User login
Does sildenafil improve antidepressant-related sexual dysfunction in women?
Assessment of female sexual dysfunction is difficult
The four areas of potential abnormality in sexual function, as described in the Diagnostic and Statistical Manual of Mental Disorders–IV, include desire, arousal, orgasm, and pain and are more difficult to differentiate in women than in men. In addition, the variability of the hormonal environment makes it challenging to study the physiologic mechanisms of some of these conditions.
Nurnberg and colleagues sought to eliminate most of the variables by including only premenopausal women who had normal hormone levels at baseline (mean estradiol level, 67 pg/mL; FSH
Selective and nonselective SRIs have been associated with sexual side effects in up to 70% of women—specifically, delayed or absent orgasm, decreased arousal and lubrication, and diminished libido.
Details of the study
This prospective, randomized, double-blind, placebo-controlled trial aimed to assess the efficacy of 50 to 100 mg of sildenafil in reversing new-onset sexual dysfunction in women adequately treated for their underlying major depression. Ninety-eight women (average age, 37 years) were randomized, with 37 and 39 completing the 8-week trial in the placebo and active treatment arms, respectively.
Utilizing an intent-to-treat statistical analysis, the authors found that women taking an average dosage of 91.7 mg of sildenafil experienced significant improvement in overall sexual satisfaction, lubrication, and ability to reach orgasm. Four different scales of sexual function were utilized in the analysis, each of which has been well validated in previous studies. Of note, 28% of women taking sildenafil and 73% of those taking placebo reported no improvement in sexual function throughout the 8-week study.
In this study, responders had slightly (and statistically significantly) higher levels of both free testosterone (1.44 pg/mL vs 1.04 pg/mL) and thyroxine (8.83 ng/dL vs 7.75 ng/dL) than nonresponders in the treatment arm.
These findings are the first to demonstrate efficacy in women
Physiologically, these findings make sense. In estrogen-replete women, type-5 phosphodiesterase inhibitors are thought to increase blood flow, engorgement, and lubrication to the genitalia, resulting in improved sensation and arousal.
By eliminating or controlling for many of the confounders in earlier studies of sildenafil in women, this well-designed trial has demonstrated, for the first time, significant improvement in women who have sexual complaints related to antidepressant medication.
This study shows that treatment with one of the type-5 phosphodiesterase inhibitors for arousal or orgasmic problems, or both, makes sense. It is critical to ensure that baseline hormone levels are well within normal premenopausal ranges and that underlying depression or relationship issues are not contributing to low libido if you are to expect improvement.
Women who have new-onset sexual dysfunction after taking SRIs are most likely to respond. Look for subtle thyroid problems or low testosterone levels in nonresponders.—BARBARA S. LEVY, MD
Assessment of female sexual dysfunction is difficult
The four areas of potential abnormality in sexual function, as described in the Diagnostic and Statistical Manual of Mental Disorders–IV, include desire, arousal, orgasm, and pain and are more difficult to differentiate in women than in men. In addition, the variability of the hormonal environment makes it challenging to study the physiologic mechanisms of some of these conditions.
Nurnberg and colleagues sought to eliminate most of the variables by including only premenopausal women who had normal hormone levels at baseline (mean estradiol level, 67 pg/mL; FSH
Selective and nonselective SRIs have been associated with sexual side effects in up to 70% of women—specifically, delayed or absent orgasm, decreased arousal and lubrication, and diminished libido.
Details of the study
This prospective, randomized, double-blind, placebo-controlled trial aimed to assess the efficacy of 50 to 100 mg of sildenafil in reversing new-onset sexual dysfunction in women adequately treated for their underlying major depression. Ninety-eight women (average age, 37 years) were randomized, with 37 and 39 completing the 8-week trial in the placebo and active treatment arms, respectively.
Utilizing an intent-to-treat statistical analysis, the authors found that women taking an average dosage of 91.7 mg of sildenafil experienced significant improvement in overall sexual satisfaction, lubrication, and ability to reach orgasm. Four different scales of sexual function were utilized in the analysis, each of which has been well validated in previous studies. Of note, 28% of women taking sildenafil and 73% of those taking placebo reported no improvement in sexual function throughout the 8-week study.
In this study, responders had slightly (and statistically significantly) higher levels of both free testosterone (1.44 pg/mL vs 1.04 pg/mL) and thyroxine (8.83 ng/dL vs 7.75 ng/dL) than nonresponders in the treatment arm.
These findings are the first to demonstrate efficacy in women
Physiologically, these findings make sense. In estrogen-replete women, type-5 phosphodiesterase inhibitors are thought to increase blood flow, engorgement, and lubrication to the genitalia, resulting in improved sensation and arousal.
By eliminating or controlling for many of the confounders in earlier studies of sildenafil in women, this well-designed trial has demonstrated, for the first time, significant improvement in women who have sexual complaints related to antidepressant medication.
This study shows that treatment with one of the type-5 phosphodiesterase inhibitors for arousal or orgasmic problems, or both, makes sense. It is critical to ensure that baseline hormone levels are well within normal premenopausal ranges and that underlying depression or relationship issues are not contributing to low libido if you are to expect improvement.
Women who have new-onset sexual dysfunction after taking SRIs are most likely to respond. Look for subtle thyroid problems or low testosterone levels in nonresponders.—BARBARA S. LEVY, MD
Assessment of female sexual dysfunction is difficult
The four areas of potential abnormality in sexual function, as described in the Diagnostic and Statistical Manual of Mental Disorders–IV, include desire, arousal, orgasm, and pain and are more difficult to differentiate in women than in men. In addition, the variability of the hormonal environment makes it challenging to study the physiologic mechanisms of some of these conditions.
Nurnberg and colleagues sought to eliminate most of the variables by including only premenopausal women who had normal hormone levels at baseline (mean estradiol level, 67 pg/mL; FSH
Selective and nonselective SRIs have been associated with sexual side effects in up to 70% of women—specifically, delayed or absent orgasm, decreased arousal and lubrication, and diminished libido.
Details of the study
This prospective, randomized, double-blind, placebo-controlled trial aimed to assess the efficacy of 50 to 100 mg of sildenafil in reversing new-onset sexual dysfunction in women adequately treated for their underlying major depression. Ninety-eight women (average age, 37 years) were randomized, with 37 and 39 completing the 8-week trial in the placebo and active treatment arms, respectively.
Utilizing an intent-to-treat statistical analysis, the authors found that women taking an average dosage of 91.7 mg of sildenafil experienced significant improvement in overall sexual satisfaction, lubrication, and ability to reach orgasm. Four different scales of sexual function were utilized in the analysis, each of which has been well validated in previous studies. Of note, 28% of women taking sildenafil and 73% of those taking placebo reported no improvement in sexual function throughout the 8-week study.
In this study, responders had slightly (and statistically significantly) higher levels of both free testosterone (1.44 pg/mL vs 1.04 pg/mL) and thyroxine (8.83 ng/dL vs 7.75 ng/dL) than nonresponders in the treatment arm.
These findings are the first to demonstrate efficacy in women
Physiologically, these findings make sense. In estrogen-replete women, type-5 phosphodiesterase inhibitors are thought to increase blood flow, engorgement, and lubrication to the genitalia, resulting in improved sensation and arousal.
By eliminating or controlling for many of the confounders in earlier studies of sildenafil in women, this well-designed trial has demonstrated, for the first time, significant improvement in women who have sexual complaints related to antidepressant medication.
This study shows that treatment with one of the type-5 phosphodiesterase inhibitors for arousal or orgasmic problems, or both, makes sense. It is critical to ensure that baseline hormone levels are well within normal premenopausal ranges and that underlying depression or relationship issues are not contributing to low libido if you are to expect improvement.
Women who have new-onset sexual dysfunction after taking SRIs are most likely to respond. Look for subtle thyroid problems or low testosterone levels in nonresponders.—BARBARA S. LEVY, MD
ROUNDTABLE: PART 1 OF 2: Using mesh to repair prolapse calls for more than a kit—it takes skill
MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.
SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.
VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.
MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
Mesh kits for repairing prolapse are proliferating like crazy, just as they did for midurethral sling procedures. But mesh augmentation of prolapse surgeries requires more than a prepackaged assortment of tools and materials. In this article, moderator Mickey M. Karram, MD, and a panel of nationally recognized urogynecologists and urologists describe the literature on mesh augmentation and discuss indications, contraindications, techniques, applicable cases, and the considerable training required.
In Part 2, which will appear in the February issue of OBG Management, the panel tackles the thorny topic of complications, including erosion, extrusion, foreshortening of the vagina, dyspareunia, and pain. Their discussion focuses on ways to avoid these problems, and methods for correcting them.
Do we have enough data?
DR. KARRAM: To start, let’s quickly review the peer-reviewed literature on the use of mesh augmentation during surgery for pelvic organ prolapse.
DR. WALTERS: Until recently, most data concerned open abdominal sacrocolpopexy (ASC) using polypropylene or Merseline mesh. There is significant clinical experience with this operation, and multiple cohort studies show long-term cure rates of 78% to 100% for apical prolapse.1
At least two randomized controlled trials have compared open ASC with sutured vaginal colpopexy procedures, and ASC is certainly equal to—perhaps better than—all transvaginal sutured repairs.2,3
With ASC, most recurrences affect the distal half of the vagina and involve one or more of the following:
- anterior or posterior vaginal wall prolapse (or both)
- stress urinary incontinence (SUI)
- distal rectocele.1,3
Mesh erosion occurs in 3.4% of cases and is usually easily managed.1 Other complications, including bowel injury, tend to be related to access, regardless of whether the operation is performed via laparotomy or laparoscopy.
Robotic sacrocolpopexy has become popular in recent years, and we will probably see data on this approach as we gain experience.
When it comes to vaginal mesh kits, the peer-reviewed literature is just beginning to expand, with many studies being presented at international meetings. For anterior and, possibly, apical vaginal prolapse, the cure rate after use of a mesh kit appears to be as high as, or higher than, the rate for sutured repairs.4 This high rate of anatomic cure is balanced somewhat by additional cost and complications involving mesh and the kits.
For posterior vaginal wall prolapse and rectocele, I firmly believe, based on our research and that of others, that sutured repairs are superior to graft-augmented surgery.5
DR. KARRAM: What are the indications and contraindications for mesh augmentation of prolapse repair ( FIGURES 1 and 2 )?
DR. LUCENTE: I believe mesh is indicated in any patient in need of surgical repair of pelvic organ prolapse who is seeking optimal durability and is willing to accept the known risks of the surgery.
The issue becomes more complex when it comes to contraindications. Absolute contraindications are fairly obvious; they include medically unstable patients and those who may have an inactive infectious process within the pelvis or even undiagnosed abnormal uterine bleeding.
At our center, because the potential for dyspareunia and pelvic discomfort is our biggest concern, we have developed a profile of the patient who is more likely to develop these complaints. The profile includes any patient who has a chronic pain disorder of any type, but especially chronic pelvic pain disorders such as endometriosis and vulvodynia. Other risk factors appear to be a history of pelvic surgery involving any permanent material, suture or mesh, and young age.
So if we have a patient in her late 30s who has undergone reconstructive surgery using permanent sutures and who has an element of chronic pelvic pain, we would counsel her strongly to consider surgical options other than the use of synthetic mesh.
DR. WALTERS: The main indications for mesh-augmented prolapse repair are recurrent posthysterectomy vaginal vault prolapse, for which I usually perform ASC, and recurrent cystocele or anteriorapical prolapse, for which I use one of the anterior mesh kits.
I still think sutured repairs—by that, I mean uterosacral ligament or sacrospinous colpopexy with sutured rectocele repair—work best for recurrent posterior wall and posteriorapical prolapse. I don’t use mesh augmentation for rectocele.
The main contraindication to mesh augmentation, as I see it, is a history of mesh complications. If I am repairing a mesh complication such as erosion or pain, I do not place another mesh.
Medical issues that might increase mesh complications, such as diabetes, steroid use, or severe vaginal atrophy, would, at the very least, make me consider carefully whether mesh augmentation is appropriate. The literature is not clear on this, so mesh could still be used if the surgeon thinks it is necessary.
DR. KARRAM: I haven’t found a definitive indication for mesh augmentation. We have used biologic meshes empirically, but I am not convinced that they really add long-term durability, regardless of whether they are used in the anterior or posterior vaginal segment.
Our published durability rate for traditional suture-type repairs is in the range of 85% at 5 years out.6 Even if I assumed that mesh would give me 100% 5-year durability, this rate would have to be at the expense of some erosion, pain, and other complications unique to mesh. I do not think that the potential improvement in durability is worth these potential complications.
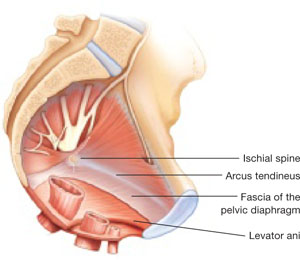
FIGURE 1 When the pelvic support system is intact, prolapse is rare
In the normal pelvis, organs are supported by a complex web of muscles, fascia, and connective tissue. 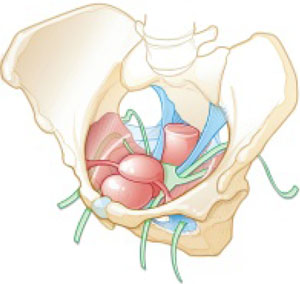
FIGURE 2 Mesh augmentation seeks to enhance the durability of repair
One type of mesh in final position. Mesh-augmented repair restores the vaginal apex and lends support to the walls of the vagina.
DR. KARRAM: If you are doing a lot of mesh repairs, you are obviously content with the results and feel that the few complications you are seeing are outweighed by the advantages mesh confers. How do you avoid extrusion and avert creation of a painful vagina?
DR. RAZ: Most of our cases are recurrent prolapse after failed vaginal or abdominal repair. I am indeed using a significant amount of soft polypropylene mesh for reconstructive procedures. As with the use of any other synthetic material, low-grade infection can develop after a few weeks or months. I use copious irrigation with antibiotic solution during reconstruction.
To avoid extrusion, I perform deep, rather than superficial, dissection of the vaginal wall to allow for better coverage of the mesh. For posterior mesh reconstruction, I cover the mesh with pararectal fascia to prevent erosion.
For mesh-augmented procedures, I cut the mesh myself in the operating room ( FIGURE 3 ). For a sling, I use a 10 cm × 1 cm soft polypropylene mesh. For a grade 3 or 4 cystocele, I use a trapezoid of soft polypropylene mesh with several points of fixation:
- at the sacrouterine ligament
- lateral to the obturator fascia
- distal to the bladder neck.
I always repair the vault at the same time.
For vault prolapse, I use a segment of soft polypropylene mesh in the shape of an apron with two arms (1 cm × 4 cm) and a central segment (4 cm × 7 cm). I support the vault using number 1-0 delayed absorbable suture and mesh. From outside the vaginal wall, in the posterolateral deep vaginal wall (inside the peritoneum), I incorporate the origin of the sacrouterine ligament and one arm of the mesh in the groove between the colon and levator ani, 15 cm from the introitus. I bring the suture 1 cm from the original entrance. A separate set of sutures brings the perirectal fascia together with the sacrouterine ligaments and perivesical fascia to close the peritoneal cavity. I tie the vault-suspension sutures, providing support to the cuff in a high posterior position (12 to 15 cm from the introitus).
In selected cases of significant recurrent rectocele, I use a rectangle of soft polypropylene mesh anchored to the origin of the sacrouterine ligament and distal to the perineal membrane. The mesh is covered by the pararectal fascia.
We have not seen vaginal, urethral, or bladder erosion in 1,800 cases of our distal urethral Prolene sling procedure using 10 cm × 1 cm soft mesh. In patients who have significant cystocele, vault prolapse, and recurrent rectocele, our vaginal erosion rate is 3%. We have never encountered rectal, bladder, or bowel perforation using our technique.
DR. LUCENTE: We often use mesh and are more than simply content with our results—we are extremely pleased, and so are our patients. Having said that, our techniques have definitely evolved over the past few years, as we’ve focused on how to decrease exposure and, more recently, optimize sexual function and vaginal comfort.
First, to avoid exposure, the most critical step is precise hydrodissection and distention of the true vesicovaginal space. This step can only be achieved through careful tactile guidance of the needle tip into the space, where it should remain while hydrodissection is performed. Always remember, sharp dissection “follows” hydrodissection. If you place the needle bevel within the vaginal wall, you will “split” the vaginal wall—as during standard colporrhaphy—which will lead to a high exposure rate.
Second, to avoid dyspareunia, it’s essential to pay close attention to POP-Q measurements, especially vaginal length, to ensure that the reconstruction restores the same length without foreshortening. This approach entails leaving the cervix in most patients who have a shorter vagina, and making sure that the mesh is secured above the ischial spine in younger, sexually active patients who have demonstrated a higher risk of postoperative deep, penetrating dyspareunia, compared with older, less sexually active patients.
Also paramount is to ensure that you have manually displaced the vagina inwardly as much as possible before deploying or setting the mesh. If you simply try to suture secure the mesh with the vagina incised open, without the ability to deploy the mesh with a closed, displaced vagina (to mimic deep penetration), it is difficult, if not impossible, to properly set the mesh for optimal comfort.
In the early days of midurethral pubovaginal slings using polypropylene, the adage was “looser is better than tighter.” This is even truer for transvaginal mesh.
DR. KARRAM: Dr. Walters, please describe your current surgical procedure of choice without mesh and explain why you haven’t adopted mesh for routine repairs.
DR. WALTERS: About 20% of my prolapse surgeries—usually for posthysterectomy or recurrent vaginal vault prolapse—involve ASC with placement of polypropylene mesh. I perform most of these cases through a Pfannenstiel incision, but I’ve also done them laparoscopically. Several of my partners perform ASC laparoscopically and robotically.
For the other 80% of my patients who have prolapse, I perform repairs transvaginally, usually using high bilateral uterosacralligament vaginal-vault suspension. We have learned to suture higher and slightly more medial on the uterosacral ligaments to attain greater vaginal depth and minimize ureteral obstruction. We use two or three sutures on each uterosacral ligament, usually a combination of permanent and delayed absorbable sutures.
I am also performing more sacrospinous ligament suspensions because this operation is being studied by the Pelvic Floor Disorders Network. Properly performed, it is an excellent surgery for apical prolapse. But, as with most of our surgeries for prolapse, recurrent anterior wall prolapse remains a problem.
Like you, Dr. Karram, we’ve studied our group’s anatomic and functional outcomes very carefully for more than 10 years and are mostly satisfied with our cure and complication rates. Although our anatomic outcomes with these surgeries are not always perfect, our reoperation rate for prolapse is only about 5%, with a high level of satisfaction in 88% to 92% of patients.
DR. RAZ: Unaugmented reconstruction fails in more than 30% of cases. Some patients who have significant prolapse and attenuated tissue think that this tissue will become healthier or stronger after reconstructive surgery, but that isn’t the case. In these situations, excision and plication make no clinical sense.
The problem is that we have yet to identify the ideal surrogate for poor-quality tissue. Most of us use polypropylene mesh in different variations. We need a better material that will be nonimmunogenic, well tolerated, and easily incorporated without erosion. Xenograft-like derivatives of dermis, or allografts such as cadaveric fascia, have failed over the long term because the body reabsorbs the graft without forming any new connective tissue.
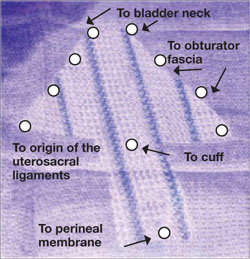
FIGURE 3 Mesh can be cut in the OR to custom-fit a patient
Hand-cut mesh and points of placement.
PHOTO: SHLOMO RAZ, MD
Is a kit a valuable aid?
DR. KARRAM: If a surgeon wants to augment a repair, what are the advantages of a packaged mesh kit, compared with simply cutting the mesh and performing surgery without a kit?
DR. WALTERS: The advantages of a packaged mesh kit are the convenience involved and the ability to consistently perform the same operation with the same product. That facilitates learning, teaching, and research. It also helps us understand the published literature a little better because “custom” prolapse repairs are operator-dependent and difficult to apply generally to a population of surgeons.
These advantages are most clearly apparent with midurethral sling mesh kits, which have almost revolutionized surgery for stress incontinence. I don’t believe mesh kits for prolapse are there yet, but they certainly have potential.
DR. RAZ: I’m opposed to the use of kits. They are industry-driven. One company has made $1 billion selling them. Imagine a patient who undergoes placement of a sling kit ($1,000), cystocele kit ($1,500), and posterior mesh kit ($1,500). How can our healthcare system sustain this burden, especially when there is no real evidence that a kit improves the operation, and given the incredible complication rate that we see?
Moreover, the kits contain a single-use needle and passer and a precut segment of polypropylene mesh. But every patient is different and requires a unique size or shape of mesh. I don’t believe that a surgeon who knows pelvic anatomy needs a kit to perform mesh-augmented reconstruction. We can buy the same segment of mesh for $200 to $400, cut it as needed, and perform the same operation advertised by industry.
For surgeons who prefer a kit, the tools that are included should be made reusable.
DR. LUCENTE: In my opinion, the primary advantage of a commercially available transvaginal mesh delivery system—notice, I avoided the word “kit,” because I think there are plenty of negative connotations associated with it—is the ability to deliver the mesh in a “tension”-free manner.
One alternative that many people pursue is cutting the mesh to size and using sutures to hold it in place while tissue ingrowth occurs. However, the hernia literature suggests that suturing mesh in place increases the risk of postoperative discomfort at the site of implantation. The true cause of the discomfort remains unclear, but it is thought to arise from nerve tethering or traction at the pre-committed points of attachment before the host tissue and mesh interface have adjusted or settled with tissue ingrowth.
All neuropathic complications of mesh implantation have been shown in the current hernia literature to be increased with the use of sutures.7 Also, as previously mentioned, it is extremely difficult to set or adjust the mesh with the vaginal incision remaining “open,” which is a downside to suture techniques.
What training is necessary to use a kit?
DR. KARRAM: Mesh kits are aggressively promoted by industry, with close to half a dozen different kits to be available soon. What is the minimum amount of training one should have before utilizing these kits?
DR. WALTERS: The surgeon should at least know how to perform traditional sutured prolapse repairs and SUI surgery and be able to perform cystoscopy. Ideally, the surgeon should undergo training on a cadaver with a skilled and experienced user of the mesh kit. The surgeon also should carefully review the risks and benefits of mesh kits with the patient and inform the patient that he or she is in the early learning curve of a particular surgery. The informed patient should have a right to refuse mesh-augmented prolapse surgery after the consent process.
DR. LUCENTE: I’m glad you asked this question. I strongly believe that surgical expertise and proficiency within gynecology need to be more effectively addressed by us all. We have a situation in our field in which techniques and technology are widening the gap between what is possible and what the surgeon is comfortable doing safely.
It’s incumbent on all of us, especially those who are in a leadership position as a chairperson or chief of a division, to work with our physician staff and faculty to optimize surgical skill and patient outcomes, including safety, with new technologies.
As for the minimal amount of training needed, that’s extremely variable. It depends on the current skill set of the physician and his or her ability to pick up the mechanics of the surgery as it is taught through a cadaver lab or preceptorship. It’s regrettable that some physicians lack the objectivity and insight to judge their own skill set. This, again, is the time for a chairperson or chief of a division to step up to the plate and ensure proper credentialing and demonstration of proficiency.
It is unrealistic to expect industry to decide who should or should not utilize this truly breakthrough technology. That is our responsibility as physicians.
DR. KARRAM: At a minimum, I think any surgeon utilizing a kit should have a firm understanding of pelvic floor anatomy and experience performing traditional repairs:
- intraperitoneal procedures such as Mc-Call culdoplasty and uterosacral suspension
- sacrospinous suspension
- retropubic procedures and anti-incontinence operations such as pubovaginal slings.
This three-dimensional understanding of the pelvic floor is mandatory if one is to assume that blind passage of trocars through potentially dangerous spaces is the wave of the future.
DR. RAZ: You need to be a pelvic surgeon, know your anatomy, and know how to manage complications if you are going to use one of these kits. You should stick to the surgery that works best in your hands. Industry cannot teach you to be a good pelvic surgeon; it takes lifelong experience.
DR. KARRAM: If you have a patient who is sexually inactive with pelvic organ prolapse, would you prefer a mesh repair or an obliterative procedure? And why?
DR. WALTERS: If the patient is sexually inactive—especially if she is older and definitely will not be in the future—it makes absolutely no sense to perform a mesh-augmented repair. A traditional, somewhat tight, sutured repair works fine in this setting and carries very low risk.
In fact, our group and others have found that, in carefully selected patients, partial colpectomy and colpocleisis procedures (without grafts) have among the highest cure and satisfaction rates of all surgeries we perform for prolapse; they also have relatively low risk.8 Recurrent prolapse after an obliterative procedure is rare; most of the dissatisfaction relates to postoperative voiding difficulties or persistent or de novo urinary incontinence.
DR. KARRAM: I also prefer an obliterative procedure. I see no reason to bring in the cost and potential for complications that mesh repair entails. An obliterative procedure should produce an anatomic success rate close to 100%, with minimal complications. It also can be performed quickly with minimal anesthesia and convalescence.
DR. LUCENTE: My response is based on a clinical study that my associate, Dr. Miles Murphy, has performed, comparing a transvaginal mesh procedure with a LaForte operation for severe pelvic organ prolapse.9 Both patient groups were well satisfied with the result, and success rates were comparable. However, the group that underwent the transvaginal mesh procedure had a shorter operative time.
As a result of these studies, we tend to prefer transvaginal mesh repair. Even though the woman may be sexually inactive, the procedure preserves vaginal function, and we all know that life has a way of being unpredictable. Her situation may change so that she once again desires sexual function.
However, for a very elderly woman—one in her late 80s or 90s—who has severe or extreme prolapse with a very large procidentia and vaginal length measuring, say, 13 cm beyond the introitus, I do prefer an obliterative procedure.
DR. RAZ: I agree. I would not offer a sexually inactive patient an obliterative procedure. You never know what the future will hold.
Mesh repair can be performed safely, provided the surgeon has good knowledge of anatomic landmarks and knows how to manage any potential complications that may arise.
1. Nygaard IE, McCreery R, Brubaker L, et al. Pelvic Floor Disorders Network. Abdominal sacrocolpopexy: a comprehensive review. Obstet Gynecol. 2004;104:805-823.
2. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421;discussion 1421-1422.
3. Maher CF, Qatawneh AM, Dwyer PL, Carey MP, Cornish A, Schluter PJ. Abdominal sacral colpopexy or vaginal sacrospinous colpopexy for vaginal vault prolapse: a prospective randomized study. Am J Obstet Gynecol. 2004;190:20-26.
4. Murphy M. Society of Gynecologic Surgeons Systematic Review Group. Clinical practice guidelines on vaginal graft use from the Society of Gynecologic Surgeons. Obstet Gynecol. 2008;112:1123-1130.
5. Paraiso MF, Barber MD, Muir TW, Walters MD. Rectocele repair: a randomized trial of three surgical procedures including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
6. Silva WA, Pauls RN, Segal JL, Rooney CM, Kleeman SD, Karram MM. Uterosacral ligament vault suspension: five-year outcomes. Obstet Gynecol 2006;108:255-263.
7. EU Hernia Trialists Collaboration. Repair of groin hernia with synthetic mesh: meta-analysis of randomized controlled trials. Ann Surg. 2002;235:322-332.
8. Barber MD, Amundsen C, Paraiso MFR, Weidner A, Romero A, Walters MD. Quality of life after surgery for genital prolapse in elderly women: obliterative and reconstructive surgery. Int Urogynecol J. 2007;18:799-806.
9. Murphy M, van Raalte H, Mercurio E, Haff R, Wiseman B, Lucente VR. Incontinence-related quality of life and sexual function following the tension-free vaginal tape vs the “inside-out” tension-free vaginal tape obturator. Int Urogynecol J. 2008;19:481-487.
MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.
SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.
VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.
MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
Mesh kits for repairing prolapse are proliferating like crazy, just as they did for midurethral sling procedures. But mesh augmentation of prolapse surgeries requires more than a prepackaged assortment of tools and materials. In this article, moderator Mickey M. Karram, MD, and a panel of nationally recognized urogynecologists and urologists describe the literature on mesh augmentation and discuss indications, contraindications, techniques, applicable cases, and the considerable training required.
In Part 2, which will appear in the February issue of OBG Management, the panel tackles the thorny topic of complications, including erosion, extrusion, foreshortening of the vagina, dyspareunia, and pain. Their discussion focuses on ways to avoid these problems, and methods for correcting them.
Do we have enough data?
DR. KARRAM: To start, let’s quickly review the peer-reviewed literature on the use of mesh augmentation during surgery for pelvic organ prolapse.
DR. WALTERS: Until recently, most data concerned open abdominal sacrocolpopexy (ASC) using polypropylene or Merseline mesh. There is significant clinical experience with this operation, and multiple cohort studies show long-term cure rates of 78% to 100% for apical prolapse.1
At least two randomized controlled trials have compared open ASC with sutured vaginal colpopexy procedures, and ASC is certainly equal to—perhaps better than—all transvaginal sutured repairs.2,3
With ASC, most recurrences affect the distal half of the vagina and involve one or more of the following:
- anterior or posterior vaginal wall prolapse (or both)
- stress urinary incontinence (SUI)
- distal rectocele.1,3
Mesh erosion occurs in 3.4% of cases and is usually easily managed.1 Other complications, including bowel injury, tend to be related to access, regardless of whether the operation is performed via laparotomy or laparoscopy.
Robotic sacrocolpopexy has become popular in recent years, and we will probably see data on this approach as we gain experience.
When it comes to vaginal mesh kits, the peer-reviewed literature is just beginning to expand, with many studies being presented at international meetings. For anterior and, possibly, apical vaginal prolapse, the cure rate after use of a mesh kit appears to be as high as, or higher than, the rate for sutured repairs.4 This high rate of anatomic cure is balanced somewhat by additional cost and complications involving mesh and the kits.
For posterior vaginal wall prolapse and rectocele, I firmly believe, based on our research and that of others, that sutured repairs are superior to graft-augmented surgery.5
DR. KARRAM: What are the indications and contraindications for mesh augmentation of prolapse repair ( FIGURES 1 and 2 )?
DR. LUCENTE: I believe mesh is indicated in any patient in need of surgical repair of pelvic organ prolapse who is seeking optimal durability and is willing to accept the known risks of the surgery.
The issue becomes more complex when it comes to contraindications. Absolute contraindications are fairly obvious; they include medically unstable patients and those who may have an inactive infectious process within the pelvis or even undiagnosed abnormal uterine bleeding.
At our center, because the potential for dyspareunia and pelvic discomfort is our biggest concern, we have developed a profile of the patient who is more likely to develop these complaints. The profile includes any patient who has a chronic pain disorder of any type, but especially chronic pelvic pain disorders such as endometriosis and vulvodynia. Other risk factors appear to be a history of pelvic surgery involving any permanent material, suture or mesh, and young age.
So if we have a patient in her late 30s who has undergone reconstructive surgery using permanent sutures and who has an element of chronic pelvic pain, we would counsel her strongly to consider surgical options other than the use of synthetic mesh.
DR. WALTERS: The main indications for mesh-augmented prolapse repair are recurrent posthysterectomy vaginal vault prolapse, for which I usually perform ASC, and recurrent cystocele or anteriorapical prolapse, for which I use one of the anterior mesh kits.
I still think sutured repairs—by that, I mean uterosacral ligament or sacrospinous colpopexy with sutured rectocele repair—work best for recurrent posterior wall and posteriorapical prolapse. I don’t use mesh augmentation for rectocele.
The main contraindication to mesh augmentation, as I see it, is a history of mesh complications. If I am repairing a mesh complication such as erosion or pain, I do not place another mesh.
Medical issues that might increase mesh complications, such as diabetes, steroid use, or severe vaginal atrophy, would, at the very least, make me consider carefully whether mesh augmentation is appropriate. The literature is not clear on this, so mesh could still be used if the surgeon thinks it is necessary.
DR. KARRAM: I haven’t found a definitive indication for mesh augmentation. We have used biologic meshes empirically, but I am not convinced that they really add long-term durability, regardless of whether they are used in the anterior or posterior vaginal segment.
Our published durability rate for traditional suture-type repairs is in the range of 85% at 5 years out.6 Even if I assumed that mesh would give me 100% 5-year durability, this rate would have to be at the expense of some erosion, pain, and other complications unique to mesh. I do not think that the potential improvement in durability is worth these potential complications.

FIGURE 1 When the pelvic support system is intact, prolapse is rare
In the normal pelvis, organs are supported by a complex web of muscles, fascia, and connective tissue. 
FIGURE 2 Mesh augmentation seeks to enhance the durability of repair
One type of mesh in final position. Mesh-augmented repair restores the vaginal apex and lends support to the walls of the vagina.
DR. KARRAM: If you are doing a lot of mesh repairs, you are obviously content with the results and feel that the few complications you are seeing are outweighed by the advantages mesh confers. How do you avoid extrusion and avert creation of a painful vagina?
DR. RAZ: Most of our cases are recurrent prolapse after failed vaginal or abdominal repair. I am indeed using a significant amount of soft polypropylene mesh for reconstructive procedures. As with the use of any other synthetic material, low-grade infection can develop after a few weeks or months. I use copious irrigation with antibiotic solution during reconstruction.
To avoid extrusion, I perform deep, rather than superficial, dissection of the vaginal wall to allow for better coverage of the mesh. For posterior mesh reconstruction, I cover the mesh with pararectal fascia to prevent erosion.
For mesh-augmented procedures, I cut the mesh myself in the operating room ( FIGURE 3 ). For a sling, I use a 10 cm × 1 cm soft polypropylene mesh. For a grade 3 or 4 cystocele, I use a trapezoid of soft polypropylene mesh with several points of fixation:
- at the sacrouterine ligament
- lateral to the obturator fascia
- distal to the bladder neck.
I always repair the vault at the same time.
For vault prolapse, I use a segment of soft polypropylene mesh in the shape of an apron with two arms (1 cm × 4 cm) and a central segment (4 cm × 7 cm). I support the vault using number 1-0 delayed absorbable suture and mesh. From outside the vaginal wall, in the posterolateral deep vaginal wall (inside the peritoneum), I incorporate the origin of the sacrouterine ligament and one arm of the mesh in the groove between the colon and levator ani, 15 cm from the introitus. I bring the suture 1 cm from the original entrance. A separate set of sutures brings the perirectal fascia together with the sacrouterine ligaments and perivesical fascia to close the peritoneal cavity. I tie the vault-suspension sutures, providing support to the cuff in a high posterior position (12 to 15 cm from the introitus).
In selected cases of significant recurrent rectocele, I use a rectangle of soft polypropylene mesh anchored to the origin of the sacrouterine ligament and distal to the perineal membrane. The mesh is covered by the pararectal fascia.
We have not seen vaginal, urethral, or bladder erosion in 1,800 cases of our distal urethral Prolene sling procedure using 10 cm × 1 cm soft mesh. In patients who have significant cystocele, vault prolapse, and recurrent rectocele, our vaginal erosion rate is 3%. We have never encountered rectal, bladder, or bowel perforation using our technique.
DR. LUCENTE: We often use mesh and are more than simply content with our results—we are extremely pleased, and so are our patients. Having said that, our techniques have definitely evolved over the past few years, as we’ve focused on how to decrease exposure and, more recently, optimize sexual function and vaginal comfort.
First, to avoid exposure, the most critical step is precise hydrodissection and distention of the true vesicovaginal space. This step can only be achieved through careful tactile guidance of the needle tip into the space, where it should remain while hydrodissection is performed. Always remember, sharp dissection “follows” hydrodissection. If you place the needle bevel within the vaginal wall, you will “split” the vaginal wall—as during standard colporrhaphy—which will lead to a high exposure rate.
Second, to avoid dyspareunia, it’s essential to pay close attention to POP-Q measurements, especially vaginal length, to ensure that the reconstruction restores the same length without foreshortening. This approach entails leaving the cervix in most patients who have a shorter vagina, and making sure that the mesh is secured above the ischial spine in younger, sexually active patients who have demonstrated a higher risk of postoperative deep, penetrating dyspareunia, compared with older, less sexually active patients.
Also paramount is to ensure that you have manually displaced the vagina inwardly as much as possible before deploying or setting the mesh. If you simply try to suture secure the mesh with the vagina incised open, without the ability to deploy the mesh with a closed, displaced vagina (to mimic deep penetration), it is difficult, if not impossible, to properly set the mesh for optimal comfort.
In the early days of midurethral pubovaginal slings using polypropylene, the adage was “looser is better than tighter.” This is even truer for transvaginal mesh.
DR. KARRAM: Dr. Walters, please describe your current surgical procedure of choice without mesh and explain why you haven’t adopted mesh for routine repairs.
DR. WALTERS: About 20% of my prolapse surgeries—usually for posthysterectomy or recurrent vaginal vault prolapse—involve ASC with placement of polypropylene mesh. I perform most of these cases through a Pfannenstiel incision, but I’ve also done them laparoscopically. Several of my partners perform ASC laparoscopically and robotically.
For the other 80% of my patients who have prolapse, I perform repairs transvaginally, usually using high bilateral uterosacralligament vaginal-vault suspension. We have learned to suture higher and slightly more medial on the uterosacral ligaments to attain greater vaginal depth and minimize ureteral obstruction. We use two or three sutures on each uterosacral ligament, usually a combination of permanent and delayed absorbable sutures.
I am also performing more sacrospinous ligament suspensions because this operation is being studied by the Pelvic Floor Disorders Network. Properly performed, it is an excellent surgery for apical prolapse. But, as with most of our surgeries for prolapse, recurrent anterior wall prolapse remains a problem.
Like you, Dr. Karram, we’ve studied our group’s anatomic and functional outcomes very carefully for more than 10 years and are mostly satisfied with our cure and complication rates. Although our anatomic outcomes with these surgeries are not always perfect, our reoperation rate for prolapse is only about 5%, with a high level of satisfaction in 88% to 92% of patients.
DR. RAZ: Unaugmented reconstruction fails in more than 30% of cases. Some patients who have significant prolapse and attenuated tissue think that this tissue will become healthier or stronger after reconstructive surgery, but that isn’t the case. In these situations, excision and plication make no clinical sense.
The problem is that we have yet to identify the ideal surrogate for poor-quality tissue. Most of us use polypropylene mesh in different variations. We need a better material that will be nonimmunogenic, well tolerated, and easily incorporated without erosion. Xenograft-like derivatives of dermis, or allografts such as cadaveric fascia, have failed over the long term because the body reabsorbs the graft without forming any new connective tissue.

FIGURE 3 Mesh can be cut in the OR to custom-fit a patient
Hand-cut mesh and points of placement.
PHOTO: SHLOMO RAZ, MD
Is a kit a valuable aid?
DR. KARRAM: If a surgeon wants to augment a repair, what are the advantages of a packaged mesh kit, compared with simply cutting the mesh and performing surgery without a kit?
DR. WALTERS: The advantages of a packaged mesh kit are the convenience involved and the ability to consistently perform the same operation with the same product. That facilitates learning, teaching, and research. It also helps us understand the published literature a little better because “custom” prolapse repairs are operator-dependent and difficult to apply generally to a population of surgeons.
These advantages are most clearly apparent with midurethral sling mesh kits, which have almost revolutionized surgery for stress incontinence. I don’t believe mesh kits for prolapse are there yet, but they certainly have potential.
DR. RAZ: I’m opposed to the use of kits. They are industry-driven. One company has made $1 billion selling them. Imagine a patient who undergoes placement of a sling kit ($1,000), cystocele kit ($1,500), and posterior mesh kit ($1,500). How can our healthcare system sustain this burden, especially when there is no real evidence that a kit improves the operation, and given the incredible complication rate that we see?
Moreover, the kits contain a single-use needle and passer and a precut segment of polypropylene mesh. But every patient is different and requires a unique size or shape of mesh. I don’t believe that a surgeon who knows pelvic anatomy needs a kit to perform mesh-augmented reconstruction. We can buy the same segment of mesh for $200 to $400, cut it as needed, and perform the same operation advertised by industry.
For surgeons who prefer a kit, the tools that are included should be made reusable.
DR. LUCENTE: In my opinion, the primary advantage of a commercially available transvaginal mesh delivery system—notice, I avoided the word “kit,” because I think there are plenty of negative connotations associated with it—is the ability to deliver the mesh in a “tension”-free manner.
One alternative that many people pursue is cutting the mesh to size and using sutures to hold it in place while tissue ingrowth occurs. However, the hernia literature suggests that suturing mesh in place increases the risk of postoperative discomfort at the site of implantation. The true cause of the discomfort remains unclear, but it is thought to arise from nerve tethering or traction at the pre-committed points of attachment before the host tissue and mesh interface have adjusted or settled with tissue ingrowth.
All neuropathic complications of mesh implantation have been shown in the current hernia literature to be increased with the use of sutures.7 Also, as previously mentioned, it is extremely difficult to set or adjust the mesh with the vaginal incision remaining “open,” which is a downside to suture techniques.
What training is necessary to use a kit?
DR. KARRAM: Mesh kits are aggressively promoted by industry, with close to half a dozen different kits to be available soon. What is the minimum amount of training one should have before utilizing these kits?
DR. WALTERS: The surgeon should at least know how to perform traditional sutured prolapse repairs and SUI surgery and be able to perform cystoscopy. Ideally, the surgeon should undergo training on a cadaver with a skilled and experienced user of the mesh kit. The surgeon also should carefully review the risks and benefits of mesh kits with the patient and inform the patient that he or she is in the early learning curve of a particular surgery. The informed patient should have a right to refuse mesh-augmented prolapse surgery after the consent process.
DR. LUCENTE: I’m glad you asked this question. I strongly believe that surgical expertise and proficiency within gynecology need to be more effectively addressed by us all. We have a situation in our field in which techniques and technology are widening the gap between what is possible and what the surgeon is comfortable doing safely.
It’s incumbent on all of us, especially those who are in a leadership position as a chairperson or chief of a division, to work with our physician staff and faculty to optimize surgical skill and patient outcomes, including safety, with new technologies.
As for the minimal amount of training needed, that’s extremely variable. It depends on the current skill set of the physician and his or her ability to pick up the mechanics of the surgery as it is taught through a cadaver lab or preceptorship. It’s regrettable that some physicians lack the objectivity and insight to judge their own skill set. This, again, is the time for a chairperson or chief of a division to step up to the plate and ensure proper credentialing and demonstration of proficiency.
It is unrealistic to expect industry to decide who should or should not utilize this truly breakthrough technology. That is our responsibility as physicians.
DR. KARRAM: At a minimum, I think any surgeon utilizing a kit should have a firm understanding of pelvic floor anatomy and experience performing traditional repairs:
- intraperitoneal procedures such as Mc-Call culdoplasty and uterosacral suspension
- sacrospinous suspension
- retropubic procedures and anti-incontinence operations such as pubovaginal slings.
This three-dimensional understanding of the pelvic floor is mandatory if one is to assume that blind passage of trocars through potentially dangerous spaces is the wave of the future.
DR. RAZ: You need to be a pelvic surgeon, know your anatomy, and know how to manage complications if you are going to use one of these kits. You should stick to the surgery that works best in your hands. Industry cannot teach you to be a good pelvic surgeon; it takes lifelong experience.
DR. KARRAM: If you have a patient who is sexually inactive with pelvic organ prolapse, would you prefer a mesh repair or an obliterative procedure? And why?
DR. WALTERS: If the patient is sexually inactive—especially if she is older and definitely will not be in the future—it makes absolutely no sense to perform a mesh-augmented repair. A traditional, somewhat tight, sutured repair works fine in this setting and carries very low risk.
In fact, our group and others have found that, in carefully selected patients, partial colpectomy and colpocleisis procedures (without grafts) have among the highest cure and satisfaction rates of all surgeries we perform for prolapse; they also have relatively low risk.8 Recurrent prolapse after an obliterative procedure is rare; most of the dissatisfaction relates to postoperative voiding difficulties or persistent or de novo urinary incontinence.
DR. KARRAM: I also prefer an obliterative procedure. I see no reason to bring in the cost and potential for complications that mesh repair entails. An obliterative procedure should produce an anatomic success rate close to 100%, with minimal complications. It also can be performed quickly with minimal anesthesia and convalescence.
DR. LUCENTE: My response is based on a clinical study that my associate, Dr. Miles Murphy, has performed, comparing a transvaginal mesh procedure with a LaForte operation for severe pelvic organ prolapse.9 Both patient groups were well satisfied with the result, and success rates were comparable. However, the group that underwent the transvaginal mesh procedure had a shorter operative time.
As a result of these studies, we tend to prefer transvaginal mesh repair. Even though the woman may be sexually inactive, the procedure preserves vaginal function, and we all know that life has a way of being unpredictable. Her situation may change so that she once again desires sexual function.
However, for a very elderly woman—one in her late 80s or 90s—who has severe or extreme prolapse with a very large procidentia and vaginal length measuring, say, 13 cm beyond the introitus, I do prefer an obliterative procedure.
DR. RAZ: I agree. I would not offer a sexually inactive patient an obliterative procedure. You never know what the future will hold.
Mesh repair can be performed safely, provided the surgeon has good knowledge of anatomic landmarks and knows how to manage any potential complications that may arise.
MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.
SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.
VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.
MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
Mesh kits for repairing prolapse are proliferating like crazy, just as they did for midurethral sling procedures. But mesh augmentation of prolapse surgeries requires more than a prepackaged assortment of tools and materials. In this article, moderator Mickey M. Karram, MD, and a panel of nationally recognized urogynecologists and urologists describe the literature on mesh augmentation and discuss indications, contraindications, techniques, applicable cases, and the considerable training required.
In Part 2, which will appear in the February issue of OBG Management, the panel tackles the thorny topic of complications, including erosion, extrusion, foreshortening of the vagina, dyspareunia, and pain. Their discussion focuses on ways to avoid these problems, and methods for correcting them.
Do we have enough data?
DR. KARRAM: To start, let’s quickly review the peer-reviewed literature on the use of mesh augmentation during surgery for pelvic organ prolapse.
DR. WALTERS: Until recently, most data concerned open abdominal sacrocolpopexy (ASC) using polypropylene or Merseline mesh. There is significant clinical experience with this operation, and multiple cohort studies show long-term cure rates of 78% to 100% for apical prolapse.1
At least two randomized controlled trials have compared open ASC with sutured vaginal colpopexy procedures, and ASC is certainly equal to—perhaps better than—all transvaginal sutured repairs.2,3
With ASC, most recurrences affect the distal half of the vagina and involve one or more of the following:
- anterior or posterior vaginal wall prolapse (or both)
- stress urinary incontinence (SUI)
- distal rectocele.1,3
Mesh erosion occurs in 3.4% of cases and is usually easily managed.1 Other complications, including bowel injury, tend to be related to access, regardless of whether the operation is performed via laparotomy or laparoscopy.
Robotic sacrocolpopexy has become popular in recent years, and we will probably see data on this approach as we gain experience.
When it comes to vaginal mesh kits, the peer-reviewed literature is just beginning to expand, with many studies being presented at international meetings. For anterior and, possibly, apical vaginal prolapse, the cure rate after use of a mesh kit appears to be as high as, or higher than, the rate for sutured repairs.4 This high rate of anatomic cure is balanced somewhat by additional cost and complications involving mesh and the kits.
For posterior vaginal wall prolapse and rectocele, I firmly believe, based on our research and that of others, that sutured repairs are superior to graft-augmented surgery.5
DR. KARRAM: What are the indications and contraindications for mesh augmentation of prolapse repair ( FIGURES 1 and 2 )?
DR. LUCENTE: I believe mesh is indicated in any patient in need of surgical repair of pelvic organ prolapse who is seeking optimal durability and is willing to accept the known risks of the surgery.
The issue becomes more complex when it comes to contraindications. Absolute contraindications are fairly obvious; they include medically unstable patients and those who may have an inactive infectious process within the pelvis or even undiagnosed abnormal uterine bleeding.
At our center, because the potential for dyspareunia and pelvic discomfort is our biggest concern, we have developed a profile of the patient who is more likely to develop these complaints. The profile includes any patient who has a chronic pain disorder of any type, but especially chronic pelvic pain disorders such as endometriosis and vulvodynia. Other risk factors appear to be a history of pelvic surgery involving any permanent material, suture or mesh, and young age.
So if we have a patient in her late 30s who has undergone reconstructive surgery using permanent sutures and who has an element of chronic pelvic pain, we would counsel her strongly to consider surgical options other than the use of synthetic mesh.
DR. WALTERS: The main indications for mesh-augmented prolapse repair are recurrent posthysterectomy vaginal vault prolapse, for which I usually perform ASC, and recurrent cystocele or anteriorapical prolapse, for which I use one of the anterior mesh kits.
I still think sutured repairs—by that, I mean uterosacral ligament or sacrospinous colpopexy with sutured rectocele repair—work best for recurrent posterior wall and posteriorapical prolapse. I don’t use mesh augmentation for rectocele.
The main contraindication to mesh augmentation, as I see it, is a history of mesh complications. If I am repairing a mesh complication such as erosion or pain, I do not place another mesh.
Medical issues that might increase mesh complications, such as diabetes, steroid use, or severe vaginal atrophy, would, at the very least, make me consider carefully whether mesh augmentation is appropriate. The literature is not clear on this, so mesh could still be used if the surgeon thinks it is necessary.
DR. KARRAM: I haven’t found a definitive indication for mesh augmentation. We have used biologic meshes empirically, but I am not convinced that they really add long-term durability, regardless of whether they are used in the anterior or posterior vaginal segment.
Our published durability rate for traditional suture-type repairs is in the range of 85% at 5 years out.6 Even if I assumed that mesh would give me 100% 5-year durability, this rate would have to be at the expense of some erosion, pain, and other complications unique to mesh. I do not think that the potential improvement in durability is worth these potential complications.

FIGURE 1 When the pelvic support system is intact, prolapse is rare
In the normal pelvis, organs are supported by a complex web of muscles, fascia, and connective tissue. 
FIGURE 2 Mesh augmentation seeks to enhance the durability of repair
One type of mesh in final position. Mesh-augmented repair restores the vaginal apex and lends support to the walls of the vagina.
DR. KARRAM: If you are doing a lot of mesh repairs, you are obviously content with the results and feel that the few complications you are seeing are outweighed by the advantages mesh confers. How do you avoid extrusion and avert creation of a painful vagina?
DR. RAZ: Most of our cases are recurrent prolapse after failed vaginal or abdominal repair. I am indeed using a significant amount of soft polypropylene mesh for reconstructive procedures. As with the use of any other synthetic material, low-grade infection can develop after a few weeks or months. I use copious irrigation with antibiotic solution during reconstruction.
To avoid extrusion, I perform deep, rather than superficial, dissection of the vaginal wall to allow for better coverage of the mesh. For posterior mesh reconstruction, I cover the mesh with pararectal fascia to prevent erosion.
For mesh-augmented procedures, I cut the mesh myself in the operating room ( FIGURE 3 ). For a sling, I use a 10 cm × 1 cm soft polypropylene mesh. For a grade 3 or 4 cystocele, I use a trapezoid of soft polypropylene mesh with several points of fixation:
- at the sacrouterine ligament
- lateral to the obturator fascia
- distal to the bladder neck.
I always repair the vault at the same time.
For vault prolapse, I use a segment of soft polypropylene mesh in the shape of an apron with two arms (1 cm × 4 cm) and a central segment (4 cm × 7 cm). I support the vault using number 1-0 delayed absorbable suture and mesh. From outside the vaginal wall, in the posterolateral deep vaginal wall (inside the peritoneum), I incorporate the origin of the sacrouterine ligament and one arm of the mesh in the groove between the colon and levator ani, 15 cm from the introitus. I bring the suture 1 cm from the original entrance. A separate set of sutures brings the perirectal fascia together with the sacrouterine ligaments and perivesical fascia to close the peritoneal cavity. I tie the vault-suspension sutures, providing support to the cuff in a high posterior position (12 to 15 cm from the introitus).
In selected cases of significant recurrent rectocele, I use a rectangle of soft polypropylene mesh anchored to the origin of the sacrouterine ligament and distal to the perineal membrane. The mesh is covered by the pararectal fascia.
We have not seen vaginal, urethral, or bladder erosion in 1,800 cases of our distal urethral Prolene sling procedure using 10 cm × 1 cm soft mesh. In patients who have significant cystocele, vault prolapse, and recurrent rectocele, our vaginal erosion rate is 3%. We have never encountered rectal, bladder, or bowel perforation using our technique.
DR. LUCENTE: We often use mesh and are more than simply content with our results—we are extremely pleased, and so are our patients. Having said that, our techniques have definitely evolved over the past few years, as we’ve focused on how to decrease exposure and, more recently, optimize sexual function and vaginal comfort.
First, to avoid exposure, the most critical step is precise hydrodissection and distention of the true vesicovaginal space. This step can only be achieved through careful tactile guidance of the needle tip into the space, where it should remain while hydrodissection is performed. Always remember, sharp dissection “follows” hydrodissection. If you place the needle bevel within the vaginal wall, you will “split” the vaginal wall—as during standard colporrhaphy—which will lead to a high exposure rate.
Second, to avoid dyspareunia, it’s essential to pay close attention to POP-Q measurements, especially vaginal length, to ensure that the reconstruction restores the same length without foreshortening. This approach entails leaving the cervix in most patients who have a shorter vagina, and making sure that the mesh is secured above the ischial spine in younger, sexually active patients who have demonstrated a higher risk of postoperative deep, penetrating dyspareunia, compared with older, less sexually active patients.
Also paramount is to ensure that you have manually displaced the vagina inwardly as much as possible before deploying or setting the mesh. If you simply try to suture secure the mesh with the vagina incised open, without the ability to deploy the mesh with a closed, displaced vagina (to mimic deep penetration), it is difficult, if not impossible, to properly set the mesh for optimal comfort.
In the early days of midurethral pubovaginal slings using polypropylene, the adage was “looser is better than tighter.” This is even truer for transvaginal mesh.
DR. KARRAM: Dr. Walters, please describe your current surgical procedure of choice without mesh and explain why you haven’t adopted mesh for routine repairs.
DR. WALTERS: About 20% of my prolapse surgeries—usually for posthysterectomy or recurrent vaginal vault prolapse—involve ASC with placement of polypropylene mesh. I perform most of these cases through a Pfannenstiel incision, but I’ve also done them laparoscopically. Several of my partners perform ASC laparoscopically and robotically.
For the other 80% of my patients who have prolapse, I perform repairs transvaginally, usually using high bilateral uterosacralligament vaginal-vault suspension. We have learned to suture higher and slightly more medial on the uterosacral ligaments to attain greater vaginal depth and minimize ureteral obstruction. We use two or three sutures on each uterosacral ligament, usually a combination of permanent and delayed absorbable sutures.
I am also performing more sacrospinous ligament suspensions because this operation is being studied by the Pelvic Floor Disorders Network. Properly performed, it is an excellent surgery for apical prolapse. But, as with most of our surgeries for prolapse, recurrent anterior wall prolapse remains a problem.
Like you, Dr. Karram, we’ve studied our group’s anatomic and functional outcomes very carefully for more than 10 years and are mostly satisfied with our cure and complication rates. Although our anatomic outcomes with these surgeries are not always perfect, our reoperation rate for prolapse is only about 5%, with a high level of satisfaction in 88% to 92% of patients.
DR. RAZ: Unaugmented reconstruction fails in more than 30% of cases. Some patients who have significant prolapse and attenuated tissue think that this tissue will become healthier or stronger after reconstructive surgery, but that isn’t the case. In these situations, excision and plication make no clinical sense.
The problem is that we have yet to identify the ideal surrogate for poor-quality tissue. Most of us use polypropylene mesh in different variations. We need a better material that will be nonimmunogenic, well tolerated, and easily incorporated without erosion. Xenograft-like derivatives of dermis, or allografts such as cadaveric fascia, have failed over the long term because the body reabsorbs the graft without forming any new connective tissue.

FIGURE 3 Mesh can be cut in the OR to custom-fit a patient
Hand-cut mesh and points of placement.
PHOTO: SHLOMO RAZ, MD
Is a kit a valuable aid?
DR. KARRAM: If a surgeon wants to augment a repair, what are the advantages of a packaged mesh kit, compared with simply cutting the mesh and performing surgery without a kit?
DR. WALTERS: The advantages of a packaged mesh kit are the convenience involved and the ability to consistently perform the same operation with the same product. That facilitates learning, teaching, and research. It also helps us understand the published literature a little better because “custom” prolapse repairs are operator-dependent and difficult to apply generally to a population of surgeons.
These advantages are most clearly apparent with midurethral sling mesh kits, which have almost revolutionized surgery for stress incontinence. I don’t believe mesh kits for prolapse are there yet, but they certainly have potential.
DR. RAZ: I’m opposed to the use of kits. They are industry-driven. One company has made $1 billion selling them. Imagine a patient who undergoes placement of a sling kit ($1,000), cystocele kit ($1,500), and posterior mesh kit ($1,500). How can our healthcare system sustain this burden, especially when there is no real evidence that a kit improves the operation, and given the incredible complication rate that we see?
Moreover, the kits contain a single-use needle and passer and a precut segment of polypropylene mesh. But every patient is different and requires a unique size or shape of mesh. I don’t believe that a surgeon who knows pelvic anatomy needs a kit to perform mesh-augmented reconstruction. We can buy the same segment of mesh for $200 to $400, cut it as needed, and perform the same operation advertised by industry.
For surgeons who prefer a kit, the tools that are included should be made reusable.
DR. LUCENTE: In my opinion, the primary advantage of a commercially available transvaginal mesh delivery system—notice, I avoided the word “kit,” because I think there are plenty of negative connotations associated with it—is the ability to deliver the mesh in a “tension”-free manner.
One alternative that many people pursue is cutting the mesh to size and using sutures to hold it in place while tissue ingrowth occurs. However, the hernia literature suggests that suturing mesh in place increases the risk of postoperative discomfort at the site of implantation. The true cause of the discomfort remains unclear, but it is thought to arise from nerve tethering or traction at the pre-committed points of attachment before the host tissue and mesh interface have adjusted or settled with tissue ingrowth.
All neuropathic complications of mesh implantation have been shown in the current hernia literature to be increased with the use of sutures.7 Also, as previously mentioned, it is extremely difficult to set or adjust the mesh with the vaginal incision remaining “open,” which is a downside to suture techniques.
What training is necessary to use a kit?
DR. KARRAM: Mesh kits are aggressively promoted by industry, with close to half a dozen different kits to be available soon. What is the minimum amount of training one should have before utilizing these kits?
DR. WALTERS: The surgeon should at least know how to perform traditional sutured prolapse repairs and SUI surgery and be able to perform cystoscopy. Ideally, the surgeon should undergo training on a cadaver with a skilled and experienced user of the mesh kit. The surgeon also should carefully review the risks and benefits of mesh kits with the patient and inform the patient that he or she is in the early learning curve of a particular surgery. The informed patient should have a right to refuse mesh-augmented prolapse surgery after the consent process.
DR. LUCENTE: I’m glad you asked this question. I strongly believe that surgical expertise and proficiency within gynecology need to be more effectively addressed by us all. We have a situation in our field in which techniques and technology are widening the gap between what is possible and what the surgeon is comfortable doing safely.
It’s incumbent on all of us, especially those who are in a leadership position as a chairperson or chief of a division, to work with our physician staff and faculty to optimize surgical skill and patient outcomes, including safety, with new technologies.
As for the minimal amount of training needed, that’s extremely variable. It depends on the current skill set of the physician and his or her ability to pick up the mechanics of the surgery as it is taught through a cadaver lab or preceptorship. It’s regrettable that some physicians lack the objectivity and insight to judge their own skill set. This, again, is the time for a chairperson or chief of a division to step up to the plate and ensure proper credentialing and demonstration of proficiency.
It is unrealistic to expect industry to decide who should or should not utilize this truly breakthrough technology. That is our responsibility as physicians.
DR. KARRAM: At a minimum, I think any surgeon utilizing a kit should have a firm understanding of pelvic floor anatomy and experience performing traditional repairs:
- intraperitoneal procedures such as Mc-Call culdoplasty and uterosacral suspension
- sacrospinous suspension
- retropubic procedures and anti-incontinence operations such as pubovaginal slings.
This three-dimensional understanding of the pelvic floor is mandatory if one is to assume that blind passage of trocars through potentially dangerous spaces is the wave of the future.
DR. RAZ: You need to be a pelvic surgeon, know your anatomy, and know how to manage complications if you are going to use one of these kits. You should stick to the surgery that works best in your hands. Industry cannot teach you to be a good pelvic surgeon; it takes lifelong experience.
DR. KARRAM: If you have a patient who is sexually inactive with pelvic organ prolapse, would you prefer a mesh repair or an obliterative procedure? And why?
DR. WALTERS: If the patient is sexually inactive—especially if she is older and definitely will not be in the future—it makes absolutely no sense to perform a mesh-augmented repair. A traditional, somewhat tight, sutured repair works fine in this setting and carries very low risk.
In fact, our group and others have found that, in carefully selected patients, partial colpectomy and colpocleisis procedures (without grafts) have among the highest cure and satisfaction rates of all surgeries we perform for prolapse; they also have relatively low risk.8 Recurrent prolapse after an obliterative procedure is rare; most of the dissatisfaction relates to postoperative voiding difficulties or persistent or de novo urinary incontinence.
DR. KARRAM: I also prefer an obliterative procedure. I see no reason to bring in the cost and potential for complications that mesh repair entails. An obliterative procedure should produce an anatomic success rate close to 100%, with minimal complications. It also can be performed quickly with minimal anesthesia and convalescence.
DR. LUCENTE: My response is based on a clinical study that my associate, Dr. Miles Murphy, has performed, comparing a transvaginal mesh procedure with a LaForte operation for severe pelvic organ prolapse.9 Both patient groups were well satisfied with the result, and success rates were comparable. However, the group that underwent the transvaginal mesh procedure had a shorter operative time.
As a result of these studies, we tend to prefer transvaginal mesh repair. Even though the woman may be sexually inactive, the procedure preserves vaginal function, and we all know that life has a way of being unpredictable. Her situation may change so that she once again desires sexual function.
However, for a very elderly woman—one in her late 80s or 90s—who has severe or extreme prolapse with a very large procidentia and vaginal length measuring, say, 13 cm beyond the introitus, I do prefer an obliterative procedure.
DR. RAZ: I agree. I would not offer a sexually inactive patient an obliterative procedure. You never know what the future will hold.
Mesh repair can be performed safely, provided the surgeon has good knowledge of anatomic landmarks and knows how to manage any potential complications that may arise.
1. Nygaard IE, McCreery R, Brubaker L, et al. Pelvic Floor Disorders Network. Abdominal sacrocolpopexy: a comprehensive review. Obstet Gynecol. 2004;104:805-823.
2. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421;discussion 1421-1422.
3. Maher CF, Qatawneh AM, Dwyer PL, Carey MP, Cornish A, Schluter PJ. Abdominal sacral colpopexy or vaginal sacrospinous colpopexy for vaginal vault prolapse: a prospective randomized study. Am J Obstet Gynecol. 2004;190:20-26.
4. Murphy M. Society of Gynecologic Surgeons Systematic Review Group. Clinical practice guidelines on vaginal graft use from the Society of Gynecologic Surgeons. Obstet Gynecol. 2008;112:1123-1130.
5. Paraiso MF, Barber MD, Muir TW, Walters MD. Rectocele repair: a randomized trial of three surgical procedures including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
6. Silva WA, Pauls RN, Segal JL, Rooney CM, Kleeman SD, Karram MM. Uterosacral ligament vault suspension: five-year outcomes. Obstet Gynecol 2006;108:255-263.
7. EU Hernia Trialists Collaboration. Repair of groin hernia with synthetic mesh: meta-analysis of randomized controlled trials. Ann Surg. 2002;235:322-332.
8. Barber MD, Amundsen C, Paraiso MFR, Weidner A, Romero A, Walters MD. Quality of life after surgery for genital prolapse in elderly women: obliterative and reconstructive surgery. Int Urogynecol J. 2007;18:799-806.
9. Murphy M, van Raalte H, Mercurio E, Haff R, Wiseman B, Lucente VR. Incontinence-related quality of life and sexual function following the tension-free vaginal tape vs the “inside-out” tension-free vaginal tape obturator. Int Urogynecol J. 2008;19:481-487.
1. Nygaard IE, McCreery R, Brubaker L, et al. Pelvic Floor Disorders Network. Abdominal sacrocolpopexy: a comprehensive review. Obstet Gynecol. 2004;104:805-823.
2. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421;discussion 1421-1422.
3. Maher CF, Qatawneh AM, Dwyer PL, Carey MP, Cornish A, Schluter PJ. Abdominal sacral colpopexy or vaginal sacrospinous colpopexy for vaginal vault prolapse: a prospective randomized study. Am J Obstet Gynecol. 2004;190:20-26.
4. Murphy M. Society of Gynecologic Surgeons Systematic Review Group. Clinical practice guidelines on vaginal graft use from the Society of Gynecologic Surgeons. Obstet Gynecol. 2008;112:1123-1130.
5. Paraiso MF, Barber MD, Muir TW, Walters MD. Rectocele repair: a randomized trial of three surgical procedures including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
6. Silva WA, Pauls RN, Segal JL, Rooney CM, Kleeman SD, Karram MM. Uterosacral ligament vault suspension: five-year outcomes. Obstet Gynecol 2006;108:255-263.
7. EU Hernia Trialists Collaboration. Repair of groin hernia with synthetic mesh: meta-analysis of randomized controlled trials. Ann Surg. 2002;235:322-332.
8. Barber MD, Amundsen C, Paraiso MFR, Weidner A, Romero A, Walters MD. Quality of life after surgery for genital prolapse in elderly women: obliterative and reconstructive surgery. Int Urogynecol J. 2007;18:799-806.
9. Murphy M, van Raalte H, Mercurio E, Haff R, Wiseman B, Lucente VR. Incontinence-related quality of life and sexual function following the tension-free vaginal tape vs the “inside-out” tension-free vaginal tape obturator. Int Urogynecol J. 2008;19:481-487.
Is long-term outcome with TVT comparable to that of laparoscopic Burch colposuspension?
Just 2 years ago, when Brubaker and colleagues published initial findings from the colpopexy and urinary reduction efforts (CARE) trial in the New England Journal of Medicine,1 Burch colposuspension was a well-established anti-incontinence procedure utilized by many urogynecologists. The procedure remains a reliable intervention, although midurethral sling procedures have surpassed it in popularity and (some would say) efficacy. This issue’s installment of Examining the Evidence highlights two recent investigations of the antiincontinence procedure:
- 2-year follow-up from the CARE trial, which compared sacrocolpopexy, with and without a concomitant Burch procedure, in women who did not have symptoms of stress urinary incontinence (SUI) at the time of surgery
- a comparison of laparoscopic Burch colposuspension and the tension-free vaginal tape (TVT) technique.
Details of the study
Seventy-two women were originally enrolled from 1999 to 2002; 74% of them (25 in the TVT group and 28 in the laparoscopic Burch group) were available for long-term followup 4 to 8 years after surgery. Fifty-seven percent (16/28) of women had subjective urinary incontinence after laparoscopic Burch colposuspension versus 48% (12/25) after TVT. There were no differences between the groups in subjective or objective findings or urinary incontinence. However, the study was severely underpowered to be able to show any difference between the groups.
These cure rates are low, but the authors note that only 11% of the laparoscopic Burch group and 8% of the TVT group had bothersome SUI. Quality of life on the urogenital distress inventory and incontinence impact questionnaire short forms was improved in both groups equally by 2 years and maintained throughout the rest of the trial.
These poor objective results are similar to those found in a 5-year follow-up by Ward and Hilton of their prospective, randomized, controlled trial of Burch versus TVT procedures. There, only 39% of the TVT group and 46% of the Burch group reported no incontinence.
The original trial by Paraiso and colleagues3 showed better outcomes in the TVT group 1 year after surgery, but that difference did not remain 4 to 8 years later. One explanation for that observation may be type-II error resulting from the small number of subjects in the trial.
Laparoscopic Burch colposuspension and the TVT procedure appear to have equal long-term outcomes. Because placing TVT is less invasive, however, it may be the preferable procedure until larger trials or meta-analyses conclusively determine which operation is superior.—PETER K. SAND, MD
1. Ulmsten U, Henriksson L, Johnson P, Varhos G. An ambulatory surgical procedure under local anesthesia for treatment of female urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 1996;7:81-86.
2. Ward KL, Hilton P. On behalf of the UK and Ireland TVT Trial Group. A prospective multicenter randomized trial of tension-free vaginal tape and colposuspension for primary urodynamic stress incontinence: two-year follow-up. Am J Obstet Gynecol. 2004;190:324-331.
3. Paraiso MF, Walters MD, Karram MM, Barber MD. Laparoscopic Burch colposuspension versus tension-free vaginal tape: a randomized trial. Obstet Gynecol. 2004;104:1249-1258.
Just 2 years ago, when Brubaker and colleagues published initial findings from the colpopexy and urinary reduction efforts (CARE) trial in the New England Journal of Medicine,1 Burch colposuspension was a well-established anti-incontinence procedure utilized by many urogynecologists. The procedure remains a reliable intervention, although midurethral sling procedures have surpassed it in popularity and (some would say) efficacy. This issue’s installment of Examining the Evidence highlights two recent investigations of the antiincontinence procedure:
- 2-year follow-up from the CARE trial, which compared sacrocolpopexy, with and without a concomitant Burch procedure, in women who did not have symptoms of stress urinary incontinence (SUI) at the time of surgery
- a comparison of laparoscopic Burch colposuspension and the tension-free vaginal tape (TVT) technique.
Details of the study
Seventy-two women were originally enrolled from 1999 to 2002; 74% of them (25 in the TVT group and 28 in the laparoscopic Burch group) were available for long-term followup 4 to 8 years after surgery. Fifty-seven percent (16/28) of women had subjective urinary incontinence after laparoscopic Burch colposuspension versus 48% (12/25) after TVT. There were no differences between the groups in subjective or objective findings or urinary incontinence. However, the study was severely underpowered to be able to show any difference between the groups.
These cure rates are low, but the authors note that only 11% of the laparoscopic Burch group and 8% of the TVT group had bothersome SUI. Quality of life on the urogenital distress inventory and incontinence impact questionnaire short forms was improved in both groups equally by 2 years and maintained throughout the rest of the trial.
These poor objective results are similar to those found in a 5-year follow-up by Ward and Hilton of their prospective, randomized, controlled trial of Burch versus TVT procedures. There, only 39% of the TVT group and 46% of the Burch group reported no incontinence.
The original trial by Paraiso and colleagues3 showed better outcomes in the TVT group 1 year after surgery, but that difference did not remain 4 to 8 years later. One explanation for that observation may be type-II error resulting from the small number of subjects in the trial.
Laparoscopic Burch colposuspension and the TVT procedure appear to have equal long-term outcomes. Because placing TVT is less invasive, however, it may be the preferable procedure until larger trials or meta-analyses conclusively determine which operation is superior.—PETER K. SAND, MD
Just 2 years ago, when Brubaker and colleagues published initial findings from the colpopexy and urinary reduction efforts (CARE) trial in the New England Journal of Medicine,1 Burch colposuspension was a well-established anti-incontinence procedure utilized by many urogynecologists. The procedure remains a reliable intervention, although midurethral sling procedures have surpassed it in popularity and (some would say) efficacy. This issue’s installment of Examining the Evidence highlights two recent investigations of the antiincontinence procedure:
- 2-year follow-up from the CARE trial, which compared sacrocolpopexy, with and without a concomitant Burch procedure, in women who did not have symptoms of stress urinary incontinence (SUI) at the time of surgery
- a comparison of laparoscopic Burch colposuspension and the tension-free vaginal tape (TVT) technique.
Details of the study
Seventy-two women were originally enrolled from 1999 to 2002; 74% of them (25 in the TVT group and 28 in the laparoscopic Burch group) were available for long-term followup 4 to 8 years after surgery. Fifty-seven percent (16/28) of women had subjective urinary incontinence after laparoscopic Burch colposuspension versus 48% (12/25) after TVT. There were no differences between the groups in subjective or objective findings or urinary incontinence. However, the study was severely underpowered to be able to show any difference between the groups.
These cure rates are low, but the authors note that only 11% of the laparoscopic Burch group and 8% of the TVT group had bothersome SUI. Quality of life on the urogenital distress inventory and incontinence impact questionnaire short forms was improved in both groups equally by 2 years and maintained throughout the rest of the trial.
These poor objective results are similar to those found in a 5-year follow-up by Ward and Hilton of their prospective, randomized, controlled trial of Burch versus TVT procedures. There, only 39% of the TVT group and 46% of the Burch group reported no incontinence.
The original trial by Paraiso and colleagues3 showed better outcomes in the TVT group 1 year after surgery, but that difference did not remain 4 to 8 years later. One explanation for that observation may be type-II error resulting from the small number of subjects in the trial.
Laparoscopic Burch colposuspension and the TVT procedure appear to have equal long-term outcomes. Because placing TVT is less invasive, however, it may be the preferable procedure until larger trials or meta-analyses conclusively determine which operation is superior.—PETER K. SAND, MD
1. Ulmsten U, Henriksson L, Johnson P, Varhos G. An ambulatory surgical procedure under local anesthesia for treatment of female urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 1996;7:81-86.
2. Ward KL, Hilton P. On behalf of the UK and Ireland TVT Trial Group. A prospective multicenter randomized trial of tension-free vaginal tape and colposuspension for primary urodynamic stress incontinence: two-year follow-up. Am J Obstet Gynecol. 2004;190:324-331.
3. Paraiso MF, Walters MD, Karram MM, Barber MD. Laparoscopic Burch colposuspension versus tension-free vaginal tape: a randomized trial. Obstet Gynecol. 2004;104:1249-1258.
1. Ulmsten U, Henriksson L, Johnson P, Varhos G. An ambulatory surgical procedure under local anesthesia for treatment of female urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 1996;7:81-86.
2. Ward KL, Hilton P. On behalf of the UK and Ireland TVT Trial Group. A prospective multicenter randomized trial of tension-free vaginal tape and colposuspension for primary urodynamic stress incontinence: two-year follow-up. Am J Obstet Gynecol. 2004;190:324-331.
3. Paraiso MF, Walters MD, Karram MM, Barber MD. Laparoscopic Burch colposuspension versus tension-free vaginal tape: a randomized trial. Obstet Gynecol. 2004;104:1249-1258.
Is Burch colposuspension needed at the time of sacrocolpopexy to prevent SUI?
Just 2 years ago, when Brubaker and colleagues published initial findings from the colpopexy and urinary reduction efforts (CARE) trial in the New England Journal of Medicine,1 Burch colposuspension was a well-established anti-incontinence procedure utilized by many urogynecologists. The procedure remains a reliable intervention, although midurethral sling procedures have surpassed it in popularity and (some would say) efficacy. This issue’s installment of Examining the Evidence highlights two recent investigations of the antiincontinence procedure:
- 2-year follow-up from the CARE trial, which compared sacrocolpopexy, with and without a concomitant Burch procedure, in women who did not have symptoms of stress urinary incontinence (SUI) at the time of surgery
- a comparison of laparoscopic Burch colposuspension and the tension-free vaginal tape (TVT) technique.
The CARE trial was designed to determine whether prophylactic concomitant Burch colposuspension is indicated in these women when they undergo planned abdominal sacrocolpopexy. In this latest update, CARE investigators present 2-year outcomes.
Functional and anatomic findings
The addition of a Burch procedure at the time of sacrocolpopexy reduced the rate of postoperative SUI symptoms to a greater degree than sacrocolpopexy alone (control group) did, with 32% of women in the Burch group experiencing symptoms (versus 45.2% of controls; p=.026). Women in the Burch arm also had a lower rate of bothersome SUI (11.6% versus 25.2%; p=.004) and a tendency to a lower rate of urge symptoms (32% versus 44.5%; p=.085). In addition, the study demonstrated that sacrocolpopexy played a beneficial role in reducing bothersome irritative and obstructive urinary symptoms after surgery, regardless of concomitant Burch.
Anatomic outcomes were similar in both arms of the trial, with the apex within 2 cm of total vaginal length in 95% of women (p=.18).
Strengths, limitations of this study
Concomitant prophylactic Burch colposuspension confers sustained protection against SUI in women who are continent at the time of sacrocolpopexy. And midurethral synthetic slings—which require a shorter operative time and hospital stay and carry a low rate of perioperative complication—offer success rates similar, if not superior, to Burch. Both procedures require specialized training to perform safely.
It is reasonable to consider a prophylactic, concomitant anti-incontinence procedure in the form of a Burch colposuspension at the time of sacrocolpopexy. We believe that a concomitant or staged (interval) midurethral sling operation is a sound alternative 1) depending on the patient’s preference and 2) after review of the available evidence and potential risks and benefits.—BELA I. KUDISH, MD, MS, AND CHERYL IGLESIA, MD
1. Brubaker L, Cundiff GW, Fine P, et al. For the Pelvic Floor Disorders Network. Abdominal sacrocolpopexy with Burch colposuspension to reduce urinary stress incontinence. N Engl J Med. 2006;354:1557-1566.
2. Fatton B. Is there any evidence to advocate SUI prevention in continent women undergoing prolapse repair? An overview. Int Urogynecol J Pelvic Floor Dysfunct. 2008 [Epub ahead of print].
Just 2 years ago, when Brubaker and colleagues published initial findings from the colpopexy and urinary reduction efforts (CARE) trial in the New England Journal of Medicine,1 Burch colposuspension was a well-established anti-incontinence procedure utilized by many urogynecologists. The procedure remains a reliable intervention, although midurethral sling procedures have surpassed it in popularity and (some would say) efficacy. This issue’s installment of Examining the Evidence highlights two recent investigations of the antiincontinence procedure:
- 2-year follow-up from the CARE trial, which compared sacrocolpopexy, with and without a concomitant Burch procedure, in women who did not have symptoms of stress urinary incontinence (SUI) at the time of surgery
- a comparison of laparoscopic Burch colposuspension and the tension-free vaginal tape (TVT) technique.
The CARE trial was designed to determine whether prophylactic concomitant Burch colposuspension is indicated in these women when they undergo planned abdominal sacrocolpopexy. In this latest update, CARE investigators present 2-year outcomes.
Functional and anatomic findings
The addition of a Burch procedure at the time of sacrocolpopexy reduced the rate of postoperative SUI symptoms to a greater degree than sacrocolpopexy alone (control group) did, with 32% of women in the Burch group experiencing symptoms (versus 45.2% of controls; p=.026). Women in the Burch arm also had a lower rate of bothersome SUI (11.6% versus 25.2%; p=.004) and a tendency to a lower rate of urge symptoms (32% versus 44.5%; p=.085). In addition, the study demonstrated that sacrocolpopexy played a beneficial role in reducing bothersome irritative and obstructive urinary symptoms after surgery, regardless of concomitant Burch.
Anatomic outcomes were similar in both arms of the trial, with the apex within 2 cm of total vaginal length in 95% of women (p=.18).
Strengths, limitations of this study
Concomitant prophylactic Burch colposuspension confers sustained protection against SUI in women who are continent at the time of sacrocolpopexy. And midurethral synthetic slings—which require a shorter operative time and hospital stay and carry a low rate of perioperative complication—offer success rates similar, if not superior, to Burch. Both procedures require specialized training to perform safely.
It is reasonable to consider a prophylactic, concomitant anti-incontinence procedure in the form of a Burch colposuspension at the time of sacrocolpopexy. We believe that a concomitant or staged (interval) midurethral sling operation is a sound alternative 1) depending on the patient’s preference and 2) after review of the available evidence and potential risks and benefits.—BELA I. KUDISH, MD, MS, AND CHERYL IGLESIA, MD
Just 2 years ago, when Brubaker and colleagues published initial findings from the colpopexy and urinary reduction efforts (CARE) trial in the New England Journal of Medicine,1 Burch colposuspension was a well-established anti-incontinence procedure utilized by many urogynecologists. The procedure remains a reliable intervention, although midurethral sling procedures have surpassed it in popularity and (some would say) efficacy. This issue’s installment of Examining the Evidence highlights two recent investigations of the antiincontinence procedure:
- 2-year follow-up from the CARE trial, which compared sacrocolpopexy, with and without a concomitant Burch procedure, in women who did not have symptoms of stress urinary incontinence (SUI) at the time of surgery
- a comparison of laparoscopic Burch colposuspension and the tension-free vaginal tape (TVT) technique.
The CARE trial was designed to determine whether prophylactic concomitant Burch colposuspension is indicated in these women when they undergo planned abdominal sacrocolpopexy. In this latest update, CARE investigators present 2-year outcomes.
Functional and anatomic findings
The addition of a Burch procedure at the time of sacrocolpopexy reduced the rate of postoperative SUI symptoms to a greater degree than sacrocolpopexy alone (control group) did, with 32% of women in the Burch group experiencing symptoms (versus 45.2% of controls; p=.026). Women in the Burch arm also had a lower rate of bothersome SUI (11.6% versus 25.2%; p=.004) and a tendency to a lower rate of urge symptoms (32% versus 44.5%; p=.085). In addition, the study demonstrated that sacrocolpopexy played a beneficial role in reducing bothersome irritative and obstructive urinary symptoms after surgery, regardless of concomitant Burch.
Anatomic outcomes were similar in both arms of the trial, with the apex within 2 cm of total vaginal length in 95% of women (p=.18).
Strengths, limitations of this study
Concomitant prophylactic Burch colposuspension confers sustained protection against SUI in women who are continent at the time of sacrocolpopexy. And midurethral synthetic slings—which require a shorter operative time and hospital stay and carry a low rate of perioperative complication—offer success rates similar, if not superior, to Burch. Both procedures require specialized training to perform safely.
It is reasonable to consider a prophylactic, concomitant anti-incontinence procedure in the form of a Burch colposuspension at the time of sacrocolpopexy. We believe that a concomitant or staged (interval) midurethral sling operation is a sound alternative 1) depending on the patient’s preference and 2) after review of the available evidence and potential risks and benefits.—BELA I. KUDISH, MD, MS, AND CHERYL IGLESIA, MD
1. Brubaker L, Cundiff GW, Fine P, et al. For the Pelvic Floor Disorders Network. Abdominal sacrocolpopexy with Burch colposuspension to reduce urinary stress incontinence. N Engl J Med. 2006;354:1557-1566.
2. Fatton B. Is there any evidence to advocate SUI prevention in continent women undergoing prolapse repair? An overview. Int Urogynecol J Pelvic Floor Dysfunct. 2008 [Epub ahead of print].
1. Brubaker L, Cundiff GW, Fine P, et al. For the Pelvic Floor Disorders Network. Abdominal sacrocolpopexy with Burch colposuspension to reduce urinary stress incontinence. N Engl J Med. 2006;354:1557-1566.
2. Fatton B. Is there any evidence to advocate SUI prevention in continent women undergoing prolapse repair? An overview. Int Urogynecol J Pelvic Floor Dysfunct. 2008 [Epub ahead of print].
FDA alert: Transvaginal placement of surgical mesh carries serious risks
These complications have been documented in more than 1,000 reports from nine surgical mesh manufacturers over the past 3 years. Besides the complications described above, they include:
- recurrent prolapse or incontinence (or both)
- bowel, bladder, and blood-vessel perforation during insertion
- vaginal scarring.
In many cases, additional surgery was required, as were intravenous therapy, blood transfusion, and drainage of hematoma or abscess.
Who is at risk?
Although the FDA has not determined whether specific patient characteristics increase the risk of complication, it notes that poor health overall and low estrogen levels may contribute. Other potential variables include the specific mesh material (as well as its size and shape), the surgical technique used, and whether concomitant procedures were undertaken.
The FDA advises physicians to…
- obtain specialized training for each mesh-placement technique
- watch for potential adverse events, especially erosion and infection
- watch for complications associated with surgery itself, such as bowel perforation
- tell the patient that surgical-mesh implantation is permanent, and warn her of potential complications, including the possible need for additional surgery
- give each patient a written copy of patient labeling from the surgical mesh manufacturer.
• www.fda.gov/cdrh/safety/102008-surgicalmesh.html (health-care providers)
• www.fda.gov/cdrh/consumer/surgicalmesh-popsui.html (consumers)
Also, return here in January 2009, when OBG Management features a roundtable on using mesh in prolapse repair, moderated by Mickey M. Karram, MD.
These complications have been documented in more than 1,000 reports from nine surgical mesh manufacturers over the past 3 years. Besides the complications described above, they include:
- recurrent prolapse or incontinence (or both)
- bowel, bladder, and blood-vessel perforation during insertion
- vaginal scarring.
In many cases, additional surgery was required, as were intravenous therapy, blood transfusion, and drainage of hematoma or abscess.
Who is at risk?
Although the FDA has not determined whether specific patient characteristics increase the risk of complication, it notes that poor health overall and low estrogen levels may contribute. Other potential variables include the specific mesh material (as well as its size and shape), the surgical technique used, and whether concomitant procedures were undertaken.
The FDA advises physicians to…
- obtain specialized training for each mesh-placement technique
- watch for potential adverse events, especially erosion and infection
- watch for complications associated with surgery itself, such as bowel perforation
- tell the patient that surgical-mesh implantation is permanent, and warn her of potential complications, including the possible need for additional surgery
- give each patient a written copy of patient labeling from the surgical mesh manufacturer.
• www.fda.gov/cdrh/safety/102008-surgicalmesh.html (health-care providers)
• www.fda.gov/cdrh/consumer/surgicalmesh-popsui.html (consumers)
Also, return here in January 2009, when OBG Management features a roundtable on using mesh in prolapse repair, moderated by Mickey M. Karram, MD.
These complications have been documented in more than 1,000 reports from nine surgical mesh manufacturers over the past 3 years. Besides the complications described above, they include:
- recurrent prolapse or incontinence (or both)
- bowel, bladder, and blood-vessel perforation during insertion
- vaginal scarring.
In many cases, additional surgery was required, as were intravenous therapy, blood transfusion, and drainage of hematoma or abscess.
Who is at risk?
Although the FDA has not determined whether specific patient characteristics increase the risk of complication, it notes that poor health overall and low estrogen levels may contribute. Other potential variables include the specific mesh material (as well as its size and shape), the surgical technique used, and whether concomitant procedures were undertaken.
The FDA advises physicians to…
- obtain specialized training for each mesh-placement technique
- watch for potential adverse events, especially erosion and infection
- watch for complications associated with surgery itself, such as bowel perforation
- tell the patient that surgical-mesh implantation is permanent, and warn her of potential complications, including the possible need for additional surgery
- give each patient a written copy of patient labeling from the surgical mesh manufacturer.
• www.fda.gov/cdrh/safety/102008-surgicalmesh.html (health-care providers)
• www.fda.gov/cdrh/consumer/surgicalmesh-popsui.html (consumers)
Also, return here in January 2009, when OBG Management features a roundtable on using mesh in prolapse repair, moderated by Mickey M. Karram, MD.
Timing of antibiotic prophylaxis for C-section: Better before incision?
There is already a solid body of literature to support preoperative antibiotic prophylaxis for other clean or clean-contaminated surgery.1 There is also substantive evidence that both elective and intrapartum C-sections benefit from antibiotic prophylaxis, with a statistically and clinically significant reduction in endometritis, wound infection, and composite morbidities, compared with no treatment.2 There has been resistance to pre-operative antibiotics for C-section because of a theoretical risk of masking neonatal sepsis.
Strengths of the study
- It was powered sufficiently to answer the fundamental question: Is there a benefit to the mother or risk to the newborn from preoperative antibiotics?
- The studies included were homogenous
- The analysis had biological plausibility
- The findings are congruent with other studies of antibiotic prophylaxis
- The same antibiotic was used in all studies, and both labored and elective cesarean deliveries were included.
Still a question about precise timing
More studies are needed to determine whether the dose of antibiotics should be weight-based and repeated for prolonged cases, as has been suggested for other surgical procedures. The optimal window for preoperative antibiotics also needs to be delineated. For scheduled procedures, including cesarean delivery, there is the luxury of timing the antibiotics fairly precisely 1 or 2 hours before skin incision. Intrapartum cesarean delivery is often unpredictable, is sometimes done in an urgent manner, and arguably carries an even higher risk of infectious morbidity.
This meta-analysis provides good evidence that we should change our practice to administer prophylactic antibiotics 1) for all cesarean deliveries and 2) before the skin incision whenever possible.—AVIVA LEE-PARRITZ, MD
1. Classen DC, Evans RS, Pestotnick SL, Horn SD, Menlove RL, Burke JP. The timing of prophylactic antibiotics and the risk of surgical wound infection. N Engl J Med. 1992;326:281-286.
2. Smaill F, Hofmeyr GJ. Antibiotic prophylaxis for cesarean section. Cochrane Database Syst Rev. 2002;(3):CD000933.-
There is already a solid body of literature to support preoperative antibiotic prophylaxis for other clean or clean-contaminated surgery.1 There is also substantive evidence that both elective and intrapartum C-sections benefit from antibiotic prophylaxis, with a statistically and clinically significant reduction in endometritis, wound infection, and composite morbidities, compared with no treatment.2 There has been resistance to pre-operative antibiotics for C-section because of a theoretical risk of masking neonatal sepsis.
Strengths of the study
- It was powered sufficiently to answer the fundamental question: Is there a benefit to the mother or risk to the newborn from preoperative antibiotics?
- The studies included were homogenous
- The analysis had biological plausibility
- The findings are congruent with other studies of antibiotic prophylaxis
- The same antibiotic was used in all studies, and both labored and elective cesarean deliveries were included.
Still a question about precise timing
More studies are needed to determine whether the dose of antibiotics should be weight-based and repeated for prolonged cases, as has been suggested for other surgical procedures. The optimal window for preoperative antibiotics also needs to be delineated. For scheduled procedures, including cesarean delivery, there is the luxury of timing the antibiotics fairly precisely 1 or 2 hours before skin incision. Intrapartum cesarean delivery is often unpredictable, is sometimes done in an urgent manner, and arguably carries an even higher risk of infectious morbidity.
This meta-analysis provides good evidence that we should change our practice to administer prophylactic antibiotics 1) for all cesarean deliveries and 2) before the skin incision whenever possible.—AVIVA LEE-PARRITZ, MD
There is already a solid body of literature to support preoperative antibiotic prophylaxis for other clean or clean-contaminated surgery.1 There is also substantive evidence that both elective and intrapartum C-sections benefit from antibiotic prophylaxis, with a statistically and clinically significant reduction in endometritis, wound infection, and composite morbidities, compared with no treatment.2 There has been resistance to pre-operative antibiotics for C-section because of a theoretical risk of masking neonatal sepsis.
Strengths of the study
- It was powered sufficiently to answer the fundamental question: Is there a benefit to the mother or risk to the newborn from preoperative antibiotics?
- The studies included were homogenous
- The analysis had biological plausibility
- The findings are congruent with other studies of antibiotic prophylaxis
- The same antibiotic was used in all studies, and both labored and elective cesarean deliveries were included.
Still a question about precise timing
More studies are needed to determine whether the dose of antibiotics should be weight-based and repeated for prolonged cases, as has been suggested for other surgical procedures. The optimal window for preoperative antibiotics also needs to be delineated. For scheduled procedures, including cesarean delivery, there is the luxury of timing the antibiotics fairly precisely 1 or 2 hours before skin incision. Intrapartum cesarean delivery is often unpredictable, is sometimes done in an urgent manner, and arguably carries an even higher risk of infectious morbidity.
This meta-analysis provides good evidence that we should change our practice to administer prophylactic antibiotics 1) for all cesarean deliveries and 2) before the skin incision whenever possible.—AVIVA LEE-PARRITZ, MD
1. Classen DC, Evans RS, Pestotnick SL, Horn SD, Menlove RL, Burke JP. The timing of prophylactic antibiotics and the risk of surgical wound infection. N Engl J Med. 1992;326:281-286.
2. Smaill F, Hofmeyr GJ. Antibiotic prophylaxis for cesarean section. Cochrane Database Syst Rev. 2002;(3):CD000933.-
1. Classen DC, Evans RS, Pestotnick SL, Horn SD, Menlove RL, Burke JP. The timing of prophylactic antibiotics and the risk of surgical wound infection. N Engl J Med. 1992;326:281-286.
2. Smaill F, Hofmeyr GJ. Antibiotic prophylaxis for cesarean section. Cochrane Database Syst Rev. 2002;(3):CD000933.-
Does the HRT-associated risk of breast cancer vary by histologic type and hormone regimen?
Using data from their large population-based, frequency-matched control study from Germany (3,464 breast cancer cases, 6,657 matched controls), they found a higher risk of lobular cancer with use of norethisterone- and levonorgestrel-derived progestogens than with progestogens derived from progesterone.
Other studies have also suggested that women who use estrogen–progestogen therapy (EPT) are more likely to be given a diagnosis of lobular, rather than ductal, cancer, and that various dosages, routes of administration, frequency of use, and type of estrogen or progestogen differentially affect the type of breast cancer—although the reason for this intriguing observation is unknown.
Fournier and associates, for example, found that EPT regimens containing progesterone or dydrogesterone were associated with a lower risk of breast cancer than other types of progestogen.1 The Women’s Health Initiative (WHI) evaluated only one type of oral EPT and found too few lobular cancers to answer the question of whether breast cancers found in women in the EPT group differed by histologic type.2
Li and colleagues3 and Flesch-Janys and associates found no statistically significant risk of breast cancer 5 years after EPT was discontinued. Heiss and colleagues reported that, 2.4 years after cessation of EPT, the risk of breast cancer remained stable for prior users in the WHI, compared with women taking placebo (5-year data are not yet available).4
Estrogen-only therapy in the WHI was not associated with an increased risk after 6.7 years of use.5
In the study by Flesch-Janys and associates, 67.9% of cases and 59.5% of controls had ever used HRT. Cases had a higher age at menopause and lower parity, and were less likely to have breastfed, more likely to have a family history of breast cancer, and more likely to have had benign breast disease.
Study design may invite bias
Case-control methodology is appropriate to study rare cases, examine conditions that develop over time, and generate preliminary hypotheses. Bias can occur, however, depending on the quality of existing records or self-recall and whether it is possible to collect information from all eligible controls.
Recall bias can occur because cases tend to provide more accurate information than controls. In the study by Flesch-Janys and colleagues, trained interviewers conducted face-to-face interviews to limit recall bias, but the low response rate, particularly among controls, is of concern, as is the variety of HRT preparations used by participants. The lack of verification of histologic subtype or re-review of histology by blinded pathologists limits the generalizability of the findings.
Lobular carcinoma, found more often in this study, accounts for only about 15% of invasive breast cancers. It is usually hormonally sensitive and considered more treatable than ductal cancer. However, it is more difficult to detect by mammography and physical examination.
Encourage your patients to establish their risk of breast cancer using the Gail model or another tool; to perform breast examination; to undergo mammography; and, in a setting of high risk, to consider magnetic resonance imaging of the breasts. Counsel patients that the risk of breast cancer may be diminished by regular exercise, weight loss, smoking cessation, limitation of alcohol, and adequate intake of vitamin D.
If hormone therapy is indicated in a given patient, using a lower dosage or, potentially, different types of estrogen–progestogen regimens could minimize the risk of breast cancer, although more research on these measures is needed before recommendations can be made.
For women who are at high risk of breast cancer, you can offer Food and Drug Administration-approved chemoprevention with tamoxifen or raloxifene.—JOANN V. PINKERTON, MD
1. Fournier A, Fabre A, Mesrine S, Boutron-Rualt M-C, Berrino F, Clavel-Chapelon F. Use of different postmenopausal hormone therapies and risk of histology- and hormone receptor-defined invasive breast cancer. J Clin Oncol. 2008;26:1260-1268.
2. Anderson GL, Chlebowski RT, Rossouw JE, et al. Prior hormone therapy and breast cancer risk in the Women’s Health Initiative randomized trial of estrogen plus progestin. Maturitas. 2006;55:103-115.
3. Li CI, Malone KE, Porter PL, et al. Relationship between menopausal hormone therapy and risk of ductal, lobular, and ductal–lobular breast carcinomas. Cancer Epidemiol Biomarkers Prev. 2008;17:43-50.
4. Heiss G, Wallace R, Anderson GL, et al. For the WHI Investigators. Health risks and benefits 3 years after stopping randomized treatment with estrogen and progestin. JAMA. 2008;299:1036-1045.
5. Prentice RL, Chlebowski RT, Stefanick ML, et al. Conjugated equine estrogens and breast cancer risk in the Women’s Health Initiative clinical trial and observational study. Am J Epidemiol. 2008;167:1407-1415.
Using data from their large population-based, frequency-matched control study from Germany (3,464 breast cancer cases, 6,657 matched controls), they found a higher risk of lobular cancer with use of norethisterone- and levonorgestrel-derived progestogens than with progestogens derived from progesterone.
Other studies have also suggested that women who use estrogen–progestogen therapy (EPT) are more likely to be given a diagnosis of lobular, rather than ductal, cancer, and that various dosages, routes of administration, frequency of use, and type of estrogen or progestogen differentially affect the type of breast cancer—although the reason for this intriguing observation is unknown.
Fournier and associates, for example, found that EPT regimens containing progesterone or dydrogesterone were associated with a lower risk of breast cancer than other types of progestogen.1 The Women’s Health Initiative (WHI) evaluated only one type of oral EPT and found too few lobular cancers to answer the question of whether breast cancers found in women in the EPT group differed by histologic type.2
Li and colleagues3 and Flesch-Janys and associates found no statistically significant risk of breast cancer 5 years after EPT was discontinued. Heiss and colleagues reported that, 2.4 years after cessation of EPT, the risk of breast cancer remained stable for prior users in the WHI, compared with women taking placebo (5-year data are not yet available).4
Estrogen-only therapy in the WHI was not associated with an increased risk after 6.7 years of use.5
In the study by Flesch-Janys and associates, 67.9% of cases and 59.5% of controls had ever used HRT. Cases had a higher age at menopause and lower parity, and were less likely to have breastfed, more likely to have a family history of breast cancer, and more likely to have had benign breast disease.
Study design may invite bias
Case-control methodology is appropriate to study rare cases, examine conditions that develop over time, and generate preliminary hypotheses. Bias can occur, however, depending on the quality of existing records or self-recall and whether it is possible to collect information from all eligible controls.
Recall bias can occur because cases tend to provide more accurate information than controls. In the study by Flesch-Janys and colleagues, trained interviewers conducted face-to-face interviews to limit recall bias, but the low response rate, particularly among controls, is of concern, as is the variety of HRT preparations used by participants. The lack of verification of histologic subtype or re-review of histology by blinded pathologists limits the generalizability of the findings.
Lobular carcinoma, found more often in this study, accounts for only about 15% of invasive breast cancers. It is usually hormonally sensitive and considered more treatable than ductal cancer. However, it is more difficult to detect by mammography and physical examination.
Encourage your patients to establish their risk of breast cancer using the Gail model or another tool; to perform breast examination; to undergo mammography; and, in a setting of high risk, to consider magnetic resonance imaging of the breasts. Counsel patients that the risk of breast cancer may be diminished by regular exercise, weight loss, smoking cessation, limitation of alcohol, and adequate intake of vitamin D.
If hormone therapy is indicated in a given patient, using a lower dosage or, potentially, different types of estrogen–progestogen regimens could minimize the risk of breast cancer, although more research on these measures is needed before recommendations can be made.
For women who are at high risk of breast cancer, you can offer Food and Drug Administration-approved chemoprevention with tamoxifen or raloxifene.—JOANN V. PINKERTON, MD
Using data from their large population-based, frequency-matched control study from Germany (3,464 breast cancer cases, 6,657 matched controls), they found a higher risk of lobular cancer with use of norethisterone- and levonorgestrel-derived progestogens than with progestogens derived from progesterone.
Other studies have also suggested that women who use estrogen–progestogen therapy (EPT) are more likely to be given a diagnosis of lobular, rather than ductal, cancer, and that various dosages, routes of administration, frequency of use, and type of estrogen or progestogen differentially affect the type of breast cancer—although the reason for this intriguing observation is unknown.
Fournier and associates, for example, found that EPT regimens containing progesterone or dydrogesterone were associated with a lower risk of breast cancer than other types of progestogen.1 The Women’s Health Initiative (WHI) evaluated only one type of oral EPT and found too few lobular cancers to answer the question of whether breast cancers found in women in the EPT group differed by histologic type.2
Li and colleagues3 and Flesch-Janys and associates found no statistically significant risk of breast cancer 5 years after EPT was discontinued. Heiss and colleagues reported that, 2.4 years after cessation of EPT, the risk of breast cancer remained stable for prior users in the WHI, compared with women taking placebo (5-year data are not yet available).4
Estrogen-only therapy in the WHI was not associated with an increased risk after 6.7 years of use.5
In the study by Flesch-Janys and associates, 67.9% of cases and 59.5% of controls had ever used HRT. Cases had a higher age at menopause and lower parity, and were less likely to have breastfed, more likely to have a family history of breast cancer, and more likely to have had benign breast disease.
Study design may invite bias
Case-control methodology is appropriate to study rare cases, examine conditions that develop over time, and generate preliminary hypotheses. Bias can occur, however, depending on the quality of existing records or self-recall and whether it is possible to collect information from all eligible controls.
Recall bias can occur because cases tend to provide more accurate information than controls. In the study by Flesch-Janys and colleagues, trained interviewers conducted face-to-face interviews to limit recall bias, but the low response rate, particularly among controls, is of concern, as is the variety of HRT preparations used by participants. The lack of verification of histologic subtype or re-review of histology by blinded pathologists limits the generalizability of the findings.
Lobular carcinoma, found more often in this study, accounts for only about 15% of invasive breast cancers. It is usually hormonally sensitive and considered more treatable than ductal cancer. However, it is more difficult to detect by mammography and physical examination.
Encourage your patients to establish their risk of breast cancer using the Gail model or another tool; to perform breast examination; to undergo mammography; and, in a setting of high risk, to consider magnetic resonance imaging of the breasts. Counsel patients that the risk of breast cancer may be diminished by regular exercise, weight loss, smoking cessation, limitation of alcohol, and adequate intake of vitamin D.
If hormone therapy is indicated in a given patient, using a lower dosage or, potentially, different types of estrogen–progestogen regimens could minimize the risk of breast cancer, although more research on these measures is needed before recommendations can be made.
For women who are at high risk of breast cancer, you can offer Food and Drug Administration-approved chemoprevention with tamoxifen or raloxifene.—JOANN V. PINKERTON, MD
1. Fournier A, Fabre A, Mesrine S, Boutron-Rualt M-C, Berrino F, Clavel-Chapelon F. Use of different postmenopausal hormone therapies and risk of histology- and hormone receptor-defined invasive breast cancer. J Clin Oncol. 2008;26:1260-1268.
2. Anderson GL, Chlebowski RT, Rossouw JE, et al. Prior hormone therapy and breast cancer risk in the Women’s Health Initiative randomized trial of estrogen plus progestin. Maturitas. 2006;55:103-115.
3. Li CI, Malone KE, Porter PL, et al. Relationship between menopausal hormone therapy and risk of ductal, lobular, and ductal–lobular breast carcinomas. Cancer Epidemiol Biomarkers Prev. 2008;17:43-50.
4. Heiss G, Wallace R, Anderson GL, et al. For the WHI Investigators. Health risks and benefits 3 years after stopping randomized treatment with estrogen and progestin. JAMA. 2008;299:1036-1045.
5. Prentice RL, Chlebowski RT, Stefanick ML, et al. Conjugated equine estrogens and breast cancer risk in the Women’s Health Initiative clinical trial and observational study. Am J Epidemiol. 2008;167:1407-1415.
1. Fournier A, Fabre A, Mesrine S, Boutron-Rualt M-C, Berrino F, Clavel-Chapelon F. Use of different postmenopausal hormone therapies and risk of histology- and hormone receptor-defined invasive breast cancer. J Clin Oncol. 2008;26:1260-1268.
2. Anderson GL, Chlebowski RT, Rossouw JE, et al. Prior hormone therapy and breast cancer risk in the Women’s Health Initiative randomized trial of estrogen plus progestin. Maturitas. 2006;55:103-115.
3. Li CI, Malone KE, Porter PL, et al. Relationship between menopausal hormone therapy and risk of ductal, lobular, and ductal–lobular breast carcinomas. Cancer Epidemiol Biomarkers Prev. 2008;17:43-50.
4. Heiss G, Wallace R, Anderson GL, et al. For the WHI Investigators. Health risks and benefits 3 years after stopping randomized treatment with estrogen and progestin. JAMA. 2008;299:1036-1045.
5. Prentice RL, Chlebowski RT, Stefanick ML, et al. Conjugated equine estrogens and breast cancer risk in the Women’s Health Initiative clinical trial and observational study. Am J Epidemiol. 2008;167:1407-1415.
Does menopausal estrogen therapy increase the risk of benign proliferative breast disease?
EXPERT COMMENTARY
Benign proliferative breast disease (BPBD) is a relatively common condition that is diagnosed by biopsy. It is important because of the economic and psychological burdens of biopsy and because BPBD may be a precursor to breast cancer. Some experts believe that the earliest phase of a continuum leading to invasive cancer is BPBD without atypia, followed by BPBD with atypia, in situ cancer, and then invasive disease.
Population was derived from WHI study
In this report, the risk of incident BPBD was compared among hysterectomized women 50 to 79 years old who participated in the Women’s Health Initiative (WHI). These women were randomized to conjugated equine estrogen (CEE) or placebo and monitored for, on average, 6.9 years of follow-up. They underwent baseline and annual clinical breast examination and mammography.
Overall, 232 new cases of BPBD were diagnosed among 10,739 women—approximately two thirds of them in the group randomized to CEE. Compared with women randomized to placebo, women taking CEE had twice the risk of being diagnosed with BPBD without atypia (hazard ratio [HR], 2.34; 95% confidence interval [CI], 1.71–3.20). For the subgroup of women with BPBD in whom histopathology was moderately extensive or florid (but lacking atypia), risk was similarly elevated (HR, 2.22).
Use of CEE was not associated with a substantially increased risk of atypical hyperplasia.
Limitations of the study
This trial tested only one estrogen regimen and dosage. It also was terminated earlier than expected, which may have rendered the risk estimates less precise. In addition, a significant number of participants stopped taking CEE during the trial.
This study found a significantly heightened risk of BPBD with use of unopposed estrogen, raising concerns about the potential for subsequent breast cancer. However, the same WHI estrogen-only trial from which this study1 derives, as well as the Nurses Health Study,2 found no increased risk of invasive breast cancer with estrogen therapy.
The authors of this WHI report suggest that longer follow-up of participants may help resolve this contradiction. In the meantime, some clinicians may choose to advise women who are beginning unopposed estrogen therapy that they face an elevated risk of breast biopsy for benign breast disease.—ANDREW M. KAUNITZ, MD
1. Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy. JAMA. 2004;291:1701-1712.
2. Chen WY, Manson JE, Hankinson SE, et al. Unopposed estrogen therapy and the risk of invasive breast cancer. Arch Intern Med. 2006;166:1027-1032.
EXPERT COMMENTARY
Benign proliferative breast disease (BPBD) is a relatively common condition that is diagnosed by biopsy. It is important because of the economic and psychological burdens of biopsy and because BPBD may be a precursor to breast cancer. Some experts believe that the earliest phase of a continuum leading to invasive cancer is BPBD without atypia, followed by BPBD with atypia, in situ cancer, and then invasive disease.
Population was derived from WHI study
In this report, the risk of incident BPBD was compared among hysterectomized women 50 to 79 years old who participated in the Women’s Health Initiative (WHI). These women were randomized to conjugated equine estrogen (CEE) or placebo and monitored for, on average, 6.9 years of follow-up. They underwent baseline and annual clinical breast examination and mammography.
Overall, 232 new cases of BPBD were diagnosed among 10,739 women—approximately two thirds of them in the group randomized to CEE. Compared with women randomized to placebo, women taking CEE had twice the risk of being diagnosed with BPBD without atypia (hazard ratio [HR], 2.34; 95% confidence interval [CI], 1.71–3.20). For the subgroup of women with BPBD in whom histopathology was moderately extensive or florid (but lacking atypia), risk was similarly elevated (HR, 2.22).
Use of CEE was not associated with a substantially increased risk of atypical hyperplasia.
Limitations of the study
This trial tested only one estrogen regimen and dosage. It also was terminated earlier than expected, which may have rendered the risk estimates less precise. In addition, a significant number of participants stopped taking CEE during the trial.
This study found a significantly heightened risk of BPBD with use of unopposed estrogen, raising concerns about the potential for subsequent breast cancer. However, the same WHI estrogen-only trial from which this study1 derives, as well as the Nurses Health Study,2 found no increased risk of invasive breast cancer with estrogen therapy.
The authors of this WHI report suggest that longer follow-up of participants may help resolve this contradiction. In the meantime, some clinicians may choose to advise women who are beginning unopposed estrogen therapy that they face an elevated risk of breast biopsy for benign breast disease.—ANDREW M. KAUNITZ, MD
EXPERT COMMENTARY
Benign proliferative breast disease (BPBD) is a relatively common condition that is diagnosed by biopsy. It is important because of the economic and psychological burdens of biopsy and because BPBD may be a precursor to breast cancer. Some experts believe that the earliest phase of a continuum leading to invasive cancer is BPBD without atypia, followed by BPBD with atypia, in situ cancer, and then invasive disease.
Population was derived from WHI study
In this report, the risk of incident BPBD was compared among hysterectomized women 50 to 79 years old who participated in the Women’s Health Initiative (WHI). These women were randomized to conjugated equine estrogen (CEE) or placebo and monitored for, on average, 6.9 years of follow-up. They underwent baseline and annual clinical breast examination and mammography.
Overall, 232 new cases of BPBD were diagnosed among 10,739 women—approximately two thirds of them in the group randomized to CEE. Compared with women randomized to placebo, women taking CEE had twice the risk of being diagnosed with BPBD without atypia (hazard ratio [HR], 2.34; 95% confidence interval [CI], 1.71–3.20). For the subgroup of women with BPBD in whom histopathology was moderately extensive or florid (but lacking atypia), risk was similarly elevated (HR, 2.22).
Use of CEE was not associated with a substantially increased risk of atypical hyperplasia.
Limitations of the study
This trial tested only one estrogen regimen and dosage. It also was terminated earlier than expected, which may have rendered the risk estimates less precise. In addition, a significant number of participants stopped taking CEE during the trial.
This study found a significantly heightened risk of BPBD with use of unopposed estrogen, raising concerns about the potential for subsequent breast cancer. However, the same WHI estrogen-only trial from which this study1 derives, as well as the Nurses Health Study,2 found no increased risk of invasive breast cancer with estrogen therapy.
The authors of this WHI report suggest that longer follow-up of participants may help resolve this contradiction. In the meantime, some clinicians may choose to advise women who are beginning unopposed estrogen therapy that they face an elevated risk of breast biopsy for benign breast disease.—ANDREW M. KAUNITZ, MD
1. Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy. JAMA. 2004;291:1701-1712.
2. Chen WY, Manson JE, Hankinson SE, et al. Unopposed estrogen therapy and the risk of invasive breast cancer. Arch Intern Med. 2006;166:1027-1032.
1. Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy. JAMA. 2004;291:1701-1712.
2. Chen WY, Manson JE, Hankinson SE, et al. Unopposed estrogen therapy and the risk of invasive breast cancer. Arch Intern Med. 2006;166:1027-1032.
Does vaginal progesterone reduce the risk of preterm birth in a high-risk woman?
EXPERT COMMENTARY
Two studies published in 2003 rekindled interest in the use of progestational agents for the prevention of recurrent preterm birth. Da Fonseca and colleagues randomized patients who had risk factors for premature birth to daily vaginal administration of a natural progesterone suppository or placebo and demonstrated a significant reduction in birth before 34 weeks.1 In a larger, multicenter trial in the United States, Meis and associates randomized patients who had a prior preterm birth to weekly administration of intramuscular 17α-progesterone caproate or placebo.2 Compared with the control group, the treatment group had a one-third reduction in births before 37 weeks, and a near 50% reduction in deliveries before 32 weeks’ gestation.
Since these studies were published, there has been much enthusiasm about the use of progesterone to reduce the incidence of preterm birth, but obstacles remain to widespread implementation of recommendations. First, 17α-progesterone caproate is not approved by the Food and Drug Administration for any indication in this country, and is not available from any major drug manufacturer. Physicians who have adopted its use for patients who have a history of preterm birth have to obtain it from a compounding pharmacy.
Use of a vaginally administered preparation would likely overcome these obstacles and meet with greater patient acceptance, but widespread use awaits further research confirming its efficacy.
In outcomes, few differences between treatment and placebo groups
O’Brien and colleagues recruited women between 18 and 23 weeks’ gestation who had previously delivered prematurely and assigned them to daily vaginal administration of 90 mg of progesterone (in gel) or placebo. Treatment was administered until 37 weeks’ gestation, delivery, or premature rupture of membranes. Outcomes were similar in the two groups, with no statistical differences in delivery before 32 or 37 weeks’ gestation, or in mean birth weight.
Nor did the groups differ in the incidence of admission for preterm labor, use of tocolytic therapy, neonatal morbidity, or NICU admission.
The authors concluded that vaginally administered progesterone does not reduce the frequency of recurrent preterm birth in women who have a history of spontaneous preterm birth.
The negative results of this well-designed study should persuade you not to adopt the vaginal route of progesterone administration as a means of preventing recurrent preterm birth. Although data have been published that vaginal progesterone reduces the rate of preterm birth in patients with a very short cervical length (≤15 mm), this is a small fraction of the patients at risk.
The final word isn’t in yet, but studies under way may shed light on the optimal dosing and route of administration for different subpopulations of patients—so that further benefit can be achieved.—ALAN M. PEACEMAN, MD
1. Da Fonseca EB, Bittar RE, Carvalho MH, Zugaib M. Prophylactic administration of progesterone by vaginal suppository to reduce the incidence of spontaneous preterm birth in women at increased risk. Am J Obstet Gynecol. 2003;188:419-424.
2. Meis PJ, Klebanoff M, Thom E, et al. For the National Institute of Child Health and Human Development Maternal–Fetal Medicine Units Network. Prevention of recurrent preterm delivery by 17-alpha-hydroxyprogesterone caproate. N Engl J Med. 2003;348:2379-2385.
EXPERT COMMENTARY
Two studies published in 2003 rekindled interest in the use of progestational agents for the prevention of recurrent preterm birth. Da Fonseca and colleagues randomized patients who had risk factors for premature birth to daily vaginal administration of a natural progesterone suppository or placebo and demonstrated a significant reduction in birth before 34 weeks.1 In a larger, multicenter trial in the United States, Meis and associates randomized patients who had a prior preterm birth to weekly administration of intramuscular 17α-progesterone caproate or placebo.2 Compared with the control group, the treatment group had a one-third reduction in births before 37 weeks, and a near 50% reduction in deliveries before 32 weeks’ gestation.
Since these studies were published, there has been much enthusiasm about the use of progesterone to reduce the incidence of preterm birth, but obstacles remain to widespread implementation of recommendations. First, 17α-progesterone caproate is not approved by the Food and Drug Administration for any indication in this country, and is not available from any major drug manufacturer. Physicians who have adopted its use for patients who have a history of preterm birth have to obtain it from a compounding pharmacy.
Use of a vaginally administered preparation would likely overcome these obstacles and meet with greater patient acceptance, but widespread use awaits further research confirming its efficacy.
In outcomes, few differences between treatment and placebo groups
O’Brien and colleagues recruited women between 18 and 23 weeks’ gestation who had previously delivered prematurely and assigned them to daily vaginal administration of 90 mg of progesterone (in gel) or placebo. Treatment was administered until 37 weeks’ gestation, delivery, or premature rupture of membranes. Outcomes were similar in the two groups, with no statistical differences in delivery before 32 or 37 weeks’ gestation, or in mean birth weight.
Nor did the groups differ in the incidence of admission for preterm labor, use of tocolytic therapy, neonatal morbidity, or NICU admission.
The authors concluded that vaginally administered progesterone does not reduce the frequency of recurrent preterm birth in women who have a history of spontaneous preterm birth.
The negative results of this well-designed study should persuade you not to adopt the vaginal route of progesterone administration as a means of preventing recurrent preterm birth. Although data have been published that vaginal progesterone reduces the rate of preterm birth in patients with a very short cervical length (≤15 mm), this is a small fraction of the patients at risk.
The final word isn’t in yet, but studies under way may shed light on the optimal dosing and route of administration for different subpopulations of patients—so that further benefit can be achieved.—ALAN M. PEACEMAN, MD
EXPERT COMMENTARY
Two studies published in 2003 rekindled interest in the use of progestational agents for the prevention of recurrent preterm birth. Da Fonseca and colleagues randomized patients who had risk factors for premature birth to daily vaginal administration of a natural progesterone suppository or placebo and demonstrated a significant reduction in birth before 34 weeks.1 In a larger, multicenter trial in the United States, Meis and associates randomized patients who had a prior preterm birth to weekly administration of intramuscular 17α-progesterone caproate or placebo.2 Compared with the control group, the treatment group had a one-third reduction in births before 37 weeks, and a near 50% reduction in deliveries before 32 weeks’ gestation.
Since these studies were published, there has been much enthusiasm about the use of progesterone to reduce the incidence of preterm birth, but obstacles remain to widespread implementation of recommendations. First, 17α-progesterone caproate is not approved by the Food and Drug Administration for any indication in this country, and is not available from any major drug manufacturer. Physicians who have adopted its use for patients who have a history of preterm birth have to obtain it from a compounding pharmacy.
Use of a vaginally administered preparation would likely overcome these obstacles and meet with greater patient acceptance, but widespread use awaits further research confirming its efficacy.
In outcomes, few differences between treatment and placebo groups
O’Brien and colleagues recruited women between 18 and 23 weeks’ gestation who had previously delivered prematurely and assigned them to daily vaginal administration of 90 mg of progesterone (in gel) or placebo. Treatment was administered until 37 weeks’ gestation, delivery, or premature rupture of membranes. Outcomes were similar in the two groups, with no statistical differences in delivery before 32 or 37 weeks’ gestation, or in mean birth weight.
Nor did the groups differ in the incidence of admission for preterm labor, use of tocolytic therapy, neonatal morbidity, or NICU admission.
The authors concluded that vaginally administered progesterone does not reduce the frequency of recurrent preterm birth in women who have a history of spontaneous preterm birth.
The negative results of this well-designed study should persuade you not to adopt the vaginal route of progesterone administration as a means of preventing recurrent preterm birth. Although data have been published that vaginal progesterone reduces the rate of preterm birth in patients with a very short cervical length (≤15 mm), this is a small fraction of the patients at risk.
The final word isn’t in yet, but studies under way may shed light on the optimal dosing and route of administration for different subpopulations of patients—so that further benefit can be achieved.—ALAN M. PEACEMAN, MD
1. Da Fonseca EB, Bittar RE, Carvalho MH, Zugaib M. Prophylactic administration of progesterone by vaginal suppository to reduce the incidence of spontaneous preterm birth in women at increased risk. Am J Obstet Gynecol. 2003;188:419-424.
2. Meis PJ, Klebanoff M, Thom E, et al. For the National Institute of Child Health and Human Development Maternal–Fetal Medicine Units Network. Prevention of recurrent preterm delivery by 17-alpha-hydroxyprogesterone caproate. N Engl J Med. 2003;348:2379-2385.
1. Da Fonseca EB, Bittar RE, Carvalho MH, Zugaib M. Prophylactic administration of progesterone by vaginal suppository to reduce the incidence of spontaneous preterm birth in women at increased risk. Am J Obstet Gynecol. 2003;188:419-424.
2. Meis PJ, Klebanoff M, Thom E, et al. For the National Institute of Child Health and Human Development Maternal–Fetal Medicine Units Network. Prevention of recurrent preterm delivery by 17-alpha-hydroxyprogesterone caproate. N Engl J Med. 2003;348:2379-2385.
Do some women with CIN 3 test negative for high-risk HPV?
EXPERT COMMENTARY
When it comes to cervical cancer prevention, screening and diagnostic tests have limits to their accuracy. Cancer-prevention strategies work because we assess patients over time and because we accept a small number of false positives as necessary to minimize the loss of sensitivity; in that way, we also minimize cancer-related morbidity.1
Link between high-risk HPV and cancer is a given
Cervical cancer is invariably linked to high-risk HPV. Women who are not truly infected with high-risk HPV are believed to have no risk for cervical cancer.2 Assay-based endpoints such as HPV DNA testing have good accuracy indices, whereas cytologic and histologic endpoints are subject to greater human error.
A CIN 3 lesion should never occur in a woman who is negative for high-risk HPV DNA. When the combination is found, which is rare, one of two mechanisms is involved:
- a falsely negative HPV test
- a falsely positive diagnosis of CIN 3.
In ALTS, falsely positive histology was probably to blame
The CIN 3 detected in women who tested negative for high-risk HPV DNA (i.e., after borderline cytology rather than because of a high-grade squamous intraepithelial lesion [HSIL]) was probably associated with falsely positive histology rather than falsely negative HPV testing.
CIN 3 in women who tested negative for high-risk HPV DNA was more likely to be:
- diagnosed at exit
- from a center where CIN 3 diagnoses were not confirmed by the Pathology
and less likely to be:
- associated with a referral Pap test classified as LSIL than as ASCUS
- associated with an enrollment Pap test classified as HSIL
- symptomatic
- associated with high-grade Cervigrams.
These women also were likely to test negative for HPV DNA using Linear Array.
In reviewing cases of CIN 3 in the 33 women who tested negative for high-risk HPV DNA, investigators found only 8 cases attributable to falsely-negative HPV testing, whereas 12 were related to incident HPV infection. Eight cases were caused by histologic overcall; 5 represented non–high-risk HPV that was unlikely to progress to cancer.
HPV testing has good, though not perfect, sensitivity for CIN 3. These findings certainly do not suggest that HPV testing is insufficiently accurate for clinical use.
Counsel women who have borderline cytology and who test negative for high-risk HPV to undergo continued testing according to the guidelines of the American Society for Colposcopy and Cervical Pathology.—DAVID G. MUTCH, MD
1. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin. Clinical management guidelines for obstetricians-gynecologists. Number 61, April 2005. Human papillomavirus. Obstet Gynecol. 2005;105:905-918.
2. Kjaer S, Høgdall E, Frederiksen K, et al. The absolute risk of cervical abnormalities in high-risk human papillomavirus-positive, cytologically normal women over a 10-year period. Cancer Res. 2006;66:10630-10636.
3. Safaeian M, Solomon D, Wacholder S, Schiffman M, Castle P. Risk of precancer and follow-up management strategies for women with human papillomavirus-negative atypical squamous cells of undetermined significance. Obstet Gynecol. 2007;109:1325-1331.
4. Schiffman M, Wheeler CM, Dasgupta A, Solomon D, Castle PE. for The ALTS Group. A comparison of a prototype PCR assay and hybrid capture 2 for detection of carcinogenic human papillomavirus DNA in women with equivocal or mildly abnormal Papanicolaou smears. Am J Clin Pathol. 2005;124:722-732.
5. Eltoum IA, Chhieng DC, Crowe DR, Roberson J, Jin G, Broker TR. Significance and possible causes of false-negative results of reflex human papillomavirus infection testing. Cancer. 2007;111:154-159.
EXPERT COMMENTARY
When it comes to cervical cancer prevention, screening and diagnostic tests have limits to their accuracy. Cancer-prevention strategies work because we assess patients over time and because we accept a small number of false positives as necessary to minimize the loss of sensitivity; in that way, we also minimize cancer-related morbidity.1
Link between high-risk HPV and cancer is a given
Cervical cancer is invariably linked to high-risk HPV. Women who are not truly infected with high-risk HPV are believed to have no risk for cervical cancer.2 Assay-based endpoints such as HPV DNA testing have good accuracy indices, whereas cytologic and histologic endpoints are subject to greater human error.
A CIN 3 lesion should never occur in a woman who is negative for high-risk HPV DNA. When the combination is found, which is rare, one of two mechanisms is involved:
- a falsely negative HPV test
- a falsely positive diagnosis of CIN 3.
In ALTS, falsely positive histology was probably to blame
The CIN 3 detected in women who tested negative for high-risk HPV DNA (i.e., after borderline cytology rather than because of a high-grade squamous intraepithelial lesion [HSIL]) was probably associated with falsely positive histology rather than falsely negative HPV testing.
CIN 3 in women who tested negative for high-risk HPV DNA was more likely to be:
- diagnosed at exit
- from a center where CIN 3 diagnoses were not confirmed by the Pathology
and less likely to be:
- associated with a referral Pap test classified as LSIL than as ASCUS
- associated with an enrollment Pap test classified as HSIL
- symptomatic
- associated with high-grade Cervigrams.
These women also were likely to test negative for HPV DNA using Linear Array.
In reviewing cases of CIN 3 in the 33 women who tested negative for high-risk HPV DNA, investigators found only 8 cases attributable to falsely-negative HPV testing, whereas 12 were related to incident HPV infection. Eight cases were caused by histologic overcall; 5 represented non–high-risk HPV that was unlikely to progress to cancer.
HPV testing has good, though not perfect, sensitivity for CIN 3. These findings certainly do not suggest that HPV testing is insufficiently accurate for clinical use.
Counsel women who have borderline cytology and who test negative for high-risk HPV to undergo continued testing according to the guidelines of the American Society for Colposcopy and Cervical Pathology.—DAVID G. MUTCH, MD
EXPERT COMMENTARY
When it comes to cervical cancer prevention, screening and diagnostic tests have limits to their accuracy. Cancer-prevention strategies work because we assess patients over time and because we accept a small number of false positives as necessary to minimize the loss of sensitivity; in that way, we also minimize cancer-related morbidity.1
Link between high-risk HPV and cancer is a given
Cervical cancer is invariably linked to high-risk HPV. Women who are not truly infected with high-risk HPV are believed to have no risk for cervical cancer.2 Assay-based endpoints such as HPV DNA testing have good accuracy indices, whereas cytologic and histologic endpoints are subject to greater human error.
A CIN 3 lesion should never occur in a woman who is negative for high-risk HPV DNA. When the combination is found, which is rare, one of two mechanisms is involved:
- a falsely negative HPV test
- a falsely positive diagnosis of CIN 3.
In ALTS, falsely positive histology was probably to blame
The CIN 3 detected in women who tested negative for high-risk HPV DNA (i.e., after borderline cytology rather than because of a high-grade squamous intraepithelial lesion [HSIL]) was probably associated with falsely positive histology rather than falsely negative HPV testing.
CIN 3 in women who tested negative for high-risk HPV DNA was more likely to be:
- diagnosed at exit
- from a center where CIN 3 diagnoses were not confirmed by the Pathology
and less likely to be:
- associated with a referral Pap test classified as LSIL than as ASCUS
- associated with an enrollment Pap test classified as HSIL
- symptomatic
- associated with high-grade Cervigrams.
These women also were likely to test negative for HPV DNA using Linear Array.
In reviewing cases of CIN 3 in the 33 women who tested negative for high-risk HPV DNA, investigators found only 8 cases attributable to falsely-negative HPV testing, whereas 12 were related to incident HPV infection. Eight cases were caused by histologic overcall; 5 represented non–high-risk HPV that was unlikely to progress to cancer.
HPV testing has good, though not perfect, sensitivity for CIN 3. These findings certainly do not suggest that HPV testing is insufficiently accurate for clinical use.
Counsel women who have borderline cytology and who test negative for high-risk HPV to undergo continued testing according to the guidelines of the American Society for Colposcopy and Cervical Pathology.—DAVID G. MUTCH, MD
1. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin. Clinical management guidelines for obstetricians-gynecologists. Number 61, April 2005. Human papillomavirus. Obstet Gynecol. 2005;105:905-918.
2. Kjaer S, Høgdall E, Frederiksen K, et al. The absolute risk of cervical abnormalities in high-risk human papillomavirus-positive, cytologically normal women over a 10-year period. Cancer Res. 2006;66:10630-10636.
3. Safaeian M, Solomon D, Wacholder S, Schiffman M, Castle P. Risk of precancer and follow-up management strategies for women with human papillomavirus-negative atypical squamous cells of undetermined significance. Obstet Gynecol. 2007;109:1325-1331.
4. Schiffman M, Wheeler CM, Dasgupta A, Solomon D, Castle PE. for The ALTS Group. A comparison of a prototype PCR assay and hybrid capture 2 for detection of carcinogenic human papillomavirus DNA in women with equivocal or mildly abnormal Papanicolaou smears. Am J Clin Pathol. 2005;124:722-732.
5. Eltoum IA, Chhieng DC, Crowe DR, Roberson J, Jin G, Broker TR. Significance and possible causes of false-negative results of reflex human papillomavirus infection testing. Cancer. 2007;111:154-159.
1. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin. Clinical management guidelines for obstetricians-gynecologists. Number 61, April 2005. Human papillomavirus. Obstet Gynecol. 2005;105:905-918.
2. Kjaer S, Høgdall E, Frederiksen K, et al. The absolute risk of cervical abnormalities in high-risk human papillomavirus-positive, cytologically normal women over a 10-year period. Cancer Res. 2006;66:10630-10636.
3. Safaeian M, Solomon D, Wacholder S, Schiffman M, Castle P. Risk of precancer and follow-up management strategies for women with human papillomavirus-negative atypical squamous cells of undetermined significance. Obstet Gynecol. 2007;109:1325-1331.
4. Schiffman M, Wheeler CM, Dasgupta A, Solomon D, Castle PE. for The ALTS Group. A comparison of a prototype PCR assay and hybrid capture 2 for detection of carcinogenic human papillomavirus DNA in women with equivocal or mildly abnormal Papanicolaou smears. Am J Clin Pathol. 2005;124:722-732.
5. Eltoum IA, Chhieng DC, Crowe DR, Roberson J, Jin G, Broker TR. Significance and possible causes of false-negative results of reflex human papillomavirus infection testing. Cancer. 2007;111:154-159.