User login
Does US measurement of cervical length to determine the need for cerclage reduce preterm delivery?
In their discussion of the findings, Simcox and colleagues assert that a trial to settle the questions frequently raised in the cerclage debate would require thousands of patients and may not be feasible.
Rigorous trial is impressive,
but doesn’t settle key questions
The authors performed an excellent randomized, controlled trial, and their intention-to-treat analysis is laudable. They are also to be congratulated for remaining focused on the primary outcome of delivery before 34 weeks’ gestation. It is notable that the primary outcome was essentially the same in each group, regardless of the treatment, be it 1) US screening and cerclage for cervical length 2) no screening and cerclage for historical indications. I recall a conference on prematurity from the mid-1980s that included, as one of its conclusions, the observation that as many as 70% of patients who have historical “indications” for cerclage will deliver at term in their next pregnancy if left untreated.
Unresolved questions in regard to cervical cerclage include:
- What is the best way to determine who is a candidate?
- What is the best type of cerclage?
- What is the most appropriate outcome to be measured?
- Is there a place in practice for “universal” screening of cervical length?
- What is the true cost (in terms of both dollars and morbidity) of intervention versus no intervention?
- What are the medicolegal implications of each approach?
High-risk women may benefit from US imaging, but the data from this study do not support that conclusion. Nor is the best type of cerclage defined, though there is ample opinion on this topic.
Is 34 weeks’ gestation the appropriate primary outcome? More and more, we read about late preterm or so-called near-term outcomes being less optimal than they once were thought to be—though delivery at 34 to 37 weeks would seem to be preferable to delivery at less than 34 weeks.
The cost of each approach is unclear. How many “unnecessary” cerclages would be needed to prevent one very-low-birth-weight delivery? And how “risky” is elective cerclage placement in skilled hands?
Finally, not many patients or physicians are likely to want to embrace a wait-and-see approach if they have already had one or more adverse outcomes, and the risk of doing nothing may be considerably greater in medicolegal terms than the risk of proceeding with what may be an unnecessary intervention that ends in a term or near-term delivery.
On the basis of these results, I think the practitioner should rely on history to make a clinical judgment about the need for cerclage. Ultrasonographic imaging may not only be of little help, but it may lead to greater intervention than would otherwise be needed. Perhaps a return to clinical basics, such as detailed history taking and physical examination, is a good message for these economic times.—JOHN T. REPKE, MD
In their discussion of the findings, Simcox and colleagues assert that a trial to settle the questions frequently raised in the cerclage debate would require thousands of patients and may not be feasible.
Rigorous trial is impressive,
but doesn’t settle key questions
The authors performed an excellent randomized, controlled trial, and their intention-to-treat analysis is laudable. They are also to be congratulated for remaining focused on the primary outcome of delivery before 34 weeks’ gestation. It is notable that the primary outcome was essentially the same in each group, regardless of the treatment, be it 1) US screening and cerclage for cervical length 2) no screening and cerclage for historical indications. I recall a conference on prematurity from the mid-1980s that included, as one of its conclusions, the observation that as many as 70% of patients who have historical “indications” for cerclage will deliver at term in their next pregnancy if left untreated.
Unresolved questions in regard to cervical cerclage include:
- What is the best way to determine who is a candidate?
- What is the best type of cerclage?
- What is the most appropriate outcome to be measured?
- Is there a place in practice for “universal” screening of cervical length?
- What is the true cost (in terms of both dollars and morbidity) of intervention versus no intervention?
- What are the medicolegal implications of each approach?
High-risk women may benefit from US imaging, but the data from this study do not support that conclusion. Nor is the best type of cerclage defined, though there is ample opinion on this topic.
Is 34 weeks’ gestation the appropriate primary outcome? More and more, we read about late preterm or so-called near-term outcomes being less optimal than they once were thought to be—though delivery at 34 to 37 weeks would seem to be preferable to delivery at less than 34 weeks.
The cost of each approach is unclear. How many “unnecessary” cerclages would be needed to prevent one very-low-birth-weight delivery? And how “risky” is elective cerclage placement in skilled hands?
Finally, not many patients or physicians are likely to want to embrace a wait-and-see approach if they have already had one or more adverse outcomes, and the risk of doing nothing may be considerably greater in medicolegal terms than the risk of proceeding with what may be an unnecessary intervention that ends in a term or near-term delivery.
On the basis of these results, I think the practitioner should rely on history to make a clinical judgment about the need for cerclage. Ultrasonographic imaging may not only be of little help, but it may lead to greater intervention than would otherwise be needed. Perhaps a return to clinical basics, such as detailed history taking and physical examination, is a good message for these economic times.—JOHN T. REPKE, MD
In their discussion of the findings, Simcox and colleagues assert that a trial to settle the questions frequently raised in the cerclage debate would require thousands of patients and may not be feasible.
Rigorous trial is impressive,
but doesn’t settle key questions
The authors performed an excellent randomized, controlled trial, and their intention-to-treat analysis is laudable. They are also to be congratulated for remaining focused on the primary outcome of delivery before 34 weeks’ gestation. It is notable that the primary outcome was essentially the same in each group, regardless of the treatment, be it 1) US screening and cerclage for cervical length 2) no screening and cerclage for historical indications. I recall a conference on prematurity from the mid-1980s that included, as one of its conclusions, the observation that as many as 70% of patients who have historical “indications” for cerclage will deliver at term in their next pregnancy if left untreated.
Unresolved questions in regard to cervical cerclage include:
- What is the best way to determine who is a candidate?
- What is the best type of cerclage?
- What is the most appropriate outcome to be measured?
- Is there a place in practice for “universal” screening of cervical length?
- What is the true cost (in terms of both dollars and morbidity) of intervention versus no intervention?
- What are the medicolegal implications of each approach?
High-risk women may benefit from US imaging, but the data from this study do not support that conclusion. Nor is the best type of cerclage defined, though there is ample opinion on this topic.
Is 34 weeks’ gestation the appropriate primary outcome? More and more, we read about late preterm or so-called near-term outcomes being less optimal than they once were thought to be—though delivery at 34 to 37 weeks would seem to be preferable to delivery at less than 34 weeks.
The cost of each approach is unclear. How many “unnecessary” cerclages would be needed to prevent one very-low-birth-weight delivery? And how “risky” is elective cerclage placement in skilled hands?
Finally, not many patients or physicians are likely to want to embrace a wait-and-see approach if they have already had one or more adverse outcomes, and the risk of doing nothing may be considerably greater in medicolegal terms than the risk of proceeding with what may be an unnecessary intervention that ends in a term or near-term delivery.
On the basis of these results, I think the practitioner should rely on history to make a clinical judgment about the need for cerclage. Ultrasonographic imaging may not only be of little help, but it may lead to greater intervention than would otherwise be needed. Perhaps a return to clinical basics, such as detailed history taking and physical examination, is a good message for these economic times.—JOHN T. REPKE, MD
Can ovulation induction be accelerated in women who have PCOS-related infertility?
Polycystic ovary syndrome is the most common cause of anovulatory infertility, and expert consensus points to clomiphene as first-line therapy.1
Under the conventional protocol, clomiphene is given early in the follicular phase, with midluteal monitoring for ovulation. If ovulation is not detected, progestin is administered to induce a withdrawal bleed, and the dosage of clomiphene is increased in the next cycle. Under this protocol, the clomiphene regimen can last as long as 90 days.
Hurst and colleagues propose a 28-day dosage-escalation method, relying on earlier ultrasonography to document follicular development and, in its absence, immediately “rechallenging” the patient with a higher dosage of clomiphene (FIGURE). They present intriguing data from a small, preliminary case series of 31 subjects who used this stair-step protocol. Seventy-four percent of these women ovulated by the end of the 28-day monitoring period, compared with 34% in 89 days among a historical control group using the traditional protocol.
Although a single stair-step cycle was more expensive than a traditional cycle, it has the potential to be less expensive when the rate of ovulation is taken into account.
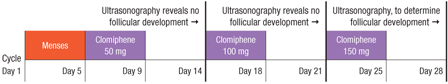
FIGURE Stair-step clomiphene protocol skips progestin administration
Accelerated regimen may have a number of negatives
At first glance, what’s not to like about a protocol that increases the likelihood of ovulation with significant savings in time and cost for the patient?
First, there are methodologic concerns when a control group is not studied by means of a randomized, controlled trial—or even at the same center as the treatment arm—but is merely created from published data from a textbook.
Second, the goal of infertility therapy is live birth, not ovulation. Our studies have demonstrated significant differences in fecundity for each successful ovulation using different medications, suggesting that not every ovulation is the same.2 In the study by Hurst and colleagues, fecundity (live birth by ovulation rate) was 13%, compared with 10% using clomiphene in our large, multi-center trial—not much of an improvement, although, admittedly, the study by Hurst and colleagues was underpowered to address this question.2
Third, there are concerns about potential adverse effects of the accelerated clomiphene regimen on the patient or fetus. Clomiphene has a prolonged half-life of 5 to 7 days, with some metabolites persisting for months. What are the cumulative effects of such aggressive dosage escalation over such a short period of time?
The current Clomid package insert recommends a maximum dosage of 500 mg/cycle. A nonresponder in the stair-step protocol could consume 1,500 mg of clomiphene over a 20-day period.
Hurst and colleagues do not mention side effects, but it would be reasonable to expect an increased rate of hot flashes or mood changes. And what about more concerning signs such as visual changes?
Although clomiphene has no known human teratogenic effects (it is listed as pregnancy category “X”), the stair-step protocol could theoretically produce higher levels of fetal metabolites during the period of organ-ogenesis, with unknown effects.
Before we rush to adopt this accelerated regimen, we need studies that better address the maternal–fetal risk–benefit ratio. However, this study does provide evidence that a progestin withdrawal bleed is not mandatory in the nonresponder.—RICHARD S. LEGRO, MD
1. Thessaloniki ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Consensus on infertility treatment related to polycystic ovary syndrome. Fertil Steril. 2008;89:505-522.
2. Legro RS, Barnhart HX, Schlaff WD. Cooperative Multicenter Reproductive Medicine Network. Clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N Engl J Med. 2007;356:551-566.
Polycystic ovary syndrome is the most common cause of anovulatory infertility, and expert consensus points to clomiphene as first-line therapy.1
Under the conventional protocol, clomiphene is given early in the follicular phase, with midluteal monitoring for ovulation. If ovulation is not detected, progestin is administered to induce a withdrawal bleed, and the dosage of clomiphene is increased in the next cycle. Under this protocol, the clomiphene regimen can last as long as 90 days.
Hurst and colleagues propose a 28-day dosage-escalation method, relying on earlier ultrasonography to document follicular development and, in its absence, immediately “rechallenging” the patient with a higher dosage of clomiphene (FIGURE). They present intriguing data from a small, preliminary case series of 31 subjects who used this stair-step protocol. Seventy-four percent of these women ovulated by the end of the 28-day monitoring period, compared with 34% in 89 days among a historical control group using the traditional protocol.
Although a single stair-step cycle was more expensive than a traditional cycle, it has the potential to be less expensive when the rate of ovulation is taken into account.

FIGURE Stair-step clomiphene protocol skips progestin administration
Accelerated regimen may have a number of negatives
At first glance, what’s not to like about a protocol that increases the likelihood of ovulation with significant savings in time and cost for the patient?
First, there are methodologic concerns when a control group is not studied by means of a randomized, controlled trial—or even at the same center as the treatment arm—but is merely created from published data from a textbook.
Second, the goal of infertility therapy is live birth, not ovulation. Our studies have demonstrated significant differences in fecundity for each successful ovulation using different medications, suggesting that not every ovulation is the same.2 In the study by Hurst and colleagues, fecundity (live birth by ovulation rate) was 13%, compared with 10% using clomiphene in our large, multi-center trial—not much of an improvement, although, admittedly, the study by Hurst and colleagues was underpowered to address this question.2
Third, there are concerns about potential adverse effects of the accelerated clomiphene regimen on the patient or fetus. Clomiphene has a prolonged half-life of 5 to 7 days, with some metabolites persisting for months. What are the cumulative effects of such aggressive dosage escalation over such a short period of time?
The current Clomid package insert recommends a maximum dosage of 500 mg/cycle. A nonresponder in the stair-step protocol could consume 1,500 mg of clomiphene over a 20-day period.
Hurst and colleagues do not mention side effects, but it would be reasonable to expect an increased rate of hot flashes or mood changes. And what about more concerning signs such as visual changes?
Although clomiphene has no known human teratogenic effects (it is listed as pregnancy category “X”), the stair-step protocol could theoretically produce higher levels of fetal metabolites during the period of organ-ogenesis, with unknown effects.
Before we rush to adopt this accelerated regimen, we need studies that better address the maternal–fetal risk–benefit ratio. However, this study does provide evidence that a progestin withdrawal bleed is not mandatory in the nonresponder.—RICHARD S. LEGRO, MD
Polycystic ovary syndrome is the most common cause of anovulatory infertility, and expert consensus points to clomiphene as first-line therapy.1
Under the conventional protocol, clomiphene is given early in the follicular phase, with midluteal monitoring for ovulation. If ovulation is not detected, progestin is administered to induce a withdrawal bleed, and the dosage of clomiphene is increased in the next cycle. Under this protocol, the clomiphene regimen can last as long as 90 days.
Hurst and colleagues propose a 28-day dosage-escalation method, relying on earlier ultrasonography to document follicular development and, in its absence, immediately “rechallenging” the patient with a higher dosage of clomiphene (FIGURE). They present intriguing data from a small, preliminary case series of 31 subjects who used this stair-step protocol. Seventy-four percent of these women ovulated by the end of the 28-day monitoring period, compared with 34% in 89 days among a historical control group using the traditional protocol.
Although a single stair-step cycle was more expensive than a traditional cycle, it has the potential to be less expensive when the rate of ovulation is taken into account.

FIGURE Stair-step clomiphene protocol skips progestin administration
Accelerated regimen may have a number of negatives
At first glance, what’s not to like about a protocol that increases the likelihood of ovulation with significant savings in time and cost for the patient?
First, there are methodologic concerns when a control group is not studied by means of a randomized, controlled trial—or even at the same center as the treatment arm—but is merely created from published data from a textbook.
Second, the goal of infertility therapy is live birth, not ovulation. Our studies have demonstrated significant differences in fecundity for each successful ovulation using different medications, suggesting that not every ovulation is the same.2 In the study by Hurst and colleagues, fecundity (live birth by ovulation rate) was 13%, compared with 10% using clomiphene in our large, multi-center trial—not much of an improvement, although, admittedly, the study by Hurst and colleagues was underpowered to address this question.2
Third, there are concerns about potential adverse effects of the accelerated clomiphene regimen on the patient or fetus. Clomiphene has a prolonged half-life of 5 to 7 days, with some metabolites persisting for months. What are the cumulative effects of such aggressive dosage escalation over such a short period of time?
The current Clomid package insert recommends a maximum dosage of 500 mg/cycle. A nonresponder in the stair-step protocol could consume 1,500 mg of clomiphene over a 20-day period.
Hurst and colleagues do not mention side effects, but it would be reasonable to expect an increased rate of hot flashes or mood changes. And what about more concerning signs such as visual changes?
Although clomiphene has no known human teratogenic effects (it is listed as pregnancy category “X”), the stair-step protocol could theoretically produce higher levels of fetal metabolites during the period of organ-ogenesis, with unknown effects.
Before we rush to adopt this accelerated regimen, we need studies that better address the maternal–fetal risk–benefit ratio. However, this study does provide evidence that a progestin withdrawal bleed is not mandatory in the nonresponder.—RICHARD S. LEGRO, MD
1. Thessaloniki ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Consensus on infertility treatment related to polycystic ovary syndrome. Fertil Steril. 2008;89:505-522.
2. Legro RS, Barnhart HX, Schlaff WD. Cooperative Multicenter Reproductive Medicine Network. Clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N Engl J Med. 2007;356:551-566.
1. Thessaloniki ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Consensus on infertility treatment related to polycystic ovary syndrome. Fertil Steril. 2008;89:505-522.
2. Legro RS, Barnhart HX, Schlaff WD. Cooperative Multicenter Reproductive Medicine Network. Clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N Engl J Med. 2007;356:551-566.
Guidelines confirm safety of pregnancy in women who have epilepsy—with caveats
Presence of seizures in pregnancy elevates risk of preterm birth
Women with epilepsy who have seizures during pregnancy appear more likely to give birth to preterm, small, or low-birth-weight babies than women without epilepsy, according to a report by Chen and colleagues in the August issue of Archives of Neurology.
Some previous studies had reported a link between adverse pregnancy outcomes and a mother’s epilepsy, but others found no association, the authors note.
“Our study further illuminates these conflicting data to suggest that it is the seizures themselves that seem to contribute greatly to the increased risk of infants being delivered preterm, of low birth weight and small for gestational age. For women who remained seizure-free throughout pregnancy, null or mild risk was identified, compared with unaffected women,” the authors write.
View the study at http://archneur.ama-assn.org/.
First-trimester exposure to the antiepileptic drug valproate increases the risk of major congenital malformation, particularly neural tube defects and facial clefts, according to recent guidelines developed by the American Academy of Neurology and the American Epilepsy Society.1-3 The guidelines recommend that women who have epilepsy avoid taking valproate during pregnancy.
“Good evidence shows that valproate is linked to an increased risk for fetal malformations and decreased thinking skills in children, whether used by itself or with other medications,” said lead guideline author Cynthia Harden, MD, director of the Epilepsy Division at the University of Miami’s Miller School of Medicine and member of the American Academy of Neurology.
The guidelines also suggest that, if at all possible, women who have epilepsy avoid taking more than one epilepsy drug at a time during pregnancy because the use of more than one antiseizure medication increases the risk of birth defects.
In addition, the guidelines recommend that physicians avoid prescribing the epilepsy drugs phenytoin and phenobarbital during pregnancy to lower the risk of diminished cognitive skills in children.
It is estimated that approximately 500,000 women of childbearing age in the United States have epilepsy, and that 3 to 5 of every 1,000 births are to women who have epilepsy. Most people who have epilepsy have well-controlled seizures and are otherwise healthy, said Harden.
Safe pregnancy is likely in women who have epilepsy
Aside from the risks associated with valproate, phenytoin, phenobarbital, and polytherapy, pregnancy in women who have epilepsy appears to be relatively safe.
“Overall, what we found should be very reassuring to every woman with epilepsy planning to become pregnant,” said Harden.
“These guidelines show that women with epilepsy are not at a substantially increased risk of having a cesarean section, late-pregnancy bleeding, or premature contractions or premature labor and delivery. Also, if a woman is seizure-free 9 months before she becomes pregnant, it’s likely that she will not have any seizures during the pregnancy.”
As a safeguard, measure blood levels of antiseizure drugs
Harden recommended that pregnant women who have epilepsy consider having their blood tested regularly.
“Levels of seizure medications in the blood tend to drop during pregnancy, so checking these levels and adjusting the medication doses should help to keep the levels in the effective range and the pregnant woman seizure-free.”
Guidelines cover range of issues
Here is a summary of the other main recommendations in the guidelines:
Avoid certain drugs; discourage smoking
- Besides avoiding valproate and antiepileptic drug polytherapy during the first trimester, women who have epilepsy should avoid these regimens throughout pregnancy to prevent adverse cognitive outcomes in the infant.
- Women who take antiepileptic drugs are probably at increased risk of a small-for-gestational-age baby and, possibly, delivering a newborn with an Apgar score below 7 at 1 minute.
- Women who have epilepsy and who smoke may increase the risk that they will develop premature contractions, premature labor, and premature delivery.
Monitor levels of some drugs
- Monitor levels of lamotrigine, carbamazepine, and phenytoin during pregnancy. Also monitor levels of levetiracetam and oxcarbazepine (a monohydroxy derivative). Blood levels of antiepileptic drugs tend to drop during pregnancy, and the dosage may need to be adjusted.
Seizure-free pregnancy is possible
- Counsel women who have epilepsy that remaining free from seizures for at least 9 months before pregnancy greatly increases the likelihood that they will remain seizure-free during pregnancy.
Folic acid may be beneficial
- Consider giving women who have epilepsy at least 0.4 mg of folic acid daily before they become pregnant, as it appears likely to lower the risk of major congenital malformation. It is unclear whether a higher daily dosage offers greater protective benefits.
Counsel the mother about breastfeeding concerns
- Women who have epilepsy and who choose to breastfeed should be counseled that primidone and levetiracetam probably pass into breast milk in significant amounts. In addition, gabapentin, lamotrigine, and topiramate may pass into breast milk in significant amounts. In contrast, valproate, phenobarbital, phenytoin, and carbamazepine probably do not pass into breast milk in clinically important amounts.
“For too long, women living with epilepsy have feared the added risk of premature birth and other consequences of both their epilepsy and their medications,” said Howard R. Soule, PhD, chief science officer for the Milken Family Foundation. “The results of this project will help relieve the worries of these women and their families.”
For more on the guidelines, visit the American Academy of Neurology Web site at: www.aan.com/index.cfm?axon=redirect&&path=/go/practice/guidelines.
1. Harden CL, Hopp J, Ting TY, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): I. Obstetrical complications and change in seizure frequency. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1229-1236.
2. Harden CL, Meador KJ, Pennell PB, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): II. Teratogenesis and perinatal outcomes. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1237-1246.
3. Harden CL, Pennell PB, Koppel BS, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): Vitamin K, folic acid, blood levels, and breast-feeding. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1247-1255.
Presence of seizures in pregnancy elevates risk of preterm birth
Women with epilepsy who have seizures during pregnancy appear more likely to give birth to preterm, small, or low-birth-weight babies than women without epilepsy, according to a report by Chen and colleagues in the August issue of Archives of Neurology.
Some previous studies had reported a link between adverse pregnancy outcomes and a mother’s epilepsy, but others found no association, the authors note.
“Our study further illuminates these conflicting data to suggest that it is the seizures themselves that seem to contribute greatly to the increased risk of infants being delivered preterm, of low birth weight and small for gestational age. For women who remained seizure-free throughout pregnancy, null or mild risk was identified, compared with unaffected women,” the authors write.
View the study at http://archneur.ama-assn.org/.
First-trimester exposure to the antiepileptic drug valproate increases the risk of major congenital malformation, particularly neural tube defects and facial clefts, according to recent guidelines developed by the American Academy of Neurology and the American Epilepsy Society.1-3 The guidelines recommend that women who have epilepsy avoid taking valproate during pregnancy.
“Good evidence shows that valproate is linked to an increased risk for fetal malformations and decreased thinking skills in children, whether used by itself or with other medications,” said lead guideline author Cynthia Harden, MD, director of the Epilepsy Division at the University of Miami’s Miller School of Medicine and member of the American Academy of Neurology.
The guidelines also suggest that, if at all possible, women who have epilepsy avoid taking more than one epilepsy drug at a time during pregnancy because the use of more than one antiseizure medication increases the risk of birth defects.
In addition, the guidelines recommend that physicians avoid prescribing the epilepsy drugs phenytoin and phenobarbital during pregnancy to lower the risk of diminished cognitive skills in children.
It is estimated that approximately 500,000 women of childbearing age in the United States have epilepsy, and that 3 to 5 of every 1,000 births are to women who have epilepsy. Most people who have epilepsy have well-controlled seizures and are otherwise healthy, said Harden.
Safe pregnancy is likely in women who have epilepsy
Aside from the risks associated with valproate, phenytoin, phenobarbital, and polytherapy, pregnancy in women who have epilepsy appears to be relatively safe.
“Overall, what we found should be very reassuring to every woman with epilepsy planning to become pregnant,” said Harden.
“These guidelines show that women with epilepsy are not at a substantially increased risk of having a cesarean section, late-pregnancy bleeding, or premature contractions or premature labor and delivery. Also, if a woman is seizure-free 9 months before she becomes pregnant, it’s likely that she will not have any seizures during the pregnancy.”
As a safeguard, measure blood levels of antiseizure drugs
Harden recommended that pregnant women who have epilepsy consider having their blood tested regularly.
“Levels of seizure medications in the blood tend to drop during pregnancy, so checking these levels and adjusting the medication doses should help to keep the levels in the effective range and the pregnant woman seizure-free.”
Guidelines cover range of issues
Here is a summary of the other main recommendations in the guidelines:
Avoid certain drugs; discourage smoking
- Besides avoiding valproate and antiepileptic drug polytherapy during the first trimester, women who have epilepsy should avoid these regimens throughout pregnancy to prevent adverse cognitive outcomes in the infant.
- Women who take antiepileptic drugs are probably at increased risk of a small-for-gestational-age baby and, possibly, delivering a newborn with an Apgar score below 7 at 1 minute.
- Women who have epilepsy and who smoke may increase the risk that they will develop premature contractions, premature labor, and premature delivery.
Monitor levels of some drugs
- Monitor levels of lamotrigine, carbamazepine, and phenytoin during pregnancy. Also monitor levels of levetiracetam and oxcarbazepine (a monohydroxy derivative). Blood levels of antiepileptic drugs tend to drop during pregnancy, and the dosage may need to be adjusted.
Seizure-free pregnancy is possible
- Counsel women who have epilepsy that remaining free from seizures for at least 9 months before pregnancy greatly increases the likelihood that they will remain seizure-free during pregnancy.
Folic acid may be beneficial
- Consider giving women who have epilepsy at least 0.4 mg of folic acid daily before they become pregnant, as it appears likely to lower the risk of major congenital malformation. It is unclear whether a higher daily dosage offers greater protective benefits.
Counsel the mother about breastfeeding concerns
- Women who have epilepsy and who choose to breastfeed should be counseled that primidone and levetiracetam probably pass into breast milk in significant amounts. In addition, gabapentin, lamotrigine, and topiramate may pass into breast milk in significant amounts. In contrast, valproate, phenobarbital, phenytoin, and carbamazepine probably do not pass into breast milk in clinically important amounts.
“For too long, women living with epilepsy have feared the added risk of premature birth and other consequences of both their epilepsy and their medications,” said Howard R. Soule, PhD, chief science officer for the Milken Family Foundation. “The results of this project will help relieve the worries of these women and their families.”
For more on the guidelines, visit the American Academy of Neurology Web site at: www.aan.com/index.cfm?axon=redirect&&path=/go/practice/guidelines.
Presence of seizures in pregnancy elevates risk of preterm birth
Women with epilepsy who have seizures during pregnancy appear more likely to give birth to preterm, small, or low-birth-weight babies than women without epilepsy, according to a report by Chen and colleagues in the August issue of Archives of Neurology.
Some previous studies had reported a link between adverse pregnancy outcomes and a mother’s epilepsy, but others found no association, the authors note.
“Our study further illuminates these conflicting data to suggest that it is the seizures themselves that seem to contribute greatly to the increased risk of infants being delivered preterm, of low birth weight and small for gestational age. For women who remained seizure-free throughout pregnancy, null or mild risk was identified, compared with unaffected women,” the authors write.
View the study at http://archneur.ama-assn.org/.
First-trimester exposure to the antiepileptic drug valproate increases the risk of major congenital malformation, particularly neural tube defects and facial clefts, according to recent guidelines developed by the American Academy of Neurology and the American Epilepsy Society.1-3 The guidelines recommend that women who have epilepsy avoid taking valproate during pregnancy.
“Good evidence shows that valproate is linked to an increased risk for fetal malformations and decreased thinking skills in children, whether used by itself or with other medications,” said lead guideline author Cynthia Harden, MD, director of the Epilepsy Division at the University of Miami’s Miller School of Medicine and member of the American Academy of Neurology.
The guidelines also suggest that, if at all possible, women who have epilepsy avoid taking more than one epilepsy drug at a time during pregnancy because the use of more than one antiseizure medication increases the risk of birth defects.
In addition, the guidelines recommend that physicians avoid prescribing the epilepsy drugs phenytoin and phenobarbital during pregnancy to lower the risk of diminished cognitive skills in children.
It is estimated that approximately 500,000 women of childbearing age in the United States have epilepsy, and that 3 to 5 of every 1,000 births are to women who have epilepsy. Most people who have epilepsy have well-controlled seizures and are otherwise healthy, said Harden.
Safe pregnancy is likely in women who have epilepsy
Aside from the risks associated with valproate, phenytoin, phenobarbital, and polytherapy, pregnancy in women who have epilepsy appears to be relatively safe.
“Overall, what we found should be very reassuring to every woman with epilepsy planning to become pregnant,” said Harden.
“These guidelines show that women with epilepsy are not at a substantially increased risk of having a cesarean section, late-pregnancy bleeding, or premature contractions or premature labor and delivery. Also, if a woman is seizure-free 9 months before she becomes pregnant, it’s likely that she will not have any seizures during the pregnancy.”
As a safeguard, measure blood levels of antiseizure drugs
Harden recommended that pregnant women who have epilepsy consider having their blood tested regularly.
“Levels of seizure medications in the blood tend to drop during pregnancy, so checking these levels and adjusting the medication doses should help to keep the levels in the effective range and the pregnant woman seizure-free.”
Guidelines cover range of issues
Here is a summary of the other main recommendations in the guidelines:
Avoid certain drugs; discourage smoking
- Besides avoiding valproate and antiepileptic drug polytherapy during the first trimester, women who have epilepsy should avoid these regimens throughout pregnancy to prevent adverse cognitive outcomes in the infant.
- Women who take antiepileptic drugs are probably at increased risk of a small-for-gestational-age baby and, possibly, delivering a newborn with an Apgar score below 7 at 1 minute.
- Women who have epilepsy and who smoke may increase the risk that they will develop premature contractions, premature labor, and premature delivery.
Monitor levels of some drugs
- Monitor levels of lamotrigine, carbamazepine, and phenytoin during pregnancy. Also monitor levels of levetiracetam and oxcarbazepine (a monohydroxy derivative). Blood levels of antiepileptic drugs tend to drop during pregnancy, and the dosage may need to be adjusted.
Seizure-free pregnancy is possible
- Counsel women who have epilepsy that remaining free from seizures for at least 9 months before pregnancy greatly increases the likelihood that they will remain seizure-free during pregnancy.
Folic acid may be beneficial
- Consider giving women who have epilepsy at least 0.4 mg of folic acid daily before they become pregnant, as it appears likely to lower the risk of major congenital malformation. It is unclear whether a higher daily dosage offers greater protective benefits.
Counsel the mother about breastfeeding concerns
- Women who have epilepsy and who choose to breastfeed should be counseled that primidone and levetiracetam probably pass into breast milk in significant amounts. In addition, gabapentin, lamotrigine, and topiramate may pass into breast milk in significant amounts. In contrast, valproate, phenobarbital, phenytoin, and carbamazepine probably do not pass into breast milk in clinically important amounts.
“For too long, women living with epilepsy have feared the added risk of premature birth and other consequences of both their epilepsy and their medications,” said Howard R. Soule, PhD, chief science officer for the Milken Family Foundation. “The results of this project will help relieve the worries of these women and their families.”
For more on the guidelines, visit the American Academy of Neurology Web site at: www.aan.com/index.cfm?axon=redirect&&path=/go/practice/guidelines.
1. Harden CL, Hopp J, Ting TY, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): I. Obstetrical complications and change in seizure frequency. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1229-1236.
2. Harden CL, Meador KJ, Pennell PB, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): II. Teratogenesis and perinatal outcomes. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1237-1246.
3. Harden CL, Pennell PB, Koppel BS, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): Vitamin K, folic acid, blood levels, and breast-feeding. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1247-1255.
1. Harden CL, Hopp J, Ting TY, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): I. Obstetrical complications and change in seizure frequency. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1229-1236.
2. Harden CL, Meador KJ, Pennell PB, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): II. Teratogenesis and perinatal outcomes. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1237-1246.
3. Harden CL, Pennell PB, Koppel BS, et al. Special report: Management issues for women with epilepsy—focus on pregnancy (evidence-based review): Vitamin K, folic acid, blood levels, and breast-feeding. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1247-1255.
IN THIS ARTICLE
Is the LNG–IUS as effective as endometrial ablation in relieving menorrhagia?
Historically, hysterectomy was the only alternative to medical treatment. However, endometrial ablation and the LNG-IUS have both proved to be effective, and less invasive, therapies.
Until recently, the average ObGyn in the United States placed only about four intrauterine devices (IUDs) a year. This low volume made many practitioners reluctant to prescribe the IUD, but we are seeing a resurgence in use.
Details of the study
This analysis, which meets Quality of Reporting of Meta-analyses (QUOROM) guidelines, was restricted to studies that included pre- and posttreatment assessment of menstrual blood loss using the Pictorial Blood Loss Assessment Chart (PBLAC).1 Although the PBLAC has limitations, it is one of the more practical, objective measures available. The PBLAC score does not yield an exact measure of blood loss, but it has been found to correlate well with menstrual blood volume. When it is used to evaluate menstrual blood loss of 80 mL or more, its specificity and sensitivity exceed 80%.
In reviewing randomized, controlled trials for this study, the authors found only a small number (n=6) that met their criteria, and those studies involved a relatively small number of patients (LNG-IUS, n=196; endometrial ablation, n=194), limiting the statistical power of this investigation.
Both treatment modalities were associated with a reduction in menstrual blood loss, and the degree of the reduction was similar between modalities at 6, 12, and 24 months.
The treatment failure rate was not time-adjusted; nor was the study powered to address the question of failure.
Only two studies assessed the PBLAC score at 6 months, five did so at 12 months, and only two did so at 24 months. The small number of women who had 24 months of follow-up limits the strength of the conclusion.
How this study compares with other investigations
As Kaunitz and colleagues note in their meta-analysis, studies comparing the LNG-IUS with endometrial ablation have produced conflicting findings about the reduction of menstrual blood loss: Some have found the modalities to be equally effective, others have found the LNG-IUS to be more effective, and still others have demonstrated higher efficacy for endometrial ablation. A recent Cochrane review reported that the LNG-IUS produced smaller mean reductions in menstrual blood loss than endometrial ablation did.2
When quality of life is the main consideration, data point to equivalence of options
The reduction of menstrual blood loss is only one area of focus in the treatment of menorrhagia, part of the larger goal of improving quality of life. Two Cochrane reviews concluded there is no difference between the LNG-IUS and endometrial ablation in regard to satisfaction rates or quality of life.2,3 Five studies reported quality of life scores; all five found them to be equivalent between modalities.
Although the data presented in this study are not definitive, the findings do support the growing body of data suggesting that these two treatments are, in some respects, equivalent options. At the same time, they are different procedures with distinct risks and considerations. When deciding between them, a patient’s desire for fertility may tip the scales in favor of the LNG-IUS.—MATTHEW R. HOPKINS, MD
1. Moher D, Cook DJ, Eastwood S, Olkin I, Rennie D, Stroup DF. Improving the quality of reports of meta-analyses of randomised controlled trials: the QUOROM statement. Quality of Reporting of Meta-analyses. Lancet. 1999;354:1896-1900.
2. Lethaby AE, Cooke I, Rees M. Progesterone on progestogen-releasing intrauterine systems for heavy menstrual bleeding. Cochrane Database Syst Rev. 2005;(4):CD002126.-
3. Marjoribanks J, Lethaby A, Farquhar C. Surgery versus medical therapy for heavy menstrual bleeding. Cochrane Database Syst Rev. 2003;(2):CD003855.-
Historically, hysterectomy was the only alternative to medical treatment. However, endometrial ablation and the LNG-IUS have both proved to be effective, and less invasive, therapies.
Until recently, the average ObGyn in the United States placed only about four intrauterine devices (IUDs) a year. This low volume made many practitioners reluctant to prescribe the IUD, but we are seeing a resurgence in use.
Details of the study
This analysis, which meets Quality of Reporting of Meta-analyses (QUOROM) guidelines, was restricted to studies that included pre- and posttreatment assessment of menstrual blood loss using the Pictorial Blood Loss Assessment Chart (PBLAC).1 Although the PBLAC has limitations, it is one of the more practical, objective measures available. The PBLAC score does not yield an exact measure of blood loss, but it has been found to correlate well with menstrual blood volume. When it is used to evaluate menstrual blood loss of 80 mL or more, its specificity and sensitivity exceed 80%.
In reviewing randomized, controlled trials for this study, the authors found only a small number (n=6) that met their criteria, and those studies involved a relatively small number of patients (LNG-IUS, n=196; endometrial ablation, n=194), limiting the statistical power of this investigation.
Both treatment modalities were associated with a reduction in menstrual blood loss, and the degree of the reduction was similar between modalities at 6, 12, and 24 months.
The treatment failure rate was not time-adjusted; nor was the study powered to address the question of failure.
Only two studies assessed the PBLAC score at 6 months, five did so at 12 months, and only two did so at 24 months. The small number of women who had 24 months of follow-up limits the strength of the conclusion.
How this study compares with other investigations
As Kaunitz and colleagues note in their meta-analysis, studies comparing the LNG-IUS with endometrial ablation have produced conflicting findings about the reduction of menstrual blood loss: Some have found the modalities to be equally effective, others have found the LNG-IUS to be more effective, and still others have demonstrated higher efficacy for endometrial ablation. A recent Cochrane review reported that the LNG-IUS produced smaller mean reductions in menstrual blood loss than endometrial ablation did.2
When quality of life is the main consideration, data point to equivalence of options
The reduction of menstrual blood loss is only one area of focus in the treatment of menorrhagia, part of the larger goal of improving quality of life. Two Cochrane reviews concluded there is no difference between the LNG-IUS and endometrial ablation in regard to satisfaction rates or quality of life.2,3 Five studies reported quality of life scores; all five found them to be equivalent between modalities.
Although the data presented in this study are not definitive, the findings do support the growing body of data suggesting that these two treatments are, in some respects, equivalent options. At the same time, they are different procedures with distinct risks and considerations. When deciding between them, a patient’s desire for fertility may tip the scales in favor of the LNG-IUS.—MATTHEW R. HOPKINS, MD
Historically, hysterectomy was the only alternative to medical treatment. However, endometrial ablation and the LNG-IUS have both proved to be effective, and less invasive, therapies.
Until recently, the average ObGyn in the United States placed only about four intrauterine devices (IUDs) a year. This low volume made many practitioners reluctant to prescribe the IUD, but we are seeing a resurgence in use.
Details of the study
This analysis, which meets Quality of Reporting of Meta-analyses (QUOROM) guidelines, was restricted to studies that included pre- and posttreatment assessment of menstrual blood loss using the Pictorial Blood Loss Assessment Chart (PBLAC).1 Although the PBLAC has limitations, it is one of the more practical, objective measures available. The PBLAC score does not yield an exact measure of blood loss, but it has been found to correlate well with menstrual blood volume. When it is used to evaluate menstrual blood loss of 80 mL or more, its specificity and sensitivity exceed 80%.
In reviewing randomized, controlled trials for this study, the authors found only a small number (n=6) that met their criteria, and those studies involved a relatively small number of patients (LNG-IUS, n=196; endometrial ablation, n=194), limiting the statistical power of this investigation.
Both treatment modalities were associated with a reduction in menstrual blood loss, and the degree of the reduction was similar between modalities at 6, 12, and 24 months.
The treatment failure rate was not time-adjusted; nor was the study powered to address the question of failure.
Only two studies assessed the PBLAC score at 6 months, five did so at 12 months, and only two did so at 24 months. The small number of women who had 24 months of follow-up limits the strength of the conclusion.
How this study compares with other investigations
As Kaunitz and colleagues note in their meta-analysis, studies comparing the LNG-IUS with endometrial ablation have produced conflicting findings about the reduction of menstrual blood loss: Some have found the modalities to be equally effective, others have found the LNG-IUS to be more effective, and still others have demonstrated higher efficacy for endometrial ablation. A recent Cochrane review reported that the LNG-IUS produced smaller mean reductions in menstrual blood loss than endometrial ablation did.2
When quality of life is the main consideration, data point to equivalence of options
The reduction of menstrual blood loss is only one area of focus in the treatment of menorrhagia, part of the larger goal of improving quality of life. Two Cochrane reviews concluded there is no difference between the LNG-IUS and endometrial ablation in regard to satisfaction rates or quality of life.2,3 Five studies reported quality of life scores; all five found them to be equivalent between modalities.
Although the data presented in this study are not definitive, the findings do support the growing body of data suggesting that these two treatments are, in some respects, equivalent options. At the same time, they are different procedures with distinct risks and considerations. When deciding between them, a patient’s desire for fertility may tip the scales in favor of the LNG-IUS.—MATTHEW R. HOPKINS, MD
1. Moher D, Cook DJ, Eastwood S, Olkin I, Rennie D, Stroup DF. Improving the quality of reports of meta-analyses of randomised controlled trials: the QUOROM statement. Quality of Reporting of Meta-analyses. Lancet. 1999;354:1896-1900.
2. Lethaby AE, Cooke I, Rees M. Progesterone on progestogen-releasing intrauterine systems for heavy menstrual bleeding. Cochrane Database Syst Rev. 2005;(4):CD002126.-
3. Marjoribanks J, Lethaby A, Farquhar C. Surgery versus medical therapy for heavy menstrual bleeding. Cochrane Database Syst Rev. 2003;(2):CD003855.-
1. Moher D, Cook DJ, Eastwood S, Olkin I, Rennie D, Stroup DF. Improving the quality of reports of meta-analyses of randomised controlled trials: the QUOROM statement. Quality of Reporting of Meta-analyses. Lancet. 1999;354:1896-1900.
2. Lethaby AE, Cooke I, Rees M. Progesterone on progestogen-releasing intrauterine systems for heavy menstrual bleeding. Cochrane Database Syst Rev. 2005;(4):CD002126.-
3. Marjoribanks J, Lethaby A, Farquhar C. Surgery versus medical therapy for heavy menstrual bleeding. Cochrane Database Syst Rev. 2003;(2):CD003855.-
Chronic pelvic pain: 11 critical questions about causes and care
Dr. Howard is a consultant to Ortho Women’s Health & Urology and a speaker for Abbott Pharmaceutical.
CASE: Multisystem involvement
makes diagnosis and treatment thorny
Sara B. is a 26-year-old gravida 4, para 3, abortus 1 who visits your office to be evaluated for chronic pelvic pain. She says her pain is most intense before and during her period and with intercourse. It is located primarily in her abdominopelvic area, but radiates to her lower extremities and lumbosacral back. It appears to be related to bowel function and meets Rome II criteria for irritable bowel syndrome (criteria developed by a panel of experts convened by the Rome Foundation).
Sara B. reports that she voids at least 20 times a day and once during the night. She has a history of depression, for which she takes sertraline (Zoloft), but no history of physical or sexual abuse. When she underwent laparoscopy more than 1 year ago, endometriosis was diagnosed visually.
Upon physical examination, you identify 13 positive fibromyalgia points, moderate tenderness of the posterior levator ani muscles, severe tenderness of the bladder, and moderate tenderness of the uterine fundus. You also find moderate tenderness in the adnexa and uterosacral ligaments bilaterally. Your tentative diagnosis: endometriosis, interstitial cystitis, fibromyalgia, and irritable bowel syndrome.
How do you confirm the diagnosis? And what treatment should you offer to her?
Chronic pelvic pain (CPP) is anything but simple. Sara B.’s case illustrates some of the complexity involved in the diagnostic evaluation and treatment of this disorder. Very rarely is the pain localized to one organ or system. More commonly, it involves multiple organs or anatomic areas within the pelvic region.
To confirm the diagnosis in Sara’s case, the next step would be a potassium chloride sensitivity test for interstitial cystitis. I would also start her on desipramine for fibromyalgia, and perform laparoscopy and cystoscopy with hydrodistention to explore the diagnosis further.
In Sara’s case, let’s assume that the repeat laparoscopy reveals glomerulations of the bladder but no recurrent endometriosis. I would administer oral pentosan polysulfate sodium and instill heparin and lidocaine in her bladder to improve her voiding pattern significantly (to the range of four to six times a day without nocturia). I would also prescribe continuous oral contraceptives to suppress her menses and alleviate some of her pain. In addition, I would be interested to see what a transjugular pelvic venogram would reveal. If it were to suggest severe pelvic congestion syndrome, I might perform embolization of both ovarian veins to provide additional relief.
Clearly, when confronted with a case as intricate as Sara’s, there are many ways to organize your thinking about the potential diagnoses that may cause or contribute to CPP. This article focuses on anatomic and mechanistic bases for evaluation of this disorder as a means of tailoring treatment appropriately. It explores these topics by addressing 11 critical questions, ranging from how pain is described to what to do about it.
1. How is pain defined?
Pain is an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage.1
Pain is defined in this way to make it clear that it is not just a sensory experience, but both a sensory and emotional experience. This means that the pain is always subjective and is not the same in all individuals—nor does it remain the same in the same person.
Individuals base their descriptions of pain on their unique prior experience of it. Many people report pain in the absence of tissue damage or any likely pathophysiologic cause, often for psychological reasons. If they regard their experience as pain and report it as they would pain caused by tissue damage, it should be accepted as pain. In defining pain, it is best to deliberately avoid tying pain to the stimulus.
What about CPP? There is no generally accepted definition. The American College of Obstetricians and Gynecologists (ACOG) defines it as noncyclic pain of at least 6 months’ duration that localizes to the anatomic pelvis, lumbosacral back, buttocks, or anterior abdominal wall at or below the umbilicus and that is severe enough to cause functional disability or lead to medical care.2
2. How many women suffer chronic pelvic pain?
Chronic pelvic pain is more common than is generally recognized. Here are some estimates:
- A US study conducted by the Gallup organization found that 15% of women 18 to 50 years old had CPP3
- A survey of women in family medicine and ObGyn offices found that 39% had CPP, although only 8% reported having it more often than “sometimes”4
- The Oxfordshire Women’s Health Study, a postal questionnaire survey of a random sample of women 18 to 49 years old in the general UK population, found a prevalence of 24%5
- A primary-care database in a UK study of women 15 to 73 years old found a prevalence of 38 cases for every 1,000 women. (The database contained annualized data that excluded women who had only dysmenorrhea or dyspareunia.) Although the study likely underestimated the prevalence of CPP, the finding does make it possible to compare prevalences in the same population: asthma (37/1,000), back pain (41/1,000), and migraine (21/1,000).6
3. What are the main types of pain involved?
They are nociceptive, inflammatory, and neuropathic pain.
Nociceptive pain occurs in response to a noxious stimulus that alerts the organism to impending tissue injury. One way to think of nociceptive pain is as “normal” or physiologic pain (FIGURE).
Acute pelvic pain is usually nociceptive in origin. CPP is usually not solely nociceptive in origin. It often involves inflammatory or neuropathic pain, or both (TABLE 1).
Inflammatory pain arises in response to tissue injury and the resulting inflammatory process. In some cases, the inflammatory response is actually a source of tissue injury (e.g., rheumatoid arthritis). Inflammatory pain may be an important mechanism in both acute and chronic pelvic pain.
Neuropathic pain is produced by damage to or dysfunction of neurons in the peripheral or central nervous system. It is not physiologic and is often a significant mechanism in the generation of CPP.
An understanding of inflammatory and neuropathic mechanisms is not esoteric, but has clinical significance.
FIGURE Noxious stimulus is often the trigger in acute and chronic pain
Nociceptive pain is a response to a noxious stimulus, alerting the organism to impending tissue injury. Four fundamental processes are involved in nociceptive pain: transduction, in which the stimulus is converted to a biochemical signal; transmission, in which the signal is transported from the peripheral nervous system to the dorsal ganglion and central nervous system; modulation, in which the intensity of the signal is increased or decreased; and perception, in which the organism experiences the pain.TABLE 1
Anatomic and mechanistic classification of pain
Central
|
Peripheral
|
4. Is chronic pelvic pain a disease—or a symptom?
My experience caring for patients who have CPP suggests that chronic pain is a disease, whereas acute pain is a symptom. This concept is controversial in gynecology, and CPP is often labeled as only a symptom, not a diagnosis.7 The search for one underlying disease means that the woman who has CPP frequently undergoes multiple surgical and other invasive procedures, often with incomplete or insignificant diagnoses and responses.
The assumption that CPP is always due to a specific pathologic process in somatic structures or viscera (nociceptive pain) excludes the possibility that CPP can be caused by prolonged or permanent dysfunction of the peripheral or central nervous system, or both (neuropathic pain), or by psychological mechanisms (central pain). Clinical knowledge lags behind basic science in this area and is not at all concrete.
Our ability to accurately diagnose neuropathic or inflammatory pain leaves room for improvement.
5. Is chronic pelvic pain a gynecologic disorder?
Gynecologists have traditionally thought of CPP as either gynecologic or nongynecologic in origin, but this framework has very limited clinical utility. An anatomic and mechanistic classification (TABLE 1) represents a far richer strategic approach to the diagnostic evaluation of CPP, allowing more comprehensive and effective treatment.
6. What distinguishes visceral from somatic pain?
In addition to recognizing the importance of nociceptive, inflammatory, and neuropathic mechanisms in the generation of CPP, it is useful to classify potential causes anatomically (TABLE 1). In the broadest anatomic categories, pain may be central or peripheral, or both. Central pain can be psychogenic or neurogenic, and peripheral pain can be visceral or somatic.
Visceral sources of CPP include the reproductive, genitourinary, and gastrointestinal (GI) tracts. Mechanistically, as has been discussed, visceral pain can be neuropathic, inflammatory, or nociceptive.
Potential somatic sources of CPP are myofascial, skeletal, and cutaneous. Mechanisms leading to somatic CPP can be neuropathic, inflammatory, or nociceptive.
Somatic pain is better understood than visceral pain, but knowledge about the latter has been expanding rapidly. Several characteristics distinguish visceral pain:
- Not all viscera generate pain, possibly owing to a lack of sensory receptors or appropriate nociceptive stimulus
- Visceral pain is not always linked to injury and, therefore, may be functional
- Visceral pain frequently results in somatic referral of pain, possibly due to central convergence of visceral and somatic afferents
- Visceral pain tends to be diffuse or poorly localized, probably because of the low concentration of nociceptive afferents within viscera (only 2% to 10% of total afferents to the spinal cord originate from visceral nociceptors).8
It is not clear whether there are visceral neurons dedicated solely to nociception; it appears that viscera utilize sympathetic and parasympathetic neurons as nociceptors. It also is important to note that the stimuli that activate somatic nociceptors—cutting, crushing, and burning, for example—do not generally cause visceral pain. Visceral nociceptive pain is generated in response to:
- distention of a viscous or organ capsule
- spasm of visceral muscular fibers
- ischemia from vascular disturbances
- hemorrhage
- neoplasm
- inflammation
- traction on mesentery.
Another characteristic that distinguishes visceral from somatic nociception: Visceral nociception utilizes a dorsal midline pathway within the central nervous system, in addition to the lateral spinothalamic tract pathway utilized by somatic nociception.
Although this anatomic and mechanistic classification is clinically useful in the diagnostic evaluation of CPP, it is an oversimplification. Most patients—like Sara B., described in the opening case—have multiple anatomic and mechanistic causes of their pain.
7. What are the primary visceral causes of chronic pelvic pain?
A limited number of visceral and somatic diagnoses are backed by level-A evidence as having a causal relationship with CPP (TABLE 2). A few are discussed here.
TABLE 2
Disorders that may cause CPP or make it worse*
Reproductive tract
|
Urinary tract
|
Gastrointestinal tract
|
Musculoskeletal system
|
| Depression |
| Somatization disorder |
| * Disorders with Level-A evidence, i.e., good and consistent scientific evidence of a causal relationship to CPP |
Disorders of the reproductive tract
Endometriosis is the most common gynecologic diagnosis in women who have CPP. There is significant epidemiologic evidence that endometriosis causes CPP. There is also strong evidence that endometriosis is a risk factor for CPP.
For example, human and animal experimental data suggest that women who have endometriosis have more episodes of urinary calculosis—and more severe pain—than women who do not have endometriosis.9,10 They also are more likely to report vaginal pain than are women who do not have endometriosis, and that vaginal pain is more likely to be severe.
Such viscerovisceral interactions may play a significant role in CPP in women and may explain why some women who have a history of endometriosis have persistent pelvic pain after their endometriosis is gone, or even appear to develop other pain syndromes, such as interstitial cystitis.11
Our introductory case illustrates these concepts. Sara B.’s history is classic for endometriosis-associated pelvic pain; that was her original diagnosis. Although her pelvic pain recurred and persisted, a repeat laparoscopy found no endometriosis—but it did reveal evidence of interstitial cystitis and painful bladder syndrome (IC/PBS). Could Sara’s current pain be neuropathic or inflammatory?
Treatment of IC/PBS targets neuropathic and inflammatory pain mechanisms, but this approach has not been fully explored for endometriosis. Might the visceral pain mechanisms be as important as the end-organ diagnoses? Clearly, this area merits further attention in gynecology.
Pelvic inflammatory disease (PID) often causes CPP. Approximately 18% to 35% of all women who have acute PID develop CPP.12,13 The actual mechanisms by which CPP results from PID are not known, but it seems likely that both inflammatory and neuropathic mechanisms are important. Adhesive disease secondary to PID may also contribute to CPP by generating nociceptive pain. The route of treatment of PID—parenteral or oral antibiotics—does not appear to affect the odds of developing CPP.
Pelvic congestion syndrome is a controversial diagnosis that is uncommon in the United States. However, a well-designed study from Turkey suggests that about 40% of women who consult a gynecologist about CPP may have this syndrome.14 In Sara’s case, pelvic congestion syndrome was diagnosed after venous dilation and delayed emptying were confirmed by selective ovarian venography (transcervical venography is another option).15 This approach is recommended.
According to the data, the most effective treatment of pelvic congestion syndrome is a high-dose progestin or gonadotropin-releasing hormone (GnRH) agonist.14,16 Only observational data back treatment with ovarian venous embolization, which was performed in Sara’s case.8
Urinary tract disorders
Interstitial cystitis/painful bladder syndrome is the most common urologic diagnosis among women who have CPP. Recent evidence suggests that 38% to 81% of women who are given a diagnosis of a reproductive-tract disorder may in fact have IC/PBS.17,18 Much of the recent evidence regarding interstitial cystitis suggests that inflammatory and neuropathic mechanisms are crucial in the generation of CPP; therefore, much of the treatment focuses on inflammatory and neuropathic pain.19,20
For example, among the treatments that alleviate IC/PBS to some degree are:
- amitriptyline, widely used for neuropathic pain21
- gapabentin, an anticonvulsant used to treat neuropathic pain22
- antihistamines directed at inflammation23
- intravesical instillation of a local anesthetic agent, which may target both inflammatory and neuropathic pain mechanisms.24
Although these therapies have not been widely studied for their efficacy in gynecologic disorders, they are likely to produce similar results.
Disorders of the GI tract
Irritable bowel syndrome is the most common GI diagnosis in women who have CPP. It is a clinical diagnosis, usually based on the Rome III criteria (TABLE 3). (Sara B. was evaluated when Rome II criteria were in use.)
Data from a primary-care database in the United Kingdom suggest that irritable bowel syndrome may be the most common diagnosis in women who have CPP (about 38% of patients).25 In some cases, irritable bowel syndrome presents primarily with lower abdominal or pelvic pain, so it must be considered in the differential diagnosis of CPP. It seems likely that the pain in irritable bowel syndrome is not simply nociceptive, but that inflammatory and nociceptive mechanisms play an important role, as well.26,27
TABLE 3
Rome III criteria for irritable bowel syndrome
| Two or more criteria must be present to make the diagnosis. |
Over the past 3 months, have you had at least 3 days when you have had abdominal pain or discomfort that:
|
8. What are the main somatic causes of chronic pelvic pain?
Abdominal wall myofascial pain syndrome
When there are trigger points and myofascial pain of the lower abdominal wall muscles or pelvic floor muscles, they often present as CPP.
The underlying mechanisms responsible for myofascial pain syndrome are not clear. Nociceptive pain seems to be an important mechanism, but it is not clear whether inflammatory and neuropathic changes occur in some patients with this syndrome.
Many women who have myofascial pain syndrome and CPP respond poorly to traditional treatment with physical therapy and trigger-point injections; this may be due to inflammatory or neuropathic changes, or both.
Pelvic floor tension myalgia
Pain due to abnormal tension of the pelvic floor muscles is well-described. In many cases, pelvic floor tension myalgia is a secondary phenomenon, as pelvic floor muscles react to the persistent presence of pelvic pain, which often has a visceral basis. In other cases, pelvic floor tension myalgia is a primary phenomenon and most likely represents myofascial pain syndrome of one or more of the pelvic floor muscles.
9. Are multiple anatomic sites and mechanisms the “norm”?
They may not be the norm, but it is not unusual to discover multiple diagnoses when evaluating a patient for CPP. Most published studies of women from primary-care practices suggest that 25% to 50% of patients have more than one diagnosis,5,25,28 and anecdotal experience from referral practices suggests that most women in such practices have more than one diagnosis. The most common diagnoses in most published series are endometriosis, adhesions, irritable bowel syndrome, and interstitial cystitis.18,29-31 The absence of somatic diagnoses in these series probably reflects the gynecologist’s tendency to concentrate on visceral elements in CPP.
10. When multiple systems are involved, is the pain greater?
Yes. Women who have more than one organ system involved in CPP have greater pain than women who have only one system involved. For example, 43% of patients who have CPP without GI or urologic symptoms had moderate or severe pain (mean visual analog score of 3.8), whereas 71% of women who had CPP and both GI and urologic symptoms had moderate to severe pain (mean visual analog scale score of 5.4).28
Pain is also more consistent in women who have multisystem symptoms. Women who have CPP are more likely than the general population to have dysmenorrhea (81% versus 58%) and dyspareunia (41% versus 14%). The severity of pain with intercourse and with menses is greater in women who have CPP and GI and urologic symptoms than in those who have no GI and urologic symptoms.
11. How is treatment affected by multiple diagnoses?
The presence of multiple diagnoses often reflects neuropathic changes and neuropathic pain. An accurate diagnosis of all pain generators, including neuropathic pain, seems vital to improving our management and treatment of women who have CPP.
For example, in Sara B.’s case, I prescribed norethindrone acetate to suppress menses, based on her history of endometriosis-associated pelvic pain and menstrual exacerbation of her symptoms. I prescribed oral pentosan polysulfate sodium and intravesical lidocaine and heparin for interstitial cystitis/painful bladder syndrome. And I gave Sara amitriptyline for both fibromyalgia and interstitial cystitis/painful bladder syndrome (as well as suspected neuropathic pain).
I also recommended a low-fat, high-fiber diet to help alleviate her irritable bowel syndrome.
1. International Association for the Study of Pain. IASP Pain Terminology. Available at: http://www.iasp-pain.org/AM/Template.cfm?Section=Pain_Definitions&Template=/CM/HTMLDisplay.cfm&ContentID=1728. Accessed July 10, 2009.
2. Chronic pelvic pain. ACOG Practice Bulletin No. 51. Obstet. Gynecol. 2004;103:589-605.
3. Mathias SD, Kuppermann M, Liberman RF, Lipschutz RC, Steege JF. Chronic pelvic pain: prevalence, health-related quality of life, and economic correlates. Obstet Gynecol. 1996;87:321-327.
4. Jamieson DJ, Steege JF. The prevalence of dysmenorrhea, dyspareunia, pelvic pain, and irritable bowel syndrome in primary care practices. Obstet Gynecol. 1996;87:55-58.
5. Zondervan KT, Yudkin PL, Vessey MP, et al. The community prevalence of chronic pelvic pain in women and associated illness behaviour. Br J Gen Pract. 2001;51:541-547.
6. Zondervan KT, Yudkin PL, Vessey MP, Dawes MG, Barlow DH, Kennedy SH. Prevalence and incidence of chronic pelvic pain in primary care: evidence from a national general practice database. Br J Obstet Gynaecol. 1999;106:1149-1155.
7. Scialli AR, Barbieri RL, Glasser MH, Olive DL, Winkel CA. Chronic Pelvic Pain: An Integrated Approach. APGO Education Series on Women’s Health Issues. Washington, DC: Association of Professors of Gynecology and Obstetrics; 2000.
8. Maleux G, Stockx L, Wilms G, Marchal G. Ovarian vein embolization for the treatment of pelvic congestion syndrome: long-term technical and clinical results. J Vasc Interv Radiol. 2000;11:859-864.
9. Giamberardino MA, De Laurentis S, Affaitati G, Lerza R, Lapenna D, Vecchiet L. Modulation of pain and hyperalgesia from the urinary tract by algogenic conditions of the reproductive organs in women. Neurosci Lett. 2001;304:61-64.
10. Giamberardino MA, Berkley KJ, Affaitati G, et al. Influence of endometriosis on pain behaviors and muscle hyperalgesia induced by a ureteral calculosis in female rats. Pain. 2002;95:247-257.
11. Parsons CL, Bullen M, Kahn BS, Stanford EJ, Willems JJ. Gynecologic presentation of interstitial cystitis as detected by intravesical potassium sensitivity. Obstet Gynecol. 2001;98:127-132.
12. Weström L. Effect of acute pelvic inflammatory disease on fertility. Am J Obstet Gynecol. 1975;121:707-713.
13. Ness RB, Soper DE, Holley RL, et al. Effectiveness of inpatient and outpatient treatment strategies for women with pelvic inflammatory disease: results from the Pelvic Inflammatory Disease Evaluation and Clinical Health (PEACH) Randomized Trial. Am J Obstet Gynecol. 2002;186:929-937.
14. Soysal ME, Soysal S, Vicdan K, Ozer S. A randomized controlled trial of goserelin and medroxyprogesterone acetate in the treatment of pelvic congestion. Hum Reprod. 2001;16:931-939.
15. Beard RW, Highman JH, Pearce S, Reginald PW. Diagnosis of pelvic varicosities in women with chronic pelvic pain. Lancet. 1984;2:946-949.
16. Farquhar CM, Rogers V, Franks S, Pearce S, Wadsworth J, Beard RW. A randomized controlled trial of medroxyprogesterone acetate and psychotherapy for the treatment of pelvic congestion. Br J Obstet Gynaecol. 1989;96:1153-1162.
17. Clemons JL, Arya LA, Myers DL. Diagnosing interstitial cystitis in women with chronic pelvic pain. Obstet Gynecol. 2002;100:337-341.
18. Parsons CL, Dell J, Stanford EJ, Bullen M, Kahn BS, Willems JJ. The prevalence of interstitial cystitis in gynecologic patients with pelvic pain, as detected by intravesical potassium sensitivity. Am J Obstet Gynecol. 2002;187:1395-1400.
19. Wesselmann U. Interstitial cystitis: a chronic visceral pain syndrome. Urology. 2001;57:32-39.
20. Butrick CW. Interstitial cystitis and chronic pelvic pain: new insights in neuropathology, diagnosis, and treatment. Clin Obstet Gynecol. 2003;46:811-823.
21. van Ophoven A, Pokupic S, Heinecke A, Hertle L. A prospective, randomized, placebo controlled, double-blind study of amitriptyline for the treatment of interstitial cystitis. J Urol. 2004;172:533-536.
22. Sasaki K, Smith CP, Chuang YC, Lee JY, Kim JC, Chancellor MB. Oral gabapentin (Neurontin) treatment of refractory genitourinary tract pain. Tech Urol. 2001;7:47-49.
23. Sant GR, Propert KJ, Hanno PM, et al. Interstitial Cystitis Clinical Trials Group. A pilot clinical trial of oral pentosan polysulfate and oral hydroxyzine in patients with interstitial cystitis. J Urol. 2003;170:810-815.
24. Parsons CL. Successful downregulation of bladder sensory nerves with combination of heparin and alkalinized lidocaine in patients with interstitial cystitis. Urology. 2005;65:45-48.
25. Zondervan KT, Yudkin PL, Vessey MP, Dawes MG, Barlow DH, Kennedy SH. Patterns of diagnosis and referral in women consulting for chronic pelvic pain in UK primary care. Br J Obstet Gynaecol. 1999;106:1156-1161.
26. Kwan CL, Diamant NE, Mikula K, Davis KD. Characteristics of rectal perception are altered in irritable bowel syndrome. Pain. 2005;113:160-171.
27. Wilder-Smith CH, Schindler D, Lovblad K, Redmond SM, Nirkko A. Brain functional magnetic resonance imaging of rectal pain and activation of endogenous inhibitory mechanisms in irritable bowel syndrome patient subgroups and healthy controls. Gut. 2004;53:1595-1601.
28. Zondervan KT, Yudkin PL, Vessey MP, et al. Chronic pelvic pain in the community—symptoms, investigations, and diagnoses. Am J Obstet Gynecol. 2001;184:1149-1155.
29. Williams RE, Hartmann KE, Sandler RS, Miller WC, Steege JF. Prevalence and characteristics of irritable bowel syndrome among women with chronic pelvic pain. Obstet Gynecol. 2004;104:452-458.
30. Howard FM. The role of laparoscopy in chronic pelvic pain: promise and pitfalls. Obstet Gynecol Surv. 1993;48:357-387.
31. Chung MK, Chung RR, Gordon D, Jennings C. The evil twins of chronic pelvic pain syndrome: endometriosis and interstitial cystitis. JSLS. 2002;6:311-314.
Dr. Howard is a consultant to Ortho Women’s Health & Urology and a speaker for Abbott Pharmaceutical.
CASE: Multisystem involvement
makes diagnosis and treatment thorny
Sara B. is a 26-year-old gravida 4, para 3, abortus 1 who visits your office to be evaluated for chronic pelvic pain. She says her pain is most intense before and during her period and with intercourse. It is located primarily in her abdominopelvic area, but radiates to her lower extremities and lumbosacral back. It appears to be related to bowel function and meets Rome II criteria for irritable bowel syndrome (criteria developed by a panel of experts convened by the Rome Foundation).
Sara B. reports that she voids at least 20 times a day and once during the night. She has a history of depression, for which she takes sertraline (Zoloft), but no history of physical or sexual abuse. When she underwent laparoscopy more than 1 year ago, endometriosis was diagnosed visually.
Upon physical examination, you identify 13 positive fibromyalgia points, moderate tenderness of the posterior levator ani muscles, severe tenderness of the bladder, and moderate tenderness of the uterine fundus. You also find moderate tenderness in the adnexa and uterosacral ligaments bilaterally. Your tentative diagnosis: endometriosis, interstitial cystitis, fibromyalgia, and irritable bowel syndrome.
How do you confirm the diagnosis? And what treatment should you offer to her?
Chronic pelvic pain (CPP) is anything but simple. Sara B.’s case illustrates some of the complexity involved in the diagnostic evaluation and treatment of this disorder. Very rarely is the pain localized to one organ or system. More commonly, it involves multiple organs or anatomic areas within the pelvic region.
To confirm the diagnosis in Sara’s case, the next step would be a potassium chloride sensitivity test for interstitial cystitis. I would also start her on desipramine for fibromyalgia, and perform laparoscopy and cystoscopy with hydrodistention to explore the diagnosis further.
In Sara’s case, let’s assume that the repeat laparoscopy reveals glomerulations of the bladder but no recurrent endometriosis. I would administer oral pentosan polysulfate sodium and instill heparin and lidocaine in her bladder to improve her voiding pattern significantly (to the range of four to six times a day without nocturia). I would also prescribe continuous oral contraceptives to suppress her menses and alleviate some of her pain. In addition, I would be interested to see what a transjugular pelvic venogram would reveal. If it were to suggest severe pelvic congestion syndrome, I might perform embolization of both ovarian veins to provide additional relief.
Clearly, when confronted with a case as intricate as Sara’s, there are many ways to organize your thinking about the potential diagnoses that may cause or contribute to CPP. This article focuses on anatomic and mechanistic bases for evaluation of this disorder as a means of tailoring treatment appropriately. It explores these topics by addressing 11 critical questions, ranging from how pain is described to what to do about it.
1. How is pain defined?
Pain is an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage.1
Pain is defined in this way to make it clear that it is not just a sensory experience, but both a sensory and emotional experience. This means that the pain is always subjective and is not the same in all individuals—nor does it remain the same in the same person.
Individuals base their descriptions of pain on their unique prior experience of it. Many people report pain in the absence of tissue damage or any likely pathophysiologic cause, often for psychological reasons. If they regard their experience as pain and report it as they would pain caused by tissue damage, it should be accepted as pain. In defining pain, it is best to deliberately avoid tying pain to the stimulus.
What about CPP? There is no generally accepted definition. The American College of Obstetricians and Gynecologists (ACOG) defines it as noncyclic pain of at least 6 months’ duration that localizes to the anatomic pelvis, lumbosacral back, buttocks, or anterior abdominal wall at or below the umbilicus and that is severe enough to cause functional disability or lead to medical care.2
2. How many women suffer chronic pelvic pain?
Chronic pelvic pain is more common than is generally recognized. Here are some estimates:
- A US study conducted by the Gallup organization found that 15% of women 18 to 50 years old had CPP3
- A survey of women in family medicine and ObGyn offices found that 39% had CPP, although only 8% reported having it more often than “sometimes”4
- The Oxfordshire Women’s Health Study, a postal questionnaire survey of a random sample of women 18 to 49 years old in the general UK population, found a prevalence of 24%5
- A primary-care database in a UK study of women 15 to 73 years old found a prevalence of 38 cases for every 1,000 women. (The database contained annualized data that excluded women who had only dysmenorrhea or dyspareunia.) Although the study likely underestimated the prevalence of CPP, the finding does make it possible to compare prevalences in the same population: asthma (37/1,000), back pain (41/1,000), and migraine (21/1,000).6
3. What are the main types of pain involved?
They are nociceptive, inflammatory, and neuropathic pain.
Nociceptive pain occurs in response to a noxious stimulus that alerts the organism to impending tissue injury. One way to think of nociceptive pain is as “normal” or physiologic pain (FIGURE).
Acute pelvic pain is usually nociceptive in origin. CPP is usually not solely nociceptive in origin. It often involves inflammatory or neuropathic pain, or both (TABLE 1).
Inflammatory pain arises in response to tissue injury and the resulting inflammatory process. In some cases, the inflammatory response is actually a source of tissue injury (e.g., rheumatoid arthritis). Inflammatory pain may be an important mechanism in both acute and chronic pelvic pain.
Neuropathic pain is produced by damage to or dysfunction of neurons in the peripheral or central nervous system. It is not physiologic and is often a significant mechanism in the generation of CPP.
An understanding of inflammatory and neuropathic mechanisms is not esoteric, but has clinical significance.
FIGURE Noxious stimulus is often the trigger in acute and chronic pain
Nociceptive pain is a response to a noxious stimulus, alerting the organism to impending tissue injury. Four fundamental processes are involved in nociceptive pain: transduction, in which the stimulus is converted to a biochemical signal; transmission, in which the signal is transported from the peripheral nervous system to the dorsal ganglion and central nervous system; modulation, in which the intensity of the signal is increased or decreased; and perception, in which the organism experiences the pain.TABLE 1
Anatomic and mechanistic classification of pain
Central
|
Peripheral
|
4. Is chronic pelvic pain a disease—or a symptom?
My experience caring for patients who have CPP suggests that chronic pain is a disease, whereas acute pain is a symptom. This concept is controversial in gynecology, and CPP is often labeled as only a symptom, not a diagnosis.7 The search for one underlying disease means that the woman who has CPP frequently undergoes multiple surgical and other invasive procedures, often with incomplete or insignificant diagnoses and responses.
The assumption that CPP is always due to a specific pathologic process in somatic structures or viscera (nociceptive pain) excludes the possibility that CPP can be caused by prolonged or permanent dysfunction of the peripheral or central nervous system, or both (neuropathic pain), or by psychological mechanisms (central pain). Clinical knowledge lags behind basic science in this area and is not at all concrete.
Our ability to accurately diagnose neuropathic or inflammatory pain leaves room for improvement.
5. Is chronic pelvic pain a gynecologic disorder?
Gynecologists have traditionally thought of CPP as either gynecologic or nongynecologic in origin, but this framework has very limited clinical utility. An anatomic and mechanistic classification (TABLE 1) represents a far richer strategic approach to the diagnostic evaluation of CPP, allowing more comprehensive and effective treatment.
6. What distinguishes visceral from somatic pain?
In addition to recognizing the importance of nociceptive, inflammatory, and neuropathic mechanisms in the generation of CPP, it is useful to classify potential causes anatomically (TABLE 1). In the broadest anatomic categories, pain may be central or peripheral, or both. Central pain can be psychogenic or neurogenic, and peripheral pain can be visceral or somatic.
Visceral sources of CPP include the reproductive, genitourinary, and gastrointestinal (GI) tracts. Mechanistically, as has been discussed, visceral pain can be neuropathic, inflammatory, or nociceptive.
Potential somatic sources of CPP are myofascial, skeletal, and cutaneous. Mechanisms leading to somatic CPP can be neuropathic, inflammatory, or nociceptive.
Somatic pain is better understood than visceral pain, but knowledge about the latter has been expanding rapidly. Several characteristics distinguish visceral pain:
- Not all viscera generate pain, possibly owing to a lack of sensory receptors or appropriate nociceptive stimulus
- Visceral pain is not always linked to injury and, therefore, may be functional
- Visceral pain frequently results in somatic referral of pain, possibly due to central convergence of visceral and somatic afferents
- Visceral pain tends to be diffuse or poorly localized, probably because of the low concentration of nociceptive afferents within viscera (only 2% to 10% of total afferents to the spinal cord originate from visceral nociceptors).8
It is not clear whether there are visceral neurons dedicated solely to nociception; it appears that viscera utilize sympathetic and parasympathetic neurons as nociceptors. It also is important to note that the stimuli that activate somatic nociceptors—cutting, crushing, and burning, for example—do not generally cause visceral pain. Visceral nociceptive pain is generated in response to:
- distention of a viscous or organ capsule
- spasm of visceral muscular fibers
- ischemia from vascular disturbances
- hemorrhage
- neoplasm
- inflammation
- traction on mesentery.
Another characteristic that distinguishes visceral from somatic nociception: Visceral nociception utilizes a dorsal midline pathway within the central nervous system, in addition to the lateral spinothalamic tract pathway utilized by somatic nociception.
Although this anatomic and mechanistic classification is clinically useful in the diagnostic evaluation of CPP, it is an oversimplification. Most patients—like Sara B., described in the opening case—have multiple anatomic and mechanistic causes of their pain.
7. What are the primary visceral causes of chronic pelvic pain?
A limited number of visceral and somatic diagnoses are backed by level-A evidence as having a causal relationship with CPP (TABLE 2). A few are discussed here.
TABLE 2
Disorders that may cause CPP or make it worse*
Reproductive tract
|
Urinary tract
|
Gastrointestinal tract
|
Musculoskeletal system
|
| Depression |
| Somatization disorder |
| * Disorders with Level-A evidence, i.e., good and consistent scientific evidence of a causal relationship to CPP |
Disorders of the reproductive tract
Endometriosis is the most common gynecologic diagnosis in women who have CPP. There is significant epidemiologic evidence that endometriosis causes CPP. There is also strong evidence that endometriosis is a risk factor for CPP.
For example, human and animal experimental data suggest that women who have endometriosis have more episodes of urinary calculosis—and more severe pain—than women who do not have endometriosis.9,10 They also are more likely to report vaginal pain than are women who do not have endometriosis, and that vaginal pain is more likely to be severe.
Such viscerovisceral interactions may play a significant role in CPP in women and may explain why some women who have a history of endometriosis have persistent pelvic pain after their endometriosis is gone, or even appear to develop other pain syndromes, such as interstitial cystitis.11
Our introductory case illustrates these concepts. Sara B.’s history is classic for endometriosis-associated pelvic pain; that was her original diagnosis. Although her pelvic pain recurred and persisted, a repeat laparoscopy found no endometriosis—but it did reveal evidence of interstitial cystitis and painful bladder syndrome (IC/PBS). Could Sara’s current pain be neuropathic or inflammatory?
Treatment of IC/PBS targets neuropathic and inflammatory pain mechanisms, but this approach has not been fully explored for endometriosis. Might the visceral pain mechanisms be as important as the end-organ diagnoses? Clearly, this area merits further attention in gynecology.
Pelvic inflammatory disease (PID) often causes CPP. Approximately 18% to 35% of all women who have acute PID develop CPP.12,13 The actual mechanisms by which CPP results from PID are not known, but it seems likely that both inflammatory and neuropathic mechanisms are important. Adhesive disease secondary to PID may also contribute to CPP by generating nociceptive pain. The route of treatment of PID—parenteral or oral antibiotics—does not appear to affect the odds of developing CPP.
Pelvic congestion syndrome is a controversial diagnosis that is uncommon in the United States. However, a well-designed study from Turkey suggests that about 40% of women who consult a gynecologist about CPP may have this syndrome.14 In Sara’s case, pelvic congestion syndrome was diagnosed after venous dilation and delayed emptying were confirmed by selective ovarian venography (transcervical venography is another option).15 This approach is recommended.
According to the data, the most effective treatment of pelvic congestion syndrome is a high-dose progestin or gonadotropin-releasing hormone (GnRH) agonist.14,16 Only observational data back treatment with ovarian venous embolization, which was performed in Sara’s case.8
Urinary tract disorders
Interstitial cystitis/painful bladder syndrome is the most common urologic diagnosis among women who have CPP. Recent evidence suggests that 38% to 81% of women who are given a diagnosis of a reproductive-tract disorder may in fact have IC/PBS.17,18 Much of the recent evidence regarding interstitial cystitis suggests that inflammatory and neuropathic mechanisms are crucial in the generation of CPP; therefore, much of the treatment focuses on inflammatory and neuropathic pain.19,20
For example, among the treatments that alleviate IC/PBS to some degree are:
- amitriptyline, widely used for neuropathic pain21
- gapabentin, an anticonvulsant used to treat neuropathic pain22
- antihistamines directed at inflammation23
- intravesical instillation of a local anesthetic agent, which may target both inflammatory and neuropathic pain mechanisms.24
Although these therapies have not been widely studied for their efficacy in gynecologic disorders, they are likely to produce similar results.
Disorders of the GI tract
Irritable bowel syndrome is the most common GI diagnosis in women who have CPP. It is a clinical diagnosis, usually based on the Rome III criteria (TABLE 3). (Sara B. was evaluated when Rome II criteria were in use.)
Data from a primary-care database in the United Kingdom suggest that irritable bowel syndrome may be the most common diagnosis in women who have CPP (about 38% of patients).25 In some cases, irritable bowel syndrome presents primarily with lower abdominal or pelvic pain, so it must be considered in the differential diagnosis of CPP. It seems likely that the pain in irritable bowel syndrome is not simply nociceptive, but that inflammatory and nociceptive mechanisms play an important role, as well.26,27
TABLE 3
Rome III criteria for irritable bowel syndrome
| Two or more criteria must be present to make the diagnosis. |
Over the past 3 months, have you had at least 3 days when you have had abdominal pain or discomfort that:
|
8. What are the main somatic causes of chronic pelvic pain?
Abdominal wall myofascial pain syndrome
When there are trigger points and myofascial pain of the lower abdominal wall muscles or pelvic floor muscles, they often present as CPP.
The underlying mechanisms responsible for myofascial pain syndrome are not clear. Nociceptive pain seems to be an important mechanism, but it is not clear whether inflammatory and neuropathic changes occur in some patients with this syndrome.
Many women who have myofascial pain syndrome and CPP respond poorly to traditional treatment with physical therapy and trigger-point injections; this may be due to inflammatory or neuropathic changes, or both.
Pelvic floor tension myalgia
Pain due to abnormal tension of the pelvic floor muscles is well-described. In many cases, pelvic floor tension myalgia is a secondary phenomenon, as pelvic floor muscles react to the persistent presence of pelvic pain, which often has a visceral basis. In other cases, pelvic floor tension myalgia is a primary phenomenon and most likely represents myofascial pain syndrome of one or more of the pelvic floor muscles.
9. Are multiple anatomic sites and mechanisms the “norm”?
They may not be the norm, but it is not unusual to discover multiple diagnoses when evaluating a patient for CPP. Most published studies of women from primary-care practices suggest that 25% to 50% of patients have more than one diagnosis,5,25,28 and anecdotal experience from referral practices suggests that most women in such practices have more than one diagnosis. The most common diagnoses in most published series are endometriosis, adhesions, irritable bowel syndrome, and interstitial cystitis.18,29-31 The absence of somatic diagnoses in these series probably reflects the gynecologist’s tendency to concentrate on visceral elements in CPP.
10. When multiple systems are involved, is the pain greater?
Yes. Women who have more than one organ system involved in CPP have greater pain than women who have only one system involved. For example, 43% of patients who have CPP without GI or urologic symptoms had moderate or severe pain (mean visual analog score of 3.8), whereas 71% of women who had CPP and both GI and urologic symptoms had moderate to severe pain (mean visual analog scale score of 5.4).28
Pain is also more consistent in women who have multisystem symptoms. Women who have CPP are more likely than the general population to have dysmenorrhea (81% versus 58%) and dyspareunia (41% versus 14%). The severity of pain with intercourse and with menses is greater in women who have CPP and GI and urologic symptoms than in those who have no GI and urologic symptoms.
11. How is treatment affected by multiple diagnoses?
The presence of multiple diagnoses often reflects neuropathic changes and neuropathic pain. An accurate diagnosis of all pain generators, including neuropathic pain, seems vital to improving our management and treatment of women who have CPP.
For example, in Sara B.’s case, I prescribed norethindrone acetate to suppress menses, based on her history of endometriosis-associated pelvic pain and menstrual exacerbation of her symptoms. I prescribed oral pentosan polysulfate sodium and intravesical lidocaine and heparin for interstitial cystitis/painful bladder syndrome. And I gave Sara amitriptyline for both fibromyalgia and interstitial cystitis/painful bladder syndrome (as well as suspected neuropathic pain).
I also recommended a low-fat, high-fiber diet to help alleviate her irritable bowel syndrome.
Dr. Howard is a consultant to Ortho Women’s Health & Urology and a speaker for Abbott Pharmaceutical.
CASE: Multisystem involvement
makes diagnosis and treatment thorny
Sara B. is a 26-year-old gravida 4, para 3, abortus 1 who visits your office to be evaluated for chronic pelvic pain. She says her pain is most intense before and during her period and with intercourse. It is located primarily in her abdominopelvic area, but radiates to her lower extremities and lumbosacral back. It appears to be related to bowel function and meets Rome II criteria for irritable bowel syndrome (criteria developed by a panel of experts convened by the Rome Foundation).
Sara B. reports that she voids at least 20 times a day and once during the night. She has a history of depression, for which she takes sertraline (Zoloft), but no history of physical or sexual abuse. When she underwent laparoscopy more than 1 year ago, endometriosis was diagnosed visually.
Upon physical examination, you identify 13 positive fibromyalgia points, moderate tenderness of the posterior levator ani muscles, severe tenderness of the bladder, and moderate tenderness of the uterine fundus. You also find moderate tenderness in the adnexa and uterosacral ligaments bilaterally. Your tentative diagnosis: endometriosis, interstitial cystitis, fibromyalgia, and irritable bowel syndrome.
How do you confirm the diagnosis? And what treatment should you offer to her?
Chronic pelvic pain (CPP) is anything but simple. Sara B.’s case illustrates some of the complexity involved in the diagnostic evaluation and treatment of this disorder. Very rarely is the pain localized to one organ or system. More commonly, it involves multiple organs or anatomic areas within the pelvic region.
To confirm the diagnosis in Sara’s case, the next step would be a potassium chloride sensitivity test for interstitial cystitis. I would also start her on desipramine for fibromyalgia, and perform laparoscopy and cystoscopy with hydrodistention to explore the diagnosis further.
In Sara’s case, let’s assume that the repeat laparoscopy reveals glomerulations of the bladder but no recurrent endometriosis. I would administer oral pentosan polysulfate sodium and instill heparin and lidocaine in her bladder to improve her voiding pattern significantly (to the range of four to six times a day without nocturia). I would also prescribe continuous oral contraceptives to suppress her menses and alleviate some of her pain. In addition, I would be interested to see what a transjugular pelvic venogram would reveal. If it were to suggest severe pelvic congestion syndrome, I might perform embolization of both ovarian veins to provide additional relief.
Clearly, when confronted with a case as intricate as Sara’s, there are many ways to organize your thinking about the potential diagnoses that may cause or contribute to CPP. This article focuses on anatomic and mechanistic bases for evaluation of this disorder as a means of tailoring treatment appropriately. It explores these topics by addressing 11 critical questions, ranging from how pain is described to what to do about it.
1. How is pain defined?
Pain is an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage.1
Pain is defined in this way to make it clear that it is not just a sensory experience, but both a sensory and emotional experience. This means that the pain is always subjective and is not the same in all individuals—nor does it remain the same in the same person.
Individuals base their descriptions of pain on their unique prior experience of it. Many people report pain in the absence of tissue damage or any likely pathophysiologic cause, often for psychological reasons. If they regard their experience as pain and report it as they would pain caused by tissue damage, it should be accepted as pain. In defining pain, it is best to deliberately avoid tying pain to the stimulus.
What about CPP? There is no generally accepted definition. The American College of Obstetricians and Gynecologists (ACOG) defines it as noncyclic pain of at least 6 months’ duration that localizes to the anatomic pelvis, lumbosacral back, buttocks, or anterior abdominal wall at or below the umbilicus and that is severe enough to cause functional disability or lead to medical care.2
2. How many women suffer chronic pelvic pain?
Chronic pelvic pain is more common than is generally recognized. Here are some estimates:
- A US study conducted by the Gallup organization found that 15% of women 18 to 50 years old had CPP3
- A survey of women in family medicine and ObGyn offices found that 39% had CPP, although only 8% reported having it more often than “sometimes”4
- The Oxfordshire Women’s Health Study, a postal questionnaire survey of a random sample of women 18 to 49 years old in the general UK population, found a prevalence of 24%5
- A primary-care database in a UK study of women 15 to 73 years old found a prevalence of 38 cases for every 1,000 women. (The database contained annualized data that excluded women who had only dysmenorrhea or dyspareunia.) Although the study likely underestimated the prevalence of CPP, the finding does make it possible to compare prevalences in the same population: asthma (37/1,000), back pain (41/1,000), and migraine (21/1,000).6
3. What are the main types of pain involved?
They are nociceptive, inflammatory, and neuropathic pain.
Nociceptive pain occurs in response to a noxious stimulus that alerts the organism to impending tissue injury. One way to think of nociceptive pain is as “normal” or physiologic pain (FIGURE).
Acute pelvic pain is usually nociceptive in origin. CPP is usually not solely nociceptive in origin. It often involves inflammatory or neuropathic pain, or both (TABLE 1).
Inflammatory pain arises in response to tissue injury and the resulting inflammatory process. In some cases, the inflammatory response is actually a source of tissue injury (e.g., rheumatoid arthritis). Inflammatory pain may be an important mechanism in both acute and chronic pelvic pain.
Neuropathic pain is produced by damage to or dysfunction of neurons in the peripheral or central nervous system. It is not physiologic and is often a significant mechanism in the generation of CPP.
An understanding of inflammatory and neuropathic mechanisms is not esoteric, but has clinical significance.
FIGURE Noxious stimulus is often the trigger in acute and chronic pain
Nociceptive pain is a response to a noxious stimulus, alerting the organism to impending tissue injury. Four fundamental processes are involved in nociceptive pain: transduction, in which the stimulus is converted to a biochemical signal; transmission, in which the signal is transported from the peripheral nervous system to the dorsal ganglion and central nervous system; modulation, in which the intensity of the signal is increased or decreased; and perception, in which the organism experiences the pain.TABLE 1
Anatomic and mechanistic classification of pain
Central
|
Peripheral
|
4. Is chronic pelvic pain a disease—or a symptom?
My experience caring for patients who have CPP suggests that chronic pain is a disease, whereas acute pain is a symptom. This concept is controversial in gynecology, and CPP is often labeled as only a symptom, not a diagnosis.7 The search for one underlying disease means that the woman who has CPP frequently undergoes multiple surgical and other invasive procedures, often with incomplete or insignificant diagnoses and responses.
The assumption that CPP is always due to a specific pathologic process in somatic structures or viscera (nociceptive pain) excludes the possibility that CPP can be caused by prolonged or permanent dysfunction of the peripheral or central nervous system, or both (neuropathic pain), or by psychological mechanisms (central pain). Clinical knowledge lags behind basic science in this area and is not at all concrete.
Our ability to accurately diagnose neuropathic or inflammatory pain leaves room for improvement.
5. Is chronic pelvic pain a gynecologic disorder?
Gynecologists have traditionally thought of CPP as either gynecologic or nongynecologic in origin, but this framework has very limited clinical utility. An anatomic and mechanistic classification (TABLE 1) represents a far richer strategic approach to the diagnostic evaluation of CPP, allowing more comprehensive and effective treatment.
6. What distinguishes visceral from somatic pain?
In addition to recognizing the importance of nociceptive, inflammatory, and neuropathic mechanisms in the generation of CPP, it is useful to classify potential causes anatomically (TABLE 1). In the broadest anatomic categories, pain may be central or peripheral, or both. Central pain can be psychogenic or neurogenic, and peripheral pain can be visceral or somatic.
Visceral sources of CPP include the reproductive, genitourinary, and gastrointestinal (GI) tracts. Mechanistically, as has been discussed, visceral pain can be neuropathic, inflammatory, or nociceptive.
Potential somatic sources of CPP are myofascial, skeletal, and cutaneous. Mechanisms leading to somatic CPP can be neuropathic, inflammatory, or nociceptive.
Somatic pain is better understood than visceral pain, but knowledge about the latter has been expanding rapidly. Several characteristics distinguish visceral pain:
- Not all viscera generate pain, possibly owing to a lack of sensory receptors or appropriate nociceptive stimulus
- Visceral pain is not always linked to injury and, therefore, may be functional
- Visceral pain frequently results in somatic referral of pain, possibly due to central convergence of visceral and somatic afferents
- Visceral pain tends to be diffuse or poorly localized, probably because of the low concentration of nociceptive afferents within viscera (only 2% to 10% of total afferents to the spinal cord originate from visceral nociceptors).8
It is not clear whether there are visceral neurons dedicated solely to nociception; it appears that viscera utilize sympathetic and parasympathetic neurons as nociceptors. It also is important to note that the stimuli that activate somatic nociceptors—cutting, crushing, and burning, for example—do not generally cause visceral pain. Visceral nociceptive pain is generated in response to:
- distention of a viscous or organ capsule
- spasm of visceral muscular fibers
- ischemia from vascular disturbances
- hemorrhage
- neoplasm
- inflammation
- traction on mesentery.
Another characteristic that distinguishes visceral from somatic nociception: Visceral nociception utilizes a dorsal midline pathway within the central nervous system, in addition to the lateral spinothalamic tract pathway utilized by somatic nociception.
Although this anatomic and mechanistic classification is clinically useful in the diagnostic evaluation of CPP, it is an oversimplification. Most patients—like Sara B., described in the opening case—have multiple anatomic and mechanistic causes of their pain.
7. What are the primary visceral causes of chronic pelvic pain?
A limited number of visceral and somatic diagnoses are backed by level-A evidence as having a causal relationship with CPP (TABLE 2). A few are discussed here.
TABLE 2
Disorders that may cause CPP or make it worse*
Reproductive tract
|
Urinary tract
|
Gastrointestinal tract
|
Musculoskeletal system
|
| Depression |
| Somatization disorder |
| * Disorders with Level-A evidence, i.e., good and consistent scientific evidence of a causal relationship to CPP |
Disorders of the reproductive tract
Endometriosis is the most common gynecologic diagnosis in women who have CPP. There is significant epidemiologic evidence that endometriosis causes CPP. There is also strong evidence that endometriosis is a risk factor for CPP.
For example, human and animal experimental data suggest that women who have endometriosis have more episodes of urinary calculosis—and more severe pain—than women who do not have endometriosis.9,10 They also are more likely to report vaginal pain than are women who do not have endometriosis, and that vaginal pain is more likely to be severe.
Such viscerovisceral interactions may play a significant role in CPP in women and may explain why some women who have a history of endometriosis have persistent pelvic pain after their endometriosis is gone, or even appear to develop other pain syndromes, such as interstitial cystitis.11
Our introductory case illustrates these concepts. Sara B.’s history is classic for endometriosis-associated pelvic pain; that was her original diagnosis. Although her pelvic pain recurred and persisted, a repeat laparoscopy found no endometriosis—but it did reveal evidence of interstitial cystitis and painful bladder syndrome (IC/PBS). Could Sara’s current pain be neuropathic or inflammatory?
Treatment of IC/PBS targets neuropathic and inflammatory pain mechanisms, but this approach has not been fully explored for endometriosis. Might the visceral pain mechanisms be as important as the end-organ diagnoses? Clearly, this area merits further attention in gynecology.
Pelvic inflammatory disease (PID) often causes CPP. Approximately 18% to 35% of all women who have acute PID develop CPP.12,13 The actual mechanisms by which CPP results from PID are not known, but it seems likely that both inflammatory and neuropathic mechanisms are important. Adhesive disease secondary to PID may also contribute to CPP by generating nociceptive pain. The route of treatment of PID—parenteral or oral antibiotics—does not appear to affect the odds of developing CPP.
Pelvic congestion syndrome is a controversial diagnosis that is uncommon in the United States. However, a well-designed study from Turkey suggests that about 40% of women who consult a gynecologist about CPP may have this syndrome.14 In Sara’s case, pelvic congestion syndrome was diagnosed after venous dilation and delayed emptying were confirmed by selective ovarian venography (transcervical venography is another option).15 This approach is recommended.
According to the data, the most effective treatment of pelvic congestion syndrome is a high-dose progestin or gonadotropin-releasing hormone (GnRH) agonist.14,16 Only observational data back treatment with ovarian venous embolization, which was performed in Sara’s case.8
Urinary tract disorders
Interstitial cystitis/painful bladder syndrome is the most common urologic diagnosis among women who have CPP. Recent evidence suggests that 38% to 81% of women who are given a diagnosis of a reproductive-tract disorder may in fact have IC/PBS.17,18 Much of the recent evidence regarding interstitial cystitis suggests that inflammatory and neuropathic mechanisms are crucial in the generation of CPP; therefore, much of the treatment focuses on inflammatory and neuropathic pain.19,20
For example, among the treatments that alleviate IC/PBS to some degree are:
- amitriptyline, widely used for neuropathic pain21
- gapabentin, an anticonvulsant used to treat neuropathic pain22
- antihistamines directed at inflammation23
- intravesical instillation of a local anesthetic agent, which may target both inflammatory and neuropathic pain mechanisms.24
Although these therapies have not been widely studied for their efficacy in gynecologic disorders, they are likely to produce similar results.
Disorders of the GI tract
Irritable bowel syndrome is the most common GI diagnosis in women who have CPP. It is a clinical diagnosis, usually based on the Rome III criteria (TABLE 3). (Sara B. was evaluated when Rome II criteria were in use.)
Data from a primary-care database in the United Kingdom suggest that irritable bowel syndrome may be the most common diagnosis in women who have CPP (about 38% of patients).25 In some cases, irritable bowel syndrome presents primarily with lower abdominal or pelvic pain, so it must be considered in the differential diagnosis of CPP. It seems likely that the pain in irritable bowel syndrome is not simply nociceptive, but that inflammatory and nociceptive mechanisms play an important role, as well.26,27
TABLE 3
Rome III criteria for irritable bowel syndrome
| Two or more criteria must be present to make the diagnosis. |
Over the past 3 months, have you had at least 3 days when you have had abdominal pain or discomfort that:
|
8. What are the main somatic causes of chronic pelvic pain?
Abdominal wall myofascial pain syndrome
When there are trigger points and myofascial pain of the lower abdominal wall muscles or pelvic floor muscles, they often present as CPP.
The underlying mechanisms responsible for myofascial pain syndrome are not clear. Nociceptive pain seems to be an important mechanism, but it is not clear whether inflammatory and neuropathic changes occur in some patients with this syndrome.
Many women who have myofascial pain syndrome and CPP respond poorly to traditional treatment with physical therapy and trigger-point injections; this may be due to inflammatory or neuropathic changes, or both.
Pelvic floor tension myalgia
Pain due to abnormal tension of the pelvic floor muscles is well-described. In many cases, pelvic floor tension myalgia is a secondary phenomenon, as pelvic floor muscles react to the persistent presence of pelvic pain, which often has a visceral basis. In other cases, pelvic floor tension myalgia is a primary phenomenon and most likely represents myofascial pain syndrome of one or more of the pelvic floor muscles.
9. Are multiple anatomic sites and mechanisms the “norm”?
They may not be the norm, but it is not unusual to discover multiple diagnoses when evaluating a patient for CPP. Most published studies of women from primary-care practices suggest that 25% to 50% of patients have more than one diagnosis,5,25,28 and anecdotal experience from referral practices suggests that most women in such practices have more than one diagnosis. The most common diagnoses in most published series are endometriosis, adhesions, irritable bowel syndrome, and interstitial cystitis.18,29-31 The absence of somatic diagnoses in these series probably reflects the gynecologist’s tendency to concentrate on visceral elements in CPP.
10. When multiple systems are involved, is the pain greater?
Yes. Women who have more than one organ system involved in CPP have greater pain than women who have only one system involved. For example, 43% of patients who have CPP without GI or urologic symptoms had moderate or severe pain (mean visual analog score of 3.8), whereas 71% of women who had CPP and both GI and urologic symptoms had moderate to severe pain (mean visual analog scale score of 5.4).28
Pain is also more consistent in women who have multisystem symptoms. Women who have CPP are more likely than the general population to have dysmenorrhea (81% versus 58%) and dyspareunia (41% versus 14%). The severity of pain with intercourse and with menses is greater in women who have CPP and GI and urologic symptoms than in those who have no GI and urologic symptoms.
11. How is treatment affected by multiple diagnoses?
The presence of multiple diagnoses often reflects neuropathic changes and neuropathic pain. An accurate diagnosis of all pain generators, including neuropathic pain, seems vital to improving our management and treatment of women who have CPP.
For example, in Sara B.’s case, I prescribed norethindrone acetate to suppress menses, based on her history of endometriosis-associated pelvic pain and menstrual exacerbation of her symptoms. I prescribed oral pentosan polysulfate sodium and intravesical lidocaine and heparin for interstitial cystitis/painful bladder syndrome. And I gave Sara amitriptyline for both fibromyalgia and interstitial cystitis/painful bladder syndrome (as well as suspected neuropathic pain).
I also recommended a low-fat, high-fiber diet to help alleviate her irritable bowel syndrome.
1. International Association for the Study of Pain. IASP Pain Terminology. Available at: http://www.iasp-pain.org/AM/Template.cfm?Section=Pain_Definitions&Template=/CM/HTMLDisplay.cfm&ContentID=1728. Accessed July 10, 2009.
2. Chronic pelvic pain. ACOG Practice Bulletin No. 51. Obstet. Gynecol. 2004;103:589-605.
3. Mathias SD, Kuppermann M, Liberman RF, Lipschutz RC, Steege JF. Chronic pelvic pain: prevalence, health-related quality of life, and economic correlates. Obstet Gynecol. 1996;87:321-327.
4. Jamieson DJ, Steege JF. The prevalence of dysmenorrhea, dyspareunia, pelvic pain, and irritable bowel syndrome in primary care practices. Obstet Gynecol. 1996;87:55-58.
5. Zondervan KT, Yudkin PL, Vessey MP, et al. The community prevalence of chronic pelvic pain in women and associated illness behaviour. Br J Gen Pract. 2001;51:541-547.
6. Zondervan KT, Yudkin PL, Vessey MP, Dawes MG, Barlow DH, Kennedy SH. Prevalence and incidence of chronic pelvic pain in primary care: evidence from a national general practice database. Br J Obstet Gynaecol. 1999;106:1149-1155.
7. Scialli AR, Barbieri RL, Glasser MH, Olive DL, Winkel CA. Chronic Pelvic Pain: An Integrated Approach. APGO Education Series on Women’s Health Issues. Washington, DC: Association of Professors of Gynecology and Obstetrics; 2000.
8. Maleux G, Stockx L, Wilms G, Marchal G. Ovarian vein embolization for the treatment of pelvic congestion syndrome: long-term technical and clinical results. J Vasc Interv Radiol. 2000;11:859-864.
9. Giamberardino MA, De Laurentis S, Affaitati G, Lerza R, Lapenna D, Vecchiet L. Modulation of pain and hyperalgesia from the urinary tract by algogenic conditions of the reproductive organs in women. Neurosci Lett. 2001;304:61-64.
10. Giamberardino MA, Berkley KJ, Affaitati G, et al. Influence of endometriosis on pain behaviors and muscle hyperalgesia induced by a ureteral calculosis in female rats. Pain. 2002;95:247-257.
11. Parsons CL, Bullen M, Kahn BS, Stanford EJ, Willems JJ. Gynecologic presentation of interstitial cystitis as detected by intravesical potassium sensitivity. Obstet Gynecol. 2001;98:127-132.
12. Weström L. Effect of acute pelvic inflammatory disease on fertility. Am J Obstet Gynecol. 1975;121:707-713.
13. Ness RB, Soper DE, Holley RL, et al. Effectiveness of inpatient and outpatient treatment strategies for women with pelvic inflammatory disease: results from the Pelvic Inflammatory Disease Evaluation and Clinical Health (PEACH) Randomized Trial. Am J Obstet Gynecol. 2002;186:929-937.
14. Soysal ME, Soysal S, Vicdan K, Ozer S. A randomized controlled trial of goserelin and medroxyprogesterone acetate in the treatment of pelvic congestion. Hum Reprod. 2001;16:931-939.
15. Beard RW, Highman JH, Pearce S, Reginald PW. Diagnosis of pelvic varicosities in women with chronic pelvic pain. Lancet. 1984;2:946-949.
16. Farquhar CM, Rogers V, Franks S, Pearce S, Wadsworth J, Beard RW. A randomized controlled trial of medroxyprogesterone acetate and psychotherapy for the treatment of pelvic congestion. Br J Obstet Gynaecol. 1989;96:1153-1162.
17. Clemons JL, Arya LA, Myers DL. Diagnosing interstitial cystitis in women with chronic pelvic pain. Obstet Gynecol. 2002;100:337-341.
18. Parsons CL, Dell J, Stanford EJ, Bullen M, Kahn BS, Willems JJ. The prevalence of interstitial cystitis in gynecologic patients with pelvic pain, as detected by intravesical potassium sensitivity. Am J Obstet Gynecol. 2002;187:1395-1400.
19. Wesselmann U. Interstitial cystitis: a chronic visceral pain syndrome. Urology. 2001;57:32-39.
20. Butrick CW. Interstitial cystitis and chronic pelvic pain: new insights in neuropathology, diagnosis, and treatment. Clin Obstet Gynecol. 2003;46:811-823.
21. van Ophoven A, Pokupic S, Heinecke A, Hertle L. A prospective, randomized, placebo controlled, double-blind study of amitriptyline for the treatment of interstitial cystitis. J Urol. 2004;172:533-536.
22. Sasaki K, Smith CP, Chuang YC, Lee JY, Kim JC, Chancellor MB. Oral gabapentin (Neurontin) treatment of refractory genitourinary tract pain. Tech Urol. 2001;7:47-49.
23. Sant GR, Propert KJ, Hanno PM, et al. Interstitial Cystitis Clinical Trials Group. A pilot clinical trial of oral pentosan polysulfate and oral hydroxyzine in patients with interstitial cystitis. J Urol. 2003;170:810-815.
24. Parsons CL. Successful downregulation of bladder sensory nerves with combination of heparin and alkalinized lidocaine in patients with interstitial cystitis. Urology. 2005;65:45-48.
25. Zondervan KT, Yudkin PL, Vessey MP, Dawes MG, Barlow DH, Kennedy SH. Patterns of diagnosis and referral in women consulting for chronic pelvic pain in UK primary care. Br J Obstet Gynaecol. 1999;106:1156-1161.
26. Kwan CL, Diamant NE, Mikula K, Davis KD. Characteristics of rectal perception are altered in irritable bowel syndrome. Pain. 2005;113:160-171.
27. Wilder-Smith CH, Schindler D, Lovblad K, Redmond SM, Nirkko A. Brain functional magnetic resonance imaging of rectal pain and activation of endogenous inhibitory mechanisms in irritable bowel syndrome patient subgroups and healthy controls. Gut. 2004;53:1595-1601.
28. Zondervan KT, Yudkin PL, Vessey MP, et al. Chronic pelvic pain in the community—symptoms, investigations, and diagnoses. Am J Obstet Gynecol. 2001;184:1149-1155.
29. Williams RE, Hartmann KE, Sandler RS, Miller WC, Steege JF. Prevalence and characteristics of irritable bowel syndrome among women with chronic pelvic pain. Obstet Gynecol. 2004;104:452-458.
30. Howard FM. The role of laparoscopy in chronic pelvic pain: promise and pitfalls. Obstet Gynecol Surv. 1993;48:357-387.
31. Chung MK, Chung RR, Gordon D, Jennings C. The evil twins of chronic pelvic pain syndrome: endometriosis and interstitial cystitis. JSLS. 2002;6:311-314.
1. International Association for the Study of Pain. IASP Pain Terminology. Available at: http://www.iasp-pain.org/AM/Template.cfm?Section=Pain_Definitions&Template=/CM/HTMLDisplay.cfm&ContentID=1728. Accessed July 10, 2009.
2. Chronic pelvic pain. ACOG Practice Bulletin No. 51. Obstet. Gynecol. 2004;103:589-605.
3. Mathias SD, Kuppermann M, Liberman RF, Lipschutz RC, Steege JF. Chronic pelvic pain: prevalence, health-related quality of life, and economic correlates. Obstet Gynecol. 1996;87:321-327.
4. Jamieson DJ, Steege JF. The prevalence of dysmenorrhea, dyspareunia, pelvic pain, and irritable bowel syndrome in primary care practices. Obstet Gynecol. 1996;87:55-58.
5. Zondervan KT, Yudkin PL, Vessey MP, et al. The community prevalence of chronic pelvic pain in women and associated illness behaviour. Br J Gen Pract. 2001;51:541-547.
6. Zondervan KT, Yudkin PL, Vessey MP, Dawes MG, Barlow DH, Kennedy SH. Prevalence and incidence of chronic pelvic pain in primary care: evidence from a national general practice database. Br J Obstet Gynaecol. 1999;106:1149-1155.
7. Scialli AR, Barbieri RL, Glasser MH, Olive DL, Winkel CA. Chronic Pelvic Pain: An Integrated Approach. APGO Education Series on Women’s Health Issues. Washington, DC: Association of Professors of Gynecology and Obstetrics; 2000.
8. Maleux G, Stockx L, Wilms G, Marchal G. Ovarian vein embolization for the treatment of pelvic congestion syndrome: long-term technical and clinical results. J Vasc Interv Radiol. 2000;11:859-864.
9. Giamberardino MA, De Laurentis S, Affaitati G, Lerza R, Lapenna D, Vecchiet L. Modulation of pain and hyperalgesia from the urinary tract by algogenic conditions of the reproductive organs in women. Neurosci Lett. 2001;304:61-64.
10. Giamberardino MA, Berkley KJ, Affaitati G, et al. Influence of endometriosis on pain behaviors and muscle hyperalgesia induced by a ureteral calculosis in female rats. Pain. 2002;95:247-257.
11. Parsons CL, Bullen M, Kahn BS, Stanford EJ, Willems JJ. Gynecologic presentation of interstitial cystitis as detected by intravesical potassium sensitivity. Obstet Gynecol. 2001;98:127-132.
12. Weström L. Effect of acute pelvic inflammatory disease on fertility. Am J Obstet Gynecol. 1975;121:707-713.
13. Ness RB, Soper DE, Holley RL, et al. Effectiveness of inpatient and outpatient treatment strategies for women with pelvic inflammatory disease: results from the Pelvic Inflammatory Disease Evaluation and Clinical Health (PEACH) Randomized Trial. Am J Obstet Gynecol. 2002;186:929-937.
14. Soysal ME, Soysal S, Vicdan K, Ozer S. A randomized controlled trial of goserelin and medroxyprogesterone acetate in the treatment of pelvic congestion. Hum Reprod. 2001;16:931-939.
15. Beard RW, Highman JH, Pearce S, Reginald PW. Diagnosis of pelvic varicosities in women with chronic pelvic pain. Lancet. 1984;2:946-949.
16. Farquhar CM, Rogers V, Franks S, Pearce S, Wadsworth J, Beard RW. A randomized controlled trial of medroxyprogesterone acetate and psychotherapy for the treatment of pelvic congestion. Br J Obstet Gynaecol. 1989;96:1153-1162.
17. Clemons JL, Arya LA, Myers DL. Diagnosing interstitial cystitis in women with chronic pelvic pain. Obstet Gynecol. 2002;100:337-341.
18. Parsons CL, Dell J, Stanford EJ, Bullen M, Kahn BS, Willems JJ. The prevalence of interstitial cystitis in gynecologic patients with pelvic pain, as detected by intravesical potassium sensitivity. Am J Obstet Gynecol. 2002;187:1395-1400.
19. Wesselmann U. Interstitial cystitis: a chronic visceral pain syndrome. Urology. 2001;57:32-39.
20. Butrick CW. Interstitial cystitis and chronic pelvic pain: new insights in neuropathology, diagnosis, and treatment. Clin Obstet Gynecol. 2003;46:811-823.
21. van Ophoven A, Pokupic S, Heinecke A, Hertle L. A prospective, randomized, placebo controlled, double-blind study of amitriptyline for the treatment of interstitial cystitis. J Urol. 2004;172:533-536.
22. Sasaki K, Smith CP, Chuang YC, Lee JY, Kim JC, Chancellor MB. Oral gabapentin (Neurontin) treatment of refractory genitourinary tract pain. Tech Urol. 2001;7:47-49.
23. Sant GR, Propert KJ, Hanno PM, et al. Interstitial Cystitis Clinical Trials Group. A pilot clinical trial of oral pentosan polysulfate and oral hydroxyzine in patients with interstitial cystitis. J Urol. 2003;170:810-815.
24. Parsons CL. Successful downregulation of bladder sensory nerves with combination of heparin and alkalinized lidocaine in patients with interstitial cystitis. Urology. 2005;65:45-48.
25. Zondervan KT, Yudkin PL, Vessey MP, Dawes MG, Barlow DH, Kennedy SH. Patterns of diagnosis and referral in women consulting for chronic pelvic pain in UK primary care. Br J Obstet Gynaecol. 1999;106:1156-1161.
26. Kwan CL, Diamant NE, Mikula K, Davis KD. Characteristics of rectal perception are altered in irritable bowel syndrome. Pain. 2005;113:160-171.
27. Wilder-Smith CH, Schindler D, Lovblad K, Redmond SM, Nirkko A. Brain functional magnetic resonance imaging of rectal pain and activation of endogenous inhibitory mechanisms in irritable bowel syndrome patient subgroups and healthy controls. Gut. 2004;53:1595-1601.
28. Zondervan KT, Yudkin PL, Vessey MP, et al. Chronic pelvic pain in the community—symptoms, investigations, and diagnoses. Am J Obstet Gynecol. 2001;184:1149-1155.
29. Williams RE, Hartmann KE, Sandler RS, Miller WC, Steege JF. Prevalence and characteristics of irritable bowel syndrome among women with chronic pelvic pain. Obstet Gynecol. 2004;104:452-458.
30. Howard FM. The role of laparoscopy in chronic pelvic pain: promise and pitfalls. Obstet Gynecol Surv. 1993;48:357-387.
31. Chung MK, Chung RR, Gordon D, Jennings C. The evil twins of chronic pelvic pain syndrome: endometriosis and interstitial cystitis. JSLS. 2002;6:311-314.
Are staples or sutures better for closing the skin at cesarean delivery?
The rate of cesarean delivery in the United States reached its highest level yet— 31.8%—in 2007.1 At more than 1.2 million procedures each year, cesarean delivery is the most common major surgery performed, but few studies have explored the techniques involved. Recent investigations have focused on whether it is advisable to close the peritoneum at the time of cesarean section,2 how to perform and close the hysterotomy,3 and the timing of prophylactic antibiotics.4,5 But we need more evidence to optimize outcomes.
Rousseau and colleagues have made a commendable effort to sift through one of the many unanswered questions regarding cesarean technique: What is the best skin closure? Their prospective, randomized, controlled trial compared staple closure with subcuticular closure using 4-0 Monocryl.
The authors did many things we have come to expect from clinical trials, including:
- a priori sample-size calculation to ensure adequate statistical power
- randomization by group of 8
- stratification of randomization by primary and repeat cesarean delivery
- assessment of wound cosmesis in a blinded, masked fashion.
They found a “statistically significant” difference in the pain score at 6 postoperative weeks between staples and sutures, with staples having the lower mean score (0.2 vs 0.5; P=.04). They also demonstrated shorter operative time for staple closure (32 vs 41 minutes; P<.001).
When such a study is published, it is easy to assume that the issue has been settled and to change or not change practice, depending on your existing technique—but that is often unwise. Every study has limitations. Even when statistically significant benefits are demonstrated, as they are in this study, it may not always be clear whether your patients match the patients in the study, or whether your technique matches what has been administered during the investigation.
In this case, a few problems need to be pointed out:
- Although the evaluation of cosmesis was by masked clinicians, assessment of the primary outcome—pain—was conducted by the patients themselves, who were not masked. One can easily see that awareness of a suture retained beneath the skin might bias a patient’s perception of pain and discomfort. It would be relatively easy to mask the type of closure—even from patients—on postoperative day 1, but masking would become much more difficult when the staples needed to be removed.
- The issue of statistical analysis can sometimes be dull, but is occasionally paramount in determining validity of a study. In this case, the primary outcome—the pain scale—was considered a continuous outcome and compared using a Student’s t-test. An important assumption in this test is that the data are normally distributed. However, the authors do not make it clear whether they tested the data for normalcy. Particularly at the 6-week evaluation, when the mean value was between 0 and 1 for both groups, it seems unlikely that the data were normally distributed. As a result, the difference in pain scores—0.2 vs 0.5—could have been driven by a few high values in one group. Statistically, this would have been easy to manage by changing the comparison to a Wilcoxon rank-sum test.
Despite these limitations, it does seem unlikely that the pain at 6 weeks would have been worse in the staple group.
These findings contrast those of another study of the same topic, which found less pain in the subcuticular suture group.6 In that unmasked study, subcuticular closure was determined to be more “cosmetically attractive” by the patients and their physicians. Again, one needs to be concerned about bias.
To echo the latest Cochrane review of this topic, there is insufficient evidence—even after this investigation—to systematically recommend one type of skin closure over another. However, given the masked evaluation of the wounds and the clear lack of difference in their appearance in this study, cosmesis alone does not seem to be sufficient reason to utilize subcuticular sutures to close the skin at cesarean delivery. In fact, the shorter operative time documented in the staple-closure group in this study could tip the scale in favor of using staples for this procedure.
Clearly, we need many more investigations of surgical technique and perioperative care in regard to cesarean delivery. Although I hope that cesarean section does not remain the most common surgical procedure, it seems likely that it will always be a large part of obstetric care. Therefore, optimization of outcomes merits attention.—AARON B. CAUGHEY, MD, PHD
1. Hamilton BE, Martin JA, Ventura SJ. Birth: Preliminary data for 2007. Natl Vital Stat Rep. 2009;57:1-23.
2. Lyell DJ, Caughey AB, Chu E, Daniels K. Peritoneal closure at primary cesarean delivery and adhesions. Obstet Gynecol. 2005;106:275-280.
3. Dodd JM, Anderson ER, Gates S. Surgical techniques for uterine incision and uterine closure at the time of caesarean section. Cochrane Database Syst Rev. 2008;Jul 16;(3):CD004732.-
4. Sullivan SA, Smith T, Chang E, Hulsey T, Vandorsten JP, Soper D. Administration of cefazolin prior to skin incision is superior to cefazolin at cord clamping in preventing postcesarean infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2007;196:455.e1-455.e5.
5. Kaimal AJ, Zlatnik MG, Cheng YW, et al. Effect of change in policy regarding the timing of prophylactic antibiotics on the rate of postcesarean delivery surgical-site infections. Am J Obstet Gynecol. 2008;199:310.e1-310.e5.
6. Frishman GN, Schwartz T, Hogan JW. Closure of Pfannenstiel skin incisions. Staples vs. subcuticular suture. J Reprod Med. 1997;42(10):627-630.
The rate of cesarean delivery in the United States reached its highest level yet— 31.8%—in 2007.1 At more than 1.2 million procedures each year, cesarean delivery is the most common major surgery performed, but few studies have explored the techniques involved. Recent investigations have focused on whether it is advisable to close the peritoneum at the time of cesarean section,2 how to perform and close the hysterotomy,3 and the timing of prophylactic antibiotics.4,5 But we need more evidence to optimize outcomes.
Rousseau and colleagues have made a commendable effort to sift through one of the many unanswered questions regarding cesarean technique: What is the best skin closure? Their prospective, randomized, controlled trial compared staple closure with subcuticular closure using 4-0 Monocryl.
The authors did many things we have come to expect from clinical trials, including:
- a priori sample-size calculation to ensure adequate statistical power
- randomization by group of 8
- stratification of randomization by primary and repeat cesarean delivery
- assessment of wound cosmesis in a blinded, masked fashion.
They found a “statistically significant” difference in the pain score at 6 postoperative weeks between staples and sutures, with staples having the lower mean score (0.2 vs 0.5; P=.04). They also demonstrated shorter operative time for staple closure (32 vs 41 minutes; P<.001).
When such a study is published, it is easy to assume that the issue has been settled and to change or not change practice, depending on your existing technique—but that is often unwise. Every study has limitations. Even when statistically significant benefits are demonstrated, as they are in this study, it may not always be clear whether your patients match the patients in the study, or whether your technique matches what has been administered during the investigation.
In this case, a few problems need to be pointed out:
- Although the evaluation of cosmesis was by masked clinicians, assessment of the primary outcome—pain—was conducted by the patients themselves, who were not masked. One can easily see that awareness of a suture retained beneath the skin might bias a patient’s perception of pain and discomfort. It would be relatively easy to mask the type of closure—even from patients—on postoperative day 1, but masking would become much more difficult when the staples needed to be removed.
- The issue of statistical analysis can sometimes be dull, but is occasionally paramount in determining validity of a study. In this case, the primary outcome—the pain scale—was considered a continuous outcome and compared using a Student’s t-test. An important assumption in this test is that the data are normally distributed. However, the authors do not make it clear whether they tested the data for normalcy. Particularly at the 6-week evaluation, when the mean value was between 0 and 1 for both groups, it seems unlikely that the data were normally distributed. As a result, the difference in pain scores—0.2 vs 0.5—could have been driven by a few high values in one group. Statistically, this would have been easy to manage by changing the comparison to a Wilcoxon rank-sum test.
Despite these limitations, it does seem unlikely that the pain at 6 weeks would have been worse in the staple group.
These findings contrast those of another study of the same topic, which found less pain in the subcuticular suture group.6 In that unmasked study, subcuticular closure was determined to be more “cosmetically attractive” by the patients and their physicians. Again, one needs to be concerned about bias.
To echo the latest Cochrane review of this topic, there is insufficient evidence—even after this investigation—to systematically recommend one type of skin closure over another. However, given the masked evaluation of the wounds and the clear lack of difference in their appearance in this study, cosmesis alone does not seem to be sufficient reason to utilize subcuticular sutures to close the skin at cesarean delivery. In fact, the shorter operative time documented in the staple-closure group in this study could tip the scale in favor of using staples for this procedure.
Clearly, we need many more investigations of surgical technique and perioperative care in regard to cesarean delivery. Although I hope that cesarean section does not remain the most common surgical procedure, it seems likely that it will always be a large part of obstetric care. Therefore, optimization of outcomes merits attention.—AARON B. CAUGHEY, MD, PHD
The rate of cesarean delivery in the United States reached its highest level yet— 31.8%—in 2007.1 At more than 1.2 million procedures each year, cesarean delivery is the most common major surgery performed, but few studies have explored the techniques involved. Recent investigations have focused on whether it is advisable to close the peritoneum at the time of cesarean section,2 how to perform and close the hysterotomy,3 and the timing of prophylactic antibiotics.4,5 But we need more evidence to optimize outcomes.
Rousseau and colleagues have made a commendable effort to sift through one of the many unanswered questions regarding cesarean technique: What is the best skin closure? Their prospective, randomized, controlled trial compared staple closure with subcuticular closure using 4-0 Monocryl.
The authors did many things we have come to expect from clinical trials, including:
- a priori sample-size calculation to ensure adequate statistical power
- randomization by group of 8
- stratification of randomization by primary and repeat cesarean delivery
- assessment of wound cosmesis in a blinded, masked fashion.
They found a “statistically significant” difference in the pain score at 6 postoperative weeks between staples and sutures, with staples having the lower mean score (0.2 vs 0.5; P=.04). They also demonstrated shorter operative time for staple closure (32 vs 41 minutes; P<.001).
When such a study is published, it is easy to assume that the issue has been settled and to change or not change practice, depending on your existing technique—but that is often unwise. Every study has limitations. Even when statistically significant benefits are demonstrated, as they are in this study, it may not always be clear whether your patients match the patients in the study, or whether your technique matches what has been administered during the investigation.
In this case, a few problems need to be pointed out:
- Although the evaluation of cosmesis was by masked clinicians, assessment of the primary outcome—pain—was conducted by the patients themselves, who were not masked. One can easily see that awareness of a suture retained beneath the skin might bias a patient’s perception of pain and discomfort. It would be relatively easy to mask the type of closure—even from patients—on postoperative day 1, but masking would become much more difficult when the staples needed to be removed.
- The issue of statistical analysis can sometimes be dull, but is occasionally paramount in determining validity of a study. In this case, the primary outcome—the pain scale—was considered a continuous outcome and compared using a Student’s t-test. An important assumption in this test is that the data are normally distributed. However, the authors do not make it clear whether they tested the data for normalcy. Particularly at the 6-week evaluation, when the mean value was between 0 and 1 for both groups, it seems unlikely that the data were normally distributed. As a result, the difference in pain scores—0.2 vs 0.5—could have been driven by a few high values in one group. Statistically, this would have been easy to manage by changing the comparison to a Wilcoxon rank-sum test.
Despite these limitations, it does seem unlikely that the pain at 6 weeks would have been worse in the staple group.
These findings contrast those of another study of the same topic, which found less pain in the subcuticular suture group.6 In that unmasked study, subcuticular closure was determined to be more “cosmetically attractive” by the patients and their physicians. Again, one needs to be concerned about bias.
To echo the latest Cochrane review of this topic, there is insufficient evidence—even after this investigation—to systematically recommend one type of skin closure over another. However, given the masked evaluation of the wounds and the clear lack of difference in their appearance in this study, cosmesis alone does not seem to be sufficient reason to utilize subcuticular sutures to close the skin at cesarean delivery. In fact, the shorter operative time documented in the staple-closure group in this study could tip the scale in favor of using staples for this procedure.
Clearly, we need many more investigations of surgical technique and perioperative care in regard to cesarean delivery. Although I hope that cesarean section does not remain the most common surgical procedure, it seems likely that it will always be a large part of obstetric care. Therefore, optimization of outcomes merits attention.—AARON B. CAUGHEY, MD, PHD
1. Hamilton BE, Martin JA, Ventura SJ. Birth: Preliminary data for 2007. Natl Vital Stat Rep. 2009;57:1-23.
2. Lyell DJ, Caughey AB, Chu E, Daniels K. Peritoneal closure at primary cesarean delivery and adhesions. Obstet Gynecol. 2005;106:275-280.
3. Dodd JM, Anderson ER, Gates S. Surgical techniques for uterine incision and uterine closure at the time of caesarean section. Cochrane Database Syst Rev. 2008;Jul 16;(3):CD004732.-
4. Sullivan SA, Smith T, Chang E, Hulsey T, Vandorsten JP, Soper D. Administration of cefazolin prior to skin incision is superior to cefazolin at cord clamping in preventing postcesarean infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2007;196:455.e1-455.e5.
5. Kaimal AJ, Zlatnik MG, Cheng YW, et al. Effect of change in policy regarding the timing of prophylactic antibiotics on the rate of postcesarean delivery surgical-site infections. Am J Obstet Gynecol. 2008;199:310.e1-310.e5.
6. Frishman GN, Schwartz T, Hogan JW. Closure of Pfannenstiel skin incisions. Staples vs. subcuticular suture. J Reprod Med. 1997;42(10):627-630.
1. Hamilton BE, Martin JA, Ventura SJ. Birth: Preliminary data for 2007. Natl Vital Stat Rep. 2009;57:1-23.
2. Lyell DJ, Caughey AB, Chu E, Daniels K. Peritoneal closure at primary cesarean delivery and adhesions. Obstet Gynecol. 2005;106:275-280.
3. Dodd JM, Anderson ER, Gates S. Surgical techniques for uterine incision and uterine closure at the time of caesarean section. Cochrane Database Syst Rev. 2008;Jul 16;(3):CD004732.-
4. Sullivan SA, Smith T, Chang E, Hulsey T, Vandorsten JP, Soper D. Administration of cefazolin prior to skin incision is superior to cefazolin at cord clamping in preventing postcesarean infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2007;196:455.e1-455.e5.
5. Kaimal AJ, Zlatnik MG, Cheng YW, et al. Effect of change in policy regarding the timing of prophylactic antibiotics on the rate of postcesarean delivery surgical-site infections. Am J Obstet Gynecol. 2008;199:310.e1-310.e5.
6. Frishman GN, Schwartz T, Hogan JW. Closure of Pfannenstiel skin incisions. Staples vs. subcuticular suture. J Reprod Med. 1997;42(10):627-630.
Does cognitive function decline during the menopausal transition?
These important findings from SWAN confirm the memory problems reported by many women during the menopausal transition. They also build on the findings of clinical trials of HT in younger menopausal women, which explored the effect of HT on cardiovascular health, to underscore the critical role that timing of such therapy can play.1,2
Other cohort studies of middle-aged women in the United States have found the menopausal transition to be associated with undesirable changes in mood3,4; some experts have described the endocrine shifts that accompany menopause as “hormonal chaos.”5
Details of the study
Participants were 42 to 52 years old and had an intact uterus and at least one ovary at entry. They were followed for 4 years, with delineation of the menopausal stage (i.e., premenopause, early and late perimenopause, and menopause) and assessment of hormone use prior to the final menstrual period and after menopause. The outcome was longitudinal performance in three cognitive domains:
- processing speed—assessed using the Symbol Digit Modalities Test. Premenopausal, early perimenopausal, and postmenopausal women improved with repeated administration of this test, but late perimenopausal women did not. Prior use of HT improved the score, whereas late use of HT reduced it.
- verbal memory—evaluated via the East Boston Memory Test. Test scores increased during premenopause and postmenopause but not during early or late perimenopause. Prior use of HT improved the test score, but late use reduced the score.
- working memory—assessed using the Digit Span Backward test. This domain did not vary by stage of menopause.
Participants who initiated menopausal HT or oral contraceptives prior to the last menstrual period were excluded from the analysis during use of HT. They were allowed to reenter the study, however, once HT or oral contraceptives were discontinued.
As the cognitive component of SWAN began, the mean age of participants was 50 years, and 8% were premenopausal, 49% were early perimenopausal, 12% were late perimenopausal, and 27% were postmenopausal. In addition, 4% were both postmenopausal and current users of HT.
Because perimenopausal HT should include a dosage of progestin adequate to suppress ovulation in order to prevent iatrogenic irregular uterine bleeding, women who are healthy, lean, nonsmoking, and still having menstrual periods can safely use a conventional oral contraceptive. Options for other symptomatic perimenopausal women include a continuous oral menopausal regimen formulated with 5 μg of ethinyl estradiol and 1 mg of norethindrone acetate (Femhrt 1/5) or 1 mg of estradiol and 0.5 mg of norethindrone acetate (Activella 1/0.5 or generic).—ANDREW M. KAUNITZ, MD
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. For the WHI and WHI-CACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Freeman EW, Sammel MD, Lin H, et al. Symptoms associated with the menopausal transition and reproductive hormones in midlife women. Obstet Gynecol. 2007;110(2 Pt 1):230-240.
4. Woods NF, Smith-DiJulio K, Percival DB, Tao EY, Mariella A, Mitchell S. Depressed mood during the menopausal transition and early postmenopause: observations from the Seattle Midlife Women’s Health Study. Menopause. 2008;15:223-232.
5. Berga SL. Disordered folliculogenesis during the menopausal transition: explaining chaos. Menopause. 2009;16:11-12.
These important findings from SWAN confirm the memory problems reported by many women during the menopausal transition. They also build on the findings of clinical trials of HT in younger menopausal women, which explored the effect of HT on cardiovascular health, to underscore the critical role that timing of such therapy can play.1,2
Other cohort studies of middle-aged women in the United States have found the menopausal transition to be associated with undesirable changes in mood3,4; some experts have described the endocrine shifts that accompany menopause as “hormonal chaos.”5
Details of the study
Participants were 42 to 52 years old and had an intact uterus and at least one ovary at entry. They were followed for 4 years, with delineation of the menopausal stage (i.e., premenopause, early and late perimenopause, and menopause) and assessment of hormone use prior to the final menstrual period and after menopause. The outcome was longitudinal performance in three cognitive domains:
- processing speed—assessed using the Symbol Digit Modalities Test. Premenopausal, early perimenopausal, and postmenopausal women improved with repeated administration of this test, but late perimenopausal women did not. Prior use of HT improved the score, whereas late use of HT reduced it.
- verbal memory—evaluated via the East Boston Memory Test. Test scores increased during premenopause and postmenopause but not during early or late perimenopause. Prior use of HT improved the test score, but late use reduced the score.
- working memory—assessed using the Digit Span Backward test. This domain did not vary by stage of menopause.
Participants who initiated menopausal HT or oral contraceptives prior to the last menstrual period were excluded from the analysis during use of HT. They were allowed to reenter the study, however, once HT or oral contraceptives were discontinued.
As the cognitive component of SWAN began, the mean age of participants was 50 years, and 8% were premenopausal, 49% were early perimenopausal, 12% were late perimenopausal, and 27% were postmenopausal. In addition, 4% were both postmenopausal and current users of HT.
Because perimenopausal HT should include a dosage of progestin adequate to suppress ovulation in order to prevent iatrogenic irregular uterine bleeding, women who are healthy, lean, nonsmoking, and still having menstrual periods can safely use a conventional oral contraceptive. Options for other symptomatic perimenopausal women include a continuous oral menopausal regimen formulated with 5 μg of ethinyl estradiol and 1 mg of norethindrone acetate (Femhrt 1/5) or 1 mg of estradiol and 0.5 mg of norethindrone acetate (Activella 1/0.5 or generic).—ANDREW M. KAUNITZ, MD
These important findings from SWAN confirm the memory problems reported by many women during the menopausal transition. They also build on the findings of clinical trials of HT in younger menopausal women, which explored the effect of HT on cardiovascular health, to underscore the critical role that timing of such therapy can play.1,2
Other cohort studies of middle-aged women in the United States have found the menopausal transition to be associated with undesirable changes in mood3,4; some experts have described the endocrine shifts that accompany menopause as “hormonal chaos.”5
Details of the study
Participants were 42 to 52 years old and had an intact uterus and at least one ovary at entry. They were followed for 4 years, with delineation of the menopausal stage (i.e., premenopause, early and late perimenopause, and menopause) and assessment of hormone use prior to the final menstrual period and after menopause. The outcome was longitudinal performance in three cognitive domains:
- processing speed—assessed using the Symbol Digit Modalities Test. Premenopausal, early perimenopausal, and postmenopausal women improved with repeated administration of this test, but late perimenopausal women did not. Prior use of HT improved the score, whereas late use of HT reduced it.
- verbal memory—evaluated via the East Boston Memory Test. Test scores increased during premenopause and postmenopause but not during early or late perimenopause. Prior use of HT improved the test score, but late use reduced the score.
- working memory—assessed using the Digit Span Backward test. This domain did not vary by stage of menopause.
Participants who initiated menopausal HT or oral contraceptives prior to the last menstrual period were excluded from the analysis during use of HT. They were allowed to reenter the study, however, once HT or oral contraceptives were discontinued.
As the cognitive component of SWAN began, the mean age of participants was 50 years, and 8% were premenopausal, 49% were early perimenopausal, 12% were late perimenopausal, and 27% were postmenopausal. In addition, 4% were both postmenopausal and current users of HT.
Because perimenopausal HT should include a dosage of progestin adequate to suppress ovulation in order to prevent iatrogenic irregular uterine bleeding, women who are healthy, lean, nonsmoking, and still having menstrual periods can safely use a conventional oral contraceptive. Options for other symptomatic perimenopausal women include a continuous oral menopausal regimen formulated with 5 μg of ethinyl estradiol and 1 mg of norethindrone acetate (Femhrt 1/5) or 1 mg of estradiol and 0.5 mg of norethindrone acetate (Activella 1/0.5 or generic).—ANDREW M. KAUNITZ, MD
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. For the WHI and WHI-CACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Freeman EW, Sammel MD, Lin H, et al. Symptoms associated with the menopausal transition and reproductive hormones in midlife women. Obstet Gynecol. 2007;110(2 Pt 1):230-240.
4. Woods NF, Smith-DiJulio K, Percival DB, Tao EY, Mariella A, Mitchell S. Depressed mood during the menopausal transition and early postmenopause: observations from the Seattle Midlife Women’s Health Study. Menopause. 2008;15:223-232.
5. Berga SL. Disordered folliculogenesis during the menopausal transition: explaining chaos. Menopause. 2009;16:11-12.
1. Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465-1477.
2. Manson JE, Allison MA, Rossouw JE, et al. For the WHI and WHI-CACS Investigators. Estrogen therapy and coronary-artery calcification. N Engl J Med. 2007;356:2591-2602.
3. Freeman EW, Sammel MD, Lin H, et al. Symptoms associated with the menopausal transition and reproductive hormones in midlife women. Obstet Gynecol. 2007;110(2 Pt 1):230-240.
4. Woods NF, Smith-DiJulio K, Percival DB, Tao EY, Mariella A, Mitchell S. Depressed mood during the menopausal transition and early postmenopause: observations from the Seattle Midlife Women’s Health Study. Menopause. 2008;15:223-232.
5. Berga SL. Disordered folliculogenesis during the menopausal transition: explaining chaos. Menopause. 2009;16:11-12.
Is ovarian Ca screening effective in postmenopausal women?
Ovarian cancer is relatively rare, but late diagnosis leads to a higher death rate than the death rate observed with other gynecologic malignancies. Detection at an early stage (I or II) is associated with higher survival than detection at a later stage (III or IV).
Earlier studies have suggested that ovarian cancer screening with the serum biomarker CA 125 and transvaginal ultrasonography (TVS) may help the clinician diagnose ovarian cancer at an earlier stage.
British study finds screening to be effective
In the United Kingdom Collaborative Trial of Ovarian Cancer Screening, postmenopausal women 50 to 74 years old were randomized to one of the following:
- no screening (n=101,359)
- annual assessment of CA 125 using a proprietary risk-of-cancer algorithm, with TVS as a second-line test (multimodal screening, or MMS) (n=50,078)
- annual TVS in a 2:1:1 ratio (n=48,230). (A scan was considered abnormal when one or both ovaries had complex morphology or a simple cyst exceeded 60 cm3 in size.)
The mean age at screening was 60.6 years, and 96.5% of participants were white.
Women who underwent MMS were triaged according to their estimated risk of developing ovarian cancer, based on the CA 125 level and age-specific risk estimates. Women in the group with the lowest risk of ovarian cancer continued annual assessment of CA 125, whereas those at highest risk underwent repeat measurement of CA 125, followed by TVS if the repeat assay suggested elevated risk. If TVS findings were also abnormal, the patient underwent clinical evaluation.
In the MMS and TVS groups, 8.7% and 12.0% of subjects, respectively, underwent clinical assessment, and surgery was performed in 0.2% and 1.8%, respectively. A primary ovarian or tubal cancer was diagnosed in 42 and 45 women in the MMS and TVS groups, respectively. In addition, 8 (MMS) and 20 (TVS) borderline malignancies were identified.
Overall, 48.3% of invasive cancers were diagnosed during stage I or II, with no difference in the distribution of stages between MMS and TVS groups. The positive predictive value (PPV) of MMS and TVS for primary invasive epithelial and tubal cancers was 35.1% and 2.8%, respectively. The ratio of surgery to case of invasive ovarian cancer was 2.9:1 (MMS) and 35:1 (TVS).
In the US study, women underwent both CA 125 assessment and TVS
The US study involved four annual rounds of screening in women 55 to 74 years old, who underwent both CA 125 measurement and TVS imaging in each round. A CA 125 level of 35 U/mL or above, or ovarian volume greater than 10 cm3 (or detection of an ovarian cyst with complex morphology) was considered abnormal.
Of 34,621 women randomized to screening, 30,630 underwent at least one screen during the four rounds. Almost two thirds of participants were 55 to 64 years old, and almost 89% were non-Hispanic white.
The percentage of women who had at least one positive screen decreased over the four rounds of screening, from 5.8% in year 1 to 4.9%, 4.6%, and 4.5% in years 2 through 4, respectively. In each round of screening, TVS was more likely to be positive than was CA 125 measurement (e.g., 4.6% vs 1.4% in the first round).
Of the 28,746 women who underwent the initial (prevalence) screen, 1,675 (5.8%) had positive findings, 566 (1.97% of those who were screened) underwent surgery, and 27 neoplasms were detected (0.06% of those who were screened), including 18 ovarian or primary peritoneal invasive cancers. Nine borderline tumors were also identified.
The PPV of a positive screen in the first round was 1.1%. Of the 18 invasive cancers identified in this round, 16.7% were diagnosed during stage I or II, and 83.4% were identified during stage III or IV. The ratio of surgery to cases of invasive ovarian cancer was 31.4:1.
Why did the trials have different findings?
The findings of the UK study suggest that an MMS strategy of CA 125 assessment, followed selectively by TVS, can detect early-stage ovarian cancer with an acceptable PPV and ratio of surgery to case of invasive cancer.
In contrast, the US findings are discouraging because of the low PPV, the large percentage of malignancies detected at an advanced stage, and the high ratio of surgery to cases of invasive cancer.
Although the studies had different primary outcomes—ovarian and tubal cancers in the UK and ovarian and primary peritoneal cancers in the US—a majority of invasive malignancies detected in both studies were ovarian.
In the US study, the poor performance of screening may be due, in part, to universal rather than selective use of TVS; that modality generates substantially more false positives than does CA 125. The US study also defined an ovarian abnormality more broadly (volume greater than 10 cm3) than the British study did (volume greater than 60 cm3), which may have lowered the PPV in the US trial.
As investigators in the UK continue to follow participants and report their findings, we will learn more about the value of MMS, including its impact on cancer mortality, possibly as soon as 2014.
Until we have further data on the value of ovarian cancer screening in postmenopausal women, we should continue the current practice of screening only symptomatic or very-high-risk women for ovarian cancer.—ANDREW M. KAUNITZ, MD
Ovarian cancer is relatively rare, but late diagnosis leads to a higher death rate than the death rate observed with other gynecologic malignancies. Detection at an early stage (I or II) is associated with higher survival than detection at a later stage (III or IV).
Earlier studies have suggested that ovarian cancer screening with the serum biomarker CA 125 and transvaginal ultrasonography (TVS) may help the clinician diagnose ovarian cancer at an earlier stage.
British study finds screening to be effective
In the United Kingdom Collaborative Trial of Ovarian Cancer Screening, postmenopausal women 50 to 74 years old were randomized to one of the following:
- no screening (n=101,359)
- annual assessment of CA 125 using a proprietary risk-of-cancer algorithm, with TVS as a second-line test (multimodal screening, or MMS) (n=50,078)
- annual TVS in a 2:1:1 ratio (n=48,230). (A scan was considered abnormal when one or both ovaries had complex morphology or a simple cyst exceeded 60 cm3 in size.)
The mean age at screening was 60.6 years, and 96.5% of participants were white.
Women who underwent MMS were triaged according to their estimated risk of developing ovarian cancer, based on the CA 125 level and age-specific risk estimates. Women in the group with the lowest risk of ovarian cancer continued annual assessment of CA 125, whereas those at highest risk underwent repeat measurement of CA 125, followed by TVS if the repeat assay suggested elevated risk. If TVS findings were also abnormal, the patient underwent clinical evaluation.
In the MMS and TVS groups, 8.7% and 12.0% of subjects, respectively, underwent clinical assessment, and surgery was performed in 0.2% and 1.8%, respectively. A primary ovarian or tubal cancer was diagnosed in 42 and 45 women in the MMS and TVS groups, respectively. In addition, 8 (MMS) and 20 (TVS) borderline malignancies were identified.
Overall, 48.3% of invasive cancers were diagnosed during stage I or II, with no difference in the distribution of stages between MMS and TVS groups. The positive predictive value (PPV) of MMS and TVS for primary invasive epithelial and tubal cancers was 35.1% and 2.8%, respectively. The ratio of surgery to case of invasive ovarian cancer was 2.9:1 (MMS) and 35:1 (TVS).
In the US study, women underwent both CA 125 assessment and TVS
The US study involved four annual rounds of screening in women 55 to 74 years old, who underwent both CA 125 measurement and TVS imaging in each round. A CA 125 level of 35 U/mL or above, or ovarian volume greater than 10 cm3 (or detection of an ovarian cyst with complex morphology) was considered abnormal.
Of 34,621 women randomized to screening, 30,630 underwent at least one screen during the four rounds. Almost two thirds of participants were 55 to 64 years old, and almost 89% were non-Hispanic white.
The percentage of women who had at least one positive screen decreased over the four rounds of screening, from 5.8% in year 1 to 4.9%, 4.6%, and 4.5% in years 2 through 4, respectively. In each round of screening, TVS was more likely to be positive than was CA 125 measurement (e.g., 4.6% vs 1.4% in the first round).
Of the 28,746 women who underwent the initial (prevalence) screen, 1,675 (5.8%) had positive findings, 566 (1.97% of those who were screened) underwent surgery, and 27 neoplasms were detected (0.06% of those who were screened), including 18 ovarian or primary peritoneal invasive cancers. Nine borderline tumors were also identified.
The PPV of a positive screen in the first round was 1.1%. Of the 18 invasive cancers identified in this round, 16.7% were diagnosed during stage I or II, and 83.4% were identified during stage III or IV. The ratio of surgery to cases of invasive ovarian cancer was 31.4:1.
Why did the trials have different findings?
The findings of the UK study suggest that an MMS strategy of CA 125 assessment, followed selectively by TVS, can detect early-stage ovarian cancer with an acceptable PPV and ratio of surgery to case of invasive cancer.
In contrast, the US findings are discouraging because of the low PPV, the large percentage of malignancies detected at an advanced stage, and the high ratio of surgery to cases of invasive cancer.
Although the studies had different primary outcomes—ovarian and tubal cancers in the UK and ovarian and primary peritoneal cancers in the US—a majority of invasive malignancies detected in both studies were ovarian.
In the US study, the poor performance of screening may be due, in part, to universal rather than selective use of TVS; that modality generates substantially more false positives than does CA 125. The US study also defined an ovarian abnormality more broadly (volume greater than 10 cm3) than the British study did (volume greater than 60 cm3), which may have lowered the PPV in the US trial.
As investigators in the UK continue to follow participants and report their findings, we will learn more about the value of MMS, including its impact on cancer mortality, possibly as soon as 2014.
Until we have further data on the value of ovarian cancer screening in postmenopausal women, we should continue the current practice of screening only symptomatic or very-high-risk women for ovarian cancer.—ANDREW M. KAUNITZ, MD
Ovarian cancer is relatively rare, but late diagnosis leads to a higher death rate than the death rate observed with other gynecologic malignancies. Detection at an early stage (I or II) is associated with higher survival than detection at a later stage (III or IV).
Earlier studies have suggested that ovarian cancer screening with the serum biomarker CA 125 and transvaginal ultrasonography (TVS) may help the clinician diagnose ovarian cancer at an earlier stage.
British study finds screening to be effective
In the United Kingdom Collaborative Trial of Ovarian Cancer Screening, postmenopausal women 50 to 74 years old were randomized to one of the following:
- no screening (n=101,359)
- annual assessment of CA 125 using a proprietary risk-of-cancer algorithm, with TVS as a second-line test (multimodal screening, or MMS) (n=50,078)
- annual TVS in a 2:1:1 ratio (n=48,230). (A scan was considered abnormal when one or both ovaries had complex morphology or a simple cyst exceeded 60 cm3 in size.)
The mean age at screening was 60.6 years, and 96.5% of participants were white.
Women who underwent MMS were triaged according to their estimated risk of developing ovarian cancer, based on the CA 125 level and age-specific risk estimates. Women in the group with the lowest risk of ovarian cancer continued annual assessment of CA 125, whereas those at highest risk underwent repeat measurement of CA 125, followed by TVS if the repeat assay suggested elevated risk. If TVS findings were also abnormal, the patient underwent clinical evaluation.
In the MMS and TVS groups, 8.7% and 12.0% of subjects, respectively, underwent clinical assessment, and surgery was performed in 0.2% and 1.8%, respectively. A primary ovarian or tubal cancer was diagnosed in 42 and 45 women in the MMS and TVS groups, respectively. In addition, 8 (MMS) and 20 (TVS) borderline malignancies were identified.
Overall, 48.3% of invasive cancers were diagnosed during stage I or II, with no difference in the distribution of stages between MMS and TVS groups. The positive predictive value (PPV) of MMS and TVS for primary invasive epithelial and tubal cancers was 35.1% and 2.8%, respectively. The ratio of surgery to case of invasive ovarian cancer was 2.9:1 (MMS) and 35:1 (TVS).
In the US study, women underwent both CA 125 assessment and TVS
The US study involved four annual rounds of screening in women 55 to 74 years old, who underwent both CA 125 measurement and TVS imaging in each round. A CA 125 level of 35 U/mL or above, or ovarian volume greater than 10 cm3 (or detection of an ovarian cyst with complex morphology) was considered abnormal.
Of 34,621 women randomized to screening, 30,630 underwent at least one screen during the four rounds. Almost two thirds of participants were 55 to 64 years old, and almost 89% were non-Hispanic white.
The percentage of women who had at least one positive screen decreased over the four rounds of screening, from 5.8% in year 1 to 4.9%, 4.6%, and 4.5% in years 2 through 4, respectively. In each round of screening, TVS was more likely to be positive than was CA 125 measurement (e.g., 4.6% vs 1.4% in the first round).
Of the 28,746 women who underwent the initial (prevalence) screen, 1,675 (5.8%) had positive findings, 566 (1.97% of those who were screened) underwent surgery, and 27 neoplasms were detected (0.06% of those who were screened), including 18 ovarian or primary peritoneal invasive cancers. Nine borderline tumors were also identified.
The PPV of a positive screen in the first round was 1.1%. Of the 18 invasive cancers identified in this round, 16.7% were diagnosed during stage I or II, and 83.4% were identified during stage III or IV. The ratio of surgery to cases of invasive ovarian cancer was 31.4:1.
Why did the trials have different findings?
The findings of the UK study suggest that an MMS strategy of CA 125 assessment, followed selectively by TVS, can detect early-stage ovarian cancer with an acceptable PPV and ratio of surgery to case of invasive cancer.
In contrast, the US findings are discouraging because of the low PPV, the large percentage of malignancies detected at an advanced stage, and the high ratio of surgery to cases of invasive cancer.
Although the studies had different primary outcomes—ovarian and tubal cancers in the UK and ovarian and primary peritoneal cancers in the US—a majority of invasive malignancies detected in both studies were ovarian.
In the US study, the poor performance of screening may be due, in part, to universal rather than selective use of TVS; that modality generates substantially more false positives than does CA 125. The US study also defined an ovarian abnormality more broadly (volume greater than 10 cm3) than the British study did (volume greater than 60 cm3), which may have lowered the PPV in the US trial.
As investigators in the UK continue to follow participants and report their findings, we will learn more about the value of MMS, including its impact on cancer mortality, possibly as soon as 2014.
Until we have further data on the value of ovarian cancer screening in postmenopausal women, we should continue the current practice of screening only symptomatic or very-high-risk women for ovarian cancer.—ANDREW M. KAUNITZ, MD
Should primary cervical cancer screening of women 30 years and older include HPV testing?
- conventional Pap test
- HPV test (Hybrid Capture 2)
- visual inspection of the cervix with acetic acid (VIA)
- no screen (the current standard in India).
Within 8 years of a single HPV test, the incidence of both advanced cervical cancer and cervical cancer mortality declined significantly. In contrast, neither a single Pap test nor a single VIA had a substantial impact on either the incidence of advanced cervical cancer or mortality. Not a single cancer occurred among the 90% of women who tested negative for HPV at enrollment.
Properly timed, the HPV test can avert more advanced cervical cancers and deaths in developing countries than other screening methods
The burden of cervical cancer is shouldered largely by the developing world, where an estimated 80% of the nearly 500,000 cervical cancers occur annually, and where screening has been too costly and complicated to instigate. This study provides the first evidence that a single screen with an HPV test, best performed 15 to 20 years after the median age of first intercourse, would save more lives than other options and may be affordable.
A low-cost, simple, and highly sensitive HPV test (careHPV test) has been developed, with financial backing from the Bill and Melinda Gates Foundation, through the Alliance for Cervical Cancer Prevention. This test provides results within 3 hours and is now being used in demonstration projects in several countries. This appears to offer a way to prevent thousands of deaths from cervical cancer worldwide.
In the United States, cotesting is more effective—and affordable—than the Pap test alone
Castle and associates provide in-depth information on cotesting in women 30 years and older using both the Pap and HPV tests in primary screening in a US setting.
A major concern about cotesting has been that the addition of the HPV test might burden the system with too many positive results, but this study demonstrates otherwise. In the general population of women of this age, the number of HPV-positive results is not burdensome.
The authors point out that, for the 90% of women who tested negative on both the Pap and HPV tests, the risk of incipient precancer or cancer is likely to be very low for the next 10 years or so. Therefore, most women who undergo cotesting could be safely screened at 3-year intervals, and those screened more irregularly would likely be better protected than women screened by cytology alone. Extending the interval makes cotesting more cost-effective and has other benefits, as well.
Rate of HPV-positive findings varied by age
Among women 30 years of age and older, there is some variation in the rate of HPV-positive results. In this study, HPV-positive results were found in:
- 10.8% of women 30 to 34 years old
- 8.0% of women 35 to 39
- 6.3% of women 40 to 44
- 4.9% of women 45 to 49
- 4.3% of women 50 to 54
- 3.9% of women 55 to 59
- 3.7% of women 60 to 69
- 5.3% of women 80 years and older.
Findings are in line with other studies
The study by Castle and colleagues is the largest general-population screening investigation of cotesting published so far. The rate of HPV-positive and Pap-negative findings was 3.99% for the group of women 30 years and older, which is right on target with the 3.7% rate for cotesting demonstrated in the Netherlands and the 4% rate reported in a US survey by the College of American Pathologists. All women of this age in the Kaiser system were given the option of continuing to get an annual Pap or switching to cotesting every 3 years; 91.6% chose the cotesting option.
More and more, data point to cotesting as the screen of choice for women 30 years and older, with the screening interval extended to 3 years for any patient who tests negative on both HPV and Pap tests.—J. THOMAS COX, MD
- conventional Pap test
- HPV test (Hybrid Capture 2)
- visual inspection of the cervix with acetic acid (VIA)
- no screen (the current standard in India).
Within 8 years of a single HPV test, the incidence of both advanced cervical cancer and cervical cancer mortality declined significantly. In contrast, neither a single Pap test nor a single VIA had a substantial impact on either the incidence of advanced cervical cancer or mortality. Not a single cancer occurred among the 90% of women who tested negative for HPV at enrollment.
Properly timed, the HPV test can avert more advanced cervical cancers and deaths in developing countries than other screening methods
The burden of cervical cancer is shouldered largely by the developing world, where an estimated 80% of the nearly 500,000 cervical cancers occur annually, and where screening has been too costly and complicated to instigate. This study provides the first evidence that a single screen with an HPV test, best performed 15 to 20 years after the median age of first intercourse, would save more lives than other options and may be affordable.
A low-cost, simple, and highly sensitive HPV test (careHPV test) has been developed, with financial backing from the Bill and Melinda Gates Foundation, through the Alliance for Cervical Cancer Prevention. This test provides results within 3 hours and is now being used in demonstration projects in several countries. This appears to offer a way to prevent thousands of deaths from cervical cancer worldwide.
In the United States, cotesting is more effective—and affordable—than the Pap test alone
Castle and associates provide in-depth information on cotesting in women 30 years and older using both the Pap and HPV tests in primary screening in a US setting.
A major concern about cotesting has been that the addition of the HPV test might burden the system with too many positive results, but this study demonstrates otherwise. In the general population of women of this age, the number of HPV-positive results is not burdensome.
The authors point out that, for the 90% of women who tested negative on both the Pap and HPV tests, the risk of incipient precancer or cancer is likely to be very low for the next 10 years or so. Therefore, most women who undergo cotesting could be safely screened at 3-year intervals, and those screened more irregularly would likely be better protected than women screened by cytology alone. Extending the interval makes cotesting more cost-effective and has other benefits, as well.
Rate of HPV-positive findings varied by age
Among women 30 years of age and older, there is some variation in the rate of HPV-positive results. In this study, HPV-positive results were found in:
- 10.8% of women 30 to 34 years old
- 8.0% of women 35 to 39
- 6.3% of women 40 to 44
- 4.9% of women 45 to 49
- 4.3% of women 50 to 54
- 3.9% of women 55 to 59
- 3.7% of women 60 to 69
- 5.3% of women 80 years and older.
Findings are in line with other studies
The study by Castle and colleagues is the largest general-population screening investigation of cotesting published so far. The rate of HPV-positive and Pap-negative findings was 3.99% for the group of women 30 years and older, which is right on target with the 3.7% rate for cotesting demonstrated in the Netherlands and the 4% rate reported in a US survey by the College of American Pathologists. All women of this age in the Kaiser system were given the option of continuing to get an annual Pap or switching to cotesting every 3 years; 91.6% chose the cotesting option.
More and more, data point to cotesting as the screen of choice for women 30 years and older, with the screening interval extended to 3 years for any patient who tests negative on both HPV and Pap tests.—J. THOMAS COX, MD
- conventional Pap test
- HPV test (Hybrid Capture 2)
- visual inspection of the cervix with acetic acid (VIA)
- no screen (the current standard in India).
Within 8 years of a single HPV test, the incidence of both advanced cervical cancer and cervical cancer mortality declined significantly. In contrast, neither a single Pap test nor a single VIA had a substantial impact on either the incidence of advanced cervical cancer or mortality. Not a single cancer occurred among the 90% of women who tested negative for HPV at enrollment.
Properly timed, the HPV test can avert more advanced cervical cancers and deaths in developing countries than other screening methods
The burden of cervical cancer is shouldered largely by the developing world, where an estimated 80% of the nearly 500,000 cervical cancers occur annually, and where screening has been too costly and complicated to instigate. This study provides the first evidence that a single screen with an HPV test, best performed 15 to 20 years after the median age of first intercourse, would save more lives than other options and may be affordable.
A low-cost, simple, and highly sensitive HPV test (careHPV test) has been developed, with financial backing from the Bill and Melinda Gates Foundation, through the Alliance for Cervical Cancer Prevention. This test provides results within 3 hours and is now being used in demonstration projects in several countries. This appears to offer a way to prevent thousands of deaths from cervical cancer worldwide.
In the United States, cotesting is more effective—and affordable—than the Pap test alone
Castle and associates provide in-depth information on cotesting in women 30 years and older using both the Pap and HPV tests in primary screening in a US setting.
A major concern about cotesting has been that the addition of the HPV test might burden the system with too many positive results, but this study demonstrates otherwise. In the general population of women of this age, the number of HPV-positive results is not burdensome.
The authors point out that, for the 90% of women who tested negative on both the Pap and HPV tests, the risk of incipient precancer or cancer is likely to be very low for the next 10 years or so. Therefore, most women who undergo cotesting could be safely screened at 3-year intervals, and those screened more irregularly would likely be better protected than women screened by cytology alone. Extending the interval makes cotesting more cost-effective and has other benefits, as well.
Rate of HPV-positive findings varied by age
Among women 30 years of age and older, there is some variation in the rate of HPV-positive results. In this study, HPV-positive results were found in:
- 10.8% of women 30 to 34 years old
- 8.0% of women 35 to 39
- 6.3% of women 40 to 44
- 4.9% of women 45 to 49
- 4.3% of women 50 to 54
- 3.9% of women 55 to 59
- 3.7% of women 60 to 69
- 5.3% of women 80 years and older.
Findings are in line with other studies
The study by Castle and colleagues is the largest general-population screening investigation of cotesting published so far. The rate of HPV-positive and Pap-negative findings was 3.99% for the group of women 30 years and older, which is right on target with the 3.7% rate for cotesting demonstrated in the Netherlands and the 4% rate reported in a US survey by the College of American Pathologists. All women of this age in the Kaiser system were given the option of continuing to get an annual Pap or switching to cotesting every 3 years; 91.6% chose the cotesting option.
More and more, data point to cotesting as the screen of choice for women 30 years and older, with the screening interval extended to 3 years for any patient who tests negative on both HPV and Pap tests.—J. THOMAS COX, MD
What is the 5-year cumulative failure rate of global endometrial ablation?
Abnormal uterine bleeding (AUB) among women of reproductive age has an enormous impact on quality of life and sexual function and consumes many health-care dollars in its evaluation and management.
If a woman has completed childbearing and has a uterus of normal size without intracavitary pathology, options include:
- the levonorgestrel-releasing intrauterine system (Mirena)
- hormonal contraception (both combination and progestin-only)
- nonsteroidal anti-inflammatory drugs
- cyclic progesterone therapy.
If the patient fails, refuses, or has contra-indications to nonsurgical therapy and seeks surgical intervention, endometrial ablation is a viable option. However, she should be informed that she may need additional treatment, resume menstruation, or develop a complication. She also should be apprised of the potential for pregnancy.
If she demands amenorrhea, total hysterectomy is the only option.
Age and other variables were predictors of outcome
The ability to predict outcomes of global endometrial ablation is clinically useful and may help the patient decide between ablation and hysterectomy. In the study by ElNashar and colleagues, women were more likely to achieve amenorrhea if they:
- were 45 years of age or older
- had a uterus shorter than 9 cm
- had endometrium thinner than 4 mm
- underwent radiofrequency ablation.
Women who were more likely to fail:
- were younger than 45 years
- had parity of 5 or higher
- had a history of tubal ligation
- had a history of dysmenorrhea.
The study included 816 women who underwent global endometrial ablation—455 in the model-development arm, and 361 in the validation arm. Three pregnancies occurred (all ended in spontaneous first-trimester abortion), 23 women (5%) complained of pelvic pain, and no patients died or developed endometrial cancer. Overall, 45 women in the model-development arm underwent hysterectomy—28 for persistent bleeding, 12 for persistent pain, and five for other indications.
Study size was a strength
Also valuable was long-term follow-up using an established registry. Among the weaknesses of the study was the fact that only two types of ablation were used.
This study confirms what many people have intuitively believed about endometrial ablation: It rarely causes permanent amenorrhea regardless of the system selected. In the original FDA clinical trials that included three other devices, the amenorrhea rate ranged from 22% to 55%, but patient satisfaction was greater than 90% in all devices studied.
Because most of the women in this study were white, further validation of this model among other races is needed.
When a patient seeks surgical intervention for AUB, assess her expectations. Ask her, “If we can make your periods return to normal or reduce monthly blood flow below normal, would you be happy with the outcome?” If she answers, “Yes,” endometrial ablation is an option. The younger the patient, the greater is the likelihood that additional surgery will eventually be necessary. If, on the other hand, she demands amenorrhea, the only option is hysterectomy with removal of the cervix.
When endometrial ablation is planned, perform preoperative imaging with saline infusion sonography or hysteroscopy to exclude intracavitary pathology, and perform preoperative endometrial biopsy to exclude pre-malignant or malignant disease. In addition, assess preoperative dysmenorrhea closely to avoid ablation in a woman who may have adenomyosis.
Also evaluate women for bleeding diathesis, such as von Willebrand’s disease, prior to ablation.- LINDA D. BRADLEY, MD
Abnormal uterine bleeding (AUB) among women of reproductive age has an enormous impact on quality of life and sexual function and consumes many health-care dollars in its evaluation and management.
If a woman has completed childbearing and has a uterus of normal size without intracavitary pathology, options include:
- the levonorgestrel-releasing intrauterine system (Mirena)
- hormonal contraception (both combination and progestin-only)
- nonsteroidal anti-inflammatory drugs
- cyclic progesterone therapy.
If the patient fails, refuses, or has contra-indications to nonsurgical therapy and seeks surgical intervention, endometrial ablation is a viable option. However, she should be informed that she may need additional treatment, resume menstruation, or develop a complication. She also should be apprised of the potential for pregnancy.
If she demands amenorrhea, total hysterectomy is the only option.
Age and other variables were predictors of outcome
The ability to predict outcomes of global endometrial ablation is clinically useful and may help the patient decide between ablation and hysterectomy. In the study by ElNashar and colleagues, women were more likely to achieve amenorrhea if they:
- were 45 years of age or older
- had a uterus shorter than 9 cm
- had endometrium thinner than 4 mm
- underwent radiofrequency ablation.
Women who were more likely to fail:
- were younger than 45 years
- had parity of 5 or higher
- had a history of tubal ligation
- had a history of dysmenorrhea.
The study included 816 women who underwent global endometrial ablation—455 in the model-development arm, and 361 in the validation arm. Three pregnancies occurred (all ended in spontaneous first-trimester abortion), 23 women (5%) complained of pelvic pain, and no patients died or developed endometrial cancer. Overall, 45 women in the model-development arm underwent hysterectomy—28 for persistent bleeding, 12 for persistent pain, and five for other indications.
Study size was a strength
Also valuable was long-term follow-up using an established registry. Among the weaknesses of the study was the fact that only two types of ablation were used.
This study confirms what many people have intuitively believed about endometrial ablation: It rarely causes permanent amenorrhea regardless of the system selected. In the original FDA clinical trials that included three other devices, the amenorrhea rate ranged from 22% to 55%, but patient satisfaction was greater than 90% in all devices studied.
Because most of the women in this study were white, further validation of this model among other races is needed.
When a patient seeks surgical intervention for AUB, assess her expectations. Ask her, “If we can make your periods return to normal or reduce monthly blood flow below normal, would you be happy with the outcome?” If she answers, “Yes,” endometrial ablation is an option. The younger the patient, the greater is the likelihood that additional surgery will eventually be necessary. If, on the other hand, she demands amenorrhea, the only option is hysterectomy with removal of the cervix.
When endometrial ablation is planned, perform preoperative imaging with saline infusion sonography or hysteroscopy to exclude intracavitary pathology, and perform preoperative endometrial biopsy to exclude pre-malignant or malignant disease. In addition, assess preoperative dysmenorrhea closely to avoid ablation in a woman who may have adenomyosis.
Also evaluate women for bleeding diathesis, such as von Willebrand’s disease, prior to ablation.- LINDA D. BRADLEY, MD
Abnormal uterine bleeding (AUB) among women of reproductive age has an enormous impact on quality of life and sexual function and consumes many health-care dollars in its evaluation and management.
If a woman has completed childbearing and has a uterus of normal size without intracavitary pathology, options include:
- the levonorgestrel-releasing intrauterine system (Mirena)
- hormonal contraception (both combination and progestin-only)
- nonsteroidal anti-inflammatory drugs
- cyclic progesterone therapy.
If the patient fails, refuses, or has contra-indications to nonsurgical therapy and seeks surgical intervention, endometrial ablation is a viable option. However, she should be informed that she may need additional treatment, resume menstruation, or develop a complication. She also should be apprised of the potential for pregnancy.
If she demands amenorrhea, total hysterectomy is the only option.
Age and other variables were predictors of outcome
The ability to predict outcomes of global endometrial ablation is clinically useful and may help the patient decide between ablation and hysterectomy. In the study by ElNashar and colleagues, women were more likely to achieve amenorrhea if they:
- were 45 years of age or older
- had a uterus shorter than 9 cm
- had endometrium thinner than 4 mm
- underwent radiofrequency ablation.
Women who were more likely to fail:
- were younger than 45 years
- had parity of 5 or higher
- had a history of tubal ligation
- had a history of dysmenorrhea.
The study included 816 women who underwent global endometrial ablation—455 in the model-development arm, and 361 in the validation arm. Three pregnancies occurred (all ended in spontaneous first-trimester abortion), 23 women (5%) complained of pelvic pain, and no patients died or developed endometrial cancer. Overall, 45 women in the model-development arm underwent hysterectomy—28 for persistent bleeding, 12 for persistent pain, and five for other indications.
Study size was a strength
Also valuable was long-term follow-up using an established registry. Among the weaknesses of the study was the fact that only two types of ablation were used.
This study confirms what many people have intuitively believed about endometrial ablation: It rarely causes permanent amenorrhea regardless of the system selected. In the original FDA clinical trials that included three other devices, the amenorrhea rate ranged from 22% to 55%, but patient satisfaction was greater than 90% in all devices studied.
Because most of the women in this study were white, further validation of this model among other races is needed.
When a patient seeks surgical intervention for AUB, assess her expectations. Ask her, “If we can make your periods return to normal or reduce monthly blood flow below normal, would you be happy with the outcome?” If she answers, “Yes,” endometrial ablation is an option. The younger the patient, the greater is the likelihood that additional surgery will eventually be necessary. If, on the other hand, she demands amenorrhea, the only option is hysterectomy with removal of the cervix.
When endometrial ablation is planned, perform preoperative imaging with saline infusion sonography or hysteroscopy to exclude intracavitary pathology, and perform preoperative endometrial biopsy to exclude pre-malignant or malignant disease. In addition, assess preoperative dysmenorrhea closely to avoid ablation in a woman who may have adenomyosis.
Also evaluate women for bleeding diathesis, such as von Willebrand’s disease, prior to ablation.- LINDA D. BRADLEY, MD
