User login
Outcomes From the Use of Cefazolin for Surgical Prophylaxis in Patients Allergic to Penicillin
Outcomes From the Use of Cefazolin for Surgical Prophylaxis in Patients Allergic to Penicillin
Given its safety profile and bactericidal activity against the predominant organisms causing surgical site infections (SSIs), cefazolin remains the most popular choice for surgical prophylaxis.1 Cefazolin offers protection against the pathogens most likely to contaminate the surgical site while minimizing inappropriate methicillin- resistant Staphylococcus aureus coverage that occurs with alternatives such as vancomycin and clindamycin. Documented allergies to Β-lactam antibiotics have historically forced clinicians to avoid the use of cephalosporins due to the potential risk of cross-reactivity. True type 1 (immunoglobin E [IgE]-mediated) cross-allergic reactions between penicillin and cephalosporins are rare, and previously reported data indicate cross-reactivity as a result of antibody recognition is more closely related to the side-chain identity rather than the Β-lactam ring.2,3
About 10% of US patients report having a penicillin allergy; however, < 1% of the population has a true IgE-mediated allergic reaction.4 Previous research that has challenged penicillin allergies with cefazolin for surgical prophylaxis has reported minimal rates of allergic reactions.2-5
In previous trials, patients with a history of delayed skin reactions, such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), were excluded. Additionally, patients with an allergy to cefazolin including those with urticaria, angioedema, bronchospasm, or anaphylaxis, were excluded from perioperative retrial of cefazolin. Grant et al found that cefazolin can be safely given to patients with IgE-mediated reactions to penicillin and other cephalosporins due to a structurally different side chain.3
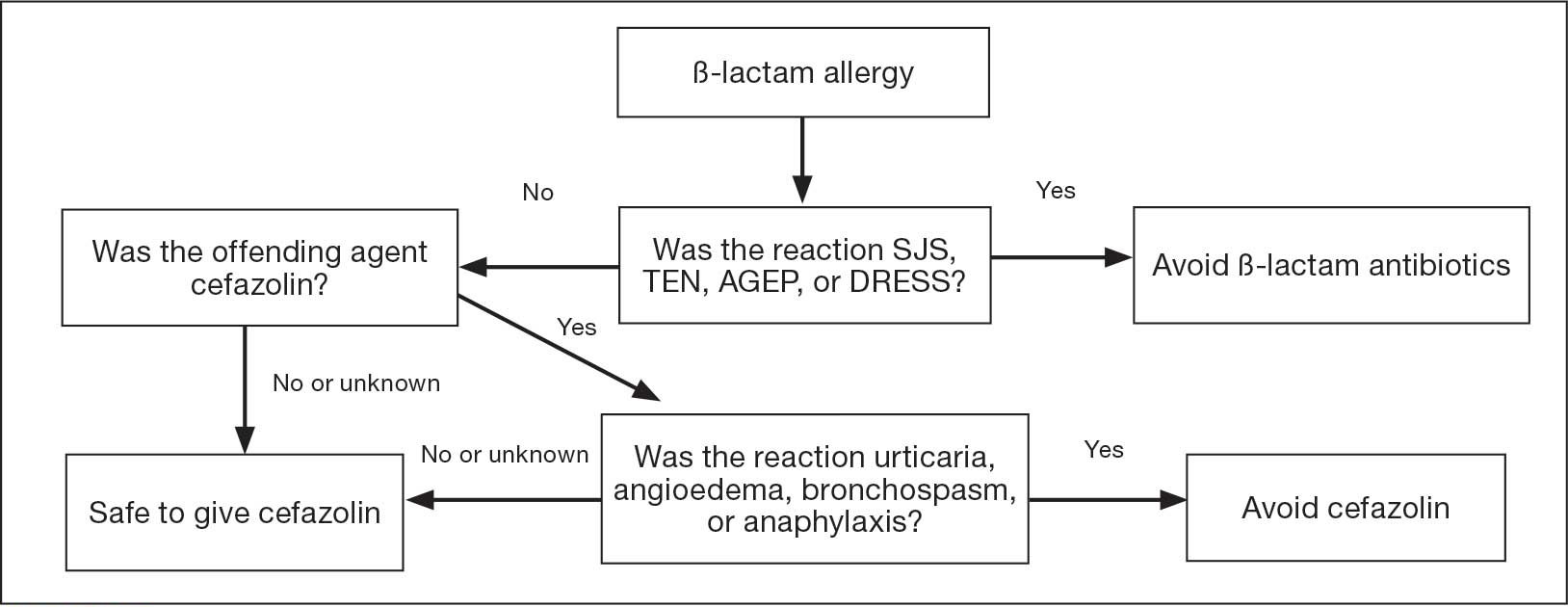
In January 2023, the Veteran Health Indiana (VHI) pharmacy team in conjunction with surgery, infectious disease, and anesthesiology, implemented a screening tool as an amendment to perioperative antibiotic guidance to help determine which patients with a documented penicillin allergy could be candidates for perioperative cefazolin. The implemented screening tool (Allergy Clarification for Cefazolin Evidence-Based Prescribing Tool) has been described by Lam et al, who reported that an increased proportion of patients with documented penicillin allergy received cefazolin without more adverse drug reactions (ADRs).5 Patients with a Β-lactam allergy were eligible to receive cefazolin unless the ADR was SJS, TEN, or DRESS, or the offending agent was cefazolin and the patient experienced urticaria, angioedema, bronchospasm, or anaphylaxis. If the reaction was not from cefazolin or was unknown, patients were eligible to receive cefazolin (Figure).
To date, minimal data exist to evaluate the incidence of ADRs when cefazolin is given perioperatively to patients with a previously documented penicillin allergy. The purpose of this study was to evaluate the incidence of allergic ADRs in patients who had a documented penicillin allergy and received periprocedural antibiotics.
Methods
This single-center, retrospective chart review used the US Department of Veterans Affairs (VA) Computerized Patient Record System (CPRS) to identify patients with a documented penicillin allergy who underwent an operation and received periprocedural antibiotics between February 1, 2023, and January 31, 2024. This study was reviewed and approved by the Indiana University Health Institutional Review Board and the VHI Research and Development Committee.
Patients were enrolled if they were aged ≥ 18 years, had a documented penicillin allergy, underwent a surgical intervention, and received perioperative antibiotics during the study period. Patients were excluded if they had a documented penicillin allergy resulting in severe delayed skin reactions (ie, SJS, TEN, or DRESS). These criteria produced 197 surgical procedures. Data were collected for each surgical procedure, so patients could be included more than once. Patient history of allergic reaction to penicillin was obtained through CPRS.
The primary endpoint was the percentage of allergic ADRs in patients with penicillin allergies receiving cefazolin perioperatively. Secondary outcomes included the appropriateness of the antibiotic regimen in congruence with American System of Health Pharmacists (ASHP) recommendations, incidence of SSIs within 30 days of the procedure, incidence of ADRs in those with a history of anaphylaxis vs nonanaphylaxis allergy, incidence of allergic reaction requiring pharmacologic and nonpharmacologic interventions, and incidence of acute kidney injury (AKI). AKI was defined as an increase in serum creatinine by ≥ 0.3 mg/dL within 48 hours or an increase in serum creatinine to ≥ 1.5 times baseline.
Demographic data included sex, age, race, preoperative serum creatinine, and postoperative serum creatinine. Anaphylaxis was defined as an acute onset of illness (within minutes to several hours) with involvement of skin, mucosal tissue, or both involving either respiratory compromise or reduced blood pressures. Allergic reactions were defined as facial, tongue, throat, airway, lip, mouth, periorbital, or eye swelling, urticaria, angioedema, dyspnea, anaphylaxis, or a positive penicillin skin test. Additionally, data collected included the description and severity of postprophylactic antibiotic reaction, antibiotic choice, interventions required for the allergic reaction, SSI occurrence, date of SSI, operating specialty, and postoperative change in renal function.
Descriptive statistics, including mean, SD, and percentages were reported for baseline characteristics of the study population. Percentages were used to demonstrate the differences in primary and secondary outcomes for each study group. Fisher exact tests were used for incidence of ADRs in patients with penicillin allergy who received cefazolin and reported incidence of SSIs.
Results
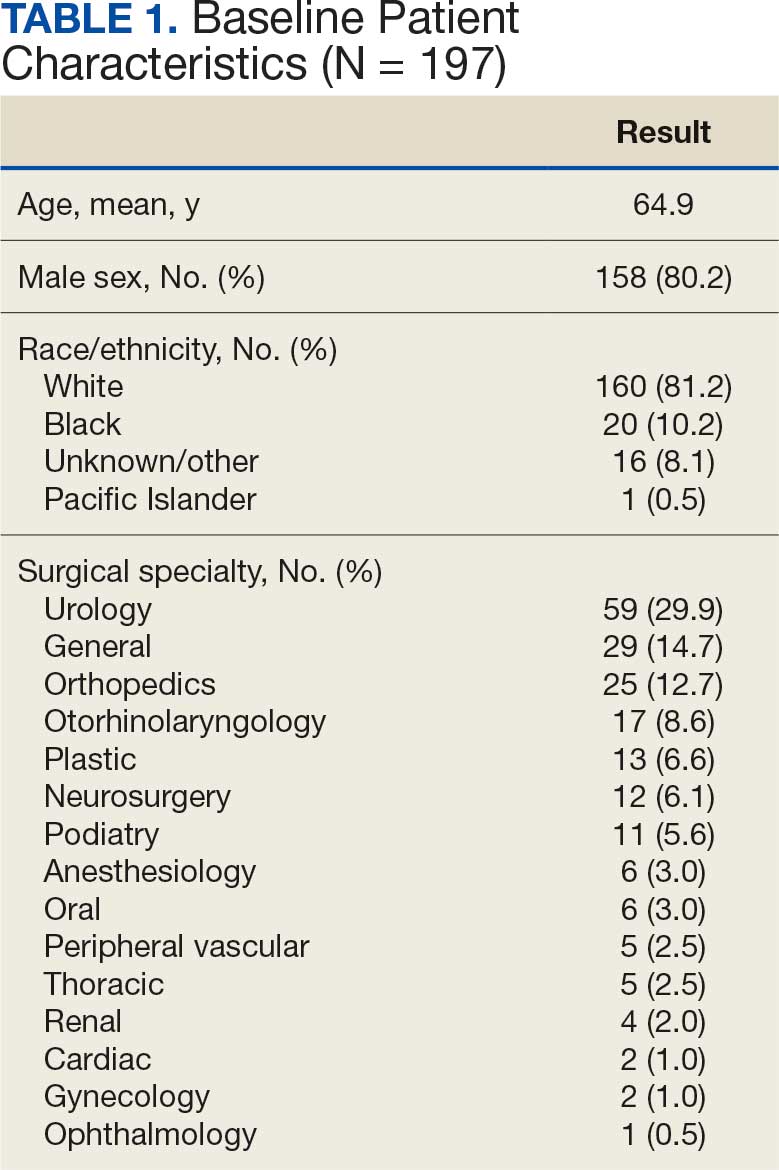
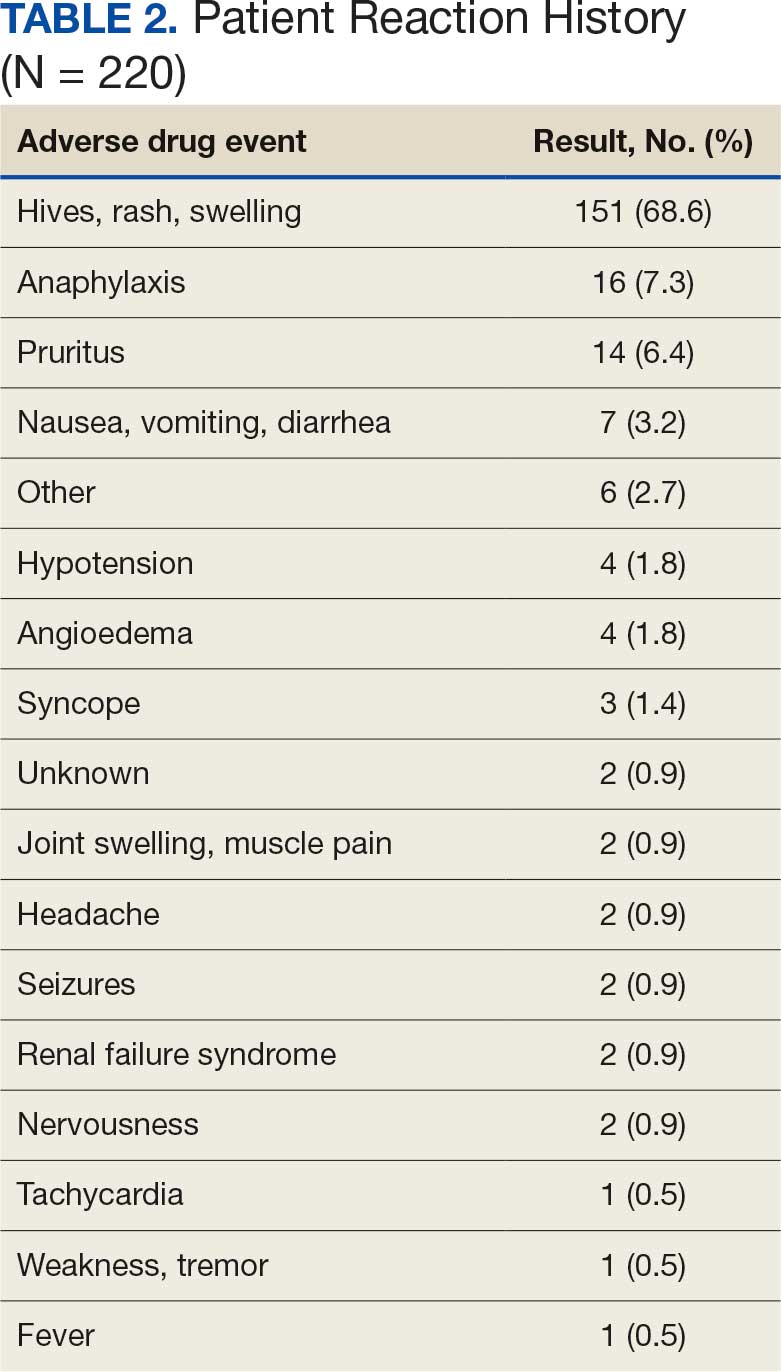
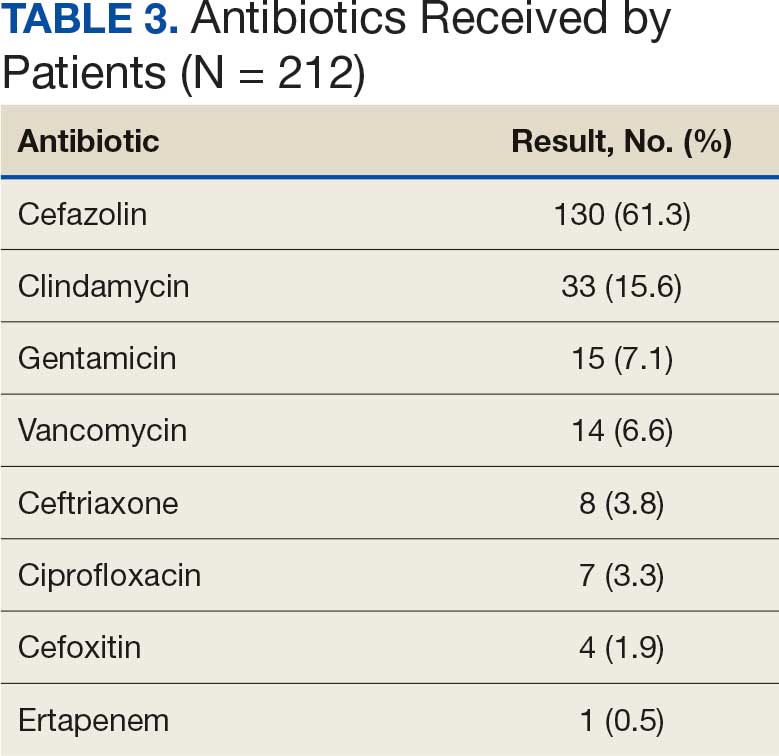
A total of 197 surgical procedures in patients with a documented penicillin allergy were included; 127 procedures used cefazolin perioperatively, 3 procedures used cefazolin plus gentamicin, and 67 procedures used other antibiotics. Most patients were White (n = 160; 81.2%), male (n = 158; 80.2%), and had a mean age of 64.9 years. Urology was the most common surgical specialty (n = 59; 29.9%) (Table 1). Of the 16 patients with documented penicillin anaphylaxis reaction, 8 received cefazolin and 8 received a different antibiotic. A total of 181 patients reported a nonanaphylaxis allergy. One hundred fifty-one patients (68.6%) reported a reaction history of hives, rash, or swelling (Table 2). Patients could report ≥ 1 reaction. The most prevalent antibiotics used were cefazolin, which was used by 130 patients (61.3%), and clindamycin which was used by 33 patients (15.6%) (Table 3). Patients could receive ≥ 1 antibiotic.
For the primary outcome, the incidence of allergic reactions in patients allergic to penicillin, there was no incidence of allergic reactions in either the cefazolin or other group. Given the absence of reactions, no interventions were required.
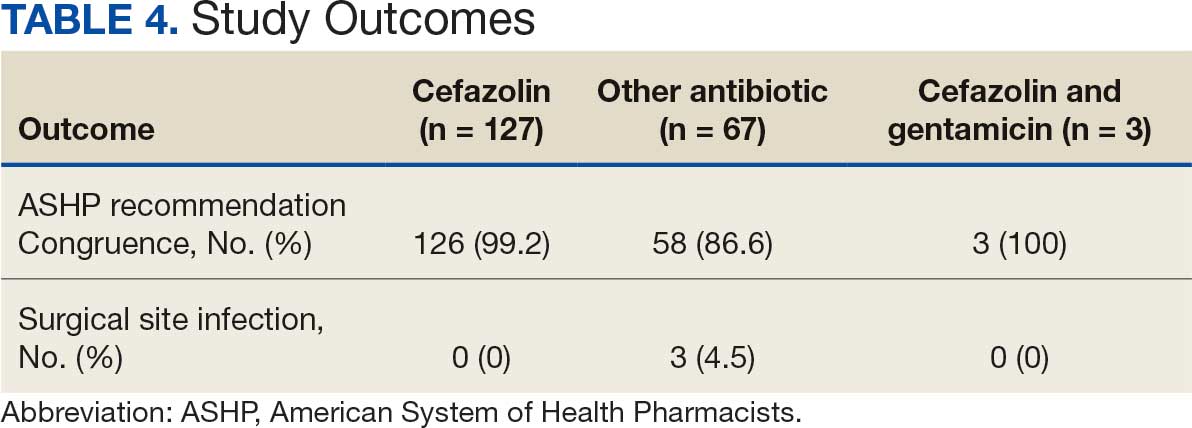
There were no ADRs in those with history of anaphylaxis or nonanaphylaxis allergy. In the cefazolin group, 126 of 127 surgical procedure regimens (99.2%) were congruent with ASHP recommendations, all 3 surgical procedures regimens in the cefazolin plus gentamicin group were congruent with ASHP recommendations, and 58 of 67 surgical procedure regimens (86.6%) in the other antibiotic group were congruent with ASHP recommendations. None of the 127 patients in the cefazolin group or of the 3 patients in the cefazolin plus gentamicin group reported an SSI, and 3 of 67 patients (4.5%) had an SSI in the other antibiotic group. One procedure that resulted in SSI was not congruent with ASHP recommendations. Twenty-four patients had 2 serum creatinine levels drawn within 48 hours of surgery. One of 12 patients (8.3%) and 0 of 12 patients had an AKI in the cefazolin and other antibiotic group, respectively (Table 4).
Discussion
Implementation of a screening tool at VHI allowed patients with documented penicillin allergy, including anaphylaxis, to receive cefazolin perioperatively. Broad spectrum antibiotics such as vancomycin, clindamycin, and fluoroquinolones are frequently used in patients allergic to penicillin, which can increase health care costs, risk of toxicity, and antimicrobial resistance.4 There was no incidence of allergic reactions noted in patients allergic to penicillin who received cefazolin. When comparing the incidence of observed allergic reactions to received perioperative antibiotics in the cefazolin group to previously published literature, no difference in allergy rates (P = .09) was found.3 Most antibiotics administered were congruent with ASHP guideline recommendations, and most patients eligible for cefazolin received it perioperatively.
Similar to this study, Goodman et al concluded that cefazolin appears to be a safe regimen in patients with documented penicillin anaphylactic reaction for surgical prophylaxis with only 1 (0.2%) potential allergic reaction.6 Patients who received cefazolin perioperatively had a statistically significant decrease in SSI rates. There were no clinically or statistically significant differences found between the proportion of allergic reactions or ADRs when compared to alternative antibiotics. Lessard et al concluded that a pharmacist-led interdisciplinary collaborative practice agreement increased cefazolin use in patients allergic to penicillin, including those with urticaria and anaphylaxis, with no reported ADRs.7 This study further demonstrated the safety of cefazolin use in patients with anaphylaxis to penicillin.
Limitations
This study’s single-center, retrospective design, patient population, and small sample size limit the generalizability of its results. The data collected are dependent on documentation in the chart. No ADRs were reported from the antibiotics patients received perioperatively. When considering safety data, information such as serum creatinine were available only in CPRS and some patients did not receive a postprocedure serum creatinine level. Additionally, this study did not investigate whether there was an increase in preferred preoperative antimicrobial prophylaxis after implementation of this protocol.
Conclusions
The results of this study support the use of cefazolin perioperatively in patients allergic to penicillin, including those with a history of anaphylaxis. Additional research should be conducted to validate data given the low incidence of ADRs. The primary outcome did not reach statistical significance, but the results may be clinically significant from a stewardship and safety perspective. VHI continues to use the screening tool described in this article.
- Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm. 2013;70:195-283. doi:10.2146/ajhp120568
- Romano A, Valluzzi RL, Caruso C, et al. Tolerability of cefazolin and ceftibuten in patients with IgE-mediated aminopenicillin allergy. J Allergy Clin Immunol Pract. 2020;8:1989-1993.e2. doi:10.1016/j.jaip.2020.02.025
- Grant JM, Song WHC, Shajari S, et al. Safety of administering cefazolin versus other antibiotics in penicillin- allergic patients for surgical prophylaxis at a major Canadian teaching hospital. Surgery. 2021;170:783-789. doi:10.1016/j.surg.2021.03.022
- Centers for Disease Control and Prevention. Clinical Features of Penicillin Allergy. August 25, 2025. Accessed January 6, 2026. https://www.cdc.gov/antibiotic-use/hcp/clinical-signs/index.html
- Lam PW, Tarighi P, Elligsen M, et al. Impact of the allergy clarification for cefazolin evidence-based prescribing tool on receipt of preferred perioperative prophylaxis: an interrupted time series study. Clin Infect Dis. 2020;71:2955- 2957. doi:10.1093/cid/ciaa516
- Goodman EJ, Morgan MJ, Johnson Pa, et al. Cephalosporins can be given to penicillin-allergic patients who do not exhibit an anaphylactic response. J Clin Anesth. 2001;13:561-564. doi:10.1016/s0952-8180(01)00329-4
- Lessard S, Huiras C, Dababneh A, et al. Pharmacist adjustment of preoperative antibiotic orders to the preferred preoperative antibiotic cefazolin for patients with penicillin allergy labeling. Am J Health Syst Pharm. 2023;80:532- 536. doi:10.1093/ajhp/zxac385
Given its safety profile and bactericidal activity against the predominant organisms causing surgical site infections (SSIs), cefazolin remains the most popular choice for surgical prophylaxis.1 Cefazolin offers protection against the pathogens most likely to contaminate the surgical site while minimizing inappropriate methicillin- resistant Staphylococcus aureus coverage that occurs with alternatives such as vancomycin and clindamycin. Documented allergies to Β-lactam antibiotics have historically forced clinicians to avoid the use of cephalosporins due to the potential risk of cross-reactivity. True type 1 (immunoglobin E [IgE]-mediated) cross-allergic reactions between penicillin and cephalosporins are rare, and previously reported data indicate cross-reactivity as a result of antibody recognition is more closely related to the side-chain identity rather than the Β-lactam ring.2,3
About 10% of US patients report having a penicillin allergy; however, < 1% of the population has a true IgE-mediated allergic reaction.4 Previous research that has challenged penicillin allergies with cefazolin for surgical prophylaxis has reported minimal rates of allergic reactions.2-5
In previous trials, patients with a history of delayed skin reactions, such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), were excluded. Additionally, patients with an allergy to cefazolin including those with urticaria, angioedema, bronchospasm, or anaphylaxis, were excluded from perioperative retrial of cefazolin. Grant et al found that cefazolin can be safely given to patients with IgE-mediated reactions to penicillin and other cephalosporins due to a structurally different side chain.3
In January 2023, the Veteran Health Indiana (VHI) pharmacy team in conjunction with surgery, infectious disease, and anesthesiology, implemented a screening tool as an amendment to perioperative antibiotic guidance to help determine which patients with a documented penicillin allergy could be candidates for perioperative cefazolin. The implemented screening tool (Allergy Clarification for Cefazolin Evidence-Based Prescribing Tool) has been described by Lam et al, who reported that an increased proportion of patients with documented penicillin allergy received cefazolin without more adverse drug reactions (ADRs).5 Patients with a Β-lactam allergy were eligible to receive cefazolin unless the ADR was SJS, TEN, or DRESS, or the offending agent was cefazolin and the patient experienced urticaria, angioedema, bronchospasm, or anaphylaxis. If the reaction was not from cefazolin or was unknown, patients were eligible to receive cefazolin (Figure).

To date, minimal data exist to evaluate the incidence of ADRs when cefazolin is given perioperatively to patients with a previously documented penicillin allergy. The purpose of this study was to evaluate the incidence of allergic ADRs in patients who had a documented penicillin allergy and received periprocedural antibiotics.
Methods
This single-center, retrospective chart review used the US Department of Veterans Affairs (VA) Computerized Patient Record System (CPRS) to identify patients with a documented penicillin allergy who underwent an operation and received periprocedural antibiotics between February 1, 2023, and January 31, 2024. This study was reviewed and approved by the Indiana University Health Institutional Review Board and the VHI Research and Development Committee.
Patients were enrolled if they were aged ≥ 18 years, had a documented penicillin allergy, underwent a surgical intervention, and received perioperative antibiotics during the study period. Patients were excluded if they had a documented penicillin allergy resulting in severe delayed skin reactions (ie, SJS, TEN, or DRESS). These criteria produced 197 surgical procedures. Data were collected for each surgical procedure, so patients could be included more than once. Patient history of allergic reaction to penicillin was obtained through CPRS.
The primary endpoint was the percentage of allergic ADRs in patients with penicillin allergies receiving cefazolin perioperatively. Secondary outcomes included the appropriateness of the antibiotic regimen in congruence with American System of Health Pharmacists (ASHP) recommendations, incidence of SSIs within 30 days of the procedure, incidence of ADRs in those with a history of anaphylaxis vs nonanaphylaxis allergy, incidence of allergic reaction requiring pharmacologic and nonpharmacologic interventions, and incidence of acute kidney injury (AKI). AKI was defined as an increase in serum creatinine by ≥ 0.3 mg/dL within 48 hours or an increase in serum creatinine to ≥ 1.5 times baseline.
Demographic data included sex, age, race, preoperative serum creatinine, and postoperative serum creatinine. Anaphylaxis was defined as an acute onset of illness (within minutes to several hours) with involvement of skin, mucosal tissue, or both involving either respiratory compromise or reduced blood pressures. Allergic reactions were defined as facial, tongue, throat, airway, lip, mouth, periorbital, or eye swelling, urticaria, angioedema, dyspnea, anaphylaxis, or a positive penicillin skin test. Additionally, data collected included the description and severity of postprophylactic antibiotic reaction, antibiotic choice, interventions required for the allergic reaction, SSI occurrence, date of SSI, operating specialty, and postoperative change in renal function.
Descriptive statistics, including mean, SD, and percentages were reported for baseline characteristics of the study population. Percentages were used to demonstrate the differences in primary and secondary outcomes for each study group. Fisher exact tests were used for incidence of ADRs in patients with penicillin allergy who received cefazolin and reported incidence of SSIs.
Results
A total of 197 surgical procedures in patients with a documented penicillin allergy were included; 127 procedures used cefazolin perioperatively, 3 procedures used cefazolin plus gentamicin, and 67 procedures used other antibiotics. Most patients were White (n = 160; 81.2%), male (n = 158; 80.2%), and had a mean age of 64.9 years. Urology was the most common surgical specialty (n = 59; 29.9%) (Table 1). Of the 16 patients with documented penicillin anaphylaxis reaction, 8 received cefazolin and 8 received a different antibiotic. A total of 181 patients reported a nonanaphylaxis allergy. One hundred fifty-one patients (68.6%) reported a reaction history of hives, rash, or swelling (Table 2). Patients could report ≥ 1 reaction. The most prevalent antibiotics used were cefazolin, which was used by 130 patients (61.3%), and clindamycin which was used by 33 patients (15.6%) (Table 3). Patients could receive ≥ 1 antibiotic.



For the primary outcome, the incidence of allergic reactions in patients allergic to penicillin, there was no incidence of allergic reactions in either the cefazolin or other group. Given the absence of reactions, no interventions were required.
There were no ADRs in those with history of anaphylaxis or nonanaphylaxis allergy. In the cefazolin group, 126 of 127 surgical procedure regimens (99.2%) were congruent with ASHP recommendations, all 3 surgical procedures regimens in the cefazolin plus gentamicin group were congruent with ASHP recommendations, and 58 of 67 surgical procedure regimens (86.6%) in the other antibiotic group were congruent with ASHP recommendations. None of the 127 patients in the cefazolin group or of the 3 patients in the cefazolin plus gentamicin group reported an SSI, and 3 of 67 patients (4.5%) had an SSI in the other antibiotic group. One procedure that resulted in SSI was not congruent with ASHP recommendations. Twenty-four patients had 2 serum creatinine levels drawn within 48 hours of surgery. One of 12 patients (8.3%) and 0 of 12 patients had an AKI in the cefazolin and other antibiotic group, respectively (Table 4).

Discussion
Implementation of a screening tool at VHI allowed patients with documented penicillin allergy, including anaphylaxis, to receive cefazolin perioperatively. Broad spectrum antibiotics such as vancomycin, clindamycin, and fluoroquinolones are frequently used in patients allergic to penicillin, which can increase health care costs, risk of toxicity, and antimicrobial resistance.4 There was no incidence of allergic reactions noted in patients allergic to penicillin who received cefazolin. When comparing the incidence of observed allergic reactions to received perioperative antibiotics in the cefazolin group to previously published literature, no difference in allergy rates (P = .09) was found.3 Most antibiotics administered were congruent with ASHP guideline recommendations, and most patients eligible for cefazolin received it perioperatively.
Similar to this study, Goodman et al concluded that cefazolin appears to be a safe regimen in patients with documented penicillin anaphylactic reaction for surgical prophylaxis with only 1 (0.2%) potential allergic reaction.6 Patients who received cefazolin perioperatively had a statistically significant decrease in SSI rates. There were no clinically or statistically significant differences found between the proportion of allergic reactions or ADRs when compared to alternative antibiotics. Lessard et al concluded that a pharmacist-led interdisciplinary collaborative practice agreement increased cefazolin use in patients allergic to penicillin, including those with urticaria and anaphylaxis, with no reported ADRs.7 This study further demonstrated the safety of cefazolin use in patients with anaphylaxis to penicillin.
Limitations
This study’s single-center, retrospective design, patient population, and small sample size limit the generalizability of its results. The data collected are dependent on documentation in the chart. No ADRs were reported from the antibiotics patients received perioperatively. When considering safety data, information such as serum creatinine were available only in CPRS and some patients did not receive a postprocedure serum creatinine level. Additionally, this study did not investigate whether there was an increase in preferred preoperative antimicrobial prophylaxis after implementation of this protocol.
Conclusions
The results of this study support the use of cefazolin perioperatively in patients allergic to penicillin, including those with a history of anaphylaxis. Additional research should be conducted to validate data given the low incidence of ADRs. The primary outcome did not reach statistical significance, but the results may be clinically significant from a stewardship and safety perspective. VHI continues to use the screening tool described in this article.
Given its safety profile and bactericidal activity against the predominant organisms causing surgical site infections (SSIs), cefazolin remains the most popular choice for surgical prophylaxis.1 Cefazolin offers protection against the pathogens most likely to contaminate the surgical site while minimizing inappropriate methicillin- resistant Staphylococcus aureus coverage that occurs with alternatives such as vancomycin and clindamycin. Documented allergies to Β-lactam antibiotics have historically forced clinicians to avoid the use of cephalosporins due to the potential risk of cross-reactivity. True type 1 (immunoglobin E [IgE]-mediated) cross-allergic reactions between penicillin and cephalosporins are rare, and previously reported data indicate cross-reactivity as a result of antibody recognition is more closely related to the side-chain identity rather than the Β-lactam ring.2,3
About 10% of US patients report having a penicillin allergy; however, < 1% of the population has a true IgE-mediated allergic reaction.4 Previous research that has challenged penicillin allergies with cefazolin for surgical prophylaxis has reported minimal rates of allergic reactions.2-5
In previous trials, patients with a history of delayed skin reactions, such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), were excluded. Additionally, patients with an allergy to cefazolin including those with urticaria, angioedema, bronchospasm, or anaphylaxis, were excluded from perioperative retrial of cefazolin. Grant et al found that cefazolin can be safely given to patients with IgE-mediated reactions to penicillin and other cephalosporins due to a structurally different side chain.3
In January 2023, the Veteran Health Indiana (VHI) pharmacy team in conjunction with surgery, infectious disease, and anesthesiology, implemented a screening tool as an amendment to perioperative antibiotic guidance to help determine which patients with a documented penicillin allergy could be candidates for perioperative cefazolin. The implemented screening tool (Allergy Clarification for Cefazolin Evidence-Based Prescribing Tool) has been described by Lam et al, who reported that an increased proportion of patients with documented penicillin allergy received cefazolin without more adverse drug reactions (ADRs).5 Patients with a Β-lactam allergy were eligible to receive cefazolin unless the ADR was SJS, TEN, or DRESS, or the offending agent was cefazolin and the patient experienced urticaria, angioedema, bronchospasm, or anaphylaxis. If the reaction was not from cefazolin or was unknown, patients were eligible to receive cefazolin (Figure).

To date, minimal data exist to evaluate the incidence of ADRs when cefazolin is given perioperatively to patients with a previously documented penicillin allergy. The purpose of this study was to evaluate the incidence of allergic ADRs in patients who had a documented penicillin allergy and received periprocedural antibiotics.
Methods
This single-center, retrospective chart review used the US Department of Veterans Affairs (VA) Computerized Patient Record System (CPRS) to identify patients with a documented penicillin allergy who underwent an operation and received periprocedural antibiotics between February 1, 2023, and January 31, 2024. This study was reviewed and approved by the Indiana University Health Institutional Review Board and the VHI Research and Development Committee.
Patients were enrolled if they were aged ≥ 18 years, had a documented penicillin allergy, underwent a surgical intervention, and received perioperative antibiotics during the study period. Patients were excluded if they had a documented penicillin allergy resulting in severe delayed skin reactions (ie, SJS, TEN, or DRESS). These criteria produced 197 surgical procedures. Data were collected for each surgical procedure, so patients could be included more than once. Patient history of allergic reaction to penicillin was obtained through CPRS.
The primary endpoint was the percentage of allergic ADRs in patients with penicillin allergies receiving cefazolin perioperatively. Secondary outcomes included the appropriateness of the antibiotic regimen in congruence with American System of Health Pharmacists (ASHP) recommendations, incidence of SSIs within 30 days of the procedure, incidence of ADRs in those with a history of anaphylaxis vs nonanaphylaxis allergy, incidence of allergic reaction requiring pharmacologic and nonpharmacologic interventions, and incidence of acute kidney injury (AKI). AKI was defined as an increase in serum creatinine by ≥ 0.3 mg/dL within 48 hours or an increase in serum creatinine to ≥ 1.5 times baseline.
Demographic data included sex, age, race, preoperative serum creatinine, and postoperative serum creatinine. Anaphylaxis was defined as an acute onset of illness (within minutes to several hours) with involvement of skin, mucosal tissue, or both involving either respiratory compromise or reduced blood pressures. Allergic reactions were defined as facial, tongue, throat, airway, lip, mouth, periorbital, or eye swelling, urticaria, angioedema, dyspnea, anaphylaxis, or a positive penicillin skin test. Additionally, data collected included the description and severity of postprophylactic antibiotic reaction, antibiotic choice, interventions required for the allergic reaction, SSI occurrence, date of SSI, operating specialty, and postoperative change in renal function.
Descriptive statistics, including mean, SD, and percentages were reported for baseline characteristics of the study population. Percentages were used to demonstrate the differences in primary and secondary outcomes for each study group. Fisher exact tests were used for incidence of ADRs in patients with penicillin allergy who received cefazolin and reported incidence of SSIs.
Results
A total of 197 surgical procedures in patients with a documented penicillin allergy were included; 127 procedures used cefazolin perioperatively, 3 procedures used cefazolin plus gentamicin, and 67 procedures used other antibiotics. Most patients were White (n = 160; 81.2%), male (n = 158; 80.2%), and had a mean age of 64.9 years. Urology was the most common surgical specialty (n = 59; 29.9%) (Table 1). Of the 16 patients with documented penicillin anaphylaxis reaction, 8 received cefazolin and 8 received a different antibiotic. A total of 181 patients reported a nonanaphylaxis allergy. One hundred fifty-one patients (68.6%) reported a reaction history of hives, rash, or swelling (Table 2). Patients could report ≥ 1 reaction. The most prevalent antibiotics used were cefazolin, which was used by 130 patients (61.3%), and clindamycin which was used by 33 patients (15.6%) (Table 3). Patients could receive ≥ 1 antibiotic.



For the primary outcome, the incidence of allergic reactions in patients allergic to penicillin, there was no incidence of allergic reactions in either the cefazolin or other group. Given the absence of reactions, no interventions were required.
There were no ADRs in those with history of anaphylaxis or nonanaphylaxis allergy. In the cefazolin group, 126 of 127 surgical procedure regimens (99.2%) were congruent with ASHP recommendations, all 3 surgical procedures regimens in the cefazolin plus gentamicin group were congruent with ASHP recommendations, and 58 of 67 surgical procedure regimens (86.6%) in the other antibiotic group were congruent with ASHP recommendations. None of the 127 patients in the cefazolin group or of the 3 patients in the cefazolin plus gentamicin group reported an SSI, and 3 of 67 patients (4.5%) had an SSI in the other antibiotic group. One procedure that resulted in SSI was not congruent with ASHP recommendations. Twenty-four patients had 2 serum creatinine levels drawn within 48 hours of surgery. One of 12 patients (8.3%) and 0 of 12 patients had an AKI in the cefazolin and other antibiotic group, respectively (Table 4).

Discussion
Implementation of a screening tool at VHI allowed patients with documented penicillin allergy, including anaphylaxis, to receive cefazolin perioperatively. Broad spectrum antibiotics such as vancomycin, clindamycin, and fluoroquinolones are frequently used in patients allergic to penicillin, which can increase health care costs, risk of toxicity, and antimicrobial resistance.4 There was no incidence of allergic reactions noted in patients allergic to penicillin who received cefazolin. When comparing the incidence of observed allergic reactions to received perioperative antibiotics in the cefazolin group to previously published literature, no difference in allergy rates (P = .09) was found.3 Most antibiotics administered were congruent with ASHP guideline recommendations, and most patients eligible for cefazolin received it perioperatively.
Similar to this study, Goodman et al concluded that cefazolin appears to be a safe regimen in patients with documented penicillin anaphylactic reaction for surgical prophylaxis with only 1 (0.2%) potential allergic reaction.6 Patients who received cefazolin perioperatively had a statistically significant decrease in SSI rates. There were no clinically or statistically significant differences found between the proportion of allergic reactions or ADRs when compared to alternative antibiotics. Lessard et al concluded that a pharmacist-led interdisciplinary collaborative practice agreement increased cefazolin use in patients allergic to penicillin, including those with urticaria and anaphylaxis, with no reported ADRs.7 This study further demonstrated the safety of cefazolin use in patients with anaphylaxis to penicillin.
Limitations
This study’s single-center, retrospective design, patient population, and small sample size limit the generalizability of its results. The data collected are dependent on documentation in the chart. No ADRs were reported from the antibiotics patients received perioperatively. When considering safety data, information such as serum creatinine were available only in CPRS and some patients did not receive a postprocedure serum creatinine level. Additionally, this study did not investigate whether there was an increase in preferred preoperative antimicrobial prophylaxis after implementation of this protocol.
Conclusions
The results of this study support the use of cefazolin perioperatively in patients allergic to penicillin, including those with a history of anaphylaxis. Additional research should be conducted to validate data given the low incidence of ADRs. The primary outcome did not reach statistical significance, but the results may be clinically significant from a stewardship and safety perspective. VHI continues to use the screening tool described in this article.
- Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm. 2013;70:195-283. doi:10.2146/ajhp120568
- Romano A, Valluzzi RL, Caruso C, et al. Tolerability of cefazolin and ceftibuten in patients with IgE-mediated aminopenicillin allergy. J Allergy Clin Immunol Pract. 2020;8:1989-1993.e2. doi:10.1016/j.jaip.2020.02.025
- Grant JM, Song WHC, Shajari S, et al. Safety of administering cefazolin versus other antibiotics in penicillin- allergic patients for surgical prophylaxis at a major Canadian teaching hospital. Surgery. 2021;170:783-789. doi:10.1016/j.surg.2021.03.022
- Centers for Disease Control and Prevention. Clinical Features of Penicillin Allergy. August 25, 2025. Accessed January 6, 2026. https://www.cdc.gov/antibiotic-use/hcp/clinical-signs/index.html
- Lam PW, Tarighi P, Elligsen M, et al. Impact of the allergy clarification for cefazolin evidence-based prescribing tool on receipt of preferred perioperative prophylaxis: an interrupted time series study. Clin Infect Dis. 2020;71:2955- 2957. doi:10.1093/cid/ciaa516
- Goodman EJ, Morgan MJ, Johnson Pa, et al. Cephalosporins can be given to penicillin-allergic patients who do not exhibit an anaphylactic response. J Clin Anesth. 2001;13:561-564. doi:10.1016/s0952-8180(01)00329-4
- Lessard S, Huiras C, Dababneh A, et al. Pharmacist adjustment of preoperative antibiotic orders to the preferred preoperative antibiotic cefazolin for patients with penicillin allergy labeling. Am J Health Syst Pharm. 2023;80:532- 536. doi:10.1093/ajhp/zxac385
- Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm. 2013;70:195-283. doi:10.2146/ajhp120568
- Romano A, Valluzzi RL, Caruso C, et al. Tolerability of cefazolin and ceftibuten in patients with IgE-mediated aminopenicillin allergy. J Allergy Clin Immunol Pract. 2020;8:1989-1993.e2. doi:10.1016/j.jaip.2020.02.025
- Grant JM, Song WHC, Shajari S, et al. Safety of administering cefazolin versus other antibiotics in penicillin- allergic patients for surgical prophylaxis at a major Canadian teaching hospital. Surgery. 2021;170:783-789. doi:10.1016/j.surg.2021.03.022
- Centers for Disease Control and Prevention. Clinical Features of Penicillin Allergy. August 25, 2025. Accessed January 6, 2026. https://www.cdc.gov/antibiotic-use/hcp/clinical-signs/index.html
- Lam PW, Tarighi P, Elligsen M, et al. Impact of the allergy clarification for cefazolin evidence-based prescribing tool on receipt of preferred perioperative prophylaxis: an interrupted time series study. Clin Infect Dis. 2020;71:2955- 2957. doi:10.1093/cid/ciaa516
- Goodman EJ, Morgan MJ, Johnson Pa, et al. Cephalosporins can be given to penicillin-allergic patients who do not exhibit an anaphylactic response. J Clin Anesth. 2001;13:561-564. doi:10.1016/s0952-8180(01)00329-4
- Lessard S, Huiras C, Dababneh A, et al. Pharmacist adjustment of preoperative antibiotic orders to the preferred preoperative antibiotic cefazolin for patients with penicillin allergy labeling. Am J Health Syst Pharm. 2023;80:532- 536. doi:10.1093/ajhp/zxac385
Outcomes From the Use of Cefazolin for Surgical Prophylaxis in Patients Allergic to Penicillin
Outcomes From the Use of Cefazolin for Surgical Prophylaxis in Patients Allergic to Penicillin