User login
Centers for Disease Control and Prevention (CDC): Advisory Committee on Immunization Practices (ACIP)
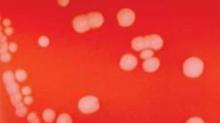
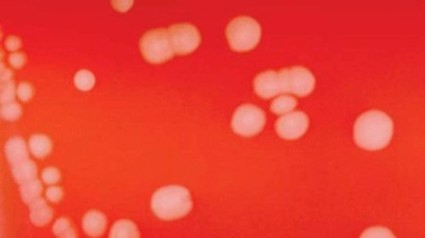
CDC okays group meningitis vaccination in emergency
Large-scale vaccination with a still-unapproved vaccine should be considered if multiple cases of serogroup B meningitis occur within 6 months in a community, a workgroup of the Centers for Disease Control and Prevention recommended June 26.
In groups of fewer than 5,000 people, vaccination should be considered if two or more cases present within 6 months. In larger communities, vaccination may be implemented if three or more cases present over 6 months, Jessica MacNeil said at a meeting of the CDC’s Advisory Committee on Immunization Practices.
The recommendation, which will be posted on the CDC’s meningitis web pages, was developed in response to last year’s group B meningitis outbreaks at the University of California, Santa Barbara, and at Princeton University, said Ms. MacNeil, an epidemiologist at the CDC and liaison with ACIP’s Meningococcal Outbreak Working Group.
When the outbreaks occurred, the CDC sponsored an expanded-access Investigational New Drug (IND) application so that students at both campuses could receive the unapproved vaccine.
The workgroup was convened in January and has conducted biweekly meetings to develop some interim guidance that would allow prompt response to future outbreaks in the absence of an approved vaccine.
"There are delays in getting a vaccination campaign in place under an IND," Ms. MacNeil said. "Therefore, we need to consider options early. We recognize that waiting for additional cases to occur before acting is difficult for organizations, so we need to have clear steps for each case already identified."
The proposed guideline begins with testing clinical specimens of Neisseria meningitides. Serogrouping should be initiated with 24 hours of collection. Labs that can’t do so within 72 hours need to transfer the specimen to a lab that can or get samples to the CDC within 24 hours.
The CDC will work with state and local health departments in determining whether or not a large-scale vaccination is appropriate. That decision involves several considerations: the number of cases, the size of the community involved, the strains present in the cases, the feasibility and cost of vaccinating, and vaccine availability.
For groups of fewer than 5,000, one case of meningitis should activate routine public health practices: serogrouping, a case investigation, and chemoprophylaxis for close contacts. The isolates should be saved.
Once two or more cases have been identified, specimens should be sent to the CDC for molecular genotyping. If they are identified as serogroup B and, thus, likely to be covered by the MenB vaccine, local health officials may consult with CDC about using the vaccine under the expanded access IND program.
In communities of 5,000 or more, a single case will prompt the same routine public health practices. If two cases occur within 6 months, the isolates should be tested. If they are both serogroup B, the CDC should be notified and the situation carefully monitored. If three or more cases develop and the isolates are all serogroup B, then the IND vaccination campaign can be considered.
If the CDC-sponsored IND is activated, any organization that provides the vaccine must appoint a local coinvestigator. The group will be required to participate in safety follow-up studies as required by the program.
Ms. MacNeil added that the universities that have implemented MenB vaccination campaigns are bearing the cost of obtaining and administering the vaccine.
She also discussed the value of large-scale chemoprophylaxis. A literature review identified 18 instances in which mass chemoprophylaxis had been used as an outbreak response. These suggested that chemoprophylaxis resulted in decreases in carriage soon after, but no permanent clearance.
"Mass chemoprophylaxis is not recommended as a stand-alone measure to control outbreaks of meningococcal disease," she said. "It may be considered as an interim measure to reduce carriage and transmission in the period before potential protection from vaccination can be achieved."
However, she added, chemoprophylaxis only would be effective in small or closed populations with limited mixing from the outside – like jails or residential facilities.
"It would be difficult to ensure that everyone in the target group could receive treatment within a short time frame," she said. And dosing everyone in a population opens up the possibility of idiosyncratic adverse reactions and interactions with other commonly used medications, including antidepressants.
"In many outbreak settings, particularly when a small, closed, at-risk population can’t be defined, the disadvantages outweigh the possible benefits," she said.
She said the guidance will be published within a few days on the CDC’s meningitis pages.
Large-scale vaccination with a still-unapproved vaccine should be considered if multiple cases of serogroup B meningitis occur within 6 months in a community, a workgroup of the Centers for Disease Control and Prevention recommended June 26.
In groups of fewer than 5,000 people, vaccination should be considered if two or more cases present within 6 months. In larger communities, vaccination may be implemented if three or more cases present over 6 months, Jessica MacNeil said at a meeting of the CDC’s Advisory Committee on Immunization Practices.
The recommendation, which will be posted on the CDC’s meningitis web pages, was developed in response to last year’s group B meningitis outbreaks at the University of California, Santa Barbara, and at Princeton University, said Ms. MacNeil, an epidemiologist at the CDC and liaison with ACIP’s Meningococcal Outbreak Working Group.
When the outbreaks occurred, the CDC sponsored an expanded-access Investigational New Drug (IND) application so that students at both campuses could receive the unapproved vaccine.
The workgroup was convened in January and has conducted biweekly meetings to develop some interim guidance that would allow prompt response to future outbreaks in the absence of an approved vaccine.
"There are delays in getting a vaccination campaign in place under an IND," Ms. MacNeil said. "Therefore, we need to consider options early. We recognize that waiting for additional cases to occur before acting is difficult for organizations, so we need to have clear steps for each case already identified."
The proposed guideline begins with testing clinical specimens of Neisseria meningitides. Serogrouping should be initiated with 24 hours of collection. Labs that can’t do so within 72 hours need to transfer the specimen to a lab that can or get samples to the CDC within 24 hours.
The CDC will work with state and local health departments in determining whether or not a large-scale vaccination is appropriate. That decision involves several considerations: the number of cases, the size of the community involved, the strains present in the cases, the feasibility and cost of vaccinating, and vaccine availability.
For groups of fewer than 5,000, one case of meningitis should activate routine public health practices: serogrouping, a case investigation, and chemoprophylaxis for close contacts. The isolates should be saved.
Once two or more cases have been identified, specimens should be sent to the CDC for molecular genotyping. If they are identified as serogroup B and, thus, likely to be covered by the MenB vaccine, local health officials may consult with CDC about using the vaccine under the expanded access IND program.
In communities of 5,000 or more, a single case will prompt the same routine public health practices. If two cases occur within 6 months, the isolates should be tested. If they are both serogroup B, the CDC should be notified and the situation carefully monitored. If three or more cases develop and the isolates are all serogroup B, then the IND vaccination campaign can be considered.
If the CDC-sponsored IND is activated, any organization that provides the vaccine must appoint a local coinvestigator. The group will be required to participate in safety follow-up studies as required by the program.
Ms. MacNeil added that the universities that have implemented MenB vaccination campaigns are bearing the cost of obtaining and administering the vaccine.
She also discussed the value of large-scale chemoprophylaxis. A literature review identified 18 instances in which mass chemoprophylaxis had been used as an outbreak response. These suggested that chemoprophylaxis resulted in decreases in carriage soon after, but no permanent clearance.
"Mass chemoprophylaxis is not recommended as a stand-alone measure to control outbreaks of meningococcal disease," she said. "It may be considered as an interim measure to reduce carriage and transmission in the period before potential protection from vaccination can be achieved."
However, she added, chemoprophylaxis only would be effective in small or closed populations with limited mixing from the outside – like jails or residential facilities.
"It would be difficult to ensure that everyone in the target group could receive treatment within a short time frame," she said. And dosing everyone in a population opens up the possibility of idiosyncratic adverse reactions and interactions with other commonly used medications, including antidepressants.
"In many outbreak settings, particularly when a small, closed, at-risk population can’t be defined, the disadvantages outweigh the possible benefits," she said.
She said the guidance will be published within a few days on the CDC’s meningitis pages.
Large-scale vaccination with a still-unapproved vaccine should be considered if multiple cases of serogroup B meningitis occur within 6 months in a community, a workgroup of the Centers for Disease Control and Prevention recommended June 26.
In groups of fewer than 5,000 people, vaccination should be considered if two or more cases present within 6 months. In larger communities, vaccination may be implemented if three or more cases present over 6 months, Jessica MacNeil said at a meeting of the CDC’s Advisory Committee on Immunization Practices.
The recommendation, which will be posted on the CDC’s meningitis web pages, was developed in response to last year’s group B meningitis outbreaks at the University of California, Santa Barbara, and at Princeton University, said Ms. MacNeil, an epidemiologist at the CDC and liaison with ACIP’s Meningococcal Outbreak Working Group.
When the outbreaks occurred, the CDC sponsored an expanded-access Investigational New Drug (IND) application so that students at both campuses could receive the unapproved vaccine.
The workgroup was convened in January and has conducted biweekly meetings to develop some interim guidance that would allow prompt response to future outbreaks in the absence of an approved vaccine.
"There are delays in getting a vaccination campaign in place under an IND," Ms. MacNeil said. "Therefore, we need to consider options early. We recognize that waiting for additional cases to occur before acting is difficult for organizations, so we need to have clear steps for each case already identified."
The proposed guideline begins with testing clinical specimens of Neisseria meningitides. Serogrouping should be initiated with 24 hours of collection. Labs that can’t do so within 72 hours need to transfer the specimen to a lab that can or get samples to the CDC within 24 hours.
The CDC will work with state and local health departments in determining whether or not a large-scale vaccination is appropriate. That decision involves several considerations: the number of cases, the size of the community involved, the strains present in the cases, the feasibility and cost of vaccinating, and vaccine availability.
For groups of fewer than 5,000, one case of meningitis should activate routine public health practices: serogrouping, a case investigation, and chemoprophylaxis for close contacts. The isolates should be saved.
Once two or more cases have been identified, specimens should be sent to the CDC for molecular genotyping. If they are identified as serogroup B and, thus, likely to be covered by the MenB vaccine, local health officials may consult with CDC about using the vaccine under the expanded access IND program.
In communities of 5,000 or more, a single case will prompt the same routine public health practices. If two cases occur within 6 months, the isolates should be tested. If they are both serogroup B, the CDC should be notified and the situation carefully monitored. If three or more cases develop and the isolates are all serogroup B, then the IND vaccination campaign can be considered.
If the CDC-sponsored IND is activated, any organization that provides the vaccine must appoint a local coinvestigator. The group will be required to participate in safety follow-up studies as required by the program.
Ms. MacNeil added that the universities that have implemented MenB vaccination campaigns are bearing the cost of obtaining and administering the vaccine.
She also discussed the value of large-scale chemoprophylaxis. A literature review identified 18 instances in which mass chemoprophylaxis had been used as an outbreak response. These suggested that chemoprophylaxis resulted in decreases in carriage soon after, but no permanent clearance.
"Mass chemoprophylaxis is not recommended as a stand-alone measure to control outbreaks of meningococcal disease," she said. "It may be considered as an interim measure to reduce carriage and transmission in the period before potential protection from vaccination can be achieved."
However, she added, chemoprophylaxis only would be effective in small or closed populations with limited mixing from the outside – like jails or residential facilities.
"It would be difficult to ensure that everyone in the target group could receive treatment within a short time frame," she said. And dosing everyone in a population opens up the possibility of idiosyncratic adverse reactions and interactions with other commonly used medications, including antidepressants.
"In many outbreak settings, particularly when a small, closed, at-risk population can’t be defined, the disadvantages outweigh the possible benefits," she said.
She said the guidance will be published within a few days on the CDC’s meningitis pages.
FROM AN ACIP MEETING
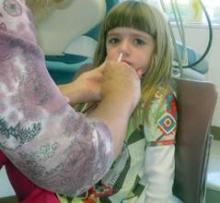
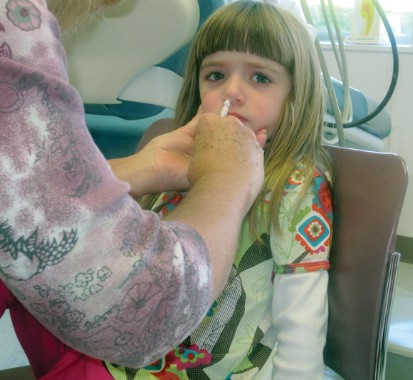
CDC panel: For children, live attenuated intranasal flu vaccine is best
The live attenuated intranasal flu vaccine is the preferred method of immunizing healthy children aged 2-8 years against seasonal influenza, a federal panel has recommended.
At a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices, committee members unanimously agreed that healthy children with no contraindications should receive the quadrivalent live attenuated vaccine (LAIV) rather than an inactivated vaccine, provided that the LAIV is readily available.
"If the LAIV is not immediately available, [inactivated influenza vaccine (IIV)] should be used," the recommendation reads. "Vaccination should not be delayed in order to procure LAIV."
All children aged 6 months or older should receive influenza vaccine, according to the recommendation. "Influenza vaccine should not be delayed to procure a specific vaccine preparation if an appropriate one is already available."
The committee approved the recommendation after hearing an evidence review that found the LAIV to be "moderately" more effective for this age group than the inactivated vaccine was. Although the data are not plentiful, they suggest that it would probably prevent about 47 more lab-confirmed cases of flu per 1,000 children than the IIV, Dr. Lisa Grohskopf said
"The benefits appear to outweigh the potential harms, with modestly better efficacy," and no appreciable increase in adverse events associated with the live vaccine, said Dr. Grohskopf, who is a medical officer in the influenza division of the Centers for Disease Control and Prevention in Atlanta.
Her review was based on two studies – one published in 2006 and one in 2007. The earlier study was an open-label trial that randomized about 2,300 children aged 6-71 months to either the LAIV or an IIV. The much larger 2007 study randomized about 16,000 children aged 6-59 months to the two vaccine types or to matching placebos.
The pooled analysis was limited to healthy children aged 24-59 months who were without a history of asthma or wheezing. The primary endpoint of lab-confirmed influenza infection was significantly less likely among children who had received the LAIV (hazard ratio, 0.47). The secondary endpoint of influenza-associated otitis media was also significantly less likely to occur among those who got the LAIV (HR, 0.47).
Neither of the vaccines was associated with any medically significant wheezing or fever, Dr. Grohskopf said.
She also examined these same endpoints in children who had a history of wheezing or asthma. The LAIV significantly reduced the risk of lab-confirmed influenza in this group (HR, 0.53). There were no significantly increased risks of medically significant wheezing in the children, regardless of whether or not they had experienced wheezing within the past 12 months.
However, Dr. Grohskopf said, she and her colleagues didn’t feel that the studies were strong enough to make any initial recommendations to the committee about whether the LAIV could safely be used in children with a history of wheezing or asthma. The studies didn’t drill down into data on this group enough, and the data are now too old to be completely reliable for this population.
Asthma and wheezing are not strict contraindications to the LAIV, she said, but ACIP does not recommend it for children with those issues or for those with any chronic conditions that might predispose them to complications from the flu.
The committee also heard that the strains included in the 2014-2015 vaccine will be the same as last season’s. Because of this, children who received at least one dose of the 2013-2014 vaccine will only need one dose of this year’s vaccine to ensure immunity. "This is analogous to what happened in the 2011-2012 season, when the vaccine strains were unchanged from the previous season," Dr. Grohskopf said.
She also touched on the issue of cost. "We haven’t done a formal cost-effectiveness analysis," she said.
"It’s likely to be a very complex undertaking because of the large number of products out there. It will probably be a few years before that could be done."
The best estimates available now can be drawn from the 2013-2014 private sector costs associated with CDC’s Vaccines for Children program. "The cost of the LAIV intranasal vaccine is $22.70/dose, and for the inactivated vaccine, there is a broad range of variability, from $7.65-$14.81/dose for the trivalent to $14.90-$21.09/dose for the quadrivalent."
On Twitter @alz_gal
The live attenuated intranasal flu vaccine is the preferred method of immunizing healthy children aged 2-8 years against seasonal influenza, a federal panel has recommended.
At a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices, committee members unanimously agreed that healthy children with no contraindications should receive the quadrivalent live attenuated vaccine (LAIV) rather than an inactivated vaccine, provided that the LAIV is readily available.
"If the LAIV is not immediately available, [inactivated influenza vaccine (IIV)] should be used," the recommendation reads. "Vaccination should not be delayed in order to procure LAIV."
All children aged 6 months or older should receive influenza vaccine, according to the recommendation. "Influenza vaccine should not be delayed to procure a specific vaccine preparation if an appropriate one is already available."
The committee approved the recommendation after hearing an evidence review that found the LAIV to be "moderately" more effective for this age group than the inactivated vaccine was. Although the data are not plentiful, they suggest that it would probably prevent about 47 more lab-confirmed cases of flu per 1,000 children than the IIV, Dr. Lisa Grohskopf said
"The benefits appear to outweigh the potential harms, with modestly better efficacy," and no appreciable increase in adverse events associated with the live vaccine, said Dr. Grohskopf, who is a medical officer in the influenza division of the Centers for Disease Control and Prevention in Atlanta.
Her review was based on two studies – one published in 2006 and one in 2007. The earlier study was an open-label trial that randomized about 2,300 children aged 6-71 months to either the LAIV or an IIV. The much larger 2007 study randomized about 16,000 children aged 6-59 months to the two vaccine types or to matching placebos.
The pooled analysis was limited to healthy children aged 24-59 months who were without a history of asthma or wheezing. The primary endpoint of lab-confirmed influenza infection was significantly less likely among children who had received the LAIV (hazard ratio, 0.47). The secondary endpoint of influenza-associated otitis media was also significantly less likely to occur among those who got the LAIV (HR, 0.47).
Neither of the vaccines was associated with any medically significant wheezing or fever, Dr. Grohskopf said.
She also examined these same endpoints in children who had a history of wheezing or asthma. The LAIV significantly reduced the risk of lab-confirmed influenza in this group (HR, 0.53). There were no significantly increased risks of medically significant wheezing in the children, regardless of whether or not they had experienced wheezing within the past 12 months.
However, Dr. Grohskopf said, she and her colleagues didn’t feel that the studies were strong enough to make any initial recommendations to the committee about whether the LAIV could safely be used in children with a history of wheezing or asthma. The studies didn’t drill down into data on this group enough, and the data are now too old to be completely reliable for this population.
Asthma and wheezing are not strict contraindications to the LAIV, she said, but ACIP does not recommend it for children with those issues or for those with any chronic conditions that might predispose them to complications from the flu.
The committee also heard that the strains included in the 2014-2015 vaccine will be the same as last season’s. Because of this, children who received at least one dose of the 2013-2014 vaccine will only need one dose of this year’s vaccine to ensure immunity. "This is analogous to what happened in the 2011-2012 season, when the vaccine strains were unchanged from the previous season," Dr. Grohskopf said.
She also touched on the issue of cost. "We haven’t done a formal cost-effectiveness analysis," she said.
"It’s likely to be a very complex undertaking because of the large number of products out there. It will probably be a few years before that could be done."
The best estimates available now can be drawn from the 2013-2014 private sector costs associated with CDC’s Vaccines for Children program. "The cost of the LAIV intranasal vaccine is $22.70/dose, and for the inactivated vaccine, there is a broad range of variability, from $7.65-$14.81/dose for the trivalent to $14.90-$21.09/dose for the quadrivalent."
On Twitter @alz_gal
The live attenuated intranasal flu vaccine is the preferred method of immunizing healthy children aged 2-8 years against seasonal influenza, a federal panel has recommended.
At a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices, committee members unanimously agreed that healthy children with no contraindications should receive the quadrivalent live attenuated vaccine (LAIV) rather than an inactivated vaccine, provided that the LAIV is readily available.
"If the LAIV is not immediately available, [inactivated influenza vaccine (IIV)] should be used," the recommendation reads. "Vaccination should not be delayed in order to procure LAIV."
All children aged 6 months or older should receive influenza vaccine, according to the recommendation. "Influenza vaccine should not be delayed to procure a specific vaccine preparation if an appropriate one is already available."
The committee approved the recommendation after hearing an evidence review that found the LAIV to be "moderately" more effective for this age group than the inactivated vaccine was. Although the data are not plentiful, they suggest that it would probably prevent about 47 more lab-confirmed cases of flu per 1,000 children than the IIV, Dr. Lisa Grohskopf said
"The benefits appear to outweigh the potential harms, with modestly better efficacy," and no appreciable increase in adverse events associated with the live vaccine, said Dr. Grohskopf, who is a medical officer in the influenza division of the Centers for Disease Control and Prevention in Atlanta.
Her review was based on two studies – one published in 2006 and one in 2007. The earlier study was an open-label trial that randomized about 2,300 children aged 6-71 months to either the LAIV or an IIV. The much larger 2007 study randomized about 16,000 children aged 6-59 months to the two vaccine types or to matching placebos.
The pooled analysis was limited to healthy children aged 24-59 months who were without a history of asthma or wheezing. The primary endpoint of lab-confirmed influenza infection was significantly less likely among children who had received the LAIV (hazard ratio, 0.47). The secondary endpoint of influenza-associated otitis media was also significantly less likely to occur among those who got the LAIV (HR, 0.47).
Neither of the vaccines was associated with any medically significant wheezing or fever, Dr. Grohskopf said.
She also examined these same endpoints in children who had a history of wheezing or asthma. The LAIV significantly reduced the risk of lab-confirmed influenza in this group (HR, 0.53). There were no significantly increased risks of medically significant wheezing in the children, regardless of whether or not they had experienced wheezing within the past 12 months.
However, Dr. Grohskopf said, she and her colleagues didn’t feel that the studies were strong enough to make any initial recommendations to the committee about whether the LAIV could safely be used in children with a history of wheezing or asthma. The studies didn’t drill down into data on this group enough, and the data are now too old to be completely reliable for this population.
Asthma and wheezing are not strict contraindications to the LAIV, she said, but ACIP does not recommend it for children with those issues or for those with any chronic conditions that might predispose them to complications from the flu.
The committee also heard that the strains included in the 2014-2015 vaccine will be the same as last season’s. Because of this, children who received at least one dose of the 2013-2014 vaccine will only need one dose of this year’s vaccine to ensure immunity. "This is analogous to what happened in the 2011-2012 season, when the vaccine strains were unchanged from the previous season," Dr. Grohskopf said.
She also touched on the issue of cost. "We haven’t done a formal cost-effectiveness analysis," she said.
"It’s likely to be a very complex undertaking because of the large number of products out there. It will probably be a few years before that could be done."
The best estimates available now can be drawn from the 2013-2014 private sector costs associated with CDC’s Vaccines for Children program. "The cost of the LAIV intranasal vaccine is $22.70/dose, and for the inactivated vaccine, there is a broad range of variability, from $7.65-$14.81/dose for the trivalent to $14.90-$21.09/dose for the quadrivalent."
On Twitter @alz_gal
FROM AN ACIP MEETING