User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Back to school: How pediatricians can help LGBTQ youth
September every year means one thing to students across the country: Summer break is over, and it is time to go back to school. For LGBTQ youth, this can be both a blessing and a curse. Schools can be a refuge from being stuck at home with unsupportive family, but it also can mean returning to hallways full of harassment from other students and/or staff. Groups such as a gender-sexuality alliance (GSA) or a chapter of the Gay, Lesbian, and Straight Education Network (GLSEN) can provide a safe space for these students at school. Pediatricians can play an important role in ensuring that their patients know about access to these resources.
Gender-sexuality alliances, or gay-straight alliances as they have been more commonly known, have been around since the late 1980s. The first one was founded at Concord Academy in Massachusetts in 1988 by a straight student who was upset at how her gay classmates were being treated. Today’s GSAs continue this mission to create a welcoming environment for students of all gender identities and sexual orientations to gather, increase awareness on their campus of LGBTQ issues, and make the school environment safer for all students. According to the GSA network, there are over 4,000 active GSAs today in the United States located in 40 states.1
GLSEN was founded in 1990 initially as a network of gay and lesbian educators who wanted to create safer spaces in schools for LGBTQ students. Over the last 30 years, GLSEN continues to support this mission but has expanded into research and advocacy as well. There are currently 43 chapters of GLSEN in 30 states.2 GLSEN sponsors a number of national events throughout the year to raise awareness of LGBTQ issues in schools, including No Name Calling Week and the Day of Silence. Many chapters provide mentoring to local GSAs and volunteering as a mentor can be a great way for pediatricians to become involved in their local schools.
You may be asking yourself, why are GSAs important? According to GLSEN’s 2017 National School Climate Survey, nearly 35% of LGBTQ students missed at least 1 day of school in the previous month because of feeling unsafe, and nearly 57% of students reported hearing homophobic remarks from teachers and staff at their school.3 Around 10% of LGBTQ students reported being physically assaulted based on their sexual orientation and/or gender identity. Those LGBTQ students who experienced discrimination based on their sexual orientation and/or gender identity were more likely to have lower grade point averages and were more likely to be disciplined than those students who had not experienced discrimination.3 The cumulative effect of these negative experiences at school lead a sizable portion of affected students to drop out of school and possibly not pursue postsecondary education. This then leads to decreased job opportunities or career advancement, which could then lead to unemployment or low-wage jobs. Creating safe spaces for education to take place can have a lasting effect on the lives of LGBTQ students.
The 53% of students who reported having a GSA at their school in the National School Climate survey were less likely to report hearing negative comments about LGBTQ students, were less likely to miss school, experienced lower levels of victimization, and reported higher levels of supportive teachers and staff. All of these factors taken together ensure that LGBTQ students are more likely to complete their high school education. Russell B. Toomey, PhD, and colleagues were able to show that LGBTQ students with a perceived effective GSA were two times more likely than those without an effective GSA to attain a college education.4 Research also has shown that the presence of a GSA can have a beneficial impact on reducing bullying in general for all students, whether they identify as LGBTQ or not.5
What active steps can a pediatrician take to support their LGBTQ students? First, If the families run into trouble from the school, have your social workers help them connect with legal resources, as many court cases have established precedent that public schools cannot have a blanket ban on GSAs solely because they focus on LGBTQ issues. Second, if your patient has a GSA at their school and seems to be struggling with his/her sexual orientation and/or gender identity, encourage that student to consider attending their GSA so that they are able to spend time with other students like themselves. Third, as many schools will be starting virtually this year, you can provide your LGBTQ patients with a list of local online groups that students can participate in virtually if their school’s GSA is not meeting (see my LGBTQ Youth Consult column entitled, “Resources for LGBTQ youth during challenging times” at mdedge.com/pediatrics for a few ideas).* Lastly, be an active advocate in your own local school district for the inclusion of comprehensive nondiscrimination policies and the presence of GSAs for students. These small steps can go a long way to helping your LGBTQ patients thrive and succeed in school.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. Dr. Cooper has no relevant financial disclosures. Email him at [email protected].
References
1. gsanetwork.org/mission-vision-history/.
2. www.glsen.org/find_chapter?field_chapter_state_target_id=All.
3. live-glsen-website.pantheonsite.io/sites/default/files/2019-10/GLSEN-2017-National-School-Climate-Survey-NSCS-Full-Report.pdf.
4. Appl Dev Sci. 2011 Nov 7;15(4):175-85.
5.www.usnews.com/news/articles/2016-08-04/gay-straight-alliances-in-schools-pay-off-for-all-students-study-finds.
*This article was updated 8/17/2020.
September every year means one thing to students across the country: Summer break is over, and it is time to go back to school. For LGBTQ youth, this can be both a blessing and a curse. Schools can be a refuge from being stuck at home with unsupportive family, but it also can mean returning to hallways full of harassment from other students and/or staff. Groups such as a gender-sexuality alliance (GSA) or a chapter of the Gay, Lesbian, and Straight Education Network (GLSEN) can provide a safe space for these students at school. Pediatricians can play an important role in ensuring that their patients know about access to these resources.
Gender-sexuality alliances, or gay-straight alliances as they have been more commonly known, have been around since the late 1980s. The first one was founded at Concord Academy in Massachusetts in 1988 by a straight student who was upset at how her gay classmates were being treated. Today’s GSAs continue this mission to create a welcoming environment for students of all gender identities and sexual orientations to gather, increase awareness on their campus of LGBTQ issues, and make the school environment safer for all students. According to the GSA network, there are over 4,000 active GSAs today in the United States located in 40 states.1
GLSEN was founded in 1990 initially as a network of gay and lesbian educators who wanted to create safer spaces in schools for LGBTQ students. Over the last 30 years, GLSEN continues to support this mission but has expanded into research and advocacy as well. There are currently 43 chapters of GLSEN in 30 states.2 GLSEN sponsors a number of national events throughout the year to raise awareness of LGBTQ issues in schools, including No Name Calling Week and the Day of Silence. Many chapters provide mentoring to local GSAs and volunteering as a mentor can be a great way for pediatricians to become involved in their local schools.
You may be asking yourself, why are GSAs important? According to GLSEN’s 2017 National School Climate Survey, nearly 35% of LGBTQ students missed at least 1 day of school in the previous month because of feeling unsafe, and nearly 57% of students reported hearing homophobic remarks from teachers and staff at their school.3 Around 10% of LGBTQ students reported being physically assaulted based on their sexual orientation and/or gender identity. Those LGBTQ students who experienced discrimination based on their sexual orientation and/or gender identity were more likely to have lower grade point averages and were more likely to be disciplined than those students who had not experienced discrimination.3 The cumulative effect of these negative experiences at school lead a sizable portion of affected students to drop out of school and possibly not pursue postsecondary education. This then leads to decreased job opportunities or career advancement, which could then lead to unemployment or low-wage jobs. Creating safe spaces for education to take place can have a lasting effect on the lives of LGBTQ students.
The 53% of students who reported having a GSA at their school in the National School Climate survey were less likely to report hearing negative comments about LGBTQ students, were less likely to miss school, experienced lower levels of victimization, and reported higher levels of supportive teachers and staff. All of these factors taken together ensure that LGBTQ students are more likely to complete their high school education. Russell B. Toomey, PhD, and colleagues were able to show that LGBTQ students with a perceived effective GSA were two times more likely than those without an effective GSA to attain a college education.4 Research also has shown that the presence of a GSA can have a beneficial impact on reducing bullying in general for all students, whether they identify as LGBTQ or not.5
What active steps can a pediatrician take to support their LGBTQ students? First, If the families run into trouble from the school, have your social workers help them connect with legal resources, as many court cases have established precedent that public schools cannot have a blanket ban on GSAs solely because they focus on LGBTQ issues. Second, if your patient has a GSA at their school and seems to be struggling with his/her sexual orientation and/or gender identity, encourage that student to consider attending their GSA so that they are able to spend time with other students like themselves. Third, as many schools will be starting virtually this year, you can provide your LGBTQ patients with a list of local online groups that students can participate in virtually if their school’s GSA is not meeting (see my LGBTQ Youth Consult column entitled, “Resources for LGBTQ youth during challenging times” at mdedge.com/pediatrics for a few ideas).* Lastly, be an active advocate in your own local school district for the inclusion of comprehensive nondiscrimination policies and the presence of GSAs for students. These small steps can go a long way to helping your LGBTQ patients thrive and succeed in school.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. Dr. Cooper has no relevant financial disclosures. Email him at [email protected].
References
1. gsanetwork.org/mission-vision-history/.
2. www.glsen.org/find_chapter?field_chapter_state_target_id=All.
3. live-glsen-website.pantheonsite.io/sites/default/files/2019-10/GLSEN-2017-National-School-Climate-Survey-NSCS-Full-Report.pdf.
4. Appl Dev Sci. 2011 Nov 7;15(4):175-85.
5.www.usnews.com/news/articles/2016-08-04/gay-straight-alliances-in-schools-pay-off-for-all-students-study-finds.
*This article was updated 8/17/2020.
September every year means one thing to students across the country: Summer break is over, and it is time to go back to school. For LGBTQ youth, this can be both a blessing and a curse. Schools can be a refuge from being stuck at home with unsupportive family, but it also can mean returning to hallways full of harassment from other students and/or staff. Groups such as a gender-sexuality alliance (GSA) or a chapter of the Gay, Lesbian, and Straight Education Network (GLSEN) can provide a safe space for these students at school. Pediatricians can play an important role in ensuring that their patients know about access to these resources.
Gender-sexuality alliances, or gay-straight alliances as they have been more commonly known, have been around since the late 1980s. The first one was founded at Concord Academy in Massachusetts in 1988 by a straight student who was upset at how her gay classmates were being treated. Today’s GSAs continue this mission to create a welcoming environment for students of all gender identities and sexual orientations to gather, increase awareness on their campus of LGBTQ issues, and make the school environment safer for all students. According to the GSA network, there are over 4,000 active GSAs today in the United States located in 40 states.1
GLSEN was founded in 1990 initially as a network of gay and lesbian educators who wanted to create safer spaces in schools for LGBTQ students. Over the last 30 years, GLSEN continues to support this mission but has expanded into research and advocacy as well. There are currently 43 chapters of GLSEN in 30 states.2 GLSEN sponsors a number of national events throughout the year to raise awareness of LGBTQ issues in schools, including No Name Calling Week and the Day of Silence. Many chapters provide mentoring to local GSAs and volunteering as a mentor can be a great way for pediatricians to become involved in their local schools.
You may be asking yourself, why are GSAs important? According to GLSEN’s 2017 National School Climate Survey, nearly 35% of LGBTQ students missed at least 1 day of school in the previous month because of feeling unsafe, and nearly 57% of students reported hearing homophobic remarks from teachers and staff at their school.3 Around 10% of LGBTQ students reported being physically assaulted based on their sexual orientation and/or gender identity. Those LGBTQ students who experienced discrimination based on their sexual orientation and/or gender identity were more likely to have lower grade point averages and were more likely to be disciplined than those students who had not experienced discrimination.3 The cumulative effect of these negative experiences at school lead a sizable portion of affected students to drop out of school and possibly not pursue postsecondary education. This then leads to decreased job opportunities or career advancement, which could then lead to unemployment or low-wage jobs. Creating safe spaces for education to take place can have a lasting effect on the lives of LGBTQ students.
The 53% of students who reported having a GSA at their school in the National School Climate survey were less likely to report hearing negative comments about LGBTQ students, were less likely to miss school, experienced lower levels of victimization, and reported higher levels of supportive teachers and staff. All of these factors taken together ensure that LGBTQ students are more likely to complete their high school education. Russell B. Toomey, PhD, and colleagues were able to show that LGBTQ students with a perceived effective GSA were two times more likely than those without an effective GSA to attain a college education.4 Research also has shown that the presence of a GSA can have a beneficial impact on reducing bullying in general for all students, whether they identify as LGBTQ or not.5
What active steps can a pediatrician take to support their LGBTQ students? First, If the families run into trouble from the school, have your social workers help them connect with legal resources, as many court cases have established precedent that public schools cannot have a blanket ban on GSAs solely because they focus on LGBTQ issues. Second, if your patient has a GSA at their school and seems to be struggling with his/her sexual orientation and/or gender identity, encourage that student to consider attending their GSA so that they are able to spend time with other students like themselves. Third, as many schools will be starting virtually this year, you can provide your LGBTQ patients with a list of local online groups that students can participate in virtually if their school’s GSA is not meeting (see my LGBTQ Youth Consult column entitled, “Resources for LGBTQ youth during challenging times” at mdedge.com/pediatrics for a few ideas).* Lastly, be an active advocate in your own local school district for the inclusion of comprehensive nondiscrimination policies and the presence of GSAs for students. These small steps can go a long way to helping your LGBTQ patients thrive and succeed in school.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas. Dr. Cooper has no relevant financial disclosures. Email him at [email protected].
References
1. gsanetwork.org/mission-vision-history/.
2. www.glsen.org/find_chapter?field_chapter_state_target_id=All.
3. live-glsen-website.pantheonsite.io/sites/default/files/2019-10/GLSEN-2017-National-School-Climate-Survey-NSCS-Full-Report.pdf.
4. Appl Dev Sci. 2011 Nov 7;15(4):175-85.
5.www.usnews.com/news/articles/2016-08-04/gay-straight-alliances-in-schools-pay-off-for-all-students-study-finds.
*This article was updated 8/17/2020.
Long-lasting COVID-19 symptoms: Patients want answers
Q&A with Dr. Sachin Gupta
For some patients, a bout of COVID-19 may not be over after hospital discharge, acute symptoms subside, or a couple of tests for SARS-CoV-2 come back negative. Those who have reached these milestones of conquering the disease may find that their recovery journey has only begun. Debilitating symptoms such as fatigue, headache, and dyspnea may linger for weeks or longer. Patients with persistent symptoms, often referred to as “long haulers” in reference to the duration of their recovery, are looking for answers about their condition and when their COVID-19 illness will finally resolve.
Long-haul patients organize
What started as an accumulation of anecdotal evidence in social media, blogs, and the mainstream press about slow recovery and long-lasting symptoms of COVID-19 is now the focus of clinical trials in the population of recovering patients. Projects such as the COVID Symptom Study, initiated by the Massachusetts General Hospital, Boston; the Harvard School of Public Health, Boston; King’s College London; and Stanford (Calif.) University, are collecting data on symptoms from millions of patients and will eventually contribute to a better understanding of prolonged recovery.
Patients looking for answers have created groups on social media such as Facebook to exchange information about their experiences (e.g., Survivor Corps, COVID-19 Support Group, COVID-19 Recovered Survivors). Recovering patients have created patient-led research organizations (Body Politic COVID-19 Support Group) to explore persistent symptoms and begin to create data for research.
Some data on lingering symptoms
A small study of 143 previously hospitalized, recovering patients in Italy found that 87.4% of the cohort had at least one persistent symptom 2 months or longer after initial onset and at more than a month after discharge. In this sample, only 5% had been intubated. (JAMA 2020 Jul 9. doi: 10.1001/jama.2020.12603).
One study found that even patients who have had relatively mild symptoms and were not hospitalized can have persistent symptoms. The Centers for Disease Control and Prevention conducted a survey of adults who tested positive for the positive reverse transcription–polymerase chain reaction test for SARS-CoV-2 and found that, among the 292 respondents, 35% were still feeling the impact of the disease 2-3 weeks after testing. Fatigue (71%), cough (61%), and headache (61%) were the most commonly reported symptoms. The survey found that delayed recovery was evident in nearly a quarter of 18- to 34-year-olds and in a third of 35- to 49-year-olds who were not sick enough to require hospitalization (MMWR. 2020 Jul 24. doi: 10.15585/mmwr.mm6930e1).
Sachin Gupta, MD, FCCP, ATSF, a pulmonologist and member of the CHEST Physician editorial advisory board, has treated patients with COVID-19 and shared some of his thoughts on the problem of prolonged symptoms of COVID-19.
Q: Should clinicians expect to see COVID-19 patients who have symptoms persisting weeks after they are diagnosed?
Dr. Gupta: I think clinicians, especially in primary care, are already seeing many patients with lingering symptoms, both respiratory and nonrespiratory related, and debility. A few patients here in the San Francisco Bay Area that I have spoken with 4-6 weeks out from their acute illness have complained of persisting, though improving, fatigue and cough. Early studies are confirming this as a topical issue. There may be other long-lasting sequelae of COVID-19 beyond the common mild lingering symptoms. It will also be important to consider (and get more data on) to what degree asymptomatic patients develop some degree of mild inflammatory and subsequent fibrotic changes in organs like the lungs and heart
Q: How does the recovery phase of COVID-19 compare with recovery from severe influenza or ARDS?
Dr. Gupta: Most prior influenza and acute respiratory distress syndrome (ARDS) studies have provided initial follow-up at 3 months and beyond, so technically speaking, it is a little difficult to compare the symptomatology patterns in the JAMA study of 2 months on follow-up. Nevertheless, the key takeaway is that, even though few patients in the study had ARDS requiring intubation (severe disease), many patients with milder disease had significant lingering symptoms (55% with three or more symptoms) at 2 months.
This fits logically with the premise, which we have some limited data on with ARDS (N Engl J Med. 2003;348:683-93. doi: 10.1056/NEJMoa022450) and severe influenza infection survivors (Nature Sci Rep. 2017;7:17275. doi: 10.1038/s41598-017-17497-6) that varying degrees of the inflammation cascade triggered by certain viruses can lead to changes in important patient-reported outcomes, and objective measures such as pulmonary function over the long term.
Q: What can you do for patients with lingering symptoms of COVID-19 or what can you tell them about their symptoms?
Dr. Gupta: For many patients, there is fear, given the novel nature of the virus/pandemic, that their symptoms may persist long term. Acknowledgment of their symptoms is validating and important for us to recognize as we learn more about the virus. As we are finding, many patients are going online to find answers, after sometimes feeling rushed or dismissed initially in the clinical setting.
In my experience, the bar is fairly high for most patients to reach out to their physicians with complaints of lingering symptoms after acute infection. For the ones who do reach out, they tend to have either a greater constellation of symptoms or higher severity of one or two key symptoms. After assessing and, when appropriate, ruling out secondary infections or newly developed conditions, I shift toward symptom management. I encourage such patients to build up slowly. I suggest they work first on their activities of daily living (bathing, grooming), then their instrumental activities of daily living (cooking, cleaning, checking the mail), and then to engage, based on their tolerance of symptoms, to light purposeful exercise. There are many online resources for at-home exercise activities that I recommend to patients who are more debilitated; some larger centers are beginning to offer some forms of telepulmonary rehab.
Based on what we know about other causes of viral pneumonitis and ARDS, I ask such symptomatic patients to expect a slow, gradual, and in most cases a complete recovery, and depending on the individual case, I recommend pulmonary function tests and imaging that may be helpful to track that progress.
I remind myself, and patients, that our understanding may change as we learn more over time. Checking in at set intervals, even if not in person but through a phone call, can go a long way in a setting where we do not have a specific therapy, other than gradual exercise training, to help these patients recover faster. Reassurance and encouragement are vital for patients who are struggling with the lingering burden of disease and who may find it difficult to return to work or function as usual at home. The final point is to be mindful of our patient’s mental health and, where our reassurance is not enough, to consider appropriate mental health referrals.
Q: Can you handle this kind of problem with telemedicine or which patients with lingering symptoms need to come into the office – or failing that, the ED?
Dr. Gupta: Telemedicine in the outpatient setting provides a helpful tool to assess and manage patients, in my experience, with limited and straightforward complaints. Its scope is limited diagnostically (assessing symptoms and signs) as is its reach (ability to connect with elderly, disabled, or patients without/limited telemedicine access). In many instances, telemedicine limits our ability to connect with patients emotionally and build trust. Many patients who have gone through the acute illness that we see in pulmonary clinic on follow-up are older in age, and for many, video visits are not a practical solution. Telemedicine visits can sometimes present challenges for me as well in terms of thoroughly conveying lifestyle and symptom management strategies. Health literacy is typically easier to gauge and address in person.
For patients with any degree of enduring dyspnea, more so in the acute phase, I recommend home pulse oximetry for monitoring their oxygen saturation if it is financially and technically feasible for them to obtain one. Sending a patient to the ED is an option of last resort, but one that is necessary in some cases. I expect patients with lingering symptoms to tell me that symptoms may be persisting, hopefully gradually improving, and not getting worse. If post–COVID-19 symptoms such as fever, dyspnea, fatigue, or lightheadedness are new or worsening, particularly rapidly, the safest and best option I advise patients is to go to the ED for further assessment and testing. Postviral bacterial pneumonia is something we should consider, and there is some potential for aspergillosis as well.
Q: Do you have any concerns about patients with asthma, chronic obstructive pulmonary disease, or other pulmonary issues having lingering symptoms that may mask exacerbations or may cause exacerbation of their disease?
Dr. Gupta: So far, patients with chronic lung conditions do not appear to have not been disproportionately affected by the pandemic in terms of absolute numbers or percentage wise compared to the general public. I think that sheltering in place has been readily followed by many of these patients, and in addition, I assume better adherence to their maintenance therapies has likely helped. The very few cases of patients with underlying chronic obstructive pulmonary disease and interstitial lung disease that I have seen have fared very poorly when they were diagnosed with COVID-19 in the hospital. There are emerging data about short-term outcomes from severe COVID-19 infection in patients with interstitial lung disease in Europe (medRxiv. 2020 Jul 17. doi: 10.1101/2020.07.15.20152967), and from physicians treating pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension (Ann Am Thorac Soc. 2020 Jul 29. doi: 10.1513/AnnalsATS.202005-521OC). But so far, little has been published on the outcomes of mild disease in these patients with chronic lung disease.
Q: It’s still early days to know the significance of lingering symptoms. But at what point do you begin to consider the possibility of some kind of relapse? And what is your next move if the symptoms get worse?
Dr. Gupta: COVID-19 recurrence, whether because of reinfection or relapse, is a potential concern but not one that is very commonly seen so far in my purview. Generally, symptoms of post–COVID-19 infection that are lingering trend toward getting better, even if slowly. If post–COVID-19 infection symptoms are progressing (particularly if rapidly), that would be a strong indication to evaluate that patient in the ED (less likely in clinic), reswab them for SARS-CoV-2, and obtain further testing such as blood work and imaging. A significant challenge from a research perspective will be determining if coinfection with another virus is playing a role as we move closer to the fall season.
Q&A with Dr. Sachin Gupta
Q&A with Dr. Sachin Gupta
For some patients, a bout of COVID-19 may not be over after hospital discharge, acute symptoms subside, or a couple of tests for SARS-CoV-2 come back negative. Those who have reached these milestones of conquering the disease may find that their recovery journey has only begun. Debilitating symptoms such as fatigue, headache, and dyspnea may linger for weeks or longer. Patients with persistent symptoms, often referred to as “long haulers” in reference to the duration of their recovery, are looking for answers about their condition and when their COVID-19 illness will finally resolve.
Long-haul patients organize
What started as an accumulation of anecdotal evidence in social media, blogs, and the mainstream press about slow recovery and long-lasting symptoms of COVID-19 is now the focus of clinical trials in the population of recovering patients. Projects such as the COVID Symptom Study, initiated by the Massachusetts General Hospital, Boston; the Harvard School of Public Health, Boston; King’s College London; and Stanford (Calif.) University, are collecting data on symptoms from millions of patients and will eventually contribute to a better understanding of prolonged recovery.
Patients looking for answers have created groups on social media such as Facebook to exchange information about their experiences (e.g., Survivor Corps, COVID-19 Support Group, COVID-19 Recovered Survivors). Recovering patients have created patient-led research organizations (Body Politic COVID-19 Support Group) to explore persistent symptoms and begin to create data for research.
Some data on lingering symptoms
A small study of 143 previously hospitalized, recovering patients in Italy found that 87.4% of the cohort had at least one persistent symptom 2 months or longer after initial onset and at more than a month after discharge. In this sample, only 5% had been intubated. (JAMA 2020 Jul 9. doi: 10.1001/jama.2020.12603).
One study found that even patients who have had relatively mild symptoms and were not hospitalized can have persistent symptoms. The Centers for Disease Control and Prevention conducted a survey of adults who tested positive for the positive reverse transcription–polymerase chain reaction test for SARS-CoV-2 and found that, among the 292 respondents, 35% were still feeling the impact of the disease 2-3 weeks after testing. Fatigue (71%), cough (61%), and headache (61%) were the most commonly reported symptoms. The survey found that delayed recovery was evident in nearly a quarter of 18- to 34-year-olds and in a third of 35- to 49-year-olds who were not sick enough to require hospitalization (MMWR. 2020 Jul 24. doi: 10.15585/mmwr.mm6930e1).
Sachin Gupta, MD, FCCP, ATSF, a pulmonologist and member of the CHEST Physician editorial advisory board, has treated patients with COVID-19 and shared some of his thoughts on the problem of prolonged symptoms of COVID-19.
Q: Should clinicians expect to see COVID-19 patients who have symptoms persisting weeks after they are diagnosed?
Dr. Gupta: I think clinicians, especially in primary care, are already seeing many patients with lingering symptoms, both respiratory and nonrespiratory related, and debility. A few patients here in the San Francisco Bay Area that I have spoken with 4-6 weeks out from their acute illness have complained of persisting, though improving, fatigue and cough. Early studies are confirming this as a topical issue. There may be other long-lasting sequelae of COVID-19 beyond the common mild lingering symptoms. It will also be important to consider (and get more data on) to what degree asymptomatic patients develop some degree of mild inflammatory and subsequent fibrotic changes in organs like the lungs and heart
Q: How does the recovery phase of COVID-19 compare with recovery from severe influenza or ARDS?
Dr. Gupta: Most prior influenza and acute respiratory distress syndrome (ARDS) studies have provided initial follow-up at 3 months and beyond, so technically speaking, it is a little difficult to compare the symptomatology patterns in the JAMA study of 2 months on follow-up. Nevertheless, the key takeaway is that, even though few patients in the study had ARDS requiring intubation (severe disease), many patients with milder disease had significant lingering symptoms (55% with three or more symptoms) at 2 months.
This fits logically with the premise, which we have some limited data on with ARDS (N Engl J Med. 2003;348:683-93. doi: 10.1056/NEJMoa022450) and severe influenza infection survivors (Nature Sci Rep. 2017;7:17275. doi: 10.1038/s41598-017-17497-6) that varying degrees of the inflammation cascade triggered by certain viruses can lead to changes in important patient-reported outcomes, and objective measures such as pulmonary function over the long term.
Q: What can you do for patients with lingering symptoms of COVID-19 or what can you tell them about their symptoms?
Dr. Gupta: For many patients, there is fear, given the novel nature of the virus/pandemic, that their symptoms may persist long term. Acknowledgment of their symptoms is validating and important for us to recognize as we learn more about the virus. As we are finding, many patients are going online to find answers, after sometimes feeling rushed or dismissed initially in the clinical setting.
In my experience, the bar is fairly high for most patients to reach out to their physicians with complaints of lingering symptoms after acute infection. For the ones who do reach out, they tend to have either a greater constellation of symptoms or higher severity of one or two key symptoms. After assessing and, when appropriate, ruling out secondary infections or newly developed conditions, I shift toward symptom management. I encourage such patients to build up slowly. I suggest they work first on their activities of daily living (bathing, grooming), then their instrumental activities of daily living (cooking, cleaning, checking the mail), and then to engage, based on their tolerance of symptoms, to light purposeful exercise. There are many online resources for at-home exercise activities that I recommend to patients who are more debilitated; some larger centers are beginning to offer some forms of telepulmonary rehab.
Based on what we know about other causes of viral pneumonitis and ARDS, I ask such symptomatic patients to expect a slow, gradual, and in most cases a complete recovery, and depending on the individual case, I recommend pulmonary function tests and imaging that may be helpful to track that progress.
I remind myself, and patients, that our understanding may change as we learn more over time. Checking in at set intervals, even if not in person but through a phone call, can go a long way in a setting where we do not have a specific therapy, other than gradual exercise training, to help these patients recover faster. Reassurance and encouragement are vital for patients who are struggling with the lingering burden of disease and who may find it difficult to return to work or function as usual at home. The final point is to be mindful of our patient’s mental health and, where our reassurance is not enough, to consider appropriate mental health referrals.
Q: Can you handle this kind of problem with telemedicine or which patients with lingering symptoms need to come into the office – or failing that, the ED?
Dr. Gupta: Telemedicine in the outpatient setting provides a helpful tool to assess and manage patients, in my experience, with limited and straightforward complaints. Its scope is limited diagnostically (assessing symptoms and signs) as is its reach (ability to connect with elderly, disabled, or patients without/limited telemedicine access). In many instances, telemedicine limits our ability to connect with patients emotionally and build trust. Many patients who have gone through the acute illness that we see in pulmonary clinic on follow-up are older in age, and for many, video visits are not a practical solution. Telemedicine visits can sometimes present challenges for me as well in terms of thoroughly conveying lifestyle and symptom management strategies. Health literacy is typically easier to gauge and address in person.
For patients with any degree of enduring dyspnea, more so in the acute phase, I recommend home pulse oximetry for monitoring their oxygen saturation if it is financially and technically feasible for them to obtain one. Sending a patient to the ED is an option of last resort, but one that is necessary in some cases. I expect patients with lingering symptoms to tell me that symptoms may be persisting, hopefully gradually improving, and not getting worse. If post–COVID-19 symptoms such as fever, dyspnea, fatigue, or lightheadedness are new or worsening, particularly rapidly, the safest and best option I advise patients is to go to the ED for further assessment and testing. Postviral bacterial pneumonia is something we should consider, and there is some potential for aspergillosis as well.
Q: Do you have any concerns about patients with asthma, chronic obstructive pulmonary disease, or other pulmonary issues having lingering symptoms that may mask exacerbations or may cause exacerbation of their disease?
Dr. Gupta: So far, patients with chronic lung conditions do not appear to have not been disproportionately affected by the pandemic in terms of absolute numbers or percentage wise compared to the general public. I think that sheltering in place has been readily followed by many of these patients, and in addition, I assume better adherence to their maintenance therapies has likely helped. The very few cases of patients with underlying chronic obstructive pulmonary disease and interstitial lung disease that I have seen have fared very poorly when they were diagnosed with COVID-19 in the hospital. There are emerging data about short-term outcomes from severe COVID-19 infection in patients with interstitial lung disease in Europe (medRxiv. 2020 Jul 17. doi: 10.1101/2020.07.15.20152967), and from physicians treating pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension (Ann Am Thorac Soc. 2020 Jul 29. doi: 10.1513/AnnalsATS.202005-521OC). But so far, little has been published on the outcomes of mild disease in these patients with chronic lung disease.
Q: It’s still early days to know the significance of lingering symptoms. But at what point do you begin to consider the possibility of some kind of relapse? And what is your next move if the symptoms get worse?
Dr. Gupta: COVID-19 recurrence, whether because of reinfection or relapse, is a potential concern but not one that is very commonly seen so far in my purview. Generally, symptoms of post–COVID-19 infection that are lingering trend toward getting better, even if slowly. If post–COVID-19 infection symptoms are progressing (particularly if rapidly), that would be a strong indication to evaluate that patient in the ED (less likely in clinic), reswab them for SARS-CoV-2, and obtain further testing such as blood work and imaging. A significant challenge from a research perspective will be determining if coinfection with another virus is playing a role as we move closer to the fall season.
For some patients, a bout of COVID-19 may not be over after hospital discharge, acute symptoms subside, or a couple of tests for SARS-CoV-2 come back negative. Those who have reached these milestones of conquering the disease may find that their recovery journey has only begun. Debilitating symptoms such as fatigue, headache, and dyspnea may linger for weeks or longer. Patients with persistent symptoms, often referred to as “long haulers” in reference to the duration of their recovery, are looking for answers about their condition and when their COVID-19 illness will finally resolve.
Long-haul patients organize
What started as an accumulation of anecdotal evidence in social media, blogs, and the mainstream press about slow recovery and long-lasting symptoms of COVID-19 is now the focus of clinical trials in the population of recovering patients. Projects such as the COVID Symptom Study, initiated by the Massachusetts General Hospital, Boston; the Harvard School of Public Health, Boston; King’s College London; and Stanford (Calif.) University, are collecting data on symptoms from millions of patients and will eventually contribute to a better understanding of prolonged recovery.
Patients looking for answers have created groups on social media such as Facebook to exchange information about their experiences (e.g., Survivor Corps, COVID-19 Support Group, COVID-19 Recovered Survivors). Recovering patients have created patient-led research organizations (Body Politic COVID-19 Support Group) to explore persistent symptoms and begin to create data for research.
Some data on lingering symptoms
A small study of 143 previously hospitalized, recovering patients in Italy found that 87.4% of the cohort had at least one persistent symptom 2 months or longer after initial onset and at more than a month after discharge. In this sample, only 5% had been intubated. (JAMA 2020 Jul 9. doi: 10.1001/jama.2020.12603).
One study found that even patients who have had relatively mild symptoms and were not hospitalized can have persistent symptoms. The Centers for Disease Control and Prevention conducted a survey of adults who tested positive for the positive reverse transcription–polymerase chain reaction test for SARS-CoV-2 and found that, among the 292 respondents, 35% were still feeling the impact of the disease 2-3 weeks after testing. Fatigue (71%), cough (61%), and headache (61%) were the most commonly reported symptoms. The survey found that delayed recovery was evident in nearly a quarter of 18- to 34-year-olds and in a third of 35- to 49-year-olds who were not sick enough to require hospitalization (MMWR. 2020 Jul 24. doi: 10.15585/mmwr.mm6930e1).
Sachin Gupta, MD, FCCP, ATSF, a pulmonologist and member of the CHEST Physician editorial advisory board, has treated patients with COVID-19 and shared some of his thoughts on the problem of prolonged symptoms of COVID-19.
Q: Should clinicians expect to see COVID-19 patients who have symptoms persisting weeks after they are diagnosed?
Dr. Gupta: I think clinicians, especially in primary care, are already seeing many patients with lingering symptoms, both respiratory and nonrespiratory related, and debility. A few patients here in the San Francisco Bay Area that I have spoken with 4-6 weeks out from their acute illness have complained of persisting, though improving, fatigue and cough. Early studies are confirming this as a topical issue. There may be other long-lasting sequelae of COVID-19 beyond the common mild lingering symptoms. It will also be important to consider (and get more data on) to what degree asymptomatic patients develop some degree of mild inflammatory and subsequent fibrotic changes in organs like the lungs and heart
Q: How does the recovery phase of COVID-19 compare with recovery from severe influenza or ARDS?
Dr. Gupta: Most prior influenza and acute respiratory distress syndrome (ARDS) studies have provided initial follow-up at 3 months and beyond, so technically speaking, it is a little difficult to compare the symptomatology patterns in the JAMA study of 2 months on follow-up. Nevertheless, the key takeaway is that, even though few patients in the study had ARDS requiring intubation (severe disease), many patients with milder disease had significant lingering symptoms (55% with three or more symptoms) at 2 months.
This fits logically with the premise, which we have some limited data on with ARDS (N Engl J Med. 2003;348:683-93. doi: 10.1056/NEJMoa022450) and severe influenza infection survivors (Nature Sci Rep. 2017;7:17275. doi: 10.1038/s41598-017-17497-6) that varying degrees of the inflammation cascade triggered by certain viruses can lead to changes in important patient-reported outcomes, and objective measures such as pulmonary function over the long term.
Q: What can you do for patients with lingering symptoms of COVID-19 or what can you tell them about their symptoms?
Dr. Gupta: For many patients, there is fear, given the novel nature of the virus/pandemic, that their symptoms may persist long term. Acknowledgment of their symptoms is validating and important for us to recognize as we learn more about the virus. As we are finding, many patients are going online to find answers, after sometimes feeling rushed or dismissed initially in the clinical setting.
In my experience, the bar is fairly high for most patients to reach out to their physicians with complaints of lingering symptoms after acute infection. For the ones who do reach out, they tend to have either a greater constellation of symptoms or higher severity of one or two key symptoms. After assessing and, when appropriate, ruling out secondary infections or newly developed conditions, I shift toward symptom management. I encourage such patients to build up slowly. I suggest they work first on their activities of daily living (bathing, grooming), then their instrumental activities of daily living (cooking, cleaning, checking the mail), and then to engage, based on their tolerance of symptoms, to light purposeful exercise. There are many online resources for at-home exercise activities that I recommend to patients who are more debilitated; some larger centers are beginning to offer some forms of telepulmonary rehab.
Based on what we know about other causes of viral pneumonitis and ARDS, I ask such symptomatic patients to expect a slow, gradual, and in most cases a complete recovery, and depending on the individual case, I recommend pulmonary function tests and imaging that may be helpful to track that progress.
I remind myself, and patients, that our understanding may change as we learn more over time. Checking in at set intervals, even if not in person but through a phone call, can go a long way in a setting where we do not have a specific therapy, other than gradual exercise training, to help these patients recover faster. Reassurance and encouragement are vital for patients who are struggling with the lingering burden of disease and who may find it difficult to return to work or function as usual at home. The final point is to be mindful of our patient’s mental health and, where our reassurance is not enough, to consider appropriate mental health referrals.
Q: Can you handle this kind of problem with telemedicine or which patients with lingering symptoms need to come into the office – or failing that, the ED?
Dr. Gupta: Telemedicine in the outpatient setting provides a helpful tool to assess and manage patients, in my experience, with limited and straightforward complaints. Its scope is limited diagnostically (assessing symptoms and signs) as is its reach (ability to connect with elderly, disabled, or patients without/limited telemedicine access). In many instances, telemedicine limits our ability to connect with patients emotionally and build trust. Many patients who have gone through the acute illness that we see in pulmonary clinic on follow-up are older in age, and for many, video visits are not a practical solution. Telemedicine visits can sometimes present challenges for me as well in terms of thoroughly conveying lifestyle and symptom management strategies. Health literacy is typically easier to gauge and address in person.
For patients with any degree of enduring dyspnea, more so in the acute phase, I recommend home pulse oximetry for monitoring their oxygen saturation if it is financially and technically feasible for them to obtain one. Sending a patient to the ED is an option of last resort, but one that is necessary in some cases. I expect patients with lingering symptoms to tell me that symptoms may be persisting, hopefully gradually improving, and not getting worse. If post–COVID-19 symptoms such as fever, dyspnea, fatigue, or lightheadedness are new or worsening, particularly rapidly, the safest and best option I advise patients is to go to the ED for further assessment and testing. Postviral bacterial pneumonia is something we should consider, and there is some potential for aspergillosis as well.
Q: Do you have any concerns about patients with asthma, chronic obstructive pulmonary disease, or other pulmonary issues having lingering symptoms that may mask exacerbations or may cause exacerbation of their disease?
Dr. Gupta: So far, patients with chronic lung conditions do not appear to have not been disproportionately affected by the pandemic in terms of absolute numbers or percentage wise compared to the general public. I think that sheltering in place has been readily followed by many of these patients, and in addition, I assume better adherence to their maintenance therapies has likely helped. The very few cases of patients with underlying chronic obstructive pulmonary disease and interstitial lung disease that I have seen have fared very poorly when they were diagnosed with COVID-19 in the hospital. There are emerging data about short-term outcomes from severe COVID-19 infection in patients with interstitial lung disease in Europe (medRxiv. 2020 Jul 17. doi: 10.1101/2020.07.15.20152967), and from physicians treating pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension (Ann Am Thorac Soc. 2020 Jul 29. doi: 10.1513/AnnalsATS.202005-521OC). But so far, little has been published on the outcomes of mild disease in these patients with chronic lung disease.
Q: It’s still early days to know the significance of lingering symptoms. But at what point do you begin to consider the possibility of some kind of relapse? And what is your next move if the symptoms get worse?
Dr. Gupta: COVID-19 recurrence, whether because of reinfection or relapse, is a potential concern but not one that is very commonly seen so far in my purview. Generally, symptoms of post–COVID-19 infection that are lingering trend toward getting better, even if slowly. If post–COVID-19 infection symptoms are progressing (particularly if rapidly), that would be a strong indication to evaluate that patient in the ED (less likely in clinic), reswab them for SARS-CoV-2, and obtain further testing such as blood work and imaging. A significant challenge from a research perspective will be determining if coinfection with another virus is playing a role as we move closer to the fall season.
FDA approves first oral treatment for spinal muscular atrophy
This marks the first approval of an oral therapy for the rare and devastating condition.
Risdiplam, marketed by Roche and PTC Therapeutics, provides “an important treatment option for patients with SMA, following the approval of the first treatment for this devastating disease less than 4 years ago,” Billy Dunn, MD, director of the Office of Neuroscience in the Center for Drug Evaluation and Research at the FDA, said in a release from the agency.
The approval was based on the results from two trials. In the open-label FIREFISH study of infantile-onset SMA, 7 (41%) of the 17 participants (mean baseline age, 6.7 months) were able to sit independently for more than 5 seconds after 12 months of treatment with risdiplam. This was a “meaningful difference from the natural progression of the disease because all untreated infants with infantile-onset SMA cannot sit independently,” the FDA noted. In addition, 81% of the participants were alive after 23 or more months of treatment – and without need of permanent ventilation.
The second study was the randomized controlled trial known as SUNFISH and included 180 patients with SMA between the ages of 2 and 25 years. Those who received the study drug had an average 1.36 increase from baseline on a motor function measure versus a 0.19 decrease in function for those who received placebo.
The FDA noted that the most common treatment-related adverse events (AEs) include fever, diarrhea, rash, ulcers of the mouth, arthralgia, and urinary tract infections. Additional AEs reported in some patients with infantile-onset SMA included upper respiratory tract infection, pneumonia, constipation, and vomiting.
The drug received fast track designation and priority review from the FDA, as well as orphan drug designation.
‘Eagerly awaited’
“Today marks an incredibly important moment for the broader SMA patient community that had been in dire need of safe and effective treatment options,” Stuart W. Peltz, PhD, chief executive officer of PTC Therapeutics, said in a company statement.
“Given [that] the majority of people with SMA in the U.S. remain untreated, we believe Evrysdi, with its favorable clinical profile and oral administration, may offer meaningful benefits for many living with this rare neurological disease,” Levi Garraway, MD, PhD, chief medical officer and head of global product development for Genentech, added in the company’s press release. Genentech is a member of the Roche Group.
The drug is continuing to be studied in more than 450 individuals as part of a “large and robust clinical trial program in SMA,” the company reports. These participants are between the ages of 2 months and 60 years.
“The approval of Evrysdi is an eagerly awaited milestone for our community. We appreciate Genentech’s commitment to … developing a treatment that can be administered at home,” Kenneth Hobby, president of the nonprofit Cure SMA, said in the same release.
In May 2019, the FDA approved the first gene therapy for SMA – the infusion drug onasemnogene abeparvovec-xioi (Zolgensma, AveXis Inc).
Genentech announced that the new oral drug will be available in the United States within 2 weeks “for direct delivery to patients’ homes.”
A version of this article originally appeared on Medscape.com.
This marks the first approval of an oral therapy for the rare and devastating condition.
Risdiplam, marketed by Roche and PTC Therapeutics, provides “an important treatment option for patients with SMA, following the approval of the first treatment for this devastating disease less than 4 years ago,” Billy Dunn, MD, director of the Office of Neuroscience in the Center for Drug Evaluation and Research at the FDA, said in a release from the agency.
The approval was based on the results from two trials. In the open-label FIREFISH study of infantile-onset SMA, 7 (41%) of the 17 participants (mean baseline age, 6.7 months) were able to sit independently for more than 5 seconds after 12 months of treatment with risdiplam. This was a “meaningful difference from the natural progression of the disease because all untreated infants with infantile-onset SMA cannot sit independently,” the FDA noted. In addition, 81% of the participants were alive after 23 or more months of treatment – and without need of permanent ventilation.
The second study was the randomized controlled trial known as SUNFISH and included 180 patients with SMA between the ages of 2 and 25 years. Those who received the study drug had an average 1.36 increase from baseline on a motor function measure versus a 0.19 decrease in function for those who received placebo.
The FDA noted that the most common treatment-related adverse events (AEs) include fever, diarrhea, rash, ulcers of the mouth, arthralgia, and urinary tract infections. Additional AEs reported in some patients with infantile-onset SMA included upper respiratory tract infection, pneumonia, constipation, and vomiting.
The drug received fast track designation and priority review from the FDA, as well as orphan drug designation.
‘Eagerly awaited’
“Today marks an incredibly important moment for the broader SMA patient community that had been in dire need of safe and effective treatment options,” Stuart W. Peltz, PhD, chief executive officer of PTC Therapeutics, said in a company statement.
“Given [that] the majority of people with SMA in the U.S. remain untreated, we believe Evrysdi, with its favorable clinical profile and oral administration, may offer meaningful benefits for many living with this rare neurological disease,” Levi Garraway, MD, PhD, chief medical officer and head of global product development for Genentech, added in the company’s press release. Genentech is a member of the Roche Group.
The drug is continuing to be studied in more than 450 individuals as part of a “large and robust clinical trial program in SMA,” the company reports. These participants are between the ages of 2 months and 60 years.
“The approval of Evrysdi is an eagerly awaited milestone for our community. We appreciate Genentech’s commitment to … developing a treatment that can be administered at home,” Kenneth Hobby, president of the nonprofit Cure SMA, said in the same release.
In May 2019, the FDA approved the first gene therapy for SMA – the infusion drug onasemnogene abeparvovec-xioi (Zolgensma, AveXis Inc).
Genentech announced that the new oral drug will be available in the United States within 2 weeks “for direct delivery to patients’ homes.”
A version of this article originally appeared on Medscape.com.
This marks the first approval of an oral therapy for the rare and devastating condition.
Risdiplam, marketed by Roche and PTC Therapeutics, provides “an important treatment option for patients with SMA, following the approval of the first treatment for this devastating disease less than 4 years ago,” Billy Dunn, MD, director of the Office of Neuroscience in the Center for Drug Evaluation and Research at the FDA, said in a release from the agency.
The approval was based on the results from two trials. In the open-label FIREFISH study of infantile-onset SMA, 7 (41%) of the 17 participants (mean baseline age, 6.7 months) were able to sit independently for more than 5 seconds after 12 months of treatment with risdiplam. This was a “meaningful difference from the natural progression of the disease because all untreated infants with infantile-onset SMA cannot sit independently,” the FDA noted. In addition, 81% of the participants were alive after 23 or more months of treatment – and without need of permanent ventilation.
The second study was the randomized controlled trial known as SUNFISH and included 180 patients with SMA between the ages of 2 and 25 years. Those who received the study drug had an average 1.36 increase from baseline on a motor function measure versus a 0.19 decrease in function for those who received placebo.
The FDA noted that the most common treatment-related adverse events (AEs) include fever, diarrhea, rash, ulcers of the mouth, arthralgia, and urinary tract infections. Additional AEs reported in some patients with infantile-onset SMA included upper respiratory tract infection, pneumonia, constipation, and vomiting.
The drug received fast track designation and priority review from the FDA, as well as orphan drug designation.
‘Eagerly awaited’
“Today marks an incredibly important moment for the broader SMA patient community that had been in dire need of safe and effective treatment options,” Stuart W. Peltz, PhD, chief executive officer of PTC Therapeutics, said in a company statement.
“Given [that] the majority of people with SMA in the U.S. remain untreated, we believe Evrysdi, with its favorable clinical profile and oral administration, may offer meaningful benefits for many living with this rare neurological disease,” Levi Garraway, MD, PhD, chief medical officer and head of global product development for Genentech, added in the company’s press release. Genentech is a member of the Roche Group.
The drug is continuing to be studied in more than 450 individuals as part of a “large and robust clinical trial program in SMA,” the company reports. These participants are between the ages of 2 months and 60 years.
“The approval of Evrysdi is an eagerly awaited milestone for our community. We appreciate Genentech’s commitment to … developing a treatment that can be administered at home,” Kenneth Hobby, president of the nonprofit Cure SMA, said in the same release.
In May 2019, the FDA approved the first gene therapy for SMA – the infusion drug onasemnogene abeparvovec-xioi (Zolgensma, AveXis Inc).
Genentech announced that the new oral drug will be available in the United States within 2 weeks “for direct delivery to patients’ homes.”
A version of this article originally appeared on Medscape.com.
Sleep problems in young children linked to lower QOL in later years
Sleep problems in children from birth to middle childhood may lead to decreased emotional well-being and quality of life by the time a child is 10-11 years old, a recent longitudinal study has found.
The effects of these impairments increased over time and included internalizing and externalizing concerns, self-control, and quality of life, but did not appear to significantly affect cognitive or academic skills, according to Ariel A. Williamson, PhD, DBSM, of Children’s Hospital of Philadelphia, and colleagues. While children with consistent sleep problems experienced the worse outcomes, mild sleep problems also were associated with impairment, the researchers said.
“The range of impairments across academic and psychosocial domains in middle childhood indicate that it is important to screen for sleep problems consistently over the course of a child’s development, especially to target children who experience persistent sleep problems over time,” said Dr. Williamson in a press release.
The researchers examined data from 5,107 children in the Longitudinal Study of Australian Children – Birth Cohort, where sleep problems and well-being outcomes were measured at multiple time points. Behaviors such as difficulty getting off to sleep at night, not happy to sleep alone, and waking during the night were defined as sleep problems. The investigators found five main domains of sleep issues: children who had persistent sleep problems through middle childhood (7.7%), limited sleep problems as an infant or during preschool (9.0%), mild sleep problems over time (14.4%), increased sleep problems during middle childhood (17.0%), and a group that did not experience sleep problems (51.9%).
Caregivers reported sleep issues in the cohort, while well-being outcomes were reported by caregivers and teachers, and tasks were completed by the children at 10-11 years of age. Dr. Williamson and colleagues examined well-being in terms of emotional and behavioral functioning, health-related quality of life, cognitive skills, and academic achievement.
Different reports from teacher and caregivers
Teacher and caregivers reported different effects in children with persistent sleep problems. Teachers reported moderate internalizing (effect size, –0.65; 95% confidence interval [CI],–0.87 to –0.43; P < .001) and externalizing concerns (ES, –0.40; 95% CI, –0.58 to –0.21; P less than .001), compared with children who did not have sleep problems, whereas caregivers reported large internalizing (ES, –0.75; 95% CI, –0.92 to –0.57; P less than .001) and externalizing concerns (ES, –0.70; 95% CI, –0.86 to –0.53; P < .001). Children with persistent sleep problems had moderate impairment of self-control as reported by caregivers, compared with children with no sleep problems (ES, –0.37; 95% CI, –0.52 to –0.21; P < .001). Psychosocial and health-related quality of life reported by caregivers were worse in children with persistent sleep problems, compared with children who did not have sleep problems (ES range, –0.78 to –0.90; 95% CI, –1.06 to –0.56; P < .001).
For children who exhibited increased sleep problems in middle childhood, caregivers (ES for both, –0.61; 95% CI, –0.76 to –0.46; P < .001) and teachers (ES range, –0.29 to –0.39; 95% CI, –0.53 to –0.15; P < .001) reported greater rates of internalizing and externalizing symptoms, compared with children who had no sleep issues.
Small impairments in internalizing internal or externalizing symptoms were seen in children who had limited sleep problems as an infant or in preschool (ES, –0.12; 95% CI, –0.23 to –0.01; P < .05) as reported by teachers, and in children with mild sleep problems over time (ES, –0.19; 95% CI, –0.30 to –0.08; P < .001) as reported by caregivers. There were no significant impairments in self-control for children in either the infant or preschool impairment group or in the group of children with mild sleep problems.
Across all groups, sleep problems did not significantly impair nonverbal reasoning, and most areas of academic competencies were not significantly impaired among groups except in language and literacy, and mathematical thinking for children with persistent sleep problems (ES, –0.41 for both; 95% CI, –0.60 to –0.23; P < .001). Children with increased sleep problems during middle childhood “had few academic and cognitive impairments,” and academic impairments among children with mild sleep problems were not significant.
Expert opinion
Brandon M. Seay MD, FAAP, pediatric pulmonologist and sleep specialist at Children’s Healthcare of Atlanta, said in an interview that the study is one of the first to offer longitudinal data for impairment in children with sleep problems. He said the paper emphasizes the need for recognizing when children are demonstrating sleep problems. “It just shows that problems that aren’t dealt with earlier on definitely have bigger impacts on sleep as you go through life,” he said.
Although primary care physicians and pediatricians should be already asking questions about sleep through anticipatory guidance, he said, intervening earlier for sleep problems is important. He noted children who exhibit sleep problems over time are more likely to have issues in handling their emotions and eventually may develop cognitive issues. “[W]e know that if these problems continue to go through, this paper’s showing us that they have worse effects down the road,” he said.
Impact of the COVID-19 crisis
These problems may also be worsened by the COVID-19 pandemic. Dr. Seay noted that with many parents working from home, sleep schedules can be affected and parents may also be co-sleeping with their children, which can cause chronic insomnia and early waking. To help address sleep issues, especially ones that may have arisen during COVID-19, parents should make sure their children show up for primary care visits to report problems, and clinicians should make a sleep routine a focus of conversations around sleep problems.
Prior to the pandemic, “we already were hitting upon that in sleep clinic, making sure [they] get the same schedule every day,” said Dr. Seay. For parents with children who have “issues with insomnia or waking up during the night, having that routine in place does help to mitigate that a little bit, so if that routine is not there, it can actually exacerbate the issues.”
This study was funded by the Australian federal government. The authors report no relevant conflicts of interest. Dr. Seay reports no relevant conflicts of interest.
SOURCE: Williamson AA et al. J Child Psychol Psychiatry. 2020 Jul 26. doi:10.1111/jcpp.13303.
Sleep problems in children from birth to middle childhood may lead to decreased emotional well-being and quality of life by the time a child is 10-11 years old, a recent longitudinal study has found.
The effects of these impairments increased over time and included internalizing and externalizing concerns, self-control, and quality of life, but did not appear to significantly affect cognitive or academic skills, according to Ariel A. Williamson, PhD, DBSM, of Children’s Hospital of Philadelphia, and colleagues. While children with consistent sleep problems experienced the worse outcomes, mild sleep problems also were associated with impairment, the researchers said.
“The range of impairments across academic and psychosocial domains in middle childhood indicate that it is important to screen for sleep problems consistently over the course of a child’s development, especially to target children who experience persistent sleep problems over time,” said Dr. Williamson in a press release.
The researchers examined data from 5,107 children in the Longitudinal Study of Australian Children – Birth Cohort, where sleep problems and well-being outcomes were measured at multiple time points. Behaviors such as difficulty getting off to sleep at night, not happy to sleep alone, and waking during the night were defined as sleep problems. The investigators found five main domains of sleep issues: children who had persistent sleep problems through middle childhood (7.7%), limited sleep problems as an infant or during preschool (9.0%), mild sleep problems over time (14.4%), increased sleep problems during middle childhood (17.0%), and a group that did not experience sleep problems (51.9%).
Caregivers reported sleep issues in the cohort, while well-being outcomes were reported by caregivers and teachers, and tasks were completed by the children at 10-11 years of age. Dr. Williamson and colleagues examined well-being in terms of emotional and behavioral functioning, health-related quality of life, cognitive skills, and academic achievement.
Different reports from teacher and caregivers
Teacher and caregivers reported different effects in children with persistent sleep problems. Teachers reported moderate internalizing (effect size, –0.65; 95% confidence interval [CI],–0.87 to –0.43; P < .001) and externalizing concerns (ES, –0.40; 95% CI, –0.58 to –0.21; P less than .001), compared with children who did not have sleep problems, whereas caregivers reported large internalizing (ES, –0.75; 95% CI, –0.92 to –0.57; P less than .001) and externalizing concerns (ES, –0.70; 95% CI, –0.86 to –0.53; P < .001). Children with persistent sleep problems had moderate impairment of self-control as reported by caregivers, compared with children with no sleep problems (ES, –0.37; 95% CI, –0.52 to –0.21; P < .001). Psychosocial and health-related quality of life reported by caregivers were worse in children with persistent sleep problems, compared with children who did not have sleep problems (ES range, –0.78 to –0.90; 95% CI, –1.06 to –0.56; P < .001).
For children who exhibited increased sleep problems in middle childhood, caregivers (ES for both, –0.61; 95% CI, –0.76 to –0.46; P < .001) and teachers (ES range, –0.29 to –0.39; 95% CI, –0.53 to –0.15; P < .001) reported greater rates of internalizing and externalizing symptoms, compared with children who had no sleep issues.
Small impairments in internalizing internal or externalizing symptoms were seen in children who had limited sleep problems as an infant or in preschool (ES, –0.12; 95% CI, –0.23 to –0.01; P < .05) as reported by teachers, and in children with mild sleep problems over time (ES, –0.19; 95% CI, –0.30 to –0.08; P < .001) as reported by caregivers. There were no significant impairments in self-control for children in either the infant or preschool impairment group or in the group of children with mild sleep problems.
Across all groups, sleep problems did not significantly impair nonverbal reasoning, and most areas of academic competencies were not significantly impaired among groups except in language and literacy, and mathematical thinking for children with persistent sleep problems (ES, –0.41 for both; 95% CI, –0.60 to –0.23; P < .001). Children with increased sleep problems during middle childhood “had few academic and cognitive impairments,” and academic impairments among children with mild sleep problems were not significant.
Expert opinion
Brandon M. Seay MD, FAAP, pediatric pulmonologist and sleep specialist at Children’s Healthcare of Atlanta, said in an interview that the study is one of the first to offer longitudinal data for impairment in children with sleep problems. He said the paper emphasizes the need for recognizing when children are demonstrating sleep problems. “It just shows that problems that aren’t dealt with earlier on definitely have bigger impacts on sleep as you go through life,” he said.
Although primary care physicians and pediatricians should be already asking questions about sleep through anticipatory guidance, he said, intervening earlier for sleep problems is important. He noted children who exhibit sleep problems over time are more likely to have issues in handling their emotions and eventually may develop cognitive issues. “[W]e know that if these problems continue to go through, this paper’s showing us that they have worse effects down the road,” he said.
Impact of the COVID-19 crisis
These problems may also be worsened by the COVID-19 pandemic. Dr. Seay noted that with many parents working from home, sleep schedules can be affected and parents may also be co-sleeping with their children, which can cause chronic insomnia and early waking. To help address sleep issues, especially ones that may have arisen during COVID-19, parents should make sure their children show up for primary care visits to report problems, and clinicians should make a sleep routine a focus of conversations around sleep problems.
Prior to the pandemic, “we already were hitting upon that in sleep clinic, making sure [they] get the same schedule every day,” said Dr. Seay. For parents with children who have “issues with insomnia or waking up during the night, having that routine in place does help to mitigate that a little bit, so if that routine is not there, it can actually exacerbate the issues.”
This study was funded by the Australian federal government. The authors report no relevant conflicts of interest. Dr. Seay reports no relevant conflicts of interest.
SOURCE: Williamson AA et al. J Child Psychol Psychiatry. 2020 Jul 26. doi:10.1111/jcpp.13303.
Sleep problems in children from birth to middle childhood may lead to decreased emotional well-being and quality of life by the time a child is 10-11 years old, a recent longitudinal study has found.
The effects of these impairments increased over time and included internalizing and externalizing concerns, self-control, and quality of life, but did not appear to significantly affect cognitive or academic skills, according to Ariel A. Williamson, PhD, DBSM, of Children’s Hospital of Philadelphia, and colleagues. While children with consistent sleep problems experienced the worse outcomes, mild sleep problems also were associated with impairment, the researchers said.
“The range of impairments across academic and psychosocial domains in middle childhood indicate that it is important to screen for sleep problems consistently over the course of a child’s development, especially to target children who experience persistent sleep problems over time,” said Dr. Williamson in a press release.
The researchers examined data from 5,107 children in the Longitudinal Study of Australian Children – Birth Cohort, where sleep problems and well-being outcomes were measured at multiple time points. Behaviors such as difficulty getting off to sleep at night, not happy to sleep alone, and waking during the night were defined as sleep problems. The investigators found five main domains of sleep issues: children who had persistent sleep problems through middle childhood (7.7%), limited sleep problems as an infant or during preschool (9.0%), mild sleep problems over time (14.4%), increased sleep problems during middle childhood (17.0%), and a group that did not experience sleep problems (51.9%).
Caregivers reported sleep issues in the cohort, while well-being outcomes were reported by caregivers and teachers, and tasks were completed by the children at 10-11 years of age. Dr. Williamson and colleagues examined well-being in terms of emotional and behavioral functioning, health-related quality of life, cognitive skills, and academic achievement.
Different reports from teacher and caregivers
Teacher and caregivers reported different effects in children with persistent sleep problems. Teachers reported moderate internalizing (effect size, –0.65; 95% confidence interval [CI],–0.87 to –0.43; P < .001) and externalizing concerns (ES, –0.40; 95% CI, –0.58 to –0.21; P less than .001), compared with children who did not have sleep problems, whereas caregivers reported large internalizing (ES, –0.75; 95% CI, –0.92 to –0.57; P less than .001) and externalizing concerns (ES, –0.70; 95% CI, –0.86 to –0.53; P < .001). Children with persistent sleep problems had moderate impairment of self-control as reported by caregivers, compared with children with no sleep problems (ES, –0.37; 95% CI, –0.52 to –0.21; P < .001). Psychosocial and health-related quality of life reported by caregivers were worse in children with persistent sleep problems, compared with children who did not have sleep problems (ES range, –0.78 to –0.90; 95% CI, –1.06 to –0.56; P < .001).
For children who exhibited increased sleep problems in middle childhood, caregivers (ES for both, –0.61; 95% CI, –0.76 to –0.46; P < .001) and teachers (ES range, –0.29 to –0.39; 95% CI, –0.53 to –0.15; P < .001) reported greater rates of internalizing and externalizing symptoms, compared with children who had no sleep issues.
Small impairments in internalizing internal or externalizing symptoms were seen in children who had limited sleep problems as an infant or in preschool (ES, –0.12; 95% CI, –0.23 to –0.01; P < .05) as reported by teachers, and in children with mild sleep problems over time (ES, –0.19; 95% CI, –0.30 to –0.08; P < .001) as reported by caregivers. There were no significant impairments in self-control for children in either the infant or preschool impairment group or in the group of children with mild sleep problems.
Across all groups, sleep problems did not significantly impair nonverbal reasoning, and most areas of academic competencies were not significantly impaired among groups except in language and literacy, and mathematical thinking for children with persistent sleep problems (ES, –0.41 for both; 95% CI, –0.60 to –0.23; P < .001). Children with increased sleep problems during middle childhood “had few academic and cognitive impairments,” and academic impairments among children with mild sleep problems were not significant.
Expert opinion
Brandon M. Seay MD, FAAP, pediatric pulmonologist and sleep specialist at Children’s Healthcare of Atlanta, said in an interview that the study is one of the first to offer longitudinal data for impairment in children with sleep problems. He said the paper emphasizes the need for recognizing when children are demonstrating sleep problems. “It just shows that problems that aren’t dealt with earlier on definitely have bigger impacts on sleep as you go through life,” he said.
Although primary care physicians and pediatricians should be already asking questions about sleep through anticipatory guidance, he said, intervening earlier for sleep problems is important. He noted children who exhibit sleep problems over time are more likely to have issues in handling their emotions and eventually may develop cognitive issues. “[W]e know that if these problems continue to go through, this paper’s showing us that they have worse effects down the road,” he said.
Impact of the COVID-19 crisis
These problems may also be worsened by the COVID-19 pandemic. Dr. Seay noted that with many parents working from home, sleep schedules can be affected and parents may also be co-sleeping with their children, which can cause chronic insomnia and early waking. To help address sleep issues, especially ones that may have arisen during COVID-19, parents should make sure their children show up for primary care visits to report problems, and clinicians should make a sleep routine a focus of conversations around sleep problems.
Prior to the pandemic, “we already were hitting upon that in sleep clinic, making sure [they] get the same schedule every day,” said Dr. Seay. For parents with children who have “issues with insomnia or waking up during the night, having that routine in place does help to mitigate that a little bit, so if that routine is not there, it can actually exacerbate the issues.”
This study was funded by the Australian federal government. The authors report no relevant conflicts of interest. Dr. Seay reports no relevant conflicts of interest.
SOURCE: Williamson AA et al. J Child Psychol Psychiatry. 2020 Jul 26. doi:10.1111/jcpp.13303.
FROM JOURNAL OF CHILD PSYCHOLOGY AND PSYCHIATRY
COVID-19 cases in children nearly doubled in just 4 weeks
The cumulative number of new COVID-19 cases among children in the United States jumped by 90% during a recent 4-week period, according to a report that confirms children are not immune to the coronavirus.
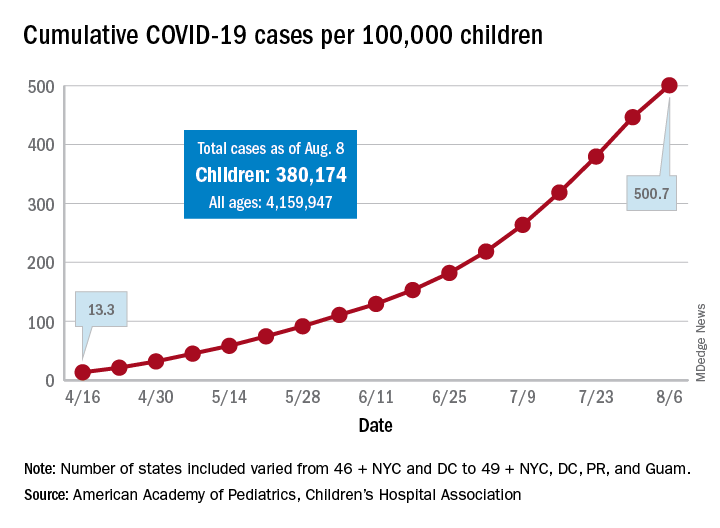
“In areas with rapid community spread, it’s likely that more children will also be infected, and these data show that,” Sally Goza, MD, president of the American Academy of Pediatrics, said in a written statement. “I urge people to wear cloth face coverings and be diligent in social distancing and hand-washing. It is up to us to make the difference, community by community.”
The joint report from the AAP and the Children’s Hospital Association draws on data from state and local health departments in 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
The cumulative number of COVID-19 cases in children as of Aug. 6, 2020, was 380,174, and that number is 90% higher – an increase of 179,990 cases – than the total on July 9, just 4 weeks earlier, the two organizations said in the report.
and 27 states out of 47 with available data now report that over 10% of their cases were children, with Wyoming the highest at 16.5% and New Jersey the lowest at 2.9%, the report data show.
Alabama has a higher percentage of 22.5%, but the state has been reporting cases in individuals aged 0-24 years as child cases since May 7. The report’s findings are somewhat limited by differences in reporting among the states and by “gaps in the data they are reporting [that affect] how the data can be interpreted,” the AAP said in its statement.
The cumulative number of cases per 100,000 children has risen from 13.3 in mid-April, when the total number was 9,259 cases, to 500.7 per 100,000 as of Aug. 6, and there are now 21 states, along with the District of Columbia, reporting a rate of over 500 cases per 100,000 children. Arizona has the highest rate at 1,206.4, followed by South Carolina (1,074.4) and Tennessee (1,050.8), the AAP and the CHA said.
In New York City, the early epicenter of the pandemic, the 390.5 cases per 100,000 children have been reported, and in New Jersey, which joined New York in the initial surge of cases, the number is 269.5. As of Aug. 6, Hawaii had the fewest cases of any state at 91.2 per 100,000, according to the report.
Children continue to represent a very low proportion of COVID-19 deaths, “but as case counts rise across the board, that is likely to impact more children with severe illness as well,” Sean O’Leary, MD, MPH, vice chair of the AAP’s committee on infectious diseases, said in the AAP statement.
It is possible that “some of the increase in numbers of cases in children could be due to more testing. Early in the pandemic, testing only occurred for the sickest individuals. Now that there is more testing capacity … the numbers reflect a broader slice of the population, including children who may have mild or few symptoms,” the AAP suggested.
This article was updated on 8/17/2020.
The cumulative number of new COVID-19 cases among children in the United States jumped by 90% during a recent 4-week period, according to a report that confirms children are not immune to the coronavirus.

“In areas with rapid community spread, it’s likely that more children will also be infected, and these data show that,” Sally Goza, MD, president of the American Academy of Pediatrics, said in a written statement. “I urge people to wear cloth face coverings and be diligent in social distancing and hand-washing. It is up to us to make the difference, community by community.”
The joint report from the AAP and the Children’s Hospital Association draws on data from state and local health departments in 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
The cumulative number of COVID-19 cases in children as of Aug. 6, 2020, was 380,174, and that number is 90% higher – an increase of 179,990 cases – than the total on July 9, just 4 weeks earlier, the two organizations said in the report.
and 27 states out of 47 with available data now report that over 10% of their cases were children, with Wyoming the highest at 16.5% and New Jersey the lowest at 2.9%, the report data show.
Alabama has a higher percentage of 22.5%, but the state has been reporting cases in individuals aged 0-24 years as child cases since May 7. The report’s findings are somewhat limited by differences in reporting among the states and by “gaps in the data they are reporting [that affect] how the data can be interpreted,” the AAP said in its statement.
The cumulative number of cases per 100,000 children has risen from 13.3 in mid-April, when the total number was 9,259 cases, to 500.7 per 100,000 as of Aug. 6, and there are now 21 states, along with the District of Columbia, reporting a rate of over 500 cases per 100,000 children. Arizona has the highest rate at 1,206.4, followed by South Carolina (1,074.4) and Tennessee (1,050.8), the AAP and the CHA said.
In New York City, the early epicenter of the pandemic, the 390.5 cases per 100,000 children have been reported, and in New Jersey, which joined New York in the initial surge of cases, the number is 269.5. As of Aug. 6, Hawaii had the fewest cases of any state at 91.2 per 100,000, according to the report.
Children continue to represent a very low proportion of COVID-19 deaths, “but as case counts rise across the board, that is likely to impact more children with severe illness as well,” Sean O’Leary, MD, MPH, vice chair of the AAP’s committee on infectious diseases, said in the AAP statement.
It is possible that “some of the increase in numbers of cases in children could be due to more testing. Early in the pandemic, testing only occurred for the sickest individuals. Now that there is more testing capacity … the numbers reflect a broader slice of the population, including children who may have mild or few symptoms,” the AAP suggested.
This article was updated on 8/17/2020.
The cumulative number of new COVID-19 cases among children in the United States jumped by 90% during a recent 4-week period, according to a report that confirms children are not immune to the coronavirus.

“In areas with rapid community spread, it’s likely that more children will also be infected, and these data show that,” Sally Goza, MD, president of the American Academy of Pediatrics, said in a written statement. “I urge people to wear cloth face coverings and be diligent in social distancing and hand-washing. It is up to us to make the difference, community by community.”
The joint report from the AAP and the Children’s Hospital Association draws on data from state and local health departments in 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
The cumulative number of COVID-19 cases in children as of Aug. 6, 2020, was 380,174, and that number is 90% higher – an increase of 179,990 cases – than the total on July 9, just 4 weeks earlier, the two organizations said in the report.
and 27 states out of 47 with available data now report that over 10% of their cases were children, with Wyoming the highest at 16.5% and New Jersey the lowest at 2.9%, the report data show.
Alabama has a higher percentage of 22.5%, but the state has been reporting cases in individuals aged 0-24 years as child cases since May 7. The report’s findings are somewhat limited by differences in reporting among the states and by “gaps in the data they are reporting [that affect] how the data can be interpreted,” the AAP said in its statement.
The cumulative number of cases per 100,000 children has risen from 13.3 in mid-April, when the total number was 9,259 cases, to 500.7 per 100,000 as of Aug. 6, and there are now 21 states, along with the District of Columbia, reporting a rate of over 500 cases per 100,000 children. Arizona has the highest rate at 1,206.4, followed by South Carolina (1,074.4) and Tennessee (1,050.8), the AAP and the CHA said.
In New York City, the early epicenter of the pandemic, the 390.5 cases per 100,000 children have been reported, and in New Jersey, which joined New York in the initial surge of cases, the number is 269.5. As of Aug. 6, Hawaii had the fewest cases of any state at 91.2 per 100,000, according to the report.
Children continue to represent a very low proportion of COVID-19 deaths, “but as case counts rise across the board, that is likely to impact more children with severe illness as well,” Sean O’Leary, MD, MPH, vice chair of the AAP’s committee on infectious diseases, said in the AAP statement.
It is possible that “some of the increase in numbers of cases in children could be due to more testing. Early in the pandemic, testing only occurred for the sickest individuals. Now that there is more testing capacity … the numbers reflect a broader slice of the population, including children who may have mild or few symptoms,” the AAP suggested.
This article was updated on 8/17/2020.
Rapid cycle pediatric simulation exercises promise improved readiness
Focused repetition builds sustained skill
A methodical, constructive, goal-oriented rapid repetition of emergency response simulations has emerged as a dominant strategy for pediatric readiness in the hospital setting, according to a detailed description of one such program at the virtual Pediatric Hospital Medicine.
Rather than a single run-through followed by a lengthy debriefing, which has been a traditional approach, short simulations done rapidly and repeatedly until skills are mastered improve skill development, according to Jeanmarie Schied, MD, of the department of pediatrics, University of Chicago Medicine.
“This method utilizes repetitions to develop muscle memory much like an athlete who ‘practices, practices, practices’ until it becomes second nature,” Dr. Schied explained.
Dr. Schied credited this approach to Elizabeth Hunt, MD, PhD, director of the Johns Hopkins Medicine Simulation Center. The method created by Dr. Hunt is called Rapid Cycle Deliberate Practice (RCDP). At the University of Chicago, where the same principles are being applied, “we have had great success,” Dr. Schied said.
Deficiencies in the traditional approach prompted the change. It has been shown that when experienced residents who have performed multiple simulations are compared to new residents with limited experience or when those certified in Pediatric Advanced Life Support (PAL) are compared to those who are not, they “do not necessarily do better” in the metrics used in simulations to measure competence, according to Dr. Schied.
With the RDCP, learners get multiple chances to master skills.
“Everyone makes mistakes, and letting the participants know this ahead of time puts people at ease,” Dr. Schied said. “People want to know they will have a chance to rewind and do it right.”
In setting up an effective simulation program, the first step is a needs assessment. By first gauging the skill and experience level of those scheduled to participate, Dr. Schied said the program can be tailored to the audience.
The next step is formulating learning objectives. Dr. Schied recommended creating these objectives for the case overall and for each phase of the simulation as it progresses from basic clinical assessments through the specific interventions appropriate for the diagnosis.
Within these objectives there are additional goals. For example, the team should work to administer care within prespecified benchmarks, such as an elapsed time of 60 seconds or less for oxygenation or a time of 180 seconds or less for defibrillation, according to Dr. Schied.
Yet, Dr. Schied suggested that enforcing these goals on initial run-throughs might not be appropriate.
“Let the scenario run longer so you can see the deficits,” Dr. Schied said. If, for example, chest compression is not being done correctly, she recommended interrupting the process to provide immediate and direct feedback. In critiquing the performance, Dr. Schied advised against a critical or punitive tone.
“Inform the learners that they are in a safe environment,” she said. It is essential to identify errors so that they can be corrected on the next run of the practice simulation, but Dr. Schied advised instructors to “be nonjudgmental.” Praise is appropriate when warranted, but she also warned, “don’t sugarcoat” a substandard performance.
During the simulation, team leaders should employ action phrases, meaning that the problem and the action needed are expressed at the same time, according to Dr. Schied. Examples include, “the patient is not breathing, start bagging,” or “there is no pulse, start compression.”
“When the team gets used to these action-linked phrases, studies show that they react in a more timely fashion,” Dr. Schied explained at the event sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
In the study by Dr. Hunt that established the effectiveness of RDCP, 51 pediatric residents who had previously participated in a cardiopulmonary arrest simulation were retested again after being retrained with the RDCP methodology (Resuscitation 2014;85:945-51).
RDCP “was associated with improvement in performance of key measures of quality life support and progressive acquisition of resuscitation skills,” according to Dr. Hunt, who has published frequently on resuscitation training in pediatrics.
Prior to RDCP, traditional methods produced “little improvement” in resuscitation skills when measured over the course of pediatric residency, according to Dr. Hunt. After RDCP, third-year residents were shown to be “significantly more likely than first-years to defibrillate within 2 minutes,” she reported.
However, there are other strategies to improve retention of skills, according to Dr. Schied. For example, it is important to conduct simulations when the staff can focus. Specifically, Dr. Schied recommended conducting simulations immediately after a staff meeting or before a scheduled shift so that clinical responsibilities will not interfere or divert the learner’s attention. She also recommended conducting key simulations quarterly.
“Studies have shown that knowledge deterioration related to resuscitation begins about 4 months after the last simulation,” she said.
In addition to building the skills of individual participants, Dr. Schied emphasized the importance of also developing effective team dynamics and active communication. In the debriefing that should follow every simulation, she recommended encouraging a discussion of strengths and weaknesses of the team response.
Pediatric emergency simulation scenarios are readily available on multiple sites found on the Internet,” Dr. Schied said. She recommended documenting performance so the data are available for subsequent analysis.
Focused repetition builds sustained skill
Focused repetition builds sustained skill
A methodical, constructive, goal-oriented rapid repetition of emergency response simulations has emerged as a dominant strategy for pediatric readiness in the hospital setting, according to a detailed description of one such program at the virtual Pediatric Hospital Medicine.
Rather than a single run-through followed by a lengthy debriefing, which has been a traditional approach, short simulations done rapidly and repeatedly until skills are mastered improve skill development, according to Jeanmarie Schied, MD, of the department of pediatrics, University of Chicago Medicine.
“This method utilizes repetitions to develop muscle memory much like an athlete who ‘practices, practices, practices’ until it becomes second nature,” Dr. Schied explained.
Dr. Schied credited this approach to Elizabeth Hunt, MD, PhD, director of the Johns Hopkins Medicine Simulation Center. The method created by Dr. Hunt is called Rapid Cycle Deliberate Practice (RCDP). At the University of Chicago, where the same principles are being applied, “we have had great success,” Dr. Schied said.
Deficiencies in the traditional approach prompted the change. It has been shown that when experienced residents who have performed multiple simulations are compared to new residents with limited experience or when those certified in Pediatric Advanced Life Support (PAL) are compared to those who are not, they “do not necessarily do better” in the metrics used in simulations to measure competence, according to Dr. Schied.
With the RDCP, learners get multiple chances to master skills.
“Everyone makes mistakes, and letting the participants know this ahead of time puts people at ease,” Dr. Schied said. “People want to know they will have a chance to rewind and do it right.”
In setting up an effective simulation program, the first step is a needs assessment. By first gauging the skill and experience level of those scheduled to participate, Dr. Schied said the program can be tailored to the audience.
The next step is formulating learning objectives. Dr. Schied recommended creating these objectives for the case overall and for each phase of the simulation as it progresses from basic clinical assessments through the specific interventions appropriate for the diagnosis.
Within these objectives there are additional goals. For example, the team should work to administer care within prespecified benchmarks, such as an elapsed time of 60 seconds or less for oxygenation or a time of 180 seconds or less for defibrillation, according to Dr. Schied.
Yet, Dr. Schied suggested that enforcing these goals on initial run-throughs might not be appropriate.
“Let the scenario run longer so you can see the deficits,” Dr. Schied said. If, for example, chest compression is not being done correctly, she recommended interrupting the process to provide immediate and direct feedback. In critiquing the performance, Dr. Schied advised against a critical or punitive tone.
“Inform the learners that they are in a safe environment,” she said. It is essential to identify errors so that they can be corrected on the next run of the practice simulation, but Dr. Schied advised instructors to “be nonjudgmental.” Praise is appropriate when warranted, but she also warned, “don’t sugarcoat” a substandard performance.
During the simulation, team leaders should employ action phrases, meaning that the problem and the action needed are expressed at the same time, according to Dr. Schied. Examples include, “the patient is not breathing, start bagging,” or “there is no pulse, start compression.”
“When the team gets used to these action-linked phrases, studies show that they react in a more timely fashion,” Dr. Schied explained at the event sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
In the study by Dr. Hunt that established the effectiveness of RDCP, 51 pediatric residents who had previously participated in a cardiopulmonary arrest simulation were retested again after being retrained with the RDCP methodology (Resuscitation 2014;85:945-51).
RDCP “was associated with improvement in performance of key measures of quality life support and progressive acquisition of resuscitation skills,” according to Dr. Hunt, who has published frequently on resuscitation training in pediatrics.
Prior to RDCP, traditional methods produced “little improvement” in resuscitation skills when measured over the course of pediatric residency, according to Dr. Hunt. After RDCP, third-year residents were shown to be “significantly more likely than first-years to defibrillate within 2 minutes,” she reported.
However, there are other strategies to improve retention of skills, according to Dr. Schied. For example, it is important to conduct simulations when the staff can focus. Specifically, Dr. Schied recommended conducting simulations immediately after a staff meeting or before a scheduled shift so that clinical responsibilities will not interfere or divert the learner’s attention. She also recommended conducting key simulations quarterly.
“Studies have shown that knowledge deterioration related to resuscitation begins about 4 months after the last simulation,” she said.
In addition to building the skills of individual participants, Dr. Schied emphasized the importance of also developing effective team dynamics and active communication. In the debriefing that should follow every simulation, she recommended encouraging a discussion of strengths and weaknesses of the team response.
Pediatric emergency simulation scenarios are readily available on multiple sites found on the Internet,” Dr. Schied said. She recommended documenting performance so the data are available for subsequent analysis.
A methodical, constructive, goal-oriented rapid repetition of emergency response simulations has emerged as a dominant strategy for pediatric readiness in the hospital setting, according to a detailed description of one such program at the virtual Pediatric Hospital Medicine.
Rather than a single run-through followed by a lengthy debriefing, which has been a traditional approach, short simulations done rapidly and repeatedly until skills are mastered improve skill development, according to Jeanmarie Schied, MD, of the department of pediatrics, University of Chicago Medicine.
“This method utilizes repetitions to develop muscle memory much like an athlete who ‘practices, practices, practices’ until it becomes second nature,” Dr. Schied explained.
Dr. Schied credited this approach to Elizabeth Hunt, MD, PhD, director of the Johns Hopkins Medicine Simulation Center. The method created by Dr. Hunt is called Rapid Cycle Deliberate Practice (RCDP). At the University of Chicago, where the same principles are being applied, “we have had great success,” Dr. Schied said.
Deficiencies in the traditional approach prompted the change. It has been shown that when experienced residents who have performed multiple simulations are compared to new residents with limited experience or when those certified in Pediatric Advanced Life Support (PAL) are compared to those who are not, they “do not necessarily do better” in the metrics used in simulations to measure competence, according to Dr. Schied.
With the RDCP, learners get multiple chances to master skills.
“Everyone makes mistakes, and letting the participants know this ahead of time puts people at ease,” Dr. Schied said. “People want to know they will have a chance to rewind and do it right.”
In setting up an effective simulation program, the first step is a needs assessment. By first gauging the skill and experience level of those scheduled to participate, Dr. Schied said the program can be tailored to the audience.
The next step is formulating learning objectives. Dr. Schied recommended creating these objectives for the case overall and for each phase of the simulation as it progresses from basic clinical assessments through the specific interventions appropriate for the diagnosis.
Within these objectives there are additional goals. For example, the team should work to administer care within prespecified benchmarks, such as an elapsed time of 60 seconds or less for oxygenation or a time of 180 seconds or less for defibrillation, according to Dr. Schied.
Yet, Dr. Schied suggested that enforcing these goals on initial run-throughs might not be appropriate.
“Let the scenario run longer so you can see the deficits,” Dr. Schied said. If, for example, chest compression is not being done correctly, she recommended interrupting the process to provide immediate and direct feedback. In critiquing the performance, Dr. Schied advised against a critical or punitive tone.
“Inform the learners that they are in a safe environment,” she said. It is essential to identify errors so that they can be corrected on the next run of the practice simulation, but Dr. Schied advised instructors to “be nonjudgmental.” Praise is appropriate when warranted, but she also warned, “don’t sugarcoat” a substandard performance.
During the simulation, team leaders should employ action phrases, meaning that the problem and the action needed are expressed at the same time, according to Dr. Schied. Examples include, “the patient is not breathing, start bagging,” or “there is no pulse, start compression.”
“When the team gets used to these action-linked phrases, studies show that they react in a more timely fashion,” Dr. Schied explained at the event sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
In the study by Dr. Hunt that established the effectiveness of RDCP, 51 pediatric residents who had previously participated in a cardiopulmonary arrest simulation were retested again after being retrained with the RDCP methodology (Resuscitation 2014;85:945-51).
RDCP “was associated with improvement in performance of key measures of quality life support and progressive acquisition of resuscitation skills,” according to Dr. Hunt, who has published frequently on resuscitation training in pediatrics.
Prior to RDCP, traditional methods produced “little improvement” in resuscitation skills when measured over the course of pediatric residency, according to Dr. Hunt. After RDCP, third-year residents were shown to be “significantly more likely than first-years to defibrillate within 2 minutes,” she reported.
However, there are other strategies to improve retention of skills, according to Dr. Schied. For example, it is important to conduct simulations when the staff can focus. Specifically, Dr. Schied recommended conducting simulations immediately after a staff meeting or before a scheduled shift so that clinical responsibilities will not interfere or divert the learner’s attention. She also recommended conducting key simulations quarterly.
“Studies have shown that knowledge deterioration related to resuscitation begins about 4 months after the last simulation,” she said.
In addition to building the skills of individual participants, Dr. Schied emphasized the importance of also developing effective team dynamics and active communication. In the debriefing that should follow every simulation, she recommended encouraging a discussion of strengths and weaknesses of the team response.
Pediatric emergency simulation scenarios are readily available on multiple sites found on the Internet,” Dr. Schied said. She recommended documenting performance so the data are available for subsequent analysis.
FROM PHM20 VIRTUAL
Most clinicians undertreat childhood lichen sclerosus
In the clinical experience of Libby Edwards, MD, the diagnosis of lichen sclerosus in a young girl often triggers worry from patients and parents alike.
“The parents are worried about the ramifications of genital diseases and they’re worried about scarring,” she said during the virtual annual meeting of the Society for Pediatric Dermatology.
Meanwhile, during the initial assessment, physicians tend to think about sexual abuse or sexually transmitted diseases as the primary culprit. “It’s really important that you consider those issues, but they’re not usually what’s going on,” said Dr. Edwards, a dermatologist who practices in Charlotte, N.C. “Also, for some reason we jump to yeast as a cause of diseases in the genital area. If the child is out of diapers and hasn’t reached puberty, it’s almost never yeast. Do a culture. Try and prove yeast. If it doesn’t respond to treatment for yeast, it’s not going to be yeast. Reassure, and don’t forget to reassure.”
. Lichen sclerosus presents classically as white, fragile plaques. “Textbooks say that there is cigarette paper-like crinkling of skin,” Dr. Edwards said. “I think of it being more like cellophane paper. In children, we often see it as smooth, kind of waxy and shiny, compared to adults. Children usually present with pruritus and irritation.”
Lichen sclerosus often starts in the clitoral area and on the perineum, and often with an edematous clitoral hood. “It often eventuates into clitoral phimosis, meaning that there is midline adhesion so that the clitoris is buried,” she said. “In adults, seeing this clitoral phimosis is a reliable sign of a scarring dermatosis – most often lichen sclerosus. But you can’t say that in children, because little girls will often have scarring over the clitoris. It’s just physiologic and means nothing, and it will go away at puberty. Certainly, sometimes this white discoloration can have crinkling. Purpura and tearing are common; if you look at lichen sclerosus histologically it looks like a thin epithelium that’s stretched over gelatin. Any rubbing and scratching can cause bleeding in the skin.”
Clinical appearance of well demarcated white skin with texture change drives the diagnosis. “It can be hard to tell from vitiligo at times, but there always should be texture change – whether it’s crinkling, whether it’s waxy, whether it’s smooth – and it’s symptomatic,” she said.
A biopsy is not usually required. “I think a good picture [of the affected area] or some sort of objective description in the chart is important, because most children do so well that in a few months there’s no sign of it, and the next provider [they see] may not believe that they ever had it,” she said.
The recommended initial treatment for lichen sclerosus in girls is a tiny amount of a superpotent topical corticosteroid ointment such as clobetasol or halobetasol one to two times daily until the skin is clear, which usually takes 2-4 months. “You do not treat these children until they’re comfortable, because that may be a week,” Dr. Edwards said. “You treat these children until the skin looks normal. Then you need to keep treating them, because if you don’t, the skin will relapse, even though they might not have symptoms.”
Following initial treatment, she recommends use of a superpotent corticosteroid once per day three times a week, or a midpotency steroid like triamcinolone ointment 0.1% every day. In her clinical experience, if lesions clear and remain clear with long-term treatment through puberty, the chances are good that they’ll stay clear if the medication is stopped.
“There are no studies on what to do after a patient clears,” said Dr. Edwards, chief of dermatology at Carolinas Medical Center, Charlotte, and adjunct clinical professor of dermatology at the University of North Carolina, Chapel Hill. “We have been informed by trial and error. If a child is totally clear after puberty, I will stop their medication and see them back every 3 months for about a year and a half. If they stay clear after a year and a half, I find that they stay clear. I wonder what happens at menopause. We surely don’t know.”
With consistent topical treatment, many patients will have clearing in one area of affected skin after a month or two, and it will take 3 or 4 months for the remaining area to clear. “I tend to see patients back every 6-8 weeks until they’re clear,” she said. “I do not like the idea of sending people out and saying, ‘use this medication twice a day for a month, then once a day for a month, then three times a week, then as needed.’
For patients concerned about the long-term use of topical steroids, the immunosuppressants tacrolimus and pimecrolimus are options. “They are often irritating on the vulva, but can work better than steroids for extragenital disease,” Dr. Edwards said. “Parents sometimes object to the use of a corticosteroid, but because these produce slower benefit and often burn with application, you can remind the parents that tacrolimus and pimecrolimus are not without side effects and are labeled as being associated with cancer. That often will prompt a parent to be willing to use a topical steroid. You can also point to studies that show the safety of topical steroids.”
Intralesional steroids are useful for thick lesions, but Dr. Edwards said that she has never had to use them in a child with lichen sclerosus. “I have found methotrexate to be useful in some people, but there is not one study on genital lichen sclerosus and methotrexate,” she said. “I find that about one in five patients with recalcitrant vulvar lichen sclerosus has had some benefit from methotrexate,” she added, noting that fractional CO2 laser “is showing promise in these patients.”
Dr. Edwards concluded her remarks by noting that she has never cared for a child with vulvar lichen sclerosus who didn’t respond to topical super potent steroids, “except due to poor compliance.”
She reported having no relevant financial disclosures.
In the clinical experience of Libby Edwards, MD, the diagnosis of lichen sclerosus in a young girl often triggers worry from patients and parents alike.
“The parents are worried about the ramifications of genital diseases and they’re worried about scarring,” she said during the virtual annual meeting of the Society for Pediatric Dermatology.
Meanwhile, during the initial assessment, physicians tend to think about sexual abuse or sexually transmitted diseases as the primary culprit. “It’s really important that you consider those issues, but they’re not usually what’s going on,” said Dr. Edwards, a dermatologist who practices in Charlotte, N.C. “Also, for some reason we jump to yeast as a cause of diseases in the genital area. If the child is out of diapers and hasn’t reached puberty, it’s almost never yeast. Do a culture. Try and prove yeast. If it doesn’t respond to treatment for yeast, it’s not going to be yeast. Reassure, and don’t forget to reassure.”
. Lichen sclerosus presents classically as white, fragile plaques. “Textbooks say that there is cigarette paper-like crinkling of skin,” Dr. Edwards said. “I think of it being more like cellophane paper. In children, we often see it as smooth, kind of waxy and shiny, compared to adults. Children usually present with pruritus and irritation.”
Lichen sclerosus often starts in the clitoral area and on the perineum, and often with an edematous clitoral hood. “It often eventuates into clitoral phimosis, meaning that there is midline adhesion so that the clitoris is buried,” she said. “In adults, seeing this clitoral phimosis is a reliable sign of a scarring dermatosis – most often lichen sclerosus. But you can’t say that in children, because little girls will often have scarring over the clitoris. It’s just physiologic and means nothing, and it will go away at puberty. Certainly, sometimes this white discoloration can have crinkling. Purpura and tearing are common; if you look at lichen sclerosus histologically it looks like a thin epithelium that’s stretched over gelatin. Any rubbing and scratching can cause bleeding in the skin.”
Clinical appearance of well demarcated white skin with texture change drives the diagnosis. “It can be hard to tell from vitiligo at times, but there always should be texture change – whether it’s crinkling, whether it’s waxy, whether it’s smooth – and it’s symptomatic,” she said.
A biopsy is not usually required. “I think a good picture [of the affected area] or some sort of objective description in the chart is important, because most children do so well that in a few months there’s no sign of it, and the next provider [they see] may not believe that they ever had it,” she said.
The recommended initial treatment for lichen sclerosus in girls is a tiny amount of a superpotent topical corticosteroid ointment such as clobetasol or halobetasol one to two times daily until the skin is clear, which usually takes 2-4 months. “You do not treat these children until they’re comfortable, because that may be a week,” Dr. Edwards said. “You treat these children until the skin looks normal. Then you need to keep treating them, because if you don’t, the skin will relapse, even though they might not have symptoms.”
Following initial treatment, she recommends use of a superpotent corticosteroid once per day three times a week, or a midpotency steroid like triamcinolone ointment 0.1% every day. In her clinical experience, if lesions clear and remain clear with long-term treatment through puberty, the chances are good that they’ll stay clear if the medication is stopped.
“There are no studies on what to do after a patient clears,” said Dr. Edwards, chief of dermatology at Carolinas Medical Center, Charlotte, and adjunct clinical professor of dermatology at the University of North Carolina, Chapel Hill. “We have been informed by trial and error. If a child is totally clear after puberty, I will stop their medication and see them back every 3 months for about a year and a half. If they stay clear after a year and a half, I find that they stay clear. I wonder what happens at menopause. We surely don’t know.”
With consistent topical treatment, many patients will have clearing in one area of affected skin after a month or two, and it will take 3 or 4 months for the remaining area to clear. “I tend to see patients back every 6-8 weeks until they’re clear,” she said. “I do not like the idea of sending people out and saying, ‘use this medication twice a day for a month, then once a day for a month, then three times a week, then as needed.’
For patients concerned about the long-term use of topical steroids, the immunosuppressants tacrolimus and pimecrolimus are options. “They are often irritating on the vulva, but can work better than steroids for extragenital disease,” Dr. Edwards said. “Parents sometimes object to the use of a corticosteroid, but because these produce slower benefit and often burn with application, you can remind the parents that tacrolimus and pimecrolimus are not without side effects and are labeled as being associated with cancer. That often will prompt a parent to be willing to use a topical steroid. You can also point to studies that show the safety of topical steroids.”
Intralesional steroids are useful for thick lesions, but Dr. Edwards said that she has never had to use them in a child with lichen sclerosus. “I have found methotrexate to be useful in some people, but there is not one study on genital lichen sclerosus and methotrexate,” she said. “I find that about one in five patients with recalcitrant vulvar lichen sclerosus has had some benefit from methotrexate,” she added, noting that fractional CO2 laser “is showing promise in these patients.”
Dr. Edwards concluded her remarks by noting that she has never cared for a child with vulvar lichen sclerosus who didn’t respond to topical super potent steroids, “except due to poor compliance.”
She reported having no relevant financial disclosures.
In the clinical experience of Libby Edwards, MD, the diagnosis of lichen sclerosus in a young girl often triggers worry from patients and parents alike.
“The parents are worried about the ramifications of genital diseases and they’re worried about scarring,” she said during the virtual annual meeting of the Society for Pediatric Dermatology.
Meanwhile, during the initial assessment, physicians tend to think about sexual abuse or sexually transmitted diseases as the primary culprit. “It’s really important that you consider those issues, but they’re not usually what’s going on,” said Dr. Edwards, a dermatologist who practices in Charlotte, N.C. “Also, for some reason we jump to yeast as a cause of diseases in the genital area. If the child is out of diapers and hasn’t reached puberty, it’s almost never yeast. Do a culture. Try and prove yeast. If it doesn’t respond to treatment for yeast, it’s not going to be yeast. Reassure, and don’t forget to reassure.”
. Lichen sclerosus presents classically as white, fragile plaques. “Textbooks say that there is cigarette paper-like crinkling of skin,” Dr. Edwards said. “I think of it being more like cellophane paper. In children, we often see it as smooth, kind of waxy and shiny, compared to adults. Children usually present with pruritus and irritation.”
Lichen sclerosus often starts in the clitoral area and on the perineum, and often with an edematous clitoral hood. “It often eventuates into clitoral phimosis, meaning that there is midline adhesion so that the clitoris is buried,” she said. “In adults, seeing this clitoral phimosis is a reliable sign of a scarring dermatosis – most often lichen sclerosus. But you can’t say that in children, because little girls will often have scarring over the clitoris. It’s just physiologic and means nothing, and it will go away at puberty. Certainly, sometimes this white discoloration can have crinkling. Purpura and tearing are common; if you look at lichen sclerosus histologically it looks like a thin epithelium that’s stretched over gelatin. Any rubbing and scratching can cause bleeding in the skin.”
Clinical appearance of well demarcated white skin with texture change drives the diagnosis. “It can be hard to tell from vitiligo at times, but there always should be texture change – whether it’s crinkling, whether it’s waxy, whether it’s smooth – and it’s symptomatic,” she said.
A biopsy is not usually required. “I think a good picture [of the affected area] or some sort of objective description in the chart is important, because most children do so well that in a few months there’s no sign of it, and the next provider [they see] may not believe that they ever had it,” she said.
The recommended initial treatment for lichen sclerosus in girls is a tiny amount of a superpotent topical corticosteroid ointment such as clobetasol or halobetasol one to two times daily until the skin is clear, which usually takes 2-4 months. “You do not treat these children until they’re comfortable, because that may be a week,” Dr. Edwards said. “You treat these children until the skin looks normal. Then you need to keep treating them, because if you don’t, the skin will relapse, even though they might not have symptoms.”
Following initial treatment, she recommends use of a superpotent corticosteroid once per day three times a week, or a midpotency steroid like triamcinolone ointment 0.1% every day. In her clinical experience, if lesions clear and remain clear with long-term treatment through puberty, the chances are good that they’ll stay clear if the medication is stopped.
“There are no studies on what to do after a patient clears,” said Dr. Edwards, chief of dermatology at Carolinas Medical Center, Charlotte, and adjunct clinical professor of dermatology at the University of North Carolina, Chapel Hill. “We have been informed by trial and error. If a child is totally clear after puberty, I will stop their medication and see them back every 3 months for about a year and a half. If they stay clear after a year and a half, I find that they stay clear. I wonder what happens at menopause. We surely don’t know.”
With consistent topical treatment, many patients will have clearing in one area of affected skin after a month or two, and it will take 3 or 4 months for the remaining area to clear. “I tend to see patients back every 6-8 weeks until they’re clear,” she said. “I do not like the idea of sending people out and saying, ‘use this medication twice a day for a month, then once a day for a month, then three times a week, then as needed.’
For patients concerned about the long-term use of topical steroids, the immunosuppressants tacrolimus and pimecrolimus are options. “They are often irritating on the vulva, but can work better than steroids for extragenital disease,” Dr. Edwards said. “Parents sometimes object to the use of a corticosteroid, but because these produce slower benefit and often burn with application, you can remind the parents that tacrolimus and pimecrolimus are not without side effects and are labeled as being associated with cancer. That often will prompt a parent to be willing to use a topical steroid. You can also point to studies that show the safety of topical steroids.”
Intralesional steroids are useful for thick lesions, but Dr. Edwards said that she has never had to use them in a child with lichen sclerosus. “I have found methotrexate to be useful in some people, but there is not one study on genital lichen sclerosus and methotrexate,” she said. “I find that about one in five patients with recalcitrant vulvar lichen sclerosus has had some benefit from methotrexate,” she added, noting that fractional CO2 laser “is showing promise in these patients.”
Dr. Edwards concluded her remarks by noting that she has never cared for a child with vulvar lichen sclerosus who didn’t respond to topical super potent steroids, “except due to poor compliance.”
She reported having no relevant financial disclosures.
FROM SPD 2020
Antibiotic resistance: Personal responsibility in somewhat short supply
Most primary care physicians agree that antibiotic resistance and inappropriate prescribing are problems in the United States, but they are much less inclined to recognize these issues in their own practices, according to the results of a nationwide survey.
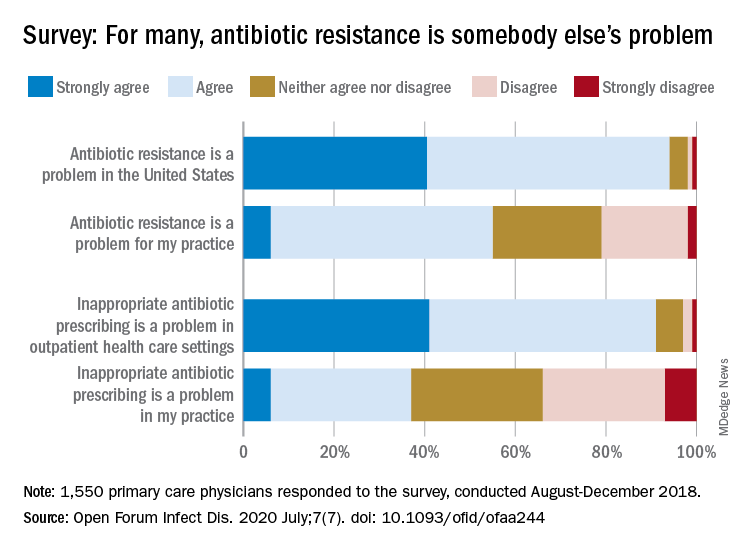
Rachel M. Zetts, MPH, of the Pew Charitable Trusts, Washington, D.C., and associates wrote in Open Forum Infectious Diseases.
Almost all (94%) of the 1,550 internists, family physicians, and pediatricians who responded to the survey said that antibiotic resistance is a national problem, and nearly that many (91%) agreed that “inappropriate antibiotic prescribing is a problem in outpatient health care settings,” the investigators acknowledged.
Narrowing the focus to their own practices, however, changed some opinions. At that level, only 55% of the respondents said that resistance was a problem for their practices, and just 37% said that there any sort of inappropriate prescribing going on, based on data from the survey, which was conducted from August to October 2018 by Pew and the American Medical Association.
Antibiotic stewardship, defined as activities meant to ensure appropriate prescribing of antibiotics, should include “staff and patient education, clinician-level antibiotic prescribing feedback, and communications training on how to discuss antibiotic prescribing with patients,” Ms. Zetts and associates explained.
The need for such stewardship in health care settings was acknowledged by 72% of respondents, but 53% of those surveyed also said that all they need to do to support such efforts “is to talk with their patients about the value of an antibiotic for their symptoms,” they noted.
The bacteria, it seems, are not the only ones with some resistance. Half of the primary care physicians believe that it would be difficult to fairly and accurately track the appropriate use of antibiotics, and 52% agreed with the statement that “practice-based reporting requirements for antibiotic use would be too onerous,” the researchers pointed out.
“Antibiotic resistance is an impending public health crisis. We are seeing today, as we respond to the COVID-19 pandemic, what our health system looks like with no or limited treatments available to tackle an outbreak. … We must all remain vigilant in combating the spread of antibiotic resistant bacteria and be prudent when prescribing antibiotics,” AMA President Susan R. Bailey, MD, said in a written statement.
SOURCE: Zetts RM et al. Open Forum Infect Dis. 2020 July;7(7). doi: 10.1093/ofid/ofaa244.
Most primary care physicians agree that antibiotic resistance and inappropriate prescribing are problems in the United States, but they are much less inclined to recognize these issues in their own practices, according to the results of a nationwide survey.

Rachel M. Zetts, MPH, of the Pew Charitable Trusts, Washington, D.C., and associates wrote in Open Forum Infectious Diseases.
Almost all (94%) of the 1,550 internists, family physicians, and pediatricians who responded to the survey said that antibiotic resistance is a national problem, and nearly that many (91%) agreed that “inappropriate antibiotic prescribing is a problem in outpatient health care settings,” the investigators acknowledged.
Narrowing the focus to their own practices, however, changed some opinions. At that level, only 55% of the respondents said that resistance was a problem for their practices, and just 37% said that there any sort of inappropriate prescribing going on, based on data from the survey, which was conducted from August to October 2018 by Pew and the American Medical Association.
Antibiotic stewardship, defined as activities meant to ensure appropriate prescribing of antibiotics, should include “staff and patient education, clinician-level antibiotic prescribing feedback, and communications training on how to discuss antibiotic prescribing with patients,” Ms. Zetts and associates explained.
The need for such stewardship in health care settings was acknowledged by 72% of respondents, but 53% of those surveyed also said that all they need to do to support such efforts “is to talk with their patients about the value of an antibiotic for their symptoms,” they noted.
The bacteria, it seems, are not the only ones with some resistance. Half of the primary care physicians believe that it would be difficult to fairly and accurately track the appropriate use of antibiotics, and 52% agreed with the statement that “practice-based reporting requirements for antibiotic use would be too onerous,” the researchers pointed out.
“Antibiotic resistance is an impending public health crisis. We are seeing today, as we respond to the COVID-19 pandemic, what our health system looks like with no or limited treatments available to tackle an outbreak. … We must all remain vigilant in combating the spread of antibiotic resistant bacteria and be prudent when prescribing antibiotics,” AMA President Susan R. Bailey, MD, said in a written statement.
SOURCE: Zetts RM et al. Open Forum Infect Dis. 2020 July;7(7). doi: 10.1093/ofid/ofaa244.
Most primary care physicians agree that antibiotic resistance and inappropriate prescribing are problems in the United States, but they are much less inclined to recognize these issues in their own practices, according to the results of a nationwide survey.

Rachel M. Zetts, MPH, of the Pew Charitable Trusts, Washington, D.C., and associates wrote in Open Forum Infectious Diseases.
Almost all (94%) of the 1,550 internists, family physicians, and pediatricians who responded to the survey said that antibiotic resistance is a national problem, and nearly that many (91%) agreed that “inappropriate antibiotic prescribing is a problem in outpatient health care settings,” the investigators acknowledged.
Narrowing the focus to their own practices, however, changed some opinions. At that level, only 55% of the respondents said that resistance was a problem for their practices, and just 37% said that there any sort of inappropriate prescribing going on, based on data from the survey, which was conducted from August to October 2018 by Pew and the American Medical Association.
Antibiotic stewardship, defined as activities meant to ensure appropriate prescribing of antibiotics, should include “staff and patient education, clinician-level antibiotic prescribing feedback, and communications training on how to discuss antibiotic prescribing with patients,” Ms. Zetts and associates explained.
The need for such stewardship in health care settings was acknowledged by 72% of respondents, but 53% of those surveyed also said that all they need to do to support such efforts “is to talk with their patients about the value of an antibiotic for their symptoms,” they noted.
The bacteria, it seems, are not the only ones with some resistance. Half of the primary care physicians believe that it would be difficult to fairly and accurately track the appropriate use of antibiotics, and 52% agreed with the statement that “practice-based reporting requirements for antibiotic use would be too onerous,” the researchers pointed out.
“Antibiotic resistance is an impending public health crisis. We are seeing today, as we respond to the COVID-19 pandemic, what our health system looks like with no or limited treatments available to tackle an outbreak. … We must all remain vigilant in combating the spread of antibiotic resistant bacteria and be prudent when prescribing antibiotics,” AMA President Susan R. Bailey, MD, said in a written statement.
SOURCE: Zetts RM et al. Open Forum Infect Dis. 2020 July;7(7). doi: 10.1093/ofid/ofaa244.
FROM OPEN FORUM INFECTIOUS DISEASES
Pandemic hampers reopening of joint replacement gold mine
Dr. Ira Weintraub, a recently retired orthopedic surgeon who now works at a medical billing consultancy, saw a hip replacement bill for over $400,000 earlier this year.
“The patient stayed in the hospital 17 days, which is only 17 times normal. The bill got paid,” mused Weintraub, chief medical officer of Portland, Oregon-based WellRithms, which helps self-funded employers and workers’ compensation insurers make sense of large, complex medical bills and ensure they pay the fair amount.
Charges like that go a long way toward explaining why hospitals are eager to restore joint replacements to pre-COVID levels as quickly as possible – an eagerness tempered only by safety concerns amid a resurgence of the coronavirus in some regions of the country. Revenue losses at hospitals and outpatient surgery centers may have exceeded $5 billion from canceled knee and hip replacements alone during a roughly two-month hiatus on elective procedures earlier this year.
The cost of joint replacement surgery varies widely – though, on average, it is in the tens, not hundreds, of thousands of dollars. Still, given the high and rapidly growing volume, it’s easy to see why joint replacement operations have become a vital chunk of revenue at most U.S. hospitals.
The rate of knee and hip replacements more than doubled from 2000 to 2015, according to inpatient discharge data from the Agency for Healthcare Research and Quality. And that growth is likely to continue: Knee replacements are expected to triple between now and 2040, with hip replacements not far behind, according to projections published last year in the Journal of Rheumatology.
Joint procedures are usually not emergencies, and they were among the first to be scrubbed or delayed when hospitals froze elective surgeries in March – and again in July in some areas plagued by renewed COVID outbreaks.
“Without orthopedic volumes returning to something near their pre-pandemic levels, it will make it difficult for health systems to get back to anywhere near break-even from a bottom-line perspective,” said Stephen Thome, a principal in health care consulting at Grant Thornton, an advisory, audit and tax firm.
It’s impossible to know exactly how much knee and hip replacements are worth to hospitals, because no definitive data on total volume or price exists.
But using published estimates of volume, extrapolating average commercial payments from published Medicare rates based on a study, and making an educated guess of patient coinsurance, Thome helped KHN arrive at an annual market value for American hospitals and surgery centers of between $15.5 billion and $21.5 billion for knee replacements alone.
That suggests a revenue loss of $1.3 billion to $1.8 billion per month for the period the surgeries were shut down. These figures include ambulatory surgery centers not owned by hospitals, which also suspended most operations in late March, all of April and into May.
If you add hip replacements, which account for about half the volume of knees and are paid at similar rates, the total annual value rises to a range of $23 billion to $32 billion, with monthly revenue losses from $1.9 billion to $2.7 billion.
The American Hospital Association projects total revenue lost at U.S. hospitals will reach $323 billion by year’s end, not counting additional losses from surgeries canceled during the current coronavirus spike. That amount is partially offset by $69 billion in federal relief dollars hospitals have received so far, according to the association. The California Hospital Association puts the net revenue loss for hospitals in that state at about $10.5 billion, said spokesperson Jan Emerson-Shea.
Hospitals resumed joint replacement surgeries in early to mid-May, with the timing and ramp-up speed varying by region and hospital. Some hospitals restored volume quickly; others took a more cautious route and continue to lose revenue. Still others have had to shut down again.
At the NYU Langone Orthopedic Hospital in New York City, “people are starting to come in and you see the operating rooms full again,” said Dr. Claudette Lajam, chief orthopedic safety officer.
At St. Jude Medical Center in Fullerton, California, where the coronavirus is raging, inpatient joint replacements resumed in the second or third week of May – cautiously at first, but volume is “very close to pre-pandemic levels at this point,” said Dr. Kevin Khajavi, chairman of the hospital’s orthopedic surgery department. However, “we are constantly monitoring the situation to determine if we have to scale back once again,” he said.
In large swaths of Texas, elective surgeries were once again suspended in July because of the COVID-19 resurgence. The same is true at many hospitals in Florida, Alabama, South Carolina and Nevada.
The Mayo Clinic in Phoenix suspended nonemergency joint replacement surgeries in early July. It resumed outpatient replacement procedures the week of July 27, but still has not resumed nonemergency inpatient procedures, said Dr. Mark Spangehl, an orthopedic surgeon there. In terms of medical urgency, joint replacements are “at the bottom of the totem pole,” Spangehl said.
In terms of cash flow, however, joint replacements are decidedly not at the bottom of the totem pole. They have become a cash cow as the number of patients undergoing them has skyrocketed in recent decades.
The volume is being driven by an aging population, an epidemic of obesity and a significant rise in the number of younger people replacing joints worn out by years of sports and exercise.
It’s also being driven by the cash. Once only done in hospitals, the operations are now increasingly performed at ambulatory surgery centers – especially on younger, healthier patients who don’t require hospitalization.
The surgery centers are often physician-owned, but private equity groups such as Bain Capital and KKR & Co. have taken an interest in them, drawn by their high growth potential, robust financial returns and ability to offer competitive prices.
“[G]enerally the savings should be very good – but I do see a lot of outlier surgery centers where they are charging exorbitant amounts of money – $100,000 wouldn’t be too much,” said WellRithm’s Weintraub, who co-owned such a surgery center in Portland.
Fear of catching the coronavirus in a hospital is reinforcing the outpatient trend. Matthew Davis, a 58-year-old resident of Washington, was scheduled for a hip replacement on March 30 but got cold feet because of COVID-19, and canceled just before all elective surgeries were halted. When it came time to reschedule in June, he overcame his reservations in large part because the surgeon planned to perform the procedure at a free-standing surgery center.
“That was key to me – avoiding an overnight hospital stay to minimize my exposure,” Davis said. “These joint replacements are almost industrial-scale. They are cranking out joint replacements 9 to 5. I went in at 6:30 a.m. and I was walking out the door at 11:30.”
Acutely aware of the financial benefits, hospitals and surgery clinics have been marketing joint replacements for years, competing for coveted rankings and running ads that show healthy aging people, all smiles, engaged in vigorous activity.
However, a 2014 study concluded that one-third of knee replacements were not warranted, mainly because the symptoms of the patients were not severe enough to justify the procedures.
“The whole marketing of health care is so manipulative to the consuming public,” said Lisa McGiffert, a longtime consumer advocate and co-founder of the Patient Safety Action Network. “People might be encouraged to get a knee replacement, when in reality something less invasive could have improved their condition.”
McGiffert recounted a conversation with an orthopedic surgeon in Washington state who told her about a patient who requested a knee replacement, even though he had not tried any lower-impact treatments to fix the problem. “I asked the surgeon, ‘You didn’t do it, did you?’ And he said, ‘Of course I did. He would just have gone to somebody else.’ ”
This Kaiser Health News story first published on California Healthline, a service of the California Health Care Foundation.
Dr. Ira Weintraub, a recently retired orthopedic surgeon who now works at a medical billing consultancy, saw a hip replacement bill for over $400,000 earlier this year.
“The patient stayed in the hospital 17 days, which is only 17 times normal. The bill got paid,” mused Weintraub, chief medical officer of Portland, Oregon-based WellRithms, which helps self-funded employers and workers’ compensation insurers make sense of large, complex medical bills and ensure they pay the fair amount.
Charges like that go a long way toward explaining why hospitals are eager to restore joint replacements to pre-COVID levels as quickly as possible – an eagerness tempered only by safety concerns amid a resurgence of the coronavirus in some regions of the country. Revenue losses at hospitals and outpatient surgery centers may have exceeded $5 billion from canceled knee and hip replacements alone during a roughly two-month hiatus on elective procedures earlier this year.
The cost of joint replacement surgery varies widely – though, on average, it is in the tens, not hundreds, of thousands of dollars. Still, given the high and rapidly growing volume, it’s easy to see why joint replacement operations have become a vital chunk of revenue at most U.S. hospitals.
The rate of knee and hip replacements more than doubled from 2000 to 2015, according to inpatient discharge data from the Agency for Healthcare Research and Quality. And that growth is likely to continue: Knee replacements are expected to triple between now and 2040, with hip replacements not far behind, according to projections published last year in the Journal of Rheumatology.
Joint procedures are usually not emergencies, and they were among the first to be scrubbed or delayed when hospitals froze elective surgeries in March – and again in July in some areas plagued by renewed COVID outbreaks.
“Without orthopedic volumes returning to something near their pre-pandemic levels, it will make it difficult for health systems to get back to anywhere near break-even from a bottom-line perspective,” said Stephen Thome, a principal in health care consulting at Grant Thornton, an advisory, audit and tax firm.
It’s impossible to know exactly how much knee and hip replacements are worth to hospitals, because no definitive data on total volume or price exists.
But using published estimates of volume, extrapolating average commercial payments from published Medicare rates based on a study, and making an educated guess of patient coinsurance, Thome helped KHN arrive at an annual market value for American hospitals and surgery centers of between $15.5 billion and $21.5 billion for knee replacements alone.
That suggests a revenue loss of $1.3 billion to $1.8 billion per month for the period the surgeries were shut down. These figures include ambulatory surgery centers not owned by hospitals, which also suspended most operations in late March, all of April and into May.
If you add hip replacements, which account for about half the volume of knees and are paid at similar rates, the total annual value rises to a range of $23 billion to $32 billion, with monthly revenue losses from $1.9 billion to $2.7 billion.
The American Hospital Association projects total revenue lost at U.S. hospitals will reach $323 billion by year’s end, not counting additional losses from surgeries canceled during the current coronavirus spike. That amount is partially offset by $69 billion in federal relief dollars hospitals have received so far, according to the association. The California Hospital Association puts the net revenue loss for hospitals in that state at about $10.5 billion, said spokesperson Jan Emerson-Shea.
Hospitals resumed joint replacement surgeries in early to mid-May, with the timing and ramp-up speed varying by region and hospital. Some hospitals restored volume quickly; others took a more cautious route and continue to lose revenue. Still others have had to shut down again.
At the NYU Langone Orthopedic Hospital in New York City, “people are starting to come in and you see the operating rooms full again,” said Dr. Claudette Lajam, chief orthopedic safety officer.
At St. Jude Medical Center in Fullerton, California, where the coronavirus is raging, inpatient joint replacements resumed in the second or third week of May – cautiously at first, but volume is “very close to pre-pandemic levels at this point,” said Dr. Kevin Khajavi, chairman of the hospital’s orthopedic surgery department. However, “we are constantly monitoring the situation to determine if we have to scale back once again,” he said.
In large swaths of Texas, elective surgeries were once again suspended in July because of the COVID-19 resurgence. The same is true at many hospitals in Florida, Alabama, South Carolina and Nevada.
The Mayo Clinic in Phoenix suspended nonemergency joint replacement surgeries in early July. It resumed outpatient replacement procedures the week of July 27, but still has not resumed nonemergency inpatient procedures, said Dr. Mark Spangehl, an orthopedic surgeon there. In terms of medical urgency, joint replacements are “at the bottom of the totem pole,” Spangehl said.
In terms of cash flow, however, joint replacements are decidedly not at the bottom of the totem pole. They have become a cash cow as the number of patients undergoing them has skyrocketed in recent decades.
The volume is being driven by an aging population, an epidemic of obesity and a significant rise in the number of younger people replacing joints worn out by years of sports and exercise.
It’s also being driven by the cash. Once only done in hospitals, the operations are now increasingly performed at ambulatory surgery centers – especially on younger, healthier patients who don’t require hospitalization.
The surgery centers are often physician-owned, but private equity groups such as Bain Capital and KKR & Co. have taken an interest in them, drawn by their high growth potential, robust financial returns and ability to offer competitive prices.
“[G]enerally the savings should be very good – but I do see a lot of outlier surgery centers where they are charging exorbitant amounts of money – $100,000 wouldn’t be too much,” said WellRithm’s Weintraub, who co-owned such a surgery center in Portland.
Fear of catching the coronavirus in a hospital is reinforcing the outpatient trend. Matthew Davis, a 58-year-old resident of Washington, was scheduled for a hip replacement on March 30 but got cold feet because of COVID-19, and canceled just before all elective surgeries were halted. When it came time to reschedule in June, he overcame his reservations in large part because the surgeon planned to perform the procedure at a free-standing surgery center.
“That was key to me – avoiding an overnight hospital stay to minimize my exposure,” Davis said. “These joint replacements are almost industrial-scale. They are cranking out joint replacements 9 to 5. I went in at 6:30 a.m. and I was walking out the door at 11:30.”
Acutely aware of the financial benefits, hospitals and surgery clinics have been marketing joint replacements for years, competing for coveted rankings and running ads that show healthy aging people, all smiles, engaged in vigorous activity.
However, a 2014 study concluded that one-third of knee replacements were not warranted, mainly because the symptoms of the patients were not severe enough to justify the procedures.
“The whole marketing of health care is so manipulative to the consuming public,” said Lisa McGiffert, a longtime consumer advocate and co-founder of the Patient Safety Action Network. “People might be encouraged to get a knee replacement, when in reality something less invasive could have improved their condition.”
McGiffert recounted a conversation with an orthopedic surgeon in Washington state who told her about a patient who requested a knee replacement, even though he had not tried any lower-impact treatments to fix the problem. “I asked the surgeon, ‘You didn’t do it, did you?’ And he said, ‘Of course I did. He would just have gone to somebody else.’ ”
This Kaiser Health News story first published on California Healthline, a service of the California Health Care Foundation.
Dr. Ira Weintraub, a recently retired orthopedic surgeon who now works at a medical billing consultancy, saw a hip replacement bill for over $400,000 earlier this year.
“The patient stayed in the hospital 17 days, which is only 17 times normal. The bill got paid,” mused Weintraub, chief medical officer of Portland, Oregon-based WellRithms, which helps self-funded employers and workers’ compensation insurers make sense of large, complex medical bills and ensure they pay the fair amount.
Charges like that go a long way toward explaining why hospitals are eager to restore joint replacements to pre-COVID levels as quickly as possible – an eagerness tempered only by safety concerns amid a resurgence of the coronavirus in some regions of the country. Revenue losses at hospitals and outpatient surgery centers may have exceeded $5 billion from canceled knee and hip replacements alone during a roughly two-month hiatus on elective procedures earlier this year.
The cost of joint replacement surgery varies widely – though, on average, it is in the tens, not hundreds, of thousands of dollars. Still, given the high and rapidly growing volume, it’s easy to see why joint replacement operations have become a vital chunk of revenue at most U.S. hospitals.
The rate of knee and hip replacements more than doubled from 2000 to 2015, according to inpatient discharge data from the Agency for Healthcare Research and Quality. And that growth is likely to continue: Knee replacements are expected to triple between now and 2040, with hip replacements not far behind, according to projections published last year in the Journal of Rheumatology.
Joint procedures are usually not emergencies, and they were among the first to be scrubbed or delayed when hospitals froze elective surgeries in March – and again in July in some areas plagued by renewed COVID outbreaks.
“Without orthopedic volumes returning to something near their pre-pandemic levels, it will make it difficult for health systems to get back to anywhere near break-even from a bottom-line perspective,” said Stephen Thome, a principal in health care consulting at Grant Thornton, an advisory, audit and tax firm.
It’s impossible to know exactly how much knee and hip replacements are worth to hospitals, because no definitive data on total volume or price exists.
But using published estimates of volume, extrapolating average commercial payments from published Medicare rates based on a study, and making an educated guess of patient coinsurance, Thome helped KHN arrive at an annual market value for American hospitals and surgery centers of between $15.5 billion and $21.5 billion for knee replacements alone.
That suggests a revenue loss of $1.3 billion to $1.8 billion per month for the period the surgeries were shut down. These figures include ambulatory surgery centers not owned by hospitals, which also suspended most operations in late March, all of April and into May.
If you add hip replacements, which account for about half the volume of knees and are paid at similar rates, the total annual value rises to a range of $23 billion to $32 billion, with monthly revenue losses from $1.9 billion to $2.7 billion.
The American Hospital Association projects total revenue lost at U.S. hospitals will reach $323 billion by year’s end, not counting additional losses from surgeries canceled during the current coronavirus spike. That amount is partially offset by $69 billion in federal relief dollars hospitals have received so far, according to the association. The California Hospital Association puts the net revenue loss for hospitals in that state at about $10.5 billion, said spokesperson Jan Emerson-Shea.
Hospitals resumed joint replacement surgeries in early to mid-May, with the timing and ramp-up speed varying by region and hospital. Some hospitals restored volume quickly; others took a more cautious route and continue to lose revenue. Still others have had to shut down again.
At the NYU Langone Orthopedic Hospital in New York City, “people are starting to come in and you see the operating rooms full again,” said Dr. Claudette Lajam, chief orthopedic safety officer.
At St. Jude Medical Center in Fullerton, California, where the coronavirus is raging, inpatient joint replacements resumed in the second or third week of May – cautiously at first, but volume is “very close to pre-pandemic levels at this point,” said Dr. Kevin Khajavi, chairman of the hospital’s orthopedic surgery department. However, “we are constantly monitoring the situation to determine if we have to scale back once again,” he said.
In large swaths of Texas, elective surgeries were once again suspended in July because of the COVID-19 resurgence. The same is true at many hospitals in Florida, Alabama, South Carolina and Nevada.
The Mayo Clinic in Phoenix suspended nonemergency joint replacement surgeries in early July. It resumed outpatient replacement procedures the week of July 27, but still has not resumed nonemergency inpatient procedures, said Dr. Mark Spangehl, an orthopedic surgeon there. In terms of medical urgency, joint replacements are “at the bottom of the totem pole,” Spangehl said.
In terms of cash flow, however, joint replacements are decidedly not at the bottom of the totem pole. They have become a cash cow as the number of patients undergoing them has skyrocketed in recent decades.
The volume is being driven by an aging population, an epidemic of obesity and a significant rise in the number of younger people replacing joints worn out by years of sports and exercise.
It’s also being driven by the cash. Once only done in hospitals, the operations are now increasingly performed at ambulatory surgery centers – especially on younger, healthier patients who don’t require hospitalization.
The surgery centers are often physician-owned, but private equity groups such as Bain Capital and KKR & Co. have taken an interest in them, drawn by their high growth potential, robust financial returns and ability to offer competitive prices.
“[G]enerally the savings should be very good – but I do see a lot of outlier surgery centers where they are charging exorbitant amounts of money – $100,000 wouldn’t be too much,” said WellRithm’s Weintraub, who co-owned such a surgery center in Portland.
Fear of catching the coronavirus in a hospital is reinforcing the outpatient trend. Matthew Davis, a 58-year-old resident of Washington, was scheduled for a hip replacement on March 30 but got cold feet because of COVID-19, and canceled just before all elective surgeries were halted. When it came time to reschedule in June, he overcame his reservations in large part because the surgeon planned to perform the procedure at a free-standing surgery center.
“That was key to me – avoiding an overnight hospital stay to minimize my exposure,” Davis said. “These joint replacements are almost industrial-scale. They are cranking out joint replacements 9 to 5. I went in at 6:30 a.m. and I was walking out the door at 11:30.”
Acutely aware of the financial benefits, hospitals and surgery clinics have been marketing joint replacements for years, competing for coveted rankings and running ads that show healthy aging people, all smiles, engaged in vigorous activity.
However, a 2014 study concluded that one-third of knee replacements were not warranted, mainly because the symptoms of the patients were not severe enough to justify the procedures.
“The whole marketing of health care is so manipulative to the consuming public,” said Lisa McGiffert, a longtime consumer advocate and co-founder of the Patient Safety Action Network. “People might be encouraged to get a knee replacement, when in reality something less invasive could have improved their condition.”
McGiffert recounted a conversation with an orthopedic surgeon in Washington state who told her about a patient who requested a knee replacement, even though he had not tried any lower-impact treatments to fix the problem. “I asked the surgeon, ‘You didn’t do it, did you?’ And he said, ‘Of course I did. He would just have gone to somebody else.’ ”
This Kaiser Health News story first published on California Healthline, a service of the California Health Care Foundation.
Experimental nonstimulant effective, fast-acting for ADHD
The experimental nonstimulant medication viloxazine extended-release, known as SPN-812, reduced symptoms of attention-deficit/hyperactivity disorder (ADHD) as soon as 1 week after dosing and was well tolerated in a randomized, placebo-controlled phase 3 study that included more than 400 children.
In addition to its fast onset of action, the fact that it was effective for both inattentive and hyperactive/impulsive clusters of symptoms is “impressive,” study investigator Andrew Cutler, MD, clinical professor of psychiatry, SUNY Upstate Medical University, Syracuse, N.Y., said in an interviews.
Also noteworthy was the improvement in measures of quality of life and function, “especially function in the areas of school, home life, family relations, and peer relationships, which can be really disrupted with ADHD,” Dr. Cutler said.
The findings were published online July 25 in Clinical Therapeutics.
Novel modulating agent
Viloxazine extended-release is a novel multimodal serotonergic and noradrenergic modulating agent with activity at serotonin receptors and the norepinephrine transporter.
About two-thirds were boys.
All participants had an ADHD-Rating Scale–5 (ADHD-RS-5) score of at least 28 and a Clinical Global Impression–Severity score of at least 4. None had taken ADHD medication for at least 1 week prior to randomization.
The intent-to-treat population included 460 children. Of these, 155 were randomly assigned to receive placebo, 147 to receive viloxazine 100 mg, and 158 to receive viloxazine 200 mg.
The primary efficacy endpoint was change from baseline in ADHD-RS-5 total score at week 6. Score changes for both the 100-mg (P = .0004) and the 200-mg (P < .0001) viloxazine groups met statistical significance compared with the placebo group.
Change from baseline in both the ADHD-RS-5 inattention and hyperactivity/impulsivity subscale scores was also significantly reduced in the 100-mg (P = .0006 and .0026, respectively) and 200-mg (P < .0001 and P < .0001, respectively) treatment groups compared with the placebo group.
Improvements occurred after 1 week of treatment and were maintained throughout the 6-week trial, “indicating an early and sustained effect,” the investigators wrote.
FDA target action date
The Clinical Global Impression–Improvement (CGI-I) score at 6 weeks was significantly improved in those receiving 100 mg (P = .0020) and 200 mg (P < .0001) of the active treatment compared with placebo.
The CGI-I responder rate, the percentage of children with a CGI-I score of 1 (very much improved) or 2 (much improved), was significantly higher at 6 weeks with viloxazine 100 mg and 200 mg vs. placebo (45% and 51% vs. 30%, respectively; P = .0065 and P = .0002).
These standard investigator-rated assessments were supported by two parent self-rated assessments: the Conners 3–Parent Short Form and the Weiss Functional Impairment Rating Scale–Parent Form.
Parents noted improvement not only in their children’s ADHD symptoms but also in ADHD-associated learning problems, executive functioning, defiance/aggression, peer relations, and functioning in different settings.
At both doses, once-daily viloxazine was generally well tolerated, with a low rate of discontinuation because of adverse events (<5%). Most adverse events were characterized as mild or moderate in severity and included somnolence (8.9%), decreased appetite (6.0%), and headache (5.4%).
On the basis of results of this study and others, the Food and Drug Administration accepted the company’s new drug application for viloxazine extended-release for ADHD in children and adolescents. The application has a target action date of Nov. 8, 2020.
Potential advantages
Commenting on the study in an interview, Dean Elbe, PharmD, clinical pharmacy specialist, child and adolescent mental health, BC Children’s Hospital, Vancouver, B.C., said that use of viloxazine to treat ADHD is “interesting.”
Dr. Elbe, who was not involved with the current research, noted that “it is actually an old drug that has been around since the mid-1970s in Europe as an antidepressant. It was removed from the market due to poor sales, not safety issues.”
Overall, on the basis of this study, viloxazine has potential to offer “modest improvements” over atomoxetine (Strattera), and the dosing may be “more straightforward and somewhat less challenging than with atomoxetine, with no taper up and no adjustment for poor 2D6 metabolizers,” Dr. Elbe noted.
“The onset of action appears somewhat quicker than we typically see with atomoxetine, so that is also helpful for parent and clinician acceptance and partially overcomes a perceived barrier with atomoxetine,” he said.
Dr. Elbe said he wonders, however, whether viloxazine will show “real-world clinical utility for both hyperactive-impulsive as well as inattentive symptoms. Although the study shows efficacy in both symptom clusters, so did the atomoxetine RCTs, and this has not been the clinical impression for atomoxetine.”
The study was funded by Supernus Pharmaceuticals. Dr. Cutler is a consultant for Supernus, as well as for Adlon Therapeutics, Aevi Genomics, Akili Interactive, Arbor Pharmaceuticals, Ironshore, KemPharm, Lundbeck, Neos Therapeutics, NLS Pharma, Otsuka, Purdue, Shire, Sunovion, Takeda, and Tris Pharma. He has received speaker/promotional honoraria from Adlon Therapeutics, Arbor Pharmaceuticals, Lundbeck, Neos Therapeutics, Otsuka, Shire, Sunovion, Takeda, and Tris Pharma and has received research grants from Aevi Genomics, Akili Interactive, Arbor Pharmaceuticals, Ironshore, KemPharm, Lundbeck, Neos Therapeutics, Otsuka, Purdue, Shire, Sunovion, Supernus Pharmaceuticals, Takeda, and Tris Pharma. A complete list of disclosures for the other authors is available in the original article. Dr. Elbe has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The experimental nonstimulant medication viloxazine extended-release, known as SPN-812, reduced symptoms of attention-deficit/hyperactivity disorder (ADHD) as soon as 1 week after dosing and was well tolerated in a randomized, placebo-controlled phase 3 study that included more than 400 children.
In addition to its fast onset of action, the fact that it was effective for both inattentive and hyperactive/impulsive clusters of symptoms is “impressive,” study investigator Andrew Cutler, MD, clinical professor of psychiatry, SUNY Upstate Medical University, Syracuse, N.Y., said in an interviews.
Also noteworthy was the improvement in measures of quality of life and function, “especially function in the areas of school, home life, family relations, and peer relationships, which can be really disrupted with ADHD,” Dr. Cutler said.
The findings were published online July 25 in Clinical Therapeutics.
Novel modulating agent
Viloxazine extended-release is a novel multimodal serotonergic and noradrenergic modulating agent with activity at serotonin receptors and the norepinephrine transporter.
About two-thirds were boys.
All participants had an ADHD-Rating Scale–5 (ADHD-RS-5) score of at least 28 and a Clinical Global Impression–Severity score of at least 4. None had taken ADHD medication for at least 1 week prior to randomization.
The intent-to-treat population included 460 children. Of these, 155 were randomly assigned to receive placebo, 147 to receive viloxazine 100 mg, and 158 to receive viloxazine 200 mg.
The primary efficacy endpoint was change from baseline in ADHD-RS-5 total score at week 6. Score changes for both the 100-mg (P = .0004) and the 200-mg (P < .0001) viloxazine groups met statistical significance compared with the placebo group.
Change from baseline in both the ADHD-RS-5 inattention and hyperactivity/impulsivity subscale scores was also significantly reduced in the 100-mg (P = .0006 and .0026, respectively) and 200-mg (P < .0001 and P < .0001, respectively) treatment groups compared with the placebo group.
Improvements occurred after 1 week of treatment and were maintained throughout the 6-week trial, “indicating an early and sustained effect,” the investigators wrote.
FDA target action date
The Clinical Global Impression–Improvement (CGI-I) score at 6 weeks was significantly improved in those receiving 100 mg (P = .0020) and 200 mg (P < .0001) of the active treatment compared with placebo.
The CGI-I responder rate, the percentage of children with a CGI-I score of 1 (very much improved) or 2 (much improved), was significantly higher at 6 weeks with viloxazine 100 mg and 200 mg vs. placebo (45% and 51% vs. 30%, respectively; P = .0065 and P = .0002).
These standard investigator-rated assessments were supported by two parent self-rated assessments: the Conners 3–Parent Short Form and the Weiss Functional Impairment Rating Scale–Parent Form.
Parents noted improvement not only in their children’s ADHD symptoms but also in ADHD-associated learning problems, executive functioning, defiance/aggression, peer relations, and functioning in different settings.
At both doses, once-daily viloxazine was generally well tolerated, with a low rate of discontinuation because of adverse events (<5%). Most adverse events were characterized as mild or moderate in severity and included somnolence (8.9%), decreased appetite (6.0%), and headache (5.4%).
On the basis of results of this study and others, the Food and Drug Administration accepted the company’s new drug application for viloxazine extended-release for ADHD in children and adolescents. The application has a target action date of Nov. 8, 2020.
Potential advantages
Commenting on the study in an interview, Dean Elbe, PharmD, clinical pharmacy specialist, child and adolescent mental health, BC Children’s Hospital, Vancouver, B.C., said that use of viloxazine to treat ADHD is “interesting.”
Dr. Elbe, who was not involved with the current research, noted that “it is actually an old drug that has been around since the mid-1970s in Europe as an antidepressant. It was removed from the market due to poor sales, not safety issues.”
Overall, on the basis of this study, viloxazine has potential to offer “modest improvements” over atomoxetine (Strattera), and the dosing may be “more straightforward and somewhat less challenging than with atomoxetine, with no taper up and no adjustment for poor 2D6 metabolizers,” Dr. Elbe noted.
“The onset of action appears somewhat quicker than we typically see with atomoxetine, so that is also helpful for parent and clinician acceptance and partially overcomes a perceived barrier with atomoxetine,” he said.
Dr. Elbe said he wonders, however, whether viloxazine will show “real-world clinical utility for both hyperactive-impulsive as well as inattentive symptoms. Although the study shows efficacy in both symptom clusters, so did the atomoxetine RCTs, and this has not been the clinical impression for atomoxetine.”
The study was funded by Supernus Pharmaceuticals. Dr. Cutler is a consultant for Supernus, as well as for Adlon Therapeutics, Aevi Genomics, Akili Interactive, Arbor Pharmaceuticals, Ironshore, KemPharm, Lundbeck, Neos Therapeutics, NLS Pharma, Otsuka, Purdue, Shire, Sunovion, Takeda, and Tris Pharma. He has received speaker/promotional honoraria from Adlon Therapeutics, Arbor Pharmaceuticals, Lundbeck, Neos Therapeutics, Otsuka, Shire, Sunovion, Takeda, and Tris Pharma and has received research grants from Aevi Genomics, Akili Interactive, Arbor Pharmaceuticals, Ironshore, KemPharm, Lundbeck, Neos Therapeutics, Otsuka, Purdue, Shire, Sunovion, Supernus Pharmaceuticals, Takeda, and Tris Pharma. A complete list of disclosures for the other authors is available in the original article. Dr. Elbe has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The experimental nonstimulant medication viloxazine extended-release, known as SPN-812, reduced symptoms of attention-deficit/hyperactivity disorder (ADHD) as soon as 1 week after dosing and was well tolerated in a randomized, placebo-controlled phase 3 study that included more than 400 children.
In addition to its fast onset of action, the fact that it was effective for both inattentive and hyperactive/impulsive clusters of symptoms is “impressive,” study investigator Andrew Cutler, MD, clinical professor of psychiatry, SUNY Upstate Medical University, Syracuse, N.Y., said in an interviews.
Also noteworthy was the improvement in measures of quality of life and function, “especially function in the areas of school, home life, family relations, and peer relationships, which can be really disrupted with ADHD,” Dr. Cutler said.
The findings were published online July 25 in Clinical Therapeutics.
Novel modulating agent
Viloxazine extended-release is a novel multimodal serotonergic and noradrenergic modulating agent with activity at serotonin receptors and the norepinephrine transporter.
About two-thirds were boys.
All participants had an ADHD-Rating Scale–5 (ADHD-RS-5) score of at least 28 and a Clinical Global Impression–Severity score of at least 4. None had taken ADHD medication for at least 1 week prior to randomization.
The intent-to-treat population included 460 children. Of these, 155 were randomly assigned to receive placebo, 147 to receive viloxazine 100 mg, and 158 to receive viloxazine 200 mg.
The primary efficacy endpoint was change from baseline in ADHD-RS-5 total score at week 6. Score changes for both the 100-mg (P = .0004) and the 200-mg (P < .0001) viloxazine groups met statistical significance compared with the placebo group.
Change from baseline in both the ADHD-RS-5 inattention and hyperactivity/impulsivity subscale scores was also significantly reduced in the 100-mg (P = .0006 and .0026, respectively) and 200-mg (P < .0001 and P < .0001, respectively) treatment groups compared with the placebo group.
Improvements occurred after 1 week of treatment and were maintained throughout the 6-week trial, “indicating an early and sustained effect,” the investigators wrote.
FDA target action date
The Clinical Global Impression–Improvement (CGI-I) score at 6 weeks was significantly improved in those receiving 100 mg (P = .0020) and 200 mg (P < .0001) of the active treatment compared with placebo.
The CGI-I responder rate, the percentage of children with a CGI-I score of 1 (very much improved) or 2 (much improved), was significantly higher at 6 weeks with viloxazine 100 mg and 200 mg vs. placebo (45% and 51% vs. 30%, respectively; P = .0065 and P = .0002).
These standard investigator-rated assessments were supported by two parent self-rated assessments: the Conners 3–Parent Short Form and the Weiss Functional Impairment Rating Scale–Parent Form.
Parents noted improvement not only in their children’s ADHD symptoms but also in ADHD-associated learning problems, executive functioning, defiance/aggression, peer relations, and functioning in different settings.
At both doses, once-daily viloxazine was generally well tolerated, with a low rate of discontinuation because of adverse events (<5%). Most adverse events were characterized as mild or moderate in severity and included somnolence (8.9%), decreased appetite (6.0%), and headache (5.4%).
On the basis of results of this study and others, the Food and Drug Administration accepted the company’s new drug application for viloxazine extended-release for ADHD in children and adolescents. The application has a target action date of Nov. 8, 2020.
Potential advantages
Commenting on the study in an interview, Dean Elbe, PharmD, clinical pharmacy specialist, child and adolescent mental health, BC Children’s Hospital, Vancouver, B.C., said that use of viloxazine to treat ADHD is “interesting.”
Dr. Elbe, who was not involved with the current research, noted that “it is actually an old drug that has been around since the mid-1970s in Europe as an antidepressant. It was removed from the market due to poor sales, not safety issues.”
Overall, on the basis of this study, viloxazine has potential to offer “modest improvements” over atomoxetine (Strattera), and the dosing may be “more straightforward and somewhat less challenging than with atomoxetine, with no taper up and no adjustment for poor 2D6 metabolizers,” Dr. Elbe noted.
“The onset of action appears somewhat quicker than we typically see with atomoxetine, so that is also helpful for parent and clinician acceptance and partially overcomes a perceived barrier with atomoxetine,” he said.
Dr. Elbe said he wonders, however, whether viloxazine will show “real-world clinical utility for both hyperactive-impulsive as well as inattentive symptoms. Although the study shows efficacy in both symptom clusters, so did the atomoxetine RCTs, and this has not been the clinical impression for atomoxetine.”
The study was funded by Supernus Pharmaceuticals. Dr. Cutler is a consultant for Supernus, as well as for Adlon Therapeutics, Aevi Genomics, Akili Interactive, Arbor Pharmaceuticals, Ironshore, KemPharm, Lundbeck, Neos Therapeutics, NLS Pharma, Otsuka, Purdue, Shire, Sunovion, Takeda, and Tris Pharma. He has received speaker/promotional honoraria from Adlon Therapeutics, Arbor Pharmaceuticals, Lundbeck, Neos Therapeutics, Otsuka, Shire, Sunovion, Takeda, and Tris Pharma and has received research grants from Aevi Genomics, Akili Interactive, Arbor Pharmaceuticals, Ironshore, KemPharm, Lundbeck, Neos Therapeutics, Otsuka, Purdue, Shire, Sunovion, Supernus Pharmaceuticals, Takeda, and Tris Pharma. A complete list of disclosures for the other authors is available in the original article. Dr. Elbe has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.












