User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Pulse oximeters lead to less oxygen supplementation for people of color
The new research suggests that skin color–related differences in pulse oximeter readings are in fact impacting clinical decision-making, lead author Eric R. Gottlieb, MD, of Brigham and Women’s Hospital and Massachusetts Institute of Technology, both in Boston, and colleagues wrote. This suggests that technology needs to updated to improve health equity, they continued, in their paper published in JAMA Internal Medicine.
“It has been known for decades that these readings are affected by various surface pigmentations, including nail polish and skin melanin, which may affect light absorption and scattering,” the investigators wrote. “This increases the risk of hidden hypoxemia [among patients with darker skin], in which patients have falsely elevated SpO2 readings, usually defined as 92% or greater, with a blood hemoglobin oxygen saturation less than 88%.”
Although published reports on this phenomenon date back to the 1980s, clinical significance has been largely discounted, they said, citing a 2008 paper on the topic, which stated that “oximetry need not have exact accuracy” to determine if a patient needs oxygen supplementation.
‘We’re not providing equal care’
Questioning the validity of this statement, Dr. Gottlieb and colleagues conducted a retrospective cohort study involving 3,069 patients admitted to intensive care at the Beth Israel Deaconess Medical Center in Boston between 2008 and 2019, thereby excluding patients treated during the COVID-19 pandemic. The population consisted of four races/ethnicities: White (87%), Black (7%), Hispanic (4%), and Asian (3%).
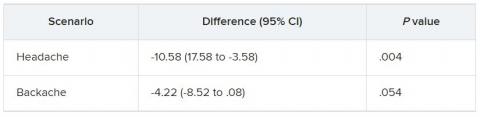
Aligning with previous studies, multivariable linear regression analyses showed that Asian, Black, and Hispanic patients had significantly higher SpO2 readings than White patients in relation to hemoglobin oxygen saturation values, suggesting falsely elevated readings.
Further modeling showed that these same patient groups also received lower oxygen delivery rates, which were not explained directly by race/ethnicity, but instead were mediated by the discrepancy between SpO2 and hemoglobin oxygen saturation values. In other words, physicians were responding consistently to pulse oximetry readings, rather than exhibiting a direct racial/ethnic bias in their clinical decision-making.
“We’re not providing equal care,” Dr. Gottlieb said in an interview. “It’s not that the patients are sicker, or have other socioeconomic explanations for why this happens to them. It’s us. It’s our technology. And that’s something that really has to be fixed.”
The investigators offered a cautionary view of corrective algorithms, as these “have exacerbated disparities and are subject to ethical concerns;” for example, with glomerular filtration rate estimations in Black patients.
Dr. Gottlieb also cautioned against action by individual physicians, who may now be inclined to change how they interpret pulse oximeter readings based on a patient’s race or ethnicity.
“I don’t think that we can expect physicians, every time they see a patient, to be second guessing whether the number basically reflects the truth,” he said.
Instead, Dr. Gottlieb suggested that the burden of change rests upon the shoulders of institutions, including hospitals and device manufacturers, both of which “really need to take the responsibility” for making sure that pulse oximeters are “equitable and have similar performance across races.”
While Dr. Gottlieb said that skin color likely plays the greatest role in measurement discrepancies, he encouraged stakeholders “to think broadly about this, and not just assume that it’s entirely skin color,” noting a small amount of evidence indicating that blood chemistry may also play a role. Still, he predicted that colorimetry – the direct measurement of skin color – will probably be incorporated into pulse oximeters of the future.
Black patients 3X more likely to have hidden hypoxia than White patients
Michael Sjoding, MD, of the University of Michigan, Ann Arbor, was one of the first to raise awareness of skin color–related issues with pulse oximeters during the throes of the COVID-19 pandemic. His study, which involved more than 10,000 patients, showed that Black patients were threefold more likely to have hidden hypoxia than White patients.
The present study shows that such discrepancies are indeed clinically significant, Dr. Sjoding said in an interview. And these data are needed, he added, to bring about change.
“What is being asked is potentially a big deal,” Dr. Sjoding said. “Pulse oximeters are everywhere, and it would be a big undertaking to redesign pulse oximeters and purchase new pulse oximeters. You need a compelling body of evidence to do that. I think it’s there now, clearly. So I’m hopeful that we’re going to finally move forward, towards having devices that we are confident work accurately in everyone.”
Why it has taken so long to gather this evidence, however, is a thornier topic, considering race-related discrepancies in pulse oximeter readings were first documented more than 3 decades ago.
“We sort of rediscovered something that had been known and had been described in the past,” Dr. Sjoding said. He explained how he and many of his colleagues had completed pulmonary fellowships, yet none of them knew of these potential issues with pulse oximeters until they began to observe differences in their own patients during the pandemic.
“I’ll give previous generations of researchers the benefit of the doubt,” Dr. Sjoding said, pointing out that techniques in data gathering and analysis have advanced considerably over the years. “The types of studies that were done before were very different than what we did.”
Yet Dr. Sjoding entertained the possibility that other factors may have been at play.
“I think definitely there’s a social commentary on prioritization of research,” he said.
The study was supported by grants from the National Institutes of Health. The investigators and Dr. Sjoding reported no conflicts of interest.
The new research suggests that skin color–related differences in pulse oximeter readings are in fact impacting clinical decision-making, lead author Eric R. Gottlieb, MD, of Brigham and Women’s Hospital and Massachusetts Institute of Technology, both in Boston, and colleagues wrote. This suggests that technology needs to updated to improve health equity, they continued, in their paper published in JAMA Internal Medicine.
“It has been known for decades that these readings are affected by various surface pigmentations, including nail polish and skin melanin, which may affect light absorption and scattering,” the investigators wrote. “This increases the risk of hidden hypoxemia [among patients with darker skin], in which patients have falsely elevated SpO2 readings, usually defined as 92% or greater, with a blood hemoglobin oxygen saturation less than 88%.”
Although published reports on this phenomenon date back to the 1980s, clinical significance has been largely discounted, they said, citing a 2008 paper on the topic, which stated that “oximetry need not have exact accuracy” to determine if a patient needs oxygen supplementation.
‘We’re not providing equal care’
Questioning the validity of this statement, Dr. Gottlieb and colleagues conducted a retrospective cohort study involving 3,069 patients admitted to intensive care at the Beth Israel Deaconess Medical Center in Boston between 2008 and 2019, thereby excluding patients treated during the COVID-19 pandemic. The population consisted of four races/ethnicities: White (87%), Black (7%), Hispanic (4%), and Asian (3%).
Aligning with previous studies, multivariable linear regression analyses showed that Asian, Black, and Hispanic patients had significantly higher SpO2 readings than White patients in relation to hemoglobin oxygen saturation values, suggesting falsely elevated readings.
Further modeling showed that these same patient groups also received lower oxygen delivery rates, which were not explained directly by race/ethnicity, but instead were mediated by the discrepancy between SpO2 and hemoglobin oxygen saturation values. In other words, physicians were responding consistently to pulse oximetry readings, rather than exhibiting a direct racial/ethnic bias in their clinical decision-making.
“We’re not providing equal care,” Dr. Gottlieb said in an interview. “It’s not that the patients are sicker, or have other socioeconomic explanations for why this happens to them. It’s us. It’s our technology. And that’s something that really has to be fixed.”
The investigators offered a cautionary view of corrective algorithms, as these “have exacerbated disparities and are subject to ethical concerns;” for example, with glomerular filtration rate estimations in Black patients.
Dr. Gottlieb also cautioned against action by individual physicians, who may now be inclined to change how they interpret pulse oximeter readings based on a patient’s race or ethnicity.
“I don’t think that we can expect physicians, every time they see a patient, to be second guessing whether the number basically reflects the truth,” he said.
Instead, Dr. Gottlieb suggested that the burden of change rests upon the shoulders of institutions, including hospitals and device manufacturers, both of which “really need to take the responsibility” for making sure that pulse oximeters are “equitable and have similar performance across races.”
While Dr. Gottlieb said that skin color likely plays the greatest role in measurement discrepancies, he encouraged stakeholders “to think broadly about this, and not just assume that it’s entirely skin color,” noting a small amount of evidence indicating that blood chemistry may also play a role. Still, he predicted that colorimetry – the direct measurement of skin color – will probably be incorporated into pulse oximeters of the future.
Black patients 3X more likely to have hidden hypoxia than White patients
Michael Sjoding, MD, of the University of Michigan, Ann Arbor, was one of the first to raise awareness of skin color–related issues with pulse oximeters during the throes of the COVID-19 pandemic. His study, which involved more than 10,000 patients, showed that Black patients were threefold more likely to have hidden hypoxia than White patients.
The present study shows that such discrepancies are indeed clinically significant, Dr. Sjoding said in an interview. And these data are needed, he added, to bring about change.
“What is being asked is potentially a big deal,” Dr. Sjoding said. “Pulse oximeters are everywhere, and it would be a big undertaking to redesign pulse oximeters and purchase new pulse oximeters. You need a compelling body of evidence to do that. I think it’s there now, clearly. So I’m hopeful that we’re going to finally move forward, towards having devices that we are confident work accurately in everyone.”
Why it has taken so long to gather this evidence, however, is a thornier topic, considering race-related discrepancies in pulse oximeter readings were first documented more than 3 decades ago.
“We sort of rediscovered something that had been known and had been described in the past,” Dr. Sjoding said. He explained how he and many of his colleagues had completed pulmonary fellowships, yet none of them knew of these potential issues with pulse oximeters until they began to observe differences in their own patients during the pandemic.
“I’ll give previous generations of researchers the benefit of the doubt,” Dr. Sjoding said, pointing out that techniques in data gathering and analysis have advanced considerably over the years. “The types of studies that were done before were very different than what we did.”
Yet Dr. Sjoding entertained the possibility that other factors may have been at play.
“I think definitely there’s a social commentary on prioritization of research,” he said.
The study was supported by grants from the National Institutes of Health. The investigators and Dr. Sjoding reported no conflicts of interest.
The new research suggests that skin color–related differences in pulse oximeter readings are in fact impacting clinical decision-making, lead author Eric R. Gottlieb, MD, of Brigham and Women’s Hospital and Massachusetts Institute of Technology, both in Boston, and colleagues wrote. This suggests that technology needs to updated to improve health equity, they continued, in their paper published in JAMA Internal Medicine.
“It has been known for decades that these readings are affected by various surface pigmentations, including nail polish and skin melanin, which may affect light absorption and scattering,” the investigators wrote. “This increases the risk of hidden hypoxemia [among patients with darker skin], in which patients have falsely elevated SpO2 readings, usually defined as 92% or greater, with a blood hemoglobin oxygen saturation less than 88%.”
Although published reports on this phenomenon date back to the 1980s, clinical significance has been largely discounted, they said, citing a 2008 paper on the topic, which stated that “oximetry need not have exact accuracy” to determine if a patient needs oxygen supplementation.
‘We’re not providing equal care’
Questioning the validity of this statement, Dr. Gottlieb and colleagues conducted a retrospective cohort study involving 3,069 patients admitted to intensive care at the Beth Israel Deaconess Medical Center in Boston between 2008 and 2019, thereby excluding patients treated during the COVID-19 pandemic. The population consisted of four races/ethnicities: White (87%), Black (7%), Hispanic (4%), and Asian (3%).
Aligning with previous studies, multivariable linear regression analyses showed that Asian, Black, and Hispanic patients had significantly higher SpO2 readings than White patients in relation to hemoglobin oxygen saturation values, suggesting falsely elevated readings.
Further modeling showed that these same patient groups also received lower oxygen delivery rates, which were not explained directly by race/ethnicity, but instead were mediated by the discrepancy between SpO2 and hemoglobin oxygen saturation values. In other words, physicians were responding consistently to pulse oximetry readings, rather than exhibiting a direct racial/ethnic bias in their clinical decision-making.
“We’re not providing equal care,” Dr. Gottlieb said in an interview. “It’s not that the patients are sicker, or have other socioeconomic explanations for why this happens to them. It’s us. It’s our technology. And that’s something that really has to be fixed.”
The investigators offered a cautionary view of corrective algorithms, as these “have exacerbated disparities and are subject to ethical concerns;” for example, with glomerular filtration rate estimations in Black patients.
Dr. Gottlieb also cautioned against action by individual physicians, who may now be inclined to change how they interpret pulse oximeter readings based on a patient’s race or ethnicity.
“I don’t think that we can expect physicians, every time they see a patient, to be second guessing whether the number basically reflects the truth,” he said.
Instead, Dr. Gottlieb suggested that the burden of change rests upon the shoulders of institutions, including hospitals and device manufacturers, both of which “really need to take the responsibility” for making sure that pulse oximeters are “equitable and have similar performance across races.”
While Dr. Gottlieb said that skin color likely plays the greatest role in measurement discrepancies, he encouraged stakeholders “to think broadly about this, and not just assume that it’s entirely skin color,” noting a small amount of evidence indicating that blood chemistry may also play a role. Still, he predicted that colorimetry – the direct measurement of skin color – will probably be incorporated into pulse oximeters of the future.
Black patients 3X more likely to have hidden hypoxia than White patients
Michael Sjoding, MD, of the University of Michigan, Ann Arbor, was one of the first to raise awareness of skin color–related issues with pulse oximeters during the throes of the COVID-19 pandemic. His study, which involved more than 10,000 patients, showed that Black patients were threefold more likely to have hidden hypoxia than White patients.
The present study shows that such discrepancies are indeed clinically significant, Dr. Sjoding said in an interview. And these data are needed, he added, to bring about change.
“What is being asked is potentially a big deal,” Dr. Sjoding said. “Pulse oximeters are everywhere, and it would be a big undertaking to redesign pulse oximeters and purchase new pulse oximeters. You need a compelling body of evidence to do that. I think it’s there now, clearly. So I’m hopeful that we’re going to finally move forward, towards having devices that we are confident work accurately in everyone.”
Why it has taken so long to gather this evidence, however, is a thornier topic, considering race-related discrepancies in pulse oximeter readings were first documented more than 3 decades ago.
“We sort of rediscovered something that had been known and had been described in the past,” Dr. Sjoding said. He explained how he and many of his colleagues had completed pulmonary fellowships, yet none of them knew of these potential issues with pulse oximeters until they began to observe differences in their own patients during the pandemic.
“I’ll give previous generations of researchers the benefit of the doubt,” Dr. Sjoding said, pointing out that techniques in data gathering and analysis have advanced considerably over the years. “The types of studies that were done before were very different than what we did.”
Yet Dr. Sjoding entertained the possibility that other factors may have been at play.
“I think definitely there’s a social commentary on prioritization of research,” he said.
The study was supported by grants from the National Institutes of Health. The investigators and Dr. Sjoding reported no conflicts of interest.
FROM JAMA INTERNAL MEDICINE
High deductible insurance linked to delayed advanced cancer diagnosis
In oncology, delayed care may result in a failed opportunity to achieve remission. Delays in diagnosis can result in patients having to undergo more extensive surgery, radiation exposure, or more intensive drug therapy than if their disease had been detected at an early stage.
Now, researchers at Harvard Medical School, Boston, report that
Using national insurance claims data, the authors conducted an observational study to examine what happened when some workers with employer-based insurance were switched from low-deductible to high-deductible plans, compared with a control group of workers who remained on low-deductible plans.
After the switch, workers shunted into high-deductible plans had a longer time to first diagnosis of a metastatic cancer, indicating delayed detection of advanced disease, compared with controls. The difference translated into a delay in diagnosis of metastatic disease of nearly 5 months, reported Nico Trad, BA, a fourth-year medical student at Dana-Farber Cancer Institute, Boston.
“The takeaway here is that these plans were associated with delayed detection of metastatic cancer. We did not assess the mechanism, but it’s a reasonable assumption to make that increased cost-sharing is having some adverse impacts on people’s willingness to seek care. And although we didn’t study potential impacts, we might anticipate that a delayed diagnosis might also lead to delayed engagement with palliative care,” he said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“A delay in initiation of symptom-relieving therapies and a delayed presentation might also lead to greater dissemination of disease throughout the body, which also has the potential to limit therapeutic options,” he added.
‘Deductible relief day’
Mr. Trad said that in 2022 more than half of employees are covered by high-deductible health plans, compared with only about 10% in 2006.
This major shift in cost burden coincided with President Joseph Biden’s announcement in early 2022 of the “Cancer Moonshot,” program with the goal of reducing cancer mortality by 50% over the next 25 years.
“Part of that is cancer prevention and control, which involves timely detection of cancer so that we can treat it early and have better outcomes,” he said.
High-deductible health plans ostensibly provide motivation for patients to shop for lower-priced care and avoid unnecessary or low-quality care, but making patients shell out more upfront before their insurance kicks in, while it reduced health care utilization, can also reduce the quality of care, he said.
In 2022, “Deductible Relief Day,” the day in which the average patient has satisfied the deductible and insurance starts to pick up more of the tab, occurred in mid-May, compared with late February in 2006.
Insurance claims data
Mr. Trad and colleagues used health insurance claims data from a nationally representative cohort of privately insured patients in a national commercial and Medicare Advantage database. They excluded patients 65 and older who were eligible for Medicare because it does not have high-deductible options.
The study cohort included 345,401 adults from the ages of 18 to 64 whose employers mandated a switch from a low-deductible plan which was defined as $500 or less, to a high-deductible plan defined as $1,000 or more. Controls were 1,654,775 contemporaneous adults whose employers offered only low-deductible plans. Both groups had a 1-year baseline period when all members were enrolled in low deductible plans.
To minimize the possibility of confounding, the investigators matched the participants by age, gender, race/ethnicity, morbidity according to Adjusted Clinical Group score, poverty level, geographic region, employer size, baseline primary cancer, baseline medical and pharmacy costs, and follow-up duration.
During the baseline period, the hazard ratio for time to a first observed metastatic cancer diagnosis in the main cohort, compared with controls, was 0.96 with a nonsignificant P value, indicating no difference in the time to diagnosis between the groups.
During a maximum 13.5 years of follow-up, however, the participants who had been switched after a year to a high-deductible plan had a significantly longer time to first metastatic diagnosis (HR, 0.88; P = .01), indicating delayed diagnosis relative to controls. This difference translated to a delay of 4.6 months associated with the higher out-of-pocket costs plans.
According to a systematic review and meta-analysis published online in 2020, a 1-month delay in treatment for many types of cancer can translate into a 6% to 13% higher risk for death, a risk that continues to increase with further delays.
The investigators acknowledged that the study was limited by the use of retrospective claims-based data, which not contain information on how the patients fared after diagnosis.
“I would say in terms of policy relevance that this really points to the need for new and innovative insurance models that, No. 1, reduce the cost-sharing burden for patients so that they’re not deterred from seeking care, and No. 2, that align rather than contradict the goal of improving population-level survival from cancer,” Mr. Trad said.
Further evidence of a flawed system
The study adds to an already strong body of evidence showing that high-deductible plans can have a negative impact on health, said Sara R. Collins, vice president for health care coverage and access at the Commonwealth Fund, a New York–based private foundation dedicated to improving health care.
“This is really the latest evidence on top of years of research that shows that high-deductible health plans lead people to make decisions that are not in the best interest of their health,” said Ms. Collins, who is not affiliated with the study presented at ASCO.
“We have a health care cost problem in the United States that far exceeds that of other high-income countries. Insurers try to solve it by shifting the costs to consumers and using other measures to restrict people’s use of health care, and often needed health care like this. The result is less access to needed care, and long-term adverse health consequences and their associated costs to patients and the health system generally,” she said.
The real driver of health care costs is not utilization, but the prices that insurers and providers negotiate in their service contracts, she explained.
“Prices are the central problem, insurers have control over those prices in their negotiations with providers. So unless we can gain control of that driver, patients are going to continue to suffer unnecessarily from both the short- and long-term effects of insurers who use tools to reduce their access to care,” she said.
In oncology, delayed care may result in a failed opportunity to achieve remission. Delays in diagnosis can result in patients having to undergo more extensive surgery, radiation exposure, or more intensive drug therapy than if their disease had been detected at an early stage.
Now, researchers at Harvard Medical School, Boston, report that
Using national insurance claims data, the authors conducted an observational study to examine what happened when some workers with employer-based insurance were switched from low-deductible to high-deductible plans, compared with a control group of workers who remained on low-deductible plans.
After the switch, workers shunted into high-deductible plans had a longer time to first diagnosis of a metastatic cancer, indicating delayed detection of advanced disease, compared with controls. The difference translated into a delay in diagnosis of metastatic disease of nearly 5 months, reported Nico Trad, BA, a fourth-year medical student at Dana-Farber Cancer Institute, Boston.
“The takeaway here is that these plans were associated with delayed detection of metastatic cancer. We did not assess the mechanism, but it’s a reasonable assumption to make that increased cost-sharing is having some adverse impacts on people’s willingness to seek care. And although we didn’t study potential impacts, we might anticipate that a delayed diagnosis might also lead to delayed engagement with palliative care,” he said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“A delay in initiation of symptom-relieving therapies and a delayed presentation might also lead to greater dissemination of disease throughout the body, which also has the potential to limit therapeutic options,” he added.
‘Deductible relief day’
Mr. Trad said that in 2022 more than half of employees are covered by high-deductible health plans, compared with only about 10% in 2006.
This major shift in cost burden coincided with President Joseph Biden’s announcement in early 2022 of the “Cancer Moonshot,” program with the goal of reducing cancer mortality by 50% over the next 25 years.
“Part of that is cancer prevention and control, which involves timely detection of cancer so that we can treat it early and have better outcomes,” he said.
High-deductible health plans ostensibly provide motivation for patients to shop for lower-priced care and avoid unnecessary or low-quality care, but making patients shell out more upfront before their insurance kicks in, while it reduced health care utilization, can also reduce the quality of care, he said.
In 2022, “Deductible Relief Day,” the day in which the average patient has satisfied the deductible and insurance starts to pick up more of the tab, occurred in mid-May, compared with late February in 2006.
Insurance claims data
Mr. Trad and colleagues used health insurance claims data from a nationally representative cohort of privately insured patients in a national commercial and Medicare Advantage database. They excluded patients 65 and older who were eligible for Medicare because it does not have high-deductible options.
The study cohort included 345,401 adults from the ages of 18 to 64 whose employers mandated a switch from a low-deductible plan which was defined as $500 or less, to a high-deductible plan defined as $1,000 or more. Controls were 1,654,775 contemporaneous adults whose employers offered only low-deductible plans. Both groups had a 1-year baseline period when all members were enrolled in low deductible plans.
To minimize the possibility of confounding, the investigators matched the participants by age, gender, race/ethnicity, morbidity according to Adjusted Clinical Group score, poverty level, geographic region, employer size, baseline primary cancer, baseline medical and pharmacy costs, and follow-up duration.
During the baseline period, the hazard ratio for time to a first observed metastatic cancer diagnosis in the main cohort, compared with controls, was 0.96 with a nonsignificant P value, indicating no difference in the time to diagnosis between the groups.
During a maximum 13.5 years of follow-up, however, the participants who had been switched after a year to a high-deductible plan had a significantly longer time to first metastatic diagnosis (HR, 0.88; P = .01), indicating delayed diagnosis relative to controls. This difference translated to a delay of 4.6 months associated with the higher out-of-pocket costs plans.
According to a systematic review and meta-analysis published online in 2020, a 1-month delay in treatment for many types of cancer can translate into a 6% to 13% higher risk for death, a risk that continues to increase with further delays.
The investigators acknowledged that the study was limited by the use of retrospective claims-based data, which not contain information on how the patients fared after diagnosis.
“I would say in terms of policy relevance that this really points to the need for new and innovative insurance models that, No. 1, reduce the cost-sharing burden for patients so that they’re not deterred from seeking care, and No. 2, that align rather than contradict the goal of improving population-level survival from cancer,” Mr. Trad said.
Further evidence of a flawed system
The study adds to an already strong body of evidence showing that high-deductible plans can have a negative impact on health, said Sara R. Collins, vice president for health care coverage and access at the Commonwealth Fund, a New York–based private foundation dedicated to improving health care.
“This is really the latest evidence on top of years of research that shows that high-deductible health plans lead people to make decisions that are not in the best interest of their health,” said Ms. Collins, who is not affiliated with the study presented at ASCO.
“We have a health care cost problem in the United States that far exceeds that of other high-income countries. Insurers try to solve it by shifting the costs to consumers and using other measures to restrict people’s use of health care, and often needed health care like this. The result is less access to needed care, and long-term adverse health consequences and their associated costs to patients and the health system generally,” she said.
The real driver of health care costs is not utilization, but the prices that insurers and providers negotiate in their service contracts, she explained.
“Prices are the central problem, insurers have control over those prices in their negotiations with providers. So unless we can gain control of that driver, patients are going to continue to suffer unnecessarily from both the short- and long-term effects of insurers who use tools to reduce their access to care,” she said.
In oncology, delayed care may result in a failed opportunity to achieve remission. Delays in diagnosis can result in patients having to undergo more extensive surgery, radiation exposure, or more intensive drug therapy than if their disease had been detected at an early stage.
Now, researchers at Harvard Medical School, Boston, report that
Using national insurance claims data, the authors conducted an observational study to examine what happened when some workers with employer-based insurance were switched from low-deductible to high-deductible plans, compared with a control group of workers who remained on low-deductible plans.
After the switch, workers shunted into high-deductible plans had a longer time to first diagnosis of a metastatic cancer, indicating delayed detection of advanced disease, compared with controls. The difference translated into a delay in diagnosis of metastatic disease of nearly 5 months, reported Nico Trad, BA, a fourth-year medical student at Dana-Farber Cancer Institute, Boston.
“The takeaway here is that these plans were associated with delayed detection of metastatic cancer. We did not assess the mechanism, but it’s a reasonable assumption to make that increased cost-sharing is having some adverse impacts on people’s willingness to seek care. And although we didn’t study potential impacts, we might anticipate that a delayed diagnosis might also lead to delayed engagement with palliative care,” he said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“A delay in initiation of symptom-relieving therapies and a delayed presentation might also lead to greater dissemination of disease throughout the body, which also has the potential to limit therapeutic options,” he added.
‘Deductible relief day’
Mr. Trad said that in 2022 more than half of employees are covered by high-deductible health plans, compared with only about 10% in 2006.
This major shift in cost burden coincided with President Joseph Biden’s announcement in early 2022 of the “Cancer Moonshot,” program with the goal of reducing cancer mortality by 50% over the next 25 years.
“Part of that is cancer prevention and control, which involves timely detection of cancer so that we can treat it early and have better outcomes,” he said.
High-deductible health plans ostensibly provide motivation for patients to shop for lower-priced care and avoid unnecessary or low-quality care, but making patients shell out more upfront before their insurance kicks in, while it reduced health care utilization, can also reduce the quality of care, he said.
In 2022, “Deductible Relief Day,” the day in which the average patient has satisfied the deductible and insurance starts to pick up more of the tab, occurred in mid-May, compared with late February in 2006.
Insurance claims data
Mr. Trad and colleagues used health insurance claims data from a nationally representative cohort of privately insured patients in a national commercial and Medicare Advantage database. They excluded patients 65 and older who were eligible for Medicare because it does not have high-deductible options.
The study cohort included 345,401 adults from the ages of 18 to 64 whose employers mandated a switch from a low-deductible plan which was defined as $500 or less, to a high-deductible plan defined as $1,000 or more. Controls were 1,654,775 contemporaneous adults whose employers offered only low-deductible plans. Both groups had a 1-year baseline period when all members were enrolled in low deductible plans.
To minimize the possibility of confounding, the investigators matched the participants by age, gender, race/ethnicity, morbidity according to Adjusted Clinical Group score, poverty level, geographic region, employer size, baseline primary cancer, baseline medical and pharmacy costs, and follow-up duration.
During the baseline period, the hazard ratio for time to a first observed metastatic cancer diagnosis in the main cohort, compared with controls, was 0.96 with a nonsignificant P value, indicating no difference in the time to diagnosis between the groups.
During a maximum 13.5 years of follow-up, however, the participants who had been switched after a year to a high-deductible plan had a significantly longer time to first metastatic diagnosis (HR, 0.88; P = .01), indicating delayed diagnosis relative to controls. This difference translated to a delay of 4.6 months associated with the higher out-of-pocket costs plans.
According to a systematic review and meta-analysis published online in 2020, a 1-month delay in treatment for many types of cancer can translate into a 6% to 13% higher risk for death, a risk that continues to increase with further delays.
The investigators acknowledged that the study was limited by the use of retrospective claims-based data, which not contain information on how the patients fared after diagnosis.
“I would say in terms of policy relevance that this really points to the need for new and innovative insurance models that, No. 1, reduce the cost-sharing burden for patients so that they’re not deterred from seeking care, and No. 2, that align rather than contradict the goal of improving population-level survival from cancer,” Mr. Trad said.
Further evidence of a flawed system
The study adds to an already strong body of evidence showing that high-deductible plans can have a negative impact on health, said Sara R. Collins, vice president for health care coverage and access at the Commonwealth Fund, a New York–based private foundation dedicated to improving health care.
“This is really the latest evidence on top of years of research that shows that high-deductible health plans lead people to make decisions that are not in the best interest of their health,” said Ms. Collins, who is not affiliated with the study presented at ASCO.
“We have a health care cost problem in the United States that far exceeds that of other high-income countries. Insurers try to solve it by shifting the costs to consumers and using other measures to restrict people’s use of health care, and often needed health care like this. The result is less access to needed care, and long-term adverse health consequences and their associated costs to patients and the health system generally,” she said.
The real driver of health care costs is not utilization, but the prices that insurers and providers negotiate in their service contracts, she explained.
“Prices are the central problem, insurers have control over those prices in their negotiations with providers. So unless we can gain control of that driver, patients are going to continue to suffer unnecessarily from both the short- and long-term effects of insurers who use tools to reduce their access to care,” she said.
FROM ASCO 2022
Children and COVID: Vaccination a harder sell in the summer
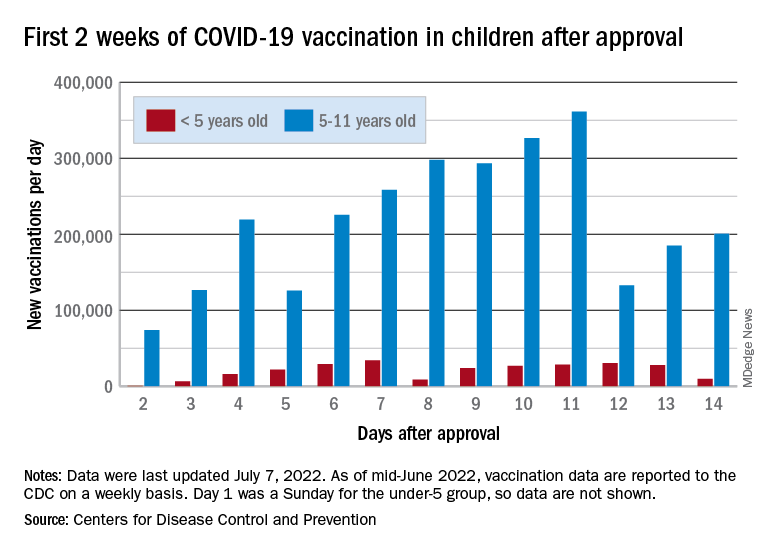
The COVID-19 vaccination effort in the youngest children has begun much more slowly than the most recent rollout for older children, according to the Centers for Disease Control and Prevention.
in early November of 2021, based on CDC data last updated on July 7.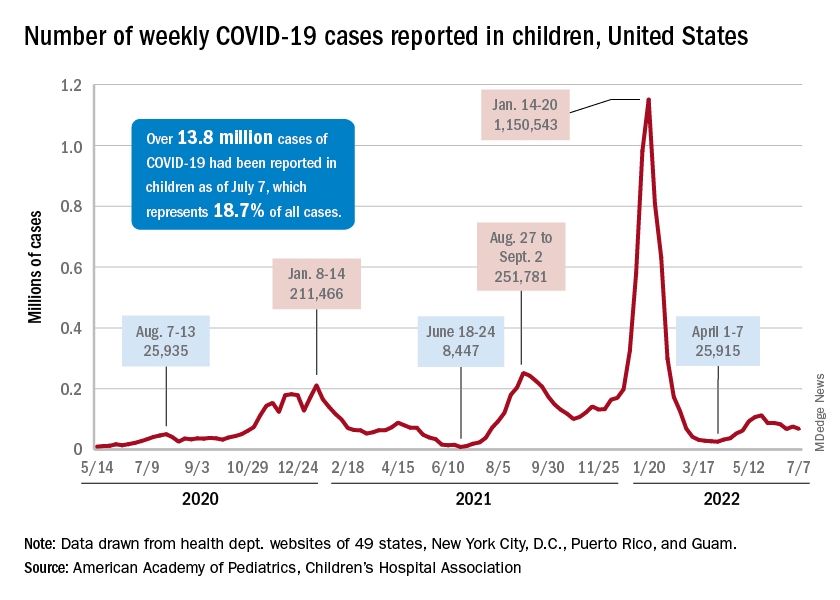
That approval, of course, came between the Delta and Omicron surges, when awareness was higher. The low initial uptake among those under age 5, however, was not unexpected by the Biden administration. “That number in and of itself is very much in line with our expectation, and we’re eager to continue working closely with partners to build on this start,” a senior administration official told ABC News.
With approval of the vaccine occurring after the school year was over, parents’ thoughts have been focused more on vacations and less on vaccinations. “Even before these vaccines officially became available, this was going to be a different rollout; it was going to take more time,” the official explained.
Incidence measures continue on different paths
New COVID-19 cases dropped during the latest reporting week (July 1-7), returning to the downward trend that began in late May and then stopped for 1 week (June 24-30), when cases were up by 12.4%, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Children also represent a smaller share of cases, probably because of underreporting. “There has been a notable decline in the portion of reported weekly COVID-19 cases that are children,” the two groups said in their weekly COVID report. Although “cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases.”
Other measures, however, have been rising slowly but steadily since the spring. New admissions of patients aged 0-17 years with confirmed COVID, which were down to 0.13 per 100,000 population in early April, had climbed to 0.39 per 100,000 by July 7, the CDC said on its COVID Data Tracker.
Emergency department visits continue to show the same upward trend, despite a small decline in early June. A COVID diagnosis was involved in just 0.5% of ED visits in children aged 0-11 years on March 26, but by July 6 the rate was 4.7%. Increases were not as high among older children: From 0.3% on March 26 to 2.5% on July 6 for those aged 12-15 and from 0.3% to 2.4% for 16- and 17-year-olds, according to the CDC.
The COVID-19 vaccination effort in the youngest children has begun much more slowly than the most recent rollout for older children, according to the Centers for Disease Control and Prevention.
in early November of 2021, based on CDC data last updated on July 7.
That approval, of course, came between the Delta and Omicron surges, when awareness was higher. The low initial uptake among those under age 5, however, was not unexpected by the Biden administration. “That number in and of itself is very much in line with our expectation, and we’re eager to continue working closely with partners to build on this start,” a senior administration official told ABC News.
With approval of the vaccine occurring after the school year was over, parents’ thoughts have been focused more on vacations and less on vaccinations. “Even before these vaccines officially became available, this was going to be a different rollout; it was going to take more time,” the official explained.
Incidence measures continue on different paths
New COVID-19 cases dropped during the latest reporting week (July 1-7), returning to the downward trend that began in late May and then stopped for 1 week (June 24-30), when cases were up by 12.4%, according to the American Academy of Pediatrics and the Children’s Hospital Association.

Children also represent a smaller share of cases, probably because of underreporting. “There has been a notable decline in the portion of reported weekly COVID-19 cases that are children,” the two groups said in their weekly COVID report. Although “cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases.”
Other measures, however, have been rising slowly but steadily since the spring. New admissions of patients aged 0-17 years with confirmed COVID, which were down to 0.13 per 100,000 population in early April, had climbed to 0.39 per 100,000 by July 7, the CDC said on its COVID Data Tracker.
Emergency department visits continue to show the same upward trend, despite a small decline in early June. A COVID diagnosis was involved in just 0.5% of ED visits in children aged 0-11 years on March 26, but by July 6 the rate was 4.7%. Increases were not as high among older children: From 0.3% on March 26 to 2.5% on July 6 for those aged 12-15 and from 0.3% to 2.4% for 16- and 17-year-olds, according to the CDC.
The COVID-19 vaccination effort in the youngest children has begun much more slowly than the most recent rollout for older children, according to the Centers for Disease Control and Prevention.
in early November of 2021, based on CDC data last updated on July 7.
That approval, of course, came between the Delta and Omicron surges, when awareness was higher. The low initial uptake among those under age 5, however, was not unexpected by the Biden administration. “That number in and of itself is very much in line with our expectation, and we’re eager to continue working closely with partners to build on this start,” a senior administration official told ABC News.
With approval of the vaccine occurring after the school year was over, parents’ thoughts have been focused more on vacations and less on vaccinations. “Even before these vaccines officially became available, this was going to be a different rollout; it was going to take more time,” the official explained.
Incidence measures continue on different paths
New COVID-19 cases dropped during the latest reporting week (July 1-7), returning to the downward trend that began in late May and then stopped for 1 week (June 24-30), when cases were up by 12.4%, according to the American Academy of Pediatrics and the Children’s Hospital Association.

Children also represent a smaller share of cases, probably because of underreporting. “There has been a notable decline in the portion of reported weekly COVID-19 cases that are children,” the two groups said in their weekly COVID report. Although “cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases.”
Other measures, however, have been rising slowly but steadily since the spring. New admissions of patients aged 0-17 years with confirmed COVID, which were down to 0.13 per 100,000 population in early April, had climbed to 0.39 per 100,000 by July 7, the CDC said on its COVID Data Tracker.
Emergency department visits continue to show the same upward trend, despite a small decline in early June. A COVID diagnosis was involved in just 0.5% of ED visits in children aged 0-11 years on March 26, but by July 6 the rate was 4.7%. Increases were not as high among older children: From 0.3% on March 26 to 2.5% on July 6 for those aged 12-15 and from 0.3% to 2.4% for 16- and 17-year-olds, according to the CDC.
Aggression toward health care providers common during pandemic
After an aggressive event or abuse occurred, 56% of providers considered changing their care tasks, and more than a third considered quitting their profession.
“Aggression of any sort against health care providers is not a new social phenomenon, and it has existed as far as medicine and health care is reported in literature. However, the phenomenon of aggression against health care providers during the pandemic grew worse,” senior study author Adrian Baranchuk, MD, a professor of medicine at Queen’s University, Kingston, Ont., told this news organization.
The study was published online in Current Problems in Cardiology
Survey snapshot
Dr. Baranchuk and colleagues, with the support of the Inter-American Society of Cardiology, developed a survey to characterize the frequency and types of abuse that frontline health professionals faced. They invited health care professionals from Latin America who had provided care since March 2020 to participate.
Between January and February 2022, 3,544 participants from 19 countries took the survey. Among them, 70.8% were physicians, 16% were nurses, and 13.2% were other health team members, such as administrative staff and technicians. About 58.5% were women, and 74.7% provided direct care to patients with COVID-19.
Overall, 54.8% of respondents reported acts of aggression. Of this group, 95.6% reported verbal abuse, 11.1% reported physical abuse, and 19.9% reported other types of abuse, including microaggressions.
About 13% of respondents reported experiencing some form of aggression daily, 26.4% experienced abuse weekly, and 38.8% reported violence a few times per month. Typically, the incidents involved patients’ relatives or both the patients and their relatives.
Nearly half of those who reported abuse experienced psychosomatic symptoms after the event, and 12% sought psychological care.
Administrative staff were 3.5 times more likely to experience abuse than other health care workers. Doctors and nurses were about twice as likely to experience abuse.
In addition, women, younger staff, and those who worked directly with COVID-19 patients were more likely to report abuse.
‘Shocking results’
Dr. Baranchuk, a native of Argentina, said people initially celebrated doctors and nurses for keeping communities safe. In several countries across Latin America, for instance, people lit candles, applauded at certain hours, and posted support on social media. As pandemic-related policies changed, however, health care providers faced unrest as people grew tired of wearing masks, maintaining social distance, and obeying restrictions at public spaces such as clubs and restaurants.
“This fatigue toward the social changes grew, but people didn’t have a specific target, and slowly and gradually, health care providers became the target of frustration and hate,” said Dr. Baranchuk. “In areas of the world where legislation is more flexible and less strict in charging individuals with poor or unacceptable behavior toward members of the health care team, aggression and microaggression became more frequent.”
“The results we obtained were more shocking than we expected,” Sebastián García-Zamora, MD, the lead study author and head of the coronary care unit at the Delta Clinic, Buenos Aires, said in an interview.
Dr. García-Zamora, also the coordinator of the International Society of Electrocardiology Young Community, noted the particularly high numbers of reports among young health care workers and women.
“Unfortunately, young women seem to be the most vulnerable staff to suffering violence, regardless of the work they perform in the health system,” he said. “Notably, less than one in four health team members that suffered workplace violence pursued legal action based on the events.”
The research team is now conducting additional analyses on the different types of aggression based on gender, region, and task performed by the health care team. They’re trying to understand who is most vulnerable to physical attacks, as well as the consequences.
“The most important thing to highlight is that this problem exists, it is more frequent than we think, and we can only solve it if we all get involved in it,” Dr. García-Zamora said.
‘Complete systematic failure’
Health care workers in certain communities faced more aggression as well. In a CMAJ Open study published in November 2021, Asian Canadian and Asian American health care workers experienced discrimination, racial microaggressions, threats of violence, and violent acts during the pandemic. Women and frontline workers with direct patient contact were more likely to face verbal and physical abuse.
“This highlights that we need to continue the fight against misogyny, racism, and health care worker discrimination,” lead study author Zhida Shang, a medical student at McGill University, Montreal, told this news organization.
“As we are managing to live with the COVID-19 pandemic, it is important to study our successes and shortcomings. I sincerely believe that during the pandemic, the treatment of various racialized communities, including Asian Americans and Asian Canadians, was a complete systematic failure,” he said. “It is crucial to continue to examine, reflect, and learn from these lessons so that there will be equitable outcomes during the next public health emergency.”
The study was conducted without funding support. Dr. Baranchuk, Dr. García-Zamora, and Ms. Shang report no relevant disclosures.
A version of this article first appeared on Medscape.com.
After an aggressive event or abuse occurred, 56% of providers considered changing their care tasks, and more than a third considered quitting their profession.
“Aggression of any sort against health care providers is not a new social phenomenon, and it has existed as far as medicine and health care is reported in literature. However, the phenomenon of aggression against health care providers during the pandemic grew worse,” senior study author Adrian Baranchuk, MD, a professor of medicine at Queen’s University, Kingston, Ont., told this news organization.
The study was published online in Current Problems in Cardiology
Survey snapshot
Dr. Baranchuk and colleagues, with the support of the Inter-American Society of Cardiology, developed a survey to characterize the frequency and types of abuse that frontline health professionals faced. They invited health care professionals from Latin America who had provided care since March 2020 to participate.
Between January and February 2022, 3,544 participants from 19 countries took the survey. Among them, 70.8% were physicians, 16% were nurses, and 13.2% were other health team members, such as administrative staff and technicians. About 58.5% were women, and 74.7% provided direct care to patients with COVID-19.
Overall, 54.8% of respondents reported acts of aggression. Of this group, 95.6% reported verbal abuse, 11.1% reported physical abuse, and 19.9% reported other types of abuse, including microaggressions.
About 13% of respondents reported experiencing some form of aggression daily, 26.4% experienced abuse weekly, and 38.8% reported violence a few times per month. Typically, the incidents involved patients’ relatives or both the patients and their relatives.
Nearly half of those who reported abuse experienced psychosomatic symptoms after the event, and 12% sought psychological care.
Administrative staff were 3.5 times more likely to experience abuse than other health care workers. Doctors and nurses were about twice as likely to experience abuse.
In addition, women, younger staff, and those who worked directly with COVID-19 patients were more likely to report abuse.
‘Shocking results’
Dr. Baranchuk, a native of Argentina, said people initially celebrated doctors and nurses for keeping communities safe. In several countries across Latin America, for instance, people lit candles, applauded at certain hours, and posted support on social media. As pandemic-related policies changed, however, health care providers faced unrest as people grew tired of wearing masks, maintaining social distance, and obeying restrictions at public spaces such as clubs and restaurants.
“This fatigue toward the social changes grew, but people didn’t have a specific target, and slowly and gradually, health care providers became the target of frustration and hate,” said Dr. Baranchuk. “In areas of the world where legislation is more flexible and less strict in charging individuals with poor or unacceptable behavior toward members of the health care team, aggression and microaggression became more frequent.”
“The results we obtained were more shocking than we expected,” Sebastián García-Zamora, MD, the lead study author and head of the coronary care unit at the Delta Clinic, Buenos Aires, said in an interview.
Dr. García-Zamora, also the coordinator of the International Society of Electrocardiology Young Community, noted the particularly high numbers of reports among young health care workers and women.
“Unfortunately, young women seem to be the most vulnerable staff to suffering violence, regardless of the work they perform in the health system,” he said. “Notably, less than one in four health team members that suffered workplace violence pursued legal action based on the events.”
The research team is now conducting additional analyses on the different types of aggression based on gender, region, and task performed by the health care team. They’re trying to understand who is most vulnerable to physical attacks, as well as the consequences.
“The most important thing to highlight is that this problem exists, it is more frequent than we think, and we can only solve it if we all get involved in it,” Dr. García-Zamora said.
‘Complete systematic failure’
Health care workers in certain communities faced more aggression as well. In a CMAJ Open study published in November 2021, Asian Canadian and Asian American health care workers experienced discrimination, racial microaggressions, threats of violence, and violent acts during the pandemic. Women and frontline workers with direct patient contact were more likely to face verbal and physical abuse.
“This highlights that we need to continue the fight against misogyny, racism, and health care worker discrimination,” lead study author Zhida Shang, a medical student at McGill University, Montreal, told this news organization.
“As we are managing to live with the COVID-19 pandemic, it is important to study our successes and shortcomings. I sincerely believe that during the pandemic, the treatment of various racialized communities, including Asian Americans and Asian Canadians, was a complete systematic failure,” he said. “It is crucial to continue to examine, reflect, and learn from these lessons so that there will be equitable outcomes during the next public health emergency.”
The study was conducted without funding support. Dr. Baranchuk, Dr. García-Zamora, and Ms. Shang report no relevant disclosures.
A version of this article first appeared on Medscape.com.
After an aggressive event or abuse occurred, 56% of providers considered changing their care tasks, and more than a third considered quitting their profession.
“Aggression of any sort against health care providers is not a new social phenomenon, and it has existed as far as medicine and health care is reported in literature. However, the phenomenon of aggression against health care providers during the pandemic grew worse,” senior study author Adrian Baranchuk, MD, a professor of medicine at Queen’s University, Kingston, Ont., told this news organization.
The study was published online in Current Problems in Cardiology
Survey snapshot
Dr. Baranchuk and colleagues, with the support of the Inter-American Society of Cardiology, developed a survey to characterize the frequency and types of abuse that frontline health professionals faced. They invited health care professionals from Latin America who had provided care since March 2020 to participate.
Between January and February 2022, 3,544 participants from 19 countries took the survey. Among them, 70.8% were physicians, 16% were nurses, and 13.2% were other health team members, such as administrative staff and technicians. About 58.5% were women, and 74.7% provided direct care to patients with COVID-19.
Overall, 54.8% of respondents reported acts of aggression. Of this group, 95.6% reported verbal abuse, 11.1% reported physical abuse, and 19.9% reported other types of abuse, including microaggressions.
About 13% of respondents reported experiencing some form of aggression daily, 26.4% experienced abuse weekly, and 38.8% reported violence a few times per month. Typically, the incidents involved patients’ relatives or both the patients and their relatives.
Nearly half of those who reported abuse experienced psychosomatic symptoms after the event, and 12% sought psychological care.
Administrative staff were 3.5 times more likely to experience abuse than other health care workers. Doctors and nurses were about twice as likely to experience abuse.
In addition, women, younger staff, and those who worked directly with COVID-19 patients were more likely to report abuse.
‘Shocking results’
Dr. Baranchuk, a native of Argentina, said people initially celebrated doctors and nurses for keeping communities safe. In several countries across Latin America, for instance, people lit candles, applauded at certain hours, and posted support on social media. As pandemic-related policies changed, however, health care providers faced unrest as people grew tired of wearing masks, maintaining social distance, and obeying restrictions at public spaces such as clubs and restaurants.
“This fatigue toward the social changes grew, but people didn’t have a specific target, and slowly and gradually, health care providers became the target of frustration and hate,” said Dr. Baranchuk. “In areas of the world where legislation is more flexible and less strict in charging individuals with poor or unacceptable behavior toward members of the health care team, aggression and microaggression became more frequent.”
“The results we obtained were more shocking than we expected,” Sebastián García-Zamora, MD, the lead study author and head of the coronary care unit at the Delta Clinic, Buenos Aires, said in an interview.
Dr. García-Zamora, also the coordinator of the International Society of Electrocardiology Young Community, noted the particularly high numbers of reports among young health care workers and women.
“Unfortunately, young women seem to be the most vulnerable staff to suffering violence, regardless of the work they perform in the health system,” he said. “Notably, less than one in four health team members that suffered workplace violence pursued legal action based on the events.”
The research team is now conducting additional analyses on the different types of aggression based on gender, region, and task performed by the health care team. They’re trying to understand who is most vulnerable to physical attacks, as well as the consequences.
“The most important thing to highlight is that this problem exists, it is more frequent than we think, and we can only solve it if we all get involved in it,” Dr. García-Zamora said.
‘Complete systematic failure’
Health care workers in certain communities faced more aggression as well. In a CMAJ Open study published in November 2021, Asian Canadian and Asian American health care workers experienced discrimination, racial microaggressions, threats of violence, and violent acts during the pandemic. Women and frontline workers with direct patient contact were more likely to face verbal and physical abuse.
“This highlights that we need to continue the fight against misogyny, racism, and health care worker discrimination,” lead study author Zhida Shang, a medical student at McGill University, Montreal, told this news organization.
“As we are managing to live with the COVID-19 pandemic, it is important to study our successes and shortcomings. I sincerely believe that during the pandemic, the treatment of various racialized communities, including Asian Americans and Asian Canadians, was a complete systematic failure,” he said. “It is crucial to continue to examine, reflect, and learn from these lessons so that there will be equitable outcomes during the next public health emergency.”
The study was conducted without funding support. Dr. Baranchuk, Dr. García-Zamora, and Ms. Shang report no relevant disclosures.
A version of this article first appeared on Medscape.com.
BA.4 and BA.5 subvariants are more evasive of antibodies, but not of cellular immunity
The picture around the BA.4 and BA.5 subvariants of Omicron has been really confusing in that the pair is driving up cases but global COVID-19 deaths remain at their lowest level since the beginning of the pandemic. Explaining the two components of the immune response – antibodies versus cellular immune responses – can help us understand where we are in the pandemic and future booster options.
These two subvariants of Omicron, as of July 5, make up more than half of the COVID-19 strains in the United States and are expected to keep increasing. One of two reasons can lead to a variant or subvariant becoming dominant strain: increased transmissibility or evasion of antibodies.
Although BA.4 and BA.5 could be more transmissible than other subvariants of Omicron (which is already very transmissible), this has not yet been established in experiments showing increased affinity for the human receptor or in animal models. What we do know is that BA.4 and BA.5 seem to evade neutralizing antibodies conferred by the vaccines or even prior BA.1 infection (an earlier subvariant of Omicron), which could be the reason we are seeing so many reinfections now. Of note, BA.1 infection conferred antibodies that protected against subsequent BA.2 infection, so we did not see the same spike in cases in the United States with BA.2 (after a large BA.1 spike over the winter) earlier this spring.
Okay, so isn’t evasion of antibodies a bad thing? Of course it is but, luckily, our immune system is “redundant” and doesn›t just rely on antibodies to protect us from infection. In fact, antibodies (such as IgA, which is the mucosal antibody most prevalent in the nose and mouth, and IgG, which is the most prevalent antibody in the bloodstream) are our first line of COVID-19 defense in the nasal mucosa. Therefore, mild upper respiratory infections will be common as BA.4/BA.5 evade our nasal antibodies. Luckily, the rate of severe disease is remaining low throughout the world, probably because of the high amounts of cellular immunity to the virus. B and T cells are our protectors from severe disease.
For instance, two-dose vaccines are still conferring high rates of protection from severe disease with the BA.4 and BA.5 variants, with 87% protection against hospitalization per South Africa data. This is probably attributable to the fact that T-cell immunity from the vaccines remains protective across variants “from Alpha to Omicron,” as described by a recent and elegant paper.
Data from Qatar show that natural infection (even occurring up to 14 months ago) remains very protective (97.3%) against severe disease with the current circulating subvariants, including BA.4 and BA.5. Again, this is probably attributable to T cells which specifically amplify in response to a piece of the virus and help recruit cells to attack the pathogen directly.
The original BA.1 subvariant of Omicron has 26-32 mutations along its spike protein that differ from the “ancestral strain,” and BA.4 and BA.5 variants have a few more. Our T-cell response, even across a mutated spike protein, is so robust that we have not seen Omicron yet able to evade the many T cells (which we produce from the vaccines or infection) that descend upon the mutated virus to fight severe disease. Antibody-producing memory B cells, generated by the vaccines (or prior infection), have been shown to actually adapt their immune response to the variant to which they are exposed.
Therefore, the story of the BA.4 and BA.5 subvariants seems to remain about antibodies vs. cellular immunity. Our immunity in the United States is growing and is from both vaccination and natural infection, with 78.3% of the population having had at least one dose of the vaccine and at least 60% of adults (and 75% of children 0-18) having been exposed to the virus by February 2022, per the Centers for Disease Control and Prevention (with exposure probably much higher now in July 2022 after subsequent Omicron subvariants waves).
So, what about Omicron-specific boosters? A booster shot will just raise antibodies temporarily, but their effectiveness wanes several months later. Moreover, a booster shot against the ancestral strain is not very effective in neutralizing BA.4 and BA.5 (with a prior BA.1 Omicron infection being more effective than a booster). Luckily, Pfizer has promised a BA.4/BA.5-specific mRNA vaccine by October, and Moderna has promised a bivalent vaccine containing BA.4/BA.5 mRNA sequences around the same time. A vaccine that specifically increases antibodies against the most prevalent circulating strain should be important as a booster for those who are predisposed to severe breakthrough infections (for example, those with immunocompromise or older individuals with multiple comorbidities). Moreover, BA.4/BA.5–specific booster vaccines may help prevent mild infections for many individuals. Finally, any booster (or exposure) should diversify and broaden T-cell responses to the virus, and a booster shot will also expand the potency of B cells, making them better able to respond to the newest subvariants as we continue to live with COVID-19.
Monica Gandhi, MD, MPH, is an infectious diseases doctor, professor of medicine, and associate chief in the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco.
A version of this article first appeared on Medscape.com.
The picture around the BA.4 and BA.5 subvariants of Omicron has been really confusing in that the pair is driving up cases but global COVID-19 deaths remain at their lowest level since the beginning of the pandemic. Explaining the two components of the immune response – antibodies versus cellular immune responses – can help us understand where we are in the pandemic and future booster options.
These two subvariants of Omicron, as of July 5, make up more than half of the COVID-19 strains in the United States and are expected to keep increasing. One of two reasons can lead to a variant or subvariant becoming dominant strain: increased transmissibility or evasion of antibodies.
Although BA.4 and BA.5 could be more transmissible than other subvariants of Omicron (which is already very transmissible), this has not yet been established in experiments showing increased affinity for the human receptor or in animal models. What we do know is that BA.4 and BA.5 seem to evade neutralizing antibodies conferred by the vaccines or even prior BA.1 infection (an earlier subvariant of Omicron), which could be the reason we are seeing so many reinfections now. Of note, BA.1 infection conferred antibodies that protected against subsequent BA.2 infection, so we did not see the same spike in cases in the United States with BA.2 (after a large BA.1 spike over the winter) earlier this spring.
Okay, so isn’t evasion of antibodies a bad thing? Of course it is but, luckily, our immune system is “redundant” and doesn›t just rely on antibodies to protect us from infection. In fact, antibodies (such as IgA, which is the mucosal antibody most prevalent in the nose and mouth, and IgG, which is the most prevalent antibody in the bloodstream) are our first line of COVID-19 defense in the nasal mucosa. Therefore, mild upper respiratory infections will be common as BA.4/BA.5 evade our nasal antibodies. Luckily, the rate of severe disease is remaining low throughout the world, probably because of the high amounts of cellular immunity to the virus. B and T cells are our protectors from severe disease.
For instance, two-dose vaccines are still conferring high rates of protection from severe disease with the BA.4 and BA.5 variants, with 87% protection against hospitalization per South Africa data. This is probably attributable to the fact that T-cell immunity from the vaccines remains protective across variants “from Alpha to Omicron,” as described by a recent and elegant paper.
Data from Qatar show that natural infection (even occurring up to 14 months ago) remains very protective (97.3%) against severe disease with the current circulating subvariants, including BA.4 and BA.5. Again, this is probably attributable to T cells which specifically amplify in response to a piece of the virus and help recruit cells to attack the pathogen directly.
The original BA.1 subvariant of Omicron has 26-32 mutations along its spike protein that differ from the “ancestral strain,” and BA.4 and BA.5 variants have a few more. Our T-cell response, even across a mutated spike protein, is so robust that we have not seen Omicron yet able to evade the many T cells (which we produce from the vaccines or infection) that descend upon the mutated virus to fight severe disease. Antibody-producing memory B cells, generated by the vaccines (or prior infection), have been shown to actually adapt their immune response to the variant to which they are exposed.
Therefore, the story of the BA.4 and BA.5 subvariants seems to remain about antibodies vs. cellular immunity. Our immunity in the United States is growing and is from both vaccination and natural infection, with 78.3% of the population having had at least one dose of the vaccine and at least 60% of adults (and 75% of children 0-18) having been exposed to the virus by February 2022, per the Centers for Disease Control and Prevention (with exposure probably much higher now in July 2022 after subsequent Omicron subvariants waves).
So, what about Omicron-specific boosters? A booster shot will just raise antibodies temporarily, but their effectiveness wanes several months later. Moreover, a booster shot against the ancestral strain is not very effective in neutralizing BA.4 and BA.5 (with a prior BA.1 Omicron infection being more effective than a booster). Luckily, Pfizer has promised a BA.4/BA.5-specific mRNA vaccine by October, and Moderna has promised a bivalent vaccine containing BA.4/BA.5 mRNA sequences around the same time. A vaccine that specifically increases antibodies against the most prevalent circulating strain should be important as a booster for those who are predisposed to severe breakthrough infections (for example, those with immunocompromise or older individuals with multiple comorbidities). Moreover, BA.4/BA.5–specific booster vaccines may help prevent mild infections for many individuals. Finally, any booster (or exposure) should diversify and broaden T-cell responses to the virus, and a booster shot will also expand the potency of B cells, making them better able to respond to the newest subvariants as we continue to live with COVID-19.
Monica Gandhi, MD, MPH, is an infectious diseases doctor, professor of medicine, and associate chief in the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco.
A version of this article first appeared on Medscape.com.
The picture around the BA.4 and BA.5 subvariants of Omicron has been really confusing in that the pair is driving up cases but global COVID-19 deaths remain at their lowest level since the beginning of the pandemic. Explaining the two components of the immune response – antibodies versus cellular immune responses – can help us understand where we are in the pandemic and future booster options.
These two subvariants of Omicron, as of July 5, make up more than half of the COVID-19 strains in the United States and are expected to keep increasing. One of two reasons can lead to a variant or subvariant becoming dominant strain: increased transmissibility or evasion of antibodies.
Although BA.4 and BA.5 could be more transmissible than other subvariants of Omicron (which is already very transmissible), this has not yet been established in experiments showing increased affinity for the human receptor or in animal models. What we do know is that BA.4 and BA.5 seem to evade neutralizing antibodies conferred by the vaccines or even prior BA.1 infection (an earlier subvariant of Omicron), which could be the reason we are seeing so many reinfections now. Of note, BA.1 infection conferred antibodies that protected against subsequent BA.2 infection, so we did not see the same spike in cases in the United States with BA.2 (after a large BA.1 spike over the winter) earlier this spring.
Okay, so isn’t evasion of antibodies a bad thing? Of course it is but, luckily, our immune system is “redundant” and doesn›t just rely on antibodies to protect us from infection. In fact, antibodies (such as IgA, which is the mucosal antibody most prevalent in the nose and mouth, and IgG, which is the most prevalent antibody in the bloodstream) are our first line of COVID-19 defense in the nasal mucosa. Therefore, mild upper respiratory infections will be common as BA.4/BA.5 evade our nasal antibodies. Luckily, the rate of severe disease is remaining low throughout the world, probably because of the high amounts of cellular immunity to the virus. B and T cells are our protectors from severe disease.
For instance, two-dose vaccines are still conferring high rates of protection from severe disease with the BA.4 and BA.5 variants, with 87% protection against hospitalization per South Africa data. This is probably attributable to the fact that T-cell immunity from the vaccines remains protective across variants “from Alpha to Omicron,” as described by a recent and elegant paper.
Data from Qatar show that natural infection (even occurring up to 14 months ago) remains very protective (97.3%) against severe disease with the current circulating subvariants, including BA.4 and BA.5. Again, this is probably attributable to T cells which specifically amplify in response to a piece of the virus and help recruit cells to attack the pathogen directly.
The original BA.1 subvariant of Omicron has 26-32 mutations along its spike protein that differ from the “ancestral strain,” and BA.4 and BA.5 variants have a few more. Our T-cell response, even across a mutated spike protein, is so robust that we have not seen Omicron yet able to evade the many T cells (which we produce from the vaccines or infection) that descend upon the mutated virus to fight severe disease. Antibody-producing memory B cells, generated by the vaccines (or prior infection), have been shown to actually adapt their immune response to the variant to which they are exposed.
Therefore, the story of the BA.4 and BA.5 subvariants seems to remain about antibodies vs. cellular immunity. Our immunity in the United States is growing and is from both vaccination and natural infection, with 78.3% of the population having had at least one dose of the vaccine and at least 60% of adults (and 75% of children 0-18) having been exposed to the virus by February 2022, per the Centers for Disease Control and Prevention (with exposure probably much higher now in July 2022 after subsequent Omicron subvariants waves).
So, what about Omicron-specific boosters? A booster shot will just raise antibodies temporarily, but their effectiveness wanes several months later. Moreover, a booster shot against the ancestral strain is not very effective in neutralizing BA.4 and BA.5 (with a prior BA.1 Omicron infection being more effective than a booster). Luckily, Pfizer has promised a BA.4/BA.5-specific mRNA vaccine by October, and Moderna has promised a bivalent vaccine containing BA.4/BA.5 mRNA sequences around the same time. A vaccine that specifically increases antibodies against the most prevalent circulating strain should be important as a booster for those who are predisposed to severe breakthrough infections (for example, those with immunocompromise or older individuals with multiple comorbidities). Moreover, BA.4/BA.5–specific booster vaccines may help prevent mild infections for many individuals. Finally, any booster (or exposure) should diversify and broaden T-cell responses to the virus, and a booster shot will also expand the potency of B cells, making them better able to respond to the newest subvariants as we continue to live with COVID-19.
Monica Gandhi, MD, MPH, is an infectious diseases doctor, professor of medicine, and associate chief in the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco.
A version of this article first appeared on Medscape.com.
Obesity links to faster fading of COVID vaccine protection
Researchers published the study covered in this summary on medRxiv.org as a preprint that has not yet been peer reviewed.
Key takeaways
- The study results suggest that
- The findings documented evidence of reduced neutralizing antibody capacity 6 months after primary vaccination in people with severe obesity.
- This was a large study involving about more than 3.5 million people who had received at least two doses of COVID-19 vaccine, including more than 650,000 with obesity.
Why this matters
- Obesity is associated with comorbidities that independently increase the risk for severe COVID-19, including type 2 diabetes, chronic kidney disease, and heart failure.
- The authors concluded that additional or more frequent booster doses are likely to be required to maintain protection among people with obesity against COVID-19.
Study design
- Prospective longitudinal study of the incidence and severity of COVID-19 infections and immune responses in a cohort of more than 3.5 million adults from a Scottish healthcare database who received two or three doses of COVID-19 vaccine. The data came from the study, centered at the University of Edinburgh.
- About 16% had obesity with a body mass index of 30-39.9 kg/m2, and an additional 3% had severe obesity with a BMI of 40 or greater.
- Although not specified in this preprint, another said that the vaccines administered in Scotland have been the Pfizer-BioNTech and Oxford-AstraZeneca formulations.
Key results
- Between Sept. 14, 2020, and March 19, 2022, 10,983 people (0.3% of the total cohort; 6.0 events per 1,000 person-years) had severe COVID-19, consisting of 9,733 who were hospitalized and 2,207 who died (957 of those hospitalized also died).
- People with obesity or severe obesity were at higher risk of hospitalization or death from COVID-19 after both a second and third (booster) dose of vaccine.
- Compared with those with normal weight, those with severe obesity (BMI higher than 40) were at significantly increased risk for severe COVID-19 after a second vaccine dose, with an adjusted rate ratio 1.76, whereas those with standard obesity (BMI, 30-40) were at a modestly but significantly increased risk with an adjusted rate ratio of 1.11.
- Breakthrough infections after the second dose for those with severe obesity, obesity, and normal weight occurred on average at 10 weeks, 15 weeks, and 20 weeks, respectively.
- Interaction testing showed that vaccine effectiveness significantly diminished over time across BMI groups, and protection waned more rapidly as BMI increased.
- Results from immunophenotyping studies run in a subgroup of several dozen subjects with severe obesity or normal weight showed significant decrements in the robustness of antibody responses in those with severe obesity 6 months after a second or third vaccine dose.
Limitations
- The authors did not specify any limitations.
Disclosures
- The study received no commercial funding.
- One author received funding from Wellcome.
This is a summary of a preprint research study , “Accelerated waning of the humoral response to SARS-CoV-2 vaccines in obesity,” published by researchers primarily at the University of Cambridge (England), on medRxiv. This study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.
Researchers published the study covered in this summary on medRxiv.org as a preprint that has not yet been peer reviewed.
Key takeaways
- The study results suggest that
- The findings documented evidence of reduced neutralizing antibody capacity 6 months after primary vaccination in people with severe obesity.
- This was a large study involving about more than 3.5 million people who had received at least two doses of COVID-19 vaccine, including more than 650,000 with obesity.
Why this matters
- Obesity is associated with comorbidities that independently increase the risk for severe COVID-19, including type 2 diabetes, chronic kidney disease, and heart failure.
- The authors concluded that additional or more frequent booster doses are likely to be required to maintain protection among people with obesity against COVID-19.
Study design
- Prospective longitudinal study of the incidence and severity of COVID-19 infections and immune responses in a cohort of more than 3.5 million adults from a Scottish healthcare database who received two or three doses of COVID-19 vaccine. The data came from the study, centered at the University of Edinburgh.
- About 16% had obesity with a body mass index of 30-39.9 kg/m2, and an additional 3% had severe obesity with a BMI of 40 or greater.
- Although not specified in this preprint, another said that the vaccines administered in Scotland have been the Pfizer-BioNTech and Oxford-AstraZeneca formulations.
Key results
- Between Sept. 14, 2020, and March 19, 2022, 10,983 people (0.3% of the total cohort; 6.0 events per 1,000 person-years) had severe COVID-19, consisting of 9,733 who were hospitalized and 2,207 who died (957 of those hospitalized also died).
- People with obesity or severe obesity were at higher risk of hospitalization or death from COVID-19 after both a second and third (booster) dose of vaccine.
- Compared with those with normal weight, those with severe obesity (BMI higher than 40) were at significantly increased risk for severe COVID-19 after a second vaccine dose, with an adjusted rate ratio 1.76, whereas those with standard obesity (BMI, 30-40) were at a modestly but significantly increased risk with an adjusted rate ratio of 1.11.
- Breakthrough infections after the second dose for those with severe obesity, obesity, and normal weight occurred on average at 10 weeks, 15 weeks, and 20 weeks, respectively.
- Interaction testing showed that vaccine effectiveness significantly diminished over time across BMI groups, and protection waned more rapidly as BMI increased.
- Results from immunophenotyping studies run in a subgroup of several dozen subjects with severe obesity or normal weight showed significant decrements in the robustness of antibody responses in those with severe obesity 6 months after a second or third vaccine dose.
Limitations
- The authors did not specify any limitations.
Disclosures
- The study received no commercial funding.
- One author received funding from Wellcome.
This is a summary of a preprint research study , “Accelerated waning of the humoral response to SARS-CoV-2 vaccines in obesity,” published by researchers primarily at the University of Cambridge (England), on medRxiv. This study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.
Researchers published the study covered in this summary on medRxiv.org as a preprint that has not yet been peer reviewed.
Key takeaways
- The study results suggest that
- The findings documented evidence of reduced neutralizing antibody capacity 6 months after primary vaccination in people with severe obesity.
- This was a large study involving about more than 3.5 million people who had received at least two doses of COVID-19 vaccine, including more than 650,000 with obesity.
Why this matters
- Obesity is associated with comorbidities that independently increase the risk for severe COVID-19, including type 2 diabetes, chronic kidney disease, and heart failure.
- The authors concluded that additional or more frequent booster doses are likely to be required to maintain protection among people with obesity against COVID-19.
Study design
- Prospective longitudinal study of the incidence and severity of COVID-19 infections and immune responses in a cohort of more than 3.5 million adults from a Scottish healthcare database who received two or three doses of COVID-19 vaccine. The data came from the study, centered at the University of Edinburgh.
- About 16% had obesity with a body mass index of 30-39.9 kg/m2, and an additional 3% had severe obesity with a BMI of 40 or greater.
- Although not specified in this preprint, another said that the vaccines administered in Scotland have been the Pfizer-BioNTech and Oxford-AstraZeneca formulations.
Key results
- Between Sept. 14, 2020, and March 19, 2022, 10,983 people (0.3% of the total cohort; 6.0 events per 1,000 person-years) had severe COVID-19, consisting of 9,733 who were hospitalized and 2,207 who died (957 of those hospitalized also died).
- People with obesity or severe obesity were at higher risk of hospitalization or death from COVID-19 after both a second and third (booster) dose of vaccine.
- Compared with those with normal weight, those with severe obesity (BMI higher than 40) were at significantly increased risk for severe COVID-19 after a second vaccine dose, with an adjusted rate ratio 1.76, whereas those with standard obesity (BMI, 30-40) were at a modestly but significantly increased risk with an adjusted rate ratio of 1.11.
- Breakthrough infections after the second dose for those with severe obesity, obesity, and normal weight occurred on average at 10 weeks, 15 weeks, and 20 weeks, respectively.
- Interaction testing showed that vaccine effectiveness significantly diminished over time across BMI groups, and protection waned more rapidly as BMI increased.
- Results from immunophenotyping studies run in a subgroup of several dozen subjects with severe obesity or normal weight showed significant decrements in the robustness of antibody responses in those with severe obesity 6 months after a second or third vaccine dose.
Limitations
- The authors did not specify any limitations.
Disclosures
- The study received no commercial funding.
- One author received funding from Wellcome.
This is a summary of a preprint research study , “Accelerated waning of the humoral response to SARS-CoV-2 vaccines in obesity,” published by researchers primarily at the University of Cambridge (England), on medRxiv. This study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.
Long COVID-19 in children and adolescents: What do we know?
Among scientists, the existence of long COVID-19 in children and adolescents has been the subject of debate.
Published by a Mexican multidisciplinary group in Scientific Reports, the first study is a systematic review and meta-analysis. It identified mood symptoms as the most prevalent clinical manifestations of long COVID-19 in children and adolescents. These symptoms included sadness, tension, anger, depression, and anxiety (16.50%); fatigue (9.66%); and sleep disorders (8.42%).
The second study, LongCOVIDKidsDK, was conducted in Denmark. It compared 11,000 children younger than 14 years who had tested positive for COVID-19 with 33,000 children who had no history of COVID-19. The study was published in The Lancet Child and Adolescent Health.
Definitions are changing
In their meta-analysis, the researchers estimated the prevalence and counted signs and symptoms of long COVID-19, as defined by the United Kingdom’s National Institute for Health and Care Excellence. Long COVID-19 was defined as the presence of one or more symptoms more than 4 weeks after SARS-CoV-2 infection. For search terms, the researchers used “COVID-19,” “COVID,” “SARSCOV-2,” “coronavirus,” “long COVID,” “postCOVID,” “PASC,” “long-haulers,” “prolonged,” “post-acute,” “persistent,” “convalescent,” “sequelae,” and “postviral.”
Of the 8,373 citations returned by the search as of Feb. 10, 2022, 21 prospective studies, 2 of them on preprint servers, met the authors’ selection criteria. Those studies included a total of 80,071 children and adolescents younger than 18 years.
In the meta-analysis, the prevalence of long COVID-19 among children and adolescents was reported to be 25.24% (95% confidence interval, 18.17-33.02; I2, 99.61%), regardless of whether the case had been asymptomatic, mild, moderate, severe, or serious. For patients who had been hospitalized, the prevalence was 29.19% (95% CI, 17.83-41.98; I2, 80.84%).
These numbers, while striking, are not the focus of the study, according to first author Sandra Lopez-Leon, MD, PhD, associate professor of pharmacoepidemiology at Rutgers University, New Brunswick, N.J. “It’s important that we don’t focus on that 25%,” she said in an interview. “It’s a disease that we’re learning about, we’re at a time when the definitions are still changing, and, depending on when it is measured, a different number will be given. The message we want to give is that long COVID-19 exists, it’s happening in children and adolescents, and patients need this recognition. And also to show that it can affect the whole body.”
The study showed that the children and adolescents who presented with SARS-CoV-2 infection were at higher risk of subsequent long dyspnea, anosmia/ageusia, or fever, compared with control persons.
In total, in the studies that were included, more than 40 long-term clinical manifestations associated with COVID-19 in the pediatric population were identified.
The most common symptoms among children aged 0-3 years were mood swings, skin rashes, and stomachaches. In 4- to 11-year-olds, the most common symptoms were mood swings, trouble remembering or concentrating, and skin rashes. In 12- to 14-year-olds, they were fatigue, mood swings, and trouble remembering or concentrating. These data are based on parent responses.
The list of signs and symptoms also includes headache, respiratory symptoms, cognitive symptoms (such as decreased concentration, learning difficulties, confusion, and memory loss), loss of appetite, and smell disorder (hyposmia, anosmia, hyperosmia, parosmia, and phantom smell).
In the studies, the prevalence of the following symptoms was less than 5%: hyperhidrosis, chest pain, dizziness, cough, myalgia/arthralgia, changes in body weight, taste disorder, otalgia (tinnitus, ear pain, vertigo), ophthalmologic symptoms (conjunctivitis, dry eye, blurred vision, photophobia, pain), dermatologic symptoms (dry skin, itchy skin, rashes, hives, hair loss), urinary symptoms, abdominal pain, throat pain, chest tightness, variations in heart rate, palpitations, constipation, dysphonia, fever, diarrhea, vomiting/nausea, menstrual changes, neurological abnormalities, speech disorders, and dysphagia.
The authors made it clear that the frequency and severity of these symptoms can fluctuate from one patient to another.
“The meta-analysis is important because it brings together 21 studies selected from more than 8,000 articles – and in them, a large number of children – to study the most common manifestations of long COVID-19,” Gabriela Ensinck, MD, head of the infectious diseases department at the Víctor J. Vilela Children’s Hospital in Rosario, Argentina, told this news organization. Dr. Ensinck did not participate in the study. “The important thing here is that long COVID-19 exists in pediatrics. And that it is a prolongation of signs or symptoms over time, a time for which there is no single definition.”
“It’s a snapshot of all the symptoms that can remain after COVID-19,” Dr. Lopez-Leon explained. “The meta-analysis seeks to see if there’s an association between having had COVID-19 and having the symptoms, but at no time does it speak of causality.”
The prevalence of symptoms largely depends on the time since the onset of acute COVID-19. Most symptoms improve over time. In the studies that were included in the meta-analysis, the follow-up time varied between 1 and 13 months. It is important to understand what symptoms are associated with each period after the onset of infection, the authors said.
Danish parent survey
The Danish study LongCOVIDKidsDK followed the World Health Organization criteria for long COVID-19 and included children and adolescents aged 0-14 years who received a diagnosis of COVID-19 and who experienced symptoms that lasted at least 2 months.
Between July 20, 2021, and Sept. 15, 2021, a questionnaire was sent to 38,152 case patients and 147,212 control persons. Of this group, 10,997 (28.8%) case patients and 33,016 (22.4%) control persons answered the survey.
Children who had been diagnosed with SARS-CoV-2 infection were more likely to experience long-lasting symptoms than children who had never been diagnosed. Approximately one-third of children with a positive SARS-CoV-2 test experienced symptoms that were not present before infection. Children who experienced long-lasting symptoms included 40% of children diagnosed with COVID-19 and 27% of control persons aged 0-3 years, 38% of case patients and 34% of control persons aged 4-11 years, and 46% of case patients and 41% of control persons aged 12-14 years.
Interestingly, those diagnosed with COVID-19 reported fewer psychological and social problems than those in the control group. Among the oldest (aged 12-14 years), quality of life scores were higher and anxiety scores were lower for those who had tested positive for SARS-CoV-2.
More information needed
Given the diversity of symptoms in the meta-analysis and the LongCOVIDKidsDK study, a multidisciplinary approach is imperative. Dr. Lopez-Leon suggests that there is a need to raise awareness among parents, clinicians, researchers, and the health system about the conditions that can occur after COVID-19. Clinicians must better understand the sequelae to provide targeted care and treatment. The authors of the Danish study recommend establishing clinics for long COVID-19 with multispecialty care.
Maren J. Heilskov Rytter, PhD, associate professor of clinical medicine at the University of Copenhagen, wrote an editorial in The Lancet Child and Adolescent Health about the Danish study. Until it is clarified whether SARS-CoV-2 does indeed cause persistent symptoms, she wrote, “it seems excessive and premature to establish specific multidisciplinary clinics for children with long COVID-19.”
Dr. Rytter highlighted the difficulty of interpreting LongCOVIDKidsDK data, owing to recall bias, the failure to exclude other causes of symptoms in the cases analyzed, and the number of symptoms in the control persons. In addition, the data analyzed in Denmark are of limited clinical relevance, she said, given a greater presence of mild symptoms and, paradoxically, a higher quality of life.
She concluded, “In the majority of children with nonspecific symptoms after COVID-19, the symptoms presented are more likely to have been caused by something other than COVID-19, and if they are related to COVID-19, they are likely to go away over time.”
Dr. Ensinck, who is coauthor of the Argentine Ministry of Health’s guide for long COVID-19 monitoring for children and adolescents and who represented the Infectious Diseases Committee of the Argentine Society of Pediatrics, highlighted another aspect of the problem. “What should be taken into account in these data is to see how much the confinement contributed. Children are the ones who suffered the most in the period in which schools were closed; they could not meet their peers, they had sick relatives, they felt fear. … all this must be taken into account.”
There is as yet no agreement on how to define and diagnose long COVID-19 in adults, a population that has been studied more closely. Part of the problem is that long COVID-19 has been linked to more than 200 symptoms, which can range in severity from inconvenient to debilitating, can last for months or years, and can recur, sometimes months after apparent recovery. Thus, there are still disparate answers to basic questions about the syndrome’s frequency and its effects on vaccination, reinfection, and the latest variant of SARS-CoV-2.
This article has been translated from the Medscape Spanish edition. A version appeared on Medscape.com.
Among scientists, the existence of long COVID-19 in children and adolescents has been the subject of debate.
Published by a Mexican multidisciplinary group in Scientific Reports, the first study is a systematic review and meta-analysis. It identified mood symptoms as the most prevalent clinical manifestations of long COVID-19 in children and adolescents. These symptoms included sadness, tension, anger, depression, and anxiety (16.50%); fatigue (9.66%); and sleep disorders (8.42%).
The second study, LongCOVIDKidsDK, was conducted in Denmark. It compared 11,000 children younger than 14 years who had tested positive for COVID-19 with 33,000 children who had no history of COVID-19. The study was published in The Lancet Child and Adolescent Health.
Definitions are changing
In their meta-analysis, the researchers estimated the prevalence and counted signs and symptoms of long COVID-19, as defined by the United Kingdom’s National Institute for Health and Care Excellence. Long COVID-19 was defined as the presence of one or more symptoms more than 4 weeks after SARS-CoV-2 infection. For search terms, the researchers used “COVID-19,” “COVID,” “SARSCOV-2,” “coronavirus,” “long COVID,” “postCOVID,” “PASC,” “long-haulers,” “prolonged,” “post-acute,” “persistent,” “convalescent,” “sequelae,” and “postviral.”
Of the 8,373 citations returned by the search as of Feb. 10, 2022, 21 prospective studies, 2 of them on preprint servers, met the authors’ selection criteria. Those studies included a total of 80,071 children and adolescents younger than 18 years.
In the meta-analysis, the prevalence of long COVID-19 among children and adolescents was reported to be 25.24% (95% confidence interval, 18.17-33.02; I2, 99.61%), regardless of whether the case had been asymptomatic, mild, moderate, severe, or serious. For patients who had been hospitalized, the prevalence was 29.19% (95% CI, 17.83-41.98; I2, 80.84%).
These numbers, while striking, are not the focus of the study, according to first author Sandra Lopez-Leon, MD, PhD, associate professor of pharmacoepidemiology at Rutgers University, New Brunswick, N.J. “It’s important that we don’t focus on that 25%,” she said in an interview. “It’s a disease that we’re learning about, we’re at a time when the definitions are still changing, and, depending on when it is measured, a different number will be given. The message we want to give is that long COVID-19 exists, it’s happening in children and adolescents, and patients need this recognition. And also to show that it can affect the whole body.”
The study showed that the children and adolescents who presented with SARS-CoV-2 infection were at higher risk of subsequent long dyspnea, anosmia/ageusia, or fever, compared with control persons.
In total, in the studies that were included, more than 40 long-term clinical manifestations associated with COVID-19 in the pediatric population were identified.
The most common symptoms among children aged 0-3 years were mood swings, skin rashes, and stomachaches. In 4- to 11-year-olds, the most common symptoms were mood swings, trouble remembering or concentrating, and skin rashes. In 12- to 14-year-olds, they were fatigue, mood swings, and trouble remembering or concentrating. These data are based on parent responses.
The list of signs and symptoms also includes headache, respiratory symptoms, cognitive symptoms (such as decreased concentration, learning difficulties, confusion, and memory loss), loss of appetite, and smell disorder (hyposmia, anosmia, hyperosmia, parosmia, and phantom smell).
In the studies, the prevalence of the following symptoms was less than 5%: hyperhidrosis, chest pain, dizziness, cough, myalgia/arthralgia, changes in body weight, taste disorder, otalgia (tinnitus, ear pain, vertigo), ophthalmologic symptoms (conjunctivitis, dry eye, blurred vision, photophobia, pain), dermatologic symptoms (dry skin, itchy skin, rashes, hives, hair loss), urinary symptoms, abdominal pain, throat pain, chest tightness, variations in heart rate, palpitations, constipation, dysphonia, fever, diarrhea, vomiting/nausea, menstrual changes, neurological abnormalities, speech disorders, and dysphagia.
The authors made it clear that the frequency and severity of these symptoms can fluctuate from one patient to another.
“The meta-analysis is important because it brings together 21 studies selected from more than 8,000 articles – and in them, a large number of children – to study the most common manifestations of long COVID-19,” Gabriela Ensinck, MD, head of the infectious diseases department at the Víctor J. Vilela Children’s Hospital in Rosario, Argentina, told this news organization. Dr. Ensinck did not participate in the study. “The important thing here is that long COVID-19 exists in pediatrics. And that it is a prolongation of signs or symptoms over time, a time for which there is no single definition.”
“It’s a snapshot of all the symptoms that can remain after COVID-19,” Dr. Lopez-Leon explained. “The meta-analysis seeks to see if there’s an association between having had COVID-19 and having the symptoms, but at no time does it speak of causality.”
The prevalence of symptoms largely depends on the time since the onset of acute COVID-19. Most symptoms improve over time. In the studies that were included in the meta-analysis, the follow-up time varied between 1 and 13 months. It is important to understand what symptoms are associated with each period after the onset of infection, the authors said.
Danish parent survey
The Danish study LongCOVIDKidsDK followed the World Health Organization criteria for long COVID-19 and included children and adolescents aged 0-14 years who received a diagnosis of COVID-19 and who experienced symptoms that lasted at least 2 months.
Between July 20, 2021, and Sept. 15, 2021, a questionnaire was sent to 38,152 case patients and 147,212 control persons. Of this group, 10,997 (28.8%) case patients and 33,016 (22.4%) control persons answered the survey.
Children who had been diagnosed with SARS-CoV-2 infection were more likely to experience long-lasting symptoms than children who had never been diagnosed. Approximately one-third of children with a positive SARS-CoV-2 test experienced symptoms that were not present before infection. Children who experienced long-lasting symptoms included 40% of children diagnosed with COVID-19 and 27% of control persons aged 0-3 years, 38% of case patients and 34% of control persons aged 4-11 years, and 46% of case patients and 41% of control persons aged 12-14 years.
Interestingly, those diagnosed with COVID-19 reported fewer psychological and social problems than those in the control group. Among the oldest (aged 12-14 years), quality of life scores were higher and anxiety scores were lower for those who had tested positive for SARS-CoV-2.
More information needed
Given the diversity of symptoms in the meta-analysis and the LongCOVIDKidsDK study, a multidisciplinary approach is imperative. Dr. Lopez-Leon suggests that there is a need to raise awareness among parents, clinicians, researchers, and the health system about the conditions that can occur after COVID-19. Clinicians must better understand the sequelae to provide targeted care and treatment. The authors of the Danish study recommend establishing clinics for long COVID-19 with multispecialty care.
Maren J. Heilskov Rytter, PhD, associate professor of clinical medicine at the University of Copenhagen, wrote an editorial in The Lancet Child and Adolescent Health about the Danish study. Until it is clarified whether SARS-CoV-2 does indeed cause persistent symptoms, she wrote, “it seems excessive and premature to establish specific multidisciplinary clinics for children with long COVID-19.”
Dr. Rytter highlighted the difficulty of interpreting LongCOVIDKidsDK data, owing to recall bias, the failure to exclude other causes of symptoms in the cases analyzed, and the number of symptoms in the control persons. In addition, the data analyzed in Denmark are of limited clinical relevance, she said, given a greater presence of mild symptoms and, paradoxically, a higher quality of life.
She concluded, “In the majority of children with nonspecific symptoms after COVID-19, the symptoms presented are more likely to have been caused by something other than COVID-19, and if they are related to COVID-19, they are likely to go away over time.”
Dr. Ensinck, who is coauthor of the Argentine Ministry of Health’s guide for long COVID-19 monitoring for children and adolescents and who represented the Infectious Diseases Committee of the Argentine Society of Pediatrics, highlighted another aspect of the problem. “What should be taken into account in these data is to see how much the confinement contributed. Children are the ones who suffered the most in the period in which schools were closed; they could not meet their peers, they had sick relatives, they felt fear. … all this must be taken into account.”
There is as yet no agreement on how to define and diagnose long COVID-19 in adults, a population that has been studied more closely. Part of the problem is that long COVID-19 has been linked to more than 200 symptoms, which can range in severity from inconvenient to debilitating, can last for months or years, and can recur, sometimes months after apparent recovery. Thus, there are still disparate answers to basic questions about the syndrome’s frequency and its effects on vaccination, reinfection, and the latest variant of SARS-CoV-2.
This article has been translated from the Medscape Spanish edition. A version appeared on Medscape.com.
Among scientists, the existence of long COVID-19 in children and adolescents has been the subject of debate.
Published by a Mexican multidisciplinary group in Scientific Reports, the first study is a systematic review and meta-analysis. It identified mood symptoms as the most prevalent clinical manifestations of long COVID-19 in children and adolescents. These symptoms included sadness, tension, anger, depression, and anxiety (16.50%); fatigue (9.66%); and sleep disorders (8.42%).
The second study, LongCOVIDKidsDK, was conducted in Denmark. It compared 11,000 children younger than 14 years who had tested positive for COVID-19 with 33,000 children who had no history of COVID-19. The study was published in The Lancet Child and Adolescent Health.
Definitions are changing
In their meta-analysis, the researchers estimated the prevalence and counted signs and symptoms of long COVID-19, as defined by the United Kingdom’s National Institute for Health and Care Excellence. Long COVID-19 was defined as the presence of one or more symptoms more than 4 weeks after SARS-CoV-2 infection. For search terms, the researchers used “COVID-19,” “COVID,” “SARSCOV-2,” “coronavirus,” “long COVID,” “postCOVID,” “PASC,” “long-haulers,” “prolonged,” “post-acute,” “persistent,” “convalescent,” “sequelae,” and “postviral.”
Of the 8,373 citations returned by the search as of Feb. 10, 2022, 21 prospective studies, 2 of them on preprint servers, met the authors’ selection criteria. Those studies included a total of 80,071 children and adolescents younger than 18 years.
In the meta-analysis, the prevalence of long COVID-19 among children and adolescents was reported to be 25.24% (95% confidence interval, 18.17-33.02; I2, 99.61%), regardless of whether the case had been asymptomatic, mild, moderate, severe, or serious. For patients who had been hospitalized, the prevalence was 29.19% (95% CI, 17.83-41.98; I2, 80.84%).
These numbers, while striking, are not the focus of the study, according to first author Sandra Lopez-Leon, MD, PhD, associate professor of pharmacoepidemiology at Rutgers University, New Brunswick, N.J. “It’s important that we don’t focus on that 25%,” she said in an interview. “It’s a disease that we’re learning about, we’re at a time when the definitions are still changing, and, depending on when it is measured, a different number will be given. The message we want to give is that long COVID-19 exists, it’s happening in children and adolescents, and patients need this recognition. And also to show that it can affect the whole body.”
The study showed that the children and adolescents who presented with SARS-CoV-2 infection were at higher risk of subsequent long dyspnea, anosmia/ageusia, or fever, compared with control persons.
In total, in the studies that were included, more than 40 long-term clinical manifestations associated with COVID-19 in the pediatric population were identified.
The most common symptoms among children aged 0-3 years were mood swings, skin rashes, and stomachaches. In 4- to 11-year-olds, the most common symptoms were mood swings, trouble remembering or concentrating, and skin rashes. In 12- to 14-year-olds, they were fatigue, mood swings, and trouble remembering or concentrating. These data are based on parent responses.
The list of signs and symptoms also includes headache, respiratory symptoms, cognitive symptoms (such as decreased concentration, learning difficulties, confusion, and memory loss), loss of appetite, and smell disorder (hyposmia, anosmia, hyperosmia, parosmia, and phantom smell).
In the studies, the prevalence of the following symptoms was less than 5%: hyperhidrosis, chest pain, dizziness, cough, myalgia/arthralgia, changes in body weight, taste disorder, otalgia (tinnitus, ear pain, vertigo), ophthalmologic symptoms (conjunctivitis, dry eye, blurred vision, photophobia, pain), dermatologic symptoms (dry skin, itchy skin, rashes, hives, hair loss), urinary symptoms, abdominal pain, throat pain, chest tightness, variations in heart rate, palpitations, constipation, dysphonia, fever, diarrhea, vomiting/nausea, menstrual changes, neurological abnormalities, speech disorders, and dysphagia.
The authors made it clear that the frequency and severity of these symptoms can fluctuate from one patient to another.
“The meta-analysis is important because it brings together 21 studies selected from more than 8,000 articles – and in them, a large number of children – to study the most common manifestations of long COVID-19,” Gabriela Ensinck, MD, head of the infectious diseases department at the Víctor J. Vilela Children’s Hospital in Rosario, Argentina, told this news organization. Dr. Ensinck did not participate in the study. “The important thing here is that long COVID-19 exists in pediatrics. And that it is a prolongation of signs or symptoms over time, a time for which there is no single definition.”
“It’s a snapshot of all the symptoms that can remain after COVID-19,” Dr. Lopez-Leon explained. “The meta-analysis seeks to see if there’s an association between having had COVID-19 and having the symptoms, but at no time does it speak of causality.”
The prevalence of symptoms largely depends on the time since the onset of acute COVID-19. Most symptoms improve over time. In the studies that were included in the meta-analysis, the follow-up time varied between 1 and 13 months. It is important to understand what symptoms are associated with each period after the onset of infection, the authors said.
Danish parent survey
The Danish study LongCOVIDKidsDK followed the World Health Organization criteria for long COVID-19 and included children and adolescents aged 0-14 years who received a diagnosis of COVID-19 and who experienced symptoms that lasted at least 2 months.
Between July 20, 2021, and Sept. 15, 2021, a questionnaire was sent to 38,152 case patients and 147,212 control persons. Of this group, 10,997 (28.8%) case patients and 33,016 (22.4%) control persons answered the survey.
Children who had been diagnosed with SARS-CoV-2 infection were more likely to experience long-lasting symptoms than children who had never been diagnosed. Approximately one-third of children with a positive SARS-CoV-2 test experienced symptoms that were not present before infection. Children who experienced long-lasting symptoms included 40% of children diagnosed with COVID-19 and 27% of control persons aged 0-3 years, 38% of case patients and 34% of control persons aged 4-11 years, and 46% of case patients and 41% of control persons aged 12-14 years.
Interestingly, those diagnosed with COVID-19 reported fewer psychological and social problems than those in the control group. Among the oldest (aged 12-14 years), quality of life scores were higher and anxiety scores were lower for those who had tested positive for SARS-CoV-2.
More information needed
Given the diversity of symptoms in the meta-analysis and the LongCOVIDKidsDK study, a multidisciplinary approach is imperative. Dr. Lopez-Leon suggests that there is a need to raise awareness among parents, clinicians, researchers, and the health system about the conditions that can occur after COVID-19. Clinicians must better understand the sequelae to provide targeted care and treatment. The authors of the Danish study recommend establishing clinics for long COVID-19 with multispecialty care.
Maren J. Heilskov Rytter, PhD, associate professor of clinical medicine at the University of Copenhagen, wrote an editorial in The Lancet Child and Adolescent Health about the Danish study. Until it is clarified whether SARS-CoV-2 does indeed cause persistent symptoms, she wrote, “it seems excessive and premature to establish specific multidisciplinary clinics for children with long COVID-19.”
Dr. Rytter highlighted the difficulty of interpreting LongCOVIDKidsDK data, owing to recall bias, the failure to exclude other causes of symptoms in the cases analyzed, and the number of symptoms in the control persons. In addition, the data analyzed in Denmark are of limited clinical relevance, she said, given a greater presence of mild symptoms and, paradoxically, a higher quality of life.
She concluded, “In the majority of children with nonspecific symptoms after COVID-19, the symptoms presented are more likely to have been caused by something other than COVID-19, and if they are related to COVID-19, they are likely to go away over time.”
Dr. Ensinck, who is coauthor of the Argentine Ministry of Health’s guide for long COVID-19 monitoring for children and adolescents and who represented the Infectious Diseases Committee of the Argentine Society of Pediatrics, highlighted another aspect of the problem. “What should be taken into account in these data is to see how much the confinement contributed. Children are the ones who suffered the most in the period in which schools were closed; they could not meet their peers, they had sick relatives, they felt fear. … all this must be taken into account.”
There is as yet no agreement on how to define and diagnose long COVID-19 in adults, a population that has been studied more closely. Part of the problem is that long COVID-19 has been linked to more than 200 symptoms, which can range in severity from inconvenient to debilitating, can last for months or years, and can recur, sometimes months after apparent recovery. Thus, there are still disparate answers to basic questions about the syndrome’s frequency and its effects on vaccination, reinfection, and the latest variant of SARS-CoV-2.
This article has been translated from the Medscape Spanish edition. A version appeared on Medscape.com.
FROM SCIENTIFIC REPORTS AND THE LANCET CHILD AND ADOLESCENT HEALTH
Sleep-deprived physicians less empathetic to patient pain?
new research suggests.
In the first of two studies, resident physicians were presented with two hypothetical scenarios involving a patient who complains of pain. They were asked about their likelihood of prescribing pain medication. The test was given to one group of residents who were just starting their day and to another group who were at the end of their night shift after being on call for 26 hours.
Results showed that the night shift residents were less likely than their daytime counterparts to say they would prescribe pain medication to the patients.
In further analysis of discharge notes from more than 13,000 electronic records of patients presenting with pain complaints at hospitals in Israel and the United States, the likelihood of an analgesic being prescribed during the night shift was 11% lower in Israel and 9% lower in the United States, compared with the day shift.
“Pain management is a major challenge, and a doctor’s perception of a patient’s subjective pain is susceptible to bias,” coinvestigator David Gozal, MD, the Marie M. and Harry L. Smith Endowed Chair of Child Health, University of Missouri–Columbia, said in a press release.
“This study demonstrated that night shift work is an important and previously unrecognized source of bias in pain management, likely stemming from impaired perception of pain,” Dr. Gozal added.
The findings were published online in the Proceedings of the National Academy of Sciences.
‘Directional’ differences
Senior investigator Alex Gileles-Hillel, MD, senior pediatric pulmonologist and sleep researcher at Hadassah University Medical Center, Jerusalem, said in an interview that physicians must make “complex assessments of patients’ subjective pain experience” – and the “subjective nature of pain management decisions can give rise to various biases.”
Dr. Gileles-Hillel has previously researched the cognitive toll of night shift work on physicians.
“It’s pretty established, for example, not to drive when sleep deprived because cognition is impaired,” he said. The current study explored whether sleep deprivation could affect areas other than cognition, including emotions and empathy.
The researchers used “two complementary approaches.” First, they administered tests to measure empathy and pain management decisions in 67 resident physicians at Hadassah Medical Centers either following a 26-hour night shift that began at 8:00 a.m. the day before (n = 36) or immediately before starting the workday (n = 31).
There were no significant differences in demographic, sleep, or burnout measures between the two groups, except that night shift physicians had slept less than those in the daytime group (2.93 vs. 5.96 hours).
Participants completed two tasks. In the empathy-for-pain task, they rated their emotional reactions to pictures of individuals in pain. In the empathy accuracy task, they were asked to assess the feelings of videotaped individuals telling emotional stories.
They were then presented with two clinical scenarios: a female patient with a headache and a male patient with a backache. Following that, they were asked to assess the magnitude of the patients’ pain and how likely they would be to prescribe pain medication.
In the empathy-for-pain task, physicians’ empathy scores were significantly lower in the night shift group than in the day group (difference, –0.83; 95% CI, –1.55 to –0.10; P = .026). There were no significant differences between the groups in the empathy accuracy task.
In both scenarios, physicians in the night shift group assessed the patient’s pain as weaker in comparison with physicians in the day group. There was a statistically significant difference in the headache scenario but not the backache scenario.
In the headache scenario, the propensity of the physicians to prescribe analgesics was “directionally lower” but did not reach statistical significance. In the backache scenario, there was no significant difference between the groups’ prescribing propensities.
In both scenarios, pain assessment was positively correlated with the propensity to prescribe analgesics.
Despite the lack of statistical significance, the findings “documented a negative effect of night shift work on physician empathy for pain and a positive association between physician assessment of patient pain and the propensity to prescribe analgesics,” the investigators wrote.
Need for naps?
The researchers then analyzed analgesic prescription patterns drawn from three datasets of discharge notes of patients presenting to the emergency department with pain complaints (n = 13,482) at two branches of Hadassah-Hebrew University Medical Center and the University of Missouri Health Center.
The researchers collected data, including discharge time, medications patients were prescribed upon discharge, and patients’ subjective pain rating on a scale of 0-10 on a visual analogue scale (VAS).
Although patients’ VAS scores did not differ with respect to time or shift, patients were discharged with significantly less prescribed analgesics during the night shift in comparison with the day shift.
No similar differences in prescriptions between night shifts and day shifts were found for nonanalgesic medications, such as for diabetes or blood pressure. This suggests “the effect was specific to pain,” Dr. Gileles-Hillel said.
The pattern remained significant after controlling for potential confounders, including patient and physician variables and emergency department characteristics.
In addition, patients seen during night shifts received fewer analgesics, particularly opioids, than recommended by the World Health Organization for pain management.
“The first study enabled us to measure empathy for pain directly and examine our hypothesis in a controlled environment, while the second enabled us to test the implications by examining real-life pain management decisions,” Dr. Gileles-Hillel said.
“Physicians need to be aware of this,” he noted. “I try to be aware when I’m taking calls [at night] that I’m less empathetic to others and I might be more brief or angry with others.”
On a “house management level, perhaps institutions should try to schedule naps either before or during overnight call. A nap might give a boost and reboot not only to cognitive but also to emotional resources,” Dr. Gileles-Hillel added.
Compromised safety
In a comment, Eti Ben Simon, PhD, a postdoctoral fellow at the Center for Human Sleep Science, University of California, Berkeley, called the study “an important contribution to a growing list of studies that reveal how long night shifts reduce overall safety” for both patients and clinicians.
“It’s time to abandon the notion that the human brain can function as normal after being deprived of sleep for 24 hours,” said Dr. Ben Simon, who was not involved with the research.
“This is especially true in medicine, where we trust others to take care of us and feel our pain. These functions are simply not possible without adequate sleep,” she added.
Also commenting, Kannan Ramar, MD, president of the American Academy of Sleep Medicine, suggested that being cognizant of these findings “may help providers to mitigate this bias” of underprescribing pain medications when treating their patients.
Dr. Ramar, who is also a critical care specialist, pulmonologist, and sleep medicine specialist at Mayo Clinic, Rochester, Minn., was not involved with the research.
He noted that “further studies that systematically evaluate this further in a prospective and blinded way will be important.”
The research was supported in part by grants from the Israel Science Foundation, Joy Ventures, the Recanati Fund at the Jerusalem School of Business at the Hebrew University, and a fellowship from the Azrieli Foundation and received grant support to various investigators from the NIH, the Leda J. Sears Foundation, and the University of Missouri. The investigators, Ramar, and Ben Simon have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
In the first of two studies, resident physicians were presented with two hypothetical scenarios involving a patient who complains of pain. They were asked about their likelihood of prescribing pain medication. The test was given to one group of residents who were just starting their day and to another group who were at the end of their night shift after being on call for 26 hours.
Results showed that the night shift residents were less likely than their daytime counterparts to say they would prescribe pain medication to the patients.
In further analysis of discharge notes from more than 13,000 electronic records of patients presenting with pain complaints at hospitals in Israel and the United States, the likelihood of an analgesic being prescribed during the night shift was 11% lower in Israel and 9% lower in the United States, compared with the day shift.
“Pain management is a major challenge, and a doctor’s perception of a patient’s subjective pain is susceptible to bias,” coinvestigator David Gozal, MD, the Marie M. and Harry L. Smith Endowed Chair of Child Health, University of Missouri–Columbia, said in a press release.
“This study demonstrated that night shift work is an important and previously unrecognized source of bias in pain management, likely stemming from impaired perception of pain,” Dr. Gozal added.
The findings were published online in the Proceedings of the National Academy of Sciences.
‘Directional’ differences
Senior investigator Alex Gileles-Hillel, MD, senior pediatric pulmonologist and sleep researcher at Hadassah University Medical Center, Jerusalem, said in an interview that physicians must make “complex assessments of patients’ subjective pain experience” – and the “subjective nature of pain management decisions can give rise to various biases.”
Dr. Gileles-Hillel has previously researched the cognitive toll of night shift work on physicians.
“It’s pretty established, for example, not to drive when sleep deprived because cognition is impaired,” he said. The current study explored whether sleep deprivation could affect areas other than cognition, including emotions and empathy.
The researchers used “two complementary approaches.” First, they administered tests to measure empathy and pain management decisions in 67 resident physicians at Hadassah Medical Centers either following a 26-hour night shift that began at 8:00 a.m. the day before (n = 36) or immediately before starting the workday (n = 31).
There were no significant differences in demographic, sleep, or burnout measures between the two groups, except that night shift physicians had slept less than those in the daytime group (2.93 vs. 5.96 hours).
Participants completed two tasks. In the empathy-for-pain task, they rated their emotional reactions to pictures of individuals in pain. In the empathy accuracy task, they were asked to assess the feelings of videotaped individuals telling emotional stories.
They were then presented with two clinical scenarios: a female patient with a headache and a male patient with a backache. Following that, they were asked to assess the magnitude of the patients’ pain and how likely they would be to prescribe pain medication.
In the empathy-for-pain task, physicians’ empathy scores were significantly lower in the night shift group than in the day group (difference, –0.83; 95% CI, –1.55 to –0.10; P = .026). There were no significant differences between the groups in the empathy accuracy task.
In both scenarios, physicians in the night shift group assessed the patient’s pain as weaker in comparison with physicians in the day group. There was a statistically significant difference in the headache scenario but not the backache scenario.
In the headache scenario, the propensity of the physicians to prescribe analgesics was “directionally lower” but did not reach statistical significance. In the backache scenario, there was no significant difference between the groups’ prescribing propensities.
In both scenarios, pain assessment was positively correlated with the propensity to prescribe analgesics.
Despite the lack of statistical significance, the findings “documented a negative effect of night shift work on physician empathy for pain and a positive association between physician assessment of patient pain and the propensity to prescribe analgesics,” the investigators wrote.
Need for naps?
The researchers then analyzed analgesic prescription patterns drawn from three datasets of discharge notes of patients presenting to the emergency department with pain complaints (n = 13,482) at two branches of Hadassah-Hebrew University Medical Center and the University of Missouri Health Center.
The researchers collected data, including discharge time, medications patients were prescribed upon discharge, and patients’ subjective pain rating on a scale of 0-10 on a visual analogue scale (VAS).
Although patients’ VAS scores did not differ with respect to time or shift, patients were discharged with significantly less prescribed analgesics during the night shift in comparison with the day shift.
No similar differences in prescriptions between night shifts and day shifts were found for nonanalgesic medications, such as for diabetes or blood pressure. This suggests “the effect was specific to pain,” Dr. Gileles-Hillel said.
The pattern remained significant after controlling for potential confounders, including patient and physician variables and emergency department characteristics.
In addition, patients seen during night shifts received fewer analgesics, particularly opioids, than recommended by the World Health Organization for pain management.
“The first study enabled us to measure empathy for pain directly and examine our hypothesis in a controlled environment, while the second enabled us to test the implications by examining real-life pain management decisions,” Dr. Gileles-Hillel said.
“Physicians need to be aware of this,” he noted. “I try to be aware when I’m taking calls [at night] that I’m less empathetic to others and I might be more brief or angry with others.”
On a “house management level, perhaps institutions should try to schedule naps either before or during overnight call. A nap might give a boost and reboot not only to cognitive but also to emotional resources,” Dr. Gileles-Hillel added.
Compromised safety
In a comment, Eti Ben Simon, PhD, a postdoctoral fellow at the Center for Human Sleep Science, University of California, Berkeley, called the study “an important contribution to a growing list of studies that reveal how long night shifts reduce overall safety” for both patients and clinicians.
“It’s time to abandon the notion that the human brain can function as normal after being deprived of sleep for 24 hours,” said Dr. Ben Simon, who was not involved with the research.
“This is especially true in medicine, where we trust others to take care of us and feel our pain. These functions are simply not possible without adequate sleep,” she added.
Also commenting, Kannan Ramar, MD, president of the American Academy of Sleep Medicine, suggested that being cognizant of these findings “may help providers to mitigate this bias” of underprescribing pain medications when treating their patients.
Dr. Ramar, who is also a critical care specialist, pulmonologist, and sleep medicine specialist at Mayo Clinic, Rochester, Minn., was not involved with the research.
He noted that “further studies that systematically evaluate this further in a prospective and blinded way will be important.”
The research was supported in part by grants from the Israel Science Foundation, Joy Ventures, the Recanati Fund at the Jerusalem School of Business at the Hebrew University, and a fellowship from the Azrieli Foundation and received grant support to various investigators from the NIH, the Leda J. Sears Foundation, and the University of Missouri. The investigators, Ramar, and Ben Simon have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
In the first of two studies, resident physicians were presented with two hypothetical scenarios involving a patient who complains of pain. They were asked about their likelihood of prescribing pain medication. The test was given to one group of residents who were just starting their day and to another group who were at the end of their night shift after being on call for 26 hours.
Results showed that the night shift residents were less likely than their daytime counterparts to say they would prescribe pain medication to the patients.
In further analysis of discharge notes from more than 13,000 electronic records of patients presenting with pain complaints at hospitals in Israel and the United States, the likelihood of an analgesic being prescribed during the night shift was 11% lower in Israel and 9% lower in the United States, compared with the day shift.
“Pain management is a major challenge, and a doctor’s perception of a patient’s subjective pain is susceptible to bias,” coinvestigator David Gozal, MD, the Marie M. and Harry L. Smith Endowed Chair of Child Health, University of Missouri–Columbia, said in a press release.
“This study demonstrated that night shift work is an important and previously unrecognized source of bias in pain management, likely stemming from impaired perception of pain,” Dr. Gozal added.
The findings were published online in the Proceedings of the National Academy of Sciences.
‘Directional’ differences
Senior investigator Alex Gileles-Hillel, MD, senior pediatric pulmonologist and sleep researcher at Hadassah University Medical Center, Jerusalem, said in an interview that physicians must make “complex assessments of patients’ subjective pain experience” – and the “subjective nature of pain management decisions can give rise to various biases.”
Dr. Gileles-Hillel has previously researched the cognitive toll of night shift work on physicians.
“It’s pretty established, for example, not to drive when sleep deprived because cognition is impaired,” he said. The current study explored whether sleep deprivation could affect areas other than cognition, including emotions and empathy.
The researchers used “two complementary approaches.” First, they administered tests to measure empathy and pain management decisions in 67 resident physicians at Hadassah Medical Centers either following a 26-hour night shift that began at 8:00 a.m. the day before (n = 36) or immediately before starting the workday (n = 31).
There were no significant differences in demographic, sleep, or burnout measures between the two groups, except that night shift physicians had slept less than those in the daytime group (2.93 vs. 5.96 hours).
Participants completed two tasks. In the empathy-for-pain task, they rated their emotional reactions to pictures of individuals in pain. In the empathy accuracy task, they were asked to assess the feelings of videotaped individuals telling emotional stories.
They were then presented with two clinical scenarios: a female patient with a headache and a male patient with a backache. Following that, they were asked to assess the magnitude of the patients’ pain and how likely they would be to prescribe pain medication.
In the empathy-for-pain task, physicians’ empathy scores were significantly lower in the night shift group than in the day group (difference, –0.83; 95% CI, –1.55 to –0.10; P = .026). There were no significant differences between the groups in the empathy accuracy task.
In both scenarios, physicians in the night shift group assessed the patient’s pain as weaker in comparison with physicians in the day group. There was a statistically significant difference in the headache scenario but not the backache scenario.
In the headache scenario, the propensity of the physicians to prescribe analgesics was “directionally lower” but did not reach statistical significance. In the backache scenario, there was no significant difference between the groups’ prescribing propensities.
In both scenarios, pain assessment was positively correlated with the propensity to prescribe analgesics.
Despite the lack of statistical significance, the findings “documented a negative effect of night shift work on physician empathy for pain and a positive association between physician assessment of patient pain and the propensity to prescribe analgesics,” the investigators wrote.
Need for naps?
The researchers then analyzed analgesic prescription patterns drawn from three datasets of discharge notes of patients presenting to the emergency department with pain complaints (n = 13,482) at two branches of Hadassah-Hebrew University Medical Center and the University of Missouri Health Center.
The researchers collected data, including discharge time, medications patients were prescribed upon discharge, and patients’ subjective pain rating on a scale of 0-10 on a visual analogue scale (VAS).
Although patients’ VAS scores did not differ with respect to time or shift, patients were discharged with significantly less prescribed analgesics during the night shift in comparison with the day shift.
No similar differences in prescriptions between night shifts and day shifts were found for nonanalgesic medications, such as for diabetes or blood pressure. This suggests “the effect was specific to pain,” Dr. Gileles-Hillel said.
The pattern remained significant after controlling for potential confounders, including patient and physician variables and emergency department characteristics.
In addition, patients seen during night shifts received fewer analgesics, particularly opioids, than recommended by the World Health Organization for pain management.
“The first study enabled us to measure empathy for pain directly and examine our hypothesis in a controlled environment, while the second enabled us to test the implications by examining real-life pain management decisions,” Dr. Gileles-Hillel said.
“Physicians need to be aware of this,” he noted. “I try to be aware when I’m taking calls [at night] that I’m less empathetic to others and I might be more brief or angry with others.”
On a “house management level, perhaps institutions should try to schedule naps either before or during overnight call. A nap might give a boost and reboot not only to cognitive but also to emotional resources,” Dr. Gileles-Hillel added.
Compromised safety
In a comment, Eti Ben Simon, PhD, a postdoctoral fellow at the Center for Human Sleep Science, University of California, Berkeley, called the study “an important contribution to a growing list of studies that reveal how long night shifts reduce overall safety” for both patients and clinicians.
“It’s time to abandon the notion that the human brain can function as normal after being deprived of sleep for 24 hours,” said Dr. Ben Simon, who was not involved with the research.
“This is especially true in medicine, where we trust others to take care of us and feel our pain. These functions are simply not possible without adequate sleep,” she added.
Also commenting, Kannan Ramar, MD, president of the American Academy of Sleep Medicine, suggested that being cognizant of these findings “may help providers to mitigate this bias” of underprescribing pain medications when treating their patients.
Dr. Ramar, who is also a critical care specialist, pulmonologist, and sleep medicine specialist at Mayo Clinic, Rochester, Minn., was not involved with the research.
He noted that “further studies that systematically evaluate this further in a prospective and blinded way will be important.”
The research was supported in part by grants from the Israel Science Foundation, Joy Ventures, the Recanati Fund at the Jerusalem School of Business at the Hebrew University, and a fellowship from the Azrieli Foundation and received grant support to various investigators from the NIH, the Leda J. Sears Foundation, and the University of Missouri. The investigators, Ramar, and Ben Simon have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES
New European guidelines ‘drastically’ reduce statin eligibility
New risk thresholds used to guide statin therapy for primary prevention of atherosclerotic cardiovascular disease in the latest European guidelines dramatically reduce eligibility for statin use in low-risk countries, a new study has found.
The authors reported that and essentially eliminate a statin indication in women.
“We have guidelines in place to try to prevent cardiovascular disease but the risk threshold in this new guideline means that almost nobody qualifies for treatment in many countries, which will lead to almost no prevention of future cardiovascular disease in those countries,” lead author Martin Bødtker Mortensen, MD, PhD, Aarhus (Denmark) University Hospital, commented in an interview.
“We argue that the risk thresholds need to be lowered to get the statin eligibility in European countries to be in line with thresholds in the U.K. and U.S., which are based on randomized, controlled trials,” he added.
The study was published online in JAMA Cardiology.
An accompanying editorial describes the results of the study as “alarming,” and, if confirmed, said the guidelines should be revisited to “prevent a step backwards in the use of statins in primary prevention.”
For the study, Dr. Mortensen and colleagues set out to compare the clinical performance of the new European prevention guidelines with American College of Cardiology/American Heart Association, United Kingdom–National Institute for Health and Care Excellence, and the 2019 European guidelines in a contemporary European cohort of 66,909 apparently healthy individuals from the Copenhagen General Population Study.
During the 9-year follow-up, a range of 2,962-4,277 nonfatal and fatal cardiovascular events was observed, as defined by the models in the various guidelines.
Results showed that although the new 2021 European guidelines introduced a new and improved risk model, known as SCORE2, the updated age-specific recommendations dramatically reduced eligibility for a class I recommendation for statin therapy to only 4% of individuals, aged 40-69 years, and less than 1% of women.
This is in sharp contrast to the previous 2019 European guidelines as well as current UK-NICE and US-ACC/AHA guidelines that provide class I/strong recommendations to 20%, 26%, and 34% of individuals, respectively, with better clinical performance in both men and women, the authors report.
The researchers also reported other analyses in which the sensitivity of the new European guidelines was improved considerably by lowering the treatment thresholds.
Dr. Mortensen explained to this news organization that the original SCORE risk model used in ESC guidelines was problematic as it only predicts the 10-year risk of fatal atherosclerotic cardiovascular events, whereas those from the United States and United Kingdom used both fatal and nonfatal cardiovascular events.
“Now the ESC has updated its model and the new model is much better in that it predicts both fatal and nonfatal events, and the predicted risk correlates well with the actual risk. So that’s a big step forward. However, the new thresholds for statin treatment are far too high for low-risk European countries because very few individuals will now qualify for statin therapy,” he said.
“The problem is that, if we use these guidelines, the vast majority of those individuals who will develop cardiovascular disease within 10 years will not be assigned statin therapy that can reduce this risk. There will be lots of individuals who are at high risk of cardiovascular disease, but these guidelines will not identify them as needing to take a statin,” Dr. Mortensen commented.
“If we use the U.K. or U.S. guidelines, far more people in these low-risk European countries would be eligible for statin therapy and we would prevent far more events than if we use the new ESC guidelines,” he added.
Dr. Mortensen explained that the problem arises from having four different risk score models in Europe for areas at different risk, but they all use the same risk thresholds for statin treatment.
“In general, Eastern European countries have higher risk than Western European countries, so these guidelines may work quite well in Eastern European countries but in low-risk Western European countries, where the low-risk score model is used, very few people will qualify for statin therapy,” he said.
While Dr. Mortensen is not against the idea of different risk models in areas that have different risks, he says this needs to be accompanied by different risk thresholds in the different risk areas.
Asked whether there is an argument that most individuals in low-risk countries may not need to take a statin, Dr. Mortensen countered: “One of the reasons the risk is low in many of these European countries is the high use of preventative medication. So, if a threshold that is too high is used most people will not take a statin anymore and the risk in these countries will increase again.”
Authors of the accompanying editorial, Ann Marie Navar, MD, PhD, University of Texas Southwestern Medical Center, Dallas; Gregg C. Fonarow, MD, University of California, Los Angeles; and Michael J. Pencina, PhD, Duke University Medical Center, Durham, N.C., agreed with Dr. Mortensen that the problems appear to arise from use of a risk score that is highly influenced by regional cardiovascular burden.
They point out that under the current guidelines, a 55-year-old woman (smoker; systolic blood pressure, 130 mm Hg; non–HDL cholesterol, 4.0 mmol/L) would have a 10-year predicted risk of having a cardiovascular event of 5% in Denmark but a predicted risk of 18% in Romania.
“While there may be regional differences in environmental risk factors, location alone should not cause a fourfold difference in an individual’s predicted cardiovascular risk,” they wrote.
The editorialists also elaborated on Dr. Mortensen’s point that the new guideline creates a system that eventually becomes a victim of its own success.
“As countries are successful in implementing statin therapy to lower CVD, CVD rates drop, and progressively fewer individuals are then eligible for the very therapy that contributed to the decline in CVD in the first place,” they noted.
The editorialists called for the analysis to be replicated in other low-risk countries and extended to higher-risk regions, with a focus on potential overtreatment of men and older adults.
“If confirmed, the present findings should be a catalyst for the ESC to revisit or augment their current guidelines to prevent a step backward in the use of statins in primary prevention,” they concluded.
This news organization asked the ESC for a response to the findings, but did not comment by press time.
This work was supported by the Lundbeck Foundation, Herlev and Gentofte Hospital, Copenhagen University Hospital, the Copenhagen County Foundation, and Aarhus University, Denmark. Dr. Mortensen reported no disclosures.
A version of this article first appeared on Medscape.com.
New risk thresholds used to guide statin therapy for primary prevention of atherosclerotic cardiovascular disease in the latest European guidelines dramatically reduce eligibility for statin use in low-risk countries, a new study has found.
The authors reported that and essentially eliminate a statin indication in women.
“We have guidelines in place to try to prevent cardiovascular disease but the risk threshold in this new guideline means that almost nobody qualifies for treatment in many countries, which will lead to almost no prevention of future cardiovascular disease in those countries,” lead author Martin Bødtker Mortensen, MD, PhD, Aarhus (Denmark) University Hospital, commented in an interview.
“We argue that the risk thresholds need to be lowered to get the statin eligibility in European countries to be in line with thresholds in the U.K. and U.S., which are based on randomized, controlled trials,” he added.
The study was published online in JAMA Cardiology.
An accompanying editorial describes the results of the study as “alarming,” and, if confirmed, said the guidelines should be revisited to “prevent a step backwards in the use of statins in primary prevention.”
For the study, Dr. Mortensen and colleagues set out to compare the clinical performance of the new European prevention guidelines with American College of Cardiology/American Heart Association, United Kingdom–National Institute for Health and Care Excellence, and the 2019 European guidelines in a contemporary European cohort of 66,909 apparently healthy individuals from the Copenhagen General Population Study.
During the 9-year follow-up, a range of 2,962-4,277 nonfatal and fatal cardiovascular events was observed, as defined by the models in the various guidelines.
Results showed that although the new 2021 European guidelines introduced a new and improved risk model, known as SCORE2, the updated age-specific recommendations dramatically reduced eligibility for a class I recommendation for statin therapy to only 4% of individuals, aged 40-69 years, and less than 1% of women.
This is in sharp contrast to the previous 2019 European guidelines as well as current UK-NICE and US-ACC/AHA guidelines that provide class I/strong recommendations to 20%, 26%, and 34% of individuals, respectively, with better clinical performance in both men and women, the authors report.
The researchers also reported other analyses in which the sensitivity of the new European guidelines was improved considerably by lowering the treatment thresholds.
Dr. Mortensen explained to this news organization that the original SCORE risk model used in ESC guidelines was problematic as it only predicts the 10-year risk of fatal atherosclerotic cardiovascular events, whereas those from the United States and United Kingdom used both fatal and nonfatal cardiovascular events.
“Now the ESC has updated its model and the new model is much better in that it predicts both fatal and nonfatal events, and the predicted risk correlates well with the actual risk. So that’s a big step forward. However, the new thresholds for statin treatment are far too high for low-risk European countries because very few individuals will now qualify for statin therapy,” he said.
“The problem is that, if we use these guidelines, the vast majority of those individuals who will develop cardiovascular disease within 10 years will not be assigned statin therapy that can reduce this risk. There will be lots of individuals who are at high risk of cardiovascular disease, but these guidelines will not identify them as needing to take a statin,” Dr. Mortensen commented.
“If we use the U.K. or U.S. guidelines, far more people in these low-risk European countries would be eligible for statin therapy and we would prevent far more events than if we use the new ESC guidelines,” he added.
Dr. Mortensen explained that the problem arises from having four different risk score models in Europe for areas at different risk, but they all use the same risk thresholds for statin treatment.
“In general, Eastern European countries have higher risk than Western European countries, so these guidelines may work quite well in Eastern European countries but in low-risk Western European countries, where the low-risk score model is used, very few people will qualify for statin therapy,” he said.
While Dr. Mortensen is not against the idea of different risk models in areas that have different risks, he says this needs to be accompanied by different risk thresholds in the different risk areas.
Asked whether there is an argument that most individuals in low-risk countries may not need to take a statin, Dr. Mortensen countered: “One of the reasons the risk is low in many of these European countries is the high use of preventative medication. So, if a threshold that is too high is used most people will not take a statin anymore and the risk in these countries will increase again.”
Authors of the accompanying editorial, Ann Marie Navar, MD, PhD, University of Texas Southwestern Medical Center, Dallas; Gregg C. Fonarow, MD, University of California, Los Angeles; and Michael J. Pencina, PhD, Duke University Medical Center, Durham, N.C., agreed with Dr. Mortensen that the problems appear to arise from use of a risk score that is highly influenced by regional cardiovascular burden.
They point out that under the current guidelines, a 55-year-old woman (smoker; systolic blood pressure, 130 mm Hg; non–HDL cholesterol, 4.0 mmol/L) would have a 10-year predicted risk of having a cardiovascular event of 5% in Denmark but a predicted risk of 18% in Romania.
“While there may be regional differences in environmental risk factors, location alone should not cause a fourfold difference in an individual’s predicted cardiovascular risk,” they wrote.
The editorialists also elaborated on Dr. Mortensen’s point that the new guideline creates a system that eventually becomes a victim of its own success.
“As countries are successful in implementing statin therapy to lower CVD, CVD rates drop, and progressively fewer individuals are then eligible for the very therapy that contributed to the decline in CVD in the first place,” they noted.
The editorialists called for the analysis to be replicated in other low-risk countries and extended to higher-risk regions, with a focus on potential overtreatment of men and older adults.
“If confirmed, the present findings should be a catalyst for the ESC to revisit or augment their current guidelines to prevent a step backward in the use of statins in primary prevention,” they concluded.
This news organization asked the ESC for a response to the findings, but did not comment by press time.
This work was supported by the Lundbeck Foundation, Herlev and Gentofte Hospital, Copenhagen University Hospital, the Copenhagen County Foundation, and Aarhus University, Denmark. Dr. Mortensen reported no disclosures.
A version of this article first appeared on Medscape.com.
New risk thresholds used to guide statin therapy for primary prevention of atherosclerotic cardiovascular disease in the latest European guidelines dramatically reduce eligibility for statin use in low-risk countries, a new study has found.
The authors reported that and essentially eliminate a statin indication in women.
“We have guidelines in place to try to prevent cardiovascular disease but the risk threshold in this new guideline means that almost nobody qualifies for treatment in many countries, which will lead to almost no prevention of future cardiovascular disease in those countries,” lead author Martin Bødtker Mortensen, MD, PhD, Aarhus (Denmark) University Hospital, commented in an interview.
“We argue that the risk thresholds need to be lowered to get the statin eligibility in European countries to be in line with thresholds in the U.K. and U.S., which are based on randomized, controlled trials,” he added.
The study was published online in JAMA Cardiology.
An accompanying editorial describes the results of the study as “alarming,” and, if confirmed, said the guidelines should be revisited to “prevent a step backwards in the use of statins in primary prevention.”
For the study, Dr. Mortensen and colleagues set out to compare the clinical performance of the new European prevention guidelines with American College of Cardiology/American Heart Association, United Kingdom–National Institute for Health and Care Excellence, and the 2019 European guidelines in a contemporary European cohort of 66,909 apparently healthy individuals from the Copenhagen General Population Study.
During the 9-year follow-up, a range of 2,962-4,277 nonfatal and fatal cardiovascular events was observed, as defined by the models in the various guidelines.
Results showed that although the new 2021 European guidelines introduced a new and improved risk model, known as SCORE2, the updated age-specific recommendations dramatically reduced eligibility for a class I recommendation for statin therapy to only 4% of individuals, aged 40-69 years, and less than 1% of women.
This is in sharp contrast to the previous 2019 European guidelines as well as current UK-NICE and US-ACC/AHA guidelines that provide class I/strong recommendations to 20%, 26%, and 34% of individuals, respectively, with better clinical performance in both men and women, the authors report.
The researchers also reported other analyses in which the sensitivity of the new European guidelines was improved considerably by lowering the treatment thresholds.
Dr. Mortensen explained to this news organization that the original SCORE risk model used in ESC guidelines was problematic as it only predicts the 10-year risk of fatal atherosclerotic cardiovascular events, whereas those from the United States and United Kingdom used both fatal and nonfatal cardiovascular events.
“Now the ESC has updated its model and the new model is much better in that it predicts both fatal and nonfatal events, and the predicted risk correlates well with the actual risk. So that’s a big step forward. However, the new thresholds for statin treatment are far too high for low-risk European countries because very few individuals will now qualify for statin therapy,” he said.
“The problem is that, if we use these guidelines, the vast majority of those individuals who will develop cardiovascular disease within 10 years will not be assigned statin therapy that can reduce this risk. There will be lots of individuals who are at high risk of cardiovascular disease, but these guidelines will not identify them as needing to take a statin,” Dr. Mortensen commented.
“If we use the U.K. or U.S. guidelines, far more people in these low-risk European countries would be eligible for statin therapy and we would prevent far more events than if we use the new ESC guidelines,” he added.
Dr. Mortensen explained that the problem arises from having four different risk score models in Europe for areas at different risk, but they all use the same risk thresholds for statin treatment.
“In general, Eastern European countries have higher risk than Western European countries, so these guidelines may work quite well in Eastern European countries but in low-risk Western European countries, where the low-risk score model is used, very few people will qualify for statin therapy,” he said.
While Dr. Mortensen is not against the idea of different risk models in areas that have different risks, he says this needs to be accompanied by different risk thresholds in the different risk areas.
Asked whether there is an argument that most individuals in low-risk countries may not need to take a statin, Dr. Mortensen countered: “One of the reasons the risk is low in many of these European countries is the high use of preventative medication. So, if a threshold that is too high is used most people will not take a statin anymore and the risk in these countries will increase again.”
Authors of the accompanying editorial, Ann Marie Navar, MD, PhD, University of Texas Southwestern Medical Center, Dallas; Gregg C. Fonarow, MD, University of California, Los Angeles; and Michael J. Pencina, PhD, Duke University Medical Center, Durham, N.C., agreed with Dr. Mortensen that the problems appear to arise from use of a risk score that is highly influenced by regional cardiovascular burden.
They point out that under the current guidelines, a 55-year-old woman (smoker; systolic blood pressure, 130 mm Hg; non–HDL cholesterol, 4.0 mmol/L) would have a 10-year predicted risk of having a cardiovascular event of 5% in Denmark but a predicted risk of 18% in Romania.
“While there may be regional differences in environmental risk factors, location alone should not cause a fourfold difference in an individual’s predicted cardiovascular risk,” they wrote.
The editorialists also elaborated on Dr. Mortensen’s point that the new guideline creates a system that eventually becomes a victim of its own success.
“As countries are successful in implementing statin therapy to lower CVD, CVD rates drop, and progressively fewer individuals are then eligible for the very therapy that contributed to the decline in CVD in the first place,” they noted.
The editorialists called for the analysis to be replicated in other low-risk countries and extended to higher-risk regions, with a focus on potential overtreatment of men and older adults.
“If confirmed, the present findings should be a catalyst for the ESC to revisit or augment their current guidelines to prevent a step backward in the use of statins in primary prevention,” they concluded.
This news organization asked the ESC for a response to the findings, but did not comment by press time.
This work was supported by the Lundbeck Foundation, Herlev and Gentofte Hospital, Copenhagen University Hospital, the Copenhagen County Foundation, and Aarhus University, Denmark. Dr. Mortensen reported no disclosures.
A version of this article first appeared on Medscape.com.
FROM JAMA CARDIOLOGY
California will make low-cost insulin, Gov. Newsom says
On July 7, he said he had just signed a state budget that includes $50 million for development of the insulin and another $50 million for a place to make it.
“Nothing, nothing epitomizes market failures more than the cost of insulin,” Gov. Newsom said in a video posted on the governor’s Twitter page. He noted that many Americans have out-of-pocket costs ranging from $300 to $500 per month for insulin, which is used to treat diabetes.
“In California, we know people should not go into debt to receive lifesaving medication,” he said.
Gov. Newsom said that when he first took office, he signed an executive order to launch California’s own prescription drug system and that the insulin initiative is the first step toward making that happen.
People who take insulin have long complained about its high price. A November 2021 report from The Lancet said 25% of the insulin patients in the United States struggle to pay for it.
The cost of insulin for patients with insurance ranges from $334 to $1,000 a month, ABC News said, citing the Kaiser Family Foundation.
Legislation in Congress would bring down the cost of insulin if passed, with one bill capping costs at $35 per month for patients with health insurance. But The Hill reported that some Republicans oppose the legislation because it would interfere with free markets and raise costs for drug companies.
The CDC says 37.3 million people in the United States – about 11.3% of the population – have diabetes, with 8.5 million of them undiagnosed.
A version of this article first appeared on WebMD.com.
On July 7, he said he had just signed a state budget that includes $50 million for development of the insulin and another $50 million for a place to make it.
“Nothing, nothing epitomizes market failures more than the cost of insulin,” Gov. Newsom said in a video posted on the governor’s Twitter page. He noted that many Americans have out-of-pocket costs ranging from $300 to $500 per month for insulin, which is used to treat diabetes.
“In California, we know people should not go into debt to receive lifesaving medication,” he said.
Gov. Newsom said that when he first took office, he signed an executive order to launch California’s own prescription drug system and that the insulin initiative is the first step toward making that happen.
People who take insulin have long complained about its high price. A November 2021 report from The Lancet said 25% of the insulin patients in the United States struggle to pay for it.
The cost of insulin for patients with insurance ranges from $334 to $1,000 a month, ABC News said, citing the Kaiser Family Foundation.
Legislation in Congress would bring down the cost of insulin if passed, with one bill capping costs at $35 per month for patients with health insurance. But The Hill reported that some Republicans oppose the legislation because it would interfere with free markets and raise costs for drug companies.
The CDC says 37.3 million people in the United States – about 11.3% of the population – have diabetes, with 8.5 million of them undiagnosed.
A version of this article first appeared on WebMD.com.
On July 7, he said he had just signed a state budget that includes $50 million for development of the insulin and another $50 million for a place to make it.
“Nothing, nothing epitomizes market failures more than the cost of insulin,” Gov. Newsom said in a video posted on the governor’s Twitter page. He noted that many Americans have out-of-pocket costs ranging from $300 to $500 per month for insulin, which is used to treat diabetes.
“In California, we know people should not go into debt to receive lifesaving medication,” he said.
Gov. Newsom said that when he first took office, he signed an executive order to launch California’s own prescription drug system and that the insulin initiative is the first step toward making that happen.
People who take insulin have long complained about its high price. A November 2021 report from The Lancet said 25% of the insulin patients in the United States struggle to pay for it.
The cost of insulin for patients with insurance ranges from $334 to $1,000 a month, ABC News said, citing the Kaiser Family Foundation.
Legislation in Congress would bring down the cost of insulin if passed, with one bill capping costs at $35 per month for patients with health insurance. But The Hill reported that some Republicans oppose the legislation because it would interfere with free markets and raise costs for drug companies.
The CDC says 37.3 million people in the United States – about 11.3% of the population – have diabetes, with 8.5 million of them undiagnosed.
A version of this article first appeared on WebMD.com.