User login
Hypochlorous Acid: A Multipurpose New Addition to the Military Med Bag?
Hypochlorous Acid: A Multipurpose New Addition to the Military Med Bag?
Exogenously, hypochlorous acid (HOCl) is a powerful oxidizing agent formed from chlorine dissolved in water. Within the body, it is part of the immune response, created by activated leukocytes, which form HOCl from hydrogen peroxide and chloride. HOCl has been used as a disinfectant in wound care due to its antimicrobial properties via inhibition of DNA synthesis, protein synthesis, and decreased adenosine triphosphate production. It specifically targets bacteria by blocking bacterial cell wall synthesis and decreasing DNA replication.1
During the COVID-19 pandemic, HOCl was recommended by the US Environmental Protection Agency as a disinfectant.2 HOCl can be purchased from a supplier, though its major limitation is its shelf life. The main environmental factors affecting its stability are sunlight exposure, temperatures > 25 °C, and air exposure. HOCl is stable and most potent when the pH falls between 3.5 and 5.5.3 It is best stored in a cool, dark environment to maintain efficacy for 2 weeks. Rossi-Fedele et al found that when exposed to sunlight, chlorine reduction starts on day 4, whereas solutions kept in dark storage remained more stable, with this process starting after day 14.4
HOCl can also be made on-site via a machine, which ranges in price from a portable version costing < $200 to a large commercial option that can cost $7000 to $25,000. HOCl is produced by mixing noniodinated salt and water, and using electrolysis, which generally takes less than 10 minutes before it is ready for use.2 Given the cost and nonreusable nature of disinfecting wipes, HOCl may be more worthwhile for economic and disposal purposes in the long term.
Different concentrations of HOCl are readily available commercially. Because topical application of 1% HOCl may cause skin irritation, solutions with lower concentrations have been developed including Vashe (0.03% HOCl; SteadMed), PhaseOne (0.025% HOCl solution; IHT), OCuSOFT (0.02% HOCl; OCuSOFT), Bruder (0.02% HOCl; Bruder Healthcare), Acuicyn (0.01% HOCl solution in dilute saline; Sonoma Pharmaceuticals), and Avenova (0.01% HOCl solution; NovaBay Pharmaceuticals).5
Aside from its surface utility, HOCl has been researched for its beneficial effects on skin. HOCl has been shown to be helpful intraoperatively and postoperatively in improving adverse effects (AEs) after hair restoration, including erythema and pruritus, and in optimizing healing by reducing inflammation, likely due to its antimicrobial properties and ability to promote oxygenation.6 Bucko et al demonstrated that Microcyn scar gel (with HOCl) was a superior nonirritating, nontoxic method of not only improving scar appearance (vascularity, scar height, and pliability) but also reducing scar symptoms of pain and pruritus in comparison to 100% silicone scar gel (traditional application used to improve scarring).7 Zhang et al demonstrated that HOCl consistently improved symptom relief of blepharitis, including meibomian gland, eyelash, and eyelid redness, irritation, and appearance in comparison as well as were better tolerated in comparison to traditional recommendations of eyelid compresses and wash (tea tree oil, diluted baby shampoo, and topical antibiotics).8 In children with moderate to severe atopic dermatitis, Majewski et al compared a traditional bleach bath with a body wash containing hypochlorite (NaOCl; hypochlorous acid in alkaline aqueous solution). The body wash proved to be more convenient (showering vs 10-minute bath) and significantly improved symptoms while reducing the need for topical corticosteroids (common treatment modality for atopic dermatitis).9
The skin is the body’s primary defense against both dermatologic and respiratory infections. The face is especially vulnerable to microbes via airborne or environmental transmission, mechanical irritation, and touch. In the military environment, personal protective equipment (PPE) or uniform items may increase the risk of dermatologic conditions such as allergic or irritant dermatitis, infection, and friction blisters.
In a literature review of 312 dermatologic articles published between 2002 and 2022, Singal and Lipner found that among deployed soldiers serving in hot and dry climates, dermatitis and eczematous conditions were the most common, whereas bacterial and fungal conditions were most common in hot and humid settings. In the nondeployed setting, dermatitis and eczematous, acne, and fungal infections were the most common skin conditions. This is reflected by the unique circumstances that service members face at home and while deployed, when they may be more vulnerable to developing new or worsening chronic skin conditions depending on the environment (access to shelter, humid vs dry environments), and decreased access or time for hygiene (shared quarters at home in barracks or on deployment). Occupation-related conditions also play a large role in military dermatologic conditions.10
Dever et al noted the unique risks and exposures in the environment itself (plants, arthropods) as well as uniform items (protective gear) that carry an increased risk of friction irritation and dermatitis. Occupational exposures commonly associated with irritant contact dermatitis include alcohols, oils, fuel, disinfectants, and solvents. Chemicals in military uniforms themselves (eg, formaldehyde resins, disperse dyes, and chromate-containing dyes) also have the potential to cause allergic contact dermatitis, which can be challenging to address given the emphasis on uniformity and standards.11 PPE also may exacerbate rosacea and acne.
Some pathologies are associated specifically with bacteria, such as Cutibacterium acnes, as seen in acne vulgaris. Colonization of bacteria on the face may create biofilms that are difficult to detect, may be resistant to antibiotic therapy, and are implicated in other dermatologic conditions, such as persistent wounds, atopic dermatitis, and candidiasis.12
Biofilm and antibiotic resistance already pose a risk to patient care, but the unique environmental conditions and exposures of military settings can amplify this risk in the military population.13 Using HOCl in austere environments or the field for wound care may help reduce microbial load and the subsequent need for systemic antibiotics which carry the risk of gastrointestinal AEs and resistance.1
An optimized healing rate would support operational objectives by enabling service members to remain on full duty and avoid medications, which may prevent them from special duty, such as aviation. Sakarya et al found that HOCl solution enhanced wound healing in contrast with povidone-iodine (PI), while a study by Dharap et al discussed how HOCl provided major improvement in ulcer wound size (and infection), as well as significant reduction of inflammation.13
Anagnostopoulos et al studied the efficacy of 0.01% HOCl vs other disinfectants (5% PI, 4% chlorhexidine gluconate [CHG] and 70% isopropyl alcohol [IPA]) against common skin organisms, including methicillin-susceptible Staphylococcus aureus and methicillin-susceptible Staphylococcus epidermidis. The study found that HOCl had at least equal if not greater efficacy to PI, CHG, and IPA depending on the bacterial strain, demonstrating immediate bactericidal effects.14
Furthermore, HOCl has been shown to be useful in suturing and wound closure by reducing microbial load when soaked gauze is placed in wound beds prior to closure, while not harming surrounding tissue.15 This would be especially advantageous for military health care when specialist follow-up would be delayed or to prevent infection risk while en route to higher care. Aside from its disinfectant strength, it’s also well tolerated. HOCl studies on human tissue demonstrate its efficacy to prevent irritation and AEs while also preventing infection and promoting wound healing.
Gozukucuk and Cakiroglu studied the use of HOCl as a skin disinfectant before neonatal circumcision and demonstrated fewer adverse effects compared with the more commonly used PI. Neonates treated with PI prior to circumcision resulted in greater postoperative edema and increased duration of wound healing compared with infants treated with HOCl.16 Furthermore, studies have shown that PI can lead to irritant dermatitis or chemical burns if not properly dried or if it becomes pooled because of occlusion dressings.17
Aside from its indicated use for infection or wound care, anti-inflammatory properties of HOCl also may be beneficial for off-label use in preventing flareups of chronic conditions as well as for treating symptoms while awaiting specialist evaluation. This might be the case during US-based training exercises, in remote locations without nearby dermatologists, or during virtual care because of internet constraints. For chronic conditions such as rosacea or atopic dermatitis, which research has shown are related to mast cell activation and degranulation and cytokine release, HOCl has been shown to reduce histamine, neutrophil-generated leukotrienes, in addition to interleukin-6 and interleukin-2 to improve symptoms by reducing inflammation.18
Limitations of HOCl to explore would be extending its shelf life, exploring its various forms (eg, spray, topical) and storage limitations, and training of the machine and materials needed to be made in-house if not purchased. There are also no official guidelines for clinicians to recommend HOCl to patients, and research should be expanded on its use in humans, though it generally is well tolerated without AEs. HOCl has the potential to be a potent, nontoxic, inexpensive tool in med bags or at austere clinics to help maintain a sterile space for procedures, prevent infection while rendering care, and help with exacerbations or prevent flare-ups of chronic conditions such as psoriasis, acne, and atopic dermatitis while specialist care is pending.
- Natarelli N, et al. Hypochlorous acid: applications in dermatology. J Integr Dermatol. December 22, 2022. Accessed March 2, 2026. https://www.jintegrativederm.org/article/56663-hypochlorous-acid-applications-in-dermatology
- Block MS, Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg. 2020;78:1461-1466. doi:10.1016/j.joms.2020.06.029
- Menta N, Vidal SI, Friedman A. Hypochlorous acid: a blast from the past. J Drugs Dermatol. 2024;23:909-910.
- Rossi-Fedele G, Dogramaci E, Steier L, et al. Some factors influencing the stability of Sterilox®, a super-oxidised water. Br Dent J. 2011;210:E23. doi:10.1038/sj.bdj.2011.143
- Tran AQ, Topilow N, Rong A, et al. Comparison of skin antiseptic agents and the role of 0.01% hypochlorous acid. Aesthet Surg J. 2021;41:1170-1175. doi:10.1093/asj/sjaa322
- Stough D. Topical stabilized super-oxidized hypochlorous acid for wound healing in hair restoration surgery: a real-time usage-controlled trial evaluating safety, efficacy, and tolerability. J Drugs Dermatol. 2023;22:1191-1196. doi:10.36849/JDD.7172
- Bucko AD, Draelos Z, Dubois JC, Jones TM. A doubleblind, randomized study to compare Microcyn scar management hydrogel, K103163, and Kelo-cote scar gel for hypertrophic or keloid scars. Dermatologist. 2015;23:113-122.
- Zhang H, Wu Y, Wan X, et al. Effect of hypochlorous acid on blepharitis through ultrasonic atomization: a randomized clinical trial. J Clin Med. 2023;12(3):1164. doi:10.3390/jcm12031164
- Majewski S, Bhattacharya T, Asztalos M, et al. Sodium hypochlorite body wash in the management of Staphylococcus aureus-colonized moderate-to-severe atopic dermatitis in infants, children, and adolescents. Pediatr Dermatol. 2019;36:442-447. doi:10.1111/pde.13842
- Singal A, Lipner SR. A review of skin disease in military soldiers: challenges and potential solutions. Ann Med. 2023;55:2267425. doi:10.1080/07853890.2023.2267425
- Dever TT, Walters M, Jacob S. Contact dermatitis in military personnel. Dermatitis. 2011;22:313-319. doi:10.2310/6620.2011.11024
- Nowbuth AA, Armstrong J, Cloete T, et al. A potential benefit of hypochlorous acid-facial sanitisation: a review. Preprints. 2021. doi:10.20944/preprints202107.0129.v2
- Gold MH, Andriessen A, Bhatia AC, et al. Topical stabilized hypochlorous acid: the future gold standard for wound care and scar management in dermatologic and plastic surgery procedures. J Cosmet Dermatol. 2020;19:270-277. doi:10.1111/jocd.13280
- Anagnostopoulos AG, Rong A, Miller D, et al. 0.01% hypochlorous acid as an alternative skin antiseptic: an in vitro comparison. Dermatol Surg. 2018;44:1489-1493. doi:10.1097/DSS.0000000000001594
- Odom EB, Mundschenk MB, Hard KA, et al. The utility of hypochlorous acid wound therapy in wound bed preparation and skin graft salvage. Plast Reconstr Surg. 2019;143:677e-678e. doi:10.1097/PRS.0000000000005359
- Gozukucuk A, Cakiroglu B. Comparison of hypochlorous acid and povidone-iodine as a disinfectant in neonatal circumcision. J Pediatr Urol. 2022;18:341.e1-341.e5. doi:10.1016/j.jpurol.2022.03.011
- Borrego L, Hernández N, Hernández Z, et al. Povidoneiodine-induced postsurgical irritant contact dermatitis localized outside of the surgical incision area: report of 27 cases and a literature review. Int J Dermatol. 2016;55:540- 545. doi:10.1111/ijd.12957
- Del Rosso JQ, Bhatia N. Status report on topical hypochlorous acid: clinical relevance of specific formulations, potential modes of action, and study outcomes. J Clin Aesthet Dermatol. 2018;11:36-39.
Exogenously, hypochlorous acid (HOCl) is a powerful oxidizing agent formed from chlorine dissolved in water. Within the body, it is part of the immune response, created by activated leukocytes, which form HOCl from hydrogen peroxide and chloride. HOCl has been used as a disinfectant in wound care due to its antimicrobial properties via inhibition of DNA synthesis, protein synthesis, and decreased adenosine triphosphate production. It specifically targets bacteria by blocking bacterial cell wall synthesis and decreasing DNA replication.1
During the COVID-19 pandemic, HOCl was recommended by the US Environmental Protection Agency as a disinfectant.2 HOCl can be purchased from a supplier, though its major limitation is its shelf life. The main environmental factors affecting its stability are sunlight exposure, temperatures > 25 °C, and air exposure. HOCl is stable and most potent when the pH falls between 3.5 and 5.5.3 It is best stored in a cool, dark environment to maintain efficacy for 2 weeks. Rossi-Fedele et al found that when exposed to sunlight, chlorine reduction starts on day 4, whereas solutions kept in dark storage remained more stable, with this process starting after day 14.4
HOCl can also be made on-site via a machine, which ranges in price from a portable version costing < $200 to a large commercial option that can cost $7000 to $25,000. HOCl is produced by mixing noniodinated salt and water, and using electrolysis, which generally takes less than 10 minutes before it is ready for use.2 Given the cost and nonreusable nature of disinfecting wipes, HOCl may be more worthwhile for economic and disposal purposes in the long term.
Different concentrations of HOCl are readily available commercially. Because topical application of 1% HOCl may cause skin irritation, solutions with lower concentrations have been developed including Vashe (0.03% HOCl; SteadMed), PhaseOne (0.025% HOCl solution; IHT), OCuSOFT (0.02% HOCl; OCuSOFT), Bruder (0.02% HOCl; Bruder Healthcare), Acuicyn (0.01% HOCl solution in dilute saline; Sonoma Pharmaceuticals), and Avenova (0.01% HOCl solution; NovaBay Pharmaceuticals).5
Aside from its surface utility, HOCl has been researched for its beneficial effects on skin. HOCl has been shown to be helpful intraoperatively and postoperatively in improving adverse effects (AEs) after hair restoration, including erythema and pruritus, and in optimizing healing by reducing inflammation, likely due to its antimicrobial properties and ability to promote oxygenation.6 Bucko et al demonstrated that Microcyn scar gel (with HOCl) was a superior nonirritating, nontoxic method of not only improving scar appearance (vascularity, scar height, and pliability) but also reducing scar symptoms of pain and pruritus in comparison to 100% silicone scar gel (traditional application used to improve scarring).7 Zhang et al demonstrated that HOCl consistently improved symptom relief of blepharitis, including meibomian gland, eyelash, and eyelid redness, irritation, and appearance in comparison as well as were better tolerated in comparison to traditional recommendations of eyelid compresses and wash (tea tree oil, diluted baby shampoo, and topical antibiotics).8 In children with moderate to severe atopic dermatitis, Majewski et al compared a traditional bleach bath with a body wash containing hypochlorite (NaOCl; hypochlorous acid in alkaline aqueous solution). The body wash proved to be more convenient (showering vs 10-minute bath) and significantly improved symptoms while reducing the need for topical corticosteroids (common treatment modality for atopic dermatitis).9
The skin is the body’s primary defense against both dermatologic and respiratory infections. The face is especially vulnerable to microbes via airborne or environmental transmission, mechanical irritation, and touch. In the military environment, personal protective equipment (PPE) or uniform items may increase the risk of dermatologic conditions such as allergic or irritant dermatitis, infection, and friction blisters.
In a literature review of 312 dermatologic articles published between 2002 and 2022, Singal and Lipner found that among deployed soldiers serving in hot and dry climates, dermatitis and eczematous conditions were the most common, whereas bacterial and fungal conditions were most common in hot and humid settings. In the nondeployed setting, dermatitis and eczematous, acne, and fungal infections were the most common skin conditions. This is reflected by the unique circumstances that service members face at home and while deployed, when they may be more vulnerable to developing new or worsening chronic skin conditions depending on the environment (access to shelter, humid vs dry environments), and decreased access or time for hygiene (shared quarters at home in barracks or on deployment). Occupation-related conditions also play a large role in military dermatologic conditions.10
Dever et al noted the unique risks and exposures in the environment itself (plants, arthropods) as well as uniform items (protective gear) that carry an increased risk of friction irritation and dermatitis. Occupational exposures commonly associated with irritant contact dermatitis include alcohols, oils, fuel, disinfectants, and solvents. Chemicals in military uniforms themselves (eg, formaldehyde resins, disperse dyes, and chromate-containing dyes) also have the potential to cause allergic contact dermatitis, which can be challenging to address given the emphasis on uniformity and standards.11 PPE also may exacerbate rosacea and acne.
Some pathologies are associated specifically with bacteria, such as Cutibacterium acnes, as seen in acne vulgaris. Colonization of bacteria on the face may create biofilms that are difficult to detect, may be resistant to antibiotic therapy, and are implicated in other dermatologic conditions, such as persistent wounds, atopic dermatitis, and candidiasis.12
Biofilm and antibiotic resistance already pose a risk to patient care, but the unique environmental conditions and exposures of military settings can amplify this risk in the military population.13 Using HOCl in austere environments or the field for wound care may help reduce microbial load and the subsequent need for systemic antibiotics which carry the risk of gastrointestinal AEs and resistance.1
An optimized healing rate would support operational objectives by enabling service members to remain on full duty and avoid medications, which may prevent them from special duty, such as aviation. Sakarya et al found that HOCl solution enhanced wound healing in contrast with povidone-iodine (PI), while a study by Dharap et al discussed how HOCl provided major improvement in ulcer wound size (and infection), as well as significant reduction of inflammation.13
Anagnostopoulos et al studied the efficacy of 0.01% HOCl vs other disinfectants (5% PI, 4% chlorhexidine gluconate [CHG] and 70% isopropyl alcohol [IPA]) against common skin organisms, including methicillin-susceptible Staphylococcus aureus and methicillin-susceptible Staphylococcus epidermidis. The study found that HOCl had at least equal if not greater efficacy to PI, CHG, and IPA depending on the bacterial strain, demonstrating immediate bactericidal effects.14
Furthermore, HOCl has been shown to be useful in suturing and wound closure by reducing microbial load when soaked gauze is placed in wound beds prior to closure, while not harming surrounding tissue.15 This would be especially advantageous for military health care when specialist follow-up would be delayed or to prevent infection risk while en route to higher care. Aside from its disinfectant strength, it’s also well tolerated. HOCl studies on human tissue demonstrate its efficacy to prevent irritation and AEs while also preventing infection and promoting wound healing.
Gozukucuk and Cakiroglu studied the use of HOCl as a skin disinfectant before neonatal circumcision and demonstrated fewer adverse effects compared with the more commonly used PI. Neonates treated with PI prior to circumcision resulted in greater postoperative edema and increased duration of wound healing compared with infants treated with HOCl.16 Furthermore, studies have shown that PI can lead to irritant dermatitis or chemical burns if not properly dried or if it becomes pooled because of occlusion dressings.17
Aside from its indicated use for infection or wound care, anti-inflammatory properties of HOCl also may be beneficial for off-label use in preventing flareups of chronic conditions as well as for treating symptoms while awaiting specialist evaluation. This might be the case during US-based training exercises, in remote locations without nearby dermatologists, or during virtual care because of internet constraints. For chronic conditions such as rosacea or atopic dermatitis, which research has shown are related to mast cell activation and degranulation and cytokine release, HOCl has been shown to reduce histamine, neutrophil-generated leukotrienes, in addition to interleukin-6 and interleukin-2 to improve symptoms by reducing inflammation.18
Limitations of HOCl to explore would be extending its shelf life, exploring its various forms (eg, spray, topical) and storage limitations, and training of the machine and materials needed to be made in-house if not purchased. There are also no official guidelines for clinicians to recommend HOCl to patients, and research should be expanded on its use in humans, though it generally is well tolerated without AEs. HOCl has the potential to be a potent, nontoxic, inexpensive tool in med bags or at austere clinics to help maintain a sterile space for procedures, prevent infection while rendering care, and help with exacerbations or prevent flare-ups of chronic conditions such as psoriasis, acne, and atopic dermatitis while specialist care is pending.
Exogenously, hypochlorous acid (HOCl) is a powerful oxidizing agent formed from chlorine dissolved in water. Within the body, it is part of the immune response, created by activated leukocytes, which form HOCl from hydrogen peroxide and chloride. HOCl has been used as a disinfectant in wound care due to its antimicrobial properties via inhibition of DNA synthesis, protein synthesis, and decreased adenosine triphosphate production. It specifically targets bacteria by blocking bacterial cell wall synthesis and decreasing DNA replication.1
During the COVID-19 pandemic, HOCl was recommended by the US Environmental Protection Agency as a disinfectant.2 HOCl can be purchased from a supplier, though its major limitation is its shelf life. The main environmental factors affecting its stability are sunlight exposure, temperatures > 25 °C, and air exposure. HOCl is stable and most potent when the pH falls between 3.5 and 5.5.3 It is best stored in a cool, dark environment to maintain efficacy for 2 weeks. Rossi-Fedele et al found that when exposed to sunlight, chlorine reduction starts on day 4, whereas solutions kept in dark storage remained more stable, with this process starting after day 14.4
HOCl can also be made on-site via a machine, which ranges in price from a portable version costing < $200 to a large commercial option that can cost $7000 to $25,000. HOCl is produced by mixing noniodinated salt and water, and using electrolysis, which generally takes less than 10 minutes before it is ready for use.2 Given the cost and nonreusable nature of disinfecting wipes, HOCl may be more worthwhile for economic and disposal purposes in the long term.
Different concentrations of HOCl are readily available commercially. Because topical application of 1% HOCl may cause skin irritation, solutions with lower concentrations have been developed including Vashe (0.03% HOCl; SteadMed), PhaseOne (0.025% HOCl solution; IHT), OCuSOFT (0.02% HOCl; OCuSOFT), Bruder (0.02% HOCl; Bruder Healthcare), Acuicyn (0.01% HOCl solution in dilute saline; Sonoma Pharmaceuticals), and Avenova (0.01% HOCl solution; NovaBay Pharmaceuticals).5
Aside from its surface utility, HOCl has been researched for its beneficial effects on skin. HOCl has been shown to be helpful intraoperatively and postoperatively in improving adverse effects (AEs) after hair restoration, including erythema and pruritus, and in optimizing healing by reducing inflammation, likely due to its antimicrobial properties and ability to promote oxygenation.6 Bucko et al demonstrated that Microcyn scar gel (with HOCl) was a superior nonirritating, nontoxic method of not only improving scar appearance (vascularity, scar height, and pliability) but also reducing scar symptoms of pain and pruritus in comparison to 100% silicone scar gel (traditional application used to improve scarring).7 Zhang et al demonstrated that HOCl consistently improved symptom relief of blepharitis, including meibomian gland, eyelash, and eyelid redness, irritation, and appearance in comparison as well as were better tolerated in comparison to traditional recommendations of eyelid compresses and wash (tea tree oil, diluted baby shampoo, and topical antibiotics).8 In children with moderate to severe atopic dermatitis, Majewski et al compared a traditional bleach bath with a body wash containing hypochlorite (NaOCl; hypochlorous acid in alkaline aqueous solution). The body wash proved to be more convenient (showering vs 10-minute bath) and significantly improved symptoms while reducing the need for topical corticosteroids (common treatment modality for atopic dermatitis).9
The skin is the body’s primary defense against both dermatologic and respiratory infections. The face is especially vulnerable to microbes via airborne or environmental transmission, mechanical irritation, and touch. In the military environment, personal protective equipment (PPE) or uniform items may increase the risk of dermatologic conditions such as allergic or irritant dermatitis, infection, and friction blisters.
In a literature review of 312 dermatologic articles published between 2002 and 2022, Singal and Lipner found that among deployed soldiers serving in hot and dry climates, dermatitis and eczematous conditions were the most common, whereas bacterial and fungal conditions were most common in hot and humid settings. In the nondeployed setting, dermatitis and eczematous, acne, and fungal infections were the most common skin conditions. This is reflected by the unique circumstances that service members face at home and while deployed, when they may be more vulnerable to developing new or worsening chronic skin conditions depending on the environment (access to shelter, humid vs dry environments), and decreased access or time for hygiene (shared quarters at home in barracks or on deployment). Occupation-related conditions also play a large role in military dermatologic conditions.10
Dever et al noted the unique risks and exposures in the environment itself (plants, arthropods) as well as uniform items (protective gear) that carry an increased risk of friction irritation and dermatitis. Occupational exposures commonly associated with irritant contact dermatitis include alcohols, oils, fuel, disinfectants, and solvents. Chemicals in military uniforms themselves (eg, formaldehyde resins, disperse dyes, and chromate-containing dyes) also have the potential to cause allergic contact dermatitis, which can be challenging to address given the emphasis on uniformity and standards.11 PPE also may exacerbate rosacea and acne.
Some pathologies are associated specifically with bacteria, such as Cutibacterium acnes, as seen in acne vulgaris. Colonization of bacteria on the face may create biofilms that are difficult to detect, may be resistant to antibiotic therapy, and are implicated in other dermatologic conditions, such as persistent wounds, atopic dermatitis, and candidiasis.12
Biofilm and antibiotic resistance already pose a risk to patient care, but the unique environmental conditions and exposures of military settings can amplify this risk in the military population.13 Using HOCl in austere environments or the field for wound care may help reduce microbial load and the subsequent need for systemic antibiotics which carry the risk of gastrointestinal AEs and resistance.1
An optimized healing rate would support operational objectives by enabling service members to remain on full duty and avoid medications, which may prevent them from special duty, such as aviation. Sakarya et al found that HOCl solution enhanced wound healing in contrast with povidone-iodine (PI), while a study by Dharap et al discussed how HOCl provided major improvement in ulcer wound size (and infection), as well as significant reduction of inflammation.13
Anagnostopoulos et al studied the efficacy of 0.01% HOCl vs other disinfectants (5% PI, 4% chlorhexidine gluconate [CHG] and 70% isopropyl alcohol [IPA]) against common skin organisms, including methicillin-susceptible Staphylococcus aureus and methicillin-susceptible Staphylococcus epidermidis. The study found that HOCl had at least equal if not greater efficacy to PI, CHG, and IPA depending on the bacterial strain, demonstrating immediate bactericidal effects.14
Furthermore, HOCl has been shown to be useful in suturing and wound closure by reducing microbial load when soaked gauze is placed in wound beds prior to closure, while not harming surrounding tissue.15 This would be especially advantageous for military health care when specialist follow-up would be delayed or to prevent infection risk while en route to higher care. Aside from its disinfectant strength, it’s also well tolerated. HOCl studies on human tissue demonstrate its efficacy to prevent irritation and AEs while also preventing infection and promoting wound healing.
Gozukucuk and Cakiroglu studied the use of HOCl as a skin disinfectant before neonatal circumcision and demonstrated fewer adverse effects compared with the more commonly used PI. Neonates treated with PI prior to circumcision resulted in greater postoperative edema and increased duration of wound healing compared with infants treated with HOCl.16 Furthermore, studies have shown that PI can lead to irritant dermatitis or chemical burns if not properly dried or if it becomes pooled because of occlusion dressings.17
Aside from its indicated use for infection or wound care, anti-inflammatory properties of HOCl also may be beneficial for off-label use in preventing flareups of chronic conditions as well as for treating symptoms while awaiting specialist evaluation. This might be the case during US-based training exercises, in remote locations without nearby dermatologists, or during virtual care because of internet constraints. For chronic conditions such as rosacea or atopic dermatitis, which research has shown are related to mast cell activation and degranulation and cytokine release, HOCl has been shown to reduce histamine, neutrophil-generated leukotrienes, in addition to interleukin-6 and interleukin-2 to improve symptoms by reducing inflammation.18
Limitations of HOCl to explore would be extending its shelf life, exploring its various forms (eg, spray, topical) and storage limitations, and training of the machine and materials needed to be made in-house if not purchased. There are also no official guidelines for clinicians to recommend HOCl to patients, and research should be expanded on its use in humans, though it generally is well tolerated without AEs. HOCl has the potential to be a potent, nontoxic, inexpensive tool in med bags or at austere clinics to help maintain a sterile space for procedures, prevent infection while rendering care, and help with exacerbations or prevent flare-ups of chronic conditions such as psoriasis, acne, and atopic dermatitis while specialist care is pending.
- Natarelli N, et al. Hypochlorous acid: applications in dermatology. J Integr Dermatol. December 22, 2022. Accessed March 2, 2026. https://www.jintegrativederm.org/article/56663-hypochlorous-acid-applications-in-dermatology
- Block MS, Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg. 2020;78:1461-1466. doi:10.1016/j.joms.2020.06.029
- Menta N, Vidal SI, Friedman A. Hypochlorous acid: a blast from the past. J Drugs Dermatol. 2024;23:909-910.
- Rossi-Fedele G, Dogramaci E, Steier L, et al. Some factors influencing the stability of Sterilox®, a super-oxidised water. Br Dent J. 2011;210:E23. doi:10.1038/sj.bdj.2011.143
- Tran AQ, Topilow N, Rong A, et al. Comparison of skin antiseptic agents and the role of 0.01% hypochlorous acid. Aesthet Surg J. 2021;41:1170-1175. doi:10.1093/asj/sjaa322
- Stough D. Topical stabilized super-oxidized hypochlorous acid for wound healing in hair restoration surgery: a real-time usage-controlled trial evaluating safety, efficacy, and tolerability. J Drugs Dermatol. 2023;22:1191-1196. doi:10.36849/JDD.7172
- Bucko AD, Draelos Z, Dubois JC, Jones TM. A doubleblind, randomized study to compare Microcyn scar management hydrogel, K103163, and Kelo-cote scar gel for hypertrophic or keloid scars. Dermatologist. 2015;23:113-122.
- Zhang H, Wu Y, Wan X, et al. Effect of hypochlorous acid on blepharitis through ultrasonic atomization: a randomized clinical trial. J Clin Med. 2023;12(3):1164. doi:10.3390/jcm12031164
- Majewski S, Bhattacharya T, Asztalos M, et al. Sodium hypochlorite body wash in the management of Staphylococcus aureus-colonized moderate-to-severe atopic dermatitis in infants, children, and adolescents. Pediatr Dermatol. 2019;36:442-447. doi:10.1111/pde.13842
- Singal A, Lipner SR. A review of skin disease in military soldiers: challenges and potential solutions. Ann Med. 2023;55:2267425. doi:10.1080/07853890.2023.2267425
- Dever TT, Walters M, Jacob S. Contact dermatitis in military personnel. Dermatitis. 2011;22:313-319. doi:10.2310/6620.2011.11024
- Nowbuth AA, Armstrong J, Cloete T, et al. A potential benefit of hypochlorous acid-facial sanitisation: a review. Preprints. 2021. doi:10.20944/preprints202107.0129.v2
- Gold MH, Andriessen A, Bhatia AC, et al. Topical stabilized hypochlorous acid: the future gold standard for wound care and scar management in dermatologic and plastic surgery procedures. J Cosmet Dermatol. 2020;19:270-277. doi:10.1111/jocd.13280
- Anagnostopoulos AG, Rong A, Miller D, et al. 0.01% hypochlorous acid as an alternative skin antiseptic: an in vitro comparison. Dermatol Surg. 2018;44:1489-1493. doi:10.1097/DSS.0000000000001594
- Odom EB, Mundschenk MB, Hard KA, et al. The utility of hypochlorous acid wound therapy in wound bed preparation and skin graft salvage. Plast Reconstr Surg. 2019;143:677e-678e. doi:10.1097/PRS.0000000000005359
- Gozukucuk A, Cakiroglu B. Comparison of hypochlorous acid and povidone-iodine as a disinfectant in neonatal circumcision. J Pediatr Urol. 2022;18:341.e1-341.e5. doi:10.1016/j.jpurol.2022.03.011
- Borrego L, Hernández N, Hernández Z, et al. Povidoneiodine-induced postsurgical irritant contact dermatitis localized outside of the surgical incision area: report of 27 cases and a literature review. Int J Dermatol. 2016;55:540- 545. doi:10.1111/ijd.12957
- Del Rosso JQ, Bhatia N. Status report on topical hypochlorous acid: clinical relevance of specific formulations, potential modes of action, and study outcomes. J Clin Aesthet Dermatol. 2018;11:36-39.
- Natarelli N, et al. Hypochlorous acid: applications in dermatology. J Integr Dermatol. December 22, 2022. Accessed March 2, 2026. https://www.jintegrativederm.org/article/56663-hypochlorous-acid-applications-in-dermatology
- Block MS, Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg. 2020;78:1461-1466. doi:10.1016/j.joms.2020.06.029
- Menta N, Vidal SI, Friedman A. Hypochlorous acid: a blast from the past. J Drugs Dermatol. 2024;23:909-910.
- Rossi-Fedele G, Dogramaci E, Steier L, et al. Some factors influencing the stability of Sterilox®, a super-oxidised water. Br Dent J. 2011;210:E23. doi:10.1038/sj.bdj.2011.143
- Tran AQ, Topilow N, Rong A, et al. Comparison of skin antiseptic agents and the role of 0.01% hypochlorous acid. Aesthet Surg J. 2021;41:1170-1175. doi:10.1093/asj/sjaa322
- Stough D. Topical stabilized super-oxidized hypochlorous acid for wound healing in hair restoration surgery: a real-time usage-controlled trial evaluating safety, efficacy, and tolerability. J Drugs Dermatol. 2023;22:1191-1196. doi:10.36849/JDD.7172
- Bucko AD, Draelos Z, Dubois JC, Jones TM. A doubleblind, randomized study to compare Microcyn scar management hydrogel, K103163, and Kelo-cote scar gel for hypertrophic or keloid scars. Dermatologist. 2015;23:113-122.
- Zhang H, Wu Y, Wan X, et al. Effect of hypochlorous acid on blepharitis through ultrasonic atomization: a randomized clinical trial. J Clin Med. 2023;12(3):1164. doi:10.3390/jcm12031164
- Majewski S, Bhattacharya T, Asztalos M, et al. Sodium hypochlorite body wash in the management of Staphylococcus aureus-colonized moderate-to-severe atopic dermatitis in infants, children, and adolescents. Pediatr Dermatol. 2019;36:442-447. doi:10.1111/pde.13842
- Singal A, Lipner SR. A review of skin disease in military soldiers: challenges and potential solutions. Ann Med. 2023;55:2267425. doi:10.1080/07853890.2023.2267425
- Dever TT, Walters M, Jacob S. Contact dermatitis in military personnel. Dermatitis. 2011;22:313-319. doi:10.2310/6620.2011.11024
- Nowbuth AA, Armstrong J, Cloete T, et al. A potential benefit of hypochlorous acid-facial sanitisation: a review. Preprints. 2021. doi:10.20944/preprints202107.0129.v2
- Gold MH, Andriessen A, Bhatia AC, et al. Topical stabilized hypochlorous acid: the future gold standard for wound care and scar management in dermatologic and plastic surgery procedures. J Cosmet Dermatol. 2020;19:270-277. doi:10.1111/jocd.13280
- Anagnostopoulos AG, Rong A, Miller D, et al. 0.01% hypochlorous acid as an alternative skin antiseptic: an in vitro comparison. Dermatol Surg. 2018;44:1489-1493. doi:10.1097/DSS.0000000000001594
- Odom EB, Mundschenk MB, Hard KA, et al. The utility of hypochlorous acid wound therapy in wound bed preparation and skin graft salvage. Plast Reconstr Surg. 2019;143:677e-678e. doi:10.1097/PRS.0000000000005359
- Gozukucuk A, Cakiroglu B. Comparison of hypochlorous acid and povidone-iodine as a disinfectant in neonatal circumcision. J Pediatr Urol. 2022;18:341.e1-341.e5. doi:10.1016/j.jpurol.2022.03.011
- Borrego L, Hernández N, Hernández Z, et al. Povidoneiodine-induced postsurgical irritant contact dermatitis localized outside of the surgical incision area: report of 27 cases and a literature review. Int J Dermatol. 2016;55:540- 545. doi:10.1111/ijd.12957
- Del Rosso JQ, Bhatia N. Status report on topical hypochlorous acid: clinical relevance of specific formulations, potential modes of action, and study outcomes. J Clin Aesthet Dermatol. 2018;11:36-39.
Hypochlorous Acid: A Multipurpose New Addition to the Military Med Bag?
Hypochlorous Acid: A Multipurpose New Addition to the Military Med Bag?
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
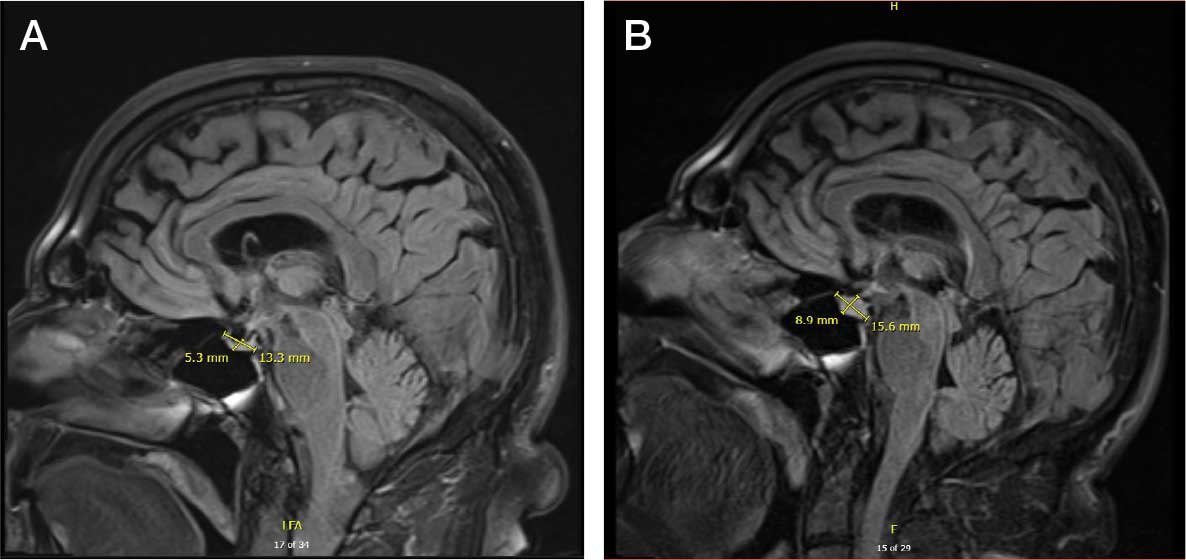
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).
The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.
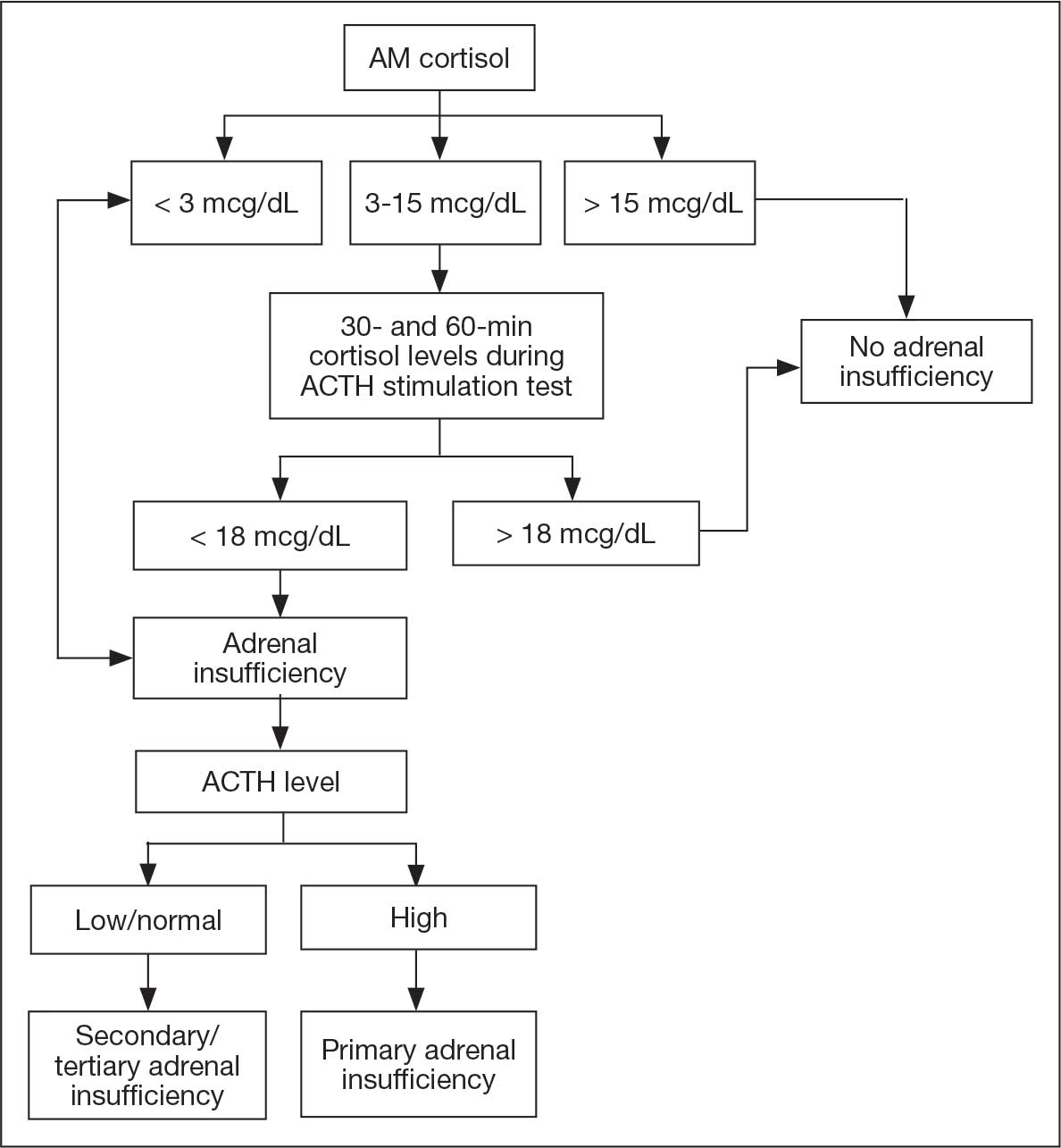
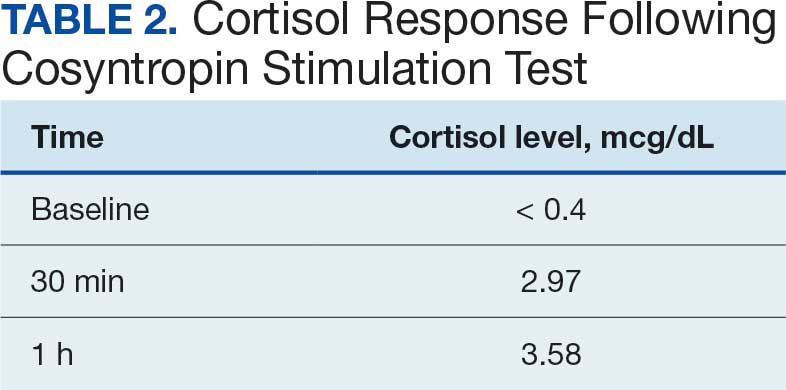
One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).
The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).

The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.

One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).

The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).

The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.

One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).

The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Recliner Butt
Recliner Butt
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
Recliner Butt
Recliner Butt
An 89-year-old male veteran with a history of obesity (body mass index, 33), osteoarthritis, anemia, pulmonary embolism, and urinary incontinence presented for evaluation of gluteal skin lesions (Figure). The patient had poor mobility and spent most of the day in a recliner chair. He also slept in the recliner due to chronic dyspnea and orthopnea.
The gluteal region demonstrated purplish discoloration with blanchable erythema and superficial ulcerations. The affected area was not pruritic and had remained unchanged for 3 months.
A punch biopsy of the discolored gluteal area was performed. Histopathologic examination revealed hyperkeratosis, orthokeratosis, irregular acanthosis, and mild spongiosis. Vascular proliferation and papillary dermal edema were also noted.
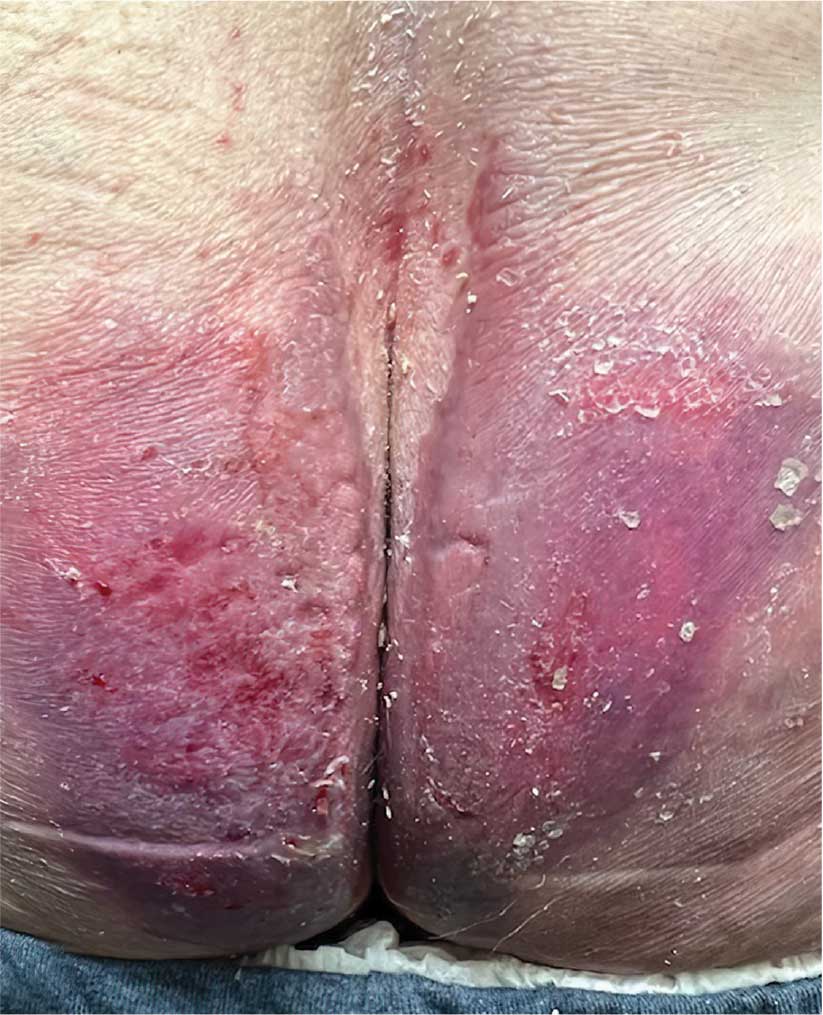
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Melanoma Leads Skin Cancer Malpractice Cases Over 95 Years
Melanoma Leads Skin Cancer Malpractice Cases Over 95 Years
TOPLINE:
In a review of physician-related malpractice cases from 1930 to 2025, melanoma was the most frequently litigated skin cancer, and failure or delay in diagnosis was the most common allegation, with documented death in nearly one third of cases.
METHODOLOGY:
Researchers conducted a review of physician-related medicolegal cases involving skin cancer using the LexisNexis legal database and identified 188 unique cases from 1930 through May 2025.
Cases were included if physicians were named as defendants and the litigation centered on diagnosis or management of a cutaneous malignancy.
Study outcomes examined case characteristics including cancer type, practice setting, defendant specialty, primary allegations, clinical outcomes, and case verdicts across the US.
TAKEAWAY:
Melanoma accounted for 49.5% of litigated cases, followed by squamous cell carcinoma (21.6%), basal cell carcinoma (14.2%), unspecified skin cancer (11.6%), and other rare tumors (3.1%). Death was reported in 29.8% of cases and metastatic disease in 39.9%.
Failure or delay in diagnosis was the leading allegation (38.1%), followed by treatment or management errors (24.2%), misdiagnosis (11.4%), “deliberate indifference” (8.3%), inadequate informed consent (7.5%), and pathology-related errors (7.2%).
Family physicians were the most common defendants (27.5%), followed by dermatologists, including Mohs surgeons (20.1%), and pathologists or dermatopathologists (14.4%), followed by general or plastic surgeons (7.9%), and internists (4.4%). Most cases originated in private practices (59.7%), and New York (16.0%) and California (13.3%) were the states with the most cases.
Among 109 closed cases, 5.5% resulted in plaintiff verdicts, whereas defense verdicts predominated in 55.0%. Plaintiff awards ranged from $10,000 to $4.25 million.
IN PRACTICE:
“This comprehensive review demonstrates that melanoma is the most frequently litigated skin cancer, particularly in cases involving metastatic disease or death, and that family physicians are the most commonly named defendants overall,” the authors wrote. “By examining both allegations and outcomes,” they added, “this analysis provides a pragmatic assessment of real-world litigation exposure and the clinical scenarios that expose physicians to legal proceedings, financial cost, reputational harm, and psychological burden, regardless of case disposition.”
SOURCE:
The study was led by Ghassan Barnawi, MD, Division of Dermatology, McGill University in Montreal, Quebec, Canada, and was published online on February 20, 2026, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study relied on published court decisions, which likely underestimated malpractice burden by excluding settlements and unreported claims.
DISCLOSURES:
The study did not receive any funding. The authors reported having no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
The study had no commercial funding. The authors had no relevant disclosures.
A version of this article first appeared on Medscape.com
TOPLINE:
In a review of physician-related malpractice cases from 1930 to 2025, melanoma was the most frequently litigated skin cancer, and failure or delay in diagnosis was the most common allegation, with documented death in nearly one third of cases.
METHODOLOGY:
Researchers conducted a review of physician-related medicolegal cases involving skin cancer using the LexisNexis legal database and identified 188 unique cases from 1930 through May 2025.
Cases were included if physicians were named as defendants and the litigation centered on diagnosis or management of a cutaneous malignancy.
Study outcomes examined case characteristics including cancer type, practice setting, defendant specialty, primary allegations, clinical outcomes, and case verdicts across the US.
TAKEAWAY:
Melanoma accounted for 49.5% of litigated cases, followed by squamous cell carcinoma (21.6%), basal cell carcinoma (14.2%), unspecified skin cancer (11.6%), and other rare tumors (3.1%). Death was reported in 29.8% of cases and metastatic disease in 39.9%.
Failure or delay in diagnosis was the leading allegation (38.1%), followed by treatment or management errors (24.2%), misdiagnosis (11.4%), “deliberate indifference” (8.3%), inadequate informed consent (7.5%), and pathology-related errors (7.2%).
Family physicians were the most common defendants (27.5%), followed by dermatologists, including Mohs surgeons (20.1%), and pathologists or dermatopathologists (14.4%), followed by general or plastic surgeons (7.9%), and internists (4.4%). Most cases originated in private practices (59.7%), and New York (16.0%) and California (13.3%) were the states with the most cases.
Among 109 closed cases, 5.5% resulted in plaintiff verdicts, whereas defense verdicts predominated in 55.0%. Plaintiff awards ranged from $10,000 to $4.25 million.
IN PRACTICE:
“This comprehensive review demonstrates that melanoma is the most frequently litigated skin cancer, particularly in cases involving metastatic disease or death, and that family physicians are the most commonly named defendants overall,” the authors wrote. “By examining both allegations and outcomes,” they added, “this analysis provides a pragmatic assessment of real-world litigation exposure and the clinical scenarios that expose physicians to legal proceedings, financial cost, reputational harm, and psychological burden, regardless of case disposition.”
SOURCE:
The study was led by Ghassan Barnawi, MD, Division of Dermatology, McGill University in Montreal, Quebec, Canada, and was published online on February 20, 2026, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study relied on published court decisions, which likely underestimated malpractice burden by excluding settlements and unreported claims.
DISCLOSURES:
The study did not receive any funding. The authors reported having no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
The study had no commercial funding. The authors had no relevant disclosures.
A version of this article first appeared on Medscape.com
TOPLINE:
In a review of physician-related malpractice cases from 1930 to 2025, melanoma was the most frequently litigated skin cancer, and failure or delay in diagnosis was the most common allegation, with documented death in nearly one third of cases.
METHODOLOGY:
Researchers conducted a review of physician-related medicolegal cases involving skin cancer using the LexisNexis legal database and identified 188 unique cases from 1930 through May 2025.
Cases were included if physicians were named as defendants and the litigation centered on diagnosis or management of a cutaneous malignancy.
Study outcomes examined case characteristics including cancer type, practice setting, defendant specialty, primary allegations, clinical outcomes, and case verdicts across the US.
TAKEAWAY:
Melanoma accounted for 49.5% of litigated cases, followed by squamous cell carcinoma (21.6%), basal cell carcinoma (14.2%), unspecified skin cancer (11.6%), and other rare tumors (3.1%). Death was reported in 29.8% of cases and metastatic disease in 39.9%.
Failure or delay in diagnosis was the leading allegation (38.1%), followed by treatment or management errors (24.2%), misdiagnosis (11.4%), “deliberate indifference” (8.3%), inadequate informed consent (7.5%), and pathology-related errors (7.2%).
Family physicians were the most common defendants (27.5%), followed by dermatologists, including Mohs surgeons (20.1%), and pathologists or dermatopathologists (14.4%), followed by general or plastic surgeons (7.9%), and internists (4.4%). Most cases originated in private practices (59.7%), and New York (16.0%) and California (13.3%) were the states with the most cases.
Among 109 closed cases, 5.5% resulted in plaintiff verdicts, whereas defense verdicts predominated in 55.0%. Plaintiff awards ranged from $10,000 to $4.25 million.
IN PRACTICE:
“This comprehensive review demonstrates that melanoma is the most frequently litigated skin cancer, particularly in cases involving metastatic disease or death, and that family physicians are the most commonly named defendants overall,” the authors wrote. “By examining both allegations and outcomes,” they added, “this analysis provides a pragmatic assessment of real-world litigation exposure and the clinical scenarios that expose physicians to legal proceedings, financial cost, reputational harm, and psychological burden, regardless of case disposition.”
SOURCE:
The study was led by Ghassan Barnawi, MD, Division of Dermatology, McGill University in Montreal, Quebec, Canada, and was published online on February 20, 2026, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study relied on published court decisions, which likely underestimated malpractice burden by excluding settlements and unreported claims.
DISCLOSURES:
The study did not receive any funding. The authors reported having no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
The study had no commercial funding. The authors had no relevant disclosures.
A version of this article first appeared on Medscape.com
Melanoma Leads Skin Cancer Malpractice Cases Over 95 Years
Melanoma Leads Skin Cancer Malpractice Cases Over 95 Years
Cross-Sectional Analysis of Biologic Use in the Treatment of Veterans With Hidradenitis Suppurativa
Cross-Sectional Analysis of Biologic Use in the Treatment of Veterans With Hidradenitis Suppurativa
Hidradenitis suppurativa (HS) is a chronic, inflammatory skin disorder characterized by painful nodules, abscesses, and tunnels predominantly affecting intertriginous areas of the body.1,2 The condition poses significant challenges in terms of diagnosis, treatment, and quality of life for affected individuals. Various systemic therapies have been explored to manage this debilitating condition, with the emergence of biologic agents offering hope for improved outcomes. In 2015, adalimumab (ADA) was the first biologic approved by the US Food and Drug Administration (FDA) for the treatment of HS, followed by secukinumab in 2023 and bimekizumab in 2024. However, the off-label use of other biologics and/or tumor necrosis factor inhibitors such as infliximab (IFX) has become common practice.3
Although these therapies have demonstrated promising results in the treatment of HS, their widespread use may be hindered by accessibility and cost barriers. Orenstein et al analyzed data from the IBM Explorys platform from 2015 to 2020 and found that only 1.8% of patients diagnosed with HS had been prescribed ADA or IFX.4 More recently, Garg et al examined IBM MarketScan and IBM US Medicaid data from 2015 to 2018 to evaluate trends in clinical care and treatment. The prevalence of ADA and IFX prescriptions among patients with HS ranged from 2.3% to 8.0% (ADA) and 0.7% to 0.9% (IFX) for patients with commercial insurance, and 1.4% to 4.8% (ADA) and 0.5% to 0.7% (IFX) for patients with Medicaid.5 Biologics are often expensive, and the high cost associated with these therapies has been identified as a significant barrier to access for patients with HS, particularly those who lack adequate insurance coverage or face financial constraints.6
Furthermore, these barriers, particularly the financial barriers, are potentially compounded by the demographics of patients most notably affected by HS. In the US, a disproportionate incidence of HS has been noted in specific groups and age ranges, including women, individuals aged 18 to 29 years, and Black individuals.4 Orenstein et al found a statistically significant difference in use of ADA and IFX biologics based on age, sex, and race.4
The aim of this study was to examine the use of 2 biologics (ADA and IFX) in the Veterans Health Administration (VHA), a unique population in which financial barriers are reduced due to the single-payer government health care system structure. This design allowed for improved isolation and evaluation of variation in ADA and/or IFX prescription rates by demographics and health-related factors among patients with HS. To our knowledge, no studies have analyzed these metrics within the VHA.
Methods
This retrospective, cross-sectional analysis of VHA patients used data from the US Department of Veterans Affairs (VA) Corporate Data Warehouse, a data repository that provides access to longitudinal national electronic health record data for all veterans receiving care through VHA facilities. This study received ethical approval from institutional review boards at the Minneapolis Veterans Affairs Health Care System and VA Salt Lake City Healthcare System. Patient information was deidentified, and patient consent was not required.
Patients with HS were identified using ≥ 1 International Classification of Diseases (ICD) diagnostic code: (ICD-9 [705.83] or ICD-10 [L73.2]) between January 1, 2011, and December 31, 2021. The study included patients aged ≥ 18 years as of January 1, 2011, with ≥ 2 patient encounters during the postdiagnosis follow-up period, and with ≥ 1 encounter 6 months postindex. Patients with a biologic prescription prior to HS diagnosis were excluded. For this study, the term biologics refers to ADA and/or IFX prescriptions, unless otherwise specified. Only ADA and IFX were included in this analysis because ADA, a tumor necrosis factor (TNF)-á inhibitor, was the only FDA-approved medication at the time of the search, and IFX is another common TNF-α inhibitor used for the treatment of HS.
Statistical Analysis
We calculated logistic regression using SAS 9.4 (SAS Institute, Cary, NC). For each variable, the univariate relationship with biologic prescriptions was examined first, followed by the multivariate relationship controlling for all other variables. The following variables were controlled for in the multivariate models and were chosen a priori: sex, age, race, ethnicity, US region, hospital setting, current or previous tobacco use, obesity (defined as body mass index [BMI] ≥ 30), and Charlson Comorbidity Index (CCI).7
Results
Using ICD codes, we identified 29,483 individuals with ≥ 1 HS diagnosis (Figure 1). Of those identified, 1537 patients (5.21%) had been prescribed ≥ 1 biologic. The cohort was predominantly White (60.56%), male (75.27%), obese (59.34%), and had a history of current or previous tobacco use (73.47%) (Table 1). There were significant adjusted differences in prescription rates among veterans with HS based on age, race, and BMI. Notably, there was an age-dependent reduction in the odds of being prescribed a biologic in patients with HS. Compared with patients aged 18 to 44 years, patients aged 45 to 64 years (adjusted odds ratio [aOR], 0.63; 95% CI, 0.54–0.74; P < .001) and patients aged ≥ 65 years (aOR, 0.36; 95% CI, 0.27–0.48; P < .001) had significantly lower odds of receiving a biologic prescription (Table 2). Compared with White patients with HS, Native Hawaiian (NH) or Pacific Islander (PI) patients were less likely to be prescribed a biologic (aOR, 0.23; 95% CI, 0.06–0.92; P = .04). Patients with obesity had significantly higher odds of receiving a biologic prescription compared with patients without obesity (aOR, 1.47; 95% CI, 1.27– 1.71; P < .001).
Included in Analysis.
After adjusting for the variables listed in Table 1, there were no significant differences in biologic prescription rates for men compared with women (aOR, 0.97; 95% CI, 0.83-1.12; P = .68). We observed slight variations in biologic prescriptions between US regions (Midwest 5.0%, East 4.2%, South 5.8%, West 4.6%), none of which were significantly different in the fully adjusted model. No statistically significant differences were found in biologic prescriptions between urban and rural VA settings (5.4% vs 4.8%; aOR, 1.06; 95% CI, 0.90–1.24; P = .47). Tobacco use was not associated with the rate of biologic prescription receipt (aOR, 1.14; 95% CI, 0.97–1.34; P = .11). After adjusting for other variables (as outlined in Table 2), no significant differences were found between CCI of 0 and 1 (aOR, 0.97; 95% CI, 0.82–1.16; P = .77) or between CCI of 0 and 2 (aOR, 0.89; 95% CI, 0.74–1.07; P = .22).7
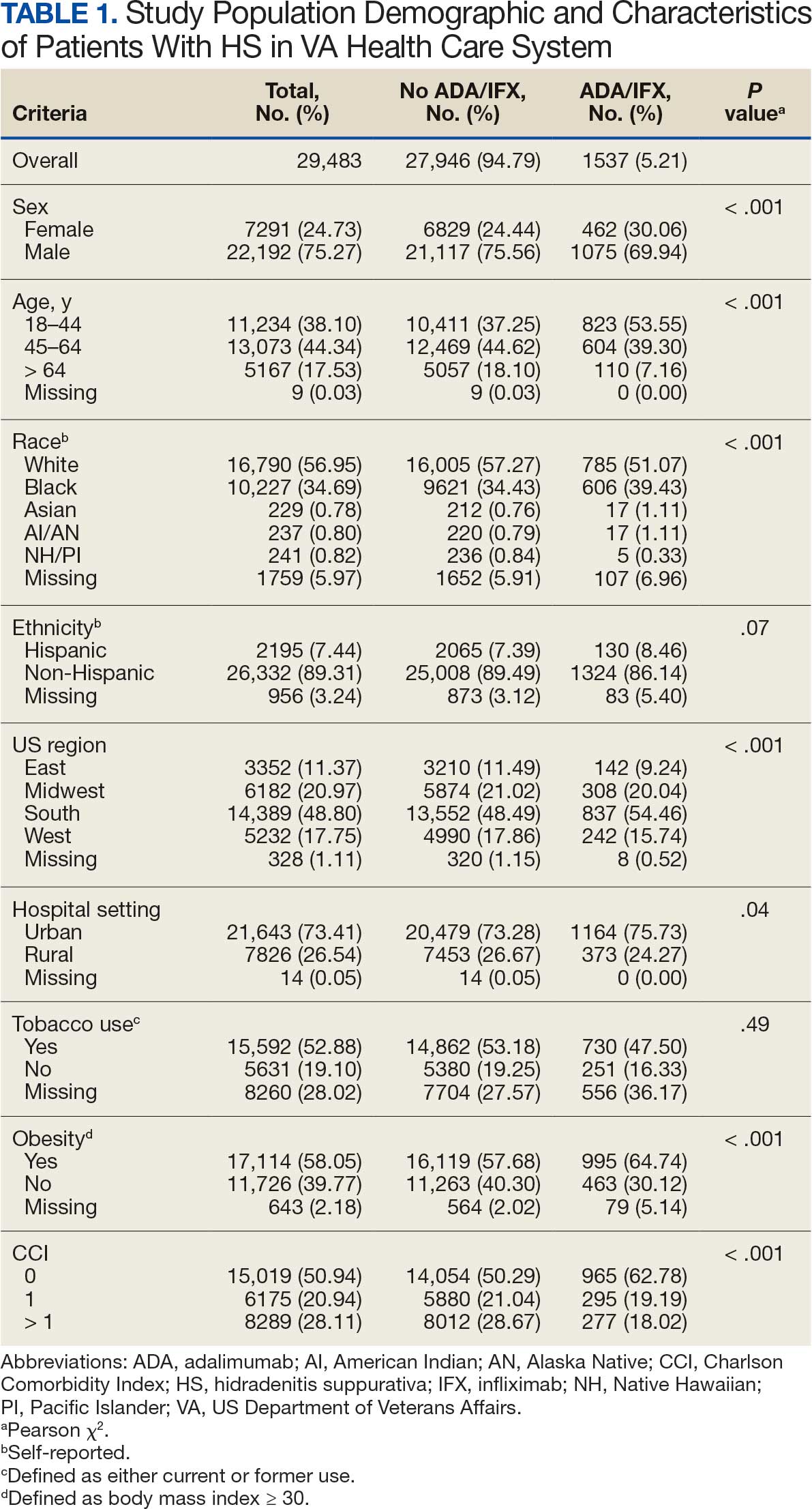
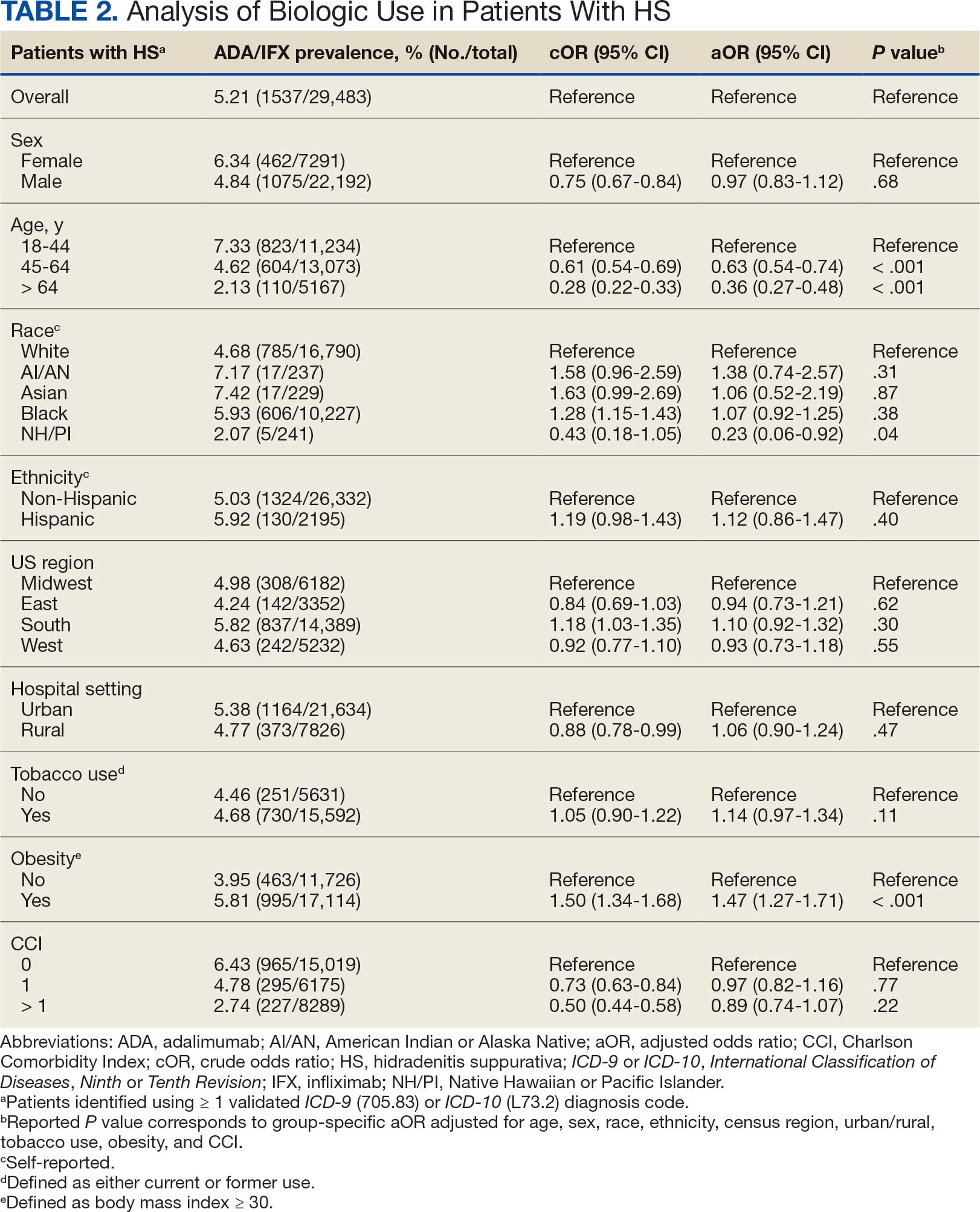
Discussion
The aim of the study was to ascertain potential discrepancies in biologic prescription patterns among patients with HS in the VHA by demographic and lifestyle behavior modifiers. Veteran cohorts are unique in composition, consisting predominantly of older White men within a single-payer health care system. The prevalence of biologic prescriptions in this population was low (5.2%), consistent with prior studies (1.8%–8.9%).4,5
We found a significant difference in ADA/IFX prescription patterns between White patients and NH/PI patients (aOR, 0.23; 95% CI, 0.06-0.92; P = .04). Further replication of this result is needed due to the small number of NH/PI patients included in the study (n = 241). Notably, we did not find a significant difference in the odds of Black patients being prescribed a biologic compared with White patients (aOR, 1.07; 95% CI, 0.92–1.25; P = .38), consistent with prior studies.4
In line with prior studies, age was associated with the likelihood of receiving a biologic prescription.4 Using the multivariate model adjusting for variables listed in Table 1, including CCI, patients aged 45 to 64 years and > 64 years were less likely to be prescribed a biologic than patients aged 18 to 44 years. HS disease activity could be a potential confounding variable, as HS severity may subside in some people with increasing age or menopause.8
Because different regions in the US have different sociopolitical ideologies and governing legislation, we hypothesized that there may be dissimilarities in the prevalence rates of biologic prescribing across various US regions. However, no significant differences were found in prescription patterns among US regions or between rural and urban settings. Previous research has demonstrated discernible disparities in both dermatologic care and clinical outcomes based on hospital setting (ie, urban vs rural).9-11
Tobacco use has been demonstrated to be associated with the development of HS.12 In a large retrospective analysis, Garg et al reported increased odds of receiving a new HS diagnosis in known tobacco users (aOR, 1.9; 95% CI, 1.8–2.0).13 The extent to which tobacco use affects HS severity is less understood. While some studies have found an association between smoking and HS severity, other analyses have failed to find this association.14,15 The effects of smoking cessation on the disease course of HS are unknown.16 This analysis, found no significant difference in prescriptions for biologics among patients with HS comparing current or previous tobacco users with nonusers.
There is a known positive correlation between increasing BMI and HS prevalence and severity that may be explained by the downstream effects of adipose tissue secretion of proinflammatory mediators and insulin resistance in the setting of chronic inflammation.12 This analysis found that patients with HS and obesity were 1.47 times more likely to be prescribed a biologic than patients with HS without obesity, which may be confounded by increased HS severity among patients with obesity. The initial concern when analyzing tobacco use and obesity was that clinician bias may result in a decrease in the prevalence of biologic use in these demographics, which was not supported in this study.
Although we identified few disparities, the results demonstrated a substantial underutilization of biologic therapies (5.2%), similar to the other US civilian studies (1.8-8.9%).4,5 While there is no current universal, standardized severity scoring system to evaluate HS (it is difficult to objectively define moderate to severe HS), estimates have shown that 40.3% to 65.8% of patients with HS have Hurley stage II or III.17-19 Therefore, only a small percentage of patients with moderate to severe disease were prescribed the only FDA-approved medication during this time period. The persistence of this underutilization within a medical system that reduces financial barriers suggests that nonfinancial barriers have a notable role in the underutilization of biologics.
For instance, risk of adverse events, particularly lymphoma and infection, has been cited by patients as a reason to avoid biologics. Additionally, treatment fatigue reduced some patients’ willingness to try new treatments, as did lack of knowledge about treatment options.6,20 Other reported barriers included the frequency of injections and fear of needles.6 Additionally, within the VA, ADA may require prior authorization at the local facility level.21 An established relationship with a dermatologist has been shown to significantly increase the odds of being prescribed a biologic medication in the face of these barriers.4 Future system-wide quality improvement initiatives could be implemented to identify patients with HS not followed by dermatology, with the goal of establishing care with a dermatologist.
Limitations
Limitations to this study include an inability to categorize HS disease severity and assess the degree to which disease severity confounded study findings, particularly in relation to tobacco use and obesity. The generalizability of this study is also limited because of the demographic characteristics of the veteran patient population, which is predominantly older, White, and male, whereas HS disproportionately affects younger, Black, and female individuals in the US.22 Despite these limitations, this study contributes valuable insights into the use of biologic therapies for veteran populations with HS using a national dataset.
Conclusions
This study was performed within a single-payer government medical system, likely reducing or removing the financial barriers that some patient populations may face when pursuing biologics for HS treatment. However, the prevalence of biologic use in this population was low overall (5.2%), suggesting that other factors play a role in the underutilization of biologics in HS. Consistent with previous studies, younger individuals were more likely to be prescribed a biologic, and no difference in prescription rates between Black and White patients was observed. Unlike previous studies, no significant difference in prescription rates between men and women was observed.
- Goldburg SR, Strober BE, Payette MJ. Hidradenitis suppurativa: epidemiology, clinical presentation, and pathogenesis. J Am Acad Dermatol. 2020;82:1045-1058. doi:10.1016/j.jaad.2019.08.090
- Tchero H, Herlin C, Bekara F, et al. Hidradenitis suppurativa: a systematic review and meta-analysis of therapeutic interventions. Indian J Dermatol Venereol Leprol. 2019;85:248-257. doi:10.4103/ijdvl.IJDVL_69_18
- Shih T, Lee K, Grogan T, et al. Infliximab in hidradenitis suppurativa: a systematic review and meta-analysis. Dermatol Ther. 2022;35:e15691. doi:10.1111/dth.15691
- Orenstein LAV, Wright S, Strunk A, et al. Low prescription of tumor necrosis alpha inhibitors in hidradenitis suppurativa: a cross-sectional analysis. J Am Acad Dermatol. 2021;84:1399-1401. doi:10.1016/j.jaad.2020.07.108
- Garg A, Naik HB, Alavi A, et al. Real-world findings on the characteristics and treatment exposures of patients with hidradenitis suppurativa from US claims data. Dermatol Ther (Heidelb). 2023;13:581-594. doi:10.1007/s13555-022-00872-1
- De DR, Shih T, Fixsen D, et al. Biologic use in hidradenitis suppurativa: patient perspectives and barriers. J Dermatolog Treat. 2022;33:3060-3062. doi:10.1080/09546634.2022.2089336
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- von der Werth JM, Williams HC. The natural history of hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2000;14:389-392. doi:10.1046/j.1468-3083.2000.00087.x
- Silverberg JI, Barbarot S, Gadkari A, et al. Atopic dermatitis in the pediatric population: a cross-sectional, international epidemiologic study. Ann Allergy Asthma Immunol. 2021;126:417-428.e2. doi:10.1016/j.anai.2020.12.020
- Wu YP, Parsons B, Jo Y, et al. Outdoor activities and sunburn among urban and rural families in a Western region of the US: implications for skin cancer prevention. Prev Med Rep. 2022;29:101914. doi:10.1016/j.pmedr.2022.101914
- Mannschreck DB, Li X, Okoye G. Rural melanoma patients in Maryland do not present with more advanced disease than urban patients. Dermatol Online J. 2021;27. doi:10.5070/D327553607
- Garg A, Malviya N, Strunk A, et al. Comorbidity screening in hidradenitis suppurativa: evidence-based recommendations from the US and Canadian Hidradenitis Suppurativa Foundations. J Am Acad Dermatol. 2022;86:1092-1101. doi:10.1016/j.jaad.2021.01.059
- Garg A, Papagermanos V, Midura M, et al. Incidence of hidradenitis suppurativa among tobacco smokers: a population- based retrospective analysis in the U.S.A. Br J Dermatol. 2018;178:709-714. doi:10.1111/bjd.15939
- Sartorius K, Emtestam L, Jemec GBE, et al. Objective scoring of hidradenitis suppurativa reflecting the role of tobacco smoking and obesity. Br J Dermatol. 2009;161:831- 839. doi:10.1111/j.1365-2133.2009.09198.x
- Canoui-Poitrine F, Revuz JE, Wolkenstein P, et al. Clinical characteristics of a series of 302 French patients with hidradenitis suppurativa, with an analysis of factors associated with disease severity. J Am Acad Dermatol. 2009;61:51-57. doi:10.1016/j.jaad.2009.02.013
- Dufour DN, Emtestam L, Jemec GB. Hidradenitis suppurativa: a common and burdensome, yet under-recognised, inflammatory skin disease. Postgrad Med J. 2014;90:216- 221. doi:10.1136/postgradmedj-2013-131994
- Vazquez BG, Alikhan A, Weaver AL, et al. Incidence of hidradenitis suppurativa and associated factors: a population- based study of Olmsted County, Minnesota. J Invest Dermatol. 2013;133:97-103. doi:10.1038/jid.2012.255
- Vanlaerhoven AMJD, Ardon CB, van Straalen KR, et al. Hurley III hidradenitis suppurativa has an aggressive disease course. Dermatology. 2018;234:232-233. doi:10.1159/000491547
- Shahi V, Alikhan A, Vazquez BG, et al. Prevalence of hidradenitis suppurativa: a population-based study in Olmsted County, Minnesota. Dermatology. 2014;229:154-158. doi:10.1159/000363381
- Salame N, Sow YN, Siira MR, et al. Factors affecting treatment selection among patients with hidradenitis suppurativa. JAMA Dermatol. 2024;160:179. doi:10.1001/jamadermatol.2023.5425
- VA Formulary Advisor: ADALIMUMAB-BWWD INJ,SOLN. US Department of Veterans Affairs. Updated December 17, 2025. Accessed January 15, 2026. https://www.va.gov/formularyadvisor/drugs/4042383-ADALIMUMAB-BWWD-INJ-SOLN
- Garg A, Lavian J, Lin G, et al. Incidence of hidradenitis suppurativa in the United States: a sex- and age-adjusted population analysis. J Am Acad Dermatol. 2017;77:118- 122. doi:10.1016/j.jaad.2017.02.005
Hidradenitis suppurativa (HS) is a chronic, inflammatory skin disorder characterized by painful nodules, abscesses, and tunnels predominantly affecting intertriginous areas of the body.1,2 The condition poses significant challenges in terms of diagnosis, treatment, and quality of life for affected individuals. Various systemic therapies have been explored to manage this debilitating condition, with the emergence of biologic agents offering hope for improved outcomes. In 2015, adalimumab (ADA) was the first biologic approved by the US Food and Drug Administration (FDA) for the treatment of HS, followed by secukinumab in 2023 and bimekizumab in 2024. However, the off-label use of other biologics and/or tumor necrosis factor inhibitors such as infliximab (IFX) has become common practice.3
Although these therapies have demonstrated promising results in the treatment of HS, their widespread use may be hindered by accessibility and cost barriers. Orenstein et al analyzed data from the IBM Explorys platform from 2015 to 2020 and found that only 1.8% of patients diagnosed with HS had been prescribed ADA or IFX.4 More recently, Garg et al examined IBM MarketScan and IBM US Medicaid data from 2015 to 2018 to evaluate trends in clinical care and treatment. The prevalence of ADA and IFX prescriptions among patients with HS ranged from 2.3% to 8.0% (ADA) and 0.7% to 0.9% (IFX) for patients with commercial insurance, and 1.4% to 4.8% (ADA) and 0.5% to 0.7% (IFX) for patients with Medicaid.5 Biologics are often expensive, and the high cost associated with these therapies has been identified as a significant barrier to access for patients with HS, particularly those who lack adequate insurance coverage or face financial constraints.6
Furthermore, these barriers, particularly the financial barriers, are potentially compounded by the demographics of patients most notably affected by HS. In the US, a disproportionate incidence of HS has been noted in specific groups and age ranges, including women, individuals aged 18 to 29 years, and Black individuals.4 Orenstein et al found a statistically significant difference in use of ADA and IFX biologics based on age, sex, and race.4
The aim of this study was to examine the use of 2 biologics (ADA and IFX) in the Veterans Health Administration (VHA), a unique population in which financial barriers are reduced due to the single-payer government health care system structure. This design allowed for improved isolation and evaluation of variation in ADA and/or IFX prescription rates by demographics and health-related factors among patients with HS. To our knowledge, no studies have analyzed these metrics within the VHA.
Methods
This retrospective, cross-sectional analysis of VHA patients used data from the US Department of Veterans Affairs (VA) Corporate Data Warehouse, a data repository that provides access to longitudinal national electronic health record data for all veterans receiving care through VHA facilities. This study received ethical approval from institutional review boards at the Minneapolis Veterans Affairs Health Care System and VA Salt Lake City Healthcare System. Patient information was deidentified, and patient consent was not required.
Patients with HS were identified using ≥ 1 International Classification of Diseases (ICD) diagnostic code: (ICD-9 [705.83] or ICD-10 [L73.2]) between January 1, 2011, and December 31, 2021. The study included patients aged ≥ 18 years as of January 1, 2011, with ≥ 2 patient encounters during the postdiagnosis follow-up period, and with ≥ 1 encounter 6 months postindex. Patients with a biologic prescription prior to HS diagnosis were excluded. For this study, the term biologics refers to ADA and/or IFX prescriptions, unless otherwise specified. Only ADA and IFX were included in this analysis because ADA, a tumor necrosis factor (TNF)-á inhibitor, was the only FDA-approved medication at the time of the search, and IFX is another common TNF-α inhibitor used for the treatment of HS.
Statistical Analysis
We calculated logistic regression using SAS 9.4 (SAS Institute, Cary, NC). For each variable, the univariate relationship with biologic prescriptions was examined first, followed by the multivariate relationship controlling for all other variables. The following variables were controlled for in the multivariate models and were chosen a priori: sex, age, race, ethnicity, US region, hospital setting, current or previous tobacco use, obesity (defined as body mass index [BMI] ≥ 30), and Charlson Comorbidity Index (CCI).7
Results
Using ICD codes, we identified 29,483 individuals with ≥ 1 HS diagnosis (Figure 1). Of those identified, 1537 patients (5.21%) had been prescribed ≥ 1 biologic. The cohort was predominantly White (60.56%), male (75.27%), obese (59.34%), and had a history of current or previous tobacco use (73.47%) (Table 1). There were significant adjusted differences in prescription rates among veterans with HS based on age, race, and BMI. Notably, there was an age-dependent reduction in the odds of being prescribed a biologic in patients with HS. Compared with patients aged 18 to 44 years, patients aged 45 to 64 years (adjusted odds ratio [aOR], 0.63; 95% CI, 0.54–0.74; P < .001) and patients aged ≥ 65 years (aOR, 0.36; 95% CI, 0.27–0.48; P < .001) had significantly lower odds of receiving a biologic prescription (Table 2). Compared with White patients with HS, Native Hawaiian (NH) or Pacific Islander (PI) patients were less likely to be prescribed a biologic (aOR, 0.23; 95% CI, 0.06–0.92; P = .04). Patients with obesity had significantly higher odds of receiving a biologic prescription compared with patients without obesity (aOR, 1.47; 95% CI, 1.27– 1.71; P < .001).

Included in Analysis.
After adjusting for the variables listed in Table 1, there were no significant differences in biologic prescription rates for men compared with women (aOR, 0.97; 95% CI, 0.83-1.12; P = .68). We observed slight variations in biologic prescriptions between US regions (Midwest 5.0%, East 4.2%, South 5.8%, West 4.6%), none of which were significantly different in the fully adjusted model. No statistically significant differences were found in biologic prescriptions between urban and rural VA settings (5.4% vs 4.8%; aOR, 1.06; 95% CI, 0.90–1.24; P = .47). Tobacco use was not associated with the rate of biologic prescription receipt (aOR, 1.14; 95% CI, 0.97–1.34; P = .11). After adjusting for other variables (as outlined in Table 2), no significant differences were found between CCI of 0 and 1 (aOR, 0.97; 95% CI, 0.82–1.16; P = .77) or between CCI of 0 and 2 (aOR, 0.89; 95% CI, 0.74–1.07; P = .22).7


Discussion
The aim of the study was to ascertain potential discrepancies in biologic prescription patterns among patients with HS in the VHA by demographic and lifestyle behavior modifiers. Veteran cohorts are unique in composition, consisting predominantly of older White men within a single-payer health care system. The prevalence of biologic prescriptions in this population was low (5.2%), consistent with prior studies (1.8%–8.9%).4,5
We found a significant difference in ADA/IFX prescription patterns between White patients and NH/PI patients (aOR, 0.23; 95% CI, 0.06-0.92; P = .04). Further replication of this result is needed due to the small number of NH/PI patients included in the study (n = 241). Notably, we did not find a significant difference in the odds of Black patients being prescribed a biologic compared with White patients (aOR, 1.07; 95% CI, 0.92–1.25; P = .38), consistent with prior studies.4
In line with prior studies, age was associated with the likelihood of receiving a biologic prescription.4 Using the multivariate model adjusting for variables listed in Table 1, including CCI, patients aged 45 to 64 years and > 64 years were less likely to be prescribed a biologic than patients aged 18 to 44 years. HS disease activity could be a potential confounding variable, as HS severity may subside in some people with increasing age or menopause.8
Because different regions in the US have different sociopolitical ideologies and governing legislation, we hypothesized that there may be dissimilarities in the prevalence rates of biologic prescribing across various US regions. However, no significant differences were found in prescription patterns among US regions or between rural and urban settings. Previous research has demonstrated discernible disparities in both dermatologic care and clinical outcomes based on hospital setting (ie, urban vs rural).9-11
Tobacco use has been demonstrated to be associated with the development of HS.12 In a large retrospective analysis, Garg et al reported increased odds of receiving a new HS diagnosis in known tobacco users (aOR, 1.9; 95% CI, 1.8–2.0).13 The extent to which tobacco use affects HS severity is less understood. While some studies have found an association between smoking and HS severity, other analyses have failed to find this association.14,15 The effects of smoking cessation on the disease course of HS are unknown.16 This analysis, found no significant difference in prescriptions for biologics among patients with HS comparing current or previous tobacco users with nonusers.
There is a known positive correlation between increasing BMI and HS prevalence and severity that may be explained by the downstream effects of adipose tissue secretion of proinflammatory mediators and insulin resistance in the setting of chronic inflammation.12 This analysis found that patients with HS and obesity were 1.47 times more likely to be prescribed a biologic than patients with HS without obesity, which may be confounded by increased HS severity among patients with obesity. The initial concern when analyzing tobacco use and obesity was that clinician bias may result in a decrease in the prevalence of biologic use in these demographics, which was not supported in this study.
Although we identified few disparities, the results demonstrated a substantial underutilization of biologic therapies (5.2%), similar to the other US civilian studies (1.8-8.9%).4,5 While there is no current universal, standardized severity scoring system to evaluate HS (it is difficult to objectively define moderate to severe HS), estimates have shown that 40.3% to 65.8% of patients with HS have Hurley stage II or III.17-19 Therefore, only a small percentage of patients with moderate to severe disease were prescribed the only FDA-approved medication during this time period. The persistence of this underutilization within a medical system that reduces financial barriers suggests that nonfinancial barriers have a notable role in the underutilization of biologics.
For instance, risk of adverse events, particularly lymphoma and infection, has been cited by patients as a reason to avoid biologics. Additionally, treatment fatigue reduced some patients’ willingness to try new treatments, as did lack of knowledge about treatment options.6,20 Other reported barriers included the frequency of injections and fear of needles.6 Additionally, within the VA, ADA may require prior authorization at the local facility level.21 An established relationship with a dermatologist has been shown to significantly increase the odds of being prescribed a biologic medication in the face of these barriers.4 Future system-wide quality improvement initiatives could be implemented to identify patients with HS not followed by dermatology, with the goal of establishing care with a dermatologist.
Limitations
Limitations to this study include an inability to categorize HS disease severity and assess the degree to which disease severity confounded study findings, particularly in relation to tobacco use and obesity. The generalizability of this study is also limited because of the demographic characteristics of the veteran patient population, which is predominantly older, White, and male, whereas HS disproportionately affects younger, Black, and female individuals in the US.22 Despite these limitations, this study contributes valuable insights into the use of biologic therapies for veteran populations with HS using a national dataset.
Conclusions
This study was performed within a single-payer government medical system, likely reducing or removing the financial barriers that some patient populations may face when pursuing biologics for HS treatment. However, the prevalence of biologic use in this population was low overall (5.2%), suggesting that other factors play a role in the underutilization of biologics in HS. Consistent with previous studies, younger individuals were more likely to be prescribed a biologic, and no difference in prescription rates between Black and White patients was observed. Unlike previous studies, no significant difference in prescription rates between men and women was observed.
Hidradenitis suppurativa (HS) is a chronic, inflammatory skin disorder characterized by painful nodules, abscesses, and tunnels predominantly affecting intertriginous areas of the body.1,2 The condition poses significant challenges in terms of diagnosis, treatment, and quality of life for affected individuals. Various systemic therapies have been explored to manage this debilitating condition, with the emergence of biologic agents offering hope for improved outcomes. In 2015, adalimumab (ADA) was the first biologic approved by the US Food and Drug Administration (FDA) for the treatment of HS, followed by secukinumab in 2023 and bimekizumab in 2024. However, the off-label use of other biologics and/or tumor necrosis factor inhibitors such as infliximab (IFX) has become common practice.3
Although these therapies have demonstrated promising results in the treatment of HS, their widespread use may be hindered by accessibility and cost barriers. Orenstein et al analyzed data from the IBM Explorys platform from 2015 to 2020 and found that only 1.8% of patients diagnosed with HS had been prescribed ADA or IFX.4 More recently, Garg et al examined IBM MarketScan and IBM US Medicaid data from 2015 to 2018 to evaluate trends in clinical care and treatment. The prevalence of ADA and IFX prescriptions among patients with HS ranged from 2.3% to 8.0% (ADA) and 0.7% to 0.9% (IFX) for patients with commercial insurance, and 1.4% to 4.8% (ADA) and 0.5% to 0.7% (IFX) for patients with Medicaid.5 Biologics are often expensive, and the high cost associated with these therapies has been identified as a significant barrier to access for patients with HS, particularly those who lack adequate insurance coverage or face financial constraints.6
Furthermore, these barriers, particularly the financial barriers, are potentially compounded by the demographics of patients most notably affected by HS. In the US, a disproportionate incidence of HS has been noted in specific groups and age ranges, including women, individuals aged 18 to 29 years, and Black individuals.4 Orenstein et al found a statistically significant difference in use of ADA and IFX biologics based on age, sex, and race.4
The aim of this study was to examine the use of 2 biologics (ADA and IFX) in the Veterans Health Administration (VHA), a unique population in which financial barriers are reduced due to the single-payer government health care system structure. This design allowed for improved isolation and evaluation of variation in ADA and/or IFX prescription rates by demographics and health-related factors among patients with HS. To our knowledge, no studies have analyzed these metrics within the VHA.
Methods
This retrospective, cross-sectional analysis of VHA patients used data from the US Department of Veterans Affairs (VA) Corporate Data Warehouse, a data repository that provides access to longitudinal national electronic health record data for all veterans receiving care through VHA facilities. This study received ethical approval from institutional review boards at the Minneapolis Veterans Affairs Health Care System and VA Salt Lake City Healthcare System. Patient information was deidentified, and patient consent was not required.
Patients with HS were identified using ≥ 1 International Classification of Diseases (ICD) diagnostic code: (ICD-9 [705.83] or ICD-10 [L73.2]) between January 1, 2011, and December 31, 2021. The study included patients aged ≥ 18 years as of January 1, 2011, with ≥ 2 patient encounters during the postdiagnosis follow-up period, and with ≥ 1 encounter 6 months postindex. Patients with a biologic prescription prior to HS diagnosis were excluded. For this study, the term biologics refers to ADA and/or IFX prescriptions, unless otherwise specified. Only ADA and IFX were included in this analysis because ADA, a tumor necrosis factor (TNF)-á inhibitor, was the only FDA-approved medication at the time of the search, and IFX is another common TNF-α inhibitor used for the treatment of HS.
Statistical Analysis
We calculated logistic regression using SAS 9.4 (SAS Institute, Cary, NC). For each variable, the univariate relationship with biologic prescriptions was examined first, followed by the multivariate relationship controlling for all other variables. The following variables were controlled for in the multivariate models and were chosen a priori: sex, age, race, ethnicity, US region, hospital setting, current or previous tobacco use, obesity (defined as body mass index [BMI] ≥ 30), and Charlson Comorbidity Index (CCI).7
Results
Using ICD codes, we identified 29,483 individuals with ≥ 1 HS diagnosis (Figure 1). Of those identified, 1537 patients (5.21%) had been prescribed ≥ 1 biologic. The cohort was predominantly White (60.56%), male (75.27%), obese (59.34%), and had a history of current or previous tobacco use (73.47%) (Table 1). There were significant adjusted differences in prescription rates among veterans with HS based on age, race, and BMI. Notably, there was an age-dependent reduction in the odds of being prescribed a biologic in patients with HS. Compared with patients aged 18 to 44 years, patients aged 45 to 64 years (adjusted odds ratio [aOR], 0.63; 95% CI, 0.54–0.74; P < .001) and patients aged ≥ 65 years (aOR, 0.36; 95% CI, 0.27–0.48; P < .001) had significantly lower odds of receiving a biologic prescription (Table 2). Compared with White patients with HS, Native Hawaiian (NH) or Pacific Islander (PI) patients were less likely to be prescribed a biologic (aOR, 0.23; 95% CI, 0.06–0.92; P = .04). Patients with obesity had significantly higher odds of receiving a biologic prescription compared with patients without obesity (aOR, 1.47; 95% CI, 1.27– 1.71; P < .001).

Included in Analysis.
After adjusting for the variables listed in Table 1, there were no significant differences in biologic prescription rates for men compared with women (aOR, 0.97; 95% CI, 0.83-1.12; P = .68). We observed slight variations in biologic prescriptions between US regions (Midwest 5.0%, East 4.2%, South 5.8%, West 4.6%), none of which were significantly different in the fully adjusted model. No statistically significant differences were found in biologic prescriptions between urban and rural VA settings (5.4% vs 4.8%; aOR, 1.06; 95% CI, 0.90–1.24; P = .47). Tobacco use was not associated with the rate of biologic prescription receipt (aOR, 1.14; 95% CI, 0.97–1.34; P = .11). After adjusting for other variables (as outlined in Table 2), no significant differences were found between CCI of 0 and 1 (aOR, 0.97; 95% CI, 0.82–1.16; P = .77) or between CCI of 0 and 2 (aOR, 0.89; 95% CI, 0.74–1.07; P = .22).7


Discussion
The aim of the study was to ascertain potential discrepancies in biologic prescription patterns among patients with HS in the VHA by demographic and lifestyle behavior modifiers. Veteran cohorts are unique in composition, consisting predominantly of older White men within a single-payer health care system. The prevalence of biologic prescriptions in this population was low (5.2%), consistent with prior studies (1.8%–8.9%).4,5
We found a significant difference in ADA/IFX prescription patterns between White patients and NH/PI patients (aOR, 0.23; 95% CI, 0.06-0.92; P = .04). Further replication of this result is needed due to the small number of NH/PI patients included in the study (n = 241). Notably, we did not find a significant difference in the odds of Black patients being prescribed a biologic compared with White patients (aOR, 1.07; 95% CI, 0.92–1.25; P = .38), consistent with prior studies.4
In line with prior studies, age was associated with the likelihood of receiving a biologic prescription.4 Using the multivariate model adjusting for variables listed in Table 1, including CCI, patients aged 45 to 64 years and > 64 years were less likely to be prescribed a biologic than patients aged 18 to 44 years. HS disease activity could be a potential confounding variable, as HS severity may subside in some people with increasing age or menopause.8
Because different regions in the US have different sociopolitical ideologies and governing legislation, we hypothesized that there may be dissimilarities in the prevalence rates of biologic prescribing across various US regions. However, no significant differences were found in prescription patterns among US regions or between rural and urban settings. Previous research has demonstrated discernible disparities in both dermatologic care and clinical outcomes based on hospital setting (ie, urban vs rural).9-11
Tobacco use has been demonstrated to be associated with the development of HS.12 In a large retrospective analysis, Garg et al reported increased odds of receiving a new HS diagnosis in known tobacco users (aOR, 1.9; 95% CI, 1.8–2.0).13 The extent to which tobacco use affects HS severity is less understood. While some studies have found an association between smoking and HS severity, other analyses have failed to find this association.14,15 The effects of smoking cessation on the disease course of HS are unknown.16 This analysis, found no significant difference in prescriptions for biologics among patients with HS comparing current or previous tobacco users with nonusers.
There is a known positive correlation between increasing BMI and HS prevalence and severity that may be explained by the downstream effects of adipose tissue secretion of proinflammatory mediators and insulin resistance in the setting of chronic inflammation.12 This analysis found that patients with HS and obesity were 1.47 times more likely to be prescribed a biologic than patients with HS without obesity, which may be confounded by increased HS severity among patients with obesity. The initial concern when analyzing tobacco use and obesity was that clinician bias may result in a decrease in the prevalence of biologic use in these demographics, which was not supported in this study.
Although we identified few disparities, the results demonstrated a substantial underutilization of biologic therapies (5.2%), similar to the other US civilian studies (1.8-8.9%).4,5 While there is no current universal, standardized severity scoring system to evaluate HS (it is difficult to objectively define moderate to severe HS), estimates have shown that 40.3% to 65.8% of patients with HS have Hurley stage II or III.17-19 Therefore, only a small percentage of patients with moderate to severe disease were prescribed the only FDA-approved medication during this time period. The persistence of this underutilization within a medical system that reduces financial barriers suggests that nonfinancial barriers have a notable role in the underutilization of biologics.
For instance, risk of adverse events, particularly lymphoma and infection, has been cited by patients as a reason to avoid biologics. Additionally, treatment fatigue reduced some patients’ willingness to try new treatments, as did lack of knowledge about treatment options.6,20 Other reported barriers included the frequency of injections and fear of needles.6 Additionally, within the VA, ADA may require prior authorization at the local facility level.21 An established relationship with a dermatologist has been shown to significantly increase the odds of being prescribed a biologic medication in the face of these barriers.4 Future system-wide quality improvement initiatives could be implemented to identify patients with HS not followed by dermatology, with the goal of establishing care with a dermatologist.
Limitations
Limitations to this study include an inability to categorize HS disease severity and assess the degree to which disease severity confounded study findings, particularly in relation to tobacco use and obesity. The generalizability of this study is also limited because of the demographic characteristics of the veteran patient population, which is predominantly older, White, and male, whereas HS disproportionately affects younger, Black, and female individuals in the US.22 Despite these limitations, this study contributes valuable insights into the use of biologic therapies for veteran populations with HS using a national dataset.
Conclusions
This study was performed within a single-payer government medical system, likely reducing or removing the financial barriers that some patient populations may face when pursuing biologics for HS treatment. However, the prevalence of biologic use in this population was low overall (5.2%), suggesting that other factors play a role in the underutilization of biologics in HS. Consistent with previous studies, younger individuals were more likely to be prescribed a biologic, and no difference in prescription rates between Black and White patients was observed. Unlike previous studies, no significant difference in prescription rates between men and women was observed.
- Goldburg SR, Strober BE, Payette MJ. Hidradenitis suppurativa: epidemiology, clinical presentation, and pathogenesis. J Am Acad Dermatol. 2020;82:1045-1058. doi:10.1016/j.jaad.2019.08.090
- Tchero H, Herlin C, Bekara F, et al. Hidradenitis suppurativa: a systematic review and meta-analysis of therapeutic interventions. Indian J Dermatol Venereol Leprol. 2019;85:248-257. doi:10.4103/ijdvl.IJDVL_69_18
- Shih T, Lee K, Grogan T, et al. Infliximab in hidradenitis suppurativa: a systematic review and meta-analysis. Dermatol Ther. 2022;35:e15691. doi:10.1111/dth.15691
- Orenstein LAV, Wright S, Strunk A, et al. Low prescription of tumor necrosis alpha inhibitors in hidradenitis suppurativa: a cross-sectional analysis. J Am Acad Dermatol. 2021;84:1399-1401. doi:10.1016/j.jaad.2020.07.108
- Garg A, Naik HB, Alavi A, et al. Real-world findings on the characteristics and treatment exposures of patients with hidradenitis suppurativa from US claims data. Dermatol Ther (Heidelb). 2023;13:581-594. doi:10.1007/s13555-022-00872-1
- De DR, Shih T, Fixsen D, et al. Biologic use in hidradenitis suppurativa: patient perspectives and barriers. J Dermatolog Treat. 2022;33:3060-3062. doi:10.1080/09546634.2022.2089336
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- von der Werth JM, Williams HC. The natural history of hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2000;14:389-392. doi:10.1046/j.1468-3083.2000.00087.x
- Silverberg JI, Barbarot S, Gadkari A, et al. Atopic dermatitis in the pediatric population: a cross-sectional, international epidemiologic study. Ann Allergy Asthma Immunol. 2021;126:417-428.e2. doi:10.1016/j.anai.2020.12.020
- Wu YP, Parsons B, Jo Y, et al. Outdoor activities and sunburn among urban and rural families in a Western region of the US: implications for skin cancer prevention. Prev Med Rep. 2022;29:101914. doi:10.1016/j.pmedr.2022.101914
- Mannschreck DB, Li X, Okoye G. Rural melanoma patients in Maryland do not present with more advanced disease than urban patients. Dermatol Online J. 2021;27. doi:10.5070/D327553607
- Garg A, Malviya N, Strunk A, et al. Comorbidity screening in hidradenitis suppurativa: evidence-based recommendations from the US and Canadian Hidradenitis Suppurativa Foundations. J Am Acad Dermatol. 2022;86:1092-1101. doi:10.1016/j.jaad.2021.01.059
- Garg A, Papagermanos V, Midura M, et al. Incidence of hidradenitis suppurativa among tobacco smokers: a population- based retrospective analysis in the U.S.A. Br J Dermatol. 2018;178:709-714. doi:10.1111/bjd.15939
- Sartorius K, Emtestam L, Jemec GBE, et al. Objective scoring of hidradenitis suppurativa reflecting the role of tobacco smoking and obesity. Br J Dermatol. 2009;161:831- 839. doi:10.1111/j.1365-2133.2009.09198.x
- Canoui-Poitrine F, Revuz JE, Wolkenstein P, et al. Clinical characteristics of a series of 302 French patients with hidradenitis suppurativa, with an analysis of factors associated with disease severity. J Am Acad Dermatol. 2009;61:51-57. doi:10.1016/j.jaad.2009.02.013
- Dufour DN, Emtestam L, Jemec GB. Hidradenitis suppurativa: a common and burdensome, yet under-recognised, inflammatory skin disease. Postgrad Med J. 2014;90:216- 221. doi:10.1136/postgradmedj-2013-131994
- Vazquez BG, Alikhan A, Weaver AL, et al. Incidence of hidradenitis suppurativa and associated factors: a population- based study of Olmsted County, Minnesota. J Invest Dermatol. 2013;133:97-103. doi:10.1038/jid.2012.255
- Vanlaerhoven AMJD, Ardon CB, van Straalen KR, et al. Hurley III hidradenitis suppurativa has an aggressive disease course. Dermatology. 2018;234:232-233. doi:10.1159/000491547
- Shahi V, Alikhan A, Vazquez BG, et al. Prevalence of hidradenitis suppurativa: a population-based study in Olmsted County, Minnesota. Dermatology. 2014;229:154-158. doi:10.1159/000363381
- Salame N, Sow YN, Siira MR, et al. Factors affecting treatment selection among patients with hidradenitis suppurativa. JAMA Dermatol. 2024;160:179. doi:10.1001/jamadermatol.2023.5425
- VA Formulary Advisor: ADALIMUMAB-BWWD INJ,SOLN. US Department of Veterans Affairs. Updated December 17, 2025. Accessed January 15, 2026. https://www.va.gov/formularyadvisor/drugs/4042383-ADALIMUMAB-BWWD-INJ-SOLN
- Garg A, Lavian J, Lin G, et al. Incidence of hidradenitis suppurativa in the United States: a sex- and age-adjusted population analysis. J Am Acad Dermatol. 2017;77:118- 122. doi:10.1016/j.jaad.2017.02.005
- Goldburg SR, Strober BE, Payette MJ. Hidradenitis suppurativa: epidemiology, clinical presentation, and pathogenesis. J Am Acad Dermatol. 2020;82:1045-1058. doi:10.1016/j.jaad.2019.08.090
- Tchero H, Herlin C, Bekara F, et al. Hidradenitis suppurativa: a systematic review and meta-analysis of therapeutic interventions. Indian J Dermatol Venereol Leprol. 2019;85:248-257. doi:10.4103/ijdvl.IJDVL_69_18
- Shih T, Lee K, Grogan T, et al. Infliximab in hidradenitis suppurativa: a systematic review and meta-analysis. Dermatol Ther. 2022;35:e15691. doi:10.1111/dth.15691
- Orenstein LAV, Wright S, Strunk A, et al. Low prescription of tumor necrosis alpha inhibitors in hidradenitis suppurativa: a cross-sectional analysis. J Am Acad Dermatol. 2021;84:1399-1401. doi:10.1016/j.jaad.2020.07.108
- Garg A, Naik HB, Alavi A, et al. Real-world findings on the characteristics and treatment exposures of patients with hidradenitis suppurativa from US claims data. Dermatol Ther (Heidelb). 2023;13:581-594. doi:10.1007/s13555-022-00872-1
- De DR, Shih T, Fixsen D, et al. Biologic use in hidradenitis suppurativa: patient perspectives and barriers. J Dermatolog Treat. 2022;33:3060-3062. doi:10.1080/09546634.2022.2089336
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- von der Werth JM, Williams HC. The natural history of hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2000;14:389-392. doi:10.1046/j.1468-3083.2000.00087.x
- Silverberg JI, Barbarot S, Gadkari A, et al. Atopic dermatitis in the pediatric population: a cross-sectional, international epidemiologic study. Ann Allergy Asthma Immunol. 2021;126:417-428.e2. doi:10.1016/j.anai.2020.12.020
- Wu YP, Parsons B, Jo Y, et al. Outdoor activities and sunburn among urban and rural families in a Western region of the US: implications for skin cancer prevention. Prev Med Rep. 2022;29:101914. doi:10.1016/j.pmedr.2022.101914
- Mannschreck DB, Li X, Okoye G. Rural melanoma patients in Maryland do not present with more advanced disease than urban patients. Dermatol Online J. 2021;27. doi:10.5070/D327553607
- Garg A, Malviya N, Strunk A, et al. Comorbidity screening in hidradenitis suppurativa: evidence-based recommendations from the US and Canadian Hidradenitis Suppurativa Foundations. J Am Acad Dermatol. 2022;86:1092-1101. doi:10.1016/j.jaad.2021.01.059
- Garg A, Papagermanos V, Midura M, et al. Incidence of hidradenitis suppurativa among tobacco smokers: a population- based retrospective analysis in the U.S.A. Br J Dermatol. 2018;178:709-714. doi:10.1111/bjd.15939
- Sartorius K, Emtestam L, Jemec GBE, et al. Objective scoring of hidradenitis suppurativa reflecting the role of tobacco smoking and obesity. Br J Dermatol. 2009;161:831- 839. doi:10.1111/j.1365-2133.2009.09198.x
- Canoui-Poitrine F, Revuz JE, Wolkenstein P, et al. Clinical characteristics of a series of 302 French patients with hidradenitis suppurativa, with an analysis of factors associated with disease severity. J Am Acad Dermatol. 2009;61:51-57. doi:10.1016/j.jaad.2009.02.013
- Dufour DN, Emtestam L, Jemec GB. Hidradenitis suppurativa: a common and burdensome, yet under-recognised, inflammatory skin disease. Postgrad Med J. 2014;90:216- 221. doi:10.1136/postgradmedj-2013-131994
- Vazquez BG, Alikhan A, Weaver AL, et al. Incidence of hidradenitis suppurativa and associated factors: a population- based study of Olmsted County, Minnesota. J Invest Dermatol. 2013;133:97-103. doi:10.1038/jid.2012.255
- Vanlaerhoven AMJD, Ardon CB, van Straalen KR, et al. Hurley III hidradenitis suppurativa has an aggressive disease course. Dermatology. 2018;234:232-233. doi:10.1159/000491547
- Shahi V, Alikhan A, Vazquez BG, et al. Prevalence of hidradenitis suppurativa: a population-based study in Olmsted County, Minnesota. Dermatology. 2014;229:154-158. doi:10.1159/000363381
- Salame N, Sow YN, Siira MR, et al. Factors affecting treatment selection among patients with hidradenitis suppurativa. JAMA Dermatol. 2024;160:179. doi:10.1001/jamadermatol.2023.5425
- VA Formulary Advisor: ADALIMUMAB-BWWD INJ,SOLN. US Department of Veterans Affairs. Updated December 17, 2025. Accessed January 15, 2026. https://www.va.gov/formularyadvisor/drugs/4042383-ADALIMUMAB-BWWD-INJ-SOLN
- Garg A, Lavian J, Lin G, et al. Incidence of hidradenitis suppurativa in the United States: a sex- and age-adjusted population analysis. J Am Acad Dermatol. 2017;77:118- 122. doi:10.1016/j.jaad.2017.02.005
Cross-Sectional Analysis of Biologic Use in the Treatment of Veterans With Hidradenitis Suppurativa
Cross-Sectional Analysis of Biologic Use in the Treatment of Veterans With Hidradenitis Suppurativa
Early Infantile Hemangioma Diagnosis Is Key in Skin of Color
Early Infantile Hemangioma Diagnosis Is Key in Skin of Color
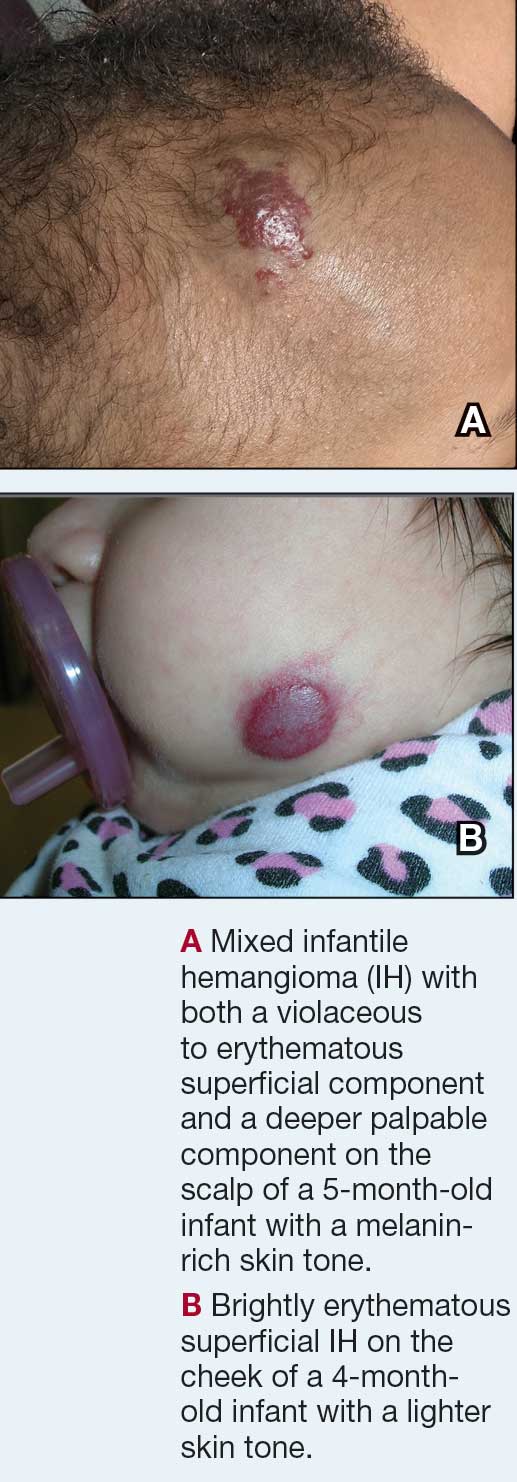
Richard P. Usatine, MD
Infantile hemangioma (IH) is the most common vascular tumor of infancy, appearing within the first few weeks of life and typically reaching peak size by age 3 to 5 months.1 It classically manifests as a raised or flat bright-red lesion in the upper dermis of the skin and/or subcutaneous tissue and can vary in number, size, shape, and location.2 It is characterized by a rapid proliferative phase, especially between 5 and 8 weeks of age, followed by gradual spontaneous regression over 1 to 10 years.1-3
Infantile hemangiomas are categorized based on depth (superficial, deep, or mixed) and distribution pattern (focal, multifocal, segmental, or indeterminate).4 In most cases, complete regression occurs by age 4 years, but there can be residual telangiectasia, fibrofatty tissue, and/or scarring.1,4 About 10% to 15% of IHs result in complications that require medical intervention (eg, visual, airway, or auditory compromise; ulceration; disfigurement); ideally, these patients should be referred to a specialist by 5 weeks of age.4 Prompt assessment of IH severity is essential to prevent or mitigate potential complications and ultimately improve outcomes.3 Social drivers of health contribute to delayed diagnosis and management of hemangiomas, leading to increased complications in some patient populations.5-7
Epidemiology
Infantile hemangiomas are estimated to manifest in 4.5% of infants in the United States.1 The most common type is superficial IH, typically found on the head or neck.5 Risk factors in infants include female sex, White race, premature birth, and low birth weight (< 1000 g).1,3 Maternal risk factors include advanced gestational age (ie, > 35 years), multiple gestations, family history of IH, tobacco use, use of progesterone therapy during pregnancy, and pre-eclampsia.1,3
Focal IH typically manifests as a single localized lesion that can occur anywhere on the body.2,3 In contrast, segmental IH manifests in a linear pattern and/or is distributed on a large anatomic area, most commonly on the face and less frequently the extremities and trunk.
Key Clinical Features
Superficial IH in patients with darker skin tones may appear as a dark-red or violaceous papule or plaque compared to bright red in lighter skin tones.5 Deep IH may appear as a soft, round, flesh-colored or blue-hued subcutaneous mass, the color of which may be harder to appreciate in those with darker skin tones.5
Worth Noting
Complications from IH may require imaging, close follow-up, systemic therapy, multidisciplinary care, and advanced health literacy and patient/family navigation. Multifocal IHs (≥ 5 lesions) are more likely to be associated with infantile hepatic hemangiomas.2,3 Large (> 5 cm) segmental IHs on the face and lumbosacral area require further evaluation for PHACES (posterior fossa malformation, hemangiomas, arterial anomalies, cardiac defects, eye anomalies, and sternal raphe/cleft defects) and LUMBAR (lower-body segmental IH; urogenital anomalies and ulceration; myelopathy; bony deformities; anorectal malformations and arterial anomalies; and renal anomalies) syndromes, which are more common in patients of Hispanic ethnicity.2,3
The Infantile Hemangioma Referral Score is a recently validated tool that can assist primary care physicians in timely referral of IHs requiring early specialist intervention.4,9 It takes into account the location, number, and size of the lesions and the age of the patient; these factors help to determine which IHs may be managed conservatively vs those that may require treatment to prevent life-threatening complications.1-3
Systemic corticosteroids historically have been the primary treatment for IH; however, in the past decade, propranolol oral solution (4.28 mg/mL) has become the first-line therapy for most infants requiring systemic management.10 It is the only medication approved by the US Food and Drug Administration for proliferating IH, with treatment initiation as young as 5 weeks corrected age.11 As a nonselective beta-blocker, propranolol is believed to reduce IHs through vasoconstriction or by inhibition of angiogenesis.1,4,10
For small superficial IHs, treatment options include timolol maleate ophthalmic solution 0.5% (one drop applied twice daily to the IH) or pulsed dye laser therapy.4,10 Surgical excision typically is avoided during infancy due to concerns about anesthetic risks and potential blood loss.4,10 Surgery is reserved for cases involving residual fibrofatty tissue, postinvolution scarring, obstruction of vital structures, or lesions in aesthetically sensitive areas as well as when propranolol is contraindicated.4,10
Health Disparity Highlight
Infants with skin of color and those of lower socioeconomic status (SES) face a heightened risk for delayed diagnosis and more advanced disease at the initial evaluation for IH.5,7 Access barriers such as geographic limitations to specialty services, lack of insurance, underinsurance, and language differences impact timely diagnosis and treatment.5,6 Implementation of telemedicine services in areas with limited access to specialists can facilitate early evaluation and risk stratification for IH.12
A retrospective cohort study of 804 children seen at a large academic hospital found that those of lower SES were more likely to seek care after 3 months of age than their higher-SES counterparts.6 Those who presented after 6 months of age also had higher IH severity scores compared to their counterparts with higher SES.6 Delayed access to care may cause children to miss the critical treatment window during the rapid proliferative growth phase.6,12 However, children insured through Medicaid or the Children’s Health Insurance Program who participated in institutional care management programs (which assist in scheduling specialty care appointments within the institution) sought treatment earlier regardless of their SES, suggesting that such programs may help reduce disparities in timely access for children of lower SES.6
An epidemiologic study analyzing the demographics of children hospitalized across the United States demonstrated that Black infants with IH were more likely to belong to the lowest income quartile compared with White infants or those of other races. They also were 2 times older on average at initial presentation (1.8 vs 1.0 years), experienced longer hospitalizations (16.4 vs 13.8 days), and underwent more IH-related procedures than White infants and infants of other races (2.4, 1.9, and 2.1, respectively).7
These and other factors may contribute to missed windows of opportunity for timely treatment of high-risk IHs in patients with darker skin tones and/or those facing challenges stemming from social drivers of health.
- Léauté-Labrèze C, Harper JI, Hoeger PH. Infantile haemangioma. Lancet. 2017;390:85-94.
- Mitra R, Fitzsimons HL, Hale T, et al. Recent advances in understanding the molecular basis of infantile haemangioma development. Br J Dermatol. 2024;191:661-669.
- Rodríguez Bandera AI, Sebaratnam DF, Wargon O, et al. Infantile hemangioma. Part 1: epidemiology, pathogenesis, clinical presentation and assessment. J Am Acad Dermatol. 2021;85:1379-1392.
- Sebaratnam DF, Rodríguez Bandera AL, Wong LCF, et al. Infantile hemangioma. Part 2: management. J Am Acad Dermatol. 2021;85:1395-1404.
- Taye ME, Shah J, Seiverling EV, et al. Diagnosis of vascular anomalies in patients with skin of color. J Clin Aesthet Dermatol. 2024;17:54-62.
- Lie E, Psoter KJ, Püttgen KB. Lower socioeconomic status is associated with delayed access to care for infantile hemangioma: a cohort study. J Am Acad Dermatol. 2023;88:E221-E230.
- Kumar KD, Desai AD, Shah VP, et al. Racial discrepancies in presentation of hospitalized infantile hemangioma cases using the Kids’ Inpatient Database. Health Sci Rep. 2023;6:E1092.
- Chiller KG, Passaro D, Frieden IJ. Hemangiomas of infancy: clinical characteristics, morphologic subtypes, and their relationship to race, ethnicity, and sex. Arch Dermatol. 2002;138:1567.
- Léauté-Labrèze C, Baselga Torres E, Weibel L, et al. The infantile hemangioma referral score: a validated tool for physicians. Pediatrics. 2020;145:E20191628.
- Macca L, Altavilla D, Di Bartolomeo L, et al. Update on treatment of infantile hemangiomas: what’s new in the last five years? Front Pharmacol. 2022;13:879602.
- Krowchuk DP, Frieden IJ, Mancini AJ, et al. Clinical practice guideline for the management of infantile hemangiomas. Pediatrics. 2019;143:E20183475.
- Frieden IJ, Püttgen KB, Drolet BA, et al. Management of infantile hemangiomas during the COVID pandemic. Pediatr Dermatol. 2020;37:412-418.

Richard P. Usatine, MD
Infantile hemangioma (IH) is the most common vascular tumor of infancy, appearing within the first few weeks of life and typically reaching peak size by age 3 to 5 months.1 It classically manifests as a raised or flat bright-red lesion in the upper dermis of the skin and/or subcutaneous tissue and can vary in number, size, shape, and location.2 It is characterized by a rapid proliferative phase, especially between 5 and 8 weeks of age, followed by gradual spontaneous regression over 1 to 10 years.1-3
Infantile hemangiomas are categorized based on depth (superficial, deep, or mixed) and distribution pattern (focal, multifocal, segmental, or indeterminate).4 In most cases, complete regression occurs by age 4 years, but there can be residual telangiectasia, fibrofatty tissue, and/or scarring.1,4 About 10% to 15% of IHs result in complications that require medical intervention (eg, visual, airway, or auditory compromise; ulceration; disfigurement); ideally, these patients should be referred to a specialist by 5 weeks of age.4 Prompt assessment of IH severity is essential to prevent or mitigate potential complications and ultimately improve outcomes.3 Social drivers of health contribute to delayed diagnosis and management of hemangiomas, leading to increased complications in some patient populations.5-7
Epidemiology
Infantile hemangiomas are estimated to manifest in 4.5% of infants in the United States.1 The most common type is superficial IH, typically found on the head or neck.5 Risk factors in infants include female sex, White race, premature birth, and low birth weight (< 1000 g).1,3 Maternal risk factors include advanced gestational age (ie, > 35 years), multiple gestations, family history of IH, tobacco use, use of progesterone therapy during pregnancy, and pre-eclampsia.1,3
Focal IH typically manifests as a single localized lesion that can occur anywhere on the body.2,3 In contrast, segmental IH manifests in a linear pattern and/or is distributed on a large anatomic area, most commonly on the face and less frequently the extremities and trunk.
Key Clinical Features
Superficial IH in patients with darker skin tones may appear as a dark-red or violaceous papule or plaque compared to bright red in lighter skin tones.5 Deep IH may appear as a soft, round, flesh-colored or blue-hued subcutaneous mass, the color of which may be harder to appreciate in those with darker skin tones.5
Worth Noting
Complications from IH may require imaging, close follow-up, systemic therapy, multidisciplinary care, and advanced health literacy and patient/family navigation. Multifocal IHs (≥ 5 lesions) are more likely to be associated with infantile hepatic hemangiomas.2,3 Large (> 5 cm) segmental IHs on the face and lumbosacral area require further evaluation for PHACES (posterior fossa malformation, hemangiomas, arterial anomalies, cardiac defects, eye anomalies, and sternal raphe/cleft defects) and LUMBAR (lower-body segmental IH; urogenital anomalies and ulceration; myelopathy; bony deformities; anorectal malformations and arterial anomalies; and renal anomalies) syndromes, which are more common in patients of Hispanic ethnicity.2,3
The Infantile Hemangioma Referral Score is a recently validated tool that can assist primary care physicians in timely referral of IHs requiring early specialist intervention.4,9 It takes into account the location, number, and size of the lesions and the age of the patient; these factors help to determine which IHs may be managed conservatively vs those that may require treatment to prevent life-threatening complications.1-3
Systemic corticosteroids historically have been the primary treatment for IH; however, in the past decade, propranolol oral solution (4.28 mg/mL) has become the first-line therapy for most infants requiring systemic management.10 It is the only medication approved by the US Food and Drug Administration for proliferating IH, with treatment initiation as young as 5 weeks corrected age.11 As a nonselective beta-blocker, propranolol is believed to reduce IHs through vasoconstriction or by inhibition of angiogenesis.1,4,10
For small superficial IHs, treatment options include timolol maleate ophthalmic solution 0.5% (one drop applied twice daily to the IH) or pulsed dye laser therapy.4,10 Surgical excision typically is avoided during infancy due to concerns about anesthetic risks and potential blood loss.4,10 Surgery is reserved for cases involving residual fibrofatty tissue, postinvolution scarring, obstruction of vital structures, or lesions in aesthetically sensitive areas as well as when propranolol is contraindicated.4,10
Health Disparity Highlight
Infants with skin of color and those of lower socioeconomic status (SES) face a heightened risk for delayed diagnosis and more advanced disease at the initial evaluation for IH.5,7 Access barriers such as geographic limitations to specialty services, lack of insurance, underinsurance, and language differences impact timely diagnosis and treatment.5,6 Implementation of telemedicine services in areas with limited access to specialists can facilitate early evaluation and risk stratification for IH.12
A retrospective cohort study of 804 children seen at a large academic hospital found that those of lower SES were more likely to seek care after 3 months of age than their higher-SES counterparts.6 Those who presented after 6 months of age also had higher IH severity scores compared to their counterparts with higher SES.6 Delayed access to care may cause children to miss the critical treatment window during the rapid proliferative growth phase.6,12 However, children insured through Medicaid or the Children’s Health Insurance Program who participated in institutional care management programs (which assist in scheduling specialty care appointments within the institution) sought treatment earlier regardless of their SES, suggesting that such programs may help reduce disparities in timely access for children of lower SES.6
An epidemiologic study analyzing the demographics of children hospitalized across the United States demonstrated that Black infants with IH were more likely to belong to the lowest income quartile compared with White infants or those of other races. They also were 2 times older on average at initial presentation (1.8 vs 1.0 years), experienced longer hospitalizations (16.4 vs 13.8 days), and underwent more IH-related procedures than White infants and infants of other races (2.4, 1.9, and 2.1, respectively).7
These and other factors may contribute to missed windows of opportunity for timely treatment of high-risk IHs in patients with darker skin tones and/or those facing challenges stemming from social drivers of health.

Richard P. Usatine, MD
Infantile hemangioma (IH) is the most common vascular tumor of infancy, appearing within the first few weeks of life and typically reaching peak size by age 3 to 5 months.1 It classically manifests as a raised or flat bright-red lesion in the upper dermis of the skin and/or subcutaneous tissue and can vary in number, size, shape, and location.2 It is characterized by a rapid proliferative phase, especially between 5 and 8 weeks of age, followed by gradual spontaneous regression over 1 to 10 years.1-3
Infantile hemangiomas are categorized based on depth (superficial, deep, or mixed) and distribution pattern (focal, multifocal, segmental, or indeterminate).4 In most cases, complete regression occurs by age 4 years, but there can be residual telangiectasia, fibrofatty tissue, and/or scarring.1,4 About 10% to 15% of IHs result in complications that require medical intervention (eg, visual, airway, or auditory compromise; ulceration; disfigurement); ideally, these patients should be referred to a specialist by 5 weeks of age.4 Prompt assessment of IH severity is essential to prevent or mitigate potential complications and ultimately improve outcomes.3 Social drivers of health contribute to delayed diagnosis and management of hemangiomas, leading to increased complications in some patient populations.5-7
Epidemiology
Infantile hemangiomas are estimated to manifest in 4.5% of infants in the United States.1 The most common type is superficial IH, typically found on the head or neck.5 Risk factors in infants include female sex, White race, premature birth, and low birth weight (< 1000 g).1,3 Maternal risk factors include advanced gestational age (ie, > 35 years), multiple gestations, family history of IH, tobacco use, use of progesterone therapy during pregnancy, and pre-eclampsia.1,3
Focal IH typically manifests as a single localized lesion that can occur anywhere on the body.2,3 In contrast, segmental IH manifests in a linear pattern and/or is distributed on a large anatomic area, most commonly on the face and less frequently the extremities and trunk.
Key Clinical Features
Superficial IH in patients with darker skin tones may appear as a dark-red or violaceous papule or plaque compared to bright red in lighter skin tones.5 Deep IH may appear as a soft, round, flesh-colored or blue-hued subcutaneous mass, the color of which may be harder to appreciate in those with darker skin tones.5
Worth Noting
Complications from IH may require imaging, close follow-up, systemic therapy, multidisciplinary care, and advanced health literacy and patient/family navigation. Multifocal IHs (≥ 5 lesions) are more likely to be associated with infantile hepatic hemangiomas.2,3 Large (> 5 cm) segmental IHs on the face and lumbosacral area require further evaluation for PHACES (posterior fossa malformation, hemangiomas, arterial anomalies, cardiac defects, eye anomalies, and sternal raphe/cleft defects) and LUMBAR (lower-body segmental IH; urogenital anomalies and ulceration; myelopathy; bony deformities; anorectal malformations and arterial anomalies; and renal anomalies) syndromes, which are more common in patients of Hispanic ethnicity.2,3
The Infantile Hemangioma Referral Score is a recently validated tool that can assist primary care physicians in timely referral of IHs requiring early specialist intervention.4,9 It takes into account the location, number, and size of the lesions and the age of the patient; these factors help to determine which IHs may be managed conservatively vs those that may require treatment to prevent life-threatening complications.1-3
Systemic corticosteroids historically have been the primary treatment for IH; however, in the past decade, propranolol oral solution (4.28 mg/mL) has become the first-line therapy for most infants requiring systemic management.10 It is the only medication approved by the US Food and Drug Administration for proliferating IH, with treatment initiation as young as 5 weeks corrected age.11 As a nonselective beta-blocker, propranolol is believed to reduce IHs through vasoconstriction or by inhibition of angiogenesis.1,4,10
For small superficial IHs, treatment options include timolol maleate ophthalmic solution 0.5% (one drop applied twice daily to the IH) or pulsed dye laser therapy.4,10 Surgical excision typically is avoided during infancy due to concerns about anesthetic risks and potential blood loss.4,10 Surgery is reserved for cases involving residual fibrofatty tissue, postinvolution scarring, obstruction of vital structures, or lesions in aesthetically sensitive areas as well as when propranolol is contraindicated.4,10
Health Disparity Highlight
Infants with skin of color and those of lower socioeconomic status (SES) face a heightened risk for delayed diagnosis and more advanced disease at the initial evaluation for IH.5,7 Access barriers such as geographic limitations to specialty services, lack of insurance, underinsurance, and language differences impact timely diagnosis and treatment.5,6 Implementation of telemedicine services in areas with limited access to specialists can facilitate early evaluation and risk stratification for IH.12
A retrospective cohort study of 804 children seen at a large academic hospital found that those of lower SES were more likely to seek care after 3 months of age than their higher-SES counterparts.6 Those who presented after 6 months of age also had higher IH severity scores compared to their counterparts with higher SES.6 Delayed access to care may cause children to miss the critical treatment window during the rapid proliferative growth phase.6,12 However, children insured through Medicaid or the Children’s Health Insurance Program who participated in institutional care management programs (which assist in scheduling specialty care appointments within the institution) sought treatment earlier regardless of their SES, suggesting that such programs may help reduce disparities in timely access for children of lower SES.6
An epidemiologic study analyzing the demographics of children hospitalized across the United States demonstrated that Black infants with IH were more likely to belong to the lowest income quartile compared with White infants or those of other races. They also were 2 times older on average at initial presentation (1.8 vs 1.0 years), experienced longer hospitalizations (16.4 vs 13.8 days), and underwent more IH-related procedures than White infants and infants of other races (2.4, 1.9, and 2.1, respectively).7
These and other factors may contribute to missed windows of opportunity for timely treatment of high-risk IHs in patients with darker skin tones and/or those facing challenges stemming from social drivers of health.
- Léauté-Labrèze C, Harper JI, Hoeger PH. Infantile haemangioma. Lancet. 2017;390:85-94.
- Mitra R, Fitzsimons HL, Hale T, et al. Recent advances in understanding the molecular basis of infantile haemangioma development. Br J Dermatol. 2024;191:661-669.
- Rodríguez Bandera AI, Sebaratnam DF, Wargon O, et al. Infantile hemangioma. Part 1: epidemiology, pathogenesis, clinical presentation and assessment. J Am Acad Dermatol. 2021;85:1379-1392.
- Sebaratnam DF, Rodríguez Bandera AL, Wong LCF, et al. Infantile hemangioma. Part 2: management. J Am Acad Dermatol. 2021;85:1395-1404.
- Taye ME, Shah J, Seiverling EV, et al. Diagnosis of vascular anomalies in patients with skin of color. J Clin Aesthet Dermatol. 2024;17:54-62.
- Lie E, Psoter KJ, Püttgen KB. Lower socioeconomic status is associated with delayed access to care for infantile hemangioma: a cohort study. J Am Acad Dermatol. 2023;88:E221-E230.
- Kumar KD, Desai AD, Shah VP, et al. Racial discrepancies in presentation of hospitalized infantile hemangioma cases using the Kids’ Inpatient Database. Health Sci Rep. 2023;6:E1092.
- Chiller KG, Passaro D, Frieden IJ. Hemangiomas of infancy: clinical characteristics, morphologic subtypes, and their relationship to race, ethnicity, and sex. Arch Dermatol. 2002;138:1567.
- Léauté-Labrèze C, Baselga Torres E, Weibel L, et al. The infantile hemangioma referral score: a validated tool for physicians. Pediatrics. 2020;145:E20191628.
- Macca L, Altavilla D, Di Bartolomeo L, et al. Update on treatment of infantile hemangiomas: what’s new in the last five years? Front Pharmacol. 2022;13:879602.
- Krowchuk DP, Frieden IJ, Mancini AJ, et al. Clinical practice guideline for the management of infantile hemangiomas. Pediatrics. 2019;143:E20183475.
- Frieden IJ, Püttgen KB, Drolet BA, et al. Management of infantile hemangiomas during the COVID pandemic. Pediatr Dermatol. 2020;37:412-418.
- Léauté-Labrèze C, Harper JI, Hoeger PH. Infantile haemangioma. Lancet. 2017;390:85-94.
- Mitra R, Fitzsimons HL, Hale T, et al. Recent advances in understanding the molecular basis of infantile haemangioma development. Br J Dermatol. 2024;191:661-669.
- Rodríguez Bandera AI, Sebaratnam DF, Wargon O, et al. Infantile hemangioma. Part 1: epidemiology, pathogenesis, clinical presentation and assessment. J Am Acad Dermatol. 2021;85:1379-1392.
- Sebaratnam DF, Rodríguez Bandera AL, Wong LCF, et al. Infantile hemangioma. Part 2: management. J Am Acad Dermatol. 2021;85:1395-1404.
- Taye ME, Shah J, Seiverling EV, et al. Diagnosis of vascular anomalies in patients with skin of color. J Clin Aesthet Dermatol. 2024;17:54-62.
- Lie E, Psoter KJ, Püttgen KB. Lower socioeconomic status is associated with delayed access to care for infantile hemangioma: a cohort study. J Am Acad Dermatol. 2023;88:E221-E230.
- Kumar KD, Desai AD, Shah VP, et al. Racial discrepancies in presentation of hospitalized infantile hemangioma cases using the Kids’ Inpatient Database. Health Sci Rep. 2023;6:E1092.
- Chiller KG, Passaro D, Frieden IJ. Hemangiomas of infancy: clinical characteristics, morphologic subtypes, and their relationship to race, ethnicity, and sex. Arch Dermatol. 2002;138:1567.
- Léauté-Labrèze C, Baselga Torres E, Weibel L, et al. The infantile hemangioma referral score: a validated tool for physicians. Pediatrics. 2020;145:E20191628.
- Macca L, Altavilla D, Di Bartolomeo L, et al. Update on treatment of infantile hemangiomas: what’s new in the last five years? Front Pharmacol. 2022;13:879602.
- Krowchuk DP, Frieden IJ, Mancini AJ, et al. Clinical practice guideline for the management of infantile hemangiomas. Pediatrics. 2019;143:E20183475.
- Frieden IJ, Püttgen KB, Drolet BA, et al. Management of infantile hemangiomas during the COVID pandemic. Pediatr Dermatol. 2020;37:412-418.
Early Infantile Hemangioma Diagnosis Is Key in Skin of Color
Early Infantile Hemangioma Diagnosis Is Key in Skin of Color
Atrophic Areas on the Axillary and Anogenital Anatomy
Atrophic Areas on the Axillary and Anogenital Anatomy
Discussion
A diagnosis of lichen sclerosus (LS) was made based on clinical and dermoscopic features, followed by confirmation with histology. The patient’s presentation included typical signs and symptoms of LS: itching, burning, intermittent bleeding, perianal hemorrhage, fusion of the clitoral head, and fissures. Other presentations can include dyspareunia, erosions, and excoriations; however, these symptoms and signs were not reported or seen in this patient.
LS typically affects the anogenital region and has 2 peak incidences: in preadolescent teens and during the fifth to sixth decade of life.1 This patient presented with a case of extragenital LS, which is less common than the classic presentation of LS that affects the genitals. This variant’s epidemiology differs, as it is less common in children and more common in postmenopausal women.2 Extragenital LS presents as white, atrophic plaques with a predilection for sites including the trunk, breasts, upper arms, and sites of physical trauma, with symptoms of dryness and pruritus. Over time, the papules can coalesce and form ivory, scar-like papules or plaques with a wrinkled surface. In advanced stages, telangiectasia or follicular plugging can be present, along with flattening of the dermal-epidermal junction. This flat interface is fragile and can result in bullae that may become hemorrhagic.
Cutaneous squamous cell carcinoma (SCC) may infrequently arise from LS, similar to other chronic inflammatory dermatoses.3 Lichen planus is typically not associated with an increased risk of SCC, except in the oral and hypertrophic variants. However, LS may be considered a premalignant process, and many vulvar SCC cases are noted to have adjacent LS lesions.3
Autoimmune and genetic factors contribute to the pathogenesis of LS. Extracellular matrix protein 1 (ECM1) binds molecules of the basement membrane zone and dermis, contributing to the structure and integrity of skin. Autoantibodies against ECM1 and other antigens of the basement membrane zone, including BP180 and BP320, were found in LS.2 HLA-DQ7 major histocompatibility complex class II antigens have been associated with LS.1
On histologic examination, the epidermis of LS is atrophic with hyperkeratosis. The dermis shows homogenization and sclerosis of superficial collagen with a band-like lymphocytic infiltrate below the sclerosis. The basal layer is thickened, showing basal cell vacuolization and hydropic degeneration.4
First-line treatment for genital and extragenital variants of LS is high-potency topical steroids for 3 months or until the skin texture and color resolve (ie, clobetasol 0.05% cream or ointment). The second-line treatment is a topical calcineurin inhibitor. These treatments are used for management. They are not cures for LS, as relapse is possible after the initial treatment course is completed. Adverse effects of high potency topical steroids are skin burning, skin atrophy, and fragility, telangiectasia. The adverse effects of topical calcineurin inhibitors are stinging and burning on application.
Other Diagnostic Considerations
Inverse psoriasis (IP) is a variant of psoriasis that presents as erythematous, well-demarcated plaques with minimal scale in intertriginous areas and flexural surfaces. Localized dermatophyte, candidal, or bacterial infections can trigger IP.5 It occurs in about 3% to 7% of patients with plaque psoriasis and is thought to form due to koebnerization via mechanical friction of flexural zones.6 The patient described in this case did not have IP because IP would be more likely to present as a well-demarcated erythematous plaque rather than a patch.
Histologically, IP shows regular psoriasiform acanthosis and hypogranulosis of the epidermis, Munro microabscess, spongiform pustules of Kogoj, dilated tortuous dermal vessels, and thinning of the suprapapillary plates.5
Lichen planus pigmentosus-inversus (LPPI) is also known as lichen planus pigmentosus—intertriginous variant. This variant of lichen planus pigmentosus presents as multiple gray to dark brown macules and patches with poorly defined borders in a linear distribution limited to intertriginous areas, flexural surfaces, or following the lines of Blaschko.7 About 20% of cases present with frontal fibrosing alopecia. It is most common in individuals with intermediate and darker skin pigmentation, has a higher prevalence in females, and typically occurs within the third and fifth decades of life. Friction is a common trigger of LPPI.7 A diagnosis of LPPI is incorrect because the lesions would present as gray to dark brown macules, as opposed to the shiny white atrophic thin papules with surrounding pink and purple patches seen in this case.
Histologically, while both LS and LPPI share band-like lymphocytic infiltrate and basal cell vacuolization, findings in the dermis differ. LPPI shows melanophages and prominent melanin incontinence, while LS shows homogenization and sclerosis of superficial collagen.1,8 LPPI also shows absence of compensatory keratinocyte proliferation.
Morphea is an inflammatory disease that affects the dermis and subcutaneous fat, resulting in sclerosis that appears scarlike. Its prevalence increases with age and has a 4:1 prevalence in females, with the plaque type being the most common variant. 9 The typical presentation of plaque-type morphea is an insidious onset of asymptomatic, slightly elevated, erythematous or violaceous, slightly edematous plaques with centrifugal expansion. The center of the plaque may become sclerotic and indurated, acquiring a shiny white color with a peripheral “lilac” ring. Trunk and upper extremity involvement is common. Morphea is associated with increased antisingle-stranded DNA, antitopoisomerase IIa, antiphospholipid, antifibrillin-1, and antihistone antibodies. Triggers of morphea are believed to be localized insults to the skin, including mechanical trauma, injections, vaccinations, and irradiation.9 This answer is incorrect because the patient’s lesions were pruritic and had genital involvement, which are not typical of morphea. Morphea can be differentiated with based on symptoms (lack of pruritus, pain, burning), morphology of lesions (induration versus atrophy), dermoscopy (fibrotic beams with less scale and hemorrhage vs keratotic follicular plugs), and histopathology (depth of inflammation in superficial and deep dermis).
Histology of morphea can differ based on the stage, whether the lesion is sampled in the inflammatory margin or central sclerosis, and the depth of affected skin. At the inflammatory margin, vascular changes, including endothelial swelling and edema, are present, as well as CD4+ T cells, eosinophils, plasma cells, and mast cells surrounding smaller blood vessels. In late stages, the inflammatory infiltrate is no longer present, the epidermis appears regular, and there is a flattened dermal-epidermal junction. Distinct features include homogenous collagen bundles that replace many dermal structures, with atrophic eccrine glands that appear “trapped” in the thickened dermis, and homogenized and hyalinized subcutis.9
Mycosis fungoides (MF) is the most common type of cutaneous T-cell lymphoma and presents as annular, erythematous or hypopigmented patches and plaques with fine scale and tumors on the buttocks and sun-protected areas of the limbs and trunk. Lesions can appear with prominent poikiloderma or atrophic or lichenified skin.10 It is most common in males of African descent aged 50 to 55 years. The etiology is largely unknown but believed to be multifactorial. This answer is incorrect because the lesions in this patient appeared more atrophic, were less well demarcated, and lacked the scale that would be present in MF.
On histology, both LS and MF show band-like lymphocytic infiltrate, however MF lacks the homogenization and sclerosis of superficial collagen that is present in the dermis of LS. Also, MF demonstrates epidermotropism of atypical lymphocytes forming Pautrier microabscess.10
Primary Care Role
Primary care physicians can diagnose and treat LS. Referral to dermatology is not mandatory. Note that topical steroids can be used daily for up to 12 weeks. In LS, early treatment is associated with improved outcomes and minimizes the risk of irreversible skin changes.11 Follow-up during the treatment period is recommended to monitor subjective and objective response to treatment. Follow-up after the initial treatment is recommended since LS is typically chronic, can relapse, and SCC can infrequently arise from LS lesions.11
- Tran DA, Tan X, Macri CJ, Goldstein AT, Fu SW. Lichen sclerosus: an autoimmunopathogenic and genomic enigma with emerging genetic and immune targets. Int J Biol Sci. 2019;15:1429-1439. doi:10.7150/ijbs.34613
- De Luca DA, Papara C, Vorobyev A, et al. Lichen sclerosus: the 2023 update. Front Med (Lausanne). 2023;10:1106318. doi:10.3389/fmed.2023.1106318
- Kuraitis D, Murina A. Squamous cell carcinoma arising in chronic inflammatory dermatoses. Cutis. 2024;113:29-34. doi:10.12788/cutis.0914
- Gaertner E, Elstein W. Lichen planus pigmentosus-inversus: case report and review of an unusual entity. Dermatol Online J. 2012;18:11.
- Micali G, Verzì AE, Giuffrida G, et al. Inverse psoriasis: from diagnosis to current treatment options. Clin Cosmet Investig Dermatol. 2019;12:953-959. doi:10.2147/CCID.S189000
- Syed ZU, Khachemoune A. Inverse psoriasis: case presentation and review. Am J Clin Dermatol. 2011;12:143-146. doi:10.2165/11532060-000000000-00000
- Robles-Méndez JC, Rizo-Frías P, Herz-Ruelas ME, et al. Lichen planus pigmentosus and its variants: review and update. Int J Dermatol. 2018;57:505-514. doi:10.1111/ijd.13806
- Vinay K, Kumar S, Bishnoi A, et al. A clinico-demographic study of 344 patients with lichen planus pigmentosus seen in a tertiary care center in India over an 8-year period. Int J Dermatol. 2020;59:245-252. doi:10.1111/ijd.14540
- Papara C, De Luca DA, Bieber K, et al. Morphea: the 2023 update. Front Med (Lausanne). 2023;10:1108623. doi:10.3389/fmed.2023.1108623
- Zinzani PL, Ferreri AJ, Cerroni L. Mycosis fungoides. Cri t Rev Oncol Hematol. 2008;65:172-182. doi:10.1016/j.critrevonc.2007.08.004
- Lee A, Bradford J, Fischer G. Long-term management of adult vulvar lichen sclerosus: a prospective cohort study of 507 women. JAMA Dermatol. 2015;151(10):1061-1067. doi:10.1001/jamadermatol.2015.0643
Discussion
A diagnosis of lichen sclerosus (LS) was made based on clinical and dermoscopic features, followed by confirmation with histology. The patient’s presentation included typical signs and symptoms of LS: itching, burning, intermittent bleeding, perianal hemorrhage, fusion of the clitoral head, and fissures. Other presentations can include dyspareunia, erosions, and excoriations; however, these symptoms and signs were not reported or seen in this patient.
LS typically affects the anogenital region and has 2 peak incidences: in preadolescent teens and during the fifth to sixth decade of life.1 This patient presented with a case of extragenital LS, which is less common than the classic presentation of LS that affects the genitals. This variant’s epidemiology differs, as it is less common in children and more common in postmenopausal women.2 Extragenital LS presents as white, atrophic plaques with a predilection for sites including the trunk, breasts, upper arms, and sites of physical trauma, with symptoms of dryness and pruritus. Over time, the papules can coalesce and form ivory, scar-like papules or plaques with a wrinkled surface. In advanced stages, telangiectasia or follicular plugging can be present, along with flattening of the dermal-epidermal junction. This flat interface is fragile and can result in bullae that may become hemorrhagic.
Cutaneous squamous cell carcinoma (SCC) may infrequently arise from LS, similar to other chronic inflammatory dermatoses.3 Lichen planus is typically not associated with an increased risk of SCC, except in the oral and hypertrophic variants. However, LS may be considered a premalignant process, and many vulvar SCC cases are noted to have adjacent LS lesions.3
Autoimmune and genetic factors contribute to the pathogenesis of LS. Extracellular matrix protein 1 (ECM1) binds molecules of the basement membrane zone and dermis, contributing to the structure and integrity of skin. Autoantibodies against ECM1 and other antigens of the basement membrane zone, including BP180 and BP320, were found in LS.2 HLA-DQ7 major histocompatibility complex class II antigens have been associated with LS.1
On histologic examination, the epidermis of LS is atrophic with hyperkeratosis. The dermis shows homogenization and sclerosis of superficial collagen with a band-like lymphocytic infiltrate below the sclerosis. The basal layer is thickened, showing basal cell vacuolization and hydropic degeneration.4
First-line treatment for genital and extragenital variants of LS is high-potency topical steroids for 3 months or until the skin texture and color resolve (ie, clobetasol 0.05% cream or ointment). The second-line treatment is a topical calcineurin inhibitor. These treatments are used for management. They are not cures for LS, as relapse is possible after the initial treatment course is completed. Adverse effects of high potency topical steroids are skin burning, skin atrophy, and fragility, telangiectasia. The adverse effects of topical calcineurin inhibitors are stinging and burning on application.
Other Diagnostic Considerations
Inverse psoriasis (IP) is a variant of psoriasis that presents as erythematous, well-demarcated plaques with minimal scale in intertriginous areas and flexural surfaces. Localized dermatophyte, candidal, or bacterial infections can trigger IP.5 It occurs in about 3% to 7% of patients with plaque psoriasis and is thought to form due to koebnerization via mechanical friction of flexural zones.6 The patient described in this case did not have IP because IP would be more likely to present as a well-demarcated erythematous plaque rather than a patch.
Histologically, IP shows regular psoriasiform acanthosis and hypogranulosis of the epidermis, Munro microabscess, spongiform pustules of Kogoj, dilated tortuous dermal vessels, and thinning of the suprapapillary plates.5
Lichen planus pigmentosus-inversus (LPPI) is also known as lichen planus pigmentosus—intertriginous variant. This variant of lichen planus pigmentosus presents as multiple gray to dark brown macules and patches with poorly defined borders in a linear distribution limited to intertriginous areas, flexural surfaces, or following the lines of Blaschko.7 About 20% of cases present with frontal fibrosing alopecia. It is most common in individuals with intermediate and darker skin pigmentation, has a higher prevalence in females, and typically occurs within the third and fifth decades of life. Friction is a common trigger of LPPI.7 A diagnosis of LPPI is incorrect because the lesions would present as gray to dark brown macules, as opposed to the shiny white atrophic thin papules with surrounding pink and purple patches seen in this case.
Histologically, while both LS and LPPI share band-like lymphocytic infiltrate and basal cell vacuolization, findings in the dermis differ. LPPI shows melanophages and prominent melanin incontinence, while LS shows homogenization and sclerosis of superficial collagen.1,8 LPPI also shows absence of compensatory keratinocyte proliferation.
Morphea is an inflammatory disease that affects the dermis and subcutaneous fat, resulting in sclerosis that appears scarlike. Its prevalence increases with age and has a 4:1 prevalence in females, with the plaque type being the most common variant. 9 The typical presentation of plaque-type morphea is an insidious onset of asymptomatic, slightly elevated, erythematous or violaceous, slightly edematous plaques with centrifugal expansion. The center of the plaque may become sclerotic and indurated, acquiring a shiny white color with a peripheral “lilac” ring. Trunk and upper extremity involvement is common. Morphea is associated with increased antisingle-stranded DNA, antitopoisomerase IIa, antiphospholipid, antifibrillin-1, and antihistone antibodies. Triggers of morphea are believed to be localized insults to the skin, including mechanical trauma, injections, vaccinations, and irradiation.9 This answer is incorrect because the patient’s lesions were pruritic and had genital involvement, which are not typical of morphea. Morphea can be differentiated with based on symptoms (lack of pruritus, pain, burning), morphology of lesions (induration versus atrophy), dermoscopy (fibrotic beams with less scale and hemorrhage vs keratotic follicular plugs), and histopathology (depth of inflammation in superficial and deep dermis).
Histology of morphea can differ based on the stage, whether the lesion is sampled in the inflammatory margin or central sclerosis, and the depth of affected skin. At the inflammatory margin, vascular changes, including endothelial swelling and edema, are present, as well as CD4+ T cells, eosinophils, plasma cells, and mast cells surrounding smaller blood vessels. In late stages, the inflammatory infiltrate is no longer present, the epidermis appears regular, and there is a flattened dermal-epidermal junction. Distinct features include homogenous collagen bundles that replace many dermal structures, with atrophic eccrine glands that appear “trapped” in the thickened dermis, and homogenized and hyalinized subcutis.9
Mycosis fungoides (MF) is the most common type of cutaneous T-cell lymphoma and presents as annular, erythematous or hypopigmented patches and plaques with fine scale and tumors on the buttocks and sun-protected areas of the limbs and trunk. Lesions can appear with prominent poikiloderma or atrophic or lichenified skin.10 It is most common in males of African descent aged 50 to 55 years. The etiology is largely unknown but believed to be multifactorial. This answer is incorrect because the lesions in this patient appeared more atrophic, were less well demarcated, and lacked the scale that would be present in MF.
On histology, both LS and MF show band-like lymphocytic infiltrate, however MF lacks the homogenization and sclerosis of superficial collagen that is present in the dermis of LS. Also, MF demonstrates epidermotropism of atypical lymphocytes forming Pautrier microabscess.10
Primary Care Role
Primary care physicians can diagnose and treat LS. Referral to dermatology is not mandatory. Note that topical steroids can be used daily for up to 12 weeks. In LS, early treatment is associated with improved outcomes and minimizes the risk of irreversible skin changes.11 Follow-up during the treatment period is recommended to monitor subjective and objective response to treatment. Follow-up after the initial treatment is recommended since LS is typically chronic, can relapse, and SCC can infrequently arise from LS lesions.11
Discussion
A diagnosis of lichen sclerosus (LS) was made based on clinical and dermoscopic features, followed by confirmation with histology. The patient’s presentation included typical signs and symptoms of LS: itching, burning, intermittent bleeding, perianal hemorrhage, fusion of the clitoral head, and fissures. Other presentations can include dyspareunia, erosions, and excoriations; however, these symptoms and signs were not reported or seen in this patient.
LS typically affects the anogenital region and has 2 peak incidences: in preadolescent teens and during the fifth to sixth decade of life.1 This patient presented with a case of extragenital LS, which is less common than the classic presentation of LS that affects the genitals. This variant’s epidemiology differs, as it is less common in children and more common in postmenopausal women.2 Extragenital LS presents as white, atrophic plaques with a predilection for sites including the trunk, breasts, upper arms, and sites of physical trauma, with symptoms of dryness and pruritus. Over time, the papules can coalesce and form ivory, scar-like papules or plaques with a wrinkled surface. In advanced stages, telangiectasia or follicular plugging can be present, along with flattening of the dermal-epidermal junction. This flat interface is fragile and can result in bullae that may become hemorrhagic.
Cutaneous squamous cell carcinoma (SCC) may infrequently arise from LS, similar to other chronic inflammatory dermatoses.3 Lichen planus is typically not associated with an increased risk of SCC, except in the oral and hypertrophic variants. However, LS may be considered a premalignant process, and many vulvar SCC cases are noted to have adjacent LS lesions.3
Autoimmune and genetic factors contribute to the pathogenesis of LS. Extracellular matrix protein 1 (ECM1) binds molecules of the basement membrane zone and dermis, contributing to the structure and integrity of skin. Autoantibodies against ECM1 and other antigens of the basement membrane zone, including BP180 and BP320, were found in LS.2 HLA-DQ7 major histocompatibility complex class II antigens have been associated with LS.1
On histologic examination, the epidermis of LS is atrophic with hyperkeratosis. The dermis shows homogenization and sclerosis of superficial collagen with a band-like lymphocytic infiltrate below the sclerosis. The basal layer is thickened, showing basal cell vacuolization and hydropic degeneration.4
First-line treatment for genital and extragenital variants of LS is high-potency topical steroids for 3 months or until the skin texture and color resolve (ie, clobetasol 0.05% cream or ointment). The second-line treatment is a topical calcineurin inhibitor. These treatments are used for management. They are not cures for LS, as relapse is possible after the initial treatment course is completed. Adverse effects of high potency topical steroids are skin burning, skin atrophy, and fragility, telangiectasia. The adverse effects of topical calcineurin inhibitors are stinging and burning on application.
Other Diagnostic Considerations
Inverse psoriasis (IP) is a variant of psoriasis that presents as erythematous, well-demarcated plaques with minimal scale in intertriginous areas and flexural surfaces. Localized dermatophyte, candidal, or bacterial infections can trigger IP.5 It occurs in about 3% to 7% of patients with plaque psoriasis and is thought to form due to koebnerization via mechanical friction of flexural zones.6 The patient described in this case did not have IP because IP would be more likely to present as a well-demarcated erythematous plaque rather than a patch.
Histologically, IP shows regular psoriasiform acanthosis and hypogranulosis of the epidermis, Munro microabscess, spongiform pustules of Kogoj, dilated tortuous dermal vessels, and thinning of the suprapapillary plates.5
Lichen planus pigmentosus-inversus (LPPI) is also known as lichen planus pigmentosus—intertriginous variant. This variant of lichen planus pigmentosus presents as multiple gray to dark brown macules and patches with poorly defined borders in a linear distribution limited to intertriginous areas, flexural surfaces, or following the lines of Blaschko.7 About 20% of cases present with frontal fibrosing alopecia. It is most common in individuals with intermediate and darker skin pigmentation, has a higher prevalence in females, and typically occurs within the third and fifth decades of life. Friction is a common trigger of LPPI.7 A diagnosis of LPPI is incorrect because the lesions would present as gray to dark brown macules, as opposed to the shiny white atrophic thin papules with surrounding pink and purple patches seen in this case.
Histologically, while both LS and LPPI share band-like lymphocytic infiltrate and basal cell vacuolization, findings in the dermis differ. LPPI shows melanophages and prominent melanin incontinence, while LS shows homogenization and sclerosis of superficial collagen.1,8 LPPI also shows absence of compensatory keratinocyte proliferation.
Morphea is an inflammatory disease that affects the dermis and subcutaneous fat, resulting in sclerosis that appears scarlike. Its prevalence increases with age and has a 4:1 prevalence in females, with the plaque type being the most common variant. 9 The typical presentation of plaque-type morphea is an insidious onset of asymptomatic, slightly elevated, erythematous or violaceous, slightly edematous plaques with centrifugal expansion. The center of the plaque may become sclerotic and indurated, acquiring a shiny white color with a peripheral “lilac” ring. Trunk and upper extremity involvement is common. Morphea is associated with increased antisingle-stranded DNA, antitopoisomerase IIa, antiphospholipid, antifibrillin-1, and antihistone antibodies. Triggers of morphea are believed to be localized insults to the skin, including mechanical trauma, injections, vaccinations, and irradiation.9 This answer is incorrect because the patient’s lesions were pruritic and had genital involvement, which are not typical of morphea. Morphea can be differentiated with based on symptoms (lack of pruritus, pain, burning), morphology of lesions (induration versus atrophy), dermoscopy (fibrotic beams with less scale and hemorrhage vs keratotic follicular plugs), and histopathology (depth of inflammation in superficial and deep dermis).
Histology of morphea can differ based on the stage, whether the lesion is sampled in the inflammatory margin or central sclerosis, and the depth of affected skin. At the inflammatory margin, vascular changes, including endothelial swelling and edema, are present, as well as CD4+ T cells, eosinophils, plasma cells, and mast cells surrounding smaller blood vessels. In late stages, the inflammatory infiltrate is no longer present, the epidermis appears regular, and there is a flattened dermal-epidermal junction. Distinct features include homogenous collagen bundles that replace many dermal structures, with atrophic eccrine glands that appear “trapped” in the thickened dermis, and homogenized and hyalinized subcutis.9
Mycosis fungoides (MF) is the most common type of cutaneous T-cell lymphoma and presents as annular, erythematous or hypopigmented patches and plaques with fine scale and tumors on the buttocks and sun-protected areas of the limbs and trunk. Lesions can appear with prominent poikiloderma or atrophic or lichenified skin.10 It is most common in males of African descent aged 50 to 55 years. The etiology is largely unknown but believed to be multifactorial. This answer is incorrect because the lesions in this patient appeared more atrophic, were less well demarcated, and lacked the scale that would be present in MF.
On histology, both LS and MF show band-like lymphocytic infiltrate, however MF lacks the homogenization and sclerosis of superficial collagen that is present in the dermis of LS. Also, MF demonstrates epidermotropism of atypical lymphocytes forming Pautrier microabscess.10
Primary Care Role
Primary care physicians can diagnose and treat LS. Referral to dermatology is not mandatory. Note that topical steroids can be used daily for up to 12 weeks. In LS, early treatment is associated with improved outcomes and minimizes the risk of irreversible skin changes.11 Follow-up during the treatment period is recommended to monitor subjective and objective response to treatment. Follow-up after the initial treatment is recommended since LS is typically chronic, can relapse, and SCC can infrequently arise from LS lesions.11
- Tran DA, Tan X, Macri CJ, Goldstein AT, Fu SW. Lichen sclerosus: an autoimmunopathogenic and genomic enigma with emerging genetic and immune targets. Int J Biol Sci. 2019;15:1429-1439. doi:10.7150/ijbs.34613
- De Luca DA, Papara C, Vorobyev A, et al. Lichen sclerosus: the 2023 update. Front Med (Lausanne). 2023;10:1106318. doi:10.3389/fmed.2023.1106318
- Kuraitis D, Murina A. Squamous cell carcinoma arising in chronic inflammatory dermatoses. Cutis. 2024;113:29-34. doi:10.12788/cutis.0914
- Gaertner E, Elstein W. Lichen planus pigmentosus-inversus: case report and review of an unusual entity. Dermatol Online J. 2012;18:11.
- Micali G, Verzì AE, Giuffrida G, et al. Inverse psoriasis: from diagnosis to current treatment options. Clin Cosmet Investig Dermatol. 2019;12:953-959. doi:10.2147/CCID.S189000
- Syed ZU, Khachemoune A. Inverse psoriasis: case presentation and review. Am J Clin Dermatol. 2011;12:143-146. doi:10.2165/11532060-000000000-00000
- Robles-Méndez JC, Rizo-Frías P, Herz-Ruelas ME, et al. Lichen planus pigmentosus and its variants: review and update. Int J Dermatol. 2018;57:505-514. doi:10.1111/ijd.13806
- Vinay K, Kumar S, Bishnoi A, et al. A clinico-demographic study of 344 patients with lichen planus pigmentosus seen in a tertiary care center in India over an 8-year period. Int J Dermatol. 2020;59:245-252. doi:10.1111/ijd.14540
- Papara C, De Luca DA, Bieber K, et al. Morphea: the 2023 update. Front Med (Lausanne). 2023;10:1108623. doi:10.3389/fmed.2023.1108623
- Zinzani PL, Ferreri AJ, Cerroni L. Mycosis fungoides. Cri t Rev Oncol Hematol. 2008;65:172-182. doi:10.1016/j.critrevonc.2007.08.004
- Lee A, Bradford J, Fischer G. Long-term management of adult vulvar lichen sclerosus: a prospective cohort study of 507 women. JAMA Dermatol. 2015;151(10):1061-1067. doi:10.1001/jamadermatol.2015.0643
- Tran DA, Tan X, Macri CJ, Goldstein AT, Fu SW. Lichen sclerosus: an autoimmunopathogenic and genomic enigma with emerging genetic and immune targets. Int J Biol Sci. 2019;15:1429-1439. doi:10.7150/ijbs.34613
- De Luca DA, Papara C, Vorobyev A, et al. Lichen sclerosus: the 2023 update. Front Med (Lausanne). 2023;10:1106318. doi:10.3389/fmed.2023.1106318
- Kuraitis D, Murina A. Squamous cell carcinoma arising in chronic inflammatory dermatoses. Cutis. 2024;113:29-34. doi:10.12788/cutis.0914
- Gaertner E, Elstein W. Lichen planus pigmentosus-inversus: case report and review of an unusual entity. Dermatol Online J. 2012;18:11.
- Micali G, Verzì AE, Giuffrida G, et al. Inverse psoriasis: from diagnosis to current treatment options. Clin Cosmet Investig Dermatol. 2019;12:953-959. doi:10.2147/CCID.S189000
- Syed ZU, Khachemoune A. Inverse psoriasis: case presentation and review. Am J Clin Dermatol. 2011;12:143-146. doi:10.2165/11532060-000000000-00000
- Robles-Méndez JC, Rizo-Frías P, Herz-Ruelas ME, et al. Lichen planus pigmentosus and its variants: review and update. Int J Dermatol. 2018;57:505-514. doi:10.1111/ijd.13806
- Vinay K, Kumar S, Bishnoi A, et al. A clinico-demographic study of 344 patients with lichen planus pigmentosus seen in a tertiary care center in India over an 8-year period. Int J Dermatol. 2020;59:245-252. doi:10.1111/ijd.14540
- Papara C, De Luca DA, Bieber K, et al. Morphea: the 2023 update. Front Med (Lausanne). 2023;10:1108623. doi:10.3389/fmed.2023.1108623
- Zinzani PL, Ferreri AJ, Cerroni L. Mycosis fungoides. Cri t Rev Oncol Hematol. 2008;65:172-182. doi:10.1016/j.critrevonc.2007.08.004
- Lee A, Bradford J, Fischer G. Long-term management of adult vulvar lichen sclerosus: a prospective cohort study of 507 women. JAMA Dermatol. 2015;151(10):1061-1067. doi:10.1001/jamadermatol.2015.0643
Atrophic Areas on the Axillary and Anogenital Anatomy
Atrophic Areas on the Axillary and Anogenital Anatomy
A 62-year-old woman presented for a fullbody skin examination and was found to have a rash in her axillae and inframammary regions. The rash was intermittently pruritic, and the patient felt that the inframammary rash had started from contact with brassiere underwires. She had no oral lesions but noted intermittent burning and itching of the vaginal folds and intermittent bleeding near her anus. Physical examination revealed confluent, shiny, white, atrophic, thin papules with surrounding pink and purple patches on bilateral axillae, bilateral inframammary folds, bilateral inner thighs, and on the clitoral hood and labia minora. There was also an hourglass-shaped erythematous patch involving the vagina and anus. A small fissure was noted perianally, and small hemorrhage was noted on the clitoral head, with fusion of the clitoral head and superior labia minora (Figures 1 and 2).
lesion from punch biopsy of the patient’s left axilla.
sclerosus plaque showing bright white grouped dots
on a pink background with follicular plugging and linear
branching vessels.
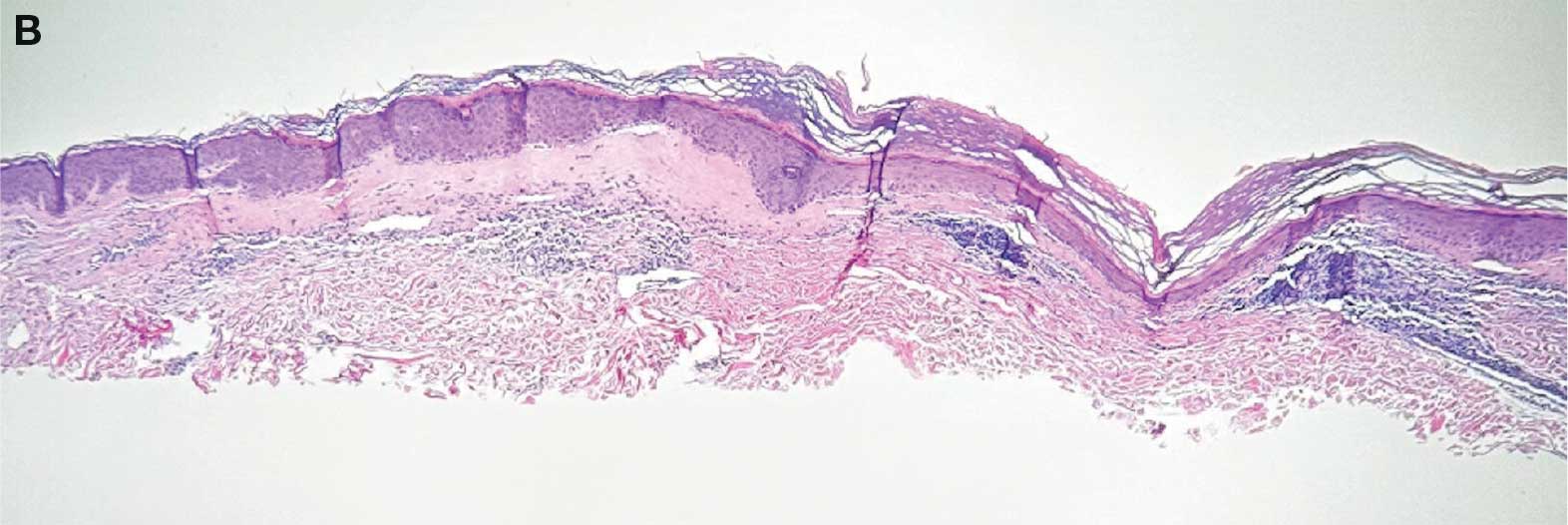
showing a compact corneal layer with a pale papillary
dermis and an underlying lymphocytic infiltrate. These
findings give the “red, white, and blue” appearance.
Low power 20× magnification.
nsbp;
Does Ethnicity Affect Skin Cancer Risk?
Does Ethnicity Affect Skin Cancer Risk?
TOPLINE:
The incidence of skin cancer in England varied by ethnicity: White individuals had higher rates of melanoma, cutaneous squamous cell carcinoma, and basal cell carcinoma than Asian or Black individuals. In contrast, acral lentiginous melanoma was most common among Black individuals, whereas cutaneous T-cell lymphoma and Kaposi sarcoma were highest among those in the "Other" ethnic group.
METHODOLOGY:
- Researchers analysed all cases of cutaneous melanoma (melanoma and acral lentiginous melanoma), basal cell carcinoma, cutaneous squamous cell carcinoma, cutaneous T-cell lymphoma, and Kaposi sarcoma using data from the NHS National Disease Registration Service cancer registry between 2013 and 2020.
- Data collection incorporated ethnicity information from multiple health care datasets, including Clinical Outcomes and Services Dataset, Patient Administration System, Radiotherapy Dataset, Diagnostic Imaging Dataset, and Hospital Episode Statistics.
- A population analysis categorised patients into 7 standardised ethnic groups (on the basis of Office for National Statistics classifications): White, Asian, Chinese, Black, mixed, other, and unknown groups, with ethnicity data being self-reported by patients.
- Outcomes included European age-standardised rates calculated using the 2013 European Standard Population and reported per 100,000 person-years (PYs).
TAKEAWAY:
- White Individuals had 13-fold higher rates of cutaneous squamous cell carcinoma (61.75 per 100,000 PYs), 26-fold and 27-fold higher rates of basal cell carcinoma (153.69 per 100,000 PYs), and 33-fold and 16-fold higher rates of cutaneous melanoma (27.29 per 100,000 PYs) than Asian and Black individuals, respectively.
- Black individuals had the highest incidence of acral lentiginous melanoma (0.85 per 100,000 PYs), and those in the other ethnic group had the highest incidence of cutaneous T-cell lymphoma (1.74 per 100,000 PYs) and Kaposi sarcoma (1.57 per 100,000 PYs).
- The presentation of early-stage melanoma was low among Asian (53.5%), Black (62.4%), mixed (62.5%), and other (76.4%) ethnic groups compared to that among White ethnicities (79.8%).
- Acral lentiginous melanomas were less likely to get urgent suspected cancer pathway referrals than overall melanoma (40.1% vs 44.6%; P < .001) and more likely to be diagnosed late than overall melanoma (stage I/II at diagnosis; 72% vs 80%; P < .0001).
IN PRACTICE:
"The findings emphasise the need for better, targeted ethnicity data collection strategies to address incidence, outcomes and health care equity for not just skin cancer but all health conditions in underserved populations," the authors wrote. "While projects like the Global Burden of Disease have improved global health care reporting, continuous audit and improvement of collected data are essential to provide better care across people of all ethnicities."
SOURCE:
This study was led by Shehnaz Ahmed, British Association of Dermatologists, London, England. It was published online on September 10, 2025, in the British Journal of Dermatology.
LIMITATIONS:
Census data collection after every 10 years could have contributed to inaccurate population estimates and incidence rates. Small sample sizes in certain ethnic groups could have led to potential confounders, requiring a cautious interpretation of relative incidence. The NHS data included only self-reported ethnicity data with no available details of skin phototypes, skin tones, or racial ancestry. This study lacked granular ethnicity census data and stage data for basal cell carcinoma, cutaneous small cell carcinoma, and Kaposi sarcoma.
DISCLOSURES:
This research was supported through a partnership between the British Association of Dermatologists and NHS England's National Disease Registration Service. Two authors reported being employees of the British Association of Dermatologists.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
The incidence of skin cancer in England varied by ethnicity: White individuals had higher rates of melanoma, cutaneous squamous cell carcinoma, and basal cell carcinoma than Asian or Black individuals. In contrast, acral lentiginous melanoma was most common among Black individuals, whereas cutaneous T-cell lymphoma and Kaposi sarcoma were highest among those in the "Other" ethnic group.
METHODOLOGY:
- Researchers analysed all cases of cutaneous melanoma (melanoma and acral lentiginous melanoma), basal cell carcinoma, cutaneous squamous cell carcinoma, cutaneous T-cell lymphoma, and Kaposi sarcoma using data from the NHS National Disease Registration Service cancer registry between 2013 and 2020.
- Data collection incorporated ethnicity information from multiple health care datasets, including Clinical Outcomes and Services Dataset, Patient Administration System, Radiotherapy Dataset, Diagnostic Imaging Dataset, and Hospital Episode Statistics.
- A population analysis categorised patients into 7 standardised ethnic groups (on the basis of Office for National Statistics classifications): White, Asian, Chinese, Black, mixed, other, and unknown groups, with ethnicity data being self-reported by patients.
- Outcomes included European age-standardised rates calculated using the 2013 European Standard Population and reported per 100,000 person-years (PYs).
TAKEAWAY:
- White Individuals had 13-fold higher rates of cutaneous squamous cell carcinoma (61.75 per 100,000 PYs), 26-fold and 27-fold higher rates of basal cell carcinoma (153.69 per 100,000 PYs), and 33-fold and 16-fold higher rates of cutaneous melanoma (27.29 per 100,000 PYs) than Asian and Black individuals, respectively.
- Black individuals had the highest incidence of acral lentiginous melanoma (0.85 per 100,000 PYs), and those in the other ethnic group had the highest incidence of cutaneous T-cell lymphoma (1.74 per 100,000 PYs) and Kaposi sarcoma (1.57 per 100,000 PYs).
- The presentation of early-stage melanoma was low among Asian (53.5%), Black (62.4%), mixed (62.5%), and other (76.4%) ethnic groups compared to that among White ethnicities (79.8%).
- Acral lentiginous melanomas were less likely to get urgent suspected cancer pathway referrals than overall melanoma (40.1% vs 44.6%; P < .001) and more likely to be diagnosed late than overall melanoma (stage I/II at diagnosis; 72% vs 80%; P < .0001).
IN PRACTICE:
"The findings emphasise the need for better, targeted ethnicity data collection strategies to address incidence, outcomes and health care equity for not just skin cancer but all health conditions in underserved populations," the authors wrote. "While projects like the Global Burden of Disease have improved global health care reporting, continuous audit and improvement of collected data are essential to provide better care across people of all ethnicities."
SOURCE:
This study was led by Shehnaz Ahmed, British Association of Dermatologists, London, England. It was published online on September 10, 2025, in the British Journal of Dermatology.
LIMITATIONS:
Census data collection after every 10 years could have contributed to inaccurate population estimates and incidence rates. Small sample sizes in certain ethnic groups could have led to potential confounders, requiring a cautious interpretation of relative incidence. The NHS data included only self-reported ethnicity data with no available details of skin phototypes, skin tones, or racial ancestry. This study lacked granular ethnicity census data and stage data for basal cell carcinoma, cutaneous small cell carcinoma, and Kaposi sarcoma.
DISCLOSURES:
This research was supported through a partnership between the British Association of Dermatologists and NHS England's National Disease Registration Service. Two authors reported being employees of the British Association of Dermatologists.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
The incidence of skin cancer in England varied by ethnicity: White individuals had higher rates of melanoma, cutaneous squamous cell carcinoma, and basal cell carcinoma than Asian or Black individuals. In contrast, acral lentiginous melanoma was most common among Black individuals, whereas cutaneous T-cell lymphoma and Kaposi sarcoma were highest among those in the "Other" ethnic group.
METHODOLOGY:
- Researchers analysed all cases of cutaneous melanoma (melanoma and acral lentiginous melanoma), basal cell carcinoma, cutaneous squamous cell carcinoma, cutaneous T-cell lymphoma, and Kaposi sarcoma using data from the NHS National Disease Registration Service cancer registry between 2013 and 2020.
- Data collection incorporated ethnicity information from multiple health care datasets, including Clinical Outcomes and Services Dataset, Patient Administration System, Radiotherapy Dataset, Diagnostic Imaging Dataset, and Hospital Episode Statistics.
- A population analysis categorised patients into 7 standardised ethnic groups (on the basis of Office for National Statistics classifications): White, Asian, Chinese, Black, mixed, other, and unknown groups, with ethnicity data being self-reported by patients.
- Outcomes included European age-standardised rates calculated using the 2013 European Standard Population and reported per 100,000 person-years (PYs).
TAKEAWAY:
- White Individuals had 13-fold higher rates of cutaneous squamous cell carcinoma (61.75 per 100,000 PYs), 26-fold and 27-fold higher rates of basal cell carcinoma (153.69 per 100,000 PYs), and 33-fold and 16-fold higher rates of cutaneous melanoma (27.29 per 100,000 PYs) than Asian and Black individuals, respectively.
- Black individuals had the highest incidence of acral lentiginous melanoma (0.85 per 100,000 PYs), and those in the other ethnic group had the highest incidence of cutaneous T-cell lymphoma (1.74 per 100,000 PYs) and Kaposi sarcoma (1.57 per 100,000 PYs).
- The presentation of early-stage melanoma was low among Asian (53.5%), Black (62.4%), mixed (62.5%), and other (76.4%) ethnic groups compared to that among White ethnicities (79.8%).
- Acral lentiginous melanomas were less likely to get urgent suspected cancer pathway referrals than overall melanoma (40.1% vs 44.6%; P < .001) and more likely to be diagnosed late than overall melanoma (stage I/II at diagnosis; 72% vs 80%; P < .0001).
IN PRACTICE:
"The findings emphasise the need for better, targeted ethnicity data collection strategies to address incidence, outcomes and health care equity for not just skin cancer but all health conditions in underserved populations," the authors wrote. "While projects like the Global Burden of Disease have improved global health care reporting, continuous audit and improvement of collected data are essential to provide better care across people of all ethnicities."
SOURCE:
This study was led by Shehnaz Ahmed, British Association of Dermatologists, London, England. It was published online on September 10, 2025, in the British Journal of Dermatology.
LIMITATIONS:
Census data collection after every 10 years could have contributed to inaccurate population estimates and incidence rates. Small sample sizes in certain ethnic groups could have led to potential confounders, requiring a cautious interpretation of relative incidence. The NHS data included only self-reported ethnicity data with no available details of skin phototypes, skin tones, or racial ancestry. This study lacked granular ethnicity census data and stage data for basal cell carcinoma, cutaneous small cell carcinoma, and Kaposi sarcoma.
DISCLOSURES:
This research was supported through a partnership between the British Association of Dermatologists and NHS England's National Disease Registration Service. Two authors reported being employees of the British Association of Dermatologists.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Does Ethnicity Affect Skin Cancer Risk?
Does Ethnicity Affect Skin Cancer Risk?
Consider Cultural Practices and Barriers to Care When Treating Alopecia Areata
Consider Cultural Practices and Barriers to Care When Treating Alopecia Areata
The Comparison
A. Alopecia areata in a young girl with a lighter skin tone. The fine white vellus hairs are signs of regrowth.
B. Alopecia areata in a 49-year-old man with tightly coiled hair and darker skin tone. Coiled white hairs are noted in the alopecia patches.
young girl with a lighter skin
tone. The fine white vellus
hairs are signs of regrowth. Photographs courtesy of
Richard P. Usatine, MD.

49-year-old man with tightly
coiled hair and darker skin
tone. Coiled white hairs
are noted in the alopecia
patches. Photographs courtesy of
Richard P. Usatine, MD.
Alopecia areata (AA) is a common autoimmune condition characterized by hair loss resulting from a T cell–mediated attack on the hair follicles. It manifests as nonscarring patches of hair loss on the scalp, eyebrows, eyelashes, and beard area as well as more extensive complete loss of scalp and body hair. While AA may affect individuals of any age, most patients develop their first patch(es) of hair loss during childhood.1 The treatment landscape for AA has evolved considerably in recent years, but barriers to access to newer treatments persist.
Epidemiology
AA is most prevalent among pediatric and adult individuals of African, Asian, or Hispanic/Latino descent.2-4 In some studies, Black individuals had higher odds and Asian individuals had lower odds of developing AA, while other studies have reported the highest standardized prevalence among Asian individuals.5 In the United States, AA affects about 1.47% of adults and as many as 0.11% of children.6-8 In Black patients, AA often manifests early with a female predominance.5
AA frequently is associated with autoimmune comorbidities, the most common being thyroid disease.3,5 In Black patients, AA is associated with more atopic comorbidities, including asthma, atopic dermatitis, and allergic rhinitis.5
Key Clinical Features
AA clinically manifests similarly across different skin tones; however, in patients with more tightly coiled or curly hair, the extent of scalp hair loss may be underestimated without a full examination. Culturally sensitive approaches to hair and scalp evaluation are essential, especially for Black women, whose hair care practices and scalp conditions may be overlooked or misunderstood during visits to evaluate hair loss. A thoughtful history and gentle examination of the hair and scalp that considers hair texture, cultural practices such as head coverings (eg, headwraps, turbans, hijabs), use of hair adornments (eg, clips, beads, bows), traditional braiding, and use of natural oils or herbal treatments, as well as styling methods including tight hairstyles, use of heat styling tools (eg, flat irons, curling irons), chemical application (eg, straighteners, hair color), and washing or styling frequency can improve diagnostic accuracy and help build trust in the patient-provider relationship.
Classic signs of AA visualized with dermoscopy include yellow and/or black dots on the scalp and exclamation point hairs. The appearance of fine white vellus hairs within the alopecic patches also may indicate early regrowth. On scalp trichoscopy, black dots are more prominent, and yellow dots are less prominent, in individuals with darker skin tones vs lighter skin tones.9
Worth Noting
In addition to a full examination of the scalp, documenting the extent of hair loss using validated severity scales, including the severity of alopecia tool (SALT), AA severity index (AASI), clinician-reported outcome assessment, and patient-reported outcome measures, can standardize disease severity assessment, facilitate timely insurance or medication approvals, and support objective tracking of treatment response, which may ultimately enhance access to care.10
Prompt treatment of AA is essential. Not surprisingly, patients given a diagnosis of AA may experience considerable emotional and psychological distress—regardless of the extent of the loss.11 Treatment options include mid- to high-potency topical or intralesional corticosteroids and newer and more targeted systemic options, including 3 Janus kinase (JAK) inhibitors—baricitinib, ritlecitinib, and deuruxolitinib—for more extensive disease.12 Treatment with intralesional corticosteroids may cause transient hypopigmentation, which may be more noticeable in patients with darker skin tones. Delays in treatment with JAK inhibitors can lead to a less-than-optimal response. Of the 3 JAK inhibitors that are approved by the US Food and Drug Administration for AA, only ritlecitinib is approved for children 12 years and older, leaving a therapeutic gap for younger patients that often leads to uncomfortable scalp injections, delayed or no treatment, off-label use of JAK inhibitors as well as the pairing of off-label dupilumab with oral minoxidil.12
Based on adult data, patients with severe disease and a shorter duration of hair loss (ie, < 4 years) tend to respond better to JAK inhibitors than those experiencing hair loss for longer periods. Also, those with more severe AA tend to have poorer outcomes than those with less severe disease.13 If treatment proves less than optimal, wigs and hair pieces may need to be considered. It is worth noting that some insurance companies will cover the cost of wigs for patients when prescribed as cranial prostheses.
Health Disparity Highlight
Health disparities in AA can be influenced by socioeconomic status and access to care. Patients from lower-income backgrounds often face barriers to accessing dermatologic care and treatments such as JAK inhibitors, which may remain inaccessible due to high costs and insurance limitations.14 These barriers can intersect with other factors such as age, sex, and race, potentially exacerbating disparities. Women with skin of color in underserved communities may experience delayed diagnosis, limited treatment options, and greater psychosocial distress from hair loss.14 Addressing these inequities requires advocacy, education for both patients and clinicians, and improved access to treatment to ensure comprehensive care for all patients.
- Kara T, Topkarcı Z. Interactions between posttraumatic stress disorder and alopecia areata in child with trauma exposure: two case reports. Int J Trichology. 2018;10:131-134. doi:10.4103/ijt.ijt_2_18
- Sy N, Mastacouris N, Strunk A, et al. Overall and racial and ethnic subgroup prevalences of alopecia areata, alopecia totalis, and alopecia universalis. JAMA Dermatol. 2023;159:419-423.
- Lee H, Jung SJ, Patel AB, et al. Racial characteristics of alopecia areata in the United States. J Am Acad Dermatol. 2020;83:1064-1070.
- Feaster B, McMichael AJ. Epidemiology of alopecia areata in Black patients: a retrospective chart review. J Am Acad Dermatol. 2022;87:1121-1123.
- Lee HH, Gwillim E, Patel KR, et al. Epidemiology of alopecia areata, ophiasis, totalis, and universalis: a systematic review and meta-analysis. J Am Acad Dermatol. 2020;82:675-682.
- Mostaghimi A, Gao W, Ray M, et al. Trends in prevalence and incidence of alopecia areata, alopecia totalis, and alopecia universalis among adults and children in a US employer-sponsored insured population. JAMA Dermatol. 2023;159:411-418.
- Adhanom R, Ansbro B, Castelo-Soccio L. Epidemiology of pediatric alopecia areata. Pediatr Dermatol. 2025;42 suppl 1(suppl 1):12-23.
- Karampinis E, Toli O, Georgopoulou KE, et al. Exploring pediatric dermatology in skin of color: focus on dermoscopy. Life (Basel). 2024;14:1604.
- King BA, Senna MM, Ohyama M, et al. Defining severity in alopecia areata: current perspectives and a multidimensional framework. Dermatol Ther (Heidelb). 2022;12:825-834.
- Toussi A, Barton VR, Le ST, et al. Psychosocial and psychiatric comorbidities and health-related quality of life in alopecia areata: a systematic review. J Am Acad Dermatol. 2021;85:162-175.
- Kalil L, Welch D, Heath CR, et al. Systemic therapies for pediatric alopecia areata. Pediatr Dermatol. 2025;42 suppl 1:36-42.
- King BA, Craiglow BG. Janus kinase inhibitors for alopecia areata. J Am Acad Dermatol. 2023;89:S29-S32.
- Klein EJ, Taiwò D, Kakpovbia E, et al. Disparities in Janus kinase inhibitor access for alopecia areata: a retrospective analysis. Int J Womens Dermatol. 2024;10:E155.
- McKenzie PL, Maltenfort M, Bruckner AL, et al. Evaluation of the prevalence and incidence of pediatric alopecia areata using electronic health record data. JAMA Dermatol. 2022;158:547-551. doi:10.1001/jamadermatol.2022.0351
The Comparison
A. Alopecia areata in a young girl with a lighter skin tone. The fine white vellus hairs are signs of regrowth.
B. Alopecia areata in a 49-year-old man with tightly coiled hair and darker skin tone. Coiled white hairs are noted in the alopecia patches.

young girl with a lighter skin
tone. The fine white vellus
hairs are signs of regrowth. Photographs courtesy of
Richard P. Usatine, MD.

49-year-old man with tightly
coiled hair and darker skin
tone. Coiled white hairs
are noted in the alopecia
patches. Photographs courtesy of
Richard P. Usatine, MD.
Alopecia areata (AA) is a common autoimmune condition characterized by hair loss resulting from a T cell–mediated attack on the hair follicles. It manifests as nonscarring patches of hair loss on the scalp, eyebrows, eyelashes, and beard area as well as more extensive complete loss of scalp and body hair. While AA may affect individuals of any age, most patients develop their first patch(es) of hair loss during childhood.1 The treatment landscape for AA has evolved considerably in recent years, but barriers to access to newer treatments persist.
Epidemiology
AA is most prevalent among pediatric and adult individuals of African, Asian, or Hispanic/Latino descent.2-4 In some studies, Black individuals had higher odds and Asian individuals had lower odds of developing AA, while other studies have reported the highest standardized prevalence among Asian individuals.5 In the United States, AA affects about 1.47% of adults and as many as 0.11% of children.6-8 In Black patients, AA often manifests early with a female predominance.5
AA frequently is associated with autoimmune comorbidities, the most common being thyroid disease.3,5 In Black patients, AA is associated with more atopic comorbidities, including asthma, atopic dermatitis, and allergic rhinitis.5
Key Clinical Features
AA clinically manifests similarly across different skin tones; however, in patients with more tightly coiled or curly hair, the extent of scalp hair loss may be underestimated without a full examination. Culturally sensitive approaches to hair and scalp evaluation are essential, especially for Black women, whose hair care practices and scalp conditions may be overlooked or misunderstood during visits to evaluate hair loss. A thoughtful history and gentle examination of the hair and scalp that considers hair texture, cultural practices such as head coverings (eg, headwraps, turbans, hijabs), use of hair adornments (eg, clips, beads, bows), traditional braiding, and use of natural oils or herbal treatments, as well as styling methods including tight hairstyles, use of heat styling tools (eg, flat irons, curling irons), chemical application (eg, straighteners, hair color), and washing or styling frequency can improve diagnostic accuracy and help build trust in the patient-provider relationship.
Classic signs of AA visualized with dermoscopy include yellow and/or black dots on the scalp and exclamation point hairs. The appearance of fine white vellus hairs within the alopecic patches also may indicate early regrowth. On scalp trichoscopy, black dots are more prominent, and yellow dots are less prominent, in individuals with darker skin tones vs lighter skin tones.9
Worth Noting
In addition to a full examination of the scalp, documenting the extent of hair loss using validated severity scales, including the severity of alopecia tool (SALT), AA severity index (AASI), clinician-reported outcome assessment, and patient-reported outcome measures, can standardize disease severity assessment, facilitate timely insurance or medication approvals, and support objective tracking of treatment response, which may ultimately enhance access to care.10
Prompt treatment of AA is essential. Not surprisingly, patients given a diagnosis of AA may experience considerable emotional and psychological distress—regardless of the extent of the loss.11 Treatment options include mid- to high-potency topical or intralesional corticosteroids and newer and more targeted systemic options, including 3 Janus kinase (JAK) inhibitors—baricitinib, ritlecitinib, and deuruxolitinib—for more extensive disease.12 Treatment with intralesional corticosteroids may cause transient hypopigmentation, which may be more noticeable in patients with darker skin tones. Delays in treatment with JAK inhibitors can lead to a less-than-optimal response. Of the 3 JAK inhibitors that are approved by the US Food and Drug Administration for AA, only ritlecitinib is approved for children 12 years and older, leaving a therapeutic gap for younger patients that often leads to uncomfortable scalp injections, delayed or no treatment, off-label use of JAK inhibitors as well as the pairing of off-label dupilumab with oral minoxidil.12
Based on adult data, patients with severe disease and a shorter duration of hair loss (ie, < 4 years) tend to respond better to JAK inhibitors than those experiencing hair loss for longer periods. Also, those with more severe AA tend to have poorer outcomes than those with less severe disease.13 If treatment proves less than optimal, wigs and hair pieces may need to be considered. It is worth noting that some insurance companies will cover the cost of wigs for patients when prescribed as cranial prostheses.
Health Disparity Highlight
Health disparities in AA can be influenced by socioeconomic status and access to care. Patients from lower-income backgrounds often face barriers to accessing dermatologic care and treatments such as JAK inhibitors, which may remain inaccessible due to high costs and insurance limitations.14 These barriers can intersect with other factors such as age, sex, and race, potentially exacerbating disparities. Women with skin of color in underserved communities may experience delayed diagnosis, limited treatment options, and greater psychosocial distress from hair loss.14 Addressing these inequities requires advocacy, education for both patients and clinicians, and improved access to treatment to ensure comprehensive care for all patients.
The Comparison
A. Alopecia areata in a young girl with a lighter skin tone. The fine white vellus hairs are signs of regrowth.
B. Alopecia areata in a 49-year-old man with tightly coiled hair and darker skin tone. Coiled white hairs are noted in the alopecia patches.

young girl with a lighter skin
tone. The fine white vellus
hairs are signs of regrowth. Photographs courtesy of
Richard P. Usatine, MD.

49-year-old man with tightly
coiled hair and darker skin
tone. Coiled white hairs
are noted in the alopecia
patches. Photographs courtesy of
Richard P. Usatine, MD.
Alopecia areata (AA) is a common autoimmune condition characterized by hair loss resulting from a T cell–mediated attack on the hair follicles. It manifests as nonscarring patches of hair loss on the scalp, eyebrows, eyelashes, and beard area as well as more extensive complete loss of scalp and body hair. While AA may affect individuals of any age, most patients develop their first patch(es) of hair loss during childhood.1 The treatment landscape for AA has evolved considerably in recent years, but barriers to access to newer treatments persist.
Epidemiology
AA is most prevalent among pediatric and adult individuals of African, Asian, or Hispanic/Latino descent.2-4 In some studies, Black individuals had higher odds and Asian individuals had lower odds of developing AA, while other studies have reported the highest standardized prevalence among Asian individuals.5 In the United States, AA affects about 1.47% of adults and as many as 0.11% of children.6-8 In Black patients, AA often manifests early with a female predominance.5
AA frequently is associated with autoimmune comorbidities, the most common being thyroid disease.3,5 In Black patients, AA is associated with more atopic comorbidities, including asthma, atopic dermatitis, and allergic rhinitis.5
Key Clinical Features
AA clinically manifests similarly across different skin tones; however, in patients with more tightly coiled or curly hair, the extent of scalp hair loss may be underestimated without a full examination. Culturally sensitive approaches to hair and scalp evaluation are essential, especially for Black women, whose hair care practices and scalp conditions may be overlooked or misunderstood during visits to evaluate hair loss. A thoughtful history and gentle examination of the hair and scalp that considers hair texture, cultural practices such as head coverings (eg, headwraps, turbans, hijabs), use of hair adornments (eg, clips, beads, bows), traditional braiding, and use of natural oils or herbal treatments, as well as styling methods including tight hairstyles, use of heat styling tools (eg, flat irons, curling irons), chemical application (eg, straighteners, hair color), and washing or styling frequency can improve diagnostic accuracy and help build trust in the patient-provider relationship.
Classic signs of AA visualized with dermoscopy include yellow and/or black dots on the scalp and exclamation point hairs. The appearance of fine white vellus hairs within the alopecic patches also may indicate early regrowth. On scalp trichoscopy, black dots are more prominent, and yellow dots are less prominent, in individuals with darker skin tones vs lighter skin tones.9
Worth Noting
In addition to a full examination of the scalp, documenting the extent of hair loss using validated severity scales, including the severity of alopecia tool (SALT), AA severity index (AASI), clinician-reported outcome assessment, and patient-reported outcome measures, can standardize disease severity assessment, facilitate timely insurance or medication approvals, and support objective tracking of treatment response, which may ultimately enhance access to care.10
Prompt treatment of AA is essential. Not surprisingly, patients given a diagnosis of AA may experience considerable emotional and psychological distress—regardless of the extent of the loss.11 Treatment options include mid- to high-potency topical or intralesional corticosteroids and newer and more targeted systemic options, including 3 Janus kinase (JAK) inhibitors—baricitinib, ritlecitinib, and deuruxolitinib—for more extensive disease.12 Treatment with intralesional corticosteroids may cause transient hypopigmentation, which may be more noticeable in patients with darker skin tones. Delays in treatment with JAK inhibitors can lead to a less-than-optimal response. Of the 3 JAK inhibitors that are approved by the US Food and Drug Administration for AA, only ritlecitinib is approved for children 12 years and older, leaving a therapeutic gap for younger patients that often leads to uncomfortable scalp injections, delayed or no treatment, off-label use of JAK inhibitors as well as the pairing of off-label dupilumab with oral minoxidil.12
Based on adult data, patients with severe disease and a shorter duration of hair loss (ie, < 4 years) tend to respond better to JAK inhibitors than those experiencing hair loss for longer periods. Also, those with more severe AA tend to have poorer outcomes than those with less severe disease.13 If treatment proves less than optimal, wigs and hair pieces may need to be considered. It is worth noting that some insurance companies will cover the cost of wigs for patients when prescribed as cranial prostheses.
Health Disparity Highlight
Health disparities in AA can be influenced by socioeconomic status and access to care. Patients from lower-income backgrounds often face barriers to accessing dermatologic care and treatments such as JAK inhibitors, which may remain inaccessible due to high costs and insurance limitations.14 These barriers can intersect with other factors such as age, sex, and race, potentially exacerbating disparities. Women with skin of color in underserved communities may experience delayed diagnosis, limited treatment options, and greater psychosocial distress from hair loss.14 Addressing these inequities requires advocacy, education for both patients and clinicians, and improved access to treatment to ensure comprehensive care for all patients.
- Kara T, Topkarcı Z. Interactions between posttraumatic stress disorder and alopecia areata in child with trauma exposure: two case reports. Int J Trichology. 2018;10:131-134. doi:10.4103/ijt.ijt_2_18
- Sy N, Mastacouris N, Strunk A, et al. Overall and racial and ethnic subgroup prevalences of alopecia areata, alopecia totalis, and alopecia universalis. JAMA Dermatol. 2023;159:419-423.
- Lee H, Jung SJ, Patel AB, et al. Racial characteristics of alopecia areata in the United States. J Am Acad Dermatol. 2020;83:1064-1070.
- Feaster B, McMichael AJ. Epidemiology of alopecia areata in Black patients: a retrospective chart review. J Am Acad Dermatol. 2022;87:1121-1123.
- Lee HH, Gwillim E, Patel KR, et al. Epidemiology of alopecia areata, ophiasis, totalis, and universalis: a systematic review and meta-analysis. J Am Acad Dermatol. 2020;82:675-682.
- Mostaghimi A, Gao W, Ray M, et al. Trends in prevalence and incidence of alopecia areata, alopecia totalis, and alopecia universalis among adults and children in a US employer-sponsored insured population. JAMA Dermatol. 2023;159:411-418.
- Adhanom R, Ansbro B, Castelo-Soccio L. Epidemiology of pediatric alopecia areata. Pediatr Dermatol. 2025;42 suppl 1(suppl 1):12-23.
- Karampinis E, Toli O, Georgopoulou KE, et al. Exploring pediatric dermatology in skin of color: focus on dermoscopy. Life (Basel). 2024;14:1604.
- King BA, Senna MM, Ohyama M, et al. Defining severity in alopecia areata: current perspectives and a multidimensional framework. Dermatol Ther (Heidelb). 2022;12:825-834.
- Toussi A, Barton VR, Le ST, et al. Psychosocial and psychiatric comorbidities and health-related quality of life in alopecia areata: a systematic review. J Am Acad Dermatol. 2021;85:162-175.
- Kalil L, Welch D, Heath CR, et al. Systemic therapies for pediatric alopecia areata. Pediatr Dermatol. 2025;42 suppl 1:36-42.
- King BA, Craiglow BG. Janus kinase inhibitors for alopecia areata. J Am Acad Dermatol. 2023;89:S29-S32.
- Klein EJ, Taiwò D, Kakpovbia E, et al. Disparities in Janus kinase inhibitor access for alopecia areata: a retrospective analysis. Int J Womens Dermatol. 2024;10:E155.
- McKenzie PL, Maltenfort M, Bruckner AL, et al. Evaluation of the prevalence and incidence of pediatric alopecia areata using electronic health record data. JAMA Dermatol. 2022;158:547-551. doi:10.1001/jamadermatol.2022.0351
- Kara T, Topkarcı Z. Interactions between posttraumatic stress disorder and alopecia areata in child with trauma exposure: two case reports. Int J Trichology. 2018;10:131-134. doi:10.4103/ijt.ijt_2_18
- Sy N, Mastacouris N, Strunk A, et al. Overall and racial and ethnic subgroup prevalences of alopecia areata, alopecia totalis, and alopecia universalis. JAMA Dermatol. 2023;159:419-423.
- Lee H, Jung SJ, Patel AB, et al. Racial characteristics of alopecia areata in the United States. J Am Acad Dermatol. 2020;83:1064-1070.
- Feaster B, McMichael AJ. Epidemiology of alopecia areata in Black patients: a retrospective chart review. J Am Acad Dermatol. 2022;87:1121-1123.
- Lee HH, Gwillim E, Patel KR, et al. Epidemiology of alopecia areata, ophiasis, totalis, and universalis: a systematic review and meta-analysis. J Am Acad Dermatol. 2020;82:675-682.
- Mostaghimi A, Gao W, Ray M, et al. Trends in prevalence and incidence of alopecia areata, alopecia totalis, and alopecia universalis among adults and children in a US employer-sponsored insured population. JAMA Dermatol. 2023;159:411-418.
- Adhanom R, Ansbro B, Castelo-Soccio L. Epidemiology of pediatric alopecia areata. Pediatr Dermatol. 2025;42 suppl 1(suppl 1):12-23.
- Karampinis E, Toli O, Georgopoulou KE, et al. Exploring pediatric dermatology in skin of color: focus on dermoscopy. Life (Basel). 2024;14:1604.
- King BA, Senna MM, Ohyama M, et al. Defining severity in alopecia areata: current perspectives and a multidimensional framework. Dermatol Ther (Heidelb). 2022;12:825-834.
- Toussi A, Barton VR, Le ST, et al. Psychosocial and psychiatric comorbidities and health-related quality of life in alopecia areata: a systematic review. J Am Acad Dermatol. 2021;85:162-175.
- Kalil L, Welch D, Heath CR, et al. Systemic therapies for pediatric alopecia areata. Pediatr Dermatol. 2025;42 suppl 1:36-42.
- King BA, Craiglow BG. Janus kinase inhibitors for alopecia areata. J Am Acad Dermatol. 2023;89:S29-S32.
- Klein EJ, Taiwò D, Kakpovbia E, et al. Disparities in Janus kinase inhibitor access for alopecia areata: a retrospective analysis. Int J Womens Dermatol. 2024;10:E155.
- McKenzie PL, Maltenfort M, Bruckner AL, et al. Evaluation of the prevalence and incidence of pediatric alopecia areata using electronic health record data. JAMA Dermatol. 2022;158:547-551. doi:10.1001/jamadermatol.2022.0351
Consider Cultural Practices and Barriers to Care When Treating Alopecia Areata
Consider Cultural Practices and Barriers to Care When Treating Alopecia Areata