User login
How Experts Predicts This COVID and Flu Season Will Unfold
What’s the outlook for COVID-19 and flu this fall and winter? It’ll probably be a lot like last year, experts say.
“We currently expect this flu season to be comparable to last year’s season,” said Adrienne Keen, PhD, of the Centers for Disease Control and Prevention’s (CDC) Center for Forecasting and Outbreak Analytics. “We expect this year’s COVID-19 season peak to be similar to last year’s or lower.” The CDC is still analyzing COVID surveillance data from the summer and will update the forecast as more is learned.
For COVID, that means it won’t be as bad as the pandemic years, and for the flu, it’s a typical pre-pandemic season. But status quo does not mean great.
Between October 2023 and April 2024, as many as 75 million people got the flu in the United States, according to CDC estimates, resulting in up to 900,000 hospitalizations and between 17,000 and 100,000 deaths. In 2023, about 900,000 Americans were hospitalized with COVID and 75,000 died.
Getting vaccinated remains crucial, public health officials stressed.
Predicting COVID
Two key predictors of how bad an upcoming COVID season will be are the cycling of new variants and the population’s immunity (protection from an infectious disease that happens when a population is immune through vaccination or previous infection).
When new variants go up and immunity goes down, “we tend to see the increase in cases,” said Michael T. Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy and a professor of public health at the University of Minnesota, Minneapolis. But if the number of variants goes down and immunity levels go up, the outlook is more favorable.
The new COVID variant called XEC has been found in at least 25 states. On September 27, the CDC added the variant to the COVID tracker. It now accounts for 6% of US cases. This was expected, as the variant has been circulating in Europe, said Amesh Adalja, MD, a senior scholar and infectious disease expert at the Center for Health Security at Johns Hopkins University, Baltimore, Maryland.
“There will always be a new variant appearing, and one falling,” he said. “So the fact that this is happening is not surprising.”
Meanwhile, the summer COVID surge has provided postinfection immunity for some people. “What’s likely is, we are going to see substantial protection of the population for several months based on previous infection and in some cases vaccination,” Dr. Osterholm said. That means protection from serious illness, hospitalizations, and deaths (but not necessarily infection). That protection could last through the year or into early 2025.
The timing of 2024’s winter surge will likely be a bit later than 2023’s, said Andrew Pekosz, PhD, a professor and vice chair of molecular microbiology and immunology at Johns Hopkins University, Baltimore, “peaking just after the Christmas/New Year holiday.”
During the 2023-2024 season, weekly COVID hospitalizations peaked the week of Dec. 30, said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill and a member of the COVID-19 Scenario Modeling Hub.
But variants are unpredictable. “There’s a chance that the XEC variant may take off and spread, or might not,” said Dr. Adalja. As of September 28, the Omicron variant KP.3.1.1 was leading, accounting for 58.7% of US cases, according to the CDC.
While Dr. Adalja agreed that 2024’s COVID season will probably be like 2023’s, “we have to be prepared for cases and hospitalizations going up,” he said, “but not to the point of a crisis.” A return to lockdowns and social distancing is unlikely.
Still, older adults and others at higher risk of getting very sick from COVID should consider masking during travel, said Rajendram Rajnarayanan, PhD, MSc, an associate professor at the New York Institute of Technology College of Osteopathic Medicine at Arkansas State University, Jonesboro.
Flu Forecasts
Predicting flu season this early is hard, said Jeffrey Shaman, PhD, a professor of environmental health sciences and professor of climate at Colombia University, New York.
“You can look at the CDC forecast and use it as a very loose guide right now,” said Dr. Shaman, who won the CDC’s first “Predict the Influenza Season Challenge” in 2014. “Until there is actually flu, it’s like trying to predict the landfall of a hurricane.” Flu activity remained low as of September 14 (the most current data available), according to the CDC.
When flu activity picks up, typically in mid-October or November, experts look at the dominant strain, exposure to similar strains in previous years, and how well-matched the current flu vaccine is to that dominant strain, Dr. Shaman said. Vaccine makers must make an educated guess months in advance regarding which strain to target, to allow time for production.
The vaccination rate plays a role, too, but that tends to remain constant, Dr. Shaman said. According to the CDC, less than half of adults age 18 and up got a flu vaccination last year.
Experts also consider flu patterns in the Southern Hemisphere, where 2024 flu activity has mostly involved two subtypes of influenza A — H1N1 and H3N2 — and some influenza B, the CDC found.
How Well Do This Year’s Vaccines and Viruses Match Up?
The FDA has authorized three updated COVID vaccines for this fall. Novavax targets the JN.1 strain of SARS-CoV-2, the virus that causes COVID-19. Both mRNA vaccines, Moderna and Pfizer, target KP.2, a descendant of JN.1. All three target current predominant variants, and any one of them is recommended by the CDC.
The vaccines are a good “though not perfect match to virtually all the circulating variants of SARS-CoV-2,” said Dr. Pekosz.
Experts said that the shots will protect against the XEC variant.
“XEC and its sublineages are expected to be the dominant fall/winter variant group,” said Dr. Rajnarayanan.
This year’s flu vaccines, all trivalent (protecting against three viruses), will target the three strains expected to circulate — H1N1, H3N2, and influenza B (Victoria), according to the CDC.
People should still get vaccinated, Dr. Adalja said, and use home tests for flu and COVID and take antivirals promptly when needed. The goal should not be status quo but rather fewer COVID and flu hospitalizations and deaths.
A version of this article first appeared on WebMD.com.
What’s the outlook for COVID-19 and flu this fall and winter? It’ll probably be a lot like last year, experts say.
“We currently expect this flu season to be comparable to last year’s season,” said Adrienne Keen, PhD, of the Centers for Disease Control and Prevention’s (CDC) Center for Forecasting and Outbreak Analytics. “We expect this year’s COVID-19 season peak to be similar to last year’s or lower.” The CDC is still analyzing COVID surveillance data from the summer and will update the forecast as more is learned.
For COVID, that means it won’t be as bad as the pandemic years, and for the flu, it’s a typical pre-pandemic season. But status quo does not mean great.
Between October 2023 and April 2024, as many as 75 million people got the flu in the United States, according to CDC estimates, resulting in up to 900,000 hospitalizations and between 17,000 and 100,000 deaths. In 2023, about 900,000 Americans were hospitalized with COVID and 75,000 died.
Getting vaccinated remains crucial, public health officials stressed.
Predicting COVID
Two key predictors of how bad an upcoming COVID season will be are the cycling of new variants and the population’s immunity (protection from an infectious disease that happens when a population is immune through vaccination or previous infection).
When new variants go up and immunity goes down, “we tend to see the increase in cases,” said Michael T. Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy and a professor of public health at the University of Minnesota, Minneapolis. But if the number of variants goes down and immunity levels go up, the outlook is more favorable.
The new COVID variant called XEC has been found in at least 25 states. On September 27, the CDC added the variant to the COVID tracker. It now accounts for 6% of US cases. This was expected, as the variant has been circulating in Europe, said Amesh Adalja, MD, a senior scholar and infectious disease expert at the Center for Health Security at Johns Hopkins University, Baltimore, Maryland.
“There will always be a new variant appearing, and one falling,” he said. “So the fact that this is happening is not surprising.”
Meanwhile, the summer COVID surge has provided postinfection immunity for some people. “What’s likely is, we are going to see substantial protection of the population for several months based on previous infection and in some cases vaccination,” Dr. Osterholm said. That means protection from serious illness, hospitalizations, and deaths (but not necessarily infection). That protection could last through the year or into early 2025.
The timing of 2024’s winter surge will likely be a bit later than 2023’s, said Andrew Pekosz, PhD, a professor and vice chair of molecular microbiology and immunology at Johns Hopkins University, Baltimore, “peaking just after the Christmas/New Year holiday.”
During the 2023-2024 season, weekly COVID hospitalizations peaked the week of Dec. 30, said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill and a member of the COVID-19 Scenario Modeling Hub.
But variants are unpredictable. “There’s a chance that the XEC variant may take off and spread, or might not,” said Dr. Adalja. As of September 28, the Omicron variant KP.3.1.1 was leading, accounting for 58.7% of US cases, according to the CDC.
While Dr. Adalja agreed that 2024’s COVID season will probably be like 2023’s, “we have to be prepared for cases and hospitalizations going up,” he said, “but not to the point of a crisis.” A return to lockdowns and social distancing is unlikely.
Still, older adults and others at higher risk of getting very sick from COVID should consider masking during travel, said Rajendram Rajnarayanan, PhD, MSc, an associate professor at the New York Institute of Technology College of Osteopathic Medicine at Arkansas State University, Jonesboro.
Flu Forecasts
Predicting flu season this early is hard, said Jeffrey Shaman, PhD, a professor of environmental health sciences and professor of climate at Colombia University, New York.
“You can look at the CDC forecast and use it as a very loose guide right now,” said Dr. Shaman, who won the CDC’s first “Predict the Influenza Season Challenge” in 2014. “Until there is actually flu, it’s like trying to predict the landfall of a hurricane.” Flu activity remained low as of September 14 (the most current data available), according to the CDC.
When flu activity picks up, typically in mid-October or November, experts look at the dominant strain, exposure to similar strains in previous years, and how well-matched the current flu vaccine is to that dominant strain, Dr. Shaman said. Vaccine makers must make an educated guess months in advance regarding which strain to target, to allow time for production.
The vaccination rate plays a role, too, but that tends to remain constant, Dr. Shaman said. According to the CDC, less than half of adults age 18 and up got a flu vaccination last year.
Experts also consider flu patterns in the Southern Hemisphere, where 2024 flu activity has mostly involved two subtypes of influenza A — H1N1 and H3N2 — and some influenza B, the CDC found.
How Well Do This Year’s Vaccines and Viruses Match Up?
The FDA has authorized three updated COVID vaccines for this fall. Novavax targets the JN.1 strain of SARS-CoV-2, the virus that causes COVID-19. Both mRNA vaccines, Moderna and Pfizer, target KP.2, a descendant of JN.1. All three target current predominant variants, and any one of them is recommended by the CDC.
The vaccines are a good “though not perfect match to virtually all the circulating variants of SARS-CoV-2,” said Dr. Pekosz.
Experts said that the shots will protect against the XEC variant.
“XEC and its sublineages are expected to be the dominant fall/winter variant group,” said Dr. Rajnarayanan.
This year’s flu vaccines, all trivalent (protecting against three viruses), will target the three strains expected to circulate — H1N1, H3N2, and influenza B (Victoria), according to the CDC.
People should still get vaccinated, Dr. Adalja said, and use home tests for flu and COVID and take antivirals promptly when needed. The goal should not be status quo but rather fewer COVID and flu hospitalizations and deaths.
A version of this article first appeared on WebMD.com.
What’s the outlook for COVID-19 and flu this fall and winter? It’ll probably be a lot like last year, experts say.
“We currently expect this flu season to be comparable to last year’s season,” said Adrienne Keen, PhD, of the Centers for Disease Control and Prevention’s (CDC) Center for Forecasting and Outbreak Analytics. “We expect this year’s COVID-19 season peak to be similar to last year’s or lower.” The CDC is still analyzing COVID surveillance data from the summer and will update the forecast as more is learned.
For COVID, that means it won’t be as bad as the pandemic years, and for the flu, it’s a typical pre-pandemic season. But status quo does not mean great.
Between October 2023 and April 2024, as many as 75 million people got the flu in the United States, according to CDC estimates, resulting in up to 900,000 hospitalizations and between 17,000 and 100,000 deaths. In 2023, about 900,000 Americans were hospitalized with COVID and 75,000 died.
Getting vaccinated remains crucial, public health officials stressed.
Predicting COVID
Two key predictors of how bad an upcoming COVID season will be are the cycling of new variants and the population’s immunity (protection from an infectious disease that happens when a population is immune through vaccination or previous infection).
When new variants go up and immunity goes down, “we tend to see the increase in cases,” said Michael T. Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy and a professor of public health at the University of Minnesota, Minneapolis. But if the number of variants goes down and immunity levels go up, the outlook is more favorable.
The new COVID variant called XEC has been found in at least 25 states. On September 27, the CDC added the variant to the COVID tracker. It now accounts for 6% of US cases. This was expected, as the variant has been circulating in Europe, said Amesh Adalja, MD, a senior scholar and infectious disease expert at the Center for Health Security at Johns Hopkins University, Baltimore, Maryland.
“There will always be a new variant appearing, and one falling,” he said. “So the fact that this is happening is not surprising.”
Meanwhile, the summer COVID surge has provided postinfection immunity for some people. “What’s likely is, we are going to see substantial protection of the population for several months based on previous infection and in some cases vaccination,” Dr. Osterholm said. That means protection from serious illness, hospitalizations, and deaths (but not necessarily infection). That protection could last through the year or into early 2025.
The timing of 2024’s winter surge will likely be a bit later than 2023’s, said Andrew Pekosz, PhD, a professor and vice chair of molecular microbiology and immunology at Johns Hopkins University, Baltimore, “peaking just after the Christmas/New Year holiday.”
During the 2023-2024 season, weekly COVID hospitalizations peaked the week of Dec. 30, said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill and a member of the COVID-19 Scenario Modeling Hub.
But variants are unpredictable. “There’s a chance that the XEC variant may take off and spread, or might not,” said Dr. Adalja. As of September 28, the Omicron variant KP.3.1.1 was leading, accounting for 58.7% of US cases, according to the CDC.
While Dr. Adalja agreed that 2024’s COVID season will probably be like 2023’s, “we have to be prepared for cases and hospitalizations going up,” he said, “but not to the point of a crisis.” A return to lockdowns and social distancing is unlikely.
Still, older adults and others at higher risk of getting very sick from COVID should consider masking during travel, said Rajendram Rajnarayanan, PhD, MSc, an associate professor at the New York Institute of Technology College of Osteopathic Medicine at Arkansas State University, Jonesboro.
Flu Forecasts
Predicting flu season this early is hard, said Jeffrey Shaman, PhD, a professor of environmental health sciences and professor of climate at Colombia University, New York.
“You can look at the CDC forecast and use it as a very loose guide right now,” said Dr. Shaman, who won the CDC’s first “Predict the Influenza Season Challenge” in 2014. “Until there is actually flu, it’s like trying to predict the landfall of a hurricane.” Flu activity remained low as of September 14 (the most current data available), according to the CDC.
When flu activity picks up, typically in mid-October or November, experts look at the dominant strain, exposure to similar strains in previous years, and how well-matched the current flu vaccine is to that dominant strain, Dr. Shaman said. Vaccine makers must make an educated guess months in advance regarding which strain to target, to allow time for production.
The vaccination rate plays a role, too, but that tends to remain constant, Dr. Shaman said. According to the CDC, less than half of adults age 18 and up got a flu vaccination last year.
Experts also consider flu patterns in the Southern Hemisphere, where 2024 flu activity has mostly involved two subtypes of influenza A — H1N1 and H3N2 — and some influenza B, the CDC found.
How Well Do This Year’s Vaccines and Viruses Match Up?
The FDA has authorized three updated COVID vaccines for this fall. Novavax targets the JN.1 strain of SARS-CoV-2, the virus that causes COVID-19. Both mRNA vaccines, Moderna and Pfizer, target KP.2, a descendant of JN.1. All three target current predominant variants, and any one of them is recommended by the CDC.
The vaccines are a good “though not perfect match to virtually all the circulating variants of SARS-CoV-2,” said Dr. Pekosz.
Experts said that the shots will protect against the XEC variant.
“XEC and its sublineages are expected to be the dominant fall/winter variant group,” said Dr. Rajnarayanan.
This year’s flu vaccines, all trivalent (protecting against three viruses), will target the three strains expected to circulate — H1N1, H3N2, and influenza B (Victoria), according to the CDC.
People should still get vaccinated, Dr. Adalja said, and use home tests for flu and COVID and take antivirals promptly when needed. The goal should not be status quo but rather fewer COVID and flu hospitalizations and deaths.
A version of this article first appeared on WebMD.com.
Guidance for Practicing Primary Care: World Health Organization’s Updated Influenza Guidelines for 2024
As primary care physicians, we are often the first ones patients see when they become infected with influenza. According to Centers for Disease Control and Prevention statistics, approximately 5%-20% of the US population will be infected with influenza every year. Additionally, more than 200,000 of these patients will be hospitalized because of complications related to influenza.
Earlier in September, the World Health Organization (WHO) issued its latest clinical practice guidelines for influenza for the 2024-2025 season. This is a 213-page document aimed at healthcare providers who treat patients infected with influenza. It includes treatment for those with severe and nonsevere influenza infections, those in both the outpatient and hospitalized setting, as well as medication prophylaxis for those exposed to the virus. Additionally, it defines risk estimates for those who are at risk of being hospitalized or dying. In contrast, previous updates focused on management of severe influenza or those at risk of severe influenza.
These guidelines cover recommendations regarding all the antiviral medications for treating influenza used around the world. For the purpose of this article, we will focus on those most commonly used in the United States.
A newer medication discussed was baloxavir. It is recommended to be used for patients with nonsevere influenza who are at high risk for progression to severe disease. The advice is to not use it for those with little risk of progression to severe disease. Oseltamivir is recommended for those with severe infection.
The guidelines recommend against using antibiotics for those who have a low likelihood of having a bacterial coinfection. As primary care doctors, we often prescribe medications to help with symptoms. These guidelines recommend against the use of corticosteroids and antibiotics but did advise that NSAIDs could be used for symptom relief.
One of the important parts of these guidelines is prevention in patients who have been exposed but are asymptomatic. They recommend baloxavir or oseltamivir but only for those patients who are at high risk of being hospitalized if they were to become infected. Any of the antivirals can be used for patients who are exposed to the novel influenza A, which is associated with a higher mortality rate. Caution when prescribing antivirals is recommended in immunocompromised patients because there is more drug resistance seen in these patients.
These updates also discuss the use of different influenza tests. In the outpatient setting, primary doctors don’t have time for test results that may take 2 days to come back. Only rapid tests make the sense in the primary care setting. Additionally, in the age of COVID, it is important to make an accurate diagnosis so we should be testing patients. There is resistance seen with the antivirals we prescribe for influenza so prescribing them empirically without a confirmed diagnosis of influenza may be doing more harm than good.
One gap in these recommendations is vaccination. This topic was not covered at all. It would be helpful to have a strategy in place to prevent infection in populations rather than focusing just on exposed individuals. A discussion of when and who and to vaccinate would be helpful. Research into the effectiveness of vaccines is key and more accurate development of a season’s influenza vaccine would be beneficial. Currently, there is much vaccine misinformation being spread around. Education and information regarding influenza vaccines, especially coming from WHO, is crucial.
Another failure of these recommendations is that the guidelines apply only to those who present within a few days of becoming symptomatic. As family doctors, we know many of our patients self-treat or consult Google. They often don’t come for medical care until they’ve been sick for a week or longer. There are no guidelines for these patients.
In general, these guidelines are comprehensive and do a great job discussing the current medications available. However, more is needed to increase vaccination rates. Patients need to know that if they may be sick with influenza, they need to seek medical care as soon as possible. We, as family doctors, need to do a better job of risk-stratifying our patients and prescribing prophylactic medication when suitable. Every infection we prevent aids in the health of our community and the global population at large.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no relevant conflicts of interest.
As primary care physicians, we are often the first ones patients see when they become infected with influenza. According to Centers for Disease Control and Prevention statistics, approximately 5%-20% of the US population will be infected with influenza every year. Additionally, more than 200,000 of these patients will be hospitalized because of complications related to influenza.
Earlier in September, the World Health Organization (WHO) issued its latest clinical practice guidelines for influenza for the 2024-2025 season. This is a 213-page document aimed at healthcare providers who treat patients infected with influenza. It includes treatment for those with severe and nonsevere influenza infections, those in both the outpatient and hospitalized setting, as well as medication prophylaxis for those exposed to the virus. Additionally, it defines risk estimates for those who are at risk of being hospitalized or dying. In contrast, previous updates focused on management of severe influenza or those at risk of severe influenza.
These guidelines cover recommendations regarding all the antiviral medications for treating influenza used around the world. For the purpose of this article, we will focus on those most commonly used in the United States.
A newer medication discussed was baloxavir. It is recommended to be used for patients with nonsevere influenza who are at high risk for progression to severe disease. The advice is to not use it for those with little risk of progression to severe disease. Oseltamivir is recommended for those with severe infection.
The guidelines recommend against using antibiotics for those who have a low likelihood of having a bacterial coinfection. As primary care doctors, we often prescribe medications to help with symptoms. These guidelines recommend against the use of corticosteroids and antibiotics but did advise that NSAIDs could be used for symptom relief.
One of the important parts of these guidelines is prevention in patients who have been exposed but are asymptomatic. They recommend baloxavir or oseltamivir but only for those patients who are at high risk of being hospitalized if they were to become infected. Any of the antivirals can be used for patients who are exposed to the novel influenza A, which is associated with a higher mortality rate. Caution when prescribing antivirals is recommended in immunocompromised patients because there is more drug resistance seen in these patients.
These updates also discuss the use of different influenza tests. In the outpatient setting, primary doctors don’t have time for test results that may take 2 days to come back. Only rapid tests make the sense in the primary care setting. Additionally, in the age of COVID, it is important to make an accurate diagnosis so we should be testing patients. There is resistance seen with the antivirals we prescribe for influenza so prescribing them empirically without a confirmed diagnosis of influenza may be doing more harm than good.
One gap in these recommendations is vaccination. This topic was not covered at all. It would be helpful to have a strategy in place to prevent infection in populations rather than focusing just on exposed individuals. A discussion of when and who and to vaccinate would be helpful. Research into the effectiveness of vaccines is key and more accurate development of a season’s influenza vaccine would be beneficial. Currently, there is much vaccine misinformation being spread around. Education and information regarding influenza vaccines, especially coming from WHO, is crucial.
Another failure of these recommendations is that the guidelines apply only to those who present within a few days of becoming symptomatic. As family doctors, we know many of our patients self-treat or consult Google. They often don’t come for medical care until they’ve been sick for a week or longer. There are no guidelines for these patients.
In general, these guidelines are comprehensive and do a great job discussing the current medications available. However, more is needed to increase vaccination rates. Patients need to know that if they may be sick with influenza, they need to seek medical care as soon as possible. We, as family doctors, need to do a better job of risk-stratifying our patients and prescribing prophylactic medication when suitable. Every infection we prevent aids in the health of our community and the global population at large.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no relevant conflicts of interest.
As primary care physicians, we are often the first ones patients see when they become infected with influenza. According to Centers for Disease Control and Prevention statistics, approximately 5%-20% of the US population will be infected with influenza every year. Additionally, more than 200,000 of these patients will be hospitalized because of complications related to influenza.
Earlier in September, the World Health Organization (WHO) issued its latest clinical practice guidelines for influenza for the 2024-2025 season. This is a 213-page document aimed at healthcare providers who treat patients infected with influenza. It includes treatment for those with severe and nonsevere influenza infections, those in both the outpatient and hospitalized setting, as well as medication prophylaxis for those exposed to the virus. Additionally, it defines risk estimates for those who are at risk of being hospitalized or dying. In contrast, previous updates focused on management of severe influenza or those at risk of severe influenza.
These guidelines cover recommendations regarding all the antiviral medications for treating influenza used around the world. For the purpose of this article, we will focus on those most commonly used in the United States.
A newer medication discussed was baloxavir. It is recommended to be used for patients with nonsevere influenza who are at high risk for progression to severe disease. The advice is to not use it for those with little risk of progression to severe disease. Oseltamivir is recommended for those with severe infection.
The guidelines recommend against using antibiotics for those who have a low likelihood of having a bacterial coinfection. As primary care doctors, we often prescribe medications to help with symptoms. These guidelines recommend against the use of corticosteroids and antibiotics but did advise that NSAIDs could be used for symptom relief.
One of the important parts of these guidelines is prevention in patients who have been exposed but are asymptomatic. They recommend baloxavir or oseltamivir but only for those patients who are at high risk of being hospitalized if they were to become infected. Any of the antivirals can be used for patients who are exposed to the novel influenza A, which is associated with a higher mortality rate. Caution when prescribing antivirals is recommended in immunocompromised patients because there is more drug resistance seen in these patients.
These updates also discuss the use of different influenza tests. In the outpatient setting, primary doctors don’t have time for test results that may take 2 days to come back. Only rapid tests make the sense in the primary care setting. Additionally, in the age of COVID, it is important to make an accurate diagnosis so we should be testing patients. There is resistance seen with the antivirals we prescribe for influenza so prescribing them empirically without a confirmed diagnosis of influenza may be doing more harm than good.
One gap in these recommendations is vaccination. This topic was not covered at all. It would be helpful to have a strategy in place to prevent infection in populations rather than focusing just on exposed individuals. A discussion of when and who and to vaccinate would be helpful. Research into the effectiveness of vaccines is key and more accurate development of a season’s influenza vaccine would be beneficial. Currently, there is much vaccine misinformation being spread around. Education and information regarding influenza vaccines, especially coming from WHO, is crucial.
Another failure of these recommendations is that the guidelines apply only to those who present within a few days of becoming symptomatic. As family doctors, we know many of our patients self-treat or consult Google. They often don’t come for medical care until they’ve been sick for a week or longer. There are no guidelines for these patients.
In general, these guidelines are comprehensive and do a great job discussing the current medications available. However, more is needed to increase vaccination rates. Patients need to know that if they may be sick with influenza, they need to seek medical care as soon as possible. We, as family doctors, need to do a better job of risk-stratifying our patients and prescribing prophylactic medication when suitable. Every infection we prevent aids in the health of our community and the global population at large.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no relevant conflicts of interest.
Species Possibly Responsible for COVID Pandemic Identified
The origin of the COVID-19 pandemic has sparked much debate, and various hypotheses have been put forward.
“My colleagues and I have examined the issue with an open mind, taking into account all possible hypotheses. The laboratory origin hypothesis was legitimate and deserved to be investigated,” Florence Débarre, PhD, a research director at the French National Center for Scientific Research at the Institute of Ecology and Environmental Sciences in Paris, France, told this news organization. Nevertheless,
“We studied data from environmental samples taken at the Huanan market in Wuhan shortly after its closure in early 2020,” said Dr. Débarre. The data were shared by the Chinese Center for Disease Control and Prevention on open and public databases. They include the raw genetic sequences of more than 800 samples collected at the Huanan market, on cages and carts, on the floors and walls of the stalls, and in the pipes and sewers.
These data allowed researchers to highlight the co-presence at this location of genetic material from the SARS-CoV-2 virus and certain wild animals. Masked palm civets, which are wild canids similar to foxes, with a dark facial mask similar to that of raccoons, and civets, small carnivorous mammals close to mongooses, were at the site.
“These species were already involved in the emergence of the SARS epidemic in the early 2000s and considered to facilitate the transmission of the virus from animals to humans,” said Dr. Débarre.
These animals were identified based on their DNA and located in the southwest part of the market, which is also a hotspot where many samples tested positive for SARS-CoV-2.
“There is a particular stall where the virus and the animals were found,” she said.
Since the data used are based on environmental samples, it is not possible to formally demonstrate that the animals were infected, but the discovery of virus samples located in the same place as the genetic material of these animals suggests that they were.
“There were samples taken from some animals at the market, but not from others, as they had already been evacuated when the sampling services arrived,” said Dr. Débarre. These results add to a large body of evidence that all points in the same direction: an animal origin at the Wuhan market.
The team also found other zoonotic viruses, such as avian flu. “This study confirms that live animal markets pose a high health risk, especially when they are at the heart of urban centers,” said Dr. Débarre. “It can provide avenues for prevention, particularly by limiting interactions between humans and wild fauna.”
This story was translated from the Medscape French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
The origin of the COVID-19 pandemic has sparked much debate, and various hypotheses have been put forward.
“My colleagues and I have examined the issue with an open mind, taking into account all possible hypotheses. The laboratory origin hypothesis was legitimate and deserved to be investigated,” Florence Débarre, PhD, a research director at the French National Center for Scientific Research at the Institute of Ecology and Environmental Sciences in Paris, France, told this news organization. Nevertheless,
“We studied data from environmental samples taken at the Huanan market in Wuhan shortly after its closure in early 2020,” said Dr. Débarre. The data were shared by the Chinese Center for Disease Control and Prevention on open and public databases. They include the raw genetic sequences of more than 800 samples collected at the Huanan market, on cages and carts, on the floors and walls of the stalls, and in the pipes and sewers.
These data allowed researchers to highlight the co-presence at this location of genetic material from the SARS-CoV-2 virus and certain wild animals. Masked palm civets, which are wild canids similar to foxes, with a dark facial mask similar to that of raccoons, and civets, small carnivorous mammals close to mongooses, were at the site.
“These species were already involved in the emergence of the SARS epidemic in the early 2000s and considered to facilitate the transmission of the virus from animals to humans,” said Dr. Débarre.
These animals were identified based on their DNA and located in the southwest part of the market, which is also a hotspot where many samples tested positive for SARS-CoV-2.
“There is a particular stall where the virus and the animals were found,” she said.
Since the data used are based on environmental samples, it is not possible to formally demonstrate that the animals were infected, but the discovery of virus samples located in the same place as the genetic material of these animals suggests that they were.
“There were samples taken from some animals at the market, but not from others, as they had already been evacuated when the sampling services arrived,” said Dr. Débarre. These results add to a large body of evidence that all points in the same direction: an animal origin at the Wuhan market.
The team also found other zoonotic viruses, such as avian flu. “This study confirms that live animal markets pose a high health risk, especially when they are at the heart of urban centers,” said Dr. Débarre. “It can provide avenues for prevention, particularly by limiting interactions between humans and wild fauna.”
This story was translated from the Medscape French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
The origin of the COVID-19 pandemic has sparked much debate, and various hypotheses have been put forward.
“My colleagues and I have examined the issue with an open mind, taking into account all possible hypotheses. The laboratory origin hypothesis was legitimate and deserved to be investigated,” Florence Débarre, PhD, a research director at the French National Center for Scientific Research at the Institute of Ecology and Environmental Sciences in Paris, France, told this news organization. Nevertheless,
“We studied data from environmental samples taken at the Huanan market in Wuhan shortly after its closure in early 2020,” said Dr. Débarre. The data were shared by the Chinese Center for Disease Control and Prevention on open and public databases. They include the raw genetic sequences of more than 800 samples collected at the Huanan market, on cages and carts, on the floors and walls of the stalls, and in the pipes and sewers.
These data allowed researchers to highlight the co-presence at this location of genetic material from the SARS-CoV-2 virus and certain wild animals. Masked palm civets, which are wild canids similar to foxes, with a dark facial mask similar to that of raccoons, and civets, small carnivorous mammals close to mongooses, were at the site.
“These species were already involved in the emergence of the SARS epidemic in the early 2000s and considered to facilitate the transmission of the virus from animals to humans,” said Dr. Débarre.
These animals were identified based on their DNA and located in the southwest part of the market, which is also a hotspot where many samples tested positive for SARS-CoV-2.
“There is a particular stall where the virus and the animals were found,” she said.
Since the data used are based on environmental samples, it is not possible to formally demonstrate that the animals were infected, but the discovery of virus samples located in the same place as the genetic material of these animals suggests that they were.
“There were samples taken from some animals at the market, but not from others, as they had already been evacuated when the sampling services arrived,” said Dr. Débarre. These results add to a large body of evidence that all points in the same direction: an animal origin at the Wuhan market.
The team also found other zoonotic viruses, such as avian flu. “This study confirms that live animal markets pose a high health risk, especially when they are at the heart of urban centers,” said Dr. Débarre. “It can provide avenues for prevention, particularly by limiting interactions between humans and wild fauna.”
This story was translated from the Medscape French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Nasal Staph Aureus Carriage Linked to Surgical Infections
published in the August issue of Open Forum Infectious Diseases.
“This was a pan-European study with many hospitals, many different clinical settings, and as far as I’m aware, it hasn’t been done before. [The new study] covers a lot of European countries and a lot of surgical specialties,” said lead author Jan Kluytmans, MD. The study also captures the current state of preventive strategies in surgery, such as changes in air flow, dress, and skin preparation, he added.
The study included 5004 patients from 33 hospitals in ten European countries, of whom 67.3% were found to be SA carriers. The median age was 65 years, and 49.8% of patients were male. Open cardiac, and knee and hip prosthesis surgeries made up the largest fraction, but there were 12 types of surgery included in the study.
There were 100 SA surgical site or blood infections. The researchers found an association between surgical site or blood infection and SA carriage at any site (adjusted hazard ratio [aHR], 4.6; 95% CI, 2.1-10.0) and nasal SA carriage (aHR, 4.2; 95% CI, 2.0-8.6). Extranasal SA carriage was not associated with an increased infection risk.
Each 1-unit increase in nasal bacteria was associated with an increase in infection risk (aHR, 1.23; 95% CI, 1.05-1.43).
A strength of the study is that it is the largest prospective study yet conducted on SA carriage in surgical patients, but the researchers were unable to do a subgroup of methicillin-resistant SA (MRSA) due to small numbers of infections.
The study confirms the value of the decolonization strategy, which the World Health Organization has endorsed with the highest level of scientific evidence that is available in preventive strategies in surgery. WHO strongly recommends decolonization for cardiothoracic and orthopedic surgery using intranasal applications of mupirocin 2% ointment with or without a combination of chlorhexidine gluconate body wash. It has a conditional recommendation for a similar procedure before other types of surgery.
However, “It is not widely practiced, and although that was not a surprise to me, I think it’s really disappointing to see that proven effective strategies are not being practiced,” said Dr. Kluytmans, professor of medical microbiology at University Medical Center Utrecht, Utrecht University, the Netherlands. “If I would come into surgery being a carrier, and not be decolonized, I would really be quite angry because it puts you at risk, which is preventable. I think that’s something we owe to our patients,” he said.
He said that some may have concerns about the potential for decolonization to contribute to antibiotic resistance, but the short-term prophylaxis — typically a few days — should not foster resistance, according to Dr. Kluytmans. “If you use it short term, just before surgery, it has been shown in many studies that resistance isn’t a big problem and it can be monitored.”
The link specifically to SA nasal carriage is a mystery, according to Dr. Kluytmans. “It puzzles me still how it gets from the nares to the wound during surgery. So that’s my million-dollar question that I would like to resolve. We would like to study it, but we haven’t quite a bright idea how to do that,” he said.
The results are compelling, according to Heather Evans, MD, who was asked for comment. “On the face of it, this looks like a no-brainer. We should be decolonizing all patients that go to the operating room, and it’s not a terribly unpleasant thing for a patient to undergo to have decolonization done. Particularly for patients who are at higher risk for having a severe complication, like someone that has an operation that’s involving an implant, for example, I think it really makes a lot of sense to do this low-cost intervention for those patients,” said Dr. Evans, professor of medicine at The Medical University of South Carolina as well as the president of the Surgical Infection Society.
She noted that many facilities test for methicillin-resistant SA, but usual not SA more broadly. “This is a very interesting and compelling study that makes us rethink that, and maybe it isn’t even worth testing to see if you have staph aureus, maybe we should just be putting Betadine in everyone’s nostrils when they come to the operating room. It just seems like it would be a pretty low-cost intervention and something that could potentially have a big impact,” said Dr. Evans.
Although she was impressed by the study, Dr. Evans noted that the researchers tested for carriage at sites unrelated to the surgical site. “It really made me wonder if it would have added even more credibility to the study if there had been a sample taken after surgical prep was done to demonstrate that there is actually no staph aureus present on the skin at the time that the wound was made,” she said.
The question ties into the recent “Trojan horse” hypothesis, which suggests that endemic carriage of bacteria is responsible for most surgical site infections, rather than the long-held belief that operating room contamination is to blame. “That would sort of fly with this study, that the patient is walking around with Staph aureus and not necessarily on their skin or at their surgical site, but it’s endemic in their body,” said Dr. Evans.
Dr. Kluytmans and Dr. Evans have no relevant financial disclosures.
published in the August issue of Open Forum Infectious Diseases.
“This was a pan-European study with many hospitals, many different clinical settings, and as far as I’m aware, it hasn’t been done before. [The new study] covers a lot of European countries and a lot of surgical specialties,” said lead author Jan Kluytmans, MD. The study also captures the current state of preventive strategies in surgery, such as changes in air flow, dress, and skin preparation, he added.
The study included 5004 patients from 33 hospitals in ten European countries, of whom 67.3% were found to be SA carriers. The median age was 65 years, and 49.8% of patients were male. Open cardiac, and knee and hip prosthesis surgeries made up the largest fraction, but there were 12 types of surgery included in the study.
There were 100 SA surgical site or blood infections. The researchers found an association between surgical site or blood infection and SA carriage at any site (adjusted hazard ratio [aHR], 4.6; 95% CI, 2.1-10.0) and nasal SA carriage (aHR, 4.2; 95% CI, 2.0-8.6). Extranasal SA carriage was not associated with an increased infection risk.
Each 1-unit increase in nasal bacteria was associated with an increase in infection risk (aHR, 1.23; 95% CI, 1.05-1.43).
A strength of the study is that it is the largest prospective study yet conducted on SA carriage in surgical patients, but the researchers were unable to do a subgroup of methicillin-resistant SA (MRSA) due to small numbers of infections.
The study confirms the value of the decolonization strategy, which the World Health Organization has endorsed with the highest level of scientific evidence that is available in preventive strategies in surgery. WHO strongly recommends decolonization for cardiothoracic and orthopedic surgery using intranasal applications of mupirocin 2% ointment with or without a combination of chlorhexidine gluconate body wash. It has a conditional recommendation for a similar procedure before other types of surgery.
However, “It is not widely practiced, and although that was not a surprise to me, I think it’s really disappointing to see that proven effective strategies are not being practiced,” said Dr. Kluytmans, professor of medical microbiology at University Medical Center Utrecht, Utrecht University, the Netherlands. “If I would come into surgery being a carrier, and not be decolonized, I would really be quite angry because it puts you at risk, which is preventable. I think that’s something we owe to our patients,” he said.
He said that some may have concerns about the potential for decolonization to contribute to antibiotic resistance, but the short-term prophylaxis — typically a few days — should not foster resistance, according to Dr. Kluytmans. “If you use it short term, just before surgery, it has been shown in many studies that resistance isn’t a big problem and it can be monitored.”
The link specifically to SA nasal carriage is a mystery, according to Dr. Kluytmans. “It puzzles me still how it gets from the nares to the wound during surgery. So that’s my million-dollar question that I would like to resolve. We would like to study it, but we haven’t quite a bright idea how to do that,” he said.
The results are compelling, according to Heather Evans, MD, who was asked for comment. “On the face of it, this looks like a no-brainer. We should be decolonizing all patients that go to the operating room, and it’s not a terribly unpleasant thing for a patient to undergo to have decolonization done. Particularly for patients who are at higher risk for having a severe complication, like someone that has an operation that’s involving an implant, for example, I think it really makes a lot of sense to do this low-cost intervention for those patients,” said Dr. Evans, professor of medicine at The Medical University of South Carolina as well as the president of the Surgical Infection Society.
She noted that many facilities test for methicillin-resistant SA, but usual not SA more broadly. “This is a very interesting and compelling study that makes us rethink that, and maybe it isn’t even worth testing to see if you have staph aureus, maybe we should just be putting Betadine in everyone’s nostrils when they come to the operating room. It just seems like it would be a pretty low-cost intervention and something that could potentially have a big impact,” said Dr. Evans.
Although she was impressed by the study, Dr. Evans noted that the researchers tested for carriage at sites unrelated to the surgical site. “It really made me wonder if it would have added even more credibility to the study if there had been a sample taken after surgical prep was done to demonstrate that there is actually no staph aureus present on the skin at the time that the wound was made,” she said.
The question ties into the recent “Trojan horse” hypothesis, which suggests that endemic carriage of bacteria is responsible for most surgical site infections, rather than the long-held belief that operating room contamination is to blame. “That would sort of fly with this study, that the patient is walking around with Staph aureus and not necessarily on their skin or at their surgical site, but it’s endemic in their body,” said Dr. Evans.
Dr. Kluytmans and Dr. Evans have no relevant financial disclosures.
published in the August issue of Open Forum Infectious Diseases.
“This was a pan-European study with many hospitals, many different clinical settings, and as far as I’m aware, it hasn’t been done before. [The new study] covers a lot of European countries and a lot of surgical specialties,” said lead author Jan Kluytmans, MD. The study also captures the current state of preventive strategies in surgery, such as changes in air flow, dress, and skin preparation, he added.
The study included 5004 patients from 33 hospitals in ten European countries, of whom 67.3% were found to be SA carriers. The median age was 65 years, and 49.8% of patients were male. Open cardiac, and knee and hip prosthesis surgeries made up the largest fraction, but there were 12 types of surgery included in the study.
There were 100 SA surgical site or blood infections. The researchers found an association between surgical site or blood infection and SA carriage at any site (adjusted hazard ratio [aHR], 4.6; 95% CI, 2.1-10.0) and nasal SA carriage (aHR, 4.2; 95% CI, 2.0-8.6). Extranasal SA carriage was not associated with an increased infection risk.
Each 1-unit increase in nasal bacteria was associated with an increase in infection risk (aHR, 1.23; 95% CI, 1.05-1.43).
A strength of the study is that it is the largest prospective study yet conducted on SA carriage in surgical patients, but the researchers were unable to do a subgroup of methicillin-resistant SA (MRSA) due to small numbers of infections.
The study confirms the value of the decolonization strategy, which the World Health Organization has endorsed with the highest level of scientific evidence that is available in preventive strategies in surgery. WHO strongly recommends decolonization for cardiothoracic and orthopedic surgery using intranasal applications of mupirocin 2% ointment with or without a combination of chlorhexidine gluconate body wash. It has a conditional recommendation for a similar procedure before other types of surgery.
However, “It is not widely practiced, and although that was not a surprise to me, I think it’s really disappointing to see that proven effective strategies are not being practiced,” said Dr. Kluytmans, professor of medical microbiology at University Medical Center Utrecht, Utrecht University, the Netherlands. “If I would come into surgery being a carrier, and not be decolonized, I would really be quite angry because it puts you at risk, which is preventable. I think that’s something we owe to our patients,” he said.
He said that some may have concerns about the potential for decolonization to contribute to antibiotic resistance, but the short-term prophylaxis — typically a few days — should not foster resistance, according to Dr. Kluytmans. “If you use it short term, just before surgery, it has been shown in many studies that resistance isn’t a big problem and it can be monitored.”
The link specifically to SA nasal carriage is a mystery, according to Dr. Kluytmans. “It puzzles me still how it gets from the nares to the wound during surgery. So that’s my million-dollar question that I would like to resolve. We would like to study it, but we haven’t quite a bright idea how to do that,” he said.
The results are compelling, according to Heather Evans, MD, who was asked for comment. “On the face of it, this looks like a no-brainer. We should be decolonizing all patients that go to the operating room, and it’s not a terribly unpleasant thing for a patient to undergo to have decolonization done. Particularly for patients who are at higher risk for having a severe complication, like someone that has an operation that’s involving an implant, for example, I think it really makes a lot of sense to do this low-cost intervention for those patients,” said Dr. Evans, professor of medicine at The Medical University of South Carolina as well as the president of the Surgical Infection Society.
She noted that many facilities test for methicillin-resistant SA, but usual not SA more broadly. “This is a very interesting and compelling study that makes us rethink that, and maybe it isn’t even worth testing to see if you have staph aureus, maybe we should just be putting Betadine in everyone’s nostrils when they come to the operating room. It just seems like it would be a pretty low-cost intervention and something that could potentially have a big impact,” said Dr. Evans.
Although she was impressed by the study, Dr. Evans noted that the researchers tested for carriage at sites unrelated to the surgical site. “It really made me wonder if it would have added even more credibility to the study if there had been a sample taken after surgical prep was done to demonstrate that there is actually no staph aureus present on the skin at the time that the wound was made,” she said.
The question ties into the recent “Trojan horse” hypothesis, which suggests that endemic carriage of bacteria is responsible for most surgical site infections, rather than the long-held belief that operating room contamination is to blame. “That would sort of fly with this study, that the patient is walking around with Staph aureus and not necessarily on their skin or at their surgical site, but it’s endemic in their body,” said Dr. Evans.
Dr. Kluytmans and Dr. Evans have no relevant financial disclosures.
Pertussis Rates Up Compared With Recent Years
, according to data from the Centers for Disease Control and Prevention (CDC). Reports from several states illustrate this trend, thought to be due to reduced immunity across the country.
The Alaska Department of Health issued a statement on its website about the significant increase in pertussis cases in the state during the summer, with 90 cases in July and 61 in August, compared with 24 in June and a total of 26 cases in 2023.
Similarly, the Florida Department of Health reported a pertussis increase in July 2024 that was higher than the June 2024 case count and also above the previous 5-year average.
Experts in these and other states suggest that several factors are driving the nationwide increase, including the fact that fewer people are consistently wearing masks. The mass masking during the COVID-19 pandemic caused a significant drop in pertussis, but the latest data suggest a return to prepandemic levels, and waning immunity likely plays a role as well.
Pertussis, also known as whooping cough, typically begins with symptoms similar to those of the common cold, including runny nose, sneezing, mild fever, and cough, according to the CDC. However, babies with whooping cough may experience trouble breathing rather than a cough. The coughing fits often associated with pertussis may not start until 2 weeks after the onset of other symptoms, according to the CDC.
Those who have been vaccinated against pertussis can still become infected, but the risk is lower, and the illness, if it occurs, is likely to be milder. Complications such as apnea, pneumonia, and convulsions can occur in babies younger than 1 year, especially if they have not been vaccinated, according to the CDC.
Beyond Easing Pandemic Precautions
Many respiratory-based infections dipped during the COVID-19 pandemic, almost certainly from the multifactorial interventions of masking, distancing, and the general lack of comingling, said David J. Cennimo, MD, associate professor of medicine & pediatrics in the Division of Infectious Diseases at Rutgers New Jersey Medical School, Newark, New Jersey, in an interview.
The number of cases of many of these diseases returned to previous levels after COVID-19 restrictions were lifted, he said.
“However, we know pertussis immunity wanes over time. Children get DTaP at 2, 4, 6, and 15 months, and a Tdap booster at 11-12 years old gets them to adulthood,” Dr. Cennimo said. Adults should be getting a Tdap every 10 years, he added.
The latest available CDC data indicate that Tdap vaccine coverage in adults is approximately 40%, which means that there may be a large number of susceptible people who can become infected and propagate to others, said Dr. Cennimo.
Not Just the Young Ones
A recent pertussis outbreak among college students in Virginia highlighted the fact that the infection can affect all ages, and that the effectiveness of childhood vaccines may decrease over time. The majority of the recently diagnosed cases occurred in individuals who had been previously vaccinated, according to a press release from the Virginia Department of Health.
Clinical Clues
The initial stage of pertussis infection looks like a common cold with symptoms of upper respiratory infection, Dr. Cennimo told this news organization. “Unless there is reason to suspect pertussis exposure, it would almost certainly be missed,” he noted.
The characteristic barking/seal-like cough is mostly seen in children, said Dr. Cennimo. Adults and children can experience coughing fits that can lead to shortness of breath and/or vomiting, which would raise suspicion for pertussis, but is not universally present, he said. The convalescent stage of pertussis can be prolonged and is characterized by chronic coughing. “In the past, pertussis had been called the 100-day cough,” and at that point, treatment is ineffective, Dr. Cennimo said.
In clinical practice, “I advise everyone to get the Tdap vaccine every 10 years,” and remember that the “Td” is the every 10-year tetanus shot as well, Dr. Cennimo told this news organization. Reassure patients that the Tdap can be given with other vaccines, he said, and remind patients that, as with any of the respiratory illnesses, they should stay home if sick, cover a cough, consider wearing a mask in public, and wash hands frequently, he said.
Dr. Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
, according to data from the Centers for Disease Control and Prevention (CDC). Reports from several states illustrate this trend, thought to be due to reduced immunity across the country.
The Alaska Department of Health issued a statement on its website about the significant increase in pertussis cases in the state during the summer, with 90 cases in July and 61 in August, compared with 24 in June and a total of 26 cases in 2023.
Similarly, the Florida Department of Health reported a pertussis increase in July 2024 that was higher than the June 2024 case count and also above the previous 5-year average.
Experts in these and other states suggest that several factors are driving the nationwide increase, including the fact that fewer people are consistently wearing masks. The mass masking during the COVID-19 pandemic caused a significant drop in pertussis, but the latest data suggest a return to prepandemic levels, and waning immunity likely plays a role as well.
Pertussis, also known as whooping cough, typically begins with symptoms similar to those of the common cold, including runny nose, sneezing, mild fever, and cough, according to the CDC. However, babies with whooping cough may experience trouble breathing rather than a cough. The coughing fits often associated with pertussis may not start until 2 weeks after the onset of other symptoms, according to the CDC.
Those who have been vaccinated against pertussis can still become infected, but the risk is lower, and the illness, if it occurs, is likely to be milder. Complications such as apnea, pneumonia, and convulsions can occur in babies younger than 1 year, especially if they have not been vaccinated, according to the CDC.
Beyond Easing Pandemic Precautions
Many respiratory-based infections dipped during the COVID-19 pandemic, almost certainly from the multifactorial interventions of masking, distancing, and the general lack of comingling, said David J. Cennimo, MD, associate professor of medicine & pediatrics in the Division of Infectious Diseases at Rutgers New Jersey Medical School, Newark, New Jersey, in an interview.
The number of cases of many of these diseases returned to previous levels after COVID-19 restrictions were lifted, he said.
“However, we know pertussis immunity wanes over time. Children get DTaP at 2, 4, 6, and 15 months, and a Tdap booster at 11-12 years old gets them to adulthood,” Dr. Cennimo said. Adults should be getting a Tdap every 10 years, he added.
The latest available CDC data indicate that Tdap vaccine coverage in adults is approximately 40%, which means that there may be a large number of susceptible people who can become infected and propagate to others, said Dr. Cennimo.
Not Just the Young Ones
A recent pertussis outbreak among college students in Virginia highlighted the fact that the infection can affect all ages, and that the effectiveness of childhood vaccines may decrease over time. The majority of the recently diagnosed cases occurred in individuals who had been previously vaccinated, according to a press release from the Virginia Department of Health.
Clinical Clues
The initial stage of pertussis infection looks like a common cold with symptoms of upper respiratory infection, Dr. Cennimo told this news organization. “Unless there is reason to suspect pertussis exposure, it would almost certainly be missed,” he noted.
The characteristic barking/seal-like cough is mostly seen in children, said Dr. Cennimo. Adults and children can experience coughing fits that can lead to shortness of breath and/or vomiting, which would raise suspicion for pertussis, but is not universally present, he said. The convalescent stage of pertussis can be prolonged and is characterized by chronic coughing. “In the past, pertussis had been called the 100-day cough,” and at that point, treatment is ineffective, Dr. Cennimo said.
In clinical practice, “I advise everyone to get the Tdap vaccine every 10 years,” and remember that the “Td” is the every 10-year tetanus shot as well, Dr. Cennimo told this news organization. Reassure patients that the Tdap can be given with other vaccines, he said, and remind patients that, as with any of the respiratory illnesses, they should stay home if sick, cover a cough, consider wearing a mask in public, and wash hands frequently, he said.
Dr. Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
, according to data from the Centers for Disease Control and Prevention (CDC). Reports from several states illustrate this trend, thought to be due to reduced immunity across the country.
The Alaska Department of Health issued a statement on its website about the significant increase in pertussis cases in the state during the summer, with 90 cases in July and 61 in August, compared with 24 in June and a total of 26 cases in 2023.
Similarly, the Florida Department of Health reported a pertussis increase in July 2024 that was higher than the June 2024 case count and also above the previous 5-year average.
Experts in these and other states suggest that several factors are driving the nationwide increase, including the fact that fewer people are consistently wearing masks. The mass masking during the COVID-19 pandemic caused a significant drop in pertussis, but the latest data suggest a return to prepandemic levels, and waning immunity likely plays a role as well.
Pertussis, also known as whooping cough, typically begins with symptoms similar to those of the common cold, including runny nose, sneezing, mild fever, and cough, according to the CDC. However, babies with whooping cough may experience trouble breathing rather than a cough. The coughing fits often associated with pertussis may not start until 2 weeks after the onset of other symptoms, according to the CDC.
Those who have been vaccinated against pertussis can still become infected, but the risk is lower, and the illness, if it occurs, is likely to be milder. Complications such as apnea, pneumonia, and convulsions can occur in babies younger than 1 year, especially if they have not been vaccinated, according to the CDC.
Beyond Easing Pandemic Precautions
Many respiratory-based infections dipped during the COVID-19 pandemic, almost certainly from the multifactorial interventions of masking, distancing, and the general lack of comingling, said David J. Cennimo, MD, associate professor of medicine & pediatrics in the Division of Infectious Diseases at Rutgers New Jersey Medical School, Newark, New Jersey, in an interview.
The number of cases of many of these diseases returned to previous levels after COVID-19 restrictions were lifted, he said.
“However, we know pertussis immunity wanes over time. Children get DTaP at 2, 4, 6, and 15 months, and a Tdap booster at 11-12 years old gets them to adulthood,” Dr. Cennimo said. Adults should be getting a Tdap every 10 years, he added.
The latest available CDC data indicate that Tdap vaccine coverage in adults is approximately 40%, which means that there may be a large number of susceptible people who can become infected and propagate to others, said Dr. Cennimo.
Not Just the Young Ones
A recent pertussis outbreak among college students in Virginia highlighted the fact that the infection can affect all ages, and that the effectiveness of childhood vaccines may decrease over time. The majority of the recently diagnosed cases occurred in individuals who had been previously vaccinated, according to a press release from the Virginia Department of Health.
Clinical Clues
The initial stage of pertussis infection looks like a common cold with symptoms of upper respiratory infection, Dr. Cennimo told this news organization. “Unless there is reason to suspect pertussis exposure, it would almost certainly be missed,” he noted.
The characteristic barking/seal-like cough is mostly seen in children, said Dr. Cennimo. Adults and children can experience coughing fits that can lead to shortness of breath and/or vomiting, which would raise suspicion for pertussis, but is not universally present, he said. The convalescent stage of pertussis can be prolonged and is characterized by chronic coughing. “In the past, pertussis had been called the 100-day cough,” and at that point, treatment is ineffective, Dr. Cennimo said.
In clinical practice, “I advise everyone to get the Tdap vaccine every 10 years,” and remember that the “Td” is the every 10-year tetanus shot as well, Dr. Cennimo told this news organization. Reassure patients that the Tdap can be given with other vaccines, he said, and remind patients that, as with any of the respiratory illnesses, they should stay home if sick, cover a cough, consider wearing a mask in public, and wash hands frequently, he said.
Dr. Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
A Rare Case of a Splenic Abscess as the Origin of Illness in Exudative Pleural Effusion
Splenic abscesses are a rare occurrence that represent a marginal proportion of intra-abdominal infections. One study found splenic abscesses in only 0.14% to 0.70% of autopsies and none of the 540 abdominal abscesses they examined originated in the spleen.1 Patients with splenic abscesses tend to present with nonspecific symptoms such as fevers, chills, and abdominal pain.2 Imaging modalities such as abdominal ultrasound and computed tomography (CT) are vital to the workup and diagnosis identification.2 Splenic abscesses are generally associated with another underlying process, as seen in patients who are affected by endocarditis, trauma, metastatic infection, splenic infarction, or neoplasia.2
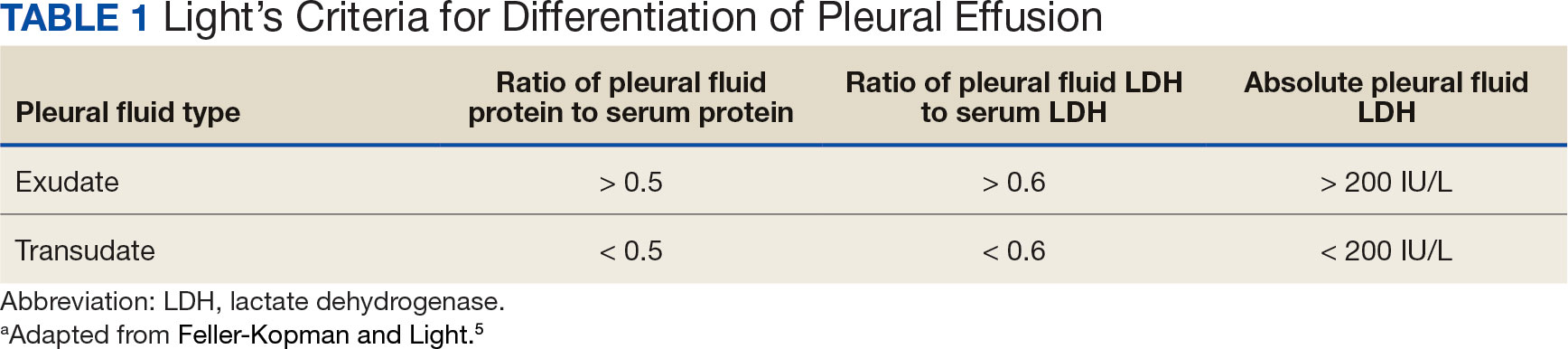
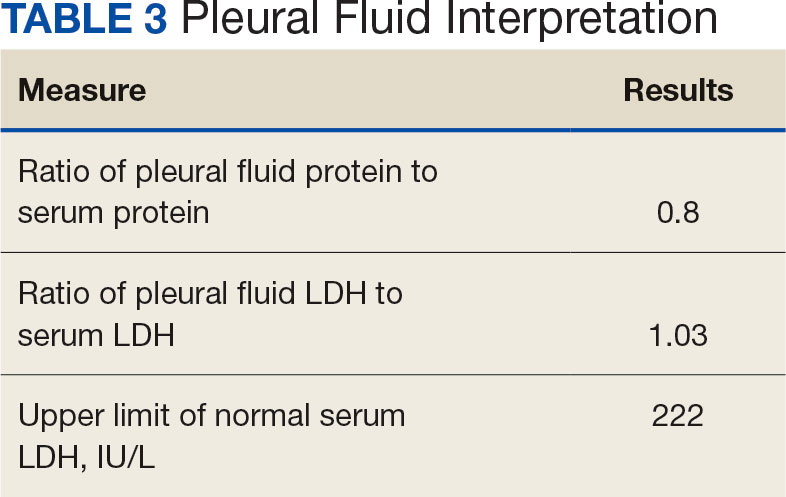
Pleural effusions, or the buildup of fluid within the pleural space, is a common condition typically secondary to another disease.3 Clinical identification of the primary condition may be challenging.3 In the absence of a clear etiology, such as obvious signs of congestive heart failure, further differentiation relies upon pleural fluid analysis, beginning with the distinction between exudate (inflammatory) and transudate (noninflammatory). 3,4 This distinction can be made using Light’s criteria, which relies on protein and lactate dehydrogenase (LDH) ratios between the pleural fluid and serum (Table 1).5 Though rare, half of splenic abscesses are associated with pleural effusion.6 As an inflammatory condition, splenic abscesses have been classically described as a cause of exudative pleural effusions.5,6
A myelodysplastic syndrome is a group of diseases that arise from malignant hematopoietic stem cells, leading to the proliferation of the malignant cells and faulty production of other bone marrow products.7 These disorders can range from single to multilineage dysplasia. Cells are often left in an immature blast form, unable to function appropriately, and vulnerable to destruction. Patients with myeloproliferative disorders frequently suffer from leukopenia and infections attributable to known quantitative and qualitative defects of neutrophils.8
CASE PRESENTATION
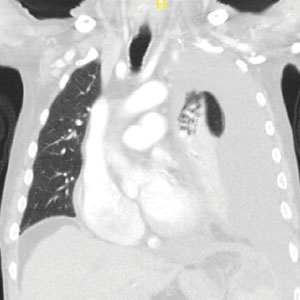
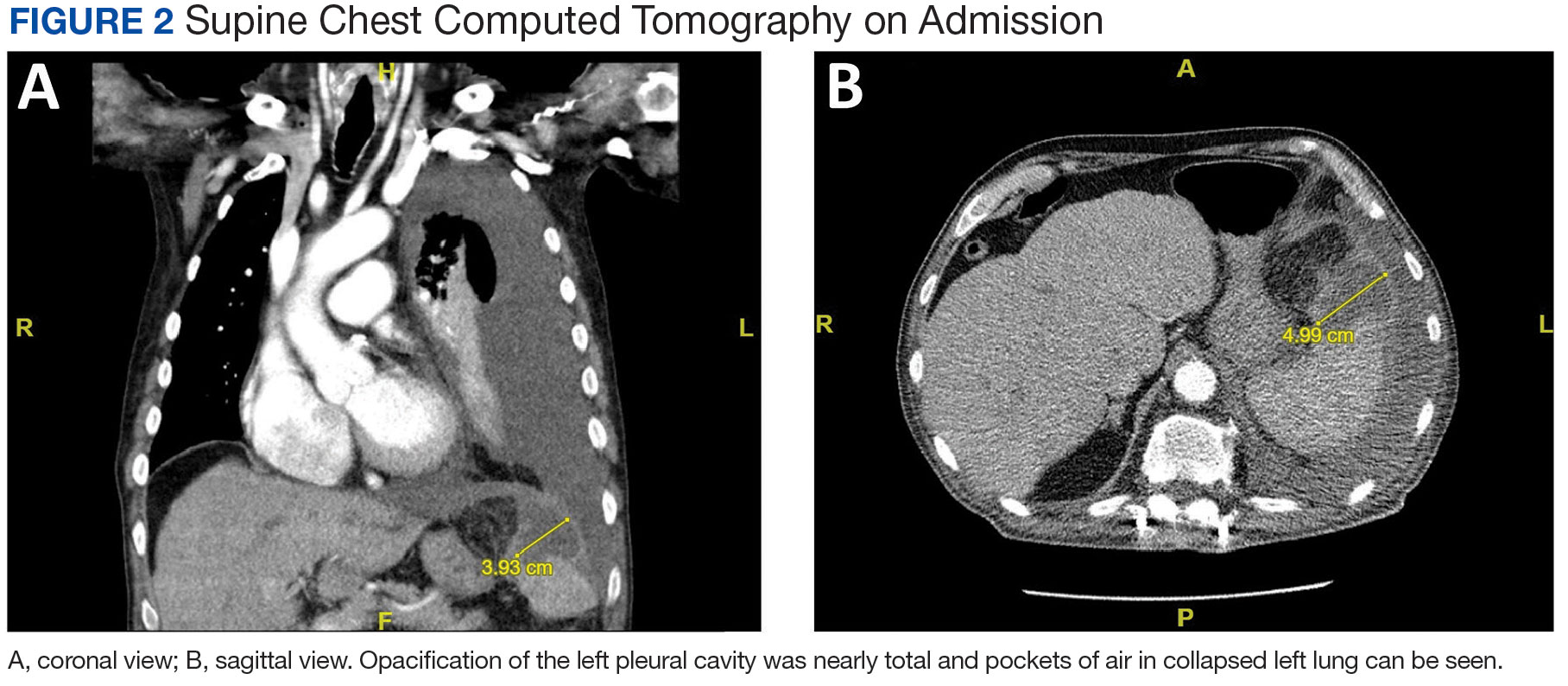
A male aged 80 years presented to the Central Texas Veterans Affairs Hospital (CTVAH) with shortness of breath, weight loss, and fever. On admission, his medical history was notable for atrial fibrillation, myelodysplastic syndrome, hypertension, hyperlipidemia, stable ascending aortic aneurysm, and Vitamin B12 deficiency. A chest CT showed a large left pleural effusion (Figure 1). Additionally, the radiology report noted a nonspecific 4- to 5-cm lobulated subdiaphragmatic mass within the anterior dome of the spleen with surrounding soft tissue swelling and splenomegaly (Figure 2).
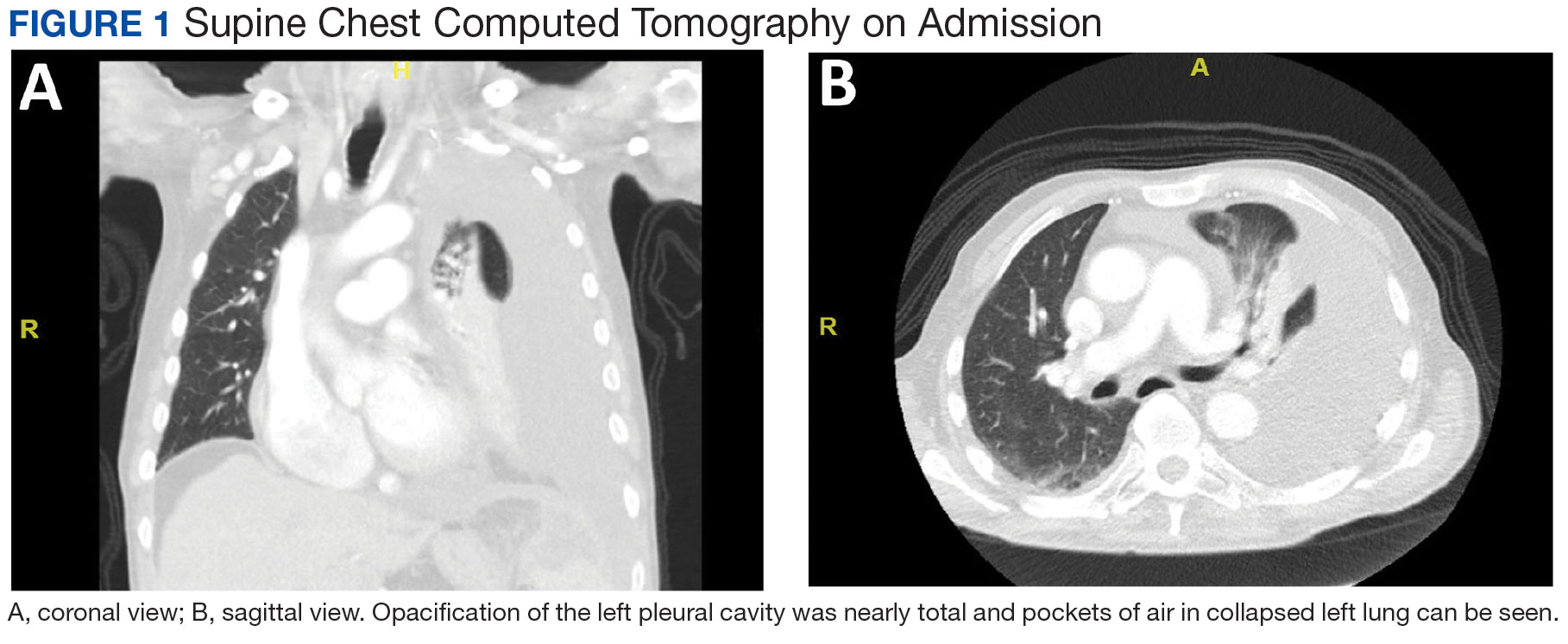
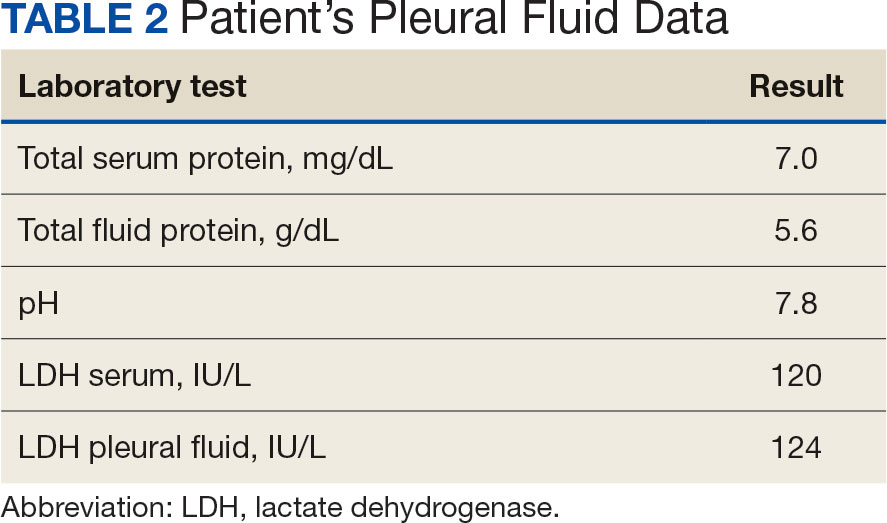
Initial thoracentesis was performed with 1500 mL of straw-colored fluid negative for bacteria, fungi, malignancy, and acid-fast organisms (Tables 2 and 3). The pleural effusion persisted, requiring a second thoracentesis 2 days later that was positive for Escherichia coli (E coli). Given the exudative nature and positive culture, a chest tube was placed, and the pleural effusion was therefore felt to be an empyema, arousing suspicion that the splenic mass seen on CT was an abscess. The site was accessed by interventional radiology, purulent fluid aspirated, and a drain was placed. Cultures grew E coli sensitive to ceftriaxone. Despite receiving intravenous ceftriaxone 2 g daily, the pleural effusion became further complicated due to chest tube obstruction and persistent drainage.
The patient was discharged to Baylor Scott & White Medical Center in Temple, Texas where he underwent decortication with cardiothoracic surgery with several pleural adhesions noted. Following surgery the patient was readmitted to CTVAH and continued ceftriaxone therapy following the infectious disease specialist's recommendation. He was discharged with plans to return to CTVAH for continued care. The patient was readmitted and transitioned to oral levofloxacin 500 mg daily and received physical and occupational therapy. He showed dramatic improvement on this regimen, with a 3-week follow-up CT that indicated only a small left pleural effusion and a 28 mm × 11 mm × 10 mm lesion in the anterior superior spleen. The patient had not returned for further evaluation by thoracic surgery; however, he has continued to see CTVAH primary care without reported recurrence of symptoms.
DISCUSSION
Splenic abscesses are a rare condition typically characterized by hematogenous spread of bacteria from another source, most commonly the endocardium.2 Other differential diagnoses include bacteremia or spread from an intra-abdominal site.2 Staphylococcus aureus and E coli are the most common bacteria seen in splenic abscesses. 2 Treatment includes antibiotics, percutaneous drainage, and, as a last resort, splenectomy.2
Our patient was found to have grown E coli, but no source indicative of spread was identified. He had negative blood cultures, negative findings for intra-abdominal pathologies on CT scans, and a negative echocardiogram for endocarditis. A bronchoscopy showed no evidence of a source from the lungs, and specimens taken from the pleural adhesions were negative for malignancy and bacteria.
This patient had risk factors for the illness, namely his history of being immunocompromised secondary to myelodysplastic syndrome.7 Accordingly, the patient showed persistent leukopenia with neutropenia and lymphocytopenia, which would not be expected for most patients with such an extensive infection. 8 While being immunocompromised undoubtedly contributed to the severity of the patient’s presentation and slow recovery, it does not explain the etiology or origin of his infection. This patient differs from current literature in that his splenic abscess was truly idiopathic rather than resulting from an alternative source.
Complications of splenic abscesses include pleural effusions, as seen with this patient, as well as pneumonia, pneumothorax, hemorrhage, subphrenic abscess, and intraabdominal perforation, among others.2 We determined conclusively that the patient’s pleural effusion was secondary to the splenic abscess, and excluded other bacterial foci strongly suggests that the spleen was the origin of the illness.
CONCLUSIONS
This case suggests splenic abscesses should be considered when evaluating pleural effusion. It further demonstrates that the spleen may be the central source of infection in the absence of iatrogenic inoculation or bacteremia. We hope our findings may lead to earlier identification in similar scenarios and improved patient outcomes in a multidisciplinary approach.
- Lee WS, Choi ST, Kim KK. Splenic abscess: a single institution study and review of the literature. Yonsei Med J. 2011;52(2):288-292. doi:10.3349/ymj.2011.52.2.288
- Lotfollahzadeh S, Mathew G, Zemaitis MR. Splenic Abscess. In: StatPearls. StatPearls Publishing; June 3, 2023.
- Jany B, Welte T. Pleural effusion in adults-etiology, diagnosis, and treatment. Dtsch Arztebl Int. 2019;116(21):377- 386. doi:10.3238/arztebl.2019.0377
- Light RW. Pleural effusions. Med Clin North Am. 2011;95(6):1055-1070. doi:10.1016/j.mcna.2011.08.005
- Feller-Kopman D, Light R. Pleural Disease. N Engl J Med. 2018;378(18):1754. doi:10.1056/NEJMc1803858
- Ferreiro L, Casal A, Toubes ME, et al. Pleural effusion due to nonmalignant gastrointestinal disease. ERJ Open Res. 2023;9(3):00290-2022. doi:10.1183/23120541.00290-2022
- Hasserjian RP. Myelodysplastic syndrome updated. Pathobiology. 2019;86(1):7-13. doi:10.1159/000489702
- Toma A, Fenaux P, Dreyfus F, Cordonnier C. Infections in myelodysplastic syndromes. Haematologica. 2012;97(10):1459- 1470. doi:10.3324/haematol2012.063420
Splenic abscesses are a rare occurrence that represent a marginal proportion of intra-abdominal infections. One study found splenic abscesses in only 0.14% to 0.70% of autopsies and none of the 540 abdominal abscesses they examined originated in the spleen.1 Patients with splenic abscesses tend to present with nonspecific symptoms such as fevers, chills, and abdominal pain.2 Imaging modalities such as abdominal ultrasound and computed tomography (CT) are vital to the workup and diagnosis identification.2 Splenic abscesses are generally associated with another underlying process, as seen in patients who are affected by endocarditis, trauma, metastatic infection, splenic infarction, or neoplasia.2
Pleural effusions, or the buildup of fluid within the pleural space, is a common condition typically secondary to another disease.3 Clinical identification of the primary condition may be challenging.3 In the absence of a clear etiology, such as obvious signs of congestive heart failure, further differentiation relies upon pleural fluid analysis, beginning with the distinction between exudate (inflammatory) and transudate (noninflammatory). 3,4 This distinction can be made using Light’s criteria, which relies on protein and lactate dehydrogenase (LDH) ratios between the pleural fluid and serum (Table 1).5 Though rare, half of splenic abscesses are associated with pleural effusion.6 As an inflammatory condition, splenic abscesses have been classically described as a cause of exudative pleural effusions.5,6

A myelodysplastic syndrome is a group of diseases that arise from malignant hematopoietic stem cells, leading to the proliferation of the malignant cells and faulty production of other bone marrow products.7 These disorders can range from single to multilineage dysplasia. Cells are often left in an immature blast form, unable to function appropriately, and vulnerable to destruction. Patients with myeloproliferative disorders frequently suffer from leukopenia and infections attributable to known quantitative and qualitative defects of neutrophils.8
CASE PRESENTATION
A male aged 80 years presented to the Central Texas Veterans Affairs Hospital (CTVAH) with shortness of breath, weight loss, and fever. On admission, his medical history was notable for atrial fibrillation, myelodysplastic syndrome, hypertension, hyperlipidemia, stable ascending aortic aneurysm, and Vitamin B12 deficiency. A chest CT showed a large left pleural effusion (Figure 1). Additionally, the radiology report noted a nonspecific 4- to 5-cm lobulated subdiaphragmatic mass within the anterior dome of the spleen with surrounding soft tissue swelling and splenomegaly (Figure 2).


Initial thoracentesis was performed with 1500 mL of straw-colored fluid negative for bacteria, fungi, malignancy, and acid-fast organisms (Tables 2 and 3). The pleural effusion persisted, requiring a second thoracentesis 2 days later that was positive for Escherichia coli (E coli). Given the exudative nature and positive culture, a chest tube was placed, and the pleural effusion was therefore felt to be an empyema, arousing suspicion that the splenic mass seen on CT was an abscess. The site was accessed by interventional radiology, purulent fluid aspirated, and a drain was placed. Cultures grew E coli sensitive to ceftriaxone. Despite receiving intravenous ceftriaxone 2 g daily, the pleural effusion became further complicated due to chest tube obstruction and persistent drainage.


The patient was discharged to Baylor Scott & White Medical Center in Temple, Texas where he underwent decortication with cardiothoracic surgery with several pleural adhesions noted. Following surgery the patient was readmitted to CTVAH and continued ceftriaxone therapy following the infectious disease specialist's recommendation. He was discharged with plans to return to CTVAH for continued care. The patient was readmitted and transitioned to oral levofloxacin 500 mg daily and received physical and occupational therapy. He showed dramatic improvement on this regimen, with a 3-week follow-up CT that indicated only a small left pleural effusion and a 28 mm × 11 mm × 10 mm lesion in the anterior superior spleen. The patient had not returned for further evaluation by thoracic surgery; however, he has continued to see CTVAH primary care without reported recurrence of symptoms.
DISCUSSION
Splenic abscesses are a rare condition typically characterized by hematogenous spread of bacteria from another source, most commonly the endocardium.2 Other differential diagnoses include bacteremia or spread from an intra-abdominal site.2 Staphylococcus aureus and E coli are the most common bacteria seen in splenic abscesses. 2 Treatment includes antibiotics, percutaneous drainage, and, as a last resort, splenectomy.2
Our patient was found to have grown E coli, but no source indicative of spread was identified. He had negative blood cultures, negative findings for intra-abdominal pathologies on CT scans, and a negative echocardiogram for endocarditis. A bronchoscopy showed no evidence of a source from the lungs, and specimens taken from the pleural adhesions were negative for malignancy and bacteria.
This patient had risk factors for the illness, namely his history of being immunocompromised secondary to myelodysplastic syndrome.7 Accordingly, the patient showed persistent leukopenia with neutropenia and lymphocytopenia, which would not be expected for most patients with such an extensive infection. 8 While being immunocompromised undoubtedly contributed to the severity of the patient’s presentation and slow recovery, it does not explain the etiology or origin of his infection. This patient differs from current literature in that his splenic abscess was truly idiopathic rather than resulting from an alternative source.
Complications of splenic abscesses include pleural effusions, as seen with this patient, as well as pneumonia, pneumothorax, hemorrhage, subphrenic abscess, and intraabdominal perforation, among others.2 We determined conclusively that the patient’s pleural effusion was secondary to the splenic abscess, and excluded other bacterial foci strongly suggests that the spleen was the origin of the illness.
CONCLUSIONS
This case suggests splenic abscesses should be considered when evaluating pleural effusion. It further demonstrates that the spleen may be the central source of infection in the absence of iatrogenic inoculation or bacteremia. We hope our findings may lead to earlier identification in similar scenarios and improved patient outcomes in a multidisciplinary approach.
Splenic abscesses are a rare occurrence that represent a marginal proportion of intra-abdominal infections. One study found splenic abscesses in only 0.14% to 0.70% of autopsies and none of the 540 abdominal abscesses they examined originated in the spleen.1 Patients with splenic abscesses tend to present with nonspecific symptoms such as fevers, chills, and abdominal pain.2 Imaging modalities such as abdominal ultrasound and computed tomography (CT) are vital to the workup and diagnosis identification.2 Splenic abscesses are generally associated with another underlying process, as seen in patients who are affected by endocarditis, trauma, metastatic infection, splenic infarction, or neoplasia.2
Pleural effusions, or the buildup of fluid within the pleural space, is a common condition typically secondary to another disease.3 Clinical identification of the primary condition may be challenging.3 In the absence of a clear etiology, such as obvious signs of congestive heart failure, further differentiation relies upon pleural fluid analysis, beginning with the distinction between exudate (inflammatory) and transudate (noninflammatory). 3,4 This distinction can be made using Light’s criteria, which relies on protein and lactate dehydrogenase (LDH) ratios between the pleural fluid and serum (Table 1).5 Though rare, half of splenic abscesses are associated with pleural effusion.6 As an inflammatory condition, splenic abscesses have been classically described as a cause of exudative pleural effusions.5,6

A myelodysplastic syndrome is a group of diseases that arise from malignant hematopoietic stem cells, leading to the proliferation of the malignant cells and faulty production of other bone marrow products.7 These disorders can range from single to multilineage dysplasia. Cells are often left in an immature blast form, unable to function appropriately, and vulnerable to destruction. Patients with myeloproliferative disorders frequently suffer from leukopenia and infections attributable to known quantitative and qualitative defects of neutrophils.8
CASE PRESENTATION
A male aged 80 years presented to the Central Texas Veterans Affairs Hospital (CTVAH) with shortness of breath, weight loss, and fever. On admission, his medical history was notable for atrial fibrillation, myelodysplastic syndrome, hypertension, hyperlipidemia, stable ascending aortic aneurysm, and Vitamin B12 deficiency. A chest CT showed a large left pleural effusion (Figure 1). Additionally, the radiology report noted a nonspecific 4- to 5-cm lobulated subdiaphragmatic mass within the anterior dome of the spleen with surrounding soft tissue swelling and splenomegaly (Figure 2).


Initial thoracentesis was performed with 1500 mL of straw-colored fluid negative for bacteria, fungi, malignancy, and acid-fast organisms (Tables 2 and 3). The pleural effusion persisted, requiring a second thoracentesis 2 days later that was positive for Escherichia coli (E coli). Given the exudative nature and positive culture, a chest tube was placed, and the pleural effusion was therefore felt to be an empyema, arousing suspicion that the splenic mass seen on CT was an abscess. The site was accessed by interventional radiology, purulent fluid aspirated, and a drain was placed. Cultures grew E coli sensitive to ceftriaxone. Despite receiving intravenous ceftriaxone 2 g daily, the pleural effusion became further complicated due to chest tube obstruction and persistent drainage.


The patient was discharged to Baylor Scott & White Medical Center in Temple, Texas where he underwent decortication with cardiothoracic surgery with several pleural adhesions noted. Following surgery the patient was readmitted to CTVAH and continued ceftriaxone therapy following the infectious disease specialist's recommendation. He was discharged with plans to return to CTVAH for continued care. The patient was readmitted and transitioned to oral levofloxacin 500 mg daily and received physical and occupational therapy. He showed dramatic improvement on this regimen, with a 3-week follow-up CT that indicated only a small left pleural effusion and a 28 mm × 11 mm × 10 mm lesion in the anterior superior spleen. The patient had not returned for further evaluation by thoracic surgery; however, he has continued to see CTVAH primary care without reported recurrence of symptoms.
DISCUSSION
Splenic abscesses are a rare condition typically characterized by hematogenous spread of bacteria from another source, most commonly the endocardium.2 Other differential diagnoses include bacteremia or spread from an intra-abdominal site.2 Staphylococcus aureus and E coli are the most common bacteria seen in splenic abscesses. 2 Treatment includes antibiotics, percutaneous drainage, and, as a last resort, splenectomy.2
Our patient was found to have grown E coli, but no source indicative of spread was identified. He had negative blood cultures, negative findings for intra-abdominal pathologies on CT scans, and a negative echocardiogram for endocarditis. A bronchoscopy showed no evidence of a source from the lungs, and specimens taken from the pleural adhesions were negative for malignancy and bacteria.
This patient had risk factors for the illness, namely his history of being immunocompromised secondary to myelodysplastic syndrome.7 Accordingly, the patient showed persistent leukopenia with neutropenia and lymphocytopenia, which would not be expected for most patients with such an extensive infection. 8 While being immunocompromised undoubtedly contributed to the severity of the patient’s presentation and slow recovery, it does not explain the etiology or origin of his infection. This patient differs from current literature in that his splenic abscess was truly idiopathic rather than resulting from an alternative source.
Complications of splenic abscesses include pleural effusions, as seen with this patient, as well as pneumonia, pneumothorax, hemorrhage, subphrenic abscess, and intraabdominal perforation, among others.2 We determined conclusively that the patient’s pleural effusion was secondary to the splenic abscess, and excluded other bacterial foci strongly suggests that the spleen was the origin of the illness.
CONCLUSIONS
This case suggests splenic abscesses should be considered when evaluating pleural effusion. It further demonstrates that the spleen may be the central source of infection in the absence of iatrogenic inoculation or bacteremia. We hope our findings may lead to earlier identification in similar scenarios and improved patient outcomes in a multidisciplinary approach.
- Lee WS, Choi ST, Kim KK. Splenic abscess: a single institution study and review of the literature. Yonsei Med J. 2011;52(2):288-292. doi:10.3349/ymj.2011.52.2.288
- Lotfollahzadeh S, Mathew G, Zemaitis MR. Splenic Abscess. In: StatPearls. StatPearls Publishing; June 3, 2023.
- Jany B, Welte T. Pleural effusion in adults-etiology, diagnosis, and treatment. Dtsch Arztebl Int. 2019;116(21):377- 386. doi:10.3238/arztebl.2019.0377
- Light RW. Pleural effusions. Med Clin North Am. 2011;95(6):1055-1070. doi:10.1016/j.mcna.2011.08.005
- Feller-Kopman D, Light R. Pleural Disease. N Engl J Med. 2018;378(18):1754. doi:10.1056/NEJMc1803858
- Ferreiro L, Casal A, Toubes ME, et al. Pleural effusion due to nonmalignant gastrointestinal disease. ERJ Open Res. 2023;9(3):00290-2022. doi:10.1183/23120541.00290-2022
- Hasserjian RP. Myelodysplastic syndrome updated. Pathobiology. 2019;86(1):7-13. doi:10.1159/000489702
- Toma A, Fenaux P, Dreyfus F, Cordonnier C. Infections in myelodysplastic syndromes. Haematologica. 2012;97(10):1459- 1470. doi:10.3324/haematol2012.063420
- Lee WS, Choi ST, Kim KK. Splenic abscess: a single institution study and review of the literature. Yonsei Med J. 2011;52(2):288-292. doi:10.3349/ymj.2011.52.2.288
- Lotfollahzadeh S, Mathew G, Zemaitis MR. Splenic Abscess. In: StatPearls. StatPearls Publishing; June 3, 2023.
- Jany B, Welte T. Pleural effusion in adults-etiology, diagnosis, and treatment. Dtsch Arztebl Int. 2019;116(21):377- 386. doi:10.3238/arztebl.2019.0377
- Light RW. Pleural effusions. Med Clin North Am. 2011;95(6):1055-1070. doi:10.1016/j.mcna.2011.08.005
- Feller-Kopman D, Light R. Pleural Disease. N Engl J Med. 2018;378(18):1754. doi:10.1056/NEJMc1803858
- Ferreiro L, Casal A, Toubes ME, et al. Pleural effusion due to nonmalignant gastrointestinal disease. ERJ Open Res. 2023;9(3):00290-2022. doi:10.1183/23120541.00290-2022
- Hasserjian RP. Myelodysplastic syndrome updated. Pathobiology. 2019;86(1):7-13. doi:10.1159/000489702
- Toma A, Fenaux P, Dreyfus F, Cordonnier C. Infections in myelodysplastic syndromes. Haematologica. 2012;97(10):1459- 1470. doi:10.3324/haematol2012.063420
Myth of the Month: Vitamin C vs the Common Cold
Case: A 38-year-old presents for acute onset runny nose, cough, and fever for the last 3 days. Her children at home have a similar presentation. She believes that she has been managing her symptoms well with Tylenol and rest. The patient is up to date on her COVID and flu shots and was wondering if there was anything else she could have done to prevent her symptoms. She saw a commercial about vitamin C supplements boosting the immune system and was wondering about their efficacy. How would you respond?
Studies of Vitamin C
Linus Pauling, FRS, did a summary of four relatively small published studies of vitamin C and concluded that vitamin C supplementation helped prevent and lessen colds.1 He mentioned a placebo-controlled study of vitamin C with viral inoculation which did not show any effect. His overall conclusion of efficacy for vitamin C led to the widespread belief that vitamin C was a proven effective therapy to prevent and treat the common cold. Since then, multiple trials and studies have examined the effect of vitamin C on the prevention and treatment of colds.
The Cochrane Review conducted a meta-analysis comparing 29 placebo-controlled trials involving 11,306 participants.2 Criteria included vitamin C supplementation of 0.2 g-1 g/day to study its efficacy in preventing the common cold. The analysis showed that supplemental vitamin C did not significantly reduce the incidence of colds. However, there was a statistically significant 8% reduction in adults and 14% in children in the duration of colds. In terms of treatment, there was no evidence of vitamin C’s efficacy.
A 2001 study conducted a small double-blind, randomized control trial to evaluate large doses of vitamin C as treatment for the common cold.3 Volunteers were divided and instructed to take varying doses ranging from 1 to 3 g of vitamin C vs a placebo at the onset of cold-like symptoms. Subjects were expected to assess the duration and severity of their cold. The data showed no significant difference in the severity or duration of cold symptoms between small or large vitamin C doses or placebo.
A more recent meta-analysis by Hemilä and Chalker looked at 10 placebo-controlled trials of vitamin C for the prevention and treatment of colds.4 The analysis showed a small 15% reduction in more severe cold symptoms.
Summary
While vitamin C is safe, there is no evidence for its ability to prevent the common cold. Although the Cochrane review and more a recent meta-analysis by Hemilä and Chalker demonstrated statistical significance in shortening the duration of symptoms, it was a minimal reduction with little clinical significance. .
Ms. Ibabao is a fourth year medical student at the University of Washington School of Medicine; Dr. Paauw is Professor of Medicine, Rathmann Family Foundation Endowed Chair Patient-centered Clinical Education, at the University of Washington School of Medicine, Seattle. They have no conflicts of interest.
References
1. Pauling L. The significance of the evidence about ascorbic acid and the common cold. Proc Natl Acad Sci USA. 1971;68:2678-2671.
2. Hemilä H, Chalker E. Vitamin C for preventing and treating the common cold. Cochrane Database of Systematic Reviews. 2013;1(1).
3. Audera C et al. Mega‐dose vitamin C in treatment of the common cold: a randomised controlled trial. Med J Australia. 2001;175(7):359-362.
4. Hemilä H, Chalker E. Vitamin C reduces the severity of common colds: a meta-analysis. BMC Public Health. 2023;23:2468.
Case: A 38-year-old presents for acute onset runny nose, cough, and fever for the last 3 days. Her children at home have a similar presentation. She believes that she has been managing her symptoms well with Tylenol and rest. The patient is up to date on her COVID and flu shots and was wondering if there was anything else she could have done to prevent her symptoms. She saw a commercial about vitamin C supplements boosting the immune system and was wondering about their efficacy. How would you respond?
Studies of Vitamin C
Linus Pauling, FRS, did a summary of four relatively small published studies of vitamin C and concluded that vitamin C supplementation helped prevent and lessen colds.1 He mentioned a placebo-controlled study of vitamin C with viral inoculation which did not show any effect. His overall conclusion of efficacy for vitamin C led to the widespread belief that vitamin C was a proven effective therapy to prevent and treat the common cold. Since then, multiple trials and studies have examined the effect of vitamin C on the prevention and treatment of colds.
The Cochrane Review conducted a meta-analysis comparing 29 placebo-controlled trials involving 11,306 participants.2 Criteria included vitamin C supplementation of 0.2 g-1 g/day to study its efficacy in preventing the common cold. The analysis showed that supplemental vitamin C did not significantly reduce the incidence of colds. However, there was a statistically significant 8% reduction in adults and 14% in children in the duration of colds. In terms of treatment, there was no evidence of vitamin C’s efficacy.
A 2001 study conducted a small double-blind, randomized control trial to evaluate large doses of vitamin C as treatment for the common cold.3 Volunteers were divided and instructed to take varying doses ranging from 1 to 3 g of vitamin C vs a placebo at the onset of cold-like symptoms. Subjects were expected to assess the duration and severity of their cold. The data showed no significant difference in the severity or duration of cold symptoms between small or large vitamin C doses or placebo.
A more recent meta-analysis by Hemilä and Chalker looked at 10 placebo-controlled trials of vitamin C for the prevention and treatment of colds.4 The analysis showed a small 15% reduction in more severe cold symptoms.
Summary
While vitamin C is safe, there is no evidence for its ability to prevent the common cold. Although the Cochrane review and more a recent meta-analysis by Hemilä and Chalker demonstrated statistical significance in shortening the duration of symptoms, it was a minimal reduction with little clinical significance. .
Ms. Ibabao is a fourth year medical student at the University of Washington School of Medicine; Dr. Paauw is Professor of Medicine, Rathmann Family Foundation Endowed Chair Patient-centered Clinical Education, at the University of Washington School of Medicine, Seattle. They have no conflicts of interest.
References
1. Pauling L. The significance of the evidence about ascorbic acid and the common cold. Proc Natl Acad Sci USA. 1971;68:2678-2671.
2. Hemilä H, Chalker E. Vitamin C for preventing and treating the common cold. Cochrane Database of Systematic Reviews. 2013;1(1).
3. Audera C et al. Mega‐dose vitamin C in treatment of the common cold: a randomised controlled trial. Med J Australia. 2001;175(7):359-362.
4. Hemilä H, Chalker E. Vitamin C reduces the severity of common colds: a meta-analysis. BMC Public Health. 2023;23:2468.
Case: A 38-year-old presents for acute onset runny nose, cough, and fever for the last 3 days. Her children at home have a similar presentation. She believes that she has been managing her symptoms well with Tylenol and rest. The patient is up to date on her COVID and flu shots and was wondering if there was anything else she could have done to prevent her symptoms. She saw a commercial about vitamin C supplements boosting the immune system and was wondering about their efficacy. How would you respond?
Studies of Vitamin C
Linus Pauling, FRS, did a summary of four relatively small published studies of vitamin C and concluded that vitamin C supplementation helped prevent and lessen colds.1 He mentioned a placebo-controlled study of vitamin C with viral inoculation which did not show any effect. His overall conclusion of efficacy for vitamin C led to the widespread belief that vitamin C was a proven effective therapy to prevent and treat the common cold. Since then, multiple trials and studies have examined the effect of vitamin C on the prevention and treatment of colds.
The Cochrane Review conducted a meta-analysis comparing 29 placebo-controlled trials involving 11,306 participants.2 Criteria included vitamin C supplementation of 0.2 g-1 g/day to study its efficacy in preventing the common cold. The analysis showed that supplemental vitamin C did not significantly reduce the incidence of colds. However, there was a statistically significant 8% reduction in adults and 14% in children in the duration of colds. In terms of treatment, there was no evidence of vitamin C’s efficacy.
A 2001 study conducted a small double-blind, randomized control trial to evaluate large doses of vitamin C as treatment for the common cold.3 Volunteers were divided and instructed to take varying doses ranging from 1 to 3 g of vitamin C vs a placebo at the onset of cold-like symptoms. Subjects were expected to assess the duration and severity of their cold. The data showed no significant difference in the severity or duration of cold symptoms between small or large vitamin C doses or placebo.
A more recent meta-analysis by Hemilä and Chalker looked at 10 placebo-controlled trials of vitamin C for the prevention and treatment of colds.4 The analysis showed a small 15% reduction in more severe cold symptoms.
Summary
While vitamin C is safe, there is no evidence for its ability to prevent the common cold. Although the Cochrane review and more a recent meta-analysis by Hemilä and Chalker demonstrated statistical significance in shortening the duration of symptoms, it was a minimal reduction with little clinical significance. .
Ms. Ibabao is a fourth year medical student at the University of Washington School of Medicine; Dr. Paauw is Professor of Medicine, Rathmann Family Foundation Endowed Chair Patient-centered Clinical Education, at the University of Washington School of Medicine, Seattle. They have no conflicts of interest.
References
1. Pauling L. The significance of the evidence about ascorbic acid and the common cold. Proc Natl Acad Sci USA. 1971;68:2678-2671.
2. Hemilä H, Chalker E. Vitamin C for preventing and treating the common cold. Cochrane Database of Systematic Reviews. 2013;1(1).
3. Audera C et al. Mega‐dose vitamin C in treatment of the common cold: a randomised controlled trial. Med J Australia. 2001;175(7):359-362.
4. Hemilä H, Chalker E. Vitamin C reduces the severity of common colds: a meta-analysis. BMC Public Health. 2023;23:2468.
Whooping Cough Rising Fast, Especially Among Teens
Whooping cough is surging in the United States, with four times as many cases reported so far this year, compared to all of 2023.
The CDC said 14,569 cases had been reported as of Sept. 14, compared to 3475 in all of 2023.
Whooping cough, also called pertussis, is a respiratory illness spread through coughing, sneezing, or breathing very close to another person. Babies are given the DTaP vaccine to protect against whooping cough, diphtheria, and tetanus. Because the vaccine effectiveness wanes faster for whooping cough than the two other illnesses, boosters are recommended every decade or so.
Why the Whooping Cough Vaccine Is Important
Whooping cough is a very contagious bacteria, so vaccination is an important step to avoid it.
But many children in their tweens aren’t getting boosters, and that age group is driving the whooping cough outbreak.
“With the increase in vaccine hesitancy that has been going on since the COVID-19 pandemic, we’re seeing outbreaks occurring in kids who are not vaccinated,” Tina Tan, MD, president-elect of the Infectious Diseases Society of America, told NBC News.
Also, people are not social distancing the way they did during the height of the COVID pandemic, when whooping cough numbers went down.
“Levels of pertussis dropped dramatically when we were all masking, and now this huge increase is getting us back to pre-pandemic levels, and probably a little above that,” Thomas Murray, MD, a Yale Medicine pediatric infectious diseases specialist, said in a news release from the school. “It’s a contagious respiratory virus that can spread fairly quickly through the population.”
FDA advisers were scheduled to meet Sept. 20 to discuss developing more effective boosters for whooping cough.
A version of this article appeared on WebMD.com.
Whooping cough is surging in the United States, with four times as many cases reported so far this year, compared to all of 2023.
The CDC said 14,569 cases had been reported as of Sept. 14, compared to 3475 in all of 2023.
Whooping cough, also called pertussis, is a respiratory illness spread through coughing, sneezing, or breathing very close to another person. Babies are given the DTaP vaccine to protect against whooping cough, diphtheria, and tetanus. Because the vaccine effectiveness wanes faster for whooping cough than the two other illnesses, boosters are recommended every decade or so.
Why the Whooping Cough Vaccine Is Important
Whooping cough is a very contagious bacteria, so vaccination is an important step to avoid it.
But many children in their tweens aren’t getting boosters, and that age group is driving the whooping cough outbreak.
“With the increase in vaccine hesitancy that has been going on since the COVID-19 pandemic, we’re seeing outbreaks occurring in kids who are not vaccinated,” Tina Tan, MD, president-elect of the Infectious Diseases Society of America, told NBC News.
Also, people are not social distancing the way they did during the height of the COVID pandemic, when whooping cough numbers went down.
“Levels of pertussis dropped dramatically when we were all masking, and now this huge increase is getting us back to pre-pandemic levels, and probably a little above that,” Thomas Murray, MD, a Yale Medicine pediatric infectious diseases specialist, said in a news release from the school. “It’s a contagious respiratory virus that can spread fairly quickly through the population.”
FDA advisers were scheduled to meet Sept. 20 to discuss developing more effective boosters for whooping cough.
A version of this article appeared on WebMD.com.
Whooping cough is surging in the United States, with four times as many cases reported so far this year, compared to all of 2023.
The CDC said 14,569 cases had been reported as of Sept. 14, compared to 3475 in all of 2023.
Whooping cough, also called pertussis, is a respiratory illness spread through coughing, sneezing, or breathing very close to another person. Babies are given the DTaP vaccine to protect against whooping cough, diphtheria, and tetanus. Because the vaccine effectiveness wanes faster for whooping cough than the two other illnesses, boosters are recommended every decade or so.
Why the Whooping Cough Vaccine Is Important
Whooping cough is a very contagious bacteria, so vaccination is an important step to avoid it.
But many children in their tweens aren’t getting boosters, and that age group is driving the whooping cough outbreak.
“With the increase in vaccine hesitancy that has been going on since the COVID-19 pandemic, we’re seeing outbreaks occurring in kids who are not vaccinated,” Tina Tan, MD, president-elect of the Infectious Diseases Society of America, told NBC News.
Also, people are not social distancing the way they did during the height of the COVID pandemic, when whooping cough numbers went down.
“Levels of pertussis dropped dramatically when we were all masking, and now this huge increase is getting us back to pre-pandemic levels, and probably a little above that,” Thomas Murray, MD, a Yale Medicine pediatric infectious diseases specialist, said in a news release from the school. “It’s a contagious respiratory virus that can spread fairly quickly through the population.”
FDA advisers were scheduled to meet Sept. 20 to discuss developing more effective boosters for whooping cough.
A version of this article appeared on WebMD.com.
Identifying Drug-Induced Rashes in Skin of Color: Heightened Awareness Can Accelerate Diagnosis
NEW YORK — Because of their heterogeneity in appearance, to speed the diagnosis.
This risk for a delayed or missed diagnosis in patients with darker skin is shared across skin rashes, but drug-induced hypersensitivity syndrome (DIHS) is a telling example, according to Joanna Harp, MD, director of the Inpatient Dermatology Consult Service, NewYork–Presbyterian Hospital, New York City.
DIHS, also known as a drug reaction with eosinophilia and systemic symptoms, is a type IV hypersensitivity reaction, Dr. Harp explained. While the fact that this disorder does not always include eosinophilia prompted the DIHS acronym, the maculopapular rash often serves as a critical clue of the underlying etiology.
In patients with darker skin, DIHS skin manifestations “can look different, can be more severe, and can have worse outcomes,” Dr. Harp said. As with other skin rashes that are primarily erythematous, the DIHS rash is often more subtle in Black-skinned patients, typically appearing gray or violaceous rather than red.
“The high amount of scale can be a clue,” said Dr. Harp, speaking at the 2024 Skin of Color Update. Scale is particularly prominent among Black patients, she said, because of the greater relative transepidermal water loss than lighter skin, increasing dryness and susceptibility to scale.
The maculopapular rash is “similar to a simple drug eruption, although it is usually more impressive,” she said. Emphasizing that DIHS is a systemic disease, she noted that the characteristic rash is typically accompanied by inflammation in multiple organs that not only includes the mucous membranes but can include major organs such as the lungs, kidneys, and heart.
In patients with DIHS and many of the even more serious types of rashes traced to drug exposures, such as Stevens-Johnson syndrome (SJS) or erythema multiforme, the delay to appearance of the rash from the time of exposure can be the most confusing element.
“It can be months for some drugs such as allopurinol,” said Dr. Harp, pointing out that Black and Asian patients are more likely to carry the HLA-B*5801 genotype, a known risk factor for allopurinol hypersensitivity.
Signs of AGEP Can Be Subtle in Black Patients
Some of the same principles for diagnosing drug-induced rash in darker skin can also be applied to acute generalized exanthematous pustulosis (AGEP), another type IV hypersensitivity reaction. Like all drug-induced rashes, the earlier AGEP is recognized and treated, the better the outcome, but in Black patients, the signs can be subtle.
“The onset is usually fast and occurs in 1-2 days after [the causative drug] exposure,” said Dr. Harp, adding that antibiotics, such as cephalosporins or penicillin, and calcium channel blockers are among the prominent causes of AGEP.
One of the hallmark signs of early-onset AGEP are tiny erythematous pustules in flexural areas, such as the neck or the armpits. The issue of detecting erythema in darker skin is also relevant to this area, but there is an additional problem, according to Dr. Harp. The pustules often dry up quickly, leaving a neutrophilic scale that further complicates the effort to see the characteristic erythema.
“If you see a lot of scale, look for erythema underneath. Think of inflammation,” Dr. Harp said, explaining that the clinical appearance evolves quickly. “If you do not see the pustules, it does not mean they were not there; you just missed them.”
In addition to the flexural areas, “AGEP loves the ears, the face, and the geographic tongue,” she said, offering several pearls to help with the diagnosis. These include side lighting to make papules easier to see, pressing on the skin to highlight the difference between erythematous skin and blanched skin, and checking less pigmented skin, such as on the hands and feet, which makes erythema easier to see.
Steroids are often the first-line treatment for drug-induced skin rashes, but Dr. Harp moves to etanercept or cyclosporine for the most serious drug reactions, such as SJS and toxic epidermal necrolysis.
Etanercept is typically her first choice because patients with systemic hypersensitivity reactions with major organ involvement are often quite ill, making cyclosporine harder to use. In her experience, etanercept has been well tolerated.
Conversely, she cautioned against the use of intravenous immunoglobulin (IVIG). Although this has been used traditionally for severe drug hypersensitivity reactions, “the data are not there,” she said. The data are stronger for a combination of high-dose steroids and IVIG, but she thinks even these data are inconsistent and not as strong as the data supporting etanercept or cyclosporine. She encouraged centers still using IVIG to consider alternatives.
After drug sensitivity reactions are controlled, follow-up care is particularly important for Black patients who face greater risks for sequelae, such as hypopigmentation, hyperpigmentation, or keloids. She recommended aggressive use of emollients and sunscreens for an extended period after lesions resolve to lessen these risks.
Differences in the manifestations of drug-induced skin rashes by race and ethnicity are important and perhaps underappreciated, agreed Shawn Kwatra, MD, professor and chairman of the Department of Dermatology, University of Maryland, Baltimore.
Asked to comment at the meeting, Dr. Kwatra said that he appreciated Dr. Harp’s effort to translate published data and her experience into an overview that increases awareness of the risk for missed or delayed diagnoses of drug-induced rashes in skin of color. He noted that the strategies to identify erythema and pustules, such as increased suspicion in skin of color and the extra steps to rule them out, such as the use of side lighting in the case of pustules for AGEP, are simple and practical.
Dr. Harp and Dr. Kwatra had no relevant disclosures.
A version of this article appeared on Medscape.com.
NEW YORK — Because of their heterogeneity in appearance, to speed the diagnosis.
This risk for a delayed or missed diagnosis in patients with darker skin is shared across skin rashes, but drug-induced hypersensitivity syndrome (DIHS) is a telling example, according to Joanna Harp, MD, director of the Inpatient Dermatology Consult Service, NewYork–Presbyterian Hospital, New York City.
DIHS, also known as a drug reaction with eosinophilia and systemic symptoms, is a type IV hypersensitivity reaction, Dr. Harp explained. While the fact that this disorder does not always include eosinophilia prompted the DIHS acronym, the maculopapular rash often serves as a critical clue of the underlying etiology.
In patients with darker skin, DIHS skin manifestations “can look different, can be more severe, and can have worse outcomes,” Dr. Harp said. As with other skin rashes that are primarily erythematous, the DIHS rash is often more subtle in Black-skinned patients, typically appearing gray or violaceous rather than red.
“The high amount of scale can be a clue,” said Dr. Harp, speaking at the 2024 Skin of Color Update. Scale is particularly prominent among Black patients, she said, because of the greater relative transepidermal water loss than lighter skin, increasing dryness and susceptibility to scale.
The maculopapular rash is “similar to a simple drug eruption, although it is usually more impressive,” she said. Emphasizing that DIHS is a systemic disease, she noted that the characteristic rash is typically accompanied by inflammation in multiple organs that not only includes the mucous membranes but can include major organs such as the lungs, kidneys, and heart.
In patients with DIHS and many of the even more serious types of rashes traced to drug exposures, such as Stevens-Johnson syndrome (SJS) or erythema multiforme, the delay to appearance of the rash from the time of exposure can be the most confusing element.
“It can be months for some drugs such as allopurinol,” said Dr. Harp, pointing out that Black and Asian patients are more likely to carry the HLA-B*5801 genotype, a known risk factor for allopurinol hypersensitivity.
Signs of AGEP Can Be Subtle in Black Patients
Some of the same principles for diagnosing drug-induced rash in darker skin can also be applied to acute generalized exanthematous pustulosis (AGEP), another type IV hypersensitivity reaction. Like all drug-induced rashes, the earlier AGEP is recognized and treated, the better the outcome, but in Black patients, the signs can be subtle.
“The onset is usually fast and occurs in 1-2 days after [the causative drug] exposure,” said Dr. Harp, adding that antibiotics, such as cephalosporins or penicillin, and calcium channel blockers are among the prominent causes of AGEP.
One of the hallmark signs of early-onset AGEP are tiny erythematous pustules in flexural areas, such as the neck or the armpits. The issue of detecting erythema in darker skin is also relevant to this area, but there is an additional problem, according to Dr. Harp. The pustules often dry up quickly, leaving a neutrophilic scale that further complicates the effort to see the characteristic erythema.
“If you see a lot of scale, look for erythema underneath. Think of inflammation,” Dr. Harp said, explaining that the clinical appearance evolves quickly. “If you do not see the pustules, it does not mean they were not there; you just missed them.”
In addition to the flexural areas, “AGEP loves the ears, the face, and the geographic tongue,” she said, offering several pearls to help with the diagnosis. These include side lighting to make papules easier to see, pressing on the skin to highlight the difference between erythematous skin and blanched skin, and checking less pigmented skin, such as on the hands and feet, which makes erythema easier to see.
Steroids are often the first-line treatment for drug-induced skin rashes, but Dr. Harp moves to etanercept or cyclosporine for the most serious drug reactions, such as SJS and toxic epidermal necrolysis.
Etanercept is typically her first choice because patients with systemic hypersensitivity reactions with major organ involvement are often quite ill, making cyclosporine harder to use. In her experience, etanercept has been well tolerated.
Conversely, she cautioned against the use of intravenous immunoglobulin (IVIG). Although this has been used traditionally for severe drug hypersensitivity reactions, “the data are not there,” she said. The data are stronger for a combination of high-dose steroids and IVIG, but she thinks even these data are inconsistent and not as strong as the data supporting etanercept or cyclosporine. She encouraged centers still using IVIG to consider alternatives.
After drug sensitivity reactions are controlled, follow-up care is particularly important for Black patients who face greater risks for sequelae, such as hypopigmentation, hyperpigmentation, or keloids. She recommended aggressive use of emollients and sunscreens for an extended period after lesions resolve to lessen these risks.
Differences in the manifestations of drug-induced skin rashes by race and ethnicity are important and perhaps underappreciated, agreed Shawn Kwatra, MD, professor and chairman of the Department of Dermatology, University of Maryland, Baltimore.
Asked to comment at the meeting, Dr. Kwatra said that he appreciated Dr. Harp’s effort to translate published data and her experience into an overview that increases awareness of the risk for missed or delayed diagnoses of drug-induced rashes in skin of color. He noted that the strategies to identify erythema and pustules, such as increased suspicion in skin of color and the extra steps to rule them out, such as the use of side lighting in the case of pustules for AGEP, are simple and practical.
Dr. Harp and Dr. Kwatra had no relevant disclosures.
A version of this article appeared on Medscape.com.
NEW YORK — Because of their heterogeneity in appearance, to speed the diagnosis.
This risk for a delayed or missed diagnosis in patients with darker skin is shared across skin rashes, but drug-induced hypersensitivity syndrome (DIHS) is a telling example, according to Joanna Harp, MD, director of the Inpatient Dermatology Consult Service, NewYork–Presbyterian Hospital, New York City.
DIHS, also known as a drug reaction with eosinophilia and systemic symptoms, is a type IV hypersensitivity reaction, Dr. Harp explained. While the fact that this disorder does not always include eosinophilia prompted the DIHS acronym, the maculopapular rash often serves as a critical clue of the underlying etiology.
In patients with darker skin, DIHS skin manifestations “can look different, can be more severe, and can have worse outcomes,” Dr. Harp said. As with other skin rashes that are primarily erythematous, the DIHS rash is often more subtle in Black-skinned patients, typically appearing gray or violaceous rather than red.
“The high amount of scale can be a clue,” said Dr. Harp, speaking at the 2024 Skin of Color Update. Scale is particularly prominent among Black patients, she said, because of the greater relative transepidermal water loss than lighter skin, increasing dryness and susceptibility to scale.
The maculopapular rash is “similar to a simple drug eruption, although it is usually more impressive,” she said. Emphasizing that DIHS is a systemic disease, she noted that the characteristic rash is typically accompanied by inflammation in multiple organs that not only includes the mucous membranes but can include major organs such as the lungs, kidneys, and heart.
In patients with DIHS and many of the even more serious types of rashes traced to drug exposures, such as Stevens-Johnson syndrome (SJS) or erythema multiforme, the delay to appearance of the rash from the time of exposure can be the most confusing element.
“It can be months for some drugs such as allopurinol,” said Dr. Harp, pointing out that Black and Asian patients are more likely to carry the HLA-B*5801 genotype, a known risk factor for allopurinol hypersensitivity.
Signs of AGEP Can Be Subtle in Black Patients
Some of the same principles for diagnosing drug-induced rash in darker skin can also be applied to acute generalized exanthematous pustulosis (AGEP), another type IV hypersensitivity reaction. Like all drug-induced rashes, the earlier AGEP is recognized and treated, the better the outcome, but in Black patients, the signs can be subtle.
“The onset is usually fast and occurs in 1-2 days after [the causative drug] exposure,” said Dr. Harp, adding that antibiotics, such as cephalosporins or penicillin, and calcium channel blockers are among the prominent causes of AGEP.
One of the hallmark signs of early-onset AGEP are tiny erythematous pustules in flexural areas, such as the neck or the armpits. The issue of detecting erythema in darker skin is also relevant to this area, but there is an additional problem, according to Dr. Harp. The pustules often dry up quickly, leaving a neutrophilic scale that further complicates the effort to see the characteristic erythema.
“If you see a lot of scale, look for erythema underneath. Think of inflammation,” Dr. Harp said, explaining that the clinical appearance evolves quickly. “If you do not see the pustules, it does not mean they were not there; you just missed them.”
In addition to the flexural areas, “AGEP loves the ears, the face, and the geographic tongue,” she said, offering several pearls to help with the diagnosis. These include side lighting to make papules easier to see, pressing on the skin to highlight the difference between erythematous skin and blanched skin, and checking less pigmented skin, such as on the hands and feet, which makes erythema easier to see.
Steroids are often the first-line treatment for drug-induced skin rashes, but Dr. Harp moves to etanercept or cyclosporine for the most serious drug reactions, such as SJS and toxic epidermal necrolysis.
Etanercept is typically her first choice because patients with systemic hypersensitivity reactions with major organ involvement are often quite ill, making cyclosporine harder to use. In her experience, etanercept has been well tolerated.
Conversely, she cautioned against the use of intravenous immunoglobulin (IVIG). Although this has been used traditionally for severe drug hypersensitivity reactions, “the data are not there,” she said. The data are stronger for a combination of high-dose steroids and IVIG, but she thinks even these data are inconsistent and not as strong as the data supporting etanercept or cyclosporine. She encouraged centers still using IVIG to consider alternatives.
After drug sensitivity reactions are controlled, follow-up care is particularly important for Black patients who face greater risks for sequelae, such as hypopigmentation, hyperpigmentation, or keloids. She recommended aggressive use of emollients and sunscreens for an extended period after lesions resolve to lessen these risks.
Differences in the manifestations of drug-induced skin rashes by race and ethnicity are important and perhaps underappreciated, agreed Shawn Kwatra, MD, professor and chairman of the Department of Dermatology, University of Maryland, Baltimore.
Asked to comment at the meeting, Dr. Kwatra said that he appreciated Dr. Harp’s effort to translate published data and her experience into an overview that increases awareness of the risk for missed or delayed diagnoses of drug-induced rashes in skin of color. He noted that the strategies to identify erythema and pustules, such as increased suspicion in skin of color and the extra steps to rule them out, such as the use of side lighting in the case of pustules for AGEP, are simple and practical.
Dr. Harp and Dr. Kwatra had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM SOC 2024
Nonscaly Red-Brown Macules on the Feet and Ankles
THE DIAGNOSIS: Secondary Syphilis
Histopathology demonstrated a mild superficial perivascular and interstitial infiltrate composed of lymphocytes, histiocytes, and rare plasma cells with a background of extravasated erythrocytes (Figure, A). Treponema pallidum staining highlighted multiple spirochetes along the dermoepidermal junction and in the superficial dermis (Figure, B). Direct immunofluorescence was negative. Laboratory workup revealed a reactive rapid plasma reagin screen with a titer of 1:16 and positive IgG and IgM treponemal antibodies. The patient was diagnosed with secondary syphilis and was treated with a single dose of 2.4 million U of intramuscular benzathine penicillin G, with notable improvement of the rash and arthritis symptoms at 2-week follow-up.
Syphilis is a sexually transmitted infection caused by the spirochete T pallidum that progresses through active and latent stages. The incidence of both the primary and secondary stages of syphilis was at a historic low in the year 2000 and has increased annually since then.1 Syphilis is more common in men, and men who have sex with men (MSM) are disproportionately affected. Although the incidence of syphilis in MSM has increased since 2000, rates have slowed, with slight decreases in this population between 2019 and 2020.1 Conversely, rates among women have increased substantially in recent years, suggesting a more recent epidemic affecting heterosexual men and women.2
Classically, the primary stage of syphilis manifests as an asymptomatic papule followed by a painless ulcer (chancre) that heals spontaneously. The secondary stage of syphilis results from dissemination of T pallidum and is characterized by a wide range of mucocutaneous manifestations and prodromal symptoms. The most common cutaneous manifestation is a diffuse, nonpruritic, papulosquamous rash with red-brown scaly macules or papules on the trunk and extremities.3 The palms and soles commonly are involved. Mucosal patches, “snail-track” ulcers in the mouth, and condylomata lata are the characteristic mucosal lesions of secondary syphilis. Mucocutaneous findings typically are preceded by systemic signs including fever, malaise, myalgia, and generalized lymphadenopathy. However, syphilis is considered “the great mimicker,” with new reports of unusual presentations of the disease. In addition to papulosquamous morphologies, pustular, targetoid, psoriasiform, and noduloulcerative (also known as lues maligna) forms of syphilis have been reported.3-5
The histopathologic features of secondary syphilis also are variable. Classically, secondary syphilis demonstrates vacuolar interface dermatitis and acanthosis with slender elongated rete ridges. Other well-known features include endothelial swelling and the presence of plasma cells in most cases.6 However, the histopathologic features of secondary syphilis may vary depending on the morphology of the skin eruption and when the biopsy is taken. Our patient lacked the classic histopathologic features of secondary syphilis. However, because syphilis was in the clinical differential diagnosis, a treponemal stain was ordered and confirmed the diagnosis. Immunohistochemical stains using antibodies to treponemal antigens have a reported sensitivity of 71% to 100% and are highly specific.7 Although the combination of endothelial swelling, interstitial inflammation, irregular acanthosis, and elongated rete ridges should raise the possibility of syphilis, a treponemal stain may be useful to identify spirochetes if clinical suspicion exists.8
Given our patient’s known history of GPA, leukocytoclastic vasculitis was high on the list of differential diagnoses. However, leukocytoclastic vasculitis most classically manifests as petechiae and palpable purpura, and unlike in secondary syphilis, the palms and soles are less commonly involved. Because our patient’s rash was mainly localized to the lower limbs, the differential also included 2 pigmented purpuric dermatoses (PPDs): progressive pigmentary purpura (Schamberg disease) and purpura annularis telangiectodes (Majocchi disease). Progressive pigmentary purpura is the most common manifestation of PPD and appears as cayenne pepper–colored macules that coalesce into golden brown–pigmented patches on the legs.9 Purpura annularis telangiectodes is another variant of PPD that manifests as pinpoint telangiectatic macules that progress to annular hyperpigmented patches with central clearing. Although PPDs frequently occur on the lower extremities, reports of plantar involvement are rare.10 Annular lichen planus manifests as violaceous papules with a clear center; however, it would be atypical for these lesions to be restricted to the feet and ankles. Palmoplantar lichen planus can mimic secondary syphilis clinically, but these cases manifest as hyperkeratotic pruritic papules on the palms and soles in contrast to the faint brown asymptomatic macules noted in our case.11
Our case highlights an unusual presentation of secondary syphilis and demonstrates the challenge of diagnosing this entity on clinical presentation alone. Because this patient lacked the classic clinical and histopathologic features of secondary syphilis, a skin biopsy with positive immunohistochemical staining for treponemal antigens was necessary to make the diagnosis. Given the variability in presentation of secondary syphilis, a biopsy or serologic testing may be necessary to make a proper diagnosis.
- Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2020. Accessed September 4, 2024. https://www.cdc.gov/std/statistics/2020/2020-SR-4-10-2023.pdf
- Ghanem KG, Ram S, Rice PA. The modern epidemic of syphilis. N Engl J Med. 2020;382:845-854. doi:10.1056/NEJMra1901593
- Forrestel AK, Kovarik CL, Katz KA. Sexually acquired syphilis: historical aspects, microbiology, epidemiology, and clinical manifestations. J Am Acad Dermatol. 2020;82:1-14. doi:10.1016/j.jaad.2019.02.073
- Wu MC, Hsu CK, Lee JY, et al. Erythema multiforme-like secondary syphilis in a HIV-positive bisexual man. Acta Derm Venereol. 2010;90:647-648. doi:10.2340/00015555-0920
- Kopelman H, Lin A, Jorizzo JL. A pemphigus-like presentation of secondary syphilis. JAAD Case Rep. 2019;5:861-864. doi:10.1016/j.jdcr.2019.07.021
- Liu XK, Li J. Histologic features of secondary syphilis. Dermatology. 2020;236:145-150. doi:10.1159/000502641
- Forrestel AK, Kovarik CL, Katz KA. Sexually acquired syphilis: laboratory diagnosis, management, and prevention. J Am Acad Dermatol. 2020;82:17-28. doi:10.1016/j.jaad.2019.02.074
- Flamm A, Parikh K, Xie Q, et al. Histologic features of secondary syphilis: a multicenter retrospective review. J Am Acad Dermatol. 2015;73:1025-1030. doi:10.1016/j.jaad.2015.08.062
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410. doi:10.5021/ad.2015.27.4.404
- Sivendran M, Mowad C. Hyperpigmented patches on shins, palms, and soles. JAMA Dermatol. 2013;149:223. doi:10.1001/2013.jamadermatol.652a
- Kim YS, Kim MH, Kim CW, et al. A case of palmoplantar lichen planus mimicking secondary syphilis. Ann Dermatol. 2009;21:429-431.doi:10.5021/ad.2009.21.4.429
THE DIAGNOSIS: Secondary Syphilis
Histopathology demonstrated a mild superficial perivascular and interstitial infiltrate composed of lymphocytes, histiocytes, and rare plasma cells with a background of extravasated erythrocytes (Figure, A). Treponema pallidum staining highlighted multiple spirochetes along the dermoepidermal junction and in the superficial dermis (Figure, B). Direct immunofluorescence was negative. Laboratory workup revealed a reactive rapid plasma reagin screen with a titer of 1:16 and positive IgG and IgM treponemal antibodies. The patient was diagnosed with secondary syphilis and was treated with a single dose of 2.4 million U of intramuscular benzathine penicillin G, with notable improvement of the rash and arthritis symptoms at 2-week follow-up.

Syphilis is a sexually transmitted infection caused by the spirochete T pallidum that progresses through active and latent stages. The incidence of both the primary and secondary stages of syphilis was at a historic low in the year 2000 and has increased annually since then.1 Syphilis is more common in men, and men who have sex with men (MSM) are disproportionately affected. Although the incidence of syphilis in MSM has increased since 2000, rates have slowed, with slight decreases in this population between 2019 and 2020.1 Conversely, rates among women have increased substantially in recent years, suggesting a more recent epidemic affecting heterosexual men and women.2
Classically, the primary stage of syphilis manifests as an asymptomatic papule followed by a painless ulcer (chancre) that heals spontaneously. The secondary stage of syphilis results from dissemination of T pallidum and is characterized by a wide range of mucocutaneous manifestations and prodromal symptoms. The most common cutaneous manifestation is a diffuse, nonpruritic, papulosquamous rash with red-brown scaly macules or papules on the trunk and extremities.3 The palms and soles commonly are involved. Mucosal patches, “snail-track” ulcers in the mouth, and condylomata lata are the characteristic mucosal lesions of secondary syphilis. Mucocutaneous findings typically are preceded by systemic signs including fever, malaise, myalgia, and generalized lymphadenopathy. However, syphilis is considered “the great mimicker,” with new reports of unusual presentations of the disease. In addition to papulosquamous morphologies, pustular, targetoid, psoriasiform, and noduloulcerative (also known as lues maligna) forms of syphilis have been reported.3-5
The histopathologic features of secondary syphilis also are variable. Classically, secondary syphilis demonstrates vacuolar interface dermatitis and acanthosis with slender elongated rete ridges. Other well-known features include endothelial swelling and the presence of plasma cells in most cases.6 However, the histopathologic features of secondary syphilis may vary depending on the morphology of the skin eruption and when the biopsy is taken. Our patient lacked the classic histopathologic features of secondary syphilis. However, because syphilis was in the clinical differential diagnosis, a treponemal stain was ordered and confirmed the diagnosis. Immunohistochemical stains using antibodies to treponemal antigens have a reported sensitivity of 71% to 100% and are highly specific.7 Although the combination of endothelial swelling, interstitial inflammation, irregular acanthosis, and elongated rete ridges should raise the possibility of syphilis, a treponemal stain may be useful to identify spirochetes if clinical suspicion exists.8
Given our patient’s known history of GPA, leukocytoclastic vasculitis was high on the list of differential diagnoses. However, leukocytoclastic vasculitis most classically manifests as petechiae and palpable purpura, and unlike in secondary syphilis, the palms and soles are less commonly involved. Because our patient’s rash was mainly localized to the lower limbs, the differential also included 2 pigmented purpuric dermatoses (PPDs): progressive pigmentary purpura (Schamberg disease) and purpura annularis telangiectodes (Majocchi disease). Progressive pigmentary purpura is the most common manifestation of PPD and appears as cayenne pepper–colored macules that coalesce into golden brown–pigmented patches on the legs.9 Purpura annularis telangiectodes is another variant of PPD that manifests as pinpoint telangiectatic macules that progress to annular hyperpigmented patches with central clearing. Although PPDs frequently occur on the lower extremities, reports of plantar involvement are rare.10 Annular lichen planus manifests as violaceous papules with a clear center; however, it would be atypical for these lesions to be restricted to the feet and ankles. Palmoplantar lichen planus can mimic secondary syphilis clinically, but these cases manifest as hyperkeratotic pruritic papules on the palms and soles in contrast to the faint brown asymptomatic macules noted in our case.11
Our case highlights an unusual presentation of secondary syphilis and demonstrates the challenge of diagnosing this entity on clinical presentation alone. Because this patient lacked the classic clinical and histopathologic features of secondary syphilis, a skin biopsy with positive immunohistochemical staining for treponemal antigens was necessary to make the diagnosis. Given the variability in presentation of secondary syphilis, a biopsy or serologic testing may be necessary to make a proper diagnosis.
THE DIAGNOSIS: Secondary Syphilis
Histopathology demonstrated a mild superficial perivascular and interstitial infiltrate composed of lymphocytes, histiocytes, and rare plasma cells with a background of extravasated erythrocytes (Figure, A). Treponema pallidum staining highlighted multiple spirochetes along the dermoepidermal junction and in the superficial dermis (Figure, B). Direct immunofluorescence was negative. Laboratory workup revealed a reactive rapid plasma reagin screen with a titer of 1:16 and positive IgG and IgM treponemal antibodies. The patient was diagnosed with secondary syphilis and was treated with a single dose of 2.4 million U of intramuscular benzathine penicillin G, with notable improvement of the rash and arthritis symptoms at 2-week follow-up.

Syphilis is a sexually transmitted infection caused by the spirochete T pallidum that progresses through active and latent stages. The incidence of both the primary and secondary stages of syphilis was at a historic low in the year 2000 and has increased annually since then.1 Syphilis is more common in men, and men who have sex with men (MSM) are disproportionately affected. Although the incidence of syphilis in MSM has increased since 2000, rates have slowed, with slight decreases in this population between 2019 and 2020.1 Conversely, rates among women have increased substantially in recent years, suggesting a more recent epidemic affecting heterosexual men and women.2
Classically, the primary stage of syphilis manifests as an asymptomatic papule followed by a painless ulcer (chancre) that heals spontaneously. The secondary stage of syphilis results from dissemination of T pallidum and is characterized by a wide range of mucocutaneous manifestations and prodromal symptoms. The most common cutaneous manifestation is a diffuse, nonpruritic, papulosquamous rash with red-brown scaly macules or papules on the trunk and extremities.3 The palms and soles commonly are involved. Mucosal patches, “snail-track” ulcers in the mouth, and condylomata lata are the characteristic mucosal lesions of secondary syphilis. Mucocutaneous findings typically are preceded by systemic signs including fever, malaise, myalgia, and generalized lymphadenopathy. However, syphilis is considered “the great mimicker,” with new reports of unusual presentations of the disease. In addition to papulosquamous morphologies, pustular, targetoid, psoriasiform, and noduloulcerative (also known as lues maligna) forms of syphilis have been reported.3-5
The histopathologic features of secondary syphilis also are variable. Classically, secondary syphilis demonstrates vacuolar interface dermatitis and acanthosis with slender elongated rete ridges. Other well-known features include endothelial swelling and the presence of plasma cells in most cases.6 However, the histopathologic features of secondary syphilis may vary depending on the morphology of the skin eruption and when the biopsy is taken. Our patient lacked the classic histopathologic features of secondary syphilis. However, because syphilis was in the clinical differential diagnosis, a treponemal stain was ordered and confirmed the diagnosis. Immunohistochemical stains using antibodies to treponemal antigens have a reported sensitivity of 71% to 100% and are highly specific.7 Although the combination of endothelial swelling, interstitial inflammation, irregular acanthosis, and elongated rete ridges should raise the possibility of syphilis, a treponemal stain may be useful to identify spirochetes if clinical suspicion exists.8
Given our patient’s known history of GPA, leukocytoclastic vasculitis was high on the list of differential diagnoses. However, leukocytoclastic vasculitis most classically manifests as petechiae and palpable purpura, and unlike in secondary syphilis, the palms and soles are less commonly involved. Because our patient’s rash was mainly localized to the lower limbs, the differential also included 2 pigmented purpuric dermatoses (PPDs): progressive pigmentary purpura (Schamberg disease) and purpura annularis telangiectodes (Majocchi disease). Progressive pigmentary purpura is the most common manifestation of PPD and appears as cayenne pepper–colored macules that coalesce into golden brown–pigmented patches on the legs.9 Purpura annularis telangiectodes is another variant of PPD that manifests as pinpoint telangiectatic macules that progress to annular hyperpigmented patches with central clearing. Although PPDs frequently occur on the lower extremities, reports of plantar involvement are rare.10 Annular lichen planus manifests as violaceous papules with a clear center; however, it would be atypical for these lesions to be restricted to the feet and ankles. Palmoplantar lichen planus can mimic secondary syphilis clinically, but these cases manifest as hyperkeratotic pruritic papules on the palms and soles in contrast to the faint brown asymptomatic macules noted in our case.11
Our case highlights an unusual presentation of secondary syphilis and demonstrates the challenge of diagnosing this entity on clinical presentation alone. Because this patient lacked the classic clinical and histopathologic features of secondary syphilis, a skin biopsy with positive immunohistochemical staining for treponemal antigens was necessary to make the diagnosis. Given the variability in presentation of secondary syphilis, a biopsy or serologic testing may be necessary to make a proper diagnosis.
- Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2020. Accessed September 4, 2024. https://www.cdc.gov/std/statistics/2020/2020-SR-4-10-2023.pdf
- Ghanem KG, Ram S, Rice PA. The modern epidemic of syphilis. N Engl J Med. 2020;382:845-854. doi:10.1056/NEJMra1901593
- Forrestel AK, Kovarik CL, Katz KA. Sexually acquired syphilis: historical aspects, microbiology, epidemiology, and clinical manifestations. J Am Acad Dermatol. 2020;82:1-14. doi:10.1016/j.jaad.2019.02.073
- Wu MC, Hsu CK, Lee JY, et al. Erythema multiforme-like secondary syphilis in a HIV-positive bisexual man. Acta Derm Venereol. 2010;90:647-648. doi:10.2340/00015555-0920
- Kopelman H, Lin A, Jorizzo JL. A pemphigus-like presentation of secondary syphilis. JAAD Case Rep. 2019;5:861-864. doi:10.1016/j.jdcr.2019.07.021
- Liu XK, Li J. Histologic features of secondary syphilis. Dermatology. 2020;236:145-150. doi:10.1159/000502641
- Forrestel AK, Kovarik CL, Katz KA. Sexually acquired syphilis: laboratory diagnosis, management, and prevention. J Am Acad Dermatol. 2020;82:17-28. doi:10.1016/j.jaad.2019.02.074
- Flamm A, Parikh K, Xie Q, et al. Histologic features of secondary syphilis: a multicenter retrospective review. J Am Acad Dermatol. 2015;73:1025-1030. doi:10.1016/j.jaad.2015.08.062
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410. doi:10.5021/ad.2015.27.4.404
- Sivendran M, Mowad C. Hyperpigmented patches on shins, palms, and soles. JAMA Dermatol. 2013;149:223. doi:10.1001/2013.jamadermatol.652a
- Kim YS, Kim MH, Kim CW, et al. A case of palmoplantar lichen planus mimicking secondary syphilis. Ann Dermatol. 2009;21:429-431.doi:10.5021/ad.2009.21.4.429
- Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2020. Accessed September 4, 2024. https://www.cdc.gov/std/statistics/2020/2020-SR-4-10-2023.pdf
- Ghanem KG, Ram S, Rice PA. The modern epidemic of syphilis. N Engl J Med. 2020;382:845-854. doi:10.1056/NEJMra1901593
- Forrestel AK, Kovarik CL, Katz KA. Sexually acquired syphilis: historical aspects, microbiology, epidemiology, and clinical manifestations. J Am Acad Dermatol. 2020;82:1-14. doi:10.1016/j.jaad.2019.02.073
- Wu MC, Hsu CK, Lee JY, et al. Erythema multiforme-like secondary syphilis in a HIV-positive bisexual man. Acta Derm Venereol. 2010;90:647-648. doi:10.2340/00015555-0920
- Kopelman H, Lin A, Jorizzo JL. A pemphigus-like presentation of secondary syphilis. JAAD Case Rep. 2019;5:861-864. doi:10.1016/j.jdcr.2019.07.021
- Liu XK, Li J. Histologic features of secondary syphilis. Dermatology. 2020;236:145-150. doi:10.1159/000502641
- Forrestel AK, Kovarik CL, Katz KA. Sexually acquired syphilis: laboratory diagnosis, management, and prevention. J Am Acad Dermatol. 2020;82:17-28. doi:10.1016/j.jaad.2019.02.074
- Flamm A, Parikh K, Xie Q, et al. Histologic features of secondary syphilis: a multicenter retrospective review. J Am Acad Dermatol. 2015;73:1025-1030. doi:10.1016/j.jaad.2015.08.062
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410. doi:10.5021/ad.2015.27.4.404
- Sivendran M, Mowad C. Hyperpigmented patches on shins, palms, and soles. JAMA Dermatol. 2013;149:223. doi:10.1001/2013.jamadermatol.652a
- Kim YS, Kim MH, Kim CW, et al. A case of palmoplantar lichen planus mimicking secondary syphilis. Ann Dermatol. 2009;21:429-431.doi:10.5021/ad.2009.21.4.429
A 59-year-old man presented with a nontender nonpruritic rash on the feet of 2 days’ duration. The patient had a several-year history of granulomatosis with polyangiitis (GPA) and was taking methotrexate and prednisone. The rash appeared suddenly—first on the right foot and then on the left foot—and was preceded by 1 week of worsening polyarthralgia, most notably in the ankles. He denied any fever, chills, sore throat, or weight loss. His typical GPA symptoms included inflammatory arthritis, scleritis, leukocytoclastic vasculitis, and sinonasal and renal involvement. He recently experienced exacerbation of inflammatory arthritis that required an increase in the prednisone dosage (from 40 mg to 60 mg daily), but there were no other GPA symptoms. He had a history of multiple female sexual partners but no known history of HIV and no recent testing for sexually transmitted infections. Hepatitis C antibody testing performed 5 years earlier was nonreactive. He denied any illicit drug use, recent travel, sick contacts, or new medications.
Dermatologic examination revealed nonscaly, clustered, red-brown macules, some with central clearing, on the medial and lateral aspects of the feet and ankles with a few faint copper-colored macules on the palms and soles. The ankles had full range of motion with no edema or effusion. There were no oral or genital lesions. The remainder of the skin examination was normal. Punch biopsies of skin on the left foot were obtained for histopathology and direct immunofluorescence.