User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Pharmacist BP telemonitoring cut cardiovascular events, turned profit
PHILADELPHIA – A home blood pressure telemonitoring program featuring pharmacist management of patients with uncontrolled hypertension reduced cardiovascular events by half and was cost saving over the course of 5 years, even though the intervention ended after year 1, Karen L. Margolis, MD, reported at the American Heart Association scientific sessions.
“The return on investment was 126%. That means that for every dollar spent on the intervention, that dollar was recouped by $1.00 plus another $1.26,” explained Dr. Margolis, a general internist who serves as executive director for research at the HealthPartners Institute in Bloomington, Minn., and professor of medicine at the University of Minnesota, Minneapolis.
She presented 5-year follow-up data from the Hyperlink (Home Blood Pressure Telemonitoring and Case Management to Control Hypertension) study, a cluster randomized controlled trial involving 16 primary care clinics. Half of the clinics were randomized to the intervention, which entailed home blood pressure telemonitoring and pharmacist-led case management in collaboration with the primary care team. The other eight clinics provided usual care. The intervention portion of the trial, which lasted for 12 months, included 450 adults with uncontrolled hypertension as defined by repeated on-treatment blood pressure readings of 140/90 mm Hg or more. Participants’ baseline mean blood pressure was 148/85 mm Hg while on an average of one and a half antihypertensive drug classes. On average, pharmacists ended up adding one additional drug from a different antihypertensive drug class to achieve improved blood pressure control.
The details of the intervention and the short-term blood pressure results have previously been reported (JAMA. 2013 Jul 3;310[1]:46-56). Briefly, 6 months into the study, patients in the intervention arm averaged 11/6 mm Hg lower blood pressure than did the usual care controls. At 12 months – when the intervention ended – the between-group difference was similar at 10/5 mm Hg. At 18 months, the difference, while attenuated, remained significant at 7/3 mm Hg in favor of the intervention group. However, at 54 months, the intervention group’s advantage – a 3–mm Hg lower SBP and a 1–mm Hg lower DBP than in controls – was no longer significant.
The exciting new findings Dr. Margolis presented at the AHA scientific sessions focused on 5-year outcomes. Since HealthPartners is an integrated health care system, follow-up was essentially complete.
“None of the other telemetry studies I’m aware of have published anything on cardiovascular events. And we were somewhat surprised when we looked at our data to see fairly substantial differences in our primary outcome,” she noted.
That outcome was a composite of MI, stroke, heart failure, or cardiovascular death occurring over 5 years. The rate was 4.4% in the intervention group and nearly double at 8.6% in controls. That translated to a 51% relative risk reduction. The biggest difference was in stroke: 4 cases in the intervention arm, 12 in usual care controls.
The 5-year coronary revascularization rate was 5.3% in the intervention arm and 10.4% in controls, for a 52% relative risk reduction.
A major caveat regarding the Hyperlink trial was that, even at 450 patients and 5 years of follow-up, the study was underpowered to show significant differences in event rates, with P =.09 for the primary endpoint.
That being said, the financial results were striking. The intervention cost $1,511 per patient in 2017 U.S. dollars. The cost of treatment for major adverse cardiovascular events totaled $758,000 in the intervention group and $1,538,000 in usual care controls. That works out to $3,420 less per patient in the intervention arm. Offset by the cost of the intervention, that spells a net savings of $1,908 per patient achieved by implementing the year-long intervention. It’s a rare instance in health care of an intervention that actually makes money.
These results were unusual enough that Dr. Margolis and her coinvestigators decided to feed their wealth of SBP readings into a microsimulation model, which they ran 1,000 times. The model predicted – in light of the fact that patients in the intervention group were on average 2 years older than the controls were – that the expected reduction in the primary endpoint was 12% rather than the observed 51% relative risk reduction.
How to explain the discrepancy? The Hyperlink results could have been due to chance. Or it could be, Dr. Margolis surmised, that the pharmacists helped accomplish improvements in other cardiovascular risk factors, such as hyperlipidemia, smoking, or sedentary behavior. That’s unknown, since the investigators focused on changes in blood pressure only. Future studies of home telemonitoring and pharmacist case management of uncontrolled hypertension should be powered to detect significant differences in cardiovascular events and should track additional risk factors, she concluded.
She reported having no financial conflicts regarding the study.
SOURCE: Margolis KL. AHA 2019. Abstract MDP232.
PHILADELPHIA – A home blood pressure telemonitoring program featuring pharmacist management of patients with uncontrolled hypertension reduced cardiovascular events by half and was cost saving over the course of 5 years, even though the intervention ended after year 1, Karen L. Margolis, MD, reported at the American Heart Association scientific sessions.
“The return on investment was 126%. That means that for every dollar spent on the intervention, that dollar was recouped by $1.00 plus another $1.26,” explained Dr. Margolis, a general internist who serves as executive director for research at the HealthPartners Institute in Bloomington, Minn., and professor of medicine at the University of Minnesota, Minneapolis.
She presented 5-year follow-up data from the Hyperlink (Home Blood Pressure Telemonitoring and Case Management to Control Hypertension) study, a cluster randomized controlled trial involving 16 primary care clinics. Half of the clinics were randomized to the intervention, which entailed home blood pressure telemonitoring and pharmacist-led case management in collaboration with the primary care team. The other eight clinics provided usual care. The intervention portion of the trial, which lasted for 12 months, included 450 adults with uncontrolled hypertension as defined by repeated on-treatment blood pressure readings of 140/90 mm Hg or more. Participants’ baseline mean blood pressure was 148/85 mm Hg while on an average of one and a half antihypertensive drug classes. On average, pharmacists ended up adding one additional drug from a different antihypertensive drug class to achieve improved blood pressure control.
The details of the intervention and the short-term blood pressure results have previously been reported (JAMA. 2013 Jul 3;310[1]:46-56). Briefly, 6 months into the study, patients in the intervention arm averaged 11/6 mm Hg lower blood pressure than did the usual care controls. At 12 months – when the intervention ended – the between-group difference was similar at 10/5 mm Hg. At 18 months, the difference, while attenuated, remained significant at 7/3 mm Hg in favor of the intervention group. However, at 54 months, the intervention group’s advantage – a 3–mm Hg lower SBP and a 1–mm Hg lower DBP than in controls – was no longer significant.
The exciting new findings Dr. Margolis presented at the AHA scientific sessions focused on 5-year outcomes. Since HealthPartners is an integrated health care system, follow-up was essentially complete.
“None of the other telemetry studies I’m aware of have published anything on cardiovascular events. And we were somewhat surprised when we looked at our data to see fairly substantial differences in our primary outcome,” she noted.
That outcome was a composite of MI, stroke, heart failure, or cardiovascular death occurring over 5 years. The rate was 4.4% in the intervention group and nearly double at 8.6% in controls. That translated to a 51% relative risk reduction. The biggest difference was in stroke: 4 cases in the intervention arm, 12 in usual care controls.
The 5-year coronary revascularization rate was 5.3% in the intervention arm and 10.4% in controls, for a 52% relative risk reduction.
A major caveat regarding the Hyperlink trial was that, even at 450 patients and 5 years of follow-up, the study was underpowered to show significant differences in event rates, with P =.09 for the primary endpoint.
That being said, the financial results were striking. The intervention cost $1,511 per patient in 2017 U.S. dollars. The cost of treatment for major adverse cardiovascular events totaled $758,000 in the intervention group and $1,538,000 in usual care controls. That works out to $3,420 less per patient in the intervention arm. Offset by the cost of the intervention, that spells a net savings of $1,908 per patient achieved by implementing the year-long intervention. It’s a rare instance in health care of an intervention that actually makes money.
These results were unusual enough that Dr. Margolis and her coinvestigators decided to feed their wealth of SBP readings into a microsimulation model, which they ran 1,000 times. The model predicted – in light of the fact that patients in the intervention group were on average 2 years older than the controls were – that the expected reduction in the primary endpoint was 12% rather than the observed 51% relative risk reduction.
How to explain the discrepancy? The Hyperlink results could have been due to chance. Or it could be, Dr. Margolis surmised, that the pharmacists helped accomplish improvements in other cardiovascular risk factors, such as hyperlipidemia, smoking, or sedentary behavior. That’s unknown, since the investigators focused on changes in blood pressure only. Future studies of home telemonitoring and pharmacist case management of uncontrolled hypertension should be powered to detect significant differences in cardiovascular events and should track additional risk factors, she concluded.
She reported having no financial conflicts regarding the study.
SOURCE: Margolis KL. AHA 2019. Abstract MDP232.
PHILADELPHIA – A home blood pressure telemonitoring program featuring pharmacist management of patients with uncontrolled hypertension reduced cardiovascular events by half and was cost saving over the course of 5 years, even though the intervention ended after year 1, Karen L. Margolis, MD, reported at the American Heart Association scientific sessions.
“The return on investment was 126%. That means that for every dollar spent on the intervention, that dollar was recouped by $1.00 plus another $1.26,” explained Dr. Margolis, a general internist who serves as executive director for research at the HealthPartners Institute in Bloomington, Minn., and professor of medicine at the University of Minnesota, Minneapolis.
She presented 5-year follow-up data from the Hyperlink (Home Blood Pressure Telemonitoring and Case Management to Control Hypertension) study, a cluster randomized controlled trial involving 16 primary care clinics. Half of the clinics were randomized to the intervention, which entailed home blood pressure telemonitoring and pharmacist-led case management in collaboration with the primary care team. The other eight clinics provided usual care. The intervention portion of the trial, which lasted for 12 months, included 450 adults with uncontrolled hypertension as defined by repeated on-treatment blood pressure readings of 140/90 mm Hg or more. Participants’ baseline mean blood pressure was 148/85 mm Hg while on an average of one and a half antihypertensive drug classes. On average, pharmacists ended up adding one additional drug from a different antihypertensive drug class to achieve improved blood pressure control.
The details of the intervention and the short-term blood pressure results have previously been reported (JAMA. 2013 Jul 3;310[1]:46-56). Briefly, 6 months into the study, patients in the intervention arm averaged 11/6 mm Hg lower blood pressure than did the usual care controls. At 12 months – when the intervention ended – the between-group difference was similar at 10/5 mm Hg. At 18 months, the difference, while attenuated, remained significant at 7/3 mm Hg in favor of the intervention group. However, at 54 months, the intervention group’s advantage – a 3–mm Hg lower SBP and a 1–mm Hg lower DBP than in controls – was no longer significant.
The exciting new findings Dr. Margolis presented at the AHA scientific sessions focused on 5-year outcomes. Since HealthPartners is an integrated health care system, follow-up was essentially complete.
“None of the other telemetry studies I’m aware of have published anything on cardiovascular events. And we were somewhat surprised when we looked at our data to see fairly substantial differences in our primary outcome,” she noted.
That outcome was a composite of MI, stroke, heart failure, or cardiovascular death occurring over 5 years. The rate was 4.4% in the intervention group and nearly double at 8.6% in controls. That translated to a 51% relative risk reduction. The biggest difference was in stroke: 4 cases in the intervention arm, 12 in usual care controls.
The 5-year coronary revascularization rate was 5.3% in the intervention arm and 10.4% in controls, for a 52% relative risk reduction.
A major caveat regarding the Hyperlink trial was that, even at 450 patients and 5 years of follow-up, the study was underpowered to show significant differences in event rates, with P =.09 for the primary endpoint.
That being said, the financial results were striking. The intervention cost $1,511 per patient in 2017 U.S. dollars. The cost of treatment for major adverse cardiovascular events totaled $758,000 in the intervention group and $1,538,000 in usual care controls. That works out to $3,420 less per patient in the intervention arm. Offset by the cost of the intervention, that spells a net savings of $1,908 per patient achieved by implementing the year-long intervention. It’s a rare instance in health care of an intervention that actually makes money.
These results were unusual enough that Dr. Margolis and her coinvestigators decided to feed their wealth of SBP readings into a microsimulation model, which they ran 1,000 times. The model predicted – in light of the fact that patients in the intervention group were on average 2 years older than the controls were – that the expected reduction in the primary endpoint was 12% rather than the observed 51% relative risk reduction.
How to explain the discrepancy? The Hyperlink results could have been due to chance. Or it could be, Dr. Margolis surmised, that the pharmacists helped accomplish improvements in other cardiovascular risk factors, such as hyperlipidemia, smoking, or sedentary behavior. That’s unknown, since the investigators focused on changes in blood pressure only. Future studies of home telemonitoring and pharmacist case management of uncontrolled hypertension should be powered to detect significant differences in cardiovascular events and should track additional risk factors, she concluded.
She reported having no financial conflicts regarding the study.
SOURCE: Margolis KL. AHA 2019. Abstract MDP232.
REPORTING FROM THE AHA SCIENTIFIC SESSIONS
ID consult for Candida bloodstream infections can reduce mortality risk
findings from a large retrospective study suggest.
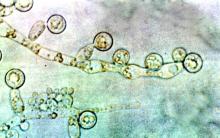
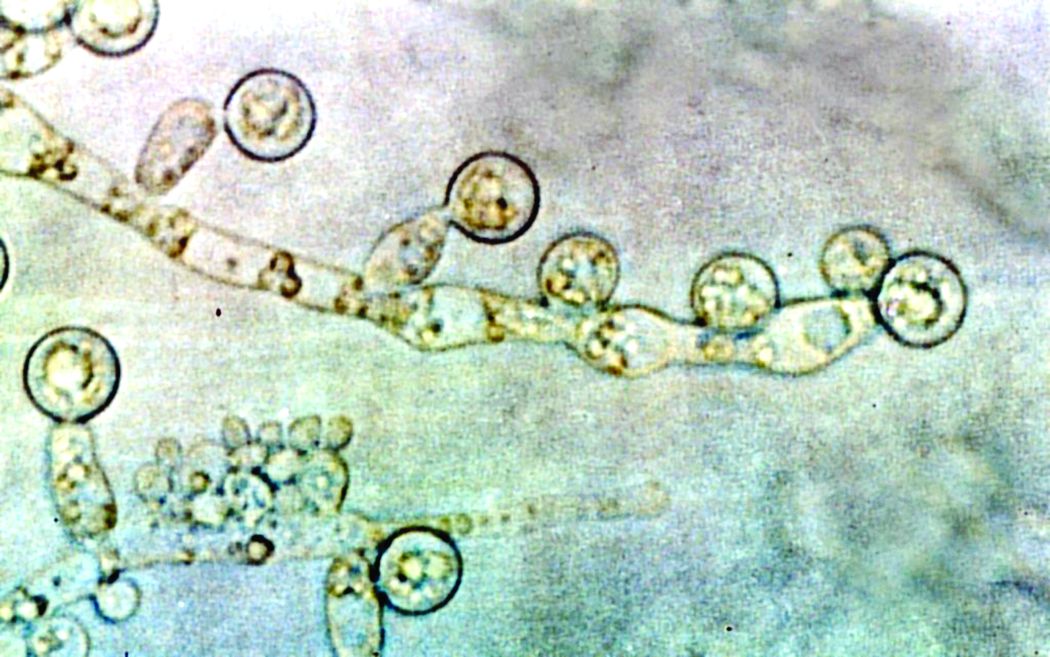
Mortality attributable to Candida bloodstream infection ranges between 15% and 47%, and delay in initiation of appropriate treatment has been associated with increased mortality. Previous small studies showed that ID consultation has conferred benefits to patients with Candida bloodstream infections. Carlos Mejia-Chew, MD, and colleagues from Washington University, St. Louis, sought to explore this further by performing a retrospective, single-center cohort study of 1,691 patients aged 18 years or older with Candida bloodstream infection from 2002 to 2015. They analyzed demographics, comorbidities, predisposing factors, all-cause mortality, antifungal use, central-line removal, and ophthalmological and echocardiographic evaluation in order to compare 90-day all-cause mortality between individuals with and without an ID consultation.
They found that those patients who received an ID consult for a Candida bloodstream infection had a significantly lower 90-day mortality rate than did those who did not (29% vs. 51%).
With a model using inverse weighting by the propensity score, they found that ID consultation was associated with a hazard ratio of 0.81 for mortality (95% confidence interval, 0.73-0.91; P less than .0001). In the ID consultation group, the median duration of antifungal therapy was significantly longer (18 vs. 14 days; P less than .0001); central-line removal was significantly more common (76% vs. 59%; P less than .0001); echocardiography use was more frequent (57% vs. 33%; P less than .0001); and ophthalmological examinations were performed more often (53% vs. 17%; P less than .0001). Importantly, fewer patients in the ID consultation group were untreated (2% vs. 14%; P less than .0001).
In an accompanying commentary, Katrien Lagrou, MD, and Eric Van Wijngaerden, MD, of the department of microbiology, immunology and transplantation, University Hospitals Leuven (Belgium) stated: “We think that the high proportion of patients (14%) with a Candida bloodstream infection who did not receive any antifungal treatment and did not have an infectious disease consultation is a particularly alarming finding. ... Ninety-day mortality in these untreated patients was high (67%).”
“We believe every hospital should have an expert management strategy addressing all individual cases of candidaemia. The need for such expert management should be incorporated in all future candidaemia management guidelines,” they concluded.
The study was funded by the Astellas Global Development Pharma, the Washington University Institute of Clinical and Translational Sciences, and the Agency for Healthcare Research and Quality. Several of the authors had financial connections to Astellas Global Development or other pharmaceutical companies. Dr. Lagrou and Dr. Van Wijngaerden both reported receiving personal fees and nonfinancial support from a number of pharmaceutical companies, but all outside the scope of the study.
SOURCE: Mejia-Chew C et al. Lancet Infect Dis. 2019;19:1336-44.
findings from a large retrospective study suggest.
Mortality attributable to Candida bloodstream infection ranges between 15% and 47%, and delay in initiation of appropriate treatment has been associated with increased mortality. Previous small studies showed that ID consultation has conferred benefits to patients with Candida bloodstream infections. Carlos Mejia-Chew, MD, and colleagues from Washington University, St. Louis, sought to explore this further by performing a retrospective, single-center cohort study of 1,691 patients aged 18 years or older with Candida bloodstream infection from 2002 to 2015. They analyzed demographics, comorbidities, predisposing factors, all-cause mortality, antifungal use, central-line removal, and ophthalmological and echocardiographic evaluation in order to compare 90-day all-cause mortality between individuals with and without an ID consultation.
They found that those patients who received an ID consult for a Candida bloodstream infection had a significantly lower 90-day mortality rate than did those who did not (29% vs. 51%).
With a model using inverse weighting by the propensity score, they found that ID consultation was associated with a hazard ratio of 0.81 for mortality (95% confidence interval, 0.73-0.91; P less than .0001). In the ID consultation group, the median duration of antifungal therapy was significantly longer (18 vs. 14 days; P less than .0001); central-line removal was significantly more common (76% vs. 59%; P less than .0001); echocardiography use was more frequent (57% vs. 33%; P less than .0001); and ophthalmological examinations were performed more often (53% vs. 17%; P less than .0001). Importantly, fewer patients in the ID consultation group were untreated (2% vs. 14%; P less than .0001).
In an accompanying commentary, Katrien Lagrou, MD, and Eric Van Wijngaerden, MD, of the department of microbiology, immunology and transplantation, University Hospitals Leuven (Belgium) stated: “We think that the high proportion of patients (14%) with a Candida bloodstream infection who did not receive any antifungal treatment and did not have an infectious disease consultation is a particularly alarming finding. ... Ninety-day mortality in these untreated patients was high (67%).”
“We believe every hospital should have an expert management strategy addressing all individual cases of candidaemia. The need for such expert management should be incorporated in all future candidaemia management guidelines,” they concluded.
The study was funded by the Astellas Global Development Pharma, the Washington University Institute of Clinical and Translational Sciences, and the Agency for Healthcare Research and Quality. Several of the authors had financial connections to Astellas Global Development or other pharmaceutical companies. Dr. Lagrou and Dr. Van Wijngaerden both reported receiving personal fees and nonfinancial support from a number of pharmaceutical companies, but all outside the scope of the study.
SOURCE: Mejia-Chew C et al. Lancet Infect Dis. 2019;19:1336-44.
findings from a large retrospective study suggest.
Mortality attributable to Candida bloodstream infection ranges between 15% and 47%, and delay in initiation of appropriate treatment has been associated with increased mortality. Previous small studies showed that ID consultation has conferred benefits to patients with Candida bloodstream infections. Carlos Mejia-Chew, MD, and colleagues from Washington University, St. Louis, sought to explore this further by performing a retrospective, single-center cohort study of 1,691 patients aged 18 years or older with Candida bloodstream infection from 2002 to 2015. They analyzed demographics, comorbidities, predisposing factors, all-cause mortality, antifungal use, central-line removal, and ophthalmological and echocardiographic evaluation in order to compare 90-day all-cause mortality between individuals with and without an ID consultation.
They found that those patients who received an ID consult for a Candida bloodstream infection had a significantly lower 90-day mortality rate than did those who did not (29% vs. 51%).
With a model using inverse weighting by the propensity score, they found that ID consultation was associated with a hazard ratio of 0.81 for mortality (95% confidence interval, 0.73-0.91; P less than .0001). In the ID consultation group, the median duration of antifungal therapy was significantly longer (18 vs. 14 days; P less than .0001); central-line removal was significantly more common (76% vs. 59%; P less than .0001); echocardiography use was more frequent (57% vs. 33%; P less than .0001); and ophthalmological examinations were performed more often (53% vs. 17%; P less than .0001). Importantly, fewer patients in the ID consultation group were untreated (2% vs. 14%; P less than .0001).
In an accompanying commentary, Katrien Lagrou, MD, and Eric Van Wijngaerden, MD, of the department of microbiology, immunology and transplantation, University Hospitals Leuven (Belgium) stated: “We think that the high proportion of patients (14%) with a Candida bloodstream infection who did not receive any antifungal treatment and did not have an infectious disease consultation is a particularly alarming finding. ... Ninety-day mortality in these untreated patients was high (67%).”
“We believe every hospital should have an expert management strategy addressing all individual cases of candidaemia. The need for such expert management should be incorporated in all future candidaemia management guidelines,” they concluded.
The study was funded by the Astellas Global Development Pharma, the Washington University Institute of Clinical and Translational Sciences, and the Agency for Healthcare Research and Quality. Several of the authors had financial connections to Astellas Global Development or other pharmaceutical companies. Dr. Lagrou and Dr. Van Wijngaerden both reported receiving personal fees and nonfinancial support from a number of pharmaceutical companies, but all outside the scope of the study.
SOURCE: Mejia-Chew C et al. Lancet Infect Dis. 2019;19:1336-44.
FROM LANCET: INFECTIOUS DISEASES
Adolescent alcohol, opioid misuse linked to risky behaviors
Binge drinking and misuse of opioids led to risky behavior during adolescence, two studies from the journal Pediatrics highlighted. And the binge drinking in high school may predict risky driving behaviors up to 4 years after high school.
Federico E. Vaca, MD, of the developmental neurocognitive driving simulation research center at Yale University, New Haven, Conn., and colleagues examined the associations between risky driving behaviors and binge drinking of 2,785 adolescents in the nationally representative, longitudinal NEXT Generation Health Study. The researchers studied the effects of binge drinking on driving while impaired (DWI), riding with an impaired driver (RWI), blackouts, extreme binge drinking, and risky driving.
The adolescents were studied across seven waves, with Wave 1 beginning in the 2009-2010 school year (10th grade; mean age, 16 years), and data extended up to 4 years after high school. Of all adolescents enrolled, 91% completed Wave 1, 88% completed Wave 2, 86% completed Wave 3 (12th grade), 78% completed Wave 4, 79% completed Wave 5, 84% completed Wave 6, and 83% completed Wave 7 (4 years after leaving high school) of the study.
High school binge drinking predicts later risky behavior
About one-quarter of adolescents reported binge drinking in Waves 1-3, with an incidence of 27% in Wave 1, 24% in Wave 2, and 27% in Wave 3. Adolescents who reported binge drinking in Wave 3 had a higher likelihood of DWI in subsequent waves, with nearly six times higher odds in Wave 5 and more than twice as likely in Wave 7, researchers said. Binge drinking in Wave 3 also was associated with greater than four times higher odds of RWI in Wave 4, and more than two and a half times higher odds of RWI in Wave 7. Among adolescents who reported binge drinking across 3 years in high school, there was a higher likelihood of extreme binge drinking in Wave 7, and higher likelihood of risky driving after graduating.
Impact of parental knowledge of drinking
in some waves. Father monitoring knowledge of drinking in Waves 1-3 lowered the odds of DWI by 30% in Wave 5 and 20% in Wave 6, while also lowering the odds of RWI in Wave 4 and Wave 7 by 20%.
Mother knowledge of drinking in Waves 1-3 was associated with 60% lower odds of DWI in Wave 4, but did not lower odds in any wave for RWI.
Overall, parental support for not drinking lowered odds for DWI by 40% in Waves 4 and 5, and by 30% in Wave 7 while also lowering odds of RWI in Wave 4 by 20%.
The results are consistent with other studies examining risky driving behavior and binge drinking in adolescent populations, but researchers noted that “to an important but limited extent, parental practices while the teenager is in high school may protect against DWI, RWI, and blackouts as adolescents move into early adulthood.”
“Our findings are relevant to prevention programs that seek to incorporate alcohol screening with intentional inquiry about binge drinking. Moreover, our results may be instructive to programs that seek to leverage facets of parental practices to reduce health-risk contexts for youth,” Dr. Vaca and colleagues concluded. “Such prevention activities coupled with strengthening of policies and practices reducing adolescents’ access to alcohol could reduce later major alcohol-related health-risk behaviors and their consequences.”
Opioid misuse and risky behavior
In a second study, Devika Bhatia, MD, of the University of Colorado at Denver, Aurora, and colleagues examined opioid misuse in a nationally-representative sample of 14,765 adolescents from the Centers for Disease Control and Prevention’s 2017 Youth Risk Behavior Surveillance Survey. The researchers measured opioid misuse by categorizing adolescents into groups based on whether they had ever misused prescription opioids and whether they had engaged in risky driving behavior, violent behavior, risky sexual behavior, had a history of substance abuse, or attempted suicide.
Dr. Bhatia and colleagues found 14% of adolescents in the study reported misusing opioids, with an overrepresentation of 17-year-old and 18-year-old participants reporting opioid misuse (P less than .0001). there were no statistically significant difference between those who misused opioids and those who did not in terms of race, ethnicity, or sex.
Those adolescents who reported misusing opioids were 2.8 times more likely to not use a seatbelt; were 2.8 times more likely to have RWI; were 5.8 times more likely to have DWI; or 2.3 times more likely to have texted or emailed while driving. In each of these cases, P was less than .0001.
Adolescents who misused opioids also had significantly increased odds of engaging in risky sexual behaviors such as having sex before 13 years (3.9 times); having sex with four or more partners (4.8 times); using substances before sex (3.6 times); and not using a condom before sex (2.0 times). In each of these cases, P was less than .0001.
Additionally, adolescents in this category were between 5.4 times and 22.3 times more likely to use other substances (P less than .0001 for 10 variables); 4.9 times more likely to have attempted suicide (P less than .0001); or more likely to have engaged in violent behavior such as getting into physical fights (4.0 times), carrying a weapon (3.4 times) or a gun (5.1 times) within the last 30 days. In the four latter cases, P was less than .0001.
“With the ongoing opioid epidemic, pediatricians and child psychiatrists are likely to be more attuned to opioid misuse in their patients,” Dr. Bhatia and colleagues concluded. “If youth are screening positive for opioid misuse, pediatricians, nurses, social workers, child psychiatrists, and other providers assessing adolescents may have a new, broad range of other risky behaviors for which to screen regardless of the direction of the association.”
Substance use screening for treating substance use disorder traditionally has been is provided by a specialist, Jessica A. Kulak, PhD, MPH, said in an interview. “However, integration of care services may help to change societal norms around problematic substance use – both by decreasing stigma associated with substance use, as well as increasing clinicians’ preparedness, knowledge, and confidence in preventing and intervening on adolescents’ substance experimentation and use.” She recommended that clinicians in primary care improve their training by using the Substance Abuse and Mental Health Services Administration’s Screening, Brief Intervention, and Referral to Treatment program, which is available as a free online course.
Confidentiality is important in adolescent health, said Dr. Kulak, who is an assistant professor in the department of health, nutrition, and dietetics at State University of New York at Buffalo. “When discussing sensitive topics, such as binge drinking and opioid misuse, adolescents may fear that these or other risky activities may be disclosed to parents or law enforcement officials. Therefore, adolescent health providers should be aware of local, state, and federal laws pertaining to the confidentiality of minors.”
She added, “adolescents are often susceptible to others’ influences, so having open communication and support from a trusted adult – be it a parent or clinician – may also be protective against risky behaviors.”
The study by Vaca et al. was funded by the National Institutes of Health with support from the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development; the National Heart, Lung, and Blood Institute; the National Institute on Alcohol Abuse and Alcoholism; the National Institute on Drug Abuse; and the Maternal and Child Health Bureau of the Health Resources and Services Administration. The study by Bhatia et al. had no external funding. The authors from both studies reported no relevant financial disclosures. Dr. Kulak said she had no financial disclosures or other conflicts of interest.
SOURCE: Vaca FE et al. Pediatrics. 2020; doi: 10.1542/peds.2018-4095. Bhatia D et al. Pediatrics. 2020; doi: 10.1542/peds.2019-2470.
These newly published reports indicate the high prevalence of risky behaviors and their associations – cross-sectionally and longitudinally – with major threats to adolescent health – so asking about alcohol use, opioid misuse, and associated health risks is truly “in the lane” of clinicians, school professionals, and parents who see and care about adolescents.
At this point, I think it’s incontrovertible that clinicians should screen adolescents to learn about their physical, emotional, and behavioral health. And they should seek opportunities for professional training, skills development, and expansion of their professional networks so they are able to address – individually or collaboratively via referrals – the behavioral and psychosocial health risks of their patients.
The good news is that there is growing awareness of the importance of using validated screening tools to identify patient behavioral health risks – including those pertaining to adolescent and young adult alcohol use and opioid misuse. “Best practice” dictates that screening approaches rely on asking questions using structured tools; intuition and “just winging it” are not effective or reliable for identifying patient behavior. Forward-looking clinics and practices could be asking patients to report about health behaviors in the waiting room (on a computer tablet, for example), or even remotely (using a secure app or data collection tool) in advance of a visit. Asking should be periodic – since behaviors can change fairly rapidly among young people. The benefit is that patient-reported information can be processed in advance to cue clinician follow-up and intervention. And youth tend to share more about their behaviors when they are asked electronically, rather than face to face. Intelligent screens can provide near real-time estimation of risk – to support in-office brief intervention tailored to the risk level of a young person or to trigger follow-up.
These studies indicate that binge alcohol use and misuse of prescription opioids among adolescents are real, pervasive, and deserving of our considered attention. There is no magic bullet. However busy clinicians may have a significant role to play in identifying and addressing these problems.
Elissa Weitzman, ScD, MSc, is an associate professor of pediatrics at Harvard Medical School, Boston, and an associate scientist based in adolescent/young adult medicine and the computational health informatics program at Boston Children’s Hospital. She was asked to comment on the articles by Vaca et al. and Bhatia et al. Dr. Weitzman said she had no relevant financial disclosures.
These newly published reports indicate the high prevalence of risky behaviors and their associations – cross-sectionally and longitudinally – with major threats to adolescent health – so asking about alcohol use, opioid misuse, and associated health risks is truly “in the lane” of clinicians, school professionals, and parents who see and care about adolescents.
At this point, I think it’s incontrovertible that clinicians should screen adolescents to learn about their physical, emotional, and behavioral health. And they should seek opportunities for professional training, skills development, and expansion of their professional networks so they are able to address – individually or collaboratively via referrals – the behavioral and psychosocial health risks of their patients.
The good news is that there is growing awareness of the importance of using validated screening tools to identify patient behavioral health risks – including those pertaining to adolescent and young adult alcohol use and opioid misuse. “Best practice” dictates that screening approaches rely on asking questions using structured tools; intuition and “just winging it” are not effective or reliable for identifying patient behavior. Forward-looking clinics and practices could be asking patients to report about health behaviors in the waiting room (on a computer tablet, for example), or even remotely (using a secure app or data collection tool) in advance of a visit. Asking should be periodic – since behaviors can change fairly rapidly among young people. The benefit is that patient-reported information can be processed in advance to cue clinician follow-up and intervention. And youth tend to share more about their behaviors when they are asked electronically, rather than face to face. Intelligent screens can provide near real-time estimation of risk – to support in-office brief intervention tailored to the risk level of a young person or to trigger follow-up.
These studies indicate that binge alcohol use and misuse of prescription opioids among adolescents are real, pervasive, and deserving of our considered attention. There is no magic bullet. However busy clinicians may have a significant role to play in identifying and addressing these problems.
Elissa Weitzman, ScD, MSc, is an associate professor of pediatrics at Harvard Medical School, Boston, and an associate scientist based in adolescent/young adult medicine and the computational health informatics program at Boston Children’s Hospital. She was asked to comment on the articles by Vaca et al. and Bhatia et al. Dr. Weitzman said she had no relevant financial disclosures.
These newly published reports indicate the high prevalence of risky behaviors and their associations – cross-sectionally and longitudinally – with major threats to adolescent health – so asking about alcohol use, opioid misuse, and associated health risks is truly “in the lane” of clinicians, school professionals, and parents who see and care about adolescents.
At this point, I think it’s incontrovertible that clinicians should screen adolescents to learn about their physical, emotional, and behavioral health. And they should seek opportunities for professional training, skills development, and expansion of their professional networks so they are able to address – individually or collaboratively via referrals – the behavioral and psychosocial health risks of their patients.
The good news is that there is growing awareness of the importance of using validated screening tools to identify patient behavioral health risks – including those pertaining to adolescent and young adult alcohol use and opioid misuse. “Best practice” dictates that screening approaches rely on asking questions using structured tools; intuition and “just winging it” are not effective or reliable for identifying patient behavior. Forward-looking clinics and practices could be asking patients to report about health behaviors in the waiting room (on a computer tablet, for example), or even remotely (using a secure app or data collection tool) in advance of a visit. Asking should be periodic – since behaviors can change fairly rapidly among young people. The benefit is that patient-reported information can be processed in advance to cue clinician follow-up and intervention. And youth tend to share more about their behaviors when they are asked electronically, rather than face to face. Intelligent screens can provide near real-time estimation of risk – to support in-office brief intervention tailored to the risk level of a young person or to trigger follow-up.
These studies indicate that binge alcohol use and misuse of prescription opioids among adolescents are real, pervasive, and deserving of our considered attention. There is no magic bullet. However busy clinicians may have a significant role to play in identifying and addressing these problems.
Elissa Weitzman, ScD, MSc, is an associate professor of pediatrics at Harvard Medical School, Boston, and an associate scientist based in adolescent/young adult medicine and the computational health informatics program at Boston Children’s Hospital. She was asked to comment on the articles by Vaca et al. and Bhatia et al. Dr. Weitzman said she had no relevant financial disclosures.
Binge drinking and misuse of opioids led to risky behavior during adolescence, two studies from the journal Pediatrics highlighted. And the binge drinking in high school may predict risky driving behaviors up to 4 years after high school.
Federico E. Vaca, MD, of the developmental neurocognitive driving simulation research center at Yale University, New Haven, Conn., and colleagues examined the associations between risky driving behaviors and binge drinking of 2,785 adolescents in the nationally representative, longitudinal NEXT Generation Health Study. The researchers studied the effects of binge drinking on driving while impaired (DWI), riding with an impaired driver (RWI), blackouts, extreme binge drinking, and risky driving.
The adolescents were studied across seven waves, with Wave 1 beginning in the 2009-2010 school year (10th grade; mean age, 16 years), and data extended up to 4 years after high school. Of all adolescents enrolled, 91% completed Wave 1, 88% completed Wave 2, 86% completed Wave 3 (12th grade), 78% completed Wave 4, 79% completed Wave 5, 84% completed Wave 6, and 83% completed Wave 7 (4 years after leaving high school) of the study.
High school binge drinking predicts later risky behavior
About one-quarter of adolescents reported binge drinking in Waves 1-3, with an incidence of 27% in Wave 1, 24% in Wave 2, and 27% in Wave 3. Adolescents who reported binge drinking in Wave 3 had a higher likelihood of DWI in subsequent waves, with nearly six times higher odds in Wave 5 and more than twice as likely in Wave 7, researchers said. Binge drinking in Wave 3 also was associated with greater than four times higher odds of RWI in Wave 4, and more than two and a half times higher odds of RWI in Wave 7. Among adolescents who reported binge drinking across 3 years in high school, there was a higher likelihood of extreme binge drinking in Wave 7, and higher likelihood of risky driving after graduating.
Impact of parental knowledge of drinking
in some waves. Father monitoring knowledge of drinking in Waves 1-3 lowered the odds of DWI by 30% in Wave 5 and 20% in Wave 6, while also lowering the odds of RWI in Wave 4 and Wave 7 by 20%.
Mother knowledge of drinking in Waves 1-3 was associated with 60% lower odds of DWI in Wave 4, but did not lower odds in any wave for RWI.
Overall, parental support for not drinking lowered odds for DWI by 40% in Waves 4 and 5, and by 30% in Wave 7 while also lowering odds of RWI in Wave 4 by 20%.
The results are consistent with other studies examining risky driving behavior and binge drinking in adolescent populations, but researchers noted that “to an important but limited extent, parental practices while the teenager is in high school may protect against DWI, RWI, and blackouts as adolescents move into early adulthood.”
“Our findings are relevant to prevention programs that seek to incorporate alcohol screening with intentional inquiry about binge drinking. Moreover, our results may be instructive to programs that seek to leverage facets of parental practices to reduce health-risk contexts for youth,” Dr. Vaca and colleagues concluded. “Such prevention activities coupled with strengthening of policies and practices reducing adolescents’ access to alcohol could reduce later major alcohol-related health-risk behaviors and their consequences.”
Opioid misuse and risky behavior
In a second study, Devika Bhatia, MD, of the University of Colorado at Denver, Aurora, and colleagues examined opioid misuse in a nationally-representative sample of 14,765 adolescents from the Centers for Disease Control and Prevention’s 2017 Youth Risk Behavior Surveillance Survey. The researchers measured opioid misuse by categorizing adolescents into groups based on whether they had ever misused prescription opioids and whether they had engaged in risky driving behavior, violent behavior, risky sexual behavior, had a history of substance abuse, or attempted suicide.
Dr. Bhatia and colleagues found 14% of adolescents in the study reported misusing opioids, with an overrepresentation of 17-year-old and 18-year-old participants reporting opioid misuse (P less than .0001). there were no statistically significant difference between those who misused opioids and those who did not in terms of race, ethnicity, or sex.
Those adolescents who reported misusing opioids were 2.8 times more likely to not use a seatbelt; were 2.8 times more likely to have RWI; were 5.8 times more likely to have DWI; or 2.3 times more likely to have texted or emailed while driving. In each of these cases, P was less than .0001.
Adolescents who misused opioids also had significantly increased odds of engaging in risky sexual behaviors such as having sex before 13 years (3.9 times); having sex with four or more partners (4.8 times); using substances before sex (3.6 times); and not using a condom before sex (2.0 times). In each of these cases, P was less than .0001.
Additionally, adolescents in this category were between 5.4 times and 22.3 times more likely to use other substances (P less than .0001 for 10 variables); 4.9 times more likely to have attempted suicide (P less than .0001); or more likely to have engaged in violent behavior such as getting into physical fights (4.0 times), carrying a weapon (3.4 times) or a gun (5.1 times) within the last 30 days. In the four latter cases, P was less than .0001.
“With the ongoing opioid epidemic, pediatricians and child psychiatrists are likely to be more attuned to opioid misuse in their patients,” Dr. Bhatia and colleagues concluded. “If youth are screening positive for opioid misuse, pediatricians, nurses, social workers, child psychiatrists, and other providers assessing adolescents may have a new, broad range of other risky behaviors for which to screen regardless of the direction of the association.”
Substance use screening for treating substance use disorder traditionally has been is provided by a specialist, Jessica A. Kulak, PhD, MPH, said in an interview. “However, integration of care services may help to change societal norms around problematic substance use – both by decreasing stigma associated with substance use, as well as increasing clinicians’ preparedness, knowledge, and confidence in preventing and intervening on adolescents’ substance experimentation and use.” She recommended that clinicians in primary care improve their training by using the Substance Abuse and Mental Health Services Administration’s Screening, Brief Intervention, and Referral to Treatment program, which is available as a free online course.
Confidentiality is important in adolescent health, said Dr. Kulak, who is an assistant professor in the department of health, nutrition, and dietetics at State University of New York at Buffalo. “When discussing sensitive topics, such as binge drinking and opioid misuse, adolescents may fear that these or other risky activities may be disclosed to parents or law enforcement officials. Therefore, adolescent health providers should be aware of local, state, and federal laws pertaining to the confidentiality of minors.”
She added, “adolescents are often susceptible to others’ influences, so having open communication and support from a trusted adult – be it a parent or clinician – may also be protective against risky behaviors.”
The study by Vaca et al. was funded by the National Institutes of Health with support from the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development; the National Heart, Lung, and Blood Institute; the National Institute on Alcohol Abuse and Alcoholism; the National Institute on Drug Abuse; and the Maternal and Child Health Bureau of the Health Resources and Services Administration. The study by Bhatia et al. had no external funding. The authors from both studies reported no relevant financial disclosures. Dr. Kulak said she had no financial disclosures or other conflicts of interest.
SOURCE: Vaca FE et al. Pediatrics. 2020; doi: 10.1542/peds.2018-4095. Bhatia D et al. Pediatrics. 2020; doi: 10.1542/peds.2019-2470.
Binge drinking and misuse of opioids led to risky behavior during adolescence, two studies from the journal Pediatrics highlighted. And the binge drinking in high school may predict risky driving behaviors up to 4 years after high school.
Federico E. Vaca, MD, of the developmental neurocognitive driving simulation research center at Yale University, New Haven, Conn., and colleagues examined the associations between risky driving behaviors and binge drinking of 2,785 adolescents in the nationally representative, longitudinal NEXT Generation Health Study. The researchers studied the effects of binge drinking on driving while impaired (DWI), riding with an impaired driver (RWI), blackouts, extreme binge drinking, and risky driving.
The adolescents were studied across seven waves, with Wave 1 beginning in the 2009-2010 school year (10th grade; mean age, 16 years), and data extended up to 4 years after high school. Of all adolescents enrolled, 91% completed Wave 1, 88% completed Wave 2, 86% completed Wave 3 (12th grade), 78% completed Wave 4, 79% completed Wave 5, 84% completed Wave 6, and 83% completed Wave 7 (4 years after leaving high school) of the study.
High school binge drinking predicts later risky behavior
About one-quarter of adolescents reported binge drinking in Waves 1-3, with an incidence of 27% in Wave 1, 24% in Wave 2, and 27% in Wave 3. Adolescents who reported binge drinking in Wave 3 had a higher likelihood of DWI in subsequent waves, with nearly six times higher odds in Wave 5 and more than twice as likely in Wave 7, researchers said. Binge drinking in Wave 3 also was associated with greater than four times higher odds of RWI in Wave 4, and more than two and a half times higher odds of RWI in Wave 7. Among adolescents who reported binge drinking across 3 years in high school, there was a higher likelihood of extreme binge drinking in Wave 7, and higher likelihood of risky driving after graduating.
Impact of parental knowledge of drinking
in some waves. Father monitoring knowledge of drinking in Waves 1-3 lowered the odds of DWI by 30% in Wave 5 and 20% in Wave 6, while also lowering the odds of RWI in Wave 4 and Wave 7 by 20%.
Mother knowledge of drinking in Waves 1-3 was associated with 60% lower odds of DWI in Wave 4, but did not lower odds in any wave for RWI.
Overall, parental support for not drinking lowered odds for DWI by 40% in Waves 4 and 5, and by 30% in Wave 7 while also lowering odds of RWI in Wave 4 by 20%.
The results are consistent with other studies examining risky driving behavior and binge drinking in adolescent populations, but researchers noted that “to an important but limited extent, parental practices while the teenager is in high school may protect against DWI, RWI, and blackouts as adolescents move into early adulthood.”
“Our findings are relevant to prevention programs that seek to incorporate alcohol screening with intentional inquiry about binge drinking. Moreover, our results may be instructive to programs that seek to leverage facets of parental practices to reduce health-risk contexts for youth,” Dr. Vaca and colleagues concluded. “Such prevention activities coupled with strengthening of policies and practices reducing adolescents’ access to alcohol could reduce later major alcohol-related health-risk behaviors and their consequences.”
Opioid misuse and risky behavior
In a second study, Devika Bhatia, MD, of the University of Colorado at Denver, Aurora, and colleagues examined opioid misuse in a nationally-representative sample of 14,765 adolescents from the Centers for Disease Control and Prevention’s 2017 Youth Risk Behavior Surveillance Survey. The researchers measured opioid misuse by categorizing adolescents into groups based on whether they had ever misused prescription opioids and whether they had engaged in risky driving behavior, violent behavior, risky sexual behavior, had a history of substance abuse, or attempted suicide.
Dr. Bhatia and colleagues found 14% of adolescents in the study reported misusing opioids, with an overrepresentation of 17-year-old and 18-year-old participants reporting opioid misuse (P less than .0001). there were no statistically significant difference between those who misused opioids and those who did not in terms of race, ethnicity, or sex.
Those adolescents who reported misusing opioids were 2.8 times more likely to not use a seatbelt; were 2.8 times more likely to have RWI; were 5.8 times more likely to have DWI; or 2.3 times more likely to have texted or emailed while driving. In each of these cases, P was less than .0001.
Adolescents who misused opioids also had significantly increased odds of engaging in risky sexual behaviors such as having sex before 13 years (3.9 times); having sex with four or more partners (4.8 times); using substances before sex (3.6 times); and not using a condom before sex (2.0 times). In each of these cases, P was less than .0001.
Additionally, adolescents in this category were between 5.4 times and 22.3 times more likely to use other substances (P less than .0001 for 10 variables); 4.9 times more likely to have attempted suicide (P less than .0001); or more likely to have engaged in violent behavior such as getting into physical fights (4.0 times), carrying a weapon (3.4 times) or a gun (5.1 times) within the last 30 days. In the four latter cases, P was less than .0001.
“With the ongoing opioid epidemic, pediatricians and child psychiatrists are likely to be more attuned to opioid misuse in their patients,” Dr. Bhatia and colleagues concluded. “If youth are screening positive for opioid misuse, pediatricians, nurses, social workers, child psychiatrists, and other providers assessing adolescents may have a new, broad range of other risky behaviors for which to screen regardless of the direction of the association.”
Substance use screening for treating substance use disorder traditionally has been is provided by a specialist, Jessica A. Kulak, PhD, MPH, said in an interview. “However, integration of care services may help to change societal norms around problematic substance use – both by decreasing stigma associated with substance use, as well as increasing clinicians’ preparedness, knowledge, and confidence in preventing and intervening on adolescents’ substance experimentation and use.” She recommended that clinicians in primary care improve their training by using the Substance Abuse and Mental Health Services Administration’s Screening, Brief Intervention, and Referral to Treatment program, which is available as a free online course.
Confidentiality is important in adolescent health, said Dr. Kulak, who is an assistant professor in the department of health, nutrition, and dietetics at State University of New York at Buffalo. “When discussing sensitive topics, such as binge drinking and opioid misuse, adolescents may fear that these or other risky activities may be disclosed to parents or law enforcement officials. Therefore, adolescent health providers should be aware of local, state, and federal laws pertaining to the confidentiality of minors.”
She added, “adolescents are often susceptible to others’ influences, so having open communication and support from a trusted adult – be it a parent or clinician – may also be protective against risky behaviors.”
The study by Vaca et al. was funded by the National Institutes of Health with support from the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development; the National Heart, Lung, and Blood Institute; the National Institute on Alcohol Abuse and Alcoholism; the National Institute on Drug Abuse; and the Maternal and Child Health Bureau of the Health Resources and Services Administration. The study by Bhatia et al. had no external funding. The authors from both studies reported no relevant financial disclosures. Dr. Kulak said she had no financial disclosures or other conflicts of interest.
SOURCE: Vaca FE et al. Pediatrics. 2020; doi: 10.1542/peds.2018-4095. Bhatia D et al. Pediatrics. 2020; doi: 10.1542/peds.2019-2470.
FROM PEDIATRICS
FDA Awards Grant To Study Temozolomide in Gist
A phase 2 study of temozolomide in gastrointestinal stromal tumors (GIST) received one of the 12 grants awarded in October by the US Food and Drug Administration (FDA) to enhance the development of medical products for patients with rare diseases. Jason Sicklick, MD, and the University of California San Diego in La Jolla will receive $1.5 million over 3 years to conduct the phase 2 study (NCT03556384). The objective of the study is to determine the efficacy at 6 months of temozolomide therapy in patients with the SDH-mutant/deficient subtype. Temozolomide is approved by the FDA to treat newly diagnosed glioblastoma multiforme and refractory anaplastic astrocytomas. It is not approved for the treatment of SDH-mutant/deficient GIST.
The FDA awarded the grants through the Orphan Products Clinical Trials Grants Program. Other orphan diseases receiving grants included glomerulopathy, gliomas, Fanconi anemia, sickle cell respiratory complications, Duchenne muscular dystrophy, HPV associated respiratory papillomatosis, refractory viral infections and T-cell immunodeficiency, oral cancer, retinoblastoma, cerebellar brain tumors, and acute myeloid leukemia.
A phase 2 study of temozolomide in gastrointestinal stromal tumors (GIST) received one of the 12 grants awarded in October by the US Food and Drug Administration (FDA) to enhance the development of medical products for patients with rare diseases. Jason Sicklick, MD, and the University of California San Diego in La Jolla will receive $1.5 million over 3 years to conduct the phase 2 study (NCT03556384). The objective of the study is to determine the efficacy at 6 months of temozolomide therapy in patients with the SDH-mutant/deficient subtype. Temozolomide is approved by the FDA to treat newly diagnosed glioblastoma multiforme and refractory anaplastic astrocytomas. It is not approved for the treatment of SDH-mutant/deficient GIST.
The FDA awarded the grants through the Orphan Products Clinical Trials Grants Program. Other orphan diseases receiving grants included glomerulopathy, gliomas, Fanconi anemia, sickle cell respiratory complications, Duchenne muscular dystrophy, HPV associated respiratory papillomatosis, refractory viral infections and T-cell immunodeficiency, oral cancer, retinoblastoma, cerebellar brain tumors, and acute myeloid leukemia.
A phase 2 study of temozolomide in gastrointestinal stromal tumors (GIST) received one of the 12 grants awarded in October by the US Food and Drug Administration (FDA) to enhance the development of medical products for patients with rare diseases. Jason Sicklick, MD, and the University of California San Diego in La Jolla will receive $1.5 million over 3 years to conduct the phase 2 study (NCT03556384). The objective of the study is to determine the efficacy at 6 months of temozolomide therapy in patients with the SDH-mutant/deficient subtype. Temozolomide is approved by the FDA to treat newly diagnosed glioblastoma multiforme and refractory anaplastic astrocytomas. It is not approved for the treatment of SDH-mutant/deficient GIST.
The FDA awarded the grants through the Orphan Products Clinical Trials Grants Program. Other orphan diseases receiving grants included glomerulopathy, gliomas, Fanconi anemia, sickle cell respiratory complications, Duchenne muscular dystrophy, HPV associated respiratory papillomatosis, refractory viral infections and T-cell immunodeficiency, oral cancer, retinoblastoma, cerebellar brain tumors, and acute myeloid leukemia.
Genomic profiling of AML and MDS yields prognostic clues
ORLANDO – A genome-wide study of blood and bone marrow samples from more than 1,300 adults with myeloid disorders has both confirmed the role of known or suspected driver mutations and uncovered new associations that could inform clinical care for patients with acute myeloid leukemia and myelodysplastic syndrome.
“Integration of mutational and expression data is important to refine subytpes and constellations of mutations with prognostic significance,” Ilaria Iacobucci, PhD, of St. Jude Children’s Research Hospital in Memphis said during a late-breaking abstract session at the annual meeting of the American Society of Hematology.
Her team conducted an analysis combining full genomic sequencing and gene-expression profiles in blood and bone marrow samples from 598 adults with acute myeloid leukemia (AML) and 706 with myelodysplastic syndrome (MDS).
The goals of the study were to provide “unbiased analysis of AML and MDS by integrated genomic and transcriptome data and clinico-pathologic features and clinical outcome” and to identify and define myeloid leukemia subtypes with diagnostic, prognostic, and therapeutic significance, she said.
The median age of the MDS cohort was 73.2 years (range 23.3-93.1). According to 2016 World Health Organization criteria, 37% had a diagnosis of MDS with excess blasts, 26.3% had MDS with ring sideroblasts, 20.9% had MDS with multilineage dysplasia, 14.6% had MDS with deletion 5q, and 1.1% had unclassifiable MDS.
The median age of the AML cohort was 68 years. Of this group, 31.7% had a diagnosis of AML not otherwise specified, 29.9% had known cytogenetic alterations, 27.3% had NPM1-mutated AML, and 9.7% had RUNX1-mutated disease.
Samples from all patients underwent tumor whole-genome sequencing and whole-transcriptome sequencing.
The combined sequencing confirmed a diagnosis of AML with recurrent genetic abnormalities in 11% of cases. These patients had disease with distinct gene-expression profiles and favorable prognosis. The sequencing identified combinations of mutations in genes linked with specific AML subtypes.
For example, combinations of mutations in KIT, ZBTB7A, ASXL2, RAD21, CSF3R, and DNM2 were associated with RUNX1-RUNXT1 leukemia, whereas mutations in FLT3, DDX54, WT1, and CALR in promyelocytic leukemia/retinoic acid receptor alpha were associated with promyelocytic leukemia, and KIT and BCORL1 mutations were associated with CBFB-rearranged leukemia.
In addition to rounding up the usual genomic suspects, the investigators also identified combinations that are associated with prognosis. Notably, NPM1 mutations were found in 27.4% of AML and 1% of MDS cases, and these mutations were characterized by four gene-expression signatures that were associated with different combinations of cooperating mutations in cohesin and signaling genes, and with outcome.
They found that patients with co-occurring NPM1 and FLT3 mutations had worse prognosis than those with mutations only in NPM1, whereas patients with NPM1 mutations co-occurring with cohesin gene mutations had better outcomes.
At a briefing prior to her presentation of the data, Dr. Iacobucci explained how her group’s findings might inform treatment, including the possibility of preventing development of AML in patients with MDS.
“What we are doing, in addition to the genomic part, is also establishing a repository of patient-derived xenografts, so in this way we can have the genome information, and we can have the biological material in vivo to test different therapies,” she said.
In an interview, Andrew H. Wei, MBBS, PhD, from the Alfred Hospital in Melbourne, who was not involved in the genomic study, commented on the role of sequencing in treatment of patients with myeloid malignancies.
“I think the future is that as the leukemia evolves, our therapy will evolve along with it. Furthermore, we now have the potential to measure many of these mutations with much higher sensitivity than just whole-genome sequencing, so we can imagine a future whereby we can track and measure these mutations as they rise in the patient’s bone marrow or blood before the patients becomes sick with florid leukemia, and it gives us the potential to predictably alter our management before they become sick,” he said.
The study was supported by St. Jude Children’s Research Hospital and the Leukemia and Lymphoma Society. Dr. Iacobucci and Dr. Wei reported having no relevant disclosures.
SOURCE: Iacobucci I et al. ASH 2019, Abstract LBA-4.
ORLANDO – A genome-wide study of blood and bone marrow samples from more than 1,300 adults with myeloid disorders has both confirmed the role of known or suspected driver mutations and uncovered new associations that could inform clinical care for patients with acute myeloid leukemia and myelodysplastic syndrome.
“Integration of mutational and expression data is important to refine subytpes and constellations of mutations with prognostic significance,” Ilaria Iacobucci, PhD, of St. Jude Children’s Research Hospital in Memphis said during a late-breaking abstract session at the annual meeting of the American Society of Hematology.
Her team conducted an analysis combining full genomic sequencing and gene-expression profiles in blood and bone marrow samples from 598 adults with acute myeloid leukemia (AML) and 706 with myelodysplastic syndrome (MDS).
The goals of the study were to provide “unbiased analysis of AML and MDS by integrated genomic and transcriptome data and clinico-pathologic features and clinical outcome” and to identify and define myeloid leukemia subtypes with diagnostic, prognostic, and therapeutic significance, she said.
The median age of the MDS cohort was 73.2 years (range 23.3-93.1). According to 2016 World Health Organization criteria, 37% had a diagnosis of MDS with excess blasts, 26.3% had MDS with ring sideroblasts, 20.9% had MDS with multilineage dysplasia, 14.6% had MDS with deletion 5q, and 1.1% had unclassifiable MDS.
The median age of the AML cohort was 68 years. Of this group, 31.7% had a diagnosis of AML not otherwise specified, 29.9% had known cytogenetic alterations, 27.3% had NPM1-mutated AML, and 9.7% had RUNX1-mutated disease.
Samples from all patients underwent tumor whole-genome sequencing and whole-transcriptome sequencing.
The combined sequencing confirmed a diagnosis of AML with recurrent genetic abnormalities in 11% of cases. These patients had disease with distinct gene-expression profiles and favorable prognosis. The sequencing identified combinations of mutations in genes linked with specific AML subtypes.
For example, combinations of mutations in KIT, ZBTB7A, ASXL2, RAD21, CSF3R, and DNM2 were associated with RUNX1-RUNXT1 leukemia, whereas mutations in FLT3, DDX54, WT1, and CALR in promyelocytic leukemia/retinoic acid receptor alpha were associated with promyelocytic leukemia, and KIT and BCORL1 mutations were associated with CBFB-rearranged leukemia.
In addition to rounding up the usual genomic suspects, the investigators also identified combinations that are associated with prognosis. Notably, NPM1 mutations were found in 27.4% of AML and 1% of MDS cases, and these mutations were characterized by four gene-expression signatures that were associated with different combinations of cooperating mutations in cohesin and signaling genes, and with outcome.
They found that patients with co-occurring NPM1 and FLT3 mutations had worse prognosis than those with mutations only in NPM1, whereas patients with NPM1 mutations co-occurring with cohesin gene mutations had better outcomes.
At a briefing prior to her presentation of the data, Dr. Iacobucci explained how her group’s findings might inform treatment, including the possibility of preventing development of AML in patients with MDS.
“What we are doing, in addition to the genomic part, is also establishing a repository of patient-derived xenografts, so in this way we can have the genome information, and we can have the biological material in vivo to test different therapies,” she said.
In an interview, Andrew H. Wei, MBBS, PhD, from the Alfred Hospital in Melbourne, who was not involved in the genomic study, commented on the role of sequencing in treatment of patients with myeloid malignancies.
“I think the future is that as the leukemia evolves, our therapy will evolve along with it. Furthermore, we now have the potential to measure many of these mutations with much higher sensitivity than just whole-genome sequencing, so we can imagine a future whereby we can track and measure these mutations as they rise in the patient’s bone marrow or blood before the patients becomes sick with florid leukemia, and it gives us the potential to predictably alter our management before they become sick,” he said.
The study was supported by St. Jude Children’s Research Hospital and the Leukemia and Lymphoma Society. Dr. Iacobucci and Dr. Wei reported having no relevant disclosures.
SOURCE: Iacobucci I et al. ASH 2019, Abstract LBA-4.
ORLANDO – A genome-wide study of blood and bone marrow samples from more than 1,300 adults with myeloid disorders has both confirmed the role of known or suspected driver mutations and uncovered new associations that could inform clinical care for patients with acute myeloid leukemia and myelodysplastic syndrome.
“Integration of mutational and expression data is important to refine subytpes and constellations of mutations with prognostic significance,” Ilaria Iacobucci, PhD, of St. Jude Children’s Research Hospital in Memphis said during a late-breaking abstract session at the annual meeting of the American Society of Hematology.
Her team conducted an analysis combining full genomic sequencing and gene-expression profiles in blood and bone marrow samples from 598 adults with acute myeloid leukemia (AML) and 706 with myelodysplastic syndrome (MDS).
The goals of the study were to provide “unbiased analysis of AML and MDS by integrated genomic and transcriptome data and clinico-pathologic features and clinical outcome” and to identify and define myeloid leukemia subtypes with diagnostic, prognostic, and therapeutic significance, she said.
The median age of the MDS cohort was 73.2 years (range 23.3-93.1). According to 2016 World Health Organization criteria, 37% had a diagnosis of MDS with excess blasts, 26.3% had MDS with ring sideroblasts, 20.9% had MDS with multilineage dysplasia, 14.6% had MDS with deletion 5q, and 1.1% had unclassifiable MDS.
The median age of the AML cohort was 68 years. Of this group, 31.7% had a diagnosis of AML not otherwise specified, 29.9% had known cytogenetic alterations, 27.3% had NPM1-mutated AML, and 9.7% had RUNX1-mutated disease.
Samples from all patients underwent tumor whole-genome sequencing and whole-transcriptome sequencing.
The combined sequencing confirmed a diagnosis of AML with recurrent genetic abnormalities in 11% of cases. These patients had disease with distinct gene-expression profiles and favorable prognosis. The sequencing identified combinations of mutations in genes linked with specific AML subtypes.
For example, combinations of mutations in KIT, ZBTB7A, ASXL2, RAD21, CSF3R, and DNM2 were associated with RUNX1-RUNXT1 leukemia, whereas mutations in FLT3, DDX54, WT1, and CALR in promyelocytic leukemia/retinoic acid receptor alpha were associated with promyelocytic leukemia, and KIT and BCORL1 mutations were associated with CBFB-rearranged leukemia.
In addition to rounding up the usual genomic suspects, the investigators also identified combinations that are associated with prognosis. Notably, NPM1 mutations were found in 27.4% of AML and 1% of MDS cases, and these mutations were characterized by four gene-expression signatures that were associated with different combinations of cooperating mutations in cohesin and signaling genes, and with outcome.
They found that patients with co-occurring NPM1 and FLT3 mutations had worse prognosis than those with mutations only in NPM1, whereas patients with NPM1 mutations co-occurring with cohesin gene mutations had better outcomes.
At a briefing prior to her presentation of the data, Dr. Iacobucci explained how her group’s findings might inform treatment, including the possibility of preventing development of AML in patients with MDS.
“What we are doing, in addition to the genomic part, is also establishing a repository of patient-derived xenografts, so in this way we can have the genome information, and we can have the biological material in vivo to test different therapies,” she said.
In an interview, Andrew H. Wei, MBBS, PhD, from the Alfred Hospital in Melbourne, who was not involved in the genomic study, commented on the role of sequencing in treatment of patients with myeloid malignancies.
“I think the future is that as the leukemia evolves, our therapy will evolve along with it. Furthermore, we now have the potential to measure many of these mutations with much higher sensitivity than just whole-genome sequencing, so we can imagine a future whereby we can track and measure these mutations as they rise in the patient’s bone marrow or blood before the patients becomes sick with florid leukemia, and it gives us the potential to predictably alter our management before they become sick,” he said.
The study was supported by St. Jude Children’s Research Hospital and the Leukemia and Lymphoma Society. Dr. Iacobucci and Dr. Wei reported having no relevant disclosures.
SOURCE: Iacobucci I et al. ASH 2019, Abstract LBA-4.
REPORTING FROM ASH 2019
HRS urges consumers to direct questions about wearables’ data to clinicians
With much of the public now wearing devices on their wrists or elsewhere capable of recording a range of vital signs, including heart rate abnormalities, the Heart Rhythm Society launched a guide for American consumers about wearables and the data they collect during a session on Jan. 9 at CES 2020 in Las Vegas.
While providing a succinct but comprehensive overview of the types of wearables and the health metrics they can record, the main and recurring message of the 10-page e-pamphlet is that, when a layperson has a question or concern about their data, the best course is to consult a clinician.
The “Guidance for Wearable Health Solutions,” produced by the Heart Rhythm Society (HRS) along with the Consumer Technology Association (CTA, which presents the annual CES exhibition), cautions that “most wearables are primarily suited for fitness and wellness,” and stresses that wearables “are not a substitute for medical devices prescribed by a clinician.” And in all cases, the document advises, when questions arise about the data – including an apparently high heart rate; a reading the device identifies as abnormal; and when symptoms appear such as a rapid heart rate, dizziness, or fluttering or flopping of the heart – the response that the guidance advocates is consistent: Talk with your clinician.
“Heart Rhythm Society members are seeing more and more patients with their own data collected by wearables,” said Nassir F. Marrouche, MD, professor of medicine and director of electrophysiology at Tulane University, New Orleans, and a member of the panel that wrote the guidance document for the HRS and CTA. “Every provider is dealing with consumer wearable data. The need is important for consumers to be supported. Consumers and patients are buying over-the-counter devices and using them for diagnosis and management, with little to no guidance, and we want to help them feel supported in managing their data and understand what to do with it,” Dr. Marrouche said in an interview.
“This is a new reality in medicine; the direction of information is changing. Consumers are collecting data themselves and coming to physicians already informed. There is a new shift in how information is collected, shared, and used.” Dr. Marrouche was 1 of 5 cardiac electrophysiologists who served on the 11-member writing group.
The new document for consumers “addresses an unmet need,” and the HRS collaboration with the CTA was “a unique opportunity to develop useful guidance that supports education and empowers consumers,” said Christina Wurster, chief strategy officer for the HRS in Washington and a member of the writing panel. “The questions outlined in the document are questions our members receive daily. The document is a resource they can direct people to.”
The HRS and CTA will “partner with consumer advocacy groups and professional societies to further disseminate the document,” added Ms. Wurster. “We’ll also have a strong push on social media to reach consumer audiences and drive awareness of this new resource,” she said in an interview. In addition, HRS “has strategic partnerships with other societies and will aim to work with them for dissemination, including societies related to internal medicine, emergency medicine, cardiology, and nursing, as well as also working with patient and consumer advocacy groups to reach the public.” The CTA will also actively publicize and disseminate the guidance document through their members.
Clinical guidelines play catch-up
Ironically, the HRS has issued this guidance to the public and has told people to take their wearable-collected heart data to clinicians before the HRS or any other medical group has advised clinicians on how they should handle, interpret, and use heart rhythm data collected this way.
Presumably, many if not most of the people with questions about their heart data from wearables are asymptomatic, because symptoms are what usually drive patients with a cardiac arrhythmia to consult a physician – they don’t wait to see what their device tells them. But the best way to manage asymptomatic arrhythmias like atrial fibrillation (AFib) remains a big clinical uncertainty today, with no evidence base as a guide, although several studies exploring this question are in progress.
“There are no clear and definitive data showing that treating subclinical atrial fibrillation improves outcomes. That’s what we need, and until we get these data you won’t see strong recommendations in guidelines” to screen patients for asymptomatic AFib or other arrhythmias, said Sana M. Al-Khatib, MD, a cardiac electrophysiologist and professor of medicine at Duke University, Durham, N.C., during the 2019 American Heart Association scientific sessions in Philadelphia in a talk about wearables and guidelines.
“If you intervene with silent AFib, do you improve outcomes? That evidence is lacking,” she said. Another shortcoming of current evidence is a clear understanding of what AFib burden warrants intervention, added Dr. Al-Khatib. “We see high-rate AFib episodes recorded in patients with implanted cardiac devices [and no symptoms], and we don’t know what to do with that either.”
The closest any existing guideline from a medical society comes to currently endorsing screening for AFib by a wearable is the 2016 European Society of Cardiology’s AFib management guidelines, which give “opportunistic screening” among people aged older than 65 years a IB recommendation, but specifically for screening by taking a patient’s pulse or with a ECG recording, with no mention of the screening role for wearables (Eur Heart J. 2016 Oct 7;37[38]:2893-967), Dr. Al-Khatib noted.
The most extensive data on screening for asymptomatic AFib in an unselected population came in the recently reported results from the Apple Heart Study, which enrolled more than 419,000 people monitored by a smart watch for a median of 117 days. During this screening, 2,161 people (0.52%) received a notification of having an irregular pulse (including 3.1% of those who were aged at least 65 years), which triggered more intensive assessment with an ECG patch for a median of 13 days in 450 of the 2,161 screening positives (21%) who agreed to participate in this follow-up. Among those 450 people, the patch test identified 34% as having actual AFib (N Engl J Med. 2019 Nov 14;381[20]:1909-17). But while this study provided evidence that screening for an irregular heartbeat with a wearable can identify AFib with some level of success, the results did not address whether this approach improved short- or long-term patient outcomes.
In addition, what the Apple Heart Study results showed was that this sort of screening results in a relatively large volume of follow-up testing. Of the 2,161 participants who received an irregular pulse notification, 1,376 (64%) returned a 90-day survey. Of these, 787 (57%) reported contact with a health care provider outside the study, 28% were prescribed a new medication, 33% were recommended to see a specialist (such as a cardiologist), and 36% were recommended to have additional testing.
“The results raise the question that a lot of resources were used,” to assess patients with a positive screening result, noted Paul A. Heidenreich, MD, a cardiologist and professor of medicine at Stanford (Calif.) University who studies quality of care for patients with heart disease. He estimated that, in the Apple Heart Study, each of the more than 2,000 patients who screening positive for an irregular heartbeat and underwent subsequent assessment ran up about $700 worth of follow-up testing. But he added that, in the case of AFib, the primary intervention that many previously undiagnosed AFib patients receive is some sort of anticoagulation for stroke prevention. Moreover, because this intervention is so effective there is a lot of money to play with to make AFib screening cost effective, as judged by typical, contemporary metrics of cost efficacy that value a quality-adjusted life-year (QALY) gain as reasonable for society to pay if the cost of an incremental QALY is $50,000-$150,000.
If the benchmark is a cost that’s within $50,000/QALY, then an average follow-up cost of $116/person to assess screened positives can fall within this cost ceiling. If the benchmark is $150,000/QALY, then follow-up costs can run as high as $491/person screened, said Dr. Heidenreich during the same AHA session where Dr. Al-Khatib spoke last November.
Despite this good news for screening for AFib with a wearable from a cost-effectiveness perspective, “there is so much uncertainty regarding the benefit and the consequences of incidental findings that we need an outcomes study before widespread implementation” of this type of screening, Dr. Heidenreich concluded. “We need an outcomes study to feel comfortable” with screening. “There is a huge potential for extra care that we don’t understand.”
Dr. Marrouche agreed that collecting adequate evidence to drive changes in clinical guidelines on how to use data from wearables has lagged behind the rapid spread of wearables and the information they can produce among the American public. “Outcomes and evidence will support guidelines development, but in the meantime, we’re offering education to clinicians, patients, and consumers. Consumers own their data, and they can share them with whomever they choose.”
The document notes that people who use wearables are, in general, “enthusiastic about tracking their data, not only for their own use, but also to share” with others, often on social media websites.
“We cannot control that, but our goal in the document is focused on the clinical relevance [of the data] and to help people better understand their data and use it in a meaningful and safe way,” Dr. Marrouche said.
Dr. Marrouche has been a consultant to, advisor to, or received research support from Abbott, Biosense Webster, Biotronik, GE Healthcare, Medtronic, Preventice, Sanofi-Aventis, Siemens, and Vytronus. Ms. Wurster is an employee of the Heart Rhythm Society. Dr. Al-Khatib has been a consultant to Milestone Pharmaceuticals and Medtronic, and she has also received other financial benefits from Medtronic. Dr. Heidenreich had no disclosures,
With much of the public now wearing devices on their wrists or elsewhere capable of recording a range of vital signs, including heart rate abnormalities, the Heart Rhythm Society launched a guide for American consumers about wearables and the data they collect during a session on Jan. 9 at CES 2020 in Las Vegas.
While providing a succinct but comprehensive overview of the types of wearables and the health metrics they can record, the main and recurring message of the 10-page e-pamphlet is that, when a layperson has a question or concern about their data, the best course is to consult a clinician.
The “Guidance for Wearable Health Solutions,” produced by the Heart Rhythm Society (HRS) along with the Consumer Technology Association (CTA, which presents the annual CES exhibition), cautions that “most wearables are primarily suited for fitness and wellness,” and stresses that wearables “are not a substitute for medical devices prescribed by a clinician.” And in all cases, the document advises, when questions arise about the data – including an apparently high heart rate; a reading the device identifies as abnormal; and when symptoms appear such as a rapid heart rate, dizziness, or fluttering or flopping of the heart – the response that the guidance advocates is consistent: Talk with your clinician.
“Heart Rhythm Society members are seeing more and more patients with their own data collected by wearables,” said Nassir F. Marrouche, MD, professor of medicine and director of electrophysiology at Tulane University, New Orleans, and a member of the panel that wrote the guidance document for the HRS and CTA. “Every provider is dealing with consumer wearable data. The need is important for consumers to be supported. Consumers and patients are buying over-the-counter devices and using them for diagnosis and management, with little to no guidance, and we want to help them feel supported in managing their data and understand what to do with it,” Dr. Marrouche said in an interview.
“This is a new reality in medicine; the direction of information is changing. Consumers are collecting data themselves and coming to physicians already informed. There is a new shift in how information is collected, shared, and used.” Dr. Marrouche was 1 of 5 cardiac electrophysiologists who served on the 11-member writing group.
The new document for consumers “addresses an unmet need,” and the HRS collaboration with the CTA was “a unique opportunity to develop useful guidance that supports education and empowers consumers,” said Christina Wurster, chief strategy officer for the HRS in Washington and a member of the writing panel. “The questions outlined in the document are questions our members receive daily. The document is a resource they can direct people to.”
The HRS and CTA will “partner with consumer advocacy groups and professional societies to further disseminate the document,” added Ms. Wurster. “We’ll also have a strong push on social media to reach consumer audiences and drive awareness of this new resource,” she said in an interview. In addition, HRS “has strategic partnerships with other societies and will aim to work with them for dissemination, including societies related to internal medicine, emergency medicine, cardiology, and nursing, as well as also working with patient and consumer advocacy groups to reach the public.” The CTA will also actively publicize and disseminate the guidance document through their members.
Clinical guidelines play catch-up
Ironically, the HRS has issued this guidance to the public and has told people to take their wearable-collected heart data to clinicians before the HRS or any other medical group has advised clinicians on how they should handle, interpret, and use heart rhythm data collected this way.
Presumably, many if not most of the people with questions about their heart data from wearables are asymptomatic, because symptoms are what usually drive patients with a cardiac arrhythmia to consult a physician – they don’t wait to see what their device tells them. But the best way to manage asymptomatic arrhythmias like atrial fibrillation (AFib) remains a big clinical uncertainty today, with no evidence base as a guide, although several studies exploring this question are in progress.
“There are no clear and definitive data showing that treating subclinical atrial fibrillation improves outcomes. That’s what we need, and until we get these data you won’t see strong recommendations in guidelines” to screen patients for asymptomatic AFib or other arrhythmias, said Sana M. Al-Khatib, MD, a cardiac electrophysiologist and professor of medicine at Duke University, Durham, N.C., during the 2019 American Heart Association scientific sessions in Philadelphia in a talk about wearables and guidelines.
“If you intervene with silent AFib, do you improve outcomes? That evidence is lacking,” she said. Another shortcoming of current evidence is a clear understanding of what AFib burden warrants intervention, added Dr. Al-Khatib. “We see high-rate AFib episodes recorded in patients with implanted cardiac devices [and no symptoms], and we don’t know what to do with that either.”
The closest any existing guideline from a medical society comes to currently endorsing screening for AFib by a wearable is the 2016 European Society of Cardiology’s AFib management guidelines, which give “opportunistic screening” among people aged older than 65 years a IB recommendation, but specifically for screening by taking a patient’s pulse or with a ECG recording, with no mention of the screening role for wearables (Eur Heart J. 2016 Oct 7;37[38]:2893-967), Dr. Al-Khatib noted.
The most extensive data on screening for asymptomatic AFib in an unselected population came in the recently reported results from the Apple Heart Study, which enrolled more than 419,000 people monitored by a smart watch for a median of 117 days. During this screening, 2,161 people (0.52%) received a notification of having an irregular pulse (including 3.1% of those who were aged at least 65 years), which triggered more intensive assessment with an ECG patch for a median of 13 days in 450 of the 2,161 screening positives (21%) who agreed to participate in this follow-up. Among those 450 people, the patch test identified 34% as having actual AFib (N Engl J Med. 2019 Nov 14;381[20]:1909-17). But while this study provided evidence that screening for an irregular heartbeat with a wearable can identify AFib with some level of success, the results did not address whether this approach improved short- or long-term patient outcomes.
In addition, what the Apple Heart Study results showed was that this sort of screening results in a relatively large volume of follow-up testing. Of the 2,161 participants who received an irregular pulse notification, 1,376 (64%) returned a 90-day survey. Of these, 787 (57%) reported contact with a health care provider outside the study, 28% were prescribed a new medication, 33% were recommended to see a specialist (such as a cardiologist), and 36% were recommended to have additional testing.
“The results raise the question that a lot of resources were used,” to assess patients with a positive screening result, noted Paul A. Heidenreich, MD, a cardiologist and professor of medicine at Stanford (Calif.) University who studies quality of care for patients with heart disease. He estimated that, in the Apple Heart Study, each of the more than 2,000 patients who screening positive for an irregular heartbeat and underwent subsequent assessment ran up about $700 worth of follow-up testing. But he added that, in the case of AFib, the primary intervention that many previously undiagnosed AFib patients receive is some sort of anticoagulation for stroke prevention. Moreover, because this intervention is so effective there is a lot of money to play with to make AFib screening cost effective, as judged by typical, contemporary metrics of cost efficacy that value a quality-adjusted life-year (QALY) gain as reasonable for society to pay if the cost of an incremental QALY is $50,000-$150,000.
If the benchmark is a cost that’s within $50,000/QALY, then an average follow-up cost of $116/person to assess screened positives can fall within this cost ceiling. If the benchmark is $150,000/QALY, then follow-up costs can run as high as $491/person screened, said Dr. Heidenreich during the same AHA session where Dr. Al-Khatib spoke last November.
Despite this good news for screening for AFib with a wearable from a cost-effectiveness perspective, “there is so much uncertainty regarding the benefit and the consequences of incidental findings that we need an outcomes study before widespread implementation” of this type of screening, Dr. Heidenreich concluded. “We need an outcomes study to feel comfortable” with screening. “There is a huge potential for extra care that we don’t understand.”
Dr. Marrouche agreed that collecting adequate evidence to drive changes in clinical guidelines on how to use data from wearables has lagged behind the rapid spread of wearables and the information they can produce among the American public. “Outcomes and evidence will support guidelines development, but in the meantime, we’re offering education to clinicians, patients, and consumers. Consumers own their data, and they can share them with whomever they choose.”
The document notes that people who use wearables are, in general, “enthusiastic about tracking their data, not only for their own use, but also to share” with others, often on social media websites.
“We cannot control that, but our goal in the document is focused on the clinical relevance [of the data] and to help people better understand their data and use it in a meaningful and safe way,” Dr. Marrouche said.
Dr. Marrouche has been a consultant to, advisor to, or received research support from Abbott, Biosense Webster, Biotronik, GE Healthcare, Medtronic, Preventice, Sanofi-Aventis, Siemens, and Vytronus. Ms. Wurster is an employee of the Heart Rhythm Society. Dr. Al-Khatib has been a consultant to Milestone Pharmaceuticals and Medtronic, and she has also received other financial benefits from Medtronic. Dr. Heidenreich had no disclosures,
With much of the public now wearing devices on their wrists or elsewhere capable of recording a range of vital signs, including heart rate abnormalities, the Heart Rhythm Society launched a guide for American consumers about wearables and the data they collect during a session on Jan. 9 at CES 2020 in Las Vegas.
While providing a succinct but comprehensive overview of the types of wearables and the health metrics they can record, the main and recurring message of the 10-page e-pamphlet is that, when a layperson has a question or concern about their data, the best course is to consult a clinician.
The “Guidance for Wearable Health Solutions,” produced by the Heart Rhythm Society (HRS) along with the Consumer Technology Association (CTA, which presents the annual CES exhibition), cautions that “most wearables are primarily suited for fitness and wellness,” and stresses that wearables “are not a substitute for medical devices prescribed by a clinician.” And in all cases, the document advises, when questions arise about the data – including an apparently high heart rate; a reading the device identifies as abnormal; and when symptoms appear such as a rapid heart rate, dizziness, or fluttering or flopping of the heart – the response that the guidance advocates is consistent: Talk with your clinician.
“Heart Rhythm Society members are seeing more and more patients with their own data collected by wearables,” said Nassir F. Marrouche, MD, professor of medicine and director of electrophysiology at Tulane University, New Orleans, and a member of the panel that wrote the guidance document for the HRS and CTA. “Every provider is dealing with consumer wearable data. The need is important for consumers to be supported. Consumers and patients are buying over-the-counter devices and using them for diagnosis and management, with little to no guidance, and we want to help them feel supported in managing their data and understand what to do with it,” Dr. Marrouche said in an interview.
“This is a new reality in medicine; the direction of information is changing. Consumers are collecting data themselves and coming to physicians already informed. There is a new shift in how information is collected, shared, and used.” Dr. Marrouche was 1 of 5 cardiac electrophysiologists who served on the 11-member writing group.
The new document for consumers “addresses an unmet need,” and the HRS collaboration with the CTA was “a unique opportunity to develop useful guidance that supports education and empowers consumers,” said Christina Wurster, chief strategy officer for the HRS in Washington and a member of the writing panel. “The questions outlined in the document are questions our members receive daily. The document is a resource they can direct people to.”
The HRS and CTA will “partner with consumer advocacy groups and professional societies to further disseminate the document,” added Ms. Wurster. “We’ll also have a strong push on social media to reach consumer audiences and drive awareness of this new resource,” she said in an interview. In addition, HRS “has strategic partnerships with other societies and will aim to work with them for dissemination, including societies related to internal medicine, emergency medicine, cardiology, and nursing, as well as also working with patient and consumer advocacy groups to reach the public.” The CTA will also actively publicize and disseminate the guidance document through their members.
Clinical guidelines play catch-up
Ironically, the HRS has issued this guidance to the public and has told people to take their wearable-collected heart data to clinicians before the HRS or any other medical group has advised clinicians on how they should handle, interpret, and use heart rhythm data collected this way.
Presumably, many if not most of the people with questions about their heart data from wearables are asymptomatic, because symptoms are what usually drive patients with a cardiac arrhythmia to consult a physician – they don’t wait to see what their device tells them. But the best way to manage asymptomatic arrhythmias like atrial fibrillation (AFib) remains a big clinical uncertainty today, with no evidence base as a guide, although several studies exploring this question are in progress.
“There are no clear and definitive data showing that treating subclinical atrial fibrillation improves outcomes. That’s what we need, and until we get these data you won’t see strong recommendations in guidelines” to screen patients for asymptomatic AFib or other arrhythmias, said Sana M. Al-Khatib, MD, a cardiac electrophysiologist and professor of medicine at Duke University, Durham, N.C., during the 2019 American Heart Association scientific sessions in Philadelphia in a talk about wearables and guidelines.
“If you intervene with silent AFib, do you improve outcomes? That evidence is lacking,” she said. Another shortcoming of current evidence is a clear understanding of what AFib burden warrants intervention, added Dr. Al-Khatib. “We see high-rate AFib episodes recorded in patients with implanted cardiac devices [and no symptoms], and we don’t know what to do with that either.”
The closest any existing guideline from a medical society comes to currently endorsing screening for AFib by a wearable is the 2016 European Society of Cardiology’s AFib management guidelines, which give “opportunistic screening” among people aged older than 65 years a IB recommendation, but specifically for screening by taking a patient’s pulse or with a ECG recording, with no mention of the screening role for wearables (Eur Heart J. 2016 Oct 7;37[38]:2893-967), Dr. Al-Khatib noted.
The most extensive data on screening for asymptomatic AFib in an unselected population came in the recently reported results from the Apple Heart Study, which enrolled more than 419,000 people monitored by a smart watch for a median of 117 days. During this screening, 2,161 people (0.52%) received a notification of having an irregular pulse (including 3.1% of those who were aged at least 65 years), which triggered more intensive assessment with an ECG patch for a median of 13 days in 450 of the 2,161 screening positives (21%) who agreed to participate in this follow-up. Among those 450 people, the patch test identified 34% as having actual AFib (N Engl J Med. 2019 Nov 14;381[20]:1909-17). But while this study provided evidence that screening for an irregular heartbeat with a wearable can identify AFib with some level of success, the results did not address whether this approach improved short- or long-term patient outcomes.
In addition, what the Apple Heart Study results showed was that this sort of screening results in a relatively large volume of follow-up testing. Of the 2,161 participants who received an irregular pulse notification, 1,376 (64%) returned a 90-day survey. Of these, 787 (57%) reported contact with a health care provider outside the study, 28% were prescribed a new medication, 33% were recommended to see a specialist (such as a cardiologist), and 36% were recommended to have additional testing.
“The results raise the question that a lot of resources were used,” to assess patients with a positive screening result, noted Paul A. Heidenreich, MD, a cardiologist and professor of medicine at Stanford (Calif.) University who studies quality of care for patients with heart disease. He estimated that, in the Apple Heart Study, each of the more than 2,000 patients who screening positive for an irregular heartbeat and underwent subsequent assessment ran up about $700 worth of follow-up testing. But he added that, in the case of AFib, the primary intervention that many previously undiagnosed AFib patients receive is some sort of anticoagulation for stroke prevention. Moreover, because this intervention is so effective there is a lot of money to play with to make AFib screening cost effective, as judged by typical, contemporary metrics of cost efficacy that value a quality-adjusted life-year (QALY) gain as reasonable for society to pay if the cost of an incremental QALY is $50,000-$150,000.
If the benchmark is a cost that’s within $50,000/QALY, then an average follow-up cost of $116/person to assess screened positives can fall within this cost ceiling. If the benchmark is $150,000/QALY, then follow-up costs can run as high as $491/person screened, said Dr. Heidenreich during the same AHA session where Dr. Al-Khatib spoke last November.
Despite this good news for screening for AFib with a wearable from a cost-effectiveness perspective, “there is so much uncertainty regarding the benefit and the consequences of incidental findings that we need an outcomes study before widespread implementation” of this type of screening, Dr. Heidenreich concluded. “We need an outcomes study to feel comfortable” with screening. “There is a huge potential for extra care that we don’t understand.”
Dr. Marrouche agreed that collecting adequate evidence to drive changes in clinical guidelines on how to use data from wearables has lagged behind the rapid spread of wearables and the information they can produce among the American public. “Outcomes and evidence will support guidelines development, but in the meantime, we’re offering education to clinicians, patients, and consumers. Consumers own their data, and they can share them with whomever they choose.”
The document notes that people who use wearables are, in general, “enthusiastic about tracking their data, not only for their own use, but also to share” with others, often on social media websites.
“We cannot control that, but our goal in the document is focused on the clinical relevance [of the data] and to help people better understand their data and use it in a meaningful and safe way,” Dr. Marrouche said.
Dr. Marrouche has been a consultant to, advisor to, or received research support from Abbott, Biosense Webster, Biotronik, GE Healthcare, Medtronic, Preventice, Sanofi-Aventis, Siemens, and Vytronus. Ms. Wurster is an employee of the Heart Rhythm Society. Dr. Al-Khatib has been a consultant to Milestone Pharmaceuticals and Medtronic, and she has also received other financial benefits from Medtronic. Dr. Heidenreich had no disclosures,
Evidence builds for bariatric surgery’s role in cancer prevention
LAS VEGAS – The ability of bariatric surgery and substantial subsequent weight loss to cut the incidence of a variety of obesity-related cancers and other malignancies received further confirmation in results from two studies reported at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
In one study, 2,107 adults enrolled in the Longitudinal Assessment of Bariatric Surgery (LABS-2) study showed a statistically significant halving of the cancer incidence during 7 years of follow-up in patients who underwent bariatric surgery and had a reduction of at least 20% in their presurgical body mass index (BMI), compared with patients in the study who underwent bariatric surgery but lost less weight, reported Andrea M. Stroud, MD, a bariatric surgeon at the Oregon Health & Science University, Portland.
In the second study, analysis of about 1.7 million hospitalized U.S. patients in the National Inpatient Sample showed that the incidence of an obesity-related cancer was 21% higher in more than 1.4 million obese individuals (BMI, 35 kg/m2 or greater) with no history of bariatric surgery, compared with nearly 247,000 people in the same database with a history of both obesity and bariatric surgery, said Juliana Henrique, MD, a bariatric surgeon at the Cleveland Clinic Florida in Weston.
The study reported by Dr. Henrique focused specifically on the 13 cancer types identified by the Centers for Disease Control and Prevention as having an incidence that links with overweight and obesity (Morb Mortal Wkly Rep. 2017;66[39]:1052-8), whereas the study presented by Dr. Stroud included all incident cancers during follow-up, but which were predominantly obesity related, with breast cancer – an obesity-related malignancy – having the highest incidence. Overall, 40% of all U.S. cancers in 2014 were obesity related, according to the CDC’s report.
“A number of studies have shown decreases in cancer rates after bariatric surgery, especially female cancers like breast and ovarian,” commented John Scott, MD, director of metabolic and bariatric surgery for Prism Health–Upstate in Greenville, S.C. “These two reports build on that.”
The evidence for weight loss after bariatric surgery as a means to cut the risk of a first or recurrent cancer has become strong enough for some patients to see cancer prophylaxis as a prime reason to undergo the procedure, said surgeons at the meeting.
Bariatric surgery and subsequent weight loss “is a substantial preventive factor for cancer, especially in patients who have obesity and diabetes,” commented Theresa LaMasters, MD, a bariatric surgeon in West Des Moines, Iowa. “It might not just be weight loss. It’s likely a multifactorial effect, including reduced inflammation after bariatric surgery, but weight loss is a component” of the effect, Dr. LaMasters said in an interview. It is now common for her to see patients seeking bariatric surgery because of a family or personal history of cancer. “Patients are trying to reduce their future risk” for cancer with bariatric surgery, she added.
The LABS-2 study enrolled 2,458 patients who were part of the first LABS cohort, LABS-1, but followed them longer term. The data Dr. Stroud reported came from 2,107 of the LABS-2 patients without a history of cancer, no cancer diagnosed in the first year after bariatric surgery, and longer-term follow-up of 7 years. About three-quarters of the patients underwent gastric bypass, with the rest undergoing laparoscopic gastric band placement. Nearly half of those included had diabetes. Their average BMI was 45-50 kg/m2.
Dr. Stroud and associates ran an analysis that divided the populations into tertiles based on percentage of baseline body mass lost at 12 months after surgery and cancer-free survival during the 7 years after the 12-month follow-up. The incidence of cancer was 51% lower in patients who lost 20%-34% of their BMI, compared with those who lost less than 20%, a statistically significant difference, and patients who lost 35% or more of their BMI had a 31% reduced cancer rate, compared with those who lost less than 20%, a difference that was not statistically significant, Dr. Stroud reported. The patients who lost less weight after surgery mostly underwent gastric banding, whereas those who lost more mostly underwent gastric bypass.
The analysis reported by Dr. Henrique used data collected in the U.S. National Inpatient Sample during 2010-2014, which totaled more than 7 million patients hospitalized for cancer, including 1,423,367 with a history of obesity and 246,668 with obesity who had undergone bariatric surgery. Those without bariatric surgery had a 21% higher rate of developing obesity-related cancers after adjustment for many baseline demographic and clinical features, Dr. Henrique said. The cancer protection after bariatric surgery was especially notable in the subset of patients in the sample with a genetic predisposition to developing cancer.
LABS-1 and LABS-2 were funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Stroud and Dr. Henrique had no disclosures.
SOURCES: Stroud AM et al. Obesity Week, Abstract A107; Henrique J et al. Obesity Week, Abstract A108.
LAS VEGAS – The ability of bariatric surgery and substantial subsequent weight loss to cut the incidence of a variety of obesity-related cancers and other malignancies received further confirmation in results from two studies reported at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
In one study, 2,107 adults enrolled in the Longitudinal Assessment of Bariatric Surgery (LABS-2) study showed a statistically significant halving of the cancer incidence during 7 years of follow-up in patients who underwent bariatric surgery and had a reduction of at least 20% in their presurgical body mass index (BMI), compared with patients in the study who underwent bariatric surgery but lost less weight, reported Andrea M. Stroud, MD, a bariatric surgeon at the Oregon Health & Science University, Portland.
In the second study, analysis of about 1.7 million hospitalized U.S. patients in the National Inpatient Sample showed that the incidence of an obesity-related cancer was 21% higher in more than 1.4 million obese individuals (BMI, 35 kg/m2 or greater) with no history of bariatric surgery, compared with nearly 247,000 people in the same database with a history of both obesity and bariatric surgery, said Juliana Henrique, MD, a bariatric surgeon at the Cleveland Clinic Florida in Weston.
The study reported by Dr. Henrique focused specifically on the 13 cancer types identified by the Centers for Disease Control and Prevention as having an incidence that links with overweight and obesity (Morb Mortal Wkly Rep. 2017;66[39]:1052-8), whereas the study presented by Dr. Stroud included all incident cancers during follow-up, but which were predominantly obesity related, with breast cancer – an obesity-related malignancy – having the highest incidence. Overall, 40% of all U.S. cancers in 2014 were obesity related, according to the CDC’s report.
“A number of studies have shown decreases in cancer rates after bariatric surgery, especially female cancers like breast and ovarian,” commented John Scott, MD, director of metabolic and bariatric surgery for Prism Health–Upstate in Greenville, S.C. “These two reports build on that.”
The evidence for weight loss after bariatric surgery as a means to cut the risk of a first or recurrent cancer has become strong enough for some patients to see cancer prophylaxis as a prime reason to undergo the procedure, said surgeons at the meeting.
Bariatric surgery and subsequent weight loss “is a substantial preventive factor for cancer, especially in patients who have obesity and diabetes,” commented Theresa LaMasters, MD, a bariatric surgeon in West Des Moines, Iowa. “It might not just be weight loss. It’s likely a multifactorial effect, including reduced inflammation after bariatric surgery, but weight loss is a component” of the effect, Dr. LaMasters said in an interview. It is now common for her to see patients seeking bariatric surgery because of a family or personal history of cancer. “Patients are trying to reduce their future risk” for cancer with bariatric surgery, she added.
The LABS-2 study enrolled 2,458 patients who were part of the first LABS cohort, LABS-1, but followed them longer term. The data Dr. Stroud reported came from 2,107 of the LABS-2 patients without a history of cancer, no cancer diagnosed in the first year after bariatric surgery, and longer-term follow-up of 7 years. About three-quarters of the patients underwent gastric bypass, with the rest undergoing laparoscopic gastric band placement. Nearly half of those included had diabetes. Their average BMI was 45-50 kg/m2.
Dr. Stroud and associates ran an analysis that divided the populations into tertiles based on percentage of baseline body mass lost at 12 months after surgery and cancer-free survival during the 7 years after the 12-month follow-up. The incidence of cancer was 51% lower in patients who lost 20%-34% of their BMI, compared with those who lost less than 20%, a statistically significant difference, and patients who lost 35% or more of their BMI had a 31% reduced cancer rate, compared with those who lost less than 20%, a difference that was not statistically significant, Dr. Stroud reported. The patients who lost less weight after surgery mostly underwent gastric banding, whereas those who lost more mostly underwent gastric bypass.
The analysis reported by Dr. Henrique used data collected in the U.S. National Inpatient Sample during 2010-2014, which totaled more than 7 million patients hospitalized for cancer, including 1,423,367 with a history of obesity and 246,668 with obesity who had undergone bariatric surgery. Those without bariatric surgery had a 21% higher rate of developing obesity-related cancers after adjustment for many baseline demographic and clinical features, Dr. Henrique said. The cancer protection after bariatric surgery was especially notable in the subset of patients in the sample with a genetic predisposition to developing cancer.
LABS-1 and LABS-2 were funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Stroud and Dr. Henrique had no disclosures.
SOURCES: Stroud AM et al. Obesity Week, Abstract A107; Henrique J et al. Obesity Week, Abstract A108.
LAS VEGAS – The ability of bariatric surgery and substantial subsequent weight loss to cut the incidence of a variety of obesity-related cancers and other malignancies received further confirmation in results from two studies reported at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
In one study, 2,107 adults enrolled in the Longitudinal Assessment of Bariatric Surgery (LABS-2) study showed a statistically significant halving of the cancer incidence during 7 years of follow-up in patients who underwent bariatric surgery and had a reduction of at least 20% in their presurgical body mass index (BMI), compared with patients in the study who underwent bariatric surgery but lost less weight, reported Andrea M. Stroud, MD, a bariatric surgeon at the Oregon Health & Science University, Portland.
In the second study, analysis of about 1.7 million hospitalized U.S. patients in the National Inpatient Sample showed that the incidence of an obesity-related cancer was 21% higher in more than 1.4 million obese individuals (BMI, 35 kg/m2 or greater) with no history of bariatric surgery, compared with nearly 247,000 people in the same database with a history of both obesity and bariatric surgery, said Juliana Henrique, MD, a bariatric surgeon at the Cleveland Clinic Florida in Weston.
The study reported by Dr. Henrique focused specifically on the 13 cancer types identified by the Centers for Disease Control and Prevention as having an incidence that links with overweight and obesity (Morb Mortal Wkly Rep. 2017;66[39]:1052-8), whereas the study presented by Dr. Stroud included all incident cancers during follow-up, but which were predominantly obesity related, with breast cancer – an obesity-related malignancy – having the highest incidence. Overall, 40% of all U.S. cancers in 2014 were obesity related, according to the CDC’s report.
“A number of studies have shown decreases in cancer rates after bariatric surgery, especially female cancers like breast and ovarian,” commented John Scott, MD, director of metabolic and bariatric surgery for Prism Health–Upstate in Greenville, S.C. “These two reports build on that.”
The evidence for weight loss after bariatric surgery as a means to cut the risk of a first or recurrent cancer has become strong enough for some patients to see cancer prophylaxis as a prime reason to undergo the procedure, said surgeons at the meeting.
Bariatric surgery and subsequent weight loss “is a substantial preventive factor for cancer, especially in patients who have obesity and diabetes,” commented Theresa LaMasters, MD, a bariatric surgeon in West Des Moines, Iowa. “It might not just be weight loss. It’s likely a multifactorial effect, including reduced inflammation after bariatric surgery, but weight loss is a component” of the effect, Dr. LaMasters said in an interview. It is now common for her to see patients seeking bariatric surgery because of a family or personal history of cancer. “Patients are trying to reduce their future risk” for cancer with bariatric surgery, she added.
The LABS-2 study enrolled 2,458 patients who were part of the first LABS cohort, LABS-1, but followed them longer term. The data Dr. Stroud reported came from 2,107 of the LABS-2 patients without a history of cancer, no cancer diagnosed in the first year after bariatric surgery, and longer-term follow-up of 7 years. About three-quarters of the patients underwent gastric bypass, with the rest undergoing laparoscopic gastric band placement. Nearly half of those included had diabetes. Their average BMI was 45-50 kg/m2.
Dr. Stroud and associates ran an analysis that divided the populations into tertiles based on percentage of baseline body mass lost at 12 months after surgery and cancer-free survival during the 7 years after the 12-month follow-up. The incidence of cancer was 51% lower in patients who lost 20%-34% of their BMI, compared with those who lost less than 20%, a statistically significant difference, and patients who lost 35% or more of their BMI had a 31% reduced cancer rate, compared with those who lost less than 20%, a difference that was not statistically significant, Dr. Stroud reported. The patients who lost less weight after surgery mostly underwent gastric banding, whereas those who lost more mostly underwent gastric bypass.
The analysis reported by Dr. Henrique used data collected in the U.S. National Inpatient Sample during 2010-2014, which totaled more than 7 million patients hospitalized for cancer, including 1,423,367 with a history of obesity and 246,668 with obesity who had undergone bariatric surgery. Those without bariatric surgery had a 21% higher rate of developing obesity-related cancers after adjustment for many baseline demographic and clinical features, Dr. Henrique said. The cancer protection after bariatric surgery was especially notable in the subset of patients in the sample with a genetic predisposition to developing cancer.
LABS-1 and LABS-2 were funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Stroud and Dr. Henrique had no disclosures.
SOURCES: Stroud AM et al. Obesity Week, Abstract A107; Henrique J et al. Obesity Week, Abstract A108.
REPORTING FROM OBESITY WEEK 2019
Carbohydrate restriction a viable choice for reversal of type 2 diabetes, expert says
LOS ANGELES – according to Sarah Hallberg, DO.
“Nutritional ketosis supports diabetes reversal by reducing insulin resistance while providing an alternative fuel to glucose with favorable signaling properties,” she said at the World Congress on Insulin Resistance, Diabetes, and Cardiovascular Disease.
Low-carbohydrate nutritional patterns including ketosis have extensive clinical trial evidence for improvement of type 2 diabetes, including preliminary results from a 5-year study of 465 patients enrolled in the Indiana Type 2 Diabetes Reversal Trial that Dr. Hallberg is overseeing in her role as medical director and founder of the medically supervised weight-loss program at Indiana University Health Arnett, Lafayette.
“The ketogenic diet is not a fad diet, it’s what we used to treat people with before the advent of insulin,” said Dr. Hallberg, who has been recommending and counseling patients with type 2 diabetes to follow a ketogenic diet for nearly 10 years. “Of course, insulin has been wonderful. It’s saved so many people with type 1 diabetes. But we also misused it in type 2 diabetes. Instead of counseling people the way we used to about the food that they’re taking in to control their blood sugar, we’ve just been putting [them] on medication, including insulin.”
The American Diabetes Association and other organizations have updated their guidelines to include low-carbohydrate eating patterns for type 2 diabetes treatment, she continued. Veterans Affairs/Department of Defense recommend carbohydrate levels as low as 14%.
Dr. Hallberg, who is also medical director for Virta Health, defined a very-low-carbohydrate or ketogenic diet as less than 50 g of carbohydrates per day, or fewer than 10% of calories consumed. A low-carbohydrate diet is 51-130 g of carbohydrates per day, or 25% or fewer calories consumed, whereas anything above 25% calories consumed is a not a low-carbohydrate diet. A well-formulated ketogenic diet, she continued, consists of 5%-10% carbohydrates (or less than 50 g), 15%-20% protein, and 70%-80% fat. The carbohydrates include 5-10 g per day of protein-based food, 10-15 g of vegetables, 5-10 g of nuts/seeds, 5-10 g of fruits, and 5-10 g of miscellaneous nutrients. “When we’re talking about a total carbohydrate intake per day of under 50 g, you can get a lot of vegetables and nuts in,” she said. “I like to tell my patients they’re not eating GPS: no grains, no potatoes, and no sugar.”
Recently, Dr. Hallberg and colleagues published a review in which they sought to evaluate the appropriateness of sources cited in the ADA’s guidelines on eating patterns for the management of type 2 diabetes, identify additional relevant sources, and evaluate the evidence (Diabetes Obes Metab. 2019;21[8]:1769-79). “We looked at how much evidence there is for the low-carb diet, the Mediterranean diet, the DASH [Dietary Approaches to Stop Hypertension] diet, and a plant-based diet,” she said. “We found a wide variation in the evidence for each eating pattern, but the low-carb eating pattern for diabetes has so much more evidence than any of the other eating patterns.”
In an earlier study, researchers followed 10 inpatients with diabetes in a metabolic ward for 3 weeks. Their mean age was 51 years, and their mean body mass index was 40.3 kg/m2. The patients were fed a standard diet for 7 days, then a low-carbohydrate diet (21 g per day) for 14 days (Ann Intern Med 2005; 142[6]:403-11). After 2 weeks of the low-carbohydrate diet, their mean fasting blood glucose dropped from 7.5 to 6.3 mmol/L, and their mean hemoglobin A1c (HbA1c) fell from 7.3% to 6.8%. “The levels came down very fast,” said Dr. Hallberg, who was not involved with the study. “This is an important part of the intervention, because when you get a patient who’s tried everything, who’s injecting hundreds of units of insulin every day, you can make a huge difference in the first couple of weeks. It is not unusual for us to pull patients off of 200-plus units of insulin. This is as motivating as all get out. It also affects their pocketbook right away. This is one of the reasons our patients are able to sustain a ketogenic diet along with support: early motivation and satisfaction.”
In a longer-term trial, researchers evaluated the impact of a ketogenic diet in 64 obese patients with diabetes over the course of 56 weeks (Moll Cell Biochem. 2007;302[1-2]:249-56). The body weight, body mass index, and levels of blood glucose, total cholesterol, LDL cholesterol, triglycerides, and urea showed a significant decrease from week 1 to week 56 (P less than .0001), while the level of HDL cholesterol increased significantly (P less than .0001).
A separate trial conducted in Israel evaluated the effects of a low-carbohydrate diet, compared with a Mediterranean or low-fat diet in 322 moderately obese patients over the course of 2 years (N Engl J Med. 2008;359:229-41). The rate of adherence to a study diet was 85% at 2 years. The mean weight change was greatest for those on the low-carbohydrate diet, followed by the Mediterranean and low-fat diets. Fasting glucose was best for those on the Mediterranean diet at the end of 2 years, whereas change in HbA1c was best among those on the low-carbohydrate diet.
Another study randomized patients to a low-carbohydrate ketogenic diet (less than 20 g per day with no calorie restriction) or to a low–glycemic index diet (55% carbohydrate restriction of 500 kcal from baseline) over the course of 24 weeks (Nutr Metab [Lond]. 2008 Dec 19. doi:10.1186/1743-7075-5-36). Between baseline and week 24, the mean HbA1c fell from 8.8% to 7.3% in the very-low-carbohydrate diet group, and from 8.3% to 7.8% in the low–glycemic diet group, for a between-group comparison P value of .03. In addition, 95% of patients in the low-carbohydrate diet group were able to reduce or eliminate the number of medications they were taking, compared with 62% of patients in the low–glycemic diet group (P less than .01).
Dr. Hallberg and colleagues are currently in year 4 of the 5-year Indiana Type 2 Diabetes Reversal Study, a prospective, nonrandomized, controlled trial of carbohydrate restriction in 465 patients, making it the largest and longest study of its kind. Of the 465 patients, 387 are in the continuous-care arm, which consists of a diet from Virta Health based on principles of nutritional ketosis, and 87 patients in a usual care arm who are followed for 2 years. The trial includes patients who have been prescribed insulin and who have been diagnosed with diabetes for an average of 8 years.
At the meeting, Dr. Hallberg presented preliminary results based on 2 years of data collection. The retention rate was 83% at 1 year and 74% at 2 years. In the treatment arm, the researchers observed that the level of beta hydroxybutyrate, or evidence of ketogenesis, was the same at 2 years as it had been at 1 year. “So, people were still following the diet, as well as being engaged,” she said.
At the end of 2 years, the mean HbA1c reduction was 0.9, the mean reduction for the Homeostatic Model Assessment of Insulin Resistance was 32%, and 55% of completers experienced reversal of their diabetes. Overall, 91% of insulin users reduced or eliminated their use of insulin, and the average weight loss was 10% of baseline weight. “Medication reduction was across the board,” she added. “This is huge from a cost-savings and a patient-satisfaction standpoint. We were improving A1c levels in patients who have had diabetes for an average of over 8 years while we were getting [them] off medication, including insulin. Low carb is now the standard of care.”
Even patients who did not experience a reversal of their diabetes were conferred a benefit. They had an average reduction of 1.2 in HbA1c level, to 7%; their average weight loss was 9.8%; 45% of patients eliminated their diabetes prescriptions; 81% reduced or eliminated their use of insulin; there was an average reduction of 27% in triglyceride levels; and they had a 17% reduction in their 10-year risk score for atherosclerotic cardiovascular disease.
In the overall cohort, the 10-year Atherosclerotic Cardiovascular Disease risk score improved by 12%; almost all markers for cardiovascular disease improved at 1 year. “We were giving these patients appropriate support, which I think is key,” Dr. Hallberg said. “No matter what you do, you have to have a high-touch intervention, and supply that through technology. We do better than medication adherence. Putting patients on a carbohydrate-restricted diet with the appropriate support works for sustainability.”
Dr. Hallberg disclosed that she is an employee of Virta Health and that she is an adviser for Simply Good Foods.
LOS ANGELES – according to Sarah Hallberg, DO.
“Nutritional ketosis supports diabetes reversal by reducing insulin resistance while providing an alternative fuel to glucose with favorable signaling properties,” she said at the World Congress on Insulin Resistance, Diabetes, and Cardiovascular Disease.
Low-carbohydrate nutritional patterns including ketosis have extensive clinical trial evidence for improvement of type 2 diabetes, including preliminary results from a 5-year study of 465 patients enrolled in the Indiana Type 2 Diabetes Reversal Trial that Dr. Hallberg is overseeing in her role as medical director and founder of the medically supervised weight-loss program at Indiana University Health Arnett, Lafayette.
“The ketogenic diet is not a fad diet, it’s what we used to treat people with before the advent of insulin,” said Dr. Hallberg, who has been recommending and counseling patients with type 2 diabetes to follow a ketogenic diet for nearly 10 years. “Of course, insulin has been wonderful. It’s saved so many people with type 1 diabetes. But we also misused it in type 2 diabetes. Instead of counseling people the way we used to about the food that they’re taking in to control their blood sugar, we’ve just been putting [them] on medication, including insulin.”
The American Diabetes Association and other organizations have updated their guidelines to include low-carbohydrate eating patterns for type 2 diabetes treatment, she continued. Veterans Affairs/Department of Defense recommend carbohydrate levels as low as 14%.
Dr. Hallberg, who is also medical director for Virta Health, defined a very-low-carbohydrate or ketogenic diet as less than 50 g of carbohydrates per day, or fewer than 10% of calories consumed. A low-carbohydrate diet is 51-130 g of carbohydrates per day, or 25% or fewer calories consumed, whereas anything above 25% calories consumed is a not a low-carbohydrate diet. A well-formulated ketogenic diet, she continued, consists of 5%-10% carbohydrates (or less than 50 g), 15%-20% protein, and 70%-80% fat. The carbohydrates include 5-10 g per day of protein-based food, 10-15 g of vegetables, 5-10 g of nuts/seeds, 5-10 g of fruits, and 5-10 g of miscellaneous nutrients. “When we’re talking about a total carbohydrate intake per day of under 50 g, you can get a lot of vegetables and nuts in,” she said. “I like to tell my patients they’re not eating GPS: no grains, no potatoes, and no sugar.”
Recently, Dr. Hallberg and colleagues published a review in which they sought to evaluate the appropriateness of sources cited in the ADA’s guidelines on eating patterns for the management of type 2 diabetes, identify additional relevant sources, and evaluate the evidence (Diabetes Obes Metab. 2019;21[8]:1769-79). “We looked at how much evidence there is for the low-carb diet, the Mediterranean diet, the DASH [Dietary Approaches to Stop Hypertension] diet, and a plant-based diet,” she said. “We found a wide variation in the evidence for each eating pattern, but the low-carb eating pattern for diabetes has so much more evidence than any of the other eating patterns.”
In an earlier study, researchers followed 10 inpatients with diabetes in a metabolic ward for 3 weeks. Their mean age was 51 years, and their mean body mass index was 40.3 kg/m2. The patients were fed a standard diet for 7 days, then a low-carbohydrate diet (21 g per day) for 14 days (Ann Intern Med 2005; 142[6]:403-11). After 2 weeks of the low-carbohydrate diet, their mean fasting blood glucose dropped from 7.5 to 6.3 mmol/L, and their mean hemoglobin A1c (HbA1c) fell from 7.3% to 6.8%. “The levels came down very fast,” said Dr. Hallberg, who was not involved with the study. “This is an important part of the intervention, because when you get a patient who’s tried everything, who’s injecting hundreds of units of insulin every day, you can make a huge difference in the first couple of weeks. It is not unusual for us to pull patients off of 200-plus units of insulin. This is as motivating as all get out. It also affects their pocketbook right away. This is one of the reasons our patients are able to sustain a ketogenic diet along with support: early motivation and satisfaction.”
In a longer-term trial, researchers evaluated the impact of a ketogenic diet in 64 obese patients with diabetes over the course of 56 weeks (Moll Cell Biochem. 2007;302[1-2]:249-56). The body weight, body mass index, and levels of blood glucose, total cholesterol, LDL cholesterol, triglycerides, and urea showed a significant decrease from week 1 to week 56 (P less than .0001), while the level of HDL cholesterol increased significantly (P less than .0001).
A separate trial conducted in Israel evaluated the effects of a low-carbohydrate diet, compared with a Mediterranean or low-fat diet in 322 moderately obese patients over the course of 2 years (N Engl J Med. 2008;359:229-41). The rate of adherence to a study diet was 85% at 2 years. The mean weight change was greatest for those on the low-carbohydrate diet, followed by the Mediterranean and low-fat diets. Fasting glucose was best for those on the Mediterranean diet at the end of 2 years, whereas change in HbA1c was best among those on the low-carbohydrate diet.
Another study randomized patients to a low-carbohydrate ketogenic diet (less than 20 g per day with no calorie restriction) or to a low–glycemic index diet (55% carbohydrate restriction of 500 kcal from baseline) over the course of 24 weeks (Nutr Metab [Lond]. 2008 Dec 19. doi:10.1186/1743-7075-5-36). Between baseline and week 24, the mean HbA1c fell from 8.8% to 7.3% in the very-low-carbohydrate diet group, and from 8.3% to 7.8% in the low–glycemic diet group, for a between-group comparison P value of .03. In addition, 95% of patients in the low-carbohydrate diet group were able to reduce or eliminate the number of medications they were taking, compared with 62% of patients in the low–glycemic diet group (P less than .01).
Dr. Hallberg and colleagues are currently in year 4 of the 5-year Indiana Type 2 Diabetes Reversal Study, a prospective, nonrandomized, controlled trial of carbohydrate restriction in 465 patients, making it the largest and longest study of its kind. Of the 465 patients, 387 are in the continuous-care arm, which consists of a diet from Virta Health based on principles of nutritional ketosis, and 87 patients in a usual care arm who are followed for 2 years. The trial includes patients who have been prescribed insulin and who have been diagnosed with diabetes for an average of 8 years.
At the meeting, Dr. Hallberg presented preliminary results based on 2 years of data collection. The retention rate was 83% at 1 year and 74% at 2 years. In the treatment arm, the researchers observed that the level of beta hydroxybutyrate, or evidence of ketogenesis, was the same at 2 years as it had been at 1 year. “So, people were still following the diet, as well as being engaged,” she said.
At the end of 2 years, the mean HbA1c reduction was 0.9, the mean reduction for the Homeostatic Model Assessment of Insulin Resistance was 32%, and 55% of completers experienced reversal of their diabetes. Overall, 91% of insulin users reduced or eliminated their use of insulin, and the average weight loss was 10% of baseline weight. “Medication reduction was across the board,” she added. “This is huge from a cost-savings and a patient-satisfaction standpoint. We were improving A1c levels in patients who have had diabetes for an average of over 8 years while we were getting [them] off medication, including insulin. Low carb is now the standard of care.”
Even patients who did not experience a reversal of their diabetes were conferred a benefit. They had an average reduction of 1.2 in HbA1c level, to 7%; their average weight loss was 9.8%; 45% of patients eliminated their diabetes prescriptions; 81% reduced or eliminated their use of insulin; there was an average reduction of 27% in triglyceride levels; and they had a 17% reduction in their 10-year risk score for atherosclerotic cardiovascular disease.
In the overall cohort, the 10-year Atherosclerotic Cardiovascular Disease risk score improved by 12%; almost all markers for cardiovascular disease improved at 1 year. “We were giving these patients appropriate support, which I think is key,” Dr. Hallberg said. “No matter what you do, you have to have a high-touch intervention, and supply that through technology. We do better than medication adherence. Putting patients on a carbohydrate-restricted diet with the appropriate support works for sustainability.”
Dr. Hallberg disclosed that she is an employee of Virta Health and that she is an adviser for Simply Good Foods.
LOS ANGELES – according to Sarah Hallberg, DO.
“Nutritional ketosis supports diabetes reversal by reducing insulin resistance while providing an alternative fuel to glucose with favorable signaling properties,” she said at the World Congress on Insulin Resistance, Diabetes, and Cardiovascular Disease.
Low-carbohydrate nutritional patterns including ketosis have extensive clinical trial evidence for improvement of type 2 diabetes, including preliminary results from a 5-year study of 465 patients enrolled in the Indiana Type 2 Diabetes Reversal Trial that Dr. Hallberg is overseeing in her role as medical director and founder of the medically supervised weight-loss program at Indiana University Health Arnett, Lafayette.
“The ketogenic diet is not a fad diet, it’s what we used to treat people with before the advent of insulin,” said Dr. Hallberg, who has been recommending and counseling patients with type 2 diabetes to follow a ketogenic diet for nearly 10 years. “Of course, insulin has been wonderful. It’s saved so many people with type 1 diabetes. But we also misused it in type 2 diabetes. Instead of counseling people the way we used to about the food that they’re taking in to control their blood sugar, we’ve just been putting [them] on medication, including insulin.”
The American Diabetes Association and other organizations have updated their guidelines to include low-carbohydrate eating patterns for type 2 diabetes treatment, she continued. Veterans Affairs/Department of Defense recommend carbohydrate levels as low as 14%.
Dr. Hallberg, who is also medical director for Virta Health, defined a very-low-carbohydrate or ketogenic diet as less than 50 g of carbohydrates per day, or fewer than 10% of calories consumed. A low-carbohydrate diet is 51-130 g of carbohydrates per day, or 25% or fewer calories consumed, whereas anything above 25% calories consumed is a not a low-carbohydrate diet. A well-formulated ketogenic diet, she continued, consists of 5%-10% carbohydrates (or less than 50 g), 15%-20% protein, and 70%-80% fat. The carbohydrates include 5-10 g per day of protein-based food, 10-15 g of vegetables, 5-10 g of nuts/seeds, 5-10 g of fruits, and 5-10 g of miscellaneous nutrients. “When we’re talking about a total carbohydrate intake per day of under 50 g, you can get a lot of vegetables and nuts in,” she said. “I like to tell my patients they’re not eating GPS: no grains, no potatoes, and no sugar.”
Recently, Dr. Hallberg and colleagues published a review in which they sought to evaluate the appropriateness of sources cited in the ADA’s guidelines on eating patterns for the management of type 2 diabetes, identify additional relevant sources, and evaluate the evidence (Diabetes Obes Metab. 2019;21[8]:1769-79). “We looked at how much evidence there is for the low-carb diet, the Mediterranean diet, the DASH [Dietary Approaches to Stop Hypertension] diet, and a plant-based diet,” she said. “We found a wide variation in the evidence for each eating pattern, but the low-carb eating pattern for diabetes has so much more evidence than any of the other eating patterns.”
In an earlier study, researchers followed 10 inpatients with diabetes in a metabolic ward for 3 weeks. Their mean age was 51 years, and their mean body mass index was 40.3 kg/m2. The patients were fed a standard diet for 7 days, then a low-carbohydrate diet (21 g per day) for 14 days (Ann Intern Med 2005; 142[6]:403-11). After 2 weeks of the low-carbohydrate diet, their mean fasting blood glucose dropped from 7.5 to 6.3 mmol/L, and their mean hemoglobin A1c (HbA1c) fell from 7.3% to 6.8%. “The levels came down very fast,” said Dr. Hallberg, who was not involved with the study. “This is an important part of the intervention, because when you get a patient who’s tried everything, who’s injecting hundreds of units of insulin every day, you can make a huge difference in the first couple of weeks. It is not unusual for us to pull patients off of 200-plus units of insulin. This is as motivating as all get out. It also affects their pocketbook right away. This is one of the reasons our patients are able to sustain a ketogenic diet along with support: early motivation and satisfaction.”
In a longer-term trial, researchers evaluated the impact of a ketogenic diet in 64 obese patients with diabetes over the course of 56 weeks (Moll Cell Biochem. 2007;302[1-2]:249-56). The body weight, body mass index, and levels of blood glucose, total cholesterol, LDL cholesterol, triglycerides, and urea showed a significant decrease from week 1 to week 56 (P less than .0001), while the level of HDL cholesterol increased significantly (P less than .0001).
A separate trial conducted in Israel evaluated the effects of a low-carbohydrate diet, compared with a Mediterranean or low-fat diet in 322 moderately obese patients over the course of 2 years (N Engl J Med. 2008;359:229-41). The rate of adherence to a study diet was 85% at 2 years. The mean weight change was greatest for those on the low-carbohydrate diet, followed by the Mediterranean and low-fat diets. Fasting glucose was best for those on the Mediterranean diet at the end of 2 years, whereas change in HbA1c was best among those on the low-carbohydrate diet.
Another study randomized patients to a low-carbohydrate ketogenic diet (less than 20 g per day with no calorie restriction) or to a low–glycemic index diet (55% carbohydrate restriction of 500 kcal from baseline) over the course of 24 weeks (Nutr Metab [Lond]. 2008 Dec 19. doi:10.1186/1743-7075-5-36). Between baseline and week 24, the mean HbA1c fell from 8.8% to 7.3% in the very-low-carbohydrate diet group, and from 8.3% to 7.8% in the low–glycemic diet group, for a between-group comparison P value of .03. In addition, 95% of patients in the low-carbohydrate diet group were able to reduce or eliminate the number of medications they were taking, compared with 62% of patients in the low–glycemic diet group (P less than .01).
Dr. Hallberg and colleagues are currently in year 4 of the 5-year Indiana Type 2 Diabetes Reversal Study, a prospective, nonrandomized, controlled trial of carbohydrate restriction in 465 patients, making it the largest and longest study of its kind. Of the 465 patients, 387 are in the continuous-care arm, which consists of a diet from Virta Health based on principles of nutritional ketosis, and 87 patients in a usual care arm who are followed for 2 years. The trial includes patients who have been prescribed insulin and who have been diagnosed with diabetes for an average of 8 years.
At the meeting, Dr. Hallberg presented preliminary results based on 2 years of data collection. The retention rate was 83% at 1 year and 74% at 2 years. In the treatment arm, the researchers observed that the level of beta hydroxybutyrate, or evidence of ketogenesis, was the same at 2 years as it had been at 1 year. “So, people were still following the diet, as well as being engaged,” she said.
At the end of 2 years, the mean HbA1c reduction was 0.9, the mean reduction for the Homeostatic Model Assessment of Insulin Resistance was 32%, and 55% of completers experienced reversal of their diabetes. Overall, 91% of insulin users reduced or eliminated their use of insulin, and the average weight loss was 10% of baseline weight. “Medication reduction was across the board,” she added. “This is huge from a cost-savings and a patient-satisfaction standpoint. We were improving A1c levels in patients who have had diabetes for an average of over 8 years while we were getting [them] off medication, including insulin. Low carb is now the standard of care.”
Even patients who did not experience a reversal of their diabetes were conferred a benefit. They had an average reduction of 1.2 in HbA1c level, to 7%; their average weight loss was 9.8%; 45% of patients eliminated their diabetes prescriptions; 81% reduced or eliminated their use of insulin; there was an average reduction of 27% in triglyceride levels; and they had a 17% reduction in their 10-year risk score for atherosclerotic cardiovascular disease.
In the overall cohort, the 10-year Atherosclerotic Cardiovascular Disease risk score improved by 12%; almost all markers for cardiovascular disease improved at 1 year. “We were giving these patients appropriate support, which I think is key,” Dr. Hallberg said. “No matter what you do, you have to have a high-touch intervention, and supply that through technology. We do better than medication adherence. Putting patients on a carbohydrate-restricted diet with the appropriate support works for sustainability.”
Dr. Hallberg disclosed that she is an employee of Virta Health and that she is an adviser for Simply Good Foods.
EXPERT ANALYSIS FROM WCIRDC 2019
Analyses clarify who benefits from ARNI-ARB combination
PHILADELPHIA – Two clinical trials of the combination therapy of the neprilysin inhibitor sacubitril and the angiotensin II receptor blocker valsartan in patients with heart failure and reduced ejection fraction found that it lowered rates of all-cause death, compared to a renin-angiotensin-system inhibitor alone.
Furthermore, the treatment produced a more beneficial effect in women, who are more prone to heart failure and preserved ejection fraction (HFpEF), lead investigators reported at the American Heart Association scientific sessions.
A prespecified subgroup analysis of 4,796 patients in the PARAGON-HF trial found that the sacubitril/valsartan, or sac/val, combination had a significantly more beneficial risk reduction of first and recurrent hospitalizations for heart failure, as well as cardiovascular death, in women than men. A prespecified pooled analysis of 13,195 patients in the PARAGON-HF and the PARADIGM-HF trials also found women derived a greater benefit from the combination therapy than men, but also concluded that patients with heart failure and even mildly reduced ejection fraction had better outcomes. The results of both studies were published simultaneously with the presentations on Nov. 17 in Circulation (doi: 10.1161/circulationaha.119.044491; doi: 10.1161/circulationaha.119.044586).
The findings underscore the effectiveness of sac/val combination in patients with HF and EF in the lower ranges, defined as 40% or less, commented discussant Lynne Warner Stevenson, MD, of Vanderbilt Heart and Vascular Institute in Nashville, Tenn. “We all agree now that the use of sacubitril/valsartan is very appropriate to improve outcomes in those patients, even if they’ve never been hospitalized,” she said in an interview.
PARAGON-HF subanalysis
John J.V. McMurray, MD, of the University of Glasgow presented the PARAGON-HF subgroup analysis. He said it initially focused on 12 subgroups, but that only two baseline variables showed a modified effect of sac/val: sex and left-ventricle ejection fraction (LVEF). The findings, he said, “stood up in a very robust, multivariable analysis.”
The women in the subgroup analysis were older, had higher baseline New York Heart Association class status, and worse quality of life as measured by Kansas City Cardiomyopathy Questionnaire clinical summary score. At baseline, women also had higher average LVEF (59% vs. 56%), lower N-terminal prohormone brain natriuretic peptide levels, and higher rates of renal dysfunction and chronic kidney disease, but lower incidence of a previous MI and coronary artery disease. Prestudy treatments were similar between the sexes.
In terms of the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – “there was an apparent 27% relative risk reduction in women and no overall effect in men,” Dr. McMurray said of the treatment group. “The difference was driven completely by hospitalizations.” Rates of CV death were similar between the valsartan-only and sac/val groups in both men and women, he said.
In the analysis of LVEF, women in the treatment group seemed to cross over to a heightened risk of hospitalization and CV death at an LVEF in the 60%-65% range, Dr. McMurray said, whereas men made that cross over in the 50%-55% range. “It looks as though women might be getting more benefit from this treatment up to a higher EF than in men,” he said.
However, the differences between men and women did not hold up in the analysis of secondary outcomes. At 8 months, women in the sac/val group had a 0.6-point greater decline than did the valsartan-only patients in KCCQ-CSS score, whereas men on sac/val had a 2.8-point lesser decline than did those on valsartan only. Similar differences were seen between the treatment and valsartan-only groups within the sexes, with women showing a noticeable improvement surpassing the men.
Posttreatment hypotension rates in both sexes were higher in the sac/val groups, and the risk of renal dysfunction was a bit less in both treatment groups. Women in the treatment group had significantly higher rates of angioedema than did the valsartan-only group and men in either group.
“Compared to valsartan, it’s important to say that sacubitril/valsartan seemed to reduce the risk of heart failure and hospitalization more in women than men, but we didn’t find a similar differential for other endpoints,” Dr. McMurray said. “Therefore, we’re not sure this is a real effect or a chance finding. It’s very statistically robust, but it could still be a chance finding.”
A possible explanation could be than men may not be responding to sac/val, or that valsartan alone may be more effective in men than women, he said. “This possible effect modification of sac/val vs. valsartan by sex deserves further investigation,” he said.
PARAGON-HF and PARADIGM-HF pooled analysis
Likewise, the prespecified pooled analysis of the PARADIGM-HF and PARAGON-HF trials found a greater benefit of sac/val in women, according to results presented by Scott D. Solomon, MD, of Brigham and Women’s Hospital in Boston. Where PARAGON-HF compared combination therapy with valsartan 160 mg twice daily alone, PARADIGM-HF used enalapril 10 mg twice daily alone as the comparator renin-angiotensin-system (RAS) inhibitor.
“These data suggest that the therapeutic effect of sacubitril/valsartan vs. RAS inhibition alone appear to extend to patients with heart failure and mildly reduced EF, with therapeutic benefits that extend to a higher left-ventricle EF range in women compared to men,” Dr. Solomon said.
The pooled analysis divided patients into six different EF groups: up to 22.5%, then in 10-point increments from 22.5% to 62.5%, and 62.5% or greater. PARADIGM-HF enrolled patients age 18 years and older, whereas PARAGON-HF involved those aged 50 years and older.
The analysis showed that, as LVEF rates increased across the EF groups, the rates of the primary composite outcome – HF hospitalizations, CV death, and all-cause mortality – decreased, but the decline was greatest for CV death and less so for HF hospitalization. And while rates of all-cause mortality decreased as EF increased, rates of non-CV death increased substantially with increasing LVEF.
“For each of these endpoints, there are significant benefits to sacubitril/valsartan in the pooled analysis, and this includes HF hospitalization, CV death, either total or first events, and all-cause mortality, which was reduced overall by 12% in the combination group,” Dr. Solomon said. That benefit was seen in the first five categories of EF, but all but disappeared in the highest category (at least 62.5%), he said.
At the lower end of the EF spectrum, the effect of sac/val is more pronounced and similar for men and women, Dr. Solomon said. “But as EF goes up, we see an attenuation of that effect in both men and women, but it occurs at a different point,” he said. “Women seem to derive a benefit to a higher ejection fraction than men.” As in Dr. McMurray’s research, the benefit seems to extend to LVEF of 55%-60% in men and 65%-70% in women.
“These findings were driven by an observed benefit in patients with chronic heart failure and LVEF below the normal range,” he said. “The benefit in the EF range above the ranking ‘reduced’ but below normal was driven primarily by reduction in HF hospitalization.”
Dr. Stevenson said that these findings indicate that a previous hospitalization for HF with preserved EF may be a telling marker for the effectiveness of sac/val. “As opposed to the patient who has exertional dyspnea but has never decompensated to the level needing hospitalization, if they have pEF, our current analyses would suggest sac/val may not offer them much benefit,” she said.
In real-world practice, cost would be an issue, Dr. Stevenson said. “This drug is very expensive; the majority of patients pay more than $100 a month in out-of-pocket costs, and we have to recognize this is not a therapy that everyone can afford,” she said in an interview. “In many areas, and particularly in the disadvantaged populations, this is not going to be a therapy that we’re going to be able to offer everyone, and that gives me great concern as we move toward trying to treat the whole disease that we’re developing therapies that will be limited by finance rather than by physiology. That’s a major call to action for all of us.”
Novartis sponsored the studies. Dr. McMurray has no disclosures. Dr. Solomon disclosed financial relationships with trial sponsor Novartis along with numerous pharmaceutical companies and the National Heart, Lung, and Blood Institute.
SOURCE: McMurray JJ and Solomon SD. AHA 2019, Late Breaking Science Session 5.
PHILADELPHIA – Two clinical trials of the combination therapy of the neprilysin inhibitor sacubitril and the angiotensin II receptor blocker valsartan in patients with heart failure and reduced ejection fraction found that it lowered rates of all-cause death, compared to a renin-angiotensin-system inhibitor alone.
Furthermore, the treatment produced a more beneficial effect in women, who are more prone to heart failure and preserved ejection fraction (HFpEF), lead investigators reported at the American Heart Association scientific sessions.
A prespecified subgroup analysis of 4,796 patients in the PARAGON-HF trial found that the sacubitril/valsartan, or sac/val, combination had a significantly more beneficial risk reduction of first and recurrent hospitalizations for heart failure, as well as cardiovascular death, in women than men. A prespecified pooled analysis of 13,195 patients in the PARAGON-HF and the PARADIGM-HF trials also found women derived a greater benefit from the combination therapy than men, but also concluded that patients with heart failure and even mildly reduced ejection fraction had better outcomes. The results of both studies were published simultaneously with the presentations on Nov. 17 in Circulation (doi: 10.1161/circulationaha.119.044491; doi: 10.1161/circulationaha.119.044586).
The findings underscore the effectiveness of sac/val combination in patients with HF and EF in the lower ranges, defined as 40% or less, commented discussant Lynne Warner Stevenson, MD, of Vanderbilt Heart and Vascular Institute in Nashville, Tenn. “We all agree now that the use of sacubitril/valsartan is very appropriate to improve outcomes in those patients, even if they’ve never been hospitalized,” she said in an interview.
PARAGON-HF subanalysis
John J.V. McMurray, MD, of the University of Glasgow presented the PARAGON-HF subgroup analysis. He said it initially focused on 12 subgroups, but that only two baseline variables showed a modified effect of sac/val: sex and left-ventricle ejection fraction (LVEF). The findings, he said, “stood up in a very robust, multivariable analysis.”
The women in the subgroup analysis were older, had higher baseline New York Heart Association class status, and worse quality of life as measured by Kansas City Cardiomyopathy Questionnaire clinical summary score. At baseline, women also had higher average LVEF (59% vs. 56%), lower N-terminal prohormone brain natriuretic peptide levels, and higher rates of renal dysfunction and chronic kidney disease, but lower incidence of a previous MI and coronary artery disease. Prestudy treatments were similar between the sexes.
In terms of the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – “there was an apparent 27% relative risk reduction in women and no overall effect in men,” Dr. McMurray said of the treatment group. “The difference was driven completely by hospitalizations.” Rates of CV death were similar between the valsartan-only and sac/val groups in both men and women, he said.
In the analysis of LVEF, women in the treatment group seemed to cross over to a heightened risk of hospitalization and CV death at an LVEF in the 60%-65% range, Dr. McMurray said, whereas men made that cross over in the 50%-55% range. “It looks as though women might be getting more benefit from this treatment up to a higher EF than in men,” he said.
However, the differences between men and women did not hold up in the analysis of secondary outcomes. At 8 months, women in the sac/val group had a 0.6-point greater decline than did the valsartan-only patients in KCCQ-CSS score, whereas men on sac/val had a 2.8-point lesser decline than did those on valsartan only. Similar differences were seen between the treatment and valsartan-only groups within the sexes, with women showing a noticeable improvement surpassing the men.
Posttreatment hypotension rates in both sexes were higher in the sac/val groups, and the risk of renal dysfunction was a bit less in both treatment groups. Women in the treatment group had significantly higher rates of angioedema than did the valsartan-only group and men in either group.
“Compared to valsartan, it’s important to say that sacubitril/valsartan seemed to reduce the risk of heart failure and hospitalization more in women than men, but we didn’t find a similar differential for other endpoints,” Dr. McMurray said. “Therefore, we’re not sure this is a real effect or a chance finding. It’s very statistically robust, but it could still be a chance finding.”
A possible explanation could be than men may not be responding to sac/val, or that valsartan alone may be more effective in men than women, he said. “This possible effect modification of sac/val vs. valsartan by sex deserves further investigation,” he said.
PARAGON-HF and PARADIGM-HF pooled analysis
Likewise, the prespecified pooled analysis of the PARADIGM-HF and PARAGON-HF trials found a greater benefit of sac/val in women, according to results presented by Scott D. Solomon, MD, of Brigham and Women’s Hospital in Boston. Where PARAGON-HF compared combination therapy with valsartan 160 mg twice daily alone, PARADIGM-HF used enalapril 10 mg twice daily alone as the comparator renin-angiotensin-system (RAS) inhibitor.
“These data suggest that the therapeutic effect of sacubitril/valsartan vs. RAS inhibition alone appear to extend to patients with heart failure and mildly reduced EF, with therapeutic benefits that extend to a higher left-ventricle EF range in women compared to men,” Dr. Solomon said.
The pooled analysis divided patients into six different EF groups: up to 22.5%, then in 10-point increments from 22.5% to 62.5%, and 62.5% or greater. PARADIGM-HF enrolled patients age 18 years and older, whereas PARAGON-HF involved those aged 50 years and older.
The analysis showed that, as LVEF rates increased across the EF groups, the rates of the primary composite outcome – HF hospitalizations, CV death, and all-cause mortality – decreased, but the decline was greatest for CV death and less so for HF hospitalization. And while rates of all-cause mortality decreased as EF increased, rates of non-CV death increased substantially with increasing LVEF.
“For each of these endpoints, there are significant benefits to sacubitril/valsartan in the pooled analysis, and this includes HF hospitalization, CV death, either total or first events, and all-cause mortality, which was reduced overall by 12% in the combination group,” Dr. Solomon said. That benefit was seen in the first five categories of EF, but all but disappeared in the highest category (at least 62.5%), he said.
At the lower end of the EF spectrum, the effect of sac/val is more pronounced and similar for men and women, Dr. Solomon said. “But as EF goes up, we see an attenuation of that effect in both men and women, but it occurs at a different point,” he said. “Women seem to derive a benefit to a higher ejection fraction than men.” As in Dr. McMurray’s research, the benefit seems to extend to LVEF of 55%-60% in men and 65%-70% in women.
“These findings were driven by an observed benefit in patients with chronic heart failure and LVEF below the normal range,” he said. “The benefit in the EF range above the ranking ‘reduced’ but below normal was driven primarily by reduction in HF hospitalization.”
Dr. Stevenson said that these findings indicate that a previous hospitalization for HF with preserved EF may be a telling marker for the effectiveness of sac/val. “As opposed to the patient who has exertional dyspnea but has never decompensated to the level needing hospitalization, if they have pEF, our current analyses would suggest sac/val may not offer them much benefit,” she said.
In real-world practice, cost would be an issue, Dr. Stevenson said. “This drug is very expensive; the majority of patients pay more than $100 a month in out-of-pocket costs, and we have to recognize this is not a therapy that everyone can afford,” she said in an interview. “In many areas, and particularly in the disadvantaged populations, this is not going to be a therapy that we’re going to be able to offer everyone, and that gives me great concern as we move toward trying to treat the whole disease that we’re developing therapies that will be limited by finance rather than by physiology. That’s a major call to action for all of us.”
Novartis sponsored the studies. Dr. McMurray has no disclosures. Dr. Solomon disclosed financial relationships with trial sponsor Novartis along with numerous pharmaceutical companies and the National Heart, Lung, and Blood Institute.
SOURCE: McMurray JJ and Solomon SD. AHA 2019, Late Breaking Science Session 5.
PHILADELPHIA – Two clinical trials of the combination therapy of the neprilysin inhibitor sacubitril and the angiotensin II receptor blocker valsartan in patients with heart failure and reduced ejection fraction found that it lowered rates of all-cause death, compared to a renin-angiotensin-system inhibitor alone.
Furthermore, the treatment produced a more beneficial effect in women, who are more prone to heart failure and preserved ejection fraction (HFpEF), lead investigators reported at the American Heart Association scientific sessions.
A prespecified subgroup analysis of 4,796 patients in the PARAGON-HF trial found that the sacubitril/valsartan, or sac/val, combination had a significantly more beneficial risk reduction of first and recurrent hospitalizations for heart failure, as well as cardiovascular death, in women than men. A prespecified pooled analysis of 13,195 patients in the PARAGON-HF and the PARADIGM-HF trials also found women derived a greater benefit from the combination therapy than men, but also concluded that patients with heart failure and even mildly reduced ejection fraction had better outcomes. The results of both studies were published simultaneously with the presentations on Nov. 17 in Circulation (doi: 10.1161/circulationaha.119.044491; doi: 10.1161/circulationaha.119.044586).
The findings underscore the effectiveness of sac/val combination in patients with HF and EF in the lower ranges, defined as 40% or less, commented discussant Lynne Warner Stevenson, MD, of Vanderbilt Heart and Vascular Institute in Nashville, Tenn. “We all agree now that the use of sacubitril/valsartan is very appropriate to improve outcomes in those patients, even if they’ve never been hospitalized,” she said in an interview.
PARAGON-HF subanalysis
John J.V. McMurray, MD, of the University of Glasgow presented the PARAGON-HF subgroup analysis. He said it initially focused on 12 subgroups, but that only two baseline variables showed a modified effect of sac/val: sex and left-ventricle ejection fraction (LVEF). The findings, he said, “stood up in a very robust, multivariable analysis.”
The women in the subgroup analysis were older, had higher baseline New York Heart Association class status, and worse quality of life as measured by Kansas City Cardiomyopathy Questionnaire clinical summary score. At baseline, women also had higher average LVEF (59% vs. 56%), lower N-terminal prohormone brain natriuretic peptide levels, and higher rates of renal dysfunction and chronic kidney disease, but lower incidence of a previous MI and coronary artery disease. Prestudy treatments were similar between the sexes.
In terms of the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – “there was an apparent 27% relative risk reduction in women and no overall effect in men,” Dr. McMurray said of the treatment group. “The difference was driven completely by hospitalizations.” Rates of CV death were similar between the valsartan-only and sac/val groups in both men and women, he said.
In the analysis of LVEF, women in the treatment group seemed to cross over to a heightened risk of hospitalization and CV death at an LVEF in the 60%-65% range, Dr. McMurray said, whereas men made that cross over in the 50%-55% range. “It looks as though women might be getting more benefit from this treatment up to a higher EF than in men,” he said.
However, the differences between men and women did not hold up in the analysis of secondary outcomes. At 8 months, women in the sac/val group had a 0.6-point greater decline than did the valsartan-only patients in KCCQ-CSS score, whereas men on sac/val had a 2.8-point lesser decline than did those on valsartan only. Similar differences were seen between the treatment and valsartan-only groups within the sexes, with women showing a noticeable improvement surpassing the men.
Posttreatment hypotension rates in both sexes were higher in the sac/val groups, and the risk of renal dysfunction was a bit less in both treatment groups. Women in the treatment group had significantly higher rates of angioedema than did the valsartan-only group and men in either group.
“Compared to valsartan, it’s important to say that sacubitril/valsartan seemed to reduce the risk of heart failure and hospitalization more in women than men, but we didn’t find a similar differential for other endpoints,” Dr. McMurray said. “Therefore, we’re not sure this is a real effect or a chance finding. It’s very statistically robust, but it could still be a chance finding.”
A possible explanation could be than men may not be responding to sac/val, or that valsartan alone may be more effective in men than women, he said. “This possible effect modification of sac/val vs. valsartan by sex deserves further investigation,” he said.
PARAGON-HF and PARADIGM-HF pooled analysis
Likewise, the prespecified pooled analysis of the PARADIGM-HF and PARAGON-HF trials found a greater benefit of sac/val in women, according to results presented by Scott D. Solomon, MD, of Brigham and Women’s Hospital in Boston. Where PARAGON-HF compared combination therapy with valsartan 160 mg twice daily alone, PARADIGM-HF used enalapril 10 mg twice daily alone as the comparator renin-angiotensin-system (RAS) inhibitor.
“These data suggest that the therapeutic effect of sacubitril/valsartan vs. RAS inhibition alone appear to extend to patients with heart failure and mildly reduced EF, with therapeutic benefits that extend to a higher left-ventricle EF range in women compared to men,” Dr. Solomon said.
The pooled analysis divided patients into six different EF groups: up to 22.5%, then in 10-point increments from 22.5% to 62.5%, and 62.5% or greater. PARADIGM-HF enrolled patients age 18 years and older, whereas PARAGON-HF involved those aged 50 years and older.
The analysis showed that, as LVEF rates increased across the EF groups, the rates of the primary composite outcome – HF hospitalizations, CV death, and all-cause mortality – decreased, but the decline was greatest for CV death and less so for HF hospitalization. And while rates of all-cause mortality decreased as EF increased, rates of non-CV death increased substantially with increasing LVEF.
“For each of these endpoints, there are significant benefits to sacubitril/valsartan in the pooled analysis, and this includes HF hospitalization, CV death, either total or first events, and all-cause mortality, which was reduced overall by 12% in the combination group,” Dr. Solomon said. That benefit was seen in the first five categories of EF, but all but disappeared in the highest category (at least 62.5%), he said.
At the lower end of the EF spectrum, the effect of sac/val is more pronounced and similar for men and women, Dr. Solomon said. “But as EF goes up, we see an attenuation of that effect in both men and women, but it occurs at a different point,” he said. “Women seem to derive a benefit to a higher ejection fraction than men.” As in Dr. McMurray’s research, the benefit seems to extend to LVEF of 55%-60% in men and 65%-70% in women.
“These findings were driven by an observed benefit in patients with chronic heart failure and LVEF below the normal range,” he said. “The benefit in the EF range above the ranking ‘reduced’ but below normal was driven primarily by reduction in HF hospitalization.”
Dr. Stevenson said that these findings indicate that a previous hospitalization for HF with preserved EF may be a telling marker for the effectiveness of sac/val. “As opposed to the patient who has exertional dyspnea but has never decompensated to the level needing hospitalization, if they have pEF, our current analyses would suggest sac/val may not offer them much benefit,” she said.
In real-world practice, cost would be an issue, Dr. Stevenson said. “This drug is very expensive; the majority of patients pay more than $100 a month in out-of-pocket costs, and we have to recognize this is not a therapy that everyone can afford,” she said in an interview. “In many areas, and particularly in the disadvantaged populations, this is not going to be a therapy that we’re going to be able to offer everyone, and that gives me great concern as we move toward trying to treat the whole disease that we’re developing therapies that will be limited by finance rather than by physiology. That’s a major call to action for all of us.”
Novartis sponsored the studies. Dr. McMurray has no disclosures. Dr. Solomon disclosed financial relationships with trial sponsor Novartis along with numerous pharmaceutical companies and the National Heart, Lung, and Blood Institute.
SOURCE: McMurray JJ and Solomon SD. AHA 2019, Late Breaking Science Session 5.
REPORTING FROM THE AHA SCIENTIFIC SESSIONS
Score predicts bariatric surgery’s benefits for obesity, type 2 diabetes
LAS VEGAS –
The Individualized Diabetes Complications risk score “can provide personalized, evidence-based risk information for patients with type 2 diabetes and obesity about their future cardiovascular disease outcomes and mortality with and without metabolic surgery,” Ali Aminian, MD, said at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
Although the calculator needs validation in a prospective, randomized study to document its impact on practice, it is now available on two separate websites and as a downloadable app, said Dr. Aminian, a surgeon at the Cleveland Clinic.
The calculator inputs data for 26 distinct, “readily available” demographic and clinical entries, and based on that, estimates the patient’s 10-year risk for all-cause death, diabetic kidney disease, cerebrovascular disease, heart failure, and coronary artery disease if no surgery occurs or after some type of metabolic or bariatric surgery. The calculator does not currently have the ability to individualize predicted risks based on the specific type of metabolic surgery performed, but that is planned as a future refinement of the score.
“We validated the model in the nonsurgical patients, which showed it was very accurate. The next step is to run a randomized trial to see how useful the calculator is” for assisting in patients’ decision making, Dr. Aminian said.
The data for deriving the risk calculator, and for a preliminary validation of it, came from 13,722 patients with obesity (body mass index, 30 kg/m2 or greater) and type 2 diabetes, who were managed at the Cleveland Clinic during 1998-2017, drawn from more than 287,000 such patients in the clinic’s database. The study focused on 2,287 patients who underwent metabolic (bariatric) surgery and 11,435 patients from the same database who did not have surgery and matched by propensity scoring on a 5:1 basis with those who had surgery. The two cohorts this created matched well for age (about 54 years), sex (about two-thirds women), body mass index (about 44 kg/m2), and the prevalence of various comorbidities at baseline.
Dr. Aminian and associates then analyzed the incidence of all-cause mortality and various cardiovascular disease endpoints, as well as nephropathy during follow-up, through December 2018. Patients who had undergone metabolic surgery showed statistically significant reductions in the incidence of each of those events, compared with patients who did not have surgery (JAMA. 2019;322[13]:1271-82).
The investigators used these findings to create their model for calculating a patient’s risk score. For example, to calculate an estimate for the 10-year risk from all-cause mortality, the results showed that the most powerful risk factors were age; baseline body mass index, heart failure, and need for insulin; and smoking status. For the endpoint of nephropathy, the most important factors were estimated glomerular filtration rate at baseline and age. Identified risk factors could account for about 80% of the 10-year risk for all-cause death and for about 75% of the risk for developing nephropathy during 10 years, based on the area-under-the-curve values the model produced.
The risk score may help patients better understand the potential role that metabolic surgery can have in reducing their future event risk, thereby helping them better appreciate the benefit they stand to gain from undergoing surgery, Dr. Aminian said.
The calculator is available at a website maintained by the Cleveland Clinic, at a site of the American Society for Metabolic and Bariatric Surgery, and in app stores, he said.
The work was partially funded by Medtronic. Dr. Aminian has received grants from Medtronic.
SOURCE: Aminian A et al. Obesity Week 2019, Abstract A101.
LAS VEGAS –
The Individualized Diabetes Complications risk score “can provide personalized, evidence-based risk information for patients with type 2 diabetes and obesity about their future cardiovascular disease outcomes and mortality with and without metabolic surgery,” Ali Aminian, MD, said at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
Although the calculator needs validation in a prospective, randomized study to document its impact on practice, it is now available on two separate websites and as a downloadable app, said Dr. Aminian, a surgeon at the Cleveland Clinic.
The calculator inputs data for 26 distinct, “readily available” demographic and clinical entries, and based on that, estimates the patient’s 10-year risk for all-cause death, diabetic kidney disease, cerebrovascular disease, heart failure, and coronary artery disease if no surgery occurs or after some type of metabolic or bariatric surgery. The calculator does not currently have the ability to individualize predicted risks based on the specific type of metabolic surgery performed, but that is planned as a future refinement of the score.
“We validated the model in the nonsurgical patients, which showed it was very accurate. The next step is to run a randomized trial to see how useful the calculator is” for assisting in patients’ decision making, Dr. Aminian said.
The data for deriving the risk calculator, and for a preliminary validation of it, came from 13,722 patients with obesity (body mass index, 30 kg/m2 or greater) and type 2 diabetes, who were managed at the Cleveland Clinic during 1998-2017, drawn from more than 287,000 such patients in the clinic’s database. The study focused on 2,287 patients who underwent metabolic (bariatric) surgery and 11,435 patients from the same database who did not have surgery and matched by propensity scoring on a 5:1 basis with those who had surgery. The two cohorts this created matched well for age (about 54 years), sex (about two-thirds women), body mass index (about 44 kg/m2), and the prevalence of various comorbidities at baseline.
Dr. Aminian and associates then analyzed the incidence of all-cause mortality and various cardiovascular disease endpoints, as well as nephropathy during follow-up, through December 2018. Patients who had undergone metabolic surgery showed statistically significant reductions in the incidence of each of those events, compared with patients who did not have surgery (JAMA. 2019;322[13]:1271-82).
The investigators used these findings to create their model for calculating a patient’s risk score. For example, to calculate an estimate for the 10-year risk from all-cause mortality, the results showed that the most powerful risk factors were age; baseline body mass index, heart failure, and need for insulin; and smoking status. For the endpoint of nephropathy, the most important factors were estimated glomerular filtration rate at baseline and age. Identified risk factors could account for about 80% of the 10-year risk for all-cause death and for about 75% of the risk for developing nephropathy during 10 years, based on the area-under-the-curve values the model produced.
The risk score may help patients better understand the potential role that metabolic surgery can have in reducing their future event risk, thereby helping them better appreciate the benefit they stand to gain from undergoing surgery, Dr. Aminian said.
The calculator is available at a website maintained by the Cleveland Clinic, at a site of the American Society for Metabolic and Bariatric Surgery, and in app stores, he said.
The work was partially funded by Medtronic. Dr. Aminian has received grants from Medtronic.
SOURCE: Aminian A et al. Obesity Week 2019, Abstract A101.
LAS VEGAS –
The Individualized Diabetes Complications risk score “can provide personalized, evidence-based risk information for patients with type 2 diabetes and obesity about their future cardiovascular disease outcomes and mortality with and without metabolic surgery,” Ali Aminian, MD, said at a meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
Although the calculator needs validation in a prospective, randomized study to document its impact on practice, it is now available on two separate websites and as a downloadable app, said Dr. Aminian, a surgeon at the Cleveland Clinic.
The calculator inputs data for 26 distinct, “readily available” demographic and clinical entries, and based on that, estimates the patient’s 10-year risk for all-cause death, diabetic kidney disease, cerebrovascular disease, heart failure, and coronary artery disease if no surgery occurs or after some type of metabolic or bariatric surgery. The calculator does not currently have the ability to individualize predicted risks based on the specific type of metabolic surgery performed, but that is planned as a future refinement of the score.
“We validated the model in the nonsurgical patients, which showed it was very accurate. The next step is to run a randomized trial to see how useful the calculator is” for assisting in patients’ decision making, Dr. Aminian said.
The data for deriving the risk calculator, and for a preliminary validation of it, came from 13,722 patients with obesity (body mass index, 30 kg/m2 or greater) and type 2 diabetes, who were managed at the Cleveland Clinic during 1998-2017, drawn from more than 287,000 such patients in the clinic’s database. The study focused on 2,287 patients who underwent metabolic (bariatric) surgery and 11,435 patients from the same database who did not have surgery and matched by propensity scoring on a 5:1 basis with those who had surgery. The two cohorts this created matched well for age (about 54 years), sex (about two-thirds women), body mass index (about 44 kg/m2), and the prevalence of various comorbidities at baseline.
Dr. Aminian and associates then analyzed the incidence of all-cause mortality and various cardiovascular disease endpoints, as well as nephropathy during follow-up, through December 2018. Patients who had undergone metabolic surgery showed statistically significant reductions in the incidence of each of those events, compared with patients who did not have surgery (JAMA. 2019;322[13]:1271-82).
The investigators used these findings to create their model for calculating a patient’s risk score. For example, to calculate an estimate for the 10-year risk from all-cause mortality, the results showed that the most powerful risk factors were age; baseline body mass index, heart failure, and need for insulin; and smoking status. For the endpoint of nephropathy, the most important factors were estimated glomerular filtration rate at baseline and age. Identified risk factors could account for about 80% of the 10-year risk for all-cause death and for about 75% of the risk for developing nephropathy during 10 years, based on the area-under-the-curve values the model produced.
The risk score may help patients better understand the potential role that metabolic surgery can have in reducing their future event risk, thereby helping them better appreciate the benefit they stand to gain from undergoing surgery, Dr. Aminian said.
The calculator is available at a website maintained by the Cleveland Clinic, at a site of the American Society for Metabolic and Bariatric Surgery, and in app stores, he said.
The work was partially funded by Medtronic. Dr. Aminian has received grants from Medtronic.
SOURCE: Aminian A et al. Obesity Week 2019, Abstract A101.
REPORTING FROM OBESITY WEEK 2019