User login
Study tracks distant metastatic patterns of Merkel cell carcinoma
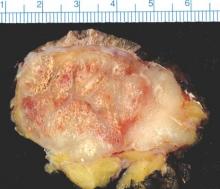
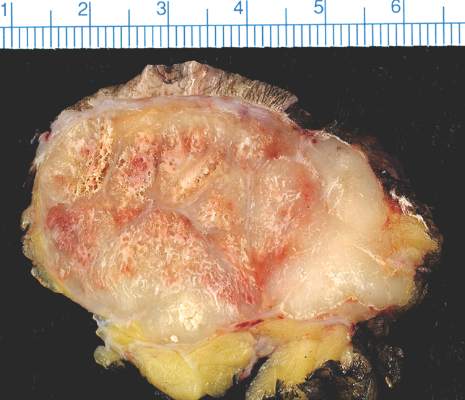
SCOTTSDALE, ARIZ. – Distant metastatic sites of Merkel cell carcinoma most often involved the supraclavicular, retroperitoneal, and iliac lymph nodes, in a single center retrospective study of 305 patients.
Merkel cell carcinoma metastases “clearly favored distant nodes, but the distribution of other metastatic sites was distinct from other cancers, such as melanoma,” Jamiluddin Qazi, an undergraduate student at the University of Washington, Seattle, said at the annual meeting of the Society for Investigative Dermatology. The findings could help guide imaging and other surveillance of patients after they develop a primary Merkel cell tumor, he added.
About 2,000 individuals in the United States are diagnosed with Merkel cell carcinoma every year. About 40% of these patients develop metastatic disease, which has a 5-year survival rate of less than 25%, Mr. Qazi noted. Indeed, median survival after diagnosis of metastatic Merkel cell carcinoma was only 9.5 months in one recent study (J Cutan Pathol. 2010;37:20-7). Programmed death 1 (PD-1) blockade with pembrolizumab (Keytruda) can potentially improve survival (N Engl J Med. 2016; 374:2542-52), “but there is no consensus regarding follow-up for Merkel cell carcinoma. The 2016 National Comprehensive Cancer Network (NCCN) guidelines recommend ‘follow up as clinically indicated,’ and a lack of data has led to ambiguity,” Mr. Qazi said.
Working with oncologists and radiologists at the Seattle Cancer Care Alliance, he analyzed a tissue and clinical database of 442 initial distant Merkel cell carcinoma metastases among 305 patients. Initial distant metastases were defined as the first lesions detected beyond the regional lymph nodes of the primary tumor. A total of 69% of patients had one initial distant metastasis, 19% had two concurrently identified lesions, 9% had three lesions, and 4% had at least four lesions, Mr. Qazi reported.
“Merkel cell carcinoma seemed to metastasize to unusual places, but clearly preferred the distant lymph nodes. In all, 26% of metastases localized there, most commonly to the supraclavicular, retroperitoneal, and iliac nodes,” he said. The next most common site of distant metastasis was the liver (15% of lesions), followed by the skin and bone (13% of lesions each), lung (6%), and pancreas (5%). Less common sites included the heart, spleen, abdominal muscle, brain, kidneys, adrenal glands, gonad, chest wall, and stomach.
Comparing these findings with a similar study in melanoma (J Oncol 2012. doi: 10.1155/2012/647684) showed that both cancers have about the same chances of metastasizing to the liver, bone, kidneys, adrenal glands, and stomach, Mr. Qazi said. However, Merkel cell carcinoma was less likely to metastasize to the brain (3% of lesions, vs. 12% for melanoma) and lung (6% vs. 14%), and was more likely to metastasize to the pancreas (5% vs. 1%).
Now the investigators are working to link metastatic sites with factors such as the location of the primary tumor, the presence or absence of lymphovascular invasion, and the status of the immune system and Merkel polyomavirus infection, said Mr. Qazi. They also are analyzing time from diagnosis or treatment to metastasis to help guide decisions about when to order follow-up imaging. Ultimately, they hope to create an online tool that enables clinicians to describe a primary Merkel cell carcinoma and rapidly receive automated information about the most likely timing and location of metastasis.
The National Institutes of Health supported the study. Mr. Qazi had no conflicts of interest.
SCOTTSDALE, ARIZ. – Distant metastatic sites of Merkel cell carcinoma most often involved the supraclavicular, retroperitoneal, and iliac lymph nodes, in a single center retrospective study of 305 patients.
Merkel cell carcinoma metastases “clearly favored distant nodes, but the distribution of other metastatic sites was distinct from other cancers, such as melanoma,” Jamiluddin Qazi, an undergraduate student at the University of Washington, Seattle, said at the annual meeting of the Society for Investigative Dermatology. The findings could help guide imaging and other surveillance of patients after they develop a primary Merkel cell tumor, he added.
About 2,000 individuals in the United States are diagnosed with Merkel cell carcinoma every year. About 40% of these patients develop metastatic disease, which has a 5-year survival rate of less than 25%, Mr. Qazi noted. Indeed, median survival after diagnosis of metastatic Merkel cell carcinoma was only 9.5 months in one recent study (J Cutan Pathol. 2010;37:20-7). Programmed death 1 (PD-1) blockade with pembrolizumab (Keytruda) can potentially improve survival (N Engl J Med. 2016; 374:2542-52), “but there is no consensus regarding follow-up for Merkel cell carcinoma. The 2016 National Comprehensive Cancer Network (NCCN) guidelines recommend ‘follow up as clinically indicated,’ and a lack of data has led to ambiguity,” Mr. Qazi said.
Working with oncologists and radiologists at the Seattle Cancer Care Alliance, he analyzed a tissue and clinical database of 442 initial distant Merkel cell carcinoma metastases among 305 patients. Initial distant metastases were defined as the first lesions detected beyond the regional lymph nodes of the primary tumor. A total of 69% of patients had one initial distant metastasis, 19% had two concurrently identified lesions, 9% had three lesions, and 4% had at least four lesions, Mr. Qazi reported.
“Merkel cell carcinoma seemed to metastasize to unusual places, but clearly preferred the distant lymph nodes. In all, 26% of metastases localized there, most commonly to the supraclavicular, retroperitoneal, and iliac nodes,” he said. The next most common site of distant metastasis was the liver (15% of lesions), followed by the skin and bone (13% of lesions each), lung (6%), and pancreas (5%). Less common sites included the heart, spleen, abdominal muscle, brain, kidneys, adrenal glands, gonad, chest wall, and stomach.
Comparing these findings with a similar study in melanoma (J Oncol 2012. doi: 10.1155/2012/647684) showed that both cancers have about the same chances of metastasizing to the liver, bone, kidneys, adrenal glands, and stomach, Mr. Qazi said. However, Merkel cell carcinoma was less likely to metastasize to the brain (3% of lesions, vs. 12% for melanoma) and lung (6% vs. 14%), and was more likely to metastasize to the pancreas (5% vs. 1%).
Now the investigators are working to link metastatic sites with factors such as the location of the primary tumor, the presence or absence of lymphovascular invasion, and the status of the immune system and Merkel polyomavirus infection, said Mr. Qazi. They also are analyzing time from diagnosis or treatment to metastasis to help guide decisions about when to order follow-up imaging. Ultimately, they hope to create an online tool that enables clinicians to describe a primary Merkel cell carcinoma and rapidly receive automated information about the most likely timing and location of metastasis.
The National Institutes of Health supported the study. Mr. Qazi had no conflicts of interest.
SCOTTSDALE, ARIZ. – Distant metastatic sites of Merkel cell carcinoma most often involved the supraclavicular, retroperitoneal, and iliac lymph nodes, in a single center retrospective study of 305 patients.
Merkel cell carcinoma metastases “clearly favored distant nodes, but the distribution of other metastatic sites was distinct from other cancers, such as melanoma,” Jamiluddin Qazi, an undergraduate student at the University of Washington, Seattle, said at the annual meeting of the Society for Investigative Dermatology. The findings could help guide imaging and other surveillance of patients after they develop a primary Merkel cell tumor, he added.
About 2,000 individuals in the United States are diagnosed with Merkel cell carcinoma every year. About 40% of these patients develop metastatic disease, which has a 5-year survival rate of less than 25%, Mr. Qazi noted. Indeed, median survival after diagnosis of metastatic Merkel cell carcinoma was only 9.5 months in one recent study (J Cutan Pathol. 2010;37:20-7). Programmed death 1 (PD-1) blockade with pembrolizumab (Keytruda) can potentially improve survival (N Engl J Med. 2016; 374:2542-52), “but there is no consensus regarding follow-up for Merkel cell carcinoma. The 2016 National Comprehensive Cancer Network (NCCN) guidelines recommend ‘follow up as clinically indicated,’ and a lack of data has led to ambiguity,” Mr. Qazi said.
Working with oncologists and radiologists at the Seattle Cancer Care Alliance, he analyzed a tissue and clinical database of 442 initial distant Merkel cell carcinoma metastases among 305 patients. Initial distant metastases were defined as the first lesions detected beyond the regional lymph nodes of the primary tumor. A total of 69% of patients had one initial distant metastasis, 19% had two concurrently identified lesions, 9% had three lesions, and 4% had at least four lesions, Mr. Qazi reported.
“Merkel cell carcinoma seemed to metastasize to unusual places, but clearly preferred the distant lymph nodes. In all, 26% of metastases localized there, most commonly to the supraclavicular, retroperitoneal, and iliac nodes,” he said. The next most common site of distant metastasis was the liver (15% of lesions), followed by the skin and bone (13% of lesions each), lung (6%), and pancreas (5%). Less common sites included the heart, spleen, abdominal muscle, brain, kidneys, adrenal glands, gonad, chest wall, and stomach.
Comparing these findings with a similar study in melanoma (J Oncol 2012. doi: 10.1155/2012/647684) showed that both cancers have about the same chances of metastasizing to the liver, bone, kidneys, adrenal glands, and stomach, Mr. Qazi said. However, Merkel cell carcinoma was less likely to metastasize to the brain (3% of lesions, vs. 12% for melanoma) and lung (6% vs. 14%), and was more likely to metastasize to the pancreas (5% vs. 1%).
Now the investigators are working to link metastatic sites with factors such as the location of the primary tumor, the presence or absence of lymphovascular invasion, and the status of the immune system and Merkel polyomavirus infection, said Mr. Qazi. They also are analyzing time from diagnosis or treatment to metastasis to help guide decisions about when to order follow-up imaging. Ultimately, they hope to create an online tool that enables clinicians to describe a primary Merkel cell carcinoma and rapidly receive automated information about the most likely timing and location of metastasis.
The National Institutes of Health supported the study. Mr. Qazi had no conflicts of interest.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: Distant metastases of Merkel cell carcinoma most often involve the lymph nodes, followed by the liver, skin, and bone.
Major finding: Distant lymph node metastases comprised 26% of lesions, the liver comprised 15%, and skin and bone made up 13% each.
Data source: A single-center retrospective study of 442 initial distant metastases of Merkel cell carcinoma among 305 patients.
Disclosures: The National Institutes of Health supported the study. Mr. Qazi had no conflicts of interest.
US Completes "Largest Takedown" of Federal Health Insurance Fraud
WASHINGTON - The U.S. Justice Department said Wednesday that federal law enforcement officials have hit a milestone in 2016 by completing the "largest takedown ever" against defendants allegedly trying to defraud Medicare and other federal insurance programs.
The 2016 takedown involves 301 defendants and a loss amount of $900 million, the department said. That exceeds a record last year, when 243 defendants faced charges in a combined $712 million in losses.
Among the defendants charged in the takedown include two owners of a group of outpatient clinics and a patient recruiter who stand accused of filing $36 million in fraudulent claims for physical therapy and other services that were not medically necessary.
To find patients, the Justice Department alleges the clinic operators and the recruiter targeted poor drug addicts and offered them narcotics so they could bill them for services that were never provided.
Another case that was highlighted on Wednesday involved home health fraud. In that case, a doctor was indicted for billing $38 million for home health services that were not needed or ever provided.
The Justice Department said that about 50 percent of the cases in the 2016 take down involve some form of home health fraud, and about 25 percent involve pharmacy fraud.
WASHINGTON - The U.S. Justice Department said Wednesday that federal law enforcement officials have hit a milestone in 2016 by completing the "largest takedown ever" against defendants allegedly trying to defraud Medicare and other federal insurance programs.
The 2016 takedown involves 301 defendants and a loss amount of $900 million, the department said. That exceeds a record last year, when 243 defendants faced charges in a combined $712 million in losses.
Among the defendants charged in the takedown include two owners of a group of outpatient clinics and a patient recruiter who stand accused of filing $36 million in fraudulent claims for physical therapy and other services that were not medically necessary.
To find patients, the Justice Department alleges the clinic operators and the recruiter targeted poor drug addicts and offered them narcotics so they could bill them for services that were never provided.
Another case that was highlighted on Wednesday involved home health fraud. In that case, a doctor was indicted for billing $38 million for home health services that were not needed or ever provided.
The Justice Department said that about 50 percent of the cases in the 2016 take down involve some form of home health fraud, and about 25 percent involve pharmacy fraud.
WASHINGTON - The U.S. Justice Department said Wednesday that federal law enforcement officials have hit a milestone in 2016 by completing the "largest takedown ever" against defendants allegedly trying to defraud Medicare and other federal insurance programs.
The 2016 takedown involves 301 defendants and a loss amount of $900 million, the department said. That exceeds a record last year, when 243 defendants faced charges in a combined $712 million in losses.
Among the defendants charged in the takedown include two owners of a group of outpatient clinics and a patient recruiter who stand accused of filing $36 million in fraudulent claims for physical therapy and other services that were not medically necessary.
To find patients, the Justice Department alleges the clinic operators and the recruiter targeted poor drug addicts and offered them narcotics so they could bill them for services that were never provided.
Another case that was highlighted on Wednesday involved home health fraud. In that case, a doctor was indicted for billing $38 million for home health services that were not needed or ever provided.
The Justice Department said that about 50 percent of the cases in the 2016 take down involve some form of home health fraud, and about 25 percent involve pharmacy fraud.
Nonwhite Race, Lower Socioeconomic Status Predicts Persistently Active AD
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
AT THE 2016 SID ANNUAL MEETING
Nonwhite race, lower socioeconomic status predicts persistently active AD
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: Persistently active atopic dermatitis is associated with nonwhite race, annual household income under $50,000, female sex, and history of atopy.
Major finding: Nonwhite race and history of atopy each lowered the odds of complete disease control by about 43% (odds ratios, 0.53; P less than .05).
Data source: A longitudinal cohort study of 6,237 patients aged 2-26 years from the Pediatric Eczema Elective Registry (PEER).
Disclosures: Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
Nonwhite race, lower socioeconomic status predicts persistently active AD
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
SCOTTSDALE, ARIZ. –Among patients with atopic dermatitis, persistently active disease was significantly more common among females of nonwhite race with a history of atopy than among patients without these characteristics, in an analysis of survey data from the Pediatric Elective Eczema Registry.
Annual household income under $50,000 also was a significant predictor of persistently active eczema, according to Katrina Abuabara, MD, of the department of dermatology, University of California, San Francisco, and her associates, who reported their results in a poster at the annual meeting of the Society for Investigative Dermatology.
Atopic dermatitis often persists into adulthood, but few studies have explored contributors to poor disease control. To help fill that gap, the investigators analyzed 65,237 surveys from the Pediatric Eczema Elective Registry (PEER), which tracks children and young adults aged 2-26 years with physician-diagnosed atopic dermatitis. The average age of the 6,237 patients was 7 years at enrollment (standard deviation, 4 years). They were followed at 6-month intervals for up to 10 years, with an average of about 10 surveys per respondent (standard deviation, 6.3 surveys).
In all, 4,607 patients (74% of the cohort) returned surveys spanning early childhood through their mid-20s. Only 15% of patients had “resolving” disease, meaning that as they aged, they increasingly reported complete disease control for periods of 6 months and longer.
The remaining 85% of patients had persistently active disease. In this group, 54% were female, 77% had a household income under $50,000 per year, 71% were nonwhite, and 75% had a history of atopy. Each of these characteristics significantly increased the odds of persistently active atopic dermatitis in the multivariable model (P less than .05 for each association).
Nonwhite race and history of atopy were the strongest predictors of persistently active disease – each lowered the odds of complete disease control by almost 50% (odds ratio, 0.53). Furthermore, females had 37% lower odds of complete disease control compared with males (OR, 0.63), and individuals with household income under $50,000 had 16% lower odds of complete disease control compared with those with higher annual incomes (OR, 0.84).
The link between lower socioeconomic status and persistently active eczema belies previous findings, the researchers noted. Those studies found that individuals of higher socioeconomic status were at greater risk for developing atopic dermatitis, but “failed to account for the chronic nature of the disease. In contrast, our results suggest that atopic dermatitis persistence may be associated with lower income and nonwhite race, and highlight the importance of longitudinal studies that permit analysis of mechanisms of disease control over time.”
Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: Persistently active atopic dermatitis is associated with nonwhite race, annual household income under $50,000, female sex, and history of atopy.
Major finding: Nonwhite race and history of atopy each lowered the odds of complete disease control by about 43% (odds ratios, 0.53; P less than .05).
Data source: A longitudinal cohort study of 6,237 patients aged 2-26 years from the Pediatric Eczema Elective Registry (PEER).
Disclosures: Dr. Abuabara received a grant from the Clinical & Translational Science Institute of UCSF. She had no disclosures.
Cluster Seizures Are Shorter Than Isolated Seizures
To determine the unique characteristics of cluster seizures, investigators looked at 92 subjects, 83% of whom had at least one seizure cluster. They found that seizures occurring within a cluster were significantly shorter than the last seizure that occurred in the cluster. They also discovered that the terminal seizure in a cluster is similar to duration in isolated seizures that are not part of a cluster. Finally, the researchers found that cluster seizures are more likely to occur in the frontal and temporal lobes.
Ferastraoaru V, Schulze-Bonhage A, Lipton RB, Dumpelmann M, Legatt AD, Hunt SR. Termination of seizure clusters is related to duration of focal seizures. Epilepsia. 2016;57(6):889-895.
To determine the unique characteristics of cluster seizures, investigators looked at 92 subjects, 83% of whom had at least one seizure cluster. They found that seizures occurring within a cluster were significantly shorter than the last seizure that occurred in the cluster. They also discovered that the terminal seizure in a cluster is similar to duration in isolated seizures that are not part of a cluster. Finally, the researchers found that cluster seizures are more likely to occur in the frontal and temporal lobes.
Ferastraoaru V, Schulze-Bonhage A, Lipton RB, Dumpelmann M, Legatt AD, Hunt SR. Termination of seizure clusters is related to duration of focal seizures. Epilepsia. 2016;57(6):889-895.
To determine the unique characteristics of cluster seizures, investigators looked at 92 subjects, 83% of whom had at least one seizure cluster. They found that seizures occurring within a cluster were significantly shorter than the last seizure that occurred in the cluster. They also discovered that the terminal seizure in a cluster is similar to duration in isolated seizures that are not part of a cluster. Finally, the researchers found that cluster seizures are more likely to occur in the frontal and temporal lobes.
Ferastraoaru V, Schulze-Bonhage A, Lipton RB, Dumpelmann M, Legatt AD, Hunt SR. Termination of seizure clusters is related to duration of focal seizures. Epilepsia. 2016;57(6):889-895.
Gene Variants Linked to Posttraumatic Seizures
To determine if genetic variation influences the susceptibility to traumatic brain injury and the subsequent posttraumatic seizures, Anne Ritter and her associates from the University of Pittsburgh analyzed the relationship between posttraumatic seizures and single nucleotide polymorphisms (SNPs). Thirty two SNPs were evaluated within SLC1A1 and SLC1A6, which are protein coding genes for glutamate transporters. (Glutamate transporters control glutamate levels and excitatory neurotransmission and have been associated with traumatic brain injury.) The analysis found that among 253 individuals, 49 had experienced posttraumatic seizures. Within this smaller group, they found genotypes at SNP rs10974620 (SLC1A1) linked to the time to the first seizure during a three year follow-up. And after factoring in several confounding variables, rs10974620 remained statistically significant (P = .017).
Rittner AC, Kammerer CM, Brooks MM, Conley YP, Wagner AM. Genetic variation in neuronal glutamate transport genes and associations with posttraumatic seizure. Epilepsia. 2016;57(6):984-993.
To determine if genetic variation influences the susceptibility to traumatic brain injury and the subsequent posttraumatic seizures, Anne Ritter and her associates from the University of Pittsburgh analyzed the relationship between posttraumatic seizures and single nucleotide polymorphisms (SNPs). Thirty two SNPs were evaluated within SLC1A1 and SLC1A6, which are protein coding genes for glutamate transporters. (Glutamate transporters control glutamate levels and excitatory neurotransmission and have been associated with traumatic brain injury.) The analysis found that among 253 individuals, 49 had experienced posttraumatic seizures. Within this smaller group, they found genotypes at SNP rs10974620 (SLC1A1) linked to the time to the first seizure during a three year follow-up. And after factoring in several confounding variables, rs10974620 remained statistically significant (P = .017).
Rittner AC, Kammerer CM, Brooks MM, Conley YP, Wagner AM. Genetic variation in neuronal glutamate transport genes and associations with posttraumatic seizure. Epilepsia. 2016;57(6):984-993.
To determine if genetic variation influences the susceptibility to traumatic brain injury and the subsequent posttraumatic seizures, Anne Ritter and her associates from the University of Pittsburgh analyzed the relationship between posttraumatic seizures and single nucleotide polymorphisms (SNPs). Thirty two SNPs were evaluated within SLC1A1 and SLC1A6, which are protein coding genes for glutamate transporters. (Glutamate transporters control glutamate levels and excitatory neurotransmission and have been associated with traumatic brain injury.) The analysis found that among 253 individuals, 49 had experienced posttraumatic seizures. Within this smaller group, they found genotypes at SNP rs10974620 (SLC1A1) linked to the time to the first seizure during a three year follow-up. And after factoring in several confounding variables, rs10974620 remained statistically significant (P = .017).
Rittner AC, Kammerer CM, Brooks MM, Conley YP, Wagner AM. Genetic variation in neuronal glutamate transport genes and associations with posttraumatic seizure. Epilepsia. 2016;57(6):984-993.
A Patient of Child-Bearing Age
Nikesh Ardeshna, MD
Dr. Ardeshna is the Medical Director of Adult Epilepsy Services at Royal Oak Hospital, Beaumont Health System, in Royal Oak, Michigan.
A 20-year-old female with a history of primary generalized epilepsy presents for establishment of care, as the patient’s insurance had changed. She is currently taking levetiracetam (Keppra) 1500 mg twice daily and divalproex (Depakote) 500 mg once daily by mouth at bedtime. The patient’s last breakthrough seizure was 10 months ago, at which time divalproex was added. The patient has a history of anxiety and depression, for which she is taking escitalopram (Lexapro) 10 mg once daily. The patient also has a history of “sporadic” headaches. The patient used to be on oral contraceptives, but about 3 months ago an intrauterine device was placed.
Questions:
Given the patient’s gender and age, is this the ideal antiepileptic drug (AED) regimen for this patient?
Items to consider: The patient is a young female of child-bearing age. Divalproex has been shown to be among the least safe AEDs during pregnancy due to a higher risk for birth defects and cognitive side effects on the child.
Ideally, an AED other than divalproex should be chosen. Divalproex also has numerous other side effects.
Polytherapy and higher doses of AEDs can increase the risk of teratogenic side effects.
Levetiracetam is considered to be a relatively safe AED for pregnancy.
The goal should be to simplify the patient’s AED regimen to monotherapy and minimize the dose.
Nikesh Ardeshna, MD
Dr. Ardeshna is the Medical Director of Adult Epilepsy Services at Royal Oak Hospital, Beaumont Health System, in Royal Oak, Michigan.
A 20-year-old female with a history of primary generalized epilepsy presents for establishment of care, as the patient’s insurance had changed. She is currently taking levetiracetam (Keppra) 1500 mg twice daily and divalproex (Depakote) 500 mg once daily by mouth at bedtime. The patient’s last breakthrough seizure was 10 months ago, at which time divalproex was added. The patient has a history of anxiety and depression, for which she is taking escitalopram (Lexapro) 10 mg once daily. The patient also has a history of “sporadic” headaches. The patient used to be on oral contraceptives, but about 3 months ago an intrauterine device was placed.
Questions:
Given the patient’s gender and age, is this the ideal antiepileptic drug (AED) regimen for this patient?
Items to consider: The patient is a young female of child-bearing age. Divalproex has been shown to be among the least safe AEDs during pregnancy due to a higher risk for birth defects and cognitive side effects on the child.
Ideally, an AED other than divalproex should be chosen. Divalproex also has numerous other side effects.
Polytherapy and higher doses of AEDs can increase the risk of teratogenic side effects.
Levetiracetam is considered to be a relatively safe AED for pregnancy.
The goal should be to simplify the patient’s AED regimen to monotherapy and minimize the dose.
Nikesh Ardeshna, MD
Dr. Ardeshna is the Medical Director of Adult Epilepsy Services at Royal Oak Hospital, Beaumont Health System, in Royal Oak, Michigan.
A 20-year-old female with a history of primary generalized epilepsy presents for establishment of care, as the patient’s insurance had changed. She is currently taking levetiracetam (Keppra) 1500 mg twice daily and divalproex (Depakote) 500 mg once daily by mouth at bedtime. The patient’s last breakthrough seizure was 10 months ago, at which time divalproex was added. The patient has a history of anxiety and depression, for which she is taking escitalopram (Lexapro) 10 mg once daily. The patient also has a history of “sporadic” headaches. The patient used to be on oral contraceptives, but about 3 months ago an intrauterine device was placed.
Questions:
Given the patient’s gender and age, is this the ideal antiepileptic drug (AED) regimen for this patient?
Items to consider: The patient is a young female of child-bearing age. Divalproex has been shown to be among the least safe AEDs during pregnancy due to a higher risk for birth defects and cognitive side effects on the child.
Ideally, an AED other than divalproex should be chosen. Divalproex also has numerous other side effects.
Polytherapy and higher doses of AEDs can increase the risk of teratogenic side effects.
Levetiracetam is considered to be a relatively safe AED for pregnancy.
The goal should be to simplify the patient’s AED regimen to monotherapy and minimize the dose.
Etanercept biosimilar proves effective, tolerated in phase III trial
LONDON – Biosimilar etanercept (CHS-0214) is as effective and well tolerated as etanercept (Enbrel) for the treatment of rheumatoid arthritis according to the results of a randomized, double-blind, phase III trial conducted in 13 countries.
The primary endpoint of an American College of Rheumatology (ACR) 20 at 24 weeks was achieved by 91% of patients given CHS-0214 and 90.6% of those given etanercept, giving a treatment difference of just 0.4%. The percentages of patients achieving ACR 50 (67.6% and 63.7%) and ACR 70 (38.3% and 37.9%) were also comparable.
These are the first clinical data to be presented on this biosimilar, which is a fusion protein comprising the soluble human p75 tumor necrosis factor (TNF) receptor and the Fc region of human immunoglobulin G1.
“Like all biosimilars CHD-0214 has undergone extensive analytical characterization, which had demonstrated highly similar structure and function to etanercept,” study investigator James O’Dell, MD, of the University of Nebraska Medical Center, Omaha, said at the European Congress of Rheumatology. “Studies have demonstrated that CHS-0214 is similar to etanercept with regard to in vitro pharmacology, in vivo pharmacokinetics, and toxicology,” he observed.
A total of 644 patients with moderate to severe rheumatoid arthritis who had an inadequate response to methotrexate were enrolled in the phase III trial and randomized to receive CHS-0214 or etanercept at a subcutaneous dose of 50 mg once a week for 24 weeks. After the double-blind period had ended, patients could continue on open-label CHS-0214 for another 24 weeks. Assessment of efficacy was performed on 512 patients as an issue with the production of the biosimilar resulted in a dosing interruption for 132 patients.
Remission, defined as a DAS28-CRP (Disease Activity Score 28–C-reactive protein) score of less than 2.6, was achieved in a similar percentage of subjects in the CHS-0214 and etanercept groups, at 40.6% and 42.4%, respectively.
There was a similar percentage of any adverse event (60.8% vs. 65%) occurring among patients receiving the biosimilar and those getting etanercept. Treatment-related adverse events (16.4% vs. 21.9%), and treatment-related serious adverse events (0.9% vs. 0.3%) were also comparable. Drug-drug antibodies occurred in numerically fewer patients treated with the biosimilar than with etanercept (1.3% vs. 4.7%).
Asked after his presentation about why he thought the ACR responses seen in the study were so high, Dr. O’Dell said: “We were surprised by that.” There are a number of potential explanations, he suggested. “We had a remarkable completion rate in the trial, 95%, so we lost very few patients, and we are in the process of analyzing other trials to see if that is a major factor.” In addition, patients were on relatively lower doses of methotrexate than in some other trials “because a quarter of them came from Japan,” he said. Patients were also all biologic naive and many were recruited from countries where they don’t have the opportunity to be exposed to a lot of therapies.
“The studies on all biosimilars so far suggest that they are indeed biosimilar,” Dr. O’Dell said in an interview. So how might clinicians begin to choose between the various biosimilars? “Well, there is very, very little data to compare any biologic to another biologic,” he answered.
“You haven’t seen studies comparing one TNF inhibitor to another TNF inhibitor. So it’s not surprising that we don’t have data that compare this biosimilar to that biosimilar,” he added. Such studies are probably also unlikely to ever be conducted so the choice of biosimilar, like anti-TNF therapy is probably going to be dictated by national and local guidelines, and medical insurance policies.
“Today, the biosimilars look biosimilar, so I have no problems if somebody tells me I have to start a biosimilar as opposed to the innovator product. I do have a problem if they tell me I have to switch, because that’s interchangeability and that’s a whole different story,” he noted.
Dr. O’Dell observed, however, that data from the DANBIO registry presented during the same session had shown that an enforced national switch from infliximab (Remicade) to biosimilar infliximab had shown that this did not appear to pose a problem in routine care in Denmark. Indeed 3 months’ clinical outcomes in patients with RA, psoriatic arthritis, or axial spondyloarthritis who were switched appeared to be comparable. Whether this is true across all the biosimilars needs to be established.
Coherus Biosciences funded the study. Dr. O’Dell has been a study investigator for, received research grants from, or acted as a speaker or consultant to Coherus Biosciences, Medac, Eli Lilly, Bristol-Myers Squibb, GlaxoSmithKline, Antares, and Crescendo.
LONDON – Biosimilar etanercept (CHS-0214) is as effective and well tolerated as etanercept (Enbrel) for the treatment of rheumatoid arthritis according to the results of a randomized, double-blind, phase III trial conducted in 13 countries.
The primary endpoint of an American College of Rheumatology (ACR) 20 at 24 weeks was achieved by 91% of patients given CHS-0214 and 90.6% of those given etanercept, giving a treatment difference of just 0.4%. The percentages of patients achieving ACR 50 (67.6% and 63.7%) and ACR 70 (38.3% and 37.9%) were also comparable.
These are the first clinical data to be presented on this biosimilar, which is a fusion protein comprising the soluble human p75 tumor necrosis factor (TNF) receptor and the Fc region of human immunoglobulin G1.
“Like all biosimilars CHD-0214 has undergone extensive analytical characterization, which had demonstrated highly similar structure and function to etanercept,” study investigator James O’Dell, MD, of the University of Nebraska Medical Center, Omaha, said at the European Congress of Rheumatology. “Studies have demonstrated that CHS-0214 is similar to etanercept with regard to in vitro pharmacology, in vivo pharmacokinetics, and toxicology,” he observed.
A total of 644 patients with moderate to severe rheumatoid arthritis who had an inadequate response to methotrexate were enrolled in the phase III trial and randomized to receive CHS-0214 or etanercept at a subcutaneous dose of 50 mg once a week for 24 weeks. After the double-blind period had ended, patients could continue on open-label CHS-0214 for another 24 weeks. Assessment of efficacy was performed on 512 patients as an issue with the production of the biosimilar resulted in a dosing interruption for 132 patients.
Remission, defined as a DAS28-CRP (Disease Activity Score 28–C-reactive protein) score of less than 2.6, was achieved in a similar percentage of subjects in the CHS-0214 and etanercept groups, at 40.6% and 42.4%, respectively.
There was a similar percentage of any adverse event (60.8% vs. 65%) occurring among patients receiving the biosimilar and those getting etanercept. Treatment-related adverse events (16.4% vs. 21.9%), and treatment-related serious adverse events (0.9% vs. 0.3%) were also comparable. Drug-drug antibodies occurred in numerically fewer patients treated with the biosimilar than with etanercept (1.3% vs. 4.7%).
Asked after his presentation about why he thought the ACR responses seen in the study were so high, Dr. O’Dell said: “We were surprised by that.” There are a number of potential explanations, he suggested. “We had a remarkable completion rate in the trial, 95%, so we lost very few patients, and we are in the process of analyzing other trials to see if that is a major factor.” In addition, patients were on relatively lower doses of methotrexate than in some other trials “because a quarter of them came from Japan,” he said. Patients were also all biologic naive and many were recruited from countries where they don’t have the opportunity to be exposed to a lot of therapies.
“The studies on all biosimilars so far suggest that they are indeed biosimilar,” Dr. O’Dell said in an interview. So how might clinicians begin to choose between the various biosimilars? “Well, there is very, very little data to compare any biologic to another biologic,” he answered.
“You haven’t seen studies comparing one TNF inhibitor to another TNF inhibitor. So it’s not surprising that we don’t have data that compare this biosimilar to that biosimilar,” he added. Such studies are probably also unlikely to ever be conducted so the choice of biosimilar, like anti-TNF therapy is probably going to be dictated by national and local guidelines, and medical insurance policies.
“Today, the biosimilars look biosimilar, so I have no problems if somebody tells me I have to start a biosimilar as opposed to the innovator product. I do have a problem if they tell me I have to switch, because that’s interchangeability and that’s a whole different story,” he noted.
Dr. O’Dell observed, however, that data from the DANBIO registry presented during the same session had shown that an enforced national switch from infliximab (Remicade) to biosimilar infliximab had shown that this did not appear to pose a problem in routine care in Denmark. Indeed 3 months’ clinical outcomes in patients with RA, psoriatic arthritis, or axial spondyloarthritis who were switched appeared to be comparable. Whether this is true across all the biosimilars needs to be established.
Coherus Biosciences funded the study. Dr. O’Dell has been a study investigator for, received research grants from, or acted as a speaker or consultant to Coherus Biosciences, Medac, Eli Lilly, Bristol-Myers Squibb, GlaxoSmithKline, Antares, and Crescendo.
LONDON – Biosimilar etanercept (CHS-0214) is as effective and well tolerated as etanercept (Enbrel) for the treatment of rheumatoid arthritis according to the results of a randomized, double-blind, phase III trial conducted in 13 countries.
The primary endpoint of an American College of Rheumatology (ACR) 20 at 24 weeks was achieved by 91% of patients given CHS-0214 and 90.6% of those given etanercept, giving a treatment difference of just 0.4%. The percentages of patients achieving ACR 50 (67.6% and 63.7%) and ACR 70 (38.3% and 37.9%) were also comparable.
These are the first clinical data to be presented on this biosimilar, which is a fusion protein comprising the soluble human p75 tumor necrosis factor (TNF) receptor and the Fc region of human immunoglobulin G1.
“Like all biosimilars CHD-0214 has undergone extensive analytical characterization, which had demonstrated highly similar structure and function to etanercept,” study investigator James O’Dell, MD, of the University of Nebraska Medical Center, Omaha, said at the European Congress of Rheumatology. “Studies have demonstrated that CHS-0214 is similar to etanercept with regard to in vitro pharmacology, in vivo pharmacokinetics, and toxicology,” he observed.
A total of 644 patients with moderate to severe rheumatoid arthritis who had an inadequate response to methotrexate were enrolled in the phase III trial and randomized to receive CHS-0214 or etanercept at a subcutaneous dose of 50 mg once a week for 24 weeks. After the double-blind period had ended, patients could continue on open-label CHS-0214 for another 24 weeks. Assessment of efficacy was performed on 512 patients as an issue with the production of the biosimilar resulted in a dosing interruption for 132 patients.
Remission, defined as a DAS28-CRP (Disease Activity Score 28–C-reactive protein) score of less than 2.6, was achieved in a similar percentage of subjects in the CHS-0214 and etanercept groups, at 40.6% and 42.4%, respectively.
There was a similar percentage of any adverse event (60.8% vs. 65%) occurring among patients receiving the biosimilar and those getting etanercept. Treatment-related adverse events (16.4% vs. 21.9%), and treatment-related serious adverse events (0.9% vs. 0.3%) were also comparable. Drug-drug antibodies occurred in numerically fewer patients treated with the biosimilar than with etanercept (1.3% vs. 4.7%).
Asked after his presentation about why he thought the ACR responses seen in the study were so high, Dr. O’Dell said: “We were surprised by that.” There are a number of potential explanations, he suggested. “We had a remarkable completion rate in the trial, 95%, so we lost very few patients, and we are in the process of analyzing other trials to see if that is a major factor.” In addition, patients were on relatively lower doses of methotrexate than in some other trials “because a quarter of them came from Japan,” he said. Patients were also all biologic naive and many were recruited from countries where they don’t have the opportunity to be exposed to a lot of therapies.
“The studies on all biosimilars so far suggest that they are indeed biosimilar,” Dr. O’Dell said in an interview. So how might clinicians begin to choose between the various biosimilars? “Well, there is very, very little data to compare any biologic to another biologic,” he answered.
“You haven’t seen studies comparing one TNF inhibitor to another TNF inhibitor. So it’s not surprising that we don’t have data that compare this biosimilar to that biosimilar,” he added. Such studies are probably also unlikely to ever be conducted so the choice of biosimilar, like anti-TNF therapy is probably going to be dictated by national and local guidelines, and medical insurance policies.
“Today, the biosimilars look biosimilar, so I have no problems if somebody tells me I have to start a biosimilar as opposed to the innovator product. I do have a problem if they tell me I have to switch, because that’s interchangeability and that’s a whole different story,” he noted.
Dr. O’Dell observed, however, that data from the DANBIO registry presented during the same session had shown that an enforced national switch from infliximab (Remicade) to biosimilar infliximab had shown that this did not appear to pose a problem in routine care in Denmark. Indeed 3 months’ clinical outcomes in patients with RA, psoriatic arthritis, or axial spondyloarthritis who were switched appeared to be comparable. Whether this is true across all the biosimilars needs to be established.
Coherus Biosciences funded the study. Dr. O’Dell has been a study investigator for, received research grants from, or acted as a speaker or consultant to Coherus Biosciences, Medac, Eli Lilly, Bristol-Myers Squibb, GlaxoSmithKline, Antares, and Crescendo.
AT THE EULAR 2016 CONGRESS
Key clinical point: Biosimilar etanercept (CHS-0214) was shown to be as effective and well tolerated as etanercept.
Major finding: ACR 20 at 24 weeks (primary endpoint) was achieved by 91% given the biosimilar and 90.6% of those given etanercept.
Data source: Multicenter, randomized, double-blind, phase III trial comparing the etanercept biosimilar CHS-0215 with etanercept in 644 patients with rheumatoid arthritis.
Disclosures: Coherus Biosciences funded the study. Dr. O’Dell has been a study investigator for, received research grants from, or acted as a speaker or consultant to Coherus Biosciences, Medac, Eli Lilly, Bristol-Myers Squibb, GlaxoSmithKline, Antares, and Crescendo.
New Mexico high court states assisted suicide is not a right
On June 30, New Mexico became the latest state to throw out a challenge to a law banning physician-assisted suicide. In Morris v. Brandenburg, proponents of the right-to-die movement contended that medical aid in dying was a fundamental right, meaning that any law that restricted the right should be presumed to be invalid unless the state had a compelling reason for the restriction.
In a unanimous decision, the New Mexico Supreme Court held that there was no such right under that state’s constitution, and that even if the right had existed, the state had several compelling reasons to restrict it: to protect “the integrity and ethics of the medical profession”; to protect “vulnerable groups – including the poor, the elderly, and disabled persons – from the risk of subtle coercion and undue influence in end-of-life situations, including pressures associated with the substantial financial burden of end-of-life health care costs”; and to protect “against voluntary or involuntary euthanasia because if physician aid in dying is a constitutional right, it must be made available to everyone, even when a duly appointed surrogate makes the decision, and even when the patient is unable to self-administer the life-ending medication.”1
Let me repeat that last point:
If aid in dying is a constitutional right, then medical euthanasia is inevitable. In other words, if a patient is too disabled to commit suicide on his or her own, a physician would be obligated to kill the patient.
Proponents will dismiss this last statement as alarmist and unfounded. They will point to a 20-year history of legalized aid in dying in Oregon, and insist there has never been a problem and no attempt to expand the practice to include euthanasia.
What they don’t mention is that such an attempt would be politically disastrous for the right-to-die movement. In 2015, 25 states and the District of Columbia considered legalization bills.2 After careful consideration and debate, these bills failed in every state except California, and in that state it passed only because of an extraordinary and unusual legislative maneuver. The bill initially failed in regular session, but passed in special session when the California Medical Association (CMA) shifted to a neutral stance. That shift would never have happened had CMA members been aware of the legal inevitability of euthanasia. At this stage of the political process, the goal of the right-to-die movement is to get laws on the books in as many states as possible before pushing for more.
The New Mexico opinion echoes a similar decision issued this past May by a New York appellate court. The plaintiffs in that case were three terminally ill New Yorkers and five doctors, as well as two euthanasia advocacy groups. The arguments were virtually identical to those made in New Mexico, asserting a fundamental right to medical aid in dying based upon personal autonomy. They additionally sought an exception to New York criminal law, which defined assisted suicide as second-degree manslaughter. In Myers v. Schneiderman, the court observed that no appellate court had ever found medical aid in dying to be a fundamental right and that no new developments justified a change in this interpretation.3 While the court acknowledged some change in public opinion as reflected in telephone surveys, it correctly gave little weight to this argument: “Plaintiffs fail to allege whether those public polls reflect the opinion of people who are fully informed of the arguments espoused by those who caution against permitting aid-in-dying, such as those articulated in the New York State Task Force on Life and the Law.”
This task force issued the following recommendation in its 1994 report,4 “When Death is Sought: Assisted Suicide and Euthanasia in the Medical Context”: “The Task Force members unanimously recommend that existing law should not be changed to permit assisted suicide or euthanasia. Legalizing assisted suicide and euthanasia would pose profound risks to many individuals who are ill and vulnerable. The Task Force members concluded that the potential dangers of this dramatic change in public policy would outweigh any benefit that might be achieved” (Chapter 6, p. 120).
The New York aid-in-dying bill did not progress out of committee by the time the session ended in June.
Lastly, both of these cases concluded that medical aid in dying was suicide. In Myers, Judge Angela M. Mazzarelli stated: “The word ‘suicide’ has a straightforward meaning, and a dictionary is hardly necessary … It is traditionally defined as ‘the act or instance of taking one’s own life voluntarily and intentionally.’ ... Whatever label one puts on the act that plaintiffs are asking us to permit, it unquestionably fits that literal description.”
Regardless of the label used – aid in dying, assisted suicide, or self-deliverance – it is not a civil right.
Resources
1. http://www.nmcompcomm.us/nmcases/nmsc/slips/SC35,478.pdf
2. https://www.deathwithdignity.org/assisted-dying-chronology/
3. http://www.nycourts.gov/reporter/3dseries/2016/2016_03457.htm
4. https://www.health.ny.gov/regulations/task_force/reports_publications/when_death_is_sought/
Dr. Hanson is a forensic psychiatrist and coauthor of “Shrink Rap: Three Psychiatrists Explain Their Work.” The opinions expressed are those of the author only, and do not represent those of any of Dr. Hanson’s employers or consultees, including the Maryland Department of Health and Mental Hygiene or the Maryland Division of Correction.
On June 30, New Mexico became the latest state to throw out a challenge to a law banning physician-assisted suicide. In Morris v. Brandenburg, proponents of the right-to-die movement contended that medical aid in dying was a fundamental right, meaning that any law that restricted the right should be presumed to be invalid unless the state had a compelling reason for the restriction.
In a unanimous decision, the New Mexico Supreme Court held that there was no such right under that state’s constitution, and that even if the right had existed, the state had several compelling reasons to restrict it: to protect “the integrity and ethics of the medical profession”; to protect “vulnerable groups – including the poor, the elderly, and disabled persons – from the risk of subtle coercion and undue influence in end-of-life situations, including pressures associated with the substantial financial burden of end-of-life health care costs”; and to protect “against voluntary or involuntary euthanasia because if physician aid in dying is a constitutional right, it must be made available to everyone, even when a duly appointed surrogate makes the decision, and even when the patient is unable to self-administer the life-ending medication.”1
Let me repeat that last point:
If aid in dying is a constitutional right, then medical euthanasia is inevitable. In other words, if a patient is too disabled to commit suicide on his or her own, a physician would be obligated to kill the patient.
Proponents will dismiss this last statement as alarmist and unfounded. They will point to a 20-year history of legalized aid in dying in Oregon, and insist there has never been a problem and no attempt to expand the practice to include euthanasia.
What they don’t mention is that such an attempt would be politically disastrous for the right-to-die movement. In 2015, 25 states and the District of Columbia considered legalization bills.2 After careful consideration and debate, these bills failed in every state except California, and in that state it passed only because of an extraordinary and unusual legislative maneuver. The bill initially failed in regular session, but passed in special session when the California Medical Association (CMA) shifted to a neutral stance. That shift would never have happened had CMA members been aware of the legal inevitability of euthanasia. At this stage of the political process, the goal of the right-to-die movement is to get laws on the books in as many states as possible before pushing for more.
The New Mexico opinion echoes a similar decision issued this past May by a New York appellate court. The plaintiffs in that case were three terminally ill New Yorkers and five doctors, as well as two euthanasia advocacy groups. The arguments were virtually identical to those made in New Mexico, asserting a fundamental right to medical aid in dying based upon personal autonomy. They additionally sought an exception to New York criminal law, which defined assisted suicide as second-degree manslaughter. In Myers v. Schneiderman, the court observed that no appellate court had ever found medical aid in dying to be a fundamental right and that no new developments justified a change in this interpretation.3 While the court acknowledged some change in public opinion as reflected in telephone surveys, it correctly gave little weight to this argument: “Plaintiffs fail to allege whether those public polls reflect the opinion of people who are fully informed of the arguments espoused by those who caution against permitting aid-in-dying, such as those articulated in the New York State Task Force on Life and the Law.”
This task force issued the following recommendation in its 1994 report,4 “When Death is Sought: Assisted Suicide and Euthanasia in the Medical Context”: “The Task Force members unanimously recommend that existing law should not be changed to permit assisted suicide or euthanasia. Legalizing assisted suicide and euthanasia would pose profound risks to many individuals who are ill and vulnerable. The Task Force members concluded that the potential dangers of this dramatic change in public policy would outweigh any benefit that might be achieved” (Chapter 6, p. 120).
The New York aid-in-dying bill did not progress out of committee by the time the session ended in June.
Lastly, both of these cases concluded that medical aid in dying was suicide. In Myers, Judge Angela M. Mazzarelli stated: “The word ‘suicide’ has a straightforward meaning, and a dictionary is hardly necessary … It is traditionally defined as ‘the act or instance of taking one’s own life voluntarily and intentionally.’ ... Whatever label one puts on the act that plaintiffs are asking us to permit, it unquestionably fits that literal description.”
Regardless of the label used – aid in dying, assisted suicide, or self-deliverance – it is not a civil right.
Resources
1. http://www.nmcompcomm.us/nmcases/nmsc/slips/SC35,478.pdf
2. https://www.deathwithdignity.org/assisted-dying-chronology/
3. http://www.nycourts.gov/reporter/3dseries/2016/2016_03457.htm
4. https://www.health.ny.gov/regulations/task_force/reports_publications/when_death_is_sought/
Dr. Hanson is a forensic psychiatrist and coauthor of “Shrink Rap: Three Psychiatrists Explain Their Work.” The opinions expressed are those of the author only, and do not represent those of any of Dr. Hanson’s employers or consultees, including the Maryland Department of Health and Mental Hygiene or the Maryland Division of Correction.
On June 30, New Mexico became the latest state to throw out a challenge to a law banning physician-assisted suicide. In Morris v. Brandenburg, proponents of the right-to-die movement contended that medical aid in dying was a fundamental right, meaning that any law that restricted the right should be presumed to be invalid unless the state had a compelling reason for the restriction.
In a unanimous decision, the New Mexico Supreme Court held that there was no such right under that state’s constitution, and that even if the right had existed, the state had several compelling reasons to restrict it: to protect “the integrity and ethics of the medical profession”; to protect “vulnerable groups – including the poor, the elderly, and disabled persons – from the risk of subtle coercion and undue influence in end-of-life situations, including pressures associated with the substantial financial burden of end-of-life health care costs”; and to protect “against voluntary or involuntary euthanasia because if physician aid in dying is a constitutional right, it must be made available to everyone, even when a duly appointed surrogate makes the decision, and even when the patient is unable to self-administer the life-ending medication.”1
Let me repeat that last point:
If aid in dying is a constitutional right, then medical euthanasia is inevitable. In other words, if a patient is too disabled to commit suicide on his or her own, a physician would be obligated to kill the patient.
Proponents will dismiss this last statement as alarmist and unfounded. They will point to a 20-year history of legalized aid in dying in Oregon, and insist there has never been a problem and no attempt to expand the practice to include euthanasia.
What they don’t mention is that such an attempt would be politically disastrous for the right-to-die movement. In 2015, 25 states and the District of Columbia considered legalization bills.2 After careful consideration and debate, these bills failed in every state except California, and in that state it passed only because of an extraordinary and unusual legislative maneuver. The bill initially failed in regular session, but passed in special session when the California Medical Association (CMA) shifted to a neutral stance. That shift would never have happened had CMA members been aware of the legal inevitability of euthanasia. At this stage of the political process, the goal of the right-to-die movement is to get laws on the books in as many states as possible before pushing for more.
The New Mexico opinion echoes a similar decision issued this past May by a New York appellate court. The plaintiffs in that case were three terminally ill New Yorkers and five doctors, as well as two euthanasia advocacy groups. The arguments were virtually identical to those made in New Mexico, asserting a fundamental right to medical aid in dying based upon personal autonomy. They additionally sought an exception to New York criminal law, which defined assisted suicide as second-degree manslaughter. In Myers v. Schneiderman, the court observed that no appellate court had ever found medical aid in dying to be a fundamental right and that no new developments justified a change in this interpretation.3 While the court acknowledged some change in public opinion as reflected in telephone surveys, it correctly gave little weight to this argument: “Plaintiffs fail to allege whether those public polls reflect the opinion of people who are fully informed of the arguments espoused by those who caution against permitting aid-in-dying, such as those articulated in the New York State Task Force on Life and the Law.”
This task force issued the following recommendation in its 1994 report,4 “When Death is Sought: Assisted Suicide and Euthanasia in the Medical Context”: “The Task Force members unanimously recommend that existing law should not be changed to permit assisted suicide or euthanasia. Legalizing assisted suicide and euthanasia would pose profound risks to many individuals who are ill and vulnerable. The Task Force members concluded that the potential dangers of this dramatic change in public policy would outweigh any benefit that might be achieved” (Chapter 6, p. 120).
The New York aid-in-dying bill did not progress out of committee by the time the session ended in June.
Lastly, both of these cases concluded that medical aid in dying was suicide. In Myers, Judge Angela M. Mazzarelli stated: “The word ‘suicide’ has a straightforward meaning, and a dictionary is hardly necessary … It is traditionally defined as ‘the act or instance of taking one’s own life voluntarily and intentionally.’ ... Whatever label one puts on the act that plaintiffs are asking us to permit, it unquestionably fits that literal description.”
Regardless of the label used – aid in dying, assisted suicide, or self-deliverance – it is not a civil right.
Resources
1. http://www.nmcompcomm.us/nmcases/nmsc/slips/SC35,478.pdf
2. https://www.deathwithdignity.org/assisted-dying-chronology/
3. http://www.nycourts.gov/reporter/3dseries/2016/2016_03457.htm
4. https://www.health.ny.gov/regulations/task_force/reports_publications/when_death_is_sought/
Dr. Hanson is a forensic psychiatrist and coauthor of “Shrink Rap: Three Psychiatrists Explain Their Work.” The opinions expressed are those of the author only, and do not represent those of any of Dr. Hanson’s employers or consultees, including the Maryland Department of Health and Mental Hygiene or the Maryland Division of Correction.