User login
What is the optimal timing of elective cesarean delivery at term?
Unless fetal lung maturity has been confirmed, elective cesarean delivery before 39 weeks’ gestation is associated with a higher rate of neonatal respiratory problems. In this observational study, performed at 19 US centers from 1999 to 2002, and funded by the National Institutes of Health, infants of women who underwent elective cesarean delivery at 37 or more weeks’ gestation were assessed for the primary outcome—a composite neonatal outcome that included the occurrence of any of the following:
- death
- respiratory distress syndrome
- transient tachypnea of the newborn
- hypoglycemia
- newborn sepsis
- confirmed seizures
- necrotizing enterocolitis
- hypoxic–ischemic encephalopathy
- cardiopulmonary resuscitation or ventilator support within 24 hours of birth
- umbilical cord–blood arterial pH
- 5-minute Apgar score of 3 or below
- admission to NICU
- hospitalization for 5 or more days.
Of 13,258 elective cesarean deliveries performed at term, 35.8% occurred before 39 completed weeks of gestation (6.3% at 37 weeks, 29.5% at 38 weeks) and 49.1% at 39 weeks. Women who delivered before 39 weeks were more likely to be married, white, and to have initiated prenatal care early.
Compared with infants delivered at 39 weeks, those born at 37 to 38 weeks’ gestation had a greater risk of the primary (composite) outcome. At 37 weeks, the adjusted odds ratio (OR) was 2.1 (95% confidence interval [CI], 1.7–2.5). At 38 weeks, the adjusted OR was 1.5 (1.3–1.7).
The authors estimated that, at 37 weeks’ gestation, postponing elective delivery until 39 weeks might prevent 48% of cases of the primary outcome; this percentage was estimated to be 27% at 38 weeks’ gestation.
Patient preference determines timing in some cases
Cesarean delivery accounts for almost one third of US births, and most women who have had such a delivery opt to repeat it in their next pregnancy. As an editorial accompanying this article points out, women in this study who delivered before 39 weeks were more likely to be private patients and had likely asked their own obstetrician to perform the delivery.1 Obstetricians who wish to promote patient satisfaction are likely to honor such a request, recognizing that waiting until later would increase the likelihood of labor, which would exclude the possibility of an elective procedure.
Limitations of the study
Because this study lacked data about testing for fetal lung maturity, it is unclear whether the higher rate of adverse outcomes with elective cesarean delivery before 39 weeks could be explained by failure to assess for fetal lung maturity.
It also appears that the delay of delivery to 39 weeks or beyond may be associated with an increased risk of stillbirth. In other populations, this risk has been estimated to be roughly 0.5 of every 1,000 births for each advancing week of gestation.
Obstetricians and their patients should weigh the known risks of elective cesarean delivery before 39 weeks’ gestation against the small risk of late stillbirth. At the same time, it is important to factor in the patient’s preferences about when delivery occurs and who performs it.—ANDREW M. KAUNITZ, MD
1. Greene MF. Making small risks even smaller [editorial]. N Engl J Med. 2009;360:183-184.
Unless fetal lung maturity has been confirmed, elective cesarean delivery before 39 weeks’ gestation is associated with a higher rate of neonatal respiratory problems. In this observational study, performed at 19 US centers from 1999 to 2002, and funded by the National Institutes of Health, infants of women who underwent elective cesarean delivery at 37 or more weeks’ gestation were assessed for the primary outcome—a composite neonatal outcome that included the occurrence of any of the following:
- death
- respiratory distress syndrome
- transient tachypnea of the newborn
- hypoglycemia
- newborn sepsis
- confirmed seizures
- necrotizing enterocolitis
- hypoxic–ischemic encephalopathy
- cardiopulmonary resuscitation or ventilator support within 24 hours of birth
- umbilical cord–blood arterial pH
- 5-minute Apgar score of 3 or below
- admission to NICU
- hospitalization for 5 or more days.
Of 13,258 elective cesarean deliveries performed at term, 35.8% occurred before 39 completed weeks of gestation (6.3% at 37 weeks, 29.5% at 38 weeks) and 49.1% at 39 weeks. Women who delivered before 39 weeks were more likely to be married, white, and to have initiated prenatal care early.
Compared with infants delivered at 39 weeks, those born at 37 to 38 weeks’ gestation had a greater risk of the primary (composite) outcome. At 37 weeks, the adjusted odds ratio (OR) was 2.1 (95% confidence interval [CI], 1.7–2.5). At 38 weeks, the adjusted OR was 1.5 (1.3–1.7).
The authors estimated that, at 37 weeks’ gestation, postponing elective delivery until 39 weeks might prevent 48% of cases of the primary outcome; this percentage was estimated to be 27% at 38 weeks’ gestation.
Patient preference determines timing in some cases
Cesarean delivery accounts for almost one third of US births, and most women who have had such a delivery opt to repeat it in their next pregnancy. As an editorial accompanying this article points out, women in this study who delivered before 39 weeks were more likely to be private patients and had likely asked their own obstetrician to perform the delivery.1 Obstetricians who wish to promote patient satisfaction are likely to honor such a request, recognizing that waiting until later would increase the likelihood of labor, which would exclude the possibility of an elective procedure.
Limitations of the study
Because this study lacked data about testing for fetal lung maturity, it is unclear whether the higher rate of adverse outcomes with elective cesarean delivery before 39 weeks could be explained by failure to assess for fetal lung maturity.
It also appears that the delay of delivery to 39 weeks or beyond may be associated with an increased risk of stillbirth. In other populations, this risk has been estimated to be roughly 0.5 of every 1,000 births for each advancing week of gestation.
Obstetricians and their patients should weigh the known risks of elective cesarean delivery before 39 weeks’ gestation against the small risk of late stillbirth. At the same time, it is important to factor in the patient’s preferences about when delivery occurs and who performs it.—ANDREW M. KAUNITZ, MD
Unless fetal lung maturity has been confirmed, elective cesarean delivery before 39 weeks’ gestation is associated with a higher rate of neonatal respiratory problems. In this observational study, performed at 19 US centers from 1999 to 2002, and funded by the National Institutes of Health, infants of women who underwent elective cesarean delivery at 37 or more weeks’ gestation were assessed for the primary outcome—a composite neonatal outcome that included the occurrence of any of the following:
- death
- respiratory distress syndrome
- transient tachypnea of the newborn
- hypoglycemia
- newborn sepsis
- confirmed seizures
- necrotizing enterocolitis
- hypoxic–ischemic encephalopathy
- cardiopulmonary resuscitation or ventilator support within 24 hours of birth
- umbilical cord–blood arterial pH
- 5-minute Apgar score of 3 or below
- admission to NICU
- hospitalization for 5 or more days.
Of 13,258 elective cesarean deliveries performed at term, 35.8% occurred before 39 completed weeks of gestation (6.3% at 37 weeks, 29.5% at 38 weeks) and 49.1% at 39 weeks. Women who delivered before 39 weeks were more likely to be married, white, and to have initiated prenatal care early.
Compared with infants delivered at 39 weeks, those born at 37 to 38 weeks’ gestation had a greater risk of the primary (composite) outcome. At 37 weeks, the adjusted odds ratio (OR) was 2.1 (95% confidence interval [CI], 1.7–2.5). At 38 weeks, the adjusted OR was 1.5 (1.3–1.7).
The authors estimated that, at 37 weeks’ gestation, postponing elective delivery until 39 weeks might prevent 48% of cases of the primary outcome; this percentage was estimated to be 27% at 38 weeks’ gestation.
Patient preference determines timing in some cases
Cesarean delivery accounts for almost one third of US births, and most women who have had such a delivery opt to repeat it in their next pregnancy. As an editorial accompanying this article points out, women in this study who delivered before 39 weeks were more likely to be private patients and had likely asked their own obstetrician to perform the delivery.1 Obstetricians who wish to promote patient satisfaction are likely to honor such a request, recognizing that waiting until later would increase the likelihood of labor, which would exclude the possibility of an elective procedure.
Limitations of the study
Because this study lacked data about testing for fetal lung maturity, it is unclear whether the higher rate of adverse outcomes with elective cesarean delivery before 39 weeks could be explained by failure to assess for fetal lung maturity.
It also appears that the delay of delivery to 39 weeks or beyond may be associated with an increased risk of stillbirth. In other populations, this risk has been estimated to be roughly 0.5 of every 1,000 births for each advancing week of gestation.
Obstetricians and their patients should weigh the known risks of elective cesarean delivery before 39 weeks’ gestation against the small risk of late stillbirth. At the same time, it is important to factor in the patient’s preferences about when delivery occurs and who performs it.—ANDREW M. KAUNITZ, MD
1. Greene MF. Making small risks even smaller [editorial]. N Engl J Med. 2009;360:183-184.
1. Greene MF. Making small risks even smaller [editorial]. N Engl J Med. 2009;360:183-184.
Can a change in practice patterns reduce the number of OB malpractice claims?
Most obstetricians have been sued at some point in their career. The cost of these lawsuits can be measured in both financial and human terms—from the expense of mounting a defense to enormous stress and loss of physician productivity. Fear of litigation also leads to the practice of defensive medicine, driving up the cost of health care and further limiting our procedural options.
Suggestions for reform, such as modifying the tort system and reining in medical experts, have met with limited success. Neither an obstetric practice nor a hospital—let alone an individual obstetrician—can hope to effect these kinds of changes. The best solution for mitigating the cost of obstetric litigation is to make care safer. Safer obstetrics may lower obstetricians’ costs while reducing the financial and emotional toll that injury takes on our patients and their families.
A few caveats
The case series by Clark and colleagues is limited because it does not include a comparison group or address cases that were dropped, defended successfully, or filed despite good care. Nor do the authors fully explain how “substandard” care is defined or the evidence on which this finding is based.
In obstetric liability cases, decades may pass between the delivery of an infant and the delivery of a jury verdict, which limits the applicability of these findings to modern obstetric care.
As a case series, this study is hypothesis-generating. It offers four practice- or hospital-level solutions without proving that they work. The authors’ expert opinion should stimulate analytic research that utilizes a comparison group so that associations can be made.
Despite these weaknesses, the conclusions that:
- most cases were a result of substandard care
- documentation is often lacking or inadequate
- use of oxytocin should be standardized
- obstetric coverage should be 24/7
The blame game can be destructive
We live in a blame-based society, exemplified by the obstetric-litigation environment. The study by Clark and colleagues is a welcome step toward realigning our priorities and lowering costs by eliminating preventable errors.
The authors offer four practical recommendations to lower the number of obstetric liability claims:
- Deliver in a facility with 24-hour in-house obstetric coverage
- Adhere to published high-risk medication protocols, especially for oxytocin, misoprostol, and magnesium sulfate
- Limit vaginal birth after cesarean (VBAC) to spontaneous labors progressing without augmentation and without repetitive moderate or severe variable decelerations
- Use a comprehensive, standardized procedure note in cases of shoulder dystocia.
Although this study does not provide proof, it does suggest that it will cost obstetricians much more if they do not follow these four simple recommendations.—JASON K. BAXTER, MD, MSCP, AND LOUIS WEINSTEIN, MD
Most obstetricians have been sued at some point in their career. The cost of these lawsuits can be measured in both financial and human terms—from the expense of mounting a defense to enormous stress and loss of physician productivity. Fear of litigation also leads to the practice of defensive medicine, driving up the cost of health care and further limiting our procedural options.
Suggestions for reform, such as modifying the tort system and reining in medical experts, have met with limited success. Neither an obstetric practice nor a hospital—let alone an individual obstetrician—can hope to effect these kinds of changes. The best solution for mitigating the cost of obstetric litigation is to make care safer. Safer obstetrics may lower obstetricians’ costs while reducing the financial and emotional toll that injury takes on our patients and their families.
A few caveats
The case series by Clark and colleagues is limited because it does not include a comparison group or address cases that were dropped, defended successfully, or filed despite good care. Nor do the authors fully explain how “substandard” care is defined or the evidence on which this finding is based.
In obstetric liability cases, decades may pass between the delivery of an infant and the delivery of a jury verdict, which limits the applicability of these findings to modern obstetric care.
As a case series, this study is hypothesis-generating. It offers four practice- or hospital-level solutions without proving that they work. The authors’ expert opinion should stimulate analytic research that utilizes a comparison group so that associations can be made.
Despite these weaknesses, the conclusions that:
- most cases were a result of substandard care
- documentation is often lacking or inadequate
- use of oxytocin should be standardized
- obstetric coverage should be 24/7
The blame game can be destructive
We live in a blame-based society, exemplified by the obstetric-litigation environment. The study by Clark and colleagues is a welcome step toward realigning our priorities and lowering costs by eliminating preventable errors.
The authors offer four practical recommendations to lower the number of obstetric liability claims:
- Deliver in a facility with 24-hour in-house obstetric coverage
- Adhere to published high-risk medication protocols, especially for oxytocin, misoprostol, and magnesium sulfate
- Limit vaginal birth after cesarean (VBAC) to spontaneous labors progressing without augmentation and without repetitive moderate or severe variable decelerations
- Use a comprehensive, standardized procedure note in cases of shoulder dystocia.
Although this study does not provide proof, it does suggest that it will cost obstetricians much more if they do not follow these four simple recommendations.—JASON K. BAXTER, MD, MSCP, AND LOUIS WEINSTEIN, MD
Most obstetricians have been sued at some point in their career. The cost of these lawsuits can be measured in both financial and human terms—from the expense of mounting a defense to enormous stress and loss of physician productivity. Fear of litigation also leads to the practice of defensive medicine, driving up the cost of health care and further limiting our procedural options.
Suggestions for reform, such as modifying the tort system and reining in medical experts, have met with limited success. Neither an obstetric practice nor a hospital—let alone an individual obstetrician—can hope to effect these kinds of changes. The best solution for mitigating the cost of obstetric litigation is to make care safer. Safer obstetrics may lower obstetricians’ costs while reducing the financial and emotional toll that injury takes on our patients and their families.
A few caveats
The case series by Clark and colleagues is limited because it does not include a comparison group or address cases that were dropped, defended successfully, or filed despite good care. Nor do the authors fully explain how “substandard” care is defined or the evidence on which this finding is based.
In obstetric liability cases, decades may pass between the delivery of an infant and the delivery of a jury verdict, which limits the applicability of these findings to modern obstetric care.
As a case series, this study is hypothesis-generating. It offers four practice- or hospital-level solutions without proving that they work. The authors’ expert opinion should stimulate analytic research that utilizes a comparison group so that associations can be made.
Despite these weaknesses, the conclusions that:
- most cases were a result of substandard care
- documentation is often lacking or inadequate
- use of oxytocin should be standardized
- obstetric coverage should be 24/7
The blame game can be destructive
We live in a blame-based society, exemplified by the obstetric-litigation environment. The study by Clark and colleagues is a welcome step toward realigning our priorities and lowering costs by eliminating preventable errors.
The authors offer four practical recommendations to lower the number of obstetric liability claims:
- Deliver in a facility with 24-hour in-house obstetric coverage
- Adhere to published high-risk medication protocols, especially for oxytocin, misoprostol, and magnesium sulfate
- Limit vaginal birth after cesarean (VBAC) to spontaneous labors progressing without augmentation and without repetitive moderate or severe variable decelerations
- Use a comprehensive, standardized procedure note in cases of shoulder dystocia.
Although this study does not provide proof, it does suggest that it will cost obstetricians much more if they do not follow these four simple recommendations.—JASON K. BAXTER, MD, MSCP, AND LOUIS WEINSTEIN, MD
What percentage of cerebral palsy cases might be associated with intrapartum asphyxia?
In his editorial, Freeman acknowledges the complexity of the issue and allows that some cases of CP are clearly caused by substandard intrapartum care, but he leaves many essential questions unanswered.1 Similarly, the meta-analysis itself offers multiple explanations of the possible causes of CP but relatively few conclusions that can be applied to any specific case of CP, when the cause in that case is unclear.
Authors focus on cord pH below 7.0
To be sure, an umbilical artery pH of less than 7.0 at birth is concerning, but even at this level of acidosis, results are conflicting. Combining data from multiple studies, the authors concluded that the incidence of significant neonatal neurologic morbidity and mortality was significant among nonanomalous infants who had such a pH level (23.1%). However, the remaining 76.9% of infants were neurologically normal at the time of hospital discharge.
Nor did Apgar scores predict asphyxial complications when cord pH was less than 7.0. However, the combination of an Apgar score of 3 or less and cord pH below 7.0 was a sensitive predictor of serious neonatal morbidity.
Questions abound—but not answers
It was thought that electronic fetal monitoring would eliminate intrapartum stillbirth and reduce the incidence of CP—but neither goal has been achieved. Moreover, the presence of meconium, long associated with nonreassuring fetal status, was found in one study to have no association with CP.2
As for the role of infection, inflammation, and intrapartum fever, Eastman and DeLeon suggested as early as 1955 that intrapartum fever was seven times more likely in mothers of children who were later diagnosed with CP than in mothers of normal children—and intrapartum fever, infection, and neonatal fever remain prime suspects in the CP mystery.3
How does the average clinical obstetrician interpret and use these results? How does our legal system use these results?
It depends. The overwhelming majority of nonanomalous term infants do well. For the few who develop CP, there often is an accepted reason for the diagnosis.
Graham and colleagues conclude that only 14.5% of CP cases are associated with intrapartum asphyxia. The dilemma? For that 14.5%—or even the remaining 85.5%—our ability to determine the true cause of CP in any given case is unreliable. Who or what test can conclusively eliminate intrapartum asphyxia as a medically probable cause?
The answers are disheartening.
- Recognize that prematurity and infection are the leading risk factors for cerebral palsy (CP) in nonanomalous infants.
- Be cognizant of the lack of predictive value—both positive and negative—of current methodologies, such as Apgar score and pH level, in regard to CP.
- Remember that neither the introduction of electronic fetal monitoring nor the increase in the cesarean delivery rate from 5.5% in 1970 to 31.1% in 2006 has appreciably altered the rate of CP in the United States.
- Don’t discount the importance of the first 20 minutes of postnatal life. They are perhaps at least as important as the final 20 minutes of fetal life. When delivering a patient who has a risk factor for CP, do not hesitate to request the presence of a skilled neonatologist for assistance with newborn resuscitation.—JOHN T. REPKE, MD
1. Freeman RK. Medical and legal implications for necessary requirements to diagnose damaging hypoxic–ischemic encephalopathy leading to later cerebral palsy. Am J Obstet Gynecol. 2008;199:585-586.
2. Nelson KB, Grether JK. Potentially asphyxiating conditions and spastic cerebral palsy in infants of normal birth weight. Am J Obstet Gynecol. 1998;179:507-513.
3. Eastman NJ, DeLeon M. The etiology of cerebral palsy. Am J Obstet Gynecol. 1955;69:950-961.
In his editorial, Freeman acknowledges the complexity of the issue and allows that some cases of CP are clearly caused by substandard intrapartum care, but he leaves many essential questions unanswered.1 Similarly, the meta-analysis itself offers multiple explanations of the possible causes of CP but relatively few conclusions that can be applied to any specific case of CP, when the cause in that case is unclear.
Authors focus on cord pH below 7.0
To be sure, an umbilical artery pH of less than 7.0 at birth is concerning, but even at this level of acidosis, results are conflicting. Combining data from multiple studies, the authors concluded that the incidence of significant neonatal neurologic morbidity and mortality was significant among nonanomalous infants who had such a pH level (23.1%). However, the remaining 76.9% of infants were neurologically normal at the time of hospital discharge.
Nor did Apgar scores predict asphyxial complications when cord pH was less than 7.0. However, the combination of an Apgar score of 3 or less and cord pH below 7.0 was a sensitive predictor of serious neonatal morbidity.
Questions abound—but not answers
It was thought that electronic fetal monitoring would eliminate intrapartum stillbirth and reduce the incidence of CP—but neither goal has been achieved. Moreover, the presence of meconium, long associated with nonreassuring fetal status, was found in one study to have no association with CP.2
As for the role of infection, inflammation, and intrapartum fever, Eastman and DeLeon suggested as early as 1955 that intrapartum fever was seven times more likely in mothers of children who were later diagnosed with CP than in mothers of normal children—and intrapartum fever, infection, and neonatal fever remain prime suspects in the CP mystery.3
How does the average clinical obstetrician interpret and use these results? How does our legal system use these results?
It depends. The overwhelming majority of nonanomalous term infants do well. For the few who develop CP, there often is an accepted reason for the diagnosis.
Graham and colleagues conclude that only 14.5% of CP cases are associated with intrapartum asphyxia. The dilemma? For that 14.5%—or even the remaining 85.5%—our ability to determine the true cause of CP in any given case is unreliable. Who or what test can conclusively eliminate intrapartum asphyxia as a medically probable cause?
The answers are disheartening.
- Recognize that prematurity and infection are the leading risk factors for cerebral palsy (CP) in nonanomalous infants.
- Be cognizant of the lack of predictive value—both positive and negative—of current methodologies, such as Apgar score and pH level, in regard to CP.
- Remember that neither the introduction of electronic fetal monitoring nor the increase in the cesarean delivery rate from 5.5% in 1970 to 31.1% in 2006 has appreciably altered the rate of CP in the United States.
- Don’t discount the importance of the first 20 minutes of postnatal life. They are perhaps at least as important as the final 20 minutes of fetal life. When delivering a patient who has a risk factor for CP, do not hesitate to request the presence of a skilled neonatologist for assistance with newborn resuscitation.—JOHN T. REPKE, MD
In his editorial, Freeman acknowledges the complexity of the issue and allows that some cases of CP are clearly caused by substandard intrapartum care, but he leaves many essential questions unanswered.1 Similarly, the meta-analysis itself offers multiple explanations of the possible causes of CP but relatively few conclusions that can be applied to any specific case of CP, when the cause in that case is unclear.
Authors focus on cord pH below 7.0
To be sure, an umbilical artery pH of less than 7.0 at birth is concerning, but even at this level of acidosis, results are conflicting. Combining data from multiple studies, the authors concluded that the incidence of significant neonatal neurologic morbidity and mortality was significant among nonanomalous infants who had such a pH level (23.1%). However, the remaining 76.9% of infants were neurologically normal at the time of hospital discharge.
Nor did Apgar scores predict asphyxial complications when cord pH was less than 7.0. However, the combination of an Apgar score of 3 or less and cord pH below 7.0 was a sensitive predictor of serious neonatal morbidity.
Questions abound—but not answers
It was thought that electronic fetal monitoring would eliminate intrapartum stillbirth and reduce the incidence of CP—but neither goal has been achieved. Moreover, the presence of meconium, long associated with nonreassuring fetal status, was found in one study to have no association with CP.2
As for the role of infection, inflammation, and intrapartum fever, Eastman and DeLeon suggested as early as 1955 that intrapartum fever was seven times more likely in mothers of children who were later diagnosed with CP than in mothers of normal children—and intrapartum fever, infection, and neonatal fever remain prime suspects in the CP mystery.3
How does the average clinical obstetrician interpret and use these results? How does our legal system use these results?
It depends. The overwhelming majority of nonanomalous term infants do well. For the few who develop CP, there often is an accepted reason for the diagnosis.
Graham and colleagues conclude that only 14.5% of CP cases are associated with intrapartum asphyxia. The dilemma? For that 14.5%—or even the remaining 85.5%—our ability to determine the true cause of CP in any given case is unreliable. Who or what test can conclusively eliminate intrapartum asphyxia as a medically probable cause?
The answers are disheartening.
- Recognize that prematurity and infection are the leading risk factors for cerebral palsy (CP) in nonanomalous infants.
- Be cognizant of the lack of predictive value—both positive and negative—of current methodologies, such as Apgar score and pH level, in regard to CP.
- Remember that neither the introduction of electronic fetal monitoring nor the increase in the cesarean delivery rate from 5.5% in 1970 to 31.1% in 2006 has appreciably altered the rate of CP in the United States.
- Don’t discount the importance of the first 20 minutes of postnatal life. They are perhaps at least as important as the final 20 minutes of fetal life. When delivering a patient who has a risk factor for CP, do not hesitate to request the presence of a skilled neonatologist for assistance with newborn resuscitation.—JOHN T. REPKE, MD
1. Freeman RK. Medical and legal implications for necessary requirements to diagnose damaging hypoxic–ischemic encephalopathy leading to later cerebral palsy. Am J Obstet Gynecol. 2008;199:585-586.
2. Nelson KB, Grether JK. Potentially asphyxiating conditions and spastic cerebral palsy in infants of normal birth weight. Am J Obstet Gynecol. 1998;179:507-513.
3. Eastman NJ, DeLeon M. The etiology of cerebral palsy. Am J Obstet Gynecol. 1955;69:950-961.
1. Freeman RK. Medical and legal implications for necessary requirements to diagnose damaging hypoxic–ischemic encephalopathy leading to later cerebral palsy. Am J Obstet Gynecol. 2008;199:585-586.
2. Nelson KB, Grether JK. Potentially asphyxiating conditions and spastic cerebral palsy in infants of normal birth weight. Am J Obstet Gynecol. 1998;179:507-513.
3. Eastman NJ, DeLeon M. The etiology of cerebral palsy. Am J Obstet Gynecol. 1955;69:950-961.
Should cystoscopy be routine at the time of hysterectomy?
The overall rate of ureteral injury at the time of hysterectomy and other gynecologic procedures for benign disease has been estimated at 8.8 injuries for every 1,000 procedures, with the highest rate (17.3/1,000) occurring during laparoscopic hysterectomy with bilateral salpingo-oophorectomy (BSO). The rate of bladder injury is estimated at 16.3 for every 1,000 gynecologic procedures and 29.2 for every 1,000 laparoscopic hysterectomies with BSO.1
Cystoscopy is a low-risk procedure that may be beneficial in surgeries associated with a high rate (1% to 2%) of lower urinary tract injury.2 Early detection of bladder or ureteral injury is preferable to avoid postoperative complications such as fistula formation, loss of renal function, and other complications requiring additional surgery and prolonged hospitalization. Early detection also reduces medicolegal risk.
Ibeanu and associates point out that many gynecologic surgeons do not perform cystoscopy routinely, because of either a lack of training or difficulty obtaining privileges to perform this urologic procedure. They also note that the benefits of cystoscopy clearly outweigh the risks.
Only 25.6% of injuries were detected by visual inspection
Roughly one in four injuries to the bladder and ureter were detected without the aid of cystoscopy; the rest were identified using cystoscopy.
Twenty-four cases of bladder injury (2.9%) and 15 cases of ureteral injury (1.8%) were identified at the time of rigid diagnostic cystoscopy after hysterectomy. The majority (544) of the hysterectomies were abdominal, followed by vaginal hysterectomy (227) and laparoscopically assisted vaginal hysterectomy (61) ( TABLE ).
Most ureteral injuries (80%) occurred at the level of the uterine artery. The ureter is difficult to visualize or palpate once it goes under the uterine artery and courses along the anterior vagina before entry into the urinary bladder.
Ureteral injury also was common at the level of the infundibulopelvic ligament. One patient developed vesicovaginal fistula postoperatively that was missed on initial cystoscopy.
TABLE
Injury rate, by hysterectomy procedure
| Type of procedure | Bladder injury | Ureteral injury |
|---|---|---|
| Total abdominal hysterectomy | 2.3% | 1.7% |
| Total vaginal hysterectomy (alone) | 1.8% | 0.9% |
| Total vaginal hysterectomy (with prolapse procedures) | 2.6% | 1.7% |
| Laparoscopically assisted vaginal hysterectomy | 3.3% | 0 |
Cystoscopy is imperfect, but effective, and its cost is justifiable
Cystoscopy should be performed routinely after any gynecologic procedure associated with a high risk of injury, such as difficult bladder or ureteral dissection. Findings that justify cystoscopy include de novo hematuria and air in the Foley bag during laparoscopy.
Although cystoscopy may not identify all injuries, its benefits likely outweigh any additional cost associated with the procedure when a high rate of injury is likely (greater than, say, 1.5%).3
This study was conducted over 8 years at three academic practices, so it may not be possible to generalize its findings broadly across practitioners.
Perform cystoscopy to verify integrity of the lower urinary tract at the time of hysterectomy for benign disease. Appropriate training to detect and repair injury is required to optimize surgical outcomes.—CHERYL IGLESIA, MD
1. Gilmour DT, Das S, Flowerdew G. Rates of urinary tract injury from gynecologic surgery and the role of intraoperative cystoscopy. Obstet Gynecol. 2006;107:1366-1372.
2. American College of Obstetricians and Gynecologists. ACOG Committee Opinion. Number 372. July 2007. The role of cystourethroscopy in the generalist obstetrician-gynecologist practice. Obstet Gynecol. 2007;110:221–224.
3. Visco AG, Taber KH, Weidner AC, Barber MD, Myers ER. Cost-effectiveness of universal cystoscopy to identify ureteral injury at hysterectomy. Obstet Gynecol. 2001;97:685-692.
The overall rate of ureteral injury at the time of hysterectomy and other gynecologic procedures for benign disease has been estimated at 8.8 injuries for every 1,000 procedures, with the highest rate (17.3/1,000) occurring during laparoscopic hysterectomy with bilateral salpingo-oophorectomy (BSO). The rate of bladder injury is estimated at 16.3 for every 1,000 gynecologic procedures and 29.2 for every 1,000 laparoscopic hysterectomies with BSO.1
Cystoscopy is a low-risk procedure that may be beneficial in surgeries associated with a high rate (1% to 2%) of lower urinary tract injury.2 Early detection of bladder or ureteral injury is preferable to avoid postoperative complications such as fistula formation, loss of renal function, and other complications requiring additional surgery and prolonged hospitalization. Early detection also reduces medicolegal risk.
Ibeanu and associates point out that many gynecologic surgeons do not perform cystoscopy routinely, because of either a lack of training or difficulty obtaining privileges to perform this urologic procedure. They also note that the benefits of cystoscopy clearly outweigh the risks.
Only 25.6% of injuries were detected by visual inspection
Roughly one in four injuries to the bladder and ureter were detected without the aid of cystoscopy; the rest were identified using cystoscopy.
Twenty-four cases of bladder injury (2.9%) and 15 cases of ureteral injury (1.8%) were identified at the time of rigid diagnostic cystoscopy after hysterectomy. The majority (544) of the hysterectomies were abdominal, followed by vaginal hysterectomy (227) and laparoscopically assisted vaginal hysterectomy (61) ( TABLE ).
Most ureteral injuries (80%) occurred at the level of the uterine artery. The ureter is difficult to visualize or palpate once it goes under the uterine artery and courses along the anterior vagina before entry into the urinary bladder.
Ureteral injury also was common at the level of the infundibulopelvic ligament. One patient developed vesicovaginal fistula postoperatively that was missed on initial cystoscopy.
TABLE
Injury rate, by hysterectomy procedure
| Type of procedure | Bladder injury | Ureteral injury |
|---|---|---|
| Total abdominal hysterectomy | 2.3% | 1.7% |
| Total vaginal hysterectomy (alone) | 1.8% | 0.9% |
| Total vaginal hysterectomy (with prolapse procedures) | 2.6% | 1.7% |
| Laparoscopically assisted vaginal hysterectomy | 3.3% | 0 |
Cystoscopy is imperfect, but effective, and its cost is justifiable
Cystoscopy should be performed routinely after any gynecologic procedure associated with a high risk of injury, such as difficult bladder or ureteral dissection. Findings that justify cystoscopy include de novo hematuria and air in the Foley bag during laparoscopy.
Although cystoscopy may not identify all injuries, its benefits likely outweigh any additional cost associated with the procedure when a high rate of injury is likely (greater than, say, 1.5%).3
This study was conducted over 8 years at three academic practices, so it may not be possible to generalize its findings broadly across practitioners.
Perform cystoscopy to verify integrity of the lower urinary tract at the time of hysterectomy for benign disease. Appropriate training to detect and repair injury is required to optimize surgical outcomes.—CHERYL IGLESIA, MD
The overall rate of ureteral injury at the time of hysterectomy and other gynecologic procedures for benign disease has been estimated at 8.8 injuries for every 1,000 procedures, with the highest rate (17.3/1,000) occurring during laparoscopic hysterectomy with bilateral salpingo-oophorectomy (BSO). The rate of bladder injury is estimated at 16.3 for every 1,000 gynecologic procedures and 29.2 for every 1,000 laparoscopic hysterectomies with BSO.1
Cystoscopy is a low-risk procedure that may be beneficial in surgeries associated with a high rate (1% to 2%) of lower urinary tract injury.2 Early detection of bladder or ureteral injury is preferable to avoid postoperative complications such as fistula formation, loss of renal function, and other complications requiring additional surgery and prolonged hospitalization. Early detection also reduces medicolegal risk.
Ibeanu and associates point out that many gynecologic surgeons do not perform cystoscopy routinely, because of either a lack of training or difficulty obtaining privileges to perform this urologic procedure. They also note that the benefits of cystoscopy clearly outweigh the risks.
Only 25.6% of injuries were detected by visual inspection
Roughly one in four injuries to the bladder and ureter were detected without the aid of cystoscopy; the rest were identified using cystoscopy.
Twenty-four cases of bladder injury (2.9%) and 15 cases of ureteral injury (1.8%) were identified at the time of rigid diagnostic cystoscopy after hysterectomy. The majority (544) of the hysterectomies were abdominal, followed by vaginal hysterectomy (227) and laparoscopically assisted vaginal hysterectomy (61) ( TABLE ).
Most ureteral injuries (80%) occurred at the level of the uterine artery. The ureter is difficult to visualize or palpate once it goes under the uterine artery and courses along the anterior vagina before entry into the urinary bladder.
Ureteral injury also was common at the level of the infundibulopelvic ligament. One patient developed vesicovaginal fistula postoperatively that was missed on initial cystoscopy.
TABLE
Injury rate, by hysterectomy procedure
| Type of procedure | Bladder injury | Ureteral injury |
|---|---|---|
| Total abdominal hysterectomy | 2.3% | 1.7% |
| Total vaginal hysterectomy (alone) | 1.8% | 0.9% |
| Total vaginal hysterectomy (with prolapse procedures) | 2.6% | 1.7% |
| Laparoscopically assisted vaginal hysterectomy | 3.3% | 0 |
Cystoscopy is imperfect, but effective, and its cost is justifiable
Cystoscopy should be performed routinely after any gynecologic procedure associated with a high risk of injury, such as difficult bladder or ureteral dissection. Findings that justify cystoscopy include de novo hematuria and air in the Foley bag during laparoscopy.
Although cystoscopy may not identify all injuries, its benefits likely outweigh any additional cost associated with the procedure when a high rate of injury is likely (greater than, say, 1.5%).3
This study was conducted over 8 years at three academic practices, so it may not be possible to generalize its findings broadly across practitioners.
Perform cystoscopy to verify integrity of the lower urinary tract at the time of hysterectomy for benign disease. Appropriate training to detect and repair injury is required to optimize surgical outcomes.—CHERYL IGLESIA, MD
1. Gilmour DT, Das S, Flowerdew G. Rates of urinary tract injury from gynecologic surgery and the role of intraoperative cystoscopy. Obstet Gynecol. 2006;107:1366-1372.
2. American College of Obstetricians and Gynecologists. ACOG Committee Opinion. Number 372. July 2007. The role of cystourethroscopy in the generalist obstetrician-gynecologist practice. Obstet Gynecol. 2007;110:221–224.
3. Visco AG, Taber KH, Weidner AC, Barber MD, Myers ER. Cost-effectiveness of universal cystoscopy to identify ureteral injury at hysterectomy. Obstet Gynecol. 2001;97:685-692.
1. Gilmour DT, Das S, Flowerdew G. Rates of urinary tract injury from gynecologic surgery and the role of intraoperative cystoscopy. Obstet Gynecol. 2006;107:1366-1372.
2. American College of Obstetricians and Gynecologists. ACOG Committee Opinion. Number 372. July 2007. The role of cystourethroscopy in the generalist obstetrician-gynecologist practice. Obstet Gynecol. 2007;110:221–224.
3. Visco AG, Taber KH, Weidner AC, Barber MD, Myers ER. Cost-effectiveness of universal cystoscopy to identify ureteral injury at hysterectomy. Obstet Gynecol. 2001;97:685-692.
Are new tools for correcting prolapse and incontinence better just because they’re new?
From my vantage point, it appears that economic factors are playing an increasingly important role in how pelvic organ prolapse (POP) and urinary incontinence (UI) are managed—particularly, in regard to the use of surgical devices. As such, the topic of treating POP and UI deserves our attention to ensure that we make the best decisions for our patients.
Now, I’m a staunch supporter of innovation in treatment; certainly, there is room for improvement in current approaches—particularly in surgery—for treating POP and UI. At the same time, I strongly believe that innovation must be demonstrated to be an improvement before it is incorporated into practice. Although innovation is commonly taken on faith, we should know better than to equate “new” with “better” until evidence, gathered through clinical research, has demonstrated this conclusively. A look at the US Food and Drug Administration’s (FDA’s) process for clearing medical devices for clinical use reveals that such a standard often doesn’t apply—and this should matter to us.
The meaning of 510(k)
Most medical devices are evaluated through an FDA clearance mechanism known as the 510(k) process. This is wholly distinct from the agency’s drug approval process with which most of us are familiar. It’s beyond the scope of this commentary for me to go into detail about 510(k); if you are interested, see two recent commentaries1,2 and visit http://www.fda.gov/cdrh/devadvice/314.html .
In a nutshell, the 510(k) process requires only that an applicant demonstrate that a new medical device has “substantial equivalence” to an already legally marketed device, known as the predicate, which may also have been cleared only through the 510(k) process. That means it’s possible to have generations of products cleared on the basis of one predicate device that was itself never studied adequately.
Indeed, this is the case with most medical devices that have been sold for the surgical treatment of POP and UI—from before the ProteGen Sling (Boston Scientific), through Tension-Free Vaginal Tape (TVT) (Gynecare), and continuing with the newest devices.
The story of the ProteGen Sling ( FIGURE ) offers a cautionary tale about what can go wrong when new devices are cleared by the FDA through 510(k), rather than evaluated through rigorous clinical trials, as drugs are. More recently, experience with the ObTape (Mentor Corporation) followed virtually the same trajectory of events; the product was pulled from the market in 2006 and is now the focus of lawsuits nationwide.
Fortunately, for our patients, experience with TVT (Gynecare) has been favorable. Although TVT was also cleared by the FDA through 510(k), clinical research performed after TVT was introduced has demonstrated its effectiveness and relative safety. Indeed, TVT has revolutionized the treatment of stress UI in women—and, even, our understanding of its etiology.
Several companies are capitalizing on the success of TVT by introducing competing products that are designed to be 1) similar enough to ride on the coattails of TVT yet 2) different enough to capture their own share of market—without evidence of safety or effectiveness required. Even Gynecare (part of Ethicon Women’s Health and Urology, a subsidiary of Johnson & Johnson) has introduced TVT SECUR to compete with its own TVT—again, without independent evidence of safety or effectiveness.
The current market in devices for stress UI is a moving target that makes it nearly impossible—even for research organizations, such as the federally funded Pelvic Floor Disorders Network, that are independent of industry—to develop and implement sound clinical trials of those devices. Why do I say “moving target”? First, there are no assurances that any device chosen for study will remain the same for the duration of a trial. Second, there is no way to foresee which products will be abandoned over the time required for a large clinical trial.
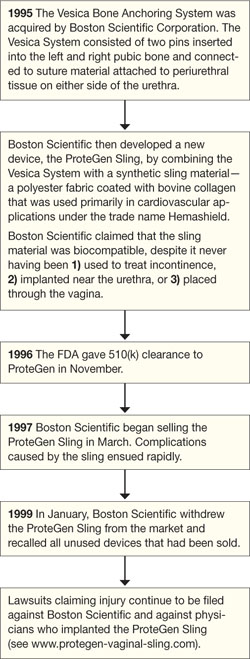
FIGURE The saga of the ProteGen Sling
Transparency over what might be considered “experimental”
Until the FDA changes its process—to one in which 1) medical devices are adequately assessed before they reach market and 2) postmarketing surveillance is required—it’s our duty to insist on evidence of safety and effectiveness before adopting the latest and greatest products that companies have to offer.
Of all the questions that a patient might ask before treatment, three of the most important, surely, are:
- “Will this help me?”
- “If it helps me, how long will it help?”
- “Whether or not this treatment helps me, what risks—in the short-term and over the long-term—does it present?”
Until we can provide our patients with answers that are supported by evidence, products that lack such evidence should be considered experimental, and patients should be counseled accordingly.
Some patients may accept what they’ve been advised are new and unproven treatments—in the way that some physicians are early adopters. Nevertheless, I am concerned that some clinicians do not appear to appreciate the true lack of evidence that accompanies most marketed devices for prolapse and incontinence. They may mistake the FDA 510(k) process of clearance for something similar to the agency’s extended and complex drug approval process. They may accept claims made in industry-produced white papers that are often largely promotional materials, and fail to look further into those claims.
Now, more than ever and above all else, we must stand between marketing and our patients’ safety. We are familiar with the toll that prolapse and incontinence, as chronic conditions, take on our patients; yet it’s that very chronic nature that should lead us to adopt patience and caution in accepting new treatments before they have been adequately studied.
If we cannot always rely on industry to provide clear information about the risks and benefits of new devices, neither can we routinely look to our professional organizations for unbiased information. Often, professional organizations accept cash contributions from industry, raising the question of conflict of interest that may undermine their actions when the priorities of industry do not align with the goal of safeguarding patients’ well-being.
In an unprecedented example of how a professional association can interfere with its own, expert-authored clinical practice guidelines, the American College of Obstetricians and Gynecologists (ACOG) more than a year ago rescinded one of its published guidelines on POP (Issue 79, February 20073 ) and replaced it with a new guideline (Issue 85, September 20074 ). The new guideline is nearly identical to the prior one—save for one sentence, in which “experimental” is deleted in a discussion of kits of trocar-based synthetic materials sold for the surgical treatment of pelvic organ prolapse (see the EXCERPT ).
The deletion is crucial because offering informed consent for surgery requires a patient to accept risks in balance with an expectation of benefit. A patient cannot be appropriately informed when no evidence of benefit exists and evidence of postoperative risk is extremely limited.
Now, I am not declaring that ACOG acted with bias because of a financial conflict of interest with industry in this instance; the fact that a financial conflict of interest exists for ACOG, however, cannot be disputed if one examines the College’s Annual Report, where contributors are listed. (For a comprehensive, if disillusioning, treatise on the many effects of financial conflict of interest within medicine, I recommend the book On The Take.5 )
Differences between the two bulletins are marked in boldface
Bulletin #79 (original wording): “Given the limited data and frequent changes in the marketed products (particularly with regard to type of mesh material itself, which is most closely associated with several of the postoperative risks especially mesh erosion), if clinicians recommend these procedures before evidence of their risk-benefit is fully understood, the procedures should be considered experimental and patients consented for surgery with that understanding.”
Bulletin #84 (revised wording): “Given the limited data and frequent changes in the marketed products for vaginal surgery for prolapse repair (particularly with regard to type of mesh material itself, which is most closely associated with several of the postoperative risks especially mesh erosion), patients should consent to surgery with an understanding of the post-operative risks and lack of long-term outcomes data.”
Case: Radiofrequency therapy. Even when clinical experience demonstrates lack of effectiveness or an unacceptable rate of complications associated with certain techniques or devices, unequivocal evidence of such problems does not always appear in the literature. One example of this is how a technique was translated into a treatment for incontinence by way of its use in other fields.
Radiofrequency has, among other uses, been used to ablate nerves in intractable chronic pain and to address joint instability in orthopedic surgery. Radiofrequency energy was then, by extension, applied transvaginally to tissue (known as “endopelvic” fascia, of a distinctly different nature than parietal fascia involved in orthopedic procedures) surrounding the urethra. The goal was to coagulate “supporting” tissues and “correct” urethral hypermobility that purportedly causes stress incontinence.
Marketing of the SURx Transvaginal System (CooperSurgical, Inc.) began in 2002, followed by reports of success. One industry-sponsored study, for example, reported a 73% rate of either continence or improvement after 12 months.6
Despite such favorable early results, however, in 2006 CooperSurgical decided to abandon this system, citing “technique-dependent” results of the procedure. Since then, independent research has shown a very low initial success rate that declines rapidly—within weeks or months—of treatment.7,8
A different radiofrequency technique continues to be marketed—the Renessa System™ (NovaSys Medical), which uses a urethral catheter-mounted system to deliver radiofrequency energy through the urethral mucosa to the submucosa and adjacent tissues. Once again, initial reports of industry-sponsored research showed promising results; one study of 110 patients reported 74% achieved continence or improvement after 1 year.9 In a follow-up report of 21 of the original 110 patients, “improvement” was reported in 74% after 3 years.10 Independent research has yet to be reported in the literature.
Of particular concern, no data exist on the long-term effect of denaturing collagen in the urethra and adjacent tissues in relation to UI, other aspects of bladder function, or sexual function. An apparent lack of immediate complications cannot be equated with safety; we need long-term studies to determine whether urethral function is affected adversely compared with that in untreated women and women treated with surgery.
Bring on innovation—in context!
For those who consider my argument anti-innovation, let me repeat: I believe strongly in innovation to improve care for our patients. Am I anti-industry? Only when there is an unbridled race to profit from marketing products without safeguards to ensure, first and foremost, the safety of our patients and, second, their long-term effectiveness. Knowingly or unknowingly, patients then become the guinea pigs on whom these products are tested—just not in the appropriate context of clinical research and informed consent for participation.
Instead (as happened in the US Public Health Service’s Tuskegee syphilis experiments), patients serve as research subjects without their consent when they receive untested products and undergo unproven treatments. And because clinicians are the conduit through which patients receive untested and unproven treatments, who is ultimately responsible for the outcome?
Industry brings innovation to clinical practice. But it is incumbent on clinicians to recognize, with unflinching honesty, the bottom line on which industry operates. Prolapse and incontinence are deeply distressing for our patients, but these chronic conditions are not life-threatening; virtually all women who suffer these conditions have been symptomatic for years before they come for care. I see no need, except to increase that bottom line, to rush products to market before they have been evaluated sufficiently to determine whether “new” is actually “better.”
For clinicians who style themselves as early adopters, remember: It’s not you, but your patient, who is “adopting” a foreign material and having it placed deep in the most intimate area of her body—a foreign material intended to stay for life (except for those unfortunate patients who must have it removed). Above all, we must do no harm—an elusive goal when some of us try to attract patients by being the first to use a product before evidence of its risk and utility have been established in practice.
Does this kind of talk encourage litigation?
Does a commentary like this one provide fodder for plaintiff attorneys who are seeking grounds for product liability lawsuits against manufacturers and malpractice claims against clinicians? Please! Spend a moment on the Web, and you will see that the lawyers are already busy—especially since the FDA’s October 2008 alert about complications with surgical mesh for prolapse and incontinence. [See “FDA alert: Transvaginal placement of surgical mesh carries serious risks,” in the December 2008 issue of OBG Management, at www.obgmanagement.com.] It’s worth noting how these lawyers see themselves: They would likely tell you that they “provide an important service in protecting patients from unscrupulous manufacturers who profit from the vulnerability of people seeking treatment for distressing conditions.” As clinicians, are we absolutely sure that we can say the same of ourselves?
Is it wrong to harp on what happened in the past?
Why revisit events surrounding, for example, the ProteGen Sling? My reply is another question: Where is the evidence that such sequences of events are in the past? Among clinicians, who knows which is best in a collection of kits that changes from one month to the next, without their promoters skipping a beat in proclaiming theirs as the “best”? It isn’t shameful to admit that one doesn’t know which one is best; but it is a shame to act as if one does know, especially when the risk falls to another. The names change; the events are the same.
What is the solution to this problem?
Businesses succeed only when their products are purchased. If clinicians refused to be participants whenever the device industry introduces unproven treatments into the market, industry would be compelled to test their products beforehand. Patients would benefit—by being able to make truly informed choices, with adequate information about risk and benefit. Clinicians would benefit—by being able to provide the most effective treatment without sacrificing their integrity in the process. Ultimately, industry would benefit, by profiting appropriately from products that truly help our patients. Is that an impossible wish?
1. Goldman HB. Is new always better? Curr Urol Rep. 2007;8(4):253-254.
2. Ostergard DR. Lessons from the past: directions for the future. Do new marketed surgical procedures and grafts produce ethical, personal liability, and legal concerns for physicians? Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:591-598.
3. ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 79: Pelvic organ prolapse. Obstet Gynecol. 2007;109(2 Pt 1):461-473.
4. ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 85: Pelvic organ prolapse. Obstet Gynecol. 2007;110:717-729.
5. Kassirer JP. On the Take: How Medicine’s Complicity with Big Business Can Endanger Your Health. New York: Oxford University Press; 2005.
6. Dmochowski RR, Avon M, Ross J, et al. Transvaginal radiofrequency treatment of the endopelvic fascia: a prospective evaluation for the treatment of genuine stress urinary incontinence. J Urol. 2003;169:1028-1032.
7. Buchsbaum GM, McConville J, Korni R, Duecy EE. Outcome of transvaginal radiofrequency for treatment of women with stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18(3):263-265.
8. Ismail SI. Radiofrequency remodelling of the endopelvic fascia is not an effective procedure for urodynamic stress incontinence in women. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1205-1209.
9. Appell RA, Juma S, Wells WG, et al. Transurethral radio-frequency energy collagen micro-remodeling for the treatment of female stress urinary incontinence. Neurourol Urodyn. 2006;25(4):331-336.
10. Appell RA, Singh G, Klimberg IW, et al. Nonsurgical radiofrequency collagen denaturation for stress urinary incontinence: retrospective 3-year evaluation. Expert Rev Med Devices. 2007;4:455-461.
From my vantage point, it appears that economic factors are playing an increasingly important role in how pelvic organ prolapse (POP) and urinary incontinence (UI) are managed—particularly, in regard to the use of surgical devices. As such, the topic of treating POP and UI deserves our attention to ensure that we make the best decisions for our patients.
Now, I’m a staunch supporter of innovation in treatment; certainly, there is room for improvement in current approaches—particularly in surgery—for treating POP and UI. At the same time, I strongly believe that innovation must be demonstrated to be an improvement before it is incorporated into practice. Although innovation is commonly taken on faith, we should know better than to equate “new” with “better” until evidence, gathered through clinical research, has demonstrated this conclusively. A look at the US Food and Drug Administration’s (FDA’s) process for clearing medical devices for clinical use reveals that such a standard often doesn’t apply—and this should matter to us.
The meaning of 510(k)
Most medical devices are evaluated through an FDA clearance mechanism known as the 510(k) process. This is wholly distinct from the agency’s drug approval process with which most of us are familiar. It’s beyond the scope of this commentary for me to go into detail about 510(k); if you are interested, see two recent commentaries1,2 and visit http://www.fda.gov/cdrh/devadvice/314.html .
In a nutshell, the 510(k) process requires only that an applicant demonstrate that a new medical device has “substantial equivalence” to an already legally marketed device, known as the predicate, which may also have been cleared only through the 510(k) process. That means it’s possible to have generations of products cleared on the basis of one predicate device that was itself never studied adequately.
Indeed, this is the case with most medical devices that have been sold for the surgical treatment of POP and UI—from before the ProteGen Sling (Boston Scientific), through Tension-Free Vaginal Tape (TVT) (Gynecare), and continuing with the newest devices.
The story of the ProteGen Sling ( FIGURE ) offers a cautionary tale about what can go wrong when new devices are cleared by the FDA through 510(k), rather than evaluated through rigorous clinical trials, as drugs are. More recently, experience with the ObTape (Mentor Corporation) followed virtually the same trajectory of events; the product was pulled from the market in 2006 and is now the focus of lawsuits nationwide.
Fortunately, for our patients, experience with TVT (Gynecare) has been favorable. Although TVT was also cleared by the FDA through 510(k), clinical research performed after TVT was introduced has demonstrated its effectiveness and relative safety. Indeed, TVT has revolutionized the treatment of stress UI in women—and, even, our understanding of its etiology.
Several companies are capitalizing on the success of TVT by introducing competing products that are designed to be 1) similar enough to ride on the coattails of TVT yet 2) different enough to capture their own share of market—without evidence of safety or effectiveness required. Even Gynecare (part of Ethicon Women’s Health and Urology, a subsidiary of Johnson & Johnson) has introduced TVT SECUR to compete with its own TVT—again, without independent evidence of safety or effectiveness.
The current market in devices for stress UI is a moving target that makes it nearly impossible—even for research organizations, such as the federally funded Pelvic Floor Disorders Network, that are independent of industry—to develop and implement sound clinical trials of those devices. Why do I say “moving target”? First, there are no assurances that any device chosen for study will remain the same for the duration of a trial. Second, there is no way to foresee which products will be abandoned over the time required for a large clinical trial.

FIGURE The saga of the ProteGen Sling
Transparency over what might be considered “experimental”
Until the FDA changes its process—to one in which 1) medical devices are adequately assessed before they reach market and 2) postmarketing surveillance is required—it’s our duty to insist on evidence of safety and effectiveness before adopting the latest and greatest products that companies have to offer.
Of all the questions that a patient might ask before treatment, three of the most important, surely, are:
- “Will this help me?”
- “If it helps me, how long will it help?”
- “Whether or not this treatment helps me, what risks—in the short-term and over the long-term—does it present?”
Until we can provide our patients with answers that are supported by evidence, products that lack such evidence should be considered experimental, and patients should be counseled accordingly.
Some patients may accept what they’ve been advised are new and unproven treatments—in the way that some physicians are early adopters. Nevertheless, I am concerned that some clinicians do not appear to appreciate the true lack of evidence that accompanies most marketed devices for prolapse and incontinence. They may mistake the FDA 510(k) process of clearance for something similar to the agency’s extended and complex drug approval process. They may accept claims made in industry-produced white papers that are often largely promotional materials, and fail to look further into those claims.
Now, more than ever and above all else, we must stand between marketing and our patients’ safety. We are familiar with the toll that prolapse and incontinence, as chronic conditions, take on our patients; yet it’s that very chronic nature that should lead us to adopt patience and caution in accepting new treatments before they have been adequately studied.
If we cannot always rely on industry to provide clear information about the risks and benefits of new devices, neither can we routinely look to our professional organizations for unbiased information. Often, professional organizations accept cash contributions from industry, raising the question of conflict of interest that may undermine their actions when the priorities of industry do not align with the goal of safeguarding patients’ well-being.
In an unprecedented example of how a professional association can interfere with its own, expert-authored clinical practice guidelines, the American College of Obstetricians and Gynecologists (ACOG) more than a year ago rescinded one of its published guidelines on POP (Issue 79, February 20073 ) and replaced it with a new guideline (Issue 85, September 20074 ). The new guideline is nearly identical to the prior one—save for one sentence, in which “experimental” is deleted in a discussion of kits of trocar-based synthetic materials sold for the surgical treatment of pelvic organ prolapse (see the EXCERPT ).
The deletion is crucial because offering informed consent for surgery requires a patient to accept risks in balance with an expectation of benefit. A patient cannot be appropriately informed when no evidence of benefit exists and evidence of postoperative risk is extremely limited.
Now, I am not declaring that ACOG acted with bias because of a financial conflict of interest with industry in this instance; the fact that a financial conflict of interest exists for ACOG, however, cannot be disputed if one examines the College’s Annual Report, where contributors are listed. (For a comprehensive, if disillusioning, treatise on the many effects of financial conflict of interest within medicine, I recommend the book On The Take.5 )
Differences between the two bulletins are marked in boldface
Bulletin #79 (original wording): “Given the limited data and frequent changes in the marketed products (particularly with regard to type of mesh material itself, which is most closely associated with several of the postoperative risks especially mesh erosion), if clinicians recommend these procedures before evidence of their risk-benefit is fully understood, the procedures should be considered experimental and patients consented for surgery with that understanding.”
Bulletin #84 (revised wording): “Given the limited data and frequent changes in the marketed products for vaginal surgery for prolapse repair (particularly with regard to type of mesh material itself, which is most closely associated with several of the postoperative risks especially mesh erosion), patients should consent to surgery with an understanding of the post-operative risks and lack of long-term outcomes data.”
Case: Radiofrequency therapy. Even when clinical experience demonstrates lack of effectiveness or an unacceptable rate of complications associated with certain techniques or devices, unequivocal evidence of such problems does not always appear in the literature. One example of this is how a technique was translated into a treatment for incontinence by way of its use in other fields.
Radiofrequency has, among other uses, been used to ablate nerves in intractable chronic pain and to address joint instability in orthopedic surgery. Radiofrequency energy was then, by extension, applied transvaginally to tissue (known as “endopelvic” fascia, of a distinctly different nature than parietal fascia involved in orthopedic procedures) surrounding the urethra. The goal was to coagulate “supporting” tissues and “correct” urethral hypermobility that purportedly causes stress incontinence.
Marketing of the SURx Transvaginal System (CooperSurgical, Inc.) began in 2002, followed by reports of success. One industry-sponsored study, for example, reported a 73% rate of either continence or improvement after 12 months.6
Despite such favorable early results, however, in 2006 CooperSurgical decided to abandon this system, citing “technique-dependent” results of the procedure. Since then, independent research has shown a very low initial success rate that declines rapidly—within weeks or months—of treatment.7,8
A different radiofrequency technique continues to be marketed—the Renessa System™ (NovaSys Medical), which uses a urethral catheter-mounted system to deliver radiofrequency energy through the urethral mucosa to the submucosa and adjacent tissues. Once again, initial reports of industry-sponsored research showed promising results; one study of 110 patients reported 74% achieved continence or improvement after 1 year.9 In a follow-up report of 21 of the original 110 patients, “improvement” was reported in 74% after 3 years.10 Independent research has yet to be reported in the literature.
Of particular concern, no data exist on the long-term effect of denaturing collagen in the urethra and adjacent tissues in relation to UI, other aspects of bladder function, or sexual function. An apparent lack of immediate complications cannot be equated with safety; we need long-term studies to determine whether urethral function is affected adversely compared with that in untreated women and women treated with surgery.
Bring on innovation—in context!
For those who consider my argument anti-innovation, let me repeat: I believe strongly in innovation to improve care for our patients. Am I anti-industry? Only when there is an unbridled race to profit from marketing products without safeguards to ensure, first and foremost, the safety of our patients and, second, their long-term effectiveness. Knowingly or unknowingly, patients then become the guinea pigs on whom these products are tested—just not in the appropriate context of clinical research and informed consent for participation.
Instead (as happened in the US Public Health Service’s Tuskegee syphilis experiments), patients serve as research subjects without their consent when they receive untested products and undergo unproven treatments. And because clinicians are the conduit through which patients receive untested and unproven treatments, who is ultimately responsible for the outcome?
Industry brings innovation to clinical practice. But it is incumbent on clinicians to recognize, with unflinching honesty, the bottom line on which industry operates. Prolapse and incontinence are deeply distressing for our patients, but these chronic conditions are not life-threatening; virtually all women who suffer these conditions have been symptomatic for years before they come for care. I see no need, except to increase that bottom line, to rush products to market before they have been evaluated sufficiently to determine whether “new” is actually “better.”
For clinicians who style themselves as early adopters, remember: It’s not you, but your patient, who is “adopting” a foreign material and having it placed deep in the most intimate area of her body—a foreign material intended to stay for life (except for those unfortunate patients who must have it removed). Above all, we must do no harm—an elusive goal when some of us try to attract patients by being the first to use a product before evidence of its risk and utility have been established in practice.
Does this kind of talk encourage litigation?
Does a commentary like this one provide fodder for plaintiff attorneys who are seeking grounds for product liability lawsuits against manufacturers and malpractice claims against clinicians? Please! Spend a moment on the Web, and you will see that the lawyers are already busy—especially since the FDA’s October 2008 alert about complications with surgical mesh for prolapse and incontinence. [See “FDA alert: Transvaginal placement of surgical mesh carries serious risks,” in the December 2008 issue of OBG Management, at www.obgmanagement.com.] It’s worth noting how these lawyers see themselves: They would likely tell you that they “provide an important service in protecting patients from unscrupulous manufacturers who profit from the vulnerability of people seeking treatment for distressing conditions.” As clinicians, are we absolutely sure that we can say the same of ourselves?
Is it wrong to harp on what happened in the past?
Why revisit events surrounding, for example, the ProteGen Sling? My reply is another question: Where is the evidence that such sequences of events are in the past? Among clinicians, who knows which is best in a collection of kits that changes from one month to the next, without their promoters skipping a beat in proclaiming theirs as the “best”? It isn’t shameful to admit that one doesn’t know which one is best; but it is a shame to act as if one does know, especially when the risk falls to another. The names change; the events are the same.
What is the solution to this problem?
Businesses succeed only when their products are purchased. If clinicians refused to be participants whenever the device industry introduces unproven treatments into the market, industry would be compelled to test their products beforehand. Patients would benefit—by being able to make truly informed choices, with adequate information about risk and benefit. Clinicians would benefit—by being able to provide the most effective treatment without sacrificing their integrity in the process. Ultimately, industry would benefit, by profiting appropriately from products that truly help our patients. Is that an impossible wish?
From my vantage point, it appears that economic factors are playing an increasingly important role in how pelvic organ prolapse (POP) and urinary incontinence (UI) are managed—particularly, in regard to the use of surgical devices. As such, the topic of treating POP and UI deserves our attention to ensure that we make the best decisions for our patients.
Now, I’m a staunch supporter of innovation in treatment; certainly, there is room for improvement in current approaches—particularly in surgery—for treating POP and UI. At the same time, I strongly believe that innovation must be demonstrated to be an improvement before it is incorporated into practice. Although innovation is commonly taken on faith, we should know better than to equate “new” with “better” until evidence, gathered through clinical research, has demonstrated this conclusively. A look at the US Food and Drug Administration’s (FDA’s) process for clearing medical devices for clinical use reveals that such a standard often doesn’t apply—and this should matter to us.
The meaning of 510(k)
Most medical devices are evaluated through an FDA clearance mechanism known as the 510(k) process. This is wholly distinct from the agency’s drug approval process with which most of us are familiar. It’s beyond the scope of this commentary for me to go into detail about 510(k); if you are interested, see two recent commentaries1,2 and visit http://www.fda.gov/cdrh/devadvice/314.html .
In a nutshell, the 510(k) process requires only that an applicant demonstrate that a new medical device has “substantial equivalence” to an already legally marketed device, known as the predicate, which may also have been cleared only through the 510(k) process. That means it’s possible to have generations of products cleared on the basis of one predicate device that was itself never studied adequately.
Indeed, this is the case with most medical devices that have been sold for the surgical treatment of POP and UI—from before the ProteGen Sling (Boston Scientific), through Tension-Free Vaginal Tape (TVT) (Gynecare), and continuing with the newest devices.
The story of the ProteGen Sling ( FIGURE ) offers a cautionary tale about what can go wrong when new devices are cleared by the FDA through 510(k), rather than evaluated through rigorous clinical trials, as drugs are. More recently, experience with the ObTape (Mentor Corporation) followed virtually the same trajectory of events; the product was pulled from the market in 2006 and is now the focus of lawsuits nationwide.
Fortunately, for our patients, experience with TVT (Gynecare) has been favorable. Although TVT was also cleared by the FDA through 510(k), clinical research performed after TVT was introduced has demonstrated its effectiveness and relative safety. Indeed, TVT has revolutionized the treatment of stress UI in women—and, even, our understanding of its etiology.
Several companies are capitalizing on the success of TVT by introducing competing products that are designed to be 1) similar enough to ride on the coattails of TVT yet 2) different enough to capture their own share of market—without evidence of safety or effectiveness required. Even Gynecare (part of Ethicon Women’s Health and Urology, a subsidiary of Johnson & Johnson) has introduced TVT SECUR to compete with its own TVT—again, without independent evidence of safety or effectiveness.
The current market in devices for stress UI is a moving target that makes it nearly impossible—even for research organizations, such as the federally funded Pelvic Floor Disorders Network, that are independent of industry—to develop and implement sound clinical trials of those devices. Why do I say “moving target”? First, there are no assurances that any device chosen for study will remain the same for the duration of a trial. Second, there is no way to foresee which products will be abandoned over the time required for a large clinical trial.

FIGURE The saga of the ProteGen Sling
Transparency over what might be considered “experimental”
Until the FDA changes its process—to one in which 1) medical devices are adequately assessed before they reach market and 2) postmarketing surveillance is required—it’s our duty to insist on evidence of safety and effectiveness before adopting the latest and greatest products that companies have to offer.
Of all the questions that a patient might ask before treatment, three of the most important, surely, are:
- “Will this help me?”
- “If it helps me, how long will it help?”
- “Whether or not this treatment helps me, what risks—in the short-term and over the long-term—does it present?”
Until we can provide our patients with answers that are supported by evidence, products that lack such evidence should be considered experimental, and patients should be counseled accordingly.
Some patients may accept what they’ve been advised are new and unproven treatments—in the way that some physicians are early adopters. Nevertheless, I am concerned that some clinicians do not appear to appreciate the true lack of evidence that accompanies most marketed devices for prolapse and incontinence. They may mistake the FDA 510(k) process of clearance for something similar to the agency’s extended and complex drug approval process. They may accept claims made in industry-produced white papers that are often largely promotional materials, and fail to look further into those claims.
Now, more than ever and above all else, we must stand between marketing and our patients’ safety. We are familiar with the toll that prolapse and incontinence, as chronic conditions, take on our patients; yet it’s that very chronic nature that should lead us to adopt patience and caution in accepting new treatments before they have been adequately studied.
If we cannot always rely on industry to provide clear information about the risks and benefits of new devices, neither can we routinely look to our professional organizations for unbiased information. Often, professional organizations accept cash contributions from industry, raising the question of conflict of interest that may undermine their actions when the priorities of industry do not align with the goal of safeguarding patients’ well-being.
In an unprecedented example of how a professional association can interfere with its own, expert-authored clinical practice guidelines, the American College of Obstetricians and Gynecologists (ACOG) more than a year ago rescinded one of its published guidelines on POP (Issue 79, February 20073 ) and replaced it with a new guideline (Issue 85, September 20074 ). The new guideline is nearly identical to the prior one—save for one sentence, in which “experimental” is deleted in a discussion of kits of trocar-based synthetic materials sold for the surgical treatment of pelvic organ prolapse (see the EXCERPT ).
The deletion is crucial because offering informed consent for surgery requires a patient to accept risks in balance with an expectation of benefit. A patient cannot be appropriately informed when no evidence of benefit exists and evidence of postoperative risk is extremely limited.
Now, I am not declaring that ACOG acted with bias because of a financial conflict of interest with industry in this instance; the fact that a financial conflict of interest exists for ACOG, however, cannot be disputed if one examines the College’s Annual Report, where contributors are listed. (For a comprehensive, if disillusioning, treatise on the many effects of financial conflict of interest within medicine, I recommend the book On The Take.5 )
Differences between the two bulletins are marked in boldface
Bulletin #79 (original wording): “Given the limited data and frequent changes in the marketed products (particularly with regard to type of mesh material itself, which is most closely associated with several of the postoperative risks especially mesh erosion), if clinicians recommend these procedures before evidence of their risk-benefit is fully understood, the procedures should be considered experimental and patients consented for surgery with that understanding.”
Bulletin #84 (revised wording): “Given the limited data and frequent changes in the marketed products for vaginal surgery for prolapse repair (particularly with regard to type of mesh material itself, which is most closely associated with several of the postoperative risks especially mesh erosion), patients should consent to surgery with an understanding of the post-operative risks and lack of long-term outcomes data.”
Case: Radiofrequency therapy. Even when clinical experience demonstrates lack of effectiveness or an unacceptable rate of complications associated with certain techniques or devices, unequivocal evidence of such problems does not always appear in the literature. One example of this is how a technique was translated into a treatment for incontinence by way of its use in other fields.
Radiofrequency has, among other uses, been used to ablate nerves in intractable chronic pain and to address joint instability in orthopedic surgery. Radiofrequency energy was then, by extension, applied transvaginally to tissue (known as “endopelvic” fascia, of a distinctly different nature than parietal fascia involved in orthopedic procedures) surrounding the urethra. The goal was to coagulate “supporting” tissues and “correct” urethral hypermobility that purportedly causes stress incontinence.
Marketing of the SURx Transvaginal System (CooperSurgical, Inc.) began in 2002, followed by reports of success. One industry-sponsored study, for example, reported a 73% rate of either continence or improvement after 12 months.6
Despite such favorable early results, however, in 2006 CooperSurgical decided to abandon this system, citing “technique-dependent” results of the procedure. Since then, independent research has shown a very low initial success rate that declines rapidly—within weeks or months—of treatment.7,8
A different radiofrequency technique continues to be marketed—the Renessa System™ (NovaSys Medical), which uses a urethral catheter-mounted system to deliver radiofrequency energy through the urethral mucosa to the submucosa and adjacent tissues. Once again, initial reports of industry-sponsored research showed promising results; one study of 110 patients reported 74% achieved continence or improvement after 1 year.9 In a follow-up report of 21 of the original 110 patients, “improvement” was reported in 74% after 3 years.10 Independent research has yet to be reported in the literature.
Of particular concern, no data exist on the long-term effect of denaturing collagen in the urethra and adjacent tissues in relation to UI, other aspects of bladder function, or sexual function. An apparent lack of immediate complications cannot be equated with safety; we need long-term studies to determine whether urethral function is affected adversely compared with that in untreated women and women treated with surgery.
Bring on innovation—in context!
For those who consider my argument anti-innovation, let me repeat: I believe strongly in innovation to improve care for our patients. Am I anti-industry? Only when there is an unbridled race to profit from marketing products without safeguards to ensure, first and foremost, the safety of our patients and, second, their long-term effectiveness. Knowingly or unknowingly, patients then become the guinea pigs on whom these products are tested—just not in the appropriate context of clinical research and informed consent for participation.
Instead (as happened in the US Public Health Service’s Tuskegee syphilis experiments), patients serve as research subjects without their consent when they receive untested products and undergo unproven treatments. And because clinicians are the conduit through which patients receive untested and unproven treatments, who is ultimately responsible for the outcome?
Industry brings innovation to clinical practice. But it is incumbent on clinicians to recognize, with unflinching honesty, the bottom line on which industry operates. Prolapse and incontinence are deeply distressing for our patients, but these chronic conditions are not life-threatening; virtually all women who suffer these conditions have been symptomatic for years before they come for care. I see no need, except to increase that bottom line, to rush products to market before they have been evaluated sufficiently to determine whether “new” is actually “better.”
For clinicians who style themselves as early adopters, remember: It’s not you, but your patient, who is “adopting” a foreign material and having it placed deep in the most intimate area of her body—a foreign material intended to stay for life (except for those unfortunate patients who must have it removed). Above all, we must do no harm—an elusive goal when some of us try to attract patients by being the first to use a product before evidence of its risk and utility have been established in practice.
Does this kind of talk encourage litigation?
Does a commentary like this one provide fodder for plaintiff attorneys who are seeking grounds for product liability lawsuits against manufacturers and malpractice claims against clinicians? Please! Spend a moment on the Web, and you will see that the lawyers are already busy—especially since the FDA’s October 2008 alert about complications with surgical mesh for prolapse and incontinence. [See “FDA alert: Transvaginal placement of surgical mesh carries serious risks,” in the December 2008 issue of OBG Management, at www.obgmanagement.com.] It’s worth noting how these lawyers see themselves: They would likely tell you that they “provide an important service in protecting patients from unscrupulous manufacturers who profit from the vulnerability of people seeking treatment for distressing conditions.” As clinicians, are we absolutely sure that we can say the same of ourselves?
Is it wrong to harp on what happened in the past?
Why revisit events surrounding, for example, the ProteGen Sling? My reply is another question: Where is the evidence that such sequences of events are in the past? Among clinicians, who knows which is best in a collection of kits that changes from one month to the next, without their promoters skipping a beat in proclaiming theirs as the “best”? It isn’t shameful to admit that one doesn’t know which one is best; but it is a shame to act as if one does know, especially when the risk falls to another. The names change; the events are the same.
What is the solution to this problem?
Businesses succeed only when their products are purchased. If clinicians refused to be participants whenever the device industry introduces unproven treatments into the market, industry would be compelled to test their products beforehand. Patients would benefit—by being able to make truly informed choices, with adequate information about risk and benefit. Clinicians would benefit—by being able to provide the most effective treatment without sacrificing their integrity in the process. Ultimately, industry would benefit, by profiting appropriately from products that truly help our patients. Is that an impossible wish?
1. Goldman HB. Is new always better? Curr Urol Rep. 2007;8(4):253-254.
2. Ostergard DR. Lessons from the past: directions for the future. Do new marketed surgical procedures and grafts produce ethical, personal liability, and legal concerns for physicians? Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:591-598.
3. ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 79: Pelvic organ prolapse. Obstet Gynecol. 2007;109(2 Pt 1):461-473.
4. ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 85: Pelvic organ prolapse. Obstet Gynecol. 2007;110:717-729.
5. Kassirer JP. On the Take: How Medicine’s Complicity with Big Business Can Endanger Your Health. New York: Oxford University Press; 2005.
6. Dmochowski RR, Avon M, Ross J, et al. Transvaginal radiofrequency treatment of the endopelvic fascia: a prospective evaluation for the treatment of genuine stress urinary incontinence. J Urol. 2003;169:1028-1032.
7. Buchsbaum GM, McConville J, Korni R, Duecy EE. Outcome of transvaginal radiofrequency for treatment of women with stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18(3):263-265.
8. Ismail SI. Radiofrequency remodelling of the endopelvic fascia is not an effective procedure for urodynamic stress incontinence in women. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1205-1209.
9. Appell RA, Juma S, Wells WG, et al. Transurethral radio-frequency energy collagen micro-remodeling for the treatment of female stress urinary incontinence. Neurourol Urodyn. 2006;25(4):331-336.
10. Appell RA, Singh G, Klimberg IW, et al. Nonsurgical radiofrequency collagen denaturation for stress urinary incontinence: retrospective 3-year evaluation. Expert Rev Med Devices. 2007;4:455-461.
1. Goldman HB. Is new always better? Curr Urol Rep. 2007;8(4):253-254.
2. Ostergard DR. Lessons from the past: directions for the future. Do new marketed surgical procedures and grafts produce ethical, personal liability, and legal concerns for physicians? Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:591-598.
3. ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 79: Pelvic organ prolapse. Obstet Gynecol. 2007;109(2 Pt 1):461-473.
4. ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 85: Pelvic organ prolapse. Obstet Gynecol. 2007;110:717-729.
5. Kassirer JP. On the Take: How Medicine’s Complicity with Big Business Can Endanger Your Health. New York: Oxford University Press; 2005.
6. Dmochowski RR, Avon M, Ross J, et al. Transvaginal radiofrequency treatment of the endopelvic fascia: a prospective evaluation for the treatment of genuine stress urinary incontinence. J Urol. 2003;169:1028-1032.
7. Buchsbaum GM, McConville J, Korni R, Duecy EE. Outcome of transvaginal radiofrequency for treatment of women with stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18(3):263-265.
8. Ismail SI. Radiofrequency remodelling of the endopelvic fascia is not an effective procedure for urodynamic stress incontinence in women. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1205-1209.
9. Appell RA, Juma S, Wells WG, et al. Transurethral radio-frequency energy collagen micro-remodeling for the treatment of female stress urinary incontinence. Neurourol Urodyn. 2006;25(4):331-336.
10. Appell RA, Singh G, Klimberg IW, et al. Nonsurgical radiofrequency collagen denaturation for stress urinary incontinence: retrospective 3-year evaluation. Expert Rev Med Devices. 2007;4:455-461.
Are new tools for correcting prolapse and incontinence better just because they’re new?
From my vantage point, it appears that economic factors are playing an increasingly important role in how pelvic organ prolapse (POP) and urinary incontinence (UI) are managed—particularly, in regard to the use of surgical devices. As such, the topic of treating POP and UI deserves our attention to ensure that we make the best decisions for our patients.
Now, I’m a staunch supporter of innovation in treatment; certainly, there is room for improvement in current approaches—particularly in surgery—for treating POP and UI. At the same time, I strongly believe that innovation must be demonstrated to be an improvement before it is incorporated into practice. Although innovation is commonly taken on faith, we should know better than to equate “new” with “better” until evidence, gathered through clinical research, has demonstrated this conclusively. A look at the US Food and Drug Administration’s (FDA’s) process for clearing medical devices for clinical use reveals that such a standard often doesn’t apply—and this should matter to us.
The meaning of 510(k)
Most medical devices are evaluated through an FDA clearance mechanism known as the 510(k) process. This is wholly distinct from the agency’s drug approval process with which most of us are familiar. It’s beyond the scope of this commentary for me to go into detail about 510(k); if you are interested, see two recent commentaries“FDA alert: Transvaginal placement of surgical mesh carries serious risks,” in the December 2008 issue of OBG Management.] It’s worth noting how these lawyers see themselves: They would likely tell you that they “provide an important service in protecting patients from unscrupulous manufacturers who profit from the vulnerability of people seeking treatment for distressing conditions.” As clinicians, are we absolutely sure that we can say the same of ourselves?
Is it wrong to harp on what happened in the past?
Why revisit events surrounding, for example, the ProteGen Sling? My reply is another question: Where is the evidence that such sequences of events are in the past? Among clinicians, who knows which is best in a collection of kits that changes from one month to the next, without their promoters skipping a beat in proclaiming theirs as the “best”? It isn’t shameful to admit that one doesn’t know which one is best; but it is a shame to act as if one does know, especially when the risk falls to another. The names change; the events are the same.
What is the solution to this problem?
Businesses succeed only when their products are purchased. If clinicians refused to be participants whenever the device industry introduces unproven treatments into the market, industry would be compelled to test their products beforehand. Patients would benefit—by being able to make truly informed choices, with adequate information about risk and benefit. Clinicians would benefit—by being able to provide the most effective treatment without sacrificing their integrity in the process. Ultimately, industry would benefit, by profiting appropriately from products that truly help our patients. Is that an impossible wish?
- Goldman HB. Is new always better? Curr Urol Rep. 2007;8(4):253-254.
- Ostergard DR. Lessons from the past: directions for the future. Do new marketed surgical procedures and grafts produce ethical, personal liability, and legal concerns for physicians? Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:591-598.
- ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 79: Pelvic organ prolapse. Obstet Gynecol. 2007;109(2 Pt 1):461-473.
- ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 85: Pelvic organ prolapse. Obstet Gynecol. 2007;110:717-729.
- Kassirer JP. On the Take: How Medicine’s Complicity with Big Business Can Endanger Your Health. New York: Oxford University Press; 2005.
- Dmochowski RR, Avon M, Ross J, et al. Transvaginal radiofrequency treatment of the endopelvic fascia: a prospective evaluation for the treatment of genuine stress urinary incontinence. J Urol. 2003;169:1028-1032.
- Buchsbaum GM, McConville J, Korni R, Duecy EE. Outcome of transvaginal radiofrequency for treatment of women with stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18(3):263-265.
- Ismail SI. Radiofrequency remodelling of the endopelvic fascia is not an effective procedure for urodynamic stress incontinence in women. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1205-1209.
- Appell RA, Juma S, Wells WG, et al. Transurethral radio-frequency energy collagen micro-remodeling for the treatment of female stress urinary incontinence. Neurourol Urodyn. 2006;25(4):331-336.
- Appell RA, Singh G, Klimberg IW, et al. Nonsurgical radiofrequency collagen denaturation for stress urinary incontinence: retrospective 3-year evaluation. Expert Rev Med Devices. 2007;4:455-461.
From my vantage point, it appears that economic factors are playing an increasingly important role in how pelvic organ prolapse (POP) and urinary incontinence (UI) are managed—particularly, in regard to the use of surgical devices. As such, the topic of treating POP and UI deserves our attention to ensure that we make the best decisions for our patients.
Now, I’m a staunch supporter of innovation in treatment; certainly, there is room for improvement in current approaches—particularly in surgery—for treating POP and UI. At the same time, I strongly believe that innovation must be demonstrated to be an improvement before it is incorporated into practice. Although innovation is commonly taken on faith, we should know better than to equate “new” with “better” until evidence, gathered through clinical research, has demonstrated this conclusively. A look at the US Food and Drug Administration’s (FDA’s) process for clearing medical devices for clinical use reveals that such a standard often doesn’t apply—and this should matter to us.
The meaning of 510(k)
Most medical devices are evaluated through an FDA clearance mechanism known as the 510(k) process. This is wholly distinct from the agency’s drug approval process with which most of us are familiar. It’s beyond the scope of this commentary for me to go into detail about 510(k); if you are interested, see two recent commentaries“FDA alert: Transvaginal placement of surgical mesh carries serious risks,” in the December 2008 issue of OBG Management.] It’s worth noting how these lawyers see themselves: They would likely tell you that they “provide an important service in protecting patients from unscrupulous manufacturers who profit from the vulnerability of people seeking treatment for distressing conditions.” As clinicians, are we absolutely sure that we can say the same of ourselves?
Is it wrong to harp on what happened in the past?
Why revisit events surrounding, for example, the ProteGen Sling? My reply is another question: Where is the evidence that such sequences of events are in the past? Among clinicians, who knows which is best in a collection of kits that changes from one month to the next, without their promoters skipping a beat in proclaiming theirs as the “best”? It isn’t shameful to admit that one doesn’t know which one is best; but it is a shame to act as if one does know, especially when the risk falls to another. The names change; the events are the same.
What is the solution to this problem?
Businesses succeed only when their products are purchased. If clinicians refused to be participants whenever the device industry introduces unproven treatments into the market, industry would be compelled to test their products beforehand. Patients would benefit—by being able to make truly informed choices, with adequate information about risk and benefit. Clinicians would benefit—by being able to provide the most effective treatment without sacrificing their integrity in the process. Ultimately, industry would benefit, by profiting appropriately from products that truly help our patients. Is that an impossible wish?
From my vantage point, it appears that economic factors are playing an increasingly important role in how pelvic organ prolapse (POP) and urinary incontinence (UI) are managed—particularly, in regard to the use of surgical devices. As such, the topic of treating POP and UI deserves our attention to ensure that we make the best decisions for our patients.
Now, I’m a staunch supporter of innovation in treatment; certainly, there is room for improvement in current approaches—particularly in surgery—for treating POP and UI. At the same time, I strongly believe that innovation must be demonstrated to be an improvement before it is incorporated into practice. Although innovation is commonly taken on faith, we should know better than to equate “new” with “better” until evidence, gathered through clinical research, has demonstrated this conclusively. A look at the US Food and Drug Administration’s (FDA’s) process for clearing medical devices for clinical use reveals that such a standard often doesn’t apply—and this should matter to us.
The meaning of 510(k)
Most medical devices are evaluated through an FDA clearance mechanism known as the 510(k) process. This is wholly distinct from the agency’s drug approval process with which most of us are familiar. It’s beyond the scope of this commentary for me to go into detail about 510(k); if you are interested, see two recent commentaries“FDA alert: Transvaginal placement of surgical mesh carries serious risks,” in the December 2008 issue of OBG Management.] It’s worth noting how these lawyers see themselves: They would likely tell you that they “provide an important service in protecting patients from unscrupulous manufacturers who profit from the vulnerability of people seeking treatment for distressing conditions.” As clinicians, are we absolutely sure that we can say the same of ourselves?
Is it wrong to harp on what happened in the past?
Why revisit events surrounding, for example, the ProteGen Sling? My reply is another question: Where is the evidence that such sequences of events are in the past? Among clinicians, who knows which is best in a collection of kits that changes from one month to the next, without their promoters skipping a beat in proclaiming theirs as the “best”? It isn’t shameful to admit that one doesn’t know which one is best; but it is a shame to act as if one does know, especially when the risk falls to another. The names change; the events are the same.
What is the solution to this problem?
Businesses succeed only when their products are purchased. If clinicians refused to be participants whenever the device industry introduces unproven treatments into the market, industry would be compelled to test their products beforehand. Patients would benefit—by being able to make truly informed choices, with adequate information about risk and benefit. Clinicians would benefit—by being able to provide the most effective treatment without sacrificing their integrity in the process. Ultimately, industry would benefit, by profiting appropriately from products that truly help our patients. Is that an impossible wish?
- Goldman HB. Is new always better? Curr Urol Rep. 2007;8(4):253-254.
- Ostergard DR. Lessons from the past: directions for the future. Do new marketed surgical procedures and grafts produce ethical, personal liability, and legal concerns for physicians? Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:591-598.
- ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 79: Pelvic organ prolapse. Obstet Gynecol. 2007;109(2 Pt 1):461-473.
- ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 85: Pelvic organ prolapse. Obstet Gynecol. 2007;110:717-729.
- Kassirer JP. On the Take: How Medicine’s Complicity with Big Business Can Endanger Your Health. New York: Oxford University Press; 2005.
- Dmochowski RR, Avon M, Ross J, et al. Transvaginal radiofrequency treatment of the endopelvic fascia: a prospective evaluation for the treatment of genuine stress urinary incontinence. J Urol. 2003;169:1028-1032.
- Buchsbaum GM, McConville J, Korni R, Duecy EE. Outcome of transvaginal radiofrequency for treatment of women with stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18(3):263-265.
- Ismail SI. Radiofrequency remodelling of the endopelvic fascia is not an effective procedure for urodynamic stress incontinence in women. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1205-1209.
- Appell RA, Juma S, Wells WG, et al. Transurethral radio-frequency energy collagen micro-remodeling for the treatment of female stress urinary incontinence. Neurourol Urodyn. 2006;25(4):331-336.
- Appell RA, Singh G, Klimberg IW, et al. Nonsurgical radiofrequency collagen denaturation for stress urinary incontinence: retrospective 3-year evaluation. Expert Rev Med Devices. 2007;4:455-461.
- Goldman HB. Is new always better? Curr Urol Rep. 2007;8(4):253-254.
- Ostergard DR. Lessons from the past: directions for the future. Do new marketed surgical procedures and grafts produce ethical, personal liability, and legal concerns for physicians? Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:591-598.
- ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 79: Pelvic organ prolapse. Obstet Gynecol. 2007;109(2 Pt 1):461-473.
- ACOG Committee on Practice Bulletins–Gynecology, American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 85: Pelvic organ prolapse. Obstet Gynecol. 2007;110:717-729.
- Kassirer JP. On the Take: How Medicine’s Complicity with Big Business Can Endanger Your Health. New York: Oxford University Press; 2005.
- Dmochowski RR, Avon M, Ross J, et al. Transvaginal radiofrequency treatment of the endopelvic fascia: a prospective evaluation for the treatment of genuine stress urinary incontinence. J Urol. 2003;169:1028-1032.
- Buchsbaum GM, McConville J, Korni R, Duecy EE. Outcome of transvaginal radiofrequency for treatment of women with stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18(3):263-265.
- Ismail SI. Radiofrequency remodelling of the endopelvic fascia is not an effective procedure for urodynamic stress incontinence in women. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1205-1209.
- Appell RA, Juma S, Wells WG, et al. Transurethral radio-frequency energy collagen micro-remodeling for the treatment of female stress urinary incontinence. Neurourol Urodyn. 2006;25(4):331-336.
- Appell RA, Singh G, Klimberg IW, et al. Nonsurgical radiofrequency collagen denaturation for stress urinary incontinence: retrospective 3-year evaluation. Expert Rev Med Devices. 2007;4:455-461.
Is hysterectomy definitive treatment for high-grade intraepithelial neoplasia?
The cervix is most susceptible at the transformation zone, but the remainder of the squamous epithelium of the lower genital tract may be at risk in susceptible persons.
What is the residual risk?
Who should we watch?
If the cervix is removed during hysterectomy after CIN 2+ is diagnosed, what is the residual risk to the patient? How should she be managed?
These are key questions, despite the fact that hysterectomy is not as commonly performed to treat high-grade CIN as it is for other indications, such as abnormal bleeding, pain, endometriosis, and fibroids. Hysterectomy for these other conditions without a concomitant history of neoplasia obviates the need for cervical cancer screening via cytology or HPV testing, or both. When CIN is the reason for the hysterectomy, however, or the patient has a history of CIN 2+, there is some residual risk, with the most common sequela being VAIN and, if the VAIN remains undiscovered, vaginal carcinoma. But how do we identify the patient at high risk for VAIN so that we can provide extra resources (screening or colposcopy services)?
Older women merit closer attention
One strength of this study is the fact that it statistically identifiled “older women” as a high-risk subset more likely to develop VAIN 2+. In the study, the mean age of women who were likely to develop VAIN 2+ was 61 years, compared with 46.9 years for women likely to remain free of disease.
The median interval between hysterectomy and diagnosis of VAIN 2+ was 35 months (range, 5–103 months). This information allows us to anticipate an optimal window in which to focus extra screening.
Retrospective design is a weakness
The retrospective design of this review of pathology data from multiple practices in multiple records is a limitation. The exclusion of patients who had coexisting VAIN or a history of VAIN is laudable, but bias is possible. The quality of the documentation of examination of the remainder of the lower genital tract in patients who had abnormal cervical screening during colposcopy (which led to the diagnosis of CIN) is subject to extreme variability, compared with a prospective design that would have defined the elements of vaginal and vulvar colposcopic examination.
In other words, I am concerned that these are truly incident—rather than persistent—lesions following hysterectomy.
How would HPV testing come into play?
This study and the other published studies that address the subject of hysterectomy in the treatment of CIN 2+ did not use HPV testing. If they had, it likely would have provided more information about the true risk of recurrence and helped determine the best screening interval.
The American Society for Colposcopy and Cervical Pathology (ASCCP) guidelines from 2006 do not address this particular scenario, but American College of Obstetricians and Gynecologists (ACOG) guidelines do recommend annual Papanicolaou testing following hysterectomy for CIN until three consecutive tests are negative.1
How reliable are screening methods when the vagina is the target tissue?
Cytologic sampling, HPV test sampling, and colposcopy of the “at-risk” transformation zone or endocervical canal are easier to accomplish than are examination and testing of the entire vaginal vault. The latter are more prone to error and lack evidence-based data to substantiate our practice.
Nevertheless, excellent practice recommendations regarding vaginal colposcopy can be found at the ASCCP Web site at www.asccp.org/edu/practice/vagina.shtml.
We should always carefully examine the entire lower genital tract during colposcopy following referral for abnormal cytology or another abnormal screening test—whether or not the patient has a cervix.-NEAL M.LONKY, MD, MPH
1. Cervical cytology screening. ACOG Practice Bulletin #45. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2003;102:417-427.
The cervix is most susceptible at the transformation zone, but the remainder of the squamous epithelium of the lower genital tract may be at risk in susceptible persons.
What is the residual risk?
Who should we watch?
If the cervix is removed during hysterectomy after CIN 2+ is diagnosed, what is the residual risk to the patient? How should she be managed?
These are key questions, despite the fact that hysterectomy is not as commonly performed to treat high-grade CIN as it is for other indications, such as abnormal bleeding, pain, endometriosis, and fibroids. Hysterectomy for these other conditions without a concomitant history of neoplasia obviates the need for cervical cancer screening via cytology or HPV testing, or both. When CIN is the reason for the hysterectomy, however, or the patient has a history of CIN 2+, there is some residual risk, with the most common sequela being VAIN and, if the VAIN remains undiscovered, vaginal carcinoma. But how do we identify the patient at high risk for VAIN so that we can provide extra resources (screening or colposcopy services)?
Older women merit closer attention
One strength of this study is the fact that it statistically identifiled “older women” as a high-risk subset more likely to develop VAIN 2+. In the study, the mean age of women who were likely to develop VAIN 2+ was 61 years, compared with 46.9 years for women likely to remain free of disease.
The median interval between hysterectomy and diagnosis of VAIN 2+ was 35 months (range, 5–103 months). This information allows us to anticipate an optimal window in which to focus extra screening.
Retrospective design is a weakness
The retrospective design of this review of pathology data from multiple practices in multiple records is a limitation. The exclusion of patients who had coexisting VAIN or a history of VAIN is laudable, but bias is possible. The quality of the documentation of examination of the remainder of the lower genital tract in patients who had abnormal cervical screening during colposcopy (which led to the diagnosis of CIN) is subject to extreme variability, compared with a prospective design that would have defined the elements of vaginal and vulvar colposcopic examination.
In other words, I am concerned that these are truly incident—rather than persistent—lesions following hysterectomy.
How would HPV testing come into play?
This study and the other published studies that address the subject of hysterectomy in the treatment of CIN 2+ did not use HPV testing. If they had, it likely would have provided more information about the true risk of recurrence and helped determine the best screening interval.
The American Society for Colposcopy and Cervical Pathology (ASCCP) guidelines from 2006 do not address this particular scenario, but American College of Obstetricians and Gynecologists (ACOG) guidelines do recommend annual Papanicolaou testing following hysterectomy for CIN until three consecutive tests are negative.1
How reliable are screening methods when the vagina is the target tissue?
Cytologic sampling, HPV test sampling, and colposcopy of the “at-risk” transformation zone or endocervical canal are easier to accomplish than are examination and testing of the entire vaginal vault. The latter are more prone to error and lack evidence-based data to substantiate our practice.
Nevertheless, excellent practice recommendations regarding vaginal colposcopy can be found at the ASCCP Web site at www.asccp.org/edu/practice/vagina.shtml.
We should always carefully examine the entire lower genital tract during colposcopy following referral for abnormal cytology or another abnormal screening test—whether or not the patient has a cervix.-NEAL M.LONKY, MD, MPH
The cervix is most susceptible at the transformation zone, but the remainder of the squamous epithelium of the lower genital tract may be at risk in susceptible persons.
What is the residual risk?
Who should we watch?
If the cervix is removed during hysterectomy after CIN 2+ is diagnosed, what is the residual risk to the patient? How should she be managed?
These are key questions, despite the fact that hysterectomy is not as commonly performed to treat high-grade CIN as it is for other indications, such as abnormal bleeding, pain, endometriosis, and fibroids. Hysterectomy for these other conditions without a concomitant history of neoplasia obviates the need for cervical cancer screening via cytology or HPV testing, or both. When CIN is the reason for the hysterectomy, however, or the patient has a history of CIN 2+, there is some residual risk, with the most common sequela being VAIN and, if the VAIN remains undiscovered, vaginal carcinoma. But how do we identify the patient at high risk for VAIN so that we can provide extra resources (screening or colposcopy services)?
Older women merit closer attention
One strength of this study is the fact that it statistically identifiled “older women” as a high-risk subset more likely to develop VAIN 2+. In the study, the mean age of women who were likely to develop VAIN 2+ was 61 years, compared with 46.9 years for women likely to remain free of disease.
The median interval between hysterectomy and diagnosis of VAIN 2+ was 35 months (range, 5–103 months). This information allows us to anticipate an optimal window in which to focus extra screening.
Retrospective design is a weakness
The retrospective design of this review of pathology data from multiple practices in multiple records is a limitation. The exclusion of patients who had coexisting VAIN or a history of VAIN is laudable, but bias is possible. The quality of the documentation of examination of the remainder of the lower genital tract in patients who had abnormal cervical screening during colposcopy (which led to the diagnosis of CIN) is subject to extreme variability, compared with a prospective design that would have defined the elements of vaginal and vulvar colposcopic examination.
In other words, I am concerned that these are truly incident—rather than persistent—lesions following hysterectomy.
How would HPV testing come into play?
This study and the other published studies that address the subject of hysterectomy in the treatment of CIN 2+ did not use HPV testing. If they had, it likely would have provided more information about the true risk of recurrence and helped determine the best screening interval.
The American Society for Colposcopy and Cervical Pathology (ASCCP) guidelines from 2006 do not address this particular scenario, but American College of Obstetricians and Gynecologists (ACOG) guidelines do recommend annual Papanicolaou testing following hysterectomy for CIN until three consecutive tests are negative.1
How reliable are screening methods when the vagina is the target tissue?
Cytologic sampling, HPV test sampling, and colposcopy of the “at-risk” transformation zone or endocervical canal are easier to accomplish than are examination and testing of the entire vaginal vault. The latter are more prone to error and lack evidence-based data to substantiate our practice.
Nevertheless, excellent practice recommendations regarding vaginal colposcopy can be found at the ASCCP Web site at www.asccp.org/edu/practice/vagina.shtml.
We should always carefully examine the entire lower genital tract during colposcopy following referral for abnormal cytology or another abnormal screening test—whether or not the patient has a cervix.-NEAL M.LONKY, MD, MPH
1. Cervical cytology screening. ACOG Practice Bulletin #45. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2003;102:417-427.
1. Cervical cytology screening. ACOG Practice Bulletin #45. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2003;102:417-427.
Is MRSA a common isolate from vulvar abscesses?
The vulva is especially susceptible to colonization by MRSA owing to its proximity to the rectum and to trauma caused by shaving, waxing, sexual contact, and use of personal hygiene products.
Moreover, obese and disabled women may have difficulty cleaning the vulva adequately; poor hygiene is also associated with MRSA colonization.
MRSA wasn’t the only pathogen identified
Gram-negative organisms, such as Proteus mirabilis and Escherichia coli, and gram-positive organisms, such as Enterococcus and group B Streptococcus also were isolated from patients in this study.
Although most of the vulvar abscesses in this study were colonized with MRSA, that fact was not apparent until specimens were cultured. No presenting signs or symptoms distinguished patients who had MRSA from those who did not.
Nor were women with MRSA more likely to require hospitalization or experience complications from treatment. Rather, hospitalization was more likely in women who had such comorbidities as:
- diabetes
- hypertension
- an initial serum glucose level above 200 mg/dL
- a larger abscess (mean, 5.2 cm in diameter)
- an elevated white blood cell count (≥12×103/µL).
Management of MRSA-colonized vulvar abscess primarily involved incision and drainage. Most inpatients also received intravenous vancomycin or clindamycin. Among outpatients, trimethoprim-sulfamethoxazole, an antibiotic regimen known to be eff ective against MRSA, was given in selected cases.
Limitations of this study
This study was conducted in a large county hospital in San Antonio that served primarily low-income Hispanic patients. Findings may therefore apply only to this population or geographic region.
Several variables were either not presented or inadequately discussed in the published study. For example, 26 of the subjects were pregnant. Should they have been included in the overall analysis? Does pregnancy alter the immune system—thereby becoming a risk factor for MRSA-colonized vulvar abscess? Was antibiotic selection different for pregnant patients than it was for nonpregnant patients?
The article also fails to provide much information on the prevalence of sexually transmitted infections (STI) in this population. Only 41% of the 133 women who had their abscess cultured were screened for STI. If these patients were infected with HIV (AIDS-defined), Chlamydia trachomatis, gonorrhea, genital herpes, or other STI pathogen, how would this have changed the data and outcomes?
Last, it is unclear whether the 10 cases of recurrent vulvar abscess identified in this study came from the inpatient or outpatient group.
A number of practice points can be gleaned from this study:
- When any patient has a vulvar abscess, culture it for aerobic and anaerobic microorganisms, including MRSA
- Because highly resistant organisms are becoming increasingly common in the community as well as the hospital, it is critical that you be familiar with your hospital’s antibiotic biogram, which delineates the organisms that are causing infection as well as susceptibility patterns
- Incision and drainage are the mainstay of management of MRSA-colonized vulvar abscess
- When deciding whether to treat a patient as an inpatient, consider medical conditions such as diabetes, HIV infection, obesity, and other conditions that compromise the immune system
- When selecting an antibiotic, choose one that includes coverage of MRSA as well as gram-negative enteric and other gram-positive organisms
- To prevent the spread of MRSA, incorporate proper hand washing and other infection-control measures into routine procedures. Also, decontaminate areas in which patients undergo incision and drainage to prevent transmission of MRSA to staff and other patients.—DAVID A. BAKER, MD
The vulva is especially susceptible to colonization by MRSA owing to its proximity to the rectum and to trauma caused by shaving, waxing, sexual contact, and use of personal hygiene products.
Moreover, obese and disabled women may have difficulty cleaning the vulva adequately; poor hygiene is also associated with MRSA colonization.
MRSA wasn’t the only pathogen identified
Gram-negative organisms, such as Proteus mirabilis and Escherichia coli, and gram-positive organisms, such as Enterococcus and group B Streptococcus also were isolated from patients in this study.
Although most of the vulvar abscesses in this study were colonized with MRSA, that fact was not apparent until specimens were cultured. No presenting signs or symptoms distinguished patients who had MRSA from those who did not.
Nor were women with MRSA more likely to require hospitalization or experience complications from treatment. Rather, hospitalization was more likely in women who had such comorbidities as:
- diabetes
- hypertension
- an initial serum glucose level above 200 mg/dL
- a larger abscess (mean, 5.2 cm in diameter)
- an elevated white blood cell count (≥12×103/µL).
Management of MRSA-colonized vulvar abscess primarily involved incision and drainage. Most inpatients also received intravenous vancomycin or clindamycin. Among outpatients, trimethoprim-sulfamethoxazole, an antibiotic regimen known to be eff ective against MRSA, was given in selected cases.
Limitations of this study
This study was conducted in a large county hospital in San Antonio that served primarily low-income Hispanic patients. Findings may therefore apply only to this population or geographic region.
Several variables were either not presented or inadequately discussed in the published study. For example, 26 of the subjects were pregnant. Should they have been included in the overall analysis? Does pregnancy alter the immune system—thereby becoming a risk factor for MRSA-colonized vulvar abscess? Was antibiotic selection different for pregnant patients than it was for nonpregnant patients?
The article also fails to provide much information on the prevalence of sexually transmitted infections (STI) in this population. Only 41% of the 133 women who had their abscess cultured were screened for STI. If these patients were infected with HIV (AIDS-defined), Chlamydia trachomatis, gonorrhea, genital herpes, or other STI pathogen, how would this have changed the data and outcomes?
Last, it is unclear whether the 10 cases of recurrent vulvar abscess identified in this study came from the inpatient or outpatient group.
A number of practice points can be gleaned from this study:
- When any patient has a vulvar abscess, culture it for aerobic and anaerobic microorganisms, including MRSA
- Because highly resistant organisms are becoming increasingly common in the community as well as the hospital, it is critical that you be familiar with your hospital’s antibiotic biogram, which delineates the organisms that are causing infection as well as susceptibility patterns
- Incision and drainage are the mainstay of management of MRSA-colonized vulvar abscess
- When deciding whether to treat a patient as an inpatient, consider medical conditions such as diabetes, HIV infection, obesity, and other conditions that compromise the immune system
- When selecting an antibiotic, choose one that includes coverage of MRSA as well as gram-negative enteric and other gram-positive organisms
- To prevent the spread of MRSA, incorporate proper hand washing and other infection-control measures into routine procedures. Also, decontaminate areas in which patients undergo incision and drainage to prevent transmission of MRSA to staff and other patients.—DAVID A. BAKER, MD
The vulva is especially susceptible to colonization by MRSA owing to its proximity to the rectum and to trauma caused by shaving, waxing, sexual contact, and use of personal hygiene products.
Moreover, obese and disabled women may have difficulty cleaning the vulva adequately; poor hygiene is also associated with MRSA colonization.
MRSA wasn’t the only pathogen identified
Gram-negative organisms, such as Proteus mirabilis and Escherichia coli, and gram-positive organisms, such as Enterococcus and group B Streptococcus also were isolated from patients in this study.
Although most of the vulvar abscesses in this study were colonized with MRSA, that fact was not apparent until specimens were cultured. No presenting signs or symptoms distinguished patients who had MRSA from those who did not.
Nor were women with MRSA more likely to require hospitalization or experience complications from treatment. Rather, hospitalization was more likely in women who had such comorbidities as:
- diabetes
- hypertension
- an initial serum glucose level above 200 mg/dL
- a larger abscess (mean, 5.2 cm in diameter)
- an elevated white blood cell count (≥12×103/µL).
Management of MRSA-colonized vulvar abscess primarily involved incision and drainage. Most inpatients also received intravenous vancomycin or clindamycin. Among outpatients, trimethoprim-sulfamethoxazole, an antibiotic regimen known to be eff ective against MRSA, was given in selected cases.
Limitations of this study
This study was conducted in a large county hospital in San Antonio that served primarily low-income Hispanic patients. Findings may therefore apply only to this population or geographic region.
Several variables were either not presented or inadequately discussed in the published study. For example, 26 of the subjects were pregnant. Should they have been included in the overall analysis? Does pregnancy alter the immune system—thereby becoming a risk factor for MRSA-colonized vulvar abscess? Was antibiotic selection different for pregnant patients than it was for nonpregnant patients?
The article also fails to provide much information on the prevalence of sexually transmitted infections (STI) in this population. Only 41% of the 133 women who had their abscess cultured were screened for STI. If these patients were infected with HIV (AIDS-defined), Chlamydia trachomatis, gonorrhea, genital herpes, or other STI pathogen, how would this have changed the data and outcomes?
Last, it is unclear whether the 10 cases of recurrent vulvar abscess identified in this study came from the inpatient or outpatient group.
A number of practice points can be gleaned from this study:
- When any patient has a vulvar abscess, culture it for aerobic and anaerobic microorganisms, including MRSA
- Because highly resistant organisms are becoming increasingly common in the community as well as the hospital, it is critical that you be familiar with your hospital’s antibiotic biogram, which delineates the organisms that are causing infection as well as susceptibility patterns
- Incision and drainage are the mainstay of management of MRSA-colonized vulvar abscess
- When deciding whether to treat a patient as an inpatient, consider medical conditions such as diabetes, HIV infection, obesity, and other conditions that compromise the immune system
- When selecting an antibiotic, choose one that includes coverage of MRSA as well as gram-negative enteric and other gram-positive organisms
- To prevent the spread of MRSA, incorporate proper hand washing and other infection-control measures into routine procedures. Also, decontaminate areas in which patients undergo incision and drainage to prevent transmission of MRSA to staff and other patients.—DAVID A. BAKER, MD
ROUNDTABLE PART 2 OF 2: Using mesh to repair prolapse: Averting, managing complications
Hear Dr Phillips discuss the key points of this series
Vaginal placement of mesh for the correction of pelvic organ prolapse is not an entirely benign procedure. As Mickey M. Karram, MD, and an expert panel discuss in this article—the second of a two-part series—complications secondary to mesh placement can be a challenge to correct and often make life miserable for patients who experience them. Here, these experts address mesh erosion, extrusion, and other serious complications; discuss ways to prevent them; and offer strategies for managing them when they arise.
In Part 1, which appeared in the January 2009 issue of OBG Management, the panel discussed the increasing use of mesh in prolapse repair—in particular, the proliferation of mesh kits.
How common is erosion?
DR. KARRAM: The literature seems to indicate that, even in the best of hands, there is an extrusion, or erosion, rate of between 5% and 17% when mesh is used. Would you agree with this statistic?
DR. LUCENTE: Not completely. The vaginal exposure rate can be as low as 2%, as reported by our center and others, when the mesh is properly placed below all histologic layers of the vaginal wall, as it is when it is “delivered” to the pelvis via the transabdominal route.1,2
At the other end of the scale, an exposure rate above 17% has been reported when mesh is improperly placed within the vaginal wall—that is, just below the mucosa, as some surgeons have described in the methodology section of their abstract or article.3,4

MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.

SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.

VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.

MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
We have found that complete, full-thickness dissection of the vaginal wall into the true pelvic space (vesicovaginal and rectovaginal), utilizing small vaginal incisions and limiting hysterectomy and the trimming of vaginal mucosa, can promote a very low vaginal-exposure rate.
DR. WALTERS: Some surgeons tell me that their own extrusion or erosion rate is lower than the published rate of 5% to 17%, but it is impossible to be certain of the long-term outcome in any patient unless she is followed carefully. The patient may consult another physician about her complications. The primary surgeon—even an expert—often does not know the actual mesh complication rate.
That said, I am sure that some surgeons are particularly adept at using mesh kits for prolapse repair, thereby keeping their mesh complication rate low. The 5% to 17% number is what most gynecologic surgeons should expect for their patients.
DR. RAZ: The complication rates are clearly underreported since very few centers of excellence report on complications and the majority of users don’t report them. Also, the reported complication rate concerns short-term erosion. I imagine that, as time passes and vaginal tissue becomes more atrophic, the incidence of erosion will increase.
Are simple measures enough to resolve erosion?
DR. KARRAM: There seems to be a general perception that most extrusions or erosions can be easily managed in the office by placing estrogen or trimming. In our experience, that approach has been successful in a minority of cases only.
What have you seen?
DR. WALTERS: At the Cleveland Clinic, as at most tertiary care referral centers, we often see the worst cases of extrusion or erosion related to mesh. Estrogen helps in some cases of simple mesh exposure, especially after sacrocolpopexy. If estrogen is going to be effective, however, the problem should clear up relatively quickly; if it isn’t effective after a month or two of therapy, estrogen is unlikely to ever be successful.
When it comes to related problems, such as ridges or strictures in the vagina, dyspareunia, penile pain with insertion, and vaginal burning pain, I have not found simple trimming and estrogen to be effective.
DR. KARRAM: It’s also unlikely that simple excision or placement of estrogen will be successful over the long term. When an extrusion or erosion occurs, we are generally seeing only the tip of the iceberg. That’s because mesh is placed in a certain plane. Although only part of the mesh may be exposed, the entire mesh is likely to be affected because it lies in the same plane.
Also, because of the special nature of vaginal flora, it is unlikely that a foreign body is going to be successfully managed by simple excision or placement of estrogen.
DR. LUCENTE: Management of vaginal exposure really depends on the size of the exposure, its location, and whether there is underlying infection or ischemia of host tissue. When the exposure is small (<1 cm in diameter) and in the midline, with the mesh lying flat below the plane of the vaginal wall, we have been very successful using a conservative approach.
However, even the tiniest of exposures needs to be surgically excised if it traverses the vaginal sulcus. Obviously, any mesh erosion into viscera such as the bladder and bowel also requires surgical intervention. Host-tissue factors always play a contributing role.
I also want to point out that the manner in which exposure is managed depends to some extent on whether the mesh was properly placed. Exposures that arise when mesh is implanted improperly are difficult to correct and usually require complete removal.
Although we, too, started off with an exposure rate around 8%, it is now very low, thanks to technical advancements.
DR. RAZ: A very small vaginal erosion of a mesh sling can sometimes be managed in the office by excision. The cases referred to our service generally involve more extensive areas of exposure that will not be resolved by local treatment.
Is risk of injury operator-dependent?
DR. KARRAM: We’re all seeing very severe complications secondary to mesh placement. Would each of you give your opinion as to whether the severe complications such as significant pain, dyspareunia, and injury of important structures are mostly technical or inherent to mesh placement. Would they happen in the best of hands?
DR. LUCENTE: The more severe complications, for the most part, are very much related to technique. Not that they cannot happen in the very best of hands, but they are extremely rare when technique is meticulous.
Over a 4-year period, after well over 1,000 transvaginal mesh surgeries at our center, we had no death, ICU admission, or transfusion, and our intraoperative complication rate was only 3%, most commonly involving simple cystotomy without long-term consequence. This compares very favorably to the nearly 12% complication rate reported recently in the CARE trial for abdominal sacral colpopexy.5
Our primary challenge today is preventing postoperative dyspareunia. Our rate of new-onset dyspareunia is approximately 3.5%. This complication is, I think, more likely to be related to the inherent material properties of mesh, such as elasticity and flexural rigidity, and to host-tissue response to the material itself.
DR. RAZ: I think that the majority of complications are operator-dependent. Thin dissection of the vaginal wall and unrecognized bladder, urethral, and vaginal perforation are the most common reasons for the complications. Mesh does not move after surgery; if there is a problem, it means that the mesh was misplaced.
Another problem is that industry, in an effort to sell more kits, is pushing physicians who are unfamiliar with the principles of pelvic reconstruction to perform this complex procedure. Repair of major vaginal prolapse is not a simple sling procedure.
In addition, there is a greater likelihood of complications in patients who have severe atrophic tissues. These patients should not be candidates for mesh reconstruction.
DR. WALTERS: Many of the complications that we see with mesh are certainly operator-dependent. For example, mesh that is placed under too much tension leaves the vagina tight and stiff, and mesh that is placed with ripples and ridges causes irregularities in the vagina that are often painful, especially during intercourse.
I do not believe that mesh “erodes” into the bladder, urethra, or rectum, but that it is placed there inadvertently and overlooked intraoperatively (FIGURES 1 and 2), Visceral erosion can occur if the primary surgeon made a cystotomy or proctotomy before proceeding with the mesh kit, and the mesh eventually wore through the repaired area.
There are also some problems that are inherent to mesh, and that occur even in the best hands and after surgeries that are performed very competently. Some mesh exposures are inevitable, as are some cases of dyspareunia and rare cases of vaginal burning and pain. In addition, I am seeing more de novo SUI [stress urinary incontinence] with anterior mesh kits. Although this is not really a complication, it does lead to dissatisfaction in patients and merits efforts to prevent it.
DR. KARRAM: Yes. With the current state of mesh, I believe pain and dyspareunia are almost inevitable in some cases.
DR. LUCENTE: Another problem that is currently underaddressed is scar plating along the surface of the mesh. Such plating forms more readily in the absence of mechanical movement or distention during the early stages of wound healing. To make a comparison, even the best reconstructive orthopedic surgeons cannot achieve optimal functional outcomes with an implant surgery without intense postoperative physical therapy, which may simply involve range of motion or movement.
Most everyone is familiar with the capsular fibrosis and contraction that develop around a breast implant if there isn’t immediate postoperative massaging of the breast tissue and implant during wound repair. I am confident that the rate of dyspareunia will decline over time if specialists in reconstructive pelvic surgery pay closer attention to optimizing vaginal length, preserving the cervix (in women with relatively shorter vaginal length), and ensuring optimal apical attachment (that is, above the ischial spine) in younger, sexually active patients.
DR. RAZ: I think it is the surgeon rather than the surgery who causes most complications. In its effort to sell kits, industry sometimes puts them in the hands of surgeons who are not well prepared for the task. This operation can be quite complex, and you cannot create a pelvic surgeon from a physician who is unfamiliar with the anatomy. If you cannot manage the potential complications, you should not perform this type of surgery.
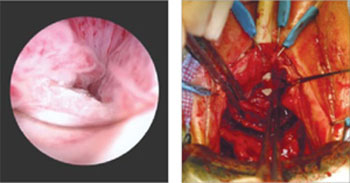
FIGURE 1 When mesh “erodes” into the urethra
Two images of mesh in the urethra. There is some uncertainty here whether mesh that has penetrated the urethra eroded through vaginal tissue or was placed there inadvertently and overlooked intraoperatively.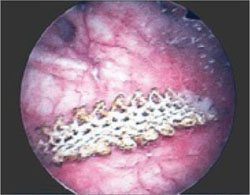
FIGURE 2 Mesh in the bladder
A segment of tension-free vaginal tape has penetrated into the bladder.
Should mesh be removed at the time of injury?
DR. KARRAM: As we discuss specific complications, let’s start with the most severe, which I would say relate to the inadvertent placement of mesh through important structures such as bowel, bladder, or ureters. If this were to happen and be diagnosed intraoperatively, what would you recommend that the surgeon do—abort the procedure or simply remove the mesh or trocar and attempt to pass it again safely?
DR. LUCENTE: That is a difficult question to answer because so much depends on various intraoperative factors.
I am much more comfortable proceeding with surgery after intraoperative bladder injury than after bowel or rectal injury. We have successfully corrected cystotomies that were small, did not encroach on the ureter, and were easily repaired without tension—and we have seen no fistula formation as a result.
The key is to maintain a high index of suspicion throughout the procedure. We have always diagnosed injuries before mesh is delivered—either during dissection or during passage of the needle or trocar. We have not experienced any ureteral injuries aside from “kinking” of one ureter, which was easily corrected with simple readjustment of the mesh.
If, at any time, we were concerned about potential infection, fistula, or a more severe complication that would be aggravated by proceeding with the operation, we would abort the procedure. However, we would be likely to proceed with an alternative operation to address the pelvic-support defect so that the patient would not awaken with intraoperative injury and no surgical treatment for her primary complaint.
We conduct informed consent in such a way as to preserve our flexibility to adapt the surgical plan to execute the reparative work that is necessary despite the development of a non–life-threatening complication during surgery. In the event of any injury to the bowel that would involve gross spillage of fecal material, of course, I would abort placement of synthetic mesh.
DR. WALTERS: If I placed one of the trocars through the bladder or bowel, I would probably remove it, reposition it, and continue with the surgery. With bladder perforation, this approach is generally no problem, but I would usually leave a Foley catheter in place for 1 week of continuous bladder drainage.
If I placed the trocar through the rectum, I would probably oversew the proctotomy, irrigate the space, and continue with the mesh repair. If I had an outright laceration in the bladder or rectum as part of the dissection, I would repair it and consider converting the surgery to prolapse repair without mesh.
The most dreaded complication: the foreshortened vagina
DR. KARRAM: It would seem that the most difficult complication to deal with is the foreshortened, firm, painful vagina. A patient who has these problems may be perceived, at times, as a pelvic “cripple.” Is this an accepted, albeit rare, complication? Or can it be avoided?
DR. LUCENTE: This is the most feared complication arising from the use of synthetic mesh. I do believe it can almost always be avoided—but I never say never. The key is to pay full attention to considerations of vaginal length before surgery, including, first, preservation of the cervix, and, second, placing the mesh loosely, properly sized, and attached with optimization of apical support to preserve vaginal length.
I also believe that use of second-generation meshes that are lighter, more elastic, and more flexible helps reduce this complication when the mesh is properly placed by a surgeon well trained in the technique.
When the vagina is foreshortened, the sooner it is revised, the better the chance that pain will resolve, whether the mesh is removed or released.
DR. RAZ: Mesh infection, capsular formation, dissection of a thin vaginal wall, and excess vaginal-wall excision lead to the short, firm, and painful vagina. The use and abuse of mesh has created a new subspecialty to manage mesh complications. The PFS syndrome (painful, firm, and short vagina) is one of the most difficult complications to treat because, in many cases, it cannot be reversed without major surgery.
DR. WALTERS: Women who have a foreshortened, firm, or painful vagina after mesh augmentation almost always need to have the mesh removed with reconstruction of the vaginal canal. I have never seen a successful outcome in this type of patient without complete or near-complete removal of the mesh.
1. van Raalte H, Lucente V, Haff R, Murphy M. Prolift: an innovative delivery system for transvaginal placement of synthetic grafts for the repair of pelvic organ prolapse. J Pelvic Med Surg .2007;13:351-360.
2. Murphy M, Raders JL, Haff R, Yeager M, Lucente V. Early U.S. experience with vaginal extraperitoneal colpopexy using propylene graft (Prolift) for the treatment of pelvic organ prolapse. J Pelvic Med Surg .2006;12:104-105.
3. Nguyen JM, Burchette RJ. Outcome after anterior vaginal prolapse repair: a randomized controlled trial. Obstet Gynecol. 2008;111:891-898.
4. Nieminen K, Hiltunen R, Heiskanen E, et al. Symptom resolution and sexual function after anterior vaginal wall repair with or without polypropylene mesh. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1611-1616.
5. Brubaker L, Nygaard I, Richter HE, et al. Two-year outcomes after sacrocolpopexy with and without Burch to prevent stress urinary incontinence. Obstet Gynecol. 2008;112:49-55.
Hear Dr Phillips discuss the key points of this series
Vaginal placement of mesh for the correction of pelvic organ prolapse is not an entirely benign procedure. As Mickey M. Karram, MD, and an expert panel discuss in this article—the second of a two-part series—complications secondary to mesh placement can be a challenge to correct and often make life miserable for patients who experience them. Here, these experts address mesh erosion, extrusion, and other serious complications; discuss ways to prevent them; and offer strategies for managing them when they arise.
In Part 1, which appeared in the January 2009 issue of OBG Management, the panel discussed the increasing use of mesh in prolapse repair—in particular, the proliferation of mesh kits.
How common is erosion?
DR. KARRAM: The literature seems to indicate that, even in the best of hands, there is an extrusion, or erosion, rate of between 5% and 17% when mesh is used. Would you agree with this statistic?
DR. LUCENTE: Not completely. The vaginal exposure rate can be as low as 2%, as reported by our center and others, when the mesh is properly placed below all histologic layers of the vaginal wall, as it is when it is “delivered” to the pelvis via the transabdominal route.1,2
At the other end of the scale, an exposure rate above 17% has been reported when mesh is improperly placed within the vaginal wall—that is, just below the mucosa, as some surgeons have described in the methodology section of their abstract or article.3,4

MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.

SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.

VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.

MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
We have found that complete, full-thickness dissection of the vaginal wall into the true pelvic space (vesicovaginal and rectovaginal), utilizing small vaginal incisions and limiting hysterectomy and the trimming of vaginal mucosa, can promote a very low vaginal-exposure rate.
DR. WALTERS: Some surgeons tell me that their own extrusion or erosion rate is lower than the published rate of 5% to 17%, but it is impossible to be certain of the long-term outcome in any patient unless she is followed carefully. The patient may consult another physician about her complications. The primary surgeon—even an expert—often does not know the actual mesh complication rate.
That said, I am sure that some surgeons are particularly adept at using mesh kits for prolapse repair, thereby keeping their mesh complication rate low. The 5% to 17% number is what most gynecologic surgeons should expect for their patients.
DR. RAZ: The complication rates are clearly underreported since very few centers of excellence report on complications and the majority of users don’t report them. Also, the reported complication rate concerns short-term erosion. I imagine that, as time passes and vaginal tissue becomes more atrophic, the incidence of erosion will increase.
Are simple measures enough to resolve erosion?
DR. KARRAM: There seems to be a general perception that most extrusions or erosions can be easily managed in the office by placing estrogen or trimming. In our experience, that approach has been successful in a minority of cases only.
What have you seen?
DR. WALTERS: At the Cleveland Clinic, as at most tertiary care referral centers, we often see the worst cases of extrusion or erosion related to mesh. Estrogen helps in some cases of simple mesh exposure, especially after sacrocolpopexy. If estrogen is going to be effective, however, the problem should clear up relatively quickly; if it isn’t effective after a month or two of therapy, estrogen is unlikely to ever be successful.
When it comes to related problems, such as ridges or strictures in the vagina, dyspareunia, penile pain with insertion, and vaginal burning pain, I have not found simple trimming and estrogen to be effective.
DR. KARRAM: It’s also unlikely that simple excision or placement of estrogen will be successful over the long term. When an extrusion or erosion occurs, we are generally seeing only the tip of the iceberg. That’s because mesh is placed in a certain plane. Although only part of the mesh may be exposed, the entire mesh is likely to be affected because it lies in the same plane.
Also, because of the special nature of vaginal flora, it is unlikely that a foreign body is going to be successfully managed by simple excision or placement of estrogen.
DR. LUCENTE: Management of vaginal exposure really depends on the size of the exposure, its location, and whether there is underlying infection or ischemia of host tissue. When the exposure is small (<1 cm in diameter) and in the midline, with the mesh lying flat below the plane of the vaginal wall, we have been very successful using a conservative approach.
However, even the tiniest of exposures needs to be surgically excised if it traverses the vaginal sulcus. Obviously, any mesh erosion into viscera such as the bladder and bowel also requires surgical intervention. Host-tissue factors always play a contributing role.
I also want to point out that the manner in which exposure is managed depends to some extent on whether the mesh was properly placed. Exposures that arise when mesh is implanted improperly are difficult to correct and usually require complete removal.
Although we, too, started off with an exposure rate around 8%, it is now very low, thanks to technical advancements.
DR. RAZ: A very small vaginal erosion of a mesh sling can sometimes be managed in the office by excision. The cases referred to our service generally involve more extensive areas of exposure that will not be resolved by local treatment.
Is risk of injury operator-dependent?
DR. KARRAM: We’re all seeing very severe complications secondary to mesh placement. Would each of you give your opinion as to whether the severe complications such as significant pain, dyspareunia, and injury of important structures are mostly technical or inherent to mesh placement. Would they happen in the best of hands?
DR. LUCENTE: The more severe complications, for the most part, are very much related to technique. Not that they cannot happen in the very best of hands, but they are extremely rare when technique is meticulous.
Over a 4-year period, after well over 1,000 transvaginal mesh surgeries at our center, we had no death, ICU admission, or transfusion, and our intraoperative complication rate was only 3%, most commonly involving simple cystotomy without long-term consequence. This compares very favorably to the nearly 12% complication rate reported recently in the CARE trial for abdominal sacral colpopexy.5
Our primary challenge today is preventing postoperative dyspareunia. Our rate of new-onset dyspareunia is approximately 3.5%. This complication is, I think, more likely to be related to the inherent material properties of mesh, such as elasticity and flexural rigidity, and to host-tissue response to the material itself.
DR. RAZ: I think that the majority of complications are operator-dependent. Thin dissection of the vaginal wall and unrecognized bladder, urethral, and vaginal perforation are the most common reasons for the complications. Mesh does not move after surgery; if there is a problem, it means that the mesh was misplaced.
Another problem is that industry, in an effort to sell more kits, is pushing physicians who are unfamiliar with the principles of pelvic reconstruction to perform this complex procedure. Repair of major vaginal prolapse is not a simple sling procedure.
In addition, there is a greater likelihood of complications in patients who have severe atrophic tissues. These patients should not be candidates for mesh reconstruction.
DR. WALTERS: Many of the complications that we see with mesh are certainly operator-dependent. For example, mesh that is placed under too much tension leaves the vagina tight and stiff, and mesh that is placed with ripples and ridges causes irregularities in the vagina that are often painful, especially during intercourse.
I do not believe that mesh “erodes” into the bladder, urethra, or rectum, but that it is placed there inadvertently and overlooked intraoperatively (FIGURES 1 and 2), Visceral erosion can occur if the primary surgeon made a cystotomy or proctotomy before proceeding with the mesh kit, and the mesh eventually wore through the repaired area.
There are also some problems that are inherent to mesh, and that occur even in the best hands and after surgeries that are performed very competently. Some mesh exposures are inevitable, as are some cases of dyspareunia and rare cases of vaginal burning and pain. In addition, I am seeing more de novo SUI [stress urinary incontinence] with anterior mesh kits. Although this is not really a complication, it does lead to dissatisfaction in patients and merits efforts to prevent it.
DR. KARRAM: Yes. With the current state of mesh, I believe pain and dyspareunia are almost inevitable in some cases.
DR. LUCENTE: Another problem that is currently underaddressed is scar plating along the surface of the mesh. Such plating forms more readily in the absence of mechanical movement or distention during the early stages of wound healing. To make a comparison, even the best reconstructive orthopedic surgeons cannot achieve optimal functional outcomes with an implant surgery without intense postoperative physical therapy, which may simply involve range of motion or movement.
Most everyone is familiar with the capsular fibrosis and contraction that develop around a breast implant if there isn’t immediate postoperative massaging of the breast tissue and implant during wound repair. I am confident that the rate of dyspareunia will decline over time if specialists in reconstructive pelvic surgery pay closer attention to optimizing vaginal length, preserving the cervix (in women with relatively shorter vaginal length), and ensuring optimal apical attachment (that is, above the ischial spine) in younger, sexually active patients.
DR. RAZ: I think it is the surgeon rather than the surgery who causes most complications. In its effort to sell kits, industry sometimes puts them in the hands of surgeons who are not well prepared for the task. This operation can be quite complex, and you cannot create a pelvic surgeon from a physician who is unfamiliar with the anatomy. If you cannot manage the potential complications, you should not perform this type of surgery.

FIGURE 1 When mesh “erodes” into the urethra
Two images of mesh in the urethra. There is some uncertainty here whether mesh that has penetrated the urethra eroded through vaginal tissue or was placed there inadvertently and overlooked intraoperatively.
FIGURE 2 Mesh in the bladder
A segment of tension-free vaginal tape has penetrated into the bladder.
Should mesh be removed at the time of injury?
DR. KARRAM: As we discuss specific complications, let’s start with the most severe, which I would say relate to the inadvertent placement of mesh through important structures such as bowel, bladder, or ureters. If this were to happen and be diagnosed intraoperatively, what would you recommend that the surgeon do—abort the procedure or simply remove the mesh or trocar and attempt to pass it again safely?
DR. LUCENTE: That is a difficult question to answer because so much depends on various intraoperative factors.
I am much more comfortable proceeding with surgery after intraoperative bladder injury than after bowel or rectal injury. We have successfully corrected cystotomies that were small, did not encroach on the ureter, and were easily repaired without tension—and we have seen no fistula formation as a result.
The key is to maintain a high index of suspicion throughout the procedure. We have always diagnosed injuries before mesh is delivered—either during dissection or during passage of the needle or trocar. We have not experienced any ureteral injuries aside from “kinking” of one ureter, which was easily corrected with simple readjustment of the mesh.
If, at any time, we were concerned about potential infection, fistula, or a more severe complication that would be aggravated by proceeding with the operation, we would abort the procedure. However, we would be likely to proceed with an alternative operation to address the pelvic-support defect so that the patient would not awaken with intraoperative injury and no surgical treatment for her primary complaint.
We conduct informed consent in such a way as to preserve our flexibility to adapt the surgical plan to execute the reparative work that is necessary despite the development of a non–life-threatening complication during surgery. In the event of any injury to the bowel that would involve gross spillage of fecal material, of course, I would abort placement of synthetic mesh.
DR. WALTERS: If I placed one of the trocars through the bladder or bowel, I would probably remove it, reposition it, and continue with the surgery. With bladder perforation, this approach is generally no problem, but I would usually leave a Foley catheter in place for 1 week of continuous bladder drainage.
If I placed the trocar through the rectum, I would probably oversew the proctotomy, irrigate the space, and continue with the mesh repair. If I had an outright laceration in the bladder or rectum as part of the dissection, I would repair it and consider converting the surgery to prolapse repair without mesh.
The most dreaded complication: the foreshortened vagina
DR. KARRAM: It would seem that the most difficult complication to deal with is the foreshortened, firm, painful vagina. A patient who has these problems may be perceived, at times, as a pelvic “cripple.” Is this an accepted, albeit rare, complication? Or can it be avoided?
DR. LUCENTE: This is the most feared complication arising from the use of synthetic mesh. I do believe it can almost always be avoided—but I never say never. The key is to pay full attention to considerations of vaginal length before surgery, including, first, preservation of the cervix, and, second, placing the mesh loosely, properly sized, and attached with optimization of apical support to preserve vaginal length.
I also believe that use of second-generation meshes that are lighter, more elastic, and more flexible helps reduce this complication when the mesh is properly placed by a surgeon well trained in the technique.
When the vagina is foreshortened, the sooner it is revised, the better the chance that pain will resolve, whether the mesh is removed or released.
DR. RAZ: Mesh infection, capsular formation, dissection of a thin vaginal wall, and excess vaginal-wall excision lead to the short, firm, and painful vagina. The use and abuse of mesh has created a new subspecialty to manage mesh complications. The PFS syndrome (painful, firm, and short vagina) is one of the most difficult complications to treat because, in many cases, it cannot be reversed without major surgery.
DR. WALTERS: Women who have a foreshortened, firm, or painful vagina after mesh augmentation almost always need to have the mesh removed with reconstruction of the vaginal canal. I have never seen a successful outcome in this type of patient without complete or near-complete removal of the mesh.
Hear Dr Phillips discuss the key points of this series
Vaginal placement of mesh for the correction of pelvic organ prolapse is not an entirely benign procedure. As Mickey M. Karram, MD, and an expert panel discuss in this article—the second of a two-part series—complications secondary to mesh placement can be a challenge to correct and often make life miserable for patients who experience them. Here, these experts address mesh erosion, extrusion, and other serious complications; discuss ways to prevent them; and offer strategies for managing them when they arise.
In Part 1, which appeared in the January 2009 issue of OBG Management, the panel discussed the increasing use of mesh in prolapse repair—in particular, the proliferation of mesh kits.
How common is erosion?
DR. KARRAM: The literature seems to indicate that, even in the best of hands, there is an extrusion, or erosion, rate of between 5% and 17% when mesh is used. Would you agree with this statistic?
DR. LUCENTE: Not completely. The vaginal exposure rate can be as low as 2%, as reported by our center and others, when the mesh is properly placed below all histologic layers of the vaginal wall, as it is when it is “delivered” to the pelvis via the transabdominal route.1,2
At the other end of the scale, an exposure rate above 17% has been reported when mesh is improperly placed within the vaginal wall—that is, just below the mucosa, as some surgeons have described in the methodology section of their abstract or article.3,4

MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.

SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.

VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.

MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
We have found that complete, full-thickness dissection of the vaginal wall into the true pelvic space (vesicovaginal and rectovaginal), utilizing small vaginal incisions and limiting hysterectomy and the trimming of vaginal mucosa, can promote a very low vaginal-exposure rate.
DR. WALTERS: Some surgeons tell me that their own extrusion or erosion rate is lower than the published rate of 5% to 17%, but it is impossible to be certain of the long-term outcome in any patient unless she is followed carefully. The patient may consult another physician about her complications. The primary surgeon—even an expert—often does not know the actual mesh complication rate.
That said, I am sure that some surgeons are particularly adept at using mesh kits for prolapse repair, thereby keeping their mesh complication rate low. The 5% to 17% number is what most gynecologic surgeons should expect for their patients.
DR. RAZ: The complication rates are clearly underreported since very few centers of excellence report on complications and the majority of users don’t report them. Also, the reported complication rate concerns short-term erosion. I imagine that, as time passes and vaginal tissue becomes more atrophic, the incidence of erosion will increase.
Are simple measures enough to resolve erosion?
DR. KARRAM: There seems to be a general perception that most extrusions or erosions can be easily managed in the office by placing estrogen or trimming. In our experience, that approach has been successful in a minority of cases only.
What have you seen?
DR. WALTERS: At the Cleveland Clinic, as at most tertiary care referral centers, we often see the worst cases of extrusion or erosion related to mesh. Estrogen helps in some cases of simple mesh exposure, especially after sacrocolpopexy. If estrogen is going to be effective, however, the problem should clear up relatively quickly; if it isn’t effective after a month or two of therapy, estrogen is unlikely to ever be successful.
When it comes to related problems, such as ridges or strictures in the vagina, dyspareunia, penile pain with insertion, and vaginal burning pain, I have not found simple trimming and estrogen to be effective.
DR. KARRAM: It’s also unlikely that simple excision or placement of estrogen will be successful over the long term. When an extrusion or erosion occurs, we are generally seeing only the tip of the iceberg. That’s because mesh is placed in a certain plane. Although only part of the mesh may be exposed, the entire mesh is likely to be affected because it lies in the same plane.
Also, because of the special nature of vaginal flora, it is unlikely that a foreign body is going to be successfully managed by simple excision or placement of estrogen.
DR. LUCENTE: Management of vaginal exposure really depends on the size of the exposure, its location, and whether there is underlying infection or ischemia of host tissue. When the exposure is small (<1 cm in diameter) and in the midline, with the mesh lying flat below the plane of the vaginal wall, we have been very successful using a conservative approach.
However, even the tiniest of exposures needs to be surgically excised if it traverses the vaginal sulcus. Obviously, any mesh erosion into viscera such as the bladder and bowel also requires surgical intervention. Host-tissue factors always play a contributing role.
I also want to point out that the manner in which exposure is managed depends to some extent on whether the mesh was properly placed. Exposures that arise when mesh is implanted improperly are difficult to correct and usually require complete removal.
Although we, too, started off with an exposure rate around 8%, it is now very low, thanks to technical advancements.
DR. RAZ: A very small vaginal erosion of a mesh sling can sometimes be managed in the office by excision. The cases referred to our service generally involve more extensive areas of exposure that will not be resolved by local treatment.
Is risk of injury operator-dependent?
DR. KARRAM: We’re all seeing very severe complications secondary to mesh placement. Would each of you give your opinion as to whether the severe complications such as significant pain, dyspareunia, and injury of important structures are mostly technical or inherent to mesh placement. Would they happen in the best of hands?
DR. LUCENTE: The more severe complications, for the most part, are very much related to technique. Not that they cannot happen in the very best of hands, but they are extremely rare when technique is meticulous.
Over a 4-year period, after well over 1,000 transvaginal mesh surgeries at our center, we had no death, ICU admission, or transfusion, and our intraoperative complication rate was only 3%, most commonly involving simple cystotomy without long-term consequence. This compares very favorably to the nearly 12% complication rate reported recently in the CARE trial for abdominal sacral colpopexy.5
Our primary challenge today is preventing postoperative dyspareunia. Our rate of new-onset dyspareunia is approximately 3.5%. This complication is, I think, more likely to be related to the inherent material properties of mesh, such as elasticity and flexural rigidity, and to host-tissue response to the material itself.
DR. RAZ: I think that the majority of complications are operator-dependent. Thin dissection of the vaginal wall and unrecognized bladder, urethral, and vaginal perforation are the most common reasons for the complications. Mesh does not move after surgery; if there is a problem, it means that the mesh was misplaced.
Another problem is that industry, in an effort to sell more kits, is pushing physicians who are unfamiliar with the principles of pelvic reconstruction to perform this complex procedure. Repair of major vaginal prolapse is not a simple sling procedure.
In addition, there is a greater likelihood of complications in patients who have severe atrophic tissues. These patients should not be candidates for mesh reconstruction.
DR. WALTERS: Many of the complications that we see with mesh are certainly operator-dependent. For example, mesh that is placed under too much tension leaves the vagina tight and stiff, and mesh that is placed with ripples and ridges causes irregularities in the vagina that are often painful, especially during intercourse.
I do not believe that mesh “erodes” into the bladder, urethra, or rectum, but that it is placed there inadvertently and overlooked intraoperatively (FIGURES 1 and 2), Visceral erosion can occur if the primary surgeon made a cystotomy or proctotomy before proceeding with the mesh kit, and the mesh eventually wore through the repaired area.
There are also some problems that are inherent to mesh, and that occur even in the best hands and after surgeries that are performed very competently. Some mesh exposures are inevitable, as are some cases of dyspareunia and rare cases of vaginal burning and pain. In addition, I am seeing more de novo SUI [stress urinary incontinence] with anterior mesh kits. Although this is not really a complication, it does lead to dissatisfaction in patients and merits efforts to prevent it.
DR. KARRAM: Yes. With the current state of mesh, I believe pain and dyspareunia are almost inevitable in some cases.
DR. LUCENTE: Another problem that is currently underaddressed is scar plating along the surface of the mesh. Such plating forms more readily in the absence of mechanical movement or distention during the early stages of wound healing. To make a comparison, even the best reconstructive orthopedic surgeons cannot achieve optimal functional outcomes with an implant surgery without intense postoperative physical therapy, which may simply involve range of motion or movement.
Most everyone is familiar with the capsular fibrosis and contraction that develop around a breast implant if there isn’t immediate postoperative massaging of the breast tissue and implant during wound repair. I am confident that the rate of dyspareunia will decline over time if specialists in reconstructive pelvic surgery pay closer attention to optimizing vaginal length, preserving the cervix (in women with relatively shorter vaginal length), and ensuring optimal apical attachment (that is, above the ischial spine) in younger, sexually active patients.
DR. RAZ: I think it is the surgeon rather than the surgery who causes most complications. In its effort to sell kits, industry sometimes puts them in the hands of surgeons who are not well prepared for the task. This operation can be quite complex, and you cannot create a pelvic surgeon from a physician who is unfamiliar with the anatomy. If you cannot manage the potential complications, you should not perform this type of surgery.

FIGURE 1 When mesh “erodes” into the urethra
Two images of mesh in the urethra. There is some uncertainty here whether mesh that has penetrated the urethra eroded through vaginal tissue or was placed there inadvertently and overlooked intraoperatively.
FIGURE 2 Mesh in the bladder
A segment of tension-free vaginal tape has penetrated into the bladder.
Should mesh be removed at the time of injury?
DR. KARRAM: As we discuss specific complications, let’s start with the most severe, which I would say relate to the inadvertent placement of mesh through important structures such as bowel, bladder, or ureters. If this were to happen and be diagnosed intraoperatively, what would you recommend that the surgeon do—abort the procedure or simply remove the mesh or trocar and attempt to pass it again safely?
DR. LUCENTE: That is a difficult question to answer because so much depends on various intraoperative factors.
I am much more comfortable proceeding with surgery after intraoperative bladder injury than after bowel or rectal injury. We have successfully corrected cystotomies that were small, did not encroach on the ureter, and were easily repaired without tension—and we have seen no fistula formation as a result.
The key is to maintain a high index of suspicion throughout the procedure. We have always diagnosed injuries before mesh is delivered—either during dissection or during passage of the needle or trocar. We have not experienced any ureteral injuries aside from “kinking” of one ureter, which was easily corrected with simple readjustment of the mesh.
If, at any time, we were concerned about potential infection, fistula, or a more severe complication that would be aggravated by proceeding with the operation, we would abort the procedure. However, we would be likely to proceed with an alternative operation to address the pelvic-support defect so that the patient would not awaken with intraoperative injury and no surgical treatment for her primary complaint.
We conduct informed consent in such a way as to preserve our flexibility to adapt the surgical plan to execute the reparative work that is necessary despite the development of a non–life-threatening complication during surgery. In the event of any injury to the bowel that would involve gross spillage of fecal material, of course, I would abort placement of synthetic mesh.
DR. WALTERS: If I placed one of the trocars through the bladder or bowel, I would probably remove it, reposition it, and continue with the surgery. With bladder perforation, this approach is generally no problem, but I would usually leave a Foley catheter in place for 1 week of continuous bladder drainage.
If I placed the trocar through the rectum, I would probably oversew the proctotomy, irrigate the space, and continue with the mesh repair. If I had an outright laceration in the bladder or rectum as part of the dissection, I would repair it and consider converting the surgery to prolapse repair without mesh.
The most dreaded complication: the foreshortened vagina
DR. KARRAM: It would seem that the most difficult complication to deal with is the foreshortened, firm, painful vagina. A patient who has these problems may be perceived, at times, as a pelvic “cripple.” Is this an accepted, albeit rare, complication? Or can it be avoided?
DR. LUCENTE: This is the most feared complication arising from the use of synthetic mesh. I do believe it can almost always be avoided—but I never say never. The key is to pay full attention to considerations of vaginal length before surgery, including, first, preservation of the cervix, and, second, placing the mesh loosely, properly sized, and attached with optimization of apical support to preserve vaginal length.
I also believe that use of second-generation meshes that are lighter, more elastic, and more flexible helps reduce this complication when the mesh is properly placed by a surgeon well trained in the technique.
When the vagina is foreshortened, the sooner it is revised, the better the chance that pain will resolve, whether the mesh is removed or released.
DR. RAZ: Mesh infection, capsular formation, dissection of a thin vaginal wall, and excess vaginal-wall excision lead to the short, firm, and painful vagina. The use and abuse of mesh has created a new subspecialty to manage mesh complications. The PFS syndrome (painful, firm, and short vagina) is one of the most difficult complications to treat because, in many cases, it cannot be reversed without major surgery.
DR. WALTERS: Women who have a foreshortened, firm, or painful vagina after mesh augmentation almost always need to have the mesh removed with reconstruction of the vaginal canal. I have never seen a successful outcome in this type of patient without complete or near-complete removal of the mesh.
1. van Raalte H, Lucente V, Haff R, Murphy M. Prolift: an innovative delivery system for transvaginal placement of synthetic grafts for the repair of pelvic organ prolapse. J Pelvic Med Surg .2007;13:351-360.
2. Murphy M, Raders JL, Haff R, Yeager M, Lucente V. Early U.S. experience with vaginal extraperitoneal colpopexy using propylene graft (Prolift) for the treatment of pelvic organ prolapse. J Pelvic Med Surg .2006;12:104-105.
3. Nguyen JM, Burchette RJ. Outcome after anterior vaginal prolapse repair: a randomized controlled trial. Obstet Gynecol. 2008;111:891-898.
4. Nieminen K, Hiltunen R, Heiskanen E, et al. Symptom resolution and sexual function after anterior vaginal wall repair with or without polypropylene mesh. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1611-1616.
5. Brubaker L, Nygaard I, Richter HE, et al. Two-year outcomes after sacrocolpopexy with and without Burch to prevent stress urinary incontinence. Obstet Gynecol. 2008;112:49-55.
1. van Raalte H, Lucente V, Haff R, Murphy M. Prolift: an innovative delivery system for transvaginal placement of synthetic grafts for the repair of pelvic organ prolapse. J Pelvic Med Surg .2007;13:351-360.
2. Murphy M, Raders JL, Haff R, Yeager M, Lucente V. Early U.S. experience with vaginal extraperitoneal colpopexy using propylene graft (Prolift) for the treatment of pelvic organ prolapse. J Pelvic Med Surg .2006;12:104-105.
3. Nguyen JM, Burchette RJ. Outcome after anterior vaginal prolapse repair: a randomized controlled trial. Obstet Gynecol. 2008;111:891-898.
4. Nieminen K, Hiltunen R, Heiskanen E, et al. Symptom resolution and sexual function after anterior vaginal wall repair with or without polypropylene mesh. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:1611-1616.
5. Brubaker L, Nygaard I, Richter HE, et al. Two-year outcomes after sacrocolpopexy with and without Burch to prevent stress urinary incontinence. Obstet Gynecol. 2008;112:49-55.
Is personal distress an important measure when assessing sexual dysfunction?
In the survey described by this study, which had a response rate of 63% (n=31,581), female heads of household 18 years of age and older were asked to evaluate their sexual function using the female short-form Changes in Sexual Functioning Questionnaire and the Female Sexual Distress Scale. The prevalence of any sexual problem was 44.2%, with the most common problems being:
- low desire (38.7%)
- low arousal (26.1%)
- orgasmic dysfunction (20.5%).
Dyspareunia was not assessed in this survey because a physical examination is required.
How the findings broke down by age
The prevalence of sexual problems increased with age:
- Among women 18 to 44 years old, 27.2% reported a problem with desire, arousal, orgasm, or a combination of the three.
- Among women 45 to 64 years old, the prevalence was 44.6%.
- Among women 65 years and older, the prevalence of one or more of these problems was 80.1%.
- highest (14.8%) among respondents 45 to 64 years old
- lowest (8.9%) among women 65 years or older
- intermediate (10.8%) in women 18 to 44 years old.
Medical problems associated with a higher prevalence of distressing problems of desire were depression, thyroid dysfunction, anxiety, and urinary incontinence.
Strengths and weaknesses of the study
The large sample size, wide age range, and use of verified instruments to measure sexual problems and related distress were all strengths of this study.
However, to increase response rates to “sensitive” questions, the authors used a research panel that was not randomly chosen. As a result, respondents may have been more health-conscious and self-aware than otherwise would have been the case; they also may have had more time to answer mailed questionnaires.
The fact that sexual problems and distress were self-reported without clinical evaluation also may have biased the findings slightly. Because the study was cross-sectional, cause and effect could not be established.
Although sexual problems are common among women in the United States, this survey confirms that distress caused by these problems is considerably less widespread. Sexual problems increase with age, but related distress is most common in women at midlife (45 to 64 years old).
Women’s health clinicians who elicit a history of sexual dysfunction should determine the level of distress that is present before deciding to address the problem.—ANDREW M. KAUNITZ, MD
1. Laumann EO, Paik A, Rosen RC. Sexual dysfunction in the United States: prevalence and predictors [published erratum appears in JAMA. 1999;281:1174]. JAMA. 1999;281:537-544.
In the survey described by this study, which had a response rate of 63% (n=31,581), female heads of household 18 years of age and older were asked to evaluate their sexual function using the female short-form Changes in Sexual Functioning Questionnaire and the Female Sexual Distress Scale. The prevalence of any sexual problem was 44.2%, with the most common problems being:
- low desire (38.7%)
- low arousal (26.1%)
- orgasmic dysfunction (20.5%).
Dyspareunia was not assessed in this survey because a physical examination is required.
How the findings broke down by age
The prevalence of sexual problems increased with age:
- Among women 18 to 44 years old, 27.2% reported a problem with desire, arousal, orgasm, or a combination of the three.
- Among women 45 to 64 years old, the prevalence was 44.6%.
- Among women 65 years and older, the prevalence of one or more of these problems was 80.1%.
- highest (14.8%) among respondents 45 to 64 years old
- lowest (8.9%) among women 65 years or older
- intermediate (10.8%) in women 18 to 44 years old.
Medical problems associated with a higher prevalence of distressing problems of desire were depression, thyroid dysfunction, anxiety, and urinary incontinence.
Strengths and weaknesses of the study
The large sample size, wide age range, and use of verified instruments to measure sexual problems and related distress were all strengths of this study.
However, to increase response rates to “sensitive” questions, the authors used a research panel that was not randomly chosen. As a result, respondents may have been more health-conscious and self-aware than otherwise would have been the case; they also may have had more time to answer mailed questionnaires.
The fact that sexual problems and distress were self-reported without clinical evaluation also may have biased the findings slightly. Because the study was cross-sectional, cause and effect could not be established.
Although sexual problems are common among women in the United States, this survey confirms that distress caused by these problems is considerably less widespread. Sexual problems increase with age, but related distress is most common in women at midlife (45 to 64 years old).
Women’s health clinicians who elicit a history of sexual dysfunction should determine the level of distress that is present before deciding to address the problem.—ANDREW M. KAUNITZ, MD
In the survey described by this study, which had a response rate of 63% (n=31,581), female heads of household 18 years of age and older were asked to evaluate their sexual function using the female short-form Changes in Sexual Functioning Questionnaire and the Female Sexual Distress Scale. The prevalence of any sexual problem was 44.2%, with the most common problems being:
- low desire (38.7%)
- low arousal (26.1%)
- orgasmic dysfunction (20.5%).
Dyspareunia was not assessed in this survey because a physical examination is required.
How the findings broke down by age
The prevalence of sexual problems increased with age:
- Among women 18 to 44 years old, 27.2% reported a problem with desire, arousal, orgasm, or a combination of the three.
- Among women 45 to 64 years old, the prevalence was 44.6%.
- Among women 65 years and older, the prevalence of one or more of these problems was 80.1%.
- highest (14.8%) among respondents 45 to 64 years old
- lowest (8.9%) among women 65 years or older
- intermediate (10.8%) in women 18 to 44 years old.
Medical problems associated with a higher prevalence of distressing problems of desire were depression, thyroid dysfunction, anxiety, and urinary incontinence.
Strengths and weaknesses of the study
The large sample size, wide age range, and use of verified instruments to measure sexual problems and related distress were all strengths of this study.
However, to increase response rates to “sensitive” questions, the authors used a research panel that was not randomly chosen. As a result, respondents may have been more health-conscious and self-aware than otherwise would have been the case; they also may have had more time to answer mailed questionnaires.
The fact that sexual problems and distress were self-reported without clinical evaluation also may have biased the findings slightly. Because the study was cross-sectional, cause and effect could not be established.
Although sexual problems are common among women in the United States, this survey confirms that distress caused by these problems is considerably less widespread. Sexual problems increase with age, but related distress is most common in women at midlife (45 to 64 years old).
Women’s health clinicians who elicit a history of sexual dysfunction should determine the level of distress that is present before deciding to address the problem.—ANDREW M. KAUNITZ, MD
1. Laumann EO, Paik A, Rosen RC. Sexual dysfunction in the United States: prevalence and predictors [published erratum appears in JAMA. 1999;281:1174]. JAMA. 1999;281:537-544.
1. Laumann EO, Paik A, Rosen RC. Sexual dysfunction in the United States: prevalence and predictors [published erratum appears in JAMA. 1999;281:1174]. JAMA. 1999;281:537-544.