User login
COVID-19: When health care personnel become patients
according to the Centers for Disease Control and Prevention.
That number, however, is probably an underestimation because health care personnel (HCP) status was available for just over 49,000 of the 315,000 COVID-19 cases reported to the CDC as of April 9. Of the cases with known HCP status, 9,282 (19%) were health care personnel, Matthew J. Stuckey, PhD, and the CDC’s COVID-19 Response Team said.
“The number of cases in HCP reported here must be considered a lower bound because additional cases likely have gone unidentified or unreported,” they said.
The median age of the nearly 9,300 HCP with COVID-19 was 42 years, and the majority (55%) were aged 16-44 years; another 21% were 45-54, 18% were 55-64, and 6% were age 65 and over. The oldest group, however, represented 10 of the 27 known HCP deaths, the investigators reported in the Morbidity and Mortality Weekly Report.
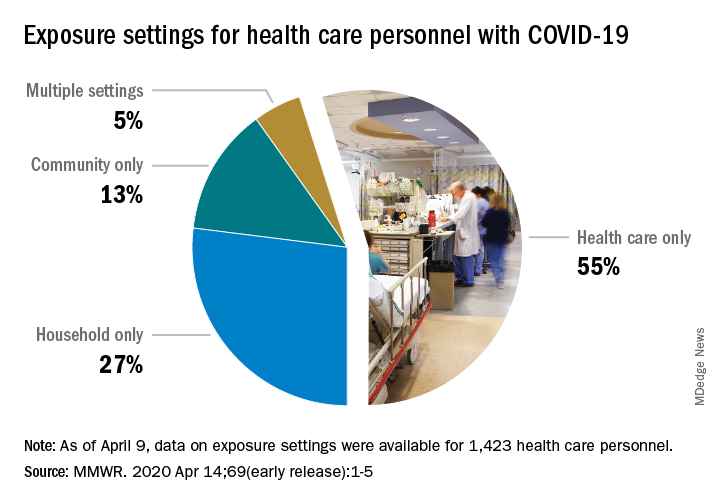
The majority of infected HCP (55%) reported exposure to a COVID-19 patient in the health care setting, but “there were also known exposures in households and in the community, highlighting the potential for exposure in multiple settings, especially as community transmission increases,” the response team said.
Since “contact tracing after recognized occupational exposures likely will fail to identify many HCP at risk for developing COVID-19,” other measures will probably be needed to “reduce the risk for infected HCP transmitting the virus to colleagues and patients,” they added.
HCP with COVID-19 were less likely to be hospitalized (8%-10%) than the overall population (21%-31%), which “might reflect the younger median age … of HCP patients, compared with that of reported COVID-19 patients overall, as well as prioritization of HCP for testing, which might identify less-severe illness,” the investigators suggested.
The prevalence of underlying conditions in HCP patients, 38%, was the same as all patients with COVID-19, and 92% of the HCP patients presented with fever, cough, or shortness of breath. Two-thirds of all HCP reported muscle aches, and 65% reported headache, the CDC response team noted.
“It is critical to make every effort to ensure the health and safety of this essential national workforce of approximately 18 million HCP, both at work and in the community,” they wrote.
SOURCE: Stuckey MJ et al. MMWR. Apr 14;69(early release):1-5.
according to the Centers for Disease Control and Prevention.

That number, however, is probably an underestimation because health care personnel (HCP) status was available for just over 49,000 of the 315,000 COVID-19 cases reported to the CDC as of April 9. Of the cases with known HCP status, 9,282 (19%) were health care personnel, Matthew J. Stuckey, PhD, and the CDC’s COVID-19 Response Team said.
“The number of cases in HCP reported here must be considered a lower bound because additional cases likely have gone unidentified or unreported,” they said.
The median age of the nearly 9,300 HCP with COVID-19 was 42 years, and the majority (55%) were aged 16-44 years; another 21% were 45-54, 18% were 55-64, and 6% were age 65 and over. The oldest group, however, represented 10 of the 27 known HCP deaths, the investigators reported in the Morbidity and Mortality Weekly Report.
The majority of infected HCP (55%) reported exposure to a COVID-19 patient in the health care setting, but “there were also known exposures in households and in the community, highlighting the potential for exposure in multiple settings, especially as community transmission increases,” the response team said.
Since “contact tracing after recognized occupational exposures likely will fail to identify many HCP at risk for developing COVID-19,” other measures will probably be needed to “reduce the risk for infected HCP transmitting the virus to colleagues and patients,” they added.
HCP with COVID-19 were less likely to be hospitalized (8%-10%) than the overall population (21%-31%), which “might reflect the younger median age … of HCP patients, compared with that of reported COVID-19 patients overall, as well as prioritization of HCP for testing, which might identify less-severe illness,” the investigators suggested.
The prevalence of underlying conditions in HCP patients, 38%, was the same as all patients with COVID-19, and 92% of the HCP patients presented with fever, cough, or shortness of breath. Two-thirds of all HCP reported muscle aches, and 65% reported headache, the CDC response team noted.
“It is critical to make every effort to ensure the health and safety of this essential national workforce of approximately 18 million HCP, both at work and in the community,” they wrote.
SOURCE: Stuckey MJ et al. MMWR. Apr 14;69(early release):1-5.
according to the Centers for Disease Control and Prevention.

That number, however, is probably an underestimation because health care personnel (HCP) status was available for just over 49,000 of the 315,000 COVID-19 cases reported to the CDC as of April 9. Of the cases with known HCP status, 9,282 (19%) were health care personnel, Matthew J. Stuckey, PhD, and the CDC’s COVID-19 Response Team said.
“The number of cases in HCP reported here must be considered a lower bound because additional cases likely have gone unidentified or unreported,” they said.
The median age of the nearly 9,300 HCP with COVID-19 was 42 years, and the majority (55%) were aged 16-44 years; another 21% were 45-54, 18% were 55-64, and 6% were age 65 and over. The oldest group, however, represented 10 of the 27 known HCP deaths, the investigators reported in the Morbidity and Mortality Weekly Report.
The majority of infected HCP (55%) reported exposure to a COVID-19 patient in the health care setting, but “there were also known exposures in households and in the community, highlighting the potential for exposure in multiple settings, especially as community transmission increases,” the response team said.
Since “contact tracing after recognized occupational exposures likely will fail to identify many HCP at risk for developing COVID-19,” other measures will probably be needed to “reduce the risk for infected HCP transmitting the virus to colleagues and patients,” they added.
HCP with COVID-19 were less likely to be hospitalized (8%-10%) than the overall population (21%-31%), which “might reflect the younger median age … of HCP patients, compared with that of reported COVID-19 patients overall, as well as prioritization of HCP for testing, which might identify less-severe illness,” the investigators suggested.
The prevalence of underlying conditions in HCP patients, 38%, was the same as all patients with COVID-19, and 92% of the HCP patients presented with fever, cough, or shortness of breath. Two-thirds of all HCP reported muscle aches, and 65% reported headache, the CDC response team noted.
“It is critical to make every effort to ensure the health and safety of this essential national workforce of approximately 18 million HCP, both at work and in the community,” they wrote.
SOURCE: Stuckey MJ et al. MMWR. Apr 14;69(early release):1-5.
FROM THE MMWR
The role of FOAM and social networks in COVID-19
“Uncertainty creates weakness. Uncertainty makes one tentative, if not fearful, and tentative steps, even when in the right direction, may not overcome significant obstacles.”1
Recently, I spent my vacation time quarantined reading “The Great Influenza,” which recounts the history of the 1918 pandemic. Despite over a century of scientific and medical progress, the parallels to our current situation are indisputable. Just as in 1918, we are limiting social gatherings, quarantining, wearing face masks, and living with the fear and anxiety of keeping ourselves and our families safe. In 1918, use of aspirin, quinine, and digitalis therapies in a desperate search for relief despite limited evidence mirror the current use of hydroxychloroquine, azithromycin, and lopinavir/ritonavir. While there are many similarities between the two situations, in this pandemic our channels for dissemination of scientific literature are better developed, and online networks are enabling physicians across the globe to communicate their experience and findings in near real time.
During this time of uncertainty, our understanding of COVID-19 evolves daily. Without the advantage of robust randomized, controlled trials and large-scale studies to guide us, we are forced to rely on pattern recognition for surveillance and anecdotal or limited case-based accounts to guide clinical care. Fortunately, free open-access medical education (FOAM) and social networks offer a significant advantage in our ability to collect and disseminate information.
Free open access medical education
The concept of FOAM started in 2012 with the intent of creating a collaborative and constantly evolving community to provide open-access medical education. It encompasses multiple platforms – blogs, podcasts, videos, and social media – and features content experts from across the globe. Since its inception, FOAM has grown in popularity and use, especially within emergency medicine and critical care communities, as an adjunct for asynchronous learning.2,3
In a time where knowledge of COVID-19 is dynamically changing, traditional sources like textbooks, journals, and organizational guidelines often lag behind real-time clinical experience and needs. Additionally, many clinicians are now being tasked with taking care of patient populations and a new critical illness profile with which they are not comfortable. It is challenging to find a well-curated and updated repository of information to answer questions surrounding pathophysiology, critical care, ventilator management, caring for adult patients, and personal protective equipment (PPE). During this rapidly evolving reality, FOAM is becoming the ideal modality for timely and efficient sharing of reviews of current literature, expert discussions, and clinical practice guidelines.
A few self-directed hours on EMCrit’s Internet Book of Critical Care’s COVID-19 chapter reveals a bastion of content regarding diagnosis, pathophysiology, transmission, therapies, and ventilator strategies.4 It includes references to major journals and recommendations from international societies. Websites like EMCrit and REBEL EM are updated daily with podcasts, videos, and blog posts surrounding the latest highly debated topics in COVID-19 management.5 Podcasts like EM:RAP and Peds RAP have made COVID segments discussing important topics like pharmacotherapy, telemedicine, and pregnancy available for free.6,7 Many networks, institutions, and individual physicians have created and posted videos online on critical care topics and refreshers.
Social networks
Online social networks composed of international physicians within Facebook and LinkedIn serve as miniature publishing houses. First-hand accounts of patient presentations and patient care act as case reports. As similar accounts accumulate, they become case series. Patterns emerge and new hypotheses are generated, debated, and critiqued through this informal peer review. Personal accounts of frustration with lack of PPE, fear of exposing loved ones, distress at being separated from family, and grief of witnessing multiple patients die alone are opinion and perspective articles.
These networks offer the space for sharing. Those who have had the experience of caring for the surge of COVID-19 patients offer advice and words of caution to those who have yet to experience it. Protocols from a multitude of institutions on triage, surge, disposition, and end-of-life care are disseminated, serving as templates for those that have not yet developed their own. There is an impressive variety of innovative, do-it-yourself projects surrounding PPE, intubation boxes, and three-dimensionally printed ventilator parts.
Finally, these networks provide emotional support. There are offers to ship additional PPE, videos of cities cheering as clinicians go to work, stories of triumph and recovery, pictures depicting ongoing wellness activities, and the occasional much-needed humorous anecdote or illustration. These networks reinforce the message that our lives continue despite this upheaval, and we are not alone in this struggle.
The end of the passage in The Great Influenza concludes with: “Ultimately a scientist has nothing to believe in but the process of inquiry. To move forcefully and aggressively even while uncertain requires a confidence and strength deeper than physical courage.”
They represent a highly adaptable, evolving, and collaborative global community’s determination to persevere through time of uncertainty together.
Dr. Ren is a pediatric emergency medicine fellow at Children’s National Hospital, Washington. Dr. Simpson is a pediatric emergency medicine attending and medical director of emergency preparedness at the hospital. They reported that they do not have any disclosures or conflicts of interest. Email Dr. Ren and Dr. Simpson at [email protected].
References
1. “The Great Influenza: The Story of the Deadliest Pandemic in History.” (New York: Penguin Books, 2005, pp. 261-62).
2. Emerg Med J. 2014 Oct;31(e1):e76-7.
3. Acad Med. 2014 Apr;89(4):598-601.
4. “The Internet Book of Critical Care: COVID-19.” EMCrit Project.
5. “Covid-19.” REBEL EM-Emergency Medicine Blog.
6. “EM:RAP COVID-19 Resources.” EM RAP: Emergency Medicine Reviews and Perspectives.
7. “Episodes.” Peds RAP, Hippo Education.
“Uncertainty creates weakness. Uncertainty makes one tentative, if not fearful, and tentative steps, even when in the right direction, may not overcome significant obstacles.”1
Recently, I spent my vacation time quarantined reading “The Great Influenza,” which recounts the history of the 1918 pandemic. Despite over a century of scientific and medical progress, the parallels to our current situation are indisputable. Just as in 1918, we are limiting social gatherings, quarantining, wearing face masks, and living with the fear and anxiety of keeping ourselves and our families safe. In 1918, use of aspirin, quinine, and digitalis therapies in a desperate search for relief despite limited evidence mirror the current use of hydroxychloroquine, azithromycin, and lopinavir/ritonavir. While there are many similarities between the two situations, in this pandemic our channels for dissemination of scientific literature are better developed, and online networks are enabling physicians across the globe to communicate their experience and findings in near real time.
During this time of uncertainty, our understanding of COVID-19 evolves daily. Without the advantage of robust randomized, controlled trials and large-scale studies to guide us, we are forced to rely on pattern recognition for surveillance and anecdotal or limited case-based accounts to guide clinical care. Fortunately, free open-access medical education (FOAM) and social networks offer a significant advantage in our ability to collect and disseminate information.
Free open access medical education
The concept of FOAM started in 2012 with the intent of creating a collaborative and constantly evolving community to provide open-access medical education. It encompasses multiple platforms – blogs, podcasts, videos, and social media – and features content experts from across the globe. Since its inception, FOAM has grown in popularity and use, especially within emergency medicine and critical care communities, as an adjunct for asynchronous learning.2,3
In a time where knowledge of COVID-19 is dynamically changing, traditional sources like textbooks, journals, and organizational guidelines often lag behind real-time clinical experience and needs. Additionally, many clinicians are now being tasked with taking care of patient populations and a new critical illness profile with which they are not comfortable. It is challenging to find a well-curated and updated repository of information to answer questions surrounding pathophysiology, critical care, ventilator management, caring for adult patients, and personal protective equipment (PPE). During this rapidly evolving reality, FOAM is becoming the ideal modality for timely and efficient sharing of reviews of current literature, expert discussions, and clinical practice guidelines.
A few self-directed hours on EMCrit’s Internet Book of Critical Care’s COVID-19 chapter reveals a bastion of content regarding diagnosis, pathophysiology, transmission, therapies, and ventilator strategies.4 It includes references to major journals and recommendations from international societies. Websites like EMCrit and REBEL EM are updated daily with podcasts, videos, and blog posts surrounding the latest highly debated topics in COVID-19 management.5 Podcasts like EM:RAP and Peds RAP have made COVID segments discussing important topics like pharmacotherapy, telemedicine, and pregnancy available for free.6,7 Many networks, institutions, and individual physicians have created and posted videos online on critical care topics and refreshers.
Social networks
Online social networks composed of international physicians within Facebook and LinkedIn serve as miniature publishing houses. First-hand accounts of patient presentations and patient care act as case reports. As similar accounts accumulate, they become case series. Patterns emerge and new hypotheses are generated, debated, and critiqued through this informal peer review. Personal accounts of frustration with lack of PPE, fear of exposing loved ones, distress at being separated from family, and grief of witnessing multiple patients die alone are opinion and perspective articles.
These networks offer the space for sharing. Those who have had the experience of caring for the surge of COVID-19 patients offer advice and words of caution to those who have yet to experience it. Protocols from a multitude of institutions on triage, surge, disposition, and end-of-life care are disseminated, serving as templates for those that have not yet developed their own. There is an impressive variety of innovative, do-it-yourself projects surrounding PPE, intubation boxes, and three-dimensionally printed ventilator parts.
Finally, these networks provide emotional support. There are offers to ship additional PPE, videos of cities cheering as clinicians go to work, stories of triumph and recovery, pictures depicting ongoing wellness activities, and the occasional much-needed humorous anecdote or illustration. These networks reinforce the message that our lives continue despite this upheaval, and we are not alone in this struggle.
The end of the passage in The Great Influenza concludes with: “Ultimately a scientist has nothing to believe in but the process of inquiry. To move forcefully and aggressively even while uncertain requires a confidence and strength deeper than physical courage.”
They represent a highly adaptable, evolving, and collaborative global community’s determination to persevere through time of uncertainty together.
Dr. Ren is a pediatric emergency medicine fellow at Children’s National Hospital, Washington. Dr. Simpson is a pediatric emergency medicine attending and medical director of emergency preparedness at the hospital. They reported that they do not have any disclosures or conflicts of interest. Email Dr. Ren and Dr. Simpson at [email protected].
References
1. “The Great Influenza: The Story of the Deadliest Pandemic in History.” (New York: Penguin Books, 2005, pp. 261-62).
2. Emerg Med J. 2014 Oct;31(e1):e76-7.
3. Acad Med. 2014 Apr;89(4):598-601.
4. “The Internet Book of Critical Care: COVID-19.” EMCrit Project.
5. “Covid-19.” REBEL EM-Emergency Medicine Blog.
6. “EM:RAP COVID-19 Resources.” EM RAP: Emergency Medicine Reviews and Perspectives.
7. “Episodes.” Peds RAP, Hippo Education.
“Uncertainty creates weakness. Uncertainty makes one tentative, if not fearful, and tentative steps, even when in the right direction, may not overcome significant obstacles.”1
Recently, I spent my vacation time quarantined reading “The Great Influenza,” which recounts the history of the 1918 pandemic. Despite over a century of scientific and medical progress, the parallels to our current situation are indisputable. Just as in 1918, we are limiting social gatherings, quarantining, wearing face masks, and living with the fear and anxiety of keeping ourselves and our families safe. In 1918, use of aspirin, quinine, and digitalis therapies in a desperate search for relief despite limited evidence mirror the current use of hydroxychloroquine, azithromycin, and lopinavir/ritonavir. While there are many similarities between the two situations, in this pandemic our channels for dissemination of scientific literature are better developed, and online networks are enabling physicians across the globe to communicate their experience and findings in near real time.
During this time of uncertainty, our understanding of COVID-19 evolves daily. Without the advantage of robust randomized, controlled trials and large-scale studies to guide us, we are forced to rely on pattern recognition for surveillance and anecdotal or limited case-based accounts to guide clinical care. Fortunately, free open-access medical education (FOAM) and social networks offer a significant advantage in our ability to collect and disseminate information.
Free open access medical education
The concept of FOAM started in 2012 with the intent of creating a collaborative and constantly evolving community to provide open-access medical education. It encompasses multiple platforms – blogs, podcasts, videos, and social media – and features content experts from across the globe. Since its inception, FOAM has grown in popularity and use, especially within emergency medicine and critical care communities, as an adjunct for asynchronous learning.2,3
In a time where knowledge of COVID-19 is dynamically changing, traditional sources like textbooks, journals, and organizational guidelines often lag behind real-time clinical experience and needs. Additionally, many clinicians are now being tasked with taking care of patient populations and a new critical illness profile with which they are not comfortable. It is challenging to find a well-curated and updated repository of information to answer questions surrounding pathophysiology, critical care, ventilator management, caring for adult patients, and personal protective equipment (PPE). During this rapidly evolving reality, FOAM is becoming the ideal modality for timely and efficient sharing of reviews of current literature, expert discussions, and clinical practice guidelines.
A few self-directed hours on EMCrit’s Internet Book of Critical Care’s COVID-19 chapter reveals a bastion of content regarding diagnosis, pathophysiology, transmission, therapies, and ventilator strategies.4 It includes references to major journals and recommendations from international societies. Websites like EMCrit and REBEL EM are updated daily with podcasts, videos, and blog posts surrounding the latest highly debated topics in COVID-19 management.5 Podcasts like EM:RAP and Peds RAP have made COVID segments discussing important topics like pharmacotherapy, telemedicine, and pregnancy available for free.6,7 Many networks, institutions, and individual physicians have created and posted videos online on critical care topics and refreshers.
Social networks
Online social networks composed of international physicians within Facebook and LinkedIn serve as miniature publishing houses. First-hand accounts of patient presentations and patient care act as case reports. As similar accounts accumulate, they become case series. Patterns emerge and new hypotheses are generated, debated, and critiqued through this informal peer review. Personal accounts of frustration with lack of PPE, fear of exposing loved ones, distress at being separated from family, and grief of witnessing multiple patients die alone are opinion and perspective articles.
These networks offer the space for sharing. Those who have had the experience of caring for the surge of COVID-19 patients offer advice and words of caution to those who have yet to experience it. Protocols from a multitude of institutions on triage, surge, disposition, and end-of-life care are disseminated, serving as templates for those that have not yet developed their own. There is an impressive variety of innovative, do-it-yourself projects surrounding PPE, intubation boxes, and three-dimensionally printed ventilator parts.
Finally, these networks provide emotional support. There are offers to ship additional PPE, videos of cities cheering as clinicians go to work, stories of triumph and recovery, pictures depicting ongoing wellness activities, and the occasional much-needed humorous anecdote or illustration. These networks reinforce the message that our lives continue despite this upheaval, and we are not alone in this struggle.
The end of the passage in The Great Influenza concludes with: “Ultimately a scientist has nothing to believe in but the process of inquiry. To move forcefully and aggressively even while uncertain requires a confidence and strength deeper than physical courage.”
They represent a highly adaptable, evolving, and collaborative global community’s determination to persevere through time of uncertainty together.
Dr. Ren is a pediatric emergency medicine fellow at Children’s National Hospital, Washington. Dr. Simpson is a pediatric emergency medicine attending and medical director of emergency preparedness at the hospital. They reported that they do not have any disclosures or conflicts of interest. Email Dr. Ren and Dr. Simpson at [email protected].
References
1. “The Great Influenza: The Story of the Deadliest Pandemic in History.” (New York: Penguin Books, 2005, pp. 261-62).
2. Emerg Med J. 2014 Oct;31(e1):e76-7.
3. Acad Med. 2014 Apr;89(4):598-601.
4. “The Internet Book of Critical Care: COVID-19.” EMCrit Project.
5. “Covid-19.” REBEL EM-Emergency Medicine Blog.
6. “EM:RAP COVID-19 Resources.” EM RAP: Emergency Medicine Reviews and Perspectives.
7. “Episodes.” Peds RAP, Hippo Education.
FDA approves emergency use of saliva test to detect COVID-19
As the race to develop rapid testing for COVID-19 expands, the Food and Drug Administration has granted emergency approval for an approach that uses saliva as the primary test biomaterial.
According to a document provided to the FDA, the Rutgers Clinical Genomics Laboratory TaqPath SARS-CoV-2 Assay is intended for the qualitative detection of nucleic acid from SARS-CoV-2 in oropharyngeal (throat) swab, nasopharyngeal swab, anterior nasal swab, mid-turbinate nasal swab from individuals suspected of COVID-19 by their health care clinicians. To expand on this assay, Rutgers University–based RUCDR Infinite Biologics developed a saliva collection method in partnership with Spectrum Solutions and Accurate Diagnostic Labs.
The document states that Samples are transported for RNA extraction and are tested within 48 hours of collection. In saliva samples obtained from 60 patients evaluated by the researchers, all were in agreement with the presence of COVID-19.
“If shown to be as accurate as nasopharyngeal and oropharyngeal samples, saliva as a biomatrix offers the advantage of not generating aerosols or creating as many respiratory droplets during specimen procurement, therefore decreasing the risk of transmission to the health care worker doing the testing,” said Matthew P. Cheng, MDCM, of the division of infectious diseases at McGill University Health Centre, Montreal, who was not involved in development of the test but who has written about diagnostic testing for the virus.
“Also, it may be easy enough for patients to do saliva self-collection at home. However, it is important to note that SARS-CoV-2 tests on saliva have not yet undergone the more rigorous evaluation of full FDA authorization, and saliva is not a preferred specimen type of the FDA nor the [Centers for Disease Control and Prevention] for respiratory virus testing.”
In a prepared statement, Andrew I. Brooks, PhD, chief operating officer at RUCDR Infinite Biologics, said the saliva collection method enables clinicians to preserve personal protective equipment for use in patient care instead of testing. “We can significantly increase the number of people tested each and every day as self-collection of saliva is quicker and more scalable than swab collections,” he said. “All of this combined will have a tremendous impact on testing in New Jersey and across the United States.”
The tests are currently available to the RWJBarnabas Health network, based in West Orange, N.J., which has partnered with Rutgers University.
As the race to develop rapid testing for COVID-19 expands, the Food and Drug Administration has granted emergency approval for an approach that uses saliva as the primary test biomaterial.
According to a document provided to the FDA, the Rutgers Clinical Genomics Laboratory TaqPath SARS-CoV-2 Assay is intended for the qualitative detection of nucleic acid from SARS-CoV-2 in oropharyngeal (throat) swab, nasopharyngeal swab, anterior nasal swab, mid-turbinate nasal swab from individuals suspected of COVID-19 by their health care clinicians. To expand on this assay, Rutgers University–based RUCDR Infinite Biologics developed a saliva collection method in partnership with Spectrum Solutions and Accurate Diagnostic Labs.
The document states that Samples are transported for RNA extraction and are tested within 48 hours of collection. In saliva samples obtained from 60 patients evaluated by the researchers, all were in agreement with the presence of COVID-19.
“If shown to be as accurate as nasopharyngeal and oropharyngeal samples, saliva as a biomatrix offers the advantage of not generating aerosols or creating as many respiratory droplets during specimen procurement, therefore decreasing the risk of transmission to the health care worker doing the testing,” said Matthew P. Cheng, MDCM, of the division of infectious diseases at McGill University Health Centre, Montreal, who was not involved in development of the test but who has written about diagnostic testing for the virus.
“Also, it may be easy enough for patients to do saliva self-collection at home. However, it is important to note that SARS-CoV-2 tests on saliva have not yet undergone the more rigorous evaluation of full FDA authorization, and saliva is not a preferred specimen type of the FDA nor the [Centers for Disease Control and Prevention] for respiratory virus testing.”
In a prepared statement, Andrew I. Brooks, PhD, chief operating officer at RUCDR Infinite Biologics, said the saliva collection method enables clinicians to preserve personal protective equipment for use in patient care instead of testing. “We can significantly increase the number of people tested each and every day as self-collection of saliva is quicker and more scalable than swab collections,” he said. “All of this combined will have a tremendous impact on testing in New Jersey and across the United States.”
The tests are currently available to the RWJBarnabas Health network, based in West Orange, N.J., which has partnered with Rutgers University.
As the race to develop rapid testing for COVID-19 expands, the Food and Drug Administration has granted emergency approval for an approach that uses saliva as the primary test biomaterial.
According to a document provided to the FDA, the Rutgers Clinical Genomics Laboratory TaqPath SARS-CoV-2 Assay is intended for the qualitative detection of nucleic acid from SARS-CoV-2 in oropharyngeal (throat) swab, nasopharyngeal swab, anterior nasal swab, mid-turbinate nasal swab from individuals suspected of COVID-19 by their health care clinicians. To expand on this assay, Rutgers University–based RUCDR Infinite Biologics developed a saliva collection method in partnership with Spectrum Solutions and Accurate Diagnostic Labs.
The document states that Samples are transported for RNA extraction and are tested within 48 hours of collection. In saliva samples obtained from 60 patients evaluated by the researchers, all were in agreement with the presence of COVID-19.
“If shown to be as accurate as nasopharyngeal and oropharyngeal samples, saliva as a biomatrix offers the advantage of not generating aerosols or creating as many respiratory droplets during specimen procurement, therefore decreasing the risk of transmission to the health care worker doing the testing,” said Matthew P. Cheng, MDCM, of the division of infectious diseases at McGill University Health Centre, Montreal, who was not involved in development of the test but who has written about diagnostic testing for the virus.
“Also, it may be easy enough for patients to do saliva self-collection at home. However, it is important to note that SARS-CoV-2 tests on saliva have not yet undergone the more rigorous evaluation of full FDA authorization, and saliva is not a preferred specimen type of the FDA nor the [Centers for Disease Control and Prevention] for respiratory virus testing.”
In a prepared statement, Andrew I. Brooks, PhD, chief operating officer at RUCDR Infinite Biologics, said the saliva collection method enables clinicians to preserve personal protective equipment for use in patient care instead of testing. “We can significantly increase the number of people tested each and every day as self-collection of saliva is quicker and more scalable than swab collections,” he said. “All of this combined will have a tremendous impact on testing in New Jersey and across the United States.”
The tests are currently available to the RWJBarnabas Health network, based in West Orange, N.J., which has partnered with Rutgers University.
Resources for LGBTQ youth during challenging times
If you are anything like me, March 1 came and went as just another first day of the month. Few of us could have imagined that our day-to-day way of life would soon be upended, and our country would be in the midst of the COVID-19 pandemic. While there is considerable anxiety around protecting our individual health, social distancing and the physical isolation that comes from it have cut off a vital source of support for many of our lesbian, gay, bisexual, transgender, and questioning (or queer) (LGBTQ) youth. Shared experiences with other young people like themselves provide these youth with a sense of community that they may not find in their schools, towns, etc.
LGBTQ youth already face increased rates of anxiety and depression compared with their heterosexual and cisgender peers. According to the 2017 Youth Risk Behavior Survey, 63% of LGB youth nationwide reported feeling sad or hopeless compared with 28% of their heterosexual peers. While quarantined at home, many of these youth now are stuck for many more hours per day with families who may not accept them for who they are. Previous research by Ryan et al. shows that LGB adolescents who have higher rates of family rejection are nearly six times more likely to have higher rates of depression and more than eight times more likely to attempt suicide than their peers who come from families with low or no levels of rejection (Pediatrics. 2009 Jan;123[1]:346-52). Going to school for roughly 8 hours a day allows some of these youth an escape from what is otherwise an unpleasant home situation. In addition, educators and other school staff may be among the only allies that a student has in his/her life, and school cancellations remove students from access to these important people.
Due to stay-at-home orders and physical distancing measures, lack of in-person access to medical and psychological care can be distressing for many LGBTQ youth. While many practices have been able to convert to audiovisual telemedicine visits, not all of them have the resources or capability to do so. Consequently, LGBTQ youth may have reduced access to support services that help to bolster their social and emotional health. In addition, many trans youth suffer from physical dysphoria that can make it distressing to see themselves on camera doing teletherapy and so they wish to avoid it for this reason.
This is not to say that everything is bleak. LGBTQ youth can also be resilient in times of stress and worry. “The LGBTQ community has a long history of overcoming adversity and utilizing challenges to build an even stronger sense of community. This pandemic will create yet another opportunity for us to highlight existing health disparities and to support our LGBTQ young people in finding creative responses,” said Heather Newby, LCSW, clinical social worker for the GENECIS (GENder Education and Care Interdisciplinary Support) Program at Children’s Medical Center Dallas. In addition, she reported that many LGBTQ advocacy groups have created excellent online support networks and resources to provide nationwide, regional, and local help.
During these challenging times, there are a number of resources that LGBTQ youth can turn to while trying to maintain their connection to their peers. First, many local LGBTQ service organizations have moved their in-person support groups to a virtual or online platform. Check with your local service organization to see what they are offering during these times. National organizations, such as Gender Spectrum, continue to have online groups as well that youth can participate in. Second, many virtual mental health helplines, such as those through the Trevor Project, remain staffed should LGBTQ youth need to access their services (1-866-488-7386, plus text and chat). They can be reached 24/7 to help those whose mental health has been affected during this pandemic. Third, youth can continue to stay connected to their friends through means such as Zoom, FaceTime, or other virtual audiovisual tools. Lastly, some youth have taken to meeting in school parking lots, mall parking lots, etc., and staying at least 6 feet apart so that they can still see their friends in person.
While the current times may be challenging, they will pass and we will be able to return to those activities that bring us joy. Do not hesitate to reach out if you need help. As Rainer Maria Rilke once said, “In the difficult, we must have our joys, our happiness, our dreams: There against the depth of this background, they stand out, there for the first time we see how beautiful they are.”
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. He has no relevant financial disclosures. Dr. Cooper is on Twitter @teendocmbc. Email him at [email protected].
If you are anything like me, March 1 came and went as just another first day of the month. Few of us could have imagined that our day-to-day way of life would soon be upended, and our country would be in the midst of the COVID-19 pandemic. While there is considerable anxiety around protecting our individual health, social distancing and the physical isolation that comes from it have cut off a vital source of support for many of our lesbian, gay, bisexual, transgender, and questioning (or queer) (LGBTQ) youth. Shared experiences with other young people like themselves provide these youth with a sense of community that they may not find in their schools, towns, etc.
LGBTQ youth already face increased rates of anxiety and depression compared with their heterosexual and cisgender peers. According to the 2017 Youth Risk Behavior Survey, 63% of LGB youth nationwide reported feeling sad or hopeless compared with 28% of their heterosexual peers. While quarantined at home, many of these youth now are stuck for many more hours per day with families who may not accept them for who they are. Previous research by Ryan et al. shows that LGB adolescents who have higher rates of family rejection are nearly six times more likely to have higher rates of depression and more than eight times more likely to attempt suicide than their peers who come from families with low or no levels of rejection (Pediatrics. 2009 Jan;123[1]:346-52). Going to school for roughly 8 hours a day allows some of these youth an escape from what is otherwise an unpleasant home situation. In addition, educators and other school staff may be among the only allies that a student has in his/her life, and school cancellations remove students from access to these important people.
Due to stay-at-home orders and physical distancing measures, lack of in-person access to medical and psychological care can be distressing for many LGBTQ youth. While many practices have been able to convert to audiovisual telemedicine visits, not all of them have the resources or capability to do so. Consequently, LGBTQ youth may have reduced access to support services that help to bolster their social and emotional health. In addition, many trans youth suffer from physical dysphoria that can make it distressing to see themselves on camera doing teletherapy and so they wish to avoid it for this reason.
This is not to say that everything is bleak. LGBTQ youth can also be resilient in times of stress and worry. “The LGBTQ community has a long history of overcoming adversity and utilizing challenges to build an even stronger sense of community. This pandemic will create yet another opportunity for us to highlight existing health disparities and to support our LGBTQ young people in finding creative responses,” said Heather Newby, LCSW, clinical social worker for the GENECIS (GENder Education and Care Interdisciplinary Support) Program at Children’s Medical Center Dallas. In addition, she reported that many LGBTQ advocacy groups have created excellent online support networks and resources to provide nationwide, regional, and local help.
During these challenging times, there are a number of resources that LGBTQ youth can turn to while trying to maintain their connection to their peers. First, many local LGBTQ service organizations have moved their in-person support groups to a virtual or online platform. Check with your local service organization to see what they are offering during these times. National organizations, such as Gender Spectrum, continue to have online groups as well that youth can participate in. Second, many virtual mental health helplines, such as those through the Trevor Project, remain staffed should LGBTQ youth need to access their services (1-866-488-7386, plus text and chat). They can be reached 24/7 to help those whose mental health has been affected during this pandemic. Third, youth can continue to stay connected to their friends through means such as Zoom, FaceTime, or other virtual audiovisual tools. Lastly, some youth have taken to meeting in school parking lots, mall parking lots, etc., and staying at least 6 feet apart so that they can still see their friends in person.
While the current times may be challenging, they will pass and we will be able to return to those activities that bring us joy. Do not hesitate to reach out if you need help. As Rainer Maria Rilke once said, “In the difficult, we must have our joys, our happiness, our dreams: There against the depth of this background, they stand out, there for the first time we see how beautiful they are.”
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. He has no relevant financial disclosures. Dr. Cooper is on Twitter @teendocmbc. Email him at [email protected].
If you are anything like me, March 1 came and went as just another first day of the month. Few of us could have imagined that our day-to-day way of life would soon be upended, and our country would be in the midst of the COVID-19 pandemic. While there is considerable anxiety around protecting our individual health, social distancing and the physical isolation that comes from it have cut off a vital source of support for many of our lesbian, gay, bisexual, transgender, and questioning (or queer) (LGBTQ) youth. Shared experiences with other young people like themselves provide these youth with a sense of community that they may not find in their schools, towns, etc.
LGBTQ youth already face increased rates of anxiety and depression compared with their heterosexual and cisgender peers. According to the 2017 Youth Risk Behavior Survey, 63% of LGB youth nationwide reported feeling sad or hopeless compared with 28% of their heterosexual peers. While quarantined at home, many of these youth now are stuck for many more hours per day with families who may not accept them for who they are. Previous research by Ryan et al. shows that LGB adolescents who have higher rates of family rejection are nearly six times more likely to have higher rates of depression and more than eight times more likely to attempt suicide than their peers who come from families with low or no levels of rejection (Pediatrics. 2009 Jan;123[1]:346-52). Going to school for roughly 8 hours a day allows some of these youth an escape from what is otherwise an unpleasant home situation. In addition, educators and other school staff may be among the only allies that a student has in his/her life, and school cancellations remove students from access to these important people.
Due to stay-at-home orders and physical distancing measures, lack of in-person access to medical and psychological care can be distressing for many LGBTQ youth. While many practices have been able to convert to audiovisual telemedicine visits, not all of them have the resources or capability to do so. Consequently, LGBTQ youth may have reduced access to support services that help to bolster their social and emotional health. In addition, many trans youth suffer from physical dysphoria that can make it distressing to see themselves on camera doing teletherapy and so they wish to avoid it for this reason.
This is not to say that everything is bleak. LGBTQ youth can also be resilient in times of stress and worry. “The LGBTQ community has a long history of overcoming adversity and utilizing challenges to build an even stronger sense of community. This pandemic will create yet another opportunity for us to highlight existing health disparities and to support our LGBTQ young people in finding creative responses,” said Heather Newby, LCSW, clinical social worker for the GENECIS (GENder Education and Care Interdisciplinary Support) Program at Children’s Medical Center Dallas. In addition, she reported that many LGBTQ advocacy groups have created excellent online support networks and resources to provide nationwide, regional, and local help.
During these challenging times, there are a number of resources that LGBTQ youth can turn to while trying to maintain their connection to their peers. First, many local LGBTQ service organizations have moved their in-person support groups to a virtual or online platform. Check with your local service organization to see what they are offering during these times. National organizations, such as Gender Spectrum, continue to have online groups as well that youth can participate in. Second, many virtual mental health helplines, such as those through the Trevor Project, remain staffed should LGBTQ youth need to access their services (1-866-488-7386, plus text and chat). They can be reached 24/7 to help those whose mental health has been affected during this pandemic. Third, youth can continue to stay connected to their friends through means such as Zoom, FaceTime, or other virtual audiovisual tools. Lastly, some youth have taken to meeting in school parking lots, mall parking lots, etc., and staying at least 6 feet apart so that they can still see their friends in person.
While the current times may be challenging, they will pass and we will be able to return to those activities that bring us joy. Do not hesitate to reach out if you need help. As Rainer Maria Rilke once said, “In the difficult, we must have our joys, our happiness, our dreams: There against the depth of this background, they stand out, there for the first time we see how beautiful they are.”
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. He has no relevant financial disclosures. Dr. Cooper is on Twitter @teendocmbc. Email him at [email protected].
Social distancing comes to the medicine wards
As the coronavirus pandemic has swept across America, so have advisories for social distancing. As of April 2, stay-at-home orders had been given in 38 states and parts of 7 more, affecting about 300 million people. Most of these people have been asked to maintain 6 feet of separation to anyone outside their immediate family and to avoid all avoidable contacts.
Typical hospital medicine patients at an academic hospital, however, traditionally receive visits from their hospitalist, an intern, a resident, and sometimes several medical students, pharmacists, and case managers. At University of California, San Diego, Health, many of these visits would occur during Focused Interdisciplinary Team rounds, with providers moving together in close proximity.
Asymptomatic and presymptomatic spread of coronavirus have been documented, which means distancing is a good idea for everyone. The risks of traditional patient visits during the coronavirus pandemic include spread to both patients (at high risk of complications) and staff (taken out of the workforce during surge times). Even if coronavirus were not a risk, visits to isolation rooms consume PPE, which is in short supply.
In response to the pandemic, UCSD Hospital Medicine drafted guidelines for the reduction of patient contacts. Our slide presentations and written guidelines were then distributed to physicians, nurses, pharmacists, and other staff by our pandemic response command center. Key points include the following:
- Target one in-person MD visit per day for stable patients. This means that attending reexaminations of patients seen by residents, nurse practitioners, physician assistants, and so on would not be done for billing or teaching purposes, only when clinically necessary.
- Use phone or video conferencing for follow-up discussions unless direct patient contact is needed.
- Consider skipping daily exams on patients who do not require them, such as patients awaiting placement or stably receiving long courses of antibiotics. Interview them remotely or from the door instead.
- Conduct team rounds, patient discussions, and handoffs with all members 6 feet apart or by telephone or video. Avoid shared work rooms. Substitute video conferences for in-person meetings. Use EMR embedded messaging to reduce face-to-face discussions.
- Check if a patient is ready for a visit before donning PPE to avoid waste.
- Explain to patients that distancing is being conducted to protect them. In our experience, when patients are asked about distancing, they welcome the changes.
We have also considered that most patient visits are generated by nurses and assistants. To increase distancing and reduce PPE waste, we have encouraged nurses and pharmacists to maximize their use of remote communication with patients and to suggest changes to care plans and come up with creative solutions to reduce traffic. We specifically suggested the following changes to routine care:
- Reduce frequency of taking vital signs, such as just daily or as needed, in stable patients (for example, those awaiting placement).
- Reduce checks for alcohol withdrawal and neurologic status as soon as possible, and stop fingersticks in patients with well-controlled diabetes not receiving insulin.
- Substitute less frequently administered medications where appropriate if doing so would reduce room traffic (such as enoxaparin for heparin, ceftriaxone for cefazolin, naproxen for ibuprofen, or patient-controlled analgesia for as needed morphine).
- Place intravenous pumps in halls if needed – luckily, our situation has not required these measures in San Diego.
- Explore the possibility of increased patient self-management (self-dosed insulin or inhalers) where medically appropriate.
- Eliminate food service and janitorial trips to isolation rooms unless requested by registered nurse.
There are clear downsides to medical distancing for hospital medicine patients. Patients might have delayed diagnosis of new conditions or inadequate management of conditions requiring frequent assessment, such as alcohol withdrawal. Opportunities for miscommunication (either patient-provider or provider-provider) may be increased with distancing. Isolation also comes with emotional costs such as stress and feelings of isolation or abandonment. Given the dynamic nature of the pandemic response, we are continually reevaluating our distancing guidelines to administer the safest and most effective hospital care possible as we approach California’s expected peak coronavirus infection period.
Dr. Jenkins is professor and chair of the Patient Safety Committee in the Division of Hospital Medicine at UCSD. Dr. Seymann is clinical professor and vice chief for academic affairs, UCSD division of hospital medicine. Dr. Horman and Dr. Bell are hospitalists and associate professors of medicine at UC San Diego Health.
As the coronavirus pandemic has swept across America, so have advisories for social distancing. As of April 2, stay-at-home orders had been given in 38 states and parts of 7 more, affecting about 300 million people. Most of these people have been asked to maintain 6 feet of separation to anyone outside their immediate family and to avoid all avoidable contacts.
Typical hospital medicine patients at an academic hospital, however, traditionally receive visits from their hospitalist, an intern, a resident, and sometimes several medical students, pharmacists, and case managers. At University of California, San Diego, Health, many of these visits would occur during Focused Interdisciplinary Team rounds, with providers moving together in close proximity.
Asymptomatic and presymptomatic spread of coronavirus have been documented, which means distancing is a good idea for everyone. The risks of traditional patient visits during the coronavirus pandemic include spread to both patients (at high risk of complications) and staff (taken out of the workforce during surge times). Even if coronavirus were not a risk, visits to isolation rooms consume PPE, which is in short supply.
In response to the pandemic, UCSD Hospital Medicine drafted guidelines for the reduction of patient contacts. Our slide presentations and written guidelines were then distributed to physicians, nurses, pharmacists, and other staff by our pandemic response command center. Key points include the following:
- Target one in-person MD visit per day for stable patients. This means that attending reexaminations of patients seen by residents, nurse practitioners, physician assistants, and so on would not be done for billing or teaching purposes, only when clinically necessary.
- Use phone or video conferencing for follow-up discussions unless direct patient contact is needed.
- Consider skipping daily exams on patients who do not require them, such as patients awaiting placement or stably receiving long courses of antibiotics. Interview them remotely or from the door instead.
- Conduct team rounds, patient discussions, and handoffs with all members 6 feet apart or by telephone or video. Avoid shared work rooms. Substitute video conferences for in-person meetings. Use EMR embedded messaging to reduce face-to-face discussions.
- Check if a patient is ready for a visit before donning PPE to avoid waste.
- Explain to patients that distancing is being conducted to protect them. In our experience, when patients are asked about distancing, they welcome the changes.
We have also considered that most patient visits are generated by nurses and assistants. To increase distancing and reduce PPE waste, we have encouraged nurses and pharmacists to maximize their use of remote communication with patients and to suggest changes to care plans and come up with creative solutions to reduce traffic. We specifically suggested the following changes to routine care:
- Reduce frequency of taking vital signs, such as just daily or as needed, in stable patients (for example, those awaiting placement).
- Reduce checks for alcohol withdrawal and neurologic status as soon as possible, and stop fingersticks in patients with well-controlled diabetes not receiving insulin.
- Substitute less frequently administered medications where appropriate if doing so would reduce room traffic (such as enoxaparin for heparin, ceftriaxone for cefazolin, naproxen for ibuprofen, or patient-controlled analgesia for as needed morphine).
- Place intravenous pumps in halls if needed – luckily, our situation has not required these measures in San Diego.
- Explore the possibility of increased patient self-management (self-dosed insulin or inhalers) where medically appropriate.
- Eliminate food service and janitorial trips to isolation rooms unless requested by registered nurse.
There are clear downsides to medical distancing for hospital medicine patients. Patients might have delayed diagnosis of new conditions or inadequate management of conditions requiring frequent assessment, such as alcohol withdrawal. Opportunities for miscommunication (either patient-provider or provider-provider) may be increased with distancing. Isolation also comes with emotional costs such as stress and feelings of isolation or abandonment. Given the dynamic nature of the pandemic response, we are continually reevaluating our distancing guidelines to administer the safest and most effective hospital care possible as we approach California’s expected peak coronavirus infection period.
Dr. Jenkins is professor and chair of the Patient Safety Committee in the Division of Hospital Medicine at UCSD. Dr. Seymann is clinical professor and vice chief for academic affairs, UCSD division of hospital medicine. Dr. Horman and Dr. Bell are hospitalists and associate professors of medicine at UC San Diego Health.
As the coronavirus pandemic has swept across America, so have advisories for social distancing. As of April 2, stay-at-home orders had been given in 38 states and parts of 7 more, affecting about 300 million people. Most of these people have been asked to maintain 6 feet of separation to anyone outside their immediate family and to avoid all avoidable contacts.
Typical hospital medicine patients at an academic hospital, however, traditionally receive visits from their hospitalist, an intern, a resident, and sometimes several medical students, pharmacists, and case managers. At University of California, San Diego, Health, many of these visits would occur during Focused Interdisciplinary Team rounds, with providers moving together in close proximity.
Asymptomatic and presymptomatic spread of coronavirus have been documented, which means distancing is a good idea for everyone. The risks of traditional patient visits during the coronavirus pandemic include spread to both patients (at high risk of complications) and staff (taken out of the workforce during surge times). Even if coronavirus were not a risk, visits to isolation rooms consume PPE, which is in short supply.
In response to the pandemic, UCSD Hospital Medicine drafted guidelines for the reduction of patient contacts. Our slide presentations and written guidelines were then distributed to physicians, nurses, pharmacists, and other staff by our pandemic response command center. Key points include the following:
- Target one in-person MD visit per day for stable patients. This means that attending reexaminations of patients seen by residents, nurse practitioners, physician assistants, and so on would not be done for billing or teaching purposes, only when clinically necessary.
- Use phone or video conferencing for follow-up discussions unless direct patient contact is needed.
- Consider skipping daily exams on patients who do not require them, such as patients awaiting placement or stably receiving long courses of antibiotics. Interview them remotely or from the door instead.
- Conduct team rounds, patient discussions, and handoffs with all members 6 feet apart or by telephone or video. Avoid shared work rooms. Substitute video conferences for in-person meetings. Use EMR embedded messaging to reduce face-to-face discussions.
- Check if a patient is ready for a visit before donning PPE to avoid waste.
- Explain to patients that distancing is being conducted to protect them. In our experience, when patients are asked about distancing, they welcome the changes.
We have also considered that most patient visits are generated by nurses and assistants. To increase distancing and reduce PPE waste, we have encouraged nurses and pharmacists to maximize their use of remote communication with patients and to suggest changes to care plans and come up with creative solutions to reduce traffic. We specifically suggested the following changes to routine care:
- Reduce frequency of taking vital signs, such as just daily or as needed, in stable patients (for example, those awaiting placement).
- Reduce checks for alcohol withdrawal and neurologic status as soon as possible, and stop fingersticks in patients with well-controlled diabetes not receiving insulin.
- Substitute less frequently administered medications where appropriate if doing so would reduce room traffic (such as enoxaparin for heparin, ceftriaxone for cefazolin, naproxen for ibuprofen, or patient-controlled analgesia for as needed morphine).
- Place intravenous pumps in halls if needed – luckily, our situation has not required these measures in San Diego.
- Explore the possibility of increased patient self-management (self-dosed insulin or inhalers) where medically appropriate.
- Eliminate food service and janitorial trips to isolation rooms unless requested by registered nurse.
There are clear downsides to medical distancing for hospital medicine patients. Patients might have delayed diagnosis of new conditions or inadequate management of conditions requiring frequent assessment, such as alcohol withdrawal. Opportunities for miscommunication (either patient-provider or provider-provider) may be increased with distancing. Isolation also comes with emotional costs such as stress and feelings of isolation or abandonment. Given the dynamic nature of the pandemic response, we are continually reevaluating our distancing guidelines to administer the safest and most effective hospital care possible as we approach California’s expected peak coronavirus infection period.
Dr. Jenkins is professor and chair of the Patient Safety Committee in the Division of Hospital Medicine at UCSD. Dr. Seymann is clinical professor and vice chief for academic affairs, UCSD division of hospital medicine. Dr. Horman and Dr. Bell are hospitalists and associate professors of medicine at UC San Diego Health.
Most e-consults not followed by specialist visit
Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
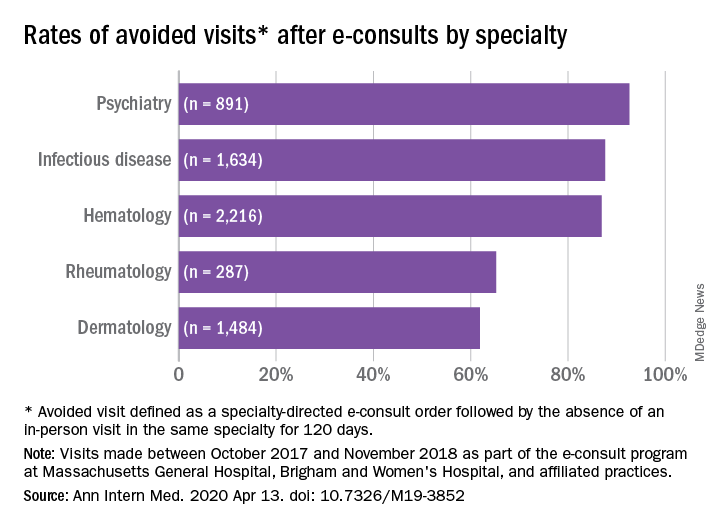
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.

Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.

Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.
FROM ANNALS OF INTERNAL MEDICINE
SARS-CoV-2 may confound seasons, persist in warmer months, report shows
Although conflicting, the available data indicate that SARS-CoV-2 could continue to spread in warmer spring and summer months in the US, according to a new report from the National Academies of Science, Engineering, and Medicine (NAS).
Current data suggest that the novel coronavirus may be transmitted less efficiently in higher temperatures and humidity, but the studies are not conclusive because of poor data quality, confounding factors, and the relatively short existence of the pandemic, which makes it difficult to determine its true course, writes David A. Relman, MD, a member of the NAS’ Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats, in a rapid expert consultation letter to the White House Office of Science and Technology Policy on April 7.
A number of factors could influence whether SARS-CoV-2 follows the same seasonal pattern as the influenza virus and other seasonal coronaviruses, which wane during warmer months, writes Relman, a professor of microbiology and immunology at Stanford University in California.
But he pointed out that previous coronavirus strains that have caused serious illness – SARS-CoV and MERS-CoV – “have not demonstrated any evidence of seasonality following their emergence.”
Relman cites an example from the current outbreak: “Given that countries currently in ‘summer’ climates, such as Australia and Iran, are experiencing rapid virus spread, a decrease in cases with increases in humidity and temperature elsewhere should not be assumed…Additional studies as the pandemic unfolds could shed more light on the effects of climate on transmission,” he writes.
And even if SARS-CoV-2 turns out to be less infectious in warmer months, “given the lack of host immunity globally, this reduction in transmission efficiency may not lead to a significant reduction in disease spread without the concomitant adoption of major public health interventions,” writes Relman.
Conflicting Data
Relman cites a handful of studies indicating that, on the one hand, SARS-CoV-2 has declined with increasing humidity and temperatures, but that conversely, infectivity has increased in warmer, more humid climates.
A recent study in China, published on the repository and international journal site SSRN, found that while increased temperatures and humidity decreased the infectivity, “the average R0 (R naught) was still close to 2 at maximum temperatures and humidity in their data set, suggesting that the virus will still spread exponentially at higher temperatures and humidity,” said Relman.
Several other studies found higher growth rates in temperate regions. One study, still in preprint on MedRxiv, looked at 310 geographic regions across 116 countries, and shows an inverse relationship between temperature and humidity and the incidence of COVID-19.
All the available studies so far have significant limitations, including limitation in time and location, confounding factors having to do with geography, access to and the quality of public health and health care systems, human behavior, and the availability of testing, said Relman.
However, he said, “it is useful to note that pandemic influenza strains have not exhibited the typical seasonal pattern of endemic/epidemic strains,” and, regardless of whether they started in a warmer or a cooler month, “all had a peak second wave approximately six months after the emergence in the human population.”
Worrisome Persistence on Masks
Seasonality can also be potentially gauged in the laboratory. Most of the studies on environmental persistence of SARS-CoV-2 have been conducted using virus grown in tissue culture. But that, too, is an imperfect method.
Virus disseminated into the environment from naturally infected humans likely has different survival properties than virus grown in culture, said Relman.
In addition, many labs cannot, or fail to, control and vary relative humidity, the committee letter noted. The aerosol studies so far have used humidity levels of 50% to 65%, which is more favorable to decay, while respiratory fluid is more likely to protect against infectivity, and the 20%-to-40% wintertime indoor humidity in temperate regions is more favorable for virus survival.
Even with these caveats, the committee cited worrisome studies on SARS-CoV-2 survival.
In a study published April 2 online in The Lancet, Hong Kong researchers reported significant reductions in virus in culture starting with temperatures at 37°C (98.6°F) or above.
On surfaces at a room temperature of 22°C (71.6°F) with a relative humidity of 65%, there was no infectious virus on printing paper or tissue papers after just 3 hours. It took 4 days for an infectious level to break down on glass and money, and 7 days for stainless steel and plastic. But after 7 days, investigators found 0.1% of the original inoculum on the outside of a surgical mask.
“The persistence of infectious virus on PPE is concerning,” writes Relman, noting that more studies are needed to guide healthcare workers, especially on what might be used to disinfect personal protective equipment “when they cannot be discarded after single use.”
Chad Roy, PhD, a researcher from Tulane University National Primate Research Center in New Orleans, Louisiana, told Relman by phone that in experiments where the virus was suspended as an aerosol at a temperature of 23°C (73.4° F) and about 50% humidity, SARS-CoV-2 had a longer half-life than the influenza virus, SARS-CoV-1, monkeypox virus, and Mycobacterium tuberculosis.
“This result is also concerning, but quite preliminary,” writes Relman.
This article first appeared on Medscape.com.
Although conflicting, the available data indicate that SARS-CoV-2 could continue to spread in warmer spring and summer months in the US, according to a new report from the National Academies of Science, Engineering, and Medicine (NAS).
Current data suggest that the novel coronavirus may be transmitted less efficiently in higher temperatures and humidity, but the studies are not conclusive because of poor data quality, confounding factors, and the relatively short existence of the pandemic, which makes it difficult to determine its true course, writes David A. Relman, MD, a member of the NAS’ Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats, in a rapid expert consultation letter to the White House Office of Science and Technology Policy on April 7.
A number of factors could influence whether SARS-CoV-2 follows the same seasonal pattern as the influenza virus and other seasonal coronaviruses, which wane during warmer months, writes Relman, a professor of microbiology and immunology at Stanford University in California.
But he pointed out that previous coronavirus strains that have caused serious illness – SARS-CoV and MERS-CoV – “have not demonstrated any evidence of seasonality following their emergence.”
Relman cites an example from the current outbreak: “Given that countries currently in ‘summer’ climates, such as Australia and Iran, are experiencing rapid virus spread, a decrease in cases with increases in humidity and temperature elsewhere should not be assumed…Additional studies as the pandemic unfolds could shed more light on the effects of climate on transmission,” he writes.
And even if SARS-CoV-2 turns out to be less infectious in warmer months, “given the lack of host immunity globally, this reduction in transmission efficiency may not lead to a significant reduction in disease spread without the concomitant adoption of major public health interventions,” writes Relman.
Conflicting Data
Relman cites a handful of studies indicating that, on the one hand, SARS-CoV-2 has declined with increasing humidity and temperatures, but that conversely, infectivity has increased in warmer, more humid climates.
A recent study in China, published on the repository and international journal site SSRN, found that while increased temperatures and humidity decreased the infectivity, “the average R0 (R naught) was still close to 2 at maximum temperatures and humidity in their data set, suggesting that the virus will still spread exponentially at higher temperatures and humidity,” said Relman.
Several other studies found higher growth rates in temperate regions. One study, still in preprint on MedRxiv, looked at 310 geographic regions across 116 countries, and shows an inverse relationship between temperature and humidity and the incidence of COVID-19.
All the available studies so far have significant limitations, including limitation in time and location, confounding factors having to do with geography, access to and the quality of public health and health care systems, human behavior, and the availability of testing, said Relman.
However, he said, “it is useful to note that pandemic influenza strains have not exhibited the typical seasonal pattern of endemic/epidemic strains,” and, regardless of whether they started in a warmer or a cooler month, “all had a peak second wave approximately six months after the emergence in the human population.”
Worrisome Persistence on Masks
Seasonality can also be potentially gauged in the laboratory. Most of the studies on environmental persistence of SARS-CoV-2 have been conducted using virus grown in tissue culture. But that, too, is an imperfect method.
Virus disseminated into the environment from naturally infected humans likely has different survival properties than virus grown in culture, said Relman.
In addition, many labs cannot, or fail to, control and vary relative humidity, the committee letter noted. The aerosol studies so far have used humidity levels of 50% to 65%, which is more favorable to decay, while respiratory fluid is more likely to protect against infectivity, and the 20%-to-40% wintertime indoor humidity in temperate regions is more favorable for virus survival.
Even with these caveats, the committee cited worrisome studies on SARS-CoV-2 survival.
In a study published April 2 online in The Lancet, Hong Kong researchers reported significant reductions in virus in culture starting with temperatures at 37°C (98.6°F) or above.
On surfaces at a room temperature of 22°C (71.6°F) with a relative humidity of 65%, there was no infectious virus on printing paper or tissue papers after just 3 hours. It took 4 days for an infectious level to break down on glass and money, and 7 days for stainless steel and plastic. But after 7 days, investigators found 0.1% of the original inoculum on the outside of a surgical mask.
“The persistence of infectious virus on PPE is concerning,” writes Relman, noting that more studies are needed to guide healthcare workers, especially on what might be used to disinfect personal protective equipment “when they cannot be discarded after single use.”
Chad Roy, PhD, a researcher from Tulane University National Primate Research Center in New Orleans, Louisiana, told Relman by phone that in experiments where the virus was suspended as an aerosol at a temperature of 23°C (73.4° F) and about 50% humidity, SARS-CoV-2 had a longer half-life than the influenza virus, SARS-CoV-1, monkeypox virus, and Mycobacterium tuberculosis.
“This result is also concerning, but quite preliminary,” writes Relman.
This article first appeared on Medscape.com.
Although conflicting, the available data indicate that SARS-CoV-2 could continue to spread in warmer spring and summer months in the US, according to a new report from the National Academies of Science, Engineering, and Medicine (NAS).
Current data suggest that the novel coronavirus may be transmitted less efficiently in higher temperatures and humidity, but the studies are not conclusive because of poor data quality, confounding factors, and the relatively short existence of the pandemic, which makes it difficult to determine its true course, writes David A. Relman, MD, a member of the NAS’ Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats, in a rapid expert consultation letter to the White House Office of Science and Technology Policy on April 7.
A number of factors could influence whether SARS-CoV-2 follows the same seasonal pattern as the influenza virus and other seasonal coronaviruses, which wane during warmer months, writes Relman, a professor of microbiology and immunology at Stanford University in California.
But he pointed out that previous coronavirus strains that have caused serious illness – SARS-CoV and MERS-CoV – “have not demonstrated any evidence of seasonality following their emergence.”
Relman cites an example from the current outbreak: “Given that countries currently in ‘summer’ climates, such as Australia and Iran, are experiencing rapid virus spread, a decrease in cases with increases in humidity and temperature elsewhere should not be assumed…Additional studies as the pandemic unfolds could shed more light on the effects of climate on transmission,” he writes.
And even if SARS-CoV-2 turns out to be less infectious in warmer months, “given the lack of host immunity globally, this reduction in transmission efficiency may not lead to a significant reduction in disease spread without the concomitant adoption of major public health interventions,” writes Relman.
Conflicting Data
Relman cites a handful of studies indicating that, on the one hand, SARS-CoV-2 has declined with increasing humidity and temperatures, but that conversely, infectivity has increased in warmer, more humid climates.
A recent study in China, published on the repository and international journal site SSRN, found that while increased temperatures and humidity decreased the infectivity, “the average R0 (R naught) was still close to 2 at maximum temperatures and humidity in their data set, suggesting that the virus will still spread exponentially at higher temperatures and humidity,” said Relman.
Several other studies found higher growth rates in temperate regions. One study, still in preprint on MedRxiv, looked at 310 geographic regions across 116 countries, and shows an inverse relationship between temperature and humidity and the incidence of COVID-19.
All the available studies so far have significant limitations, including limitation in time and location, confounding factors having to do with geography, access to and the quality of public health and health care systems, human behavior, and the availability of testing, said Relman.
However, he said, “it is useful to note that pandemic influenza strains have not exhibited the typical seasonal pattern of endemic/epidemic strains,” and, regardless of whether they started in a warmer or a cooler month, “all had a peak second wave approximately six months after the emergence in the human population.”
Worrisome Persistence on Masks
Seasonality can also be potentially gauged in the laboratory. Most of the studies on environmental persistence of SARS-CoV-2 have been conducted using virus grown in tissue culture. But that, too, is an imperfect method.
Virus disseminated into the environment from naturally infected humans likely has different survival properties than virus grown in culture, said Relman.
In addition, many labs cannot, or fail to, control and vary relative humidity, the committee letter noted. The aerosol studies so far have used humidity levels of 50% to 65%, which is more favorable to decay, while respiratory fluid is more likely to protect against infectivity, and the 20%-to-40% wintertime indoor humidity in temperate regions is more favorable for virus survival.
Even with these caveats, the committee cited worrisome studies on SARS-CoV-2 survival.
In a study published April 2 online in The Lancet, Hong Kong researchers reported significant reductions in virus in culture starting with temperatures at 37°C (98.6°F) or above.
On surfaces at a room temperature of 22°C (71.6°F) with a relative humidity of 65%, there was no infectious virus on printing paper or tissue papers after just 3 hours. It took 4 days for an infectious level to break down on glass and money, and 7 days for stainless steel and plastic. But after 7 days, investigators found 0.1% of the original inoculum on the outside of a surgical mask.
“The persistence of infectious virus on PPE is concerning,” writes Relman, noting that more studies are needed to guide healthcare workers, especially on what might be used to disinfect personal protective equipment “when they cannot be discarded after single use.”
Chad Roy, PhD, a researcher from Tulane University National Primate Research Center in New Orleans, Louisiana, told Relman by phone that in experiments where the virus was suspended as an aerosol at a temperature of 23°C (73.4° F) and about 50% humidity, SARS-CoV-2 had a longer half-life than the influenza virus, SARS-CoV-1, monkeypox virus, and Mycobacterium tuberculosis.
“This result is also concerning, but quite preliminary,” writes Relman.
This article first appeared on Medscape.com.
What do early remdesivir data suggest?
New data on the investigational antiviral drug remdesivir (Gilead) suggest clinical improvement in 36 of 53 patients (68%) hospitalized for severe COVID-19, according to a new study published online April 10 in the New England Journal of Medicine.
But experts are warning that these data come from compassionate use in a wide variety of patients, with no randomization and no control group.
“It is impossible to know the outcome for this relatively small group of patients had they not received remdesivir,” commented Stephen Griffin, PhD, associate professor at the University of Leeds School of Medicine, United Kingdom, who was not involved with the study.
“As the authors point out, a randomized clinical trial is necessary to determine the true effectiveness of this drug,” Griffin added in comments he provided to the Science Media Centre in London. Such trials are underway.
“The data from this paper are almost uninterpretable,” said Stephen Evans, MSc, FRCP, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who provided comments to the Science Media Centre.
Evans notes that the authors describe multiple caveats that limit interpretation of the results, including the small sample size, the relatively short follow-up, missing data, no follow-up on eight patients, and lack of a randomized control group.
Meanwhile, Josh Farkas, MD, who writes the PulmCrit blog, details his criticisms in a piece entitled, “Eleven reasons the NEJM paper on remdesivir reveals nothing.” Beyond the issues the authors list, he points out several more, including cherry picking of patients. “Remdesivir was aggressively sought-after by thousands of patients with COVID-19,” he writes. “Of these patients, 61 ended up receiving the drug. Why did these patients receive medication, out of scores of patients applying to receive it?”
Also, there are no follow-up data for 8 of the 61 patients who received an initial dose of the drug, leaving 53 for the published analysis, continues Farkas, who is an assistant professor of pulmonary and critical care medicine at the University of Vermont in Burlington.
“What happened to these patients? Did they die from anaphylaxis? Did they get well, sign out against medical advice, and go party? This is unknown — but I’m worried that these patients actually didn’t fare so well,” Farkas writes.
Farkas, like Evans and Griffin, concludes that the data are largely unusable. “Until [a randomized controlled trial] is performed, further compassionate use of remdesivir probably isn’t justified,” he writes.
Data from Compassionate Use Program
The data in the NEJM article come from a compassionate use program set up by Gilead. The company says it has provided emergency access to remdesivir for several hundred patients in the United States, Europe, and Japan.
The authors, led by Jonathan Grein, MD, from Cedars–Sinai Medical Center, Los Angeles, California, report on 61 patients who received remdesivir as part of this program.
The authors, several of whom are employees of Gilead, note that data on 8 patients could not be analyzed (including 7 patients with no posttreatment data and 1 with a dosing error).
Of the 53 patients whose data were included, 22 were in the United States, 22 in Europe or Canada, and 9 in Japan.
These were patients hospitalized with COVID-19 who had confirmed SARS-CoV-2 infection and had an oxygen saturation of 94% or less while they were breathing ambient air, or who were receiving oxygen support.
Patients received a 10-day course of remdesivir, consisting of 200 mg administered intravenously on day 1, followed by 100 mg daily for the remaining 9 days of treatment.
At baseline, 30 patients (57%) were receiving mechanical ventilation and 4 (8%) were receiving extracorporeal membrane oxygenation.
During a median follow-up of 18 days, 36 patients (68%) had an improvement in oxygen-support class, including 17 (57%) of 30 patients receiving mechanical ventilation who were extubated.
A total of 25 patients (47%) were discharged, and 7 patients (13%) died; mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
While the authors acknowledge limitations of the data they collected, they nevertheless comment that “comparisons with contemporaneous cohorts from the literature, in whom general care is expected to be consistent with that of our cohort, suggest that remdesivir may have clinical benefit in patients with severe COVID-19.”
“Currently there is no proven treatment for COVID-19. We cannot draw definitive conclusions from these data, but the observations from this group of hospitalized patients who received remdesivir are hopeful,” said Grein in a Cedars–Sinai press release. “We look forward to the results of controlled clinical trials to potentially validate these findings.”
Experts are not convinced, however.
“The drug was being used in patients who were severely ill, but reporting on 61 out of several hundred makes it clear that generalizations about the efficacy and safety must be treated with great caution,” said Evans. “There is some evidence suggesting efficacy, but we simply do not know what would have happened to these patients had they not been given the drug.”
“I would say it’s impossible to discern whether there is a treatment effect or not,” said Duncan Richards, MA, DM, FRCP, clinical pharmacologist and professor of clinical therapeutics, University of Oxford, UK. “This is in part due to the mixed patient population, ranging from those needing low dose oxygen, who are more likely to survive anyway, to much more severe cases ... [who] show a much more mixed picture.”
“There are ongoing large international randomized controlled trials with remdesivir — we really need to see those data, “ he said in comments to Science Media Centre. “Safe and effective treatments for COVID-19 are critically needed and should be expedited wherever possible, but it’s important not to compromise on the quality of the research.”
Multiple coauthors are employees of Gilead, the company developing remdesivir. Griffin, Evans, and Farkas have disclosed no relevant financial relationships. Richards consults for GlaxoSmithKline in the field of drug safety. GSK does not manufacture any of the products mentioned.
N Engl J Med. 2020 Apr 10. Full text.
This article first appeared on Medscape.com.
New data on the investigational antiviral drug remdesivir (Gilead) suggest clinical improvement in 36 of 53 patients (68%) hospitalized for severe COVID-19, according to a new study published online April 10 in the New England Journal of Medicine.
But experts are warning that these data come from compassionate use in a wide variety of patients, with no randomization and no control group.
“It is impossible to know the outcome for this relatively small group of patients had they not received remdesivir,” commented Stephen Griffin, PhD, associate professor at the University of Leeds School of Medicine, United Kingdom, who was not involved with the study.
“As the authors point out, a randomized clinical trial is necessary to determine the true effectiveness of this drug,” Griffin added in comments he provided to the Science Media Centre in London. Such trials are underway.
“The data from this paper are almost uninterpretable,” said Stephen Evans, MSc, FRCP, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who provided comments to the Science Media Centre.
Evans notes that the authors describe multiple caveats that limit interpretation of the results, including the small sample size, the relatively short follow-up, missing data, no follow-up on eight patients, and lack of a randomized control group.
Meanwhile, Josh Farkas, MD, who writes the PulmCrit blog, details his criticisms in a piece entitled, “Eleven reasons the NEJM paper on remdesivir reveals nothing.” Beyond the issues the authors list, he points out several more, including cherry picking of patients. “Remdesivir was aggressively sought-after by thousands of patients with COVID-19,” he writes. “Of these patients, 61 ended up receiving the drug. Why did these patients receive medication, out of scores of patients applying to receive it?”
Also, there are no follow-up data for 8 of the 61 patients who received an initial dose of the drug, leaving 53 for the published analysis, continues Farkas, who is an assistant professor of pulmonary and critical care medicine at the University of Vermont in Burlington.
“What happened to these patients? Did they die from anaphylaxis? Did they get well, sign out against medical advice, and go party? This is unknown — but I’m worried that these patients actually didn’t fare so well,” Farkas writes.
Farkas, like Evans and Griffin, concludes that the data are largely unusable. “Until [a randomized controlled trial] is performed, further compassionate use of remdesivir probably isn’t justified,” he writes.
Data from Compassionate Use Program
The data in the NEJM article come from a compassionate use program set up by Gilead. The company says it has provided emergency access to remdesivir for several hundred patients in the United States, Europe, and Japan.
The authors, led by Jonathan Grein, MD, from Cedars–Sinai Medical Center, Los Angeles, California, report on 61 patients who received remdesivir as part of this program.
The authors, several of whom are employees of Gilead, note that data on 8 patients could not be analyzed (including 7 patients with no posttreatment data and 1 with a dosing error).
Of the 53 patients whose data were included, 22 were in the United States, 22 in Europe or Canada, and 9 in Japan.
These were patients hospitalized with COVID-19 who had confirmed SARS-CoV-2 infection and had an oxygen saturation of 94% or less while they were breathing ambient air, or who were receiving oxygen support.
Patients received a 10-day course of remdesivir, consisting of 200 mg administered intravenously on day 1, followed by 100 mg daily for the remaining 9 days of treatment.
At baseline, 30 patients (57%) were receiving mechanical ventilation and 4 (8%) were receiving extracorporeal membrane oxygenation.
During a median follow-up of 18 days, 36 patients (68%) had an improvement in oxygen-support class, including 17 (57%) of 30 patients receiving mechanical ventilation who were extubated.
A total of 25 patients (47%) were discharged, and 7 patients (13%) died; mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
While the authors acknowledge limitations of the data they collected, they nevertheless comment that “comparisons with contemporaneous cohorts from the literature, in whom general care is expected to be consistent with that of our cohort, suggest that remdesivir may have clinical benefit in patients with severe COVID-19.”
“Currently there is no proven treatment for COVID-19. We cannot draw definitive conclusions from these data, but the observations from this group of hospitalized patients who received remdesivir are hopeful,” said Grein in a Cedars–Sinai press release. “We look forward to the results of controlled clinical trials to potentially validate these findings.”
Experts are not convinced, however.
“The drug was being used in patients who were severely ill, but reporting on 61 out of several hundred makes it clear that generalizations about the efficacy and safety must be treated with great caution,” said Evans. “There is some evidence suggesting efficacy, but we simply do not know what would have happened to these patients had they not been given the drug.”
“I would say it’s impossible to discern whether there is a treatment effect or not,” said Duncan Richards, MA, DM, FRCP, clinical pharmacologist and professor of clinical therapeutics, University of Oxford, UK. “This is in part due to the mixed patient population, ranging from those needing low dose oxygen, who are more likely to survive anyway, to much more severe cases ... [who] show a much more mixed picture.”
“There are ongoing large international randomized controlled trials with remdesivir — we really need to see those data, “ he said in comments to Science Media Centre. “Safe and effective treatments for COVID-19 are critically needed and should be expedited wherever possible, but it’s important not to compromise on the quality of the research.”
Multiple coauthors are employees of Gilead, the company developing remdesivir. Griffin, Evans, and Farkas have disclosed no relevant financial relationships. Richards consults for GlaxoSmithKline in the field of drug safety. GSK does not manufacture any of the products mentioned.
N Engl J Med. 2020 Apr 10. Full text.
This article first appeared on Medscape.com.
New data on the investigational antiviral drug remdesivir (Gilead) suggest clinical improvement in 36 of 53 patients (68%) hospitalized for severe COVID-19, according to a new study published online April 10 in the New England Journal of Medicine.
But experts are warning that these data come from compassionate use in a wide variety of patients, with no randomization and no control group.
“It is impossible to know the outcome for this relatively small group of patients had they not received remdesivir,” commented Stephen Griffin, PhD, associate professor at the University of Leeds School of Medicine, United Kingdom, who was not involved with the study.
“As the authors point out, a randomized clinical trial is necessary to determine the true effectiveness of this drug,” Griffin added in comments he provided to the Science Media Centre in London. Such trials are underway.
“The data from this paper are almost uninterpretable,” said Stephen Evans, MSc, FRCP, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who provided comments to the Science Media Centre.
Evans notes that the authors describe multiple caveats that limit interpretation of the results, including the small sample size, the relatively short follow-up, missing data, no follow-up on eight patients, and lack of a randomized control group.
Meanwhile, Josh Farkas, MD, who writes the PulmCrit blog, details his criticisms in a piece entitled, “Eleven reasons the NEJM paper on remdesivir reveals nothing.” Beyond the issues the authors list, he points out several more, including cherry picking of patients. “Remdesivir was aggressively sought-after by thousands of patients with COVID-19,” he writes. “Of these patients, 61 ended up receiving the drug. Why did these patients receive medication, out of scores of patients applying to receive it?”
Also, there are no follow-up data for 8 of the 61 patients who received an initial dose of the drug, leaving 53 for the published analysis, continues Farkas, who is an assistant professor of pulmonary and critical care medicine at the University of Vermont in Burlington.
“What happened to these patients? Did they die from anaphylaxis? Did they get well, sign out against medical advice, and go party? This is unknown — but I’m worried that these patients actually didn’t fare so well,” Farkas writes.
Farkas, like Evans and Griffin, concludes that the data are largely unusable. “Until [a randomized controlled trial] is performed, further compassionate use of remdesivir probably isn’t justified,” he writes.
Data from Compassionate Use Program
The data in the NEJM article come from a compassionate use program set up by Gilead. The company says it has provided emergency access to remdesivir for several hundred patients in the United States, Europe, and Japan.
The authors, led by Jonathan Grein, MD, from Cedars–Sinai Medical Center, Los Angeles, California, report on 61 patients who received remdesivir as part of this program.
The authors, several of whom are employees of Gilead, note that data on 8 patients could not be analyzed (including 7 patients with no posttreatment data and 1 with a dosing error).
Of the 53 patients whose data were included, 22 were in the United States, 22 in Europe or Canada, and 9 in Japan.
These were patients hospitalized with COVID-19 who had confirmed SARS-CoV-2 infection and had an oxygen saturation of 94% or less while they were breathing ambient air, or who were receiving oxygen support.
Patients received a 10-day course of remdesivir, consisting of 200 mg administered intravenously on day 1, followed by 100 mg daily for the remaining 9 days of treatment.
At baseline, 30 patients (57%) were receiving mechanical ventilation and 4 (8%) were receiving extracorporeal membrane oxygenation.
During a median follow-up of 18 days, 36 patients (68%) had an improvement in oxygen-support class, including 17 (57%) of 30 patients receiving mechanical ventilation who were extubated.
A total of 25 patients (47%) were discharged, and 7 patients (13%) died; mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
While the authors acknowledge limitations of the data they collected, they nevertheless comment that “comparisons with contemporaneous cohorts from the literature, in whom general care is expected to be consistent with that of our cohort, suggest that remdesivir may have clinical benefit in patients with severe COVID-19.”
“Currently there is no proven treatment for COVID-19. We cannot draw definitive conclusions from these data, but the observations from this group of hospitalized patients who received remdesivir are hopeful,” said Grein in a Cedars–Sinai press release. “We look forward to the results of controlled clinical trials to potentially validate these findings.”
Experts are not convinced, however.
“The drug was being used in patients who were severely ill, but reporting on 61 out of several hundred makes it clear that generalizations about the efficacy and safety must be treated with great caution,” said Evans. “There is some evidence suggesting efficacy, but we simply do not know what would have happened to these patients had they not been given the drug.”
“I would say it’s impossible to discern whether there is a treatment effect or not,” said Duncan Richards, MA, DM, FRCP, clinical pharmacologist and professor of clinical therapeutics, University of Oxford, UK. “This is in part due to the mixed patient population, ranging from those needing low dose oxygen, who are more likely to survive anyway, to much more severe cases ... [who] show a much more mixed picture.”
“There are ongoing large international randomized controlled trials with remdesivir — we really need to see those data, “ he said in comments to Science Media Centre. “Safe and effective treatments for COVID-19 are critically needed and should be expedited wherever possible, but it’s important not to compromise on the quality of the research.”
Multiple coauthors are employees of Gilead, the company developing remdesivir. Griffin, Evans, and Farkas have disclosed no relevant financial relationships. Richards consults for GlaxoSmithKline in the field of drug safety. GSK does not manufacture any of the products mentioned.
N Engl J Med. 2020 Apr 10. Full text.
This article first appeared on Medscape.com.
Remdesivir tops list of promising COVID-19 treatments in review of nearly 300 trials
, according to authors of a recent review covering nearly 300 active clinical treatment trials underway for the disease.
Remdesivir, which has potent in vitro activity against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is not approved by the Food and Drug Administration and is currently being tested in randomized trials, according to the review authors, led by James M. Sanders, PhD, of the department of pharmacy at University of Texas Southwestern Medical Center in Dallas.
By contrast, oseltamivir has not demonstrated efficacy against the virus, corticosteroids are not recommended, and promising data from a small French hydroxychloroquine study are balanced by “several major limitations” including small sample size and exclusion of early dropouts from the analysis, among others, Dr. Sanders and coauthors said in their report.
“These limitations coupled with concerns of additive cardiotoxicity with combination therapy [i.e., hydroxychloroquine with azithromycin] do not support adoption of this regimen without additional studies,” the researchers wrote. Their report is in JAMA.
Dr. Sanders and colleagues identified 291 COVID-19–specific studies listed in ClinicalTrials.gov through April 2, including 29 placebo-controlled trials.
This might represent just a sliver of the treatments that could combat COVID-19, according to the researchers, who said more than 3,000 small-molecule drug candidates with potential activity against human coronaviruses have been identified.
“This large amount of potential agents will hopefully yield more candidate therapeutics in the race to find effective treatments or preventive strategies against COVID-19,” said Dr. Sanders and coauthors.
Remdesivir for COVID-19
Remdesivir, an investigational nucleotide analog, is one promising agent because of its broad-spectrum and potent activity against SARS-CoV-2 and other novel coronaviruses, they said, adding that phase 1 trials demonstrated the drug was well tolerated without observed liver or kidney toxicity.
There have been “successful” case reports of remdesivir use in COVID-19, and at least five ongoing clinical trials are evaluating the drug’s safety and antiviral activity in this disease. Among those studies is a National Institutes of Health–sponsored adaptive, randomized, placebo-controlled trial that will provide data on the use of remdesivir versus supportive care.
“As the results from randomized controlled trials are anticipated, inclusion of this agent for treatment of COVID-19 may be considered,” Dr. Sanders and colleagues wrote in their report. To date, remdesivir remains investigational and needs to be obtained via compassionate use, through expanded access, or by participating in a clinical trial, they added.
Hydroxychloroquine and chloroquine
Among the published hydroxychloroquine studies is a “promising” 36-patient open-label nonrandomized French study, in which the antimalarial agent given every 8 hours improved virologic clearance by day 6 versus controls (70% vs. 12.5%, respectively), the review authors said. Moreover, viral clearance was 100% for 6 patients who received hydroxychloroquine plus azithromycin, compared to 57% (8 of 14) for patients treated with hydroxychloroquine alone. However, that study had several important limitations, including the small sample size, variable viral loads at baseline between groups, and a lack of safety and clinical outcomes reporting, according to the investigators. Moreover, six patients in the hydroxychloroquine group were taken out of the analysis because of early treatment stoppage due to medical intolerance or critical illness, the authors noted.
One prospective study including 30 patients in China demonstrated no difference in virologic outcomes for patients randomized to hydroxychloroquine plus standard of care versus standard of care alone, they added. There is also a case series of more than 100 patients with COVID-19 that reportedly improved viral clearance and reduced disease progression, though they said results haven’t been published or presented beyond a news briefing in China.
Randomized, controlled trials of chloroquine and hydroxychloroquine for COVID-19 treatment are underway, and studies are planned or enrolling to look at chloroquine prophylaxis in health care personnel and hydroxychloroquine for postexposure prophylaxis, authors said.
In results from one of those randomized trials, just reported, a higher dose of chloroquine was associated with a cardiac adverse event and an increased mortality risk, leading to the closure of that study arm. In the parallel, double-blinded, phase IIb clinical trial, patients in Brazil with SARS-CoV-2 infection received low or high doses of chloroquine plus ceftriaxone and azithromycin. According to the preprint publication, a higher rate of heart rate–corrected QT interval (QTc) prolongation and a “trend toward higher lethality” was observed in the high-dose group, leading investigators to “strongly recommend” the higher dose be abandoned.
“No apparent benefit of chloroquine was seen regarding lethality in our patients so far, but we will still enroll patients in the low chloroquine dose group to complete the originally planned sample size,” said investigators of the study, which at the time of the report had enrolled 81 out of an anticipated 440 patients.
Other COVID-19 pharmacologic therapies under study
Treatments of note in the review included the following:
- Tocilizumab. This monoclonal antibody IL-6 receptor antagonist, approved by the FDA for treatment of rheumatoid arthritis and for cytokine release syndrome related to chimeric antigen receptor (CAR) T-cell therapy, has yielded success in small series of patients with severe cases of COVID-19, according to authors. In one 21-patient report, 91% had clinical improvement, usually after a single dose. In China, tocilizumab is included in COVID-19 treatment guidelines, and several randomized clinical trials are underway in China including patients with COVID-19 with severe pneumonia.
- Immunoglobulin therapy. Antibodies from recovered COVID-19 patients could help with free virus and infected cell immune clearance, the authors said, adding that further studies are warranted beyond a few small published case series that suggest promise. Furthermore, on March 24 the FDA released guidance for screening donors for COVID-19 convalescent plasma and on emergency investigational new drug applications based on this modality.
- Lopinavir/ritonavir. Despite demonstrated in vitro activity against other novel coronaviruses, there is no published in vitro data for lopinavir/ritonavir in SARS-CoV-2, and likely a “limited role” for this combination anticipated in treating COVID-19, according to the review authors. In an open-label randomized clinical trial published in the New England Journal of Medicine (2020 Mar 18. doi: 10.1056/NEJMoa2001282), there were no differences in clinical improvement, viral clearance, or mortality for antiviral treatment versus standard care. Delayed treatment initiation may explain the ineffectiveness, though a subgroup analysis didn’t show a shorter time to clinical improvement for those who got the treatment earlier.
- Ribavirin. Likewise, this antiviral medication has efficacy and safety data suggesting “limited value” for treatment of COVID-19. Treatment of SARS yielded “inconclusive results” for ribavirin, which was also associated with substantial toxicity that included hemolytic anemia in 60% of SARS patients.
- Oseltamivir. While it may treat influenza, it has no documented activity against SARS-CoV-2 in vitro: “This agent has no role in the management of COVID-19 once influenza has been excluded,” said Dr. Sanders and coauthors.
- Corticosteroids. They could decrease inflammatory responses in the lung, but they could also lead to delays in viral clearance and increases in secondary infection risk. Guidelines for COVID-19 say to avoid corticosteroids, and the authors of the review concur, saying that potential harms and lack of proven benefit mean they usually should not be used outside of a randomized clinical trial setting.
- Vaccines. Clearly, vaccines represent the “most effective long-term strategy” to prevent future COVID-19 outbreaks, though at least 12-18 months would be required until vaccines can be widely deployed, authors said.
Dr. Sanders reported no potential conflicts. Senior author James B. Cutrell, MD, also of the University of Texas Southwestern Medical Center, reported nonfinancial support from Gilead and Regeneron outside of the study. No other authors reported disclosures.
, according to authors of a recent review covering nearly 300 active clinical treatment trials underway for the disease.
Remdesivir, which has potent in vitro activity against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is not approved by the Food and Drug Administration and is currently being tested in randomized trials, according to the review authors, led by James M. Sanders, PhD, of the department of pharmacy at University of Texas Southwestern Medical Center in Dallas.
By contrast, oseltamivir has not demonstrated efficacy against the virus, corticosteroids are not recommended, and promising data from a small French hydroxychloroquine study are balanced by “several major limitations” including small sample size and exclusion of early dropouts from the analysis, among others, Dr. Sanders and coauthors said in their report.
“These limitations coupled with concerns of additive cardiotoxicity with combination therapy [i.e., hydroxychloroquine with azithromycin] do not support adoption of this regimen without additional studies,” the researchers wrote. Their report is in JAMA.
Dr. Sanders and colleagues identified 291 COVID-19–specific studies listed in ClinicalTrials.gov through April 2, including 29 placebo-controlled trials.
This might represent just a sliver of the treatments that could combat COVID-19, according to the researchers, who said more than 3,000 small-molecule drug candidates with potential activity against human coronaviruses have been identified.
“This large amount of potential agents will hopefully yield more candidate therapeutics in the race to find effective treatments or preventive strategies against COVID-19,” said Dr. Sanders and coauthors.
Remdesivir for COVID-19
Remdesivir, an investigational nucleotide analog, is one promising agent because of its broad-spectrum and potent activity against SARS-CoV-2 and other novel coronaviruses, they said, adding that phase 1 trials demonstrated the drug was well tolerated without observed liver or kidney toxicity.
There have been “successful” case reports of remdesivir use in COVID-19, and at least five ongoing clinical trials are evaluating the drug’s safety and antiviral activity in this disease. Among those studies is a National Institutes of Health–sponsored adaptive, randomized, placebo-controlled trial that will provide data on the use of remdesivir versus supportive care.
“As the results from randomized controlled trials are anticipated, inclusion of this agent for treatment of COVID-19 may be considered,” Dr. Sanders and colleagues wrote in their report. To date, remdesivir remains investigational and needs to be obtained via compassionate use, through expanded access, or by participating in a clinical trial, they added.
Hydroxychloroquine and chloroquine
Among the published hydroxychloroquine studies is a “promising” 36-patient open-label nonrandomized French study, in which the antimalarial agent given every 8 hours improved virologic clearance by day 6 versus controls (70% vs. 12.5%, respectively), the review authors said. Moreover, viral clearance was 100% for 6 patients who received hydroxychloroquine plus azithromycin, compared to 57% (8 of 14) for patients treated with hydroxychloroquine alone. However, that study had several important limitations, including the small sample size, variable viral loads at baseline between groups, and a lack of safety and clinical outcomes reporting, according to the investigators. Moreover, six patients in the hydroxychloroquine group were taken out of the analysis because of early treatment stoppage due to medical intolerance or critical illness, the authors noted.
One prospective study including 30 patients in China demonstrated no difference in virologic outcomes for patients randomized to hydroxychloroquine plus standard of care versus standard of care alone, they added. There is also a case series of more than 100 patients with COVID-19 that reportedly improved viral clearance and reduced disease progression, though they said results haven’t been published or presented beyond a news briefing in China.
Randomized, controlled trials of chloroquine and hydroxychloroquine for COVID-19 treatment are underway, and studies are planned or enrolling to look at chloroquine prophylaxis in health care personnel and hydroxychloroquine for postexposure prophylaxis, authors said.
In results from one of those randomized trials, just reported, a higher dose of chloroquine was associated with a cardiac adverse event and an increased mortality risk, leading to the closure of that study arm. In the parallel, double-blinded, phase IIb clinical trial, patients in Brazil with SARS-CoV-2 infection received low or high doses of chloroquine plus ceftriaxone and azithromycin. According to the preprint publication, a higher rate of heart rate–corrected QT interval (QTc) prolongation and a “trend toward higher lethality” was observed in the high-dose group, leading investigators to “strongly recommend” the higher dose be abandoned.
“No apparent benefit of chloroquine was seen regarding lethality in our patients so far, but we will still enroll patients in the low chloroquine dose group to complete the originally planned sample size,” said investigators of the study, which at the time of the report had enrolled 81 out of an anticipated 440 patients.
Other COVID-19 pharmacologic therapies under study
Treatments of note in the review included the following:
- Tocilizumab. This monoclonal antibody IL-6 receptor antagonist, approved by the FDA for treatment of rheumatoid arthritis and for cytokine release syndrome related to chimeric antigen receptor (CAR) T-cell therapy, has yielded success in small series of patients with severe cases of COVID-19, according to authors. In one 21-patient report, 91% had clinical improvement, usually after a single dose. In China, tocilizumab is included in COVID-19 treatment guidelines, and several randomized clinical trials are underway in China including patients with COVID-19 with severe pneumonia.
- Immunoglobulin therapy. Antibodies from recovered COVID-19 patients could help with free virus and infected cell immune clearance, the authors said, adding that further studies are warranted beyond a few small published case series that suggest promise. Furthermore, on March 24 the FDA released guidance for screening donors for COVID-19 convalescent plasma and on emergency investigational new drug applications based on this modality.
- Lopinavir/ritonavir. Despite demonstrated in vitro activity against other novel coronaviruses, there is no published in vitro data for lopinavir/ritonavir in SARS-CoV-2, and likely a “limited role” for this combination anticipated in treating COVID-19, according to the review authors. In an open-label randomized clinical trial published in the New England Journal of Medicine (2020 Mar 18. doi: 10.1056/NEJMoa2001282), there were no differences in clinical improvement, viral clearance, or mortality for antiviral treatment versus standard care. Delayed treatment initiation may explain the ineffectiveness, though a subgroup analysis didn’t show a shorter time to clinical improvement for those who got the treatment earlier.
- Ribavirin. Likewise, this antiviral medication has efficacy and safety data suggesting “limited value” for treatment of COVID-19. Treatment of SARS yielded “inconclusive results” for ribavirin, which was also associated with substantial toxicity that included hemolytic anemia in 60% of SARS patients.
- Oseltamivir. While it may treat influenza, it has no documented activity against SARS-CoV-2 in vitro: “This agent has no role in the management of COVID-19 once influenza has been excluded,” said Dr. Sanders and coauthors.
- Corticosteroids. They could decrease inflammatory responses in the lung, but they could also lead to delays in viral clearance and increases in secondary infection risk. Guidelines for COVID-19 say to avoid corticosteroids, and the authors of the review concur, saying that potential harms and lack of proven benefit mean they usually should not be used outside of a randomized clinical trial setting.
- Vaccines. Clearly, vaccines represent the “most effective long-term strategy” to prevent future COVID-19 outbreaks, though at least 12-18 months would be required until vaccines can be widely deployed, authors said.
Dr. Sanders reported no potential conflicts. Senior author James B. Cutrell, MD, also of the University of Texas Southwestern Medical Center, reported nonfinancial support from Gilead and Regeneron outside of the study. No other authors reported disclosures.
, according to authors of a recent review covering nearly 300 active clinical treatment trials underway for the disease.
Remdesivir, which has potent in vitro activity against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is not approved by the Food and Drug Administration and is currently being tested in randomized trials, according to the review authors, led by James M. Sanders, PhD, of the department of pharmacy at University of Texas Southwestern Medical Center in Dallas.
By contrast, oseltamivir has not demonstrated efficacy against the virus, corticosteroids are not recommended, and promising data from a small French hydroxychloroquine study are balanced by “several major limitations” including small sample size and exclusion of early dropouts from the analysis, among others, Dr. Sanders and coauthors said in their report.
“These limitations coupled with concerns of additive cardiotoxicity with combination therapy [i.e., hydroxychloroquine with azithromycin] do not support adoption of this regimen without additional studies,” the researchers wrote. Their report is in JAMA.
Dr. Sanders and colleagues identified 291 COVID-19–specific studies listed in ClinicalTrials.gov through April 2, including 29 placebo-controlled trials.
This might represent just a sliver of the treatments that could combat COVID-19, according to the researchers, who said more than 3,000 small-molecule drug candidates with potential activity against human coronaviruses have been identified.
“This large amount of potential agents will hopefully yield more candidate therapeutics in the race to find effective treatments or preventive strategies against COVID-19,” said Dr. Sanders and coauthors.
Remdesivir for COVID-19
Remdesivir, an investigational nucleotide analog, is one promising agent because of its broad-spectrum and potent activity against SARS-CoV-2 and other novel coronaviruses, they said, adding that phase 1 trials demonstrated the drug was well tolerated without observed liver or kidney toxicity.
There have been “successful” case reports of remdesivir use in COVID-19, and at least five ongoing clinical trials are evaluating the drug’s safety and antiviral activity in this disease. Among those studies is a National Institutes of Health–sponsored adaptive, randomized, placebo-controlled trial that will provide data on the use of remdesivir versus supportive care.
“As the results from randomized controlled trials are anticipated, inclusion of this agent for treatment of COVID-19 may be considered,” Dr. Sanders and colleagues wrote in their report. To date, remdesivir remains investigational and needs to be obtained via compassionate use, through expanded access, or by participating in a clinical trial, they added.
Hydroxychloroquine and chloroquine
Among the published hydroxychloroquine studies is a “promising” 36-patient open-label nonrandomized French study, in which the antimalarial agent given every 8 hours improved virologic clearance by day 6 versus controls (70% vs. 12.5%, respectively), the review authors said. Moreover, viral clearance was 100% for 6 patients who received hydroxychloroquine plus azithromycin, compared to 57% (8 of 14) for patients treated with hydroxychloroquine alone. However, that study had several important limitations, including the small sample size, variable viral loads at baseline between groups, and a lack of safety and clinical outcomes reporting, according to the investigators. Moreover, six patients in the hydroxychloroquine group were taken out of the analysis because of early treatment stoppage due to medical intolerance or critical illness, the authors noted.
One prospective study including 30 patients in China demonstrated no difference in virologic outcomes for patients randomized to hydroxychloroquine plus standard of care versus standard of care alone, they added. There is also a case series of more than 100 patients with COVID-19 that reportedly improved viral clearance and reduced disease progression, though they said results haven’t been published or presented beyond a news briefing in China.
Randomized, controlled trials of chloroquine and hydroxychloroquine for COVID-19 treatment are underway, and studies are planned or enrolling to look at chloroquine prophylaxis in health care personnel and hydroxychloroquine for postexposure prophylaxis, authors said.
In results from one of those randomized trials, just reported, a higher dose of chloroquine was associated with a cardiac adverse event and an increased mortality risk, leading to the closure of that study arm. In the parallel, double-blinded, phase IIb clinical trial, patients in Brazil with SARS-CoV-2 infection received low or high doses of chloroquine plus ceftriaxone and azithromycin. According to the preprint publication, a higher rate of heart rate–corrected QT interval (QTc) prolongation and a “trend toward higher lethality” was observed in the high-dose group, leading investigators to “strongly recommend” the higher dose be abandoned.
“No apparent benefit of chloroquine was seen regarding lethality in our patients so far, but we will still enroll patients in the low chloroquine dose group to complete the originally planned sample size,” said investigators of the study, which at the time of the report had enrolled 81 out of an anticipated 440 patients.
Other COVID-19 pharmacologic therapies under study
Treatments of note in the review included the following:
- Tocilizumab. This monoclonal antibody IL-6 receptor antagonist, approved by the FDA for treatment of rheumatoid arthritis and for cytokine release syndrome related to chimeric antigen receptor (CAR) T-cell therapy, has yielded success in small series of patients with severe cases of COVID-19, according to authors. In one 21-patient report, 91% had clinical improvement, usually after a single dose. In China, tocilizumab is included in COVID-19 treatment guidelines, and several randomized clinical trials are underway in China including patients with COVID-19 with severe pneumonia.
- Immunoglobulin therapy. Antibodies from recovered COVID-19 patients could help with free virus and infected cell immune clearance, the authors said, adding that further studies are warranted beyond a few small published case series that suggest promise. Furthermore, on March 24 the FDA released guidance for screening donors for COVID-19 convalescent plasma and on emergency investigational new drug applications based on this modality.
- Lopinavir/ritonavir. Despite demonstrated in vitro activity against other novel coronaviruses, there is no published in vitro data for lopinavir/ritonavir in SARS-CoV-2, and likely a “limited role” for this combination anticipated in treating COVID-19, according to the review authors. In an open-label randomized clinical trial published in the New England Journal of Medicine (2020 Mar 18. doi: 10.1056/NEJMoa2001282), there were no differences in clinical improvement, viral clearance, or mortality for antiviral treatment versus standard care. Delayed treatment initiation may explain the ineffectiveness, though a subgroup analysis didn’t show a shorter time to clinical improvement for those who got the treatment earlier.
- Ribavirin. Likewise, this antiviral medication has efficacy and safety data suggesting “limited value” for treatment of COVID-19. Treatment of SARS yielded “inconclusive results” for ribavirin, which was also associated with substantial toxicity that included hemolytic anemia in 60% of SARS patients.
- Oseltamivir. While it may treat influenza, it has no documented activity against SARS-CoV-2 in vitro: “This agent has no role in the management of COVID-19 once influenza has been excluded,” said Dr. Sanders and coauthors.
- Corticosteroids. They could decrease inflammatory responses in the lung, but they could also lead to delays in viral clearance and increases in secondary infection risk. Guidelines for COVID-19 say to avoid corticosteroids, and the authors of the review concur, saying that potential harms and lack of proven benefit mean they usually should not be used outside of a randomized clinical trial setting.
- Vaccines. Clearly, vaccines represent the “most effective long-term strategy” to prevent future COVID-19 outbreaks, though at least 12-18 months would be required until vaccines can be widely deployed, authors said.
Dr. Sanders reported no potential conflicts. Senior author James B. Cutrell, MD, also of the University of Texas Southwestern Medical Center, reported nonfinancial support from Gilead and Regeneron outside of the study. No other authors reported disclosures.
FROM JAMA
COVID-19 pandemic brings unexpected pediatric consequences
As physicians and advanced practitioners, we have been preparing to face COVID-19 – anticipating increasing volumes of patients with fevers, cough, and shortness of breath, and potential surges in emergency departments (EDs) and primary care offices. Fortunately, while COVID-19 has demonstrated more mild symptoms in pediatric patients, the heightened public health fears and mandated social isolation have created some unforeseen consequences for pediatric patients. This article presents cases encountered over the course of 2 weeks in our ED that shed light on the unexpected ramifications of living in the time of a pandemic. These encounters should remind us as providers to be diligent and thorough in giving guidance to families during a time when face-to-face medicine has become increasingly difficult and limited.
These stories have been modified to protect patient confidentiality.
Case 1
A 2-week-old full-term infant arrived in the ED after having a fever for 48 hours. The patient’s mother reported that she had called the pediatrician yesterday to ask for advice on treating the fever and was instructed to give acetaminophen and bring the infant into the ED for testing.
When we asked mom why she did not bring the infant in yesterday, she stated that the fever went down with acetaminophen, and the baby was drinking well and urinating normally. Mostly, she was afraid to bring the child into the ED given concern for COVID-19; however, when the fever persisted today, she came in. During the work-up, the infant was noted to have focal seizures and was ultimately diagnosed with bacterial meningitis.
Takeaway: Families may be hesitant to follow pediatrician’s advice to seek medical attention at an ED or doctor’s office because of the fear of being exposed to COVID-19.
- If something is urgent or emergent, be sure to stress the importance to families that the advice is non-negotiable for their child’s health.
- Attempt to call ahead for patients who might be more vulnerable in waiting rooms or overcrowded hospitals.
Case 2
A 5-month-old baby presented to the ED with new-onset seizures. Immediate bedside blood work performed demonstrated a normal blood glucose, but the baby was profoundly hyponatremic. Upon asking the mother if the baby has had any vomiting, diarrhea, or difficulty tolerating feeds, she says that she has been diluting formula because all the stores were out of formula. Today, she gave the baby plain water because they were completely out of formula.
Takeaway: With economists estimating unemployment rates in the United States at 13% at press time (the worst since the Great Depression), many families may lack resources to purchase necessities.
- Even if families have the ability to purchase necessities, they may be difficult to find or unavailable (e.g., formula, medications, diapers).
- Consider reaching out to patients in your practice to ask about their ability to find essentials and with advice on what to do if they run out of formula or diapers, or who they should contact if they cannot refill a medication.
- Are you in a position to speak with your mayor or local council to ensure there are regulations on the hoarding of essential items?
- In a time when breast milk or formula is not available for children younger than 1 year of age, what will you recommend for families? There are no current American Academy of Pediatrics’ guidelines.
Case 3
A school-aged girl was helping her mother sanitize the home during the COVID-19 pandemic. She had her gloves on, her commercial antiseptic cleaner ready to go, but it was not spraying. She turned the bottle around to check the nozzle and sprayed herself in the eyes. The family presented to the ED for alkaline burn to her eyes, which required copious irrigation.
Takeaway: Children are spending more time in the house with access to button batteries, choking hazards, and cleaning supplies.
- Cleaning products can cause chemical burns. These products should not be used by young children.
Case 4
A school-aged boy arrived via emergency medical services (EMS) for altered mental status. He told his father he was feeling dizzy and then lost consciousness. EMS noticed that he had some tonic movements of his lower extremities, and when he arrived in the ED, he had eye deviation and was unresponsive.
Work-up ultimately demonstrated that this patient had a seizure and a dangerously elevated ethanol level from drinking an entire bottle of hand sanitizer. Hand sanitizer may contain high concentrations of ethyl alcohol or isopropyl alcohol, which when ingested can cause intoxication or poisoning.
Takeaway: Many products that we may view as harmless can be toxic if ingested in large amounts.
- Consider making a list of products that families may have acquired and have around the home during this COVID-19 pandemic and instruct families to make sure dangerous items (e.g., acetaminophen, aspirin, hand sanitizer, lighters, firearms, batteries) are locked up and/or out of reach of children.
- Make sure families know the Poison Control phone number (800-222-1222).
Case 5
An adolescent female currently being treated with immunosuppressants arrived from home with fever. Her medical history revealed that the patient’s guardian recently passed away from suspected COVID-19. The patient was tested and is herself found to be positive for COVID-19. The patient is currently being cared for by relatives who also live in the same home. They require extensive education and teaching regarding the patient’s medication regimen, while also dealing with the loss of their loved one and the fear of personal exposure.
Takeaway: Communicate with families – especially those with special health care needs – about issues of guardianship in case a child’s primary caretaker falls ill.
- Discuss with families about having easily accessible lists of medications and medical conditions.
- Involve social work and child life specialists to help children and their families deal with life-altering changes and losses suffered during this time, as well as fears related to mortality and exposure.
Case 6
A 3-year-old boy arrived covered in bruises and complaining of stomachache. While the mother denies any known abuse, she states that her significant other has been getting more and more “worked up having to deal with the child’s behavior all day every day.” The preschool the child previously attended has closed due to the pandemic.
Takeaway: Abuse is more common when the parents perceive that there is little community support and when families feel a lack of connection to the community.1 Huang et al. examined the relationship between the economy and nonaccidental trauma, showing a doubling in the rate of nonaccidental head trauma during economic recession.2
- Allow families to know that they are not alone and that child care is difficult
- Offer advice on what caretakers can do if they feel alone or at their mental or physical limit.
- Provide strategies on your practice’s website if a situation at home becomes tense and strained.
Case 7
An adolescent female arrived to the ED with increased suicidality. She normally follows with her psychiatrist once a month and her therapist once a week. Since the beginning of COVID-19 restrictions, she has been using telemedicine for her therapy visits. While previously doing well, she reports that her suicidal ideations have worsened because of feeling isolated from her friends now that school is out and she is not allowed to see them. Although compliant with her medications, her thoughts have increased to the point where she has to be admitted to inpatient psychiatry.
Takeaway: Anxiety, depression, and suicide may increase in a down economy. After the 2008 global economic crisis, rates of suicide drastically increased.3
- Recognize the limitations of telemedicine (technology limitations, patient cooperation, etc.)
- Social isolation may contribute to worsening mental health
- Know when to advise patients to seek in-person evaluation and care for medical and mental health concerns.
Pediatricians are at the forefront of preventative medicine. Families rely on pediatricians for trustworthy and accurate anticipatory guidance, a need that is only heightened during times of local and national stress. The social isolation, fear, and lack of resources accompanying this pandemic have serious consequences for our families. What can you and your practice do to keep children safe in the time of COVID-19?
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Hospital in Washington. Dr. Rachel Hatcliffe is an attending physician at the hospital. Neither physician had any relevant financial disclosures. Email Dr. DesPain and/or Dr. Hatcliffe at [email protected].
References
1. Child Dev. 1978;49:604-16.
2. J Neurosurg Pediatr 2011 Aug;8(2):171-6.
3. BMJ 2013;347:f5239.
As physicians and advanced practitioners, we have been preparing to face COVID-19 – anticipating increasing volumes of patients with fevers, cough, and shortness of breath, and potential surges in emergency departments (EDs) and primary care offices. Fortunately, while COVID-19 has demonstrated more mild symptoms in pediatric patients, the heightened public health fears and mandated social isolation have created some unforeseen consequences for pediatric patients. This article presents cases encountered over the course of 2 weeks in our ED that shed light on the unexpected ramifications of living in the time of a pandemic. These encounters should remind us as providers to be diligent and thorough in giving guidance to families during a time when face-to-face medicine has become increasingly difficult and limited.
These stories have been modified to protect patient confidentiality.
Case 1
A 2-week-old full-term infant arrived in the ED after having a fever for 48 hours. The patient’s mother reported that she had called the pediatrician yesterday to ask for advice on treating the fever and was instructed to give acetaminophen and bring the infant into the ED for testing.
When we asked mom why she did not bring the infant in yesterday, she stated that the fever went down with acetaminophen, and the baby was drinking well and urinating normally. Mostly, she was afraid to bring the child into the ED given concern for COVID-19; however, when the fever persisted today, she came in. During the work-up, the infant was noted to have focal seizures and was ultimately diagnosed with bacterial meningitis.
Takeaway: Families may be hesitant to follow pediatrician’s advice to seek medical attention at an ED or doctor’s office because of the fear of being exposed to COVID-19.
- If something is urgent or emergent, be sure to stress the importance to families that the advice is non-negotiable for their child’s health.
- Attempt to call ahead for patients who might be more vulnerable in waiting rooms or overcrowded hospitals.
Case 2
A 5-month-old baby presented to the ED with new-onset seizures. Immediate bedside blood work performed demonstrated a normal blood glucose, but the baby was profoundly hyponatremic. Upon asking the mother if the baby has had any vomiting, diarrhea, or difficulty tolerating feeds, she says that she has been diluting formula because all the stores were out of formula. Today, she gave the baby plain water because they were completely out of formula.
Takeaway: With economists estimating unemployment rates in the United States at 13% at press time (the worst since the Great Depression), many families may lack resources to purchase necessities.
- Even if families have the ability to purchase necessities, they may be difficult to find or unavailable (e.g., formula, medications, diapers).
- Consider reaching out to patients in your practice to ask about their ability to find essentials and with advice on what to do if they run out of formula or diapers, or who they should contact if they cannot refill a medication.
- Are you in a position to speak with your mayor or local council to ensure there are regulations on the hoarding of essential items?
- In a time when breast milk or formula is not available for children younger than 1 year of age, what will you recommend for families? There are no current American Academy of Pediatrics’ guidelines.
Case 3
A school-aged girl was helping her mother sanitize the home during the COVID-19 pandemic. She had her gloves on, her commercial antiseptic cleaner ready to go, but it was not spraying. She turned the bottle around to check the nozzle and sprayed herself in the eyes. The family presented to the ED for alkaline burn to her eyes, which required copious irrigation.
Takeaway: Children are spending more time in the house with access to button batteries, choking hazards, and cleaning supplies.
- Cleaning products can cause chemical burns. These products should not be used by young children.
Case 4
A school-aged boy arrived via emergency medical services (EMS) for altered mental status. He told his father he was feeling dizzy and then lost consciousness. EMS noticed that he had some tonic movements of his lower extremities, and when he arrived in the ED, he had eye deviation and was unresponsive.
Work-up ultimately demonstrated that this patient had a seizure and a dangerously elevated ethanol level from drinking an entire bottle of hand sanitizer. Hand sanitizer may contain high concentrations of ethyl alcohol or isopropyl alcohol, which when ingested can cause intoxication or poisoning.
Takeaway: Many products that we may view as harmless can be toxic if ingested in large amounts.
- Consider making a list of products that families may have acquired and have around the home during this COVID-19 pandemic and instruct families to make sure dangerous items (e.g., acetaminophen, aspirin, hand sanitizer, lighters, firearms, batteries) are locked up and/or out of reach of children.
- Make sure families know the Poison Control phone number (800-222-1222).
Case 5
An adolescent female currently being treated with immunosuppressants arrived from home with fever. Her medical history revealed that the patient’s guardian recently passed away from suspected COVID-19. The patient was tested and is herself found to be positive for COVID-19. The patient is currently being cared for by relatives who also live in the same home. They require extensive education and teaching regarding the patient’s medication regimen, while also dealing with the loss of their loved one and the fear of personal exposure.
Takeaway: Communicate with families – especially those with special health care needs – about issues of guardianship in case a child’s primary caretaker falls ill.
- Discuss with families about having easily accessible lists of medications and medical conditions.
- Involve social work and child life specialists to help children and their families deal with life-altering changes and losses suffered during this time, as well as fears related to mortality and exposure.
Case 6
A 3-year-old boy arrived covered in bruises and complaining of stomachache. While the mother denies any known abuse, she states that her significant other has been getting more and more “worked up having to deal with the child’s behavior all day every day.” The preschool the child previously attended has closed due to the pandemic.
Takeaway: Abuse is more common when the parents perceive that there is little community support and when families feel a lack of connection to the community.1 Huang et al. examined the relationship between the economy and nonaccidental trauma, showing a doubling in the rate of nonaccidental head trauma during economic recession.2
- Allow families to know that they are not alone and that child care is difficult
- Offer advice on what caretakers can do if they feel alone or at their mental or physical limit.
- Provide strategies on your practice’s website if a situation at home becomes tense and strained.
Case 7
An adolescent female arrived to the ED with increased suicidality. She normally follows with her psychiatrist once a month and her therapist once a week. Since the beginning of COVID-19 restrictions, she has been using telemedicine for her therapy visits. While previously doing well, she reports that her suicidal ideations have worsened because of feeling isolated from her friends now that school is out and she is not allowed to see them. Although compliant with her medications, her thoughts have increased to the point where she has to be admitted to inpatient psychiatry.
Takeaway: Anxiety, depression, and suicide may increase in a down economy. After the 2008 global economic crisis, rates of suicide drastically increased.3
- Recognize the limitations of telemedicine (technology limitations, patient cooperation, etc.)
- Social isolation may contribute to worsening mental health
- Know when to advise patients to seek in-person evaluation and care for medical and mental health concerns.
Pediatricians are at the forefront of preventative medicine. Families rely on pediatricians for trustworthy and accurate anticipatory guidance, a need that is only heightened during times of local and national stress. The social isolation, fear, and lack of resources accompanying this pandemic have serious consequences for our families. What can you and your practice do to keep children safe in the time of COVID-19?
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Hospital in Washington. Dr. Rachel Hatcliffe is an attending physician at the hospital. Neither physician had any relevant financial disclosures. Email Dr. DesPain and/or Dr. Hatcliffe at [email protected].
References
1. Child Dev. 1978;49:604-16.
2. J Neurosurg Pediatr 2011 Aug;8(2):171-6.
3. BMJ 2013;347:f5239.
As physicians and advanced practitioners, we have been preparing to face COVID-19 – anticipating increasing volumes of patients with fevers, cough, and shortness of breath, and potential surges in emergency departments (EDs) and primary care offices. Fortunately, while COVID-19 has demonstrated more mild symptoms in pediatric patients, the heightened public health fears and mandated social isolation have created some unforeseen consequences for pediatric patients. This article presents cases encountered over the course of 2 weeks in our ED that shed light on the unexpected ramifications of living in the time of a pandemic. These encounters should remind us as providers to be diligent and thorough in giving guidance to families during a time when face-to-face medicine has become increasingly difficult and limited.
These stories have been modified to protect patient confidentiality.
Case 1
A 2-week-old full-term infant arrived in the ED after having a fever for 48 hours. The patient’s mother reported that she had called the pediatrician yesterday to ask for advice on treating the fever and was instructed to give acetaminophen and bring the infant into the ED for testing.
When we asked mom why she did not bring the infant in yesterday, she stated that the fever went down with acetaminophen, and the baby was drinking well and urinating normally. Mostly, she was afraid to bring the child into the ED given concern for COVID-19; however, when the fever persisted today, she came in. During the work-up, the infant was noted to have focal seizures and was ultimately diagnosed with bacterial meningitis.
Takeaway: Families may be hesitant to follow pediatrician’s advice to seek medical attention at an ED or doctor’s office because of the fear of being exposed to COVID-19.
- If something is urgent or emergent, be sure to stress the importance to families that the advice is non-negotiable for their child’s health.
- Attempt to call ahead for patients who might be more vulnerable in waiting rooms or overcrowded hospitals.
Case 2
A 5-month-old baby presented to the ED with new-onset seizures. Immediate bedside blood work performed demonstrated a normal blood glucose, but the baby was profoundly hyponatremic. Upon asking the mother if the baby has had any vomiting, diarrhea, or difficulty tolerating feeds, she says that she has been diluting formula because all the stores were out of formula. Today, she gave the baby plain water because they were completely out of formula.
Takeaway: With economists estimating unemployment rates in the United States at 13% at press time (the worst since the Great Depression), many families may lack resources to purchase necessities.
- Even if families have the ability to purchase necessities, they may be difficult to find or unavailable (e.g., formula, medications, diapers).
- Consider reaching out to patients in your practice to ask about their ability to find essentials and with advice on what to do if they run out of formula or diapers, or who they should contact if they cannot refill a medication.
- Are you in a position to speak with your mayor or local council to ensure there are regulations on the hoarding of essential items?
- In a time when breast milk or formula is not available for children younger than 1 year of age, what will you recommend for families? There are no current American Academy of Pediatrics’ guidelines.
Case 3
A school-aged girl was helping her mother sanitize the home during the COVID-19 pandemic. She had her gloves on, her commercial antiseptic cleaner ready to go, but it was not spraying. She turned the bottle around to check the nozzle and sprayed herself in the eyes. The family presented to the ED for alkaline burn to her eyes, which required copious irrigation.
Takeaway: Children are spending more time in the house with access to button batteries, choking hazards, and cleaning supplies.
- Cleaning products can cause chemical burns. These products should not be used by young children.
Case 4
A school-aged boy arrived via emergency medical services (EMS) for altered mental status. He told his father he was feeling dizzy and then lost consciousness. EMS noticed that he had some tonic movements of his lower extremities, and when he arrived in the ED, he had eye deviation and was unresponsive.
Work-up ultimately demonstrated that this patient had a seizure and a dangerously elevated ethanol level from drinking an entire bottle of hand sanitizer. Hand sanitizer may contain high concentrations of ethyl alcohol or isopropyl alcohol, which when ingested can cause intoxication or poisoning.
Takeaway: Many products that we may view as harmless can be toxic if ingested in large amounts.
- Consider making a list of products that families may have acquired and have around the home during this COVID-19 pandemic and instruct families to make sure dangerous items (e.g., acetaminophen, aspirin, hand sanitizer, lighters, firearms, batteries) are locked up and/or out of reach of children.
- Make sure families know the Poison Control phone number (800-222-1222).
Case 5
An adolescent female currently being treated with immunosuppressants arrived from home with fever. Her medical history revealed that the patient’s guardian recently passed away from suspected COVID-19. The patient was tested and is herself found to be positive for COVID-19. The patient is currently being cared for by relatives who also live in the same home. They require extensive education and teaching regarding the patient’s medication regimen, while also dealing with the loss of their loved one and the fear of personal exposure.
Takeaway: Communicate with families – especially those with special health care needs – about issues of guardianship in case a child’s primary caretaker falls ill.
- Discuss with families about having easily accessible lists of medications and medical conditions.
- Involve social work and child life specialists to help children and their families deal with life-altering changes and losses suffered during this time, as well as fears related to mortality and exposure.
Case 6
A 3-year-old boy arrived covered in bruises and complaining of stomachache. While the mother denies any known abuse, she states that her significant other has been getting more and more “worked up having to deal with the child’s behavior all day every day.” The preschool the child previously attended has closed due to the pandemic.
Takeaway: Abuse is more common when the parents perceive that there is little community support and when families feel a lack of connection to the community.1 Huang et al. examined the relationship between the economy and nonaccidental trauma, showing a doubling in the rate of nonaccidental head trauma during economic recession.2
- Allow families to know that they are not alone and that child care is difficult
- Offer advice on what caretakers can do if they feel alone or at their mental or physical limit.
- Provide strategies on your practice’s website if a situation at home becomes tense and strained.
Case 7
An adolescent female arrived to the ED with increased suicidality. She normally follows with her psychiatrist once a month and her therapist once a week. Since the beginning of COVID-19 restrictions, she has been using telemedicine for her therapy visits. While previously doing well, she reports that her suicidal ideations have worsened because of feeling isolated from her friends now that school is out and she is not allowed to see them. Although compliant with her medications, her thoughts have increased to the point where she has to be admitted to inpatient psychiatry.
Takeaway: Anxiety, depression, and suicide may increase in a down economy. After the 2008 global economic crisis, rates of suicide drastically increased.3
- Recognize the limitations of telemedicine (technology limitations, patient cooperation, etc.)
- Social isolation may contribute to worsening mental health
- Know when to advise patients to seek in-person evaluation and care for medical and mental health concerns.
Pediatricians are at the forefront of preventative medicine. Families rely on pediatricians for trustworthy and accurate anticipatory guidance, a need that is only heightened during times of local and national stress. The social isolation, fear, and lack of resources accompanying this pandemic have serious consequences for our families. What can you and your practice do to keep children safe in the time of COVID-19?
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Hospital in Washington. Dr. Rachel Hatcliffe is an attending physician at the hospital. Neither physician had any relevant financial disclosures. Email Dr. DesPain and/or Dr. Hatcliffe at [email protected].
References
1. Child Dev. 1978;49:604-16.
2. J Neurosurg Pediatr 2011 Aug;8(2):171-6.
3. BMJ 2013;347:f5239.