User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
New deep dive into Paxlovid interactions with CVD meds
Nirmatrelvir/ritonavir (Paxlovid) has been a game changer for high-risk patients with early COVID-19 symptoms but has significant interactions with commonly used cardiovascular medications, a new paper cautions.
COVID-19 patients with cardiovascular disease (CVD) or risk factors such as diabetes, hypertension, and chronic kidney disease are at high risk of severe disease and account for the lion’s share of those receiving Paxlovid. Data from the initial EPIC-HR trial and recent real-world data also suggest they’re among the most likely to benefit from the oral antiviral, regardless of their COVID-19 vaccination status.
“But at the same time, it unfortunately interacts with many very commonly prescribed cardiovascular medications and with many of them in a very clinically meaningful way, which may lead to serious adverse consequences,” senior author Sarju Ganatra, MD, said in an interview. “So, while it’s being prescribed with a good intention to help these people, we may actually end up doing more harm than good.
“We don’t want to deter people from getting their necessary COVID-19 treatment, which is excellent for the most part these days as an outpatient,” he added. “So, we felt the need to make a comprehensive list of cardiac medications and level of interactions with Paxlovid and also to help the clinicians and prescribers at the point of care to make the clinical decision of what modifications they may need to do.”
The paper, published online in the Journal of the American College of Cardiology, details drug-drug interactions with some 80 CV medications including statins, antihypertensive agents, heart failure therapies, and antiplatelet/anticoagulants.
It also includes a color-coded figure denoting whether a drug is safe to coadminister with Paxlovid, may potentially interact and require a dose adjustment or temporary discontinuation, or is contraindicated.
Among the commonly used blood thinners, for example, the paper notes that Paxlovid significantly increases drug levels of the direct oral anticoagulants (DOACs) apixaban, rivaroxaban, edoxaban, and dabigatran and, thus, increases the risk of bleeding.
“It can still be administered, if it’s necessary, but the dose of the DOAC either needs to be reduced or held depending on what they are getting it for, whether they’re getting it for pulmonary embolism or atrial fibrillation, and we adjust for all those things in the table in the paper,” said Dr. Ganatra, from Lahey Hospital and Medical Center, Burlington, Mass.
When the DOAC can’t be interrupted or dose adjusted, however, Paxlovid should not be given, the experts said. The antiviral is safe to use with enoxaparin, a low-molecular-weight heparin, but can increase or decrease levels of warfarin and should be used with close international normalized ratio monitoring.
For patients on antiplatelet agents, clinicians are advised to avoid prescribing nirmatrelvir/ritonavir to those on ticagrelor or clopidogrel unless the agents can be replaced by prasugrel.
Ritonavir – an inhibitor of cytochrome P 450 enzymes, particularly CYP3A4 – poses an increased risk of bleeding when given with ticagrelor, a CYP3A4 substrate, and decreases the active metabolite of clopidogrel, cutting its platelet inhibition by 20%. Although there’s a twofold decrease in the maximum concentration of prasugrel in patients on ritonavir, this does not affect its antiplatelet activity, the paper explains.
Among the lipid-lowering agents, experts suggested temporarily withholding atorvastatin, rosuvastatin, simvastatin, and lovastatin because of an increased risk for myopathy and liver toxicity but say that other statins, fibrates, ezetimibe, and the proprotein convertase subtilisin/kexin type 9 inhibitors evolocumab and alirocumab are safe to coadminister with Paxlovid.
While statins typically leave the body within hours, most of the antiarrhythmic drugs, except for sotalol, are not safe to give with Paxlovid, Dr. Ganatra said. It’s technically not feasible to hold these drugs because most have long half-lives, reaching about 100 days, for example, for amiodarone.
“It’s going to hang around in your system for a long time, so you don’t want to be falsely reassured that you’re holding the drug and it’s going to be fine to go back slowly,” he said. “You need to look for alternative therapies in those scenarios for COVID-19 treatment, which could be other antivirals, or a monoclonal antibody individualized to the patient’s risk.”
Although there’s limited clinical information regarding interaction-related adverse events with Paxlovid, the team used pharmacokinetics and pharmacodynamics data to provide the guidance. Serious adverse events are also well documented for ritonavir, which has been prescribed for years to treat HIV, Dr. Ganatra noted.
The Infectious Disease Society of America also published guidance on the management of potential drug interactions with Paxlovid in May and, earlier in October, the Food and Drug Administration updated its Paxlovid patient eligibility screening checklist.
Still, most prescribers are actually primary care physicians and even pharmacists, who may not be completely attuned, said Dr. Ganatra, who noted that some centers have started programs to help connect primary care physicians with their cardiology colleagues to check on CV drugs in their COVID-19 patients.
“We need to be thinking more broadly and at a system level where the hospital or health care system leverages the electronic health record systems,” he said. “Most of them are sophisticated enough to incorporate simple drug-drug interaction information, so if you try to prescribe someone Paxlovid and it’s a heart transplant patient who is on immunosuppressive therapy or a patient on a blood thinner, then it should give you a warning ... or at least give them a link to our paper or other valuable resources.
“If someone is on a blood thinner and the blood thinner level goes up by ninefold, we can only imagine what we would be dealing with,” Dr. Ganatra said. “So, these interactions should be taken very seriously and I think it’s worth the time and investment.”
The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Nirmatrelvir/ritonavir (Paxlovid) has been a game changer for high-risk patients with early COVID-19 symptoms but has significant interactions with commonly used cardiovascular medications, a new paper cautions.
COVID-19 patients with cardiovascular disease (CVD) or risk factors such as diabetes, hypertension, and chronic kidney disease are at high risk of severe disease and account for the lion’s share of those receiving Paxlovid. Data from the initial EPIC-HR trial and recent real-world data also suggest they’re among the most likely to benefit from the oral antiviral, regardless of their COVID-19 vaccination status.
“But at the same time, it unfortunately interacts with many very commonly prescribed cardiovascular medications and with many of them in a very clinically meaningful way, which may lead to serious adverse consequences,” senior author Sarju Ganatra, MD, said in an interview. “So, while it’s being prescribed with a good intention to help these people, we may actually end up doing more harm than good.
“We don’t want to deter people from getting their necessary COVID-19 treatment, which is excellent for the most part these days as an outpatient,” he added. “So, we felt the need to make a comprehensive list of cardiac medications and level of interactions with Paxlovid and also to help the clinicians and prescribers at the point of care to make the clinical decision of what modifications they may need to do.”
The paper, published online in the Journal of the American College of Cardiology, details drug-drug interactions with some 80 CV medications including statins, antihypertensive agents, heart failure therapies, and antiplatelet/anticoagulants.
It also includes a color-coded figure denoting whether a drug is safe to coadminister with Paxlovid, may potentially interact and require a dose adjustment or temporary discontinuation, or is contraindicated.
Among the commonly used blood thinners, for example, the paper notes that Paxlovid significantly increases drug levels of the direct oral anticoagulants (DOACs) apixaban, rivaroxaban, edoxaban, and dabigatran and, thus, increases the risk of bleeding.
“It can still be administered, if it’s necessary, but the dose of the DOAC either needs to be reduced or held depending on what they are getting it for, whether they’re getting it for pulmonary embolism or atrial fibrillation, and we adjust for all those things in the table in the paper,” said Dr. Ganatra, from Lahey Hospital and Medical Center, Burlington, Mass.
When the DOAC can’t be interrupted or dose adjusted, however, Paxlovid should not be given, the experts said. The antiviral is safe to use with enoxaparin, a low-molecular-weight heparin, but can increase or decrease levels of warfarin and should be used with close international normalized ratio monitoring.
For patients on antiplatelet agents, clinicians are advised to avoid prescribing nirmatrelvir/ritonavir to those on ticagrelor or clopidogrel unless the agents can be replaced by prasugrel.
Ritonavir – an inhibitor of cytochrome P 450 enzymes, particularly CYP3A4 – poses an increased risk of bleeding when given with ticagrelor, a CYP3A4 substrate, and decreases the active metabolite of clopidogrel, cutting its platelet inhibition by 20%. Although there’s a twofold decrease in the maximum concentration of prasugrel in patients on ritonavir, this does not affect its antiplatelet activity, the paper explains.
Among the lipid-lowering agents, experts suggested temporarily withholding atorvastatin, rosuvastatin, simvastatin, and lovastatin because of an increased risk for myopathy and liver toxicity but say that other statins, fibrates, ezetimibe, and the proprotein convertase subtilisin/kexin type 9 inhibitors evolocumab and alirocumab are safe to coadminister with Paxlovid.
While statins typically leave the body within hours, most of the antiarrhythmic drugs, except for sotalol, are not safe to give with Paxlovid, Dr. Ganatra said. It’s technically not feasible to hold these drugs because most have long half-lives, reaching about 100 days, for example, for amiodarone.
“It’s going to hang around in your system for a long time, so you don’t want to be falsely reassured that you’re holding the drug and it’s going to be fine to go back slowly,” he said. “You need to look for alternative therapies in those scenarios for COVID-19 treatment, which could be other antivirals, or a monoclonal antibody individualized to the patient’s risk.”
Although there’s limited clinical information regarding interaction-related adverse events with Paxlovid, the team used pharmacokinetics and pharmacodynamics data to provide the guidance. Serious adverse events are also well documented for ritonavir, which has been prescribed for years to treat HIV, Dr. Ganatra noted.
The Infectious Disease Society of America also published guidance on the management of potential drug interactions with Paxlovid in May and, earlier in October, the Food and Drug Administration updated its Paxlovid patient eligibility screening checklist.
Still, most prescribers are actually primary care physicians and even pharmacists, who may not be completely attuned, said Dr. Ganatra, who noted that some centers have started programs to help connect primary care physicians with their cardiology colleagues to check on CV drugs in their COVID-19 patients.
“We need to be thinking more broadly and at a system level where the hospital or health care system leverages the electronic health record systems,” he said. “Most of them are sophisticated enough to incorporate simple drug-drug interaction information, so if you try to prescribe someone Paxlovid and it’s a heart transplant patient who is on immunosuppressive therapy or a patient on a blood thinner, then it should give you a warning ... or at least give them a link to our paper or other valuable resources.
“If someone is on a blood thinner and the blood thinner level goes up by ninefold, we can only imagine what we would be dealing with,” Dr. Ganatra said. “So, these interactions should be taken very seriously and I think it’s worth the time and investment.”
The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Nirmatrelvir/ritonavir (Paxlovid) has been a game changer for high-risk patients with early COVID-19 symptoms but has significant interactions with commonly used cardiovascular medications, a new paper cautions.
COVID-19 patients with cardiovascular disease (CVD) or risk factors such as diabetes, hypertension, and chronic kidney disease are at high risk of severe disease and account for the lion’s share of those receiving Paxlovid. Data from the initial EPIC-HR trial and recent real-world data also suggest they’re among the most likely to benefit from the oral antiviral, regardless of their COVID-19 vaccination status.
“But at the same time, it unfortunately interacts with many very commonly prescribed cardiovascular medications and with many of them in a very clinically meaningful way, which may lead to serious adverse consequences,” senior author Sarju Ganatra, MD, said in an interview. “So, while it’s being prescribed with a good intention to help these people, we may actually end up doing more harm than good.
“We don’t want to deter people from getting their necessary COVID-19 treatment, which is excellent for the most part these days as an outpatient,” he added. “So, we felt the need to make a comprehensive list of cardiac medications and level of interactions with Paxlovid and also to help the clinicians and prescribers at the point of care to make the clinical decision of what modifications they may need to do.”
The paper, published online in the Journal of the American College of Cardiology, details drug-drug interactions with some 80 CV medications including statins, antihypertensive agents, heart failure therapies, and antiplatelet/anticoagulants.
It also includes a color-coded figure denoting whether a drug is safe to coadminister with Paxlovid, may potentially interact and require a dose adjustment or temporary discontinuation, or is contraindicated.
Among the commonly used blood thinners, for example, the paper notes that Paxlovid significantly increases drug levels of the direct oral anticoagulants (DOACs) apixaban, rivaroxaban, edoxaban, and dabigatran and, thus, increases the risk of bleeding.
“It can still be administered, if it’s necessary, but the dose of the DOAC either needs to be reduced or held depending on what they are getting it for, whether they’re getting it for pulmonary embolism or atrial fibrillation, and we adjust for all those things in the table in the paper,” said Dr. Ganatra, from Lahey Hospital and Medical Center, Burlington, Mass.
When the DOAC can’t be interrupted or dose adjusted, however, Paxlovid should not be given, the experts said. The antiviral is safe to use with enoxaparin, a low-molecular-weight heparin, but can increase or decrease levels of warfarin and should be used with close international normalized ratio monitoring.
For patients on antiplatelet agents, clinicians are advised to avoid prescribing nirmatrelvir/ritonavir to those on ticagrelor or clopidogrel unless the agents can be replaced by prasugrel.
Ritonavir – an inhibitor of cytochrome P 450 enzymes, particularly CYP3A4 – poses an increased risk of bleeding when given with ticagrelor, a CYP3A4 substrate, and decreases the active metabolite of clopidogrel, cutting its platelet inhibition by 20%. Although there’s a twofold decrease in the maximum concentration of prasugrel in patients on ritonavir, this does not affect its antiplatelet activity, the paper explains.
Among the lipid-lowering agents, experts suggested temporarily withholding atorvastatin, rosuvastatin, simvastatin, and lovastatin because of an increased risk for myopathy and liver toxicity but say that other statins, fibrates, ezetimibe, and the proprotein convertase subtilisin/kexin type 9 inhibitors evolocumab and alirocumab are safe to coadminister with Paxlovid.
While statins typically leave the body within hours, most of the antiarrhythmic drugs, except for sotalol, are not safe to give with Paxlovid, Dr. Ganatra said. It’s technically not feasible to hold these drugs because most have long half-lives, reaching about 100 days, for example, for amiodarone.
“It’s going to hang around in your system for a long time, so you don’t want to be falsely reassured that you’re holding the drug and it’s going to be fine to go back slowly,” he said. “You need to look for alternative therapies in those scenarios for COVID-19 treatment, which could be other antivirals, or a monoclonal antibody individualized to the patient’s risk.”
Although there’s limited clinical information regarding interaction-related adverse events with Paxlovid, the team used pharmacokinetics and pharmacodynamics data to provide the guidance. Serious adverse events are also well documented for ritonavir, which has been prescribed for years to treat HIV, Dr. Ganatra noted.
The Infectious Disease Society of America also published guidance on the management of potential drug interactions with Paxlovid in May and, earlier in October, the Food and Drug Administration updated its Paxlovid patient eligibility screening checklist.
Still, most prescribers are actually primary care physicians and even pharmacists, who may not be completely attuned, said Dr. Ganatra, who noted that some centers have started programs to help connect primary care physicians with their cardiology colleagues to check on CV drugs in their COVID-19 patients.
“We need to be thinking more broadly and at a system level where the hospital or health care system leverages the electronic health record systems,” he said. “Most of them are sophisticated enough to incorporate simple drug-drug interaction information, so if you try to prescribe someone Paxlovid and it’s a heart transplant patient who is on immunosuppressive therapy or a patient on a blood thinner, then it should give you a warning ... or at least give them a link to our paper or other valuable resources.
“If someone is on a blood thinner and the blood thinner level goes up by ninefold, we can only imagine what we would be dealing with,” Dr. Ganatra said. “So, these interactions should be taken very seriously and I think it’s worth the time and investment.”
The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Starting a blog
. Health information is one of the most popular topics people search for online. Starting a physician blog can provide your practice with promotional and marketing benefits that you may have a difficult time finding elsewhere. A blog can be an effective way to drive traffic to your website, establish yourself as an authority or expert in a particular area, and stay on the radar with your patients. However, there are a few things you should think about before you start.
Start by determining what you want to accomplish. Do you want to reach quantitative milestones, like a certain number of followers, or are you looking to increase your website traffic from potential patients? One goal will probably be to augment the health knowledge of your patients. Decide early on what your benchmarks will be and how you will track them.
Next, determine who your potential readers are. Initially, most will probably be local (your existing patient base and their family and friends), but your audience may expand geographically as your blog gains in popularity.
By now, you probably realize that blogging will require a significant commitment, over and above the time needed to write the content. Decide whether you have the time and energy to take this on yourself, or whether help will be needed. Ideally, you should have one person in charge of all your social media efforts, so that everything is consistent and has the same voice. That person can be in-house, or you can outsource to any of the many companies that administer blogs and other media functions. (As always, I have no financial interest in any company or service mentioned in this column.)
The advantage of hiring an outside administrator is that a professionally designed blog will be far more attractive and polished than anything you could build yourself. Furthermore, an experienced designer will employ “search engine optimization” (SEO), meaning that content will be created using key words and phrases that will make it readily visible to search engine users.
You can leave design and SEO to the pros, but don’t delegate the content itself; as captain of the ship you are responsible for all the facts and opinions on your blog. You may not be up to writing everything yourself, but anything you don’t write personally needs to be scrutinized by you personally to make sure that it is factually accurate and reflects your personal view. And remember that, once it’s online, it’s online forever; consider the ramifications of anything you post on any site – yours or others – before hitting the “send” button. “The most damaging item about you,” one consultant told me, “could well be something you post yourself.” Just ask any of several prominent politicians who have famously sabotaged their own careers online.
That said, don’t be shy about creating content. Patients appreciate factual information, but they value your opinions too. Give people content that will be of interest or benefit to them. This can include health-related tips, reminders, suggestions, whatever. If they are interested in it, they will keep reading and may even share it with others. You should also write about subjects – medical and otherwise – that interest you personally. If you have expertise in a particular field, be sure to write about that.
Your practice is a local business, so localize your blog to attract people from your area. Be sure to include local city keywords in your writing. You may also want to post about local events in which your practice is involved.
Try to avoid political diatribes. While most physicians have strong political opinions, and some are not shy about expressing them, there are many venues that are more appropriate for those discussions than medical blogs. Also avoid outright sales pitches. It’s fine to describe procedures that you offer, but aggressive solicitation will only turn readers off.
Keep any medical advice in general terms; don’t use any specific examples that might make a patient identifiable and generate a HIPAA violation.
If you are having trouble growing your readership, use your practice’s Facebook page to push blog updates into patients’ feeds. Additionally, track Twitter hashtags that are relevant to your practice, and use them to find existing online communities with an interest in your blog’s topics.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
*This article was updated 10/17/2022.
. Health information is one of the most popular topics people search for online. Starting a physician blog can provide your practice with promotional and marketing benefits that you may have a difficult time finding elsewhere. A blog can be an effective way to drive traffic to your website, establish yourself as an authority or expert in a particular area, and stay on the radar with your patients. However, there are a few things you should think about before you start.
Start by determining what you want to accomplish. Do you want to reach quantitative milestones, like a certain number of followers, or are you looking to increase your website traffic from potential patients? One goal will probably be to augment the health knowledge of your patients. Decide early on what your benchmarks will be and how you will track them.
Next, determine who your potential readers are. Initially, most will probably be local (your existing patient base and their family and friends), but your audience may expand geographically as your blog gains in popularity.
By now, you probably realize that blogging will require a significant commitment, over and above the time needed to write the content. Decide whether you have the time and energy to take this on yourself, or whether help will be needed. Ideally, you should have one person in charge of all your social media efforts, so that everything is consistent and has the same voice. That person can be in-house, or you can outsource to any of the many companies that administer blogs and other media functions. (As always, I have no financial interest in any company or service mentioned in this column.)
The advantage of hiring an outside administrator is that a professionally designed blog will be far more attractive and polished than anything you could build yourself. Furthermore, an experienced designer will employ “search engine optimization” (SEO), meaning that content will be created using key words and phrases that will make it readily visible to search engine users.
You can leave design and SEO to the pros, but don’t delegate the content itself; as captain of the ship you are responsible for all the facts and opinions on your blog. You may not be up to writing everything yourself, but anything you don’t write personally needs to be scrutinized by you personally to make sure that it is factually accurate and reflects your personal view. And remember that, once it’s online, it’s online forever; consider the ramifications of anything you post on any site – yours or others – before hitting the “send” button. “The most damaging item about you,” one consultant told me, “could well be something you post yourself.” Just ask any of several prominent politicians who have famously sabotaged their own careers online.
That said, don’t be shy about creating content. Patients appreciate factual information, but they value your opinions too. Give people content that will be of interest or benefit to them. This can include health-related tips, reminders, suggestions, whatever. If they are interested in it, they will keep reading and may even share it with others. You should also write about subjects – medical and otherwise – that interest you personally. If you have expertise in a particular field, be sure to write about that.
Your practice is a local business, so localize your blog to attract people from your area. Be sure to include local city keywords in your writing. You may also want to post about local events in which your practice is involved.
Try to avoid political diatribes. While most physicians have strong political opinions, and some are not shy about expressing them, there are many venues that are more appropriate for those discussions than medical blogs. Also avoid outright sales pitches. It’s fine to describe procedures that you offer, but aggressive solicitation will only turn readers off.
Keep any medical advice in general terms; don’t use any specific examples that might make a patient identifiable and generate a HIPAA violation.
If you are having trouble growing your readership, use your practice’s Facebook page to push blog updates into patients’ feeds. Additionally, track Twitter hashtags that are relevant to your practice, and use them to find existing online communities with an interest in your blog’s topics.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
*This article was updated 10/17/2022.
. Health information is one of the most popular topics people search for online. Starting a physician blog can provide your practice with promotional and marketing benefits that you may have a difficult time finding elsewhere. A blog can be an effective way to drive traffic to your website, establish yourself as an authority or expert in a particular area, and stay on the radar with your patients. However, there are a few things you should think about before you start.
Start by determining what you want to accomplish. Do you want to reach quantitative milestones, like a certain number of followers, or are you looking to increase your website traffic from potential patients? One goal will probably be to augment the health knowledge of your patients. Decide early on what your benchmarks will be and how you will track them.
Next, determine who your potential readers are. Initially, most will probably be local (your existing patient base and their family and friends), but your audience may expand geographically as your blog gains in popularity.
By now, you probably realize that blogging will require a significant commitment, over and above the time needed to write the content. Decide whether you have the time and energy to take this on yourself, or whether help will be needed. Ideally, you should have one person in charge of all your social media efforts, so that everything is consistent and has the same voice. That person can be in-house, or you can outsource to any of the many companies that administer blogs and other media functions. (As always, I have no financial interest in any company or service mentioned in this column.)
The advantage of hiring an outside administrator is that a professionally designed blog will be far more attractive and polished than anything you could build yourself. Furthermore, an experienced designer will employ “search engine optimization” (SEO), meaning that content will be created using key words and phrases that will make it readily visible to search engine users.
You can leave design and SEO to the pros, but don’t delegate the content itself; as captain of the ship you are responsible for all the facts and opinions on your blog. You may not be up to writing everything yourself, but anything you don’t write personally needs to be scrutinized by you personally to make sure that it is factually accurate and reflects your personal view. And remember that, once it’s online, it’s online forever; consider the ramifications of anything you post on any site – yours or others – before hitting the “send” button. “The most damaging item about you,” one consultant told me, “could well be something you post yourself.” Just ask any of several prominent politicians who have famously sabotaged their own careers online.
That said, don’t be shy about creating content. Patients appreciate factual information, but they value your opinions too. Give people content that will be of interest or benefit to them. This can include health-related tips, reminders, suggestions, whatever. If they are interested in it, they will keep reading and may even share it with others. You should also write about subjects – medical and otherwise – that interest you personally. If you have expertise in a particular field, be sure to write about that.
Your practice is a local business, so localize your blog to attract people from your area. Be sure to include local city keywords in your writing. You may also want to post about local events in which your practice is involved.
Try to avoid political diatribes. While most physicians have strong political opinions, and some are not shy about expressing them, there are many venues that are more appropriate for those discussions than medical blogs. Also avoid outright sales pitches. It’s fine to describe procedures that you offer, but aggressive solicitation will only turn readers off.
Keep any medical advice in general terms; don’t use any specific examples that might make a patient identifiable and generate a HIPAA violation.
If you are having trouble growing your readership, use your practice’s Facebook page to push blog updates into patients’ feeds. Additionally, track Twitter hashtags that are relevant to your practice, and use them to find existing online communities with an interest in your blog’s topics.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
*This article was updated 10/17/2022.
Tourette syndrome: Diagnosis is key for best care
Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), obsessive-compulsive disorder (OCD), and autism spectrum disorder (ASD) share significant overlap in symptomatology, and it can be challenging at times to distinguish between these conditions. Being able to do so, however, can help guide more targeted interventions and accommodations to optimize a patient’s level of functioning.
Case example
A healthy, bright 6-year-old boy is referred by his family doctor to an academic medical center for a full team evaluation because of suspicion of ASD, after having already been diagnosed with ADHD at the age of 5. His difficulties with inattention, impulsivity, and hyperactivity, as well as his behavioral rigidities and sensory avoidant and sensory seeking behaviors have caused functional impairments for him in his kindergarten classroom. He has been penalized with removal of recess on more than one occasion. A low dose of a stimulant had been tried but resulted in a perceived increase in disruptive behaviors.
The boy, while hyperkinetic and often paying poor attention, is quite capable of high-quality and well-modulated eye contact paired with typical social referencing and reciprocity when actively engaging with the examiner and his parents. He does have a reported history of serial fixated interests and some repetitive behaviors but is also noted to be flexible in his interpersonal style, maintains other varied and typical interests, easily directs affect, utilizes a wide array of fluid gestures paired naturally with verbal communication, and shares enjoyment with smoothly coordinated gaze. He has mild articulation errors but uses pronouns appropriately and has no scripted speech or echolalia, though does engage in some whispered palilalia intermittently.
He is generally quite cooperative and redirectable when focused and has a completely normal physical and neurologic examination. During the visit, the doctor notices the boy making an intermittent honking sound, which parents report as an attention-seeking strategy during times of stress. Further physician-guided information gathering around other repetitive noises and movements elicits a history of engagement in repetitive hand-to-groin movements, some exaggerated blinking, and a number of other waxing and waning subtle motor and phonic tics with onset in preschool. These noises and movements have generally been identified as “fidgeting” and “misbehaving” by well-meaning caregivers in the home and school environments.
Both Tourette syndrome and ASD are more common in males, with stereotyped patterns of movements and behaviors; anxious, obsessive, and compulsive behaviors resulting in behavioral rigidities; sensory sensitivities; and increased rates of hyperkinesis with decreased impulse control which result in increased sensory-seeking behaviors. Diagnostic criteria for Tourette syndrome are met when a child has had multiple motor tics and at least one phonic tic present for at least 1 year, with tic-free intervals lasting no longer than 3 months, and with onset before the age of 18. Typically, tics emerge in late preschool and early grade school, and some children even develop repetitive movements as early as toddlerhood. Tics tend to worsen around the peripubertal era, then often generally improve in the teen years. Tic types, frequency, and severity general fluctuate over time.
Forty percent of children with Tourette syndrome also meet criteria for OCD, with many more having OCD traits, and about 65% of children with Tourette syndrome also meet criteria for ADHD, with many more having ADHD traits. OCD can lead to more rigid and directive social interactions in children as well as obsessive interests, just as ADHD can lead to less socially attuned and less cooperative behaviors, even in children who do not meet criteria for ASD.
For example, a child with OCD in the absence of ASD may still “police” other kids in class and be overly focused on the rules of a game, which may become a social liability. Likewise, a child with ADHD in the absence of ASD may be so distractible that focusing on what other kids are saying and their paired facial expressions is compromised, leading to poor-quality social reciprocity during interactions with peers. Given the remarkable overlap in shared symptoms, it is essential for pediatric providers to consider Tourette syndrome in the differential for any child with repetitive movements and behaviors in addition to ASD and a wide array of other neurodevelopment differences, including global developmental delays and intellectual disabilities. This is of particular importance as the diagnosis of Tourette syndrome can be used to gain access to developmental disability services if the condition has resulted in true adaptive impairments.
It is determined that the boy does in fact meet criteria for ADHD, but also for OCD and Tourette syndrome. Both his Autism Diagnostic Observation Schedule and DSM-5–influenced autism interview are found to be in the nonclinical ranges, given his quality of communication, social engagement, imaginative play, and varied interests. A diagnosis of ASD is not felt to be an appropriate conceptualization of his neurodevelopmental differences. He is started on a low dose of guanfacine, which induces a decline in tics, impulsivity, and hyperkinesis. He is given a 504 plan in school that includes scheduled “tic breaks,” sensory fidgets for use in the classroom, extra movement opportunities as needed, and utilization of a gentle cueing system between him and his teacher for low-key redirection of disruptive behaviors. He is no longer penalized for inattention or tics, and his 504 plan protects him from the use of recess removal as a behavioral modification strategy.
His parents enroll him in the community swim program for extra exercise, focus on decreasing screen time, and give him an earlier bedtime to help decrease his tics and rigidities, while improving his ability to self-regulate. Eventually, a low dose of a newer-generation stimulant is added to his guanfacine, with excellent results and only a mild increase in tolerable tics.
The child in the vignette did well with a 504 plan based on his medical diagnoses, though if related learning difficulties had persisted, eligibility under Other Health Impaired could be used to provide eligibility for an Individualized Education Plan. Alpha-agonists can be helpful for symptom control in those with Tourette syndrome by simultaneously treating tics, hyperkinesis, and impulsivity, while decreasing the risk of tic exacerbation with use of stimulants. Overall, understanding the neurodiversity related to Tourette syndrome can help providers advocate for home and community-based supports to optimize general functioning and quality of life.
Dr. Roth is a developmental and behavioral pediatrician in Eugene, Ore. She has no conflicts of interest.
References
Darrow S et al. J Am Acad Child Adolescent Psych. 2017;56(7):610-7.
AAP Section on Developmental and Behavioral Pediatrics. Developmental and Behavioral Pediatrics. Voigt RG et al, eds. 2018: American Academy of Pediatrics.
Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), obsessive-compulsive disorder (OCD), and autism spectrum disorder (ASD) share significant overlap in symptomatology, and it can be challenging at times to distinguish between these conditions. Being able to do so, however, can help guide more targeted interventions and accommodations to optimize a patient’s level of functioning.
Case example
A healthy, bright 6-year-old boy is referred by his family doctor to an academic medical center for a full team evaluation because of suspicion of ASD, after having already been diagnosed with ADHD at the age of 5. His difficulties with inattention, impulsivity, and hyperactivity, as well as his behavioral rigidities and sensory avoidant and sensory seeking behaviors have caused functional impairments for him in his kindergarten classroom. He has been penalized with removal of recess on more than one occasion. A low dose of a stimulant had been tried but resulted in a perceived increase in disruptive behaviors.
The boy, while hyperkinetic and often paying poor attention, is quite capable of high-quality and well-modulated eye contact paired with typical social referencing and reciprocity when actively engaging with the examiner and his parents. He does have a reported history of serial fixated interests and some repetitive behaviors but is also noted to be flexible in his interpersonal style, maintains other varied and typical interests, easily directs affect, utilizes a wide array of fluid gestures paired naturally with verbal communication, and shares enjoyment with smoothly coordinated gaze. He has mild articulation errors but uses pronouns appropriately and has no scripted speech or echolalia, though does engage in some whispered palilalia intermittently.
He is generally quite cooperative and redirectable when focused and has a completely normal physical and neurologic examination. During the visit, the doctor notices the boy making an intermittent honking sound, which parents report as an attention-seeking strategy during times of stress. Further physician-guided information gathering around other repetitive noises and movements elicits a history of engagement in repetitive hand-to-groin movements, some exaggerated blinking, and a number of other waxing and waning subtle motor and phonic tics with onset in preschool. These noises and movements have generally been identified as “fidgeting” and “misbehaving” by well-meaning caregivers in the home and school environments.
Both Tourette syndrome and ASD are more common in males, with stereotyped patterns of movements and behaviors; anxious, obsessive, and compulsive behaviors resulting in behavioral rigidities; sensory sensitivities; and increased rates of hyperkinesis with decreased impulse control which result in increased sensory-seeking behaviors. Diagnostic criteria for Tourette syndrome are met when a child has had multiple motor tics and at least one phonic tic present for at least 1 year, with tic-free intervals lasting no longer than 3 months, and with onset before the age of 18. Typically, tics emerge in late preschool and early grade school, and some children even develop repetitive movements as early as toddlerhood. Tics tend to worsen around the peripubertal era, then often generally improve in the teen years. Tic types, frequency, and severity general fluctuate over time.
Forty percent of children with Tourette syndrome also meet criteria for OCD, with many more having OCD traits, and about 65% of children with Tourette syndrome also meet criteria for ADHD, with many more having ADHD traits. OCD can lead to more rigid and directive social interactions in children as well as obsessive interests, just as ADHD can lead to less socially attuned and less cooperative behaviors, even in children who do not meet criteria for ASD.
For example, a child with OCD in the absence of ASD may still “police” other kids in class and be overly focused on the rules of a game, which may become a social liability. Likewise, a child with ADHD in the absence of ASD may be so distractible that focusing on what other kids are saying and their paired facial expressions is compromised, leading to poor-quality social reciprocity during interactions with peers. Given the remarkable overlap in shared symptoms, it is essential for pediatric providers to consider Tourette syndrome in the differential for any child with repetitive movements and behaviors in addition to ASD and a wide array of other neurodevelopment differences, including global developmental delays and intellectual disabilities. This is of particular importance as the diagnosis of Tourette syndrome can be used to gain access to developmental disability services if the condition has resulted in true adaptive impairments.
It is determined that the boy does in fact meet criteria for ADHD, but also for OCD and Tourette syndrome. Both his Autism Diagnostic Observation Schedule and DSM-5–influenced autism interview are found to be in the nonclinical ranges, given his quality of communication, social engagement, imaginative play, and varied interests. A diagnosis of ASD is not felt to be an appropriate conceptualization of his neurodevelopmental differences. He is started on a low dose of guanfacine, which induces a decline in tics, impulsivity, and hyperkinesis. He is given a 504 plan in school that includes scheduled “tic breaks,” sensory fidgets for use in the classroom, extra movement opportunities as needed, and utilization of a gentle cueing system between him and his teacher for low-key redirection of disruptive behaviors. He is no longer penalized for inattention or tics, and his 504 plan protects him from the use of recess removal as a behavioral modification strategy.
His parents enroll him in the community swim program for extra exercise, focus on decreasing screen time, and give him an earlier bedtime to help decrease his tics and rigidities, while improving his ability to self-regulate. Eventually, a low dose of a newer-generation stimulant is added to his guanfacine, with excellent results and only a mild increase in tolerable tics.
The child in the vignette did well with a 504 plan based on his medical diagnoses, though if related learning difficulties had persisted, eligibility under Other Health Impaired could be used to provide eligibility for an Individualized Education Plan. Alpha-agonists can be helpful for symptom control in those with Tourette syndrome by simultaneously treating tics, hyperkinesis, and impulsivity, while decreasing the risk of tic exacerbation with use of stimulants. Overall, understanding the neurodiversity related to Tourette syndrome can help providers advocate for home and community-based supports to optimize general functioning and quality of life.
Dr. Roth is a developmental and behavioral pediatrician in Eugene, Ore. She has no conflicts of interest.
References
Darrow S et al. J Am Acad Child Adolescent Psych. 2017;56(7):610-7.
AAP Section on Developmental and Behavioral Pediatrics. Developmental and Behavioral Pediatrics. Voigt RG et al, eds. 2018: American Academy of Pediatrics.
Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), obsessive-compulsive disorder (OCD), and autism spectrum disorder (ASD) share significant overlap in symptomatology, and it can be challenging at times to distinguish between these conditions. Being able to do so, however, can help guide more targeted interventions and accommodations to optimize a patient’s level of functioning.
Case example
A healthy, bright 6-year-old boy is referred by his family doctor to an academic medical center for a full team evaluation because of suspicion of ASD, after having already been diagnosed with ADHD at the age of 5. His difficulties with inattention, impulsivity, and hyperactivity, as well as his behavioral rigidities and sensory avoidant and sensory seeking behaviors have caused functional impairments for him in his kindergarten classroom. He has been penalized with removal of recess on more than one occasion. A low dose of a stimulant had been tried but resulted in a perceived increase in disruptive behaviors.
The boy, while hyperkinetic and often paying poor attention, is quite capable of high-quality and well-modulated eye contact paired with typical social referencing and reciprocity when actively engaging with the examiner and his parents. He does have a reported history of serial fixated interests and some repetitive behaviors but is also noted to be flexible in his interpersonal style, maintains other varied and typical interests, easily directs affect, utilizes a wide array of fluid gestures paired naturally with verbal communication, and shares enjoyment with smoothly coordinated gaze. He has mild articulation errors but uses pronouns appropriately and has no scripted speech or echolalia, though does engage in some whispered palilalia intermittently.
He is generally quite cooperative and redirectable when focused and has a completely normal physical and neurologic examination. During the visit, the doctor notices the boy making an intermittent honking sound, which parents report as an attention-seeking strategy during times of stress. Further physician-guided information gathering around other repetitive noises and movements elicits a history of engagement in repetitive hand-to-groin movements, some exaggerated blinking, and a number of other waxing and waning subtle motor and phonic tics with onset in preschool. These noises and movements have generally been identified as “fidgeting” and “misbehaving” by well-meaning caregivers in the home and school environments.
Both Tourette syndrome and ASD are more common in males, with stereotyped patterns of movements and behaviors; anxious, obsessive, and compulsive behaviors resulting in behavioral rigidities; sensory sensitivities; and increased rates of hyperkinesis with decreased impulse control which result in increased sensory-seeking behaviors. Diagnostic criteria for Tourette syndrome are met when a child has had multiple motor tics and at least one phonic tic present for at least 1 year, with tic-free intervals lasting no longer than 3 months, and with onset before the age of 18. Typically, tics emerge in late preschool and early grade school, and some children even develop repetitive movements as early as toddlerhood. Tics tend to worsen around the peripubertal era, then often generally improve in the teen years. Tic types, frequency, and severity general fluctuate over time.
Forty percent of children with Tourette syndrome also meet criteria for OCD, with many more having OCD traits, and about 65% of children with Tourette syndrome also meet criteria for ADHD, with many more having ADHD traits. OCD can lead to more rigid and directive social interactions in children as well as obsessive interests, just as ADHD can lead to less socially attuned and less cooperative behaviors, even in children who do not meet criteria for ASD.
For example, a child with OCD in the absence of ASD may still “police” other kids in class and be overly focused on the rules of a game, which may become a social liability. Likewise, a child with ADHD in the absence of ASD may be so distractible that focusing on what other kids are saying and their paired facial expressions is compromised, leading to poor-quality social reciprocity during interactions with peers. Given the remarkable overlap in shared symptoms, it is essential for pediatric providers to consider Tourette syndrome in the differential for any child with repetitive movements and behaviors in addition to ASD and a wide array of other neurodevelopment differences, including global developmental delays and intellectual disabilities. This is of particular importance as the diagnosis of Tourette syndrome can be used to gain access to developmental disability services if the condition has resulted in true adaptive impairments.
It is determined that the boy does in fact meet criteria for ADHD, but also for OCD and Tourette syndrome. Both his Autism Diagnostic Observation Schedule and DSM-5–influenced autism interview are found to be in the nonclinical ranges, given his quality of communication, social engagement, imaginative play, and varied interests. A diagnosis of ASD is not felt to be an appropriate conceptualization of his neurodevelopmental differences. He is started on a low dose of guanfacine, which induces a decline in tics, impulsivity, and hyperkinesis. He is given a 504 plan in school that includes scheduled “tic breaks,” sensory fidgets for use in the classroom, extra movement opportunities as needed, and utilization of a gentle cueing system between him and his teacher for low-key redirection of disruptive behaviors. He is no longer penalized for inattention or tics, and his 504 plan protects him from the use of recess removal as a behavioral modification strategy.
His parents enroll him in the community swim program for extra exercise, focus on decreasing screen time, and give him an earlier bedtime to help decrease his tics and rigidities, while improving his ability to self-regulate. Eventually, a low dose of a newer-generation stimulant is added to his guanfacine, with excellent results and only a mild increase in tolerable tics.
The child in the vignette did well with a 504 plan based on his medical diagnoses, though if related learning difficulties had persisted, eligibility under Other Health Impaired could be used to provide eligibility for an Individualized Education Plan. Alpha-agonists can be helpful for symptom control in those with Tourette syndrome by simultaneously treating tics, hyperkinesis, and impulsivity, while decreasing the risk of tic exacerbation with use of stimulants. Overall, understanding the neurodiversity related to Tourette syndrome can help providers advocate for home and community-based supports to optimize general functioning and quality of life.
Dr. Roth is a developmental and behavioral pediatrician in Eugene, Ore. She has no conflicts of interest.
References
Darrow S et al. J Am Acad Child Adolescent Psych. 2017;56(7):610-7.
AAP Section on Developmental and Behavioral Pediatrics. Developmental and Behavioral Pediatrics. Voigt RG et al, eds. 2018: American Academy of Pediatrics.
The WPATH guidelines for treatment of adolescents with gender dysphoria have changed
The World Professional Association for Transgender Health (WPATH) is an interdisciplinary professional and educational organization devoted to transgender health. One of their activities is to produce the Standards of Care (SOC) for treatment of individuals with gender dysphoria. According to WPATH, the SOC “articulate a professional consensus about the psychiatric, psychological, medical, and surgical management of gender dysphoria and help professionals understand the parameters within which they may offer assistance to those with these conditions.” Many clinicians around the world use these guidelines to help them care for patients with gender dysphoria and diverse gender expressions.
The most recent SOC, version 8, were released on Sept. 15, 2022, after a 2-year postponement because of the pandemic. These new standards represent the first update to the SOC since version 7, which was released in 2012. Given how recent this update is, this column will attempt to summarize the changes in the new guidelines that affect children and adolescents.
One of the major differences between SOC versions 7 and 8 is that version 8 now includes a chapter specifically dedicated to the care of adolescents. Version 7 lumped children and adolescents together into one chapter. This is an important distinction for SOC 8, as it highlights that care for prepubertal youth is simply social in nature and distinct from that of pubertal adolescents. Social transition includes things such as using an affirmed name/pronouns and changing hair style and clothes. It does not include medications of any kind. Allowing these youth the time and space to explore the natural gender diversity of childhood leads to improved psychological outcomes over time and reduces adversity. Psychological support, where indicated, should be offered to gender-diverse children and their families to explore the persistence, consistence, and insistence of that child’s gender identity.
Once a child reaches puberty, medications may come into play as part of an adolescent’s transition. SOC 7 had established a minimum age of 16 before any partially reversible medications (testosterone, estrogen) were started as part of a patient’s medical transition. Starting with SOC 8, a minimum age has been removed for the initiation of gender-affirming hormone therapy. However, a patient must still have begun their natal puberty before any medication is started. A specific age was removed to acknowledge that maturity in adolescents occurs on a continuum and at different ages. SOC 8 guidelines continue to recommend that the individual’s emotional, cognitive, and psychosocial development be taken into account when determining their ability to provide consent for treatment. These individuals should still undergo a comprehensive assessment, as described below.
Similar to SOC 7, SOC 8 continues to stress the importance of a comprehensive, multidisciplinary evaluation of those adolescents who seek medical therapy as part of their transition. This allows for the exploration of additional coexisting causes of gender dysphoria, such as anxiety, depression, or other mental health conditions. If these exist, then they must be appropriately treated before any gender-affirming medical treatment is initiated. Assessments should be performed by clinicians who have training and expertise with the developmental trajectory of adolescents, as well as with common mental health conditions. These assessments are also critical, as SOC 8 acknowledges a rise in the number of adolescents who may not have had gender-diverse expression in childhood.
SOC 8 and the Endocrine Society Guidelines (see references) provide physicians and other health care professionals with a road map for addressing the needs of transgender and gender-diverse persons. By referencing these guidelines when taking care of these patients, physicians and other health care professionals will know that they are providing the most up-to-date, evidence-based care.
Dr. M. Brett Cooper is an assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
SOC 8: https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
SOC 7: https://www.wpath.org/media/cms/Documents/SOC%20v7/SOC%20V7_English2012.pdf?_t=1613669341
Endocrine Society Gender Affirming Care Guidelines: https://academic.oup.com/jcem/article/102/11/3869/4157558?login=false
The World Professional Association for Transgender Health (WPATH) is an interdisciplinary professional and educational organization devoted to transgender health. One of their activities is to produce the Standards of Care (SOC) for treatment of individuals with gender dysphoria. According to WPATH, the SOC “articulate a professional consensus about the psychiatric, psychological, medical, and surgical management of gender dysphoria and help professionals understand the parameters within which they may offer assistance to those with these conditions.” Many clinicians around the world use these guidelines to help them care for patients with gender dysphoria and diverse gender expressions.
The most recent SOC, version 8, were released on Sept. 15, 2022, after a 2-year postponement because of the pandemic. These new standards represent the first update to the SOC since version 7, which was released in 2012. Given how recent this update is, this column will attempt to summarize the changes in the new guidelines that affect children and adolescents.
One of the major differences between SOC versions 7 and 8 is that version 8 now includes a chapter specifically dedicated to the care of adolescents. Version 7 lumped children and adolescents together into one chapter. This is an important distinction for SOC 8, as it highlights that care for prepubertal youth is simply social in nature and distinct from that of pubertal adolescents. Social transition includes things such as using an affirmed name/pronouns and changing hair style and clothes. It does not include medications of any kind. Allowing these youth the time and space to explore the natural gender diversity of childhood leads to improved psychological outcomes over time and reduces adversity. Psychological support, where indicated, should be offered to gender-diverse children and their families to explore the persistence, consistence, and insistence of that child’s gender identity.
Once a child reaches puberty, medications may come into play as part of an adolescent’s transition. SOC 7 had established a minimum age of 16 before any partially reversible medications (testosterone, estrogen) were started as part of a patient’s medical transition. Starting with SOC 8, a minimum age has been removed for the initiation of gender-affirming hormone therapy. However, a patient must still have begun their natal puberty before any medication is started. A specific age was removed to acknowledge that maturity in adolescents occurs on a continuum and at different ages. SOC 8 guidelines continue to recommend that the individual’s emotional, cognitive, and psychosocial development be taken into account when determining their ability to provide consent for treatment. These individuals should still undergo a comprehensive assessment, as described below.
Similar to SOC 7, SOC 8 continues to stress the importance of a comprehensive, multidisciplinary evaluation of those adolescents who seek medical therapy as part of their transition. This allows for the exploration of additional coexisting causes of gender dysphoria, such as anxiety, depression, or other mental health conditions. If these exist, then they must be appropriately treated before any gender-affirming medical treatment is initiated. Assessments should be performed by clinicians who have training and expertise with the developmental trajectory of adolescents, as well as with common mental health conditions. These assessments are also critical, as SOC 8 acknowledges a rise in the number of adolescents who may not have had gender-diverse expression in childhood.
SOC 8 and the Endocrine Society Guidelines (see references) provide physicians and other health care professionals with a road map for addressing the needs of transgender and gender-diverse persons. By referencing these guidelines when taking care of these patients, physicians and other health care professionals will know that they are providing the most up-to-date, evidence-based care.
Dr. M. Brett Cooper is an assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
SOC 8: https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
SOC 7: https://www.wpath.org/media/cms/Documents/SOC%20v7/SOC%20V7_English2012.pdf?_t=1613669341
Endocrine Society Gender Affirming Care Guidelines: https://academic.oup.com/jcem/article/102/11/3869/4157558?login=false
The World Professional Association for Transgender Health (WPATH) is an interdisciplinary professional and educational organization devoted to transgender health. One of their activities is to produce the Standards of Care (SOC) for treatment of individuals with gender dysphoria. According to WPATH, the SOC “articulate a professional consensus about the psychiatric, psychological, medical, and surgical management of gender dysphoria and help professionals understand the parameters within which they may offer assistance to those with these conditions.” Many clinicians around the world use these guidelines to help them care for patients with gender dysphoria and diverse gender expressions.
The most recent SOC, version 8, were released on Sept. 15, 2022, after a 2-year postponement because of the pandemic. These new standards represent the first update to the SOC since version 7, which was released in 2012. Given how recent this update is, this column will attempt to summarize the changes in the new guidelines that affect children and adolescents.
One of the major differences between SOC versions 7 and 8 is that version 8 now includes a chapter specifically dedicated to the care of adolescents. Version 7 lumped children and adolescents together into one chapter. This is an important distinction for SOC 8, as it highlights that care for prepubertal youth is simply social in nature and distinct from that of pubertal adolescents. Social transition includes things such as using an affirmed name/pronouns and changing hair style and clothes. It does not include medications of any kind. Allowing these youth the time and space to explore the natural gender diversity of childhood leads to improved psychological outcomes over time and reduces adversity. Psychological support, where indicated, should be offered to gender-diverse children and their families to explore the persistence, consistence, and insistence of that child’s gender identity.
Once a child reaches puberty, medications may come into play as part of an adolescent’s transition. SOC 7 had established a minimum age of 16 before any partially reversible medications (testosterone, estrogen) were started as part of a patient’s medical transition. Starting with SOC 8, a minimum age has been removed for the initiation of gender-affirming hormone therapy. However, a patient must still have begun their natal puberty before any medication is started. A specific age was removed to acknowledge that maturity in adolescents occurs on a continuum and at different ages. SOC 8 guidelines continue to recommend that the individual’s emotional, cognitive, and psychosocial development be taken into account when determining their ability to provide consent for treatment. These individuals should still undergo a comprehensive assessment, as described below.
Similar to SOC 7, SOC 8 continues to stress the importance of a comprehensive, multidisciplinary evaluation of those adolescents who seek medical therapy as part of their transition. This allows for the exploration of additional coexisting causes of gender dysphoria, such as anxiety, depression, or other mental health conditions. If these exist, then they must be appropriately treated before any gender-affirming medical treatment is initiated. Assessments should be performed by clinicians who have training and expertise with the developmental trajectory of adolescents, as well as with common mental health conditions. These assessments are also critical, as SOC 8 acknowledges a rise in the number of adolescents who may not have had gender-diverse expression in childhood.
SOC 8 and the Endocrine Society Guidelines (see references) provide physicians and other health care professionals with a road map for addressing the needs of transgender and gender-diverse persons. By referencing these guidelines when taking care of these patients, physicians and other health care professionals will know that they are providing the most up-to-date, evidence-based care.
Dr. M. Brett Cooper is an assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
SOC 8: https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
SOC 7: https://www.wpath.org/media/cms/Documents/SOC%20v7/SOC%20V7_English2012.pdf?_t=1613669341
Endocrine Society Gender Affirming Care Guidelines: https://academic.oup.com/jcem/article/102/11/3869/4157558?login=false
The urgent need to diagnose Sanfilippo syndrome at an early age
Sanfilippo syndrome is a rare inherited neurodegenerative metabolic disorder for which there are no approved therapies. Symptoms of the more severe subtypes typically begin within the first years of life, rapidly producing serious and progressive physical and cognitive deficits. The underlying pathophysiology is targetable, but the delay in diagnosis of this as well as other lysosomal storage disorders (LSDs) is slowing progress toward effective therapies.
“Lack of awareness and the delays to diagnosis have been a real challenge for us. There is reason for cautious optimism about treatments now in or approaching clinical studies, but to evaluate efficacy on cognitive outcomes we need to enroll more children at a very young age, before loss of milestones,” according to Cara O’Neill, MD, a co-founder and chief science officer of Cure Sanfilippo Foundation.
Epidemiology and description
Sanfilippo syndrome, like the more than 50 other LSDs, is caused by a gene mutation that leads to an enzyme deficiency in the lysosome.1 In the case of Sanfilippo syndrome, also known as mucopolysaccharidosis (MPS III), there are hundreds of mutations that can lead to Sanfilippo by altering the function of one of the four genes essential to degradation of heparan sulfate.2 Lysosomal accumulation of heparan sulfate drives a broad spectrum of progressive and largely irreversible symptoms that typically begin with somatic manifestations, such as bowel dysfunction and recurrent ear and upper respiratory infections.
Impairment of the central nervous system (CNS) usually occurs early in life, halting physical and mental development. As it progresses, accumulation of heparan sulfate in a variety of cells leads to a cascade of abnormal cellular signaling and dysfunction. Disruption of these processes, which are critical for normal neurodevelopment, result in loss of the developmental skills already gained and eventually loss of brain tissue.3 Although life expectancy has improved with supportive care, survival into adulthood is typically limited to milder forms.4
Over the past several years, progress in this and other LSDs has yielded therapeutic targets, including those involving gene repair and enzyme replacement. Already approved for use in some LSDs, these therapies have also shown promise in the experimental setting for Sanfilippo syndrome, leading to several completed clinical trials.5
So far, none of these treatments has advanced beyond clinical trials in Sanfilippo syndrome, but there have been favorable changes in the markers of disease, suggesting that better methods of treatment delivery and/or more sensitive tools to measure clinical change might lead to evidence of disease attenuation. However, the promise of treatment in all cases has been to prevent, slow, or halt progression, not to reverse it. This point is important, because it indicates that degree of benefit will depend on enrolling patients early in life. Even if effective therapies are identified, few patients will benefit without strategies to accelerate diagnosis.
In fact, “one study6 reported that the average age of diagnosis for Sanfilippo syndrome has not improved over the past 30 years,” according to Dr. O’Neill. She indicated that this has been frustrating, given the availability of clinical trials on which progress is dependent. There is no widely accepted protocol for who and when to test for Sanfilippo syndrome or other LSDs, but Dr. O’Neill’s organization is among those advocating for strategies to detect these diseases earlier, including screening at birth.
Almost by definition, the clinical diagnosis of rare diseases poses a challenge. With nonspecific symptoms and a broad range of potential diagnoses, diseases with a low incidence are not the first ones that are typically considered. In the case of Sanfilippo syndrome, published studies indicate incidence rates at or below 1 per 70,000 live births.7 However, the incidence rates have been highly variable not only by geographical regions but even across neighboring countries where genetic risk would be expected to be similar.
In Europe, for example, epidemiologic studies suggest the lifetime risk of MPS IIIA is approximately two times greater in Germany and the Netherlands relative to France and Sweden.7 It is possible that the methodology for identifying cases might be a more important factor than differences in genetic risk to explain this variability. Many experts, including Dr. O’Neill, believe that prevalence figures for Sanfilippo syndrome are typically underestimates because of the frequency with which LSDs are attributed to other pathology.
“For these types of rare disorders, a clinician might only see a single case over a career, and the symptoms can vary in presentation and severity with many alternatives to consider in the differential diagnosis,” Dr. O’Neill explained. She cited case reports in which symptoms of Sanfilippo syndrome after a period of initial normal development has been initially attributed to autism, which is a comorbid feature of the disease, idiopathic developmental delay, or other nonprogressive disorders until further clinical deterioration leads to additional testing. The implication is that LSDs must be considered far earlier despite their rarity.
For the least common of the four clinical subtypes, MPS IIIC and MPS IIID, the median ages of diagnosis have ranged from 4.5 to 19 years of age.7 This is likely a reflection of a slower progression and a later onset of clinical manifestations.
For the more rapidly progressing and typically more severe subtypes, MPS IIIA and MPS IIIB, the diagnosis is typically made earlier. In one review of epidemiologic studies in different countries, the earliest reported median age at diagnosis was 2.5 years,7 a point at which significant disease progression is likely to have already occurred. If the promise of treatments in development is prevention of disease progression, disability in many patients might be substantial if the time to diagnosis is not reduced.
Screening and testing
Independent of the potential to enroll children in clinical trials, early diagnosis also advances the opportunities for supportive care to lessen the burden of the disease on patients and families. Perhaps even more important, early diagnosis is vital to family planning. Since the American pediatrician Sylvester Sanfilippo, MD, first described this syndrome in 1963,7 the genetic profile and many of the features of the disease have become well characterized.8
“One reason to emphasize the importance of early diagnosis is the heritability of this disorder. With prompt diagnosis, genetic counseling can be offered to families to provide them with critical information for future family planning and for cascade testing of other potentially affected siblings,” Dr. O’Neill reported. The inheritance pattern of Sanfilippo syndrome is autosomal recessive.3 In families with an affected child, the risk for any subsequent child to have the same disorder is 25%. The chance of a sibling to be unaffected and not a carrier is also 25%. There is a 50% chance of a sibling to be a carrier but asymptomatic. Of priorities, spreading awareness has been a critical mission of the Cure Sanfilippo Foundation since it was founded 8 years ago, according to Glenn O’Neill, the president. He and his wife, Dr. O’Neill, who is a pediatrician, founded the organization after their own child’s diagnosis of Sanfilippo syndrome. Creating awareness is fundamental to the mission of attracting funds for research, but support to patients and their families as well as early enrollment in clinical trials are among other initiatives being pursued by the foundation to improve care and prognosis.
These strategies include some novel ideas, including an algorithm based on artificial intelligence (AI) that can accelerate suspicion of Sanfilippo syndrome in advance of laboratory or genetic testing, according to Dr. O’Neill. She reported that the facial phenotype, which is observed in a high proportion of but not in all Sanfilippo patients, includes coarse facial features such as puffiness around the eyes, heavy eyebrows, full lips, and macrocephaly.9 Interpretation of photos for AI-based analysis is enhanced when combined with other clinical symptoms.

“The Foundation was involved in honing such a tool by submitting the photos that were used to teach the AI to recognize the Sanfilippo syndrome phenotype,” Dr. O’Neill said. The AI-based tool (Face2Gene.com) is available from FDNA, a company that has been involved in analyzing complex phenotypic and genomic information to guide diagnosis and therapeutic strategies for an array of diseases, not just Sanfilippo syndrome.
The preferred method for diagnosis is biochemical or genetic testing. Of these, urine testing for elevated levels of heparan sulfate glycosaminoglycans (GAG) can be useful for screening, although false-negative tests occur. Analysis of the blood can be performed to detect abnormal levels or activity of the enzymes that break down this GAG. In addition, genetic testing can be performed on blood, fibroblast, buccal swab, or saliva samples. Genetic testing of the blood is the most frequently performed.
For the four MPS III subtypes – MPS IIIA, IIIB, IIIC, and IIID – the presence of two pathogenic mutations in the SGSH (17q25.3), NAGLU (17q21.2), HGSNAT (8p11.21), and GNS (12q14.3) genes, respectively, are likely diagnostic, but enzymatic testing or GAG analysis should be performed to confirm disease status, according to Dr. O’Neill, who said that global consensus based clinical care guidelines led by the Foundation were recently accepted for publication and also include a section on the approach to diagnosis.
While laboratory testing is sensitive, urinary excretion of GAG can be variable, with the potential for ambiguous results. Typically, biochemical and genetic testing provide more reliable results for the diagnosis. They can be readily performed in utero or at the time of birth. In addition, gene panels can permit the diagnosis of multiple types of LSDs, not just Sanfilippo, making screening a cost-effective strategy to consider multiple diseases with overlapping symptoms when an LSD is suspected. Dr. O’Neill said clinical guidelines recommend confirmation of enzyme deficiency or evidence of GAG substrate accumulation as confirmatory tests when genetic testing is positive.
“Ultimately, our goal is to promote universal screening at birth for these serious genetic disorders affecting children,” Dr. O’Neill said.
“We are in a catch-22 when it comes to newborn screening. Currently our federal system requires there be an available treatment before recommending routine screening for a disease. However, it is extremely difficult to power trials with patients who are most likely to show benefit in a trial setting without that very early diagnosis. Universal newborn screening would pave the way for accelerated drug development for children,” she added.
In the meantime, Dr. O’Neill suggests that clinicians should employ a low threshold of suspicion to pursue diagnostic studies of LSDs in infants and children with developmental delays or otherwise unexplained progressive disorders.
Importantly, clinicians can now act quickly on their suspicions and order testing without concern for delays or denial by insurers through a special program, according to Dr. O’Neill. Free genetic testing, offered by the Invitae Corporation, evaluates a panel of 58 genes associated with lysosomal disorders, permitting detection of Sanfilippo syndrome and other LSDs, according to Dr. O’Neill. The Invitae testing is typically performed on 3 mL of whole blood delivered to a central testing facility.
“Results can be obtained within a few weeks or sooner. This can seem like a long wait for families, but it is much more efficient than ordering tests sequentially,” Dr. O’Neill said.
Diagnosis: Signs and symptoms
Despite the differences in progression of the MPS III subtypes, the clinical characteristics are more similar than different. In all patients, prenatal and infant development are typically normal. The initial signs of disease can be found in the newborn, such as neonatal tachypnea, through the early infancy period, such as macrocephaly. However, these are not commonly recognized until about age 1 or soon after in those with MPS IIIA and IIIB.3 Speech delay is the first developmental delay seen in most patients. In those with MPS IIIC, initial symptoms are typically detected at age 3 or later and progress more slowly.10,11 The same is likely to be true of MPS IIID, although this subtype is less well characterized than the other three.7
Although many organs can be involved, degeneration of the CNS is regarded as the most characteristic.3 In aggressive disease, this includes slower acquisition of and failure to meet developmental milestones with progressive intellectual disability, while behavioral difficulties are a more common initial compliant in children with milder disease.13,14 These behavioral changes include hyperactivity, inattention, autistic behaviors, worsening safety awareness, and in some cases aggressive behavior that can be destructive. Sleep disturbances are common.15Because of variability inherent in descriptions of relatively small numbers of patients, the characterization of each of the MPS III subgroups is based on a limited number of small studies, but most patients demonstrate behavior disorders, have coarse facial features, and develop speech delay, according to a survey conducted of published studies.7 Collectively, abnormal behavior was identified as an early symptom in 77% of those with MPS IIIA, 69% of those with IIIB, and 77% of those with IIIC.
For MPS IIIA, loss of speech was observed at a median age of 3.8 years and loss of walking ability at 10.4 years. The median survival has been reported to range between 13 and 18 years. In children with MPS IIIB, the median age of speech loss was reported to about the same age, while loss of walking ability occurred at 11 years. In one study of MPS IIIB, 24% of patients had developed dementia by age 6 years, and the reported median survival has ranged between 17 and 19 years. For MPS IIIC, the onset of clinical symptoms has been observed at a median age of 3.5 years with evidence of cognitive loss observed in 33% of children by the age of 6 years. The median survival has ranged from 19 to 34 years in three studies tracing the natural history of this MPS III subtype.
The differential diagnosis reasonably includes other types of mucopolysaccharidosis disorders with cognitive impairment, including Hurler, Hunter, or Sly syndromes, other neurodevelopmental disorders, and inborn errors of metabolism. The heterogeneity of the features makes definitive laboratory or genetic testing, rather than the effort to differentiate clinical features, appropriate for a definitive diagnosis.
Once the diagnosis is made, other examinations for the common complications of Sanfilippo syndrome are appropriate. Abdominal imaging is appropriate for detecting complications in the gastrointestinal tract, including hepatomegaly, which has been reported in more than half of patients with MPS IIIA and IIIB and in 39% of patients with IIIC.7 In patients with breathing concerns at night and/or sleep disturbance, polysomnography can be useful for identifying sleep apnea and nocturnal seizure activity. In children suspected of seizures, EEG is appropriate. In one study, 66% of patients with MPS IIIA developed seizure activity.16 This has been less commonly reported in MPS IIIB and IIIC, ranging from 8% to 13%.15
Formal hearing evaluation is indicated for any child with speech delays. Hearing loss typically develops after the newborn period in Sanfilippo and may affect peak language acquisition if not treated, according to Dr. O’Neill.
Radiographic studies for dysostosis multiplex or other skeletal abnormalities are also appropriate based on clinical presentation.
Treatment: Present and future
In the absence of treatments to improve the prognosis of Sanfilippo syndrome, current management is based on supportive care and managing organ-specific complications. However, several strategies have proven viable in experimental models and led to clinical trials. None of these therapies has reached approval yet, but several have been associated with attenuation of biomarkers of MPS III disease activity.
Of nearly 30 Sanfilippo clinical trials conducted over the past 20 years, at least 9 have now been completed.5 In addition to studying gene therapy and enzyme replacement therapy, these trials have included stem cell transplantation and substrate reduction therapy, for which the goal is to reduce synthesis of the heparan sulfate GAG to prevent accumulation.5 Of this latter approach, promising initial results with genistein, an isoflavone that breaks down heparan sulfate, reached a phase 3 evaluation.18 Although heparan sulfate levels in the CNS were non-significantly reduced over the course of the trial, the reduction was not sufficient to attenuate cognitive decline.
In other LSDs, several forms of enzyme replacement therapy are now approved. In Fabry disease, for example, recombinant alpha-galactosidase A has now been used for more than 15 years.19 Clinical benefit has not yet been demonstrated in patients with Sanfilippo syndrome because of the difficulty of delivering these therapies past the blood-brain barrier. Several strategies have been pursued. For example, intrathecal delivery of recombinant heparan-N-sulfatase reduced CNS levels of GAG heparan sulfate in one phase 2B study, but it approached but fell short of the statistical significance for the primary endpoint of predefined cognitive stabilization.20 The signal of activity and generally acceptable tolerability has encouraged further study, including an ongoing study with promising interim results of intracerebroventricular enzyme replacement in MPS IIIB, according to Dr. O’Neill.
Acceptable safety and promising activity on disease biomarkers have also been seen with gene therapy in clinical trials. In one study that showed attenuation of brain atrophy, there was moderate improvement in behavior and sleep in three of the four patients enrolled.21 Other studies using various strategies for gene delivery have also produced signals of activity against the underlying pathology, generating persistent interest in ongoing and planned clinical studies with this form of treatment.22Unmodified hematopoietic stem cell transplantation (HSCT), an approach that has demonstrated efficacy when delivered early in the course of other LSDs, such as Hurler syndrome,23 has not yet been associated with significant activity in clinical studies of MPS III, including those that initiated treatment prior to the onset of neurological symptoms.24 However, promising early results have been reported in a study of gene-modified HSCT, which overexpresses the MPS IIIA enzyme.
“The clinical trial landscape fluctuates quite a bit, so I always encourage clinicians and families to check back often for updates. Patient organizations can also be helpful for understanding the most up-to-date and emerging trial options,” Dr. O’Neill reported.
Although it is expected that the greatest benefit would be derived from treatments initiated before or very early after the onset of symptoms, based on the limited potential for reversing cognitive loss, Dr. O’Neill said that she and others are also striving to offer treatments for individuals now living with Sanfilippo syndrome.
“We have to be willing to test treatments that are symptomatic in nature. To that aim, the Cure Sanfilippo Foundation has sponsored a study of a CNS-penetrating anti-inflammatory agent in advanced-disease patients more than 4 years of age,” Dr. O’Neill said. This group of patients typically been ineligible for clinical trials in the past. Dr. O’Neill hopes to change this orientation.
“It is important to highlight that all patients deserve our efforts to improve their quality of life and alleviate suffering, regardless of how old they are or how progressed in the disease they happen to be,” she said.
However, whether the goal is enrollment before or early in disease or later in disease progression, the challenge of enrolling sufficient numbers of patients to confirm clinical activity has been and continues to be a hurdle to progress.
“Clinical studies in Sanfilippo enroll relatively small numbers of patients, often 20 or less,” said Dr. O’Neill, explaining one of the reasons why her organization has been so active in raising awareness and funding such studies. For patients and families, the Cure Sanfilippo Foundation can offer a variety of guidance and support, but information about opportunities for clinical trial participation is a key resource they provide for families and their physicians.
Conclusion
For most children with Sanfilippo syndrome, life expectancy is limited. However, the characterization of the genetic causes and the biochemistry of the subtypes has led to several viable therapeutic approaches under development. There has been progress in delivery of therapeutic enzymes to the CNS, and there is substantial optimism that more progress is coming. One issue for treatment development, is the last of a clear regulatory pathway addressing important biomarkers of pathology, such as heparan sulfate burden. Developing treatments that address this issue or impaired enzyme activity levels have promise for preventing progression, particularly if started in infancy. However, the effort to draw awareness to this disease is the first step toward accelerating the time to an early diagnosis and subsequent opportunities to enroll in clinical trials.
References
1. Sun A. Lysosomal storage disease overview. Ann Transl Med. 2018 Dec;6(24):476. doi: 10.21037/atm.2018.11.39.
2. Andrade F et al. Sanfilippo syndrome: Overall review. Pediatr Int. 2015 Jun;57(3):331-8. doi: 10.1111/ped.12636.
3. Fedele AO. Sanfilippo syndrome: Causes, consequences, and treatments. Appl Clin Genet. 2015 Nov 25;8:269-81. doi: 10.2147/TACG.S57672.
4. Lavery C et al. Mortality in patients with Sanfilippo syndrome. Orphanet J Rare Dis. 2017 Oct 23;12(1):168. doi: 10.1186/s13023-017-0717-y.
5. Pearse Y et al. A cure for Sanfilippo syndrome? A summary of current therapeutic approaches and their promise. Med Res Arch. 2020 Feb 1;8(2). doi: 10.18103/mra.v8i2.2045.
6. Kuiper GA et al. Failure to shorten the diagnostic delay in two ultrao-rphan diseases (mucopolysaccharidosis types I and III): potential causes and implication. Orphanet J Rare Dis. 2018;13:2. Doi: 10.1186/s13023-017-0733-y.
7. Zelei T et al. Epidemiology of Sanfilippo syndrome: Results of a systematic literature review. Orphanet J Rare Dis. 2018 Apr 10;13(1):53. doi: 10.1186/s13023-018-0796-4.
8. Wagner VF, Northrup H. Mucopolysaccaharidosis type III. Gene Reviews. 2019 Sep 19. University of Washington, Seattle. https://www.ncbi.nlm.nih.gov/books/NBK546574/8.
9. O’Neill C et al. Natural history of facial features observed in Sanfilippo syndrome (MPS IIIB) using a next generation phenotyping tool. Mol Genet Metab. 2019 Feb;126:S112.
10. Ruijter GJ et al. Clinical and genetic spectrum of Sanfilippo type C (MPS IIIC) disease in the Netherlands. Mol Genet Metab. 2008 Feb;93(2):104-11. doi: 10.1016/j.ymgme.2007.09.011.
11. Valstar MJ et al. Mucopolysaccharidosis type IIID: 12 new patients and 15 novel mutations. Hum Mutat. 2010 May;31(5):E1348-60. doi: 10.1002/humu.21234.
12. Nijmeijer SCM. The attenuated end of phenotypic spectrum in MPS III: from late-onset stable cognitive impairment to non-neuronopathic phenotype. Orphanet J Rare Dis. 2019;14:249. Doi10.1186/s13023-019-1232-0.
13. Nidiffer FD, Kelly TE. Developmental and degenerative patterns associated with cognitive, behavioural and motor difficulties in the Sanfilippo syndrome: An epidemiological study. J Ment Defic Res. 1983 Sep;27 (Pt 3):185-203. doi: 10.1111/j.1365-2788.1983.tb00291.x.
14. Bax MC, Colville GA. Behaviour in mucopolysaccharide disorders. Arch Dis Child. 1995 Jul;73(1):77-81. doi: 10.1136/adc.73.1.77.
15. Fraser J et al. Sleep disturbance in mucopolysaccharidosis type III (Sanfilippo syndrome): A survey of managing clinicians. Clin Genet. 2002 Nov;62(5):418-21. doi: 10.1034/j.1399-0004.2002.620512.x.
16. Valstar MJ et al. Mucopolysaccharidosis type IIIA: Clinical spectrum and genotype-phenotype correlations. Ann Neurol. 2010 Dec;68(6):876-87. doi: 10.1002/ana.22092.
17. Heron B et al. Incidence and natural history of mucopolysaccharidosis type III in France and comparison with United Kingdom and Greece. Am J Med Genet A. 2011 Jan;155A(1):58-68. doi: 10.1002/ajmg.a.33779.
18. Delgadillo V et al. Genistein supplementation in patients affected by Sanfilippo disease. J Inherit Metab Dis. 2011 Oct;34(5):1039-44. doi: 10.1007/s10545-011-9342-4.
19. van der Veen SJ et al. Developments in the treatment of Fabry disease. J Inherit Metab Dis. 2020 Sep;43(5):908-21. doi: 10.1002/jimd.12228.
20. Wijburg FA et al. Intrathecal heparan-N-sulfatase in patients with Sanfilippo syndrome type A: A phase IIb randomized trial. Mol Genet Metab. 2019 Feb;126(2):121-30. doi: 10.1016/j.ymgme.2018.10.006.
21. Tardieu M et al. Intracerebral administration of adeno-associated viral vector serotype rh.10 carrying human SGSH and SUMF1 cDNAs in children with mucopolysaccharidosis type IIIA disease: Results of a phase I/II trial. Hum Gene Ther. 2014 Jun;25(6):506-16. doi: 10.1089/hum.2013.238.
22. Marco S et al. In vivo gene therapy for mucopolysaccharidosis type III (Sanfilippo syndrome): A new treatment horizon. Hum Gene Ther. 2019 Oct;30(10):1211-1121. doi: 10.1089/hum.2019.217.
23. Taylor M et al. Hematopoietic stem cell transplantation for mucopolysaccharidoses: Past, present, and future. Biol Blood Marrow Transplant. 2019 Jul;25(7):e226-e246. doi: 10.1016/j.bbmt.2019.02.012.
24. Sivakumur P, Wraith JE. Bone marrow transplantation in mucopolysaccharidosis type IIIA: A comparison of an early treated patient with his untreated sibling. J Inherit Metab Dis. 1999 Oct;22(7):849-50. doi: 10.1023/a:1005526628598.
Sanfilippo syndrome is a rare inherited neurodegenerative metabolic disorder for which there are no approved therapies. Symptoms of the more severe subtypes typically begin within the first years of life, rapidly producing serious and progressive physical and cognitive deficits. The underlying pathophysiology is targetable, but the delay in diagnosis of this as well as other lysosomal storage disorders (LSDs) is slowing progress toward effective therapies.
“Lack of awareness and the delays to diagnosis have been a real challenge for us. There is reason for cautious optimism about treatments now in or approaching clinical studies, but to evaluate efficacy on cognitive outcomes we need to enroll more children at a very young age, before loss of milestones,” according to Cara O’Neill, MD, a co-founder and chief science officer of Cure Sanfilippo Foundation.
Epidemiology and description
Sanfilippo syndrome, like the more than 50 other LSDs, is caused by a gene mutation that leads to an enzyme deficiency in the lysosome.1 In the case of Sanfilippo syndrome, also known as mucopolysaccharidosis (MPS III), there are hundreds of mutations that can lead to Sanfilippo by altering the function of one of the four genes essential to degradation of heparan sulfate.2 Lysosomal accumulation of heparan sulfate drives a broad spectrum of progressive and largely irreversible symptoms that typically begin with somatic manifestations, such as bowel dysfunction and recurrent ear and upper respiratory infections.
Impairment of the central nervous system (CNS) usually occurs early in life, halting physical and mental development. As it progresses, accumulation of heparan sulfate in a variety of cells leads to a cascade of abnormal cellular signaling and dysfunction. Disruption of these processes, which are critical for normal neurodevelopment, result in loss of the developmental skills already gained and eventually loss of brain tissue.3 Although life expectancy has improved with supportive care, survival into adulthood is typically limited to milder forms.4
Over the past several years, progress in this and other LSDs has yielded therapeutic targets, including those involving gene repair and enzyme replacement. Already approved for use in some LSDs, these therapies have also shown promise in the experimental setting for Sanfilippo syndrome, leading to several completed clinical trials.5
So far, none of these treatments has advanced beyond clinical trials in Sanfilippo syndrome, but there have been favorable changes in the markers of disease, suggesting that better methods of treatment delivery and/or more sensitive tools to measure clinical change might lead to evidence of disease attenuation. However, the promise of treatment in all cases has been to prevent, slow, or halt progression, not to reverse it. This point is important, because it indicates that degree of benefit will depend on enrolling patients early in life. Even if effective therapies are identified, few patients will benefit without strategies to accelerate diagnosis.
In fact, “one study6 reported that the average age of diagnosis for Sanfilippo syndrome has not improved over the past 30 years,” according to Dr. O’Neill. She indicated that this has been frustrating, given the availability of clinical trials on which progress is dependent. There is no widely accepted protocol for who and when to test for Sanfilippo syndrome or other LSDs, but Dr. O’Neill’s organization is among those advocating for strategies to detect these diseases earlier, including screening at birth.
Almost by definition, the clinical diagnosis of rare diseases poses a challenge. With nonspecific symptoms and a broad range of potential diagnoses, diseases with a low incidence are not the first ones that are typically considered. In the case of Sanfilippo syndrome, published studies indicate incidence rates at or below 1 per 70,000 live births.7 However, the incidence rates have been highly variable not only by geographical regions but even across neighboring countries where genetic risk would be expected to be similar.
In Europe, for example, epidemiologic studies suggest the lifetime risk of MPS IIIA is approximately two times greater in Germany and the Netherlands relative to France and Sweden.7 It is possible that the methodology for identifying cases might be a more important factor than differences in genetic risk to explain this variability. Many experts, including Dr. O’Neill, believe that prevalence figures for Sanfilippo syndrome are typically underestimates because of the frequency with which LSDs are attributed to other pathology.
“For these types of rare disorders, a clinician might only see a single case over a career, and the symptoms can vary in presentation and severity with many alternatives to consider in the differential diagnosis,” Dr. O’Neill explained. She cited case reports in which symptoms of Sanfilippo syndrome after a period of initial normal development has been initially attributed to autism, which is a comorbid feature of the disease, idiopathic developmental delay, or other nonprogressive disorders until further clinical deterioration leads to additional testing. The implication is that LSDs must be considered far earlier despite their rarity.
For the least common of the four clinical subtypes, MPS IIIC and MPS IIID, the median ages of diagnosis have ranged from 4.5 to 19 years of age.7 This is likely a reflection of a slower progression and a later onset of clinical manifestations.
For the more rapidly progressing and typically more severe subtypes, MPS IIIA and MPS IIIB, the diagnosis is typically made earlier. In one review of epidemiologic studies in different countries, the earliest reported median age at diagnosis was 2.5 years,7 a point at which significant disease progression is likely to have already occurred. If the promise of treatments in development is prevention of disease progression, disability in many patients might be substantial if the time to diagnosis is not reduced.
Screening and testing
Independent of the potential to enroll children in clinical trials, early diagnosis also advances the opportunities for supportive care to lessen the burden of the disease on patients and families. Perhaps even more important, early diagnosis is vital to family planning. Since the American pediatrician Sylvester Sanfilippo, MD, first described this syndrome in 1963,7 the genetic profile and many of the features of the disease have become well characterized.8
“One reason to emphasize the importance of early diagnosis is the heritability of this disorder. With prompt diagnosis, genetic counseling can be offered to families to provide them with critical information for future family planning and for cascade testing of other potentially affected siblings,” Dr. O’Neill reported. The inheritance pattern of Sanfilippo syndrome is autosomal recessive.3 In families with an affected child, the risk for any subsequent child to have the same disorder is 25%. The chance of a sibling to be unaffected and not a carrier is also 25%. There is a 50% chance of a sibling to be a carrier but asymptomatic. Of priorities, spreading awareness has been a critical mission of the Cure Sanfilippo Foundation since it was founded 8 years ago, according to Glenn O’Neill, the president. He and his wife, Dr. O’Neill, who is a pediatrician, founded the organization after their own child’s diagnosis of Sanfilippo syndrome. Creating awareness is fundamental to the mission of attracting funds for research, but support to patients and their families as well as early enrollment in clinical trials are among other initiatives being pursued by the foundation to improve care and prognosis.
These strategies include some novel ideas, including an algorithm based on artificial intelligence (AI) that can accelerate suspicion of Sanfilippo syndrome in advance of laboratory or genetic testing, according to Dr. O’Neill. She reported that the facial phenotype, which is observed in a high proportion of but not in all Sanfilippo patients, includes coarse facial features such as puffiness around the eyes, heavy eyebrows, full lips, and macrocephaly.9 Interpretation of photos for AI-based analysis is enhanced when combined with other clinical symptoms.

“The Foundation was involved in honing such a tool by submitting the photos that were used to teach the AI to recognize the Sanfilippo syndrome phenotype,” Dr. O’Neill said. The AI-based tool (Face2Gene.com) is available from FDNA, a company that has been involved in analyzing complex phenotypic and genomic information to guide diagnosis and therapeutic strategies for an array of diseases, not just Sanfilippo syndrome.
The preferred method for diagnosis is biochemical or genetic testing. Of these, urine testing for elevated levels of heparan sulfate glycosaminoglycans (GAG) can be useful for screening, although false-negative tests occur. Analysis of the blood can be performed to detect abnormal levels or activity of the enzymes that break down this GAG. In addition, genetic testing can be performed on blood, fibroblast, buccal swab, or saliva samples. Genetic testing of the blood is the most frequently performed.
For the four MPS III subtypes – MPS IIIA, IIIB, IIIC, and IIID – the presence of two pathogenic mutations in the SGSH (17q25.3), NAGLU (17q21.2), HGSNAT (8p11.21), and GNS (12q14.3) genes, respectively, are likely diagnostic, but enzymatic testing or GAG analysis should be performed to confirm disease status, according to Dr. O’Neill, who said that global consensus based clinical care guidelines led by the Foundation were recently accepted for publication and also include a section on the approach to diagnosis.
While laboratory testing is sensitive, urinary excretion of GAG can be variable, with the potential for ambiguous results. Typically, biochemical and genetic testing provide more reliable results for the diagnosis. They can be readily performed in utero or at the time of birth. In addition, gene panels can permit the diagnosis of multiple types of LSDs, not just Sanfilippo, making screening a cost-effective strategy to consider multiple diseases with overlapping symptoms when an LSD is suspected. Dr. O’Neill said clinical guidelines recommend confirmation of enzyme deficiency or evidence of GAG substrate accumulation as confirmatory tests when genetic testing is positive.
“Ultimately, our goal is to promote universal screening at birth for these serious genetic disorders affecting children,” Dr. O’Neill said.
“We are in a catch-22 when it comes to newborn screening. Currently our federal system requires there be an available treatment before recommending routine screening for a disease. However, it is extremely difficult to power trials with patients who are most likely to show benefit in a trial setting without that very early diagnosis. Universal newborn screening would pave the way for accelerated drug development for children,” she added.
In the meantime, Dr. O’Neill suggests that clinicians should employ a low threshold of suspicion to pursue diagnostic studies of LSDs in infants and children with developmental delays or otherwise unexplained progressive disorders.
Importantly, clinicians can now act quickly on their suspicions and order testing without concern for delays or denial by insurers through a special program, according to Dr. O’Neill. Free genetic testing, offered by the Invitae Corporation, evaluates a panel of 58 genes associated with lysosomal disorders, permitting detection of Sanfilippo syndrome and other LSDs, according to Dr. O’Neill. The Invitae testing is typically performed on 3 mL of whole blood delivered to a central testing facility.
“Results can be obtained within a few weeks or sooner. This can seem like a long wait for families, but it is much more efficient than ordering tests sequentially,” Dr. O’Neill said.
Diagnosis: Signs and symptoms
Despite the differences in progression of the MPS III subtypes, the clinical characteristics are more similar than different. In all patients, prenatal and infant development are typically normal. The initial signs of disease can be found in the newborn, such as neonatal tachypnea, through the early infancy period, such as macrocephaly. However, these are not commonly recognized until about age 1 or soon after in those with MPS IIIA and IIIB.3 Speech delay is the first developmental delay seen in most patients. In those with MPS IIIC, initial symptoms are typically detected at age 3 or later and progress more slowly.10,11 The same is likely to be true of MPS IIID, although this subtype is less well characterized than the other three.7
Although many organs can be involved, degeneration of the CNS is regarded as the most characteristic.3 In aggressive disease, this includes slower acquisition of and failure to meet developmental milestones with progressive intellectual disability, while behavioral difficulties are a more common initial compliant in children with milder disease.13,14 These behavioral changes include hyperactivity, inattention, autistic behaviors, worsening safety awareness, and in some cases aggressive behavior that can be destructive. Sleep disturbances are common.15Because of variability inherent in descriptions of relatively small numbers of patients, the characterization of each of the MPS III subgroups is based on a limited number of small studies, but most patients demonstrate behavior disorders, have coarse facial features, and develop speech delay, according to a survey conducted of published studies.7 Collectively, abnormal behavior was identified as an early symptom in 77% of those with MPS IIIA, 69% of those with IIIB, and 77% of those with IIIC.
For MPS IIIA, loss of speech was observed at a median age of 3.8 years and loss of walking ability at 10.4 years. The median survival has been reported to range between 13 and 18 years. In children with MPS IIIB, the median age of speech loss was reported to about the same age, while loss of walking ability occurred at 11 years. In one study of MPS IIIB, 24% of patients had developed dementia by age 6 years, and the reported median survival has ranged between 17 and 19 years. For MPS IIIC, the onset of clinical symptoms has been observed at a median age of 3.5 years with evidence of cognitive loss observed in 33% of children by the age of 6 years. The median survival has ranged from 19 to 34 years in three studies tracing the natural history of this MPS III subtype.
The differential diagnosis reasonably includes other types of mucopolysaccharidosis disorders with cognitive impairment, including Hurler, Hunter, or Sly syndromes, other neurodevelopmental disorders, and inborn errors of metabolism. The heterogeneity of the features makes definitive laboratory or genetic testing, rather than the effort to differentiate clinical features, appropriate for a definitive diagnosis.
Once the diagnosis is made, other examinations for the common complications of Sanfilippo syndrome are appropriate. Abdominal imaging is appropriate for detecting complications in the gastrointestinal tract, including hepatomegaly, which has been reported in more than half of patients with MPS IIIA and IIIB and in 39% of patients with IIIC.7 In patients with breathing concerns at night and/or sleep disturbance, polysomnography can be useful for identifying sleep apnea and nocturnal seizure activity. In children suspected of seizures, EEG is appropriate. In one study, 66% of patients with MPS IIIA developed seizure activity.16 This has been less commonly reported in MPS IIIB and IIIC, ranging from 8% to 13%.15
Formal hearing evaluation is indicated for any child with speech delays. Hearing loss typically develops after the newborn period in Sanfilippo and may affect peak language acquisition if not treated, according to Dr. O’Neill.
Radiographic studies for dysostosis multiplex or other skeletal abnormalities are also appropriate based on clinical presentation.
Treatment: Present and future
In the absence of treatments to improve the prognosis of Sanfilippo syndrome, current management is based on supportive care and managing organ-specific complications. However, several strategies have proven viable in experimental models and led to clinical trials. None of these therapies has reached approval yet, but several have been associated with attenuation of biomarkers of MPS III disease activity.
Of nearly 30 Sanfilippo clinical trials conducted over the past 20 years, at least 9 have now been completed.5 In addition to studying gene therapy and enzyme replacement therapy, these trials have included stem cell transplantation and substrate reduction therapy, for which the goal is to reduce synthesis of the heparan sulfate GAG to prevent accumulation.5 Of this latter approach, promising initial results with genistein, an isoflavone that breaks down heparan sulfate, reached a phase 3 evaluation.18 Although heparan sulfate levels in the CNS were non-significantly reduced over the course of the trial, the reduction was not sufficient to attenuate cognitive decline.
In other LSDs, several forms of enzyme replacement therapy are now approved. In Fabry disease, for example, recombinant alpha-galactosidase A has now been used for more than 15 years.19 Clinical benefit has not yet been demonstrated in patients with Sanfilippo syndrome because of the difficulty of delivering these therapies past the blood-brain barrier. Several strategies have been pursued. For example, intrathecal delivery of recombinant heparan-N-sulfatase reduced CNS levels of GAG heparan sulfate in one phase 2B study, but it approached but fell short of the statistical significance for the primary endpoint of predefined cognitive stabilization.20 The signal of activity and generally acceptable tolerability has encouraged further study, including an ongoing study with promising interim results of intracerebroventricular enzyme replacement in MPS IIIB, according to Dr. O’Neill.
Acceptable safety and promising activity on disease biomarkers have also been seen with gene therapy in clinical trials. In one study that showed attenuation of brain atrophy, there was moderate improvement in behavior and sleep in three of the four patients enrolled.21 Other studies using various strategies for gene delivery have also produced signals of activity against the underlying pathology, generating persistent interest in ongoing and planned clinical studies with this form of treatment.22Unmodified hematopoietic stem cell transplantation (HSCT), an approach that has demonstrated efficacy when delivered early in the course of other LSDs, such as Hurler syndrome,23 has not yet been associated with significant activity in clinical studies of MPS III, including those that initiated treatment prior to the onset of neurological symptoms.24 However, promising early results have been reported in a study of gene-modified HSCT, which overexpresses the MPS IIIA enzyme.
“The clinical trial landscape fluctuates quite a bit, so I always encourage clinicians and families to check back often for updates. Patient organizations can also be helpful for understanding the most up-to-date and emerging trial options,” Dr. O’Neill reported.
Although it is expected that the greatest benefit would be derived from treatments initiated before or very early after the onset of symptoms, based on the limited potential for reversing cognitive loss, Dr. O’Neill said that she and others are also striving to offer treatments for individuals now living with Sanfilippo syndrome.
“We have to be willing to test treatments that are symptomatic in nature. To that aim, the Cure Sanfilippo Foundation has sponsored a study of a CNS-penetrating anti-inflammatory agent in advanced-disease patients more than 4 years of age,” Dr. O’Neill said. This group of patients typically been ineligible for clinical trials in the past. Dr. O’Neill hopes to change this orientation.
“It is important to highlight that all patients deserve our efforts to improve their quality of life and alleviate suffering, regardless of how old they are or how progressed in the disease they happen to be,” she said.
However, whether the goal is enrollment before or early in disease or later in disease progression, the challenge of enrolling sufficient numbers of patients to confirm clinical activity has been and continues to be a hurdle to progress.
“Clinical studies in Sanfilippo enroll relatively small numbers of patients, often 20 or less,” said Dr. O’Neill, explaining one of the reasons why her organization has been so active in raising awareness and funding such studies. For patients and families, the Cure Sanfilippo Foundation can offer a variety of guidance and support, but information about opportunities for clinical trial participation is a key resource they provide for families and their physicians.
Conclusion
For most children with Sanfilippo syndrome, life expectancy is limited. However, the characterization of the genetic causes and the biochemistry of the subtypes has led to several viable therapeutic approaches under development. There has been progress in delivery of therapeutic enzymes to the CNS, and there is substantial optimism that more progress is coming. One issue for treatment development, is the last of a clear regulatory pathway addressing important biomarkers of pathology, such as heparan sulfate burden. Developing treatments that address this issue or impaired enzyme activity levels have promise for preventing progression, particularly if started in infancy. However, the effort to draw awareness to this disease is the first step toward accelerating the time to an early diagnosis and subsequent opportunities to enroll in clinical trials.
References
1. Sun A. Lysosomal storage disease overview. Ann Transl Med. 2018 Dec;6(24):476. doi: 10.21037/atm.2018.11.39.
2. Andrade F et al. Sanfilippo syndrome: Overall review. Pediatr Int. 2015 Jun;57(3):331-8. doi: 10.1111/ped.12636.
3. Fedele AO. Sanfilippo syndrome: Causes, consequences, and treatments. Appl Clin Genet. 2015 Nov 25;8:269-81. doi: 10.2147/TACG.S57672.
4. Lavery C et al. Mortality in patients with Sanfilippo syndrome. Orphanet J Rare Dis. 2017 Oct 23;12(1):168. doi: 10.1186/s13023-017-0717-y.
5. Pearse Y et al. A cure for Sanfilippo syndrome? A summary of current therapeutic approaches and their promise. Med Res Arch. 2020 Feb 1;8(2). doi: 10.18103/mra.v8i2.2045.
6. Kuiper GA et al. Failure to shorten the diagnostic delay in two ultrao-rphan diseases (mucopolysaccharidosis types I and III): potential causes and implication. Orphanet J Rare Dis. 2018;13:2. Doi: 10.1186/s13023-017-0733-y.
7. Zelei T et al. Epidemiology of Sanfilippo syndrome: Results of a systematic literature review. Orphanet J Rare Dis. 2018 Apr 10;13(1):53. doi: 10.1186/s13023-018-0796-4.
8. Wagner VF, Northrup H. Mucopolysaccaharidosis type III. Gene Reviews. 2019 Sep 19. University of Washington, Seattle. https://www.ncbi.nlm.nih.gov/books/NBK546574/8.
9. O’Neill C et al. Natural history of facial features observed in Sanfilippo syndrome (MPS IIIB) using a next generation phenotyping tool. Mol Genet Metab. 2019 Feb;126:S112.
10. Ruijter GJ et al. Clinical and genetic spectrum of Sanfilippo type C (MPS IIIC) disease in the Netherlands. Mol Genet Metab. 2008 Feb;93(2):104-11. doi: 10.1016/j.ymgme.2007.09.011.
11. Valstar MJ et al. Mucopolysaccharidosis type IIID: 12 new patients and 15 novel mutations. Hum Mutat. 2010 May;31(5):E1348-60. doi: 10.1002/humu.21234.
12. Nijmeijer SCM. The attenuated end of phenotypic spectrum in MPS III: from late-onset stable cognitive impairment to non-neuronopathic phenotype. Orphanet J Rare Dis. 2019;14:249. Doi10.1186/s13023-019-1232-0.
13. Nidiffer FD, Kelly TE. Developmental and degenerative patterns associated with cognitive, behavioural and motor difficulties in the Sanfilippo syndrome: An epidemiological study. J Ment Defic Res. 1983 Sep;27 (Pt 3):185-203. doi: 10.1111/j.1365-2788.1983.tb00291.x.
14. Bax MC, Colville GA. Behaviour in mucopolysaccharide disorders. Arch Dis Child. 1995 Jul;73(1):77-81. doi: 10.1136/adc.73.1.77.
15. Fraser J et al. Sleep disturbance in mucopolysaccharidosis type III (Sanfilippo syndrome): A survey of managing clinicians. Clin Genet. 2002 Nov;62(5):418-21. doi: 10.1034/j.1399-0004.2002.620512.x.
16. Valstar MJ et al. Mucopolysaccharidosis type IIIA: Clinical spectrum and genotype-phenotype correlations. Ann Neurol. 2010 Dec;68(6):876-87. doi: 10.1002/ana.22092.
17. Heron B et al. Incidence and natural history of mucopolysaccharidosis type III in France and comparison with United Kingdom and Greece. Am J Med Genet A. 2011 Jan;155A(1):58-68. doi: 10.1002/ajmg.a.33779.
18. Delgadillo V et al. Genistein supplementation in patients affected by Sanfilippo disease. J Inherit Metab Dis. 2011 Oct;34(5):1039-44. doi: 10.1007/s10545-011-9342-4.
19. van der Veen SJ et al. Developments in the treatment of Fabry disease. J Inherit Metab Dis. 2020 Sep;43(5):908-21. doi: 10.1002/jimd.12228.
20. Wijburg FA et al. Intrathecal heparan-N-sulfatase in patients with Sanfilippo syndrome type A: A phase IIb randomized trial. Mol Genet Metab. 2019 Feb;126(2):121-30. doi: 10.1016/j.ymgme.2018.10.006.
21. Tardieu M et al. Intracerebral administration of adeno-associated viral vector serotype rh.10 carrying human SGSH and SUMF1 cDNAs in children with mucopolysaccharidosis type IIIA disease: Results of a phase I/II trial. Hum Gene Ther. 2014 Jun;25(6):506-16. doi: 10.1089/hum.2013.238.
22. Marco S et al. In vivo gene therapy for mucopolysaccharidosis type III (Sanfilippo syndrome): A new treatment horizon. Hum Gene Ther. 2019 Oct;30(10):1211-1121. doi: 10.1089/hum.2019.217.
23. Taylor M et al. Hematopoietic stem cell transplantation for mucopolysaccharidoses: Past, present, and future. Biol Blood Marrow Transplant. 2019 Jul;25(7):e226-e246. doi: 10.1016/j.bbmt.2019.02.012.
24. Sivakumur P, Wraith JE. Bone marrow transplantation in mucopolysaccharidosis type IIIA: A comparison of an early treated patient with his untreated sibling. J Inherit Metab Dis. 1999 Oct;22(7):849-50. doi: 10.1023/a:1005526628598.
Sanfilippo syndrome is a rare inherited neurodegenerative metabolic disorder for which there are no approved therapies. Symptoms of the more severe subtypes typically begin within the first years of life, rapidly producing serious and progressive physical and cognitive deficits. The underlying pathophysiology is targetable, but the delay in diagnosis of this as well as other lysosomal storage disorders (LSDs) is slowing progress toward effective therapies.
“Lack of awareness and the delays to diagnosis have been a real challenge for us. There is reason for cautious optimism about treatments now in or approaching clinical studies, but to evaluate efficacy on cognitive outcomes we need to enroll more children at a very young age, before loss of milestones,” according to Cara O’Neill, MD, a co-founder and chief science officer of Cure Sanfilippo Foundation.
Epidemiology and description
Sanfilippo syndrome, like the more than 50 other LSDs, is caused by a gene mutation that leads to an enzyme deficiency in the lysosome.1 In the case of Sanfilippo syndrome, also known as mucopolysaccharidosis (MPS III), there are hundreds of mutations that can lead to Sanfilippo by altering the function of one of the four genes essential to degradation of heparan sulfate.2 Lysosomal accumulation of heparan sulfate drives a broad spectrum of progressive and largely irreversible symptoms that typically begin with somatic manifestations, such as bowel dysfunction and recurrent ear and upper respiratory infections.
Impairment of the central nervous system (CNS) usually occurs early in life, halting physical and mental development. As it progresses, accumulation of heparan sulfate in a variety of cells leads to a cascade of abnormal cellular signaling and dysfunction. Disruption of these processes, which are critical for normal neurodevelopment, result in loss of the developmental skills already gained and eventually loss of brain tissue.3 Although life expectancy has improved with supportive care, survival into adulthood is typically limited to milder forms.4
Over the past several years, progress in this and other LSDs has yielded therapeutic targets, including those involving gene repair and enzyme replacement. Already approved for use in some LSDs, these therapies have also shown promise in the experimental setting for Sanfilippo syndrome, leading to several completed clinical trials.5
So far, none of these treatments has advanced beyond clinical trials in Sanfilippo syndrome, but there have been favorable changes in the markers of disease, suggesting that better methods of treatment delivery and/or more sensitive tools to measure clinical change might lead to evidence of disease attenuation. However, the promise of treatment in all cases has been to prevent, slow, or halt progression, not to reverse it. This point is important, because it indicates that degree of benefit will depend on enrolling patients early in life. Even if effective therapies are identified, few patients will benefit without strategies to accelerate diagnosis.
In fact, “one study6 reported that the average age of diagnosis for Sanfilippo syndrome has not improved over the past 30 years,” according to Dr. O’Neill. She indicated that this has been frustrating, given the availability of clinical trials on which progress is dependent. There is no widely accepted protocol for who and when to test for Sanfilippo syndrome or other LSDs, but Dr. O’Neill’s organization is among those advocating for strategies to detect these diseases earlier, including screening at birth.
Almost by definition, the clinical diagnosis of rare diseases poses a challenge. With nonspecific symptoms and a broad range of potential diagnoses, diseases with a low incidence are not the first ones that are typically considered. In the case of Sanfilippo syndrome, published studies indicate incidence rates at or below 1 per 70,000 live births.7 However, the incidence rates have been highly variable not only by geographical regions but even across neighboring countries where genetic risk would be expected to be similar.
In Europe, for example, epidemiologic studies suggest the lifetime risk of MPS IIIA is approximately two times greater in Germany and the Netherlands relative to France and Sweden.7 It is possible that the methodology for identifying cases might be a more important factor than differences in genetic risk to explain this variability. Many experts, including Dr. O’Neill, believe that prevalence figures for Sanfilippo syndrome are typically underestimates because of the frequency with which LSDs are attributed to other pathology.
“For these types of rare disorders, a clinician might only see a single case over a career, and the symptoms can vary in presentation and severity with many alternatives to consider in the differential diagnosis,” Dr. O’Neill explained. She cited case reports in which symptoms of Sanfilippo syndrome after a period of initial normal development has been initially attributed to autism, which is a comorbid feature of the disease, idiopathic developmental delay, or other nonprogressive disorders until further clinical deterioration leads to additional testing. The implication is that LSDs must be considered far earlier despite their rarity.
For the least common of the four clinical subtypes, MPS IIIC and MPS IIID, the median ages of diagnosis have ranged from 4.5 to 19 years of age.7 This is likely a reflection of a slower progression and a later onset of clinical manifestations.
For the more rapidly progressing and typically more severe subtypes, MPS IIIA and MPS IIIB, the diagnosis is typically made earlier. In one review of epidemiologic studies in different countries, the earliest reported median age at diagnosis was 2.5 years,7 a point at which significant disease progression is likely to have already occurred. If the promise of treatments in development is prevention of disease progression, disability in many patients might be substantial if the time to diagnosis is not reduced.
Screening and testing
Independent of the potential to enroll children in clinical trials, early diagnosis also advances the opportunities for supportive care to lessen the burden of the disease on patients and families. Perhaps even more important, early diagnosis is vital to family planning. Since the American pediatrician Sylvester Sanfilippo, MD, first described this syndrome in 1963,7 the genetic profile and many of the features of the disease have become well characterized.8
“One reason to emphasize the importance of early diagnosis is the heritability of this disorder. With prompt diagnosis, genetic counseling can be offered to families to provide them with critical information for future family planning and for cascade testing of other potentially affected siblings,” Dr. O’Neill reported. The inheritance pattern of Sanfilippo syndrome is autosomal recessive.3 In families with an affected child, the risk for any subsequent child to have the same disorder is 25%. The chance of a sibling to be unaffected and not a carrier is also 25%. There is a 50% chance of a sibling to be a carrier but asymptomatic. Of priorities, spreading awareness has been a critical mission of the Cure Sanfilippo Foundation since it was founded 8 years ago, according to Glenn O’Neill, the president. He and his wife, Dr. O’Neill, who is a pediatrician, founded the organization after their own child’s diagnosis of Sanfilippo syndrome. Creating awareness is fundamental to the mission of attracting funds for research, but support to patients and their families as well as early enrollment in clinical trials are among other initiatives being pursued by the foundation to improve care and prognosis.
These strategies include some novel ideas, including an algorithm based on artificial intelligence (AI) that can accelerate suspicion of Sanfilippo syndrome in advance of laboratory or genetic testing, according to Dr. O’Neill. She reported that the facial phenotype, which is observed in a high proportion of but not in all Sanfilippo patients, includes coarse facial features such as puffiness around the eyes, heavy eyebrows, full lips, and macrocephaly.9 Interpretation of photos for AI-based analysis is enhanced when combined with other clinical symptoms.

“The Foundation was involved in honing such a tool by submitting the photos that were used to teach the AI to recognize the Sanfilippo syndrome phenotype,” Dr. O’Neill said. The AI-based tool (Face2Gene.com) is available from FDNA, a company that has been involved in analyzing complex phenotypic and genomic information to guide diagnosis and therapeutic strategies for an array of diseases, not just Sanfilippo syndrome.
The preferred method for diagnosis is biochemical or genetic testing. Of these, urine testing for elevated levels of heparan sulfate glycosaminoglycans (GAG) can be useful for screening, although false-negative tests occur. Analysis of the blood can be performed to detect abnormal levels or activity of the enzymes that break down this GAG. In addition, genetic testing can be performed on blood, fibroblast, buccal swab, or saliva samples. Genetic testing of the blood is the most frequently performed.
For the four MPS III subtypes – MPS IIIA, IIIB, IIIC, and IIID – the presence of two pathogenic mutations in the SGSH (17q25.3), NAGLU (17q21.2), HGSNAT (8p11.21), and GNS (12q14.3) genes, respectively, are likely diagnostic, but enzymatic testing or GAG analysis should be performed to confirm disease status, according to Dr. O’Neill, who said that global consensus based clinical care guidelines led by the Foundation were recently accepted for publication and also include a section on the approach to diagnosis.
While laboratory testing is sensitive, urinary excretion of GAG can be variable, with the potential for ambiguous results. Typically, biochemical and genetic testing provide more reliable results for the diagnosis. They can be readily performed in utero or at the time of birth. In addition, gene panels can permit the diagnosis of multiple types of LSDs, not just Sanfilippo, making screening a cost-effective strategy to consider multiple diseases with overlapping symptoms when an LSD is suspected. Dr. O’Neill said clinical guidelines recommend confirmation of enzyme deficiency or evidence of GAG substrate accumulation as confirmatory tests when genetic testing is positive.
“Ultimately, our goal is to promote universal screening at birth for these serious genetic disorders affecting children,” Dr. O’Neill said.
“We are in a catch-22 when it comes to newborn screening. Currently our federal system requires there be an available treatment before recommending routine screening for a disease. However, it is extremely difficult to power trials with patients who are most likely to show benefit in a trial setting without that very early diagnosis. Universal newborn screening would pave the way for accelerated drug development for children,” she added.
In the meantime, Dr. O’Neill suggests that clinicians should employ a low threshold of suspicion to pursue diagnostic studies of LSDs in infants and children with developmental delays or otherwise unexplained progressive disorders.
Importantly, clinicians can now act quickly on their suspicions and order testing without concern for delays or denial by insurers through a special program, according to Dr. O’Neill. Free genetic testing, offered by the Invitae Corporation, evaluates a panel of 58 genes associated with lysosomal disorders, permitting detection of Sanfilippo syndrome and other LSDs, according to Dr. O’Neill. The Invitae testing is typically performed on 3 mL of whole blood delivered to a central testing facility.
“Results can be obtained within a few weeks or sooner. This can seem like a long wait for families, but it is much more efficient than ordering tests sequentially,” Dr. O’Neill said.
Diagnosis: Signs and symptoms
Despite the differences in progression of the MPS III subtypes, the clinical characteristics are more similar than different. In all patients, prenatal and infant development are typically normal. The initial signs of disease can be found in the newborn, such as neonatal tachypnea, through the early infancy period, such as macrocephaly. However, these are not commonly recognized until about age 1 or soon after in those with MPS IIIA and IIIB.3 Speech delay is the first developmental delay seen in most patients. In those with MPS IIIC, initial symptoms are typically detected at age 3 or later and progress more slowly.10,11 The same is likely to be true of MPS IIID, although this subtype is less well characterized than the other three.7
Although many organs can be involved, degeneration of the CNS is regarded as the most characteristic.3 In aggressive disease, this includes slower acquisition of and failure to meet developmental milestones with progressive intellectual disability, while behavioral difficulties are a more common initial compliant in children with milder disease.13,14 These behavioral changes include hyperactivity, inattention, autistic behaviors, worsening safety awareness, and in some cases aggressive behavior that can be destructive. Sleep disturbances are common.15Because of variability inherent in descriptions of relatively small numbers of patients, the characterization of each of the MPS III subgroups is based on a limited number of small studies, but most patients demonstrate behavior disorders, have coarse facial features, and develop speech delay, according to a survey conducted of published studies.7 Collectively, abnormal behavior was identified as an early symptom in 77% of those with MPS IIIA, 69% of those with IIIB, and 77% of those with IIIC.
For MPS IIIA, loss of speech was observed at a median age of 3.8 years and loss of walking ability at 10.4 years. The median survival has been reported to range between 13 and 18 years. In children with MPS IIIB, the median age of speech loss was reported to about the same age, while loss of walking ability occurred at 11 years. In one study of MPS IIIB, 24% of patients had developed dementia by age 6 years, and the reported median survival has ranged between 17 and 19 years. For MPS IIIC, the onset of clinical symptoms has been observed at a median age of 3.5 years with evidence of cognitive loss observed in 33% of children by the age of 6 years. The median survival has ranged from 19 to 34 years in three studies tracing the natural history of this MPS III subtype.
The differential diagnosis reasonably includes other types of mucopolysaccharidosis disorders with cognitive impairment, including Hurler, Hunter, or Sly syndromes, other neurodevelopmental disorders, and inborn errors of metabolism. The heterogeneity of the features makes definitive laboratory or genetic testing, rather than the effort to differentiate clinical features, appropriate for a definitive diagnosis.
Once the diagnosis is made, other examinations for the common complications of Sanfilippo syndrome are appropriate. Abdominal imaging is appropriate for detecting complications in the gastrointestinal tract, including hepatomegaly, which has been reported in more than half of patients with MPS IIIA and IIIB and in 39% of patients with IIIC.7 In patients with breathing concerns at night and/or sleep disturbance, polysomnography can be useful for identifying sleep apnea and nocturnal seizure activity. In children suspected of seizures, EEG is appropriate. In one study, 66% of patients with MPS IIIA developed seizure activity.16 This has been less commonly reported in MPS IIIB and IIIC, ranging from 8% to 13%.15
Formal hearing evaluation is indicated for any child with speech delays. Hearing loss typically develops after the newborn period in Sanfilippo and may affect peak language acquisition if not treated, according to Dr. O’Neill.
Radiographic studies for dysostosis multiplex or other skeletal abnormalities are also appropriate based on clinical presentation.
Treatment: Present and future
In the absence of treatments to improve the prognosis of Sanfilippo syndrome, current management is based on supportive care and managing organ-specific complications. However, several strategies have proven viable in experimental models and led to clinical trials. None of these therapies has reached approval yet, but several have been associated with attenuation of biomarkers of MPS III disease activity.
Of nearly 30 Sanfilippo clinical trials conducted over the past 20 years, at least 9 have now been completed.5 In addition to studying gene therapy and enzyme replacement therapy, these trials have included stem cell transplantation and substrate reduction therapy, for which the goal is to reduce synthesis of the heparan sulfate GAG to prevent accumulation.5 Of this latter approach, promising initial results with genistein, an isoflavone that breaks down heparan sulfate, reached a phase 3 evaluation.18 Although heparan sulfate levels in the CNS were non-significantly reduced over the course of the trial, the reduction was not sufficient to attenuate cognitive decline.
In other LSDs, several forms of enzyme replacement therapy are now approved. In Fabry disease, for example, recombinant alpha-galactosidase A has now been used for more than 15 years.19 Clinical benefit has not yet been demonstrated in patients with Sanfilippo syndrome because of the difficulty of delivering these therapies past the blood-brain barrier. Several strategies have been pursued. For example, intrathecal delivery of recombinant heparan-N-sulfatase reduced CNS levels of GAG heparan sulfate in one phase 2B study, but it approached but fell short of the statistical significance for the primary endpoint of predefined cognitive stabilization.20 The signal of activity and generally acceptable tolerability has encouraged further study, including an ongoing study with promising interim results of intracerebroventricular enzyme replacement in MPS IIIB, according to Dr. O’Neill.
Acceptable safety and promising activity on disease biomarkers have also been seen with gene therapy in clinical trials. In one study that showed attenuation of brain atrophy, there was moderate improvement in behavior and sleep in three of the four patients enrolled.21 Other studies using various strategies for gene delivery have also produced signals of activity against the underlying pathology, generating persistent interest in ongoing and planned clinical studies with this form of treatment.22Unmodified hematopoietic stem cell transplantation (HSCT), an approach that has demonstrated efficacy when delivered early in the course of other LSDs, such as Hurler syndrome,23 has not yet been associated with significant activity in clinical studies of MPS III, including those that initiated treatment prior to the onset of neurological symptoms.24 However, promising early results have been reported in a study of gene-modified HSCT, which overexpresses the MPS IIIA enzyme.
“The clinical trial landscape fluctuates quite a bit, so I always encourage clinicians and families to check back often for updates. Patient organizations can also be helpful for understanding the most up-to-date and emerging trial options,” Dr. O’Neill reported.
Although it is expected that the greatest benefit would be derived from treatments initiated before or very early after the onset of symptoms, based on the limited potential for reversing cognitive loss, Dr. O’Neill said that she and others are also striving to offer treatments for individuals now living with Sanfilippo syndrome.
“We have to be willing to test treatments that are symptomatic in nature. To that aim, the Cure Sanfilippo Foundation has sponsored a study of a CNS-penetrating anti-inflammatory agent in advanced-disease patients more than 4 years of age,” Dr. O’Neill said. This group of patients typically been ineligible for clinical trials in the past. Dr. O’Neill hopes to change this orientation.
“It is important to highlight that all patients deserve our efforts to improve their quality of life and alleviate suffering, regardless of how old they are or how progressed in the disease they happen to be,” she said.
However, whether the goal is enrollment before or early in disease or later in disease progression, the challenge of enrolling sufficient numbers of patients to confirm clinical activity has been and continues to be a hurdle to progress.
“Clinical studies in Sanfilippo enroll relatively small numbers of patients, often 20 or less,” said Dr. O’Neill, explaining one of the reasons why her organization has been so active in raising awareness and funding such studies. For patients and families, the Cure Sanfilippo Foundation can offer a variety of guidance and support, but information about opportunities for clinical trial participation is a key resource they provide for families and their physicians.
Conclusion
For most children with Sanfilippo syndrome, life expectancy is limited. However, the characterization of the genetic causes and the biochemistry of the subtypes has led to several viable therapeutic approaches under development. There has been progress in delivery of therapeutic enzymes to the CNS, and there is substantial optimism that more progress is coming. One issue for treatment development, is the last of a clear regulatory pathway addressing important biomarkers of pathology, such as heparan sulfate burden. Developing treatments that address this issue or impaired enzyme activity levels have promise for preventing progression, particularly if started in infancy. However, the effort to draw awareness to this disease is the first step toward accelerating the time to an early diagnosis and subsequent opportunities to enroll in clinical trials.
References
1. Sun A. Lysosomal storage disease overview. Ann Transl Med. 2018 Dec;6(24):476. doi: 10.21037/atm.2018.11.39.
2. Andrade F et al. Sanfilippo syndrome: Overall review. Pediatr Int. 2015 Jun;57(3):331-8. doi: 10.1111/ped.12636.
3. Fedele AO. Sanfilippo syndrome: Causes, consequences, and treatments. Appl Clin Genet. 2015 Nov 25;8:269-81. doi: 10.2147/TACG.S57672.
4. Lavery C et al. Mortality in patients with Sanfilippo syndrome. Orphanet J Rare Dis. 2017 Oct 23;12(1):168. doi: 10.1186/s13023-017-0717-y.
5. Pearse Y et al. A cure for Sanfilippo syndrome? A summary of current therapeutic approaches and their promise. Med Res Arch. 2020 Feb 1;8(2). doi: 10.18103/mra.v8i2.2045.
6. Kuiper GA et al. Failure to shorten the diagnostic delay in two ultrao-rphan diseases (mucopolysaccharidosis types I and III): potential causes and implication. Orphanet J Rare Dis. 2018;13:2. Doi: 10.1186/s13023-017-0733-y.
7. Zelei T et al. Epidemiology of Sanfilippo syndrome: Results of a systematic literature review. Orphanet J Rare Dis. 2018 Apr 10;13(1):53. doi: 10.1186/s13023-018-0796-4.
8. Wagner VF, Northrup H. Mucopolysaccaharidosis type III. Gene Reviews. 2019 Sep 19. University of Washington, Seattle. https://www.ncbi.nlm.nih.gov/books/NBK546574/8.
9. O’Neill C et al. Natural history of facial features observed in Sanfilippo syndrome (MPS IIIB) using a next generation phenotyping tool. Mol Genet Metab. 2019 Feb;126:S112.
10. Ruijter GJ et al. Clinical and genetic spectrum of Sanfilippo type C (MPS IIIC) disease in the Netherlands. Mol Genet Metab. 2008 Feb;93(2):104-11. doi: 10.1016/j.ymgme.2007.09.011.
11. Valstar MJ et al. Mucopolysaccharidosis type IIID: 12 new patients and 15 novel mutations. Hum Mutat. 2010 May;31(5):E1348-60. doi: 10.1002/humu.21234.
12. Nijmeijer SCM. The attenuated end of phenotypic spectrum in MPS III: from late-onset stable cognitive impairment to non-neuronopathic phenotype. Orphanet J Rare Dis. 2019;14:249. Doi10.1186/s13023-019-1232-0.
13. Nidiffer FD, Kelly TE. Developmental and degenerative patterns associated with cognitive, behavioural and motor difficulties in the Sanfilippo syndrome: An epidemiological study. J Ment Defic Res. 1983 Sep;27 (Pt 3):185-203. doi: 10.1111/j.1365-2788.1983.tb00291.x.
14. Bax MC, Colville GA. Behaviour in mucopolysaccharide disorders. Arch Dis Child. 1995 Jul;73(1):77-81. doi: 10.1136/adc.73.1.77.
15. Fraser J et al. Sleep disturbance in mucopolysaccharidosis type III (Sanfilippo syndrome): A survey of managing clinicians. Clin Genet. 2002 Nov;62(5):418-21. doi: 10.1034/j.1399-0004.2002.620512.x.
16. Valstar MJ et al. Mucopolysaccharidosis type IIIA: Clinical spectrum and genotype-phenotype correlations. Ann Neurol. 2010 Dec;68(6):876-87. doi: 10.1002/ana.22092.
17. Heron B et al. Incidence and natural history of mucopolysaccharidosis type III in France and comparison with United Kingdom and Greece. Am J Med Genet A. 2011 Jan;155A(1):58-68. doi: 10.1002/ajmg.a.33779.
18. Delgadillo V et al. Genistein supplementation in patients affected by Sanfilippo disease. J Inherit Metab Dis. 2011 Oct;34(5):1039-44. doi: 10.1007/s10545-011-9342-4.
19. van der Veen SJ et al. Developments in the treatment of Fabry disease. J Inherit Metab Dis. 2020 Sep;43(5):908-21. doi: 10.1002/jimd.12228.
20. Wijburg FA et al. Intrathecal heparan-N-sulfatase in patients with Sanfilippo syndrome type A: A phase IIb randomized trial. Mol Genet Metab. 2019 Feb;126(2):121-30. doi: 10.1016/j.ymgme.2018.10.006.
21. Tardieu M et al. Intracerebral administration of adeno-associated viral vector serotype rh.10 carrying human SGSH and SUMF1 cDNAs in children with mucopolysaccharidosis type IIIA disease: Results of a phase I/II trial. Hum Gene Ther. 2014 Jun;25(6):506-16. doi: 10.1089/hum.2013.238.
22. Marco S et al. In vivo gene therapy for mucopolysaccharidosis type III (Sanfilippo syndrome): A new treatment horizon. Hum Gene Ther. 2019 Oct;30(10):1211-1121. doi: 10.1089/hum.2019.217.
23. Taylor M et al. Hematopoietic stem cell transplantation for mucopolysaccharidoses: Past, present, and future. Biol Blood Marrow Transplant. 2019 Jul;25(7):e226-e246. doi: 10.1016/j.bbmt.2019.02.012.
24. Sivakumur P, Wraith JE. Bone marrow transplantation in mucopolysaccharidosis type IIIA: A comparison of an early treated patient with his untreated sibling. J Inherit Metab Dis. 1999 Oct;22(7):849-50. doi: 10.1023/a:1005526628598.
Novel gene-based therapies for neuromuscular diseases
Neuromuscular diseases (NMDs) are a broad classification of heterogeneous groups of disorders characterized by progressive muscle weakness resulting from muscle or nerve dysfunction.1 Diagnosis is based on symptoms and a full medical history, as well as on muscle and imaging tests (including electromyography, nerve-conduction studies, magnetic resonance imaging, muscle biopsy, and blood tests) to confirm or rule out specific NMDs.2 Early diagnosis of NMDs can be difficult because symptoms overlap with those of many other diseases.
Although individually, NMDs are rare, collectively, they affect approximately 250,000 people in the United States. Disease types vary in regard to cause, symptoms, prevalence, age of onset, progression, and severity. Functional impairment from any NMD can lead to lifelong morbidities and shortened life expectancy.1,3
Treatment options for NMDs are limited; most target symptoms, not disease progression. Although there is a need for safe and effective gene-based therapies for NMDs, there are challenges to developing and delivering such treatments that have impeded clinical success. These include a lack of understanding about disease pathology and drug targets, limited animal model systems, and few reliable biomarkers that are predictive of therapeutic success.4,5
Notwithstanding that challenges remain, our understanding of gene expression in NMDs has greatly advanced in the past few decades. This progress has translated into promising results in the gene-therapy field – thereby setting the stage for therapeutic approaches that use novel gene-delivery and gene-manipulation tools.6 These novel approaches include nonviral strategies, such as antisense oligonucleotides (ASOs), and viral-based strategies, such as adeno-associated virus (AAV)-mediated gene silencing and AAV-mediated gene delivery.
In this article, we highlight advancements in the clinical development of gene-based therapies for NMDs. We focus on amyotrophic lateral sclerosis (ALS), spinal muscular atrophy (SMA), and Duchenne muscular dystrophy (DMD) because of recent clinical successes in developing such therapies.1,6,7 We also catalog completed and ongoing clinical trials for ALS, SMA, and DMD (Tables 1-3).
Amyotrophic lateral sclerosis
ALS is caused by progressive degeneration of upper- and lower-motor neurons, which eventually leads to respiratory failure and death 3 to 5 years after disease onset.7-9 There are two subtypes: Familial ALS (10% of cases) and sporadic ALS (90% of cases). Commonly mutated ALS-associated genes6,8 are:
- Superoxide dismutase type 1 (SOD1).
- Chromosome 9 open reading frame 72 (C9orf72).
- Transactive response DNA-binding protein 43 (TARDBP).
- Fused in sarcoma (FUS).
SOD1-targeted therapy is being studied, with early evidence of clinical success. Mutations in SOD1 account for 10% to 20% of familial ALS cases and 1% to 2% of sporadic ALS cases.6,10 10 Mutations in C9orf72 account for 25 to 40% of familial ALS cases and 7% of sporadic ALS cases.8,9,11 Mutations in TARDBP account for 3% of familial ALS cases and 2% of sporadic cases.12 Mutations in FUS account for 4% of familial ALS cases and 1% of sporadic cases. Overall, these mutant proteins can trigger neurotoxicity, thus inducing motor-neuron death.6,10
Treatment of ALS
Two treatments for ALS are Food and Drug Administration approved: riluzole (Rilutek), approved in 1995, and edaravone (Radicava), approved in 2017.
Riluzole is an oral anti-excitotoxic glutamate antagonist.11 Approval of riluzole was based on the results of two studies that demonstrated a 2- to 3-month survival benefit.10,14 For patients who have difficulty swallowing, an oral suspension (Tiglutik, approved in 2018) and an oral film (Exservan, approved in 2019) are available.
Edaravone is a free-radical scavenger that decreases oxidative stress and is administered intravenously (IV).9,13,14 Findings from clinical trials suggest functional improvement or slower decline in function for some patients.
Although these two agents demonstrate modest therapeutic benefit, neither reverses progression of disease.10,14
Gene-based therapy for ALS
Many non-viral strategies, including antisense oligonucleotide (ASO), monoclonal antibodies, reverse transcriptase inhibitors, and HGF gene replacement therapy are used as therapeutic approaches to SOD1, C9orf72, and FUS gene mutations in ALS patients, and are being evaluated in clinical studies14,15 (Table 113-17).
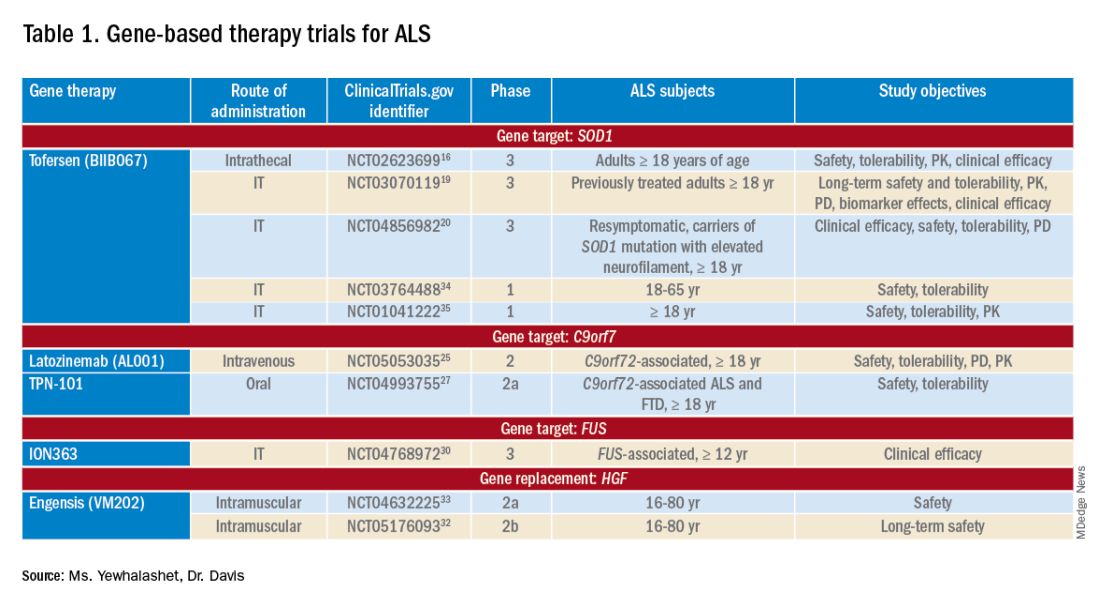
Tofersen, also known as BIIB067, is an investigational ASO, administered by intrathecal (IT) injection, that binds to SOD1 mRNA, thus reducing its protein levels.16 Tofersen was evaluated in the VALOR phase 3 study (ClinicalTrials.gov Identifier: NCT02623699), a three-part randomized, double-blind, placebo-controlled trial: single ascending dose (Part A), multiple ascending dose (B), and fixed dose (C).10 In Parts A and B, 48 participants received five IT injections of tofersen or placebo over 12 weeks and were followed for an additional 12 weeks. Reduction in SOD1 protein production and neurofilament level in cerebrospinal fluid (CSF) (a potential biomarker of motor-neuron degeneration) was observed, which determined the fixed-dose for Part C.16,17
Part C examined the efficacy, safety and tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) of tofersen, compared with placebo, in adults with ALS who had a confirmed SOD1 mutation.17 A total of 108 participants were enrolled; 60 were identified as “faster-progressing”; 48, as “slower-progressing.”18 The primary endpoint of Part C was change from baseline to Week 28 on the Revised ALS Functional Rating Scale (ALSFRS-R) total score. (ALSFRS-R measures overall clinical effect; the score ranges from 0 [no function] to 4 [full function].17)
Tofersen failed to meet the primary efficacy outcome because statistically significant findings were lacking in the faster-progressing population, as measured by joint-rank analysis (difference of 1.2 on the ALSFRS-R score; P = .97). However, trends favoring tofersen were observed across key secondary clinical outcome measures18:
- Change from baseline in CSF SOD1 protein concentration.17 Percent reduction in the total SOD1 protein level was much higher in the tofersen-treated group than in the control group (38% more than controls in the faster-progressing population; 26% more than controls in the slower-progressing population).18
- Change from baseline in neurofilament light-chain concentration in plasma.17,18 Percent reduction in the level of neurofilament light chain was also observed to be higher in the tofersen-treated group than in the control group (67% more than controls in the faster-progressing population and 48% more than controls in the slower-progressing population).18
Because of these encouraging results, VALOR participants were moved to the ongoing open-label extension trial of tofersen (ClinicalTri-als.gov Identifier: NCT03070119), in which both groups were treated with the active agent.
These data suggest that early tofersen treatment might slow decline in faster-progressing patients and stabilize clinical function in slower-progressing patients.18,19 Overall, most adverse events (AEs) in the trial among patients receiving active treatment were of mild or moderate severity, and were largely consistent with either disease progression or lumbar puncture–related complications.18
Because data from VALOR suggested potential benefit from tofersen, the ATLAS trial (ClinicalTrials.gov Identifier: NCT04856982) is investigating the clinical value of presymptomatic treatment and the optimal timing of initiation of therapy.20,21 ATLAS is a phase 3, randomized, placebo-controlled trial that examines the clinical efficacy, safety, and tolerability of tofersen in presymptomatic adult carriers of SOD1 mutation who have an elevated neurofilament light-chain concentration.21 ATLAS will also evaluate the efficacy of tofersen when initiated before, rather than after, ALS manifests clinically. Enrollment is still open for this trial.20,21
Latozinemab, also known as AL001, is a first-in-class monoclonal antibody, administered by IV infusion, that elevates levels of progranulin, a key regulator of the immune activity and lysosomal function in the brain.22,23 Latozinemab limits progranulin endocytosis and degradation by sortilin inhibition.22 Progranulin gene mutations can reduce progranulin expression (by 50 to 70 percent reduction), which may cause neuro-degeneration due to abnormal accumulation of TAR-DNA-binding protein 43 (TDP-43) in the brain cells.22,24 TDP-43 pathology has also been shown to be associated with C9orf72 mutations.23 Although the mechanism is not fully understood, the role of progranulin deficiency in TDP-43 pathology is believed to be associated with neurodegenerative diseases like ALS.11,23,24,43 Previous animal models of chronic neurodegenera-tion have demonstrated how increased progranulin levels can be protective against TDP-43 pathology, increasing neuronal development and survival, thus potentially slowing disease progression.23,24,43 Currently, latozinemab is being investigated in a randomized, double-blind, placebo-controlled, multicenter phase 2 trial (ClinicalTrials.gov Identifier: NCT05053035). Approximately, 45 C90rf72-associated ALS participants (≥ 18 years of age) will receive latozinemab or placebo infusions every 4 weeks (for 24 weeks). Study endpoints include safety, tolerability, PK, PD, as well as plasma, and CSF progranulin levels.25 In previous studies, latozinemab demonstrated encouraging results in frontotemporal dementia (FTD) patients who carry a progranulin mutation. Because FTD was revealed to have significant genetic overlap with ALS, there is disease-modifying potential for latozinemab in ALS patients.23,24
TPN-101 is a nucleoside analog reverse transcriptase inhibitor, administered orally, that was originally developed for human immunodeficiency virus (HIV) treatment. However, due to recent findings suggesting retrotransposon activity contributing to neurodegeneration in TDP-43 mediated diseases, including ALS and FTD, TNP-101 is being repurposed.26 The safety and tolerability of TNP-101 are currently being evaluated in C9orf72-associated ALS and FTD patients (≥ 18 years of age). The study is a randomized, double-blind, placebo-controlled paral-lel-group phase 2a trial (ClinicalTrials.gov Identifier: NCT04993755) The study includes a screening period of 6 weeks, double-blind treatment period of 24 weeks, an open-label treatment period of 24 weeks, and 4 weeks of the post-treatment follow-up visit. Study endpoints include the incidence and severity of spontaneously reported treatment-emergent adverse events (TEAEs) associated with TNP-101 and placebo for a to-tal of 48 weeks.27
ION363 is an investigational ASO, administered by IT injection, that selectively targets one of the FUS mutations (p.P525L), which is responsible for earlier disease onset and rapid ALS progression.28,29 The clinical efficacy of ION363, specifically in clinical function and survival is being assessed in FUS-associated ALS patients (≥ 12 years of age). This randomized phase 3 study (ClinicalTrials.gov Identifier: NCT04768972) includes two parts; part 1 will consist of participants receiving a multi-dose regimen (1 dose every 4-12 weeks) of ION363 or placebo for 61 weeks followed by an open-label extension treatment period in part 2, which will consist of participants receiving ION363 (every 12 weeks) for 85 weeks. The primary endpoint of the study is the change from baseline to day 505 in functional impairment, using ALS Functional Rating Scale-Revised (ALSFRS-R). This measures functional disease severity, specifically in bulbar function, gross motor skills, fine motor skills, and respiratory. The score for all 12 questions can range from 0 (no function) to 4 (full function) with a total possible score of 48.30
Engensis, also known as VM202, is a non-viral gene therapy, administered by intramuscular (IM) injection, that uses a plasmid to deliver the hepatocyte growth factor (HGF) gene to promote HGF protein production. The HGF protein plays a role in angiogenesis, the previous of muscle atrophy, and the promotion of neuronal survival and growth. Based on preclinical studies, increasing HGF protein production has been shown to reduce neurodegeneration, thus potentially halting or slowing ALS progression.31 Currently, the safety of engensis is being evaluated in ALS patients (18-80 years of age) in the REViVALS phase 2a (ClinicalTrials.gov Identifier: NCT04632225)/2b (ClinicalTrial.gov Identifier: NCT05176093).32,33 The ReViVALS trial is a double-blind, randomized, placebo-controlled, multi-center study. The phase 2a study endpoints include the incidence of TEAEs, treatment-emergent serious adverse events (TESAEs), injection site reactions, and clinically significant labor-atory values post-treatment (engensis vs placebo group) for 180 days.33 A phase 2b study will evaluate the long-term safety of engensis for an additional 6 months. Study endpoints include the incidence of AEs, changes from baseline in ALSFRS-R scores to evaluate improvement in muscle function, changes from baseline in quality of life using the ALS patient assessment questionnaire, time to all-cause mortality compared to placebo, etc.32
Spinal muscular atrophy
SMA is a hereditary lower motor-neuron disease caused (in 95% of cases) by deletions or, less commonly, by mutations of the survival motor neuron 1 (SMN1) gene on chromosome 5q13 that encodes the SMN protein.6 Reduction in expression of the SMN protein causes motor neurons to degenerate.36-38 Because of a large inverted duplication in chromosome 5q, two variants of SMN (SMN1 and SMN2) exist on each allele. The paralog gene, SMN2, also produces the SMN protein – although at a lower level (10% to 20% of total SMN protein production) than SMN1 does.
A single nucleotide substitution in SMN2 alters splicing and suppresses transcription of exon 7, resulting in a shortened mRNA strand that yields a truncated SMN protein product.6,37,39 SMA is classified based on age of onset and maximum motor abilities achieved, ranging from the most severe (Type 0) to mildest (Type 4) disease.36,40 Because SMA patients lack functional SMN1 (due to polymorphisms), disease severity is determined by copy numbers of SMN2.6,39
Gene-based therapy for SMA
Three FDA-approved SMN treatments demonstrate clinically meaningful benefit in SMA: SMN2-targeting nusinersen [Spinraza] and risdiplam [Evrysdi], and SMN1-targeting onasemnogene abeparvovec-xioi [Zolgensma]38 Additional approaches to SMA treatment are through SMN-independent therapies, which target muscle and nerve function. Research has strongly suggested that combined SMA therapies, specifically approved SMN-targeted and investigational SMN-independent treatments, such as GYM329 (also known as RO7204239) may be the best strategy to treat all ages, stages, and types of SMA.41 (Table 226-41).
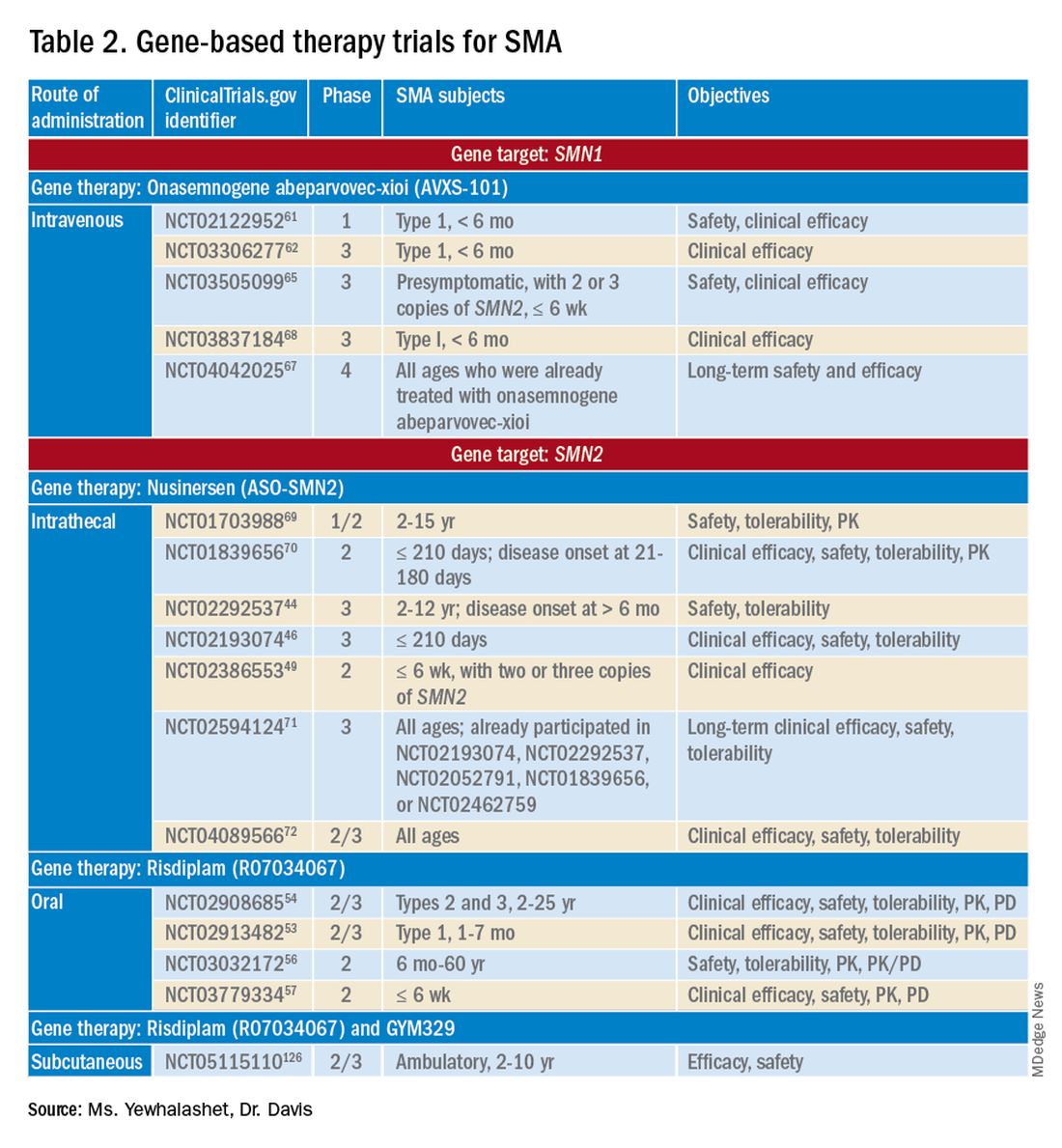
Agents that modulate SMN2. Nusinersen, approved by the FDA in 2016, was the first treatment indicated for all SMA types in pediatric and adult patients.42 The agent is an ASO that targets exon 7 of SMN2, thus stabilizing transcription. Inclusion of exon 7 increases SMN protein production, improving motor function.6,38 Nusinersen is a lifelong treatment that requires IT administration every 4 months because it cannot cross the blood-brain barrier.38,43
Pivotal clinical studies that led to approval of nusinersen include CHERISH (ClinicalTrial.gov Identifier: NCT02292537) and ENDEAR (ClinicalTrial.gov Identifier: NCT02193074) studies.
CHERISH was a phase 3, randomized, double-blind, sham procedure–controlled trial that examined the clinical efficacy and safety of nusinersen in 126 participants with later-onset SMA (2-12 years of age). The primary endpoint was the change from baseline using the Hammersmith Functional Motor Scale Expanded (HFMSE) at 15 months. HFMSE looks at 33 activities to assess improvement in motor function. The study met the primary efficacy outcome, demonstrating statistically significant (P = .0000001) improvement in overall motor function. The nusinersen group showed a 3.9-point increase in the HFMSE score from baseline, which indicates improvement, compared with a 1.0-point decline from baseline in the control group.46,47
ENDEAR was also a randomized, double-blind, sham procedure–controlled phase 3 trial, which investigated the efficacy and safety of nusinersen in 121 participants with early-onset SMA Type 1 (≤ 210 days of age). Coprimary endpoints were:
- Percentage of motor milestones responders, as determined using Section 2 of the Hammersmith Infant Neurological Examination–Part 2.
- Event-free survival (that is, avoidance of combined endpoint of death or permanent ventilation).
ENDEAR met the first primary efficacy outcome, demonstrating statistically significant (P < .0001) improvement in motor milestones (head control, rolling, independent sitting, and standing). By 13 months of age, approximately 51% of nusinersen-treated participants showed improvement, compared with none in the control group.46,47
The second primary endpoint was also met, with a statistically significant (P = .005) 47% decrease in mortality or permanent ventilation use.46-48
The NURTURE (ClinicalTrial.gov Identifier: NCT02386553) study is also investigating the efficacy and safety of nusinersen. An ongoing, open-label, supportive phase 2 trial, NURTURE is evaluating the efficacy and safety of multiple doses of nusinersen in 25 presymptomatic SMA patients (≤ 6 weeks of age). The primary endpoint of this study is time to death or respiratory intervention.49 Interim results demonstrate that 100% of presymptomatic infants are functioning without respiratory intervention after median follow-up of 2.9 years.46-48
Although nusinersen has been shown to be generally safe in clinical studies, development of lumbar puncture–related complications, as well as the need for sedation during IT administration, might affect treatment tolerability in some patients.39
Risdiplam was approved by the FDA in 2020 as the first orally administered small-molecule treatment of SMA (for patients ≤ 2 months of age).52 Risdiplam is a SMN2 splicing modifier, binding to the 5’ splice site of intron 7 and exonic splicing enhancer 2 in exon 7 of SMN2 pre-mRNA. This alternative splicing increases efficiency in SMN2 gene transcription, thus increasing SMN protein production in motor-neuron cells.36 An important advantage of risdiplam is the convenience of oral administration: A large percentage of SMA patients (that is, those with Type 2 disease) have severe scoliosis, which can further complicate therapy or deter patients from using a treatment that is administered through the IT route.40
FDA approval of risdiplam was based on clinical data from two pivotal studies, FIREFISH (ClinicalTrial.gov Identifier: NCT02913482) and SUNFISH (ClinicalTrial.gov Identifier: NCT02908685).53-54
FIREFISH is an open-label, phase 2/3 ongoing trial in infants (1-7 months of age) with SMA Type 1. The study comprises two parts; Part 1 determined the dose of risdiplam used in Part 2, which assessed the efficacy and safety of risdiplam for 24 months. The primary endpoint was the percentage of infants sitting without support for 5 seconds after 12 months of treatment using the gross motor scale of the Bayley Scales of Infant and Toddler Development–Third Edition. A statistically significant (P < .0001) therapeutic benefit was observed in motor milestones. Approximately 29% of infants achieved the motor milestone of independent sitting for 5 seconds, which had not been observed in the natural history of SMA.53-55
SUNFISH is an ongoing randomized, double-blind, placebo-controlled trial of risdiplam in adult and pediatric patients with SMA Types 2 and 3 (2-25 years old). This phase 2/3 study comprises two parts: Part 1 determined the dose (for 12 weeks) to be used for confirmatory Part 2 (for 12 to 24 months). The primary endpoint was the change from baseline on the 32-item Motor Function Measure at 12 months. The study met its primary endpoint, demonstrating statistically significant (P = .0156) improvement in motor function scores, with a 1.36-point increase in the risdiplam group, compared with a 0.19-point decrease in the control group.54,55
Ongoing risdiplam clinical trials also include JEWELFISH (ClinicalTrial.gov Identifier: NCT03032172) and RAINBOW (ClinicalTrial.gov Identifier: NCT03779334).56-57 JEWELFISH is an open-label, phase 2 trial assessing the safety of risdiplam in patients (6 months to 60 years old) who received prior treatment. The study has completed recruitment; results are pending.56 RAINBOW is an ongoing, open-label, single-arm, phase 2 trial, evaluating the clinical efficacy and safety of risdiplam in SMA-presymptomatic newborns (≤ 6 weeks old). The study is open for enrollment.57 Overall, interim results for JEWELFISH and RAINBOW appear promising.
In addition, combined SMA therapies, specifically risdiplam and GYM329 are currently being investigated to address the underlying cause and symptoms of SMA concurrently.58 GYM329, is an investigational anti-myostatin antibody, selectively binding preforms of myostatin - pro-myostatin and latent myostatin, thus improving muscle mass and strength for SMA patients.59 The safety and efficacy of GYM329 in combination with risdiplam is currently being investigated in 180 ambulant participants with SMA (2-10 years of age) in the MANATEE (ClinicalTrial.gov Identifier: NCT05115110) phase 2/3 trial. The MANATEE study is a two-part, seamless, randomized, placebo-controlled, double-blind trial. Part 1 will assess the safety of the combination treatment in approximately 36 participants; participants will receive both GYM329 (every 4 weeks) by subcutaneous (SC) injection into the abdomen and risdiplam (once per day) for 24 weeks followed by a 72-week open-label treatment period. 54,58 The outcome measures include the incidence of AEs, percentage change from baseline in the contractile area of skeletal muscle (in dominant thigh and calf), change from baseline in RHS total score, and incidence of change from baseline in serum concentration (total myostatin, free latent myostatin, and mature myostatin) etc.54 Part 2 will be conducted on 144 participants, specifically assessing the efficacy and safety of the optimal dose of GYM329 selected from Part 1 (combined with risdiplam) for 72 weeks. Once the treatment period is completed in either part, participants can partake in a 2-year open-label extension period.54,58 Other outcome measures include change from baseline in lean muscle mass (assessed by full body dual-energy X- ray absorptiometry (DXA) scan), in time taken to walk/run 10 meters (measured by RHS), in time taken to rise from the floor (measured by RHS), etc.54 Overall, this combination treatment has the potential to further improve SMA patient outcomes and will be further investigated in other patient populations (including non-ambulant patients and a broader age range) in the future.58
An agent that alters SMN1 expression. Onasemnogene abeparvovec-xioi, FDA approved in 2019, was the first gene-replacement therapy indicated for treating SMA in children ≤ 2 years old.60 Treatment utilizes an AAV vector type 9 (AAV9) to deliver a functional copy of SMN1 into target motor-neuron cells, thus increasing SMN protein production and improving motor function. This AAV serotype is ideal because it crosses the blood-brain barrier. Treatment is administered as a one-time IV fusion.38,39,43
FDA approval was based on the STR1VE (ClinicalTrial.gov Identifier: NCT03306277) phase 3 study and START (ClinicalTrial.gov Identifier: NCT02122952) phase 1 study.61,62 START was the first trial to investigate the safety and efficacy of onasemnogene abeparvovec-xioi in SMA Type 1 infants (< 6 months old). Results demonstrated remarkable clinical benefit, including 100% permanent ventilation-free survival and a 92% (11 of 12 patients) rate of improvement in motor function. Improvement in development milestones was also observed: 92% (11 of 12 patients) could sit without support for 5 seconds and 75% (9 of 12) could sit without support for 30 seconds.14,61,63
The efficacy of onasemnogene abeparvovec-xioi seen in STR1VE was consistent with what was observed in START. STRIVE, a phase 3 open-label, single-dose trial, examined treatment efficacy and safety in 22 symptomatic infants (< 6 months old) with SMA Type 1 (one or two SMN2 copies). The primary endpoint was 30 seconds of independent sitting and event-free survival. Patients were followed for as long as 18 months. Treatment showed statistically significant (P < .0001) improvement in motor milestone development and event-free survival, which had not been observed in SMA Type 1 historically. Approximately 59% (13 of 22 patients) could sit independently for 30 seconds at 18 months of age. At 14 months of age, 91% (20 of 22 patients) were alive and achieved independence from ventilatory support.34,35,53
Although many clinical studies suggest that onasemnogene abeparvovec-xioi can slow disease progression, the benefits and risks of long-term effects are still unknown. A 15-year observational study is investigating the long-term therapeutic effects and potential complications of onasemnogene abeparvovec-xioi. Participants in START were invited to enroll in this long-term follow-up study (ClinicalTrial.gov Identifier: NCT04042025).66-67
Duchenne muscular dystrophy
DMD is the most common muscular dystrophy of childhood. With an X-linked pattern of inheritance, DMD is seen mostly in young males (1 in every 3,500 male births).38,39,73 DMD is caused by mutation of the dystrophin encoding gene, or DMD, on the X chromosome. Deletion of one or more exons of DMD prevents production of the dystrophin protein, which leads to muscle degeneration.38,39,43 Common DMD deletion hotspots are exon 51 (20% of cases), exon 53 (13% of cases), exon 44 (11% of cases), and exon 45 (12% of cases).74 Nonsense mutations, which account for another 10% of DMD cases, occur when premature termination codons are found in the DMD gene. Those mutations yield truncated dystrophin protein products.39,66
Therapy for DMD
There are many therapeutic options for DMD, including deflazacort (Emflaza), FDA approved in 2017, which has been shown to reduce inflammation and immune system activity in DMD patients (≥ 5 years old). Deflazacort is a corticosteroid prodrug; its active metabolite acts on the glucocorticoid receptor to exert anti-inflammatory and immunosuppressive effects. Studies have shown that muscle strength scores over 6-12 months and average time to loss of ambulation numerically favored deflazacort over placebo.74,75
Gene-based therapy for DMD
Mutation-specific therapeutic approaches, such as exon skipping and nonsense suppression, have shown promise for the treatment of DMD (Table 358-79):
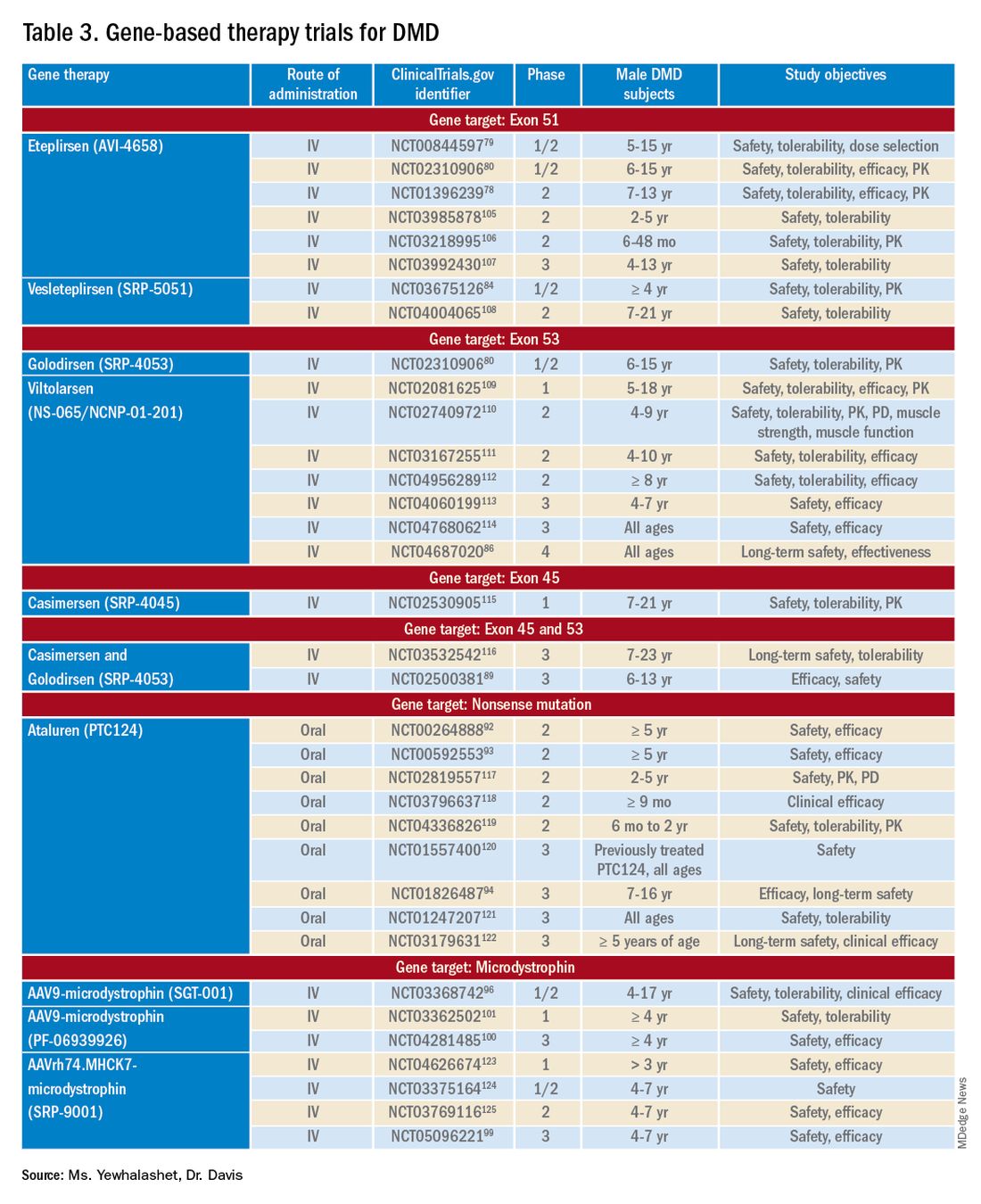
- ASO-mediated exon skipping allows one or more exons to be omitted from the mutated DMD mRNA.74,75 Effective FDA-approved ASOs include golodirsen [Vyondys 53], viltolarsen [Viltepso], and casimersen [Amondys 45].74
- An example of therapeutic suppression of nonsense mutations is ataluren [Translarna], an investigational agent that can promote premature termination codon read-through in DMD patients.66
Another potential treatment approach is through the use of AAV gene transfer to treat DMD. However, because DMD is too large for the AAV vector (packaging size, 5.0 kb), microdystrophin genes (3.5-4 kb, are used as an alternative to fit into a single AAV vector.39,76
Exon skipping targeting exon 51. Eteplirsen, approved in 2016, is indicated for the treatment of DMD patients with the confirmed DMD gene mutation that is amenable to exon 51 skipping. Eteplirsen binds to exon 51 of dystrophin pre-mRNA, causing it to be skipped, thus, restoring the reading frame in patients with DMD gene mutation amenable to exon 51 skipping. This exclusion promotes dystrophin production. Though the dystrophin protein is still functional, it is shortened.38,77 Treatment is administered IV, once a week (over 35-60 minutes). Eteplirsen’s accelerated approval was based on 3 clinical studies (ClinicalTrial.gov Identifier: NCT01396239, NCT01540409, and NCT00844597.) 78-81 The data demonstrated an increased expression of dystrophin in skeletal muscles in some DMD patients treated with eteplirsen. Though the clinical benefit of eteplirsen (including improved motor function) was not established, it was concluded by the FDA that the data were reasonably likely to predict clinical benefit. Continued approval for this indication may depend on the verification of a clinical benefit in confirmatory trials. Ongoing clinical trials include (ClinicalTrial.gov Identifier: NCT03992430 (MIS51ON), NCT03218995, and NCT03218995).77,81,82
Vesleteplirsen, is an investigational agent that is designed for DMD patients who are amendable to exon 51 skip-ping. The mechanism of action of vesleteplirsen appears to be similar to that of eteplirsen.83 The ongoing MOMENTUM (ClinicalTrial.gov Identifier: NCT04004065) phase 2 trial is assessing the safety and tolerability of vesleteplirsen at multiple-ascending dose levels (administered via IV infusion) in 60 participants (7-21 years of age). The study consists of two parts; participants receive escalating dose levels of vesleteplirsen (every 4 weeks) for 72 weeks during part A and participants receive the selected doses from part A (every 4 weeks) for 2 years during part B. Study endpoints include the number of AEs (up to 75 weeks) and the change from baseline to week 28 in dystrophin protein level. 84 Serious AEs of reversible hypomagnesemia were observed in part B, and as a result, the study protocol was amended to include magnesium supplementation and monitoring of magnesium levels.83
Exon skipping targeting exon 53. Golodirsen, FDA approved in 2019, is indicated for the treatment of DMD in patients who have a confirmed DMD mutation that is amenable to exon 53 skipping. The mechanism of action is similar to eteplirsen, however, golodirsen is designed to bind to exon 53.38,39 Treatment is administered by IV infusion over 35-60 minutes.
Approval of golodirsen was based primarily on a two-part, phase 1/2 clinical trial (ClinicalTrial.gov Identifier: NCT02310906). Part 1 was a randomized, placebo-controlled, dose-titration study that assessed multiple-dose efficacy in 12 DMD male patients, 6 to 15 years old, with deletions that were amenable to exon 53 skipping.
Part 2 was an open-label trial in 12 DMD patients from Part 1 of the trial plus 13 newly enrolled male DMD patients who were also amenable to exon 53 skipping and who had not already received treatment. Primary endpoints were change from baseline in total distance walked during the 6-minute walk test at Week 144 and dystrophin protein levels (measured by western blot testing) at Week 48. A statistically significant increase in the mean dystrophin level was observed, from a baseline 0.10% mean dystrophin level to a 1.02% mean dystrophin level after 48 weeks of treatment (P < .001). Common reported adverse events associated with golodirsen were headache, fever, abdominal pain, rash, and dermatitis. Renal toxicity was observed in preclinical studies of golodirsen but not in clinical studies.80,85
Viltolarsen, approved in 2020, is also indicated for the treatment of DMD in patients with deletions amenable to exon 53 skipping. The mechanism of action and administration (IV infusion over 60 minutes) are similar to that of golodirsen.
Approval of viltolarsen was based on two phase 2 clinical trials (ClinicalTrial.gov Identifier: NCT02740972 and NCT03167255) in a total of 32 patients. NCT02740972 was a randomized, double-blind, placebo-controlled, dose-finding study that evaluated the clinical efficacy of viltolarsen in 16 male DMD patients (4-9 years old) for 24 weeks.
NCT03167255 was an open-label study that evaluated the safety and tolerability of viltolarsen in DMD male patients (5-18 years old) for 192 weeks. The efficacy endpoint was the change in dystrophin production from baseline after 24 weeks of treatment. A statistically significant increase in the mean dystrophin level was observed, from a 0.6% mean dystrophin level at baseline to a 5.9% mean dystrophin level at Week 25 (P = .01). The most common adverse events observed were upper respiratory tract infection, cough, fever, and injection-site reaction.86-87
Exon skipping targeting exon 45. Casimersen was approved in 2021 for the treatment of DMD in patients with deletions amenable to exon 45 skipping.88 Treatment is administered by IV infusion over 30-60 minutes. Approval was based on an increase in dystrophin production in skeletal muscle in treated patients. Clinical benefit was reported in interim results from the ESSENCE (ClinicalTrial.gov Identifier: NCT02500381) study, an ongoing double-blind, placebo-controlled phase 3 trial that is evaluating the efficacy of casimersen, compared with placebo, in male participants (6-13 years old) for 48 weeks. Efficacy is based on the change from baseline dystrophin intensity level, determined by immunohistochemistry, at Week 48.
Interim results from ESSENCE show a statistically significant increase in dystrophin production in the casimersen group, from a 0.9% mean dystrophin level at baseline to a 1.7% mean dystrophin level at Week 48 (P = .004); in the control group, a 0.54% mean dystrophin level at baseline increased to a 0.76% mean dystrophin level at Week 48 (P = .09). Common adverse events have included respiratory tract infection, headache, arthralgia, fever, and oropharyngeal pain. Renal toxicity was observed in preclinical data but not in clinical studies.60,84
Targeting nonsense mutations. Ataluren is an investigational, orally administered nonsense mutation suppression therapy (through the read-through of stop codons).37 Early clinical evidence supporting the use of ataluren in DMD was seen in an open-label, dose-ranging, phase 2a study (ClinicalTrial.gov Identifier: NCT00264888) in male DMD patients (≥ 5 years old) caused by nonsense mutation. The study demonstrated a modest (61% ) increase in dystrophin expression in 23 of 38 patients after 28 days of treatment.37,91,92
However, a phase 2b randomized, double-blind, placebo-controlled trial (ClinicalTrial.gov Identifier: NCT00592553) and a subsequent confirmatory ACT DMD phase 3 study (ClinicalTrial.gov Identifier: NCT01826487) did not meet their primary endpoint of improvement in ambulation after 48 weeks as measured by the 6-minute walk test.37,93,94 In ACT DMD, approximately 74% of the ataluren group did not experience disease progression, compared with 56% of the control group (P = 0386), measured by a change in the 6-minute walk test, which assessed ambulatory decline.37,95
Based on limited data showing that ataluren is effective and well tolerated, the European Medicines Agency has given conditional approval for clinical use of the drug in Europe. However, ataluren was rejected by the FDA as a candidate therapy for DMD in the United States.22 Late-stage clinical studies of ataluren are ongoing in the United States.
AAV gene transfer with microdystrophin. Limitations on traditional gene-replacement therapy prompted exploration of gene-editing strategies for treating DMD, including using AAV-based vectors to transfer microdystrophin, an engineered version of DMD, into target muscles.43 The microdystrophin gene is designed to produce a functional, truncated form of dystrophin, thus improving muscular function.
There are 3 ongoing investigational microdystrophin gene therapies that are in clinical development (ClinicalTrial.gov Identifier: NCT03368742 (IGNITE DMD), NCT04281485 (CIFFREO), and NCT05096221 (EMBARK)).38,82
IGNITE DMD is a phase 1/2 randomized, controlled, single-ascending dose trial evaluating the safety and efficacy of a SGT-001, single IV infusion of AAV9 vector containing a microdystrophin construct in DMD patients (4-17 years old) for 12 months. At the conclusion of the trial, treatment and control groups will be followed for 5 years. The primary efficacy endpoint is the change from baseline in microdystrophin protein production in muscle-biopsy material, using western blot testing.96 Long-term interim data on biopsy findings from three patients demonstrated clinical evidence of durable microdystrophin protein expression after 2 years of treatment.96,97
The CIFFREO trial will assess the safety and efficacy of the PF-06939926 microdystrophin gene therapy, an investigational AAV9 containing microdystrophin, in approximately 99 ambulatory DMD patients (4-7 years of age). The study is a randomized, double-blind, placebo-controlled, multicenter phase 3 trial. The primary efficacy end-point is the change from baseline in the North Star Ambulatory Assessment (NSAA), which measures gross motor function. This will be assessed at 52 weeks; all study participants will be followed for a total of 5 years post-treatment.98,99,100 Due to unexpected patient death (in a non-ambulatory cohort) in the phase 1b (in a non-ambulatory cohort) in the phase 1b (ClinicalTrial.gov Identifier: (NCT03362502) trial, microdystrophin gene therapy was immediately placed on clinical hold.101,102 The amended study protocol required that all participants undergo one week of in-hospital observation after receiving treatment.102
The EMBARK study is a global, randomized, double-blind, placebo-controlled, phase 3 trial that is evaluating the safety and efficacy of SRP-9001, which is a rAAVrh74.MHCK7.microdystrophin gene therapy. The AAV vector (rAAVrh74) contains the microdystrophin construct, driven by the skeletal and cardiac muscle–specific promoter, MHCK7.98,99 In the EMBARK study, approximately 120 participants with DMD (4-7 years of age) will be enrolled. The primary efficacy endpoint includes the change from baseline to week 52 in the NSAA total score.99 Based on SRP-9001, data demonstrating consistent statistically significant functional improvements in NSAA total scores and timed function tests (after one-year post- treatment) in DMD patients from previous studies and an integrated analysis from multiple studies (ClinicalTrial.gov Identifier: NCT03375164, NCT03769116, and NCT04626674), the ongoing EMBARK has great promise.103,104
Challenges ahead, but advancements realized
Novel gene-based therapies show significant potential for transforming the treatment of NMDs. The complex pathologies of NMDs have been a huge challenge to disease management in an area once considered unremediable by gene-based therapy. However, advancements in precision medicine – specifically, gene-delivery systems (for example, AAV9 and AAVrh74 vectors) combined with gene modification strategies (ASOs and AAV-mediated silencing) – have the potential to, first, revolutionize standards of care for sporadic and inherited NMDs and, second, significantly reduce disease burden.6
What will be determined to be the “best” therapeutic approach will, likely, vary from NMD to NMD; further investigation is required to determine which agents offer optimal clinical efficacy and safety profiles.43 Furthermore, the key to therapeutic success will continue to be early detection and diagnosis – first, by better understanding disease pathology and drug targets and, second, by validation of reliable biomarkers that are predictive of therapeutic benefit.4,5
To sum up, development challenges remain, but therapeutic approaches to ALS, SMA, and DMD that utilize novel gene-delivery and gene-manipulation tools show great promise.
Ms. Yewhalashet is a student in the masters of business and science program, with a concentration in healthcare economics, at Keck Graduate Institute Henry E. Riggs School of Applied Life Sciences, Claremont, Calif. Dr. Davis is professor of practice in clinical and regulatory affairs, Keck Graduate Institute Henry E. Riggs School of Applied Life Sciences.
References
1. Aitken M et al. Understanding neuromuscular disease care. IQVIA [Internet]. Oct 30, 2018. Accessed Mar 1, 2022. https://www.iqvia.com/insights/the-iqvia-institute/reports/understanding-neuromuscular-disease-care.
2. National Institute of Neurological Disorders and Stroke. Neurological diagnostic tests and procedures fact sheet. Updated Nov 15, 2021. Ac-cessed Mar 1, 2022. http://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Neurological-Diagnostic-Tests-and-Procedures-Fact.
3. Deenen JCW et al. The epidemiology of neuromuscular disorders: A comprehensive overview of the literature. J Neuromuscul Dis. 2015;2(1):73-85.
4. Cavazzoni P. The path forward: Advancing treatments and cures for neurodegenerative diseases. U.S. Food and Drug Administration. Jul 29, 2021. Accessed Mar 1, 2022. http://www.fda.gov/news-events/congressional-testimony/path-forward-advancing-treatments-and-cures-neurodegenerative-diseases-07292021.
5. Martier R, Konstantinova P. Gene therapy for neurodegenerative diseases: Slowing down the ticking clock. Front Neurosci. 2020 Sep 18;14:580179. doi: 10.3389/fnins.2020.580179.
6. Sun J, Roy S. Gene-based therapies for neurodegenerative diseases. Nat Neurosci. 2021 Mar;24(3):297-311. doi:10.1038/s41593-020-00778-1.
7. Amado DA, Davidson BL. Gene therapy for ALS: A review. Mol Ther. 2021 Dec 1;29(12):3345-58. doi:10.1016/j.ymthe.2021.04.008.
8. Yun Y, Ha Y. CRISPR/Cas9-mediated gene correction to understand ALS. Int J Mol Sci. 2020;21(11):3801. doi:10.3390/ijms21113801.
9. National Institute of Neurological Disorders and Stroke. Amyotrophic lateral sclerosis (ALS) fact sheet. Updated Nov 15, 2021. Accessed Mar 1, 2022. http://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Amyotrophic-Lateral-Sclerosis-ALS-Fact-Sheet.
10. Cappella M et al. Gene therapy for ALS – A perspective. Int J Mol Sci. 2019;20(18):4388. doi:10.3390/ijms20184388.
11. Abramzon YA, Fratta P, Traynor BJ, Chia R. The Overlapping Genetics of Amyotrophic Lateral Sclerosis and Frontotemporal Dementia. Front Neurosci. 2020;14. Accessed August 18, 2022. https://www.frontiersin.org/articles/10.3389/fnins.2020.00042
12. Giannini M, Bayona-Feliu A, Sproviero D, Barroso SI, Cereda C, Aguilera A. TDP-43 mutations link Amyotrophic Lateral Sclerosis with R-loop homeostasis and R loop-mediated DNA damage. PLOS Genet. 2020;16(12):e1009260. doi:10.1371/journal.pgen.1009260
13. FDA-approved drugs for treating ALS. The ALS Association [Internet]. Accessed Mar 1, 2022. http://www.als.org/navigating-als/living-with-als/fda-approved-drugs.
14. Jensen TL et al. Current and future prospects for gene therapy for rare genetic diseases affecting the brain and spinal cord. Front Mol Neurosci. 2021 Oct 6;14:695937. doi:10.3389/fnmol.2021.695937.
15. ALS Gene Targeted Therapies. The ALS Association. Accessed August 22, 2022. https://www.als.org/understanding-als/who-gets-als/genetic-testing/als-gene-targeted-therapies
16. Tofersen for ALS clears phase 1/2 trial, now in phase 3. Advances in Motion. Massachusetts General Hospital [Internet]. Sep 30, 2020. Accessed Mar 1, 2022. https://advances.massgeneral.org/neuro/journal.aspx?id=1699.17. Biogen. A study to evaluate the efficacy, safety, tol-erability, pharmacokinetics, and pharmacodynamics of BIIB067 administered to adult subjects with amyotrophic lateral sclerosis and confirmed superoxide dismutase 1 mutation. ClinicalTrials.gov Identifier: NCT02623699. Updated Jul 25, 2021. Accessed Feb 17, 2022. https://clinicaltrials.gov/ct2/show/NCT02623699.
18. Biogen. Biogen announces topline results from the tofersen phase 3 study and its open-label Extension in SOD1-ALS. Press release. Oct 17, 2021. Accessed Mar 1, 2022. https://investors.biogen.com/news-releases/news-release-details/biogen-announces-topline-results-tofersen-phase-3-study-and-its.
19. Biogen. An extension study to assess the long-term safety, tolerability, pharmacokinetics, and effect on disease progression of BIIB067 ad-ministered to previously treated adults with amyotrophic lateral sclerosis caused by superoxide dismutase 1 mutation. ClinicalTrials.gov Identi-fier: NCT03070119. Updated Sep 10, 2021. Accessed Feb 17, 2022. https://clinicaltrials.gov/ct2/show/NCT03070119.
20. MS MW. #AANAM – ATLAS Trial to Assess Tofersen in Presymptomatic SOD1 ALS. Accessed February 19, 2022. https://alsnewstoday.com/news-posts/2021/04/23/aanam-atlas-clinical-trial- tofersen-presymptomatic-sod1-als-patients/
21.Biogen. A phase 3 randomized, placebo-controlled trial with a longitudinal natural history run-in and open-label extension to evaluate BIIB067 initiated in clinically presymptomatic adults with a confirmed superoxide dismutase 1 mutation. ClinicalTrials.gov Identifier: NCT04856982. Updated Feb 18, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04856982.
22. Latozinemab | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/latozinemab
23. Alector Presents AL001 (latozinemab) Data from the FTD-C9orf72 Cohort of the INFRONT-2 Phase 2 Clinical Trial | Alector. Accessed August 18, 2022. https://investors.alector.com/news- releas-es/news-release-details/alector-presents-al001-latozinemab-data-ftd-c9orf72-cohort/
24. Alector Announces First Participant Dosed in Phase 2 Study Evaluating AL001 in Amyotrophic Lateral Sclerosis (ALS) | Alector. Accessed August 18, 2022. https://investors.alector.com/news- releases/news-release-details/lector-announces-first-participant-dosed-phase-2-study-0/ 25. A Phase 2 Study to Evaluate AL001 in C9orf72-Associated ALS - Full Text View - ClinicalTrials.gov. Accessed August 19, 2022. https://clinicaltrials.gov/ct2/show/NCT05053035
26.TPN-101 | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/tpn- 101
27. Transposon Therapeutics, Inc. A Phase 2a Study of TPN-101 in Patients With Amyotrophic Lateral Sclerosis (ALS) and/or Frontotemporal Dementia (FTD) Associated With Hexanucleotide Repeat Expansion in the C9orf72 Gene (C9ORF72 ALS/FTD). clinicaltrials.gov; 2022. Ac-cessed August 17, 2022. https://clinicaltrials.gov/ct2/show/NCT04993755
28. Kerk SY, Bai Y, Smith J, et al. Homozygous ALS-linked FUS P525L mutations cell- autonomously perturb transcriptome profile and chem-oreceptor signaling in human iPSC microglia. Stem Cell Rep. 2022;17(3):678-692. doi:10.1016/j.stemcr.2022.01.004
29. ION363 | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/ion363 30. Ionis Pharmaceuticals, Inc. A Phase 1-3 Study to Evaluate the Efficacy, Safety, Pharmacokinetics and Pharmacodynamics of Intrathecally Administered ION363 in Amyo-trophic Lateral Sclerosis Patients With Fused in Sarcoma Mutations (FUS-ALS). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04768972
31. PhD LF. Engensis (VM202) - ALS News Today. Accessed August 19, 2022. https://alsnewstoday.com/vm202/
32. Helixmith Co., Ltd. A 6-Month Extension Study Following Protocol VMALS-002-2 (A Phase 2a, Double-Blind, Randomized, Place-bo-Controlled, Multicenter Study to Assess the Safety of Engensis in Participants With Amyotrophic Lateral Sclerosis). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT05176093 33. Safety of Engensis in Participants With Amyotrophic Lateral Sclerosis - Full Text View - ClinicalTrials.gov. Accessed August 19, 2022. https://clinicaltrials.gov/ct2/show/NCT04632225
34. Biogen. A phase 1, safety, tolerability, and distribution study of a microdose of radiolabeled BIIB067 co-administered with BIIB067 to healthy adults. ClinicalTrials.gov Identifier: NCT03764488. Updated Jul 19, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03764488.
35. Ionis Pharmaceuticals Inc. A phase 1, double-blind, placebo-controlled, dose-escalation study of the safety, tolerability, and pharmacokinet-ics of ISIS 333611 administered intrathecally to patients with familial amyotrophic lateral sclerosis due to superoxide dismutase 1 gene muta-tions. ClinicalTrials.gov Identifier: NCT01041222. Updated Apr 13, 2012. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01041222.
36. Messina S, Sframeli M. New treatments in spinal muscular atrophy: Positive results and new challenges. J Clin Med. 2020;9(7):2222. doi:10.3390/jcm9072222.
37. Scoto M et al. Genetic therapies for inherited neuromuscular disorders. Lancet Child Adolesc Health. 2018 Aug;2(8):600-9. doi:10.1016/S2352-4642(18)30140-8.
38. Abreu NJ, Waldrop MA. Overview of gene therapy in spinal muscular atrophy and Duchenne muscular dystrophy. Pediatr Pulmonol. 2021 Apr;56(4):710-20. doi:10.1002/ppul.25055.
39. Brandsema J, Cappa R. Genetically targeted therapies for inherited neuromuscular disorders. Practical Neurology [Internet]. Jul/Aug 2021:69-73. Accessed Mar 1, 2022. https://practicalneurology.com/articles/2021-july-aug/genetically-targeted-therapies-for-inherited-neuromuscular-disorders/pdf.
40. Ojala KS et al. In search of a cure: The development of therapeutics to alter the progression of spinal muscular atrophy. Brain Sci. 2021;11(2):194. doi:10.3390/brainsci11020194.
41. McCall S. Cure SMA Releases Updated Drug Pipeline. Cure SMA. Published December 13, 2021. Accessed August 21, 2022. https://www.curesma.org/cure-sma-releases-updated-drug-pipeline- 2021/ 42. FDA approves first drug for spinal muscular atrophy. U.S. Food and Drug Administration. News release. Dec 23, 2016. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-spinal-muscular-atrophy.43. Kirschner J. Postnatal gene therapy for neuromuscular diseases – Opportunities and limitations. J Perinat Med. 2021 Sep;49(8):1011-5. doi:10.1515/jpm-2021-0435.
43. Terryn J, Verfaillie CM, Van Damme P. Tweaking Progranulin Expression: Therapeutic Avenues and Opportunities. Front Mol Neurosci. 2021;14. Accessed September 4, 2022. https://www.frontiersin.org/articles/10.3389/fnmol.2021.71303144.
44. Biogen. A phase 3, randomized, double-blind, sham-procedure controlled study to assess the clinical efficacy and safety of ISIS 396443 administered intrathecally in patients with later-onset spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02292537. Updated Feb 17, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/study/NCT02292537.
45. Why Spinraza/later-onset studies. SPINRAZA® (nusinersen) [Internet]. Accessed Mar 1, 2022. www.spinraza.com/en_us/home/why-spinraza/later-onset-studies.html#scroll-tabs.
46. Biogen. A Phase 3, Randomized, Double-Blind, Sham-Procedure Controlled Study to Assess the Clinical Efficacy and Safety of ISIS 396443 Administered Intrathecally in Patients With Infantile- Onset Spinal Muscular Atrophy. clinicaltrials.gov; 2021. Accessed February 10, 2022. https://clinicaltrials.gov/ct2/show/results/NCT02193074
47. Early-onset SMA (Type 1) | SPINRAZA® (nusinersen). Accessed Mar 1, 2022. https://www.spinraza-hcp.com/en_us/home/why-spinraza/about-spinraza.html.
48. Finkel RS et al; ENDEAR Study Group. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N Engl J Med. 2017;377(18):1723-32. doi: 10.1056/NEJMoa1702752.
49. Biogen. An open-label study to assess the efficacy, safety, tolerability, and pharmacokinetics of multiple doses of ISIS 396443 delivered intrathecally to subjects with genetically diagnosed and presymptomatic spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02386553. Updated Nov 18, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02386553.
50. De Vivo DC et al; NURTURE Study Group. Nusinersen initiated in infants during the presymptomatic stage of spinal muscular atrophy: In-terim efficacy and safety results from the phase 2 NURTURE study. Neuromuscul Disord. 2019 Nov;29(11):842-56. doi:10.1016/j.nmd.2019.09.007.
51. Why Spinraza/presymptomatic study. SPINRAZA® (nusinersen) [Internet]. Accessed Feb 22, 2022. www.spinraza.com/en_us/home/why-spinraza/presymptomatic-study.html#scroll-tabs.
52. FDA approves oral treatment for spinal muscular atrophy. U.S. Food and Drug Administration. News release. Aug 7, 2020. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-oral-treatment-spinal-muscular-atrophy.
53. Hoffmann-La Roche. A two-part seamless, open-label, multicenter study to investigate the safety, tolerability, pharmacokinetics, pharmaco-dynamics and efficacy of risdiplam (RO7034067) in infants with type 1 spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02913482. Updated Jan 21, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02913482.
54. Hoffmann-La Roche. A two-part seamless, multi-center randomized, placebo-controlled, double-blind study to investigate the safety, tolera-bility, pharmacokinetics, pharmacodynamics and efficacy of risdiplam (RO7034067) in type 2 and 3 spinal muscular atrophy patients. Clinical-Trials.gov Identifier: NCT02908685. Updated Dec 28, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02908685.
55. Genentech. Genentech’s risdiplam shows significant improvement in survival and motor milestones in infants with type 1 spinal muscular atrophy (SMA). Press release. Apr 27, 2020. Accessed Mar 1, 2022. http://www.gene.com/media/press-releases/14847/2020-04-27/genentechs-risdiplam-shows-significant-i
56. Hoffmann-La Roche. An open-label study to investigate the safety, tolerability, and pharmacokinetics/pharmacodynamics of risdiplam (RO7034067) in adult and pediatric patients with spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT03032172. Updated Jan 27, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03032172.
57. Hoffmann-La Roche. An open-label study of risdiplam in infants with genetically diagnosed and presymptomatic spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT03779334. Updated Jan 27, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03779334.
58. McCall S. Update on Genentech/Roche Initiation of MANATEE Clinical Study. Cure SMA. Published October 20, 2021. Accessed August 20, 2022. https://www.curesma.org/update-on- genentech-roche-initiation-of-manatee-clinical-study/
59. Abati E, Manini A, Comi GP, Corti S. Inhibition of myostatin and related signaling pathways for the treatment of muscle atrophy in motor neuron diseases. Cell Mol Life Sci. 2022;79(7):374. doi:10.1007/s00018-022-04408-w
60. FDA approves innovative gene therapy to treat pediatric patients with spinal muscular atrophy, a rare disease and leading genetic cause of infant mortality. U.S. Food and Drug Administration. News release. May 24, 2019. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-innovative-gene-therapy-treat-pediatric-patients-spinal-muscular-atrophy-rare-disease.
61. Novartis Gene Therapies. Phase I gene transfer clinical trial for spinal muscular atrophy type 1 delivering AVXS-101. ClinicalTrials.gov Identifier: NCT02122952. Updated Jun 14, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02122952.
62. Novartis Gene Therapies. Phase 3, open-label, single-arm, single-dose gene replacement therapy clinical trial for patients with spinal mus-cular atrophy type 1 with one or two SMN2 copies delivering AVXS-101 by intravenous infusion. ClinicalTrials.gov Identifier: NCT03306277. Updated Jun 14, 2021. Accessed Feb 21, 2022. https://clinicaltrials.gov/ct2/show/NCT03306277.
63. Mendell JR et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N Engl J Med. 2017;377(18):1713-22. doi:10.1056/NEJMoa1706198.
64. Symptomatic study results. ZOLGENSMA [Internet]. Updated Nov 2021. Accessed Mar 1, 2022. Error! Hyperlink reference not valid..
65. Novartis Gene Therapies. A global study of a single, one-time dose of AVXS-101 delivered to infants with genetically diagnosed and pre-symptomatic spinal muscular atrophy with multiple copies of SMN2. ClinicalTrials.gov Identifier: NCT03505099. Updated Jan 1, 2022. Ac-cessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03505099.
66. Chiu W et al. Current genetics and potential gene-targeting therapeutics for neuromuscular diseases. Int J Mol Sci. 2020 Dec;21(24):9589. doi:10.3390/ijms21249589.
67. Novartis Gene Therapies. A long-term follow-up study of patients in the clinical trials for spinal muscular atrophy receiving AVXS-101. Clini-calTrials.gov Identifier: NCT04042025. Updated Jun 9, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04042025.
68. Novartis Gene Therapies. Phase 3, open-label, single-arm, single-dose gene replacement therapy clinical trial for patients with spinal mus-cular atrophy type 1 with one or two SMN2 copies delivering AVXS-101 by intravenous infusion. ClinicalTrials.gov Identifier: NCT0383718. Up-dated Jan 11, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03837184.
69. Biogen. An open-label, dose escalation study to assess the safety, tolerability and dose-range finding of multiple doses of ISIS 396443 de-livered intrathecally to patients with spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT01703988. Updated Apr 13, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01703988.
70. Biogen. A study to assess the efficacy, safety, tolerability, and pharmacokinetics of multiple doses of ISIS 396443 delivered intrathecally to patients with infantile-onset spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT01839656. Updated Feb 17, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01839656.
71. Biogen. An open-label extension study for patients with spinal muscular atrophy who previously participated in investigational studies of ISIS 396443. ClinicalTrials.gov Identifier: NCT02594124. Updated Nov 15, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02594124.
72. Biogen. Escalating dose and randomized, controlled study of nusinersen (BIIB058) in participants with spinal muscular atrophy. ClinicalTri-als.gov Identifier: NCT04089566. Updated Feb 24, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04089566.
73. National Center for Advancing Translational Sciences. Duchenne muscular dystrophy. Genetic and Rare Diseases Information Center. Up-dated Nov 2, 2020. Accessed Mar 1, 2022. https://rarediseases.info.nih.gov/diseases/6291/duchenne-muscular-dystrophy.
74. Matsuo M. Antisense oligonucleotide-mediated exon-skipping therapies: Precision medicine spreading from Duchenne muscular dystrophy. JMA J. 2021 Jul 15;4(3):232-40. doi:10.31662/jmaj.2021-0019.
75. FDA approves drug to treat Duchenne muscular dystrophy. U.S. Food and Drug Administration. News release. Feb 9, 2017. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-drug-treat-duchenne-muscular-dystrophy.74.
76. Duan D. Dystrophin gene replacement and gene repair therapy for Duchenne muscular dystrophy in 2016: An interview. Hum Gene Ther Clin Dev. 2016 Mar;27(1):9-18. doi:10.1089/humc.2016.001.
77. EXONDYS 51®. Parent Project Muscular Dystrophy. Accessed August 21, 2022. https://www.parentprojectmd.org/drug-development-pipeline/exondys-51/
78. Sarepta Therapeutics, Inc. A Randomized, Double-Blind, Placebo-Controlled, Multiple Dose Efficacy, Safety, Tolerability and Pharmacoki-netics Study of AVI-4658(Eteplirsen),in the Treatment of Ambulant Subjects With Duchenne Muscular Dystrophy. clinicaltrials.gov; 2020. Ac-cessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT01396239
79. Sarepta Therapeutics, Inc. Clinical Study to Assess the Safety Fo AVI-4658 in Subjects With Duchenne Muscular Dystrophy Due to a Frame-Shift Mutation Amenable to Correction by Skipping Exon 51. clinicaltrials.gov; 2015. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/study/NCT00844597
80. Sarepta Therapeutics, Inc. A 2-part, randomized, double-blind, placebo-controlled, dose-titration, safety, tolerability, and pharmacokinetics study (Part 1) followed by an open-label efficacy and safety evaluation (Part 2) of SRP-4053 in patients with Duchenne muscular dystrophy amenable to exon 53 skipping. ClinicalTrials.gov Identifier: NCT02310906. Updated Oct 19, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/results/NCT02310906.
81. Commissioner O of the. FDA grants accelerated approval to first drug for Duchenne muscular dystrophy. FDA. Published March 24, 2020. Accessed August 21, 2022. hDuchenne Muscular Dystrophy Amenable to Exon 51-Skipping Treatment. clinicaltrials.gov; 2022. Accessed Au-gust 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04004065
109. National Center of Neurology and Psychiatry, Japan. Exploratory study of NS-065/NCNP-01 in Duchenne muscular dystrophy. ClinicalTri-als.gov Identifier: NCT02081625; Updated Feb 26, 2020. Accessed Mar 2, 2022. https://clinicaltrialsttps://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-first-drug-duchenne-muscular- dys-trophy
82. Duchenne Drug Development Pipeline. Parent Project Muscular Dystrophy. Accessed August 21, 2022. https://www.parentprojectmd.org/duchenne-drug-development-pipeline/
83. Sarepta Therapeutics Provides Update on SRP-5051 for the Treatment of Duchenne Muscular Dystrophy | Sarepta Therapeutics, Inc. Ac-cessed August 22, 2022. https://investorrelations.sarepta.com/news-releases/news-release-details/sarepta-therapeutics- pro-vides-update-srp-5051-treatment-duchenne
84. Sarepta Therapeutics, Inc. An Open-Label Extension Study for Patients With Duchenne Muscular Dystrophy Who Participated in Studies of SRP-5051. clinicaltrials.gov; 2021. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03675126
85. VYONDYS 53. Prescribing information. Sarepta Therapeutics Inc.; 2019. Accessed Mar 2, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211970s000lbl.pdf.
86. NS Pharma Inc. Long-term use of viltolarsen in boys with Duchenne muscular dystrophy in clinical practice (VILT-502). ClinicalTrials.gov Identifier: NCT04687020. Updated Nov 22, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04687020.
87. VILTEPSO. Prescribing information. NS Pharma; 2020. Accessed Mar 2, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2020/212154s000lbl.pdf.
88. FDA approves targeted treatment for rare Duchenne muscular dystrophy mutation. U.S. Food and Drug Administration. News release. Feb 25, 2021. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-targeted-treatment-rare-duchenne-muscular-dystrophy-mutation-0.
89. Sarepta Therapeutics Inc. A double-blind, placebo-controlled, multi-center study with an open-label extension to evaluate the efficacy and safety of SRP-4045 and SRP-4053 in patients with Duchenne muscular dystrophy. Clinicaltrials.gov Identifier: NCT02500381. Updated Aug 19, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02500381.
90. AMONDYS 45. Prescribing information. Sarepta Therapeutics Inc.; 2021. Accessed Feb 22, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2021/213026lbl.pdf.
91. Finkel RS et al. Phase 2a study of ataluren-mediated dystrophin production in patients with nonsense mutation Duchenne muscular dys-trophy. PLoS ONE. 2013;8(12):e81302. doi:10.1371/journal.pone.0081302.
92. PTC Therapeutics. A phase 2 study of PTC124 as an oral treatment for nonsense-mutation-mediated Duchenne muscular dystrophy. Clini-calTrials.gov Identifier: NCT00264888. Updated Jan 14, 2009. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT00264888.
93. PTC Therapeutics. A phase 2B efficacy and safety study of PTC124 in subjects with nonsense-mutation-mediated Duchenne and Becker muscular dystrophy. ClinicalTrials.gov Identifier: NCT00592553. Updated Apr 7, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT00592553.
94. PTC Therapeutics. A phase 3 efficacy and safety study of ataluren in patients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01826487. Updated Aug 4, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01826487.
95. Bushby K et al; PTC124-GD-007-DMD Study Group. Ataluren treatment of patients with nonsense mutation dystrophinopathy. Muscle Nerve. 2014 Oct;50(4):477-87. doi:10.1002/mus.24332.
96. Solid Biosciences LLC. A randomized, controlled, open-label, single-ascending dose, phase I/II study to investigate the safety and tolerabil-ity, and efficacy of intravenous SGT-001 in male adolescents and children with Duchenne muscular dystrophy. ClinicalTrials.gov Identifier: NCT03368742. Updated Aug 24, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03368742.
97. Solid Biosciences reports 1.5-year data from patients in the ongoing IGNITE DMD phase I/II clinical trial of SGT-001. Press release. Solid Biosciences. Sep 27, 2021. Accessed Mar 2, 2022. http://www.solidbio.com/about/media/press-releases/solid-biosciences-reports-1-5-year-data-from-patients-in-the-ongoing-ignite-dmd-phase-i-ii-clinical-trial-of-sgt-001.
98. Potter RA et al. Dose-escalation study of systemically delivered rAAVrh74.MHCK7.microdystrophin in the mdx mouse model of Duchenne muscular dystrophy. Hum Gene Ther. 2021 Apr;32(7-8):375-89. doi:10.1089/hum.2019.255.
99. Sarepta Therapeutics, Inc. A Phase 3 Multinational, Randomized, Double-Blind, Placebo- Controlled Systemic Gene Delivery Study to Evaluate the Safety and Efficacy of SRP-9001 in Patients With Duchenne Muscular Dystrophy (EMBARK). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT05096221
100. Pfizer. A PHASE 3, MULTICENTER, RANDOMIZED, DOUBLE-BLIND, PLACEBO CONTROLLED STUDY TO EVALUATE THE SAFETY AND EFFICACY OF PF 06939926 FOR THE TREATMENT OF DUCHENNE MUSCULAR DYSTROPHY. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04281485
101. Pfizer. A phase 1B multicenter open-label, single ascending dose study to evaluate the safety and tolerability of PF-06939926 in ambula-tory and non-ambulatory subjects with Duchenne muscular dystrophy. ClinicalTrials.gov Identifier: NCT03362502. Updated Mar 2, 2022. Ac-cessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03362502.
102. MS MW. Phase 3 CIFFREO DMD Gene Therapy Trial Slated to Begin in June in US. Accessed August 21, 2022. https://musculardystrophynews.com/news/phase-3-trial-of-pfizers-gene-therapy- expected-to-open-in-us-in-june/
103. SRP-9001. Parent Project Muscular Dystrophy. Accessed August 22, 2022. https://www.parentprojectmd.org/drug-development-pipeline/srp-9001-micro-dystrophin-gene- transfer/
104. Sarepta Therapeutics’ Investigational Gene Therapy SRP-9001 for Duchenne Muscular Dystrophy Demonstrates Significant Functional Improvements Across Multiple Studies | Sarepta Therapeutics, Inc. Accessed August 22, 2022. https://investorrelations.sarepta.com/news-releases/news-release- details/sarepta-therapeutics-investigational-gene-therapy-srp-9001
105. Sarepta Therapeutics, Inc. An Open-Label Safety, Tolerability, and Efficacy Study of Eteplirsen in Patients With Duchenne Muscular Dys-trophy Who Have Completed Study 4658-102.clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03985878
106. Sarepta Therapeutics, Inc. An Open-Label Safety, Tolerability, and Pharmacokinetics Study of Eteplirsen in Young Patients With Duchenne Mus-cular Dystrophy Amenable to Exon 51 Skipping. clinicaltrials.gov; 2021. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03218995
107.Sarepta Therapeutics, Inc. A Randomized, Double-Blind, Dose Finding and Comparison Study of the Safety and Efficacy of a High Dose of Eteplirsen, Preceded by an Open-Label Dose Escalation, in Patients With Duchenne Muscular Dystrophy With Deletion Mutations Amenable to Exon 51 Skipping. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03992430
108. Sarepta Therapeutics, Inc. A Phase 2, Two-Part, Multiple-Ascending-Dose Study of SRP-5051 for Dose Determination, Then Dose Ex-pansion, in Patients With .gov/ct2/show/NCT02081625.
110. NS Pharma Inc. A phase II, dose finding study to assess the safety, tolerability, pharmacokinetics, and pharmacodynamics of NS-065/NCNP-01 in boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT02740972. Updated Dec 7, 2021. Ac-cessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02740972.
111. NS Pharma Inc. A phase II, open-label, extension study to assess the safety and efficacy of NS-065/NCNP-01 in boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT03167255. Updated Nov 24, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03167255.
112. NS Pharma Inc. A phase 2 open label study to assess the safety, tolerability, and efficacy of viltolarsen in ambulant and non-ambulant boys with Duchenne muscular dystrophy (DMD) compared with natural history controls. ClinicalTrials.gov Identifier: NCT04956289. Updated Feb 1, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04956289.
113. NS Pharma Inc. A phase 3 randomized, double-blind, placebo-controlled, multi-center study to assess the efficacy and safety of viltolarsen in ambulant boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT04060199. Updated Nov 16, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04060199.
114. NS Pharma Inc. A phase 3, multi-center, open-label extension study to assess the safety and efficacy of viltolarsen in ambulant boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT04768062. Updated Nov 16, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04768062.
115. Sarepta Therapeutics Inc. A randomized, double-blind, placebo-controlled, dose-titration, safety, tolerability, and pharmacokinetics study followed by an open-label safety and efficacy evaluation of SRP-4045 in advanced-stage patients with Duchenne muscular dystrophy amena-ble to exon 45 skipping. ClinicalTrials.gov Identifier: NCT02530905. Updated May 17, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02530905.
116. Sarepta Therapeutics Inc. Long-term, open-label extension study for patients with Duchenne muscular dystrophy enrolled in clinical trials evaluating casimersen or golodirsen. ClinicalTrials.gov Identifier: NCT03532542. Updated Dec 20, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03532542.
117. PTC Therapeutics. A phase 2 study of the safety, pharmacokinetics, and pharmacodynamics of ataluren (PTC124®) in patients aged ≥2 to <5 years old with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT02819557. Updated Aug 28, 2020. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02819557.
118. PTC Therapeutics. Phase 2, non-interventional, clinical study to assess dystrophin levels in subjects with nonsense mutation Duchenne muscular dystrophy who have been treated with ataluren for ≥ 9 months. ClinicalTrials.gov Identifier: NCT03796637. Updated Apr 10, 2020. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03796637.
119. PTC Therapeutics. An Open-Label Study Evaluating the Safety and Pharmacokinetics of Ataluren in Children From ≥6 Months to <2 Years of Age With Nonsense Mutation Duchenne Muscular Dystrophy. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04336826 120. PTC Therapeutics. An open-label study for previously treated ataluren (PTC124®) pa-tients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01557400. Updated Nov 25, 2020. Accessed Feb 21, 2022. https://clinicaltrials.gov/ct2/show/NCT01557400.
121. PTC Therapeutics. An open-label, safety study for ataluren (PTC124) patients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01247207. Updated Feb 16, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT01247207.
122. PTC Therapeutics. A phase 3, randomized, double-blind, placebo-controlled efficacy and safety study of ataluren in patients with non-sense mutation Duchenne muscular dystrophy and open-label extension. ClinicalTrials.gov Identifier: NCT03179631. Updated Feb 8, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03179631.
123. Sarepta Therapeutics, Inc. An Open-Label, Systemic Gene Delivery Study Using Commercial Process Material to Evaluate the Safety of and Expression From SRP-9001 in Subjects With Duchenne Muscular Dystrophy (ENDEAVOR). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04626674
124. Sarepta Therapeutics, Inc. Systemic Gene Delivery Phase I/IIa Clinical Trial for Duchenne Muscular Dystrophy Using RAA-Vrh74.MHCK7.Micro-Dystrophin (MicroDys-IV-001). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03375164
125. Sarepta Therapeutics Inc. A multicenter, randomized, double-blind, placebo-controlled trial for Duchenne muscular dystrophy using SRP-9001. ClinicalTrials.gov Identifier: NCT03769116. Updated Dec 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03769116.
126. Hoffmann-La Roche. A Two-Part, Seamless, Multi-Center, Randomized, Placebo-Controlled, Double-Blind Study to Investigate the Safety, Tolerability, Pharmacokinetics, Pharmacodynamics and Efficacy of RO7204239 in Combination With Risdiplam (RO7034067) in Ambulant Pa-tients With Spinal Muscular Atrophy. clinicaltrials.gov; 2022. Accessed September 1, 2022. https://clinicaltrials.gov/ct2/show/NCT05115110
Neuromuscular diseases (NMDs) are a broad classification of heterogeneous groups of disorders characterized by progressive muscle weakness resulting from muscle or nerve dysfunction.1 Diagnosis is based on symptoms and a full medical history, as well as on muscle and imaging tests (including electromyography, nerve-conduction studies, magnetic resonance imaging, muscle biopsy, and blood tests) to confirm or rule out specific NMDs.2 Early diagnosis of NMDs can be difficult because symptoms overlap with those of many other diseases.
Although individually, NMDs are rare, collectively, they affect approximately 250,000 people in the United States. Disease types vary in regard to cause, symptoms, prevalence, age of onset, progression, and severity. Functional impairment from any NMD can lead to lifelong morbidities and shortened life expectancy.1,3
Treatment options for NMDs are limited; most target symptoms, not disease progression. Although there is a need for safe and effective gene-based therapies for NMDs, there are challenges to developing and delivering such treatments that have impeded clinical success. These include a lack of understanding about disease pathology and drug targets, limited animal model systems, and few reliable biomarkers that are predictive of therapeutic success.4,5
Notwithstanding that challenges remain, our understanding of gene expression in NMDs has greatly advanced in the past few decades. This progress has translated into promising results in the gene-therapy field – thereby setting the stage for therapeutic approaches that use novel gene-delivery and gene-manipulation tools.6 These novel approaches include nonviral strategies, such as antisense oligonucleotides (ASOs), and viral-based strategies, such as adeno-associated virus (AAV)-mediated gene silencing and AAV-mediated gene delivery.
In this article, we highlight advancements in the clinical development of gene-based therapies for NMDs. We focus on amyotrophic lateral sclerosis (ALS), spinal muscular atrophy (SMA), and Duchenne muscular dystrophy (DMD) because of recent clinical successes in developing such therapies.1,6,7 We also catalog completed and ongoing clinical trials for ALS, SMA, and DMD (Tables 1-3).
Amyotrophic lateral sclerosis
ALS is caused by progressive degeneration of upper- and lower-motor neurons, which eventually leads to respiratory failure and death 3 to 5 years after disease onset.7-9 There are two subtypes: Familial ALS (10% of cases) and sporadic ALS (90% of cases). Commonly mutated ALS-associated genes6,8 are:
- Superoxide dismutase type 1 (SOD1).
- Chromosome 9 open reading frame 72 (C9orf72).
- Transactive response DNA-binding protein 43 (TARDBP).
- Fused in sarcoma (FUS).
SOD1-targeted therapy is being studied, with early evidence of clinical success. Mutations in SOD1 account for 10% to 20% of familial ALS cases and 1% to 2% of sporadic ALS cases.6,10 10 Mutations in C9orf72 account for 25 to 40% of familial ALS cases and 7% of sporadic ALS cases.8,9,11 Mutations in TARDBP account for 3% of familial ALS cases and 2% of sporadic cases.12 Mutations in FUS account for 4% of familial ALS cases and 1% of sporadic cases. Overall, these mutant proteins can trigger neurotoxicity, thus inducing motor-neuron death.6,10
Treatment of ALS
Two treatments for ALS are Food and Drug Administration approved: riluzole (Rilutek), approved in 1995, and edaravone (Radicava), approved in 2017.
Riluzole is an oral anti-excitotoxic glutamate antagonist.11 Approval of riluzole was based on the results of two studies that demonstrated a 2- to 3-month survival benefit.10,14 For patients who have difficulty swallowing, an oral suspension (Tiglutik, approved in 2018) and an oral film (Exservan, approved in 2019) are available.
Edaravone is a free-radical scavenger that decreases oxidative stress and is administered intravenously (IV).9,13,14 Findings from clinical trials suggest functional improvement or slower decline in function for some patients.
Although these two agents demonstrate modest therapeutic benefit, neither reverses progression of disease.10,14
Gene-based therapy for ALS
Many non-viral strategies, including antisense oligonucleotide (ASO), monoclonal antibodies, reverse transcriptase inhibitors, and HGF gene replacement therapy are used as therapeutic approaches to SOD1, C9orf72, and FUS gene mutations in ALS patients, and are being evaluated in clinical studies14,15 (Table 113-17).

Tofersen, also known as BIIB067, is an investigational ASO, administered by intrathecal (IT) injection, that binds to SOD1 mRNA, thus reducing its protein levels.16 Tofersen was evaluated in the VALOR phase 3 study (ClinicalTrials.gov Identifier: NCT02623699), a three-part randomized, double-blind, placebo-controlled trial: single ascending dose (Part A), multiple ascending dose (B), and fixed dose (C).10 In Parts A and B, 48 participants received five IT injections of tofersen or placebo over 12 weeks and were followed for an additional 12 weeks. Reduction in SOD1 protein production and neurofilament level in cerebrospinal fluid (CSF) (a potential biomarker of motor-neuron degeneration) was observed, which determined the fixed-dose for Part C.16,17
Part C examined the efficacy, safety and tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) of tofersen, compared with placebo, in adults with ALS who had a confirmed SOD1 mutation.17 A total of 108 participants were enrolled; 60 were identified as “faster-progressing”; 48, as “slower-progressing.”18 The primary endpoint of Part C was change from baseline to Week 28 on the Revised ALS Functional Rating Scale (ALSFRS-R) total score. (ALSFRS-R measures overall clinical effect; the score ranges from 0 [no function] to 4 [full function].17)
Tofersen failed to meet the primary efficacy outcome because statistically significant findings were lacking in the faster-progressing population, as measured by joint-rank analysis (difference of 1.2 on the ALSFRS-R score; P = .97). However, trends favoring tofersen were observed across key secondary clinical outcome measures18:
- Change from baseline in CSF SOD1 protein concentration.17 Percent reduction in the total SOD1 protein level was much higher in the tofersen-treated group than in the control group (38% more than controls in the faster-progressing population; 26% more than controls in the slower-progressing population).18
- Change from baseline in neurofilament light-chain concentration in plasma.17,18 Percent reduction in the level of neurofilament light chain was also observed to be higher in the tofersen-treated group than in the control group (67% more than controls in the faster-progressing population and 48% more than controls in the slower-progressing population).18
Because of these encouraging results, VALOR participants were moved to the ongoing open-label extension trial of tofersen (ClinicalTri-als.gov Identifier: NCT03070119), in which both groups were treated with the active agent.
These data suggest that early tofersen treatment might slow decline in faster-progressing patients and stabilize clinical function in slower-progressing patients.18,19 Overall, most adverse events (AEs) in the trial among patients receiving active treatment were of mild or moderate severity, and were largely consistent with either disease progression or lumbar puncture–related complications.18
Because data from VALOR suggested potential benefit from tofersen, the ATLAS trial (ClinicalTrials.gov Identifier: NCT04856982) is investigating the clinical value of presymptomatic treatment and the optimal timing of initiation of therapy.20,21 ATLAS is a phase 3, randomized, placebo-controlled trial that examines the clinical efficacy, safety, and tolerability of tofersen in presymptomatic adult carriers of SOD1 mutation who have an elevated neurofilament light-chain concentration.21 ATLAS will also evaluate the efficacy of tofersen when initiated before, rather than after, ALS manifests clinically. Enrollment is still open for this trial.20,21
Latozinemab, also known as AL001, is a first-in-class monoclonal antibody, administered by IV infusion, that elevates levels of progranulin, a key regulator of the immune activity and lysosomal function in the brain.22,23 Latozinemab limits progranulin endocytosis and degradation by sortilin inhibition.22 Progranulin gene mutations can reduce progranulin expression (by 50 to 70 percent reduction), which may cause neuro-degeneration due to abnormal accumulation of TAR-DNA-binding protein 43 (TDP-43) in the brain cells.22,24 TDP-43 pathology has also been shown to be associated with C9orf72 mutations.23 Although the mechanism is not fully understood, the role of progranulin deficiency in TDP-43 pathology is believed to be associated with neurodegenerative diseases like ALS.11,23,24,43 Previous animal models of chronic neurodegenera-tion have demonstrated how increased progranulin levels can be protective against TDP-43 pathology, increasing neuronal development and survival, thus potentially slowing disease progression.23,24,43 Currently, latozinemab is being investigated in a randomized, double-blind, placebo-controlled, multicenter phase 2 trial (ClinicalTrials.gov Identifier: NCT05053035). Approximately, 45 C90rf72-associated ALS participants (≥ 18 years of age) will receive latozinemab or placebo infusions every 4 weeks (for 24 weeks). Study endpoints include safety, tolerability, PK, PD, as well as plasma, and CSF progranulin levels.25 In previous studies, latozinemab demonstrated encouraging results in frontotemporal dementia (FTD) patients who carry a progranulin mutation. Because FTD was revealed to have significant genetic overlap with ALS, there is disease-modifying potential for latozinemab in ALS patients.23,24
TPN-101 is a nucleoside analog reverse transcriptase inhibitor, administered orally, that was originally developed for human immunodeficiency virus (HIV) treatment. However, due to recent findings suggesting retrotransposon activity contributing to neurodegeneration in TDP-43 mediated diseases, including ALS and FTD, TNP-101 is being repurposed.26 The safety and tolerability of TNP-101 are currently being evaluated in C9orf72-associated ALS and FTD patients (≥ 18 years of age). The study is a randomized, double-blind, placebo-controlled paral-lel-group phase 2a trial (ClinicalTrials.gov Identifier: NCT04993755) The study includes a screening period of 6 weeks, double-blind treatment period of 24 weeks, an open-label treatment period of 24 weeks, and 4 weeks of the post-treatment follow-up visit. Study endpoints include the incidence and severity of spontaneously reported treatment-emergent adverse events (TEAEs) associated with TNP-101 and placebo for a to-tal of 48 weeks.27
ION363 is an investigational ASO, administered by IT injection, that selectively targets one of the FUS mutations (p.P525L), which is responsible for earlier disease onset and rapid ALS progression.28,29 The clinical efficacy of ION363, specifically in clinical function and survival is being assessed in FUS-associated ALS patients (≥ 12 years of age). This randomized phase 3 study (ClinicalTrials.gov Identifier: NCT04768972) includes two parts; part 1 will consist of participants receiving a multi-dose regimen (1 dose every 4-12 weeks) of ION363 or placebo for 61 weeks followed by an open-label extension treatment period in part 2, which will consist of participants receiving ION363 (every 12 weeks) for 85 weeks. The primary endpoint of the study is the change from baseline to day 505 in functional impairment, using ALS Functional Rating Scale-Revised (ALSFRS-R). This measures functional disease severity, specifically in bulbar function, gross motor skills, fine motor skills, and respiratory. The score for all 12 questions can range from 0 (no function) to 4 (full function) with a total possible score of 48.30
Engensis, also known as VM202, is a non-viral gene therapy, administered by intramuscular (IM) injection, that uses a plasmid to deliver the hepatocyte growth factor (HGF) gene to promote HGF protein production. The HGF protein plays a role in angiogenesis, the previous of muscle atrophy, and the promotion of neuronal survival and growth. Based on preclinical studies, increasing HGF protein production has been shown to reduce neurodegeneration, thus potentially halting or slowing ALS progression.31 Currently, the safety of engensis is being evaluated in ALS patients (18-80 years of age) in the REViVALS phase 2a (ClinicalTrials.gov Identifier: NCT04632225)/2b (ClinicalTrial.gov Identifier: NCT05176093).32,33 The ReViVALS trial is a double-blind, randomized, placebo-controlled, multi-center study. The phase 2a study endpoints include the incidence of TEAEs, treatment-emergent serious adverse events (TESAEs), injection site reactions, and clinically significant labor-atory values post-treatment (engensis vs placebo group) for 180 days.33 A phase 2b study will evaluate the long-term safety of engensis for an additional 6 months. Study endpoints include the incidence of AEs, changes from baseline in ALSFRS-R scores to evaluate improvement in muscle function, changes from baseline in quality of life using the ALS patient assessment questionnaire, time to all-cause mortality compared to placebo, etc.32
Spinal muscular atrophy
SMA is a hereditary lower motor-neuron disease caused (in 95% of cases) by deletions or, less commonly, by mutations of the survival motor neuron 1 (SMN1) gene on chromosome 5q13 that encodes the SMN protein.6 Reduction in expression of the SMN protein causes motor neurons to degenerate.36-38 Because of a large inverted duplication in chromosome 5q, two variants of SMN (SMN1 and SMN2) exist on each allele. The paralog gene, SMN2, also produces the SMN protein – although at a lower level (10% to 20% of total SMN protein production) than SMN1 does.
A single nucleotide substitution in SMN2 alters splicing and suppresses transcription of exon 7, resulting in a shortened mRNA strand that yields a truncated SMN protein product.6,37,39 SMA is classified based on age of onset and maximum motor abilities achieved, ranging from the most severe (Type 0) to mildest (Type 4) disease.36,40 Because SMA patients lack functional SMN1 (due to polymorphisms), disease severity is determined by copy numbers of SMN2.6,39
Gene-based therapy for SMA
Three FDA-approved SMN treatments demonstrate clinically meaningful benefit in SMA: SMN2-targeting nusinersen [Spinraza] and risdiplam [Evrysdi], and SMN1-targeting onasemnogene abeparvovec-xioi [Zolgensma]38 Additional approaches to SMA treatment are through SMN-independent therapies, which target muscle and nerve function. Research has strongly suggested that combined SMA therapies, specifically approved SMN-targeted and investigational SMN-independent treatments, such as GYM329 (also known as RO7204239) may be the best strategy to treat all ages, stages, and types of SMA.41 (Table 226-41).

Agents that modulate SMN2. Nusinersen, approved by the FDA in 2016, was the first treatment indicated for all SMA types in pediatric and adult patients.42 The agent is an ASO that targets exon 7 of SMN2, thus stabilizing transcription. Inclusion of exon 7 increases SMN protein production, improving motor function.6,38 Nusinersen is a lifelong treatment that requires IT administration every 4 months because it cannot cross the blood-brain barrier.38,43
Pivotal clinical studies that led to approval of nusinersen include CHERISH (ClinicalTrial.gov Identifier: NCT02292537) and ENDEAR (ClinicalTrial.gov Identifier: NCT02193074) studies.
CHERISH was a phase 3, randomized, double-blind, sham procedure–controlled trial that examined the clinical efficacy and safety of nusinersen in 126 participants with later-onset SMA (2-12 years of age). The primary endpoint was the change from baseline using the Hammersmith Functional Motor Scale Expanded (HFMSE) at 15 months. HFMSE looks at 33 activities to assess improvement in motor function. The study met the primary efficacy outcome, demonstrating statistically significant (P = .0000001) improvement in overall motor function. The nusinersen group showed a 3.9-point increase in the HFMSE score from baseline, which indicates improvement, compared with a 1.0-point decline from baseline in the control group.46,47
ENDEAR was also a randomized, double-blind, sham procedure–controlled phase 3 trial, which investigated the efficacy and safety of nusinersen in 121 participants with early-onset SMA Type 1 (≤ 210 days of age). Coprimary endpoints were:
- Percentage of motor milestones responders, as determined using Section 2 of the Hammersmith Infant Neurological Examination–Part 2.
- Event-free survival (that is, avoidance of combined endpoint of death or permanent ventilation).
ENDEAR met the first primary efficacy outcome, demonstrating statistically significant (P < .0001) improvement in motor milestones (head control, rolling, independent sitting, and standing). By 13 months of age, approximately 51% of nusinersen-treated participants showed improvement, compared with none in the control group.46,47
The second primary endpoint was also met, with a statistically significant (P = .005) 47% decrease in mortality or permanent ventilation use.46-48
The NURTURE (ClinicalTrial.gov Identifier: NCT02386553) study is also investigating the efficacy and safety of nusinersen. An ongoing, open-label, supportive phase 2 trial, NURTURE is evaluating the efficacy and safety of multiple doses of nusinersen in 25 presymptomatic SMA patients (≤ 6 weeks of age). The primary endpoint of this study is time to death or respiratory intervention.49 Interim results demonstrate that 100% of presymptomatic infants are functioning without respiratory intervention after median follow-up of 2.9 years.46-48
Although nusinersen has been shown to be generally safe in clinical studies, development of lumbar puncture–related complications, as well as the need for sedation during IT administration, might affect treatment tolerability in some patients.39
Risdiplam was approved by the FDA in 2020 as the first orally administered small-molecule treatment of SMA (for patients ≤ 2 months of age).52 Risdiplam is a SMN2 splicing modifier, binding to the 5’ splice site of intron 7 and exonic splicing enhancer 2 in exon 7 of SMN2 pre-mRNA. This alternative splicing increases efficiency in SMN2 gene transcription, thus increasing SMN protein production in motor-neuron cells.36 An important advantage of risdiplam is the convenience of oral administration: A large percentage of SMA patients (that is, those with Type 2 disease) have severe scoliosis, which can further complicate therapy or deter patients from using a treatment that is administered through the IT route.40
FDA approval of risdiplam was based on clinical data from two pivotal studies, FIREFISH (ClinicalTrial.gov Identifier: NCT02913482) and SUNFISH (ClinicalTrial.gov Identifier: NCT02908685).53-54
FIREFISH is an open-label, phase 2/3 ongoing trial in infants (1-7 months of age) with SMA Type 1. The study comprises two parts; Part 1 determined the dose of risdiplam used in Part 2, which assessed the efficacy and safety of risdiplam for 24 months. The primary endpoint was the percentage of infants sitting without support for 5 seconds after 12 months of treatment using the gross motor scale of the Bayley Scales of Infant and Toddler Development–Third Edition. A statistically significant (P < .0001) therapeutic benefit was observed in motor milestones. Approximately 29% of infants achieved the motor milestone of independent sitting for 5 seconds, which had not been observed in the natural history of SMA.53-55
SUNFISH is an ongoing randomized, double-blind, placebo-controlled trial of risdiplam in adult and pediatric patients with SMA Types 2 and 3 (2-25 years old). This phase 2/3 study comprises two parts: Part 1 determined the dose (for 12 weeks) to be used for confirmatory Part 2 (for 12 to 24 months). The primary endpoint was the change from baseline on the 32-item Motor Function Measure at 12 months. The study met its primary endpoint, demonstrating statistically significant (P = .0156) improvement in motor function scores, with a 1.36-point increase in the risdiplam group, compared with a 0.19-point decrease in the control group.54,55
Ongoing risdiplam clinical trials also include JEWELFISH (ClinicalTrial.gov Identifier: NCT03032172) and RAINBOW (ClinicalTrial.gov Identifier: NCT03779334).56-57 JEWELFISH is an open-label, phase 2 trial assessing the safety of risdiplam in patients (6 months to 60 years old) who received prior treatment. The study has completed recruitment; results are pending.56 RAINBOW is an ongoing, open-label, single-arm, phase 2 trial, evaluating the clinical efficacy and safety of risdiplam in SMA-presymptomatic newborns (≤ 6 weeks old). The study is open for enrollment.57 Overall, interim results for JEWELFISH and RAINBOW appear promising.
In addition, combined SMA therapies, specifically risdiplam and GYM329 are currently being investigated to address the underlying cause and symptoms of SMA concurrently.58 GYM329, is an investigational anti-myostatin antibody, selectively binding preforms of myostatin - pro-myostatin and latent myostatin, thus improving muscle mass and strength for SMA patients.59 The safety and efficacy of GYM329 in combination with risdiplam is currently being investigated in 180 ambulant participants with SMA (2-10 years of age) in the MANATEE (ClinicalTrial.gov Identifier: NCT05115110) phase 2/3 trial. The MANATEE study is a two-part, seamless, randomized, placebo-controlled, double-blind trial. Part 1 will assess the safety of the combination treatment in approximately 36 participants; participants will receive both GYM329 (every 4 weeks) by subcutaneous (SC) injection into the abdomen and risdiplam (once per day) for 24 weeks followed by a 72-week open-label treatment period. 54,58 The outcome measures include the incidence of AEs, percentage change from baseline in the contractile area of skeletal muscle (in dominant thigh and calf), change from baseline in RHS total score, and incidence of change from baseline in serum concentration (total myostatin, free latent myostatin, and mature myostatin) etc.54 Part 2 will be conducted on 144 participants, specifically assessing the efficacy and safety of the optimal dose of GYM329 selected from Part 1 (combined with risdiplam) for 72 weeks. Once the treatment period is completed in either part, participants can partake in a 2-year open-label extension period.54,58 Other outcome measures include change from baseline in lean muscle mass (assessed by full body dual-energy X- ray absorptiometry (DXA) scan), in time taken to walk/run 10 meters (measured by RHS), in time taken to rise from the floor (measured by RHS), etc.54 Overall, this combination treatment has the potential to further improve SMA patient outcomes and will be further investigated in other patient populations (including non-ambulant patients and a broader age range) in the future.58
An agent that alters SMN1 expression. Onasemnogene abeparvovec-xioi, FDA approved in 2019, was the first gene-replacement therapy indicated for treating SMA in children ≤ 2 years old.60 Treatment utilizes an AAV vector type 9 (AAV9) to deliver a functional copy of SMN1 into target motor-neuron cells, thus increasing SMN protein production and improving motor function. This AAV serotype is ideal because it crosses the blood-brain barrier. Treatment is administered as a one-time IV fusion.38,39,43
FDA approval was based on the STR1VE (ClinicalTrial.gov Identifier: NCT03306277) phase 3 study and START (ClinicalTrial.gov Identifier: NCT02122952) phase 1 study.61,62 START was the first trial to investigate the safety and efficacy of onasemnogene abeparvovec-xioi in SMA Type 1 infants (< 6 months old). Results demonstrated remarkable clinical benefit, including 100% permanent ventilation-free survival and a 92% (11 of 12 patients) rate of improvement in motor function. Improvement in development milestones was also observed: 92% (11 of 12 patients) could sit without support for 5 seconds and 75% (9 of 12) could sit without support for 30 seconds.14,61,63
The efficacy of onasemnogene abeparvovec-xioi seen in STR1VE was consistent with what was observed in START. STRIVE, a phase 3 open-label, single-dose trial, examined treatment efficacy and safety in 22 symptomatic infants (< 6 months old) with SMA Type 1 (one or two SMN2 copies). The primary endpoint was 30 seconds of independent sitting and event-free survival. Patients were followed for as long as 18 months. Treatment showed statistically significant (P < .0001) improvement in motor milestone development and event-free survival, which had not been observed in SMA Type 1 historically. Approximately 59% (13 of 22 patients) could sit independently for 30 seconds at 18 months of age. At 14 months of age, 91% (20 of 22 patients) were alive and achieved independence from ventilatory support.34,35,53
Although many clinical studies suggest that onasemnogene abeparvovec-xioi can slow disease progression, the benefits and risks of long-term effects are still unknown. A 15-year observational study is investigating the long-term therapeutic effects and potential complications of onasemnogene abeparvovec-xioi. Participants in START were invited to enroll in this long-term follow-up study (ClinicalTrial.gov Identifier: NCT04042025).66-67
Duchenne muscular dystrophy
DMD is the most common muscular dystrophy of childhood. With an X-linked pattern of inheritance, DMD is seen mostly in young males (1 in every 3,500 male births).38,39,73 DMD is caused by mutation of the dystrophin encoding gene, or DMD, on the X chromosome. Deletion of one or more exons of DMD prevents production of the dystrophin protein, which leads to muscle degeneration.38,39,43 Common DMD deletion hotspots are exon 51 (20% of cases), exon 53 (13% of cases), exon 44 (11% of cases), and exon 45 (12% of cases).74 Nonsense mutations, which account for another 10% of DMD cases, occur when premature termination codons are found in the DMD gene. Those mutations yield truncated dystrophin protein products.39,66
Therapy for DMD
There are many therapeutic options for DMD, including deflazacort (Emflaza), FDA approved in 2017, which has been shown to reduce inflammation and immune system activity in DMD patients (≥ 5 years old). Deflazacort is a corticosteroid prodrug; its active metabolite acts on the glucocorticoid receptor to exert anti-inflammatory and immunosuppressive effects. Studies have shown that muscle strength scores over 6-12 months and average time to loss of ambulation numerically favored deflazacort over placebo.74,75
Gene-based therapy for DMD
Mutation-specific therapeutic approaches, such as exon skipping and nonsense suppression, have shown promise for the treatment of DMD (Table 358-79):

- ASO-mediated exon skipping allows one or more exons to be omitted from the mutated DMD mRNA.74,75 Effective FDA-approved ASOs include golodirsen [Vyondys 53], viltolarsen [Viltepso], and casimersen [Amondys 45].74
- An example of therapeutic suppression of nonsense mutations is ataluren [Translarna], an investigational agent that can promote premature termination codon read-through in DMD patients.66
Another potential treatment approach is through the use of AAV gene transfer to treat DMD. However, because DMD is too large for the AAV vector (packaging size, 5.0 kb), microdystrophin genes (3.5-4 kb, are used as an alternative to fit into a single AAV vector.39,76
Exon skipping targeting exon 51. Eteplirsen, approved in 2016, is indicated for the treatment of DMD patients with the confirmed DMD gene mutation that is amenable to exon 51 skipping. Eteplirsen binds to exon 51 of dystrophin pre-mRNA, causing it to be skipped, thus, restoring the reading frame in patients with DMD gene mutation amenable to exon 51 skipping. This exclusion promotes dystrophin production. Though the dystrophin protein is still functional, it is shortened.38,77 Treatment is administered IV, once a week (over 35-60 minutes). Eteplirsen’s accelerated approval was based on 3 clinical studies (ClinicalTrial.gov Identifier: NCT01396239, NCT01540409, and NCT00844597.) 78-81 The data demonstrated an increased expression of dystrophin in skeletal muscles in some DMD patients treated with eteplirsen. Though the clinical benefit of eteplirsen (including improved motor function) was not established, it was concluded by the FDA that the data were reasonably likely to predict clinical benefit. Continued approval for this indication may depend on the verification of a clinical benefit in confirmatory trials. Ongoing clinical trials include (ClinicalTrial.gov Identifier: NCT03992430 (MIS51ON), NCT03218995, and NCT03218995).77,81,82
Vesleteplirsen, is an investigational agent that is designed for DMD patients who are amendable to exon 51 skip-ping. The mechanism of action of vesleteplirsen appears to be similar to that of eteplirsen.83 The ongoing MOMENTUM (ClinicalTrial.gov Identifier: NCT04004065) phase 2 trial is assessing the safety and tolerability of vesleteplirsen at multiple-ascending dose levels (administered via IV infusion) in 60 participants (7-21 years of age). The study consists of two parts; participants receive escalating dose levels of vesleteplirsen (every 4 weeks) for 72 weeks during part A and participants receive the selected doses from part A (every 4 weeks) for 2 years during part B. Study endpoints include the number of AEs (up to 75 weeks) and the change from baseline to week 28 in dystrophin protein level. 84 Serious AEs of reversible hypomagnesemia were observed in part B, and as a result, the study protocol was amended to include magnesium supplementation and monitoring of magnesium levels.83
Exon skipping targeting exon 53. Golodirsen, FDA approved in 2019, is indicated for the treatment of DMD in patients who have a confirmed DMD mutation that is amenable to exon 53 skipping. The mechanism of action is similar to eteplirsen, however, golodirsen is designed to bind to exon 53.38,39 Treatment is administered by IV infusion over 35-60 minutes.
Approval of golodirsen was based primarily on a two-part, phase 1/2 clinical trial (ClinicalTrial.gov Identifier: NCT02310906). Part 1 was a randomized, placebo-controlled, dose-titration study that assessed multiple-dose efficacy in 12 DMD male patients, 6 to 15 years old, with deletions that were amenable to exon 53 skipping.
Part 2 was an open-label trial in 12 DMD patients from Part 1 of the trial plus 13 newly enrolled male DMD patients who were also amenable to exon 53 skipping and who had not already received treatment. Primary endpoints were change from baseline in total distance walked during the 6-minute walk test at Week 144 and dystrophin protein levels (measured by western blot testing) at Week 48. A statistically significant increase in the mean dystrophin level was observed, from a baseline 0.10% mean dystrophin level to a 1.02% mean dystrophin level after 48 weeks of treatment (P < .001). Common reported adverse events associated with golodirsen were headache, fever, abdominal pain, rash, and dermatitis. Renal toxicity was observed in preclinical studies of golodirsen but not in clinical studies.80,85
Viltolarsen, approved in 2020, is also indicated for the treatment of DMD in patients with deletions amenable to exon 53 skipping. The mechanism of action and administration (IV infusion over 60 minutes) are similar to that of golodirsen.
Approval of viltolarsen was based on two phase 2 clinical trials (ClinicalTrial.gov Identifier: NCT02740972 and NCT03167255) in a total of 32 patients. NCT02740972 was a randomized, double-blind, placebo-controlled, dose-finding study that evaluated the clinical efficacy of viltolarsen in 16 male DMD patients (4-9 years old) for 24 weeks.
NCT03167255 was an open-label study that evaluated the safety and tolerability of viltolarsen in DMD male patients (5-18 years old) for 192 weeks. The efficacy endpoint was the change in dystrophin production from baseline after 24 weeks of treatment. A statistically significant increase in the mean dystrophin level was observed, from a 0.6% mean dystrophin level at baseline to a 5.9% mean dystrophin level at Week 25 (P = .01). The most common adverse events observed were upper respiratory tract infection, cough, fever, and injection-site reaction.86-87
Exon skipping targeting exon 45. Casimersen was approved in 2021 for the treatment of DMD in patients with deletions amenable to exon 45 skipping.88 Treatment is administered by IV infusion over 30-60 minutes. Approval was based on an increase in dystrophin production in skeletal muscle in treated patients. Clinical benefit was reported in interim results from the ESSENCE (ClinicalTrial.gov Identifier: NCT02500381) study, an ongoing double-blind, placebo-controlled phase 3 trial that is evaluating the efficacy of casimersen, compared with placebo, in male participants (6-13 years old) for 48 weeks. Efficacy is based on the change from baseline dystrophin intensity level, determined by immunohistochemistry, at Week 48.
Interim results from ESSENCE show a statistically significant increase in dystrophin production in the casimersen group, from a 0.9% mean dystrophin level at baseline to a 1.7% mean dystrophin level at Week 48 (P = .004); in the control group, a 0.54% mean dystrophin level at baseline increased to a 0.76% mean dystrophin level at Week 48 (P = .09). Common adverse events have included respiratory tract infection, headache, arthralgia, fever, and oropharyngeal pain. Renal toxicity was observed in preclinical data but not in clinical studies.60,84
Targeting nonsense mutations. Ataluren is an investigational, orally administered nonsense mutation suppression therapy (through the read-through of stop codons).37 Early clinical evidence supporting the use of ataluren in DMD was seen in an open-label, dose-ranging, phase 2a study (ClinicalTrial.gov Identifier: NCT00264888) in male DMD patients (≥ 5 years old) caused by nonsense mutation. The study demonstrated a modest (61% ) increase in dystrophin expression in 23 of 38 patients after 28 days of treatment.37,91,92
However, a phase 2b randomized, double-blind, placebo-controlled trial (ClinicalTrial.gov Identifier: NCT00592553) and a subsequent confirmatory ACT DMD phase 3 study (ClinicalTrial.gov Identifier: NCT01826487) did not meet their primary endpoint of improvement in ambulation after 48 weeks as measured by the 6-minute walk test.37,93,94 In ACT DMD, approximately 74% of the ataluren group did not experience disease progression, compared with 56% of the control group (P = 0386), measured by a change in the 6-minute walk test, which assessed ambulatory decline.37,95
Based on limited data showing that ataluren is effective and well tolerated, the European Medicines Agency has given conditional approval for clinical use of the drug in Europe. However, ataluren was rejected by the FDA as a candidate therapy for DMD in the United States.22 Late-stage clinical studies of ataluren are ongoing in the United States.
AAV gene transfer with microdystrophin. Limitations on traditional gene-replacement therapy prompted exploration of gene-editing strategies for treating DMD, including using AAV-based vectors to transfer microdystrophin, an engineered version of DMD, into target muscles.43 The microdystrophin gene is designed to produce a functional, truncated form of dystrophin, thus improving muscular function.
There are 3 ongoing investigational microdystrophin gene therapies that are in clinical development (ClinicalTrial.gov Identifier: NCT03368742 (IGNITE DMD), NCT04281485 (CIFFREO), and NCT05096221 (EMBARK)).38,82
IGNITE DMD is a phase 1/2 randomized, controlled, single-ascending dose trial evaluating the safety and efficacy of a SGT-001, single IV infusion of AAV9 vector containing a microdystrophin construct in DMD patients (4-17 years old) for 12 months. At the conclusion of the trial, treatment and control groups will be followed for 5 years. The primary efficacy endpoint is the change from baseline in microdystrophin protein production in muscle-biopsy material, using western blot testing.96 Long-term interim data on biopsy findings from three patients demonstrated clinical evidence of durable microdystrophin protein expression after 2 years of treatment.96,97
The CIFFREO trial will assess the safety and efficacy of the PF-06939926 microdystrophin gene therapy, an investigational AAV9 containing microdystrophin, in approximately 99 ambulatory DMD patients (4-7 years of age). The study is a randomized, double-blind, placebo-controlled, multicenter phase 3 trial. The primary efficacy end-point is the change from baseline in the North Star Ambulatory Assessment (NSAA), which measures gross motor function. This will be assessed at 52 weeks; all study participants will be followed for a total of 5 years post-treatment.98,99,100 Due to unexpected patient death (in a non-ambulatory cohort) in the phase 1b (in a non-ambulatory cohort) in the phase 1b (ClinicalTrial.gov Identifier: (NCT03362502) trial, microdystrophin gene therapy was immediately placed on clinical hold.101,102 The amended study protocol required that all participants undergo one week of in-hospital observation after receiving treatment.102
The EMBARK study is a global, randomized, double-blind, placebo-controlled, phase 3 trial that is evaluating the safety and efficacy of SRP-9001, which is a rAAVrh74.MHCK7.microdystrophin gene therapy. The AAV vector (rAAVrh74) contains the microdystrophin construct, driven by the skeletal and cardiac muscle–specific promoter, MHCK7.98,99 In the EMBARK study, approximately 120 participants with DMD (4-7 years of age) will be enrolled. The primary efficacy endpoint includes the change from baseline to week 52 in the NSAA total score.99 Based on SRP-9001, data demonstrating consistent statistically significant functional improvements in NSAA total scores and timed function tests (after one-year post- treatment) in DMD patients from previous studies and an integrated analysis from multiple studies (ClinicalTrial.gov Identifier: NCT03375164, NCT03769116, and NCT04626674), the ongoing EMBARK has great promise.103,104
Challenges ahead, but advancements realized
Novel gene-based therapies show significant potential for transforming the treatment of NMDs. The complex pathologies of NMDs have been a huge challenge to disease management in an area once considered unremediable by gene-based therapy. However, advancements in precision medicine – specifically, gene-delivery systems (for example, AAV9 and AAVrh74 vectors) combined with gene modification strategies (ASOs and AAV-mediated silencing) – have the potential to, first, revolutionize standards of care for sporadic and inherited NMDs and, second, significantly reduce disease burden.6
What will be determined to be the “best” therapeutic approach will, likely, vary from NMD to NMD; further investigation is required to determine which agents offer optimal clinical efficacy and safety profiles.43 Furthermore, the key to therapeutic success will continue to be early detection and diagnosis – first, by better understanding disease pathology and drug targets and, second, by validation of reliable biomarkers that are predictive of therapeutic benefit.4,5
To sum up, development challenges remain, but therapeutic approaches to ALS, SMA, and DMD that utilize novel gene-delivery and gene-manipulation tools show great promise.
Ms. Yewhalashet is a student in the masters of business and science program, with a concentration in healthcare economics, at Keck Graduate Institute Henry E. Riggs School of Applied Life Sciences, Claremont, Calif. Dr. Davis is professor of practice in clinical and regulatory affairs, Keck Graduate Institute Henry E. Riggs School of Applied Life Sciences.
References
1. Aitken M et al. Understanding neuromuscular disease care. IQVIA [Internet]. Oct 30, 2018. Accessed Mar 1, 2022. https://www.iqvia.com/insights/the-iqvia-institute/reports/understanding-neuromuscular-disease-care.
2. National Institute of Neurological Disorders and Stroke. Neurological diagnostic tests and procedures fact sheet. Updated Nov 15, 2021. Ac-cessed Mar 1, 2022. http://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Neurological-Diagnostic-Tests-and-Procedures-Fact.
3. Deenen JCW et al. The epidemiology of neuromuscular disorders: A comprehensive overview of the literature. J Neuromuscul Dis. 2015;2(1):73-85.
4. Cavazzoni P. The path forward: Advancing treatments and cures for neurodegenerative diseases. U.S. Food and Drug Administration. Jul 29, 2021. Accessed Mar 1, 2022. http://www.fda.gov/news-events/congressional-testimony/path-forward-advancing-treatments-and-cures-neurodegenerative-diseases-07292021.
5. Martier R, Konstantinova P. Gene therapy for neurodegenerative diseases: Slowing down the ticking clock. Front Neurosci. 2020 Sep 18;14:580179. doi: 10.3389/fnins.2020.580179.
6. Sun J, Roy S. Gene-based therapies for neurodegenerative diseases. Nat Neurosci. 2021 Mar;24(3):297-311. doi:10.1038/s41593-020-00778-1.
7. Amado DA, Davidson BL. Gene therapy for ALS: A review. Mol Ther. 2021 Dec 1;29(12):3345-58. doi:10.1016/j.ymthe.2021.04.008.
8. Yun Y, Ha Y. CRISPR/Cas9-mediated gene correction to understand ALS. Int J Mol Sci. 2020;21(11):3801. doi:10.3390/ijms21113801.
9. National Institute of Neurological Disorders and Stroke. Amyotrophic lateral sclerosis (ALS) fact sheet. Updated Nov 15, 2021. Accessed Mar 1, 2022. http://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Amyotrophic-Lateral-Sclerosis-ALS-Fact-Sheet.
10. Cappella M et al. Gene therapy for ALS – A perspective. Int J Mol Sci. 2019;20(18):4388. doi:10.3390/ijms20184388.
11. Abramzon YA, Fratta P, Traynor BJ, Chia R. The Overlapping Genetics of Amyotrophic Lateral Sclerosis and Frontotemporal Dementia. Front Neurosci. 2020;14. Accessed August 18, 2022. https://www.frontiersin.org/articles/10.3389/fnins.2020.00042
12. Giannini M, Bayona-Feliu A, Sproviero D, Barroso SI, Cereda C, Aguilera A. TDP-43 mutations link Amyotrophic Lateral Sclerosis with R-loop homeostasis and R loop-mediated DNA damage. PLOS Genet. 2020;16(12):e1009260. doi:10.1371/journal.pgen.1009260
13. FDA-approved drugs for treating ALS. The ALS Association [Internet]. Accessed Mar 1, 2022. http://www.als.org/navigating-als/living-with-als/fda-approved-drugs.
14. Jensen TL et al. Current and future prospects for gene therapy for rare genetic diseases affecting the brain and spinal cord. Front Mol Neurosci. 2021 Oct 6;14:695937. doi:10.3389/fnmol.2021.695937.
15. ALS Gene Targeted Therapies. The ALS Association. Accessed August 22, 2022. https://www.als.org/understanding-als/who-gets-als/genetic-testing/als-gene-targeted-therapies
16. Tofersen for ALS clears phase 1/2 trial, now in phase 3. Advances in Motion. Massachusetts General Hospital [Internet]. Sep 30, 2020. Accessed Mar 1, 2022. https://advances.massgeneral.org/neuro/journal.aspx?id=1699.17. Biogen. A study to evaluate the efficacy, safety, tol-erability, pharmacokinetics, and pharmacodynamics of BIIB067 administered to adult subjects with amyotrophic lateral sclerosis and confirmed superoxide dismutase 1 mutation. ClinicalTrials.gov Identifier: NCT02623699. Updated Jul 25, 2021. Accessed Feb 17, 2022. https://clinicaltrials.gov/ct2/show/NCT02623699.
18. Biogen. Biogen announces topline results from the tofersen phase 3 study and its open-label Extension in SOD1-ALS. Press release. Oct 17, 2021. Accessed Mar 1, 2022. https://investors.biogen.com/news-releases/news-release-details/biogen-announces-topline-results-tofersen-phase-3-study-and-its.
19. Biogen. An extension study to assess the long-term safety, tolerability, pharmacokinetics, and effect on disease progression of BIIB067 ad-ministered to previously treated adults with amyotrophic lateral sclerosis caused by superoxide dismutase 1 mutation. ClinicalTrials.gov Identi-fier: NCT03070119. Updated Sep 10, 2021. Accessed Feb 17, 2022. https://clinicaltrials.gov/ct2/show/NCT03070119.
20. MS MW. #AANAM – ATLAS Trial to Assess Tofersen in Presymptomatic SOD1 ALS. Accessed February 19, 2022. https://alsnewstoday.com/news-posts/2021/04/23/aanam-atlas-clinical-trial- tofersen-presymptomatic-sod1-als-patients/
21.Biogen. A phase 3 randomized, placebo-controlled trial with a longitudinal natural history run-in and open-label extension to evaluate BIIB067 initiated in clinically presymptomatic adults with a confirmed superoxide dismutase 1 mutation. ClinicalTrials.gov Identifier: NCT04856982. Updated Feb 18, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04856982.
22. Latozinemab | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/latozinemab
23. Alector Presents AL001 (latozinemab) Data from the FTD-C9orf72 Cohort of the INFRONT-2 Phase 2 Clinical Trial | Alector. Accessed August 18, 2022. https://investors.alector.com/news- releas-es/news-release-details/alector-presents-al001-latozinemab-data-ftd-c9orf72-cohort/
24. Alector Announces First Participant Dosed in Phase 2 Study Evaluating AL001 in Amyotrophic Lateral Sclerosis (ALS) | Alector. Accessed August 18, 2022. https://investors.alector.com/news- releases/news-release-details/lector-announces-first-participant-dosed-phase-2-study-0/ 25. A Phase 2 Study to Evaluate AL001 in C9orf72-Associated ALS - Full Text View - ClinicalTrials.gov. Accessed August 19, 2022. https://clinicaltrials.gov/ct2/show/NCT05053035
26.TPN-101 | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/tpn- 101
27. Transposon Therapeutics, Inc. A Phase 2a Study of TPN-101 in Patients With Amyotrophic Lateral Sclerosis (ALS) and/or Frontotemporal Dementia (FTD) Associated With Hexanucleotide Repeat Expansion in the C9orf72 Gene (C9ORF72 ALS/FTD). clinicaltrials.gov; 2022. Ac-cessed August 17, 2022. https://clinicaltrials.gov/ct2/show/NCT04993755
28. Kerk SY, Bai Y, Smith J, et al. Homozygous ALS-linked FUS P525L mutations cell- autonomously perturb transcriptome profile and chem-oreceptor signaling in human iPSC microglia. Stem Cell Rep. 2022;17(3):678-692. doi:10.1016/j.stemcr.2022.01.004
29. ION363 | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/ion363 30. Ionis Pharmaceuticals, Inc. A Phase 1-3 Study to Evaluate the Efficacy, Safety, Pharmacokinetics and Pharmacodynamics of Intrathecally Administered ION363 in Amyo-trophic Lateral Sclerosis Patients With Fused in Sarcoma Mutations (FUS-ALS). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04768972
31. PhD LF. Engensis (VM202) - ALS News Today. Accessed August 19, 2022. https://alsnewstoday.com/vm202/
32. Helixmith Co., Ltd. A 6-Month Extension Study Following Protocol VMALS-002-2 (A Phase 2a, Double-Blind, Randomized, Place-bo-Controlled, Multicenter Study to Assess the Safety of Engensis in Participants With Amyotrophic Lateral Sclerosis). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT05176093 33. Safety of Engensis in Participants With Amyotrophic Lateral Sclerosis - Full Text View - ClinicalTrials.gov. Accessed August 19, 2022. https://clinicaltrials.gov/ct2/show/NCT04632225
34. Biogen. A phase 1, safety, tolerability, and distribution study of a microdose of radiolabeled BIIB067 co-administered with BIIB067 to healthy adults. ClinicalTrials.gov Identifier: NCT03764488. Updated Jul 19, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03764488.
35. Ionis Pharmaceuticals Inc. A phase 1, double-blind, placebo-controlled, dose-escalation study of the safety, tolerability, and pharmacokinet-ics of ISIS 333611 administered intrathecally to patients with familial amyotrophic lateral sclerosis due to superoxide dismutase 1 gene muta-tions. ClinicalTrials.gov Identifier: NCT01041222. Updated Apr 13, 2012. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01041222.
36. Messina S, Sframeli M. New treatments in spinal muscular atrophy: Positive results and new challenges. J Clin Med. 2020;9(7):2222. doi:10.3390/jcm9072222.
37. Scoto M et al. Genetic therapies for inherited neuromuscular disorders. Lancet Child Adolesc Health. 2018 Aug;2(8):600-9. doi:10.1016/S2352-4642(18)30140-8.
38. Abreu NJ, Waldrop MA. Overview of gene therapy in spinal muscular atrophy and Duchenne muscular dystrophy. Pediatr Pulmonol. 2021 Apr;56(4):710-20. doi:10.1002/ppul.25055.
39. Brandsema J, Cappa R. Genetically targeted therapies for inherited neuromuscular disorders. Practical Neurology [Internet]. Jul/Aug 2021:69-73. Accessed Mar 1, 2022. https://practicalneurology.com/articles/2021-july-aug/genetically-targeted-therapies-for-inherited-neuromuscular-disorders/pdf.
40. Ojala KS et al. In search of a cure: The development of therapeutics to alter the progression of spinal muscular atrophy. Brain Sci. 2021;11(2):194. doi:10.3390/brainsci11020194.
41. McCall S. Cure SMA Releases Updated Drug Pipeline. Cure SMA. Published December 13, 2021. Accessed August 21, 2022. https://www.curesma.org/cure-sma-releases-updated-drug-pipeline- 2021/ 42. FDA approves first drug for spinal muscular atrophy. U.S. Food and Drug Administration. News release. Dec 23, 2016. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-spinal-muscular-atrophy.43. Kirschner J. Postnatal gene therapy for neuromuscular diseases – Opportunities and limitations. J Perinat Med. 2021 Sep;49(8):1011-5. doi:10.1515/jpm-2021-0435.
43. Terryn J, Verfaillie CM, Van Damme P. Tweaking Progranulin Expression: Therapeutic Avenues and Opportunities. Front Mol Neurosci. 2021;14. Accessed September 4, 2022. https://www.frontiersin.org/articles/10.3389/fnmol.2021.71303144.
44. Biogen. A phase 3, randomized, double-blind, sham-procedure controlled study to assess the clinical efficacy and safety of ISIS 396443 administered intrathecally in patients with later-onset spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02292537. Updated Feb 17, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/study/NCT02292537.
45. Why Spinraza/later-onset studies. SPINRAZA® (nusinersen) [Internet]. Accessed Mar 1, 2022. www.spinraza.com/en_us/home/why-spinraza/later-onset-studies.html#scroll-tabs.
46. Biogen. A Phase 3, Randomized, Double-Blind, Sham-Procedure Controlled Study to Assess the Clinical Efficacy and Safety of ISIS 396443 Administered Intrathecally in Patients With Infantile- Onset Spinal Muscular Atrophy. clinicaltrials.gov; 2021. Accessed February 10, 2022. https://clinicaltrials.gov/ct2/show/results/NCT02193074
47. Early-onset SMA (Type 1) | SPINRAZA® (nusinersen). Accessed Mar 1, 2022. https://www.spinraza-hcp.com/en_us/home/why-spinraza/about-spinraza.html.
48. Finkel RS et al; ENDEAR Study Group. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N Engl J Med. 2017;377(18):1723-32. doi: 10.1056/NEJMoa1702752.
49. Biogen. An open-label study to assess the efficacy, safety, tolerability, and pharmacokinetics of multiple doses of ISIS 396443 delivered intrathecally to subjects with genetically diagnosed and presymptomatic spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02386553. Updated Nov 18, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02386553.
50. De Vivo DC et al; NURTURE Study Group. Nusinersen initiated in infants during the presymptomatic stage of spinal muscular atrophy: In-terim efficacy and safety results from the phase 2 NURTURE study. Neuromuscul Disord. 2019 Nov;29(11):842-56. doi:10.1016/j.nmd.2019.09.007.
51. Why Spinraza/presymptomatic study. SPINRAZA® (nusinersen) [Internet]. Accessed Feb 22, 2022. www.spinraza.com/en_us/home/why-spinraza/presymptomatic-study.html#scroll-tabs.
52. FDA approves oral treatment for spinal muscular atrophy. U.S. Food and Drug Administration. News release. Aug 7, 2020. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-oral-treatment-spinal-muscular-atrophy.
53. Hoffmann-La Roche. A two-part seamless, open-label, multicenter study to investigate the safety, tolerability, pharmacokinetics, pharmaco-dynamics and efficacy of risdiplam (RO7034067) in infants with type 1 spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02913482. Updated Jan 21, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02913482.
54. Hoffmann-La Roche. A two-part seamless, multi-center randomized, placebo-controlled, double-blind study to investigate the safety, tolera-bility, pharmacokinetics, pharmacodynamics and efficacy of risdiplam (RO7034067) in type 2 and 3 spinal muscular atrophy patients. Clinical-Trials.gov Identifier: NCT02908685. Updated Dec 28, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02908685.
55. Genentech. Genentech’s risdiplam shows significant improvement in survival and motor milestones in infants with type 1 spinal muscular atrophy (SMA). Press release. Apr 27, 2020. Accessed Mar 1, 2022. http://www.gene.com/media/press-releases/14847/2020-04-27/genentechs-risdiplam-shows-significant-i
56. Hoffmann-La Roche. An open-label study to investigate the safety, tolerability, and pharmacokinetics/pharmacodynamics of risdiplam (RO7034067) in adult and pediatric patients with spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT03032172. Updated Jan 27, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03032172.
57. Hoffmann-La Roche. An open-label study of risdiplam in infants with genetically diagnosed and presymptomatic spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT03779334. Updated Jan 27, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03779334.
58. McCall S. Update on Genentech/Roche Initiation of MANATEE Clinical Study. Cure SMA. Published October 20, 2021. Accessed August 20, 2022. https://www.curesma.org/update-on- genentech-roche-initiation-of-manatee-clinical-study/
59. Abati E, Manini A, Comi GP, Corti S. Inhibition of myostatin and related signaling pathways for the treatment of muscle atrophy in motor neuron diseases. Cell Mol Life Sci. 2022;79(7):374. doi:10.1007/s00018-022-04408-w
60. FDA approves innovative gene therapy to treat pediatric patients with spinal muscular atrophy, a rare disease and leading genetic cause of infant mortality. U.S. Food and Drug Administration. News release. May 24, 2019. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-innovative-gene-therapy-treat-pediatric-patients-spinal-muscular-atrophy-rare-disease.
61. Novartis Gene Therapies. Phase I gene transfer clinical trial for spinal muscular atrophy type 1 delivering AVXS-101. ClinicalTrials.gov Identifier: NCT02122952. Updated Jun 14, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02122952.
62. Novartis Gene Therapies. Phase 3, open-label, single-arm, single-dose gene replacement therapy clinical trial for patients with spinal mus-cular atrophy type 1 with one or two SMN2 copies delivering AVXS-101 by intravenous infusion. ClinicalTrials.gov Identifier: NCT03306277. Updated Jun 14, 2021. Accessed Feb 21, 2022. https://clinicaltrials.gov/ct2/show/NCT03306277.
63. Mendell JR et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N Engl J Med. 2017;377(18):1713-22. doi:10.1056/NEJMoa1706198.
64. Symptomatic study results. ZOLGENSMA [Internet]. Updated Nov 2021. Accessed Mar 1, 2022. Error! Hyperlink reference not valid..
65. Novartis Gene Therapies. A global study of a single, one-time dose of AVXS-101 delivered to infants with genetically diagnosed and pre-symptomatic spinal muscular atrophy with multiple copies of SMN2. ClinicalTrials.gov Identifier: NCT03505099. Updated Jan 1, 2022. Ac-cessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03505099.
66. Chiu W et al. Current genetics and potential gene-targeting therapeutics for neuromuscular diseases. Int J Mol Sci. 2020 Dec;21(24):9589. doi:10.3390/ijms21249589.
67. Novartis Gene Therapies. A long-term follow-up study of patients in the clinical trials for spinal muscular atrophy receiving AVXS-101. Clini-calTrials.gov Identifier: NCT04042025. Updated Jun 9, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04042025.
68. Novartis Gene Therapies. Phase 3, open-label, single-arm, single-dose gene replacement therapy clinical trial for patients with spinal mus-cular atrophy type 1 with one or two SMN2 copies delivering AVXS-101 by intravenous infusion. ClinicalTrials.gov Identifier: NCT0383718. Up-dated Jan 11, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03837184.
69. Biogen. An open-label, dose escalation study to assess the safety, tolerability and dose-range finding of multiple doses of ISIS 396443 de-livered intrathecally to patients with spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT01703988. Updated Apr 13, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01703988.
70. Biogen. A study to assess the efficacy, safety, tolerability, and pharmacokinetics of multiple doses of ISIS 396443 delivered intrathecally to patients with infantile-onset spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT01839656. Updated Feb 17, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01839656.
71. Biogen. An open-label extension study for patients with spinal muscular atrophy who previously participated in investigational studies of ISIS 396443. ClinicalTrials.gov Identifier: NCT02594124. Updated Nov 15, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02594124.
72. Biogen. Escalating dose and randomized, controlled study of nusinersen (BIIB058) in participants with spinal muscular atrophy. ClinicalTri-als.gov Identifier: NCT04089566. Updated Feb 24, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04089566.
73. National Center for Advancing Translational Sciences. Duchenne muscular dystrophy. Genetic and Rare Diseases Information Center. Up-dated Nov 2, 2020. Accessed Mar 1, 2022. https://rarediseases.info.nih.gov/diseases/6291/duchenne-muscular-dystrophy.
74. Matsuo M. Antisense oligonucleotide-mediated exon-skipping therapies: Precision medicine spreading from Duchenne muscular dystrophy. JMA J. 2021 Jul 15;4(3):232-40. doi:10.31662/jmaj.2021-0019.
75. FDA approves drug to treat Duchenne muscular dystrophy. U.S. Food and Drug Administration. News release. Feb 9, 2017. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-drug-treat-duchenne-muscular-dystrophy.74.
76. Duan D. Dystrophin gene replacement and gene repair therapy for Duchenne muscular dystrophy in 2016: An interview. Hum Gene Ther Clin Dev. 2016 Mar;27(1):9-18. doi:10.1089/humc.2016.001.
77. EXONDYS 51®. Parent Project Muscular Dystrophy. Accessed August 21, 2022. https://www.parentprojectmd.org/drug-development-pipeline/exondys-51/
78. Sarepta Therapeutics, Inc. A Randomized, Double-Blind, Placebo-Controlled, Multiple Dose Efficacy, Safety, Tolerability and Pharmacoki-netics Study of AVI-4658(Eteplirsen),in the Treatment of Ambulant Subjects With Duchenne Muscular Dystrophy. clinicaltrials.gov; 2020. Ac-cessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT01396239
79. Sarepta Therapeutics, Inc. Clinical Study to Assess the Safety Fo AVI-4658 in Subjects With Duchenne Muscular Dystrophy Due to a Frame-Shift Mutation Amenable to Correction by Skipping Exon 51. clinicaltrials.gov; 2015. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/study/NCT00844597
80. Sarepta Therapeutics, Inc. A 2-part, randomized, double-blind, placebo-controlled, dose-titration, safety, tolerability, and pharmacokinetics study (Part 1) followed by an open-label efficacy and safety evaluation (Part 2) of SRP-4053 in patients with Duchenne muscular dystrophy amenable to exon 53 skipping. ClinicalTrials.gov Identifier: NCT02310906. Updated Oct 19, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/results/NCT02310906.
81. Commissioner O of the. FDA grants accelerated approval to first drug for Duchenne muscular dystrophy. FDA. Published March 24, 2020. Accessed August 21, 2022. hDuchenne Muscular Dystrophy Amenable to Exon 51-Skipping Treatment. clinicaltrials.gov; 2022. Accessed Au-gust 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04004065
109. National Center of Neurology and Psychiatry, Japan. Exploratory study of NS-065/NCNP-01 in Duchenne muscular dystrophy. ClinicalTri-als.gov Identifier: NCT02081625; Updated Feb 26, 2020. Accessed Mar 2, 2022. https://clinicaltrialsttps://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-first-drug-duchenne-muscular- dys-trophy
82. Duchenne Drug Development Pipeline. Parent Project Muscular Dystrophy. Accessed August 21, 2022. https://www.parentprojectmd.org/duchenne-drug-development-pipeline/
83. Sarepta Therapeutics Provides Update on SRP-5051 for the Treatment of Duchenne Muscular Dystrophy | Sarepta Therapeutics, Inc. Ac-cessed August 22, 2022. https://investorrelations.sarepta.com/news-releases/news-release-details/sarepta-therapeutics- pro-vides-update-srp-5051-treatment-duchenne
84. Sarepta Therapeutics, Inc. An Open-Label Extension Study for Patients With Duchenne Muscular Dystrophy Who Participated in Studies of SRP-5051. clinicaltrials.gov; 2021. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03675126
85. VYONDYS 53. Prescribing information. Sarepta Therapeutics Inc.; 2019. Accessed Mar 2, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211970s000lbl.pdf.
86. NS Pharma Inc. Long-term use of viltolarsen in boys with Duchenne muscular dystrophy in clinical practice (VILT-502). ClinicalTrials.gov Identifier: NCT04687020. Updated Nov 22, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04687020.
87. VILTEPSO. Prescribing information. NS Pharma; 2020. Accessed Mar 2, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2020/212154s000lbl.pdf.
88. FDA approves targeted treatment for rare Duchenne muscular dystrophy mutation. U.S. Food and Drug Administration. News release. Feb 25, 2021. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-targeted-treatment-rare-duchenne-muscular-dystrophy-mutation-0.
89. Sarepta Therapeutics Inc. A double-blind, placebo-controlled, multi-center study with an open-label extension to evaluate the efficacy and safety of SRP-4045 and SRP-4053 in patients with Duchenne muscular dystrophy. Clinicaltrials.gov Identifier: NCT02500381. Updated Aug 19, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02500381.
90. AMONDYS 45. Prescribing information. Sarepta Therapeutics Inc.; 2021. Accessed Feb 22, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2021/213026lbl.pdf.
91. Finkel RS et al. Phase 2a study of ataluren-mediated dystrophin production in patients with nonsense mutation Duchenne muscular dys-trophy. PLoS ONE. 2013;8(12):e81302. doi:10.1371/journal.pone.0081302.
92. PTC Therapeutics. A phase 2 study of PTC124 as an oral treatment for nonsense-mutation-mediated Duchenne muscular dystrophy. Clini-calTrials.gov Identifier: NCT00264888. Updated Jan 14, 2009. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT00264888.
93. PTC Therapeutics. A phase 2B efficacy and safety study of PTC124 in subjects with nonsense-mutation-mediated Duchenne and Becker muscular dystrophy. ClinicalTrials.gov Identifier: NCT00592553. Updated Apr 7, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT00592553.
94. PTC Therapeutics. A phase 3 efficacy and safety study of ataluren in patients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01826487. Updated Aug 4, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01826487.
95. Bushby K et al; PTC124-GD-007-DMD Study Group. Ataluren treatment of patients with nonsense mutation dystrophinopathy. Muscle Nerve. 2014 Oct;50(4):477-87. doi:10.1002/mus.24332.
96. Solid Biosciences LLC. A randomized, controlled, open-label, single-ascending dose, phase I/II study to investigate the safety and tolerabil-ity, and efficacy of intravenous SGT-001 in male adolescents and children with Duchenne muscular dystrophy. ClinicalTrials.gov Identifier: NCT03368742. Updated Aug 24, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03368742.
97. Solid Biosciences reports 1.5-year data from patients in the ongoing IGNITE DMD phase I/II clinical trial of SGT-001. Press release. Solid Biosciences. Sep 27, 2021. Accessed Mar 2, 2022. http://www.solidbio.com/about/media/press-releases/solid-biosciences-reports-1-5-year-data-from-patients-in-the-ongoing-ignite-dmd-phase-i-ii-clinical-trial-of-sgt-001.
98. Potter RA et al. Dose-escalation study of systemically delivered rAAVrh74.MHCK7.microdystrophin in the mdx mouse model of Duchenne muscular dystrophy. Hum Gene Ther. 2021 Apr;32(7-8):375-89. doi:10.1089/hum.2019.255.
99. Sarepta Therapeutics, Inc. A Phase 3 Multinational, Randomized, Double-Blind, Placebo- Controlled Systemic Gene Delivery Study to Evaluate the Safety and Efficacy of SRP-9001 in Patients With Duchenne Muscular Dystrophy (EMBARK). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT05096221
100. Pfizer. A PHASE 3, MULTICENTER, RANDOMIZED, DOUBLE-BLIND, PLACEBO CONTROLLED STUDY TO EVALUATE THE SAFETY AND EFFICACY OF PF 06939926 FOR THE TREATMENT OF DUCHENNE MUSCULAR DYSTROPHY. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04281485
101. Pfizer. A phase 1B multicenter open-label, single ascending dose study to evaluate the safety and tolerability of PF-06939926 in ambula-tory and non-ambulatory subjects with Duchenne muscular dystrophy. ClinicalTrials.gov Identifier: NCT03362502. Updated Mar 2, 2022. Ac-cessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03362502.
102. MS MW. Phase 3 CIFFREO DMD Gene Therapy Trial Slated to Begin in June in US. Accessed August 21, 2022. https://musculardystrophynews.com/news/phase-3-trial-of-pfizers-gene-therapy- expected-to-open-in-us-in-june/
103. SRP-9001. Parent Project Muscular Dystrophy. Accessed August 22, 2022. https://www.parentprojectmd.org/drug-development-pipeline/srp-9001-micro-dystrophin-gene- transfer/
104. Sarepta Therapeutics’ Investigational Gene Therapy SRP-9001 for Duchenne Muscular Dystrophy Demonstrates Significant Functional Improvements Across Multiple Studies | Sarepta Therapeutics, Inc. Accessed August 22, 2022. https://investorrelations.sarepta.com/news-releases/news-release- details/sarepta-therapeutics-investigational-gene-therapy-srp-9001
105. Sarepta Therapeutics, Inc. An Open-Label Safety, Tolerability, and Efficacy Study of Eteplirsen in Patients With Duchenne Muscular Dys-trophy Who Have Completed Study 4658-102.clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03985878
106. Sarepta Therapeutics, Inc. An Open-Label Safety, Tolerability, and Pharmacokinetics Study of Eteplirsen in Young Patients With Duchenne Mus-cular Dystrophy Amenable to Exon 51 Skipping. clinicaltrials.gov; 2021. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03218995
107.Sarepta Therapeutics, Inc. A Randomized, Double-Blind, Dose Finding and Comparison Study of the Safety and Efficacy of a High Dose of Eteplirsen, Preceded by an Open-Label Dose Escalation, in Patients With Duchenne Muscular Dystrophy With Deletion Mutations Amenable to Exon 51 Skipping. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03992430
108. Sarepta Therapeutics, Inc. A Phase 2, Two-Part, Multiple-Ascending-Dose Study of SRP-5051 for Dose Determination, Then Dose Ex-pansion, in Patients With .gov/ct2/show/NCT02081625.
110. NS Pharma Inc. A phase II, dose finding study to assess the safety, tolerability, pharmacokinetics, and pharmacodynamics of NS-065/NCNP-01 in boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT02740972. Updated Dec 7, 2021. Ac-cessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02740972.
111. NS Pharma Inc. A phase II, open-label, extension study to assess the safety and efficacy of NS-065/NCNP-01 in boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT03167255. Updated Nov 24, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03167255.
112. NS Pharma Inc. A phase 2 open label study to assess the safety, tolerability, and efficacy of viltolarsen in ambulant and non-ambulant boys with Duchenne muscular dystrophy (DMD) compared with natural history controls. ClinicalTrials.gov Identifier: NCT04956289. Updated Feb 1, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04956289.
113. NS Pharma Inc. A phase 3 randomized, double-blind, placebo-controlled, multi-center study to assess the efficacy and safety of viltolarsen in ambulant boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT04060199. Updated Nov 16, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04060199.
114. NS Pharma Inc. A phase 3, multi-center, open-label extension study to assess the safety and efficacy of viltolarsen in ambulant boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT04768062. Updated Nov 16, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04768062.
115. Sarepta Therapeutics Inc. A randomized, double-blind, placebo-controlled, dose-titration, safety, tolerability, and pharmacokinetics study followed by an open-label safety and efficacy evaluation of SRP-4045 in advanced-stage patients with Duchenne muscular dystrophy amena-ble to exon 45 skipping. ClinicalTrials.gov Identifier: NCT02530905. Updated May 17, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02530905.
116. Sarepta Therapeutics Inc. Long-term, open-label extension study for patients with Duchenne muscular dystrophy enrolled in clinical trials evaluating casimersen or golodirsen. ClinicalTrials.gov Identifier: NCT03532542. Updated Dec 20, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03532542.
117. PTC Therapeutics. A phase 2 study of the safety, pharmacokinetics, and pharmacodynamics of ataluren (PTC124®) in patients aged ≥2 to <5 years old with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT02819557. Updated Aug 28, 2020. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02819557.
118. PTC Therapeutics. Phase 2, non-interventional, clinical study to assess dystrophin levels in subjects with nonsense mutation Duchenne muscular dystrophy who have been treated with ataluren for ≥ 9 months. ClinicalTrials.gov Identifier: NCT03796637. Updated Apr 10, 2020. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03796637.
119. PTC Therapeutics. An Open-Label Study Evaluating the Safety and Pharmacokinetics of Ataluren in Children From ≥6 Months to <2 Years of Age With Nonsense Mutation Duchenne Muscular Dystrophy. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04336826 120. PTC Therapeutics. An open-label study for previously treated ataluren (PTC124®) pa-tients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01557400. Updated Nov 25, 2020. Accessed Feb 21, 2022. https://clinicaltrials.gov/ct2/show/NCT01557400.
121. PTC Therapeutics. An open-label, safety study for ataluren (PTC124) patients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01247207. Updated Feb 16, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT01247207.
122. PTC Therapeutics. A phase 3, randomized, double-blind, placebo-controlled efficacy and safety study of ataluren in patients with non-sense mutation Duchenne muscular dystrophy and open-label extension. ClinicalTrials.gov Identifier: NCT03179631. Updated Feb 8, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03179631.
123. Sarepta Therapeutics, Inc. An Open-Label, Systemic Gene Delivery Study Using Commercial Process Material to Evaluate the Safety of and Expression From SRP-9001 in Subjects With Duchenne Muscular Dystrophy (ENDEAVOR). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04626674
124. Sarepta Therapeutics, Inc. Systemic Gene Delivery Phase I/IIa Clinical Trial for Duchenne Muscular Dystrophy Using RAA-Vrh74.MHCK7.Micro-Dystrophin (MicroDys-IV-001). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03375164
125. Sarepta Therapeutics Inc. A multicenter, randomized, double-blind, placebo-controlled trial for Duchenne muscular dystrophy using SRP-9001. ClinicalTrials.gov Identifier: NCT03769116. Updated Dec 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03769116.
126. Hoffmann-La Roche. A Two-Part, Seamless, Multi-Center, Randomized, Placebo-Controlled, Double-Blind Study to Investigate the Safety, Tolerability, Pharmacokinetics, Pharmacodynamics and Efficacy of RO7204239 in Combination With Risdiplam (RO7034067) in Ambulant Pa-tients With Spinal Muscular Atrophy. clinicaltrials.gov; 2022. Accessed September 1, 2022. https://clinicaltrials.gov/ct2/show/NCT05115110
Neuromuscular diseases (NMDs) are a broad classification of heterogeneous groups of disorders characterized by progressive muscle weakness resulting from muscle or nerve dysfunction.1 Diagnosis is based on symptoms and a full medical history, as well as on muscle and imaging tests (including electromyography, nerve-conduction studies, magnetic resonance imaging, muscle biopsy, and blood tests) to confirm or rule out specific NMDs.2 Early diagnosis of NMDs can be difficult because symptoms overlap with those of many other diseases.
Although individually, NMDs are rare, collectively, they affect approximately 250,000 people in the United States. Disease types vary in regard to cause, symptoms, prevalence, age of onset, progression, and severity. Functional impairment from any NMD can lead to lifelong morbidities and shortened life expectancy.1,3
Treatment options for NMDs are limited; most target symptoms, not disease progression. Although there is a need for safe and effective gene-based therapies for NMDs, there are challenges to developing and delivering such treatments that have impeded clinical success. These include a lack of understanding about disease pathology and drug targets, limited animal model systems, and few reliable biomarkers that are predictive of therapeutic success.4,5
Notwithstanding that challenges remain, our understanding of gene expression in NMDs has greatly advanced in the past few decades. This progress has translated into promising results in the gene-therapy field – thereby setting the stage for therapeutic approaches that use novel gene-delivery and gene-manipulation tools.6 These novel approaches include nonviral strategies, such as antisense oligonucleotides (ASOs), and viral-based strategies, such as adeno-associated virus (AAV)-mediated gene silencing and AAV-mediated gene delivery.
In this article, we highlight advancements in the clinical development of gene-based therapies for NMDs. We focus on amyotrophic lateral sclerosis (ALS), spinal muscular atrophy (SMA), and Duchenne muscular dystrophy (DMD) because of recent clinical successes in developing such therapies.1,6,7 We also catalog completed and ongoing clinical trials for ALS, SMA, and DMD (Tables 1-3).
Amyotrophic lateral sclerosis
ALS is caused by progressive degeneration of upper- and lower-motor neurons, which eventually leads to respiratory failure and death 3 to 5 years after disease onset.7-9 There are two subtypes: Familial ALS (10% of cases) and sporadic ALS (90% of cases). Commonly mutated ALS-associated genes6,8 are:
- Superoxide dismutase type 1 (SOD1).
- Chromosome 9 open reading frame 72 (C9orf72).
- Transactive response DNA-binding protein 43 (TARDBP).
- Fused in sarcoma (FUS).
SOD1-targeted therapy is being studied, with early evidence of clinical success. Mutations in SOD1 account for 10% to 20% of familial ALS cases and 1% to 2% of sporadic ALS cases.6,10 10 Mutations in C9orf72 account for 25 to 40% of familial ALS cases and 7% of sporadic ALS cases.8,9,11 Mutations in TARDBP account for 3% of familial ALS cases and 2% of sporadic cases.12 Mutations in FUS account for 4% of familial ALS cases and 1% of sporadic cases. Overall, these mutant proteins can trigger neurotoxicity, thus inducing motor-neuron death.6,10
Treatment of ALS
Two treatments for ALS are Food and Drug Administration approved: riluzole (Rilutek), approved in 1995, and edaravone (Radicava), approved in 2017.
Riluzole is an oral anti-excitotoxic glutamate antagonist.11 Approval of riluzole was based on the results of two studies that demonstrated a 2- to 3-month survival benefit.10,14 For patients who have difficulty swallowing, an oral suspension (Tiglutik, approved in 2018) and an oral film (Exservan, approved in 2019) are available.
Edaravone is a free-radical scavenger that decreases oxidative stress and is administered intravenously (IV).9,13,14 Findings from clinical trials suggest functional improvement or slower decline in function for some patients.
Although these two agents demonstrate modest therapeutic benefit, neither reverses progression of disease.10,14
Gene-based therapy for ALS
Many non-viral strategies, including antisense oligonucleotide (ASO), monoclonal antibodies, reverse transcriptase inhibitors, and HGF gene replacement therapy are used as therapeutic approaches to SOD1, C9orf72, and FUS gene mutations in ALS patients, and are being evaluated in clinical studies14,15 (Table 113-17).

Tofersen, also known as BIIB067, is an investigational ASO, administered by intrathecal (IT) injection, that binds to SOD1 mRNA, thus reducing its protein levels.16 Tofersen was evaluated in the VALOR phase 3 study (ClinicalTrials.gov Identifier: NCT02623699), a three-part randomized, double-blind, placebo-controlled trial: single ascending dose (Part A), multiple ascending dose (B), and fixed dose (C).10 In Parts A and B, 48 participants received five IT injections of tofersen or placebo over 12 weeks and were followed for an additional 12 weeks. Reduction in SOD1 protein production and neurofilament level in cerebrospinal fluid (CSF) (a potential biomarker of motor-neuron degeneration) was observed, which determined the fixed-dose for Part C.16,17
Part C examined the efficacy, safety and tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) of tofersen, compared with placebo, in adults with ALS who had a confirmed SOD1 mutation.17 A total of 108 participants were enrolled; 60 were identified as “faster-progressing”; 48, as “slower-progressing.”18 The primary endpoint of Part C was change from baseline to Week 28 on the Revised ALS Functional Rating Scale (ALSFRS-R) total score. (ALSFRS-R measures overall clinical effect; the score ranges from 0 [no function] to 4 [full function].17)
Tofersen failed to meet the primary efficacy outcome because statistically significant findings were lacking in the faster-progressing population, as measured by joint-rank analysis (difference of 1.2 on the ALSFRS-R score; P = .97). However, trends favoring tofersen were observed across key secondary clinical outcome measures18:
- Change from baseline in CSF SOD1 protein concentration.17 Percent reduction in the total SOD1 protein level was much higher in the tofersen-treated group than in the control group (38% more than controls in the faster-progressing population; 26% more than controls in the slower-progressing population).18
- Change from baseline in neurofilament light-chain concentration in plasma.17,18 Percent reduction in the level of neurofilament light chain was also observed to be higher in the tofersen-treated group than in the control group (67% more than controls in the faster-progressing population and 48% more than controls in the slower-progressing population).18
Because of these encouraging results, VALOR participants were moved to the ongoing open-label extension trial of tofersen (ClinicalTri-als.gov Identifier: NCT03070119), in which both groups were treated with the active agent.
These data suggest that early tofersen treatment might slow decline in faster-progressing patients and stabilize clinical function in slower-progressing patients.18,19 Overall, most adverse events (AEs) in the trial among patients receiving active treatment were of mild or moderate severity, and were largely consistent with either disease progression or lumbar puncture–related complications.18
Because data from VALOR suggested potential benefit from tofersen, the ATLAS trial (ClinicalTrials.gov Identifier: NCT04856982) is investigating the clinical value of presymptomatic treatment and the optimal timing of initiation of therapy.20,21 ATLAS is a phase 3, randomized, placebo-controlled trial that examines the clinical efficacy, safety, and tolerability of tofersen in presymptomatic adult carriers of SOD1 mutation who have an elevated neurofilament light-chain concentration.21 ATLAS will also evaluate the efficacy of tofersen when initiated before, rather than after, ALS manifests clinically. Enrollment is still open for this trial.20,21
Latozinemab, also known as AL001, is a first-in-class monoclonal antibody, administered by IV infusion, that elevates levels of progranulin, a key regulator of the immune activity and lysosomal function in the brain.22,23 Latozinemab limits progranulin endocytosis and degradation by sortilin inhibition.22 Progranulin gene mutations can reduce progranulin expression (by 50 to 70 percent reduction), which may cause neuro-degeneration due to abnormal accumulation of TAR-DNA-binding protein 43 (TDP-43) in the brain cells.22,24 TDP-43 pathology has also been shown to be associated with C9orf72 mutations.23 Although the mechanism is not fully understood, the role of progranulin deficiency in TDP-43 pathology is believed to be associated with neurodegenerative diseases like ALS.11,23,24,43 Previous animal models of chronic neurodegenera-tion have demonstrated how increased progranulin levels can be protective against TDP-43 pathology, increasing neuronal development and survival, thus potentially slowing disease progression.23,24,43 Currently, latozinemab is being investigated in a randomized, double-blind, placebo-controlled, multicenter phase 2 trial (ClinicalTrials.gov Identifier: NCT05053035). Approximately, 45 C90rf72-associated ALS participants (≥ 18 years of age) will receive latozinemab or placebo infusions every 4 weeks (for 24 weeks). Study endpoints include safety, tolerability, PK, PD, as well as plasma, and CSF progranulin levels.25 In previous studies, latozinemab demonstrated encouraging results in frontotemporal dementia (FTD) patients who carry a progranulin mutation. Because FTD was revealed to have significant genetic overlap with ALS, there is disease-modifying potential for latozinemab in ALS patients.23,24
TPN-101 is a nucleoside analog reverse transcriptase inhibitor, administered orally, that was originally developed for human immunodeficiency virus (HIV) treatment. However, due to recent findings suggesting retrotransposon activity contributing to neurodegeneration in TDP-43 mediated diseases, including ALS and FTD, TNP-101 is being repurposed.26 The safety and tolerability of TNP-101 are currently being evaluated in C9orf72-associated ALS and FTD patients (≥ 18 years of age). The study is a randomized, double-blind, placebo-controlled paral-lel-group phase 2a trial (ClinicalTrials.gov Identifier: NCT04993755) The study includes a screening period of 6 weeks, double-blind treatment period of 24 weeks, an open-label treatment period of 24 weeks, and 4 weeks of the post-treatment follow-up visit. Study endpoints include the incidence and severity of spontaneously reported treatment-emergent adverse events (TEAEs) associated with TNP-101 and placebo for a to-tal of 48 weeks.27
ION363 is an investigational ASO, administered by IT injection, that selectively targets one of the FUS mutations (p.P525L), which is responsible for earlier disease onset and rapid ALS progression.28,29 The clinical efficacy of ION363, specifically in clinical function and survival is being assessed in FUS-associated ALS patients (≥ 12 years of age). This randomized phase 3 study (ClinicalTrials.gov Identifier: NCT04768972) includes two parts; part 1 will consist of participants receiving a multi-dose regimen (1 dose every 4-12 weeks) of ION363 or placebo for 61 weeks followed by an open-label extension treatment period in part 2, which will consist of participants receiving ION363 (every 12 weeks) for 85 weeks. The primary endpoint of the study is the change from baseline to day 505 in functional impairment, using ALS Functional Rating Scale-Revised (ALSFRS-R). This measures functional disease severity, specifically in bulbar function, gross motor skills, fine motor skills, and respiratory. The score for all 12 questions can range from 0 (no function) to 4 (full function) with a total possible score of 48.30
Engensis, also known as VM202, is a non-viral gene therapy, administered by intramuscular (IM) injection, that uses a plasmid to deliver the hepatocyte growth factor (HGF) gene to promote HGF protein production. The HGF protein plays a role in angiogenesis, the previous of muscle atrophy, and the promotion of neuronal survival and growth. Based on preclinical studies, increasing HGF protein production has been shown to reduce neurodegeneration, thus potentially halting or slowing ALS progression.31 Currently, the safety of engensis is being evaluated in ALS patients (18-80 years of age) in the REViVALS phase 2a (ClinicalTrials.gov Identifier: NCT04632225)/2b (ClinicalTrial.gov Identifier: NCT05176093).32,33 The ReViVALS trial is a double-blind, randomized, placebo-controlled, multi-center study. The phase 2a study endpoints include the incidence of TEAEs, treatment-emergent serious adverse events (TESAEs), injection site reactions, and clinically significant labor-atory values post-treatment (engensis vs placebo group) for 180 days.33 A phase 2b study will evaluate the long-term safety of engensis for an additional 6 months. Study endpoints include the incidence of AEs, changes from baseline in ALSFRS-R scores to evaluate improvement in muscle function, changes from baseline in quality of life using the ALS patient assessment questionnaire, time to all-cause mortality compared to placebo, etc.32
Spinal muscular atrophy
SMA is a hereditary lower motor-neuron disease caused (in 95% of cases) by deletions or, less commonly, by mutations of the survival motor neuron 1 (SMN1) gene on chromosome 5q13 that encodes the SMN protein.6 Reduction in expression of the SMN protein causes motor neurons to degenerate.36-38 Because of a large inverted duplication in chromosome 5q, two variants of SMN (SMN1 and SMN2) exist on each allele. The paralog gene, SMN2, also produces the SMN protein – although at a lower level (10% to 20% of total SMN protein production) than SMN1 does.
A single nucleotide substitution in SMN2 alters splicing and suppresses transcription of exon 7, resulting in a shortened mRNA strand that yields a truncated SMN protein product.6,37,39 SMA is classified based on age of onset and maximum motor abilities achieved, ranging from the most severe (Type 0) to mildest (Type 4) disease.36,40 Because SMA patients lack functional SMN1 (due to polymorphisms), disease severity is determined by copy numbers of SMN2.6,39
Gene-based therapy for SMA
Three FDA-approved SMN treatments demonstrate clinically meaningful benefit in SMA: SMN2-targeting nusinersen [Spinraza] and risdiplam [Evrysdi], and SMN1-targeting onasemnogene abeparvovec-xioi [Zolgensma]38 Additional approaches to SMA treatment are through SMN-independent therapies, which target muscle and nerve function. Research has strongly suggested that combined SMA therapies, specifically approved SMN-targeted and investigational SMN-independent treatments, such as GYM329 (also known as RO7204239) may be the best strategy to treat all ages, stages, and types of SMA.41 (Table 226-41).

Agents that modulate SMN2. Nusinersen, approved by the FDA in 2016, was the first treatment indicated for all SMA types in pediatric and adult patients.42 The agent is an ASO that targets exon 7 of SMN2, thus stabilizing transcription. Inclusion of exon 7 increases SMN protein production, improving motor function.6,38 Nusinersen is a lifelong treatment that requires IT administration every 4 months because it cannot cross the blood-brain barrier.38,43
Pivotal clinical studies that led to approval of nusinersen include CHERISH (ClinicalTrial.gov Identifier: NCT02292537) and ENDEAR (ClinicalTrial.gov Identifier: NCT02193074) studies.
CHERISH was a phase 3, randomized, double-blind, sham procedure–controlled trial that examined the clinical efficacy and safety of nusinersen in 126 participants with later-onset SMA (2-12 years of age). The primary endpoint was the change from baseline using the Hammersmith Functional Motor Scale Expanded (HFMSE) at 15 months. HFMSE looks at 33 activities to assess improvement in motor function. The study met the primary efficacy outcome, demonstrating statistically significant (P = .0000001) improvement in overall motor function. The nusinersen group showed a 3.9-point increase in the HFMSE score from baseline, which indicates improvement, compared with a 1.0-point decline from baseline in the control group.46,47
ENDEAR was also a randomized, double-blind, sham procedure–controlled phase 3 trial, which investigated the efficacy and safety of nusinersen in 121 participants with early-onset SMA Type 1 (≤ 210 days of age). Coprimary endpoints were:
- Percentage of motor milestones responders, as determined using Section 2 of the Hammersmith Infant Neurological Examination–Part 2.
- Event-free survival (that is, avoidance of combined endpoint of death or permanent ventilation).
ENDEAR met the first primary efficacy outcome, demonstrating statistically significant (P < .0001) improvement in motor milestones (head control, rolling, independent sitting, and standing). By 13 months of age, approximately 51% of nusinersen-treated participants showed improvement, compared with none in the control group.46,47
The second primary endpoint was also met, with a statistically significant (P = .005) 47% decrease in mortality or permanent ventilation use.46-48
The NURTURE (ClinicalTrial.gov Identifier: NCT02386553) study is also investigating the efficacy and safety of nusinersen. An ongoing, open-label, supportive phase 2 trial, NURTURE is evaluating the efficacy and safety of multiple doses of nusinersen in 25 presymptomatic SMA patients (≤ 6 weeks of age). The primary endpoint of this study is time to death or respiratory intervention.49 Interim results demonstrate that 100% of presymptomatic infants are functioning without respiratory intervention after median follow-up of 2.9 years.46-48
Although nusinersen has been shown to be generally safe in clinical studies, development of lumbar puncture–related complications, as well as the need for sedation during IT administration, might affect treatment tolerability in some patients.39
Risdiplam was approved by the FDA in 2020 as the first orally administered small-molecule treatment of SMA (for patients ≤ 2 months of age).52 Risdiplam is a SMN2 splicing modifier, binding to the 5’ splice site of intron 7 and exonic splicing enhancer 2 in exon 7 of SMN2 pre-mRNA. This alternative splicing increases efficiency in SMN2 gene transcription, thus increasing SMN protein production in motor-neuron cells.36 An important advantage of risdiplam is the convenience of oral administration: A large percentage of SMA patients (that is, those with Type 2 disease) have severe scoliosis, which can further complicate therapy or deter patients from using a treatment that is administered through the IT route.40
FDA approval of risdiplam was based on clinical data from two pivotal studies, FIREFISH (ClinicalTrial.gov Identifier: NCT02913482) and SUNFISH (ClinicalTrial.gov Identifier: NCT02908685).53-54
FIREFISH is an open-label, phase 2/3 ongoing trial in infants (1-7 months of age) with SMA Type 1. The study comprises two parts; Part 1 determined the dose of risdiplam used in Part 2, which assessed the efficacy and safety of risdiplam for 24 months. The primary endpoint was the percentage of infants sitting without support for 5 seconds after 12 months of treatment using the gross motor scale of the Bayley Scales of Infant and Toddler Development–Third Edition. A statistically significant (P < .0001) therapeutic benefit was observed in motor milestones. Approximately 29% of infants achieved the motor milestone of independent sitting for 5 seconds, which had not been observed in the natural history of SMA.53-55
SUNFISH is an ongoing randomized, double-blind, placebo-controlled trial of risdiplam in adult and pediatric patients with SMA Types 2 and 3 (2-25 years old). This phase 2/3 study comprises two parts: Part 1 determined the dose (for 12 weeks) to be used for confirmatory Part 2 (for 12 to 24 months). The primary endpoint was the change from baseline on the 32-item Motor Function Measure at 12 months. The study met its primary endpoint, demonstrating statistically significant (P = .0156) improvement in motor function scores, with a 1.36-point increase in the risdiplam group, compared with a 0.19-point decrease in the control group.54,55
Ongoing risdiplam clinical trials also include JEWELFISH (ClinicalTrial.gov Identifier: NCT03032172) and RAINBOW (ClinicalTrial.gov Identifier: NCT03779334).56-57 JEWELFISH is an open-label, phase 2 trial assessing the safety of risdiplam in patients (6 months to 60 years old) who received prior treatment. The study has completed recruitment; results are pending.56 RAINBOW is an ongoing, open-label, single-arm, phase 2 trial, evaluating the clinical efficacy and safety of risdiplam in SMA-presymptomatic newborns (≤ 6 weeks old). The study is open for enrollment.57 Overall, interim results for JEWELFISH and RAINBOW appear promising.
In addition, combined SMA therapies, specifically risdiplam and GYM329 are currently being investigated to address the underlying cause and symptoms of SMA concurrently.58 GYM329, is an investigational anti-myostatin antibody, selectively binding preforms of myostatin - pro-myostatin and latent myostatin, thus improving muscle mass and strength for SMA patients.59 The safety and efficacy of GYM329 in combination with risdiplam is currently being investigated in 180 ambulant participants with SMA (2-10 years of age) in the MANATEE (ClinicalTrial.gov Identifier: NCT05115110) phase 2/3 trial. The MANATEE study is a two-part, seamless, randomized, placebo-controlled, double-blind trial. Part 1 will assess the safety of the combination treatment in approximately 36 participants; participants will receive both GYM329 (every 4 weeks) by subcutaneous (SC) injection into the abdomen and risdiplam (once per day) for 24 weeks followed by a 72-week open-label treatment period. 54,58 The outcome measures include the incidence of AEs, percentage change from baseline in the contractile area of skeletal muscle (in dominant thigh and calf), change from baseline in RHS total score, and incidence of change from baseline in serum concentration (total myostatin, free latent myostatin, and mature myostatin) etc.54 Part 2 will be conducted on 144 participants, specifically assessing the efficacy and safety of the optimal dose of GYM329 selected from Part 1 (combined with risdiplam) for 72 weeks. Once the treatment period is completed in either part, participants can partake in a 2-year open-label extension period.54,58 Other outcome measures include change from baseline in lean muscle mass (assessed by full body dual-energy X- ray absorptiometry (DXA) scan), in time taken to walk/run 10 meters (measured by RHS), in time taken to rise from the floor (measured by RHS), etc.54 Overall, this combination treatment has the potential to further improve SMA patient outcomes and will be further investigated in other patient populations (including non-ambulant patients and a broader age range) in the future.58
An agent that alters SMN1 expression. Onasemnogene abeparvovec-xioi, FDA approved in 2019, was the first gene-replacement therapy indicated for treating SMA in children ≤ 2 years old.60 Treatment utilizes an AAV vector type 9 (AAV9) to deliver a functional copy of SMN1 into target motor-neuron cells, thus increasing SMN protein production and improving motor function. This AAV serotype is ideal because it crosses the blood-brain barrier. Treatment is administered as a one-time IV fusion.38,39,43
FDA approval was based on the STR1VE (ClinicalTrial.gov Identifier: NCT03306277) phase 3 study and START (ClinicalTrial.gov Identifier: NCT02122952) phase 1 study.61,62 START was the first trial to investigate the safety and efficacy of onasemnogene abeparvovec-xioi in SMA Type 1 infants (< 6 months old). Results demonstrated remarkable clinical benefit, including 100% permanent ventilation-free survival and a 92% (11 of 12 patients) rate of improvement in motor function. Improvement in development milestones was also observed: 92% (11 of 12 patients) could sit without support for 5 seconds and 75% (9 of 12) could sit without support for 30 seconds.14,61,63
The efficacy of onasemnogene abeparvovec-xioi seen in STR1VE was consistent with what was observed in START. STRIVE, a phase 3 open-label, single-dose trial, examined treatment efficacy and safety in 22 symptomatic infants (< 6 months old) with SMA Type 1 (one or two SMN2 copies). The primary endpoint was 30 seconds of independent sitting and event-free survival. Patients were followed for as long as 18 months. Treatment showed statistically significant (P < .0001) improvement in motor milestone development and event-free survival, which had not been observed in SMA Type 1 historically. Approximately 59% (13 of 22 patients) could sit independently for 30 seconds at 18 months of age. At 14 months of age, 91% (20 of 22 patients) were alive and achieved independence from ventilatory support.34,35,53
Although many clinical studies suggest that onasemnogene abeparvovec-xioi can slow disease progression, the benefits and risks of long-term effects are still unknown. A 15-year observational study is investigating the long-term therapeutic effects and potential complications of onasemnogene abeparvovec-xioi. Participants in START were invited to enroll in this long-term follow-up study (ClinicalTrial.gov Identifier: NCT04042025).66-67
Duchenne muscular dystrophy
DMD is the most common muscular dystrophy of childhood. With an X-linked pattern of inheritance, DMD is seen mostly in young males (1 in every 3,500 male births).38,39,73 DMD is caused by mutation of the dystrophin encoding gene, or DMD, on the X chromosome. Deletion of one or more exons of DMD prevents production of the dystrophin protein, which leads to muscle degeneration.38,39,43 Common DMD deletion hotspots are exon 51 (20% of cases), exon 53 (13% of cases), exon 44 (11% of cases), and exon 45 (12% of cases).74 Nonsense mutations, which account for another 10% of DMD cases, occur when premature termination codons are found in the DMD gene. Those mutations yield truncated dystrophin protein products.39,66
Therapy for DMD
There are many therapeutic options for DMD, including deflazacort (Emflaza), FDA approved in 2017, which has been shown to reduce inflammation and immune system activity in DMD patients (≥ 5 years old). Deflazacort is a corticosteroid prodrug; its active metabolite acts on the glucocorticoid receptor to exert anti-inflammatory and immunosuppressive effects. Studies have shown that muscle strength scores over 6-12 months and average time to loss of ambulation numerically favored deflazacort over placebo.74,75
Gene-based therapy for DMD
Mutation-specific therapeutic approaches, such as exon skipping and nonsense suppression, have shown promise for the treatment of DMD (Table 358-79):

- ASO-mediated exon skipping allows one or more exons to be omitted from the mutated DMD mRNA.74,75 Effective FDA-approved ASOs include golodirsen [Vyondys 53], viltolarsen [Viltepso], and casimersen [Amondys 45].74
- An example of therapeutic suppression of nonsense mutations is ataluren [Translarna], an investigational agent that can promote premature termination codon read-through in DMD patients.66
Another potential treatment approach is through the use of AAV gene transfer to treat DMD. However, because DMD is too large for the AAV vector (packaging size, 5.0 kb), microdystrophin genes (3.5-4 kb, are used as an alternative to fit into a single AAV vector.39,76
Exon skipping targeting exon 51. Eteplirsen, approved in 2016, is indicated for the treatment of DMD patients with the confirmed DMD gene mutation that is amenable to exon 51 skipping. Eteplirsen binds to exon 51 of dystrophin pre-mRNA, causing it to be skipped, thus, restoring the reading frame in patients with DMD gene mutation amenable to exon 51 skipping. This exclusion promotes dystrophin production. Though the dystrophin protein is still functional, it is shortened.38,77 Treatment is administered IV, once a week (over 35-60 minutes). Eteplirsen’s accelerated approval was based on 3 clinical studies (ClinicalTrial.gov Identifier: NCT01396239, NCT01540409, and NCT00844597.) 78-81 The data demonstrated an increased expression of dystrophin in skeletal muscles in some DMD patients treated with eteplirsen. Though the clinical benefit of eteplirsen (including improved motor function) was not established, it was concluded by the FDA that the data were reasonably likely to predict clinical benefit. Continued approval for this indication may depend on the verification of a clinical benefit in confirmatory trials. Ongoing clinical trials include (ClinicalTrial.gov Identifier: NCT03992430 (MIS51ON), NCT03218995, and NCT03218995).77,81,82
Vesleteplirsen, is an investigational agent that is designed for DMD patients who are amendable to exon 51 skip-ping. The mechanism of action of vesleteplirsen appears to be similar to that of eteplirsen.83 The ongoing MOMENTUM (ClinicalTrial.gov Identifier: NCT04004065) phase 2 trial is assessing the safety and tolerability of vesleteplirsen at multiple-ascending dose levels (administered via IV infusion) in 60 participants (7-21 years of age). The study consists of two parts; participants receive escalating dose levels of vesleteplirsen (every 4 weeks) for 72 weeks during part A and participants receive the selected doses from part A (every 4 weeks) for 2 years during part B. Study endpoints include the number of AEs (up to 75 weeks) and the change from baseline to week 28 in dystrophin protein level. 84 Serious AEs of reversible hypomagnesemia were observed in part B, and as a result, the study protocol was amended to include magnesium supplementation and monitoring of magnesium levels.83
Exon skipping targeting exon 53. Golodirsen, FDA approved in 2019, is indicated for the treatment of DMD in patients who have a confirmed DMD mutation that is amenable to exon 53 skipping. The mechanism of action is similar to eteplirsen, however, golodirsen is designed to bind to exon 53.38,39 Treatment is administered by IV infusion over 35-60 minutes.
Approval of golodirsen was based primarily on a two-part, phase 1/2 clinical trial (ClinicalTrial.gov Identifier: NCT02310906). Part 1 was a randomized, placebo-controlled, dose-titration study that assessed multiple-dose efficacy in 12 DMD male patients, 6 to 15 years old, with deletions that were amenable to exon 53 skipping.
Part 2 was an open-label trial in 12 DMD patients from Part 1 of the trial plus 13 newly enrolled male DMD patients who were also amenable to exon 53 skipping and who had not already received treatment. Primary endpoints were change from baseline in total distance walked during the 6-minute walk test at Week 144 and dystrophin protein levels (measured by western blot testing) at Week 48. A statistically significant increase in the mean dystrophin level was observed, from a baseline 0.10% mean dystrophin level to a 1.02% mean dystrophin level after 48 weeks of treatment (P < .001). Common reported adverse events associated with golodirsen were headache, fever, abdominal pain, rash, and dermatitis. Renal toxicity was observed in preclinical studies of golodirsen but not in clinical studies.80,85
Viltolarsen, approved in 2020, is also indicated for the treatment of DMD in patients with deletions amenable to exon 53 skipping. The mechanism of action and administration (IV infusion over 60 minutes) are similar to that of golodirsen.
Approval of viltolarsen was based on two phase 2 clinical trials (ClinicalTrial.gov Identifier: NCT02740972 and NCT03167255) in a total of 32 patients. NCT02740972 was a randomized, double-blind, placebo-controlled, dose-finding study that evaluated the clinical efficacy of viltolarsen in 16 male DMD patients (4-9 years old) for 24 weeks.
NCT03167255 was an open-label study that evaluated the safety and tolerability of viltolarsen in DMD male patients (5-18 years old) for 192 weeks. The efficacy endpoint was the change in dystrophin production from baseline after 24 weeks of treatment. A statistically significant increase in the mean dystrophin level was observed, from a 0.6% mean dystrophin level at baseline to a 5.9% mean dystrophin level at Week 25 (P = .01). The most common adverse events observed were upper respiratory tract infection, cough, fever, and injection-site reaction.86-87
Exon skipping targeting exon 45. Casimersen was approved in 2021 for the treatment of DMD in patients with deletions amenable to exon 45 skipping.88 Treatment is administered by IV infusion over 30-60 minutes. Approval was based on an increase in dystrophin production in skeletal muscle in treated patients. Clinical benefit was reported in interim results from the ESSENCE (ClinicalTrial.gov Identifier: NCT02500381) study, an ongoing double-blind, placebo-controlled phase 3 trial that is evaluating the efficacy of casimersen, compared with placebo, in male participants (6-13 years old) for 48 weeks. Efficacy is based on the change from baseline dystrophin intensity level, determined by immunohistochemistry, at Week 48.
Interim results from ESSENCE show a statistically significant increase in dystrophin production in the casimersen group, from a 0.9% mean dystrophin level at baseline to a 1.7% mean dystrophin level at Week 48 (P = .004); in the control group, a 0.54% mean dystrophin level at baseline increased to a 0.76% mean dystrophin level at Week 48 (P = .09). Common adverse events have included respiratory tract infection, headache, arthralgia, fever, and oropharyngeal pain. Renal toxicity was observed in preclinical data but not in clinical studies.60,84
Targeting nonsense mutations. Ataluren is an investigational, orally administered nonsense mutation suppression therapy (through the read-through of stop codons).37 Early clinical evidence supporting the use of ataluren in DMD was seen in an open-label, dose-ranging, phase 2a study (ClinicalTrial.gov Identifier: NCT00264888) in male DMD patients (≥ 5 years old) caused by nonsense mutation. The study demonstrated a modest (61% ) increase in dystrophin expression in 23 of 38 patients after 28 days of treatment.37,91,92
However, a phase 2b randomized, double-blind, placebo-controlled trial (ClinicalTrial.gov Identifier: NCT00592553) and a subsequent confirmatory ACT DMD phase 3 study (ClinicalTrial.gov Identifier: NCT01826487) did not meet their primary endpoint of improvement in ambulation after 48 weeks as measured by the 6-minute walk test.37,93,94 In ACT DMD, approximately 74% of the ataluren group did not experience disease progression, compared with 56% of the control group (P = 0386), measured by a change in the 6-minute walk test, which assessed ambulatory decline.37,95
Based on limited data showing that ataluren is effective and well tolerated, the European Medicines Agency has given conditional approval for clinical use of the drug in Europe. However, ataluren was rejected by the FDA as a candidate therapy for DMD in the United States.22 Late-stage clinical studies of ataluren are ongoing in the United States.
AAV gene transfer with microdystrophin. Limitations on traditional gene-replacement therapy prompted exploration of gene-editing strategies for treating DMD, including using AAV-based vectors to transfer microdystrophin, an engineered version of DMD, into target muscles.43 The microdystrophin gene is designed to produce a functional, truncated form of dystrophin, thus improving muscular function.
There are 3 ongoing investigational microdystrophin gene therapies that are in clinical development (ClinicalTrial.gov Identifier: NCT03368742 (IGNITE DMD), NCT04281485 (CIFFREO), and NCT05096221 (EMBARK)).38,82
IGNITE DMD is a phase 1/2 randomized, controlled, single-ascending dose trial evaluating the safety and efficacy of a SGT-001, single IV infusion of AAV9 vector containing a microdystrophin construct in DMD patients (4-17 years old) for 12 months. At the conclusion of the trial, treatment and control groups will be followed for 5 years. The primary efficacy endpoint is the change from baseline in microdystrophin protein production in muscle-biopsy material, using western blot testing.96 Long-term interim data on biopsy findings from three patients demonstrated clinical evidence of durable microdystrophin protein expression after 2 years of treatment.96,97
The CIFFREO trial will assess the safety and efficacy of the PF-06939926 microdystrophin gene therapy, an investigational AAV9 containing microdystrophin, in approximately 99 ambulatory DMD patients (4-7 years of age). The study is a randomized, double-blind, placebo-controlled, multicenter phase 3 trial. The primary efficacy end-point is the change from baseline in the North Star Ambulatory Assessment (NSAA), which measures gross motor function. This will be assessed at 52 weeks; all study participants will be followed for a total of 5 years post-treatment.98,99,100 Due to unexpected patient death (in a non-ambulatory cohort) in the phase 1b (in a non-ambulatory cohort) in the phase 1b (ClinicalTrial.gov Identifier: (NCT03362502) trial, microdystrophin gene therapy was immediately placed on clinical hold.101,102 The amended study protocol required that all participants undergo one week of in-hospital observation after receiving treatment.102
The EMBARK study is a global, randomized, double-blind, placebo-controlled, phase 3 trial that is evaluating the safety and efficacy of SRP-9001, which is a rAAVrh74.MHCK7.microdystrophin gene therapy. The AAV vector (rAAVrh74) contains the microdystrophin construct, driven by the skeletal and cardiac muscle–specific promoter, MHCK7.98,99 In the EMBARK study, approximately 120 participants with DMD (4-7 years of age) will be enrolled. The primary efficacy endpoint includes the change from baseline to week 52 in the NSAA total score.99 Based on SRP-9001, data demonstrating consistent statistically significant functional improvements in NSAA total scores and timed function tests (after one-year post- treatment) in DMD patients from previous studies and an integrated analysis from multiple studies (ClinicalTrial.gov Identifier: NCT03375164, NCT03769116, and NCT04626674), the ongoing EMBARK has great promise.103,104
Challenges ahead, but advancements realized
Novel gene-based therapies show significant potential for transforming the treatment of NMDs. The complex pathologies of NMDs have been a huge challenge to disease management in an area once considered unremediable by gene-based therapy. However, advancements in precision medicine – specifically, gene-delivery systems (for example, AAV9 and AAVrh74 vectors) combined with gene modification strategies (ASOs and AAV-mediated silencing) – have the potential to, first, revolutionize standards of care for sporadic and inherited NMDs and, second, significantly reduce disease burden.6
What will be determined to be the “best” therapeutic approach will, likely, vary from NMD to NMD; further investigation is required to determine which agents offer optimal clinical efficacy and safety profiles.43 Furthermore, the key to therapeutic success will continue to be early detection and diagnosis – first, by better understanding disease pathology and drug targets and, second, by validation of reliable biomarkers that are predictive of therapeutic benefit.4,5
To sum up, development challenges remain, but therapeutic approaches to ALS, SMA, and DMD that utilize novel gene-delivery and gene-manipulation tools show great promise.
Ms. Yewhalashet is a student in the masters of business and science program, with a concentration in healthcare economics, at Keck Graduate Institute Henry E. Riggs School of Applied Life Sciences, Claremont, Calif. Dr. Davis is professor of practice in clinical and regulatory affairs, Keck Graduate Institute Henry E. Riggs School of Applied Life Sciences.
References
1. Aitken M et al. Understanding neuromuscular disease care. IQVIA [Internet]. Oct 30, 2018. Accessed Mar 1, 2022. https://www.iqvia.com/insights/the-iqvia-institute/reports/understanding-neuromuscular-disease-care.
2. National Institute of Neurological Disorders and Stroke. Neurological diagnostic tests and procedures fact sheet. Updated Nov 15, 2021. Ac-cessed Mar 1, 2022. http://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Neurological-Diagnostic-Tests-and-Procedures-Fact.
3. Deenen JCW et al. The epidemiology of neuromuscular disorders: A comprehensive overview of the literature. J Neuromuscul Dis. 2015;2(1):73-85.
4. Cavazzoni P. The path forward: Advancing treatments and cures for neurodegenerative diseases. U.S. Food and Drug Administration. Jul 29, 2021. Accessed Mar 1, 2022. http://www.fda.gov/news-events/congressional-testimony/path-forward-advancing-treatments-and-cures-neurodegenerative-diseases-07292021.
5. Martier R, Konstantinova P. Gene therapy for neurodegenerative diseases: Slowing down the ticking clock. Front Neurosci. 2020 Sep 18;14:580179. doi: 10.3389/fnins.2020.580179.
6. Sun J, Roy S. Gene-based therapies for neurodegenerative diseases. Nat Neurosci. 2021 Mar;24(3):297-311. doi:10.1038/s41593-020-00778-1.
7. Amado DA, Davidson BL. Gene therapy for ALS: A review. Mol Ther. 2021 Dec 1;29(12):3345-58. doi:10.1016/j.ymthe.2021.04.008.
8. Yun Y, Ha Y. CRISPR/Cas9-mediated gene correction to understand ALS. Int J Mol Sci. 2020;21(11):3801. doi:10.3390/ijms21113801.
9. National Institute of Neurological Disorders and Stroke. Amyotrophic lateral sclerosis (ALS) fact sheet. Updated Nov 15, 2021. Accessed Mar 1, 2022. http://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Amyotrophic-Lateral-Sclerosis-ALS-Fact-Sheet.
10. Cappella M et al. Gene therapy for ALS – A perspective. Int J Mol Sci. 2019;20(18):4388. doi:10.3390/ijms20184388.
11. Abramzon YA, Fratta P, Traynor BJ, Chia R. The Overlapping Genetics of Amyotrophic Lateral Sclerosis and Frontotemporal Dementia. Front Neurosci. 2020;14. Accessed August 18, 2022. https://www.frontiersin.org/articles/10.3389/fnins.2020.00042
12. Giannini M, Bayona-Feliu A, Sproviero D, Barroso SI, Cereda C, Aguilera A. TDP-43 mutations link Amyotrophic Lateral Sclerosis with R-loop homeostasis and R loop-mediated DNA damage. PLOS Genet. 2020;16(12):e1009260. doi:10.1371/journal.pgen.1009260
13. FDA-approved drugs for treating ALS. The ALS Association [Internet]. Accessed Mar 1, 2022. http://www.als.org/navigating-als/living-with-als/fda-approved-drugs.
14. Jensen TL et al. Current and future prospects for gene therapy for rare genetic diseases affecting the brain and spinal cord. Front Mol Neurosci. 2021 Oct 6;14:695937. doi:10.3389/fnmol.2021.695937.
15. ALS Gene Targeted Therapies. The ALS Association. Accessed August 22, 2022. https://www.als.org/understanding-als/who-gets-als/genetic-testing/als-gene-targeted-therapies
16. Tofersen for ALS clears phase 1/2 trial, now in phase 3. Advances in Motion. Massachusetts General Hospital [Internet]. Sep 30, 2020. Accessed Mar 1, 2022. https://advances.massgeneral.org/neuro/journal.aspx?id=1699.17. Biogen. A study to evaluate the efficacy, safety, tol-erability, pharmacokinetics, and pharmacodynamics of BIIB067 administered to adult subjects with amyotrophic lateral sclerosis and confirmed superoxide dismutase 1 mutation. ClinicalTrials.gov Identifier: NCT02623699. Updated Jul 25, 2021. Accessed Feb 17, 2022. https://clinicaltrials.gov/ct2/show/NCT02623699.
18. Biogen. Biogen announces topline results from the tofersen phase 3 study and its open-label Extension in SOD1-ALS. Press release. Oct 17, 2021. Accessed Mar 1, 2022. https://investors.biogen.com/news-releases/news-release-details/biogen-announces-topline-results-tofersen-phase-3-study-and-its.
19. Biogen. An extension study to assess the long-term safety, tolerability, pharmacokinetics, and effect on disease progression of BIIB067 ad-ministered to previously treated adults with amyotrophic lateral sclerosis caused by superoxide dismutase 1 mutation. ClinicalTrials.gov Identi-fier: NCT03070119. Updated Sep 10, 2021. Accessed Feb 17, 2022. https://clinicaltrials.gov/ct2/show/NCT03070119.
20. MS MW. #AANAM – ATLAS Trial to Assess Tofersen in Presymptomatic SOD1 ALS. Accessed February 19, 2022. https://alsnewstoday.com/news-posts/2021/04/23/aanam-atlas-clinical-trial- tofersen-presymptomatic-sod1-als-patients/
21.Biogen. A phase 3 randomized, placebo-controlled trial with a longitudinal natural history run-in and open-label extension to evaluate BIIB067 initiated in clinically presymptomatic adults with a confirmed superoxide dismutase 1 mutation. ClinicalTrials.gov Identifier: NCT04856982. Updated Feb 18, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04856982.
22. Latozinemab | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/latozinemab
23. Alector Presents AL001 (latozinemab) Data from the FTD-C9orf72 Cohort of the INFRONT-2 Phase 2 Clinical Trial | Alector. Accessed August 18, 2022. https://investors.alector.com/news- releas-es/news-release-details/alector-presents-al001-latozinemab-data-ftd-c9orf72-cohort/
24. Alector Announces First Participant Dosed in Phase 2 Study Evaluating AL001 in Amyotrophic Lateral Sclerosis (ALS) | Alector. Accessed August 18, 2022. https://investors.alector.com/news- releases/news-release-details/lector-announces-first-participant-dosed-phase-2-study-0/ 25. A Phase 2 Study to Evaluate AL001 in C9orf72-Associated ALS - Full Text View - ClinicalTrials.gov. Accessed August 19, 2022. https://clinicaltrials.gov/ct2/show/NCT05053035
26.TPN-101 | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/tpn- 101
27. Transposon Therapeutics, Inc. A Phase 2a Study of TPN-101 in Patients With Amyotrophic Lateral Sclerosis (ALS) and/or Frontotemporal Dementia (FTD) Associated With Hexanucleotide Repeat Expansion in the C9orf72 Gene (C9ORF72 ALS/FTD). clinicaltrials.gov; 2022. Ac-cessed August 17, 2022. https://clinicaltrials.gov/ct2/show/NCT04993755
28. Kerk SY, Bai Y, Smith J, et al. Homozygous ALS-linked FUS P525L mutations cell- autonomously perturb transcriptome profile and chem-oreceptor signaling in human iPSC microglia. Stem Cell Rep. 2022;17(3):678-692. doi:10.1016/j.stemcr.2022.01.004
29. ION363 | ALZFORUM. Accessed August 19, 2022. https://www.alzforum.org/therapeutics/ion363 30. Ionis Pharmaceuticals, Inc. A Phase 1-3 Study to Evaluate the Efficacy, Safety, Pharmacokinetics and Pharmacodynamics of Intrathecally Administered ION363 in Amyo-trophic Lateral Sclerosis Patients With Fused in Sarcoma Mutations (FUS-ALS). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04768972
31. PhD LF. Engensis (VM202) - ALS News Today. Accessed August 19, 2022. https://alsnewstoday.com/vm202/
32. Helixmith Co., Ltd. A 6-Month Extension Study Following Protocol VMALS-002-2 (A Phase 2a, Double-Blind, Randomized, Place-bo-Controlled, Multicenter Study to Assess the Safety of Engensis in Participants With Amyotrophic Lateral Sclerosis). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT05176093 33. Safety of Engensis in Participants With Amyotrophic Lateral Sclerosis - Full Text View - ClinicalTrials.gov. Accessed August 19, 2022. https://clinicaltrials.gov/ct2/show/NCT04632225
34. Biogen. A phase 1, safety, tolerability, and distribution study of a microdose of radiolabeled BIIB067 co-administered with BIIB067 to healthy adults. ClinicalTrials.gov Identifier: NCT03764488. Updated Jul 19, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03764488.
35. Ionis Pharmaceuticals Inc. A phase 1, double-blind, placebo-controlled, dose-escalation study of the safety, tolerability, and pharmacokinet-ics of ISIS 333611 administered intrathecally to patients with familial amyotrophic lateral sclerosis due to superoxide dismutase 1 gene muta-tions. ClinicalTrials.gov Identifier: NCT01041222. Updated Apr 13, 2012. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01041222.
36. Messina S, Sframeli M. New treatments in spinal muscular atrophy: Positive results and new challenges. J Clin Med. 2020;9(7):2222. doi:10.3390/jcm9072222.
37. Scoto M et al. Genetic therapies for inherited neuromuscular disorders. Lancet Child Adolesc Health. 2018 Aug;2(8):600-9. doi:10.1016/S2352-4642(18)30140-8.
38. Abreu NJ, Waldrop MA. Overview of gene therapy in spinal muscular atrophy and Duchenne muscular dystrophy. Pediatr Pulmonol. 2021 Apr;56(4):710-20. doi:10.1002/ppul.25055.
39. Brandsema J, Cappa R. Genetically targeted therapies for inherited neuromuscular disorders. Practical Neurology [Internet]. Jul/Aug 2021:69-73. Accessed Mar 1, 2022. https://practicalneurology.com/articles/2021-july-aug/genetically-targeted-therapies-for-inherited-neuromuscular-disorders/pdf.
40. Ojala KS et al. In search of a cure: The development of therapeutics to alter the progression of spinal muscular atrophy. Brain Sci. 2021;11(2):194. doi:10.3390/brainsci11020194.
41. McCall S. Cure SMA Releases Updated Drug Pipeline. Cure SMA. Published December 13, 2021. Accessed August 21, 2022. https://www.curesma.org/cure-sma-releases-updated-drug-pipeline- 2021/ 42. FDA approves first drug for spinal muscular atrophy. U.S. Food and Drug Administration. News release. Dec 23, 2016. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-spinal-muscular-atrophy.43. Kirschner J. Postnatal gene therapy for neuromuscular diseases – Opportunities and limitations. J Perinat Med. 2021 Sep;49(8):1011-5. doi:10.1515/jpm-2021-0435.
43. Terryn J, Verfaillie CM, Van Damme P. Tweaking Progranulin Expression: Therapeutic Avenues and Opportunities. Front Mol Neurosci. 2021;14. Accessed September 4, 2022. https://www.frontiersin.org/articles/10.3389/fnmol.2021.71303144.
44. Biogen. A phase 3, randomized, double-blind, sham-procedure controlled study to assess the clinical efficacy and safety of ISIS 396443 administered intrathecally in patients with later-onset spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02292537. Updated Feb 17, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/study/NCT02292537.
45. Why Spinraza/later-onset studies. SPINRAZA® (nusinersen) [Internet]. Accessed Mar 1, 2022. www.spinraza.com/en_us/home/why-spinraza/later-onset-studies.html#scroll-tabs.
46. Biogen. A Phase 3, Randomized, Double-Blind, Sham-Procedure Controlled Study to Assess the Clinical Efficacy and Safety of ISIS 396443 Administered Intrathecally in Patients With Infantile- Onset Spinal Muscular Atrophy. clinicaltrials.gov; 2021. Accessed February 10, 2022. https://clinicaltrials.gov/ct2/show/results/NCT02193074
47. Early-onset SMA (Type 1) | SPINRAZA® (nusinersen). Accessed Mar 1, 2022. https://www.spinraza-hcp.com/en_us/home/why-spinraza/about-spinraza.html.
48. Finkel RS et al; ENDEAR Study Group. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N Engl J Med. 2017;377(18):1723-32. doi: 10.1056/NEJMoa1702752.
49. Biogen. An open-label study to assess the efficacy, safety, tolerability, and pharmacokinetics of multiple doses of ISIS 396443 delivered intrathecally to subjects with genetically diagnosed and presymptomatic spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02386553. Updated Nov 18, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02386553.
50. De Vivo DC et al; NURTURE Study Group. Nusinersen initiated in infants during the presymptomatic stage of spinal muscular atrophy: In-terim efficacy and safety results from the phase 2 NURTURE study. Neuromuscul Disord. 2019 Nov;29(11):842-56. doi:10.1016/j.nmd.2019.09.007.
51. Why Spinraza/presymptomatic study. SPINRAZA® (nusinersen) [Internet]. Accessed Feb 22, 2022. www.spinraza.com/en_us/home/why-spinraza/presymptomatic-study.html#scroll-tabs.
52. FDA approves oral treatment for spinal muscular atrophy. U.S. Food and Drug Administration. News release. Aug 7, 2020. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-oral-treatment-spinal-muscular-atrophy.
53. Hoffmann-La Roche. A two-part seamless, open-label, multicenter study to investigate the safety, tolerability, pharmacokinetics, pharmaco-dynamics and efficacy of risdiplam (RO7034067) in infants with type 1 spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT02913482. Updated Jan 21, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02913482.
54. Hoffmann-La Roche. A two-part seamless, multi-center randomized, placebo-controlled, double-blind study to investigate the safety, tolera-bility, pharmacokinetics, pharmacodynamics and efficacy of risdiplam (RO7034067) in type 2 and 3 spinal muscular atrophy patients. Clinical-Trials.gov Identifier: NCT02908685. Updated Dec 28, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02908685.
55. Genentech. Genentech’s risdiplam shows significant improvement in survival and motor milestones in infants with type 1 spinal muscular atrophy (SMA). Press release. Apr 27, 2020. Accessed Mar 1, 2022. http://www.gene.com/media/press-releases/14847/2020-04-27/genentechs-risdiplam-shows-significant-i
56. Hoffmann-La Roche. An open-label study to investigate the safety, tolerability, and pharmacokinetics/pharmacodynamics of risdiplam (RO7034067) in adult and pediatric patients with spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT03032172. Updated Jan 27, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03032172.
57. Hoffmann-La Roche. An open-label study of risdiplam in infants with genetically diagnosed and presymptomatic spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT03779334. Updated Jan 27, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03779334.
58. McCall S. Update on Genentech/Roche Initiation of MANATEE Clinical Study. Cure SMA. Published October 20, 2021. Accessed August 20, 2022. https://www.curesma.org/update-on- genentech-roche-initiation-of-manatee-clinical-study/
59. Abati E, Manini A, Comi GP, Corti S. Inhibition of myostatin and related signaling pathways for the treatment of muscle atrophy in motor neuron diseases. Cell Mol Life Sci. 2022;79(7):374. doi:10.1007/s00018-022-04408-w
60. FDA approves innovative gene therapy to treat pediatric patients with spinal muscular atrophy, a rare disease and leading genetic cause of infant mortality. U.S. Food and Drug Administration. News release. May 24, 2019. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-innovative-gene-therapy-treat-pediatric-patients-spinal-muscular-atrophy-rare-disease.
61. Novartis Gene Therapies. Phase I gene transfer clinical trial for spinal muscular atrophy type 1 delivering AVXS-101. ClinicalTrials.gov Identifier: NCT02122952. Updated Jun 14, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02122952.
62. Novartis Gene Therapies. Phase 3, open-label, single-arm, single-dose gene replacement therapy clinical trial for patients with spinal mus-cular atrophy type 1 with one or two SMN2 copies delivering AVXS-101 by intravenous infusion. ClinicalTrials.gov Identifier: NCT03306277. Updated Jun 14, 2021. Accessed Feb 21, 2022. https://clinicaltrials.gov/ct2/show/NCT03306277.
63. Mendell JR et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N Engl J Med. 2017;377(18):1713-22. doi:10.1056/NEJMoa1706198.
64. Symptomatic study results. ZOLGENSMA [Internet]. Updated Nov 2021. Accessed Mar 1, 2022. Error! Hyperlink reference not valid..
65. Novartis Gene Therapies. A global study of a single, one-time dose of AVXS-101 delivered to infants with genetically diagnosed and pre-symptomatic spinal muscular atrophy with multiple copies of SMN2. ClinicalTrials.gov Identifier: NCT03505099. Updated Jan 1, 2022. Ac-cessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03505099.
66. Chiu W et al. Current genetics and potential gene-targeting therapeutics for neuromuscular diseases. Int J Mol Sci. 2020 Dec;21(24):9589. doi:10.3390/ijms21249589.
67. Novartis Gene Therapies. A long-term follow-up study of patients in the clinical trials for spinal muscular atrophy receiving AVXS-101. Clini-calTrials.gov Identifier: NCT04042025. Updated Jun 9, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04042025.
68. Novartis Gene Therapies. Phase 3, open-label, single-arm, single-dose gene replacement therapy clinical trial for patients with spinal mus-cular atrophy type 1 with one or two SMN2 copies delivering AVXS-101 by intravenous infusion. ClinicalTrials.gov Identifier: NCT0383718. Up-dated Jan 11, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03837184.
69. Biogen. An open-label, dose escalation study to assess the safety, tolerability and dose-range finding of multiple doses of ISIS 396443 de-livered intrathecally to patients with spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT01703988. Updated Apr 13, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01703988.
70. Biogen. A study to assess the efficacy, safety, tolerability, and pharmacokinetics of multiple doses of ISIS 396443 delivered intrathecally to patients with infantile-onset spinal muscular atrophy. ClinicalTrials.gov Identifier: NCT01839656. Updated Feb 17, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01839656.
71. Biogen. An open-label extension study for patients with spinal muscular atrophy who previously participated in investigational studies of ISIS 396443. ClinicalTrials.gov Identifier: NCT02594124. Updated Nov 15, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02594124.
72. Biogen. Escalating dose and randomized, controlled study of nusinersen (BIIB058) in participants with spinal muscular atrophy. ClinicalTri-als.gov Identifier: NCT04089566. Updated Feb 24, 2022. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04089566.
73. National Center for Advancing Translational Sciences. Duchenne muscular dystrophy. Genetic and Rare Diseases Information Center. Up-dated Nov 2, 2020. Accessed Mar 1, 2022. https://rarediseases.info.nih.gov/diseases/6291/duchenne-muscular-dystrophy.
74. Matsuo M. Antisense oligonucleotide-mediated exon-skipping therapies: Precision medicine spreading from Duchenne muscular dystrophy. JMA J. 2021 Jul 15;4(3):232-40. doi:10.31662/jmaj.2021-0019.
75. FDA approves drug to treat Duchenne muscular dystrophy. U.S. Food and Drug Administration. News release. Feb 9, 2017. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-drug-treat-duchenne-muscular-dystrophy.74.
76. Duan D. Dystrophin gene replacement and gene repair therapy for Duchenne muscular dystrophy in 2016: An interview. Hum Gene Ther Clin Dev. 2016 Mar;27(1):9-18. doi:10.1089/humc.2016.001.
77. EXONDYS 51®. Parent Project Muscular Dystrophy. Accessed August 21, 2022. https://www.parentprojectmd.org/drug-development-pipeline/exondys-51/
78. Sarepta Therapeutics, Inc. A Randomized, Double-Blind, Placebo-Controlled, Multiple Dose Efficacy, Safety, Tolerability and Pharmacoki-netics Study of AVI-4658(Eteplirsen),in the Treatment of Ambulant Subjects With Duchenne Muscular Dystrophy. clinicaltrials.gov; 2020. Ac-cessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT01396239
79. Sarepta Therapeutics, Inc. Clinical Study to Assess the Safety Fo AVI-4658 in Subjects With Duchenne Muscular Dystrophy Due to a Frame-Shift Mutation Amenable to Correction by Skipping Exon 51. clinicaltrials.gov; 2015. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/study/NCT00844597
80. Sarepta Therapeutics, Inc. A 2-part, randomized, double-blind, placebo-controlled, dose-titration, safety, tolerability, and pharmacokinetics study (Part 1) followed by an open-label efficacy and safety evaluation (Part 2) of SRP-4053 in patients with Duchenne muscular dystrophy amenable to exon 53 skipping. ClinicalTrials.gov Identifier: NCT02310906. Updated Oct 19, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/results/NCT02310906.
81. Commissioner O of the. FDA grants accelerated approval to first drug for Duchenne muscular dystrophy. FDA. Published March 24, 2020. Accessed August 21, 2022. hDuchenne Muscular Dystrophy Amenable to Exon 51-Skipping Treatment. clinicaltrials.gov; 2022. Accessed Au-gust 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04004065
109. National Center of Neurology and Psychiatry, Japan. Exploratory study of NS-065/NCNP-01 in Duchenne muscular dystrophy. ClinicalTri-als.gov Identifier: NCT02081625; Updated Feb 26, 2020. Accessed Mar 2, 2022. https://clinicaltrialsttps://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-first-drug-duchenne-muscular- dys-trophy
82. Duchenne Drug Development Pipeline. Parent Project Muscular Dystrophy. Accessed August 21, 2022. https://www.parentprojectmd.org/duchenne-drug-development-pipeline/
83. Sarepta Therapeutics Provides Update on SRP-5051 for the Treatment of Duchenne Muscular Dystrophy | Sarepta Therapeutics, Inc. Ac-cessed August 22, 2022. https://investorrelations.sarepta.com/news-releases/news-release-details/sarepta-therapeutics- pro-vides-update-srp-5051-treatment-duchenne
84. Sarepta Therapeutics, Inc. An Open-Label Extension Study for Patients With Duchenne Muscular Dystrophy Who Participated in Studies of SRP-5051. clinicaltrials.gov; 2021. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03675126
85. VYONDYS 53. Prescribing information. Sarepta Therapeutics Inc.; 2019. Accessed Mar 2, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211970s000lbl.pdf.
86. NS Pharma Inc. Long-term use of viltolarsen in boys with Duchenne muscular dystrophy in clinical practice (VILT-502). ClinicalTrials.gov Identifier: NCT04687020. Updated Nov 22, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04687020.
87. VILTEPSO. Prescribing information. NS Pharma; 2020. Accessed Mar 2, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2020/212154s000lbl.pdf.
88. FDA approves targeted treatment for rare Duchenne muscular dystrophy mutation. U.S. Food and Drug Administration. News release. Feb 25, 2021. Accessed Mar 1, 2022. http://www.fda.gov/news-events/press-announcements/fda-approves-targeted-treatment-rare-duchenne-muscular-dystrophy-mutation-0.
89. Sarepta Therapeutics Inc. A double-blind, placebo-controlled, multi-center study with an open-label extension to evaluate the efficacy and safety of SRP-4045 and SRP-4053 in patients with Duchenne muscular dystrophy. Clinicaltrials.gov Identifier: NCT02500381. Updated Aug 19, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT02500381.
90. AMONDYS 45. Prescribing information. Sarepta Therapeutics Inc.; 2021. Accessed Feb 22, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/label/2021/213026lbl.pdf.
91. Finkel RS et al. Phase 2a study of ataluren-mediated dystrophin production in patients with nonsense mutation Duchenne muscular dys-trophy. PLoS ONE. 2013;8(12):e81302. doi:10.1371/journal.pone.0081302.
92. PTC Therapeutics. A phase 2 study of PTC124 as an oral treatment for nonsense-mutation-mediated Duchenne muscular dystrophy. Clini-calTrials.gov Identifier: NCT00264888. Updated Jan 14, 2009. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT00264888.
93. PTC Therapeutics. A phase 2B efficacy and safety study of PTC124 in subjects with nonsense-mutation-mediated Duchenne and Becker muscular dystrophy. ClinicalTrials.gov Identifier: NCT00592553. Updated Apr 7, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT00592553.
94. PTC Therapeutics. A phase 3 efficacy and safety study of ataluren in patients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01826487. Updated Aug 4, 2020. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT01826487.
95. Bushby K et al; PTC124-GD-007-DMD Study Group. Ataluren treatment of patients with nonsense mutation dystrophinopathy. Muscle Nerve. 2014 Oct;50(4):477-87. doi:10.1002/mus.24332.
96. Solid Biosciences LLC. A randomized, controlled, open-label, single-ascending dose, phase I/II study to investigate the safety and tolerabil-ity, and efficacy of intravenous SGT-001 in male adolescents and children with Duchenne muscular dystrophy. ClinicalTrials.gov Identifier: NCT03368742. Updated Aug 24, 2021. Accessed Mar 1, 2022. https://clinicaltrials.gov/ct2/show/NCT03368742.
97. Solid Biosciences reports 1.5-year data from patients in the ongoing IGNITE DMD phase I/II clinical trial of SGT-001. Press release. Solid Biosciences. Sep 27, 2021. Accessed Mar 2, 2022. http://www.solidbio.com/about/media/press-releases/solid-biosciences-reports-1-5-year-data-from-patients-in-the-ongoing-ignite-dmd-phase-i-ii-clinical-trial-of-sgt-001.
98. Potter RA et al. Dose-escalation study of systemically delivered rAAVrh74.MHCK7.microdystrophin in the mdx mouse model of Duchenne muscular dystrophy. Hum Gene Ther. 2021 Apr;32(7-8):375-89. doi:10.1089/hum.2019.255.
99. Sarepta Therapeutics, Inc. A Phase 3 Multinational, Randomized, Double-Blind, Placebo- Controlled Systemic Gene Delivery Study to Evaluate the Safety and Efficacy of SRP-9001 in Patients With Duchenne Muscular Dystrophy (EMBARK). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT05096221
100. Pfizer. A PHASE 3, MULTICENTER, RANDOMIZED, DOUBLE-BLIND, PLACEBO CONTROLLED STUDY TO EVALUATE THE SAFETY AND EFFICACY OF PF 06939926 FOR THE TREATMENT OF DUCHENNE MUSCULAR DYSTROPHY. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04281485
101. Pfizer. A phase 1B multicenter open-label, single ascending dose study to evaluate the safety and tolerability of PF-06939926 in ambula-tory and non-ambulatory subjects with Duchenne muscular dystrophy. ClinicalTrials.gov Identifier: NCT03362502. Updated Mar 2, 2022. Ac-cessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03362502.
102. MS MW. Phase 3 CIFFREO DMD Gene Therapy Trial Slated to Begin in June in US. Accessed August 21, 2022. https://musculardystrophynews.com/news/phase-3-trial-of-pfizers-gene-therapy- expected-to-open-in-us-in-june/
103. SRP-9001. Parent Project Muscular Dystrophy. Accessed August 22, 2022. https://www.parentprojectmd.org/drug-development-pipeline/srp-9001-micro-dystrophin-gene- transfer/
104. Sarepta Therapeutics’ Investigational Gene Therapy SRP-9001 for Duchenne Muscular Dystrophy Demonstrates Significant Functional Improvements Across Multiple Studies | Sarepta Therapeutics, Inc. Accessed August 22, 2022. https://investorrelations.sarepta.com/news-releases/news-release- details/sarepta-therapeutics-investigational-gene-therapy-srp-9001
105. Sarepta Therapeutics, Inc. An Open-Label Safety, Tolerability, and Efficacy Study of Eteplirsen in Patients With Duchenne Muscular Dys-trophy Who Have Completed Study 4658-102.clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03985878
106. Sarepta Therapeutics, Inc. An Open-Label Safety, Tolerability, and Pharmacokinetics Study of Eteplirsen in Young Patients With Duchenne Mus-cular Dystrophy Amenable to Exon 51 Skipping. clinicaltrials.gov; 2021. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03218995
107.Sarepta Therapeutics, Inc. A Randomized, Double-Blind, Dose Finding and Comparison Study of the Safety and Efficacy of a High Dose of Eteplirsen, Preceded by an Open-Label Dose Escalation, in Patients With Duchenne Muscular Dystrophy With Deletion Mutations Amenable to Exon 51 Skipping. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03992430
108. Sarepta Therapeutics, Inc. A Phase 2, Two-Part, Multiple-Ascending-Dose Study of SRP-5051 for Dose Determination, Then Dose Ex-pansion, in Patients With .gov/ct2/show/NCT02081625.
110. NS Pharma Inc. A phase II, dose finding study to assess the safety, tolerability, pharmacokinetics, and pharmacodynamics of NS-065/NCNP-01 in boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT02740972. Updated Dec 7, 2021. Ac-cessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02740972.
111. NS Pharma Inc. A phase II, open-label, extension study to assess the safety and efficacy of NS-065/NCNP-01 in boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT03167255. Updated Nov 24, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03167255.
112. NS Pharma Inc. A phase 2 open label study to assess the safety, tolerability, and efficacy of viltolarsen in ambulant and non-ambulant boys with Duchenne muscular dystrophy (DMD) compared with natural history controls. ClinicalTrials.gov Identifier: NCT04956289. Updated Feb 1, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04956289.
113. NS Pharma Inc. A phase 3 randomized, double-blind, placebo-controlled, multi-center study to assess the efficacy and safety of viltolarsen in ambulant boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT04060199. Updated Nov 16, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04060199.
114. NS Pharma Inc. A phase 3, multi-center, open-label extension study to assess the safety and efficacy of viltolarsen in ambulant boys with Duchenne muscular dystrophy (DMD). ClinicalTrials.gov Identifier: NCT04768062. Updated Nov 16, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT04768062.
115. Sarepta Therapeutics Inc. A randomized, double-blind, placebo-controlled, dose-titration, safety, tolerability, and pharmacokinetics study followed by an open-label safety and efficacy evaluation of SRP-4045 in advanced-stage patients with Duchenne muscular dystrophy amena-ble to exon 45 skipping. ClinicalTrials.gov Identifier: NCT02530905. Updated May 17, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02530905.
116. Sarepta Therapeutics Inc. Long-term, open-label extension study for patients with Duchenne muscular dystrophy enrolled in clinical trials evaluating casimersen or golodirsen. ClinicalTrials.gov Identifier: NCT03532542. Updated Dec 20, 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03532542.
117. PTC Therapeutics. A phase 2 study of the safety, pharmacokinetics, and pharmacodynamics of ataluren (PTC124®) in patients aged ≥2 to <5 years old with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT02819557. Updated Aug 28, 2020. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT02819557.
118. PTC Therapeutics. Phase 2, non-interventional, clinical study to assess dystrophin levels in subjects with nonsense mutation Duchenne muscular dystrophy who have been treated with ataluren for ≥ 9 months. ClinicalTrials.gov Identifier: NCT03796637. Updated Apr 10, 2020. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03796637.
119. PTC Therapeutics. An Open-Label Study Evaluating the Safety and Pharmacokinetics of Ataluren in Children From ≥6 Months to <2 Years of Age With Nonsense Mutation Duchenne Muscular Dystrophy. clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04336826 120. PTC Therapeutics. An open-label study for previously treated ataluren (PTC124®) pa-tients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01557400. Updated Nov 25, 2020. Accessed Feb 21, 2022. https://clinicaltrials.gov/ct2/show/NCT01557400.
121. PTC Therapeutics. An open-label, safety study for ataluren (PTC124) patients with nonsense mutation dystrophinopathy. ClinicalTrials.gov Identifier: NCT01247207. Updated Feb 16, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT01247207.
122. PTC Therapeutics. A phase 3, randomized, double-blind, placebo-controlled efficacy and safety study of ataluren in patients with non-sense mutation Duchenne muscular dystrophy and open-label extension. ClinicalTrials.gov Identifier: NCT03179631. Updated Feb 8, 2022. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03179631.
123. Sarepta Therapeutics, Inc. An Open-Label, Systemic Gene Delivery Study Using Commercial Process Material to Evaluate the Safety of and Expression From SRP-9001 in Subjects With Duchenne Muscular Dystrophy (ENDEAVOR). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT04626674
124. Sarepta Therapeutics, Inc. Systemic Gene Delivery Phase I/IIa Clinical Trial for Duchenne Muscular Dystrophy Using RAA-Vrh74.MHCK7.Micro-Dystrophin (MicroDys-IV-001). clinicaltrials.gov; 2022. Accessed August 18, 2022. https://clinicaltrials.gov/ct2/show/NCT03375164
125. Sarepta Therapeutics Inc. A multicenter, randomized, double-blind, placebo-controlled trial for Duchenne muscular dystrophy using SRP-9001. ClinicalTrials.gov Identifier: NCT03769116. Updated Dec 2021. Accessed Mar 2, 2022. https://clinicaltrials.gov/ct2/show/NCT03769116.
126. Hoffmann-La Roche. A Two-Part, Seamless, Multi-Center, Randomized, Placebo-Controlled, Double-Blind Study to Investigate the Safety, Tolerability, Pharmacokinetics, Pharmacodynamics and Efficacy of RO7204239 in Combination With Risdiplam (RO7034067) in Ambulant Pa-tients With Spinal Muscular Atrophy. clinicaltrials.gov; 2022. Accessed September 1, 2022. https://clinicaltrials.gov/ct2/show/NCT05115110
NORD Rare Disease Centers of Excellence: A new network seeks to break down barriers in rare disease care
In November 2021, the National Organization for Rare Disorders (NORD) announced that it had designated 31 institutions across the United States as “NORD Rare Disease Centers of Excellence.” More than just a stamp of approval, the new NORD network aims to change the way rare diseases are diagnosed and treated, creating more efficient pathways for collaboration among physicians, while helping patients get better care closer to home.
To understand better how the nascent network can benefit patients and clinicians, Neurology Reviews/MDedge Neurology spoke with Ed Neilan, MD, PhD, NORD’s chief scientific and medical officer. Dr. Neilan, a pediatrician and geneticist, is a former president of the medical staff at Boston Children’s Hospital and also served as head of global medical affairs for rare neurology at Sanofi Genzyme.
How did NORD choose its 31 centers?
We were looking for places that had both broad capabilities and deep expertise, where it was reasonable to expect that a patient with almost any condition could go and, without too many missteps or delays, get the right diagnosis or the right treatment. We also sought sites that were educating the next generation of rare disease specialists across departments. The sites had to be involved in research, because that moves the field forward, and sometimes it’s the only way to get a really impactful treatment for the 95% of rare diseases that don’t have an FDA-approved treatment. NORD sent a letter inviting different centers to apply, along with an application that had 120 questions. Most of the questions sought information about what kinds of expertise or services were available on-site, so that patients don’t have to go elsewhere to get, let’s say, a brain MRI scan or to see an immunologist. We wanted each site to be a place where you could go for almost any problem, at any age, and expect that while you’re being seen, and receiving treatment, it can also contribute to the education of the next generation of rare disease specialists and to research.
Several of the members of the network comprise more than one institution: They’re a children’s hospital combined with another facility.
Children’s hospitals, which are highly specialized and able to care for rare things in children, couldn’t apply by themselves. They had to apply in partnership with a center that could provide adult care as patients got older; otherwise, their care model would be incomplete. We’ve had some small victories already just by asking these questions and outlining this sort of approach. At one institution in the Great Plains, the director told us that he had been trying for years to get permission to hire someone who could make appointments across three different hospitals – a children’s hospital and two adult hospitals. He’d wanted to ensure that patients with rare and genetic diseases were seen in the appropriate places, and thanks to the NORD designation, he finally can. Now, regardless of age, the same office staff can handle the arrangements, and the patient will be scheduled in the right place.
You make clear that these are different from disease-specific centers of excellence – you specifically chose the 31 centers for their breadth of expertise. There’s no way to represent all 7,000 rare diseases equally, and disease-specific centers of excellence, which already exist for hemophilia, muscular dystrophy, cystic fibrosis, and some other conditions, have a very important role. We’re not aiming to compete with any other existing resources. What we are seeking to do is to fill the unmet need of, “What if there are no such designations for the disease that you’re concerned about?” Our goal was to find places that could help with unanswered questions, whether diagnostic questions or treatment questions. To identify places where a patient could reasonably expect to go and have a deeper dive – maybe an interdisciplinary deep dive.
The delay to diagnosis can be years in rare diseases. How can the network help speed up diagnoses?
With all these experts on different diseases, we hope to develop some better diagnostic algorithms within the network. Another thing we can do is to share resources. With 31 sites, everybody’s seeing patients with unknown diagnoses. Everyone is seeing patients for whom they would maybe like to get a whole genome done, or a whole exome done, but they are often encountering stiff resistance from insurance companies.
Meanwhile some sites, but not all 31, have multimillion-dollar grants to do sequencing and other kinds of advanced diagnostic tests to solve unknown cases. And there are people at those sites who say, “We need more samples. Can you get us samples from the other sites?”
One of the main things we aim to do is share information, including information about available diagnostic resources. We want all 31 sites to know which sites have funding and programs that enable them to study samples for other sites. We also want to know what criteria they’re putting on it. Someone might say: “I’ve got a grant to sequence genomes for people with unexplained seizures. Send me all your unexplained seizures.” Somebody else might have a grant for unexplained GI diseases. So, we want to put on our intranet a resource for the 31 sites, kind of a cookbook for – when if you can’t get it paid for by insurance, but you really think you need a particular special test – who might be able to do it for you within the network.
This would seem to benefit research across sites as well.
Yes, but we also want to share clinical advice and expertise for direct patient benefit. So, it doesn’t always have to fulfill the goals of a specific research project. For example, we might be able to create an undiagnosed patient quality improvement database across all 31 sites that could compliantly let Drs. X and Y know that they’re each seeing a patient with the same rare thing.
But let’s say you want to move the field forward by discovering a new disease. Rare genetic diseases are now being discovered at the rate of about 250 a year, so about 5 per week across the world. With two or three unrelated patients who have the same disease and a whole exome sequence, you can potentially discover a disease. Maybe you’ve found one unique patient with a genetic variant of possible significance, but you can’t be 100% sure, and you may not be able to convince your colleagues, or journal editors, until you find other cases. You need those two or three ultrarare patients. Within this network, a lot of sites want to share information about their ultrarare patients and be able to put together additional instances of the same thing, to prove that it is a real disease, to learn more about it and how to diagnose, manage, and treat it.
Part of the idea with a nationwide network is that patients aren’t going to have to move around among these centers of excellence, is that correct? They’re going to be seen at the closest ones, and it’s the expertise that is mobile.
Yes, that’s right. While we can’t eliminate the need for travel, what we are trying to do is increase the sharing of expertise, to improve results for patients while limiting the need for traveling very long distances. As a geneticist I’ve been on both the requesting and the receiving end of consultations with doctors at other sites, sometimes very far away, especially for ultrarare conditions for which any one physician’s experience is limited. We all try to honor these sorts of requests, but insurance doesn’t reimburse it and so hospitals don’t give doctors much credit for it.
We want to ultimately find ways to incentivize this type of collaboration. Hopefully we can get agreements with insurance companies to allow intersite consultations within our network, recognizing that they don’t want to pay for the patient to be seen out of state, but you also want the patient to get the best possible medical advice. This might require legislative changes in the long run. But what we can do more readily is create a culture within this network of mutual consultation and sharing of clinical experience. Outside of such a network, the idea of “cold calling” somebody, whom you may never have met, and asking them for help and free advice is a little bit of a bar, right? We want to lower that bar.
Can patients get telemedicine consults with physicians across the network?
NORD supports having telemedicine options for everybody regardless of diagnosis, rare or not, and we support legislation that would continue access and reimbursement for telemedicine post pandemic. I hope we can get that, or at least preserve telemedicine for rare diseases, for which there are often not enough, or sometimes not any, expert providers in the same state. Ultimately, we want patients to be able to get the expert assessments and advice they need. For rare diseases, that sometimes means battling back and forth with an insurance provider, seeking permission to see an expert clinician a thousand miles away. By sharing medical expertise, and through telemedicine when that’s allowed, we hope to reduce the need for that. But the telemedicine environment is still evolving and somewhat uncertain.
How will the network’s physician collaborations take place?
One of the important things NORD is providing to the network is an information technology setup and intranet across the 31 sites. That intranet is where center staff will go to access the network’s internal resources, including live and recorded case conferences. In those case conferences you can present a case you haven’t been able to solve. Experts you may have only heard of by reputation will now be streamed to your computer as part of the nationwide network. It benefits the patient because you get additional expert opinions, but it also benefits the physicians because we have this collegial space for discussion and learning. We’ll be linked by frequent meetings – some in person, most virtual – a common culture, and a common intranet.
On the intranet, we will also have a growing set of useful databases, links, and documents that are available to all members. These will be progressively updated with help from experts at the centers, so that clinicians can more directly learn from each other, instead of separately reinventing the wheel. The way things usually work, when you see a patient with an ultrarare condition that you’re not that familiar with, is that you tell them what little you can, then schedule them to come back in a few weeks. In the meantime, usually in your off time, you spend hours searching PubMed and other sources and you try to piece things together, to figure out what’s known that might help your patient. But imagine that this has already been figured out by someone else in the network. You can see on the network a list of articles the other expert read and found helpful in addressing this problem. And you then reach out directly to that other expert.
In recent months you’ve had one-on-one meetings with all 31 directors at the sites, and after that you convened 11 working groups. What are you trying to achieve?
Once the sites were chosen, we aimed to talk quickly and honestly about what everyone needed, what everyone saw as the biggest problems to tackle in rare diseases. Two things were very rewarding about those phone calls: one, all the centers were very enthusiastic, and two, they pretty much all agreed on what the key unmet needs are for rare disease patients and the practitioners trying to help them. So, we empaneled working groups of expert volunteers enthusiastic to work on each of those problems. These groups collectively comprise more than 200 volunteers – faculty, staff, and trainees – from the different sites nationwide. Each group is working on a key unmet need in rare diseases, and each group will be given its own space on our file-sharing platform, where they can share information and co-develop new ideas and documents. When something they produce is good enough to start to be a practice resource, such as a draft treatment guideline that the working group now wants to try in the real world, but it’s not yet ready to be published, they can share it and have it tested by all 31 sites through the dedicated intranet we are building for the network.
Jennie Smith is a freelance journalist specializing in medicine and health.
In November 2021, the National Organization for Rare Disorders (NORD) announced that it had designated 31 institutions across the United States as “NORD Rare Disease Centers of Excellence.” More than just a stamp of approval, the new NORD network aims to change the way rare diseases are diagnosed and treated, creating more efficient pathways for collaboration among physicians, while helping patients get better care closer to home.
To understand better how the nascent network can benefit patients and clinicians, Neurology Reviews/MDedge Neurology spoke with Ed Neilan, MD, PhD, NORD’s chief scientific and medical officer. Dr. Neilan, a pediatrician and geneticist, is a former president of the medical staff at Boston Children’s Hospital and also served as head of global medical affairs for rare neurology at Sanofi Genzyme.
How did NORD choose its 31 centers?
We were looking for places that had both broad capabilities and deep expertise, where it was reasonable to expect that a patient with almost any condition could go and, without too many missteps or delays, get the right diagnosis or the right treatment. We also sought sites that were educating the next generation of rare disease specialists across departments. The sites had to be involved in research, because that moves the field forward, and sometimes it’s the only way to get a really impactful treatment for the 95% of rare diseases that don’t have an FDA-approved treatment. NORD sent a letter inviting different centers to apply, along with an application that had 120 questions. Most of the questions sought information about what kinds of expertise or services were available on-site, so that patients don’t have to go elsewhere to get, let’s say, a brain MRI scan or to see an immunologist. We wanted each site to be a place where you could go for almost any problem, at any age, and expect that while you’re being seen, and receiving treatment, it can also contribute to the education of the next generation of rare disease specialists and to research.
Several of the members of the network comprise more than one institution: They’re a children’s hospital combined with another facility.
Children’s hospitals, which are highly specialized and able to care for rare things in children, couldn’t apply by themselves. They had to apply in partnership with a center that could provide adult care as patients got older; otherwise, their care model would be incomplete. We’ve had some small victories already just by asking these questions and outlining this sort of approach. At one institution in the Great Plains, the director told us that he had been trying for years to get permission to hire someone who could make appointments across three different hospitals – a children’s hospital and two adult hospitals. He’d wanted to ensure that patients with rare and genetic diseases were seen in the appropriate places, and thanks to the NORD designation, he finally can. Now, regardless of age, the same office staff can handle the arrangements, and the patient will be scheduled in the right place.
You make clear that these are different from disease-specific centers of excellence – you specifically chose the 31 centers for their breadth of expertise. There’s no way to represent all 7,000 rare diseases equally, and disease-specific centers of excellence, which already exist for hemophilia, muscular dystrophy, cystic fibrosis, and some other conditions, have a very important role. We’re not aiming to compete with any other existing resources. What we are seeking to do is to fill the unmet need of, “What if there are no such designations for the disease that you’re concerned about?” Our goal was to find places that could help with unanswered questions, whether diagnostic questions or treatment questions. To identify places where a patient could reasonably expect to go and have a deeper dive – maybe an interdisciplinary deep dive.
The delay to diagnosis can be years in rare diseases. How can the network help speed up diagnoses?
With all these experts on different diseases, we hope to develop some better diagnostic algorithms within the network. Another thing we can do is to share resources. With 31 sites, everybody’s seeing patients with unknown diagnoses. Everyone is seeing patients for whom they would maybe like to get a whole genome done, or a whole exome done, but they are often encountering stiff resistance from insurance companies.
Meanwhile some sites, but not all 31, have multimillion-dollar grants to do sequencing and other kinds of advanced diagnostic tests to solve unknown cases. And there are people at those sites who say, “We need more samples. Can you get us samples from the other sites?”
One of the main things we aim to do is share information, including information about available diagnostic resources. We want all 31 sites to know which sites have funding and programs that enable them to study samples for other sites. We also want to know what criteria they’re putting on it. Someone might say: “I’ve got a grant to sequence genomes for people with unexplained seizures. Send me all your unexplained seizures.” Somebody else might have a grant for unexplained GI diseases. So, we want to put on our intranet a resource for the 31 sites, kind of a cookbook for – when if you can’t get it paid for by insurance, but you really think you need a particular special test – who might be able to do it for you within the network.
This would seem to benefit research across sites as well.
Yes, but we also want to share clinical advice and expertise for direct patient benefit. So, it doesn’t always have to fulfill the goals of a specific research project. For example, we might be able to create an undiagnosed patient quality improvement database across all 31 sites that could compliantly let Drs. X and Y know that they’re each seeing a patient with the same rare thing.
But let’s say you want to move the field forward by discovering a new disease. Rare genetic diseases are now being discovered at the rate of about 250 a year, so about 5 per week across the world. With two or three unrelated patients who have the same disease and a whole exome sequence, you can potentially discover a disease. Maybe you’ve found one unique patient with a genetic variant of possible significance, but you can’t be 100% sure, and you may not be able to convince your colleagues, or journal editors, until you find other cases. You need those two or three ultrarare patients. Within this network, a lot of sites want to share information about their ultrarare patients and be able to put together additional instances of the same thing, to prove that it is a real disease, to learn more about it and how to diagnose, manage, and treat it.
Part of the idea with a nationwide network is that patients aren’t going to have to move around among these centers of excellence, is that correct? They’re going to be seen at the closest ones, and it’s the expertise that is mobile.
Yes, that’s right. While we can’t eliminate the need for travel, what we are trying to do is increase the sharing of expertise, to improve results for patients while limiting the need for traveling very long distances. As a geneticist I’ve been on both the requesting and the receiving end of consultations with doctors at other sites, sometimes very far away, especially for ultrarare conditions for which any one physician’s experience is limited. We all try to honor these sorts of requests, but insurance doesn’t reimburse it and so hospitals don’t give doctors much credit for it.
We want to ultimately find ways to incentivize this type of collaboration. Hopefully we can get agreements with insurance companies to allow intersite consultations within our network, recognizing that they don’t want to pay for the patient to be seen out of state, but you also want the patient to get the best possible medical advice. This might require legislative changes in the long run. But what we can do more readily is create a culture within this network of mutual consultation and sharing of clinical experience. Outside of such a network, the idea of “cold calling” somebody, whom you may never have met, and asking them for help and free advice is a little bit of a bar, right? We want to lower that bar.
Can patients get telemedicine consults with physicians across the network?
NORD supports having telemedicine options for everybody regardless of diagnosis, rare or not, and we support legislation that would continue access and reimbursement for telemedicine post pandemic. I hope we can get that, or at least preserve telemedicine for rare diseases, for which there are often not enough, or sometimes not any, expert providers in the same state. Ultimately, we want patients to be able to get the expert assessments and advice they need. For rare diseases, that sometimes means battling back and forth with an insurance provider, seeking permission to see an expert clinician a thousand miles away. By sharing medical expertise, and through telemedicine when that’s allowed, we hope to reduce the need for that. But the telemedicine environment is still evolving and somewhat uncertain.
How will the network’s physician collaborations take place?
One of the important things NORD is providing to the network is an information technology setup and intranet across the 31 sites. That intranet is where center staff will go to access the network’s internal resources, including live and recorded case conferences. In those case conferences you can present a case you haven’t been able to solve. Experts you may have only heard of by reputation will now be streamed to your computer as part of the nationwide network. It benefits the patient because you get additional expert opinions, but it also benefits the physicians because we have this collegial space for discussion and learning. We’ll be linked by frequent meetings – some in person, most virtual – a common culture, and a common intranet.
On the intranet, we will also have a growing set of useful databases, links, and documents that are available to all members. These will be progressively updated with help from experts at the centers, so that clinicians can more directly learn from each other, instead of separately reinventing the wheel. The way things usually work, when you see a patient with an ultrarare condition that you’re not that familiar with, is that you tell them what little you can, then schedule them to come back in a few weeks. In the meantime, usually in your off time, you spend hours searching PubMed and other sources and you try to piece things together, to figure out what’s known that might help your patient. But imagine that this has already been figured out by someone else in the network. You can see on the network a list of articles the other expert read and found helpful in addressing this problem. And you then reach out directly to that other expert.
In recent months you’ve had one-on-one meetings with all 31 directors at the sites, and after that you convened 11 working groups. What are you trying to achieve?
Once the sites were chosen, we aimed to talk quickly and honestly about what everyone needed, what everyone saw as the biggest problems to tackle in rare diseases. Two things were very rewarding about those phone calls: one, all the centers were very enthusiastic, and two, they pretty much all agreed on what the key unmet needs are for rare disease patients and the practitioners trying to help them. So, we empaneled working groups of expert volunteers enthusiastic to work on each of those problems. These groups collectively comprise more than 200 volunteers – faculty, staff, and trainees – from the different sites nationwide. Each group is working on a key unmet need in rare diseases, and each group will be given its own space on our file-sharing platform, where they can share information and co-develop new ideas and documents. When something they produce is good enough to start to be a practice resource, such as a draft treatment guideline that the working group now wants to try in the real world, but it’s not yet ready to be published, they can share it and have it tested by all 31 sites through the dedicated intranet we are building for the network.
Jennie Smith is a freelance journalist specializing in medicine and health.
In November 2021, the National Organization for Rare Disorders (NORD) announced that it had designated 31 institutions across the United States as “NORD Rare Disease Centers of Excellence.” More than just a stamp of approval, the new NORD network aims to change the way rare diseases are diagnosed and treated, creating more efficient pathways for collaboration among physicians, while helping patients get better care closer to home.
To understand better how the nascent network can benefit patients and clinicians, Neurology Reviews/MDedge Neurology spoke with Ed Neilan, MD, PhD, NORD’s chief scientific and medical officer. Dr. Neilan, a pediatrician and geneticist, is a former president of the medical staff at Boston Children’s Hospital and also served as head of global medical affairs for rare neurology at Sanofi Genzyme.
How did NORD choose its 31 centers?
We were looking for places that had both broad capabilities and deep expertise, where it was reasonable to expect that a patient with almost any condition could go and, without too many missteps or delays, get the right diagnosis or the right treatment. We also sought sites that were educating the next generation of rare disease specialists across departments. The sites had to be involved in research, because that moves the field forward, and sometimes it’s the only way to get a really impactful treatment for the 95% of rare diseases that don’t have an FDA-approved treatment. NORD sent a letter inviting different centers to apply, along with an application that had 120 questions. Most of the questions sought information about what kinds of expertise or services were available on-site, so that patients don’t have to go elsewhere to get, let’s say, a brain MRI scan or to see an immunologist. We wanted each site to be a place where you could go for almost any problem, at any age, and expect that while you’re being seen, and receiving treatment, it can also contribute to the education of the next generation of rare disease specialists and to research.
Several of the members of the network comprise more than one institution: They’re a children’s hospital combined with another facility.
Children’s hospitals, which are highly specialized and able to care for rare things in children, couldn’t apply by themselves. They had to apply in partnership with a center that could provide adult care as patients got older; otherwise, their care model would be incomplete. We’ve had some small victories already just by asking these questions and outlining this sort of approach. At one institution in the Great Plains, the director told us that he had been trying for years to get permission to hire someone who could make appointments across three different hospitals – a children’s hospital and two adult hospitals. He’d wanted to ensure that patients with rare and genetic diseases were seen in the appropriate places, and thanks to the NORD designation, he finally can. Now, regardless of age, the same office staff can handle the arrangements, and the patient will be scheduled in the right place.
You make clear that these are different from disease-specific centers of excellence – you specifically chose the 31 centers for their breadth of expertise. There’s no way to represent all 7,000 rare diseases equally, and disease-specific centers of excellence, which already exist for hemophilia, muscular dystrophy, cystic fibrosis, and some other conditions, have a very important role. We’re not aiming to compete with any other existing resources. What we are seeking to do is to fill the unmet need of, “What if there are no such designations for the disease that you’re concerned about?” Our goal was to find places that could help with unanswered questions, whether diagnostic questions or treatment questions. To identify places where a patient could reasonably expect to go and have a deeper dive – maybe an interdisciplinary deep dive.
The delay to diagnosis can be years in rare diseases. How can the network help speed up diagnoses?
With all these experts on different diseases, we hope to develop some better diagnostic algorithms within the network. Another thing we can do is to share resources. With 31 sites, everybody’s seeing patients with unknown diagnoses. Everyone is seeing patients for whom they would maybe like to get a whole genome done, or a whole exome done, but they are often encountering stiff resistance from insurance companies.
Meanwhile some sites, but not all 31, have multimillion-dollar grants to do sequencing and other kinds of advanced diagnostic tests to solve unknown cases. And there are people at those sites who say, “We need more samples. Can you get us samples from the other sites?”
One of the main things we aim to do is share information, including information about available diagnostic resources. We want all 31 sites to know which sites have funding and programs that enable them to study samples for other sites. We also want to know what criteria they’re putting on it. Someone might say: “I’ve got a grant to sequence genomes for people with unexplained seizures. Send me all your unexplained seizures.” Somebody else might have a grant for unexplained GI diseases. So, we want to put on our intranet a resource for the 31 sites, kind of a cookbook for – when if you can’t get it paid for by insurance, but you really think you need a particular special test – who might be able to do it for you within the network.
This would seem to benefit research across sites as well.
Yes, but we also want to share clinical advice and expertise for direct patient benefit. So, it doesn’t always have to fulfill the goals of a specific research project. For example, we might be able to create an undiagnosed patient quality improvement database across all 31 sites that could compliantly let Drs. X and Y know that they’re each seeing a patient with the same rare thing.
But let’s say you want to move the field forward by discovering a new disease. Rare genetic diseases are now being discovered at the rate of about 250 a year, so about 5 per week across the world. With two or three unrelated patients who have the same disease and a whole exome sequence, you can potentially discover a disease. Maybe you’ve found one unique patient with a genetic variant of possible significance, but you can’t be 100% sure, and you may not be able to convince your colleagues, or journal editors, until you find other cases. You need those two or three ultrarare patients. Within this network, a lot of sites want to share information about their ultrarare patients and be able to put together additional instances of the same thing, to prove that it is a real disease, to learn more about it and how to diagnose, manage, and treat it.
Part of the idea with a nationwide network is that patients aren’t going to have to move around among these centers of excellence, is that correct? They’re going to be seen at the closest ones, and it’s the expertise that is mobile.
Yes, that’s right. While we can’t eliminate the need for travel, what we are trying to do is increase the sharing of expertise, to improve results for patients while limiting the need for traveling very long distances. As a geneticist I’ve been on both the requesting and the receiving end of consultations with doctors at other sites, sometimes very far away, especially for ultrarare conditions for which any one physician’s experience is limited. We all try to honor these sorts of requests, but insurance doesn’t reimburse it and so hospitals don’t give doctors much credit for it.
We want to ultimately find ways to incentivize this type of collaboration. Hopefully we can get agreements with insurance companies to allow intersite consultations within our network, recognizing that they don’t want to pay for the patient to be seen out of state, but you also want the patient to get the best possible medical advice. This might require legislative changes in the long run. But what we can do more readily is create a culture within this network of mutual consultation and sharing of clinical experience. Outside of such a network, the idea of “cold calling” somebody, whom you may never have met, and asking them for help and free advice is a little bit of a bar, right? We want to lower that bar.
Can patients get telemedicine consults with physicians across the network?
NORD supports having telemedicine options for everybody regardless of diagnosis, rare or not, and we support legislation that would continue access and reimbursement for telemedicine post pandemic. I hope we can get that, or at least preserve telemedicine for rare diseases, for which there are often not enough, or sometimes not any, expert providers in the same state. Ultimately, we want patients to be able to get the expert assessments and advice they need. For rare diseases, that sometimes means battling back and forth with an insurance provider, seeking permission to see an expert clinician a thousand miles away. By sharing medical expertise, and through telemedicine when that’s allowed, we hope to reduce the need for that. But the telemedicine environment is still evolving and somewhat uncertain.
How will the network’s physician collaborations take place?
One of the important things NORD is providing to the network is an information technology setup and intranet across the 31 sites. That intranet is where center staff will go to access the network’s internal resources, including live and recorded case conferences. In those case conferences you can present a case you haven’t been able to solve. Experts you may have only heard of by reputation will now be streamed to your computer as part of the nationwide network. It benefits the patient because you get additional expert opinions, but it also benefits the physicians because we have this collegial space for discussion and learning. We’ll be linked by frequent meetings – some in person, most virtual – a common culture, and a common intranet.
On the intranet, we will also have a growing set of useful databases, links, and documents that are available to all members. These will be progressively updated with help from experts at the centers, so that clinicians can more directly learn from each other, instead of separately reinventing the wheel. The way things usually work, when you see a patient with an ultrarare condition that you’re not that familiar with, is that you tell them what little you can, then schedule them to come back in a few weeks. In the meantime, usually in your off time, you spend hours searching PubMed and other sources and you try to piece things together, to figure out what’s known that might help your patient. But imagine that this has already been figured out by someone else in the network. You can see on the network a list of articles the other expert read and found helpful in addressing this problem. And you then reach out directly to that other expert.
In recent months you’ve had one-on-one meetings with all 31 directors at the sites, and after that you convened 11 working groups. What are you trying to achieve?
Once the sites were chosen, we aimed to talk quickly and honestly about what everyone needed, what everyone saw as the biggest problems to tackle in rare diseases. Two things were very rewarding about those phone calls: one, all the centers were very enthusiastic, and two, they pretty much all agreed on what the key unmet needs are for rare disease patients and the practitioners trying to help them. So, we empaneled working groups of expert volunteers enthusiastic to work on each of those problems. These groups collectively comprise more than 200 volunteers – faculty, staff, and trainees – from the different sites nationwide. Each group is working on a key unmet need in rare diseases, and each group will be given its own space on our file-sharing platform, where they can share information and co-develop new ideas and documents. When something they produce is good enough to start to be a practice resource, such as a draft treatment guideline that the working group now wants to try in the real world, but it’s not yet ready to be published, they can share it and have it tested by all 31 sites through the dedicated intranet we are building for the network.
Jennie Smith is a freelance journalist specializing in medicine and health.
Spinal muscular atrophy: Patient care in the age of genetically targeted therapy
In 2016, the U.S. Food and Drug Administration approved nusinersen, the first treatment for spinal muscular atrophy (SMA). Until then, SMA had a mortality rate nearly double that of the general population.1 Two-thirds of patients were symptomatic within 6 months of birth and, in the absence of mechanical ventilation and other support, had a nearly 100% mortality rate by age 2.2
Five years later, there are three approved treatments for SMA, all of which have been shown to slow or even halt disease progression in many patients. Neurologists, whose SMA patient population once consisted almost entirely of children, are now treating more adults with the disease. Indeed, more than half of all people alive with SMA in the United States today are adults, according to Cure SMA.
“Managing SMA used to be clinic follow-ups where we were doing our best supportive care and watching people fall apart before our eyes,” said John Brandsema, MD, a physician and neuromuscular section head at the Children’s Hospital of Philadelphia. “Today, what we see in the vast majority of people is that they are either the same as they were before – which is completely against the natural history of this disease and something to be celebrated – or that people are really better with their function. It totally changes everything in the clinic.”
Among those changes are a more proactive approach to rehabilitation and an even greater emphasis on personalized medicine and multidisciplinary care. But there is also a need for updated treatment guidelines, a new classification system to measure disease severity, specific biomarkers to guide therapy choices, more data on long-term efficacy of existing therapeutics, new medications to complement those therapies, and a deeper understanding of a disease that may have treatment options but still has no cure.
Advances in early diagnosis
Patients with SMA lack a working copy of the survival motor neuron 1 (SMN1) gene, which provides instructions for producing a protein called SMN that is critical for the maintenance and function of motor neurons. Without this protein, motor neurons eventually die, causing debilitating and progressive muscle weakness that affects the ability to walk, eat, and breathe. SMA is rare, affecting about 1 in 10,000 newborns.
In approximately 96% of patients, SMA is caused by homozygous loss of the SMN1 gene. People with SMA have at least one copy of the SMN2 gene, sometimes called a “backup” gene, that also produces SMN protein. However, a single nucleotide difference between SMN2 and SMN1 causes about 90% of the protein produced by SMN2 to be truncated and less stable. Even with multiple copies of SMN2 present, as is the case with many infants with SMA, the amount of functional protein produced isn’t enough to compensate for the loss of SMN1.3
All three approved medications are SMN up-regulators and work to increase the amount of functional SMN protein. Starting these medications early, even before symptoms present, is critical to preserve motor function. Early treatment depends on early diagnosis, which became more widespread after 2018 when SMA was added to the federally Recommended Uniform Screening Panel for newborns. As of July 1, 2022, 47 states have incorporated SMA newborn screening into their state panel, ensuring that 97% of all infants born in the United States undergo SMA screening shortly after birth. Screening in the remaining states – Hawaii, Nevada, and South Carolina – and Washington, D.C. is expected by mid-2023.
SMA newborn screening is a PCR-based assay that detects homozygous SMN1 gene deletion found in about 95% of all people with SMA. The remaining 5% of cases are caused by various genetic mutations that can only be detected with gene sequencing. In these cases, and in children who don’t undergo SMA newborn screening, the disease is usually identified when symptoms are noticed by a parent, pediatrician, or primary care provider. But a study found that in 2018 only 52.7% of pediatricians correctly identified genetic testing as a requirement for a definitive diagnosis of SMA; in 2019, with a larger sample size, that number decreased to 45%.4 The lack of awareness of diagnostic requirements for SMA could contribute to delays in diagnosis, said Mary Schroth, MD, chief medical officer for Cure SMA and a coauthor of the study.
“In our world, suspicion of SMA in an infant is an emergency situation,” Dr. Schroth said. “These babies need to be referred immediately and have genetic testing so that treatment can begin as soon as possible.”
Based on the study findings, Dr. Schroth and others with Cure SMA launched a new tool in 2021 designed to help pediatricians, primary care physicians, and parents identify early signs of SMA, so that a referral to a pediatric neurologist happens quickly. Called SMArt Moves, the educational resource features videos and a checklist to help increase early detection in infants who had a negative SMA newborn screening result or did not receive SMA screening at birth.5
Who to treat, when, and with which treatment
For many patients, having multiple effective treatment options means that SMA is no longer a fatal disease in early childhood, but one that can be managed into adolescence and adulthood. The question for clinicians is, who do they treat, when, and with which treatment?
Studies have long shown that the number of copies of the backup gene that a patient has is inversely associated with disease severity.6 In 2018, a group of SMA experts published a treatment algorithm to help guide decision-making following a positive SMA newborn screening.7 The treatment guidelines were updated in 2020 based on clinical trial data for presymptomatic infants, and current recommendations include immediate treatment for infants with two to four copies of the SMN2 gene.8 For patients with only one copy of SMN2, most of whom will likely be symptomatic at birth, the guidelines recommend that treatment decisions be made jointly between the clinician and the family.7,8
Some suggest that the number of SMN2 copies a patient has should also be a factor in determining phenotype, which has started a conversation on the development of a new classification system.9 The original classification system for disease severity – Types 0-4 – was based on age of onset and degree of motor function achieved, with Type 0 developing prenatally and being the most severe and Type 4 developing in adulthood. Type 1 is the most common, affecting more than half of all people with SMA, followed by Types 2-4. In 2018, updated consensus care guidelines offered a revised classification system that better reflected disease progression in the age of therapy. The functional motor outcomes include nonsitters (historically Type I), sitters (historically Type 2/3), and walkers (historically Type 3/4).10,11 These guidelines are a start, but clinicians say more revision is needed.
“Types 1, 2, 3, 4 were based on function – getting to a certain point and then losing it, but now that we can treat this disease, people will shift categories based on therapeutic response or based on normal development that is possible now that the neurologic piece has been stabilized,” Dr. Brandsema said. “We need to completely change our thinking around all these different aspects of SMA management.”
While discussions of a new classification system for SMA are underway, another effort to update treatment recommendations is closer to completion. Led by Cure SMA, a group of about 50 physician experts in the United States and Europe who specialize in SMA are revising guidelines for diagnosis and treatment, the first time the recommendations have been updated since 2018. The updated recommendations, which should be published later this year, will focus on diagnosis and treatment considerations.
“We have three treatments that are available, and there are specific FDA indications for each of those, but it’s not totally clear just how those medications should be used or applied to different clinical situations,” said Dr. Schroth. “We’re in a rapid phase of learning right now in the SMA community, trying to understand how these treatments alter physiology and disease outcomes and how to best use the tools that we now have available to us. In parallel with clinical treatments, we have to be doing the best care we can to optimize the outcomes for those treatments.”
Research advances in 2021
Although all three drugs approved to treat SMA – nusinersen (Spinraza; Biogen), onasemnogene abeparvovec-xioi gene replacement therapy (Zolgensma; Novartis Gene Therapies), and risdiplam (Evrysdi, Genentech/Roche) – are highly effective, there are still unanswered questions and unmet needs. New research findings from 2021 focused on higher dosing, different drug-delivery methods, combination therapy, and complementary therapeutics to address SMA comorbidities.
Higher-dose nusinersen. The first drug approved to treat SMA, nusinersen is an antisense oligonucleotide approved for all ages and all SMA types. It works by altering splicing of the SMN2 gene pre-mRNA to make more complete SMN protein. Given as an intrathecal (IT) injection, four “loading doses” are administered within the first 2 months of treatment, followed by a maintenance dose every 4 months for the duration of the individual’s life.
Reports from patients of waning effects of nusinersen just prior to follow-up treatment have led some clinicians to ask if a higher dose may be needed. A study underway seeks to address that issue.
DEVOTE is a phase 2/3 trial to study the safety and efficacy of high-dose nusinersen in patients with SMA. Preliminary findings reported in 2021 found no adverse events among patients treated with 28 mg of nusinersen for 161-257 days.12 Another analysis from this trial found that higher doses are associated with greater decrease of plasma phosphorylated neurofilament heavy chain (pNF-H) levels in patients with SMA and may lead to clinically meaningful improvement in motor function beyond that observed with the approved 12 mg dose.13 The trial is ongoing.
Another trial, ASCEND, is a phase 3B study assessing higher dose nusinersen in patients previously treated with risdiplam. Recruitment for that trial began in October 2021.
Long-term efficacy and IT administration of SMA therapy. Several studies are looking at the long-term efficacy and alternate routes of administration of onasemnogene abeparvovec and other SMA therapies.
A one-time gene replacement therapy delivered via an IV infusion replaces the function of the missing or nonworking SMN1 gene with a new, working copy of the SMN1 gene. FDA approved in 2019, it is authorized for use in patients with SMA up to 2 years of age.
The latest data from an ongoing, long-term follow-up safety study of onasemnogene abeparvovec, published in May 2021, suggest that the treatment’s effects persist more than 5 years after treatment. Researchers followed 13 infants with symptomatic SMA type 1 since the beginning of the phase 1 clinical trial of the gene transfer therapy. All patients who received the therapeutic dose maintained their baseline motor function, and two of the patients actually improved without other SMN-targeted treatment. At a median 6.2 years after they received treatment, all were alive and none needed permanent ventilation.14
After a 2-year hold by the FDA, a study of IT administration of onasemnogene abeparvovec is now enrolling patients. Citing concerns from animal studies that IT administration might result in dorsal root ganglia injury, the FDA issued a partial hold on the STRONG trial in 2019. Following positive study results in nonhuman primates, the FDA announced the trial can continue. Novartis is launching a new phase 3 STEER trial to test the drug delivered intrathecally in patients aged 2-18 years with Type 2 SMA. IT administration could allow the gene therapy to be used safely and effectively in more patients with SMA.
Efficacy of risdiplam in more patients. The first oral treatment for SMA was approved by the FDA in 2020. It’s given once per day in patients with SMA of all ages and disease types. The drug increases functional SMN protein production by the SMN2 gene.
A July 2021 publication of the results of the FIREFISH study found that infants with Type I SMA treated with risdiplam for 12 months were significantly more likely to achieve motor milestones, such as sitting without support, compared with untreated infants with Type 1 SMA.15 Risdiplam is also effective in older patients with Type 2 or 3 SMA, according to results published in December from the SUNFISH clinical trial.16 Another study, RAINBOWFISH, is studying safety and efficacy at 24 months in presymptomatic infants started on treatment at up to 6 weeks of age.
The efficacy of risdiplam in previously treated patients is the subject of JEWELFISH, an ongoing study in patients 6 months to 60 years with SMA. Preliminary data presented at the 2020 Virtual SMA Research and Clinical Care Meeting suggest treatment with risdiplam led to a median two-fold increase in the amount of blood SMN protein levels after 4 weeks, which was sustained for at least 24 months.17
Combination therapy. Among the more eagerly awaited results are those from studies of combination therapies, including those that combine approved SMN up-regulators with new non–SMN-targeted therapeutics.
“We’re seeing that while these three approved therapies have dramatic results, especially for infants who are treated presymptomatically, there are still unmet medical needs in those patients, particularly for older teens and adults whose disease may have progressed before they were able to start therapy,” said Jackie Glascock, PhD, vice president of research for Cure SMA.
Of particular interest are studies of myostatin inhibitors, therapeutics that block the production of the protein myostatin. Myostatin acts on muscle cells to reduce muscle growth. Animal studies suggest that inhibiting myostatin increases muscle mass, which could be important in patients with muscle loss due to SMA.
Three experimental myostatin inhibitors are currently in clinical trials. MANATEE is a global phase 2-3 trial that aims to evaluate the safety and efficacy of the antimyostatin antibody GYM329 (RO7204239) in combination with risdiplam. SAPPHIRE is a phase 3 trial of apitegromab (SRK-015) in combination with nusinersen or risdiplam. RESILIANT is a phase 3 trial of tadefgrobep alfa in combination with other treatments.
A trial is underway to study the efficacy and safety of nusinersen in patients with persistent symptoms of SMA after treatment with the gene therapy. The phase 4 study, RESPOND, is enrolling children aged 2-36 months.
What’s needed next
Despite the advances in treatment and patient care, Dr. Brandsema, Dr. Schroth, and Dr. Glascock note that there remain unmet needs in the SMA community in a variety of areas.
Increased focus on adults with SMA. Before nusinersen, treatment of SMA mainly involved treating its symptoms. Many patients stopped seeing their neurologist, relying more heavily on pulmonary care specialists and/or primary care providers to address breathing, nutrition, and mobility problems. “Now with the approval of these treatments, they’re coming back to see their neurologists and are becoming more visible in the SMA community,” Dr. Schroth said.
Despite this re-emergence, a 2020 meta-analysis of studies on adults with SMA found a paucity of data on physical and occupational therapy, respiratory management, mental health care, and palliative care.18
“There is just so much work we need to do in the area of adult clinical care of SMA.”
Treatment algorithms. While the development of the newborn screening algorithm and revised patient care guidelines are helpful resources, clinicians still face uncertainty when choosing which therapy will work best for their patients. Treatment algorithms that help clinicians figure out what therapy or combination of therapies will offer the best outcomes for individual patients are desperately needed, Dr. Brandsema said.
“Each person’s experience of this disease is so unique to the individual based partly on their genetics and partly on the factors about what got them into care and how compliant they are with everything we’re trying to do to help them,” he said. “Biomarkers would help clinicians create personalized treatment plans for each patient.”
More basic science. While scientists have a good understanding of the SMN gene, there are many unanswered questions about the function of the SMN protein and its relationship to motor neuron loss. SMN is a ubiquitously expressed protein, and its function in other cell types is largely unknown. Despite all of the research advances, there is much basic science left to be done.
“We are strongly advocating to regulatory authorities that these aren’t cures and we need to continue to invest in the basic research,” Dr. Glascock said. “These biological questions that pertain to SMN and its function and expression really drive drug development. I really think that understanding those pathways better will lead us to more druggable targets.”
Two deaths from liver failure linked to spinal muscular atrophy drug
Two children taking the gene therapy drug onasemnogene abeparvovec (Zolgensma, Novartis) for spinal muscular atrophy (SMA) have died from acute liver failure, according to a statement issued by the drug’s manufacturer.
The patients were 4 months and 28 months of age and lived in Russia and Kazakhstan. They died 5-6 weeks after infusion with Zolgensma and approximately 1-10 days after the initiation of a corticosteroid taper.
These are the first known fatal cases of acute liver failure associated with the drug, which the company notes was a known side effect included in the product label and in a boxed warning in the United States.
“Following two recent patient fatalities, and in alignment with health authorities, we will be updating the labeling to specify that fatal acute liver failure has been reported,” the statement reads.
“While this is important safety information, it is not a new safety signal,” it adds.
Rare genetic disorder
SMA is a rare genetic disorder that affects about 1 in 10,000 newborns. Patients with SMA lack a working copy of the survival motor neuron 1 (SMN1) gene, which encodes a protein called SMN that is critical for the maintenance and function of motor neurons.
Without this protein, motor neurons eventually die, causing debilitating and progressive muscle weakness that affects the ability to walk, eat, and breathe.
Zolgensma, a one-time gene replacement therapy delivered via intravenous infusion, replaces the function of the missing or nonworking SMN1 gene with a new, working copy of the SMN1 gene.
The first gene therapy treatment for SMA, it was approved by the U.S. Food and Drug Administration in 2019 for patients with SMA up to 2 years of age. It is also the most expensive drug in the world, costing about $2.1 million for a one-time treatment.
“We have notified health authorities in all markets where Zolgensma is used, including the FDA, and are communicating to relevant healthcare professionals as an additional step in markets where this action is supported by health authorities,” the manufacturer’s statement says.
Studies have suggested that the treatment’s effects persist more than 5 years after infusion.
Clinical trials currently underway by Novartis are studying the drug’s long-term efficacy and safety and its potential use in older patients.
The company is also leading the phase 3 clinical trial STEER to test intrathecal (IT) administration of the drug in patients ages 2-18 years who have type 2 SMA.
That trial began late last year after the FDA lifted a 2-year partial hold on an earlier study. The FDA halted the STRONG trial in 2019, citing concerns from animal studies that IT administration may result in dorsal root ganglia injury. The partial hold was released last fall following positive study results in nonhuman primates.
None of the current trials will be affected by the two deaths reported, according to a Novartis spokesperson.
Kelli Whitlock Burton is a staff writer/reporter for Medscape Neurology and MDedge Neurology.
References
1. Viscidi E et al. Comparative all-cause mortality among a large population of patients with spinal muscular atrophy versus matched controls. Neurol Ther. 2022 Mar;11(1):449-457. doi: 10.1007/s40120-021-00307-7.
2. Finkel RS et al. Observational study of spinal muscular atrophy type I and implications for clinical trials. Neurology. 2014 Aug 26;83(9):810-817. doi: 10.1212/WNL.0000000000000741.
3. Klotz J et al. Advances in the therapy of spinal muscular atrophy. J Pediatr. 2021 Sep;236:13-20.e1. doi: 10.1016/j.jpeds.2021.06.033.
4. Curry M et al. Awareness screening and referral patterns among pediatricians in the United States related to early clinical features of spinal muscular atrophy (SMA). BMC Pediatr. 2021 May;21(1):236. doi: 10.1186/s12887-021-02692-2.
5. SMArt Moves. https://smartmoves.curesma.org/
6. Swoboda KJ et al. Natural history of denervation in SMA: Relation to age, SMN2 copy number, and function. Ann Neurol. 2005 May;57(5):704-12. doi: 10.1002/ana.20473.
7. Glascock J et al. Treatment algorithm for infants diagnosed with spinal muscular atrophy through newborn screening. J Neuromuscul Dis. 2018;5(2):145-158. doi: 10.3233/JND-180304.
8. Glascock J et al. Revised recommendations for the treatment of infants diagnosed with spinal muscular atrophy via newborn screening who have 4 copies of SMN2. J Neuromuscul Dis. 2020;7(2):97-100. doi: 10.3233/JND-190468.
9. Talbot K, Tizzano EF. The clinical landscape for SMA in a new therapeutic era. Gene Ther. 2017 Sep;24(9):529-533. doi: 10.1038/gt.2017.52.
10. Mercuri E et al. Diagnosis and management of spinal muscular atrophy: Part 1: Recommendations for diagnosis, rehabilitation, orthopedic and nutritional care. Neuromuscul Disord. 2018 Feb;28(2):103-115. doi: 10.1016/j.nmd.2017.11.005.
11. Finkel RS et al. Diagnosis and management of spinal muscular atrophy: Part 2: Pulmonary and acute care; medications, supplements and immunizations; other organ systems; and ethics. Neuromuscul Disord. 2018 Mar;28(3):197-207. doi: 10.1016/j.nmd.2017.11.004.
12. Pascual SI et al. Ongoing phase 2/3 DEVOTE (232SM203) randomized, controlled study to explore high-dose nusinersen in SMA: Part A interim results and Part B enrollment update. Presented at MDA Clinical and Scientific Conference 2021, Mar 15-18.
13. Finkel RS et al. Scientific rationale for a higher dose of nusinersen. Presented at 2021 Cure SMA Annual Meeting, Jun 9-11. Abstract P46.
14. Mendell JR et al. Five-year extension results of the phase 1 START trial of onasemnogene abeparvovec in spinal muscular atrophy. JAMA Neurol. 2021 Jul;78(7):834-841. doi: 10.1001/jamaneurol.2021.1272.
15. Darras BT et al. Risdiplam-treated infants with type 1 spinal muscular atrophy versus historical controls. N Engl J Med. 2021 Jul 29;385(5):427-435. doi: 10.1056/NEJMoa2102047.
16. Mercuri E et al. Safety and efficacy of once-daily risdiplam in type 2 and non-ambulant type 3 spinal muscular atrophy (SUNFISH part 2): A phase 3, double-blind, randomised, placebo-controlled trial. Lancet Neurol. 2022 Jan;21(1):42-52. doi: 10.1016/S1474-4422(21)00367-7. Erratum in: Lancet Neurol. 2022 Feb;21(2):e2. doi: 10.1016/S1474-4422(22)00006-0. Correction in: Lancet Neurol. 2022 Mar;21(3):e3. doi: 10.1016/S1474-4422(22)00038-2.
17. Genentech announces 2-year risdiplam data from SUNFISH and new data from JEWELFISH in infants, children and adults with SMA. https://www.curesma.org/genentech-risdiplam-data-conference-2020/
18. Wan HWY et al. Health, wellbeing and lived experiences of adults with SMA: a scoping systematic review. Orphanet J Rare Dis. 2020;15(1):70. doi: 10.1186/s13023-020-1339-3.
In 2016, the U.S. Food and Drug Administration approved nusinersen, the first treatment for spinal muscular atrophy (SMA). Until then, SMA had a mortality rate nearly double that of the general population.1 Two-thirds of patients were symptomatic within 6 months of birth and, in the absence of mechanical ventilation and other support, had a nearly 100% mortality rate by age 2.2
Five years later, there are three approved treatments for SMA, all of which have been shown to slow or even halt disease progression in many patients. Neurologists, whose SMA patient population once consisted almost entirely of children, are now treating more adults with the disease. Indeed, more than half of all people alive with SMA in the United States today are adults, according to Cure SMA.
“Managing SMA used to be clinic follow-ups where we were doing our best supportive care and watching people fall apart before our eyes,” said John Brandsema, MD, a physician and neuromuscular section head at the Children’s Hospital of Philadelphia. “Today, what we see in the vast majority of people is that they are either the same as they were before – which is completely against the natural history of this disease and something to be celebrated – or that people are really better with their function. It totally changes everything in the clinic.”
Among those changes are a more proactive approach to rehabilitation and an even greater emphasis on personalized medicine and multidisciplinary care. But there is also a need for updated treatment guidelines, a new classification system to measure disease severity, specific biomarkers to guide therapy choices, more data on long-term efficacy of existing therapeutics, new medications to complement those therapies, and a deeper understanding of a disease that may have treatment options but still has no cure.
Advances in early diagnosis
Patients with SMA lack a working copy of the survival motor neuron 1 (SMN1) gene, which provides instructions for producing a protein called SMN that is critical for the maintenance and function of motor neurons. Without this protein, motor neurons eventually die, causing debilitating and progressive muscle weakness that affects the ability to walk, eat, and breathe. SMA is rare, affecting about 1 in 10,000 newborns.
In approximately 96% of patients, SMA is caused by homozygous loss of the SMN1 gene. People with SMA have at least one copy of the SMN2 gene, sometimes called a “backup” gene, that also produces SMN protein. However, a single nucleotide difference between SMN2 and SMN1 causes about 90% of the protein produced by SMN2 to be truncated and less stable. Even with multiple copies of SMN2 present, as is the case with many infants with SMA, the amount of functional protein produced isn’t enough to compensate for the loss of SMN1.3
All three approved medications are SMN up-regulators and work to increase the amount of functional SMN protein. Starting these medications early, even before symptoms present, is critical to preserve motor function. Early treatment depends on early diagnosis, which became more widespread after 2018 when SMA was added to the federally Recommended Uniform Screening Panel for newborns. As of July 1, 2022, 47 states have incorporated SMA newborn screening into their state panel, ensuring that 97% of all infants born in the United States undergo SMA screening shortly after birth. Screening in the remaining states – Hawaii, Nevada, and South Carolina – and Washington, D.C. is expected by mid-2023.
SMA newborn screening is a PCR-based assay that detects homozygous SMN1 gene deletion found in about 95% of all people with SMA. The remaining 5% of cases are caused by various genetic mutations that can only be detected with gene sequencing. In these cases, and in children who don’t undergo SMA newborn screening, the disease is usually identified when symptoms are noticed by a parent, pediatrician, or primary care provider. But a study found that in 2018 only 52.7% of pediatricians correctly identified genetic testing as a requirement for a definitive diagnosis of SMA; in 2019, with a larger sample size, that number decreased to 45%.4 The lack of awareness of diagnostic requirements for SMA could contribute to delays in diagnosis, said Mary Schroth, MD, chief medical officer for Cure SMA and a coauthor of the study.
“In our world, suspicion of SMA in an infant is an emergency situation,” Dr. Schroth said. “These babies need to be referred immediately and have genetic testing so that treatment can begin as soon as possible.”
Based on the study findings, Dr. Schroth and others with Cure SMA launched a new tool in 2021 designed to help pediatricians, primary care physicians, and parents identify early signs of SMA, so that a referral to a pediatric neurologist happens quickly. Called SMArt Moves, the educational resource features videos and a checklist to help increase early detection in infants who had a negative SMA newborn screening result or did not receive SMA screening at birth.5
Who to treat, when, and with which treatment
For many patients, having multiple effective treatment options means that SMA is no longer a fatal disease in early childhood, but one that can be managed into adolescence and adulthood. The question for clinicians is, who do they treat, when, and with which treatment?
Studies have long shown that the number of copies of the backup gene that a patient has is inversely associated with disease severity.6 In 2018, a group of SMA experts published a treatment algorithm to help guide decision-making following a positive SMA newborn screening.7 The treatment guidelines were updated in 2020 based on clinical trial data for presymptomatic infants, and current recommendations include immediate treatment for infants with two to four copies of the SMN2 gene.8 For patients with only one copy of SMN2, most of whom will likely be symptomatic at birth, the guidelines recommend that treatment decisions be made jointly between the clinician and the family.7,8
Some suggest that the number of SMN2 copies a patient has should also be a factor in determining phenotype, which has started a conversation on the development of a new classification system.9 The original classification system for disease severity – Types 0-4 – was based on age of onset and degree of motor function achieved, with Type 0 developing prenatally and being the most severe and Type 4 developing in adulthood. Type 1 is the most common, affecting more than half of all people with SMA, followed by Types 2-4. In 2018, updated consensus care guidelines offered a revised classification system that better reflected disease progression in the age of therapy. The functional motor outcomes include nonsitters (historically Type I), sitters (historically Type 2/3), and walkers (historically Type 3/4).10,11 These guidelines are a start, but clinicians say more revision is needed.
“Types 1, 2, 3, 4 were based on function – getting to a certain point and then losing it, but now that we can treat this disease, people will shift categories based on therapeutic response or based on normal development that is possible now that the neurologic piece has been stabilized,” Dr. Brandsema said. “We need to completely change our thinking around all these different aspects of SMA management.”
While discussions of a new classification system for SMA are underway, another effort to update treatment recommendations is closer to completion. Led by Cure SMA, a group of about 50 physician experts in the United States and Europe who specialize in SMA are revising guidelines for diagnosis and treatment, the first time the recommendations have been updated since 2018. The updated recommendations, which should be published later this year, will focus on diagnosis and treatment considerations.
“We have three treatments that are available, and there are specific FDA indications for each of those, but it’s not totally clear just how those medications should be used or applied to different clinical situations,” said Dr. Schroth. “We’re in a rapid phase of learning right now in the SMA community, trying to understand how these treatments alter physiology and disease outcomes and how to best use the tools that we now have available to us. In parallel with clinical treatments, we have to be doing the best care we can to optimize the outcomes for those treatments.”
Research advances in 2021
Although all three drugs approved to treat SMA – nusinersen (Spinraza; Biogen), onasemnogene abeparvovec-xioi gene replacement therapy (Zolgensma; Novartis Gene Therapies), and risdiplam (Evrysdi, Genentech/Roche) – are highly effective, there are still unanswered questions and unmet needs. New research findings from 2021 focused on higher dosing, different drug-delivery methods, combination therapy, and complementary therapeutics to address SMA comorbidities.
Higher-dose nusinersen. The first drug approved to treat SMA, nusinersen is an antisense oligonucleotide approved for all ages and all SMA types. It works by altering splicing of the SMN2 gene pre-mRNA to make more complete SMN protein. Given as an intrathecal (IT) injection, four “loading doses” are administered within the first 2 months of treatment, followed by a maintenance dose every 4 months for the duration of the individual’s life.
Reports from patients of waning effects of nusinersen just prior to follow-up treatment have led some clinicians to ask if a higher dose may be needed. A study underway seeks to address that issue.
DEVOTE is a phase 2/3 trial to study the safety and efficacy of high-dose nusinersen in patients with SMA. Preliminary findings reported in 2021 found no adverse events among patients treated with 28 mg of nusinersen for 161-257 days.12 Another analysis from this trial found that higher doses are associated with greater decrease of plasma phosphorylated neurofilament heavy chain (pNF-H) levels in patients with SMA and may lead to clinically meaningful improvement in motor function beyond that observed with the approved 12 mg dose.13 The trial is ongoing.
Another trial, ASCEND, is a phase 3B study assessing higher dose nusinersen in patients previously treated with risdiplam. Recruitment for that trial began in October 2021.
Long-term efficacy and IT administration of SMA therapy. Several studies are looking at the long-term efficacy and alternate routes of administration of onasemnogene abeparvovec and other SMA therapies.
A one-time gene replacement therapy delivered via an IV infusion replaces the function of the missing or nonworking SMN1 gene with a new, working copy of the SMN1 gene. FDA approved in 2019, it is authorized for use in patients with SMA up to 2 years of age.
The latest data from an ongoing, long-term follow-up safety study of onasemnogene abeparvovec, published in May 2021, suggest that the treatment’s effects persist more than 5 years after treatment. Researchers followed 13 infants with symptomatic SMA type 1 since the beginning of the phase 1 clinical trial of the gene transfer therapy. All patients who received the therapeutic dose maintained their baseline motor function, and two of the patients actually improved without other SMN-targeted treatment. At a median 6.2 years after they received treatment, all were alive and none needed permanent ventilation.14
After a 2-year hold by the FDA, a study of IT administration of onasemnogene abeparvovec is now enrolling patients. Citing concerns from animal studies that IT administration might result in dorsal root ganglia injury, the FDA issued a partial hold on the STRONG trial in 2019. Following positive study results in nonhuman primates, the FDA announced the trial can continue. Novartis is launching a new phase 3 STEER trial to test the drug delivered intrathecally in patients aged 2-18 years with Type 2 SMA. IT administration could allow the gene therapy to be used safely and effectively in more patients with SMA.
Efficacy of risdiplam in more patients. The first oral treatment for SMA was approved by the FDA in 2020. It’s given once per day in patients with SMA of all ages and disease types. The drug increases functional SMN protein production by the SMN2 gene.
A July 2021 publication of the results of the FIREFISH study found that infants with Type I SMA treated with risdiplam for 12 months were significantly more likely to achieve motor milestones, such as sitting without support, compared with untreated infants with Type 1 SMA.15 Risdiplam is also effective in older patients with Type 2 or 3 SMA, according to results published in December from the SUNFISH clinical trial.16 Another study, RAINBOWFISH, is studying safety and efficacy at 24 months in presymptomatic infants started on treatment at up to 6 weeks of age.
The efficacy of risdiplam in previously treated patients is the subject of JEWELFISH, an ongoing study in patients 6 months to 60 years with SMA. Preliminary data presented at the 2020 Virtual SMA Research and Clinical Care Meeting suggest treatment with risdiplam led to a median two-fold increase in the amount of blood SMN protein levels after 4 weeks, which was sustained for at least 24 months.17
Combination therapy. Among the more eagerly awaited results are those from studies of combination therapies, including those that combine approved SMN up-regulators with new non–SMN-targeted therapeutics.
“We’re seeing that while these three approved therapies have dramatic results, especially for infants who are treated presymptomatically, there are still unmet medical needs in those patients, particularly for older teens and adults whose disease may have progressed before they were able to start therapy,” said Jackie Glascock, PhD, vice president of research for Cure SMA.
Of particular interest are studies of myostatin inhibitors, therapeutics that block the production of the protein myostatin. Myostatin acts on muscle cells to reduce muscle growth. Animal studies suggest that inhibiting myostatin increases muscle mass, which could be important in patients with muscle loss due to SMA.
Three experimental myostatin inhibitors are currently in clinical trials. MANATEE is a global phase 2-3 trial that aims to evaluate the safety and efficacy of the antimyostatin antibody GYM329 (RO7204239) in combination with risdiplam. SAPPHIRE is a phase 3 trial of apitegromab (SRK-015) in combination with nusinersen or risdiplam. RESILIANT is a phase 3 trial of tadefgrobep alfa in combination with other treatments.
A trial is underway to study the efficacy and safety of nusinersen in patients with persistent symptoms of SMA after treatment with the gene therapy. The phase 4 study, RESPOND, is enrolling children aged 2-36 months.
What’s needed next
Despite the advances in treatment and patient care, Dr. Brandsema, Dr. Schroth, and Dr. Glascock note that there remain unmet needs in the SMA community in a variety of areas.
Increased focus on adults with SMA. Before nusinersen, treatment of SMA mainly involved treating its symptoms. Many patients stopped seeing their neurologist, relying more heavily on pulmonary care specialists and/or primary care providers to address breathing, nutrition, and mobility problems. “Now with the approval of these treatments, they’re coming back to see their neurologists and are becoming more visible in the SMA community,” Dr. Schroth said.
Despite this re-emergence, a 2020 meta-analysis of studies on adults with SMA found a paucity of data on physical and occupational therapy, respiratory management, mental health care, and palliative care.18
“There is just so much work we need to do in the area of adult clinical care of SMA.”
Treatment algorithms. While the development of the newborn screening algorithm and revised patient care guidelines are helpful resources, clinicians still face uncertainty when choosing which therapy will work best for their patients. Treatment algorithms that help clinicians figure out what therapy or combination of therapies will offer the best outcomes for individual patients are desperately needed, Dr. Brandsema said.
“Each person’s experience of this disease is so unique to the individual based partly on their genetics and partly on the factors about what got them into care and how compliant they are with everything we’re trying to do to help them,” he said. “Biomarkers would help clinicians create personalized treatment plans for each patient.”
More basic science. While scientists have a good understanding of the SMN gene, there are many unanswered questions about the function of the SMN protein and its relationship to motor neuron loss. SMN is a ubiquitously expressed protein, and its function in other cell types is largely unknown. Despite all of the research advances, there is much basic science left to be done.
“We are strongly advocating to regulatory authorities that these aren’t cures and we need to continue to invest in the basic research,” Dr. Glascock said. “These biological questions that pertain to SMN and its function and expression really drive drug development. I really think that understanding those pathways better will lead us to more druggable targets.”
Two deaths from liver failure linked to spinal muscular atrophy drug
Two children taking the gene therapy drug onasemnogene abeparvovec (Zolgensma, Novartis) for spinal muscular atrophy (SMA) have died from acute liver failure, according to a statement issued by the drug’s manufacturer.
The patients were 4 months and 28 months of age and lived in Russia and Kazakhstan. They died 5-6 weeks after infusion with Zolgensma and approximately 1-10 days after the initiation of a corticosteroid taper.
These are the first known fatal cases of acute liver failure associated with the drug, which the company notes was a known side effect included in the product label and in a boxed warning in the United States.
“Following two recent patient fatalities, and in alignment with health authorities, we will be updating the labeling to specify that fatal acute liver failure has been reported,” the statement reads.
“While this is important safety information, it is not a new safety signal,” it adds.
Rare genetic disorder
SMA is a rare genetic disorder that affects about 1 in 10,000 newborns. Patients with SMA lack a working copy of the survival motor neuron 1 (SMN1) gene, which encodes a protein called SMN that is critical for the maintenance and function of motor neurons.
Without this protein, motor neurons eventually die, causing debilitating and progressive muscle weakness that affects the ability to walk, eat, and breathe.
Zolgensma, a one-time gene replacement therapy delivered via intravenous infusion, replaces the function of the missing or nonworking SMN1 gene with a new, working copy of the SMN1 gene.
The first gene therapy treatment for SMA, it was approved by the U.S. Food and Drug Administration in 2019 for patients with SMA up to 2 years of age. It is also the most expensive drug in the world, costing about $2.1 million for a one-time treatment.
“We have notified health authorities in all markets where Zolgensma is used, including the FDA, and are communicating to relevant healthcare professionals as an additional step in markets where this action is supported by health authorities,” the manufacturer’s statement says.
Studies have suggested that the treatment’s effects persist more than 5 years after infusion.
Clinical trials currently underway by Novartis are studying the drug’s long-term efficacy and safety and its potential use in older patients.
The company is also leading the phase 3 clinical trial STEER to test intrathecal (IT) administration of the drug in patients ages 2-18 years who have type 2 SMA.
That trial began late last year after the FDA lifted a 2-year partial hold on an earlier study. The FDA halted the STRONG trial in 2019, citing concerns from animal studies that IT administration may result in dorsal root ganglia injury. The partial hold was released last fall following positive study results in nonhuman primates.
None of the current trials will be affected by the two deaths reported, according to a Novartis spokesperson.
Kelli Whitlock Burton is a staff writer/reporter for Medscape Neurology and MDedge Neurology.
References
1. Viscidi E et al. Comparative all-cause mortality among a large population of patients with spinal muscular atrophy versus matched controls. Neurol Ther. 2022 Mar;11(1):449-457. doi: 10.1007/s40120-021-00307-7.
2. Finkel RS et al. Observational study of spinal muscular atrophy type I and implications for clinical trials. Neurology. 2014 Aug 26;83(9):810-817. doi: 10.1212/WNL.0000000000000741.
3. Klotz J et al. Advances in the therapy of spinal muscular atrophy. J Pediatr. 2021 Sep;236:13-20.e1. doi: 10.1016/j.jpeds.2021.06.033.
4. Curry M et al. Awareness screening and referral patterns among pediatricians in the United States related to early clinical features of spinal muscular atrophy (SMA). BMC Pediatr. 2021 May;21(1):236. doi: 10.1186/s12887-021-02692-2.
5. SMArt Moves. https://smartmoves.curesma.org/
6. Swoboda KJ et al. Natural history of denervation in SMA: Relation to age, SMN2 copy number, and function. Ann Neurol. 2005 May;57(5):704-12. doi: 10.1002/ana.20473.
7. Glascock J et al. Treatment algorithm for infants diagnosed with spinal muscular atrophy through newborn screening. J Neuromuscul Dis. 2018;5(2):145-158. doi: 10.3233/JND-180304.
8. Glascock J et al. Revised recommendations for the treatment of infants diagnosed with spinal muscular atrophy via newborn screening who have 4 copies of SMN2. J Neuromuscul Dis. 2020;7(2):97-100. doi: 10.3233/JND-190468.
9. Talbot K, Tizzano EF. The clinical landscape for SMA in a new therapeutic era. Gene Ther. 2017 Sep;24(9):529-533. doi: 10.1038/gt.2017.52.
10. Mercuri E et al. Diagnosis and management of spinal muscular atrophy: Part 1: Recommendations for diagnosis, rehabilitation, orthopedic and nutritional care. Neuromuscul Disord. 2018 Feb;28(2):103-115. doi: 10.1016/j.nmd.2017.11.005.
11. Finkel RS et al. Diagnosis and management of spinal muscular atrophy: Part 2: Pulmonary and acute care; medications, supplements and immunizations; other organ systems; and ethics. Neuromuscul Disord. 2018 Mar;28(3):197-207. doi: 10.1016/j.nmd.2017.11.004.
12. Pascual SI et al. Ongoing phase 2/3 DEVOTE (232SM203) randomized, controlled study to explore high-dose nusinersen in SMA: Part A interim results and Part B enrollment update. Presented at MDA Clinical and Scientific Conference 2021, Mar 15-18.
13. Finkel RS et al. Scientific rationale for a higher dose of nusinersen. Presented at 2021 Cure SMA Annual Meeting, Jun 9-11. Abstract P46.
14. Mendell JR et al. Five-year extension results of the phase 1 START trial of onasemnogene abeparvovec in spinal muscular atrophy. JAMA Neurol. 2021 Jul;78(7):834-841. doi: 10.1001/jamaneurol.2021.1272.
15. Darras BT et al. Risdiplam-treated infants with type 1 spinal muscular atrophy versus historical controls. N Engl J Med. 2021 Jul 29;385(5):427-435. doi: 10.1056/NEJMoa2102047.
16. Mercuri E et al. Safety and efficacy of once-daily risdiplam in type 2 and non-ambulant type 3 spinal muscular atrophy (SUNFISH part 2): A phase 3, double-blind, randomised, placebo-controlled trial. Lancet Neurol. 2022 Jan;21(1):42-52. doi: 10.1016/S1474-4422(21)00367-7. Erratum in: Lancet Neurol. 2022 Feb;21(2):e2. doi: 10.1016/S1474-4422(22)00006-0. Correction in: Lancet Neurol. 2022 Mar;21(3):e3. doi: 10.1016/S1474-4422(22)00038-2.
17. Genentech announces 2-year risdiplam data from SUNFISH and new data from JEWELFISH in infants, children and adults with SMA. https://www.curesma.org/genentech-risdiplam-data-conference-2020/
18. Wan HWY et al. Health, wellbeing and lived experiences of adults with SMA: a scoping systematic review. Orphanet J Rare Dis. 2020;15(1):70. doi: 10.1186/s13023-020-1339-3.
In 2016, the U.S. Food and Drug Administration approved nusinersen, the first treatment for spinal muscular atrophy (SMA). Until then, SMA had a mortality rate nearly double that of the general population.1 Two-thirds of patients were symptomatic within 6 months of birth and, in the absence of mechanical ventilation and other support, had a nearly 100% mortality rate by age 2.2
Five years later, there are three approved treatments for SMA, all of which have been shown to slow or even halt disease progression in many patients. Neurologists, whose SMA patient population once consisted almost entirely of children, are now treating more adults with the disease. Indeed, more than half of all people alive with SMA in the United States today are adults, according to Cure SMA.
“Managing SMA used to be clinic follow-ups where we were doing our best supportive care and watching people fall apart before our eyes,” said John Brandsema, MD, a physician and neuromuscular section head at the Children’s Hospital of Philadelphia. “Today, what we see in the vast majority of people is that they are either the same as they were before – which is completely against the natural history of this disease and something to be celebrated – or that people are really better with their function. It totally changes everything in the clinic.”
Among those changes are a more proactive approach to rehabilitation and an even greater emphasis on personalized medicine and multidisciplinary care. But there is also a need for updated treatment guidelines, a new classification system to measure disease severity, specific biomarkers to guide therapy choices, more data on long-term efficacy of existing therapeutics, new medications to complement those therapies, and a deeper understanding of a disease that may have treatment options but still has no cure.
Advances in early diagnosis
Patients with SMA lack a working copy of the survival motor neuron 1 (SMN1) gene, which provides instructions for producing a protein called SMN that is critical for the maintenance and function of motor neurons. Without this protein, motor neurons eventually die, causing debilitating and progressive muscle weakness that affects the ability to walk, eat, and breathe. SMA is rare, affecting about 1 in 10,000 newborns.
In approximately 96% of patients, SMA is caused by homozygous loss of the SMN1 gene. People with SMA have at least one copy of the SMN2 gene, sometimes called a “backup” gene, that also produces SMN protein. However, a single nucleotide difference between SMN2 and SMN1 causes about 90% of the protein produced by SMN2 to be truncated and less stable. Even with multiple copies of SMN2 present, as is the case with many infants with SMA, the amount of functional protein produced isn’t enough to compensate for the loss of SMN1.3
All three approved medications are SMN up-regulators and work to increase the amount of functional SMN protein. Starting these medications early, even before symptoms present, is critical to preserve motor function. Early treatment depends on early diagnosis, which became more widespread after 2018 when SMA was added to the federally Recommended Uniform Screening Panel for newborns. As of July 1, 2022, 47 states have incorporated SMA newborn screening into their state panel, ensuring that 97% of all infants born in the United States undergo SMA screening shortly after birth. Screening in the remaining states – Hawaii, Nevada, and South Carolina – and Washington, D.C. is expected by mid-2023.
SMA newborn screening is a PCR-based assay that detects homozygous SMN1 gene deletion found in about 95% of all people with SMA. The remaining 5% of cases are caused by various genetic mutations that can only be detected with gene sequencing. In these cases, and in children who don’t undergo SMA newborn screening, the disease is usually identified when symptoms are noticed by a parent, pediatrician, or primary care provider. But a study found that in 2018 only 52.7% of pediatricians correctly identified genetic testing as a requirement for a definitive diagnosis of SMA; in 2019, with a larger sample size, that number decreased to 45%.4 The lack of awareness of diagnostic requirements for SMA could contribute to delays in diagnosis, said Mary Schroth, MD, chief medical officer for Cure SMA and a coauthor of the study.
“In our world, suspicion of SMA in an infant is an emergency situation,” Dr. Schroth said. “These babies need to be referred immediately and have genetic testing so that treatment can begin as soon as possible.”
Based on the study findings, Dr. Schroth and others with Cure SMA launched a new tool in 2021 designed to help pediatricians, primary care physicians, and parents identify early signs of SMA, so that a referral to a pediatric neurologist happens quickly. Called SMArt Moves, the educational resource features videos and a checklist to help increase early detection in infants who had a negative SMA newborn screening result or did not receive SMA screening at birth.5
Who to treat, when, and with which treatment
For many patients, having multiple effective treatment options means that SMA is no longer a fatal disease in early childhood, but one that can be managed into adolescence and adulthood. The question for clinicians is, who do they treat, when, and with which treatment?
Studies have long shown that the number of copies of the backup gene that a patient has is inversely associated with disease severity.6 In 2018, a group of SMA experts published a treatment algorithm to help guide decision-making following a positive SMA newborn screening.7 The treatment guidelines were updated in 2020 based on clinical trial data for presymptomatic infants, and current recommendations include immediate treatment for infants with two to four copies of the SMN2 gene.8 For patients with only one copy of SMN2, most of whom will likely be symptomatic at birth, the guidelines recommend that treatment decisions be made jointly between the clinician and the family.7,8
Some suggest that the number of SMN2 copies a patient has should also be a factor in determining phenotype, which has started a conversation on the development of a new classification system.9 The original classification system for disease severity – Types 0-4 – was based on age of onset and degree of motor function achieved, with Type 0 developing prenatally and being the most severe and Type 4 developing in adulthood. Type 1 is the most common, affecting more than half of all people with SMA, followed by Types 2-4. In 2018, updated consensus care guidelines offered a revised classification system that better reflected disease progression in the age of therapy. The functional motor outcomes include nonsitters (historically Type I), sitters (historically Type 2/3), and walkers (historically Type 3/4).10,11 These guidelines are a start, but clinicians say more revision is needed.
“Types 1, 2, 3, 4 were based on function – getting to a certain point and then losing it, but now that we can treat this disease, people will shift categories based on therapeutic response or based on normal development that is possible now that the neurologic piece has been stabilized,” Dr. Brandsema said. “We need to completely change our thinking around all these different aspects of SMA management.”
While discussions of a new classification system for SMA are underway, another effort to update treatment recommendations is closer to completion. Led by Cure SMA, a group of about 50 physician experts in the United States and Europe who specialize in SMA are revising guidelines for diagnosis and treatment, the first time the recommendations have been updated since 2018. The updated recommendations, which should be published later this year, will focus on diagnosis and treatment considerations.
“We have three treatments that are available, and there are specific FDA indications for each of those, but it’s not totally clear just how those medications should be used or applied to different clinical situations,” said Dr. Schroth. “We’re in a rapid phase of learning right now in the SMA community, trying to understand how these treatments alter physiology and disease outcomes and how to best use the tools that we now have available to us. In parallel with clinical treatments, we have to be doing the best care we can to optimize the outcomes for those treatments.”
Research advances in 2021
Although all three drugs approved to treat SMA – nusinersen (Spinraza; Biogen), onasemnogene abeparvovec-xioi gene replacement therapy (Zolgensma; Novartis Gene Therapies), and risdiplam (Evrysdi, Genentech/Roche) – are highly effective, there are still unanswered questions and unmet needs. New research findings from 2021 focused on higher dosing, different drug-delivery methods, combination therapy, and complementary therapeutics to address SMA comorbidities.
Higher-dose nusinersen. The first drug approved to treat SMA, nusinersen is an antisense oligonucleotide approved for all ages and all SMA types. It works by altering splicing of the SMN2 gene pre-mRNA to make more complete SMN protein. Given as an intrathecal (IT) injection, four “loading doses” are administered within the first 2 months of treatment, followed by a maintenance dose every 4 months for the duration of the individual’s life.
Reports from patients of waning effects of nusinersen just prior to follow-up treatment have led some clinicians to ask if a higher dose may be needed. A study underway seeks to address that issue.
DEVOTE is a phase 2/3 trial to study the safety and efficacy of high-dose nusinersen in patients with SMA. Preliminary findings reported in 2021 found no adverse events among patients treated with 28 mg of nusinersen for 161-257 days.12 Another analysis from this trial found that higher doses are associated with greater decrease of plasma phosphorylated neurofilament heavy chain (pNF-H) levels in patients with SMA and may lead to clinically meaningful improvement in motor function beyond that observed with the approved 12 mg dose.13 The trial is ongoing.
Another trial, ASCEND, is a phase 3B study assessing higher dose nusinersen in patients previously treated with risdiplam. Recruitment for that trial began in October 2021.
Long-term efficacy and IT administration of SMA therapy. Several studies are looking at the long-term efficacy and alternate routes of administration of onasemnogene abeparvovec and other SMA therapies.
A one-time gene replacement therapy delivered via an IV infusion replaces the function of the missing or nonworking SMN1 gene with a new, working copy of the SMN1 gene. FDA approved in 2019, it is authorized for use in patients with SMA up to 2 years of age.
The latest data from an ongoing, long-term follow-up safety study of onasemnogene abeparvovec, published in May 2021, suggest that the treatment’s effects persist more than 5 years after treatment. Researchers followed 13 infants with symptomatic SMA type 1 since the beginning of the phase 1 clinical trial of the gene transfer therapy. All patients who received the therapeutic dose maintained their baseline motor function, and two of the patients actually improved without other SMN-targeted treatment. At a median 6.2 years after they received treatment, all were alive and none needed permanent ventilation.14
After a 2-year hold by the FDA, a study of IT administration of onasemnogene abeparvovec is now enrolling patients. Citing concerns from animal studies that IT administration might result in dorsal root ganglia injury, the FDA issued a partial hold on the STRONG trial in 2019. Following positive study results in nonhuman primates, the FDA announced the trial can continue. Novartis is launching a new phase 3 STEER trial to test the drug delivered intrathecally in patients aged 2-18 years with Type 2 SMA. IT administration could allow the gene therapy to be used safely and effectively in more patients with SMA.
Efficacy of risdiplam in more patients. The first oral treatment for SMA was approved by the FDA in 2020. It’s given once per day in patients with SMA of all ages and disease types. The drug increases functional SMN protein production by the SMN2 gene.
A July 2021 publication of the results of the FIREFISH study found that infants with Type I SMA treated with risdiplam for 12 months were significantly more likely to achieve motor milestones, such as sitting without support, compared with untreated infants with Type 1 SMA.15 Risdiplam is also effective in older patients with Type 2 or 3 SMA, according to results published in December from the SUNFISH clinical trial.16 Another study, RAINBOWFISH, is studying safety and efficacy at 24 months in presymptomatic infants started on treatment at up to 6 weeks of age.
The efficacy of risdiplam in previously treated patients is the subject of JEWELFISH, an ongoing study in patients 6 months to 60 years with SMA. Preliminary data presented at the 2020 Virtual SMA Research and Clinical Care Meeting suggest treatment with risdiplam led to a median two-fold increase in the amount of blood SMN protein levels after 4 weeks, which was sustained for at least 24 months.17
Combination therapy. Among the more eagerly awaited results are those from studies of combination therapies, including those that combine approved SMN up-regulators with new non–SMN-targeted therapeutics.
“We’re seeing that while these three approved therapies have dramatic results, especially for infants who are treated presymptomatically, there are still unmet medical needs in those patients, particularly for older teens and adults whose disease may have progressed before they were able to start therapy,” said Jackie Glascock, PhD, vice president of research for Cure SMA.
Of particular interest are studies of myostatin inhibitors, therapeutics that block the production of the protein myostatin. Myostatin acts on muscle cells to reduce muscle growth. Animal studies suggest that inhibiting myostatin increases muscle mass, which could be important in patients with muscle loss due to SMA.
Three experimental myostatin inhibitors are currently in clinical trials. MANATEE is a global phase 2-3 trial that aims to evaluate the safety and efficacy of the antimyostatin antibody GYM329 (RO7204239) in combination with risdiplam. SAPPHIRE is a phase 3 trial of apitegromab (SRK-015) in combination with nusinersen or risdiplam. RESILIANT is a phase 3 trial of tadefgrobep alfa in combination with other treatments.
A trial is underway to study the efficacy and safety of nusinersen in patients with persistent symptoms of SMA after treatment with the gene therapy. The phase 4 study, RESPOND, is enrolling children aged 2-36 months.
What’s needed next
Despite the advances in treatment and patient care, Dr. Brandsema, Dr. Schroth, and Dr. Glascock note that there remain unmet needs in the SMA community in a variety of areas.
Increased focus on adults with SMA. Before nusinersen, treatment of SMA mainly involved treating its symptoms. Many patients stopped seeing their neurologist, relying more heavily on pulmonary care specialists and/or primary care providers to address breathing, nutrition, and mobility problems. “Now with the approval of these treatments, they’re coming back to see their neurologists and are becoming more visible in the SMA community,” Dr. Schroth said.
Despite this re-emergence, a 2020 meta-analysis of studies on adults with SMA found a paucity of data on physical and occupational therapy, respiratory management, mental health care, and palliative care.18
“There is just so much work we need to do in the area of adult clinical care of SMA.”
Treatment algorithms. While the development of the newborn screening algorithm and revised patient care guidelines are helpful resources, clinicians still face uncertainty when choosing which therapy will work best for their patients. Treatment algorithms that help clinicians figure out what therapy or combination of therapies will offer the best outcomes for individual patients are desperately needed, Dr. Brandsema said.
“Each person’s experience of this disease is so unique to the individual based partly on their genetics and partly on the factors about what got them into care and how compliant they are with everything we’re trying to do to help them,” he said. “Biomarkers would help clinicians create personalized treatment plans for each patient.”
More basic science. While scientists have a good understanding of the SMN gene, there are many unanswered questions about the function of the SMN protein and its relationship to motor neuron loss. SMN is a ubiquitously expressed protein, and its function in other cell types is largely unknown. Despite all of the research advances, there is much basic science left to be done.
“We are strongly advocating to regulatory authorities that these aren’t cures and we need to continue to invest in the basic research,” Dr. Glascock said. “These biological questions that pertain to SMN and its function and expression really drive drug development. I really think that understanding those pathways better will lead us to more druggable targets.”
Two deaths from liver failure linked to spinal muscular atrophy drug
Two children taking the gene therapy drug onasemnogene abeparvovec (Zolgensma, Novartis) for spinal muscular atrophy (SMA) have died from acute liver failure, according to a statement issued by the drug’s manufacturer.
The patients were 4 months and 28 months of age and lived in Russia and Kazakhstan. They died 5-6 weeks after infusion with Zolgensma and approximately 1-10 days after the initiation of a corticosteroid taper.
These are the first known fatal cases of acute liver failure associated with the drug, which the company notes was a known side effect included in the product label and in a boxed warning in the United States.
“Following two recent patient fatalities, and in alignment with health authorities, we will be updating the labeling to specify that fatal acute liver failure has been reported,” the statement reads.
“While this is important safety information, it is not a new safety signal,” it adds.
Rare genetic disorder
SMA is a rare genetic disorder that affects about 1 in 10,000 newborns. Patients with SMA lack a working copy of the survival motor neuron 1 (SMN1) gene, which encodes a protein called SMN that is critical for the maintenance and function of motor neurons.
Without this protein, motor neurons eventually die, causing debilitating and progressive muscle weakness that affects the ability to walk, eat, and breathe.
Zolgensma, a one-time gene replacement therapy delivered via intravenous infusion, replaces the function of the missing or nonworking SMN1 gene with a new, working copy of the SMN1 gene.
The first gene therapy treatment for SMA, it was approved by the U.S. Food and Drug Administration in 2019 for patients with SMA up to 2 years of age. It is also the most expensive drug in the world, costing about $2.1 million for a one-time treatment.
“We have notified health authorities in all markets where Zolgensma is used, including the FDA, and are communicating to relevant healthcare professionals as an additional step in markets where this action is supported by health authorities,” the manufacturer’s statement says.
Studies have suggested that the treatment’s effects persist more than 5 years after infusion.
Clinical trials currently underway by Novartis are studying the drug’s long-term efficacy and safety and its potential use in older patients.
The company is also leading the phase 3 clinical trial STEER to test intrathecal (IT) administration of the drug in patients ages 2-18 years who have type 2 SMA.
That trial began late last year after the FDA lifted a 2-year partial hold on an earlier study. The FDA halted the STRONG trial in 2019, citing concerns from animal studies that IT administration may result in dorsal root ganglia injury. The partial hold was released last fall following positive study results in nonhuman primates.
None of the current trials will be affected by the two deaths reported, according to a Novartis spokesperson.
Kelli Whitlock Burton is a staff writer/reporter for Medscape Neurology and MDedge Neurology.
References
1. Viscidi E et al. Comparative all-cause mortality among a large population of patients with spinal muscular atrophy versus matched controls. Neurol Ther. 2022 Mar;11(1):449-457. doi: 10.1007/s40120-021-00307-7.
2. Finkel RS et al. Observational study of spinal muscular atrophy type I and implications for clinical trials. Neurology. 2014 Aug 26;83(9):810-817. doi: 10.1212/WNL.0000000000000741.
3. Klotz J et al. Advances in the therapy of spinal muscular atrophy. J Pediatr. 2021 Sep;236:13-20.e1. doi: 10.1016/j.jpeds.2021.06.033.
4. Curry M et al. Awareness screening and referral patterns among pediatricians in the United States related to early clinical features of spinal muscular atrophy (SMA). BMC Pediatr. 2021 May;21(1):236. doi: 10.1186/s12887-021-02692-2.
5. SMArt Moves. https://smartmoves.curesma.org/
6. Swoboda KJ et al. Natural history of denervation in SMA: Relation to age, SMN2 copy number, and function. Ann Neurol. 2005 May;57(5):704-12. doi: 10.1002/ana.20473.
7. Glascock J et al. Treatment algorithm for infants diagnosed with spinal muscular atrophy through newborn screening. J Neuromuscul Dis. 2018;5(2):145-158. doi: 10.3233/JND-180304.
8. Glascock J et al. Revised recommendations for the treatment of infants diagnosed with spinal muscular atrophy via newborn screening who have 4 copies of SMN2. J Neuromuscul Dis. 2020;7(2):97-100. doi: 10.3233/JND-190468.
9. Talbot K, Tizzano EF. The clinical landscape for SMA in a new therapeutic era. Gene Ther. 2017 Sep;24(9):529-533. doi: 10.1038/gt.2017.52.
10. Mercuri E et al. Diagnosis and management of spinal muscular atrophy: Part 1: Recommendations for diagnosis, rehabilitation, orthopedic and nutritional care. Neuromuscul Disord. 2018 Feb;28(2):103-115. doi: 10.1016/j.nmd.2017.11.005.
11. Finkel RS et al. Diagnosis and management of spinal muscular atrophy: Part 2: Pulmonary and acute care; medications, supplements and immunizations; other organ systems; and ethics. Neuromuscul Disord. 2018 Mar;28(3):197-207. doi: 10.1016/j.nmd.2017.11.004.
12. Pascual SI et al. Ongoing phase 2/3 DEVOTE (232SM203) randomized, controlled study to explore high-dose nusinersen in SMA: Part A interim results and Part B enrollment update. Presented at MDA Clinical and Scientific Conference 2021, Mar 15-18.
13. Finkel RS et al. Scientific rationale for a higher dose of nusinersen. Presented at 2021 Cure SMA Annual Meeting, Jun 9-11. Abstract P46.
14. Mendell JR et al. Five-year extension results of the phase 1 START trial of onasemnogene abeparvovec in spinal muscular atrophy. JAMA Neurol. 2021 Jul;78(7):834-841. doi: 10.1001/jamaneurol.2021.1272.
15. Darras BT et al. Risdiplam-treated infants with type 1 spinal muscular atrophy versus historical controls. N Engl J Med. 2021 Jul 29;385(5):427-435. doi: 10.1056/NEJMoa2102047.
16. Mercuri E et al. Safety and efficacy of once-daily risdiplam in type 2 and non-ambulant type 3 spinal muscular atrophy (SUNFISH part 2): A phase 3, double-blind, randomised, placebo-controlled trial. Lancet Neurol. 2022 Jan;21(1):42-52. doi: 10.1016/S1474-4422(21)00367-7. Erratum in: Lancet Neurol. 2022 Feb;21(2):e2. doi: 10.1016/S1474-4422(22)00006-0. Correction in: Lancet Neurol. 2022 Mar;21(3):e3. doi: 10.1016/S1474-4422(22)00038-2.
17. Genentech announces 2-year risdiplam data from SUNFISH and new data from JEWELFISH in infants, children and adults with SMA. https://www.curesma.org/genentech-risdiplam-data-conference-2020/
18. Wan HWY et al. Health, wellbeing and lived experiences of adults with SMA: a scoping systematic review. Orphanet J Rare Dis. 2020;15(1):70. doi: 10.1186/s13023-020-1339-3.
Rare disease patient advocacy groups empowered by data
With the goal of advancing treatment of rare neurological diseases – or rare diseases of any type – the National Organization for Rare Disorders (NORD) has launched innovative new research initiatives in recent years to help patient advocacy organizations develop a precious asset: data to support better understanding of diseases and research that might lead to life-altering diagnostics or treatments.
“Most rare diseases still don’t have approved therapies, and the problem is often a lack of the basic information needed to advance research,” explained Aliza Fink, DSc, the director of research programs at NORD. “Our goal is to help patient organizations play a key role in the collection, analysis, and sharing of data to support better understanding of how a disease presents, its natural history, the types and severity of symptoms, and other unanswered questions.”
Over the past 2 decades, the Internet, social media, and other communications resources have provided patient organizations with unprecedented reach. As a result, these organizations are in a unique position to connect patients and caregivers around the world – those dealing with even the rarest of rare diseases – and become a repository of information on the disease and the patient experience.
Since the late 1980s, NORD has had a research grants program, and the grants this program provides to academic researchers have led to numerous significant discoveries and publications, as well as to two products that ultimately were approved by FDA. More recently, however, NORD’s research programs have been expanded to include an initiative known as IAMRARE, in which patient advocacy organizations are trained to conduct observational research and host natural history studies and registries on a platform developed by NORD.
“We work with the patient groups to determine what types of data would be most important to drive research, help develop the methodology for data collection, and advise them on protocols for supporting the quality and integrity of the data,” Dr. Fink said. “By systematically collecting data from the patients and families they serve, these groups are in a position to contribute enormously to understanding the disease and advancing research.”
NORD also helps with the practical aspects of conducting research of sufficient quality to be publishable, such as providing groups with guidelines and best practices for developing medical advisory committees, creating templates and materials to streamline their project’s submission to institutional review boards, ensuring data security and privacy in accordance with Health Insurance Portability and Accountability Act criteria, and developing other expected standards for data collection and analysis.
Unlike even academic medical centers with an interest in a given rare disease, leading patient advocacy groups for these specific disorders have unmatched access to affected patients and families. This includes patients being managed in diverse settings or those not yet receiving care at all. By harnessing this patient population to record the signs, symptoms, disease course, and other information, the patient advocacy groups can contribute greatly to the pool of available data and ultimately what is known about the disease.
Data empowers research
While NORD helps groups through the IAMRARE program to become research-ready and guides them in developing research protocols and goals, the data are ultimately owned by the patient advocacy groups themselves. This helps to ensure that the patient voice is heard. By controlling data collection and dissemination, the advocacy groups can take a leading role in defining the goals of research, including what outcome measures are important to them and what they agree are the most promising avenues for research to achieve those goals.
“By collecting the data to understand the disease, it sets the stage for the next steps in research,” explained Debbie Drell, the director of membership for NORD. She noted that IAMRARE has grown steadily since its inception in 2014 and that there are now close to 40 advocacy groups participating.
The value of this initiative is not difficult to grasp. Even though direct participation in research was not generally part of the agenda for some advocacy groups when IAMRARE was conceived, Ms. Drell said that this initiative is a compelling perk of becoming involved with NORD. Groups that elect to become research-ready in order to participate in IAMRARE fall into a category of membership that requires specific organizational structures – such as a medical advisory board – and NORD provides templates and guidance to help them meet these qualifications to successfully become research-ready.
Collaboration leads to progress
NORD was founded by an ad hoc committee of patient organizations that played a key role in enactment of the Orphan Drug Act nearly 40 years ago. Shortly after the Orphan Drug Act was passed by Congress and signed into law by President Ronald Reagan in 1983, the ad hoc committee formally united to create NORD to continue the momentum of this initial collaboration and support the rare disease community. According to Mary Dunkle, a senior advisor at NORD, passage of the Orphan Drug Act, which is widely considered a major driver of progress in development of treatments for rare diseases, made the advantages of their cooperation clear.
“The groups had so many issues in common across the spectrum of diseases that they decided to continue their collaboration,” she explained. ”They realized that, while each disease is rare, the challenges they present to patients, families, clinicians, and researchers have many similarities.”
The definition of rare disease, according to the National Institutes of Health, is a disorder that affects fewer than 200,000 people in the United States. More than 7,000 such disorders have been identified. Approximately one-third of rare diseases are neurological. Whether neurological or affecting different or multiple organ systems, most – perhaps 75%-80% – involve a genetic component, according to Ms. Dunkle.
Research reaps rewards
Altogether, today there are more than 1,000 patient organizations that provide various types of support and services for patients and caregivers affected by rare diseases. Approximately one-third of these organizations are members of NORD. For organizations that don’t yet meet the membership criteria or for other reasons have not yet formally joined NORD, there are still many opportunities to get involved and to learn best practices to strengthen their governance, infrastructure, and capacity to support their members.
Of these, the IAMRARE program is one of the best examples of ways to get involved. Beyond the many other ways these groups help patients and families cope with challenging diseases, participation in research takes rare disorder advocacy to a different level. Objective data can attract the attention of those with the resources to further study the disease, while also giving advocacy groups a seat at the table when researchers or industry become interested.
“Why create a registry? It removes competition between academic centers or industry working on their own. It creates one central source for data-sharing, and the advantage is that advocacy groups have a trusted relationship with the patient community because they are not-for-profit, community-run, and patient-driven,” Ms. Drell explained.
The registry platforms developed for IAMRARE are customizable. With guidance from NORD, the advocacy groups themselves decide what data to collect and what questions they wish to answer, according to Dr. Fink. Once the registries are created, patients and caregivers participate by responding to survey questions on disease onset, progression over time, types and severity of symptoms, and other topics. The data can be de-identified for research purposes. The advocacy groups decide how and when to share the data, including whether to publish findings.
“Some of the groups have been very successful in getting the data published and leveraging their results to drive research forward, but there is variability in the extent of dissemination across the groups,” said Dr. Fink. She noted that many of the registries that NORD has helped set up involve groups whose officers have had little or no prior research experience.
“We have advocacy groups that have had biomedical researchers on staff and other groups that are coming to research completely new,” Dr. Fink said. In trying to get them up to speed on quality data collection, “We try to meet them where they are,” she added, indicating that leading groups to a research-ready status is not just about logistics but can sometimes involve an organizational reorientation.
The examples of peer-reviewed publications that can be directly traced to IAMRARE registries are growing. One example is a registry on Prader-Willi syndrome, which is a complex neurodevelopmental disorder characterized by failure to thrive and by multiple endocrine abnormalities. The registry was developed in NORD’s IAMRARE program by the Foundation for Prader-Willi Research, a nonprofit created in 2003 by parents of children with this disorder.
By 2019, when the first data from the Global Prader-Willi Syndrome Registry were published, they drew from 23,550 surveys completed for 1,696 separate cases of the disorder in 37 countries. The surveys provided some preliminary findings on demographics and on the genetic subtypes most commonly encountered, as well as simply proof that the registry was viable. From its inception in 2015, a significant proportion of the Prader-Willi population in the United States had been enrolled, according to the study authors. With time, the serial accumulation of more data on more cases will be invaluable for documenting disease characteristics. It will be a constantly maturing resource even after fundamental questions on disease impact and prognosis are addressed.
Data accumulation
Only about 10% of rare diseases currently have approved treatments, but there is widespread belief in the rare community that collecting and analyzing the data that can promote understanding of the biology of the disease and identify therapeutic targets could accelerate the development of treatments for diseases that currently have none.
Therefore, data accumulation has become central to the mission of NORD. In addition to IAMRARE, the organization has embarked on several other important initiatives in data accumulation for rare diseases. One is the Rare Disease Cures Accelerator – Data and Analytics Platform (RDCA-DAP), an initiative in which NORD is partnering with the Critical Path Institute. The goal of this program is to gather disparate pools of existing data in a standardized format to increase their power.
“With funding from the Food and Drug Administration, we have helped to support this platform, which is designed specifically to provide a centralized structure for combining and sharing of data,” according to Dr. Fink. In RDCA-DAP, patient-level data is being assembled from a variety of resources, including academic centers, industry, registries, observational studies, and clinical trials. The program was launched in September 2021. In some cases, gaining access to data includes resolving privacy issues or addressing the proprietary concerns of those who currently have the data, but the value of the combined data is a compelling argument for participation.
“What we are trying to do is pull together the data from their current silos into one platform, and then make it generally available,” said Dr. Fink. As with IAMRARE, RDCA-DAP offers enormous potential.
“The primary challenge for those studying rare diseases is the small numbers of patients. Randomized clinical trials for some of these diseases are simply not feasible because there are not enough subjects to power two study arms,” said Dr. Fink in explaining why NORD has turned to novel strategies for data generation. One strategy for maximizing the potential value of data from these small populations of patients is data-sharing. For RDCA-DAP, data access will be open to all stakeholders after scientific review and approval.
“Anyone can get an account and request data from the platform,” said Dr. Fink, who expects this to spur more and novel types of research in rare disorders.
Another example of recent NORD initiatives to advance research and understanding of rare diseases is a study of metachromatic leukodystrophy (MLD) that is now enrolling patients, which also represents a partnership with the FDA. For this study, which is known as the HOME study, NORD hosts a platform where patients and caregivers enter data to capture the natural history of this disease. All MLD patients, even if they are already participating in a clinical trial or another registry, are invited. As with the IAMRARE registries, surveys capture patient or caregiver responses entered from a computer or smart device.
“We have always believed that the fact that so many rare diseases don’t have treatments or are not even being studied by researchers doesn’t reflect a lack of interest among academic or industry researchers. Rather, it reflects a lack of data to support research and to provide a fundamental understanding of the disease,” Dr. Fink said. “If NORD’s expanded research programs can draw the patient community together to provide that crucially needed data, we will have provided an important and essential service to patients, patient organizations, and researchers alike.”
Theodore Bosworth is a freelance journalist and editor specializing in medicine and health.
With the goal of advancing treatment of rare neurological diseases – or rare diseases of any type – the National Organization for Rare Disorders (NORD) has launched innovative new research initiatives in recent years to help patient advocacy organizations develop a precious asset: data to support better understanding of diseases and research that might lead to life-altering diagnostics or treatments.
“Most rare diseases still don’t have approved therapies, and the problem is often a lack of the basic information needed to advance research,” explained Aliza Fink, DSc, the director of research programs at NORD. “Our goal is to help patient organizations play a key role in the collection, analysis, and sharing of data to support better understanding of how a disease presents, its natural history, the types and severity of symptoms, and other unanswered questions.”
Over the past 2 decades, the Internet, social media, and other communications resources have provided patient organizations with unprecedented reach. As a result, these organizations are in a unique position to connect patients and caregivers around the world – those dealing with even the rarest of rare diseases – and become a repository of information on the disease and the patient experience.
Since the late 1980s, NORD has had a research grants program, and the grants this program provides to academic researchers have led to numerous significant discoveries and publications, as well as to two products that ultimately were approved by FDA. More recently, however, NORD’s research programs have been expanded to include an initiative known as IAMRARE, in which patient advocacy organizations are trained to conduct observational research and host natural history studies and registries on a platform developed by NORD.
“We work with the patient groups to determine what types of data would be most important to drive research, help develop the methodology for data collection, and advise them on protocols for supporting the quality and integrity of the data,” Dr. Fink said. “By systematically collecting data from the patients and families they serve, these groups are in a position to contribute enormously to understanding the disease and advancing research.”
NORD also helps with the practical aspects of conducting research of sufficient quality to be publishable, such as providing groups with guidelines and best practices for developing medical advisory committees, creating templates and materials to streamline their project’s submission to institutional review boards, ensuring data security and privacy in accordance with Health Insurance Portability and Accountability Act criteria, and developing other expected standards for data collection and analysis.
Unlike even academic medical centers with an interest in a given rare disease, leading patient advocacy groups for these specific disorders have unmatched access to affected patients and families. This includes patients being managed in diverse settings or those not yet receiving care at all. By harnessing this patient population to record the signs, symptoms, disease course, and other information, the patient advocacy groups can contribute greatly to the pool of available data and ultimately what is known about the disease.
Data empowers research
While NORD helps groups through the IAMRARE program to become research-ready and guides them in developing research protocols and goals, the data are ultimately owned by the patient advocacy groups themselves. This helps to ensure that the patient voice is heard. By controlling data collection and dissemination, the advocacy groups can take a leading role in defining the goals of research, including what outcome measures are important to them and what they agree are the most promising avenues for research to achieve those goals.
“By collecting the data to understand the disease, it sets the stage for the next steps in research,” explained Debbie Drell, the director of membership for NORD. She noted that IAMRARE has grown steadily since its inception in 2014 and that there are now close to 40 advocacy groups participating.
The value of this initiative is not difficult to grasp. Even though direct participation in research was not generally part of the agenda for some advocacy groups when IAMRARE was conceived, Ms. Drell said that this initiative is a compelling perk of becoming involved with NORD. Groups that elect to become research-ready in order to participate in IAMRARE fall into a category of membership that requires specific organizational structures – such as a medical advisory board – and NORD provides templates and guidance to help them meet these qualifications to successfully become research-ready.
Collaboration leads to progress
NORD was founded by an ad hoc committee of patient organizations that played a key role in enactment of the Orphan Drug Act nearly 40 years ago. Shortly after the Orphan Drug Act was passed by Congress and signed into law by President Ronald Reagan in 1983, the ad hoc committee formally united to create NORD to continue the momentum of this initial collaboration and support the rare disease community. According to Mary Dunkle, a senior advisor at NORD, passage of the Orphan Drug Act, which is widely considered a major driver of progress in development of treatments for rare diseases, made the advantages of their cooperation clear.
“The groups had so many issues in common across the spectrum of diseases that they decided to continue their collaboration,” she explained. ”They realized that, while each disease is rare, the challenges they present to patients, families, clinicians, and researchers have many similarities.”
The definition of rare disease, according to the National Institutes of Health, is a disorder that affects fewer than 200,000 people in the United States. More than 7,000 such disorders have been identified. Approximately one-third of rare diseases are neurological. Whether neurological or affecting different or multiple organ systems, most – perhaps 75%-80% – involve a genetic component, according to Ms. Dunkle.
Research reaps rewards
Altogether, today there are more than 1,000 patient organizations that provide various types of support and services for patients and caregivers affected by rare diseases. Approximately one-third of these organizations are members of NORD. For organizations that don’t yet meet the membership criteria or for other reasons have not yet formally joined NORD, there are still many opportunities to get involved and to learn best practices to strengthen their governance, infrastructure, and capacity to support their members.
Of these, the IAMRARE program is one of the best examples of ways to get involved. Beyond the many other ways these groups help patients and families cope with challenging diseases, participation in research takes rare disorder advocacy to a different level. Objective data can attract the attention of those with the resources to further study the disease, while also giving advocacy groups a seat at the table when researchers or industry become interested.
“Why create a registry? It removes competition between academic centers or industry working on their own. It creates one central source for data-sharing, and the advantage is that advocacy groups have a trusted relationship with the patient community because they are not-for-profit, community-run, and patient-driven,” Ms. Drell explained.
The registry platforms developed for IAMRARE are customizable. With guidance from NORD, the advocacy groups themselves decide what data to collect and what questions they wish to answer, according to Dr. Fink. Once the registries are created, patients and caregivers participate by responding to survey questions on disease onset, progression over time, types and severity of symptoms, and other topics. The data can be de-identified for research purposes. The advocacy groups decide how and when to share the data, including whether to publish findings.
“Some of the groups have been very successful in getting the data published and leveraging their results to drive research forward, but there is variability in the extent of dissemination across the groups,” said Dr. Fink. She noted that many of the registries that NORD has helped set up involve groups whose officers have had little or no prior research experience.
“We have advocacy groups that have had biomedical researchers on staff and other groups that are coming to research completely new,” Dr. Fink said. In trying to get them up to speed on quality data collection, “We try to meet them where they are,” she added, indicating that leading groups to a research-ready status is not just about logistics but can sometimes involve an organizational reorientation.
The examples of peer-reviewed publications that can be directly traced to IAMRARE registries are growing. One example is a registry on Prader-Willi syndrome, which is a complex neurodevelopmental disorder characterized by failure to thrive and by multiple endocrine abnormalities. The registry was developed in NORD’s IAMRARE program by the Foundation for Prader-Willi Research, a nonprofit created in 2003 by parents of children with this disorder.
By 2019, when the first data from the Global Prader-Willi Syndrome Registry were published, they drew from 23,550 surveys completed for 1,696 separate cases of the disorder in 37 countries. The surveys provided some preliminary findings on demographics and on the genetic subtypes most commonly encountered, as well as simply proof that the registry was viable. From its inception in 2015, a significant proportion of the Prader-Willi population in the United States had been enrolled, according to the study authors. With time, the serial accumulation of more data on more cases will be invaluable for documenting disease characteristics. It will be a constantly maturing resource even after fundamental questions on disease impact and prognosis are addressed.
Data accumulation
Only about 10% of rare diseases currently have approved treatments, but there is widespread belief in the rare community that collecting and analyzing the data that can promote understanding of the biology of the disease and identify therapeutic targets could accelerate the development of treatments for diseases that currently have none.
Therefore, data accumulation has become central to the mission of NORD. In addition to IAMRARE, the organization has embarked on several other important initiatives in data accumulation for rare diseases. One is the Rare Disease Cures Accelerator – Data and Analytics Platform (RDCA-DAP), an initiative in which NORD is partnering with the Critical Path Institute. The goal of this program is to gather disparate pools of existing data in a standardized format to increase their power.
“With funding from the Food and Drug Administration, we have helped to support this platform, which is designed specifically to provide a centralized structure for combining and sharing of data,” according to Dr. Fink. In RDCA-DAP, patient-level data is being assembled from a variety of resources, including academic centers, industry, registries, observational studies, and clinical trials. The program was launched in September 2021. In some cases, gaining access to data includes resolving privacy issues or addressing the proprietary concerns of those who currently have the data, but the value of the combined data is a compelling argument for participation.
“What we are trying to do is pull together the data from their current silos into one platform, and then make it generally available,” said Dr. Fink. As with IAMRARE, RDCA-DAP offers enormous potential.
“The primary challenge for those studying rare diseases is the small numbers of patients. Randomized clinical trials for some of these diseases are simply not feasible because there are not enough subjects to power two study arms,” said Dr. Fink in explaining why NORD has turned to novel strategies for data generation. One strategy for maximizing the potential value of data from these small populations of patients is data-sharing. For RDCA-DAP, data access will be open to all stakeholders after scientific review and approval.
“Anyone can get an account and request data from the platform,” said Dr. Fink, who expects this to spur more and novel types of research in rare disorders.
Another example of recent NORD initiatives to advance research and understanding of rare diseases is a study of metachromatic leukodystrophy (MLD) that is now enrolling patients, which also represents a partnership with the FDA. For this study, which is known as the HOME study, NORD hosts a platform where patients and caregivers enter data to capture the natural history of this disease. All MLD patients, even if they are already participating in a clinical trial or another registry, are invited. As with the IAMRARE registries, surveys capture patient or caregiver responses entered from a computer or smart device.
“We have always believed that the fact that so many rare diseases don’t have treatments or are not even being studied by researchers doesn’t reflect a lack of interest among academic or industry researchers. Rather, it reflects a lack of data to support research and to provide a fundamental understanding of the disease,” Dr. Fink said. “If NORD’s expanded research programs can draw the patient community together to provide that crucially needed data, we will have provided an important and essential service to patients, patient organizations, and researchers alike.”
Theodore Bosworth is a freelance journalist and editor specializing in medicine and health.
With the goal of advancing treatment of rare neurological diseases – or rare diseases of any type – the National Organization for Rare Disorders (NORD) has launched innovative new research initiatives in recent years to help patient advocacy organizations develop a precious asset: data to support better understanding of diseases and research that might lead to life-altering diagnostics or treatments.
“Most rare diseases still don’t have approved therapies, and the problem is often a lack of the basic information needed to advance research,” explained Aliza Fink, DSc, the director of research programs at NORD. “Our goal is to help patient organizations play a key role in the collection, analysis, and sharing of data to support better understanding of how a disease presents, its natural history, the types and severity of symptoms, and other unanswered questions.”
Over the past 2 decades, the Internet, social media, and other communications resources have provided patient organizations with unprecedented reach. As a result, these organizations are in a unique position to connect patients and caregivers around the world – those dealing with even the rarest of rare diseases – and become a repository of information on the disease and the patient experience.
Since the late 1980s, NORD has had a research grants program, and the grants this program provides to academic researchers have led to numerous significant discoveries and publications, as well as to two products that ultimately were approved by FDA. More recently, however, NORD’s research programs have been expanded to include an initiative known as IAMRARE, in which patient advocacy organizations are trained to conduct observational research and host natural history studies and registries on a platform developed by NORD.
“We work with the patient groups to determine what types of data would be most important to drive research, help develop the methodology for data collection, and advise them on protocols for supporting the quality and integrity of the data,” Dr. Fink said. “By systematically collecting data from the patients and families they serve, these groups are in a position to contribute enormously to understanding the disease and advancing research.”
NORD also helps with the practical aspects of conducting research of sufficient quality to be publishable, such as providing groups with guidelines and best practices for developing medical advisory committees, creating templates and materials to streamline their project’s submission to institutional review boards, ensuring data security and privacy in accordance with Health Insurance Portability and Accountability Act criteria, and developing other expected standards for data collection and analysis.
Unlike even academic medical centers with an interest in a given rare disease, leading patient advocacy groups for these specific disorders have unmatched access to affected patients and families. This includes patients being managed in diverse settings or those not yet receiving care at all. By harnessing this patient population to record the signs, symptoms, disease course, and other information, the patient advocacy groups can contribute greatly to the pool of available data and ultimately what is known about the disease.
Data empowers research
While NORD helps groups through the IAMRARE program to become research-ready and guides them in developing research protocols and goals, the data are ultimately owned by the patient advocacy groups themselves. This helps to ensure that the patient voice is heard. By controlling data collection and dissemination, the advocacy groups can take a leading role in defining the goals of research, including what outcome measures are important to them and what they agree are the most promising avenues for research to achieve those goals.
“By collecting the data to understand the disease, it sets the stage for the next steps in research,” explained Debbie Drell, the director of membership for NORD. She noted that IAMRARE has grown steadily since its inception in 2014 and that there are now close to 40 advocacy groups participating.
The value of this initiative is not difficult to grasp. Even though direct participation in research was not generally part of the agenda for some advocacy groups when IAMRARE was conceived, Ms. Drell said that this initiative is a compelling perk of becoming involved with NORD. Groups that elect to become research-ready in order to participate in IAMRARE fall into a category of membership that requires specific organizational structures – such as a medical advisory board – and NORD provides templates and guidance to help them meet these qualifications to successfully become research-ready.
Collaboration leads to progress
NORD was founded by an ad hoc committee of patient organizations that played a key role in enactment of the Orphan Drug Act nearly 40 years ago. Shortly after the Orphan Drug Act was passed by Congress and signed into law by President Ronald Reagan in 1983, the ad hoc committee formally united to create NORD to continue the momentum of this initial collaboration and support the rare disease community. According to Mary Dunkle, a senior advisor at NORD, passage of the Orphan Drug Act, which is widely considered a major driver of progress in development of treatments for rare diseases, made the advantages of their cooperation clear.
“The groups had so many issues in common across the spectrum of diseases that they decided to continue their collaboration,” she explained. ”They realized that, while each disease is rare, the challenges they present to patients, families, clinicians, and researchers have many similarities.”
The definition of rare disease, according to the National Institutes of Health, is a disorder that affects fewer than 200,000 people in the United States. More than 7,000 such disorders have been identified. Approximately one-third of rare diseases are neurological. Whether neurological or affecting different or multiple organ systems, most – perhaps 75%-80% – involve a genetic component, according to Ms. Dunkle.
Research reaps rewards
Altogether, today there are more than 1,000 patient organizations that provide various types of support and services for patients and caregivers affected by rare diseases. Approximately one-third of these organizations are members of NORD. For organizations that don’t yet meet the membership criteria or for other reasons have not yet formally joined NORD, there are still many opportunities to get involved and to learn best practices to strengthen their governance, infrastructure, and capacity to support their members.
Of these, the IAMRARE program is one of the best examples of ways to get involved. Beyond the many other ways these groups help patients and families cope with challenging diseases, participation in research takes rare disorder advocacy to a different level. Objective data can attract the attention of those with the resources to further study the disease, while also giving advocacy groups a seat at the table when researchers or industry become interested.
“Why create a registry? It removes competition between academic centers or industry working on their own. It creates one central source for data-sharing, and the advantage is that advocacy groups have a trusted relationship with the patient community because they are not-for-profit, community-run, and patient-driven,” Ms. Drell explained.
The registry platforms developed for IAMRARE are customizable. With guidance from NORD, the advocacy groups themselves decide what data to collect and what questions they wish to answer, according to Dr. Fink. Once the registries are created, patients and caregivers participate by responding to survey questions on disease onset, progression over time, types and severity of symptoms, and other topics. The data can be de-identified for research purposes. The advocacy groups decide how and when to share the data, including whether to publish findings.
“Some of the groups have been very successful in getting the data published and leveraging their results to drive research forward, but there is variability in the extent of dissemination across the groups,” said Dr. Fink. She noted that many of the registries that NORD has helped set up involve groups whose officers have had little or no prior research experience.
“We have advocacy groups that have had biomedical researchers on staff and other groups that are coming to research completely new,” Dr. Fink said. In trying to get them up to speed on quality data collection, “We try to meet them where they are,” she added, indicating that leading groups to a research-ready status is not just about logistics but can sometimes involve an organizational reorientation.
The examples of peer-reviewed publications that can be directly traced to IAMRARE registries are growing. One example is a registry on Prader-Willi syndrome, which is a complex neurodevelopmental disorder characterized by failure to thrive and by multiple endocrine abnormalities. The registry was developed in NORD’s IAMRARE program by the Foundation for Prader-Willi Research, a nonprofit created in 2003 by parents of children with this disorder.
By 2019, when the first data from the Global Prader-Willi Syndrome Registry were published, they drew from 23,550 surveys completed for 1,696 separate cases of the disorder in 37 countries. The surveys provided some preliminary findings on demographics and on the genetic subtypes most commonly encountered, as well as simply proof that the registry was viable. From its inception in 2015, a significant proportion of the Prader-Willi population in the United States had been enrolled, according to the study authors. With time, the serial accumulation of more data on more cases will be invaluable for documenting disease characteristics. It will be a constantly maturing resource even after fundamental questions on disease impact and prognosis are addressed.
Data accumulation
Only about 10% of rare diseases currently have approved treatments, but there is widespread belief in the rare community that collecting and analyzing the data that can promote understanding of the biology of the disease and identify therapeutic targets could accelerate the development of treatments for diseases that currently have none.
Therefore, data accumulation has become central to the mission of NORD. In addition to IAMRARE, the organization has embarked on several other important initiatives in data accumulation for rare diseases. One is the Rare Disease Cures Accelerator – Data and Analytics Platform (RDCA-DAP), an initiative in which NORD is partnering with the Critical Path Institute. The goal of this program is to gather disparate pools of existing data in a standardized format to increase their power.
“With funding from the Food and Drug Administration, we have helped to support this platform, which is designed specifically to provide a centralized structure for combining and sharing of data,” according to Dr. Fink. In RDCA-DAP, patient-level data is being assembled from a variety of resources, including academic centers, industry, registries, observational studies, and clinical trials. The program was launched in September 2021. In some cases, gaining access to data includes resolving privacy issues or addressing the proprietary concerns of those who currently have the data, but the value of the combined data is a compelling argument for participation.
“What we are trying to do is pull together the data from their current silos into one platform, and then make it generally available,” said Dr. Fink. As with IAMRARE, RDCA-DAP offers enormous potential.
“The primary challenge for those studying rare diseases is the small numbers of patients. Randomized clinical trials for some of these diseases are simply not feasible because there are not enough subjects to power two study arms,” said Dr. Fink in explaining why NORD has turned to novel strategies for data generation. One strategy for maximizing the potential value of data from these small populations of patients is data-sharing. For RDCA-DAP, data access will be open to all stakeholders after scientific review and approval.
“Anyone can get an account and request data from the platform,” said Dr. Fink, who expects this to spur more and novel types of research in rare disorders.
Another example of recent NORD initiatives to advance research and understanding of rare diseases is a study of metachromatic leukodystrophy (MLD) that is now enrolling patients, which also represents a partnership with the FDA. For this study, which is known as the HOME study, NORD hosts a platform where patients and caregivers enter data to capture the natural history of this disease. All MLD patients, even if they are already participating in a clinical trial or another registry, are invited. As with the IAMRARE registries, surveys capture patient or caregiver responses entered from a computer or smart device.
“We have always believed that the fact that so many rare diseases don’t have treatments or are not even being studied by researchers doesn’t reflect a lack of interest among academic or industry researchers. Rather, it reflects a lack of data to support research and to provide a fundamental understanding of the disease,” Dr. Fink said. “If NORD’s expanded research programs can draw the patient community together to provide that crucially needed data, we will have provided an important and essential service to patients, patient organizations, and researchers alike.”
Theodore Bosworth is a freelance journalist and editor specializing in medicine and health.
Rett syndrome: Looking to the future and the promise of gene therapy
The dream of curing genetic disorders has been a persistent but elusive goal, even before the human genome was mapped. Once mapping of the human genome was complete in 2001, an entirely new avenue of potential treatments and cures for genetic diseases and disorders was opened.1,2
The disorders best suited for targeted gene therapy are monogenic; however, tools and delivery methods for editing the human genome were limited and difficult to apply, until the advent of the CRISPR system in 2012.3 CRISPR (an acronym of clustered regularly interspaced short palindromic repeats) has changed the way in which gene therapy strategies are pursued, with dozens of companies leveraging a variety of platforms to create potentially life-changing therapies for devastating rare diseases and disorders.
One of the rare monogenic disorders that is embracing multiple gene therapy approaches is Rett syndrome, a rare, debilitating neurodevelopmental disorder. In this review, we explore the molecular cause of Rett syndrome, disease presentation, current treatments, ongoing clinical trials, and therapies that are on the horizon.
Underlying molecular cause
Rett syndrome is caused by mutations in, or the absence of, the MECP2 gene, which produces methyl-CpG binding protein 2 (MECP2). The syndrome was first described clinically in 1954 by the Austrian physician Andreas Rett; it would take until 1982 before the disorder was officially named, eponymously, in a seminal paper by Hagberg.4 After Hagberg’s characterization, Rett syndrome became the predominant global clinical diagnosis identified among cognitively impaired females, with an incidence of 1 in every 10,000 to 15,000.4
In 1999, mutations in, and deletions of, MECP2 were identified as the cause of Rett syndrome.4,5 MECP2 is located on the X chromosome, in the Xq28 region, making Rett syndrome an X-linked dominant disorder.6 Rett syndrome is seen predominantly in females who are mosaic for mutant or deleted MECP2. Random X chromosome inactivation results in some cells expressing the mutant MECP2 allele and other cells expressing the normal functioning MECP2 allele; the percentage of cells expressing the normal allele correlates with the degree of syndrome severity.7-9
The incidence of Rett syndrome is much lower in males, in whom the syndrome was originally thought to be lethal; many observed male cases are either mosaic or occur in XXY males.10,11
Approximately 95% of cases of Rett syndrome are due to de novo mutations in MECP2, with a handful of specific mutations and large deletions accounting for more than 85% of cases.12 The fact that Rett syndrome is monogenic and most cases are caused by, in total, only a handful of mutations or deletions makes the syndrome a promising candidate for gene therapy.
At the molecular level, it has been observed that the MECP2 mutations of Rett syndrome lead to loss of gene function, thus disrupting the ability of the MECP2 nuclear protein to regulate global gene transcription through its binding to methylated DNA sites.12 A large percentage of these missense and nonsense mutations lead to a truncated or nonfunctional protein.12
One of the ways in which MECP2 regulates transcription is as a component of heterochromatin condensates and by separation of heterochromatin and euchromatin.13-15 It has been observed that the cells of Rett syndrome patients have an altered chromatin state, potentially contributing to transcriptional dysregulation.16,17 Several mutations observed in Rett syndrome patients occur in crucial domains for heterochromatin condensate formation, which helps explain this cellular phenotype.13 Introduction of a engineered “mini” MECP2 in a murine model of Rett syndrome has resulted in partial rescue of heterochromatin condensate formation and transcriptional regulation – fostering the hypothesis that correcting those genetic changes could lead to a potential therapy.18
Beyond the role of MECP2 in heterochromatin condensate formation, the gene interacts with more than 40 proteins that have diverse roles in cellular function, epigenetic modulation, and neuronal development. This volume of interactions contributes to MECP2 being a global gene regulatory protein that has far-reaching effects on transcriptional regulation across the genome.19-22
Epigenetic dysregulation has been associated with neurodevelopmental and neuropsychiatric disorders.23 Both insulin-like growth factor 1 (IGF-1) and brain-derived neurotrophic factor are transcriptional targets of MECP2, and are involved in neuronal differentiation, synaptic function, and neurite outgrowth.12 This helps explain the neurodevelopmental phenotypes observed in MECP2-mutated patients.
Notably, although Rett syndrome patients experience neurodevelopmental phenotypes at the cellular level, neuronal death is not readily observed. That observation provides hope that an interventional therapy after onset of symptoms might still be of benefit.
Presentation
Early neurotypical development. A hallmark of Rett syndrome is neurotypical physical and mental development until 6 to 24 months of age.
Stagnation is the first stage of the syndrome, involving a small but rapid decline in habitual milestones, such motor and language skills.12 Subtle signs, such as microcephaly and hypotonia, can also arise at this time but might be missed.24
Rapid regression follows stagnation. Speech and motor delays and impaired gait and breathing occur;12,25 purposeful hand skills are lost, replaced by repetitive hand-wringing movements that are a hallmark of the syndrome.12,24 Seizures are observed; they become more common during the next stage.12
Plateau. Language advances can be observed, but further deficits are seen in motor skills and hand coordination.12
Late motor deterioration stage. Late physical deficits develop, leading to lifelong impairments. The physical deficits observed are the result of severe muscle weakness, usually resulting in wheelchair dependency.12
Plateau. Patients then reach a second plateau. Regression stops; deficient physical and cognitive states stabilize and are maintained.25
At all stages of Rett syndrome, the following are observed:
- Gastrointestinal problems.
- Sleep disturbances.
- Abnormal cardiorespiratory coupling.
- Greater-than-expected mortality.12
Final regression. The patient is fully dependent for the rest of their lifespan, partially due to seizure activity.26,27
A life-changing diagnosis
A diagnosis of Rett syndrome is life-changing for a patient’s family; access to supportive groups of other patients and their families is extremely beneficial. Two helpful organizations – the Rett Syndrome Research Trust28 and International Rett Syndrome Foundation,29 – offer patient support and community and fund research.
Because X chromosome inactivation is random in Rett syndrome, the individual patient can present with a wide variety of phenotypic combinations – making the patient, and their needs, unique.12 During stages of regression, patients often experience emotional dysregulation and anxiety, which is attributable to their increasing physical difficulties.30 They often exhibit combinations of uncontrolled movements, including repetitive rocking, scratching, and self-injurious behavior.30 For most, regression subsides after the first 5 years of alternating development and regression; after that, their ultimate symptoms persist for life.25
As patients mature, they need to be monitored for proper nutrition and scoliosis.25 As adults, they are at risk of pneumonia, respiratory distress, status epilepticus, osteopenia, and lack of adequate food or water because of impaired ability to feed.25
The lifespan of Rett syndrome patients has increased, thanks to improvements in health care, advances in technology, and early genetic testing, which allows for earlier diagnosis, intervention, and management of symptoms.
Current treatments
When a female patient presents with regression and loss of milestones, sequencing of MECP2 is performed to verify whether Rett syndrome is the cause, by detecting any of the known mutations. Multiplex ligation-dependent probe amplification is also performed to detect major deletions.25
All available treatments for Rett syndrome are symptomatic; intensive early intervention is practiced.31 Multidisciplinary management – medical, psychiatric, and physical – is introduced almost immediately after diagnosis. Following diagnosis, patients are prescribed anti-seizure, sleep, and anxiety medications.31 Electroencephalography can be performed to identify seizure type. Neuromuscular blockage drugs can be prescribed to help with gait and stereotypic hand movements.25
Handguards or splints to the elbows can be prescribed by an occupational therapist to improve hand movement.25 Physical therapy can improve mobility; hydrotherapy and hippotherapy have been successful in helping to maintain mobility and muscle support.32,33 Nutritional management is implemented to control caloric intake and maintain the vitamin D level.31 Some patients experience constipation and urinary retention, putting them at risk of nephrolithiasis.
Once the signs and symptoms of Rett syndrome progress, and milestones regress to a certain point, patients need constant, full-time care for the rest of their lives.34 As symptomatic interventions have greatly improved patient outcomes and it has been shown that about 70% can reach adulthood with a potential lifespan of about 50 years.25
Although there is no cure for Rett syndrome and treatments are symptomatic, ongoing studies – both clinical and preclinical – offer promise that treatments will be developed that work at molecular and genetic levels.
Clinical trials
Advances in understanding of Rett syndrome have led to many therapies in clinical trials, several of which show promise.
Trofinetide. One of the most promising targets for downstream therapy, mentioned earlier, is IGF-1, which was the target of a successful phase 3 clinical trial, LAVENDER (sponsored by Acadia Pharmaceuticals).35,36 This trial studied trofinetide, a synthetic IGF-1 analog that inhibits neuroinflammation, restores glial function, corrects synaptic deficiencies, and regulates oxidative stress response.12,37,38 Initial results from phase 2 and phase 3 trials indicate improved scores for treated patients in the Rett syndrome Behaviour Questionnaire (RSBQ) and Clinical Global Impression–Improvement (CGI-I) scores, while also showing improvements in the Communication and Symbolic Behavior Scales Developmental Profile Infant–Toddler Checklist–Social composite score.36,39
The most common adverse events seen with trofinetide were diarrhea and vomiting.
Acadia Pharmaceuticals has filed for approval of a new drug application for trofinetide with the Food and Drug Administration, for which the company has been granted Fast Track Status and orphan drug designations. Most (95%) subjects in the phase 3 LAVENDER trial elected to continue taking trofinetide in the subsequent open-label Lilac and Lilac-2 extension studies.36 A current open-label phase 2/3 trial is recruiting patients 2 to 5 years of age to evaluate trofinetide.40 It is expected that, in the near future, this could be a drug given to Rett patients as an FDA-approved treatment.
Blarcamesine. Another small molecule drug, blarcamesine (also known as ANAVEX2-73), a sigma-1 receptor agonist, produced promising results in phase 2 clinical trials in adult Rett syndrome patients. The drug is in a phase 2/3 clinical trial for pediatric Rett syndrome patients (sponsored by Anavex Life Sciences).41-43
Phase 2 results indicated statistically significant and clinically meaningful improvement in RSBQ and CGI-I scores with blarcamesine. Improvement was initially observed within 4 weeks after the start of treatment and was sustained throughout the study. The drug was shown to be well tolerated, with minimal adverse effects; no serious adverse events were recorded. These results were observed in adult patients, demonstrating that improvements in Rett syndrome are possible even after regression.
Blarcamesine activates the sigma 1 receptor, which is pivotal to restoring cellular homeostasis and restoring neuroplasticity – deficiencies of which have been linked to autophagy and glutamate toxicity. The drug has also been explored as a potential treatment for other neurological disorders.44-47 Improvements in blarcamesine-treated patients further correlated with lower levels of glutamate in cerebrospinal fluid, which is a Rett syndrome biomarker, supporting the proposition that behavioral improvements were due to drug intervention.48,49 The phase 2 trial was modified into a phase 3 trial and additional endpoints were added.41-43
All patients in the phase 2 adult trial elected to continue in the extension study.
Based on these promising data, Anavex is pursuing an approval pathway for adult patients, while continuing dosage optimization phase 2/3 trials and recruitment for a pediatric trial.42,43
Is the future about gene therapy?
TSHA-102 (miniMECP2). Taysha Gene Therapies is developing a promising gene therapy, TSHA-102, for Rett syndrome, and is aiming to begin phase 1/2 clinical trials in 2022.50 The technology for this therapy relies on the delivery of a fragment of MECP2 (known as miniMECP2), which is regulated by a built-in microRNA regulator (miR-responsive auto-regulatory element, or miRARE) to help ameliorate MECP2 dosage toxicity. (Overexpression of MECP2 is toxic to neurons, which has made traditional [so to speak] gene replacement therapy difficult in Rett syndrome: Levels of MECP2 need to be tightly regulated, and the Taysha microRNA technology regulates levels of miniMECP2, thus reducing toxicity.)
The Taysha microRNA technology has yielded promising results in mouse studies for Rett syndrome; results indicate a lengthening of lifespan and delayed onset of gait abnormalities.51 TSHA-102 is in the preclinical stage but offers promise that it will be the first gene therapy for Rett syndrome to enter clinical trials.
As the field of gene therapy advances, several promising technologies are on the horizon that could potentially have disease-altering impacts on Rett syndrome. These therapies are divided into two broad categories: those at the gene level and those at the transcription and protein level. A few of these approaches are highlighted below.
Gene replacement involves adding a full or partial copy of MECP2 to neuronal cells. This type of therapy presents challenges, from delivery of the new gene to dosage concerns, because MECP2 can be toxic if overexpressed.52-54 Groundbreaking work was done in mouse models involving truncated MECP2, exhibiting phenotypic rescue and validating the gene-replacement approach.18 This strategy is being pursued by Neurogene, which has a uinique technology that allows for tuning of the gene’s expression to get the correct protein levels in the patient. Promising data was presented this year at the American Society of Gene and Cell Therapy conference.55
Early gene replacement therapy studies also laid the foundation for the minMECP2 and microRNA approach being used by Taysha Gene Therapies (discussed above).51
“Correcting” DNA mutations. A different approach at the genetic level involves “correcting” mutations in MECP2 at the DNA level. This is possible because, in a large subset of Rett syndrome patients who have the same missense or nonsense mutations, by using CRISPR, a gene editing tool (discussed above) a single base pair can be corrected.56,57 Previous research, in a Rett syndrome-model of induced pluripotent stem cells, showed that this type of editing is possible – and effective.52 An approach with particular promise involves use of a class of CRISPR proteins known as base editors that are able to specifically alter a single base of DNA.57 The technique has the potential to address many of the mutations seen in Rett syndrome; research on this type of technology is being pursued by Beam Therapeutics, and has the potential to impact Rett syndrome.58
Another promising “correction” approach is exonic editing, in which a much larger section of DNA – potentially, exons 3 and 4, which, taken together, comprise 97% of known MECP2 mutations seen in Rett syndrome – are replaced.59
In both CRISPR and exonic editing therapeutic approaches, endogenous levels of MECP2 expression would be maintained. Of note, both approaches are being pursued for use in treating other genetic disorders, which provides a boost in scaling-up work on addressing safety and efficacy concerns that accompany gene-editing approaches.58 One advantage to the DNA correction approach is that is has the potential to be a “one-and-done” treatment.
“Correcting” RNA. Beyond directly editing DNA, several therapeutic approaches are exploring the ability to edit RNA or to provide the protein directly to cells as enzyme replacement therapy. Such an RNA correction strategy leverages a technology that takes advantage of cells’ natural RNA editor, known as adenosine deaminase acting on RNA (ADAR), which corrects errors in cells’ RNA by providing specific guides to the cell. ADAR can be targeted to fix mutations in the MECP2 RNA transcript, resulting in a “corrected” MECP2 protein.60,61 This technology has delivered promising proof-of-concept evidence in cells and in murine models, and is in the therapeutic pipeline at VICO Therapeutics.62
Shape Therapeutics has also leveraged ADAR to “correct” mutated RNA; Rett syndrome is among the top priorities in the company’s pipeline.
Worth noting is that there are several advantages to the “correction” approach:
- Leveraging internal repair mechanisms minimizes the immune response.
- The flexibility of correction means that it can be used to address many of the mutations that cause Rett syndrome.63
Enzyme replacement therapy, in which the MECP2 protein produced by MECP2 would be directly replaced, is being explored in Rett syndrome patients. This technology has been used successfully in Pompe disease; however, Rett syndrome presents its own challenge because MECP2 needs to be delivered to the brain and neuronal cells.64
Where does this work stand? The technologies described in this section are in preclinical stages of study. Nonetheless, it is expected that several will enter human clinical trials during the next 5 years.
Conclusion
A diagnosis of Rett syndrome is a life-altering event for patients and their family. But there is more hope than ever for effective therapies and, eventually, a cure.
Multiple late-stage clinical trials in progress are demonstrating promising results from therapeutic products, with minimal adverse events. Remarkably, these interventions have delivered improvements to adult patients after regression has stabilized. With rapid progress being made in the field of gene therapy, several technologies for which are focused on Rett syndrome, a hopeful picture is emerging: that therapeutic intervention will be possible before regression, thus effectively treating and, potentially, even curing Rett syndrome.
The landscape is broadening. Add to this hope for approved therapies is the fact that Rett syndrome isn’t the only target being pursued with such strategies; in fact, researchers in the larger field of neurodevelopmental disorder study are working together to find common solutions to shared challenges – from how therapies are designed and delivered to how toxicity is minimized. Much of what is being explored in the Rett syndrome field is also under investigation in other neurodevelopmental syndromes, including Angelman, Prader-Willi, chromosome 15q11.2-13.1 duplication (dup15q), and Fragile X syndrome. This kind of parallel investigation benefits all parties and optimizes a treatment platform so that it can be applied across more than a single disorder.
Like many monogenic disorders, Rett syndrome is entering an exciting stage – at which the words “treatment” and “cure” can be spoken with intent and vision, not just wide-eyed optimism. These words portend real promise for patients who carry the weight of a diagnosis of Rett syndrome, and for their families.
Ms. Ambrose is a student in the master’s of science in human genetics and genomic data analytics program, Keck Graduate Institute, Claremont, Calif. Dr. Bailus is an assistant professor of genetics, Keck Graduate Institute. The authors report no conflict of interest related to this article.
References
1. Lander ES et al; . Initial sequencing and analysis of the human genome. Nature. 2001 Feb 15;409(6822):860-921. doi: 10.1038/35057062.
2. Venter JC et al. The sequence of the human genome. Science. 2001 Feb 16;291(5507):1304-51. doi: 10.1126/science.1058040.
3. Jinek M et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012 Aug 17;337(6096):816-21. doi: 10.1126/science.1225829.
4. Percy A. The American history of Rett syndrome. Pediatr Neurol. 2014 Jan;50(1):1-3. doi: 10.1016/j.pediatrneurol.2013.08.018.
5. Amir RE et al. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet. 1999 Oct;23(2):185-8. doi: 10.1038/13810.
6. Pitzianti MB et al. Rett syndrome in males: The different clinical course in two brothers with the same microduplication MECP2 Xq28. Int J Environ Res Public Health. 2019 Aug;16(17):3075. doi: 10.3390/ijerph16173075.
7. Ribeiro MC, MacDonald JL. Sex differences in Mecp2-mutant Rett syndrome model mice and the impact of cellular mosaicism in phenotype development. Brain Res. 2020 Feb 15;1729:146644. doi: 10.1016/j.brainres.2019.146644.
8. Bao X et al. X chromosome inactivation in Rett syndrome and its correlations with MECP2 mutations and phenotype. J Child Neurol. 2008 Jan;23(1):22-5. doi: 10.1177/0883073807307077.
9. Knudsen GPS et al. Increased skewing of X chromosome inactivation in Rett syndrome patients and their mothers. Eur J Hum Genet. 2006 Jul;14(11):1189-94. doi: 10.1038/sj.ejhg.5201682.
10. Chahil G et al. Rett syndrome in males: A case report and review of literature. Cureus. 2018;10(10):e3414. doi: 10.7759/cureus.3414.
11. Reichow B et al. Brief report: Systematic review of Rett syndrome in males. J Autism Dev Disord. 2015 Oct;45(10):3377-83. doi: 10.1007/s10803-015-2519-1.
12. Vashi N, Justice MJ. Treating Rett syndrome: From mouse models to human therapies. Mamm Genome. 2019 Jun;30(5-6):90-110. doi: 10.1007/s00335-019-09793-5.
13. Li CH et al. MeCP2 links heterochromatin condensates and neurodevelopmental disease. Nature. 2020 Oct;586(7829):440-4. doi: 10.1038/s41586-020-2574-4.
14. Schmidt A et al. MeCP2 and chromatin compartmentalization. Cells. 2020 Apr;9(4):878. doi: 10.3390/cells9040878.
15. Wang L et al. Rett syndrome-causing mutations compromise MeCP2-mediated liquid-liquid phase separation of chromatin. Cell Res. 2020 May;30(5):393-407. doi: 10.1038/s41422-020-0288-7.
16. Lin P et al. Transcriptome analysis of human brain tissue identifies reduced expression of complement complex C1Q genes in Rett syndrome. BMC Genomics. 2016;17:427. doi: 10.1186/s12864-016-2746-7.
17. Tudor M et al. Transcriptional profiling of a mouse model for Rett syndrome reveals subtle transcriptional changes in the brain. Proc Natl Acad Sci U S A. 2002 Nov 26;99(24):15536-41. doi: 10.1073/pnas.242566899.
18. Tillotson R et al. Radically truncated MeCP2 rescues Rett syndrome–like neurological defects. Nature. 2017 Oct 19;550(7676):398-401. doi: 10.1038/nature24058.
19. Connolly DR, Zhou Z. Genomic insights into MeCP2 function: A role for the maintenance of chromatin architecture. Curr Opin Neurobiol. 2019 Dec;59:174-9. doi: 10.1016/j.conb.2019.07.002.
20. Johnson BS et al. Biotin tagging of MeCP2 in mice reveals contextual insights into the Rett syndrome transcriptome. Nat Med. 2017 Oct;23(10):1203-14. doi: 10.1038/nm.4406.
21. Gabel HW et al. Disruption of DNA-methylation–dependent long gene repression in Rett syndrome. Nature. 2015 Jun 4;522(7554):89-93. doi: 10.1038/nature14319.
22. Lyst MJ, Bird A. Rett syndrome: A complex disorder with simple roots. Nat Rev Genet. 2015 May;16(5):261-75. doi: 10.1038/nrg3897.
23. Kuehner JN et al. Epigenetic regulations in neuropsychiatric disorders. Front Genet. 2019 Apr 4;10:268. doi: 10.3389/fgene.2019.00268.
24. Pejhan S, Rastegar M. Role of DNA methyl-CpG-binding protein MeCP2 in Rett syndrome pathobiology and mechanism of disease. Biomolecules. 2021 Jan;11(1):75. doi: 10.3390/biom11010075.
25. Fu C et al. Consensus guidelines on managing Rett syndrome across the lifespan. BMJ Paediatr Open. 2020;4(1):e000717. doi: 10.1136/bmjpo-2020-000717.
26. Operto FF et al. Epilepsy and genetic in Rett syndrome: A review. Brain Behav. 2019 May;9(5):e01250. doi: 10.1002/brb3.1250.
27. Nissenkorn A et al. Epilepsy in Rett syndrome – The experience of a National Rett Center. Epilepsia. 2010 Jul;51(7):1252-8. doi: 10.1111/j.1528-1167.2010.02597.x.
28. Welcome to the Rett cure community. Rett Syndrome Research Trust [Internet]. Updated Feb 8, 2022. Accessed Feb 23, 2022. https://reverserett.org.
29. About Rett syndrome. International Rett Syndrome Foundation [Internet]. Updated Jan 4, 2022. Accessed Feb 23, 2022. http://www.rettsyndrome.org.
30. Singh J, Santosh P. Key issues in Rett syndrome: Emotional, behavioural and autonomic dysregulation (EBAD) – A target for clinical trials. Orphanet J Rare Dis. 2018 Jul 31;13(1):128. doi: 10.1186/s13023-018-0873-8.
31. Banerjee A et al. Towards a better diagnosis and treatment of Rett syndrome: A model synaptic disorder. Brain. 2019 Feb 1;142(2):239-48. doi: 10.1093/brain/awy323.
32. Ager S et al. Parental experiences of scoliosis management in Rett syndrome. Disabil Rehabil. 2009 Sep 19;31(23):1917-24. doi: 10.1080/09638280902846392.
33. Budden SS. Management of Rett syndrome: A ten year experience. Neuropediatrics. 1995;26(2):75-7. doi: 10.1055/s-2007-979727.
34. Ip JPK et al. Rett syndrome: Insights into genetic, molecular and circuit mechanisms. Nat Rev Neurosci. 2018 Jun;19(6):368-82. doi: 10.1038/s41583-018-0006-3.
35. Acadia Pharmaceuticals Inc. Study of trofinetide for the treatment of girls and women with Rett syndrome (LAVENDER™). ClinicalTrials.gov identifier: NCT04181723. Updated Feb 17, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT04181723.
36. Acadia Pharmaceuticals announces positive top-line results from the pivotal phase 3 LAVENDER trial of trofinetide in Rett syndrome. Press release. Acadia Pharmaceuticals Inc. Dec 6, 2021. Accessed Feb 23, 2022. https://ir.acadia-pharm.com/news-releases/news-release-details/acadia-pharmaceuticals-announces-positive-top-line-results-1.
37. Copping NA et al. Emerging gene and small molecule therapies for the neurodevelopmental disorder Angelman syndrome. Neurotherapeutics. 2021 Jul;18(3):1535-47. doi: 10.1007/s13311-021-01082-x.
38. Riikonen R. Insulin-like growth factors in the pathogenesis of neurological diseases in children. Int J Mol Sci. 2017 Sep;18(10):2056. doi: 10.3390/ijms18102056.
39. Glaze DG et al; Rett 002 Study Group. Double-blind, randomized, placebo-controlled study of trofinetide in pediatric Rett syndrome. Neurology. 2019 April 16;92(16):e1912-e1925. doi: 10.1212/WNL.0000000000007316.
40. Acadia Pharmaceuticals Inc. An open-label study of trofinetide for the treatment of girls two to five years of age who have Rett syndrome (DAFFODIL™). ClinicalTrials.gov Identifier: NCT04988867. Updated Jan 24, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT04988867.
41. Anavex Life Sciences announces ANAVEX®2-73 meets primary and secondary endpoints in clinical trial. Press release. Anavex Life Sciences Corp. Dec 15, 2020. Accessed Feb 23, 2022. http://www.anavex.com/post/anavex-life-sciences-announces-anavex-2-73-meets-primary-and-secondary-endpoints-in-clinical-trial.
42. Anavex Life Sciences Corp. ANAVEX2-73 study in patients with Rett syndrome (AVATAR). ClinicalTrials.gov Identifier: NCT03941444. Updated Jan 27, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT03941444.
43. Anavex Life Sciences Corp. ANAVEX2-73 study in pediatric patients with Rett syndrome (EXCELLENCE). ClinicalTrials.gov Identifier: NCT04304482. Updated Sep 28, 2021. Accessed Feb 23, 2022. http://www.clinicaltrials.gov/ct2/show/NCT04304482.
44.Christ MG et al. The Sigma-1 receptor at the crossroad of proteostasis, neurodegeneration, and autophagy. Trends Neurosci. 2020 Feb;43(2):79-81. doi: 10.1016/j.tins.2019.12.002.
45. Kaufmann WE et al. ANAVEX®2-73 (blarcamesine), a sigma-1 receptor agonist, ameliorates neurologic impairments in a mouse model of Rett syndrome. Pharmacol Biochem Behav. 2019 Dec;187:172796. doi: 10.1016/j.pbb.2019.172796.
46. Brimson JM et al. Dipentylammonium binds to the sigma-1 receptor and protects against glutamate toxicity, attenuates dopamine toxicity and potentiates neurite outgrowth in various cultured cell lines. Neurotox Res. 2018 Aug;34(2):263-72. doi: 10.1007/s12640-018-9883-5.
47. Kourrich S et al. The sigma-1 receptor: roles in neuronal plasticity and disease. Trends Neurosci. 2012 Dec;35(12):762-71. doi: 10.1016/j.tins.2012.09.007.
48. Lappalainen R, Riikonen RS. High levels of cerebrospinal fluid glutamate in Rett syndrome. Pediatr Neurol. 1996 Oct;15(3):213-6. doi: 10.1016/s0887-8994(96)00218-4.
49. Hamberger A et al. Elevated CSF glutamate in Rett syndrome. Neuropediatrics. 1992;23(4):212-3. doi: 10.1055/s-2008-1071344.
50. Inacio P. FDA acts to support development of potential gene therapy, TSHA-102. Rett Syndrome News [Internet]. Oct 16, 2020. Accessed Feb 23, 2022. https://rettsyndromenews.com/2020/10/16/fda-grants-orphan-drug-rare-pediatric-disease-status-to-tsha-102-potential-rett-gene-therapy.
51. Sinnett SE et al. Engineered microRNA-based regulatory element permits safe high-dose miniMECP2 gene therapy in Rett mice. Brain. 2021 Nov 29;144(10):3005-19. doi: 10.1093/brain/awab182.
52. Le TTH et al. Efficient and precise CRISPR/Cas9-mediated MECP2 modifications in human-induced pluripotent stem cells. Front Genet. 2019 Jul 2;10:625. doi: 10.3389/fgene.2019.00625.
53. Koerner MV et al. Toxicity of overexpressed MeCP2 is independent of HDAC3 activity. Genes Dev. 2018;32(23-24):1514-24. doi: 10.1101/gad.320325.118.
54. Heckman LD et al. Rett-causing mutations reveal two domains critical for MeCP2 function and for toxicity in MECP2 duplication syndrome mice. Elife. 2014;3:e02676. doi: 10.7554/eLife.02676.
55. Neurogene announces new development program in Rett syndrome utilizing novel EXACT technology platform [Internet]. Accessed Aug 12, 2022. https://www.neurogene.com/press-releases/neurogene-announces-new-development-program-in-rett-syndrome-utilizing-novel-exact-technology-platform/
56. Anzalone AV et al. Genome editing with CRISPR-Cas nucleases, base editors, transposases and prime editors. Nat Biotechnol. 2020 Jul;38(7):824-44. doi: 10.1038/s41587-020-0561-9.
57. Gaudelli NM, Komor AC, Rees HA, et al. Programmable base editing of A●T to G●C in genomic DNA without DNA cleavage. Nature. 2017 Nov 23;551(7681):464-71. doi: 10.1038/nature24644.
58. Coenraads M. How RSRT is driving the search for a Rett cure. Rett Syndrome Research Trust [Internet]. Dec 7, 2021. Accessed Feb 23, 2022. https://rettnews.org/articles/how-rsrt-is-driving-the-search-for-a-rett-cure.
59. Cutting-edge technologies to repair the underlying mutations that cause Rett. Rett Syndrome Research Trust [Internet]. Updated Nov 3, 2021. Accessed Feb 23, 2022. https://reverserett.org/research/cures/gene-editing.
60. Sinnamon JR et al. In vivo repair of a protein underlying a neurological disorder by programmable RNA editing. Cell Rep. 2020 Jul 14;32(2):107878. doi: 10.1016/j.celrep.2020.107878.
61. Sinnamon JR et al. Site-directed RNA repair of endogenous Mecp2 RNA in neurons. Proc Natl Acad Sci U S A. 2017 Oct 31;114(44):E9395-E9402. doi: 10.1073/pnas.1715320114.
62. Pipeline. VICO Therapeutics [Internet]. Updated Nov 5, 2021. Accessed Feb 23, 2022. https://vicotx.com/pipeline.
63. Therapeutics platform. Shape Therapeutics [Internet]. Updated Feb 20, 2021. Accessed Feb 23, 2022.
https://live-shapetx.pantheonsite.io/therapeutics-platform.
64. Koeberl DD et al. Glycogen storage disease types I and II: Treatment updates. J Inherit Metab Dis. 2007 Apr;30(2):159-64. doi: 10.1007/s10545-007-0519-9.
The dream of curing genetic disorders has been a persistent but elusive goal, even before the human genome was mapped. Once mapping of the human genome was complete in 2001, an entirely new avenue of potential treatments and cures for genetic diseases and disorders was opened.1,2
The disorders best suited for targeted gene therapy are monogenic; however, tools and delivery methods for editing the human genome were limited and difficult to apply, until the advent of the CRISPR system in 2012.3 CRISPR (an acronym of clustered regularly interspaced short palindromic repeats) has changed the way in which gene therapy strategies are pursued, with dozens of companies leveraging a variety of platforms to create potentially life-changing therapies for devastating rare diseases and disorders.
One of the rare monogenic disorders that is embracing multiple gene therapy approaches is Rett syndrome, a rare, debilitating neurodevelopmental disorder. In this review, we explore the molecular cause of Rett syndrome, disease presentation, current treatments, ongoing clinical trials, and therapies that are on the horizon.
Underlying molecular cause
Rett syndrome is caused by mutations in, or the absence of, the MECP2 gene, which produces methyl-CpG binding protein 2 (MECP2). The syndrome was first described clinically in 1954 by the Austrian physician Andreas Rett; it would take until 1982 before the disorder was officially named, eponymously, in a seminal paper by Hagberg.4 After Hagberg’s characterization, Rett syndrome became the predominant global clinical diagnosis identified among cognitively impaired females, with an incidence of 1 in every 10,000 to 15,000.4
In 1999, mutations in, and deletions of, MECP2 were identified as the cause of Rett syndrome.4,5 MECP2 is located on the X chromosome, in the Xq28 region, making Rett syndrome an X-linked dominant disorder.6 Rett syndrome is seen predominantly in females who are mosaic for mutant or deleted MECP2. Random X chromosome inactivation results in some cells expressing the mutant MECP2 allele and other cells expressing the normal functioning MECP2 allele; the percentage of cells expressing the normal allele correlates with the degree of syndrome severity.7-9
The incidence of Rett syndrome is much lower in males, in whom the syndrome was originally thought to be lethal; many observed male cases are either mosaic or occur in XXY males.10,11
Approximately 95% of cases of Rett syndrome are due to de novo mutations in MECP2, with a handful of specific mutations and large deletions accounting for more than 85% of cases.12 The fact that Rett syndrome is monogenic and most cases are caused by, in total, only a handful of mutations or deletions makes the syndrome a promising candidate for gene therapy.
At the molecular level, it has been observed that the MECP2 mutations of Rett syndrome lead to loss of gene function, thus disrupting the ability of the MECP2 nuclear protein to regulate global gene transcription through its binding to methylated DNA sites.12 A large percentage of these missense and nonsense mutations lead to a truncated or nonfunctional protein.12
One of the ways in which MECP2 regulates transcription is as a component of heterochromatin condensates and by separation of heterochromatin and euchromatin.13-15 It has been observed that the cells of Rett syndrome patients have an altered chromatin state, potentially contributing to transcriptional dysregulation.16,17 Several mutations observed in Rett syndrome patients occur in crucial domains for heterochromatin condensate formation, which helps explain this cellular phenotype.13 Introduction of a engineered “mini” MECP2 in a murine model of Rett syndrome has resulted in partial rescue of heterochromatin condensate formation and transcriptional regulation – fostering the hypothesis that correcting those genetic changes could lead to a potential therapy.18
Beyond the role of MECP2 in heterochromatin condensate formation, the gene interacts with more than 40 proteins that have diverse roles in cellular function, epigenetic modulation, and neuronal development. This volume of interactions contributes to MECP2 being a global gene regulatory protein that has far-reaching effects on transcriptional regulation across the genome.19-22
Epigenetic dysregulation has been associated with neurodevelopmental and neuropsychiatric disorders.23 Both insulin-like growth factor 1 (IGF-1) and brain-derived neurotrophic factor are transcriptional targets of MECP2, and are involved in neuronal differentiation, synaptic function, and neurite outgrowth.12 This helps explain the neurodevelopmental phenotypes observed in MECP2-mutated patients.
Notably, although Rett syndrome patients experience neurodevelopmental phenotypes at the cellular level, neuronal death is not readily observed. That observation provides hope that an interventional therapy after onset of symptoms might still be of benefit.
Presentation
Early neurotypical development. A hallmark of Rett syndrome is neurotypical physical and mental development until 6 to 24 months of age.
Stagnation is the first stage of the syndrome, involving a small but rapid decline in habitual milestones, such motor and language skills.12 Subtle signs, such as microcephaly and hypotonia, can also arise at this time but might be missed.24
Rapid regression follows stagnation. Speech and motor delays and impaired gait and breathing occur;12,25 purposeful hand skills are lost, replaced by repetitive hand-wringing movements that are a hallmark of the syndrome.12,24 Seizures are observed; they become more common during the next stage.12
Plateau. Language advances can be observed, but further deficits are seen in motor skills and hand coordination.12
Late motor deterioration stage. Late physical deficits develop, leading to lifelong impairments. The physical deficits observed are the result of severe muscle weakness, usually resulting in wheelchair dependency.12
Plateau. Patients then reach a second plateau. Regression stops; deficient physical and cognitive states stabilize and are maintained.25
At all stages of Rett syndrome, the following are observed:
- Gastrointestinal problems.
- Sleep disturbances.
- Abnormal cardiorespiratory coupling.
- Greater-than-expected mortality.12
Final regression. The patient is fully dependent for the rest of their lifespan, partially due to seizure activity.26,27
A life-changing diagnosis
A diagnosis of Rett syndrome is life-changing for a patient’s family; access to supportive groups of other patients and their families is extremely beneficial. Two helpful organizations – the Rett Syndrome Research Trust28 and International Rett Syndrome Foundation,29 – offer patient support and community and fund research.
Because X chromosome inactivation is random in Rett syndrome, the individual patient can present with a wide variety of phenotypic combinations – making the patient, and their needs, unique.12 During stages of regression, patients often experience emotional dysregulation and anxiety, which is attributable to their increasing physical difficulties.30 They often exhibit combinations of uncontrolled movements, including repetitive rocking, scratching, and self-injurious behavior.30 For most, regression subsides after the first 5 years of alternating development and regression; after that, their ultimate symptoms persist for life.25
As patients mature, they need to be monitored for proper nutrition and scoliosis.25 As adults, they are at risk of pneumonia, respiratory distress, status epilepticus, osteopenia, and lack of adequate food or water because of impaired ability to feed.25
The lifespan of Rett syndrome patients has increased, thanks to improvements in health care, advances in technology, and early genetic testing, which allows for earlier diagnosis, intervention, and management of symptoms.
Current treatments
When a female patient presents with regression and loss of milestones, sequencing of MECP2 is performed to verify whether Rett syndrome is the cause, by detecting any of the known mutations. Multiplex ligation-dependent probe amplification is also performed to detect major deletions.25
All available treatments for Rett syndrome are symptomatic; intensive early intervention is practiced.31 Multidisciplinary management – medical, psychiatric, and physical – is introduced almost immediately after diagnosis. Following diagnosis, patients are prescribed anti-seizure, sleep, and anxiety medications.31 Electroencephalography can be performed to identify seizure type. Neuromuscular blockage drugs can be prescribed to help with gait and stereotypic hand movements.25
Handguards or splints to the elbows can be prescribed by an occupational therapist to improve hand movement.25 Physical therapy can improve mobility; hydrotherapy and hippotherapy have been successful in helping to maintain mobility and muscle support.32,33 Nutritional management is implemented to control caloric intake and maintain the vitamin D level.31 Some patients experience constipation and urinary retention, putting them at risk of nephrolithiasis.
Once the signs and symptoms of Rett syndrome progress, and milestones regress to a certain point, patients need constant, full-time care for the rest of their lives.34 As symptomatic interventions have greatly improved patient outcomes and it has been shown that about 70% can reach adulthood with a potential lifespan of about 50 years.25
Although there is no cure for Rett syndrome and treatments are symptomatic, ongoing studies – both clinical and preclinical – offer promise that treatments will be developed that work at molecular and genetic levels.
Clinical trials
Advances in understanding of Rett syndrome have led to many therapies in clinical trials, several of which show promise.
Trofinetide. One of the most promising targets for downstream therapy, mentioned earlier, is IGF-1, which was the target of a successful phase 3 clinical trial, LAVENDER (sponsored by Acadia Pharmaceuticals).35,36 This trial studied trofinetide, a synthetic IGF-1 analog that inhibits neuroinflammation, restores glial function, corrects synaptic deficiencies, and regulates oxidative stress response.12,37,38 Initial results from phase 2 and phase 3 trials indicate improved scores for treated patients in the Rett syndrome Behaviour Questionnaire (RSBQ) and Clinical Global Impression–Improvement (CGI-I) scores, while also showing improvements in the Communication and Symbolic Behavior Scales Developmental Profile Infant–Toddler Checklist–Social composite score.36,39
The most common adverse events seen with trofinetide were diarrhea and vomiting.
Acadia Pharmaceuticals has filed for approval of a new drug application for trofinetide with the Food and Drug Administration, for which the company has been granted Fast Track Status and orphan drug designations. Most (95%) subjects in the phase 3 LAVENDER trial elected to continue taking trofinetide in the subsequent open-label Lilac and Lilac-2 extension studies.36 A current open-label phase 2/3 trial is recruiting patients 2 to 5 years of age to evaluate trofinetide.40 It is expected that, in the near future, this could be a drug given to Rett patients as an FDA-approved treatment.
Blarcamesine. Another small molecule drug, blarcamesine (also known as ANAVEX2-73), a sigma-1 receptor agonist, produced promising results in phase 2 clinical trials in adult Rett syndrome patients. The drug is in a phase 2/3 clinical trial for pediatric Rett syndrome patients (sponsored by Anavex Life Sciences).41-43
Phase 2 results indicated statistically significant and clinically meaningful improvement in RSBQ and CGI-I scores with blarcamesine. Improvement was initially observed within 4 weeks after the start of treatment and was sustained throughout the study. The drug was shown to be well tolerated, with minimal adverse effects; no serious adverse events were recorded. These results were observed in adult patients, demonstrating that improvements in Rett syndrome are possible even after regression.
Blarcamesine activates the sigma 1 receptor, which is pivotal to restoring cellular homeostasis and restoring neuroplasticity – deficiencies of which have been linked to autophagy and glutamate toxicity. The drug has also been explored as a potential treatment for other neurological disorders.44-47 Improvements in blarcamesine-treated patients further correlated with lower levels of glutamate in cerebrospinal fluid, which is a Rett syndrome biomarker, supporting the proposition that behavioral improvements were due to drug intervention.48,49 The phase 2 trial was modified into a phase 3 trial and additional endpoints were added.41-43
All patients in the phase 2 adult trial elected to continue in the extension study.
Based on these promising data, Anavex is pursuing an approval pathway for adult patients, while continuing dosage optimization phase 2/3 trials and recruitment for a pediatric trial.42,43
Is the future about gene therapy?
TSHA-102 (miniMECP2). Taysha Gene Therapies is developing a promising gene therapy, TSHA-102, for Rett syndrome, and is aiming to begin phase 1/2 clinical trials in 2022.50 The technology for this therapy relies on the delivery of a fragment of MECP2 (known as miniMECP2), which is regulated by a built-in microRNA regulator (miR-responsive auto-regulatory element, or miRARE) to help ameliorate MECP2 dosage toxicity. (Overexpression of MECP2 is toxic to neurons, which has made traditional [so to speak] gene replacement therapy difficult in Rett syndrome: Levels of MECP2 need to be tightly regulated, and the Taysha microRNA technology regulates levels of miniMECP2, thus reducing toxicity.)
The Taysha microRNA technology has yielded promising results in mouse studies for Rett syndrome; results indicate a lengthening of lifespan and delayed onset of gait abnormalities.51 TSHA-102 is in the preclinical stage but offers promise that it will be the first gene therapy for Rett syndrome to enter clinical trials.
As the field of gene therapy advances, several promising technologies are on the horizon that could potentially have disease-altering impacts on Rett syndrome. These therapies are divided into two broad categories: those at the gene level and those at the transcription and protein level. A few of these approaches are highlighted below.
Gene replacement involves adding a full or partial copy of MECP2 to neuronal cells. This type of therapy presents challenges, from delivery of the new gene to dosage concerns, because MECP2 can be toxic if overexpressed.52-54 Groundbreaking work was done in mouse models involving truncated MECP2, exhibiting phenotypic rescue and validating the gene-replacement approach.18 This strategy is being pursued by Neurogene, which has a uinique technology that allows for tuning of the gene’s expression to get the correct protein levels in the patient. Promising data was presented this year at the American Society of Gene and Cell Therapy conference.55
Early gene replacement therapy studies also laid the foundation for the minMECP2 and microRNA approach being used by Taysha Gene Therapies (discussed above).51
“Correcting” DNA mutations. A different approach at the genetic level involves “correcting” mutations in MECP2 at the DNA level. This is possible because, in a large subset of Rett syndrome patients who have the same missense or nonsense mutations, by using CRISPR, a gene editing tool (discussed above) a single base pair can be corrected.56,57 Previous research, in a Rett syndrome-model of induced pluripotent stem cells, showed that this type of editing is possible – and effective.52 An approach with particular promise involves use of a class of CRISPR proteins known as base editors that are able to specifically alter a single base of DNA.57 The technique has the potential to address many of the mutations seen in Rett syndrome; research on this type of technology is being pursued by Beam Therapeutics, and has the potential to impact Rett syndrome.58
Another promising “correction” approach is exonic editing, in which a much larger section of DNA – potentially, exons 3 and 4, which, taken together, comprise 97% of known MECP2 mutations seen in Rett syndrome – are replaced.59
In both CRISPR and exonic editing therapeutic approaches, endogenous levels of MECP2 expression would be maintained. Of note, both approaches are being pursued for use in treating other genetic disorders, which provides a boost in scaling-up work on addressing safety and efficacy concerns that accompany gene-editing approaches.58 One advantage to the DNA correction approach is that is has the potential to be a “one-and-done” treatment.
“Correcting” RNA. Beyond directly editing DNA, several therapeutic approaches are exploring the ability to edit RNA or to provide the protein directly to cells as enzyme replacement therapy. Such an RNA correction strategy leverages a technology that takes advantage of cells’ natural RNA editor, known as adenosine deaminase acting on RNA (ADAR), which corrects errors in cells’ RNA by providing specific guides to the cell. ADAR can be targeted to fix mutations in the MECP2 RNA transcript, resulting in a “corrected” MECP2 protein.60,61 This technology has delivered promising proof-of-concept evidence in cells and in murine models, and is in the therapeutic pipeline at VICO Therapeutics.62
Shape Therapeutics has also leveraged ADAR to “correct” mutated RNA; Rett syndrome is among the top priorities in the company’s pipeline.
Worth noting is that there are several advantages to the “correction” approach:
- Leveraging internal repair mechanisms minimizes the immune response.
- The flexibility of correction means that it can be used to address many of the mutations that cause Rett syndrome.63
Enzyme replacement therapy, in which the MECP2 protein produced by MECP2 would be directly replaced, is being explored in Rett syndrome patients. This technology has been used successfully in Pompe disease; however, Rett syndrome presents its own challenge because MECP2 needs to be delivered to the brain and neuronal cells.64
Where does this work stand? The technologies described in this section are in preclinical stages of study. Nonetheless, it is expected that several will enter human clinical trials during the next 5 years.
Conclusion
A diagnosis of Rett syndrome is a life-altering event for patients and their family. But there is more hope than ever for effective therapies and, eventually, a cure.
Multiple late-stage clinical trials in progress are demonstrating promising results from therapeutic products, with minimal adverse events. Remarkably, these interventions have delivered improvements to adult patients after regression has stabilized. With rapid progress being made in the field of gene therapy, several technologies for which are focused on Rett syndrome, a hopeful picture is emerging: that therapeutic intervention will be possible before regression, thus effectively treating and, potentially, even curing Rett syndrome.
The landscape is broadening. Add to this hope for approved therapies is the fact that Rett syndrome isn’t the only target being pursued with such strategies; in fact, researchers in the larger field of neurodevelopmental disorder study are working together to find common solutions to shared challenges – from how therapies are designed and delivered to how toxicity is minimized. Much of what is being explored in the Rett syndrome field is also under investigation in other neurodevelopmental syndromes, including Angelman, Prader-Willi, chromosome 15q11.2-13.1 duplication (dup15q), and Fragile X syndrome. This kind of parallel investigation benefits all parties and optimizes a treatment platform so that it can be applied across more than a single disorder.
Like many monogenic disorders, Rett syndrome is entering an exciting stage – at which the words “treatment” and “cure” can be spoken with intent and vision, not just wide-eyed optimism. These words portend real promise for patients who carry the weight of a diagnosis of Rett syndrome, and for their families.
Ms. Ambrose is a student in the master’s of science in human genetics and genomic data analytics program, Keck Graduate Institute, Claremont, Calif. Dr. Bailus is an assistant professor of genetics, Keck Graduate Institute. The authors report no conflict of interest related to this article.
References
1. Lander ES et al; . Initial sequencing and analysis of the human genome. Nature. 2001 Feb 15;409(6822):860-921. doi: 10.1038/35057062.
2. Venter JC et al. The sequence of the human genome. Science. 2001 Feb 16;291(5507):1304-51. doi: 10.1126/science.1058040.
3. Jinek M et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012 Aug 17;337(6096):816-21. doi: 10.1126/science.1225829.
4. Percy A. The American history of Rett syndrome. Pediatr Neurol. 2014 Jan;50(1):1-3. doi: 10.1016/j.pediatrneurol.2013.08.018.
5. Amir RE et al. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet. 1999 Oct;23(2):185-8. doi: 10.1038/13810.
6. Pitzianti MB et al. Rett syndrome in males: The different clinical course in two brothers with the same microduplication MECP2 Xq28. Int J Environ Res Public Health. 2019 Aug;16(17):3075. doi: 10.3390/ijerph16173075.
7. Ribeiro MC, MacDonald JL. Sex differences in Mecp2-mutant Rett syndrome model mice and the impact of cellular mosaicism in phenotype development. Brain Res. 2020 Feb 15;1729:146644. doi: 10.1016/j.brainres.2019.146644.
8. Bao X et al. X chromosome inactivation in Rett syndrome and its correlations with MECP2 mutations and phenotype. J Child Neurol. 2008 Jan;23(1):22-5. doi: 10.1177/0883073807307077.
9. Knudsen GPS et al. Increased skewing of X chromosome inactivation in Rett syndrome patients and their mothers. Eur J Hum Genet. 2006 Jul;14(11):1189-94. doi: 10.1038/sj.ejhg.5201682.
10. Chahil G et al. Rett syndrome in males: A case report and review of literature. Cureus. 2018;10(10):e3414. doi: 10.7759/cureus.3414.
11. Reichow B et al. Brief report: Systematic review of Rett syndrome in males. J Autism Dev Disord. 2015 Oct;45(10):3377-83. doi: 10.1007/s10803-015-2519-1.
12. Vashi N, Justice MJ. Treating Rett syndrome: From mouse models to human therapies. Mamm Genome. 2019 Jun;30(5-6):90-110. doi: 10.1007/s00335-019-09793-5.
13. Li CH et al. MeCP2 links heterochromatin condensates and neurodevelopmental disease. Nature. 2020 Oct;586(7829):440-4. doi: 10.1038/s41586-020-2574-4.
14. Schmidt A et al. MeCP2 and chromatin compartmentalization. Cells. 2020 Apr;9(4):878. doi: 10.3390/cells9040878.
15. Wang L et al. Rett syndrome-causing mutations compromise MeCP2-mediated liquid-liquid phase separation of chromatin. Cell Res. 2020 May;30(5):393-407. doi: 10.1038/s41422-020-0288-7.
16. Lin P et al. Transcriptome analysis of human brain tissue identifies reduced expression of complement complex C1Q genes in Rett syndrome. BMC Genomics. 2016;17:427. doi: 10.1186/s12864-016-2746-7.
17. Tudor M et al. Transcriptional profiling of a mouse model for Rett syndrome reveals subtle transcriptional changes in the brain. Proc Natl Acad Sci U S A. 2002 Nov 26;99(24):15536-41. doi: 10.1073/pnas.242566899.
18. Tillotson R et al. Radically truncated MeCP2 rescues Rett syndrome–like neurological defects. Nature. 2017 Oct 19;550(7676):398-401. doi: 10.1038/nature24058.
19. Connolly DR, Zhou Z. Genomic insights into MeCP2 function: A role for the maintenance of chromatin architecture. Curr Opin Neurobiol. 2019 Dec;59:174-9. doi: 10.1016/j.conb.2019.07.002.
20. Johnson BS et al. Biotin tagging of MeCP2 in mice reveals contextual insights into the Rett syndrome transcriptome. Nat Med. 2017 Oct;23(10):1203-14. doi: 10.1038/nm.4406.
21. Gabel HW et al. Disruption of DNA-methylation–dependent long gene repression in Rett syndrome. Nature. 2015 Jun 4;522(7554):89-93. doi: 10.1038/nature14319.
22. Lyst MJ, Bird A. Rett syndrome: A complex disorder with simple roots. Nat Rev Genet. 2015 May;16(5):261-75. doi: 10.1038/nrg3897.
23. Kuehner JN et al. Epigenetic regulations in neuropsychiatric disorders. Front Genet. 2019 Apr 4;10:268. doi: 10.3389/fgene.2019.00268.
24. Pejhan S, Rastegar M. Role of DNA methyl-CpG-binding protein MeCP2 in Rett syndrome pathobiology and mechanism of disease. Biomolecules. 2021 Jan;11(1):75. doi: 10.3390/biom11010075.
25. Fu C et al. Consensus guidelines on managing Rett syndrome across the lifespan. BMJ Paediatr Open. 2020;4(1):e000717. doi: 10.1136/bmjpo-2020-000717.
26. Operto FF et al. Epilepsy and genetic in Rett syndrome: A review. Brain Behav. 2019 May;9(5):e01250. doi: 10.1002/brb3.1250.
27. Nissenkorn A et al. Epilepsy in Rett syndrome – The experience of a National Rett Center. Epilepsia. 2010 Jul;51(7):1252-8. doi: 10.1111/j.1528-1167.2010.02597.x.
28. Welcome to the Rett cure community. Rett Syndrome Research Trust [Internet]. Updated Feb 8, 2022. Accessed Feb 23, 2022. https://reverserett.org.
29. About Rett syndrome. International Rett Syndrome Foundation [Internet]. Updated Jan 4, 2022. Accessed Feb 23, 2022. http://www.rettsyndrome.org.
30. Singh J, Santosh P. Key issues in Rett syndrome: Emotional, behavioural and autonomic dysregulation (EBAD) – A target for clinical trials. Orphanet J Rare Dis. 2018 Jul 31;13(1):128. doi: 10.1186/s13023-018-0873-8.
31. Banerjee A et al. Towards a better diagnosis and treatment of Rett syndrome: A model synaptic disorder. Brain. 2019 Feb 1;142(2):239-48. doi: 10.1093/brain/awy323.
32. Ager S et al. Parental experiences of scoliosis management in Rett syndrome. Disabil Rehabil. 2009 Sep 19;31(23):1917-24. doi: 10.1080/09638280902846392.
33. Budden SS. Management of Rett syndrome: A ten year experience. Neuropediatrics. 1995;26(2):75-7. doi: 10.1055/s-2007-979727.
34. Ip JPK et al. Rett syndrome: Insights into genetic, molecular and circuit mechanisms. Nat Rev Neurosci. 2018 Jun;19(6):368-82. doi: 10.1038/s41583-018-0006-3.
35. Acadia Pharmaceuticals Inc. Study of trofinetide for the treatment of girls and women with Rett syndrome (LAVENDER™). ClinicalTrials.gov identifier: NCT04181723. Updated Feb 17, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT04181723.
36. Acadia Pharmaceuticals announces positive top-line results from the pivotal phase 3 LAVENDER trial of trofinetide in Rett syndrome. Press release. Acadia Pharmaceuticals Inc. Dec 6, 2021. Accessed Feb 23, 2022. https://ir.acadia-pharm.com/news-releases/news-release-details/acadia-pharmaceuticals-announces-positive-top-line-results-1.
37. Copping NA et al. Emerging gene and small molecule therapies for the neurodevelopmental disorder Angelman syndrome. Neurotherapeutics. 2021 Jul;18(3):1535-47. doi: 10.1007/s13311-021-01082-x.
38. Riikonen R. Insulin-like growth factors in the pathogenesis of neurological diseases in children. Int J Mol Sci. 2017 Sep;18(10):2056. doi: 10.3390/ijms18102056.
39. Glaze DG et al; Rett 002 Study Group. Double-blind, randomized, placebo-controlled study of trofinetide in pediatric Rett syndrome. Neurology. 2019 April 16;92(16):e1912-e1925. doi: 10.1212/WNL.0000000000007316.
40. Acadia Pharmaceuticals Inc. An open-label study of trofinetide for the treatment of girls two to five years of age who have Rett syndrome (DAFFODIL™). ClinicalTrials.gov Identifier: NCT04988867. Updated Jan 24, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT04988867.
41. Anavex Life Sciences announces ANAVEX®2-73 meets primary and secondary endpoints in clinical trial. Press release. Anavex Life Sciences Corp. Dec 15, 2020. Accessed Feb 23, 2022. http://www.anavex.com/post/anavex-life-sciences-announces-anavex-2-73-meets-primary-and-secondary-endpoints-in-clinical-trial.
42. Anavex Life Sciences Corp. ANAVEX2-73 study in patients with Rett syndrome (AVATAR). ClinicalTrials.gov Identifier: NCT03941444. Updated Jan 27, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT03941444.
43. Anavex Life Sciences Corp. ANAVEX2-73 study in pediatric patients with Rett syndrome (EXCELLENCE). ClinicalTrials.gov Identifier: NCT04304482. Updated Sep 28, 2021. Accessed Feb 23, 2022. http://www.clinicaltrials.gov/ct2/show/NCT04304482.
44.Christ MG et al. The Sigma-1 receptor at the crossroad of proteostasis, neurodegeneration, and autophagy. Trends Neurosci. 2020 Feb;43(2):79-81. doi: 10.1016/j.tins.2019.12.002.
45. Kaufmann WE et al. ANAVEX®2-73 (blarcamesine), a sigma-1 receptor agonist, ameliorates neurologic impairments in a mouse model of Rett syndrome. Pharmacol Biochem Behav. 2019 Dec;187:172796. doi: 10.1016/j.pbb.2019.172796.
46. Brimson JM et al. Dipentylammonium binds to the sigma-1 receptor and protects against glutamate toxicity, attenuates dopamine toxicity and potentiates neurite outgrowth in various cultured cell lines. Neurotox Res. 2018 Aug;34(2):263-72. doi: 10.1007/s12640-018-9883-5.
47. Kourrich S et al. The sigma-1 receptor: roles in neuronal plasticity and disease. Trends Neurosci. 2012 Dec;35(12):762-71. doi: 10.1016/j.tins.2012.09.007.
48. Lappalainen R, Riikonen RS. High levels of cerebrospinal fluid glutamate in Rett syndrome. Pediatr Neurol. 1996 Oct;15(3):213-6. doi: 10.1016/s0887-8994(96)00218-4.
49. Hamberger A et al. Elevated CSF glutamate in Rett syndrome. Neuropediatrics. 1992;23(4):212-3. doi: 10.1055/s-2008-1071344.
50. Inacio P. FDA acts to support development of potential gene therapy, TSHA-102. Rett Syndrome News [Internet]. Oct 16, 2020. Accessed Feb 23, 2022. https://rettsyndromenews.com/2020/10/16/fda-grants-orphan-drug-rare-pediatric-disease-status-to-tsha-102-potential-rett-gene-therapy.
51. Sinnett SE et al. Engineered microRNA-based regulatory element permits safe high-dose miniMECP2 gene therapy in Rett mice. Brain. 2021 Nov 29;144(10):3005-19. doi: 10.1093/brain/awab182.
52. Le TTH et al. Efficient and precise CRISPR/Cas9-mediated MECP2 modifications in human-induced pluripotent stem cells. Front Genet. 2019 Jul 2;10:625. doi: 10.3389/fgene.2019.00625.
53. Koerner MV et al. Toxicity of overexpressed MeCP2 is independent of HDAC3 activity. Genes Dev. 2018;32(23-24):1514-24. doi: 10.1101/gad.320325.118.
54. Heckman LD et al. Rett-causing mutations reveal two domains critical for MeCP2 function and for toxicity in MECP2 duplication syndrome mice. Elife. 2014;3:e02676. doi: 10.7554/eLife.02676.
55. Neurogene announces new development program in Rett syndrome utilizing novel EXACT technology platform [Internet]. Accessed Aug 12, 2022. https://www.neurogene.com/press-releases/neurogene-announces-new-development-program-in-rett-syndrome-utilizing-novel-exact-technology-platform/
56. Anzalone AV et al. Genome editing with CRISPR-Cas nucleases, base editors, transposases and prime editors. Nat Biotechnol. 2020 Jul;38(7):824-44. doi: 10.1038/s41587-020-0561-9.
57. Gaudelli NM, Komor AC, Rees HA, et al. Programmable base editing of A●T to G●C in genomic DNA without DNA cleavage. Nature. 2017 Nov 23;551(7681):464-71. doi: 10.1038/nature24644.
58. Coenraads M. How RSRT is driving the search for a Rett cure. Rett Syndrome Research Trust [Internet]. Dec 7, 2021. Accessed Feb 23, 2022. https://rettnews.org/articles/how-rsrt-is-driving-the-search-for-a-rett-cure.
59. Cutting-edge technologies to repair the underlying mutations that cause Rett. Rett Syndrome Research Trust [Internet]. Updated Nov 3, 2021. Accessed Feb 23, 2022. https://reverserett.org/research/cures/gene-editing.
60. Sinnamon JR et al. In vivo repair of a protein underlying a neurological disorder by programmable RNA editing. Cell Rep. 2020 Jul 14;32(2):107878. doi: 10.1016/j.celrep.2020.107878.
61. Sinnamon JR et al. Site-directed RNA repair of endogenous Mecp2 RNA in neurons. Proc Natl Acad Sci U S A. 2017 Oct 31;114(44):E9395-E9402. doi: 10.1073/pnas.1715320114.
62. Pipeline. VICO Therapeutics [Internet]. Updated Nov 5, 2021. Accessed Feb 23, 2022. https://vicotx.com/pipeline.
63. Therapeutics platform. Shape Therapeutics [Internet]. Updated Feb 20, 2021. Accessed Feb 23, 2022.
https://live-shapetx.pantheonsite.io/therapeutics-platform.
64. Koeberl DD et al. Glycogen storage disease types I and II: Treatment updates. J Inherit Metab Dis. 2007 Apr;30(2):159-64. doi: 10.1007/s10545-007-0519-9.
The dream of curing genetic disorders has been a persistent but elusive goal, even before the human genome was mapped. Once mapping of the human genome was complete in 2001, an entirely new avenue of potential treatments and cures for genetic diseases and disorders was opened.1,2
The disorders best suited for targeted gene therapy are monogenic; however, tools and delivery methods for editing the human genome were limited and difficult to apply, until the advent of the CRISPR system in 2012.3 CRISPR (an acronym of clustered regularly interspaced short palindromic repeats) has changed the way in which gene therapy strategies are pursued, with dozens of companies leveraging a variety of platforms to create potentially life-changing therapies for devastating rare diseases and disorders.
One of the rare monogenic disorders that is embracing multiple gene therapy approaches is Rett syndrome, a rare, debilitating neurodevelopmental disorder. In this review, we explore the molecular cause of Rett syndrome, disease presentation, current treatments, ongoing clinical trials, and therapies that are on the horizon.
Underlying molecular cause
Rett syndrome is caused by mutations in, or the absence of, the MECP2 gene, which produces methyl-CpG binding protein 2 (MECP2). The syndrome was first described clinically in 1954 by the Austrian physician Andreas Rett; it would take until 1982 before the disorder was officially named, eponymously, in a seminal paper by Hagberg.4 After Hagberg’s characterization, Rett syndrome became the predominant global clinical diagnosis identified among cognitively impaired females, with an incidence of 1 in every 10,000 to 15,000.4
In 1999, mutations in, and deletions of, MECP2 were identified as the cause of Rett syndrome.4,5 MECP2 is located on the X chromosome, in the Xq28 region, making Rett syndrome an X-linked dominant disorder.6 Rett syndrome is seen predominantly in females who are mosaic for mutant or deleted MECP2. Random X chromosome inactivation results in some cells expressing the mutant MECP2 allele and other cells expressing the normal functioning MECP2 allele; the percentage of cells expressing the normal allele correlates with the degree of syndrome severity.7-9
The incidence of Rett syndrome is much lower in males, in whom the syndrome was originally thought to be lethal; many observed male cases are either mosaic or occur in XXY males.10,11
Approximately 95% of cases of Rett syndrome are due to de novo mutations in MECP2, with a handful of specific mutations and large deletions accounting for more than 85% of cases.12 The fact that Rett syndrome is monogenic and most cases are caused by, in total, only a handful of mutations or deletions makes the syndrome a promising candidate for gene therapy.
At the molecular level, it has been observed that the MECP2 mutations of Rett syndrome lead to loss of gene function, thus disrupting the ability of the MECP2 nuclear protein to regulate global gene transcription through its binding to methylated DNA sites.12 A large percentage of these missense and nonsense mutations lead to a truncated or nonfunctional protein.12
One of the ways in which MECP2 regulates transcription is as a component of heterochromatin condensates and by separation of heterochromatin and euchromatin.13-15 It has been observed that the cells of Rett syndrome patients have an altered chromatin state, potentially contributing to transcriptional dysregulation.16,17 Several mutations observed in Rett syndrome patients occur in crucial domains for heterochromatin condensate formation, which helps explain this cellular phenotype.13 Introduction of a engineered “mini” MECP2 in a murine model of Rett syndrome has resulted in partial rescue of heterochromatin condensate formation and transcriptional regulation – fostering the hypothesis that correcting those genetic changes could lead to a potential therapy.18
Beyond the role of MECP2 in heterochromatin condensate formation, the gene interacts with more than 40 proteins that have diverse roles in cellular function, epigenetic modulation, and neuronal development. This volume of interactions contributes to MECP2 being a global gene regulatory protein that has far-reaching effects on transcriptional regulation across the genome.19-22
Epigenetic dysregulation has been associated with neurodevelopmental and neuropsychiatric disorders.23 Both insulin-like growth factor 1 (IGF-1) and brain-derived neurotrophic factor are transcriptional targets of MECP2, and are involved in neuronal differentiation, synaptic function, and neurite outgrowth.12 This helps explain the neurodevelopmental phenotypes observed in MECP2-mutated patients.
Notably, although Rett syndrome patients experience neurodevelopmental phenotypes at the cellular level, neuronal death is not readily observed. That observation provides hope that an interventional therapy after onset of symptoms might still be of benefit.
Presentation
Early neurotypical development. A hallmark of Rett syndrome is neurotypical physical and mental development until 6 to 24 months of age.
Stagnation is the first stage of the syndrome, involving a small but rapid decline in habitual milestones, such motor and language skills.12 Subtle signs, such as microcephaly and hypotonia, can also arise at this time but might be missed.24
Rapid regression follows stagnation. Speech and motor delays and impaired gait and breathing occur;12,25 purposeful hand skills are lost, replaced by repetitive hand-wringing movements that are a hallmark of the syndrome.12,24 Seizures are observed; they become more common during the next stage.12
Plateau. Language advances can be observed, but further deficits are seen in motor skills and hand coordination.12
Late motor deterioration stage. Late physical deficits develop, leading to lifelong impairments. The physical deficits observed are the result of severe muscle weakness, usually resulting in wheelchair dependency.12
Plateau. Patients then reach a second plateau. Regression stops; deficient physical and cognitive states stabilize and are maintained.25
At all stages of Rett syndrome, the following are observed:
- Gastrointestinal problems.
- Sleep disturbances.
- Abnormal cardiorespiratory coupling.
- Greater-than-expected mortality.12
Final regression. The patient is fully dependent for the rest of their lifespan, partially due to seizure activity.26,27
A life-changing diagnosis
A diagnosis of Rett syndrome is life-changing for a patient’s family; access to supportive groups of other patients and their families is extremely beneficial. Two helpful organizations – the Rett Syndrome Research Trust28 and International Rett Syndrome Foundation,29 – offer patient support and community and fund research.
Because X chromosome inactivation is random in Rett syndrome, the individual patient can present with a wide variety of phenotypic combinations – making the patient, and their needs, unique.12 During stages of regression, patients often experience emotional dysregulation and anxiety, which is attributable to their increasing physical difficulties.30 They often exhibit combinations of uncontrolled movements, including repetitive rocking, scratching, and self-injurious behavior.30 For most, regression subsides after the first 5 years of alternating development and regression; after that, their ultimate symptoms persist for life.25
As patients mature, they need to be monitored for proper nutrition and scoliosis.25 As adults, they are at risk of pneumonia, respiratory distress, status epilepticus, osteopenia, and lack of adequate food or water because of impaired ability to feed.25
The lifespan of Rett syndrome patients has increased, thanks to improvements in health care, advances in technology, and early genetic testing, which allows for earlier diagnosis, intervention, and management of symptoms.
Current treatments
When a female patient presents with regression and loss of milestones, sequencing of MECP2 is performed to verify whether Rett syndrome is the cause, by detecting any of the known mutations. Multiplex ligation-dependent probe amplification is also performed to detect major deletions.25
All available treatments for Rett syndrome are symptomatic; intensive early intervention is practiced.31 Multidisciplinary management – medical, psychiatric, and physical – is introduced almost immediately after diagnosis. Following diagnosis, patients are prescribed anti-seizure, sleep, and anxiety medications.31 Electroencephalography can be performed to identify seizure type. Neuromuscular blockage drugs can be prescribed to help with gait and stereotypic hand movements.25
Handguards or splints to the elbows can be prescribed by an occupational therapist to improve hand movement.25 Physical therapy can improve mobility; hydrotherapy and hippotherapy have been successful in helping to maintain mobility and muscle support.32,33 Nutritional management is implemented to control caloric intake and maintain the vitamin D level.31 Some patients experience constipation and urinary retention, putting them at risk of nephrolithiasis.
Once the signs and symptoms of Rett syndrome progress, and milestones regress to a certain point, patients need constant, full-time care for the rest of their lives.34 As symptomatic interventions have greatly improved patient outcomes and it has been shown that about 70% can reach adulthood with a potential lifespan of about 50 years.25
Although there is no cure for Rett syndrome and treatments are symptomatic, ongoing studies – both clinical and preclinical – offer promise that treatments will be developed that work at molecular and genetic levels.
Clinical trials
Advances in understanding of Rett syndrome have led to many therapies in clinical trials, several of which show promise.
Trofinetide. One of the most promising targets for downstream therapy, mentioned earlier, is IGF-1, which was the target of a successful phase 3 clinical trial, LAVENDER (sponsored by Acadia Pharmaceuticals).35,36 This trial studied trofinetide, a synthetic IGF-1 analog that inhibits neuroinflammation, restores glial function, corrects synaptic deficiencies, and regulates oxidative stress response.12,37,38 Initial results from phase 2 and phase 3 trials indicate improved scores for treated patients in the Rett syndrome Behaviour Questionnaire (RSBQ) and Clinical Global Impression–Improvement (CGI-I) scores, while also showing improvements in the Communication and Symbolic Behavior Scales Developmental Profile Infant–Toddler Checklist–Social composite score.36,39
The most common adverse events seen with trofinetide were diarrhea and vomiting.
Acadia Pharmaceuticals has filed for approval of a new drug application for trofinetide with the Food and Drug Administration, for which the company has been granted Fast Track Status and orphan drug designations. Most (95%) subjects in the phase 3 LAVENDER trial elected to continue taking trofinetide in the subsequent open-label Lilac and Lilac-2 extension studies.36 A current open-label phase 2/3 trial is recruiting patients 2 to 5 years of age to evaluate trofinetide.40 It is expected that, in the near future, this could be a drug given to Rett patients as an FDA-approved treatment.
Blarcamesine. Another small molecule drug, blarcamesine (also known as ANAVEX2-73), a sigma-1 receptor agonist, produced promising results in phase 2 clinical trials in adult Rett syndrome patients. The drug is in a phase 2/3 clinical trial for pediatric Rett syndrome patients (sponsored by Anavex Life Sciences).41-43
Phase 2 results indicated statistically significant and clinically meaningful improvement in RSBQ and CGI-I scores with blarcamesine. Improvement was initially observed within 4 weeks after the start of treatment and was sustained throughout the study. The drug was shown to be well tolerated, with minimal adverse effects; no serious adverse events were recorded. These results were observed in adult patients, demonstrating that improvements in Rett syndrome are possible even after regression.
Blarcamesine activates the sigma 1 receptor, which is pivotal to restoring cellular homeostasis and restoring neuroplasticity – deficiencies of which have been linked to autophagy and glutamate toxicity. The drug has also been explored as a potential treatment for other neurological disorders.44-47 Improvements in blarcamesine-treated patients further correlated with lower levels of glutamate in cerebrospinal fluid, which is a Rett syndrome biomarker, supporting the proposition that behavioral improvements were due to drug intervention.48,49 The phase 2 trial was modified into a phase 3 trial and additional endpoints were added.41-43
All patients in the phase 2 adult trial elected to continue in the extension study.
Based on these promising data, Anavex is pursuing an approval pathway for adult patients, while continuing dosage optimization phase 2/3 trials and recruitment for a pediatric trial.42,43
Is the future about gene therapy?
TSHA-102 (miniMECP2). Taysha Gene Therapies is developing a promising gene therapy, TSHA-102, for Rett syndrome, and is aiming to begin phase 1/2 clinical trials in 2022.50 The technology for this therapy relies on the delivery of a fragment of MECP2 (known as miniMECP2), which is regulated by a built-in microRNA regulator (miR-responsive auto-regulatory element, or miRARE) to help ameliorate MECP2 dosage toxicity. (Overexpression of MECP2 is toxic to neurons, which has made traditional [so to speak] gene replacement therapy difficult in Rett syndrome: Levels of MECP2 need to be tightly regulated, and the Taysha microRNA technology regulates levels of miniMECP2, thus reducing toxicity.)
The Taysha microRNA technology has yielded promising results in mouse studies for Rett syndrome; results indicate a lengthening of lifespan and delayed onset of gait abnormalities.51 TSHA-102 is in the preclinical stage but offers promise that it will be the first gene therapy for Rett syndrome to enter clinical trials.
As the field of gene therapy advances, several promising technologies are on the horizon that could potentially have disease-altering impacts on Rett syndrome. These therapies are divided into two broad categories: those at the gene level and those at the transcription and protein level. A few of these approaches are highlighted below.
Gene replacement involves adding a full or partial copy of MECP2 to neuronal cells. This type of therapy presents challenges, from delivery of the new gene to dosage concerns, because MECP2 can be toxic if overexpressed.52-54 Groundbreaking work was done in mouse models involving truncated MECP2, exhibiting phenotypic rescue and validating the gene-replacement approach.18 This strategy is being pursued by Neurogene, which has a uinique technology that allows for tuning of the gene’s expression to get the correct protein levels in the patient. Promising data was presented this year at the American Society of Gene and Cell Therapy conference.55
Early gene replacement therapy studies also laid the foundation for the minMECP2 and microRNA approach being used by Taysha Gene Therapies (discussed above).51
“Correcting” DNA mutations. A different approach at the genetic level involves “correcting” mutations in MECP2 at the DNA level. This is possible because, in a large subset of Rett syndrome patients who have the same missense or nonsense mutations, by using CRISPR, a gene editing tool (discussed above) a single base pair can be corrected.56,57 Previous research, in a Rett syndrome-model of induced pluripotent stem cells, showed that this type of editing is possible – and effective.52 An approach with particular promise involves use of a class of CRISPR proteins known as base editors that are able to specifically alter a single base of DNA.57 The technique has the potential to address many of the mutations seen in Rett syndrome; research on this type of technology is being pursued by Beam Therapeutics, and has the potential to impact Rett syndrome.58
Another promising “correction” approach is exonic editing, in which a much larger section of DNA – potentially, exons 3 and 4, which, taken together, comprise 97% of known MECP2 mutations seen in Rett syndrome – are replaced.59
In both CRISPR and exonic editing therapeutic approaches, endogenous levels of MECP2 expression would be maintained. Of note, both approaches are being pursued for use in treating other genetic disorders, which provides a boost in scaling-up work on addressing safety and efficacy concerns that accompany gene-editing approaches.58 One advantage to the DNA correction approach is that is has the potential to be a “one-and-done” treatment.
“Correcting” RNA. Beyond directly editing DNA, several therapeutic approaches are exploring the ability to edit RNA or to provide the protein directly to cells as enzyme replacement therapy. Such an RNA correction strategy leverages a technology that takes advantage of cells’ natural RNA editor, known as adenosine deaminase acting on RNA (ADAR), which corrects errors in cells’ RNA by providing specific guides to the cell. ADAR can be targeted to fix mutations in the MECP2 RNA transcript, resulting in a “corrected” MECP2 protein.60,61 This technology has delivered promising proof-of-concept evidence in cells and in murine models, and is in the therapeutic pipeline at VICO Therapeutics.62
Shape Therapeutics has also leveraged ADAR to “correct” mutated RNA; Rett syndrome is among the top priorities in the company’s pipeline.
Worth noting is that there are several advantages to the “correction” approach:
- Leveraging internal repair mechanisms minimizes the immune response.
- The flexibility of correction means that it can be used to address many of the mutations that cause Rett syndrome.63
Enzyme replacement therapy, in which the MECP2 protein produced by MECP2 would be directly replaced, is being explored in Rett syndrome patients. This technology has been used successfully in Pompe disease; however, Rett syndrome presents its own challenge because MECP2 needs to be delivered to the brain and neuronal cells.64
Where does this work stand? The technologies described in this section are in preclinical stages of study. Nonetheless, it is expected that several will enter human clinical trials during the next 5 years.
Conclusion
A diagnosis of Rett syndrome is a life-altering event for patients and their family. But there is more hope than ever for effective therapies and, eventually, a cure.
Multiple late-stage clinical trials in progress are demonstrating promising results from therapeutic products, with minimal adverse events. Remarkably, these interventions have delivered improvements to adult patients after regression has stabilized. With rapid progress being made in the field of gene therapy, several technologies for which are focused on Rett syndrome, a hopeful picture is emerging: that therapeutic intervention will be possible before regression, thus effectively treating and, potentially, even curing Rett syndrome.
The landscape is broadening. Add to this hope for approved therapies is the fact that Rett syndrome isn’t the only target being pursued with such strategies; in fact, researchers in the larger field of neurodevelopmental disorder study are working together to find common solutions to shared challenges – from how therapies are designed and delivered to how toxicity is minimized. Much of what is being explored in the Rett syndrome field is also under investigation in other neurodevelopmental syndromes, including Angelman, Prader-Willi, chromosome 15q11.2-13.1 duplication (dup15q), and Fragile X syndrome. This kind of parallel investigation benefits all parties and optimizes a treatment platform so that it can be applied across more than a single disorder.
Like many monogenic disorders, Rett syndrome is entering an exciting stage – at which the words “treatment” and “cure” can be spoken with intent and vision, not just wide-eyed optimism. These words portend real promise for patients who carry the weight of a diagnosis of Rett syndrome, and for their families.
Ms. Ambrose is a student in the master’s of science in human genetics and genomic data analytics program, Keck Graduate Institute, Claremont, Calif. Dr. Bailus is an assistant professor of genetics, Keck Graduate Institute. The authors report no conflict of interest related to this article.
References
1. Lander ES et al; . Initial sequencing and analysis of the human genome. Nature. 2001 Feb 15;409(6822):860-921. doi: 10.1038/35057062.
2. Venter JC et al. The sequence of the human genome. Science. 2001 Feb 16;291(5507):1304-51. doi: 10.1126/science.1058040.
3. Jinek M et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012 Aug 17;337(6096):816-21. doi: 10.1126/science.1225829.
4. Percy A. The American history of Rett syndrome. Pediatr Neurol. 2014 Jan;50(1):1-3. doi: 10.1016/j.pediatrneurol.2013.08.018.
5. Amir RE et al. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet. 1999 Oct;23(2):185-8. doi: 10.1038/13810.
6. Pitzianti MB et al. Rett syndrome in males: The different clinical course in two brothers with the same microduplication MECP2 Xq28. Int J Environ Res Public Health. 2019 Aug;16(17):3075. doi: 10.3390/ijerph16173075.
7. Ribeiro MC, MacDonald JL. Sex differences in Mecp2-mutant Rett syndrome model mice and the impact of cellular mosaicism in phenotype development. Brain Res. 2020 Feb 15;1729:146644. doi: 10.1016/j.brainres.2019.146644.
8. Bao X et al. X chromosome inactivation in Rett syndrome and its correlations with MECP2 mutations and phenotype. J Child Neurol. 2008 Jan;23(1):22-5. doi: 10.1177/0883073807307077.
9. Knudsen GPS et al. Increased skewing of X chromosome inactivation in Rett syndrome patients and their mothers. Eur J Hum Genet. 2006 Jul;14(11):1189-94. doi: 10.1038/sj.ejhg.5201682.
10. Chahil G et al. Rett syndrome in males: A case report and review of literature. Cureus. 2018;10(10):e3414. doi: 10.7759/cureus.3414.
11. Reichow B et al. Brief report: Systematic review of Rett syndrome in males. J Autism Dev Disord. 2015 Oct;45(10):3377-83. doi: 10.1007/s10803-015-2519-1.
12. Vashi N, Justice MJ. Treating Rett syndrome: From mouse models to human therapies. Mamm Genome. 2019 Jun;30(5-6):90-110. doi: 10.1007/s00335-019-09793-5.
13. Li CH et al. MeCP2 links heterochromatin condensates and neurodevelopmental disease. Nature. 2020 Oct;586(7829):440-4. doi: 10.1038/s41586-020-2574-4.
14. Schmidt A et al. MeCP2 and chromatin compartmentalization. Cells. 2020 Apr;9(4):878. doi: 10.3390/cells9040878.
15. Wang L et al. Rett syndrome-causing mutations compromise MeCP2-mediated liquid-liquid phase separation of chromatin. Cell Res. 2020 May;30(5):393-407. doi: 10.1038/s41422-020-0288-7.
16. Lin P et al. Transcriptome analysis of human brain tissue identifies reduced expression of complement complex C1Q genes in Rett syndrome. BMC Genomics. 2016;17:427. doi: 10.1186/s12864-016-2746-7.
17. Tudor M et al. Transcriptional profiling of a mouse model for Rett syndrome reveals subtle transcriptional changes in the brain. Proc Natl Acad Sci U S A. 2002 Nov 26;99(24):15536-41. doi: 10.1073/pnas.242566899.
18. Tillotson R et al. Radically truncated MeCP2 rescues Rett syndrome–like neurological defects. Nature. 2017 Oct 19;550(7676):398-401. doi: 10.1038/nature24058.
19. Connolly DR, Zhou Z. Genomic insights into MeCP2 function: A role for the maintenance of chromatin architecture. Curr Opin Neurobiol. 2019 Dec;59:174-9. doi: 10.1016/j.conb.2019.07.002.
20. Johnson BS et al. Biotin tagging of MeCP2 in mice reveals contextual insights into the Rett syndrome transcriptome. Nat Med. 2017 Oct;23(10):1203-14. doi: 10.1038/nm.4406.
21. Gabel HW et al. Disruption of DNA-methylation–dependent long gene repression in Rett syndrome. Nature. 2015 Jun 4;522(7554):89-93. doi: 10.1038/nature14319.
22. Lyst MJ, Bird A. Rett syndrome: A complex disorder with simple roots. Nat Rev Genet. 2015 May;16(5):261-75. doi: 10.1038/nrg3897.
23. Kuehner JN et al. Epigenetic regulations in neuropsychiatric disorders. Front Genet. 2019 Apr 4;10:268. doi: 10.3389/fgene.2019.00268.
24. Pejhan S, Rastegar M. Role of DNA methyl-CpG-binding protein MeCP2 in Rett syndrome pathobiology and mechanism of disease. Biomolecules. 2021 Jan;11(1):75. doi: 10.3390/biom11010075.
25. Fu C et al. Consensus guidelines on managing Rett syndrome across the lifespan. BMJ Paediatr Open. 2020;4(1):e000717. doi: 10.1136/bmjpo-2020-000717.
26. Operto FF et al. Epilepsy and genetic in Rett syndrome: A review. Brain Behav. 2019 May;9(5):e01250. doi: 10.1002/brb3.1250.
27. Nissenkorn A et al. Epilepsy in Rett syndrome – The experience of a National Rett Center. Epilepsia. 2010 Jul;51(7):1252-8. doi: 10.1111/j.1528-1167.2010.02597.x.
28. Welcome to the Rett cure community. Rett Syndrome Research Trust [Internet]. Updated Feb 8, 2022. Accessed Feb 23, 2022. https://reverserett.org.
29. About Rett syndrome. International Rett Syndrome Foundation [Internet]. Updated Jan 4, 2022. Accessed Feb 23, 2022. http://www.rettsyndrome.org.
30. Singh J, Santosh P. Key issues in Rett syndrome: Emotional, behavioural and autonomic dysregulation (EBAD) – A target for clinical trials. Orphanet J Rare Dis. 2018 Jul 31;13(1):128. doi: 10.1186/s13023-018-0873-8.
31. Banerjee A et al. Towards a better diagnosis and treatment of Rett syndrome: A model synaptic disorder. Brain. 2019 Feb 1;142(2):239-48. doi: 10.1093/brain/awy323.
32. Ager S et al. Parental experiences of scoliosis management in Rett syndrome. Disabil Rehabil. 2009 Sep 19;31(23):1917-24. doi: 10.1080/09638280902846392.
33. Budden SS. Management of Rett syndrome: A ten year experience. Neuropediatrics. 1995;26(2):75-7. doi: 10.1055/s-2007-979727.
34. Ip JPK et al. Rett syndrome: Insights into genetic, molecular and circuit mechanisms. Nat Rev Neurosci. 2018 Jun;19(6):368-82. doi: 10.1038/s41583-018-0006-3.
35. Acadia Pharmaceuticals Inc. Study of trofinetide for the treatment of girls and women with Rett syndrome (LAVENDER™). ClinicalTrials.gov identifier: NCT04181723. Updated Feb 17, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT04181723.
36. Acadia Pharmaceuticals announces positive top-line results from the pivotal phase 3 LAVENDER trial of trofinetide in Rett syndrome. Press release. Acadia Pharmaceuticals Inc. Dec 6, 2021. Accessed Feb 23, 2022. https://ir.acadia-pharm.com/news-releases/news-release-details/acadia-pharmaceuticals-announces-positive-top-line-results-1.
37. Copping NA et al. Emerging gene and small molecule therapies for the neurodevelopmental disorder Angelman syndrome. Neurotherapeutics. 2021 Jul;18(3):1535-47. doi: 10.1007/s13311-021-01082-x.
38. Riikonen R. Insulin-like growth factors in the pathogenesis of neurological diseases in children. Int J Mol Sci. 2017 Sep;18(10):2056. doi: 10.3390/ijms18102056.
39. Glaze DG et al; Rett 002 Study Group. Double-blind, randomized, placebo-controlled study of trofinetide in pediatric Rett syndrome. Neurology. 2019 April 16;92(16):e1912-e1925. doi: 10.1212/WNL.0000000000007316.
40. Acadia Pharmaceuticals Inc. An open-label study of trofinetide for the treatment of girls two to five years of age who have Rett syndrome (DAFFODIL™). ClinicalTrials.gov Identifier: NCT04988867. Updated Jan 24, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT04988867.
41. Anavex Life Sciences announces ANAVEX®2-73 meets primary and secondary endpoints in clinical trial. Press release. Anavex Life Sciences Corp. Dec 15, 2020. Accessed Feb 23, 2022. http://www.anavex.com/post/anavex-life-sciences-announces-anavex-2-73-meets-primary-and-secondary-endpoints-in-clinical-trial.
42. Anavex Life Sciences Corp. ANAVEX2-73 study in patients with Rett syndrome (AVATAR). ClinicalTrials.gov Identifier: NCT03941444. Updated Jan 27, 2022. Accessed Feb 23, 2022. https://clinicaltrials.gov/ct2/show/NCT03941444.
43. Anavex Life Sciences Corp. ANAVEX2-73 study in pediatric patients with Rett syndrome (EXCELLENCE). ClinicalTrials.gov Identifier: NCT04304482. Updated Sep 28, 2021. Accessed Feb 23, 2022. http://www.clinicaltrials.gov/ct2/show/NCT04304482.
44.Christ MG et al. The Sigma-1 receptor at the crossroad of proteostasis, neurodegeneration, and autophagy. Trends Neurosci. 2020 Feb;43(2):79-81. doi: 10.1016/j.tins.2019.12.002.
45. Kaufmann WE et al. ANAVEX®2-73 (blarcamesine), a sigma-1 receptor agonist, ameliorates neurologic impairments in a mouse model of Rett syndrome. Pharmacol Biochem Behav. 2019 Dec;187:172796. doi: 10.1016/j.pbb.2019.172796.
46. Brimson JM et al. Dipentylammonium binds to the sigma-1 receptor and protects against glutamate toxicity, attenuates dopamine toxicity and potentiates neurite outgrowth in various cultured cell lines. Neurotox Res. 2018 Aug;34(2):263-72. doi: 10.1007/s12640-018-9883-5.
47. Kourrich S et al. The sigma-1 receptor: roles in neuronal plasticity and disease. Trends Neurosci. 2012 Dec;35(12):762-71. doi: 10.1016/j.tins.2012.09.007.
48. Lappalainen R, Riikonen RS. High levels of cerebrospinal fluid glutamate in Rett syndrome. Pediatr Neurol. 1996 Oct;15(3):213-6. doi: 10.1016/s0887-8994(96)00218-4.
49. Hamberger A et al. Elevated CSF glutamate in Rett syndrome. Neuropediatrics. 1992;23(4):212-3. doi: 10.1055/s-2008-1071344.
50. Inacio P. FDA acts to support development of potential gene therapy, TSHA-102. Rett Syndrome News [Internet]. Oct 16, 2020. Accessed Feb 23, 2022. https://rettsyndromenews.com/2020/10/16/fda-grants-orphan-drug-rare-pediatric-disease-status-to-tsha-102-potential-rett-gene-therapy.
51. Sinnett SE et al. Engineered microRNA-based regulatory element permits safe high-dose miniMECP2 gene therapy in Rett mice. Brain. 2021 Nov 29;144(10):3005-19. doi: 10.1093/brain/awab182.
52. Le TTH et al. Efficient and precise CRISPR/Cas9-mediated MECP2 modifications in human-induced pluripotent stem cells. Front Genet. 2019 Jul 2;10:625. doi: 10.3389/fgene.2019.00625.
53. Koerner MV et al. Toxicity of overexpressed MeCP2 is independent of HDAC3 activity. Genes Dev. 2018;32(23-24):1514-24. doi: 10.1101/gad.320325.118.
54. Heckman LD et al. Rett-causing mutations reveal two domains critical for MeCP2 function and for toxicity in MECP2 duplication syndrome mice. Elife. 2014;3:e02676. doi: 10.7554/eLife.02676.
55. Neurogene announces new development program in Rett syndrome utilizing novel EXACT technology platform [Internet]. Accessed Aug 12, 2022. https://www.neurogene.com/press-releases/neurogene-announces-new-development-program-in-rett-syndrome-utilizing-novel-exact-technology-platform/
56. Anzalone AV et al. Genome editing with CRISPR-Cas nucleases, base editors, transposases and prime editors. Nat Biotechnol. 2020 Jul;38(7):824-44. doi: 10.1038/s41587-020-0561-9.
57. Gaudelli NM, Komor AC, Rees HA, et al. Programmable base editing of A●T to G●C in genomic DNA without DNA cleavage. Nature. 2017 Nov 23;551(7681):464-71. doi: 10.1038/nature24644.
58. Coenraads M. How RSRT is driving the search for a Rett cure. Rett Syndrome Research Trust [Internet]. Dec 7, 2021. Accessed Feb 23, 2022. https://rettnews.org/articles/how-rsrt-is-driving-the-search-for-a-rett-cure.
59. Cutting-edge technologies to repair the underlying mutations that cause Rett. Rett Syndrome Research Trust [Internet]. Updated Nov 3, 2021. Accessed Feb 23, 2022. https://reverserett.org/research/cures/gene-editing.
60. Sinnamon JR et al. In vivo repair of a protein underlying a neurological disorder by programmable RNA editing. Cell Rep. 2020 Jul 14;32(2):107878. doi: 10.1016/j.celrep.2020.107878.
61. Sinnamon JR et al. Site-directed RNA repair of endogenous Mecp2 RNA in neurons. Proc Natl Acad Sci U S A. 2017 Oct 31;114(44):E9395-E9402. doi: 10.1073/pnas.1715320114.
62. Pipeline. VICO Therapeutics [Internet]. Updated Nov 5, 2021. Accessed Feb 23, 2022. https://vicotx.com/pipeline.
63. Therapeutics platform. Shape Therapeutics [Internet]. Updated Feb 20, 2021. Accessed Feb 23, 2022.
https://live-shapetx.pantheonsite.io/therapeutics-platform.
64. Koeberl DD et al. Glycogen storage disease types I and II: Treatment updates. J Inherit Metab Dis. 2007 Apr;30(2):159-64. doi: 10.1007/s10545-007-0519-9.