User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
COVID-19 burdens follow patients after discharge
COVID-19 patients who survive their hospitalization don’t leave the disease behind upon discharge, as a significant percentage died within 60 days of discharge, with an ICU admission heightening the risk, according to an observational study of 38 Michigan hospitals. What’s more, many of them were burdened with health and emotional challenges ranging from hospital readmission to job loss and financial problems.
“These data confirm that the toll of COVID-19 extends well beyond hospitalization, a finding consistent with long-term sequelae from sepsis and other severe respiratory viral illnesses,” wrote lead author Vineet Chopra, MBBS, of the University of Michigan, Ann Arbor, and colleagues (Ann Intern Med. 2020 Nov 11: doi: 10.7326/M20-5661)
The researchers found that 29.2% of all patients hospitalized for COVID-19 from March 16 to July 1 died. The observational cohort study included 1,648 COVID-19 patients hospitalized at 38 Michigan hospitals participating in a statewide collaborative.
The bulk of those deaths occurred during hospitalization: 24.2% of patients (n = 398). Of the 1,250 patients discharged, 78% (n = 975) went home and 12.6% (n = 158) went to a skilled nursing facility, with the remainder unaccounted for. Within 60 days of discharge, 6.7% (n = 84) of hospitalized survivors had died and 15.2% (n = 189) were readmitted. The researchers gathered 60-day postdischarge data via a telephone survey, contacting 41.8% (n = 488) of discharged patients.
Outcomes were even worse for discharged patients who spent time in the ICU. The death rate among this group was 10.4% (17 of 165) after discharge. That resulted in an overall study death rate of 63.5% (n = 257) for the 405 patients who were in the ICU.
While the study data were in the first wave of the novel coronavirus, the findings have relevance today, said Mary Jo Farmer, MD, PhD, FCCP, directory of pulmonary hypertension services at Baystate Health in Springfield, Mass.
“This is the best information we have to date,” she said. “We have to continue to have an open mind and expect that this information may change as the virus possibly mutates as it spreads, and we should continue doing these types of outcomes studies at 90 days, 120 days, etc.”
The median age of study patients was 62, with a range of 50-72. The three leading comorbidities among discharged patients were hypertension (n = 800, 64%), diabetes (34.9%, n = 436), and cardiovascular disease (24.1%, n = 301).
Poor postdischarge outcomes weren’t limited to mortality and readmission. Almost 19% (n = 92) reported new or worsening cardiopulmonary symptoms such as cough and dyspnea, 13.3% had a persistent loss of taste or smell, and 12% (n = 58) reported more difficulty with daily living tasks.
The after-effects were not only physical. Nearly half of discharged patients (48.7%, n = 238) reported emotional effects and almost 6% (n = 28) sought mental health care. Among the 40% (n = 195) employed before they were hospitalized, 36% (n = 78) couldn’t return to work because of health issues or layoffs. Sixty percent (n = 117) of the pre-employed discharged patients did return to work, but 25% (n = 30) did so with reduced hours or modified job duties because of health problems.
Financial problems were also a burden. More than a third, 36.7% (n = 179), reported some financial impact from their hospitalization. About 10% (n = 47) said they used most or all of their savings, and 7% (n = 35) said they resorted to rationing necessities such as food or medications.
The researchers noted that one in five patients had no primary care follow-up at 2 months post discharge. “Collectively, these findings suggest that better models to support COVID-19 survivors are necessary,” said Dr. Chopra and colleagues.
The postdischarge course for patients involves two humps, said Sachin Gupta, MD, FCCP a pulmonary and critical care specialist at Alameda Health System in Oakland, Calif.: Getting over the hospitalization itself and the recovery phase. “As you look at the median age of the survivors, elderly patients who survive a hospital stay are still going to have a period of recovery, and like any viral illness that leads to someone being hospitalized, when you have an elderly patient with comorbidities, not all of them can make it over that final hump.”
He echoed the study authors’ call for better postdischarge support for COVID-19 patients. “There’s typically, although not at every hospital, a one-size-fits-all discharge planning process,” Dr. Gupta said. “For older patients, particularly with comorbid conditions, close follow-up after discharge is important.”
Dr. Farmer noted that one challenge in discharge support may be a matter of personnel. “The providers of this care might be fearful of patients who have had COVID-19 – Do the patients remain contagious? What if symptoms of COVID-19 return such as dry cough, fever? – and of contracting the disease themselves,” she said.
The findings regarding the emotional status of discharged patients should factor into discharge planning, she added. “Providers of posthospital care need to be educated in the emotional impact of this disease (e.g., the patients may feel ostracized or that no one wants to be around them) to assist in their recovery.”
Dr. Chopra and Dr. Farmer have no financial relationships to disclose. Dr. Gupta is an employee and shareholder of Genentech.
SOURCE: Chopra V et al. Ann Intern Med. 2020 Nov 11. doi: 10.7326/M20-5661.
COVID-19 patients who survive their hospitalization don’t leave the disease behind upon discharge, as a significant percentage died within 60 days of discharge, with an ICU admission heightening the risk, according to an observational study of 38 Michigan hospitals. What’s more, many of them were burdened with health and emotional challenges ranging from hospital readmission to job loss and financial problems.
“These data confirm that the toll of COVID-19 extends well beyond hospitalization, a finding consistent with long-term sequelae from sepsis and other severe respiratory viral illnesses,” wrote lead author Vineet Chopra, MBBS, of the University of Michigan, Ann Arbor, and colleagues (Ann Intern Med. 2020 Nov 11: doi: 10.7326/M20-5661)
The researchers found that 29.2% of all patients hospitalized for COVID-19 from March 16 to July 1 died. The observational cohort study included 1,648 COVID-19 patients hospitalized at 38 Michigan hospitals participating in a statewide collaborative.
The bulk of those deaths occurred during hospitalization: 24.2% of patients (n = 398). Of the 1,250 patients discharged, 78% (n = 975) went home and 12.6% (n = 158) went to a skilled nursing facility, with the remainder unaccounted for. Within 60 days of discharge, 6.7% (n = 84) of hospitalized survivors had died and 15.2% (n = 189) were readmitted. The researchers gathered 60-day postdischarge data via a telephone survey, contacting 41.8% (n = 488) of discharged patients.
Outcomes were even worse for discharged patients who spent time in the ICU. The death rate among this group was 10.4% (17 of 165) after discharge. That resulted in an overall study death rate of 63.5% (n = 257) for the 405 patients who were in the ICU.
While the study data were in the first wave of the novel coronavirus, the findings have relevance today, said Mary Jo Farmer, MD, PhD, FCCP, directory of pulmonary hypertension services at Baystate Health in Springfield, Mass.
“This is the best information we have to date,” she said. “We have to continue to have an open mind and expect that this information may change as the virus possibly mutates as it spreads, and we should continue doing these types of outcomes studies at 90 days, 120 days, etc.”
The median age of study patients was 62, with a range of 50-72. The three leading comorbidities among discharged patients were hypertension (n = 800, 64%), diabetes (34.9%, n = 436), and cardiovascular disease (24.1%, n = 301).
Poor postdischarge outcomes weren’t limited to mortality and readmission. Almost 19% (n = 92) reported new or worsening cardiopulmonary symptoms such as cough and dyspnea, 13.3% had a persistent loss of taste or smell, and 12% (n = 58) reported more difficulty with daily living tasks.
The after-effects were not only physical. Nearly half of discharged patients (48.7%, n = 238) reported emotional effects and almost 6% (n = 28) sought mental health care. Among the 40% (n = 195) employed before they were hospitalized, 36% (n = 78) couldn’t return to work because of health issues or layoffs. Sixty percent (n = 117) of the pre-employed discharged patients did return to work, but 25% (n = 30) did so with reduced hours or modified job duties because of health problems.
Financial problems were also a burden. More than a third, 36.7% (n = 179), reported some financial impact from their hospitalization. About 10% (n = 47) said they used most or all of their savings, and 7% (n = 35) said they resorted to rationing necessities such as food or medications.
The researchers noted that one in five patients had no primary care follow-up at 2 months post discharge. “Collectively, these findings suggest that better models to support COVID-19 survivors are necessary,” said Dr. Chopra and colleagues.
The postdischarge course for patients involves two humps, said Sachin Gupta, MD, FCCP a pulmonary and critical care specialist at Alameda Health System in Oakland, Calif.: Getting over the hospitalization itself and the recovery phase. “As you look at the median age of the survivors, elderly patients who survive a hospital stay are still going to have a period of recovery, and like any viral illness that leads to someone being hospitalized, when you have an elderly patient with comorbidities, not all of them can make it over that final hump.”
He echoed the study authors’ call for better postdischarge support for COVID-19 patients. “There’s typically, although not at every hospital, a one-size-fits-all discharge planning process,” Dr. Gupta said. “For older patients, particularly with comorbid conditions, close follow-up after discharge is important.”
Dr. Farmer noted that one challenge in discharge support may be a matter of personnel. “The providers of this care might be fearful of patients who have had COVID-19 – Do the patients remain contagious? What if symptoms of COVID-19 return such as dry cough, fever? – and of contracting the disease themselves,” she said.
The findings regarding the emotional status of discharged patients should factor into discharge planning, she added. “Providers of posthospital care need to be educated in the emotional impact of this disease (e.g., the patients may feel ostracized or that no one wants to be around them) to assist in their recovery.”
Dr. Chopra and Dr. Farmer have no financial relationships to disclose. Dr. Gupta is an employee and shareholder of Genentech.
SOURCE: Chopra V et al. Ann Intern Med. 2020 Nov 11. doi: 10.7326/M20-5661.
COVID-19 patients who survive their hospitalization don’t leave the disease behind upon discharge, as a significant percentage died within 60 days of discharge, with an ICU admission heightening the risk, according to an observational study of 38 Michigan hospitals. What’s more, many of them were burdened with health and emotional challenges ranging from hospital readmission to job loss and financial problems.
“These data confirm that the toll of COVID-19 extends well beyond hospitalization, a finding consistent with long-term sequelae from sepsis and other severe respiratory viral illnesses,” wrote lead author Vineet Chopra, MBBS, of the University of Michigan, Ann Arbor, and colleagues (Ann Intern Med. 2020 Nov 11: doi: 10.7326/M20-5661)
The researchers found that 29.2% of all patients hospitalized for COVID-19 from March 16 to July 1 died. The observational cohort study included 1,648 COVID-19 patients hospitalized at 38 Michigan hospitals participating in a statewide collaborative.
The bulk of those deaths occurred during hospitalization: 24.2% of patients (n = 398). Of the 1,250 patients discharged, 78% (n = 975) went home and 12.6% (n = 158) went to a skilled nursing facility, with the remainder unaccounted for. Within 60 days of discharge, 6.7% (n = 84) of hospitalized survivors had died and 15.2% (n = 189) were readmitted. The researchers gathered 60-day postdischarge data via a telephone survey, contacting 41.8% (n = 488) of discharged patients.
Outcomes were even worse for discharged patients who spent time in the ICU. The death rate among this group was 10.4% (17 of 165) after discharge. That resulted in an overall study death rate of 63.5% (n = 257) for the 405 patients who were in the ICU.
While the study data were in the first wave of the novel coronavirus, the findings have relevance today, said Mary Jo Farmer, MD, PhD, FCCP, directory of pulmonary hypertension services at Baystate Health in Springfield, Mass.
“This is the best information we have to date,” she said. “We have to continue to have an open mind and expect that this information may change as the virus possibly mutates as it spreads, and we should continue doing these types of outcomes studies at 90 days, 120 days, etc.”
The median age of study patients was 62, with a range of 50-72. The three leading comorbidities among discharged patients were hypertension (n = 800, 64%), diabetes (34.9%, n = 436), and cardiovascular disease (24.1%, n = 301).
Poor postdischarge outcomes weren’t limited to mortality and readmission. Almost 19% (n = 92) reported new or worsening cardiopulmonary symptoms such as cough and dyspnea, 13.3% had a persistent loss of taste or smell, and 12% (n = 58) reported more difficulty with daily living tasks.
The after-effects were not only physical. Nearly half of discharged patients (48.7%, n = 238) reported emotional effects and almost 6% (n = 28) sought mental health care. Among the 40% (n = 195) employed before they were hospitalized, 36% (n = 78) couldn’t return to work because of health issues or layoffs. Sixty percent (n = 117) of the pre-employed discharged patients did return to work, but 25% (n = 30) did so with reduced hours or modified job duties because of health problems.
Financial problems were also a burden. More than a third, 36.7% (n = 179), reported some financial impact from their hospitalization. About 10% (n = 47) said they used most or all of their savings, and 7% (n = 35) said they resorted to rationing necessities such as food or medications.
The researchers noted that one in five patients had no primary care follow-up at 2 months post discharge. “Collectively, these findings suggest that better models to support COVID-19 survivors are necessary,” said Dr. Chopra and colleagues.
The postdischarge course for patients involves two humps, said Sachin Gupta, MD, FCCP a pulmonary and critical care specialist at Alameda Health System in Oakland, Calif.: Getting over the hospitalization itself and the recovery phase. “As you look at the median age of the survivors, elderly patients who survive a hospital stay are still going to have a period of recovery, and like any viral illness that leads to someone being hospitalized, when you have an elderly patient with comorbidities, not all of them can make it over that final hump.”
He echoed the study authors’ call for better postdischarge support for COVID-19 patients. “There’s typically, although not at every hospital, a one-size-fits-all discharge planning process,” Dr. Gupta said. “For older patients, particularly with comorbid conditions, close follow-up after discharge is important.”
Dr. Farmer noted that one challenge in discharge support may be a matter of personnel. “The providers of this care might be fearful of patients who have had COVID-19 – Do the patients remain contagious? What if symptoms of COVID-19 return such as dry cough, fever? – and of contracting the disease themselves,” she said.
The findings regarding the emotional status of discharged patients should factor into discharge planning, she added. “Providers of posthospital care need to be educated in the emotional impact of this disease (e.g., the patients may feel ostracized or that no one wants to be around them) to assist in their recovery.”
Dr. Chopra and Dr. Farmer have no financial relationships to disclose. Dr. Gupta is an employee and shareholder of Genentech.
SOURCE: Chopra V et al. Ann Intern Med. 2020 Nov 11. doi: 10.7326/M20-5661.
FROM ANNALS OF INTERNAL MEDICINE
Pediatric News welcomes Dr. Lessin to the board
Dr. Lessin has been a practicing pediatric clinician for the past 39 years at The Children’s Medical Group. In 1997, he was a founding partner of one of the first private practice “supergroups” by merging two competing pediatric practices into one and expanding it to 25 clinicians, with eight offices in three counties in New York state’s Mid-Hudson Valley. The group provides pediatric care to more than 30,000 children and has a nearly 90-year history, across its various incarnations, providing such care.
Dr. Lessin received his medical degree from Stanford (Calif.) University and trained in pediatrics at Yale-New Haven (Conn.) Medical Center. He has been active in national policy and leadership in the American Academy of Pediatrics, having served on the executive committee of the Section of Administration and Practice Management, the national Committee on Practice and Ambulatory Medicine, and his current appointment to the national Private Payer Advocacy Advisory Committee. In those roles he has authored several national policy statements and clinical guidelines, including “Increasing Immunization Coverage,” “Immunizing Parents and Other Close Family Contacts in the Pediatric Office Setting,” “Instrument-Based Pediatric Vision Screening Policy Statement,” and most recently, “Clinical Practice Guideline for the Diagnosis, Evaluation, and Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents.” He is also the coeditor of the AAP’s ADHD toolkit for clinicians published in 2019. Dr. Lessin served as the director of clinical research for his group for 5 years and the medical director for the practice for 10 years.
He has served as a faculty member at numerous local and regional pediatric meetings. He has been a faculty member at the AAP’s annual national conference and exposition for the past decade, speaking on a variety of topics. Dr. Lessin also has been an invited speaker internationally at pediatric conferences in India and Egypt. He has participated in more than a dozen medical missions to developing countries in Latin America, the Caribbean, Africa, and Vietnam. His expertise includes practice management, the business of medicine, immunizations, ADHD, and liability topics. He has been a testifying expert witness for both defense and plaintiff in medical malpractice litigation for more than 30 years. He founded and served as president of a medical independent practice association that began with 12 physicians and grew to over 3,000 doctors. He is also a certified managed care executive. His most recent interest has been becoming a professional voice-over actor!
While performing all of the above, Dr. Lessin is a dedicated community pediatrician whose first love and primary goal has remained providing the highest quality medical care to children while helping his colleagues manage their businesses in order to be able to survive and continue to provide such care.
Dr. Lessin has been a practicing pediatric clinician for the past 39 years at The Children’s Medical Group. In 1997, he was a founding partner of one of the first private practice “supergroups” by merging two competing pediatric practices into one and expanding it to 25 clinicians, with eight offices in three counties in New York state’s Mid-Hudson Valley. The group provides pediatric care to more than 30,000 children and has a nearly 90-year history, across its various incarnations, providing such care.
Dr. Lessin received his medical degree from Stanford (Calif.) University and trained in pediatrics at Yale-New Haven (Conn.) Medical Center. He has been active in national policy and leadership in the American Academy of Pediatrics, having served on the executive committee of the Section of Administration and Practice Management, the national Committee on Practice and Ambulatory Medicine, and his current appointment to the national Private Payer Advocacy Advisory Committee. In those roles he has authored several national policy statements and clinical guidelines, including “Increasing Immunization Coverage,” “Immunizing Parents and Other Close Family Contacts in the Pediatric Office Setting,” “Instrument-Based Pediatric Vision Screening Policy Statement,” and most recently, “Clinical Practice Guideline for the Diagnosis, Evaluation, and Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents.” He is also the coeditor of the AAP’s ADHD toolkit for clinicians published in 2019. Dr. Lessin served as the director of clinical research for his group for 5 years and the medical director for the practice for 10 years.
He has served as a faculty member at numerous local and regional pediatric meetings. He has been a faculty member at the AAP’s annual national conference and exposition for the past decade, speaking on a variety of topics. Dr. Lessin also has been an invited speaker internationally at pediatric conferences in India and Egypt. He has participated in more than a dozen medical missions to developing countries in Latin America, the Caribbean, Africa, and Vietnam. His expertise includes practice management, the business of medicine, immunizations, ADHD, and liability topics. He has been a testifying expert witness for both defense and plaintiff in medical malpractice litigation for more than 30 years. He founded and served as president of a medical independent practice association that began with 12 physicians and grew to over 3,000 doctors. He is also a certified managed care executive. His most recent interest has been becoming a professional voice-over actor!
While performing all of the above, Dr. Lessin is a dedicated community pediatrician whose first love and primary goal has remained providing the highest quality medical care to children while helping his colleagues manage their businesses in order to be able to survive and continue to provide such care.
Dr. Lessin has been a practicing pediatric clinician for the past 39 years at The Children’s Medical Group. In 1997, he was a founding partner of one of the first private practice “supergroups” by merging two competing pediatric practices into one and expanding it to 25 clinicians, with eight offices in three counties in New York state’s Mid-Hudson Valley. The group provides pediatric care to more than 30,000 children and has a nearly 90-year history, across its various incarnations, providing such care.
Dr. Lessin received his medical degree from Stanford (Calif.) University and trained in pediatrics at Yale-New Haven (Conn.) Medical Center. He has been active in national policy and leadership in the American Academy of Pediatrics, having served on the executive committee of the Section of Administration and Practice Management, the national Committee on Practice and Ambulatory Medicine, and his current appointment to the national Private Payer Advocacy Advisory Committee. In those roles he has authored several national policy statements and clinical guidelines, including “Increasing Immunization Coverage,” “Immunizing Parents and Other Close Family Contacts in the Pediatric Office Setting,” “Instrument-Based Pediatric Vision Screening Policy Statement,” and most recently, “Clinical Practice Guideline for the Diagnosis, Evaluation, and Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents.” He is also the coeditor of the AAP’s ADHD toolkit for clinicians published in 2019. Dr. Lessin served as the director of clinical research for his group for 5 years and the medical director for the practice for 10 years.
He has served as a faculty member at numerous local and regional pediatric meetings. He has been a faculty member at the AAP’s annual national conference and exposition for the past decade, speaking on a variety of topics. Dr. Lessin also has been an invited speaker internationally at pediatric conferences in India and Egypt. He has participated in more than a dozen medical missions to developing countries in Latin America, the Caribbean, Africa, and Vietnam. His expertise includes practice management, the business of medicine, immunizations, ADHD, and liability topics. He has been a testifying expert witness for both defense and plaintiff in medical malpractice litigation for more than 30 years. He founded and served as president of a medical independent practice association that began with 12 physicians and grew to over 3,000 doctors. He is also a certified managed care executive. His most recent interest has been becoming a professional voice-over actor!
While performing all of the above, Dr. Lessin is a dedicated community pediatrician whose first love and primary goal has remained providing the highest quality medical care to children while helping his colleagues manage their businesses in order to be able to survive and continue to provide such care.
Metapneumovirus infections clinically indistinguishable from flu, RSV
The all-consuming news about SARS-CoV-2 and COVID-19 has overshadowed other viral pathogens that are the cause of severe or fatal lower respiratory infections (LRI) including human metapneumovirus (HMPV).
“MPV is really a leading cause of LRI not just in children but in adults, with high mortality rates in the frail elderly, long-term care facilities, and cancer patients with pneumonia, “ said John Williams, MD, from the department of pediatric infectious diseases at the University of Pittsburgh Medical Center.
“Right now we have no effective antivirals. There are monoclonal antibodies in development that my group and others have discovered. In fact, some of these treat MPV and RSV [respiratory syncytial virus], so we may have good options,” he said in an online presentation during an annual scientific meeting on infectious diseases.
The virus preys, wolf-like, on the most vulnerable patients, including children and frail elderly adults, as well as other adults with predisposing conditions, he said.
HMPV causes acute respiratory illnesses in approximately 2%-11% of hospitalized adults, 3%-25% of organ transplant recipients or cancer patients, 4%-12% of chronic obstructive pulmonary disease exacerbations, 5%-20% of asthma exacerbations, and it has been identified in multiple outbreaks at long-term care facilities.
Relative newcomer
Metapneumovirus was isolated and discovered from children with respiratory tract disease in the early 2000s. Once included in the family of paramyxoviruses (including measles, mumps, Nipah virus, and parainfluenza virus 1-4), HMPV and RSV are now classified as pneumoviruses, based on gene order and other characteristics, Dr. Williams explained.
Various studies have consistently placed the prevalence of HMPV ranging from 5%-14% in young children with LRI, children hospitalized for wheezing, adults with cancer and LRI, adults with asthma admissions, children with upper respiratory infections, and children hospitalized in the United States and Jordan for LRI, as well as children hospitalized in the United States and Peru with acute respiratory infections.
A study tracking respiratory infections in a Rochester, N.Y., cohort from 1999 through 2003 showed that healthy elderly patients had and annual incidence of HMPV infections of 5.9%, compared with 9.1% for high-risk patients, 13.1% for young patients, and 8.5% among hospitalized adult patients.
“These percentages are virtually identical to what has been seen in the same cohort for respiratory syncytial virus, so in this multiyear prospective cohort, metapneumovirus was as common as RSV,” Dr. Williams said.
Although the incidences of both HMPV and RSV were lower among hospitalized adults “clinically, we can’t tell these respiratory viruses apart. If we know it’s circulating we can make a guess, but we really can’t discriminate them,” he added.
In the Rochester cohort the frequency of clinical symptoms – including congestion, sore throat, cough, sputum production, dyspnea, and fever – were similar among patients infected with HMPV, RSV, or influenza A, with the exception of a slightly higher incidence of wheezing (80%) with HMPV, compared with influenza.
“I can tell you as a pediatrician, this is absolutely true in children, that metapneumovirus is indistinguishable from other respiratory viruses in kids,” he said.
Fatalities among older adults
As noted before, HMPV can cause severe and fatal illness in adults. For example, during an outbreak in North Dakota in 2016, 3 of 27 hospitalized adults with HMPV (median age, 69 years) died, and 10 required mechanical or noninvasive ventilation.
In a study from Korea comparing outcomes of severe HMPV-associated community-acquired pneumonia (CAP) with those of severe influenza-associated CAP, the investigators found that 30- and 60-day mortality rates were similar between the groups, at 24% of patients with HMPV-associated CAP and 32.1% for influenza-associated CAP, and 32% versus 38.5%, respectively.
Patients at high risk for severe disease or death from HMPV infection include those over 65 years, especially frail elderly, patients with chronic obstructive pulmonary disease, immunocompromised patients, and those with cardiopulmonary diseases such as congestive heart failure.
Supportive care only
“Do we have anything for treatment? The short answer is, No,” Dr. Williams.
Supportive care is currently the only effective approach for patients with severe HMPV infection.
Ribavirin, used to treat patients with acute RSV infection, has poor in vitro activity against HMPV and poor oral bioavailability and hemolysis, and there are no randomized controlled trials to support its use in this situation.
“It really can’t be recommended, and I don’t recommend it,” he said.
Virology may still help
Mark J. Siedner, MD, an infectious diseases physician at Mass General and associate professor of medicine at Harvard Medical School, both in Boston, who was not involved in the study, said that, despite the inability to clinically distinguish HMPV from RSV or influenza A, there is still clinical value to identifying HMPV infections.
“We spend millions of dollar each year treating people for upper respiratory tract infections, often with antibacterials, sometimes with antivirals, but those have costs to the health care system, and they also have costs in terms of drug resistance,” he said in an interview seeking objective commentary.
“Diagnostic tests that determine the actual source or the cause of these upper respiratory tract infections and encourage both patients and physicians not to be using antibiotics have value,” he said.
Identifying the pathogen can also help clinicians take appropriate infection-control precautions to prevent patient-to-clinician or patient-to-patient transmission of viral infections, he added.
Dr. Williams’ research is supported by the National Institutes of Health, Henry L. Hillman Foundation, and Asher Krop Memorial Fund of Children’s Hospital of Pittsburgh. Dr. Williams and Dr. Siedner reported no relevant conflict of interest disclosures.
The all-consuming news about SARS-CoV-2 and COVID-19 has overshadowed other viral pathogens that are the cause of severe or fatal lower respiratory infections (LRI) including human metapneumovirus (HMPV).
“MPV is really a leading cause of LRI not just in children but in adults, with high mortality rates in the frail elderly, long-term care facilities, and cancer patients with pneumonia, “ said John Williams, MD, from the department of pediatric infectious diseases at the University of Pittsburgh Medical Center.
“Right now we have no effective antivirals. There are monoclonal antibodies in development that my group and others have discovered. In fact, some of these treat MPV and RSV [respiratory syncytial virus], so we may have good options,” he said in an online presentation during an annual scientific meeting on infectious diseases.
The virus preys, wolf-like, on the most vulnerable patients, including children and frail elderly adults, as well as other adults with predisposing conditions, he said.
HMPV causes acute respiratory illnesses in approximately 2%-11% of hospitalized adults, 3%-25% of organ transplant recipients or cancer patients, 4%-12% of chronic obstructive pulmonary disease exacerbations, 5%-20% of asthma exacerbations, and it has been identified in multiple outbreaks at long-term care facilities.
Relative newcomer
Metapneumovirus was isolated and discovered from children with respiratory tract disease in the early 2000s. Once included in the family of paramyxoviruses (including measles, mumps, Nipah virus, and parainfluenza virus 1-4), HMPV and RSV are now classified as pneumoviruses, based on gene order and other characteristics, Dr. Williams explained.
Various studies have consistently placed the prevalence of HMPV ranging from 5%-14% in young children with LRI, children hospitalized for wheezing, adults with cancer and LRI, adults with asthma admissions, children with upper respiratory infections, and children hospitalized in the United States and Jordan for LRI, as well as children hospitalized in the United States and Peru with acute respiratory infections.
A study tracking respiratory infections in a Rochester, N.Y., cohort from 1999 through 2003 showed that healthy elderly patients had and annual incidence of HMPV infections of 5.9%, compared with 9.1% for high-risk patients, 13.1% for young patients, and 8.5% among hospitalized adult patients.
“These percentages are virtually identical to what has been seen in the same cohort for respiratory syncytial virus, so in this multiyear prospective cohort, metapneumovirus was as common as RSV,” Dr. Williams said.
Although the incidences of both HMPV and RSV were lower among hospitalized adults “clinically, we can’t tell these respiratory viruses apart. If we know it’s circulating we can make a guess, but we really can’t discriminate them,” he added.
In the Rochester cohort the frequency of clinical symptoms – including congestion, sore throat, cough, sputum production, dyspnea, and fever – were similar among patients infected with HMPV, RSV, or influenza A, with the exception of a slightly higher incidence of wheezing (80%) with HMPV, compared with influenza.
“I can tell you as a pediatrician, this is absolutely true in children, that metapneumovirus is indistinguishable from other respiratory viruses in kids,” he said.
Fatalities among older adults
As noted before, HMPV can cause severe and fatal illness in adults. For example, during an outbreak in North Dakota in 2016, 3 of 27 hospitalized adults with HMPV (median age, 69 years) died, and 10 required mechanical or noninvasive ventilation.
In a study from Korea comparing outcomes of severe HMPV-associated community-acquired pneumonia (CAP) with those of severe influenza-associated CAP, the investigators found that 30- and 60-day mortality rates were similar between the groups, at 24% of patients with HMPV-associated CAP and 32.1% for influenza-associated CAP, and 32% versus 38.5%, respectively.
Patients at high risk for severe disease or death from HMPV infection include those over 65 years, especially frail elderly, patients with chronic obstructive pulmonary disease, immunocompromised patients, and those with cardiopulmonary diseases such as congestive heart failure.
Supportive care only
“Do we have anything for treatment? The short answer is, No,” Dr. Williams.
Supportive care is currently the only effective approach for patients with severe HMPV infection.
Ribavirin, used to treat patients with acute RSV infection, has poor in vitro activity against HMPV and poor oral bioavailability and hemolysis, and there are no randomized controlled trials to support its use in this situation.
“It really can’t be recommended, and I don’t recommend it,” he said.
Virology may still help
Mark J. Siedner, MD, an infectious diseases physician at Mass General and associate professor of medicine at Harvard Medical School, both in Boston, who was not involved in the study, said that, despite the inability to clinically distinguish HMPV from RSV or influenza A, there is still clinical value to identifying HMPV infections.
“We spend millions of dollar each year treating people for upper respiratory tract infections, often with antibacterials, sometimes with antivirals, but those have costs to the health care system, and they also have costs in terms of drug resistance,” he said in an interview seeking objective commentary.
“Diagnostic tests that determine the actual source or the cause of these upper respiratory tract infections and encourage both patients and physicians not to be using antibiotics have value,” he said.
Identifying the pathogen can also help clinicians take appropriate infection-control precautions to prevent patient-to-clinician or patient-to-patient transmission of viral infections, he added.
Dr. Williams’ research is supported by the National Institutes of Health, Henry L. Hillman Foundation, and Asher Krop Memorial Fund of Children’s Hospital of Pittsburgh. Dr. Williams and Dr. Siedner reported no relevant conflict of interest disclosures.
The all-consuming news about SARS-CoV-2 and COVID-19 has overshadowed other viral pathogens that are the cause of severe or fatal lower respiratory infections (LRI) including human metapneumovirus (HMPV).
“MPV is really a leading cause of LRI not just in children but in adults, with high mortality rates in the frail elderly, long-term care facilities, and cancer patients with pneumonia, “ said John Williams, MD, from the department of pediatric infectious diseases at the University of Pittsburgh Medical Center.
“Right now we have no effective antivirals. There are monoclonal antibodies in development that my group and others have discovered. In fact, some of these treat MPV and RSV [respiratory syncytial virus], so we may have good options,” he said in an online presentation during an annual scientific meeting on infectious diseases.
The virus preys, wolf-like, on the most vulnerable patients, including children and frail elderly adults, as well as other adults with predisposing conditions, he said.
HMPV causes acute respiratory illnesses in approximately 2%-11% of hospitalized adults, 3%-25% of organ transplant recipients or cancer patients, 4%-12% of chronic obstructive pulmonary disease exacerbations, 5%-20% of asthma exacerbations, and it has been identified in multiple outbreaks at long-term care facilities.
Relative newcomer
Metapneumovirus was isolated and discovered from children with respiratory tract disease in the early 2000s. Once included in the family of paramyxoviruses (including measles, mumps, Nipah virus, and parainfluenza virus 1-4), HMPV and RSV are now classified as pneumoviruses, based on gene order and other characteristics, Dr. Williams explained.
Various studies have consistently placed the prevalence of HMPV ranging from 5%-14% in young children with LRI, children hospitalized for wheezing, adults with cancer and LRI, adults with asthma admissions, children with upper respiratory infections, and children hospitalized in the United States and Jordan for LRI, as well as children hospitalized in the United States and Peru with acute respiratory infections.
A study tracking respiratory infections in a Rochester, N.Y., cohort from 1999 through 2003 showed that healthy elderly patients had and annual incidence of HMPV infections of 5.9%, compared with 9.1% for high-risk patients, 13.1% for young patients, and 8.5% among hospitalized adult patients.
“These percentages are virtually identical to what has been seen in the same cohort for respiratory syncytial virus, so in this multiyear prospective cohort, metapneumovirus was as common as RSV,” Dr. Williams said.
Although the incidences of both HMPV and RSV were lower among hospitalized adults “clinically, we can’t tell these respiratory viruses apart. If we know it’s circulating we can make a guess, but we really can’t discriminate them,” he added.
In the Rochester cohort the frequency of clinical symptoms – including congestion, sore throat, cough, sputum production, dyspnea, and fever – were similar among patients infected with HMPV, RSV, or influenza A, with the exception of a slightly higher incidence of wheezing (80%) with HMPV, compared with influenza.
“I can tell you as a pediatrician, this is absolutely true in children, that metapneumovirus is indistinguishable from other respiratory viruses in kids,” he said.
Fatalities among older adults
As noted before, HMPV can cause severe and fatal illness in adults. For example, during an outbreak in North Dakota in 2016, 3 of 27 hospitalized adults with HMPV (median age, 69 years) died, and 10 required mechanical or noninvasive ventilation.
In a study from Korea comparing outcomes of severe HMPV-associated community-acquired pneumonia (CAP) with those of severe influenza-associated CAP, the investigators found that 30- and 60-day mortality rates were similar between the groups, at 24% of patients with HMPV-associated CAP and 32.1% for influenza-associated CAP, and 32% versus 38.5%, respectively.
Patients at high risk for severe disease or death from HMPV infection include those over 65 years, especially frail elderly, patients with chronic obstructive pulmonary disease, immunocompromised patients, and those with cardiopulmonary diseases such as congestive heart failure.
Supportive care only
“Do we have anything for treatment? The short answer is, No,” Dr. Williams.
Supportive care is currently the only effective approach for patients with severe HMPV infection.
Ribavirin, used to treat patients with acute RSV infection, has poor in vitro activity against HMPV and poor oral bioavailability and hemolysis, and there are no randomized controlled trials to support its use in this situation.
“It really can’t be recommended, and I don’t recommend it,” he said.
Virology may still help
Mark J. Siedner, MD, an infectious diseases physician at Mass General and associate professor of medicine at Harvard Medical School, both in Boston, who was not involved in the study, said that, despite the inability to clinically distinguish HMPV from RSV or influenza A, there is still clinical value to identifying HMPV infections.
“We spend millions of dollar each year treating people for upper respiratory tract infections, often with antibacterials, sometimes with antivirals, but those have costs to the health care system, and they also have costs in terms of drug resistance,” he said in an interview seeking objective commentary.
“Diagnostic tests that determine the actual source or the cause of these upper respiratory tract infections and encourage both patients and physicians not to be using antibiotics have value,” he said.
Identifying the pathogen can also help clinicians take appropriate infection-control precautions to prevent patient-to-clinician or patient-to-patient transmission of viral infections, he added.
Dr. Williams’ research is supported by the National Institutes of Health, Henry L. Hillman Foundation, and Asher Krop Memorial Fund of Children’s Hospital of Pittsburgh. Dr. Williams and Dr. Siedner reported no relevant conflict of interest disclosures.
FROM IDWEEK 2020
'Tragic' milestone: 1 million children with COVID-19
The number of new cases soared in the past week as the United States exceeded 1 million children infected with the coronavirus, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the first time, the number of cases in children for the week ending Nov. 12 passed 100,000, and it didn’t stop until it reached 111,946, bringing the total for the pandemic to 1,039,464 reported cases in 49 states (New York is not reporting ages), the District of Columbia, New York City, and Guam, the AAP and the CHA said in their weekly COVID-19 update.
“As a pediatrician who has practiced medicine for over 3 decades, I find this number staggering and tragic. We haven’t seen a virus flash through our communities in this way since before we had vaccines for measles and polio,” AAP President Sally Goza, MD, said in a written statement.
The previous 1-week high of almost 74,000 cases came just last week, and that number had surpassed the previous week’s new high of 61,000. The number of cumulative child cases, meanwhile, has doubled since Sept. 3, when it was just over 513,000. Children now represent 11.5% of all COVID-19 cases since the start of the pandemic in the jurisdictions reporting age distribution, the AAP and CHA said.
For the week ending Nov. 12, COVID-19 cases children made up 14% of cases nationally, rising from 13% the week before and reversing a decline that started in mid-October, the AAP/CHA data show.
The two groups continue to note the rarity of severe illness in children, but the number of deaths nationally had its biggest 1-week increase since late July, as the total rose from 123 to 133 in the 42 states reporting such data by age, as well as New York City. The cumulative hospitalization rate for children decreased slightly in the past week and is now down to 1.6% in the 23 states (and NYC) with available data, the AAP and CHA said.
The AAP called on elected leaders to enact a national strategy to combat the spread of the virus and urged health authorities to do more to collect data on longer-term impacts on children.
We’re very concerned about how this will impact all children, including toddlers who are missing key educational opportunities, as well as adolescents who may be at higher risk for anxiety and depression,” Dr. Goza said.
The number of new cases soared in the past week as the United States exceeded 1 million children infected with the coronavirus, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the first time, the number of cases in children for the week ending Nov. 12 passed 100,000, and it didn’t stop until it reached 111,946, bringing the total for the pandemic to 1,039,464 reported cases in 49 states (New York is not reporting ages), the District of Columbia, New York City, and Guam, the AAP and the CHA said in their weekly COVID-19 update.
“As a pediatrician who has practiced medicine for over 3 decades, I find this number staggering and tragic. We haven’t seen a virus flash through our communities in this way since before we had vaccines for measles and polio,” AAP President Sally Goza, MD, said in a written statement.
The previous 1-week high of almost 74,000 cases came just last week, and that number had surpassed the previous week’s new high of 61,000. The number of cumulative child cases, meanwhile, has doubled since Sept. 3, when it was just over 513,000. Children now represent 11.5% of all COVID-19 cases since the start of the pandemic in the jurisdictions reporting age distribution, the AAP and CHA said.
For the week ending Nov. 12, COVID-19 cases children made up 14% of cases nationally, rising from 13% the week before and reversing a decline that started in mid-October, the AAP/CHA data show.
The two groups continue to note the rarity of severe illness in children, but the number of deaths nationally had its biggest 1-week increase since late July, as the total rose from 123 to 133 in the 42 states reporting such data by age, as well as New York City. The cumulative hospitalization rate for children decreased slightly in the past week and is now down to 1.6% in the 23 states (and NYC) with available data, the AAP and CHA said.
The AAP called on elected leaders to enact a national strategy to combat the spread of the virus and urged health authorities to do more to collect data on longer-term impacts on children.
We’re very concerned about how this will impact all children, including toddlers who are missing key educational opportunities, as well as adolescents who may be at higher risk for anxiety and depression,” Dr. Goza said.
The number of new cases soared in the past week as the United States exceeded 1 million children infected with the coronavirus, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the first time, the number of cases in children for the week ending Nov. 12 passed 100,000, and it didn’t stop until it reached 111,946, bringing the total for the pandemic to 1,039,464 reported cases in 49 states (New York is not reporting ages), the District of Columbia, New York City, and Guam, the AAP and the CHA said in their weekly COVID-19 update.
“As a pediatrician who has practiced medicine for over 3 decades, I find this number staggering and tragic. We haven’t seen a virus flash through our communities in this way since before we had vaccines for measles and polio,” AAP President Sally Goza, MD, said in a written statement.
The previous 1-week high of almost 74,000 cases came just last week, and that number had surpassed the previous week’s new high of 61,000. The number of cumulative child cases, meanwhile, has doubled since Sept. 3, when it was just over 513,000. Children now represent 11.5% of all COVID-19 cases since the start of the pandemic in the jurisdictions reporting age distribution, the AAP and CHA said.
For the week ending Nov. 12, COVID-19 cases children made up 14% of cases nationally, rising from 13% the week before and reversing a decline that started in mid-October, the AAP/CHA data show.
The two groups continue to note the rarity of severe illness in children, but the number of deaths nationally had its biggest 1-week increase since late July, as the total rose from 123 to 133 in the 42 states reporting such data by age, as well as New York City. The cumulative hospitalization rate for children decreased slightly in the past week and is now down to 1.6% in the 23 states (and NYC) with available data, the AAP and CHA said.
The AAP called on elected leaders to enact a national strategy to combat the spread of the virus and urged health authorities to do more to collect data on longer-term impacts on children.
We’re very concerned about how this will impact all children, including toddlers who are missing key educational opportunities, as well as adolescents who may be at higher risk for anxiety and depression,” Dr. Goza said.
Open enrollment 2021: A big start for HealthCare.gov
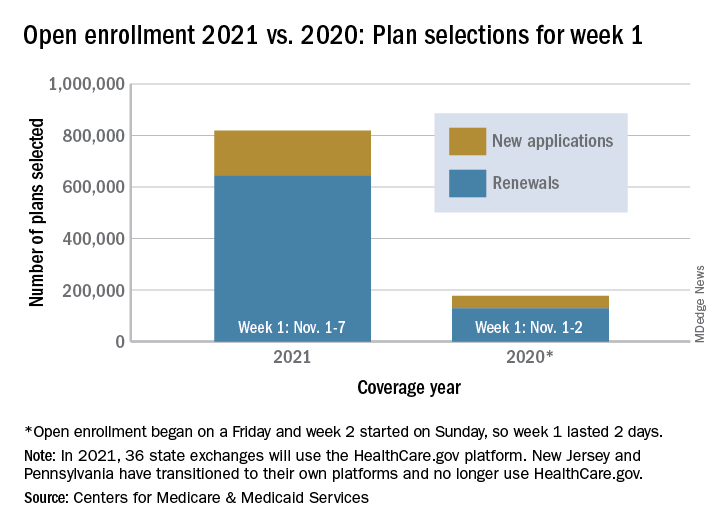
Over 818,000 plans were selected for the 2021 coverage year during the first week, Nov.1-7, of this year’s open enrollment on the federal health insurance exchange, according to the Centers for Medicare & Medicaid Services.
The bulk of those plans, nearly 79%, were renewals by consumers who had coverage through the federal exchange this year. The balance covers new plans selected by individuals who were not covered through HealthCare.gov this year, the CMS noted in a written statement.
The total enrollment for week 1 marks a considerable increase over last year’s first week of open enrollment, which saw approximately 177,000 plans selected, but Nov. 1 fell on a Friday in 2019, so that total represents only 2 days since weeks are tracked as running from Sunday to Saturday, the CMS explained.
For the 2021 benefit year, the HealthCare.gov platform covers 36 states, down from 38 for the 2020 benefit year, because New Jersey and Pennsylvania have “transitioned to their own state-based exchange platforms,” the CMS noted, adding that the two accounted for 7% of all plans selected last year.
“The final number of plan selections associated with enrollment activity during a reporting period may change due to plan modifications or cancellations,” CMS said, and its weekly snapshot “does not report the number of consumers who have paid premiums to effectuate their enrollment.”
This year’s open-enrollment period on HealthCare.gov is scheduled to conclude Dec. 15.
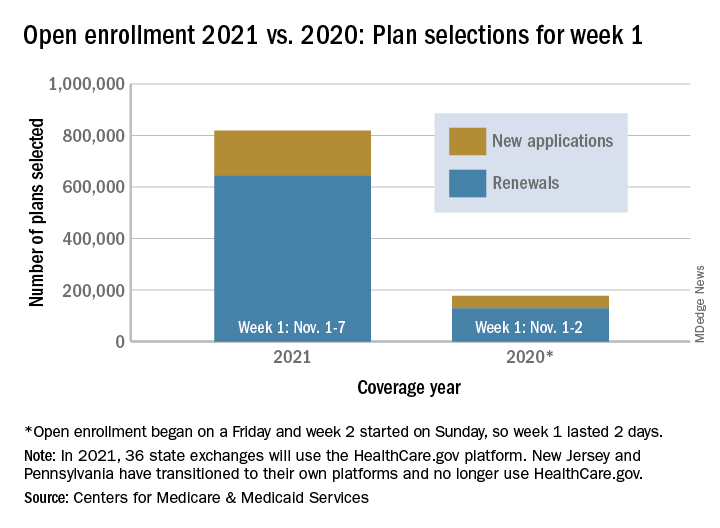
Over 818,000 plans were selected for the 2021 coverage year during the first week, Nov.1-7, of this year’s open enrollment on the federal health insurance exchange, according to the Centers for Medicare & Medicaid Services.

The bulk of those plans, nearly 79%, were renewals by consumers who had coverage through the federal exchange this year. The balance covers new plans selected by individuals who were not covered through HealthCare.gov this year, the CMS noted in a written statement.
The total enrollment for week 1 marks a considerable increase over last year’s first week of open enrollment, which saw approximately 177,000 plans selected, but Nov. 1 fell on a Friday in 2019, so that total represents only 2 days since weeks are tracked as running from Sunday to Saturday, the CMS explained.
For the 2021 benefit year, the HealthCare.gov platform covers 36 states, down from 38 for the 2020 benefit year, because New Jersey and Pennsylvania have “transitioned to their own state-based exchange platforms,” the CMS noted, adding that the two accounted for 7% of all plans selected last year.
“The final number of plan selections associated with enrollment activity during a reporting period may change due to plan modifications or cancellations,” CMS said, and its weekly snapshot “does not report the number of consumers who have paid premiums to effectuate their enrollment.”
This year’s open-enrollment period on HealthCare.gov is scheduled to conclude Dec. 15.
Over 818,000 plans were selected for the 2021 coverage year during the first week, Nov.1-7, of this year’s open enrollment on the federal health insurance exchange, according to the Centers for Medicare & Medicaid Services.

The bulk of those plans, nearly 79%, were renewals by consumers who had coverage through the federal exchange this year. The balance covers new plans selected by individuals who were not covered through HealthCare.gov this year, the CMS noted in a written statement.
The total enrollment for week 1 marks a considerable increase over last year’s first week of open enrollment, which saw approximately 177,000 plans selected, but Nov. 1 fell on a Friday in 2019, so that total represents only 2 days since weeks are tracked as running from Sunday to Saturday, the CMS explained.
For the 2021 benefit year, the HealthCare.gov platform covers 36 states, down from 38 for the 2020 benefit year, because New Jersey and Pennsylvania have “transitioned to their own state-based exchange platforms,” the CMS noted, adding that the two accounted for 7% of all plans selected last year.
“The final number of plan selections associated with enrollment activity during a reporting period may change due to plan modifications or cancellations,” CMS said, and its weekly snapshot “does not report the number of consumers who have paid premiums to effectuate their enrollment.”
This year’s open-enrollment period on HealthCare.gov is scheduled to conclude Dec. 15.
FDA-approved peanut immunotherapy protocol comes with a cost
Peanut allergy immunotherapy now comes with approval from the US Food and Drug Administration (FDA), but it also comes with protocols, standards, and paperwork. Whether it will be widely adopted has yet to be determined.
A few dozen allergists around the world have been offering food allergy immunotherapy for many years, having developed their own measuring techniques using store-bought food.
But the vast majority of allergists are not interested in developing home-grown treatments, not only because it involves research and development, but also because it comes with legal risks.
“Finally we have another treatment option,” said Edwin Kim, MD, from the UNC Allergy and Immunology Clinic in Chapel Hill, N.C. “This is what we were waiting for. It’s not cowboy stuff; this works.”
In January, the FDA approved peanut allergen powder (Palforzia) for patients 4-17 years of age, as reported by Medscape Medical News.
The pill contains measured doses of peanut flour and comes with a protocol that will allow allergists to bring patients to a peanut tolerance of 300 mg (about one peanut) and a black-box warning about anaphylaxis risk.
And before allergists can prescribe it, they must take a Risk Evaluation and Mitigation Strategy course to learn about dosing and the allergic reaction protocol.
“That may scare some away,” said Dr. Kim, who discussed the FDA-approved option during his presentation at the American College of Allergy, Asthma & Immunology 2020 Annual Scientific Meeting.
Allergic reaction, including the potential for anaphylaxis, has always been an issue with immunotherapy.
“People make the argument that there is a difference” between an expected allergic reaction – such as one that occurs after the administration of immunotherapy – and an unexpected reaction, he said. Because an expected reaction can be treated quickly, “some feel these expected reactions don’t matter so much.”
“Others say a reaction is a reaction” and argue that if, a treatment causes reaction, then it doesn’t make sense, he explained.
It comes down to patients – they must be willing to take a risk to develop tolerance and improve their quality of life – and the allergists willing to treat them.
The peanut powder involves paperwork, preauthorization forms, denials of care, a higher price tag, regimented procedures, and a prerequisite number of visits with patients. “Not everyone will want to do this,” said Dr. Kim.
The regimen involves three phases. During initial dose escalation, five doses are administered in the office on day 1. Then, over the next 6 months, updoses are administered during 11 in-office sessions and a 300-mg tolerance is achieved. Finally, to maintain tolerance to one peanut, daily doses are administered at home.
The drug cost alone is about $4,200 a year, according to Institute for Clinical and Economic Review. Peanut flour from the grocery store is cheaper, but comes with the risk of bacteria or other contamination.
“This product offers some reassurance, and that matters,” Dr. Kim said.
It’s good to have more options for food allergy treatment. “We need a more proactive way to treat food allergy; avoidance is not good enough,” he explained. “And presumably, at some point, the patient will be able to eat a grocery-store peanut instead of buying the pills.”
The art of medicine
But not all allergists will be able to make the protocol work. And it’s not clear whether there is room to alter treatment and offer patients with a higher tolerance a higher starting dose. What we do know, though, is that “the product leaves little room for ‘the art of medicine,’ ” Kim said.
That art is practiced by Arnon Elizur, MD, from the Shamir Medical Center in Tzrifin, Israel, but it’s backed by a rigid home-grown protocol.
Since 2010, he has treated 1,800 patients for peanut allergy, updosing slowly to a tolerance of 3,000 mg of peanut, the equivalent of 10 peanuts. He keeps the maintenance dose at four peanuts (1,200 mg). His center takes a personalized approach, starting patients on the highest dose they can tolerate and working up, with daily patient check-ins from home and a staff available around the clock to answer questions and deal with reactions.
“We aim for full sensitization,” Dr. Elizur said in an interview.
The peanut pill is “a big step forward” for immunotherapy, he said. It is “a standardized product, checked for bacteria and allergen content, which is available to a wide community of physicians.”
But, he pointed out, “it’s expensive.” And it’s only for peanut. “There are millions of food-allergic patients around the world dying from adverse reactions to many different kinds of food. We don’t want to wait for years for a product for all of them. We can use the actual food.”
He questions the lifelong maintenance protocol with a daily 300-mg pill. “If you can’t eat a peanut, why would you buy a drug that’s a peanut?” he asked.
He also said he’s disappointed that the product is not indicated for adults.
At the Shamir clinic, reactions are closely monitored. “Some are mild, others we treat with autoinjectors, epinephrine,” he reported. “Those are the most undesirable.”
Data from his center show that reactions occur in about 15% of patients. But his treatment success rates are good. In an average of 8 months, he is able to get 80% of his adult patients to full sensitization.
But it’s not for all patients or for all clinics, he acknowledged. “We continue to look at this balance in quality of life throughout the process. Our goal is to improve the quality of life threshold.”
Treatment that involves “native food” is “a lot of work” and requires “a lot of investment,” Dr. Elizur said. His center uses a web reporting system to maintain a 24/7 dialogue with patients, “and we look at the reports every day.” They also have a physician on call at all times. “Not everyone can commit to providing care throughout the day and night.”
His center charges the equivalent of $US3,000 per food allergy treated. “That’s whether it takes 6 months or 2 years,” he said.
There are more than 1,000 people on his 3-year waiting list.
“This is the first year that the American College of Allergy, Asthma, and Immunology is not hosting a pro–con debate on oral immunotherapy,” Dr. Kim pointed out. “We have a therapy now.”
However, the pandemic has slowed treatment uptake. “Immunotherapy is not easy to do, whether it’s FDA approved or not,” he explained. With at least 11 doctor visits in the first 6 months – each visit is between 30 minutes and 2-3 hours long – it hasn’t been possible to set up this year. “It’s not ideal.”
It will be interesting to see “how this will roll out and how it will be adopted,” Dr. Kim said. “From a food allergy point of view, the next 12 months are going to be very interesting.”
Dr. Kim reports receiving consulting honorarium from Aimmune, the maker of Palforzia; being on the clinical medical advisory board for DBV Technologies; and consulting for Aimmune, Allakos, Allergenis, DBV, Duke Clinical Research Institute, Ukko Incorporated, Vibrant America, and Kenota Health. Dr. Elizur has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Peanut allergy immunotherapy now comes with approval from the US Food and Drug Administration (FDA), but it also comes with protocols, standards, and paperwork. Whether it will be widely adopted has yet to be determined.
A few dozen allergists around the world have been offering food allergy immunotherapy for many years, having developed their own measuring techniques using store-bought food.
But the vast majority of allergists are not interested in developing home-grown treatments, not only because it involves research and development, but also because it comes with legal risks.
“Finally we have another treatment option,” said Edwin Kim, MD, from the UNC Allergy and Immunology Clinic in Chapel Hill, N.C. “This is what we were waiting for. It’s not cowboy stuff; this works.”
In January, the FDA approved peanut allergen powder (Palforzia) for patients 4-17 years of age, as reported by Medscape Medical News.
The pill contains measured doses of peanut flour and comes with a protocol that will allow allergists to bring patients to a peanut tolerance of 300 mg (about one peanut) and a black-box warning about anaphylaxis risk.
And before allergists can prescribe it, they must take a Risk Evaluation and Mitigation Strategy course to learn about dosing and the allergic reaction protocol.
“That may scare some away,” said Dr. Kim, who discussed the FDA-approved option during his presentation at the American College of Allergy, Asthma & Immunology 2020 Annual Scientific Meeting.
Allergic reaction, including the potential for anaphylaxis, has always been an issue with immunotherapy.
“People make the argument that there is a difference” between an expected allergic reaction – such as one that occurs after the administration of immunotherapy – and an unexpected reaction, he said. Because an expected reaction can be treated quickly, “some feel these expected reactions don’t matter so much.”
“Others say a reaction is a reaction” and argue that if, a treatment causes reaction, then it doesn’t make sense, he explained.
It comes down to patients – they must be willing to take a risk to develop tolerance and improve their quality of life – and the allergists willing to treat them.
The peanut powder involves paperwork, preauthorization forms, denials of care, a higher price tag, regimented procedures, and a prerequisite number of visits with patients. “Not everyone will want to do this,” said Dr. Kim.
The regimen involves three phases. During initial dose escalation, five doses are administered in the office on day 1. Then, over the next 6 months, updoses are administered during 11 in-office sessions and a 300-mg tolerance is achieved. Finally, to maintain tolerance to one peanut, daily doses are administered at home.
The drug cost alone is about $4,200 a year, according to Institute for Clinical and Economic Review. Peanut flour from the grocery store is cheaper, but comes with the risk of bacteria or other contamination.
“This product offers some reassurance, and that matters,” Dr. Kim said.
It’s good to have more options for food allergy treatment. “We need a more proactive way to treat food allergy; avoidance is not good enough,” he explained. “And presumably, at some point, the patient will be able to eat a grocery-store peanut instead of buying the pills.”
The art of medicine
But not all allergists will be able to make the protocol work. And it’s not clear whether there is room to alter treatment and offer patients with a higher tolerance a higher starting dose. What we do know, though, is that “the product leaves little room for ‘the art of medicine,’ ” Kim said.
That art is practiced by Arnon Elizur, MD, from the Shamir Medical Center in Tzrifin, Israel, but it’s backed by a rigid home-grown protocol.
Since 2010, he has treated 1,800 patients for peanut allergy, updosing slowly to a tolerance of 3,000 mg of peanut, the equivalent of 10 peanuts. He keeps the maintenance dose at four peanuts (1,200 mg). His center takes a personalized approach, starting patients on the highest dose they can tolerate and working up, with daily patient check-ins from home and a staff available around the clock to answer questions and deal with reactions.
“We aim for full sensitization,” Dr. Elizur said in an interview.
The peanut pill is “a big step forward” for immunotherapy, he said. It is “a standardized product, checked for bacteria and allergen content, which is available to a wide community of physicians.”
But, he pointed out, “it’s expensive.” And it’s only for peanut. “There are millions of food-allergic patients around the world dying from adverse reactions to many different kinds of food. We don’t want to wait for years for a product for all of them. We can use the actual food.”
He questions the lifelong maintenance protocol with a daily 300-mg pill. “If you can’t eat a peanut, why would you buy a drug that’s a peanut?” he asked.
He also said he’s disappointed that the product is not indicated for adults.
At the Shamir clinic, reactions are closely monitored. “Some are mild, others we treat with autoinjectors, epinephrine,” he reported. “Those are the most undesirable.”
Data from his center show that reactions occur in about 15% of patients. But his treatment success rates are good. In an average of 8 months, he is able to get 80% of his adult patients to full sensitization.
But it’s not for all patients or for all clinics, he acknowledged. “We continue to look at this balance in quality of life throughout the process. Our goal is to improve the quality of life threshold.”
Treatment that involves “native food” is “a lot of work” and requires “a lot of investment,” Dr. Elizur said. His center uses a web reporting system to maintain a 24/7 dialogue with patients, “and we look at the reports every day.” They also have a physician on call at all times. “Not everyone can commit to providing care throughout the day and night.”
His center charges the equivalent of $US3,000 per food allergy treated. “That’s whether it takes 6 months or 2 years,” he said.
There are more than 1,000 people on his 3-year waiting list.
“This is the first year that the American College of Allergy, Asthma, and Immunology is not hosting a pro–con debate on oral immunotherapy,” Dr. Kim pointed out. “We have a therapy now.”
However, the pandemic has slowed treatment uptake. “Immunotherapy is not easy to do, whether it’s FDA approved or not,” he explained. With at least 11 doctor visits in the first 6 months – each visit is between 30 minutes and 2-3 hours long – it hasn’t been possible to set up this year. “It’s not ideal.”
It will be interesting to see “how this will roll out and how it will be adopted,” Dr. Kim said. “From a food allergy point of view, the next 12 months are going to be very interesting.”
Dr. Kim reports receiving consulting honorarium from Aimmune, the maker of Palforzia; being on the clinical medical advisory board for DBV Technologies; and consulting for Aimmune, Allakos, Allergenis, DBV, Duke Clinical Research Institute, Ukko Incorporated, Vibrant America, and Kenota Health. Dr. Elizur has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Peanut allergy immunotherapy now comes with approval from the US Food and Drug Administration (FDA), but it also comes with protocols, standards, and paperwork. Whether it will be widely adopted has yet to be determined.
A few dozen allergists around the world have been offering food allergy immunotherapy for many years, having developed their own measuring techniques using store-bought food.
But the vast majority of allergists are not interested in developing home-grown treatments, not only because it involves research and development, but also because it comes with legal risks.
“Finally we have another treatment option,” said Edwin Kim, MD, from the UNC Allergy and Immunology Clinic in Chapel Hill, N.C. “This is what we were waiting for. It’s not cowboy stuff; this works.”
In January, the FDA approved peanut allergen powder (Palforzia) for patients 4-17 years of age, as reported by Medscape Medical News.
The pill contains measured doses of peanut flour and comes with a protocol that will allow allergists to bring patients to a peanut tolerance of 300 mg (about one peanut) and a black-box warning about anaphylaxis risk.
And before allergists can prescribe it, they must take a Risk Evaluation and Mitigation Strategy course to learn about dosing and the allergic reaction protocol.
“That may scare some away,” said Dr. Kim, who discussed the FDA-approved option during his presentation at the American College of Allergy, Asthma & Immunology 2020 Annual Scientific Meeting.
Allergic reaction, including the potential for anaphylaxis, has always been an issue with immunotherapy.
“People make the argument that there is a difference” between an expected allergic reaction – such as one that occurs after the administration of immunotherapy – and an unexpected reaction, he said. Because an expected reaction can be treated quickly, “some feel these expected reactions don’t matter so much.”
“Others say a reaction is a reaction” and argue that if, a treatment causes reaction, then it doesn’t make sense, he explained.
It comes down to patients – they must be willing to take a risk to develop tolerance and improve their quality of life – and the allergists willing to treat them.
The peanut powder involves paperwork, preauthorization forms, denials of care, a higher price tag, regimented procedures, and a prerequisite number of visits with patients. “Not everyone will want to do this,” said Dr. Kim.
The regimen involves three phases. During initial dose escalation, five doses are administered in the office on day 1. Then, over the next 6 months, updoses are administered during 11 in-office sessions and a 300-mg tolerance is achieved. Finally, to maintain tolerance to one peanut, daily doses are administered at home.
The drug cost alone is about $4,200 a year, according to Institute for Clinical and Economic Review. Peanut flour from the grocery store is cheaper, but comes with the risk of bacteria or other contamination.
“This product offers some reassurance, and that matters,” Dr. Kim said.
It’s good to have more options for food allergy treatment. “We need a more proactive way to treat food allergy; avoidance is not good enough,” he explained. “And presumably, at some point, the patient will be able to eat a grocery-store peanut instead of buying the pills.”
The art of medicine
But not all allergists will be able to make the protocol work. And it’s not clear whether there is room to alter treatment and offer patients with a higher tolerance a higher starting dose. What we do know, though, is that “the product leaves little room for ‘the art of medicine,’ ” Kim said.
That art is practiced by Arnon Elizur, MD, from the Shamir Medical Center in Tzrifin, Israel, but it’s backed by a rigid home-grown protocol.
Since 2010, he has treated 1,800 patients for peanut allergy, updosing slowly to a tolerance of 3,000 mg of peanut, the equivalent of 10 peanuts. He keeps the maintenance dose at four peanuts (1,200 mg). His center takes a personalized approach, starting patients on the highest dose they can tolerate and working up, with daily patient check-ins from home and a staff available around the clock to answer questions and deal with reactions.
“We aim for full sensitization,” Dr. Elizur said in an interview.
The peanut pill is “a big step forward” for immunotherapy, he said. It is “a standardized product, checked for bacteria and allergen content, which is available to a wide community of physicians.”
But, he pointed out, “it’s expensive.” And it’s only for peanut. “There are millions of food-allergic patients around the world dying from adverse reactions to many different kinds of food. We don’t want to wait for years for a product for all of them. We can use the actual food.”
He questions the lifelong maintenance protocol with a daily 300-mg pill. “If you can’t eat a peanut, why would you buy a drug that’s a peanut?” he asked.
He also said he’s disappointed that the product is not indicated for adults.
At the Shamir clinic, reactions are closely monitored. “Some are mild, others we treat with autoinjectors, epinephrine,” he reported. “Those are the most undesirable.”
Data from his center show that reactions occur in about 15% of patients. But his treatment success rates are good. In an average of 8 months, he is able to get 80% of his adult patients to full sensitization.
But it’s not for all patients or for all clinics, he acknowledged. “We continue to look at this balance in quality of life throughout the process. Our goal is to improve the quality of life threshold.”
Treatment that involves “native food” is “a lot of work” and requires “a lot of investment,” Dr. Elizur said. His center uses a web reporting system to maintain a 24/7 dialogue with patients, “and we look at the reports every day.” They also have a physician on call at all times. “Not everyone can commit to providing care throughout the day and night.”
His center charges the equivalent of $US3,000 per food allergy treated. “That’s whether it takes 6 months or 2 years,” he said.
There are more than 1,000 people on his 3-year waiting list.
“This is the first year that the American College of Allergy, Asthma, and Immunology is not hosting a pro–con debate on oral immunotherapy,” Dr. Kim pointed out. “We have a therapy now.”
However, the pandemic has slowed treatment uptake. “Immunotherapy is not easy to do, whether it’s FDA approved or not,” he explained. With at least 11 doctor visits in the first 6 months – each visit is between 30 minutes and 2-3 hours long – it hasn’t been possible to set up this year. “It’s not ideal.”
It will be interesting to see “how this will roll out and how it will be adopted,” Dr. Kim said. “From a food allergy point of view, the next 12 months are going to be very interesting.”
Dr. Kim reports receiving consulting honorarium from Aimmune, the maker of Palforzia; being on the clinical medical advisory board for DBV Technologies; and consulting for Aimmune, Allakos, Allergenis, DBV, Duke Clinical Research Institute, Ukko Incorporated, Vibrant America, and Kenota Health. Dr. Elizur has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Moderna: Interim data show 94.5% efficacy for COVID-19 vaccine, will seek FDA EUA
The Moderna mRNA-1273 vaccine, in development to prevent COVID-19, yielded 94.5% efficacy in early results and is generally well tolerated, the company announced early Monday. The product can be stored at refrigeration temperatures common to many physician offices, pharmacies, and hospitals.
The first interim results of the phase 3 COVE trial included 95 participants with confirmed COVID-19. An independent data safety monitoring board, which was appointed by the National Institutes of Health, informed Moderna that 90 of the patients who were positive for COVID-19 were in a placebo group and that 5 patients were in the mRNA-1273 vaccine group, resulting in a vaccine efficacy of 94.5% (P < .0001).
Interim data included 11 patients with severe COVID-19, all of whom were in the placebo group.
“This positive interim analysis from our phase 3 study has given us the first clinical validation that our vaccine can prevent COVID-19 disease, including severe disease,” said Stéphane Bancel, CEO of Moderna, said in a statement.
The vaccine met its primary study endpoint, which was based on adjudicated data that were collected starting 2 weeks after the second dose of mRNA-1273. The interim study population included people who could be at higher risk for COVID-19, including 15 adults aged 65 years and older and 20 participants from diverse communities.
Safety data
The DSMB also reviewed safety data for the COVE study interim results. The vaccine was generally safe and well tolerated, as determined on the basis of solicited adverse events. Most adverse events were mild to moderate and were generally short-lived, according to a company news release.
Injection-site pain was reported in 2.7% of participants after the first dose. After the second dose, 9.7% of participants reported fatigue, 8.9% reported myalgia, 5.2% reported arthralgia, 4.5% reported headache, 4.1% reported pain, and 2.0% reported erythema or redness at the injection site.
Moderna plans to request emergency-use authorization (EUA) from the Food and Drug Administration in the coming weeks. The company expects that the EUA will be based on more data from the COVE study, including a final analysis of 151 patients with a median follow-up of more than 2 months. Moderna also plans to seek authorizations from global regulatory agencies.
The company expects to have approximately 20 million doses of mRNA-1273 ready to ship in the United States by the end of the year. In addition, the company says it remains on track to manufacture between 500 million and 1 billion doses globally in 2021.
Moderna is developing distribution plans in conjunction with the Centers for Disease Control and Prevention, the federal government’s Operation Warp Speed, and McKesson, a COVID-19 vaccine distributor contracted by the U.S. government.
Refrigeration requirements
The mRNA-1273 vaccine can be shipped and stored for up to 6 months at –20° C (about –4° F), a temperature maintained in most home or medical freezers, according to Moderna. The company expects that, after the product thaws, it will remain stable at standard refrigerator temperatures of 2°-8° C (36°-46° F) for up to 30 days within the 6-month shelf life.
Because the mRNA-1273 vaccine is stable at these refrigerator temperatures, it can be stored at most physicians’ offices, pharmacies, and hospitals, the company noted. In contrast, the similar Pfizer BTN162b2 vaccine – early results for which showed a 90% efficacy rate – requires shipment and storage at “deep-freeze” conditions of –70° C or –80° C, which is more challenging from a logistic point of view.
Moderna’s mRNA-1273 can be kept at room temperature for up to 12 hours after removal from a refrigerator for patient administration. The vaccine will not require dilution prior to use.
More than 30,000 people aged older than 18 years in the United States are enrolled in the COVE study. The research is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases and the Biomedical Advanced Research and Development Authority, part of the Office of the Assistant Secretary for Preparedness and Response at the Department of Health & Human Services.
A version of this article originally appeared on Medscape.com.
The Moderna mRNA-1273 vaccine, in development to prevent COVID-19, yielded 94.5% efficacy in early results and is generally well tolerated, the company announced early Monday. The product can be stored at refrigeration temperatures common to many physician offices, pharmacies, and hospitals.
The first interim results of the phase 3 COVE trial included 95 participants with confirmed COVID-19. An independent data safety monitoring board, which was appointed by the National Institutes of Health, informed Moderna that 90 of the patients who were positive for COVID-19 were in a placebo group and that 5 patients were in the mRNA-1273 vaccine group, resulting in a vaccine efficacy of 94.5% (P < .0001).
Interim data included 11 patients with severe COVID-19, all of whom were in the placebo group.
“This positive interim analysis from our phase 3 study has given us the first clinical validation that our vaccine can prevent COVID-19 disease, including severe disease,” said Stéphane Bancel, CEO of Moderna, said in a statement.
The vaccine met its primary study endpoint, which was based on adjudicated data that were collected starting 2 weeks after the second dose of mRNA-1273. The interim study population included people who could be at higher risk for COVID-19, including 15 adults aged 65 years and older and 20 participants from diverse communities.
Safety data
The DSMB also reviewed safety data for the COVE study interim results. The vaccine was generally safe and well tolerated, as determined on the basis of solicited adverse events. Most adverse events were mild to moderate and were generally short-lived, according to a company news release.
Injection-site pain was reported in 2.7% of participants after the first dose. After the second dose, 9.7% of participants reported fatigue, 8.9% reported myalgia, 5.2% reported arthralgia, 4.5% reported headache, 4.1% reported pain, and 2.0% reported erythema or redness at the injection site.
Moderna plans to request emergency-use authorization (EUA) from the Food and Drug Administration in the coming weeks. The company expects that the EUA will be based on more data from the COVE study, including a final analysis of 151 patients with a median follow-up of more than 2 months. Moderna also plans to seek authorizations from global regulatory agencies.
The company expects to have approximately 20 million doses of mRNA-1273 ready to ship in the United States by the end of the year. In addition, the company says it remains on track to manufacture between 500 million and 1 billion doses globally in 2021.
Moderna is developing distribution plans in conjunction with the Centers for Disease Control and Prevention, the federal government’s Operation Warp Speed, and McKesson, a COVID-19 vaccine distributor contracted by the U.S. government.
Refrigeration requirements
The mRNA-1273 vaccine can be shipped and stored for up to 6 months at –20° C (about –4° F), a temperature maintained in most home or medical freezers, according to Moderna. The company expects that, after the product thaws, it will remain stable at standard refrigerator temperatures of 2°-8° C (36°-46° F) for up to 30 days within the 6-month shelf life.
Because the mRNA-1273 vaccine is stable at these refrigerator temperatures, it can be stored at most physicians’ offices, pharmacies, and hospitals, the company noted. In contrast, the similar Pfizer BTN162b2 vaccine – early results for which showed a 90% efficacy rate – requires shipment and storage at “deep-freeze” conditions of –70° C or –80° C, which is more challenging from a logistic point of view.
Moderna’s mRNA-1273 can be kept at room temperature for up to 12 hours after removal from a refrigerator for patient administration. The vaccine will not require dilution prior to use.
More than 30,000 people aged older than 18 years in the United States are enrolled in the COVE study. The research is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases and the Biomedical Advanced Research and Development Authority, part of the Office of the Assistant Secretary for Preparedness and Response at the Department of Health & Human Services.
A version of this article originally appeared on Medscape.com.
The Moderna mRNA-1273 vaccine, in development to prevent COVID-19, yielded 94.5% efficacy in early results and is generally well tolerated, the company announced early Monday. The product can be stored at refrigeration temperatures common to many physician offices, pharmacies, and hospitals.
The first interim results of the phase 3 COVE trial included 95 participants with confirmed COVID-19. An independent data safety monitoring board, which was appointed by the National Institutes of Health, informed Moderna that 90 of the patients who were positive for COVID-19 were in a placebo group and that 5 patients were in the mRNA-1273 vaccine group, resulting in a vaccine efficacy of 94.5% (P < .0001).
Interim data included 11 patients with severe COVID-19, all of whom were in the placebo group.
“This positive interim analysis from our phase 3 study has given us the first clinical validation that our vaccine can prevent COVID-19 disease, including severe disease,” said Stéphane Bancel, CEO of Moderna, said in a statement.
The vaccine met its primary study endpoint, which was based on adjudicated data that were collected starting 2 weeks after the second dose of mRNA-1273. The interim study population included people who could be at higher risk for COVID-19, including 15 adults aged 65 years and older and 20 participants from diverse communities.
Safety data
The DSMB also reviewed safety data for the COVE study interim results. The vaccine was generally safe and well tolerated, as determined on the basis of solicited adverse events. Most adverse events were mild to moderate and were generally short-lived, according to a company news release.
Injection-site pain was reported in 2.7% of participants after the first dose. After the second dose, 9.7% of participants reported fatigue, 8.9% reported myalgia, 5.2% reported arthralgia, 4.5% reported headache, 4.1% reported pain, and 2.0% reported erythema or redness at the injection site.
Moderna plans to request emergency-use authorization (EUA) from the Food and Drug Administration in the coming weeks. The company expects that the EUA will be based on more data from the COVE study, including a final analysis of 151 patients with a median follow-up of more than 2 months. Moderna also plans to seek authorizations from global regulatory agencies.
The company expects to have approximately 20 million doses of mRNA-1273 ready to ship in the United States by the end of the year. In addition, the company says it remains on track to manufacture between 500 million and 1 billion doses globally in 2021.
Moderna is developing distribution plans in conjunction with the Centers for Disease Control and Prevention, the federal government’s Operation Warp Speed, and McKesson, a COVID-19 vaccine distributor contracted by the U.S. government.
Refrigeration requirements
The mRNA-1273 vaccine can be shipped and stored for up to 6 months at –20° C (about –4° F), a temperature maintained in most home or medical freezers, according to Moderna. The company expects that, after the product thaws, it will remain stable at standard refrigerator temperatures of 2°-8° C (36°-46° F) for up to 30 days within the 6-month shelf life.
Because the mRNA-1273 vaccine is stable at these refrigerator temperatures, it can be stored at most physicians’ offices, pharmacies, and hospitals, the company noted. In contrast, the similar Pfizer BTN162b2 vaccine – early results for which showed a 90% efficacy rate – requires shipment and storage at “deep-freeze” conditions of –70° C or –80° C, which is more challenging from a logistic point of view.
Moderna’s mRNA-1273 can be kept at room temperature for up to 12 hours after removal from a refrigerator for patient administration. The vaccine will not require dilution prior to use.
More than 30,000 people aged older than 18 years in the United States are enrolled in the COVE study. The research is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases and the Biomedical Advanced Research and Development Authority, part of the Office of the Assistant Secretary for Preparedness and Response at the Department of Health & Human Services.
A version of this article originally appeared on Medscape.com.
Poor image quality may limit televulvology care
Seeing patients with vulvar problems via telemedicine can lead to efficient and successful care, but there are challenges and limitations with this approach, doctors are finding.
Image quality is one key factor that determines whether a clinician can assess and manage a condition remotely, said Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif. Other issues may be especially relevant to televulvology, including privacy concerns.
“Who is helping with the positioning? Who is the photographer? Is the patient comfortable with having photos taken of this part of their body and submitted, even if they know it is submitted securely? Because they might not be,” Dr. Venkatesan said in a lecture at a virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
When quality photographs from referring providers are available, Dr. Venkatesan has conducted virtual new consultations. “But sometimes I will do a virtual telemedicine visit as the first visit and then figure out, okay, this isn’t really sufficient. I need to see them in person.”
Melissa Mauskar, MD, assistant professor of dermatology and obstetrics and gynecology at the University of Texas Southwestern Medical Center, Dallas, described a case early on during the COVID-19 pandemic that illustrates a limitation of virtual visits.
A patient sent in a photograph that appeared to show lichen sclerosus. “There looked like some classic lichen sclerosus changes,” Dr. Mauskar said during a discussion at the meeting. “But she was having a lot of pain, and after a week, her pain still was not better.”
Dr. Mauskar brought the patient into the office and ultimately diagnosed a squamous cell carcinoma. “What I thought was a normal erosion was actually an ulcerated plaque,” she said.
Like Dr. Venkatesan, Dr. Mauskar has found that image quality can be uneven. Photographs may be out of focus. Video visits have been a mixed bag. Some are successful. Other times, Dr. Mauskar has to tell the patient she needs to see her in the office.
Certain clinical scenarios require a vaginal exam, Dr. Venkatesan noted. Although some type of assessment may be possible if a patient is with a primary care provider during the telemedicine visit, the examination may not be equivalent. Doctors also should anticipate where a patient might go to have a biopsy if one is necessary.
Another telemedicine caveat pertains to patient counseling. When using store-and-forward telemedicine systems, advising patients in a written report can be challenging. “Is there an easy way ... to counsel patients how to apply their topical medications?” Dr. Venkatesan said.
Excellent care is possible
Vulvology is a small part of Dr. Venkatesan’s general dermatology practice, which has used telemedicine extensively since the pandemic.
In recent years, Dr. Venkatesan’s clinic began encouraging providers in their health system to submit photographs with referrals. “That has really paid off now because we have been able to help provide a lot of excellent quality care for patients without them having to come in,” she said. “We may be able to say: ‘These are excellent photos. We know what this patient has. We can manage it. They don’t need to come see us in person.’ ” That could be the case for certain types of acne, eczema, and psoriasis.
In other cases, they may be able to provide initial advice remotely but still want to see the patient. For a patient with severe acne, “I may be able to tell the referring doctor: ‘Please start the patient on these three medicines. It will take 2 months for those medicines to start working and then we will plan to have an in-person dermatology visit.’ ” In this case, telemedicine essentially replaces one in-person visit.
If photographs are poor, the differential diagnosis is broad, a procedure is required, the doctor needs to touch the lesion, or more involved history taking or counseling are required, the patient may need to go into the office.
Beyond its public health advantages during a pandemic, telemedicine can improve access for patients who live far away, lack transportation, or are unable to take time off from work. It also can decrease patient wait times. “Once we started doing some telemedicine work … we went from having a 5-month wait time for patients to see us in person to a 72-hour wait time for providing some care for patients if they had good photos as part of their referral,” Dr. Venkatesan said.
Telemedicine has been used in inpatient and outpatient dermatology settings. Primary care providers who consult with dermatologists using a store-and-forward telemedicine system may improve their dermatology knowledge and feel more confident in their ability to diagnose and manage dermatologic conditions, research indicates.
In obstetrics and gynecology, telemedicine may play a role in preconception, contraception, and medical abortion care, prenatal visits, well-woman exams, mental health, and pre- and postoperative counseling, a recent review suggests.
Image quality is key
“Quality of the image is so critical for being able to provide good care, especially in such a visual exam field as dermatology,” Dr. Venkatesan said.
To that end, doctors have offered recommendations on how to photograph skin conditions. A guide shared by the mobile telehealth system company ClickMedix suggests focusing on the area of importance, capturing the extent of involvement, and including involved and uninvolved areas.
Good lighting and checking the image resolution can help, Dr. Venkatesan offered. Nevertheless, patients may have difficulty photographing themselves. If a patient is with their primary care doctor, “we are much more likely to be able to get good quality photos,” she said.
Dr. Venkatesan is a paid consultant for DirectDerm, a store-and-forward teledermatology company. Dr. Mauskar had no relevant disclosures.
Seeing patients with vulvar problems via telemedicine can lead to efficient and successful care, but there are challenges and limitations with this approach, doctors are finding.
Image quality is one key factor that determines whether a clinician can assess and manage a condition remotely, said Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif. Other issues may be especially relevant to televulvology, including privacy concerns.
“Who is helping with the positioning? Who is the photographer? Is the patient comfortable with having photos taken of this part of their body and submitted, even if they know it is submitted securely? Because they might not be,” Dr. Venkatesan said in a lecture at a virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
When quality photographs from referring providers are available, Dr. Venkatesan has conducted virtual new consultations. “But sometimes I will do a virtual telemedicine visit as the first visit and then figure out, okay, this isn’t really sufficient. I need to see them in person.”
Melissa Mauskar, MD, assistant professor of dermatology and obstetrics and gynecology at the University of Texas Southwestern Medical Center, Dallas, described a case early on during the COVID-19 pandemic that illustrates a limitation of virtual visits.
A patient sent in a photograph that appeared to show lichen sclerosus. “There looked like some classic lichen sclerosus changes,” Dr. Mauskar said during a discussion at the meeting. “But she was having a lot of pain, and after a week, her pain still was not better.”
Dr. Mauskar brought the patient into the office and ultimately diagnosed a squamous cell carcinoma. “What I thought was a normal erosion was actually an ulcerated plaque,” she said.
Like Dr. Venkatesan, Dr. Mauskar has found that image quality can be uneven. Photographs may be out of focus. Video visits have been a mixed bag. Some are successful. Other times, Dr. Mauskar has to tell the patient she needs to see her in the office.
Certain clinical scenarios require a vaginal exam, Dr. Venkatesan noted. Although some type of assessment may be possible if a patient is with a primary care provider during the telemedicine visit, the examination may not be equivalent. Doctors also should anticipate where a patient might go to have a biopsy if one is necessary.
Another telemedicine caveat pertains to patient counseling. When using store-and-forward telemedicine systems, advising patients in a written report can be challenging. “Is there an easy way ... to counsel patients how to apply their topical medications?” Dr. Venkatesan said.
Excellent care is possible
Vulvology is a small part of Dr. Venkatesan’s general dermatology practice, which has used telemedicine extensively since the pandemic.
In recent years, Dr. Venkatesan’s clinic began encouraging providers in their health system to submit photographs with referrals. “That has really paid off now because we have been able to help provide a lot of excellent quality care for patients without them having to come in,” she said. “We may be able to say: ‘These are excellent photos. We know what this patient has. We can manage it. They don’t need to come see us in person.’ ” That could be the case for certain types of acne, eczema, and psoriasis.
In other cases, they may be able to provide initial advice remotely but still want to see the patient. For a patient with severe acne, “I may be able to tell the referring doctor: ‘Please start the patient on these three medicines. It will take 2 months for those medicines to start working and then we will plan to have an in-person dermatology visit.’ ” In this case, telemedicine essentially replaces one in-person visit.
If photographs are poor, the differential diagnosis is broad, a procedure is required, the doctor needs to touch the lesion, or more involved history taking or counseling are required, the patient may need to go into the office.
Beyond its public health advantages during a pandemic, telemedicine can improve access for patients who live far away, lack transportation, or are unable to take time off from work. It also can decrease patient wait times. “Once we started doing some telemedicine work … we went from having a 5-month wait time for patients to see us in person to a 72-hour wait time for providing some care for patients if they had good photos as part of their referral,” Dr. Venkatesan said.
Telemedicine has been used in inpatient and outpatient dermatology settings. Primary care providers who consult with dermatologists using a store-and-forward telemedicine system may improve their dermatology knowledge and feel more confident in their ability to diagnose and manage dermatologic conditions, research indicates.
In obstetrics and gynecology, telemedicine may play a role in preconception, contraception, and medical abortion care, prenatal visits, well-woman exams, mental health, and pre- and postoperative counseling, a recent review suggests.
Image quality is key
“Quality of the image is so critical for being able to provide good care, especially in such a visual exam field as dermatology,” Dr. Venkatesan said.
To that end, doctors have offered recommendations on how to photograph skin conditions. A guide shared by the mobile telehealth system company ClickMedix suggests focusing on the area of importance, capturing the extent of involvement, and including involved and uninvolved areas.
Good lighting and checking the image resolution can help, Dr. Venkatesan offered. Nevertheless, patients may have difficulty photographing themselves. If a patient is with their primary care doctor, “we are much more likely to be able to get good quality photos,” she said.
Dr. Venkatesan is a paid consultant for DirectDerm, a store-and-forward teledermatology company. Dr. Mauskar had no relevant disclosures.
Seeing patients with vulvar problems via telemedicine can lead to efficient and successful care, but there are challenges and limitations with this approach, doctors are finding.
Image quality is one key factor that determines whether a clinician can assess and manage a condition remotely, said Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif. Other issues may be especially relevant to televulvology, including privacy concerns.
“Who is helping with the positioning? Who is the photographer? Is the patient comfortable with having photos taken of this part of their body and submitted, even if they know it is submitted securely? Because they might not be,” Dr. Venkatesan said in a lecture at a virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
When quality photographs from referring providers are available, Dr. Venkatesan has conducted virtual new consultations. “But sometimes I will do a virtual telemedicine visit as the first visit and then figure out, okay, this isn’t really sufficient. I need to see them in person.”
Melissa Mauskar, MD, assistant professor of dermatology and obstetrics and gynecology at the University of Texas Southwestern Medical Center, Dallas, described a case early on during the COVID-19 pandemic that illustrates a limitation of virtual visits.
A patient sent in a photograph that appeared to show lichen sclerosus. “There looked like some classic lichen sclerosus changes,” Dr. Mauskar said during a discussion at the meeting. “But she was having a lot of pain, and after a week, her pain still was not better.”
Dr. Mauskar brought the patient into the office and ultimately diagnosed a squamous cell carcinoma. “What I thought was a normal erosion was actually an ulcerated plaque,” she said.
Like Dr. Venkatesan, Dr. Mauskar has found that image quality can be uneven. Photographs may be out of focus. Video visits have been a mixed bag. Some are successful. Other times, Dr. Mauskar has to tell the patient she needs to see her in the office.
Certain clinical scenarios require a vaginal exam, Dr. Venkatesan noted. Although some type of assessment may be possible if a patient is with a primary care provider during the telemedicine visit, the examination may not be equivalent. Doctors also should anticipate where a patient might go to have a biopsy if one is necessary.
Another telemedicine caveat pertains to patient counseling. When using store-and-forward telemedicine systems, advising patients in a written report can be challenging. “Is there an easy way ... to counsel patients how to apply their topical medications?” Dr. Venkatesan said.
Excellent care is possible
Vulvology is a small part of Dr. Venkatesan’s general dermatology practice, which has used telemedicine extensively since the pandemic.
In recent years, Dr. Venkatesan’s clinic began encouraging providers in their health system to submit photographs with referrals. “That has really paid off now because we have been able to help provide a lot of excellent quality care for patients without them having to come in,” she said. “We may be able to say: ‘These are excellent photos. We know what this patient has. We can manage it. They don’t need to come see us in person.’ ” That could be the case for certain types of acne, eczema, and psoriasis.
In other cases, they may be able to provide initial advice remotely but still want to see the patient. For a patient with severe acne, “I may be able to tell the referring doctor: ‘Please start the patient on these three medicines. It will take 2 months for those medicines to start working and then we will plan to have an in-person dermatology visit.’ ” In this case, telemedicine essentially replaces one in-person visit.
If photographs are poor, the differential diagnosis is broad, a procedure is required, the doctor needs to touch the lesion, or more involved history taking or counseling are required, the patient may need to go into the office.
Beyond its public health advantages during a pandemic, telemedicine can improve access for patients who live far away, lack transportation, or are unable to take time off from work. It also can decrease patient wait times. “Once we started doing some telemedicine work … we went from having a 5-month wait time for patients to see us in person to a 72-hour wait time for providing some care for patients if they had good photos as part of their referral,” Dr. Venkatesan said.
Telemedicine has been used in inpatient and outpatient dermatology settings. Primary care providers who consult with dermatologists using a store-and-forward telemedicine system may improve their dermatology knowledge and feel more confident in their ability to diagnose and manage dermatologic conditions, research indicates.
In obstetrics and gynecology, telemedicine may play a role in preconception, contraception, and medical abortion care, prenatal visits, well-woman exams, mental health, and pre- and postoperative counseling, a recent review suggests.
Image quality is key
“Quality of the image is so critical for being able to provide good care, especially in such a visual exam field as dermatology,” Dr. Venkatesan said.
To that end, doctors have offered recommendations on how to photograph skin conditions. A guide shared by the mobile telehealth system company ClickMedix suggests focusing on the area of importance, capturing the extent of involvement, and including involved and uninvolved areas.
Good lighting and checking the image resolution can help, Dr. Venkatesan offered. Nevertheless, patients may have difficulty photographing themselves. If a patient is with their primary care doctor, “we are much more likely to be able to get good quality photos,” she said.
Dr. Venkatesan is a paid consultant for DirectDerm, a store-and-forward teledermatology company. Dr. Mauskar had no relevant disclosures.
FROM THE ISSVD BIENNIAL CONFERENCE
Lancet panel calls for urgent global action to combat diabetes
The article was published online Nov. 12, just ahead of World Diabetes Day.
Of the 463 million people with diabetes worldwide in 2019, 80% live in low- and middle-income countries. The condition reduces life expectancy in middle-aged adults by 4-10 years, including increasing the risk of death from cardiovascular disease, kidney disease, and cancer by up to threefold. It is also a leading cause of nontraumatic amputation and blindness.
Use of evidence-based interventions, if implemented and managed properly, could prevent thousands of deaths globally every day, stressed the commission.
“There is an enormous amount of knowledge that we have amassed over the years. We need good preventive care and we need to ensure that diabetes patients, once diagnosed, have good continuous care. There is an urgent need for decision-makers, policymakers, and payers to make things happen,” the leader of the multidisciplinary commission, Juliana C.N. Chan, MBChB, MD, said in an interview.
And now diabetes has emerged as a major risk factor for death from COVID-19, particularly in the setting of inadequate glycemic control.
“COVID-19 has exposed the vulnerability of individuals with diabetes,” said Dr. Chan, of the Hong Kong Institute of Diabetes and Obesity. “We should use the pandemic as an opportunity to implement solutions.”
Physician education key, trickling down to field workers and patients
First on the agenda, she says, should be “physician education. There are many primary care providers and internal medicine physicians whose knowledge needs to be updated.”
“Then doctors need to transfer this information to other people, such as nurses and community field workers. We cannot just rely on doctors; we need to train nonmedics” so that knowledge about how to prevent, treat, and manage diabetes long term is communicated right down the health care chain, she explained.
“They need to know how to look at people’s eyes and feet, how to do blood and urine tests, and how to collect data. Then they need to educate patients on what they should be doing, on how to practice self-care,” she added.
“We need to change our way of thinking, redesign clinic flow and how you build a team. And those care teams need to know how to collect data, and then use that data to monitor patients and to stratify individual risk, to ensure that what has been said has been done, as well as to inform practice and policies” through, for example, the establishment of diabetes registers.
The focus needs to be on “lifelong integrated care, the right treatment at the right time,” she emphasized. History-taking, clinical and laboratory assessments, as well as monitoring of macrovascular and microvascular complications, comorbidities, and medications, are all key.
Just a few simple things, if properly implemented, could make a big difference, Dr. Chan stressed.
For example, implementing a structured lifestyle intervention and use of metformin can each prevent or delay type 2 diabetes in individuals with impaired glucose tolerance by 30%-50%, and sustained weight reduction in patients with obesity by 15 kg (33 lb) or more can induce remission of type 2 diabetes for up to 2 years.
And there are plenty of medications that are “very affordable even in low- and middle-income countries” to treat diabetes and associated risk factors, including metformin, “statins, and RAS inhibitors,” she noted.
For instance, the 10 low- and middle-income countries with the greatest burden of diabetes (China, India, Brazil, Mexico, Indonesia, Egypt, Pakistan, Bangladesh, Turkey, Thailand) account for 217 million cases of type 2 diabetes, representing nearly 50% of all diabetes cases.
The commission estimated that 3.2 million of these individuals would die in 3 years if not treated, with 1.3 million of these deaths due to cardiovascular disease.
By reducing hemoglobin A1c, blood pressure, and LDL-cholesterol through achieving a diagnosis rate of 50%, ensuring access to essential medicines in at least 70% of patients, and with a support system to sustain reductions in these risk factors over 3 years, up to 800,000 premature deaths could be avoided.
People with type 1 diabetes dying; WHO launches initiative
In an accompanying commentary (2020 Nov 12. doi: 10.1016/S0140-6736[20]32378-3), Katie Dain, chief executive officer of the Noncommunicable Diseases (NCD) Alliance, points out that only half of people living with diabetes around the world – and just one in seven in Africa – have reliable access to insulin.
“Lots of people with type 1 diabetes are still dying due to lack of insulin,” Dr. Chan said in an interview. “We need to elevate basic care to intermediate and ensure that basal-bolus insulin and glucose-monitoring tools are available and that patients are trained in self-care. In that way, 80% of type 1 diabetes deaths could be prevented.”
Ms. 3Dain agrees, stressing, “Political rhetoric and commitments have yet to translate into sufficient and sustainable action for people living with diabetes worldwide, and particularly for those in [low- and middle-income countries].”
The Lancet Commission document also emphasizes the importance of support for pregnant women with diabetes and attention to the psychosocial needs of people with diabetes.
And it stresses society-, population-, and community-based strategies for type 2 diabetes prevention including health awareness programs, food policies, and broad use of nonphysician personnel to deliver diabetes prevention efforts.
In tandem with World Diabetes Day, the World Health Organization will announce the development of the WHO Global Diabetes Compact, which will be launched in April 2021.
This will aim to implement the commission’s recommendations through partnerships with governments, care providers, patient advocates, and nongovernmental organizations.
Together, they will “support countries to mobilize resources and accelerate structural transformations, which will enable the scale-up of access to essential diabetes medicines and technologies, inclusion of diagnosis and treatment of diabetes in primary health care and universal health coverage packages, and reduction of major population-level diabetes risk factors such as obesity,” according to another Lancet editorial accompanying the report.
“The evidence-base for improving diabetes prevention and care is strong. The question now for diabetes advocates is how to achieve the comprehensive, systems-level change needed to translate this evidence into action.”
Dr. Chan has reported receiving grants from AstraZeneca, Lilly, Lee Powder, Hua Medicine, and Qualigenics, as well as grants and personal fees from Bayer, Boehringer Ingelheim, Sanofi, Novartis, Merck, and MSD outside the submitted work. She has reported being the chief executive officer (pro bono) of the Asia Diabetes Foundation and a cofounder of GemVCare. She also holds a patent for genetic markers for diabetes and its complications. Ms. Dain has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The article was published online Nov. 12, just ahead of World Diabetes Day.
Of the 463 million people with diabetes worldwide in 2019, 80% live in low- and middle-income countries. The condition reduces life expectancy in middle-aged adults by 4-10 years, including increasing the risk of death from cardiovascular disease, kidney disease, and cancer by up to threefold. It is also a leading cause of nontraumatic amputation and blindness.
Use of evidence-based interventions, if implemented and managed properly, could prevent thousands of deaths globally every day, stressed the commission.
“There is an enormous amount of knowledge that we have amassed over the years. We need good preventive care and we need to ensure that diabetes patients, once diagnosed, have good continuous care. There is an urgent need for decision-makers, policymakers, and payers to make things happen,” the leader of the multidisciplinary commission, Juliana C.N. Chan, MBChB, MD, said in an interview.
And now diabetes has emerged as a major risk factor for death from COVID-19, particularly in the setting of inadequate glycemic control.
“COVID-19 has exposed the vulnerability of individuals with diabetes,” said Dr. Chan, of the Hong Kong Institute of Diabetes and Obesity. “We should use the pandemic as an opportunity to implement solutions.”
Physician education key, trickling down to field workers and patients
First on the agenda, she says, should be “physician education. There are many primary care providers and internal medicine physicians whose knowledge needs to be updated.”
“Then doctors need to transfer this information to other people, such as nurses and community field workers. We cannot just rely on doctors; we need to train nonmedics” so that knowledge about how to prevent, treat, and manage diabetes long term is communicated right down the health care chain, she explained.
“They need to know how to look at people’s eyes and feet, how to do blood and urine tests, and how to collect data. Then they need to educate patients on what they should be doing, on how to practice self-care,” she added.
“We need to change our way of thinking, redesign clinic flow and how you build a team. And those care teams need to know how to collect data, and then use that data to monitor patients and to stratify individual risk, to ensure that what has been said has been done, as well as to inform practice and policies” through, for example, the establishment of diabetes registers.
The focus needs to be on “lifelong integrated care, the right treatment at the right time,” she emphasized. History-taking, clinical and laboratory assessments, as well as monitoring of macrovascular and microvascular complications, comorbidities, and medications, are all key.
Just a few simple things, if properly implemented, could make a big difference, Dr. Chan stressed.
For example, implementing a structured lifestyle intervention and use of metformin can each prevent or delay type 2 diabetes in individuals with impaired glucose tolerance by 30%-50%, and sustained weight reduction in patients with obesity by 15 kg (33 lb) or more can induce remission of type 2 diabetes for up to 2 years.
And there are plenty of medications that are “very affordable even in low- and middle-income countries” to treat diabetes and associated risk factors, including metformin, “statins, and RAS inhibitors,” she noted.
For instance, the 10 low- and middle-income countries with the greatest burden of diabetes (China, India, Brazil, Mexico, Indonesia, Egypt, Pakistan, Bangladesh, Turkey, Thailand) account for 217 million cases of type 2 diabetes, representing nearly 50% of all diabetes cases.
The commission estimated that 3.2 million of these individuals would die in 3 years if not treated, with 1.3 million of these deaths due to cardiovascular disease.
By reducing hemoglobin A1c, blood pressure, and LDL-cholesterol through achieving a diagnosis rate of 50%, ensuring access to essential medicines in at least 70% of patients, and with a support system to sustain reductions in these risk factors over 3 years, up to 800,000 premature deaths could be avoided.
People with type 1 diabetes dying; WHO launches initiative
In an accompanying commentary (2020 Nov 12. doi: 10.1016/S0140-6736[20]32378-3), Katie Dain, chief executive officer of the Noncommunicable Diseases (NCD) Alliance, points out that only half of people living with diabetes around the world – and just one in seven in Africa – have reliable access to insulin.
“Lots of people with type 1 diabetes are still dying due to lack of insulin,” Dr. Chan said in an interview. “We need to elevate basic care to intermediate and ensure that basal-bolus insulin and glucose-monitoring tools are available and that patients are trained in self-care. In that way, 80% of type 1 diabetes deaths could be prevented.”
Ms. 3Dain agrees, stressing, “Political rhetoric and commitments have yet to translate into sufficient and sustainable action for people living with diabetes worldwide, and particularly for those in [low- and middle-income countries].”
The Lancet Commission document also emphasizes the importance of support for pregnant women with diabetes and attention to the psychosocial needs of people with diabetes.
And it stresses society-, population-, and community-based strategies for type 2 diabetes prevention including health awareness programs, food policies, and broad use of nonphysician personnel to deliver diabetes prevention efforts.
In tandem with World Diabetes Day, the World Health Organization will announce the development of the WHO Global Diabetes Compact, which will be launched in April 2021.
This will aim to implement the commission’s recommendations through partnerships with governments, care providers, patient advocates, and nongovernmental organizations.
Together, they will “support countries to mobilize resources and accelerate structural transformations, which will enable the scale-up of access to essential diabetes medicines and technologies, inclusion of diagnosis and treatment of diabetes in primary health care and universal health coverage packages, and reduction of major population-level diabetes risk factors such as obesity,” according to another Lancet editorial accompanying the report.
“The evidence-base for improving diabetes prevention and care is strong. The question now for diabetes advocates is how to achieve the comprehensive, systems-level change needed to translate this evidence into action.”
Dr. Chan has reported receiving grants from AstraZeneca, Lilly, Lee Powder, Hua Medicine, and Qualigenics, as well as grants and personal fees from Bayer, Boehringer Ingelheim, Sanofi, Novartis, Merck, and MSD outside the submitted work. She has reported being the chief executive officer (pro bono) of the Asia Diabetes Foundation and a cofounder of GemVCare. She also holds a patent for genetic markers for diabetes and its complications. Ms. Dain has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The article was published online Nov. 12, just ahead of World Diabetes Day.
Of the 463 million people with diabetes worldwide in 2019, 80% live in low- and middle-income countries. The condition reduces life expectancy in middle-aged adults by 4-10 years, including increasing the risk of death from cardiovascular disease, kidney disease, and cancer by up to threefold. It is also a leading cause of nontraumatic amputation and blindness.
Use of evidence-based interventions, if implemented and managed properly, could prevent thousands of deaths globally every day, stressed the commission.
“There is an enormous amount of knowledge that we have amassed over the years. We need good preventive care and we need to ensure that diabetes patients, once diagnosed, have good continuous care. There is an urgent need for decision-makers, policymakers, and payers to make things happen,” the leader of the multidisciplinary commission, Juliana C.N. Chan, MBChB, MD, said in an interview.
And now diabetes has emerged as a major risk factor for death from COVID-19, particularly in the setting of inadequate glycemic control.
“COVID-19 has exposed the vulnerability of individuals with diabetes,” said Dr. Chan, of the Hong Kong Institute of Diabetes and Obesity. “We should use the pandemic as an opportunity to implement solutions.”
Physician education key, trickling down to field workers and patients
First on the agenda, she says, should be “physician education. There are many primary care providers and internal medicine physicians whose knowledge needs to be updated.”
“Then doctors need to transfer this information to other people, such as nurses and community field workers. We cannot just rely on doctors; we need to train nonmedics” so that knowledge about how to prevent, treat, and manage diabetes long term is communicated right down the health care chain, she explained.
“They need to know how to look at people’s eyes and feet, how to do blood and urine tests, and how to collect data. Then they need to educate patients on what they should be doing, on how to practice self-care,” she added.
“We need to change our way of thinking, redesign clinic flow and how you build a team. And those care teams need to know how to collect data, and then use that data to monitor patients and to stratify individual risk, to ensure that what has been said has been done, as well as to inform practice and policies” through, for example, the establishment of diabetes registers.
The focus needs to be on “lifelong integrated care, the right treatment at the right time,” she emphasized. History-taking, clinical and laboratory assessments, as well as monitoring of macrovascular and microvascular complications, comorbidities, and medications, are all key.
Just a few simple things, if properly implemented, could make a big difference, Dr. Chan stressed.
For example, implementing a structured lifestyle intervention and use of metformin can each prevent or delay type 2 diabetes in individuals with impaired glucose tolerance by 30%-50%, and sustained weight reduction in patients with obesity by 15 kg (33 lb) or more can induce remission of type 2 diabetes for up to 2 years.
And there are plenty of medications that are “very affordable even in low- and middle-income countries” to treat diabetes and associated risk factors, including metformin, “statins, and RAS inhibitors,” she noted.
For instance, the 10 low- and middle-income countries with the greatest burden of diabetes (China, India, Brazil, Mexico, Indonesia, Egypt, Pakistan, Bangladesh, Turkey, Thailand) account for 217 million cases of type 2 diabetes, representing nearly 50% of all diabetes cases.
The commission estimated that 3.2 million of these individuals would die in 3 years if not treated, with 1.3 million of these deaths due to cardiovascular disease.
By reducing hemoglobin A1c, blood pressure, and LDL-cholesterol through achieving a diagnosis rate of 50%, ensuring access to essential medicines in at least 70% of patients, and with a support system to sustain reductions in these risk factors over 3 years, up to 800,000 premature deaths could be avoided.
People with type 1 diabetes dying; WHO launches initiative
In an accompanying commentary (2020 Nov 12. doi: 10.1016/S0140-6736[20]32378-3), Katie Dain, chief executive officer of the Noncommunicable Diseases (NCD) Alliance, points out that only half of people living with diabetes around the world – and just one in seven in Africa – have reliable access to insulin.
“Lots of people with type 1 diabetes are still dying due to lack of insulin,” Dr. Chan said in an interview. “We need to elevate basic care to intermediate and ensure that basal-bolus insulin and glucose-monitoring tools are available and that patients are trained in self-care. In that way, 80% of type 1 diabetes deaths could be prevented.”
Ms. 3Dain agrees, stressing, “Political rhetoric and commitments have yet to translate into sufficient and sustainable action for people living with diabetes worldwide, and particularly for those in [low- and middle-income countries].”
The Lancet Commission document also emphasizes the importance of support for pregnant women with diabetes and attention to the psychosocial needs of people with diabetes.
And it stresses society-, population-, and community-based strategies for type 2 diabetes prevention including health awareness programs, food policies, and broad use of nonphysician personnel to deliver diabetes prevention efforts.
In tandem with World Diabetes Day, the World Health Organization will announce the development of the WHO Global Diabetes Compact, which will be launched in April 2021.
This will aim to implement the commission’s recommendations through partnerships with governments, care providers, patient advocates, and nongovernmental organizations.
Together, they will “support countries to mobilize resources and accelerate structural transformations, which will enable the scale-up of access to essential diabetes medicines and technologies, inclusion of diagnosis and treatment of diabetes in primary health care and universal health coverage packages, and reduction of major population-level diabetes risk factors such as obesity,” according to another Lancet editorial accompanying the report.
“The evidence-base for improving diabetes prevention and care is strong. The question now for diabetes advocates is how to achieve the comprehensive, systems-level change needed to translate this evidence into action.”
Dr. Chan has reported receiving grants from AstraZeneca, Lilly, Lee Powder, Hua Medicine, and Qualigenics, as well as grants and personal fees from Bayer, Boehringer Ingelheim, Sanofi, Novartis, Merck, and MSD outside the submitted work. She has reported being the chief executive officer (pro bono) of the Asia Diabetes Foundation and a cofounder of GemVCare. She also holds a patent for genetic markers for diabetes and its complications. Ms. Dain has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Pediatric News board welcomes back Dr. Breach Washington
She currently is employed as a medical director at WellCare of North Carolina/Centene. Her career spans 32 years in Charlotte, N.C., where she has practiced pediatrics and held medical director positions in a large health care system and a managed care organization, as well as worked in private practice and in a public health clinic.
A native of North Babylon, N.Y., she received her undergraduate degree at Hofstra University, Hempstead, N.Y., and her medical degree at George Washington University, Washington. Dr. Breach Washington has been an advocate for children, access to health care, and diversity and inclusion.
An active member of the American Academy of Pediatrics, Dr. Breach Washington served as an elected representative to the AAP National Nominating Committee, following her term as president of the North Carolina chapter of the AAP.
She is a past pediatric section chair of the National Medical Association and past president of the Charlotte Medical, Dental, and Pharmaceutical Society. She has served on the boards of the Simmons Branch YMCA, the Mecklenburg County Medical Society, and the Teen Health Connection (a facility of Carolinas Medical Center), plus many associated committees.
Dr. Breach Washington serves the community as an active life member of Alpha Kappa Alpha Sorority, Jack and Jill of America, and a member of The Links. She is the recipient of several awards for her commitment to diversity and inclusion, philanthropy, and community service.
Dr. Breach Washington is married and has an adult daughter. She is an avid fan of all sports, a dancer, and enjoys theater, fine dining, and travel.
She currently is employed as a medical director at WellCare of North Carolina/Centene. Her career spans 32 years in Charlotte, N.C., where she has practiced pediatrics and held medical director positions in a large health care system and a managed care organization, as well as worked in private practice and in a public health clinic.
A native of North Babylon, N.Y., she received her undergraduate degree at Hofstra University, Hempstead, N.Y., and her medical degree at George Washington University, Washington. Dr. Breach Washington has been an advocate for children, access to health care, and diversity and inclusion.
An active member of the American Academy of Pediatrics, Dr. Breach Washington served as an elected representative to the AAP National Nominating Committee, following her term as president of the North Carolina chapter of the AAP.
She is a past pediatric section chair of the National Medical Association and past president of the Charlotte Medical, Dental, and Pharmaceutical Society. She has served on the boards of the Simmons Branch YMCA, the Mecklenburg County Medical Society, and the Teen Health Connection (a facility of Carolinas Medical Center), plus many associated committees.
Dr. Breach Washington serves the community as an active life member of Alpha Kappa Alpha Sorority, Jack and Jill of America, and a member of The Links. She is the recipient of several awards for her commitment to diversity and inclusion, philanthropy, and community service.
Dr. Breach Washington is married and has an adult daughter. She is an avid fan of all sports, a dancer, and enjoys theater, fine dining, and travel.
She currently is employed as a medical director at WellCare of North Carolina/Centene. Her career spans 32 years in Charlotte, N.C., where she has practiced pediatrics and held medical director positions in a large health care system and a managed care organization, as well as worked in private practice and in a public health clinic.
A native of North Babylon, N.Y., she received her undergraduate degree at Hofstra University, Hempstead, N.Y., and her medical degree at George Washington University, Washington. Dr. Breach Washington has been an advocate for children, access to health care, and diversity and inclusion.
An active member of the American Academy of Pediatrics, Dr. Breach Washington served as an elected representative to the AAP National Nominating Committee, following her term as president of the North Carolina chapter of the AAP.
She is a past pediatric section chair of the National Medical Association and past president of the Charlotte Medical, Dental, and Pharmaceutical Society. She has served on the boards of the Simmons Branch YMCA, the Mecklenburg County Medical Society, and the Teen Health Connection (a facility of Carolinas Medical Center), plus many associated committees.
Dr. Breach Washington serves the community as an active life member of Alpha Kappa Alpha Sorority, Jack and Jill of America, and a member of The Links. She is the recipient of several awards for her commitment to diversity and inclusion, philanthropy, and community service.
Dr. Breach Washington is married and has an adult daughter. She is an avid fan of all sports, a dancer, and enjoys theater, fine dining, and travel.