User login
The Hospitalist only
Performance Partnership
With 37,000 individual members and nearly 5,000 hospitals and other provider groups on its roster, the American Hospital Association (AHA) is a major player in national healthcare debates and in shaping policies aimed at improving quality.
John Combes, MD, AHA senior vice president and president and chief operating officer of the association-affiliated Center for Healthcare Governance, serves on several national advisory groups on medical ethics, palliative care, and reducing medication errors.
Among his many duties, he is a principal investigator for a national project aimed at reducing hospital-acquired infections called “On the CUSP: Stop Bloodstream Infections,” sponsored by the Agency for Healthcare Research and Quality (AHRQ). (CUSP is the Comprehensive Unit-based Safety Program, developed by Johns Hopkins University and the Michigan Hospital Association.)
Dr. Combes recently talked with The Hospitalist about the AHA’s vision for healthcare reform, integrated care, and the role of hospitalists in redesigning hospital-based care.
Question: What are the AHA’s biggest priorities over the next year?
Answer: Healthcare reform and making sure that we can increase coverage for patients without insurance. There are 48 million uninsured in this country, and we are very supportive of increasing that coverage to make sure that people have good access to healthcare.
Q: The AHA has stated that “clinical integration holds the promise of greater quality and improved efficiency in delivering patient-centered care.” What’s your vision for clinical integration?
A: What we recognize is that in a reformed delivery system, we have to have a lot of partnerships between hospitals and clinicians—physicians in particular—and between hospitals and other facilities, such as long-term care facilities and post-acute facilities. We need to be able to bring better-coordinated care that meets the patient’s needs, and we need to work with each other to constantly improve that care. So that’s why we’re looking at an integrated delivery system. In our minds, it really means one registration, one bill, one experience for the patient.
Practically speaking, if you look at the healthcare reform legislation … there are pilots in there for accountable-care organizations (ACOs) and other payment reforms. And we’re very interested in making sure that hospitals can participate and take a leadership role in the development of those kinds of new structures.
Q: What role do you expect hospitalists to play in the continued drive for higher quality and more efficient care?
A: I think hospitalists can become a critical partner with the hospital in helping us redesign inpatient care to make it more efficient and effective. Additionally, hospitalists have a key role in engaging and keeping involved the community-based PCP, and making sure that they are considered part of the care team, even though they may not be present in the hospital, since they have the continuing responsibility for the patient.
I think as we look at other models of care delivery, such as the patient-centered medical home, it’s critical that hospitalists really develop some strong relationships and communication networks with those groups as well, so that the care for the patient can become seamless and transitions are not as dangerous as they’ve been in the past, in terms of missed opportunities and missed handoffs.

Q: What are the necessary ingredients for any successful quality incentive payment program?
A: One of our big concerns is that there are lots of regulatory obstacles to true integration, where you can design some of those payment structures in terms of gainsharing and also in terms of payment for high-level-quality performance. One of the concerns of the AHM is to make sure that as we pursue these new models of care that require high levels of integration, we also look at some regulatory relief.
The promise is that if we can integrate the delivery system, we can then get focused on improving care and then rewarding high-quality delivery of care. And that can come through incentive programs or pay-for-performance programs and things of that nature that can be worked out between the hospitals and the physicians.
Q: What can be done to reduce the rates of hospital-acquired infections?
A: The idea of CUSP is that you create teams and a culture on units that will then implement the evidence-based intervention—in this case, eliminating central-line infections.
Hospitalists can play a critical role in helping create that culture of mutual accountability at the team level [and] holding each other accountable to use the evidence-based techniques for, in this case, line insertion, or for any kind of safety intervention. I think eliminating infections is a goal that’s achievable. I think we have come to the understanding over the last five or so years that these complications are avoidable in many, many cases, and that it takes teamwork, communication, and use of evidence-based procedures to get the work done.
Q: What can be done to help reduce preventable hospital readmissions?
A: There are so many things that go into readmissions. And the issue is: What is truly preventable in terms of treatments within the hospital, the coordination of discharge, and aftercare followup? A lot of readmissions are related to social determinants of health. And those have to do with people’s ability to afford their medications, people’s ability to access care, people’s home environment, and things of that nature. It’s going to take an approach by hospitals on those things that are controllable in partnerships with the physicians. But for many, many readmissions, it’s related to other issues that we as a society really have to hold ourselves accountable to.
Q: Some critics have charged that the overuse of medical technology is helping to drive up healthcare costs. Would there be more of a role for hospitals in decision-making about the appropriateness of tests within a model like an ACO?
A: In an ACO, that’s a partnership between hospitals and physicians operating as one entity. So that’s the difference, because there, everybody is aligned to make sure that we deliver the most effective care. There’s going to be much more time spent on physicians ordering the most appropriate technology or treatments for that condition that will deliver value to the patient and to the payor of that care.
But that’s in a totally integrated system. Right now we don’t have that. So where the interests of the physicians may be different from the interests of the hospital and the intentions are not aligned, it’s very hard to get at talking about what’s the most effective care.
Q: Is there a measure that hasn’t received as much attention that you would like to see more focus on to help improve the quality or cost-effectiveness of healthcare?
A: I think the one area that we’re always challenged with—and I think we’ve seen it in the healthcare debate, and I think it’s an appropriate role for us as healthcare providers to pay attention to—is palliative and end-of-life care. I don’t think we’ve done enough work, as a profession, to make sure that we deliver very-high-quality care of patients with chronic and acute catastrophic illnesses.
We need to better understand what the needs of those patients are, to ask them to work with us to set the goals with them, what they want from us.
So I think it’s an opportunity for us to have a real partnership with patients at a critical time in their lives. TH
Bryn Nelson is a freelance medical writer based in Seattle.
With 37,000 individual members and nearly 5,000 hospitals and other provider groups on its roster, the American Hospital Association (AHA) is a major player in national healthcare debates and in shaping policies aimed at improving quality.
John Combes, MD, AHA senior vice president and president and chief operating officer of the association-affiliated Center for Healthcare Governance, serves on several national advisory groups on medical ethics, palliative care, and reducing medication errors.
Among his many duties, he is a principal investigator for a national project aimed at reducing hospital-acquired infections called “On the CUSP: Stop Bloodstream Infections,” sponsored by the Agency for Healthcare Research and Quality (AHRQ). (CUSP is the Comprehensive Unit-based Safety Program, developed by Johns Hopkins University and the Michigan Hospital Association.)
Dr. Combes recently talked with The Hospitalist about the AHA’s vision for healthcare reform, integrated care, and the role of hospitalists in redesigning hospital-based care.
Question: What are the AHA’s biggest priorities over the next year?
Answer: Healthcare reform and making sure that we can increase coverage for patients without insurance. There are 48 million uninsured in this country, and we are very supportive of increasing that coverage to make sure that people have good access to healthcare.
Q: The AHA has stated that “clinical integration holds the promise of greater quality and improved efficiency in delivering patient-centered care.” What’s your vision for clinical integration?
A: What we recognize is that in a reformed delivery system, we have to have a lot of partnerships between hospitals and clinicians—physicians in particular—and between hospitals and other facilities, such as long-term care facilities and post-acute facilities. We need to be able to bring better-coordinated care that meets the patient’s needs, and we need to work with each other to constantly improve that care. So that’s why we’re looking at an integrated delivery system. In our minds, it really means one registration, one bill, one experience for the patient.
Practically speaking, if you look at the healthcare reform legislation … there are pilots in there for accountable-care organizations (ACOs) and other payment reforms. And we’re very interested in making sure that hospitals can participate and take a leadership role in the development of those kinds of new structures.
Q: What role do you expect hospitalists to play in the continued drive for higher quality and more efficient care?
A: I think hospitalists can become a critical partner with the hospital in helping us redesign inpatient care to make it more efficient and effective. Additionally, hospitalists have a key role in engaging and keeping involved the community-based PCP, and making sure that they are considered part of the care team, even though they may not be present in the hospital, since they have the continuing responsibility for the patient.
I think as we look at other models of care delivery, such as the patient-centered medical home, it’s critical that hospitalists really develop some strong relationships and communication networks with those groups as well, so that the care for the patient can become seamless and transitions are not as dangerous as they’ve been in the past, in terms of missed opportunities and missed handoffs.

Q: What are the necessary ingredients for any successful quality incentive payment program?
A: One of our big concerns is that there are lots of regulatory obstacles to true integration, where you can design some of those payment structures in terms of gainsharing and also in terms of payment for high-level-quality performance. One of the concerns of the AHM is to make sure that as we pursue these new models of care that require high levels of integration, we also look at some regulatory relief.
The promise is that if we can integrate the delivery system, we can then get focused on improving care and then rewarding high-quality delivery of care. And that can come through incentive programs or pay-for-performance programs and things of that nature that can be worked out between the hospitals and the physicians.
Q: What can be done to reduce the rates of hospital-acquired infections?
A: The idea of CUSP is that you create teams and a culture on units that will then implement the evidence-based intervention—in this case, eliminating central-line infections.
Hospitalists can play a critical role in helping create that culture of mutual accountability at the team level [and] holding each other accountable to use the evidence-based techniques for, in this case, line insertion, or for any kind of safety intervention. I think eliminating infections is a goal that’s achievable. I think we have come to the understanding over the last five or so years that these complications are avoidable in many, many cases, and that it takes teamwork, communication, and use of evidence-based procedures to get the work done.
Q: What can be done to help reduce preventable hospital readmissions?
A: There are so many things that go into readmissions. And the issue is: What is truly preventable in terms of treatments within the hospital, the coordination of discharge, and aftercare followup? A lot of readmissions are related to social determinants of health. And those have to do with people’s ability to afford their medications, people’s ability to access care, people’s home environment, and things of that nature. It’s going to take an approach by hospitals on those things that are controllable in partnerships with the physicians. But for many, many readmissions, it’s related to other issues that we as a society really have to hold ourselves accountable to.
Q: Some critics have charged that the overuse of medical technology is helping to drive up healthcare costs. Would there be more of a role for hospitals in decision-making about the appropriateness of tests within a model like an ACO?
A: In an ACO, that’s a partnership between hospitals and physicians operating as one entity. So that’s the difference, because there, everybody is aligned to make sure that we deliver the most effective care. There’s going to be much more time spent on physicians ordering the most appropriate technology or treatments for that condition that will deliver value to the patient and to the payor of that care.
But that’s in a totally integrated system. Right now we don’t have that. So where the interests of the physicians may be different from the interests of the hospital and the intentions are not aligned, it’s very hard to get at talking about what’s the most effective care.
Q: Is there a measure that hasn’t received as much attention that you would like to see more focus on to help improve the quality or cost-effectiveness of healthcare?
A: I think the one area that we’re always challenged with—and I think we’ve seen it in the healthcare debate, and I think it’s an appropriate role for us as healthcare providers to pay attention to—is palliative and end-of-life care. I don’t think we’ve done enough work, as a profession, to make sure that we deliver very-high-quality care of patients with chronic and acute catastrophic illnesses.
We need to better understand what the needs of those patients are, to ask them to work with us to set the goals with them, what they want from us.
So I think it’s an opportunity for us to have a real partnership with patients at a critical time in their lives. TH
Bryn Nelson is a freelance medical writer based in Seattle.
With 37,000 individual members and nearly 5,000 hospitals and other provider groups on its roster, the American Hospital Association (AHA) is a major player in national healthcare debates and in shaping policies aimed at improving quality.
John Combes, MD, AHA senior vice president and president and chief operating officer of the association-affiliated Center for Healthcare Governance, serves on several national advisory groups on medical ethics, palliative care, and reducing medication errors.
Among his many duties, he is a principal investigator for a national project aimed at reducing hospital-acquired infections called “On the CUSP: Stop Bloodstream Infections,” sponsored by the Agency for Healthcare Research and Quality (AHRQ). (CUSP is the Comprehensive Unit-based Safety Program, developed by Johns Hopkins University and the Michigan Hospital Association.)
Dr. Combes recently talked with The Hospitalist about the AHA’s vision for healthcare reform, integrated care, and the role of hospitalists in redesigning hospital-based care.
Question: What are the AHA’s biggest priorities over the next year?
Answer: Healthcare reform and making sure that we can increase coverage for patients without insurance. There are 48 million uninsured in this country, and we are very supportive of increasing that coverage to make sure that people have good access to healthcare.
Q: The AHA has stated that “clinical integration holds the promise of greater quality and improved efficiency in delivering patient-centered care.” What’s your vision for clinical integration?
A: What we recognize is that in a reformed delivery system, we have to have a lot of partnerships between hospitals and clinicians—physicians in particular—and between hospitals and other facilities, such as long-term care facilities and post-acute facilities. We need to be able to bring better-coordinated care that meets the patient’s needs, and we need to work with each other to constantly improve that care. So that’s why we’re looking at an integrated delivery system. In our minds, it really means one registration, one bill, one experience for the patient.
Practically speaking, if you look at the healthcare reform legislation … there are pilots in there for accountable-care organizations (ACOs) and other payment reforms. And we’re very interested in making sure that hospitals can participate and take a leadership role in the development of those kinds of new structures.
Q: What role do you expect hospitalists to play in the continued drive for higher quality and more efficient care?
A: I think hospitalists can become a critical partner with the hospital in helping us redesign inpatient care to make it more efficient and effective. Additionally, hospitalists have a key role in engaging and keeping involved the community-based PCP, and making sure that they are considered part of the care team, even though they may not be present in the hospital, since they have the continuing responsibility for the patient.
I think as we look at other models of care delivery, such as the patient-centered medical home, it’s critical that hospitalists really develop some strong relationships and communication networks with those groups as well, so that the care for the patient can become seamless and transitions are not as dangerous as they’ve been in the past, in terms of missed opportunities and missed handoffs.

Q: What are the necessary ingredients for any successful quality incentive payment program?
A: One of our big concerns is that there are lots of regulatory obstacles to true integration, where you can design some of those payment structures in terms of gainsharing and also in terms of payment for high-level-quality performance. One of the concerns of the AHM is to make sure that as we pursue these new models of care that require high levels of integration, we also look at some regulatory relief.
The promise is that if we can integrate the delivery system, we can then get focused on improving care and then rewarding high-quality delivery of care. And that can come through incentive programs or pay-for-performance programs and things of that nature that can be worked out between the hospitals and the physicians.
Q: What can be done to reduce the rates of hospital-acquired infections?
A: The idea of CUSP is that you create teams and a culture on units that will then implement the evidence-based intervention—in this case, eliminating central-line infections.
Hospitalists can play a critical role in helping create that culture of mutual accountability at the team level [and] holding each other accountable to use the evidence-based techniques for, in this case, line insertion, or for any kind of safety intervention. I think eliminating infections is a goal that’s achievable. I think we have come to the understanding over the last five or so years that these complications are avoidable in many, many cases, and that it takes teamwork, communication, and use of evidence-based procedures to get the work done.
Q: What can be done to help reduce preventable hospital readmissions?
A: There are so many things that go into readmissions. And the issue is: What is truly preventable in terms of treatments within the hospital, the coordination of discharge, and aftercare followup? A lot of readmissions are related to social determinants of health. And those have to do with people’s ability to afford their medications, people’s ability to access care, people’s home environment, and things of that nature. It’s going to take an approach by hospitals on those things that are controllable in partnerships with the physicians. But for many, many readmissions, it’s related to other issues that we as a society really have to hold ourselves accountable to.
Q: Some critics have charged that the overuse of medical technology is helping to drive up healthcare costs. Would there be more of a role for hospitals in decision-making about the appropriateness of tests within a model like an ACO?
A: In an ACO, that’s a partnership between hospitals and physicians operating as one entity. So that’s the difference, because there, everybody is aligned to make sure that we deliver the most effective care. There’s going to be much more time spent on physicians ordering the most appropriate technology or treatments for that condition that will deliver value to the patient and to the payor of that care.
But that’s in a totally integrated system. Right now we don’t have that. So where the interests of the physicians may be different from the interests of the hospital and the intentions are not aligned, it’s very hard to get at talking about what’s the most effective care.
Q: Is there a measure that hasn’t received as much attention that you would like to see more focus on to help improve the quality or cost-effectiveness of healthcare?
A: I think the one area that we’re always challenged with—and I think we’ve seen it in the healthcare debate, and I think it’s an appropriate role for us as healthcare providers to pay attention to—is palliative and end-of-life care. I don’t think we’ve done enough work, as a profession, to make sure that we deliver very-high-quality care of patients with chronic and acute catastrophic illnesses.
We need to better understand what the needs of those patients are, to ask them to work with us to set the goals with them, what they want from us.
So I think it’s an opportunity for us to have a real partnership with patients at a critical time in their lives. TH
Bryn Nelson is a freelance medical writer based in Seattle.
Practice Management Session
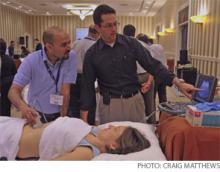
PHYSICIAN ASSISTANTS (PAs) and nurse practitioners can do almost anything a physician can do, and many have skill sets physicians lack, according to David Friar, MD, FHM, president of Hospitalists of Northwest Michigan based in Traverse City.
“As we go forward, with continued physician shortages and with the growing responsibilities of the hospitalist movement, we are going to need more and more people with different skill sets,” Dr. Friar said during his presentation at HM10. “I think one of those important areas is nonphysician providers.”
A quick survey of the 300 or so hospitalists at the session showed most HM groups employ NPPs, but less than a third of those thought they were “using NPPs well.” Dr. Friar, who has worked with NPs and PAs for 14 years, said he has found NPPs “to be an integral part of our practice. They have become indispensable to us in the way we provide services to our hospitals and patients.”
Still, many hospitalist groups waste NPP potential, Dr. Friar explained. He suggested HM groups evaluate their NPP roster and duties, and make necessary changes. “Make sure you treat them as if they are part of the team. That is very important,” he said. “NPPs can and should take care of patients throughout all stages of the hospital stay, from admission to discharge.”
When hiring NPPs, look for team players who are flexible and willing to learn. Make sure the NPP knows their limits and is comfortable asking for help. Target local training programs or other departments in the hospital as fertile ground for new hires. But, most importantly, know what you are getting when you hire an NPP.
“Don’t underestimate the cost of the inexperienced NPP in terms of training. If you think it’s two weeks or two months, you are fooling yourself,” Dr. Friar emphasized, adding an inexperienced NPP could take from six months to two years to reach full competency in hospitalist service. “Know the tradeoffs,” he said. “It might be more important for you to hire the more experienced—and more costly—NPP so you don’t have to spend two years to get them up to speed.” HM10
PHYSICIAN ASSISTANTS (PAs) and nurse practitioners can do almost anything a physician can do, and many have skill sets physicians lack, according to David Friar, MD, FHM, president of Hospitalists of Northwest Michigan based in Traverse City.
“As we go forward, with continued physician shortages and with the growing responsibilities of the hospitalist movement, we are going to need more and more people with different skill sets,” Dr. Friar said during his presentation at HM10. “I think one of those important areas is nonphysician providers.”
A quick survey of the 300 or so hospitalists at the session showed most HM groups employ NPPs, but less than a third of those thought they were “using NPPs well.” Dr. Friar, who has worked with NPs and PAs for 14 years, said he has found NPPs “to be an integral part of our practice. They have become indispensable to us in the way we provide services to our hospitals and patients.”
Still, many hospitalist groups waste NPP potential, Dr. Friar explained. He suggested HM groups evaluate their NPP roster and duties, and make necessary changes. “Make sure you treat them as if they are part of the team. That is very important,” he said. “NPPs can and should take care of patients throughout all stages of the hospital stay, from admission to discharge.”
When hiring NPPs, look for team players who are flexible and willing to learn. Make sure the NPP knows their limits and is comfortable asking for help. Target local training programs or other departments in the hospital as fertile ground for new hires. But, most importantly, know what you are getting when you hire an NPP.
“Don’t underestimate the cost of the inexperienced NPP in terms of training. If you think it’s two weeks or two months, you are fooling yourself,” Dr. Friar emphasized, adding an inexperienced NPP could take from six months to two years to reach full competency in hospitalist service. “Know the tradeoffs,” he said. “It might be more important for you to hire the more experienced—and more costly—NPP so you don’t have to spend two years to get them up to speed.” HM10
PHYSICIAN ASSISTANTS (PAs) and nurse practitioners can do almost anything a physician can do, and many have skill sets physicians lack, according to David Friar, MD, FHM, president of Hospitalists of Northwest Michigan based in Traverse City.
“As we go forward, with continued physician shortages and with the growing responsibilities of the hospitalist movement, we are going to need more and more people with different skill sets,” Dr. Friar said during his presentation at HM10. “I think one of those important areas is nonphysician providers.”
A quick survey of the 300 or so hospitalists at the session showed most HM groups employ NPPs, but less than a third of those thought they were “using NPPs well.” Dr. Friar, who has worked with NPs and PAs for 14 years, said he has found NPPs “to be an integral part of our practice. They have become indispensable to us in the way we provide services to our hospitals and patients.”
Still, many hospitalist groups waste NPP potential, Dr. Friar explained. He suggested HM groups evaluate their NPP roster and duties, and make necessary changes. “Make sure you treat them as if they are part of the team. That is very important,” he said. “NPPs can and should take care of patients throughout all stages of the hospital stay, from admission to discharge.”
When hiring NPPs, look for team players who are flexible and willing to learn. Make sure the NPP knows their limits and is comfortable asking for help. Target local training programs or other departments in the hospital as fertile ground for new hires. But, most importantly, know what you are getting when you hire an NPP.
“Don’t underestimate the cost of the inexperienced NPP in terms of training. If you think it’s two weeks or two months, you are fooling yourself,” Dr. Friar emphasized, adding an inexperienced NPP could take from six months to two years to reach full competency in hospitalist service. “Know the tradeoffs,” he said. “It might be more important for you to hire the more experienced—and more costly—NPP so you don’t have to spend two years to get them up to speed.” HM10
Practice Management Session
IN THEORY, unit-based de-ployment of hospitalists is a perfect answer to the struggles of navigating, say, a 16-patient census that includes seven units on four floors. But in the real world, it’s not.
“Just placing hospitalists on a unit and giving them patients isn’t the answer,” said Russell L. Holman, MD, SFHM, chief operating officer for Cogent Healthcare in Brentwood, Tenn., and past president of SHM. “Structure has to support a deliberate strategy. Think of what your strategic goals are. … Don’t just implement a new structure and let that be the end.”
—Russell L. Holman, MD, SFHM, chief operating officer, Cogent Healthcare, Brentwood, Tenn., past president of SHM
Dr. Holman led a panel, “The Case for Unit-Based Hospitalists: Benefits and Challenges,” in which HM experts agreed that tracking the efficacy of the setup is key to success.
Although the benefits are usually clear—less time spent traveling from floor to floor and more direct communication between physicians and nonphysician providers (NPPs)—the challenges can be numerous, including:
- Fairness. The first complaint of most HM groups switching to a unit-based approach is that it unfairly distributes patient loads, leading to daytime shifts for which one physician starts with a patient census of eight, while a colleague starts with 15.
- Interunit transfers. By creating defined geographic areas, a patient’s movement from one unit to another becomes another transition of care and brings with it those issues.
- Buy-in from other stakeholders. Physician assistants (PA), nursing staff, and others are affected by geographic alignment. Make sure to pitch quantifiable goals—increased productivity, increased touch time with patients, reduced staff turnover—when instituting the new approach.
Kevin O’Leary, MD, MS, associate chief of hospital medicine at Northwestern University’s Feinberg School of Medicine in Chicago, urges physicians to be practical, and not to expect the unit-based approach to be a panacea. “This is really the first step,” he said. HM10
IN THEORY, unit-based de-ployment of hospitalists is a perfect answer to the struggles of navigating, say, a 16-patient census that includes seven units on four floors. But in the real world, it’s not.
“Just placing hospitalists on a unit and giving them patients isn’t the answer,” said Russell L. Holman, MD, SFHM, chief operating officer for Cogent Healthcare in Brentwood, Tenn., and past president of SHM. “Structure has to support a deliberate strategy. Think of what your strategic goals are. … Don’t just implement a new structure and let that be the end.”
—Russell L. Holman, MD, SFHM, chief operating officer, Cogent Healthcare, Brentwood, Tenn., past president of SHM
Dr. Holman led a panel, “The Case for Unit-Based Hospitalists: Benefits and Challenges,” in which HM experts agreed that tracking the efficacy of the setup is key to success.
Although the benefits are usually clear—less time spent traveling from floor to floor and more direct communication between physicians and nonphysician providers (NPPs)—the challenges can be numerous, including:
- Fairness. The first complaint of most HM groups switching to a unit-based approach is that it unfairly distributes patient loads, leading to daytime shifts for which one physician starts with a patient census of eight, while a colleague starts with 15.
- Interunit transfers. By creating defined geographic areas, a patient’s movement from one unit to another becomes another transition of care and brings with it those issues.
- Buy-in from other stakeholders. Physician assistants (PA), nursing staff, and others are affected by geographic alignment. Make sure to pitch quantifiable goals—increased productivity, increased touch time with patients, reduced staff turnover—when instituting the new approach.
Kevin O’Leary, MD, MS, associate chief of hospital medicine at Northwestern University’s Feinberg School of Medicine in Chicago, urges physicians to be practical, and not to expect the unit-based approach to be a panacea. “This is really the first step,” he said. HM10
IN THEORY, unit-based de-ployment of hospitalists is a perfect answer to the struggles of navigating, say, a 16-patient census that includes seven units on four floors. But in the real world, it’s not.
“Just placing hospitalists on a unit and giving them patients isn’t the answer,” said Russell L. Holman, MD, SFHM, chief operating officer for Cogent Healthcare in Brentwood, Tenn., and past president of SHM. “Structure has to support a deliberate strategy. Think of what your strategic goals are. … Don’t just implement a new structure and let that be the end.”
—Russell L. Holman, MD, SFHM, chief operating officer, Cogent Healthcare, Brentwood, Tenn., past president of SHM
Dr. Holman led a panel, “The Case for Unit-Based Hospitalists: Benefits and Challenges,” in which HM experts agreed that tracking the efficacy of the setup is key to success.
Although the benefits are usually clear—less time spent traveling from floor to floor and more direct communication between physicians and nonphysician providers (NPPs)—the challenges can be numerous, including:
- Fairness. The first complaint of most HM groups switching to a unit-based approach is that it unfairly distributes patient loads, leading to daytime shifts for which one physician starts with a patient census of eight, while a colleague starts with 15.
- Interunit transfers. By creating defined geographic areas, a patient’s movement from one unit to another becomes another transition of care and brings with it those issues.
- Buy-in from other stakeholders. Physician assistants (PA), nursing staff, and others are affected by geographic alignment. Make sure to pitch quantifiable goals—increased productivity, increased touch time with patients, reduced staff turnover—when instituting the new approach.
Kevin O’Leary, MD, MS, associate chief of hospital medicine at Northwestern University’s Feinberg School of Medicine in Chicago, urges physicians to be practical, and not to expect the unit-based approach to be a panacea. “This is really the first step,” he said. HM10
Professional Advice
NATIONAL HARBOR, Md.—;Amie Dlouhy, RN, BSN, hospitalist program manager with Saint Mary’s Health Care in Grand Rapids, Mich., couldn’t scribble notes furiously enough during the practice-management pre-course at HM10. Dlouhy was promoted to her new position as an administrator some six weeks before the annual meeting at the Gaylord National Resort & Convention Center in early April.
So the first-time meeting attendee decided she would jot down as many tips as she could. She quickly realized the trip was worth it, as she learned that a departmental dashboard is a relatively simple way to gather key information in one place. She also likes the idea of drawing up a brochure that tells patients what they can expect from their hospitalists—and perhaps vice versa. And what new HM group leader doesn’t want advice on building a schedule that adds individualized wrinkles to the “seven-on, seven-off” structure?
“It is a business and you need to treat it as if it’s a business,” Dlouhy said. “It’s an ongoing process, and you want to make sure you have a concrete foundation.”
The tidbits Dlouhy gleaned from her pre-course were among scores of nuggets discussed during eight of the accredited educational sessions. This year’s pre-courses boosted to a new high of 20 the number of Category 1 credits physicians could earn toward the American Medical Association’s (AMA) Physician Recognition Award. Last year, the total was 15.
Offering more classes—and more varied topics—worked pretty well, as this year’s slate of pre-courses was more popular than ever, according to SHM officials. At HM09 in Chicago, more than 800 attendees participated in six sessions. At HM10, the total attendance was roughly 10% higher.
A main driver of the growth was the addition of two new courses—“Essential Neurology for the Hospitalist” and “Early Career Hospitalist: Skills for Success.” Another was a packed room of hospitalists answering questions—some right, some wrong—and preparing for the new Focused Practice in Hospital Medicine (FPHM) via the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC). The learning session pre-course debuted last year, but the new HM pathway to board recertification helped push attendance higher this year.
“The nice thing about the audience-response system is that you can actually see that not everybody is always going straight to the right answer on all of the questions,” said Julius Yang, MD, PhD, a hospitalist at Beth Israel Deaconess Medical Center in Boston and the MOC course director. “It’s really serving as an important refresher of our medical knowledge base.”
Dr. Yang said the “mini-retreat” environment of an annual convention is the perfect place to focus on granular professional development. “Trying to do these types of MOCs when you’re working to keep current with all of your other duties, you don’t get as much out of it,” Dr. Yang said. “Here, you get it all.”
—Troy Ahlstrom, MD, FHM, Hospitalists of Northwest Michigan, Traverse City
He adds that those physicians who take the time and spend the money to travel for an educational session tend to be very focused on taking advantage of the program, not just showing up to be counted.
“All of these [questions] are very much directed at growing as a hospitalist,” Dr. Yang said. “It’s a different focus than the rest of the meeting. This is about every individual bringing something back to their institution.”
That’s what keeps bringing Troy Ahlstrom, MD, FHM, back. Dr. Ahlstrom, of Hos-pitalists of Northwest Michigan in Traverse City, has been to three annual meetings, and he said he tries to hit a pre-course every time. Last year, it was a session on how to more completely capture costs from billing and coding.
This year: “Comprehensive Critical Care in 2010: An Interactive Course.” The former appealed to him given that every HM group needs to capture as many of its charges as possible, and the latter because his group helps staff the critical-care units of three hospitals.
Several physicians noted that the critical-care pre-course was particularly appealing to attendees, as more hospitalists are handling those duties at their respective institutions. The format was popular, too, and was structured in the same way as the ABIM learning session, with course director David Schul-man, MD, MPH, chief of pulmonary and critical-care medicine at Emory University Hospital in Atlanta, leading a room full of hospitalists through a multiple-choice exam.
Dr. Ahlstrom and others noted that aside from the engagement in education that the daylong pre-courses offer, the sessions are set up with take-home guides, reference materials, and earnest pledges for mentoring from speakers and SHM staff.
“Most medical meetings have a scientific focus with a couple of practical aspects,” Dr. Ahlstrom said. “SHM’s meeting is very practical. It presents research, but it’s research you will use in your practice.”
Gerald Johnson, MD, a hospitalist at Texoma Medical Center in Denison, Texas, signed up for the “Best Practices in Managing a Hospital Medicine Program” pre-course during his first visit to an SHM meeting. A hospitalist for about four years, Dr. Johnson decided to take the pre-course at the urging of senior colleagues. He said the most helpful lessons he gleaned were about compensation plans, scheduling, and staffing.
“It’s not one person getting up there and presenting ‘This is how it needs to be done,’ ” Dr. Johnson said. “They present you with several ways. It really gives you something to adapt to your personal environment.”
Dr. Johnson, who gushed about “the gurus” of HM leading his session, also likes the fact that people with both a financial pedigree and a background in clinical work present the information. In fact, several attendees of the best-practices session noted that the attention to both medicine and management helps fill in the gaps between being a clinician and being a businessman.
“You’ve got to do both well,” Dr. Ahlstrom said. “You’ve got to take good care of patients. But in order to take good care of patients, you have to run a good business model, too.” HM10
Richard Quinn is a freelance writer based in New Jersey.
NATIONAL HARBOR, Md.—;Amie Dlouhy, RN, BSN, hospitalist program manager with Saint Mary’s Health Care in Grand Rapids, Mich., couldn’t scribble notes furiously enough during the practice-management pre-course at HM10. Dlouhy was promoted to her new position as an administrator some six weeks before the annual meeting at the Gaylord National Resort & Convention Center in early April.
So the first-time meeting attendee decided she would jot down as many tips as she could. She quickly realized the trip was worth it, as she learned that a departmental dashboard is a relatively simple way to gather key information in one place. She also likes the idea of drawing up a brochure that tells patients what they can expect from their hospitalists—and perhaps vice versa. And what new HM group leader doesn’t want advice on building a schedule that adds individualized wrinkles to the “seven-on, seven-off” structure?
“It is a business and you need to treat it as if it’s a business,” Dlouhy said. “It’s an ongoing process, and you want to make sure you have a concrete foundation.”
The tidbits Dlouhy gleaned from her pre-course were among scores of nuggets discussed during eight of the accredited educational sessions. This year’s pre-courses boosted to a new high of 20 the number of Category 1 credits physicians could earn toward the American Medical Association’s (AMA) Physician Recognition Award. Last year, the total was 15.
Offering more classes—and more varied topics—worked pretty well, as this year’s slate of pre-courses was more popular than ever, according to SHM officials. At HM09 in Chicago, more than 800 attendees participated in six sessions. At HM10, the total attendance was roughly 10% higher.
A main driver of the growth was the addition of two new courses—“Essential Neurology for the Hospitalist” and “Early Career Hospitalist: Skills for Success.” Another was a packed room of hospitalists answering questions—some right, some wrong—and preparing for the new Focused Practice in Hospital Medicine (FPHM) via the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC). The learning session pre-course debuted last year, but the new HM pathway to board recertification helped push attendance higher this year.
“The nice thing about the audience-response system is that you can actually see that not everybody is always going straight to the right answer on all of the questions,” said Julius Yang, MD, PhD, a hospitalist at Beth Israel Deaconess Medical Center in Boston and the MOC course director. “It’s really serving as an important refresher of our medical knowledge base.”
Dr. Yang said the “mini-retreat” environment of an annual convention is the perfect place to focus on granular professional development. “Trying to do these types of MOCs when you’re working to keep current with all of your other duties, you don’t get as much out of it,” Dr. Yang said. “Here, you get it all.”
—Troy Ahlstrom, MD, FHM, Hospitalists of Northwest Michigan, Traverse City
He adds that those physicians who take the time and spend the money to travel for an educational session tend to be very focused on taking advantage of the program, not just showing up to be counted.
“All of these [questions] are very much directed at growing as a hospitalist,” Dr. Yang said. “It’s a different focus than the rest of the meeting. This is about every individual bringing something back to their institution.”
That’s what keeps bringing Troy Ahlstrom, MD, FHM, back. Dr. Ahlstrom, of Hos-pitalists of Northwest Michigan in Traverse City, has been to three annual meetings, and he said he tries to hit a pre-course every time. Last year, it was a session on how to more completely capture costs from billing and coding.
This year: “Comprehensive Critical Care in 2010: An Interactive Course.” The former appealed to him given that every HM group needs to capture as many of its charges as possible, and the latter because his group helps staff the critical-care units of three hospitals.
Several physicians noted that the critical-care pre-course was particularly appealing to attendees, as more hospitalists are handling those duties at their respective institutions. The format was popular, too, and was structured in the same way as the ABIM learning session, with course director David Schul-man, MD, MPH, chief of pulmonary and critical-care medicine at Emory University Hospital in Atlanta, leading a room full of hospitalists through a multiple-choice exam.
Dr. Ahlstrom and others noted that aside from the engagement in education that the daylong pre-courses offer, the sessions are set up with take-home guides, reference materials, and earnest pledges for mentoring from speakers and SHM staff.
“Most medical meetings have a scientific focus with a couple of practical aspects,” Dr. Ahlstrom said. “SHM’s meeting is very practical. It presents research, but it’s research you will use in your practice.”
Gerald Johnson, MD, a hospitalist at Texoma Medical Center in Denison, Texas, signed up for the “Best Practices in Managing a Hospital Medicine Program” pre-course during his first visit to an SHM meeting. A hospitalist for about four years, Dr. Johnson decided to take the pre-course at the urging of senior colleagues. He said the most helpful lessons he gleaned were about compensation plans, scheduling, and staffing.
“It’s not one person getting up there and presenting ‘This is how it needs to be done,’ ” Dr. Johnson said. “They present you with several ways. It really gives you something to adapt to your personal environment.”
Dr. Johnson, who gushed about “the gurus” of HM leading his session, also likes the fact that people with both a financial pedigree and a background in clinical work present the information. In fact, several attendees of the best-practices session noted that the attention to both medicine and management helps fill in the gaps between being a clinician and being a businessman.
“You’ve got to do both well,” Dr. Ahlstrom said. “You’ve got to take good care of patients. But in order to take good care of patients, you have to run a good business model, too.” HM10
Richard Quinn is a freelance writer based in New Jersey.
NATIONAL HARBOR, Md.—;Amie Dlouhy, RN, BSN, hospitalist program manager with Saint Mary’s Health Care in Grand Rapids, Mich., couldn’t scribble notes furiously enough during the practice-management pre-course at HM10. Dlouhy was promoted to her new position as an administrator some six weeks before the annual meeting at the Gaylord National Resort & Convention Center in early April.
So the first-time meeting attendee decided she would jot down as many tips as she could. She quickly realized the trip was worth it, as she learned that a departmental dashboard is a relatively simple way to gather key information in one place. She also likes the idea of drawing up a brochure that tells patients what they can expect from their hospitalists—and perhaps vice versa. And what new HM group leader doesn’t want advice on building a schedule that adds individualized wrinkles to the “seven-on, seven-off” structure?
“It is a business and you need to treat it as if it’s a business,” Dlouhy said. “It’s an ongoing process, and you want to make sure you have a concrete foundation.”
The tidbits Dlouhy gleaned from her pre-course were among scores of nuggets discussed during eight of the accredited educational sessions. This year’s pre-courses boosted to a new high of 20 the number of Category 1 credits physicians could earn toward the American Medical Association’s (AMA) Physician Recognition Award. Last year, the total was 15.
Offering more classes—and more varied topics—worked pretty well, as this year’s slate of pre-courses was more popular than ever, according to SHM officials. At HM09 in Chicago, more than 800 attendees participated in six sessions. At HM10, the total attendance was roughly 10% higher.
A main driver of the growth was the addition of two new courses—“Essential Neurology for the Hospitalist” and “Early Career Hospitalist: Skills for Success.” Another was a packed room of hospitalists answering questions—some right, some wrong—and preparing for the new Focused Practice in Hospital Medicine (FPHM) via the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC). The learning session pre-course debuted last year, but the new HM pathway to board recertification helped push attendance higher this year.
“The nice thing about the audience-response system is that you can actually see that not everybody is always going straight to the right answer on all of the questions,” said Julius Yang, MD, PhD, a hospitalist at Beth Israel Deaconess Medical Center in Boston and the MOC course director. “It’s really serving as an important refresher of our medical knowledge base.”
Dr. Yang said the “mini-retreat” environment of an annual convention is the perfect place to focus on granular professional development. “Trying to do these types of MOCs when you’re working to keep current with all of your other duties, you don’t get as much out of it,” Dr. Yang said. “Here, you get it all.”
—Troy Ahlstrom, MD, FHM, Hospitalists of Northwest Michigan, Traverse City
He adds that those physicians who take the time and spend the money to travel for an educational session tend to be very focused on taking advantage of the program, not just showing up to be counted.
“All of these [questions] are very much directed at growing as a hospitalist,” Dr. Yang said. “It’s a different focus than the rest of the meeting. This is about every individual bringing something back to their institution.”
That’s what keeps bringing Troy Ahlstrom, MD, FHM, back. Dr. Ahlstrom, of Hos-pitalists of Northwest Michigan in Traverse City, has been to three annual meetings, and he said he tries to hit a pre-course every time. Last year, it was a session on how to more completely capture costs from billing and coding.
This year: “Comprehensive Critical Care in 2010: An Interactive Course.” The former appealed to him given that every HM group needs to capture as many of its charges as possible, and the latter because his group helps staff the critical-care units of three hospitals.
Several physicians noted that the critical-care pre-course was particularly appealing to attendees, as more hospitalists are handling those duties at their respective institutions. The format was popular, too, and was structured in the same way as the ABIM learning session, with course director David Schul-man, MD, MPH, chief of pulmonary and critical-care medicine at Emory University Hospital in Atlanta, leading a room full of hospitalists through a multiple-choice exam.
Dr. Ahlstrom and others noted that aside from the engagement in education that the daylong pre-courses offer, the sessions are set up with take-home guides, reference materials, and earnest pledges for mentoring from speakers and SHM staff.
“Most medical meetings have a scientific focus with a couple of practical aspects,” Dr. Ahlstrom said. “SHM’s meeting is very practical. It presents research, but it’s research you will use in your practice.”
Gerald Johnson, MD, a hospitalist at Texoma Medical Center in Denison, Texas, signed up for the “Best Practices in Managing a Hospital Medicine Program” pre-course during his first visit to an SHM meeting. A hospitalist for about four years, Dr. Johnson decided to take the pre-course at the urging of senior colleagues. He said the most helpful lessons he gleaned were about compensation plans, scheduling, and staffing.
“It’s not one person getting up there and presenting ‘This is how it needs to be done,’ ” Dr. Johnson said. “They present you with several ways. It really gives you something to adapt to your personal environment.”
Dr. Johnson, who gushed about “the gurus” of HM leading his session, also likes the fact that people with both a financial pedigree and a background in clinical work present the information. In fact, several attendees of the best-practices session noted that the attention to both medicine and management helps fill in the gaps between being a clinician and being a businessman.
“You’ve got to do both well,” Dr. Ahlstrom said. “You’ve got to take good care of patients. But in order to take good care of patients, you have to run a good business model, too.” HM10
Richard Quinn is a freelance writer based in New Jersey.
Continuity Conundrum
Editor’s note: Third of a three-part series.
In the two monthly columns preceding this one, I’ve provided an overview of some ways hospitalist groups distribute new referrals among the providers. This month, I’ll review things that cause some groups to make exceptions to their typical method of distributing patients, and turn from how patients are distributed over 24 hours to thoughts about how they might be assigned over the course of consecutive days worked by a doctor.
Equitable Exceptions
There are a number of reasons groups decide to depart from their typical method of assigning patients. These include:
- “Bouncebacks”;
- One hospitalist is at the cap, others aren’t;
- Consult requested of a specific hospitalist;
- Hospitalists with unique skills (e.g., ICU expertise); and
- A patient “fires” the hospitalist.
There isn’t a standard “hospitalist way” of dealing with these issues, and each group will need to work out its own system. The most common of these issues is “bouncebacks.” Every group should try to have patients readmitted within three or four days of discharge go back to the discharging hospitalist. However, this proves difficult in many cases for several reasons, most commonly because the original discharging doctor might not be working when the patient returns.
The Alpha & Omega
Nearly every hospitalist practice makes some effort to maximize continuity between a single hospitalist and patient over the course of a hospital stay. But the effect of the method of patient assignment on continuity often is overlooked.
A reasonable way to think about or measure continuity is to estimate the portion of patients seen by the group that see the same hospitalist for each daytime visit over the course of their stay. (Assume that in most HM groups the same hospitalist can’t make both day and night visits over the course of the hospital stay. So, just for simplicity, I’ve intentionally left night visits, including an initial admission visit at night, out of the continuity calculation.) Plug the numbers for your practice into the formula (see Figure 1, right) and see what you get.
If a hospitalist always works seven consecutive day shifts (e.g., a seven-on/seven-off schedule) and the hospitalist’s patients have an average LOS of 4.2 days, then 54% of patients will see the same hospitalist for all daytime visits, and 46% will experience at least one handoff. (To keep things simple, I’m ignoring the effect on continuity of patients being admitted by an “admitter” or nocturnist who doesn’t see the patient subsequently.)
Changing the number of consecutive day shifts a hospitalist works has the most significant impact on continuity, but just how many consecutive days can one work routinely before fatigue and burnout—not too mention increased errors and decreased patient satisfaction—become a problem? (Many hospitalists make the mistake of trying to stuff what might be a reasonable annual workload into the smallest number of shifts possible with the goal of maximizing the number of days off. That means each worked day will be very busy, making it really hard to work many consecutive days. But you always have the option of titrating out that same annual workload over more days so that each day is less busy and it becomes easier to work more consecutive days.)
An often-overlooked way to improve continuity without having to work more consecutive day shifts is to have a hospitalist who is early in their series of worked days take on more new admissions and consults, and perhaps exempt that doctor from taking on new referrals for the last day or two he or she is on service. Eric Howell, MD, FHM, an SHM board member, calls this method “slam and dwindle.” This has been the approach I’ve experienced my whole career, and it is hard for me to imagine doing it any other way.
Here’s how it might work: Let’s say Dr. Petty always works seven consecutive day shifts, and on the first day he picks up a list of patients remaining from the doctor he’s replacing. To keep things simple, let’s assume he’s not in a large group, and during his first day of seven days on service he accepts and “keeps” all new referrals to the practice. On each successive day, he might assume the care of some new patients, but none on days six and seven. This means he takes on a disproportionately large number of new referrals at the beginning of his consecutive worked days, or “front-loads” new referrals. And because many of these patients will discharge before the end of his seven days and he takes on no new patients on days six and seven, his census will drop a lot before he rotates off, which in turn means there will be few patients who will have to get to know a new doctor on the first day Dr. Petty starts his seven-off schedule.
This system of patient distribution means continuity improved without requiring Dr. Petty to work more consecutive day shifts. Even though he works seven consecutive days and his average (or median) LOS is 4.2, as in the example above, his continuity will be much better than 54%. In fact, as many as 70% to 80% of Dr. Petty’s patients will see him for every daytime visit during their stay.
Other benefits of assigning more patients early and none late in a series of worked days are that on his last day of service, he will have more time to “tee up” patients for the next doctor, including preparing for patients anticipated to discharge the next day (e.g., dictate discharge summary, complete paperwork, etc.), and might be able to wrap up a little earlier that day. And when rotating back on service, he will pick up a small list of patients left by Dr. Tench, maybe fewer than eight, rather than the group’s average daily load of 15 patients per doctor, so he will have the capacity to admit a lot of patients that day.
I think there are three main reasons this isn’t a more common approach:
- Many HM groups just haven’t considered it.
- HM groups might have a schedule that has all doctors rotate off/on the same days each week. For example, all doctors rotate off on Tuesdays and are replaced by new doctors on Wednesday. That makes it impossible to exempt a doctor from taking on new referrals on the last day of service because all of the group’s doctors have their last day on Tuesday. These groups could stagger the day each doctor rotates off—one on Monday, one on Tuesday, and so on.
- Every doctor is so busy each day that it wouldn’t be feasible to exempt any individual doctor from taking on new patients, even if they are off the next day.
Despite the difficulties implementing a system of front-loading new referrals, I think most hospitalists would find that they like it. Because it reduces handoffs, it reduces, at least modestly, the group’s overall workload and probably benefits the group’s quality and patient satisfaction. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Editor’s note: Third of a three-part series.
In the two monthly columns preceding this one, I’ve provided an overview of some ways hospitalist groups distribute new referrals among the providers. This month, I’ll review things that cause some groups to make exceptions to their typical method of distributing patients, and turn from how patients are distributed over 24 hours to thoughts about how they might be assigned over the course of consecutive days worked by a doctor.
Equitable Exceptions
There are a number of reasons groups decide to depart from their typical method of assigning patients. These include:
- “Bouncebacks”;
- One hospitalist is at the cap, others aren’t;
- Consult requested of a specific hospitalist;
- Hospitalists with unique skills (e.g., ICU expertise); and
- A patient “fires” the hospitalist.
There isn’t a standard “hospitalist way” of dealing with these issues, and each group will need to work out its own system. The most common of these issues is “bouncebacks.” Every group should try to have patients readmitted within three or four days of discharge go back to the discharging hospitalist. However, this proves difficult in many cases for several reasons, most commonly because the original discharging doctor might not be working when the patient returns.
The Alpha & Omega
Nearly every hospitalist practice makes some effort to maximize continuity between a single hospitalist and patient over the course of a hospital stay. But the effect of the method of patient assignment on continuity often is overlooked.
A reasonable way to think about or measure continuity is to estimate the portion of patients seen by the group that see the same hospitalist for each daytime visit over the course of their stay. (Assume that in most HM groups the same hospitalist can’t make both day and night visits over the course of the hospital stay. So, just for simplicity, I’ve intentionally left night visits, including an initial admission visit at night, out of the continuity calculation.) Plug the numbers for your practice into the formula (see Figure 1, right) and see what you get.
If a hospitalist always works seven consecutive day shifts (e.g., a seven-on/seven-off schedule) and the hospitalist’s patients have an average LOS of 4.2 days, then 54% of patients will see the same hospitalist for all daytime visits, and 46% will experience at least one handoff. (To keep things simple, I’m ignoring the effect on continuity of patients being admitted by an “admitter” or nocturnist who doesn’t see the patient subsequently.)
Changing the number of consecutive day shifts a hospitalist works has the most significant impact on continuity, but just how many consecutive days can one work routinely before fatigue and burnout—not too mention increased errors and decreased patient satisfaction—become a problem? (Many hospitalists make the mistake of trying to stuff what might be a reasonable annual workload into the smallest number of shifts possible with the goal of maximizing the number of days off. That means each worked day will be very busy, making it really hard to work many consecutive days. But you always have the option of titrating out that same annual workload over more days so that each day is less busy and it becomes easier to work more consecutive days.)
An often-overlooked way to improve continuity without having to work more consecutive day shifts is to have a hospitalist who is early in their series of worked days take on more new admissions and consults, and perhaps exempt that doctor from taking on new referrals for the last day or two he or she is on service. Eric Howell, MD, FHM, an SHM board member, calls this method “slam and dwindle.” This has been the approach I’ve experienced my whole career, and it is hard for me to imagine doing it any other way.
Here’s how it might work: Let’s say Dr. Petty always works seven consecutive day shifts, and on the first day he picks up a list of patients remaining from the doctor he’s replacing. To keep things simple, let’s assume he’s not in a large group, and during his first day of seven days on service he accepts and “keeps” all new referrals to the practice. On each successive day, he might assume the care of some new patients, but none on days six and seven. This means he takes on a disproportionately large number of new referrals at the beginning of his consecutive worked days, or “front-loads” new referrals. And because many of these patients will discharge before the end of his seven days and he takes on no new patients on days six and seven, his census will drop a lot before he rotates off, which in turn means there will be few patients who will have to get to know a new doctor on the first day Dr. Petty starts his seven-off schedule.
This system of patient distribution means continuity improved without requiring Dr. Petty to work more consecutive day shifts. Even though he works seven consecutive days and his average (or median) LOS is 4.2, as in the example above, his continuity will be much better than 54%. In fact, as many as 70% to 80% of Dr. Petty’s patients will see him for every daytime visit during their stay.
Other benefits of assigning more patients early and none late in a series of worked days are that on his last day of service, he will have more time to “tee up” patients for the next doctor, including preparing for patients anticipated to discharge the next day (e.g., dictate discharge summary, complete paperwork, etc.), and might be able to wrap up a little earlier that day. And when rotating back on service, he will pick up a small list of patients left by Dr. Tench, maybe fewer than eight, rather than the group’s average daily load of 15 patients per doctor, so he will have the capacity to admit a lot of patients that day.
I think there are three main reasons this isn’t a more common approach:
- Many HM groups just haven’t considered it.
- HM groups might have a schedule that has all doctors rotate off/on the same days each week. For example, all doctors rotate off on Tuesdays and are replaced by new doctors on Wednesday. That makes it impossible to exempt a doctor from taking on new referrals on the last day of service because all of the group’s doctors have their last day on Tuesday. These groups could stagger the day each doctor rotates off—one on Monday, one on Tuesday, and so on.
- Every doctor is so busy each day that it wouldn’t be feasible to exempt any individual doctor from taking on new patients, even if they are off the next day.
Despite the difficulties implementing a system of front-loading new referrals, I think most hospitalists would find that they like it. Because it reduces handoffs, it reduces, at least modestly, the group’s overall workload and probably benefits the group’s quality and patient satisfaction. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Editor’s note: Third of a three-part series.
In the two monthly columns preceding this one, I’ve provided an overview of some ways hospitalist groups distribute new referrals among the providers. This month, I’ll review things that cause some groups to make exceptions to their typical method of distributing patients, and turn from how patients are distributed over 24 hours to thoughts about how they might be assigned over the course of consecutive days worked by a doctor.
Equitable Exceptions
There are a number of reasons groups decide to depart from their typical method of assigning patients. These include:
- “Bouncebacks”;
- One hospitalist is at the cap, others aren’t;
- Consult requested of a specific hospitalist;
- Hospitalists with unique skills (e.g., ICU expertise); and
- A patient “fires” the hospitalist.
There isn’t a standard “hospitalist way” of dealing with these issues, and each group will need to work out its own system. The most common of these issues is “bouncebacks.” Every group should try to have patients readmitted within three or four days of discharge go back to the discharging hospitalist. However, this proves difficult in many cases for several reasons, most commonly because the original discharging doctor might not be working when the patient returns.
The Alpha & Omega
Nearly every hospitalist practice makes some effort to maximize continuity between a single hospitalist and patient over the course of a hospital stay. But the effect of the method of patient assignment on continuity often is overlooked.
A reasonable way to think about or measure continuity is to estimate the portion of patients seen by the group that see the same hospitalist for each daytime visit over the course of their stay. (Assume that in most HM groups the same hospitalist can’t make both day and night visits over the course of the hospital stay. So, just for simplicity, I’ve intentionally left night visits, including an initial admission visit at night, out of the continuity calculation.) Plug the numbers for your practice into the formula (see Figure 1, right) and see what you get.
If a hospitalist always works seven consecutive day shifts (e.g., a seven-on/seven-off schedule) and the hospitalist’s patients have an average LOS of 4.2 days, then 54% of patients will see the same hospitalist for all daytime visits, and 46% will experience at least one handoff. (To keep things simple, I’m ignoring the effect on continuity of patients being admitted by an “admitter” or nocturnist who doesn’t see the patient subsequently.)
Changing the number of consecutive day shifts a hospitalist works has the most significant impact on continuity, but just how many consecutive days can one work routinely before fatigue and burnout—not too mention increased errors and decreased patient satisfaction—become a problem? (Many hospitalists make the mistake of trying to stuff what might be a reasonable annual workload into the smallest number of shifts possible with the goal of maximizing the number of days off. That means each worked day will be very busy, making it really hard to work many consecutive days. But you always have the option of titrating out that same annual workload over more days so that each day is less busy and it becomes easier to work more consecutive days.)
An often-overlooked way to improve continuity without having to work more consecutive day shifts is to have a hospitalist who is early in their series of worked days take on more new admissions and consults, and perhaps exempt that doctor from taking on new referrals for the last day or two he or she is on service. Eric Howell, MD, FHM, an SHM board member, calls this method “slam and dwindle.” This has been the approach I’ve experienced my whole career, and it is hard for me to imagine doing it any other way.
Here’s how it might work: Let’s say Dr. Petty always works seven consecutive day shifts, and on the first day he picks up a list of patients remaining from the doctor he’s replacing. To keep things simple, let’s assume he’s not in a large group, and during his first day of seven days on service he accepts and “keeps” all new referrals to the practice. On each successive day, he might assume the care of some new patients, but none on days six and seven. This means he takes on a disproportionately large number of new referrals at the beginning of his consecutive worked days, or “front-loads” new referrals. And because many of these patients will discharge before the end of his seven days and he takes on no new patients on days six and seven, his census will drop a lot before he rotates off, which in turn means there will be few patients who will have to get to know a new doctor on the first day Dr. Petty starts his seven-off schedule.
This system of patient distribution means continuity improved without requiring Dr. Petty to work more consecutive day shifts. Even though he works seven consecutive days and his average (or median) LOS is 4.2, as in the example above, his continuity will be much better than 54%. In fact, as many as 70% to 80% of Dr. Petty’s patients will see him for every daytime visit during their stay.
Other benefits of assigning more patients early and none late in a series of worked days are that on his last day of service, he will have more time to “tee up” patients for the next doctor, including preparing for patients anticipated to discharge the next day (e.g., dictate discharge summary, complete paperwork, etc.), and might be able to wrap up a little earlier that day. And when rotating back on service, he will pick up a small list of patients left by Dr. Tench, maybe fewer than eight, rather than the group’s average daily load of 15 patients per doctor, so he will have the capacity to admit a lot of patients that day.
I think there are three main reasons this isn’t a more common approach:
- Many HM groups just haven’t considered it.
- HM groups might have a schedule that has all doctors rotate off/on the same days each week. For example, all doctors rotate off on Tuesdays and are replaced by new doctors on Wednesday. That makes it impossible to exempt a doctor from taking on new referrals on the last day of service because all of the group’s doctors have their last day on Tuesday. These groups could stagger the day each doctor rotates off—one on Monday, one on Tuesday, and so on.
- Every doctor is so busy each day that it wouldn’t be feasible to exempt any individual doctor from taking on new patients, even if they are off the next day.
Despite the difficulties implementing a system of front-loading new referrals, I think most hospitalists would find that they like it. Because it reduces handoffs, it reduces, at least modestly, the group’s overall workload and probably benefits the group’s quality and patient satisfaction. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
New Referral Distribution
Editor’s note: Second of a three-part series.
As I mentioned last month, there isn’t a proven best method to use when distributing new referrals among your group’s providers. The popular methods fall along a continuum of being focused on daily, or continuous, leveling of patient loads between providers (“load leveling”) at one end; at the other end of the continuum is having a doctor be “on” for all new referrals for a predetermined time period, and accepting that patient volumes might be uneven day to day but tend to even out over long periods.
There might not be any reason to change your group’s approach to patient assignment, but you should always be thinking about how your own methods might be changed or improved. I have shared (“Bigger Isn’t Always Better,” June 2009, p. 46) my concern that some groups invest far too much time in a morning load-leveling and handoff conference. Make sure your group is using only as much time as needed.
Air-Traffic Controllers
Many large groups (e.g., more than 15 full-time equivalents) that assign patients to providers in sequence, like dealing a deck of cards, have a designated provider who holds the triage pager and serves as “air-traffic controller.” This person typically takes incoming calls about all new referrals, jots down the relevant clinical data, keeps track of which hospitalist is due to take the next patient, pages that person, and repeats the clinical information. As I’ve written before (“How to Hire and Use Clerical Staff,” June 2007, p. 73), many practices have found that during business hours, they can hand this role to a clerical person who simply takes down the name and phone number of the doctor making the referral, then pages that information to the hospitalist due to get the next patient. The hospitalist then calls and speaks directly with the referring doctor.
Small- to medium-sized groups can eliminate entirely the need for any such “air-traffic control” function if they assign all new referrals to a single doctor for specified periods of time. For example, from 7 a.m. to 3 p.m. today, all new referrals go to Dr. Glass, and from 3 p.m. to 11 p.m., they go to Dr. Cage.
Admitter-Rounder Duties
Many—maybe most?—large groups separate daytime admitter and rounder functions so that on any given day, a hospitalist does one but not both. The principal advantages of this approach are reducing the stress on, and possibly increasing the efficiency of, rounding doctors by shielding them from the unpredictable and time-consuming interruption of needing to admit a new patient. And a daytime doctor who only does admissions might be able to start seeing a patient in the ED more quickly than one who is busy making rounds.
Any increased availability of admitters to the ED could be offset by their lack of surge capacity leading to a bottleneck in ED throughput when there are many patients to admit at the same time and a limited number of admitters (often only one). Such a bottleneck would be much less likely if all daytime doctors (i.e., the rounders) were available to see admissions rather than just admitters.
Continuity of care suffers when a group has separate admitters and rounders, because no patients will be seen by the same doctor on the day of admission and the day following. This method requires a handoff from the day of admission to the next day. Such a handoff might be unavoidable for patients admitted during the night, but this doesn’t have to occur when patients are admitted during the daytime.
Who’s Seeing this Patient?
It seems to make sense to wait until each morning to distribute patients. That allows the practice to know just how many new patients there are, and they can be distributed according to complexity and whether a hospitalist has formed a previous relationship with that patient. But it means that no one at the hospital will know which hospitalist is caring for the patient until later in the morning. For example, if the radiologist is over-reading a study done during the night and finds something worthy of a phone call to the hospitalist, no one is sure who should get the call. A patient might develop hypoglycemia shortly after the hospitalist night shift is over, but the nurse doesn’t know which hospitalist to call.
And, perhaps most importantly, if patients aren’t distributed until the start of the day shift, the night hospitalist can’t tell the patient and family which hospitalist to expect the next morning. To test the significance of this issue, I conducted an experiment while working our group’s late-evening admitter shift. I concluded my visit with each admitted patient by explaining, “I am on-call for our group tonight, so I will be off recovering tomorrow. Therefore, I won’t see you again, but one of my partners will take over in the morning. Do you have any questions for me?” Every patient I admitted had the same question. “What is that doctor’s name?”
How does your group answer a patient who asks which hospitalist will be in the next day? If your method is load-leveling in the morning, then the best answer your night admitting doctor can give is probably to say: “I don’t know which of my partners will be in. There are several working tomorrow, and at the start of the day, they will divide up the patients who come in tonight depending on how busy each of them is. But all the doctors in our group are terrific and will take good care of you.”
I’m told the same thing when I get my hair cut: You’ll get whichever “hair artist” is up next. I put up with it at the hair place because it costs less than $15. But I still find it a little irritating. I’m sure all the barbers aren’t equally skilled or diligent, and I want the best one. (Maybe I shouldn’t care since there isn’t much that can be done with my hair.) I’m pretty sure patients feel the same way about which doctor they get. The public is convinced there is a wide variety in the quality of doctors, and they want a good one. If you have to tell them theirs is being assigned by lottery, they won’t be as happy than if you can provide the name and a little information about the doctor they can expect to see the next day.
When the patients I admit late last evening ask who would see them the next day, I’m glad when I can provide a name and a little more information. I say something like, “I won’t see you after tonight, but my partner, Dr. Shawn Lee, will be instead. That means you’re getting an upgrade! Not only is he a really nice guy, he’s voted one of Seattle’s best doctors every year. He’ll do a great job for you.”
To make this communication effective, the night doctor has to know which hospitalist takes over the next morning and has a list indicating which day doctor will get the first, second, third new patient, and so on, admitted during the night. This is possible if patients are assigned by a predetermined algorithm, or if the day doctors have their load-leveling meeting at the end of each day shift, rather than in the morning, to create a list telling the night doctor which day hospitalist he should admit the first and subsequent patents to. That way, the night doctor can write in the admitting orders at 1 a.m. “admit to Dr. X.” This eliminates confusion on the part of other hospital staff who need to know who to call about a patient after the start of the day shift.
Next month I will look at special circumstances, and some pros and cons of having an individual hospitalist take on the care of more patients at the beginning of consecutive day shifts, and exempting them from taking on new patients on the last day or two before rotating off. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Editor’s note: Second of a three-part series.
As I mentioned last month, there isn’t a proven best method to use when distributing new referrals among your group’s providers. The popular methods fall along a continuum of being focused on daily, or continuous, leveling of patient loads between providers (“load leveling”) at one end; at the other end of the continuum is having a doctor be “on” for all new referrals for a predetermined time period, and accepting that patient volumes might be uneven day to day but tend to even out over long periods.
There might not be any reason to change your group’s approach to patient assignment, but you should always be thinking about how your own methods might be changed or improved. I have shared (“Bigger Isn’t Always Better,” June 2009, p. 46) my concern that some groups invest far too much time in a morning load-leveling and handoff conference. Make sure your group is using only as much time as needed.
Air-Traffic Controllers
Many large groups (e.g., more than 15 full-time equivalents) that assign patients to providers in sequence, like dealing a deck of cards, have a designated provider who holds the triage pager and serves as “air-traffic controller.” This person typically takes incoming calls about all new referrals, jots down the relevant clinical data, keeps track of which hospitalist is due to take the next patient, pages that person, and repeats the clinical information. As I’ve written before (“How to Hire and Use Clerical Staff,” June 2007, p. 73), many practices have found that during business hours, they can hand this role to a clerical person who simply takes down the name and phone number of the doctor making the referral, then pages that information to the hospitalist due to get the next patient. The hospitalist then calls and speaks directly with the referring doctor.
Small- to medium-sized groups can eliminate entirely the need for any such “air-traffic control” function if they assign all new referrals to a single doctor for specified periods of time. For example, from 7 a.m. to 3 p.m. today, all new referrals go to Dr. Glass, and from 3 p.m. to 11 p.m., they go to Dr. Cage.
Admitter-Rounder Duties
Many—maybe most?—large groups separate daytime admitter and rounder functions so that on any given day, a hospitalist does one but not both. The principal advantages of this approach are reducing the stress on, and possibly increasing the efficiency of, rounding doctors by shielding them from the unpredictable and time-consuming interruption of needing to admit a new patient. And a daytime doctor who only does admissions might be able to start seeing a patient in the ED more quickly than one who is busy making rounds.
Any increased availability of admitters to the ED could be offset by their lack of surge capacity leading to a bottleneck in ED throughput when there are many patients to admit at the same time and a limited number of admitters (often only one). Such a bottleneck would be much less likely if all daytime doctors (i.e., the rounders) were available to see admissions rather than just admitters.
Continuity of care suffers when a group has separate admitters and rounders, because no patients will be seen by the same doctor on the day of admission and the day following. This method requires a handoff from the day of admission to the next day. Such a handoff might be unavoidable for patients admitted during the night, but this doesn’t have to occur when patients are admitted during the daytime.
Who’s Seeing this Patient?
It seems to make sense to wait until each morning to distribute patients. That allows the practice to know just how many new patients there are, and they can be distributed according to complexity and whether a hospitalist has formed a previous relationship with that patient. But it means that no one at the hospital will know which hospitalist is caring for the patient until later in the morning. For example, if the radiologist is over-reading a study done during the night and finds something worthy of a phone call to the hospitalist, no one is sure who should get the call. A patient might develop hypoglycemia shortly after the hospitalist night shift is over, but the nurse doesn’t know which hospitalist to call.
And, perhaps most importantly, if patients aren’t distributed until the start of the day shift, the night hospitalist can’t tell the patient and family which hospitalist to expect the next morning. To test the significance of this issue, I conducted an experiment while working our group’s late-evening admitter shift. I concluded my visit with each admitted patient by explaining, “I am on-call for our group tonight, so I will be off recovering tomorrow. Therefore, I won’t see you again, but one of my partners will take over in the morning. Do you have any questions for me?” Every patient I admitted had the same question. “What is that doctor’s name?”
How does your group answer a patient who asks which hospitalist will be in the next day? If your method is load-leveling in the morning, then the best answer your night admitting doctor can give is probably to say: “I don’t know which of my partners will be in. There are several working tomorrow, and at the start of the day, they will divide up the patients who come in tonight depending on how busy each of them is. But all the doctors in our group are terrific and will take good care of you.”
I’m told the same thing when I get my hair cut: You’ll get whichever “hair artist” is up next. I put up with it at the hair place because it costs less than $15. But I still find it a little irritating. I’m sure all the barbers aren’t equally skilled or diligent, and I want the best one. (Maybe I shouldn’t care since there isn’t much that can be done with my hair.) I’m pretty sure patients feel the same way about which doctor they get. The public is convinced there is a wide variety in the quality of doctors, and they want a good one. If you have to tell them theirs is being assigned by lottery, they won’t be as happy than if you can provide the name and a little information about the doctor they can expect to see the next day.
When the patients I admit late last evening ask who would see them the next day, I’m glad when I can provide a name and a little more information. I say something like, “I won’t see you after tonight, but my partner, Dr. Shawn Lee, will be instead. That means you’re getting an upgrade! Not only is he a really nice guy, he’s voted one of Seattle’s best doctors every year. He’ll do a great job for you.”
To make this communication effective, the night doctor has to know which hospitalist takes over the next morning and has a list indicating which day doctor will get the first, second, third new patient, and so on, admitted during the night. This is possible if patients are assigned by a predetermined algorithm, or if the day doctors have their load-leveling meeting at the end of each day shift, rather than in the morning, to create a list telling the night doctor which day hospitalist he should admit the first and subsequent patents to. That way, the night doctor can write in the admitting orders at 1 a.m. “admit to Dr. X.” This eliminates confusion on the part of other hospital staff who need to know who to call about a patient after the start of the day shift.
Next month I will look at special circumstances, and some pros and cons of having an individual hospitalist take on the care of more patients at the beginning of consecutive day shifts, and exempting them from taking on new patients on the last day or two before rotating off. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Editor’s note: Second of a three-part series.
As I mentioned last month, there isn’t a proven best method to use when distributing new referrals among your group’s providers. The popular methods fall along a continuum of being focused on daily, or continuous, leveling of patient loads between providers (“load leveling”) at one end; at the other end of the continuum is having a doctor be “on” for all new referrals for a predetermined time period, and accepting that patient volumes might be uneven day to day but tend to even out over long periods.
There might not be any reason to change your group’s approach to patient assignment, but you should always be thinking about how your own methods might be changed or improved. I have shared (“Bigger Isn’t Always Better,” June 2009, p. 46) my concern that some groups invest far too much time in a morning load-leveling and handoff conference. Make sure your group is using only as much time as needed.
Air-Traffic Controllers
Many large groups (e.g., more than 15 full-time equivalents) that assign patients to providers in sequence, like dealing a deck of cards, have a designated provider who holds the triage pager and serves as “air-traffic controller.” This person typically takes incoming calls about all new referrals, jots down the relevant clinical data, keeps track of which hospitalist is due to take the next patient, pages that person, and repeats the clinical information. As I’ve written before (“How to Hire and Use Clerical Staff,” June 2007, p. 73), many practices have found that during business hours, they can hand this role to a clerical person who simply takes down the name and phone number of the doctor making the referral, then pages that information to the hospitalist due to get the next patient. The hospitalist then calls and speaks directly with the referring doctor.
Small- to medium-sized groups can eliminate entirely the need for any such “air-traffic control” function if they assign all new referrals to a single doctor for specified periods of time. For example, from 7 a.m. to 3 p.m. today, all new referrals go to Dr. Glass, and from 3 p.m. to 11 p.m., they go to Dr. Cage.
Admitter-Rounder Duties
Many—maybe most?—large groups separate daytime admitter and rounder functions so that on any given day, a hospitalist does one but not both. The principal advantages of this approach are reducing the stress on, and possibly increasing the efficiency of, rounding doctors by shielding them from the unpredictable and time-consuming interruption of needing to admit a new patient. And a daytime doctor who only does admissions might be able to start seeing a patient in the ED more quickly than one who is busy making rounds.
Any increased availability of admitters to the ED could be offset by their lack of surge capacity leading to a bottleneck in ED throughput when there are many patients to admit at the same time and a limited number of admitters (often only one). Such a bottleneck would be much less likely if all daytime doctors (i.e., the rounders) were available to see admissions rather than just admitters.
Continuity of care suffers when a group has separate admitters and rounders, because no patients will be seen by the same doctor on the day of admission and the day following. This method requires a handoff from the day of admission to the next day. Such a handoff might be unavoidable for patients admitted during the night, but this doesn’t have to occur when patients are admitted during the daytime.
Who’s Seeing this Patient?
It seems to make sense to wait until each morning to distribute patients. That allows the practice to know just how many new patients there are, and they can be distributed according to complexity and whether a hospitalist has formed a previous relationship with that patient. But it means that no one at the hospital will know which hospitalist is caring for the patient until later in the morning. For example, if the radiologist is over-reading a study done during the night and finds something worthy of a phone call to the hospitalist, no one is sure who should get the call. A patient might develop hypoglycemia shortly after the hospitalist night shift is over, but the nurse doesn’t know which hospitalist to call.
And, perhaps most importantly, if patients aren’t distributed until the start of the day shift, the night hospitalist can’t tell the patient and family which hospitalist to expect the next morning. To test the significance of this issue, I conducted an experiment while working our group’s late-evening admitter shift. I concluded my visit with each admitted patient by explaining, “I am on-call for our group tonight, so I will be off recovering tomorrow. Therefore, I won’t see you again, but one of my partners will take over in the morning. Do you have any questions for me?” Every patient I admitted had the same question. “What is that doctor’s name?”
How does your group answer a patient who asks which hospitalist will be in the next day? If your method is load-leveling in the morning, then the best answer your night admitting doctor can give is probably to say: “I don’t know which of my partners will be in. There are several working tomorrow, and at the start of the day, they will divide up the patients who come in tonight depending on how busy each of them is. But all the doctors in our group are terrific and will take good care of you.”
I’m told the same thing when I get my hair cut: You’ll get whichever “hair artist” is up next. I put up with it at the hair place because it costs less than $15. But I still find it a little irritating. I’m sure all the barbers aren’t equally skilled or diligent, and I want the best one. (Maybe I shouldn’t care since there isn’t much that can be done with my hair.) I’m pretty sure patients feel the same way about which doctor they get. The public is convinced there is a wide variety in the quality of doctors, and they want a good one. If you have to tell them theirs is being assigned by lottery, they won’t be as happy than if you can provide the name and a little information about the doctor they can expect to see the next day.
When the patients I admit late last evening ask who would see them the next day, I’m glad when I can provide a name and a little more information. I say something like, “I won’t see you after tonight, but my partner, Dr. Shawn Lee, will be instead. That means you’re getting an upgrade! Not only is he a really nice guy, he’s voted one of Seattle’s best doctors every year. He’ll do a great job for you.”
To make this communication effective, the night doctor has to know which hospitalist takes over the next morning and has a list indicating which day doctor will get the first, second, third new patient, and so on, admitted during the night. This is possible if patients are assigned by a predetermined algorithm, or if the day doctors have their load-leveling meeting at the end of each day shift, rather than in the morning, to create a list telling the night doctor which day hospitalist he should admit the first and subsequent patents to. That way, the night doctor can write in the admitting orders at 1 a.m. “admit to Dr. X.” This eliminates confusion on the part of other hospital staff who need to know who to call about a patient after the start of the day shift.
Next month I will look at special circumstances, and some pros and cons of having an individual hospitalist take on the care of more patients at the beginning of consecutive day shifts, and exempting them from taking on new patients on the last day or two before rotating off. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Should Hospitalists Who Fail to Provide a Standard of Care Be Paid for Subsequent Care?
A 72-year-old male with a history of CHF is admitted for elective total hip arthroplasty. On postoperative day one, he develops dyspnea and hypoxia, and is diagnosed with acute pulmonary edema by the hospitalist co-managing his care. Furosemide is prescribed, and he improves, and by day four is ready for discharge following another dose of diuretics. Overnight, he develops acute onset of shortness of breath and is diagnosed with a pulmonary embolism (PE). Under new regulations, the hospital will not be reimbursed for the extra cost associated with subsequent patient care. Should the hospitalist be paid for the subsequent care?
PRO
Nonpayment won’t improve quality or significantly decrease costs
The real essence of the question raised in the clinical case above is “Should doctors profit from errors?” The answer might be “It’s better than the alternative.” Allow me to explain. There essentially are two reasons to withhold payment in this scenario: one, as a mechanism for promoting quality; two, as a mechanism for decreasing costs to the payor.
The quality argument assumes the physician will deliver higher-quality care (i.e., prescribe chemical thromboprophylaxis) if a threat of nonpayment exists. This concept is simply hogwash. If expensive medical malpractice threats fail as quality-improvement (QI) mechanisms, it is absurd to think withholding a few subsequent-care charges will generate better results.
The key issue is the type of error involved. As defined by Lucien Leape, MD, in his celebrated 1994 article on medical errors, “mistakes” reflect failures during attentional behaviors, or incorrect choices.1 “Slips” reflect lapses in concentration. “Slips occur in the face of competing sensory or emotional distractions, fatigue, and stress,” and “reducing the risk of slips requires attention to the designs of protocols, devices, and work environments.”
Misjudging the type of error—in this case, a slip (find me a hospitalist who doesn’t know total hip arthroplasty requires thrombophrophylaxis)—and misapplying corrective actions will have little to no effect on outcomes. Thus, pay-withholding schemes can have a negative net effect by diverting resources from QI projects that truly improve patient outcomes.
Withholding payment in this case generates approximately $160 in direct savings to the payor (assuming Medicare payments for one 99233 and two 99232 subsequent care visits), yet the operational costs are not negligible and must be factored into the equation. The payor needs to first determine who is truly at fault: the hospitalist or the orthopedic surgeon. Answering that question requires the payor to review the co-management agreement, perhaps aided by an attorney. That’s a costly endeavor.
For the sake of argument, let’s assume in this case the hospitalist is at fault. The next step is determining if the hospitalist who failed to prescribe prophylaxis prior to the PE is the same hospitalist caring for the patient after the PE. It is inappropriate to withhold payment to hospitalist A if hospitalist B made the error. Again, significant manpower will be required to determine fault, as this is not information one finds on a UB-04 claim form submitted to Medicare.
Further eroding the $160 savings is the cost of determining whether a contraindication exists: Bleeding ulcer? Subdural hematoma? Heparinoid allergy? Let us not forget the additional costs in copying, shipping, warehousing, and eventual shredding of the records. One can readily see that the operational costs can quickly negate the $160 anticipated savings. In fact, it’s likely a negative return on investment.
Clearly, withholding payment in this scenario is an ineffective mechanism for improving quality or decreasing cost. I am not generally a proponent of rewarding failure, and perhaps as we usher in a new era of healthcare reform, the system will be redesigned in such a way that better aligns quality and cost-control measures. However, under the current system, payment denial as outlined above likely does more harm than good.
CON
Withhold payment when medical errors are easily identifiable
When I first learned of the proposal to withhold Medicare payment for hospital-acquired conditions (HACs), I had mixed emotions. On the one hand, I firmly believe that physicians should be accountable for their work; on the other hand, this policy seems to conflict sharply with the “no blame” mantra that has been prevalent in patient safety for more than a decade.2 More recently, though, many have argued for balancing the pursuit of system fixes for quality and patient-safety issues with the development of a culture of accountability.3
In theory, the HACs should meet the following criteria: They should be high-cost conditions, high-volume conditions, or both; they should be identifiable through ICD-9-CM coding as complicating conditions (CCs) or major complicating conditions (MCCs) that result in a higher-paying MS-DRG; and they should be reasonably preventable through the application of evidence-based guidelines. Some HACs are jaw-dropping lapses in care (e.g., leaving foreign bodies in during surgery). Other HACs seem to me to be much less preventable, especially fall injuries and catheter-associated urinary tract infections (UTIs). Several experts have written eloquently regarding the limitations of these new measures, particularly emphasizing the potential for increased administrative burden on hospitals and the potential for unintended consequences.4,5
However, in the case described above involving a hospitalist, I have no reservations in limiting payment to the provider. To me, failing to prescribe VTE prophylaxis in an elderly, immobilized, post-op hip replacement patient with a CHF exacerbation is the hospitalist’s equivalent to a surgeon leaving behind a sponge in an appendectomy. It also meets the elements outlined in the HAC withholding program:
- It is high-cost. The 2007 MS-DRG payment for elective hip arthroplasty was $9,863, but adding an MCC increased that cost by one-third.6
- It is readily identifiable, though one concern might be that hospitals would perform unnecessary pre-operative testing to identify asymptomatic DVT, incurring increased testing and treatment costs and increasing the incidence of bleeding complications.
- It is very preventable. Without thromboprophylaxis, 40% to 60% of hip arthroplasty patients will develop an asymptomatic DVT, and 1 in 300 will die from a PE. However, such fatal events are exceedingly rare with appropriate prevention.7
Ultimately, I think a policy of nonpayment for this case keeps with the culture of accountability we need to foster in healthcare. The financial implications of nonpayment will drive hospital innovation and force the hospital to police provider behavior in more effective ways. This is likely to be a painful process, similar to the tribulations experienced with implementing pay-for-performance programs. The Centers for Medicare and Medicaid Services (CMS) needs to be flexible in adding—and removing—new HACs based on good evidence.
Regardless, the goal of achieving a safer, more effective healthcare system remains.
References
- Leape LL. Error in medicine. JAMA. 1994;272(23):1851-1857.
- Institute of Medicine. To Err Is Human: Building a Safer Healthcare System. Washington, D.C.: National Academies Press; 2000.
- Wachter RM, Pronovost PJ. Balancing “no blame” with accountability in patient safety. N Engl J Med. 2009;361:1401-1406.
- Saint S, Meddings JA, Calfee D, Kowalski CP, Krein SL. Catheter-associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877-884.
- Inouye SK, Brown CJ, Tinetti ME. Medicare nonpayment, hospital falls, and unintended consequences. N Engl J Med. 2009;360(23):2390-2393.
- Wachter RM, Foster NE, Dudley RA. Medicare’s decision to withhold payment for hospital errors: the devil is in the det. Jt Comm J Qual Patient Saf. 2008;34(2):116-123.
- Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):381S-453S.
The opinions expressed herein are those of the authors and do not represent those of SHM or The Hospitalist.
A 72-year-old male with a history of CHF is admitted for elective total hip arthroplasty. On postoperative day one, he develops dyspnea and hypoxia, and is diagnosed with acute pulmonary edema by the hospitalist co-managing his care. Furosemide is prescribed, and he improves, and by day four is ready for discharge following another dose of diuretics. Overnight, he develops acute onset of shortness of breath and is diagnosed with a pulmonary embolism (PE). Under new regulations, the hospital will not be reimbursed for the extra cost associated with subsequent patient care. Should the hospitalist be paid for the subsequent care?
PRO
Nonpayment won’t improve quality or significantly decrease costs
The real essence of the question raised in the clinical case above is “Should doctors profit from errors?” The answer might be “It’s better than the alternative.” Allow me to explain. There essentially are two reasons to withhold payment in this scenario: one, as a mechanism for promoting quality; two, as a mechanism for decreasing costs to the payor.
The quality argument assumes the physician will deliver higher-quality care (i.e., prescribe chemical thromboprophylaxis) if a threat of nonpayment exists. This concept is simply hogwash. If expensive medical malpractice threats fail as quality-improvement (QI) mechanisms, it is absurd to think withholding a few subsequent-care charges will generate better results.
The key issue is the type of error involved. As defined by Lucien Leape, MD, in his celebrated 1994 article on medical errors, “mistakes” reflect failures during attentional behaviors, or incorrect choices.1 “Slips” reflect lapses in concentration. “Slips occur in the face of competing sensory or emotional distractions, fatigue, and stress,” and “reducing the risk of slips requires attention to the designs of protocols, devices, and work environments.”
Misjudging the type of error—in this case, a slip (find me a hospitalist who doesn’t know total hip arthroplasty requires thrombophrophylaxis)—and misapplying corrective actions will have little to no effect on outcomes. Thus, pay-withholding schemes can have a negative net effect by diverting resources from QI projects that truly improve patient outcomes.
Withholding payment in this case generates approximately $160 in direct savings to the payor (assuming Medicare payments for one 99233 and two 99232 subsequent care visits), yet the operational costs are not negligible and must be factored into the equation. The payor needs to first determine who is truly at fault: the hospitalist or the orthopedic surgeon. Answering that question requires the payor to review the co-management agreement, perhaps aided by an attorney. That’s a costly endeavor.
For the sake of argument, let’s assume in this case the hospitalist is at fault. The next step is determining if the hospitalist who failed to prescribe prophylaxis prior to the PE is the same hospitalist caring for the patient after the PE. It is inappropriate to withhold payment to hospitalist A if hospitalist B made the error. Again, significant manpower will be required to determine fault, as this is not information one finds on a UB-04 claim form submitted to Medicare.
Further eroding the $160 savings is the cost of determining whether a contraindication exists: Bleeding ulcer? Subdural hematoma? Heparinoid allergy? Let us not forget the additional costs in copying, shipping, warehousing, and eventual shredding of the records. One can readily see that the operational costs can quickly negate the $160 anticipated savings. In fact, it’s likely a negative return on investment.
Clearly, withholding payment in this scenario is an ineffective mechanism for improving quality or decreasing cost. I am not generally a proponent of rewarding failure, and perhaps as we usher in a new era of healthcare reform, the system will be redesigned in such a way that better aligns quality and cost-control measures. However, under the current system, payment denial as outlined above likely does more harm than good.
CON
Withhold payment when medical errors are easily identifiable
When I first learned of the proposal to withhold Medicare payment for hospital-acquired conditions (HACs), I had mixed emotions. On the one hand, I firmly believe that physicians should be accountable for their work; on the other hand, this policy seems to conflict sharply with the “no blame” mantra that has been prevalent in patient safety for more than a decade.2 More recently, though, many have argued for balancing the pursuit of system fixes for quality and patient-safety issues with the development of a culture of accountability.3
In theory, the HACs should meet the following criteria: They should be high-cost conditions, high-volume conditions, or both; they should be identifiable through ICD-9-CM coding as complicating conditions (CCs) or major complicating conditions (MCCs) that result in a higher-paying MS-DRG; and they should be reasonably preventable through the application of evidence-based guidelines. Some HACs are jaw-dropping lapses in care (e.g., leaving foreign bodies in during surgery). Other HACs seem to me to be much less preventable, especially fall injuries and catheter-associated urinary tract infections (UTIs). Several experts have written eloquently regarding the limitations of these new measures, particularly emphasizing the potential for increased administrative burden on hospitals and the potential for unintended consequences.4,5
However, in the case described above involving a hospitalist, I have no reservations in limiting payment to the provider. To me, failing to prescribe VTE prophylaxis in an elderly, immobilized, post-op hip replacement patient with a CHF exacerbation is the hospitalist’s equivalent to a surgeon leaving behind a sponge in an appendectomy. It also meets the elements outlined in the HAC withholding program:
- It is high-cost. The 2007 MS-DRG payment for elective hip arthroplasty was $9,863, but adding an MCC increased that cost by one-third.6
- It is readily identifiable, though one concern might be that hospitals would perform unnecessary pre-operative testing to identify asymptomatic DVT, incurring increased testing and treatment costs and increasing the incidence of bleeding complications.
- It is very preventable. Without thromboprophylaxis, 40% to 60% of hip arthroplasty patients will develop an asymptomatic DVT, and 1 in 300 will die from a PE. However, such fatal events are exceedingly rare with appropriate prevention.7
Ultimately, I think a policy of nonpayment for this case keeps with the culture of accountability we need to foster in healthcare. The financial implications of nonpayment will drive hospital innovation and force the hospital to police provider behavior in more effective ways. This is likely to be a painful process, similar to the tribulations experienced with implementing pay-for-performance programs. The Centers for Medicare and Medicaid Services (CMS) needs to be flexible in adding—and removing—new HACs based on good evidence.
Regardless, the goal of achieving a safer, more effective healthcare system remains.
References
- Leape LL. Error in medicine. JAMA. 1994;272(23):1851-1857.
- Institute of Medicine. To Err Is Human: Building a Safer Healthcare System. Washington, D.C.: National Academies Press; 2000.
- Wachter RM, Pronovost PJ. Balancing “no blame” with accountability in patient safety. N Engl J Med. 2009;361:1401-1406.
- Saint S, Meddings JA, Calfee D, Kowalski CP, Krein SL. Catheter-associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877-884.
- Inouye SK, Brown CJ, Tinetti ME. Medicare nonpayment, hospital falls, and unintended consequences. N Engl J Med. 2009;360(23):2390-2393.
- Wachter RM, Foster NE, Dudley RA. Medicare’s decision to withhold payment for hospital errors: the devil is in the det. Jt Comm J Qual Patient Saf. 2008;34(2):116-123.
- Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):381S-453S.
The opinions expressed herein are those of the authors and do not represent those of SHM or The Hospitalist.
A 72-year-old male with a history of CHF is admitted for elective total hip arthroplasty. On postoperative day one, he develops dyspnea and hypoxia, and is diagnosed with acute pulmonary edema by the hospitalist co-managing his care. Furosemide is prescribed, and he improves, and by day four is ready for discharge following another dose of diuretics. Overnight, he develops acute onset of shortness of breath and is diagnosed with a pulmonary embolism (PE). Under new regulations, the hospital will not be reimbursed for the extra cost associated with subsequent patient care. Should the hospitalist be paid for the subsequent care?
PRO
Nonpayment won’t improve quality or significantly decrease costs
The real essence of the question raised in the clinical case above is “Should doctors profit from errors?” The answer might be “It’s better than the alternative.” Allow me to explain. There essentially are two reasons to withhold payment in this scenario: one, as a mechanism for promoting quality; two, as a mechanism for decreasing costs to the payor.
The quality argument assumes the physician will deliver higher-quality care (i.e., prescribe chemical thromboprophylaxis) if a threat of nonpayment exists. This concept is simply hogwash. If expensive medical malpractice threats fail as quality-improvement (QI) mechanisms, it is absurd to think withholding a few subsequent-care charges will generate better results.
The key issue is the type of error involved. As defined by Lucien Leape, MD, in his celebrated 1994 article on medical errors, “mistakes” reflect failures during attentional behaviors, or incorrect choices.1 “Slips” reflect lapses in concentration. “Slips occur in the face of competing sensory or emotional distractions, fatigue, and stress,” and “reducing the risk of slips requires attention to the designs of protocols, devices, and work environments.”
Misjudging the type of error—in this case, a slip (find me a hospitalist who doesn’t know total hip arthroplasty requires thrombophrophylaxis)—and misapplying corrective actions will have little to no effect on outcomes. Thus, pay-withholding schemes can have a negative net effect by diverting resources from QI projects that truly improve patient outcomes.
Withholding payment in this case generates approximately $160 in direct savings to the payor (assuming Medicare payments for one 99233 and two 99232 subsequent care visits), yet the operational costs are not negligible and must be factored into the equation. The payor needs to first determine who is truly at fault: the hospitalist or the orthopedic surgeon. Answering that question requires the payor to review the co-management agreement, perhaps aided by an attorney. That’s a costly endeavor.
For the sake of argument, let’s assume in this case the hospitalist is at fault. The next step is determining if the hospitalist who failed to prescribe prophylaxis prior to the PE is the same hospitalist caring for the patient after the PE. It is inappropriate to withhold payment to hospitalist A if hospitalist B made the error. Again, significant manpower will be required to determine fault, as this is not information one finds on a UB-04 claim form submitted to Medicare.
Further eroding the $160 savings is the cost of determining whether a contraindication exists: Bleeding ulcer? Subdural hematoma? Heparinoid allergy? Let us not forget the additional costs in copying, shipping, warehousing, and eventual shredding of the records. One can readily see that the operational costs can quickly negate the $160 anticipated savings. In fact, it’s likely a negative return on investment.
Clearly, withholding payment in this scenario is an ineffective mechanism for improving quality or decreasing cost. I am not generally a proponent of rewarding failure, and perhaps as we usher in a new era of healthcare reform, the system will be redesigned in such a way that better aligns quality and cost-control measures. However, under the current system, payment denial as outlined above likely does more harm than good.
CON
Withhold payment when medical errors are easily identifiable
When I first learned of the proposal to withhold Medicare payment for hospital-acquired conditions (HACs), I had mixed emotions. On the one hand, I firmly believe that physicians should be accountable for their work; on the other hand, this policy seems to conflict sharply with the “no blame” mantra that has been prevalent in patient safety for more than a decade.2 More recently, though, many have argued for balancing the pursuit of system fixes for quality and patient-safety issues with the development of a culture of accountability.3
In theory, the HACs should meet the following criteria: They should be high-cost conditions, high-volume conditions, or both; they should be identifiable through ICD-9-CM coding as complicating conditions (CCs) or major complicating conditions (MCCs) that result in a higher-paying MS-DRG; and they should be reasonably preventable through the application of evidence-based guidelines. Some HACs are jaw-dropping lapses in care (e.g., leaving foreign bodies in during surgery). Other HACs seem to me to be much less preventable, especially fall injuries and catheter-associated urinary tract infections (UTIs). Several experts have written eloquently regarding the limitations of these new measures, particularly emphasizing the potential for increased administrative burden on hospitals and the potential for unintended consequences.4,5
However, in the case described above involving a hospitalist, I have no reservations in limiting payment to the provider. To me, failing to prescribe VTE prophylaxis in an elderly, immobilized, post-op hip replacement patient with a CHF exacerbation is the hospitalist’s equivalent to a surgeon leaving behind a sponge in an appendectomy. It also meets the elements outlined in the HAC withholding program:
- It is high-cost. The 2007 MS-DRG payment for elective hip arthroplasty was $9,863, but adding an MCC increased that cost by one-third.6
- It is readily identifiable, though one concern might be that hospitals would perform unnecessary pre-operative testing to identify asymptomatic DVT, incurring increased testing and treatment costs and increasing the incidence of bleeding complications.
- It is very preventable. Without thromboprophylaxis, 40% to 60% of hip arthroplasty patients will develop an asymptomatic DVT, and 1 in 300 will die from a PE. However, such fatal events are exceedingly rare with appropriate prevention.7
Ultimately, I think a policy of nonpayment for this case keeps with the culture of accountability we need to foster in healthcare. The financial implications of nonpayment will drive hospital innovation and force the hospital to police provider behavior in more effective ways. This is likely to be a painful process, similar to the tribulations experienced with implementing pay-for-performance programs. The Centers for Medicare and Medicaid Services (CMS) needs to be flexible in adding—and removing—new HACs based on good evidence.
Regardless, the goal of achieving a safer, more effective healthcare system remains.
References
- Leape LL. Error in medicine. JAMA. 1994;272(23):1851-1857.
- Institute of Medicine. To Err Is Human: Building a Safer Healthcare System. Washington, D.C.: National Academies Press; 2000.
- Wachter RM, Pronovost PJ. Balancing “no blame” with accountability in patient safety. N Engl J Med. 2009;361:1401-1406.
- Saint S, Meddings JA, Calfee D, Kowalski CP, Krein SL. Catheter-associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877-884.
- Inouye SK, Brown CJ, Tinetti ME. Medicare nonpayment, hospital falls, and unintended consequences. N Engl J Med. 2009;360(23):2390-2393.
- Wachter RM, Foster NE, Dudley RA. Medicare’s decision to withhold payment for hospital errors: the devil is in the det. Jt Comm J Qual Patient Saf. 2008;34(2):116-123.
- Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):381S-453S.
The opinions expressed herein are those of the authors and do not represent those of SHM or The Hospitalist.
Texas-Sized Tort Reform
Advocates have written open letters to politicians describing it as “the least-expensive and best-known way to lower healthcare costs.” Detractors have blogged that it has saved almost no money and instead “gutted patient rights.” Among the recent templates for whether and how to proceed on the contentious issue of tort reform, Texas has become a prime example of either the wisdom or the folly of capping medical liability payouts, depending on your vantage point.
Tort reform is backed by most doctors and the insurance industry but opposed by lawyers and consumer advocates. The Congressional Budget Office has documented increases in both medical liability premiums and average malpractice claim payments that have significantly outpaced inflation. Congress itself has largely punted on the issue, however, leaving most of the wrangling over specifics to individual states.
Which brings us to Texas. In reaction to the perception that unsustainable medical liability costs were driving away doctors and driving up healthcare costs, state voters in 2003 approved Proposition 12. Among its provisions, the state constitutional amendment capped noneconomic medical liability payouts at $250,000 in nearly all cases.
Much of the ensuing debate over whether Texas did the right thing has focused on cost: For example, will the reduction in malpractice claims translate into significant savings within the healthcare system? Is tort reform relevant in recouping the perceived waste from “defensive medicine,” in which physicians are presumed to order unnecessary tests and procedures out of fear of lawsuits?
More centrally, however, the question boils down to this: Does tort reform improve the ability of doctors to do their jobs, and the opportunity for patients to benefit from that care? So far, statistics, reports, and anecdotal information suggest that Texas has achieved the first goal but not necessarily the latter, highlighting the extreme difficulty in striking the right balance.
Mission: Predictable
Kirk A. Calhoun, MD, who became president of the University of Texas Health Science Center at Tyler in 2002, points to two principal benefits of the state’s tort reform. First, the package of reforms led to a significant number of physicians migrating to Texas and helping to deal with the state’s chronic doctor shortage. For doctors, part of that attraction was the second big benefit, a significant decrease in liability insurance premiums.
“It has resulted in making Texas a more attractive state in which to practice medicine,” Dr. Calhoun says. “As a result of those expenses going down, we are able to better invest in our primary mission, and on patient care.”
Kenneth McDaniel, a program specialist in professional liability in the Texas Department of Insurance, says the dearth of affordable or available malpractice insurance in the state had spiraled into a crisis. “In Texas, we were staring at the brink of a chasm so deep that we virtually had to do something,” he says. “We were within probably some months or a year of having almost no malpractice insurance industry at all. It had become very dire.”
McDaniel stresses that the new liability cap is only for intangibles or pain and suffering, and it leaves intact the potential for higher economic damages. “But those can be predicted,” he says. “As soon as claims became more predictable, insurers started coming back into the field.” A summary of 17 companies’ rates supplied by McDaniel includes four new arrivals to the medical liability market and the return of a fifth.
At the very least, tort reform appears to have dramatically curbed the number and cost of claims in Texas. From 2003 to 2007, malpractice payments to patients dropped by two-thirds. Liability premiums paid by doctors also have fallen, by an average of 27.5%, and more insurers have rejoined the market. “We are now back to, I would say, a pretty healthy environment,” McDaniel says.
Physician-Friendly Environs
Hospitalist Gregory Johnson, MD, chair of the Texas Medical Association’s Young Physicians Section, moved to Texas in 2002, just before the reforms were approved. “The best part about Prop 12 passing is the fact that Texas is now seen as a very physician-friendly environment,” says Johnson, who now serves as a Houston-based regional chief medical officer for Tacoma, Wash.-based Sound Physicians. The significant drops in malpractice insurance rates and lawsuits have made it far easier for him to recruit out-of-state doctors. “That basically comes off any physician’s radar as a particular concern.”
Most Texas hospitals and healthcare systems do not employ physicians directly. Instead, they contract or affiliate with private or nonprofit physicians groups. Due to that arrangement, Dr. Johnson explains, the cost of insurance premiums “becomes a much more individually based and personal issue because it’s coming out of an individual’s pocket, or a group’s pocket.”
From his own experience, Dr. Johnson says, he believes hospitalists are more willing to go to underserved parts of the state because of tort reform. Three years ago, he helped start Amarillo Hospitalist Services, a program that began with three doctors and has since grown to eight, all affiliated with Northwest Texas Hospital.
Of course, hospitalists appear to be thriving in major metropolitan areas, too. Dr. Johnson’s new employer, Sound Physicians, now operates three HM programs within Houston’s Memorial Hermann Healthcare System and employs about two-dozen physicians in all. More are on the way. “We’re actively hiring,” he says.
Mixed Outcomes
Statistics from the Texas Medical Board and Department of State Health Services confirm the anecdotal evidence that a more doctor-friendly Texas is paying dividends. Even so, they paint a somewhat more complicated picture than some commentators have portrayed in recent editorials. Doctors have indeed flocked to the state—some 11,000 since 2002 alone, an increase of 31%. That rate has far outpaced the state’s overall population growth of 14.2%.
But not all areas of the state have benefited equally from the influx.
Starr County, the third-poorest county in the U.S. based on per capita income, is among those that have fared well since 2002. Overall, its number of doctors increased from 14 to 24, a net increase of 71%, as its population rose by a projected 17%. But the next five poorest counties in Texas, accounting for nearly 86,000 residents in 2002, lost six doctors during the same time period—a 12.5% decline, even as their collective population rose by a projected 10.2%. Contrary to some public pronouncements, tort reform alone has not solved the chronic shortage of doctors in poor rural areas.
A withering report released in December by Washington, D.C.-based Public Citizen, a nonprofit consumer advocacy group, offers a harsher assessment, concluding that Texas’ “experiment with medical liability caps has failed” (www.citizen.org/publications/release.cfm?ID=7721). The report suggests that Texas’ dead-last ranking in percentage of uninsured residents (25%) and the doctor shortage in rural areas have actually grown worse since tort reform. Meanwhile, the cost of health insurance has more than doubled, while the cost of healthcare also has increased at nearly double the national average, other metrics that led to the organization’s vote of no confidence.
The impact on quality of care has been harder to assess. But Dr. Johnson and other observers say they haven’t seen any dip within hospitals. “I think that we as physicians and we as hospitalists really want to focus on our patients, and we can help to drive down those costs if we’re given the freedom to do our job,” he says. Tort reform, he adds, has helped doctors do precisely that.
Dr. Calhoun agrees. “No one wants to be sued. Everyone wants to do a good job,” he says. The threat of a lawsuit alone is only one of many factors influencing quality, he adds. But creating a more inviting environment for doctors can make a big difference by encouraging the increased use of hospitalists. “Having a hospitalist in the hospital all the time,” he says, “is an obvious quality improvement.” TH
Bryn Nelson is a freelance medical writer based in Seattle.
IMAGE SOURCE: DRASCHWARTZ/ISTOCK.COM
Advocates have written open letters to politicians describing it as “the least-expensive and best-known way to lower healthcare costs.” Detractors have blogged that it has saved almost no money and instead “gutted patient rights.” Among the recent templates for whether and how to proceed on the contentious issue of tort reform, Texas has become a prime example of either the wisdom or the folly of capping medical liability payouts, depending on your vantage point.
Tort reform is backed by most doctors and the insurance industry but opposed by lawyers and consumer advocates. The Congressional Budget Office has documented increases in both medical liability premiums and average malpractice claim payments that have significantly outpaced inflation. Congress itself has largely punted on the issue, however, leaving most of the wrangling over specifics to individual states.
Which brings us to Texas. In reaction to the perception that unsustainable medical liability costs were driving away doctors and driving up healthcare costs, state voters in 2003 approved Proposition 12. Among its provisions, the state constitutional amendment capped noneconomic medical liability payouts at $250,000 in nearly all cases.
Much of the ensuing debate over whether Texas did the right thing has focused on cost: For example, will the reduction in malpractice claims translate into significant savings within the healthcare system? Is tort reform relevant in recouping the perceived waste from “defensive medicine,” in which physicians are presumed to order unnecessary tests and procedures out of fear of lawsuits?
More centrally, however, the question boils down to this: Does tort reform improve the ability of doctors to do their jobs, and the opportunity for patients to benefit from that care? So far, statistics, reports, and anecdotal information suggest that Texas has achieved the first goal but not necessarily the latter, highlighting the extreme difficulty in striking the right balance.
Mission: Predictable
Kirk A. Calhoun, MD, who became president of the University of Texas Health Science Center at Tyler in 2002, points to two principal benefits of the state’s tort reform. First, the package of reforms led to a significant number of physicians migrating to Texas and helping to deal with the state’s chronic doctor shortage. For doctors, part of that attraction was the second big benefit, a significant decrease in liability insurance premiums.
“It has resulted in making Texas a more attractive state in which to practice medicine,” Dr. Calhoun says. “As a result of those expenses going down, we are able to better invest in our primary mission, and on patient care.”
Kenneth McDaniel, a program specialist in professional liability in the Texas Department of Insurance, says the dearth of affordable or available malpractice insurance in the state had spiraled into a crisis. “In Texas, we were staring at the brink of a chasm so deep that we virtually had to do something,” he says. “We were within probably some months or a year of having almost no malpractice insurance industry at all. It had become very dire.”
McDaniel stresses that the new liability cap is only for intangibles or pain and suffering, and it leaves intact the potential for higher economic damages. “But those can be predicted,” he says. “As soon as claims became more predictable, insurers started coming back into the field.” A summary of 17 companies’ rates supplied by McDaniel includes four new arrivals to the medical liability market and the return of a fifth.
At the very least, tort reform appears to have dramatically curbed the number and cost of claims in Texas. From 2003 to 2007, malpractice payments to patients dropped by two-thirds. Liability premiums paid by doctors also have fallen, by an average of 27.5%, and more insurers have rejoined the market. “We are now back to, I would say, a pretty healthy environment,” McDaniel says.
Physician-Friendly Environs
Hospitalist Gregory Johnson, MD, chair of the Texas Medical Association’s Young Physicians Section, moved to Texas in 2002, just before the reforms were approved. “The best part about Prop 12 passing is the fact that Texas is now seen as a very physician-friendly environment,” says Johnson, who now serves as a Houston-based regional chief medical officer for Tacoma, Wash.-based Sound Physicians. The significant drops in malpractice insurance rates and lawsuits have made it far easier for him to recruit out-of-state doctors. “That basically comes off any physician’s radar as a particular concern.”
Most Texas hospitals and healthcare systems do not employ physicians directly. Instead, they contract or affiliate with private or nonprofit physicians groups. Due to that arrangement, Dr. Johnson explains, the cost of insurance premiums “becomes a much more individually based and personal issue because it’s coming out of an individual’s pocket, or a group’s pocket.”
From his own experience, Dr. Johnson says, he believes hospitalists are more willing to go to underserved parts of the state because of tort reform. Three years ago, he helped start Amarillo Hospitalist Services, a program that began with three doctors and has since grown to eight, all affiliated with Northwest Texas Hospital.
Of course, hospitalists appear to be thriving in major metropolitan areas, too. Dr. Johnson’s new employer, Sound Physicians, now operates three HM programs within Houston’s Memorial Hermann Healthcare System and employs about two-dozen physicians in all. More are on the way. “We’re actively hiring,” he says.
Mixed Outcomes
Statistics from the Texas Medical Board and Department of State Health Services confirm the anecdotal evidence that a more doctor-friendly Texas is paying dividends. Even so, they paint a somewhat more complicated picture than some commentators have portrayed in recent editorials. Doctors have indeed flocked to the state—some 11,000 since 2002 alone, an increase of 31%. That rate has far outpaced the state’s overall population growth of 14.2%.
But not all areas of the state have benefited equally from the influx.
Starr County, the third-poorest county in the U.S. based on per capita income, is among those that have fared well since 2002. Overall, its number of doctors increased from 14 to 24, a net increase of 71%, as its population rose by a projected 17%. But the next five poorest counties in Texas, accounting for nearly 86,000 residents in 2002, lost six doctors during the same time period—a 12.5% decline, even as their collective population rose by a projected 10.2%. Contrary to some public pronouncements, tort reform alone has not solved the chronic shortage of doctors in poor rural areas.
A withering report released in December by Washington, D.C.-based Public Citizen, a nonprofit consumer advocacy group, offers a harsher assessment, concluding that Texas’ “experiment with medical liability caps has failed” (www.citizen.org/publications/release.cfm?ID=7721). The report suggests that Texas’ dead-last ranking in percentage of uninsured residents (25%) and the doctor shortage in rural areas have actually grown worse since tort reform. Meanwhile, the cost of health insurance has more than doubled, while the cost of healthcare also has increased at nearly double the national average, other metrics that led to the organization’s vote of no confidence.
The impact on quality of care has been harder to assess. But Dr. Johnson and other observers say they haven’t seen any dip within hospitals. “I think that we as physicians and we as hospitalists really want to focus on our patients, and we can help to drive down those costs if we’re given the freedom to do our job,” he says. Tort reform, he adds, has helped doctors do precisely that.
Dr. Calhoun agrees. “No one wants to be sued. Everyone wants to do a good job,” he says. The threat of a lawsuit alone is only one of many factors influencing quality, he adds. But creating a more inviting environment for doctors can make a big difference by encouraging the increased use of hospitalists. “Having a hospitalist in the hospital all the time,” he says, “is an obvious quality improvement.” TH
Bryn Nelson is a freelance medical writer based in Seattle.
IMAGE SOURCE: DRASCHWARTZ/ISTOCK.COM
Advocates have written open letters to politicians describing it as “the least-expensive and best-known way to lower healthcare costs.” Detractors have blogged that it has saved almost no money and instead “gutted patient rights.” Among the recent templates for whether and how to proceed on the contentious issue of tort reform, Texas has become a prime example of either the wisdom or the folly of capping medical liability payouts, depending on your vantage point.
Tort reform is backed by most doctors and the insurance industry but opposed by lawyers and consumer advocates. The Congressional Budget Office has documented increases in both medical liability premiums and average malpractice claim payments that have significantly outpaced inflation. Congress itself has largely punted on the issue, however, leaving most of the wrangling over specifics to individual states.
Which brings us to Texas. In reaction to the perception that unsustainable medical liability costs were driving away doctors and driving up healthcare costs, state voters in 2003 approved Proposition 12. Among its provisions, the state constitutional amendment capped noneconomic medical liability payouts at $250,000 in nearly all cases.
Much of the ensuing debate over whether Texas did the right thing has focused on cost: For example, will the reduction in malpractice claims translate into significant savings within the healthcare system? Is tort reform relevant in recouping the perceived waste from “defensive medicine,” in which physicians are presumed to order unnecessary tests and procedures out of fear of lawsuits?
More centrally, however, the question boils down to this: Does tort reform improve the ability of doctors to do their jobs, and the opportunity for patients to benefit from that care? So far, statistics, reports, and anecdotal information suggest that Texas has achieved the first goal but not necessarily the latter, highlighting the extreme difficulty in striking the right balance.
Mission: Predictable
Kirk A. Calhoun, MD, who became president of the University of Texas Health Science Center at Tyler in 2002, points to two principal benefits of the state’s tort reform. First, the package of reforms led to a significant number of physicians migrating to Texas and helping to deal with the state’s chronic doctor shortage. For doctors, part of that attraction was the second big benefit, a significant decrease in liability insurance premiums.
“It has resulted in making Texas a more attractive state in which to practice medicine,” Dr. Calhoun says. “As a result of those expenses going down, we are able to better invest in our primary mission, and on patient care.”
Kenneth McDaniel, a program specialist in professional liability in the Texas Department of Insurance, says the dearth of affordable or available malpractice insurance in the state had spiraled into a crisis. “In Texas, we were staring at the brink of a chasm so deep that we virtually had to do something,” he says. “We were within probably some months or a year of having almost no malpractice insurance industry at all. It had become very dire.”
McDaniel stresses that the new liability cap is only for intangibles or pain and suffering, and it leaves intact the potential for higher economic damages. “But those can be predicted,” he says. “As soon as claims became more predictable, insurers started coming back into the field.” A summary of 17 companies’ rates supplied by McDaniel includes four new arrivals to the medical liability market and the return of a fifth.
At the very least, tort reform appears to have dramatically curbed the number and cost of claims in Texas. From 2003 to 2007, malpractice payments to patients dropped by two-thirds. Liability premiums paid by doctors also have fallen, by an average of 27.5%, and more insurers have rejoined the market. “We are now back to, I would say, a pretty healthy environment,” McDaniel says.
Physician-Friendly Environs
Hospitalist Gregory Johnson, MD, chair of the Texas Medical Association’s Young Physicians Section, moved to Texas in 2002, just before the reforms were approved. “The best part about Prop 12 passing is the fact that Texas is now seen as a very physician-friendly environment,” says Johnson, who now serves as a Houston-based regional chief medical officer for Tacoma, Wash.-based Sound Physicians. The significant drops in malpractice insurance rates and lawsuits have made it far easier for him to recruit out-of-state doctors. “That basically comes off any physician’s radar as a particular concern.”
Most Texas hospitals and healthcare systems do not employ physicians directly. Instead, they contract or affiliate with private or nonprofit physicians groups. Due to that arrangement, Dr. Johnson explains, the cost of insurance premiums “becomes a much more individually based and personal issue because it’s coming out of an individual’s pocket, or a group’s pocket.”
From his own experience, Dr. Johnson says, he believes hospitalists are more willing to go to underserved parts of the state because of tort reform. Three years ago, he helped start Amarillo Hospitalist Services, a program that began with three doctors and has since grown to eight, all affiliated with Northwest Texas Hospital.
Of course, hospitalists appear to be thriving in major metropolitan areas, too. Dr. Johnson’s new employer, Sound Physicians, now operates three HM programs within Houston’s Memorial Hermann Healthcare System and employs about two-dozen physicians in all. More are on the way. “We’re actively hiring,” he says.
Mixed Outcomes
Statistics from the Texas Medical Board and Department of State Health Services confirm the anecdotal evidence that a more doctor-friendly Texas is paying dividends. Even so, they paint a somewhat more complicated picture than some commentators have portrayed in recent editorials. Doctors have indeed flocked to the state—some 11,000 since 2002 alone, an increase of 31%. That rate has far outpaced the state’s overall population growth of 14.2%.
But not all areas of the state have benefited equally from the influx.
Starr County, the third-poorest county in the U.S. based on per capita income, is among those that have fared well since 2002. Overall, its number of doctors increased from 14 to 24, a net increase of 71%, as its population rose by a projected 17%. But the next five poorest counties in Texas, accounting for nearly 86,000 residents in 2002, lost six doctors during the same time period—a 12.5% decline, even as their collective population rose by a projected 10.2%. Contrary to some public pronouncements, tort reform alone has not solved the chronic shortage of doctors in poor rural areas.
A withering report released in December by Washington, D.C.-based Public Citizen, a nonprofit consumer advocacy group, offers a harsher assessment, concluding that Texas’ “experiment with medical liability caps has failed” (www.citizen.org/publications/release.cfm?ID=7721). The report suggests that Texas’ dead-last ranking in percentage of uninsured residents (25%) and the doctor shortage in rural areas have actually grown worse since tort reform. Meanwhile, the cost of health insurance has more than doubled, while the cost of healthcare also has increased at nearly double the national average, other metrics that led to the organization’s vote of no confidence.
The impact on quality of care has been harder to assess. But Dr. Johnson and other observers say they haven’t seen any dip within hospitals. “I think that we as physicians and we as hospitalists really want to focus on our patients, and we can help to drive down those costs if we’re given the freedom to do our job,” he says. Tort reform, he adds, has helped doctors do precisely that.
Dr. Calhoun agrees. “No one wants to be sued. Everyone wants to do a good job,” he says. The threat of a lawsuit alone is only one of many factors influencing quality, he adds. But creating a more inviting environment for doctors can make a big difference by encouraging the increased use of hospitalists. “Having a hospitalist in the hospital all the time,” he says, “is an obvious quality improvement.” TH
Bryn Nelson is a freelance medical writer based in Seattle.
IMAGE SOURCE: DRASCHWARTZ/ISTOCK.COM
Admit Documentation
In light of the recent elimination of consultation codes from the Medicare Physician Fee Schedule, physicians of all specialties are being asked to report initial hospital care services (99221-99223) for their first encounter with a patient.1 This leaves hospitalists with questions about the billing and financial implications of reporting admissions services.
Here’s a typical scenario: Dr. A admits a Medicare patient to the hospital from the ED for hyperglycemia and dehydration in the setting of uncontrolled diabetes. He performs and documents an initial hospital-care service on day one of the admission. On day two, another hospitalist, Dr. B, who works in the same HM group, sees the patient for the first time. What should each of the physicians report for their first encounter with the patient?
Each hospitalist should select the CPT code that best fits the service and their role in the case. Remember, only one physician is named “attending of record” or “admitting physician.”
When billing during the course of the hospitalization, consider all physicians of the same specialty in the same provider group as the “admitting physician/group.”
Admissions Service
On day one, Dr. A admits the patient. He performs and documents a comprehensive history, a comprehensive exam, and medical decision-making of high complexity. The documentation corresponds to the highest initial admission service, 99223. Given the recent Medicare billing changes, the attending of record is required to append modifier “AI” (principal physician of record) to the admission service (e.g., 99223-AI).
The purpose of this modifier is “to identify the physician who oversees the patient’s care from all other physicians who may be furnishing specialty care.”2 This modifier has no financial implications. It does not increase or decrease the payment associated with the reported visit level (i.e., 99223 is reimbursed at a national rate of approximately $190, with or without modifier AI).
Initial Encounter by Team Members
As previously stated, the elimination of consultation services requires physicians to report their initial hospital encounter with an initial hospital-care code (i.e., 99221-99223). However, Medicare states that “physicians in the same group practice who are in the same specialty must bill and be paid as though they were a single physician.”3 This means followup services performed on days subsequent to a group member’s initial admission service must be reported with subsequent hospital-care codes (99231-99233). Therefore, in the scenario above, Dr. B is obligated to report the appropriate subsequent hospital-care code for his patient encounter on day two.
Incomplete Documentation
Initial hospital-care services (99221-99223) require the physician to obtain, perform, and document the necessary elements of history, physical exam, and medical decision-making in support of the code reported on the claim. There are occasions when the physician’s documentation does not support the lowest code (i.e., 99221). A reasonable approach is to report the service with an unlisted E&M code (99499). “Unlisted” codes do not have a payor-recognized code description or fee. When reporting an unlisted code, the biller must manually enter a charge description (e.g., expanded problem-focused admissions service) and a fee. A payor-prompted request for documentation is likely before payment is made.
Some payors have more specific references to the situation and allow for options. Two options exist for coding services that do not meet the work and/or medical necessity requirements of 99221-99223: report an unlisted E&M service (99499); or report a subsequent hospital care code (99231-99233) that appropriately reflects physician work and medical necessity for the service, and avoids mandatory medical record submission and manual medical review.4
In fact, Medicare Administrator Contractor TrailBlazer Health’s Web site (www.trailblazerhealth.com) offers guidance to physicians who are unsure if subsequent hospital care is an appropriate choice for this dilemma: “TrailBlazer recognizes provider reluctance to miscode initial hospital care as subsequent hospital care. However, doing so is preferable in that it allows Medicare to process and pay the claims much more efficiently. For those concerned about miscoding these services, please understand that TrailBlazer will not find fault with providers who choose this option when records appropriately demonstrate the work and medical necessity of the subsequent code chosen.”4 TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She also is faculty for SHM’s inpatient coding course.
References
- CMS announces payment, policy changes for physicians services to Medicare beneficiaries in 2010. Centers for Medicare and Medicaid Services Web site. Available at: www.cms.hhs.gov/apps/media/ press/release.asp?Counter=3539&intNumPerPage=10&checkDate=&checkKey=&srchType=1&numDays=3500&srchOpt=0&srchData=&keywordType=All&chkNewsType=1%2C+2%2C+3%2C+4%2C+5&intPage=&showAll=&pYear=&year=&desc=&cboOrder=date. Accessed Nov. 12, 2009.
- Revisions to Consultation Services Payment Policy. Medicare Learning Network Web site. Available at: www.cms.hhs.gov/MLNMattersArticles/downloads/ MM6740.pdf. Accessed Jan. 16, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.5. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 16, 2010.
- Update-evaluation and management services formerly coded as consultations. Trailblazer Health Enterprises Web site. Available at: www.trailblazerhealth.com/Tools/Notices.aspx?DomainID=1. Accessed Jan. 17, 2010.
- Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2009;14-15.
In light of the recent elimination of consultation codes from the Medicare Physician Fee Schedule, physicians of all specialties are being asked to report initial hospital care services (99221-99223) for their first encounter with a patient.1 This leaves hospitalists with questions about the billing and financial implications of reporting admissions services.
Here’s a typical scenario: Dr. A admits a Medicare patient to the hospital from the ED for hyperglycemia and dehydration in the setting of uncontrolled diabetes. He performs and documents an initial hospital-care service on day one of the admission. On day two, another hospitalist, Dr. B, who works in the same HM group, sees the patient for the first time. What should each of the physicians report for their first encounter with the patient?
Each hospitalist should select the CPT code that best fits the service and their role in the case. Remember, only one physician is named “attending of record” or “admitting physician.”
When billing during the course of the hospitalization, consider all physicians of the same specialty in the same provider group as the “admitting physician/group.”
Admissions Service
On day one, Dr. A admits the patient. He performs and documents a comprehensive history, a comprehensive exam, and medical decision-making of high complexity. The documentation corresponds to the highest initial admission service, 99223. Given the recent Medicare billing changes, the attending of record is required to append modifier “AI” (principal physician of record) to the admission service (e.g., 99223-AI).
The purpose of this modifier is “to identify the physician who oversees the patient’s care from all other physicians who may be furnishing specialty care.”2 This modifier has no financial implications. It does not increase or decrease the payment associated with the reported visit level (i.e., 99223 is reimbursed at a national rate of approximately $190, with or without modifier AI).
Initial Encounter by Team Members
As previously stated, the elimination of consultation services requires physicians to report their initial hospital encounter with an initial hospital-care code (i.e., 99221-99223). However, Medicare states that “physicians in the same group practice who are in the same specialty must bill and be paid as though they were a single physician.”3 This means followup services performed on days subsequent to a group member’s initial admission service must be reported with subsequent hospital-care codes (99231-99233). Therefore, in the scenario above, Dr. B is obligated to report the appropriate subsequent hospital-care code for his patient encounter on day two.
Incomplete Documentation
Initial hospital-care services (99221-99223) require the physician to obtain, perform, and document the necessary elements of history, physical exam, and medical decision-making in support of the code reported on the claim. There are occasions when the physician’s documentation does not support the lowest code (i.e., 99221). A reasonable approach is to report the service with an unlisted E&M code (99499). “Unlisted” codes do not have a payor-recognized code description or fee. When reporting an unlisted code, the biller must manually enter a charge description (e.g., expanded problem-focused admissions service) and a fee. A payor-prompted request for documentation is likely before payment is made.
Some payors have more specific references to the situation and allow for options. Two options exist for coding services that do not meet the work and/or medical necessity requirements of 99221-99223: report an unlisted E&M service (99499); or report a subsequent hospital care code (99231-99233) that appropriately reflects physician work and medical necessity for the service, and avoids mandatory medical record submission and manual medical review.4
In fact, Medicare Administrator Contractor TrailBlazer Health’s Web site (www.trailblazerhealth.com) offers guidance to physicians who are unsure if subsequent hospital care is an appropriate choice for this dilemma: “TrailBlazer recognizes provider reluctance to miscode initial hospital care as subsequent hospital care. However, doing so is preferable in that it allows Medicare to process and pay the claims much more efficiently. For those concerned about miscoding these services, please understand that TrailBlazer will not find fault with providers who choose this option when records appropriately demonstrate the work and medical necessity of the subsequent code chosen.”4 TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She also is faculty for SHM’s inpatient coding course.
References
- CMS announces payment, policy changes for physicians services to Medicare beneficiaries in 2010. Centers for Medicare and Medicaid Services Web site. Available at: www.cms.hhs.gov/apps/media/ press/release.asp?Counter=3539&intNumPerPage=10&checkDate=&checkKey=&srchType=1&numDays=3500&srchOpt=0&srchData=&keywordType=All&chkNewsType=1%2C+2%2C+3%2C+4%2C+5&intPage=&showAll=&pYear=&year=&desc=&cboOrder=date. Accessed Nov. 12, 2009.
- Revisions to Consultation Services Payment Policy. Medicare Learning Network Web site. Available at: www.cms.hhs.gov/MLNMattersArticles/downloads/ MM6740.pdf. Accessed Jan. 16, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.5. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 16, 2010.
- Update-evaluation and management services formerly coded as consultations. Trailblazer Health Enterprises Web site. Available at: www.trailblazerhealth.com/Tools/Notices.aspx?DomainID=1. Accessed Jan. 17, 2010.
- Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2009;14-15.
In light of the recent elimination of consultation codes from the Medicare Physician Fee Schedule, physicians of all specialties are being asked to report initial hospital care services (99221-99223) for their first encounter with a patient.1 This leaves hospitalists with questions about the billing and financial implications of reporting admissions services.
Here’s a typical scenario: Dr. A admits a Medicare patient to the hospital from the ED for hyperglycemia and dehydration in the setting of uncontrolled diabetes. He performs and documents an initial hospital-care service on day one of the admission. On day two, another hospitalist, Dr. B, who works in the same HM group, sees the patient for the first time. What should each of the physicians report for their first encounter with the patient?
Each hospitalist should select the CPT code that best fits the service and their role in the case. Remember, only one physician is named “attending of record” or “admitting physician.”
When billing during the course of the hospitalization, consider all physicians of the same specialty in the same provider group as the “admitting physician/group.”
Admissions Service
On day one, Dr. A admits the patient. He performs and documents a comprehensive history, a comprehensive exam, and medical decision-making of high complexity. The documentation corresponds to the highest initial admission service, 99223. Given the recent Medicare billing changes, the attending of record is required to append modifier “AI” (principal physician of record) to the admission service (e.g., 99223-AI).
The purpose of this modifier is “to identify the physician who oversees the patient’s care from all other physicians who may be furnishing specialty care.”2 This modifier has no financial implications. It does not increase or decrease the payment associated with the reported visit level (i.e., 99223 is reimbursed at a national rate of approximately $190, with or without modifier AI).
Initial Encounter by Team Members
As previously stated, the elimination of consultation services requires physicians to report their initial hospital encounter with an initial hospital-care code (i.e., 99221-99223). However, Medicare states that “physicians in the same group practice who are in the same specialty must bill and be paid as though they were a single physician.”3 This means followup services performed on days subsequent to a group member’s initial admission service must be reported with subsequent hospital-care codes (99231-99233). Therefore, in the scenario above, Dr. B is obligated to report the appropriate subsequent hospital-care code for his patient encounter on day two.
Incomplete Documentation
Initial hospital-care services (99221-99223) require the physician to obtain, perform, and document the necessary elements of history, physical exam, and medical decision-making in support of the code reported on the claim. There are occasions when the physician’s documentation does not support the lowest code (i.e., 99221). A reasonable approach is to report the service with an unlisted E&M code (99499). “Unlisted” codes do not have a payor-recognized code description or fee. When reporting an unlisted code, the biller must manually enter a charge description (e.g., expanded problem-focused admissions service) and a fee. A payor-prompted request for documentation is likely before payment is made.
Some payors have more specific references to the situation and allow for options. Two options exist for coding services that do not meet the work and/or medical necessity requirements of 99221-99223: report an unlisted E&M service (99499); or report a subsequent hospital care code (99231-99233) that appropriately reflects physician work and medical necessity for the service, and avoids mandatory medical record submission and manual medical review.4
In fact, Medicare Administrator Contractor TrailBlazer Health’s Web site (www.trailblazerhealth.com) offers guidance to physicians who are unsure if subsequent hospital care is an appropriate choice for this dilemma: “TrailBlazer recognizes provider reluctance to miscode initial hospital care as subsequent hospital care. However, doing so is preferable in that it allows Medicare to process and pay the claims much more efficiently. For those concerned about miscoding these services, please understand that TrailBlazer will not find fault with providers who choose this option when records appropriately demonstrate the work and medical necessity of the subsequent code chosen.”4 TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She also is faculty for SHM’s inpatient coding course.
References
- CMS announces payment, policy changes for physicians services to Medicare beneficiaries in 2010. Centers for Medicare and Medicaid Services Web site. Available at: www.cms.hhs.gov/apps/media/ press/release.asp?Counter=3539&intNumPerPage=10&checkDate=&checkKey=&srchType=1&numDays=3500&srchOpt=0&srchData=&keywordType=All&chkNewsType=1%2C+2%2C+3%2C+4%2C+5&intPage=&showAll=&pYear=&year=&desc=&cboOrder=date. Accessed Nov. 12, 2009.
- Revisions to Consultation Services Payment Policy. Medicare Learning Network Web site. Available at: www.cms.hhs.gov/MLNMattersArticles/downloads/ MM6740.pdf. Accessed Jan. 16, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.5. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 16, 2010.
- Update-evaluation and management services formerly coded as consultations. Trailblazer Health Enterprises Web site. Available at: www.trailblazerhealth.com/Tools/Notices.aspx?DomainID=1. Accessed Jan. 17, 2010.
- Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2009;14-15.
When Should an IVC Filter Be Used to Treat a DVT?
Case
A 67-year-old man with a history of hypertension presents with a swollen right lower extremity. An ultrasound reveals a DVT, and he is commenced on low-molecular-weight heparin and warfarin. Two days later, he develops slurred speech and right-sided weakness. A head CT reveals an intracranial hemorrhage. When should an inferior vena cava (IVC) filter be utilized for treatment of DVT?
Overview
It is estimated that 350,000 to 600,000 Americans develop a VTE each year.1 Patients with a DVT are at high risk of developing a pulmonary embolism (PE). In a multicenter study, nearly 40% of patients admitted with a DVT had evidence of a PE on ventilation perfusion scan.2 Treatment of a DVT is aimed at preventing the extension of the DVT and embolization.3 The American College of Chest Physicians (ACCP) recommends anticoagulation as the primary DVT treatment (Grade 1A).4 However, IVC filters might be considered when anticoagulation is contraindicated.
In 1868, Trousseau created the conceptual model of surgical interruption of the IVC to prevent PE. However, it wasn’t until 1959 by Bottini that the surgical interruption was successfully performed.5 The Mobin-Uddin filter was introduced in 1967 as the first mechanical IVC filter.6 IVC filters mechanically trap the DVT, preventing emboli from traveling into the pulmonary vasculature.7
There are two classes of IVC filters: permanent filters and removable filters. Removable filters include both temporary filters and retrievable filters. Temporary filters are attached to a catheter that exits the skin and therefore must be removed due to the risk of infection and embolization.7 Retrievable filters are similar in design to permanent filters but are designed to be removed. However, this must be done with caution, as neointimal hyperplasia can prevent removal or cause vessel wall damage upon removal.8
IVC filters are inserted into the vena cava percutaneously via the femoral or jugular approach under fluoroscopy or ultrasound guidance (see Figure 1, p. 16). The filters typically are placed infrarenally, unless there is an indication for a suprarenal filter (e.g., renal vein thrombosis or IVC thrombus extending above the renal veins).7 Complete IVC thrombosis is an absolute contraindication to IVC filter placement, and the relative contraindications include significant coagulopathy and bacteremia.9
The incidence of complications related to IVC filter placement is 4% to 11%. Complications include:
- Insertion-site thrombosis;
- IVC thrombosis;
- Recurrent DVT postphlebitic syndrome;
- Filter migration;
- Erosion of the filter through the vessel wall; and
- Vena caval obstruction.10
A review of the National Hospital Discharge Survey database for trends in IVC filter use in the U.S. found a dramatic increase in the use of IVC filters from 1979 to 1999—to 49,000 patients from 2,000 patients with IVC filters in place. The indications for IVC filter use vary such that it is imperative there are well-designed trials and guidelines to guide appropriate use.11
The Evidence
The 2008 ACCP guidelines on VTE management follow a grading system that classifies recommendations as Grade 1 (strong) or Grade 2 (weak), and classifies the quality of evidence as A (high), B (moderate), or C (low).12 The ACCP guidelines’ recommended first-line treatment for a confirmed DVT is anticoagulation with subcutaneous low-molecular-weight heparin, intravenous unfractionated heparin, monitored subcutaneous heparin, fixed-dose subcutaneous unfractionated heparin, or subcutaneous fondaparinux (all Grade 1A recommendations). The ACCP recommends against the routine use of an IVC filter in addition to anticoagulants (Grade 1A). However, for patients with acute proximal DVT, if anticoagulant therapy is not possible because of the risk of bleeding, IVC filter placement is recommended (Grade 1C). If a patient requires an IVC filter for treatment of an acute DVT as an alternative to anticoagulation, it is recommended to start anticoagulant therapy once the risk of bleeding resolves (Grade 1C).4
The 2008 ACCP guidelines for IVC filter use have a few important changes from the 2004 version. First, the IVC filter placement recommendation for patients with contraindications to anticoagulation was strengthened from Grade 2C to Grade 1C. Second, the 2008 guidelines omitted the early recommendation of IVC filter use for recurrent VTE, despite adequate anticoagulation (Grade 2C).13
Only one randomized study has evaluated the efficacy of IVC filters. All other studies of IVC filters are retrospective or prospective case series.
The PREPIC study randomized 400 patients with proximal DVT considered to be at high risk for PE to receive either an IVC filter or no IVC filter. Additionally, patients were randomized to receive enoxaparin or unfractionated heparin as a bridge to warfarin therapy, which was continued for at least three months. The primary endpoints were recurrent DVT, PE, major bleeding, or death. The patients were followed up at day 12, two years, and then annually up to eight years following randomization.14 At day 12, there were fewer PEs in the group that received filters (OR 0.22, 95% CI, 0.05-0.90). However, at year two, there was no significant difference in PE development in the filter group compared with the no-filter group (OR 0.50, 95% CI, 0.19-1.33).
Additionally, at year two, the filter group was more likely to develop recurrent DVT (OR 1.87, 95% CI, 1.10-3.20). At year eight, there was a significant reduction in the number of PEs in the filter group versus the no-filter group (6.2% vs.15.1%, P=0.008). However, at eight-year followup, IVC filter use was associated with increased DVT (35.7% vs. 27.5%, P=0.042). There was no difference in mortality between the two groups.
In summary, the use of IVC filters was associated with decreased incidence of PE at eight years, offset by higher rates of recurrent DVT and no overall mortality benefit.14,15 Importantly, the indications for IVC filter use in this study differ from the current ACCP guidelines; all patients were given concomitant anticoagulation for at least three months, which might not be possible in patients for whom the ACCP recommends IVC filters.
There are no randomized studies to compare the efficacy of permanent IVC filters and retrievable filters for PE prevention. A retrospective study comparing the clinical effectiveness of the two filter types reported no difference in the rates of symptomatic PE (permanent filter 4% vs. retrievable filter 4.7%, P=0.67) or DVT (11.3% vs. 12.6%, P=0.59). In addition, the frequency of symptomatic IVC thrombosis was similar (1.1% vs. 0.5%, p=0.39).16 A paper reviewing the efficacy of IVC filters reported that permanent filters were associated with a 0%-6.2% rate of PE versus a 0%-1.9% rate with retrievable filters.7 Notably, these studies were not randomized controlled trials—rather, case series—and the indications for IVC filters were not necessarily those currently recommended by the ACCP.
Due to the long-term complications of permanent IVC filters, it is suggested that a retrievable IVC filter be used for patients with temporary contraindications to anticoagulation.17 Comerata et al created a clinical decision-making tool for picking the type of filter to employ. If the duration of contraindication to anticoagulation is short or uncertain, a retrievable filter is recommended.18 Table 1 (p. 15) outlines the recommendations for IVC filter placement.
There are no randomized controlled trials to guide the use of concomitant anticoagulation after filter insertion, although this intervention may be beneficial to prevent DVT propagation, recurrence, or IVC filter thrombosis.5 A meta-analysis of 14 studies evaluating the rates of VTE after IVC filter placement demonstrated a non-statistically significant trend toward fewer VTE events in the patients with an IVC filter and concomitant anticoagulation in comparison with those who solely had an IVC filter (OR 0.64, 95% CI, 0.35-1.2). The duration and degree of anticoagulation was not presented in all of the studies in the meta-analysis, therefore limiting the analysis.19
In addition to the ACCP guidelines, there have been other proposed indications for IVC filter use, including recurrent VTE despite anticoagulation, chronic recurrent PE with pulmonary hypertension, extensive free-floating iliofemoral thrombus, and thrombolysis of ilio-caval thrombus.20 The ACCP guidelines do not specifically address these individual indications, and at this time there are no randomized controlled trials to guide IVC filter use in these cases.
Back to the Case
Our patient developed a significant complication from anticoagulation. Current ACCP guidelines recommend an IVC filter if anticoagulant therapy is contraindicated (Grade 1C). The anticoagulation was discontinued and a retrievable IVC filter was placed. Once a patient no longer has a contraindication for anticoagulation, the ACCP recommends restarting a conventional course of anticoagulation. Thus, once the patient can tolerate anticoagulation, consideration will be given to removal of the retrievable filter.
Bottom Line
An IVC filter should be considered in patients with a DVT who have a contraindication to anticoagulation. Other indications for IVC filter use are not supported by the current literature. TH
Drs. Bhogal and Eid are hospitalist fellows and instructors at Johns Hopkins Bayview Medical Center in Baltimore. Dr. Kantsiper is a hospitalist and assistant professor at Bayview Medical Center.
References
- The Surgeon General’s Call to Action to Prevent Deep Vein Thrombosis and Pulmonary Embolism. U.S. Department of Health & Human Services Web site. Available at: www.surgeongeneral.gov/topics/deepvein/. Accessed Jan. 25, 2010.
- Moser KM, Fedullo PR, LitteJohn JK, Crawford R. Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1994;271(3):223-225.
- Bates SM, Ginsberg JS. Treatment of deep vein thrombosis. N Engl J Med. 2004;351:268-277.
- Kearon C, Kahn SR, Agnelli G, Goldhaber S, Raskob GE, Comerota AJ, American College of Chest Physicians. Antithrombotic therapy for venous theomboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):454S-545S.
- Becker DM, Philbrick JT, Selby JB. Inferior vena cava filters. Indications, safety, effectiveness. Arch Intern Med. 1992;152(10):1985-1994.
- Streiff MB. Vena caval filters: a comprehensive review. Blood. 2000;95(12):3669-3677.
- Chung J, Owen RJ. Using inferior vena cava filters to prevent pulmonary embolism. Can Fam Physician. 2008;54(1):49-55.
- Ku GH. Billett HH. Long lives, short indications. The case for removable inferior cava filters. Thromb Haemost. 2005;93(1):17-22.
- Stavropoulos WS. Inferior vena cava filters. Tech Vasc Interv Radiol. 2004;7(2):91-95.
- Crowther MA. Inferior vena cava filters in the management of venous thromboembolism. Am J Med. 2007;120(10 Suppl 2):S13–S17.
- Stein PD, Kayali F, Olson RE. Twenty-one-year trends in the use of inferior vena cava filters. Arch Intern Med. 2004;164(14):1541-1545.
- Guyatt G, Gutterman D, Baumann MH, et al. Grading strength of recommendations and quality of evidence in clinical guidelines: report from an American College of Chest Physicians task force. Chest. 2006;129(1):174-181.
- Büller HR, Agnelli G, Hull RD, Hyers TM, Prins MH, Raskob GE. Antithrombotic therapy for venous thromboembolic disease: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 Suppl):401S-428S.
- Decousus H, Leizorovicz A, Parent F, et al. A clinical trial of vena caval filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. Prévention du Risque d’Embolie Pulmonaire par Interruption Cave Study Group. N Engl J Med. 1998;338(7):409-415.
- Decousus H, Barral F, Buchmuller-Cordier A, et al. Participating centers eight-year follow-up of patients with permanent vena cava filters in the prevention of pulmonary embolism: the PREPIC randomization croup. Circulation. 2005;112:416-422.
- Kim HS, Young MJ, Narayan AK, Liddell RP, Streiff MB. A comparison of clinical outcomes with retrievable and permanent inferior vena cava filters. J Vasc Interv Radiol. 2008:19(3):393-399.
- Houman Fekrazad M, Lopes RD, Stashenko GJ, Alexander JH, Garcia D. Treatment of venous thromboembolism: guidelines translated for the clinician. J Thromb Thrombolysis. 2009; 28(3):270–275.
- Comerota AJ. Retrievable IVC filters: a decision matrix for appropriate utilization. Perspect Vasc Surg Endovasc Ther. 2006;18(1):11-17.
- Ray CE Jr, Prochazka A. The need for anticoagulation following inferior vena cava filter placement: systematic review. Cardiovasc Intervent Radiol. 2008; 31(2):316-324.
- Hajduk B, Tomkowski WZ, Malek G, Davidson BL. Vena cava filter occlusion and venous thromboembolism risk in persistently anticoagulated patients: A prospective, observational cohort study. Chest. 2009.
Case
A 67-year-old man with a history of hypertension presents with a swollen right lower extremity. An ultrasound reveals a DVT, and he is commenced on low-molecular-weight heparin and warfarin. Two days later, he develops slurred speech and right-sided weakness. A head CT reveals an intracranial hemorrhage. When should an inferior vena cava (IVC) filter be utilized for treatment of DVT?
Overview
It is estimated that 350,000 to 600,000 Americans develop a VTE each year.1 Patients with a DVT are at high risk of developing a pulmonary embolism (PE). In a multicenter study, nearly 40% of patients admitted with a DVT had evidence of a PE on ventilation perfusion scan.2 Treatment of a DVT is aimed at preventing the extension of the DVT and embolization.3 The American College of Chest Physicians (ACCP) recommends anticoagulation as the primary DVT treatment (Grade 1A).4 However, IVC filters might be considered when anticoagulation is contraindicated.
In 1868, Trousseau created the conceptual model of surgical interruption of the IVC to prevent PE. However, it wasn’t until 1959 by Bottini that the surgical interruption was successfully performed.5 The Mobin-Uddin filter was introduced in 1967 as the first mechanical IVC filter.6 IVC filters mechanically trap the DVT, preventing emboli from traveling into the pulmonary vasculature.7
There are two classes of IVC filters: permanent filters and removable filters. Removable filters include both temporary filters and retrievable filters. Temporary filters are attached to a catheter that exits the skin and therefore must be removed due to the risk of infection and embolization.7 Retrievable filters are similar in design to permanent filters but are designed to be removed. However, this must be done with caution, as neointimal hyperplasia can prevent removal or cause vessel wall damage upon removal.8
IVC filters are inserted into the vena cava percutaneously via the femoral or jugular approach under fluoroscopy or ultrasound guidance (see Figure 1, p. 16). The filters typically are placed infrarenally, unless there is an indication for a suprarenal filter (e.g., renal vein thrombosis or IVC thrombus extending above the renal veins).7 Complete IVC thrombosis is an absolute contraindication to IVC filter placement, and the relative contraindications include significant coagulopathy and bacteremia.9
The incidence of complications related to IVC filter placement is 4% to 11%. Complications include:
- Insertion-site thrombosis;
- IVC thrombosis;
- Recurrent DVT postphlebitic syndrome;
- Filter migration;
- Erosion of the filter through the vessel wall; and
- Vena caval obstruction.10
A review of the National Hospital Discharge Survey database for trends in IVC filter use in the U.S. found a dramatic increase in the use of IVC filters from 1979 to 1999—to 49,000 patients from 2,000 patients with IVC filters in place. The indications for IVC filter use vary such that it is imperative there are well-designed trials and guidelines to guide appropriate use.11
The Evidence
The 2008 ACCP guidelines on VTE management follow a grading system that classifies recommendations as Grade 1 (strong) or Grade 2 (weak), and classifies the quality of evidence as A (high), B (moderate), or C (low).12 The ACCP guidelines’ recommended first-line treatment for a confirmed DVT is anticoagulation with subcutaneous low-molecular-weight heparin, intravenous unfractionated heparin, monitored subcutaneous heparin, fixed-dose subcutaneous unfractionated heparin, or subcutaneous fondaparinux (all Grade 1A recommendations). The ACCP recommends against the routine use of an IVC filter in addition to anticoagulants (Grade 1A). However, for patients with acute proximal DVT, if anticoagulant therapy is not possible because of the risk of bleeding, IVC filter placement is recommended (Grade 1C). If a patient requires an IVC filter for treatment of an acute DVT as an alternative to anticoagulation, it is recommended to start anticoagulant therapy once the risk of bleeding resolves (Grade 1C).4
The 2008 ACCP guidelines for IVC filter use have a few important changes from the 2004 version. First, the IVC filter placement recommendation for patients with contraindications to anticoagulation was strengthened from Grade 2C to Grade 1C. Second, the 2008 guidelines omitted the early recommendation of IVC filter use for recurrent VTE, despite adequate anticoagulation (Grade 2C).13
Only one randomized study has evaluated the efficacy of IVC filters. All other studies of IVC filters are retrospective or prospective case series.
The PREPIC study randomized 400 patients with proximal DVT considered to be at high risk for PE to receive either an IVC filter or no IVC filter. Additionally, patients were randomized to receive enoxaparin or unfractionated heparin as a bridge to warfarin therapy, which was continued for at least three months. The primary endpoints were recurrent DVT, PE, major bleeding, or death. The patients were followed up at day 12, two years, and then annually up to eight years following randomization.14 At day 12, there were fewer PEs in the group that received filters (OR 0.22, 95% CI, 0.05-0.90). However, at year two, there was no significant difference in PE development in the filter group compared with the no-filter group (OR 0.50, 95% CI, 0.19-1.33).
Additionally, at year two, the filter group was more likely to develop recurrent DVT (OR 1.87, 95% CI, 1.10-3.20). At year eight, there was a significant reduction in the number of PEs in the filter group versus the no-filter group (6.2% vs.15.1%, P=0.008). However, at eight-year followup, IVC filter use was associated with increased DVT (35.7% vs. 27.5%, P=0.042). There was no difference in mortality between the two groups.
In summary, the use of IVC filters was associated with decreased incidence of PE at eight years, offset by higher rates of recurrent DVT and no overall mortality benefit.14,15 Importantly, the indications for IVC filter use in this study differ from the current ACCP guidelines; all patients were given concomitant anticoagulation for at least three months, which might not be possible in patients for whom the ACCP recommends IVC filters.
There are no randomized studies to compare the efficacy of permanent IVC filters and retrievable filters for PE prevention. A retrospective study comparing the clinical effectiveness of the two filter types reported no difference in the rates of symptomatic PE (permanent filter 4% vs. retrievable filter 4.7%, P=0.67) or DVT (11.3% vs. 12.6%, P=0.59). In addition, the frequency of symptomatic IVC thrombosis was similar (1.1% vs. 0.5%, p=0.39).16 A paper reviewing the efficacy of IVC filters reported that permanent filters were associated with a 0%-6.2% rate of PE versus a 0%-1.9% rate with retrievable filters.7 Notably, these studies were not randomized controlled trials—rather, case series—and the indications for IVC filters were not necessarily those currently recommended by the ACCP.
Due to the long-term complications of permanent IVC filters, it is suggested that a retrievable IVC filter be used for patients with temporary contraindications to anticoagulation.17 Comerata et al created a clinical decision-making tool for picking the type of filter to employ. If the duration of contraindication to anticoagulation is short or uncertain, a retrievable filter is recommended.18 Table 1 (p. 15) outlines the recommendations for IVC filter placement.
There are no randomized controlled trials to guide the use of concomitant anticoagulation after filter insertion, although this intervention may be beneficial to prevent DVT propagation, recurrence, or IVC filter thrombosis.5 A meta-analysis of 14 studies evaluating the rates of VTE after IVC filter placement demonstrated a non-statistically significant trend toward fewer VTE events in the patients with an IVC filter and concomitant anticoagulation in comparison with those who solely had an IVC filter (OR 0.64, 95% CI, 0.35-1.2). The duration and degree of anticoagulation was not presented in all of the studies in the meta-analysis, therefore limiting the analysis.19
In addition to the ACCP guidelines, there have been other proposed indications for IVC filter use, including recurrent VTE despite anticoagulation, chronic recurrent PE with pulmonary hypertension, extensive free-floating iliofemoral thrombus, and thrombolysis of ilio-caval thrombus.20 The ACCP guidelines do not specifically address these individual indications, and at this time there are no randomized controlled trials to guide IVC filter use in these cases.
Back to the Case
Our patient developed a significant complication from anticoagulation. Current ACCP guidelines recommend an IVC filter if anticoagulant therapy is contraindicated (Grade 1C). The anticoagulation was discontinued and a retrievable IVC filter was placed. Once a patient no longer has a contraindication for anticoagulation, the ACCP recommends restarting a conventional course of anticoagulation. Thus, once the patient can tolerate anticoagulation, consideration will be given to removal of the retrievable filter.
Bottom Line
An IVC filter should be considered in patients with a DVT who have a contraindication to anticoagulation. Other indications for IVC filter use are not supported by the current literature. TH
Drs. Bhogal and Eid are hospitalist fellows and instructors at Johns Hopkins Bayview Medical Center in Baltimore. Dr. Kantsiper is a hospitalist and assistant professor at Bayview Medical Center.
References
- The Surgeon General’s Call to Action to Prevent Deep Vein Thrombosis and Pulmonary Embolism. U.S. Department of Health & Human Services Web site. Available at: www.surgeongeneral.gov/topics/deepvein/. Accessed Jan. 25, 2010.
- Moser KM, Fedullo PR, LitteJohn JK, Crawford R. Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1994;271(3):223-225.
- Bates SM, Ginsberg JS. Treatment of deep vein thrombosis. N Engl J Med. 2004;351:268-277.
- Kearon C, Kahn SR, Agnelli G, Goldhaber S, Raskob GE, Comerota AJ, American College of Chest Physicians. Antithrombotic therapy for venous theomboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):454S-545S.
- Becker DM, Philbrick JT, Selby JB. Inferior vena cava filters. Indications, safety, effectiveness. Arch Intern Med. 1992;152(10):1985-1994.
- Streiff MB. Vena caval filters: a comprehensive review. Blood. 2000;95(12):3669-3677.
- Chung J, Owen RJ. Using inferior vena cava filters to prevent pulmonary embolism. Can Fam Physician. 2008;54(1):49-55.
- Ku GH. Billett HH. Long lives, short indications. The case for removable inferior cava filters. Thromb Haemost. 2005;93(1):17-22.
- Stavropoulos WS. Inferior vena cava filters. Tech Vasc Interv Radiol. 2004;7(2):91-95.
- Crowther MA. Inferior vena cava filters in the management of venous thromboembolism. Am J Med. 2007;120(10 Suppl 2):S13–S17.
- Stein PD, Kayali F, Olson RE. Twenty-one-year trends in the use of inferior vena cava filters. Arch Intern Med. 2004;164(14):1541-1545.
- Guyatt G, Gutterman D, Baumann MH, et al. Grading strength of recommendations and quality of evidence in clinical guidelines: report from an American College of Chest Physicians task force. Chest. 2006;129(1):174-181.
- Büller HR, Agnelli G, Hull RD, Hyers TM, Prins MH, Raskob GE. Antithrombotic therapy for venous thromboembolic disease: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 Suppl):401S-428S.
- Decousus H, Leizorovicz A, Parent F, et al. A clinical trial of vena caval filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. Prévention du Risque d’Embolie Pulmonaire par Interruption Cave Study Group. N Engl J Med. 1998;338(7):409-415.
- Decousus H, Barral F, Buchmuller-Cordier A, et al. Participating centers eight-year follow-up of patients with permanent vena cava filters in the prevention of pulmonary embolism: the PREPIC randomization croup. Circulation. 2005;112:416-422.
- Kim HS, Young MJ, Narayan AK, Liddell RP, Streiff MB. A comparison of clinical outcomes with retrievable and permanent inferior vena cava filters. J Vasc Interv Radiol. 2008:19(3):393-399.
- Houman Fekrazad M, Lopes RD, Stashenko GJ, Alexander JH, Garcia D. Treatment of venous thromboembolism: guidelines translated for the clinician. J Thromb Thrombolysis. 2009; 28(3):270–275.
- Comerota AJ. Retrievable IVC filters: a decision matrix for appropriate utilization. Perspect Vasc Surg Endovasc Ther. 2006;18(1):11-17.
- Ray CE Jr, Prochazka A. The need for anticoagulation following inferior vena cava filter placement: systematic review. Cardiovasc Intervent Radiol. 2008; 31(2):316-324.
- Hajduk B, Tomkowski WZ, Malek G, Davidson BL. Vena cava filter occlusion and venous thromboembolism risk in persistently anticoagulated patients: A prospective, observational cohort study. Chest. 2009.
Case
A 67-year-old man with a history of hypertension presents with a swollen right lower extremity. An ultrasound reveals a DVT, and he is commenced on low-molecular-weight heparin and warfarin. Two days later, he develops slurred speech and right-sided weakness. A head CT reveals an intracranial hemorrhage. When should an inferior vena cava (IVC) filter be utilized for treatment of DVT?
Overview
It is estimated that 350,000 to 600,000 Americans develop a VTE each year.1 Patients with a DVT are at high risk of developing a pulmonary embolism (PE). In a multicenter study, nearly 40% of patients admitted with a DVT had evidence of a PE on ventilation perfusion scan.2 Treatment of a DVT is aimed at preventing the extension of the DVT and embolization.3 The American College of Chest Physicians (ACCP) recommends anticoagulation as the primary DVT treatment (Grade 1A).4 However, IVC filters might be considered when anticoagulation is contraindicated.
In 1868, Trousseau created the conceptual model of surgical interruption of the IVC to prevent PE. However, it wasn’t until 1959 by Bottini that the surgical interruption was successfully performed.5 The Mobin-Uddin filter was introduced in 1967 as the first mechanical IVC filter.6 IVC filters mechanically trap the DVT, preventing emboli from traveling into the pulmonary vasculature.7
There are two classes of IVC filters: permanent filters and removable filters. Removable filters include both temporary filters and retrievable filters. Temporary filters are attached to a catheter that exits the skin and therefore must be removed due to the risk of infection and embolization.7 Retrievable filters are similar in design to permanent filters but are designed to be removed. However, this must be done with caution, as neointimal hyperplasia can prevent removal or cause vessel wall damage upon removal.8
IVC filters are inserted into the vena cava percutaneously via the femoral or jugular approach under fluoroscopy or ultrasound guidance (see Figure 1, p. 16). The filters typically are placed infrarenally, unless there is an indication for a suprarenal filter (e.g., renal vein thrombosis or IVC thrombus extending above the renal veins).7 Complete IVC thrombosis is an absolute contraindication to IVC filter placement, and the relative contraindications include significant coagulopathy and bacteremia.9
The incidence of complications related to IVC filter placement is 4% to 11%. Complications include:
- Insertion-site thrombosis;
- IVC thrombosis;
- Recurrent DVT postphlebitic syndrome;
- Filter migration;
- Erosion of the filter through the vessel wall; and
- Vena caval obstruction.10
A review of the National Hospital Discharge Survey database for trends in IVC filter use in the U.S. found a dramatic increase in the use of IVC filters from 1979 to 1999—to 49,000 patients from 2,000 patients with IVC filters in place. The indications for IVC filter use vary such that it is imperative there are well-designed trials and guidelines to guide appropriate use.11
The Evidence
The 2008 ACCP guidelines on VTE management follow a grading system that classifies recommendations as Grade 1 (strong) or Grade 2 (weak), and classifies the quality of evidence as A (high), B (moderate), or C (low).12 The ACCP guidelines’ recommended first-line treatment for a confirmed DVT is anticoagulation with subcutaneous low-molecular-weight heparin, intravenous unfractionated heparin, monitored subcutaneous heparin, fixed-dose subcutaneous unfractionated heparin, or subcutaneous fondaparinux (all Grade 1A recommendations). The ACCP recommends against the routine use of an IVC filter in addition to anticoagulants (Grade 1A). However, for patients with acute proximal DVT, if anticoagulant therapy is not possible because of the risk of bleeding, IVC filter placement is recommended (Grade 1C). If a patient requires an IVC filter for treatment of an acute DVT as an alternative to anticoagulation, it is recommended to start anticoagulant therapy once the risk of bleeding resolves (Grade 1C).4
The 2008 ACCP guidelines for IVC filter use have a few important changes from the 2004 version. First, the IVC filter placement recommendation for patients with contraindications to anticoagulation was strengthened from Grade 2C to Grade 1C. Second, the 2008 guidelines omitted the early recommendation of IVC filter use for recurrent VTE, despite adequate anticoagulation (Grade 2C).13
Only one randomized study has evaluated the efficacy of IVC filters. All other studies of IVC filters are retrospective or prospective case series.
The PREPIC study randomized 400 patients with proximal DVT considered to be at high risk for PE to receive either an IVC filter or no IVC filter. Additionally, patients were randomized to receive enoxaparin or unfractionated heparin as a bridge to warfarin therapy, which was continued for at least three months. The primary endpoints were recurrent DVT, PE, major bleeding, or death. The patients were followed up at day 12, two years, and then annually up to eight years following randomization.14 At day 12, there were fewer PEs in the group that received filters (OR 0.22, 95% CI, 0.05-0.90). However, at year two, there was no significant difference in PE development in the filter group compared with the no-filter group (OR 0.50, 95% CI, 0.19-1.33).
Additionally, at year two, the filter group was more likely to develop recurrent DVT (OR 1.87, 95% CI, 1.10-3.20). At year eight, there was a significant reduction in the number of PEs in the filter group versus the no-filter group (6.2% vs.15.1%, P=0.008). However, at eight-year followup, IVC filter use was associated with increased DVT (35.7% vs. 27.5%, P=0.042). There was no difference in mortality between the two groups.
In summary, the use of IVC filters was associated with decreased incidence of PE at eight years, offset by higher rates of recurrent DVT and no overall mortality benefit.14,15 Importantly, the indications for IVC filter use in this study differ from the current ACCP guidelines; all patients were given concomitant anticoagulation for at least three months, which might not be possible in patients for whom the ACCP recommends IVC filters.
There are no randomized studies to compare the efficacy of permanent IVC filters and retrievable filters for PE prevention. A retrospective study comparing the clinical effectiveness of the two filter types reported no difference in the rates of symptomatic PE (permanent filter 4% vs. retrievable filter 4.7%, P=0.67) or DVT (11.3% vs. 12.6%, P=0.59). In addition, the frequency of symptomatic IVC thrombosis was similar (1.1% vs. 0.5%, p=0.39).16 A paper reviewing the efficacy of IVC filters reported that permanent filters were associated with a 0%-6.2% rate of PE versus a 0%-1.9% rate with retrievable filters.7 Notably, these studies were not randomized controlled trials—rather, case series—and the indications for IVC filters were not necessarily those currently recommended by the ACCP.
Due to the long-term complications of permanent IVC filters, it is suggested that a retrievable IVC filter be used for patients with temporary contraindications to anticoagulation.17 Comerata et al created a clinical decision-making tool for picking the type of filter to employ. If the duration of contraindication to anticoagulation is short or uncertain, a retrievable filter is recommended.18 Table 1 (p. 15) outlines the recommendations for IVC filter placement.
There are no randomized controlled trials to guide the use of concomitant anticoagulation after filter insertion, although this intervention may be beneficial to prevent DVT propagation, recurrence, or IVC filter thrombosis.5 A meta-analysis of 14 studies evaluating the rates of VTE after IVC filter placement demonstrated a non-statistically significant trend toward fewer VTE events in the patients with an IVC filter and concomitant anticoagulation in comparison with those who solely had an IVC filter (OR 0.64, 95% CI, 0.35-1.2). The duration and degree of anticoagulation was not presented in all of the studies in the meta-analysis, therefore limiting the analysis.19
In addition to the ACCP guidelines, there have been other proposed indications for IVC filter use, including recurrent VTE despite anticoagulation, chronic recurrent PE with pulmonary hypertension, extensive free-floating iliofemoral thrombus, and thrombolysis of ilio-caval thrombus.20 The ACCP guidelines do not specifically address these individual indications, and at this time there are no randomized controlled trials to guide IVC filter use in these cases.
Back to the Case
Our patient developed a significant complication from anticoagulation. Current ACCP guidelines recommend an IVC filter if anticoagulant therapy is contraindicated (Grade 1C). The anticoagulation was discontinued and a retrievable IVC filter was placed. Once a patient no longer has a contraindication for anticoagulation, the ACCP recommends restarting a conventional course of anticoagulation. Thus, once the patient can tolerate anticoagulation, consideration will be given to removal of the retrievable filter.
Bottom Line
An IVC filter should be considered in patients with a DVT who have a contraindication to anticoagulation. Other indications for IVC filter use are not supported by the current literature. TH
Drs. Bhogal and Eid are hospitalist fellows and instructors at Johns Hopkins Bayview Medical Center in Baltimore. Dr. Kantsiper is a hospitalist and assistant professor at Bayview Medical Center.
References
- The Surgeon General’s Call to Action to Prevent Deep Vein Thrombosis and Pulmonary Embolism. U.S. Department of Health & Human Services Web site. Available at: www.surgeongeneral.gov/topics/deepvein/. Accessed Jan. 25, 2010.
- Moser KM, Fedullo PR, LitteJohn JK, Crawford R. Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1994;271(3):223-225.
- Bates SM, Ginsberg JS. Treatment of deep vein thrombosis. N Engl J Med. 2004;351:268-277.
- Kearon C, Kahn SR, Agnelli G, Goldhaber S, Raskob GE, Comerota AJ, American College of Chest Physicians. Antithrombotic therapy for venous theomboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):454S-545S.
- Becker DM, Philbrick JT, Selby JB. Inferior vena cava filters. Indications, safety, effectiveness. Arch Intern Med. 1992;152(10):1985-1994.
- Streiff MB. Vena caval filters: a comprehensive review. Blood. 2000;95(12):3669-3677.
- Chung J, Owen RJ. Using inferior vena cava filters to prevent pulmonary embolism. Can Fam Physician. 2008;54(1):49-55.
- Ku GH. Billett HH. Long lives, short indications. The case for removable inferior cava filters. Thromb Haemost. 2005;93(1):17-22.
- Stavropoulos WS. Inferior vena cava filters. Tech Vasc Interv Radiol. 2004;7(2):91-95.
- Crowther MA. Inferior vena cava filters in the management of venous thromboembolism. Am J Med. 2007;120(10 Suppl 2):S13–S17.
- Stein PD, Kayali F, Olson RE. Twenty-one-year trends in the use of inferior vena cava filters. Arch Intern Med. 2004;164(14):1541-1545.
- Guyatt G, Gutterman D, Baumann MH, et al. Grading strength of recommendations and quality of evidence in clinical guidelines: report from an American College of Chest Physicians task force. Chest. 2006;129(1):174-181.
- Büller HR, Agnelli G, Hull RD, Hyers TM, Prins MH, Raskob GE. Antithrombotic therapy for venous thromboembolic disease: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 Suppl):401S-428S.
- Decousus H, Leizorovicz A, Parent F, et al. A clinical trial of vena caval filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. Prévention du Risque d’Embolie Pulmonaire par Interruption Cave Study Group. N Engl J Med. 1998;338(7):409-415.
- Decousus H, Barral F, Buchmuller-Cordier A, et al. Participating centers eight-year follow-up of patients with permanent vena cava filters in the prevention of pulmonary embolism: the PREPIC randomization croup. Circulation. 2005;112:416-422.
- Kim HS, Young MJ, Narayan AK, Liddell RP, Streiff MB. A comparison of clinical outcomes with retrievable and permanent inferior vena cava filters. J Vasc Interv Radiol. 2008:19(3):393-399.
- Houman Fekrazad M, Lopes RD, Stashenko GJ, Alexander JH, Garcia D. Treatment of venous thromboembolism: guidelines translated for the clinician. J Thromb Thrombolysis. 2009; 28(3):270–275.
- Comerota AJ. Retrievable IVC filters: a decision matrix for appropriate utilization. Perspect Vasc Surg Endovasc Ther. 2006;18(1):11-17.
- Ray CE Jr, Prochazka A. The need for anticoagulation following inferior vena cava filter placement: systematic review. Cardiovasc Intervent Radiol. 2008; 31(2):316-324.
- Hajduk B, Tomkowski WZ, Malek G, Davidson BL. Vena cava filter occlusion and venous thromboembolism risk in persistently anticoagulated patients: A prospective, observational cohort study. Chest. 2009.