User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
Silent ischemia isn’t what it used to be
SNOWMASS, COLO. – The concept that silent myocardial ischemia is clinically detrimental has fallen by the wayside, and routine screening for this phenomenon can no longer be recommended, Patrick T. O’Gara, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
What a difference a decade or 2 can make.
“Think about where we were 25 years ago, when we worried about people who had transient ST-segment depression without angina on Holter monitoring. We would wig out, chase them down the street, try to tackle them and load them up with medications and think about balloon [percutaneous transluminal coronary angioplasty]. And now we’re at the point where it doesn’t seem to help with respect to quality of life, let alone death or myocardial infarction,” observed Dr. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, both in Boston.
The end of the line for the now-discredited notion that silent ischemia carries clinical significance approaching that of ischemia plus angina pectoris was the landmark ISCHEMIA trial, reported in November 2019 at the annual scientific sessions of the American Heart Association. This randomized trial asked the question: Is there any high-risk subgroup of patients with stable ischemic heart disease not involving the left main coronary artery for whom a strategy of routine revascularization improves hard outcomes in the current era of highly effective, guideline-directed medical therapy?
The answer turned out to be no. At 5 years of follow-up of 5,179 randomized patients with baseline stable coronary artery disease (CAD) and rigorously determined baseline moderate or severe ischemia affecting more than 10% of the myocardium, there was no difference between patients randomized to routine revascularization plus optimal medical therapy versus those on optimal medical therapy alone in the primary combined outcome of cardiovascular death, MI, heart failure, cardiac arrest, or hospitalization for unstable angina.
Of note, 35% of participants in the ISCHEMIA trial had moderate or severe silent ischemia. Like those who had angina, they achieved no additional benefit from a strategy of routine revascularization in terms of the primary outcome. ISCHEMIA participants with angina did show significant and durable improvements in quality of life and angina control with routine revascularization; however, those with silent ischemia showed little or no such improvement with an invasive strategy.
That being said, Dr. O’Gara added that he supports the ISCHEMIA investigators’ efforts to obtain funding from the National Institutes of Health for another 5 years or so of follow-up in order to determine whether revascularization actually does lead to improvement in the hard outcomes.
“Remember, in the STICH trial it took 10 years to show superiority of CABG [coronary artery bypass surgery] versus medical therapy to treat ischemic cardiomyopathy [N Engl J Med 2016; 374:1511-20]. My own view is that it’s too premature to throw the baby out with the bathwater. I think shared decision making is still very important, and I think, for many of our patients, relief of angina and improved quality of life are legitimate reasons in a low-risk situation with a good interventionalist to proceed,” he said.
Dr. O’Gara traced the history of medical thinking about silent ischemia. The notion that silent ischemia carried a clinical significance comparable with ischemia with angina gained wide credence more than 30 years ago, when investigators from the National Institutes of Health–sponsored Coronary Artery Surgery Study registry reported: “Patients with either silent or symptomatic ischemia during exercise testing have a similar risk of developing an acute myocardial infarction or sudden death – except in the three-vessel CAD subgroup, where the risk is greater in silent ischemia” (Am J Cardiol. 1988 Dec 1;62[17]:1155-8).
“This was a very important observation and led to many, many recommendations about screening and making sure that you took the expression of ST-segment depression on exercise treadmill testing pretty seriously, even if your patient did not have angina,” Dr. O’Gara recalled.
The prevailing wisdom that silent ischemia was detrimental took a hit in the Detection of Ischemia in Asymptomatic Diabetics (DIAC) trial. DIAC was conducted at a time when it had become clear that type 2 diabetes was a condition associated with increased cardiovascular risk, and that various methods of imaging were more accurate than treadmill exercise testing for the detection of underlying CAD. But when 1,123 DIAC participants with type 2 diabetes were randomized to screening with adenosine-stress radionuclide myocardial perfusion imaging or not and prospectively followed for roughly 5 years, it turned out there was no between-group difference in cardiac death or MI (JAMA. 2009 Apr 15;301[15]:1547-55).
“This pretty much put the lid on going out of one’s way to do routine screening of this nature in persons with diabetes who were considered to be at higher than average risk for the development of coronary disease,” the cardiologist commented.
Another fissure in the idea that silent ischemia was worth searching for and treating came from CLARIFY, an observational international registry of more than 20,000 individuals with stable CAD, roughly 12% of whom had silent ischemia, a figure in line with the prevalence reported in other studies. The 2-year rate of cardiovascular death or MI in the group with silent ischemia didn’t differ from the rate in patients with neither angina nor provocable ischemia. In contrast, rates of cardiovascular death or MI were significantly higher in the groups with angina but no ischemia or angina with ischemia (JAMA Intern Med. 2014 Oct;174[10]:1651-9).
“There’s something about the expression of angina that’s a very key clinical marker,” Dr. O’Gara observed.
He noted that just a few months before the ISCHEMIA trial results were released, a report from the far-smaller, randomized second Medicine, Angioplasty, or Surgery Study “threw cold water” on the notion that stress-induced ischemia in patients with multivessel CAD is a bad thing. Over 10 years of follow-up, the risk of major adverse cardiovascular events or deterioration in left ventricular function was identical in patients with or without baseline ischemia on stress testing performed after percutaneous coronary intervention, CABG surgery, or initiation of medical therapy (JAMA Intern Med. 2019 Jul 22. doi: 10.1001/jamainternmed.2019.2227).
What the guidelines say
The 6-year-old U.S. guidelines on the diagnosis and management of patients with stable ischemic heart disease are clearly out of date on the topic of silent ischemia (Circulation. 2014 Nov 4;130[19]:1749-67). The recommendations are based on expert opinion formed prior to the massive amount of new evidence that has since become available. For example, the current guidelines state as a class IIa, level of evidence C recommendation that exercise or pharmacologic stress can be useful for follow-up assessment at 2-year or greater intervals in patients with stable ischemic heart disease with prior evidence of silent ischemia.
“This is a very weak recommendation. The class of recommendation says it would be reasonable, but in the absence of an evidence base and in light of newer information, I’m not sure that it approaches even a class IIa level of recommendation,” according to Dr. O’Gara.
The 2019 European Society of Cardiology guidelines on chronic coronary syndromes are similarly weak on silent ischemia. The European guidelines state that patients with diabetes or chronic kidney disease may have a higher burden of silent ischemia, might be at higher risk for atherosclerotic cardiovascular disease events, and that periodic ECGs and functional testing every 3-5 years might be considered.
“Obviously there’s a lot of leeway there in how you wish to interpret that,” Dr. O’Gara said. “And this did not rise to the level where they’d put it in the table of recommendations, but it’s simply included as part of the explanatory text.”
What’s coming next in stable ischemic heart disease
“Nowadays all the rage has to do with coronary microvascular dysfunction,” according to Dr. O’Gara. “I think all of the research interest currently is focused on the coronary microcirculation as perhaps the next frontier in our understanding of why it is that ischemia can occur in the absence of epicardial coronary disease.”
He highly recommended a review article entitled: “Reappraisal of Ischemic Heart Disease,” in which an international trio of prominent cardiologists asserted that coronary microvascular dysfunction not only plays a pivotal pathogenic role in angina pectoris, but also in a phenomenon known as microvascular angina – that is, angina in the absence of obstructive CAD. Microvascular angina may explain the roughly one-third of patients who experience acute coronary syndrome without epicardial coronary artery stenosis or thrombosis. The authors delved into the structural and functional mechanisms underlying coronary microvascular dysfunction, while noting that effective treatment of this common phenomenon remains a major unmet need (Circulation. 2018 Oct 2;138[14]:1463-80).
Dr. O’Gara reported receiving funding from the National Heart, Lung, and Blood Institute; from Medtronic in conjunction with the ongoing pivotal APOLLO transcatheter mitral valve replacement trial; from Edwards Lifesciences for the ongoing EARLY TAVR trial; and from Medtrace Pharma, a Danish company developing an innovative form of PET diagnostic imaging.
SNOWMASS, COLO. – The concept that silent myocardial ischemia is clinically detrimental has fallen by the wayside, and routine screening for this phenomenon can no longer be recommended, Patrick T. O’Gara, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
What a difference a decade or 2 can make.
“Think about where we were 25 years ago, when we worried about people who had transient ST-segment depression without angina on Holter monitoring. We would wig out, chase them down the street, try to tackle them and load them up with medications and think about balloon [percutaneous transluminal coronary angioplasty]. And now we’re at the point where it doesn’t seem to help with respect to quality of life, let alone death or myocardial infarction,” observed Dr. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, both in Boston.
The end of the line for the now-discredited notion that silent ischemia carries clinical significance approaching that of ischemia plus angina pectoris was the landmark ISCHEMIA trial, reported in November 2019 at the annual scientific sessions of the American Heart Association. This randomized trial asked the question: Is there any high-risk subgroup of patients with stable ischemic heart disease not involving the left main coronary artery for whom a strategy of routine revascularization improves hard outcomes in the current era of highly effective, guideline-directed medical therapy?
The answer turned out to be no. At 5 years of follow-up of 5,179 randomized patients with baseline stable coronary artery disease (CAD) and rigorously determined baseline moderate or severe ischemia affecting more than 10% of the myocardium, there was no difference between patients randomized to routine revascularization plus optimal medical therapy versus those on optimal medical therapy alone in the primary combined outcome of cardiovascular death, MI, heart failure, cardiac arrest, or hospitalization for unstable angina.
Of note, 35% of participants in the ISCHEMIA trial had moderate or severe silent ischemia. Like those who had angina, they achieved no additional benefit from a strategy of routine revascularization in terms of the primary outcome. ISCHEMIA participants with angina did show significant and durable improvements in quality of life and angina control with routine revascularization; however, those with silent ischemia showed little or no such improvement with an invasive strategy.
That being said, Dr. O’Gara added that he supports the ISCHEMIA investigators’ efforts to obtain funding from the National Institutes of Health for another 5 years or so of follow-up in order to determine whether revascularization actually does lead to improvement in the hard outcomes.
“Remember, in the STICH trial it took 10 years to show superiority of CABG [coronary artery bypass surgery] versus medical therapy to treat ischemic cardiomyopathy [N Engl J Med 2016; 374:1511-20]. My own view is that it’s too premature to throw the baby out with the bathwater. I think shared decision making is still very important, and I think, for many of our patients, relief of angina and improved quality of life are legitimate reasons in a low-risk situation with a good interventionalist to proceed,” he said.
Dr. O’Gara traced the history of medical thinking about silent ischemia. The notion that silent ischemia carried a clinical significance comparable with ischemia with angina gained wide credence more than 30 years ago, when investigators from the National Institutes of Health–sponsored Coronary Artery Surgery Study registry reported: “Patients with either silent or symptomatic ischemia during exercise testing have a similar risk of developing an acute myocardial infarction or sudden death – except in the three-vessel CAD subgroup, where the risk is greater in silent ischemia” (Am J Cardiol. 1988 Dec 1;62[17]:1155-8).
“This was a very important observation and led to many, many recommendations about screening and making sure that you took the expression of ST-segment depression on exercise treadmill testing pretty seriously, even if your patient did not have angina,” Dr. O’Gara recalled.
The prevailing wisdom that silent ischemia was detrimental took a hit in the Detection of Ischemia in Asymptomatic Diabetics (DIAC) trial. DIAC was conducted at a time when it had become clear that type 2 diabetes was a condition associated with increased cardiovascular risk, and that various methods of imaging were more accurate than treadmill exercise testing for the detection of underlying CAD. But when 1,123 DIAC participants with type 2 diabetes were randomized to screening with adenosine-stress radionuclide myocardial perfusion imaging or not and prospectively followed for roughly 5 years, it turned out there was no between-group difference in cardiac death or MI (JAMA. 2009 Apr 15;301[15]:1547-55).
“This pretty much put the lid on going out of one’s way to do routine screening of this nature in persons with diabetes who were considered to be at higher than average risk for the development of coronary disease,” the cardiologist commented.
Another fissure in the idea that silent ischemia was worth searching for and treating came from CLARIFY, an observational international registry of more than 20,000 individuals with stable CAD, roughly 12% of whom had silent ischemia, a figure in line with the prevalence reported in other studies. The 2-year rate of cardiovascular death or MI in the group with silent ischemia didn’t differ from the rate in patients with neither angina nor provocable ischemia. In contrast, rates of cardiovascular death or MI were significantly higher in the groups with angina but no ischemia or angina with ischemia (JAMA Intern Med. 2014 Oct;174[10]:1651-9).
“There’s something about the expression of angina that’s a very key clinical marker,” Dr. O’Gara observed.
He noted that just a few months before the ISCHEMIA trial results were released, a report from the far-smaller, randomized second Medicine, Angioplasty, or Surgery Study “threw cold water” on the notion that stress-induced ischemia in patients with multivessel CAD is a bad thing. Over 10 years of follow-up, the risk of major adverse cardiovascular events or deterioration in left ventricular function was identical in patients with or without baseline ischemia on stress testing performed after percutaneous coronary intervention, CABG surgery, or initiation of medical therapy (JAMA Intern Med. 2019 Jul 22. doi: 10.1001/jamainternmed.2019.2227).
What the guidelines say
The 6-year-old U.S. guidelines on the diagnosis and management of patients with stable ischemic heart disease are clearly out of date on the topic of silent ischemia (Circulation. 2014 Nov 4;130[19]:1749-67). The recommendations are based on expert opinion formed prior to the massive amount of new evidence that has since become available. For example, the current guidelines state as a class IIa, level of evidence C recommendation that exercise or pharmacologic stress can be useful for follow-up assessment at 2-year or greater intervals in patients with stable ischemic heart disease with prior evidence of silent ischemia.
“This is a very weak recommendation. The class of recommendation says it would be reasonable, but in the absence of an evidence base and in light of newer information, I’m not sure that it approaches even a class IIa level of recommendation,” according to Dr. O’Gara.
The 2019 European Society of Cardiology guidelines on chronic coronary syndromes are similarly weak on silent ischemia. The European guidelines state that patients with diabetes or chronic kidney disease may have a higher burden of silent ischemia, might be at higher risk for atherosclerotic cardiovascular disease events, and that periodic ECGs and functional testing every 3-5 years might be considered.
“Obviously there’s a lot of leeway there in how you wish to interpret that,” Dr. O’Gara said. “And this did not rise to the level where they’d put it in the table of recommendations, but it’s simply included as part of the explanatory text.”
What’s coming next in stable ischemic heart disease
“Nowadays all the rage has to do with coronary microvascular dysfunction,” according to Dr. O’Gara. “I think all of the research interest currently is focused on the coronary microcirculation as perhaps the next frontier in our understanding of why it is that ischemia can occur in the absence of epicardial coronary disease.”
He highly recommended a review article entitled: “Reappraisal of Ischemic Heart Disease,” in which an international trio of prominent cardiologists asserted that coronary microvascular dysfunction not only plays a pivotal pathogenic role in angina pectoris, but also in a phenomenon known as microvascular angina – that is, angina in the absence of obstructive CAD. Microvascular angina may explain the roughly one-third of patients who experience acute coronary syndrome without epicardial coronary artery stenosis or thrombosis. The authors delved into the structural and functional mechanisms underlying coronary microvascular dysfunction, while noting that effective treatment of this common phenomenon remains a major unmet need (Circulation. 2018 Oct 2;138[14]:1463-80).
Dr. O’Gara reported receiving funding from the National Heart, Lung, and Blood Institute; from Medtronic in conjunction with the ongoing pivotal APOLLO transcatheter mitral valve replacement trial; from Edwards Lifesciences for the ongoing EARLY TAVR trial; and from Medtrace Pharma, a Danish company developing an innovative form of PET diagnostic imaging.
SNOWMASS, COLO. – The concept that silent myocardial ischemia is clinically detrimental has fallen by the wayside, and routine screening for this phenomenon can no longer be recommended, Patrick T. O’Gara, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
What a difference a decade or 2 can make.
“Think about where we were 25 years ago, when we worried about people who had transient ST-segment depression without angina on Holter monitoring. We would wig out, chase them down the street, try to tackle them and load them up with medications and think about balloon [percutaneous transluminal coronary angioplasty]. And now we’re at the point where it doesn’t seem to help with respect to quality of life, let alone death or myocardial infarction,” observed Dr. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, both in Boston.
The end of the line for the now-discredited notion that silent ischemia carries clinical significance approaching that of ischemia plus angina pectoris was the landmark ISCHEMIA trial, reported in November 2019 at the annual scientific sessions of the American Heart Association. This randomized trial asked the question: Is there any high-risk subgroup of patients with stable ischemic heart disease not involving the left main coronary artery for whom a strategy of routine revascularization improves hard outcomes in the current era of highly effective, guideline-directed medical therapy?
The answer turned out to be no. At 5 years of follow-up of 5,179 randomized patients with baseline stable coronary artery disease (CAD) and rigorously determined baseline moderate or severe ischemia affecting more than 10% of the myocardium, there was no difference between patients randomized to routine revascularization plus optimal medical therapy versus those on optimal medical therapy alone in the primary combined outcome of cardiovascular death, MI, heart failure, cardiac arrest, or hospitalization for unstable angina.
Of note, 35% of participants in the ISCHEMIA trial had moderate or severe silent ischemia. Like those who had angina, they achieved no additional benefit from a strategy of routine revascularization in terms of the primary outcome. ISCHEMIA participants with angina did show significant and durable improvements in quality of life and angina control with routine revascularization; however, those with silent ischemia showed little or no such improvement with an invasive strategy.
That being said, Dr. O’Gara added that he supports the ISCHEMIA investigators’ efforts to obtain funding from the National Institutes of Health for another 5 years or so of follow-up in order to determine whether revascularization actually does lead to improvement in the hard outcomes.
“Remember, in the STICH trial it took 10 years to show superiority of CABG [coronary artery bypass surgery] versus medical therapy to treat ischemic cardiomyopathy [N Engl J Med 2016; 374:1511-20]. My own view is that it’s too premature to throw the baby out with the bathwater. I think shared decision making is still very important, and I think, for many of our patients, relief of angina and improved quality of life are legitimate reasons in a low-risk situation with a good interventionalist to proceed,” he said.
Dr. O’Gara traced the history of medical thinking about silent ischemia. The notion that silent ischemia carried a clinical significance comparable with ischemia with angina gained wide credence more than 30 years ago, when investigators from the National Institutes of Health–sponsored Coronary Artery Surgery Study registry reported: “Patients with either silent or symptomatic ischemia during exercise testing have a similar risk of developing an acute myocardial infarction or sudden death – except in the three-vessel CAD subgroup, where the risk is greater in silent ischemia” (Am J Cardiol. 1988 Dec 1;62[17]:1155-8).
“This was a very important observation and led to many, many recommendations about screening and making sure that you took the expression of ST-segment depression on exercise treadmill testing pretty seriously, even if your patient did not have angina,” Dr. O’Gara recalled.
The prevailing wisdom that silent ischemia was detrimental took a hit in the Detection of Ischemia in Asymptomatic Diabetics (DIAC) trial. DIAC was conducted at a time when it had become clear that type 2 diabetes was a condition associated with increased cardiovascular risk, and that various methods of imaging were more accurate than treadmill exercise testing for the detection of underlying CAD. But when 1,123 DIAC participants with type 2 diabetes were randomized to screening with adenosine-stress radionuclide myocardial perfusion imaging or not and prospectively followed for roughly 5 years, it turned out there was no between-group difference in cardiac death or MI (JAMA. 2009 Apr 15;301[15]:1547-55).
“This pretty much put the lid on going out of one’s way to do routine screening of this nature in persons with diabetes who were considered to be at higher than average risk for the development of coronary disease,” the cardiologist commented.
Another fissure in the idea that silent ischemia was worth searching for and treating came from CLARIFY, an observational international registry of more than 20,000 individuals with stable CAD, roughly 12% of whom had silent ischemia, a figure in line with the prevalence reported in other studies. The 2-year rate of cardiovascular death or MI in the group with silent ischemia didn’t differ from the rate in patients with neither angina nor provocable ischemia. In contrast, rates of cardiovascular death or MI were significantly higher in the groups with angina but no ischemia or angina with ischemia (JAMA Intern Med. 2014 Oct;174[10]:1651-9).
“There’s something about the expression of angina that’s a very key clinical marker,” Dr. O’Gara observed.
He noted that just a few months before the ISCHEMIA trial results were released, a report from the far-smaller, randomized second Medicine, Angioplasty, or Surgery Study “threw cold water” on the notion that stress-induced ischemia in patients with multivessel CAD is a bad thing. Over 10 years of follow-up, the risk of major adverse cardiovascular events or deterioration in left ventricular function was identical in patients with or without baseline ischemia on stress testing performed after percutaneous coronary intervention, CABG surgery, or initiation of medical therapy (JAMA Intern Med. 2019 Jul 22. doi: 10.1001/jamainternmed.2019.2227).
What the guidelines say
The 6-year-old U.S. guidelines on the diagnosis and management of patients with stable ischemic heart disease are clearly out of date on the topic of silent ischemia (Circulation. 2014 Nov 4;130[19]:1749-67). The recommendations are based on expert opinion formed prior to the massive amount of new evidence that has since become available. For example, the current guidelines state as a class IIa, level of evidence C recommendation that exercise or pharmacologic stress can be useful for follow-up assessment at 2-year or greater intervals in patients with stable ischemic heart disease with prior evidence of silent ischemia.
“This is a very weak recommendation. The class of recommendation says it would be reasonable, but in the absence of an evidence base and in light of newer information, I’m not sure that it approaches even a class IIa level of recommendation,” according to Dr. O’Gara.
The 2019 European Society of Cardiology guidelines on chronic coronary syndromes are similarly weak on silent ischemia. The European guidelines state that patients with diabetes or chronic kidney disease may have a higher burden of silent ischemia, might be at higher risk for atherosclerotic cardiovascular disease events, and that periodic ECGs and functional testing every 3-5 years might be considered.
“Obviously there’s a lot of leeway there in how you wish to interpret that,” Dr. O’Gara said. “And this did not rise to the level where they’d put it in the table of recommendations, but it’s simply included as part of the explanatory text.”
What’s coming next in stable ischemic heart disease
“Nowadays all the rage has to do with coronary microvascular dysfunction,” according to Dr. O’Gara. “I think all of the research interest currently is focused on the coronary microcirculation as perhaps the next frontier in our understanding of why it is that ischemia can occur in the absence of epicardial coronary disease.”
He highly recommended a review article entitled: “Reappraisal of Ischemic Heart Disease,” in which an international trio of prominent cardiologists asserted that coronary microvascular dysfunction not only plays a pivotal pathogenic role in angina pectoris, but also in a phenomenon known as microvascular angina – that is, angina in the absence of obstructive CAD. Microvascular angina may explain the roughly one-third of patients who experience acute coronary syndrome without epicardial coronary artery stenosis or thrombosis. The authors delved into the structural and functional mechanisms underlying coronary microvascular dysfunction, while noting that effective treatment of this common phenomenon remains a major unmet need (Circulation. 2018 Oct 2;138[14]:1463-80).
Dr. O’Gara reported receiving funding from the National Heart, Lung, and Blood Institute; from Medtronic in conjunction with the ongoing pivotal APOLLO transcatheter mitral valve replacement trial; from Edwards Lifesciences for the ongoing EARLY TAVR trial; and from Medtrace Pharma, a Danish company developing an innovative form of PET diagnostic imaging.
EXPERT ANALYSIS FROM ACC SNOWMASS 2020
Docs weigh pulling out of MIPS over paltry payments
If you’ve knocked yourself out to earn a Merit-Based Incentive Payment System (MIPS) bonus payment, it’s pretty safe to say that getting a 1.68% payment boost probably didn’t feel like a “win” that was worth the effort.
And although it saved you from having a negative 5% payment adjustment, many physicians don’t feel that it was worth the effort.
On Jan. 6, the Centers for Medicare & Medicaid Services announced the 2020 payouts for MIPS.
Based on 2018 participation, the bonus for those who scored a perfect 100 is only a 1.68% boost in Medicare reimbursement, slightly lower than last year’s 1.88%. This decline comes as no surprise as the agency leader admits: “As the program matures, we expect that the increases in the performance thresholds in future program years will create a smaller distribution of positive payment adjustments.” Overall, more than 97% of participants avoided having a negative 5% payment adjustment.
Indeed, these bonus monies are based on a short-term appropriation of extra funds from Congress. After these temporary funds are no longer available, there will be little, if any, monies to distribute as the program is based on a “losers-feed-the-winners” construct.
It may be very tempting for many physicians to decide to ignore MIPS, with the rationale that 1.68% is not worth the effort. But don’t let your foot off the gas pedal yet, since the penalty for not participating in 2020 is a substantial 9%.
However, it is certainly time to reconsider efforts to participate at the highest level.
Should you or shouldn’t you bother with MIPS?
Let’s say you have $75,000 in revenue from Medicare Part B per year. Depending on the services you offer in your practice, that equates to 500-750 encounters with Medicare beneficiaries per year. (A reminder that MIPS affects only Part B; Medicare Advantage plans do not partake in the program.)
The recent announcement reveals that perfection would equate to an additional $1,260 per year. That’s only if you received the full 100 points; if you were simply an “exceptional performer,” the government will allot an additional $157. That’s less than you get paid for a single office visit.
The difference between perfection and compliance is approximately $1,000. Failure to participate, however, knocks $6,750 off your bottom line. Clearly, that’s a substantial financial loss that would affect most practices. Obviously, the numbers change if you have higher – or lower – Medicare revenue, but it’s important to do the math.
Why? Physicians are spending a significant amount of money to comply with the program requirements. This includes substantial payments to registries – typically $200 to >$1,000 per year – to report the quality measures for the program; electronic health record (EHR) systems, many of which require additional funding for the “upgrade” to a MIPS-compatible system, are also a sizable investment.
These hard costs pale in comparison with the time spent on understanding the ever-changing requirements of the program and the process by which your practice will implement them. Take, for example, something as innocuous as the required “Support Electronic Referral Loops by Receiving and Incorporating Health Information.”
You first must understand the elements of the measure: What is a “referral loop?” When do we need to generate one? To whom shall it be sent? What needs to be included in “health information?” What is the electronic address to which we should route the information? How do we obtain that address? Then you must determine how your EHR system captures and reports it.
Only then comes the hard part: How are we going to implement this? That’s only one of more than a dozen required elements: six quality measures, two (to four) improvement activities, and four promoting interoperability requirements. Each one of these elements has a host of requirements, all listed on multipage specification sheets.
The government does not seem to be listening. John Cullen, MD, president of the American Academy of Family Physicians, testified at the Senate Finance Committee in May 2019 that MIPS “has created a burdensome and extremely complex program that has increased practice costs ... ” Yet, later that year, CMS issued another hefty ruling that outlines significant changes to the program, despite the fact that it’s in its fourth performance year.
Turning frustration into action
Frustration or even anger may be one reaction, but now is an opportune time to determine your investment in the program. At a minimum, it’s vital to understand and meet the threshold to avoid the penalty. It’s been shifting to date, but it’s now set at 9% for perpetuity.
First, it’s crucial to check on your participation status. CMS revealed that the participation database was recently corrected for so-called inconsistencies, so it pays to double-check. It only takes seconds: Insert your NPI in the QPP Participation Status Tool to determine your eligibility for 2020.
In 2020, the threshold to avoid the penalty is 45 points. To get the 45 points, practices must participate in two improvement activities, which is not difficult as there are 118 options. That will garner 15 points. Then there are 45 points available from the quality category; you need at least 30 to reach the 45-point threshold for penalty avoidance.
Smart MIPS hacks that can help you
To obtain the additional 30 points, turn your attention to the quality category. There are 268 quality measures; choose at least six to measure. If you report directly from your EHR system, you’ll get a bonus point for each reported measure, plus one just for trying. (There are a few other opportunities for bonus points, such as improving your scores over last year.) Those bonus points give you a base with which to work, but getting to 45 will require effort to report successfully on at least a couple of the measures.
The quality category has a total of 100 points available, which are converted to 45 toward your composite score. Since you need 30 to reach that magical 45 (if 15 were attained from improvement activities), that means you must come up with 75 points in the quality category. Between the bonus points and measuring a handful of measures successfully through the year, you’ll achieve this threshold.
There are two other categories in the program: promoting interoperability (PI) and cost. The PI category mirrors the old “meaningful use” program; however, it has become increasingly difficult over the years. If you think that you can meet the required elements, you can pick up 25 more points toward your composite score.
Cost is a bit of an unknown, as the scoring is based on a retrospective review of your claims. You’ll likely pick up a few more points on this 15-point category, but there’s no method to determine performance until after the reporting period. Therefore, be cautious about relying on this category.
The best MIPS hack, however, is if you are a small practice. CMS – remarkably – defines a “small practice” as 15 or fewer eligible professionals. If you qualify under this paradigm, you have multiple options to ease compliance:
Apply for a “hardship exemption” simply on the basis of being small; the exemption relates to the promoting operability category, shifting those points to the quality category.
Gain three points per quality measure, regardless of data completeness; this compares to just one point for other physicians.
Capture all of the points available from the Improvement Activities category by confirming participation with just a single activity. (This also applies to all physicians in rural or Health Professional Shortage Areas.)
In the event that you don’t qualify as a “small practice” or you’re still falling short of the requirements, CMS allows for the ultimate “out”: You can apply for exemption on the basis of an “extreme and uncontrollable circumstance.” The applications for these exceptions open this summer.
Unless you qualify for the program exemption, it’s important to keep pace with the program to ensure that you reach the 45-point threshold. It may not, however, be worthwhile to gear up for all 100 points unless your estimate of the potential return – and what it costs you to get there – reveals otherwise. MIPS is not going anywhere; the program is written into the law.
But that doesn’t mean that CMS can’t make tweaks and updates. Hopefully, the revisions won’t create even more administrative burden as the program is quickly turning into a big stick with only a small carrot at the end.
Elizabeth Woodcock is president of Woodcock & Associates in Atlanta. She has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
If you’ve knocked yourself out to earn a Merit-Based Incentive Payment System (MIPS) bonus payment, it’s pretty safe to say that getting a 1.68% payment boost probably didn’t feel like a “win” that was worth the effort.
And although it saved you from having a negative 5% payment adjustment, many physicians don’t feel that it was worth the effort.
On Jan. 6, the Centers for Medicare & Medicaid Services announced the 2020 payouts for MIPS.
Based on 2018 participation, the bonus for those who scored a perfect 100 is only a 1.68% boost in Medicare reimbursement, slightly lower than last year’s 1.88%. This decline comes as no surprise as the agency leader admits: “As the program matures, we expect that the increases in the performance thresholds in future program years will create a smaller distribution of positive payment adjustments.” Overall, more than 97% of participants avoided having a negative 5% payment adjustment.
Indeed, these bonus monies are based on a short-term appropriation of extra funds from Congress. After these temporary funds are no longer available, there will be little, if any, monies to distribute as the program is based on a “losers-feed-the-winners” construct.
It may be very tempting for many physicians to decide to ignore MIPS, with the rationale that 1.68% is not worth the effort. But don’t let your foot off the gas pedal yet, since the penalty for not participating in 2020 is a substantial 9%.
However, it is certainly time to reconsider efforts to participate at the highest level.
Should you or shouldn’t you bother with MIPS?
Let’s say you have $75,000 in revenue from Medicare Part B per year. Depending on the services you offer in your practice, that equates to 500-750 encounters with Medicare beneficiaries per year. (A reminder that MIPS affects only Part B; Medicare Advantage plans do not partake in the program.)
The recent announcement reveals that perfection would equate to an additional $1,260 per year. That’s only if you received the full 100 points; if you were simply an “exceptional performer,” the government will allot an additional $157. That’s less than you get paid for a single office visit.
The difference between perfection and compliance is approximately $1,000. Failure to participate, however, knocks $6,750 off your bottom line. Clearly, that’s a substantial financial loss that would affect most practices. Obviously, the numbers change if you have higher – or lower – Medicare revenue, but it’s important to do the math.
Why? Physicians are spending a significant amount of money to comply with the program requirements. This includes substantial payments to registries – typically $200 to >$1,000 per year – to report the quality measures for the program; electronic health record (EHR) systems, many of which require additional funding for the “upgrade” to a MIPS-compatible system, are also a sizable investment.
These hard costs pale in comparison with the time spent on understanding the ever-changing requirements of the program and the process by which your practice will implement them. Take, for example, something as innocuous as the required “Support Electronic Referral Loops by Receiving and Incorporating Health Information.”
You first must understand the elements of the measure: What is a “referral loop?” When do we need to generate one? To whom shall it be sent? What needs to be included in “health information?” What is the electronic address to which we should route the information? How do we obtain that address? Then you must determine how your EHR system captures and reports it.
Only then comes the hard part: How are we going to implement this? That’s only one of more than a dozen required elements: six quality measures, two (to four) improvement activities, and four promoting interoperability requirements. Each one of these elements has a host of requirements, all listed on multipage specification sheets.
The government does not seem to be listening. John Cullen, MD, president of the American Academy of Family Physicians, testified at the Senate Finance Committee in May 2019 that MIPS “has created a burdensome and extremely complex program that has increased practice costs ... ” Yet, later that year, CMS issued another hefty ruling that outlines significant changes to the program, despite the fact that it’s in its fourth performance year.
Turning frustration into action
Frustration or even anger may be one reaction, but now is an opportune time to determine your investment in the program. At a minimum, it’s vital to understand and meet the threshold to avoid the penalty. It’s been shifting to date, but it’s now set at 9% for perpetuity.
First, it’s crucial to check on your participation status. CMS revealed that the participation database was recently corrected for so-called inconsistencies, so it pays to double-check. It only takes seconds: Insert your NPI in the QPP Participation Status Tool to determine your eligibility for 2020.
In 2020, the threshold to avoid the penalty is 45 points. To get the 45 points, practices must participate in two improvement activities, which is not difficult as there are 118 options. That will garner 15 points. Then there are 45 points available from the quality category; you need at least 30 to reach the 45-point threshold for penalty avoidance.
Smart MIPS hacks that can help you
To obtain the additional 30 points, turn your attention to the quality category. There are 268 quality measures; choose at least six to measure. If you report directly from your EHR system, you’ll get a bonus point for each reported measure, plus one just for trying. (There are a few other opportunities for bonus points, such as improving your scores over last year.) Those bonus points give you a base with which to work, but getting to 45 will require effort to report successfully on at least a couple of the measures.
The quality category has a total of 100 points available, which are converted to 45 toward your composite score. Since you need 30 to reach that magical 45 (if 15 were attained from improvement activities), that means you must come up with 75 points in the quality category. Between the bonus points and measuring a handful of measures successfully through the year, you’ll achieve this threshold.
There are two other categories in the program: promoting interoperability (PI) and cost. The PI category mirrors the old “meaningful use” program; however, it has become increasingly difficult over the years. If you think that you can meet the required elements, you can pick up 25 more points toward your composite score.
Cost is a bit of an unknown, as the scoring is based on a retrospective review of your claims. You’ll likely pick up a few more points on this 15-point category, but there’s no method to determine performance until after the reporting period. Therefore, be cautious about relying on this category.
The best MIPS hack, however, is if you are a small practice. CMS – remarkably – defines a “small practice” as 15 or fewer eligible professionals. If you qualify under this paradigm, you have multiple options to ease compliance:
Apply for a “hardship exemption” simply on the basis of being small; the exemption relates to the promoting operability category, shifting those points to the quality category.
Gain three points per quality measure, regardless of data completeness; this compares to just one point for other physicians.
Capture all of the points available from the Improvement Activities category by confirming participation with just a single activity. (This also applies to all physicians in rural or Health Professional Shortage Areas.)
In the event that you don’t qualify as a “small practice” or you’re still falling short of the requirements, CMS allows for the ultimate “out”: You can apply for exemption on the basis of an “extreme and uncontrollable circumstance.” The applications for these exceptions open this summer.
Unless you qualify for the program exemption, it’s important to keep pace with the program to ensure that you reach the 45-point threshold. It may not, however, be worthwhile to gear up for all 100 points unless your estimate of the potential return – and what it costs you to get there – reveals otherwise. MIPS is not going anywhere; the program is written into the law.
But that doesn’t mean that CMS can’t make tweaks and updates. Hopefully, the revisions won’t create even more administrative burden as the program is quickly turning into a big stick with only a small carrot at the end.
Elizabeth Woodcock is president of Woodcock & Associates in Atlanta. She has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
If you’ve knocked yourself out to earn a Merit-Based Incentive Payment System (MIPS) bonus payment, it’s pretty safe to say that getting a 1.68% payment boost probably didn’t feel like a “win” that was worth the effort.
And although it saved you from having a negative 5% payment adjustment, many physicians don’t feel that it was worth the effort.
On Jan. 6, the Centers for Medicare & Medicaid Services announced the 2020 payouts for MIPS.
Based on 2018 participation, the bonus for those who scored a perfect 100 is only a 1.68% boost in Medicare reimbursement, slightly lower than last year’s 1.88%. This decline comes as no surprise as the agency leader admits: “As the program matures, we expect that the increases in the performance thresholds in future program years will create a smaller distribution of positive payment adjustments.” Overall, more than 97% of participants avoided having a negative 5% payment adjustment.
Indeed, these bonus monies are based on a short-term appropriation of extra funds from Congress. After these temporary funds are no longer available, there will be little, if any, monies to distribute as the program is based on a “losers-feed-the-winners” construct.
It may be very tempting for many physicians to decide to ignore MIPS, with the rationale that 1.68% is not worth the effort. But don’t let your foot off the gas pedal yet, since the penalty for not participating in 2020 is a substantial 9%.
However, it is certainly time to reconsider efforts to participate at the highest level.
Should you or shouldn’t you bother with MIPS?
Let’s say you have $75,000 in revenue from Medicare Part B per year. Depending on the services you offer in your practice, that equates to 500-750 encounters with Medicare beneficiaries per year. (A reminder that MIPS affects only Part B; Medicare Advantage plans do not partake in the program.)
The recent announcement reveals that perfection would equate to an additional $1,260 per year. That’s only if you received the full 100 points; if you were simply an “exceptional performer,” the government will allot an additional $157. That’s less than you get paid for a single office visit.
The difference between perfection and compliance is approximately $1,000. Failure to participate, however, knocks $6,750 off your bottom line. Clearly, that’s a substantial financial loss that would affect most practices. Obviously, the numbers change if you have higher – or lower – Medicare revenue, but it’s important to do the math.
Why? Physicians are spending a significant amount of money to comply with the program requirements. This includes substantial payments to registries – typically $200 to >$1,000 per year – to report the quality measures for the program; electronic health record (EHR) systems, many of which require additional funding for the “upgrade” to a MIPS-compatible system, are also a sizable investment.
These hard costs pale in comparison with the time spent on understanding the ever-changing requirements of the program and the process by which your practice will implement them. Take, for example, something as innocuous as the required “Support Electronic Referral Loops by Receiving and Incorporating Health Information.”
You first must understand the elements of the measure: What is a “referral loop?” When do we need to generate one? To whom shall it be sent? What needs to be included in “health information?” What is the electronic address to which we should route the information? How do we obtain that address? Then you must determine how your EHR system captures and reports it.
Only then comes the hard part: How are we going to implement this? That’s only one of more than a dozen required elements: six quality measures, two (to four) improvement activities, and four promoting interoperability requirements. Each one of these elements has a host of requirements, all listed on multipage specification sheets.
The government does not seem to be listening. John Cullen, MD, president of the American Academy of Family Physicians, testified at the Senate Finance Committee in May 2019 that MIPS “has created a burdensome and extremely complex program that has increased practice costs ... ” Yet, later that year, CMS issued another hefty ruling that outlines significant changes to the program, despite the fact that it’s in its fourth performance year.
Turning frustration into action
Frustration or even anger may be one reaction, but now is an opportune time to determine your investment in the program. At a minimum, it’s vital to understand and meet the threshold to avoid the penalty. It’s been shifting to date, but it’s now set at 9% for perpetuity.
First, it’s crucial to check on your participation status. CMS revealed that the participation database was recently corrected for so-called inconsistencies, so it pays to double-check. It only takes seconds: Insert your NPI in the QPP Participation Status Tool to determine your eligibility for 2020.
In 2020, the threshold to avoid the penalty is 45 points. To get the 45 points, practices must participate in two improvement activities, which is not difficult as there are 118 options. That will garner 15 points. Then there are 45 points available from the quality category; you need at least 30 to reach the 45-point threshold for penalty avoidance.
Smart MIPS hacks that can help you
To obtain the additional 30 points, turn your attention to the quality category. There are 268 quality measures; choose at least six to measure. If you report directly from your EHR system, you’ll get a bonus point for each reported measure, plus one just for trying. (There are a few other opportunities for bonus points, such as improving your scores over last year.) Those bonus points give you a base with which to work, but getting to 45 will require effort to report successfully on at least a couple of the measures.
The quality category has a total of 100 points available, which are converted to 45 toward your composite score. Since you need 30 to reach that magical 45 (if 15 were attained from improvement activities), that means you must come up with 75 points in the quality category. Between the bonus points and measuring a handful of measures successfully through the year, you’ll achieve this threshold.
There are two other categories in the program: promoting interoperability (PI) and cost. The PI category mirrors the old “meaningful use” program; however, it has become increasingly difficult over the years. If you think that you can meet the required elements, you can pick up 25 more points toward your composite score.
Cost is a bit of an unknown, as the scoring is based on a retrospective review of your claims. You’ll likely pick up a few more points on this 15-point category, but there’s no method to determine performance until after the reporting period. Therefore, be cautious about relying on this category.
The best MIPS hack, however, is if you are a small practice. CMS – remarkably – defines a “small practice” as 15 or fewer eligible professionals. If you qualify under this paradigm, you have multiple options to ease compliance:
Apply for a “hardship exemption” simply on the basis of being small; the exemption relates to the promoting operability category, shifting those points to the quality category.
Gain three points per quality measure, regardless of data completeness; this compares to just one point for other physicians.
Capture all of the points available from the Improvement Activities category by confirming participation with just a single activity. (This also applies to all physicians in rural or Health Professional Shortage Areas.)
In the event that you don’t qualify as a “small practice” or you’re still falling short of the requirements, CMS allows for the ultimate “out”: You can apply for exemption on the basis of an “extreme and uncontrollable circumstance.” The applications for these exceptions open this summer.
Unless you qualify for the program exemption, it’s important to keep pace with the program to ensure that you reach the 45-point threshold. It may not, however, be worthwhile to gear up for all 100 points unless your estimate of the potential return – and what it costs you to get there – reveals otherwise. MIPS is not going anywhere; the program is written into the law.
But that doesn’t mean that CMS can’t make tweaks and updates. Hopefully, the revisions won’t create even more administrative burden as the program is quickly turning into a big stick with only a small carrot at the end.
Elizabeth Woodcock is president of Woodcock & Associates in Atlanta. She has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Costs are keeping Americans out of the doctor’s office
The cost of health care is keeping more Americans from seeing a doctor, even as the number of individuals with insurance coverage increases, according to a new study.
“Despite short-term gains owing to the [Affordable Care Act], over the past 20 years the portion of adults aged 18-64 years unable to see a physician owing to the cost increased, mostly because of an increase among persons with insurance,” Laura Hawks, MD, of Cambridge (Mass.) Health Alliance and Harvard Medical School in Boston and colleagues wrote in a new research report published in JAMA Internal Medicine.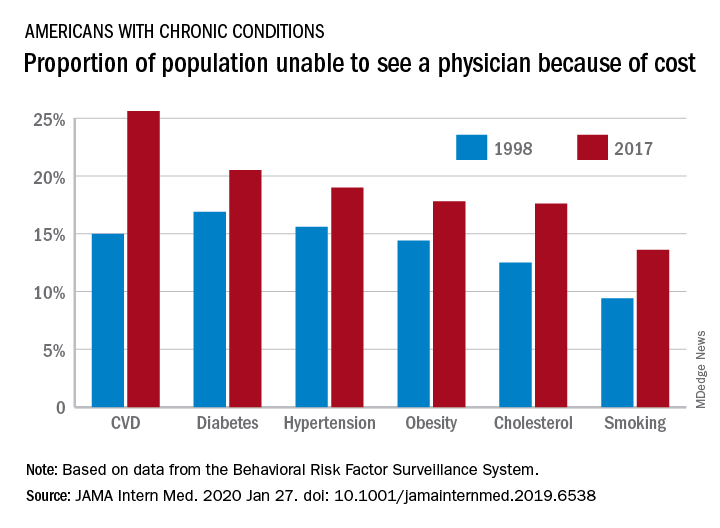
“In 2017, nearly one-fifth of individuals with any chronic condition (diabetes, obesity, or cardiovascular disease) said they were unable to see a physician owing to cost,” they continued.
Researchers examined 20 years of data (January 1998 through December 2017) from the Centers for Disease Control and Prevention’s Behavioral Risk Factor Surveillance System to identify trends in unmet need for physician and preventive services.
Among adults aged 18-64 years who responded to the survey in 1998 and 2017, uninsurance decreased by 2.1 percentage points, falling from 16.9% to 14.8%. But at the same time, the portion of adults who were unable to see a physician because of cost rose by 2.7 percentage points, from 11.4% to 15.7%. Looking specifically at adults who had insurance coverage, the researchers found that cost was a barrier for 11.5% of them in 2017, up from 7.1% in 1998.
These results come against a backdrop of growing medical costs, increasing deductibles and copayments, an increasing use of cost containment measures like prior authorization, and narrow provider networks in the wake of the transition to value-based payment structures, the authors noted.
“Our finding that financial access to physician care worsened is concerning,” Dr. Hawks and her colleagues wrote. “Persons with conditions such as diabetes, hypertension, cardiovascular disease, and poor health status risk substantial harms if they forgo physician care. Financial barriers to care have been associated with increased hospitalizations and worse health outcomes in patients with cardiovascular disease and hypertension and increased morbidity among patients with diabetes.”
One of the trends highlighted by the study authors is the growing number of employers offering plans with a high deductible.
“Enrollment in a high-deductible health plan, which has become increasingly common in the last decade, a trend uninterrupted by the ACA, is associated with forgoing needed care, especially among those of lower socioeconomic status,” the authors wrote. “Other changes in insurance benefit design, such as imposing tiered copayments and coinsurance obligations, eliminating coverage for some services (e.g., eyeglasses) and narrowing provider networks (which can force some patients to go out-of-network for care) may also have undermined the affordability of care.”
There was some positive news among the findings, however.
“The main encouraging finding from our analysis is the increase in the proportion of persons – both insured and uninsured – receiving cholesterol checks and flu shots,” Dr. Hawk and her colleagues wrote, adding that this increase “may be attributable to the increasing implementation of quality metrics, financial incentives, and improved systems for the delivery of these services.”
However, not all preventive services that had cost barriers eliminated under the ACA saw improvement, such as cancer screening. They note that the proportion of women who did not receive mammography increased during the study period and then plateaued, but did not improve following the implementation of the ACA. The authors described the reasons for this as “unclear.”
Dr. Hawks received funding support from an Institutional National Research Service award and from Cambridge Health Alliance, her employer. Other authors reported membership in Physicians for a National Health Program.
SOURCE: Hawks L et al. JAMA Intern Med. 2020 Jan 27. doi: 10.1001/jamainternmed.2019.6538.
The cost of health care is keeping more Americans from seeing a doctor, even as the number of individuals with insurance coverage increases, according to a new study.
“Despite short-term gains owing to the [Affordable Care Act], over the past 20 years the portion of adults aged 18-64 years unable to see a physician owing to the cost increased, mostly because of an increase among persons with insurance,” Laura Hawks, MD, of Cambridge (Mass.) Health Alliance and Harvard Medical School in Boston and colleagues wrote in a new research report published in JAMA Internal Medicine.
“In 2017, nearly one-fifth of individuals with any chronic condition (diabetes, obesity, or cardiovascular disease) said they were unable to see a physician owing to cost,” they continued.
Researchers examined 20 years of data (January 1998 through December 2017) from the Centers for Disease Control and Prevention’s Behavioral Risk Factor Surveillance System to identify trends in unmet need for physician and preventive services.
Among adults aged 18-64 years who responded to the survey in 1998 and 2017, uninsurance decreased by 2.1 percentage points, falling from 16.9% to 14.8%. But at the same time, the portion of adults who were unable to see a physician because of cost rose by 2.7 percentage points, from 11.4% to 15.7%. Looking specifically at adults who had insurance coverage, the researchers found that cost was a barrier for 11.5% of them in 2017, up from 7.1% in 1998.
These results come against a backdrop of growing medical costs, increasing deductibles and copayments, an increasing use of cost containment measures like prior authorization, and narrow provider networks in the wake of the transition to value-based payment structures, the authors noted.
“Our finding that financial access to physician care worsened is concerning,” Dr. Hawks and her colleagues wrote. “Persons with conditions such as diabetes, hypertension, cardiovascular disease, and poor health status risk substantial harms if they forgo physician care. Financial barriers to care have been associated with increased hospitalizations and worse health outcomes in patients with cardiovascular disease and hypertension and increased morbidity among patients with diabetes.”
One of the trends highlighted by the study authors is the growing number of employers offering plans with a high deductible.
“Enrollment in a high-deductible health plan, which has become increasingly common in the last decade, a trend uninterrupted by the ACA, is associated with forgoing needed care, especially among those of lower socioeconomic status,” the authors wrote. “Other changes in insurance benefit design, such as imposing tiered copayments and coinsurance obligations, eliminating coverage for some services (e.g., eyeglasses) and narrowing provider networks (which can force some patients to go out-of-network for care) may also have undermined the affordability of care.”
There was some positive news among the findings, however.
“The main encouraging finding from our analysis is the increase in the proportion of persons – both insured and uninsured – receiving cholesterol checks and flu shots,” Dr. Hawk and her colleagues wrote, adding that this increase “may be attributable to the increasing implementation of quality metrics, financial incentives, and improved systems for the delivery of these services.”
However, not all preventive services that had cost barriers eliminated under the ACA saw improvement, such as cancer screening. They note that the proportion of women who did not receive mammography increased during the study period and then plateaued, but did not improve following the implementation of the ACA. The authors described the reasons for this as “unclear.”
Dr. Hawks received funding support from an Institutional National Research Service award and from Cambridge Health Alliance, her employer. Other authors reported membership in Physicians for a National Health Program.
SOURCE: Hawks L et al. JAMA Intern Med. 2020 Jan 27. doi: 10.1001/jamainternmed.2019.6538.
The cost of health care is keeping more Americans from seeing a doctor, even as the number of individuals with insurance coverage increases, according to a new study.
“Despite short-term gains owing to the [Affordable Care Act], over the past 20 years the portion of adults aged 18-64 years unable to see a physician owing to the cost increased, mostly because of an increase among persons with insurance,” Laura Hawks, MD, of Cambridge (Mass.) Health Alliance and Harvard Medical School in Boston and colleagues wrote in a new research report published in JAMA Internal Medicine.
“In 2017, nearly one-fifth of individuals with any chronic condition (diabetes, obesity, or cardiovascular disease) said they were unable to see a physician owing to cost,” they continued.
Researchers examined 20 years of data (January 1998 through December 2017) from the Centers for Disease Control and Prevention’s Behavioral Risk Factor Surveillance System to identify trends in unmet need for physician and preventive services.
Among adults aged 18-64 years who responded to the survey in 1998 and 2017, uninsurance decreased by 2.1 percentage points, falling from 16.9% to 14.8%. But at the same time, the portion of adults who were unable to see a physician because of cost rose by 2.7 percentage points, from 11.4% to 15.7%. Looking specifically at adults who had insurance coverage, the researchers found that cost was a barrier for 11.5% of them in 2017, up from 7.1% in 1998.
These results come against a backdrop of growing medical costs, increasing deductibles and copayments, an increasing use of cost containment measures like prior authorization, and narrow provider networks in the wake of the transition to value-based payment structures, the authors noted.
“Our finding that financial access to physician care worsened is concerning,” Dr. Hawks and her colleagues wrote. “Persons with conditions such as diabetes, hypertension, cardiovascular disease, and poor health status risk substantial harms if they forgo physician care. Financial barriers to care have been associated with increased hospitalizations and worse health outcomes in patients with cardiovascular disease and hypertension and increased morbidity among patients with diabetes.”
One of the trends highlighted by the study authors is the growing number of employers offering plans with a high deductible.
“Enrollment in a high-deductible health plan, which has become increasingly common in the last decade, a trend uninterrupted by the ACA, is associated with forgoing needed care, especially among those of lower socioeconomic status,” the authors wrote. “Other changes in insurance benefit design, such as imposing tiered copayments and coinsurance obligations, eliminating coverage for some services (e.g., eyeglasses) and narrowing provider networks (which can force some patients to go out-of-network for care) may also have undermined the affordability of care.”
There was some positive news among the findings, however.
“The main encouraging finding from our analysis is the increase in the proportion of persons – both insured and uninsured – receiving cholesterol checks and flu shots,” Dr. Hawk and her colleagues wrote, adding that this increase “may be attributable to the increasing implementation of quality metrics, financial incentives, and improved systems for the delivery of these services.”
However, not all preventive services that had cost barriers eliminated under the ACA saw improvement, such as cancer screening. They note that the proportion of women who did not receive mammography increased during the study period and then plateaued, but did not improve following the implementation of the ACA. The authors described the reasons for this as “unclear.”
Dr. Hawks received funding support from an Institutional National Research Service award and from Cambridge Health Alliance, her employer. Other authors reported membership in Physicians for a National Health Program.
SOURCE: Hawks L et al. JAMA Intern Med. 2020 Jan 27. doi: 10.1001/jamainternmed.2019.6538.
FROM JAMA INTERNAL MEDICINE
Journal editors seek more complete disclosure from authors
A group of leading medical journal editors is seeking to improve the completeness and transparency of financial disclosure reporting with a proposed new disclosure form that puts more onus on readers to decide whether relationships and activities should influence how they view published papers.
The proposed changes are described in an editorial published simultaneously today in the Annals of Internal Medicine, British Medical Journal, Journal of the American Medical Association, The Lancet, New England Journal of Medicine, and several other journals whose editors are members of the International Committee of Medical Journal Editors (ICMJE).
“While no approach to disclosure will be perfect or foolproof, we hope the changes we propose will help promote transparency and trust,” the editorial stated (Ann Intern Med. 2020 Jan 27. doi: 10.7326/M19-3933).
The ICMJE adopted its currently used electronic form – the “ICMJE Form for the Disclosure of Potential Conflicts of Interest” – 10 years ago in an effort to create some uniformity amidst a patchwork of differing disclosure requirements for authors.
It’s not known how many journals outside of the ICMJE’s member journals routinely use the disclosure form, but the organization’s website houses an extensive list of journals whose editors or publishers have requested to be listed as following the ICMJE’s recommendations for editing, reporting, and publishing, including those concerning disclosures. The ICMJE does not “certify” journals. The full set of recommendations was updated in December 2019.
Most authors are committed to transparent reporting, but “opinions differ over which relationships or activities to report,” the editorial stated.
An author might choose to omit an item that others deem important because of a difference in opinion regarding “relevance,” confusion over definitions, or a simple oversight. Some authors may be “concerned that readers will interpret the listing of any item as a ‘potential conflict of interest’ as indicative of problematic influence and wrongdoing,” the editorial stated.
The revised form, like the current one, asks authors to disclose relationships and activities that are directly related to the reported work, as well as those that are topically related (within the broadly defined field addressed in the work). But unlike the current form, the new version provides a checklist of relationships and activities and asks authors to check ‘yes’ or ‘no’ for each one (and to name them when the answer is ‘yes’).
Items in the checklist include grants, payments/honoraria for lectures, patents issued or planned, stock/stock options, and leadership or fiduciary roles in committees, boards, or societies.
The proposed new form makes no mention of “potential conflicts of interest” or “relevancy,” per say. Authors aren’t asked to determine what might be interpreted as a potential conflict of interest, but instead are asked for a “complete listing” of what readers may find “pertinent” to their work.
“We’re trying to move away from calling everything a [potential] ‘conflict,’ ” Darren B. Taichman, MD, PhD, secretary of ICMJE and executive editor of the Annals of Internal Medicine, said in an interview. “We want to remove for authors the concern or stigma, if you will, that anything listed on a form implies that there is something wrong, because that’s just not true. … We want readers to decide what relationships are important as they interpret the work.”
Dr. Taichman said in the interview that the ICMJE’s updating of the form was more a function of “good housekeeping” and continuous appreciation of disclosure as an important issue, rather than any one specific issue, such as concern over a “relevancy” approach to disclosures.
The ICMJE is seeking feedback about its proposed form, which is available with a link for providing comments, at www.icmje.org.
Broader national efforts
Editors and others have been increasingly moving, however, toward asking for more complete disclosures where authors aren’t asked to judge “relevancy” and where readers can make decisions on their own. The American Society of Clinical Oncology, which produces the Journal of Clinical Oncology (JCO) as well as practice guidelines and continuing medical education programs, moved about 5 years ago to a system of general disclosure that asks physicians and others to disclose all financial interests and industry relationships, with no qualifiers.
Earlier in January 2020, the Accreditation Council for Continuing Medical Education issued proposed revisions to its Standards for Integrity and Independence in Accredited Continuing Education. These revisions, which are open for comment, require CME providers to collect disclosure information about all financial relationships of speakers and presenters. It’s up to the CME provider to then determine which relationships are relevant, according to the proposed document.
More change is on the way, as disclosure issues are being deliberated nationally in the wake of a highly publicized disclosure failure at Memorial Sloan Kettering Cancer Center in 2018. Chief medical officer José Baselga, MD, PhD, failed to report millions of dollars of industry payments and ownership interests in journal articles he wrote or cowrote over several years.
In February 2019, leaders from journals, academia, medical societies, and other institutions gathered in Washington for a closed-door meeting to hash out various disclosure related issues.
Hosted by the Association of American Medical Colleges and cosponsored by Memorial Sloan Kettering Cancer Center, ASCO, JAMA, and the Council of Medical Specialty Societies, the meeting led to a series of working groups that are creating additional recommendations “due out soon in 2020,” Heather Pierce, senior director of science policy and regulatory counsel for the AAMC, said in an interview.
Among the questions being discussed: What disclosures should be verified and who should do so? How can disclosures be made more complete and easier for researchers? And, “most importantly,” said Ms. Pierce, how can policy requirements across each of these sectors be aligned so that there’s more coordination and oversight – and with it, public trust?
Some critics of current disclosure policies have called for more reporting of compensation amounts, and Ms. Pierce said that this has been part of cross-sector discussions.
The ICMJE’s proposed form invites, but does not require, authors to indicate what payments were made to them or their institutions. “Part of this is due to the fact that it’s hard to define, let alone agree on, what’s an important amount,” Dr. Taichman said.
A push for registries
The ICMJE is also aiming to make the disclosure process more efficient for authors – and to eliminate inconsistent and incomplete disclosures – by accepting disclosures from web-based repositories, according to the editorial. Repositories allow authors to maintain an inventory of their relationships and activities and then create electronic disclosures that are tailored to the requirements of the ICMJE, medical societies, and other entities.
The AAMC-run repository, called Convey, is consistent with ICMJE reporting requirements and other criteria (e.g., there are no fees for individuals to enter, store, or export their data), but the development of other repositories may be helpful “for meeting regional, linguistic, and regulatory needs” of authors across the world, the editorial stated.
The Annals of Internal Medicine and the New England Journal of Medicine are both currently collecting disclosures through Convey. The platform was born from discussions that followed a 2009 Institute of Medicine report on conflicts of interest.
Signers of the ICMJE editorial include representatives of the National Library of Medicine and the World Association of Medical Editors, in addition to editors in chief and other leaders of the ICMJE member journals.
A group of leading medical journal editors is seeking to improve the completeness and transparency of financial disclosure reporting with a proposed new disclosure form that puts more onus on readers to decide whether relationships and activities should influence how they view published papers.
The proposed changes are described in an editorial published simultaneously today in the Annals of Internal Medicine, British Medical Journal, Journal of the American Medical Association, The Lancet, New England Journal of Medicine, and several other journals whose editors are members of the International Committee of Medical Journal Editors (ICMJE).
“While no approach to disclosure will be perfect or foolproof, we hope the changes we propose will help promote transparency and trust,” the editorial stated (Ann Intern Med. 2020 Jan 27. doi: 10.7326/M19-3933).
The ICMJE adopted its currently used electronic form – the “ICMJE Form for the Disclosure of Potential Conflicts of Interest” – 10 years ago in an effort to create some uniformity amidst a patchwork of differing disclosure requirements for authors.
It’s not known how many journals outside of the ICMJE’s member journals routinely use the disclosure form, but the organization’s website houses an extensive list of journals whose editors or publishers have requested to be listed as following the ICMJE’s recommendations for editing, reporting, and publishing, including those concerning disclosures. The ICMJE does not “certify” journals. The full set of recommendations was updated in December 2019.
Most authors are committed to transparent reporting, but “opinions differ over which relationships or activities to report,” the editorial stated.
An author might choose to omit an item that others deem important because of a difference in opinion regarding “relevance,” confusion over definitions, or a simple oversight. Some authors may be “concerned that readers will interpret the listing of any item as a ‘potential conflict of interest’ as indicative of problematic influence and wrongdoing,” the editorial stated.
The revised form, like the current one, asks authors to disclose relationships and activities that are directly related to the reported work, as well as those that are topically related (within the broadly defined field addressed in the work). But unlike the current form, the new version provides a checklist of relationships and activities and asks authors to check ‘yes’ or ‘no’ for each one (and to name them when the answer is ‘yes’).
Items in the checklist include grants, payments/honoraria for lectures, patents issued or planned, stock/stock options, and leadership or fiduciary roles in committees, boards, or societies.
The proposed new form makes no mention of “potential conflicts of interest” or “relevancy,” per say. Authors aren’t asked to determine what might be interpreted as a potential conflict of interest, but instead are asked for a “complete listing” of what readers may find “pertinent” to their work.
“We’re trying to move away from calling everything a [potential] ‘conflict,’ ” Darren B. Taichman, MD, PhD, secretary of ICMJE and executive editor of the Annals of Internal Medicine, said in an interview. “We want to remove for authors the concern or stigma, if you will, that anything listed on a form implies that there is something wrong, because that’s just not true. … We want readers to decide what relationships are important as they interpret the work.”
Dr. Taichman said in the interview that the ICMJE’s updating of the form was more a function of “good housekeeping” and continuous appreciation of disclosure as an important issue, rather than any one specific issue, such as concern over a “relevancy” approach to disclosures.
The ICMJE is seeking feedback about its proposed form, which is available with a link for providing comments, at www.icmje.org.
Broader national efforts
Editors and others have been increasingly moving, however, toward asking for more complete disclosures where authors aren’t asked to judge “relevancy” and where readers can make decisions on their own. The American Society of Clinical Oncology, which produces the Journal of Clinical Oncology (JCO) as well as practice guidelines and continuing medical education programs, moved about 5 years ago to a system of general disclosure that asks physicians and others to disclose all financial interests and industry relationships, with no qualifiers.
Earlier in January 2020, the Accreditation Council for Continuing Medical Education issued proposed revisions to its Standards for Integrity and Independence in Accredited Continuing Education. These revisions, which are open for comment, require CME providers to collect disclosure information about all financial relationships of speakers and presenters. It’s up to the CME provider to then determine which relationships are relevant, according to the proposed document.
More change is on the way, as disclosure issues are being deliberated nationally in the wake of a highly publicized disclosure failure at Memorial Sloan Kettering Cancer Center in 2018. Chief medical officer José Baselga, MD, PhD, failed to report millions of dollars of industry payments and ownership interests in journal articles he wrote or cowrote over several years.
In February 2019, leaders from journals, academia, medical societies, and other institutions gathered in Washington for a closed-door meeting to hash out various disclosure related issues.
Hosted by the Association of American Medical Colleges and cosponsored by Memorial Sloan Kettering Cancer Center, ASCO, JAMA, and the Council of Medical Specialty Societies, the meeting led to a series of working groups that are creating additional recommendations “due out soon in 2020,” Heather Pierce, senior director of science policy and regulatory counsel for the AAMC, said in an interview.
Among the questions being discussed: What disclosures should be verified and who should do so? How can disclosures be made more complete and easier for researchers? And, “most importantly,” said Ms. Pierce, how can policy requirements across each of these sectors be aligned so that there’s more coordination and oversight – and with it, public trust?
Some critics of current disclosure policies have called for more reporting of compensation amounts, and Ms. Pierce said that this has been part of cross-sector discussions.
The ICMJE’s proposed form invites, but does not require, authors to indicate what payments were made to them or their institutions. “Part of this is due to the fact that it’s hard to define, let alone agree on, what’s an important amount,” Dr. Taichman said.
A push for registries
The ICMJE is also aiming to make the disclosure process more efficient for authors – and to eliminate inconsistent and incomplete disclosures – by accepting disclosures from web-based repositories, according to the editorial. Repositories allow authors to maintain an inventory of their relationships and activities and then create electronic disclosures that are tailored to the requirements of the ICMJE, medical societies, and other entities.
The AAMC-run repository, called Convey, is consistent with ICMJE reporting requirements and other criteria (e.g., there are no fees for individuals to enter, store, or export their data), but the development of other repositories may be helpful “for meeting regional, linguistic, and regulatory needs” of authors across the world, the editorial stated.
The Annals of Internal Medicine and the New England Journal of Medicine are both currently collecting disclosures through Convey. The platform was born from discussions that followed a 2009 Institute of Medicine report on conflicts of interest.
Signers of the ICMJE editorial include representatives of the National Library of Medicine and the World Association of Medical Editors, in addition to editors in chief and other leaders of the ICMJE member journals.
A group of leading medical journal editors is seeking to improve the completeness and transparency of financial disclosure reporting with a proposed new disclosure form that puts more onus on readers to decide whether relationships and activities should influence how they view published papers.
The proposed changes are described in an editorial published simultaneously today in the Annals of Internal Medicine, British Medical Journal, Journal of the American Medical Association, The Lancet, New England Journal of Medicine, and several other journals whose editors are members of the International Committee of Medical Journal Editors (ICMJE).
“While no approach to disclosure will be perfect or foolproof, we hope the changes we propose will help promote transparency and trust,” the editorial stated (Ann Intern Med. 2020 Jan 27. doi: 10.7326/M19-3933).
The ICMJE adopted its currently used electronic form – the “ICMJE Form for the Disclosure of Potential Conflicts of Interest” – 10 years ago in an effort to create some uniformity amidst a patchwork of differing disclosure requirements for authors.
It’s not known how many journals outside of the ICMJE’s member journals routinely use the disclosure form, but the organization’s website houses an extensive list of journals whose editors or publishers have requested to be listed as following the ICMJE’s recommendations for editing, reporting, and publishing, including those concerning disclosures. The ICMJE does not “certify” journals. The full set of recommendations was updated in December 2019.
Most authors are committed to transparent reporting, but “opinions differ over which relationships or activities to report,” the editorial stated.
An author might choose to omit an item that others deem important because of a difference in opinion regarding “relevance,” confusion over definitions, or a simple oversight. Some authors may be “concerned that readers will interpret the listing of any item as a ‘potential conflict of interest’ as indicative of problematic influence and wrongdoing,” the editorial stated.
The revised form, like the current one, asks authors to disclose relationships and activities that are directly related to the reported work, as well as those that are topically related (within the broadly defined field addressed in the work). But unlike the current form, the new version provides a checklist of relationships and activities and asks authors to check ‘yes’ or ‘no’ for each one (and to name them when the answer is ‘yes’).
Items in the checklist include grants, payments/honoraria for lectures, patents issued or planned, stock/stock options, and leadership or fiduciary roles in committees, boards, or societies.
The proposed new form makes no mention of “potential conflicts of interest” or “relevancy,” per say. Authors aren’t asked to determine what might be interpreted as a potential conflict of interest, but instead are asked for a “complete listing” of what readers may find “pertinent” to their work.
“We’re trying to move away from calling everything a [potential] ‘conflict,’ ” Darren B. Taichman, MD, PhD, secretary of ICMJE and executive editor of the Annals of Internal Medicine, said in an interview. “We want to remove for authors the concern or stigma, if you will, that anything listed on a form implies that there is something wrong, because that’s just not true. … We want readers to decide what relationships are important as they interpret the work.”
Dr. Taichman said in the interview that the ICMJE’s updating of the form was more a function of “good housekeeping” and continuous appreciation of disclosure as an important issue, rather than any one specific issue, such as concern over a “relevancy” approach to disclosures.
The ICMJE is seeking feedback about its proposed form, which is available with a link for providing comments, at www.icmje.org.
Broader national efforts
Editors and others have been increasingly moving, however, toward asking for more complete disclosures where authors aren’t asked to judge “relevancy” and where readers can make decisions on their own. The American Society of Clinical Oncology, which produces the Journal of Clinical Oncology (JCO) as well as practice guidelines and continuing medical education programs, moved about 5 years ago to a system of general disclosure that asks physicians and others to disclose all financial interests and industry relationships, with no qualifiers.
Earlier in January 2020, the Accreditation Council for Continuing Medical Education issued proposed revisions to its Standards for Integrity and Independence in Accredited Continuing Education. These revisions, which are open for comment, require CME providers to collect disclosure information about all financial relationships of speakers and presenters. It’s up to the CME provider to then determine which relationships are relevant, according to the proposed document.
More change is on the way, as disclosure issues are being deliberated nationally in the wake of a highly publicized disclosure failure at Memorial Sloan Kettering Cancer Center in 2018. Chief medical officer José Baselga, MD, PhD, failed to report millions of dollars of industry payments and ownership interests in journal articles he wrote or cowrote over several years.
In February 2019, leaders from journals, academia, medical societies, and other institutions gathered in Washington for a closed-door meeting to hash out various disclosure related issues.
Hosted by the Association of American Medical Colleges and cosponsored by Memorial Sloan Kettering Cancer Center, ASCO, JAMA, and the Council of Medical Specialty Societies, the meeting led to a series of working groups that are creating additional recommendations “due out soon in 2020,” Heather Pierce, senior director of science policy and regulatory counsel for the AAMC, said in an interview.
Among the questions being discussed: What disclosures should be verified and who should do so? How can disclosures be made more complete and easier for researchers? And, “most importantly,” said Ms. Pierce, how can policy requirements across each of these sectors be aligned so that there’s more coordination and oversight – and with it, public trust?
Some critics of current disclosure policies have called for more reporting of compensation amounts, and Ms. Pierce said that this has been part of cross-sector discussions.
The ICMJE’s proposed form invites, but does not require, authors to indicate what payments were made to them or their institutions. “Part of this is due to the fact that it’s hard to define, let alone agree on, what’s an important amount,” Dr. Taichman said.
A push for registries
The ICMJE is also aiming to make the disclosure process more efficient for authors – and to eliminate inconsistent and incomplete disclosures – by accepting disclosures from web-based repositories, according to the editorial. Repositories allow authors to maintain an inventory of their relationships and activities and then create electronic disclosures that are tailored to the requirements of the ICMJE, medical societies, and other entities.
The AAMC-run repository, called Convey, is consistent with ICMJE reporting requirements and other criteria (e.g., there are no fees for individuals to enter, store, or export their data), but the development of other repositories may be helpful “for meeting regional, linguistic, and regulatory needs” of authors across the world, the editorial stated.
The Annals of Internal Medicine and the New England Journal of Medicine are both currently collecting disclosures through Convey. The platform was born from discussions that followed a 2009 Institute of Medicine report on conflicts of interest.
Signers of the ICMJE editorial include representatives of the National Library of Medicine and the World Association of Medical Editors, in addition to editors in chief and other leaders of the ICMJE member journals.
FROM ANNALS OF INTERNAL MEDICINE
What 2019’s top five CAD trials tell us
SNOWMASS, COLO. – A repeated theme threading through much of one prominent interventional cardiologist’s personal list of the top five coronary artery disease (CAD) trials of the past year is that aspirin is very often more trouble than it’s worth.
“For some years I’ve been concerned that the only thing that aspirin does [in patients after percutaneous coronary intervention] is increase your risk of bleeding. It doesn’t really provide any additional ischemic protection,” Malcolm R. Bell, MBBS, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’ll remind you that, when we go back to the early stent days, observed Dr. Bell, professor of medicine and vice chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
Here are the key takeaway messages from his five most important randomized trials in CAD during the last year.
AUGUSTUS
For years, cardiologists have grappled with how to best manage high-cardiovascular-risk patients with atrial fibrillation who seem like they might benefit from triple-antithrombotic therapy. AUGUSTUS supplied the answer: Don’t do it. Skip the aspirin and turn instead to a P2Y12 inhibitor plus a non–vitamin K antagonist oral anticoagulant (NOAC), rather than warfarin.
“I would like you to think of triple therapy as a triple threat. That’s really what triple therapy is all about”– a three-pronged threat to patient safety, Dr. Bell commented.
In AUGUSTUS, 4,614 patients with atrial fibrillation and CAD with an acute coronary syndrome (ACS) and/or percutaneous coronary intervention (PCI) in 33 countries were placed on a P2Y12 inhibitor – most often clopidogrel – and randomized double blind to either apixaban (Eliquis) or warfarin, and further to aspirin or placebo, for 6 months of antithrombotic therapy. The strategy of a P2Y12 inhibitor and apixaban without aspirin was the clear winner, resulting in significantly less major bleeding, mortality, and hospitalizations than treatment with a P2Y12 inhibitor and warfarin, with or without aspirin. Most importantly, ischemic event rates didn’t differ between the apixaban and warfarin groups. And patients randomized to aspirin had rates of ischemic events and death or hospitalization similar to placebo-treated controls, meaning aspirin accomplished nothing (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
Dr. Bell noted that a meta-analysis of AUGUSTUS and three smaller randomized trials including more than 10,000 AUGUSTUS-type patients with atrial fibrillation concluded that a treatment strategy utilizing a NOAC and a P2Y12 inhibitor resulted in less bleeding than warfarin plus DAPT, and at no cost in terms of excess ischemic events. Moreover, regimens without aspirin resulted in less intracranial and other major bleeding without any difference in major adverse cardiovascular events (JAMA Cardiol. 2019 Jun 19. doi: 10.1001/jamacardio.2019.1880).
A key message of these four trials is that a NOAC is preferable to warfarin, so much so that, in high-risk patients who are already on warfarin, it’s worth considering a switch to a NOAC.
“And we should really be avoiding DAPT,” Dr. Bell added.
How soon after an ACS and/or PCI should patients with atrial fibrillation stop taking aspirin?
“In AUGUSTUS, randomization occurred at a median of 6 days, so we know that half the patients stopped their aspirin by then. In our own practice, we’re just dropping the aspirin for the most part before the patient leaves the hospital. I think if you leave them with instructions to stop the aspirin in a week’s time or a month’s time it just leads to confusion. And we should also remember that half of the major bleeding after PCI or ACS happens in the first 30 days, so it doesn’t make a lot of sense to say that we should continue it for a month and then drop it,” according to the cardiologist.
SMART-CHOICE and STOPDAPT-2
These two large multicenter studies demonstrate that DAPT can safely be stopped early if needed. SMART-CHOICE from South Korea and STOPDAPT-2 from Japan each randomized roughly 3,000 patients undergoing PCI to 12 months of DAPT or to DAPT for only 3 months or 1 month, respectively, at which point the aspirin was dropped and patients in the abbreviated DAPT arm continued on P2Y12 inhibitor monotherapy, mostly clopidogrel, for the remainder of the 12 months. In the Japanese STOPDAPT-2 trial, 1 month of DAPT proved superior to 12 months of DAPT for the primary composite endpoint of cardiovascular death, MI, stroke, definite stent thrombosis, or major or minor bleeding at 12 months (JAMA. 2019 Jun 25;321[24]:2414-27). In the South Korean SMART-CHOICE trial, 3 months of DAPT was noninferior to 12 months for major adverse cardiac and cerebrovascular events, and superior in terms of bleeding risk (JAMA. 2019 Jun 25;321[24]:2428-37). Of note, roughly half of patients in the two trials were lower-risk individuals undergoing PCI for stable angina.
Dr. Bell noted that, while the TWILIGHT trial (Ticagrelor With or Without Aspirin in High-Risk Patients After PCI) didn’t make his top-five list, it certainly fits well with the two East Asian studies. The TWILIGHT investigators randomized more than 7,000 patients to 12 months of DAPT or discontinuation of aspirin after 3 months. The result: a lower incidence of clinically relevant bleeding with ticagrelor monotherapy, and with no increased risk of death, MI, or stroke, compared with 12 months of DAPT (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
“Again, I would just question what the added value of aspirin is here,” Dr. Bell commented. “Many interventional cardiologists are absolutely terrified of their patients having stent thrombosis, but with second-generation drug-eluting stents – the stents we’re putting in day in and day out – the risk of stent thrombosis is less than 1%. And in these two trials it was less than 0.5%. There’s more risk of having major bleeding events than there is of ischemia, so I think the balance is in favor of preventing bleeding. We know that major bleeding predicts short- and long-term mortality.”
COLCOT
This double-blind trial randomized 4,745 patients within 30 days post MI to low-dose colchicine or placebo on top of excellent rates of background guideline-directed medical therapy. The goal was to see if this anti-inflammatory agent could reduce cardiovascular events independent of any lipid-lowering effect, as was earlier seen with canakinumab in the CANTOS trial. It did so to a statistically significant but relatively modest degree, with a 5.5% rate of the composite cardiovascular events endpoint in the colchicine group and 7.1% in placebo-treated controls (N Engl J Med. 2019 Dec 26;381[26]:2497-505). But Dr. Bell was unimpressed.
“All-cause mortality was identical at 1.8% in both groups. So colchicine is not saving lives. In fact, the only real differences were in stroke – but the study wasn’t powered to look at stroke – and in urgent hospitalization for angina leading to revascularization, which is a soft endpoint,” he observed.
Plus, 2.5% of patients were lost to follow-up, which Dr. Bell considers “a little concerning” in a trial conducted in the current era.
“In my opinion, the evidence that colchicine is effective is weak, and I don’t think really supports the drug’s routine use post MI. We already send these patients out on numerous medications. We have to think about cost/benefit, and if a patient asks me: ‘Is this going to prevent another heart attack or make me live longer?’ I think the unequivocal answer is no,” he said.
These days colchicine is no longer an inexpensive drug, either, at an average cost of $300-$400 per month, the cardiologist added.
COMPLETE
This study randomized more than 4,000 patients with ST-segment elevation MI (STEMI) and multivessel disease to primary PCI of the culprit lesion only or to staged complete revascularization via PCI of all angiographically significant nonculprit lesions. Complete revascularization proved to be the superior strategy, with a 26% reduction in the risk of the composite of cardiovascular death or MI at a median of 3 years (N Engl J Med. 2019 Oct 10;381[15]:1411-21).
The optimal timing of the staged procedure remains unclear, since the study didn’t specify a protocol.
“I’m still a bit uncomfortable doing multivessel PCI at 2 o’clock in the morning in the setting of STEMI in someone I’ve never met before. I don’t think there’s a rush to do anything then. Often in this middle-of-the-night stuff, we miss things or we overinterpret things. I think it’s better to let the patient cool down, get to know them,” according to Dr. Bell.
EXCEL
Publication of the 5-year outcomes of the largest-ever randomized trial of PCI versus coronary artery bypass grafting (CABG) for left main coronary disease has led to furious controversy, with a few of the surgeons involved in the study opting to publically broadcast allegations of misbehavior on the part of the interventional cardiologist study leadership, charges that have been strongly denied.
The actual results are in line with findings reported from smaller randomized trials. At 5 years in EXCEL, there was no significant difference between the PCI and CABG groups in the primary composite endpoint of death, cerebrovascular accident, or MI (N Engl J Med. 2019 Nov 7;381[19]:1820-30). The all-cause mortality rate was 13% in the PCI arm and 9.9% with CABG, but this finding comes with a caveat.
“I’ll emphasize this trial was never powered to look at mortality. Neither were any of the other randomized trials. On the other hand, I don’t think you can necessarily ignore the finding of an absolute 3.1% difference,” Dr. Bell said.
PCI and CABG are both very good, mature therapies for left main disease, in his view. In the setting of more-complex coronary disease in younger patients, he often views the complete revascularization offered by surgery as the preferred option. On the other hand, in an 80-year-old with severe comorbidities, clearly PCI is attractive.
He considers the highly public nature of this interspecialty spat a regrettable black eye for the entire field of cardiovascular medicine. And he predicted that an ongoing outside neutral-party review of the study data and procedures will conclude, as he has, “there was no malfeasance at all in the trial.”
Dr. Bell reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – A repeated theme threading through much of one prominent interventional cardiologist’s personal list of the top five coronary artery disease (CAD) trials of the past year is that aspirin is very often more trouble than it’s worth.
“For some years I’ve been concerned that the only thing that aspirin does [in patients after percutaneous coronary intervention] is increase your risk of bleeding. It doesn’t really provide any additional ischemic protection,” Malcolm R. Bell, MBBS, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’ll remind you that, when we go back to the early stent days, observed Dr. Bell, professor of medicine and vice chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
Here are the key takeaway messages from his five most important randomized trials in CAD during the last year.
AUGUSTUS
For years, cardiologists have grappled with how to best manage high-cardiovascular-risk patients with atrial fibrillation who seem like they might benefit from triple-antithrombotic therapy. AUGUSTUS supplied the answer: Don’t do it. Skip the aspirin and turn instead to a P2Y12 inhibitor plus a non–vitamin K antagonist oral anticoagulant (NOAC), rather than warfarin.
“I would like you to think of triple therapy as a triple threat. That’s really what triple therapy is all about”– a three-pronged threat to patient safety, Dr. Bell commented.
In AUGUSTUS, 4,614 patients with atrial fibrillation and CAD with an acute coronary syndrome (ACS) and/or percutaneous coronary intervention (PCI) in 33 countries were placed on a P2Y12 inhibitor – most often clopidogrel – and randomized double blind to either apixaban (Eliquis) or warfarin, and further to aspirin or placebo, for 6 months of antithrombotic therapy. The strategy of a P2Y12 inhibitor and apixaban without aspirin was the clear winner, resulting in significantly less major bleeding, mortality, and hospitalizations than treatment with a P2Y12 inhibitor and warfarin, with or without aspirin. Most importantly, ischemic event rates didn’t differ between the apixaban and warfarin groups. And patients randomized to aspirin had rates of ischemic events and death or hospitalization similar to placebo-treated controls, meaning aspirin accomplished nothing (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
Dr. Bell noted that a meta-analysis of AUGUSTUS and three smaller randomized trials including more than 10,000 AUGUSTUS-type patients with atrial fibrillation concluded that a treatment strategy utilizing a NOAC and a P2Y12 inhibitor resulted in less bleeding than warfarin plus DAPT, and at no cost in terms of excess ischemic events. Moreover, regimens without aspirin resulted in less intracranial and other major bleeding without any difference in major adverse cardiovascular events (JAMA Cardiol. 2019 Jun 19. doi: 10.1001/jamacardio.2019.1880).
A key message of these four trials is that a NOAC is preferable to warfarin, so much so that, in high-risk patients who are already on warfarin, it’s worth considering a switch to a NOAC.
“And we should really be avoiding DAPT,” Dr. Bell added.
How soon after an ACS and/or PCI should patients with atrial fibrillation stop taking aspirin?
“In AUGUSTUS, randomization occurred at a median of 6 days, so we know that half the patients stopped their aspirin by then. In our own practice, we’re just dropping the aspirin for the most part before the patient leaves the hospital. I think if you leave them with instructions to stop the aspirin in a week’s time or a month’s time it just leads to confusion. And we should also remember that half of the major bleeding after PCI or ACS happens in the first 30 days, so it doesn’t make a lot of sense to say that we should continue it for a month and then drop it,” according to the cardiologist.
SMART-CHOICE and STOPDAPT-2
These two large multicenter studies demonstrate that DAPT can safely be stopped early if needed. SMART-CHOICE from South Korea and STOPDAPT-2 from Japan each randomized roughly 3,000 patients undergoing PCI to 12 months of DAPT or to DAPT for only 3 months or 1 month, respectively, at which point the aspirin was dropped and patients in the abbreviated DAPT arm continued on P2Y12 inhibitor monotherapy, mostly clopidogrel, for the remainder of the 12 months. In the Japanese STOPDAPT-2 trial, 1 month of DAPT proved superior to 12 months of DAPT for the primary composite endpoint of cardiovascular death, MI, stroke, definite stent thrombosis, or major or minor bleeding at 12 months (JAMA. 2019 Jun 25;321[24]:2414-27). In the South Korean SMART-CHOICE trial, 3 months of DAPT was noninferior to 12 months for major adverse cardiac and cerebrovascular events, and superior in terms of bleeding risk (JAMA. 2019 Jun 25;321[24]:2428-37). Of note, roughly half of patients in the two trials were lower-risk individuals undergoing PCI for stable angina.
Dr. Bell noted that, while the TWILIGHT trial (Ticagrelor With or Without Aspirin in High-Risk Patients After PCI) didn’t make his top-five list, it certainly fits well with the two East Asian studies. The TWILIGHT investigators randomized more than 7,000 patients to 12 months of DAPT or discontinuation of aspirin after 3 months. The result: a lower incidence of clinically relevant bleeding with ticagrelor monotherapy, and with no increased risk of death, MI, or stroke, compared with 12 months of DAPT (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
“Again, I would just question what the added value of aspirin is here,” Dr. Bell commented. “Many interventional cardiologists are absolutely terrified of their patients having stent thrombosis, but with second-generation drug-eluting stents – the stents we’re putting in day in and day out – the risk of stent thrombosis is less than 1%. And in these two trials it was less than 0.5%. There’s more risk of having major bleeding events than there is of ischemia, so I think the balance is in favor of preventing bleeding. We know that major bleeding predicts short- and long-term mortality.”
COLCOT
This double-blind trial randomized 4,745 patients within 30 days post MI to low-dose colchicine or placebo on top of excellent rates of background guideline-directed medical therapy. The goal was to see if this anti-inflammatory agent could reduce cardiovascular events independent of any lipid-lowering effect, as was earlier seen with canakinumab in the CANTOS trial. It did so to a statistically significant but relatively modest degree, with a 5.5% rate of the composite cardiovascular events endpoint in the colchicine group and 7.1% in placebo-treated controls (N Engl J Med. 2019 Dec 26;381[26]:2497-505). But Dr. Bell was unimpressed.
“All-cause mortality was identical at 1.8% in both groups. So colchicine is not saving lives. In fact, the only real differences were in stroke – but the study wasn’t powered to look at stroke – and in urgent hospitalization for angina leading to revascularization, which is a soft endpoint,” he observed.
Plus, 2.5% of patients were lost to follow-up, which Dr. Bell considers “a little concerning” in a trial conducted in the current era.
“In my opinion, the evidence that colchicine is effective is weak, and I don’t think really supports the drug’s routine use post MI. We already send these patients out on numerous medications. We have to think about cost/benefit, and if a patient asks me: ‘Is this going to prevent another heart attack or make me live longer?’ I think the unequivocal answer is no,” he said.
These days colchicine is no longer an inexpensive drug, either, at an average cost of $300-$400 per month, the cardiologist added.
COMPLETE
This study randomized more than 4,000 patients with ST-segment elevation MI (STEMI) and multivessel disease to primary PCI of the culprit lesion only or to staged complete revascularization via PCI of all angiographically significant nonculprit lesions. Complete revascularization proved to be the superior strategy, with a 26% reduction in the risk of the composite of cardiovascular death or MI at a median of 3 years (N Engl J Med. 2019 Oct 10;381[15]:1411-21).
The optimal timing of the staged procedure remains unclear, since the study didn’t specify a protocol.
“I’m still a bit uncomfortable doing multivessel PCI at 2 o’clock in the morning in the setting of STEMI in someone I’ve never met before. I don’t think there’s a rush to do anything then. Often in this middle-of-the-night stuff, we miss things or we overinterpret things. I think it’s better to let the patient cool down, get to know them,” according to Dr. Bell.
EXCEL
Publication of the 5-year outcomes of the largest-ever randomized trial of PCI versus coronary artery bypass grafting (CABG) for left main coronary disease has led to furious controversy, with a few of the surgeons involved in the study opting to publically broadcast allegations of misbehavior on the part of the interventional cardiologist study leadership, charges that have been strongly denied.
The actual results are in line with findings reported from smaller randomized trials. At 5 years in EXCEL, there was no significant difference between the PCI and CABG groups in the primary composite endpoint of death, cerebrovascular accident, or MI (N Engl J Med. 2019 Nov 7;381[19]:1820-30). The all-cause mortality rate was 13% in the PCI arm and 9.9% with CABG, but this finding comes with a caveat.
“I’ll emphasize this trial was never powered to look at mortality. Neither were any of the other randomized trials. On the other hand, I don’t think you can necessarily ignore the finding of an absolute 3.1% difference,” Dr. Bell said.
PCI and CABG are both very good, mature therapies for left main disease, in his view. In the setting of more-complex coronary disease in younger patients, he often views the complete revascularization offered by surgery as the preferred option. On the other hand, in an 80-year-old with severe comorbidities, clearly PCI is attractive.
He considers the highly public nature of this interspecialty spat a regrettable black eye for the entire field of cardiovascular medicine. And he predicted that an ongoing outside neutral-party review of the study data and procedures will conclude, as he has, “there was no malfeasance at all in the trial.”
Dr. Bell reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – A repeated theme threading through much of one prominent interventional cardiologist’s personal list of the top five coronary artery disease (CAD) trials of the past year is that aspirin is very often more trouble than it’s worth.
“For some years I’ve been concerned that the only thing that aspirin does [in patients after percutaneous coronary intervention] is increase your risk of bleeding. It doesn’t really provide any additional ischemic protection,” Malcolm R. Bell, MBBS, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’ll remind you that, when we go back to the early stent days, observed Dr. Bell, professor of medicine and vice chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
Here are the key takeaway messages from his five most important randomized trials in CAD during the last year.
AUGUSTUS
For years, cardiologists have grappled with how to best manage high-cardiovascular-risk patients with atrial fibrillation who seem like they might benefit from triple-antithrombotic therapy. AUGUSTUS supplied the answer: Don’t do it. Skip the aspirin and turn instead to a P2Y12 inhibitor plus a non–vitamin K antagonist oral anticoagulant (NOAC), rather than warfarin.
“I would like you to think of triple therapy as a triple threat. That’s really what triple therapy is all about”– a three-pronged threat to patient safety, Dr. Bell commented.
In AUGUSTUS, 4,614 patients with atrial fibrillation and CAD with an acute coronary syndrome (ACS) and/or percutaneous coronary intervention (PCI) in 33 countries were placed on a P2Y12 inhibitor – most often clopidogrel – and randomized double blind to either apixaban (Eliquis) or warfarin, and further to aspirin or placebo, for 6 months of antithrombotic therapy. The strategy of a P2Y12 inhibitor and apixaban without aspirin was the clear winner, resulting in significantly less major bleeding, mortality, and hospitalizations than treatment with a P2Y12 inhibitor and warfarin, with or without aspirin. Most importantly, ischemic event rates didn’t differ between the apixaban and warfarin groups. And patients randomized to aspirin had rates of ischemic events and death or hospitalization similar to placebo-treated controls, meaning aspirin accomplished nothing (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
Dr. Bell noted that a meta-analysis of AUGUSTUS and three smaller randomized trials including more than 10,000 AUGUSTUS-type patients with atrial fibrillation concluded that a treatment strategy utilizing a NOAC and a P2Y12 inhibitor resulted in less bleeding than warfarin plus DAPT, and at no cost in terms of excess ischemic events. Moreover, regimens without aspirin resulted in less intracranial and other major bleeding without any difference in major adverse cardiovascular events (JAMA Cardiol. 2019 Jun 19. doi: 10.1001/jamacardio.2019.1880).
A key message of these four trials is that a NOAC is preferable to warfarin, so much so that, in high-risk patients who are already on warfarin, it’s worth considering a switch to a NOAC.
“And we should really be avoiding DAPT,” Dr. Bell added.
How soon after an ACS and/or PCI should patients with atrial fibrillation stop taking aspirin?
“In AUGUSTUS, randomization occurred at a median of 6 days, so we know that half the patients stopped their aspirin by then. In our own practice, we’re just dropping the aspirin for the most part before the patient leaves the hospital. I think if you leave them with instructions to stop the aspirin in a week’s time or a month’s time it just leads to confusion. And we should also remember that half of the major bleeding after PCI or ACS happens in the first 30 days, so it doesn’t make a lot of sense to say that we should continue it for a month and then drop it,” according to the cardiologist.
SMART-CHOICE and STOPDAPT-2
These two large multicenter studies demonstrate that DAPT can safely be stopped early if needed. SMART-CHOICE from South Korea and STOPDAPT-2 from Japan each randomized roughly 3,000 patients undergoing PCI to 12 months of DAPT or to DAPT for only 3 months or 1 month, respectively, at which point the aspirin was dropped and patients in the abbreviated DAPT arm continued on P2Y12 inhibitor monotherapy, mostly clopidogrel, for the remainder of the 12 months. In the Japanese STOPDAPT-2 trial, 1 month of DAPT proved superior to 12 months of DAPT for the primary composite endpoint of cardiovascular death, MI, stroke, definite stent thrombosis, or major or minor bleeding at 12 months (JAMA. 2019 Jun 25;321[24]:2414-27). In the South Korean SMART-CHOICE trial, 3 months of DAPT was noninferior to 12 months for major adverse cardiac and cerebrovascular events, and superior in terms of bleeding risk (JAMA. 2019 Jun 25;321[24]:2428-37). Of note, roughly half of patients in the two trials were lower-risk individuals undergoing PCI for stable angina.
Dr. Bell noted that, while the TWILIGHT trial (Ticagrelor With or Without Aspirin in High-Risk Patients After PCI) didn’t make his top-five list, it certainly fits well with the two East Asian studies. The TWILIGHT investigators randomized more than 7,000 patients to 12 months of DAPT or discontinuation of aspirin after 3 months. The result: a lower incidence of clinically relevant bleeding with ticagrelor monotherapy, and with no increased risk of death, MI, or stroke, compared with 12 months of DAPT (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
“Again, I would just question what the added value of aspirin is here,” Dr. Bell commented. “Many interventional cardiologists are absolutely terrified of their patients having stent thrombosis, but with second-generation drug-eluting stents – the stents we’re putting in day in and day out – the risk of stent thrombosis is less than 1%. And in these two trials it was less than 0.5%. There’s more risk of having major bleeding events than there is of ischemia, so I think the balance is in favor of preventing bleeding. We know that major bleeding predicts short- and long-term mortality.”
COLCOT
This double-blind trial randomized 4,745 patients within 30 days post MI to low-dose colchicine or placebo on top of excellent rates of background guideline-directed medical therapy. The goal was to see if this anti-inflammatory agent could reduce cardiovascular events independent of any lipid-lowering effect, as was earlier seen with canakinumab in the CANTOS trial. It did so to a statistically significant but relatively modest degree, with a 5.5% rate of the composite cardiovascular events endpoint in the colchicine group and 7.1% in placebo-treated controls (N Engl J Med. 2019 Dec 26;381[26]:2497-505). But Dr. Bell was unimpressed.
“All-cause mortality was identical at 1.8% in both groups. So colchicine is not saving lives. In fact, the only real differences were in stroke – but the study wasn’t powered to look at stroke – and in urgent hospitalization for angina leading to revascularization, which is a soft endpoint,” he observed.
Plus, 2.5% of patients were lost to follow-up, which Dr. Bell considers “a little concerning” in a trial conducted in the current era.
“In my opinion, the evidence that colchicine is effective is weak, and I don’t think really supports the drug’s routine use post MI. We already send these patients out on numerous medications. We have to think about cost/benefit, and if a patient asks me: ‘Is this going to prevent another heart attack or make me live longer?’ I think the unequivocal answer is no,” he said.
These days colchicine is no longer an inexpensive drug, either, at an average cost of $300-$400 per month, the cardiologist added.
COMPLETE
This study randomized more than 4,000 patients with ST-segment elevation MI (STEMI) and multivessel disease to primary PCI of the culprit lesion only or to staged complete revascularization via PCI of all angiographically significant nonculprit lesions. Complete revascularization proved to be the superior strategy, with a 26% reduction in the risk of the composite of cardiovascular death or MI at a median of 3 years (N Engl J Med. 2019 Oct 10;381[15]:1411-21).
The optimal timing of the staged procedure remains unclear, since the study didn’t specify a protocol.
“I’m still a bit uncomfortable doing multivessel PCI at 2 o’clock in the morning in the setting of STEMI in someone I’ve never met before. I don’t think there’s a rush to do anything then. Often in this middle-of-the-night stuff, we miss things or we overinterpret things. I think it’s better to let the patient cool down, get to know them,” according to Dr. Bell.
EXCEL
Publication of the 5-year outcomes of the largest-ever randomized trial of PCI versus coronary artery bypass grafting (CABG) for left main coronary disease has led to furious controversy, with a few of the surgeons involved in the study opting to publically broadcast allegations of misbehavior on the part of the interventional cardiologist study leadership, charges that have been strongly denied.
The actual results are in line with findings reported from smaller randomized trials. At 5 years in EXCEL, there was no significant difference between the PCI and CABG groups in the primary composite endpoint of death, cerebrovascular accident, or MI (N Engl J Med. 2019 Nov 7;381[19]:1820-30). The all-cause mortality rate was 13% in the PCI arm and 9.9% with CABG, but this finding comes with a caveat.
“I’ll emphasize this trial was never powered to look at mortality. Neither were any of the other randomized trials. On the other hand, I don’t think you can necessarily ignore the finding of an absolute 3.1% difference,” Dr. Bell said.
PCI and CABG are both very good, mature therapies for left main disease, in his view. In the setting of more-complex coronary disease in younger patients, he often views the complete revascularization offered by surgery as the preferred option. On the other hand, in an 80-year-old with severe comorbidities, clearly PCI is attractive.
He considers the highly public nature of this interspecialty spat a regrettable black eye for the entire field of cardiovascular medicine. And he predicted that an ongoing outside neutral-party review of the study data and procedures will conclude, as he has, “there was no malfeasance at all in the trial.”
Dr. Bell reported having no financial conflicts regarding his presentation.
REPORTING FROM ACC SNOWMASS 2020
BP levels during endovascular stroke therapy affect neurologic outcomes
For patients with acute ischemic stroke, prolonged durations of blood pressure above or below certain thresholds during endovascular therapy may be linked to poor functional outcome, results of a retrospective study suggest.
Mean arterial blood pressure (MABP) lower than 70 mm Hg for 10 minutes or more, or higher than 90 mm Hg for 45 minutes or more, represented “critical thresholds” associated with worse neurologic outcomes, the study authors wrote in JAMA Neurology.
“These results suggest MABP may be a modifiable therapeutic target to prevent or reduce poor functional outcome in patients undergoing endovascular therapy for acute ischemic stroke, and that MABP should possibly be maintained within such narrow limits, wrote the authors, led by Mads Rasmussen, MD, PhD, of the department of anesthesia at Aarhus (Denmark) University Hospital.
The findings come from an analysis of BP data from 365 patients with acute ischemic stroke enrolled in three randomized trials evaluating different strategies for anesthesia. Among those patients, the mean age was approximately 71 years, and about 45% were women.
The investigators looked at a variety of BP-related variables during endovascular therapy to assess their impact on functional outcome, based on modified Rankin Scale (mRS) scores at 90 days.
Having an MABP below 70 mm Hg for a cumulative time of at least 10 minutes substantially increased odds of higher 90-day mRS scores (odds ratio, 1.51; 95% confidence interval, 1.02-2.22), according to Dr. Rasmussen and colleagues. The number needed to harm (NNH) at this threshold was 10; in other words, to harm 1 patient, 10 patients are needed with procedural MABP below 70 mm Hg for at least 10 minutes.
Likewise, having an MABP above 90 mm Hg for a cumulated time of at least 45 minutes significantly increased odds of higher 90-day mRS scores, with an OR of 1.49 (95% CI, 1.11-2.02) and a number needed to harm of 10.
Odds of shifting toward a worse neurologic outcome increased by 62% for every continuous 10 minutes of MABP below 70 mm Hg, and by 8% for every continuous 10 minutes above 90 mm Hg.
The maximum MABP during the procedure was significantly associated with neurologic outcomes in the study, while by contrast, maximum procedural systolic BP was not, according to the investigators.
In general, the study findings suggest that MABP is “more sensitive” than systolic BP when assessing hypotension and hypertension in these patients. However, these findings are subject to a number of limitations, the investigators wrote, including the retrospective nature of the analysis and the selected group of patients enrolled in studies designed to evaluate anesthesia strategies, not hemodynamic management.
“Randomized studies are needed to determine the optimal blood pressure management strategy during endovascular therapy,” the investigators wrote.
Dr. Rasmussen reported grant support from the Health Research Foundation of Central Denmark Region and the National Helicopter Emergency Medical Service Foundation. Coauthors reported receiving grant support from the Novo Nordisk Foundation; a research award from the Patient-Centered Outcomes Research Institute; and personal fees from Abbott Medical Sweden, I4L Innovation for Life, Boehringer Ingelheim, Medtronic, and Zoll.
SOURCE: Rasmussen M et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4838.
For patients with acute ischemic stroke, prolonged durations of blood pressure above or below certain thresholds during endovascular therapy may be linked to poor functional outcome, results of a retrospective study suggest.
Mean arterial blood pressure (MABP) lower than 70 mm Hg for 10 minutes or more, or higher than 90 mm Hg for 45 minutes or more, represented “critical thresholds” associated with worse neurologic outcomes, the study authors wrote in JAMA Neurology.
“These results suggest MABP may be a modifiable therapeutic target to prevent or reduce poor functional outcome in patients undergoing endovascular therapy for acute ischemic stroke, and that MABP should possibly be maintained within such narrow limits, wrote the authors, led by Mads Rasmussen, MD, PhD, of the department of anesthesia at Aarhus (Denmark) University Hospital.
The findings come from an analysis of BP data from 365 patients with acute ischemic stroke enrolled in three randomized trials evaluating different strategies for anesthesia. Among those patients, the mean age was approximately 71 years, and about 45% were women.
The investigators looked at a variety of BP-related variables during endovascular therapy to assess their impact on functional outcome, based on modified Rankin Scale (mRS) scores at 90 days.
Having an MABP below 70 mm Hg for a cumulative time of at least 10 minutes substantially increased odds of higher 90-day mRS scores (odds ratio, 1.51; 95% confidence interval, 1.02-2.22), according to Dr. Rasmussen and colleagues. The number needed to harm (NNH) at this threshold was 10; in other words, to harm 1 patient, 10 patients are needed with procedural MABP below 70 mm Hg for at least 10 minutes.
Likewise, having an MABP above 90 mm Hg for a cumulated time of at least 45 minutes significantly increased odds of higher 90-day mRS scores, with an OR of 1.49 (95% CI, 1.11-2.02) and a number needed to harm of 10.
Odds of shifting toward a worse neurologic outcome increased by 62% for every continuous 10 minutes of MABP below 70 mm Hg, and by 8% for every continuous 10 minutes above 90 mm Hg.
The maximum MABP during the procedure was significantly associated with neurologic outcomes in the study, while by contrast, maximum procedural systolic BP was not, according to the investigators.
In general, the study findings suggest that MABP is “more sensitive” than systolic BP when assessing hypotension and hypertension in these patients. However, these findings are subject to a number of limitations, the investigators wrote, including the retrospective nature of the analysis and the selected group of patients enrolled in studies designed to evaluate anesthesia strategies, not hemodynamic management.
“Randomized studies are needed to determine the optimal blood pressure management strategy during endovascular therapy,” the investigators wrote.
Dr. Rasmussen reported grant support from the Health Research Foundation of Central Denmark Region and the National Helicopter Emergency Medical Service Foundation. Coauthors reported receiving grant support from the Novo Nordisk Foundation; a research award from the Patient-Centered Outcomes Research Institute; and personal fees from Abbott Medical Sweden, I4L Innovation for Life, Boehringer Ingelheim, Medtronic, and Zoll.
SOURCE: Rasmussen M et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4838.
For patients with acute ischemic stroke, prolonged durations of blood pressure above or below certain thresholds during endovascular therapy may be linked to poor functional outcome, results of a retrospective study suggest.
Mean arterial blood pressure (MABP) lower than 70 mm Hg for 10 minutes or more, or higher than 90 mm Hg for 45 minutes or more, represented “critical thresholds” associated with worse neurologic outcomes, the study authors wrote in JAMA Neurology.
“These results suggest MABP may be a modifiable therapeutic target to prevent or reduce poor functional outcome in patients undergoing endovascular therapy for acute ischemic stroke, and that MABP should possibly be maintained within such narrow limits, wrote the authors, led by Mads Rasmussen, MD, PhD, of the department of anesthesia at Aarhus (Denmark) University Hospital.
The findings come from an analysis of BP data from 365 patients with acute ischemic stroke enrolled in three randomized trials evaluating different strategies for anesthesia. Among those patients, the mean age was approximately 71 years, and about 45% were women.
The investigators looked at a variety of BP-related variables during endovascular therapy to assess their impact on functional outcome, based on modified Rankin Scale (mRS) scores at 90 days.
Having an MABP below 70 mm Hg for a cumulative time of at least 10 minutes substantially increased odds of higher 90-day mRS scores (odds ratio, 1.51; 95% confidence interval, 1.02-2.22), according to Dr. Rasmussen and colleagues. The number needed to harm (NNH) at this threshold was 10; in other words, to harm 1 patient, 10 patients are needed with procedural MABP below 70 mm Hg for at least 10 minutes.
Likewise, having an MABP above 90 mm Hg for a cumulated time of at least 45 minutes significantly increased odds of higher 90-day mRS scores, with an OR of 1.49 (95% CI, 1.11-2.02) and a number needed to harm of 10.
Odds of shifting toward a worse neurologic outcome increased by 62% for every continuous 10 minutes of MABP below 70 mm Hg, and by 8% for every continuous 10 minutes above 90 mm Hg.
The maximum MABP during the procedure was significantly associated with neurologic outcomes in the study, while by contrast, maximum procedural systolic BP was not, according to the investigators.
In general, the study findings suggest that MABP is “more sensitive” than systolic BP when assessing hypotension and hypertension in these patients. However, these findings are subject to a number of limitations, the investigators wrote, including the retrospective nature of the analysis and the selected group of patients enrolled in studies designed to evaluate anesthesia strategies, not hemodynamic management.
“Randomized studies are needed to determine the optimal blood pressure management strategy during endovascular therapy,” the investigators wrote.
Dr. Rasmussen reported grant support from the Health Research Foundation of Central Denmark Region and the National Helicopter Emergency Medical Service Foundation. Coauthors reported receiving grant support from the Novo Nordisk Foundation; a research award from the Patient-Centered Outcomes Research Institute; and personal fees from Abbott Medical Sweden, I4L Innovation for Life, Boehringer Ingelheim, Medtronic, and Zoll.
SOURCE: Rasmussen M et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4838.
FROM JAMA NEUROLOGY
Opioid deaths boost donor heart supply
SNOWMASS, COLO. – The tragic opioid epidemic has “one small bright spot”: an expanding pool of eligible donor hearts for transplantation, Akshay S. Desai, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
For decades, the annual volume of heart transplantations performed in the U.S. was static because of the huge mismatch between donor organ supply and demand. But heart transplant volume has increased steadily in the last few years – a result of the opioid epidemic.
Data from the U.S. Organ Procurement and Transplantation Network show that the proportion of donor hearts obtained from individuals who died from drug intoxication climbed from a mere 1.5% in 1999 to 17.6% in 2017, the most recent year for which data are available. Meanwhile, the size of the heart transplant waiting list, which rose year after year in 2009-2015, has since declined (N Engl J Med. 2019 Feb 7;380[6]:597-9).
“What’s amazing is that, even though these patients might have historically been considered high risk in general, the organs recovered from these patients – and particularly the hearts – don’t seem to be any worse in terms of allograft survival than the organs recovered from patients who died from other causes, which are the traditional sources, like blunt head trauma, gunshot wounds, or stroke, that lead to brain death. In general, these organs are useful and do quite well,” according to Dr. Desai, medical director of the cardiomyopathy and heart failure program at Brigham and Women’s Hospital, Boston.
He highlighted several other recent developments in the field of cardiac transplantation that promise to further expand the donor heart pool, including acceptance of hepatitis C–infected donors and organ donation after circulatory rather than brain death. Dr. Desai also drew attention to the unintended perverse consequences of a recent redesign of the U.S. donor heart allocation system and discussed the impressive improvement in clinical outcomes with mechanical circulatory support. He noted that, while relatively few cardiologists practice in the highly specialized centers where heart transplants take place, virtually all cardiologists are affected by advances in heart transplantation since hundreds of thousands of the estimated 7 million Americans with heart failure have advanced disease.
Heart transplantation, he emphasized, is becoming increasingly complex. Recipients are on average older, sicker, and have more comorbidities than in times past. As a result, there is greater need for dual organ transplants: heart/lung, heart/liver, or heart/kidney. Plus, more patients come to transplantation after prior cardiac surgery for implantation of a ventricular assist device, so sensitization to blood products is a growing issue. And, of course, the pool of transplant candidates has expanded.
“We’re now forced to take patients previously considered to have contraindications to transplant; for example, diabetes was a contraindication to transplant in the early years, but now it’s the rule in 35%-40% of our patients who present with advanced heart failure,” the cardiologist noted.
Transplants from HCV-infected donors to uninfected recipients
Hearts and lungs from donors with hepatitis C viremia were traditionally deemed unsuitable for transplant. That’s all changed in the current era of highly effective direct-acting antiviral agents for the treatment of HCV infection.
In the DONATE HCV trial, Dr. Desai’s colleagues at Brigham and Women’s Hospital showed that giving HCV-uninfected recipients of hearts or lungs from HCV-viremic donors a shortened 4-week course of treatment with sofosbuvir-velpatasvir (Epclusa) beginning within a few hours after transplantation uniformly blocked viral replication. Six months after transplantation, none of the study participants had a detectable HCV viral load, and all had excellent graft function (N Engl J Med. 2019 Apr 25;380[17]:1606-17).
“This is effective prevention of HCV infection by aggressive upfront therapy,” Dr. Desai explained. “We can now take organs from HCV-viremic patients and use them in solid organ transplantation. This has led to a skyrocketing increase in donors with HCV infection, and those donations have helped us clear the waiting list.”
Donation after circulatory death
Australian transplant physicians have pioneered the use of donor hearts obtained after circulatory death in individuals with devastating neurologic injury who didn’t quite meet the criteria for brain death, which is the traditional prerequisite. In the new scenario, withdrawal of life-supporting therapy is followed by circulatory death, then the donor heart is procured and preserved via extracorporeal perfusion until transplantation.
The Australians report excellent outcomes, with rates of overall survival and rejection episodes similar to outcomes from brain-dead donors (J Am Coll Cardiol. 2019 Apr 2;73[12]:1447-59). The first U.S. heart transplant involving donation after circulatory death took place at Duke University in Durham, North Carolina. A multicenter U.S. clinical trial of this practice is underway.
If the results are positive and the practice of donation after circulatory death becomes widely implemented, the U.S. heart donor pool could increase by 30%.
Recent overhaul of donor heart allocation system may have backfired
The U.S. donor heart allocation system was redesigned in the fall of 2018 in an effort to reduce waiting times. One of the biggest changes involved breaking down the category with the highest urgency status into three new subcategories based upon sickness. Now, the highest-urgency category is for patients in cardiogenic shock who are supported by extracorporeal membrane oxygenation (ECMO) or other temporary mechanical circulatory support devices.
But an analysis of United Network for Organ Sharing (UNOS) data suggests this change has unintended adverse consequences for clinical outcomes.
Indeed, the investigators reported that the use of ECMO support is fourfold greater in the new system, the use of durable left ventricular assist devices (LVADs) as a bridge to transplant is down, and outcomes are worse. The 180-day rate of freedom from death or retransplantation was 77.9%, down significantly from 93.4% in the former system. In a multivariate analysis, patients transplanted in the new system had an adjusted 2.1-fold increased risk of death or retransplantation (J Heart Lung Transplant. 2020 Jan;39[1]:1-4).
“When you create a new listing system, you create new incentives, and people start to manage patients differently,” Dr. Desai observed. “Increasingly now, the path direct to transplant is through temporary mechanical circulatory support rather than durable mechanical circulatory support. Is that a good idea? We don’t know, but if you look at the best data, those on ECMO or percutaneous VADs have the worst outcomes. So the question of whether we should take the sickest of sick patients directly to transplant as a standard strategy has come under scrutiny.”
Improved durable LVAD technology brings impressive clinical outcomes
Results of the landmark MOMENTUM 3 randomized trial showed that 2-year clinical outcomes with the magnetically levitated centrifugal-flow HeartMate 3 LVAD now rival those of percutaneous mitral valve repair using the MitraClip device. Two-year all-cause mortality in the LVAD recipients was 22% versus 29.1% with the MitraClip in the COAPT trial and 34.9% in the MITRA-FR trial. The HeartMate 3 reduces the hemocompatibility issues that plagued earlier-generation durable LVADs, with resultant lower rates of pump thrombosis, stroke, and GI bleeding. Indeed, the outcomes in MOMENTUM 3 were so good – and so similar – with the HeartMate 3, regardless of whether the intended treatment goal was as a bridge to transplant or as lifelong destination therapy, that the investigators have recently proposed doing away with those distinctions.
“It is possible that use of arbitrary categorizations based on current or future transplant eligibility should be clinically abandoned in favor of a single preimplant strategy: to extend the survival and improve the quality of life of patients with medically refractory heart failure,” according to the investigators (JAMA Cardiol. 2020 Jan 15. doi: 10.1001/jamacardio.2019.5323).
The next step forward in LVAD technology is already on the horizon: a fully implantable device that eliminates the transcutaneous drive-line for the power supply, which is prone to infection and diminishes overall quality of life. This investigational device utilizes wireless coplanar energy transfer, with a coil ring placed around the lung and fixed to the chest wall. The implanted battery provides more than 6 hours of power without a recharge (J Heart Lung Transplant. 2019 Apr;38[4]:339-43).
“The first LVAD patient has gone swimming in Kazakhstan,” according to Dr. Desai.
Myocardial recovery in LVAD recipients remains elusive
The initial hope for LVADs was that they would not only be able to serve as a bridge to transplantation or as lifetime therapy, but that the prolonged unloading of the ventricle would enable potent medical therapy to rescue myocardial function so that the device could eventually be explanted. That does happen, but only rarely. In a large registry study, myocardial recovery occurred in only about 1% of patients on mechanical circulatory support. Attempts to enhance the process by add-on stem cell therapy have thus far been ineffective.
“For the moment, recovery is still a hope, not a reality,” the cardiologist said.
He reported serving as a consultant to more than a dozen pharmaceutical or medical device companies and receiving research grants from Alnylam, AstraZeneca, Bayer Healthcare, MyoKardia, and Novartis.
SNOWMASS, COLO. – The tragic opioid epidemic has “one small bright spot”: an expanding pool of eligible donor hearts for transplantation, Akshay S. Desai, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
For decades, the annual volume of heart transplantations performed in the U.S. was static because of the huge mismatch between donor organ supply and demand. But heart transplant volume has increased steadily in the last few years – a result of the opioid epidemic.
Data from the U.S. Organ Procurement and Transplantation Network show that the proportion of donor hearts obtained from individuals who died from drug intoxication climbed from a mere 1.5% in 1999 to 17.6% in 2017, the most recent year for which data are available. Meanwhile, the size of the heart transplant waiting list, which rose year after year in 2009-2015, has since declined (N Engl J Med. 2019 Feb 7;380[6]:597-9).
“What’s amazing is that, even though these patients might have historically been considered high risk in general, the organs recovered from these patients – and particularly the hearts – don’t seem to be any worse in terms of allograft survival than the organs recovered from patients who died from other causes, which are the traditional sources, like blunt head trauma, gunshot wounds, or stroke, that lead to brain death. In general, these organs are useful and do quite well,” according to Dr. Desai, medical director of the cardiomyopathy and heart failure program at Brigham and Women’s Hospital, Boston.
He highlighted several other recent developments in the field of cardiac transplantation that promise to further expand the donor heart pool, including acceptance of hepatitis C–infected donors and organ donation after circulatory rather than brain death. Dr. Desai also drew attention to the unintended perverse consequences of a recent redesign of the U.S. donor heart allocation system and discussed the impressive improvement in clinical outcomes with mechanical circulatory support. He noted that, while relatively few cardiologists practice in the highly specialized centers where heart transplants take place, virtually all cardiologists are affected by advances in heart transplantation since hundreds of thousands of the estimated 7 million Americans with heart failure have advanced disease.
Heart transplantation, he emphasized, is becoming increasingly complex. Recipients are on average older, sicker, and have more comorbidities than in times past. As a result, there is greater need for dual organ transplants: heart/lung, heart/liver, or heart/kidney. Plus, more patients come to transplantation after prior cardiac surgery for implantation of a ventricular assist device, so sensitization to blood products is a growing issue. And, of course, the pool of transplant candidates has expanded.
“We’re now forced to take patients previously considered to have contraindications to transplant; for example, diabetes was a contraindication to transplant in the early years, but now it’s the rule in 35%-40% of our patients who present with advanced heart failure,” the cardiologist noted.
Transplants from HCV-infected donors to uninfected recipients
Hearts and lungs from donors with hepatitis C viremia were traditionally deemed unsuitable for transplant. That’s all changed in the current era of highly effective direct-acting antiviral agents for the treatment of HCV infection.
In the DONATE HCV trial, Dr. Desai’s colleagues at Brigham and Women’s Hospital showed that giving HCV-uninfected recipients of hearts or lungs from HCV-viremic donors a shortened 4-week course of treatment with sofosbuvir-velpatasvir (Epclusa) beginning within a few hours after transplantation uniformly blocked viral replication. Six months after transplantation, none of the study participants had a detectable HCV viral load, and all had excellent graft function (N Engl J Med. 2019 Apr 25;380[17]:1606-17).
“This is effective prevention of HCV infection by aggressive upfront therapy,” Dr. Desai explained. “We can now take organs from HCV-viremic patients and use them in solid organ transplantation. This has led to a skyrocketing increase in donors with HCV infection, and those donations have helped us clear the waiting list.”
Donation after circulatory death
Australian transplant physicians have pioneered the use of donor hearts obtained after circulatory death in individuals with devastating neurologic injury who didn’t quite meet the criteria for brain death, which is the traditional prerequisite. In the new scenario, withdrawal of life-supporting therapy is followed by circulatory death, then the donor heart is procured and preserved via extracorporeal perfusion until transplantation.
The Australians report excellent outcomes, with rates of overall survival and rejection episodes similar to outcomes from brain-dead donors (J Am Coll Cardiol. 2019 Apr 2;73[12]:1447-59). The first U.S. heart transplant involving donation after circulatory death took place at Duke University in Durham, North Carolina. A multicenter U.S. clinical trial of this practice is underway.
If the results are positive and the practice of donation after circulatory death becomes widely implemented, the U.S. heart donor pool could increase by 30%.
Recent overhaul of donor heart allocation system may have backfired
The U.S. donor heart allocation system was redesigned in the fall of 2018 in an effort to reduce waiting times. One of the biggest changes involved breaking down the category with the highest urgency status into three new subcategories based upon sickness. Now, the highest-urgency category is for patients in cardiogenic shock who are supported by extracorporeal membrane oxygenation (ECMO) or other temporary mechanical circulatory support devices.
But an analysis of United Network for Organ Sharing (UNOS) data suggests this change has unintended adverse consequences for clinical outcomes.
Indeed, the investigators reported that the use of ECMO support is fourfold greater in the new system, the use of durable left ventricular assist devices (LVADs) as a bridge to transplant is down, and outcomes are worse. The 180-day rate of freedom from death or retransplantation was 77.9%, down significantly from 93.4% in the former system. In a multivariate analysis, patients transplanted in the new system had an adjusted 2.1-fold increased risk of death or retransplantation (J Heart Lung Transplant. 2020 Jan;39[1]:1-4).
“When you create a new listing system, you create new incentives, and people start to manage patients differently,” Dr. Desai observed. “Increasingly now, the path direct to transplant is through temporary mechanical circulatory support rather than durable mechanical circulatory support. Is that a good idea? We don’t know, but if you look at the best data, those on ECMO or percutaneous VADs have the worst outcomes. So the question of whether we should take the sickest of sick patients directly to transplant as a standard strategy has come under scrutiny.”
Improved durable LVAD technology brings impressive clinical outcomes
Results of the landmark MOMENTUM 3 randomized trial showed that 2-year clinical outcomes with the magnetically levitated centrifugal-flow HeartMate 3 LVAD now rival those of percutaneous mitral valve repair using the MitraClip device. Two-year all-cause mortality in the LVAD recipients was 22% versus 29.1% with the MitraClip in the COAPT trial and 34.9% in the MITRA-FR trial. The HeartMate 3 reduces the hemocompatibility issues that plagued earlier-generation durable LVADs, with resultant lower rates of pump thrombosis, stroke, and GI bleeding. Indeed, the outcomes in MOMENTUM 3 were so good – and so similar – with the HeartMate 3, regardless of whether the intended treatment goal was as a bridge to transplant or as lifelong destination therapy, that the investigators have recently proposed doing away with those distinctions.
“It is possible that use of arbitrary categorizations based on current or future transplant eligibility should be clinically abandoned in favor of a single preimplant strategy: to extend the survival and improve the quality of life of patients with medically refractory heart failure,” according to the investigators (JAMA Cardiol. 2020 Jan 15. doi: 10.1001/jamacardio.2019.5323).
The next step forward in LVAD technology is already on the horizon: a fully implantable device that eliminates the transcutaneous drive-line for the power supply, which is prone to infection and diminishes overall quality of life. This investigational device utilizes wireless coplanar energy transfer, with a coil ring placed around the lung and fixed to the chest wall. The implanted battery provides more than 6 hours of power without a recharge (J Heart Lung Transplant. 2019 Apr;38[4]:339-43).
“The first LVAD patient has gone swimming in Kazakhstan,” according to Dr. Desai.
Myocardial recovery in LVAD recipients remains elusive
The initial hope for LVADs was that they would not only be able to serve as a bridge to transplantation or as lifetime therapy, but that the prolonged unloading of the ventricle would enable potent medical therapy to rescue myocardial function so that the device could eventually be explanted. That does happen, but only rarely. In a large registry study, myocardial recovery occurred in only about 1% of patients on mechanical circulatory support. Attempts to enhance the process by add-on stem cell therapy have thus far been ineffective.
“For the moment, recovery is still a hope, not a reality,” the cardiologist said.
He reported serving as a consultant to more than a dozen pharmaceutical or medical device companies and receiving research grants from Alnylam, AstraZeneca, Bayer Healthcare, MyoKardia, and Novartis.
SNOWMASS, COLO. – The tragic opioid epidemic has “one small bright spot”: an expanding pool of eligible donor hearts for transplantation, Akshay S. Desai, MD, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
For decades, the annual volume of heart transplantations performed in the U.S. was static because of the huge mismatch between donor organ supply and demand. But heart transplant volume has increased steadily in the last few years – a result of the opioid epidemic.
Data from the U.S. Organ Procurement and Transplantation Network show that the proportion of donor hearts obtained from individuals who died from drug intoxication climbed from a mere 1.5% in 1999 to 17.6% in 2017, the most recent year for which data are available. Meanwhile, the size of the heart transplant waiting list, which rose year after year in 2009-2015, has since declined (N Engl J Med. 2019 Feb 7;380[6]:597-9).
“What’s amazing is that, even though these patients might have historically been considered high risk in general, the organs recovered from these patients – and particularly the hearts – don’t seem to be any worse in terms of allograft survival than the organs recovered from patients who died from other causes, which are the traditional sources, like blunt head trauma, gunshot wounds, or stroke, that lead to brain death. In general, these organs are useful and do quite well,” according to Dr. Desai, medical director of the cardiomyopathy and heart failure program at Brigham and Women’s Hospital, Boston.
He highlighted several other recent developments in the field of cardiac transplantation that promise to further expand the donor heart pool, including acceptance of hepatitis C–infected donors and organ donation after circulatory rather than brain death. Dr. Desai also drew attention to the unintended perverse consequences of a recent redesign of the U.S. donor heart allocation system and discussed the impressive improvement in clinical outcomes with mechanical circulatory support. He noted that, while relatively few cardiologists practice in the highly specialized centers where heart transplants take place, virtually all cardiologists are affected by advances in heart transplantation since hundreds of thousands of the estimated 7 million Americans with heart failure have advanced disease.
Heart transplantation, he emphasized, is becoming increasingly complex. Recipients are on average older, sicker, and have more comorbidities than in times past. As a result, there is greater need for dual organ transplants: heart/lung, heart/liver, or heart/kidney. Plus, more patients come to transplantation after prior cardiac surgery for implantation of a ventricular assist device, so sensitization to blood products is a growing issue. And, of course, the pool of transplant candidates has expanded.
“We’re now forced to take patients previously considered to have contraindications to transplant; for example, diabetes was a contraindication to transplant in the early years, but now it’s the rule in 35%-40% of our patients who present with advanced heart failure,” the cardiologist noted.
Transplants from HCV-infected donors to uninfected recipients
Hearts and lungs from donors with hepatitis C viremia were traditionally deemed unsuitable for transplant. That’s all changed in the current era of highly effective direct-acting antiviral agents for the treatment of HCV infection.
In the DONATE HCV trial, Dr. Desai’s colleagues at Brigham and Women’s Hospital showed that giving HCV-uninfected recipients of hearts or lungs from HCV-viremic donors a shortened 4-week course of treatment with sofosbuvir-velpatasvir (Epclusa) beginning within a few hours after transplantation uniformly blocked viral replication. Six months after transplantation, none of the study participants had a detectable HCV viral load, and all had excellent graft function (N Engl J Med. 2019 Apr 25;380[17]:1606-17).
“This is effective prevention of HCV infection by aggressive upfront therapy,” Dr. Desai explained. “We can now take organs from HCV-viremic patients and use them in solid organ transplantation. This has led to a skyrocketing increase in donors with HCV infection, and those donations have helped us clear the waiting list.”
Donation after circulatory death
Australian transplant physicians have pioneered the use of donor hearts obtained after circulatory death in individuals with devastating neurologic injury who didn’t quite meet the criteria for brain death, which is the traditional prerequisite. In the new scenario, withdrawal of life-supporting therapy is followed by circulatory death, then the donor heart is procured and preserved via extracorporeal perfusion until transplantation.
The Australians report excellent outcomes, with rates of overall survival and rejection episodes similar to outcomes from brain-dead donors (J Am Coll Cardiol. 2019 Apr 2;73[12]:1447-59). The first U.S. heart transplant involving donation after circulatory death took place at Duke University in Durham, North Carolina. A multicenter U.S. clinical trial of this practice is underway.
If the results are positive and the practice of donation after circulatory death becomes widely implemented, the U.S. heart donor pool could increase by 30%.
Recent overhaul of donor heart allocation system may have backfired
The U.S. donor heart allocation system was redesigned in the fall of 2018 in an effort to reduce waiting times. One of the biggest changes involved breaking down the category with the highest urgency status into three new subcategories based upon sickness. Now, the highest-urgency category is for patients in cardiogenic shock who are supported by extracorporeal membrane oxygenation (ECMO) or other temporary mechanical circulatory support devices.
But an analysis of United Network for Organ Sharing (UNOS) data suggests this change has unintended adverse consequences for clinical outcomes.
Indeed, the investigators reported that the use of ECMO support is fourfold greater in the new system, the use of durable left ventricular assist devices (LVADs) as a bridge to transplant is down, and outcomes are worse. The 180-day rate of freedom from death or retransplantation was 77.9%, down significantly from 93.4% in the former system. In a multivariate analysis, patients transplanted in the new system had an adjusted 2.1-fold increased risk of death or retransplantation (J Heart Lung Transplant. 2020 Jan;39[1]:1-4).
“When you create a new listing system, you create new incentives, and people start to manage patients differently,” Dr. Desai observed. “Increasingly now, the path direct to transplant is through temporary mechanical circulatory support rather than durable mechanical circulatory support. Is that a good idea? We don’t know, but if you look at the best data, those on ECMO or percutaneous VADs have the worst outcomes. So the question of whether we should take the sickest of sick patients directly to transplant as a standard strategy has come under scrutiny.”
Improved durable LVAD technology brings impressive clinical outcomes
Results of the landmark MOMENTUM 3 randomized trial showed that 2-year clinical outcomes with the magnetically levitated centrifugal-flow HeartMate 3 LVAD now rival those of percutaneous mitral valve repair using the MitraClip device. Two-year all-cause mortality in the LVAD recipients was 22% versus 29.1% with the MitraClip in the COAPT trial and 34.9% in the MITRA-FR trial. The HeartMate 3 reduces the hemocompatibility issues that plagued earlier-generation durable LVADs, with resultant lower rates of pump thrombosis, stroke, and GI bleeding. Indeed, the outcomes in MOMENTUM 3 were so good – and so similar – with the HeartMate 3, regardless of whether the intended treatment goal was as a bridge to transplant or as lifelong destination therapy, that the investigators have recently proposed doing away with those distinctions.
“It is possible that use of arbitrary categorizations based on current or future transplant eligibility should be clinically abandoned in favor of a single preimplant strategy: to extend the survival and improve the quality of life of patients with medically refractory heart failure,” according to the investigators (JAMA Cardiol. 2020 Jan 15. doi: 10.1001/jamacardio.2019.5323).
The next step forward in LVAD technology is already on the horizon: a fully implantable device that eliminates the transcutaneous drive-line for the power supply, which is prone to infection and diminishes overall quality of life. This investigational device utilizes wireless coplanar energy transfer, with a coil ring placed around the lung and fixed to the chest wall. The implanted battery provides more than 6 hours of power without a recharge (J Heart Lung Transplant. 2019 Apr;38[4]:339-43).
“The first LVAD patient has gone swimming in Kazakhstan,” according to Dr. Desai.
Myocardial recovery in LVAD recipients remains elusive
The initial hope for LVADs was that they would not only be able to serve as a bridge to transplantation or as lifetime therapy, but that the prolonged unloading of the ventricle would enable potent medical therapy to rescue myocardial function so that the device could eventually be explanted. That does happen, but only rarely. In a large registry study, myocardial recovery occurred in only about 1% of patients on mechanical circulatory support. Attempts to enhance the process by add-on stem cell therapy have thus far been ineffective.
“For the moment, recovery is still a hope, not a reality,” the cardiologist said.
He reported serving as a consultant to more than a dozen pharmaceutical or medical device companies and receiving research grants from Alnylam, AstraZeneca, Bayer Healthcare, MyoKardia, and Novartis.
EXPERT ANALYSIS FROM ACC SNOWMASS 2020
FFR use nearly halved 1-year mortality risk in ischemic heart disease
Use of fractional flow reserve significantly improved 1-year mortality rates in adults with stable ischemic heart disease, according to a review of 17,989 patients.
Although fractional flow reserve (FFR) has demonstrated value in guiding coronary revascularization, its impact on outcomes in patients with stable ischemic heart disease has not been well studied in a large population, wrote Rushi V. Parikh, MD, of the University of California, Los Angeles, and colleagues.
In a study published in the Journal of the American College of Cardiology, the researchers analyzed data from the Veterans Affairs Clinical Assessment, Reporting, and Tracking Program for adults who underwent coronary angiography between January 2009 and September 2017. The study included patients with angiographically intermediate disease, defined as 40%-69% diameter stenosis on visual inspection.
The rate of FFR use increased from 14.8% to 18.5% during the study period for all patients with intermediate lesions, and from 44% to 75% for those who had percutaneous coronary intervention, the researchers wrote.
Overall, based on hazard models, 1-year mortality was significantly lower in patients who underwent FFR, compared with those who had angiography only (2.8% vs. 5.9%; P less than 0.001). In addition, FFR use in revascularization was associated with a 43% reduced 1-year mortality risk, compared with angiography only.
The findings were limited by several factors, including the observational nature of the study, inability to distinguish between cardiovascular and noncardiovascular mortality, lack of data on the technical performance of the FFR, and a relatively short follow-up period, the researchers noted.
However, the results were strengthened by the large sample size, and support the use of FFR-guided revascularization in patients with angiographically intermediate stenosis, they wrote.
“Future registry-based studies accounting for all physiologic modalities are warranted to accurately quantify the landscape of coronary physiology-guided revascularization,” they added.
The study was supported in part by the Rocky Mountain Regional VA Medical Center in Aurora, Colo. Lead author Dr. Parikh had no financial conflicts to disclose.
SOURCE: Parikh RV et al. J Am Coll Cardiol. 2020 Feb 4;75:409-19.
Although the study suggests that the use of fractional flow reserve (FFR) has increased, it remains underused despite evidence and recommendations, wrote Julien Adjedj, MD, and Benoit Guillon, MD, in an accompanying editorial (J Am Coll Cardiol. 2020 Feb 4;75:420-1).
“Of course, time, cost, and need for hyperemia are often perceived as stumbling blocks. Yet, the real question is whether the cardiology community – not only interventional cardiologists – has truly adopted FFR (i.e., using it routinely and treating according to the results),” they wrote.
The editorialists noted that, in this study, typical predictors of FFR included younger age, multivessel or left main disease, previous history of percutaneous coronary intervention, no heart failure, and higher left ventricular ejection fraction.
“However, neither the absence of documented ischemia nor the presence of symptoms influenced the use of FFR. Significant site-level variation in FFR was observed,” they wrote. “This important finding suggests that the main reason for FFR underutilization in the contemporary era is operator belief regarding the utility of coronary physiology, and that revised reimbursement policies and additional education/training may not have a meaningful impact on FFR adoption.”
The editorialists emphasized that, although FFR use has increased, the findings of a significant decrease in mortality support additional use of FFR “and good reasons to do so.”
Dr. Adjedj is affiliated with the Centre Hospitalier Universitaire Vaudois in Lausanne, Switzerland, and had no financial conflicts to disclose. Dr. Guillon is affiliated with the University Hospital Jean Minjoz in Besançon, France, and disclosed a grant from Sanofi and participation in a conference for Abbott.
Although the study suggests that the use of fractional flow reserve (FFR) has increased, it remains underused despite evidence and recommendations, wrote Julien Adjedj, MD, and Benoit Guillon, MD, in an accompanying editorial (J Am Coll Cardiol. 2020 Feb 4;75:420-1).
“Of course, time, cost, and need for hyperemia are often perceived as stumbling blocks. Yet, the real question is whether the cardiology community – not only interventional cardiologists – has truly adopted FFR (i.e., using it routinely and treating according to the results),” they wrote.
The editorialists noted that, in this study, typical predictors of FFR included younger age, multivessel or left main disease, previous history of percutaneous coronary intervention, no heart failure, and higher left ventricular ejection fraction.
“However, neither the absence of documented ischemia nor the presence of symptoms influenced the use of FFR. Significant site-level variation in FFR was observed,” they wrote. “This important finding suggests that the main reason for FFR underutilization in the contemporary era is operator belief regarding the utility of coronary physiology, and that revised reimbursement policies and additional education/training may not have a meaningful impact on FFR adoption.”
The editorialists emphasized that, although FFR use has increased, the findings of a significant decrease in mortality support additional use of FFR “and good reasons to do so.”
Dr. Adjedj is affiliated with the Centre Hospitalier Universitaire Vaudois in Lausanne, Switzerland, and had no financial conflicts to disclose. Dr. Guillon is affiliated with the University Hospital Jean Minjoz in Besançon, France, and disclosed a grant from Sanofi and participation in a conference for Abbott.
Although the study suggests that the use of fractional flow reserve (FFR) has increased, it remains underused despite evidence and recommendations, wrote Julien Adjedj, MD, and Benoit Guillon, MD, in an accompanying editorial (J Am Coll Cardiol. 2020 Feb 4;75:420-1).
“Of course, time, cost, and need for hyperemia are often perceived as stumbling blocks. Yet, the real question is whether the cardiology community – not only interventional cardiologists – has truly adopted FFR (i.e., using it routinely and treating according to the results),” they wrote.
The editorialists noted that, in this study, typical predictors of FFR included younger age, multivessel or left main disease, previous history of percutaneous coronary intervention, no heart failure, and higher left ventricular ejection fraction.
“However, neither the absence of documented ischemia nor the presence of symptoms influenced the use of FFR. Significant site-level variation in FFR was observed,” they wrote. “This important finding suggests that the main reason for FFR underutilization in the contemporary era is operator belief regarding the utility of coronary physiology, and that revised reimbursement policies and additional education/training may not have a meaningful impact on FFR adoption.”
The editorialists emphasized that, although FFR use has increased, the findings of a significant decrease in mortality support additional use of FFR “and good reasons to do so.”
Dr. Adjedj is affiliated with the Centre Hospitalier Universitaire Vaudois in Lausanne, Switzerland, and had no financial conflicts to disclose. Dr. Guillon is affiliated with the University Hospital Jean Minjoz in Besançon, France, and disclosed a grant from Sanofi and participation in a conference for Abbott.
Use of fractional flow reserve significantly improved 1-year mortality rates in adults with stable ischemic heart disease, according to a review of 17,989 patients.
Although fractional flow reserve (FFR) has demonstrated value in guiding coronary revascularization, its impact on outcomes in patients with stable ischemic heart disease has not been well studied in a large population, wrote Rushi V. Parikh, MD, of the University of California, Los Angeles, and colleagues.
In a study published in the Journal of the American College of Cardiology, the researchers analyzed data from the Veterans Affairs Clinical Assessment, Reporting, and Tracking Program for adults who underwent coronary angiography between January 2009 and September 2017. The study included patients with angiographically intermediate disease, defined as 40%-69% diameter stenosis on visual inspection.
The rate of FFR use increased from 14.8% to 18.5% during the study period for all patients with intermediate lesions, and from 44% to 75% for those who had percutaneous coronary intervention, the researchers wrote.
Overall, based on hazard models, 1-year mortality was significantly lower in patients who underwent FFR, compared with those who had angiography only (2.8% vs. 5.9%; P less than 0.001). In addition, FFR use in revascularization was associated with a 43% reduced 1-year mortality risk, compared with angiography only.
The findings were limited by several factors, including the observational nature of the study, inability to distinguish between cardiovascular and noncardiovascular mortality, lack of data on the technical performance of the FFR, and a relatively short follow-up period, the researchers noted.
However, the results were strengthened by the large sample size, and support the use of FFR-guided revascularization in patients with angiographically intermediate stenosis, they wrote.
“Future registry-based studies accounting for all physiologic modalities are warranted to accurately quantify the landscape of coronary physiology-guided revascularization,” they added.
The study was supported in part by the Rocky Mountain Regional VA Medical Center in Aurora, Colo. Lead author Dr. Parikh had no financial conflicts to disclose.
SOURCE: Parikh RV et al. J Am Coll Cardiol. 2020 Feb 4;75:409-19.
Use of fractional flow reserve significantly improved 1-year mortality rates in adults with stable ischemic heart disease, according to a review of 17,989 patients.
Although fractional flow reserve (FFR) has demonstrated value in guiding coronary revascularization, its impact on outcomes in patients with stable ischemic heart disease has not been well studied in a large population, wrote Rushi V. Parikh, MD, of the University of California, Los Angeles, and colleagues.
In a study published in the Journal of the American College of Cardiology, the researchers analyzed data from the Veterans Affairs Clinical Assessment, Reporting, and Tracking Program for adults who underwent coronary angiography between January 2009 and September 2017. The study included patients with angiographically intermediate disease, defined as 40%-69% diameter stenosis on visual inspection.
The rate of FFR use increased from 14.8% to 18.5% during the study period for all patients with intermediate lesions, and from 44% to 75% for those who had percutaneous coronary intervention, the researchers wrote.
Overall, based on hazard models, 1-year mortality was significantly lower in patients who underwent FFR, compared with those who had angiography only (2.8% vs. 5.9%; P less than 0.001). In addition, FFR use in revascularization was associated with a 43% reduced 1-year mortality risk, compared with angiography only.
The findings were limited by several factors, including the observational nature of the study, inability to distinguish between cardiovascular and noncardiovascular mortality, lack of data on the technical performance of the FFR, and a relatively short follow-up period, the researchers noted.
However, the results were strengthened by the large sample size, and support the use of FFR-guided revascularization in patients with angiographically intermediate stenosis, they wrote.
“Future registry-based studies accounting for all physiologic modalities are warranted to accurately quantify the landscape of coronary physiology-guided revascularization,” they added.
The study was supported in part by the Rocky Mountain Regional VA Medical Center in Aurora, Colo. Lead author Dr. Parikh had no financial conflicts to disclose.
SOURCE: Parikh RV et al. J Am Coll Cardiol. 2020 Feb 4;75:409-19.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Key clinical point: Use of fractional flow reserve to assist in revascularization significantly reduced the 1-year mortality risk in patients with stable ischemic heart disease.
Major finding:
Study details: The data come from a review of 17,989 patients who underwent coronary angiography during 2009-2017.
Disclosures: The study was supported in part by the Rocky Mountain Regional Veterans Affairs Medical Center in Aurora, Colo. Lead author Dr. Parikh had no financial conflicts to disclose.
Source: Parikh RV et al. J Am Coll Cardiol. 2020 Feb 4;75:409-19.
Vigilance safely keeps AFib patients off anticoagulants post ablation
NATIONAL HARBOR, MD. – A pilot program of daily arrhythmia self-vigilance has allowed selected patients with no atrial fibrillation following a catheter ablation procedure to safely come off a regimen of daily oral anticoagulation despite having residual risk factors for ischemic stroke.
This program, which started several years ago at the University of Pennsylvania in Philadelphia, has now managed 190 patients and followed them for a median of just over 3 years, and during 576 patient-years of follow-up, just a single patient had an ischemic cerebrovascular event that occurred with no atrial fibrillation (AFib) recurrence and appeared to be caused by an atherosclerotic embolism, Francis E. Marchlinski, MD, said at the annual International AF Symposium.
Although this strategy has not yet been tested in a prospective, randomized trial, this anecdotal, single-center experience suggests that the approach is “safe and effective” for selected patients who are eager to come off of their anticoagulation regimen when they remain arrhythmia free following catheter ablation of their AFib, said Dr. Marchlinski, professor of medicine and director of electrophysiology at the University of Pennsylvania. He and his associates developed this strategy as a way to more safely allow these patients to stop taking a daily oral anticoagulant because he found that many patients were stopping on their own, with no safety strategy in place.
“Patients tell me they don’t want to be on an oral anticoagulant because a parent had a hemorrhagic stroke, and they say they’re willing to accept the risk” of having an ischemic stroke by coming off anticoagulation. “This is a way for them to do it safely,” Dr. Marchlinski said in an interview. He stressed that he only allows his patients to go this route if they understand the risk and accept their shared responsibility for vigilant, twice-daily pulse monitoring to detect resumption of an irregular heart beat.
Since 2011, Dr. Marchlinski’s program ablated 1,216 patients with AFib who then remained arrhythmia free during 3 weeks of continuous ECG monitoring following their procedure. Among these patients, 443 had a CHA2DS2-VAScscore of either 0 (men) or 1 (women) that indicated no ongoing need for oral anticoagulation according to current guidelines. Of the remaining 773 patients with a CHA2DS2-VASc score of at least 1 in men and 2 in women, the clinicians determined 583 to be ineligible for the program because of their unwillingness to accept the risk, unwillingness to comply with daily pulse checks, a history of asymptomatic AFib, a CHA2DS2-VASc score greater than 4, or a resting pulse above 90 beats per minute, leaving 190 patients eligible to participate. Among these patients, 105 (55%) had a CHA2DS2-VASc score of 2-4, which should prompt anticoagulation according to current guidelines.Participating patients committed to check their resting pulse by palpation at least twice daily and to contacting the program immediately if their resting rate spiked by more than 20 beats per minutes or in another way seemed irregular. Patients were also instructed to restart their oral anticoagulation immediately if they experienced AFib symptoms that persisted for more than 5 minutes. Many patients in the program also use a wearable device (usually a watch) to monitor their resting pulse and to generate a 30-second ECG recording that they can send as an electronic file to the University of Pennsylvania staff. “We embrace wearables,” Dr. Marchlinski said. Those without a wearable can undergo transtelephonic EEG monitoring to document a suspected arrhythmia recurrence, and all patients undergo annual monitoring by continuous ECG for at least 2 weeks.During follow-up, in addition to the 1 patient free from recurrent AFib who had an atherosclerotic embolism, 34 patients resumed anticoagulant treatment because of AFib recurrence; 12 withdrew from the program because of noncompliance or preference, or because an exclusion appeared; 29 resumed oral anticoagulation transiently but then discontinued the drug a second time when their AFib recurrence resolved; and 114 patients (60% of the starting cohort of 190) remained completely off anticoagulation during a median of 37 months. These data updated a published report from Dr. Marchlinski and his associates on their first 99 patients followed for a median of 30 months (J Cardiovasc Electrophysiol. 2019 May;30[5]:631-8).
This experience underscored the need for ongoing rhythm monitoring even in the absence of AFib symptoms, as six patients developed asymptomatic AFib detected by monitoring, including one patient whose recurrence occurred 30 months after the ablation procedure.
Dr. Marchlinski stressed the stringent selection process he applies to limit this approach to patients who are willing to faithfully monitor their pulse and symptoms daily, and who accept the risk that this approach may pose and their responsibility to stay in contact with the clinical team. The program calls patients at the 6-month mark between annual monitoring to remind them of their need for daily attention.
“Being off anticoagulants is very important to these patients,” he explained, and he highlighted the added workload this strategy places on his staff. “I think this has legs” for adoption by other cardiac arrhythmia programs, “but it depends on the time the staff is willing to spend” monitoring and following these patients, some of whom regularly send in ECG traces from their wearable devices for assessment. “It takes a village” to make this program work, he said.
Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biosense Webster, Biotronik, Boston Scientific, and Medtronic.
NATIONAL HARBOR, MD. – A pilot program of daily arrhythmia self-vigilance has allowed selected patients with no atrial fibrillation following a catheter ablation procedure to safely come off a regimen of daily oral anticoagulation despite having residual risk factors for ischemic stroke.
This program, which started several years ago at the University of Pennsylvania in Philadelphia, has now managed 190 patients and followed them for a median of just over 3 years, and during 576 patient-years of follow-up, just a single patient had an ischemic cerebrovascular event that occurred with no atrial fibrillation (AFib) recurrence and appeared to be caused by an atherosclerotic embolism, Francis E. Marchlinski, MD, said at the annual International AF Symposium.
Although this strategy has not yet been tested in a prospective, randomized trial, this anecdotal, single-center experience suggests that the approach is “safe and effective” for selected patients who are eager to come off of their anticoagulation regimen when they remain arrhythmia free following catheter ablation of their AFib, said Dr. Marchlinski, professor of medicine and director of electrophysiology at the University of Pennsylvania. He and his associates developed this strategy as a way to more safely allow these patients to stop taking a daily oral anticoagulant because he found that many patients were stopping on their own, with no safety strategy in place.
“Patients tell me they don’t want to be on an oral anticoagulant because a parent had a hemorrhagic stroke, and they say they’re willing to accept the risk” of having an ischemic stroke by coming off anticoagulation. “This is a way for them to do it safely,” Dr. Marchlinski said in an interview. He stressed that he only allows his patients to go this route if they understand the risk and accept their shared responsibility for vigilant, twice-daily pulse monitoring to detect resumption of an irregular heart beat.
Since 2011, Dr. Marchlinski’s program ablated 1,216 patients with AFib who then remained arrhythmia free during 3 weeks of continuous ECG monitoring following their procedure. Among these patients, 443 had a CHA2DS2-VAScscore of either 0 (men) or 1 (women) that indicated no ongoing need for oral anticoagulation according to current guidelines. Of the remaining 773 patients with a CHA2DS2-VASc score of at least 1 in men and 2 in women, the clinicians determined 583 to be ineligible for the program because of their unwillingness to accept the risk, unwillingness to comply with daily pulse checks, a history of asymptomatic AFib, a CHA2DS2-VASc score greater than 4, or a resting pulse above 90 beats per minute, leaving 190 patients eligible to participate. Among these patients, 105 (55%) had a CHA2DS2-VASc score of 2-4, which should prompt anticoagulation according to current guidelines.Participating patients committed to check their resting pulse by palpation at least twice daily and to contacting the program immediately if their resting rate spiked by more than 20 beats per minutes or in another way seemed irregular. Patients were also instructed to restart their oral anticoagulation immediately if they experienced AFib symptoms that persisted for more than 5 minutes. Many patients in the program also use a wearable device (usually a watch) to monitor their resting pulse and to generate a 30-second ECG recording that they can send as an electronic file to the University of Pennsylvania staff. “We embrace wearables,” Dr. Marchlinski said. Those without a wearable can undergo transtelephonic EEG monitoring to document a suspected arrhythmia recurrence, and all patients undergo annual monitoring by continuous ECG for at least 2 weeks.During follow-up, in addition to the 1 patient free from recurrent AFib who had an atherosclerotic embolism, 34 patients resumed anticoagulant treatment because of AFib recurrence; 12 withdrew from the program because of noncompliance or preference, or because an exclusion appeared; 29 resumed oral anticoagulation transiently but then discontinued the drug a second time when their AFib recurrence resolved; and 114 patients (60% of the starting cohort of 190) remained completely off anticoagulation during a median of 37 months. These data updated a published report from Dr. Marchlinski and his associates on their first 99 patients followed for a median of 30 months (J Cardiovasc Electrophysiol. 2019 May;30[5]:631-8).
This experience underscored the need for ongoing rhythm monitoring even in the absence of AFib symptoms, as six patients developed asymptomatic AFib detected by monitoring, including one patient whose recurrence occurred 30 months after the ablation procedure.
Dr. Marchlinski stressed the stringent selection process he applies to limit this approach to patients who are willing to faithfully monitor their pulse and symptoms daily, and who accept the risk that this approach may pose and their responsibility to stay in contact with the clinical team. The program calls patients at the 6-month mark between annual monitoring to remind them of their need for daily attention.
“Being off anticoagulants is very important to these patients,” he explained, and he highlighted the added workload this strategy places on his staff. “I think this has legs” for adoption by other cardiac arrhythmia programs, “but it depends on the time the staff is willing to spend” monitoring and following these patients, some of whom regularly send in ECG traces from their wearable devices for assessment. “It takes a village” to make this program work, he said.
Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biosense Webster, Biotronik, Boston Scientific, and Medtronic.
NATIONAL HARBOR, MD. – A pilot program of daily arrhythmia self-vigilance has allowed selected patients with no atrial fibrillation following a catheter ablation procedure to safely come off a regimen of daily oral anticoagulation despite having residual risk factors for ischemic stroke.
This program, which started several years ago at the University of Pennsylvania in Philadelphia, has now managed 190 patients and followed them for a median of just over 3 years, and during 576 patient-years of follow-up, just a single patient had an ischemic cerebrovascular event that occurred with no atrial fibrillation (AFib) recurrence and appeared to be caused by an atherosclerotic embolism, Francis E. Marchlinski, MD, said at the annual International AF Symposium.
Although this strategy has not yet been tested in a prospective, randomized trial, this anecdotal, single-center experience suggests that the approach is “safe and effective” for selected patients who are eager to come off of their anticoagulation regimen when they remain arrhythmia free following catheter ablation of their AFib, said Dr. Marchlinski, professor of medicine and director of electrophysiology at the University of Pennsylvania. He and his associates developed this strategy as a way to more safely allow these patients to stop taking a daily oral anticoagulant because he found that many patients were stopping on their own, with no safety strategy in place.
“Patients tell me they don’t want to be on an oral anticoagulant because a parent had a hemorrhagic stroke, and they say they’re willing to accept the risk” of having an ischemic stroke by coming off anticoagulation. “This is a way for them to do it safely,” Dr. Marchlinski said in an interview. He stressed that he only allows his patients to go this route if they understand the risk and accept their shared responsibility for vigilant, twice-daily pulse monitoring to detect resumption of an irregular heart beat.
Since 2011, Dr. Marchlinski’s program ablated 1,216 patients with AFib who then remained arrhythmia free during 3 weeks of continuous ECG monitoring following their procedure. Among these patients, 443 had a CHA2DS2-VAScscore of either 0 (men) or 1 (women) that indicated no ongoing need for oral anticoagulation according to current guidelines. Of the remaining 773 patients with a CHA2DS2-VASc score of at least 1 in men and 2 in women, the clinicians determined 583 to be ineligible for the program because of their unwillingness to accept the risk, unwillingness to comply with daily pulse checks, a history of asymptomatic AFib, a CHA2DS2-VASc score greater than 4, or a resting pulse above 90 beats per minute, leaving 190 patients eligible to participate. Among these patients, 105 (55%) had a CHA2DS2-VASc score of 2-4, which should prompt anticoagulation according to current guidelines.Participating patients committed to check their resting pulse by palpation at least twice daily and to contacting the program immediately if their resting rate spiked by more than 20 beats per minutes or in another way seemed irregular. Patients were also instructed to restart their oral anticoagulation immediately if they experienced AFib symptoms that persisted for more than 5 minutes. Many patients in the program also use a wearable device (usually a watch) to monitor their resting pulse and to generate a 30-second ECG recording that they can send as an electronic file to the University of Pennsylvania staff. “We embrace wearables,” Dr. Marchlinski said. Those without a wearable can undergo transtelephonic EEG monitoring to document a suspected arrhythmia recurrence, and all patients undergo annual monitoring by continuous ECG for at least 2 weeks.During follow-up, in addition to the 1 patient free from recurrent AFib who had an atherosclerotic embolism, 34 patients resumed anticoagulant treatment because of AFib recurrence; 12 withdrew from the program because of noncompliance or preference, or because an exclusion appeared; 29 resumed oral anticoagulation transiently but then discontinued the drug a second time when their AFib recurrence resolved; and 114 patients (60% of the starting cohort of 190) remained completely off anticoagulation during a median of 37 months. These data updated a published report from Dr. Marchlinski and his associates on their first 99 patients followed for a median of 30 months (J Cardiovasc Electrophysiol. 2019 May;30[5]:631-8).
This experience underscored the need for ongoing rhythm monitoring even in the absence of AFib symptoms, as six patients developed asymptomatic AFib detected by monitoring, including one patient whose recurrence occurred 30 months after the ablation procedure.
Dr. Marchlinski stressed the stringent selection process he applies to limit this approach to patients who are willing to faithfully monitor their pulse and symptoms daily, and who accept the risk that this approach may pose and their responsibility to stay in contact with the clinical team. The program calls patients at the 6-month mark between annual monitoring to remind them of their need for daily attention.
“Being off anticoagulants is very important to these patients,” he explained, and he highlighted the added workload this strategy places on his staff. “I think this has legs” for adoption by other cardiac arrhythmia programs, “but it depends on the time the staff is willing to spend” monitoring and following these patients, some of whom regularly send in ECG traces from their wearable devices for assessment. “It takes a village” to make this program work, he said.
Dr. Marchlinski has been a consultant to or has received honoraria from Abbott EP/St. Jude, Biosense Webster, Biotronik, Boston Scientific, and Medtronic.
REPORTING FROM THE AF SYMPOSIUM 2020
Catheter cryoablation effective for persistent AFib in pivotal trial
NATIONAL HARBOR, MD. – , setting the stage for the device to become the first to receive U.S. labeling for catheter ablation in this atrial fibrillation population.
The Arctic Front Advance cryoballoon, used on 165 patients with persistent atrial fibrillation (AFib) enrolled in the trial, produced a 55% rate of treatment success, including freedom from recurrent AFib during 12 months of follow-up, and produced one prespecified serious adverse event in the primary safety endpoint.
Both results easily surpassed the prespecified performance goals set by negotiation with the FDA, Hugh Calkins, MD, said at the annual International AF Symposium. The trial design included no control group and instead assessed safety and efficacy against prespecified standards set by the regulatory agency.
The cryoballoon “showed excellent performance. I don’t see how this could possibly be turned down by the FDA,” said Dr. Calkins, professor of medicine and director of the cardiac arrhythmia service at Johns Hopkins Medicine in Baltimore.
Cardiac electrophysiologists have for years routinely performed catheter ablation procedures on patients with persistent AFib even though the devices, based on ablation by radiofrequency or by chilling, have been labeled for use only in treating patients with paroxysmal AFib. Although this off-label use has not resulted in any problems with health insurance coverage, Dr. Calkins said, it has kept manufacturers from marketing their ablation devices for use in persistent AFib patients.
If the reported data result in labeling for the tested cryoballoon for persistant AFib patients, “it will have a big impact,” he predicted. “People have used cryoballoons for ablating persistent AFib for years, but this would put more fuel in the fire, both the [very positive] safety and efficacy data, and getting an FDA label, which is worth a lot,” he said in an interview.
But Dr. Calkins stopped short of anticipating that the results would convince operators experienced and focused on performing radiofrequency ablation to switch to cryo devices for treating persistent AFib patients. “People are pretty stuck in their ways,” he noted, and reports are expected soon from pivotal trials that are now testing various radiofrequency devices, as well as other types of cryo devices, in persistent AFib patients, so the range of device options labeled for this population may soon grow even more.
The STOP Persistent AF trial ran at 25 sites in the United States, Canada, and Japan during March 2017-August 2019, and included 165 adults with symptomatic, persistent AFib who had not responded to at least one antiarrhythmic drug and had a history of AFib episodes lasting at least 7 days but with no episodes persisting for 6 months or longer. The study excluded patients with prior ablation or left atrial surgery, a recent cerebrovascular event, substantially reduced left ventricular function, or substantial left atrial enlargement.
The enrolled patients were an average 65 years of age and 70% were men. Patients had been diagnosed with paroxysmal AFib an average of 5 years before study entry and with persistent AFib a little over 6 months before entry. The most recent AFib episode of enrolled patients averaged about 60 days, on average they had been unsuccessfully treated with just over one antiarrhythmic drug, and on average they had previously undergone about two cardioversions, after which their arrhythmia recurred.
The primary efficacy endpoint – a 55% rate of acute procedural success plus freedom from AFib recurrence during the 9 months following a 90-day blanking period immediately after ablation plus no added or increased antiarrhythmic drugs – significantly exceeded the prespecified performance goal of a 40% rate, Dr. Calkins reported. The study used the standard measure of recurrence as any 30-second or longer AFib episode detected during a weekly ECG telemonitoring session or during 48-hour ambulatory ECG monitoring at 6- and 12-month follow-ups or during in-office 12-lead ECG assessment at 3-, 6-, and 12-month follow-up. Twelve-month follow-up occurred for 145 of the enrolled patients.
The only prespecified primary safety event was one episode of cardiac perforation, which occurred during a repeat procedure. This rate of one safety event among 165 patients (0.6%) fell well within the prespecified safety performance goal of no more than 13%. In addition to this perforation, five additional serious adverse events (3%) occurred that were attributable to the cryoballoon treatment, including two cases of vascular pseudoaneurysm, one puncture-site hematoma, one case of pericarditis, and one episode of atrial tachycardia. Four additional serious adverse events occurred that were attributable to the ablation procedure (one acute cardiac failure, one postprocedure ileus, one respiratory failure, and one urinary tract infection), for an overall serious event rate of 5%.
During follow-up, 13% of patients had a repeat ablation procedure, following an initial ablation limited to pulmonary-vein isolation (PVI). The overall rate of 1-year efficacy, including the relatively low rate of need for redo ablation, “are an impressive endorsement of a PVI-only strategy” for initial ablation, Dr. Calkins said. “I’m a strong believer in PVI only for the first ablation for both paroxysmal and persistent AFib.”
He also noted that the 30-second threshold for scoring recurrent arrhythmia episodes following the 90-day blanking period after ablation was a very conservative measure of treatment failure, but it continues to define recurrence in this and many other current AFib ablation studies because it is the historical criteria for measuring ablation success or failure. It is especially important to maintain this criteria in a study that relied on prespecified performance criteria rather than a control arm for judging efficacy, Dr. Calkins said.
The study also included three quality-of-life measures. Patient scores on the Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire rose by an average of nearly 26 points from baseline to 12 months, a statistically significant and clinically meaningful increase. Scores on both the physical and mental domains of the Short Form-12 (SF-12) improved from baseline by an average of about five points on each subscale, also statistically significant and clinically meaningful changes. Patients also reported statistically significant and in some cases substantial reductions in the prevalence rates of each of five different AFib symptoms: dizziness, dyspnea, fatigue, palpitations, and rapid heartbeat.
The study was funded by Medtronic, the company that markets the tested cryoballoon (Arctic Front Advance). Dr. Calkins has been a consultant to and has received honoraria from Medtronic, as well as from Abbott, AtriCure, Boehringer Ingelheim, Boston Scientific, and Johnson & Johnson.
NATIONAL HARBOR, MD. – , setting the stage for the device to become the first to receive U.S. labeling for catheter ablation in this atrial fibrillation population.
The Arctic Front Advance cryoballoon, used on 165 patients with persistent atrial fibrillation (AFib) enrolled in the trial, produced a 55% rate of treatment success, including freedom from recurrent AFib during 12 months of follow-up, and produced one prespecified serious adverse event in the primary safety endpoint.
Both results easily surpassed the prespecified performance goals set by negotiation with the FDA, Hugh Calkins, MD, said at the annual International AF Symposium. The trial design included no control group and instead assessed safety and efficacy against prespecified standards set by the regulatory agency.
The cryoballoon “showed excellent performance. I don’t see how this could possibly be turned down by the FDA,” said Dr. Calkins, professor of medicine and director of the cardiac arrhythmia service at Johns Hopkins Medicine in Baltimore.
Cardiac electrophysiologists have for years routinely performed catheter ablation procedures on patients with persistent AFib even though the devices, based on ablation by radiofrequency or by chilling, have been labeled for use only in treating patients with paroxysmal AFib. Although this off-label use has not resulted in any problems with health insurance coverage, Dr. Calkins said, it has kept manufacturers from marketing their ablation devices for use in persistent AFib patients.
If the reported data result in labeling for the tested cryoballoon for persistant AFib patients, “it will have a big impact,” he predicted. “People have used cryoballoons for ablating persistent AFib for years, but this would put more fuel in the fire, both the [very positive] safety and efficacy data, and getting an FDA label, which is worth a lot,” he said in an interview.
But Dr. Calkins stopped short of anticipating that the results would convince operators experienced and focused on performing radiofrequency ablation to switch to cryo devices for treating persistent AFib patients. “People are pretty stuck in their ways,” he noted, and reports are expected soon from pivotal trials that are now testing various radiofrequency devices, as well as other types of cryo devices, in persistent AFib patients, so the range of device options labeled for this population may soon grow even more.
The STOP Persistent AF trial ran at 25 sites in the United States, Canada, and Japan during March 2017-August 2019, and included 165 adults with symptomatic, persistent AFib who had not responded to at least one antiarrhythmic drug and had a history of AFib episodes lasting at least 7 days but with no episodes persisting for 6 months or longer. The study excluded patients with prior ablation or left atrial surgery, a recent cerebrovascular event, substantially reduced left ventricular function, or substantial left atrial enlargement.
The enrolled patients were an average 65 years of age and 70% were men. Patients had been diagnosed with paroxysmal AFib an average of 5 years before study entry and with persistent AFib a little over 6 months before entry. The most recent AFib episode of enrolled patients averaged about 60 days, on average they had been unsuccessfully treated with just over one antiarrhythmic drug, and on average they had previously undergone about two cardioversions, after which their arrhythmia recurred.
The primary efficacy endpoint – a 55% rate of acute procedural success plus freedom from AFib recurrence during the 9 months following a 90-day blanking period immediately after ablation plus no added or increased antiarrhythmic drugs – significantly exceeded the prespecified performance goal of a 40% rate, Dr. Calkins reported. The study used the standard measure of recurrence as any 30-second or longer AFib episode detected during a weekly ECG telemonitoring session or during 48-hour ambulatory ECG monitoring at 6- and 12-month follow-ups or during in-office 12-lead ECG assessment at 3-, 6-, and 12-month follow-up. Twelve-month follow-up occurred for 145 of the enrolled patients.
The only prespecified primary safety event was one episode of cardiac perforation, which occurred during a repeat procedure. This rate of one safety event among 165 patients (0.6%) fell well within the prespecified safety performance goal of no more than 13%. In addition to this perforation, five additional serious adverse events (3%) occurred that were attributable to the cryoballoon treatment, including two cases of vascular pseudoaneurysm, one puncture-site hematoma, one case of pericarditis, and one episode of atrial tachycardia. Four additional serious adverse events occurred that were attributable to the ablation procedure (one acute cardiac failure, one postprocedure ileus, one respiratory failure, and one urinary tract infection), for an overall serious event rate of 5%.
During follow-up, 13% of patients had a repeat ablation procedure, following an initial ablation limited to pulmonary-vein isolation (PVI). The overall rate of 1-year efficacy, including the relatively low rate of need for redo ablation, “are an impressive endorsement of a PVI-only strategy” for initial ablation, Dr. Calkins said. “I’m a strong believer in PVI only for the first ablation for both paroxysmal and persistent AFib.”
He also noted that the 30-second threshold for scoring recurrent arrhythmia episodes following the 90-day blanking period after ablation was a very conservative measure of treatment failure, but it continues to define recurrence in this and many other current AFib ablation studies because it is the historical criteria for measuring ablation success or failure. It is especially important to maintain this criteria in a study that relied on prespecified performance criteria rather than a control arm for judging efficacy, Dr. Calkins said.
The study also included three quality-of-life measures. Patient scores on the Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire rose by an average of nearly 26 points from baseline to 12 months, a statistically significant and clinically meaningful increase. Scores on both the physical and mental domains of the Short Form-12 (SF-12) improved from baseline by an average of about five points on each subscale, also statistically significant and clinically meaningful changes. Patients also reported statistically significant and in some cases substantial reductions in the prevalence rates of each of five different AFib symptoms: dizziness, dyspnea, fatigue, palpitations, and rapid heartbeat.
The study was funded by Medtronic, the company that markets the tested cryoballoon (Arctic Front Advance). Dr. Calkins has been a consultant to and has received honoraria from Medtronic, as well as from Abbott, AtriCure, Boehringer Ingelheim, Boston Scientific, and Johnson & Johnson.
NATIONAL HARBOR, MD. – , setting the stage for the device to become the first to receive U.S. labeling for catheter ablation in this atrial fibrillation population.
The Arctic Front Advance cryoballoon, used on 165 patients with persistent atrial fibrillation (AFib) enrolled in the trial, produced a 55% rate of treatment success, including freedom from recurrent AFib during 12 months of follow-up, and produced one prespecified serious adverse event in the primary safety endpoint.
Both results easily surpassed the prespecified performance goals set by negotiation with the FDA, Hugh Calkins, MD, said at the annual International AF Symposium. The trial design included no control group and instead assessed safety and efficacy against prespecified standards set by the regulatory agency.
The cryoballoon “showed excellent performance. I don’t see how this could possibly be turned down by the FDA,” said Dr. Calkins, professor of medicine and director of the cardiac arrhythmia service at Johns Hopkins Medicine in Baltimore.
Cardiac electrophysiologists have for years routinely performed catheter ablation procedures on patients with persistent AFib even though the devices, based on ablation by radiofrequency or by chilling, have been labeled for use only in treating patients with paroxysmal AFib. Although this off-label use has not resulted in any problems with health insurance coverage, Dr. Calkins said, it has kept manufacturers from marketing their ablation devices for use in persistent AFib patients.
If the reported data result in labeling for the tested cryoballoon for persistant AFib patients, “it will have a big impact,” he predicted. “People have used cryoballoons for ablating persistent AFib for years, but this would put more fuel in the fire, both the [very positive] safety and efficacy data, and getting an FDA label, which is worth a lot,” he said in an interview.
But Dr. Calkins stopped short of anticipating that the results would convince operators experienced and focused on performing radiofrequency ablation to switch to cryo devices for treating persistent AFib patients. “People are pretty stuck in their ways,” he noted, and reports are expected soon from pivotal trials that are now testing various radiofrequency devices, as well as other types of cryo devices, in persistent AFib patients, so the range of device options labeled for this population may soon grow even more.
The STOP Persistent AF trial ran at 25 sites in the United States, Canada, and Japan during March 2017-August 2019, and included 165 adults with symptomatic, persistent AFib who had not responded to at least one antiarrhythmic drug and had a history of AFib episodes lasting at least 7 days but with no episodes persisting for 6 months or longer. The study excluded patients with prior ablation or left atrial surgery, a recent cerebrovascular event, substantially reduced left ventricular function, or substantial left atrial enlargement.
The enrolled patients were an average 65 years of age and 70% were men. Patients had been diagnosed with paroxysmal AFib an average of 5 years before study entry and with persistent AFib a little over 6 months before entry. The most recent AFib episode of enrolled patients averaged about 60 days, on average they had been unsuccessfully treated with just over one antiarrhythmic drug, and on average they had previously undergone about two cardioversions, after which their arrhythmia recurred.
The primary efficacy endpoint – a 55% rate of acute procedural success plus freedom from AFib recurrence during the 9 months following a 90-day blanking period immediately after ablation plus no added or increased antiarrhythmic drugs – significantly exceeded the prespecified performance goal of a 40% rate, Dr. Calkins reported. The study used the standard measure of recurrence as any 30-second or longer AFib episode detected during a weekly ECG telemonitoring session or during 48-hour ambulatory ECG monitoring at 6- and 12-month follow-ups or during in-office 12-lead ECG assessment at 3-, 6-, and 12-month follow-up. Twelve-month follow-up occurred for 145 of the enrolled patients.
The only prespecified primary safety event was one episode of cardiac perforation, which occurred during a repeat procedure. This rate of one safety event among 165 patients (0.6%) fell well within the prespecified safety performance goal of no more than 13%. In addition to this perforation, five additional serious adverse events (3%) occurred that were attributable to the cryoballoon treatment, including two cases of vascular pseudoaneurysm, one puncture-site hematoma, one case of pericarditis, and one episode of atrial tachycardia. Four additional serious adverse events occurred that were attributable to the ablation procedure (one acute cardiac failure, one postprocedure ileus, one respiratory failure, and one urinary tract infection), for an overall serious event rate of 5%.
During follow-up, 13% of patients had a repeat ablation procedure, following an initial ablation limited to pulmonary-vein isolation (PVI). The overall rate of 1-year efficacy, including the relatively low rate of need for redo ablation, “are an impressive endorsement of a PVI-only strategy” for initial ablation, Dr. Calkins said. “I’m a strong believer in PVI only for the first ablation for both paroxysmal and persistent AFib.”
He also noted that the 30-second threshold for scoring recurrent arrhythmia episodes following the 90-day blanking period after ablation was a very conservative measure of treatment failure, but it continues to define recurrence in this and many other current AFib ablation studies because it is the historical criteria for measuring ablation success or failure. It is especially important to maintain this criteria in a study that relied on prespecified performance criteria rather than a control arm for judging efficacy, Dr. Calkins said.
The study also included three quality-of-life measures. Patient scores on the Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire rose by an average of nearly 26 points from baseline to 12 months, a statistically significant and clinically meaningful increase. Scores on both the physical and mental domains of the Short Form-12 (SF-12) improved from baseline by an average of about five points on each subscale, also statistically significant and clinically meaningful changes. Patients also reported statistically significant and in some cases substantial reductions in the prevalence rates of each of five different AFib symptoms: dizziness, dyspnea, fatigue, palpitations, and rapid heartbeat.
The study was funded by Medtronic, the company that markets the tested cryoballoon (Arctic Front Advance). Dr. Calkins has been a consultant to and has received honoraria from Medtronic, as well as from Abbott, AtriCure, Boehringer Ingelheim, Boston Scientific, and Johnson & Johnson.
REPORTING FROM THE AF SYMPOSIUM 2020
Key clinical point: Catheter ablation using a cryoballoon was safe and effective for patients with persistent atrial fibrillation in a pivotal trial.
Major finding: The rate of freedom from treatment failure after 12 months was 55%, significantly exceeding the prespecified performance goal of 40%.
Study details: A multicenter, international study with 165 enrolled patients.
Disclosures: The study was funded by Medtronic, the company that markets the tested cryoballoon (Arctic Front Advance). Dr. Calkins has been a consultant to and has received honoraria from Medtronic, as well as from Abbott, AtriCure, Boehringer Ingelheim, Boston Scientific, and Johnson & Johnson.