User login
Biomarkers Pinpoint Invasive Aspergillosis in Lung Transplant Recipients
Biomarkers Pinpoint Invasive Aspergillosis in Lung Transplant Recipients
Several newly identified biomarkers can help distinguish invasive aspergillosis from aspergillus colonization in lung transplant recipients, according to data from a new study presented at the annual meeting of the International Society for Heart and Lung Transplantation.
Aspergillus, a common environmental mold, can cause potentially serious infection or asymptomatic colonization in patients who have significant lung disease or are immunosuppressed, said Aaron Mishkin, MD, associate professor of medicine at the Lewis Katz School of Medicine at Temple University, Philadelphia, who was not involved in the study.
“Determining if the aspergillus that is present is a colonizing organism vs disease is challenging clinically,” Mishkin said. Clinicians currently rely on criteria including a compatible patient, imaging findings, and a laboratory-based diagnostic such as tissue from a biopsy, cultures, polymerase chain reaction (PCR), or fungal antigen detection, said Mishkin. “Fungal antigen detection has variable specificity and sensitivity,” he noted. New biomarkers that look for an immune response could help differentiate between colonization and infection by assessing an immune-mediated inflammatory response, the hallmark of infection, he said.
To tease out potential biomarkers associated with invasive aspergillosis, Christine Ng, MS, a researcher at the University Health Network, Toronto, Ontario, Canada, and colleagues performed RNA sequencing on samples from 14 control lung transplant patients, 34 with aspergillus colonization, and seven with invasive aspergillosis. They identified potential candidate genes in 15 control samples, 17 aspergillus colonization samples, and 15 invasive aspergillosis samples.
Overall, signaling pathway analysis showed robust immune response, T-cell immunity, and leukocyte immunity in patients with invasive aspergillosis. By contrast, patients with aspergillus colonization showed enriched cellular responses (response to stimuli, epithelium development).
In a real-time quantitative PCR analysis, the researchers validated three biomarkers specific to invasive aspergillosis (IRF7, ZBP1, CYP27B1). Biomarkers AKR1C2, FGF10, and VGLL3 demonstrated specificity for aspergillus colonization. Additionally, biomarkers PTGER3, LPAR3, and COL14A1 were significant when aspergillus colonization was compared to controls but not in comparisons between invasive aspergillosis and aspergillus colonization.
The study findings were limited by the small sample size, and larger studies are needed before they can be implemented in clinical practice, the researchers wrote. However, the results suggest that the new biomarkers reveal distinct host immune patterns and may improve differentiation of aspergillosis from colonization in lung transplant recipients, they concluded.
Clinical Implications and Next Steps
RNA testing can help differentiate colonization vs infection, Mishkin said. “Colonization is not typically treated, whereas infection would be treated with an anti-fungal and, in the case of a transplant recipient, a reduction in immunosuppression,” he said. “In lung transplantation, a delicate equilibrium must be maintained between achieving optimal immunosuppression and minimizing or treating infection. Any tools that can aid in this decision-making have the potential to enhance patient outcomes,” he added.
The current study was limited by the use of data only from a single center, and the broader applicability to additional populations, broader geographic areas, and a larger number of organisms remains unknown, Mishkin said. “This type of assay does have the possibility of applicability to a larger number of fungal and even bacterial species,” he noted.
A version of this article first appeared on Medscape.com.
Several newly identified biomarkers can help distinguish invasive aspergillosis from aspergillus colonization in lung transplant recipients, according to data from a new study presented at the annual meeting of the International Society for Heart and Lung Transplantation.
Aspergillus, a common environmental mold, can cause potentially serious infection or asymptomatic colonization in patients who have significant lung disease or are immunosuppressed, said Aaron Mishkin, MD, associate professor of medicine at the Lewis Katz School of Medicine at Temple University, Philadelphia, who was not involved in the study.
“Determining if the aspergillus that is present is a colonizing organism vs disease is challenging clinically,” Mishkin said. Clinicians currently rely on criteria including a compatible patient, imaging findings, and a laboratory-based diagnostic such as tissue from a biopsy, cultures, polymerase chain reaction (PCR), or fungal antigen detection, said Mishkin. “Fungal antigen detection has variable specificity and sensitivity,” he noted. New biomarkers that look for an immune response could help differentiate between colonization and infection by assessing an immune-mediated inflammatory response, the hallmark of infection, he said.
To tease out potential biomarkers associated with invasive aspergillosis, Christine Ng, MS, a researcher at the University Health Network, Toronto, Ontario, Canada, and colleagues performed RNA sequencing on samples from 14 control lung transplant patients, 34 with aspergillus colonization, and seven with invasive aspergillosis. They identified potential candidate genes in 15 control samples, 17 aspergillus colonization samples, and 15 invasive aspergillosis samples.
Overall, signaling pathway analysis showed robust immune response, T-cell immunity, and leukocyte immunity in patients with invasive aspergillosis. By contrast, patients with aspergillus colonization showed enriched cellular responses (response to stimuli, epithelium development).
In a real-time quantitative PCR analysis, the researchers validated three biomarkers specific to invasive aspergillosis (IRF7, ZBP1, CYP27B1). Biomarkers AKR1C2, FGF10, and VGLL3 demonstrated specificity for aspergillus colonization. Additionally, biomarkers PTGER3, LPAR3, and COL14A1 were significant when aspergillus colonization was compared to controls but not in comparisons between invasive aspergillosis and aspergillus colonization.
The study findings were limited by the small sample size, and larger studies are needed before they can be implemented in clinical practice, the researchers wrote. However, the results suggest that the new biomarkers reveal distinct host immune patterns and may improve differentiation of aspergillosis from colonization in lung transplant recipients, they concluded.
Clinical Implications and Next Steps
RNA testing can help differentiate colonization vs infection, Mishkin said. “Colonization is not typically treated, whereas infection would be treated with an anti-fungal and, in the case of a transplant recipient, a reduction in immunosuppression,” he said. “In lung transplantation, a delicate equilibrium must be maintained between achieving optimal immunosuppression and minimizing or treating infection. Any tools that can aid in this decision-making have the potential to enhance patient outcomes,” he added.
The current study was limited by the use of data only from a single center, and the broader applicability to additional populations, broader geographic areas, and a larger number of organisms remains unknown, Mishkin said. “This type of assay does have the possibility of applicability to a larger number of fungal and even bacterial species,” he noted.
A version of this article first appeared on Medscape.com.
Several newly identified biomarkers can help distinguish invasive aspergillosis from aspergillus colonization in lung transplant recipients, according to data from a new study presented at the annual meeting of the International Society for Heart and Lung Transplantation.
Aspergillus, a common environmental mold, can cause potentially serious infection or asymptomatic colonization in patients who have significant lung disease or are immunosuppressed, said Aaron Mishkin, MD, associate professor of medicine at the Lewis Katz School of Medicine at Temple University, Philadelphia, who was not involved in the study.
“Determining if the aspergillus that is present is a colonizing organism vs disease is challenging clinically,” Mishkin said. Clinicians currently rely on criteria including a compatible patient, imaging findings, and a laboratory-based diagnostic such as tissue from a biopsy, cultures, polymerase chain reaction (PCR), or fungal antigen detection, said Mishkin. “Fungal antigen detection has variable specificity and sensitivity,” he noted. New biomarkers that look for an immune response could help differentiate between colonization and infection by assessing an immune-mediated inflammatory response, the hallmark of infection, he said.
To tease out potential biomarkers associated with invasive aspergillosis, Christine Ng, MS, a researcher at the University Health Network, Toronto, Ontario, Canada, and colleagues performed RNA sequencing on samples from 14 control lung transplant patients, 34 with aspergillus colonization, and seven with invasive aspergillosis. They identified potential candidate genes in 15 control samples, 17 aspergillus colonization samples, and 15 invasive aspergillosis samples.
Overall, signaling pathway analysis showed robust immune response, T-cell immunity, and leukocyte immunity in patients with invasive aspergillosis. By contrast, patients with aspergillus colonization showed enriched cellular responses (response to stimuli, epithelium development).
In a real-time quantitative PCR analysis, the researchers validated three biomarkers specific to invasive aspergillosis (IRF7, ZBP1, CYP27B1). Biomarkers AKR1C2, FGF10, and VGLL3 demonstrated specificity for aspergillus colonization. Additionally, biomarkers PTGER3, LPAR3, and COL14A1 were significant when aspergillus colonization was compared to controls but not in comparisons between invasive aspergillosis and aspergillus colonization.
The study findings were limited by the small sample size, and larger studies are needed before they can be implemented in clinical practice, the researchers wrote. However, the results suggest that the new biomarkers reveal distinct host immune patterns and may improve differentiation of aspergillosis from colonization in lung transplant recipients, they concluded.
Clinical Implications and Next Steps
RNA testing can help differentiate colonization vs infection, Mishkin said. “Colonization is not typically treated, whereas infection would be treated with an anti-fungal and, in the case of a transplant recipient, a reduction in immunosuppression,” he said. “In lung transplantation, a delicate equilibrium must be maintained between achieving optimal immunosuppression and minimizing or treating infection. Any tools that can aid in this decision-making have the potential to enhance patient outcomes,” he added.
The current study was limited by the use of data only from a single center, and the broader applicability to additional populations, broader geographic areas, and a larger number of organisms remains unknown, Mishkin said. “This type of assay does have the possibility of applicability to a larger number of fungal and even bacterial species,” he noted.
A version of this article first appeared on Medscape.com.
Biomarkers Pinpoint Invasive Aspergillosis in Lung Transplant Recipients
Biomarkers Pinpoint Invasive Aspergillosis in Lung Transplant Recipients
Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility
Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility
Self-reported penicillin allergies are common, with a prevalence of about 10% of patients, according to the Centers for Disease Control and Prevention (CDC).1 However, only about 1% of patients have a true immunoglobulin E (IgE)-mediated allergy. This issue is often further complicated by inaccurate classification of nonallergic adverse effects as an allergy, resulting in incomplete allergy documentation in the electronic health record (EHR). The cross-reactivity rate with cephalosporins (Β-lactam antibiotics) in patients reporting a penicillin allergy is < 1%, which suggests that many patients with reported penicillin allergies can safely receive them.2 Despite this, patients with self-reported penicillin allergies often receive non–Β-lactam antibiotic agents, which may be associated with an increased risk of adverse drug reactions (ADRs), increased health care costs, and inferior clinical outcomes.3
Several strategies are recommended to assess patients with self-reported penicillin allergies. According to the CDC, evaluating a patient who reports a penicillin or other Β-lactam antibiotic allergy involves 3 steps: (1) obtaining a thorough medical history, including previous exposures to penicillin or other Β-lactam antibiotic; (2) performing a skin test using the penicillin major and minor determinants; and (3) among those who have a negative penicillin skin test, performing an observed oral challenge with 250 mg amoxicillin before proceeding directly to treatment with the indicated Β-lactam therapy.4
Most existing clinical guidance for assessing patients with self-reported penicillin allergies stems from site-specific policies and primarily focuses on oral amoxicillin challenges or penicillin skin testing (PST). However, performing these tests may not be feasible at all facilities due to time constraints and lack of allergists. Therefore, alternative strategies are necessary, such as conducting detailed patient interviews. Few studies have evaluated switching to Β-lactam agents following a penicillin allergy interview alone. However, with thorough patient histories and detailed interviews, patients with reported penicillin allergies can safely use Β-lactam antibiotics.5 Implementing this procedure provides a cost-savings opportunity by not having to administer additional antibiotics for testing in addition to improving antibiotic stewardship.
The Memphis Veterans Affairs Medical Center (MVAMC) created the Allergy to Β-Lactam Evaluation (ABLE) process to clarify and remove penicillin allergies. The process involves conducting a thorough chart review and patient interview followed by completion of a note template that provides recommendations about patient allergies and Β-lactam prescribing. Mitchell et al found that the pharmacist-led process to be beneficial for addressing Β-lactam allergy clearance.6 As a result, the ABLE process was implemented at several other US Department of Veterans Affairs (VA) medical centers (VAMCs). Using the ABLE template, the purpose of this study was to evaluate the impact of a pharmacist-led penicillin allergy initiative on penicillin allergy delabeling with an interview process alone.
Methods
Prior to ABLE process implementation, there were no standardized procedures for documenting allergy histories. ABLE was implemented at the Robley Rex VAMC (RRVAMC) in November 2022. During the interview phase, patients were initially identified during admission via TheraDoc as having either a penicillin allergy or ADR. The infectious disease pharmacist or pharmacy resident interviewed patients with documented penicillin allergies or ADRs using a standardized questionnaire (eAppendix 1). Not all identified patients could be interviewed. Patients currently receiving an antibiotic were prioritized for interviews. Patients were excluded if they declined or were unable to be interviewed, although a patient’s caregiver(s) could be interviewed in person or via telephone, if the patient was not available.
Following the interview, pharmacists used guidance from the ABLE process in addition to a detailed EHR review to determine whether the patient was eligible for an allergy update or removal and/or switch to a Β-lactam antibiotic (Figure). If eligible for modification, the interviewing pharmacist made the necessary changes. A templated process note with patient-specific recommendations was entered into the Computerized Patient Record System (CPRS) and the primary care team attending physician was added as an additional signer to be alerted in the system note (eAppendix 2).
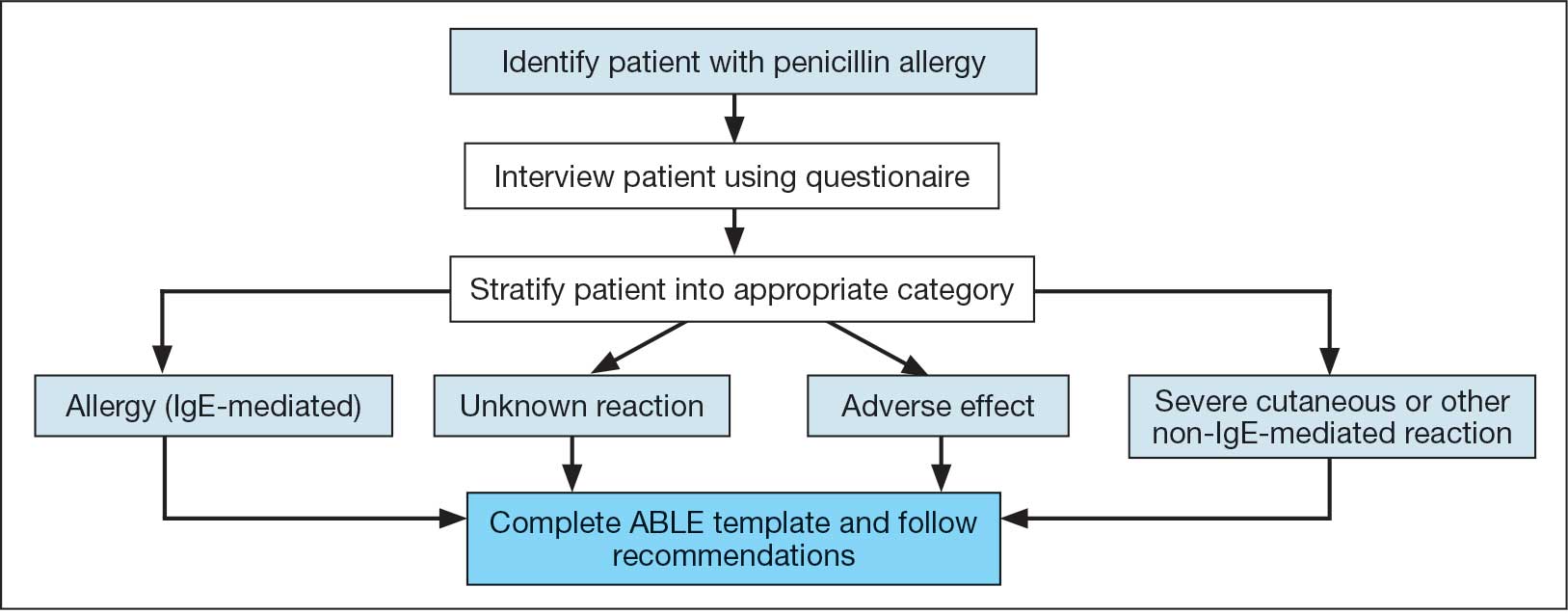
This single-center, retrospective cohort study involved review of CPRS notes and clinical interviews in the interviewed group. Hospitalized patients at the RRVAMC aged ≥ 18 years with a documented penicillin allergy or ADR were included. The historical control group consisted of patients admitted between October 31, 2019, and October 31, 2022, and the intervention group consisted of patients admitted between November 1, 2022, and March 1, 2023. Patients in the historical control group were matched 1:1 to the intervention group for penicillin allergy severity (allergy [IgE-mediated], unknown, adverse effect, severe cutaneous or other non–IgE-mediated reaction) and whether they received a noncarbapenem non–Β-lactam antibiotic.
The primary outcome was the number of patient allergies/ADRs removed or changed on patient profiles regardless of whether their antibiotic regimen was changed. This outcome was further assessed by evaluating the number of patient allergies or ADRs removed or changed on patient profiles with or without a change in antibiotic regimen. Primary outcomes were analyzed using χ2 and/ or Fisher exact tests, as appropriate to determine statistically significant differences between the interviewed group and the historical control.
Results
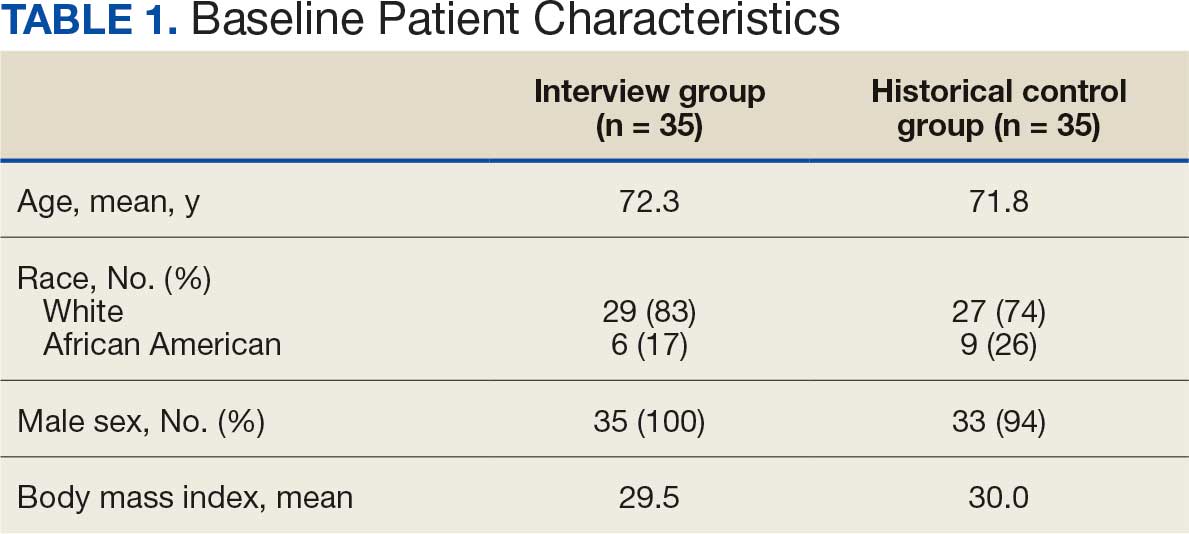
Seventy patients were included: 35 patients in the interviewed group and 35 patients in the historical control group, respectively. Both groups had a mean age of 72 years and predominantly included White male patients (Table 1). Following the interview, the allergy profile was modified for 6 patients (17%) in the interview group vs 0 patients in the control group (P = .03) (Table 2). The primary outcome was analyzed separately regardless of an antibiotic regimen change. There was not a statistically significant difference between groups when assessing patients for change in therapy (P > .99). All 6 patients with an allergy profile modification had no change in antibiotic regimen.

Discussion
This study suggests the ABLE process may be a valuable tool for adjusting penicillin allergies or ADRs within patient EHRs. In the interview group, allergies were modified in 6 (17%) patients while no patients in the control group had allergy modifications. Of the 6 allergy profile modifications, 4 allergy labels were changed from an allergy to an ADR. These patients were cleared to receive future Β-lactam antibiotics after clinicians recognized the lack of a true IgE-mediated allergic reaction. In addition, 2 of the modified allergy profiles removed the allergy designation. Although this represents a small subset of interviewed patients, it illustrates the clinical effectiveness of an interview process alone to remove penicillin allergy designations.
Previous research has assessed the impact of pharmacist intervention on penicillin allergy clarification. Mitchell et al implemented a pharmacist-driven Β-lactam allergy assessment and penicillin allergy clinic (PAC) at the MVAMC with the goal of evaluating its impact on allergy clearance. In their study, clinical pharmacy specialists evaluated patients with Β-lactam allergies, and those deemed eligible were later seen in the PAC. Among the 246 patients evaluated using the Β-lactam allergy assessment alone and who were not seen in the PAC, 25% had their penicillin allergy removed following a detailed assessment.6
Song et al evaluated the effectiveness and feasibility of a pharmacist-driven penicillin allergy delabeling pilot program without skin testing or oral challenges. Patients with penicillin allergies were interviewed by a pharmacy resident using a standardized checklist. Among the 66 patients interviewed, 12 (18%) met the criteria for delabeling and consented to removal of their allergy.7 The delabeling rates in these 2 studies are similar to the 17% rate of allergy modification in our study, although this study is the only one to compare results to a historical control group.
Harper et al evaluated the impact of a penicillin allergy assessment, including penicillin skin testing and oral amoxicillin challenges, on delabeling penicillin allergies. Pharmacists completed a penicillin allergy assessment and performed penicillin skin testing and/or oral amoxicillin challenges for eligible patients. Of 35 patients, 31 (89%) had their penicillin allergies delabeled in the EHR.8 The rate of penicillin allergy delabeling in Harper et al was likely higher than that seen in our study due to the use of oral challenge and skin testing. Regardless, a detailed penicillin allergy interview alone was effective at RRVAMC, resulting in a significant rate of allergy removal or change. This supports the use of detailed penicillin allergy assessments in settings where penicillin skin testing or oral challenges may not be feasible.
Mann et al demonstrated the effectiveness of penicillin allergy assessments in switching eligible patients to Β-lactam antibiotics. Their single-center, prospective study assessed the impact of a pharmacist-driven detailed penicillin allergy interview initiative. Interviews that evaluated potential changes to allergy profiles were conducted with 175 patients. Of these patients, 135 (77.1%) were on antimicrobial therapy and 42 (31.1%) patients receiving therapy met criteria to switch to a noncarbapenem Β-lactam antibiotic. Thirty-one patients (73.8%) switched with no signs or symptoms of intolerance demonstrating that an interview can be a valuable tool for antibiotic optimization, specifically in patients with penicillin allergy.9 No patients in our study switched antibiotic therapy, likely because only a small number of patients were eligible for transition to a noncarbapenem Β-lactam antibiotic. In the Mann et al study, non–Β-lactam antibiotics, such as fluoroquinolones and carbapenems, accounted for > 75% of the antibiotics used.
Limitations
The sample size of this study was small and its duration was short. There is a risk for selection bias as not all identified patients were able to be interviewed while admitted, but patients on antibiotics were prioritized as they were most likely to directly benefit during their current admission from a modification of their allergy. Most patients in the study were White and male, which may limit the generalizability of the results. Additionally, recommendations regarding antibiotic changes were primarily communicated to the treatment team based on a templated note in CPRS alone. Therefore, implementation of these recommendations largely relied upon nonverbal communication. Direct pharmacist-physician communication could have led to a larger impact on antimicrobial therapy changes. The interviewer’s participation in daily rounds with time allotted to discuss this topic can be considered in the future to improve these processes.
Conclusions
This study found that the ABLE process identified patients for penicillin allergy delabeling. With the high prevalence of inaccurate penicillin allergy documentation, this tool offers VA health care systems a way to empower pharmacists in allergy clarification, leading to improvements in antibiotic stewardship. Although the sample size was small, the ABLE process may provide a framework for VA clinicians. Future research has the potential to demonstrate the practicality and effectiveness this pharmacist-led penicillin allergy interview process can offer clinicians.
- Health care providers. Clinical features of penicillin allergy. Centers for Disease Control and Prevention. August 25, 2025. Accessed February 4, 2026. https://www.cdc.gov /antibiotic-use/hcp/clinical-signs/index.html
- Wrynn AF. Penicillin allergies: A guide for NPs. Nurse Pract. 2022;47:30-36. doi:10.1097/01.NPR.0000855312.11145.78
- Mohsen S, Dickinson JA, Somayaji R. Update on the adverse effects of antimicrobial therapies in community practice. Can Fam Physician. 2020;66:651-659.
- Sexually Transmitted Infections Treatment Guidelines, 2021. Managing persons who have a history of penicillin allergy. Centers for Disease Control and Prevention. September 21, 2022. Accessed February 4, 2026. https:// www.cdc.gov/std/treatment-guidelines/penicillin-allergy .htm
- Holmes AK, Bennett NT, Berry TP. Pharmacy driven assessment of appropriate antibiotic selection in patients with reported beta-lactam allergy. J Am Coll Clin Pharm. 2019;2:509-514. doi:10.1002/jac5.1135
- Mitchell AB, Ness RA, Bennett JG, et al. Implementation and impact of a Β-lactam allergy assessment protocol in a veteran population. Fed Pract. 2021;38:420-425. doi:10.12788/fp.0172
- Song YC, Nelson ZJ, Wankum MA, et al. Effectiveness and feasibility of pharmacist-driven penicillin allergy de-labeling pilot program without skin testing or oral challenges. Pharmacy (Basel). 2021;9:127. doi:10.3390/pharmacy9030127
- Harper HM, Sanchez M. Review of pharmacist driven penicillin allergy assessments and skin testing: a multicenter case-series. Hosp Pharm. 2022;57:469-473. doi:10.1177/00185787211046862
- Mann KL, Wu JY, Shah SS. Implementation of a pharmacist- driven detailed penicillin allergy interview. Ann Pharmacother. 2020;54:364-370. doi:10.1177/1060028019884874
Self-reported penicillin allergies are common, with a prevalence of about 10% of patients, according to the Centers for Disease Control and Prevention (CDC).1 However, only about 1% of patients have a true immunoglobulin E (IgE)-mediated allergy. This issue is often further complicated by inaccurate classification of nonallergic adverse effects as an allergy, resulting in incomplete allergy documentation in the electronic health record (EHR). The cross-reactivity rate with cephalosporins (Β-lactam antibiotics) in patients reporting a penicillin allergy is < 1%, which suggests that many patients with reported penicillin allergies can safely receive them.2 Despite this, patients with self-reported penicillin allergies often receive non–Β-lactam antibiotic agents, which may be associated with an increased risk of adverse drug reactions (ADRs), increased health care costs, and inferior clinical outcomes.3
Several strategies are recommended to assess patients with self-reported penicillin allergies. According to the CDC, evaluating a patient who reports a penicillin or other Β-lactam antibiotic allergy involves 3 steps: (1) obtaining a thorough medical history, including previous exposures to penicillin or other Β-lactam antibiotic; (2) performing a skin test using the penicillin major and minor determinants; and (3) among those who have a negative penicillin skin test, performing an observed oral challenge with 250 mg amoxicillin before proceeding directly to treatment with the indicated Β-lactam therapy.4
Most existing clinical guidance for assessing patients with self-reported penicillin allergies stems from site-specific policies and primarily focuses on oral amoxicillin challenges or penicillin skin testing (PST). However, performing these tests may not be feasible at all facilities due to time constraints and lack of allergists. Therefore, alternative strategies are necessary, such as conducting detailed patient interviews. Few studies have evaluated switching to Β-lactam agents following a penicillin allergy interview alone. However, with thorough patient histories and detailed interviews, patients with reported penicillin allergies can safely use Β-lactam antibiotics.5 Implementing this procedure provides a cost-savings opportunity by not having to administer additional antibiotics for testing in addition to improving antibiotic stewardship.
The Memphis Veterans Affairs Medical Center (MVAMC) created the Allergy to Β-Lactam Evaluation (ABLE) process to clarify and remove penicillin allergies. The process involves conducting a thorough chart review and patient interview followed by completion of a note template that provides recommendations about patient allergies and Β-lactam prescribing. Mitchell et al found that the pharmacist-led process to be beneficial for addressing Β-lactam allergy clearance.6 As a result, the ABLE process was implemented at several other US Department of Veterans Affairs (VA) medical centers (VAMCs). Using the ABLE template, the purpose of this study was to evaluate the impact of a pharmacist-led penicillin allergy initiative on penicillin allergy delabeling with an interview process alone.
Methods
Prior to ABLE process implementation, there were no standardized procedures for documenting allergy histories. ABLE was implemented at the Robley Rex VAMC (RRVAMC) in November 2022. During the interview phase, patients were initially identified during admission via TheraDoc as having either a penicillin allergy or ADR. The infectious disease pharmacist or pharmacy resident interviewed patients with documented penicillin allergies or ADRs using a standardized questionnaire (eAppendix 1). Not all identified patients could be interviewed. Patients currently receiving an antibiotic were prioritized for interviews. Patients were excluded if they declined or were unable to be interviewed, although a patient’s caregiver(s) could be interviewed in person or via telephone, if the patient was not available.
Following the interview, pharmacists used guidance from the ABLE process in addition to a detailed EHR review to determine whether the patient was eligible for an allergy update or removal and/or switch to a Β-lactam antibiotic (Figure). If eligible for modification, the interviewing pharmacist made the necessary changes. A templated process note with patient-specific recommendations was entered into the Computerized Patient Record System (CPRS) and the primary care team attending physician was added as an additional signer to be alerted in the system note (eAppendix 2).

This single-center, retrospective cohort study involved review of CPRS notes and clinical interviews in the interviewed group. Hospitalized patients at the RRVAMC aged ≥ 18 years with a documented penicillin allergy or ADR were included. The historical control group consisted of patients admitted between October 31, 2019, and October 31, 2022, and the intervention group consisted of patients admitted between November 1, 2022, and March 1, 2023. Patients in the historical control group were matched 1:1 to the intervention group for penicillin allergy severity (allergy [IgE-mediated], unknown, adverse effect, severe cutaneous or other non–IgE-mediated reaction) and whether they received a noncarbapenem non–Β-lactam antibiotic.
The primary outcome was the number of patient allergies/ADRs removed or changed on patient profiles regardless of whether their antibiotic regimen was changed. This outcome was further assessed by evaluating the number of patient allergies or ADRs removed or changed on patient profiles with or without a change in antibiotic regimen. Primary outcomes were analyzed using χ2 and/ or Fisher exact tests, as appropriate to determine statistically significant differences between the interviewed group and the historical control.
Results
Seventy patients were included: 35 patients in the interviewed group and 35 patients in the historical control group, respectively. Both groups had a mean age of 72 years and predominantly included White male patients (Table 1). Following the interview, the allergy profile was modified for 6 patients (17%) in the interview group vs 0 patients in the control group (P = .03) (Table 2). The primary outcome was analyzed separately regardless of an antibiotic regimen change. There was not a statistically significant difference between groups when assessing patients for change in therapy (P > .99). All 6 patients with an allergy profile modification had no change in antibiotic regimen.


Discussion
This study suggests the ABLE process may be a valuable tool for adjusting penicillin allergies or ADRs within patient EHRs. In the interview group, allergies were modified in 6 (17%) patients while no patients in the control group had allergy modifications. Of the 6 allergy profile modifications, 4 allergy labels were changed from an allergy to an ADR. These patients were cleared to receive future Β-lactam antibiotics after clinicians recognized the lack of a true IgE-mediated allergic reaction. In addition, 2 of the modified allergy profiles removed the allergy designation. Although this represents a small subset of interviewed patients, it illustrates the clinical effectiveness of an interview process alone to remove penicillin allergy designations.
Previous research has assessed the impact of pharmacist intervention on penicillin allergy clarification. Mitchell et al implemented a pharmacist-driven Β-lactam allergy assessment and penicillin allergy clinic (PAC) at the MVAMC with the goal of evaluating its impact on allergy clearance. In their study, clinical pharmacy specialists evaluated patients with Β-lactam allergies, and those deemed eligible were later seen in the PAC. Among the 246 patients evaluated using the Β-lactam allergy assessment alone and who were not seen in the PAC, 25% had their penicillin allergy removed following a detailed assessment.6
Song et al evaluated the effectiveness and feasibility of a pharmacist-driven penicillin allergy delabeling pilot program without skin testing or oral challenges. Patients with penicillin allergies were interviewed by a pharmacy resident using a standardized checklist. Among the 66 patients interviewed, 12 (18%) met the criteria for delabeling and consented to removal of their allergy.7 The delabeling rates in these 2 studies are similar to the 17% rate of allergy modification in our study, although this study is the only one to compare results to a historical control group.
Harper et al evaluated the impact of a penicillin allergy assessment, including penicillin skin testing and oral amoxicillin challenges, on delabeling penicillin allergies. Pharmacists completed a penicillin allergy assessment and performed penicillin skin testing and/or oral amoxicillin challenges for eligible patients. Of 35 patients, 31 (89%) had their penicillin allergies delabeled in the EHR.8 The rate of penicillin allergy delabeling in Harper et al was likely higher than that seen in our study due to the use of oral challenge and skin testing. Regardless, a detailed penicillin allergy interview alone was effective at RRVAMC, resulting in a significant rate of allergy removal or change. This supports the use of detailed penicillin allergy assessments in settings where penicillin skin testing or oral challenges may not be feasible.
Mann et al demonstrated the effectiveness of penicillin allergy assessments in switching eligible patients to Β-lactam antibiotics. Their single-center, prospective study assessed the impact of a pharmacist-driven detailed penicillin allergy interview initiative. Interviews that evaluated potential changes to allergy profiles were conducted with 175 patients. Of these patients, 135 (77.1%) were on antimicrobial therapy and 42 (31.1%) patients receiving therapy met criteria to switch to a noncarbapenem Β-lactam antibiotic. Thirty-one patients (73.8%) switched with no signs or symptoms of intolerance demonstrating that an interview can be a valuable tool for antibiotic optimization, specifically in patients with penicillin allergy.9 No patients in our study switched antibiotic therapy, likely because only a small number of patients were eligible for transition to a noncarbapenem Β-lactam antibiotic. In the Mann et al study, non–Β-lactam antibiotics, such as fluoroquinolones and carbapenems, accounted for > 75% of the antibiotics used.
Limitations
The sample size of this study was small and its duration was short. There is a risk for selection bias as not all identified patients were able to be interviewed while admitted, but patients on antibiotics were prioritized as they were most likely to directly benefit during their current admission from a modification of their allergy. Most patients in the study were White and male, which may limit the generalizability of the results. Additionally, recommendations regarding antibiotic changes were primarily communicated to the treatment team based on a templated note in CPRS alone. Therefore, implementation of these recommendations largely relied upon nonverbal communication. Direct pharmacist-physician communication could have led to a larger impact on antimicrobial therapy changes. The interviewer’s participation in daily rounds with time allotted to discuss this topic can be considered in the future to improve these processes.
Conclusions
This study found that the ABLE process identified patients for penicillin allergy delabeling. With the high prevalence of inaccurate penicillin allergy documentation, this tool offers VA health care systems a way to empower pharmacists in allergy clarification, leading to improvements in antibiotic stewardship. Although the sample size was small, the ABLE process may provide a framework for VA clinicians. Future research has the potential to demonstrate the practicality and effectiveness this pharmacist-led penicillin allergy interview process can offer clinicians.
Self-reported penicillin allergies are common, with a prevalence of about 10% of patients, according to the Centers for Disease Control and Prevention (CDC).1 However, only about 1% of patients have a true immunoglobulin E (IgE)-mediated allergy. This issue is often further complicated by inaccurate classification of nonallergic adverse effects as an allergy, resulting in incomplete allergy documentation in the electronic health record (EHR). The cross-reactivity rate with cephalosporins (Β-lactam antibiotics) in patients reporting a penicillin allergy is < 1%, which suggests that many patients with reported penicillin allergies can safely receive them.2 Despite this, patients with self-reported penicillin allergies often receive non–Β-lactam antibiotic agents, which may be associated with an increased risk of adverse drug reactions (ADRs), increased health care costs, and inferior clinical outcomes.3
Several strategies are recommended to assess patients with self-reported penicillin allergies. According to the CDC, evaluating a patient who reports a penicillin or other Β-lactam antibiotic allergy involves 3 steps: (1) obtaining a thorough medical history, including previous exposures to penicillin or other Β-lactam antibiotic; (2) performing a skin test using the penicillin major and minor determinants; and (3) among those who have a negative penicillin skin test, performing an observed oral challenge with 250 mg amoxicillin before proceeding directly to treatment with the indicated Β-lactam therapy.4
Most existing clinical guidance for assessing patients with self-reported penicillin allergies stems from site-specific policies and primarily focuses on oral amoxicillin challenges or penicillin skin testing (PST). However, performing these tests may not be feasible at all facilities due to time constraints and lack of allergists. Therefore, alternative strategies are necessary, such as conducting detailed patient interviews. Few studies have evaluated switching to Β-lactam agents following a penicillin allergy interview alone. However, with thorough patient histories and detailed interviews, patients with reported penicillin allergies can safely use Β-lactam antibiotics.5 Implementing this procedure provides a cost-savings opportunity by not having to administer additional antibiotics for testing in addition to improving antibiotic stewardship.
The Memphis Veterans Affairs Medical Center (MVAMC) created the Allergy to Β-Lactam Evaluation (ABLE) process to clarify and remove penicillin allergies. The process involves conducting a thorough chart review and patient interview followed by completion of a note template that provides recommendations about patient allergies and Β-lactam prescribing. Mitchell et al found that the pharmacist-led process to be beneficial for addressing Β-lactam allergy clearance.6 As a result, the ABLE process was implemented at several other US Department of Veterans Affairs (VA) medical centers (VAMCs). Using the ABLE template, the purpose of this study was to evaluate the impact of a pharmacist-led penicillin allergy initiative on penicillin allergy delabeling with an interview process alone.
Methods
Prior to ABLE process implementation, there were no standardized procedures for documenting allergy histories. ABLE was implemented at the Robley Rex VAMC (RRVAMC) in November 2022. During the interview phase, patients were initially identified during admission via TheraDoc as having either a penicillin allergy or ADR. The infectious disease pharmacist or pharmacy resident interviewed patients with documented penicillin allergies or ADRs using a standardized questionnaire (eAppendix 1). Not all identified patients could be interviewed. Patients currently receiving an antibiotic were prioritized for interviews. Patients were excluded if they declined or were unable to be interviewed, although a patient’s caregiver(s) could be interviewed in person or via telephone, if the patient was not available.
Following the interview, pharmacists used guidance from the ABLE process in addition to a detailed EHR review to determine whether the patient was eligible for an allergy update or removal and/or switch to a Β-lactam antibiotic (Figure). If eligible for modification, the interviewing pharmacist made the necessary changes. A templated process note with patient-specific recommendations was entered into the Computerized Patient Record System (CPRS) and the primary care team attending physician was added as an additional signer to be alerted in the system note (eAppendix 2).

This single-center, retrospective cohort study involved review of CPRS notes and clinical interviews in the interviewed group. Hospitalized patients at the RRVAMC aged ≥ 18 years with a documented penicillin allergy or ADR were included. The historical control group consisted of patients admitted between October 31, 2019, and October 31, 2022, and the intervention group consisted of patients admitted between November 1, 2022, and March 1, 2023. Patients in the historical control group were matched 1:1 to the intervention group for penicillin allergy severity (allergy [IgE-mediated], unknown, adverse effect, severe cutaneous or other non–IgE-mediated reaction) and whether they received a noncarbapenem non–Β-lactam antibiotic.
The primary outcome was the number of patient allergies/ADRs removed or changed on patient profiles regardless of whether their antibiotic regimen was changed. This outcome was further assessed by evaluating the number of patient allergies or ADRs removed or changed on patient profiles with or without a change in antibiotic regimen. Primary outcomes were analyzed using χ2 and/ or Fisher exact tests, as appropriate to determine statistically significant differences between the interviewed group and the historical control.
Results
Seventy patients were included: 35 patients in the interviewed group and 35 patients in the historical control group, respectively. Both groups had a mean age of 72 years and predominantly included White male patients (Table 1). Following the interview, the allergy profile was modified for 6 patients (17%) in the interview group vs 0 patients in the control group (P = .03) (Table 2). The primary outcome was analyzed separately regardless of an antibiotic regimen change. There was not a statistically significant difference between groups when assessing patients for change in therapy (P > .99). All 6 patients with an allergy profile modification had no change in antibiotic regimen.


Discussion
This study suggests the ABLE process may be a valuable tool for adjusting penicillin allergies or ADRs within patient EHRs. In the interview group, allergies were modified in 6 (17%) patients while no patients in the control group had allergy modifications. Of the 6 allergy profile modifications, 4 allergy labels were changed from an allergy to an ADR. These patients were cleared to receive future Β-lactam antibiotics after clinicians recognized the lack of a true IgE-mediated allergic reaction. In addition, 2 of the modified allergy profiles removed the allergy designation. Although this represents a small subset of interviewed patients, it illustrates the clinical effectiveness of an interview process alone to remove penicillin allergy designations.
Previous research has assessed the impact of pharmacist intervention on penicillin allergy clarification. Mitchell et al implemented a pharmacist-driven Β-lactam allergy assessment and penicillin allergy clinic (PAC) at the MVAMC with the goal of evaluating its impact on allergy clearance. In their study, clinical pharmacy specialists evaluated patients with Β-lactam allergies, and those deemed eligible were later seen in the PAC. Among the 246 patients evaluated using the Β-lactam allergy assessment alone and who were not seen in the PAC, 25% had their penicillin allergy removed following a detailed assessment.6
Song et al evaluated the effectiveness and feasibility of a pharmacist-driven penicillin allergy delabeling pilot program without skin testing or oral challenges. Patients with penicillin allergies were interviewed by a pharmacy resident using a standardized checklist. Among the 66 patients interviewed, 12 (18%) met the criteria for delabeling and consented to removal of their allergy.7 The delabeling rates in these 2 studies are similar to the 17% rate of allergy modification in our study, although this study is the only one to compare results to a historical control group.
Harper et al evaluated the impact of a penicillin allergy assessment, including penicillin skin testing and oral amoxicillin challenges, on delabeling penicillin allergies. Pharmacists completed a penicillin allergy assessment and performed penicillin skin testing and/or oral amoxicillin challenges for eligible patients. Of 35 patients, 31 (89%) had their penicillin allergies delabeled in the EHR.8 The rate of penicillin allergy delabeling in Harper et al was likely higher than that seen in our study due to the use of oral challenge and skin testing. Regardless, a detailed penicillin allergy interview alone was effective at RRVAMC, resulting in a significant rate of allergy removal or change. This supports the use of detailed penicillin allergy assessments in settings where penicillin skin testing or oral challenges may not be feasible.
Mann et al demonstrated the effectiveness of penicillin allergy assessments in switching eligible patients to Β-lactam antibiotics. Their single-center, prospective study assessed the impact of a pharmacist-driven detailed penicillin allergy interview initiative. Interviews that evaluated potential changes to allergy profiles were conducted with 175 patients. Of these patients, 135 (77.1%) were on antimicrobial therapy and 42 (31.1%) patients receiving therapy met criteria to switch to a noncarbapenem Β-lactam antibiotic. Thirty-one patients (73.8%) switched with no signs or symptoms of intolerance demonstrating that an interview can be a valuable tool for antibiotic optimization, specifically in patients with penicillin allergy.9 No patients in our study switched antibiotic therapy, likely because only a small number of patients were eligible for transition to a noncarbapenem Β-lactam antibiotic. In the Mann et al study, non–Β-lactam antibiotics, such as fluoroquinolones and carbapenems, accounted for > 75% of the antibiotics used.
Limitations
The sample size of this study was small and its duration was short. There is a risk for selection bias as not all identified patients were able to be interviewed while admitted, but patients on antibiotics were prioritized as they were most likely to directly benefit during their current admission from a modification of their allergy. Most patients in the study were White and male, which may limit the generalizability of the results. Additionally, recommendations regarding antibiotic changes were primarily communicated to the treatment team based on a templated note in CPRS alone. Therefore, implementation of these recommendations largely relied upon nonverbal communication. Direct pharmacist-physician communication could have led to a larger impact on antimicrobial therapy changes. The interviewer’s participation in daily rounds with time allotted to discuss this topic can be considered in the future to improve these processes.
Conclusions
This study found that the ABLE process identified patients for penicillin allergy delabeling. With the high prevalence of inaccurate penicillin allergy documentation, this tool offers VA health care systems a way to empower pharmacists in allergy clarification, leading to improvements in antibiotic stewardship. Although the sample size was small, the ABLE process may provide a framework for VA clinicians. Future research has the potential to demonstrate the practicality and effectiveness this pharmacist-led penicillin allergy interview process can offer clinicians.
- Health care providers. Clinical features of penicillin allergy. Centers for Disease Control and Prevention. August 25, 2025. Accessed February 4, 2026. https://www.cdc.gov /antibiotic-use/hcp/clinical-signs/index.html
- Wrynn AF. Penicillin allergies: A guide for NPs. Nurse Pract. 2022;47:30-36. doi:10.1097/01.NPR.0000855312.11145.78
- Mohsen S, Dickinson JA, Somayaji R. Update on the adverse effects of antimicrobial therapies in community practice. Can Fam Physician. 2020;66:651-659.
- Sexually Transmitted Infections Treatment Guidelines, 2021. Managing persons who have a history of penicillin allergy. Centers for Disease Control and Prevention. September 21, 2022. Accessed February 4, 2026. https:// www.cdc.gov/std/treatment-guidelines/penicillin-allergy .htm
- Holmes AK, Bennett NT, Berry TP. Pharmacy driven assessment of appropriate antibiotic selection in patients with reported beta-lactam allergy. J Am Coll Clin Pharm. 2019;2:509-514. doi:10.1002/jac5.1135
- Mitchell AB, Ness RA, Bennett JG, et al. Implementation and impact of a Β-lactam allergy assessment protocol in a veteran population. Fed Pract. 2021;38:420-425. doi:10.12788/fp.0172
- Song YC, Nelson ZJ, Wankum MA, et al. Effectiveness and feasibility of pharmacist-driven penicillin allergy de-labeling pilot program without skin testing or oral challenges. Pharmacy (Basel). 2021;9:127. doi:10.3390/pharmacy9030127
- Harper HM, Sanchez M. Review of pharmacist driven penicillin allergy assessments and skin testing: a multicenter case-series. Hosp Pharm. 2022;57:469-473. doi:10.1177/00185787211046862
- Mann KL, Wu JY, Shah SS. Implementation of a pharmacist- driven detailed penicillin allergy interview. Ann Pharmacother. 2020;54:364-370. doi:10.1177/1060028019884874
- Health care providers. Clinical features of penicillin allergy. Centers for Disease Control and Prevention. August 25, 2025. Accessed February 4, 2026. https://www.cdc.gov /antibiotic-use/hcp/clinical-signs/index.html
- Wrynn AF. Penicillin allergies: A guide for NPs. Nurse Pract. 2022;47:30-36. doi:10.1097/01.NPR.0000855312.11145.78
- Mohsen S, Dickinson JA, Somayaji R. Update on the adverse effects of antimicrobial therapies in community practice. Can Fam Physician. 2020;66:651-659.
- Sexually Transmitted Infections Treatment Guidelines, 2021. Managing persons who have a history of penicillin allergy. Centers for Disease Control and Prevention. September 21, 2022. Accessed February 4, 2026. https:// www.cdc.gov/std/treatment-guidelines/penicillin-allergy .htm
- Holmes AK, Bennett NT, Berry TP. Pharmacy driven assessment of appropriate antibiotic selection in patients with reported beta-lactam allergy. J Am Coll Clin Pharm. 2019;2:509-514. doi:10.1002/jac5.1135
- Mitchell AB, Ness RA, Bennett JG, et al. Implementation and impact of a Β-lactam allergy assessment protocol in a veteran population. Fed Pract. 2021;38:420-425. doi:10.12788/fp.0172
- Song YC, Nelson ZJ, Wankum MA, et al. Effectiveness and feasibility of pharmacist-driven penicillin allergy de-labeling pilot program without skin testing or oral challenges. Pharmacy (Basel). 2021;9:127. doi:10.3390/pharmacy9030127
- Harper HM, Sanchez M. Review of pharmacist driven penicillin allergy assessments and skin testing: a multicenter case-series. Hosp Pharm. 2022;57:469-473. doi:10.1177/00185787211046862
- Mann KL, Wu JY, Shah SS. Implementation of a pharmacist- driven detailed penicillin allergy interview. Ann Pharmacother. 2020;54:364-370. doi:10.1177/1060028019884874
Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility
Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility
Outcomes From the Use of Cefazolin for Surgical Prophylaxis in Patients Allergic to Penicillin
Outcomes From the Use of Cefazolin for Surgical Prophylaxis in Patients Allergic to Penicillin
Given its safety profile and bactericidal activity against the predominant organisms causing surgical site infections (SSIs), cefazolin remains the most popular choice for surgical prophylaxis.1 Cefazolin offers protection against the pathogens most likely to contaminate the surgical site while minimizing inappropriate methicillin- resistant Staphylococcus aureus coverage that occurs with alternatives such as vancomycin and clindamycin. Documented allergies to Β-lactam antibiotics have historically forced clinicians to avoid the use of cephalosporins due to the potential risk of cross-reactivity. True type 1 (immunoglobin E [IgE]-mediated) cross-allergic reactions between penicillin and cephalosporins are rare, and previously reported data indicate cross-reactivity as a result of antibody recognition is more closely related to the side-chain identity rather than the Β-lactam ring.2,3
About 10% of US patients report having a penicillin allergy; however, < 1% of the population has a true IgE-mediated allergic reaction.4 Previous research that has challenged penicillin allergies with cefazolin for surgical prophylaxis has reported minimal rates of allergic reactions.2-5
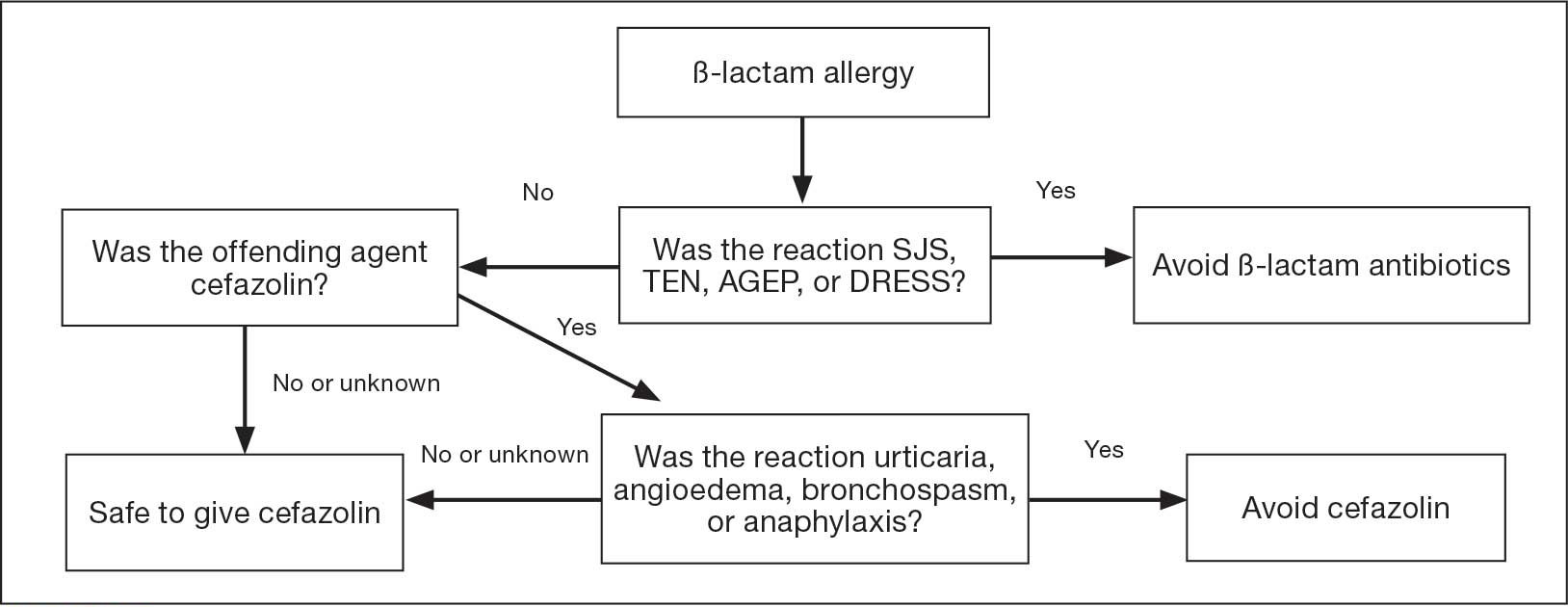
In previous trials, patients with a history of delayed skin reactions, such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), were excluded. Additionally, patients with an allergy to cefazolin including those with urticaria, angioedema, bronchospasm, or anaphylaxis, were excluded from perioperative retrial of cefazolin. Grant et al found that cefazolin can be safely given to patients with IgE-mediated reactions to penicillin and other cephalosporins due to a structurally different side chain.3
In January 2023, the Veteran Health Indiana (VHI) pharmacy team in conjunction with surgery, infectious disease, and anesthesiology, implemented a screening tool as an amendment to perioperative antibiotic guidance to help determine which patients with a documented penicillin allergy could be candidates for perioperative cefazolin. The implemented screening tool (Allergy Clarification for Cefazolin Evidence-Based Prescribing Tool) has been described by Lam et al, who reported that an increased proportion of patients with documented penicillin allergy received cefazolin without more adverse drug reactions (ADRs).5 Patients with a Β-lactam allergy were eligible to receive cefazolin unless the ADR was SJS, TEN, or DRESS, or the offending agent was cefazolin and the patient experienced urticaria, angioedema, bronchospasm, or anaphylaxis. If the reaction was not from cefazolin or was unknown, patients were eligible to receive cefazolin (Figure).
To date, minimal data exist to evaluate the incidence of ADRs when cefazolin is given perioperatively to patients with a previously documented penicillin allergy. The purpose of this study was to evaluate the incidence of allergic ADRs in patients who had a documented penicillin allergy and received periprocedural antibiotics.
Methods
This single-center, retrospective chart review used the US Department of Veterans Affairs (VA) Computerized Patient Record System (CPRS) to identify patients with a documented penicillin allergy who underwent an operation and received periprocedural antibiotics between February 1, 2023, and January 31, 2024. This study was reviewed and approved by the Indiana University Health Institutional Review Board and the VHI Research and Development Committee.
Patients were enrolled if they were aged ≥ 18 years, had a documented penicillin allergy, underwent a surgical intervention, and received perioperative antibiotics during the study period. Patients were excluded if they had a documented penicillin allergy resulting in severe delayed skin reactions (ie, SJS, TEN, or DRESS). These criteria produced 197 surgical procedures. Data were collected for each surgical procedure, so patients could be included more than once. Patient history of allergic reaction to penicillin was obtained through CPRS.
The primary endpoint was the percentage of allergic ADRs in patients with penicillin allergies receiving cefazolin perioperatively. Secondary outcomes included the appropriateness of the antibiotic regimen in congruence with American System of Health Pharmacists (ASHP) recommendations, incidence of SSIs within 30 days of the procedure, incidence of ADRs in those with a history of anaphylaxis vs nonanaphylaxis allergy, incidence of allergic reaction requiring pharmacologic and nonpharmacologic interventions, and incidence of acute kidney injury (AKI). AKI was defined as an increase in serum creatinine by ≥ 0.3 mg/dL within 48 hours or an increase in serum creatinine to ≥ 1.5 times baseline.
Demographic data included sex, age, race, preoperative serum creatinine, and postoperative serum creatinine. Anaphylaxis was defined as an acute onset of illness (within minutes to several hours) with involvement of skin, mucosal tissue, or both involving either respiratory compromise or reduced blood pressures. Allergic reactions were defined as facial, tongue, throat, airway, lip, mouth, periorbital, or eye swelling, urticaria, angioedema, dyspnea, anaphylaxis, or a positive penicillin skin test. Additionally, data collected included the description and severity of postprophylactic antibiotic reaction, antibiotic choice, interventions required for the allergic reaction, SSI occurrence, date of SSI, operating specialty, and postoperative change in renal function.
Descriptive statistics, including mean, SD, and percentages were reported for baseline characteristics of the study population. Percentages were used to demonstrate the differences in primary and secondary outcomes for each study group. Fisher exact tests were used for incidence of ADRs in patients with penicillin allergy who received cefazolin and reported incidence of SSIs.
Results
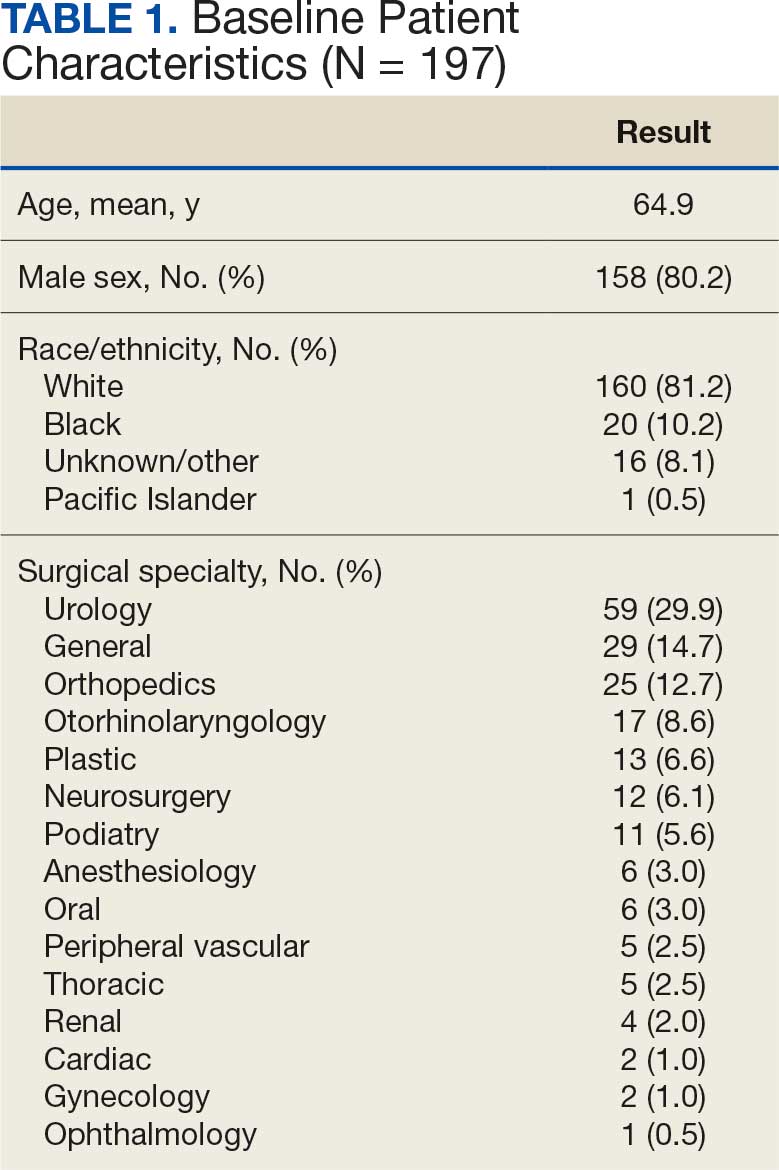
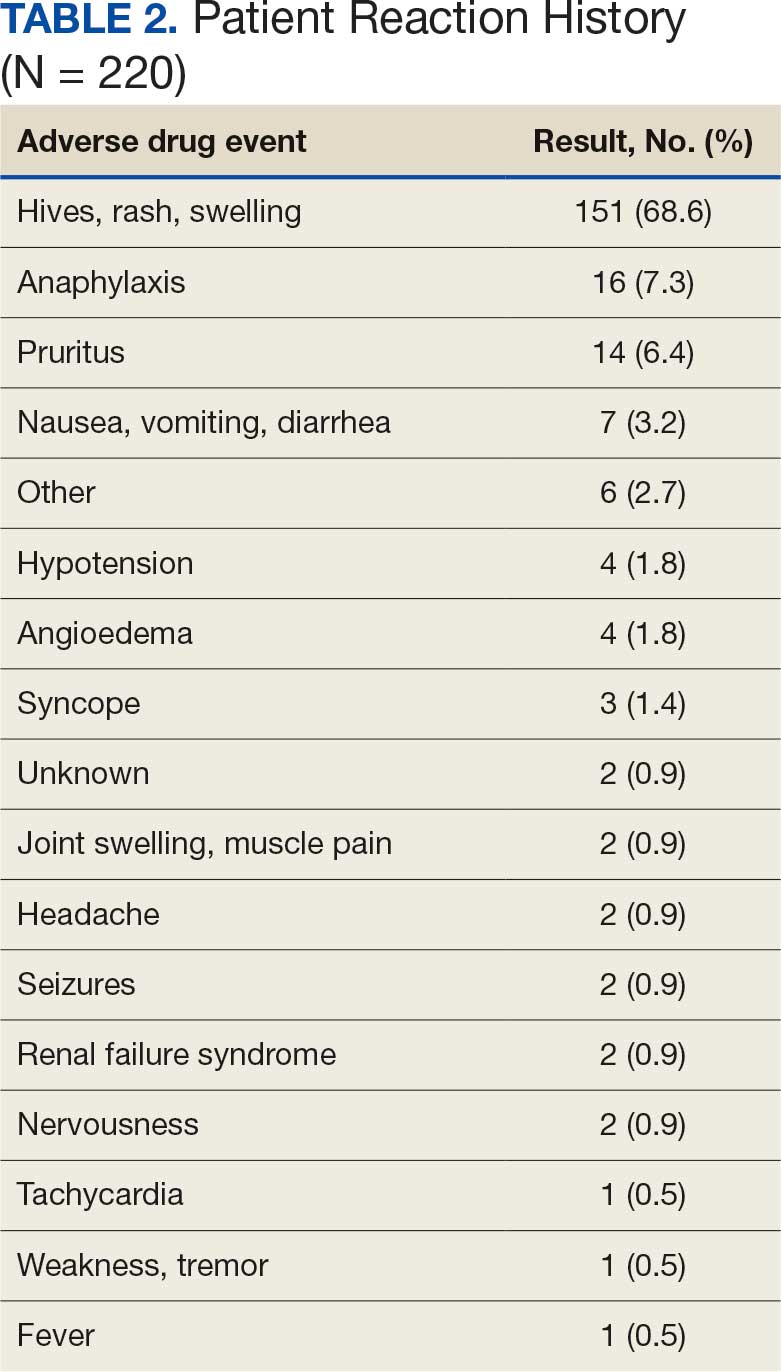
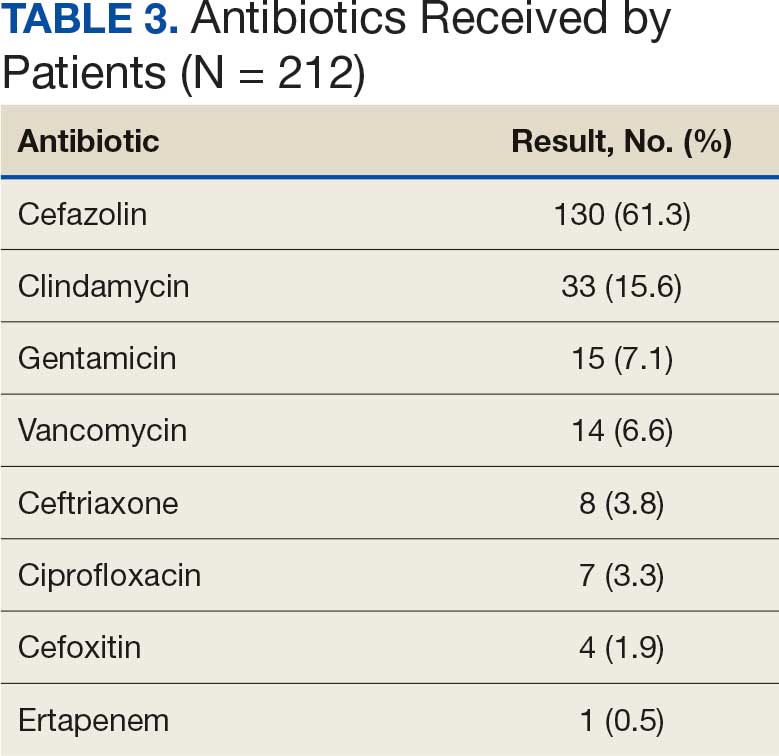
A total of 197 surgical procedures in patients with a documented penicillin allergy were included; 127 procedures used cefazolin perioperatively, 3 procedures used cefazolin plus gentamicin, and 67 procedures used other antibiotics. Most patients were White (n = 160; 81.2%), male (n = 158; 80.2%), and had a mean age of 64.9 years. Urology was the most common surgical specialty (n = 59; 29.9%) (Table 1). Of the 16 patients with documented penicillin anaphylaxis reaction, 8 received cefazolin and 8 received a different antibiotic. A total of 181 patients reported a nonanaphylaxis allergy. One hundred fifty-one patients (68.6%) reported a reaction history of hives, rash, or swelling (Table 2). Patients could report ≥ 1 reaction. The most prevalent antibiotics used were cefazolin, which was used by 130 patients (61.3%), and clindamycin which was used by 33 patients (15.6%) (Table 3). Patients could receive ≥ 1 antibiotic.
For the primary outcome, the incidence of allergic reactions in patients allergic to penicillin, there was no incidence of allergic reactions in either the cefazolin or other group. Given the absence of reactions, no interventions were required.
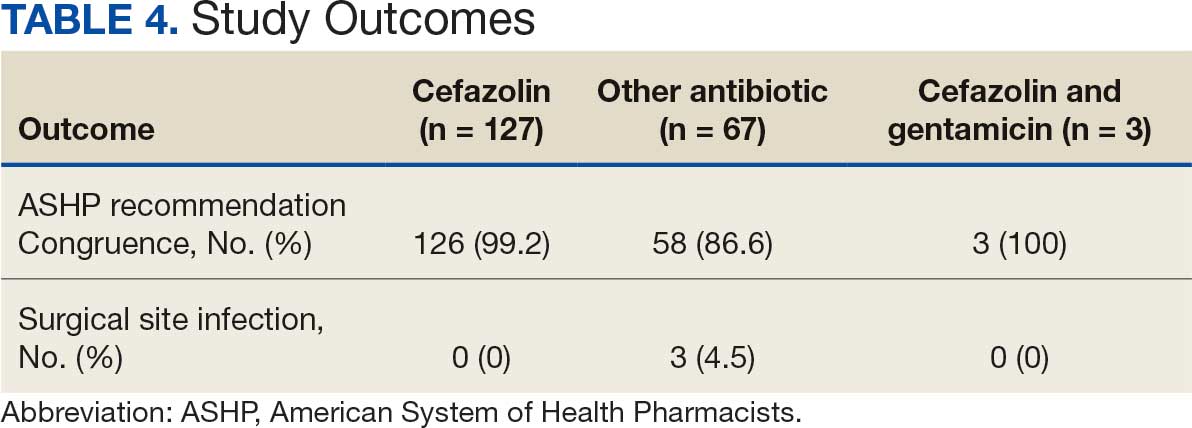
There were no ADRs in those with history of anaphylaxis or nonanaphylaxis allergy. In the cefazolin group, 126 of 127 surgical procedure regimens (99.2%) were congruent with ASHP recommendations, all 3 surgical procedures regimens in the cefazolin plus gentamicin group were congruent with ASHP recommendations, and 58 of 67 surgical procedure regimens (86.6%) in the other antibiotic group were congruent with ASHP recommendations. None of the 127 patients in the cefazolin group or of the 3 patients in the cefazolin plus gentamicin group reported an SSI, and 3 of 67 patients (4.5%) had an SSI in the other antibiotic group. One procedure that resulted in SSI was not congruent with ASHP recommendations. Twenty-four patients had 2 serum creatinine levels drawn within 48 hours of surgery. One of 12 patients (8.3%) and 0 of 12 patients had an AKI in the cefazolin and other antibiotic group, respectively (Table 4).
Discussion
Implementation of a screening tool at VHI allowed patients with documented penicillin allergy, including anaphylaxis, to receive cefazolin perioperatively. Broad spectrum antibiotics such as vancomycin, clindamycin, and fluoroquinolones are frequently used in patients allergic to penicillin, which can increase health care costs, risk of toxicity, and antimicrobial resistance.4 There was no incidence of allergic reactions noted in patients allergic to penicillin who received cefazolin. When comparing the incidence of observed allergic reactions to received perioperative antibiotics in the cefazolin group to previously published literature, no difference in allergy rates (P = .09) was found.3 Most antibiotics administered were congruent with ASHP guideline recommendations, and most patients eligible for cefazolin received it perioperatively.
Similar to this study, Goodman et al concluded that cefazolin appears to be a safe regimen in patients with documented penicillin anaphylactic reaction for surgical prophylaxis with only 1 (0.2%) potential allergic reaction.6 Patients who received cefazolin perioperatively had a statistically significant decrease in SSI rates. There were no clinically or statistically significant differences found between the proportion of allergic reactions or ADRs when compared to alternative antibiotics. Lessard et al concluded that a pharmacist-led interdisciplinary collaborative practice agreement increased cefazolin use in patients allergic to penicillin, including those with urticaria and anaphylaxis, with no reported ADRs.7 This study further demonstrated the safety of cefazolin use in patients with anaphylaxis to penicillin.
Limitations
This study’s single-center, retrospective design, patient population, and small sample size limit the generalizability of its results. The data collected are dependent on documentation in the chart. No ADRs were reported from the antibiotics patients received perioperatively. When considering safety data, information such as serum creatinine were available only in CPRS and some patients did not receive a postprocedure serum creatinine level. Additionally, this study did not investigate whether there was an increase in preferred preoperative antimicrobial prophylaxis after implementation of this protocol.
Conclusions
The results of this study support the use of cefazolin perioperatively in patients allergic to penicillin, including those with a history of anaphylaxis. Additional research should be conducted to validate data given the low incidence of ADRs. The primary outcome did not reach statistical significance, but the results may be clinically significant from a stewardship and safety perspective. VHI continues to use the screening tool described in this article.
- Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm. 2013;70:195-283. doi:10.2146/ajhp120568
- Romano A, Valluzzi RL, Caruso C, et al. Tolerability of cefazolin and ceftibuten in patients with IgE-mediated aminopenicillin allergy. J Allergy Clin Immunol Pract. 2020;8:1989-1993.e2. doi:10.1016/j.jaip.2020.02.025
- Grant JM, Song WHC, Shajari S, et al. Safety of administering cefazolin versus other antibiotics in penicillin- allergic patients for surgical prophylaxis at a major Canadian teaching hospital. Surgery. 2021;170:783-789. doi:10.1016/j.surg.2021.03.022
- Centers for Disease Control and Prevention. Clinical Features of Penicillin Allergy. August 25, 2025. Accessed January 6, 2026. https://www.cdc.gov/antibiotic-use/hcp/clinical-signs/index.html
- Lam PW, Tarighi P, Elligsen M, et al. Impact of the allergy clarification for cefazolin evidence-based prescribing tool on receipt of preferred perioperative prophylaxis: an interrupted time series study. Clin Infect Dis. 2020;71:2955- 2957. doi:10.1093/cid/ciaa516
- Goodman EJ, Morgan MJ, Johnson Pa, et al. Cephalosporins can be given to penicillin-allergic patients who do not exhibit an anaphylactic response. J Clin Anesth. 2001;13:561-564. doi:10.1016/s0952-8180(01)00329-4
- Lessard S, Huiras C, Dababneh A, et al. Pharmacist adjustment of preoperative antibiotic orders to the preferred preoperative antibiotic cefazolin for patients with penicillin allergy labeling. Am J Health Syst Pharm. 2023;80:532- 536. doi:10.1093/ajhp/zxac385
Given its safety profile and bactericidal activity against the predominant organisms causing surgical site infections (SSIs), cefazolin remains the most popular choice for surgical prophylaxis.1 Cefazolin offers protection against the pathogens most likely to contaminate the surgical site while minimizing inappropriate methicillin- resistant Staphylococcus aureus coverage that occurs with alternatives such as vancomycin and clindamycin. Documented allergies to Β-lactam antibiotics have historically forced clinicians to avoid the use of cephalosporins due to the potential risk of cross-reactivity. True type 1 (immunoglobin E [IgE]-mediated) cross-allergic reactions between penicillin and cephalosporins are rare, and previously reported data indicate cross-reactivity as a result of antibody recognition is more closely related to the side-chain identity rather than the Β-lactam ring.2,3
About 10% of US patients report having a penicillin allergy; however, < 1% of the population has a true IgE-mediated allergic reaction.4 Previous research that has challenged penicillin allergies with cefazolin for surgical prophylaxis has reported minimal rates of allergic reactions.2-5
In previous trials, patients with a history of delayed skin reactions, such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), were excluded. Additionally, patients with an allergy to cefazolin including those with urticaria, angioedema, bronchospasm, or anaphylaxis, were excluded from perioperative retrial of cefazolin. Grant et al found that cefazolin can be safely given to patients with IgE-mediated reactions to penicillin and other cephalosporins due to a structurally different side chain.3
In January 2023, the Veteran Health Indiana (VHI) pharmacy team in conjunction with surgery, infectious disease, and anesthesiology, implemented a screening tool as an amendment to perioperative antibiotic guidance to help determine which patients with a documented penicillin allergy could be candidates for perioperative cefazolin. The implemented screening tool (Allergy Clarification for Cefazolin Evidence-Based Prescribing Tool) has been described by Lam et al, who reported that an increased proportion of patients with documented penicillin allergy received cefazolin without more adverse drug reactions (ADRs).5 Patients with a Β-lactam allergy were eligible to receive cefazolin unless the ADR was SJS, TEN, or DRESS, or the offending agent was cefazolin and the patient experienced urticaria, angioedema, bronchospasm, or anaphylaxis. If the reaction was not from cefazolin or was unknown, patients were eligible to receive cefazolin (Figure).

To date, minimal data exist to evaluate the incidence of ADRs when cefazolin is given perioperatively to patients with a previously documented penicillin allergy. The purpose of this study was to evaluate the incidence of allergic ADRs in patients who had a documented penicillin allergy and received periprocedural antibiotics.
Methods
This single-center, retrospective chart review used the US Department of Veterans Affairs (VA) Computerized Patient Record System (CPRS) to identify patients with a documented penicillin allergy who underwent an operation and received periprocedural antibiotics between February 1, 2023, and January 31, 2024. This study was reviewed and approved by the Indiana University Health Institutional Review Board and the VHI Research and Development Committee.
Patients were enrolled if they were aged ≥ 18 years, had a documented penicillin allergy, underwent a surgical intervention, and received perioperative antibiotics during the study period. Patients were excluded if they had a documented penicillin allergy resulting in severe delayed skin reactions (ie, SJS, TEN, or DRESS). These criteria produced 197 surgical procedures. Data were collected for each surgical procedure, so patients could be included more than once. Patient history of allergic reaction to penicillin was obtained through CPRS.
The primary endpoint was the percentage of allergic ADRs in patients with penicillin allergies receiving cefazolin perioperatively. Secondary outcomes included the appropriateness of the antibiotic regimen in congruence with American System of Health Pharmacists (ASHP) recommendations, incidence of SSIs within 30 days of the procedure, incidence of ADRs in those with a history of anaphylaxis vs nonanaphylaxis allergy, incidence of allergic reaction requiring pharmacologic and nonpharmacologic interventions, and incidence of acute kidney injury (AKI). AKI was defined as an increase in serum creatinine by ≥ 0.3 mg/dL within 48 hours or an increase in serum creatinine to ≥ 1.5 times baseline.
Demographic data included sex, age, race, preoperative serum creatinine, and postoperative serum creatinine. Anaphylaxis was defined as an acute onset of illness (within minutes to several hours) with involvement of skin, mucosal tissue, or both involving either respiratory compromise or reduced blood pressures. Allergic reactions were defined as facial, tongue, throat, airway, lip, mouth, periorbital, or eye swelling, urticaria, angioedema, dyspnea, anaphylaxis, or a positive penicillin skin test. Additionally, data collected included the description and severity of postprophylactic antibiotic reaction, antibiotic choice, interventions required for the allergic reaction, SSI occurrence, date of SSI, operating specialty, and postoperative change in renal function.
Descriptive statistics, including mean, SD, and percentages were reported for baseline characteristics of the study population. Percentages were used to demonstrate the differences in primary and secondary outcomes for each study group. Fisher exact tests were used for incidence of ADRs in patients with penicillin allergy who received cefazolin and reported incidence of SSIs.
Results
A total of 197 surgical procedures in patients with a documented penicillin allergy were included; 127 procedures used cefazolin perioperatively, 3 procedures used cefazolin plus gentamicin, and 67 procedures used other antibiotics. Most patients were White (n = 160; 81.2%), male (n = 158; 80.2%), and had a mean age of 64.9 years. Urology was the most common surgical specialty (n = 59; 29.9%) (Table 1). Of the 16 patients with documented penicillin anaphylaxis reaction, 8 received cefazolin and 8 received a different antibiotic. A total of 181 patients reported a nonanaphylaxis allergy. One hundred fifty-one patients (68.6%) reported a reaction history of hives, rash, or swelling (Table 2). Patients could report ≥ 1 reaction. The most prevalent antibiotics used were cefazolin, which was used by 130 patients (61.3%), and clindamycin which was used by 33 patients (15.6%) (Table 3). Patients could receive ≥ 1 antibiotic.



For the primary outcome, the incidence of allergic reactions in patients allergic to penicillin, there was no incidence of allergic reactions in either the cefazolin or other group. Given the absence of reactions, no interventions were required.
There were no ADRs in those with history of anaphylaxis or nonanaphylaxis allergy. In the cefazolin group, 126 of 127 surgical procedure regimens (99.2%) were congruent with ASHP recommendations, all 3 surgical procedures regimens in the cefazolin plus gentamicin group were congruent with ASHP recommendations, and 58 of 67 surgical procedure regimens (86.6%) in the other antibiotic group were congruent with ASHP recommendations. None of the 127 patients in the cefazolin group or of the 3 patients in the cefazolin plus gentamicin group reported an SSI, and 3 of 67 patients (4.5%) had an SSI in the other antibiotic group. One procedure that resulted in SSI was not congruent with ASHP recommendations. Twenty-four patients had 2 serum creatinine levels drawn within 48 hours of surgery. One of 12 patients (8.3%) and 0 of 12 patients had an AKI in the cefazolin and other antibiotic group, respectively (Table 4).

Discussion
Implementation of a screening tool at VHI allowed patients with documented penicillin allergy, including anaphylaxis, to receive cefazolin perioperatively. Broad spectrum antibiotics such as vancomycin, clindamycin, and fluoroquinolones are frequently used in patients allergic to penicillin, which can increase health care costs, risk of toxicity, and antimicrobial resistance.4 There was no incidence of allergic reactions noted in patients allergic to penicillin who received cefazolin. When comparing the incidence of observed allergic reactions to received perioperative antibiotics in the cefazolin group to previously published literature, no difference in allergy rates (P = .09) was found.3 Most antibiotics administered were congruent with ASHP guideline recommendations, and most patients eligible for cefazolin received it perioperatively.
Similar to this study, Goodman et al concluded that cefazolin appears to be a safe regimen in patients with documented penicillin anaphylactic reaction for surgical prophylaxis with only 1 (0.2%) potential allergic reaction.6 Patients who received cefazolin perioperatively had a statistically significant decrease in SSI rates. There were no clinically or statistically significant differences found between the proportion of allergic reactions or ADRs when compared to alternative antibiotics. Lessard et al concluded that a pharmacist-led interdisciplinary collaborative practice agreement increased cefazolin use in patients allergic to penicillin, including those with urticaria and anaphylaxis, with no reported ADRs.7 This study further demonstrated the safety of cefazolin use in patients with anaphylaxis to penicillin.
Limitations
This study’s single-center, retrospective design, patient population, and small sample size limit the generalizability of its results. The data collected are dependent on documentation in the chart. No ADRs were reported from the antibiotics patients received perioperatively. When considering safety data, information such as serum creatinine were available only in CPRS and some patients did not receive a postprocedure serum creatinine level. Additionally, this study did not investigate whether there was an increase in preferred preoperative antimicrobial prophylaxis after implementation of this protocol.
Conclusions
The results of this study support the use of cefazolin perioperatively in patients allergic to penicillin, including those with a history of anaphylaxis. Additional research should be conducted to validate data given the low incidence of ADRs. The primary outcome did not reach statistical significance, but the results may be clinically significant from a stewardship and safety perspective. VHI continues to use the screening tool described in this article.
Given its safety profile and bactericidal activity against the predominant organisms causing surgical site infections (SSIs), cefazolin remains the most popular choice for surgical prophylaxis.1 Cefazolin offers protection against the pathogens most likely to contaminate the surgical site while minimizing inappropriate methicillin- resistant Staphylococcus aureus coverage that occurs with alternatives such as vancomycin and clindamycin. Documented allergies to Β-lactam antibiotics have historically forced clinicians to avoid the use of cephalosporins due to the potential risk of cross-reactivity. True type 1 (immunoglobin E [IgE]-mediated) cross-allergic reactions between penicillin and cephalosporins are rare, and previously reported data indicate cross-reactivity as a result of antibody recognition is more closely related to the side-chain identity rather than the Β-lactam ring.2,3
About 10% of US patients report having a penicillin allergy; however, < 1% of the population has a true IgE-mediated allergic reaction.4 Previous research that has challenged penicillin allergies with cefazolin for surgical prophylaxis has reported minimal rates of allergic reactions.2-5
In previous trials, patients with a history of delayed skin reactions, such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), were excluded. Additionally, patients with an allergy to cefazolin including those with urticaria, angioedema, bronchospasm, or anaphylaxis, were excluded from perioperative retrial of cefazolin. Grant et al found that cefazolin can be safely given to patients with IgE-mediated reactions to penicillin and other cephalosporins due to a structurally different side chain.3
In January 2023, the Veteran Health Indiana (VHI) pharmacy team in conjunction with surgery, infectious disease, and anesthesiology, implemented a screening tool as an amendment to perioperative antibiotic guidance to help determine which patients with a documented penicillin allergy could be candidates for perioperative cefazolin. The implemented screening tool (Allergy Clarification for Cefazolin Evidence-Based Prescribing Tool) has been described by Lam et al, who reported that an increased proportion of patients with documented penicillin allergy received cefazolin without more adverse drug reactions (ADRs).5 Patients with a Β-lactam allergy were eligible to receive cefazolin unless the ADR was SJS, TEN, or DRESS, or the offending agent was cefazolin and the patient experienced urticaria, angioedema, bronchospasm, or anaphylaxis. If the reaction was not from cefazolin or was unknown, patients were eligible to receive cefazolin (Figure).

To date, minimal data exist to evaluate the incidence of ADRs when cefazolin is given perioperatively to patients with a previously documented penicillin allergy. The purpose of this study was to evaluate the incidence of allergic ADRs in patients who had a documented penicillin allergy and received periprocedural antibiotics.
Methods
This single-center, retrospective chart review used the US Department of Veterans Affairs (VA) Computerized Patient Record System (CPRS) to identify patients with a documented penicillin allergy who underwent an operation and received periprocedural antibiotics between February 1, 2023, and January 31, 2024. This study was reviewed and approved by the Indiana University Health Institutional Review Board and the VHI Research and Development Committee.
Patients were enrolled if they were aged ≥ 18 years, had a documented penicillin allergy, underwent a surgical intervention, and received perioperative antibiotics during the study period. Patients were excluded if they had a documented penicillin allergy resulting in severe delayed skin reactions (ie, SJS, TEN, or DRESS). These criteria produced 197 surgical procedures. Data were collected for each surgical procedure, so patients could be included more than once. Patient history of allergic reaction to penicillin was obtained through CPRS.
The primary endpoint was the percentage of allergic ADRs in patients with penicillin allergies receiving cefazolin perioperatively. Secondary outcomes included the appropriateness of the antibiotic regimen in congruence with American System of Health Pharmacists (ASHP) recommendations, incidence of SSIs within 30 days of the procedure, incidence of ADRs in those with a history of anaphylaxis vs nonanaphylaxis allergy, incidence of allergic reaction requiring pharmacologic and nonpharmacologic interventions, and incidence of acute kidney injury (AKI). AKI was defined as an increase in serum creatinine by ≥ 0.3 mg/dL within 48 hours or an increase in serum creatinine to ≥ 1.5 times baseline.
Demographic data included sex, age, race, preoperative serum creatinine, and postoperative serum creatinine. Anaphylaxis was defined as an acute onset of illness (within minutes to several hours) with involvement of skin, mucosal tissue, or both involving either respiratory compromise or reduced blood pressures. Allergic reactions were defined as facial, tongue, throat, airway, lip, mouth, periorbital, or eye swelling, urticaria, angioedema, dyspnea, anaphylaxis, or a positive penicillin skin test. Additionally, data collected included the description and severity of postprophylactic antibiotic reaction, antibiotic choice, interventions required for the allergic reaction, SSI occurrence, date of SSI, operating specialty, and postoperative change in renal function.
Descriptive statistics, including mean, SD, and percentages were reported for baseline characteristics of the study population. Percentages were used to demonstrate the differences in primary and secondary outcomes for each study group. Fisher exact tests were used for incidence of ADRs in patients with penicillin allergy who received cefazolin and reported incidence of SSIs.
Results
A total of 197 surgical procedures in patients with a documented penicillin allergy were included; 127 procedures used cefazolin perioperatively, 3 procedures used cefazolin plus gentamicin, and 67 procedures used other antibiotics. Most patients were White (n = 160; 81.2%), male (n = 158; 80.2%), and had a mean age of 64.9 years. Urology was the most common surgical specialty (n = 59; 29.9%) (Table 1). Of the 16 patients with documented penicillin anaphylaxis reaction, 8 received cefazolin and 8 received a different antibiotic. A total of 181 patients reported a nonanaphylaxis allergy. One hundred fifty-one patients (68.6%) reported a reaction history of hives, rash, or swelling (Table 2). Patients could report ≥ 1 reaction. The most prevalent antibiotics used were cefazolin, which was used by 130 patients (61.3%), and clindamycin which was used by 33 patients (15.6%) (Table 3). Patients could receive ≥ 1 antibiotic.



For the primary outcome, the incidence of allergic reactions in patients allergic to penicillin, there was no incidence of allergic reactions in either the cefazolin or other group. Given the absence of reactions, no interventions were required.
There were no ADRs in those with history of anaphylaxis or nonanaphylaxis allergy. In the cefazolin group, 126 of 127 surgical procedure regimens (99.2%) were congruent with ASHP recommendations, all 3 surgical procedures regimens in the cefazolin plus gentamicin group were congruent with ASHP recommendations, and 58 of 67 surgical procedure regimens (86.6%) in the other antibiotic group were congruent with ASHP recommendations. None of the 127 patients in the cefazolin group or of the 3 patients in the cefazolin plus gentamicin group reported an SSI, and 3 of 67 patients (4.5%) had an SSI in the other antibiotic group. One procedure that resulted in SSI was not congruent with ASHP recommendations. Twenty-four patients had 2 serum creatinine levels drawn within 48 hours of surgery. One of 12 patients (8.3%) and 0 of 12 patients had an AKI in the cefazolin and other antibiotic group, respectively (Table 4).

Discussion
Implementation of a screening tool at VHI allowed patients with documented penicillin allergy, including anaphylaxis, to receive cefazolin perioperatively. Broad spectrum antibiotics such as vancomycin, clindamycin, and fluoroquinolones are frequently used in patients allergic to penicillin, which can increase health care costs, risk of toxicity, and antimicrobial resistance.4 There was no incidence of allergic reactions noted in patients allergic to penicillin who received cefazolin. When comparing the incidence of observed allergic reactions to received perioperative antibiotics in the cefazolin group to previously published literature, no difference in allergy rates (P = .09) was found.3 Most antibiotics administered were congruent with ASHP guideline recommendations, and most patients eligible for cefazolin received it perioperatively.
Similar to this study, Goodman et al concluded that cefazolin appears to be a safe regimen in patients with documented penicillin anaphylactic reaction for surgical prophylaxis with only 1 (0.2%) potential allergic reaction.6 Patients who received cefazolin perioperatively had a statistically significant decrease in SSI rates. There were no clinically or statistically significant differences found between the proportion of allergic reactions or ADRs when compared to alternative antibiotics. Lessard et al concluded that a pharmacist-led interdisciplinary collaborative practice agreement increased cefazolin use in patients allergic to penicillin, including those with urticaria and anaphylaxis, with no reported ADRs.7 This study further demonstrated the safety of cefazolin use in patients with anaphylaxis to penicillin.
Limitations
This study’s single-center, retrospective design, patient population, and small sample size limit the generalizability of its results. The data collected are dependent on documentation in the chart. No ADRs were reported from the antibiotics patients received perioperatively. When considering safety data, information such as serum creatinine were available only in CPRS and some patients did not receive a postprocedure serum creatinine level. Additionally, this study did not investigate whether there was an increase in preferred preoperative antimicrobial prophylaxis after implementation of this protocol.
Conclusions
The results of this study support the use of cefazolin perioperatively in patients allergic to penicillin, including those with a history of anaphylaxis. Additional research should be conducted to validate data given the low incidence of ADRs. The primary outcome did not reach statistical significance, but the results may be clinically significant from a stewardship and safety perspective. VHI continues to use the screening tool described in this article.
- Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm. 2013;70:195-283. doi:10.2146/ajhp120568
- Romano A, Valluzzi RL, Caruso C, et al. Tolerability of cefazolin and ceftibuten in patients with IgE-mediated aminopenicillin allergy. J Allergy Clin Immunol Pract. 2020;8:1989-1993.e2. doi:10.1016/j.jaip.2020.02.025
- Grant JM, Song WHC, Shajari S, et al. Safety of administering cefazolin versus other antibiotics in penicillin- allergic patients for surgical prophylaxis at a major Canadian teaching hospital. Surgery. 2021;170:783-789. doi:10.1016/j.surg.2021.03.022
- Centers for Disease Control and Prevention. Clinical Features of Penicillin Allergy. August 25, 2025. Accessed January 6, 2026. https://www.cdc.gov/antibiotic-use/hcp/clinical-signs/index.html
- Lam PW, Tarighi P, Elligsen M, et al. Impact of the allergy clarification for cefazolin evidence-based prescribing tool on receipt of preferred perioperative prophylaxis: an interrupted time series study. Clin Infect Dis. 2020;71:2955- 2957. doi:10.1093/cid/ciaa516
- Goodman EJ, Morgan MJ, Johnson Pa, et al. Cephalosporins can be given to penicillin-allergic patients who do not exhibit an anaphylactic response. J Clin Anesth. 2001;13:561-564. doi:10.1016/s0952-8180(01)00329-4
- Lessard S, Huiras C, Dababneh A, et al. Pharmacist adjustment of preoperative antibiotic orders to the preferred preoperative antibiotic cefazolin for patients with penicillin allergy labeling. Am J Health Syst Pharm. 2023;80:532- 536. doi:10.1093/ajhp/zxac385
- Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm. 2013;70:195-283. doi:10.2146/ajhp120568
- Romano A, Valluzzi RL, Caruso C, et al. Tolerability of cefazolin and ceftibuten in patients with IgE-mediated aminopenicillin allergy. J Allergy Clin Immunol Pract. 2020;8:1989-1993.e2. doi:10.1016/j.jaip.2020.02.025
- Grant JM, Song WHC, Shajari S, et al. Safety of administering cefazolin versus other antibiotics in penicillin- allergic patients for surgical prophylaxis at a major Canadian teaching hospital. Surgery. 2021;170:783-789. doi:10.1016/j.surg.2021.03.022
- Centers for Disease Control and Prevention. Clinical Features of Penicillin Allergy. August 25, 2025. Accessed January 6, 2026. https://www.cdc.gov/antibiotic-use/hcp/clinical-signs/index.html
- Lam PW, Tarighi P, Elligsen M, et al. Impact of the allergy clarification for cefazolin evidence-based prescribing tool on receipt of preferred perioperative prophylaxis: an interrupted time series study. Clin Infect Dis. 2020;71:2955- 2957. doi:10.1093/cid/ciaa516
- Goodman EJ, Morgan MJ, Johnson Pa, et al. Cephalosporins can be given to penicillin-allergic patients who do not exhibit an anaphylactic response. J Clin Anesth. 2001;13:561-564. doi:10.1016/s0952-8180(01)00329-4
- Lessard S, Huiras C, Dababneh A, et al. Pharmacist adjustment of preoperative antibiotic orders to the preferred preoperative antibiotic cefazolin for patients with penicillin allergy labeling. Am J Health Syst Pharm. 2023;80:532- 536. doi:10.1093/ajhp/zxac385
Outcomes From the Use of Cefazolin for Surgical Prophylaxis in Patients Allergic to Penicillin
Outcomes From the Use of Cefazolin for Surgical Prophylaxis in Patients Allergic to Penicillin
The Immune Heartache: Pericarditis Following Checkpoint Inhibition
Background
Immune checkpoint inhibitors (ICI) have changed the landscape of cancer therapy. Pembrolizumab is an ICI which targets programmed cell death protein-1 on T-cells and acts to release inhibition of the T-cell antitumor response. Pembrolizumab is approved for the treatment of multiple malignancies. However, ICI therapy may precipitate immune-related adverse events (IRAEs). We describe a unique presentation of irAE-cardiotoxicity.
Case Discussion
A 70-year-old female with a history of uterine cancer previously treated with pembrolizumab (discontinued in January) presented to the emergency department with acute onset nausea and vomiting. On arrival, she was afebrile, tachycardic, normotensive, and saturated well in room air. Labs were notable for troponin of 20, normal TSH, elevated proBNP, ESR and CRP. EKG revealed atrial fibrillation with rapid ventricular response and subtle ST changes in leads II, aVF, V4-V6. She was started on diltiazem infusion for rate control and was subsequently transitioned to oral amiodarone. Given the concern for pericarditis, NSAIDs were initiated. Transthoracic echocardiogram was notable for an ejection fraction of 58% with moderate circumferential pericardial effusion without tamponade. Given her recent ICI exposure and evolving clinical course, she was diagnosed with pembrolizumab- induced pericarditis with associated atrial fibrillation and pericardial effusion. High-dose corticosteroids and colchicine were initiated for stabilization and symptomatic improvement.
Discussion
IRAEs usually occur within 3 months of therapy but may develop later. They are classified as low-grade (1-2), high-grade (3-4), or lethal (5). Anti- PD1 therapy is frequently associated with minor IRAEs, which develop in ~70% of patients; dermatologic IRAEs are most common. Major IRAEs develop in 10-15% of patients, and lethal IRAEs may develop in up to 3%. Cardiac IRAEs are infrequent but significant. Presentation is variable and may involve the myocardium, pericardium, or conductive system. In the case of pericardial disease, high-dose IV methylprednisolone with oral steroid taper should be considered. Re-challenge with ICI therapy should only be considered if clinically stable and pericarditis or myocarditis are excluded.
Conclusions
Our patient illustrates a rare but significant IRAE associated with ICI with improvement following immunosuppressive and rate-control therapy.
Background
Immune checkpoint inhibitors (ICI) have changed the landscape of cancer therapy. Pembrolizumab is an ICI which targets programmed cell death protein-1 on T-cells and acts to release inhibition of the T-cell antitumor response. Pembrolizumab is approved for the treatment of multiple malignancies. However, ICI therapy may precipitate immune-related adverse events (IRAEs). We describe a unique presentation of irAE-cardiotoxicity.
Case Discussion
A 70-year-old female with a history of uterine cancer previously treated with pembrolizumab (discontinued in January) presented to the emergency department with acute onset nausea and vomiting. On arrival, she was afebrile, tachycardic, normotensive, and saturated well in room air. Labs were notable for troponin of 20, normal TSH, elevated proBNP, ESR and CRP. EKG revealed atrial fibrillation with rapid ventricular response and subtle ST changes in leads II, aVF, V4-V6. She was started on diltiazem infusion for rate control and was subsequently transitioned to oral amiodarone. Given the concern for pericarditis, NSAIDs were initiated. Transthoracic echocardiogram was notable for an ejection fraction of 58% with moderate circumferential pericardial effusion without tamponade. Given her recent ICI exposure and evolving clinical course, she was diagnosed with pembrolizumab- induced pericarditis with associated atrial fibrillation and pericardial effusion. High-dose corticosteroids and colchicine were initiated for stabilization and symptomatic improvement.
Discussion
IRAEs usually occur within 3 months of therapy but may develop later. They are classified as low-grade (1-2), high-grade (3-4), or lethal (5). Anti- PD1 therapy is frequently associated with minor IRAEs, which develop in ~70% of patients; dermatologic IRAEs are most common. Major IRAEs develop in 10-15% of patients, and lethal IRAEs may develop in up to 3%. Cardiac IRAEs are infrequent but significant. Presentation is variable and may involve the myocardium, pericardium, or conductive system. In the case of pericardial disease, high-dose IV methylprednisolone with oral steroid taper should be considered. Re-challenge with ICI therapy should only be considered if clinically stable and pericarditis or myocarditis are excluded.
Conclusions
Our patient illustrates a rare but significant IRAE associated with ICI with improvement following immunosuppressive and rate-control therapy.
Background
Immune checkpoint inhibitors (ICI) have changed the landscape of cancer therapy. Pembrolizumab is an ICI which targets programmed cell death protein-1 on T-cells and acts to release inhibition of the T-cell antitumor response. Pembrolizumab is approved for the treatment of multiple malignancies. However, ICI therapy may precipitate immune-related adverse events (IRAEs). We describe a unique presentation of irAE-cardiotoxicity.
Case Discussion
A 70-year-old female with a history of uterine cancer previously treated with pembrolizumab (discontinued in January) presented to the emergency department with acute onset nausea and vomiting. On arrival, she was afebrile, tachycardic, normotensive, and saturated well in room air. Labs were notable for troponin of 20, normal TSH, elevated proBNP, ESR and CRP. EKG revealed atrial fibrillation with rapid ventricular response and subtle ST changes in leads II, aVF, V4-V6. She was started on diltiazem infusion for rate control and was subsequently transitioned to oral amiodarone. Given the concern for pericarditis, NSAIDs were initiated. Transthoracic echocardiogram was notable for an ejection fraction of 58% with moderate circumferential pericardial effusion without tamponade. Given her recent ICI exposure and evolving clinical course, she was diagnosed with pembrolizumab- induced pericarditis with associated atrial fibrillation and pericardial effusion. High-dose corticosteroids and colchicine were initiated for stabilization and symptomatic improvement.
Discussion
IRAEs usually occur within 3 months of therapy but may develop later. They are classified as low-grade (1-2), high-grade (3-4), or lethal (5). Anti- PD1 therapy is frequently associated with minor IRAEs, which develop in ~70% of patients; dermatologic IRAEs are most common. Major IRAEs develop in 10-15% of patients, and lethal IRAEs may develop in up to 3%. Cardiac IRAEs are infrequent but significant. Presentation is variable and may involve the myocardium, pericardium, or conductive system. In the case of pericardial disease, high-dose IV methylprednisolone with oral steroid taper should be considered. Re-challenge with ICI therapy should only be considered if clinically stable and pericarditis or myocarditis are excluded.
Conclusions
Our patient illustrates a rare but significant IRAE associated with ICI with improvement following immunosuppressive and rate-control therapy.
Brief Immunotherapy Yields Major Survival Benefits in Advanced NSCLC: A Case Report
Background
Lung cancer, primarily non-small cell lung cancer (NSCLC), typically presents at an advanced stage with a five-year survival rate below 5%. Treatment includes platinum-based chemotherapy and targeted therapies for specific mutations, with immunotherapy significantly improving outcomes for patients with high PD-L1 expression.
Case Presentation
A 72-year-old male, diagnosed with advanced lung adenocarcinoma in 2020 after showing symptoms of brain metastases, underwent successful surgical and CyberKnife treatments. Despite no actionable genetic targets and a high PD-L1 expression of 80%, his treatment with 3-cycles of Keytruda was cut short due to a psoriatic arthritis flare-up, though it initially decreased his CEA levels significantly. Over the following years, fluctuating CEA levels and various imaging studies indicated some concerning changes, such as potential radionecrosis or recurrence of cancer in the lung. His refusal of biopsy and a preference for avoiding invasive treatments led to only surveillance. Later, an MRI showed some metastasis, and the patient agreed to a lung biopsy, which showed poorly differentiated carcinoma of pulmonary origin. The patient only agreed to restart treatment with Keytruda 4-years later after his initial treatment with Keytruda, under close rheumatological care, and received only two doses. Afterward, the patient lost follow-ups. 3-months later, Repeated CT scans of the chest, abdomen, and pelvis showed no evidence of mass or pathological lymph nodes, and repeated CEA was 3.4.
Discussion
Managing advanced lung adenocarcinoma, especially with complications like brain metastases and psoriatic arthritis, is challenging. Pembrolizumab treatment showed promise by significantly reducing CEA levels despite early discontinuation due to autoimmune side effects, indicating effective tumor response in patients with high PD-L1 expression. The case underscores the need for balancing cancer treatment with autoimmune management and highlights the importance of patient preferences in treatment plans. Ongoing surveillance and genomic profiling remain crucial for guiding therapy.
Conclusions
This case of a 70-year-old male with advanced lung adenocarcinoma highlights the significant impact of immunotherapy, particularly PD-1/ PD-L1 inhibitors like pembrolizumab, in NSCLC. Despite a brief treatment period, the patient experienced extended disease control, demonstrating the potential of immunotherapy to enhance survival and its broad applicability in oncology.
Background
Lung cancer, primarily non-small cell lung cancer (NSCLC), typically presents at an advanced stage with a five-year survival rate below 5%. Treatment includes platinum-based chemotherapy and targeted therapies for specific mutations, with immunotherapy significantly improving outcomes for patients with high PD-L1 expression.
Case Presentation
A 72-year-old male, diagnosed with advanced lung adenocarcinoma in 2020 after showing symptoms of brain metastases, underwent successful surgical and CyberKnife treatments. Despite no actionable genetic targets and a high PD-L1 expression of 80%, his treatment with 3-cycles of Keytruda was cut short due to a psoriatic arthritis flare-up, though it initially decreased his CEA levels significantly. Over the following years, fluctuating CEA levels and various imaging studies indicated some concerning changes, such as potential radionecrosis or recurrence of cancer in the lung. His refusal of biopsy and a preference for avoiding invasive treatments led to only surveillance. Later, an MRI showed some metastasis, and the patient agreed to a lung biopsy, which showed poorly differentiated carcinoma of pulmonary origin. The patient only agreed to restart treatment with Keytruda 4-years later after his initial treatment with Keytruda, under close rheumatological care, and received only two doses. Afterward, the patient lost follow-ups. 3-months later, Repeated CT scans of the chest, abdomen, and pelvis showed no evidence of mass or pathological lymph nodes, and repeated CEA was 3.4.
Discussion
Managing advanced lung adenocarcinoma, especially with complications like brain metastases and psoriatic arthritis, is challenging. Pembrolizumab treatment showed promise by significantly reducing CEA levels despite early discontinuation due to autoimmune side effects, indicating effective tumor response in patients with high PD-L1 expression. The case underscores the need for balancing cancer treatment with autoimmune management and highlights the importance of patient preferences in treatment plans. Ongoing surveillance and genomic profiling remain crucial for guiding therapy.
Conclusions
This case of a 70-year-old male with advanced lung adenocarcinoma highlights the significant impact of immunotherapy, particularly PD-1/ PD-L1 inhibitors like pembrolizumab, in NSCLC. Despite a brief treatment period, the patient experienced extended disease control, demonstrating the potential of immunotherapy to enhance survival and its broad applicability in oncology.
Background
Lung cancer, primarily non-small cell lung cancer (NSCLC), typically presents at an advanced stage with a five-year survival rate below 5%. Treatment includes platinum-based chemotherapy and targeted therapies for specific mutations, with immunotherapy significantly improving outcomes for patients with high PD-L1 expression.
Case Presentation
A 72-year-old male, diagnosed with advanced lung adenocarcinoma in 2020 after showing symptoms of brain metastases, underwent successful surgical and CyberKnife treatments. Despite no actionable genetic targets and a high PD-L1 expression of 80%, his treatment with 3-cycles of Keytruda was cut short due to a psoriatic arthritis flare-up, though it initially decreased his CEA levels significantly. Over the following years, fluctuating CEA levels and various imaging studies indicated some concerning changes, such as potential radionecrosis or recurrence of cancer in the lung. His refusal of biopsy and a preference for avoiding invasive treatments led to only surveillance. Later, an MRI showed some metastasis, and the patient agreed to a lung biopsy, which showed poorly differentiated carcinoma of pulmonary origin. The patient only agreed to restart treatment with Keytruda 4-years later after his initial treatment with Keytruda, under close rheumatological care, and received only two doses. Afterward, the patient lost follow-ups. 3-months later, Repeated CT scans of the chest, abdomen, and pelvis showed no evidence of mass or pathological lymph nodes, and repeated CEA was 3.4.
Discussion
Managing advanced lung adenocarcinoma, especially with complications like brain metastases and psoriatic arthritis, is challenging. Pembrolizumab treatment showed promise by significantly reducing CEA levels despite early discontinuation due to autoimmune side effects, indicating effective tumor response in patients with high PD-L1 expression. The case underscores the need for balancing cancer treatment with autoimmune management and highlights the importance of patient preferences in treatment plans. Ongoing surveillance and genomic profiling remain crucial for guiding therapy.
Conclusions
This case of a 70-year-old male with advanced lung adenocarcinoma highlights the significant impact of immunotherapy, particularly PD-1/ PD-L1 inhibitors like pembrolizumab, in NSCLC. Despite a brief treatment period, the patient experienced extended disease control, demonstrating the potential of immunotherapy to enhance survival and its broad applicability in oncology.
VA Ann Arbor Immunotherapy Stewardship Program
Purpose
To compare vial utilization and spending between fixed and weight-based dosing of pembrolizumab in Veterans. Promote and assess pembrolizumab extended interval dosing.
Background
FDA approved pembrolizumab label change from weight-based to fixed dosing without evidence of fixed-dosing’s superiority. Retrospective studies demonstrate equivalent outcomes for 2 mg/kg every 3 weeks (Q3W), 200 mg Q3W, 4 mg/kg every 6 weeks (Q6W), and 400 mg Q6W.
Methods
In July 2024 VAAAHS (VA Ann Arbor Healthcare System) initiated an immunotherapy stewardship quality improvement program to deprescribe unnecessary pembrolizumab units and promote extended-interval dosing. Specific interventions included order template modification and targeted outreach to key stakeholders.
Data Analysis
All pembrolizumab doses administered at VAAAHS between July 1, 2024 (launch) and March 31, 2025 (data cutoff) were extracted from EHR. Drug utilization, spending, and healthcare contact hours averted were compared to a fixed-dosing counterfactual.
Results
Sixty-three Veterans received 286 total pembrolizumab doses, of which 107 (37.4%) were Q6W and 179 (62.6%) were Q3W. In total, 741 vials were utilized, against expectation of 786 (5.7% reduction), reflecting approximately $182,000 in savings (annualized, $243,000) and 86.5% of the theoretical maximum savings were captured. Q6W’s share of all doses rose from 27.3% in July 2024 to 53.8% in March 2025. Amongst monotherapy, Q6W’s share rose from 60.0% in July 2024 to 86.7% in March 2025. Q6W adoption saved 381 Veteran-healthcare contact hours, not including travel time.
Conclusions
Stewardship efforts reduced unnecessary pembrolizumab utilization and spending while saving Veterans and VAAAHS providers’ time. Continued provider reinforcement, preparation for Oracle/ Cerner implementation, VISN expansion, refinement of pembrolizumab dose-banding, and development of dose bands for other immunotherapies are underway.
Significance
National implementation would improve Veteran convenience and quality of life, enable reductions in drug and resource costs, and enhance clinic throughput.
Purpose
To compare vial utilization and spending between fixed and weight-based dosing of pembrolizumab in Veterans. Promote and assess pembrolizumab extended interval dosing.
Background
FDA approved pembrolizumab label change from weight-based to fixed dosing without evidence of fixed-dosing’s superiority. Retrospective studies demonstrate equivalent outcomes for 2 mg/kg every 3 weeks (Q3W), 200 mg Q3W, 4 mg/kg every 6 weeks (Q6W), and 400 mg Q6W.
Methods
In July 2024 VAAAHS (VA Ann Arbor Healthcare System) initiated an immunotherapy stewardship quality improvement program to deprescribe unnecessary pembrolizumab units and promote extended-interval dosing. Specific interventions included order template modification and targeted outreach to key stakeholders.
Data Analysis
All pembrolizumab doses administered at VAAAHS between July 1, 2024 (launch) and March 31, 2025 (data cutoff) were extracted from EHR. Drug utilization, spending, and healthcare contact hours averted were compared to a fixed-dosing counterfactual.
Results
Sixty-three Veterans received 286 total pembrolizumab doses, of which 107 (37.4%) were Q6W and 179 (62.6%) were Q3W. In total, 741 vials were utilized, against expectation of 786 (5.7% reduction), reflecting approximately $182,000 in savings (annualized, $243,000) and 86.5% of the theoretical maximum savings were captured. Q6W’s share of all doses rose from 27.3% in July 2024 to 53.8% in March 2025. Amongst monotherapy, Q6W’s share rose from 60.0% in July 2024 to 86.7% in March 2025. Q6W adoption saved 381 Veteran-healthcare contact hours, not including travel time.
Conclusions
Stewardship efforts reduced unnecessary pembrolizumab utilization and spending while saving Veterans and VAAAHS providers’ time. Continued provider reinforcement, preparation for Oracle/ Cerner implementation, VISN expansion, refinement of pembrolizumab dose-banding, and development of dose bands for other immunotherapies are underway.
Significance
National implementation would improve Veteran convenience and quality of life, enable reductions in drug and resource costs, and enhance clinic throughput.
Purpose
To compare vial utilization and spending between fixed and weight-based dosing of pembrolizumab in Veterans. Promote and assess pembrolizumab extended interval dosing.
Background
FDA approved pembrolizumab label change from weight-based to fixed dosing without evidence of fixed-dosing’s superiority. Retrospective studies demonstrate equivalent outcomes for 2 mg/kg every 3 weeks (Q3W), 200 mg Q3W, 4 mg/kg every 6 weeks (Q6W), and 400 mg Q6W.
Methods
In July 2024 VAAAHS (VA Ann Arbor Healthcare System) initiated an immunotherapy stewardship quality improvement program to deprescribe unnecessary pembrolizumab units and promote extended-interval dosing. Specific interventions included order template modification and targeted outreach to key stakeholders.
Data Analysis
All pembrolizumab doses administered at VAAAHS between July 1, 2024 (launch) and March 31, 2025 (data cutoff) were extracted from EHR. Drug utilization, spending, and healthcare contact hours averted were compared to a fixed-dosing counterfactual.
Results
Sixty-three Veterans received 286 total pembrolizumab doses, of which 107 (37.4%) were Q6W and 179 (62.6%) were Q3W. In total, 741 vials were utilized, against expectation of 786 (5.7% reduction), reflecting approximately $182,000 in savings (annualized, $243,000) and 86.5% of the theoretical maximum savings were captured. Q6W’s share of all doses rose from 27.3% in July 2024 to 53.8% in March 2025. Amongst monotherapy, Q6W’s share rose from 60.0% in July 2024 to 86.7% in March 2025. Q6W adoption saved 381 Veteran-healthcare contact hours, not including travel time.
Conclusions
Stewardship efforts reduced unnecessary pembrolizumab utilization and spending while saving Veterans and VAAAHS providers’ time. Continued provider reinforcement, preparation for Oracle/ Cerner implementation, VISN expansion, refinement of pembrolizumab dose-banding, and development of dose bands for other immunotherapies are underway.
Significance
National implementation would improve Veteran convenience and quality of life, enable reductions in drug and resource costs, and enhance clinic throughput.
Proactive Penicillin Allergy Delabeling: Lessons Learned From a Quality Improvement Project
Proactive Penicillin Allergy Delabeling: Lessons Learned From a Quality Improvement Project
Penicillin allergy is common in the United States. About 9.0% to 13.8% of patients have a diagnosed penicillin allergy documented in their electronic health record. The annual incidence rates is 1.1% in males and 1.4% in females.1,2
Penicillin hypersensitivity likely wanes over time. A 1981 study found that 93% of patients who experienced an allergic reaction to penicillin had a positive skin test 7 to 12 months postreaction, but only 22% still had a positive test after 10 years.3 Confirmed type 1 hypersensitivity penicillin allergies, as demonstrated by positive skin prick testing, also are decreasing over time.4 Furthermore, many patients’ reactions may have been misdiagnosed as a penicillin allergy. Upon actual confirmatory testing of penicillin allergy, only 8.5% to 13.8% of patients believed to have a penicillin allergy were positive on skin prick testing of penicillin products.5,6 A 2024 US study found that 11% of individuals with a history of a penicillin reaction tested positive on skin testing.7
The positive predictive value of penicillin allergy skin testing is poorly defined due to the ethical dilemma of orally challenging a patient who demonstrates skin test reactivity. Due to its high negative predictive value (NPV), skin prick combined with intradermal testing has been the gold-standard test in cases of clinical concern.6 Patients with positive skin testing are assumed to be truly positive, and therefore penicillin allergic, even though false-positive results to penicillin skin testing are known to occur.8
Misdiagnosis of penicillin allergy carries substantial clinical and economic consequences. A 2011 study suggested a statistically significant 1.8% increased absolute risk of mortality and 5.5% increased absolute risk of intensive care unit admission for those labeled with penicillin allergy and admitted for an infection.9 Another study found a 14% increase in mortality associated with the diagnosis of penicillin allergy.10 In a 2014 case-control study, penicillin allergy also was associated with a 23.4% greater risk of Clostridioides difficile, 14.1% more methicillin-resistant Staphylococcus aureus, and 30.1% more vancomycin-resistant enterococci infections.11Direct cost savings during an inpatient admission for infection were as much as $609 per patient with additional indirect cost savings of up to $4254 per admission.12 When viewed from the perspective of a health care system, these costs quickly accumulate, negatively impacting the fiscal stability of our patients and placing additional financial strain on an over-burdened system.
If 10% of US patients have penicillin allergy labels, then about 33 million patients might be eligible for delabeling. There are only 6309 board-certified allergists actively practicing in the US, which could amount to about 5231 potential penicillin challenges per allergist, not even including the 3.3 million new patients per year (assuming a 1% incidence).13 Clarifying each patient’s tolerance of penicillin products will clearly require nonallergist cooperation.
The 2022 drug allergy practice parameter update recommends several consensus-based statements (CBSs) to directly address penicillin allergy.14 This guideline recommends proactive efforts to delabel patients with a reported penicillin allergy (CBS 4); advise against testing in cases where the history is inconsistent with a true allergic reaction, though a challenge may be offered (CBS 5); skin testing for those with a history of anaphylaxis or a recent reaction (CBS 6); advise against multiple-day penicillin challenges (CBS 7); advise against skin testing for pediatric patients with benign cutaneous reactions (CBS 8); and recommends direct oral challenge for adults with distant or benign cutaneous reactions (CBS 9). These recommendations create a potentially high demand for delabeling with allergy specialists. One potential solution is to perform direct oral challenges in primary care, emergency departments, and urgent care clinics.
Evidence supporting the safety of direct oral penicillin challenges in low-risk patients was initially noted in the allergy community, but now evidence for their use in primary care clinics is growing—including in children.15 In a military-specific population, an amoxicillin challenge of Marine recruits with suspected penicillin allergy revealed that only 1.5% of those challenged acutely reacted and should be considered allergic to penicillin.16 Historically, in order to refute the diagnosis of penicillin allergy, an allergist would order penicillin skin prick testing. If the test was negative, an allergist would proceed to intradermal testing and if negative again (NPV of 97.9%), proceed to a graded oral challenge.6 However, this process is not fully reproducible in most clinics because the minor determinants mixture used in skin testing is not commercially available.17 Additionally, the full skin testing procedure requires specialized training, is more time-consuming, causes more discomfort, lacks US Food and Drug Administration approval for children, and has a higher cost ($220 per test for each patient as of 2016).18 As such, the movement toward direct oral challenges is progressing. Nonetheless, the best method for primary care or emergency department clinicians to determine who the appropriate patients are for this procedure has not been fully established. Risk tools have been created in the past to help delineate low-risk patients who would be appropriate for direct oral amoxicillin challenges, but these were not widely replicated or validated.19 The PEN-FAST standardized risk score was first published in 2020 and has since been validated in different groups with additional safety data. This scoring system ranges from 0 to 5 points, assigning 2 points for a penicillin reaction within the past five (F) years, 2 points for angioedema/anaphylaxis (A) or a severe (S) cutaneous reaction, and 1 point if treatment (T) was required for the reaction. A score < 3 is considered low-risk and safe for direct oral challenge, although most of the safety data are in patients with a score of 0 or 1.20 The PEN-FAST guided direct oral challenge with an NPV of 96.3% has now been prospectively shown to be noninferior to standard skin prick test/intradermal test/graded challenge for low-risk patients with a PEN-FAST score < 3.21 The PEN-FAST validating study was conducted predominantly with an Australian population of adult White women, but now it also has been validated in children aged > 12 years, as well as in European and North American cohorts.22-24
Air Force Delabeling Program
This article describes a method for proactively, safely, and efficiently delabeling penicillin allergic patients at an Air Force clinic. This quality improvement (QI) report provides a successful model for penicillin allergy delabeling, illustrates lessons learned, and suggests next steps toward improving patient options for an invaluable antibiotic class.
The first step was to proactively delabel penicillin allergy from a population of active duty service members and their dependents. Electronic health record (EHR) allergy search functions are a helpful tool in finding patients with allergy labels. The Kadena Medical Clinic, in Okinawa, Japan, uses the Military Health System GENESIS EHR, which includes a discern reporting portal with a patient allergy search that creates a patient-specific medication allergy report. To compile the most complete database of patients with a penicillin allergy, all 15 potential allergy search options for “penicillin” were selected, as were 4 relevant options for amoxicillin (including options with clavulanate). Including so many options for specific penicillin medication allergies helps add specificity to the diagnosis in the EHR but can make aggregation of data more difficult. The
The complete compiled list was manually reviewed for high-risk patients with severe cutaneous adverse reactions (SCARs) of any age. Patients with pregnancy, unsuitable medical histories (ie, severe asthma), or taking β-blockers were excluded. Patients remaining on the list were contacted by telephone and offered appointments during a single week that was dedicated to penicillin allergy delabeling. Allergists in the Air Force are assigned to a region where they offer allergy services at clinics without a regular allergist. The allergist for the region traveled to the QI site for a 1-week campaign at an estimated cost of $4600. When the patients were contacted, they were briefly informed of the goal of the penicillin delabeling campaign, and if interested, they were scheduled for 1 of 50 available appointments that week. Patients were contacted with enough lead time to stop oral antihistamines (OAH) for ≥ 7 days before the appointment.
Patients were given an intake questionnaire and interviewed about their penicillin allergy history. This questionnaire inquired about the nature of the allergy, mental and physical health impacts of the allergy label, PEN-FAST scoring questions, and posttest attitude toward delabeling, if applicable. Patients with a PEN-FAST score < 3 were offered direct, graded oral challenge or the standard skin prick, followed by intradermal, followed by graded oral challenge protocol. Patients with PEN-FAST scores of ≥ 3 were offered skin testing prior to oral challenge protocol. Patients could decline further testing. If patients wished to proceed, they were asked to complete a written informed consent document.
Oral challenges followed a 10%/90% protocol, beginning with 50 mg of liquid amoxicillin followed by 450 mg after 15 minutes, as long as the patient remained asymptomatic. Challenge forms are available in the eAppendix . After receiving the 450-mg amoxicillin dose, the patient remained in the clinic for 60 minutes before a final clinical evaluation. If the patient remained asymptomatic after this period, the penicillin or amoxicillin allergy was marked as resolved in the EHR. The patients were given contact information for the clinic for follow-up if a delayed reaction was noted and they wished the medication allergy to be re-entered. An EHR encounter note was created for each patient detailing the allergy testing and delabeling.
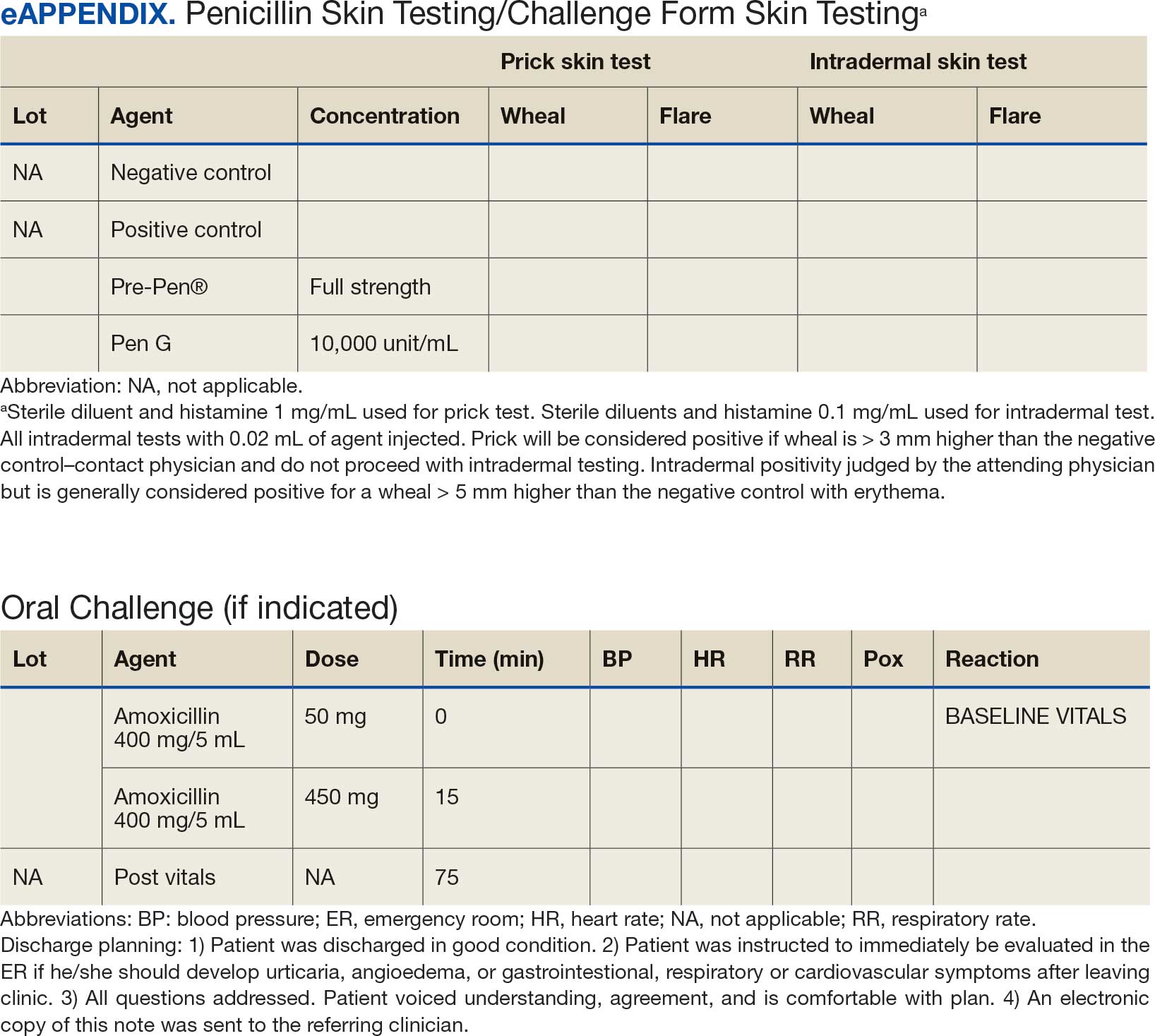
This campaign was conducted at a basic life support-only facility by a single clinician without medical technician support. An allergic reaction medication kit was available and contained OAHs, intramuscular antihistamines, intramuscular epinephrine, intramuscular corticosteroids, and short-acting β-agonists for nebulization. The facility also had an urgent care room (staffed by primary care practitioners [PCPs]) that could help establish intravenous access and administer fluids if necessary and had previously established plans for emergency patient transport to a higher level of care, if necessary.
Program Outcomes
A list of 65 patients that included both active-duty service members and dependents with penicillin or amoxicillin allergy was created. This list was reviewed by an allergist to identify high-risk individuals, which required about 90 minutes. Two patients (3%) were excluded; 1 had a history of SCAR to penicillin and 1 had a complex medical history requiring continued OAH use. Sixty-three patients were contacted via telephone, and 29 patients (46%) scheduled an appointment. One patient (2%) was identified as penicillin-tolerant during the booking process, and the penicillin allergy was removed without testing (Figure 1).
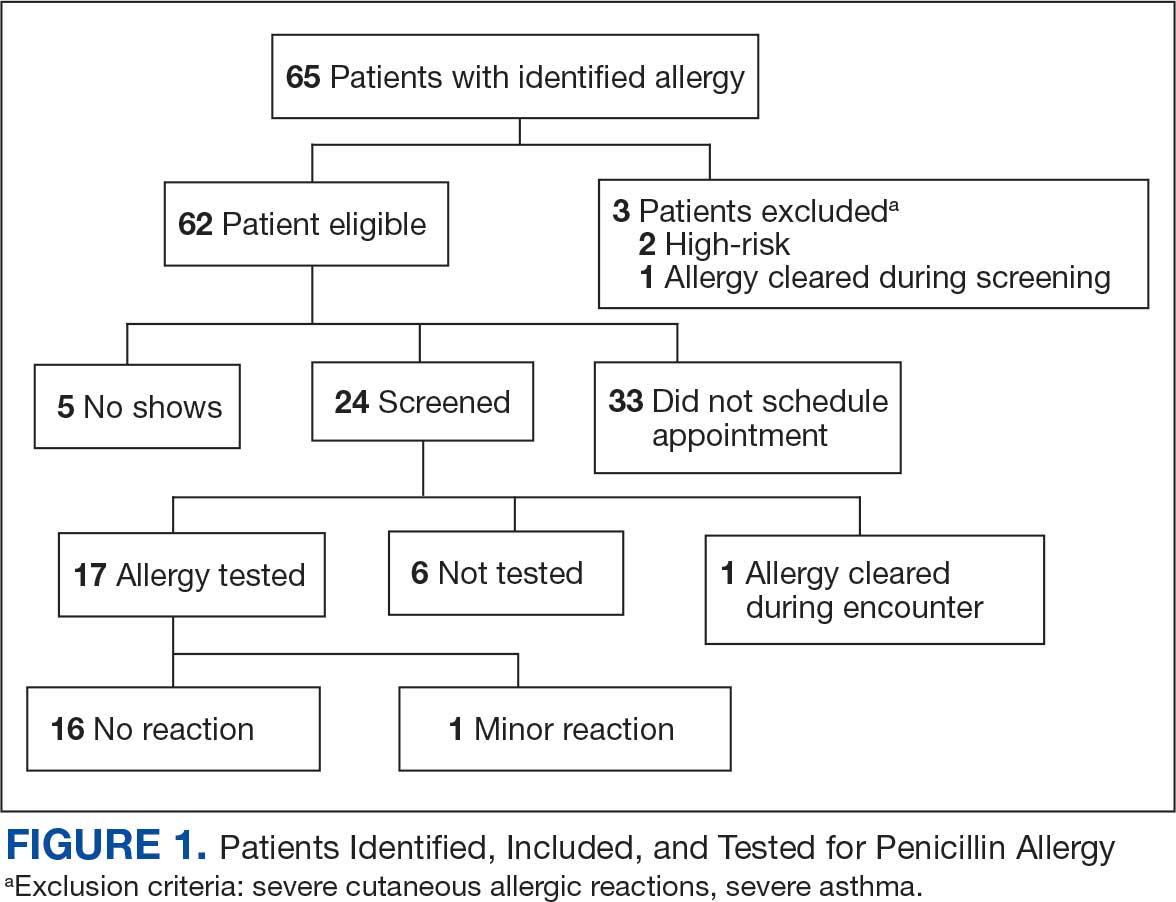
Of the 29 scheduled patients, 5 patients (17%) failed to present for care. Of the potential appointments set aside for the program, only 42% were used. One patient (4%) who was seen in clinic was delabeled based on history alone as they had previously successfully tolerated a course of amoxicillin. Four patients (17%) declined further testing with a PEN-FAST score > 2 due to a clear history of acute immunoglobulin (Ig) E-mediated reaction to a penicillin product within the past year. One patient (4%) was unable to be tested due to ongoing OAH use and 1 patient (4%) declined further penicillin testing after the discussion about risks, benefits, and alternatives to the procedures offered.
Of the 24 patients who arrived for a clinic appointment, 17 (71%) underwent penicillin allergy delabeling testing: 14 (82%) underwent direct challenge, and 3 (18%) underwent the skin testing before oral amoxicillin challenge procedure. Of the 17 who were tested, 16 (94%) tolerated a total dose of 500 mg of oral amoxicillin within the 1-hour observation period. One tested patient (6%) in the direct oral challenge group experienced an adverse reaction that was described as dull headache and hand tremor after the 50-mg dose; although it self-resolved within 15 minutes, this prompted the patient to discontinue the challenge. This adverse reaction was determined to be very unlikely IgE-mediated. None of the 3 patients who underwent the skin testing before oral challenge protocol experienced an adverse drug reaction (ADR). None of the 17 patients who received any oral amoxicillin required follow-up or reported a delayed cutaneous ADR to the challenge. No OAHs or epinephrine were used for any of the challenges.
Data collected from patient questionnaires displayed perceived health impacts of a penicillin allergy on the patient population. Patients reported a variety of ADRs to previous administration of penicillin products: 17 (71%) reported urticaria, 2 (8%) reported anaphylaxis, and 3 (13%) were unable to recall the reaction (Figure 2). Nine patients (38%) felt their initial reaction was distressing. Fifteen patients (88%) felt relief following negative testing (Table).
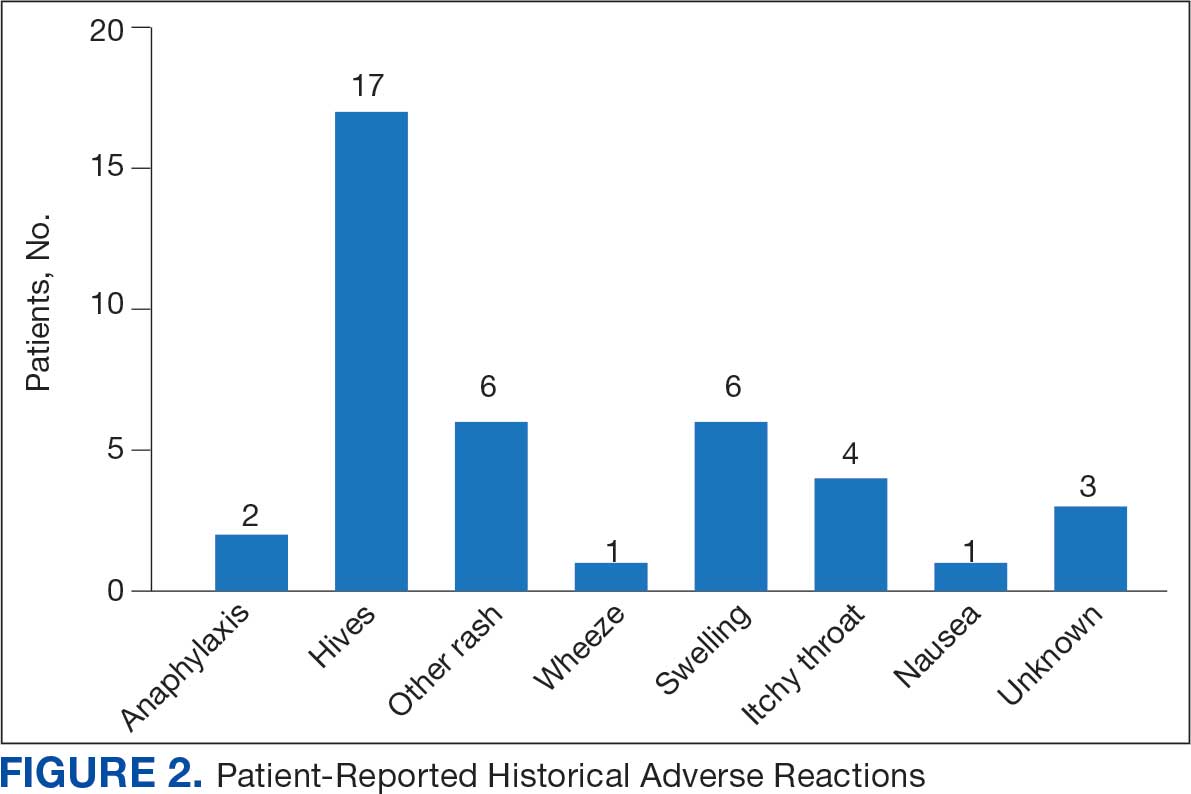

Discussion
To our knowledge, this was the first documented proactive penicillin delabeling QI project in a military clinic treating both active-duty service members and their dependents, modeled on the 2022 drug allergy guidelines.14 Several interesting lessons were learned that may improve future similar QI projects. Only 46% of patients identified as having penicillin allergy presented for evaluation, leaving 42% of available appointments unused. Without prior data on anticipated participation rates, these data provide a crude benchmark for utilization rates, which can inform future resource planning. While attempts were made to contact each patient, additional efforts to publicize the penicillin allergy delabeling campaign would have been useful to improve efficiency.
In addition, when patients with a PEN-FAST score of < 3 were educated about the risks and benefits of each procedure and offered the direct oral graded challenge and skin testing prior to oral challenge, 82% preferred the direct challenge. None of the patients who experienced a penicillin ADR in the past year wished to undergo skin testing or oral challenge, though each was educated on penicillin allergy and the possibility of testing in the future, making each encounter beneficial. Of the 17 patients tested, 16 (94%) tolerated oral amoxicillin and 1 (6%) experienced a mild, self-resolving ADR that was very unlikely of an IgE-mediated origin. Additionally, while plans and preparations for ADRs to the challenges were available, none were required. Patient questionnaires demonstrated the heterogeneity of previous ADRs and their attitude toward their allergy diagnosis. The positive impact of delabeling on patient well-being noted by 88% of patients reinforced the benefit of the effort.
This project was limited by a relatively small sample size, which may not have been large enough to detect ADRs, especially IgE-mediated allergic reactions. Herein lies the importance of having clinicians equipped to treat allergic ADRs to conduct penicillin challenges in the primary care setting. It is prudent to ensure not only proper training of physicians performing these challenges, but also appropriate equipment, medication, and response personnel. Medications that are useful include epinephrine, OAHs, albuterol, steroids, and intravenous fluids.
Having a response area and plan are essential to ensure appropriate care in the rare instance of allergic ADRs progressing to anaphylaxis. In rare cases, emergency medical services may be required and having a plan with appropriate response and transport time is essential to patient safety. This may not be practical in more rural or smaller practices. In those scenarios, it may be helpful to partner with a larger practice to send patients for delabeling or to use clinical space in closer proximity to emergency services. Perhaps an ideal setting might be urgent or emergent care centers due to high acuity resources and frequent prescription of amoxicillin antibiotics; however, this may be complicated by concurrent infections raising the incidence of delayed benign eruptions with amoxicillin ingestion and complicating the patient’s allergy records. Further training of urgent and emergent care practitioners would be helpful for proper patient education regarding antibiotic-associated reactions.
Full testing integration into other primary care clinics may be limited due to the specialized training required for complete skin testing. Nevertheless, as shown in this project, most patients may be delabeled based on a PEN-FAST evaluation followed by oral challenge alone. Incorporation in other QI projects could involve continuing medical education to train staff physicians on PEN-FAST, teaching primary care residents during training, and site visits by allergists to train local physicians on testing. This project involved training 2 PCPs to conduct skin and oral challenge testing using PEN-FAST to guide clinical decision-making with an allergist available for consultation if needed. Future projects might model a similar approach or perhaps focus on training more physicians on oral challenges alone to reach a high percentage of the target population.
Conclusions
This project demonstrates a safe, efficient, and cost-effective model for penicillin allergy delabeling in clinics without regular access to allergy services. The use of PEN-FAST allows a quick and simple method to screen patients with penicillin allergy to meet the goals of the 2022 CBSs, but data are still accumulating to validate this method of screening across populations. This project demonstrates additional support for the use of PEN-FAST, while illustrating appropriate education regarding oral testing technique and its limitations.
Using an EHR report limited the patients in the testing pool and subsequent sample size. This suggests that a primary care identification-driven enrollment in testing may offer even more benefit both in allergy detection and education of testing benefits. Oral challenges are more cost effective, shorter in duration, and have fewer training requirements when compared with antecedent skin testing, making them an ideal option for PCPs in a clinic setting. Trained PCPs may opt to offer periodic appointments for delabeling, or offer days dedicated to delabeling as many patients as possible. Penicillin delabeling is an urgent and expansive charge; this study offers a replicable model for executing this important task.
- Macy E, Poon KYT. Self-reported antibiotic allergy incidence and prevalence: age and sex effects. Am J Med. 2009;122(8):778.e1-778.e7787. doi:10.1016/j.amjmed.2009.01.034
- Zhou L, Dhopeshwarkar N, Blumenthal KG, et al. Drug allergies documented in electronic health records of a large healthcare system. Allergy. 2016;71(9):1305-1313. doi:10.1111/all.12881
- Sullivan TJ, Wedner HJ, Shatz GS, Yecies LD, Parker CW. Skin testing to detect penicillin allergy. J Allergy Clin Immunol. 1981;68(3):171-180. doi:10.1016/0091-6749(81)90180-9
- Macy E, Schatz M, Lin C, Poon KY. The falling rate of positive penicillin skin tests from 1995 to 2007. Perm J. 2009;13(2):12-18. doi:10.7812/TPP/08-073
- Fox SJ, Park MA. Penicillin skin testing is a safe and effective tool for evaluating penicillin allergy in the pediatric population. J Allergy Clin Immunol Pract. 2014;2(4):439-444. doi:10.1016/j.jaip.2014.04.013
- Solensky R, Jacobs J, Lester M, et al. Penicillin Allergy Evaluation: A Prospective, Multicenter, Open-Label Evaluation of a Comprehensive Penicillin Skin Test Kit. J Allergy Clin Immunol Pract. 2019;7(6):1876-1885.e3. doi:10.1016/j.jaip.2019.02.040 7.
- Gonzalez-Estrada A, Park MA, Accarino JJO, et al. Predicting penicillin allergy: A United States multicenter retrospective study. J Allergy Clin Immunol Pract. 2024;12(5):1181-1191.e10. doi:10.1016/j.jaip.2024.01.010
- Stüwe HT, Geissler W, Paap A, Cromwell O. The presence of latex can induce false-positive skin tests in subjects tested with penicillin determinants. Allergy. 1997;52(12):1243. doi:10.1111/j.1398-9995.1997.tb00975.x
- Charneski L, Deshpande G, Smith SW. Impact of an antimicrobial allergy label in the medical record on clinical outcomes in hospitalized patients. Pharmacotherapy. 2011;31(8):742-747. doi:10.1592/phco.31.8.742
- Blumenthal KG, Lu N, Zhang Y, Walensky RP, Choi HK. Recorded penicillin allergy and risk of mortality: a population-based matched cohort study. J Gen Intern Med. 2019;34(9):1685-1687. doi:10.1007/s11606-019-04991-y
- Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: A cohort study. J Allergy Clin Immunol. 2014;133(3):790-796. doi:10.1016/j.jaci.2013.09.021
- Mattingly TJ II, Fulton A, Lumish RA, et al. The cost of self-reported penicillin allergy: a systematic review. J Allergy Clin Immunol Pract. 2018;6(5):1649-1654.e4. doi:10.1016/j.jaip.2017.12.033
- Diplomate Statistics. American Board of Allergy and Immunology website. Published February, 18 2021. Accessed July 28, 2025. https://www.abai.org/statistics_diplomates.asp
- Khan DA, Banerji A, Blumenthal KG, et al. Drug allergy: a 2022 practice parameter update. J Allergy Clin Immunol. 2022;150(6):1333-1393. doi:10.1016/j.jaci.2022.08.028
- Mill C, Primeau MN, Medoff E, et al. Assessing the diagnostic properties of a graded oral provocation challenge for the diagnosis of immediate and nonimmediate reactions to amoxicillin in children. JAMA Pediatr. 2016;170:e160033. doi:10.1001/jamapediatrics.2016.0033
- Tucker MH, Lomas CM, Ramchandar N, Waldram JD. Amoxicillin challenge without penicillin skin testing in evaluation of penicillin allergy in a cohort of Marine recruits. J Allergy Clin Immunol Pract. 2017;5(3):813-815. doi:10.1016/j.jaip.2017.01.023
- Shenoy ES, Macy E, Rowe T, Blumenthal KG. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321(2):188–99. doi:10.1001/jama.2018.19283
- Blumenthal KG, Li Y, Banerji A, et al. The cost of penicillin allergy evaluation. J Allergy Clin Immunol Pract. 2018;6(3):1019-1027.e2. doi:10.1016/j.jaip.2017.08.006
- Banks TA, Tucker M, Macy E. Evaluating penicillin allergies without skin testing. Curr Allergy Asthma Rep. 2019;19(5):27. doi:10.1007/s11882-019-0854-6
- Trubiano JA, Vogrin S, Chua KYL, et al. Development and validation of a penicillin allergy clinical decision rule. JAMA Intern Med. 2020;180(5):745-752. doi:10.1001/jamainternmed.2020.0403
- Copaescu AM, Vogrin S, James F, et al. Efficacy of a clinical decision rule to enable direct oral challenge in patients with low-risk penicillin allergy: the PALACE randomized clinical trial. JAMA Intern Med. 2023;183(9):944-952. doi:10.1001/jamainternmed.2023.2986
- Copaescu AM, Vogrin S, Shand G, et al. Validation of the PEN-FAST score in a pediatric population. JAMA Netw Open. 2022;5(9):e2233703. doi:10.1001/jamanetworkopen.2022.33703
- Piotin A, Godet J, Trubiano JA, et al. Predictive factors of amoxicillin immediate hypersensitivity and validation of PEN-FAST clinical decision rule. Ann Allergy Asthma Immunol. 2022;128(1):27-32. doi:10.1016/j.anai.2021.07.005
- Su C, Belmont A, Liao J, et al. Evaluating the PEN-FAST clinical decision-making tool to enhance penicillin allergy delabeling. JAMA Intern Med. 2023;183(8):883-885. doi:10.1001/jamainternmed.2023.1572
Penicillin allergy is common in the United States. About 9.0% to 13.8% of patients have a diagnosed penicillin allergy documented in their electronic health record. The annual incidence rates is 1.1% in males and 1.4% in females.1,2
Penicillin hypersensitivity likely wanes over time. A 1981 study found that 93% of patients who experienced an allergic reaction to penicillin had a positive skin test 7 to 12 months postreaction, but only 22% still had a positive test after 10 years.3 Confirmed type 1 hypersensitivity penicillin allergies, as demonstrated by positive skin prick testing, also are decreasing over time.4 Furthermore, many patients’ reactions may have been misdiagnosed as a penicillin allergy. Upon actual confirmatory testing of penicillin allergy, only 8.5% to 13.8% of patients believed to have a penicillin allergy were positive on skin prick testing of penicillin products.5,6 A 2024 US study found that 11% of individuals with a history of a penicillin reaction tested positive on skin testing.7
The positive predictive value of penicillin allergy skin testing is poorly defined due to the ethical dilemma of orally challenging a patient who demonstrates skin test reactivity. Due to its high negative predictive value (NPV), skin prick combined with intradermal testing has been the gold-standard test in cases of clinical concern.6 Patients with positive skin testing are assumed to be truly positive, and therefore penicillin allergic, even though false-positive results to penicillin skin testing are known to occur.8
Misdiagnosis of penicillin allergy carries substantial clinical and economic consequences. A 2011 study suggested a statistically significant 1.8% increased absolute risk of mortality and 5.5% increased absolute risk of intensive care unit admission for those labeled with penicillin allergy and admitted for an infection.9 Another study found a 14% increase in mortality associated with the diagnosis of penicillin allergy.10 In a 2014 case-control study, penicillin allergy also was associated with a 23.4% greater risk of Clostridioides difficile, 14.1% more methicillin-resistant Staphylococcus aureus, and 30.1% more vancomycin-resistant enterococci infections.11Direct cost savings during an inpatient admission for infection were as much as $609 per patient with additional indirect cost savings of up to $4254 per admission.12 When viewed from the perspective of a health care system, these costs quickly accumulate, negatively impacting the fiscal stability of our patients and placing additional financial strain on an over-burdened system.
If 10% of US patients have penicillin allergy labels, then about 33 million patients might be eligible for delabeling. There are only 6309 board-certified allergists actively practicing in the US, which could amount to about 5231 potential penicillin challenges per allergist, not even including the 3.3 million new patients per year (assuming a 1% incidence).13 Clarifying each patient’s tolerance of penicillin products will clearly require nonallergist cooperation.
The 2022 drug allergy practice parameter update recommends several consensus-based statements (CBSs) to directly address penicillin allergy.14 This guideline recommends proactive efforts to delabel patients with a reported penicillin allergy (CBS 4); advise against testing in cases where the history is inconsistent with a true allergic reaction, though a challenge may be offered (CBS 5); skin testing for those with a history of anaphylaxis or a recent reaction (CBS 6); advise against multiple-day penicillin challenges (CBS 7); advise against skin testing for pediatric patients with benign cutaneous reactions (CBS 8); and recommends direct oral challenge for adults with distant or benign cutaneous reactions (CBS 9). These recommendations create a potentially high demand for delabeling with allergy specialists. One potential solution is to perform direct oral challenges in primary care, emergency departments, and urgent care clinics.
Evidence supporting the safety of direct oral penicillin challenges in low-risk patients was initially noted in the allergy community, but now evidence for their use in primary care clinics is growing—including in children.15 In a military-specific population, an amoxicillin challenge of Marine recruits with suspected penicillin allergy revealed that only 1.5% of those challenged acutely reacted and should be considered allergic to penicillin.16 Historically, in order to refute the diagnosis of penicillin allergy, an allergist would order penicillin skin prick testing. If the test was negative, an allergist would proceed to intradermal testing and if negative again (NPV of 97.9%), proceed to a graded oral challenge.6 However, this process is not fully reproducible in most clinics because the minor determinants mixture used in skin testing is not commercially available.17 Additionally, the full skin testing procedure requires specialized training, is more time-consuming, causes more discomfort, lacks US Food and Drug Administration approval for children, and has a higher cost ($220 per test for each patient as of 2016).18 As such, the movement toward direct oral challenges is progressing. Nonetheless, the best method for primary care or emergency department clinicians to determine who the appropriate patients are for this procedure has not been fully established. Risk tools have been created in the past to help delineate low-risk patients who would be appropriate for direct oral amoxicillin challenges, but these were not widely replicated or validated.19 The PEN-FAST standardized risk score was first published in 2020 and has since been validated in different groups with additional safety data. This scoring system ranges from 0 to 5 points, assigning 2 points for a penicillin reaction within the past five (F) years, 2 points for angioedema/anaphylaxis (A) or a severe (S) cutaneous reaction, and 1 point if treatment (T) was required for the reaction. A score < 3 is considered low-risk and safe for direct oral challenge, although most of the safety data are in patients with a score of 0 or 1.20 The PEN-FAST guided direct oral challenge with an NPV of 96.3% has now been prospectively shown to be noninferior to standard skin prick test/intradermal test/graded challenge for low-risk patients with a PEN-FAST score < 3.21 The PEN-FAST validating study was conducted predominantly with an Australian population of adult White women, but now it also has been validated in children aged > 12 years, as well as in European and North American cohorts.22-24
Air Force Delabeling Program
This article describes a method for proactively, safely, and efficiently delabeling penicillin allergic patients at an Air Force clinic. This quality improvement (QI) report provides a successful model for penicillin allergy delabeling, illustrates lessons learned, and suggests next steps toward improving patient options for an invaluable antibiotic class.
The first step was to proactively delabel penicillin allergy from a population of active duty service members and their dependents. Electronic health record (EHR) allergy search functions are a helpful tool in finding patients with allergy labels. The Kadena Medical Clinic, in Okinawa, Japan, uses the Military Health System GENESIS EHR, which includes a discern reporting portal with a patient allergy search that creates a patient-specific medication allergy report. To compile the most complete database of patients with a penicillin allergy, all 15 potential allergy search options for “penicillin” were selected, as were 4 relevant options for amoxicillin (including options with clavulanate). Including so many options for specific penicillin medication allergies helps add specificity to the diagnosis in the EHR but can make aggregation of data more difficult. The
The complete compiled list was manually reviewed for high-risk patients with severe cutaneous adverse reactions (SCARs) of any age. Patients with pregnancy, unsuitable medical histories (ie, severe asthma), or taking β-blockers were excluded. Patients remaining on the list were contacted by telephone and offered appointments during a single week that was dedicated to penicillin allergy delabeling. Allergists in the Air Force are assigned to a region where they offer allergy services at clinics without a regular allergist. The allergist for the region traveled to the QI site for a 1-week campaign at an estimated cost of $4600. When the patients were contacted, they were briefly informed of the goal of the penicillin delabeling campaign, and if interested, they were scheduled for 1 of 50 available appointments that week. Patients were contacted with enough lead time to stop oral antihistamines (OAH) for ≥ 7 days before the appointment.
Patients were given an intake questionnaire and interviewed about their penicillin allergy history. This questionnaire inquired about the nature of the allergy, mental and physical health impacts of the allergy label, PEN-FAST scoring questions, and posttest attitude toward delabeling, if applicable. Patients with a PEN-FAST score < 3 were offered direct, graded oral challenge or the standard skin prick, followed by intradermal, followed by graded oral challenge protocol. Patients with PEN-FAST scores of ≥ 3 were offered skin testing prior to oral challenge protocol. Patients could decline further testing. If patients wished to proceed, they were asked to complete a written informed consent document.
Oral challenges followed a 10%/90% protocol, beginning with 50 mg of liquid amoxicillin followed by 450 mg after 15 minutes, as long as the patient remained asymptomatic. Challenge forms are available in the eAppendix . After receiving the 450-mg amoxicillin dose, the patient remained in the clinic for 60 minutes before a final clinical evaluation. If the patient remained asymptomatic after this period, the penicillin or amoxicillin allergy was marked as resolved in the EHR. The patients were given contact information for the clinic for follow-up if a delayed reaction was noted and they wished the medication allergy to be re-entered. An EHR encounter note was created for each patient detailing the allergy testing and delabeling.

This campaign was conducted at a basic life support-only facility by a single clinician without medical technician support. An allergic reaction medication kit was available and contained OAHs, intramuscular antihistamines, intramuscular epinephrine, intramuscular corticosteroids, and short-acting β-agonists for nebulization. The facility also had an urgent care room (staffed by primary care practitioners [PCPs]) that could help establish intravenous access and administer fluids if necessary and had previously established plans for emergency patient transport to a higher level of care, if necessary.
Program Outcomes
A list of 65 patients that included both active-duty service members and dependents with penicillin or amoxicillin allergy was created. This list was reviewed by an allergist to identify high-risk individuals, which required about 90 minutes. Two patients (3%) were excluded; 1 had a history of SCAR to penicillin and 1 had a complex medical history requiring continued OAH use. Sixty-three patients were contacted via telephone, and 29 patients (46%) scheduled an appointment. One patient (2%) was identified as penicillin-tolerant during the booking process, and the penicillin allergy was removed without testing (Figure 1).

Of the 29 scheduled patients, 5 patients (17%) failed to present for care. Of the potential appointments set aside for the program, only 42% were used. One patient (4%) who was seen in clinic was delabeled based on history alone as they had previously successfully tolerated a course of amoxicillin. Four patients (17%) declined further testing with a PEN-FAST score > 2 due to a clear history of acute immunoglobulin (Ig) E-mediated reaction to a penicillin product within the past year. One patient (4%) was unable to be tested due to ongoing OAH use and 1 patient (4%) declined further penicillin testing after the discussion about risks, benefits, and alternatives to the procedures offered.
Of the 24 patients who arrived for a clinic appointment, 17 (71%) underwent penicillin allergy delabeling testing: 14 (82%) underwent direct challenge, and 3 (18%) underwent the skin testing before oral amoxicillin challenge procedure. Of the 17 who were tested, 16 (94%) tolerated a total dose of 500 mg of oral amoxicillin within the 1-hour observation period. One tested patient (6%) in the direct oral challenge group experienced an adverse reaction that was described as dull headache and hand tremor after the 50-mg dose; although it self-resolved within 15 minutes, this prompted the patient to discontinue the challenge. This adverse reaction was determined to be very unlikely IgE-mediated. None of the 3 patients who underwent the skin testing before oral challenge protocol experienced an adverse drug reaction (ADR). None of the 17 patients who received any oral amoxicillin required follow-up or reported a delayed cutaneous ADR to the challenge. No OAHs or epinephrine were used for any of the challenges.
Data collected from patient questionnaires displayed perceived health impacts of a penicillin allergy on the patient population. Patients reported a variety of ADRs to previous administration of penicillin products: 17 (71%) reported urticaria, 2 (8%) reported anaphylaxis, and 3 (13%) were unable to recall the reaction (Figure 2). Nine patients (38%) felt their initial reaction was distressing. Fifteen patients (88%) felt relief following negative testing (Table).


Discussion
To our knowledge, this was the first documented proactive penicillin delabeling QI project in a military clinic treating both active-duty service members and their dependents, modeled on the 2022 drug allergy guidelines.14 Several interesting lessons were learned that may improve future similar QI projects. Only 46% of patients identified as having penicillin allergy presented for evaluation, leaving 42% of available appointments unused. Without prior data on anticipated participation rates, these data provide a crude benchmark for utilization rates, which can inform future resource planning. While attempts were made to contact each patient, additional efforts to publicize the penicillin allergy delabeling campaign would have been useful to improve efficiency.
In addition, when patients with a PEN-FAST score of < 3 were educated about the risks and benefits of each procedure and offered the direct oral graded challenge and skin testing prior to oral challenge, 82% preferred the direct challenge. None of the patients who experienced a penicillin ADR in the past year wished to undergo skin testing or oral challenge, though each was educated on penicillin allergy and the possibility of testing in the future, making each encounter beneficial. Of the 17 patients tested, 16 (94%) tolerated oral amoxicillin and 1 (6%) experienced a mild, self-resolving ADR that was very unlikely of an IgE-mediated origin. Additionally, while plans and preparations for ADRs to the challenges were available, none were required. Patient questionnaires demonstrated the heterogeneity of previous ADRs and their attitude toward their allergy diagnosis. The positive impact of delabeling on patient well-being noted by 88% of patients reinforced the benefit of the effort.
This project was limited by a relatively small sample size, which may not have been large enough to detect ADRs, especially IgE-mediated allergic reactions. Herein lies the importance of having clinicians equipped to treat allergic ADRs to conduct penicillin challenges in the primary care setting. It is prudent to ensure not only proper training of physicians performing these challenges, but also appropriate equipment, medication, and response personnel. Medications that are useful include epinephrine, OAHs, albuterol, steroids, and intravenous fluids.
Having a response area and plan are essential to ensure appropriate care in the rare instance of allergic ADRs progressing to anaphylaxis. In rare cases, emergency medical services may be required and having a plan with appropriate response and transport time is essential to patient safety. This may not be practical in more rural or smaller practices. In those scenarios, it may be helpful to partner with a larger practice to send patients for delabeling or to use clinical space in closer proximity to emergency services. Perhaps an ideal setting might be urgent or emergent care centers due to high acuity resources and frequent prescription of amoxicillin antibiotics; however, this may be complicated by concurrent infections raising the incidence of delayed benign eruptions with amoxicillin ingestion and complicating the patient’s allergy records. Further training of urgent and emergent care practitioners would be helpful for proper patient education regarding antibiotic-associated reactions.
Full testing integration into other primary care clinics may be limited due to the specialized training required for complete skin testing. Nevertheless, as shown in this project, most patients may be delabeled based on a PEN-FAST evaluation followed by oral challenge alone. Incorporation in other QI projects could involve continuing medical education to train staff physicians on PEN-FAST, teaching primary care residents during training, and site visits by allergists to train local physicians on testing. This project involved training 2 PCPs to conduct skin and oral challenge testing using PEN-FAST to guide clinical decision-making with an allergist available for consultation if needed. Future projects might model a similar approach or perhaps focus on training more physicians on oral challenges alone to reach a high percentage of the target population.
Conclusions
This project demonstrates a safe, efficient, and cost-effective model for penicillin allergy delabeling in clinics without regular access to allergy services. The use of PEN-FAST allows a quick and simple method to screen patients with penicillin allergy to meet the goals of the 2022 CBSs, but data are still accumulating to validate this method of screening across populations. This project demonstrates additional support for the use of PEN-FAST, while illustrating appropriate education regarding oral testing technique and its limitations.
Using an EHR report limited the patients in the testing pool and subsequent sample size. This suggests that a primary care identification-driven enrollment in testing may offer even more benefit both in allergy detection and education of testing benefits. Oral challenges are more cost effective, shorter in duration, and have fewer training requirements when compared with antecedent skin testing, making them an ideal option for PCPs in a clinic setting. Trained PCPs may opt to offer periodic appointments for delabeling, or offer days dedicated to delabeling as many patients as possible. Penicillin delabeling is an urgent and expansive charge; this study offers a replicable model for executing this important task.
Penicillin allergy is common in the United States. About 9.0% to 13.8% of patients have a diagnosed penicillin allergy documented in their electronic health record. The annual incidence rates is 1.1% in males and 1.4% in females.1,2
Penicillin hypersensitivity likely wanes over time. A 1981 study found that 93% of patients who experienced an allergic reaction to penicillin had a positive skin test 7 to 12 months postreaction, but only 22% still had a positive test after 10 years.3 Confirmed type 1 hypersensitivity penicillin allergies, as demonstrated by positive skin prick testing, also are decreasing over time.4 Furthermore, many patients’ reactions may have been misdiagnosed as a penicillin allergy. Upon actual confirmatory testing of penicillin allergy, only 8.5% to 13.8% of patients believed to have a penicillin allergy were positive on skin prick testing of penicillin products.5,6 A 2024 US study found that 11% of individuals with a history of a penicillin reaction tested positive on skin testing.7
The positive predictive value of penicillin allergy skin testing is poorly defined due to the ethical dilemma of orally challenging a patient who demonstrates skin test reactivity. Due to its high negative predictive value (NPV), skin prick combined with intradermal testing has been the gold-standard test in cases of clinical concern.6 Patients with positive skin testing are assumed to be truly positive, and therefore penicillin allergic, even though false-positive results to penicillin skin testing are known to occur.8
Misdiagnosis of penicillin allergy carries substantial clinical and economic consequences. A 2011 study suggested a statistically significant 1.8% increased absolute risk of mortality and 5.5% increased absolute risk of intensive care unit admission for those labeled with penicillin allergy and admitted for an infection.9 Another study found a 14% increase in mortality associated with the diagnosis of penicillin allergy.10 In a 2014 case-control study, penicillin allergy also was associated with a 23.4% greater risk of Clostridioides difficile, 14.1% more methicillin-resistant Staphylococcus aureus, and 30.1% more vancomycin-resistant enterococci infections.11Direct cost savings during an inpatient admission for infection were as much as $609 per patient with additional indirect cost savings of up to $4254 per admission.12 When viewed from the perspective of a health care system, these costs quickly accumulate, negatively impacting the fiscal stability of our patients and placing additional financial strain on an over-burdened system.
If 10% of US patients have penicillin allergy labels, then about 33 million patients might be eligible for delabeling. There are only 6309 board-certified allergists actively practicing in the US, which could amount to about 5231 potential penicillin challenges per allergist, not even including the 3.3 million new patients per year (assuming a 1% incidence).13 Clarifying each patient’s tolerance of penicillin products will clearly require nonallergist cooperation.
The 2022 drug allergy practice parameter update recommends several consensus-based statements (CBSs) to directly address penicillin allergy.14 This guideline recommends proactive efforts to delabel patients with a reported penicillin allergy (CBS 4); advise against testing in cases where the history is inconsistent with a true allergic reaction, though a challenge may be offered (CBS 5); skin testing for those with a history of anaphylaxis or a recent reaction (CBS 6); advise against multiple-day penicillin challenges (CBS 7); advise against skin testing for pediatric patients with benign cutaneous reactions (CBS 8); and recommends direct oral challenge for adults with distant or benign cutaneous reactions (CBS 9). These recommendations create a potentially high demand for delabeling with allergy specialists. One potential solution is to perform direct oral challenges in primary care, emergency departments, and urgent care clinics.
Evidence supporting the safety of direct oral penicillin challenges in low-risk patients was initially noted in the allergy community, but now evidence for their use in primary care clinics is growing—including in children.15 In a military-specific population, an amoxicillin challenge of Marine recruits with suspected penicillin allergy revealed that only 1.5% of those challenged acutely reacted and should be considered allergic to penicillin.16 Historically, in order to refute the diagnosis of penicillin allergy, an allergist would order penicillin skin prick testing. If the test was negative, an allergist would proceed to intradermal testing and if negative again (NPV of 97.9%), proceed to a graded oral challenge.6 However, this process is not fully reproducible in most clinics because the minor determinants mixture used in skin testing is not commercially available.17 Additionally, the full skin testing procedure requires specialized training, is more time-consuming, causes more discomfort, lacks US Food and Drug Administration approval for children, and has a higher cost ($220 per test for each patient as of 2016).18 As such, the movement toward direct oral challenges is progressing. Nonetheless, the best method for primary care or emergency department clinicians to determine who the appropriate patients are for this procedure has not been fully established. Risk tools have been created in the past to help delineate low-risk patients who would be appropriate for direct oral amoxicillin challenges, but these were not widely replicated or validated.19 The PEN-FAST standardized risk score was first published in 2020 and has since been validated in different groups with additional safety data. This scoring system ranges from 0 to 5 points, assigning 2 points for a penicillin reaction within the past five (F) years, 2 points for angioedema/anaphylaxis (A) or a severe (S) cutaneous reaction, and 1 point if treatment (T) was required for the reaction. A score < 3 is considered low-risk and safe for direct oral challenge, although most of the safety data are in patients with a score of 0 or 1.20 The PEN-FAST guided direct oral challenge with an NPV of 96.3% has now been prospectively shown to be noninferior to standard skin prick test/intradermal test/graded challenge for low-risk patients with a PEN-FAST score < 3.21 The PEN-FAST validating study was conducted predominantly with an Australian population of adult White women, but now it also has been validated in children aged > 12 years, as well as in European and North American cohorts.22-24
Air Force Delabeling Program
This article describes a method for proactively, safely, and efficiently delabeling penicillin allergic patients at an Air Force clinic. This quality improvement (QI) report provides a successful model for penicillin allergy delabeling, illustrates lessons learned, and suggests next steps toward improving patient options for an invaluable antibiotic class.
The first step was to proactively delabel penicillin allergy from a population of active duty service members and their dependents. Electronic health record (EHR) allergy search functions are a helpful tool in finding patients with allergy labels. The Kadena Medical Clinic, in Okinawa, Japan, uses the Military Health System GENESIS EHR, which includes a discern reporting portal with a patient allergy search that creates a patient-specific medication allergy report. To compile the most complete database of patients with a penicillin allergy, all 15 potential allergy search options for “penicillin” were selected, as were 4 relevant options for amoxicillin (including options with clavulanate). Including so many options for specific penicillin medication allergies helps add specificity to the diagnosis in the EHR but can make aggregation of data more difficult. The
The complete compiled list was manually reviewed for high-risk patients with severe cutaneous adverse reactions (SCARs) of any age. Patients with pregnancy, unsuitable medical histories (ie, severe asthma), or taking β-blockers were excluded. Patients remaining on the list were contacted by telephone and offered appointments during a single week that was dedicated to penicillin allergy delabeling. Allergists in the Air Force are assigned to a region where they offer allergy services at clinics without a regular allergist. The allergist for the region traveled to the QI site for a 1-week campaign at an estimated cost of $4600. When the patients were contacted, they were briefly informed of the goal of the penicillin delabeling campaign, and if interested, they were scheduled for 1 of 50 available appointments that week. Patients were contacted with enough lead time to stop oral antihistamines (OAH) for ≥ 7 days before the appointment.
Patients were given an intake questionnaire and interviewed about their penicillin allergy history. This questionnaire inquired about the nature of the allergy, mental and physical health impacts of the allergy label, PEN-FAST scoring questions, and posttest attitude toward delabeling, if applicable. Patients with a PEN-FAST score < 3 were offered direct, graded oral challenge or the standard skin prick, followed by intradermal, followed by graded oral challenge protocol. Patients with PEN-FAST scores of ≥ 3 were offered skin testing prior to oral challenge protocol. Patients could decline further testing. If patients wished to proceed, they were asked to complete a written informed consent document.
Oral challenges followed a 10%/90% protocol, beginning with 50 mg of liquid amoxicillin followed by 450 mg after 15 minutes, as long as the patient remained asymptomatic. Challenge forms are available in the eAppendix . After receiving the 450-mg amoxicillin dose, the patient remained in the clinic for 60 minutes before a final clinical evaluation. If the patient remained asymptomatic after this period, the penicillin or amoxicillin allergy was marked as resolved in the EHR. The patients were given contact information for the clinic for follow-up if a delayed reaction was noted and they wished the medication allergy to be re-entered. An EHR encounter note was created for each patient detailing the allergy testing and delabeling.

This campaign was conducted at a basic life support-only facility by a single clinician without medical technician support. An allergic reaction medication kit was available and contained OAHs, intramuscular antihistamines, intramuscular epinephrine, intramuscular corticosteroids, and short-acting β-agonists for nebulization. The facility also had an urgent care room (staffed by primary care practitioners [PCPs]) that could help establish intravenous access and administer fluids if necessary and had previously established plans for emergency patient transport to a higher level of care, if necessary.
Program Outcomes
A list of 65 patients that included both active-duty service members and dependents with penicillin or amoxicillin allergy was created. This list was reviewed by an allergist to identify high-risk individuals, which required about 90 minutes. Two patients (3%) were excluded; 1 had a history of SCAR to penicillin and 1 had a complex medical history requiring continued OAH use. Sixty-three patients were contacted via telephone, and 29 patients (46%) scheduled an appointment. One patient (2%) was identified as penicillin-tolerant during the booking process, and the penicillin allergy was removed without testing (Figure 1).

Of the 29 scheduled patients, 5 patients (17%) failed to present for care. Of the potential appointments set aside for the program, only 42% were used. One patient (4%) who was seen in clinic was delabeled based on history alone as they had previously successfully tolerated a course of amoxicillin. Four patients (17%) declined further testing with a PEN-FAST score > 2 due to a clear history of acute immunoglobulin (Ig) E-mediated reaction to a penicillin product within the past year. One patient (4%) was unable to be tested due to ongoing OAH use and 1 patient (4%) declined further penicillin testing after the discussion about risks, benefits, and alternatives to the procedures offered.
Of the 24 patients who arrived for a clinic appointment, 17 (71%) underwent penicillin allergy delabeling testing: 14 (82%) underwent direct challenge, and 3 (18%) underwent the skin testing before oral amoxicillin challenge procedure. Of the 17 who were tested, 16 (94%) tolerated a total dose of 500 mg of oral amoxicillin within the 1-hour observation period. One tested patient (6%) in the direct oral challenge group experienced an adverse reaction that was described as dull headache and hand tremor after the 50-mg dose; although it self-resolved within 15 minutes, this prompted the patient to discontinue the challenge. This adverse reaction was determined to be very unlikely IgE-mediated. None of the 3 patients who underwent the skin testing before oral challenge protocol experienced an adverse drug reaction (ADR). None of the 17 patients who received any oral amoxicillin required follow-up or reported a delayed cutaneous ADR to the challenge. No OAHs or epinephrine were used for any of the challenges.
Data collected from patient questionnaires displayed perceived health impacts of a penicillin allergy on the patient population. Patients reported a variety of ADRs to previous administration of penicillin products: 17 (71%) reported urticaria, 2 (8%) reported anaphylaxis, and 3 (13%) were unable to recall the reaction (Figure 2). Nine patients (38%) felt their initial reaction was distressing. Fifteen patients (88%) felt relief following negative testing (Table).


Discussion
To our knowledge, this was the first documented proactive penicillin delabeling QI project in a military clinic treating both active-duty service members and their dependents, modeled on the 2022 drug allergy guidelines.14 Several interesting lessons were learned that may improve future similar QI projects. Only 46% of patients identified as having penicillin allergy presented for evaluation, leaving 42% of available appointments unused. Without prior data on anticipated participation rates, these data provide a crude benchmark for utilization rates, which can inform future resource planning. While attempts were made to contact each patient, additional efforts to publicize the penicillin allergy delabeling campaign would have been useful to improve efficiency.
In addition, when patients with a PEN-FAST score of < 3 were educated about the risks and benefits of each procedure and offered the direct oral graded challenge and skin testing prior to oral challenge, 82% preferred the direct challenge. None of the patients who experienced a penicillin ADR in the past year wished to undergo skin testing or oral challenge, though each was educated on penicillin allergy and the possibility of testing in the future, making each encounter beneficial. Of the 17 patients tested, 16 (94%) tolerated oral amoxicillin and 1 (6%) experienced a mild, self-resolving ADR that was very unlikely of an IgE-mediated origin. Additionally, while plans and preparations for ADRs to the challenges were available, none were required. Patient questionnaires demonstrated the heterogeneity of previous ADRs and their attitude toward their allergy diagnosis. The positive impact of delabeling on patient well-being noted by 88% of patients reinforced the benefit of the effort.
This project was limited by a relatively small sample size, which may not have been large enough to detect ADRs, especially IgE-mediated allergic reactions. Herein lies the importance of having clinicians equipped to treat allergic ADRs to conduct penicillin challenges in the primary care setting. It is prudent to ensure not only proper training of physicians performing these challenges, but also appropriate equipment, medication, and response personnel. Medications that are useful include epinephrine, OAHs, albuterol, steroids, and intravenous fluids.
Having a response area and plan are essential to ensure appropriate care in the rare instance of allergic ADRs progressing to anaphylaxis. In rare cases, emergency medical services may be required and having a plan with appropriate response and transport time is essential to patient safety. This may not be practical in more rural or smaller practices. In those scenarios, it may be helpful to partner with a larger practice to send patients for delabeling or to use clinical space in closer proximity to emergency services. Perhaps an ideal setting might be urgent or emergent care centers due to high acuity resources and frequent prescription of amoxicillin antibiotics; however, this may be complicated by concurrent infections raising the incidence of delayed benign eruptions with amoxicillin ingestion and complicating the patient’s allergy records. Further training of urgent and emergent care practitioners would be helpful for proper patient education regarding antibiotic-associated reactions.
Full testing integration into other primary care clinics may be limited due to the specialized training required for complete skin testing. Nevertheless, as shown in this project, most patients may be delabeled based on a PEN-FAST evaluation followed by oral challenge alone. Incorporation in other QI projects could involve continuing medical education to train staff physicians on PEN-FAST, teaching primary care residents during training, and site visits by allergists to train local physicians on testing. This project involved training 2 PCPs to conduct skin and oral challenge testing using PEN-FAST to guide clinical decision-making with an allergist available for consultation if needed. Future projects might model a similar approach or perhaps focus on training more physicians on oral challenges alone to reach a high percentage of the target population.
Conclusions
This project demonstrates a safe, efficient, and cost-effective model for penicillin allergy delabeling in clinics without regular access to allergy services. The use of PEN-FAST allows a quick and simple method to screen patients with penicillin allergy to meet the goals of the 2022 CBSs, but data are still accumulating to validate this method of screening across populations. This project demonstrates additional support for the use of PEN-FAST, while illustrating appropriate education regarding oral testing technique and its limitations.
Using an EHR report limited the patients in the testing pool and subsequent sample size. This suggests that a primary care identification-driven enrollment in testing may offer even more benefit both in allergy detection and education of testing benefits. Oral challenges are more cost effective, shorter in duration, and have fewer training requirements when compared with antecedent skin testing, making them an ideal option for PCPs in a clinic setting. Trained PCPs may opt to offer periodic appointments for delabeling, or offer days dedicated to delabeling as many patients as possible. Penicillin delabeling is an urgent and expansive charge; this study offers a replicable model for executing this important task.
- Macy E, Poon KYT. Self-reported antibiotic allergy incidence and prevalence: age and sex effects. Am J Med. 2009;122(8):778.e1-778.e7787. doi:10.1016/j.amjmed.2009.01.034
- Zhou L, Dhopeshwarkar N, Blumenthal KG, et al. Drug allergies documented in electronic health records of a large healthcare system. Allergy. 2016;71(9):1305-1313. doi:10.1111/all.12881
- Sullivan TJ, Wedner HJ, Shatz GS, Yecies LD, Parker CW. Skin testing to detect penicillin allergy. J Allergy Clin Immunol. 1981;68(3):171-180. doi:10.1016/0091-6749(81)90180-9
- Macy E, Schatz M, Lin C, Poon KY. The falling rate of positive penicillin skin tests from 1995 to 2007. Perm J. 2009;13(2):12-18. doi:10.7812/TPP/08-073
- Fox SJ, Park MA. Penicillin skin testing is a safe and effective tool for evaluating penicillin allergy in the pediatric population. J Allergy Clin Immunol Pract. 2014;2(4):439-444. doi:10.1016/j.jaip.2014.04.013
- Solensky R, Jacobs J, Lester M, et al. Penicillin Allergy Evaluation: A Prospective, Multicenter, Open-Label Evaluation of a Comprehensive Penicillin Skin Test Kit. J Allergy Clin Immunol Pract. 2019;7(6):1876-1885.e3. doi:10.1016/j.jaip.2019.02.040 7.
- Gonzalez-Estrada A, Park MA, Accarino JJO, et al. Predicting penicillin allergy: A United States multicenter retrospective study. J Allergy Clin Immunol Pract. 2024;12(5):1181-1191.e10. doi:10.1016/j.jaip.2024.01.010
- Stüwe HT, Geissler W, Paap A, Cromwell O. The presence of latex can induce false-positive skin tests in subjects tested with penicillin determinants. Allergy. 1997;52(12):1243. doi:10.1111/j.1398-9995.1997.tb00975.x
- Charneski L, Deshpande G, Smith SW. Impact of an antimicrobial allergy label in the medical record on clinical outcomes in hospitalized patients. Pharmacotherapy. 2011;31(8):742-747. doi:10.1592/phco.31.8.742
- Blumenthal KG, Lu N, Zhang Y, Walensky RP, Choi HK. Recorded penicillin allergy and risk of mortality: a population-based matched cohort study. J Gen Intern Med. 2019;34(9):1685-1687. doi:10.1007/s11606-019-04991-y
- Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: A cohort study. J Allergy Clin Immunol. 2014;133(3):790-796. doi:10.1016/j.jaci.2013.09.021
- Mattingly TJ II, Fulton A, Lumish RA, et al. The cost of self-reported penicillin allergy: a systematic review. J Allergy Clin Immunol Pract. 2018;6(5):1649-1654.e4. doi:10.1016/j.jaip.2017.12.033
- Diplomate Statistics. American Board of Allergy and Immunology website. Published February, 18 2021. Accessed July 28, 2025. https://www.abai.org/statistics_diplomates.asp
- Khan DA, Banerji A, Blumenthal KG, et al. Drug allergy: a 2022 practice parameter update. J Allergy Clin Immunol. 2022;150(6):1333-1393. doi:10.1016/j.jaci.2022.08.028
- Mill C, Primeau MN, Medoff E, et al. Assessing the diagnostic properties of a graded oral provocation challenge for the diagnosis of immediate and nonimmediate reactions to amoxicillin in children. JAMA Pediatr. 2016;170:e160033. doi:10.1001/jamapediatrics.2016.0033
- Tucker MH, Lomas CM, Ramchandar N, Waldram JD. Amoxicillin challenge without penicillin skin testing in evaluation of penicillin allergy in a cohort of Marine recruits. J Allergy Clin Immunol Pract. 2017;5(3):813-815. doi:10.1016/j.jaip.2017.01.023
- Shenoy ES, Macy E, Rowe T, Blumenthal KG. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321(2):188–99. doi:10.1001/jama.2018.19283
- Blumenthal KG, Li Y, Banerji A, et al. The cost of penicillin allergy evaluation. J Allergy Clin Immunol Pract. 2018;6(3):1019-1027.e2. doi:10.1016/j.jaip.2017.08.006
- Banks TA, Tucker M, Macy E. Evaluating penicillin allergies without skin testing. Curr Allergy Asthma Rep. 2019;19(5):27. doi:10.1007/s11882-019-0854-6
- Trubiano JA, Vogrin S, Chua KYL, et al. Development and validation of a penicillin allergy clinical decision rule. JAMA Intern Med. 2020;180(5):745-752. doi:10.1001/jamainternmed.2020.0403
- Copaescu AM, Vogrin S, James F, et al. Efficacy of a clinical decision rule to enable direct oral challenge in patients with low-risk penicillin allergy: the PALACE randomized clinical trial. JAMA Intern Med. 2023;183(9):944-952. doi:10.1001/jamainternmed.2023.2986
- Copaescu AM, Vogrin S, Shand G, et al. Validation of the PEN-FAST score in a pediatric population. JAMA Netw Open. 2022;5(9):e2233703. doi:10.1001/jamanetworkopen.2022.33703
- Piotin A, Godet J, Trubiano JA, et al. Predictive factors of amoxicillin immediate hypersensitivity and validation of PEN-FAST clinical decision rule. Ann Allergy Asthma Immunol. 2022;128(1):27-32. doi:10.1016/j.anai.2021.07.005
- Su C, Belmont A, Liao J, et al. Evaluating the PEN-FAST clinical decision-making tool to enhance penicillin allergy delabeling. JAMA Intern Med. 2023;183(8):883-885. doi:10.1001/jamainternmed.2023.1572
- Macy E, Poon KYT. Self-reported antibiotic allergy incidence and prevalence: age and sex effects. Am J Med. 2009;122(8):778.e1-778.e7787. doi:10.1016/j.amjmed.2009.01.034
- Zhou L, Dhopeshwarkar N, Blumenthal KG, et al. Drug allergies documented in electronic health records of a large healthcare system. Allergy. 2016;71(9):1305-1313. doi:10.1111/all.12881
- Sullivan TJ, Wedner HJ, Shatz GS, Yecies LD, Parker CW. Skin testing to detect penicillin allergy. J Allergy Clin Immunol. 1981;68(3):171-180. doi:10.1016/0091-6749(81)90180-9
- Macy E, Schatz M, Lin C, Poon KY. The falling rate of positive penicillin skin tests from 1995 to 2007. Perm J. 2009;13(2):12-18. doi:10.7812/TPP/08-073
- Fox SJ, Park MA. Penicillin skin testing is a safe and effective tool for evaluating penicillin allergy in the pediatric population. J Allergy Clin Immunol Pract. 2014;2(4):439-444. doi:10.1016/j.jaip.2014.04.013
- Solensky R, Jacobs J, Lester M, et al. Penicillin Allergy Evaluation: A Prospective, Multicenter, Open-Label Evaluation of a Comprehensive Penicillin Skin Test Kit. J Allergy Clin Immunol Pract. 2019;7(6):1876-1885.e3. doi:10.1016/j.jaip.2019.02.040 7.
- Gonzalez-Estrada A, Park MA, Accarino JJO, et al. Predicting penicillin allergy: A United States multicenter retrospective study. J Allergy Clin Immunol Pract. 2024;12(5):1181-1191.e10. doi:10.1016/j.jaip.2024.01.010
- Stüwe HT, Geissler W, Paap A, Cromwell O. The presence of latex can induce false-positive skin tests in subjects tested with penicillin determinants. Allergy. 1997;52(12):1243. doi:10.1111/j.1398-9995.1997.tb00975.x
- Charneski L, Deshpande G, Smith SW. Impact of an antimicrobial allergy label in the medical record on clinical outcomes in hospitalized patients. Pharmacotherapy. 2011;31(8):742-747. doi:10.1592/phco.31.8.742
- Blumenthal KG, Lu N, Zhang Y, Walensky RP, Choi HK. Recorded penicillin allergy and risk of mortality: a population-based matched cohort study. J Gen Intern Med. 2019;34(9):1685-1687. doi:10.1007/s11606-019-04991-y
- Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: A cohort study. J Allergy Clin Immunol. 2014;133(3):790-796. doi:10.1016/j.jaci.2013.09.021
- Mattingly TJ II, Fulton A, Lumish RA, et al. The cost of self-reported penicillin allergy: a systematic review. J Allergy Clin Immunol Pract. 2018;6(5):1649-1654.e4. doi:10.1016/j.jaip.2017.12.033
- Diplomate Statistics. American Board of Allergy and Immunology website. Published February, 18 2021. Accessed July 28, 2025. https://www.abai.org/statistics_diplomates.asp
- Khan DA, Banerji A, Blumenthal KG, et al. Drug allergy: a 2022 practice parameter update. J Allergy Clin Immunol. 2022;150(6):1333-1393. doi:10.1016/j.jaci.2022.08.028
- Mill C, Primeau MN, Medoff E, et al. Assessing the diagnostic properties of a graded oral provocation challenge for the diagnosis of immediate and nonimmediate reactions to amoxicillin in children. JAMA Pediatr. 2016;170:e160033. doi:10.1001/jamapediatrics.2016.0033
- Tucker MH, Lomas CM, Ramchandar N, Waldram JD. Amoxicillin challenge without penicillin skin testing in evaluation of penicillin allergy in a cohort of Marine recruits. J Allergy Clin Immunol Pract. 2017;5(3):813-815. doi:10.1016/j.jaip.2017.01.023
- Shenoy ES, Macy E, Rowe T, Blumenthal KG. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321(2):188–99. doi:10.1001/jama.2018.19283
- Blumenthal KG, Li Y, Banerji A, et al. The cost of penicillin allergy evaluation. J Allergy Clin Immunol Pract. 2018;6(3):1019-1027.e2. doi:10.1016/j.jaip.2017.08.006
- Banks TA, Tucker M, Macy E. Evaluating penicillin allergies without skin testing. Curr Allergy Asthma Rep. 2019;19(5):27. doi:10.1007/s11882-019-0854-6
- Trubiano JA, Vogrin S, Chua KYL, et al. Development and validation of a penicillin allergy clinical decision rule. JAMA Intern Med. 2020;180(5):745-752. doi:10.1001/jamainternmed.2020.0403
- Copaescu AM, Vogrin S, James F, et al. Efficacy of a clinical decision rule to enable direct oral challenge in patients with low-risk penicillin allergy: the PALACE randomized clinical trial. JAMA Intern Med. 2023;183(9):944-952. doi:10.1001/jamainternmed.2023.2986
- Copaescu AM, Vogrin S, Shand G, et al. Validation of the PEN-FAST score in a pediatric population. JAMA Netw Open. 2022;5(9):e2233703. doi:10.1001/jamanetworkopen.2022.33703
- Piotin A, Godet J, Trubiano JA, et al. Predictive factors of amoxicillin immediate hypersensitivity and validation of PEN-FAST clinical decision rule. Ann Allergy Asthma Immunol. 2022;128(1):27-32. doi:10.1016/j.anai.2021.07.005
- Su C, Belmont A, Liao J, et al. Evaluating the PEN-FAST clinical decision-making tool to enhance penicillin allergy delabeling. JAMA Intern Med. 2023;183(8):883-885. doi:10.1001/jamainternmed.2023.1572
Proactive Penicillin Allergy Delabeling: Lessons Learned From a Quality Improvement Project
Proactive Penicillin Allergy Delabeling: Lessons Learned From a Quality Improvement Project
Around 5% of US Population Diagnosed With Autoimmune Disease
TOPLINE:
In 2022, autoimmune diseases affected over 15 million individuals in the United States, with women nearly twice as likely to be affected as men and more than one third of affected individuals having more than one autoimmune condition.
METHODOLOGY:
- Researchers used electronic health record (EHR) data from six healthcare systems in the United States between 2011 and 2022 to estimate the prevalence of autoimmune diseases according to sex and age.
- They selected 105 autoimmune diseases from the textbook The Autoimmune Diseases and estimated their prevalence in more than 10 million individuals from these healthcare systems; these statistics were subsequently extrapolated to an estimated US population of 333.3 million.
- An individual was considered to have a diagnosis of an autoimmune disease if they had at least two diagnosis codes for the condition, with the codes being at least 30 days apart.
- A software program was developed to compute the prevalence of autoimmune diseases alone and in aggregate, enabling other researchers to replicate or modify the analysis over time.
TAKEAWAY:
- More than 15 million people, accounting for 4.6% of the US population, were diagnosed with at least one autoimmune disease from January 2011 to June 2022; 34% were diagnosed with more than one autoimmune disease.
- Sex-stratified analysis revealed that 63% of patients diagnosed with autoimmune disease were women, and only 37% were men, establishing a female-to-male ratio of 1.7:1; age-stratified analysis revealed increasing prevalence of autoimmune conditions with age, peaking in individuals aged ≥ 65 years.
- Among individuals with autoimmune diseases, 65% of patients had one condition, whereas 24% had two, 8% had three, and 2% had four or more autoimmune diseases (does not add to 100% due to rounding).
- Rheumatoid arthritis emerged as the most prevalent autoimmune disease, followed by psoriasis, type 1 diabetes, Grave’s disease, and autoimmune thyroiditis; 19 of the top 20 most prevalent autoimmune diseases occurred more frequently in women.
IN PRACTICE:
“Accurate data on the prevalence of autoimmune diseases as a category of disease and for individual autoimmune diseases are needed to further clinical and basic research to improve diagnosis, biomarkers, and therapies for these diseases, which significantly impact the US population,” the authors wrote.
SOURCE:
The study was led by Aaron H. Abend, Autoimmune Registry, Guilford, Connecticut, and was published online in The Journal of Clinical Investigation.
LIMITATIONS:
The use of EHR data presented several challenges, including potential inaccuracies in diagnosis codes and the possibility of missing patients with single diagnosis codes because of the two-code requirement. Certain autoimmune diseases evolve over time and involve nonspecific clinical signs and symptoms that can mimic other diseases, potentially resulting in underdiagnosis. Moreover, rare diseases lacking specific diagnosis codes may have been underrepresented.
DISCLOSURES:
The study received support from Autoimmune Registry; the National Institutes of Health National Center for Advancing Translational Sciences; the National Heart, Lung, and Blood Institute; and other sources. Information on potential conflicts of interest was not disclosed.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
In 2022, autoimmune diseases affected over 15 million individuals in the United States, with women nearly twice as likely to be affected as men and more than one third of affected individuals having more than one autoimmune condition.
METHODOLOGY:
- Researchers used electronic health record (EHR) data from six healthcare systems in the United States between 2011 and 2022 to estimate the prevalence of autoimmune diseases according to sex and age.
- They selected 105 autoimmune diseases from the textbook The Autoimmune Diseases and estimated their prevalence in more than 10 million individuals from these healthcare systems; these statistics were subsequently extrapolated to an estimated US population of 333.3 million.
- An individual was considered to have a diagnosis of an autoimmune disease if they had at least two diagnosis codes for the condition, with the codes being at least 30 days apart.
- A software program was developed to compute the prevalence of autoimmune diseases alone and in aggregate, enabling other researchers to replicate or modify the analysis over time.
TAKEAWAY:
- More than 15 million people, accounting for 4.6% of the US population, were diagnosed with at least one autoimmune disease from January 2011 to June 2022; 34% were diagnosed with more than one autoimmune disease.
- Sex-stratified analysis revealed that 63% of patients diagnosed with autoimmune disease were women, and only 37% were men, establishing a female-to-male ratio of 1.7:1; age-stratified analysis revealed increasing prevalence of autoimmune conditions with age, peaking in individuals aged ≥ 65 years.
- Among individuals with autoimmune diseases, 65% of patients had one condition, whereas 24% had two, 8% had three, and 2% had four or more autoimmune diseases (does not add to 100% due to rounding).
- Rheumatoid arthritis emerged as the most prevalent autoimmune disease, followed by psoriasis, type 1 diabetes, Grave’s disease, and autoimmune thyroiditis; 19 of the top 20 most prevalent autoimmune diseases occurred more frequently in women.
IN PRACTICE:
“Accurate data on the prevalence of autoimmune diseases as a category of disease and for individual autoimmune diseases are needed to further clinical and basic research to improve diagnosis, biomarkers, and therapies for these diseases, which significantly impact the US population,” the authors wrote.
SOURCE:
The study was led by Aaron H. Abend, Autoimmune Registry, Guilford, Connecticut, and was published online in The Journal of Clinical Investigation.
LIMITATIONS:
The use of EHR data presented several challenges, including potential inaccuracies in diagnosis codes and the possibility of missing patients with single diagnosis codes because of the two-code requirement. Certain autoimmune diseases evolve over time and involve nonspecific clinical signs and symptoms that can mimic other diseases, potentially resulting in underdiagnosis. Moreover, rare diseases lacking specific diagnosis codes may have been underrepresented.
DISCLOSURES:
The study received support from Autoimmune Registry; the National Institutes of Health National Center for Advancing Translational Sciences; the National Heart, Lung, and Blood Institute; and other sources. Information on potential conflicts of interest was not disclosed.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
In 2022, autoimmune diseases affected over 15 million individuals in the United States, with women nearly twice as likely to be affected as men and more than one third of affected individuals having more than one autoimmune condition.
METHODOLOGY:
- Researchers used electronic health record (EHR) data from six healthcare systems in the United States between 2011 and 2022 to estimate the prevalence of autoimmune diseases according to sex and age.
- They selected 105 autoimmune diseases from the textbook The Autoimmune Diseases and estimated their prevalence in more than 10 million individuals from these healthcare systems; these statistics were subsequently extrapolated to an estimated US population of 333.3 million.
- An individual was considered to have a diagnosis of an autoimmune disease if they had at least two diagnosis codes for the condition, with the codes being at least 30 days apart.
- A software program was developed to compute the prevalence of autoimmune diseases alone and in aggregate, enabling other researchers to replicate or modify the analysis over time.
TAKEAWAY:
- More than 15 million people, accounting for 4.6% of the US population, were diagnosed with at least one autoimmune disease from January 2011 to June 2022; 34% were diagnosed with more than one autoimmune disease.
- Sex-stratified analysis revealed that 63% of patients diagnosed with autoimmune disease were women, and only 37% were men, establishing a female-to-male ratio of 1.7:1; age-stratified analysis revealed increasing prevalence of autoimmune conditions with age, peaking in individuals aged ≥ 65 years.
- Among individuals with autoimmune diseases, 65% of patients had one condition, whereas 24% had two, 8% had three, and 2% had four or more autoimmune diseases (does not add to 100% due to rounding).
- Rheumatoid arthritis emerged as the most prevalent autoimmune disease, followed by psoriasis, type 1 diabetes, Grave’s disease, and autoimmune thyroiditis; 19 of the top 20 most prevalent autoimmune diseases occurred more frequently in women.
IN PRACTICE:
“Accurate data on the prevalence of autoimmune diseases as a category of disease and for individual autoimmune diseases are needed to further clinical and basic research to improve diagnosis, biomarkers, and therapies for these diseases, which significantly impact the US population,” the authors wrote.
SOURCE:
The study was led by Aaron H. Abend, Autoimmune Registry, Guilford, Connecticut, and was published online in The Journal of Clinical Investigation.
LIMITATIONS:
The use of EHR data presented several challenges, including potential inaccuracies in diagnosis codes and the possibility of missing patients with single diagnosis codes because of the two-code requirement. Certain autoimmune diseases evolve over time and involve nonspecific clinical signs and symptoms that can mimic other diseases, potentially resulting in underdiagnosis. Moreover, rare diseases lacking specific diagnosis codes may have been underrepresented.
DISCLOSURES:
The study received support from Autoimmune Registry; the National Institutes of Health National Center for Advancing Translational Sciences; the National Heart, Lung, and Blood Institute; and other sources. Information on potential conflicts of interest was not disclosed.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
The Protein Problem: The Unsolved Mystery of AI Drug Dev
The question has been lingering for years in medical science circles. Since 2020, when the artificial intelligence (AI) model AlphaFold made it possible to predict protein structures, would the technology open the drug discovery floodgates?
Short answer: No. At least not yet.
The longer answer goes something like this:
A drug target (such as a mutation) is like a lock. The right drug (a protein designed to bind to the mutation, stopping its activity) is the key. But proteins are fidgety and flexible.
“They’re basically molecular springs,” said Gabriel Monteiro da Silva, PhD, a computational chemistry research scientist at Genesis Therapeutics. “Your key can bend and alter the shape of the lock, and if you don’t account for that, your key might fail.”
This is the protein problem in drug development. Another issue making this challenge so vexing is that proteins don’t act in isolation. Their interactions with other proteins, ribonucleic acid, and DNA can affect how they bind to molecules and the shapes they adopt.
Newer versions of AlphaFold, such as AlphaFold Multimer and AlphaFold 3 (the code for which was recently revealed for academic use), can predict many interactions among proteins and between proteins and other molecules. But these tools still have weak points scientists are trying to overcome or work around.
“Those kinds of dynamics and multiple conformations are still quite challenging for the AI models to predict,” said James Zou, PhD, associate professor of biomedical data science at Stanford University in California.
“We’re finding more and more that the only way we can make these structures useful for drug discovery is if we incorporate dynamics, if we incorporate more physics into the model,” said Monteiro da Silva.
Monteiro da Silva spent 3 years during his PhD at Brown University, Providence, Rhode Island, running physics-based simulations in the lab, trying to understand why proteins carrying certain mutations are drug resistant. His results showed how “the changing landscape of shapes that a protein can take” prevented the drug from binding.
It took him 3 years to model just four mutations.
AI can do better — and the struggle is fascinating. By developing models that build on the predictive power of AlphaFold, scientists are uncovering new details about protein activity — insights that can lead to new therapeutics and reveal why existing ones stop working — much faster than they could with traditional methods or AlphaFold alone.
New Windows into Protein Dynamics
A notable step, “but that’s just the starting point,” said Pedro Beltrao, PhD, an associate professor at Institute of Molecular Systems Biology, ETH Zurich in Switzerland. “It’s still very difficult, given a pocket, to actually design the drug or figure out what the pocket binds.”
Going back to the lock-and-key analogy: While he was at Brown, with a team of researchers in the Rubenstein Group, Monteiro da Silva helped create a model to better understand how mutations affect “the shape and dynamics of the lock.” They manipulated the amino acid sequences of proteins, guiding their evolution. This enabled them to use AlphaFold to predict “protein ensembles” and how frequently those ensembles appear. Each ensemble represents the many different shapes a protein can take under given conditions.
“Essentially, it tries to find the most common shapes that a protein will take over an arbitrary amount of time,” Monteiro da Silva said. “If we can predict these ensembles at scale and fast, then we can screen many mutations that cause resistance and develop drugs that will not be affected by that resistance.”
To evaluate their method, the researchers focused on ABL1, a well-studied kinase that causes leukemia. ABL1 can be drugged – unless it carries or develops a mutation that causes drug resistance. Currently there are no drugs that work against proteins carrying those mutations, according to Monteiro da Silva. The researchers used their hybrid AI-meets-physics method to investigate how drugs bind to different ABL1 mutations, screening 100 mutations in just 1 month.
“It’s not going to be perfect for every one of them. But if we have 100 and we get 20 with good accuracy, that’s better than doing four over 3 years,” Monteiro da Silva said.
A forthcoming paper will make their model publicly available in “an easy-to-use graphical interface” that they hope clinicians and medicinal chemists will try out. It can also complement other AI-based tools that dig into protein dynamics, according to Monteiro da Silva.
Complementary Tools to Speed Up Discovery
Another aspect of the protein problem is scale. One protein can interact with hundreds of other proteins, which in turn may interact with hundreds more, all of which comprise the human interactome.
Feixiong Cheng, PhD, helped build PIONEER, a deep learning model that predicts the three-dimensional (3D) structure of interactions between proteins across the interactome.
Most disease mutations disrupt specific interactions between proteins, making their affinity stronger or weaker, explained Cheng. To treat a disease without causing major side effects, scientists need a precise understanding of those interactions.
“From the drug discovery perspective, we cannot just focus on single proteins. We have to understand the protein environment, in particular how the protein interacts with other proteins,” said Cheng, director of Cleveland Clinic Genome Center, Cleveland.
PIONEER helps by blending AlphaFold’s protein structure predictions with next-generation sequencing, a type of genomic research that identifies mutations in the human genome. The model predicts the 3D structure of the places where proteins interact — the binding sites, or interfaces — across the interactome.
“We tell you not only that a binds b, but where on a and where on b the two proteins interact,” said Haiyuan Yu, PhD, director of the Center for Innovative Proteomics, Cornell University, and co-creator of PIONEER.
This can help scientists understand “why a mutation, protein, or even network is a good target for therapeutic discovery,” Cheng said.
The researchers validated PIONEER’s predictions in the lab, testing the impacts of roughly 3000 mutations on 7000 pairs of interacting proteins. Based on their findings, they plan to develop and test treatments for lung and endometrial cancer.
PIONEER can also help scientists home in on how a mutation causes a disease, such as by showing recurrent mutations.
“If you find cancer mutations hitting an interface again and again and again, it means that this is likely to be driving cancer progression,” said Beltrao.
Beltrao’s lab and others have looked for recurrent mutations by using AlphaFold Multimer and AlphaFold 3 to directly model protein interactions. It’s a much slower approach (Pioneer is more than 5000 faster than AlphaFold Multimer, according to Cheng). But it could allow scientists to model interfaces that are not shown by PIONEER.
“You will need many different things to try to come up with a structural modeling of the interactome, and all these will have limitations,” said Beltrao. “Their method is a very good step forward, and there’ll be other approaches that are complementary, to continue to add details.”
And It Wouldn’t be an AI Mission Without ChatGPT
Large language models, such as ChatGPT, are another way that scientists are adding details to protein structure predictions. Zou used GPT-4 to “fine tune” a protein language model, called evolutionary scale modeling (ESM-2), which predicts protein structures directly from a protein sequence.
First, they trained ChatGPT on thousands of papers and studies containing information about the functions, biophysical properties, and disease relevance of different mutations. Next, they used the trained model to “teach” ESM-2, boosting its ability “to predict which mutations are likely to have larger effects or smaller effects,” Zou said. The same could be done for a model like AlphaFold, according to Zou.
“They are quite complementary in that the large language model contains a lot more information about the functions and the biophysics of different mutations and proteins as captured in text,” he said, whereas “you can’t give AlphaFold a piece of paper.”
Exactly how AlphaFold makes its predictions is another mystery. “It will somehow learn protein dynamics phenomenologically,” said Monteiro da Silva. He and others are trying to understand how that happens, in hopes of creating even more accurate predictive models. But for the time being, AI-based methods still need assistance from physics.
“The dream is that we achieve a state where we rely on just the fast methods, and they’re accurate enough,” he said. “But we’re so far from that.”
A version of this article first appeared on Medscape.com.
The question has been lingering for years in medical science circles. Since 2020, when the artificial intelligence (AI) model AlphaFold made it possible to predict protein structures, would the technology open the drug discovery floodgates?
Short answer: No. At least not yet.
The longer answer goes something like this:
A drug target (such as a mutation) is like a lock. The right drug (a protein designed to bind to the mutation, stopping its activity) is the key. But proteins are fidgety and flexible.
“They’re basically molecular springs,” said Gabriel Monteiro da Silva, PhD, a computational chemistry research scientist at Genesis Therapeutics. “Your key can bend and alter the shape of the lock, and if you don’t account for that, your key might fail.”
This is the protein problem in drug development. Another issue making this challenge so vexing is that proteins don’t act in isolation. Their interactions with other proteins, ribonucleic acid, and DNA can affect how they bind to molecules and the shapes they adopt.
Newer versions of AlphaFold, such as AlphaFold Multimer and AlphaFold 3 (the code for which was recently revealed for academic use), can predict many interactions among proteins and between proteins and other molecules. But these tools still have weak points scientists are trying to overcome or work around.
“Those kinds of dynamics and multiple conformations are still quite challenging for the AI models to predict,” said James Zou, PhD, associate professor of biomedical data science at Stanford University in California.
“We’re finding more and more that the only way we can make these structures useful for drug discovery is if we incorporate dynamics, if we incorporate more physics into the model,” said Monteiro da Silva.
Monteiro da Silva spent 3 years during his PhD at Brown University, Providence, Rhode Island, running physics-based simulations in the lab, trying to understand why proteins carrying certain mutations are drug resistant. His results showed how “the changing landscape of shapes that a protein can take” prevented the drug from binding.
It took him 3 years to model just four mutations.
AI can do better — and the struggle is fascinating. By developing models that build on the predictive power of AlphaFold, scientists are uncovering new details about protein activity — insights that can lead to new therapeutics and reveal why existing ones stop working — much faster than they could with traditional methods or AlphaFold alone.
New Windows into Protein Dynamics
A notable step, “but that’s just the starting point,” said Pedro Beltrao, PhD, an associate professor at Institute of Molecular Systems Biology, ETH Zurich in Switzerland. “It’s still very difficult, given a pocket, to actually design the drug or figure out what the pocket binds.”
Going back to the lock-and-key analogy: While he was at Brown, with a team of researchers in the Rubenstein Group, Monteiro da Silva helped create a model to better understand how mutations affect “the shape and dynamics of the lock.” They manipulated the amino acid sequences of proteins, guiding their evolution. This enabled them to use AlphaFold to predict “protein ensembles” and how frequently those ensembles appear. Each ensemble represents the many different shapes a protein can take under given conditions.
“Essentially, it tries to find the most common shapes that a protein will take over an arbitrary amount of time,” Monteiro da Silva said. “If we can predict these ensembles at scale and fast, then we can screen many mutations that cause resistance and develop drugs that will not be affected by that resistance.”
To evaluate their method, the researchers focused on ABL1, a well-studied kinase that causes leukemia. ABL1 can be drugged – unless it carries or develops a mutation that causes drug resistance. Currently there are no drugs that work against proteins carrying those mutations, according to Monteiro da Silva. The researchers used their hybrid AI-meets-physics method to investigate how drugs bind to different ABL1 mutations, screening 100 mutations in just 1 month.
“It’s not going to be perfect for every one of them. But if we have 100 and we get 20 with good accuracy, that’s better than doing four over 3 years,” Monteiro da Silva said.
A forthcoming paper will make their model publicly available in “an easy-to-use graphical interface” that they hope clinicians and medicinal chemists will try out. It can also complement other AI-based tools that dig into protein dynamics, according to Monteiro da Silva.
Complementary Tools to Speed Up Discovery
Another aspect of the protein problem is scale. One protein can interact with hundreds of other proteins, which in turn may interact with hundreds more, all of which comprise the human interactome.
Feixiong Cheng, PhD, helped build PIONEER, a deep learning model that predicts the three-dimensional (3D) structure of interactions between proteins across the interactome.
Most disease mutations disrupt specific interactions between proteins, making their affinity stronger or weaker, explained Cheng. To treat a disease without causing major side effects, scientists need a precise understanding of those interactions.
“From the drug discovery perspective, we cannot just focus on single proteins. We have to understand the protein environment, in particular how the protein interacts with other proteins,” said Cheng, director of Cleveland Clinic Genome Center, Cleveland.
PIONEER helps by blending AlphaFold’s protein structure predictions with next-generation sequencing, a type of genomic research that identifies mutations in the human genome. The model predicts the 3D structure of the places where proteins interact — the binding sites, or interfaces — across the interactome.
“We tell you not only that a binds b, but where on a and where on b the two proteins interact,” said Haiyuan Yu, PhD, director of the Center for Innovative Proteomics, Cornell University, and co-creator of PIONEER.
This can help scientists understand “why a mutation, protein, or even network is a good target for therapeutic discovery,” Cheng said.
The researchers validated PIONEER’s predictions in the lab, testing the impacts of roughly 3000 mutations on 7000 pairs of interacting proteins. Based on their findings, they plan to develop and test treatments for lung and endometrial cancer.
PIONEER can also help scientists home in on how a mutation causes a disease, such as by showing recurrent mutations.
“If you find cancer mutations hitting an interface again and again and again, it means that this is likely to be driving cancer progression,” said Beltrao.
Beltrao’s lab and others have looked for recurrent mutations by using AlphaFold Multimer and AlphaFold 3 to directly model protein interactions. It’s a much slower approach (Pioneer is more than 5000 faster than AlphaFold Multimer, according to Cheng). But it could allow scientists to model interfaces that are not shown by PIONEER.
“You will need many different things to try to come up with a structural modeling of the interactome, and all these will have limitations,” said Beltrao. “Their method is a very good step forward, and there’ll be other approaches that are complementary, to continue to add details.”
And It Wouldn’t be an AI Mission Without ChatGPT
Large language models, such as ChatGPT, are another way that scientists are adding details to protein structure predictions. Zou used GPT-4 to “fine tune” a protein language model, called evolutionary scale modeling (ESM-2), which predicts protein structures directly from a protein sequence.
First, they trained ChatGPT on thousands of papers and studies containing information about the functions, biophysical properties, and disease relevance of different mutations. Next, they used the trained model to “teach” ESM-2, boosting its ability “to predict which mutations are likely to have larger effects or smaller effects,” Zou said. The same could be done for a model like AlphaFold, according to Zou.
“They are quite complementary in that the large language model contains a lot more information about the functions and the biophysics of different mutations and proteins as captured in text,” he said, whereas “you can’t give AlphaFold a piece of paper.”
Exactly how AlphaFold makes its predictions is another mystery. “It will somehow learn protein dynamics phenomenologically,” said Monteiro da Silva. He and others are trying to understand how that happens, in hopes of creating even more accurate predictive models. But for the time being, AI-based methods still need assistance from physics.
“The dream is that we achieve a state where we rely on just the fast methods, and they’re accurate enough,” he said. “But we’re so far from that.”
A version of this article first appeared on Medscape.com.
The question has been lingering for years in medical science circles. Since 2020, when the artificial intelligence (AI) model AlphaFold made it possible to predict protein structures, would the technology open the drug discovery floodgates?
Short answer: No. At least not yet.
The longer answer goes something like this:
A drug target (such as a mutation) is like a lock. The right drug (a protein designed to bind to the mutation, stopping its activity) is the key. But proteins are fidgety and flexible.
“They’re basically molecular springs,” said Gabriel Monteiro da Silva, PhD, a computational chemistry research scientist at Genesis Therapeutics. “Your key can bend and alter the shape of the lock, and if you don’t account for that, your key might fail.”
This is the protein problem in drug development. Another issue making this challenge so vexing is that proteins don’t act in isolation. Their interactions with other proteins, ribonucleic acid, and DNA can affect how they bind to molecules and the shapes they adopt.
Newer versions of AlphaFold, such as AlphaFold Multimer and AlphaFold 3 (the code for which was recently revealed for academic use), can predict many interactions among proteins and between proteins and other molecules. But these tools still have weak points scientists are trying to overcome or work around.
“Those kinds of dynamics and multiple conformations are still quite challenging for the AI models to predict,” said James Zou, PhD, associate professor of biomedical data science at Stanford University in California.
“We’re finding more and more that the only way we can make these structures useful for drug discovery is if we incorporate dynamics, if we incorporate more physics into the model,” said Monteiro da Silva.
Monteiro da Silva spent 3 years during his PhD at Brown University, Providence, Rhode Island, running physics-based simulations in the lab, trying to understand why proteins carrying certain mutations are drug resistant. His results showed how “the changing landscape of shapes that a protein can take” prevented the drug from binding.
It took him 3 years to model just four mutations.
AI can do better — and the struggle is fascinating. By developing models that build on the predictive power of AlphaFold, scientists are uncovering new details about protein activity — insights that can lead to new therapeutics and reveal why existing ones stop working — much faster than they could with traditional methods or AlphaFold alone.
New Windows into Protein Dynamics
A notable step, “but that’s just the starting point,” said Pedro Beltrao, PhD, an associate professor at Institute of Molecular Systems Biology, ETH Zurich in Switzerland. “It’s still very difficult, given a pocket, to actually design the drug or figure out what the pocket binds.”
Going back to the lock-and-key analogy: While he was at Brown, with a team of researchers in the Rubenstein Group, Monteiro da Silva helped create a model to better understand how mutations affect “the shape and dynamics of the lock.” They manipulated the amino acid sequences of proteins, guiding their evolution. This enabled them to use AlphaFold to predict “protein ensembles” and how frequently those ensembles appear. Each ensemble represents the many different shapes a protein can take under given conditions.
“Essentially, it tries to find the most common shapes that a protein will take over an arbitrary amount of time,” Monteiro da Silva said. “If we can predict these ensembles at scale and fast, then we can screen many mutations that cause resistance and develop drugs that will not be affected by that resistance.”
To evaluate their method, the researchers focused on ABL1, a well-studied kinase that causes leukemia. ABL1 can be drugged – unless it carries or develops a mutation that causes drug resistance. Currently there are no drugs that work against proteins carrying those mutations, according to Monteiro da Silva. The researchers used their hybrid AI-meets-physics method to investigate how drugs bind to different ABL1 mutations, screening 100 mutations in just 1 month.
“It’s not going to be perfect for every one of them. But if we have 100 and we get 20 with good accuracy, that’s better than doing four over 3 years,” Monteiro da Silva said.
A forthcoming paper will make their model publicly available in “an easy-to-use graphical interface” that they hope clinicians and medicinal chemists will try out. It can also complement other AI-based tools that dig into protein dynamics, according to Monteiro da Silva.
Complementary Tools to Speed Up Discovery
Another aspect of the protein problem is scale. One protein can interact with hundreds of other proteins, which in turn may interact with hundreds more, all of which comprise the human interactome.
Feixiong Cheng, PhD, helped build PIONEER, a deep learning model that predicts the three-dimensional (3D) structure of interactions between proteins across the interactome.
Most disease mutations disrupt specific interactions between proteins, making their affinity stronger or weaker, explained Cheng. To treat a disease without causing major side effects, scientists need a precise understanding of those interactions.
“From the drug discovery perspective, we cannot just focus on single proteins. We have to understand the protein environment, in particular how the protein interacts with other proteins,” said Cheng, director of Cleveland Clinic Genome Center, Cleveland.
PIONEER helps by blending AlphaFold’s protein structure predictions with next-generation sequencing, a type of genomic research that identifies mutations in the human genome. The model predicts the 3D structure of the places where proteins interact — the binding sites, or interfaces — across the interactome.
“We tell you not only that a binds b, but where on a and where on b the two proteins interact,” said Haiyuan Yu, PhD, director of the Center for Innovative Proteomics, Cornell University, and co-creator of PIONEER.
This can help scientists understand “why a mutation, protein, or even network is a good target for therapeutic discovery,” Cheng said.
The researchers validated PIONEER’s predictions in the lab, testing the impacts of roughly 3000 mutations on 7000 pairs of interacting proteins. Based on their findings, they plan to develop and test treatments for lung and endometrial cancer.
PIONEER can also help scientists home in on how a mutation causes a disease, such as by showing recurrent mutations.
“If you find cancer mutations hitting an interface again and again and again, it means that this is likely to be driving cancer progression,” said Beltrao.
Beltrao’s lab and others have looked for recurrent mutations by using AlphaFold Multimer and AlphaFold 3 to directly model protein interactions. It’s a much slower approach (Pioneer is more than 5000 faster than AlphaFold Multimer, according to Cheng). But it could allow scientists to model interfaces that are not shown by PIONEER.
“You will need many different things to try to come up with a structural modeling of the interactome, and all these will have limitations,” said Beltrao. “Their method is a very good step forward, and there’ll be other approaches that are complementary, to continue to add details.”
And It Wouldn’t be an AI Mission Without ChatGPT
Large language models, such as ChatGPT, are another way that scientists are adding details to protein structure predictions. Zou used GPT-4 to “fine tune” a protein language model, called evolutionary scale modeling (ESM-2), which predicts protein structures directly from a protein sequence.
First, they trained ChatGPT on thousands of papers and studies containing information about the functions, biophysical properties, and disease relevance of different mutations. Next, they used the trained model to “teach” ESM-2, boosting its ability “to predict which mutations are likely to have larger effects or smaller effects,” Zou said. The same could be done for a model like AlphaFold, according to Zou.
“They are quite complementary in that the large language model contains a lot more information about the functions and the biophysics of different mutations and proteins as captured in text,” he said, whereas “you can’t give AlphaFold a piece of paper.”
Exactly how AlphaFold makes its predictions is another mystery. “It will somehow learn protein dynamics phenomenologically,” said Monteiro da Silva. He and others are trying to understand how that happens, in hopes of creating even more accurate predictive models. But for the time being, AI-based methods still need assistance from physics.
“The dream is that we achieve a state where we rely on just the fast methods, and they’re accurate enough,” he said. “But we’re so far from that.”
A version of this article first appeared on Medscape.com.
Alpha-Gal Syndrome: 5 Things to Know
Alpha-gal syndrome (AGS), a tickborne disease commonly called “red meat allergy,” is a serious, potentially life-threatening allergy to the carbohydrate alpha-gal. The alpha-gal carbohydrate is found in most mammals, though it is not in humans, apes, or old-world monkeys. People with AGS can have allergic reactions when they consume mammalian meat, dairy products, or other products derived from mammals. People often live with this disease for years before receiving a correct diagnosis, greatly impacting their quality of life. The number of suspected cases is also rising.
More than 110,000 suspected AGS cases were identified between 2010 and 2022, according to a Centers for Disease Control and Prevention (CDC) report.1 However, because the diagnosis requires a positive test and a clinical exam and some people may not get tested, as many as 450,000 people might be affected by AGS in the United States. Additionally, a CDC survey found that nearly half (42%) of US healthcare providers had never heard of AGS.2 Among those who had, less than one third (29%) knew how to diagnose the condition.
Here are 5 things clinicians need to know about AGS.
1. People can develop AGS after being bitten by a tick, primarily the lone star tick (Amblyomma americanum), in the United States.
In the United States, AGS is primarily associated with the bite of a lone star tick, but other kinds of ticks have not been ruled out. The majority of suspected AGS cases in the United States were reported in parts of Arkansas, Delaware, Illinois, Indiana, Kansas, Kentucky, Maryland, Mississippi, Missouri, North Carolina, Oklahoma, Tennessee, and Virginia. The lone star tick is widely distributed with established populations in Alabama, Arkansas, Connecticut, Delaware, Florida, Georgia, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana, Michigan, Minnesota, Mississippi, Missouri, Nebraska, New Hampshire, New Jersey, New York, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Texas, Virginia, and West Virginia.
While AGS is associated with tick bites, more research is needed to understand the role ticks play in starting this condition, and why certain people develop AGS. Anyone can develop AGS, but most cases have been reported in adults.
Know how to recognize the symptoms of AGS and be prepared to test, diagnose, and manage AGS, particularly in states where lone star ticks are found.
2. Tick bites are only one risk factor for developing AGS.
Many people are bitten by lone star ticks and will never develop AGS. Scientists are exploring the connection between other risk factors and developing AGS. A recent study has shown that people diagnosed with AGS may be more likely to have a family member who was also diagnosed with AGS, have another food allergy, have an allergy to stinging or biting insects, or have A or O blood types.3
Research has also shown that environmental risk factors could contribute to developing AGS,4 like living in an area with lone star ticks, remembering finding a tick on themselves, recalling multiple tick bites, living near a wooded forest, spending more time outside, or living in areas with deer, such as larger properties, wooded forests, and properties with shrubs and brush.
Ask your patient questions about other allergies and history of recent tick bites or outdoor exposure to help determine if testing for AGS is appropriate.
3. Symptoms of AGS are consistently inconsistent.
There is a spectrum of how sensitive AGS patients are to alpha-gal, and reactions are often different from person to person, which can make it difficult to diagnose. The first allergic reaction to AGS typically occurs between 1-6 months after a tick bite. Symptoms commonly appear 2-6 hours after being in contact with products containing alpha-gal, like red meat (beef, pork, lamb, venison, rabbit, or other meat from mammals), dairy, and some medications. Symptoms can range from mild to severe and include hives or itchy rash; swelling of the lips, throat, tongue, or eyelids; gastrointestinal symptoms such as nausea, vomiting, or diarrhea; heartburn or indigestion; cough, shortness of breath, or difficulty breathing; dizziness or a drop in blood pressure; or anaphylaxis.
Consider AGS if a patient reports waking up in the middle of the night with allergic symptoms after eating alpha-gal containing products for dinner, if allergic reactions are delayed, or if a patient has anaphylaxis of unknown cause, adult-onset allergy, or allergic symptoms and reports a recent tick bite.
4. Diagnosing AGS requires a combination of a blood test and a physical exam.
Diagnosing AGS requires a detailed patient history, physical exam, and a blood test to detect specific immunoglobulin E (IgE) antibodies specific to alpha-gal (alpha-gal sIgE). Tests for alpha-gal sIgE antibodies are available at several large commercial laboratories and some academic institutions. Skin tests to identify reactions to allergens like pork or beef may also be used to inform AGS diagnosis. However, a positive alpha-gal sIgE test or skin test does not mean a person has AGS. Many people, particularly those who live in regions with lone star ticks, have positive alpha-gal specific IgE tests without having AGS.
Consider the test results along with your patient’s symptoms and risk factors.
5. There is no treatment for AGS, but people can take prevention steps and AGS can be managed.
People can protect themselves and their family from AGS by preventing tick bites. Encourage your patients to use an Environmental Protection Agency–registered insect repellent outdoors, wear permethrin-treated clothing, and conduct thorough tick checks after outdoor activities.
Once a person is no longer exposed to alpha-gal containing products, they should no longer experience symptoms. People with AGS should also proactively prevent tick bites. Tick bites can trigger or reactivate AGS.
For patients who have AGS, help manage their symptoms and identify alpha-gal containing products to avoid.
Dr. Kersh is Chief of the Rickettsial Zoonoses Branch, Division of Vector-Borne Diseases, Centers for Disease Control and Prevention, Atlanta, Georgia, and disclosed no relevant conflicts of interest.
CDC resources:
About Alpha-gal Syndrome | Alpha-gal Syndrome | CDC
Clinical Testing and Diagnosis for Alpha-gal Syndrome | Alpha-gal Syndrome | CDC
Clinical Resources | Alpha-gal Syndrome | CDC
References
Thompson JM et al. MMWR Morb Mortal Wkly Rep. 2023;72:815-820.
Carpenter A et al. MMWR Morb Mortal Wkly Rep. 2023;72:809-814. Taylor ML et al. Ann Allergy, Asthma & Immunol. 2024 Jun;132(6):759.e2-764.e2. Kersh GJ et al. Ann Allergy, Asthma & Immunol. 2023 Apr;130(4):472-478.
Alpha-gal syndrome (AGS), a tickborne disease commonly called “red meat allergy,” is a serious, potentially life-threatening allergy to the carbohydrate alpha-gal. The alpha-gal carbohydrate is found in most mammals, though it is not in humans, apes, or old-world monkeys. People with AGS can have allergic reactions when they consume mammalian meat, dairy products, or other products derived from mammals. People often live with this disease for years before receiving a correct diagnosis, greatly impacting their quality of life. The number of suspected cases is also rising.
More than 110,000 suspected AGS cases were identified between 2010 and 2022, according to a Centers for Disease Control and Prevention (CDC) report.1 However, because the diagnosis requires a positive test and a clinical exam and some people may not get tested, as many as 450,000 people might be affected by AGS in the United States. Additionally, a CDC survey found that nearly half (42%) of US healthcare providers had never heard of AGS.2 Among those who had, less than one third (29%) knew how to diagnose the condition.
Here are 5 things clinicians need to know about AGS.
1. People can develop AGS after being bitten by a tick, primarily the lone star tick (Amblyomma americanum), in the United States.
In the United States, AGS is primarily associated with the bite of a lone star tick, but other kinds of ticks have not been ruled out. The majority of suspected AGS cases in the United States were reported in parts of Arkansas, Delaware, Illinois, Indiana, Kansas, Kentucky, Maryland, Mississippi, Missouri, North Carolina, Oklahoma, Tennessee, and Virginia. The lone star tick is widely distributed with established populations in Alabama, Arkansas, Connecticut, Delaware, Florida, Georgia, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana, Michigan, Minnesota, Mississippi, Missouri, Nebraska, New Hampshire, New Jersey, New York, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Texas, Virginia, and West Virginia.
While AGS is associated with tick bites, more research is needed to understand the role ticks play in starting this condition, and why certain people develop AGS. Anyone can develop AGS, but most cases have been reported in adults.
Know how to recognize the symptoms of AGS and be prepared to test, diagnose, and manage AGS, particularly in states where lone star ticks are found.
2. Tick bites are only one risk factor for developing AGS.
Many people are bitten by lone star ticks and will never develop AGS. Scientists are exploring the connection between other risk factors and developing AGS. A recent study has shown that people diagnosed with AGS may be more likely to have a family member who was also diagnosed with AGS, have another food allergy, have an allergy to stinging or biting insects, or have A or O blood types.3
Research has also shown that environmental risk factors could contribute to developing AGS,4 like living in an area with lone star ticks, remembering finding a tick on themselves, recalling multiple tick bites, living near a wooded forest, spending more time outside, or living in areas with deer, such as larger properties, wooded forests, and properties with shrubs and brush.
Ask your patient questions about other allergies and history of recent tick bites or outdoor exposure to help determine if testing for AGS is appropriate.
3. Symptoms of AGS are consistently inconsistent.
There is a spectrum of how sensitive AGS patients are to alpha-gal, and reactions are often different from person to person, which can make it difficult to diagnose. The first allergic reaction to AGS typically occurs between 1-6 months after a tick bite. Symptoms commonly appear 2-6 hours after being in contact with products containing alpha-gal, like red meat (beef, pork, lamb, venison, rabbit, or other meat from mammals), dairy, and some medications. Symptoms can range from mild to severe and include hives or itchy rash; swelling of the lips, throat, tongue, or eyelids; gastrointestinal symptoms such as nausea, vomiting, or diarrhea; heartburn or indigestion; cough, shortness of breath, or difficulty breathing; dizziness or a drop in blood pressure; or anaphylaxis.
Consider AGS if a patient reports waking up in the middle of the night with allergic symptoms after eating alpha-gal containing products for dinner, if allergic reactions are delayed, or if a patient has anaphylaxis of unknown cause, adult-onset allergy, or allergic symptoms and reports a recent tick bite.
4. Diagnosing AGS requires a combination of a blood test and a physical exam.
Diagnosing AGS requires a detailed patient history, physical exam, and a blood test to detect specific immunoglobulin E (IgE) antibodies specific to alpha-gal (alpha-gal sIgE). Tests for alpha-gal sIgE antibodies are available at several large commercial laboratories and some academic institutions. Skin tests to identify reactions to allergens like pork or beef may also be used to inform AGS diagnosis. However, a positive alpha-gal sIgE test or skin test does not mean a person has AGS. Many people, particularly those who live in regions with lone star ticks, have positive alpha-gal specific IgE tests without having AGS.
Consider the test results along with your patient’s symptoms and risk factors.
5. There is no treatment for AGS, but people can take prevention steps and AGS can be managed.
People can protect themselves and their family from AGS by preventing tick bites. Encourage your patients to use an Environmental Protection Agency–registered insect repellent outdoors, wear permethrin-treated clothing, and conduct thorough tick checks after outdoor activities.
Once a person is no longer exposed to alpha-gal containing products, they should no longer experience symptoms. People with AGS should also proactively prevent tick bites. Tick bites can trigger or reactivate AGS.
For patients who have AGS, help manage their symptoms and identify alpha-gal containing products to avoid.
Dr. Kersh is Chief of the Rickettsial Zoonoses Branch, Division of Vector-Borne Diseases, Centers for Disease Control and Prevention, Atlanta, Georgia, and disclosed no relevant conflicts of interest.
CDC resources:
About Alpha-gal Syndrome | Alpha-gal Syndrome | CDC
Clinical Testing and Diagnosis for Alpha-gal Syndrome | Alpha-gal Syndrome | CDC
Clinical Resources | Alpha-gal Syndrome | CDC
References
Thompson JM et al. MMWR Morb Mortal Wkly Rep. 2023;72:815-820.
Carpenter A et al. MMWR Morb Mortal Wkly Rep. 2023;72:809-814. Taylor ML et al. Ann Allergy, Asthma & Immunol. 2024 Jun;132(6):759.e2-764.e2. Kersh GJ et al. Ann Allergy, Asthma & Immunol. 2023 Apr;130(4):472-478.
Alpha-gal syndrome (AGS), a tickborne disease commonly called “red meat allergy,” is a serious, potentially life-threatening allergy to the carbohydrate alpha-gal. The alpha-gal carbohydrate is found in most mammals, though it is not in humans, apes, or old-world monkeys. People with AGS can have allergic reactions when they consume mammalian meat, dairy products, or other products derived from mammals. People often live with this disease for years before receiving a correct diagnosis, greatly impacting their quality of life. The number of suspected cases is also rising.
More than 110,000 suspected AGS cases were identified between 2010 and 2022, according to a Centers for Disease Control and Prevention (CDC) report.1 However, because the diagnosis requires a positive test and a clinical exam and some people may not get tested, as many as 450,000 people might be affected by AGS in the United States. Additionally, a CDC survey found that nearly half (42%) of US healthcare providers had never heard of AGS.2 Among those who had, less than one third (29%) knew how to diagnose the condition.
Here are 5 things clinicians need to know about AGS.
1. People can develop AGS after being bitten by a tick, primarily the lone star tick (Amblyomma americanum), in the United States.
In the United States, AGS is primarily associated with the bite of a lone star tick, but other kinds of ticks have not been ruled out. The majority of suspected AGS cases in the United States were reported in parts of Arkansas, Delaware, Illinois, Indiana, Kansas, Kentucky, Maryland, Mississippi, Missouri, North Carolina, Oklahoma, Tennessee, and Virginia. The lone star tick is widely distributed with established populations in Alabama, Arkansas, Connecticut, Delaware, Florida, Georgia, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana, Michigan, Minnesota, Mississippi, Missouri, Nebraska, New Hampshire, New Jersey, New York, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Texas, Virginia, and West Virginia.
While AGS is associated with tick bites, more research is needed to understand the role ticks play in starting this condition, and why certain people develop AGS. Anyone can develop AGS, but most cases have been reported in adults.
Know how to recognize the symptoms of AGS and be prepared to test, diagnose, and manage AGS, particularly in states where lone star ticks are found.
2. Tick bites are only one risk factor for developing AGS.
Many people are bitten by lone star ticks and will never develop AGS. Scientists are exploring the connection between other risk factors and developing AGS. A recent study has shown that people diagnosed with AGS may be more likely to have a family member who was also diagnosed with AGS, have another food allergy, have an allergy to stinging or biting insects, or have A or O blood types.3
Research has also shown that environmental risk factors could contribute to developing AGS,4 like living in an area with lone star ticks, remembering finding a tick on themselves, recalling multiple tick bites, living near a wooded forest, spending more time outside, or living in areas with deer, such as larger properties, wooded forests, and properties with shrubs and brush.
Ask your patient questions about other allergies and history of recent tick bites or outdoor exposure to help determine if testing for AGS is appropriate.
3. Symptoms of AGS are consistently inconsistent.
There is a spectrum of how sensitive AGS patients are to alpha-gal, and reactions are often different from person to person, which can make it difficult to diagnose. The first allergic reaction to AGS typically occurs between 1-6 months after a tick bite. Symptoms commonly appear 2-6 hours after being in contact with products containing alpha-gal, like red meat (beef, pork, lamb, venison, rabbit, or other meat from mammals), dairy, and some medications. Symptoms can range from mild to severe and include hives or itchy rash; swelling of the lips, throat, tongue, or eyelids; gastrointestinal symptoms such as nausea, vomiting, or diarrhea; heartburn or indigestion; cough, shortness of breath, or difficulty breathing; dizziness or a drop in blood pressure; or anaphylaxis.
Consider AGS if a patient reports waking up in the middle of the night with allergic symptoms after eating alpha-gal containing products for dinner, if allergic reactions are delayed, or if a patient has anaphylaxis of unknown cause, adult-onset allergy, or allergic symptoms and reports a recent tick bite.
4. Diagnosing AGS requires a combination of a blood test and a physical exam.
Diagnosing AGS requires a detailed patient history, physical exam, and a blood test to detect specific immunoglobulin E (IgE) antibodies specific to alpha-gal (alpha-gal sIgE). Tests for alpha-gal sIgE antibodies are available at several large commercial laboratories and some academic institutions. Skin tests to identify reactions to allergens like pork or beef may also be used to inform AGS diagnosis. However, a positive alpha-gal sIgE test or skin test does not mean a person has AGS. Many people, particularly those who live in regions with lone star ticks, have positive alpha-gal specific IgE tests without having AGS.
Consider the test results along with your patient’s symptoms and risk factors.
5. There is no treatment for AGS, but people can take prevention steps and AGS can be managed.
People can protect themselves and their family from AGS by preventing tick bites. Encourage your patients to use an Environmental Protection Agency–registered insect repellent outdoors, wear permethrin-treated clothing, and conduct thorough tick checks after outdoor activities.
Once a person is no longer exposed to alpha-gal containing products, they should no longer experience symptoms. People with AGS should also proactively prevent tick bites. Tick bites can trigger or reactivate AGS.
For patients who have AGS, help manage their symptoms and identify alpha-gal containing products to avoid.
Dr. Kersh is Chief of the Rickettsial Zoonoses Branch, Division of Vector-Borne Diseases, Centers for Disease Control and Prevention, Atlanta, Georgia, and disclosed no relevant conflicts of interest.
CDC resources:
About Alpha-gal Syndrome | Alpha-gal Syndrome | CDC
Clinical Testing and Diagnosis for Alpha-gal Syndrome | Alpha-gal Syndrome | CDC
Clinical Resources | Alpha-gal Syndrome | CDC
References
Thompson JM et al. MMWR Morb Mortal Wkly Rep. 2023;72:815-820.
Carpenter A et al. MMWR Morb Mortal Wkly Rep. 2023;72:809-814. Taylor ML et al. Ann Allergy, Asthma & Immunol. 2024 Jun;132(6):759.e2-764.e2. Kersh GJ et al. Ann Allergy, Asthma & Immunol. 2023 Apr;130(4):472-478.
