User login
‘Soak and smear’ not superior for kids’ atopic dermatitis
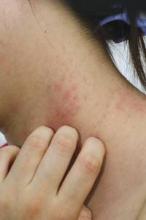
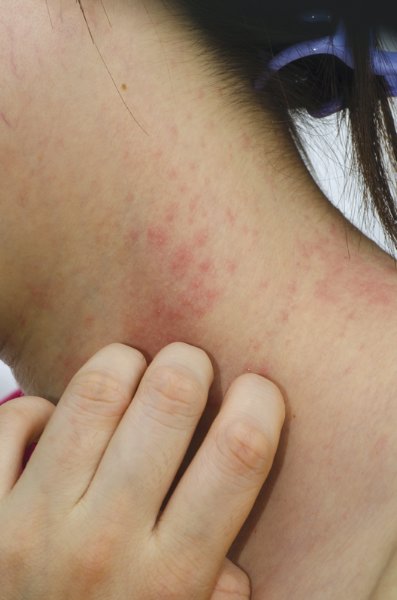
COEUR D’ALENE, IDAHO – Topical corticosteroids applied to the dry skin of children with atopic dermatitis proved as effective for clinical improvement as the soak and smear technique favored by many physicians, according to a randomized, investigator-blinded clinical trial.
"The use of corticosteroid application to prehydrated, wet skin is not more efficacious than corticosteroid application to dry skin in pediatric patients with atopic dermatitis," Dr. Richard J. Antaya reported at the annual meeting of the Society for Pediatric Dermatology. "This study suggests that 2 weeks of using either soak and smear or standard topical corticosteroid application techniques results in considerable improvement in atopic dermatitis severity," he said.
Eczema Area and Severity Index (EASI) scores improved to a similarly impressive extent – close to 85% after 2 weeks – regardless of which application method was used, added Dr. Antaya, professor of dermatology, pediatrics, and nursing and director of pediatric dermatology at Yale University in New Haven, Conn.
The study included 47 patients aged 4 months to 16 years with atopic dermatitis and a mean baseline EASI score of 15.5. All were assigned to 2 weeks of twice-daily topical steroid therapy. Those younger than age 2 years received a prescription for a 1-lb jar of hydrocortisone 2.5% ointment; older patients received 1-lb jars of triamcinolone 0.1% ointment, and, for the more sensitive face and intertriginous areas, hydrocortisone 2.5% ointment. Patients were randomized to twice-daily application of their medication to affected dry skin or to a single daily soak and smear session and one application of the medication to dry skin.
Soak and smear entails a 10-minute soak in lukewarm plain water to boost skin hydration, followed by steroid application to the wet skin. Data from several studies conducted in adults concluded that soak and smear is more effective than was conventional steroid application to dry skin. For example, a retrospective study of 28 adults referred to a tertiary dermatologic center for highly refractory atopic dermatitis or other eczematous dermatoses showed 26 of the 28 were clear or at least 90% improved after several days to 2 weeks of soak and smear sessions (Arch. Dermatol. 2005;141:1556-9).
In Dr. Antaya’s pediatric atopic dermatitis study, assessment of EASI scores was performed by a dermatologist blinded to the treatment arm. The profound and similar improvement in EASI scores in the two treatment groups was accompanied by essentially equal improvements in measures of sleep quality, itch, and overall disease impact.
Also, there was no difference in EASI score improvement between the two groups when patients were stratified according to baseline atopic dermatitis severity.
No differences between the groups were noted in treatment days missed or development of folliculitis. Neither group showed any evidence of hypothalamic-pituitary-adrenal axis suppression, he added.
Dr. Antaya attributed the marked improvement in atopic dermatitis seen in both study groups to the fact that, at the study outset, caregivers received education aimed at alleviating steroid phobia. Also, the 1-lb jars of medication encouraged treatment compliance, he noted.
Dr. Antaya reported having no financial conflicts with regard to this study.
COEUR D’ALENE, IDAHO – Topical corticosteroids applied to the dry skin of children with atopic dermatitis proved as effective for clinical improvement as the soak and smear technique favored by many physicians, according to a randomized, investigator-blinded clinical trial.
"The use of corticosteroid application to prehydrated, wet skin is not more efficacious than corticosteroid application to dry skin in pediatric patients with atopic dermatitis," Dr. Richard J. Antaya reported at the annual meeting of the Society for Pediatric Dermatology. "This study suggests that 2 weeks of using either soak and smear or standard topical corticosteroid application techniques results in considerable improvement in atopic dermatitis severity," he said.
Eczema Area and Severity Index (EASI) scores improved to a similarly impressive extent – close to 85% after 2 weeks – regardless of which application method was used, added Dr. Antaya, professor of dermatology, pediatrics, and nursing and director of pediatric dermatology at Yale University in New Haven, Conn.
The study included 47 patients aged 4 months to 16 years with atopic dermatitis and a mean baseline EASI score of 15.5. All were assigned to 2 weeks of twice-daily topical steroid therapy. Those younger than age 2 years received a prescription for a 1-lb jar of hydrocortisone 2.5% ointment; older patients received 1-lb jars of triamcinolone 0.1% ointment, and, for the more sensitive face and intertriginous areas, hydrocortisone 2.5% ointment. Patients were randomized to twice-daily application of their medication to affected dry skin or to a single daily soak and smear session and one application of the medication to dry skin.
Soak and smear entails a 10-minute soak in lukewarm plain water to boost skin hydration, followed by steroid application to the wet skin. Data from several studies conducted in adults concluded that soak and smear is more effective than was conventional steroid application to dry skin. For example, a retrospective study of 28 adults referred to a tertiary dermatologic center for highly refractory atopic dermatitis or other eczematous dermatoses showed 26 of the 28 were clear or at least 90% improved after several days to 2 weeks of soak and smear sessions (Arch. Dermatol. 2005;141:1556-9).
In Dr. Antaya’s pediatric atopic dermatitis study, assessment of EASI scores was performed by a dermatologist blinded to the treatment arm. The profound and similar improvement in EASI scores in the two treatment groups was accompanied by essentially equal improvements in measures of sleep quality, itch, and overall disease impact.
Also, there was no difference in EASI score improvement between the two groups when patients were stratified according to baseline atopic dermatitis severity.
No differences between the groups were noted in treatment days missed or development of folliculitis. Neither group showed any evidence of hypothalamic-pituitary-adrenal axis suppression, he added.
Dr. Antaya attributed the marked improvement in atopic dermatitis seen in both study groups to the fact that, at the study outset, caregivers received education aimed at alleviating steroid phobia. Also, the 1-lb jars of medication encouraged treatment compliance, he noted.
Dr. Antaya reported having no financial conflicts with regard to this study.
COEUR D’ALENE, IDAHO – Topical corticosteroids applied to the dry skin of children with atopic dermatitis proved as effective for clinical improvement as the soak and smear technique favored by many physicians, according to a randomized, investigator-blinded clinical trial.
"The use of corticosteroid application to prehydrated, wet skin is not more efficacious than corticosteroid application to dry skin in pediatric patients with atopic dermatitis," Dr. Richard J. Antaya reported at the annual meeting of the Society for Pediatric Dermatology. "This study suggests that 2 weeks of using either soak and smear or standard topical corticosteroid application techniques results in considerable improvement in atopic dermatitis severity," he said.
Eczema Area and Severity Index (EASI) scores improved to a similarly impressive extent – close to 85% after 2 weeks – regardless of which application method was used, added Dr. Antaya, professor of dermatology, pediatrics, and nursing and director of pediatric dermatology at Yale University in New Haven, Conn.
The study included 47 patients aged 4 months to 16 years with atopic dermatitis and a mean baseline EASI score of 15.5. All were assigned to 2 weeks of twice-daily topical steroid therapy. Those younger than age 2 years received a prescription for a 1-lb jar of hydrocortisone 2.5% ointment; older patients received 1-lb jars of triamcinolone 0.1% ointment, and, for the more sensitive face and intertriginous areas, hydrocortisone 2.5% ointment. Patients were randomized to twice-daily application of their medication to affected dry skin or to a single daily soak and smear session and one application of the medication to dry skin.
Soak and smear entails a 10-minute soak in lukewarm plain water to boost skin hydration, followed by steroid application to the wet skin. Data from several studies conducted in adults concluded that soak and smear is more effective than was conventional steroid application to dry skin. For example, a retrospective study of 28 adults referred to a tertiary dermatologic center for highly refractory atopic dermatitis or other eczematous dermatoses showed 26 of the 28 were clear or at least 90% improved after several days to 2 weeks of soak and smear sessions (Arch. Dermatol. 2005;141:1556-9).
In Dr. Antaya’s pediatric atopic dermatitis study, assessment of EASI scores was performed by a dermatologist blinded to the treatment arm. The profound and similar improvement in EASI scores in the two treatment groups was accompanied by essentially equal improvements in measures of sleep quality, itch, and overall disease impact.
Also, there was no difference in EASI score improvement between the two groups when patients were stratified according to baseline atopic dermatitis severity.
No differences between the groups were noted in treatment days missed or development of folliculitis. Neither group showed any evidence of hypothalamic-pituitary-adrenal axis suppression, he added.
Dr. Antaya attributed the marked improvement in atopic dermatitis seen in both study groups to the fact that, at the study outset, caregivers received education aimed at alleviating steroid phobia. Also, the 1-lb jars of medication encouraged treatment compliance, he noted.
Dr. Antaya reported having no financial conflicts with regard to this study.
AT THE SPD ANNUAL MEETING
Key clinical point: Physicians can anticipate equally profound improvement in the severity of pediatric atopic dermatitis after 2 weeks of twice-daily topical corticosteroid therapy, regardless of whether the medication is applied to prehydrated or dry skin.
Major finding: After 2 weeks of twice-daily topical corticosteroid therapy, pediatric atopic dermatitis patients showed roughly an 85% improvement in disease severity, regardless of whether the medication was applied to dry skin or using the soak and smear method.
Data source: A 2-week, randomized, investigator-blinded clinical trial of twice-daily topical corticosteroid therapy in 47 children and teens with atopic dermatitis.
Disclosures: The presenter reported having no financial conflicts regarding this study, conducted free of commercial support.
An insider’s look at the 2014 atopic dermatitis guidelines
COEUR D’ALENE, IDAHO – The 2014 American Academy of Dermatology atopic dermatitis guidelines may already need an update, according to the cochair of the guidelines panel.
The guidelines were based upon studies published through 2012. Since then, new evidence has emerged that raises the level of uncertainty regarding several key questions the panel addressed, Dr. Robert Sidbury observed at the annual meeting of the Society for Pediatric Dermatology. Among these questions: To bathe or not to bathe? Will a child outgrow atopic dermatitis?
Serving as cochair of the guidelines committee was both a reassuring and daunting experience, according to Dr. Sidbury, chief of the division of dermatology at Seattle Children’s Hospital.
It was reassuring to note that the committee members, who included 17 atopic dermatitis experts from three countries, were free from financial conflicts as they sifted through the published data for evidence-based recommendations to inform practice. But it was daunting to learn how sketchy the supporting evidence is for some of the conventional wisdom regarding atopic dermatitis management, he explained.
By way of providing what he called "a peek behind the curtains" of the guidelines-development process, Dr. Sidbury highlighted the issue of daily bathing followed by application of emollients and moisturizers. This was among the topics the panel struggled with the most, which might come as a surprise to outsiders who consider this to be standard practice, he noted.
"It seems like a very straightforward thing. Almost everyone in this room, to a person, recommends a daily bath followed by moisturizers, yet when we examined the studies we realized that recommendation isn’t based upon much evidence," he said.
Thus, the panel concluded that bathing is "suggested" for atopic dermatitis patients, while adding that "there is no standard" for the duration or frequency of bathing. The panel rated the strength of their recommendation as C, and the level of evidence as III.
"That’s a fairly weak recommendation based upon fairly week evidence," Dr. Sidbury commented.
Moreover, since publication of the guidelines, two new studies have come forth that address the question of whether bathing plus moisturizers is beneficial in atopic dermatitis. The results conflict with each other, making recommendations even more difficult.
Data from a retrospective study of 75 patients with moderate or severe atopic dermatitis suggested that a daily 15- to 20-minute bath followed by a mid-potency topical steroid and moisturizer was indeed beneficial: 79% of subjects showed marked improvement based on Investigator’s Global Assessment at week 3, and 4% were clear (Dermatitis 2014;25:56-9).
By contrast, data from a prospective trial in which 28 children with atopic dermatitis were randomized to a daily vs. twice-weekly bath followed by appropriate care indicated that, while hydration with emollients was important, bathing frequency wasn’t (Clin. Pediatr. 2014;53:677-81).
"This paper makes me feel better about the guidelines not saying, ‘You should bathe every day,’ although that’s still my own recommendation to patients," Dr. Sidbury said.
This year also has brought two conflicting studies regarding the natural history of atopic dermatitis. A large national Taiwanese population-based cohort study of children diagnosed with atopic dermatitis within the first 2 years of life and followed from birth to age 10 years concluded that 70% of these early-onset patients eventually went into remission. A total of 19% of patients did so within the first year, and 49% in less than 4 years. The median disease duration was 4.2 years (Br. J. Dermatol. 2014;170:130-5).
On the other hand, a report from the 7,157-patient, cross-sectional, longitudinal Pediatric Eczema Elective Registry (PEER) found that by age 20, only 50% of the patients had experienced at least one symptom-free period lasting 6 months or more. The investigators concluded that atopic dermatitis is probably a lifelong disease (JAMA Dermatology 2014;150:593-600).
"That’s a provocative conclusion, and a tough thing to tell a parent," Dr. Sidbury observed. "I offer parents realistic but optimistic counsel. I tell them the tendency toward xerosis, irritancy, and infection will persist – the patient in front of you is never going to want to wear a wool sweater for the rest of their life. But the incessant itch, the need for treatment, the impact on quality of life – which is really the issue at hand – hopefully will not persist."
Since the release earlier this year of the first three of the four sections of the atopic dermatitis guidelines, Dr. Sidbury and the other panelists have received considerable feedback that the guidelines didn’t adequately address the topic of topical steroid addiction.
"Some say we missed the boat in not making coherent recommendations to parents about it. We got some very pointed comments," he conceded.
He noted that a systematic review presented at last year’s International Symposium on Atopic Dermatitis concluded that topical steroid withdrawal is a real phenomenon distinct from other topical steroid side effects. It comes in two rosacea-like variants: an erythroedematous form and a papulopustular form. An atopic dermatitis patient’s report of a burning sensation upon cessation of topical steroid therapy is a red flag.
Despite the occasional missed opportunity in drawing up the first AAD atopic guidelines in 10 years, the process was richly rewarding, Dr. Sidbury said. And although experts will continue to debate the unresolved controversies in atopic dermatitis, for him the most important lesson to emerge from the panel’s comprehensive review of the evidence was strikingly clear: "Time and time again, education trumps all. Education of patients and families leads to the best outcomes. I think that’s an important lesson to take home," he said.
Dr. Sidbury had no financial conflicts to disclose.
COEUR D’ALENE, IDAHO – The 2014 American Academy of Dermatology atopic dermatitis guidelines may already need an update, according to the cochair of the guidelines panel.
The guidelines were based upon studies published through 2012. Since then, new evidence has emerged that raises the level of uncertainty regarding several key questions the panel addressed, Dr. Robert Sidbury observed at the annual meeting of the Society for Pediatric Dermatology. Among these questions: To bathe or not to bathe? Will a child outgrow atopic dermatitis?
Serving as cochair of the guidelines committee was both a reassuring and daunting experience, according to Dr. Sidbury, chief of the division of dermatology at Seattle Children’s Hospital.
It was reassuring to note that the committee members, who included 17 atopic dermatitis experts from three countries, were free from financial conflicts as they sifted through the published data for evidence-based recommendations to inform practice. But it was daunting to learn how sketchy the supporting evidence is for some of the conventional wisdom regarding atopic dermatitis management, he explained.
By way of providing what he called "a peek behind the curtains" of the guidelines-development process, Dr. Sidbury highlighted the issue of daily bathing followed by application of emollients and moisturizers. This was among the topics the panel struggled with the most, which might come as a surprise to outsiders who consider this to be standard practice, he noted.
"It seems like a very straightforward thing. Almost everyone in this room, to a person, recommends a daily bath followed by moisturizers, yet when we examined the studies we realized that recommendation isn’t based upon much evidence," he said.
Thus, the panel concluded that bathing is "suggested" for atopic dermatitis patients, while adding that "there is no standard" for the duration or frequency of bathing. The panel rated the strength of their recommendation as C, and the level of evidence as III.
"That’s a fairly weak recommendation based upon fairly week evidence," Dr. Sidbury commented.
Moreover, since publication of the guidelines, two new studies have come forth that address the question of whether bathing plus moisturizers is beneficial in atopic dermatitis. The results conflict with each other, making recommendations even more difficult.
Data from a retrospective study of 75 patients with moderate or severe atopic dermatitis suggested that a daily 15- to 20-minute bath followed by a mid-potency topical steroid and moisturizer was indeed beneficial: 79% of subjects showed marked improvement based on Investigator’s Global Assessment at week 3, and 4% were clear (Dermatitis 2014;25:56-9).
By contrast, data from a prospective trial in which 28 children with atopic dermatitis were randomized to a daily vs. twice-weekly bath followed by appropriate care indicated that, while hydration with emollients was important, bathing frequency wasn’t (Clin. Pediatr. 2014;53:677-81).
"This paper makes me feel better about the guidelines not saying, ‘You should bathe every day,’ although that’s still my own recommendation to patients," Dr. Sidbury said.
This year also has brought two conflicting studies regarding the natural history of atopic dermatitis. A large national Taiwanese population-based cohort study of children diagnosed with atopic dermatitis within the first 2 years of life and followed from birth to age 10 years concluded that 70% of these early-onset patients eventually went into remission. A total of 19% of patients did so within the first year, and 49% in less than 4 years. The median disease duration was 4.2 years (Br. J. Dermatol. 2014;170:130-5).
On the other hand, a report from the 7,157-patient, cross-sectional, longitudinal Pediatric Eczema Elective Registry (PEER) found that by age 20, only 50% of the patients had experienced at least one symptom-free period lasting 6 months or more. The investigators concluded that atopic dermatitis is probably a lifelong disease (JAMA Dermatology 2014;150:593-600).
"That’s a provocative conclusion, and a tough thing to tell a parent," Dr. Sidbury observed. "I offer parents realistic but optimistic counsel. I tell them the tendency toward xerosis, irritancy, and infection will persist – the patient in front of you is never going to want to wear a wool sweater for the rest of their life. But the incessant itch, the need for treatment, the impact on quality of life – which is really the issue at hand – hopefully will not persist."
Since the release earlier this year of the first three of the four sections of the atopic dermatitis guidelines, Dr. Sidbury and the other panelists have received considerable feedback that the guidelines didn’t adequately address the topic of topical steroid addiction.
"Some say we missed the boat in not making coherent recommendations to parents about it. We got some very pointed comments," he conceded.
He noted that a systematic review presented at last year’s International Symposium on Atopic Dermatitis concluded that topical steroid withdrawal is a real phenomenon distinct from other topical steroid side effects. It comes in two rosacea-like variants: an erythroedematous form and a papulopustular form. An atopic dermatitis patient’s report of a burning sensation upon cessation of topical steroid therapy is a red flag.
Despite the occasional missed opportunity in drawing up the first AAD atopic guidelines in 10 years, the process was richly rewarding, Dr. Sidbury said. And although experts will continue to debate the unresolved controversies in atopic dermatitis, for him the most important lesson to emerge from the panel’s comprehensive review of the evidence was strikingly clear: "Time and time again, education trumps all. Education of patients and families leads to the best outcomes. I think that’s an important lesson to take home," he said.
Dr. Sidbury had no financial conflicts to disclose.
COEUR D’ALENE, IDAHO – The 2014 American Academy of Dermatology atopic dermatitis guidelines may already need an update, according to the cochair of the guidelines panel.
The guidelines were based upon studies published through 2012. Since then, new evidence has emerged that raises the level of uncertainty regarding several key questions the panel addressed, Dr. Robert Sidbury observed at the annual meeting of the Society for Pediatric Dermatology. Among these questions: To bathe or not to bathe? Will a child outgrow atopic dermatitis?
Serving as cochair of the guidelines committee was both a reassuring and daunting experience, according to Dr. Sidbury, chief of the division of dermatology at Seattle Children’s Hospital.
It was reassuring to note that the committee members, who included 17 atopic dermatitis experts from three countries, were free from financial conflicts as they sifted through the published data for evidence-based recommendations to inform practice. But it was daunting to learn how sketchy the supporting evidence is for some of the conventional wisdom regarding atopic dermatitis management, he explained.
By way of providing what he called "a peek behind the curtains" of the guidelines-development process, Dr. Sidbury highlighted the issue of daily bathing followed by application of emollients and moisturizers. This was among the topics the panel struggled with the most, which might come as a surprise to outsiders who consider this to be standard practice, he noted.
"It seems like a very straightforward thing. Almost everyone in this room, to a person, recommends a daily bath followed by moisturizers, yet when we examined the studies we realized that recommendation isn’t based upon much evidence," he said.
Thus, the panel concluded that bathing is "suggested" for atopic dermatitis patients, while adding that "there is no standard" for the duration or frequency of bathing. The panel rated the strength of their recommendation as C, and the level of evidence as III.
"That’s a fairly weak recommendation based upon fairly week evidence," Dr. Sidbury commented.
Moreover, since publication of the guidelines, two new studies have come forth that address the question of whether bathing plus moisturizers is beneficial in atopic dermatitis. The results conflict with each other, making recommendations even more difficult.
Data from a retrospective study of 75 patients with moderate or severe atopic dermatitis suggested that a daily 15- to 20-minute bath followed by a mid-potency topical steroid and moisturizer was indeed beneficial: 79% of subjects showed marked improvement based on Investigator’s Global Assessment at week 3, and 4% were clear (Dermatitis 2014;25:56-9).
By contrast, data from a prospective trial in which 28 children with atopic dermatitis were randomized to a daily vs. twice-weekly bath followed by appropriate care indicated that, while hydration with emollients was important, bathing frequency wasn’t (Clin. Pediatr. 2014;53:677-81).
"This paper makes me feel better about the guidelines not saying, ‘You should bathe every day,’ although that’s still my own recommendation to patients," Dr. Sidbury said.
This year also has brought two conflicting studies regarding the natural history of atopic dermatitis. A large national Taiwanese population-based cohort study of children diagnosed with atopic dermatitis within the first 2 years of life and followed from birth to age 10 years concluded that 70% of these early-onset patients eventually went into remission. A total of 19% of patients did so within the first year, and 49% in less than 4 years. The median disease duration was 4.2 years (Br. J. Dermatol. 2014;170:130-5).
On the other hand, a report from the 7,157-patient, cross-sectional, longitudinal Pediatric Eczema Elective Registry (PEER) found that by age 20, only 50% of the patients had experienced at least one symptom-free period lasting 6 months or more. The investigators concluded that atopic dermatitis is probably a lifelong disease (JAMA Dermatology 2014;150:593-600).
"That’s a provocative conclusion, and a tough thing to tell a parent," Dr. Sidbury observed. "I offer parents realistic but optimistic counsel. I tell them the tendency toward xerosis, irritancy, and infection will persist – the patient in front of you is never going to want to wear a wool sweater for the rest of their life. But the incessant itch, the need for treatment, the impact on quality of life – which is really the issue at hand – hopefully will not persist."
Since the release earlier this year of the first three of the four sections of the atopic dermatitis guidelines, Dr. Sidbury and the other panelists have received considerable feedback that the guidelines didn’t adequately address the topic of topical steroid addiction.
"Some say we missed the boat in not making coherent recommendations to parents about it. We got some very pointed comments," he conceded.
He noted that a systematic review presented at last year’s International Symposium on Atopic Dermatitis concluded that topical steroid withdrawal is a real phenomenon distinct from other topical steroid side effects. It comes in two rosacea-like variants: an erythroedematous form and a papulopustular form. An atopic dermatitis patient’s report of a burning sensation upon cessation of topical steroid therapy is a red flag.
Despite the occasional missed opportunity in drawing up the first AAD atopic guidelines in 10 years, the process was richly rewarding, Dr. Sidbury said. And although experts will continue to debate the unresolved controversies in atopic dermatitis, for him the most important lesson to emerge from the panel’s comprehensive review of the evidence was strikingly clear: "Time and time again, education trumps all. Education of patients and families leads to the best outcomes. I think that’s an important lesson to take home," he said.
Dr. Sidbury had no financial conflicts to disclose.
EXPERT OPINION FROM THE SPD ANNUAL MEETING
More vitamin D didn’t beat placebo for kids’ atopic dermatitis
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
COEUR D’ALENE, IDAHO – Vitamin D insufficiency or outright deficiency is extremely common among pediatric atopic dermatitis patients, and it correlates with worse skin disease severity.
Unfortunately, vitamin D supplementation in such patients didn’t outperform placebo in improving their atopic dermatitis severity scores in a double-blind, randomized, prospective clinical trial, Dr. Irene Lara-Corrales reported at the annual meeting of the Society for Pediatric Dermatology.
In the first phase of this two-part study, 77 atopic dermatitis patients aged 1-18 years, with a mean age of 7.4 years and free of potentially confounding comorbid medical conditions, had their serum vitamin D level checked. Only 27 patients, or 35%, had a normal level, defined as greater than 72.5 nmol/L, or 30 ng/mL.
Thirty-six patients were categorized as vitamin D insufficient based upon a serum level of 32.5-72.5 nmol/L, or 15-29 ng/mL. The remaining 14 patients were vitamin D deficient, with a serum level below 32.5 nmol/L or 15 ng/mL, according to Dr. Lara-Corrales of the Hospital for Sick Children in Toronto.
Mean baseline atopic dermatitis severity scores using the SCORAD (Scoring Atopic Disease) system were 19.0 in the group with normal serum vitamin D and significantly worse at 28.8 in those who were vitamin D insufficient and 24.6 in patients who were vitamin D deficient.
In phase II of the study, 41 patients from phase I who had low vitamin D levels were randomized and double blinded to either 2,000 IU of vitamin D administered as cholecalciferol drops or to placebo drops for 3 months. The study hypothesis was that SCORAD ratings would improve markedly with vitamin D supplementation, based upon mounting evidence that serum vitamin D plays a key role in skin immune function.
But the hypothesis did not prevail. Although serum vitamin D levels did indeed increase significantly in response to 3 months of daily oral vitamin D supplementation, and recipients showed a hefty mean 15.35-point improvement in SCORAD scores, the placebo-treated controls demonstrated a near-identical mean 15.13-point SCORAD improvement as well.
Dr. Lara-Corrales noted that the study was completed only recently and that substantial collected data have yet to be analyzed. That includes information on sun exposure, nutritional intake, sunscreen use, and exposure to breast milk in early childhood, which may prove useful in interpreting the study results.
The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
AT THE SPD ANNUAL MEETING
Key clinical point: There may be more behind atopic dermatitis severity than just vitamin D deficiency.
Major finding: Correcting the atopic dermatitis patients’ low serum vitamin D with 2,000 IU of cholecalciferol for 3 months improved their atopic dermatitis severity scores, but only to the same extent as in placebo-treated controls.
Data source: This was a randomized, double-blind, single-center trial in which 40 atopic dermatitis patients aged 1-18 years with vitamin D insufficiency or deficiency were placed on vitamin D supplementation or placebo for 3 months.
Disclosures: The Society for Pediatric Dermatology and the Canadian Dermatology Foundation supported the study. Dr. Lara-Corrales reported having no financial conflicts.
Summer care for atopic skin
Summer months can be dreadful for patients with atopic dermatitis. The chlorine, heat, and humidity can lead to flares. Furthermore, noncompliance with skin care regimens because of changing summer routines, travel, and the use of hotel products can exacerbate even the calmest skin disease.
Share these tips with your patients to help them keep their atopic skin under control in the summer heat, and stop flares before they start.
• Rinse the skin well after swimming. Chlorine and saltwater can dry out the skin. Showers after swimming in chlorinated pools can help retain the skin’s natural oils.
• Avoid hot tubs. Cracks and fissures in atopic skin can become infected in hot tubs with Staphylococcus and Pseudomonas. Advise your atopic patients to avoid hot tubs, even if they claim the tubs have been cleaned.
• Bring your own products. Many soaps and shower gels available in hotels and resorts are extremely drying, and may contain ingredients that could irritate atopic skin.
• Don’t switch from thick creams to thin lotions just because it is summer. Remind your patients that a thin lotion does not provide the same occlusive and humectant properties as thicker creams, although they are not as easy to apply, and can feel thick and sticky on the skin with humidity.
• In case of an active eczema flare, topical steroids should be used and sun exposure should be avoided. Topical steroids are the most effective treatment when used correctly. However, any occurrence of hypopigmentation as a result of their use becomes more evident if the skin tans around the area of treatment.
• Wear physical sunscreen. This seems obvious, but most chemical blockers – even the formulations made for babies – can burn on cracked, inflamed skin. Instead, stress to your patients that they use a physical blocker made of pure titanium dioxide or zinc oxide on inflamed skin.
• Oral steroids and sun do not mix. Oral steroids can be potent photosensitizers. If they are needed, UV exposure should be avoided.
Dr. Talakoub and Dr. Wesley are co-contributors to a monthly Aesthetic Dermatology column in Skin & Allergy News. Dr. Talakoub is in private practice at McLean (Va.) Dermatology Center. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
Summer months can be dreadful for patients with atopic dermatitis. The chlorine, heat, and humidity can lead to flares. Furthermore, noncompliance with skin care regimens because of changing summer routines, travel, and the use of hotel products can exacerbate even the calmest skin disease.
Share these tips with your patients to help them keep their atopic skin under control in the summer heat, and stop flares before they start.
• Rinse the skin well after swimming. Chlorine and saltwater can dry out the skin. Showers after swimming in chlorinated pools can help retain the skin’s natural oils.
• Avoid hot tubs. Cracks and fissures in atopic skin can become infected in hot tubs with Staphylococcus and Pseudomonas. Advise your atopic patients to avoid hot tubs, even if they claim the tubs have been cleaned.
• Bring your own products. Many soaps and shower gels available in hotels and resorts are extremely drying, and may contain ingredients that could irritate atopic skin.
• Don’t switch from thick creams to thin lotions just because it is summer. Remind your patients that a thin lotion does not provide the same occlusive and humectant properties as thicker creams, although they are not as easy to apply, and can feel thick and sticky on the skin with humidity.
• In case of an active eczema flare, topical steroids should be used and sun exposure should be avoided. Topical steroids are the most effective treatment when used correctly. However, any occurrence of hypopigmentation as a result of their use becomes more evident if the skin tans around the area of treatment.
• Wear physical sunscreen. This seems obvious, but most chemical blockers – even the formulations made for babies – can burn on cracked, inflamed skin. Instead, stress to your patients that they use a physical blocker made of pure titanium dioxide or zinc oxide on inflamed skin.
• Oral steroids and sun do not mix. Oral steroids can be potent photosensitizers. If they are needed, UV exposure should be avoided.
Dr. Talakoub and Dr. Wesley are co-contributors to a monthly Aesthetic Dermatology column in Skin & Allergy News. Dr. Talakoub is in private practice at McLean (Va.) Dermatology Center. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
Summer months can be dreadful for patients with atopic dermatitis. The chlorine, heat, and humidity can lead to flares. Furthermore, noncompliance with skin care regimens because of changing summer routines, travel, and the use of hotel products can exacerbate even the calmest skin disease.
Share these tips with your patients to help them keep their atopic skin under control in the summer heat, and stop flares before they start.
• Rinse the skin well after swimming. Chlorine and saltwater can dry out the skin. Showers after swimming in chlorinated pools can help retain the skin’s natural oils.
• Avoid hot tubs. Cracks and fissures in atopic skin can become infected in hot tubs with Staphylococcus and Pseudomonas. Advise your atopic patients to avoid hot tubs, even if they claim the tubs have been cleaned.
• Bring your own products. Many soaps and shower gels available in hotels and resorts are extremely drying, and may contain ingredients that could irritate atopic skin.
• Don’t switch from thick creams to thin lotions just because it is summer. Remind your patients that a thin lotion does not provide the same occlusive and humectant properties as thicker creams, although they are not as easy to apply, and can feel thick and sticky on the skin with humidity.
• In case of an active eczema flare, topical steroids should be used and sun exposure should be avoided. Topical steroids are the most effective treatment when used correctly. However, any occurrence of hypopigmentation as a result of their use becomes more evident if the skin tans around the area of treatment.
• Wear physical sunscreen. This seems obvious, but most chemical blockers – even the formulations made for babies – can burn on cracked, inflamed skin. Instead, stress to your patients that they use a physical blocker made of pure titanium dioxide or zinc oxide on inflamed skin.
• Oral steroids and sun do not mix. Oral steroids can be potent photosensitizers. If they are needed, UV exposure should be avoided.
Dr. Talakoub and Dr. Wesley are co-contributors to a monthly Aesthetic Dermatology column in Skin & Allergy News. Dr. Talakoub is in private practice at McLean (Va.) Dermatology Center. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
Dupilumab produces dramatic improvement in atopic dermatitis
The monoclonal antibody dupilumab, which blocks the action of the type 2 helper T-cell (Th2) cytokines interleukin-4 and interleukin-13, produced rapid and significant improvement in all signs, symptoms, and biomarkers of atopic dermatitis in four industry-sponsored randomized clinical trials. The data were reported online July 9 in the New England Journal of Medicine.
In particular, once-weekly subcutaneous injections of dupilumab quickly yielded substantial reductions in pruritus, "a major contributor to reduced quality of life experienced by patients with atopic dermatitis," said Dr. Lisa A. Beck of the department of dermatology, University of Rochester (N.Y.) Medical Center, and her associates.
These findings "extend the potential benefit of this new biologic therapy beyond asthma to a second atopic disorder" and suggest that the treatment may be beneficial for other atopic disorders as well, they noted (N. Engl. J. Med. 2014;371:130-9 [doi:10.1056/NEJMoa1314768]).
The four double-blind, placebo-controlled trials involved a total of 207 adults aged 18 years and older in the United States and Europe who had poorly controlled moderate to severe atopic dermatitis of at least 2 years’ duration. Two studies assessed 4 weeks of monotherapy with dupilumab, one assessed 12 weeks of monotherapy, and the fourth assessed 4 weeks of dupilumab added to topical glucocorticoids.
In all four studies, dupilumab induced rapid, dose-dependent improvements in an investigator’s global assessment score, a measure of the percentage of affected body-surface area, scores on the Eczema Area and Severity Index, and scores on both the 5-D pruritus scale and an additional numerical-rating scale of pruritus. In the 12-week trial, for example, 85% of patients given dupilumab achieved at least a 50% reduction in EASI scores, and mean scores on the pruritus rating scale declined by 56%.
Adverse events in all study groups were generally mild and transient: Nasopharyngitis and headache were reported more frequently in the active-treatment groups than in the placebo groups. However, serious adverse events and adverse events leading to treatment withdrawal – chiefly skin infections and exacerbations of the underlying dermatitis – were more common in the placebo groups and were related to lack of efficacy.
This study was funded by Regeneron Pharmaceuticals and Sanofi, which also performed data analysis and assisted with writing the report. Dr. Beck reported other ties to Regeneron; her associates reported ties to numerous industry sources.
The monoclonal antibody dupilumab, which blocks the action of the type 2 helper T-cell (Th2) cytokines interleukin-4 and interleukin-13, produced rapid and significant improvement in all signs, symptoms, and biomarkers of atopic dermatitis in four industry-sponsored randomized clinical trials. The data were reported online July 9 in the New England Journal of Medicine.
In particular, once-weekly subcutaneous injections of dupilumab quickly yielded substantial reductions in pruritus, "a major contributor to reduced quality of life experienced by patients with atopic dermatitis," said Dr. Lisa A. Beck of the department of dermatology, University of Rochester (N.Y.) Medical Center, and her associates.
These findings "extend the potential benefit of this new biologic therapy beyond asthma to a second atopic disorder" and suggest that the treatment may be beneficial for other atopic disorders as well, they noted (N. Engl. J. Med. 2014;371:130-9 [doi:10.1056/NEJMoa1314768]).
The four double-blind, placebo-controlled trials involved a total of 207 adults aged 18 years and older in the United States and Europe who had poorly controlled moderate to severe atopic dermatitis of at least 2 years’ duration. Two studies assessed 4 weeks of monotherapy with dupilumab, one assessed 12 weeks of monotherapy, and the fourth assessed 4 weeks of dupilumab added to topical glucocorticoids.
In all four studies, dupilumab induced rapid, dose-dependent improvements in an investigator’s global assessment score, a measure of the percentage of affected body-surface area, scores on the Eczema Area and Severity Index, and scores on both the 5-D pruritus scale and an additional numerical-rating scale of pruritus. In the 12-week trial, for example, 85% of patients given dupilumab achieved at least a 50% reduction in EASI scores, and mean scores on the pruritus rating scale declined by 56%.
Adverse events in all study groups were generally mild and transient: Nasopharyngitis and headache were reported more frequently in the active-treatment groups than in the placebo groups. However, serious adverse events and adverse events leading to treatment withdrawal – chiefly skin infections and exacerbations of the underlying dermatitis – were more common in the placebo groups and were related to lack of efficacy.
This study was funded by Regeneron Pharmaceuticals and Sanofi, which also performed data analysis and assisted with writing the report. Dr. Beck reported other ties to Regeneron; her associates reported ties to numerous industry sources.
The monoclonal antibody dupilumab, which blocks the action of the type 2 helper T-cell (Th2) cytokines interleukin-4 and interleukin-13, produced rapid and significant improvement in all signs, symptoms, and biomarkers of atopic dermatitis in four industry-sponsored randomized clinical trials. The data were reported online July 9 in the New England Journal of Medicine.
In particular, once-weekly subcutaneous injections of dupilumab quickly yielded substantial reductions in pruritus, "a major contributor to reduced quality of life experienced by patients with atopic dermatitis," said Dr. Lisa A. Beck of the department of dermatology, University of Rochester (N.Y.) Medical Center, and her associates.
These findings "extend the potential benefit of this new biologic therapy beyond asthma to a second atopic disorder" and suggest that the treatment may be beneficial for other atopic disorders as well, they noted (N. Engl. J. Med. 2014;371:130-9 [doi:10.1056/NEJMoa1314768]).
The four double-blind, placebo-controlled trials involved a total of 207 adults aged 18 years and older in the United States and Europe who had poorly controlled moderate to severe atopic dermatitis of at least 2 years’ duration. Two studies assessed 4 weeks of monotherapy with dupilumab, one assessed 12 weeks of monotherapy, and the fourth assessed 4 weeks of dupilumab added to topical glucocorticoids.
In all four studies, dupilumab induced rapid, dose-dependent improvements in an investigator’s global assessment score, a measure of the percentage of affected body-surface area, scores on the Eczema Area and Severity Index, and scores on both the 5-D pruritus scale and an additional numerical-rating scale of pruritus. In the 12-week trial, for example, 85% of patients given dupilumab achieved at least a 50% reduction in EASI scores, and mean scores on the pruritus rating scale declined by 56%.
Adverse events in all study groups were generally mild and transient: Nasopharyngitis and headache were reported more frequently in the active-treatment groups than in the placebo groups. However, serious adverse events and adverse events leading to treatment withdrawal – chiefly skin infections and exacerbations of the underlying dermatitis – were more common in the placebo groups and were related to lack of efficacy.
This study was funded by Regeneron Pharmaceuticals and Sanofi, which also performed data analysis and assisted with writing the report. Dr. Beck reported other ties to Regeneron; her associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: By reducing pruritus, dupilumab could improve quality of life.
Major finding: In a 12-week trial, 85% of patients given dupilumab achieved at least a 50% reduction in EASI scores, and the mean scores on the pruritus rating scale declined by 56%.
Data source: Four separate double-blind randomized trials comparing 4 weeks or 12 weeks of weekly subcutaneous injections of dupilumab against placebo in 207 American and European adults with poorly controlled moderate to severe atopic dermatitis.
Disclosures: This study was funded by Regeneron Pharmaceuticals and Sanofi, which also performed data analysis and assisted with writing the report. Dr. Beck reported other ties to Regeneron; her associates reported ties to numerous industry sources.
Eczema linked to increased suicidal thoughts in teens
Adolescents with eczema were significantly more likely to report suicidal ideation than those who did not have eczema, based on data from a population-based study of more than 3,000 adolescents. The findings were published in the Journal of Investigative Dermatology.
Data from previous studies have shown an increased suicide risk in patients with skin disorders, but the impact of eczema on adolescents in particular has not been well studied, wrote Dr. Jon A. Halvorsen of the department of dermatology at the University of Oslo and his colleagues (J. Invest. Dermatol. 2014;134:1847-54).
The researchers reviewed survey data from 3,556 adolescents aged 18-19 years collected as part of the Oslo section of the Youths 2004 study.
The overall prevalence of eczema was 10% (12% in girls and 7% in boys). The overall incidence of suicidal ideation was 11%, but 16% of adolescents with eczema reported suicidal ideation vs. 9% of those with no eczema (odds ratio, 1.87). The association was even stronger in adolescents who reported eczema with itching (OR, 3.57), compared with those who did not report itching (OR, 1.06).
"The risk of suicidal ideation increased nearly fourfold in this group," the researchers noted. "Our data suggest that itch is a major predictor of psychological problems and may be a greater risk factor for these problems than chronic eczema without itch."
In addition, adolescents with eczema were significantly more likely than those without eczema to report mental health problems (assessed by the Strengths and Difficulties Questionnaire) and mental distress (assessed by the Hopkins Symptom Checklist 10) and these associations were even more significant in adolescents who reported eczema with itch, compared with those who had eczema without itch.
The study was limited by the use of self-reports, but its strengths include a large sample size with high participation, the researchers said. The results suggest that "both treatment of eczema and psychological support in this high-risk group may help to reduce suicide and mental health problems in this vulnerable population," they added.
The researchers reported no financial conflicts.
Adolescents with eczema were significantly more likely to report suicidal ideation than those who did not have eczema, based on data from a population-based study of more than 3,000 adolescents. The findings were published in the Journal of Investigative Dermatology.
Data from previous studies have shown an increased suicide risk in patients with skin disorders, but the impact of eczema on adolescents in particular has not been well studied, wrote Dr. Jon A. Halvorsen of the department of dermatology at the University of Oslo and his colleagues (J. Invest. Dermatol. 2014;134:1847-54).
The researchers reviewed survey data from 3,556 adolescents aged 18-19 years collected as part of the Oslo section of the Youths 2004 study.
The overall prevalence of eczema was 10% (12% in girls and 7% in boys). The overall incidence of suicidal ideation was 11%, but 16% of adolescents with eczema reported suicidal ideation vs. 9% of those with no eczema (odds ratio, 1.87). The association was even stronger in adolescents who reported eczema with itching (OR, 3.57), compared with those who did not report itching (OR, 1.06).
"The risk of suicidal ideation increased nearly fourfold in this group," the researchers noted. "Our data suggest that itch is a major predictor of psychological problems and may be a greater risk factor for these problems than chronic eczema without itch."
In addition, adolescents with eczema were significantly more likely than those without eczema to report mental health problems (assessed by the Strengths and Difficulties Questionnaire) and mental distress (assessed by the Hopkins Symptom Checklist 10) and these associations were even more significant in adolescents who reported eczema with itch, compared with those who had eczema without itch.
The study was limited by the use of self-reports, but its strengths include a large sample size with high participation, the researchers said. The results suggest that "both treatment of eczema and psychological support in this high-risk group may help to reduce suicide and mental health problems in this vulnerable population," they added.
The researchers reported no financial conflicts.
Adolescents with eczema were significantly more likely to report suicidal ideation than those who did not have eczema, based on data from a population-based study of more than 3,000 adolescents. The findings were published in the Journal of Investigative Dermatology.
Data from previous studies have shown an increased suicide risk in patients with skin disorders, but the impact of eczema on adolescents in particular has not been well studied, wrote Dr. Jon A. Halvorsen of the department of dermatology at the University of Oslo and his colleagues (J. Invest. Dermatol. 2014;134:1847-54).
The researchers reviewed survey data from 3,556 adolescents aged 18-19 years collected as part of the Oslo section of the Youths 2004 study.
The overall prevalence of eczema was 10% (12% in girls and 7% in boys). The overall incidence of suicidal ideation was 11%, but 16% of adolescents with eczema reported suicidal ideation vs. 9% of those with no eczema (odds ratio, 1.87). The association was even stronger in adolescents who reported eczema with itching (OR, 3.57), compared with those who did not report itching (OR, 1.06).
"The risk of suicidal ideation increased nearly fourfold in this group," the researchers noted. "Our data suggest that itch is a major predictor of psychological problems and may be a greater risk factor for these problems than chronic eczema without itch."
In addition, adolescents with eczema were significantly more likely than those without eczema to report mental health problems (assessed by the Strengths and Difficulties Questionnaire) and mental distress (assessed by the Hopkins Symptom Checklist 10) and these associations were even more significant in adolescents who reported eczema with itch, compared with those who had eczema without itch.
The study was limited by the use of self-reports, but its strengths include a large sample size with high participation, the researchers said. The results suggest that "both treatment of eczema and psychological support in this high-risk group may help to reduce suicide and mental health problems in this vulnerable population," they added.
The researchers reported no financial conflicts.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Key clinical point: Treating eczema and providing psychological support might help reduce suicidal ideation in vulnerable adolescent populations.
Major finding: Sixteen percent of adolescents with eczema reported suicidal ideation, compared with 9% of those without eczema (odds ratio, 1.87), and the association was even stronger in those reporting itching (OR, 3.57) compared with those without itching (OR, 1.06).
Data source: A population-based study of 3,556 adolescents aged 18-19 years.
Disclosures: The researchers reported no financial conflicts.
Hypochlorite wash may help pediatric atopic dermatitis
DENVER – A sodium hypochlorite wash significantly improved symptoms and quality of life for children who had atopic dermatitis with bacterial colonization, in a small study.
Staphylococcus aureus colonization occurs in the lesional and nonlesional skin of many atopic dermatitis patients, and bleach baths at a concentration of approximately 0.005% sodium hypochlorite are a common treatment, but not always an easy or convenient one, Dr. Benjamin R. Bohaty noted in a poster at the annual meeting of the American Academy of Dermatology.
In an open-label study, Dr. Bohaty of the University of Texas Health Science Center in Houston and his colleagues tested a gel cleanser containing a dilute concentration of 0.006% sodium hypochlorite wash that could be used in the bath or shower. The product is designed to be lathered onto the skin and rinsed off after 1-2 minutes. The study population included 40 children, average age 9 years, with a diagnosis of moderate to severe atopic dermatitis and S. aureus colonization. The children were instructed to use the wash once daily for 6 weeks, and they were assessed at three office visits during this period.
At 2 weeks and 6 weeks, the patients had improved an average of 34% and 44% from baseline, respectively, on the Eczema Area and Severity Index, and 23% and 34% from baseline on the Investigator’s Global Assessment scale score.
The average improvement in pruritus visual analog scale score was 31% at 2 weeks and 37% at 6 weeks.
Quality of life measures also improved during the study period. The average change in the Children’s Dermatology Life Quality Index was 41% at 2 weeks, dropping to 32% at 6 weeks, but both were significant improvements from baseline. The average improvement in the Family Dermatology Life Quality Index score was 13% at 2 weeks and 43% at 6 weeks, compared with baseline.
The significant decreases and lack of reported adverse events suggest that the wash is "a simple alternative to bleach baths," Dr. Bohaty reported.
The product is available in the United States under the label CLn Skin Care, a trademark of TopMD Skin Care, which funded the trial through research grants to the universities of the investigators.
DENVER – A sodium hypochlorite wash significantly improved symptoms and quality of life for children who had atopic dermatitis with bacterial colonization, in a small study.
Staphylococcus aureus colonization occurs in the lesional and nonlesional skin of many atopic dermatitis patients, and bleach baths at a concentration of approximately 0.005% sodium hypochlorite are a common treatment, but not always an easy or convenient one, Dr. Benjamin R. Bohaty noted in a poster at the annual meeting of the American Academy of Dermatology.
In an open-label study, Dr. Bohaty of the University of Texas Health Science Center in Houston and his colleagues tested a gel cleanser containing a dilute concentration of 0.006% sodium hypochlorite wash that could be used in the bath or shower. The product is designed to be lathered onto the skin and rinsed off after 1-2 minutes. The study population included 40 children, average age 9 years, with a diagnosis of moderate to severe atopic dermatitis and S. aureus colonization. The children were instructed to use the wash once daily for 6 weeks, and they were assessed at three office visits during this period.
At 2 weeks and 6 weeks, the patients had improved an average of 34% and 44% from baseline, respectively, on the Eczema Area and Severity Index, and 23% and 34% from baseline on the Investigator’s Global Assessment scale score.
The average improvement in pruritus visual analog scale score was 31% at 2 weeks and 37% at 6 weeks.
Quality of life measures also improved during the study period. The average change in the Children’s Dermatology Life Quality Index was 41% at 2 weeks, dropping to 32% at 6 weeks, but both were significant improvements from baseline. The average improvement in the Family Dermatology Life Quality Index score was 13% at 2 weeks and 43% at 6 weeks, compared with baseline.
The significant decreases and lack of reported adverse events suggest that the wash is "a simple alternative to bleach baths," Dr. Bohaty reported.
The product is available in the United States under the label CLn Skin Care, a trademark of TopMD Skin Care, which funded the trial through research grants to the universities of the investigators.
DENVER – A sodium hypochlorite wash significantly improved symptoms and quality of life for children who had atopic dermatitis with bacterial colonization, in a small study.
Staphylococcus aureus colonization occurs in the lesional and nonlesional skin of many atopic dermatitis patients, and bleach baths at a concentration of approximately 0.005% sodium hypochlorite are a common treatment, but not always an easy or convenient one, Dr. Benjamin R. Bohaty noted in a poster at the annual meeting of the American Academy of Dermatology.
In an open-label study, Dr. Bohaty of the University of Texas Health Science Center in Houston and his colleagues tested a gel cleanser containing a dilute concentration of 0.006% sodium hypochlorite wash that could be used in the bath or shower. The product is designed to be lathered onto the skin and rinsed off after 1-2 minutes. The study population included 40 children, average age 9 years, with a diagnosis of moderate to severe atopic dermatitis and S. aureus colonization. The children were instructed to use the wash once daily for 6 weeks, and they were assessed at three office visits during this period.
At 2 weeks and 6 weeks, the patients had improved an average of 34% and 44% from baseline, respectively, on the Eczema Area and Severity Index, and 23% and 34% from baseline on the Investigator’s Global Assessment scale score.
The average improvement in pruritus visual analog scale score was 31% at 2 weeks and 37% at 6 weeks.
Quality of life measures also improved during the study period. The average change in the Children’s Dermatology Life Quality Index was 41% at 2 weeks, dropping to 32% at 6 weeks, but both were significant improvements from baseline. The average improvement in the Family Dermatology Life Quality Index score was 13% at 2 weeks and 43% at 6 weeks, compared with baseline.
The significant decreases and lack of reported adverse events suggest that the wash is "a simple alternative to bleach baths," Dr. Bohaty reported.
The product is available in the United States under the label CLn Skin Care, a trademark of TopMD Skin Care, which funded the trial through research grants to the universities of the investigators.
FROM THE AAD ANNUAL MEETING
Key clinical point: A gel cleanser appeared to be a potentially effective and convenient alternative to bleach baths for children with atopic dermatitis.
Major finding: A gel cleanser with 0.006% sodium hypochlorite improved symptoms and quality of life by at least 20% from baseline in children with moderate to severe atopic dermatitis.
Data source: An open label study of 40 children, average age 9 years.
Disclosures: The product is available in the United States under the label CLn Skin Care, a trademark of TopMD Skin Care, which funded the trial through research grants to the universities of the investigators.
Desonide hydrogel improved itching in atopic dermatitis
Twice-daily application of 0.05% desonide hydrogel significantly improved clinical symptoms of itching and quality of life scores in 100% of patients with mild to moderate atopic dermatitis after 7 days in a small preliminary study.
"Increasingly, researchers and clinicians recognize that disrupted barrier function contributes not only to the xerotic and pruritic manifestations of AD, but also to the inflammatory cascade that underlies the disease," wrote Dr. Leon Kircik of Indiana University, Indianapolis.
Data from previous studies have shown the effectiveness of desonide hydrogel 0.05% for atopic dermatitis in children, but few studies have examined the effectiveness of the gel on itching in particular. In this study, patients applied the gel twice daily, with assessments at baseline, day 3, and day 7 (J. Drugs Dermatol. 2014;13:725-8).
At day 7, all patients achieved the primary endpoint of at least a 50% reduction in pruritus. The average Investigator’s Global Assessment (IGA) scale score was .55 (down from 2.35 at baseline), which translated to a 76% improvement from baseline. The visual analog score decreased by an average of 6.4 points, for an average reduction of 85%. Statistically significant improvements from baseline in both IGA and visual analog scores also were noted at day 3, when the average IGA score improved 27% from baseline, and the mean visual analog score showed a 53% reduction.
The study included 20 atopic dermatitis patients ranging in age from 8 to 68 years, with an average age of 25 years; 60% were black, 40% were white, and 75% were female.
The results suggest that hydrogel is an appropriate option to relieve itchiness in AD patients, and a large, randomized, double-blind controlled trial would be helpful to further study effectiveness, Dr. Kircik said.
Dr. Kircik disclosed receiving funding as an investigator, consultant, or speaker for Bayer Dermatology, manufacturer of the product tested.
Twice-daily application of 0.05% desonide hydrogel significantly improved clinical symptoms of itching and quality of life scores in 100% of patients with mild to moderate atopic dermatitis after 7 days in a small preliminary study.
"Increasingly, researchers and clinicians recognize that disrupted barrier function contributes not only to the xerotic and pruritic manifestations of AD, but also to the inflammatory cascade that underlies the disease," wrote Dr. Leon Kircik of Indiana University, Indianapolis.
Data from previous studies have shown the effectiveness of desonide hydrogel 0.05% for atopic dermatitis in children, but few studies have examined the effectiveness of the gel on itching in particular. In this study, patients applied the gel twice daily, with assessments at baseline, day 3, and day 7 (J. Drugs Dermatol. 2014;13:725-8).
At day 7, all patients achieved the primary endpoint of at least a 50% reduction in pruritus. The average Investigator’s Global Assessment (IGA) scale score was .55 (down from 2.35 at baseline), which translated to a 76% improvement from baseline. The visual analog score decreased by an average of 6.4 points, for an average reduction of 85%. Statistically significant improvements from baseline in both IGA and visual analog scores also were noted at day 3, when the average IGA score improved 27% from baseline, and the mean visual analog score showed a 53% reduction.
The study included 20 atopic dermatitis patients ranging in age from 8 to 68 years, with an average age of 25 years; 60% were black, 40% were white, and 75% were female.
The results suggest that hydrogel is an appropriate option to relieve itchiness in AD patients, and a large, randomized, double-blind controlled trial would be helpful to further study effectiveness, Dr. Kircik said.
Dr. Kircik disclosed receiving funding as an investigator, consultant, or speaker for Bayer Dermatology, manufacturer of the product tested.
Twice-daily application of 0.05% desonide hydrogel significantly improved clinical symptoms of itching and quality of life scores in 100% of patients with mild to moderate atopic dermatitis after 7 days in a small preliminary study.
"Increasingly, researchers and clinicians recognize that disrupted barrier function contributes not only to the xerotic and pruritic manifestations of AD, but also to the inflammatory cascade that underlies the disease," wrote Dr. Leon Kircik of Indiana University, Indianapolis.
Data from previous studies have shown the effectiveness of desonide hydrogel 0.05% for atopic dermatitis in children, but few studies have examined the effectiveness of the gel on itching in particular. In this study, patients applied the gel twice daily, with assessments at baseline, day 3, and day 7 (J. Drugs Dermatol. 2014;13:725-8).
At day 7, all patients achieved the primary endpoint of at least a 50% reduction in pruritus. The average Investigator’s Global Assessment (IGA) scale score was .55 (down from 2.35 at baseline), which translated to a 76% improvement from baseline. The visual analog score decreased by an average of 6.4 points, for an average reduction of 85%. Statistically significant improvements from baseline in both IGA and visual analog scores also were noted at day 3, when the average IGA score improved 27% from baseline, and the mean visual analog score showed a 53% reduction.
The study included 20 atopic dermatitis patients ranging in age from 8 to 68 years, with an average age of 25 years; 60% were black, 40% were white, and 75% were female.
The results suggest that hydrogel is an appropriate option to relieve itchiness in AD patients, and a large, randomized, double-blind controlled trial would be helpful to further study effectiveness, Dr. Kircik said.
Dr. Kircik disclosed receiving funding as an investigator, consultant, or speaker for Bayer Dermatology, manufacturer of the product tested.
FROM JOURNAL OF DRUGS IN DERMATOLOGY
Key clinical point: Many atopic dermatitis patients object to applying ointments, and a water-based gel offered a nongreasy, effective alternative that improves itching and achieved high levels of patient satisfaction.
Major finding: Desonide hydrogel 0.05% improved pruritus by at least 50% based on investigator and patient assessment scales.
Data source: An open-label pilot study of 20 individuals with mild to moderate atopic dermatitis.
Disclosures: Dr. Kircik disclosed receiving funding as an investigator, consultant, or speaker for Bayer Dermatology, manufacturer of the product tested.
Desonide hydrogel improved itching in atopic dermatitis
Twice-daily application of 0.05% desonide hydrogel significantly improved clinical symptoms of itching and quality of life scores in 100% of patients with mild to moderate atopic dermatitis after 7 days in a small preliminary study.
"Increasingly, researchers and clinicians recognize that disrupted barrier function contributes not only to the xerotic and pruritic manifestations of AD, but also to the inflammatory cascade that underlies the disease," wrote Dr. Leon Kircik of Indiana University, Indianapolis.
Data from previous studies have shown the effectiveness of desonide hydrogel 0.05% for atopic dermatitis in children, but few studies have examined the effectiveness of the gel on itching in particular. In this study, patients applied the gel twice daily, with assessments at baseline, day 3, and day 7 (J. Drugs Dermatol. 2014;13:725-8).
At day 7, all patients achieved the primary endpoint of at least a 50% reduction in pruritus. The average Investigator’s Global Assessment (IGA) scale score was .55 (down from 2.35 at baseline), which translated to a 76% improvement from baseline. The visual analog score decreased by an average of 6.4 points, for an average reduction of 85%. Statistically significant improvements from baseline in both IGA and visual analog scores also were noted at day 3, when the average IGA score improved 27% from baseline, and the mean visual analog score showed a 53% reduction.
The study included 20 atopic dermatitis patients ranging in age from 8 to 68 years, with an average age of 25 years; 60% were black, 40% were white, and 75% were female.
The results suggest that hydrogel is an appropriate option to relieve itchiness in AD patients, and a large, randomized, double-blind controlled trial would be helpful to further study effectiveness, Dr. Kircik said.
Dr. Kircik disclosed receiving funding as an investigator, consultant, or speaker for Bayer Dermatology, manufacturer of the product tested.
Twice-daily application of 0.05% desonide hydrogel significantly improved clinical symptoms of itching and quality of life scores in 100% of patients with mild to moderate atopic dermatitis after 7 days in a small preliminary study.
"Increasingly, researchers and clinicians recognize that disrupted barrier function contributes not only to the xerotic and pruritic manifestations of AD, but also to the inflammatory cascade that underlies the disease," wrote Dr. Leon Kircik of Indiana University, Indianapolis.
Data from previous studies have shown the effectiveness of desonide hydrogel 0.05% for atopic dermatitis in children, but few studies have examined the effectiveness of the gel on itching in particular. In this study, patients applied the gel twice daily, with assessments at baseline, day 3, and day 7 (J. Drugs Dermatol. 2014;13:725-8).
At day 7, all patients achieved the primary endpoint of at least a 50% reduction in pruritus. The average Investigator’s Global Assessment (IGA) scale score was .55 (down from 2.35 at baseline), which translated to a 76% improvement from baseline. The visual analog score decreased by an average of 6.4 points, for an average reduction of 85%. Statistically significant improvements from baseline in both IGA and visual analog scores also were noted at day 3, when the average IGA score improved 27% from baseline, and the mean visual analog score showed a 53% reduction.
The study included 20 atopic dermatitis patients ranging in age from 8 to 68 years, with an average age of 25 years; 60% were black, 40% were white, and 75% were female.
The results suggest that hydrogel is an appropriate option to relieve itchiness in AD patients, and a large, randomized, double-blind controlled trial would be helpful to further study effectiveness, Dr. Kircik said.
Dr. Kircik disclosed receiving funding as an investigator, consultant, or speaker for Bayer Dermatology, manufacturer of the product tested.
Twice-daily application of 0.05% desonide hydrogel significantly improved clinical symptoms of itching and quality of life scores in 100% of patients with mild to moderate atopic dermatitis after 7 days in a small preliminary study.
"Increasingly, researchers and clinicians recognize that disrupted barrier function contributes not only to the xerotic and pruritic manifestations of AD, but also to the inflammatory cascade that underlies the disease," wrote Dr. Leon Kircik of Indiana University, Indianapolis.
Data from previous studies have shown the effectiveness of desonide hydrogel 0.05% for atopic dermatitis in children, but few studies have examined the effectiveness of the gel on itching in particular. In this study, patients applied the gel twice daily, with assessments at baseline, day 3, and day 7 (J. Drugs Dermatol. 2014;13:725-8).
At day 7, all patients achieved the primary endpoint of at least a 50% reduction in pruritus. The average Investigator’s Global Assessment (IGA) scale score was .55 (down from 2.35 at baseline), which translated to a 76% improvement from baseline. The visual analog score decreased by an average of 6.4 points, for an average reduction of 85%. Statistically significant improvements from baseline in both IGA and visual analog scores also were noted at day 3, when the average IGA score improved 27% from baseline, and the mean visual analog score showed a 53% reduction.
The study included 20 atopic dermatitis patients ranging in age from 8 to 68 years, with an average age of 25 years; 60% were black, 40% were white, and 75% were female.
The results suggest that hydrogel is an appropriate option to relieve itchiness in AD patients, and a large, randomized, double-blind controlled trial would be helpful to further study effectiveness, Dr. Kircik said.
Dr. Kircik disclosed receiving funding as an investigator, consultant, or speaker for Bayer Dermatology, manufacturer of the product tested.
FROM JOURNAL OF DRUGS IN DERMATOLOGY
Key clinical point: Many atopic dermatitis patients object to applying ointments, and a water-based gel offered a nongreasy, effective alternative that improves itching and achieved high levels of patient satisfaction.
Major finding: Desonide hydrogel 0.05% improved pruritus by at least 50% based on investigator and patient assessment scales.
Data source: An open-label pilot study of 20 individuals with mild to moderate atopic dermatitis.
Disclosures: Dr. Kircik disclosed receiving funding as an investigator, consultant, or speaker for Bayer Dermatology, manufacturer of the product tested.
Atopic dermatitis update taps top therapy choices
WAIKOLOA, HAWAII – The 2014 update of the American Academy of Dermatology guidelines on atopic dermatitis take a strong stance in favor of the use of topical calcineurin inhibitors as topical corticosteroid-sparing agents, even in children less than 2 years old, where the use of the medications remains off-label.
The evidence-based AAD guidelines bestow a Class A, Level of Evidence I recommendation for the use of topical calcineurin inhibitors as topical steroid-sparing agents. The proactive use of topical calcineurin inhibitors as proactively scheduled, short-term, intermittent maintenance therapy to prevent disease flares also gets an A-I recommendation in the guidelines. The report states that there is no need to monitor blood levels of topical calcineurin inhibitors, Dr. Wynnis Tom noted at the Hawaii Dermatology Seminar, sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The guidelines emphasize the importance of having a proactive discussion with the patient and/or parents about the black box warning for topical calcineurin inhibitors, stressing the fact that interim analyses of long-term surveillance studies do not show an increase in malignancies, said Dr. Tom, a member of the working group that developed the AAD guidelines and a pediatric dermatologist at the University of California, San Diego.
The section of the guidelines devoted to topical therapies includes a detailed description of wet wrap therapy, which is useful for quickly reducing atopic dermatitis severity during flares. The use of topical antihistamines gets a strong thumbs-down in the guidelines.
The guidelines go into considerable detail about the importance of fixing the skin barrier. Bathing is recommended as an important component of therapy, with the caveat that there is no good evidence as to the optimal frequency or duration.
"You want to get the crusting off and hydrate the skin; but you do have to be careful of how long the bath lasts, because you don’t want the skin to dry out as the water evaporates," Dr. Tom said. "I find bathing even daily is good, so long as people are using moisturizers afterward. That’s the key part both for treatment and maintenance: liberal use of moisturizers."
The guideline panel determined that while moisturizers are a cornerstone of atopic dermatitis therapy, no one moisturizer product has been shown to be better than the others. And that includes the prescription devices containing ceramides or hydrolipids, which haven’t persuasively been shown to have clinical advantages over inexpensive over-the-counter moisturizers.
The 2014 atopic dermatitis guidelines are the first-ever AAD guidelines to include an entire section devoted to the diagnosis of a dermatologic disorder (J. Am. Acad. Dermatol. 2014;70:338-51). This was deemed necessary because misdiagnosis of atopic dermatitis is a problem, particularly in adults.
The guidelines stress that atopic dermatitis is a clinical diagnosis that requires ruling out conditions including contact dermatitis, cutaneous T-cell lymphoma, psoriasis, photosensitivity reactions, seborrheic dermatitis, and immune deficiency diseases. At this time, no specific biomarkers can be recommended for the diagnosis of atopic dermatitis or assessment of its severity. In particular, according to the guidelines, the popular practice of monitoring immunoglobulin E levels isn’t recommended.
The comprehensive guidelines also include a section on phototherapy and systemic agents. In addition, a section on preventing disease flares and the use of adjunctive therapies is slated for release in June.
Dr. Tom reported serving as a financially uncompensated investigator for studies sponsored by Amgen and Anacor. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The 2014 update of the American Academy of Dermatology guidelines on atopic dermatitis take a strong stance in favor of the use of topical calcineurin inhibitors as topical corticosteroid-sparing agents, even in children less than 2 years old, where the use of the medications remains off-label.
The evidence-based AAD guidelines bestow a Class A, Level of Evidence I recommendation for the use of topical calcineurin inhibitors as topical steroid-sparing agents. The proactive use of topical calcineurin inhibitors as proactively scheduled, short-term, intermittent maintenance therapy to prevent disease flares also gets an A-I recommendation in the guidelines. The report states that there is no need to monitor blood levels of topical calcineurin inhibitors, Dr. Wynnis Tom noted at the Hawaii Dermatology Seminar, sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The guidelines emphasize the importance of having a proactive discussion with the patient and/or parents about the black box warning for topical calcineurin inhibitors, stressing the fact that interim analyses of long-term surveillance studies do not show an increase in malignancies, said Dr. Tom, a member of the working group that developed the AAD guidelines and a pediatric dermatologist at the University of California, San Diego.
The section of the guidelines devoted to topical therapies includes a detailed description of wet wrap therapy, which is useful for quickly reducing atopic dermatitis severity during flares. The use of topical antihistamines gets a strong thumbs-down in the guidelines.
The guidelines go into considerable detail about the importance of fixing the skin barrier. Bathing is recommended as an important component of therapy, with the caveat that there is no good evidence as to the optimal frequency or duration.
"You want to get the crusting off and hydrate the skin; but you do have to be careful of how long the bath lasts, because you don’t want the skin to dry out as the water evaporates," Dr. Tom said. "I find bathing even daily is good, so long as people are using moisturizers afterward. That’s the key part both for treatment and maintenance: liberal use of moisturizers."
The guideline panel determined that while moisturizers are a cornerstone of atopic dermatitis therapy, no one moisturizer product has been shown to be better than the others. And that includes the prescription devices containing ceramides or hydrolipids, which haven’t persuasively been shown to have clinical advantages over inexpensive over-the-counter moisturizers.
The 2014 atopic dermatitis guidelines are the first-ever AAD guidelines to include an entire section devoted to the diagnosis of a dermatologic disorder (J. Am. Acad. Dermatol. 2014;70:338-51). This was deemed necessary because misdiagnosis of atopic dermatitis is a problem, particularly in adults.
The guidelines stress that atopic dermatitis is a clinical diagnosis that requires ruling out conditions including contact dermatitis, cutaneous T-cell lymphoma, psoriasis, photosensitivity reactions, seborrheic dermatitis, and immune deficiency diseases. At this time, no specific biomarkers can be recommended for the diagnosis of atopic dermatitis or assessment of its severity. In particular, according to the guidelines, the popular practice of monitoring immunoglobulin E levels isn’t recommended.
The comprehensive guidelines also include a section on phototherapy and systemic agents. In addition, a section on preventing disease flares and the use of adjunctive therapies is slated for release in June.
Dr. Tom reported serving as a financially uncompensated investigator for studies sponsored by Amgen and Anacor. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The 2014 update of the American Academy of Dermatology guidelines on atopic dermatitis take a strong stance in favor of the use of topical calcineurin inhibitors as topical corticosteroid-sparing agents, even in children less than 2 years old, where the use of the medications remains off-label.
The evidence-based AAD guidelines bestow a Class A, Level of Evidence I recommendation for the use of topical calcineurin inhibitors as topical steroid-sparing agents. The proactive use of topical calcineurin inhibitors as proactively scheduled, short-term, intermittent maintenance therapy to prevent disease flares also gets an A-I recommendation in the guidelines. The report states that there is no need to monitor blood levels of topical calcineurin inhibitors, Dr. Wynnis Tom noted at the Hawaii Dermatology Seminar, sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The guidelines emphasize the importance of having a proactive discussion with the patient and/or parents about the black box warning for topical calcineurin inhibitors, stressing the fact that interim analyses of long-term surveillance studies do not show an increase in malignancies, said Dr. Tom, a member of the working group that developed the AAD guidelines and a pediatric dermatologist at the University of California, San Diego.
The section of the guidelines devoted to topical therapies includes a detailed description of wet wrap therapy, which is useful for quickly reducing atopic dermatitis severity during flares. The use of topical antihistamines gets a strong thumbs-down in the guidelines.
The guidelines go into considerable detail about the importance of fixing the skin barrier. Bathing is recommended as an important component of therapy, with the caveat that there is no good evidence as to the optimal frequency or duration.
"You want to get the crusting off and hydrate the skin; but you do have to be careful of how long the bath lasts, because you don’t want the skin to dry out as the water evaporates," Dr. Tom said. "I find bathing even daily is good, so long as people are using moisturizers afterward. That’s the key part both for treatment and maintenance: liberal use of moisturizers."
The guideline panel determined that while moisturizers are a cornerstone of atopic dermatitis therapy, no one moisturizer product has been shown to be better than the others. And that includes the prescription devices containing ceramides or hydrolipids, which haven’t persuasively been shown to have clinical advantages over inexpensive over-the-counter moisturizers.
The 2014 atopic dermatitis guidelines are the first-ever AAD guidelines to include an entire section devoted to the diagnosis of a dermatologic disorder (J. Am. Acad. Dermatol. 2014;70:338-51). This was deemed necessary because misdiagnosis of atopic dermatitis is a problem, particularly in adults.
The guidelines stress that atopic dermatitis is a clinical diagnosis that requires ruling out conditions including contact dermatitis, cutaneous T-cell lymphoma, psoriasis, photosensitivity reactions, seborrheic dermatitis, and immune deficiency diseases. At this time, no specific biomarkers can be recommended for the diagnosis of atopic dermatitis or assessment of its severity. In particular, according to the guidelines, the popular practice of monitoring immunoglobulin E levels isn’t recommended.
The comprehensive guidelines also include a section on phototherapy and systemic agents. In addition, a section on preventing disease flares and the use of adjunctive therapies is slated for release in June.
Dr. Tom reported serving as a financially uncompensated investigator for studies sponsored by Amgen and Anacor. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR