User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Clinical Edge Journal Scan Commentary: RA September 2021
Herpes zoster infection is another well-known complication of RA and its treatment, including glucocorticoid therapy. An increased incidence has recently been noted in people who use JAK inhibitors, though other bDMARDs including TNF inhibitors are also known to increase risk. Redeker et al. compare the incidence of herpes zoster in people with RA using csDMARDs, bDMARDs, and tsDMARDs using a German prospective RA registry. In nearly 14,000 patients, 559 cases of herpes zoster were documented; after adjusting for age, sex, and glucocorticoid use, an increased risk was noted for treatment with monoclonal anti-TNF therapy, B-cell directed therapy, and JAK inhibitors compared to csDMARDs, whereas soluble TNF receptor fusion protein, T cell costimulation modulators and IL-6 inhibitors were not associated with a higher risk of herpes zoster compared with csDMARDs. Unfortunately, zoster vaccination status was not extracted for all patients. The study confirms what we already know with direct risk comparison between different agents and underscores the importance of vaccination in RA patients, especially those being treated with glucocorticoids and tsDMARDs.
Finally, another important consideration in the use of bDMARDs is the increase in cancer risk due to a potential reduction in immunosurveillance. Initial meta-analyses of clinical trials of anti-TNF agents highlighted an early increase in cancer risk, though later studies including meta-analyses and registry studies with longer follow-up durations have been equivocal. Huss et al. examine a Swedish registry of people with RA and no prior history of cancer and found a small increase in cancer-risk in patients with RA compared to the general population (HR 1.2). However, there was no increase in overall cancer incidence in patients treated with TNF inhibitors, rituximab, abatacept, or JAK inhibitors compared to RA patients naïve to bDMARDs and tsDMARDs. Interestingly, urinary tract cancer risk was slightly increased in several treatment groups, though the effect size was small. Considering the generally long duration of follow-up (with the exception of JAK inhibitors), this study is very reassuring regarding long-term risk of cancer of bDMARD use and useful in counseling people with RA on therapeutic risks.
Herpes zoster infection is another well-known complication of RA and its treatment, including glucocorticoid therapy. An increased incidence has recently been noted in people who use JAK inhibitors, though other bDMARDs including TNF inhibitors are also known to increase risk. Redeker et al. compare the incidence of herpes zoster in people with RA using csDMARDs, bDMARDs, and tsDMARDs using a German prospective RA registry. In nearly 14,000 patients, 559 cases of herpes zoster were documented; after adjusting for age, sex, and glucocorticoid use, an increased risk was noted for treatment with monoclonal anti-TNF therapy, B-cell directed therapy, and JAK inhibitors compared to csDMARDs, whereas soluble TNF receptor fusion protein, T cell costimulation modulators and IL-6 inhibitors were not associated with a higher risk of herpes zoster compared with csDMARDs. Unfortunately, zoster vaccination status was not extracted for all patients. The study confirms what we already know with direct risk comparison between different agents and underscores the importance of vaccination in RA patients, especially those being treated with glucocorticoids and tsDMARDs.
Finally, another important consideration in the use of bDMARDs is the increase in cancer risk due to a potential reduction in immunosurveillance. Initial meta-analyses of clinical trials of anti-TNF agents highlighted an early increase in cancer risk, though later studies including meta-analyses and registry studies with longer follow-up durations have been equivocal. Huss et al. examine a Swedish registry of people with RA and no prior history of cancer and found a small increase in cancer-risk in patients with RA compared to the general population (HR 1.2). However, there was no increase in overall cancer incidence in patients treated with TNF inhibitors, rituximab, abatacept, or JAK inhibitors compared to RA patients naïve to bDMARDs and tsDMARDs. Interestingly, urinary tract cancer risk was slightly increased in several treatment groups, though the effect size was small. Considering the generally long duration of follow-up (with the exception of JAK inhibitors), this study is very reassuring regarding long-term risk of cancer of bDMARD use and useful in counseling people with RA on therapeutic risks.
Herpes zoster infection is another well-known complication of RA and its treatment, including glucocorticoid therapy. An increased incidence has recently been noted in people who use JAK inhibitors, though other bDMARDs including TNF inhibitors are also known to increase risk. Redeker et al. compare the incidence of herpes zoster in people with RA using csDMARDs, bDMARDs, and tsDMARDs using a German prospective RA registry. In nearly 14,000 patients, 559 cases of herpes zoster were documented; after adjusting for age, sex, and glucocorticoid use, an increased risk was noted for treatment with monoclonal anti-TNF therapy, B-cell directed therapy, and JAK inhibitors compared to csDMARDs, whereas soluble TNF receptor fusion protein, T cell costimulation modulators and IL-6 inhibitors were not associated with a higher risk of herpes zoster compared with csDMARDs. Unfortunately, zoster vaccination status was not extracted for all patients. The study confirms what we already know with direct risk comparison between different agents and underscores the importance of vaccination in RA patients, especially those being treated with glucocorticoids and tsDMARDs.
Finally, another important consideration in the use of bDMARDs is the increase in cancer risk due to a potential reduction in immunosurveillance. Initial meta-analyses of clinical trials of anti-TNF agents highlighted an early increase in cancer risk, though later studies including meta-analyses and registry studies with longer follow-up durations have been equivocal. Huss et al. examine a Swedish registry of people with RA and no prior history of cancer and found a small increase in cancer-risk in patients with RA compared to the general population (HR 1.2). However, there was no increase in overall cancer incidence in patients treated with TNF inhibitors, rituximab, abatacept, or JAK inhibitors compared to RA patients naïve to bDMARDs and tsDMARDs. Interestingly, urinary tract cancer risk was slightly increased in several treatment groups, though the effect size was small. Considering the generally long duration of follow-up (with the exception of JAK inhibitors), this study is very reassuring regarding long-term risk of cancer of bDMARD use and useful in counseling people with RA on therapeutic risks.
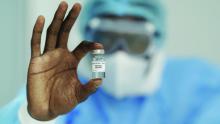
CDC panel unanimously backs Pfizer vax, fortifying FDA approval
An independent expert panel within the Centers for Disease Control and Prevention (CDC) has studied the potential benefits and risks of the Pfizer-BioNTech COVID-19 vaccine and voted unanimously to recommend the shots for all Americans ages 16 and older.
The vaccine was fully approved by the U.S. Food and Drug Administration (FDA) last week.
The inoculation is still available to teens ages 12 to 15 under an emergency use authorization from the FDA.
ACIP now sends its recommendation to the CDC Director Rochelle Walensky, MD, for her sign off.
After reviewing the evidence behind the vaccine, panel member Sarah Long, MD, a professor of pediatrics at Drexel University College of Medicine, Philadelphia, said she couldn’t recall another instance where panelists had so much data on which to base their recommendation.
“This vaccine is worthy of the trust of the American people,” she said.
Doctors across the country use vaccines in line with the recommendations made by the ACIP. Their approval typically means that private and government insurers will cover the cost of the shots. In the case of the COVID-19 vaccines, the government is already picking up the tab.
Few surprises
The panel’s independent review of the vaccine’s effectiveness from nine studies held few surprises.
They found the Pfizer vaccine prevented a COVID infection with symptoms about 90%–92% of the time, at least for the first 4 months after the second shot. Protection against hospitalization and death was even higher.
The vaccine was about 89% effective at preventing a COVID infection without symptoms, according to a pooled estimate of five studies.
The data included in the review was updated only through March 13 of this year, however, and does not reflect the impact of further waning of immunity or the impact of the Delta variant.
In making their recommendation, the panel got an update on the safety of the vaccines, which have now been used in the United States for about 9 months.
The rate of anaphylaxis has settled at around five cases for every million shots given, according to the ACIP’s review of the evidence.
Cases of myocarditis and pericarditis were more common after getting a Pfizer-BioNTech vaccine than would be expected to happen naturally in the general population, but the risk was still very rare, and elevated primarily for men younger than age 30.
Out of 17 million second doses of Pfizer-BioNTech vaccines in the United States, there have been 327 confirmed cases of myocarditis reported to the Vaccine Adverse Event Reporting System in people who are younger than age 30. The average hospital stay for a myocarditis cases is 1 to 2 days.
So far, no one in the United States diagnosed with myocarditis after vaccination has died.
What’s more, the risk of myocarditis after vaccination was dwarfed by the risk of myocarditis after a COVID infection. The risk of myocarditis after a COVID infection was 6 to 34 times higher than the risk after receiving an mRNA vaccine.
About 11% of people who get the vaccine experience a serious reaction to the shot, compared with about 3% in the placebo group. Serious reactions were defined as pain; swelling or redness at the injection site that interferes with activity; needing to visit the hospital or ER for pain; tissue necrosis, or having skin slough off; high fever; vomiting that requires hydration; persistent diarrhea; severe headache; or muscle pain/severe joint pain.
“Safe and effective”
After hearing a presentation on the state of the pandemic in the US, some panel members were struck and shaken that 38% of Americans who are eligible are still not fully vaccinated.
Pablo Sanchez, MD, a pediatrician at Nationwide Children’s Hospital in Columbus, Ohio, said, “We’re doing an abysmal job vaccinating the American people. The message has to go out that the vaccines are safe and effective.”
A version of this story first appeared on Medscape.com.
An independent expert panel within the Centers for Disease Control and Prevention (CDC) has studied the potential benefits and risks of the Pfizer-BioNTech COVID-19 vaccine and voted unanimously to recommend the shots for all Americans ages 16 and older.
The vaccine was fully approved by the U.S. Food and Drug Administration (FDA) last week.
The inoculation is still available to teens ages 12 to 15 under an emergency use authorization from the FDA.
ACIP now sends its recommendation to the CDC Director Rochelle Walensky, MD, for her sign off.
After reviewing the evidence behind the vaccine, panel member Sarah Long, MD, a professor of pediatrics at Drexel University College of Medicine, Philadelphia, said she couldn’t recall another instance where panelists had so much data on which to base their recommendation.
“This vaccine is worthy of the trust of the American people,” she said.
Doctors across the country use vaccines in line with the recommendations made by the ACIP. Their approval typically means that private and government insurers will cover the cost of the shots. In the case of the COVID-19 vaccines, the government is already picking up the tab.
Few surprises
The panel’s independent review of the vaccine’s effectiveness from nine studies held few surprises.
They found the Pfizer vaccine prevented a COVID infection with symptoms about 90%–92% of the time, at least for the first 4 months after the second shot. Protection against hospitalization and death was even higher.
The vaccine was about 89% effective at preventing a COVID infection without symptoms, according to a pooled estimate of five studies.
The data included in the review was updated only through March 13 of this year, however, and does not reflect the impact of further waning of immunity or the impact of the Delta variant.
In making their recommendation, the panel got an update on the safety of the vaccines, which have now been used in the United States for about 9 months.
The rate of anaphylaxis has settled at around five cases for every million shots given, according to the ACIP’s review of the evidence.
Cases of myocarditis and pericarditis were more common after getting a Pfizer-BioNTech vaccine than would be expected to happen naturally in the general population, but the risk was still very rare, and elevated primarily for men younger than age 30.
Out of 17 million second doses of Pfizer-BioNTech vaccines in the United States, there have been 327 confirmed cases of myocarditis reported to the Vaccine Adverse Event Reporting System in people who are younger than age 30. The average hospital stay for a myocarditis cases is 1 to 2 days.
So far, no one in the United States diagnosed with myocarditis after vaccination has died.
What’s more, the risk of myocarditis after vaccination was dwarfed by the risk of myocarditis after a COVID infection. The risk of myocarditis after a COVID infection was 6 to 34 times higher than the risk after receiving an mRNA vaccine.
About 11% of people who get the vaccine experience a serious reaction to the shot, compared with about 3% in the placebo group. Serious reactions were defined as pain; swelling or redness at the injection site that interferes with activity; needing to visit the hospital or ER for pain; tissue necrosis, or having skin slough off; high fever; vomiting that requires hydration; persistent diarrhea; severe headache; or muscle pain/severe joint pain.
“Safe and effective”
After hearing a presentation on the state of the pandemic in the US, some panel members were struck and shaken that 38% of Americans who are eligible are still not fully vaccinated.
Pablo Sanchez, MD, a pediatrician at Nationwide Children’s Hospital in Columbus, Ohio, said, “We’re doing an abysmal job vaccinating the American people. The message has to go out that the vaccines are safe and effective.”
A version of this story first appeared on Medscape.com.
An independent expert panel within the Centers for Disease Control and Prevention (CDC) has studied the potential benefits and risks of the Pfizer-BioNTech COVID-19 vaccine and voted unanimously to recommend the shots for all Americans ages 16 and older.
The vaccine was fully approved by the U.S. Food and Drug Administration (FDA) last week.
The inoculation is still available to teens ages 12 to 15 under an emergency use authorization from the FDA.
ACIP now sends its recommendation to the CDC Director Rochelle Walensky, MD, for her sign off.
After reviewing the evidence behind the vaccine, panel member Sarah Long, MD, a professor of pediatrics at Drexel University College of Medicine, Philadelphia, said she couldn’t recall another instance where panelists had so much data on which to base their recommendation.
“This vaccine is worthy of the trust of the American people,” she said.
Doctors across the country use vaccines in line with the recommendations made by the ACIP. Their approval typically means that private and government insurers will cover the cost of the shots. In the case of the COVID-19 vaccines, the government is already picking up the tab.
Few surprises
The panel’s independent review of the vaccine’s effectiveness from nine studies held few surprises.
They found the Pfizer vaccine prevented a COVID infection with symptoms about 90%–92% of the time, at least for the first 4 months after the second shot. Protection against hospitalization and death was even higher.
The vaccine was about 89% effective at preventing a COVID infection without symptoms, according to a pooled estimate of five studies.
The data included in the review was updated only through March 13 of this year, however, and does not reflect the impact of further waning of immunity or the impact of the Delta variant.
In making their recommendation, the panel got an update on the safety of the vaccines, which have now been used in the United States for about 9 months.
The rate of anaphylaxis has settled at around five cases for every million shots given, according to the ACIP’s review of the evidence.
Cases of myocarditis and pericarditis were more common after getting a Pfizer-BioNTech vaccine than would be expected to happen naturally in the general population, but the risk was still very rare, and elevated primarily for men younger than age 30.
Out of 17 million second doses of Pfizer-BioNTech vaccines in the United States, there have been 327 confirmed cases of myocarditis reported to the Vaccine Adverse Event Reporting System in people who are younger than age 30. The average hospital stay for a myocarditis cases is 1 to 2 days.
So far, no one in the United States diagnosed with myocarditis after vaccination has died.
What’s more, the risk of myocarditis after vaccination was dwarfed by the risk of myocarditis after a COVID infection. The risk of myocarditis after a COVID infection was 6 to 34 times higher than the risk after receiving an mRNA vaccine.
About 11% of people who get the vaccine experience a serious reaction to the shot, compared with about 3% in the placebo group. Serious reactions were defined as pain; swelling or redness at the injection site that interferes with activity; needing to visit the hospital or ER for pain; tissue necrosis, or having skin slough off; high fever; vomiting that requires hydration; persistent diarrhea; severe headache; or muscle pain/severe joint pain.
“Safe and effective”
After hearing a presentation on the state of the pandemic in the US, some panel members were struck and shaken that 38% of Americans who are eligible are still not fully vaccinated.
Pablo Sanchez, MD, a pediatrician at Nationwide Children’s Hospital in Columbus, Ohio, said, “We’re doing an abysmal job vaccinating the American people. The message has to go out that the vaccines are safe and effective.”
A version of this story first appeared on Medscape.com.
Biologic benefit in psoriasis might extend to arthritis prevention
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
FROM ARTHRITIS & RHEUMATOLOGY
Advocates seek to reframe masks as a disability accommodation
As governors and legislatures in states such as Texas, Florida, South Carolina, and Arkansas have banned schools and other entities from implementing mask mandates, disability rights advocates have pushed back. In federal civil rights lawsuits, they argue that bans on mask mandates violate antidiscrimination laws protecting people with disabilities.
For unvaccinated and immunosuppressed individuals, masks can provide crucial protection from SARS-CoV-2.
argues Mical Raz, MD, PhD, a professor at the University of Rochester (N.Y.) and a physician at Strong Memorial Hospital, also in Rochester, New York, in an article published in JAMA with coauthor Doron Dorfman, LLB, JSD.
This news organization talked with Dr. Raz about approaching mask requirements as disability accommodation during the COVID-19 pandemic. The following interview was lightly edited for length and clarity.
How did you come to think about mask requirements as a form of disability accommodation?
I saw a tweet from a professor at a university who said they couldn’t ask students about their vaccination status or to wear a mask. All agency was removed from the professor to take care of and protect themselves. I thought, well, that can’t be right. And ostensibly, that would be particularly dangerous for somebody with immunosuppression for whom the vaccine is not adequately protective. So, I called my friend, Doron Dorfman, and asked him to help me think through the legal part of this. We fleshed it out and wrote the article that same night.
How novel is it to view accommodations for people who are immunosuppressed through the lens of disability accommodation?
I think there has not been enough focus during the pandemic on individuals with disabilities or on how disability law can be mobilized during this pandemic to help supplement the public health law. This framework should be used a lot more because it’s good for everybody, not just for individuals with disabilities.
For example, take what’s called the “curb effect.” If you expand sidewalks, yes, it helps individuals who use a wheelchair. But it also helps me as a mom with a stroller. It helps somebody with a shopping cart, or a kid with a bike. If we adopt policies that are inclusive to those who are disadvantaged, it’s good for everybody. We should always strive to be an inclusive society, not just because it’s the right thing to do but because it really makes our society better.
How can mask requirements be used as a form of disability accommodation, as you argue in the JAMA article?
The ADA requires employers to provide reasonable accommodations for disability. In this case, the disability is your immunosuppressive status. We have an abundance of evidence showing individuals who are immunocompromised and vaccinated are still inadequately protected from the SARS-CoV-2 virus. So, there is absolute data to show individuals with immunosuppression have a disability that requires accommodation.
The ADA has a mandate requiring employers to adjust or modify policies in order to accommodate a disability. There are certain situations in which you cannot or do not need to accommodate a disability, when it would fundamentally alter the kind of employment you offer or if it’s an undue burden or hardship. But given that we’ve been wearing masks and working remotely for a year now, arguing that somehow these accommodations are no longer possible seems disingenuous.
In that way, allowing a person who’s immunocompromised to require those around them to mask is a form of modified protective policies. And in this case, those policies line up with a public health good, masking in the face of the highly contagious Delta variant ravaging our country right now.
In your view, can this argument be used in the mask debates happening right now across the country?
This argument can and should be useful for a couple of different lawsuits that are now underway in different states. I hope our article will provide further support for those suits. And I hope in school board hearings, when parents and teachers are talking about their concerns, this could be one way to argue for why we should allow mask mandates in classes. I’ve received emails from parents who said they’re going to bring this article to their school board hearing.
I also hope this could shift the narrative around the pandemic. Instead of focusing on individual responsibility – I got my vaccine shot so I’m fine – let’s focus on how we create an inclusive environment where we protect everybody, including those who cannot be vaccinated because of age or disability, or those who are vaccinated but inadequately protected because of their underlying conditions.
In the JAMA article, you talk about how our pandemic response has focused on individual health and how that individual focus can be ableist. Can you explain that point?
I think this idea that we just make our choices – like whether to get vaccinated or wear a mask, or not – and live with it really perpetuates a highly individualistic and ableist mindset. It doesn’t consider the people I admit to the hospital who are vaccinated but have a heart transplant and didn’t mount the sufficient immune response. Or even the people who chose not to be vaccinated because they were exposed to hours and hours of misinformation on TV.
We like to individualize everything, focusing on personal responsibility and choices, but a pandemic is one of those moments where everybody’s choices affect everybody else. Laying responsibility at the doorstep of each person, rather than thinking about what steps we as a society could be taking, is cheap and politically expedient. There is no public health rationale behind the bans on mask requirements in states like Texas, Iowa, and Florida. These choices are about politics. And the price is always borne by the most disadvantaged among us.
A version of this article first appeared on Medscape.com.
As governors and legislatures in states such as Texas, Florida, South Carolina, and Arkansas have banned schools and other entities from implementing mask mandates, disability rights advocates have pushed back. In federal civil rights lawsuits, they argue that bans on mask mandates violate antidiscrimination laws protecting people with disabilities.
For unvaccinated and immunosuppressed individuals, masks can provide crucial protection from SARS-CoV-2.
argues Mical Raz, MD, PhD, a professor at the University of Rochester (N.Y.) and a physician at Strong Memorial Hospital, also in Rochester, New York, in an article published in JAMA with coauthor Doron Dorfman, LLB, JSD.
This news organization talked with Dr. Raz about approaching mask requirements as disability accommodation during the COVID-19 pandemic. The following interview was lightly edited for length and clarity.
How did you come to think about mask requirements as a form of disability accommodation?
I saw a tweet from a professor at a university who said they couldn’t ask students about their vaccination status or to wear a mask. All agency was removed from the professor to take care of and protect themselves. I thought, well, that can’t be right. And ostensibly, that would be particularly dangerous for somebody with immunosuppression for whom the vaccine is not adequately protective. So, I called my friend, Doron Dorfman, and asked him to help me think through the legal part of this. We fleshed it out and wrote the article that same night.
How novel is it to view accommodations for people who are immunosuppressed through the lens of disability accommodation?
I think there has not been enough focus during the pandemic on individuals with disabilities or on how disability law can be mobilized during this pandemic to help supplement the public health law. This framework should be used a lot more because it’s good for everybody, not just for individuals with disabilities.
For example, take what’s called the “curb effect.” If you expand sidewalks, yes, it helps individuals who use a wheelchair. But it also helps me as a mom with a stroller. It helps somebody with a shopping cart, or a kid with a bike. If we adopt policies that are inclusive to those who are disadvantaged, it’s good for everybody. We should always strive to be an inclusive society, not just because it’s the right thing to do but because it really makes our society better.
How can mask requirements be used as a form of disability accommodation, as you argue in the JAMA article?
The ADA requires employers to provide reasonable accommodations for disability. In this case, the disability is your immunosuppressive status. We have an abundance of evidence showing individuals who are immunocompromised and vaccinated are still inadequately protected from the SARS-CoV-2 virus. So, there is absolute data to show individuals with immunosuppression have a disability that requires accommodation.
The ADA has a mandate requiring employers to adjust or modify policies in order to accommodate a disability. There are certain situations in which you cannot or do not need to accommodate a disability, when it would fundamentally alter the kind of employment you offer or if it’s an undue burden or hardship. But given that we’ve been wearing masks and working remotely for a year now, arguing that somehow these accommodations are no longer possible seems disingenuous.
In that way, allowing a person who’s immunocompromised to require those around them to mask is a form of modified protective policies. And in this case, those policies line up with a public health good, masking in the face of the highly contagious Delta variant ravaging our country right now.
In your view, can this argument be used in the mask debates happening right now across the country?
This argument can and should be useful for a couple of different lawsuits that are now underway in different states. I hope our article will provide further support for those suits. And I hope in school board hearings, when parents and teachers are talking about their concerns, this could be one way to argue for why we should allow mask mandates in classes. I’ve received emails from parents who said they’re going to bring this article to their school board hearing.
I also hope this could shift the narrative around the pandemic. Instead of focusing on individual responsibility – I got my vaccine shot so I’m fine – let’s focus on how we create an inclusive environment where we protect everybody, including those who cannot be vaccinated because of age or disability, or those who are vaccinated but inadequately protected because of their underlying conditions.
In the JAMA article, you talk about how our pandemic response has focused on individual health and how that individual focus can be ableist. Can you explain that point?
I think this idea that we just make our choices – like whether to get vaccinated or wear a mask, or not – and live with it really perpetuates a highly individualistic and ableist mindset. It doesn’t consider the people I admit to the hospital who are vaccinated but have a heart transplant and didn’t mount the sufficient immune response. Or even the people who chose not to be vaccinated because they were exposed to hours and hours of misinformation on TV.
We like to individualize everything, focusing on personal responsibility and choices, but a pandemic is one of those moments where everybody’s choices affect everybody else. Laying responsibility at the doorstep of each person, rather than thinking about what steps we as a society could be taking, is cheap and politically expedient. There is no public health rationale behind the bans on mask requirements in states like Texas, Iowa, and Florida. These choices are about politics. And the price is always borne by the most disadvantaged among us.
A version of this article first appeared on Medscape.com.
As governors and legislatures in states such as Texas, Florida, South Carolina, and Arkansas have banned schools and other entities from implementing mask mandates, disability rights advocates have pushed back. In federal civil rights lawsuits, they argue that bans on mask mandates violate antidiscrimination laws protecting people with disabilities.
For unvaccinated and immunosuppressed individuals, masks can provide crucial protection from SARS-CoV-2.
argues Mical Raz, MD, PhD, a professor at the University of Rochester (N.Y.) and a physician at Strong Memorial Hospital, also in Rochester, New York, in an article published in JAMA with coauthor Doron Dorfman, LLB, JSD.
This news organization talked with Dr. Raz about approaching mask requirements as disability accommodation during the COVID-19 pandemic. The following interview was lightly edited for length and clarity.
How did you come to think about mask requirements as a form of disability accommodation?
I saw a tweet from a professor at a university who said they couldn’t ask students about their vaccination status or to wear a mask. All agency was removed from the professor to take care of and protect themselves. I thought, well, that can’t be right. And ostensibly, that would be particularly dangerous for somebody with immunosuppression for whom the vaccine is not adequately protective. So, I called my friend, Doron Dorfman, and asked him to help me think through the legal part of this. We fleshed it out and wrote the article that same night.
How novel is it to view accommodations for people who are immunosuppressed through the lens of disability accommodation?
I think there has not been enough focus during the pandemic on individuals with disabilities or on how disability law can be mobilized during this pandemic to help supplement the public health law. This framework should be used a lot more because it’s good for everybody, not just for individuals with disabilities.
For example, take what’s called the “curb effect.” If you expand sidewalks, yes, it helps individuals who use a wheelchair. But it also helps me as a mom with a stroller. It helps somebody with a shopping cart, or a kid with a bike. If we adopt policies that are inclusive to those who are disadvantaged, it’s good for everybody. We should always strive to be an inclusive society, not just because it’s the right thing to do but because it really makes our society better.
How can mask requirements be used as a form of disability accommodation, as you argue in the JAMA article?
The ADA requires employers to provide reasonable accommodations for disability. In this case, the disability is your immunosuppressive status. We have an abundance of evidence showing individuals who are immunocompromised and vaccinated are still inadequately protected from the SARS-CoV-2 virus. So, there is absolute data to show individuals with immunosuppression have a disability that requires accommodation.
The ADA has a mandate requiring employers to adjust or modify policies in order to accommodate a disability. There are certain situations in which you cannot or do not need to accommodate a disability, when it would fundamentally alter the kind of employment you offer or if it’s an undue burden or hardship. But given that we’ve been wearing masks and working remotely for a year now, arguing that somehow these accommodations are no longer possible seems disingenuous.
In that way, allowing a person who’s immunocompromised to require those around them to mask is a form of modified protective policies. And in this case, those policies line up with a public health good, masking in the face of the highly contagious Delta variant ravaging our country right now.
In your view, can this argument be used in the mask debates happening right now across the country?
This argument can and should be useful for a couple of different lawsuits that are now underway in different states. I hope our article will provide further support for those suits. And I hope in school board hearings, when parents and teachers are talking about their concerns, this could be one way to argue for why we should allow mask mandates in classes. I’ve received emails from parents who said they’re going to bring this article to their school board hearing.
I also hope this could shift the narrative around the pandemic. Instead of focusing on individual responsibility – I got my vaccine shot so I’m fine – let’s focus on how we create an inclusive environment where we protect everybody, including those who cannot be vaccinated because of age or disability, or those who are vaccinated but inadequately protected because of their underlying conditions.
In the JAMA article, you talk about how our pandemic response has focused on individual health and how that individual focus can be ableist. Can you explain that point?
I think this idea that we just make our choices – like whether to get vaccinated or wear a mask, or not – and live with it really perpetuates a highly individualistic and ableist mindset. It doesn’t consider the people I admit to the hospital who are vaccinated but have a heart transplant and didn’t mount the sufficient immune response. Or even the people who chose not to be vaccinated because they were exposed to hours and hours of misinformation on TV.
We like to individualize everything, focusing on personal responsibility and choices, but a pandemic is one of those moments where everybody’s choices affect everybody else. Laying responsibility at the doorstep of each person, rather than thinking about what steps we as a society could be taking, is cheap and politically expedient. There is no public health rationale behind the bans on mask requirements in states like Texas, Iowa, and Florida. These choices are about politics. And the price is always borne by the most disadvantaged among us.
A version of this article first appeared on Medscape.com.
ACR updates COVID vaccine guidance with booster schedule
Patients on immunosuppressive or immunomodulatory therapy should receive a third dose of either the Pfizer-BioNTech COVID-19 vaccine or the Moderna COVID-19 vaccine at least 28 days after the second dose of either of these two mRNA vaccines, according to updated recommendations from the American College of Rheumatology.
The update follows the Centers for Disease Control and Prevention’s recommendation that certain immunocompromised patients receive a third dose of an mRNA vaccine to reduce their risk of contracting COVID-19.
Individuals receiving the Pfizer vaccine must be aged 12 years and older, while those receiving the Moderna vaccine must be 18 years and older, the ACR emphasized.
“These statements were based upon a dearth of high-quality data and are not intended to replace clinical judgment,” the authors wrote. “Modifications made to treatment plans, particularly in complex rheumatic disease patients, are highly disease, patient, geography, and time specific and, therefore, must be individualized as part of a shared decision-making process.”
The task force recommended using the same mRNA vaccine booster as the patient received for their initial two-dose series when possible, but notes that either mRNA vaccine is acceptable, and recommends the mRNA vaccine for patients who have yet to receive any vaccine because of the availability of the booster. The task force emphasized that they achieved no consensus on recommending a booster mRNA vaccine to patients who received a single dose of Johnson & Johnson vaccine because the safety data are uncertain.
The updated guidance also identifies the Food and Drug Administration’s emergency use authorization in August for the use of REGEN-COV monoclonal antibody treatment for emergency postexposure prophylaxis for COVID-19 in adults and adolescents aged 12 years and older who weigh at least 40 kg and are at increased risk for severe COVID-19, which includes patients receiving immunosuppressive or immunomodulatory therapies other than hydroxychloroquine. Patients who have been exposed to an individual with COVID-19 should discuss this treatment with their health care provider as an added precaution; however, the guidance emphasized that the prophylactic treatment is not a substitute for COVID-19 vaccination.
The recommendations advise clinicians to counsel their patients to refrain from taking certain immunomodulatory or immunosuppressive medications for 1-2 weeks after booster vaccination if disease activity allows, with the exception of glucocorticoids and anticytokines such as tumor necrosis factor inhibitors and others including interleukin-17, IL-12/23, IL-23, IL-1R, IL-6R antagonists, for which the task force did not achieve a consensus recommendation.
The guidance notes that patients on rituximab or other anti-CD20 medications “should discuss the optimal timing [of the booster] with their rheumatology provider” and that some practitioners measure CD19 B cells as a tool with which to time the booster and subsequent rituximab dosing. For those who elect to dose without such information, or for whom such measurement is not available or feasible, provide the booster 2-4 weeks before next anticipated rituximab dose (e.g., at month 5.0 or 5.5 for patients on an every-6-month rituximab dosing schedule).”
There was strong consensus from the task force that health care providers “should not routinely order any lab testing (e.g., antibody tests for IgM and/or IgG to spike or nucleocapsid proteins) to assess immunity to COVID-19 post vaccination, nor to assess the need for vaccination in a yet-unvaccinated person.”
“The updated information from the ACR addresses not only booster vaccination but also other important and practical issues facing rheumatology providers and their patients related to the pandemic,” said task force chair Jeffrey R. Curtis, MD, of the University of Alabama at Birmingham, in an ACR statement announcing the updates.
“Although the guidance is issued in light of the best evidence available, the science regarding COVID-19 vaccination as it affects the practice of rheumatology is undergoing rapid evolution,” he noted. “We need direct evidence such as that from randomized trials to inform the best practices of what we can do to protect our patients from SARS-CoV-2.”
The update retains the current recommendations that rheumatology patients follow all public health guidelines regarding physical distancing and other preventive measures following vaccination, but the task force did not recommend exceeding current public health guidance. “The appropriateness for continued preventive measures (e.g., masking, physical distancing) should be discussed with patients as their rheumatology providers deem appropriate,” they wrote.
The full updated version of the ACR’s COVID-19 Vaccine Clinical Guidance for Patients with Rheumatic and Musculoskeletal Diseases will be published in Arthritis & Rheumatology. The summary was developed by the ACR COVID-19 Vaccine Clinical Guidance Task Force, which included 9 rheumatologists, 2 infectious disease specialists, and 2 public health experts with current or past employment history with the CDC.
The ACR encourages clinicians with questions or concerns to email [email protected] for support.
Patients on immunosuppressive or immunomodulatory therapy should receive a third dose of either the Pfizer-BioNTech COVID-19 vaccine or the Moderna COVID-19 vaccine at least 28 days after the second dose of either of these two mRNA vaccines, according to updated recommendations from the American College of Rheumatology.
The update follows the Centers for Disease Control and Prevention’s recommendation that certain immunocompromised patients receive a third dose of an mRNA vaccine to reduce their risk of contracting COVID-19.
Individuals receiving the Pfizer vaccine must be aged 12 years and older, while those receiving the Moderna vaccine must be 18 years and older, the ACR emphasized.
“These statements were based upon a dearth of high-quality data and are not intended to replace clinical judgment,” the authors wrote. “Modifications made to treatment plans, particularly in complex rheumatic disease patients, are highly disease, patient, geography, and time specific and, therefore, must be individualized as part of a shared decision-making process.”
The task force recommended using the same mRNA vaccine booster as the patient received for their initial two-dose series when possible, but notes that either mRNA vaccine is acceptable, and recommends the mRNA vaccine for patients who have yet to receive any vaccine because of the availability of the booster. The task force emphasized that they achieved no consensus on recommending a booster mRNA vaccine to patients who received a single dose of Johnson & Johnson vaccine because the safety data are uncertain.
The updated guidance also identifies the Food and Drug Administration’s emergency use authorization in August for the use of REGEN-COV monoclonal antibody treatment for emergency postexposure prophylaxis for COVID-19 in adults and adolescents aged 12 years and older who weigh at least 40 kg and are at increased risk for severe COVID-19, which includes patients receiving immunosuppressive or immunomodulatory therapies other than hydroxychloroquine. Patients who have been exposed to an individual with COVID-19 should discuss this treatment with their health care provider as an added precaution; however, the guidance emphasized that the prophylactic treatment is not a substitute for COVID-19 vaccination.
The recommendations advise clinicians to counsel their patients to refrain from taking certain immunomodulatory or immunosuppressive medications for 1-2 weeks after booster vaccination if disease activity allows, with the exception of glucocorticoids and anticytokines such as tumor necrosis factor inhibitors and others including interleukin-17, IL-12/23, IL-23, IL-1R, IL-6R antagonists, for which the task force did not achieve a consensus recommendation.
The guidance notes that patients on rituximab or other anti-CD20 medications “should discuss the optimal timing [of the booster] with their rheumatology provider” and that some practitioners measure CD19 B cells as a tool with which to time the booster and subsequent rituximab dosing. For those who elect to dose without such information, or for whom such measurement is not available or feasible, provide the booster 2-4 weeks before next anticipated rituximab dose (e.g., at month 5.0 or 5.5 for patients on an every-6-month rituximab dosing schedule).”
There was strong consensus from the task force that health care providers “should not routinely order any lab testing (e.g., antibody tests for IgM and/or IgG to spike or nucleocapsid proteins) to assess immunity to COVID-19 post vaccination, nor to assess the need for vaccination in a yet-unvaccinated person.”
“The updated information from the ACR addresses not only booster vaccination but also other important and practical issues facing rheumatology providers and their patients related to the pandemic,” said task force chair Jeffrey R. Curtis, MD, of the University of Alabama at Birmingham, in an ACR statement announcing the updates.
“Although the guidance is issued in light of the best evidence available, the science regarding COVID-19 vaccination as it affects the practice of rheumatology is undergoing rapid evolution,” he noted. “We need direct evidence such as that from randomized trials to inform the best practices of what we can do to protect our patients from SARS-CoV-2.”
The update retains the current recommendations that rheumatology patients follow all public health guidelines regarding physical distancing and other preventive measures following vaccination, but the task force did not recommend exceeding current public health guidance. “The appropriateness for continued preventive measures (e.g., masking, physical distancing) should be discussed with patients as their rheumatology providers deem appropriate,” they wrote.
The full updated version of the ACR’s COVID-19 Vaccine Clinical Guidance for Patients with Rheumatic and Musculoskeletal Diseases will be published in Arthritis & Rheumatology. The summary was developed by the ACR COVID-19 Vaccine Clinical Guidance Task Force, which included 9 rheumatologists, 2 infectious disease specialists, and 2 public health experts with current or past employment history with the CDC.
The ACR encourages clinicians with questions or concerns to email [email protected] for support.
Patients on immunosuppressive or immunomodulatory therapy should receive a third dose of either the Pfizer-BioNTech COVID-19 vaccine or the Moderna COVID-19 vaccine at least 28 days after the second dose of either of these two mRNA vaccines, according to updated recommendations from the American College of Rheumatology.
The update follows the Centers for Disease Control and Prevention’s recommendation that certain immunocompromised patients receive a third dose of an mRNA vaccine to reduce their risk of contracting COVID-19.
Individuals receiving the Pfizer vaccine must be aged 12 years and older, while those receiving the Moderna vaccine must be 18 years and older, the ACR emphasized.
“These statements were based upon a dearth of high-quality data and are not intended to replace clinical judgment,” the authors wrote. “Modifications made to treatment plans, particularly in complex rheumatic disease patients, are highly disease, patient, geography, and time specific and, therefore, must be individualized as part of a shared decision-making process.”
The task force recommended using the same mRNA vaccine booster as the patient received for their initial two-dose series when possible, but notes that either mRNA vaccine is acceptable, and recommends the mRNA vaccine for patients who have yet to receive any vaccine because of the availability of the booster. The task force emphasized that they achieved no consensus on recommending a booster mRNA vaccine to patients who received a single dose of Johnson & Johnson vaccine because the safety data are uncertain.
The updated guidance also identifies the Food and Drug Administration’s emergency use authorization in August for the use of REGEN-COV monoclonal antibody treatment for emergency postexposure prophylaxis for COVID-19 in adults and adolescents aged 12 years and older who weigh at least 40 kg and are at increased risk for severe COVID-19, which includes patients receiving immunosuppressive or immunomodulatory therapies other than hydroxychloroquine. Patients who have been exposed to an individual with COVID-19 should discuss this treatment with their health care provider as an added precaution; however, the guidance emphasized that the prophylactic treatment is not a substitute for COVID-19 vaccination.
The recommendations advise clinicians to counsel their patients to refrain from taking certain immunomodulatory or immunosuppressive medications for 1-2 weeks after booster vaccination if disease activity allows, with the exception of glucocorticoids and anticytokines such as tumor necrosis factor inhibitors and others including interleukin-17, IL-12/23, IL-23, IL-1R, IL-6R antagonists, for which the task force did not achieve a consensus recommendation.
The guidance notes that patients on rituximab or other anti-CD20 medications “should discuss the optimal timing [of the booster] with their rheumatology provider” and that some practitioners measure CD19 B cells as a tool with which to time the booster and subsequent rituximab dosing. For those who elect to dose without such information, or for whom such measurement is not available or feasible, provide the booster 2-4 weeks before next anticipated rituximab dose (e.g., at month 5.0 or 5.5 for patients on an every-6-month rituximab dosing schedule).”
There was strong consensus from the task force that health care providers “should not routinely order any lab testing (e.g., antibody tests for IgM and/or IgG to spike or nucleocapsid proteins) to assess immunity to COVID-19 post vaccination, nor to assess the need for vaccination in a yet-unvaccinated person.”
“The updated information from the ACR addresses not only booster vaccination but also other important and practical issues facing rheumatology providers and their patients related to the pandemic,” said task force chair Jeffrey R. Curtis, MD, of the University of Alabama at Birmingham, in an ACR statement announcing the updates.
“Although the guidance is issued in light of the best evidence available, the science regarding COVID-19 vaccination as it affects the practice of rheumatology is undergoing rapid evolution,” he noted. “We need direct evidence such as that from randomized trials to inform the best practices of what we can do to protect our patients from SARS-CoV-2.”
The update retains the current recommendations that rheumatology patients follow all public health guidelines regarding physical distancing and other preventive measures following vaccination, but the task force did not recommend exceeding current public health guidance. “The appropriateness for continued preventive measures (e.g., masking, physical distancing) should be discussed with patients as their rheumatology providers deem appropriate,” they wrote.
The full updated version of the ACR’s COVID-19 Vaccine Clinical Guidance for Patients with Rheumatic and Musculoskeletal Diseases will be published in Arthritis & Rheumatology. The summary was developed by the ACR COVID-19 Vaccine Clinical Guidance Task Force, which included 9 rheumatologists, 2 infectious disease specialists, and 2 public health experts with current or past employment history with the CDC.
The ACR encourages clinicians with questions or concerns to email [email protected] for support.
Pretreatment resistin levels tied to erosive disease risk in early RA treated with DMARD and infliximab
Key clinical point: High resistin levels predict radiologically more rapidly progressing disease in patients with early active rheumatoid arthritis (RA) despite treatment with intensified disease-modifying antirheumatic drug (DMARD) combination. Adding infliximab delayed radiological progression in these patients.
Major finding: Plasma resistin levels at baseline showed positive linearity with disease activity score based on 28-joint count (P = .0072). The addition of infliximab to the DMARD combination delayed radiological progression, with no difference observed between resistin tertiles at 5 years (P = .73).
Study details: This was a post hoc analysis of the NEO-RACo trial, which enrolled 99 patients with early RA treated with intensified FIN-RACo regimen (methotrexate, sulfasalazine, hydroxychloroquine, and prednisolone). Subsequently, patients were randomly assigned to receive placebo or infliximab infusions added to the combination for 6 months.
Disclosures: The Scandinavian Rheumatology Research Foundation, the Maire Lisko Foundation, the Tampere University Hospital's Competitive Research, and others funded this study. None of the authors disclosed any potential conflict of interests.
Source: Vuolteenaho K et al. Scand J Rheumatol. 2021 Jul 15. doi: 10.1080/03009742.2021.1929456.
Key clinical point: High resistin levels predict radiologically more rapidly progressing disease in patients with early active rheumatoid arthritis (RA) despite treatment with intensified disease-modifying antirheumatic drug (DMARD) combination. Adding infliximab delayed radiological progression in these patients.
Major finding: Plasma resistin levels at baseline showed positive linearity with disease activity score based on 28-joint count (P = .0072). The addition of infliximab to the DMARD combination delayed radiological progression, with no difference observed between resistin tertiles at 5 years (P = .73).
Study details: This was a post hoc analysis of the NEO-RACo trial, which enrolled 99 patients with early RA treated with intensified FIN-RACo regimen (methotrexate, sulfasalazine, hydroxychloroquine, and prednisolone). Subsequently, patients were randomly assigned to receive placebo or infliximab infusions added to the combination for 6 months.
Disclosures: The Scandinavian Rheumatology Research Foundation, the Maire Lisko Foundation, the Tampere University Hospital's Competitive Research, and others funded this study. None of the authors disclosed any potential conflict of interests.
Source: Vuolteenaho K et al. Scand J Rheumatol. 2021 Jul 15. doi: 10.1080/03009742.2021.1929456.
Key clinical point: High resistin levels predict radiologically more rapidly progressing disease in patients with early active rheumatoid arthritis (RA) despite treatment with intensified disease-modifying antirheumatic drug (DMARD) combination. Adding infliximab delayed radiological progression in these patients.
Major finding: Plasma resistin levels at baseline showed positive linearity with disease activity score based on 28-joint count (P = .0072). The addition of infliximab to the DMARD combination delayed radiological progression, with no difference observed between resistin tertiles at 5 years (P = .73).
Study details: This was a post hoc analysis of the NEO-RACo trial, which enrolled 99 patients with early RA treated with intensified FIN-RACo regimen (methotrexate, sulfasalazine, hydroxychloroquine, and prednisolone). Subsequently, patients were randomly assigned to receive placebo or infliximab infusions added to the combination for 6 months.
Disclosures: The Scandinavian Rheumatology Research Foundation, the Maire Lisko Foundation, the Tampere University Hospital's Competitive Research, and others funded this study. None of the authors disclosed any potential conflict of interests.
Source: Vuolteenaho K et al. Scand J Rheumatol. 2021 Jul 15. doi: 10.1080/03009742.2021.1929456.
Can iguratimod be regarded as a potential alternative to methotrexate in RA?
Key clinical point: Iguratimod as monotherapy or combined therapy was remarkably effective and safe in patients with active rheumatoid arthritis (RA) compared with other disease-modifying antirheumatic drugs (DMARDs) monotherapy, primarily consisting of methotrexate.
Major finding: Compared with other DMARDs monotherapy, iguratimod monotherapy or combined therapy was associated with significantly higher American College of Rheumatology 20% response (odds ratio, 1.97; P = .002), lower Disease Activity Score in 28 joints-C-reactive protein (standard mean difference [SMD], −3.49), and a shorter duration of morning stiffness (SMD, −2.06; all P less than .001). The incidence of gastrointestinal events (P = .070), leucopenia (P = .309), transaminase elevation (P = .321), and liver damage (P = .182) was comparable between methotrexate and iguratimod monotherapy.
Study details: This was a meta-analysis of 23 randomized clinical trials involving 2,533 patients with RA.
Disclosures: This study was supported by the Ministry of Science and Technology of China, the Chinese Academy of Medical Sciences, and the Beijing Municipal Science & Technology Commission. The authors declared no conflict of interests.
Source: Hu CJ et al. J Orthop Surg Res. 2021 Jul 16. doi: 10.1186/s13018-021-02603-2.
Key clinical point: Iguratimod as monotherapy or combined therapy was remarkably effective and safe in patients with active rheumatoid arthritis (RA) compared with other disease-modifying antirheumatic drugs (DMARDs) monotherapy, primarily consisting of methotrexate.
Major finding: Compared with other DMARDs monotherapy, iguratimod monotherapy or combined therapy was associated with significantly higher American College of Rheumatology 20% response (odds ratio, 1.97; P = .002), lower Disease Activity Score in 28 joints-C-reactive protein (standard mean difference [SMD], −3.49), and a shorter duration of morning stiffness (SMD, −2.06; all P less than .001). The incidence of gastrointestinal events (P = .070), leucopenia (P = .309), transaminase elevation (P = .321), and liver damage (P = .182) was comparable between methotrexate and iguratimod monotherapy.
Study details: This was a meta-analysis of 23 randomized clinical trials involving 2,533 patients with RA.
Disclosures: This study was supported by the Ministry of Science and Technology of China, the Chinese Academy of Medical Sciences, and the Beijing Municipal Science & Technology Commission. The authors declared no conflict of interests.
Source: Hu CJ et al. J Orthop Surg Res. 2021 Jul 16. doi: 10.1186/s13018-021-02603-2.
Key clinical point: Iguratimod as monotherapy or combined therapy was remarkably effective and safe in patients with active rheumatoid arthritis (RA) compared with other disease-modifying antirheumatic drugs (DMARDs) monotherapy, primarily consisting of methotrexate.
Major finding: Compared with other DMARDs monotherapy, iguratimod monotherapy or combined therapy was associated with significantly higher American College of Rheumatology 20% response (odds ratio, 1.97; P = .002), lower Disease Activity Score in 28 joints-C-reactive protein (standard mean difference [SMD], −3.49), and a shorter duration of morning stiffness (SMD, −2.06; all P less than .001). The incidence of gastrointestinal events (P = .070), leucopenia (P = .309), transaminase elevation (P = .321), and liver damage (P = .182) was comparable between methotrexate and iguratimod monotherapy.
Study details: This was a meta-analysis of 23 randomized clinical trials involving 2,533 patients with RA.
Disclosures: This study was supported by the Ministry of Science and Technology of China, the Chinese Academy of Medical Sciences, and the Beijing Municipal Science & Technology Commission. The authors declared no conflict of interests.
Source: Hu CJ et al. J Orthop Surg Res. 2021 Jul 16. doi: 10.1186/s13018-021-02603-2.
Fostamatinib tied to higher risk for hypertension in RA
Key clinical point: Meta-analysis showed a significant association between the use of fostamatinib and a higher risk for hypertension in patients with rheumatoid arthritis (RA); however, no association was observed with elevated risk for cardiovascular events.
Major findings: Fostamatinib was associated with significantly higher risk for hypertension compared with placebo (relative risk, 3.82; 95% confidence interval [CI], 2.88-5.05); however, it was not associated with the higher risk for all-cause death (Peto odds ratio [pOR], 0.16; 95% CI, 0.02-1.24), major adverse cardiovascular events (pOR, 1.24; 95% CI, 0.26-5.97), and other forms of heart disease (pOR, 1.96; 95% CI, 0.72-5.38).
Study details: This was a meta-analysis of 12 trials including 5,618 patients with RA who were treated with oral fostamatinib.
Disclosures: This study was supported by the Post-doctoral Research and Development Fund of West China Hospital, the Clinical Research Incubation Project of West China Hospital, and the Sichuan Science and Technology Program. The authors declared no conflict of interests.
Source: Chen Y et al. Front Pharmacol. 2021 Jul 19. doi: 10.3389/fphar.2021.632551.
Key clinical point: Meta-analysis showed a significant association between the use of fostamatinib and a higher risk for hypertension in patients with rheumatoid arthritis (RA); however, no association was observed with elevated risk for cardiovascular events.
Major findings: Fostamatinib was associated with significantly higher risk for hypertension compared with placebo (relative risk, 3.82; 95% confidence interval [CI], 2.88-5.05); however, it was not associated with the higher risk for all-cause death (Peto odds ratio [pOR], 0.16; 95% CI, 0.02-1.24), major adverse cardiovascular events (pOR, 1.24; 95% CI, 0.26-5.97), and other forms of heart disease (pOR, 1.96; 95% CI, 0.72-5.38).
Study details: This was a meta-analysis of 12 trials including 5,618 patients with RA who were treated with oral fostamatinib.
Disclosures: This study was supported by the Post-doctoral Research and Development Fund of West China Hospital, the Clinical Research Incubation Project of West China Hospital, and the Sichuan Science and Technology Program. The authors declared no conflict of interests.
Source: Chen Y et al. Front Pharmacol. 2021 Jul 19. doi: 10.3389/fphar.2021.632551.
Key clinical point: Meta-analysis showed a significant association between the use of fostamatinib and a higher risk for hypertension in patients with rheumatoid arthritis (RA); however, no association was observed with elevated risk for cardiovascular events.
Major findings: Fostamatinib was associated with significantly higher risk for hypertension compared with placebo (relative risk, 3.82; 95% confidence interval [CI], 2.88-5.05); however, it was not associated with the higher risk for all-cause death (Peto odds ratio [pOR], 0.16; 95% CI, 0.02-1.24), major adverse cardiovascular events (pOR, 1.24; 95% CI, 0.26-5.97), and other forms of heart disease (pOR, 1.96; 95% CI, 0.72-5.38).
Study details: This was a meta-analysis of 12 trials including 5,618 patients with RA who were treated with oral fostamatinib.
Disclosures: This study was supported by the Post-doctoral Research and Development Fund of West China Hospital, the Clinical Research Incubation Project of West China Hospital, and the Sichuan Science and Technology Program. The authors declared no conflict of interests.
Source: Chen Y et al. Front Pharmacol. 2021 Jul 19. doi: 10.3389/fphar.2021.632551.
Fibromyalgianess tied to persistent glucocorticoid use in RA
Key clinical point: Patients with rheumatoid arthritis (RA) with high baseline fibromyalgianess were more likely to use glucocorticoid persistently at 3 months follow-up, regardless of inflammatory activity.
Major finding: After adjusting for potential confounders including noninflammatory and inflammatory factors, patients with high or very high vs low fibromyalgianess were significantly more likely to be taking prednisone (adjusted odds ratio, 4.99; 95% confidence interval, 1.20-20.73) at 3-month follow-up.
Study details: Findings are from an analysis of 97 patients with active RA from the prospective Central Pain in RA (CPIRA) cohort, who were on oral prednisone for 3 months after starting a new disease-modifying antirheumatic drug.
Disclosures: The CPIRA study was funded by the National Institute of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. YC Lee, MB Bolster, and DJ Clauw reported receiving consulting fees, speaking fees, honoraria, and/or stock ownership from various sources. All the other authors declared no conflict of interests.
Source: Wallace BI et al. Rheumatology (Oxford). 2021 Jul 22. doi: 10.1093/rheumatology/keab583.
Key clinical point: Patients with rheumatoid arthritis (RA) with high baseline fibromyalgianess were more likely to use glucocorticoid persistently at 3 months follow-up, regardless of inflammatory activity.
Major finding: After adjusting for potential confounders including noninflammatory and inflammatory factors, patients with high or very high vs low fibromyalgianess were significantly more likely to be taking prednisone (adjusted odds ratio, 4.99; 95% confidence interval, 1.20-20.73) at 3-month follow-up.
Study details: Findings are from an analysis of 97 patients with active RA from the prospective Central Pain in RA (CPIRA) cohort, who were on oral prednisone for 3 months after starting a new disease-modifying antirheumatic drug.
Disclosures: The CPIRA study was funded by the National Institute of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. YC Lee, MB Bolster, and DJ Clauw reported receiving consulting fees, speaking fees, honoraria, and/or stock ownership from various sources. All the other authors declared no conflict of interests.
Source: Wallace BI et al. Rheumatology (Oxford). 2021 Jul 22. doi: 10.1093/rheumatology/keab583.
Key clinical point: Patients with rheumatoid arthritis (RA) with high baseline fibromyalgianess were more likely to use glucocorticoid persistently at 3 months follow-up, regardless of inflammatory activity.
Major finding: After adjusting for potential confounders including noninflammatory and inflammatory factors, patients with high or very high vs low fibromyalgianess were significantly more likely to be taking prednisone (adjusted odds ratio, 4.99; 95% confidence interval, 1.20-20.73) at 3-month follow-up.
Study details: Findings are from an analysis of 97 patients with active RA from the prospective Central Pain in RA (CPIRA) cohort, who were on oral prednisone for 3 months after starting a new disease-modifying antirheumatic drug.
Disclosures: The CPIRA study was funded by the National Institute of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. YC Lee, MB Bolster, and DJ Clauw reported receiving consulting fees, speaking fees, honoraria, and/or stock ownership from various sources. All the other authors declared no conflict of interests.
Source: Wallace BI et al. Rheumatology (Oxford). 2021 Jul 22. doi: 10.1093/rheumatology/keab583.
No evidence of increased cancer incidence with long-term TNFi use in RA
Key clinical point: Long-term use of tumor necrosis factor inhibitors (TNFis) for rheumatoid arthritis (RA) in clinical practice was not associated with an increased incidence of overall cancer. However, a higher risk of developing urinary tract cancer was observed for TNFi, rituximab, and abatacept.
Major finding: Compared with biologic and targeted synthetic disease-modifying antirheumatic drug (b/tsDMARD)-naive patients, the relative risk for overall cancer was not higher among those treated with TNFis for 10 or more years (adjusted hazard ratio [aHR], 1.0; 95% confidence interval [CI], 0.8-1.2). However, the risk for urinary tract cancer was significantly higher with TNFi (aHR, 1.5; 95% CI, 1.1-1.9), rituximab (aHR, 2.1; 95% CI, 1.3-3.7), and abatacept (aHR, 2.3; 95% CI, 1.3-3.9).
Study details: This was an observational cohort study that included patients with RA (n=69,308) who received TNFis or other b/tsDMARDs, b/tsDMARDs-naïve patients with RA (n=56,233), and matched general population referents (n=109,532).
Disclosures: This study was funded by the Karolinska Institute Region Stockholm. J Askling declared research agreements with various sources. The other authors declared no conflict of interests.
Source: Huss V et al. Rheumatology (Oxford). 2021 Jul 29. doi: 10.1093/rheumatology/keab570.
Key clinical point: Long-term use of tumor necrosis factor inhibitors (TNFis) for rheumatoid arthritis (RA) in clinical practice was not associated with an increased incidence of overall cancer. However, a higher risk of developing urinary tract cancer was observed for TNFi, rituximab, and abatacept.
Major finding: Compared with biologic and targeted synthetic disease-modifying antirheumatic drug (b/tsDMARD)-naive patients, the relative risk for overall cancer was not higher among those treated with TNFis for 10 or more years (adjusted hazard ratio [aHR], 1.0; 95% confidence interval [CI], 0.8-1.2). However, the risk for urinary tract cancer was significantly higher with TNFi (aHR, 1.5; 95% CI, 1.1-1.9), rituximab (aHR, 2.1; 95% CI, 1.3-3.7), and abatacept (aHR, 2.3; 95% CI, 1.3-3.9).
Study details: This was an observational cohort study that included patients with RA (n=69,308) who received TNFis or other b/tsDMARDs, b/tsDMARDs-naïve patients with RA (n=56,233), and matched general population referents (n=109,532).
Disclosures: This study was funded by the Karolinska Institute Region Stockholm. J Askling declared research agreements with various sources. The other authors declared no conflict of interests.
Source: Huss V et al. Rheumatology (Oxford). 2021 Jul 29. doi: 10.1093/rheumatology/keab570.
Key clinical point: Long-term use of tumor necrosis factor inhibitors (TNFis) for rheumatoid arthritis (RA) in clinical practice was not associated with an increased incidence of overall cancer. However, a higher risk of developing urinary tract cancer was observed for TNFi, rituximab, and abatacept.
Major finding: Compared with biologic and targeted synthetic disease-modifying antirheumatic drug (b/tsDMARD)-naive patients, the relative risk for overall cancer was not higher among those treated with TNFis for 10 or more years (adjusted hazard ratio [aHR], 1.0; 95% confidence interval [CI], 0.8-1.2). However, the risk for urinary tract cancer was significantly higher with TNFi (aHR, 1.5; 95% CI, 1.1-1.9), rituximab (aHR, 2.1; 95% CI, 1.3-3.7), and abatacept (aHR, 2.3; 95% CI, 1.3-3.9).
Study details: This was an observational cohort study that included patients with RA (n=69,308) who received TNFis or other b/tsDMARDs, b/tsDMARDs-naïve patients with RA (n=56,233), and matched general population referents (n=109,532).
Disclosures: This study was funded by the Karolinska Institute Region Stockholm. J Askling declared research agreements with various sources. The other authors declared no conflict of interests.
Source: Huss V et al. Rheumatology (Oxford). 2021 Jul 29. doi: 10.1093/rheumatology/keab570.