User login
Probiotics showed slight promise in post-resection Crohn’s prevention
A mixture of eight probiotic bacterial strains only somewhat outperformed placebo for preventing endoscopic recurrence after ileal resection in Crohn’s disease patients, according to a multicenter, randomized trial.
After 90 days of treatment, 9.3% of patients who received the probiotic mixture (VSL#3) had developed severe endoscopic recurrence, compared with 15.7% of the placebo group (P = .19), reported Dr. Richard Fedorak of the University of Alberta, Edmonton, and his associates.
The recurrence rate for the placebo group was about two-thirds lower than what the researchers had expected based on the sample size calculation, they noted. But the probiotic blend was linked to significantly significant decreases in colonic mucosal levels of proinflammatory cytokines, they reported (Clin. Gastroenterol. Hepatol. 2014 Nov. 6 [doi:10.1016/j.cgh.2014.10.031]).
Investigators have tested probiotics as a preventive therapy for Crohn’s disease because patients with active disease have less diverse intestinal microbiota, compared with those with quiescent disease or healthy controls. Past studies of single-strain probiotics have shown them to be no better than placebo for preventing endoscopic recurrence.
But in one small study, rifampin followed by VSL#3 outperformed mesalamine at 1 year (Gastroenterology 2000;118:A781), the researchers noted. “This mixture could confer protective effects where single-strain or lactobacillus-only formulations had failed,” they hypothesized.
To test that theory, the investigators randomized 120 patients with Crohn’s disease who had undergone ileal resection and ileocolonic anastomosis to twice-daily VSL#3 or placebo. Treatment began within 30 days after surgery and continued for 90 days, after which all patients received open-label VSL#3 for another 9 months.
Among patients who had nonsevere endoscopic lesions at day 90, 1-year rates of severe endoscopic recurrence were 10% for the early VSL#3 group, compared with 26.7% for the late VSL#3 group (P = .09), said the researchers. Likewise, combined rates of severe recurrence on days 90 and 365 were not statistically different, they reported. However, the early VSL#3 group had lower mucosal levels of 13 pro-inflammatory cytokines, compared with patients who received placebo until day 90 (P < .05). Measures of Crohn’s disease activity and disease-related quality of life scores were similar for both groups.
“Early treatment with VSL#3 had a larger effect than late treatment,” concluded the investigators. “Future larger studies will be needed to confirm the effect of VSL#3 in prevention of postoperative recurrence.”
The study was funded by VSL Pharmaceuticals, the Canadian Institutes of Health Research, and Crohn’s and Colitis Foundation of Canada. Dr. Fedorak reported having served on a speakers bureau for VSL Pharmaceuticals. The other authors declared no relevant conflicts of interest.
A mixture of eight probiotic bacterial strains only somewhat outperformed placebo for preventing endoscopic recurrence after ileal resection in Crohn’s disease patients, according to a multicenter, randomized trial.
After 90 days of treatment, 9.3% of patients who received the probiotic mixture (VSL#3) had developed severe endoscopic recurrence, compared with 15.7% of the placebo group (P = .19), reported Dr. Richard Fedorak of the University of Alberta, Edmonton, and his associates.
The recurrence rate for the placebo group was about two-thirds lower than what the researchers had expected based on the sample size calculation, they noted. But the probiotic blend was linked to significantly significant decreases in colonic mucosal levels of proinflammatory cytokines, they reported (Clin. Gastroenterol. Hepatol. 2014 Nov. 6 [doi:10.1016/j.cgh.2014.10.031]).
Investigators have tested probiotics as a preventive therapy for Crohn’s disease because patients with active disease have less diverse intestinal microbiota, compared with those with quiescent disease or healthy controls. Past studies of single-strain probiotics have shown them to be no better than placebo for preventing endoscopic recurrence.
But in one small study, rifampin followed by VSL#3 outperformed mesalamine at 1 year (Gastroenterology 2000;118:A781), the researchers noted. “This mixture could confer protective effects where single-strain or lactobacillus-only formulations had failed,” they hypothesized.
To test that theory, the investigators randomized 120 patients with Crohn’s disease who had undergone ileal resection and ileocolonic anastomosis to twice-daily VSL#3 or placebo. Treatment began within 30 days after surgery and continued for 90 days, after which all patients received open-label VSL#3 for another 9 months.
Among patients who had nonsevere endoscopic lesions at day 90, 1-year rates of severe endoscopic recurrence were 10% for the early VSL#3 group, compared with 26.7% for the late VSL#3 group (P = .09), said the researchers. Likewise, combined rates of severe recurrence on days 90 and 365 were not statistically different, they reported. However, the early VSL#3 group had lower mucosal levels of 13 pro-inflammatory cytokines, compared with patients who received placebo until day 90 (P < .05). Measures of Crohn’s disease activity and disease-related quality of life scores were similar for both groups.
“Early treatment with VSL#3 had a larger effect than late treatment,” concluded the investigators. “Future larger studies will be needed to confirm the effect of VSL#3 in prevention of postoperative recurrence.”
The study was funded by VSL Pharmaceuticals, the Canadian Institutes of Health Research, and Crohn’s and Colitis Foundation of Canada. Dr. Fedorak reported having served on a speakers bureau for VSL Pharmaceuticals. The other authors declared no relevant conflicts of interest.
A mixture of eight probiotic bacterial strains only somewhat outperformed placebo for preventing endoscopic recurrence after ileal resection in Crohn’s disease patients, according to a multicenter, randomized trial.
After 90 days of treatment, 9.3% of patients who received the probiotic mixture (VSL#3) had developed severe endoscopic recurrence, compared with 15.7% of the placebo group (P = .19), reported Dr. Richard Fedorak of the University of Alberta, Edmonton, and his associates.
The recurrence rate for the placebo group was about two-thirds lower than what the researchers had expected based on the sample size calculation, they noted. But the probiotic blend was linked to significantly significant decreases in colonic mucosal levels of proinflammatory cytokines, they reported (Clin. Gastroenterol. Hepatol. 2014 Nov. 6 [doi:10.1016/j.cgh.2014.10.031]).
Investigators have tested probiotics as a preventive therapy for Crohn’s disease because patients with active disease have less diverse intestinal microbiota, compared with those with quiescent disease or healthy controls. Past studies of single-strain probiotics have shown them to be no better than placebo for preventing endoscopic recurrence.
But in one small study, rifampin followed by VSL#3 outperformed mesalamine at 1 year (Gastroenterology 2000;118:A781), the researchers noted. “This mixture could confer protective effects where single-strain or lactobacillus-only formulations had failed,” they hypothesized.
To test that theory, the investigators randomized 120 patients with Crohn’s disease who had undergone ileal resection and ileocolonic anastomosis to twice-daily VSL#3 or placebo. Treatment began within 30 days after surgery and continued for 90 days, after which all patients received open-label VSL#3 for another 9 months.
Among patients who had nonsevere endoscopic lesions at day 90, 1-year rates of severe endoscopic recurrence were 10% for the early VSL#3 group, compared with 26.7% for the late VSL#3 group (P = .09), said the researchers. Likewise, combined rates of severe recurrence on days 90 and 365 were not statistically different, they reported. However, the early VSL#3 group had lower mucosal levels of 13 pro-inflammatory cytokines, compared with patients who received placebo until day 90 (P < .05). Measures of Crohn’s disease activity and disease-related quality of life scores were similar for both groups.
“Early treatment with VSL#3 had a larger effect than late treatment,” concluded the investigators. “Future larger studies will be needed to confirm the effect of VSL#3 in prevention of postoperative recurrence.”
The study was funded by VSL Pharmaceuticals, the Canadian Institutes of Health Research, and Crohn’s and Colitis Foundation of Canada. Dr. Fedorak reported having served on a speakers bureau for VSL Pharmaceuticals. The other authors declared no relevant conflicts of interest.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: A mixture of eight bacterial probiotic strains somewhat outperformed placebo for preventing endoscopic recurrence in patients with Crohn’s disease.
Major finding: At day 90, severe endoscopic recurrence affected 9.3% of the treatment group and 15.7% of the placebo group (P =. 19)
Data source: Multicenter, randomized, double-blind study of 119 patients who had undergone ileal resection for Crohn’s disease.
Disclosures: The study was funded by VSL Pharmaceuticals, the Canadian Institutes of Health Research, and Crohn’s and Colitis Foundation of Canada. Dr. Fedorak reported having served on a speaker bureau for VSL Pharmaceuticals. The other authors declared no relevant conflicts of interest.
HALS and LAP colectomy each offer unique benefits
CHICAGO – A nationwide analysis comparing straight versus hand-assisted laparoscopic colectomy appears to have resulted in a draw.
Despite being used more commonly in patients with higher body mass index and comorbidities, hand-assisted laparoscopic surgery (HALS) resulted in a significantly shorter operative time than did straight laparoscopic surgery (LAP) (171 minutes vs. 178.8 minutes; P < .001).
On the other hand, LAP was associated with a significantly shorter length of stay (5.9 days vs. 6.0 days; P < .001), fewer complications (15.9% vs. 18.2%; P = .006), fewer specifically superficial skin infections (4.1% vs. 5.4%; P = .007), and less prolonged postoperative ileus (7.5% vs. 9.1%; P = .01).
The differences were statistically significant likely because of the high volume of patients in each group, but are unlikely to be clinically significant in terms of patient care, senior study author Dr. I. Emre Gorgun said at the annual meeting of the Central Surgical Association.
CSA incoming president Dr. Scott A. Gruber, chief of staff at the John D. Dingell VA Medical Center in Detroit, agreed. “There were very, very small differences. I think they were trying to show that the two [approaches] are virtually equivalent,” he said in an interview. “Each has its advantages and disadvantages, but the HALS technique was used in more complex, more high-risk patients,” Dr. Gruber said.
HALS bridges the learning gap between open and straight laparoscopic surgery, which has been slow to gain adoption in colorectal surgery because it is technically demanding and has a steep learning curve, said Dr. Gorgun, a colorectal surgeon at the Cleveland Clinic. HALS allows surgeons to regain tactile sensation and manual retraction, enabling them to perform even more complex operations more effectively.
Studies also suggest that HALS reduces conversion rates and preserves the short-term benefits of LAP, though a nationwide comparison of short-term outcomes using the two techniques has been absent.
To fill this knowledge gap, the investigators used the 2012 colectomy-targeted American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP
At baseline, the HALS group was significantly older than the LAP group (61.5 years vs. 60 years), had a higher body mass index (28.6 kg/m2 vs. 28 kg/m2), was more likely to be hypertensive (49.8% vs. 45.1%), and to be American Society of Anesthesiologists class III (42% vs. 37.4%) and class IV (2.8% vs. 2.7%). Steroid use for inflammatory bowel disease was significantly higher in the LAP group (6.5% vs. 5.4%).
Mortality rates were similar in the LAP and HALS groups (0.52% vs. 0.50%; P = .94), Dr. Gorgun reported.
Multivariate logistic regression adjusted for comorbid conditions lessened the degree of differences between the two groups, but continued to favor HALS for shorter operative time (odds ratio, 0.94) and favor LAP for shorter hospital stays (OR, 1.05) and less superficial surgical site infections (OR, 1.31), less prolonged post-operative ileus (OR, 1.25), and overall morbidity (OR, 1.16).
“Both LAP and HALS approaches are used in minimally invasive colorectal surgery based on their respective contributions and complement each other,” Dr. Gorgun concluded. “Implementing the best approach to decrease postoperative complication rates and increase use of MIS [minimally invasive surgery] will play a role in improving patient care and overall quality of health care.”
Dr. Gorgun, his coauthors, and Dr. Gruber reported having no financial conflicts
CHICAGO – A nationwide analysis comparing straight versus hand-assisted laparoscopic colectomy appears to have resulted in a draw.
Despite being used more commonly in patients with higher body mass index and comorbidities, hand-assisted laparoscopic surgery (HALS) resulted in a significantly shorter operative time than did straight laparoscopic surgery (LAP) (171 minutes vs. 178.8 minutes; P < .001).
On the other hand, LAP was associated with a significantly shorter length of stay (5.9 days vs. 6.0 days; P < .001), fewer complications (15.9% vs. 18.2%; P = .006), fewer specifically superficial skin infections (4.1% vs. 5.4%; P = .007), and less prolonged postoperative ileus (7.5% vs. 9.1%; P = .01).
The differences were statistically significant likely because of the high volume of patients in each group, but are unlikely to be clinically significant in terms of patient care, senior study author Dr. I. Emre Gorgun said at the annual meeting of the Central Surgical Association.
CSA incoming president Dr. Scott A. Gruber, chief of staff at the John D. Dingell VA Medical Center in Detroit, agreed. “There were very, very small differences. I think they were trying to show that the two [approaches] are virtually equivalent,” he said in an interview. “Each has its advantages and disadvantages, but the HALS technique was used in more complex, more high-risk patients,” Dr. Gruber said.
HALS bridges the learning gap between open and straight laparoscopic surgery, which has been slow to gain adoption in colorectal surgery because it is technically demanding and has a steep learning curve, said Dr. Gorgun, a colorectal surgeon at the Cleveland Clinic. HALS allows surgeons to regain tactile sensation and manual retraction, enabling them to perform even more complex operations more effectively.
Studies also suggest that HALS reduces conversion rates and preserves the short-term benefits of LAP, though a nationwide comparison of short-term outcomes using the two techniques has been absent.
To fill this knowledge gap, the investigators used the 2012 colectomy-targeted American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP
At baseline, the HALS group was significantly older than the LAP group (61.5 years vs. 60 years), had a higher body mass index (28.6 kg/m2 vs. 28 kg/m2), was more likely to be hypertensive (49.8% vs. 45.1%), and to be American Society of Anesthesiologists class III (42% vs. 37.4%) and class IV (2.8% vs. 2.7%). Steroid use for inflammatory bowel disease was significantly higher in the LAP group (6.5% vs. 5.4%).
Mortality rates were similar in the LAP and HALS groups (0.52% vs. 0.50%; P = .94), Dr. Gorgun reported.
Multivariate logistic regression adjusted for comorbid conditions lessened the degree of differences between the two groups, but continued to favor HALS for shorter operative time (odds ratio, 0.94) and favor LAP for shorter hospital stays (OR, 1.05) and less superficial surgical site infections (OR, 1.31), less prolonged post-operative ileus (OR, 1.25), and overall morbidity (OR, 1.16).
“Both LAP and HALS approaches are used in minimally invasive colorectal surgery based on their respective contributions and complement each other,” Dr. Gorgun concluded. “Implementing the best approach to decrease postoperative complication rates and increase use of MIS [minimally invasive surgery] will play a role in improving patient care and overall quality of health care.”
Dr. Gorgun, his coauthors, and Dr. Gruber reported having no financial conflicts
CHICAGO – A nationwide analysis comparing straight versus hand-assisted laparoscopic colectomy appears to have resulted in a draw.
Despite being used more commonly in patients with higher body mass index and comorbidities, hand-assisted laparoscopic surgery (HALS) resulted in a significantly shorter operative time than did straight laparoscopic surgery (LAP) (171 minutes vs. 178.8 minutes; P < .001).
On the other hand, LAP was associated with a significantly shorter length of stay (5.9 days vs. 6.0 days; P < .001), fewer complications (15.9% vs. 18.2%; P = .006), fewer specifically superficial skin infections (4.1% vs. 5.4%; P = .007), and less prolonged postoperative ileus (7.5% vs. 9.1%; P = .01).
The differences were statistically significant likely because of the high volume of patients in each group, but are unlikely to be clinically significant in terms of patient care, senior study author Dr. I. Emre Gorgun said at the annual meeting of the Central Surgical Association.
CSA incoming president Dr. Scott A. Gruber, chief of staff at the John D. Dingell VA Medical Center in Detroit, agreed. “There were very, very small differences. I think they were trying to show that the two [approaches] are virtually equivalent,” he said in an interview. “Each has its advantages and disadvantages, but the HALS technique was used in more complex, more high-risk patients,” Dr. Gruber said.
HALS bridges the learning gap between open and straight laparoscopic surgery, which has been slow to gain adoption in colorectal surgery because it is technically demanding and has a steep learning curve, said Dr. Gorgun, a colorectal surgeon at the Cleveland Clinic. HALS allows surgeons to regain tactile sensation and manual retraction, enabling them to perform even more complex operations more effectively.
Studies also suggest that HALS reduces conversion rates and preserves the short-term benefits of LAP, though a nationwide comparison of short-term outcomes using the two techniques has been absent.
To fill this knowledge gap, the investigators used the 2012 colectomy-targeted American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP
At baseline, the HALS group was significantly older than the LAP group (61.5 years vs. 60 years), had a higher body mass index (28.6 kg/m2 vs. 28 kg/m2), was more likely to be hypertensive (49.8% vs. 45.1%), and to be American Society of Anesthesiologists class III (42% vs. 37.4%) and class IV (2.8% vs. 2.7%). Steroid use for inflammatory bowel disease was significantly higher in the LAP group (6.5% vs. 5.4%).
Mortality rates were similar in the LAP and HALS groups (0.52% vs. 0.50%; P = .94), Dr. Gorgun reported.
Multivariate logistic regression adjusted for comorbid conditions lessened the degree of differences between the two groups, but continued to favor HALS for shorter operative time (odds ratio, 0.94) and favor LAP for shorter hospital stays (OR, 1.05) and less superficial surgical site infections (OR, 1.31), less prolonged post-operative ileus (OR, 1.25), and overall morbidity (OR, 1.16).
“Both LAP and HALS approaches are used in minimally invasive colorectal surgery based on their respective contributions and complement each other,” Dr. Gorgun concluded. “Implementing the best approach to decrease postoperative complication rates and increase use of MIS [minimally invasive surgery] will play a role in improving patient care and overall quality of health care.”
Dr. Gorgun, his coauthors, and Dr. Gruber reported having no financial conflicts
AT THE ANNUAL MEETING OF THE CENTRAL SURGICAL ASSOCIATION
Key clinical point: Straight and hand-assisted laparoscopic colectomy offer unique benefits, and HALS is a good option in more complex patients.
Major finding: Surgery was 7 minutes shorter with HALS, while complications were down 2.3% with LAP.
Data source: Retrospective study of 7,843 patients undergoing colectomy in the ACS NSQIP database.
Disclosures: Dr. Gorgun, his coauthors, and Dr. Gruber reported having no financial conflicts.
Colonoscopy Bowel Preparation Instructions
According to the CDC, colorectal cancer (CRC) is largely preventable but remains the second leading cancer killer for men and women in the U.S. Screening for polyps (detection of abnormal growths) and surveillance (based on prior bowel preparation quality, findings, and personal and family histories) are key elements for CRC prevention and survival.1 However, inadequate bowel preparation greatly reduces accuracy of its intended purpose: finding and removing precancerous polyps or lesions before they develop into a cancer, typically within a 10-year window. If preparation quality is not satisfactory, the ability of the endoscopist to meet national polyp detection rates is limited. These rates are currently 25% for men and 15% for women.2 Compounding poor preparation, many veterans avoid CRC screening due to anxiety, shame, and fear of what could be found.
Related: Do I Need a Colonoscopy?
About 60% of veterans presenting for colonoscopy have inadequate bowel preparation.3 Colonoscopy remains the gold standard for detection of colorectal pathology and is available to veterans without insurance preauthorization, eliminating a significant barrier to screening.1 Inadequate bowel preparation can result in missed polyps, cancelled procedures, and increased procedure time. Nonadherence to the liquid diet and high-volume, bowel-cleansing solution can lead to a repeated colonoscopy.
Two nurse practitioners (NPs) at the Philadelphia VAMC (PVAMC) gastroenterology (GE) section recognized that many veterans had poor bowel preparation in spite of preprocedure visits, written instructions, and no financial limitations. Repeated colonoscopies were impacting patient satisfaction, facility costs, and endoscopy staff morale. The NPs developed a study to examine bowel preparation outcomes after a group preprocedure class that provided comprehensive and multimedia education in comparison to standard mailed instructions. The study was approved by the Institutional Review Board. The hypothesis was that group patient education would result in better adherence to bowel preparation instructions than did mailed instructions and that better adherence would result in significantly improved colonoscopy outcomes.
Methods
This was a descriptive pilot study with a convenience sample of 200 veterans randomly selected between 2009 and 2011. The study measured 2 groups. The control group received only the mailed standard bowel preparation instructions, whereas the intervention group received the standard bowel preparation instructions and participated in a group intervention class. Eligible participants were aged 45 to 79 years and were enrolled as patients in a single center (PVAMC GE clinic).
Related: E-Consults in Gastroenterology: A Quality Improvement Project
After referral consults were initially selected for appropriate colonoscopy screening or surveillance, potential patient subjects were randomized into either the control or intervention groups by the coin toss method, followed by mailed letters inviting them to participate in the study. If subjects expressed interest, then consent was obtained. The colonoscopy procedure note was updated to reflect bowel preparation quality. All subjects were de-identified. There were about 8 endoscopists; all were board-certified gastroenterologists plus GE fellows who performed procedures at the time of the study. (Fellows rotated every 2 to 4 weeks in the GE clinic and were always accompanied by an attending gastroenterologist.)
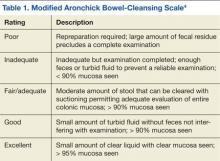
All the endoscopists were instructed in the grading system adapted from the modified Aronchick scale (Table 1).4 This scale measures the quality of bowel preparation for the entire colon: excellent (> 95% visualization of bowel mucosa); good (> 90% of mucosa was visible); fair (some semisolid stool could be suctioned out, but > 90% of mucosa was visible), and poor (semisolid stool cannot be suctioned out and < 90% of mucosa was seen). The modified Aronchick scale also has an inadequate rating, but this was not used in the study. For this study, bowel preparation that was excellent or good received a 1, a fair preparation received a 2, and poor preparation received a 3. The Pearson correlation for the modified Aronchick scale coefficients was 0.62 (P < .001). The value for the kappa statistic was 0.77 (P < .001).5
Results
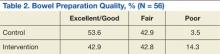
There were 77 men and 5 women enrolled in the study. The control group had 43 subjects, and the intervention group had 39 subjects. Only 28 subjects each from the control and intervention groups had the quality of bowel preparation rated by the endoscopists. In the control group, 53.6% were rated excellent or good, 42.9% were fair, and 3.5% were poor. In the intervention group, 42.9 % of preparations were excellent or good, 42.8% were fair, and 14.3% were poor (Table 2).
Preparation quality was not described in the procedure documentation for 34.9% and 28.2% of the subjects, respectively, for the control and intervention groups. There was no significant difference in no-show rates to procedures in either of the groups. Based on the data, a Fisher exact test for association was performed (P = .39), indicating there was no evidence of association between the intervention group and preparation quality.
The authors observed several recurrent themes during the group interventions. Fear of finding cancer and the perception of the procedure’s invasiveness were raised and addressed. Also misconceptions about CRC were debunked, such as the belief that a lack of bowel symptoms indicated no chance of having CRC or that only a family history indicated a risk factor. Patients discussed how much they learned about CRC, colon anatomy, and the importance of the bowel preparation. A multifaceted teaching approach was used to convey teaching points, such as flip charts, colonoscopy equipment, tours of procedure rooms, and visuals of various bowel preparation qualities. Throughout the educational intervention, humor, active listening, and reflection were woven into discussions to create a comfortable and relaxed learning environment.
Discussion and Limitations
The study results were unexpected. The authors had hypothesized that the group preprocedure educational intervention would have made a statistically significant difference in preparation quality, but it did not. In addition, the authors’ observations during the intervention led them to believe that the subjects had gained knowledge about how to correctly administer the bowel preparation.
Related: Do Age and Gender Matter in Colorectal Cancer?
A significant limitation of this pilot study was the difficulty in extrapolating meaningful data within the intervention group and between the intervention and control groups. After closely examining the raw data, the authors identified some key issues: There were only 28 subjects in each group who had bowel preparation quality described. This small sample size makes it difficult to draw meaningful conclusions. However, the education session in and of itself was clearly a positive experience for subjects, and the authors would recommend a future study with a larger sample size.
A prior power analysis would have helped this study determine a sufficient number of subjects that would be needed to determine whether the intervention had an effect. Furthermore, instead of tossing a coin to randomize the study groups, other types of randomization could have been used.
Other study limitations that came to light were:
- Variable preparation quality documentation by endoscopists;
- Limited availability of days to schedule group intervention classes;
- Some subjects did not attend the group session but still had the procedure done;
- The study invitation letter was long, and there were no financial incentives to participate;
- If pre- and postintervention testing had been conducted, effective and ineffective teaching strategies could have been identified; and
- The principal investigator also performed some of the procedures during the study, introducing potential bias.
Since the study, the authors have learned more about changes in national standards for bowel preparation administration and polyp surveillance. Preparation instructions need to be updated to reflect current recommendations for split-dose preparation administration in which the bowel preparation is taken in spaced doses, leading to better compliance and outcomes.6 Informally, patients and family have told staff that preparation instructions are difficult to understand. Following a Plan-Do-Study-Act cycle, feedback from patients should be obtained before revising and printing preparation instructions.7 This feedback could ensure that preparation instructions are written in patient-friendly, easily understood language.
Conclusion
Nursing professionals are likely to be effective in helping veterans achieve improved bowel preparation quality, because nurses have an established record as patient educators and advocates. Good bowel preparation quality is an important, achievable objective for veterans. As Mangnall reported, bowel preparation quality data are a strong nurse-sensitive quality measure that can be used to devise more effective interventions to obtain better bowel preparation results.8 As clinicians working on the frontline, nurses are well positioned to assess, intervene, and evaluate whether or not the modifications they have made to bowel preparation instructions are effective as they measure bowel preparation quality status post colonoscopy.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. American Society of Gastrointestinal Endoscopy. Media backgrounder: Colorectal cancer screening. American Society of Gastrointestinal Endoscopy Website. http://www.asge.org/PressroomIndex .aspx?id=552. Updated August 2014. Accessed January 29, 2015.
2. Lee RH. Quality colonoscopy: A matter of time, technique or technology? World J Gastroenterol. 2013;19(10):1517-1522.
3. Health Services Research & Development. 1004—Impact of a novel patient educational booklet on colonoscopy preparation in veterans. US Department of Veterans Affairs Website. http://www.hsrd .research.va.gov/meetings/2009/print_abstract .cfm?recordid=541. Accessed January 29, 2015.
4. Gurudu SR, Ratuapli S, Heigh R, DiBaise J, Leighton J, Crowell M. Quality of bowel cleansing for afternoon colonoscopy is influenced by time of administration. Am J Gastroenterol. 2010;105(11):2318-2322.
5. Rostom A, Jolicoeur E. Validation of a new scale for the assessment of bowel preparation quality. Gastrointest Endosc. 2004;59(4):482-486.
6. Cohen, LB. Split dosing of bowel preparation for colonoscopy: An analysis of its efficacy, safety and tolerability. Gastrointest Endosc. 2010;72(2):406-412.
7. Institute for Healthcare Improvement. How to improve: Model for improvement. Institute for Healthcare improvement; 2015. Institute for Healthcare Improvement Website. http://www.ihi.org/resources/Pages/HowtoImprove/default.aspx. Accessed February 5, 2015.
8. Mangnall R. Good bowel cleaning vital for effective colonoscopy. Nurse N Z. 2012;18(16):17-19.
According to the CDC, colorectal cancer (CRC) is largely preventable but remains the second leading cancer killer for men and women in the U.S. Screening for polyps (detection of abnormal growths) and surveillance (based on prior bowel preparation quality, findings, and personal and family histories) are key elements for CRC prevention and survival.1 However, inadequate bowel preparation greatly reduces accuracy of its intended purpose: finding and removing precancerous polyps or lesions before they develop into a cancer, typically within a 10-year window. If preparation quality is not satisfactory, the ability of the endoscopist to meet national polyp detection rates is limited. These rates are currently 25% for men and 15% for women.2 Compounding poor preparation, many veterans avoid CRC screening due to anxiety, shame, and fear of what could be found.
Related: Do I Need a Colonoscopy?
About 60% of veterans presenting for colonoscopy have inadequate bowel preparation.3 Colonoscopy remains the gold standard for detection of colorectal pathology and is available to veterans without insurance preauthorization, eliminating a significant barrier to screening.1 Inadequate bowel preparation can result in missed polyps, cancelled procedures, and increased procedure time. Nonadherence to the liquid diet and high-volume, bowel-cleansing solution can lead to a repeated colonoscopy.
Two nurse practitioners (NPs) at the Philadelphia VAMC (PVAMC) gastroenterology (GE) section recognized that many veterans had poor bowel preparation in spite of preprocedure visits, written instructions, and no financial limitations. Repeated colonoscopies were impacting patient satisfaction, facility costs, and endoscopy staff morale. The NPs developed a study to examine bowel preparation outcomes after a group preprocedure class that provided comprehensive and multimedia education in comparison to standard mailed instructions. The study was approved by the Institutional Review Board. The hypothesis was that group patient education would result in better adherence to bowel preparation instructions than did mailed instructions and that better adherence would result in significantly improved colonoscopy outcomes.
Methods
This was a descriptive pilot study with a convenience sample of 200 veterans randomly selected between 2009 and 2011. The study measured 2 groups. The control group received only the mailed standard bowel preparation instructions, whereas the intervention group received the standard bowel preparation instructions and participated in a group intervention class. Eligible participants were aged 45 to 79 years and were enrolled as patients in a single center (PVAMC GE clinic).
Related: E-Consults in Gastroenterology: A Quality Improvement Project
After referral consults were initially selected for appropriate colonoscopy screening or surveillance, potential patient subjects were randomized into either the control or intervention groups by the coin toss method, followed by mailed letters inviting them to participate in the study. If subjects expressed interest, then consent was obtained. The colonoscopy procedure note was updated to reflect bowel preparation quality. All subjects were de-identified. There were about 8 endoscopists; all were board-certified gastroenterologists plus GE fellows who performed procedures at the time of the study. (Fellows rotated every 2 to 4 weeks in the GE clinic and were always accompanied by an attending gastroenterologist.)
All the endoscopists were instructed in the grading system adapted from the modified Aronchick scale (Table 1).4 This scale measures the quality of bowel preparation for the entire colon: excellent (> 95% visualization of bowel mucosa); good (> 90% of mucosa was visible); fair (some semisolid stool could be suctioned out, but > 90% of mucosa was visible), and poor (semisolid stool cannot be suctioned out and < 90% of mucosa was seen). The modified Aronchick scale also has an inadequate rating, but this was not used in the study. For this study, bowel preparation that was excellent or good received a 1, a fair preparation received a 2, and poor preparation received a 3. The Pearson correlation for the modified Aronchick scale coefficients was 0.62 (P < .001). The value for the kappa statistic was 0.77 (P < .001).5
Results
There were 77 men and 5 women enrolled in the study. The control group had 43 subjects, and the intervention group had 39 subjects. Only 28 subjects each from the control and intervention groups had the quality of bowel preparation rated by the endoscopists. In the control group, 53.6% were rated excellent or good, 42.9% were fair, and 3.5% were poor. In the intervention group, 42.9 % of preparations were excellent or good, 42.8% were fair, and 14.3% were poor (Table 2).
Preparation quality was not described in the procedure documentation for 34.9% and 28.2% of the subjects, respectively, for the control and intervention groups. There was no significant difference in no-show rates to procedures in either of the groups. Based on the data, a Fisher exact test for association was performed (P = .39), indicating there was no evidence of association between the intervention group and preparation quality.
The authors observed several recurrent themes during the group interventions. Fear of finding cancer and the perception of the procedure’s invasiveness were raised and addressed. Also misconceptions about CRC were debunked, such as the belief that a lack of bowel symptoms indicated no chance of having CRC or that only a family history indicated a risk factor. Patients discussed how much they learned about CRC, colon anatomy, and the importance of the bowel preparation. A multifaceted teaching approach was used to convey teaching points, such as flip charts, colonoscopy equipment, tours of procedure rooms, and visuals of various bowel preparation qualities. Throughout the educational intervention, humor, active listening, and reflection were woven into discussions to create a comfortable and relaxed learning environment.
Discussion and Limitations
The study results were unexpected. The authors had hypothesized that the group preprocedure educational intervention would have made a statistically significant difference in preparation quality, but it did not. In addition, the authors’ observations during the intervention led them to believe that the subjects had gained knowledge about how to correctly administer the bowel preparation.
Related: Do Age and Gender Matter in Colorectal Cancer?
A significant limitation of this pilot study was the difficulty in extrapolating meaningful data within the intervention group and between the intervention and control groups. After closely examining the raw data, the authors identified some key issues: There were only 28 subjects in each group who had bowel preparation quality described. This small sample size makes it difficult to draw meaningful conclusions. However, the education session in and of itself was clearly a positive experience for subjects, and the authors would recommend a future study with a larger sample size.
A prior power analysis would have helped this study determine a sufficient number of subjects that would be needed to determine whether the intervention had an effect. Furthermore, instead of tossing a coin to randomize the study groups, other types of randomization could have been used.
Other study limitations that came to light were:
- Variable preparation quality documentation by endoscopists;
- Limited availability of days to schedule group intervention classes;
- Some subjects did not attend the group session but still had the procedure done;
- The study invitation letter was long, and there were no financial incentives to participate;
- If pre- and postintervention testing had been conducted, effective and ineffective teaching strategies could have been identified; and
- The principal investigator also performed some of the procedures during the study, introducing potential bias.
Since the study, the authors have learned more about changes in national standards for bowel preparation administration and polyp surveillance. Preparation instructions need to be updated to reflect current recommendations for split-dose preparation administration in which the bowel preparation is taken in spaced doses, leading to better compliance and outcomes.6 Informally, patients and family have told staff that preparation instructions are difficult to understand. Following a Plan-Do-Study-Act cycle, feedback from patients should be obtained before revising and printing preparation instructions.7 This feedback could ensure that preparation instructions are written in patient-friendly, easily understood language.
Conclusion
Nursing professionals are likely to be effective in helping veterans achieve improved bowel preparation quality, because nurses have an established record as patient educators and advocates. Good bowel preparation quality is an important, achievable objective for veterans. As Mangnall reported, bowel preparation quality data are a strong nurse-sensitive quality measure that can be used to devise more effective interventions to obtain better bowel preparation results.8 As clinicians working on the frontline, nurses are well positioned to assess, intervene, and evaluate whether or not the modifications they have made to bowel preparation instructions are effective as they measure bowel preparation quality status post colonoscopy.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
According to the CDC, colorectal cancer (CRC) is largely preventable but remains the second leading cancer killer for men and women in the U.S. Screening for polyps (detection of abnormal growths) and surveillance (based on prior bowel preparation quality, findings, and personal and family histories) are key elements for CRC prevention and survival.1 However, inadequate bowel preparation greatly reduces accuracy of its intended purpose: finding and removing precancerous polyps or lesions before they develop into a cancer, typically within a 10-year window. If preparation quality is not satisfactory, the ability of the endoscopist to meet national polyp detection rates is limited. These rates are currently 25% for men and 15% for women.2 Compounding poor preparation, many veterans avoid CRC screening due to anxiety, shame, and fear of what could be found.
Related: Do I Need a Colonoscopy?
About 60% of veterans presenting for colonoscopy have inadequate bowel preparation.3 Colonoscopy remains the gold standard for detection of colorectal pathology and is available to veterans without insurance preauthorization, eliminating a significant barrier to screening.1 Inadequate bowel preparation can result in missed polyps, cancelled procedures, and increased procedure time. Nonadherence to the liquid diet and high-volume, bowel-cleansing solution can lead to a repeated colonoscopy.
Two nurse practitioners (NPs) at the Philadelphia VAMC (PVAMC) gastroenterology (GE) section recognized that many veterans had poor bowel preparation in spite of preprocedure visits, written instructions, and no financial limitations. Repeated colonoscopies were impacting patient satisfaction, facility costs, and endoscopy staff morale. The NPs developed a study to examine bowel preparation outcomes after a group preprocedure class that provided comprehensive and multimedia education in comparison to standard mailed instructions. The study was approved by the Institutional Review Board. The hypothesis was that group patient education would result in better adherence to bowel preparation instructions than did mailed instructions and that better adherence would result in significantly improved colonoscopy outcomes.
Methods
This was a descriptive pilot study with a convenience sample of 200 veterans randomly selected between 2009 and 2011. The study measured 2 groups. The control group received only the mailed standard bowel preparation instructions, whereas the intervention group received the standard bowel preparation instructions and participated in a group intervention class. Eligible participants were aged 45 to 79 years and were enrolled as patients in a single center (PVAMC GE clinic).
Related: E-Consults in Gastroenterology: A Quality Improvement Project
After referral consults were initially selected for appropriate colonoscopy screening or surveillance, potential patient subjects were randomized into either the control or intervention groups by the coin toss method, followed by mailed letters inviting them to participate in the study. If subjects expressed interest, then consent was obtained. The colonoscopy procedure note was updated to reflect bowel preparation quality. All subjects were de-identified. There were about 8 endoscopists; all were board-certified gastroenterologists plus GE fellows who performed procedures at the time of the study. (Fellows rotated every 2 to 4 weeks in the GE clinic and were always accompanied by an attending gastroenterologist.)
All the endoscopists were instructed in the grading system adapted from the modified Aronchick scale (Table 1).4 This scale measures the quality of bowel preparation for the entire colon: excellent (> 95% visualization of bowel mucosa); good (> 90% of mucosa was visible); fair (some semisolid stool could be suctioned out, but > 90% of mucosa was visible), and poor (semisolid stool cannot be suctioned out and < 90% of mucosa was seen). The modified Aronchick scale also has an inadequate rating, but this was not used in the study. For this study, bowel preparation that was excellent or good received a 1, a fair preparation received a 2, and poor preparation received a 3. The Pearson correlation for the modified Aronchick scale coefficients was 0.62 (P < .001). The value for the kappa statistic was 0.77 (P < .001).5
Results
There were 77 men and 5 women enrolled in the study. The control group had 43 subjects, and the intervention group had 39 subjects. Only 28 subjects each from the control and intervention groups had the quality of bowel preparation rated by the endoscopists. In the control group, 53.6% were rated excellent or good, 42.9% were fair, and 3.5% were poor. In the intervention group, 42.9 % of preparations were excellent or good, 42.8% were fair, and 14.3% were poor (Table 2).
Preparation quality was not described in the procedure documentation for 34.9% and 28.2% of the subjects, respectively, for the control and intervention groups. There was no significant difference in no-show rates to procedures in either of the groups. Based on the data, a Fisher exact test for association was performed (P = .39), indicating there was no evidence of association between the intervention group and preparation quality.
The authors observed several recurrent themes during the group interventions. Fear of finding cancer and the perception of the procedure’s invasiveness were raised and addressed. Also misconceptions about CRC were debunked, such as the belief that a lack of bowel symptoms indicated no chance of having CRC or that only a family history indicated a risk factor. Patients discussed how much they learned about CRC, colon anatomy, and the importance of the bowel preparation. A multifaceted teaching approach was used to convey teaching points, such as flip charts, colonoscopy equipment, tours of procedure rooms, and visuals of various bowel preparation qualities. Throughout the educational intervention, humor, active listening, and reflection were woven into discussions to create a comfortable and relaxed learning environment.
Discussion and Limitations
The study results were unexpected. The authors had hypothesized that the group preprocedure educational intervention would have made a statistically significant difference in preparation quality, but it did not. In addition, the authors’ observations during the intervention led them to believe that the subjects had gained knowledge about how to correctly administer the bowel preparation.
Related: Do Age and Gender Matter in Colorectal Cancer?
A significant limitation of this pilot study was the difficulty in extrapolating meaningful data within the intervention group and between the intervention and control groups. After closely examining the raw data, the authors identified some key issues: There were only 28 subjects in each group who had bowel preparation quality described. This small sample size makes it difficult to draw meaningful conclusions. However, the education session in and of itself was clearly a positive experience for subjects, and the authors would recommend a future study with a larger sample size.
A prior power analysis would have helped this study determine a sufficient number of subjects that would be needed to determine whether the intervention had an effect. Furthermore, instead of tossing a coin to randomize the study groups, other types of randomization could have been used.
Other study limitations that came to light were:
- Variable preparation quality documentation by endoscopists;
- Limited availability of days to schedule group intervention classes;
- Some subjects did not attend the group session but still had the procedure done;
- The study invitation letter was long, and there were no financial incentives to participate;
- If pre- and postintervention testing had been conducted, effective and ineffective teaching strategies could have been identified; and
- The principal investigator also performed some of the procedures during the study, introducing potential bias.
Since the study, the authors have learned more about changes in national standards for bowel preparation administration and polyp surveillance. Preparation instructions need to be updated to reflect current recommendations for split-dose preparation administration in which the bowel preparation is taken in spaced doses, leading to better compliance and outcomes.6 Informally, patients and family have told staff that preparation instructions are difficult to understand. Following a Plan-Do-Study-Act cycle, feedback from patients should be obtained before revising and printing preparation instructions.7 This feedback could ensure that preparation instructions are written in patient-friendly, easily understood language.
Conclusion
Nursing professionals are likely to be effective in helping veterans achieve improved bowel preparation quality, because nurses have an established record as patient educators and advocates. Good bowel preparation quality is an important, achievable objective for veterans. As Mangnall reported, bowel preparation quality data are a strong nurse-sensitive quality measure that can be used to devise more effective interventions to obtain better bowel preparation results.8 As clinicians working on the frontline, nurses are well positioned to assess, intervene, and evaluate whether or not the modifications they have made to bowel preparation instructions are effective as they measure bowel preparation quality status post colonoscopy.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. American Society of Gastrointestinal Endoscopy. Media backgrounder: Colorectal cancer screening. American Society of Gastrointestinal Endoscopy Website. http://www.asge.org/PressroomIndex .aspx?id=552. Updated August 2014. Accessed January 29, 2015.
2. Lee RH. Quality colonoscopy: A matter of time, technique or technology? World J Gastroenterol. 2013;19(10):1517-1522.
3. Health Services Research & Development. 1004—Impact of a novel patient educational booklet on colonoscopy preparation in veterans. US Department of Veterans Affairs Website. http://www.hsrd .research.va.gov/meetings/2009/print_abstract .cfm?recordid=541. Accessed January 29, 2015.
4. Gurudu SR, Ratuapli S, Heigh R, DiBaise J, Leighton J, Crowell M. Quality of bowel cleansing for afternoon colonoscopy is influenced by time of administration. Am J Gastroenterol. 2010;105(11):2318-2322.
5. Rostom A, Jolicoeur E. Validation of a new scale for the assessment of bowel preparation quality. Gastrointest Endosc. 2004;59(4):482-486.
6. Cohen, LB. Split dosing of bowel preparation for colonoscopy: An analysis of its efficacy, safety and tolerability. Gastrointest Endosc. 2010;72(2):406-412.
7. Institute for Healthcare Improvement. How to improve: Model for improvement. Institute for Healthcare improvement; 2015. Institute for Healthcare Improvement Website. http://www.ihi.org/resources/Pages/HowtoImprove/default.aspx. Accessed February 5, 2015.
8. Mangnall R. Good bowel cleaning vital for effective colonoscopy. Nurse N Z. 2012;18(16):17-19.
1. American Society of Gastrointestinal Endoscopy. Media backgrounder: Colorectal cancer screening. American Society of Gastrointestinal Endoscopy Website. http://www.asge.org/PressroomIndex .aspx?id=552. Updated August 2014. Accessed January 29, 2015.
2. Lee RH. Quality colonoscopy: A matter of time, technique or technology? World J Gastroenterol. 2013;19(10):1517-1522.
3. Health Services Research & Development. 1004—Impact of a novel patient educational booklet on colonoscopy preparation in veterans. US Department of Veterans Affairs Website. http://www.hsrd .research.va.gov/meetings/2009/print_abstract .cfm?recordid=541. Accessed January 29, 2015.
4. Gurudu SR, Ratuapli S, Heigh R, DiBaise J, Leighton J, Crowell M. Quality of bowel cleansing for afternoon colonoscopy is influenced by time of administration. Am J Gastroenterol. 2010;105(11):2318-2322.
5. Rostom A, Jolicoeur E. Validation of a new scale for the assessment of bowel preparation quality. Gastrointest Endosc. 2004;59(4):482-486.
6. Cohen, LB. Split dosing of bowel preparation for colonoscopy: An analysis of its efficacy, safety and tolerability. Gastrointest Endosc. 2010;72(2):406-412.
7. Institute for Healthcare Improvement. How to improve: Model for improvement. Institute for Healthcare improvement; 2015. Institute for Healthcare Improvement Website. http://www.ihi.org/resources/Pages/HowtoImprove/default.aspx. Accessed February 5, 2015.
8. Mangnall R. Good bowel cleaning vital for effective colonoscopy. Nurse N Z. 2012;18(16):17-19.
Mesh overlay reduces hernia after colorectal surgeries
The use of a large-pore polypropylene mesh in colorectal surgery can reduce incidence of incisional hernias without contributing to wound complication risk, according to results from a randomized controlled trial.
While prophylactic polypropylene meshes have been used successfully in other types of surgeries to prevent hernias, they have been little studied in series of patients undergoing colorectal surgeries, a group for which incidence of IH is high, and particularly so with emergency procedures.
For their research, published ahead of print in Annals of Surgery (Ann. Surg.2015 Jan 8 [doi: 10.1097/SLA.0000000000001116]), Dr. Miguel Ángel García-Ureña of Henares University Hospital in Madrid, and his colleagues, recruited 107 patients with elective or emergency colorectal surgeries using a midline laparotomy approach.
Patients were randomized to either standard care (n = 54, 20 emergency) or the addition of an overlay large-pore polypropylene mesh after the closure of the abdominal wall (n = 53, 17 emergency). All operations took place at the same hospital, with 12 surgeons participating.
At 24 months’ follow-up, the control group saw 17 incisional hernias (31.5%), compared with 6 (11.3%) in the study group (P = .011). No statistically significant differences were seen for incidence of surgical site infection, seroma, evisceration, or systemic complications, and no mesh rejection was seen.
Dr. García-Ureña and colleagues used a very low-weight, large-pore polypropylene mesh after initial studies suggested large-pore meshes were better tolerated in contaminated fields, and that these could be salvaged even in the case of site infection.
The study “confirms the safe use of large-pore polypropylene meshes even in contaminated and emergency surgical procedures,” the investigators wrote in their analysis, adding that the use of mesh overlay “was cost-effective due to the number needed to treat obtained: 1 IH was prevented for every 5 prophylactic meshes that were used.”
Dr. García-Ureña and colleagues cited as limitations of their study the fact that deaths and reoperations occurred in 28% of patients before follow-up ended, the inclusion of both elective and emergency cases, and that wound length was not recorded. Further studies will be needed, they said, to determine the ideal positioning of the mesh and the best type of mesh for these procedures.
The study authors declared no conflicts of interest.
The use of a large-pore polypropylene mesh in colorectal surgery can reduce incidence of incisional hernias without contributing to wound complication risk, according to results from a randomized controlled trial.
While prophylactic polypropylene meshes have been used successfully in other types of surgeries to prevent hernias, they have been little studied in series of patients undergoing colorectal surgeries, a group for which incidence of IH is high, and particularly so with emergency procedures.
For their research, published ahead of print in Annals of Surgery (Ann. Surg.2015 Jan 8 [doi: 10.1097/SLA.0000000000001116]), Dr. Miguel Ángel García-Ureña of Henares University Hospital in Madrid, and his colleagues, recruited 107 patients with elective or emergency colorectal surgeries using a midline laparotomy approach.
Patients were randomized to either standard care (n = 54, 20 emergency) or the addition of an overlay large-pore polypropylene mesh after the closure of the abdominal wall (n = 53, 17 emergency). All operations took place at the same hospital, with 12 surgeons participating.
At 24 months’ follow-up, the control group saw 17 incisional hernias (31.5%), compared with 6 (11.3%) in the study group (P = .011). No statistically significant differences were seen for incidence of surgical site infection, seroma, evisceration, or systemic complications, and no mesh rejection was seen.
Dr. García-Ureña and colleagues used a very low-weight, large-pore polypropylene mesh after initial studies suggested large-pore meshes were better tolerated in contaminated fields, and that these could be salvaged even in the case of site infection.
The study “confirms the safe use of large-pore polypropylene meshes even in contaminated and emergency surgical procedures,” the investigators wrote in their analysis, adding that the use of mesh overlay “was cost-effective due to the number needed to treat obtained: 1 IH was prevented for every 5 prophylactic meshes that were used.”
Dr. García-Ureña and colleagues cited as limitations of their study the fact that deaths and reoperations occurred in 28% of patients before follow-up ended, the inclusion of both elective and emergency cases, and that wound length was not recorded. Further studies will be needed, they said, to determine the ideal positioning of the mesh and the best type of mesh for these procedures.
The study authors declared no conflicts of interest.
The use of a large-pore polypropylene mesh in colorectal surgery can reduce incidence of incisional hernias without contributing to wound complication risk, according to results from a randomized controlled trial.
While prophylactic polypropylene meshes have been used successfully in other types of surgeries to prevent hernias, they have been little studied in series of patients undergoing colorectal surgeries, a group for which incidence of IH is high, and particularly so with emergency procedures.
For their research, published ahead of print in Annals of Surgery (Ann. Surg.2015 Jan 8 [doi: 10.1097/SLA.0000000000001116]), Dr. Miguel Ángel García-Ureña of Henares University Hospital in Madrid, and his colleagues, recruited 107 patients with elective or emergency colorectal surgeries using a midline laparotomy approach.
Patients were randomized to either standard care (n = 54, 20 emergency) or the addition of an overlay large-pore polypropylene mesh after the closure of the abdominal wall (n = 53, 17 emergency). All operations took place at the same hospital, with 12 surgeons participating.
At 24 months’ follow-up, the control group saw 17 incisional hernias (31.5%), compared with 6 (11.3%) in the study group (P = .011). No statistically significant differences were seen for incidence of surgical site infection, seroma, evisceration, or systemic complications, and no mesh rejection was seen.
Dr. García-Ureña and colleagues used a very low-weight, large-pore polypropylene mesh after initial studies suggested large-pore meshes were better tolerated in contaminated fields, and that these could be salvaged even in the case of site infection.
The study “confirms the safe use of large-pore polypropylene meshes even in contaminated and emergency surgical procedures,” the investigators wrote in their analysis, adding that the use of mesh overlay “was cost-effective due to the number needed to treat obtained: 1 IH was prevented for every 5 prophylactic meshes that were used.”
Dr. García-Ureña and colleagues cited as limitations of their study the fact that deaths and reoperations occurred in 28% of patients before follow-up ended, the inclusion of both elective and emergency cases, and that wound length was not recorded. Further studies will be needed, they said, to determine the ideal positioning of the mesh and the best type of mesh for these procedures.
The study authors declared no conflicts of interest.
FROM ANNALS OF SURGERY
Key clinical point: Use of a large-pore polypropylene mesh overlay significantly reduced incidence of incisional hernia after colorectal surgery, without increasing complications.
Major finding: Incidence of IH was 31.5% in the standard-care group and 11.3% group randomized to mesh overlay (P = .011).
Data source: A randomized controlled trial of 107 patients undergoing emergency or elective colorectal surgeries at one surgical center.
Disclosures: The study authors declared no conflicts of interest.
Jury still out on survival benefit of resecting primary in mCRC
SAN FRANCISCO – Resecting the primary tumor in patients with metastatic colon or colorectal cancer may prolong survival. But then again, it may not.
This was the overarching take-home message from a trio of cohort studies presented at the Gastrointestinal Cancers Symposium cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology. Results were reported in a poster session.
“Whereas surgery is the primary treatment of localized colorectal cancer, resection of the primary tumor in patients with incurable metastatic disease is usually recommended for palliative purposes to manage obstruction, perforation, or bleeding,” Dr. Shahid Ahmed, lead investigator of one of the studies, noted in comments provided by e-mail. “The role of surgical resection of the primary tumor in patients with newly diagnosed incurable stage IV colorectal cancer remains controversial.”
In earlier research, he and colleagues found a survival benefit of primary resection among Canadian patients whose cancer was diagnosed between 1992 and 2005 (Cancer 2014;120:683-91). But the majority did not receive systemic therapy, and those who did were often given older regimens.
In a new study aimed at testing the association in the contemporary treatment era, the researchers analyzed data from 569 patients with stage IV colorectal cancer diagnosed between 2006 and 2010 who had a median follow-up of 11 months. Overall, 55% had resection of the primary tumor.
Among the 57% of patients who received systemic therapy, 91% received FOLFIRI or FOLFOX, 65% received bevacizumab (Avastin), and 10% received cetuximab (Erbitux) or panitumumab (Vectibix), according to Dr. Ahmed, professor of medicine, University of Saskatchewan, Canada.
Results for the entire cohort showed that median overall survival was 18 months in patients who had resection of their primary versus 4 months in those who did not (multivariate hazard ratio, 0.44; P less than .001).
Among the subgroup of patients who received chemotherapy, median survival was 27 months with primary resection versus 14 months without it (P less than .0001). And among the subgroup that specifically received FOLFIRI or FOLFOX and a biologic agent, it was 35 months with primary resection and 23 months without it (P less than .001).
“Surgical resection of primary tumor improves survival of patients with stage IV colorectal cancer, independent of other prognostic variables including age, performance status, comorbid illness, and chemotherapy,” maintained Dr. Ahmed. “The current study validates our findings and supports surgical resection of primary tumor in patients with stage IV colorectal cancer who are treated with modern chemotherapy and biologics.
“A well-designed prospective randomized trial is warranted to confirm the survival benefit conferred by the primary tumor resection,” he added, noting that two such trials in Europe – SYNCHRONOUS and CAIRO4 – are underway.
“If the magnitude of survival benefit is confirmed in these future randomized studies, surgical resection of the primary tumor could potentially be a more cost-effective intervention compared with novel systemic therapy in the management of metastatic colorectal cancer,” he concluded.
In a second study, Dr. Aaron Lewis, a surgical oncology fellow at the City of Hope, Duarte, Calif., and colleagues analyzed data from patients with stage IV colon cancer in the Surveillance, Epidemiology, and End Results (SEER) database for the years 1998 through 2011. They excluded those who died within 30 days of diagnosis or had resection of metastases. Overall, 70% of the 28,068 included patients had resection of their primary.
In multivariate analyses, patients who underwent resection had half the risk of death when compared with peers who did not have this surgery (hazard ratio, 0.49), reported Dr. Lewis.
Findings were essentially the same when the analysis was repeated in a subset of matched patients: Median survival was 17 months with resection versus 9 months without it (hazard ratio, 0.48; P less than .0001). Estimated 3-year survival was 23% and 6%, respectively.
“There are limitations, factors that we couldn’t completely control for. For example, there is no chemotherapy data in the SEER database. We didn’t know the timing of surgery in relation to chemotherapy. And we didn’t know whether these patients were asymptomatic or symptomatic,” Dr. Lewis noted in an interview. “But analysis of this huge group of patients in the United States that are getting treated shows that there is a survival benefit.”
Possible reasons why surgery might prolong life in this setting are unknown but may include the effects of tumor debulking or some enhancement of the immune response, he proposed.
To definitively confirm a survival benefit, a randomized controlled trial is needed, he agreed. “This seems to be a popular question in the literature in the last couple of years, so maybe somebody will be willing to take it on.”
In a third study, a team led by Dr. Zeinab Alawadi, a surgeon and postdoctoral fellow at the University of Texas MD Anderson Cancer Center, Houston, analyzed data from 14,399 patients in the National Cancer Data Base. They had been diagnosed with stage IV colon cancer between 2003 and 2005. The researchers excluded patients who had nonelective resection or surgery at other sites, such as metastasectomy.
The primary tumor was resected in 55% of all patients studied and in 74% of patients included in a 1-year landmark analysis done to account for early deaths related to comorbidity or disease burden, reported Dr. Alawadi.
In the entire cohort, primary resection conferred a significant survival benefit after standard multivariate adjustment (hazard ratio, 0.39) that persisted after propensity score weighting to account for treatment selection bias (hazard ratio, 0.41). The benefit was also significant, but much attenuated, in an instrumental variable analysis, another method for accounting for treatment selection bias (relative mortality rate, 0.88).
In the 1-year landmark population, primary resection conferred a smaller significant survival benefit after standard multivariate adjustment (hazard ratio, 0.60) that persisted after propensity score weighting (hazard ratio, 0.59). But there was no longer a significant benefit in the instrumental variable analysis here.
“Among the entire cohort of patients with stage 4 colon cancer, primary tumor resection offered no survival benefit over systemic chemotherapy alone when the [instrumental variable] method was applied at the 1 year landmark,” the investigators write.
“Subject to selection and survivor treatment bias, standard regression analysis may overestimate the benefit of [primary tumor resection],” they concluded.
SAN FRANCISCO – Resecting the primary tumor in patients with metastatic colon or colorectal cancer may prolong survival. But then again, it may not.
This was the overarching take-home message from a trio of cohort studies presented at the Gastrointestinal Cancers Symposium cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology. Results were reported in a poster session.
“Whereas surgery is the primary treatment of localized colorectal cancer, resection of the primary tumor in patients with incurable metastatic disease is usually recommended for palliative purposes to manage obstruction, perforation, or bleeding,” Dr. Shahid Ahmed, lead investigator of one of the studies, noted in comments provided by e-mail. “The role of surgical resection of the primary tumor in patients with newly diagnosed incurable stage IV colorectal cancer remains controversial.”
In earlier research, he and colleagues found a survival benefit of primary resection among Canadian patients whose cancer was diagnosed between 1992 and 2005 (Cancer 2014;120:683-91). But the majority did not receive systemic therapy, and those who did were often given older regimens.
In a new study aimed at testing the association in the contemporary treatment era, the researchers analyzed data from 569 patients with stage IV colorectal cancer diagnosed between 2006 and 2010 who had a median follow-up of 11 months. Overall, 55% had resection of the primary tumor.
Among the 57% of patients who received systemic therapy, 91% received FOLFIRI or FOLFOX, 65% received bevacizumab (Avastin), and 10% received cetuximab (Erbitux) or panitumumab (Vectibix), according to Dr. Ahmed, professor of medicine, University of Saskatchewan, Canada.
Results for the entire cohort showed that median overall survival was 18 months in patients who had resection of their primary versus 4 months in those who did not (multivariate hazard ratio, 0.44; P less than .001).
Among the subgroup of patients who received chemotherapy, median survival was 27 months with primary resection versus 14 months without it (P less than .0001). And among the subgroup that specifically received FOLFIRI or FOLFOX and a biologic agent, it was 35 months with primary resection and 23 months without it (P less than .001).
“Surgical resection of primary tumor improves survival of patients with stage IV colorectal cancer, independent of other prognostic variables including age, performance status, comorbid illness, and chemotherapy,” maintained Dr. Ahmed. “The current study validates our findings and supports surgical resection of primary tumor in patients with stage IV colorectal cancer who are treated with modern chemotherapy and biologics.
“A well-designed prospective randomized trial is warranted to confirm the survival benefit conferred by the primary tumor resection,” he added, noting that two such trials in Europe – SYNCHRONOUS and CAIRO4 – are underway.
“If the magnitude of survival benefit is confirmed in these future randomized studies, surgical resection of the primary tumor could potentially be a more cost-effective intervention compared with novel systemic therapy in the management of metastatic colorectal cancer,” he concluded.
In a second study, Dr. Aaron Lewis, a surgical oncology fellow at the City of Hope, Duarte, Calif., and colleagues analyzed data from patients with stage IV colon cancer in the Surveillance, Epidemiology, and End Results (SEER) database for the years 1998 through 2011. They excluded those who died within 30 days of diagnosis or had resection of metastases. Overall, 70% of the 28,068 included patients had resection of their primary.
In multivariate analyses, patients who underwent resection had half the risk of death when compared with peers who did not have this surgery (hazard ratio, 0.49), reported Dr. Lewis.
Findings were essentially the same when the analysis was repeated in a subset of matched patients: Median survival was 17 months with resection versus 9 months without it (hazard ratio, 0.48; P less than .0001). Estimated 3-year survival was 23% and 6%, respectively.
“There are limitations, factors that we couldn’t completely control for. For example, there is no chemotherapy data in the SEER database. We didn’t know the timing of surgery in relation to chemotherapy. And we didn’t know whether these patients were asymptomatic or symptomatic,” Dr. Lewis noted in an interview. “But analysis of this huge group of patients in the United States that are getting treated shows that there is a survival benefit.”
Possible reasons why surgery might prolong life in this setting are unknown but may include the effects of tumor debulking or some enhancement of the immune response, he proposed.
To definitively confirm a survival benefit, a randomized controlled trial is needed, he agreed. “This seems to be a popular question in the literature in the last couple of years, so maybe somebody will be willing to take it on.”
In a third study, a team led by Dr. Zeinab Alawadi, a surgeon and postdoctoral fellow at the University of Texas MD Anderson Cancer Center, Houston, analyzed data from 14,399 patients in the National Cancer Data Base. They had been diagnosed with stage IV colon cancer between 2003 and 2005. The researchers excluded patients who had nonelective resection or surgery at other sites, such as metastasectomy.
The primary tumor was resected in 55% of all patients studied and in 74% of patients included in a 1-year landmark analysis done to account for early deaths related to comorbidity or disease burden, reported Dr. Alawadi.
In the entire cohort, primary resection conferred a significant survival benefit after standard multivariate adjustment (hazard ratio, 0.39) that persisted after propensity score weighting to account for treatment selection bias (hazard ratio, 0.41). The benefit was also significant, but much attenuated, in an instrumental variable analysis, another method for accounting for treatment selection bias (relative mortality rate, 0.88).
In the 1-year landmark population, primary resection conferred a smaller significant survival benefit after standard multivariate adjustment (hazard ratio, 0.60) that persisted after propensity score weighting (hazard ratio, 0.59). But there was no longer a significant benefit in the instrumental variable analysis here.
“Among the entire cohort of patients with stage 4 colon cancer, primary tumor resection offered no survival benefit over systemic chemotherapy alone when the [instrumental variable] method was applied at the 1 year landmark,” the investigators write.
“Subject to selection and survivor treatment bias, standard regression analysis may overestimate the benefit of [primary tumor resection],” they concluded.
SAN FRANCISCO – Resecting the primary tumor in patients with metastatic colon or colorectal cancer may prolong survival. But then again, it may not.
This was the overarching take-home message from a trio of cohort studies presented at the Gastrointestinal Cancers Symposium cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology. Results were reported in a poster session.
“Whereas surgery is the primary treatment of localized colorectal cancer, resection of the primary tumor in patients with incurable metastatic disease is usually recommended for palliative purposes to manage obstruction, perforation, or bleeding,” Dr. Shahid Ahmed, lead investigator of one of the studies, noted in comments provided by e-mail. “The role of surgical resection of the primary tumor in patients with newly diagnosed incurable stage IV colorectal cancer remains controversial.”
In earlier research, he and colleagues found a survival benefit of primary resection among Canadian patients whose cancer was diagnosed between 1992 and 2005 (Cancer 2014;120:683-91). But the majority did not receive systemic therapy, and those who did were often given older regimens.
In a new study aimed at testing the association in the contemporary treatment era, the researchers analyzed data from 569 patients with stage IV colorectal cancer diagnosed between 2006 and 2010 who had a median follow-up of 11 months. Overall, 55% had resection of the primary tumor.
Among the 57% of patients who received systemic therapy, 91% received FOLFIRI or FOLFOX, 65% received bevacizumab (Avastin), and 10% received cetuximab (Erbitux) or panitumumab (Vectibix), according to Dr. Ahmed, professor of medicine, University of Saskatchewan, Canada.
Results for the entire cohort showed that median overall survival was 18 months in patients who had resection of their primary versus 4 months in those who did not (multivariate hazard ratio, 0.44; P less than .001).
Among the subgroup of patients who received chemotherapy, median survival was 27 months with primary resection versus 14 months without it (P less than .0001). And among the subgroup that specifically received FOLFIRI or FOLFOX and a biologic agent, it was 35 months with primary resection and 23 months without it (P less than .001).
“Surgical resection of primary tumor improves survival of patients with stage IV colorectal cancer, independent of other prognostic variables including age, performance status, comorbid illness, and chemotherapy,” maintained Dr. Ahmed. “The current study validates our findings and supports surgical resection of primary tumor in patients with stage IV colorectal cancer who are treated with modern chemotherapy and biologics.
“A well-designed prospective randomized trial is warranted to confirm the survival benefit conferred by the primary tumor resection,” he added, noting that two such trials in Europe – SYNCHRONOUS and CAIRO4 – are underway.
“If the magnitude of survival benefit is confirmed in these future randomized studies, surgical resection of the primary tumor could potentially be a more cost-effective intervention compared with novel systemic therapy in the management of metastatic colorectal cancer,” he concluded.
In a second study, Dr. Aaron Lewis, a surgical oncology fellow at the City of Hope, Duarte, Calif., and colleagues analyzed data from patients with stage IV colon cancer in the Surveillance, Epidemiology, and End Results (SEER) database for the years 1998 through 2011. They excluded those who died within 30 days of diagnosis or had resection of metastases. Overall, 70% of the 28,068 included patients had resection of their primary.
In multivariate analyses, patients who underwent resection had half the risk of death when compared with peers who did not have this surgery (hazard ratio, 0.49), reported Dr. Lewis.
Findings were essentially the same when the analysis was repeated in a subset of matched patients: Median survival was 17 months with resection versus 9 months without it (hazard ratio, 0.48; P less than .0001). Estimated 3-year survival was 23% and 6%, respectively.
“There are limitations, factors that we couldn’t completely control for. For example, there is no chemotherapy data in the SEER database. We didn’t know the timing of surgery in relation to chemotherapy. And we didn’t know whether these patients were asymptomatic or symptomatic,” Dr. Lewis noted in an interview. “But analysis of this huge group of patients in the United States that are getting treated shows that there is a survival benefit.”
Possible reasons why surgery might prolong life in this setting are unknown but may include the effects of tumor debulking or some enhancement of the immune response, he proposed.
To definitively confirm a survival benefit, a randomized controlled trial is needed, he agreed. “This seems to be a popular question in the literature in the last couple of years, so maybe somebody will be willing to take it on.”
In a third study, a team led by Dr. Zeinab Alawadi, a surgeon and postdoctoral fellow at the University of Texas MD Anderson Cancer Center, Houston, analyzed data from 14,399 patients in the National Cancer Data Base. They had been diagnosed with stage IV colon cancer between 2003 and 2005. The researchers excluded patients who had nonelective resection or surgery at other sites, such as metastasectomy.
The primary tumor was resected in 55% of all patients studied and in 74% of patients included in a 1-year landmark analysis done to account for early deaths related to comorbidity or disease burden, reported Dr. Alawadi.
In the entire cohort, primary resection conferred a significant survival benefit after standard multivariate adjustment (hazard ratio, 0.39) that persisted after propensity score weighting to account for treatment selection bias (hazard ratio, 0.41). The benefit was also significant, but much attenuated, in an instrumental variable analysis, another method for accounting for treatment selection bias (relative mortality rate, 0.88).
In the 1-year landmark population, primary resection conferred a smaller significant survival benefit after standard multivariate adjustment (hazard ratio, 0.60) that persisted after propensity score weighting (hazard ratio, 0.59). But there was no longer a significant benefit in the instrumental variable analysis here.
“Among the entire cohort of patients with stage 4 colon cancer, primary tumor resection offered no survival benefit over systemic chemotherapy alone when the [instrumental variable] method was applied at the 1 year landmark,” the investigators write.
“Subject to selection and survivor treatment bias, standard regression analysis may overestimate the benefit of [primary tumor resection],” they concluded.
AT THE GASTROINTESTINAL CANCERS SYMPOSIUM
Key clinical point: Data are mixed regarding an overall survival benefit of resecting the primary tumor.
Major finding: Two studies found a halving of the risk of death, whereas one study found lesser or even no benefit.
Data source: A trio of cohort studies in 569 patients, 28,068 patients, and 14,399 patients with metastatic colon or colorectal cancer.
Disclosures: Dr. Ahmed, Dr. Lewis, and Dr. Alawadi disclosed that they had no conflicts of interest.
Postop NSAIDs may raise anastomotic complications
Postoperative NSAIDs appear to raise the risk for anastomotic complications among patients undergoing nonelective colorectal resection, according to a report in JAMA Surgery.
“Given that other analgesic regimens are effective and well tolerated, these data may be enough from some surgeons to alter practice patterns,” said Dr. Timo W. Hakkarainen of the department of surgery at the University of Washington, Seattle, and his associates.
The recent development of intravenous formulations of NSAIDs has expanded their use in postoperative patients, chiefly because the drugs don’t carry the adverse effects of opioid analgesia, which include impaired GI motility. However, several small and single-institution studies have suggested that NSAIDs used in this setting may impair anastomotic healing and may raise the rates of leakage.
Dr. Hakkarainen and his associates examined this issue using data from a statewide surveillance system for surgical quality. They analyzed 90-day complications among 13,082 adults (mean age, 58 years) who underwent surgery of the GI tract with anastomosis at 47 participating hospitals throughout Washington State during a 5-year period. The investigators tracked the use of ibuprofen, naproxen, ketorolac tromethamine, Caldolor, celecoxib, and diclofenac during the first 24 hours after surgery; they assumed that in most cases, this involved IV NSAIDs because such patients don’t take oral medications within that time period.
Postoperative NSAIDs were given to 24% of the study population. This use was associated with a significantly increased risk for anastomotic leakage during the next 90 days, with an OR of 1.24. Further analysis showed that this association was largely restricted to the subgroup of patients who had nonelective colorectal surgery, in whom the relationship was even stronger, with an OR of 1.70. Among patients who had nonelective colorectal surgery, those who received postoperative NSAIDs had a 12.3% rate of anastomotic leakage, compared with an 8.3% rate among those who did not receive NSAIDs, the investigators said (JAMA Surg. 2015 Jan. 21 [doi:10.1001/jamasurg.2014.2239]).
This study was limited in that the records didn’t specify the dose or duration of NSAID use, didn’t take into account preoperative NSAID use, and didn’t include the timing of anastomotic leakage (immediately following surgery vs. weeks or months later). “We believe that [these] results are sufficient to suggest caution in the use of NSAIDs in the postoperative treatment of patients undergoing nonelective colorectal surgery, and highlight the importance of further evaluation of this association,” the investigators added.
Postoperative NSAIDs appear to raise the risk for anastomotic complications among patients undergoing nonelective colorectal resection, according to a report in JAMA Surgery.
“Given that other analgesic regimens are effective and well tolerated, these data may be enough from some surgeons to alter practice patterns,” said Dr. Timo W. Hakkarainen of the department of surgery at the University of Washington, Seattle, and his associates.
The recent development of intravenous formulations of NSAIDs has expanded their use in postoperative patients, chiefly because the drugs don’t carry the adverse effects of opioid analgesia, which include impaired GI motility. However, several small and single-institution studies have suggested that NSAIDs used in this setting may impair anastomotic healing and may raise the rates of leakage.
Dr. Hakkarainen and his associates examined this issue using data from a statewide surveillance system for surgical quality. They analyzed 90-day complications among 13,082 adults (mean age, 58 years) who underwent surgery of the GI tract with anastomosis at 47 participating hospitals throughout Washington State during a 5-year period. The investigators tracked the use of ibuprofen, naproxen, ketorolac tromethamine, Caldolor, celecoxib, and diclofenac during the first 24 hours after surgery; they assumed that in most cases, this involved IV NSAIDs because such patients don’t take oral medications within that time period.
Postoperative NSAIDs were given to 24% of the study population. This use was associated with a significantly increased risk for anastomotic leakage during the next 90 days, with an OR of 1.24. Further analysis showed that this association was largely restricted to the subgroup of patients who had nonelective colorectal surgery, in whom the relationship was even stronger, with an OR of 1.70. Among patients who had nonelective colorectal surgery, those who received postoperative NSAIDs had a 12.3% rate of anastomotic leakage, compared with an 8.3% rate among those who did not receive NSAIDs, the investigators said (JAMA Surg. 2015 Jan. 21 [doi:10.1001/jamasurg.2014.2239]).
This study was limited in that the records didn’t specify the dose or duration of NSAID use, didn’t take into account preoperative NSAID use, and didn’t include the timing of anastomotic leakage (immediately following surgery vs. weeks or months later). “We believe that [these] results are sufficient to suggest caution in the use of NSAIDs in the postoperative treatment of patients undergoing nonelective colorectal surgery, and highlight the importance of further evaluation of this association,” the investigators added.
Postoperative NSAIDs appear to raise the risk for anastomotic complications among patients undergoing nonelective colorectal resection, according to a report in JAMA Surgery.
“Given that other analgesic regimens are effective and well tolerated, these data may be enough from some surgeons to alter practice patterns,” said Dr. Timo W. Hakkarainen of the department of surgery at the University of Washington, Seattle, and his associates.
The recent development of intravenous formulations of NSAIDs has expanded their use in postoperative patients, chiefly because the drugs don’t carry the adverse effects of opioid analgesia, which include impaired GI motility. However, several small and single-institution studies have suggested that NSAIDs used in this setting may impair anastomotic healing and may raise the rates of leakage.
Dr. Hakkarainen and his associates examined this issue using data from a statewide surveillance system for surgical quality. They analyzed 90-day complications among 13,082 adults (mean age, 58 years) who underwent surgery of the GI tract with anastomosis at 47 participating hospitals throughout Washington State during a 5-year period. The investigators tracked the use of ibuprofen, naproxen, ketorolac tromethamine, Caldolor, celecoxib, and diclofenac during the first 24 hours after surgery; they assumed that in most cases, this involved IV NSAIDs because such patients don’t take oral medications within that time period.
Postoperative NSAIDs were given to 24% of the study population. This use was associated with a significantly increased risk for anastomotic leakage during the next 90 days, with an OR of 1.24. Further analysis showed that this association was largely restricted to the subgroup of patients who had nonelective colorectal surgery, in whom the relationship was even stronger, with an OR of 1.70. Among patients who had nonelective colorectal surgery, those who received postoperative NSAIDs had a 12.3% rate of anastomotic leakage, compared with an 8.3% rate among those who did not receive NSAIDs, the investigators said (JAMA Surg. 2015 Jan. 21 [doi:10.1001/jamasurg.2014.2239]).
This study was limited in that the records didn’t specify the dose or duration of NSAID use, didn’t take into account preoperative NSAID use, and didn’t include the timing of anastomotic leakage (immediately following surgery vs. weeks or months later). “We believe that [these] results are sufficient to suggest caution in the use of NSAIDs in the postoperative treatment of patients undergoing nonelective colorectal surgery, and highlight the importance of further evaluation of this association,” the investigators added.
Key clinical point: Postoperative NSAIDs appear to raise the risk of anastomotic complications in patients undergoing nonelective colorectal resection.
Major finding: Among patients who had nonelective colorectal surgery, those who received postoperative NSAIDs had a 12.3% rate of anastomotic leakage, compared with an 8.3% rate among those who did not receive NSAIDs.
Data source: A secondary analysis of a cohort study involving 13,082 patients who had GI-tract surgery with anastomosis at 47 hospitals in Washington State during a 5-year period.
Disclosures: The Comparative Effective Research Translation Network of the Agency for Healthcare Research and Quality, the Washington State Life Science Discovery Fund, and the National Institutes of Health supported the study. Dr. Hakkarainen and his associates reported having no financial disclosures.
Statins found to have a survival benefit in colorectal cancer
SAN FRANCISCO – Statin use confers a survival benefit in patients with colorectal cancer, suggests a systematic review and meta-analysis presented at the annual Gastrointestinal Cancers Symposium.
Investigators analyzed data from seven observational studies having a total of 64,773 patients with this cancer, a fifth of whom were using statins. Results showed that statin users had a nearly 30% reduction in the adjusted risk of all-cause mortality relative to nonusers, Dr. Arjun Gupta, the lead investigator, reported in a poster session at the meeting cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology.
“There is a lot of interest in non–cytotoxic chemotherapy drugs that can be used to treat cancer,” he said in an interview. “Thirty percent is a very, very significant number – we use chemotherapy drugs even if they have just a 10% survival benefit. So I am hopeful that this will help people.”
Accumulating preclinical data suggest that statins have anticancer actions, inhibiting cell proliferation and angiogenesis, and inducing apoptosis, according to Dr. Gupta, who is a resident in internal medicine at the University of Texas Southwestern Medical Center, Dallas. Previous studies have shown use to be associated with a decreased risk of developing colorectal cancer, but its impact on established disease has not been well assessed on a large scale.
“This is all observational data. There is no randomized controlled trial that’s been done,” he acknowledged. “But these data certainly seem to point out that this is something we can do. It’s a cheap and easy, safe drug which people take for years on end without any complications.”
At present, statins are approved by the Food and Drug Administration only for lipid-lowering indications, he noted. However, patients with colorectal cancer often have cardiovascular risk factors that make them candidates for statins.
“No one really gets a statin right now for colon cancer. We are just sort of lucky, maybe, that so many people with colon cancer have high cholesterol and are getting these drugs,” Dr. Gupta said. “But hopefully, my dream is 5 years down the line, it will be prescribed to people for this indication.”
Study results showed that in multivariate analyses, patients using statins were 26% less likely to die of any cause (hazard ratio, 0.74). Findings were similar whether they had colon cancer (hazard ratio, 0.79) or rectal cancer (hazard ratio, 0.63).
When analyses were restricted to studies that adjusted for concomitant use of aspirin and nonsteroidal anti-inflammatory drugs, statin users had significantly reduced risks of both all-cause mortality (hazard ratio, 0.74) and colorectal cancer–specific mortality (HR, 0.76), reported Dr. Gupta.
Patients who started using the medications after their cancer diagnosis had a significantly reduced risk of colorectal cancer-specific mortality (HR, 0.70) but not all-cause mortality.
Dr. Gupta disclosed that he had no relevant conflicts of interest.
SAN FRANCISCO – Statin use confers a survival benefit in patients with colorectal cancer, suggests a systematic review and meta-analysis presented at the annual Gastrointestinal Cancers Symposium.
Investigators analyzed data from seven observational studies having a total of 64,773 patients with this cancer, a fifth of whom were using statins. Results showed that statin users had a nearly 30% reduction in the adjusted risk of all-cause mortality relative to nonusers, Dr. Arjun Gupta, the lead investigator, reported in a poster session at the meeting cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology.
“There is a lot of interest in non–cytotoxic chemotherapy drugs that can be used to treat cancer,” he said in an interview. “Thirty percent is a very, very significant number – we use chemotherapy drugs even if they have just a 10% survival benefit. So I am hopeful that this will help people.”
Accumulating preclinical data suggest that statins have anticancer actions, inhibiting cell proliferation and angiogenesis, and inducing apoptosis, according to Dr. Gupta, who is a resident in internal medicine at the University of Texas Southwestern Medical Center, Dallas. Previous studies have shown use to be associated with a decreased risk of developing colorectal cancer, but its impact on established disease has not been well assessed on a large scale.
“This is all observational data. There is no randomized controlled trial that’s been done,” he acknowledged. “But these data certainly seem to point out that this is something we can do. It’s a cheap and easy, safe drug which people take for years on end without any complications.”
At present, statins are approved by the Food and Drug Administration only for lipid-lowering indications, he noted. However, patients with colorectal cancer often have cardiovascular risk factors that make them candidates for statins.
“No one really gets a statin right now for colon cancer. We are just sort of lucky, maybe, that so many people with colon cancer have high cholesterol and are getting these drugs,” Dr. Gupta said. “But hopefully, my dream is 5 years down the line, it will be prescribed to people for this indication.”
Study results showed that in multivariate analyses, patients using statins were 26% less likely to die of any cause (hazard ratio, 0.74). Findings were similar whether they had colon cancer (hazard ratio, 0.79) or rectal cancer (hazard ratio, 0.63).
When analyses were restricted to studies that adjusted for concomitant use of aspirin and nonsteroidal anti-inflammatory drugs, statin users had significantly reduced risks of both all-cause mortality (hazard ratio, 0.74) and colorectal cancer–specific mortality (HR, 0.76), reported Dr. Gupta.
Patients who started using the medications after their cancer diagnosis had a significantly reduced risk of colorectal cancer-specific mortality (HR, 0.70) but not all-cause mortality.
Dr. Gupta disclosed that he had no relevant conflicts of interest.
SAN FRANCISCO – Statin use confers a survival benefit in patients with colorectal cancer, suggests a systematic review and meta-analysis presented at the annual Gastrointestinal Cancers Symposium.
Investigators analyzed data from seven observational studies having a total of 64,773 patients with this cancer, a fifth of whom were using statins. Results showed that statin users had a nearly 30% reduction in the adjusted risk of all-cause mortality relative to nonusers, Dr. Arjun Gupta, the lead investigator, reported in a poster session at the meeting cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology.
“There is a lot of interest in non–cytotoxic chemotherapy drugs that can be used to treat cancer,” he said in an interview. “Thirty percent is a very, very significant number – we use chemotherapy drugs even if they have just a 10% survival benefit. So I am hopeful that this will help people.”
Accumulating preclinical data suggest that statins have anticancer actions, inhibiting cell proliferation and angiogenesis, and inducing apoptosis, according to Dr. Gupta, who is a resident in internal medicine at the University of Texas Southwestern Medical Center, Dallas. Previous studies have shown use to be associated with a decreased risk of developing colorectal cancer, but its impact on established disease has not been well assessed on a large scale.
“This is all observational data. There is no randomized controlled trial that’s been done,” he acknowledged. “But these data certainly seem to point out that this is something we can do. It’s a cheap and easy, safe drug which people take for years on end without any complications.”
At present, statins are approved by the Food and Drug Administration only for lipid-lowering indications, he noted. However, patients with colorectal cancer often have cardiovascular risk factors that make them candidates for statins.
“No one really gets a statin right now for colon cancer. We are just sort of lucky, maybe, that so many people with colon cancer have high cholesterol and are getting these drugs,” Dr. Gupta said. “But hopefully, my dream is 5 years down the line, it will be prescribed to people for this indication.”
Study results showed that in multivariate analyses, patients using statins were 26% less likely to die of any cause (hazard ratio, 0.74). Findings were similar whether they had colon cancer (hazard ratio, 0.79) or rectal cancer (hazard ratio, 0.63).
When analyses were restricted to studies that adjusted for concomitant use of aspirin and nonsteroidal anti-inflammatory drugs, statin users had significantly reduced risks of both all-cause mortality (hazard ratio, 0.74) and colorectal cancer–specific mortality (HR, 0.76), reported Dr. Gupta.
Patients who started using the medications after their cancer diagnosis had a significantly reduced risk of colorectal cancer-specific mortality (HR, 0.70) but not all-cause mortality.
Dr. Gupta disclosed that he had no relevant conflicts of interest.
Key clinical point: Patients with colorectal cancer who take statins are less likely to die.
Major finding: Relative to nonusers, statin users had a 26% reduction in the adjusted risk of all-cause mortality.
Data source: A systematic review and meta-analysis of seven observational studies having a total of 64,773 patients.
Disclosures: Dr. Gupta disclosed that he had no relevant conflicts of interest.
Preoperative hepatic, regional arterial chemo nets better outcomes in early CRC
SAN FRANCISCO – Giving preoperative chemotherapy directly into the tumor-feeding artery and prophylactically into the common hepatic artery to target any liver micrometastases improves outcomes in patients with early colorectal cancer undergoing curative resection. But benefit is seen mainly in patients with stage III disease.
These were among the key findings of the randomized multicenter phase III PHRAC trial (Preoperative Hepatic and Regional Arterial Chemotherapy) conducted among 688 patients in China with stage II or III colorectal cancer.
Compared with curative resection alone, followed by adjuvant systemic chemotherapy with the modified FOLFOX6 regimen, the addition of preoperative arterial chemotherapy reduced the 5-year estimated risks of disease-free survival events by 40%, of liver metastases by 61%, and of death by 41% in the trial population overall, according to data reported at the annual Gastrointestinal Cancers Symposium cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology. But stratified analyses showed that these benefits were significant only among the patients with stage III disease.
“Preoperative hepatic and regional arterial chemotherapy had no effect on colorectal cancer surgery or postoperative morbidity,” commented lead investigator Jianmin Xu, an oncologist at the Zhongshan Hospital, Fudan University, in Shanghai. This therapy “is safe and feasible and can reduce liver metastasis and improve disease-free survival and overall survival, especially for the stage III patients.”
“Dr. Xu’s study was a positive study, but we have no description of the catheter problems or the number of patients who could be treated,” commented invited discussant Dr. Nancy E. Kemeny of Memorial Sloan-Kettering Cancer Center, New York. Additionally, outcomes in the control group fell short of those seen in patients treated with FOLFOX historically.
Despite accumulating evidence of benefit, the procedure is not yet ready for incorporation into routine care, according to Dr. Kemeny. “Since we don’t have a lot of information about the problems with the catheters or other problems like that, we need a larger, international study. I think one should be done because if you can prevent recurrences right away, which is what this study is suggesting, then that’s very good for these patients. We have ways of dealing with this afterwards, but it would be nice to prevent it right way. So given the fact that we already have five studies suggesting some benefit with this type of treatment, we should move into a large randomized study,” she recommended.
A session attendee noted that although colon cancer most commonly recurs in the liver, rectal cancer most commonly recurs in the pelvis. “Therefore, chemoradiotherapy is the standard of care now [for rectal cancer]. So infusional arterial chemotherapy should be used in patients with colon cancer, I think,” he said.
The patients with rectal cancer received radiation therapy after surgery if needed, Dr. Xu replied. “In the future, we will do subgroup analyses for colon cancer and for rectal cancer,” he added.
In the trial, patients from five hospitals in China were randomized to immediate curative primary surgery or to hepatic and regional arterial chemotherapy – floxuridine (FUDR), mitomycin C, and oxaliplatin delivered to both the main tumor-supplying artery and to the common hepatic artery – followed by curative primary surgery a week later. All patients received the same adjuvant therapy.
In the intention-to-treat population, estimated 5-year disease-free survival, the trial’s primary endpoint, was 75% with preoperative arterial chemotherapy versus 61% without it (hazard ratio, 0.60; P less than .001), reported Dr. Xu, who disclosed no relevant conflicts of interest. Subgroup analyses indicated that the benefit was significant in patients with stage III disease (68% vs. 51%; HR, 0.62; P = .017) but showed only a trend in patients with stage II disease (84% vs. 74%; HR, 0.64; P = .068).
Patients in the arterial chemotherapy group also were less likely to develop liver metastases (8% vs. 18%; HR, 0.39; P less than .001) and had better overall survival (81% vs. 72%, HR, 0.59; P = .003). But subgroup analyses again showed that benefit was significant only in the stage III patients.
Efficacy findings were similar in the trial’s eligible population, which excluded patients who were found to have pathologic stage I or IV disease at surgery and patients who developed metastases within 6 months of surgery.A total of 25% of patients in the arterial chemotherapy group experienced grade 3 toxicity from the procedure. However, the rate of postoperative complications did not differ significantly between the arterial chemotherapy and control groups.
SAN FRANCISCO – Giving preoperative chemotherapy directly into the tumor-feeding artery and prophylactically into the common hepatic artery to target any liver micrometastases improves outcomes in patients with early colorectal cancer undergoing curative resection. But benefit is seen mainly in patients with stage III disease.
These were among the key findings of the randomized multicenter phase III PHRAC trial (Preoperative Hepatic and Regional Arterial Chemotherapy) conducted among 688 patients in China with stage II or III colorectal cancer.
Compared with curative resection alone, followed by adjuvant systemic chemotherapy with the modified FOLFOX6 regimen, the addition of preoperative arterial chemotherapy reduced the 5-year estimated risks of disease-free survival events by 40%, of liver metastases by 61%, and of death by 41% in the trial population overall, according to data reported at the annual Gastrointestinal Cancers Symposium cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology. But stratified analyses showed that these benefits were significant only among the patients with stage III disease.
“Preoperative hepatic and regional arterial chemotherapy had no effect on colorectal cancer surgery or postoperative morbidity,” commented lead investigator Jianmin Xu, an oncologist at the Zhongshan Hospital, Fudan University, in Shanghai. This therapy “is safe and feasible and can reduce liver metastasis and improve disease-free survival and overall survival, especially for the stage III patients.”
“Dr. Xu’s study was a positive study, but we have no description of the catheter problems or the number of patients who could be treated,” commented invited discussant Dr. Nancy E. Kemeny of Memorial Sloan-Kettering Cancer Center, New York. Additionally, outcomes in the control group fell short of those seen in patients treated with FOLFOX historically.
Despite accumulating evidence of benefit, the procedure is not yet ready for incorporation into routine care, according to Dr. Kemeny. “Since we don’t have a lot of information about the problems with the catheters or other problems like that, we need a larger, international study. I think one should be done because if you can prevent recurrences right away, which is what this study is suggesting, then that’s very good for these patients. We have ways of dealing with this afterwards, but it would be nice to prevent it right way. So given the fact that we already have five studies suggesting some benefit with this type of treatment, we should move into a large randomized study,” she recommended.
A session attendee noted that although colon cancer most commonly recurs in the liver, rectal cancer most commonly recurs in the pelvis. “Therefore, chemoradiotherapy is the standard of care now [for rectal cancer]. So infusional arterial chemotherapy should be used in patients with colon cancer, I think,” he said.
The patients with rectal cancer received radiation therapy after surgery if needed, Dr. Xu replied. “In the future, we will do subgroup analyses for colon cancer and for rectal cancer,” he added.
In the trial, patients from five hospitals in China were randomized to immediate curative primary surgery or to hepatic and regional arterial chemotherapy – floxuridine (FUDR), mitomycin C, and oxaliplatin delivered to both the main tumor-supplying artery and to the common hepatic artery – followed by curative primary surgery a week later. All patients received the same adjuvant therapy.
In the intention-to-treat population, estimated 5-year disease-free survival, the trial’s primary endpoint, was 75% with preoperative arterial chemotherapy versus 61% without it (hazard ratio, 0.60; P less than .001), reported Dr. Xu, who disclosed no relevant conflicts of interest. Subgroup analyses indicated that the benefit was significant in patients with stage III disease (68% vs. 51%; HR, 0.62; P = .017) but showed only a trend in patients with stage II disease (84% vs. 74%; HR, 0.64; P = .068).
Patients in the arterial chemotherapy group also were less likely to develop liver metastases (8% vs. 18%; HR, 0.39; P less than .001) and had better overall survival (81% vs. 72%, HR, 0.59; P = .003). But subgroup analyses again showed that benefit was significant only in the stage III patients.
Efficacy findings were similar in the trial’s eligible population, which excluded patients who were found to have pathologic stage I or IV disease at surgery and patients who developed metastases within 6 months of surgery.A total of 25% of patients in the arterial chemotherapy group experienced grade 3 toxicity from the procedure. However, the rate of postoperative complications did not differ significantly between the arterial chemotherapy and control groups.
SAN FRANCISCO – Giving preoperative chemotherapy directly into the tumor-feeding artery and prophylactically into the common hepatic artery to target any liver micrometastases improves outcomes in patients with early colorectal cancer undergoing curative resection. But benefit is seen mainly in patients with stage III disease.
These were among the key findings of the randomized multicenter phase III PHRAC trial (Preoperative Hepatic and Regional Arterial Chemotherapy) conducted among 688 patients in China with stage II or III colorectal cancer.
Compared with curative resection alone, followed by adjuvant systemic chemotherapy with the modified FOLFOX6 regimen, the addition of preoperative arterial chemotherapy reduced the 5-year estimated risks of disease-free survival events by 40%, of liver metastases by 61%, and of death by 41% in the trial population overall, according to data reported at the annual Gastrointestinal Cancers Symposium cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology. But stratified analyses showed that these benefits were significant only among the patients with stage III disease.
“Preoperative hepatic and regional arterial chemotherapy had no effect on colorectal cancer surgery or postoperative morbidity,” commented lead investigator Jianmin Xu, an oncologist at the Zhongshan Hospital, Fudan University, in Shanghai. This therapy “is safe and feasible and can reduce liver metastasis and improve disease-free survival and overall survival, especially for the stage III patients.”
“Dr. Xu’s study was a positive study, but we have no description of the catheter problems or the number of patients who could be treated,” commented invited discussant Dr. Nancy E. Kemeny of Memorial Sloan-Kettering Cancer Center, New York. Additionally, outcomes in the control group fell short of those seen in patients treated with FOLFOX historically.
Despite accumulating evidence of benefit, the procedure is not yet ready for incorporation into routine care, according to Dr. Kemeny. “Since we don’t have a lot of information about the problems with the catheters or other problems like that, we need a larger, international study. I think one should be done because if you can prevent recurrences right away, which is what this study is suggesting, then that’s very good for these patients. We have ways of dealing with this afterwards, but it would be nice to prevent it right way. So given the fact that we already have five studies suggesting some benefit with this type of treatment, we should move into a large randomized study,” she recommended.
A session attendee noted that although colon cancer most commonly recurs in the liver, rectal cancer most commonly recurs in the pelvis. “Therefore, chemoradiotherapy is the standard of care now [for rectal cancer]. So infusional arterial chemotherapy should be used in patients with colon cancer, I think,” he said.
The patients with rectal cancer received radiation therapy after surgery if needed, Dr. Xu replied. “In the future, we will do subgroup analyses for colon cancer and for rectal cancer,” he added.
In the trial, patients from five hospitals in China were randomized to immediate curative primary surgery or to hepatic and regional arterial chemotherapy – floxuridine (FUDR), mitomycin C, and oxaliplatin delivered to both the main tumor-supplying artery and to the common hepatic artery – followed by curative primary surgery a week later. All patients received the same adjuvant therapy.
In the intention-to-treat population, estimated 5-year disease-free survival, the trial’s primary endpoint, was 75% with preoperative arterial chemotherapy versus 61% without it (hazard ratio, 0.60; P less than .001), reported Dr. Xu, who disclosed no relevant conflicts of interest. Subgroup analyses indicated that the benefit was significant in patients with stage III disease (68% vs. 51%; HR, 0.62; P = .017) but showed only a trend in patients with stage II disease (84% vs. 74%; HR, 0.64; P = .068).
Patients in the arterial chemotherapy group also were less likely to develop liver metastases (8% vs. 18%; HR, 0.39; P less than .001) and had better overall survival (81% vs. 72%, HR, 0.59; P = .003). But subgroup analyses again showed that benefit was significant only in the stage III patients.
Efficacy findings were similar in the trial’s eligible population, which excluded patients who were found to have pathologic stage I or IV disease at surgery and patients who developed metastases within 6 months of surgery.A total of 25% of patients in the arterial chemotherapy group experienced grade 3 toxicity from the procedure. However, the rate of postoperative complications did not differ significantly between the arterial chemotherapy and control groups.
AT THE 2015 GASTROINTESTINAL CANCERS SYMPOSIUM
Key clinical point: Preoperative arterial chemotherapy improves outcomes, but mainly for stage III patients.
Major finding: Five-year disease-free survival was 75% with preoperative arterial chemotherapy vs. 61% without it.
Data source: A randomized phase III trial among 688 patients undergoing curative resection of stage II or III colorectal cancer.
Disclosures: Dr. Xu disclosed that he had no relevant conflicts of interest.
Stage IV CRC survival rates up, resection rates down
Although significantly fewer patients with stage IV colorectal cancer have undergone primary tumor resection (PTR) since 1988, overall rates of patient survival have improved over that period, according to the results of a study published in JAMA.
“The role of PTR for asymptomatic patients remains controversial. Some physicians advocate PTR to prevent the development of symptoms associated with an intact primary tumor,” wrote lead author Chung-Yuan Hu, Ph.D., of the University of Texas M.D. Anderson Cancer Center in Houston, and his associates. “Approximately 10%-25% of patients with intact primary tumors will develop symptoms, but there is considerable morbidity (4%-30%) and mortality (2%-10%) associated with noncurative PTR” (JAMA Surg. [doi:10.1001/jamasurg.2014.2253]).
In a retrospective cohort study, Dr. Hu and his colleagues used data on 64,157 patients diagnosed with stage IV colon or rectal cancer between Jan. 1, 1988, and Dec. 31, 2010, from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) CRC registry. The primary outcome measured was difference in PTR rates over the study period. Included subjects had either undergone PTR or not, and investigators calculated rates of PTR and median relative survival for each year. Joinpoint regression analyses were used to determine when a “significant change” in PTR rate had occurred, while logistic regression analyses were used to assess factors associated with PTR.
Findings indicated that 43,273 (67.4%) of the patients analyzed for the study had undergone PTR; overall, between 1988 and 2010, annual PTR rates declined from 74.5% to 57.4%, respectively (P < .001). “Significant change” was noted between 1998 and 2001 and from 2001 to 2010: –0.41% and –2.39%, respectively (P < .001 in both cases). However, median annual survival rates improved substantially from 1988 to 2009, changing from 8.6% to 17.8%, respectively (P < .001).
“Despite the availability of more effective chemotherapeutic options, a considerable number of patients with stage IV CRC continue to undergo PTR,” wrote the authors. “Our findings indicate potential overuse of PTR among these patients and highlight a need to better understand the clinical decisions and outcomes associated with that treatment.”
This study was supported in part by grants from the National Institute of Health’s National Cancer Institute, and the American Society of Clinical Oncology Foundation Career Development Award. Authors reported no financial conflicts of interest.
Although significantly fewer patients with stage IV colorectal cancer have undergone primary tumor resection (PTR) since 1988, overall rates of patient survival have improved over that period, according to the results of a study published in JAMA.
“The role of PTR for asymptomatic patients remains controversial. Some physicians advocate PTR to prevent the development of symptoms associated with an intact primary tumor,” wrote lead author Chung-Yuan Hu, Ph.D., of the University of Texas M.D. Anderson Cancer Center in Houston, and his associates. “Approximately 10%-25% of patients with intact primary tumors will develop symptoms, but there is considerable morbidity (4%-30%) and mortality (2%-10%) associated with noncurative PTR” (JAMA Surg. [doi:10.1001/jamasurg.2014.2253]).
In a retrospective cohort study, Dr. Hu and his colleagues used data on 64,157 patients diagnosed with stage IV colon or rectal cancer between Jan. 1, 1988, and Dec. 31, 2010, from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) CRC registry. The primary outcome measured was difference in PTR rates over the study period. Included subjects had either undergone PTR or not, and investigators calculated rates of PTR and median relative survival for each year. Joinpoint regression analyses were used to determine when a “significant change” in PTR rate had occurred, while logistic regression analyses were used to assess factors associated with PTR.
Findings indicated that 43,273 (67.4%) of the patients analyzed for the study had undergone PTR; overall, between 1988 and 2010, annual PTR rates declined from 74.5% to 57.4%, respectively (P < .001). “Significant change” was noted between 1998 and 2001 and from 2001 to 2010: –0.41% and –2.39%, respectively (P < .001 in both cases). However, median annual survival rates improved substantially from 1988 to 2009, changing from 8.6% to 17.8%, respectively (P < .001).
“Despite the availability of more effective chemotherapeutic options, a considerable number of patients with stage IV CRC continue to undergo PTR,” wrote the authors. “Our findings indicate potential overuse of PTR among these patients and highlight a need to better understand the clinical decisions and outcomes associated with that treatment.”
This study was supported in part by grants from the National Institute of Health’s National Cancer Institute, and the American Society of Clinical Oncology Foundation Career Development Award. Authors reported no financial conflicts of interest.
Although significantly fewer patients with stage IV colorectal cancer have undergone primary tumor resection (PTR) since 1988, overall rates of patient survival have improved over that period, according to the results of a study published in JAMA.
“The role of PTR for asymptomatic patients remains controversial. Some physicians advocate PTR to prevent the development of symptoms associated with an intact primary tumor,” wrote lead author Chung-Yuan Hu, Ph.D., of the University of Texas M.D. Anderson Cancer Center in Houston, and his associates. “Approximately 10%-25% of patients with intact primary tumors will develop symptoms, but there is considerable morbidity (4%-30%) and mortality (2%-10%) associated with noncurative PTR” (JAMA Surg. [doi:10.1001/jamasurg.2014.2253]).
In a retrospective cohort study, Dr. Hu and his colleagues used data on 64,157 patients diagnosed with stage IV colon or rectal cancer between Jan. 1, 1988, and Dec. 31, 2010, from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) CRC registry. The primary outcome measured was difference in PTR rates over the study period. Included subjects had either undergone PTR or not, and investigators calculated rates of PTR and median relative survival for each year. Joinpoint regression analyses were used to determine when a “significant change” in PTR rate had occurred, while logistic regression analyses were used to assess factors associated with PTR.
Findings indicated that 43,273 (67.4%) of the patients analyzed for the study had undergone PTR; overall, between 1988 and 2010, annual PTR rates declined from 74.5% to 57.4%, respectively (P < .001). “Significant change” was noted between 1998 and 2001 and from 2001 to 2010: –0.41% and –2.39%, respectively (P < .001 in both cases). However, median annual survival rates improved substantially from 1988 to 2009, changing from 8.6% to 17.8%, respectively (P < .001).
“Despite the availability of more effective chemotherapeutic options, a considerable number of patients with stage IV CRC continue to undergo PTR,” wrote the authors. “Our findings indicate potential overuse of PTR among these patients and highlight a need to better understand the clinical decisions and outcomes associated with that treatment.”
This study was supported in part by grants from the National Institute of Health’s National Cancer Institute, and the American Society of Clinical Oncology Foundation Career Development Award. Authors reported no financial conflicts of interest.
FROM JAMA SURGERY
Key clinical point: Although rates of primary tumor resection in patients with stage IV colorectal cancer have decreased in recent years, patient survival rates have improved.
Major finding: The annual rate of primary tumor resection decreased from 74.5% in 1988 to 57.4% in 2010, while median relative survival rate improved from 8.6% in 1988 to 17.8% in 2009.
Data source: Retrospective cohort study examining data on 64,157 subjects in the National Cancer Institute’s SEER CRC registry.
Disclosures: Study was supported in part by grants from the National Institute of Health’s National Cancer Institute, and the American Society of Clinical Oncology Foundation Career Development Award. Authors reported no financial conflicts of interest.
Skipping surgery is an option for some patients with rectal cancer
Patients with locally advanced rectal cancer who have a complete response to neoadjuvant therapy can skip surgery with little compromise in outcomes, according to a retrospective review that will be reported this week at the annual Gastrointestinal Cancers Symposium.
Investigators at the Memorial Sloan-Kettering Cancer Center in New York studied 145 patients with stage I to III rectal cancer who received neoadjuvant therapy there between 2006 and 2013. The meeting was cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology
After a median follow-up of 3.5 years, there were no significant differences in disease-specific or overall survival between patients who had a clinical complete response to neoadjuvant therapy of radiation and chemotherapy and skipped surgery, and patients who underwent rectal resection (the current standard strategy in the United States) with a pathologic complete response. Additionally, more than three-fourths of the group skipping surgery had preservation of rectal function.
“Nonoperative management appears to be a safe and effective treatment strategy and achieves a high rate of rectal preservation,” senior investigator Dr. Philip B. Paty, a surgical oncologist at Memorial Sloan-Kettering Cancer Center, New York, commented in a press briefing held before the symposium.
Ideally, the findings would be tested in a randomized trial, he said; however, “there’s too many factors and too much patient autonomy involved here, so that no one really believes asking people to sign up for a randomized trial where rectal resection is decided by a flip of the coin would ever accrue patients.” Short of that, rigorous prospective studies, such as a phase II trial now open at the center, will provide critical information.
Press briefing moderator Dr. Smitha S. Krishnamurthi of Case Western Reserve University, Cleveland, commented, “These are important findings for patients with rectal cancer because removal of the rectum can result in altered bowel habits or the need for permanent colostomy. This study set the bar very high, comparing the results of nonoperative management to the results seen in patients who had no cancer left under the microscope at the time of surgery, and in this study, the nonoperative management appears to compare favorably.”
“We do need longer follow-up though, to be sure that these patients will have disease-specific survival that equals what is achieved with surgery in the long term,” she cautioned, such as the conventional 5 years of observation patients typically receive. “And then of course a prospective study also would be helpful to see the effects of this approach.”
In the study, Dr. Paty and colleagues identified 73 patients who had a clinical complete response (no cancer detected on physical exam, endoscopy, or imaging) to neoadjuvant therapy of radiation and chemotherapy and—by mutual agreement of physician and patient—had nonoperative management (watchful waiting), consisting initially of follow-up at 3- to 4-month intervals by digital rectal and endoscopic exams and at 6-month intervals by imaging. They used as a comparison group 72 patients who underwent standard total mesorectal excision and had a pathologic complete response (no viable cancer cells found microscopically in the resected tissue).
Results showed that 74% of the patients who did not have surgery had a sustained clinical complete response, with no regrowth of tumor during follow-up, reported Dr. Paty. Among the other 26% whose tumors regrew, most of the recurrences were clinically detectable and all patients had successful resections with clean margins. Overall, 77% of the nonsurgical patients had rectal preservation and 98% had local control.
The nonsurgical group did not differ significantly from the surgical group with respect to 4-year rates of disease-specific survival (91% vs. 96%) and overall survival (91% vs. 95%).
The investigators plan to report quality of life data in the future, Dr. Paty said. “But I think it’s pretty obvious to everyone who’s managed these patients that if you can avoid rectal surgery, the quality of life and particularly bowel function is far superior to those who have had rectal resection.”
Successful nonoperative management hinges critically on careful patient selection, close follow-up, and use of salvage surgery, he stressed. Additionally, “the informed consent process in nonoperative management is extremely important, and I always tell patients that they are taking a slight risk. It’s hard to imagine that not operating is going to have 100% equivalent cancer outcomes as operating. That is hard to believe. So we never sell it as being absolutely as good, but …with good follow-up, the results seem to be very close if not equivalent.”
In his experience, most patients are willing to accept this option. “In fact, I have only had two patients [out of more than 100] decline nonoperative management when I thought they were candidates—both young, both with young children, both not wanting to take even the slightest risk of leaving cancer in the rectal wall or their body,” he commented.
Adoption of nonoperative management has been slow in the United States for a variety of reasons, according to Dr. Paty. “I think the bottom line is that practicing watch and wait, nonoperative management is more difficult for the surgeon. It requires first the judgment that the cancer’s gone. You have to follow the patient longer after radiation; sometimes the complete response will take up to 3 months. And there is also the medical-legal issue of deviating from the standard of care. … So I think it was operationally a difficult thing to do, it didn’t fit with the existing paradigm very well.”
But that is changing as more data roll in. “What’s happened in the last I will say 2-3 years is that there are centers publishing their experience, ours being the largest outside Brazil and the first in North America. Another group in the Netherlands has published a group of about 25 patients,” he explained. “Talking with people at meetings around the world, centers are adopting it, and I think that many leaders in clinical trials in rectal cancer recognize that this option is not only reasonable, but perhaps it’s necessary to inform patients that it is an option.”
Patients with locally advanced rectal cancer who have a complete response to neoadjuvant therapy can skip surgery with little compromise in outcomes, according to a retrospective review that will be reported this week at the annual Gastrointestinal Cancers Symposium.
Investigators at the Memorial Sloan-Kettering Cancer Center in New York studied 145 patients with stage I to III rectal cancer who received neoadjuvant therapy there between 2006 and 2013. The meeting was cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology
After a median follow-up of 3.5 years, there were no significant differences in disease-specific or overall survival between patients who had a clinical complete response to neoadjuvant therapy of radiation and chemotherapy and skipped surgery, and patients who underwent rectal resection (the current standard strategy in the United States) with a pathologic complete response. Additionally, more than three-fourths of the group skipping surgery had preservation of rectal function.
“Nonoperative management appears to be a safe and effective treatment strategy and achieves a high rate of rectal preservation,” senior investigator Dr. Philip B. Paty, a surgical oncologist at Memorial Sloan-Kettering Cancer Center, New York, commented in a press briefing held before the symposium.
Ideally, the findings would be tested in a randomized trial, he said; however, “there’s too many factors and too much patient autonomy involved here, so that no one really believes asking people to sign up for a randomized trial where rectal resection is decided by a flip of the coin would ever accrue patients.” Short of that, rigorous prospective studies, such as a phase II trial now open at the center, will provide critical information.
Press briefing moderator Dr. Smitha S. Krishnamurthi of Case Western Reserve University, Cleveland, commented, “These are important findings for patients with rectal cancer because removal of the rectum can result in altered bowel habits or the need for permanent colostomy. This study set the bar very high, comparing the results of nonoperative management to the results seen in patients who had no cancer left under the microscope at the time of surgery, and in this study, the nonoperative management appears to compare favorably.”
“We do need longer follow-up though, to be sure that these patients will have disease-specific survival that equals what is achieved with surgery in the long term,” she cautioned, such as the conventional 5 years of observation patients typically receive. “And then of course a prospective study also would be helpful to see the effects of this approach.”
In the study, Dr. Paty and colleagues identified 73 patients who had a clinical complete response (no cancer detected on physical exam, endoscopy, or imaging) to neoadjuvant therapy of radiation and chemotherapy and—by mutual agreement of physician and patient—had nonoperative management (watchful waiting), consisting initially of follow-up at 3- to 4-month intervals by digital rectal and endoscopic exams and at 6-month intervals by imaging. They used as a comparison group 72 patients who underwent standard total mesorectal excision and had a pathologic complete response (no viable cancer cells found microscopically in the resected tissue).
Results showed that 74% of the patients who did not have surgery had a sustained clinical complete response, with no regrowth of tumor during follow-up, reported Dr. Paty. Among the other 26% whose tumors regrew, most of the recurrences were clinically detectable and all patients had successful resections with clean margins. Overall, 77% of the nonsurgical patients had rectal preservation and 98% had local control.
The nonsurgical group did not differ significantly from the surgical group with respect to 4-year rates of disease-specific survival (91% vs. 96%) and overall survival (91% vs. 95%).
The investigators plan to report quality of life data in the future, Dr. Paty said. “But I think it’s pretty obvious to everyone who’s managed these patients that if you can avoid rectal surgery, the quality of life and particularly bowel function is far superior to those who have had rectal resection.”
Successful nonoperative management hinges critically on careful patient selection, close follow-up, and use of salvage surgery, he stressed. Additionally, “the informed consent process in nonoperative management is extremely important, and I always tell patients that they are taking a slight risk. It’s hard to imagine that not operating is going to have 100% equivalent cancer outcomes as operating. That is hard to believe. So we never sell it as being absolutely as good, but …with good follow-up, the results seem to be very close if not equivalent.”
In his experience, most patients are willing to accept this option. “In fact, I have only had two patients [out of more than 100] decline nonoperative management when I thought they were candidates—both young, both with young children, both not wanting to take even the slightest risk of leaving cancer in the rectal wall or their body,” he commented.
Adoption of nonoperative management has been slow in the United States for a variety of reasons, according to Dr. Paty. “I think the bottom line is that practicing watch and wait, nonoperative management is more difficult for the surgeon. It requires first the judgment that the cancer’s gone. You have to follow the patient longer after radiation; sometimes the complete response will take up to 3 months. And there is also the medical-legal issue of deviating from the standard of care. … So I think it was operationally a difficult thing to do, it didn’t fit with the existing paradigm very well.”
But that is changing as more data roll in. “What’s happened in the last I will say 2-3 years is that there are centers publishing their experience, ours being the largest outside Brazil and the first in North America. Another group in the Netherlands has published a group of about 25 patients,” he explained. “Talking with people at meetings around the world, centers are adopting it, and I think that many leaders in clinical trials in rectal cancer recognize that this option is not only reasonable, but perhaps it’s necessary to inform patients that it is an option.”
Patients with locally advanced rectal cancer who have a complete response to neoadjuvant therapy can skip surgery with little compromise in outcomes, according to a retrospective review that will be reported this week at the annual Gastrointestinal Cancers Symposium.
Investigators at the Memorial Sloan-Kettering Cancer Center in New York studied 145 patients with stage I to III rectal cancer who received neoadjuvant therapy there between 2006 and 2013. The meeting was cosponsored by the AGA Institute, the American Society of Clinical Oncology, ASTRO, and the Society of Surgical Oncology
After a median follow-up of 3.5 years, there were no significant differences in disease-specific or overall survival between patients who had a clinical complete response to neoadjuvant therapy of radiation and chemotherapy and skipped surgery, and patients who underwent rectal resection (the current standard strategy in the United States) with a pathologic complete response. Additionally, more than three-fourths of the group skipping surgery had preservation of rectal function.
“Nonoperative management appears to be a safe and effective treatment strategy and achieves a high rate of rectal preservation,” senior investigator Dr. Philip B. Paty, a surgical oncologist at Memorial Sloan-Kettering Cancer Center, New York, commented in a press briefing held before the symposium.
Ideally, the findings would be tested in a randomized trial, he said; however, “there’s too many factors and too much patient autonomy involved here, so that no one really believes asking people to sign up for a randomized trial where rectal resection is decided by a flip of the coin would ever accrue patients.” Short of that, rigorous prospective studies, such as a phase II trial now open at the center, will provide critical information.
Press briefing moderator Dr. Smitha S. Krishnamurthi of Case Western Reserve University, Cleveland, commented, “These are important findings for patients with rectal cancer because removal of the rectum can result in altered bowel habits or the need for permanent colostomy. This study set the bar very high, comparing the results of nonoperative management to the results seen in patients who had no cancer left under the microscope at the time of surgery, and in this study, the nonoperative management appears to compare favorably.”
“We do need longer follow-up though, to be sure that these patients will have disease-specific survival that equals what is achieved with surgery in the long term,” she cautioned, such as the conventional 5 years of observation patients typically receive. “And then of course a prospective study also would be helpful to see the effects of this approach.”
In the study, Dr. Paty and colleagues identified 73 patients who had a clinical complete response (no cancer detected on physical exam, endoscopy, or imaging) to neoadjuvant therapy of radiation and chemotherapy and—by mutual agreement of physician and patient—had nonoperative management (watchful waiting), consisting initially of follow-up at 3- to 4-month intervals by digital rectal and endoscopic exams and at 6-month intervals by imaging. They used as a comparison group 72 patients who underwent standard total mesorectal excision and had a pathologic complete response (no viable cancer cells found microscopically in the resected tissue).
Results showed that 74% of the patients who did not have surgery had a sustained clinical complete response, with no regrowth of tumor during follow-up, reported Dr. Paty. Among the other 26% whose tumors regrew, most of the recurrences were clinically detectable and all patients had successful resections with clean margins. Overall, 77% of the nonsurgical patients had rectal preservation and 98% had local control.
The nonsurgical group did not differ significantly from the surgical group with respect to 4-year rates of disease-specific survival (91% vs. 96%) and overall survival (91% vs. 95%).
The investigators plan to report quality of life data in the future, Dr. Paty said. “But I think it’s pretty obvious to everyone who’s managed these patients that if you can avoid rectal surgery, the quality of life and particularly bowel function is far superior to those who have had rectal resection.”
Successful nonoperative management hinges critically on careful patient selection, close follow-up, and use of salvage surgery, he stressed. Additionally, “the informed consent process in nonoperative management is extremely important, and I always tell patients that they are taking a slight risk. It’s hard to imagine that not operating is going to have 100% equivalent cancer outcomes as operating. That is hard to believe. So we never sell it as being absolutely as good, but …with good follow-up, the results seem to be very close if not equivalent.”
In his experience, most patients are willing to accept this option. “In fact, I have only had two patients [out of more than 100] decline nonoperative management when I thought they were candidates—both young, both with young children, both not wanting to take even the slightest risk of leaving cancer in the rectal wall or their body,” he commented.
Adoption of nonoperative management has been slow in the United States for a variety of reasons, according to Dr. Paty. “I think the bottom line is that practicing watch and wait, nonoperative management is more difficult for the surgeon. It requires first the judgment that the cancer’s gone. You have to follow the patient longer after radiation; sometimes the complete response will take up to 3 months. And there is also the medical-legal issue of deviating from the standard of care. … So I think it was operationally a difficult thing to do, it didn’t fit with the existing paradigm very well.”
But that is changing as more data roll in. “What’s happened in the last I will say 2-3 years is that there are centers publishing their experience, ours being the largest outside Brazil and the first in North America. Another group in the Netherlands has published a group of about 25 patients,” he explained. “Talking with people at meetings around the world, centers are adopting it, and I think that many leaders in clinical trials in rectal cancer recognize that this option is not only reasonable, but perhaps it’s necessary to inform patients that it is an option.”
AT THE GASTROINTESTINAL CANCERS SYMPOSIUM
Key clinical point: Patients with rectal cancer who have a clinical complete response to neoadjuvant therapy can safely skip surgery.
Major finding: Nonsurgical patients with clinical complete response did not differ significantly from surgical patients with pathologic complete response in terms of 4-year overall survival (91% vs. 95%) and disease-specific survival (91% vs. 96%).
Data source: A retrospective review of 145 patients given neoadjuvant therapy for stage I to III rectal cancer.
Disclosures: Dr. Paty disclosed that he had no relevant conflicts of interest.