent pulmonary hypertension, the 22 INAS cases at the study site included, among others, three children on extracorporeal membrane oxygenation, one with meconium aspiration syndrome, and one surgical case, out of the 15 included in the study. The common denominator was the need to keep infants calm and comfortable during prolonged intubation, which was a mean of 10.5 days among INAS infants versus 5 among children who didn’t go into opioid withdrawal.
INAS infants had a daily mean morphine-equivalent dose of 106.6 mg/kg, with a mean exposure of 17 days and mean cumulative dose of 1,515 mg/kg. The daily mean morphine-equivalent dose among infants who didn’t develop INAS was 42.4 mg/kg, with a mean exposure of 4 days and mean cumulative dose of 246 mg/kg.
INAS infants spent a mean of 27 days in the hospital, and it took them a mean of almost 6 days to reach full feeds, versus 15 days for the other infants full feeds by day 4. Over half of the INAS infants (12) also were on midazolam, and they had higher cumulative doses of the sedative than infants who didn’t develop INAS (mean, 2.64 mg/kg vs. 0.19 mg/kg). The findings all were statistically significant.
Dr. Dave said the most surprising finding was the variability in opioid dosing. In another example, some infants received up to 1,400 mg/kg morphine equivalents even when their fraction of inspired oxygen requirement fell below 60%, which meant that they were getting better. Other infants by that point were off opioids altogether.
“This has definitely brought awareness to my practice. Before I would say, ‘Okay, let’s just go up,’ ” when a nurse requested an opioid increase based on N-PASS scores. Now, “I try to really figure out why they think the baby needs an increase, and I may say ‘Actually, we are turning a corner now, and maybe the baby can be a little bit more awake. How do you feel about that?’ ” she said.
“My long-term goal for this project is putting some guidelines in place,” she said.
There was no industry funding for the work, and Dr. Dave didn’t have any disclosures.
REPORTING FROM PAS 2019
Key clinical point: Some infants in the NICU, especially those with persistent pulmonary hypertension, are at risk for iatrogenic neonatal abstinence syndrome.
Major finding: Of 70 infants administered morphine or fentanyl for longer than a day, almost a third (22) developed iatrogenic neonatal abstinence syndrome.
Study details: Single-center NICU chart review.
Disclosures: There was no industry funding, and the lead investigator didn’t have any relevant financial disclosures.
Young children with neuromuscular disease are vulnerable to respiratory viruses
This highlights the need for new vaccines
Influenza gets a lot of attention each winter, but respiratory syncytial virus (RSV) and other respiratory viruses have as much or more impact on pediatric populations, particularly certain high-risk groups. But currently there are no vaccines for noninfluenza respiratory viruses. That said, several are under development, for RSV and parainfluenza.
Which groups are likely to get the most benefit from these newer vaccines?
We all are aware of the extra vulnerability to respiratory viruses (RSV being the most frequent) in premature infants, those with chronic lung disease, or those with congenital heart syndromes; such vulnerable patients are not infrequently seen in routine practice. A recent report shined a brighter light on such a group.
Real-world data from a nationwide Canadian surveillance system (CARESS) was used to analyze relative risks of categories of young children who are thought to be vulnerable to respiratory viruses, with a particular focus on those with neuromuscular disease. The CARESS investigators analyzed 12 years’ data on respiratory hospitalizations from among palivizumab-prophylaxed patients (including specific data on RSV when patients were tested for RSV per standard of care).1 Unfortunately, RSV testing was not universal despite hospitalization, so the true incidence of RSV-specific hospitalizations was likely underestimated.
Nevertheless, more than 25,000 children from 2005 through 2017 were grouped into three categories of palivizumab-prophylaxed high-risk children: standard indications (SI), n = 20,335; chronic medical conditions (CMD), n = 4,063; and neuromuscular disease (NMD), n = 605. This study is notable for having a relatively large number of neuromuscular disease subjects. Two-thirds of each group were fully palivizumab adherent.
The SI group included the standard American Academy of Pediatrics–recommended groups, such as premature infants, congenital heart disease, etc.
The CMD group included conditions that lead clinicians to use palivizumab off label, such as cystic fibrosis, congenital airway anomalies, immunodeficiency, and pulmonary disorders.
The NMD participants were subdivided into two groups. Group 1 comprised general hypotonic neuromuscular diseases such as hypoxic-ischemic encephalopathy, Prader-Willi syndrome, chromosomal disorders, and migration/demyelinating diseases. Group 2 included more severe infantile neuromuscular disorders, such as spinal muscular atrophy, myotonic dystrophy, centronuclear and nemaline myopathy, mitochondrial and glycogen storage myopathies, or arthrogryposis.
Overall, 6.9% of CARESS RSV-prophylaxed subjects were hospitalized. About one in five hospitalized patients from each group was hospitalized more than once. Specific respiratory hospitalization rates for each group were 6% (n = 1,228) for SI subjects and 9.4% (n = 380) for CMD, compared with 19.2% (n = 116) for NMD subjects.
It is unclear what proportion underwent RSV testing, but a total of 334 were confirmed RSV positive: 261 were SI, 54 were CMD and 19 were NMD. The RSV-test-positive rate was 1.5% for SI, 1.6% for CMD and 3.3% for NMD; so while a higher number of SI children were RSV positive, the rate of RSV positivity was actually highest with NMD.
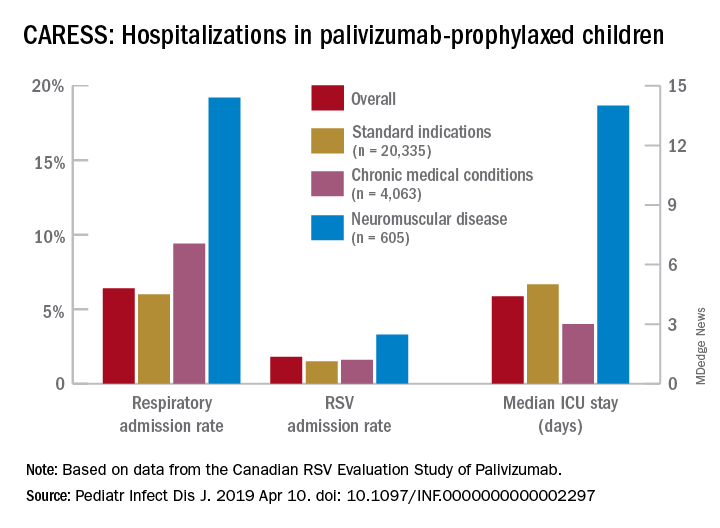
RSV-positive subjects needing ICU care among NMD patients also had longer ICU stays (median 14 days), compared with RSV-positive CMD or SI subjects (median 3 and 5 days, respectively). Further, hospitalized RSV-positive NMD subjects presented more frequently with pneumonia (42% vs. 30% for CMD and 20% for SI) while hospitalized RSV-positive SI subjects more often had apnea (17% vs. 10% for NMD and 5% for CMD, P less than .05).
These differences in the courses of NMD patients raise the question as to whether the NMD group was somehow different from the SI and CMD groups, other than muscular weakness that likely leads to less ability to clear secretions and a less efficient cough. It turns out that NMD children were older and had worse neonatal medical courses (longer hospital stays, more often ventilated, and used oxygen longer). It could be argued that these differences may have been in part due to the muscular weakness inherent in their underlying disease, but they appear to be predictors of worse respiratory infectious disease than other vulnerable populations as the NMD children get older.
Indeed, the overall risk of any respiratory admission among NMD subjects was nearly twice as high, compared with SI (hazard ratio, 1.90, P less than .0005); but the somewhat higher risk for NMD vs. CMD was not significant (HR, 1.33, P = .090). However, when looking specifically at RSV confirmed admissions, NMD had more than twice the hospitalization risk than either other group (HR, 2.26, P = .001 vs. SI; and HR, 2.74, P = .001 vs. CMD).
Further, an NMD subgroup analysis showed 1.69 times the overall respiratory hospitalization risk among the more severe vs. less severe NMD group, but a similar risk of RSV admission. The authors point out that one reason for this discrepancy may be a higher probability of aspiration causing hospitalization because of more dramatic acute events during respiratory infections in patients with more severe NMD. It also may be that palivizumab evened the playing field for RSV but not for other viruses such as parainfluenza, adenovirus, or even rhinovirus.
Nevertheless, these data tell us that risk of respiratory disease severe enough to need hospitalization continues to an older age in NMD than SI or CMD patients, well past 2 years of age. And the risk is not only from RSV. That said, RSV remains a player in some patients (particularly NMD patients) despite palivizumab prophylaxis, highlighting the need for RSV as well as parainfluenza vaccines. While these vaccines should help all young children, they seem likely to be even more beneficial for high-risk children including those with NMD, and particularly those with more severe NMD.
Eleven among 60 total candidate RSV vaccines (live attenuated, particle based, or vector based) are currently in clinical trials.2 Fewer parainfluenza vaccines are in the pipeline, but clinical trials also are underway.3-5 Approval of such vaccines is not expected until the mid-2020s, so at present we are left with providing palivizumab to our vulnerable patients while emphasizing nonmedical strategies that may help prevent respiratory viruses. These only partially successful preventive interventions include breastfeeding, avoiding secondhand smoke, and avoiding known high-risk exposures, such as large day care centers.
My hope is for quicker than projected progress on the vaccine front so that winter admissions for respiratory viruses might decrease in numbers similar to the decrease we have noted with another vaccine successful against a seasonally active pathogen – rotavirus.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatr Infect Dis J. 2019 Apr 10. doi: 10.1097/INF.0000000000002297.
2. “Advances in RSV Vaccine Research and Development – A Global Agenda.”
3. J Pediatric Infect Dis Soc. 2015 Dec;4(4): e143-6.
4. J Virol. 2015 Oct;89(20):10319-32.
5. Vaccine. 2017 Dec 18;35(51):7139-46.
This highlights the need for new vaccines
This highlights the need for new vaccines
Influenza gets a lot of attention each winter, but respiratory syncytial virus (RSV) and other respiratory viruses have as much or more impact on pediatric populations, particularly certain high-risk groups. But currently there are no vaccines for noninfluenza respiratory viruses. That said, several are under development, for RSV and parainfluenza.
Which groups are likely to get the most benefit from these newer vaccines?
We all are aware of the extra vulnerability to respiratory viruses (RSV being the most frequent) in premature infants, those with chronic lung disease, or those with congenital heart syndromes; such vulnerable patients are not infrequently seen in routine practice. A recent report shined a brighter light on such a group.
Real-world data from a nationwide Canadian surveillance system (CARESS) was used to analyze relative risks of categories of young children who are thought to be vulnerable to respiratory viruses, with a particular focus on those with neuromuscular disease. The CARESS investigators analyzed 12 years’ data on respiratory hospitalizations from among palivizumab-prophylaxed patients (including specific data on RSV when patients were tested for RSV per standard of care).1 Unfortunately, RSV testing was not universal despite hospitalization, so the true incidence of RSV-specific hospitalizations was likely underestimated.
Nevertheless, more than 25,000 children from 2005 through 2017 were grouped into three categories of palivizumab-prophylaxed high-risk children: standard indications (SI), n = 20,335; chronic medical conditions (CMD), n = 4,063; and neuromuscular disease (NMD), n = 605. This study is notable for having a relatively large number of neuromuscular disease subjects. Two-thirds of each group were fully palivizumab adherent.
The SI group included the standard American Academy of Pediatrics–recommended groups, such as premature infants, congenital heart disease, etc.
The CMD group included conditions that lead clinicians to use palivizumab off label, such as cystic fibrosis, congenital airway anomalies, immunodeficiency, and pulmonary disorders.
The NMD participants were subdivided into two groups. Group 1 comprised general hypotonic neuromuscular diseases such as hypoxic-ischemic encephalopathy, Prader-Willi syndrome, chromosomal disorders, and migration/demyelinating diseases. Group 2 included more severe infantile neuromuscular disorders, such as spinal muscular atrophy, myotonic dystrophy, centronuclear and nemaline myopathy, mitochondrial and glycogen storage myopathies, or arthrogryposis.
Overall, 6.9% of CARESS RSV-prophylaxed subjects were hospitalized. About one in five hospitalized patients from each group was hospitalized more than once. Specific respiratory hospitalization rates for each group were 6% (n = 1,228) for SI subjects and 9.4% (n = 380) for CMD, compared with 19.2% (n = 116) for NMD subjects.
It is unclear what proportion underwent RSV testing, but a total of 334 were confirmed RSV positive: 261 were SI, 54 were CMD and 19 were NMD. The RSV-test-positive rate was 1.5% for SI, 1.6% for CMD and 3.3% for NMD; so while a higher number of SI children were RSV positive, the rate of RSV positivity was actually highest with NMD.

RSV-positive subjects needing ICU care among NMD patients also had longer ICU stays (median 14 days), compared with RSV-positive CMD or SI subjects (median 3 and 5 days, respectively). Further, hospitalized RSV-positive NMD subjects presented more frequently with pneumonia (42% vs. 30% for CMD and 20% for SI) while hospitalized RSV-positive SI subjects more often had apnea (17% vs. 10% for NMD and 5% for CMD, P less than .05).
These differences in the courses of NMD patients raise the question as to whether the NMD group was somehow different from the SI and CMD groups, other than muscular weakness that likely leads to less ability to clear secretions and a less efficient cough. It turns out that NMD children were older and had worse neonatal medical courses (longer hospital stays, more often ventilated, and used oxygen longer). It could be argued that these differences may have been in part due to the muscular weakness inherent in their underlying disease, but they appear to be predictors of worse respiratory infectious disease than other vulnerable populations as the NMD children get older.
Indeed, the overall risk of any respiratory admission among NMD subjects was nearly twice as high, compared with SI (hazard ratio, 1.90, P less than .0005); but the somewhat higher risk for NMD vs. CMD was not significant (HR, 1.33, P = .090). However, when looking specifically at RSV confirmed admissions, NMD had more than twice the hospitalization risk than either other group (HR, 2.26, P = .001 vs. SI; and HR, 2.74, P = .001 vs. CMD).
Further, an NMD subgroup analysis showed 1.69 times the overall respiratory hospitalization risk among the more severe vs. less severe NMD group, but a similar risk of RSV admission. The authors point out that one reason for this discrepancy may be a higher probability of aspiration causing hospitalization because of more dramatic acute events during respiratory infections in patients with more severe NMD. It also may be that palivizumab evened the playing field for RSV but not for other viruses such as parainfluenza, adenovirus, or even rhinovirus.
Nevertheless, these data tell us that risk of respiratory disease severe enough to need hospitalization continues to an older age in NMD than SI or CMD patients, well past 2 years of age. And the risk is not only from RSV. That said, RSV remains a player in some patients (particularly NMD patients) despite palivizumab prophylaxis, highlighting the need for RSV as well as parainfluenza vaccines. While these vaccines should help all young children, they seem likely to be even more beneficial for high-risk children including those with NMD, and particularly those with more severe NMD.
Eleven among 60 total candidate RSV vaccines (live attenuated, particle based, or vector based) are currently in clinical trials.2 Fewer parainfluenza vaccines are in the pipeline, but clinical trials also are underway.3-5 Approval of such vaccines is not expected until the mid-2020s, so at present we are left with providing palivizumab to our vulnerable patients while emphasizing nonmedical strategies that may help prevent respiratory viruses. These only partially successful preventive interventions include breastfeeding, avoiding secondhand smoke, and avoiding known high-risk exposures, such as large day care centers.
My hope is for quicker than projected progress on the vaccine front so that winter admissions for respiratory viruses might decrease in numbers similar to the decrease we have noted with another vaccine successful against a seasonally active pathogen – rotavirus.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatr Infect Dis J. 2019 Apr 10. doi: 10.1097/INF.0000000000002297.
2. “Advances in RSV Vaccine Research and Development – A Global Agenda.”
3. J Pediatric Infect Dis Soc. 2015 Dec;4(4): e143-6.
4. J Virol. 2015 Oct;89(20):10319-32.
5. Vaccine. 2017 Dec 18;35(51):7139-46.
Influenza gets a lot of attention each winter, but respiratory syncytial virus (RSV) and other respiratory viruses have as much or more impact on pediatric populations, particularly certain high-risk groups. But currently there are no vaccines for noninfluenza respiratory viruses. That said, several are under development, for RSV and parainfluenza.
Which groups are likely to get the most benefit from these newer vaccines?
We all are aware of the extra vulnerability to respiratory viruses (RSV being the most frequent) in premature infants, those with chronic lung disease, or those with congenital heart syndromes; such vulnerable patients are not infrequently seen in routine practice. A recent report shined a brighter light on such a group.
Real-world data from a nationwide Canadian surveillance system (CARESS) was used to analyze relative risks of categories of young children who are thought to be vulnerable to respiratory viruses, with a particular focus on those with neuromuscular disease. The CARESS investigators analyzed 12 years’ data on respiratory hospitalizations from among palivizumab-prophylaxed patients (including specific data on RSV when patients were tested for RSV per standard of care).1 Unfortunately, RSV testing was not universal despite hospitalization, so the true incidence of RSV-specific hospitalizations was likely underestimated.
Nevertheless, more than 25,000 children from 2005 through 2017 were grouped into three categories of palivizumab-prophylaxed high-risk children: standard indications (SI), n = 20,335; chronic medical conditions (CMD), n = 4,063; and neuromuscular disease (NMD), n = 605. This study is notable for having a relatively large number of neuromuscular disease subjects. Two-thirds of each group were fully palivizumab adherent.
The SI group included the standard American Academy of Pediatrics–recommended groups, such as premature infants, congenital heart disease, etc.
The CMD group included conditions that lead clinicians to use palivizumab off label, such as cystic fibrosis, congenital airway anomalies, immunodeficiency, and pulmonary disorders.
The NMD participants were subdivided into two groups. Group 1 comprised general hypotonic neuromuscular diseases such as hypoxic-ischemic encephalopathy, Prader-Willi syndrome, chromosomal disorders, and migration/demyelinating diseases. Group 2 included more severe infantile neuromuscular disorders, such as spinal muscular atrophy, myotonic dystrophy, centronuclear and nemaline myopathy, mitochondrial and glycogen storage myopathies, or arthrogryposis.
Overall, 6.9% of CARESS RSV-prophylaxed subjects were hospitalized. About one in five hospitalized patients from each group was hospitalized more than once. Specific respiratory hospitalization rates for each group were 6% (n = 1,228) for SI subjects and 9.4% (n = 380) for CMD, compared with 19.2% (n = 116) for NMD subjects.
It is unclear what proportion underwent RSV testing, but a total of 334 were confirmed RSV positive: 261 were SI, 54 were CMD and 19 were NMD. The RSV-test-positive rate was 1.5% for SI, 1.6% for CMD and 3.3% for NMD; so while a higher number of SI children were RSV positive, the rate of RSV positivity was actually highest with NMD.

RSV-positive subjects needing ICU care among NMD patients also had longer ICU stays (median 14 days), compared with RSV-positive CMD or SI subjects (median 3 and 5 days, respectively). Further, hospitalized RSV-positive NMD subjects presented more frequently with pneumonia (42% vs. 30% for CMD and 20% for SI) while hospitalized RSV-positive SI subjects more often had apnea (17% vs. 10% for NMD and 5% for CMD, P less than .05).
These differences in the courses of NMD patients raise the question as to whether the NMD group was somehow different from the SI and CMD groups, other than muscular weakness that likely leads to less ability to clear secretions and a less efficient cough. It turns out that NMD children were older and had worse neonatal medical courses (longer hospital stays, more often ventilated, and used oxygen longer). It could be argued that these differences may have been in part due to the muscular weakness inherent in their underlying disease, but they appear to be predictors of worse respiratory infectious disease than other vulnerable populations as the NMD children get older.
Indeed, the overall risk of any respiratory admission among NMD subjects was nearly twice as high, compared with SI (hazard ratio, 1.90, P less than .0005); but the somewhat higher risk for NMD vs. CMD was not significant (HR, 1.33, P = .090). However, when looking specifically at RSV confirmed admissions, NMD had more than twice the hospitalization risk than either other group (HR, 2.26, P = .001 vs. SI; and HR, 2.74, P = .001 vs. CMD).
Further, an NMD subgroup analysis showed 1.69 times the overall respiratory hospitalization risk among the more severe vs. less severe NMD group, but a similar risk of RSV admission. The authors point out that one reason for this discrepancy may be a higher probability of aspiration causing hospitalization because of more dramatic acute events during respiratory infections in patients with more severe NMD. It also may be that palivizumab evened the playing field for RSV but not for other viruses such as parainfluenza, adenovirus, or even rhinovirus.
Nevertheless, these data tell us that risk of respiratory disease severe enough to need hospitalization continues to an older age in NMD than SI or CMD patients, well past 2 years of age. And the risk is not only from RSV. That said, RSV remains a player in some patients (particularly NMD patients) despite palivizumab prophylaxis, highlighting the need for RSV as well as parainfluenza vaccines. While these vaccines should help all young children, they seem likely to be even more beneficial for high-risk children including those with NMD, and particularly those with more severe NMD.
Eleven among 60 total candidate RSV vaccines (live attenuated, particle based, or vector based) are currently in clinical trials.2 Fewer parainfluenza vaccines are in the pipeline, but clinical trials also are underway.3-5 Approval of such vaccines is not expected until the mid-2020s, so at present we are left with providing palivizumab to our vulnerable patients while emphasizing nonmedical strategies that may help prevent respiratory viruses. These only partially successful preventive interventions include breastfeeding, avoiding secondhand smoke, and avoiding known high-risk exposures, such as large day care centers.
My hope is for quicker than projected progress on the vaccine front so that winter admissions for respiratory viruses might decrease in numbers similar to the decrease we have noted with another vaccine successful against a seasonally active pathogen – rotavirus.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatr Infect Dis J. 2019 Apr 10. doi: 10.1097/INF.0000000000002297.
2. “Advances in RSV Vaccine Research and Development – A Global Agenda.”
3. J Pediatric Infect Dis Soc. 2015 Dec;4(4): e143-6.
4. J Virol. 2015 Oct;89(20):10319-32.
5. Vaccine. 2017 Dec 18;35(51):7139-46.
Maternal immunization protects against serious RSV infection in infancy
LJUBLJANA, SLOVENIA – Passive protection of infants from severe respiratory syncytial virus lower respiratory tract infection during the first 6 months of life has convincingly been achieved through maternal immunization using a novel nanoparticle vaccine in the landmark PREPARE trial.
“I think it’s important for everyone, especially people like myself who’ve been working on maternal immunization for about 20 years, to realize that this is a historic study,” Flor M. Munoz, MD, declared in reporting the study results at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We have here for the first time a phase-3, global, randomized, placebo-controlled, observer-blinded clinical trial looking at an experimental vaccine in pregnant women for the protection of infants from a disease for which we really don’t have other potential solutions quite yet, and in a period of high vulnerability,” said Dr. Munoz, a pediatric infectious disease specialist at Baylor College of Medicine, Houston.
Indeed, respiratory syncytial virus (RSV) is the No. 2 cause of mortality worldwide during the first year of life. Moreover, most cases of severe RSV lower respiratory tract infection occur in otherwise healthy infants aged less than 5 months, when active immunization presents daunting challenges.
“While certainly mortality is uncommon in high-income countries, we do see significant hospitalization there due to severe RSV lower respiratory tract infection in the first year of life, sometimes more than other common diseases, like influenza,” she noted.
PREPARE included 4,636 women with low-risk pregnancies who were randomized 2:1 to a single intramuscular injection of the investigational RSV vaccine or placebo during gestational weeks 28-36, with efficacy assessed through the first 180 days of life. The study took place at 87 sites in 11 countries during 4 years worth of RSV seasons. Roughly half of participants were South African, one-quarter were in the United States, and the rest were drawn from nine other low-, middle-, or high-income countries in the Northern and Southern Hemispheres. The median gestational age at vaccination was 32 weeks.
The primary efficacy endpoint specified by the Food and Drug Administration – but not other regulatory agencies – was the placebo-subtracted rate of RSV lower respiratory tract infection as defined by RSV detected by reverse transcription polymerase chain reaction, along with at least one clinical manifestation of lower respiratory tract infection, oxygen saturation below 95%, and/or tachypnea. The risk of this outcome was reduced by 39% during the first 90 days of life and by 27% through 180 days in infants in the maternal immunization group, a difference which didn’t achieve statistical significance.
However, prespecified major secondary endpoints arguably of greater clinical relevance were consistently positive. Notably, when levels of transplacentally transferred neutralizing antibodies against RSV A and B were highest, with events occurring in 57 of 2,765 evaluable infants in the active treatment arm and in 53 of 1,430 controls. Similarly, there was a 40% reduction through day 180. Moreover, rates of another key secondary endpoint – RSV lower respiratory tract infection plus severe hypoxemia with an oxygen saturation below 92% – were reduced by 48% and 42% through days 90 and 180, respectively. Thus, the vaccine’s protective effect was greatest against the most severe outcomes of RSV infection in infancy, according to Dr. Munoz.
No safety signals related to this immunization strategy were seen during 1 year of follow-up of infants and 6 months for the mothers. Side effects were essentially limited to mild, self-limited injection site reactions, with zero impact on pregnancy and delivery.
An intriguing finding in an exploratory analysis was that the vaccine appeared to have ancillary benefits beyond prevention of medically significant RSV disease in the young infants. For example, the rate of all lower respiratory tract infections with severe hypoxemia – with no requirement for demonstration of RSV infection – was reduced by 46% during the first 90 days of life in the immunized group. Similarly, the rate of all-cause lower respiratory tract infection resulting in hospitalization was reduced by 28%.
“This is actually quite interesting, because these are unexpected benefits in terms of all-cause effects,” the pediatrician commented, adding that she and her coinvestigators are delving into this phenomenon in order to gain better understanding.
Additional analyses of the recently completed PREPARE study are ongoing but already have yielded some important findings. For example, women immunized before 33 weeks’ gestation had significantly greater transplacental antibody transfer than those immunized later in pregnancy, with resultant markedly greater vaccine efficacy in their offspring as well: A placebo-subtracted 70% reduction in RSV lower respiratory tract infection with severe hypoxemia through 90 days, compared with a 44% reduction associated with immunization at gestational week 33 or later. And when the interval between immunization and delivery was at least 30 days, the risk of this endpoint was reduced by 65%; in contrast, there was no significant difference between vaccine and placebo groups when time from immunization to delivery was less than 30 days.
Also noteworthy was that maternal immunization afforded no infant protection in the United States. This unanticipated finding is still under investigation, although suspicion centers around the fact that RSV seasons were generally milder there, and American women were vaccinated at a later gestational age, with a corresponding shorter interval to delivery.
The novel recombinant nanoparticle vaccine tested in PREPARE contains a nearly full-length RSV fusion protein produced in insect cells. The nanoparticles express both prefusion epitopes and epitopes common to pre- and postfusion conformations. Aluminum phosphate is employed as the adjuvant.
Novavax’s stock price has been kicked to the curb since the company earlier reported that a large phase 3 trial of the vaccine failed to meet its primary endpoint for prevention of RSV lower respiratory tract infection in older adults. Now the vaccine’s failure to meet its prespecified FDA-mandated primary endpoint in the maternal immunization study will doubtless spawn further financially dismissive headlines in the business press as well.
But pediatricians are famously advocates for children, and PREPARE received a warm welcome from the pediatric infectious disease community, regardless of investor response. Indeed, PREPARE was the only clinical trial deemed of sufficient import to be featured in the opening plenary session of ESPID 2019.
Ulrich Heininger, MD, professor of pediatrics at the University of Basel (Switzerland), who cochaired the session, jointly sponsored by ESPID and the Pediatric Infectious Diseases Society, declared, “These findings, I think, are a great step forward.”
Dr. Munoz reported receiving research grants from Janssen, the National Institutes of Health, the Centers for Disease Control and Prevention, and Novavax, which sponsored the PREPARE trial, assisted by an $89 million grant from the Bill and Melinda Gates Foundation.
LJUBLJANA, SLOVENIA – Passive protection of infants from severe respiratory syncytial virus lower respiratory tract infection during the first 6 months of life has convincingly been achieved through maternal immunization using a novel nanoparticle vaccine in the landmark PREPARE trial.
“I think it’s important for everyone, especially people like myself who’ve been working on maternal immunization for about 20 years, to realize that this is a historic study,” Flor M. Munoz, MD, declared in reporting the study results at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We have here for the first time a phase-3, global, randomized, placebo-controlled, observer-blinded clinical trial looking at an experimental vaccine in pregnant women for the protection of infants from a disease for which we really don’t have other potential solutions quite yet, and in a period of high vulnerability,” said Dr. Munoz, a pediatric infectious disease specialist at Baylor College of Medicine, Houston.
Indeed, respiratory syncytial virus (RSV) is the No. 2 cause of mortality worldwide during the first year of life. Moreover, most cases of severe RSV lower respiratory tract infection occur in otherwise healthy infants aged less than 5 months, when active immunization presents daunting challenges.
“While certainly mortality is uncommon in high-income countries, we do see significant hospitalization there due to severe RSV lower respiratory tract infection in the first year of life, sometimes more than other common diseases, like influenza,” she noted.
PREPARE included 4,636 women with low-risk pregnancies who were randomized 2:1 to a single intramuscular injection of the investigational RSV vaccine or placebo during gestational weeks 28-36, with efficacy assessed through the first 180 days of life. The study took place at 87 sites in 11 countries during 4 years worth of RSV seasons. Roughly half of participants were South African, one-quarter were in the United States, and the rest were drawn from nine other low-, middle-, or high-income countries in the Northern and Southern Hemispheres. The median gestational age at vaccination was 32 weeks.
The primary efficacy endpoint specified by the Food and Drug Administration – but not other regulatory agencies – was the placebo-subtracted rate of RSV lower respiratory tract infection as defined by RSV detected by reverse transcription polymerase chain reaction, along with at least one clinical manifestation of lower respiratory tract infection, oxygen saturation below 95%, and/or tachypnea. The risk of this outcome was reduced by 39% during the first 90 days of life and by 27% through 180 days in infants in the maternal immunization group, a difference which didn’t achieve statistical significance.
However, prespecified major secondary endpoints arguably of greater clinical relevance were consistently positive. Notably, when levels of transplacentally transferred neutralizing antibodies against RSV A and B were highest, with events occurring in 57 of 2,765 evaluable infants in the active treatment arm and in 53 of 1,430 controls. Similarly, there was a 40% reduction through day 180. Moreover, rates of another key secondary endpoint – RSV lower respiratory tract infection plus severe hypoxemia with an oxygen saturation below 92% – were reduced by 48% and 42% through days 90 and 180, respectively. Thus, the vaccine’s protective effect was greatest against the most severe outcomes of RSV infection in infancy, according to Dr. Munoz.
No safety signals related to this immunization strategy were seen during 1 year of follow-up of infants and 6 months for the mothers. Side effects were essentially limited to mild, self-limited injection site reactions, with zero impact on pregnancy and delivery.
An intriguing finding in an exploratory analysis was that the vaccine appeared to have ancillary benefits beyond prevention of medically significant RSV disease in the young infants. For example, the rate of all lower respiratory tract infections with severe hypoxemia – with no requirement for demonstration of RSV infection – was reduced by 46% during the first 90 days of life in the immunized group. Similarly, the rate of all-cause lower respiratory tract infection resulting in hospitalization was reduced by 28%.
“This is actually quite interesting, because these are unexpected benefits in terms of all-cause effects,” the pediatrician commented, adding that she and her coinvestigators are delving into this phenomenon in order to gain better understanding.
Additional analyses of the recently completed PREPARE study are ongoing but already have yielded some important findings. For example, women immunized before 33 weeks’ gestation had significantly greater transplacental antibody transfer than those immunized later in pregnancy, with resultant markedly greater vaccine efficacy in their offspring as well: A placebo-subtracted 70% reduction in RSV lower respiratory tract infection with severe hypoxemia through 90 days, compared with a 44% reduction associated with immunization at gestational week 33 or later. And when the interval between immunization and delivery was at least 30 days, the risk of this endpoint was reduced by 65%; in contrast, there was no significant difference between vaccine and placebo groups when time from immunization to delivery was less than 30 days.
Also noteworthy was that maternal immunization afforded no infant protection in the United States. This unanticipated finding is still under investigation, although suspicion centers around the fact that RSV seasons were generally milder there, and American women were vaccinated at a later gestational age, with a corresponding shorter interval to delivery.
The novel recombinant nanoparticle vaccine tested in PREPARE contains a nearly full-length RSV fusion protein produced in insect cells. The nanoparticles express both prefusion epitopes and epitopes common to pre- and postfusion conformations. Aluminum phosphate is employed as the adjuvant.
Novavax’s stock price has been kicked to the curb since the company earlier reported that a large phase 3 trial of the vaccine failed to meet its primary endpoint for prevention of RSV lower respiratory tract infection in older adults. Now the vaccine’s failure to meet its prespecified FDA-mandated primary endpoint in the maternal immunization study will doubtless spawn further financially dismissive headlines in the business press as well.
But pediatricians are famously advocates for children, and PREPARE received a warm welcome from the pediatric infectious disease community, regardless of investor response. Indeed, PREPARE was the only clinical trial deemed of sufficient import to be featured in the opening plenary session of ESPID 2019.
Ulrich Heininger, MD, professor of pediatrics at the University of Basel (Switzerland), who cochaired the session, jointly sponsored by ESPID and the Pediatric Infectious Diseases Society, declared, “These findings, I think, are a great step forward.”
Dr. Munoz reported receiving research grants from Janssen, the National Institutes of Health, the Centers for Disease Control and Prevention, and Novavax, which sponsored the PREPARE trial, assisted by an $89 million grant from the Bill and Melinda Gates Foundation.
LJUBLJANA, SLOVENIA – Passive protection of infants from severe respiratory syncytial virus lower respiratory tract infection during the first 6 months of life has convincingly been achieved through maternal immunization using a novel nanoparticle vaccine in the landmark PREPARE trial.
“I think it’s important for everyone, especially people like myself who’ve been working on maternal immunization for about 20 years, to realize that this is a historic study,” Flor M. Munoz, MD, declared in reporting the study results at the annual meeting of the European Society for Paediatric Infectious Diseases.
“We have here for the first time a phase-3, global, randomized, placebo-controlled, observer-blinded clinical trial looking at an experimental vaccine in pregnant women for the protection of infants from a disease for which we really don’t have other potential solutions quite yet, and in a period of high vulnerability,” said Dr. Munoz, a pediatric infectious disease specialist at Baylor College of Medicine, Houston.
Indeed, respiratory syncytial virus (RSV) is the No. 2 cause of mortality worldwide during the first year of life. Moreover, most cases of severe RSV lower respiratory tract infection occur in otherwise healthy infants aged less than 5 months, when active immunization presents daunting challenges.
“While certainly mortality is uncommon in high-income countries, we do see significant hospitalization there due to severe RSV lower respiratory tract infection in the first year of life, sometimes more than other common diseases, like influenza,” she noted.
PREPARE included 4,636 women with low-risk pregnancies who were randomized 2:1 to a single intramuscular injection of the investigational RSV vaccine or placebo during gestational weeks 28-36, with efficacy assessed through the first 180 days of life. The study took place at 87 sites in 11 countries during 4 years worth of RSV seasons. Roughly half of participants were South African, one-quarter were in the United States, and the rest were drawn from nine other low-, middle-, or high-income countries in the Northern and Southern Hemispheres. The median gestational age at vaccination was 32 weeks.
The primary efficacy endpoint specified by the Food and Drug Administration – but not other regulatory agencies – was the placebo-subtracted rate of RSV lower respiratory tract infection as defined by RSV detected by reverse transcription polymerase chain reaction, along with at least one clinical manifestation of lower respiratory tract infection, oxygen saturation below 95%, and/or tachypnea. The risk of this outcome was reduced by 39% during the first 90 days of life and by 27% through 180 days in infants in the maternal immunization group, a difference which didn’t achieve statistical significance.
However, prespecified major secondary endpoints arguably of greater clinical relevance were consistently positive. Notably, when levels of transplacentally transferred neutralizing antibodies against RSV A and B were highest, with events occurring in 57 of 2,765 evaluable infants in the active treatment arm and in 53 of 1,430 controls. Similarly, there was a 40% reduction through day 180. Moreover, rates of another key secondary endpoint – RSV lower respiratory tract infection plus severe hypoxemia with an oxygen saturation below 92% – were reduced by 48% and 42% through days 90 and 180, respectively. Thus, the vaccine’s protective effect was greatest against the most severe outcomes of RSV infection in infancy, according to Dr. Munoz.
No safety signals related to this immunization strategy were seen during 1 year of follow-up of infants and 6 months for the mothers. Side effects were essentially limited to mild, self-limited injection site reactions, with zero impact on pregnancy and delivery.
An intriguing finding in an exploratory analysis was that the vaccine appeared to have ancillary benefits beyond prevention of medically significant RSV disease in the young infants. For example, the rate of all lower respiratory tract infections with severe hypoxemia – with no requirement for demonstration of RSV infection – was reduced by 46% during the first 90 days of life in the immunized group. Similarly, the rate of all-cause lower respiratory tract infection resulting in hospitalization was reduced by 28%.
“This is actually quite interesting, because these are unexpected benefits in terms of all-cause effects,” the pediatrician commented, adding that she and her coinvestigators are delving into this phenomenon in order to gain better understanding.
Additional analyses of the recently completed PREPARE study are ongoing but already have yielded some important findings. For example, women immunized before 33 weeks’ gestation had significantly greater transplacental antibody transfer than those immunized later in pregnancy, with resultant markedly greater vaccine efficacy in their offspring as well: A placebo-subtracted 70% reduction in RSV lower respiratory tract infection with severe hypoxemia through 90 days, compared with a 44% reduction associated with immunization at gestational week 33 or later. And when the interval between immunization and delivery was at least 30 days, the risk of this endpoint was reduced by 65%; in contrast, there was no significant difference between vaccine and placebo groups when time from immunization to delivery was less than 30 days.
Also noteworthy was that maternal immunization afforded no infant protection in the United States. This unanticipated finding is still under investigation, although suspicion centers around the fact that RSV seasons were generally milder there, and American women were vaccinated at a later gestational age, with a corresponding shorter interval to delivery.
The novel recombinant nanoparticle vaccine tested in PREPARE contains a nearly full-length RSV fusion protein produced in insect cells. The nanoparticles express both prefusion epitopes and epitopes common to pre- and postfusion conformations. Aluminum phosphate is employed as the adjuvant.
Novavax’s stock price has been kicked to the curb since the company earlier reported that a large phase 3 trial of the vaccine failed to meet its primary endpoint for prevention of RSV lower respiratory tract infection in older adults. Now the vaccine’s failure to meet its prespecified FDA-mandated primary endpoint in the maternal immunization study will doubtless spawn further financially dismissive headlines in the business press as well.
But pediatricians are famously advocates for children, and PREPARE received a warm welcome from the pediatric infectious disease community, regardless of investor response. Indeed, PREPARE was the only clinical trial deemed of sufficient import to be featured in the opening plenary session of ESPID 2019.
Ulrich Heininger, MD, professor of pediatrics at the University of Basel (Switzerland), who cochaired the session, jointly sponsored by ESPID and the Pediatric Infectious Diseases Society, declared, “These findings, I think, are a great step forward.”
Dr. Munoz reported receiving research grants from Janssen, the National Institutes of Health, the Centers for Disease Control and Prevention, and Novavax, which sponsored the PREPARE trial, assisted by an $89 million grant from the Bill and Melinda Gates Foundation.
REPORTING FROM ESPID 2019
N.Y. hospitals report near-universal CMV screening when newborns fail hearing tests
BALTIMORE – Over the past 2 years, Northwell Health, a large medical system in the metropolitan New York area, increased cytomegalovirus screening for infants who fail hearing tests from 6.6% to 95% at five of its birth hospitals, according to a presentation at the Pediatric Academic Societies annual meeting.
Three cases of congenital cytomegalovirus (CMV) have been picked up so far. The plan is to roll the program out to all 10 of the system’s birth hospitals, where over 40,000 children are born each year.
“We feel very satisfied and proud” of the progress that’s been made at Northwell in such a short time, said Alia Chauhan, MD, a Northwell pediatrician who presented the findings.
Northwell launched its “Hearing Plus” program in 2017 to catch the infection before infants leave the hospital. Several other health systems around the country have launched similar programs, and a handful of states – including New York – now require CMV screening for infants who fail mandated hearing tests.
The issue is gaining traction because hearing loss is often the only sign of congenital CMV, so it’s a bellwether for infection. Screening children with hearing loss is an easy way to pick it up early, so steps can be taken to prevent problems down the road. As it is, congenital CMV is the leading nongenetic cause of hearing loss in infants, accounting for at least 10% of cases.
The Northwell program kicked off with an education campaign to build consensus among pediatricians, hospitalists, and nurses. A flyer was made about CMV screening for moms whose infants fail hearing tests, printed in both English and Spanish.
Initially, the program used urine PCR [polymerase chain reaction] to screen for CMV, but waiting for infants to produce a sample often delayed discharge, so a switch was soon made to saliva swab PCRs, which take seconds, with urine PCR held in reserve to confirm positive swabs.
To streamline the process, a standing order was added to the electronic records system so nurses could order saliva PCRs without having to get physician approval. “I think [that] was one of the biggest things that’s helped us,” Dr. Chauhan said.
Children who test positive must have urine confirmation within 21 days of birth; most are long gone from the hospital by then and have to be called back in. “We haven’t lost anyone to follow-up, but it can be stressful trying to get someone to come back,” she said.
Six of 449 infants have screened positive on saliva – three were false positives with negative urine screens. Of the three confirmed cases, two infants later turned out to have normal hearing on repeat testing and were otherwise asymptomatic.
These days, Dr. Chauhan said, if children have a positive saliva PCR but later turn out to have normal hearing, and are otherwise free of symptoms with no CMV risk factors, “we are not confirming with urine.”
Dr. Chauhan did not have any disclosures. No funding source was mentioned.
SOURCE: Chauhan A et al. PAS 2019. Abstract 306
BALTIMORE – Over the past 2 years, Northwell Health, a large medical system in the metropolitan New York area, increased cytomegalovirus screening for infants who fail hearing tests from 6.6% to 95% at five of its birth hospitals, according to a presentation at the Pediatric Academic Societies annual meeting.
Three cases of congenital cytomegalovirus (CMV) have been picked up so far. The plan is to roll the program out to all 10 of the system’s birth hospitals, where over 40,000 children are born each year.
“We feel very satisfied and proud” of the progress that’s been made at Northwell in such a short time, said Alia Chauhan, MD, a Northwell pediatrician who presented the findings.
Northwell launched its “Hearing Plus” program in 2017 to catch the infection before infants leave the hospital. Several other health systems around the country have launched similar programs, and a handful of states – including New York – now require CMV screening for infants who fail mandated hearing tests.
The issue is gaining traction because hearing loss is often the only sign of congenital CMV, so it’s a bellwether for infection. Screening children with hearing loss is an easy way to pick it up early, so steps can be taken to prevent problems down the road. As it is, congenital CMV is the leading nongenetic cause of hearing loss in infants, accounting for at least 10% of cases.
The Northwell program kicked off with an education campaign to build consensus among pediatricians, hospitalists, and nurses. A flyer was made about CMV screening for moms whose infants fail hearing tests, printed in both English and Spanish.
Initially, the program used urine PCR [polymerase chain reaction] to screen for CMV, but waiting for infants to produce a sample often delayed discharge, so a switch was soon made to saliva swab PCRs, which take seconds, with urine PCR held in reserve to confirm positive swabs.
To streamline the process, a standing order was added to the electronic records system so nurses could order saliva PCRs without having to get physician approval. “I think [that] was one of the biggest things that’s helped us,” Dr. Chauhan said.
Children who test positive must have urine confirmation within 21 days of birth; most are long gone from the hospital by then and have to be called back in. “We haven’t lost anyone to follow-up, but it can be stressful trying to get someone to come back,” she said.
Six of 449 infants have screened positive on saliva – three were false positives with negative urine screens. Of the three confirmed cases, two infants later turned out to have normal hearing on repeat testing and were otherwise asymptomatic.
These days, Dr. Chauhan said, if children have a positive saliva PCR but later turn out to have normal hearing, and are otherwise free of symptoms with no CMV risk factors, “we are not confirming with urine.”
Dr. Chauhan did not have any disclosures. No funding source was mentioned.
SOURCE: Chauhan A et al. PAS 2019. Abstract 306
BALTIMORE – Over the past 2 years, Northwell Health, a large medical system in the metropolitan New York area, increased cytomegalovirus screening for infants who fail hearing tests from 6.6% to 95% at five of its birth hospitals, according to a presentation at the Pediatric Academic Societies annual meeting.
Three cases of congenital cytomegalovirus (CMV) have been picked up so far. The plan is to roll the program out to all 10 of the system’s birth hospitals, where over 40,000 children are born each year.
“We feel very satisfied and proud” of the progress that’s been made at Northwell in such a short time, said Alia Chauhan, MD, a Northwell pediatrician who presented the findings.
Northwell launched its “Hearing Plus” program in 2017 to catch the infection before infants leave the hospital. Several other health systems around the country have launched similar programs, and a handful of states – including New York – now require CMV screening for infants who fail mandated hearing tests.
The issue is gaining traction because hearing loss is often the only sign of congenital CMV, so it’s a bellwether for infection. Screening children with hearing loss is an easy way to pick it up early, so steps can be taken to prevent problems down the road. As it is, congenital CMV is the leading nongenetic cause of hearing loss in infants, accounting for at least 10% of cases.
The Northwell program kicked off with an education campaign to build consensus among pediatricians, hospitalists, and nurses. A flyer was made about CMV screening for moms whose infants fail hearing tests, printed in both English and Spanish.
Initially, the program used urine PCR [polymerase chain reaction] to screen for CMV, but waiting for infants to produce a sample often delayed discharge, so a switch was soon made to saliva swab PCRs, which take seconds, with urine PCR held in reserve to confirm positive swabs.
To streamline the process, a standing order was added to the electronic records system so nurses could order saliva PCRs without having to get physician approval. “I think [that] was one of the biggest things that’s helped us,” Dr. Chauhan said.
Children who test positive must have urine confirmation within 21 days of birth; most are long gone from the hospital by then and have to be called back in. “We haven’t lost anyone to follow-up, but it can be stressful trying to get someone to come back,” she said.
Six of 449 infants have screened positive on saliva – three were false positives with negative urine screens. Of the three confirmed cases, two infants later turned out to have normal hearing on repeat testing and were otherwise asymptomatic.
These days, Dr. Chauhan said, if children have a positive saliva PCR but later turn out to have normal hearing, and are otherwise free of symptoms with no CMV risk factors, “we are not confirming with urine.”
Dr. Chauhan did not have any disclosures. No funding source was mentioned.
SOURCE: Chauhan A et al. PAS 2019. Abstract 306
REPORTING FROM PAS 2019
Key clinical point: A metropolitan N.Y. health system provides a model for how to implement cytomegalovirus screening for infants who fail hearing tests.
Major finding: .
Study details: Pre-post quality improvement project.
Disclosures: The lead investigator had no disclosures. No funding source was mentioned.
Source: Chauhan A et al. PAS 2019. Abstract 306.
A gentler approach to gastroschisis improves outcomes
BALTIMORE – a condition in which infants are born with their intestines and sometimes other organs protruding through a hole beside the umbilicus.
Neonatologists, maternal-fetal health experts, and pediatric surgeons standardized a literature-based approach that was gentler and less invasive than usual management, emphasizing sutureless closure, sometimes at bedside on the first day of life, and early feeding. Often, it turned out, that’s all that children require.
It’s made a big difference. “We reduced the number of trips to the operating room and exposure to general anesthesia. We reduced the number of babies intubated and days on the ventilator. We reduced opioid days and antibiotic days” without increasing bacteremia, and “there are probably long-term benefits beyond the NICU,” said Kara Calkins, MD, at the Pediatric Academic Societies annual meeting.
I think this is definitely ahead of the curve for NICUs. My hope is that the vast majority of universities adopt a similar approach,” said Dr. Calkins, who is an assistant professor of neonatology at the University of California, Los Angeles.
“When I was a fellow,” she explained, “we took all of these babies and intubated them right away and put them on a drip to paralyze and sedate them. We put their bowels into a silo,” essentially a plastic bag suspended by a string, in the hopes that gravity would pull the bowels back into the abdomen. More often than not, however, “the surgeon would come by every day and slowly push them” back in over a week or so. “The fear was if you did it too quickly, you’d invoke an abdominal compartment syndrome, or respiratory decompensation. You had a baby intubated for a week, sedated and paralyzed.”
Infants were kept on total parenteral nutrition for weeks, sometimes through a Broviac central catheter.
It was overkill, Dr. Calkins said, when only the intestines are out and the abdominal wall defect isn’t too large or too small, which is the case for many infants.
For those children, sutureless closure over 1-3 days is the new goal. The bowel is worked back into the abdomen and the umbilical cord is pulled to the side to approximate the edges of the wound, and tacked down; the defect then heals itself. Antibiotics are discontinued 48 hours after closure. Gastric and rectal decompression helps with reduction.
Also, “we give drops of breast milk in their cheek right away, every couple of hours starting on the first day of life. Once the output from the gastric tube is clear, we start feeds. We still give total parenteral nutrition, but through a [peripherally inserted central catheter] in the arm,” Dr. Calkins said. “Use of breast milk for this population is important” to help establish a healthy microbiome, among other reasons.
Another improvement that had been made, according to Dr. Calkins, is that if only the intestines are out, women carry their baby to term and deliver vaginally. The old practice was to deliver babies preterm by Cesarean section, she explained.
To see how it’s worked out, Dr. Calkins and her colleagues reviewed 70 gastroschisis cases managed under the new guidelines. They were uncomplicated cases, with no intestinal atresia, stricture, or ischemia.
Paralysis was avoided for silo placement in 53 infants (76%) and 32 (46%) avoided intubation. Antibiotics were discontinued in 56 (80%) within 48 hours of abdominal wall closure, and routine narcotics were discontinued in 53 infants (76%). Feeds were initiated in almost all children within 48 hours of non-bilious gastric tube output.
Compared with 168 infants treated before the changes were made, silo placement dropped from 71% to 58% of infants, and total ventilator days from a median of 5 to 2.
There was no difference in length of stay, perhaps because the “intestinal dysmotility intrinsic to gastroschisis remains a rate limiting factor for discharge,” the team concluded.
There was no industry funding, and Dr. Calkins didn’t have any disclosures.
SOURCE: Rottkamp CA et al., PAS 2019. Abstract 51.
BALTIMORE – a condition in which infants are born with their intestines and sometimes other organs protruding through a hole beside the umbilicus.
Neonatologists, maternal-fetal health experts, and pediatric surgeons standardized a literature-based approach that was gentler and less invasive than usual management, emphasizing sutureless closure, sometimes at bedside on the first day of life, and early feeding. Often, it turned out, that’s all that children require.
It’s made a big difference. “We reduced the number of trips to the operating room and exposure to general anesthesia. We reduced the number of babies intubated and days on the ventilator. We reduced opioid days and antibiotic days” without increasing bacteremia, and “there are probably long-term benefits beyond the NICU,” said Kara Calkins, MD, at the Pediatric Academic Societies annual meeting.
I think this is definitely ahead of the curve for NICUs. My hope is that the vast majority of universities adopt a similar approach,” said Dr. Calkins, who is an assistant professor of neonatology at the University of California, Los Angeles.
“When I was a fellow,” she explained, “we took all of these babies and intubated them right away and put them on a drip to paralyze and sedate them. We put their bowels into a silo,” essentially a plastic bag suspended by a string, in the hopes that gravity would pull the bowels back into the abdomen. More often than not, however, “the surgeon would come by every day and slowly push them” back in over a week or so. “The fear was if you did it too quickly, you’d invoke an abdominal compartment syndrome, or respiratory decompensation. You had a baby intubated for a week, sedated and paralyzed.”
Infants were kept on total parenteral nutrition for weeks, sometimes through a Broviac central catheter.
It was overkill, Dr. Calkins said, when only the intestines are out and the abdominal wall defect isn’t too large or too small, which is the case for many infants.
For those children, sutureless closure over 1-3 days is the new goal. The bowel is worked back into the abdomen and the umbilical cord is pulled to the side to approximate the edges of the wound, and tacked down; the defect then heals itself. Antibiotics are discontinued 48 hours after closure. Gastric and rectal decompression helps with reduction.
Also, “we give drops of breast milk in their cheek right away, every couple of hours starting on the first day of life. Once the output from the gastric tube is clear, we start feeds. We still give total parenteral nutrition, but through a [peripherally inserted central catheter] in the arm,” Dr. Calkins said. “Use of breast milk for this population is important” to help establish a healthy microbiome, among other reasons.
Another improvement that had been made, according to Dr. Calkins, is that if only the intestines are out, women carry their baby to term and deliver vaginally. The old practice was to deliver babies preterm by Cesarean section, she explained.
To see how it’s worked out, Dr. Calkins and her colleagues reviewed 70 gastroschisis cases managed under the new guidelines. They were uncomplicated cases, with no intestinal atresia, stricture, or ischemia.
Paralysis was avoided for silo placement in 53 infants (76%) and 32 (46%) avoided intubation. Antibiotics were discontinued in 56 (80%) within 48 hours of abdominal wall closure, and routine narcotics were discontinued in 53 infants (76%). Feeds were initiated in almost all children within 48 hours of non-bilious gastric tube output.
Compared with 168 infants treated before the changes were made, silo placement dropped from 71% to 58% of infants, and total ventilator days from a median of 5 to 2.
There was no difference in length of stay, perhaps because the “intestinal dysmotility intrinsic to gastroschisis remains a rate limiting factor for discharge,” the team concluded.
There was no industry funding, and Dr. Calkins didn’t have any disclosures.
SOURCE: Rottkamp CA et al., PAS 2019. Abstract 51.
BALTIMORE – a condition in which infants are born with their intestines and sometimes other organs protruding through a hole beside the umbilicus.
Neonatologists, maternal-fetal health experts, and pediatric surgeons standardized a literature-based approach that was gentler and less invasive than usual management, emphasizing sutureless closure, sometimes at bedside on the first day of life, and early feeding. Often, it turned out, that’s all that children require.
It’s made a big difference. “We reduced the number of trips to the operating room and exposure to general anesthesia. We reduced the number of babies intubated and days on the ventilator. We reduced opioid days and antibiotic days” without increasing bacteremia, and “there are probably long-term benefits beyond the NICU,” said Kara Calkins, MD, at the Pediatric Academic Societies annual meeting.
I think this is definitely ahead of the curve for NICUs. My hope is that the vast majority of universities adopt a similar approach,” said Dr. Calkins, who is an assistant professor of neonatology at the University of California, Los Angeles.
“When I was a fellow,” she explained, “we took all of these babies and intubated them right away and put them on a drip to paralyze and sedate them. We put their bowels into a silo,” essentially a plastic bag suspended by a string, in the hopes that gravity would pull the bowels back into the abdomen. More often than not, however, “the surgeon would come by every day and slowly push them” back in over a week or so. “The fear was if you did it too quickly, you’d invoke an abdominal compartment syndrome, or respiratory decompensation. You had a baby intubated for a week, sedated and paralyzed.”
Infants were kept on total parenteral nutrition for weeks, sometimes through a Broviac central catheter.
It was overkill, Dr. Calkins said, when only the intestines are out and the abdominal wall defect isn’t too large or too small, which is the case for many infants.
For those children, sutureless closure over 1-3 days is the new goal. The bowel is worked back into the abdomen and the umbilical cord is pulled to the side to approximate the edges of the wound, and tacked down; the defect then heals itself. Antibiotics are discontinued 48 hours after closure. Gastric and rectal decompression helps with reduction.
Also, “we give drops of breast milk in their cheek right away, every couple of hours starting on the first day of life. Once the output from the gastric tube is clear, we start feeds. We still give total parenteral nutrition, but through a [peripherally inserted central catheter] in the arm,” Dr. Calkins said. “Use of breast milk for this population is important” to help establish a healthy microbiome, among other reasons.
Another improvement that had been made, according to Dr. Calkins, is that if only the intestines are out, women carry their baby to term and deliver vaginally. The old practice was to deliver babies preterm by Cesarean section, she explained.
To see how it’s worked out, Dr. Calkins and her colleagues reviewed 70 gastroschisis cases managed under the new guidelines. They were uncomplicated cases, with no intestinal atresia, stricture, or ischemia.
Paralysis was avoided for silo placement in 53 infants (76%) and 32 (46%) avoided intubation. Antibiotics were discontinued in 56 (80%) within 48 hours of abdominal wall closure, and routine narcotics were discontinued in 53 infants (76%). Feeds were initiated in almost all children within 48 hours of non-bilious gastric tube output.
Compared with 168 infants treated before the changes were made, silo placement dropped from 71% to 58% of infants, and total ventilator days from a median of 5 to 2.
There was no difference in length of stay, perhaps because the “intestinal dysmotility intrinsic to gastroschisis remains a rate limiting factor for discharge,” the team concluded.
There was no industry funding, and Dr. Calkins didn’t have any disclosures.
SOURCE: Rottkamp CA et al., PAS 2019. Abstract 51.
REPORTING FROM PAS 2019
Marijuana during prenatal OUD treatment increases premature birth
BALTIMORE – Marijuana is a not a good idea during pregnancy, and it’s an even worse idea when women are being treated for opioid addiction, according to an investigation from East Tennessee State University, Mountain Home.
Marijuana use may become more common as legalization rolls out across the country, and legalization, in turn, may add to the perception that pot is harmless, and maybe a good way to take the edge off during pregnancy and prevent morning sickness, said neonatologist Darshan Shaw, MD, of the department of pediatrics at the university.
Dr. Shaw wondered how that trend might impact treatment of opioid use disorder (OUD) during pregnancy, which has also become more common. The take-home is that “if you have a pregnant patient on medically assistant therapy” for opioid addition, “you should warn them against use of marijuana. It increases the risk of prematurity and low birth weight,” he said at the Pediatric Academic Societies annual meeting.
He and his team reviewed 2,375 opioid-exposed pregnancies at six hospitals in south-central Appalachia from July 2011 to June 2016. All of the women had used opioids during pregnancy, some illegally and others for opioid use disorder (OUD) treatment or other medical issues; 108 had urine screens that were positive for tetrahydrocannabinol (THC) at the time of delivery.
Infants were born a mean of 3 days earlier in the marijuana group, and a mean of 265 g lighter. They were also more likely to be born before 37 weeks’ gestation (14% versus 6.5%); born weighing less than 2,500 g (17.6% versus 7.3%); and more likely to be admitted to the neonatal ICU (17.5% versus 7.1%).
On logistic regression to control for parity, maternal status, and tobacco and benzodiazepine use, prenatal marijuana exposure more than doubled the risk of prematurity (odds ratio, 2.35; 95% confidence interval, 1.3-4.23); tobacco and benzodiazepines did not increase the risk. Marijuana also doubled the risk of low birth weight (OR, 2.02; 95% CI, 1.18-3.47), about the same as tobacco and benzodiazepines.
The study had limitations. There was no controlling for a major confounder: the amount of opioids woman took while pregnant. These data were not available, Dr. Shaw said.
Neonatal abstinence syndrome was more common in the marijuana group (33.3% versus 18.1%), so it’s possible that women who used marijuana also used more opioids. “We suspect that opioid exposure was not uniform among all infants,” he said. There were also no data on the amount or way marijuana was used.
Marijuana-positive women were more likely to be unmarried, nulliparous, and use tobacco and benzodiazepines.
There was no industry funding for the work, and Dr. Shaw had no disclosures.
BALTIMORE – Marijuana is a not a good idea during pregnancy, and it’s an even worse idea when women are being treated for opioid addiction, according to an investigation from East Tennessee State University, Mountain Home.
Marijuana use may become more common as legalization rolls out across the country, and legalization, in turn, may add to the perception that pot is harmless, and maybe a good way to take the edge off during pregnancy and prevent morning sickness, said neonatologist Darshan Shaw, MD, of the department of pediatrics at the university.
Dr. Shaw wondered how that trend might impact treatment of opioid use disorder (OUD) during pregnancy, which has also become more common. The take-home is that “if you have a pregnant patient on medically assistant therapy” for opioid addition, “you should warn them against use of marijuana. It increases the risk of prematurity and low birth weight,” he said at the Pediatric Academic Societies annual meeting.
He and his team reviewed 2,375 opioid-exposed pregnancies at six hospitals in south-central Appalachia from July 2011 to June 2016. All of the women had used opioids during pregnancy, some illegally and others for opioid use disorder (OUD) treatment or other medical issues; 108 had urine screens that were positive for tetrahydrocannabinol (THC) at the time of delivery.
Infants were born a mean of 3 days earlier in the marijuana group, and a mean of 265 g lighter. They were also more likely to be born before 37 weeks’ gestation (14% versus 6.5%); born weighing less than 2,500 g (17.6% versus 7.3%); and more likely to be admitted to the neonatal ICU (17.5% versus 7.1%).
On logistic regression to control for parity, maternal status, and tobacco and benzodiazepine use, prenatal marijuana exposure more than doubled the risk of prematurity (odds ratio, 2.35; 95% confidence interval, 1.3-4.23); tobacco and benzodiazepines did not increase the risk. Marijuana also doubled the risk of low birth weight (OR, 2.02; 95% CI, 1.18-3.47), about the same as tobacco and benzodiazepines.
The study had limitations. There was no controlling for a major confounder: the amount of opioids woman took while pregnant. These data were not available, Dr. Shaw said.
Neonatal abstinence syndrome was more common in the marijuana group (33.3% versus 18.1%), so it’s possible that women who used marijuana also used more opioids. “We suspect that opioid exposure was not uniform among all infants,” he said. There were also no data on the amount or way marijuana was used.
Marijuana-positive women were more likely to be unmarried, nulliparous, and use tobacco and benzodiazepines.
There was no industry funding for the work, and Dr. Shaw had no disclosures.
BALTIMORE – Marijuana is a not a good idea during pregnancy, and it’s an even worse idea when women are being treated for opioid addiction, according to an investigation from East Tennessee State University, Mountain Home.
Marijuana use may become more common as legalization rolls out across the country, and legalization, in turn, may add to the perception that pot is harmless, and maybe a good way to take the edge off during pregnancy and prevent morning sickness, said neonatologist Darshan Shaw, MD, of the department of pediatrics at the university.
Dr. Shaw wondered how that trend might impact treatment of opioid use disorder (OUD) during pregnancy, which has also become more common. The take-home is that “if you have a pregnant patient on medically assistant therapy” for opioid addition, “you should warn them against use of marijuana. It increases the risk of prematurity and low birth weight,” he said at the Pediatric Academic Societies annual meeting.
He and his team reviewed 2,375 opioid-exposed pregnancies at six hospitals in south-central Appalachia from July 2011 to June 2016. All of the women had used opioids during pregnancy, some illegally and others for opioid use disorder (OUD) treatment or other medical issues; 108 had urine screens that were positive for tetrahydrocannabinol (THC) at the time of delivery.
Infants were born a mean of 3 days earlier in the marijuana group, and a mean of 265 g lighter. They were also more likely to be born before 37 weeks’ gestation (14% versus 6.5%); born weighing less than 2,500 g (17.6% versus 7.3%); and more likely to be admitted to the neonatal ICU (17.5% versus 7.1%).
On logistic regression to control for parity, maternal status, and tobacco and benzodiazepine use, prenatal marijuana exposure more than doubled the risk of prematurity (odds ratio, 2.35; 95% confidence interval, 1.3-4.23); tobacco and benzodiazepines did not increase the risk. Marijuana also doubled the risk of low birth weight (OR, 2.02; 95% CI, 1.18-3.47), about the same as tobacco and benzodiazepines.
The study had limitations. There was no controlling for a major confounder: the amount of opioids woman took while pregnant. These data were not available, Dr. Shaw said.
Neonatal abstinence syndrome was more common in the marijuana group (33.3% versus 18.1%), so it’s possible that women who used marijuana also used more opioids. “We suspect that opioid exposure was not uniform among all infants,” he said. There were also no data on the amount or way marijuana was used.
Marijuana-positive women were more likely to be unmarried, nulliparous, and use tobacco and benzodiazepines.
There was no industry funding for the work, and Dr. Shaw had no disclosures.
REPORTING FROM PAS 2019
Key clinical point: Warn pregnant women being treated for opioid use disorder to stay away from marijuana.
Major finding: Marijuana use more than doubled the risk of prematurity and low birth weight.
Study details: Review of 2,375 opioid-exposed pregnancies at six hospitals
Disclosures: There was no industry funding for the work, and the lead investigator had no disclosures.