User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Sugary drinks, rather than artificially sweetened beverages or juices, show link to IBD
Drinks sweetened with sugar – but not natural juices or drinks sweetened artificially – were linked to a higher risk of inflammatory bowel disease (IBD) in people who drank more than one a day in a study of more than 120,000 people.
IBD has previously been linked with high consumption of sugar, but population-based evidence has been inconclusive, and natural juices have not been studied, write lead author Tian Fu, with the department of gastroenterology, the Third Xiangya Hospital of Central South University in Changsha, China, and colleagues.
Their study compared the associations of sugar-sweetened drinks, artificially sweetened beverages, and natural juices (including pure fruit or vegetable juices) with IBD risk.
“As one of the major sources of free sugar, beverages have been related to inflammation-related health outcomes but received less attention in the field of IBD,” the authors wrote.
, especially Crohn’s disease (CD), but further studies are needed to confirm these findings and explore the underlying mechanism,” they write.
The study was published online in Alimentary Pharmacology and Therapeutics.
Link significant for Crohn’s disease but not ulcerative colitis
The researchers used data from 121,490 participants in the UK Biobank who did not have IBD at trial recruitment in 2006-2010. The average age of the participants was 56 years, and almost all (96.9%) were White. The researchers studied their intake of beverages with 24-hour diet recalls from 2009 to 2012.
Participants were sorted into three groups according to the consumption of each beverage: 0 unit (glasses, cans, or 250 mL/cartons) per day (reference group), 0-1 unit per day, and more than 1 unit per day.
While most (66.3%) did not drink any sugar-sweetened beverages, participants who reported drinking more than 1 unit per day were more likely to have a higher body mass index and consume higher amounts of total energy and sugar.
During an average follow-up of about 10 years, the investigators documented 510 incident IBD cases: 143 cases of CD and 367 cases of ulcerative colitis (UC).
Compared to people who did not drink sugar-sweetened beverages, those who drank at least 1 unit per day had a significantly higher IBD risk (hazard ratio, 1.51; 95% confidence interval, 1.11-2.05), but the trend was statistically nonsignificant.
The association was significant for CD (HR, 2.05; 95% CI, 1.22,3.46) but not for UC (HR, 1.31; 95% CI, 0.89-1.92). The positive association between sugar-sweetened drinks and risk of CD, but not UC, was in line with previous studies showing that dietary patterns were more associated with CD risk, the authors noted.
They also highlighted that there was no positive link between artificially sweetened beverages, natural juices, or total sugar intake and IBD risk. They noted that the inflammatory role of artificial sweeteners is still being debated.
Additionally, the effect of natural sugar in juices may be counteracted by fiber and bioactive compounds in the juices, the authors wrote.
A limitation of the study is that at baseline, all participants in the UK Biobank were older than 40 years, so the researchers could not examine any links with younger-onset IBD.
Additionally, the self-reported questionnaires are subject to recall bias, though the survey has been validated.
Study adds to previous evidence
Hasan Zaki, PhD, an assistant professor with the department of pathology at University of Texas Southwestern Medical Center, Dallas, said in an interview that the size of this population-based study adds evidence that simple sugar can increase the risk for IBD. Dr. Zaki studies the relationship of inflammatory disorders and diet and was not involved in the study.
“This study is very strong evidence of the association between high sugar and IBD,” he said. He noted that more studies are needed because there are few studies in this area and results have varied.
His lab conducted work on the subject previously in mice. In a 2020 study, they found that a high-sugar diet helped promote IBD development and gut microbiota dysfunction.
Dr. Zaki pointed out that, among people in the United Kingdom and those in the United States, diets, demographics, and IBD incidence are similar, a fact that may make the findings more generalizable.
However, studies comparing the categories of sweetened drinks should be conducted in a U.S. population to assess the results in a diverse group to see whether ethnicity plays a role, because almost all of the people in the UK group were White, he said.
Also important, Dr. Zaki said, will be follow-up studies of the link between sweet drinks and IBD in U.S. children, among whom consumption is particularly high and the IBD incidence is rising. One study showed the prevalence increased 133% from 2007 to 2016.
The results of this study should help gastroenterologists counsel patients on an ideal diet to avoid IBD or reduce IBD severity, he said.
The study was supported by the National Natural Science Foundation of China and Key Project of Research and Development Plan of Hunan Province. The study authors and Dr. Zaki report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Drinks sweetened with sugar – but not natural juices or drinks sweetened artificially – were linked to a higher risk of inflammatory bowel disease (IBD) in people who drank more than one a day in a study of more than 120,000 people.
IBD has previously been linked with high consumption of sugar, but population-based evidence has been inconclusive, and natural juices have not been studied, write lead author Tian Fu, with the department of gastroenterology, the Third Xiangya Hospital of Central South University in Changsha, China, and colleagues.
Their study compared the associations of sugar-sweetened drinks, artificially sweetened beverages, and natural juices (including pure fruit or vegetable juices) with IBD risk.
“As one of the major sources of free sugar, beverages have been related to inflammation-related health outcomes but received less attention in the field of IBD,” the authors wrote.
, especially Crohn’s disease (CD), but further studies are needed to confirm these findings and explore the underlying mechanism,” they write.
The study was published online in Alimentary Pharmacology and Therapeutics.
Link significant for Crohn’s disease but not ulcerative colitis
The researchers used data from 121,490 participants in the UK Biobank who did not have IBD at trial recruitment in 2006-2010. The average age of the participants was 56 years, and almost all (96.9%) were White. The researchers studied their intake of beverages with 24-hour diet recalls from 2009 to 2012.
Participants were sorted into three groups according to the consumption of each beverage: 0 unit (glasses, cans, or 250 mL/cartons) per day (reference group), 0-1 unit per day, and more than 1 unit per day.
While most (66.3%) did not drink any sugar-sweetened beverages, participants who reported drinking more than 1 unit per day were more likely to have a higher body mass index and consume higher amounts of total energy and sugar.
During an average follow-up of about 10 years, the investigators documented 510 incident IBD cases: 143 cases of CD and 367 cases of ulcerative colitis (UC).
Compared to people who did not drink sugar-sweetened beverages, those who drank at least 1 unit per day had a significantly higher IBD risk (hazard ratio, 1.51; 95% confidence interval, 1.11-2.05), but the trend was statistically nonsignificant.
The association was significant for CD (HR, 2.05; 95% CI, 1.22,3.46) but not for UC (HR, 1.31; 95% CI, 0.89-1.92). The positive association between sugar-sweetened drinks and risk of CD, but not UC, was in line with previous studies showing that dietary patterns were more associated with CD risk, the authors noted.
They also highlighted that there was no positive link between artificially sweetened beverages, natural juices, or total sugar intake and IBD risk. They noted that the inflammatory role of artificial sweeteners is still being debated.
Additionally, the effect of natural sugar in juices may be counteracted by fiber and bioactive compounds in the juices, the authors wrote.
A limitation of the study is that at baseline, all participants in the UK Biobank were older than 40 years, so the researchers could not examine any links with younger-onset IBD.
Additionally, the self-reported questionnaires are subject to recall bias, though the survey has been validated.
Study adds to previous evidence
Hasan Zaki, PhD, an assistant professor with the department of pathology at University of Texas Southwestern Medical Center, Dallas, said in an interview that the size of this population-based study adds evidence that simple sugar can increase the risk for IBD. Dr. Zaki studies the relationship of inflammatory disorders and diet and was not involved in the study.
“This study is very strong evidence of the association between high sugar and IBD,” he said. He noted that more studies are needed because there are few studies in this area and results have varied.
His lab conducted work on the subject previously in mice. In a 2020 study, they found that a high-sugar diet helped promote IBD development and gut microbiota dysfunction.
Dr. Zaki pointed out that, among people in the United Kingdom and those in the United States, diets, demographics, and IBD incidence are similar, a fact that may make the findings more generalizable.
However, studies comparing the categories of sweetened drinks should be conducted in a U.S. population to assess the results in a diverse group to see whether ethnicity plays a role, because almost all of the people in the UK group were White, he said.
Also important, Dr. Zaki said, will be follow-up studies of the link between sweet drinks and IBD in U.S. children, among whom consumption is particularly high and the IBD incidence is rising. One study showed the prevalence increased 133% from 2007 to 2016.
The results of this study should help gastroenterologists counsel patients on an ideal diet to avoid IBD or reduce IBD severity, he said.
The study was supported by the National Natural Science Foundation of China and Key Project of Research and Development Plan of Hunan Province. The study authors and Dr. Zaki report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Drinks sweetened with sugar – but not natural juices or drinks sweetened artificially – were linked to a higher risk of inflammatory bowel disease (IBD) in people who drank more than one a day in a study of more than 120,000 people.
IBD has previously been linked with high consumption of sugar, but population-based evidence has been inconclusive, and natural juices have not been studied, write lead author Tian Fu, with the department of gastroenterology, the Third Xiangya Hospital of Central South University in Changsha, China, and colleagues.
Their study compared the associations of sugar-sweetened drinks, artificially sweetened beverages, and natural juices (including pure fruit or vegetable juices) with IBD risk.
“As one of the major sources of free sugar, beverages have been related to inflammation-related health outcomes but received less attention in the field of IBD,” the authors wrote.
, especially Crohn’s disease (CD), but further studies are needed to confirm these findings and explore the underlying mechanism,” they write.
The study was published online in Alimentary Pharmacology and Therapeutics.
Link significant for Crohn’s disease but not ulcerative colitis
The researchers used data from 121,490 participants in the UK Biobank who did not have IBD at trial recruitment in 2006-2010. The average age of the participants was 56 years, and almost all (96.9%) were White. The researchers studied their intake of beverages with 24-hour diet recalls from 2009 to 2012.
Participants were sorted into three groups according to the consumption of each beverage: 0 unit (glasses, cans, or 250 mL/cartons) per day (reference group), 0-1 unit per day, and more than 1 unit per day.
While most (66.3%) did not drink any sugar-sweetened beverages, participants who reported drinking more than 1 unit per day were more likely to have a higher body mass index and consume higher amounts of total energy and sugar.
During an average follow-up of about 10 years, the investigators documented 510 incident IBD cases: 143 cases of CD and 367 cases of ulcerative colitis (UC).
Compared to people who did not drink sugar-sweetened beverages, those who drank at least 1 unit per day had a significantly higher IBD risk (hazard ratio, 1.51; 95% confidence interval, 1.11-2.05), but the trend was statistically nonsignificant.
The association was significant for CD (HR, 2.05; 95% CI, 1.22,3.46) but not for UC (HR, 1.31; 95% CI, 0.89-1.92). The positive association between sugar-sweetened drinks and risk of CD, but not UC, was in line with previous studies showing that dietary patterns were more associated with CD risk, the authors noted.
They also highlighted that there was no positive link between artificially sweetened beverages, natural juices, or total sugar intake and IBD risk. They noted that the inflammatory role of artificial sweeteners is still being debated.
Additionally, the effect of natural sugar in juices may be counteracted by fiber and bioactive compounds in the juices, the authors wrote.
A limitation of the study is that at baseline, all participants in the UK Biobank were older than 40 years, so the researchers could not examine any links with younger-onset IBD.
Additionally, the self-reported questionnaires are subject to recall bias, though the survey has been validated.
Study adds to previous evidence
Hasan Zaki, PhD, an assistant professor with the department of pathology at University of Texas Southwestern Medical Center, Dallas, said in an interview that the size of this population-based study adds evidence that simple sugar can increase the risk for IBD. Dr. Zaki studies the relationship of inflammatory disorders and diet and was not involved in the study.
“This study is very strong evidence of the association between high sugar and IBD,” he said. He noted that more studies are needed because there are few studies in this area and results have varied.
His lab conducted work on the subject previously in mice. In a 2020 study, they found that a high-sugar diet helped promote IBD development and gut microbiota dysfunction.
Dr. Zaki pointed out that, among people in the United Kingdom and those in the United States, diets, demographics, and IBD incidence are similar, a fact that may make the findings more generalizable.
However, studies comparing the categories of sweetened drinks should be conducted in a U.S. population to assess the results in a diverse group to see whether ethnicity plays a role, because almost all of the people in the UK group were White, he said.
Also important, Dr. Zaki said, will be follow-up studies of the link between sweet drinks and IBD in U.S. children, among whom consumption is particularly high and the IBD incidence is rising. One study showed the prevalence increased 133% from 2007 to 2016.
The results of this study should help gastroenterologists counsel patients on an ideal diet to avoid IBD or reduce IBD severity, he said.
The study was supported by the National Natural Science Foundation of China and Key Project of Research and Development Plan of Hunan Province. The study authors and Dr. Zaki report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ALIMENTARY PHARMACOLOGY AND THERAPEUTICS
VA foster program helps older vets manage COVID challenges
Susan Snead used to live in an apartment complex for older adults. The complex had a nice dayroom, and neighbors would knock on her door every now and then to check in.
But despite not being lonely, Ms. Snead, 89, did live alone in downtown Charleston, S.C. Eventually, that became dangerous.
“I fell a few times,” she says. “I had to call somebody to come and get me up.”
Sometimes help would come from the apartment complex’s office. Sometimes it came with a police escort.
Over time, needing to make those calls became a burden. Making and keeping appointments with her doctor, something she had to do regularly, as she has diabetes, got harder, too.
“It kind of wore me out,” she says. “Like you’re going up a hill.”
As she was beginning to accept she could no longer live alone, Ms. Snead, an Air Force veteran, learned about a program run by the Department of Veterans Affairs called Medical Foster Home.
Caregivers help aging veterans with activities of daily living like bathing, cooking, making and getting to appointments, getting dressed, and taking daily medication.
Caregivers can take care of up to three residents in their home at a time. While most residents are veterans, caregivers sometimes care for non-veteran residents, such as a veteran’s spouse or a caregiver’s family member.
Veterans typically pay about $1,500 to $3,000 out-of-pocket per month for the service, depending on location.
According to the VA, the concept of medical foster homes has been around since 1999, when VA hospitals across the country began reaching out to people willing to provide live-in care for veterans. The option is led by local VA hospitals, which approve caregivers and provide administrative services. There are now 517 medical foster homes, the VA says.
Much like other residential care facilities, medical foster homes get regular inspections for safety, nutrition, and more.
In 2019, Ms. Snead signed up for the program. She expected to be cared for, but she found a sense of family with her caregiver, Wilhelmina Brown, and another veteran in the home.
Ms. Brown started taking care of people – but not necessarily veterans – in 1997 when her grandmother was unable to care for herself, she says.
“My grandmama carried me to church every Sunday, she carried me to the beach – everywhere she went, she took me with her,” Ms. Brown says. As her grandmother got older, “I said, ‘I’m going to take care of her in my home.’ ”
Caring for others must come from the heart, Ms. Brown says.
She cooks her residents’ meals three times a day with dietary restrictions in mind, washes their dishes, does their laundry, remembers birthdays, and plans little parties.
“That’s my family,” Ms. Brown says.
In 2020, the COVID-19 pandemic upended the world – but at the same time, it highlighted the advantages of the medical foster home model.
Home-based primary care keeps veterans out of nursing homes – something that became particularly important as COVID-19 hit nursing homes and long-term care facilities.
Caregivers in the system were also able to help veterans, often living in rural areas, pivot and adapt to telehealth during a time of crisis.
One study, published in the journal Geriatrics, set out to identify how medical foster homes were able to deliver safe, effective health care during the early stages of the pandemic.
Researchers interviewed 37 VA care providers at 16 rural medical foster home programs across the country. The interviews took place between December 2020 and February 2021. They found medical foster home caregivers, coordinators, and health care providers communicated to move office visits to the home, helped veterans navigate telehealth, advocated to get veterans vaccinated in-home, and relied on each other to fight social isolation.
Caregivers also adapted quickly to telehealth, according to Leah Haverhals, PhD, a health research scientist and communications director for the Seattle-Denver Center of Innovation for Veteran Centered and Value Driven Care, who led the study.
Most veterans in the foster home program are older and find new technology difficult to use.
Caregivers, coordinators, and health care providers were largely new to the technology, too.
While the study found that most veterans and caregivers preferred in-person care, they were able to work together to make the best of telehealth.
“That speaks to the nature of the care being given, being able to pivot in a crisis like that,” Dr. Haverhals says.
If caregivers didn’t already have computers or telehealth-compatible devices, the VA provided iPads that would connect to the internet using cellular signals. According to the study, this helped to overcome connectivity issues that may have caused problems in rural areas.
Ms. Snead says Ms. Brown helped a lot with her telehealth calls.
“If we had to do things over the phone or with video, she was able to set that up to work with the person on the other end. She knows a lot about that stuff – about computers and things like that,” Ms. Snead says, adding that she hadn’t worked with computers since retirement in 1998.
Telehealth helped health care providers identify infections and quickly prescribe antibiotics to veterans in rural areas and provide other care that was more safely delivered in private homes.
“The findings from our study highlighted that when working together for the common goal of keeping vulnerable populations like veterans in MFHs [medical foster homes] safe during times of crisis, adaptation and collaboration facilitated the ongoing provision of high-quality care,” Dr. Haverhals’s group wrote. “Such collaboration has been shown to be critical in recent research in the United States on supporting older adults during the pandemic.”
Cari Levy, MD, PhD, a professor at the University of Colorado at Denver, Aurora, and a co-author of the study, specializes in palliative and telenursing home care for the VA.
Dr. Levy, who has worked for the VA for about 20 years, says how medical foster homes provided care during the pandemic carries lessons for civilian clinics. One of the most important lessons, she says, is that medical professionals will need to provide more care where people are, especially in populations that are too sick to get to the clinic.
“For years, there was all this hope that telehealth would expand,” but it took a pandemic to authorize approval from federal agencies to explode, she says. “I shudder to think what would have happened if we didn’t have telehealth. Fortunately, it was the right time to be able to flip a switch.”
Crisis aside, Dr. Levy says her dream would be for health care providers to do more home-based care. The model allows people to preserve the relational aspects of medicine, which can counteract a lot of the moral injury and burnout in the field, she says, adding:
“I see this as the kind of medicine many people intended to do when they got into medicine.”
A version of this article first appeared on WebMD.com.
Susan Snead used to live in an apartment complex for older adults. The complex had a nice dayroom, and neighbors would knock on her door every now and then to check in.
But despite not being lonely, Ms. Snead, 89, did live alone in downtown Charleston, S.C. Eventually, that became dangerous.
“I fell a few times,” she says. “I had to call somebody to come and get me up.”
Sometimes help would come from the apartment complex’s office. Sometimes it came with a police escort.
Over time, needing to make those calls became a burden. Making and keeping appointments with her doctor, something she had to do regularly, as she has diabetes, got harder, too.
“It kind of wore me out,” she says. “Like you’re going up a hill.”
As she was beginning to accept she could no longer live alone, Ms. Snead, an Air Force veteran, learned about a program run by the Department of Veterans Affairs called Medical Foster Home.
Caregivers help aging veterans with activities of daily living like bathing, cooking, making and getting to appointments, getting dressed, and taking daily medication.
Caregivers can take care of up to three residents in their home at a time. While most residents are veterans, caregivers sometimes care for non-veteran residents, such as a veteran’s spouse or a caregiver’s family member.
Veterans typically pay about $1,500 to $3,000 out-of-pocket per month for the service, depending on location.
According to the VA, the concept of medical foster homes has been around since 1999, when VA hospitals across the country began reaching out to people willing to provide live-in care for veterans. The option is led by local VA hospitals, which approve caregivers and provide administrative services. There are now 517 medical foster homes, the VA says.
Much like other residential care facilities, medical foster homes get regular inspections for safety, nutrition, and more.
In 2019, Ms. Snead signed up for the program. She expected to be cared for, but she found a sense of family with her caregiver, Wilhelmina Brown, and another veteran in the home.
Ms. Brown started taking care of people – but not necessarily veterans – in 1997 when her grandmother was unable to care for herself, she says.
“My grandmama carried me to church every Sunday, she carried me to the beach – everywhere she went, she took me with her,” Ms. Brown says. As her grandmother got older, “I said, ‘I’m going to take care of her in my home.’ ”
Caring for others must come from the heart, Ms. Brown says.
She cooks her residents’ meals three times a day with dietary restrictions in mind, washes their dishes, does their laundry, remembers birthdays, and plans little parties.
“That’s my family,” Ms. Brown says.
In 2020, the COVID-19 pandemic upended the world – but at the same time, it highlighted the advantages of the medical foster home model.
Home-based primary care keeps veterans out of nursing homes – something that became particularly important as COVID-19 hit nursing homes and long-term care facilities.
Caregivers in the system were also able to help veterans, often living in rural areas, pivot and adapt to telehealth during a time of crisis.
One study, published in the journal Geriatrics, set out to identify how medical foster homes were able to deliver safe, effective health care during the early stages of the pandemic.
Researchers interviewed 37 VA care providers at 16 rural medical foster home programs across the country. The interviews took place between December 2020 and February 2021. They found medical foster home caregivers, coordinators, and health care providers communicated to move office visits to the home, helped veterans navigate telehealth, advocated to get veterans vaccinated in-home, and relied on each other to fight social isolation.
Caregivers also adapted quickly to telehealth, according to Leah Haverhals, PhD, a health research scientist and communications director for the Seattle-Denver Center of Innovation for Veteran Centered and Value Driven Care, who led the study.
Most veterans in the foster home program are older and find new technology difficult to use.
Caregivers, coordinators, and health care providers were largely new to the technology, too.
While the study found that most veterans and caregivers preferred in-person care, they were able to work together to make the best of telehealth.
“That speaks to the nature of the care being given, being able to pivot in a crisis like that,” Dr. Haverhals says.
If caregivers didn’t already have computers or telehealth-compatible devices, the VA provided iPads that would connect to the internet using cellular signals. According to the study, this helped to overcome connectivity issues that may have caused problems in rural areas.
Ms. Snead says Ms. Brown helped a lot with her telehealth calls.
“If we had to do things over the phone or with video, she was able to set that up to work with the person on the other end. She knows a lot about that stuff – about computers and things like that,” Ms. Snead says, adding that she hadn’t worked with computers since retirement in 1998.
Telehealth helped health care providers identify infections and quickly prescribe antibiotics to veterans in rural areas and provide other care that was more safely delivered in private homes.
“The findings from our study highlighted that when working together for the common goal of keeping vulnerable populations like veterans in MFHs [medical foster homes] safe during times of crisis, adaptation and collaboration facilitated the ongoing provision of high-quality care,” Dr. Haverhals’s group wrote. “Such collaboration has been shown to be critical in recent research in the United States on supporting older adults during the pandemic.”
Cari Levy, MD, PhD, a professor at the University of Colorado at Denver, Aurora, and a co-author of the study, specializes in palliative and telenursing home care for the VA.
Dr. Levy, who has worked for the VA for about 20 years, says how medical foster homes provided care during the pandemic carries lessons for civilian clinics. One of the most important lessons, she says, is that medical professionals will need to provide more care where people are, especially in populations that are too sick to get to the clinic.
“For years, there was all this hope that telehealth would expand,” but it took a pandemic to authorize approval from federal agencies to explode, she says. “I shudder to think what would have happened if we didn’t have telehealth. Fortunately, it was the right time to be able to flip a switch.”
Crisis aside, Dr. Levy says her dream would be for health care providers to do more home-based care. The model allows people to preserve the relational aspects of medicine, which can counteract a lot of the moral injury and burnout in the field, she says, adding:
“I see this as the kind of medicine many people intended to do when they got into medicine.”
A version of this article first appeared on WebMD.com.
Susan Snead used to live in an apartment complex for older adults. The complex had a nice dayroom, and neighbors would knock on her door every now and then to check in.
But despite not being lonely, Ms. Snead, 89, did live alone in downtown Charleston, S.C. Eventually, that became dangerous.
“I fell a few times,” she says. “I had to call somebody to come and get me up.”
Sometimes help would come from the apartment complex’s office. Sometimes it came with a police escort.
Over time, needing to make those calls became a burden. Making and keeping appointments with her doctor, something she had to do regularly, as she has diabetes, got harder, too.
“It kind of wore me out,” she says. “Like you’re going up a hill.”
As she was beginning to accept she could no longer live alone, Ms. Snead, an Air Force veteran, learned about a program run by the Department of Veterans Affairs called Medical Foster Home.
Caregivers help aging veterans with activities of daily living like bathing, cooking, making and getting to appointments, getting dressed, and taking daily medication.
Caregivers can take care of up to three residents in their home at a time. While most residents are veterans, caregivers sometimes care for non-veteran residents, such as a veteran’s spouse or a caregiver’s family member.
Veterans typically pay about $1,500 to $3,000 out-of-pocket per month for the service, depending on location.
According to the VA, the concept of medical foster homes has been around since 1999, when VA hospitals across the country began reaching out to people willing to provide live-in care for veterans. The option is led by local VA hospitals, which approve caregivers and provide administrative services. There are now 517 medical foster homes, the VA says.
Much like other residential care facilities, medical foster homes get regular inspections for safety, nutrition, and more.
In 2019, Ms. Snead signed up for the program. She expected to be cared for, but she found a sense of family with her caregiver, Wilhelmina Brown, and another veteran in the home.
Ms. Brown started taking care of people – but not necessarily veterans – in 1997 when her grandmother was unable to care for herself, she says.
“My grandmama carried me to church every Sunday, she carried me to the beach – everywhere she went, she took me with her,” Ms. Brown says. As her grandmother got older, “I said, ‘I’m going to take care of her in my home.’ ”
Caring for others must come from the heart, Ms. Brown says.
She cooks her residents’ meals three times a day with dietary restrictions in mind, washes their dishes, does their laundry, remembers birthdays, and plans little parties.
“That’s my family,” Ms. Brown says.
In 2020, the COVID-19 pandemic upended the world – but at the same time, it highlighted the advantages of the medical foster home model.
Home-based primary care keeps veterans out of nursing homes – something that became particularly important as COVID-19 hit nursing homes and long-term care facilities.
Caregivers in the system were also able to help veterans, often living in rural areas, pivot and adapt to telehealth during a time of crisis.
One study, published in the journal Geriatrics, set out to identify how medical foster homes were able to deliver safe, effective health care during the early stages of the pandemic.
Researchers interviewed 37 VA care providers at 16 rural medical foster home programs across the country. The interviews took place between December 2020 and February 2021. They found medical foster home caregivers, coordinators, and health care providers communicated to move office visits to the home, helped veterans navigate telehealth, advocated to get veterans vaccinated in-home, and relied on each other to fight social isolation.
Caregivers also adapted quickly to telehealth, according to Leah Haverhals, PhD, a health research scientist and communications director for the Seattle-Denver Center of Innovation for Veteran Centered and Value Driven Care, who led the study.
Most veterans in the foster home program are older and find new technology difficult to use.
Caregivers, coordinators, and health care providers were largely new to the technology, too.
While the study found that most veterans and caregivers preferred in-person care, they were able to work together to make the best of telehealth.
“That speaks to the nature of the care being given, being able to pivot in a crisis like that,” Dr. Haverhals says.
If caregivers didn’t already have computers or telehealth-compatible devices, the VA provided iPads that would connect to the internet using cellular signals. According to the study, this helped to overcome connectivity issues that may have caused problems in rural areas.
Ms. Snead says Ms. Brown helped a lot with her telehealth calls.
“If we had to do things over the phone or with video, she was able to set that up to work with the person on the other end. She knows a lot about that stuff – about computers and things like that,” Ms. Snead says, adding that she hadn’t worked with computers since retirement in 1998.
Telehealth helped health care providers identify infections and quickly prescribe antibiotics to veterans in rural areas and provide other care that was more safely delivered in private homes.
“The findings from our study highlighted that when working together for the common goal of keeping vulnerable populations like veterans in MFHs [medical foster homes] safe during times of crisis, adaptation and collaboration facilitated the ongoing provision of high-quality care,” Dr. Haverhals’s group wrote. “Such collaboration has been shown to be critical in recent research in the United States on supporting older adults during the pandemic.”
Cari Levy, MD, PhD, a professor at the University of Colorado at Denver, Aurora, and a co-author of the study, specializes in palliative and telenursing home care for the VA.
Dr. Levy, who has worked for the VA for about 20 years, says how medical foster homes provided care during the pandemic carries lessons for civilian clinics. One of the most important lessons, she says, is that medical professionals will need to provide more care where people are, especially in populations that are too sick to get to the clinic.
“For years, there was all this hope that telehealth would expand,” but it took a pandemic to authorize approval from federal agencies to explode, she says. “I shudder to think what would have happened if we didn’t have telehealth. Fortunately, it was the right time to be able to flip a switch.”
Crisis aside, Dr. Levy says her dream would be for health care providers to do more home-based care. The model allows people to preserve the relational aspects of medicine, which can counteract a lot of the moral injury and burnout in the field, she says, adding:
“I see this as the kind of medicine many people intended to do when they got into medicine.”
A version of this article first appeared on WebMD.com.
Exceeding exercise guidelines boosts survival, to a point
A new study suggests that going beyond current guidance on moderate and vigorous physical activity levels may add years to one’s life.
Americans are advised to do a minimum of 150-300 minutes a week of moderate exercise or 75-150 minutes a week of vigorous exercise, or an equivalent combination of both, according to U.S. Department of Health and Human Services Physical Activity Guidelines.
Results from more than 100,000 U.S. adults followed for 30 years showed that .
Adults who reported completing four times the minimum recommended activity levels saw no clear incremental mortality benefit but also no harm, according to the study, published in the journal Circulation.
“I think we’re worried more about the lower end and people that are not even doing the minimum, but this should be reassuring to people who like to do a lot of exercise,” senior author Edward Giovannucci, MD, ScD, with the Harvard T.H. Chan School of Public Health, Boston, told this news organization.
Some studies have suggested that long-term, high-intensity exercise (e.g., marathons, triathlons, and long-distance cycling) may be associated with increased risks of atrial fibrillation, coronary artery calcification, and sudden cardiac death.
A recent analysis from the Copenhagen City Heart Study showed a U-shaped association between long-term all-cause mortality and 0 to 2.5 hours and more than 10 hours of weekly, leisure-time sports activities.
Most studies suggesting harm, however, have used only one measurement of physical activity capturing a mix of people who chronically exercise at high levels and those who do it sporadically, which possibly can be harmful, Dr. Giovannucci said. “We were better able to look at consistent long-term activity and saw there was no harm.”
The study included 116,221 participants in the Nurses’ Health Study and the Health Professionals Follow-up Study between 1988 and 2018, who completed up to 15 (median, 11) questionnaires on their health and leisure-time physical activity that were updated every 2 years.
Most were White (96%), 63% were female, and the average age and body mass index over follow-up was 66 years and 26 kg/m2. During 30 years of follow-up, there were 47,596 deaths.
‘Any effort is worthwhile’
The analysis found that individuals who met the guideline for long-term vigorous physical activity (75-150 min/week) cut their adjusted risk of death from cardiovascular disease (CVD) by a whopping 31%, from non-CVD causes by 15%, and all-causes by 19%, compared with those with no long-term vigorous activity.
Those completing two to four times the recommended minimum (150-299 min/week) had a 27%-33% lower risk of CVD mortality, 19% lower risk of non-CVD mortality, and 21%-23% lower risk of all-cause mortality.
Higher levels did not appear to further lower mortality risk. For example, 300-374 min/week of vigorous physical activity was associated with a 32% lower risk of CVD death, 18% lower risk of non-CVD death, and 22% lower risk of dying from any cause.
The analysis also found that individuals who met the guidelines for moderate physical activity had lower CVD, non-CVD, and all-cause mortality risks whether they were active 150-244 min/week (22%, 19%, and 20%, respectively) or 225-299 min/week (21%, 25%, and 20%, respectively), compared with those with almost no long-term moderate activity.
Those fitting in two to four times the recommended minimum (300-599 min/week) had a 28%-38% lower risk of CVD mortality, 25%-27% lower risk of non-CVD mortality, and 26%-31% lower risk of all-cause mortality.
The mortality benefit appeared to plateau, with 600 min/week of moderate physical activity showing associations similar to 300-599 min/week.
“The sweet spot seems to be two to four times the recommended levels but for people who are sedentary, I think one of the key messages that I give my patients is that any effort is worthwhile; that any physical activity, even less than the recommended, has some mortality reduction,” Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins University, Baltimore, said in an interview.
Indeed, individuals who reported doing just 20-74 minutes of moderate exercise per week had a 19% lower risk of dying from any cause and a 13% lower risk of dying from CVD compared with those doing less.
Current American Heart Association (AHA) recommendations are for at least 150 minutes per week of moderate-intensity aerobic exercise or 75 minutes per week of vigorous aerobic exercise, or a combination of both.
“This suggests that even more is probably better, in the range of two to four times that, so maybe we should move our targets a little bit higher, which is kind of what the Department of Health and Human Services has already done,” said Dr. Michos, who was not involved in the study.
Former AHA president Donna K. Arnett, PhD, who was not involved in the study, said in a statement that “we’ve known for a long time that moderate or intense levels of physical exercise can reduce a person’s risk of both atherosclerotic cardiovascular disease and mortality.
“We have also seen that getting more than 300 minutes of moderate-intensity aerobic physical activity or more than 150 minutes of vigorous-intensity aerobic physical exercise each week may reduce a person’s risk of atherosclerotic cardiovascular disease even further, so it makes sense that getting those extra minutes of exercise may also decrease mortality,” she added.
Mix and match
Dr. Giovannucci noted that the joint effects of the two types of exercise on mortality have not been studied and “there are some questions, for example, about whether doing a lot of moderate activity is sufficient or can you get more benefits by doing vigorous activity also.”
Joint analyses of both exercise intensities found that additional vigorous physical activity was associated with lower mortality among participants with insufficient (less than 300 min/week) levels of moderate exercise but not among those with at least 300 min/week of moderate exercise.
“The main message is that you can get essentially all of the benefit by just doing moderate exercise,” Dr. Giovannucci said. “There’s no magic benefit of doing vigorous [exercise]. But if someone wants to do vigorous, they can get the benefit in about half the time. So if you only have 2-3 hours a week to exercise and can do, say 2 or 3 hours of running, you can get pretty much the maximum benefit.”
Sensitivity analyses showed a consistent association between long-term leisure physical activity and mortality without adjustment for body mass index/calorie intake.
“Some people think the effect of exercise is to lower your body weight or keep it down, which could be one of the benefits, but even independent of that, you get benefits even if it has no effect on your weight,” he said. “So, definitely, that’s important.”
Dr. Michos pointed out that vigorous physical activity may seem daunting for many individuals but that moderate exercise can include activities such as brisk walking, ballroom dancing, active yoga, and recreational swimming.
“The nice thing is that you can really combine or substitute both and get just as similar mortality reductions with moderate physical activity, because a lot of patients may not want to do vigorous activity,” she said. “They don’t want to get on the treadmill; that’s too intimidating or stressful.”
The study was supported by the National Institutes of Health. The authors and Dr. Michos report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study suggests that going beyond current guidance on moderate and vigorous physical activity levels may add years to one’s life.
Americans are advised to do a minimum of 150-300 minutes a week of moderate exercise or 75-150 minutes a week of vigorous exercise, or an equivalent combination of both, according to U.S. Department of Health and Human Services Physical Activity Guidelines.
Results from more than 100,000 U.S. adults followed for 30 years showed that .
Adults who reported completing four times the minimum recommended activity levels saw no clear incremental mortality benefit but also no harm, according to the study, published in the journal Circulation.
“I think we’re worried more about the lower end and people that are not even doing the minimum, but this should be reassuring to people who like to do a lot of exercise,” senior author Edward Giovannucci, MD, ScD, with the Harvard T.H. Chan School of Public Health, Boston, told this news organization.
Some studies have suggested that long-term, high-intensity exercise (e.g., marathons, triathlons, and long-distance cycling) may be associated with increased risks of atrial fibrillation, coronary artery calcification, and sudden cardiac death.
A recent analysis from the Copenhagen City Heart Study showed a U-shaped association between long-term all-cause mortality and 0 to 2.5 hours and more than 10 hours of weekly, leisure-time sports activities.
Most studies suggesting harm, however, have used only one measurement of physical activity capturing a mix of people who chronically exercise at high levels and those who do it sporadically, which possibly can be harmful, Dr. Giovannucci said. “We were better able to look at consistent long-term activity and saw there was no harm.”
The study included 116,221 participants in the Nurses’ Health Study and the Health Professionals Follow-up Study between 1988 and 2018, who completed up to 15 (median, 11) questionnaires on their health and leisure-time physical activity that were updated every 2 years.
Most were White (96%), 63% were female, and the average age and body mass index over follow-up was 66 years and 26 kg/m2. During 30 years of follow-up, there were 47,596 deaths.
‘Any effort is worthwhile’
The analysis found that individuals who met the guideline for long-term vigorous physical activity (75-150 min/week) cut their adjusted risk of death from cardiovascular disease (CVD) by a whopping 31%, from non-CVD causes by 15%, and all-causes by 19%, compared with those with no long-term vigorous activity.
Those completing two to four times the recommended minimum (150-299 min/week) had a 27%-33% lower risk of CVD mortality, 19% lower risk of non-CVD mortality, and 21%-23% lower risk of all-cause mortality.
Higher levels did not appear to further lower mortality risk. For example, 300-374 min/week of vigorous physical activity was associated with a 32% lower risk of CVD death, 18% lower risk of non-CVD death, and 22% lower risk of dying from any cause.
The analysis also found that individuals who met the guidelines for moderate physical activity had lower CVD, non-CVD, and all-cause mortality risks whether they were active 150-244 min/week (22%, 19%, and 20%, respectively) or 225-299 min/week (21%, 25%, and 20%, respectively), compared with those with almost no long-term moderate activity.
Those fitting in two to four times the recommended minimum (300-599 min/week) had a 28%-38% lower risk of CVD mortality, 25%-27% lower risk of non-CVD mortality, and 26%-31% lower risk of all-cause mortality.
The mortality benefit appeared to plateau, with 600 min/week of moderate physical activity showing associations similar to 300-599 min/week.
“The sweet spot seems to be two to four times the recommended levels but for people who are sedentary, I think one of the key messages that I give my patients is that any effort is worthwhile; that any physical activity, even less than the recommended, has some mortality reduction,” Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins University, Baltimore, said in an interview.
Indeed, individuals who reported doing just 20-74 minutes of moderate exercise per week had a 19% lower risk of dying from any cause and a 13% lower risk of dying from CVD compared with those doing less.
Current American Heart Association (AHA) recommendations are for at least 150 minutes per week of moderate-intensity aerobic exercise or 75 minutes per week of vigorous aerobic exercise, or a combination of both.
“This suggests that even more is probably better, in the range of two to four times that, so maybe we should move our targets a little bit higher, which is kind of what the Department of Health and Human Services has already done,” said Dr. Michos, who was not involved in the study.
Former AHA president Donna K. Arnett, PhD, who was not involved in the study, said in a statement that “we’ve known for a long time that moderate or intense levels of physical exercise can reduce a person’s risk of both atherosclerotic cardiovascular disease and mortality.
“We have also seen that getting more than 300 minutes of moderate-intensity aerobic physical activity or more than 150 minutes of vigorous-intensity aerobic physical exercise each week may reduce a person’s risk of atherosclerotic cardiovascular disease even further, so it makes sense that getting those extra minutes of exercise may also decrease mortality,” she added.
Mix and match
Dr. Giovannucci noted that the joint effects of the two types of exercise on mortality have not been studied and “there are some questions, for example, about whether doing a lot of moderate activity is sufficient or can you get more benefits by doing vigorous activity also.”
Joint analyses of both exercise intensities found that additional vigorous physical activity was associated with lower mortality among participants with insufficient (less than 300 min/week) levels of moderate exercise but not among those with at least 300 min/week of moderate exercise.
“The main message is that you can get essentially all of the benefit by just doing moderate exercise,” Dr. Giovannucci said. “There’s no magic benefit of doing vigorous [exercise]. But if someone wants to do vigorous, they can get the benefit in about half the time. So if you only have 2-3 hours a week to exercise and can do, say 2 or 3 hours of running, you can get pretty much the maximum benefit.”
Sensitivity analyses showed a consistent association between long-term leisure physical activity and mortality without adjustment for body mass index/calorie intake.
“Some people think the effect of exercise is to lower your body weight or keep it down, which could be one of the benefits, but even independent of that, you get benefits even if it has no effect on your weight,” he said. “So, definitely, that’s important.”
Dr. Michos pointed out that vigorous physical activity may seem daunting for many individuals but that moderate exercise can include activities such as brisk walking, ballroom dancing, active yoga, and recreational swimming.
“The nice thing is that you can really combine or substitute both and get just as similar mortality reductions with moderate physical activity, because a lot of patients may not want to do vigorous activity,” she said. “They don’t want to get on the treadmill; that’s too intimidating or stressful.”
The study was supported by the National Institutes of Health. The authors and Dr. Michos report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study suggests that going beyond current guidance on moderate and vigorous physical activity levels may add years to one’s life.
Americans are advised to do a minimum of 150-300 minutes a week of moderate exercise or 75-150 minutes a week of vigorous exercise, or an equivalent combination of both, according to U.S. Department of Health and Human Services Physical Activity Guidelines.
Results from more than 100,000 U.S. adults followed for 30 years showed that .
Adults who reported completing four times the minimum recommended activity levels saw no clear incremental mortality benefit but also no harm, according to the study, published in the journal Circulation.
“I think we’re worried more about the lower end and people that are not even doing the minimum, but this should be reassuring to people who like to do a lot of exercise,” senior author Edward Giovannucci, MD, ScD, with the Harvard T.H. Chan School of Public Health, Boston, told this news organization.
Some studies have suggested that long-term, high-intensity exercise (e.g., marathons, triathlons, and long-distance cycling) may be associated with increased risks of atrial fibrillation, coronary artery calcification, and sudden cardiac death.
A recent analysis from the Copenhagen City Heart Study showed a U-shaped association between long-term all-cause mortality and 0 to 2.5 hours and more than 10 hours of weekly, leisure-time sports activities.
Most studies suggesting harm, however, have used only one measurement of physical activity capturing a mix of people who chronically exercise at high levels and those who do it sporadically, which possibly can be harmful, Dr. Giovannucci said. “We were better able to look at consistent long-term activity and saw there was no harm.”
The study included 116,221 participants in the Nurses’ Health Study and the Health Professionals Follow-up Study between 1988 and 2018, who completed up to 15 (median, 11) questionnaires on their health and leisure-time physical activity that were updated every 2 years.
Most were White (96%), 63% were female, and the average age and body mass index over follow-up was 66 years and 26 kg/m2. During 30 years of follow-up, there were 47,596 deaths.
‘Any effort is worthwhile’
The analysis found that individuals who met the guideline for long-term vigorous physical activity (75-150 min/week) cut their adjusted risk of death from cardiovascular disease (CVD) by a whopping 31%, from non-CVD causes by 15%, and all-causes by 19%, compared with those with no long-term vigorous activity.
Those completing two to four times the recommended minimum (150-299 min/week) had a 27%-33% lower risk of CVD mortality, 19% lower risk of non-CVD mortality, and 21%-23% lower risk of all-cause mortality.
Higher levels did not appear to further lower mortality risk. For example, 300-374 min/week of vigorous physical activity was associated with a 32% lower risk of CVD death, 18% lower risk of non-CVD death, and 22% lower risk of dying from any cause.
The analysis also found that individuals who met the guidelines for moderate physical activity had lower CVD, non-CVD, and all-cause mortality risks whether they were active 150-244 min/week (22%, 19%, and 20%, respectively) or 225-299 min/week (21%, 25%, and 20%, respectively), compared with those with almost no long-term moderate activity.
Those fitting in two to four times the recommended minimum (300-599 min/week) had a 28%-38% lower risk of CVD mortality, 25%-27% lower risk of non-CVD mortality, and 26%-31% lower risk of all-cause mortality.
The mortality benefit appeared to plateau, with 600 min/week of moderate physical activity showing associations similar to 300-599 min/week.
“The sweet spot seems to be two to four times the recommended levels but for people who are sedentary, I think one of the key messages that I give my patients is that any effort is worthwhile; that any physical activity, even less than the recommended, has some mortality reduction,” Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins University, Baltimore, said in an interview.
Indeed, individuals who reported doing just 20-74 minutes of moderate exercise per week had a 19% lower risk of dying from any cause and a 13% lower risk of dying from CVD compared with those doing less.
Current American Heart Association (AHA) recommendations are for at least 150 minutes per week of moderate-intensity aerobic exercise or 75 minutes per week of vigorous aerobic exercise, or a combination of both.
“This suggests that even more is probably better, in the range of two to four times that, so maybe we should move our targets a little bit higher, which is kind of what the Department of Health and Human Services has already done,” said Dr. Michos, who was not involved in the study.
Former AHA president Donna K. Arnett, PhD, who was not involved in the study, said in a statement that “we’ve known for a long time that moderate or intense levels of physical exercise can reduce a person’s risk of both atherosclerotic cardiovascular disease and mortality.
“We have also seen that getting more than 300 minutes of moderate-intensity aerobic physical activity or more than 150 minutes of vigorous-intensity aerobic physical exercise each week may reduce a person’s risk of atherosclerotic cardiovascular disease even further, so it makes sense that getting those extra minutes of exercise may also decrease mortality,” she added.
Mix and match
Dr. Giovannucci noted that the joint effects of the two types of exercise on mortality have not been studied and “there are some questions, for example, about whether doing a lot of moderate activity is sufficient or can you get more benefits by doing vigorous activity also.”
Joint analyses of both exercise intensities found that additional vigorous physical activity was associated with lower mortality among participants with insufficient (less than 300 min/week) levels of moderate exercise but not among those with at least 300 min/week of moderate exercise.
“The main message is that you can get essentially all of the benefit by just doing moderate exercise,” Dr. Giovannucci said. “There’s no magic benefit of doing vigorous [exercise]. But if someone wants to do vigorous, they can get the benefit in about half the time. So if you only have 2-3 hours a week to exercise and can do, say 2 or 3 hours of running, you can get pretty much the maximum benefit.”
Sensitivity analyses showed a consistent association between long-term leisure physical activity and mortality without adjustment for body mass index/calorie intake.
“Some people think the effect of exercise is to lower your body weight or keep it down, which could be one of the benefits, but even independent of that, you get benefits even if it has no effect on your weight,” he said. “So, definitely, that’s important.”
Dr. Michos pointed out that vigorous physical activity may seem daunting for many individuals but that moderate exercise can include activities such as brisk walking, ballroom dancing, active yoga, and recreational swimming.
“The nice thing is that you can really combine or substitute both and get just as similar mortality reductions with moderate physical activity, because a lot of patients may not want to do vigorous activity,” she said. “They don’t want to get on the treadmill; that’s too intimidating or stressful.”
The study was supported by the National Institutes of Health. The authors and Dr. Michos report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Hospital programs tackle mental health effects of long COVID
There’s little doubt that long COVID is real. Even as doctors and federal agencies struggle to define the syndrome, hospitals and health care systems are opening long COVID specialty treatment programs. As of July 25, there’s at least one long COVID center in almost every state – 48 out of 50, according to the patient advocacy group Survivor Corps.
Among the biggest challenges will be treating the mental health effects of long COVID.
Specialized centers will be tackling these problems even as the United States struggles to deal with mental health needs.
One study of COVID patients found more than one-third of them had symptoms of depression, anxiety, or PTSD 3-6 months after their initial infection. Another analysis of 30 previous studies of long COVID patients found roughly one in eight of them had severe depression – and that the risk was similar regardless of whether people were hospitalized for COVID-19.
“Many of these symptoms can emerge months into the course of long COVID illness,” said Jordan Anderson, DO, a neuropsychiatrist who sees patients at the Long COVID-19 Program at Oregon Health & Science University, Portland. Psychological symptoms are often made worse by physical setbacks like extreme fatigue and by challenges of working, caring for children, and keeping up with daily routines, he said.
“This impact is not only severe, but also chronic for many,” he said.
Like dozens of hospitals around the country, Oregon Health & Science opened its center for long COVID as it became clear that more patients would need help for ongoing physical and mental health symptoms. Today, there’s at least one long COVID center – sometimes called post-COVID care centers or clinics – in every state but Kansas and South Dakota, Survivor Corps said.
Many long COVID care centers aim to tackle both physical and mental health symptoms, said Tracy Vannorsdall, PhD, a neuropsychologist with the Johns Hopkins Post-Acute COVID-19 Team program. One goal at Hopkins is to identify patients with psychological issues that might otherwise get overlooked.
A sizable minority of patients at the Johns Hopkins center – up to about 35% – report mental health problems that they didn’t have until after they got COVID-19, Dr. Vannorsdall says. The most common mental health issues providers see are depression, anxiety, and trauma-related distress.
“Routine assessment is key,” Dr. Vannorsdall said. “If patients are not asked about their mental health symptoms, they may not spontaneously report them to their provider due to fear of stigma or simply not appreciating that there are effective treatments available for these issues.”
Fear that doctors won’t take symptoms seriously is common, says Heather Murray MD, a senior instructor in psychiatry at the University of Colorado at Denver, Aurora.
“Many patients worry their physicians, loved ones, and society will not believe them or will minimize their symptoms and suffering,” said Dr. Murray, who treats patients at the UCHealth Post-COVID Clinic.
Diagnostic tests in long COVID patients often don’t have conclusive results, which can lead doctors and patients themselves to question whether symptoms are truly “physical versus psychosomatic,” she said. “It is important that providers believe their patients and treat their symptoms, even when diagnostic tests are unrevealing.”
Growing mental health crisis
Patients often find their way to academic treatment centers after surviving severe COVID-19 infections. But a growing number of long COVID patients show up at these centers after milder cases. These patients were never hospitalized for COVID-19 but still have persistent symptoms like fatigue, thinking problems, and mood disorders.
Among the major challenges is a shortage of mental health care providers to meet the surging need for care since the start of the pandemic. Around the world, anxiety and depression surged 25% during the first year of the pandemic, according to the World Health Organization.
In the United States, 40% of adults report feelings of anxiety and depression, and one in three high school students have feelings of sadness and hopelessness, according to a March 2022 statement from the White House.
Despite this surging need for care, almost half of Americans live in areas with a severe shortage of mental health care providers, according to the Health Resources and Services Administration. As of 2019, the United States had a shortage of about 6,790 mental health providers. Since then, the shortage has worsened; it’s now about 7,500 providers.
“One of the biggest challenges for hospitals and clinics in treating mental health disorders in long COVID is the limited resources and long wait times to get in for evaluations and treatment,” said Nyaz Didehbani, PhD, a neuropsychologist who treats long COVID patients at the COVID Recover program at the University of Texas Southwestern Medical Center, Dallas.
These delays can lead to worse outcomes, Dr. Didehbani said. “Additionally, patients do not feel that they are being heard, as many providers are not aware of the mental health impact and relationship with physical and cognitive symptoms.” .
Even when doctors recognize that psychological challenges are common with long COVID, they still have to think creatively to come up with treatments that meet the unique needs of these patients, said Thida Thant, MD, an assistant professor of psychiatry at the University of Colorado who treats patients at the UCHealth Post-COVID Clinic.
“There are at least two major factors that make treating psychological issues in long COVID more complex: The fact that the pandemic is still ongoing and still so divisive throughout society, and the fact that we don’t know a single best way to treat all symptoms of long COVID,” she said.
Some common treatments for anxiety and depression, like psychotherapy and medication, can be used for long COVID patients with these conditions. But another intervention that can work wonders for many people with mood disorders – exercise – doesn’t always work for long COVID patients. That’s because many of them struggle with physical challenges like chronic fatigue and what’s known as postexertional malaise, or a worsening of symptoms after even limited physical effort.
“While we normally encourage patients to be active, have a daily routine, and to engage in physical activity as part of their mental health treatment, some long COVID patients find that their symptoms worsen after increased activity,” Dr. Vannorsdall said.
Patients who are able to reach long COVID care centers are much more apt to get mental health problems diagnosed and treated, doctors at many programs around the country agree. But many patients hardest hit by the pandemic – the poor and racial and ethnic minorities – are also less likely to have ready access to hospitals that offer these programs, said Dr. Anderson.
“Affluent, predominantly White populations are showing up in these clinics, while we know that non-White populations have disproportionally high rates of acute infection, hospitalization, and death related to the virus,” he said.
Clinics are also concentrated in academic medical centers and in urban areas, limiting options for people in rural communities who may have to drive for hours to access care, Dr. Anderson said.
“Even before long COVID, we already knew that many people live in areas where there simply aren’t enough mental health services available,” said John Zulueta, MD, an assistant professor of clinical psychiatry at the University of Illinois at Chicago who provides mental health evaluations at the UI Health Post-COVID Clinic.
“As more patients develop mental health issues associated with long COVID, it’s going to put more stress on an already stressed system,” he said.
A version of this article first appeared on WebMD.com.
There’s little doubt that long COVID is real. Even as doctors and federal agencies struggle to define the syndrome, hospitals and health care systems are opening long COVID specialty treatment programs. As of July 25, there’s at least one long COVID center in almost every state – 48 out of 50, according to the patient advocacy group Survivor Corps.
Among the biggest challenges will be treating the mental health effects of long COVID.
Specialized centers will be tackling these problems even as the United States struggles to deal with mental health needs.
One study of COVID patients found more than one-third of them had symptoms of depression, anxiety, or PTSD 3-6 months after their initial infection. Another analysis of 30 previous studies of long COVID patients found roughly one in eight of them had severe depression – and that the risk was similar regardless of whether people were hospitalized for COVID-19.
“Many of these symptoms can emerge months into the course of long COVID illness,” said Jordan Anderson, DO, a neuropsychiatrist who sees patients at the Long COVID-19 Program at Oregon Health & Science University, Portland. Psychological symptoms are often made worse by physical setbacks like extreme fatigue and by challenges of working, caring for children, and keeping up with daily routines, he said.
“This impact is not only severe, but also chronic for many,” he said.
Like dozens of hospitals around the country, Oregon Health & Science opened its center for long COVID as it became clear that more patients would need help for ongoing physical and mental health symptoms. Today, there’s at least one long COVID center – sometimes called post-COVID care centers or clinics – in every state but Kansas and South Dakota, Survivor Corps said.
Many long COVID care centers aim to tackle both physical and mental health symptoms, said Tracy Vannorsdall, PhD, a neuropsychologist with the Johns Hopkins Post-Acute COVID-19 Team program. One goal at Hopkins is to identify patients with psychological issues that might otherwise get overlooked.
A sizable minority of patients at the Johns Hopkins center – up to about 35% – report mental health problems that they didn’t have until after they got COVID-19, Dr. Vannorsdall says. The most common mental health issues providers see are depression, anxiety, and trauma-related distress.
“Routine assessment is key,” Dr. Vannorsdall said. “If patients are not asked about their mental health symptoms, they may not spontaneously report them to their provider due to fear of stigma or simply not appreciating that there are effective treatments available for these issues.”
Fear that doctors won’t take symptoms seriously is common, says Heather Murray MD, a senior instructor in psychiatry at the University of Colorado at Denver, Aurora.
“Many patients worry their physicians, loved ones, and society will not believe them or will minimize their symptoms and suffering,” said Dr. Murray, who treats patients at the UCHealth Post-COVID Clinic.
Diagnostic tests in long COVID patients often don’t have conclusive results, which can lead doctors and patients themselves to question whether symptoms are truly “physical versus psychosomatic,” she said. “It is important that providers believe their patients and treat their symptoms, even when diagnostic tests are unrevealing.”
Growing mental health crisis
Patients often find their way to academic treatment centers after surviving severe COVID-19 infections. But a growing number of long COVID patients show up at these centers after milder cases. These patients were never hospitalized for COVID-19 but still have persistent symptoms like fatigue, thinking problems, and mood disorders.
Among the major challenges is a shortage of mental health care providers to meet the surging need for care since the start of the pandemic. Around the world, anxiety and depression surged 25% during the first year of the pandemic, according to the World Health Organization.
In the United States, 40% of adults report feelings of anxiety and depression, and one in three high school students have feelings of sadness and hopelessness, according to a March 2022 statement from the White House.
Despite this surging need for care, almost half of Americans live in areas with a severe shortage of mental health care providers, according to the Health Resources and Services Administration. As of 2019, the United States had a shortage of about 6,790 mental health providers. Since then, the shortage has worsened; it’s now about 7,500 providers.
“One of the biggest challenges for hospitals and clinics in treating mental health disorders in long COVID is the limited resources and long wait times to get in for evaluations and treatment,” said Nyaz Didehbani, PhD, a neuropsychologist who treats long COVID patients at the COVID Recover program at the University of Texas Southwestern Medical Center, Dallas.
These delays can lead to worse outcomes, Dr. Didehbani said. “Additionally, patients do not feel that they are being heard, as many providers are not aware of the mental health impact and relationship with physical and cognitive symptoms.” .
Even when doctors recognize that psychological challenges are common with long COVID, they still have to think creatively to come up with treatments that meet the unique needs of these patients, said Thida Thant, MD, an assistant professor of psychiatry at the University of Colorado who treats patients at the UCHealth Post-COVID Clinic.
“There are at least two major factors that make treating psychological issues in long COVID more complex: The fact that the pandemic is still ongoing and still so divisive throughout society, and the fact that we don’t know a single best way to treat all symptoms of long COVID,” she said.
Some common treatments for anxiety and depression, like psychotherapy and medication, can be used for long COVID patients with these conditions. But another intervention that can work wonders for many people with mood disorders – exercise – doesn’t always work for long COVID patients. That’s because many of them struggle with physical challenges like chronic fatigue and what’s known as postexertional malaise, or a worsening of symptoms after even limited physical effort.
“While we normally encourage patients to be active, have a daily routine, and to engage in physical activity as part of their mental health treatment, some long COVID patients find that their symptoms worsen after increased activity,” Dr. Vannorsdall said.
Patients who are able to reach long COVID care centers are much more apt to get mental health problems diagnosed and treated, doctors at many programs around the country agree. But many patients hardest hit by the pandemic – the poor and racial and ethnic minorities – are also less likely to have ready access to hospitals that offer these programs, said Dr. Anderson.
“Affluent, predominantly White populations are showing up in these clinics, while we know that non-White populations have disproportionally high rates of acute infection, hospitalization, and death related to the virus,” he said.
Clinics are also concentrated in academic medical centers and in urban areas, limiting options for people in rural communities who may have to drive for hours to access care, Dr. Anderson said.
“Even before long COVID, we already knew that many people live in areas where there simply aren’t enough mental health services available,” said John Zulueta, MD, an assistant professor of clinical psychiatry at the University of Illinois at Chicago who provides mental health evaluations at the UI Health Post-COVID Clinic.
“As more patients develop mental health issues associated with long COVID, it’s going to put more stress on an already stressed system,” he said.
A version of this article first appeared on WebMD.com.
There’s little doubt that long COVID is real. Even as doctors and federal agencies struggle to define the syndrome, hospitals and health care systems are opening long COVID specialty treatment programs. As of July 25, there’s at least one long COVID center in almost every state – 48 out of 50, according to the patient advocacy group Survivor Corps.
Among the biggest challenges will be treating the mental health effects of long COVID.
Specialized centers will be tackling these problems even as the United States struggles to deal with mental health needs.
One study of COVID patients found more than one-third of them had symptoms of depression, anxiety, or PTSD 3-6 months after their initial infection. Another analysis of 30 previous studies of long COVID patients found roughly one in eight of them had severe depression – and that the risk was similar regardless of whether people were hospitalized for COVID-19.
“Many of these symptoms can emerge months into the course of long COVID illness,” said Jordan Anderson, DO, a neuropsychiatrist who sees patients at the Long COVID-19 Program at Oregon Health & Science University, Portland. Psychological symptoms are often made worse by physical setbacks like extreme fatigue and by challenges of working, caring for children, and keeping up with daily routines, he said.
“This impact is not only severe, but also chronic for many,” he said.
Like dozens of hospitals around the country, Oregon Health & Science opened its center for long COVID as it became clear that more patients would need help for ongoing physical and mental health symptoms. Today, there’s at least one long COVID center – sometimes called post-COVID care centers or clinics – in every state but Kansas and South Dakota, Survivor Corps said.
Many long COVID care centers aim to tackle both physical and mental health symptoms, said Tracy Vannorsdall, PhD, a neuropsychologist with the Johns Hopkins Post-Acute COVID-19 Team program. One goal at Hopkins is to identify patients with psychological issues that might otherwise get overlooked.
A sizable minority of patients at the Johns Hopkins center – up to about 35% – report mental health problems that they didn’t have until after they got COVID-19, Dr. Vannorsdall says. The most common mental health issues providers see are depression, anxiety, and trauma-related distress.
“Routine assessment is key,” Dr. Vannorsdall said. “If patients are not asked about their mental health symptoms, they may not spontaneously report them to their provider due to fear of stigma or simply not appreciating that there are effective treatments available for these issues.”
Fear that doctors won’t take symptoms seriously is common, says Heather Murray MD, a senior instructor in psychiatry at the University of Colorado at Denver, Aurora.
“Many patients worry their physicians, loved ones, and society will not believe them or will minimize their symptoms and suffering,” said Dr. Murray, who treats patients at the UCHealth Post-COVID Clinic.
Diagnostic tests in long COVID patients often don’t have conclusive results, which can lead doctors and patients themselves to question whether symptoms are truly “physical versus psychosomatic,” she said. “It is important that providers believe their patients and treat their symptoms, even when diagnostic tests are unrevealing.”
Growing mental health crisis
Patients often find their way to academic treatment centers after surviving severe COVID-19 infections. But a growing number of long COVID patients show up at these centers after milder cases. These patients were never hospitalized for COVID-19 but still have persistent symptoms like fatigue, thinking problems, and mood disorders.
Among the major challenges is a shortage of mental health care providers to meet the surging need for care since the start of the pandemic. Around the world, anxiety and depression surged 25% during the first year of the pandemic, according to the World Health Organization.
In the United States, 40% of adults report feelings of anxiety and depression, and one in three high school students have feelings of sadness and hopelessness, according to a March 2022 statement from the White House.
Despite this surging need for care, almost half of Americans live in areas with a severe shortage of mental health care providers, according to the Health Resources and Services Administration. As of 2019, the United States had a shortage of about 6,790 mental health providers. Since then, the shortage has worsened; it’s now about 7,500 providers.
“One of the biggest challenges for hospitals and clinics in treating mental health disorders in long COVID is the limited resources and long wait times to get in for evaluations and treatment,” said Nyaz Didehbani, PhD, a neuropsychologist who treats long COVID patients at the COVID Recover program at the University of Texas Southwestern Medical Center, Dallas.
These delays can lead to worse outcomes, Dr. Didehbani said. “Additionally, patients do not feel that they are being heard, as many providers are not aware of the mental health impact and relationship with physical and cognitive symptoms.” .
Even when doctors recognize that psychological challenges are common with long COVID, they still have to think creatively to come up with treatments that meet the unique needs of these patients, said Thida Thant, MD, an assistant professor of psychiatry at the University of Colorado who treats patients at the UCHealth Post-COVID Clinic.
“There are at least two major factors that make treating psychological issues in long COVID more complex: The fact that the pandemic is still ongoing and still so divisive throughout society, and the fact that we don’t know a single best way to treat all symptoms of long COVID,” she said.
Some common treatments for anxiety and depression, like psychotherapy and medication, can be used for long COVID patients with these conditions. But another intervention that can work wonders for many people with mood disorders – exercise – doesn’t always work for long COVID patients. That’s because many of them struggle with physical challenges like chronic fatigue and what’s known as postexertional malaise, or a worsening of symptoms after even limited physical effort.
“While we normally encourage patients to be active, have a daily routine, and to engage in physical activity as part of their mental health treatment, some long COVID patients find that their symptoms worsen after increased activity,” Dr. Vannorsdall said.
Patients who are able to reach long COVID care centers are much more apt to get mental health problems diagnosed and treated, doctors at many programs around the country agree. But many patients hardest hit by the pandemic – the poor and racial and ethnic minorities – are also less likely to have ready access to hospitals that offer these programs, said Dr. Anderson.
“Affluent, predominantly White populations are showing up in these clinics, while we know that non-White populations have disproportionally high rates of acute infection, hospitalization, and death related to the virus,” he said.
Clinics are also concentrated in academic medical centers and in urban areas, limiting options for people in rural communities who may have to drive for hours to access care, Dr. Anderson said.
“Even before long COVID, we already knew that many people live in areas where there simply aren’t enough mental health services available,” said John Zulueta, MD, an assistant professor of clinical psychiatry at the University of Illinois at Chicago who provides mental health evaluations at the UI Health Post-COVID Clinic.
“As more patients develop mental health issues associated with long COVID, it’s going to put more stress on an already stressed system,” he said.
A version of this article first appeared on WebMD.com.
‘Reassuring’ safety data on PPI therapy
In a novel analysis accounting for protopathic bias, proton pump inhibitor (PPI) therapy was not associated with increased risk for death due to digestive disease, cancer, cardiovascular disease (CVD), or any cause, although the jury is out on renal disease.
“There have been several studies suggesting that PPIs can cause long-term health problems and may be associated with increased mortality,” Andrew T. Chan, MD, MPH, gastroenterologist and professor of medicine, Massachusetts General Hospital and Harvard Medical School, both in Boston, told this news organization.
“We conducted this study to examine this issue using data that were better able to account for potential biases in those prior studies. We found that PPIs were generally not associated with an increased risk of mortality,” Dr. Chan said.
The study was published online in Gastroenterology.
‘Reassuring’ data
The findings are based on data collected between 2004 and 2018 from 50,156 women enrolled in the Nurses’ Health Study and 21,731 men enrolled from the Health Professionals Follow-up Study.
During the study period, 10,998 women (21.9%) and 2,945 men (13.6%) initiated PPI therapy, and PPI use increased over the study period from 6.1% to 10.0% in women and from 2.5% to 7.0% in men.
The mean age at baseline was 68.9 years for women and 68.0 years for men. During a median follow-up of 13.8 years, a total of 22,125 participants died – 4,592 of cancer, 5,404 of CVD, and 12,129 of other causes.
Unlike other studies, the researchers used a modified lag-time approach to minimize reverse causation (protopathic bias).
“Using this approach, any increased PPI use during the excluded period, which could be due to comorbid conditions prior to death, will not be considered in the quantification of the exposure, and thus, protopathic bias would be avoided,” they explain.
In the initial analysis that did not take into account lag times, PPI users had significantly higher risks for all-cause mortality and mortality due to cancer, CVD, respiratory diseases, and digestive diseases, compared with nonusers.
However, when applying lag times of up to 6 years, the associations were largely attenuated and no longer statistically significant, which “highlights the importance of carefully controlling for the influence of protopathic bias,” the researchers write.
However, despite applying lag times, PPI users remained at a significantly increased risk for mortality due to renal diseases (hazard ratio, 2.45; 95% confidence interval, 1.59-3.78).
The researchers caution, however, that they did not have reliable data on renal diseases and therefore could not adjust for confounding in the models. They call for further studies examining the risk for mortality due to renal diseases in patients using PPI therapy.
The researchers also looked at duration of PPI use and all-cause and cause-specific mortality.
For all-cause mortality and mortality due to cancer, CVD, respiratory diseases, and digestive diseases, the greatest risks were seen mostly in those who reported PPI use for 1-2 years. Longer duration of PPI use did not confer higher risk for mortality for these endpoints.
In contrast, a potential trend toward greater risk with longer duration of PPI use was observed for mortality due to renal disease. The hazard ratio was 1.68 (95% CI, 1.19-2.38) for 1 to 2 years of use and gradually increased to 2.42 (95% CI, 1.23-4.77) for 7 or more years of use.
Notably, when mortality risks were compared among PPI users and histamine H2 receptor antagonist (H2RA) users without lag time, PPI users were at increased risk for all-cause mortality and mortality due to causes other than cancer and CVD, compared with H2RA users.
But again, the strength of the associations decreased after lag time was introduced.
“This confirmed our main findings and suggested PPIs might be preferred over H2RAs in sicker patients with comorbid conditions,” the researchers write.
‘Generally safe’ when needed
Summing up, Dr. Chan said, “We think our results should be reassuring to clinicians that recommending PPIs to patients with appropriate indications will not increase their risk of death. These are generally safe drugs that when used appropriately can be very beneficial.”
Offering perspective on the study, David Johnson, MD, professor of medicine and chief of gastroenterology at the Eastern Virginia School of Medicine, Norfolk, noted that a “major continuing criticism of the allegations of harm by PPIs has been that these most commonly come from retrospective analyses of databases that were not constructed to evaluate these endpoints of harm.”
“Accordingly, these reports have multiple potentials for stratification bias and typically have low odds ratios for supporting the purported causality,” Dr. Johnson told this news organization.
“This is a well-done study design with a prospective database analysis that uses a modified lag-time approach to minimize reverse causation, that is, protopathic bias, which can occur when a pharmaceutical agent is inadvertently prescribed for an early manifestation of a disease that has not yet been diagnostically detected,” Dr. Johnson explained.
Echoing Dr. Chan, Dr. Johnson said the finding that PPI use was not associated with higher risk for all-cause mortality and mortality due to major causes is “reassuring.”
“Recognizably, too many people are taking PPIs chronically when they are not needed. If needed and appropriate, these data on continued use are reassuring,” Dr. Johnson added.
This work was supported by the National Institutes of Health and the Crohn’s and Colitis Foundation. Dr. Chan has consulted for OM1, Bayer Pharma AG, and Pfizer for topics unrelated to this study, as well as Boehringer Ingelheim for litigation related to ranitidine and cancer. Dr. Johnson reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a novel analysis accounting for protopathic bias, proton pump inhibitor (PPI) therapy was not associated with increased risk for death due to digestive disease, cancer, cardiovascular disease (CVD), or any cause, although the jury is out on renal disease.
“There have been several studies suggesting that PPIs can cause long-term health problems and may be associated with increased mortality,” Andrew T. Chan, MD, MPH, gastroenterologist and professor of medicine, Massachusetts General Hospital and Harvard Medical School, both in Boston, told this news organization.
“We conducted this study to examine this issue using data that were better able to account for potential biases in those prior studies. We found that PPIs were generally not associated with an increased risk of mortality,” Dr. Chan said.
The study was published online in Gastroenterology.
‘Reassuring’ data
The findings are based on data collected between 2004 and 2018 from 50,156 women enrolled in the Nurses’ Health Study and 21,731 men enrolled from the Health Professionals Follow-up Study.
During the study period, 10,998 women (21.9%) and 2,945 men (13.6%) initiated PPI therapy, and PPI use increased over the study period from 6.1% to 10.0% in women and from 2.5% to 7.0% in men.
The mean age at baseline was 68.9 years for women and 68.0 years for men. During a median follow-up of 13.8 years, a total of 22,125 participants died – 4,592 of cancer, 5,404 of CVD, and 12,129 of other causes.
Unlike other studies, the researchers used a modified lag-time approach to minimize reverse causation (protopathic bias).
“Using this approach, any increased PPI use during the excluded period, which could be due to comorbid conditions prior to death, will not be considered in the quantification of the exposure, and thus, protopathic bias would be avoided,” they explain.
In the initial analysis that did not take into account lag times, PPI users had significantly higher risks for all-cause mortality and mortality due to cancer, CVD, respiratory diseases, and digestive diseases, compared with nonusers.
However, when applying lag times of up to 6 years, the associations were largely attenuated and no longer statistically significant, which “highlights the importance of carefully controlling for the influence of protopathic bias,” the researchers write.
However, despite applying lag times, PPI users remained at a significantly increased risk for mortality due to renal diseases (hazard ratio, 2.45; 95% confidence interval, 1.59-3.78).
The researchers caution, however, that they did not have reliable data on renal diseases and therefore could not adjust for confounding in the models. They call for further studies examining the risk for mortality due to renal diseases in patients using PPI therapy.
The researchers also looked at duration of PPI use and all-cause and cause-specific mortality.
For all-cause mortality and mortality due to cancer, CVD, respiratory diseases, and digestive diseases, the greatest risks were seen mostly in those who reported PPI use for 1-2 years. Longer duration of PPI use did not confer higher risk for mortality for these endpoints.
In contrast, a potential trend toward greater risk with longer duration of PPI use was observed for mortality due to renal disease. The hazard ratio was 1.68 (95% CI, 1.19-2.38) for 1 to 2 years of use and gradually increased to 2.42 (95% CI, 1.23-4.77) for 7 or more years of use.
Notably, when mortality risks were compared among PPI users and histamine H2 receptor antagonist (H2RA) users without lag time, PPI users were at increased risk for all-cause mortality and mortality due to causes other than cancer and CVD, compared with H2RA users.
But again, the strength of the associations decreased after lag time was introduced.
“This confirmed our main findings and suggested PPIs might be preferred over H2RAs in sicker patients with comorbid conditions,” the researchers write.
‘Generally safe’ when needed
Summing up, Dr. Chan said, “We think our results should be reassuring to clinicians that recommending PPIs to patients with appropriate indications will not increase their risk of death. These are generally safe drugs that when used appropriately can be very beneficial.”
Offering perspective on the study, David Johnson, MD, professor of medicine and chief of gastroenterology at the Eastern Virginia School of Medicine, Norfolk, noted that a “major continuing criticism of the allegations of harm by PPIs has been that these most commonly come from retrospective analyses of databases that were not constructed to evaluate these endpoints of harm.”
“Accordingly, these reports have multiple potentials for stratification bias and typically have low odds ratios for supporting the purported causality,” Dr. Johnson told this news organization.
“This is a well-done study design with a prospective database analysis that uses a modified lag-time approach to minimize reverse causation, that is, protopathic bias, which can occur when a pharmaceutical agent is inadvertently prescribed for an early manifestation of a disease that has not yet been diagnostically detected,” Dr. Johnson explained.
Echoing Dr. Chan, Dr. Johnson said the finding that PPI use was not associated with higher risk for all-cause mortality and mortality due to major causes is “reassuring.”
“Recognizably, too many people are taking PPIs chronically when they are not needed. If needed and appropriate, these data on continued use are reassuring,” Dr. Johnson added.
This work was supported by the National Institutes of Health and the Crohn’s and Colitis Foundation. Dr. Chan has consulted for OM1, Bayer Pharma AG, and Pfizer for topics unrelated to this study, as well as Boehringer Ingelheim for litigation related to ranitidine and cancer. Dr. Johnson reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a novel analysis accounting for protopathic bias, proton pump inhibitor (PPI) therapy was not associated with increased risk for death due to digestive disease, cancer, cardiovascular disease (CVD), or any cause, although the jury is out on renal disease.
“There have been several studies suggesting that PPIs can cause long-term health problems and may be associated with increased mortality,” Andrew T. Chan, MD, MPH, gastroenterologist and professor of medicine, Massachusetts General Hospital and Harvard Medical School, both in Boston, told this news organization.
“We conducted this study to examine this issue using data that were better able to account for potential biases in those prior studies. We found that PPIs were generally not associated with an increased risk of mortality,” Dr. Chan said.
The study was published online in Gastroenterology.
‘Reassuring’ data
The findings are based on data collected between 2004 and 2018 from 50,156 women enrolled in the Nurses’ Health Study and 21,731 men enrolled from the Health Professionals Follow-up Study.
During the study period, 10,998 women (21.9%) and 2,945 men (13.6%) initiated PPI therapy, and PPI use increased over the study period from 6.1% to 10.0% in women and from 2.5% to 7.0% in men.
The mean age at baseline was 68.9 years for women and 68.0 years for men. During a median follow-up of 13.8 years, a total of 22,125 participants died – 4,592 of cancer, 5,404 of CVD, and 12,129 of other causes.
Unlike other studies, the researchers used a modified lag-time approach to minimize reverse causation (protopathic bias).
“Using this approach, any increased PPI use during the excluded period, which could be due to comorbid conditions prior to death, will not be considered in the quantification of the exposure, and thus, protopathic bias would be avoided,” they explain.
In the initial analysis that did not take into account lag times, PPI users had significantly higher risks for all-cause mortality and mortality due to cancer, CVD, respiratory diseases, and digestive diseases, compared with nonusers.
However, when applying lag times of up to 6 years, the associations were largely attenuated and no longer statistically significant, which “highlights the importance of carefully controlling for the influence of protopathic bias,” the researchers write.
However, despite applying lag times, PPI users remained at a significantly increased risk for mortality due to renal diseases (hazard ratio, 2.45; 95% confidence interval, 1.59-3.78).
The researchers caution, however, that they did not have reliable data on renal diseases and therefore could not adjust for confounding in the models. They call for further studies examining the risk for mortality due to renal diseases in patients using PPI therapy.
The researchers also looked at duration of PPI use and all-cause and cause-specific mortality.
For all-cause mortality and mortality due to cancer, CVD, respiratory diseases, and digestive diseases, the greatest risks were seen mostly in those who reported PPI use for 1-2 years. Longer duration of PPI use did not confer higher risk for mortality for these endpoints.
In contrast, a potential trend toward greater risk with longer duration of PPI use was observed for mortality due to renal disease. The hazard ratio was 1.68 (95% CI, 1.19-2.38) for 1 to 2 years of use and gradually increased to 2.42 (95% CI, 1.23-4.77) for 7 or more years of use.
Notably, when mortality risks were compared among PPI users and histamine H2 receptor antagonist (H2RA) users without lag time, PPI users were at increased risk for all-cause mortality and mortality due to causes other than cancer and CVD, compared with H2RA users.
But again, the strength of the associations decreased after lag time was introduced.
“This confirmed our main findings and suggested PPIs might be preferred over H2RAs in sicker patients with comorbid conditions,” the researchers write.
‘Generally safe’ when needed
Summing up, Dr. Chan said, “We think our results should be reassuring to clinicians that recommending PPIs to patients with appropriate indications will not increase their risk of death. These are generally safe drugs that when used appropriately can be very beneficial.”
Offering perspective on the study, David Johnson, MD, professor of medicine and chief of gastroenterology at the Eastern Virginia School of Medicine, Norfolk, noted that a “major continuing criticism of the allegations of harm by PPIs has been that these most commonly come from retrospective analyses of databases that were not constructed to evaluate these endpoints of harm.”
“Accordingly, these reports have multiple potentials for stratification bias and typically have low odds ratios for supporting the purported causality,” Dr. Johnson told this news organization.
“This is a well-done study design with a prospective database analysis that uses a modified lag-time approach to minimize reverse causation, that is, protopathic bias, which can occur when a pharmaceutical agent is inadvertently prescribed for an early manifestation of a disease that has not yet been diagnostically detected,” Dr. Johnson explained.
Echoing Dr. Chan, Dr. Johnson said the finding that PPI use was not associated with higher risk for all-cause mortality and mortality due to major causes is “reassuring.”
“Recognizably, too many people are taking PPIs chronically when they are not needed. If needed and appropriate, these data on continued use are reassuring,” Dr. Johnson added.
This work was supported by the National Institutes of Health and the Crohn’s and Colitis Foundation. Dr. Chan has consulted for OM1, Bayer Pharma AG, and Pfizer for topics unrelated to this study, as well as Boehringer Ingelheim for litigation related to ranitidine and cancer. Dr. Johnson reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM GASTROENTEROLOGY
Smartphone tool helps gauge bowel prep quality before colonoscopy
An artificial intelligence (AI) tool that runs on a smartphone can help patients scheduled for a colonoscopy evaluate independently how well they do with bowel cleansing and may be an alternative approach for evaluating bowel preparation quality before the colonoscopy, especially in the COVID-19 era.
The AI tool is a “manpower-saving” option that reduces the need for nurses to evaluate the quality of bowel preparation, say Wei Gong, MD, Southern Medical University, Shenzhen, China, and colleagues.
Having the tool on a patient’s smartphone means caregivers and nurses would not be required to assess the adequacy of bowel cleansing for patients, which, in turn, would reduce person-to-person contact and the spread of infectious diseases, they add.
The study was published online in the American Journal of Gastroenterology.
Better than do-it-yourself evaluation?
The study was conducted at two hospitals in China among consecutive patients prepping for colonoscopy. All participants received standard bowel preparation instructions and were given a leaflet with general guidelines on bowel preparation.
The leaflet included photos representing bowel preparation quality and informed patients that their stool should eventually be a yellowish clear liquid; if any cloudiness (including turbid liquid, particles, or small amounts of feces) is observed in the liquid stool, the bowel preparation is not complete.
All patients were prescribed standard polyethylene glycol electrolyte solution for bowel cleansing 4-6 hours before the colonoscopy.
After consuming the solution, all patients scanned a QR (quick response) code with a smartphone for randomization into an experimental group using the AI-convolutional neural network (AI-CNN) model or a control group using self-evaluation.
The system gave instructions for using the application, taking photos of their feces, and uploading the images.
After uploading the images, the 730 patients in the AI-CNN group automatically received a “pass” or “not pass” alert, which indicated whether their bowel preparation was adequate or not.
The 704 patients in the control group evaluated the adequacy of bowel preparation on their own according to the leaflet instructions after uploading their images.
Colonoscopists and nurses were blinded to the bowel evaluation method that each patient used.
According to the investigators, evaluation results (“pass” or “not pass”) in terms of adequacy of bowel preparation as represented by Boston Bowel Preparation Scale (BBPS) scores were consistent between the two methods (AI-CNN or self-evaluation).
Overall, there were no significant differences in the two methods in terms of mean BBPS scores, polyp detection rate, or adenoma detection rate.
In subgroup analysis, however, the mean BBPS score of patients with “pass” results was significantly higher in the AI-CNN group than in the self-evaluation control group.
This suggests that the AI-CNN model may further improve the quality of bowel preparation in patients exhibiting adequate bowel preparation, the researchers say.
The results also suggest improved bowel preparation quality of the right colon under the aid of the AI-CNN model, which may be crucial for the prevention of interval colorectal cancer.
The study did not investigate the user acceptability of the AI-CNN model.
“To improve the model and broaden its application in routine practice, evaluating its convenience, accessibility, aspects that cause users difficulty, and user satisfaction is crucial,” the study team concludes.
The study was supported by the Xiamen Medical Health Science and Technology Project and the Xiamen Chang Gung Hospital Science Project. The authors have declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An artificial intelligence (AI) tool that runs on a smartphone can help patients scheduled for a colonoscopy evaluate independently how well they do with bowel cleansing and may be an alternative approach for evaluating bowel preparation quality before the colonoscopy, especially in the COVID-19 era.
The AI tool is a “manpower-saving” option that reduces the need for nurses to evaluate the quality of bowel preparation, say Wei Gong, MD, Southern Medical University, Shenzhen, China, and colleagues.
Having the tool on a patient’s smartphone means caregivers and nurses would not be required to assess the adequacy of bowel cleansing for patients, which, in turn, would reduce person-to-person contact and the spread of infectious diseases, they add.
The study was published online in the American Journal of Gastroenterology.
Better than do-it-yourself evaluation?
The study was conducted at two hospitals in China among consecutive patients prepping for colonoscopy. All participants received standard bowel preparation instructions and were given a leaflet with general guidelines on bowel preparation.
The leaflet included photos representing bowel preparation quality and informed patients that their stool should eventually be a yellowish clear liquid; if any cloudiness (including turbid liquid, particles, or small amounts of feces) is observed in the liquid stool, the bowel preparation is not complete.
All patients were prescribed standard polyethylene glycol electrolyte solution for bowel cleansing 4-6 hours before the colonoscopy.
After consuming the solution, all patients scanned a QR (quick response) code with a smartphone for randomization into an experimental group using the AI-convolutional neural network (AI-CNN) model or a control group using self-evaluation.
The system gave instructions for using the application, taking photos of their feces, and uploading the images.
After uploading the images, the 730 patients in the AI-CNN group automatically received a “pass” or “not pass” alert, which indicated whether their bowel preparation was adequate or not.
The 704 patients in the control group evaluated the adequacy of bowel preparation on their own according to the leaflet instructions after uploading their images.
Colonoscopists and nurses were blinded to the bowel evaluation method that each patient used.
According to the investigators, evaluation results (“pass” or “not pass”) in terms of adequacy of bowel preparation as represented by Boston Bowel Preparation Scale (BBPS) scores were consistent between the two methods (AI-CNN or self-evaluation).
Overall, there were no significant differences in the two methods in terms of mean BBPS scores, polyp detection rate, or adenoma detection rate.
In subgroup analysis, however, the mean BBPS score of patients with “pass” results was significantly higher in the AI-CNN group than in the self-evaluation control group.
This suggests that the AI-CNN model may further improve the quality of bowel preparation in patients exhibiting adequate bowel preparation, the researchers say.
The results also suggest improved bowel preparation quality of the right colon under the aid of the AI-CNN model, which may be crucial for the prevention of interval colorectal cancer.
The study did not investigate the user acceptability of the AI-CNN model.
“To improve the model and broaden its application in routine practice, evaluating its convenience, accessibility, aspects that cause users difficulty, and user satisfaction is crucial,” the study team concludes.
The study was supported by the Xiamen Medical Health Science and Technology Project and the Xiamen Chang Gung Hospital Science Project. The authors have declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An artificial intelligence (AI) tool that runs on a smartphone can help patients scheduled for a colonoscopy evaluate independently how well they do with bowel cleansing and may be an alternative approach for evaluating bowel preparation quality before the colonoscopy, especially in the COVID-19 era.
The AI tool is a “manpower-saving” option that reduces the need for nurses to evaluate the quality of bowel preparation, say Wei Gong, MD, Southern Medical University, Shenzhen, China, and colleagues.
Having the tool on a patient’s smartphone means caregivers and nurses would not be required to assess the adequacy of bowel cleansing for patients, which, in turn, would reduce person-to-person contact and the spread of infectious diseases, they add.
The study was published online in the American Journal of Gastroenterology.
Better than do-it-yourself evaluation?
The study was conducted at two hospitals in China among consecutive patients prepping for colonoscopy. All participants received standard bowel preparation instructions and were given a leaflet with general guidelines on bowel preparation.
The leaflet included photos representing bowel preparation quality and informed patients that their stool should eventually be a yellowish clear liquid; if any cloudiness (including turbid liquid, particles, or small amounts of feces) is observed in the liquid stool, the bowel preparation is not complete.
All patients were prescribed standard polyethylene glycol electrolyte solution for bowel cleansing 4-6 hours before the colonoscopy.
After consuming the solution, all patients scanned a QR (quick response) code with a smartphone for randomization into an experimental group using the AI-convolutional neural network (AI-CNN) model or a control group using self-evaluation.
The system gave instructions for using the application, taking photos of their feces, and uploading the images.
After uploading the images, the 730 patients in the AI-CNN group automatically received a “pass” or “not pass” alert, which indicated whether their bowel preparation was adequate or not.
The 704 patients in the control group evaluated the adequacy of bowel preparation on their own according to the leaflet instructions after uploading their images.
Colonoscopists and nurses were blinded to the bowel evaluation method that each patient used.
According to the investigators, evaluation results (“pass” or “not pass”) in terms of adequacy of bowel preparation as represented by Boston Bowel Preparation Scale (BBPS) scores were consistent between the two methods (AI-CNN or self-evaluation).
Overall, there were no significant differences in the two methods in terms of mean BBPS scores, polyp detection rate, or adenoma detection rate.
In subgroup analysis, however, the mean BBPS score of patients with “pass” results was significantly higher in the AI-CNN group than in the self-evaluation control group.
This suggests that the AI-CNN model may further improve the quality of bowel preparation in patients exhibiting adequate bowel preparation, the researchers say.
The results also suggest improved bowel preparation quality of the right colon under the aid of the AI-CNN model, which may be crucial for the prevention of interval colorectal cancer.
The study did not investigate the user acceptability of the AI-CNN model.
“To improve the model and broaden its application in routine practice, evaluating its convenience, accessibility, aspects that cause users difficulty, and user satisfaction is crucial,” the study team concludes.
The study was supported by the Xiamen Medical Health Science and Technology Project and the Xiamen Chang Gung Hospital Science Project. The authors have declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
Metabolic syndrome raises dementia risk in under-60s
The more components of metabolic syndrome a person has in midlife seems to raise their risk of dementia, although that relationship seems to go away after age 70, a post hoc analysis of data from a major European cohort study has found.
A team of European researchers reported online in the journal Diabetes Care that the follow-up of the Whitehall II cohort study, a study of more than 10,000 civil servants in London that was established in the late 1980s, also found that cardiovascular disease (CVD) may only partially contribute to the risk of dementia in study participants.
They found that each additional metabolic syndrome component before age 60 years was linked to a 13% rise in the risk of dementia (hazard ratio, 1.13; 95% confidence interval [CI], 1.05-1.23) and, from age 60 to 70, the risk rose 8% (HR, 1.08; 95% CI, 1.00-1.16). However, in people aged 70 years and older, the relationship wasn’t statistically significant (HR, 1.04; 95% CI, 0.96-1.13]).
The study used “the latest harmonized definition” of metabolic syndrome; that is, participants were classified as having metabolic syndrome if they had three or more of the five components. As lead author Marcos D. Machado-Fragua, PhD, noted in an email interview, those components are abdominal obesity, high triglycerides, low HDL cholesterol levels, high blood pressure, and high fasting glucose.
“Our research question was on the association between metabolic syndrome and late-life dementia. We found that the presence of one metabolic syndrome component and the presence of metabolic risk before age 60, but not after, is associated with higher risk of dementia,” said Dr. Machado-Fragua, a post-doctoral researcher at the French Institute for Health and Medical Research in Paris.
The study cohort consisted of 10,308 London-based civil servants aged 35-55 years. Every 4-5 years after enrollment, from 1991 through 2016, they completed a questionnaire and had a clinical examination. The U.K. National Health Service electronic health record system tracked outcomes for all but 10 participants through March 2019.
The study identified the individual metabolic syndrome components that posed the highest risk for dementia in these three age groups:
- Age < 60 years: elevated waist circumference (HR 1.39 [95% CI 1.07, 1.81]), low HDL-C, (HR 1.30 [95% CI 1.02, 1.66]), and elevated blood pressure (HR 1.34 [95% CI 1.09, 1.63]).
- Age 60-70 years: low HDL-C (HR 1.26 [95% CI 1.02, 1.57]) and elevated fasting glucose (HR 1.40 [95% CI 1.12, 1.74]).
- Age >70 years: elevated fasting glucose (HR 1.38 [95% CI 1.07, 1.79]).
The study found that the dementia risk was significantly high in study participants under age 60 who had at least one (HR 1.99 [95% CI 1.08, 3.66]) or two (HR 1.69 [95% CI 1.12, 2.56]) metabolic syndrome components even when they didn’t have CVD.
“The present study adds to the understanding of the association between metabolic syndrome and dementia due to three novel features,” Dr. Machado-Fragua said. “First, we tested alternative thresholds to define ‘high metabolic risk,’ and findings show increased risk of dementia to start with the presence of one metabolic syndrome component. Second, assessment of metabolic syndrome components in midlife and later life allowed the examination of the role of age at prevalence of metabolic risk for incident dementia at older ages. Third, our findings showed high dementia risk in those free of cardiovascular disease during follow-up, suggesting that the association between high metabolic risk and incident dementia is not fully explained by cardiovascular disease.”
Dr. Machado-Fragua added, “For now, a cure for dementia remains elusive, making it important to think of prevention strategies. Our findings support targeting the components of the metabolic syndrome in midlife, even in those who have fewer than three of the metabolic syndrome components.”
Applicability ‘confusing’
In an interview, Yehuda Handelsman, MD, questioned the applicability of the study findings in the clinic. “Metabolic syndrome is a clinical manifestation of insulin resistance,” he said. “The more metabolic syndrome criteria a person has, the more insulin resistant that person will be. There is literature that is [suggesting] that insulin resistance is an important cause of dementia.”
The finding of a higher dementia risk before age 70, compared to afterward, makes the applicability “even more confusing,” he said. The results are even more muddled for U.S. physicians, who have moved away from the term metabolic syndrome in favor of cardiometabolic syndrome, said Dr. Handelsman, medical director and principal investigator at the Metabolic Institute of America and president of the Diabetes CardioRenal & Metabolism Institute, both in Tarzana, Calif.
Confusion also surrounds one of the components of metabolic syndrome: Waist circumference, per the harmonized definition the study used, and body mass index, which the more traditional definition uses.
Nonetheless, metabolic syndrome can be used as “kind of a risk calculator” for CVD, diabetes, and dementia, he said. One strength of the study, Dr. Handelsman said, is its size and scope, following 28 years of data. But a weakness was its observational design. “It doesn’t evaluate any true intervention to modify risk,” he said.
Dr. Machado-Fragua and coauthors have no disclosures.
The more components of metabolic syndrome a person has in midlife seems to raise their risk of dementia, although that relationship seems to go away after age 70, a post hoc analysis of data from a major European cohort study has found.
A team of European researchers reported online in the journal Diabetes Care that the follow-up of the Whitehall II cohort study, a study of more than 10,000 civil servants in London that was established in the late 1980s, also found that cardiovascular disease (CVD) may only partially contribute to the risk of dementia in study participants.
They found that each additional metabolic syndrome component before age 60 years was linked to a 13% rise in the risk of dementia (hazard ratio, 1.13; 95% confidence interval [CI], 1.05-1.23) and, from age 60 to 70, the risk rose 8% (HR, 1.08; 95% CI, 1.00-1.16). However, in people aged 70 years and older, the relationship wasn’t statistically significant (HR, 1.04; 95% CI, 0.96-1.13]).
The study used “the latest harmonized definition” of metabolic syndrome; that is, participants were classified as having metabolic syndrome if they had three or more of the five components. As lead author Marcos D. Machado-Fragua, PhD, noted in an email interview, those components are abdominal obesity, high triglycerides, low HDL cholesterol levels, high blood pressure, and high fasting glucose.
“Our research question was on the association between metabolic syndrome and late-life dementia. We found that the presence of one metabolic syndrome component and the presence of metabolic risk before age 60, but not after, is associated with higher risk of dementia,” said Dr. Machado-Fragua, a post-doctoral researcher at the French Institute for Health and Medical Research in Paris.
The study cohort consisted of 10,308 London-based civil servants aged 35-55 years. Every 4-5 years after enrollment, from 1991 through 2016, they completed a questionnaire and had a clinical examination. The U.K. National Health Service electronic health record system tracked outcomes for all but 10 participants through March 2019.
The study identified the individual metabolic syndrome components that posed the highest risk for dementia in these three age groups:
- Age < 60 years: elevated waist circumference (HR 1.39 [95% CI 1.07, 1.81]), low HDL-C, (HR 1.30 [95% CI 1.02, 1.66]), and elevated blood pressure (HR 1.34 [95% CI 1.09, 1.63]).
- Age 60-70 years: low HDL-C (HR 1.26 [95% CI 1.02, 1.57]) and elevated fasting glucose (HR 1.40 [95% CI 1.12, 1.74]).
- Age >70 years: elevated fasting glucose (HR 1.38 [95% CI 1.07, 1.79]).
The study found that the dementia risk was significantly high in study participants under age 60 who had at least one (HR 1.99 [95% CI 1.08, 3.66]) or two (HR 1.69 [95% CI 1.12, 2.56]) metabolic syndrome components even when they didn’t have CVD.
“The present study adds to the understanding of the association between metabolic syndrome and dementia due to three novel features,” Dr. Machado-Fragua said. “First, we tested alternative thresholds to define ‘high metabolic risk,’ and findings show increased risk of dementia to start with the presence of one metabolic syndrome component. Second, assessment of metabolic syndrome components in midlife and later life allowed the examination of the role of age at prevalence of metabolic risk for incident dementia at older ages. Third, our findings showed high dementia risk in those free of cardiovascular disease during follow-up, suggesting that the association between high metabolic risk and incident dementia is not fully explained by cardiovascular disease.”
Dr. Machado-Fragua added, “For now, a cure for dementia remains elusive, making it important to think of prevention strategies. Our findings support targeting the components of the metabolic syndrome in midlife, even in those who have fewer than three of the metabolic syndrome components.”
Applicability ‘confusing’
In an interview, Yehuda Handelsman, MD, questioned the applicability of the study findings in the clinic. “Metabolic syndrome is a clinical manifestation of insulin resistance,” he said. “The more metabolic syndrome criteria a person has, the more insulin resistant that person will be. There is literature that is [suggesting] that insulin resistance is an important cause of dementia.”
The finding of a higher dementia risk before age 70, compared to afterward, makes the applicability “even more confusing,” he said. The results are even more muddled for U.S. physicians, who have moved away from the term metabolic syndrome in favor of cardiometabolic syndrome, said Dr. Handelsman, medical director and principal investigator at the Metabolic Institute of America and president of the Diabetes CardioRenal & Metabolism Institute, both in Tarzana, Calif.
Confusion also surrounds one of the components of metabolic syndrome: Waist circumference, per the harmonized definition the study used, and body mass index, which the more traditional definition uses.
Nonetheless, metabolic syndrome can be used as “kind of a risk calculator” for CVD, diabetes, and dementia, he said. One strength of the study, Dr. Handelsman said, is its size and scope, following 28 years of data. But a weakness was its observational design. “It doesn’t evaluate any true intervention to modify risk,” he said.
Dr. Machado-Fragua and coauthors have no disclosures.
The more components of metabolic syndrome a person has in midlife seems to raise their risk of dementia, although that relationship seems to go away after age 70, a post hoc analysis of data from a major European cohort study has found.
A team of European researchers reported online in the journal Diabetes Care that the follow-up of the Whitehall II cohort study, a study of more than 10,000 civil servants in London that was established in the late 1980s, also found that cardiovascular disease (CVD) may only partially contribute to the risk of dementia in study participants.
They found that each additional metabolic syndrome component before age 60 years was linked to a 13% rise in the risk of dementia (hazard ratio, 1.13; 95% confidence interval [CI], 1.05-1.23) and, from age 60 to 70, the risk rose 8% (HR, 1.08; 95% CI, 1.00-1.16). However, in people aged 70 years and older, the relationship wasn’t statistically significant (HR, 1.04; 95% CI, 0.96-1.13]).
The study used “the latest harmonized definition” of metabolic syndrome; that is, participants were classified as having metabolic syndrome if they had three or more of the five components. As lead author Marcos D. Machado-Fragua, PhD, noted in an email interview, those components are abdominal obesity, high triglycerides, low HDL cholesterol levels, high blood pressure, and high fasting glucose.
“Our research question was on the association between metabolic syndrome and late-life dementia. We found that the presence of one metabolic syndrome component and the presence of metabolic risk before age 60, but not after, is associated with higher risk of dementia,” said Dr. Machado-Fragua, a post-doctoral researcher at the French Institute for Health and Medical Research in Paris.
The study cohort consisted of 10,308 London-based civil servants aged 35-55 years. Every 4-5 years after enrollment, from 1991 through 2016, they completed a questionnaire and had a clinical examination. The U.K. National Health Service electronic health record system tracked outcomes for all but 10 participants through March 2019.
The study identified the individual metabolic syndrome components that posed the highest risk for dementia in these three age groups:
- Age < 60 years: elevated waist circumference (HR 1.39 [95% CI 1.07, 1.81]), low HDL-C, (HR 1.30 [95% CI 1.02, 1.66]), and elevated blood pressure (HR 1.34 [95% CI 1.09, 1.63]).
- Age 60-70 years: low HDL-C (HR 1.26 [95% CI 1.02, 1.57]) and elevated fasting glucose (HR 1.40 [95% CI 1.12, 1.74]).
- Age >70 years: elevated fasting glucose (HR 1.38 [95% CI 1.07, 1.79]).
The study found that the dementia risk was significantly high in study participants under age 60 who had at least one (HR 1.99 [95% CI 1.08, 3.66]) or two (HR 1.69 [95% CI 1.12, 2.56]) metabolic syndrome components even when they didn’t have CVD.
“The present study adds to the understanding of the association between metabolic syndrome and dementia due to three novel features,” Dr. Machado-Fragua said. “First, we tested alternative thresholds to define ‘high metabolic risk,’ and findings show increased risk of dementia to start with the presence of one metabolic syndrome component. Second, assessment of metabolic syndrome components in midlife and later life allowed the examination of the role of age at prevalence of metabolic risk for incident dementia at older ages. Third, our findings showed high dementia risk in those free of cardiovascular disease during follow-up, suggesting that the association between high metabolic risk and incident dementia is not fully explained by cardiovascular disease.”
Dr. Machado-Fragua added, “For now, a cure for dementia remains elusive, making it important to think of prevention strategies. Our findings support targeting the components of the metabolic syndrome in midlife, even in those who have fewer than three of the metabolic syndrome components.”
Applicability ‘confusing’
In an interview, Yehuda Handelsman, MD, questioned the applicability of the study findings in the clinic. “Metabolic syndrome is a clinical manifestation of insulin resistance,” he said. “The more metabolic syndrome criteria a person has, the more insulin resistant that person will be. There is literature that is [suggesting] that insulin resistance is an important cause of dementia.”
The finding of a higher dementia risk before age 70, compared to afterward, makes the applicability “even more confusing,” he said. The results are even more muddled for U.S. physicians, who have moved away from the term metabolic syndrome in favor of cardiometabolic syndrome, said Dr. Handelsman, medical director and principal investigator at the Metabolic Institute of America and president of the Diabetes CardioRenal & Metabolism Institute, both in Tarzana, Calif.
Confusion also surrounds one of the components of metabolic syndrome: Waist circumference, per the harmonized definition the study used, and body mass index, which the more traditional definition uses.
Nonetheless, metabolic syndrome can be used as “kind of a risk calculator” for CVD, diabetes, and dementia, he said. One strength of the study, Dr. Handelsman said, is its size and scope, following 28 years of data. But a weakness was its observational design. “It doesn’t evaluate any true intervention to modify risk,” he said.
Dr. Machado-Fragua and coauthors have no disclosures.
FROM DIABETES CARE
Children and COVID: Many parents see vaccine as the greater risk
New COVID-19 cases rose for the second week in a row as cumulative cases among U.S. children passed the 14-million mark, but a recent survey shows that more than half of parents believe that the vaccine is a greater risk to children under age 5 years than the virus.
In a Kaiser Family Foundation survey conducted July 7-17, 53% of parents with children aged 6 months to 5 years said that the vaccine is “a bigger risk to their child’s health than getting infected with COVID-19, compared to 44% who say getting infected is the bigger risk,” KFF reported July 26.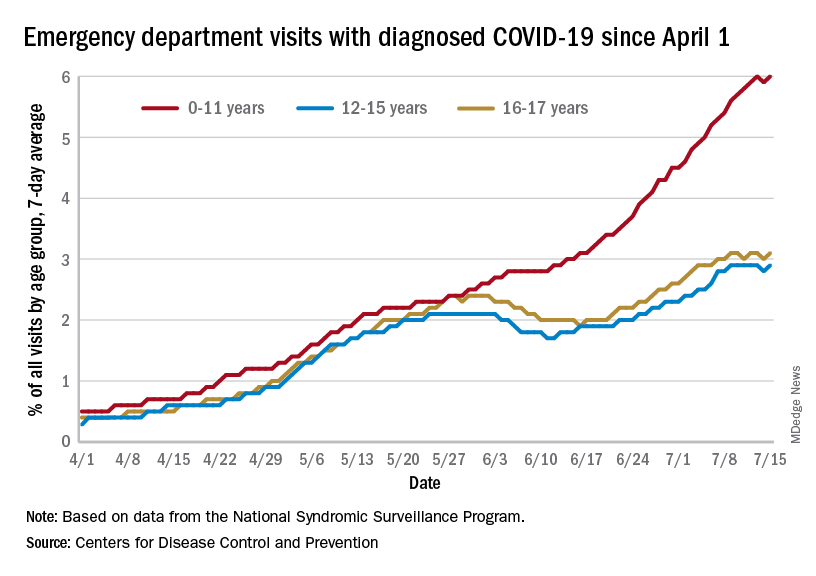
More than 4 out of 10 of respondents (43%) said that they will “definitely not” get their eligible children vaccinated, while only 7% said that their children had already received it and 10% said their children would get it as soon as possible, according to the KFF survey, which had an overall sample size of 1,847 adults, including an oversample of 471 parents of children under age 5.
Vaccine initiation has been slow in the first month since it was approved for the youngest children. Just 2.8% of all eligible children under age 5 had received an initial dose as of July 19, compared with first-month uptake figures of more than 18% for the 5- to 11-year-olds and 27% for those aged 12-15, based on data from the Centers for Disease Control and Prevention.
The current rates for vaccination in those aged 5 and older look like this: 70.2% of 12- to 17-year-olds have received at least one dose, versus 37.1% of those aged 5-11. Just over 60% of the older children were fully vaccinated as of July 19, as were 30.2% of the 5- to 11-year-olds, the CDC reported on its COVID Data Tracker.
Number of new cases hits 2-month high
Despite the vaccine, SARS-CoV-2 and its various mutations have continued with their summer travels. With 92,000 newly infected children added for the week of July 15-21, there have now been a total of 14,003,497 pediatric cases reported since the start of the pandemic, which works out to 18.6% of cases in all ages, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
The 92,000 new cases represent an increase of almost 22% over the previous week and mark the highest 1-week count since May, when the total passed 100,000 for 2 consecutive weeks. More recently the trend had seemed more stable as weekly cases dropped twice and rose twice as the total hovered around 70,000, based on the data collected by the AAP and CHA from state and territorial health departments.
A different scenario has played out for emergency department visits and hospital admissions, which have risen steadily since the beginning of April. The admission rate for children aged 0-17, which was just 0.13 new patients per 100,000 population on April 11, was up to 0.44 per 100,000 on July 21. By comparison, the highest rate reached last year during the Delta surge was 0.47 per 100,000, based on CDC data.
The 7-day average of emergency dept. visits among the youngest age group, 0-11 years, shows the same general increase as hospital admissions, but the older children have diverged form that path (see graph). For those aged 12-15 and 16-17, hospitalizations started dropping in late May and into mid-June before climbing again, although more slowly than for the youngest group, the CDC data show.
The ED visit rate with diagnosed COVID among those aged 0-11, measured at 6.1% of all visits on July 19, is, in fact, considerably higher than at any time during the Delta surge last year, when it never passed 4.0%, although much lower than peak Omicron (14.1%). That 6.1% was also higher than any other age group on that day, adults included, the CDC said.
New COVID-19 cases rose for the second week in a row as cumulative cases among U.S. children passed the 14-million mark, but a recent survey shows that more than half of parents believe that the vaccine is a greater risk to children under age 5 years than the virus.
In a Kaiser Family Foundation survey conducted July 7-17, 53% of parents with children aged 6 months to 5 years said that the vaccine is “a bigger risk to their child’s health than getting infected with COVID-19, compared to 44% who say getting infected is the bigger risk,” KFF reported July 26.
More than 4 out of 10 of respondents (43%) said that they will “definitely not” get their eligible children vaccinated, while only 7% said that their children had already received it and 10% said their children would get it as soon as possible, according to the KFF survey, which had an overall sample size of 1,847 adults, including an oversample of 471 parents of children under age 5.
Vaccine initiation has been slow in the first month since it was approved for the youngest children. Just 2.8% of all eligible children under age 5 had received an initial dose as of July 19, compared with first-month uptake figures of more than 18% for the 5- to 11-year-olds and 27% for those aged 12-15, based on data from the Centers for Disease Control and Prevention.
The current rates for vaccination in those aged 5 and older look like this: 70.2% of 12- to 17-year-olds have received at least one dose, versus 37.1% of those aged 5-11. Just over 60% of the older children were fully vaccinated as of July 19, as were 30.2% of the 5- to 11-year-olds, the CDC reported on its COVID Data Tracker.
Number of new cases hits 2-month high
Despite the vaccine, SARS-CoV-2 and its various mutations have continued with their summer travels. With 92,000 newly infected children added for the week of July 15-21, there have now been a total of 14,003,497 pediatric cases reported since the start of the pandemic, which works out to 18.6% of cases in all ages, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
The 92,000 new cases represent an increase of almost 22% over the previous week and mark the highest 1-week count since May, when the total passed 100,000 for 2 consecutive weeks. More recently the trend had seemed more stable as weekly cases dropped twice and rose twice as the total hovered around 70,000, based on the data collected by the AAP and CHA from state and territorial health departments.
A different scenario has played out for emergency department visits and hospital admissions, which have risen steadily since the beginning of April. The admission rate for children aged 0-17, which was just 0.13 new patients per 100,000 population on April 11, was up to 0.44 per 100,000 on July 21. By comparison, the highest rate reached last year during the Delta surge was 0.47 per 100,000, based on CDC data.
The 7-day average of emergency dept. visits among the youngest age group, 0-11 years, shows the same general increase as hospital admissions, but the older children have diverged form that path (see graph). For those aged 12-15 and 16-17, hospitalizations started dropping in late May and into mid-June before climbing again, although more slowly than for the youngest group, the CDC data show.
The ED visit rate with diagnosed COVID among those aged 0-11, measured at 6.1% of all visits on July 19, is, in fact, considerably higher than at any time during the Delta surge last year, when it never passed 4.0%, although much lower than peak Omicron (14.1%). That 6.1% was also higher than any other age group on that day, adults included, the CDC said.
New COVID-19 cases rose for the second week in a row as cumulative cases among U.S. children passed the 14-million mark, but a recent survey shows that more than half of parents believe that the vaccine is a greater risk to children under age 5 years than the virus.
In a Kaiser Family Foundation survey conducted July 7-17, 53% of parents with children aged 6 months to 5 years said that the vaccine is “a bigger risk to their child’s health than getting infected with COVID-19, compared to 44% who say getting infected is the bigger risk,” KFF reported July 26.
More than 4 out of 10 of respondents (43%) said that they will “definitely not” get their eligible children vaccinated, while only 7% said that their children had already received it and 10% said their children would get it as soon as possible, according to the KFF survey, which had an overall sample size of 1,847 adults, including an oversample of 471 parents of children under age 5.
Vaccine initiation has been slow in the first month since it was approved for the youngest children. Just 2.8% of all eligible children under age 5 had received an initial dose as of July 19, compared with first-month uptake figures of more than 18% for the 5- to 11-year-olds and 27% for those aged 12-15, based on data from the Centers for Disease Control and Prevention.
The current rates for vaccination in those aged 5 and older look like this: 70.2% of 12- to 17-year-olds have received at least one dose, versus 37.1% of those aged 5-11. Just over 60% of the older children were fully vaccinated as of July 19, as were 30.2% of the 5- to 11-year-olds, the CDC reported on its COVID Data Tracker.
Number of new cases hits 2-month high
Despite the vaccine, SARS-CoV-2 and its various mutations have continued with their summer travels. With 92,000 newly infected children added for the week of July 15-21, there have now been a total of 14,003,497 pediatric cases reported since the start of the pandemic, which works out to 18.6% of cases in all ages, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report.
The 92,000 new cases represent an increase of almost 22% over the previous week and mark the highest 1-week count since May, when the total passed 100,000 for 2 consecutive weeks. More recently the trend had seemed more stable as weekly cases dropped twice and rose twice as the total hovered around 70,000, based on the data collected by the AAP and CHA from state and territorial health departments.
A different scenario has played out for emergency department visits and hospital admissions, which have risen steadily since the beginning of April. The admission rate for children aged 0-17, which was just 0.13 new patients per 100,000 population on April 11, was up to 0.44 per 100,000 on July 21. By comparison, the highest rate reached last year during the Delta surge was 0.47 per 100,000, based on CDC data.
The 7-day average of emergency dept. visits among the youngest age group, 0-11 years, shows the same general increase as hospital admissions, but the older children have diverged form that path (see graph). For those aged 12-15 and 16-17, hospitalizations started dropping in late May and into mid-June before climbing again, although more slowly than for the youngest group, the CDC data show.
The ED visit rate with diagnosed COVID among those aged 0-11, measured at 6.1% of all visits on July 19, is, in fact, considerably higher than at any time during the Delta surge last year, when it never passed 4.0%, although much lower than peak Omicron (14.1%). That 6.1% was also higher than any other age group on that day, adults included, the CDC said.
Boosting hypertension screening, treatment would cut global mortality 7%
If 80% of individuals with hypertension were screened, 80% received treatment, and 80% then reached guideline-specified targets, up to 200 million cases of cardiovascular disease (CVD) and 130 million deaths could be averted by 2050, a modeling study suggests.
Achievement of the 80-80-80 target “could be one of the single most important global public health accomplishments of the coming decades,” according to the authors.
“We need to reprioritize hypertension care in our practices,” principal investigator David A. Watkins, MD, MPH, University of Washington, Seattle, told this news organization. “Only about one in five persons with hypertension around the world has their blood pressure well controlled. Oftentimes, clinicians are focused on addressing patients’ other health needs, many of which can be pressing in the short term, and we forget to talk about blood pressure, which has more than earned its reputation as ‘the silent killer.’ ”
The modeling study was published online in Nature Medicine, with lead author Sarah J. Pickersgill, MPH, also from the University of Washington.
Two interventions, three scenarios
Dr. Watkins and colleagues based their analysis on two approaches to blood pressure (BP) control shown to be beneficial: drug treatment to a systolic BP of either 130 mm Hg or 140 mm Hg or less, depending on local guidelines, and dietary sodium reduction, as recommended by the World Health Organization.
The team modeled the impacts of these interventions in 182 countries according to three scenarios:
- Business as usual (control): allowing hypertension to increase at historic rates of change and mean sodium intake to remain at current levels
- Progress: matching historically high-performing countries (for example, accelerating hypertension control by about 3% per year at intermediate levels of intervention coverage) while lowering mean sodium intake by 15% by 2030
- Aspirational: hypertension control achieved faster than historically high-performing countries (about 4% per year) and mean sodium intake decreased by 30% by 2027
The analysis suggests that in the progressive scenario, all countries could achieve 80-80-80 targets by 2050 and most countries by 2040; the aspirational scenario would have all countries meeting them by 2040. That would result in reductions in all-cause mortality of 4%-7% (76 million to 130 million deaths averted) with progressive and aspirational interventions, respectively, compared with the control scenario.
There would also be a slower rise in expected CVD from population growth and aging (110 million to 200 million cases averted). That is, the probability of dying from any CVD cause between the ages of 30 and 80 years would be reduced by 16% in the progressive scenario and 26% in the aspirational scenario.
Of note, about 83%-85% of the potential mortality reductions would result from scaling up hypertension treatment in the progressive and aspirational scenarios, respectively, with the remaining 15%-17% coming from sodium reduction, the researchers state.
Further, they propose, scaling up BP interventions could reduce CVD inequalities across countries, with low-income and lower-middle-income countries likely experiencing the largest reductions in disease rates and mortality.
Implementation barriers
“Health systems in many low- and middle-income countries have not traditionally been set up to succeed in chronic disease management in primary care,” Dr. Watkins noted. For interventions to be successful, he said, “several barriers need to be addressed, including: low population awareness of chronic diseases like hypertension and diabetes, which leads to low rates of screening and treatment; high out-of-pocket cost and low availability of medicines for chronic diseases; and need for adherence support and provider incentives for improving quality of chronic disease care in primary care settings.”
“Based on the analysis, achieving the 80-80-80 seems feasible, though actually getting there may be much more complicated. I wonder whether countries have the resources to implement the needed policies,” Rodrigo M. Carrillo-Larco, MD, researcher, department of epidemiology and biostatistics, School of Public Health, Imperial College London, told this news organization.
“It may be challenging, particularly after COVID-19, which revealed deficiencies in many health care systems, and care for hypertension may have been disturbed,” said Dr. Carrillo-Larco, who is not connected with the analysis.
That said, simplified BP screening approaches could help maximize the number of people screened overall, potentially identifying those with hypertension and raising awareness, he proposed. His team’s recent study showed that such approaches vary from country to country but are generally reliable and can be used effectively for population screening.
In addition, Dr. Carrillo-Larco said, any efforts by clinicians to improve adherence and help patients achieve BP control “would also have positive effects at the population level.”
The study was supported by a grant from the Bill & Melinda Gates Foundation, with additional funding by a grant to Dr. Watkins from Resolve to Save Lives. No conflicts of interest were declared.
A version of this article first appeared on Medscape.com.
If 80% of individuals with hypertension were screened, 80% received treatment, and 80% then reached guideline-specified targets, up to 200 million cases of cardiovascular disease (CVD) and 130 million deaths could be averted by 2050, a modeling study suggests.
Achievement of the 80-80-80 target “could be one of the single most important global public health accomplishments of the coming decades,” according to the authors.
“We need to reprioritize hypertension care in our practices,” principal investigator David A. Watkins, MD, MPH, University of Washington, Seattle, told this news organization. “Only about one in five persons with hypertension around the world has their blood pressure well controlled. Oftentimes, clinicians are focused on addressing patients’ other health needs, many of which can be pressing in the short term, and we forget to talk about blood pressure, which has more than earned its reputation as ‘the silent killer.’ ”
The modeling study was published online in Nature Medicine, with lead author Sarah J. Pickersgill, MPH, also from the University of Washington.
Two interventions, three scenarios
Dr. Watkins and colleagues based their analysis on two approaches to blood pressure (BP) control shown to be beneficial: drug treatment to a systolic BP of either 130 mm Hg or 140 mm Hg or less, depending on local guidelines, and dietary sodium reduction, as recommended by the World Health Organization.
The team modeled the impacts of these interventions in 182 countries according to three scenarios:
- Business as usual (control): allowing hypertension to increase at historic rates of change and mean sodium intake to remain at current levels
- Progress: matching historically high-performing countries (for example, accelerating hypertension control by about 3% per year at intermediate levels of intervention coverage) while lowering mean sodium intake by 15% by 2030
- Aspirational: hypertension control achieved faster than historically high-performing countries (about 4% per year) and mean sodium intake decreased by 30% by 2027
The analysis suggests that in the progressive scenario, all countries could achieve 80-80-80 targets by 2050 and most countries by 2040; the aspirational scenario would have all countries meeting them by 2040. That would result in reductions in all-cause mortality of 4%-7% (76 million to 130 million deaths averted) with progressive and aspirational interventions, respectively, compared with the control scenario.
There would also be a slower rise in expected CVD from population growth and aging (110 million to 200 million cases averted). That is, the probability of dying from any CVD cause between the ages of 30 and 80 years would be reduced by 16% in the progressive scenario and 26% in the aspirational scenario.
Of note, about 83%-85% of the potential mortality reductions would result from scaling up hypertension treatment in the progressive and aspirational scenarios, respectively, with the remaining 15%-17% coming from sodium reduction, the researchers state.
Further, they propose, scaling up BP interventions could reduce CVD inequalities across countries, with low-income and lower-middle-income countries likely experiencing the largest reductions in disease rates and mortality.
Implementation barriers
“Health systems in many low- and middle-income countries have not traditionally been set up to succeed in chronic disease management in primary care,” Dr. Watkins noted. For interventions to be successful, he said, “several barriers need to be addressed, including: low population awareness of chronic diseases like hypertension and diabetes, which leads to low rates of screening and treatment; high out-of-pocket cost and low availability of medicines for chronic diseases; and need for adherence support and provider incentives for improving quality of chronic disease care in primary care settings.”
“Based on the analysis, achieving the 80-80-80 seems feasible, though actually getting there may be much more complicated. I wonder whether countries have the resources to implement the needed policies,” Rodrigo M. Carrillo-Larco, MD, researcher, department of epidemiology and biostatistics, School of Public Health, Imperial College London, told this news organization.
“It may be challenging, particularly after COVID-19, which revealed deficiencies in many health care systems, and care for hypertension may have been disturbed,” said Dr. Carrillo-Larco, who is not connected with the analysis.
That said, simplified BP screening approaches could help maximize the number of people screened overall, potentially identifying those with hypertension and raising awareness, he proposed. His team’s recent study showed that such approaches vary from country to country but are generally reliable and can be used effectively for population screening.
In addition, Dr. Carrillo-Larco said, any efforts by clinicians to improve adherence and help patients achieve BP control “would also have positive effects at the population level.”
The study was supported by a grant from the Bill & Melinda Gates Foundation, with additional funding by a grant to Dr. Watkins from Resolve to Save Lives. No conflicts of interest were declared.
A version of this article first appeared on Medscape.com.
If 80% of individuals with hypertension were screened, 80% received treatment, and 80% then reached guideline-specified targets, up to 200 million cases of cardiovascular disease (CVD) and 130 million deaths could be averted by 2050, a modeling study suggests.
Achievement of the 80-80-80 target “could be one of the single most important global public health accomplishments of the coming decades,” according to the authors.
“We need to reprioritize hypertension care in our practices,” principal investigator David A. Watkins, MD, MPH, University of Washington, Seattle, told this news organization. “Only about one in five persons with hypertension around the world has their blood pressure well controlled. Oftentimes, clinicians are focused on addressing patients’ other health needs, many of which can be pressing in the short term, and we forget to talk about blood pressure, which has more than earned its reputation as ‘the silent killer.’ ”
The modeling study was published online in Nature Medicine, with lead author Sarah J. Pickersgill, MPH, also from the University of Washington.
Two interventions, three scenarios
Dr. Watkins and colleagues based their analysis on two approaches to blood pressure (BP) control shown to be beneficial: drug treatment to a systolic BP of either 130 mm Hg or 140 mm Hg or less, depending on local guidelines, and dietary sodium reduction, as recommended by the World Health Organization.
The team modeled the impacts of these interventions in 182 countries according to three scenarios:
- Business as usual (control): allowing hypertension to increase at historic rates of change and mean sodium intake to remain at current levels
- Progress: matching historically high-performing countries (for example, accelerating hypertension control by about 3% per year at intermediate levels of intervention coverage) while lowering mean sodium intake by 15% by 2030
- Aspirational: hypertension control achieved faster than historically high-performing countries (about 4% per year) and mean sodium intake decreased by 30% by 2027
The analysis suggests that in the progressive scenario, all countries could achieve 80-80-80 targets by 2050 and most countries by 2040; the aspirational scenario would have all countries meeting them by 2040. That would result in reductions in all-cause mortality of 4%-7% (76 million to 130 million deaths averted) with progressive and aspirational interventions, respectively, compared with the control scenario.
There would also be a slower rise in expected CVD from population growth and aging (110 million to 200 million cases averted). That is, the probability of dying from any CVD cause between the ages of 30 and 80 years would be reduced by 16% in the progressive scenario and 26% in the aspirational scenario.
Of note, about 83%-85% of the potential mortality reductions would result from scaling up hypertension treatment in the progressive and aspirational scenarios, respectively, with the remaining 15%-17% coming from sodium reduction, the researchers state.
Further, they propose, scaling up BP interventions could reduce CVD inequalities across countries, with low-income and lower-middle-income countries likely experiencing the largest reductions in disease rates and mortality.
Implementation barriers
“Health systems in many low- and middle-income countries have not traditionally been set up to succeed in chronic disease management in primary care,” Dr. Watkins noted. For interventions to be successful, he said, “several barriers need to be addressed, including: low population awareness of chronic diseases like hypertension and diabetes, which leads to low rates of screening and treatment; high out-of-pocket cost and low availability of medicines for chronic diseases; and need for adherence support and provider incentives for improving quality of chronic disease care in primary care settings.”
“Based on the analysis, achieving the 80-80-80 seems feasible, though actually getting there may be much more complicated. I wonder whether countries have the resources to implement the needed policies,” Rodrigo M. Carrillo-Larco, MD, researcher, department of epidemiology and biostatistics, School of Public Health, Imperial College London, told this news organization.
“It may be challenging, particularly after COVID-19, which revealed deficiencies in many health care systems, and care for hypertension may have been disturbed,” said Dr. Carrillo-Larco, who is not connected with the analysis.
That said, simplified BP screening approaches could help maximize the number of people screened overall, potentially identifying those with hypertension and raising awareness, he proposed. His team’s recent study showed that such approaches vary from country to country but are generally reliable and can be used effectively for population screening.
In addition, Dr. Carrillo-Larco said, any efforts by clinicians to improve adherence and help patients achieve BP control “would also have positive effects at the population level.”
The study was supported by a grant from the Bill & Melinda Gates Foundation, with additional funding by a grant to Dr. Watkins from Resolve to Save Lives. No conflicts of interest were declared.
A version of this article first appeared on Medscape.com.
U.S. News issues top hospitals list, now with expanded health equity measures
For the seventh consecutive year, the Mayo Clinic in Rochester, Minn., took the top spot in the annual honor roll of best hospitals, published July 26 by U.S. News & World Report.
The 2022 rankings, which marks the 33rd edition, showcase several methodology changes, including new ratings for ovarian, prostate, and uterine cancer surgeries that “provide patients ... with previously unavailable information to assist them in making a critical health care decision,” a news release from the publication explains.
said the release. Finally, a new metric called “home time” determines how successfully each hospital helps patients return home.
Mayo Clinic remains No. 1
For the 2022-2023 rankings and ratings, U.S. News compared more than 4,500 medical centers across the country in 15 specialties and 20 procedures and conditions. Of these, 493 were recognized as Best Regional Hospitals as a result of their overall strong performance.
The list was then narrowed to the top 20 hospitals, outlined in the honor roll below, that deliver “exceptional treatment across multiple areas of care.”
Following Mayo Clinic in the annual ranking’s top spot, Cedars-Sinai Medical Center in Los Angeles rises from No. 6 to No. 2, and New York University Langone Hospitals finish third, up from eighth in 2021.
Cleveland Clinic in Ohio holds the No. 4 spot, down two from 2021, while Johns Hopkins Hospital in Baltimore and UCLA Medical Center in Los Angeles tie for fifth place. Rounding out the top 10, in order, are: New York–Presbyterian Hospital–Columbia and Cornell, New York; Massachusetts General Hospital, Boston; Northwestern Memorial Hospital, Chicago; Stanford (Calif.) Health Care–Stanford Hospital.
The following hospitals complete the top 20 in the United States:
- 11. Barnes-Jewish Hospital, St. Louis
- 12. UCSF Medical Center, San Francisco
- 13. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
- 14. Brigham and Women’s Hospital, Boston
- 15. Houston Methodist Hospital
- 16. Mount Sinai Hospital, New York
- 17. University of Michigan Health–Michigan Medicine, Ann Arbor
- 18. Mayo Clinic–Phoenix
- 19. Vanderbilt University Medical Center, Nashville, Tenn.
- 20. Rush University Medical Center, Chicago
For the specialty rankings, the University of Texas MD Anderson Cancer Center, Houston, remains No. 1 in cancer care, the Cleveland Clinic is No. 1 in cardiology and heart surgery, and the Hospital for Special Surgery in New York is No. 1 in orthopedics.
Top five for cancer
- 1. University of Texas MD Anderson Cancer Center, Houston
- 2. Memorial Sloan Kettering Cancer Center, New York
- 3. Mayo Clinic, Rochester, Minn.
- 4. Dana-Farber/Brigham and Women’s Cancer Center, Boston
- 5. UCLA Medical Center, Los Angeles
Top five for cardiology and heart surgery
- 1. Cleveland Clinic
- 2. Mayo Clinic, Rochester, Minn.
- 3. Cedars-Sinai Medical Center, Los Angeles
- 4. New York–Presbyterian Hospital–Columbia and Cornell, New York
- 5. New York University Langone Hospitals
Top five for orthopedics
- 1. Hospital for Special Surgery, New York
- 2. Mayo Clinic, Rochester, Minn.
- 3. Cedars-Sinai Medical Center, Los Angeles
- 4. New York University Langone Hospitals
- 5. (tie) Rush University Medical Center, Chicago
- 5. (tie) UCLA Medical Center, Los Angeles
According to the news release, the procedures and conditions ratings are based entirely on objective patient care measures like survival rates, patient experience, home time, and level of nursing care. The Best Hospitals rankings consider a variety of data provided by the Centers for Medicare & Medicaid Services, American Hospital Association, professional organizations, and medical specialists.
The full report is available online.
A version of this article first appeared on Medscape.com.
For the seventh consecutive year, the Mayo Clinic in Rochester, Minn., took the top spot in the annual honor roll of best hospitals, published July 26 by U.S. News & World Report.
The 2022 rankings, which marks the 33rd edition, showcase several methodology changes, including new ratings for ovarian, prostate, and uterine cancer surgeries that “provide patients ... with previously unavailable information to assist them in making a critical health care decision,” a news release from the publication explains.
said the release. Finally, a new metric called “home time” determines how successfully each hospital helps patients return home.
Mayo Clinic remains No. 1
For the 2022-2023 rankings and ratings, U.S. News compared more than 4,500 medical centers across the country in 15 specialties and 20 procedures and conditions. Of these, 493 were recognized as Best Regional Hospitals as a result of their overall strong performance.
The list was then narrowed to the top 20 hospitals, outlined in the honor roll below, that deliver “exceptional treatment across multiple areas of care.”
Following Mayo Clinic in the annual ranking’s top spot, Cedars-Sinai Medical Center in Los Angeles rises from No. 6 to No. 2, and New York University Langone Hospitals finish third, up from eighth in 2021.
Cleveland Clinic in Ohio holds the No. 4 spot, down two from 2021, while Johns Hopkins Hospital in Baltimore and UCLA Medical Center in Los Angeles tie for fifth place. Rounding out the top 10, in order, are: New York–Presbyterian Hospital–Columbia and Cornell, New York; Massachusetts General Hospital, Boston; Northwestern Memorial Hospital, Chicago; Stanford (Calif.) Health Care–Stanford Hospital.
The following hospitals complete the top 20 in the United States:
- 11. Barnes-Jewish Hospital, St. Louis
- 12. UCSF Medical Center, San Francisco
- 13. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
- 14. Brigham and Women’s Hospital, Boston
- 15. Houston Methodist Hospital
- 16. Mount Sinai Hospital, New York
- 17. University of Michigan Health–Michigan Medicine, Ann Arbor
- 18. Mayo Clinic–Phoenix
- 19. Vanderbilt University Medical Center, Nashville, Tenn.
- 20. Rush University Medical Center, Chicago
For the specialty rankings, the University of Texas MD Anderson Cancer Center, Houston, remains No. 1 in cancer care, the Cleveland Clinic is No. 1 in cardiology and heart surgery, and the Hospital for Special Surgery in New York is No. 1 in orthopedics.
Top five for cancer
- 1. University of Texas MD Anderson Cancer Center, Houston
- 2. Memorial Sloan Kettering Cancer Center, New York
- 3. Mayo Clinic, Rochester, Minn.
- 4. Dana-Farber/Brigham and Women’s Cancer Center, Boston
- 5. UCLA Medical Center, Los Angeles
Top five for cardiology and heart surgery
- 1. Cleveland Clinic
- 2. Mayo Clinic, Rochester, Minn.
- 3. Cedars-Sinai Medical Center, Los Angeles
- 4. New York–Presbyterian Hospital–Columbia and Cornell, New York
- 5. New York University Langone Hospitals
Top five for orthopedics
- 1. Hospital for Special Surgery, New York
- 2. Mayo Clinic, Rochester, Minn.
- 3. Cedars-Sinai Medical Center, Los Angeles
- 4. New York University Langone Hospitals
- 5. (tie) Rush University Medical Center, Chicago
- 5. (tie) UCLA Medical Center, Los Angeles
According to the news release, the procedures and conditions ratings are based entirely on objective patient care measures like survival rates, patient experience, home time, and level of nursing care. The Best Hospitals rankings consider a variety of data provided by the Centers for Medicare & Medicaid Services, American Hospital Association, professional organizations, and medical specialists.
The full report is available online.
A version of this article first appeared on Medscape.com.
For the seventh consecutive year, the Mayo Clinic in Rochester, Minn., took the top spot in the annual honor roll of best hospitals, published July 26 by U.S. News & World Report.
The 2022 rankings, which marks the 33rd edition, showcase several methodology changes, including new ratings for ovarian, prostate, and uterine cancer surgeries that “provide patients ... with previously unavailable information to assist them in making a critical health care decision,” a news release from the publication explains.
said the release. Finally, a new metric called “home time” determines how successfully each hospital helps patients return home.
Mayo Clinic remains No. 1
For the 2022-2023 rankings and ratings, U.S. News compared more than 4,500 medical centers across the country in 15 specialties and 20 procedures and conditions. Of these, 493 were recognized as Best Regional Hospitals as a result of their overall strong performance.
The list was then narrowed to the top 20 hospitals, outlined in the honor roll below, that deliver “exceptional treatment across multiple areas of care.”
Following Mayo Clinic in the annual ranking’s top spot, Cedars-Sinai Medical Center in Los Angeles rises from No. 6 to No. 2, and New York University Langone Hospitals finish third, up from eighth in 2021.
Cleveland Clinic in Ohio holds the No. 4 spot, down two from 2021, while Johns Hopkins Hospital in Baltimore and UCLA Medical Center in Los Angeles tie for fifth place. Rounding out the top 10, in order, are: New York–Presbyterian Hospital–Columbia and Cornell, New York; Massachusetts General Hospital, Boston; Northwestern Memorial Hospital, Chicago; Stanford (Calif.) Health Care–Stanford Hospital.
The following hospitals complete the top 20 in the United States:
- 11. Barnes-Jewish Hospital, St. Louis
- 12. UCSF Medical Center, San Francisco
- 13. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
- 14. Brigham and Women’s Hospital, Boston
- 15. Houston Methodist Hospital
- 16. Mount Sinai Hospital, New York
- 17. University of Michigan Health–Michigan Medicine, Ann Arbor
- 18. Mayo Clinic–Phoenix
- 19. Vanderbilt University Medical Center, Nashville, Tenn.
- 20. Rush University Medical Center, Chicago
For the specialty rankings, the University of Texas MD Anderson Cancer Center, Houston, remains No. 1 in cancer care, the Cleveland Clinic is No. 1 in cardiology and heart surgery, and the Hospital for Special Surgery in New York is No. 1 in orthopedics.
Top five for cancer
- 1. University of Texas MD Anderson Cancer Center, Houston
- 2. Memorial Sloan Kettering Cancer Center, New York
- 3. Mayo Clinic, Rochester, Minn.
- 4. Dana-Farber/Brigham and Women’s Cancer Center, Boston
- 5. UCLA Medical Center, Los Angeles
Top five for cardiology and heart surgery
- 1. Cleveland Clinic
- 2. Mayo Clinic, Rochester, Minn.
- 3. Cedars-Sinai Medical Center, Los Angeles
- 4. New York–Presbyterian Hospital–Columbia and Cornell, New York
- 5. New York University Langone Hospitals
Top five for orthopedics
- 1. Hospital for Special Surgery, New York
- 2. Mayo Clinic, Rochester, Minn.
- 3. Cedars-Sinai Medical Center, Los Angeles
- 4. New York University Langone Hospitals
- 5. (tie) Rush University Medical Center, Chicago
- 5. (tie) UCLA Medical Center, Los Angeles
According to the news release, the procedures and conditions ratings are based entirely on objective patient care measures like survival rates, patient experience, home time, and level of nursing care. The Best Hospitals rankings consider a variety of data provided by the Centers for Medicare & Medicaid Services, American Hospital Association, professional organizations, and medical specialists.
The full report is available online.
A version of this article first appeared on Medscape.com.





