User login
51-year-old woman • history of Graves disease • general fatigue, palpitations, and hand tremors • Dx?
THE CASE
A 51-year-old Japanese woman presented with fever, sore throat, and dyspnea of less than 1 day’s duration. Although she had developed general fatigue, palpitations, and tremors of the hands 2 months earlier, she had not sought medical care.
Her medical history included Graves disease, which had been diagnosed 13 years earlier. She reported that her only medication was methimazole 10 mg/d. She did not have any family history of endocrinopathies or hematologic diseases.
Physical examination revealed a body temperature of 99.7 °F; heart rate, 130 beats/min; blood pressure, 182/62 mm Hg; respiratory rate, 46 breaths/min; and oxygen saturation, 100% on room air. Pharyngeal erythema was seen. Lung sounds were clear. The patient had tremors in her hands, tenderness of the thyroid gland, and exophthalmos. No leg edema or jugular vein distension was seen.
Laboratory tests indicated hyperthyroidism, with a thyroid-stimulating hormone level < 0.01 µIU/mL (normal range, 0.5-5 µIU/mL); free T3 level, 4.87 pg/mL (normal range, 2.3-4.3 pg/mL); and free T4 level, 2.97 ng/dL (normal range, 0.9-1.7 ng/dL). The patient also had a white blood cell (WBC) count of 1020 cells/µL (normal range, 3500-9000 cells/µL) and neutrophil count of 5 cells/µL (normal range, 1500-6500 cells/µL).
Other blood cell counts were normal, and a chest x-ray did not reveal any abnormal findings. In addition, there was no evidence to suggest hematologic malignancies or congenital neutropenia.
THE DIAGNOSIS
Based on the patient’s low WBC and neutrophil counts, agranulocytosis due to antithyroid drug therapy was suspected; however, this diagnosis would be highly unusual in the context of a 13-year history of therapy. Further history taking revealed that, because of her lack of financial means, unstable living conditions, and lack of understanding of the necessity for medication adherence, the patient had not taken methimazole
In consideration of these factors, a diagnosis of exacerbation of hyperthyroidism and agranulocytosis (due to methimazole restart and upper respiratory infection) was made.
Continue to: DISCUSSION
DISCUSSION
Agranulocytosis is a severe adverse event of antithyroid agents and requires prompt diagnosis and treatment. In a 26-year study at one clinic, it occurred in approximately 0.4% of patients taking antithyroid agents.1 The possible mechanisms of agranulocytosis are the direct toxicity of drugs and immune-mediated responses.2 Older age, female sex, and some HLA genotypes are reported to be associated with susceptibility to agranulocytosis.2
Although the development of agranulocytosis tends to be dose related, a small dose of antithyroid agent can sometimes cause the condition.3,4 It usually occurs within the first 3 months of treatment initiation, but occasionally patients develop agranulocytosis after long-term therapy.5 Interruption and subsequent resumption of the same antithyroid drug treatment also can be a risk factor for agranulocytosis, as in this case.5
Treatment includes drug cessation, administration of broad-spectrum antibiotics if infection is suspected, and granulocyte-colony stimulating factor (G-CSF) therapy.5
Our patient was hospitalized, and methimazole was stopped immediately. Administration of potassium iodide 50 mg/d and G-CSF was started. Meropenem 3 g/d also was administered for neutropenic fever.
The patient’s condition improved, and her WBC count increased to 1640 cells/µL on Day 8 and 10,890 cells/µL on Day 9. G-CSF was stopped on Day 12 and meropenem on Day 13. Bone marrow aspiration was not performed because of improvement in lab values and her overall condition. Although monitoring of WBC count during methimazole therapy is controversial,5 we decided to routinely monitor this patient due to the possibility of drug cross-reactivity.
Continue to: Despite repeated explanations...
Despite repeated explanations that it was dangerous for a patient who had developed agranulocytosis to take another antithyroid medication, the patient refused surgical treatment or radioiodine ablation because of her financial situation. (While all Japanese citizens are covered by a national health insurance program, patients ages 6 to 70 years are required to pay approximately 30% of medical and pharmaceutical costs.) On Day 21, potassium iodide was stopped, and propylthiouracil 300 mg/d was administered with careful follow-up. Agranulocytosis did not recur.
Immediate problem solved, but what about the future?
During her hospital stay, the medical team spoke with the patient many times, during which she expressed anxiety about her health conditions and the difficulties that she had experienced in her life. The clinicians acknowledged her concerns and assured the patient of their continuing commitment to her well-being even after discharge. The patient also was advised that she should take her medication as prescribed and that if she had a fever or sore throat, she should stop the medication and seek medical care as soon as possible. The patient accepted the medical team’s advice and expressed hope for the future.
Conversations about medication adherence. In 1 survey, about 60% of patients taking antithyroid drugs were unfamiliar with the symptoms of agranulocytosis.6 To deliver safe and effective treatment and detect conditions such as agranulocytosis at an early stage, clinicians must communicate clearly with patients who have hyperthyroidism, providing sufficient explanation and ensuring understanding on the patient’s part.
Patients may be reluctant to provide the details of medication adherence.7 Although it is common for patients to need services for socioeconomic issues,8 health care professionals sometimes fail to adequately discuss these issues with patients, especially if the patients are marginalized and/or have lower economic status.9 Cases such as ours underscore the importance of improving clinicians’ awareness and sensitivity to patients’ socioeconomic challenges.10,11
Our patient received information about welfare and other government services from a medical social worker during her hospital stay. She also was informed that she could seek assistance from medical social workers in the future if needed.
Continue to: The patient was discharged...
The patient was discharged on Day 28. After discharge, she took propylthiouracil as prescribed (300 mg/d), and her Graves disease was well controlled. Outpatient follow-up visits were performed every 1 or 2 months. No adverse events of propylthiouracil were seen in the ensuing time.
THE TAKEAWAY
Patients with chronic conditions sometimes discontinue medications, and they may not talk about it with their medical team, especially if they have socioeconomic or other difficulties in their lives. Clinicians should consider medication nonadherence and its risk factors when patients with chronic conditions develop unexpected adverse events.
We thank Jane Charbonneau, DVM, from Edanz for doing an English-language review of a draft of this manuscript.
CORRESPONDENCE
Takuya Maejima, MD, Department of General Medicine and Primary Care, University of Tsukuba Hospital, 2-1-1 Amakubo, Tsukuba, Ibaraki 305-8576 Japan; [email protected]
1. Tajiri J, Noguchi S. Antithyroid drug-induced agranulocytosis: special reference to normal white blood cell count agranulocytosis. Thyroid. 2004;14:459-462. doi: 10.1089/105072504323150787
2. Vicente N, Cardoso L, Barros L, et al. Antithyroid drug-induced agranulocytosis: state of the art on diagnosis and management. Drugs R D. 2017;17:91-96. doi: 10.1007/s40268-017-0172-1
3. Takata K, Kubota S, Fukata S, et al. Methimazole-induced agranulocytosis in patients with Graves’ disease is more frequent with an initial dose of 30 mg daily than with 15 mg daily. Thyroid. 2009;19:559-563. doi: 10.1089/thy.2008.0364
4. Tsuboi K, Ueshiba H, Shimojo M, et al. The relation of initial methimazole dose to the incidence of methimazole-induced agranulocytosis in patients with Graves’ disease. Endocr J. 2007;54:39-43. doi: 10.1507/endocrj.k05-068
5. Burch HB, Cooper DS. Management of Graves disease: a review. J Am Med Assoc. 2015;314:2544-2554. doi: 10.1001/jama.2015.16535
6. Robinson J, Richardson M, Hickey J, et al. Patient knowledge of antithyroid drug-induced agranulocytosis. Eur Thyroid J. 2014;3:245-251. doi: https://doi.org/10.1159/000367990
7. Kini V, Ho PM. Interventions to improve medication adherence: a review. J Am Med Assoc. 2018;320:2461-2473. doi: 10.1001/jama.2018.19271
8. Vest JR, Grannis SJ, Haut DP, et al. Using structured and unstructured data to identify patients’ need for services that address the social determinants of health. Int J Med Inform. 2017;107:101-106. doi: 10.1016/j.ijmedinf.2017.09.008
9. Willems S, De Maesschalck S, Deveugele M, et al. Socio-economic status of the patient and doctor-patient communication: does it make a difference? Patient Educ Couns. 2005;56:139-146. doi: 10.1016/j.pec.2004.02.011
10. The College of Family Physicians of Canada. Best advice: social determinants of health. Accessed September 15, 2023. https://patientsmedicalhome.ca/resources/best-advice-guides/best-advice-guide-social-determinants-health/
11. Hunter K, Thomson B. A scoping review of social determinants of health curricula in post-graduate medical education. Can Med Educ J. 2019;10:e61-e71. doi: 10.36834/cmej.61709
THE CASE
A 51-year-old Japanese woman presented with fever, sore throat, and dyspnea of less than 1 day’s duration. Although she had developed general fatigue, palpitations, and tremors of the hands 2 months earlier, she had not sought medical care.
Her medical history included Graves disease, which had been diagnosed 13 years earlier. She reported that her only medication was methimazole 10 mg/d. She did not have any family history of endocrinopathies or hematologic diseases.
Physical examination revealed a body temperature of 99.7 °F; heart rate, 130 beats/min; blood pressure, 182/62 mm Hg; respiratory rate, 46 breaths/min; and oxygen saturation, 100% on room air. Pharyngeal erythema was seen. Lung sounds were clear. The patient had tremors in her hands, tenderness of the thyroid gland, and exophthalmos. No leg edema or jugular vein distension was seen.
Laboratory tests indicated hyperthyroidism, with a thyroid-stimulating hormone level < 0.01 µIU/mL (normal range, 0.5-5 µIU/mL); free T3 level, 4.87 pg/mL (normal range, 2.3-4.3 pg/mL); and free T4 level, 2.97 ng/dL (normal range, 0.9-1.7 ng/dL). The patient also had a white blood cell (WBC) count of 1020 cells/µL (normal range, 3500-9000 cells/µL) and neutrophil count of 5 cells/µL (normal range, 1500-6500 cells/µL).
Other blood cell counts were normal, and a chest x-ray did not reveal any abnormal findings. In addition, there was no evidence to suggest hematologic malignancies or congenital neutropenia.
THE DIAGNOSIS
Based on the patient’s low WBC and neutrophil counts, agranulocytosis due to antithyroid drug therapy was suspected; however, this diagnosis would be highly unusual in the context of a 13-year history of therapy. Further history taking revealed that, because of her lack of financial means, unstable living conditions, and lack of understanding of the necessity for medication adherence, the patient had not taken methimazole
In consideration of these factors, a diagnosis of exacerbation of hyperthyroidism and agranulocytosis (due to methimazole restart and upper respiratory infection) was made.
Continue to: DISCUSSION
DISCUSSION
Agranulocytosis is a severe adverse event of antithyroid agents and requires prompt diagnosis and treatment. In a 26-year study at one clinic, it occurred in approximately 0.4% of patients taking antithyroid agents.1 The possible mechanisms of agranulocytosis are the direct toxicity of drugs and immune-mediated responses.2 Older age, female sex, and some HLA genotypes are reported to be associated with susceptibility to agranulocytosis.2
Although the development of agranulocytosis tends to be dose related, a small dose of antithyroid agent can sometimes cause the condition.3,4 It usually occurs within the first 3 months of treatment initiation, but occasionally patients develop agranulocytosis after long-term therapy.5 Interruption and subsequent resumption of the same antithyroid drug treatment also can be a risk factor for agranulocytosis, as in this case.5
Treatment includes drug cessation, administration of broad-spectrum antibiotics if infection is suspected, and granulocyte-colony stimulating factor (G-CSF) therapy.5
Our patient was hospitalized, and methimazole was stopped immediately. Administration of potassium iodide 50 mg/d and G-CSF was started. Meropenem 3 g/d also was administered for neutropenic fever.
The patient’s condition improved, and her WBC count increased to 1640 cells/µL on Day 8 and 10,890 cells/µL on Day 9. G-CSF was stopped on Day 12 and meropenem on Day 13. Bone marrow aspiration was not performed because of improvement in lab values and her overall condition. Although monitoring of WBC count during methimazole therapy is controversial,5 we decided to routinely monitor this patient due to the possibility of drug cross-reactivity.
Continue to: Despite repeated explanations...
Despite repeated explanations that it was dangerous for a patient who had developed agranulocytosis to take another antithyroid medication, the patient refused surgical treatment or radioiodine ablation because of her financial situation. (While all Japanese citizens are covered by a national health insurance program, patients ages 6 to 70 years are required to pay approximately 30% of medical and pharmaceutical costs.) On Day 21, potassium iodide was stopped, and propylthiouracil 300 mg/d was administered with careful follow-up. Agranulocytosis did not recur.
Immediate problem solved, but what about the future?
During her hospital stay, the medical team spoke with the patient many times, during which she expressed anxiety about her health conditions and the difficulties that she had experienced in her life. The clinicians acknowledged her concerns and assured the patient of their continuing commitment to her well-being even after discharge. The patient also was advised that she should take her medication as prescribed and that if she had a fever or sore throat, she should stop the medication and seek medical care as soon as possible. The patient accepted the medical team’s advice and expressed hope for the future.
Conversations about medication adherence. In 1 survey, about 60% of patients taking antithyroid drugs were unfamiliar with the symptoms of agranulocytosis.6 To deliver safe and effective treatment and detect conditions such as agranulocytosis at an early stage, clinicians must communicate clearly with patients who have hyperthyroidism, providing sufficient explanation and ensuring understanding on the patient’s part.
Patients may be reluctant to provide the details of medication adherence.7 Although it is common for patients to need services for socioeconomic issues,8 health care professionals sometimes fail to adequately discuss these issues with patients, especially if the patients are marginalized and/or have lower economic status.9 Cases such as ours underscore the importance of improving clinicians’ awareness and sensitivity to patients’ socioeconomic challenges.10,11
Our patient received information about welfare and other government services from a medical social worker during her hospital stay. She also was informed that she could seek assistance from medical social workers in the future if needed.
Continue to: The patient was discharged...
The patient was discharged on Day 28. After discharge, she took propylthiouracil as prescribed (300 mg/d), and her Graves disease was well controlled. Outpatient follow-up visits were performed every 1 or 2 months. No adverse events of propylthiouracil were seen in the ensuing time.
THE TAKEAWAY
Patients with chronic conditions sometimes discontinue medications, and they may not talk about it with their medical team, especially if they have socioeconomic or other difficulties in their lives. Clinicians should consider medication nonadherence and its risk factors when patients with chronic conditions develop unexpected adverse events.
We thank Jane Charbonneau, DVM, from Edanz for doing an English-language review of a draft of this manuscript.
CORRESPONDENCE
Takuya Maejima, MD, Department of General Medicine and Primary Care, University of Tsukuba Hospital, 2-1-1 Amakubo, Tsukuba, Ibaraki 305-8576 Japan; [email protected]
THE CASE
A 51-year-old Japanese woman presented with fever, sore throat, and dyspnea of less than 1 day’s duration. Although she had developed general fatigue, palpitations, and tremors of the hands 2 months earlier, she had not sought medical care.
Her medical history included Graves disease, which had been diagnosed 13 years earlier. She reported that her only medication was methimazole 10 mg/d. She did not have any family history of endocrinopathies or hematologic diseases.
Physical examination revealed a body temperature of 99.7 °F; heart rate, 130 beats/min; blood pressure, 182/62 mm Hg; respiratory rate, 46 breaths/min; and oxygen saturation, 100% on room air. Pharyngeal erythema was seen. Lung sounds were clear. The patient had tremors in her hands, tenderness of the thyroid gland, and exophthalmos. No leg edema or jugular vein distension was seen.
Laboratory tests indicated hyperthyroidism, with a thyroid-stimulating hormone level < 0.01 µIU/mL (normal range, 0.5-5 µIU/mL); free T3 level, 4.87 pg/mL (normal range, 2.3-4.3 pg/mL); and free T4 level, 2.97 ng/dL (normal range, 0.9-1.7 ng/dL). The patient also had a white blood cell (WBC) count of 1020 cells/µL (normal range, 3500-9000 cells/µL) and neutrophil count of 5 cells/µL (normal range, 1500-6500 cells/µL).
Other blood cell counts were normal, and a chest x-ray did not reveal any abnormal findings. In addition, there was no evidence to suggest hematologic malignancies or congenital neutropenia.
THE DIAGNOSIS
Based on the patient’s low WBC and neutrophil counts, agranulocytosis due to antithyroid drug therapy was suspected; however, this diagnosis would be highly unusual in the context of a 13-year history of therapy. Further history taking revealed that, because of her lack of financial means, unstable living conditions, and lack of understanding of the necessity for medication adherence, the patient had not taken methimazole
In consideration of these factors, a diagnosis of exacerbation of hyperthyroidism and agranulocytosis (due to methimazole restart and upper respiratory infection) was made.
Continue to: DISCUSSION
DISCUSSION
Agranulocytosis is a severe adverse event of antithyroid agents and requires prompt diagnosis and treatment. In a 26-year study at one clinic, it occurred in approximately 0.4% of patients taking antithyroid agents.1 The possible mechanisms of agranulocytosis are the direct toxicity of drugs and immune-mediated responses.2 Older age, female sex, and some HLA genotypes are reported to be associated with susceptibility to agranulocytosis.2
Although the development of agranulocytosis tends to be dose related, a small dose of antithyroid agent can sometimes cause the condition.3,4 It usually occurs within the first 3 months of treatment initiation, but occasionally patients develop agranulocytosis after long-term therapy.5 Interruption and subsequent resumption of the same antithyroid drug treatment also can be a risk factor for agranulocytosis, as in this case.5
Treatment includes drug cessation, administration of broad-spectrum antibiotics if infection is suspected, and granulocyte-colony stimulating factor (G-CSF) therapy.5
Our patient was hospitalized, and methimazole was stopped immediately. Administration of potassium iodide 50 mg/d and G-CSF was started. Meropenem 3 g/d also was administered for neutropenic fever.
The patient’s condition improved, and her WBC count increased to 1640 cells/µL on Day 8 and 10,890 cells/µL on Day 9. G-CSF was stopped on Day 12 and meropenem on Day 13. Bone marrow aspiration was not performed because of improvement in lab values and her overall condition. Although monitoring of WBC count during methimazole therapy is controversial,5 we decided to routinely monitor this patient due to the possibility of drug cross-reactivity.
Continue to: Despite repeated explanations...
Despite repeated explanations that it was dangerous for a patient who had developed agranulocytosis to take another antithyroid medication, the patient refused surgical treatment or radioiodine ablation because of her financial situation. (While all Japanese citizens are covered by a national health insurance program, patients ages 6 to 70 years are required to pay approximately 30% of medical and pharmaceutical costs.) On Day 21, potassium iodide was stopped, and propylthiouracil 300 mg/d was administered with careful follow-up. Agranulocytosis did not recur.
Immediate problem solved, but what about the future?
During her hospital stay, the medical team spoke with the patient many times, during which she expressed anxiety about her health conditions and the difficulties that she had experienced in her life. The clinicians acknowledged her concerns and assured the patient of their continuing commitment to her well-being even after discharge. The patient also was advised that she should take her medication as prescribed and that if she had a fever or sore throat, she should stop the medication and seek medical care as soon as possible. The patient accepted the medical team’s advice and expressed hope for the future.
Conversations about medication adherence. In 1 survey, about 60% of patients taking antithyroid drugs were unfamiliar with the symptoms of agranulocytosis.6 To deliver safe and effective treatment and detect conditions such as agranulocytosis at an early stage, clinicians must communicate clearly with patients who have hyperthyroidism, providing sufficient explanation and ensuring understanding on the patient’s part.
Patients may be reluctant to provide the details of medication adherence.7 Although it is common for patients to need services for socioeconomic issues,8 health care professionals sometimes fail to adequately discuss these issues with patients, especially if the patients are marginalized and/or have lower economic status.9 Cases such as ours underscore the importance of improving clinicians’ awareness and sensitivity to patients’ socioeconomic challenges.10,11
Our patient received information about welfare and other government services from a medical social worker during her hospital stay. She also was informed that she could seek assistance from medical social workers in the future if needed.
Continue to: The patient was discharged...
The patient was discharged on Day 28. After discharge, she took propylthiouracil as prescribed (300 mg/d), and her Graves disease was well controlled. Outpatient follow-up visits were performed every 1 or 2 months. No adverse events of propylthiouracil were seen in the ensuing time.
THE TAKEAWAY
Patients with chronic conditions sometimes discontinue medications, and they may not talk about it with their medical team, especially if they have socioeconomic or other difficulties in their lives. Clinicians should consider medication nonadherence and its risk factors when patients with chronic conditions develop unexpected adverse events.
We thank Jane Charbonneau, DVM, from Edanz for doing an English-language review of a draft of this manuscript.
CORRESPONDENCE
Takuya Maejima, MD, Department of General Medicine and Primary Care, University of Tsukuba Hospital, 2-1-1 Amakubo, Tsukuba, Ibaraki 305-8576 Japan; [email protected]
1. Tajiri J, Noguchi S. Antithyroid drug-induced agranulocytosis: special reference to normal white blood cell count agranulocytosis. Thyroid. 2004;14:459-462. doi: 10.1089/105072504323150787
2. Vicente N, Cardoso L, Barros L, et al. Antithyroid drug-induced agranulocytosis: state of the art on diagnosis and management. Drugs R D. 2017;17:91-96. doi: 10.1007/s40268-017-0172-1
3. Takata K, Kubota S, Fukata S, et al. Methimazole-induced agranulocytosis in patients with Graves’ disease is more frequent with an initial dose of 30 mg daily than with 15 mg daily. Thyroid. 2009;19:559-563. doi: 10.1089/thy.2008.0364
4. Tsuboi K, Ueshiba H, Shimojo M, et al. The relation of initial methimazole dose to the incidence of methimazole-induced agranulocytosis in patients with Graves’ disease. Endocr J. 2007;54:39-43. doi: 10.1507/endocrj.k05-068
5. Burch HB, Cooper DS. Management of Graves disease: a review. J Am Med Assoc. 2015;314:2544-2554. doi: 10.1001/jama.2015.16535
6. Robinson J, Richardson M, Hickey J, et al. Patient knowledge of antithyroid drug-induced agranulocytosis. Eur Thyroid J. 2014;3:245-251. doi: https://doi.org/10.1159/000367990
7. Kini V, Ho PM. Interventions to improve medication adherence: a review. J Am Med Assoc. 2018;320:2461-2473. doi: 10.1001/jama.2018.19271
8. Vest JR, Grannis SJ, Haut DP, et al. Using structured and unstructured data to identify patients’ need for services that address the social determinants of health. Int J Med Inform. 2017;107:101-106. doi: 10.1016/j.ijmedinf.2017.09.008
9. Willems S, De Maesschalck S, Deveugele M, et al. Socio-economic status of the patient and doctor-patient communication: does it make a difference? Patient Educ Couns. 2005;56:139-146. doi: 10.1016/j.pec.2004.02.011
10. The College of Family Physicians of Canada. Best advice: social determinants of health. Accessed September 15, 2023. https://patientsmedicalhome.ca/resources/best-advice-guides/best-advice-guide-social-determinants-health/
11. Hunter K, Thomson B. A scoping review of social determinants of health curricula in post-graduate medical education. Can Med Educ J. 2019;10:e61-e71. doi: 10.36834/cmej.61709
1. Tajiri J, Noguchi S. Antithyroid drug-induced agranulocytosis: special reference to normal white blood cell count agranulocytosis. Thyroid. 2004;14:459-462. doi: 10.1089/105072504323150787
2. Vicente N, Cardoso L, Barros L, et al. Antithyroid drug-induced agranulocytosis: state of the art on diagnosis and management. Drugs R D. 2017;17:91-96. doi: 10.1007/s40268-017-0172-1
3. Takata K, Kubota S, Fukata S, et al. Methimazole-induced agranulocytosis in patients with Graves’ disease is more frequent with an initial dose of 30 mg daily than with 15 mg daily. Thyroid. 2009;19:559-563. doi: 10.1089/thy.2008.0364
4. Tsuboi K, Ueshiba H, Shimojo M, et al. The relation of initial methimazole dose to the incidence of methimazole-induced agranulocytosis in patients with Graves’ disease. Endocr J. 2007;54:39-43. doi: 10.1507/endocrj.k05-068
5. Burch HB, Cooper DS. Management of Graves disease: a review. J Am Med Assoc. 2015;314:2544-2554. doi: 10.1001/jama.2015.16535
6. Robinson J, Richardson M, Hickey J, et al. Patient knowledge of antithyroid drug-induced agranulocytosis. Eur Thyroid J. 2014;3:245-251. doi: https://doi.org/10.1159/000367990
7. Kini V, Ho PM. Interventions to improve medication adherence: a review. J Am Med Assoc. 2018;320:2461-2473. doi: 10.1001/jama.2018.19271
8. Vest JR, Grannis SJ, Haut DP, et al. Using structured and unstructured data to identify patients’ need for services that address the social determinants of health. Int J Med Inform. 2017;107:101-106. doi: 10.1016/j.ijmedinf.2017.09.008
9. Willems S, De Maesschalck S, Deveugele M, et al. Socio-economic status of the patient and doctor-patient communication: does it make a difference? Patient Educ Couns. 2005;56:139-146. doi: 10.1016/j.pec.2004.02.011
10. The College of Family Physicians of Canada. Best advice: social determinants of health. Accessed September 15, 2023. https://patientsmedicalhome.ca/resources/best-advice-guides/best-advice-guide-social-determinants-health/
11. Hunter K, Thomson B. A scoping review of social determinants of health curricula in post-graduate medical education. Can Med Educ J. 2019;10:e61-e71. doi: 10.36834/cmej.61709
► History of Graves disease
► General fatigue, palpitations, and hand tremors
Painless nodules on legs
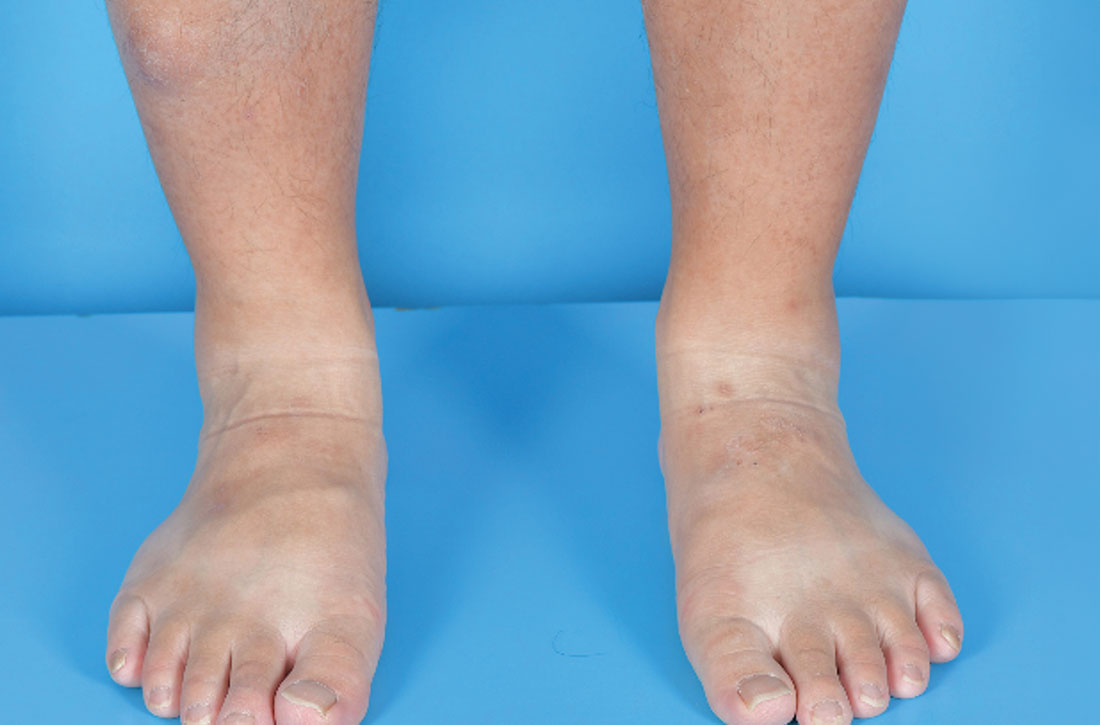
A 34-YEAR-OLD MAN presented with a 6-month history of asymptomatic, progressively enlarging subcutaneous nodules over his bilateral lower legs. He denied any history of injury, and there was no bleeding or discharge. The patient had a history of Graves disease that had been treated with radioiodine therapy 2 years prior, followed by thyroxine replacement (150 mcg/d, 5 d/wk and 125 mcg/d, 2 d/wk). At the time of presentation, his thyroid function tests indicated subclinical hypothyroidism: free T4, 21.2 pmol/L (normal range, 11.8-24.6 pmol/L) and thyroid-stimulating hormone (TSH), 14.07 mIU/L (normal range, 0.27-4.2 mIU/L).
Examination revealed nontender, soft brown nodules over the bilateral shins, with minimal overlying lichenification (FIGURE 1). There was no peau d’orange (orange peel) appearance to suggest significant edema. A punch biopsy was performed.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pretibial myxedema
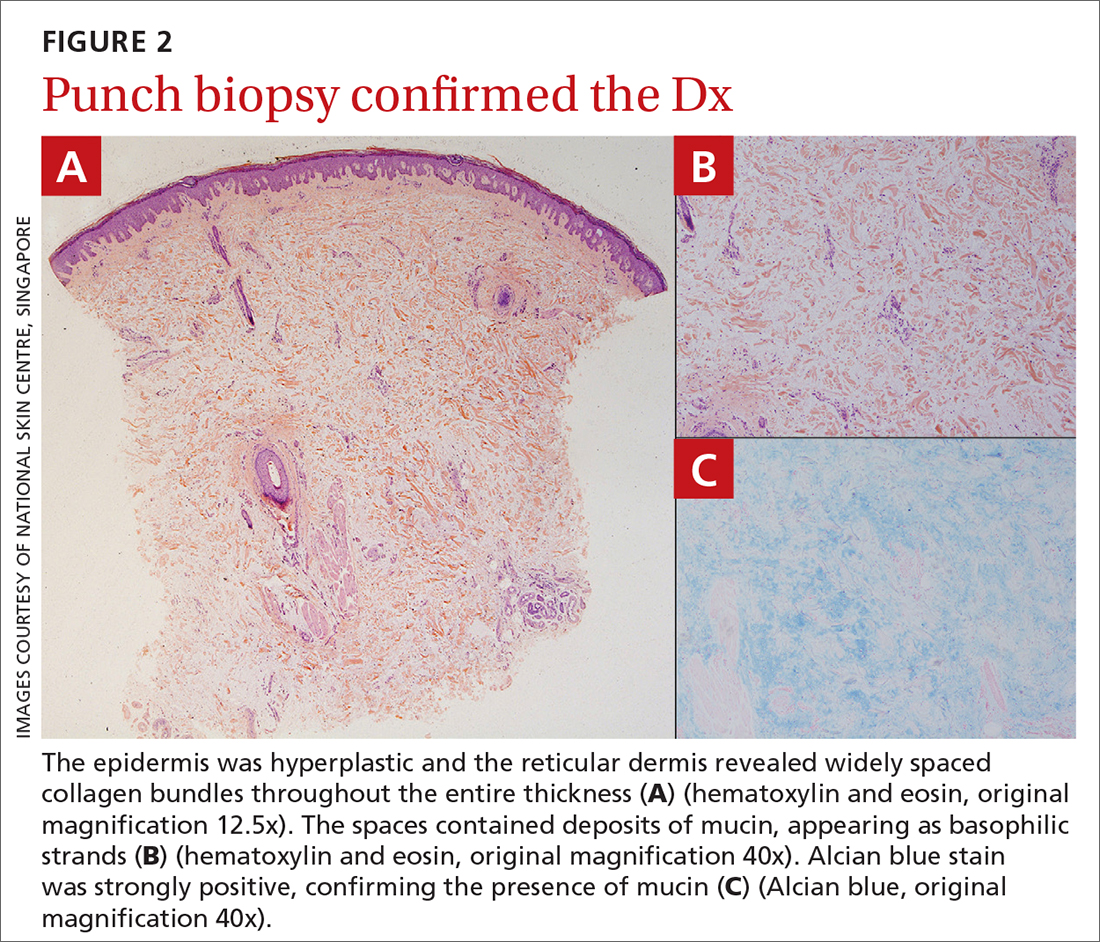
The patient’s history, paired with the results of the punch biopsy, were consistent with a diagnosis of pretibial myxedema, part of the triad of Graves disease along with thyroid ophthalmopathy and acropachy (soft-tissue swelling of the hands and clubbing of the fingers). Histopathologic findings revealed wide separation of collagen bundles throughout the entire reticular dermis without fibroplasia (FIGURE 2A). The spaces contained basophilic strands (FIGURE 2B), and the strands stained strongly positive on Alcian blue (FIGURE 2C), confirming the presence of dermal mucin. Widely separated collagen fibers and deposited mucin are indicative of pretibial myxedema. No granulomas or lymphoid proliferations were seen.
The pathogenesis of pretibial myxedema is widely postulated to be due to the stimulation of dermal fibroblasts by anti–TSH antibodies, causing overproduction of glycosaminoglycans and hyaluronic acid1 and obstructing lymphatic microcirculation, resulting in nonpitting edema.2
There are 5 distinct clinical variants of pretibial myxedema1,3:
- The diffuse form is the most common. It manifests on the lower leg with hard, nonpitting edema and cutaneous thickening.
- The plaque form manifests on the lower leg as well-demarcated erythematous or pigmented flat-topped lesions.
- The nodular form, which our patient had, typically manifests on the lower leg as well-demarcated erythematous, pigmented, or skin-colored raised, solid lesions. There may be 1 lesion or several.
- The mixed form manifests as 2 or more of the other variants.
- The elephantiasic form is the rarest and the most severe. There are widespread swollen nodules and plaques on the lower legs and/or arms.
A rare, late manifestation
Although pathognomonic for Graves disease, pretibial myxedema is a late manifestation that occurs in less than 5% of these patients.4 The most common site of involvement is the pretibial region, although less common sites include the face, arms, shoulders, abdomen, pinna, and the location of previous scars.4
While pretibial myxedema usually is associated with hyperthyroidism, it can occur after treatment (as was the case here), while the patient is in a euthyroid or hypothyroid state. Radioiodine therapy has been reported to be a trigger for pretibial myxedema in 1 case report, although the pathophysiology is not fully understood.5
Continue to: More serious conditions must be ruled out
More serious conditions must be ruled out
The differential for painless nodules includes cutaneous lymphoma and atypical infections of fungal or mycobacterial etiology.
Cutaneous lymphoma that manifests with leg tumors includes primary cutaneous anaplastic CD30+ large cell lymphoma (PCALCL) and primary cutaneous diffuse large B-cell lymphoma, leg type (PCDLBL-LT). The former may occur in young patients, whereas the latter tends to manifest in the elderly. Biopsy shows a neoplastic proliferation of atypical lymphocytes within the dermis,6 differing from our case.
Atypical infections may be detected through bacterial, mycobacterial, or fungal cultures, and may be accompanied by elevated inflammatory markers or other systemic symptoms of the infection, setting it apart from pretibial myxedema.
Treatment is simple and noninvasive
Pretibial myxedema is usually asymptomatic, with minimal morbidity. The nodular variant may resolve spontaneously; thus, therapeutic management often is reserved for severe cases or for those with cosmetic concerns. Treatment options include mid- to high-potency topical corticosteroids with an occlusive dressing for 1 to 2 weeks (or until resolution) or an intralesional triamcinolone injection (5-10 mg/mL, single or monthly until resolution), compression stockings, and pneumatic compression.2
This patient was treated with a single intralesional injection of triamcinolone 10 mg/mL. The nodules resolved within a month.
1. Thammarucha S, Sudtikoonaseth P. Nodular pretibial myxedema with Graves’ disease: a case report. Thai J Dermatol. 2021;37:30-36.
2. Singla M, Gupta A. Nodular thyroid dermopathy: not a hallmark of Graves’ disease. Am J Med. 2019;132:e521-e522. doi: 10.1016/j.amjmed.2018.11.004
3. Lan C, Wang Y, Zeng X, et al. Morphological diversity of pretibial myxedema and its mechanism of evolving process and outcome: a retrospective study of 216 cases. J Thyroid Res. 2016:2016:265217
4. doi: 10.1155/2016/2652174 4. Patil MM, Kamalanathan S, Sahoo J, et al. Pretibial myxedema. QJM. 2015;108:985. doi: 10.1093/qjmed/hcv136
5. Harvey RD, Metcalfe RA, Morteo C, et al. Acute pre-tibial myxoedema following radioiodine therapy for thyrotoxic Graves’ disease. Clin Endocrinol (Oxf). 1995;42:657-660. doi: 10.1111/j.1365-2265.1995.tb02695.x
6. Schukow C, Ahmed A. Dermatopathology, cutaneous lymphomas. StatPearls [Internet]. Updated February 16, 2023. Accessed October 23, 2023. www.ncbi.nlm.nih.gov/books/NBK589703/
A 34-YEAR-OLD MAN presented with a 6-month history of asymptomatic, progressively enlarging subcutaneous nodules over his bilateral lower legs. He denied any history of injury, and there was no bleeding or discharge. The patient had a history of Graves disease that had been treated with radioiodine therapy 2 years prior, followed by thyroxine replacement (150 mcg/d, 5 d/wk and 125 mcg/d, 2 d/wk). At the time of presentation, his thyroid function tests indicated subclinical hypothyroidism: free T4, 21.2 pmol/L (normal range, 11.8-24.6 pmol/L) and thyroid-stimulating hormone (TSH), 14.07 mIU/L (normal range, 0.27-4.2 mIU/L).
Examination revealed nontender, soft brown nodules over the bilateral shins, with minimal overlying lichenification (FIGURE 1). There was no peau d’orange (orange peel) appearance to suggest significant edema. A punch biopsy was performed.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pretibial myxedema
The patient’s history, paired with the results of the punch biopsy, were consistent with a diagnosis of pretibial myxedema, part of the triad of Graves disease along with thyroid ophthalmopathy and acropachy (soft-tissue swelling of the hands and clubbing of the fingers). Histopathologic findings revealed wide separation of collagen bundles throughout the entire reticular dermis without fibroplasia (FIGURE 2A). The spaces contained basophilic strands (FIGURE 2B), and the strands stained strongly positive on Alcian blue (FIGURE 2C), confirming the presence of dermal mucin. Widely separated collagen fibers and deposited mucin are indicative of pretibial myxedema. No granulomas or lymphoid proliferations were seen.

The pathogenesis of pretibial myxedema is widely postulated to be due to the stimulation of dermal fibroblasts by anti–TSH antibodies, causing overproduction of glycosaminoglycans and hyaluronic acid1 and obstructing lymphatic microcirculation, resulting in nonpitting edema.2
There are 5 distinct clinical variants of pretibial myxedema1,3:
- The diffuse form is the most common. It manifests on the lower leg with hard, nonpitting edema and cutaneous thickening.
- The plaque form manifests on the lower leg as well-demarcated erythematous or pigmented flat-topped lesions.
- The nodular form, which our patient had, typically manifests on the lower leg as well-demarcated erythematous, pigmented, or skin-colored raised, solid lesions. There may be 1 lesion or several.
- The mixed form manifests as 2 or more of the other variants.
- The elephantiasic form is the rarest and the most severe. There are widespread swollen nodules and plaques on the lower legs and/or arms.
A rare, late manifestation
Although pathognomonic for Graves disease, pretibial myxedema is a late manifestation that occurs in less than 5% of these patients.4 The most common site of involvement is the pretibial region, although less common sites include the face, arms, shoulders, abdomen, pinna, and the location of previous scars.4
While pretibial myxedema usually is associated with hyperthyroidism, it can occur after treatment (as was the case here), while the patient is in a euthyroid or hypothyroid state. Radioiodine therapy has been reported to be a trigger for pretibial myxedema in 1 case report, although the pathophysiology is not fully understood.5
Continue to: More serious conditions must be ruled out
More serious conditions must be ruled out
The differential for painless nodules includes cutaneous lymphoma and atypical infections of fungal or mycobacterial etiology.
Cutaneous lymphoma that manifests with leg tumors includes primary cutaneous anaplastic CD30+ large cell lymphoma (PCALCL) and primary cutaneous diffuse large B-cell lymphoma, leg type (PCDLBL-LT). The former may occur in young patients, whereas the latter tends to manifest in the elderly. Biopsy shows a neoplastic proliferation of atypical lymphocytes within the dermis,6 differing from our case.
Atypical infections may be detected through bacterial, mycobacterial, or fungal cultures, and may be accompanied by elevated inflammatory markers or other systemic symptoms of the infection, setting it apart from pretibial myxedema.
Treatment is simple and noninvasive
Pretibial myxedema is usually asymptomatic, with minimal morbidity. The nodular variant may resolve spontaneously; thus, therapeutic management often is reserved for severe cases or for those with cosmetic concerns. Treatment options include mid- to high-potency topical corticosteroids with an occlusive dressing for 1 to 2 weeks (or until resolution) or an intralesional triamcinolone injection (5-10 mg/mL, single or monthly until resolution), compression stockings, and pneumatic compression.2
This patient was treated with a single intralesional injection of triamcinolone 10 mg/mL. The nodules resolved within a month.
A 34-YEAR-OLD MAN presented with a 6-month history of asymptomatic, progressively enlarging subcutaneous nodules over his bilateral lower legs. He denied any history of injury, and there was no bleeding or discharge. The patient had a history of Graves disease that had been treated with radioiodine therapy 2 years prior, followed by thyroxine replacement (150 mcg/d, 5 d/wk and 125 mcg/d, 2 d/wk). At the time of presentation, his thyroid function tests indicated subclinical hypothyroidism: free T4, 21.2 pmol/L (normal range, 11.8-24.6 pmol/L) and thyroid-stimulating hormone (TSH), 14.07 mIU/L (normal range, 0.27-4.2 mIU/L).
Examination revealed nontender, soft brown nodules over the bilateral shins, with minimal overlying lichenification (FIGURE 1). There was no peau d’orange (orange peel) appearance to suggest significant edema. A punch biopsy was performed.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pretibial myxedema
The patient’s history, paired with the results of the punch biopsy, were consistent with a diagnosis of pretibial myxedema, part of the triad of Graves disease along with thyroid ophthalmopathy and acropachy (soft-tissue swelling of the hands and clubbing of the fingers). Histopathologic findings revealed wide separation of collagen bundles throughout the entire reticular dermis without fibroplasia (FIGURE 2A). The spaces contained basophilic strands (FIGURE 2B), and the strands stained strongly positive on Alcian blue (FIGURE 2C), confirming the presence of dermal mucin. Widely separated collagen fibers and deposited mucin are indicative of pretibial myxedema. No granulomas or lymphoid proliferations were seen.

The pathogenesis of pretibial myxedema is widely postulated to be due to the stimulation of dermal fibroblasts by anti–TSH antibodies, causing overproduction of glycosaminoglycans and hyaluronic acid1 and obstructing lymphatic microcirculation, resulting in nonpitting edema.2
There are 5 distinct clinical variants of pretibial myxedema1,3:
- The diffuse form is the most common. It manifests on the lower leg with hard, nonpitting edema and cutaneous thickening.
- The plaque form manifests on the lower leg as well-demarcated erythematous or pigmented flat-topped lesions.
- The nodular form, which our patient had, typically manifests on the lower leg as well-demarcated erythematous, pigmented, or skin-colored raised, solid lesions. There may be 1 lesion or several.
- The mixed form manifests as 2 or more of the other variants.
- The elephantiasic form is the rarest and the most severe. There are widespread swollen nodules and plaques on the lower legs and/or arms.
A rare, late manifestation
Although pathognomonic for Graves disease, pretibial myxedema is a late manifestation that occurs in less than 5% of these patients.4 The most common site of involvement is the pretibial region, although less common sites include the face, arms, shoulders, abdomen, pinna, and the location of previous scars.4
While pretibial myxedema usually is associated with hyperthyroidism, it can occur after treatment (as was the case here), while the patient is in a euthyroid or hypothyroid state. Radioiodine therapy has been reported to be a trigger for pretibial myxedema in 1 case report, although the pathophysiology is not fully understood.5
Continue to: More serious conditions must be ruled out
More serious conditions must be ruled out
The differential for painless nodules includes cutaneous lymphoma and atypical infections of fungal or mycobacterial etiology.
Cutaneous lymphoma that manifests with leg tumors includes primary cutaneous anaplastic CD30+ large cell lymphoma (PCALCL) and primary cutaneous diffuse large B-cell lymphoma, leg type (PCDLBL-LT). The former may occur in young patients, whereas the latter tends to manifest in the elderly. Biopsy shows a neoplastic proliferation of atypical lymphocytes within the dermis,6 differing from our case.
Atypical infections may be detected through bacterial, mycobacterial, or fungal cultures, and may be accompanied by elevated inflammatory markers or other systemic symptoms of the infection, setting it apart from pretibial myxedema.
Treatment is simple and noninvasive
Pretibial myxedema is usually asymptomatic, with minimal morbidity. The nodular variant may resolve spontaneously; thus, therapeutic management often is reserved for severe cases or for those with cosmetic concerns. Treatment options include mid- to high-potency topical corticosteroids with an occlusive dressing for 1 to 2 weeks (or until resolution) or an intralesional triamcinolone injection (5-10 mg/mL, single or monthly until resolution), compression stockings, and pneumatic compression.2
This patient was treated with a single intralesional injection of triamcinolone 10 mg/mL. The nodules resolved within a month.
1. Thammarucha S, Sudtikoonaseth P. Nodular pretibial myxedema with Graves’ disease: a case report. Thai J Dermatol. 2021;37:30-36.
2. Singla M, Gupta A. Nodular thyroid dermopathy: not a hallmark of Graves’ disease. Am J Med. 2019;132:e521-e522. doi: 10.1016/j.amjmed.2018.11.004
3. Lan C, Wang Y, Zeng X, et al. Morphological diversity of pretibial myxedema and its mechanism of evolving process and outcome: a retrospective study of 216 cases. J Thyroid Res. 2016:2016:265217
4. doi: 10.1155/2016/2652174 4. Patil MM, Kamalanathan S, Sahoo J, et al. Pretibial myxedema. QJM. 2015;108:985. doi: 10.1093/qjmed/hcv136
5. Harvey RD, Metcalfe RA, Morteo C, et al. Acute pre-tibial myxoedema following radioiodine therapy for thyrotoxic Graves’ disease. Clin Endocrinol (Oxf). 1995;42:657-660. doi: 10.1111/j.1365-2265.1995.tb02695.x
6. Schukow C, Ahmed A. Dermatopathology, cutaneous lymphomas. StatPearls [Internet]. Updated February 16, 2023. Accessed October 23, 2023. www.ncbi.nlm.nih.gov/books/NBK589703/
1. Thammarucha S, Sudtikoonaseth P. Nodular pretibial myxedema with Graves’ disease: a case report. Thai J Dermatol. 2021;37:30-36.
2. Singla M, Gupta A. Nodular thyroid dermopathy: not a hallmark of Graves’ disease. Am J Med. 2019;132:e521-e522. doi: 10.1016/j.amjmed.2018.11.004
3. Lan C, Wang Y, Zeng X, et al. Morphological diversity of pretibial myxedema and its mechanism of evolving process and outcome: a retrospective study of 216 cases. J Thyroid Res. 2016:2016:265217
4. doi: 10.1155/2016/2652174 4. Patil MM, Kamalanathan S, Sahoo J, et al. Pretibial myxedema. QJM. 2015;108:985. doi: 10.1093/qjmed/hcv136
5. Harvey RD, Metcalfe RA, Morteo C, et al. Acute pre-tibial myxoedema following radioiodine therapy for thyrotoxic Graves’ disease. Clin Endocrinol (Oxf). 1995;42:657-660. doi: 10.1111/j.1365-2265.1995.tb02695.x
6. Schukow C, Ahmed A. Dermatopathology, cutaneous lymphomas. StatPearls [Internet]. Updated February 16, 2023. Accessed October 23, 2023. www.ncbi.nlm.nih.gov/books/NBK589703/
AI algorithm aids egg retrieval date during fertility treatment cycles
According to the researchers, such an algorithm is needed due to the increased demand for fertility treatments, as well as the high day-to-day variability in lab workload.
According to the study investigators, predicting retrieval dates in advance for ongoing cycles is of major importance for both patients and clinicians.
“The population requiring fertility treatments, including genetic testing and fertility preservation, has massively increased, and this causes many more cycles and a high day-to-day variability in IVF activity, especially in the lab workload,” said Rohi Hourvitz, MBA, from FertilAI, an Israeli health care company focused on developing technologies that improve fertility treatments.
“We also need to accommodate and reschedule for non-working days, which causes a big issue with managing the workload in many clinics around the world,” added Mr. Hourvitz, who presented the research highlighting AI’s growing role in reproductive medicine.
In addition, AI has recently emerged as an effective tool for assisting in clinical decision-making in assisted reproductive technology, prompting further research in this space, he said.
The new study used a dataset of 9,550 predictable antagonist cycles (defined as having all necessary data) gathered from one lab with over 50 physicians between August 2018 and October 2022. The data were split into two subsets: one for training the AI model and the other for prospective testing.
To train and test the AI model, data from nearly 6,000 predictable antagonist cycles were used. Key factors used for each cycle included estrogen levels, mean follicle size, primary follicle size, and various patient demographics. Other features were considered, but Mr. Hourvitz noted that primary follicle size influenced the algorithm most, “because that is what most of us use when we want to trigger.”
Mr. Hourvitz explained that these patient data were run through an algorithm that produced a graph predicting the most probable date for a cycle retrieval.
“We could accurately predict when those ‘peak days’ were going to be happening in the clinic, and we could also give a pretty good estimate on how many cycles you’re going to have every day,” Mr. Hourvitz said, explaining that this information could help clinics more efficiently allocate resources and manage patients.
According to Mr. Hourvitz, the predictions derived from this study could improve various aspects of fertility treatments and related procedures, including better staff planning and caseload management in IVF labs, as well as higher-quality eggs at retrieval. Patients would have a clearer timeline for their treatment cycles.
Nikica Zaninovic, PhD, MS, director of the embryology lab at Weill Cornell Medical College, New York City, cautioned that the new findings are not yet ready for clinical application but emphasized the importance of more AI research focusing on the quality of oocytes, not only embryos.
“We’re so focused on the end of the process: the embryo,” Dr. Zaninovic, who was not involved in the research, said in an interview. “I think the focus should be on the beginning – the quality of eggs and sperm, not just the quantity – because that’s what the embryos will depend on.”
He noted the increasing numbers of young women in the United States undergoing egg freezing.
“Cornell is the largest academic IVF center in the United States; 20%-30% of all of the patients that we treat are actually freezing their eggs,” he said. “It’s a huge population.”
“When they come to us, they ask how many eggs they’ll need to guarantee one or two children in the future,” Dr. Zaninovic continued. “We don’t have that answer, so we always tell them [we’ll retrieve] as many as we can. That’s not the answer; we need to be more precise. We’re still lacking these tools, and I think that’s where the research will go.”
The study was funded by FertilAI. Mr. Hourvitz is a shareholder and CEO of FertilAI. Dr. Zaninovic is president of the AI Fertility Society.
A version of this article appeared on Medscape.com.
According to the researchers, such an algorithm is needed due to the increased demand for fertility treatments, as well as the high day-to-day variability in lab workload.
According to the study investigators, predicting retrieval dates in advance for ongoing cycles is of major importance for both patients and clinicians.
“The population requiring fertility treatments, including genetic testing and fertility preservation, has massively increased, and this causes many more cycles and a high day-to-day variability in IVF activity, especially in the lab workload,” said Rohi Hourvitz, MBA, from FertilAI, an Israeli health care company focused on developing technologies that improve fertility treatments.
“We also need to accommodate and reschedule for non-working days, which causes a big issue with managing the workload in many clinics around the world,” added Mr. Hourvitz, who presented the research highlighting AI’s growing role in reproductive medicine.
In addition, AI has recently emerged as an effective tool for assisting in clinical decision-making in assisted reproductive technology, prompting further research in this space, he said.
The new study used a dataset of 9,550 predictable antagonist cycles (defined as having all necessary data) gathered from one lab with over 50 physicians between August 2018 and October 2022. The data were split into two subsets: one for training the AI model and the other for prospective testing.
To train and test the AI model, data from nearly 6,000 predictable antagonist cycles were used. Key factors used for each cycle included estrogen levels, mean follicle size, primary follicle size, and various patient demographics. Other features were considered, but Mr. Hourvitz noted that primary follicle size influenced the algorithm most, “because that is what most of us use when we want to trigger.”
Mr. Hourvitz explained that these patient data were run through an algorithm that produced a graph predicting the most probable date for a cycle retrieval.
“We could accurately predict when those ‘peak days’ were going to be happening in the clinic, and we could also give a pretty good estimate on how many cycles you’re going to have every day,” Mr. Hourvitz said, explaining that this information could help clinics more efficiently allocate resources and manage patients.
According to Mr. Hourvitz, the predictions derived from this study could improve various aspects of fertility treatments and related procedures, including better staff planning and caseload management in IVF labs, as well as higher-quality eggs at retrieval. Patients would have a clearer timeline for their treatment cycles.
Nikica Zaninovic, PhD, MS, director of the embryology lab at Weill Cornell Medical College, New York City, cautioned that the new findings are not yet ready for clinical application but emphasized the importance of more AI research focusing on the quality of oocytes, not only embryos.
“We’re so focused on the end of the process: the embryo,” Dr. Zaninovic, who was not involved in the research, said in an interview. “I think the focus should be on the beginning – the quality of eggs and sperm, not just the quantity – because that’s what the embryos will depend on.”
He noted the increasing numbers of young women in the United States undergoing egg freezing.
“Cornell is the largest academic IVF center in the United States; 20%-30% of all of the patients that we treat are actually freezing their eggs,” he said. “It’s a huge population.”
“When they come to us, they ask how many eggs they’ll need to guarantee one or two children in the future,” Dr. Zaninovic continued. “We don’t have that answer, so we always tell them [we’ll retrieve] as many as we can. That’s not the answer; we need to be more precise. We’re still lacking these tools, and I think that’s where the research will go.”
The study was funded by FertilAI. Mr. Hourvitz is a shareholder and CEO of FertilAI. Dr. Zaninovic is president of the AI Fertility Society.
A version of this article appeared on Medscape.com.
According to the researchers, such an algorithm is needed due to the increased demand for fertility treatments, as well as the high day-to-day variability in lab workload.
According to the study investigators, predicting retrieval dates in advance for ongoing cycles is of major importance for both patients and clinicians.
“The population requiring fertility treatments, including genetic testing and fertility preservation, has massively increased, and this causes many more cycles and a high day-to-day variability in IVF activity, especially in the lab workload,” said Rohi Hourvitz, MBA, from FertilAI, an Israeli health care company focused on developing technologies that improve fertility treatments.
“We also need to accommodate and reschedule for non-working days, which causes a big issue with managing the workload in many clinics around the world,” added Mr. Hourvitz, who presented the research highlighting AI’s growing role in reproductive medicine.
In addition, AI has recently emerged as an effective tool for assisting in clinical decision-making in assisted reproductive technology, prompting further research in this space, he said.
The new study used a dataset of 9,550 predictable antagonist cycles (defined as having all necessary data) gathered from one lab with over 50 physicians between August 2018 and October 2022. The data were split into two subsets: one for training the AI model and the other for prospective testing.
To train and test the AI model, data from nearly 6,000 predictable antagonist cycles were used. Key factors used for each cycle included estrogen levels, mean follicle size, primary follicle size, and various patient demographics. Other features were considered, but Mr. Hourvitz noted that primary follicle size influenced the algorithm most, “because that is what most of us use when we want to trigger.”
Mr. Hourvitz explained that these patient data were run through an algorithm that produced a graph predicting the most probable date for a cycle retrieval.
“We could accurately predict when those ‘peak days’ were going to be happening in the clinic, and we could also give a pretty good estimate on how many cycles you’re going to have every day,” Mr. Hourvitz said, explaining that this information could help clinics more efficiently allocate resources and manage patients.
According to Mr. Hourvitz, the predictions derived from this study could improve various aspects of fertility treatments and related procedures, including better staff planning and caseload management in IVF labs, as well as higher-quality eggs at retrieval. Patients would have a clearer timeline for their treatment cycles.
Nikica Zaninovic, PhD, MS, director of the embryology lab at Weill Cornell Medical College, New York City, cautioned that the new findings are not yet ready for clinical application but emphasized the importance of more AI research focusing on the quality of oocytes, not only embryos.
“We’re so focused on the end of the process: the embryo,” Dr. Zaninovic, who was not involved in the research, said in an interview. “I think the focus should be on the beginning – the quality of eggs and sperm, not just the quantity – because that’s what the embryos will depend on.”
He noted the increasing numbers of young women in the United States undergoing egg freezing.
“Cornell is the largest academic IVF center in the United States; 20%-30% of all of the patients that we treat are actually freezing their eggs,” he said. “It’s a huge population.”
“When they come to us, they ask how many eggs they’ll need to guarantee one or two children in the future,” Dr. Zaninovic continued. “We don’t have that answer, so we always tell them [we’ll retrieve] as many as we can. That’s not the answer; we need to be more precise. We’re still lacking these tools, and I think that’s where the research will go.”
The study was funded by FertilAI. Mr. Hourvitz is a shareholder and CEO of FertilAI. Dr. Zaninovic is president of the AI Fertility Society.
A version of this article appeared on Medscape.com.
FROM ASRM 2023
Pervasive ‘forever chemicals’ linked to thyroid cancer?
The study suggests that higher exposure to per- and polyfluoroalkyl substances (PFAS), specifically perfluorooctanesulfonic acid (n-PFOS), may increase a person’s risk for thyroid cancer by 56%.
Several news outlets played up the findings, published online in eBioMedicine. “Dangerous ‘Forever Chemicals’ in Your Everyday Items Are Causing Cancer,” Newsweek reported.
But Gideon Meyerowitz-Katz, PhD, an epidemiologist at the University of Wollongong (Australia), voiced his skepticism.
“While it’s possible that PFAS might be causing thyroid cancer, the evidence thus far is unconvincing and probably not worth worrying about,” said Dr. Meyerowitz-Katz, who was not involved in the research.
PFAS and thyroid cancer
PFAS are a class of widely used synthetic chemicals found in many consumer and industrial products, including nonstick cookware, stain-repellent carpets, waterproof rain gear, microwave popcorn bags, and firefighting foam.
These substances have been dubbed “forever chemicals” because they do not degrade and are ubiquitous in the environment.
Exposure to endocrine-disrupting chemicals, including PFAS, has been identified as a potential risk factor for thyroid cancer, with some research linking PFAS exposure to thyroid dysfunction and carcinogenesis.
To investigate further, the researchers performed a nested case-control study of 86 patients with thyroid cancer using plasma samples collected at or before diagnosis and 86 controls without cancer who were matched on age, sex, race/ethnicity, body weight, smoking status, and year of sample collection.
Eighteen individual PFAS were measured in plasma samples; 10 were undetectable and were therefore excluded from the analysis. Of the remaining eight PFAS, only one showed a statistically significant correlation with thyroid cancer.
Specifically, the researchers found that exposure to n-PFOS was associated with a 56% increased risk for thyroid cancer among people who had a high level of the chemical in their blood (adjusted odds ratio, 1.56; P = .004). The results were similar when patients with papillary thyroid cancer only were included (aOR, 1.56; P = .009).
A separate longitudinal analysis of 31 patients diagnosed with thyroid cancer 1 year or more after plasma sample collection and 31 controls confirmed the positive association between n-PFOS and thyroid cancer (aOR, 2.67; P < .001). The longitudinal analysis also suggested correlations for a few other PFAS.
“This study supports the hypothesis that PFAS exposure may be associated with increased risk of thyroid cancer,” the authors concluded.
But in a Substack post, Dr. Meyerowitz-Katz said that it’s important to put the findings into “proper context before getting terrified about this all-new cancer risk.”
First, this study was “genuinely tiny,” with data on just 88 people with thyroid cancer and 88 controls, a limitation the researchers also acknowledged.
“That’s really not enough to do any sort of robust epidemiological analysis – you can generate interesting correlations, but what those correlations mean is anyone’s guess,” Dr. Meyerowitz-Katz said.
Even more importantly, one could easily argue that the results of this study show that most PFAS aren’t associated with thyroid cancer, given that there was no strong association for seven of the eight PFAS measured, he explained.
“There are no serious methodological concerns here, but equally there’s just not much you can reasonably gather from finding a single correlation among a vast ocean of possibilities,” Dr. Meyerowitz-Katz wrote. “Maybe there’s a correlation there, but you’d need to investigate this in much bigger samples, with more controls, and better data, to understand what that correlation means.”
Bottom line, Dr. Meyerowitz-Katz explained, is that “the link between PFAS and thyroid cancer is, at best, incredibly weak.”
Funding for the study was provided by the National Institutes of Health and The Andrea and Charles Bronfman Philanthropies. One coauthor is cofounder of Linus Biotechnology and is owner of a license agreement with NIES (Japan); received honoraria and travel compensation for lectures for the Bio-Echo and Brin foundations; and has 22 patents at various stages. Dr. Meyerowitz-Katz has no relevant disclosures.
A version of this article appeared on Medscape.com.
The study suggests that higher exposure to per- and polyfluoroalkyl substances (PFAS), specifically perfluorooctanesulfonic acid (n-PFOS), may increase a person’s risk for thyroid cancer by 56%.
Several news outlets played up the findings, published online in eBioMedicine. “Dangerous ‘Forever Chemicals’ in Your Everyday Items Are Causing Cancer,” Newsweek reported.
But Gideon Meyerowitz-Katz, PhD, an epidemiologist at the University of Wollongong (Australia), voiced his skepticism.
“While it’s possible that PFAS might be causing thyroid cancer, the evidence thus far is unconvincing and probably not worth worrying about,” said Dr. Meyerowitz-Katz, who was not involved in the research.
PFAS and thyroid cancer
PFAS are a class of widely used synthetic chemicals found in many consumer and industrial products, including nonstick cookware, stain-repellent carpets, waterproof rain gear, microwave popcorn bags, and firefighting foam.
These substances have been dubbed “forever chemicals” because they do not degrade and are ubiquitous in the environment.
Exposure to endocrine-disrupting chemicals, including PFAS, has been identified as a potential risk factor for thyroid cancer, with some research linking PFAS exposure to thyroid dysfunction and carcinogenesis.
To investigate further, the researchers performed a nested case-control study of 86 patients with thyroid cancer using plasma samples collected at or before diagnosis and 86 controls without cancer who were matched on age, sex, race/ethnicity, body weight, smoking status, and year of sample collection.
Eighteen individual PFAS were measured in plasma samples; 10 were undetectable and were therefore excluded from the analysis. Of the remaining eight PFAS, only one showed a statistically significant correlation with thyroid cancer.
Specifically, the researchers found that exposure to n-PFOS was associated with a 56% increased risk for thyroid cancer among people who had a high level of the chemical in their blood (adjusted odds ratio, 1.56; P = .004). The results were similar when patients with papillary thyroid cancer only were included (aOR, 1.56; P = .009).
A separate longitudinal analysis of 31 patients diagnosed with thyroid cancer 1 year or more after plasma sample collection and 31 controls confirmed the positive association between n-PFOS and thyroid cancer (aOR, 2.67; P < .001). The longitudinal analysis also suggested correlations for a few other PFAS.
“This study supports the hypothesis that PFAS exposure may be associated with increased risk of thyroid cancer,” the authors concluded.
But in a Substack post, Dr. Meyerowitz-Katz said that it’s important to put the findings into “proper context before getting terrified about this all-new cancer risk.”
First, this study was “genuinely tiny,” with data on just 88 people with thyroid cancer and 88 controls, a limitation the researchers also acknowledged.
“That’s really not enough to do any sort of robust epidemiological analysis – you can generate interesting correlations, but what those correlations mean is anyone’s guess,” Dr. Meyerowitz-Katz said.
Even more importantly, one could easily argue that the results of this study show that most PFAS aren’t associated with thyroid cancer, given that there was no strong association for seven of the eight PFAS measured, he explained.
“There are no serious methodological concerns here, but equally there’s just not much you can reasonably gather from finding a single correlation among a vast ocean of possibilities,” Dr. Meyerowitz-Katz wrote. “Maybe there’s a correlation there, but you’d need to investigate this in much bigger samples, with more controls, and better data, to understand what that correlation means.”
Bottom line, Dr. Meyerowitz-Katz explained, is that “the link between PFAS and thyroid cancer is, at best, incredibly weak.”
Funding for the study was provided by the National Institutes of Health and The Andrea and Charles Bronfman Philanthropies. One coauthor is cofounder of Linus Biotechnology and is owner of a license agreement with NIES (Japan); received honoraria and travel compensation for lectures for the Bio-Echo and Brin foundations; and has 22 patents at various stages. Dr. Meyerowitz-Katz has no relevant disclosures.
A version of this article appeared on Medscape.com.
The study suggests that higher exposure to per- and polyfluoroalkyl substances (PFAS), specifically perfluorooctanesulfonic acid (n-PFOS), may increase a person’s risk for thyroid cancer by 56%.
Several news outlets played up the findings, published online in eBioMedicine. “Dangerous ‘Forever Chemicals’ in Your Everyday Items Are Causing Cancer,” Newsweek reported.
But Gideon Meyerowitz-Katz, PhD, an epidemiologist at the University of Wollongong (Australia), voiced his skepticism.
“While it’s possible that PFAS might be causing thyroid cancer, the evidence thus far is unconvincing and probably not worth worrying about,” said Dr. Meyerowitz-Katz, who was not involved in the research.
PFAS and thyroid cancer
PFAS are a class of widely used synthetic chemicals found in many consumer and industrial products, including nonstick cookware, stain-repellent carpets, waterproof rain gear, microwave popcorn bags, and firefighting foam.
These substances have been dubbed “forever chemicals” because they do not degrade and are ubiquitous in the environment.
Exposure to endocrine-disrupting chemicals, including PFAS, has been identified as a potential risk factor for thyroid cancer, with some research linking PFAS exposure to thyroid dysfunction and carcinogenesis.
To investigate further, the researchers performed a nested case-control study of 86 patients with thyroid cancer using plasma samples collected at or before diagnosis and 86 controls without cancer who were matched on age, sex, race/ethnicity, body weight, smoking status, and year of sample collection.
Eighteen individual PFAS were measured in plasma samples; 10 were undetectable and were therefore excluded from the analysis. Of the remaining eight PFAS, only one showed a statistically significant correlation with thyroid cancer.
Specifically, the researchers found that exposure to n-PFOS was associated with a 56% increased risk for thyroid cancer among people who had a high level of the chemical in their blood (adjusted odds ratio, 1.56; P = .004). The results were similar when patients with papillary thyroid cancer only were included (aOR, 1.56; P = .009).
A separate longitudinal analysis of 31 patients diagnosed with thyroid cancer 1 year or more after plasma sample collection and 31 controls confirmed the positive association between n-PFOS and thyroid cancer (aOR, 2.67; P < .001). The longitudinal analysis also suggested correlations for a few other PFAS.
“This study supports the hypothesis that PFAS exposure may be associated with increased risk of thyroid cancer,” the authors concluded.
But in a Substack post, Dr. Meyerowitz-Katz said that it’s important to put the findings into “proper context before getting terrified about this all-new cancer risk.”
First, this study was “genuinely tiny,” with data on just 88 people with thyroid cancer and 88 controls, a limitation the researchers also acknowledged.
“That’s really not enough to do any sort of robust epidemiological analysis – you can generate interesting correlations, but what those correlations mean is anyone’s guess,” Dr. Meyerowitz-Katz said.
Even more importantly, one could easily argue that the results of this study show that most PFAS aren’t associated with thyroid cancer, given that there was no strong association for seven of the eight PFAS measured, he explained.
“There are no serious methodological concerns here, but equally there’s just not much you can reasonably gather from finding a single correlation among a vast ocean of possibilities,” Dr. Meyerowitz-Katz wrote. “Maybe there’s a correlation there, but you’d need to investigate this in much bigger samples, with more controls, and better data, to understand what that correlation means.”
Bottom line, Dr. Meyerowitz-Katz explained, is that “the link between PFAS and thyroid cancer is, at best, incredibly weak.”
Funding for the study was provided by the National Institutes of Health and The Andrea and Charles Bronfman Philanthropies. One coauthor is cofounder of Linus Biotechnology and is owner of a license agreement with NIES (Japan); received honoraria and travel compensation for lectures for the Bio-Echo and Brin foundations; and has 22 patents at various stages. Dr. Meyerowitz-Katz has no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM EBIOMEDICINE
Be advised: Thyroid hormones may increase risk of cognitive disorders in older adults
published in JAMA Internal Medicine.
The study found that these patients with thyrotoxicosis had a higher likelihood of incident cognitive disorder (adjusted hazard ratio, 1.39; 95% confidence interval, 1.18-1.64; P < .001). Broken down between internal and external causes of thyrotoxicosis, exogenous thyrotoxicosis continued to be a significant risk factor (aHR, 1.34: 95% CI, 1.10-1.63; P = .003), while endogenous thyrotoxicosis did not show a statistically significant risk estimates (aHR, 1.38; 95% CI, 0.96-1.98; P = .08).
The study also found that women were more likely to have low levels of thyrotropin (thyroid-stimulating hormone/TSH) than men and were more likely to be overtreated.
Previous studies looking at the correlation between hyperthyroidism and cognitive disorders often did not include participants who were already taking thyroid hormones, according to Jennifer S. Mammen, MD, PhD, assistant professor of medicine at the Asthma and Allergy Center at John Hopkins University, Baltimore, and the senior author of the study.
“The fact that we see the signal both in people who are being overtreated with thyroid hormone and in people who have endogenous hyperthyroidism is one way that we think that this supports the fact that it’s not just confounding, it’s not just bias,” Dr. Mammen said. “There’s two different sources of hyperthyroidism, and they’re both showing the same relationship.”
In the study, Dr. Mammen and colleagues analyzed electronic health records for patients aged 65 years and older who received primary care in the Johns Hopkins Community Physicians Network over a 10-year period starting in 2014. Patients had to have a minimum of two visits 30 days apart. None had a history of low TSH levels or cognitive disorder diagnoses within 6 months of their first doctor visit.
More than 65,000 patients were included in the study. Slightly more than half (56%) were female, almost 70% were White, 19.3% were Black, 4.6% were Asian, and 0.4% were American Indian. Almost 25,000 low TSH measurements among 2,710 patients were recorded during the study period. The majority of low TSH measurements were exogenous (14,875), followed by origins of unknown cause (5,833), and endogenous (4,159).
During the follow-up period, 7.2% (4,779) patients received a new cognitive disorder diagnosis, which was dementia in 77% of cases.
Dr. Mammen said primary care physicians should carefully consider whether thyroid hormone therapy is necessary for older patients, and, if so, great care should be taken to avoid overtreatment.
“This is yet another reason for us to be vigilant about not overtreating people with thyroid hormone, especially in older adults,” Dr. Mammen said. “We already know that atrial fibrillation rates are increased in people who are hyperthyroid. We know that fracture and osteoporosis is affected by hyperthyroidism. And now we also have an association with higher rates of cognitive disorders.”
Taking a cautious approach to prescribing thyroid hormone therapy for older patients is paramount, according to Jean Chen, MD, partner at Texas Diabetes & Endocrinology, who was not affiliated with the study.
“All medical providers need to be aware that the 65 and older population does not need to be treated as aggressively with their thyroid hormone,” Dr. Chen said. “We are finding more and more complications from overtreatment rather than benefit in this population.”
Often, older patients may complain of symptoms such as constipation, feeling cold, or tiredness, which can be symptoms of hypothyroidism. But these symptoms could also be from anemia, vitamin deficiencies, depression, perimenopause, menopause, insulin resistance, and sleep apnea. If necessary, Dr. Chen recommended primary care physicians consult with an endocrinologist regarding a possible treatment plan and making a differential diagnosis.
In addition, Dr. Chen said other studies have shown that treating patients with thyroid hormone either did not resolve the condition or negatively impacted anxiety, muscle strength, and bone density, or it increased the risk for arrhythmia. Therefore, it’s important to weight the risks versus the benefits.
“There’s so much gray zone here,” Dr. Chen said.
The study was supported by the Richman Family Precision Medicine Center of Excellence in Alzheimer’s Disease, the Richman Family Foundation, the Rick Sharp Alzheimer’s Foundation, the Sharp Family Foundation, among others. The work was also supported by grants from the National Institutes of Health. One coauthor reported personal fees from Karuna, MapLight Therapeutics, Axsome Therapeutics, GIA, GW Research Limited, Merck, EXCIVA, Otsuka, IntraCellular Therapies, and Medesis Pharma for consulting for treatment development in Alzheimer’s disease outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
published in JAMA Internal Medicine.
The study found that these patients with thyrotoxicosis had a higher likelihood of incident cognitive disorder (adjusted hazard ratio, 1.39; 95% confidence interval, 1.18-1.64; P < .001). Broken down between internal and external causes of thyrotoxicosis, exogenous thyrotoxicosis continued to be a significant risk factor (aHR, 1.34: 95% CI, 1.10-1.63; P = .003), while endogenous thyrotoxicosis did not show a statistically significant risk estimates (aHR, 1.38; 95% CI, 0.96-1.98; P = .08).
The study also found that women were more likely to have low levels of thyrotropin (thyroid-stimulating hormone/TSH) than men and were more likely to be overtreated.
Previous studies looking at the correlation between hyperthyroidism and cognitive disorders often did not include participants who were already taking thyroid hormones, according to Jennifer S. Mammen, MD, PhD, assistant professor of medicine at the Asthma and Allergy Center at John Hopkins University, Baltimore, and the senior author of the study.
“The fact that we see the signal both in people who are being overtreated with thyroid hormone and in people who have endogenous hyperthyroidism is one way that we think that this supports the fact that it’s not just confounding, it’s not just bias,” Dr. Mammen said. “There’s two different sources of hyperthyroidism, and they’re both showing the same relationship.”
In the study, Dr. Mammen and colleagues analyzed electronic health records for patients aged 65 years and older who received primary care in the Johns Hopkins Community Physicians Network over a 10-year period starting in 2014. Patients had to have a minimum of two visits 30 days apart. None had a history of low TSH levels or cognitive disorder diagnoses within 6 months of their first doctor visit.
More than 65,000 patients were included in the study. Slightly more than half (56%) were female, almost 70% were White, 19.3% were Black, 4.6% were Asian, and 0.4% were American Indian. Almost 25,000 low TSH measurements among 2,710 patients were recorded during the study period. The majority of low TSH measurements were exogenous (14,875), followed by origins of unknown cause (5,833), and endogenous (4,159).
During the follow-up period, 7.2% (4,779) patients received a new cognitive disorder diagnosis, which was dementia in 77% of cases.
Dr. Mammen said primary care physicians should carefully consider whether thyroid hormone therapy is necessary for older patients, and, if so, great care should be taken to avoid overtreatment.
“This is yet another reason for us to be vigilant about not overtreating people with thyroid hormone, especially in older adults,” Dr. Mammen said. “We already know that atrial fibrillation rates are increased in people who are hyperthyroid. We know that fracture and osteoporosis is affected by hyperthyroidism. And now we also have an association with higher rates of cognitive disorders.”
Taking a cautious approach to prescribing thyroid hormone therapy for older patients is paramount, according to Jean Chen, MD, partner at Texas Diabetes & Endocrinology, who was not affiliated with the study.
“All medical providers need to be aware that the 65 and older population does not need to be treated as aggressively with their thyroid hormone,” Dr. Chen said. “We are finding more and more complications from overtreatment rather than benefit in this population.”
Often, older patients may complain of symptoms such as constipation, feeling cold, or tiredness, which can be symptoms of hypothyroidism. But these symptoms could also be from anemia, vitamin deficiencies, depression, perimenopause, menopause, insulin resistance, and sleep apnea. If necessary, Dr. Chen recommended primary care physicians consult with an endocrinologist regarding a possible treatment plan and making a differential diagnosis.
In addition, Dr. Chen said other studies have shown that treating patients with thyroid hormone either did not resolve the condition or negatively impacted anxiety, muscle strength, and bone density, or it increased the risk for arrhythmia. Therefore, it’s important to weight the risks versus the benefits.
“There’s so much gray zone here,” Dr. Chen said.
The study was supported by the Richman Family Precision Medicine Center of Excellence in Alzheimer’s Disease, the Richman Family Foundation, the Rick Sharp Alzheimer’s Foundation, the Sharp Family Foundation, among others. The work was also supported by grants from the National Institutes of Health. One coauthor reported personal fees from Karuna, MapLight Therapeutics, Axsome Therapeutics, GIA, GW Research Limited, Merck, EXCIVA, Otsuka, IntraCellular Therapies, and Medesis Pharma for consulting for treatment development in Alzheimer’s disease outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
published in JAMA Internal Medicine.
The study found that these patients with thyrotoxicosis had a higher likelihood of incident cognitive disorder (adjusted hazard ratio, 1.39; 95% confidence interval, 1.18-1.64; P < .001). Broken down between internal and external causes of thyrotoxicosis, exogenous thyrotoxicosis continued to be a significant risk factor (aHR, 1.34: 95% CI, 1.10-1.63; P = .003), while endogenous thyrotoxicosis did not show a statistically significant risk estimates (aHR, 1.38; 95% CI, 0.96-1.98; P = .08).
The study also found that women were more likely to have low levels of thyrotropin (thyroid-stimulating hormone/TSH) than men and were more likely to be overtreated.
Previous studies looking at the correlation between hyperthyroidism and cognitive disorders often did not include participants who were already taking thyroid hormones, according to Jennifer S. Mammen, MD, PhD, assistant professor of medicine at the Asthma and Allergy Center at John Hopkins University, Baltimore, and the senior author of the study.
“The fact that we see the signal both in people who are being overtreated with thyroid hormone and in people who have endogenous hyperthyroidism is one way that we think that this supports the fact that it’s not just confounding, it’s not just bias,” Dr. Mammen said. “There’s two different sources of hyperthyroidism, and they’re both showing the same relationship.”
In the study, Dr. Mammen and colleagues analyzed electronic health records for patients aged 65 years and older who received primary care in the Johns Hopkins Community Physicians Network over a 10-year period starting in 2014. Patients had to have a minimum of two visits 30 days apart. None had a history of low TSH levels or cognitive disorder diagnoses within 6 months of their first doctor visit.
More than 65,000 patients were included in the study. Slightly more than half (56%) were female, almost 70% were White, 19.3% were Black, 4.6% were Asian, and 0.4% were American Indian. Almost 25,000 low TSH measurements among 2,710 patients were recorded during the study period. The majority of low TSH measurements were exogenous (14,875), followed by origins of unknown cause (5,833), and endogenous (4,159).
During the follow-up period, 7.2% (4,779) patients received a new cognitive disorder diagnosis, which was dementia in 77% of cases.
Dr. Mammen said primary care physicians should carefully consider whether thyroid hormone therapy is necessary for older patients, and, if so, great care should be taken to avoid overtreatment.
“This is yet another reason for us to be vigilant about not overtreating people with thyroid hormone, especially in older adults,” Dr. Mammen said. “We already know that atrial fibrillation rates are increased in people who are hyperthyroid. We know that fracture and osteoporosis is affected by hyperthyroidism. And now we also have an association with higher rates of cognitive disorders.”
Taking a cautious approach to prescribing thyroid hormone therapy for older patients is paramount, according to Jean Chen, MD, partner at Texas Diabetes & Endocrinology, who was not affiliated with the study.
“All medical providers need to be aware that the 65 and older population does not need to be treated as aggressively with their thyroid hormone,” Dr. Chen said. “We are finding more and more complications from overtreatment rather than benefit in this population.”
Often, older patients may complain of symptoms such as constipation, feeling cold, or tiredness, which can be symptoms of hypothyroidism. But these symptoms could also be from anemia, vitamin deficiencies, depression, perimenopause, menopause, insulin resistance, and sleep apnea. If necessary, Dr. Chen recommended primary care physicians consult with an endocrinologist regarding a possible treatment plan and making a differential diagnosis.
In addition, Dr. Chen said other studies have shown that treating patients with thyroid hormone either did not resolve the condition or negatively impacted anxiety, muscle strength, and bone density, or it increased the risk for arrhythmia. Therefore, it’s important to weight the risks versus the benefits.
“There’s so much gray zone here,” Dr. Chen said.
The study was supported by the Richman Family Precision Medicine Center of Excellence in Alzheimer’s Disease, the Richman Family Foundation, the Rick Sharp Alzheimer’s Foundation, the Sharp Family Foundation, among others. The work was also supported by grants from the National Institutes of Health. One coauthor reported personal fees from Karuna, MapLight Therapeutics, Axsome Therapeutics, GIA, GW Research Limited, Merck, EXCIVA, Otsuka, IntraCellular Therapies, and Medesis Pharma for consulting for treatment development in Alzheimer’s disease outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
FROM JAMA INTERNAL MEDICINE
No more hot flashes? AI device could stop menopause symptom
Vasomotor symptoms the sudden rises in body temperature that affect about 75% of menopausal women, have drawn interest after the approval of a new oral drug and research linking hot flashes to Alzheimer’s, heart disease, and stroke.
Now entering the discussion are researchers from the University of Massachusetts, Amherst, and Embr Labs (a Massachusetts Institute of Technology spinoff) who say they’ve developed a machine-learning algorithm that can predict a hot flash.
The device, which sells for $299, is already touted as a way to manage menopausal hot flashes.
But once the algorithm is added, the device will be able to “continuously monitor physiological signals – skin temperature, body temperature, sweating, activity level, or heart rate – and identify early indicators that a hot flash is building,” said Michael Busa, PhD, director of the Center for Human Health and Performance at UMass Amherst, who led the team that developed the algorithm.
That data would be sent to a computing platform in the cloud, where the algorithm can flag signs of an impending hot flash, Dr. Busa said. The device would automatically prompt cooling in less than a second, which could effectively stop the hot flash in its tracks or at least help to take the edge off.
Exploring cooling therapy for hot flashes
“There is always tremendous interest in anything that is nonhormonal and effective in treatment of hot flashes,” said Karen Adams, MD, an ob.gyn. and director of the menopause and healthy aging program at Stanford (Calif.) University. (Dr. Adams was not involved in developing this technology.)
Hormone therapy is the primary treatment, easing hot flashes in 3-4 weeks, Dr. Adams said. “But some women do not want to take estrogen, or should not due to medical contraindications.”
Hormone therapy is generally not recommended for people with a history of breast cancer, blood clots, or diseases of their heart or blood vessels. Recent research presented at the annual meeting of the Menopause Society found that hormone therapy may not work as well in women with obesity.
For nonhormonal treatments, the Food and Drug Administration cleared the oral med fezolinetant (Veozah) in May. Antidepressant medications can also be used as a first-line treatment in those who can’t take estrogen. Another oral drug, elinzanetant, is in late-stage clinical trials.
But there has been little clinical investigation – only two small studies, Dr. Adams said – examining cooling therapy as a treatment for hot flashes. That’s something the makers of this device hope to change.
“Despite the fact that seeking cooling relief is a woman’s immediate natural response to the onset of a hot flash, there is limited work done to understand the benefits of this natural therapy,” said Matthew Smith, PhD, chief technology officer at Embr Labs. “This is in part because the technology didn’t exist to deliver cooling in an immediate, reproducible manner.”
The algorithm’s performance has been benchmarked using data from women having hot flashes, Dr. Smith said. Results have been submitted for publication.
The Embr Wave has been shown to help menopausal women with hot flashes sleep better. It has also been tested as a therapy for hot flashes related to cancer treatment.
But to truly evaluate the device as a treatment for hot flashes, it should be tested in randomized trials including a “sham treatment arm” – where some people get the real treatment while others get the sham treatment, Dr. Adams said.
“Device studies tend to have high placebo response rates that can only be truly evaluated when there is a sham treatment in the study,” she said. “If such a device were shown to be safe and effective, we would absolutely recommend it. But we’re a long way from that.”
A version of this article appeared on WebMD.com.
Vasomotor symptoms the sudden rises in body temperature that affect about 75% of menopausal women, have drawn interest after the approval of a new oral drug and research linking hot flashes to Alzheimer’s, heart disease, and stroke.
Now entering the discussion are researchers from the University of Massachusetts, Amherst, and Embr Labs (a Massachusetts Institute of Technology spinoff) who say they’ve developed a machine-learning algorithm that can predict a hot flash.
The device, which sells for $299, is already touted as a way to manage menopausal hot flashes.
But once the algorithm is added, the device will be able to “continuously monitor physiological signals – skin temperature, body temperature, sweating, activity level, or heart rate – and identify early indicators that a hot flash is building,” said Michael Busa, PhD, director of the Center for Human Health and Performance at UMass Amherst, who led the team that developed the algorithm.
That data would be sent to a computing platform in the cloud, where the algorithm can flag signs of an impending hot flash, Dr. Busa said. The device would automatically prompt cooling in less than a second, which could effectively stop the hot flash in its tracks or at least help to take the edge off.
Exploring cooling therapy for hot flashes
“There is always tremendous interest in anything that is nonhormonal and effective in treatment of hot flashes,” said Karen Adams, MD, an ob.gyn. and director of the menopause and healthy aging program at Stanford (Calif.) University. (Dr. Adams was not involved in developing this technology.)
Hormone therapy is the primary treatment, easing hot flashes in 3-4 weeks, Dr. Adams said. “But some women do not want to take estrogen, or should not due to medical contraindications.”
Hormone therapy is generally not recommended for people with a history of breast cancer, blood clots, or diseases of their heart or blood vessels. Recent research presented at the annual meeting of the Menopause Society found that hormone therapy may not work as well in women with obesity.
For nonhormonal treatments, the Food and Drug Administration cleared the oral med fezolinetant (Veozah) in May. Antidepressant medications can also be used as a first-line treatment in those who can’t take estrogen. Another oral drug, elinzanetant, is in late-stage clinical trials.
But there has been little clinical investigation – only two small studies, Dr. Adams said – examining cooling therapy as a treatment for hot flashes. That’s something the makers of this device hope to change.
“Despite the fact that seeking cooling relief is a woman’s immediate natural response to the onset of a hot flash, there is limited work done to understand the benefits of this natural therapy,” said Matthew Smith, PhD, chief technology officer at Embr Labs. “This is in part because the technology didn’t exist to deliver cooling in an immediate, reproducible manner.”
The algorithm’s performance has been benchmarked using data from women having hot flashes, Dr. Smith said. Results have been submitted for publication.
The Embr Wave has been shown to help menopausal women with hot flashes sleep better. It has also been tested as a therapy for hot flashes related to cancer treatment.
But to truly evaluate the device as a treatment for hot flashes, it should be tested in randomized trials including a “sham treatment arm” – where some people get the real treatment while others get the sham treatment, Dr. Adams said.
“Device studies tend to have high placebo response rates that can only be truly evaluated when there is a sham treatment in the study,” she said. “If such a device were shown to be safe and effective, we would absolutely recommend it. But we’re a long way from that.”
A version of this article appeared on WebMD.com.
Vasomotor symptoms the sudden rises in body temperature that affect about 75% of menopausal women, have drawn interest after the approval of a new oral drug and research linking hot flashes to Alzheimer’s, heart disease, and stroke.
Now entering the discussion are researchers from the University of Massachusetts, Amherst, and Embr Labs (a Massachusetts Institute of Technology spinoff) who say they’ve developed a machine-learning algorithm that can predict a hot flash.
The device, which sells for $299, is already touted as a way to manage menopausal hot flashes.
But once the algorithm is added, the device will be able to “continuously monitor physiological signals – skin temperature, body temperature, sweating, activity level, or heart rate – and identify early indicators that a hot flash is building,” said Michael Busa, PhD, director of the Center for Human Health and Performance at UMass Amherst, who led the team that developed the algorithm.
That data would be sent to a computing platform in the cloud, where the algorithm can flag signs of an impending hot flash, Dr. Busa said. The device would automatically prompt cooling in less than a second, which could effectively stop the hot flash in its tracks or at least help to take the edge off.
Exploring cooling therapy for hot flashes
“There is always tremendous interest in anything that is nonhormonal and effective in treatment of hot flashes,” said Karen Adams, MD, an ob.gyn. and director of the menopause and healthy aging program at Stanford (Calif.) University. (Dr. Adams was not involved in developing this technology.)
Hormone therapy is the primary treatment, easing hot flashes in 3-4 weeks, Dr. Adams said. “But some women do not want to take estrogen, or should not due to medical contraindications.”
Hormone therapy is generally not recommended for people with a history of breast cancer, blood clots, or diseases of their heart or blood vessels. Recent research presented at the annual meeting of the Menopause Society found that hormone therapy may not work as well in women with obesity.
For nonhormonal treatments, the Food and Drug Administration cleared the oral med fezolinetant (Veozah) in May. Antidepressant medications can also be used as a first-line treatment in those who can’t take estrogen. Another oral drug, elinzanetant, is in late-stage clinical trials.
But there has been little clinical investigation – only two small studies, Dr. Adams said – examining cooling therapy as a treatment for hot flashes. That’s something the makers of this device hope to change.
“Despite the fact that seeking cooling relief is a woman’s immediate natural response to the onset of a hot flash, there is limited work done to understand the benefits of this natural therapy,” said Matthew Smith, PhD, chief technology officer at Embr Labs. “This is in part because the technology didn’t exist to deliver cooling in an immediate, reproducible manner.”
The algorithm’s performance has been benchmarked using data from women having hot flashes, Dr. Smith said. Results have been submitted for publication.
The Embr Wave has been shown to help menopausal women with hot flashes sleep better. It has also been tested as a therapy for hot flashes related to cancer treatment.
But to truly evaluate the device as a treatment for hot flashes, it should be tested in randomized trials including a “sham treatment arm” – where some people get the real treatment while others get the sham treatment, Dr. Adams said.
“Device studies tend to have high placebo response rates that can only be truly evaluated when there is a sham treatment in the study,” she said. “If such a device were shown to be safe and effective, we would absolutely recommend it. But we’re a long way from that.”
A version of this article appeared on WebMD.com.
Postmenopausal testosterone for low libido only, doctors say
Your patients may see ads claiming that testosterone replacement therapy (TRT) offers postmenopausal women health benefits beyond restored sex drive: that TRT can improve their mood, energy, and thinking and give them stronger bones and bigger muscles.
How accurate are these claims? According to six experts who talked with this news organization, not very.
“Right now in this country and around the world, testosterone’s only use in postmenopausal women is for libido,” said Adrian Sandra Dobs, MD, MHS, professor of medicine and director of the Johns Hopkins Clinical Research Network at Johns Hopkins Medicine, Baltimore.
“Treating postmenopausal women with testosterone is a rarity. Some physicians and some wellness centers make their money out of prescribing estrogen and testosterone to women in patches, gels, creams, capsules, pellets, and other forms. she added by phone.
“One has to be very careful about using testosterone in women,” Dr. Dobs cautioned. “There’s a lot of hype out there.”
Low testosterone in women has not been well studied, and no testosterone treatments for this condition have been approved by the U.S. Food and Drug Administration. Providers need to adjust male treatment data to their female patients, who require significantly lower doses than males. Contraindications and long-term side effects are poorly understood, said Mary Rosser, MD, PhD, assistant professor of women’s health and director of integrated women’s health at Columbia University Irving Medical Center, New York.
“Despite this preponderance of scientific evidence and recommendations, the myths about testosterone die hard, including that it improves women’s muscle function, endurance, and well-being,” Dr. Rosser said.
“Websites that use compounded products or pellets are not FDA-regulated; therefore, they have no responsibility to prove their claims. They can entice women into using this stuff with all kinds of promises about ‘hormone balancing’ and other meaningless terms. The Endocrine Society statement reviewed the clinical studies on testosterone for various indications surrounding physical endurance, well-being, and mental health – and the studies do not support its use,” Dr. Rosser added.
According to the Australasian Menopause Society, women’s blood testosterone levels tend to peak in their 20s, slowly decline to around 25% of peak levels at menopause, then rise again in later years.
Susan Davis, PhD, and her colleagues at Monash University, Melbourne, found in a study that TRT in postmenopausal women may improve sexual well-being and that side effects include acne and increased hair growth. But they found no benefits for cognition, bone mineral density, body composition, muscle strength, or psychological well-being, and they note that more data are needed on long-term safety.
Postmenopausal testosterone recommended for libido only
“Hypoactive sexual desire disorder (HSDD) is really the only indication for postmenopausal testosterone use,” Nanette F. Santoro, MD, professor and chair of obstetrics and gynecology at the University of Colorado School of Medicine, Aurora, noted by email. “In clinical studies using androgen gel containing testosterone, testosterone treatment has resulted in a mean of one more satisfying sexual encounter per month. Consensus statements issued by the Endocrine Society, The International Menopause Society, and the North American Menopause Society have come to similar conclusions: The only indication for androgen therapy for women is HSDD,” added Santoro, an author of the Endocrine Society statement.
“Sexual health and the sense of well-being are very much related,” Sandra Ann Carson, MD, professor of obstetrics and gynecology at Yale Medicine, New Haven, Conn., said by phone. “So we give testosterone to increase sexual desire. Testosterone is not a treatment for decreased sense of well-being alone. Women who lose their sense of well-being due to depression or other factors need to have a mental health evaluation, not testosterone.”
“Because no female product is presently approved by a national regulatory body, male formulations can be judiciously used in female doses and blood testosterone concentrations must be monitored regularly,” Dr. Rosser said. “The recommendation is for considering use of compounded testosterone for hypoactive sexual desire only; it is against use for overall health and wellness.”
“The real mischief occurs when women are exposed to doses that are supraphysiologic,” Dr. Rosser cautioned. “At high doses that approach and sometimes exceed men’s levels of testosterone, women can have deepening of the voice, adverse changes in cholesterol, and even breast atrophy. This can occur with bioidentical compounded testosterone and with testosterone pellets. The National Academies of Science, Engineering, and Medicine recommend unequivocally that such preparations not be used.”
Not all postmenopausal women should take TRT, said Meredith McClure, MD, assistant professor in the department of obstetrics and gynecology of UT Southwestern Medical School, Dallas, because it has only been shown in trials to help with HSDD.
She advised clinicians to avoid prescribing testosterone to patients who “can’t take estrogen, including if [they] have hormone-sensitive cancer, blood clot risk, liver disease, heart attack, stroke, or undiagnosed genital bleeding.”
TRT for non-libido issues may sometimes be appropriate
“Perhaps women with hip fracture or cancer cachexia could benefit from testosterone to build muscle mass,” said Dr. Dobbs, who is involved in an ongoing study of testosterone treatment in women with hip fracture. “But as yet, we have no proof that testosterone helps.”
In rare cases, Stanley G. Korenman, MD, a reproductive endocrinologist and associate dean for ethics at UCLA Health, treats postmenopausal patients with TRT for reasons other than low libido. “I have a very specialized practice in reproductive endocrinology and internal medicine and am one of very few people in the country who do this kind of management,” he said in an interview. “If my postmenopausal patients have low testosterone and lack energy, I’m willing to give them low doses. If they feel more energetic, we continue, but if they don’t, we stop. I don’t think there’s any risk whatsoever at the low level I prescribe.
“I prescribe standard gel that comes in a squirt bottle, and I suggest they take half a squirt every other day – about one-eighth of a male dose – on the sole of the foot, where hair does not grow.
“I would not prescribe testosterone for bone health. We have bisphosphonates and other much better treatments for that. And I would not prescribe it to someone who is seriously emotionally disturbed or seriously depressed. This is not a treatment for depression.”
“Postmenopausal testosterone is not ‘the latest greatest thing,’ but being very low risk, it’s worth trying once in a while, in the appropriate patient, at the right dose,” Dr. Korenman advised. He cautioned people to “avoid the longevity salespeople who sell all sorts of things in all sorts of doses to try to keep us alive forever.”
All contributors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Your patients may see ads claiming that testosterone replacement therapy (TRT) offers postmenopausal women health benefits beyond restored sex drive: that TRT can improve their mood, energy, and thinking and give them stronger bones and bigger muscles.
How accurate are these claims? According to six experts who talked with this news organization, not very.
“Right now in this country and around the world, testosterone’s only use in postmenopausal women is for libido,” said Adrian Sandra Dobs, MD, MHS, professor of medicine and director of the Johns Hopkins Clinical Research Network at Johns Hopkins Medicine, Baltimore.
“Treating postmenopausal women with testosterone is a rarity. Some physicians and some wellness centers make their money out of prescribing estrogen and testosterone to women in patches, gels, creams, capsules, pellets, and other forms. she added by phone.
“One has to be very careful about using testosterone in women,” Dr. Dobs cautioned. “There’s a lot of hype out there.”
Low testosterone in women has not been well studied, and no testosterone treatments for this condition have been approved by the U.S. Food and Drug Administration. Providers need to adjust male treatment data to their female patients, who require significantly lower doses than males. Contraindications and long-term side effects are poorly understood, said Mary Rosser, MD, PhD, assistant professor of women’s health and director of integrated women’s health at Columbia University Irving Medical Center, New York.
“Despite this preponderance of scientific evidence and recommendations, the myths about testosterone die hard, including that it improves women’s muscle function, endurance, and well-being,” Dr. Rosser said.
“Websites that use compounded products or pellets are not FDA-regulated; therefore, they have no responsibility to prove their claims. They can entice women into using this stuff with all kinds of promises about ‘hormone balancing’ and other meaningless terms. The Endocrine Society statement reviewed the clinical studies on testosterone for various indications surrounding physical endurance, well-being, and mental health – and the studies do not support its use,” Dr. Rosser added.
According to the Australasian Menopause Society, women’s blood testosterone levels tend to peak in their 20s, slowly decline to around 25% of peak levels at menopause, then rise again in later years.
Susan Davis, PhD, and her colleagues at Monash University, Melbourne, found in a study that TRT in postmenopausal women may improve sexual well-being and that side effects include acne and increased hair growth. But they found no benefits for cognition, bone mineral density, body composition, muscle strength, or psychological well-being, and they note that more data are needed on long-term safety.
Postmenopausal testosterone recommended for libido only
“Hypoactive sexual desire disorder (HSDD) is really the only indication for postmenopausal testosterone use,” Nanette F. Santoro, MD, professor and chair of obstetrics and gynecology at the University of Colorado School of Medicine, Aurora, noted by email. “In clinical studies using androgen gel containing testosterone, testosterone treatment has resulted in a mean of one more satisfying sexual encounter per month. Consensus statements issued by the Endocrine Society, The International Menopause Society, and the North American Menopause Society have come to similar conclusions: The only indication for androgen therapy for women is HSDD,” added Santoro, an author of the Endocrine Society statement.
“Sexual health and the sense of well-being are very much related,” Sandra Ann Carson, MD, professor of obstetrics and gynecology at Yale Medicine, New Haven, Conn., said by phone. “So we give testosterone to increase sexual desire. Testosterone is not a treatment for decreased sense of well-being alone. Women who lose their sense of well-being due to depression or other factors need to have a mental health evaluation, not testosterone.”
“Because no female product is presently approved by a national regulatory body, male formulations can be judiciously used in female doses and blood testosterone concentrations must be monitored regularly,” Dr. Rosser said. “The recommendation is for considering use of compounded testosterone for hypoactive sexual desire only; it is against use for overall health and wellness.”
“The real mischief occurs when women are exposed to doses that are supraphysiologic,” Dr. Rosser cautioned. “At high doses that approach and sometimes exceed men’s levels of testosterone, women can have deepening of the voice, adverse changes in cholesterol, and even breast atrophy. This can occur with bioidentical compounded testosterone and with testosterone pellets. The National Academies of Science, Engineering, and Medicine recommend unequivocally that such preparations not be used.”
Not all postmenopausal women should take TRT, said Meredith McClure, MD, assistant professor in the department of obstetrics and gynecology of UT Southwestern Medical School, Dallas, because it has only been shown in trials to help with HSDD.
She advised clinicians to avoid prescribing testosterone to patients who “can’t take estrogen, including if [they] have hormone-sensitive cancer, blood clot risk, liver disease, heart attack, stroke, or undiagnosed genital bleeding.”
TRT for non-libido issues may sometimes be appropriate
“Perhaps women with hip fracture or cancer cachexia could benefit from testosterone to build muscle mass,” said Dr. Dobbs, who is involved in an ongoing study of testosterone treatment in women with hip fracture. “But as yet, we have no proof that testosterone helps.”
In rare cases, Stanley G. Korenman, MD, a reproductive endocrinologist and associate dean for ethics at UCLA Health, treats postmenopausal patients with TRT for reasons other than low libido. “I have a very specialized practice in reproductive endocrinology and internal medicine and am one of very few people in the country who do this kind of management,” he said in an interview. “If my postmenopausal patients have low testosterone and lack energy, I’m willing to give them low doses. If they feel more energetic, we continue, but if they don’t, we stop. I don’t think there’s any risk whatsoever at the low level I prescribe.
“I prescribe standard gel that comes in a squirt bottle, and I suggest they take half a squirt every other day – about one-eighth of a male dose – on the sole of the foot, where hair does not grow.
“I would not prescribe testosterone for bone health. We have bisphosphonates and other much better treatments for that. And I would not prescribe it to someone who is seriously emotionally disturbed or seriously depressed. This is not a treatment for depression.”
“Postmenopausal testosterone is not ‘the latest greatest thing,’ but being very low risk, it’s worth trying once in a while, in the appropriate patient, at the right dose,” Dr. Korenman advised. He cautioned people to “avoid the longevity salespeople who sell all sorts of things in all sorts of doses to try to keep us alive forever.”
All contributors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Your patients may see ads claiming that testosterone replacement therapy (TRT) offers postmenopausal women health benefits beyond restored sex drive: that TRT can improve their mood, energy, and thinking and give them stronger bones and bigger muscles.
How accurate are these claims? According to six experts who talked with this news organization, not very.
“Right now in this country and around the world, testosterone’s only use in postmenopausal women is for libido,” said Adrian Sandra Dobs, MD, MHS, professor of medicine and director of the Johns Hopkins Clinical Research Network at Johns Hopkins Medicine, Baltimore.
“Treating postmenopausal women with testosterone is a rarity. Some physicians and some wellness centers make their money out of prescribing estrogen and testosterone to women in patches, gels, creams, capsules, pellets, and other forms. she added by phone.
“One has to be very careful about using testosterone in women,” Dr. Dobs cautioned. “There’s a lot of hype out there.”
Low testosterone in women has not been well studied, and no testosterone treatments for this condition have been approved by the U.S. Food and Drug Administration. Providers need to adjust male treatment data to their female patients, who require significantly lower doses than males. Contraindications and long-term side effects are poorly understood, said Mary Rosser, MD, PhD, assistant professor of women’s health and director of integrated women’s health at Columbia University Irving Medical Center, New York.
“Despite this preponderance of scientific evidence and recommendations, the myths about testosterone die hard, including that it improves women’s muscle function, endurance, and well-being,” Dr. Rosser said.
“Websites that use compounded products or pellets are not FDA-regulated; therefore, they have no responsibility to prove their claims. They can entice women into using this stuff with all kinds of promises about ‘hormone balancing’ and other meaningless terms. The Endocrine Society statement reviewed the clinical studies on testosterone for various indications surrounding physical endurance, well-being, and mental health – and the studies do not support its use,” Dr. Rosser added.
According to the Australasian Menopause Society, women’s blood testosterone levels tend to peak in their 20s, slowly decline to around 25% of peak levels at menopause, then rise again in later years.
Susan Davis, PhD, and her colleagues at Monash University, Melbourne, found in a study that TRT in postmenopausal women may improve sexual well-being and that side effects include acne and increased hair growth. But they found no benefits for cognition, bone mineral density, body composition, muscle strength, or psychological well-being, and they note that more data are needed on long-term safety.
Postmenopausal testosterone recommended for libido only
“Hypoactive sexual desire disorder (HSDD) is really the only indication for postmenopausal testosterone use,” Nanette F. Santoro, MD, professor and chair of obstetrics and gynecology at the University of Colorado School of Medicine, Aurora, noted by email. “In clinical studies using androgen gel containing testosterone, testosterone treatment has resulted in a mean of one more satisfying sexual encounter per month. Consensus statements issued by the Endocrine Society, The International Menopause Society, and the North American Menopause Society have come to similar conclusions: The only indication for androgen therapy for women is HSDD,” added Santoro, an author of the Endocrine Society statement.
“Sexual health and the sense of well-being are very much related,” Sandra Ann Carson, MD, professor of obstetrics and gynecology at Yale Medicine, New Haven, Conn., said by phone. “So we give testosterone to increase sexual desire. Testosterone is not a treatment for decreased sense of well-being alone. Women who lose their sense of well-being due to depression or other factors need to have a mental health evaluation, not testosterone.”
“Because no female product is presently approved by a national regulatory body, male formulations can be judiciously used in female doses and blood testosterone concentrations must be monitored regularly,” Dr. Rosser said. “The recommendation is for considering use of compounded testosterone for hypoactive sexual desire only; it is against use for overall health and wellness.”
“The real mischief occurs when women are exposed to doses that are supraphysiologic,” Dr. Rosser cautioned. “At high doses that approach and sometimes exceed men’s levels of testosterone, women can have deepening of the voice, adverse changes in cholesterol, and even breast atrophy. This can occur with bioidentical compounded testosterone and with testosterone pellets. The National Academies of Science, Engineering, and Medicine recommend unequivocally that such preparations not be used.”
Not all postmenopausal women should take TRT, said Meredith McClure, MD, assistant professor in the department of obstetrics and gynecology of UT Southwestern Medical School, Dallas, because it has only been shown in trials to help with HSDD.
She advised clinicians to avoid prescribing testosterone to patients who “can’t take estrogen, including if [they] have hormone-sensitive cancer, blood clot risk, liver disease, heart attack, stroke, or undiagnosed genital bleeding.”
TRT for non-libido issues may sometimes be appropriate
“Perhaps women with hip fracture or cancer cachexia could benefit from testosterone to build muscle mass,” said Dr. Dobbs, who is involved in an ongoing study of testosterone treatment in women with hip fracture. “But as yet, we have no proof that testosterone helps.”
In rare cases, Stanley G. Korenman, MD, a reproductive endocrinologist and associate dean for ethics at UCLA Health, treats postmenopausal patients with TRT for reasons other than low libido. “I have a very specialized practice in reproductive endocrinology and internal medicine and am one of very few people in the country who do this kind of management,” he said in an interview. “If my postmenopausal patients have low testosterone and lack energy, I’m willing to give them low doses. If they feel more energetic, we continue, but if they don’t, we stop. I don’t think there’s any risk whatsoever at the low level I prescribe.
“I prescribe standard gel that comes in a squirt bottle, and I suggest they take half a squirt every other day – about one-eighth of a male dose – on the sole of the foot, where hair does not grow.
“I would not prescribe testosterone for bone health. We have bisphosphonates and other much better treatments for that. And I would not prescribe it to someone who is seriously emotionally disturbed or seriously depressed. This is not a treatment for depression.”
“Postmenopausal testosterone is not ‘the latest greatest thing,’ but being very low risk, it’s worth trying once in a while, in the appropriate patient, at the right dose,” Dr. Korenman advised. He cautioned people to “avoid the longevity salespeople who sell all sorts of things in all sorts of doses to try to keep us alive forever.”
All contributors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Obesity boosts gestational diabetes risk in women with PCOS
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JOURNAL OF OBSTETRICS AND GYNAECOLOGY CANADA
Lack of racial, ethnic diversity in cryopreserved donor sperm in the U.S.
, according to a study presented at the American Society for Reproductive Medicine’s 2023 meeting.
“This really highlights the need to identify barriers to increase recruitment of these donors so that we can support family-building for all populations,” said Lauren Gibbs, MD, a resident in the department of obstetrics and gynecology at the Morehouse School of Medicine in Atlanta.
Dr. Gibbs and her colleagues compared the racial and ethnic makeup of sperm donors from online and self-reported profiles at 14 of the largest donor banks in the United States for March and April of 2023. Historical data were pulled from two large, national banks. The investigators compared these data to census estimates from 2021 for men between the ages of 18 and 44 years.
Donors who identified as Hispanic (10.9%) or Black (3.3%) were significantly underrepresented as compared to the U.S. population, of which Hispanic men compose 22% and Black men make up 13.3%.
Asian donors were overrepresented, making up 21.9% of the donors but only 6.5% of the U.S. population. White donors were proportionately represented in relation to national demographics, making up 56.6% of the donors and representing 55% of the U.S. population, according to the researchers. None of the donors identified as Native/Hawaiian/Pacific Islander or American Indian/Alaskan Natives; these groups represent 0.22% and 0.79% of the U.S. population, respectively.
“Next steps will be figuring out why this is happening and how to address it,” said Valerie L Baker, MD, director in the division of reproductive endocrinology and infertility at Johns Hopkins Medicine in Lutherville, Md., who was not involved in the study.
The study sheds light on the need to identify and address the barriers that discourage potential donors from underrepresented groups from participating in sperm donation, according to Kimball Pomeroy, PhD, scientific director at the World Egg and Sperm Bank in Scottsdale, Ariz.
“Sometimes there are inhibitors of different ethnic groups to want to act as sperm or egg donors, so trying to understand if that’s the case is important; but I’m sure a lot of it is also related to access,” Dr. Pomeroy, who was not part of the study team, said in an interview.
Longitudinal data from the two national donor banks did not indicate any significant increase or decrease in donation trends across the 5-year period from 2018 to 2022, highlighting the persisting issue of representation disparities. Dr. Gibbs said strategies need to be developed to increase recruitment of donors from underrepresented groups. Increasing the diversity of the donor pool will ultimately support family-building options for all patients, according to Dr. Gibbs.
Funding for the study was provided by the EMD Serono REI Diversity Fellowship Grant. Dr. Gibbs reports no relevant financial relationships.
, according to a study presented at the American Society for Reproductive Medicine’s 2023 meeting.
“This really highlights the need to identify barriers to increase recruitment of these donors so that we can support family-building for all populations,” said Lauren Gibbs, MD, a resident in the department of obstetrics and gynecology at the Morehouse School of Medicine in Atlanta.
Dr. Gibbs and her colleagues compared the racial and ethnic makeup of sperm donors from online and self-reported profiles at 14 of the largest donor banks in the United States for March and April of 2023. Historical data were pulled from two large, national banks. The investigators compared these data to census estimates from 2021 for men between the ages of 18 and 44 years.
Donors who identified as Hispanic (10.9%) or Black (3.3%) were significantly underrepresented as compared to the U.S. population, of which Hispanic men compose 22% and Black men make up 13.3%.
Asian donors were overrepresented, making up 21.9% of the donors but only 6.5% of the U.S. population. White donors were proportionately represented in relation to national demographics, making up 56.6% of the donors and representing 55% of the U.S. population, according to the researchers. None of the donors identified as Native/Hawaiian/Pacific Islander or American Indian/Alaskan Natives; these groups represent 0.22% and 0.79% of the U.S. population, respectively.
“Next steps will be figuring out why this is happening and how to address it,” said Valerie L Baker, MD, director in the division of reproductive endocrinology and infertility at Johns Hopkins Medicine in Lutherville, Md., who was not involved in the study.
The study sheds light on the need to identify and address the barriers that discourage potential donors from underrepresented groups from participating in sperm donation, according to Kimball Pomeroy, PhD, scientific director at the World Egg and Sperm Bank in Scottsdale, Ariz.
“Sometimes there are inhibitors of different ethnic groups to want to act as sperm or egg donors, so trying to understand if that’s the case is important; but I’m sure a lot of it is also related to access,” Dr. Pomeroy, who was not part of the study team, said in an interview.
Longitudinal data from the two national donor banks did not indicate any significant increase or decrease in donation trends across the 5-year period from 2018 to 2022, highlighting the persisting issue of representation disparities. Dr. Gibbs said strategies need to be developed to increase recruitment of donors from underrepresented groups. Increasing the diversity of the donor pool will ultimately support family-building options for all patients, according to Dr. Gibbs.
Funding for the study was provided by the EMD Serono REI Diversity Fellowship Grant. Dr. Gibbs reports no relevant financial relationships.
, according to a study presented at the American Society for Reproductive Medicine’s 2023 meeting.
“This really highlights the need to identify barriers to increase recruitment of these donors so that we can support family-building for all populations,” said Lauren Gibbs, MD, a resident in the department of obstetrics and gynecology at the Morehouse School of Medicine in Atlanta.
Dr. Gibbs and her colleagues compared the racial and ethnic makeup of sperm donors from online and self-reported profiles at 14 of the largest donor banks in the United States for March and April of 2023. Historical data were pulled from two large, national banks. The investigators compared these data to census estimates from 2021 for men between the ages of 18 and 44 years.
Donors who identified as Hispanic (10.9%) or Black (3.3%) were significantly underrepresented as compared to the U.S. population, of which Hispanic men compose 22% and Black men make up 13.3%.
Asian donors were overrepresented, making up 21.9% of the donors but only 6.5% of the U.S. population. White donors were proportionately represented in relation to national demographics, making up 56.6% of the donors and representing 55% of the U.S. population, according to the researchers. None of the donors identified as Native/Hawaiian/Pacific Islander or American Indian/Alaskan Natives; these groups represent 0.22% and 0.79% of the U.S. population, respectively.
“Next steps will be figuring out why this is happening and how to address it,” said Valerie L Baker, MD, director in the division of reproductive endocrinology and infertility at Johns Hopkins Medicine in Lutherville, Md., who was not involved in the study.
The study sheds light on the need to identify and address the barriers that discourage potential donors from underrepresented groups from participating in sperm donation, according to Kimball Pomeroy, PhD, scientific director at the World Egg and Sperm Bank in Scottsdale, Ariz.
“Sometimes there are inhibitors of different ethnic groups to want to act as sperm or egg donors, so trying to understand if that’s the case is important; but I’m sure a lot of it is also related to access,” Dr. Pomeroy, who was not part of the study team, said in an interview.
Longitudinal data from the two national donor banks did not indicate any significant increase or decrease in donation trends across the 5-year period from 2018 to 2022, highlighting the persisting issue of representation disparities. Dr. Gibbs said strategies need to be developed to increase recruitment of donors from underrepresented groups. Increasing the diversity of the donor pool will ultimately support family-building options for all patients, according to Dr. Gibbs.
Funding for the study was provided by the EMD Serono REI Diversity Fellowship Grant. Dr. Gibbs reports no relevant financial relationships.
FROM ASRM 2023
Postmenopausal stress linked to mood, cognitive symptoms
PHILADELPHIA – , according to research presented at the annual meeting of the Menopause Society (formerly the North American Menopause Society).
“This work suggests that markers of hypothalamic-pituitary-axis activation that capture total cortisol secretion over multiple months, [such as] hair cortisol, strongly correlate with cognitive performance on attention and working memory tasks, whereas measures of more acute cortisol, [such as] salivary cortisol, may be more strongly associated with depression symptom severity and verbal learning,” Christina Metcalf, PhD, an assistant professor of psychiatry in the Colorado Center for Women’s Behavioral Health and Wellness at the University of Colorado at Denver, Aurora, told attendees. “Given the associations with chronic stress, there’s a lot of potential here to increase our knowledge about how women are doing and managing stress and life stressors during this life transition,” she said.
The study involved collecting hair and saliva samples from 43 healthy women in late perimenopause or early postmenopause with an average age of 51. The participants were predominantly white and college educated. The hair sample was taken within 2 cm of the scalp, and the saliva samples were collected the day after the hair sample collection, at the start and end of a 30-minute rest period that took place between 2:00 and 3:00 p.m. local time.
All the participants had an intact uterus and at least one ovary. None of the participants were current smokers or had recent alcohol or drug dependence, and none had used hormones within the previous 6 months. The study also excluded women who were pregnant or breastfeeding, who had bleached hair or no hair, who were taking steroids, beta blockers or opioid medication, and who had recently taken NSAIDS.
Measuring hair cortisol more feasible
The study was conducted remotely, with participants using video conferencing to communicate with the study personnel and then completing study procedures at home, including 2 days of cognitive testing with the California Verbal Learning Test – Third Edition and the n-back and continuous performance tasks. The participants also completed the Center for Epidemiologic Studies Depression Scale (CES-D).
Participants with higher levels of hair cortisol and salivary cortisol also had more severe depression symptoms (P < .001). Hair cortisol was also significantly associated with attention and working memory: Women with higher levels had fewer correct answers on the 0-back and 1-back trials (P < .01) and made more mistakes on the 2-back trial (P < .001). They also scored with less specificity on the continuous performance tasks (P = .022).
Although no association existed between hair cortisol levels and verbal learning or verbal memory (P > .05), participants with higher hair cortisol did score worse on the immediate recall trials (P = .034). Salivary cortisol levels, on the other hand, showed no association with memory recall trials, attention or working memory (P > .05).
Measuring cortisol from hair samples is more feasible than using saliva samples and may offer valuable insights regarding hypothalamic-pituitary-axis activity “to consider alongside the cognitive and mental health of late peri-/early postmenopausal women,” Dr. Metcalf told attendees. The next step is to find out whether the hypothalamic-pituitary-axis axis is a modifiable biomarker that can be used to improve executive function.
The study was limited by its small population, its cross-sectional design, and the lack of covariates in the current analyses.
Monitor symptoms in midlife
Hadine Joffe, MD, MSc, a professor of psychiatry and executive director of the Mary Horrigan Connors Center for Women’s Health and Gender Biology at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said the study findings were not surprising given how common the complaints of stress and depressive symptoms are.
“Mood changes are linked with acute, immediate cortisol levels at the same point in time, and cognitive symptoms were linked to more chronically elevated cortisol levels,” Dr. Joffe said in an interview. “Women and their providers should monitor for these challenging brain symptoms in midlife as they affect performance and quality of life and are linked with changes in the HPA axis as stress biomarkers.”
Because the study is small and has a cross-sectional design, it’s not possible to determine the direction of the associations or to make any inferences about causation, Dr. Joffe said.
“We cannot make the conclusion that stress is adversely affecting mood and cognitive performance given the design limitations. It is possible that mood and cognitive issues contributed to these stress markers,” Dr. Joffe said.“However, it is known that the experience of stress is linked with vulnerability to mood and cognitive symptoms, and also that mood and cognitive symptoms induce significant stress.”
The research was funded by the Menopause Society, Colorado University, the Ludeman Family Center for Women’s Health Research, the National Institute of Mental Health, and the National Institute of Aging. Dr. Metcalf had no disclosures. Dr. Joffe has received grant support from Merck, Pfizer and Sage, and has been a consultant or advisor for Bayer, Merck and Hello Therapeutics.
PHILADELPHIA – , according to research presented at the annual meeting of the Menopause Society (formerly the North American Menopause Society).
“This work suggests that markers of hypothalamic-pituitary-axis activation that capture total cortisol secretion over multiple months, [such as] hair cortisol, strongly correlate with cognitive performance on attention and working memory tasks, whereas measures of more acute cortisol, [such as] salivary cortisol, may be more strongly associated with depression symptom severity and verbal learning,” Christina Metcalf, PhD, an assistant professor of psychiatry in the Colorado Center for Women’s Behavioral Health and Wellness at the University of Colorado at Denver, Aurora, told attendees. “Given the associations with chronic stress, there’s a lot of potential here to increase our knowledge about how women are doing and managing stress and life stressors during this life transition,” she said.
The study involved collecting hair and saliva samples from 43 healthy women in late perimenopause or early postmenopause with an average age of 51. The participants were predominantly white and college educated. The hair sample was taken within 2 cm of the scalp, and the saliva samples were collected the day after the hair sample collection, at the start and end of a 30-minute rest period that took place between 2:00 and 3:00 p.m. local time.
All the participants had an intact uterus and at least one ovary. None of the participants were current smokers or had recent alcohol or drug dependence, and none had used hormones within the previous 6 months. The study also excluded women who were pregnant or breastfeeding, who had bleached hair or no hair, who were taking steroids, beta blockers or opioid medication, and who had recently taken NSAIDS.
Measuring hair cortisol more feasible
The study was conducted remotely, with participants using video conferencing to communicate with the study personnel and then completing study procedures at home, including 2 days of cognitive testing with the California Verbal Learning Test – Third Edition and the n-back and continuous performance tasks. The participants also completed the Center for Epidemiologic Studies Depression Scale (CES-D).
Participants with higher levels of hair cortisol and salivary cortisol also had more severe depression symptoms (P < .001). Hair cortisol was also significantly associated with attention and working memory: Women with higher levels had fewer correct answers on the 0-back and 1-back trials (P < .01) and made more mistakes on the 2-back trial (P < .001). They also scored with less specificity on the continuous performance tasks (P = .022).
Although no association existed between hair cortisol levels and verbal learning or verbal memory (P > .05), participants with higher hair cortisol did score worse on the immediate recall trials (P = .034). Salivary cortisol levels, on the other hand, showed no association with memory recall trials, attention or working memory (P > .05).
Measuring cortisol from hair samples is more feasible than using saliva samples and may offer valuable insights regarding hypothalamic-pituitary-axis activity “to consider alongside the cognitive and mental health of late peri-/early postmenopausal women,” Dr. Metcalf told attendees. The next step is to find out whether the hypothalamic-pituitary-axis axis is a modifiable biomarker that can be used to improve executive function.
The study was limited by its small population, its cross-sectional design, and the lack of covariates in the current analyses.
Monitor symptoms in midlife
Hadine Joffe, MD, MSc, a professor of psychiatry and executive director of the Mary Horrigan Connors Center for Women’s Health and Gender Biology at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said the study findings were not surprising given how common the complaints of stress and depressive symptoms are.
“Mood changes are linked with acute, immediate cortisol levels at the same point in time, and cognitive symptoms were linked to more chronically elevated cortisol levels,” Dr. Joffe said in an interview. “Women and their providers should monitor for these challenging brain symptoms in midlife as they affect performance and quality of life and are linked with changes in the HPA axis as stress biomarkers.”
Because the study is small and has a cross-sectional design, it’s not possible to determine the direction of the associations or to make any inferences about causation, Dr. Joffe said.
“We cannot make the conclusion that stress is adversely affecting mood and cognitive performance given the design limitations. It is possible that mood and cognitive issues contributed to these stress markers,” Dr. Joffe said.“However, it is known that the experience of stress is linked with vulnerability to mood and cognitive symptoms, and also that mood and cognitive symptoms induce significant stress.”
The research was funded by the Menopause Society, Colorado University, the Ludeman Family Center for Women’s Health Research, the National Institute of Mental Health, and the National Institute of Aging. Dr. Metcalf had no disclosures. Dr. Joffe has received grant support from Merck, Pfizer and Sage, and has been a consultant or advisor for Bayer, Merck and Hello Therapeutics.
PHILADELPHIA – , according to research presented at the annual meeting of the Menopause Society (formerly the North American Menopause Society).
“This work suggests that markers of hypothalamic-pituitary-axis activation that capture total cortisol secretion over multiple months, [such as] hair cortisol, strongly correlate with cognitive performance on attention and working memory tasks, whereas measures of more acute cortisol, [such as] salivary cortisol, may be more strongly associated with depression symptom severity and verbal learning,” Christina Metcalf, PhD, an assistant professor of psychiatry in the Colorado Center for Women’s Behavioral Health and Wellness at the University of Colorado at Denver, Aurora, told attendees. “Given the associations with chronic stress, there’s a lot of potential here to increase our knowledge about how women are doing and managing stress and life stressors during this life transition,” she said.
The study involved collecting hair and saliva samples from 43 healthy women in late perimenopause or early postmenopause with an average age of 51. The participants were predominantly white and college educated. The hair sample was taken within 2 cm of the scalp, and the saliva samples were collected the day after the hair sample collection, at the start and end of a 30-minute rest period that took place between 2:00 and 3:00 p.m. local time.
All the participants had an intact uterus and at least one ovary. None of the participants were current smokers or had recent alcohol or drug dependence, and none had used hormones within the previous 6 months. The study also excluded women who were pregnant or breastfeeding, who had bleached hair or no hair, who were taking steroids, beta blockers or opioid medication, and who had recently taken NSAIDS.
Measuring hair cortisol more feasible
The study was conducted remotely, with participants using video conferencing to communicate with the study personnel and then completing study procedures at home, including 2 days of cognitive testing with the California Verbal Learning Test – Third Edition and the n-back and continuous performance tasks. The participants also completed the Center for Epidemiologic Studies Depression Scale (CES-D).
Participants with higher levels of hair cortisol and salivary cortisol also had more severe depression symptoms (P < .001). Hair cortisol was also significantly associated with attention and working memory: Women with higher levels had fewer correct answers on the 0-back and 1-back trials (P < .01) and made more mistakes on the 2-back trial (P < .001). They also scored with less specificity on the continuous performance tasks (P = .022).
Although no association existed between hair cortisol levels and verbal learning or verbal memory (P > .05), participants with higher hair cortisol did score worse on the immediate recall trials (P = .034). Salivary cortisol levels, on the other hand, showed no association with memory recall trials, attention or working memory (P > .05).
Measuring cortisol from hair samples is more feasible than using saliva samples and may offer valuable insights regarding hypothalamic-pituitary-axis activity “to consider alongside the cognitive and mental health of late peri-/early postmenopausal women,” Dr. Metcalf told attendees. The next step is to find out whether the hypothalamic-pituitary-axis axis is a modifiable biomarker that can be used to improve executive function.
The study was limited by its small population, its cross-sectional design, and the lack of covariates in the current analyses.
Monitor symptoms in midlife
Hadine Joffe, MD, MSc, a professor of psychiatry and executive director of the Mary Horrigan Connors Center for Women’s Health and Gender Biology at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said the study findings were not surprising given how common the complaints of stress and depressive symptoms are.
“Mood changes are linked with acute, immediate cortisol levels at the same point in time, and cognitive symptoms were linked to more chronically elevated cortisol levels,” Dr. Joffe said in an interview. “Women and their providers should monitor for these challenging brain symptoms in midlife as they affect performance and quality of life and are linked with changes in the HPA axis as stress biomarkers.”
Because the study is small and has a cross-sectional design, it’s not possible to determine the direction of the associations or to make any inferences about causation, Dr. Joffe said.
“We cannot make the conclusion that stress is adversely affecting mood and cognitive performance given the design limitations. It is possible that mood and cognitive issues contributed to these stress markers,” Dr. Joffe said.“However, it is known that the experience of stress is linked with vulnerability to mood and cognitive symptoms, and also that mood and cognitive symptoms induce significant stress.”
The research was funded by the Menopause Society, Colorado University, the Ludeman Family Center for Women’s Health Research, the National Institute of Mental Health, and the National Institute of Aging. Dr. Metcalf had no disclosures. Dr. Joffe has received grant support from Merck, Pfizer and Sage, and has been a consultant or advisor for Bayer, Merck and Hello Therapeutics.
AT NAMS 2023