User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'main-prefix')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
Reimagining psychiatric assessment and interventions as procedures
Many psychiatric physicians lament the dearth of procedures in psychiatry compared to other medical specialties such as surgery, cardiology, gastroenterology, or radiology. The few procedures in psychiatry include electroconvulsive therapy (ECT), repetitive transcranial magnetic stimulation, and vagus nerve stimulation, which are restricted to a small number of sites and not available for most psychiatric practitioners. This lack of tangible/physical procedures should not be surprising because psychiatry deals with disorders of the mind, which are invisible.
However, when one closely examines what psychiatrists do in daily practice to heal our patients, most of what we do actually qualifies as “procedures” although no hardware, machines, or gadgets are involved. Treating psychiatric brain disorders (aka mental illness) requires exquisite skills and expertise, just like medical specialties that use machines to measure or treat various body organs.
It’s time to relabel psychiatric interventions as procedures designed to improve anomalous thoughts, affect, emotions, cognition, and behavior. After giving it some thought (and with a bit of tongue in cheek), I came up with the following list of “psychiatric procedures”:
- Psychosocial exploratory laparotomy: The comprehensive psychiatric assessment and mental status exam.
- Chemotherapy: Oral or injective pharmacotherapeutic intervention.
- Psychoplastic repair: Neuroplasticity, including neurogenesis, synaptogenesis, and dendritic spine regeneration, have been shown to be associated with both psychotherapy and psychotropic medications.1,2
- Suicidectomy: Extracting the lethal urge to die by suicide.
- Anger debridement: Removing the irritability and destructive anger outbursts frequently associated with various psychopathologies.
- Anxiety ablation: Eliminating the noxious emotional state of anxiety and frightening panic attacks.
- Empathy infusion: Enabling patients to become more understanding of other people and bolstering their impaired “theory of mind.”
- Personality transplant: Replacing a maladaptive personality with a healthier one (eg, using dialectical behavior therapy for borderline personality disorder).
- Cognitive LASIK: To improve insight, analogous to how ophthalmologic LASIK improves sight.
- Mental embolectomy: Removing a blockage to repair rigid attitudes and develop “open-mindedness.”
- Behavioral dilation and curettage (D&C): To rid patients of negative attributes such as impulsivity or reckless behavior.
- Psychotherapeutic anesthesia: Numbing emotional pain or severe grief reaction.
- Social anastomosis: Helping patients who are schizoid or isolative via group therapy, an effective interpersonal and social procedure.
- Psychotherapeutic stent: To open the vessels of narrow-mindedness.
- Cortico-psychological resuscitation (CPR): For patients experiencing stress-induced behavioral arrhythmias or emotional infarction.
- Immunotherapy: Using various neuroprotective psychotropic medications with anti-inflammatory properties or employing evidence-based psychotherapy such as cognitive-behavior therapy (aka neuropsychotherapy), which have been shown to reduce inflammatory biomarkers such as C-reactive protein and cytokines.3
- Psychotherapy: A neuromodulation procedure for a variety of psychiatric disorders.4
- Neurobiological facelift: It is well established that neurogenesis, synaptogenesis, and dendritic spine sprouting are significantly increased with both neuroprotective psychotropic medications (antidepressants, lithium, valproate, and second-generationantipsychotics5) as well as with psychotherapy. There is growing evidence of “premature brain aging” in schizophrenia, bipolar disorder, and depression, with shrinkage in the volume of the cortex and subcortical regions, especially the hippocampus. Psychiatric biopsychosocial intervention rebuilds those brain regions by stimulating and replenishing the neuropil and neurogenic regions (dentate gyrus and subventricular zone). This is like performing virtual plastic surgery on a wrinkled brain and its sagging mind. MRI scans before and after ECT show a remarkable ≥10% increase in the volume of the hippocampus and amygdala, which translates to billions of new neurons, glia, and synapses.6
Reinventing psychiatric therapies as procedures may elicit sarcasm from skeptics, but when you think about it, it is justified. Excising depression is like excising a tumor, not with a scalpel, but virtually. Stabilizing the broken brain and mind after a psychotic episode (aka brain attack) is like stabilizing the heart after a myocardial infarction (aka heart attack). Just because the mind is virtual doesn’t mean it is not “real and tangible.” A desktop computer is visible, but the software that brings it to life is invisible. Healing the human mind requires multiple medical interventions by psychiatrists in hospitals and clinics, just like surgeons and endoscopists or cardiologists. Mental health care is as much procedural as other medical and surgical specialties.
One more thing: the validated clinical rating scales for various psychiatric brain disorders (eg, the Positive and Negative Syndrome Scale for schizophrenia, Montgomery-Åsberg Depression Rating Scale for depression, Young Mania Rating Scale for bipolar mania, Hamilton Anxiety Rating Scale for anxiety, Yale-Brown Obsessive Compulsive Scale for obsessive-compulsive disorder) are actual measurement procedures for the severity of the illness, just as a sphygmomanometer measures blood pressure and its improvement with treatment. There are also multiple cognitive test batteries to measure cognitive impairment.7
Finally, unlike psychiatric reimbursement, which is tethered to time, procedures are compensated more generously, irrespective of the time involved. The complexities of diagnosing and treating psychiatric brain disorders that dangerously disrupt thoughts, feelings, behavior, and cognition are just as intricate and demanding as the diagnosis and treatment of general medical and surgical conditions. They should all be equally appreciated as vital life-saving procedures for the human body, brain, and mind.
1. Nasrallah HA, Hopkins T, Pixley SK. Differential effects of antipsychotic and antidepressant drugs on neurogenic regions in rats. Brain Res. 2010;1354:23-29.
2. Tomasino B, Fabbro F. Increases in the right dorsolateral prefrontal cortex and decreases the rostral prefrontal cortex activation after-8 weeks of focused attention based mindfulness meditation. Brain Cogn. 2016;102:46-54.
3. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
4. Nasrallah HA. Optimal psychiatric treatment: Target the brain and avoid the body. Current Psychiatry. 2022;21(12):3-6.
5. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
6. Gryglewski G, Lanzenberger R, Silberbauer LR, et al. Meta-analysis of brain structural changes after electroconvulsive therapy in depression. Brain Stimul. 2021;14(4):927-937.
7. Nasrallah HA. The Cognition Self-Assessment Rating Scale for patients with schizophrenia. Current Psychiatry. 2023;22(3):30-34.
Many psychiatric physicians lament the dearth of procedures in psychiatry compared to other medical specialties such as surgery, cardiology, gastroenterology, or radiology. The few procedures in psychiatry include electroconvulsive therapy (ECT), repetitive transcranial magnetic stimulation, and vagus nerve stimulation, which are restricted to a small number of sites and not available for most psychiatric practitioners. This lack of tangible/physical procedures should not be surprising because psychiatry deals with disorders of the mind, which are invisible.
However, when one closely examines what psychiatrists do in daily practice to heal our patients, most of what we do actually qualifies as “procedures” although no hardware, machines, or gadgets are involved. Treating psychiatric brain disorders (aka mental illness) requires exquisite skills and expertise, just like medical specialties that use machines to measure or treat various body organs.
It’s time to relabel psychiatric interventions as procedures designed to improve anomalous thoughts, affect, emotions, cognition, and behavior. After giving it some thought (and with a bit of tongue in cheek), I came up with the following list of “psychiatric procedures”:
- Psychosocial exploratory laparotomy: The comprehensive psychiatric assessment and mental status exam.
- Chemotherapy: Oral or injective pharmacotherapeutic intervention.
- Psychoplastic repair: Neuroplasticity, including neurogenesis, synaptogenesis, and dendritic spine regeneration, have been shown to be associated with both psychotherapy and psychotropic medications.1,2
- Suicidectomy: Extracting the lethal urge to die by suicide.
- Anger debridement: Removing the irritability and destructive anger outbursts frequently associated with various psychopathologies.
- Anxiety ablation: Eliminating the noxious emotional state of anxiety and frightening panic attacks.
- Empathy infusion: Enabling patients to become more understanding of other people and bolstering their impaired “theory of mind.”
- Personality transplant: Replacing a maladaptive personality with a healthier one (eg, using dialectical behavior therapy for borderline personality disorder).
- Cognitive LASIK: To improve insight, analogous to how ophthalmologic LASIK improves sight.
- Mental embolectomy: Removing a blockage to repair rigid attitudes and develop “open-mindedness.”
- Behavioral dilation and curettage (D&C): To rid patients of negative attributes such as impulsivity or reckless behavior.
- Psychotherapeutic anesthesia: Numbing emotional pain or severe grief reaction.
- Social anastomosis: Helping patients who are schizoid or isolative via group therapy, an effective interpersonal and social procedure.
- Psychotherapeutic stent: To open the vessels of narrow-mindedness.
- Cortico-psychological resuscitation (CPR): For patients experiencing stress-induced behavioral arrhythmias or emotional infarction.
- Immunotherapy: Using various neuroprotective psychotropic medications with anti-inflammatory properties or employing evidence-based psychotherapy such as cognitive-behavior therapy (aka neuropsychotherapy), which have been shown to reduce inflammatory biomarkers such as C-reactive protein and cytokines.3
- Psychotherapy: A neuromodulation procedure for a variety of psychiatric disorders.4
- Neurobiological facelift: It is well established that neurogenesis, synaptogenesis, and dendritic spine sprouting are significantly increased with both neuroprotective psychotropic medications (antidepressants, lithium, valproate, and second-generationantipsychotics5) as well as with psychotherapy. There is growing evidence of “premature brain aging” in schizophrenia, bipolar disorder, and depression, with shrinkage in the volume of the cortex and subcortical regions, especially the hippocampus. Psychiatric biopsychosocial intervention rebuilds those brain regions by stimulating and replenishing the neuropil and neurogenic regions (dentate gyrus and subventricular zone). This is like performing virtual plastic surgery on a wrinkled brain and its sagging mind. MRI scans before and after ECT show a remarkable ≥10% increase in the volume of the hippocampus and amygdala, which translates to billions of new neurons, glia, and synapses.6
Reinventing psychiatric therapies as procedures may elicit sarcasm from skeptics, but when you think about it, it is justified. Excising depression is like excising a tumor, not with a scalpel, but virtually. Stabilizing the broken brain and mind after a psychotic episode (aka brain attack) is like stabilizing the heart after a myocardial infarction (aka heart attack). Just because the mind is virtual doesn’t mean it is not “real and tangible.” A desktop computer is visible, but the software that brings it to life is invisible. Healing the human mind requires multiple medical interventions by psychiatrists in hospitals and clinics, just like surgeons and endoscopists or cardiologists. Mental health care is as much procedural as other medical and surgical specialties.
One more thing: the validated clinical rating scales for various psychiatric brain disorders (eg, the Positive and Negative Syndrome Scale for schizophrenia, Montgomery-Åsberg Depression Rating Scale for depression, Young Mania Rating Scale for bipolar mania, Hamilton Anxiety Rating Scale for anxiety, Yale-Brown Obsessive Compulsive Scale for obsessive-compulsive disorder) are actual measurement procedures for the severity of the illness, just as a sphygmomanometer measures blood pressure and its improvement with treatment. There are also multiple cognitive test batteries to measure cognitive impairment.7
Finally, unlike psychiatric reimbursement, which is tethered to time, procedures are compensated more generously, irrespective of the time involved. The complexities of diagnosing and treating psychiatric brain disorders that dangerously disrupt thoughts, feelings, behavior, and cognition are just as intricate and demanding as the diagnosis and treatment of general medical and surgical conditions. They should all be equally appreciated as vital life-saving procedures for the human body, brain, and mind.
Many psychiatric physicians lament the dearth of procedures in psychiatry compared to other medical specialties such as surgery, cardiology, gastroenterology, or radiology. The few procedures in psychiatry include electroconvulsive therapy (ECT), repetitive transcranial magnetic stimulation, and vagus nerve stimulation, which are restricted to a small number of sites and not available for most psychiatric practitioners. This lack of tangible/physical procedures should not be surprising because psychiatry deals with disorders of the mind, which are invisible.
However, when one closely examines what psychiatrists do in daily practice to heal our patients, most of what we do actually qualifies as “procedures” although no hardware, machines, or gadgets are involved. Treating psychiatric brain disorders (aka mental illness) requires exquisite skills and expertise, just like medical specialties that use machines to measure or treat various body organs.
It’s time to relabel psychiatric interventions as procedures designed to improve anomalous thoughts, affect, emotions, cognition, and behavior. After giving it some thought (and with a bit of tongue in cheek), I came up with the following list of “psychiatric procedures”:
- Psychosocial exploratory laparotomy: The comprehensive psychiatric assessment and mental status exam.
- Chemotherapy: Oral or injective pharmacotherapeutic intervention.
- Psychoplastic repair: Neuroplasticity, including neurogenesis, synaptogenesis, and dendritic spine regeneration, have been shown to be associated with both psychotherapy and psychotropic medications.1,2
- Suicidectomy: Extracting the lethal urge to die by suicide.
- Anger debridement: Removing the irritability and destructive anger outbursts frequently associated with various psychopathologies.
- Anxiety ablation: Eliminating the noxious emotional state of anxiety and frightening panic attacks.
- Empathy infusion: Enabling patients to become more understanding of other people and bolstering their impaired “theory of mind.”
- Personality transplant: Replacing a maladaptive personality with a healthier one (eg, using dialectical behavior therapy for borderline personality disorder).
- Cognitive LASIK: To improve insight, analogous to how ophthalmologic LASIK improves sight.
- Mental embolectomy: Removing a blockage to repair rigid attitudes and develop “open-mindedness.”
- Behavioral dilation and curettage (D&C): To rid patients of negative attributes such as impulsivity or reckless behavior.
- Psychotherapeutic anesthesia: Numbing emotional pain or severe grief reaction.
- Social anastomosis: Helping patients who are schizoid or isolative via group therapy, an effective interpersonal and social procedure.
- Psychotherapeutic stent: To open the vessels of narrow-mindedness.
- Cortico-psychological resuscitation (CPR): For patients experiencing stress-induced behavioral arrhythmias or emotional infarction.
- Immunotherapy: Using various neuroprotective psychotropic medications with anti-inflammatory properties or employing evidence-based psychotherapy such as cognitive-behavior therapy (aka neuropsychotherapy), which have been shown to reduce inflammatory biomarkers such as C-reactive protein and cytokines.3
- Psychotherapy: A neuromodulation procedure for a variety of psychiatric disorders.4
- Neurobiological facelift: It is well established that neurogenesis, synaptogenesis, and dendritic spine sprouting are significantly increased with both neuroprotective psychotropic medications (antidepressants, lithium, valproate, and second-generationantipsychotics5) as well as with psychotherapy. There is growing evidence of “premature brain aging” in schizophrenia, bipolar disorder, and depression, with shrinkage in the volume of the cortex and subcortical regions, especially the hippocampus. Psychiatric biopsychosocial intervention rebuilds those brain regions by stimulating and replenishing the neuropil and neurogenic regions (dentate gyrus and subventricular zone). This is like performing virtual plastic surgery on a wrinkled brain and its sagging mind. MRI scans before and after ECT show a remarkable ≥10% increase in the volume of the hippocampus and amygdala, which translates to billions of new neurons, glia, and synapses.6
Reinventing psychiatric therapies as procedures may elicit sarcasm from skeptics, but when you think about it, it is justified. Excising depression is like excising a tumor, not with a scalpel, but virtually. Stabilizing the broken brain and mind after a psychotic episode (aka brain attack) is like stabilizing the heart after a myocardial infarction (aka heart attack). Just because the mind is virtual doesn’t mean it is not “real and tangible.” A desktop computer is visible, but the software that brings it to life is invisible. Healing the human mind requires multiple medical interventions by psychiatrists in hospitals and clinics, just like surgeons and endoscopists or cardiologists. Mental health care is as much procedural as other medical and surgical specialties.
One more thing: the validated clinical rating scales for various psychiatric brain disorders (eg, the Positive and Negative Syndrome Scale for schizophrenia, Montgomery-Åsberg Depression Rating Scale for depression, Young Mania Rating Scale for bipolar mania, Hamilton Anxiety Rating Scale for anxiety, Yale-Brown Obsessive Compulsive Scale for obsessive-compulsive disorder) are actual measurement procedures for the severity of the illness, just as a sphygmomanometer measures blood pressure and its improvement with treatment. There are also multiple cognitive test batteries to measure cognitive impairment.7
Finally, unlike psychiatric reimbursement, which is tethered to time, procedures are compensated more generously, irrespective of the time involved. The complexities of diagnosing and treating psychiatric brain disorders that dangerously disrupt thoughts, feelings, behavior, and cognition are just as intricate and demanding as the diagnosis and treatment of general medical and surgical conditions. They should all be equally appreciated as vital life-saving procedures for the human body, brain, and mind.
1. Nasrallah HA, Hopkins T, Pixley SK. Differential effects of antipsychotic and antidepressant drugs on neurogenic regions in rats. Brain Res. 2010;1354:23-29.
2. Tomasino B, Fabbro F. Increases in the right dorsolateral prefrontal cortex and decreases the rostral prefrontal cortex activation after-8 weeks of focused attention based mindfulness meditation. Brain Cogn. 2016;102:46-54.
3. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
4. Nasrallah HA. Optimal psychiatric treatment: Target the brain and avoid the body. Current Psychiatry. 2022;21(12):3-6.
5. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
6. Gryglewski G, Lanzenberger R, Silberbauer LR, et al. Meta-analysis of brain structural changes after electroconvulsive therapy in depression. Brain Stimul. 2021;14(4):927-937.
7. Nasrallah HA. The Cognition Self-Assessment Rating Scale for patients with schizophrenia. Current Psychiatry. 2023;22(3):30-34.
1. Nasrallah HA, Hopkins T, Pixley SK. Differential effects of antipsychotic and antidepressant drugs on neurogenic regions in rats. Brain Res. 2010;1354:23-29.
2. Tomasino B, Fabbro F. Increases in the right dorsolateral prefrontal cortex and decreases the rostral prefrontal cortex activation after-8 weeks of focused attention based mindfulness meditation. Brain Cogn. 2016;102:46-54.
3. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
4. Nasrallah HA. Optimal psychiatric treatment: Target the brain and avoid the body. Current Psychiatry. 2022;21(12):3-6.
5. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7.
6. Gryglewski G, Lanzenberger R, Silberbauer LR, et al. Meta-analysis of brain structural changes after electroconvulsive therapy in depression. Brain Stimul. 2021;14(4):927-937.
7. Nasrallah HA. The Cognition Self-Assessment Rating Scale for patients with schizophrenia. Current Psychiatry. 2023;22(3):30-34.
Psychoactive supplements: What to tell patients
Mr. D, age 41, presents to the emergency department (ED) with altered mental status and suspected intoxication. His medical history includes alcohol use disorder and spinal injury. Upon initial examination, he is confused, disorganized, and agitated. He receives IM lorazepam 4 mg to manage his agitation. His laboratory workup includes a negative screening for blood alcohol, slightly elevated creatine kinase, and urine toxicology positive for barbiturates and opioids. During re-evaluation by the consulting psychiatrist the following morning, Mr. D is alert, oriented, and calm with an organized thought process. He does not appear to be in withdrawal from any substances and tells the psychiatrist that he takes butalbital/acetaminophen/caffeine/codeine as needed for migraines. Mr. D says that 3 days before he came to the ED, he also began taking a supplement called phenibut that he purchased online for “well-being and sleep.”
Natural substances have been used throughout history as medicinal agents, sacred substances in religious rituals, and for recreational purposes.1 Supplement use in the United States is prevalent, with 57.6% of adults age ≥20 reporting supplement use in the past 30 days.2 Between 2000 and 2017, US poison control centers recorded a 74.1% increase in calls involving exposure to natural psychoactive substances, mostly driven by cases involving marijuana in adults and adolescents.3 Like synthetic drugs, herbal supplements may have psychoactive properties, including sedative, stimulant, psychedelic, euphoric, or anticholinergic effects. The variety and unregulated nature of supplements makes managing patients who use supplements particularly challenging.
Why patients use supplements
People may use supplements to treat or prevent vitamin deficiencies (eg, vitamin D, iron, calcium). Other reasons may include for promoting wellness in various disease states, for weight loss, for recreational use or misuse, or for overall well-being. In the mental health realm, patients report using supplements to treat depression, anxiety, insomnia, memory, or for vague indications such as “mood support.”4,5
Patients may view supplements as appealing alternatives to prescription medications because they are widely accessible, may be purchased over-the-counter, are inexpensive, and represent a “natural” treatment option.6 For these reasons, they may also falsely perceive supplements as categorically safe.1 People with psychiatric diagnoses may choose such alternative treatments due to a history of adverse effects or treatment failure with traditional psychiatric medications, mistrust of the health care or pharmaceutical industry, or based on the recommendations of others.7
Regulation, safety, and efficacy of dietary supplements
In the US, dietary supplements are regulated more like food products than medications. Under the Dietary Supplement Health and Education Act of 1994, the FDA regulates the quality, safety, and labeling of supplements using Current Good Manufacturing Practice regulations.8 The Federal Trade Commission monitors advertisements and marketing. Despite some regulations, dietary supplements may be adulterated or contaminated, contain unknown or toxic ingredients, have inconsistent potencies, or be sold at toxic doses.9 Importantly, supplements are not required to be evaluated for clinical efficacy. As a result, it is not known if most supplements are effective in treating the conditions for which they are promoted, mainly due to a lack of financial incentive for manufacturers to conduct large, high-quality trials.5
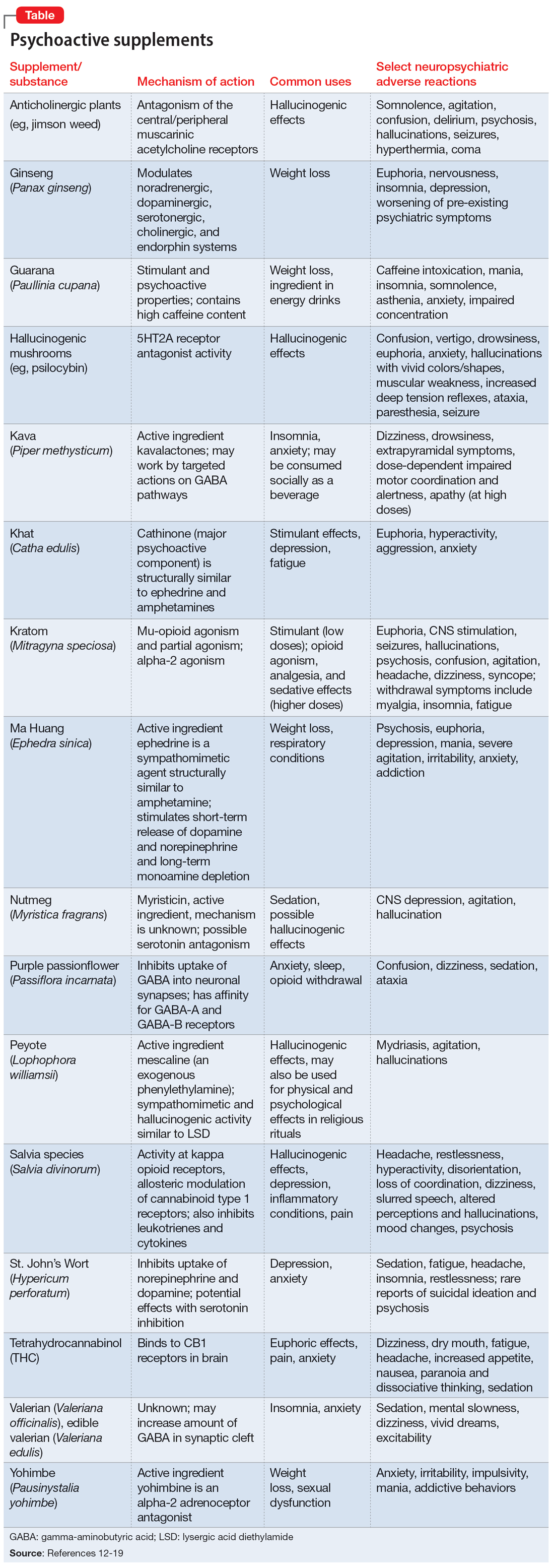
Further complicating matters is the inconsistent labeling of supplements or similar products that are easily obtainable via the internet. These products might be marketed as nutritional supplements or nootropics, which often are referred to as “cognitive enhancers” or “smart drugs.” New psychoactive substances (NPS) are drugs of misuse or abuse developed to imitate illicit drugs or controlled drug substances.10 They are sometimes referred to as “herbal highs” or “legal highs.”11 Supplements may also be labeled as performance- or image-enhancing agents and may include medications marketed to promote weight loss. This includes herbal substances (Table12-19) and medications associated with neuropsychiatric adverse effects that may be easily accessible online without a prescription.12,20
The growing popularity of the internet and social media plays an important role in the availability of supplements and nonregulated substances and may contribute to misleading claims of efficacy and safety. While many herbal supplements are available in pharmacies or supplement stores, NPS are usually sold through anonymous, low-risk means either via traditional online vendors or the deep web (parts of the internet that are not indexed via search engines). Strategies to circumvent regulation and legislative control include labeling NPS as research chemicals, fertilizers, incense, bath salts, or other identifiers and marketing them as “not for human consumption.”21 Manufacturers frequently change the chemical structures of NPS, which allows these products to exist within a legal gray area due to the lag time between when a new compound hits the market and when it is categorized as a regulated substance.10
Continue to: Another category of "supplements"...
Another category of “supplements” includes medications that are not FDA-approved but are approved for therapeutic use in other countries and readily available in the US via online sources. Such medications include phenibut, a glutamic acid derivative that functions as a gamma-aminobutyric acid-B receptor agonist in the brain, spinal cord, and autonomic nervous system. Phenibut was developed in the Soviet Union in the 1960s, and outside of the US it is prescribed for anxiolysis and other psychiatric indications.22 In the US, phenibut may be used as a nootropic or as a dietary supplement to treat anxiety, sleep problems, and other psychiatric disorders.22 It may also be used recreationally to induce euphoria. Chronic phenibut use results in tolerance and abrupt discontinuation may mimic benzodiazepine withdrawal symptoms.13,22
Educating patients about supplements
One of the most critical steps in assessing a patient’s supplement use is to directly ask them about their use of herbal or over-the-counter products. Research has consistently shown that patients are unlikely to disclose supplement use unless they are specifically asked.23,24
Additional strategies include25,26:
- Approach patients without judgment; ask open-ended questions to determine their motivations for using supplements.
- Explain the difference between supplements medically necessary to treat vitamin deficiencies (eg, vitamin D, calcium, magnesium) and those without robust clinical evidence.
- Counsel patients that many supplements with psychoactive properties, if indicated, are generally meant to be used short-term and not as substitutes for prescription medications.
- Educate patients that supplements have limited evidence regarding their safety and efficacy, but like prescription medications, supplements may cause organ damage, adverse effects, and drug-drug interactions.
- Remind patients that commonly used nutritional supplements/dietary aids, including protein or workout supplements, may contain potentially harmful ingredients.
- Utilize evidence-based resources such as the Natural Medicines Comprehensive Database14 or the National Center for Complementary and Integrative Health (https://www.nccih.nih.gov) to review levels of evidence and educate patients.
- When toxicity or withdrawal is suspected, reach out to local poison control centers for guidance.
- For a patient with a potential supplement-related substance use disorder, urine drug screens may be of limited utility and evidence is often sparse; clinicians may need to rely on primary literature such as case reports to guide management.
- If patients wish to continue taking a supplement, recommend they purchase supplements from manufacturers that have achieved the US Pharmacopeia (USP) verification mark. Products with the USP mark undergo quality assurance measures to ensure the product contains the ingredients listed on the label in the declared potency and amounts, does not contain harmful levels of contaminants, will be metabolized in the body within a specified amount of time, and has been produced in keeping with FDA Current Good Manufacturing Practice regulations.
CASE CONTINUED
In the ED, the consulting psychiatry team discusses Mr. D’s use of phenibut with him, and asks if he uses any additional supplements or nonprescription medications. Mr. D discloses he has been anxious and having trouble sleeping, and a friend recommended phenibut as a safe, natural alternative to medication. The team explains to Mr. D that phenibut’s efficacy has not been studied in the US and that based on available evidence, it is likely unsafe. It may have serious adverse effects, drug-drug interactions, and is potentially addictive.
Mr. D says he was unaware of these risks and agrees to stop taking phenibut. The treatment team discharges him from the ED with a referral for outpatient psychiatric services to address his anxiety and insomnia.
Related Resources
- Tillman B. The hidden dangers of supplements: a case of substance-induced psychosis. Current Psychiatry. 2020; 19(7):e7-e8. doi:10.12788/cp.0018
- McQueen CE. Herb–drug interactions: caution patients when changing supplements. Current Psychiatry. 2017; 16(6):38-41.
Drug Brand Names
Butalbital/acetaminophen/caffeine/codeine • Fioricet with Codeine
1. Graziano S, Orsolini L, Rotolo MC, et al. Herbal highs: review on psychoactive effects and neuropharmacology. Curr Neuropharmacol. 2017;15(5):750-761.
2. Mishra S, Stierman B, Gahche JJ, et al. Dietary supplement use among adults: United States, 2017-2018. NCHS Data Brief. 2021;(399):1-8.
3. O’Neill-Dee C, Spiller HA, Casavant MJ, et al. Natural psychoactive substance-related exposures reported to United States poison control centers, 2000-2017. Clin Toxicol (Phila). 2020;58(8):813-820.
4. Gray DC, Rutledge CM. Herbal supplements in primary care: patient perceptions, motivations, and effects on use. Holist Nurs Pract. 2013;27(1):6-12.
5. Wu K, Messamore E. Reimagining roles of dietary supplements in psychiatric care. AMA J Ethics. 2022;24(5):E437-E442.
6. Snyder FJ, Dundas ML, Kirkpatrick C, et al. Use and safety perceptions regarding herbal supplements: a study of older persons in southeast Idaho. J Nutr Elder. 2009;28(1):81-95.
7. Schulz P, Hede V. Alternative and complementary approaches in psychiatry: beliefs versus evidence. Dialogues Clin Neurosci. 2018;20(3):207-214.
8. Dietary Supplement Health and Education Act of 1994, Pub L 103-417, 103rd Cong (1993-1994).
9. Starr RR. Too little, too late: ineffective regulation of dietary supplements in the United States. Am J Public Health. 2015;105(3):478-485.
10. New psychoactive substances. Alcohol and Drug Foundation. November 10, 2021. Updated November 28, 2022. Accessed January 25, 2023. https://adf.org.au/drug-facts/new-psychoactive-substances/
11. Shafi A, Berry AJ, Sumnall H, et al. New psychoactive substances: a review and updates. Ther Adv Psychopharmacol. 2020;10:2045125320967197.
12. Bersani FS, Coviello M, Imperatori C, et al. Adverse psychiatric effects associated with herbal weight-loss products. Biomed Res Int. 2015;2015:120679.
13. IBM Micromedex POISINDEX® System. IBM Watson Health. Accessed October 3, 2022. https://www.micromedexsolutions.com
14. Natural Medicines Comprehensive Database. Therapeutic Research Center. Accessed October 3, 2022. https://naturalmedicines.therapeuticresearch.com
15. Savage KM, Stough CK, Byrne GJ, et al. Kava for the treatment of generalised anxiety disorder (K-GAD): study protocol for a randomised controlled trial. Trials. 2015;16:493.
16. Swogger MT, Smith KE, Garcia-Romeu A, et al. Understanding kratom use: a guide for healthcare providers. Front Pharmacol. 2022;13:801855.
17. Modabbernia A, Akhondzadeh S. Saffron, passionflower, valerian and sage for mental health. Psychiatr Clin North Am. 2013;36(1):85-91.
18. Coffeen U, Pellicer F. Salvia divinorum: from recreational hallucinogenic use to analgesic and anti-inflammatory action. J Pain Res. 2019;12:1069-1076.
19. National Institutes of Health, Office of Dietary Supplements. Valerian Fact Sheet for Health Professionals. Updated March 15, 2013. Accessed January 25, 2023. https://ods.od.nih.gov/factsheets/Valerian-HealthProfessional
20. An H, Sohn H, Chung S. Phentermine, sibutramine and affective disorders. Clin Psychopharmacol Neurosci. 2013;11(1):7-12.
21. Miliano C, Margiani G, Fattore L, et al. Sales and advertising channels of new psychoactive substances (NPS): internet, social networks, and smartphone apps. Brain Sci. 2018;8(7):123.
22. Hardman MI, Sprung J, Weingarten TN. Acute phenibut withdrawal: a comprehensive literature review and illustrative case report. Bosn J Basic Med Sci. 2019;19(2):125-129.
23. Guzman JR, Paterniti DA, Liu Y, et al. Factors related to disclosure and nondisclosure of dietary supplements in primary care, integrative medicine, and naturopathic medicine. J Fam Med Dis Prev. 2019;5(4):10.23937/2469-5793/1510109.
24. Foley H, Steel A, Cramer H, et al. Disclosure of complementary medicine use to medical providers: a systematic review and meta-analysis. Sci Rep. 2019;9(1):1573.
25. Aldridge Young C. ‘No miracle cures’: counseling patients about dietary supplements. Pharmacy Today. 2014;February:35.
26. United States Pharmacopeia. USP Verified Mark. Accessed January 25, 2023. https://www.usp.org/verification-services/verified-mark
Mr. D, age 41, presents to the emergency department (ED) with altered mental status and suspected intoxication. His medical history includes alcohol use disorder and spinal injury. Upon initial examination, he is confused, disorganized, and agitated. He receives IM lorazepam 4 mg to manage his agitation. His laboratory workup includes a negative screening for blood alcohol, slightly elevated creatine kinase, and urine toxicology positive for barbiturates and opioids. During re-evaluation by the consulting psychiatrist the following morning, Mr. D is alert, oriented, and calm with an organized thought process. He does not appear to be in withdrawal from any substances and tells the psychiatrist that he takes butalbital/acetaminophen/caffeine/codeine as needed for migraines. Mr. D says that 3 days before he came to the ED, he also began taking a supplement called phenibut that he purchased online for “well-being and sleep.”
Natural substances have been used throughout history as medicinal agents, sacred substances in religious rituals, and for recreational purposes.1 Supplement use in the United States is prevalent, with 57.6% of adults age ≥20 reporting supplement use in the past 30 days.2 Between 2000 and 2017, US poison control centers recorded a 74.1% increase in calls involving exposure to natural psychoactive substances, mostly driven by cases involving marijuana in adults and adolescents.3 Like synthetic drugs, herbal supplements may have psychoactive properties, including sedative, stimulant, psychedelic, euphoric, or anticholinergic effects. The variety and unregulated nature of supplements makes managing patients who use supplements particularly challenging.
Why patients use supplements
People may use supplements to treat or prevent vitamin deficiencies (eg, vitamin D, iron, calcium). Other reasons may include for promoting wellness in various disease states, for weight loss, for recreational use or misuse, or for overall well-being. In the mental health realm, patients report using supplements to treat depression, anxiety, insomnia, memory, or for vague indications such as “mood support.”4,5
Patients may view supplements as appealing alternatives to prescription medications because they are widely accessible, may be purchased over-the-counter, are inexpensive, and represent a “natural” treatment option.6 For these reasons, they may also falsely perceive supplements as categorically safe.1 People with psychiatric diagnoses may choose such alternative treatments due to a history of adverse effects or treatment failure with traditional psychiatric medications, mistrust of the health care or pharmaceutical industry, or based on the recommendations of others.7
Regulation, safety, and efficacy of dietary supplements
In the US, dietary supplements are regulated more like food products than medications. Under the Dietary Supplement Health and Education Act of 1994, the FDA regulates the quality, safety, and labeling of supplements using Current Good Manufacturing Practice regulations.8 The Federal Trade Commission monitors advertisements and marketing. Despite some regulations, dietary supplements may be adulterated or contaminated, contain unknown or toxic ingredients, have inconsistent potencies, or be sold at toxic doses.9 Importantly, supplements are not required to be evaluated for clinical efficacy. As a result, it is not known if most supplements are effective in treating the conditions for which they are promoted, mainly due to a lack of financial incentive for manufacturers to conduct large, high-quality trials.5
Further complicating matters is the inconsistent labeling of supplements or similar products that are easily obtainable via the internet. These products might be marketed as nutritional supplements or nootropics, which often are referred to as “cognitive enhancers” or “smart drugs.” New psychoactive substances (NPS) are drugs of misuse or abuse developed to imitate illicit drugs or controlled drug substances.10 They are sometimes referred to as “herbal highs” or “legal highs.”11 Supplements may also be labeled as performance- or image-enhancing agents and may include medications marketed to promote weight loss. This includes herbal substances (Table12-19) and medications associated with neuropsychiatric adverse effects that may be easily accessible online without a prescription.12,20

The growing popularity of the internet and social media plays an important role in the availability of supplements and nonregulated substances and may contribute to misleading claims of efficacy and safety. While many herbal supplements are available in pharmacies or supplement stores, NPS are usually sold through anonymous, low-risk means either via traditional online vendors or the deep web (parts of the internet that are not indexed via search engines). Strategies to circumvent regulation and legislative control include labeling NPS as research chemicals, fertilizers, incense, bath salts, or other identifiers and marketing them as “not for human consumption.”21 Manufacturers frequently change the chemical structures of NPS, which allows these products to exist within a legal gray area due to the lag time between when a new compound hits the market and when it is categorized as a regulated substance.10
Continue to: Another category of "supplements"...
Another category of “supplements” includes medications that are not FDA-approved but are approved for therapeutic use in other countries and readily available in the US via online sources. Such medications include phenibut, a glutamic acid derivative that functions as a gamma-aminobutyric acid-B receptor agonist in the brain, spinal cord, and autonomic nervous system. Phenibut was developed in the Soviet Union in the 1960s, and outside of the US it is prescribed for anxiolysis and other psychiatric indications.22 In the US, phenibut may be used as a nootropic or as a dietary supplement to treat anxiety, sleep problems, and other psychiatric disorders.22 It may also be used recreationally to induce euphoria. Chronic phenibut use results in tolerance and abrupt discontinuation may mimic benzodiazepine withdrawal symptoms.13,22
Educating patients about supplements
One of the most critical steps in assessing a patient’s supplement use is to directly ask them about their use of herbal or over-the-counter products. Research has consistently shown that patients are unlikely to disclose supplement use unless they are specifically asked.23,24
Additional strategies include25,26:
- Approach patients without judgment; ask open-ended questions to determine their motivations for using supplements.
- Explain the difference between supplements medically necessary to treat vitamin deficiencies (eg, vitamin D, calcium, magnesium) and those without robust clinical evidence.
- Counsel patients that many supplements with psychoactive properties, if indicated, are generally meant to be used short-term and not as substitutes for prescription medications.
- Educate patients that supplements have limited evidence regarding their safety and efficacy, but like prescription medications, supplements may cause organ damage, adverse effects, and drug-drug interactions.
- Remind patients that commonly used nutritional supplements/dietary aids, including protein or workout supplements, may contain potentially harmful ingredients.
- Utilize evidence-based resources such as the Natural Medicines Comprehensive Database14 or the National Center for Complementary and Integrative Health (https://www.nccih.nih.gov) to review levels of evidence and educate patients.
- When toxicity or withdrawal is suspected, reach out to local poison control centers for guidance.
- For a patient with a potential supplement-related substance use disorder, urine drug screens may be of limited utility and evidence is often sparse; clinicians may need to rely on primary literature such as case reports to guide management.
- If patients wish to continue taking a supplement, recommend they purchase supplements from manufacturers that have achieved the US Pharmacopeia (USP) verification mark. Products with the USP mark undergo quality assurance measures to ensure the product contains the ingredients listed on the label in the declared potency and amounts, does not contain harmful levels of contaminants, will be metabolized in the body within a specified amount of time, and has been produced in keeping with FDA Current Good Manufacturing Practice regulations.
CASE CONTINUED
In the ED, the consulting psychiatry team discusses Mr. D’s use of phenibut with him, and asks if he uses any additional supplements or nonprescription medications. Mr. D discloses he has been anxious and having trouble sleeping, and a friend recommended phenibut as a safe, natural alternative to medication. The team explains to Mr. D that phenibut’s efficacy has not been studied in the US and that based on available evidence, it is likely unsafe. It may have serious adverse effects, drug-drug interactions, and is potentially addictive.
Mr. D says he was unaware of these risks and agrees to stop taking phenibut. The treatment team discharges him from the ED with a referral for outpatient psychiatric services to address his anxiety and insomnia.
Related Resources
- Tillman B. The hidden dangers of supplements: a case of substance-induced psychosis. Current Psychiatry. 2020; 19(7):e7-e8. doi:10.12788/cp.0018
- McQueen CE. Herb–drug interactions: caution patients when changing supplements. Current Psychiatry. 2017; 16(6):38-41.
Drug Brand Names
Butalbital/acetaminophen/caffeine/codeine • Fioricet with Codeine
Mr. D, age 41, presents to the emergency department (ED) with altered mental status and suspected intoxication. His medical history includes alcohol use disorder and spinal injury. Upon initial examination, he is confused, disorganized, and agitated. He receives IM lorazepam 4 mg to manage his agitation. His laboratory workup includes a negative screening for blood alcohol, slightly elevated creatine kinase, and urine toxicology positive for barbiturates and opioids. During re-evaluation by the consulting psychiatrist the following morning, Mr. D is alert, oriented, and calm with an organized thought process. He does not appear to be in withdrawal from any substances and tells the psychiatrist that he takes butalbital/acetaminophen/caffeine/codeine as needed for migraines. Mr. D says that 3 days before he came to the ED, he also began taking a supplement called phenibut that he purchased online for “well-being and sleep.”
Natural substances have been used throughout history as medicinal agents, sacred substances in religious rituals, and for recreational purposes.1 Supplement use in the United States is prevalent, with 57.6% of adults age ≥20 reporting supplement use in the past 30 days.2 Between 2000 and 2017, US poison control centers recorded a 74.1% increase in calls involving exposure to natural psychoactive substances, mostly driven by cases involving marijuana in adults and adolescents.3 Like synthetic drugs, herbal supplements may have psychoactive properties, including sedative, stimulant, psychedelic, euphoric, or anticholinergic effects. The variety and unregulated nature of supplements makes managing patients who use supplements particularly challenging.
Why patients use supplements
People may use supplements to treat or prevent vitamin deficiencies (eg, vitamin D, iron, calcium). Other reasons may include for promoting wellness in various disease states, for weight loss, for recreational use or misuse, or for overall well-being. In the mental health realm, patients report using supplements to treat depression, anxiety, insomnia, memory, or for vague indications such as “mood support.”4,5
Patients may view supplements as appealing alternatives to prescription medications because they are widely accessible, may be purchased over-the-counter, are inexpensive, and represent a “natural” treatment option.6 For these reasons, they may also falsely perceive supplements as categorically safe.1 People with psychiatric diagnoses may choose such alternative treatments due to a history of adverse effects or treatment failure with traditional psychiatric medications, mistrust of the health care or pharmaceutical industry, or based on the recommendations of others.7
Regulation, safety, and efficacy of dietary supplements
In the US, dietary supplements are regulated more like food products than medications. Under the Dietary Supplement Health and Education Act of 1994, the FDA regulates the quality, safety, and labeling of supplements using Current Good Manufacturing Practice regulations.8 The Federal Trade Commission monitors advertisements and marketing. Despite some regulations, dietary supplements may be adulterated or contaminated, contain unknown or toxic ingredients, have inconsistent potencies, or be sold at toxic doses.9 Importantly, supplements are not required to be evaluated for clinical efficacy. As a result, it is not known if most supplements are effective in treating the conditions for which they are promoted, mainly due to a lack of financial incentive for manufacturers to conduct large, high-quality trials.5
Further complicating matters is the inconsistent labeling of supplements or similar products that are easily obtainable via the internet. These products might be marketed as nutritional supplements or nootropics, which often are referred to as “cognitive enhancers” or “smart drugs.” New psychoactive substances (NPS) are drugs of misuse or abuse developed to imitate illicit drugs or controlled drug substances.10 They are sometimes referred to as “herbal highs” or “legal highs.”11 Supplements may also be labeled as performance- or image-enhancing agents and may include medications marketed to promote weight loss. This includes herbal substances (Table12-19) and medications associated with neuropsychiatric adverse effects that may be easily accessible online without a prescription.12,20

The growing popularity of the internet and social media plays an important role in the availability of supplements and nonregulated substances and may contribute to misleading claims of efficacy and safety. While many herbal supplements are available in pharmacies or supplement stores, NPS are usually sold through anonymous, low-risk means either via traditional online vendors or the deep web (parts of the internet that are not indexed via search engines). Strategies to circumvent regulation and legislative control include labeling NPS as research chemicals, fertilizers, incense, bath salts, or other identifiers and marketing them as “not for human consumption.”21 Manufacturers frequently change the chemical structures of NPS, which allows these products to exist within a legal gray area due to the lag time between when a new compound hits the market and when it is categorized as a regulated substance.10
Continue to: Another category of "supplements"...
Another category of “supplements” includes medications that are not FDA-approved but are approved for therapeutic use in other countries and readily available in the US via online sources. Such medications include phenibut, a glutamic acid derivative that functions as a gamma-aminobutyric acid-B receptor agonist in the brain, spinal cord, and autonomic nervous system. Phenibut was developed in the Soviet Union in the 1960s, and outside of the US it is prescribed for anxiolysis and other psychiatric indications.22 In the US, phenibut may be used as a nootropic or as a dietary supplement to treat anxiety, sleep problems, and other psychiatric disorders.22 It may also be used recreationally to induce euphoria. Chronic phenibut use results in tolerance and abrupt discontinuation may mimic benzodiazepine withdrawal symptoms.13,22
Educating patients about supplements
One of the most critical steps in assessing a patient’s supplement use is to directly ask them about their use of herbal or over-the-counter products. Research has consistently shown that patients are unlikely to disclose supplement use unless they are specifically asked.23,24
Additional strategies include25,26:
- Approach patients without judgment; ask open-ended questions to determine their motivations for using supplements.
- Explain the difference between supplements medically necessary to treat vitamin deficiencies (eg, vitamin D, calcium, magnesium) and those without robust clinical evidence.
- Counsel patients that many supplements with psychoactive properties, if indicated, are generally meant to be used short-term and not as substitutes for prescription medications.
- Educate patients that supplements have limited evidence regarding their safety and efficacy, but like prescription medications, supplements may cause organ damage, adverse effects, and drug-drug interactions.
- Remind patients that commonly used nutritional supplements/dietary aids, including protein or workout supplements, may contain potentially harmful ingredients.
- Utilize evidence-based resources such as the Natural Medicines Comprehensive Database14 or the National Center for Complementary and Integrative Health (https://www.nccih.nih.gov) to review levels of evidence and educate patients.
- When toxicity or withdrawal is suspected, reach out to local poison control centers for guidance.
- For a patient with a potential supplement-related substance use disorder, urine drug screens may be of limited utility and evidence is often sparse; clinicians may need to rely on primary literature such as case reports to guide management.
- If patients wish to continue taking a supplement, recommend they purchase supplements from manufacturers that have achieved the US Pharmacopeia (USP) verification mark. Products with the USP mark undergo quality assurance measures to ensure the product contains the ingredients listed on the label in the declared potency and amounts, does not contain harmful levels of contaminants, will be metabolized in the body within a specified amount of time, and has been produced in keeping with FDA Current Good Manufacturing Practice regulations.
CASE CONTINUED
In the ED, the consulting psychiatry team discusses Mr. D’s use of phenibut with him, and asks if he uses any additional supplements or nonprescription medications. Mr. D discloses he has been anxious and having trouble sleeping, and a friend recommended phenibut as a safe, natural alternative to medication. The team explains to Mr. D that phenibut’s efficacy has not been studied in the US and that based on available evidence, it is likely unsafe. It may have serious adverse effects, drug-drug interactions, and is potentially addictive.
Mr. D says he was unaware of these risks and agrees to stop taking phenibut. The treatment team discharges him from the ED with a referral for outpatient psychiatric services to address his anxiety and insomnia.
Related Resources
- Tillman B. The hidden dangers of supplements: a case of substance-induced psychosis. Current Psychiatry. 2020; 19(7):e7-e8. doi:10.12788/cp.0018
- McQueen CE. Herb–drug interactions: caution patients when changing supplements. Current Psychiatry. 2017; 16(6):38-41.
Drug Brand Names
Butalbital/acetaminophen/caffeine/codeine • Fioricet with Codeine
1. Graziano S, Orsolini L, Rotolo MC, et al. Herbal highs: review on psychoactive effects and neuropharmacology. Curr Neuropharmacol. 2017;15(5):750-761.
2. Mishra S, Stierman B, Gahche JJ, et al. Dietary supplement use among adults: United States, 2017-2018. NCHS Data Brief. 2021;(399):1-8.
3. O’Neill-Dee C, Spiller HA, Casavant MJ, et al. Natural psychoactive substance-related exposures reported to United States poison control centers, 2000-2017. Clin Toxicol (Phila). 2020;58(8):813-820.
4. Gray DC, Rutledge CM. Herbal supplements in primary care: patient perceptions, motivations, and effects on use. Holist Nurs Pract. 2013;27(1):6-12.
5. Wu K, Messamore E. Reimagining roles of dietary supplements in psychiatric care. AMA J Ethics. 2022;24(5):E437-E442.
6. Snyder FJ, Dundas ML, Kirkpatrick C, et al. Use and safety perceptions regarding herbal supplements: a study of older persons in southeast Idaho. J Nutr Elder. 2009;28(1):81-95.
7. Schulz P, Hede V. Alternative and complementary approaches in psychiatry: beliefs versus evidence. Dialogues Clin Neurosci. 2018;20(3):207-214.
8. Dietary Supplement Health and Education Act of 1994, Pub L 103-417, 103rd Cong (1993-1994).
9. Starr RR. Too little, too late: ineffective regulation of dietary supplements in the United States. Am J Public Health. 2015;105(3):478-485.
10. New psychoactive substances. Alcohol and Drug Foundation. November 10, 2021. Updated November 28, 2022. Accessed January 25, 2023. https://adf.org.au/drug-facts/new-psychoactive-substances/
11. Shafi A, Berry AJ, Sumnall H, et al. New psychoactive substances: a review and updates. Ther Adv Psychopharmacol. 2020;10:2045125320967197.
12. Bersani FS, Coviello M, Imperatori C, et al. Adverse psychiatric effects associated with herbal weight-loss products. Biomed Res Int. 2015;2015:120679.
13. IBM Micromedex POISINDEX® System. IBM Watson Health. Accessed October 3, 2022. https://www.micromedexsolutions.com
14. Natural Medicines Comprehensive Database. Therapeutic Research Center. Accessed October 3, 2022. https://naturalmedicines.therapeuticresearch.com
15. Savage KM, Stough CK, Byrne GJ, et al. Kava for the treatment of generalised anxiety disorder (K-GAD): study protocol for a randomised controlled trial. Trials. 2015;16:493.
16. Swogger MT, Smith KE, Garcia-Romeu A, et al. Understanding kratom use: a guide for healthcare providers. Front Pharmacol. 2022;13:801855.
17. Modabbernia A, Akhondzadeh S. Saffron, passionflower, valerian and sage for mental health. Psychiatr Clin North Am. 2013;36(1):85-91.
18. Coffeen U, Pellicer F. Salvia divinorum: from recreational hallucinogenic use to analgesic and anti-inflammatory action. J Pain Res. 2019;12:1069-1076.
19. National Institutes of Health, Office of Dietary Supplements. Valerian Fact Sheet for Health Professionals. Updated March 15, 2013. Accessed January 25, 2023. https://ods.od.nih.gov/factsheets/Valerian-HealthProfessional
20. An H, Sohn H, Chung S. Phentermine, sibutramine and affective disorders. Clin Psychopharmacol Neurosci. 2013;11(1):7-12.
21. Miliano C, Margiani G, Fattore L, et al. Sales and advertising channels of new psychoactive substances (NPS): internet, social networks, and smartphone apps. Brain Sci. 2018;8(7):123.
22. Hardman MI, Sprung J, Weingarten TN. Acute phenibut withdrawal: a comprehensive literature review and illustrative case report. Bosn J Basic Med Sci. 2019;19(2):125-129.
23. Guzman JR, Paterniti DA, Liu Y, et al. Factors related to disclosure and nondisclosure of dietary supplements in primary care, integrative medicine, and naturopathic medicine. J Fam Med Dis Prev. 2019;5(4):10.23937/2469-5793/1510109.
24. Foley H, Steel A, Cramer H, et al. Disclosure of complementary medicine use to medical providers: a systematic review and meta-analysis. Sci Rep. 2019;9(1):1573.
25. Aldridge Young C. ‘No miracle cures’: counseling patients about dietary supplements. Pharmacy Today. 2014;February:35.
26. United States Pharmacopeia. USP Verified Mark. Accessed January 25, 2023. https://www.usp.org/verification-services/verified-mark
1. Graziano S, Orsolini L, Rotolo MC, et al. Herbal highs: review on psychoactive effects and neuropharmacology. Curr Neuropharmacol. 2017;15(5):750-761.
2. Mishra S, Stierman B, Gahche JJ, et al. Dietary supplement use among adults: United States, 2017-2018. NCHS Data Brief. 2021;(399):1-8.
3. O’Neill-Dee C, Spiller HA, Casavant MJ, et al. Natural psychoactive substance-related exposures reported to United States poison control centers, 2000-2017. Clin Toxicol (Phila). 2020;58(8):813-820.
4. Gray DC, Rutledge CM. Herbal supplements in primary care: patient perceptions, motivations, and effects on use. Holist Nurs Pract. 2013;27(1):6-12.
5. Wu K, Messamore E. Reimagining roles of dietary supplements in psychiatric care. AMA J Ethics. 2022;24(5):E437-E442.
6. Snyder FJ, Dundas ML, Kirkpatrick C, et al. Use and safety perceptions regarding herbal supplements: a study of older persons in southeast Idaho. J Nutr Elder. 2009;28(1):81-95.
7. Schulz P, Hede V. Alternative and complementary approaches in psychiatry: beliefs versus evidence. Dialogues Clin Neurosci. 2018;20(3):207-214.
8. Dietary Supplement Health and Education Act of 1994, Pub L 103-417, 103rd Cong (1993-1994).
9. Starr RR. Too little, too late: ineffective regulation of dietary supplements in the United States. Am J Public Health. 2015;105(3):478-485.
10. New psychoactive substances. Alcohol and Drug Foundation. November 10, 2021. Updated November 28, 2022. Accessed January 25, 2023. https://adf.org.au/drug-facts/new-psychoactive-substances/
11. Shafi A, Berry AJ, Sumnall H, et al. New psychoactive substances: a review and updates. Ther Adv Psychopharmacol. 2020;10:2045125320967197.
12. Bersani FS, Coviello M, Imperatori C, et al. Adverse psychiatric effects associated with herbal weight-loss products. Biomed Res Int. 2015;2015:120679.
13. IBM Micromedex POISINDEX® System. IBM Watson Health. Accessed October 3, 2022. https://www.micromedexsolutions.com
14. Natural Medicines Comprehensive Database. Therapeutic Research Center. Accessed October 3, 2022. https://naturalmedicines.therapeuticresearch.com
15. Savage KM, Stough CK, Byrne GJ, et al. Kava for the treatment of generalised anxiety disorder (K-GAD): study protocol for a randomised controlled trial. Trials. 2015;16:493.
16. Swogger MT, Smith KE, Garcia-Romeu A, et al. Understanding kratom use: a guide for healthcare providers. Front Pharmacol. 2022;13:801855.
17. Modabbernia A, Akhondzadeh S. Saffron, passionflower, valerian and sage for mental health. Psychiatr Clin North Am. 2013;36(1):85-91.
18. Coffeen U, Pellicer F. Salvia divinorum: from recreational hallucinogenic use to analgesic and anti-inflammatory action. J Pain Res. 2019;12:1069-1076.
19. National Institutes of Health, Office of Dietary Supplements. Valerian Fact Sheet for Health Professionals. Updated March 15, 2013. Accessed January 25, 2023. https://ods.od.nih.gov/factsheets/Valerian-HealthProfessional
20. An H, Sohn H, Chung S. Phentermine, sibutramine and affective disorders. Clin Psychopharmacol Neurosci. 2013;11(1):7-12.
21. Miliano C, Margiani G, Fattore L, et al. Sales and advertising channels of new psychoactive substances (NPS): internet, social networks, and smartphone apps. Brain Sci. 2018;8(7):123.
22. Hardman MI, Sprung J, Weingarten TN. Acute phenibut withdrawal: a comprehensive literature review and illustrative case report. Bosn J Basic Med Sci. 2019;19(2):125-129.
23. Guzman JR, Paterniti DA, Liu Y, et al. Factors related to disclosure and nondisclosure of dietary supplements in primary care, integrative medicine, and naturopathic medicine. J Fam Med Dis Prev. 2019;5(4):10.23937/2469-5793/1510109.
24. Foley H, Steel A, Cramer H, et al. Disclosure of complementary medicine use to medical providers: a systematic review and meta-analysis. Sci Rep. 2019;9(1):1573.
25. Aldridge Young C. ‘No miracle cures’: counseling patients about dietary supplements. Pharmacy Today. 2014;February:35.
26. United States Pharmacopeia. USP Verified Mark. Accessed January 25, 2023. https://www.usp.org/verification-services/verified-mark
Increased anxiety and depression after menstruation
CASE Increased anxiety and depression
Ms. C, age 29, has bipolar II disorder (BD II) and generalized anxiety disorder. She presents to her outpatient psychiatrist seeking relief from chronic and significant dips in her mood from Day 5 to Day 15 of her menstrual cycle. During this time, she says she experiences increased anxiety, insomnia, frequent tearfulness, and intermittent suicidal ideation.
Ms. C meticulously charts her menstrual cycle using a smartphone app and reports having a regular 28-day cycle. She says she has experienced this worsening of symptoms since the onset of menarche, but her mood generally stabilizes after Day 14 of her cycle–around the time of ovulation–and remains euthymic throughout the premenstrual period.
HISTORY Depression and a change in medication
Ms. C has a history of major depressive episodes and has experienced hypomanic episodes that lasted 1 to 2 weeks and were associated with an elevated mood, high energy, rapid speech, and increased self-confidence. Ms. C says she has chronically high anxiety associated with trouble sleeping, difficulty focusing, restlessness, and muscle tension. When she was receiving care from previous psychiatrists, treatment with lithium, quetiapine, lamotrigine, sertraline, and fluoxetine was not successful, and Ms. C said she had severe anxiety when she tried sertraline and fluoxetine. After several months of substantial mood instability and high anxiety, Ms. C responded well to pregabalin 100 mg 3 times a day, lurasidone 60 mg/d at bedtime, and gabapentin 500 mg/d at bedtime. Over the last 4 months, she reports that her overall mood has been even, and she has been coping well with her anxiety.
Ms. C is married with no children. She uses condoms for birth control. She previously tried taking a combined estrogen/progestin oral contraceptive, but stopped because she said it made her feel very depressed. Ms. C reports no history of substance use. She is employed, says she has many positive relationships, and does not have a social history suggestive of a personality disorder.
[polldaddy:11818926]
The author’s observations
Many women report worsening of mood during the premenstrual period (luteal phase). Premenstrual dysphoric disorder (PMDD) involves symptoms that develop during the luteal phase and end shortly after menstruation; this condition impacts ≤5% of women.1 The etiology of PMDD appears to involve contributions from genetics, hormones such as estrogen and progesterone, allopregnanolone (a progesterone metabolite), brain-derived neurotrophic factor, brain structural and functional differences, and hypothalamic pathways.2
Researchers have postulated that the precipitous decline in the levels of progesterone and allopregnanolone in the luteal phase may contribute to the mood symptoms of PMDD.2 Allopregnanolone is a modulator of gamma-aminobutyric acid type A (GABA-A) receptors and may exert anxiolytic and sedative effects. Women who experience PMDD may be less sensitive to the effects of allopregnanolone.3 Additionally, early luteal phase levels of estrogen may predict late luteal phase symptoms of PMDD.4 The mechanism involved may be estrogen’s effect on the serotonin system. The HPA axis may also be involved in the etiology of PMDD because patients with this condition appear to have a blunted cortisol response in reaction to stress.5 Research also has implicated immune activation and inflammation in the etiology of PMDD.6
A PMDD diagnosis should be distinguished from a premenstrual exacerbation of an underlying psychiatric condition, which occurs when a patient has an untreated primary mood or anxiety disorder that worsens during the premenstrual period. PMDD is differentiated from premenstrual syndrome by the severity of symptoms.2 The recommended first-line treatment of PMDD is an SSRI, but if an SSRI does not work, is not tolerated, or is not preferred for any other reason, recommended alternatives include combined hormone oral contraceptive pills, dutasteride, gabapentin, or various supplements.7,8 PMDD has been widely studied and is treated by both psychiatrists and gynecologists. In addition, some women report experiencing mood instability around ovulation. Kiesner9 found that 13% of women studied showed an increased negative mood state midcycle, rather than during the premenstrual period.
Continue to: Postmenstrual syndrome
Postmenstrual syndrome
Postmenstrual mood symptoms are atypical. Postmenstrual syndrome is not listed in DSM-5 or formally recognized as a medical diagnosis. Peer-reviewed research or literature on the condition is scarce to nonexistent. However, it has been discussed by physicians in articles in the lay press. One gynecologist and reproductive endocrinologist estimated that approximately 10% of women experience significant physical and emotional symptoms postmenstruation.10 An internist and women’s health specialist suggested that the cause of postmenstrual syndrome might be a surge in levels of estrogen and testosterone and may be associated with insulin resistance and polycystic ovarian syndrome, while another possible contribution could be iron deficiency caused by loss of blood from menstruation.11
TREATMENT Recommending an oral contraceptive
Ms. C’s psychiatrist does not prescribe an SSRI because he is concerned it would destabilize her BD II. The patient also had negative experiences in her past 2 trials of SSRIs.
Because the psychiatrist believes it is prudent to optimize the dosages of a patient’s current medication before starting a new medication or intervention, he considers increasing Ms. C’s dosage of lurasidone or pregabalin. The rationale for optimizing Ms. C’s current medication regimen is that greater overall mood stability would likely result in less severe postmenstrual mood symptoms. However, Ms. C does not want to increase her dosage of either medication because she is concerned about adverse effects.
Ms. C’s psychiatrist discusses the case with 2 gynecologist/obstetrician colleagues. One suggests the patient try a progesterone-only oral contraceptive and the other suggests a trial of Prometrium (a progesterone capsule used to treat endometrial hyperplasia and secondary amenorrhea). Both suggestions are based on the theory that Ms. C may be sensitive to levels of progesterone, which are low during the follicular phase and rise after ovulation; neither recommendation is evidence-based. A low level of allopregnanolone may lead to less GABAergic activity and consequently greater mood dysregulation. Some women are particularly sensitive to low levels of allopregnanolone in the follicular phase, which might lead to postmenstrual mood symptoms. Additionally, Ms. C’s previous treatment with a combined estrogen/progestin oral contraceptive may have decreased her level of allopregnanolone.12 Ultimately, Ms. C’s psychiatrist suggests that she take a progesterone-only oral contraceptive.
The author’s observations
Guidance on how to treat Ms. C’s postmenstrual symptoms came from research on how to treat PMDD in patients who have BD. In a review of managing PMDD in women with BD, Sepede et al13 presented a treatment algorithm that recommends a combined estrogen/progestin oral contraceptive as first-line treatment in euthymic patients who are already receiving an optimal dose of mood stabilizers. Sepede et al13 expressed caution about using SSRIs due to the risk of inducing mood changes, but recommended SSRIs for patients with comorbid PMDD and BD who experience a depressive episode.
Another question is which type of oral contraceptive is most effective for treating PMDD. The combined oral contraceptive drospirenone/ethinyl estradiol has the most evidence for efficacy.14 Combined oral contraceptives carry risks of venous thromboembolism, hypertension, stroke, migraines, and liver complications, and are possibly associated with certain types of cancer, such as breast and cervical cancer.15 Their use is contraindicated in patients with a history of these conditions and for women age >35 who smoke ≥15 cigarettes/d.
The limited research that has examined the efficacy of progestin-only oral contraceptives for treating PMDD has been inconclusive.16 However, progesterone-only oral contraceptives are associated with less overall risk than combined oral contraceptives, and many women opt to use progesterone-only oral contraceptives due to concerns about possible adverse effects of the combined formulations. A substantial drawback of progesterone-only oral contraceptives is they must be taken at the same time every day, and if a dose is taken late, these agents may lose their efficacy in preventing pregnancy (and a backup birth control method must be used17). Additionally, drospirenone, a progestin that is a component of many oral contraceptives, has antimineralocorticoid properties and is contraindicated in patients with kidney or adrenal gland insufficiency or liver disease. As was the case when Ms. C initially took a combined contraceptive, hormonal contraceptives can sometimes cause mood dysregulation.
Continue to: OUTCOME Improved symptoms
OUTCOME Improved symptoms
Ms. C meets with her gynecologist, who prescribes norethindrone, a progestin-only oral contraceptive. Since taking norethindrone, Ms. C reports a dramatic improvement in the mood symptoms she experiences during the postmenstrual period.
Bottom Line
Some women may experience mood symptoms during the postmenstrual period that are similar to the symptoms experienced by patients who have premenstrual dysphoric disorder (PMDD). This phenomenon has been described as postmenstrual syndrome, and though evidence is lacking, treating it similarly to PMDD may be effective.
Related Resources
- Ray P, Mandal N, Sinha VK. Change of symptoms of schizophrenia across phases of menstrual cycle. Arch Womens Ment Health. 2020;23(1):113-122. doi:10.1007/s00737-019-0952-4
- Raffi ER, Freeman MP. The etiology of premenstrual dysphoric disorder: 5 interwoven pieces. Current Psychiatry. 2017;16(9):20-28.
Drug Brand Names
Drospirenone/ethinyl estradiol • Yasmin
Dutasteride • Avodart
Fluoxetine • Prozac
Gabapentin • Neurontin
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Norethindrone • Aygestin
Pregabalin • Lyrica
Progesterone • Prometrium
Quetiapine • Seroquel
Sertraline • Zoloft
1. Epperson CN, Steiner M, Hartlage SA, et al. Premenstrual dysphoric disorder: evidence for a new category for DSM-5. Am J Psychiatry. 2012;169(5):465-475.
2. Raffi ER, Freeman MP. The etiology of premenstrual dysphoric disorder: 5 interwoven pieces. Current Psychiatry. 2017;16(9):20-28.
3. Timby E, Bäckström T, Nyberg S, et al. Women with premenstrual dysphoric disorder have altered sensitivity to allopregnanolone over the menstrual cycle compared to controls--a pilot study. Psychopharmacology (Berl). 2016;233(11):2109-2117.
4. Yen JY, Lin HC, Lin PC, et al. Early- and late-luteal-phase estrogen and progesterone levels of women with premenstrual dysphoric disorder. Int J Environ Res Public Health. 2019;16(22):4352.
5. Huang Y, Zhou R, Wu M, et al. Premenstrual syndrome is associated with blunted cortisol reactivity to the TSST. Stress. 2015;18(2):160-168.
6. Hantsoo L, Epperson CN. Premenstrual dysphoric disorder: epidemiology and treatment. Curr Psychiatry Rep. 2015;17(11):87.
7. Tiranini L, Nappi RE. Recent advances in understanding/management of premenstrual dysphoric disorder/premenstrual syndrome. Faculty Rev. 2022:11:(11). doi:10.12703/r/11-11
8. Raffi ER. Premenstrual dysphoric disorder. Current Psychiatry. 2017;16(9). Accessed January 30, 2023. https://www.mdedge.com/psychiatry/article/145089/somatic-disorders/premenstrual-dysphoric-disorder
9. Kiesner J. One woman’s low is another woman’s high: paradoxical effects of the menstrual cycle. Psychoneuroendocrinology. 2011;36(1):68-76.
10. Alnuweiri T. Feel low after your period? Postmenstrual syndrome could be the reason. Accessed January 30, 2023. https://www.wellandgood.com/pms-after-period/
11. Sharkey L. Everything you need to know about post-menstrual syndrome. Healthline. Published April 28, 2020. Accessed January 30, 2023. https://www.healthline.com/health/post-menstrual-syndrome
12. Santoru F, Berretti R, Locci A, et al. Decreased allopregnanolone induced by hormonal contraceptives is associated with a reduction in social behavior and sexual motivation in female rats. Psychopharmacology (Berl). 2014;231(17):3351-3364.
13. Sepede G, Brunetti M, Di Giannantonio M. Comorbid premenstrual dysphoric disorder in women with bipolar disorder: management challenges. Neuropsychiatr Dis Treatment. 2020;16:415-426.
14. Rapkin AJ, Korotkaya Y, Taylor KC. Contraception counseling for women with premenstrual dysphoric disorder (PMDD): current perspectives. Open Access J Contraception. 2019;10:27-39. doi:10.2147/OAJC.S183193
15. Roe AH, Bartz DA, Douglas PS. Combined estrogen-progestin contraception: side effects and health concerns. UpToDate. Accessed February 1, 2023. https://www.uptodate.com/contents/combined-estrogen-progestin-contraception-side-effects-and-health-concerns
16. Ford O, Lethaby A, Roberts H, et al. Progesterone for premenstrual syndrome. Cochrane Database Sys Rev. 2012;3:CD003415. doi:10.1002/14651858.CD003415.pub4
17. Kaunitz AM. Contraception: progestin-only pills (POPs). UpToDate. Accessed February 1, 2023. https://www.uptodate.com/contents/contraception-progestin-only-pills-pops
CASE Increased anxiety and depression
Ms. C, age 29, has bipolar II disorder (BD II) and generalized anxiety disorder. She presents to her outpatient psychiatrist seeking relief from chronic and significant dips in her mood from Day 5 to Day 15 of her menstrual cycle. During this time, she says she experiences increased anxiety, insomnia, frequent tearfulness, and intermittent suicidal ideation.
Ms. C meticulously charts her menstrual cycle using a smartphone app and reports having a regular 28-day cycle. She says she has experienced this worsening of symptoms since the onset of menarche, but her mood generally stabilizes after Day 14 of her cycle–around the time of ovulation–and remains euthymic throughout the premenstrual period.
HISTORY Depression and a change in medication
Ms. C has a history of major depressive episodes and has experienced hypomanic episodes that lasted 1 to 2 weeks and were associated with an elevated mood, high energy, rapid speech, and increased self-confidence. Ms. C says she has chronically high anxiety associated with trouble sleeping, difficulty focusing, restlessness, and muscle tension. When she was receiving care from previous psychiatrists, treatment with lithium, quetiapine, lamotrigine, sertraline, and fluoxetine was not successful, and Ms. C said she had severe anxiety when she tried sertraline and fluoxetine. After several months of substantial mood instability and high anxiety, Ms. C responded well to pregabalin 100 mg 3 times a day, lurasidone 60 mg/d at bedtime, and gabapentin 500 mg/d at bedtime. Over the last 4 months, she reports that her overall mood has been even, and she has been coping well with her anxiety.
Ms. C is married with no children. She uses condoms for birth control. She previously tried taking a combined estrogen/progestin oral contraceptive, but stopped because she said it made her feel very depressed. Ms. C reports no history of substance use. She is employed, says she has many positive relationships, and does not have a social history suggestive of a personality disorder.
[polldaddy:11818926]
The author’s observations
Many women report worsening of mood during the premenstrual period (luteal phase). Premenstrual dysphoric disorder (PMDD) involves symptoms that develop during the luteal phase and end shortly after menstruation; this condition impacts ≤5% of women.1 The etiology of PMDD appears to involve contributions from genetics, hormones such as estrogen and progesterone, allopregnanolone (a progesterone metabolite), brain-derived neurotrophic factor, brain structural and functional differences, and hypothalamic pathways.2
Researchers have postulated that the precipitous decline in the levels of progesterone and allopregnanolone in the luteal phase may contribute to the mood symptoms of PMDD.2 Allopregnanolone is a modulator of gamma-aminobutyric acid type A (GABA-A) receptors and may exert anxiolytic and sedative effects. Women who experience PMDD may be less sensitive to the effects of allopregnanolone.3 Additionally, early luteal phase levels of estrogen may predict late luteal phase symptoms of PMDD.4 The mechanism involved may be estrogen’s effect on the serotonin system. The HPA axis may also be involved in the etiology of PMDD because patients with this condition appear to have a blunted cortisol response in reaction to stress.5 Research also has implicated immune activation and inflammation in the etiology of PMDD.6
A PMDD diagnosis should be distinguished from a premenstrual exacerbation of an underlying psychiatric condition, which occurs when a patient has an untreated primary mood or anxiety disorder that worsens during the premenstrual period. PMDD is differentiated from premenstrual syndrome by the severity of symptoms.2 The recommended first-line treatment of PMDD is an SSRI, but if an SSRI does not work, is not tolerated, or is not preferred for any other reason, recommended alternatives include combined hormone oral contraceptive pills, dutasteride, gabapentin, or various supplements.7,8 PMDD has been widely studied and is treated by both psychiatrists and gynecologists. In addition, some women report experiencing mood instability around ovulation. Kiesner9 found that 13% of women studied showed an increased negative mood state midcycle, rather than during the premenstrual period.
Continue to: Postmenstrual syndrome
Postmenstrual syndrome
Postmenstrual mood symptoms are atypical. Postmenstrual syndrome is not listed in DSM-5 or formally recognized as a medical diagnosis. Peer-reviewed research or literature on the condition is scarce to nonexistent. However, it has been discussed by physicians in articles in the lay press. One gynecologist and reproductive endocrinologist estimated that approximately 10% of women experience significant physical and emotional symptoms postmenstruation.10 An internist and women’s health specialist suggested that the cause of postmenstrual syndrome might be a surge in levels of estrogen and testosterone and may be associated with insulin resistance and polycystic ovarian syndrome, while another possible contribution could be iron deficiency caused by loss of blood from menstruation.11
TREATMENT Recommending an oral contraceptive
Ms. C’s psychiatrist does not prescribe an SSRI because he is concerned it would destabilize her BD II. The patient also had negative experiences in her past 2 trials of SSRIs.
Because the psychiatrist believes it is prudent to optimize the dosages of a patient’s current medication before starting a new medication or intervention, he considers increasing Ms. C’s dosage of lurasidone or pregabalin. The rationale for optimizing Ms. C’s current medication regimen is that greater overall mood stability would likely result in less severe postmenstrual mood symptoms. However, Ms. C does not want to increase her dosage of either medication because she is concerned about adverse effects.
Ms. C’s psychiatrist discusses the case with 2 gynecologist/obstetrician colleagues. One suggests the patient try a progesterone-only oral contraceptive and the other suggests a trial of Prometrium (a progesterone capsule used to treat endometrial hyperplasia and secondary amenorrhea). Both suggestions are based on the theory that Ms. C may be sensitive to levels of progesterone, which are low during the follicular phase and rise after ovulation; neither recommendation is evidence-based. A low level of allopregnanolone may lead to less GABAergic activity and consequently greater mood dysregulation. Some women are particularly sensitive to low levels of allopregnanolone in the follicular phase, which might lead to postmenstrual mood symptoms. Additionally, Ms. C’s previous treatment with a combined estrogen/progestin oral contraceptive may have decreased her level of allopregnanolone.12 Ultimately, Ms. C’s psychiatrist suggests that she take a progesterone-only oral contraceptive.
The author’s observations
Guidance on how to treat Ms. C’s postmenstrual symptoms came from research on how to treat PMDD in patients who have BD. In a review of managing PMDD in women with BD, Sepede et al13 presented a treatment algorithm that recommends a combined estrogen/progestin oral contraceptive as first-line treatment in euthymic patients who are already receiving an optimal dose of mood stabilizers. Sepede et al13 expressed caution about using SSRIs due to the risk of inducing mood changes, but recommended SSRIs for patients with comorbid PMDD and BD who experience a depressive episode.
Another question is which type of oral contraceptive is most effective for treating PMDD. The combined oral contraceptive drospirenone/ethinyl estradiol has the most evidence for efficacy.14 Combined oral contraceptives carry risks of venous thromboembolism, hypertension, stroke, migraines, and liver complications, and are possibly associated with certain types of cancer, such as breast and cervical cancer.15 Their use is contraindicated in patients with a history of these conditions and for women age >35 who smoke ≥15 cigarettes/d.
The limited research that has examined the efficacy of progestin-only oral contraceptives for treating PMDD has been inconclusive.16 However, progesterone-only oral contraceptives are associated with less overall risk than combined oral contraceptives, and many women opt to use progesterone-only oral contraceptives due to concerns about possible adverse effects of the combined formulations. A substantial drawback of progesterone-only oral contraceptives is they must be taken at the same time every day, and if a dose is taken late, these agents may lose their efficacy in preventing pregnancy (and a backup birth control method must be used17). Additionally, drospirenone, a progestin that is a component of many oral contraceptives, has antimineralocorticoid properties and is contraindicated in patients with kidney or adrenal gland insufficiency or liver disease. As was the case when Ms. C initially took a combined contraceptive, hormonal contraceptives can sometimes cause mood dysregulation.
Continue to: OUTCOME Improved symptoms
OUTCOME Improved symptoms
Ms. C meets with her gynecologist, who prescribes norethindrone, a progestin-only oral contraceptive. Since taking norethindrone, Ms. C reports a dramatic improvement in the mood symptoms she experiences during the postmenstrual period.
Bottom Line
Some women may experience mood symptoms during the postmenstrual period that are similar to the symptoms experienced by patients who have premenstrual dysphoric disorder (PMDD). This phenomenon has been described as postmenstrual syndrome, and though evidence is lacking, treating it similarly to PMDD may be effective.
Related Resources
- Ray P, Mandal N, Sinha VK. Change of symptoms of schizophrenia across phases of menstrual cycle. Arch Womens Ment Health. 2020;23(1):113-122. doi:10.1007/s00737-019-0952-4
- Raffi ER, Freeman MP. The etiology of premenstrual dysphoric disorder: 5 interwoven pieces. Current Psychiatry. 2017;16(9):20-28.
Drug Brand Names
Drospirenone/ethinyl estradiol • Yasmin
Dutasteride • Avodart
Fluoxetine • Prozac
Gabapentin • Neurontin
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Norethindrone • Aygestin
Pregabalin • Lyrica
Progesterone • Prometrium
Quetiapine • Seroquel
Sertraline • Zoloft
CASE Increased anxiety and depression
Ms. C, age 29, has bipolar II disorder (BD II) and generalized anxiety disorder. She presents to her outpatient psychiatrist seeking relief from chronic and significant dips in her mood from Day 5 to Day 15 of her menstrual cycle. During this time, she says she experiences increased anxiety, insomnia, frequent tearfulness, and intermittent suicidal ideation.
Ms. C meticulously charts her menstrual cycle using a smartphone app and reports having a regular 28-day cycle. She says she has experienced this worsening of symptoms since the onset of menarche, but her mood generally stabilizes after Day 14 of her cycle–around the time of ovulation–and remains euthymic throughout the premenstrual period.
HISTORY Depression and a change in medication
Ms. C has a history of major depressive episodes and has experienced hypomanic episodes that lasted 1 to 2 weeks and were associated with an elevated mood, high energy, rapid speech, and increased self-confidence. Ms. C says she has chronically high anxiety associated with trouble sleeping, difficulty focusing, restlessness, and muscle tension. When she was receiving care from previous psychiatrists, treatment with lithium, quetiapine, lamotrigine, sertraline, and fluoxetine was not successful, and Ms. C said she had severe anxiety when she tried sertraline and fluoxetine. After several months of substantial mood instability and high anxiety, Ms. C responded well to pregabalin 100 mg 3 times a day, lurasidone 60 mg/d at bedtime, and gabapentin 500 mg/d at bedtime. Over the last 4 months, she reports that her overall mood has been even, and she has been coping well with her anxiety.
Ms. C is married with no children. She uses condoms for birth control. She previously tried taking a combined estrogen/progestin oral contraceptive, but stopped because she said it made her feel very depressed. Ms. C reports no history of substance use. She is employed, says she has many positive relationships, and does not have a social history suggestive of a personality disorder.
[polldaddy:11818926]
The author’s observations
Many women report worsening of mood during the premenstrual period (luteal phase). Premenstrual dysphoric disorder (PMDD) involves symptoms that develop during the luteal phase and end shortly after menstruation; this condition impacts ≤5% of women.1 The etiology of PMDD appears to involve contributions from genetics, hormones such as estrogen and progesterone, allopregnanolone (a progesterone metabolite), brain-derived neurotrophic factor, brain structural and functional differences, and hypothalamic pathways.2
Researchers have postulated that the precipitous decline in the levels of progesterone and allopregnanolone in the luteal phase may contribute to the mood symptoms of PMDD.2 Allopregnanolone is a modulator of gamma-aminobutyric acid type A (GABA-A) receptors and may exert anxiolytic and sedative effects. Women who experience PMDD may be less sensitive to the effects of allopregnanolone.3 Additionally, early luteal phase levels of estrogen may predict late luteal phase symptoms of PMDD.4 The mechanism involved may be estrogen’s effect on the serotonin system. The HPA axis may also be involved in the etiology of PMDD because patients with this condition appear to have a blunted cortisol response in reaction to stress.5 Research also has implicated immune activation and inflammation in the etiology of PMDD.6
A PMDD diagnosis should be distinguished from a premenstrual exacerbation of an underlying psychiatric condition, which occurs when a patient has an untreated primary mood or anxiety disorder that worsens during the premenstrual period. PMDD is differentiated from premenstrual syndrome by the severity of symptoms.2 The recommended first-line treatment of PMDD is an SSRI, but if an SSRI does not work, is not tolerated, or is not preferred for any other reason, recommended alternatives include combined hormone oral contraceptive pills, dutasteride, gabapentin, or various supplements.7,8 PMDD has been widely studied and is treated by both psychiatrists and gynecologists. In addition, some women report experiencing mood instability around ovulation. Kiesner9 found that 13% of women studied showed an increased negative mood state midcycle, rather than during the premenstrual period.
Continue to: Postmenstrual syndrome
Postmenstrual syndrome
Postmenstrual mood symptoms are atypical. Postmenstrual syndrome is not listed in DSM-5 or formally recognized as a medical diagnosis. Peer-reviewed research or literature on the condition is scarce to nonexistent. However, it has been discussed by physicians in articles in the lay press. One gynecologist and reproductive endocrinologist estimated that approximately 10% of women experience significant physical and emotional symptoms postmenstruation.10 An internist and women’s health specialist suggested that the cause of postmenstrual syndrome might be a surge in levels of estrogen and testosterone and may be associated with insulin resistance and polycystic ovarian syndrome, while another possible contribution could be iron deficiency caused by loss of blood from menstruation.11
TREATMENT Recommending an oral contraceptive
Ms. C’s psychiatrist does not prescribe an SSRI because he is concerned it would destabilize her BD II. The patient also had negative experiences in her past 2 trials of SSRIs.
Because the psychiatrist believes it is prudent to optimize the dosages of a patient’s current medication before starting a new medication or intervention, he considers increasing Ms. C’s dosage of lurasidone or pregabalin. The rationale for optimizing Ms. C’s current medication regimen is that greater overall mood stability would likely result in less severe postmenstrual mood symptoms. However, Ms. C does not want to increase her dosage of either medication because she is concerned about adverse effects.
Ms. C’s psychiatrist discusses the case with 2 gynecologist/obstetrician colleagues. One suggests the patient try a progesterone-only oral contraceptive and the other suggests a trial of Prometrium (a progesterone capsule used to treat endometrial hyperplasia and secondary amenorrhea). Both suggestions are based on the theory that Ms. C may be sensitive to levels of progesterone, which are low during the follicular phase and rise after ovulation; neither recommendation is evidence-based. A low level of allopregnanolone may lead to less GABAergic activity and consequently greater mood dysregulation. Some women are particularly sensitive to low levels of allopregnanolone in the follicular phase, which might lead to postmenstrual mood symptoms. Additionally, Ms. C’s previous treatment with a combined estrogen/progestin oral contraceptive may have decreased her level of allopregnanolone.12 Ultimately, Ms. C’s psychiatrist suggests that she take a progesterone-only oral contraceptive.
The author’s observations
Guidance on how to treat Ms. C’s postmenstrual symptoms came from research on how to treat PMDD in patients who have BD. In a review of managing PMDD in women with BD, Sepede et al13 presented a treatment algorithm that recommends a combined estrogen/progestin oral contraceptive as first-line treatment in euthymic patients who are already receiving an optimal dose of mood stabilizers. Sepede et al13 expressed caution about using SSRIs due to the risk of inducing mood changes, but recommended SSRIs for patients with comorbid PMDD and BD who experience a depressive episode.
Another question is which type of oral contraceptive is most effective for treating PMDD. The combined oral contraceptive drospirenone/ethinyl estradiol has the most evidence for efficacy.14 Combined oral contraceptives carry risks of venous thromboembolism, hypertension, stroke, migraines, and liver complications, and are possibly associated with certain types of cancer, such as breast and cervical cancer.15 Their use is contraindicated in patients with a history of these conditions and for women age >35 who smoke ≥15 cigarettes/d.
The limited research that has examined the efficacy of progestin-only oral contraceptives for treating PMDD has been inconclusive.16 However, progesterone-only oral contraceptives are associated with less overall risk than combined oral contraceptives, and many women opt to use progesterone-only oral contraceptives due to concerns about possible adverse effects of the combined formulations. A substantial drawback of progesterone-only oral contraceptives is they must be taken at the same time every day, and if a dose is taken late, these agents may lose their efficacy in preventing pregnancy (and a backup birth control method must be used17). Additionally, drospirenone, a progestin that is a component of many oral contraceptives, has antimineralocorticoid properties and is contraindicated in patients with kidney or adrenal gland insufficiency or liver disease. As was the case when Ms. C initially took a combined contraceptive, hormonal contraceptives can sometimes cause mood dysregulation.
Continue to: OUTCOME Improved symptoms
OUTCOME Improved symptoms
Ms. C meets with her gynecologist, who prescribes norethindrone, a progestin-only oral contraceptive. Since taking norethindrone, Ms. C reports a dramatic improvement in the mood symptoms she experiences during the postmenstrual period.
Bottom Line
Some women may experience mood symptoms during the postmenstrual period that are similar to the symptoms experienced by patients who have premenstrual dysphoric disorder (PMDD). This phenomenon has been described as postmenstrual syndrome, and though evidence is lacking, treating it similarly to PMDD may be effective.
Related Resources
- Ray P, Mandal N, Sinha VK. Change of symptoms of schizophrenia across phases of menstrual cycle. Arch Womens Ment Health. 2020;23(1):113-122. doi:10.1007/s00737-019-0952-4
- Raffi ER, Freeman MP. The etiology of premenstrual dysphoric disorder: 5 interwoven pieces. Current Psychiatry. 2017;16(9):20-28.
Drug Brand Names
Drospirenone/ethinyl estradiol • Yasmin
Dutasteride • Avodart
Fluoxetine • Prozac
Gabapentin • Neurontin
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Norethindrone • Aygestin
Pregabalin • Lyrica
Progesterone • Prometrium
Quetiapine • Seroquel
Sertraline • Zoloft
1. Epperson CN, Steiner M, Hartlage SA, et al. Premenstrual dysphoric disorder: evidence for a new category for DSM-5. Am J Psychiatry. 2012;169(5):465-475.
2. Raffi ER, Freeman MP. The etiology of premenstrual dysphoric disorder: 5 interwoven pieces. Current Psychiatry. 2017;16(9):20-28.
3. Timby E, Bäckström T, Nyberg S, et al. Women with premenstrual dysphoric disorder have altered sensitivity to allopregnanolone over the menstrual cycle compared to controls--a pilot study. Psychopharmacology (Berl). 2016;233(11):2109-2117.
4. Yen JY, Lin HC, Lin PC, et al. Early- and late-luteal-phase estrogen and progesterone levels of women with premenstrual dysphoric disorder. Int J Environ Res Public Health. 2019;16(22):4352.
5. Huang Y, Zhou R, Wu M, et al. Premenstrual syndrome is associated with blunted cortisol reactivity to the TSST. Stress. 2015;18(2):160-168.
6. Hantsoo L, Epperson CN. Premenstrual dysphoric disorder: epidemiology and treatment. Curr Psychiatry Rep. 2015;17(11):87.
7. Tiranini L, Nappi RE. Recent advances in understanding/management of premenstrual dysphoric disorder/premenstrual syndrome. Faculty Rev. 2022:11:(11). doi:10.12703/r/11-11
8. Raffi ER. Premenstrual dysphoric disorder. Current Psychiatry. 2017;16(9). Accessed January 30, 2023. https://www.mdedge.com/psychiatry/article/145089/somatic-disorders/premenstrual-dysphoric-disorder
9. Kiesner J. One woman’s low is another woman’s high: paradoxical effects of the menstrual cycle. Psychoneuroendocrinology. 2011;36(1):68-76.
10. Alnuweiri T. Feel low after your period? Postmenstrual syndrome could be the reason. Accessed January 30, 2023. https://www.wellandgood.com/pms-after-period/
11. Sharkey L. Everything you need to know about post-menstrual syndrome. Healthline. Published April 28, 2020. Accessed January 30, 2023. https://www.healthline.com/health/post-menstrual-syndrome
12. Santoru F, Berretti R, Locci A, et al. Decreased allopregnanolone induced by hormonal contraceptives is associated with a reduction in social behavior and sexual motivation in female rats. Psychopharmacology (Berl). 2014;231(17):3351-3364.
13. Sepede G, Brunetti M, Di Giannantonio M. Comorbid premenstrual dysphoric disorder in women with bipolar disorder: management challenges. Neuropsychiatr Dis Treatment. 2020;16:415-426.
14. Rapkin AJ, Korotkaya Y, Taylor KC. Contraception counseling for women with premenstrual dysphoric disorder (PMDD): current perspectives. Open Access J Contraception. 2019;10:27-39. doi:10.2147/OAJC.S183193
15. Roe AH, Bartz DA, Douglas PS. Combined estrogen-progestin contraception: side effects and health concerns. UpToDate. Accessed February 1, 2023. https://www.uptodate.com/contents/combined-estrogen-progestin-contraception-side-effects-and-health-concerns
16. Ford O, Lethaby A, Roberts H, et al. Progesterone for premenstrual syndrome. Cochrane Database Sys Rev. 2012;3:CD003415. doi:10.1002/14651858.CD003415.pub4
17. Kaunitz AM. Contraception: progestin-only pills (POPs). UpToDate. Accessed February 1, 2023. https://www.uptodate.com/contents/contraception-progestin-only-pills-pops
1. Epperson CN, Steiner M, Hartlage SA, et al. Premenstrual dysphoric disorder: evidence for a new category for DSM-5. Am J Psychiatry. 2012;169(5):465-475.
2. Raffi ER, Freeman MP. The etiology of premenstrual dysphoric disorder: 5 interwoven pieces. Current Psychiatry. 2017;16(9):20-28.
3. Timby E, Bäckström T, Nyberg S, et al. Women with premenstrual dysphoric disorder have altered sensitivity to allopregnanolone over the menstrual cycle compared to controls--a pilot study. Psychopharmacology (Berl). 2016;233(11):2109-2117.
4. Yen JY, Lin HC, Lin PC, et al. Early- and late-luteal-phase estrogen and progesterone levels of women with premenstrual dysphoric disorder. Int J Environ Res Public Health. 2019;16(22):4352.
5. Huang Y, Zhou R, Wu M, et al. Premenstrual syndrome is associated with blunted cortisol reactivity to the TSST. Stress. 2015;18(2):160-168.
6. Hantsoo L, Epperson CN. Premenstrual dysphoric disorder: epidemiology and treatment. Curr Psychiatry Rep. 2015;17(11):87.
7. Tiranini L, Nappi RE. Recent advances in understanding/management of premenstrual dysphoric disorder/premenstrual syndrome. Faculty Rev. 2022:11:(11). doi:10.12703/r/11-11
8. Raffi ER. Premenstrual dysphoric disorder. Current Psychiatry. 2017;16(9). Accessed January 30, 2023. https://www.mdedge.com/psychiatry/article/145089/somatic-disorders/premenstrual-dysphoric-disorder
9. Kiesner J. One woman’s low is another woman’s high: paradoxical effects of the menstrual cycle. Psychoneuroendocrinology. 2011;36(1):68-76.
10. Alnuweiri T. Feel low after your period? Postmenstrual syndrome could be the reason. Accessed January 30, 2023. https://www.wellandgood.com/pms-after-period/
11. Sharkey L. Everything you need to know about post-menstrual syndrome. Healthline. Published April 28, 2020. Accessed January 30, 2023. https://www.healthline.com/health/post-menstrual-syndrome
12. Santoru F, Berretti R, Locci A, et al. Decreased allopregnanolone induced by hormonal contraceptives is associated with a reduction in social behavior and sexual motivation in female rats. Psychopharmacology (Berl). 2014;231(17):3351-3364.
13. Sepede G, Brunetti M, Di Giannantonio M. Comorbid premenstrual dysphoric disorder in women with bipolar disorder: management challenges. Neuropsychiatr Dis Treatment. 2020;16:415-426.
14. Rapkin AJ, Korotkaya Y, Taylor KC. Contraception counseling for women with premenstrual dysphoric disorder (PMDD): current perspectives. Open Access J Contraception. 2019;10:27-39. doi:10.2147/OAJC.S183193
15. Roe AH, Bartz DA, Douglas PS. Combined estrogen-progestin contraception: side effects and health concerns. UpToDate. Accessed February 1, 2023. https://www.uptodate.com/contents/combined-estrogen-progestin-contraception-side-effects-and-health-concerns
16. Ford O, Lethaby A, Roberts H, et al. Progesterone for premenstrual syndrome. Cochrane Database Sys Rev. 2012;3:CD003415. doi:10.1002/14651858.CD003415.pub4
17. Kaunitz AM. Contraception: progestin-only pills (POPs). UpToDate. Accessed February 1, 2023. https://www.uptodate.com/contents/contraception-progestin-only-pills-pops
Prodromal symptoms of schizophrenia: What to look for
Schizophrenia is characterized by psychotic symptoms that typically follow a prodromal period of premonitory signs and symptoms that appear before the manifestation of the full-blown syndrome. Signs and symptoms during the prodromal phase are subsyndromal, which implies a lower degree of intensity, duration, or frequency than observed when the patient meets the full criteria for the syndrome. Early detection of prodromal symptoms can improve prognosis, but these subtle symptoms may go unrecognized.
In schizophrenia, a patient may exhibit prodromal signs and symptoms before the appearance of pathognomonic symptoms, such as delusions, hallucinations, and disorganization. The schizophrenia prodrome can be conceptualized as a period of prepsychotic disturbances depicting an alteration in the individual’s behavior and perception. Prodromal symptoms can last from weeks to years before the psychotic illness clinically manifests.1 The prodromal symptom cluster typically becomes evident during adolescence and young adulthood.2
In the mid-1990s, investigators tried to identify a “putative prodrome” for psychosis. The term “at-risk mental state” (ARMS) for psychosis is based on retrospective reports of prodromal symptoms in first-episode psychosis. Over the next 2 decades, scales such as the Comprehensive Assessment of ARMS (CAARMS)3 and the Structured Interview for Prodromal Syndrome4 were designed to enhance the objectivity and diagnostic accuracy of the ARMS. These scales have reasonable interrater reliability.5
Researchers also have attempted to stage the severity of ARMS.6 Key symptom group predictors were studied to determine which individual symptoms or cluster of symptoms are most associated with poor outcomes and progression to psychosis. Raballo et al7 found the severity of the CAARMS disorganization dimension was the strongest predictor of transition to frank psychosis. Other research suggests that approximately one-third of ARMS patients transition to psychosis within 3 years, another one-third have persistent attenuated psychotic symptoms, and the remaining one-third experience symptom remission.8,9
Despite multiple studies and meta-analyses, current scales and clinical predictors continue to be imperfect.8 Efforts to identify specific biological markers and predictors of transition to clinical psychosis have not been successful for ARMS.10,11 The Table8,9,12,13 summarizes diagnostic criteria that have been developed to more clearly identify which ARMS patients face the highest imminent risk for transition to psychosis; these have been referred to as ultra high-risk (UHR) criteria.14 These UHR criteria depict 3 categories of clinical presentation believed to confer risk of transition to psychosis: attenuated psychotic symptoms, transient psychotic symptoms, and genetic predisposition. Subsequent research found that certain additional symptom variables, as well as combinations of specific symptom clusters, conferred increased risk and improved the positive predictive sensitivity to as high as 83%.15 In addition to the UHR criteria, the Table8,9,12,13 also lists these additional variables shown to confer a high positive predictive value (PPV) of transition, alone or in combination with the UHR criterion. Thompson et al16 provide more detailed information on these later variables and their relative PPV.

What about treatment?
While discussion of the optimal treatment options for patients with prodromal symptoms of schizophrenia is beyond the scope of this article, early interventions can focus on preventing the biological, psychological, and social disruption that results from such symptoms. Establishing a therapeutic alliance with the patient while they retain insight and engaging supportive family members is a key starting point. Case management, cognitive-behavioral or supportive therapy, and treatment of comorbid mood, anxiety, or substance use disorders are helpful. There is no clear consensus on the utility of pharmacotherapy in the prodromal stage of psychosis. While scales and structured interviews can guide assessment, clinical judgment is the key driver of the appropriateness of initiating pharmacologic treatment to address symptoms. Because up to two-thirds of patients who satisfy UHR criteria do not go on to develop schizophrenia,16 clinicians should be thoughtful about the risks and benefits of antipsychotics.
1. George M, Maheshwari S, Chandran S, et al. Understanding the schizophrenia prodrome. Indian J Psychiatry. 2017;59(4):505-509.
2. Yung AR, McGorry PD. The prodromal phase of first-episode psychosis: past and current conceptualizations. Schizophr Bull. 1996;22(2):353-370.
3. Yung AR, Yuen HP, McGorry PD, et al. Mapping the onset of psychosis: the Comprehensive Assessment of At-Risk Mental States. Aust N Z J Psychiatry. 2005;39(11-12):964-971.
4. Miller TJ, McGlashan TH, Rosen JL, et al. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: predictive validity, interrater reliability, and training to reliability. Schizophr Bull. 2003;29(4):703-715.
5. Loewy RL, Pearson R, Vinogradov S, et al. Psychosis risk screening with the Prodromal Questionnaire--brief version (PQ-B). Schizophr Res. 2011;129(1):42-46.
6. Nieman DH, McGorry PD. Detection and treatment of at-risk mental state for developing a first psychosis: making up the balance. Lancet Psychiatry. 2015;2(9):825-834.
7. Raballo A, Nelson B, Thompson A, et al. The comprehensive assessment of at-risk mental states: from mapping the onset to mapping the structure. Schizophr Res. 2011;127(1-3):107-114.
8. Fusar-Poli P, Bonoldi I, Yung AR, et al. Predicting psychosis: meta-analysis of transition outcomes in individuals at high clinical risk. Arch Gen Psychiatry. 2012;69(3):220-229.
9. Cannon TD. How schizophrenia develops: cognitive and brain mechanisms underlying onset of psychosis. Trends Cogn Sci. 2015;19(12):744-756.
10. Castle DJ. Is it appropriate to treat people at high-risk of psychosis before first onset? - no. Med J Aust. 2012;196(9):557.
11. Wood SJ, Reniers RL, Heinze K. Neuroimaging findings in the at-risk mental state: a review of recent literature. Can J Psychiatry. 2013;58(1):13-18.
12. Nelson B, Yung AR. Can clinicians predict psychosis in an ultra high risk group? Aust N Z J Psychiatry. 2010;44(7):625-630.
13. Schultze-Lutter F, Michel C, Schmidt SJ, et al. EPA guidance on the early detection of clinical high risk states of psychoses. Eur Psychiatry. 2015;30(3):405-416.
14. Yung AR, Phillips LJ, Yuen HP, et al. Risk factors for psychosis in an ultra high-risk group: psychopathology and clinical features. Schizophr Res. 2004;67(2-3):131-142.
15. Ruhrmann S, Schultze-Lutter F, Salokangas RK, et al. Prediction of psychosis in adolescents and young adults at high risk: results from the prospective European prediction of psychosis study. Arch Gen Psychiatry. 2010;67(3):241-251.
16. Thompson A, Marwaha S, Broome MR. At-risk mental state for psychosis: identification and current treatment approaches. BJPsych Advances. 2016;22(3):186-193.
Schizophrenia is characterized by psychotic symptoms that typically follow a prodromal period of premonitory signs and symptoms that appear before the manifestation of the full-blown syndrome. Signs and symptoms during the prodromal phase are subsyndromal, which implies a lower degree of intensity, duration, or frequency than observed when the patient meets the full criteria for the syndrome. Early detection of prodromal symptoms can improve prognosis, but these subtle symptoms may go unrecognized.
In schizophrenia, a patient may exhibit prodromal signs and symptoms before the appearance of pathognomonic symptoms, such as delusions, hallucinations, and disorganization. The schizophrenia prodrome can be conceptualized as a period of prepsychotic disturbances depicting an alteration in the individual’s behavior and perception. Prodromal symptoms can last from weeks to years before the psychotic illness clinically manifests.1 The prodromal symptom cluster typically becomes evident during adolescence and young adulthood.2
In the mid-1990s, investigators tried to identify a “putative prodrome” for psychosis. The term “at-risk mental state” (ARMS) for psychosis is based on retrospective reports of prodromal symptoms in first-episode psychosis. Over the next 2 decades, scales such as the Comprehensive Assessment of ARMS (CAARMS)3 and the Structured Interview for Prodromal Syndrome4 were designed to enhance the objectivity and diagnostic accuracy of the ARMS. These scales have reasonable interrater reliability.5
Researchers also have attempted to stage the severity of ARMS.6 Key symptom group predictors were studied to determine which individual symptoms or cluster of symptoms are most associated with poor outcomes and progression to psychosis. Raballo et al7 found the severity of the CAARMS disorganization dimension was the strongest predictor of transition to frank psychosis. Other research suggests that approximately one-third of ARMS patients transition to psychosis within 3 years, another one-third have persistent attenuated psychotic symptoms, and the remaining one-third experience symptom remission.8,9
Despite multiple studies and meta-analyses, current scales and clinical predictors continue to be imperfect.8 Efforts to identify specific biological markers and predictors of transition to clinical psychosis have not been successful for ARMS.10,11 The Table8,9,12,13 summarizes diagnostic criteria that have been developed to more clearly identify which ARMS patients face the highest imminent risk for transition to psychosis; these have been referred to as ultra high-risk (UHR) criteria.14 These UHR criteria depict 3 categories of clinical presentation believed to confer risk of transition to psychosis: attenuated psychotic symptoms, transient psychotic symptoms, and genetic predisposition. Subsequent research found that certain additional symptom variables, as well as combinations of specific symptom clusters, conferred increased risk and improved the positive predictive sensitivity to as high as 83%.15 In addition to the UHR criteria, the Table8,9,12,13 also lists these additional variables shown to confer a high positive predictive value (PPV) of transition, alone or in combination with the UHR criterion. Thompson et al16 provide more detailed information on these later variables and their relative PPV.

What about treatment?
While discussion of the optimal treatment options for patients with prodromal symptoms of schizophrenia is beyond the scope of this article, early interventions can focus on preventing the biological, psychological, and social disruption that results from such symptoms. Establishing a therapeutic alliance with the patient while they retain insight and engaging supportive family members is a key starting point. Case management, cognitive-behavioral or supportive therapy, and treatment of comorbid mood, anxiety, or substance use disorders are helpful. There is no clear consensus on the utility of pharmacotherapy in the prodromal stage of psychosis. While scales and structured interviews can guide assessment, clinical judgment is the key driver of the appropriateness of initiating pharmacologic treatment to address symptoms. Because up to two-thirds of patients who satisfy UHR criteria do not go on to develop schizophrenia,16 clinicians should be thoughtful about the risks and benefits of antipsychotics.
Schizophrenia is characterized by psychotic symptoms that typically follow a prodromal period of premonitory signs and symptoms that appear before the manifestation of the full-blown syndrome. Signs and symptoms during the prodromal phase are subsyndromal, which implies a lower degree of intensity, duration, or frequency than observed when the patient meets the full criteria for the syndrome. Early detection of prodromal symptoms can improve prognosis, but these subtle symptoms may go unrecognized.
In schizophrenia, a patient may exhibit prodromal signs and symptoms before the appearance of pathognomonic symptoms, such as delusions, hallucinations, and disorganization. The schizophrenia prodrome can be conceptualized as a period of prepsychotic disturbances depicting an alteration in the individual’s behavior and perception. Prodromal symptoms can last from weeks to years before the psychotic illness clinically manifests.1 The prodromal symptom cluster typically becomes evident during adolescence and young adulthood.2
In the mid-1990s, investigators tried to identify a “putative prodrome” for psychosis. The term “at-risk mental state” (ARMS) for psychosis is based on retrospective reports of prodromal symptoms in first-episode psychosis. Over the next 2 decades, scales such as the Comprehensive Assessment of ARMS (CAARMS)3 and the Structured Interview for Prodromal Syndrome4 were designed to enhance the objectivity and diagnostic accuracy of the ARMS. These scales have reasonable interrater reliability.5
Researchers also have attempted to stage the severity of ARMS.6 Key symptom group predictors were studied to determine which individual symptoms or cluster of symptoms are most associated with poor outcomes and progression to psychosis. Raballo et al7 found the severity of the CAARMS disorganization dimension was the strongest predictor of transition to frank psychosis. Other research suggests that approximately one-third of ARMS patients transition to psychosis within 3 years, another one-third have persistent attenuated psychotic symptoms, and the remaining one-third experience symptom remission.8,9
Despite multiple studies and meta-analyses, current scales and clinical predictors continue to be imperfect.8 Efforts to identify specific biological markers and predictors of transition to clinical psychosis have not been successful for ARMS.10,11 The Table8,9,12,13 summarizes diagnostic criteria that have been developed to more clearly identify which ARMS patients face the highest imminent risk for transition to psychosis; these have been referred to as ultra high-risk (UHR) criteria.14 These UHR criteria depict 3 categories of clinical presentation believed to confer risk of transition to psychosis: attenuated psychotic symptoms, transient psychotic symptoms, and genetic predisposition. Subsequent research found that certain additional symptom variables, as well as combinations of specific symptom clusters, conferred increased risk and improved the positive predictive sensitivity to as high as 83%.15 In addition to the UHR criteria, the Table8,9,12,13 also lists these additional variables shown to confer a high positive predictive value (PPV) of transition, alone or in combination with the UHR criterion. Thompson et al16 provide more detailed information on these later variables and their relative PPV.

What about treatment?
While discussion of the optimal treatment options for patients with prodromal symptoms of schizophrenia is beyond the scope of this article, early interventions can focus on preventing the biological, psychological, and social disruption that results from such symptoms. Establishing a therapeutic alliance with the patient while they retain insight and engaging supportive family members is a key starting point. Case management, cognitive-behavioral or supportive therapy, and treatment of comorbid mood, anxiety, or substance use disorders are helpful. There is no clear consensus on the utility of pharmacotherapy in the prodromal stage of psychosis. While scales and structured interviews can guide assessment, clinical judgment is the key driver of the appropriateness of initiating pharmacologic treatment to address symptoms. Because up to two-thirds of patients who satisfy UHR criteria do not go on to develop schizophrenia,16 clinicians should be thoughtful about the risks and benefits of antipsychotics.
1. George M, Maheshwari S, Chandran S, et al. Understanding the schizophrenia prodrome. Indian J Psychiatry. 2017;59(4):505-509.
2. Yung AR, McGorry PD. The prodromal phase of first-episode psychosis: past and current conceptualizations. Schizophr Bull. 1996;22(2):353-370.
3. Yung AR, Yuen HP, McGorry PD, et al. Mapping the onset of psychosis: the Comprehensive Assessment of At-Risk Mental States. Aust N Z J Psychiatry. 2005;39(11-12):964-971.
4. Miller TJ, McGlashan TH, Rosen JL, et al. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: predictive validity, interrater reliability, and training to reliability. Schizophr Bull. 2003;29(4):703-715.
5. Loewy RL, Pearson R, Vinogradov S, et al. Psychosis risk screening with the Prodromal Questionnaire--brief version (PQ-B). Schizophr Res. 2011;129(1):42-46.
6. Nieman DH, McGorry PD. Detection and treatment of at-risk mental state for developing a first psychosis: making up the balance. Lancet Psychiatry. 2015;2(9):825-834.
7. Raballo A, Nelson B, Thompson A, et al. The comprehensive assessment of at-risk mental states: from mapping the onset to mapping the structure. Schizophr Res. 2011;127(1-3):107-114.
8. Fusar-Poli P, Bonoldi I, Yung AR, et al. Predicting psychosis: meta-analysis of transition outcomes in individuals at high clinical risk. Arch Gen Psychiatry. 2012;69(3):220-229.
9. Cannon TD. How schizophrenia develops: cognitive and brain mechanisms underlying onset of psychosis. Trends Cogn Sci. 2015;19(12):744-756.
10. Castle DJ. Is it appropriate to treat people at high-risk of psychosis before first onset? - no. Med J Aust. 2012;196(9):557.
11. Wood SJ, Reniers RL, Heinze K. Neuroimaging findings in the at-risk mental state: a review of recent literature. Can J Psychiatry. 2013;58(1):13-18.
12. Nelson B, Yung AR. Can clinicians predict psychosis in an ultra high risk group? Aust N Z J Psychiatry. 2010;44(7):625-630.
13. Schultze-Lutter F, Michel C, Schmidt SJ, et al. EPA guidance on the early detection of clinical high risk states of psychoses. Eur Psychiatry. 2015;30(3):405-416.
14. Yung AR, Phillips LJ, Yuen HP, et al. Risk factors for psychosis in an ultra high-risk group: psychopathology and clinical features. Schizophr Res. 2004;67(2-3):131-142.
15. Ruhrmann S, Schultze-Lutter F, Salokangas RK, et al. Prediction of psychosis in adolescents and young adults at high risk: results from the prospective European prediction of psychosis study. Arch Gen Psychiatry. 2010;67(3):241-251.
16. Thompson A, Marwaha S, Broome MR. At-risk mental state for psychosis: identification and current treatment approaches. BJPsych Advances. 2016;22(3):186-193.
1. George M, Maheshwari S, Chandran S, et al. Understanding the schizophrenia prodrome. Indian J Psychiatry. 2017;59(4):505-509.
2. Yung AR, McGorry PD. The prodromal phase of first-episode psychosis: past and current conceptualizations. Schizophr Bull. 1996;22(2):353-370.
3. Yung AR, Yuen HP, McGorry PD, et al. Mapping the onset of psychosis: the Comprehensive Assessment of At-Risk Mental States. Aust N Z J Psychiatry. 2005;39(11-12):964-971.
4. Miller TJ, McGlashan TH, Rosen JL, et al. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: predictive validity, interrater reliability, and training to reliability. Schizophr Bull. 2003;29(4):703-715.
5. Loewy RL, Pearson R, Vinogradov S, et al. Psychosis risk screening with the Prodromal Questionnaire--brief version (PQ-B). Schizophr Res. 2011;129(1):42-46.
6. Nieman DH, McGorry PD. Detection and treatment of at-risk mental state for developing a first psychosis: making up the balance. Lancet Psychiatry. 2015;2(9):825-834.
7. Raballo A, Nelson B, Thompson A, et al. The comprehensive assessment of at-risk mental states: from mapping the onset to mapping the structure. Schizophr Res. 2011;127(1-3):107-114.
8. Fusar-Poli P, Bonoldi I, Yung AR, et al. Predicting psychosis: meta-analysis of transition outcomes in individuals at high clinical risk. Arch Gen Psychiatry. 2012;69(3):220-229.
9. Cannon TD. How schizophrenia develops: cognitive and brain mechanisms underlying onset of psychosis. Trends Cogn Sci. 2015;19(12):744-756.
10. Castle DJ. Is it appropriate to treat people at high-risk of psychosis before first onset? - no. Med J Aust. 2012;196(9):557.
11. Wood SJ, Reniers RL, Heinze K. Neuroimaging findings in the at-risk mental state: a review of recent literature. Can J Psychiatry. 2013;58(1):13-18.
12. Nelson B, Yung AR. Can clinicians predict psychosis in an ultra high risk group? Aust N Z J Psychiatry. 2010;44(7):625-630.
13. Schultze-Lutter F, Michel C, Schmidt SJ, et al. EPA guidance on the early detection of clinical high risk states of psychoses. Eur Psychiatry. 2015;30(3):405-416.
14. Yung AR, Phillips LJ, Yuen HP, et al. Risk factors for psychosis in an ultra high-risk group: psychopathology and clinical features. Schizophr Res. 2004;67(2-3):131-142.
15. Ruhrmann S, Schultze-Lutter F, Salokangas RK, et al. Prediction of psychosis in adolescents and young adults at high risk: results from the prospective European prediction of psychosis study. Arch Gen Psychiatry. 2010;67(3):241-251.
16. Thompson A, Marwaha S, Broome MR. At-risk mental state for psychosis: identification and current treatment approaches. BJPsych Advances. 2016;22(3):186-193.
Generic stimulant shortage update: From bad to worse
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
I (MZP) just completed my first semester of medical school. An important lesson imparted in my coursework so far has been to remain a staunch advocate for patients. Yet compared to the rigors of medical school, over the past year it has been far more difficult to help patients locate generic Adderall. Physicians were already overburdened with administrative responsibilities stretching into burnout territory well before the shortage, and now this! Unlike paper prescriptions of old, which patients could take to any pharmacy, e-prescribing apps require selection of a specific pharmacy, and controlled substances such as stimulants require 2-factor authentication. But if the designated pharmacy does not have the medication in stock, the entire process must be repeated with an alternative pharmacy, long after the visit has concluded.
To add insult to injury, the generic stimulant shortage has grown even worse. As of February 2023, generic Adderall remained hard to find and generic Concerta was also in short supply. How did this happen? In 1985, Bulow et al¹ coined the game theory concept of “strategic substitutes,” where (for example) as beef becomes less readily accessible, consumers may switch to eating chicken as their protein. Unable to locate generic Adderall, many patients have turned to generic Concerta as a substitute psychostimulant to continue management of their attention-deficit/hyperactivity disorder.
In addition to the increase in demand, compounding the shortage is that one of the manufacturers of generic Concerta has discontinued production.² Branded methylphenidates and amphetamines, which are much more expensive than their generic counterparts, have remained in ample supply, but many insurers require trials of generics before considering coverage for more expensive brands.
Our approach to this situation
Each morning we call our local and chain pharmacies to take a census of their supply of generic stimulants. Some pharmacies refuse to release this information. Despite these census reports, we have found cases where patients have been turned away from pharmacies when they are not “regular customers,” while patients whom the pharmacies know retain access as “members.” Hence, a patient is unlikely to obtain these medications if their regular pharmacy is out of stock.
We want to share a workaround that has been effective. After unsuccessfully searching for generic stimulants at the patient’s regular pharmacy, I (RLP) write “dispense as written” for the closest branded version and file a prior authorization with the patient’s insurance company, noting “patient unable to trial any generic amphetamines or methylphenidates due to current nationwide shortage.” Even with the most difficult insurers, the response has been “a temporary 3-month authorization has been granted,” which is at least a small victory for our desperate patients and busy prescribers who are both struggling to negotiate a fragmented health care system.
1. Bulow JI, Geanakoplos JD, Klemperer PD. Multimarket oligopoly: strategic substitutes and complements. Journal of Political Economy. 1985;93(3):488-511. https://doi.org/10.1086/261312
2. US Food & Drug Administration. FDA Drug Shortages. Accessed January 7, 2023. https://www.accessdata.fda.gov/scripts/drugshortages/dsp_ActiveIngredientDetails.cfm?AI=Methylphenidate+Hydrochloride+Extended+Release+Tablets&st=d
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
I (MZP) just completed my first semester of medical school. An important lesson imparted in my coursework so far has been to remain a staunch advocate for patients. Yet compared to the rigors of medical school, over the past year it has been far more difficult to help patients locate generic Adderall. Physicians were already overburdened with administrative responsibilities stretching into burnout territory well before the shortage, and now this! Unlike paper prescriptions of old, which patients could take to any pharmacy, e-prescribing apps require selection of a specific pharmacy, and controlled substances such as stimulants require 2-factor authentication. But if the designated pharmacy does not have the medication in stock, the entire process must be repeated with an alternative pharmacy, long after the visit has concluded.
To add insult to injury, the generic stimulant shortage has grown even worse. As of February 2023, generic Adderall remained hard to find and generic Concerta was also in short supply. How did this happen? In 1985, Bulow et al¹ coined the game theory concept of “strategic substitutes,” where (for example) as beef becomes less readily accessible, consumers may switch to eating chicken as their protein. Unable to locate generic Adderall, many patients have turned to generic Concerta as a substitute psychostimulant to continue management of their attention-deficit/hyperactivity disorder.
In addition to the increase in demand, compounding the shortage is that one of the manufacturers of generic Concerta has discontinued production.² Branded methylphenidates and amphetamines, which are much more expensive than their generic counterparts, have remained in ample supply, but many insurers require trials of generics before considering coverage for more expensive brands.
Our approach to this situation
Each morning we call our local and chain pharmacies to take a census of their supply of generic stimulants. Some pharmacies refuse to release this information. Despite these census reports, we have found cases where patients have been turned away from pharmacies when they are not “regular customers,” while patients whom the pharmacies know retain access as “members.” Hence, a patient is unlikely to obtain these medications if their regular pharmacy is out of stock.
We want to share a workaround that has been effective. After unsuccessfully searching for generic stimulants at the patient’s regular pharmacy, I (RLP) write “dispense as written” for the closest branded version and file a prior authorization with the patient’s insurance company, noting “patient unable to trial any generic amphetamines or methylphenidates due to current nationwide shortage.” Even with the most difficult insurers, the response has been “a temporary 3-month authorization has been granted,” which is at least a small victory for our desperate patients and busy prescribers who are both struggling to negotiate a fragmented health care system.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
I (MZP) just completed my first semester of medical school. An important lesson imparted in my coursework so far has been to remain a staunch advocate for patients. Yet compared to the rigors of medical school, over the past year it has been far more difficult to help patients locate generic Adderall. Physicians were already overburdened with administrative responsibilities stretching into burnout territory well before the shortage, and now this! Unlike paper prescriptions of old, which patients could take to any pharmacy, e-prescribing apps require selection of a specific pharmacy, and controlled substances such as stimulants require 2-factor authentication. But if the designated pharmacy does not have the medication in stock, the entire process must be repeated with an alternative pharmacy, long after the visit has concluded.
To add insult to injury, the generic stimulant shortage has grown even worse. As of February 2023, generic Adderall remained hard to find and generic Concerta was also in short supply. How did this happen? In 1985, Bulow et al¹ coined the game theory concept of “strategic substitutes,” where (for example) as beef becomes less readily accessible, consumers may switch to eating chicken as their protein. Unable to locate generic Adderall, many patients have turned to generic Concerta as a substitute psychostimulant to continue management of their attention-deficit/hyperactivity disorder.
In addition to the increase in demand, compounding the shortage is that one of the manufacturers of generic Concerta has discontinued production.² Branded methylphenidates and amphetamines, which are much more expensive than their generic counterparts, have remained in ample supply, but many insurers require trials of generics before considering coverage for more expensive brands.
Our approach to this situation
Each morning we call our local and chain pharmacies to take a census of their supply of generic stimulants. Some pharmacies refuse to release this information. Despite these census reports, we have found cases where patients have been turned away from pharmacies when they are not “regular customers,” while patients whom the pharmacies know retain access as “members.” Hence, a patient is unlikely to obtain these medications if their regular pharmacy is out of stock.
We want to share a workaround that has been effective. After unsuccessfully searching for generic stimulants at the patient’s regular pharmacy, I (RLP) write “dispense as written” for the closest branded version and file a prior authorization with the patient’s insurance company, noting “patient unable to trial any generic amphetamines or methylphenidates due to current nationwide shortage.” Even with the most difficult insurers, the response has been “a temporary 3-month authorization has been granted,” which is at least a small victory for our desperate patients and busy prescribers who are both struggling to negotiate a fragmented health care system.
1. Bulow JI, Geanakoplos JD, Klemperer PD. Multimarket oligopoly: strategic substitutes and complements. Journal of Political Economy. 1985;93(3):488-511. https://doi.org/10.1086/261312
2. US Food & Drug Administration. FDA Drug Shortages. Accessed January 7, 2023. https://www.accessdata.fda.gov/scripts/drugshortages/dsp_ActiveIngredientDetails.cfm?AI=Methylphenidate+Hydrochloride+Extended+Release+Tablets&st=d
1. Bulow JI, Geanakoplos JD, Klemperer PD. Multimarket oligopoly: strategic substitutes and complements. Journal of Political Economy. 1985;93(3):488-511. https://doi.org/10.1086/261312
2. US Food & Drug Administration. FDA Drug Shortages. Accessed January 7, 2023. https://www.accessdata.fda.gov/scripts/drugshortages/dsp_ActiveIngredientDetails.cfm?AI=Methylphenidate+Hydrochloride+Extended+Release+Tablets&st=d
Ketamine plus psychotherapy ‘highly effective’ for PTSD
In a systematic review and meta-analysis of four studies investigating combined use of psychotherapy and ketamine for PTSD, results showed that all the studies showed a significant reduction in PTSD symptom scores.
Overall, the treatment was “highly effective, as seen by the significant improvements in symptoms on multiple measures,” Aaron E. Philipp-Muller, BScH, Centre for Neuroscience Studies, Queen’s University, Kingston, Ont., and colleagues write.
Furthermore, the study “demonstrates the potential feasibility of this treatment model and corroborates previous work,” the investigators write.
However, a limitation they note was that only 34 participants were included in the analysis.
The findings were published online in the Journal of Clinical Psychiatry.
Emerging treatment
Ketamine is an “emerging treatment for a number of psychopathologies, such as major depressive disorder and PTSD, with a higher response than other pharmacologic agents,” the researchers write.
It is hypothesized that ketamine rapidly facilitates long-term potentiation, “thereby allowing a patient to disengage from an established pattern of thought more readily,” they write.
However, ketamine has several drawbacks, including the fact that it brings only 1 week of relief for PTSD. Also, because it must be administered intravenously, it is “impractical for long-term weekly administration,” they note.
Pharmacologically enhanced psychotherapy is a potential way to prolong ketamine’s effects. Several prior studies have investigated this model using other psychedelic medications, with encouraging results.
The current investigators decided to review all literature to date on the subject of ketamine plus psychotherapy for the treatment of PTSD.
To be included, the study had to include patients diagnosed with PTSD, an intervention involving ketamine alongside any form of psychotherapy, and assessment of all patients before and after treatment using the Clinician-Administered PTSD Scale (CAPS) or the PTSD Checklist (PCL).
Four studies met inclusion criteria. Of these, two were of “moderate” quality and two were of “low” quality, based on the GRADE assessment. The studies encompassed a total of 34 patients with “diverse traumatic experiences” and included several types of ketamine administration protocols, including one used previously for treating depression and another used previously for chronic pain.
The psychotherapy modalities also differed between the studies. In two studies, patients received 12 sessions of trauma interventions using mindfulness-based extinction and reconsolidation therapy; in a third study, patients received 10 weekly sessions of prolonged exposure therapy; and in the fourth study, patients received five daily sessions of exposure therapy.
Across the studies, the psychotherapies were paired differently with ketamine administration, such as the number of ketamine administrations in conjunction with therapy.
Despite the differences in protocols, all the studies of ketamine plus psychotherapy showed a significant reduction in PTSD symptoms, with a pooled standardized mean difference (SMD) of –7.26 (95% CI, –12.28 to –2.25; P = .005) for the CAPS and a pooled SMD of –5.17 (95% CI, –7.99 to –2.35; P < .001) for the PCL.
The researchers acknowledge that the sample size was very small “due to the novelty of this research area.” This prompted the inclusion of nonrandomized studies that “lowered the quality of the evidence,” they note.
Nevertheless, “these preliminary findings indicate the potential of ketamine-assisted psychotherapy for PTSD,” the investigators write.
A promising avenue?
In a comment, Dan Iosifescu, MD, professor of psychiatry, New York University School of Medicine, called the combination of ketamine and psychotherapy in PTSD “a very promising treatment avenue.”
Dr. Iosifescu, who was not involved with the research, noted that “several PTSD-focused psychotherapies are ultimately very effective but very hard to tolerate for participants.” For example, prolonged exposure therapy has dropout rates as high as 50%.
In addition, ketamine has rapid but not sustained effects in PTSD, he said.
“So in theory, a course of ketamine could help PTSD patients improve rapidly and tolerate the psychotherapy, which could provide sustained benefits,” he added.
However, Dr. Iosifescu cautioned that the data supporting this “is very sparse for now.”
He also noted that the meta-analysis included only “four tiny studies” and had only 34 total participants. In addition, several of the studies had no comparison group and the study designs were all different – “both with respect to the administration of ketamine and to the paired PTSD psychotherapy.”
For this reason, “any conclusions are only a very preliminary suggestion that this may be a fruitful avenue,” he said.
Dr. Iosifescu added that additional studies on this topic are ongoing. The largest one at the Veterans Administration will hopefully include 100 participants and “will provide more reliable evidence for this important topic,” he said.
The study was indirectly supported by the Internal Faculty Grant from the department of psychiatry, Queen’s University. Dr. Iosifescu reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a systematic review and meta-analysis of four studies investigating combined use of psychotherapy and ketamine for PTSD, results showed that all the studies showed a significant reduction in PTSD symptom scores.
Overall, the treatment was “highly effective, as seen by the significant improvements in symptoms on multiple measures,” Aaron E. Philipp-Muller, BScH, Centre for Neuroscience Studies, Queen’s University, Kingston, Ont., and colleagues write.
Furthermore, the study “demonstrates the potential feasibility of this treatment model and corroborates previous work,” the investigators write.
However, a limitation they note was that only 34 participants were included in the analysis.
The findings were published online in the Journal of Clinical Psychiatry.
Emerging treatment
Ketamine is an “emerging treatment for a number of psychopathologies, such as major depressive disorder and PTSD, with a higher response than other pharmacologic agents,” the researchers write.
It is hypothesized that ketamine rapidly facilitates long-term potentiation, “thereby allowing a patient to disengage from an established pattern of thought more readily,” they write.
However, ketamine has several drawbacks, including the fact that it brings only 1 week of relief for PTSD. Also, because it must be administered intravenously, it is “impractical for long-term weekly administration,” they note.
Pharmacologically enhanced psychotherapy is a potential way to prolong ketamine’s effects. Several prior studies have investigated this model using other psychedelic medications, with encouraging results.
The current investigators decided to review all literature to date on the subject of ketamine plus psychotherapy for the treatment of PTSD.
To be included, the study had to include patients diagnosed with PTSD, an intervention involving ketamine alongside any form of psychotherapy, and assessment of all patients before and after treatment using the Clinician-Administered PTSD Scale (CAPS) or the PTSD Checklist (PCL).
Four studies met inclusion criteria. Of these, two were of “moderate” quality and two were of “low” quality, based on the GRADE assessment. The studies encompassed a total of 34 patients with “diverse traumatic experiences” and included several types of ketamine administration protocols, including one used previously for treating depression and another used previously for chronic pain.
The psychotherapy modalities also differed between the studies. In two studies, patients received 12 sessions of trauma interventions using mindfulness-based extinction and reconsolidation therapy; in a third study, patients received 10 weekly sessions of prolonged exposure therapy; and in the fourth study, patients received five daily sessions of exposure therapy.
Across the studies, the psychotherapies were paired differently with ketamine administration, such as the number of ketamine administrations in conjunction with therapy.
Despite the differences in protocols, all the studies of ketamine plus psychotherapy showed a significant reduction in PTSD symptoms, with a pooled standardized mean difference (SMD) of –7.26 (95% CI, –12.28 to –2.25; P = .005) for the CAPS and a pooled SMD of –5.17 (95% CI, –7.99 to –2.35; P < .001) for the PCL.
The researchers acknowledge that the sample size was very small “due to the novelty of this research area.” This prompted the inclusion of nonrandomized studies that “lowered the quality of the evidence,” they note.
Nevertheless, “these preliminary findings indicate the potential of ketamine-assisted psychotherapy for PTSD,” the investigators write.
A promising avenue?
In a comment, Dan Iosifescu, MD, professor of psychiatry, New York University School of Medicine, called the combination of ketamine and psychotherapy in PTSD “a very promising treatment avenue.”
Dr. Iosifescu, who was not involved with the research, noted that “several PTSD-focused psychotherapies are ultimately very effective but very hard to tolerate for participants.” For example, prolonged exposure therapy has dropout rates as high as 50%.
In addition, ketamine has rapid but not sustained effects in PTSD, he said.
“So in theory, a course of ketamine could help PTSD patients improve rapidly and tolerate the psychotherapy, which could provide sustained benefits,” he added.
However, Dr. Iosifescu cautioned that the data supporting this “is very sparse for now.”
He also noted that the meta-analysis included only “four tiny studies” and had only 34 total participants. In addition, several of the studies had no comparison group and the study designs were all different – “both with respect to the administration of ketamine and to the paired PTSD psychotherapy.”
For this reason, “any conclusions are only a very preliminary suggestion that this may be a fruitful avenue,” he said.
Dr. Iosifescu added that additional studies on this topic are ongoing. The largest one at the Veterans Administration will hopefully include 100 participants and “will provide more reliable evidence for this important topic,” he said.
The study was indirectly supported by the Internal Faculty Grant from the department of psychiatry, Queen’s University. Dr. Iosifescu reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a systematic review and meta-analysis of four studies investigating combined use of psychotherapy and ketamine for PTSD, results showed that all the studies showed a significant reduction in PTSD symptom scores.
Overall, the treatment was “highly effective, as seen by the significant improvements in symptoms on multiple measures,” Aaron E. Philipp-Muller, BScH, Centre for Neuroscience Studies, Queen’s University, Kingston, Ont., and colleagues write.
Furthermore, the study “demonstrates the potential feasibility of this treatment model and corroborates previous work,” the investigators write.
However, a limitation they note was that only 34 participants were included in the analysis.
The findings were published online in the Journal of Clinical Psychiatry.
Emerging treatment
Ketamine is an “emerging treatment for a number of psychopathologies, such as major depressive disorder and PTSD, with a higher response than other pharmacologic agents,” the researchers write.
It is hypothesized that ketamine rapidly facilitates long-term potentiation, “thereby allowing a patient to disengage from an established pattern of thought more readily,” they write.
However, ketamine has several drawbacks, including the fact that it brings only 1 week of relief for PTSD. Also, because it must be administered intravenously, it is “impractical for long-term weekly administration,” they note.
Pharmacologically enhanced psychotherapy is a potential way to prolong ketamine’s effects. Several prior studies have investigated this model using other psychedelic medications, with encouraging results.
The current investigators decided to review all literature to date on the subject of ketamine plus psychotherapy for the treatment of PTSD.
To be included, the study had to include patients diagnosed with PTSD, an intervention involving ketamine alongside any form of psychotherapy, and assessment of all patients before and after treatment using the Clinician-Administered PTSD Scale (CAPS) or the PTSD Checklist (PCL).
Four studies met inclusion criteria. Of these, two were of “moderate” quality and two were of “low” quality, based on the GRADE assessment. The studies encompassed a total of 34 patients with “diverse traumatic experiences” and included several types of ketamine administration protocols, including one used previously for treating depression and another used previously for chronic pain.
The psychotherapy modalities also differed between the studies. In two studies, patients received 12 sessions of trauma interventions using mindfulness-based extinction and reconsolidation therapy; in a third study, patients received 10 weekly sessions of prolonged exposure therapy; and in the fourth study, patients received five daily sessions of exposure therapy.
Across the studies, the psychotherapies were paired differently with ketamine administration, such as the number of ketamine administrations in conjunction with therapy.
Despite the differences in protocols, all the studies of ketamine plus psychotherapy showed a significant reduction in PTSD symptoms, with a pooled standardized mean difference (SMD) of –7.26 (95% CI, –12.28 to –2.25; P = .005) for the CAPS and a pooled SMD of –5.17 (95% CI, –7.99 to –2.35; P < .001) for the PCL.
The researchers acknowledge that the sample size was very small “due to the novelty of this research area.” This prompted the inclusion of nonrandomized studies that “lowered the quality of the evidence,” they note.
Nevertheless, “these preliminary findings indicate the potential of ketamine-assisted psychotherapy for PTSD,” the investigators write.
A promising avenue?
In a comment, Dan Iosifescu, MD, professor of psychiatry, New York University School of Medicine, called the combination of ketamine and psychotherapy in PTSD “a very promising treatment avenue.”
Dr. Iosifescu, who was not involved with the research, noted that “several PTSD-focused psychotherapies are ultimately very effective but very hard to tolerate for participants.” For example, prolonged exposure therapy has dropout rates as high as 50%.
In addition, ketamine has rapid but not sustained effects in PTSD, he said.
“So in theory, a course of ketamine could help PTSD patients improve rapidly and tolerate the psychotherapy, which could provide sustained benefits,” he added.
However, Dr. Iosifescu cautioned that the data supporting this “is very sparse for now.”
He also noted that the meta-analysis included only “four tiny studies” and had only 34 total participants. In addition, several of the studies had no comparison group and the study designs were all different – “both with respect to the administration of ketamine and to the paired PTSD psychotherapy.”
For this reason, “any conclusions are only a very preliminary suggestion that this may be a fruitful avenue,” he said.
Dr. Iosifescu added that additional studies on this topic are ongoing. The largest one at the Veterans Administration will hopefully include 100 participants and “will provide more reliable evidence for this important topic,” he said.
The study was indirectly supported by the Internal Faculty Grant from the department of psychiatry, Queen’s University. Dr. Iosifescu reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL PSYCHIATRY
Higher dementia risk in women explained?
a study suggests.
Prior research has found a higher lifetime dementia risk in women, and one explanation cited has been that women tend to live longer than men.
However, this new analysis of data from nearly 30,000 people in 18 countries found almost no evidence of sex differences in most known risk factors for dementia, including age.
The risk of dementia among women was significantly higher in poorer countries, pointing to economic disadvantages as a possible explanation.
“In general, we found that the greater dementia risk found in women compared to men was more pronounced in poorer countries, which points to the need for greater efforts to narrow the gaps in health disparities between women and men in these countries,” lead investigator Jessica Gong, MSc, a doctoral student at the George Institute for Global Health, Newtown, Australia, told this news organization. “It is likely that socioeconomic factors are potentially more important than biological factors when assessing dementia risk.”
The findings were published online in Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association.
Global data
Most previous studies that examined sex differences in dementia risk were conducted in high-income countries, Ms. Gong noted, leaving a gap in the literature on risk in low- and middle-income countries.
To address this issue, researchers conducted an individual participant meta-analysis of 21 studies from the Cohort Studies of Memory in an International Consortium. Data analysis included information on 29,850 people from 18 countries on six continents. None of the participants had dementia at baseline, and the average age was 71.6 years.
Over a median of 4.6 years, incident dementia was reported in 2,089 people, 66% of whom were women.
Overall, women had higher dementia risk (hazard ratio, 1.12; 95% confidence interval, 1.02-1.23) than men, but the rates were highest in low- to middle-income economies (HR, 1.73; P = .03).
Dementia risk in women was higher than in men in 14 countries. Risk was highest in Nigeria, where dementia risk was more than double in women (aHR, 2.11; 95% CI, 1.46-3.04), and lowest in Brazil, where risk was 46% lower in women than in men (aHR, 0.54; 95% CI, 0.29-1.00).
In the United States, dementia risk was 7% higher in women than men (aHR, 1.07; 0.73-1.57).
Similar risk factors
In both women and men, older age, diabetes, depression, hearing impairment, and apo E–epsilon 4 carriage were associated with a greater risk of dementia, and more years of education, higher hip circumference, current alcohol use (vs. never), and high physical activity (vs. none to minimal) were associated with a lower risk of dementia.
Among all these risk factors, sex differences were only significant for longer education and former alcohol use, with both demonstrating a stronger association in men than women.
Global dementia rates are expected to triple over the next 25 years unless steps are taken to reduce risk factors. A 2020 report found that dementia risk could be reduced by addressing 12 modifiable risk factors, including obesity, air pollution, diabetes, social isolation, and hypertension. All of these risk factors are more common in low- to middle-income countries, Ms. Gong noted.
“These findings justify ongoing efforts to support programs to improve sex and gender equity in brain health, particularly in underrepresented and underserved populations, in turn to narrow the gaps within and between country,” Ms. Gong said.
Understanding the puzzle
Commenting on the findings for Medscape Medical News, Heather Snyder, PhD, Alzheimer’s Association vice president of medical and scientific relations, said the findings add to the body of work about sex differences in dementia risk.
“This is an interesting study looking at risk factors for dementia and suggests that, while some risk factors are more pronounced in men than in women, women may be more at risk of progressing to dementia,” Dr. Snyder said. “The findings outline the importance of understanding how the underlying biology, particularly biology that differs in males and females, may be contributing to risk.”
Data on the country and geographical variations highlighted in the study also point to a potential risk influencer, she said.
“Studying geography-specific risk factors is important because it helps us understand the ‘why’ behind geographic differences in dementia risk,” Dr. Snyder said. “This type of collaboration among countries and researchers is essential for us to understand these puzzle pieces.”
Funding for the study was provided by the U.K. Medical Research Council Skills Development Fellowship, Australian National Health and Medical Research Council Investigator Grant, National Institute on Aging, among others. See the original article for full funding sources. Ms. Gong reported no relevant financial conflicts. Dr. Snyder is employed by the Alzheimer’s Association.
A version of this article originally appeared on Medscape.com.
a study suggests.
Prior research has found a higher lifetime dementia risk in women, and one explanation cited has been that women tend to live longer than men.
However, this new analysis of data from nearly 30,000 people in 18 countries found almost no evidence of sex differences in most known risk factors for dementia, including age.
The risk of dementia among women was significantly higher in poorer countries, pointing to economic disadvantages as a possible explanation.
“In general, we found that the greater dementia risk found in women compared to men was more pronounced in poorer countries, which points to the need for greater efforts to narrow the gaps in health disparities between women and men in these countries,” lead investigator Jessica Gong, MSc, a doctoral student at the George Institute for Global Health, Newtown, Australia, told this news organization. “It is likely that socioeconomic factors are potentially more important than biological factors when assessing dementia risk.”
The findings were published online in Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association.
Global data
Most previous studies that examined sex differences in dementia risk were conducted in high-income countries, Ms. Gong noted, leaving a gap in the literature on risk in low- and middle-income countries.
To address this issue, researchers conducted an individual participant meta-analysis of 21 studies from the Cohort Studies of Memory in an International Consortium. Data analysis included information on 29,850 people from 18 countries on six continents. None of the participants had dementia at baseline, and the average age was 71.6 years.
Over a median of 4.6 years, incident dementia was reported in 2,089 people, 66% of whom were women.
Overall, women had higher dementia risk (hazard ratio, 1.12; 95% confidence interval, 1.02-1.23) than men, but the rates were highest in low- to middle-income economies (HR, 1.73; P = .03).
Dementia risk in women was higher than in men in 14 countries. Risk was highest in Nigeria, where dementia risk was more than double in women (aHR, 2.11; 95% CI, 1.46-3.04), and lowest in Brazil, where risk was 46% lower in women than in men (aHR, 0.54; 95% CI, 0.29-1.00).
In the United States, dementia risk was 7% higher in women than men (aHR, 1.07; 0.73-1.57).
Similar risk factors
In both women and men, older age, diabetes, depression, hearing impairment, and apo E–epsilon 4 carriage were associated with a greater risk of dementia, and more years of education, higher hip circumference, current alcohol use (vs. never), and high physical activity (vs. none to minimal) were associated with a lower risk of dementia.
Among all these risk factors, sex differences were only significant for longer education and former alcohol use, with both demonstrating a stronger association in men than women.
Global dementia rates are expected to triple over the next 25 years unless steps are taken to reduce risk factors. A 2020 report found that dementia risk could be reduced by addressing 12 modifiable risk factors, including obesity, air pollution, diabetes, social isolation, and hypertension. All of these risk factors are more common in low- to middle-income countries, Ms. Gong noted.
“These findings justify ongoing efforts to support programs to improve sex and gender equity in brain health, particularly in underrepresented and underserved populations, in turn to narrow the gaps within and between country,” Ms. Gong said.
Understanding the puzzle
Commenting on the findings for Medscape Medical News, Heather Snyder, PhD, Alzheimer’s Association vice president of medical and scientific relations, said the findings add to the body of work about sex differences in dementia risk.
“This is an interesting study looking at risk factors for dementia and suggests that, while some risk factors are more pronounced in men than in women, women may be more at risk of progressing to dementia,” Dr. Snyder said. “The findings outline the importance of understanding how the underlying biology, particularly biology that differs in males and females, may be contributing to risk.”
Data on the country and geographical variations highlighted in the study also point to a potential risk influencer, she said.
“Studying geography-specific risk factors is important because it helps us understand the ‘why’ behind geographic differences in dementia risk,” Dr. Snyder said. “This type of collaboration among countries and researchers is essential for us to understand these puzzle pieces.”
Funding for the study was provided by the U.K. Medical Research Council Skills Development Fellowship, Australian National Health and Medical Research Council Investigator Grant, National Institute on Aging, among others. See the original article for full funding sources. Ms. Gong reported no relevant financial conflicts. Dr. Snyder is employed by the Alzheimer’s Association.
A version of this article originally appeared on Medscape.com.
a study suggests.
Prior research has found a higher lifetime dementia risk in women, and one explanation cited has been that women tend to live longer than men.
However, this new analysis of data from nearly 30,000 people in 18 countries found almost no evidence of sex differences in most known risk factors for dementia, including age.
The risk of dementia among women was significantly higher in poorer countries, pointing to economic disadvantages as a possible explanation.
“In general, we found that the greater dementia risk found in women compared to men was more pronounced in poorer countries, which points to the need for greater efforts to narrow the gaps in health disparities between women and men in these countries,” lead investigator Jessica Gong, MSc, a doctoral student at the George Institute for Global Health, Newtown, Australia, told this news organization. “It is likely that socioeconomic factors are potentially more important than biological factors when assessing dementia risk.”
The findings were published online in Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association.
Global data
Most previous studies that examined sex differences in dementia risk were conducted in high-income countries, Ms. Gong noted, leaving a gap in the literature on risk in low- and middle-income countries.
To address this issue, researchers conducted an individual participant meta-analysis of 21 studies from the Cohort Studies of Memory in an International Consortium. Data analysis included information on 29,850 people from 18 countries on six continents. None of the participants had dementia at baseline, and the average age was 71.6 years.
Over a median of 4.6 years, incident dementia was reported in 2,089 people, 66% of whom were women.
Overall, women had higher dementia risk (hazard ratio, 1.12; 95% confidence interval, 1.02-1.23) than men, but the rates were highest in low- to middle-income economies (HR, 1.73; P = .03).
Dementia risk in women was higher than in men in 14 countries. Risk was highest in Nigeria, where dementia risk was more than double in women (aHR, 2.11; 95% CI, 1.46-3.04), and lowest in Brazil, where risk was 46% lower in women than in men (aHR, 0.54; 95% CI, 0.29-1.00).
In the United States, dementia risk was 7% higher in women than men (aHR, 1.07; 0.73-1.57).
Similar risk factors
In both women and men, older age, diabetes, depression, hearing impairment, and apo E–epsilon 4 carriage were associated with a greater risk of dementia, and more years of education, higher hip circumference, current alcohol use (vs. never), and high physical activity (vs. none to minimal) were associated with a lower risk of dementia.
Among all these risk factors, sex differences were only significant for longer education and former alcohol use, with both demonstrating a stronger association in men than women.
Global dementia rates are expected to triple over the next 25 years unless steps are taken to reduce risk factors. A 2020 report found that dementia risk could be reduced by addressing 12 modifiable risk factors, including obesity, air pollution, diabetes, social isolation, and hypertension. All of these risk factors are more common in low- to middle-income countries, Ms. Gong noted.
“These findings justify ongoing efforts to support programs to improve sex and gender equity in brain health, particularly in underrepresented and underserved populations, in turn to narrow the gaps within and between country,” Ms. Gong said.
Understanding the puzzle
Commenting on the findings for Medscape Medical News, Heather Snyder, PhD, Alzheimer’s Association vice president of medical and scientific relations, said the findings add to the body of work about sex differences in dementia risk.
“This is an interesting study looking at risk factors for dementia and suggests that, while some risk factors are more pronounced in men than in women, women may be more at risk of progressing to dementia,” Dr. Snyder said. “The findings outline the importance of understanding how the underlying biology, particularly biology that differs in males and females, may be contributing to risk.”
Data on the country and geographical variations highlighted in the study also point to a potential risk influencer, she said.
“Studying geography-specific risk factors is important because it helps us understand the ‘why’ behind geographic differences in dementia risk,” Dr. Snyder said. “This type of collaboration among countries and researchers is essential for us to understand these puzzle pieces.”
Funding for the study was provided by the U.K. Medical Research Council Skills Development Fellowship, Australian National Health and Medical Research Council Investigator Grant, National Institute on Aging, among others. See the original article for full funding sources. Ms. Gong reported no relevant financial conflicts. Dr. Snyder is employed by the Alzheimer’s Association.
A version of this article originally appeared on Medscape.com.
FROM ALZHEIMER’S & DEMENTIA
Myths about smoking, diet, alcohol, and cancer persist
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
Physician pleads guilty to 52 counts in opioid scheme
Jeffrey B. Sutton, DO, a neuromuscular medicine specialist, pled guilty on January 30 in federal court to 31 counts of illegally prescribing opioids and other controlled substances, 1 count of illegally distributing controlled substances, and 20 counts of health care fraud.
Prosecutors said Dr. Sutton admitted that he ignored warnings from prescription drug management organizations, insurers, and state authorities that he was prescribing excessively high dosages of opioids.
Dr. Sutton also admitted to ignoring patient requests to lower dosages and that he also ignored signs that patients were selling prescribed medications or otherwise engaging in illicit activity, including violations of a “pain management agreement” that he required them to sign.
The fraud counts pertained to Dr. Sutton billing Medicare, Medicaid, and other insurers for medically unnecessary visits that he required of patients so that he could prescribe inappropriate or unnecessary opioids.
In the charging document shared with this news organization, prosecutors said Dr. Sutton had sex with at least three patients, including during office visits and outside of the office. Occasionally, the physician would give opioids or other controlled substances – often benzodiazepines – to these patients, without a prescription or valid medical need.
Dr. Sutton escalated the dosage for one of those patients, even as the subjective pain score did not improve and when the patient’s urine tests showed the presence of THC and buprenorphine, but not any of the prescribed medications.
Another patient came to Dr. Sutton in 2007 with a warning that she had a history of “narcotic-seeking” behavior and diagnoses of depression, anxiety, paranoid schizophrenia, and obsessive-compulsive disorder.
The patient was hospitalized in 2018 for complications from benzodiazepine use (prescribed by Dr. Sutton). She weighed 80 pounds at the time. Dr. Sutton continued to prescribe benzodiazepines and extreme doses of opioids – in excess of 2,000 morphine equivalent dose – “despite recognizing and documenting repeated instances of noncompliance with treatment for psychiatric conditions, and despite the known contraindications of long-term opioid use for patients with these mental illnesses,” according to the charging document.
Dr. Sutton continued to prescribe opioids despite two hospitalizations for overdoses, more than 20 failed urine drug screens that showed presence of illicit drugs such as cocaine, and documented excessive use of alprazolam (Xanax) and methadone.
The physician surrendered his Drug Enforcement Administration Certificate of Registration of Controlled Substances Privileges in February 2022 “as an indication of your good faith in desiring to remedy any incorrect or unlawful practices on your part,” according to a letter to Dr. Sutton from the State Medical Board of Ohio. In that September 2022 letter, the Board notified Dr. Sutton of its intention to possibly suspend or revoke his license.
Dr. Sutton did not request a hearing, and the Board permanently revoked his medical license on January 16.
The court will sentence Dr. Sutton on May 23, according to a report by WFMJ.
A version of this article originally appeared on Medscape.com.
Jeffrey B. Sutton, DO, a neuromuscular medicine specialist, pled guilty on January 30 in federal court to 31 counts of illegally prescribing opioids and other controlled substances, 1 count of illegally distributing controlled substances, and 20 counts of health care fraud.
Prosecutors said Dr. Sutton admitted that he ignored warnings from prescription drug management organizations, insurers, and state authorities that he was prescribing excessively high dosages of opioids.
Dr. Sutton also admitted to ignoring patient requests to lower dosages and that he also ignored signs that patients were selling prescribed medications or otherwise engaging in illicit activity, including violations of a “pain management agreement” that he required them to sign.
The fraud counts pertained to Dr. Sutton billing Medicare, Medicaid, and other insurers for medically unnecessary visits that he required of patients so that he could prescribe inappropriate or unnecessary opioids.
In the charging document shared with this news organization, prosecutors said Dr. Sutton had sex with at least three patients, including during office visits and outside of the office. Occasionally, the physician would give opioids or other controlled substances – often benzodiazepines – to these patients, without a prescription or valid medical need.
Dr. Sutton escalated the dosage for one of those patients, even as the subjective pain score did not improve and when the patient’s urine tests showed the presence of THC and buprenorphine, but not any of the prescribed medications.
Another patient came to Dr. Sutton in 2007 with a warning that she had a history of “narcotic-seeking” behavior and diagnoses of depression, anxiety, paranoid schizophrenia, and obsessive-compulsive disorder.
The patient was hospitalized in 2018 for complications from benzodiazepine use (prescribed by Dr. Sutton). She weighed 80 pounds at the time. Dr. Sutton continued to prescribe benzodiazepines and extreme doses of opioids – in excess of 2,000 morphine equivalent dose – “despite recognizing and documenting repeated instances of noncompliance with treatment for psychiatric conditions, and despite the known contraindications of long-term opioid use for patients with these mental illnesses,” according to the charging document.
Dr. Sutton continued to prescribe opioids despite two hospitalizations for overdoses, more than 20 failed urine drug screens that showed presence of illicit drugs such as cocaine, and documented excessive use of alprazolam (Xanax) and methadone.
The physician surrendered his Drug Enforcement Administration Certificate of Registration of Controlled Substances Privileges in February 2022 “as an indication of your good faith in desiring to remedy any incorrect or unlawful practices on your part,” according to a letter to Dr. Sutton from the State Medical Board of Ohio. In that September 2022 letter, the Board notified Dr. Sutton of its intention to possibly suspend or revoke his license.
Dr. Sutton did not request a hearing, and the Board permanently revoked his medical license on January 16.
The court will sentence Dr. Sutton on May 23, according to a report by WFMJ.
A version of this article originally appeared on Medscape.com.
Jeffrey B. Sutton, DO, a neuromuscular medicine specialist, pled guilty on January 30 in federal court to 31 counts of illegally prescribing opioids and other controlled substances, 1 count of illegally distributing controlled substances, and 20 counts of health care fraud.
Prosecutors said Dr. Sutton admitted that he ignored warnings from prescription drug management organizations, insurers, and state authorities that he was prescribing excessively high dosages of opioids.
Dr. Sutton also admitted to ignoring patient requests to lower dosages and that he also ignored signs that patients were selling prescribed medications or otherwise engaging in illicit activity, including violations of a “pain management agreement” that he required them to sign.
The fraud counts pertained to Dr. Sutton billing Medicare, Medicaid, and other insurers for medically unnecessary visits that he required of patients so that he could prescribe inappropriate or unnecessary opioids.
In the charging document shared with this news organization, prosecutors said Dr. Sutton had sex with at least three patients, including during office visits and outside of the office. Occasionally, the physician would give opioids or other controlled substances – often benzodiazepines – to these patients, without a prescription or valid medical need.
Dr. Sutton escalated the dosage for one of those patients, even as the subjective pain score did not improve and when the patient’s urine tests showed the presence of THC and buprenorphine, but not any of the prescribed medications.
Another patient came to Dr. Sutton in 2007 with a warning that she had a history of “narcotic-seeking” behavior and diagnoses of depression, anxiety, paranoid schizophrenia, and obsessive-compulsive disorder.
The patient was hospitalized in 2018 for complications from benzodiazepine use (prescribed by Dr. Sutton). She weighed 80 pounds at the time. Dr. Sutton continued to prescribe benzodiazepines and extreme doses of opioids – in excess of 2,000 morphine equivalent dose – “despite recognizing and documenting repeated instances of noncompliance with treatment for psychiatric conditions, and despite the known contraindications of long-term opioid use for patients with these mental illnesses,” according to the charging document.
Dr. Sutton continued to prescribe opioids despite two hospitalizations for overdoses, more than 20 failed urine drug screens that showed presence of illicit drugs such as cocaine, and documented excessive use of alprazolam (Xanax) and methadone.
The physician surrendered his Drug Enforcement Administration Certificate of Registration of Controlled Substances Privileges in February 2022 “as an indication of your good faith in desiring to remedy any incorrect or unlawful practices on your part,” according to a letter to Dr. Sutton from the State Medical Board of Ohio. In that September 2022 letter, the Board notified Dr. Sutton of its intention to possibly suspend or revoke his license.
Dr. Sutton did not request a hearing, and the Board permanently revoked his medical license on January 16.
The court will sentence Dr. Sutton on May 23, according to a report by WFMJ.
A version of this article originally appeared on Medscape.com.





