User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
‘Substantial’ variation in responses to BP meds
A new study has shown a substantial variation in the blood pressure response to various antihypertensive medications between individuals, raising the possibility of future personalized therapy.
“We found that using the optimal antihypertensive drug for a particular patient resulted in an average of a 4.4 mm Hg greater reduction of blood pressure compared with a random choice of the other drugs. That is quite a substantial difference, and could be equivalent to adding in another drug,” lead author Johan Sundström, MD, Uppsala (Sweden) University Hospital, told this news organization.
“These preliminary findings suggest that some people may be better treated with one antihypertensive drug rather than another. This is opening up the field of hypertension for personalized medicine,” he added.
The study was published online in the Journal of the American Medical Association.
The authors noted that despite global access to multiple classes of highly effective blood pressure-lowering drugs, only one in four women and one in five men with hypertension reach treatment targets. While most hypertension guidelines advocate combination pharmacotherapy, many patients in routine care continue to be treated with monotherapy, with adverse effects and nonadherence being important clinical problems.
“One drug often does not give enough blood pressure reduction, but patients are often reluctant to up-titrate to two drugs,” Dr. Sundström said. “While we know that the four recommended classes of antihypertensives lower blood pressure equally well on average, we don’t know if their efficacy is the same in individual patients.
“We wondered whether there could be different optimal drugs for different people, and if we could identify the optimal drug for each person then maybe more patients could get to target levels with just one drug,” he said.
The researchers conducted a randomized, double-blind, repeated crossover trial at an outpatient research clinic in Sweden, studying 280 men and women with grade 1 hypertension at low risk for cardiovascular events.
Each participant was scheduled for 2 months’ treatment in random order with each of four different classes of antihypertensive drugs: an ACE inhibitor, lisinopril; an angiotensin II blocker, candesartan; a thiazide diuretic, hydrochlorothiazide; a calcium channel blocker, amlodipine.
There were then repeated treatment periods for two drug classes to try to account for any effect of a particular event that might have affected the blood pressure at one point in time. Ambulatory daytime systolic blood pressure was measured at the end of each treatment period.
Results showed that variation in systolic blood pressure was large between treatments on average, between participants on average, within participants taking the same treatment, and between treatments in the same participant.
Overall, personalized treatment using the optimal single-drug therapy led to a 4.4–mm Hg lower systolic blood pressure in the trial population than a random choice of any of the other drug classes.
Taking into consideration that lisinopril was found to be on average the most efficacious of the drugs at the selected doses, personalized treatment compared with lisinopril still led to a 3.1–mm Hg improvement in systolic blood pressure.
The researchers noted that the mean additional blood pressure reduction achievable by using the optimal agent was of a magnitude twice that achieved by doubling the dose of a first drug, and more than half that of adding a second drug on average.
While there were only small differences between certain drugs (e.g., candesartan vs. lisinopril; amlodipine vs. hydrochlorothiazide), for all other comparisons tested, the choice was important, with particularly large gains to be made by personalizing the choice between candesartan vs. amlodipine and between lisinopril vs. amlodipine.
In addition, some people showed very large differences in response to different drugs, whereas others did not have much difference at all.
How to identify the optimal drug?
“The million-dollar question is how we identify the best drug for each individual patient,” Dr. Sundström said. “This study has opened Pandora’s box. We now need to figure out how to go forward and how we tailor treatment in each patient.”
In the study, the researchers suggest that personalizing therapy could be achieved either by identifying the phenotypic characteristics that are associated with enhanced response to one treatment vs. another or by directly measuring the individual’s responses to a series of treatments to ascertain which is most effective.
Addressing the first scenario, Dr. Sundström explained: “We can analyze the characteristics of patients who did best on each drug. There are many variables we can look at here such as age, diet, baseline blood pressure, exercise levels, smoking status, race, body weight, salt intake, and findings from genetic tests. We are going to try to look into these to see if we can find any predictors of response to various different drugs.”
For the second strategy, he suggested that patients starting pharmacologic therapy could try a few different treatments. “For example, we could give patients two different drugs and ask them to alternate treatment periods with each of them and measure their blood pressure with a home monitoring kit and record adverse effects.”
Nonadherence “is such a big problem with antihypertensives,” he added. “This approach may allow patients to be more empowered when choosing the right treatment, which should help adherence in the longer term.”
‘Proof-of-principle’
Commenting on the study in an accompanying editorial, Robert M. Carey, MD, University of Virginia Health System, Charlottesville, wrote: “At this stage, the findings are more theoretical than immediately practical for the implementation of personalized antihypertensive drug therapy, but the study does provide proof-of-principle and the authors suggest a few scenarios in which a personalized approach could be used in the future.”
He said the practical ramifications of personally targeted therapy remain unclear, given that determination of an individual’s response to a series of short test treatments before selecting long-term therapy may be considered too cumbersome, and currently few phenotypic markers are currently available that would be likely to accurately predict the individual response to a particular therapy.
Dr. Carey concluded that the results of this study “encourage the further pursuit of larger randomized trials using similar repeated crossover designs to validate this concept and eventually in trials with longer follow-up data to determine whether there is improvement in long-term clinical outcomes compared with current strategies.”
He added that the results support the possibility that personalized medical treatment of hypertension “may ultimately supplement or even supplant the current method of antihypertensive drug decision-making in the future.”
This study was supported by the Swedish Research Council; Kjell and Märta Beijer Foundation; and Anders Wiklöf. Dr. Sundström reported owning stock in Symptoms Europe AB and Anagram Kommunikation AB. Coauthor Emil Hagström, MD, PhD, reported receiving grants from Pfizer and Amgen and personal fees from Amgen, Novo Nordisk, Bayer, AstraZeneca, Amarin, and Novartis. Coauthor Ollie Östlund, PhD, reported fees from Uppsala University paid to his institution, Uppsala Clinical Research Center, for its participation in the PHYSIC trial during the conduct of the study. Dr. Carey reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study has shown a substantial variation in the blood pressure response to various antihypertensive medications between individuals, raising the possibility of future personalized therapy.
“We found that using the optimal antihypertensive drug for a particular patient resulted in an average of a 4.4 mm Hg greater reduction of blood pressure compared with a random choice of the other drugs. That is quite a substantial difference, and could be equivalent to adding in another drug,” lead author Johan Sundström, MD, Uppsala (Sweden) University Hospital, told this news organization.
“These preliminary findings suggest that some people may be better treated with one antihypertensive drug rather than another. This is opening up the field of hypertension for personalized medicine,” he added.
The study was published online in the Journal of the American Medical Association.
The authors noted that despite global access to multiple classes of highly effective blood pressure-lowering drugs, only one in four women and one in five men with hypertension reach treatment targets. While most hypertension guidelines advocate combination pharmacotherapy, many patients in routine care continue to be treated with monotherapy, with adverse effects and nonadherence being important clinical problems.
“One drug often does not give enough blood pressure reduction, but patients are often reluctant to up-titrate to two drugs,” Dr. Sundström said. “While we know that the four recommended classes of antihypertensives lower blood pressure equally well on average, we don’t know if their efficacy is the same in individual patients.
“We wondered whether there could be different optimal drugs for different people, and if we could identify the optimal drug for each person then maybe more patients could get to target levels with just one drug,” he said.
The researchers conducted a randomized, double-blind, repeated crossover trial at an outpatient research clinic in Sweden, studying 280 men and women with grade 1 hypertension at low risk for cardiovascular events.
Each participant was scheduled for 2 months’ treatment in random order with each of four different classes of antihypertensive drugs: an ACE inhibitor, lisinopril; an angiotensin II blocker, candesartan; a thiazide diuretic, hydrochlorothiazide; a calcium channel blocker, amlodipine.
There were then repeated treatment periods for two drug classes to try to account for any effect of a particular event that might have affected the blood pressure at one point in time. Ambulatory daytime systolic blood pressure was measured at the end of each treatment period.
Results showed that variation in systolic blood pressure was large between treatments on average, between participants on average, within participants taking the same treatment, and between treatments in the same participant.
Overall, personalized treatment using the optimal single-drug therapy led to a 4.4–mm Hg lower systolic blood pressure in the trial population than a random choice of any of the other drug classes.
Taking into consideration that lisinopril was found to be on average the most efficacious of the drugs at the selected doses, personalized treatment compared with lisinopril still led to a 3.1–mm Hg improvement in systolic blood pressure.
The researchers noted that the mean additional blood pressure reduction achievable by using the optimal agent was of a magnitude twice that achieved by doubling the dose of a first drug, and more than half that of adding a second drug on average.
While there were only small differences between certain drugs (e.g., candesartan vs. lisinopril; amlodipine vs. hydrochlorothiazide), for all other comparisons tested, the choice was important, with particularly large gains to be made by personalizing the choice between candesartan vs. amlodipine and between lisinopril vs. amlodipine.
In addition, some people showed very large differences in response to different drugs, whereas others did not have much difference at all.
How to identify the optimal drug?
“The million-dollar question is how we identify the best drug for each individual patient,” Dr. Sundström said. “This study has opened Pandora’s box. We now need to figure out how to go forward and how we tailor treatment in each patient.”
In the study, the researchers suggest that personalizing therapy could be achieved either by identifying the phenotypic characteristics that are associated with enhanced response to one treatment vs. another or by directly measuring the individual’s responses to a series of treatments to ascertain which is most effective.
Addressing the first scenario, Dr. Sundström explained: “We can analyze the characteristics of patients who did best on each drug. There are many variables we can look at here such as age, diet, baseline blood pressure, exercise levels, smoking status, race, body weight, salt intake, and findings from genetic tests. We are going to try to look into these to see if we can find any predictors of response to various different drugs.”
For the second strategy, he suggested that patients starting pharmacologic therapy could try a few different treatments. “For example, we could give patients two different drugs and ask them to alternate treatment periods with each of them and measure their blood pressure with a home monitoring kit and record adverse effects.”
Nonadherence “is such a big problem with antihypertensives,” he added. “This approach may allow patients to be more empowered when choosing the right treatment, which should help adherence in the longer term.”
‘Proof-of-principle’
Commenting on the study in an accompanying editorial, Robert M. Carey, MD, University of Virginia Health System, Charlottesville, wrote: “At this stage, the findings are more theoretical than immediately practical for the implementation of personalized antihypertensive drug therapy, but the study does provide proof-of-principle and the authors suggest a few scenarios in which a personalized approach could be used in the future.”
He said the practical ramifications of personally targeted therapy remain unclear, given that determination of an individual’s response to a series of short test treatments before selecting long-term therapy may be considered too cumbersome, and currently few phenotypic markers are currently available that would be likely to accurately predict the individual response to a particular therapy.
Dr. Carey concluded that the results of this study “encourage the further pursuit of larger randomized trials using similar repeated crossover designs to validate this concept and eventually in trials with longer follow-up data to determine whether there is improvement in long-term clinical outcomes compared with current strategies.”
He added that the results support the possibility that personalized medical treatment of hypertension “may ultimately supplement or even supplant the current method of antihypertensive drug decision-making in the future.”
This study was supported by the Swedish Research Council; Kjell and Märta Beijer Foundation; and Anders Wiklöf. Dr. Sundström reported owning stock in Symptoms Europe AB and Anagram Kommunikation AB. Coauthor Emil Hagström, MD, PhD, reported receiving grants from Pfizer and Amgen and personal fees from Amgen, Novo Nordisk, Bayer, AstraZeneca, Amarin, and Novartis. Coauthor Ollie Östlund, PhD, reported fees from Uppsala University paid to his institution, Uppsala Clinical Research Center, for its participation in the PHYSIC trial during the conduct of the study. Dr. Carey reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study has shown a substantial variation in the blood pressure response to various antihypertensive medications between individuals, raising the possibility of future personalized therapy.
“We found that using the optimal antihypertensive drug for a particular patient resulted in an average of a 4.4 mm Hg greater reduction of blood pressure compared with a random choice of the other drugs. That is quite a substantial difference, and could be equivalent to adding in another drug,” lead author Johan Sundström, MD, Uppsala (Sweden) University Hospital, told this news organization.
“These preliminary findings suggest that some people may be better treated with one antihypertensive drug rather than another. This is opening up the field of hypertension for personalized medicine,” he added.
The study was published online in the Journal of the American Medical Association.
The authors noted that despite global access to multiple classes of highly effective blood pressure-lowering drugs, only one in four women and one in five men with hypertension reach treatment targets. While most hypertension guidelines advocate combination pharmacotherapy, many patients in routine care continue to be treated with monotherapy, with adverse effects and nonadherence being important clinical problems.
“One drug often does not give enough blood pressure reduction, but patients are often reluctant to up-titrate to two drugs,” Dr. Sundström said. “While we know that the four recommended classes of antihypertensives lower blood pressure equally well on average, we don’t know if their efficacy is the same in individual patients.
“We wondered whether there could be different optimal drugs for different people, and if we could identify the optimal drug for each person then maybe more patients could get to target levels with just one drug,” he said.
The researchers conducted a randomized, double-blind, repeated crossover trial at an outpatient research clinic in Sweden, studying 280 men and women with grade 1 hypertension at low risk for cardiovascular events.
Each participant was scheduled for 2 months’ treatment in random order with each of four different classes of antihypertensive drugs: an ACE inhibitor, lisinopril; an angiotensin II blocker, candesartan; a thiazide diuretic, hydrochlorothiazide; a calcium channel blocker, amlodipine.
There were then repeated treatment periods for two drug classes to try to account for any effect of a particular event that might have affected the blood pressure at one point in time. Ambulatory daytime systolic blood pressure was measured at the end of each treatment period.
Results showed that variation in systolic blood pressure was large between treatments on average, between participants on average, within participants taking the same treatment, and between treatments in the same participant.
Overall, personalized treatment using the optimal single-drug therapy led to a 4.4–mm Hg lower systolic blood pressure in the trial population than a random choice of any of the other drug classes.
Taking into consideration that lisinopril was found to be on average the most efficacious of the drugs at the selected doses, personalized treatment compared with lisinopril still led to a 3.1–mm Hg improvement in systolic blood pressure.
The researchers noted that the mean additional blood pressure reduction achievable by using the optimal agent was of a magnitude twice that achieved by doubling the dose of a first drug, and more than half that of adding a second drug on average.
While there were only small differences between certain drugs (e.g., candesartan vs. lisinopril; amlodipine vs. hydrochlorothiazide), for all other comparisons tested, the choice was important, with particularly large gains to be made by personalizing the choice between candesartan vs. amlodipine and between lisinopril vs. amlodipine.
In addition, some people showed very large differences in response to different drugs, whereas others did not have much difference at all.
How to identify the optimal drug?
“The million-dollar question is how we identify the best drug for each individual patient,” Dr. Sundström said. “This study has opened Pandora’s box. We now need to figure out how to go forward and how we tailor treatment in each patient.”
In the study, the researchers suggest that personalizing therapy could be achieved either by identifying the phenotypic characteristics that are associated with enhanced response to one treatment vs. another or by directly measuring the individual’s responses to a series of treatments to ascertain which is most effective.
Addressing the first scenario, Dr. Sundström explained: “We can analyze the characteristics of patients who did best on each drug. There are many variables we can look at here such as age, diet, baseline blood pressure, exercise levels, smoking status, race, body weight, salt intake, and findings from genetic tests. We are going to try to look into these to see if we can find any predictors of response to various different drugs.”
For the second strategy, he suggested that patients starting pharmacologic therapy could try a few different treatments. “For example, we could give patients two different drugs and ask them to alternate treatment periods with each of them and measure their blood pressure with a home monitoring kit and record adverse effects.”
Nonadherence “is such a big problem with antihypertensives,” he added. “This approach may allow patients to be more empowered when choosing the right treatment, which should help adherence in the longer term.”
‘Proof-of-principle’
Commenting on the study in an accompanying editorial, Robert M. Carey, MD, University of Virginia Health System, Charlottesville, wrote: “At this stage, the findings are more theoretical than immediately practical for the implementation of personalized antihypertensive drug therapy, but the study does provide proof-of-principle and the authors suggest a few scenarios in which a personalized approach could be used in the future.”
He said the practical ramifications of personally targeted therapy remain unclear, given that determination of an individual’s response to a series of short test treatments before selecting long-term therapy may be considered too cumbersome, and currently few phenotypic markers are currently available that would be likely to accurately predict the individual response to a particular therapy.
Dr. Carey concluded that the results of this study “encourage the further pursuit of larger randomized trials using similar repeated crossover designs to validate this concept and eventually in trials with longer follow-up data to determine whether there is improvement in long-term clinical outcomes compared with current strategies.”
He added that the results support the possibility that personalized medical treatment of hypertension “may ultimately supplement or even supplant the current method of antihypertensive drug decision-making in the future.”
This study was supported by the Swedish Research Council; Kjell and Märta Beijer Foundation; and Anders Wiklöf. Dr. Sundström reported owning stock in Symptoms Europe AB and Anagram Kommunikation AB. Coauthor Emil Hagström, MD, PhD, reported receiving grants from Pfizer and Amgen and personal fees from Amgen, Novo Nordisk, Bayer, AstraZeneca, Amarin, and Novartis. Coauthor Ollie Östlund, PhD, reported fees from Uppsala University paid to his institution, Uppsala Clinical Research Center, for its participation in the PHYSIC trial during the conduct of the study. Dr. Carey reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Integrating addiction medicine with primary care cost effective: Study
Integrating buprenorphine and harm eduction tools into primary care may improve clinical outcomes, increase costs only modestly, and be cost effective in health systems, authors conclude in an original investigation in JAMA Network Open.
A team led by Raagini Jawa, MD, MPH, with the Center for Research on Healthcare, University of Pittsburgh, set out to analyze costs of the interventions versus increased benefit in extending life expectancy.
Their analysis found that, compared with the status quo, integrating buprenorphine and harm reduction kits (syringes, wound care supplies, etc.) reduced drug use–related deaths by 33% and was cost effective.
“Our results suggest that integrated addiction care in primary care has the potential to save lives and increase nonemergency health care use, which is consistent with prior literature,” the authors write. “Colocated addiction services within primary care is pragmatic and effective and has comparable quality to specialty care. We found that onsite BUP [buprenorphine prescribing] plus HR [harm reduction] provides better outcomes than BUP alone at a lower cost.”
Three strategies compared
Using a microsimulation model of 2.25 million people in the United States who inject opioids, with an average age of 44 (69% of them male), the researchers tested three strategies:
- Status quo. PCP refers to addiction care.
- BUP. PCP services plus onsite buprenorphine prescribing with referral to off-site harm reduction kits.
- BUP plus HR. PCP services plus on-site buprenorphine prescribing and harm reduction kits.
The model is the Reducing Infections Related to Drug Use Cost-Effectiveness (REDUCE) microsimulation model, which tracks serious injection-related infections, overdose, hospitalization, and death.
The status quo (referral for treatment) resulted in 1,162 overdose deaths per 10,000 people (95% credible interval, 1,144-2,303), whereas both BUP and BUP plus HR resulted in about 160 fewer deaths per 10,000 people (95% Crl for BUP, 802-1718; 95% CrI for BUP plus HR, 692-1,810).
Compared with the status quo strategy, life expectancy was lengthened with the BUP strategy by 2.65 years and BUP plus HR by 2.71 years.
Researchers found the average discounted lifetime cost per person of both the BUP strategy and the BUP plus HR strategy were higher than the average status quo.
“The dominating strategy was BUP plus HR,” the authors write. “Compared with status quo, BUP plus HR was cost effective (incremental cost-effectiveness ratio [ICER], $34,400 per life year).”
Cost for primary care practices
Comparatively, over a 5-year period, BUP plus HR was found to cost an individual PCP practice approximately $13,000.
That cost includes direct costs for resources and opportunity costs, the authors write. These costs could be offset by health care system savings.
“These costs included those for X-waiver training, which has been eliminated; thus, we expect this to cost less. Put another way, our findings inform ways to reinvest health care dollars as financial incentives for PCPs to adopt this new paradigm. Public health departments could provide grants or harm reduction kit supplies directly to PCPs to offset these costs as they do in some places with syringe service programs and/or increase Medicaid reimbursements for providing addiction care in primary care,” they write.
Data help make the case
Dinah Applewhite, MD, a primary care physician and addiction medicine specialist at Massachusetts General Hospital in Boston, who was not part of the study, said clinicians there have seen the benefits of integrating various aspects of addiction medicine into primary care but these data on outcomes and cost-effectiveness can help make the case to hospital leaders, legislators, and grant providers.
The primary care setting also provides a chance to engage patients around their injection practice and explore ways to minimize risk, she said.
“By offering them these kits, it lets them know your priority is their safety and well-being,” Dr. Applewhite said.
She noted that the linkage to primary care was low for patients who inject drugs, which speaks to the need for models in addition to this one, such as bringing primary care clinicians into syringe service programs.
“The medical establishment has a lot to learn from these programs,” she said.
Practices need support
She said it’s important to note that primary care practices need support from administrative leaders, philanthropists, and grant providers to help cover the costs.
“It’s one of the barriers to doing this,” she said. “There isn’t a mechanism to pay for this.”
Sarah Bagley, MD, a primary care physician at Boston Medical Center and medical director of BMC’s Center for Addiction Treatment for Adolescents/Young Adults Who Use Substances told this publication she was excited to see that the addition of harm reduction kits to buprenorphine seemed to have the optimal effect in improving outcomes. People with substance abuse disorders should feel they are welcome in primary care even if they are not yet ready to stop drug use, she said.
She said she was also glad to see increased life expectancy with these interventions. The news of overdose deaths contributing to a decrease in life expectancy can be overwhelming, she said.
But this study, she says, offers a road map for addressing the overdose crisis “by including harm reduction in the substance abuse care we provide.”
She pointed out that the study showed that costs increase per patient with both interventions, compared with the status quo. The study found that health care costs per person during a lifetime increased, compared with the status quo, by 69.1% for BUP and 74.3% for BUP plus HR.
But it’s important to understand the reason for that, she said: “The cost was higher because people were staying alive.”
She said it may help to compare giving optimal care to people who have substance abuse disorders with giving optimal care to people with other chronic conditions, such as diabetes, who may not always adhere to recommended diets or treatment regimens.
“We still invite those patients in and work with them based on where they are,” she said.
Growing epidemic
The researchers point to the urgent need for solutions given the U.S. opioid epidemic, which has led to increasing numbers of overdoses and injection drug use–related infections, such as infective endocarditis, and severe skin and soft tissue infections.
They point out that primary care providers are the largest clinical workforce in the United States, but few of their practices offer comprehensive addiction care onsite.
“Primary care practices are a practical place to integrate addiction services, where PCPs can prescribe buprenorphine and deliver harm reduction kits,” they write.
Coauthor Dr. Kimmel reports personal fees from Massachusetts Department of Public Health, Bureau of Substance Addiction Services Overdose Education and Prevention Program, and American Academy of Addiction Psychiatry, Opioid Response Network for harm reduction education outside the submitted work and previous consulting with Abt Associates on a Massachusetts Department of Public Health–funded project to improve access to medications for opioid use disorder treatment. Dr. Applewhite and Dr. Bagley report no relevant financial relationships.
Integrating buprenorphine and harm eduction tools into primary care may improve clinical outcomes, increase costs only modestly, and be cost effective in health systems, authors conclude in an original investigation in JAMA Network Open.
A team led by Raagini Jawa, MD, MPH, with the Center for Research on Healthcare, University of Pittsburgh, set out to analyze costs of the interventions versus increased benefit in extending life expectancy.
Their analysis found that, compared with the status quo, integrating buprenorphine and harm reduction kits (syringes, wound care supplies, etc.) reduced drug use–related deaths by 33% and was cost effective.
“Our results suggest that integrated addiction care in primary care has the potential to save lives and increase nonemergency health care use, which is consistent with prior literature,” the authors write. “Colocated addiction services within primary care is pragmatic and effective and has comparable quality to specialty care. We found that onsite BUP [buprenorphine prescribing] plus HR [harm reduction] provides better outcomes than BUP alone at a lower cost.”
Three strategies compared
Using a microsimulation model of 2.25 million people in the United States who inject opioids, with an average age of 44 (69% of them male), the researchers tested three strategies:
- Status quo. PCP refers to addiction care.
- BUP. PCP services plus onsite buprenorphine prescribing with referral to off-site harm reduction kits.
- BUP plus HR. PCP services plus on-site buprenorphine prescribing and harm reduction kits.
The model is the Reducing Infections Related to Drug Use Cost-Effectiveness (REDUCE) microsimulation model, which tracks serious injection-related infections, overdose, hospitalization, and death.
The status quo (referral for treatment) resulted in 1,162 overdose deaths per 10,000 people (95% credible interval, 1,144-2,303), whereas both BUP and BUP plus HR resulted in about 160 fewer deaths per 10,000 people (95% Crl for BUP, 802-1718; 95% CrI for BUP plus HR, 692-1,810).
Compared with the status quo strategy, life expectancy was lengthened with the BUP strategy by 2.65 years and BUP plus HR by 2.71 years.
Researchers found the average discounted lifetime cost per person of both the BUP strategy and the BUP plus HR strategy were higher than the average status quo.
“The dominating strategy was BUP plus HR,” the authors write. “Compared with status quo, BUP plus HR was cost effective (incremental cost-effectiveness ratio [ICER], $34,400 per life year).”
Cost for primary care practices
Comparatively, over a 5-year period, BUP plus HR was found to cost an individual PCP practice approximately $13,000.
That cost includes direct costs for resources and opportunity costs, the authors write. These costs could be offset by health care system savings.
“These costs included those for X-waiver training, which has been eliminated; thus, we expect this to cost less. Put another way, our findings inform ways to reinvest health care dollars as financial incentives for PCPs to adopt this new paradigm. Public health departments could provide grants or harm reduction kit supplies directly to PCPs to offset these costs as they do in some places with syringe service programs and/or increase Medicaid reimbursements for providing addiction care in primary care,” they write.
Data help make the case
Dinah Applewhite, MD, a primary care physician and addiction medicine specialist at Massachusetts General Hospital in Boston, who was not part of the study, said clinicians there have seen the benefits of integrating various aspects of addiction medicine into primary care but these data on outcomes and cost-effectiveness can help make the case to hospital leaders, legislators, and grant providers.
The primary care setting also provides a chance to engage patients around their injection practice and explore ways to minimize risk, she said.
“By offering them these kits, it lets them know your priority is their safety and well-being,” Dr. Applewhite said.
She noted that the linkage to primary care was low for patients who inject drugs, which speaks to the need for models in addition to this one, such as bringing primary care clinicians into syringe service programs.
“The medical establishment has a lot to learn from these programs,” she said.
Practices need support
She said it’s important to note that primary care practices need support from administrative leaders, philanthropists, and grant providers to help cover the costs.
“It’s one of the barriers to doing this,” she said. “There isn’t a mechanism to pay for this.”
Sarah Bagley, MD, a primary care physician at Boston Medical Center and medical director of BMC’s Center for Addiction Treatment for Adolescents/Young Adults Who Use Substances told this publication she was excited to see that the addition of harm reduction kits to buprenorphine seemed to have the optimal effect in improving outcomes. People with substance abuse disorders should feel they are welcome in primary care even if they are not yet ready to stop drug use, she said.
She said she was also glad to see increased life expectancy with these interventions. The news of overdose deaths contributing to a decrease in life expectancy can be overwhelming, she said.
But this study, she says, offers a road map for addressing the overdose crisis “by including harm reduction in the substance abuse care we provide.”
She pointed out that the study showed that costs increase per patient with both interventions, compared with the status quo. The study found that health care costs per person during a lifetime increased, compared with the status quo, by 69.1% for BUP and 74.3% for BUP plus HR.
But it’s important to understand the reason for that, she said: “The cost was higher because people were staying alive.”
She said it may help to compare giving optimal care to people who have substance abuse disorders with giving optimal care to people with other chronic conditions, such as diabetes, who may not always adhere to recommended diets or treatment regimens.
“We still invite those patients in and work with them based on where they are,” she said.
Growing epidemic
The researchers point to the urgent need for solutions given the U.S. opioid epidemic, which has led to increasing numbers of overdoses and injection drug use–related infections, such as infective endocarditis, and severe skin and soft tissue infections.
They point out that primary care providers are the largest clinical workforce in the United States, but few of their practices offer comprehensive addiction care onsite.
“Primary care practices are a practical place to integrate addiction services, where PCPs can prescribe buprenorphine and deliver harm reduction kits,” they write.
Coauthor Dr. Kimmel reports personal fees from Massachusetts Department of Public Health, Bureau of Substance Addiction Services Overdose Education and Prevention Program, and American Academy of Addiction Psychiatry, Opioid Response Network for harm reduction education outside the submitted work and previous consulting with Abt Associates on a Massachusetts Department of Public Health–funded project to improve access to medications for opioid use disorder treatment. Dr. Applewhite and Dr. Bagley report no relevant financial relationships.
Integrating buprenorphine and harm eduction tools into primary care may improve clinical outcomes, increase costs only modestly, and be cost effective in health systems, authors conclude in an original investigation in JAMA Network Open.
A team led by Raagini Jawa, MD, MPH, with the Center for Research on Healthcare, University of Pittsburgh, set out to analyze costs of the interventions versus increased benefit in extending life expectancy.
Their analysis found that, compared with the status quo, integrating buprenorphine and harm reduction kits (syringes, wound care supplies, etc.) reduced drug use–related deaths by 33% and was cost effective.
“Our results suggest that integrated addiction care in primary care has the potential to save lives and increase nonemergency health care use, which is consistent with prior literature,” the authors write. “Colocated addiction services within primary care is pragmatic and effective and has comparable quality to specialty care. We found that onsite BUP [buprenorphine prescribing] plus HR [harm reduction] provides better outcomes than BUP alone at a lower cost.”
Three strategies compared
Using a microsimulation model of 2.25 million people in the United States who inject opioids, with an average age of 44 (69% of them male), the researchers tested three strategies:
- Status quo. PCP refers to addiction care.
- BUP. PCP services plus onsite buprenorphine prescribing with referral to off-site harm reduction kits.
- BUP plus HR. PCP services plus on-site buprenorphine prescribing and harm reduction kits.
The model is the Reducing Infections Related to Drug Use Cost-Effectiveness (REDUCE) microsimulation model, which tracks serious injection-related infections, overdose, hospitalization, and death.
The status quo (referral for treatment) resulted in 1,162 overdose deaths per 10,000 people (95% credible interval, 1,144-2,303), whereas both BUP and BUP plus HR resulted in about 160 fewer deaths per 10,000 people (95% Crl for BUP, 802-1718; 95% CrI for BUP plus HR, 692-1,810).
Compared with the status quo strategy, life expectancy was lengthened with the BUP strategy by 2.65 years and BUP plus HR by 2.71 years.
Researchers found the average discounted lifetime cost per person of both the BUP strategy and the BUP plus HR strategy were higher than the average status quo.
“The dominating strategy was BUP plus HR,” the authors write. “Compared with status quo, BUP plus HR was cost effective (incremental cost-effectiveness ratio [ICER], $34,400 per life year).”
Cost for primary care practices
Comparatively, over a 5-year period, BUP plus HR was found to cost an individual PCP practice approximately $13,000.
That cost includes direct costs for resources and opportunity costs, the authors write. These costs could be offset by health care system savings.
“These costs included those for X-waiver training, which has been eliminated; thus, we expect this to cost less. Put another way, our findings inform ways to reinvest health care dollars as financial incentives for PCPs to adopt this new paradigm. Public health departments could provide grants or harm reduction kit supplies directly to PCPs to offset these costs as they do in some places with syringe service programs and/or increase Medicaid reimbursements for providing addiction care in primary care,” they write.
Data help make the case
Dinah Applewhite, MD, a primary care physician and addiction medicine specialist at Massachusetts General Hospital in Boston, who was not part of the study, said clinicians there have seen the benefits of integrating various aspects of addiction medicine into primary care but these data on outcomes and cost-effectiveness can help make the case to hospital leaders, legislators, and grant providers.
The primary care setting also provides a chance to engage patients around their injection practice and explore ways to minimize risk, she said.
“By offering them these kits, it lets them know your priority is their safety and well-being,” Dr. Applewhite said.
She noted that the linkage to primary care was low for patients who inject drugs, which speaks to the need for models in addition to this one, such as bringing primary care clinicians into syringe service programs.
“The medical establishment has a lot to learn from these programs,” she said.
Practices need support
She said it’s important to note that primary care practices need support from administrative leaders, philanthropists, and grant providers to help cover the costs.
“It’s one of the barriers to doing this,” she said. “There isn’t a mechanism to pay for this.”
Sarah Bagley, MD, a primary care physician at Boston Medical Center and medical director of BMC’s Center for Addiction Treatment for Adolescents/Young Adults Who Use Substances told this publication she was excited to see that the addition of harm reduction kits to buprenorphine seemed to have the optimal effect in improving outcomes. People with substance abuse disorders should feel they are welcome in primary care even if they are not yet ready to stop drug use, she said.
She said she was also glad to see increased life expectancy with these interventions. The news of overdose deaths contributing to a decrease in life expectancy can be overwhelming, she said.
But this study, she says, offers a road map for addressing the overdose crisis “by including harm reduction in the substance abuse care we provide.”
She pointed out that the study showed that costs increase per patient with both interventions, compared with the status quo. The study found that health care costs per person during a lifetime increased, compared with the status quo, by 69.1% for BUP and 74.3% for BUP plus HR.
But it’s important to understand the reason for that, she said: “The cost was higher because people were staying alive.”
She said it may help to compare giving optimal care to people who have substance abuse disorders with giving optimal care to people with other chronic conditions, such as diabetes, who may not always adhere to recommended diets or treatment regimens.
“We still invite those patients in and work with them based on where they are,” she said.
Growing epidemic
The researchers point to the urgent need for solutions given the U.S. opioid epidemic, which has led to increasing numbers of overdoses and injection drug use–related infections, such as infective endocarditis, and severe skin and soft tissue infections.
They point out that primary care providers are the largest clinical workforce in the United States, but few of their practices offer comprehensive addiction care onsite.
“Primary care practices are a practical place to integrate addiction services, where PCPs can prescribe buprenorphine and deliver harm reduction kits,” they write.
Coauthor Dr. Kimmel reports personal fees from Massachusetts Department of Public Health, Bureau of Substance Addiction Services Overdose Education and Prevention Program, and American Academy of Addiction Psychiatry, Opioid Response Network for harm reduction education outside the submitted work and previous consulting with Abt Associates on a Massachusetts Department of Public Health–funded project to improve access to medications for opioid use disorder treatment. Dr. Applewhite and Dr. Bagley report no relevant financial relationships.
JAMA NETWORK OPEN
ECT vs. ketamine for major depressive disorder: New data
“Although ECT is superior to ketamine for patients with a major depressive episode, our findings suggest that the therapeutic advantage may be smaller than what was demonstrated in prior analyses,” first author Vikas Menon, MD, department of psychiatry, Jawaharlal Institute of Postgraduate Medical Education & Research, Puducherry, India, told this news organization.
“This supports a recommendation for a trial of ketamine before a trial of ECT for patients with MDE, though this recommendation is limited by the small size and number of existing trials,” Dr. Menon said.
The study was published online in JAMA Psychiatry.
Questions remain
The meta-analysis included five trials of 278 adults with MDE (141 treated with ketamine and 137 with ECT).
In the main analysis, posttreatment depression ratings showed a trend for lower scores with ECT, compared with ketamine (standardized mean difference, −0.39; 95% confidence interval, −0.81 to 0.02).
In a sensitivity analysis of the two methodologically stronger trials, ECT was superior to ketamine (pooled SMD, −0.45; 95% CI, −0.75 to −0.14).
ECT was also superior to ketamine in terms of response rates (risk ratio, 1.27; 95% CI, 1.06-1.53) and remission rates (RR, 1.43; 95% CI, 1.12-1.82).
There were no significant between-group differences for number of sessions to response and remission and for cognitive outcomes.
Key limitations of the analysis were the small number of studies with limited sample sizes and a high risk of bias in all trials.
“There is a need for more comparative studies with adequate sample size in non-inferiority designs, examining a wider range of benefits and side effects and followed up for longer durations to answer clinically relevant questions about the nature and durability of observed benefits with ketamine,” said Dr. Menon.
“In patients with MDE for whom the administration of ECT is limited by restricted availability of the treatment, concerns about its cognitive adverse effects, negative patient attitudes, and other issues, clinicians may consider a trial of ketamine,” he added.
‘Important research’
Several experts offered perspective on the analysis in a statement from the U.K.-based nonprofit Science Media Centre, which was not involved with the conduct of this study.
Rupert McShane, MD, psychiatrist at the University of Oxford (England), noted that ECT and ketamine are both “potent” treatments for depression, and this meta-analysis shows that they are, “broadly speaking, equally as good as each other with perhaps a slight advantage for ECT.”
“Whether or not there is a difference depends on exactly how you define it and how you cut the data. Despite the slight advantage for ECT in this analysis, the authors support using ketamine before ECT, especially in patients who are worried about the cognitive risks of ECT. This seems sensible,” Dr. McShane said.
Allan Young, MBChB, clinical psychiatrist at King’s College London, noted that both ketamine and ECT have been shown to help some patients with treatment-resistant depression.
“Clearly the relative benefits of these two treatments need to be understood better, but this review of the existing literature suggests that ECT may benefit some more than ketamine,” said Mr. Young.
“There is evidence that ketamine with ECT may add little extra benefit, but much more work needs to be done to fully understand how these treatments fit best into the treatment pathway for major depressive episodes. However, based on this evidence, ECT clearly still merits a place in the treatment pathway,” Mr. Young added.
George Kirov, PhD, clinical professor, division of psychological medicine and clinical neurosciences, Cardiff University (England), said while the study is conducted well, most of the evidence is coming from one large trial conducted in Sweden.
“The other studies add small numbers of patients and the authors even present a sensitivity analysis after removing studies of poor quality, thus leaving only two studies and exposing even further the dependence of the results on one single study,” Dr. Kirov noted.
“The small studies should not be blamed for their size, as this is very difficult research to perform. On the other hand, the trends were in the same direction,” he added.
With those caveats in mind, Dr. Kirov said he still thinks this is “important research. It establishes the superiority of ECT against an active comparator (ketamine) which is very popular now and accepted to be quite effective.”
The study had no specific funding. Dr. Menon reports no relevant financial relationships. Dr. McShane is former chair of the ECT and Related Treatments Committee, Royal College of Psychiatrists and runs a ketamine clinic and an ECT service. Mr. Young has received compensation for lectures and advisory boards for AstraZeneca, Eli Lilly, Lundbeck, Sunovion, Servier, Livanova, Janssen, Allergan, Bionomics, Sumitomo Dainippon Pharma, COMPASS, and Sage, and has served as principal investigator on a trial of intranasal esketamine in treatment-resistant depression. Dr. Kirov has no interest to declare other than running the ECT service in Cardiff.
A version of this article first appeared on Medscape.com.
“Although ECT is superior to ketamine for patients with a major depressive episode, our findings suggest that the therapeutic advantage may be smaller than what was demonstrated in prior analyses,” first author Vikas Menon, MD, department of psychiatry, Jawaharlal Institute of Postgraduate Medical Education & Research, Puducherry, India, told this news organization.
“This supports a recommendation for a trial of ketamine before a trial of ECT for patients with MDE, though this recommendation is limited by the small size and number of existing trials,” Dr. Menon said.
The study was published online in JAMA Psychiatry.
Questions remain
The meta-analysis included five trials of 278 adults with MDE (141 treated with ketamine and 137 with ECT).
In the main analysis, posttreatment depression ratings showed a trend for lower scores with ECT, compared with ketamine (standardized mean difference, −0.39; 95% confidence interval, −0.81 to 0.02).
In a sensitivity analysis of the two methodologically stronger trials, ECT was superior to ketamine (pooled SMD, −0.45; 95% CI, −0.75 to −0.14).
ECT was also superior to ketamine in terms of response rates (risk ratio, 1.27; 95% CI, 1.06-1.53) and remission rates (RR, 1.43; 95% CI, 1.12-1.82).
There were no significant between-group differences for number of sessions to response and remission and for cognitive outcomes.
Key limitations of the analysis were the small number of studies with limited sample sizes and a high risk of bias in all trials.
“There is a need for more comparative studies with adequate sample size in non-inferiority designs, examining a wider range of benefits and side effects and followed up for longer durations to answer clinically relevant questions about the nature and durability of observed benefits with ketamine,” said Dr. Menon.
“In patients with MDE for whom the administration of ECT is limited by restricted availability of the treatment, concerns about its cognitive adverse effects, negative patient attitudes, and other issues, clinicians may consider a trial of ketamine,” he added.
‘Important research’
Several experts offered perspective on the analysis in a statement from the U.K.-based nonprofit Science Media Centre, which was not involved with the conduct of this study.
Rupert McShane, MD, psychiatrist at the University of Oxford (England), noted that ECT and ketamine are both “potent” treatments for depression, and this meta-analysis shows that they are, “broadly speaking, equally as good as each other with perhaps a slight advantage for ECT.”
“Whether or not there is a difference depends on exactly how you define it and how you cut the data. Despite the slight advantage for ECT in this analysis, the authors support using ketamine before ECT, especially in patients who are worried about the cognitive risks of ECT. This seems sensible,” Dr. McShane said.
Allan Young, MBChB, clinical psychiatrist at King’s College London, noted that both ketamine and ECT have been shown to help some patients with treatment-resistant depression.
“Clearly the relative benefits of these two treatments need to be understood better, but this review of the existing literature suggests that ECT may benefit some more than ketamine,” said Mr. Young.
“There is evidence that ketamine with ECT may add little extra benefit, but much more work needs to be done to fully understand how these treatments fit best into the treatment pathway for major depressive episodes. However, based on this evidence, ECT clearly still merits a place in the treatment pathway,” Mr. Young added.
George Kirov, PhD, clinical professor, division of psychological medicine and clinical neurosciences, Cardiff University (England), said while the study is conducted well, most of the evidence is coming from one large trial conducted in Sweden.
“The other studies add small numbers of patients and the authors even present a sensitivity analysis after removing studies of poor quality, thus leaving only two studies and exposing even further the dependence of the results on one single study,” Dr. Kirov noted.
“The small studies should not be blamed for their size, as this is very difficult research to perform. On the other hand, the trends were in the same direction,” he added.
With those caveats in mind, Dr. Kirov said he still thinks this is “important research. It establishes the superiority of ECT against an active comparator (ketamine) which is very popular now and accepted to be quite effective.”
The study had no specific funding. Dr. Menon reports no relevant financial relationships. Dr. McShane is former chair of the ECT and Related Treatments Committee, Royal College of Psychiatrists and runs a ketamine clinic and an ECT service. Mr. Young has received compensation for lectures and advisory boards for AstraZeneca, Eli Lilly, Lundbeck, Sunovion, Servier, Livanova, Janssen, Allergan, Bionomics, Sumitomo Dainippon Pharma, COMPASS, and Sage, and has served as principal investigator on a trial of intranasal esketamine in treatment-resistant depression. Dr. Kirov has no interest to declare other than running the ECT service in Cardiff.
A version of this article first appeared on Medscape.com.
“Although ECT is superior to ketamine for patients with a major depressive episode, our findings suggest that the therapeutic advantage may be smaller than what was demonstrated in prior analyses,” first author Vikas Menon, MD, department of psychiatry, Jawaharlal Institute of Postgraduate Medical Education & Research, Puducherry, India, told this news organization.
“This supports a recommendation for a trial of ketamine before a trial of ECT for patients with MDE, though this recommendation is limited by the small size and number of existing trials,” Dr. Menon said.
The study was published online in JAMA Psychiatry.
Questions remain
The meta-analysis included five trials of 278 adults with MDE (141 treated with ketamine and 137 with ECT).
In the main analysis, posttreatment depression ratings showed a trend for lower scores with ECT, compared with ketamine (standardized mean difference, −0.39; 95% confidence interval, −0.81 to 0.02).
In a sensitivity analysis of the two methodologically stronger trials, ECT was superior to ketamine (pooled SMD, −0.45; 95% CI, −0.75 to −0.14).
ECT was also superior to ketamine in terms of response rates (risk ratio, 1.27; 95% CI, 1.06-1.53) and remission rates (RR, 1.43; 95% CI, 1.12-1.82).
There were no significant between-group differences for number of sessions to response and remission and for cognitive outcomes.
Key limitations of the analysis were the small number of studies with limited sample sizes and a high risk of bias in all trials.
“There is a need for more comparative studies with adequate sample size in non-inferiority designs, examining a wider range of benefits and side effects and followed up for longer durations to answer clinically relevant questions about the nature and durability of observed benefits with ketamine,” said Dr. Menon.
“In patients with MDE for whom the administration of ECT is limited by restricted availability of the treatment, concerns about its cognitive adverse effects, negative patient attitudes, and other issues, clinicians may consider a trial of ketamine,” he added.
‘Important research’
Several experts offered perspective on the analysis in a statement from the U.K.-based nonprofit Science Media Centre, which was not involved with the conduct of this study.
Rupert McShane, MD, psychiatrist at the University of Oxford (England), noted that ECT and ketamine are both “potent” treatments for depression, and this meta-analysis shows that they are, “broadly speaking, equally as good as each other with perhaps a slight advantage for ECT.”
“Whether or not there is a difference depends on exactly how you define it and how you cut the data. Despite the slight advantage for ECT in this analysis, the authors support using ketamine before ECT, especially in patients who are worried about the cognitive risks of ECT. This seems sensible,” Dr. McShane said.
Allan Young, MBChB, clinical psychiatrist at King’s College London, noted that both ketamine and ECT have been shown to help some patients with treatment-resistant depression.
“Clearly the relative benefits of these two treatments need to be understood better, but this review of the existing literature suggests that ECT may benefit some more than ketamine,” said Mr. Young.
“There is evidence that ketamine with ECT may add little extra benefit, but much more work needs to be done to fully understand how these treatments fit best into the treatment pathway for major depressive episodes. However, based on this evidence, ECT clearly still merits a place in the treatment pathway,” Mr. Young added.
George Kirov, PhD, clinical professor, division of psychological medicine and clinical neurosciences, Cardiff University (England), said while the study is conducted well, most of the evidence is coming from one large trial conducted in Sweden.
“The other studies add small numbers of patients and the authors even present a sensitivity analysis after removing studies of poor quality, thus leaving only two studies and exposing even further the dependence of the results on one single study,” Dr. Kirov noted.
“The small studies should not be blamed for their size, as this is very difficult research to perform. On the other hand, the trends were in the same direction,” he added.
With those caveats in mind, Dr. Kirov said he still thinks this is “important research. It establishes the superiority of ECT against an active comparator (ketamine) which is very popular now and accepted to be quite effective.”
The study had no specific funding. Dr. Menon reports no relevant financial relationships. Dr. McShane is former chair of the ECT and Related Treatments Committee, Royal College of Psychiatrists and runs a ketamine clinic and an ECT service. Mr. Young has received compensation for lectures and advisory boards for AstraZeneca, Eli Lilly, Lundbeck, Sunovion, Servier, Livanova, Janssen, Allergan, Bionomics, Sumitomo Dainippon Pharma, COMPASS, and Sage, and has served as principal investigator on a trial of intranasal esketamine in treatment-resistant depression. Dr. Kirov has no interest to declare other than running the ECT service in Cardiff.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
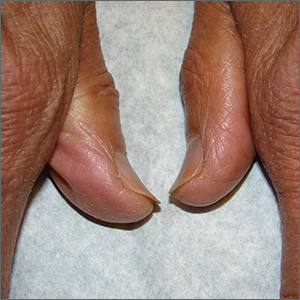
Brittle fingernails
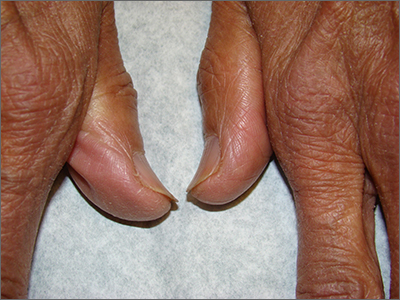
The abnormal upward curve to the fingernails was consistent with a diagnosis of koilonychia—otherwise known as spoon nails.
Koilonychia is an abnormal nail growth pattern where the distal nail matrix is depressed below its normal level, resulting in the spoon shape. The reverse, where the distal nail matrix is elevated in contrast to the proximal nail matrix, results in clubbing.1
There are multiple factors and diseases that result in koilonychia, including lichen planus, psoriasis, nutritional deficiencies (including iron deficiency anemia), and endocrinopathies.1 Lichen planus, which can cause koilonychia, often affects multiple nails and can also cause an associated central ridge pattern. Psoriasis may display a range of nail abnormalities; these include koilonychia, pitting onycholysis, and oil staining.
This patient did not have any signs or symptoms of psoriasis or lichen planus of her nails or skin. A review of her laboratory tests on file made no mention of anemia. Her chemistry profile—including liver tests, renal function tests, and protein levels—were all normal except for glucose levels, which was consistent with her prediabetes. Her thyroid function was also normal. No additional testing was performed since she had no symptoms, physical exam findings, or laboratory clues that pointed to other diseases or systemic processes.
The patient was advised to pick up over-the-counter nail strengtheners and to keep her fingernails trimmed short to minimize the likelihood of painful distal splitting that often occurs with brittle nails. Her physician advised her to follow up with the primary care team if she developed any new signs or symptoms.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Walker J, Baran R, Vélez N, et al. Koilonychia: an update on pathophysiology, differential diagnosis and clinical relevance. J Eur Acad Dermatol Venereol. 2016;30:1985-1991. doi: 10.1111/jdv.13610

The abnormal upward curve to the fingernails was consistent with a diagnosis of koilonychia—otherwise known as spoon nails.
Koilonychia is an abnormal nail growth pattern where the distal nail matrix is depressed below its normal level, resulting in the spoon shape. The reverse, where the distal nail matrix is elevated in contrast to the proximal nail matrix, results in clubbing.1
There are multiple factors and diseases that result in koilonychia, including lichen planus, psoriasis, nutritional deficiencies (including iron deficiency anemia), and endocrinopathies.1 Lichen planus, which can cause koilonychia, often affects multiple nails and can also cause an associated central ridge pattern. Psoriasis may display a range of nail abnormalities; these include koilonychia, pitting onycholysis, and oil staining.
This patient did not have any signs or symptoms of psoriasis or lichen planus of her nails or skin. A review of her laboratory tests on file made no mention of anemia. Her chemistry profile—including liver tests, renal function tests, and protein levels—were all normal except for glucose levels, which was consistent with her prediabetes. Her thyroid function was also normal. No additional testing was performed since she had no symptoms, physical exam findings, or laboratory clues that pointed to other diseases or systemic processes.
The patient was advised to pick up over-the-counter nail strengtheners and to keep her fingernails trimmed short to minimize the likelihood of painful distal splitting that often occurs with brittle nails. Her physician advised her to follow up with the primary care team if she developed any new signs or symptoms.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

The abnormal upward curve to the fingernails was consistent with a diagnosis of koilonychia—otherwise known as spoon nails.
Koilonychia is an abnormal nail growth pattern where the distal nail matrix is depressed below its normal level, resulting in the spoon shape. The reverse, where the distal nail matrix is elevated in contrast to the proximal nail matrix, results in clubbing.1
There are multiple factors and diseases that result in koilonychia, including lichen planus, psoriasis, nutritional deficiencies (including iron deficiency anemia), and endocrinopathies.1 Lichen planus, which can cause koilonychia, often affects multiple nails and can also cause an associated central ridge pattern. Psoriasis may display a range of nail abnormalities; these include koilonychia, pitting onycholysis, and oil staining.
This patient did not have any signs or symptoms of psoriasis or lichen planus of her nails or skin. A review of her laboratory tests on file made no mention of anemia. Her chemistry profile—including liver tests, renal function tests, and protein levels—were all normal except for glucose levels, which was consistent with her prediabetes. Her thyroid function was also normal. No additional testing was performed since she had no symptoms, physical exam findings, or laboratory clues that pointed to other diseases or systemic processes.
The patient was advised to pick up over-the-counter nail strengtheners and to keep her fingernails trimmed short to minimize the likelihood of painful distal splitting that often occurs with brittle nails. Her physician advised her to follow up with the primary care team if she developed any new signs or symptoms.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Walker J, Baran R, Vélez N, et al. Koilonychia: an update on pathophysiology, differential diagnosis and clinical relevance. J Eur Acad Dermatol Venereol. 2016;30:1985-1991. doi: 10.1111/jdv.13610
1. Walker J, Baran R, Vélez N, et al. Koilonychia: an update on pathophysiology, differential diagnosis and clinical relevance. J Eur Acad Dermatol Venereol. 2016;30:1985-1991. doi: 10.1111/jdv.13610
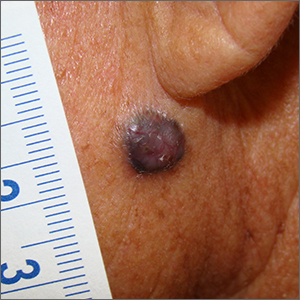
Dark facial lesion
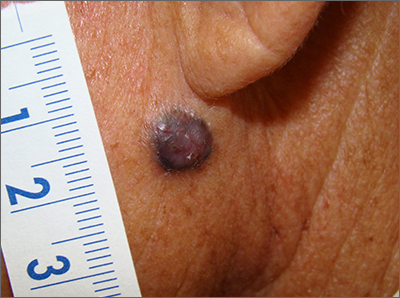
Although an elevated and pigmented lesion should be considered for possible melanoma, this one had prominent telangiectasias and was proven to be a basal cell carcinoma (BCC) on biopsy.
While the literature often focuses on light-colored skin types and the high risk of skin cancers, individuals with darker skin can also get melanoma and nonmelanoma skin cancer. Half of the BCCs in African American people are pigmented BCCs, compared to less than 10% for Caucasian individuals. Individuals who are Hispanic have twice the likelihood of pigmented BCCs as those who are Caucasian.1 Pigmented BCCs manifest as darker lesions, as occurred in this individual. Nonpigmented BCCs tend to be pink or pale in color.
Typically, superficial and very small, nodular BCCs can be successfully treated with 2 cycles of electrodesiccation and curettage. EDC should, however, be avoided in low-risk BCCs when these lesions occur in areas of secondary hair growth, such as the beard or scalp. This is because the epidermis follows the hair follicle, and in sites with deep hair follicles, EDC would have to get down to the subcutis to effectively clear the tumor.
For larger, nodular BCCs, full-thickness excision with adequate margins is warranted. For high-risk types, and those in high-risk areas near the nose, eyes, mouth, and ears, Mohs micrographic surgery is recommended to maximize the likelihood of complete excision while minimizing the loss of normal tissue.
Since the physician suspected this was a pigmented BCC, he performed a superficial shave biopsy on a small representative area of the lesion for diagnosis. This patient’s biopsy confirmed a nodular-type pigmented BCC. The lesion was removed in the office with 5-mm margins oriented along the resting skin tension lines with good closure and cosmetic results.
The patient was advised to have routine skin evaluations every 6 months due to the high risk of additional cancers. He was also advised to take oral niacinamide 500 mg twice daily, which can reduce the risk of actinic keratoses and nonmelanoma skin cancers by 15% and 23%, respectively, in those who have had lesions.2
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Higgins S, Nazemi A, Chow M, et al. Review of nonmelanoma skin cancer in African Americans, Hispanics, and Asians. Dermatol Surg. 2018;44:903-910. doi: 10.1097/DSS.0000000000001547
2. Starr P. Oral nicotinamide prevents common skin cancers in high-risk patients, reduces costs. Am Health Drug Benefits. 2015;8(spec issue):13-14.

Although an elevated and pigmented lesion should be considered for possible melanoma, this one had prominent telangiectasias and was proven to be a basal cell carcinoma (BCC) on biopsy.
While the literature often focuses on light-colored skin types and the high risk of skin cancers, individuals with darker skin can also get melanoma and nonmelanoma skin cancer. Half of the BCCs in African American people are pigmented BCCs, compared to less than 10% for Caucasian individuals. Individuals who are Hispanic have twice the likelihood of pigmented BCCs as those who are Caucasian.1 Pigmented BCCs manifest as darker lesions, as occurred in this individual. Nonpigmented BCCs tend to be pink or pale in color.
Typically, superficial and very small, nodular BCCs can be successfully treated with 2 cycles of electrodesiccation and curettage. EDC should, however, be avoided in low-risk BCCs when these lesions occur in areas of secondary hair growth, such as the beard or scalp. This is because the epidermis follows the hair follicle, and in sites with deep hair follicles, EDC would have to get down to the subcutis to effectively clear the tumor.
For larger, nodular BCCs, full-thickness excision with adequate margins is warranted. For high-risk types, and those in high-risk areas near the nose, eyes, mouth, and ears, Mohs micrographic surgery is recommended to maximize the likelihood of complete excision while minimizing the loss of normal tissue.
Since the physician suspected this was a pigmented BCC, he performed a superficial shave biopsy on a small representative area of the lesion for diagnosis. This patient’s biopsy confirmed a nodular-type pigmented BCC. The lesion was removed in the office with 5-mm margins oriented along the resting skin tension lines with good closure and cosmetic results.
The patient was advised to have routine skin evaluations every 6 months due to the high risk of additional cancers. He was also advised to take oral niacinamide 500 mg twice daily, which can reduce the risk of actinic keratoses and nonmelanoma skin cancers by 15% and 23%, respectively, in those who have had lesions.2
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

Although an elevated and pigmented lesion should be considered for possible melanoma, this one had prominent telangiectasias and was proven to be a basal cell carcinoma (BCC) on biopsy.
While the literature often focuses on light-colored skin types and the high risk of skin cancers, individuals with darker skin can also get melanoma and nonmelanoma skin cancer. Half of the BCCs in African American people are pigmented BCCs, compared to less than 10% for Caucasian individuals. Individuals who are Hispanic have twice the likelihood of pigmented BCCs as those who are Caucasian.1 Pigmented BCCs manifest as darker lesions, as occurred in this individual. Nonpigmented BCCs tend to be pink or pale in color.
Typically, superficial and very small, nodular BCCs can be successfully treated with 2 cycles of electrodesiccation and curettage. EDC should, however, be avoided in low-risk BCCs when these lesions occur in areas of secondary hair growth, such as the beard or scalp. This is because the epidermis follows the hair follicle, and in sites with deep hair follicles, EDC would have to get down to the subcutis to effectively clear the tumor.
For larger, nodular BCCs, full-thickness excision with adequate margins is warranted. For high-risk types, and those in high-risk areas near the nose, eyes, mouth, and ears, Mohs micrographic surgery is recommended to maximize the likelihood of complete excision while minimizing the loss of normal tissue.
Since the physician suspected this was a pigmented BCC, he performed a superficial shave biopsy on a small representative area of the lesion for diagnosis. This patient’s biopsy confirmed a nodular-type pigmented BCC. The lesion was removed in the office with 5-mm margins oriented along the resting skin tension lines with good closure and cosmetic results.
The patient was advised to have routine skin evaluations every 6 months due to the high risk of additional cancers. He was also advised to take oral niacinamide 500 mg twice daily, which can reduce the risk of actinic keratoses and nonmelanoma skin cancers by 15% and 23%, respectively, in those who have had lesions.2
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Higgins S, Nazemi A, Chow M, et al. Review of nonmelanoma skin cancer in African Americans, Hispanics, and Asians. Dermatol Surg. 2018;44:903-910. doi: 10.1097/DSS.0000000000001547
2. Starr P. Oral nicotinamide prevents common skin cancers in high-risk patients, reduces costs. Am Health Drug Benefits. 2015;8(spec issue):13-14.
1. Higgins S, Nazemi A, Chow M, et al. Review of nonmelanoma skin cancer in African Americans, Hispanics, and Asians. Dermatol Surg. 2018;44:903-910. doi: 10.1097/DSS.0000000000001547
2. Starr P. Oral nicotinamide prevents common skin cancers in high-risk patients, reduces costs. Am Health Drug Benefits. 2015;8(spec issue):13-14.
Long COVID: ‘On par’ with heart disease, cancer, book says
Filmmaker Gez Medinger and immunologist Danny Altmann have been dubbed by British media as “COVID’s odd couple,” and they don’t mind at all. Discussing their recent book, The Long COVID Handbook, the authors lean into their animated roles: Medinger is a passionate patient-researcher and “guinea pig” (his words) in search of his own healing, and Altmann is a no-nonsense, data-driven scientist and “Professor Boring” (as he puts it).
And the message they have about the impact of long COVID is stunning.
“The clinical burden [of long COVID] is somewhere on par with the whole of heart disease all over again, or the whole of oncology all over again, which are our biggest clinical bills concurrently,” Altmann said.
The pair met early in the pandemic, after Medinger became infected during the first wave and interviewed Altmann for his YouTube channel, which has more than 5 million views.
“Danny was one of the first people from the medical establishment to sort of stand up on the parapet and wave a flag and say, ‘Hey, guys, there’s a problem here.’ And that was incredibly validating for 2 million people in the U.K. alone who were suffering with long COVID,” Medinger said.
Their relationship works, not just for publishing one of the first definitive guides to long COVID, but also as a model for how patients with lived experiences can lead the way in medicine – from giving the condition its name to driving the medical establishment for recognition, clinical research, and therapeutic answers.
With Altmann currently leading a major research project at Imperial College London on long COVID and Medinger’s social media platform and communication skills, they’re both advancing the world’s understanding of the disease in their own way.
“We’re now more than 3 years into this completely mysterious, uncharted disease process with a whole globe full of really desperate people,” said Altmann. “It’s a living, organic thing, and yet that also demands some kind of order and collation and pulling together into some kind of sense. So I was very pleased when Gez approached me to help him with the book.”
In it, they translate everything they’ve learned about the condition that’s “scattered in 100,000 places around the globe” into a digestible format. It tells two sides of the same story: the anecdotal experiences Medinger has undergone or observed in the long COVID community through more than a dozen of his own patient-led studies, as well the hard science and research that’s amassing in the medical world.
In an interview,
What are the book’s key takeaways for you?
Medinger: “I would say we put together an incredibly comprehensive couple of chapters on the hypotheses, big picture, what’s causing long COVID. And then the nitty-gritty research for everything that we’ve found out that is going on. ... And the other part of the book that I think is particularly important, beyond the tips for managing symptoms, is the content on mental health and the impact on your emotional state and your capacity and just how huge that is. ... That has been the most powerful thing for patients when they’ve read it. And they’ve said that they’ve just been crying all the way through those chapters because suddenly they feel heard and seen.”
Altmann: “Obviously, you’d expect me to say that the parts of the book that I love most are the kind of hard-nosed, medical, mechanistic bits. ... We’ve got 150 million-plus desperate people deciding or not deciding to go and see their general practitioner, getting a fair hearing or not getting a fair hearing. And the poor doctor has never learned this in medical school, has never read a textbook on it, and hasn’t a clue what’s coming through the door.
How are they expected to know what to do? So I think the least we can do in some of those chapters is feed into their knowledge of general medicine and give them some clues. ... I think if we can explain to people what might be going on in them, and to their doctors, what on earth they might do about it, what kind of tests they might order, that helps a bit.”
How did you balance the more controversial parts of the book, including the chapter about so-called “treatments”? For instance, the book recounts Gez’s harrowing experience with ivermectin as a frightening warning. But Danny, you were nervous about even mentioning unproven and potentially dangerous treatments as things people have tried and have looked into.
Medinger: “We had to try and work out how to handle the topic, how to handle those points of view, whilst at the same time still being informative. I think the book is stronger for that chapter, too. The other thing would certainly have been to just not address the subject, but it’s one of the things that people want to know the most about. And there’s also a lot of bad information floating around out there about certain treatments. Ivermectin, for example, and this is what happened to me when I tried it. ‘Don’t do it. It’s not recommended. Please don’t.’
I think it was also very important to include because that cautionary tale really applies to every single one of those treatments that people might be hearing about that hasn’t been backed up by efficacy and safety studies.”
Altmann: “I think Gez has been quite diplomatic. That chapter was, I think, a testament to the power of the book. And the biggest test of our marriage as ‘the odd couple.’ Because when I first read the first draft of what Gez had written, I said, ‘my name can’t even be on this book. Otherwise, I’ll be sacked.’
And we had to find marriage counseling after that, and a way back to write a version of that chapter that expressed both halves of those concerns in a way that did justice to those different viewpoints. And I think that makes it quite a strong chapter.”
What do you think are the most urgent next steps in the search for solving long COVID?
Medinger: “I would personally like to try and get some sort of answer on viral persistence. ... If there’s one thing that feels like it would be treatable in theory, and would make sense why we’re still getting all of these symptoms this whole time later, it’s that, so I would like to try and establish or eliminate viral persistence. So if you gave me Elon Musk’s wealth, that’s what I would throw a bunch of the money at, trying to either eliminate or establish that.
And then, you know, the other important thing is a diagnostic test. Danny always talks about how important it is. Once you have that, it helps you suddenly open the doors to all these other things that you can do. And treatment trials. Let’s throw some meds at this so that we have an educated guess at what might work and put them into high-powered, randomized, controlled trials and see if anything comes out because from the patient perspective, I don’t think any of us wants to wait for 5 years for that stuff to start happening.”
Altmann: “I completely agree. If you go to a website, like clinicaltrials.gov, you’ll find an immense number of clinical trials on COVID. There isn’t really a shortage of them, some of them better-powered to get an answer than others.”
How do you think public policy needs to adapt for long COVID, including social safety nets such as workers’ compensation and disability benefits?
Medinger: “In terms of public policy, what I would like would be some public acknowledgment that it’s real from government sources. Just the acknowledgment that it’s real and it remains a risk even now.”
Altmann: “Nobody in politics asks my opinion. I think they’d hate to hear it. Because if I went to see them and said, well, actually, if you thought the COVID pandemic was bad, wait till you see what’s on the table now. We’ve created a disabled population in our country of 2 million, at least a portion if not more of people who are not fully contributory to the workforce anymore ... [with] legal wrangles about retirement and health insurance and pensions, and a human right to adequate health care. Which means, ideally, a purpose-built clinic where they can have their respiratory opinion and their rheumatology opinion and their endocrine opinion and their neurology opinion, all under one roof.”
You’ve both shown so much optimism. Why is that?
Altmann: “I’ve been an immunologist for a long time now, and written all my decades of grant applications, where as a community we made what, at the time, were kind of wild promises and wildly optimistic projections of how our knowledge of tumor immunity would revolutionize cancer care, and how knowledge of autoimmunity would revolutionize care of all the autoimmune diseases.
And weirdly almost every word we wrote over those 25 or 30 years came true. Cancer immunotherapy was revolutionized, and biologics for diabetes, multiple sclerosis, and arthritis were revolutionized. So if I have faith that those things came true, I have complete faith in this as well.”
Medinger: “From the patient perspective, what I would say is that we are seeing people who’ve been ill for more than 2 years recover. People are suddenly turning the corner when they might not have expected to.
And while we don’t quite know exactly why yet, and it’s not everyone, every single time I hear the story of someone saying, ‘I’m pretty much back to where I was, I feel like I’ve recovered,’ I feel great. Even if I haven’t. Because I know that every single time I hear someone say that, that just increases the probability that I will, too.”
A version of this article first appeared on WebMD.com.
Filmmaker Gez Medinger and immunologist Danny Altmann have been dubbed by British media as “COVID’s odd couple,” and they don’t mind at all. Discussing their recent book, The Long COVID Handbook, the authors lean into their animated roles: Medinger is a passionate patient-researcher and “guinea pig” (his words) in search of his own healing, and Altmann is a no-nonsense, data-driven scientist and “Professor Boring” (as he puts it).
And the message they have about the impact of long COVID is stunning.
“The clinical burden [of long COVID] is somewhere on par with the whole of heart disease all over again, or the whole of oncology all over again, which are our biggest clinical bills concurrently,” Altmann said.
The pair met early in the pandemic, after Medinger became infected during the first wave and interviewed Altmann for his YouTube channel, which has more than 5 million views.
“Danny was one of the first people from the medical establishment to sort of stand up on the parapet and wave a flag and say, ‘Hey, guys, there’s a problem here.’ And that was incredibly validating for 2 million people in the U.K. alone who were suffering with long COVID,” Medinger said.
Their relationship works, not just for publishing one of the first definitive guides to long COVID, but also as a model for how patients with lived experiences can lead the way in medicine – from giving the condition its name to driving the medical establishment for recognition, clinical research, and therapeutic answers.
With Altmann currently leading a major research project at Imperial College London on long COVID and Medinger’s social media platform and communication skills, they’re both advancing the world’s understanding of the disease in their own way.
“We’re now more than 3 years into this completely mysterious, uncharted disease process with a whole globe full of really desperate people,” said Altmann. “It’s a living, organic thing, and yet that also demands some kind of order and collation and pulling together into some kind of sense. So I was very pleased when Gez approached me to help him with the book.”
In it, they translate everything they’ve learned about the condition that’s “scattered in 100,000 places around the globe” into a digestible format. It tells two sides of the same story: the anecdotal experiences Medinger has undergone or observed in the long COVID community through more than a dozen of his own patient-led studies, as well the hard science and research that’s amassing in the medical world.
In an interview,
What are the book’s key takeaways for you?
Medinger: “I would say we put together an incredibly comprehensive couple of chapters on the hypotheses, big picture, what’s causing long COVID. And then the nitty-gritty research for everything that we’ve found out that is going on. ... And the other part of the book that I think is particularly important, beyond the tips for managing symptoms, is the content on mental health and the impact on your emotional state and your capacity and just how huge that is. ... That has been the most powerful thing for patients when they’ve read it. And they’ve said that they’ve just been crying all the way through those chapters because suddenly they feel heard and seen.”
Altmann: “Obviously, you’d expect me to say that the parts of the book that I love most are the kind of hard-nosed, medical, mechanistic bits. ... We’ve got 150 million-plus desperate people deciding or not deciding to go and see their general practitioner, getting a fair hearing or not getting a fair hearing. And the poor doctor has never learned this in medical school, has never read a textbook on it, and hasn’t a clue what’s coming through the door.
How are they expected to know what to do? So I think the least we can do in some of those chapters is feed into their knowledge of general medicine and give them some clues. ... I think if we can explain to people what might be going on in them, and to their doctors, what on earth they might do about it, what kind of tests they might order, that helps a bit.”
How did you balance the more controversial parts of the book, including the chapter about so-called “treatments”? For instance, the book recounts Gez’s harrowing experience with ivermectin as a frightening warning. But Danny, you were nervous about even mentioning unproven and potentially dangerous treatments as things people have tried and have looked into.
Medinger: “We had to try and work out how to handle the topic, how to handle those points of view, whilst at the same time still being informative. I think the book is stronger for that chapter, too. The other thing would certainly have been to just not address the subject, but it’s one of the things that people want to know the most about. And there’s also a lot of bad information floating around out there about certain treatments. Ivermectin, for example, and this is what happened to me when I tried it. ‘Don’t do it. It’s not recommended. Please don’t.’
I think it was also very important to include because that cautionary tale really applies to every single one of those treatments that people might be hearing about that hasn’t been backed up by efficacy and safety studies.”
Altmann: “I think Gez has been quite diplomatic. That chapter was, I think, a testament to the power of the book. And the biggest test of our marriage as ‘the odd couple.’ Because when I first read the first draft of what Gez had written, I said, ‘my name can’t even be on this book. Otherwise, I’ll be sacked.’
And we had to find marriage counseling after that, and a way back to write a version of that chapter that expressed both halves of those concerns in a way that did justice to those different viewpoints. And I think that makes it quite a strong chapter.”
What do you think are the most urgent next steps in the search for solving long COVID?
Medinger: “I would personally like to try and get some sort of answer on viral persistence. ... If there’s one thing that feels like it would be treatable in theory, and would make sense why we’re still getting all of these symptoms this whole time later, it’s that, so I would like to try and establish or eliminate viral persistence. So if you gave me Elon Musk’s wealth, that’s what I would throw a bunch of the money at, trying to either eliminate or establish that.
And then, you know, the other important thing is a diagnostic test. Danny always talks about how important it is. Once you have that, it helps you suddenly open the doors to all these other things that you can do. And treatment trials. Let’s throw some meds at this so that we have an educated guess at what might work and put them into high-powered, randomized, controlled trials and see if anything comes out because from the patient perspective, I don’t think any of us wants to wait for 5 years for that stuff to start happening.”
Altmann: “I completely agree. If you go to a website, like clinicaltrials.gov, you’ll find an immense number of clinical trials on COVID. There isn’t really a shortage of them, some of them better-powered to get an answer than others.”
How do you think public policy needs to adapt for long COVID, including social safety nets such as workers’ compensation and disability benefits?
Medinger: “In terms of public policy, what I would like would be some public acknowledgment that it’s real from government sources. Just the acknowledgment that it’s real and it remains a risk even now.”
Altmann: “Nobody in politics asks my opinion. I think they’d hate to hear it. Because if I went to see them and said, well, actually, if you thought the COVID pandemic was bad, wait till you see what’s on the table now. We’ve created a disabled population in our country of 2 million, at least a portion if not more of people who are not fully contributory to the workforce anymore ... [with] legal wrangles about retirement and health insurance and pensions, and a human right to adequate health care. Which means, ideally, a purpose-built clinic where they can have their respiratory opinion and their rheumatology opinion and their endocrine opinion and their neurology opinion, all under one roof.”
You’ve both shown so much optimism. Why is that?
Altmann: “I’ve been an immunologist for a long time now, and written all my decades of grant applications, where as a community we made what, at the time, were kind of wild promises and wildly optimistic projections of how our knowledge of tumor immunity would revolutionize cancer care, and how knowledge of autoimmunity would revolutionize care of all the autoimmune diseases.
And weirdly almost every word we wrote over those 25 or 30 years came true. Cancer immunotherapy was revolutionized, and biologics for diabetes, multiple sclerosis, and arthritis were revolutionized. So if I have faith that those things came true, I have complete faith in this as well.”
Medinger: “From the patient perspective, what I would say is that we are seeing people who’ve been ill for more than 2 years recover. People are suddenly turning the corner when they might not have expected to.
And while we don’t quite know exactly why yet, and it’s not everyone, every single time I hear the story of someone saying, ‘I’m pretty much back to where I was, I feel like I’ve recovered,’ I feel great. Even if I haven’t. Because I know that every single time I hear someone say that, that just increases the probability that I will, too.”
A version of this article first appeared on WebMD.com.
Filmmaker Gez Medinger and immunologist Danny Altmann have been dubbed by British media as “COVID’s odd couple,” and they don’t mind at all. Discussing their recent book, The Long COVID Handbook, the authors lean into their animated roles: Medinger is a passionate patient-researcher and “guinea pig” (his words) in search of his own healing, and Altmann is a no-nonsense, data-driven scientist and “Professor Boring” (as he puts it).
And the message they have about the impact of long COVID is stunning.
“The clinical burden [of long COVID] is somewhere on par with the whole of heart disease all over again, or the whole of oncology all over again, which are our biggest clinical bills concurrently,” Altmann said.
The pair met early in the pandemic, after Medinger became infected during the first wave and interviewed Altmann for his YouTube channel, which has more than 5 million views.
“Danny was one of the first people from the medical establishment to sort of stand up on the parapet and wave a flag and say, ‘Hey, guys, there’s a problem here.’ And that was incredibly validating for 2 million people in the U.K. alone who were suffering with long COVID,” Medinger said.
Their relationship works, not just for publishing one of the first definitive guides to long COVID, but also as a model for how patients with lived experiences can lead the way in medicine – from giving the condition its name to driving the medical establishment for recognition, clinical research, and therapeutic answers.
With Altmann currently leading a major research project at Imperial College London on long COVID and Medinger’s social media platform and communication skills, they’re both advancing the world’s understanding of the disease in their own way.
“We’re now more than 3 years into this completely mysterious, uncharted disease process with a whole globe full of really desperate people,” said Altmann. “It’s a living, organic thing, and yet that also demands some kind of order and collation and pulling together into some kind of sense. So I was very pleased when Gez approached me to help him with the book.”
In it, they translate everything they’ve learned about the condition that’s “scattered in 100,000 places around the globe” into a digestible format. It tells two sides of the same story: the anecdotal experiences Medinger has undergone or observed in the long COVID community through more than a dozen of his own patient-led studies, as well the hard science and research that’s amassing in the medical world.
In an interview,
What are the book’s key takeaways for you?
Medinger: “I would say we put together an incredibly comprehensive couple of chapters on the hypotheses, big picture, what’s causing long COVID. And then the nitty-gritty research for everything that we’ve found out that is going on. ... And the other part of the book that I think is particularly important, beyond the tips for managing symptoms, is the content on mental health and the impact on your emotional state and your capacity and just how huge that is. ... That has been the most powerful thing for patients when they’ve read it. And they’ve said that they’ve just been crying all the way through those chapters because suddenly they feel heard and seen.”
Altmann: “Obviously, you’d expect me to say that the parts of the book that I love most are the kind of hard-nosed, medical, mechanistic bits. ... We’ve got 150 million-plus desperate people deciding or not deciding to go and see their general practitioner, getting a fair hearing or not getting a fair hearing. And the poor doctor has never learned this in medical school, has never read a textbook on it, and hasn’t a clue what’s coming through the door.
How are they expected to know what to do? So I think the least we can do in some of those chapters is feed into their knowledge of general medicine and give them some clues. ... I think if we can explain to people what might be going on in them, and to their doctors, what on earth they might do about it, what kind of tests they might order, that helps a bit.”
How did you balance the more controversial parts of the book, including the chapter about so-called “treatments”? For instance, the book recounts Gez’s harrowing experience with ivermectin as a frightening warning. But Danny, you were nervous about even mentioning unproven and potentially dangerous treatments as things people have tried and have looked into.
Medinger: “We had to try and work out how to handle the topic, how to handle those points of view, whilst at the same time still being informative. I think the book is stronger for that chapter, too. The other thing would certainly have been to just not address the subject, but it’s one of the things that people want to know the most about. And there’s also a lot of bad information floating around out there about certain treatments. Ivermectin, for example, and this is what happened to me when I tried it. ‘Don’t do it. It’s not recommended. Please don’t.’
I think it was also very important to include because that cautionary tale really applies to every single one of those treatments that people might be hearing about that hasn’t been backed up by efficacy and safety studies.”
Altmann: “I think Gez has been quite diplomatic. That chapter was, I think, a testament to the power of the book. And the biggest test of our marriage as ‘the odd couple.’ Because when I first read the first draft of what Gez had written, I said, ‘my name can’t even be on this book. Otherwise, I’ll be sacked.’
And we had to find marriage counseling after that, and a way back to write a version of that chapter that expressed both halves of those concerns in a way that did justice to those different viewpoints. And I think that makes it quite a strong chapter.”
What do you think are the most urgent next steps in the search for solving long COVID?
Medinger: “I would personally like to try and get some sort of answer on viral persistence. ... If there’s one thing that feels like it would be treatable in theory, and would make sense why we’re still getting all of these symptoms this whole time later, it’s that, so I would like to try and establish or eliminate viral persistence. So if you gave me Elon Musk’s wealth, that’s what I would throw a bunch of the money at, trying to either eliminate or establish that.
And then, you know, the other important thing is a diagnostic test. Danny always talks about how important it is. Once you have that, it helps you suddenly open the doors to all these other things that you can do. And treatment trials. Let’s throw some meds at this so that we have an educated guess at what might work and put them into high-powered, randomized, controlled trials and see if anything comes out because from the patient perspective, I don’t think any of us wants to wait for 5 years for that stuff to start happening.”
Altmann: “I completely agree. If you go to a website, like clinicaltrials.gov, you’ll find an immense number of clinical trials on COVID. There isn’t really a shortage of them, some of them better-powered to get an answer than others.”
How do you think public policy needs to adapt for long COVID, including social safety nets such as workers’ compensation and disability benefits?
Medinger: “In terms of public policy, what I would like would be some public acknowledgment that it’s real from government sources. Just the acknowledgment that it’s real and it remains a risk even now.”
Altmann: “Nobody in politics asks my opinion. I think they’d hate to hear it. Because if I went to see them and said, well, actually, if you thought the COVID pandemic was bad, wait till you see what’s on the table now. We’ve created a disabled population in our country of 2 million, at least a portion if not more of people who are not fully contributory to the workforce anymore ... [with] legal wrangles about retirement and health insurance and pensions, and a human right to adequate health care. Which means, ideally, a purpose-built clinic where they can have their respiratory opinion and their rheumatology opinion and their endocrine opinion and their neurology opinion, all under one roof.”
You’ve both shown so much optimism. Why is that?
Altmann: “I’ve been an immunologist for a long time now, and written all my decades of grant applications, where as a community we made what, at the time, were kind of wild promises and wildly optimistic projections of how our knowledge of tumor immunity would revolutionize cancer care, and how knowledge of autoimmunity would revolutionize care of all the autoimmune diseases.
And weirdly almost every word we wrote over those 25 or 30 years came true. Cancer immunotherapy was revolutionized, and biologics for diabetes, multiple sclerosis, and arthritis were revolutionized. So if I have faith that those things came true, I have complete faith in this as well.”
Medinger: “From the patient perspective, what I would say is that we are seeing people who’ve been ill for more than 2 years recover. People are suddenly turning the corner when they might not have expected to.
And while we don’t quite know exactly why yet, and it’s not everyone, every single time I hear the story of someone saying, ‘I’m pretty much back to where I was, I feel like I’ve recovered,’ I feel great. Even if I haven’t. Because I know that every single time I hear someone say that, that just increases the probability that I will, too.”
A version of this article first appeared on WebMD.com.
Urban green and blue spaces linked to less psychological distress
The findings of the study, which was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology, build on a growing understanding of the relationship between types and qualities of urban environments and dementia risk.
Adithya Vegaraju, a student at Washington State University, Spokane, led the study, which looked at data from the Washington State Behavioral Risk Factor Surveillance System to assess prevalence of serious psychological distress among 42,980 Washington state residents aged 65 and over.
The data, collected between 2011 and 2019, used a self-reported questionnaire to determine serious psychological distress, which is defined as a level of mental distress considered debilitating enough to warrant treatment.
Mr. Vegaraju and his coauthor Solmaz Amiri, DDes, also of Washington State University, used ZIP codes, along with U.S. census data, to approximate the urban adults’ proximity to green and blue spaces.
After controlling for potential confounders of age, sex, ethnicity, education, and marital status, the investigators found that people living within half a mile of green or blue spaces had a 17% lower risk of experiencing serious psychological distress, compared with people living farther from these spaces, the investigators said in a news release.
Implications for cognitive decline and dementia?
Psychological distress in adults has been linked in population-based longitudinal studies to later cognitive decline and dementia. One study in older adults found the risk of dementia to be more than 50% higher among adults aged 50-70 with persistent depression. Blue and green spaces have also been investigated in relation to neurodegenerative disease among older adults; a 2022 study looking at data from some 62 million Medicare beneficiaries found those living in areas with more vegetation saw lower risk of hospitalizations for Alzheimer’s disease and related dementias.
“Since we lack effective prevention methods or treatments for mild cognitive impairment and dementia, we need to get creative in how we look at these issues,” Dr. Amiri commented in a press statement about her and Mr. Vegaraju’s findings. “Our hope is that this study showing better mental health among people living close to parks and water will trigger other studies about how these benefits work and whether this proximity can help prevent or delay mild cognitive impairment and dementia.”
The investigators acknowledged that their findings were limited by reliance on a self-reported measure of psychological distress.
A bidirectional connection with depression and dementia
In a comment, Anjum Hajat, PhD, an epidemiologist at University of Washington School of Public Health in Seattle who has also studied the relationship between green space and dementia risk in older adults, noted some further apparent limitations of the new study, for which only an abstract was available at publication.
“It has been shown that people with depression are at higher risk for dementia, but the opposite is also true,” Dr. Hajat commented. “Those with dementia are more likely to develop depression. This bidirectionality makes this study abstract difficult to interpret since the study is based on cross-sectional data: Individuals are not followed over time to see which develops first, dementia or depression.”
Additionally, Dr. Hajat noted, the data used to determine proximity to green and blue spaces did not allow for the calculation of precise distances between subjects’ homes and these spaces.
Mr. Vegaraju and Dr. Amiri’s study had no outside support, and the investigators declared no conflicts of interest. Dr. Hajat declared no conflicts of interest.
The findings of the study, which was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology, build on a growing understanding of the relationship between types and qualities of urban environments and dementia risk.
Adithya Vegaraju, a student at Washington State University, Spokane, led the study, which looked at data from the Washington State Behavioral Risk Factor Surveillance System to assess prevalence of serious psychological distress among 42,980 Washington state residents aged 65 and over.
The data, collected between 2011 and 2019, used a self-reported questionnaire to determine serious psychological distress, which is defined as a level of mental distress considered debilitating enough to warrant treatment.
Mr. Vegaraju and his coauthor Solmaz Amiri, DDes, also of Washington State University, used ZIP codes, along with U.S. census data, to approximate the urban adults’ proximity to green and blue spaces.
After controlling for potential confounders of age, sex, ethnicity, education, and marital status, the investigators found that people living within half a mile of green or blue spaces had a 17% lower risk of experiencing serious psychological distress, compared with people living farther from these spaces, the investigators said in a news release.
Implications for cognitive decline and dementia?
Psychological distress in adults has been linked in population-based longitudinal studies to later cognitive decline and dementia. One study in older adults found the risk of dementia to be more than 50% higher among adults aged 50-70 with persistent depression. Blue and green spaces have also been investigated in relation to neurodegenerative disease among older adults; a 2022 study looking at data from some 62 million Medicare beneficiaries found those living in areas with more vegetation saw lower risk of hospitalizations for Alzheimer’s disease and related dementias.
“Since we lack effective prevention methods or treatments for mild cognitive impairment and dementia, we need to get creative in how we look at these issues,” Dr. Amiri commented in a press statement about her and Mr. Vegaraju’s findings. “Our hope is that this study showing better mental health among people living close to parks and water will trigger other studies about how these benefits work and whether this proximity can help prevent or delay mild cognitive impairment and dementia.”
The investigators acknowledged that their findings were limited by reliance on a self-reported measure of psychological distress.
A bidirectional connection with depression and dementia
In a comment, Anjum Hajat, PhD, an epidemiologist at University of Washington School of Public Health in Seattle who has also studied the relationship between green space and dementia risk in older adults, noted some further apparent limitations of the new study, for which only an abstract was available at publication.
“It has been shown that people with depression are at higher risk for dementia, but the opposite is also true,” Dr. Hajat commented. “Those with dementia are more likely to develop depression. This bidirectionality makes this study abstract difficult to interpret since the study is based on cross-sectional data: Individuals are not followed over time to see which develops first, dementia or depression.”
Additionally, Dr. Hajat noted, the data used to determine proximity to green and blue spaces did not allow for the calculation of precise distances between subjects’ homes and these spaces.
Mr. Vegaraju and Dr. Amiri’s study had no outside support, and the investigators declared no conflicts of interest. Dr. Hajat declared no conflicts of interest.
The findings of the study, which was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology, build on a growing understanding of the relationship between types and qualities of urban environments and dementia risk.
Adithya Vegaraju, a student at Washington State University, Spokane, led the study, which looked at data from the Washington State Behavioral Risk Factor Surveillance System to assess prevalence of serious psychological distress among 42,980 Washington state residents aged 65 and over.
The data, collected between 2011 and 2019, used a self-reported questionnaire to determine serious psychological distress, which is defined as a level of mental distress considered debilitating enough to warrant treatment.
Mr. Vegaraju and his coauthor Solmaz Amiri, DDes, also of Washington State University, used ZIP codes, along with U.S. census data, to approximate the urban adults’ proximity to green and blue spaces.
After controlling for potential confounders of age, sex, ethnicity, education, and marital status, the investigators found that people living within half a mile of green or blue spaces had a 17% lower risk of experiencing serious psychological distress, compared with people living farther from these spaces, the investigators said in a news release.
Implications for cognitive decline and dementia?
Psychological distress in adults has been linked in population-based longitudinal studies to later cognitive decline and dementia. One study in older adults found the risk of dementia to be more than 50% higher among adults aged 50-70 with persistent depression. Blue and green spaces have also been investigated in relation to neurodegenerative disease among older adults; a 2022 study looking at data from some 62 million Medicare beneficiaries found those living in areas with more vegetation saw lower risk of hospitalizations for Alzheimer’s disease and related dementias.
“Since we lack effective prevention methods or treatments for mild cognitive impairment and dementia, we need to get creative in how we look at these issues,” Dr. Amiri commented in a press statement about her and Mr. Vegaraju’s findings. “Our hope is that this study showing better mental health among people living close to parks and water will trigger other studies about how these benefits work and whether this proximity can help prevent or delay mild cognitive impairment and dementia.”
The investigators acknowledged that their findings were limited by reliance on a self-reported measure of psychological distress.
A bidirectional connection with depression and dementia
In a comment, Anjum Hajat, PhD, an epidemiologist at University of Washington School of Public Health in Seattle who has also studied the relationship between green space and dementia risk in older adults, noted some further apparent limitations of the new study, for which only an abstract was available at publication.
“It has been shown that people with depression are at higher risk for dementia, but the opposite is also true,” Dr. Hajat commented. “Those with dementia are more likely to develop depression. This bidirectionality makes this study abstract difficult to interpret since the study is based on cross-sectional data: Individuals are not followed over time to see which develops first, dementia or depression.”
Additionally, Dr. Hajat noted, the data used to determine proximity to green and blue spaces did not allow for the calculation of precise distances between subjects’ homes and these spaces.
Mr. Vegaraju and Dr. Amiri’s study had no outside support, and the investigators declared no conflicts of interest. Dr. Hajat declared no conflicts of interest.
FROM AAN 2023
NSAID use in diabetes may worsen risk for first HF hospitalization
suggests a prospective, controlled study.
Certain subgroups may account for much of the excess risk, the results suggest, including the very elderly, patients with uncontrolled diabetes, those prescribed an NSAID for the first time, and patients already taking both a renin-angiotensin system inhibitor (RASi) and a diuretic.
Such patients with a firm indication for NSAIDs potentially could “be the ones benefiting most from closer follow-up, reduced dosage, or other mitigation strategies,” Anders Holt, MD, said in an interview.
Dr. Holt, of Copenhagen University Hospital and Herlev-Gentofte Hospital in Hellerup, Denmark, is lead author on the analysis of Danish registry data published in the Journal of the American College of Cardiology. He presented essentially the same results in preliminary form at the 2022 annual congress of the European Society of Cardiology.
HF hospitalizations linked to NSAIDs, the published report notes, are often attributed to symptoms from temporary fluid overload, often without worsening cardiac function, that stem from the drugs’ renal effects.
“One could speculate,” Dr. Holt said, that such HF events might be less severe and even associated with better outcomes, compared with other forms of heart failure.
But the current analysis provides a hint to the contrary, he observed. The 5-year mortality was similar for patients with HF linked to NSAIDs and those with other forms of HF, “which could suggest that NSAID-associated heart failure is more than transient fluid overload.”
The drugs may promote HF through direct effects on the heart by any of several proposed mechanisms, including “induction of arrhythmias and heart fibrosis, vasoconstriction, subclinical inflammation, and blood pressure elevation,” Dr. Holt said.
The current study doesn’t determine whether NSAID-associated HF stems from transient fluid overload or direct cardiac effects, but it’s “most likely both.”
In other limitations, the analysis is unable to “reliably explore” whether promotion of HF is an NSAID class effect, a “clinically relevant” point given the drugs’ varying effects on cardiovascular risk, states an accompanying editorial. Nor was it able to determine whether the drugs exert a dose-response effect on HF risk, noted Hassan Khan, MD, PhD, Norton Healthcare, Louisville, Ky., and Setor K. Kunutsor, MD, PhD, University of Leicester (England).
Still, “given the well-established relationship between the use of NSAIDs and increased HF, these findings are not unexpected because type 2 diabetes is also a major risk factor for HF.”
But it may be “premature to issue guideline recommendations based on a single observational study,” the editorialists wrote. “Further robust clinical trial evidence is needed to replicate these results and investigate the relationship of the type and dose of NSAIDs with HF risk. However, it should be realized that short-term or long-term use of NSAIDs may be detrimental to cardiovascular health.”
The analysis covered 23,308 patients from throughout Denmark with a type 2 diabetes diagnosis and no HF history who experienced a first HF hospitalization; their age averaged 76 years and 39% were women.
They served as their own controls; their NSAID exposures at two 28-day periods preceding the HF event, the one immediately before and the other preceding it by 56 days, were compared as the index and control periods, respectively.
Exposure to NSAIDs was defined as obtaining a prescription for celecoxib, diclofenac, ibuprofen, or naproxen, “as these are NSAIDs used primarily in Denmark,” the report states.
The odds ratios for HF hospitalization associated with NSAID exposure within 28 days preceding the event were 1.43 (95% confidence interval, 1.27-1.63) overall, 1.41 (95% CI, 1.16-1.71) for an NSAID given on top of both RASi and diuretics, 1.68 (95% CI, 1.00-2.88) for patients with elevated hemoglobin A1c, 1.78 (95% CI, 1.39-2.28) for those 80 or older, and 2.71 (95% CI, 1.78-4.23) for those with prior NSAID use.
That NSAID use and diabetes are each associated with increased risk for HF is well established, Dr. Holt observed. Yet the drugs had been prescribed to 16% of patients in the study.
“One of the more surprising findings, to me, was the quite substantial use of prescribed NSAIDs in a population of patients with diabetes, a patient group with a well-established cardiovascular risk,” he said.
“This patient group is only growing, so emphasis on the possible associations between even short-term NSAID use and incident heart failure is probably timely and perhaps needed.”
Dr. Holt and the study were supported by grants from Ib Mogens Kristiansens Almene Fond, Helsefonden, Snedkermester Sophus Jacobsen og hustru Astrid Jacobsen Fond, Marie og M.B. Richters Fond, and the Dagmar Marshalls Fond. Dr. Khan and Dr. Kunutsor reported no relevant relationships.
A version of this article first appeared on Medscape.com.
suggests a prospective, controlled study.
Certain subgroups may account for much of the excess risk, the results suggest, including the very elderly, patients with uncontrolled diabetes, those prescribed an NSAID for the first time, and patients already taking both a renin-angiotensin system inhibitor (RASi) and a diuretic.
Such patients with a firm indication for NSAIDs potentially could “be the ones benefiting most from closer follow-up, reduced dosage, or other mitigation strategies,” Anders Holt, MD, said in an interview.
Dr. Holt, of Copenhagen University Hospital and Herlev-Gentofte Hospital in Hellerup, Denmark, is lead author on the analysis of Danish registry data published in the Journal of the American College of Cardiology. He presented essentially the same results in preliminary form at the 2022 annual congress of the European Society of Cardiology.
HF hospitalizations linked to NSAIDs, the published report notes, are often attributed to symptoms from temporary fluid overload, often without worsening cardiac function, that stem from the drugs’ renal effects.
“One could speculate,” Dr. Holt said, that such HF events might be less severe and even associated with better outcomes, compared with other forms of heart failure.
But the current analysis provides a hint to the contrary, he observed. The 5-year mortality was similar for patients with HF linked to NSAIDs and those with other forms of HF, “which could suggest that NSAID-associated heart failure is more than transient fluid overload.”
The drugs may promote HF through direct effects on the heart by any of several proposed mechanisms, including “induction of arrhythmias and heart fibrosis, vasoconstriction, subclinical inflammation, and blood pressure elevation,” Dr. Holt said.
The current study doesn’t determine whether NSAID-associated HF stems from transient fluid overload or direct cardiac effects, but it’s “most likely both.”
In other limitations, the analysis is unable to “reliably explore” whether promotion of HF is an NSAID class effect, a “clinically relevant” point given the drugs’ varying effects on cardiovascular risk, states an accompanying editorial. Nor was it able to determine whether the drugs exert a dose-response effect on HF risk, noted Hassan Khan, MD, PhD, Norton Healthcare, Louisville, Ky., and Setor K. Kunutsor, MD, PhD, University of Leicester (England).
Still, “given the well-established relationship between the use of NSAIDs and increased HF, these findings are not unexpected because type 2 diabetes is also a major risk factor for HF.”
But it may be “premature to issue guideline recommendations based on a single observational study,” the editorialists wrote. “Further robust clinical trial evidence is needed to replicate these results and investigate the relationship of the type and dose of NSAIDs with HF risk. However, it should be realized that short-term or long-term use of NSAIDs may be detrimental to cardiovascular health.”
The analysis covered 23,308 patients from throughout Denmark with a type 2 diabetes diagnosis and no HF history who experienced a first HF hospitalization; their age averaged 76 years and 39% were women.
They served as their own controls; their NSAID exposures at two 28-day periods preceding the HF event, the one immediately before and the other preceding it by 56 days, were compared as the index and control periods, respectively.
Exposure to NSAIDs was defined as obtaining a prescription for celecoxib, diclofenac, ibuprofen, or naproxen, “as these are NSAIDs used primarily in Denmark,” the report states.
The odds ratios for HF hospitalization associated with NSAID exposure within 28 days preceding the event were 1.43 (95% confidence interval, 1.27-1.63) overall, 1.41 (95% CI, 1.16-1.71) for an NSAID given on top of both RASi and diuretics, 1.68 (95% CI, 1.00-2.88) for patients with elevated hemoglobin A1c, 1.78 (95% CI, 1.39-2.28) for those 80 or older, and 2.71 (95% CI, 1.78-4.23) for those with prior NSAID use.
That NSAID use and diabetes are each associated with increased risk for HF is well established, Dr. Holt observed. Yet the drugs had been prescribed to 16% of patients in the study.
“One of the more surprising findings, to me, was the quite substantial use of prescribed NSAIDs in a population of patients with diabetes, a patient group with a well-established cardiovascular risk,” he said.
“This patient group is only growing, so emphasis on the possible associations between even short-term NSAID use and incident heart failure is probably timely and perhaps needed.”
Dr. Holt and the study were supported by grants from Ib Mogens Kristiansens Almene Fond, Helsefonden, Snedkermester Sophus Jacobsen og hustru Astrid Jacobsen Fond, Marie og M.B. Richters Fond, and the Dagmar Marshalls Fond. Dr. Khan and Dr. Kunutsor reported no relevant relationships.
A version of this article first appeared on Medscape.com.
suggests a prospective, controlled study.
Certain subgroups may account for much of the excess risk, the results suggest, including the very elderly, patients with uncontrolled diabetes, those prescribed an NSAID for the first time, and patients already taking both a renin-angiotensin system inhibitor (RASi) and a diuretic.
Such patients with a firm indication for NSAIDs potentially could “be the ones benefiting most from closer follow-up, reduced dosage, or other mitigation strategies,” Anders Holt, MD, said in an interview.
Dr. Holt, of Copenhagen University Hospital and Herlev-Gentofte Hospital in Hellerup, Denmark, is lead author on the analysis of Danish registry data published in the Journal of the American College of Cardiology. He presented essentially the same results in preliminary form at the 2022 annual congress of the European Society of Cardiology.
HF hospitalizations linked to NSAIDs, the published report notes, are often attributed to symptoms from temporary fluid overload, often without worsening cardiac function, that stem from the drugs’ renal effects.
“One could speculate,” Dr. Holt said, that such HF events might be less severe and even associated with better outcomes, compared with other forms of heart failure.
But the current analysis provides a hint to the contrary, he observed. The 5-year mortality was similar for patients with HF linked to NSAIDs and those with other forms of HF, “which could suggest that NSAID-associated heart failure is more than transient fluid overload.”
The drugs may promote HF through direct effects on the heart by any of several proposed mechanisms, including “induction of arrhythmias and heart fibrosis, vasoconstriction, subclinical inflammation, and blood pressure elevation,” Dr. Holt said.
The current study doesn’t determine whether NSAID-associated HF stems from transient fluid overload or direct cardiac effects, but it’s “most likely both.”
In other limitations, the analysis is unable to “reliably explore” whether promotion of HF is an NSAID class effect, a “clinically relevant” point given the drugs’ varying effects on cardiovascular risk, states an accompanying editorial. Nor was it able to determine whether the drugs exert a dose-response effect on HF risk, noted Hassan Khan, MD, PhD, Norton Healthcare, Louisville, Ky., and Setor K. Kunutsor, MD, PhD, University of Leicester (England).
Still, “given the well-established relationship between the use of NSAIDs and increased HF, these findings are not unexpected because type 2 diabetes is also a major risk factor for HF.”
But it may be “premature to issue guideline recommendations based on a single observational study,” the editorialists wrote. “Further robust clinical trial evidence is needed to replicate these results and investigate the relationship of the type and dose of NSAIDs with HF risk. However, it should be realized that short-term or long-term use of NSAIDs may be detrimental to cardiovascular health.”
The analysis covered 23,308 patients from throughout Denmark with a type 2 diabetes diagnosis and no HF history who experienced a first HF hospitalization; their age averaged 76 years and 39% were women.
They served as their own controls; their NSAID exposures at two 28-day periods preceding the HF event, the one immediately before and the other preceding it by 56 days, were compared as the index and control periods, respectively.
Exposure to NSAIDs was defined as obtaining a prescription for celecoxib, diclofenac, ibuprofen, or naproxen, “as these are NSAIDs used primarily in Denmark,” the report states.
The odds ratios for HF hospitalization associated with NSAID exposure within 28 days preceding the event were 1.43 (95% confidence interval, 1.27-1.63) overall, 1.41 (95% CI, 1.16-1.71) for an NSAID given on top of both RASi and diuretics, 1.68 (95% CI, 1.00-2.88) for patients with elevated hemoglobin A1c, 1.78 (95% CI, 1.39-2.28) for those 80 or older, and 2.71 (95% CI, 1.78-4.23) for those with prior NSAID use.
That NSAID use and diabetes are each associated with increased risk for HF is well established, Dr. Holt observed. Yet the drugs had been prescribed to 16% of patients in the study.
“One of the more surprising findings, to me, was the quite substantial use of prescribed NSAIDs in a population of patients with diabetes, a patient group with a well-established cardiovascular risk,” he said.
“This patient group is only growing, so emphasis on the possible associations between even short-term NSAID use and incident heart failure is probably timely and perhaps needed.”
Dr. Holt and the study were supported by grants from Ib Mogens Kristiansens Almene Fond, Helsefonden, Snedkermester Sophus Jacobsen og hustru Astrid Jacobsen Fond, Marie og M.B. Richters Fond, and the Dagmar Marshalls Fond. Dr. Khan and Dr. Kunutsor reported no relevant relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Parents of patients with rheumatic disease, MIS-C strongly hesitant of COVID vaccination
NEW ORLEANS – Parents’ concerns about vaccinating their children against COVID-19 remain a substantial barrier to immunizing children against the disease, whether those children have chronic rheumatologic conditions or a history of multisystem inflammatory syndrome in children (MIS-C), according to two studies presented at the Pediatric Rheumatology Symposium.
Parents of children who developed MIS-C after a SARS-CoV-2 infection were particularly hesitant to vaccinate, despite strong encouragement from health care professionals at Baylor College of Medicine, Houston, said the presenter of one of the studies.
“Unfortunately, it remains unclear who is susceptible and what the mechanisms are” when it comes to MIS-C, Mariana Sanchez Villa, MS, a research coordinator at Baylor, told attendees. “Because of this, there is much hesitancy to vaccinate children with a history of MIS-C against COVID-19 out of a fear that hyperinflammation may occur.”
Ms. Sanchez Villa reported findings on the vaccination rate among patients who had been hospitalized with MIS-C. The researchers included all 295 patients who presented at the hospital with MIS-C between May 2020 and October 2022. Overall, 5% of these patients had been vaccinated against COVID-19 before they were diagnosed with MIS-C. When all these patients and their families came to outpatient follow-up appointments after discharge, the subspecialist clinicians recommended the children receive the COVID-19 vaccine 3 months after discharge. The researchers then reviewed the patients’ charts to see who did and did not receive the vaccine, which they confirmed through the state’s immunization registry.
Among the 295 patients with MIS-C, 1 died, and 99 (34%) received at least one COVID-19 vaccine dose after their diagnosis, including 7 of the 15 who had also been vaccinated prior to their MIS-C diagnosis. Just over half of the vaccinated patients (58%) were male. They received their vaccine an average 8.8 months after their hospitalization, when they were an average 10 years old, and all but one of the vaccine doses they received were the Pfizer/BioNTech mRNA vaccine.
Only 9 of the 99 vaccinated patients are fully vaccinated, defined as receiving the primary series plus the recommended boosters. Of the other patients, 13 received only one dose of the vaccine, 60 received two doses, and 17 received at least three doses of the primary series doses but no bivalent boosters. Over a subsequent average 11 months of follow-up, none of the vaccinated patients returned to the hospital with a recurrence of MIS-C or any other hyperinflammatory condition. The seven patients who had been vaccinated both before and after their MIS-C diagnosis have also not had any recurrence of a hyperinflammatory condition.
“SARS-CoV-2 vaccination is well-tolerated by children with a history of MIS-C,” the researchers concluded. Ms. Sanchez Villa referenced two other studies, in The Pediatric Infectious Disease Journal and in JAMA Network Open, with similar findings on the safety of COVID-19 vaccination in patients who have had MIS-C. “This is reassuring as SARS-CoV-2 becomes endemic and annual vaccination against SARS-CoV-2 is considered.”
Dilan Dissanayake, MD, PhD, a rheumatologist at The Hospital for Sick Children in Toronto, who attended the presentation, told this news organization that data increasingly show a “synergistic protective effect” from COVID-19 infection and vaccination. That is, “having COVID or having MIS-C once doesn’t necessarily preclude you from having it again,” thereby supporting the importance of vaccination after an MIS-C diagnosis. In talking to parents about vaccinating, he has found it most helpful for them to hear about rheumatologists’ experience regarding COVID-19 vaccination.
“Particularly as the pandemic went on, being able to comfortably say that we have this large patient group, as well as collaborators across the world who have been monitoring for any safety issues, and that all the data has been reassuring” has been most useful for parents to hear, Dr. Dissanayake said.
The other study, led by Beth Rutstein, MD, MSCE, an attending rheumatologist at Children’s Hospital of Philadelphia, focused on the population of pediatric rheumatology patients by surveying pediatric rheumatologists who were members of the Childhood Arthritis and Rheumatology Research Alliance. The survey, conducted from March to May 2022, included questions about the rheumatologists’ COVID-19 vaccination practices as well as perceptions of the vaccine by the parents of their patients.
The 219 respondents included 74% pediatric rheumatologists and 21% fellows. Nearly all the respondents (98%) believed that any disease flares after COVID-19 vaccination would be mild and/or rare, and nearly all (98%) recommend their patients be vaccinated against COVID-19.
The primary finding from the study was that “we [rheumatologists] have different concerns from the families,” coauthor and presenter Vidya Sivaraman, MD, a pediatric rheumatologist at Nationwide Children’s Hospital and the Ohio State University in Columbus, told this news organization. “We’re more worried about the efficacy of the vaccine on immunosuppressive medications,” such as rituximab, which depletes B cells, Dr. Sivaraman said, but concerns about the vaccine’s immunogenicity or efficacy were very low among parents.
Just over half the clinicians surveyed (59%) were concerned about how effective the vaccine would be for their patients, especially those receiving immunosuppressive therapy. Health care professionals were most concerned about patients on rituximab – all clinicians reported concerns about the vaccine’s effectiveness in these patients – followed by patients taking systemic corticosteroids (86%), mycophenolate mofetil (59%), and Janus kinase inhibitors (46%).
Most clinicians (88%) reported that they had temporarily modified a patient’s immunosuppressive therapy to allow for vaccination, following guidelines by the American College of Rheumatology. Aside from a small proportion of health care professionals who checked patients’ post-vaccination serology primarily for research purposes, most clinicians (82%) did not collect this serology.
In regard to adverse events, the concern cited most often by respondents was myocarditis (76%), followed by development of new autoimmune conditions (29%) and thrombosis (22%), but the clinicians ranked these adverse events as low risk.
Meanwhile, the top three concerns about vaccination among parents, as reported to physicians, were worries about side effects, lack of long-term safety data on the vaccine, and misinformation they had heard, such as anxiety about changes to their child’s genetics or vaccination causing a COVID-19 infection. “They’re seeing things on social media from other parents [saying that COVID-19 vaccines are] going to affect their fertility, so they don’t want their daughters to get it,” Dr. Sivaraman said as another example of commonly cited misinformation.
Nearly half of the respondents (47%) said more than half of their families had concerns about side effects and the lack of data on long-term outcomes after vaccination. Only 8.5% of physicians said that fewer than 10% of their families were anxious about side effects. In addition, 39% of physicians said more than half of their families had concerns about misinformation they had heard, and only 16% of physicians had heard about misinformation concerns from fewer than 10% of their patients.
Other concerns cited by parents included their child’s disease flaring; lack of data on how well the vaccine would stimulate their child’s immune system; their child having already had COVID-19; and not believing COVID-19 was a major health risk to their child. Nearly every respondent (98%) said they had parents who turned down COVID-19 vaccination, and a majority (75%) reported that more than 10% of their patients had parents who were hesitant about COVID-19 vaccination.
No external funding was noted for either study. Ms. Sanchez Villa had no relevant financial relationships, but two abstract coauthors reported financial relationships with Pfizer and Moderna, and one reported a financial relationship with Novartis. Dr. Rutstein, Dr. Sivaraman, and Dr. Dissanayake had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NEW ORLEANS – Parents’ concerns about vaccinating their children against COVID-19 remain a substantial barrier to immunizing children against the disease, whether those children have chronic rheumatologic conditions or a history of multisystem inflammatory syndrome in children (MIS-C), according to two studies presented at the Pediatric Rheumatology Symposium.
Parents of children who developed MIS-C after a SARS-CoV-2 infection were particularly hesitant to vaccinate, despite strong encouragement from health care professionals at Baylor College of Medicine, Houston, said the presenter of one of the studies.
“Unfortunately, it remains unclear who is susceptible and what the mechanisms are” when it comes to MIS-C, Mariana Sanchez Villa, MS, a research coordinator at Baylor, told attendees. “Because of this, there is much hesitancy to vaccinate children with a history of MIS-C against COVID-19 out of a fear that hyperinflammation may occur.”
Ms. Sanchez Villa reported findings on the vaccination rate among patients who had been hospitalized with MIS-C. The researchers included all 295 patients who presented at the hospital with MIS-C between May 2020 and October 2022. Overall, 5% of these patients had been vaccinated against COVID-19 before they were diagnosed with MIS-C. When all these patients and their families came to outpatient follow-up appointments after discharge, the subspecialist clinicians recommended the children receive the COVID-19 vaccine 3 months after discharge. The researchers then reviewed the patients’ charts to see who did and did not receive the vaccine, which they confirmed through the state’s immunization registry.
Among the 295 patients with MIS-C, 1 died, and 99 (34%) received at least one COVID-19 vaccine dose after their diagnosis, including 7 of the 15 who had also been vaccinated prior to their MIS-C diagnosis. Just over half of the vaccinated patients (58%) were male. They received their vaccine an average 8.8 months after their hospitalization, when they were an average 10 years old, and all but one of the vaccine doses they received were the Pfizer/BioNTech mRNA vaccine.
Only 9 of the 99 vaccinated patients are fully vaccinated, defined as receiving the primary series plus the recommended boosters. Of the other patients, 13 received only one dose of the vaccine, 60 received two doses, and 17 received at least three doses of the primary series doses but no bivalent boosters. Over a subsequent average 11 months of follow-up, none of the vaccinated patients returned to the hospital with a recurrence of MIS-C or any other hyperinflammatory condition. The seven patients who had been vaccinated both before and after their MIS-C diagnosis have also not had any recurrence of a hyperinflammatory condition.
“SARS-CoV-2 vaccination is well-tolerated by children with a history of MIS-C,” the researchers concluded. Ms. Sanchez Villa referenced two other studies, in The Pediatric Infectious Disease Journal and in JAMA Network Open, with similar findings on the safety of COVID-19 vaccination in patients who have had MIS-C. “This is reassuring as SARS-CoV-2 becomes endemic and annual vaccination against SARS-CoV-2 is considered.”
Dilan Dissanayake, MD, PhD, a rheumatologist at The Hospital for Sick Children in Toronto, who attended the presentation, told this news organization that data increasingly show a “synergistic protective effect” from COVID-19 infection and vaccination. That is, “having COVID or having MIS-C once doesn’t necessarily preclude you from having it again,” thereby supporting the importance of vaccination after an MIS-C diagnosis. In talking to parents about vaccinating, he has found it most helpful for them to hear about rheumatologists’ experience regarding COVID-19 vaccination.
“Particularly as the pandemic went on, being able to comfortably say that we have this large patient group, as well as collaborators across the world who have been monitoring for any safety issues, and that all the data has been reassuring” has been most useful for parents to hear, Dr. Dissanayake said.
The other study, led by Beth Rutstein, MD, MSCE, an attending rheumatologist at Children’s Hospital of Philadelphia, focused on the population of pediatric rheumatology patients by surveying pediatric rheumatologists who were members of the Childhood Arthritis and Rheumatology Research Alliance. The survey, conducted from March to May 2022, included questions about the rheumatologists’ COVID-19 vaccination practices as well as perceptions of the vaccine by the parents of their patients.
The 219 respondents included 74% pediatric rheumatologists and 21% fellows. Nearly all the respondents (98%) believed that any disease flares after COVID-19 vaccination would be mild and/or rare, and nearly all (98%) recommend their patients be vaccinated against COVID-19.
The primary finding from the study was that “we [rheumatologists] have different concerns from the families,” coauthor and presenter Vidya Sivaraman, MD, a pediatric rheumatologist at Nationwide Children’s Hospital and the Ohio State University in Columbus, told this news organization. “We’re more worried about the efficacy of the vaccine on immunosuppressive medications,” such as rituximab, which depletes B cells, Dr. Sivaraman said, but concerns about the vaccine’s immunogenicity or efficacy were very low among parents.
Just over half the clinicians surveyed (59%) were concerned about how effective the vaccine would be for their patients, especially those receiving immunosuppressive therapy. Health care professionals were most concerned about patients on rituximab – all clinicians reported concerns about the vaccine’s effectiveness in these patients – followed by patients taking systemic corticosteroids (86%), mycophenolate mofetil (59%), and Janus kinase inhibitors (46%).
Most clinicians (88%) reported that they had temporarily modified a patient’s immunosuppressive therapy to allow for vaccination, following guidelines by the American College of Rheumatology. Aside from a small proportion of health care professionals who checked patients’ post-vaccination serology primarily for research purposes, most clinicians (82%) did not collect this serology.
In regard to adverse events, the concern cited most often by respondents was myocarditis (76%), followed by development of new autoimmune conditions (29%) and thrombosis (22%), but the clinicians ranked these adverse events as low risk.
Meanwhile, the top three concerns about vaccination among parents, as reported to physicians, were worries about side effects, lack of long-term safety data on the vaccine, and misinformation they had heard, such as anxiety about changes to their child’s genetics or vaccination causing a COVID-19 infection. “They’re seeing things on social media from other parents [saying that COVID-19 vaccines are] going to affect their fertility, so they don’t want their daughters to get it,” Dr. Sivaraman said as another example of commonly cited misinformation.
Nearly half of the respondents (47%) said more than half of their families had concerns about side effects and the lack of data on long-term outcomes after vaccination. Only 8.5% of physicians said that fewer than 10% of their families were anxious about side effects. In addition, 39% of physicians said more than half of their families had concerns about misinformation they had heard, and only 16% of physicians had heard about misinformation concerns from fewer than 10% of their patients.
Other concerns cited by parents included their child’s disease flaring; lack of data on how well the vaccine would stimulate their child’s immune system; their child having already had COVID-19; and not believing COVID-19 was a major health risk to their child. Nearly every respondent (98%) said they had parents who turned down COVID-19 vaccination, and a majority (75%) reported that more than 10% of their patients had parents who were hesitant about COVID-19 vaccination.
No external funding was noted for either study. Ms. Sanchez Villa had no relevant financial relationships, but two abstract coauthors reported financial relationships with Pfizer and Moderna, and one reported a financial relationship with Novartis. Dr. Rutstein, Dr. Sivaraman, and Dr. Dissanayake had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NEW ORLEANS – Parents’ concerns about vaccinating their children against COVID-19 remain a substantial barrier to immunizing children against the disease, whether those children have chronic rheumatologic conditions or a history of multisystem inflammatory syndrome in children (MIS-C), according to two studies presented at the Pediatric Rheumatology Symposium.
Parents of children who developed MIS-C after a SARS-CoV-2 infection were particularly hesitant to vaccinate, despite strong encouragement from health care professionals at Baylor College of Medicine, Houston, said the presenter of one of the studies.
“Unfortunately, it remains unclear who is susceptible and what the mechanisms are” when it comes to MIS-C, Mariana Sanchez Villa, MS, a research coordinator at Baylor, told attendees. “Because of this, there is much hesitancy to vaccinate children with a history of MIS-C against COVID-19 out of a fear that hyperinflammation may occur.”
Ms. Sanchez Villa reported findings on the vaccination rate among patients who had been hospitalized with MIS-C. The researchers included all 295 patients who presented at the hospital with MIS-C between May 2020 and October 2022. Overall, 5% of these patients had been vaccinated against COVID-19 before they were diagnosed with MIS-C. When all these patients and their families came to outpatient follow-up appointments after discharge, the subspecialist clinicians recommended the children receive the COVID-19 vaccine 3 months after discharge. The researchers then reviewed the patients’ charts to see who did and did not receive the vaccine, which they confirmed through the state’s immunization registry.
Among the 295 patients with MIS-C, 1 died, and 99 (34%) received at least one COVID-19 vaccine dose after their diagnosis, including 7 of the 15 who had also been vaccinated prior to their MIS-C diagnosis. Just over half of the vaccinated patients (58%) were male. They received their vaccine an average 8.8 months after their hospitalization, when they were an average 10 years old, and all but one of the vaccine doses they received were the Pfizer/BioNTech mRNA vaccine.
Only 9 of the 99 vaccinated patients are fully vaccinated, defined as receiving the primary series plus the recommended boosters. Of the other patients, 13 received only one dose of the vaccine, 60 received two doses, and 17 received at least three doses of the primary series doses but no bivalent boosters. Over a subsequent average 11 months of follow-up, none of the vaccinated patients returned to the hospital with a recurrence of MIS-C or any other hyperinflammatory condition. The seven patients who had been vaccinated both before and after their MIS-C diagnosis have also not had any recurrence of a hyperinflammatory condition.
“SARS-CoV-2 vaccination is well-tolerated by children with a history of MIS-C,” the researchers concluded. Ms. Sanchez Villa referenced two other studies, in The Pediatric Infectious Disease Journal and in JAMA Network Open, with similar findings on the safety of COVID-19 vaccination in patients who have had MIS-C. “This is reassuring as SARS-CoV-2 becomes endemic and annual vaccination against SARS-CoV-2 is considered.”
Dilan Dissanayake, MD, PhD, a rheumatologist at The Hospital for Sick Children in Toronto, who attended the presentation, told this news organization that data increasingly show a “synergistic protective effect” from COVID-19 infection and vaccination. That is, “having COVID or having MIS-C once doesn’t necessarily preclude you from having it again,” thereby supporting the importance of vaccination after an MIS-C diagnosis. In talking to parents about vaccinating, he has found it most helpful for them to hear about rheumatologists’ experience regarding COVID-19 vaccination.
“Particularly as the pandemic went on, being able to comfortably say that we have this large patient group, as well as collaborators across the world who have been monitoring for any safety issues, and that all the data has been reassuring” has been most useful for parents to hear, Dr. Dissanayake said.
The other study, led by Beth Rutstein, MD, MSCE, an attending rheumatologist at Children’s Hospital of Philadelphia, focused on the population of pediatric rheumatology patients by surveying pediatric rheumatologists who were members of the Childhood Arthritis and Rheumatology Research Alliance. The survey, conducted from March to May 2022, included questions about the rheumatologists’ COVID-19 vaccination practices as well as perceptions of the vaccine by the parents of their patients.
The 219 respondents included 74% pediatric rheumatologists and 21% fellows. Nearly all the respondents (98%) believed that any disease flares after COVID-19 vaccination would be mild and/or rare, and nearly all (98%) recommend their patients be vaccinated against COVID-19.
The primary finding from the study was that “we [rheumatologists] have different concerns from the families,” coauthor and presenter Vidya Sivaraman, MD, a pediatric rheumatologist at Nationwide Children’s Hospital and the Ohio State University in Columbus, told this news organization. “We’re more worried about the efficacy of the vaccine on immunosuppressive medications,” such as rituximab, which depletes B cells, Dr. Sivaraman said, but concerns about the vaccine’s immunogenicity or efficacy were very low among parents.
Just over half the clinicians surveyed (59%) were concerned about how effective the vaccine would be for their patients, especially those receiving immunosuppressive therapy. Health care professionals were most concerned about patients on rituximab – all clinicians reported concerns about the vaccine’s effectiveness in these patients – followed by patients taking systemic corticosteroids (86%), mycophenolate mofetil (59%), and Janus kinase inhibitors (46%).
Most clinicians (88%) reported that they had temporarily modified a patient’s immunosuppressive therapy to allow for vaccination, following guidelines by the American College of Rheumatology. Aside from a small proportion of health care professionals who checked patients’ post-vaccination serology primarily for research purposes, most clinicians (82%) did not collect this serology.
In regard to adverse events, the concern cited most often by respondents was myocarditis (76%), followed by development of new autoimmune conditions (29%) and thrombosis (22%), but the clinicians ranked these adverse events as low risk.
Meanwhile, the top three concerns about vaccination among parents, as reported to physicians, were worries about side effects, lack of long-term safety data on the vaccine, and misinformation they had heard, such as anxiety about changes to their child’s genetics or vaccination causing a COVID-19 infection. “They’re seeing things on social media from other parents [saying that COVID-19 vaccines are] going to affect their fertility, so they don’t want their daughters to get it,” Dr. Sivaraman said as another example of commonly cited misinformation.
Nearly half of the respondents (47%) said more than half of their families had concerns about side effects and the lack of data on long-term outcomes after vaccination. Only 8.5% of physicians said that fewer than 10% of their families were anxious about side effects. In addition, 39% of physicians said more than half of their families had concerns about misinformation they had heard, and only 16% of physicians had heard about misinformation concerns from fewer than 10% of their patients.
Other concerns cited by parents included their child’s disease flaring; lack of data on how well the vaccine would stimulate their child’s immune system; their child having already had COVID-19; and not believing COVID-19 was a major health risk to their child. Nearly every respondent (98%) said they had parents who turned down COVID-19 vaccination, and a majority (75%) reported that more than 10% of their patients had parents who were hesitant about COVID-19 vaccination.
No external funding was noted for either study. Ms. Sanchez Villa had no relevant financial relationships, but two abstract coauthors reported financial relationships with Pfizer and Moderna, and one reported a financial relationship with Novartis. Dr. Rutstein, Dr. Sivaraman, and Dr. Dissanayake had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT PRSYM 2023
Long COVID hitting some states, minorities, women harder
More than one in four adults sickened by the virus go on to have long COVID, according to a new report from the U.S. Census Bureau. Overall, nearly 15% of all American adults – more than 38 million people nationwide – have had long COVID at some point since the start of the pandemic, according to the report.
The report, based on survey data collected between March 1 and 13, defines long COVID as symptoms lasting at least 3 months that people didn’t have before getting infected with the virus.
It is the second recent look at who is most likely to face long COVID. A similar study, published in March, found that women, smokers, and those who had severe COVID-19 infections are most likely to have the disorder
The Census Bureau report found that while 27% of adults nationwide have had long COVID after getting infected with the virus, the condition has impacted some states more than others. The proportion of residents hit with long COVID ranged from a low of 18.8% in New Jersey to a high of 40.7% in West Virginia.
Other states with long COVID rates well below the national average include Alaska, Maryland, New York, and Wisconsin. At the other end of the spectrum, the states with rates well above the national average include Kentucky, Mississippi, New Mexico, Nevada, South Carolina, South Dakota, and Wyoming.
Long COVID rates also varied by age, gender, and race. People in their 50s were most at risk, with about 31% of those infected by the virus going on to have long COVID, followed by those in their 40s, at more than 29%.
Far more women (almost 33%) than men (21%) with COVID infections got long COVID. And when researchers looked at long COVID rates based on gender identity, they found that transgender adults were more than twice as likely to have long COVID than cisgender males. Bisexual adults also had much higher long COVID rates than straight, gay, or lesbian people.
Long COVID was also much more common among Hispanic adults, affecting almost 29% of those infected with the virus, than among White or Black people, who had long COVID rates similar to the national average of 27%. Asian adults had lower long COVID rates than the national average, at less than 20%.
People with disabilities were also at higher risk, with long COVID rates of almost 47%, compared with 24% among adults without disabilities.
A version of this article first appeared on WebMD.com.
More than one in four adults sickened by the virus go on to have long COVID, according to a new report from the U.S. Census Bureau. Overall, nearly 15% of all American adults – more than 38 million people nationwide – have had long COVID at some point since the start of the pandemic, according to the report.
The report, based on survey data collected between March 1 and 13, defines long COVID as symptoms lasting at least 3 months that people didn’t have before getting infected with the virus.
It is the second recent look at who is most likely to face long COVID. A similar study, published in March, found that women, smokers, and those who had severe COVID-19 infections are most likely to have the disorder
The Census Bureau report found that while 27% of adults nationwide have had long COVID after getting infected with the virus, the condition has impacted some states more than others. The proportion of residents hit with long COVID ranged from a low of 18.8% in New Jersey to a high of 40.7% in West Virginia.
Other states with long COVID rates well below the national average include Alaska, Maryland, New York, and Wisconsin. At the other end of the spectrum, the states with rates well above the national average include Kentucky, Mississippi, New Mexico, Nevada, South Carolina, South Dakota, and Wyoming.
Long COVID rates also varied by age, gender, and race. People in their 50s were most at risk, with about 31% of those infected by the virus going on to have long COVID, followed by those in their 40s, at more than 29%.
Far more women (almost 33%) than men (21%) with COVID infections got long COVID. And when researchers looked at long COVID rates based on gender identity, they found that transgender adults were more than twice as likely to have long COVID than cisgender males. Bisexual adults also had much higher long COVID rates than straight, gay, or lesbian people.
Long COVID was also much more common among Hispanic adults, affecting almost 29% of those infected with the virus, than among White or Black people, who had long COVID rates similar to the national average of 27%. Asian adults had lower long COVID rates than the national average, at less than 20%.
People with disabilities were also at higher risk, with long COVID rates of almost 47%, compared with 24% among adults without disabilities.
A version of this article first appeared on WebMD.com.
More than one in four adults sickened by the virus go on to have long COVID, according to a new report from the U.S. Census Bureau. Overall, nearly 15% of all American adults – more than 38 million people nationwide – have had long COVID at some point since the start of the pandemic, according to the report.
The report, based on survey data collected between March 1 and 13, defines long COVID as symptoms lasting at least 3 months that people didn’t have before getting infected with the virus.
It is the second recent look at who is most likely to face long COVID. A similar study, published in March, found that women, smokers, and those who had severe COVID-19 infections are most likely to have the disorder
The Census Bureau report found that while 27% of adults nationwide have had long COVID after getting infected with the virus, the condition has impacted some states more than others. The proportion of residents hit with long COVID ranged from a low of 18.8% in New Jersey to a high of 40.7% in West Virginia.
Other states with long COVID rates well below the national average include Alaska, Maryland, New York, and Wisconsin. At the other end of the spectrum, the states with rates well above the national average include Kentucky, Mississippi, New Mexico, Nevada, South Carolina, South Dakota, and Wyoming.
Long COVID rates also varied by age, gender, and race. People in their 50s were most at risk, with about 31% of those infected by the virus going on to have long COVID, followed by those in their 40s, at more than 29%.
Far more women (almost 33%) than men (21%) with COVID infections got long COVID. And when researchers looked at long COVID rates based on gender identity, they found that transgender adults were more than twice as likely to have long COVID than cisgender males. Bisexual adults also had much higher long COVID rates than straight, gay, or lesbian people.
Long COVID was also much more common among Hispanic adults, affecting almost 29% of those infected with the virus, than among White or Black people, who had long COVID rates similar to the national average of 27%. Asian adults had lower long COVID rates than the national average, at less than 20%.
People with disabilities were also at higher risk, with long COVID rates of almost 47%, compared with 24% among adults without disabilities.
A version of this article first appeared on WebMD.com.