User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
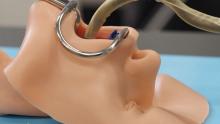
First-ever best practices for percutaneous axillary access
The Society for Cardiovascular Angiography and Interventions (SCAI) has issued the first statement on best practices for percutaneous axillary arterial access and training.
The position statement helps fill a gap amid increasing use of transaxillary access as an alternative to the femoral route for large-bore transcatheter aortic valve replacement (TAVR), endovascular aortic repair (EVAR), and mechanical circulatory support.
“The need for alternative access has increased as we are using more and more TAVR for our elderly population, and EVAR has also increased,” writing committee chair Arnold H. Seto, MD, Long Beach VA Health Care System (California) said in an interview. “There’s also a set of patients who require balloon pumps for a prolonged period, and people were using balloon pumps from the axillary approach, which were not custom-designed for that purpose.”
He noted that the evidence base leans heavily on case reports and case series, and that they were approached for guidance by a vendor developing a balloon pump specific to axillary access. “So that helped spur all of us to get together and decide to write up something on this topic, which was developing, but was certainly picking up steam rapidly.”
The statement was published in the Journal of the Society for Cardiovascular Angiography and Interventions, and it reflects the consensus of experts in heart failure, interventional cardiology and radiology, and cardiothoracic and vascular surgery. It reviews anatomic considerations and risks for percutaneous axillary access and suggests techniques for insertion, closure, and complication management.
Although the femoral artery is the most frequent access site for percutaneous large-bore procedures, the document notes that this approach may be limited in 13%-20% of patients because of prior surgeries or severe aortoiliac and/or iliofemoral atherosclerotic disease, tortuosity, or calcification.
“Absolutely, the femoral should be the predominant access site,” Dr. Seto said. Whenever there is a compromised femoral artery, “the axillary artery, which is rarely involved with atherosclerosis, makes for the most optimal alternative access. Other forms of alternative access, including transcaval and transcarotid, are possible but have their own issues and difficulties.”
Axillary access has traditionally been done through an open surgical approach, which allows for direct puncture, primary arterial repair, or placement of a sidearm conduit. Percutaneous transaxillary access avoids a surgical incision and general anesthesia and, theoretically, reduces the risk of infection, he said. It also allows for better mobility for patients, for example, who may have a balloon pump in place for weeks or even a month when waiting for a bridge to transplant.
In terms of technique, key recommendations include:
- Gaining access preferably through the left axillary
- Inserting the needle directly through the pectoralis minor into the second segment of the axillary artery
- Using a shallow-needle angle of 25-30 degrees to improve access success and decrease sheath malformation, kinking, bleeding, or vessel perforation
- Using micropuncture needles to minimize trauma to adjacent tissues
- Abducting the patient’s arm to 45-90 degrees to reduce tortuosity
- Using angiographic and ultrasound techniques to optimize vascular access
The latter point was the one area of debate among the writing committee, Dr. Seto observed. “That is one of the controversies: Should we make ultrasound mandatory? ... Everybody agreed that it can be quite useful and was likely to be useful because of its success in every other access area,” he said. “But in the absence of randomized evidence, we couldn’t make it mandatory or a strong recommendation. We just had to make it one of several options for the operator.”
The document highlights the need for familiarity with potential axillary artery complications and their management, noting that the axillary is more fragile than the femoral artery and, thus, potentially more prone to complications during instrumentation.
Data from the ARMS study in 102 patients undergoing transaxillary access for mechanical hemodynamic support reported 17 procedural complications, including 10 minor access site bleeding events, one stroke, and one pseudoaneurysm. A small study of 25 complex EVAR procedures reported a perioperative access complication rate of 8%, including one axillary artery dissection and one stenosis.
“Despite the brachial plexus being around there, there’s actually rare reports of neurologic injury and certainly none that have been permanent,” Dr. Seto said. “Also, stroke risk is probably more related to your device size and type of device rather than the approach itself.”
A significant amount of the paper is also devoted to training and privileging suggestions with an emphasis on a multidisciplinary team. The writing group recommends graduate medical education programs develop training curricula in percutaneous axillary artery access.
Those already in practice should participate in a formal training program that focuses on axillary artery anatomy, training in large bore access and closure devices, and didactic training in imaging modalities as applied to the axillary artery. Training can occur hands-on or using online simulations.
They also recommend outlining the potential need or role for proctoring and call for ongoing formal professional monitoring programs to evaluate operator outcomes using local or registry data.
“From a privileging standpoint, it was important for hospitals to be equally fair, regardless of the specialty that a requesting practitioner came from,” Dr. Seto said. “In other words, treat the vascular surgeons and interventional cardiologists and radiologists equally in terms of who has the privilege to do transaxillary access.”
The SCAI position statement has been endorsed by the American College of Cardiology, the Heart Failure Society of America, the Society of Interventional Radiology, and the Vascular & Endovascular Surgery Society.
Dr. Seto reported receiving honoraria from Getinge prior to initiation of the document. Disclosures for the rest of the writing group are available with the original article.
A version of this article first appeared on Medscape.com.
The Society for Cardiovascular Angiography and Interventions (SCAI) has issued the first statement on best practices for percutaneous axillary arterial access and training.
The position statement helps fill a gap amid increasing use of transaxillary access as an alternative to the femoral route for large-bore transcatheter aortic valve replacement (TAVR), endovascular aortic repair (EVAR), and mechanical circulatory support.
“The need for alternative access has increased as we are using more and more TAVR for our elderly population, and EVAR has also increased,” writing committee chair Arnold H. Seto, MD, Long Beach VA Health Care System (California) said in an interview. “There’s also a set of patients who require balloon pumps for a prolonged period, and people were using balloon pumps from the axillary approach, which were not custom-designed for that purpose.”
He noted that the evidence base leans heavily on case reports and case series, and that they were approached for guidance by a vendor developing a balloon pump specific to axillary access. “So that helped spur all of us to get together and decide to write up something on this topic, which was developing, but was certainly picking up steam rapidly.”
The statement was published in the Journal of the Society for Cardiovascular Angiography and Interventions, and it reflects the consensus of experts in heart failure, interventional cardiology and radiology, and cardiothoracic and vascular surgery. It reviews anatomic considerations and risks for percutaneous axillary access and suggests techniques for insertion, closure, and complication management.
Although the femoral artery is the most frequent access site for percutaneous large-bore procedures, the document notes that this approach may be limited in 13%-20% of patients because of prior surgeries or severe aortoiliac and/or iliofemoral atherosclerotic disease, tortuosity, or calcification.
“Absolutely, the femoral should be the predominant access site,” Dr. Seto said. Whenever there is a compromised femoral artery, “the axillary artery, which is rarely involved with atherosclerosis, makes for the most optimal alternative access. Other forms of alternative access, including transcaval and transcarotid, are possible but have their own issues and difficulties.”
Axillary access has traditionally been done through an open surgical approach, which allows for direct puncture, primary arterial repair, or placement of a sidearm conduit. Percutaneous transaxillary access avoids a surgical incision and general anesthesia and, theoretically, reduces the risk of infection, he said. It also allows for better mobility for patients, for example, who may have a balloon pump in place for weeks or even a month when waiting for a bridge to transplant.
In terms of technique, key recommendations include:
- Gaining access preferably through the left axillary
- Inserting the needle directly through the pectoralis minor into the second segment of the axillary artery
- Using a shallow-needle angle of 25-30 degrees to improve access success and decrease sheath malformation, kinking, bleeding, or vessel perforation
- Using micropuncture needles to minimize trauma to adjacent tissues
- Abducting the patient’s arm to 45-90 degrees to reduce tortuosity
- Using angiographic and ultrasound techniques to optimize vascular access
The latter point was the one area of debate among the writing committee, Dr. Seto observed. “That is one of the controversies: Should we make ultrasound mandatory? ... Everybody agreed that it can be quite useful and was likely to be useful because of its success in every other access area,” he said. “But in the absence of randomized evidence, we couldn’t make it mandatory or a strong recommendation. We just had to make it one of several options for the operator.”
The document highlights the need for familiarity with potential axillary artery complications and their management, noting that the axillary is more fragile than the femoral artery and, thus, potentially more prone to complications during instrumentation.
Data from the ARMS study in 102 patients undergoing transaxillary access for mechanical hemodynamic support reported 17 procedural complications, including 10 minor access site bleeding events, one stroke, and one pseudoaneurysm. A small study of 25 complex EVAR procedures reported a perioperative access complication rate of 8%, including one axillary artery dissection and one stenosis.
“Despite the brachial plexus being around there, there’s actually rare reports of neurologic injury and certainly none that have been permanent,” Dr. Seto said. “Also, stroke risk is probably more related to your device size and type of device rather than the approach itself.”
A significant amount of the paper is also devoted to training and privileging suggestions with an emphasis on a multidisciplinary team. The writing group recommends graduate medical education programs develop training curricula in percutaneous axillary artery access.
Those already in practice should participate in a formal training program that focuses on axillary artery anatomy, training in large bore access and closure devices, and didactic training in imaging modalities as applied to the axillary artery. Training can occur hands-on or using online simulations.
They also recommend outlining the potential need or role for proctoring and call for ongoing formal professional monitoring programs to evaluate operator outcomes using local or registry data.
“From a privileging standpoint, it was important for hospitals to be equally fair, regardless of the specialty that a requesting practitioner came from,” Dr. Seto said. “In other words, treat the vascular surgeons and interventional cardiologists and radiologists equally in terms of who has the privilege to do transaxillary access.”
The SCAI position statement has been endorsed by the American College of Cardiology, the Heart Failure Society of America, the Society of Interventional Radiology, and the Vascular & Endovascular Surgery Society.
Dr. Seto reported receiving honoraria from Getinge prior to initiation of the document. Disclosures for the rest of the writing group are available with the original article.
A version of this article first appeared on Medscape.com.
The Society for Cardiovascular Angiography and Interventions (SCAI) has issued the first statement on best practices for percutaneous axillary arterial access and training.
The position statement helps fill a gap amid increasing use of transaxillary access as an alternative to the femoral route for large-bore transcatheter aortic valve replacement (TAVR), endovascular aortic repair (EVAR), and mechanical circulatory support.
“The need for alternative access has increased as we are using more and more TAVR for our elderly population, and EVAR has also increased,” writing committee chair Arnold H. Seto, MD, Long Beach VA Health Care System (California) said in an interview. “There’s also a set of patients who require balloon pumps for a prolonged period, and people were using balloon pumps from the axillary approach, which were not custom-designed for that purpose.”
He noted that the evidence base leans heavily on case reports and case series, and that they were approached for guidance by a vendor developing a balloon pump specific to axillary access. “So that helped spur all of us to get together and decide to write up something on this topic, which was developing, but was certainly picking up steam rapidly.”
The statement was published in the Journal of the Society for Cardiovascular Angiography and Interventions, and it reflects the consensus of experts in heart failure, interventional cardiology and radiology, and cardiothoracic and vascular surgery. It reviews anatomic considerations and risks for percutaneous axillary access and suggests techniques for insertion, closure, and complication management.
Although the femoral artery is the most frequent access site for percutaneous large-bore procedures, the document notes that this approach may be limited in 13%-20% of patients because of prior surgeries or severe aortoiliac and/or iliofemoral atherosclerotic disease, tortuosity, or calcification.
“Absolutely, the femoral should be the predominant access site,” Dr. Seto said. Whenever there is a compromised femoral artery, “the axillary artery, which is rarely involved with atherosclerosis, makes for the most optimal alternative access. Other forms of alternative access, including transcaval and transcarotid, are possible but have their own issues and difficulties.”
Axillary access has traditionally been done through an open surgical approach, which allows for direct puncture, primary arterial repair, or placement of a sidearm conduit. Percutaneous transaxillary access avoids a surgical incision and general anesthesia and, theoretically, reduces the risk of infection, he said. It also allows for better mobility for patients, for example, who may have a balloon pump in place for weeks or even a month when waiting for a bridge to transplant.
In terms of technique, key recommendations include:
- Gaining access preferably through the left axillary
- Inserting the needle directly through the pectoralis minor into the second segment of the axillary artery
- Using a shallow-needle angle of 25-30 degrees to improve access success and decrease sheath malformation, kinking, bleeding, or vessel perforation
- Using micropuncture needles to minimize trauma to adjacent tissues
- Abducting the patient’s arm to 45-90 degrees to reduce tortuosity
- Using angiographic and ultrasound techniques to optimize vascular access
The latter point was the one area of debate among the writing committee, Dr. Seto observed. “That is one of the controversies: Should we make ultrasound mandatory? ... Everybody agreed that it can be quite useful and was likely to be useful because of its success in every other access area,” he said. “But in the absence of randomized evidence, we couldn’t make it mandatory or a strong recommendation. We just had to make it one of several options for the operator.”
The document highlights the need for familiarity with potential axillary artery complications and their management, noting that the axillary is more fragile than the femoral artery and, thus, potentially more prone to complications during instrumentation.
Data from the ARMS study in 102 patients undergoing transaxillary access for mechanical hemodynamic support reported 17 procedural complications, including 10 minor access site bleeding events, one stroke, and one pseudoaneurysm. A small study of 25 complex EVAR procedures reported a perioperative access complication rate of 8%, including one axillary artery dissection and one stenosis.
“Despite the brachial plexus being around there, there’s actually rare reports of neurologic injury and certainly none that have been permanent,” Dr. Seto said. “Also, stroke risk is probably more related to your device size and type of device rather than the approach itself.”
A significant amount of the paper is also devoted to training and privileging suggestions with an emphasis on a multidisciplinary team. The writing group recommends graduate medical education programs develop training curricula in percutaneous axillary artery access.
Those already in practice should participate in a formal training program that focuses on axillary artery anatomy, training in large bore access and closure devices, and didactic training in imaging modalities as applied to the axillary artery. Training can occur hands-on or using online simulations.
They also recommend outlining the potential need or role for proctoring and call for ongoing formal professional monitoring programs to evaluate operator outcomes using local or registry data.
“From a privileging standpoint, it was important for hospitals to be equally fair, regardless of the specialty that a requesting practitioner came from,” Dr. Seto said. “In other words, treat the vascular surgeons and interventional cardiologists and radiologists equally in terms of who has the privilege to do transaxillary access.”
The SCAI position statement has been endorsed by the American College of Cardiology, the Heart Failure Society of America, the Society of Interventional Radiology, and the Vascular & Endovascular Surgery Society.
Dr. Seto reported receiving honoraria from Getinge prior to initiation of the document. Disclosures for the rest of the writing group are available with the original article.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE SOCIETY FOR CARDIOVASCULAR ANGIOGRAPHY AND INTERVENTIONS
FDA clears mavacamten (Camzyos) for obstructive hypertrophic cardiomyopathy
The U.S. Food and Drug Administration has approved mavacamten (Camzyos, Bristol Myers Squibb) to improve functional capacity and symptoms in adults with symptomatic New York Heart Association (NYHA) class II-III obstructive hypertrophic cardiomyopathy (oHCM).
Mavacamten is the first FDA-approved allosteric and reversible inhibitor selective for cardiac myosin that targets the underlying pathophysiology of the genetic disorder. It’s available in 2.5-mg, 5-mg, 10-mg, and 15-mg capsules.
“The approval of Camzyos represents a significant milestone for appropriate symptomatic obstructive HCM patients and their families, who have long awaited a new treatment option for this chronic and progressive disease,” Anjali T. Owens, MD, medical director of the Center for Inherited Cardiac Disease and assistant professor of medicine, University of Pennsylvania, Philadelphia, said in a news release.
‘Revolutionary’ change
The approval of mavacamten was based on data from the pivotal EXPLORER-HCM and EXPLORER-LTE (long-term extension) trial of adults with symptomatic NYHA class II-III oHCM.
In EXPLORER-HCM, treatment with mavacamten over 30 weeks led to significant improvement in exercise capacity, left ventricular outflow tract (LVOT) obstruction, NYHA functional class, and health status, as reported by this news organization.
The safety and efficacy findings seen at the end of the blinded, randomized, initial 30-week phase of EXPLORER-LTE were maintained in patients who continued treatment for a median of about 62 weeks.
Mavacamten represents “an almost revolutionary change” for the treatment of oHCM, Maya E. Guglin, MD, professor of clinical medicine and an advanced heart failure physician at Indiana University, Indianapolis, said during a press briefing earlier this month at the American College of Cardiology 2022 Scientific Session earlier this month.
“Until now, there was no good medical treatment for symptomatic oHCM. This will change the landscape, and without question it will change guidelines for treating oHCM,” Dr. Guglin said.
The product information for mavacamten includes a boxed warning citing a risk for heart failure.
Echocardiogram assessments of left ventricular ejection fraction (LVEF) are required before and during treatment.
Starting mavacamten in patients with LVEF below 55% is not recommended and the drug should be interrupted if LVEF falls below 50% at any visit or if the patient experiences heart failure symptoms or worsening clinical status.
Concomitant use of mavacamten with certain cytochrome P450 inhibitors or discontinuation of certain cytochrome P450 inducers can increase the risk for heart failure attributable to systolic dysfunction. Therefore, its use is contraindicated in patients using moderate to strong CYP2C19 inhibitors or strong CYP3A4 inhibitors, and moderate to strong CYP2C19 inducers or moderate to strong CYP3A4 inducers.
Because of the risk for heart failure attributable to systolic dysfunction, mavacamten is only available through the Camzyos Risk Evaluation and Mitigation Strategy (REMS) Program.
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved mavacamten (Camzyos, Bristol Myers Squibb) to improve functional capacity and symptoms in adults with symptomatic New York Heart Association (NYHA) class II-III obstructive hypertrophic cardiomyopathy (oHCM).
Mavacamten is the first FDA-approved allosteric and reversible inhibitor selective for cardiac myosin that targets the underlying pathophysiology of the genetic disorder. It’s available in 2.5-mg, 5-mg, 10-mg, and 15-mg capsules.
“The approval of Camzyos represents a significant milestone for appropriate symptomatic obstructive HCM patients and their families, who have long awaited a new treatment option for this chronic and progressive disease,” Anjali T. Owens, MD, medical director of the Center for Inherited Cardiac Disease and assistant professor of medicine, University of Pennsylvania, Philadelphia, said in a news release.
‘Revolutionary’ change
The approval of mavacamten was based on data from the pivotal EXPLORER-HCM and EXPLORER-LTE (long-term extension) trial of adults with symptomatic NYHA class II-III oHCM.
In EXPLORER-HCM, treatment with mavacamten over 30 weeks led to significant improvement in exercise capacity, left ventricular outflow tract (LVOT) obstruction, NYHA functional class, and health status, as reported by this news organization.
The safety and efficacy findings seen at the end of the blinded, randomized, initial 30-week phase of EXPLORER-LTE were maintained in patients who continued treatment for a median of about 62 weeks.
Mavacamten represents “an almost revolutionary change” for the treatment of oHCM, Maya E. Guglin, MD, professor of clinical medicine and an advanced heart failure physician at Indiana University, Indianapolis, said during a press briefing earlier this month at the American College of Cardiology 2022 Scientific Session earlier this month.
“Until now, there was no good medical treatment for symptomatic oHCM. This will change the landscape, and without question it will change guidelines for treating oHCM,” Dr. Guglin said.
The product information for mavacamten includes a boxed warning citing a risk for heart failure.
Echocardiogram assessments of left ventricular ejection fraction (LVEF) are required before and during treatment.
Starting mavacamten in patients with LVEF below 55% is not recommended and the drug should be interrupted if LVEF falls below 50% at any visit or if the patient experiences heart failure symptoms or worsening clinical status.
Concomitant use of mavacamten with certain cytochrome P450 inhibitors or discontinuation of certain cytochrome P450 inducers can increase the risk for heart failure attributable to systolic dysfunction. Therefore, its use is contraindicated in patients using moderate to strong CYP2C19 inhibitors or strong CYP3A4 inhibitors, and moderate to strong CYP2C19 inducers or moderate to strong CYP3A4 inducers.
Because of the risk for heart failure attributable to systolic dysfunction, mavacamten is only available through the Camzyos Risk Evaluation and Mitigation Strategy (REMS) Program.
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved mavacamten (Camzyos, Bristol Myers Squibb) to improve functional capacity and symptoms in adults with symptomatic New York Heart Association (NYHA) class II-III obstructive hypertrophic cardiomyopathy (oHCM).
Mavacamten is the first FDA-approved allosteric and reversible inhibitor selective for cardiac myosin that targets the underlying pathophysiology of the genetic disorder. It’s available in 2.5-mg, 5-mg, 10-mg, and 15-mg capsules.
“The approval of Camzyos represents a significant milestone for appropriate symptomatic obstructive HCM patients and their families, who have long awaited a new treatment option for this chronic and progressive disease,” Anjali T. Owens, MD, medical director of the Center for Inherited Cardiac Disease and assistant professor of medicine, University of Pennsylvania, Philadelphia, said in a news release.
‘Revolutionary’ change
The approval of mavacamten was based on data from the pivotal EXPLORER-HCM and EXPLORER-LTE (long-term extension) trial of adults with symptomatic NYHA class II-III oHCM.
In EXPLORER-HCM, treatment with mavacamten over 30 weeks led to significant improvement in exercise capacity, left ventricular outflow tract (LVOT) obstruction, NYHA functional class, and health status, as reported by this news organization.
The safety and efficacy findings seen at the end of the blinded, randomized, initial 30-week phase of EXPLORER-LTE were maintained in patients who continued treatment for a median of about 62 weeks.
Mavacamten represents “an almost revolutionary change” for the treatment of oHCM, Maya E. Guglin, MD, professor of clinical medicine and an advanced heart failure physician at Indiana University, Indianapolis, said during a press briefing earlier this month at the American College of Cardiology 2022 Scientific Session earlier this month.
“Until now, there was no good medical treatment for symptomatic oHCM. This will change the landscape, and without question it will change guidelines for treating oHCM,” Dr. Guglin said.
The product information for mavacamten includes a boxed warning citing a risk for heart failure.
Echocardiogram assessments of left ventricular ejection fraction (LVEF) are required before and during treatment.
Starting mavacamten in patients with LVEF below 55% is not recommended and the drug should be interrupted if LVEF falls below 50% at any visit or if the patient experiences heart failure symptoms or worsening clinical status.
Concomitant use of mavacamten with certain cytochrome P450 inhibitors or discontinuation of certain cytochrome P450 inducers can increase the risk for heart failure attributable to systolic dysfunction. Therefore, its use is contraindicated in patients using moderate to strong CYP2C19 inhibitors or strong CYP3A4 inhibitors, and moderate to strong CYP2C19 inducers or moderate to strong CYP3A4 inducers.
Because of the risk for heart failure attributable to systolic dysfunction, mavacamten is only available through the Camzyos Risk Evaluation and Mitigation Strategy (REMS) Program.
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.
FDA warns of pump defect with Medtronic HVAD system
Patients implanted with the Medtronic HeartWare ventricular assist device (HVAD) System who develop pump thrombosis could have a welding defect in the internal pump causing the pump to malfunction, the Food and Drug Administration said in a letter to health care professionals.
Medtronic has sent providers an urgent medical device notice about the pump weld defect and is trying to identify which HVAD pumps are affected.
The Medtronic HVAD System was approved as a bridge to heart transplantation in 2012. Since then, it has been fraught with problems.
This past June, the company announced it was stopping all sales of the device and advised physicians to stop implanting it, as reported by this news organization.
Pump thrombosis
Medtronic has received complaints of suspected pump thrombosis in three patients with the HVAD System.
All three patients presented with one or more of the following signs or symptoms: grinding sound, transient power spikes on log files and high watt alarms, elevated lactate dehydrogenase, and low motor speed resulting in low perfusion or dizziness or lightheadedness.
Inspection of the returned pumps in these three cases identified a malfunction of the internal pump. The pumps were exchanged in all three patients. Two patients died after the pump exchange.
The FDA does not recommend the elective removal of properly functioning systems.
“Decisions about removing or exchanging the Medtronic HVAD System should be made by health care providers and patients on a case-by-case basis, considering the patient’s clinical status and surgical risks,” the agency advised.
Patients who present with one or more of the signs or symptoms of pump thrombosis should be first treated for pump thrombosis.
If symptoms fail to resolve, providers may consider whether the patient is a candidate for pump exchange, heart transplant, or pump explant for recovery, taking into account the patient’s clinical condition and surgical risks.
For patients with any of the signs and symptoms of pump thrombosis, logfiles from the controller should be uploaded to Medtronic.
The FDA is working with Medtronic to monitor for any adverse events related to pump weld defects and ensure patients with the HVAD implant continue to receive appropriate follow-up monitoring.
Problems related to the Medtronic HVAD System should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Patients implanted with the Medtronic HeartWare ventricular assist device (HVAD) System who develop pump thrombosis could have a welding defect in the internal pump causing the pump to malfunction, the Food and Drug Administration said in a letter to health care professionals.
Medtronic has sent providers an urgent medical device notice about the pump weld defect and is trying to identify which HVAD pumps are affected.
The Medtronic HVAD System was approved as a bridge to heart transplantation in 2012. Since then, it has been fraught with problems.
This past June, the company announced it was stopping all sales of the device and advised physicians to stop implanting it, as reported by this news organization.
Pump thrombosis
Medtronic has received complaints of suspected pump thrombosis in three patients with the HVAD System.
All three patients presented with one or more of the following signs or symptoms: grinding sound, transient power spikes on log files and high watt alarms, elevated lactate dehydrogenase, and low motor speed resulting in low perfusion or dizziness or lightheadedness.
Inspection of the returned pumps in these three cases identified a malfunction of the internal pump. The pumps were exchanged in all three patients. Two patients died after the pump exchange.
The FDA does not recommend the elective removal of properly functioning systems.
“Decisions about removing or exchanging the Medtronic HVAD System should be made by health care providers and patients on a case-by-case basis, considering the patient’s clinical status and surgical risks,” the agency advised.
Patients who present with one or more of the signs or symptoms of pump thrombosis should be first treated for pump thrombosis.
If symptoms fail to resolve, providers may consider whether the patient is a candidate for pump exchange, heart transplant, or pump explant for recovery, taking into account the patient’s clinical condition and surgical risks.
For patients with any of the signs and symptoms of pump thrombosis, logfiles from the controller should be uploaded to Medtronic.
The FDA is working with Medtronic to monitor for any adverse events related to pump weld defects and ensure patients with the HVAD implant continue to receive appropriate follow-up monitoring.
Problems related to the Medtronic HVAD System should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Patients implanted with the Medtronic HeartWare ventricular assist device (HVAD) System who develop pump thrombosis could have a welding defect in the internal pump causing the pump to malfunction, the Food and Drug Administration said in a letter to health care professionals.
Medtronic has sent providers an urgent medical device notice about the pump weld defect and is trying to identify which HVAD pumps are affected.
The Medtronic HVAD System was approved as a bridge to heart transplantation in 2012. Since then, it has been fraught with problems.
This past June, the company announced it was stopping all sales of the device and advised physicians to stop implanting it, as reported by this news organization.
Pump thrombosis
Medtronic has received complaints of suspected pump thrombosis in three patients with the HVAD System.
All three patients presented with one or more of the following signs or symptoms: grinding sound, transient power spikes on log files and high watt alarms, elevated lactate dehydrogenase, and low motor speed resulting in low perfusion or dizziness or lightheadedness.
Inspection of the returned pumps in these three cases identified a malfunction of the internal pump. The pumps were exchanged in all three patients. Two patients died after the pump exchange.
The FDA does not recommend the elective removal of properly functioning systems.
“Decisions about removing or exchanging the Medtronic HVAD System should be made by health care providers and patients on a case-by-case basis, considering the patient’s clinical status and surgical risks,” the agency advised.
Patients who present with one or more of the signs or symptoms of pump thrombosis should be first treated for pump thrombosis.
If symptoms fail to resolve, providers may consider whether the patient is a candidate for pump exchange, heart transplant, or pump explant for recovery, taking into account the patient’s clinical condition and surgical risks.
For patients with any of the signs and symptoms of pump thrombosis, logfiles from the controller should be uploaded to Medtronic.
The FDA is working with Medtronic to monitor for any adverse events related to pump weld defects and ensure patients with the HVAD implant continue to receive appropriate follow-up monitoring.
Problems related to the Medtronic HVAD System should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Tirzepatide succeeds in obesity in SURMOUNT-1, says Lilly
More than half of patients taking the two highest doses of tirzepatide as a once-weekly injection lost at least 20% of their body weight in the first phase 3 trial to examine this agent in patients with obesity, but without diabetes, according to preliminary top-line results from the SURMOUNT-1 trial announced by Lilly.
The full results will be reported at an upcoming medical conference and published at a later date, Lilly added.
There was much excitement in response to the news, but others have urged caution and noted that, even if tirzepatide is eventually approved for obesity, one of the major barriers to use in the United States will be insurance coverage.
“Wow (and a double Wow!) 52lb weight loss (22.5%) at highest dose of tirzepatide,” tweeted Sek Kathiresan, MD, a cardiologist who is cofounder of Verve Therapeutics and on leave from Harvard (@skathire).
“Thus far the challenge with GLP-1s [agonists] for management of obesity is that insurance usually isn’t covering them. This makes them unaffordable for most people,” replied James Marroquin, MD, of the University of Texas at Austin. (@Jamesmarroquin).
Yoni Freedhoff, MD, of the University of Ottawa (Ont.) who writes a column for this news organization on obesity, said if tirzepatide pans out, along with other similar agents already on the market for this indication, “the next few decades should see the pharmaceutical management of obesity rival its surgical management.”
Would compete with ‘game-changer’ semaglutide?
Tirzepatide has been dubbed a “twincretin” because it works not only as an agonist of the glucagonlike peptide-1 (GLP-1) receptor, but also of the glucose-dependent insulinotropic polypeptide (GIP) receptor. It has been much hyped based on the results of the series of SURPASS clinical trials, which have formed the basis of the application for type 2 diabetes approval, about which the U.S. Food and Drug Administration is expected to make a decision soon.
Several GLP-1 agonists are on the market for both type 2 diabetes and for obesity indications separately, including semaglutide (marketed as Wegovy for obesity, also a once-weekly injection) and liraglutide (Saxenda for obesity, a daily injection), both Novo Nordisk agents.
Wegovy was approved for weight loss in the United States last year, with doctors telling this news organization then that a third of patients who take the drug are likely to lose 20% or more of their starting weight, an outcome that approaches reductions seen with bariatric surgery.
Dr. Freedhoff said he’d like to see “reimbursement by insurers who will see these drugs serving as important ancillary treatments for the myriad of weight-responsive conditions they’re already covering.”
SURMOUNT-1 data: ‘Impressive body weight’ reductions
The new tirzepatide data come from the multicenter, randomized, double-blind, placebo-controlled SURMOUNT-1 trial, which included 2539 participants from the United States, Argentina, Brazil, China, India, Japan Mexico, Russia, and Taiwan. They had obesity or overweight plus at least one comorbidity but not diabetes. They were randomized to 5-mg, 10-mg, or 15-mg once-weekly tirzepatide or placebo injections for 18 months (72 weeks).
Efficacy was analyzed in two ways. Prior to factoring in drug discontinuation, participants taking tirzepatide experienced weight loss of 16.0% (35 lb/16 kg) with 5 mg, 21.4% (49 lb/22 kg) with 10 mg, and 22.5% (52 lb/24 kg) on 15 mg. In contrast, the placebo group lost just 2.4% of body weight (5 lb/2 kg).
But treatment discontinuation rates because of adverse events were 4.3%, 7.1%, 6.2%, and 2.6%, for tirzepatide 5 mg, 10 mg, 15 mg, and placebo, respectively. Overall treatment discontinuation rates were 14.3%, 16.4%, 15.1%, and 26.4%, respectively.
When efficacy was assessed regardless of treatment discontinuation, average body weight reductions were 15.0%, 19.5%, 20.9%, and 3.1% for tirzepatide 5 mg, 10 mg, 15 mg, and placebo, respectively.
More than half of patients taking tirzepatide 10 mg and 15 mg (55% and 63%, respectively) lost at least 20% of their body weight, compared with just 1.3% taking placebo.
Overall safety and tolerability were similar to those of other GLP-1 agonists, with adverse events being gastrointestinal in nature and increasing with higher doses. Nausea affected 24.6%, 33.3%, and 31.0% of the tirzepatide 5-mg, 10-mg, and 15-mg dose groups, respectively, and vomiting was experienced by 8.3%, 10.7%, and 12.2% of patients, respectively. Diarrhea and constipation were also reported more often with the drug than placebo.
“Tirzepatide delivered impressive body weight reductions in SURMOUNT-1, which could represent an important step forward for helping the patient and physician partnership treat this complex disease,” said study investigator Louis J. Aronne, MD, director of the Comprehensive Weight Control Center and the Sanford I. Weill Professor of Metabolic Research at Weill Cornell Medicine, New York, in a press release.
Further studies are ongoing for tirzepatide as a potential treatment for obesity or overweight, according to the Lilly statement. SURMOUNT is a phase 3 global clinical development program for tirzepatide that began in late 2019 with over 5,000 people with obesity or overweight across six clinical trials. Results from SURMOUNT-2, SURMOUNT-3, and SURMOUNT-4 are expected in 2023.
Tirzepatide is also being studied as a potential treatment for nonalcoholic fatty liver disease and heart failure with preserved ejection fraction. Studies of tirzepatide in obstructive sleep apnea and of morbidity/mortality in obesity are also planned.
Dr. Aronne is cofounder, chief scientific advisor, and a member of the board of directors for Intellihealth. He is also a paid scientific advisory board member for Eli Lilly. Dr. Freedhoff has served or is serving as a director, officer, partner, employee, adviser, consultant, or trustee for the Bariatric Medical Institute and Constant Health and has received a research grant from Novo Nordisk.
A version of this article first appeared on Medscape.com.
More than half of patients taking the two highest doses of tirzepatide as a once-weekly injection lost at least 20% of their body weight in the first phase 3 trial to examine this agent in patients with obesity, but without diabetes, according to preliminary top-line results from the SURMOUNT-1 trial announced by Lilly.
The full results will be reported at an upcoming medical conference and published at a later date, Lilly added.
There was much excitement in response to the news, but others have urged caution and noted that, even if tirzepatide is eventually approved for obesity, one of the major barriers to use in the United States will be insurance coverage.
“Wow (and a double Wow!) 52lb weight loss (22.5%) at highest dose of tirzepatide,” tweeted Sek Kathiresan, MD, a cardiologist who is cofounder of Verve Therapeutics and on leave from Harvard (@skathire).
“Thus far the challenge with GLP-1s [agonists] for management of obesity is that insurance usually isn’t covering them. This makes them unaffordable for most people,” replied James Marroquin, MD, of the University of Texas at Austin. (@Jamesmarroquin).
Yoni Freedhoff, MD, of the University of Ottawa (Ont.) who writes a column for this news organization on obesity, said if tirzepatide pans out, along with other similar agents already on the market for this indication, “the next few decades should see the pharmaceutical management of obesity rival its surgical management.”
Would compete with ‘game-changer’ semaglutide?
Tirzepatide has been dubbed a “twincretin” because it works not only as an agonist of the glucagonlike peptide-1 (GLP-1) receptor, but also of the glucose-dependent insulinotropic polypeptide (GIP) receptor. It has been much hyped based on the results of the series of SURPASS clinical trials, which have formed the basis of the application for type 2 diabetes approval, about which the U.S. Food and Drug Administration is expected to make a decision soon.
Several GLP-1 agonists are on the market for both type 2 diabetes and for obesity indications separately, including semaglutide (marketed as Wegovy for obesity, also a once-weekly injection) and liraglutide (Saxenda for obesity, a daily injection), both Novo Nordisk agents.
Wegovy was approved for weight loss in the United States last year, with doctors telling this news organization then that a third of patients who take the drug are likely to lose 20% or more of their starting weight, an outcome that approaches reductions seen with bariatric surgery.
Dr. Freedhoff said he’d like to see “reimbursement by insurers who will see these drugs serving as important ancillary treatments for the myriad of weight-responsive conditions they’re already covering.”
SURMOUNT-1 data: ‘Impressive body weight’ reductions
The new tirzepatide data come from the multicenter, randomized, double-blind, placebo-controlled SURMOUNT-1 trial, which included 2539 participants from the United States, Argentina, Brazil, China, India, Japan Mexico, Russia, and Taiwan. They had obesity or overweight plus at least one comorbidity but not diabetes. They were randomized to 5-mg, 10-mg, or 15-mg once-weekly tirzepatide or placebo injections for 18 months (72 weeks).
Efficacy was analyzed in two ways. Prior to factoring in drug discontinuation, participants taking tirzepatide experienced weight loss of 16.0% (35 lb/16 kg) with 5 mg, 21.4% (49 lb/22 kg) with 10 mg, and 22.5% (52 lb/24 kg) on 15 mg. In contrast, the placebo group lost just 2.4% of body weight (5 lb/2 kg).
But treatment discontinuation rates because of adverse events were 4.3%, 7.1%, 6.2%, and 2.6%, for tirzepatide 5 mg, 10 mg, 15 mg, and placebo, respectively. Overall treatment discontinuation rates were 14.3%, 16.4%, 15.1%, and 26.4%, respectively.
When efficacy was assessed regardless of treatment discontinuation, average body weight reductions were 15.0%, 19.5%, 20.9%, and 3.1% for tirzepatide 5 mg, 10 mg, 15 mg, and placebo, respectively.
More than half of patients taking tirzepatide 10 mg and 15 mg (55% and 63%, respectively) lost at least 20% of their body weight, compared with just 1.3% taking placebo.
Overall safety and tolerability were similar to those of other GLP-1 agonists, with adverse events being gastrointestinal in nature and increasing with higher doses. Nausea affected 24.6%, 33.3%, and 31.0% of the tirzepatide 5-mg, 10-mg, and 15-mg dose groups, respectively, and vomiting was experienced by 8.3%, 10.7%, and 12.2% of patients, respectively. Diarrhea and constipation were also reported more often with the drug than placebo.
“Tirzepatide delivered impressive body weight reductions in SURMOUNT-1, which could represent an important step forward for helping the patient and physician partnership treat this complex disease,” said study investigator Louis J. Aronne, MD, director of the Comprehensive Weight Control Center and the Sanford I. Weill Professor of Metabolic Research at Weill Cornell Medicine, New York, in a press release.
Further studies are ongoing for tirzepatide as a potential treatment for obesity or overweight, according to the Lilly statement. SURMOUNT is a phase 3 global clinical development program for tirzepatide that began in late 2019 with over 5,000 people with obesity or overweight across six clinical trials. Results from SURMOUNT-2, SURMOUNT-3, and SURMOUNT-4 are expected in 2023.
Tirzepatide is also being studied as a potential treatment for nonalcoholic fatty liver disease and heart failure with preserved ejection fraction. Studies of tirzepatide in obstructive sleep apnea and of morbidity/mortality in obesity are also planned.
Dr. Aronne is cofounder, chief scientific advisor, and a member of the board of directors for Intellihealth. He is also a paid scientific advisory board member for Eli Lilly. Dr. Freedhoff has served or is serving as a director, officer, partner, employee, adviser, consultant, or trustee for the Bariatric Medical Institute and Constant Health and has received a research grant from Novo Nordisk.
A version of this article first appeared on Medscape.com.
More than half of patients taking the two highest doses of tirzepatide as a once-weekly injection lost at least 20% of their body weight in the first phase 3 trial to examine this agent in patients with obesity, but without diabetes, according to preliminary top-line results from the SURMOUNT-1 trial announced by Lilly.
The full results will be reported at an upcoming medical conference and published at a later date, Lilly added.
There was much excitement in response to the news, but others have urged caution and noted that, even if tirzepatide is eventually approved for obesity, one of the major barriers to use in the United States will be insurance coverage.
“Wow (and a double Wow!) 52lb weight loss (22.5%) at highest dose of tirzepatide,” tweeted Sek Kathiresan, MD, a cardiologist who is cofounder of Verve Therapeutics and on leave from Harvard (@skathire).
“Thus far the challenge with GLP-1s [agonists] for management of obesity is that insurance usually isn’t covering them. This makes them unaffordable for most people,” replied James Marroquin, MD, of the University of Texas at Austin. (@Jamesmarroquin).
Yoni Freedhoff, MD, of the University of Ottawa (Ont.) who writes a column for this news organization on obesity, said if tirzepatide pans out, along with other similar agents already on the market for this indication, “the next few decades should see the pharmaceutical management of obesity rival its surgical management.”
Would compete with ‘game-changer’ semaglutide?
Tirzepatide has been dubbed a “twincretin” because it works not only as an agonist of the glucagonlike peptide-1 (GLP-1) receptor, but also of the glucose-dependent insulinotropic polypeptide (GIP) receptor. It has been much hyped based on the results of the series of SURPASS clinical trials, which have formed the basis of the application for type 2 diabetes approval, about which the U.S. Food and Drug Administration is expected to make a decision soon.
Several GLP-1 agonists are on the market for both type 2 diabetes and for obesity indications separately, including semaglutide (marketed as Wegovy for obesity, also a once-weekly injection) and liraglutide (Saxenda for obesity, a daily injection), both Novo Nordisk agents.
Wegovy was approved for weight loss in the United States last year, with doctors telling this news organization then that a third of patients who take the drug are likely to lose 20% or more of their starting weight, an outcome that approaches reductions seen with bariatric surgery.
Dr. Freedhoff said he’d like to see “reimbursement by insurers who will see these drugs serving as important ancillary treatments for the myriad of weight-responsive conditions they’re already covering.”
SURMOUNT-1 data: ‘Impressive body weight’ reductions
The new tirzepatide data come from the multicenter, randomized, double-blind, placebo-controlled SURMOUNT-1 trial, which included 2539 participants from the United States, Argentina, Brazil, China, India, Japan Mexico, Russia, and Taiwan. They had obesity or overweight plus at least one comorbidity but not diabetes. They were randomized to 5-mg, 10-mg, or 15-mg once-weekly tirzepatide or placebo injections for 18 months (72 weeks).
Efficacy was analyzed in two ways. Prior to factoring in drug discontinuation, participants taking tirzepatide experienced weight loss of 16.0% (35 lb/16 kg) with 5 mg, 21.4% (49 lb/22 kg) with 10 mg, and 22.5% (52 lb/24 kg) on 15 mg. In contrast, the placebo group lost just 2.4% of body weight (5 lb/2 kg).
But treatment discontinuation rates because of adverse events were 4.3%, 7.1%, 6.2%, and 2.6%, for tirzepatide 5 mg, 10 mg, 15 mg, and placebo, respectively. Overall treatment discontinuation rates were 14.3%, 16.4%, 15.1%, and 26.4%, respectively.
When efficacy was assessed regardless of treatment discontinuation, average body weight reductions were 15.0%, 19.5%, 20.9%, and 3.1% for tirzepatide 5 mg, 10 mg, 15 mg, and placebo, respectively.
More than half of patients taking tirzepatide 10 mg and 15 mg (55% and 63%, respectively) lost at least 20% of their body weight, compared with just 1.3% taking placebo.
Overall safety and tolerability were similar to those of other GLP-1 agonists, with adverse events being gastrointestinal in nature and increasing with higher doses. Nausea affected 24.6%, 33.3%, and 31.0% of the tirzepatide 5-mg, 10-mg, and 15-mg dose groups, respectively, and vomiting was experienced by 8.3%, 10.7%, and 12.2% of patients, respectively. Diarrhea and constipation were also reported more often with the drug than placebo.
“Tirzepatide delivered impressive body weight reductions in SURMOUNT-1, which could represent an important step forward for helping the patient and physician partnership treat this complex disease,” said study investigator Louis J. Aronne, MD, director of the Comprehensive Weight Control Center and the Sanford I. Weill Professor of Metabolic Research at Weill Cornell Medicine, New York, in a press release.
Further studies are ongoing for tirzepatide as a potential treatment for obesity or overweight, according to the Lilly statement. SURMOUNT is a phase 3 global clinical development program for tirzepatide that began in late 2019 with over 5,000 people with obesity or overweight across six clinical trials. Results from SURMOUNT-2, SURMOUNT-3, and SURMOUNT-4 are expected in 2023.
Tirzepatide is also being studied as a potential treatment for nonalcoholic fatty liver disease and heart failure with preserved ejection fraction. Studies of tirzepatide in obstructive sleep apnea and of morbidity/mortality in obesity are also planned.
Dr. Aronne is cofounder, chief scientific advisor, and a member of the board of directors for Intellihealth. He is also a paid scientific advisory board member for Eli Lilly. Dr. Freedhoff has served or is serving as a director, officer, partner, employee, adviser, consultant, or trustee for the Bariatric Medical Institute and Constant Health and has received a research grant from Novo Nordisk.
A version of this article first appeared on Medscape.com.
It’s time to shame the fat shamers
Fat shaming doesn’t work. If it did, obesity as we know it wouldn’t exist because if the one thing society ensures isn’t lacking for people with obesity, it’s shame. We know that fat shaming doesn’t lead to weight loss and that it’s actually correlated with weight gain: More shame leads to more gain (Puhl and Suh; Sutin and Terracciano; Tomiyama et al).
Shaming and weight stigma have far more concerning associations than weight gain. People who report experiencing more weight stigma have an increased risk for depression, anxiety, low self-esteem, poor body image, substance abuse, suicidality, unhealthy eating behaviors, disordered eating, increased caloric intake, exercise avoidance, decreased exercise motivation potentially due to heightened cortisol reactivity, elevated C-reactive protein, and elevated blood pressure.
Meanwhile, people with obesity – likely in part owing to negative weight-biased experiences in health care – are reluctant to discuss weight with their health care providers and are less likely to seek care at all for any conditions. When care is sought, people with obesity are more likely to receive substandard treatment, including receiving fewer preventive health screenings, decreased health education, and decreased time spent in appointments.
Remember that obesity is not a conscious choice
A fact that is conveniently forgotten by those who are most prone to fat shaming is that obesity, like every chronic noncommunicable disease, isn’t a choice that is consciously made by patients.
And yes, though there are lifestyle means that might affect weight, there are lifestyle means that might affect all chronic diseases – yet obesity is the only one we seem to moralize about. It’s also worth noting that other chronic diseases’ lifestyle levers tend not to be governed by thousands of genes and dozens of hormones; those trying to “lifestyle” their way out of obesity are swimming against strong physiologic currents that influence our most seminally important survival drive: eating.
But forgetting about physiologic currents, there is also staggering privilege associated with intentional perpetual behavior change around food and fitness in the name of health.
Whereas medicine and the world are right and quick to embrace the fights against racism, sexism, and homophobia, the push to confront weight bias is far rarer, despite the fact that it’s been shown to be rampant among health care professionals.
Protecting the rights of people with obesity
Perhaps though, times are changing. Movements are popping up to protect the rights of people with obesity while combating hate.
Of note, Brazil seems to have embraced a campaign to fight gordofobia — the Portuguese term used to describe weight-based discrimination. For instance, laws are being passed to ensure appropriate seating is supplied in schools for children with obesity, an annual day was formalized to promote the rights of people with obesity, preferential seating is provided on subways for people with obesity, and fines have been levied against at least one comedian for making fat jokes on the grounds of the state’s duty to protect minorities.
We need to take this fight to medicine. Given the incredibly depressing prevalence of weight bias among trainees, medical schools and residency programs should ensure countering weight bias is not only part of the curriculum but that it’s explicitly examined. National medical licensing examinations should include weight bias as well.
Though we’re closer than ever before to widely effective treatment options for obesity, it’s likely to still be decades before pharmaceutical options to treat obesity are as effective, accepted, and encouraged as medications to treat hypertension, dyslipidemia, diabetes, and more are today.
If you’re curious about your own implicit weight biases, consider taking Harvard’s Implicit Association Test for Weight. You might also want to take a few moments and review the Strategies to Overcome and Prevent Obesity Alliances’ Weight Can’t Wait guide for advice on the management of obesity in primary care.
Treat patients with obesity the same as you would those with any chronic condition.
Also, consider your physical office space. Do you have chairs suitable for patients with obesity (wide base and with arms to help patients rise)? A scale that measures up to high weights that’s in a private location? Appropriately sized blood pressure cuffs?
If not,
Examples include the family doctor who hadn’t checked my patient’s blood pressure in over a decade because he couldn’t be bothered buying an appropriately sized blood pressure cuff. Or the fertility doctor who told one of my patients that perhaps her weight reflected God’s will that she does not have children.
Finally, if reading this article about treating people with obesity the same as you would patients with other chronic, noncommunicable, lifestyle responsive diseases made you angry, there’s a great chance that you’re part of the problem.
Dr. Freedhoff, is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of The Diet Fix: Why Diets Fail and How to Make Yours Work. He has disclosed the following: He served as a director, officer, partner, employee, adviser, consultant, or trustee for Bariatric Medical Institute and Constant Health; has received research grant from Novo Nordisk, and has publicly shared opinions via Weighty Matters and social media. A version of this article first appeared on Medscape.com.
Fat shaming doesn’t work. If it did, obesity as we know it wouldn’t exist because if the one thing society ensures isn’t lacking for people with obesity, it’s shame. We know that fat shaming doesn’t lead to weight loss and that it’s actually correlated with weight gain: More shame leads to more gain (Puhl and Suh; Sutin and Terracciano; Tomiyama et al).
Shaming and weight stigma have far more concerning associations than weight gain. People who report experiencing more weight stigma have an increased risk for depression, anxiety, low self-esteem, poor body image, substance abuse, suicidality, unhealthy eating behaviors, disordered eating, increased caloric intake, exercise avoidance, decreased exercise motivation potentially due to heightened cortisol reactivity, elevated C-reactive protein, and elevated blood pressure.
Meanwhile, people with obesity – likely in part owing to negative weight-biased experiences in health care – are reluctant to discuss weight with their health care providers and are less likely to seek care at all for any conditions. When care is sought, people with obesity are more likely to receive substandard treatment, including receiving fewer preventive health screenings, decreased health education, and decreased time spent in appointments.
Remember that obesity is not a conscious choice
A fact that is conveniently forgotten by those who are most prone to fat shaming is that obesity, like every chronic noncommunicable disease, isn’t a choice that is consciously made by patients.
And yes, though there are lifestyle means that might affect weight, there are lifestyle means that might affect all chronic diseases – yet obesity is the only one we seem to moralize about. It’s also worth noting that other chronic diseases’ lifestyle levers tend not to be governed by thousands of genes and dozens of hormones; those trying to “lifestyle” their way out of obesity are swimming against strong physiologic currents that influence our most seminally important survival drive: eating.
But forgetting about physiologic currents, there is also staggering privilege associated with intentional perpetual behavior change around food and fitness in the name of health.
Whereas medicine and the world are right and quick to embrace the fights against racism, sexism, and homophobia, the push to confront weight bias is far rarer, despite the fact that it’s been shown to be rampant among health care professionals.
Protecting the rights of people with obesity
Perhaps though, times are changing. Movements are popping up to protect the rights of people with obesity while combating hate.
Of note, Brazil seems to have embraced a campaign to fight gordofobia — the Portuguese term used to describe weight-based discrimination. For instance, laws are being passed to ensure appropriate seating is supplied in schools for children with obesity, an annual day was formalized to promote the rights of people with obesity, preferential seating is provided on subways for people with obesity, and fines have been levied against at least one comedian for making fat jokes on the grounds of the state’s duty to protect minorities.
We need to take this fight to medicine. Given the incredibly depressing prevalence of weight bias among trainees, medical schools and residency programs should ensure countering weight bias is not only part of the curriculum but that it’s explicitly examined. National medical licensing examinations should include weight bias as well.
Though we’re closer than ever before to widely effective treatment options for obesity, it’s likely to still be decades before pharmaceutical options to treat obesity are as effective, accepted, and encouraged as medications to treat hypertension, dyslipidemia, diabetes, and more are today.
If you’re curious about your own implicit weight biases, consider taking Harvard’s Implicit Association Test for Weight. You might also want to take a few moments and review the Strategies to Overcome and Prevent Obesity Alliances’ Weight Can’t Wait guide for advice on the management of obesity in primary care.
Treat patients with obesity the same as you would those with any chronic condition.
Also, consider your physical office space. Do you have chairs suitable for patients with obesity (wide base and with arms to help patients rise)? A scale that measures up to high weights that’s in a private location? Appropriately sized blood pressure cuffs?
If not,
Examples include the family doctor who hadn’t checked my patient’s blood pressure in over a decade because he couldn’t be bothered buying an appropriately sized blood pressure cuff. Or the fertility doctor who told one of my patients that perhaps her weight reflected God’s will that she does not have children.
Finally, if reading this article about treating people with obesity the same as you would patients with other chronic, noncommunicable, lifestyle responsive diseases made you angry, there’s a great chance that you’re part of the problem.
Dr. Freedhoff, is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of The Diet Fix: Why Diets Fail and How to Make Yours Work. He has disclosed the following: He served as a director, officer, partner, employee, adviser, consultant, or trustee for Bariatric Medical Institute and Constant Health; has received research grant from Novo Nordisk, and has publicly shared opinions via Weighty Matters and social media. A version of this article first appeared on Medscape.com.
Fat shaming doesn’t work. If it did, obesity as we know it wouldn’t exist because if the one thing society ensures isn’t lacking for people with obesity, it’s shame. We know that fat shaming doesn’t lead to weight loss and that it’s actually correlated with weight gain: More shame leads to more gain (Puhl and Suh; Sutin and Terracciano; Tomiyama et al).
Shaming and weight stigma have far more concerning associations than weight gain. People who report experiencing more weight stigma have an increased risk for depression, anxiety, low self-esteem, poor body image, substance abuse, suicidality, unhealthy eating behaviors, disordered eating, increased caloric intake, exercise avoidance, decreased exercise motivation potentially due to heightened cortisol reactivity, elevated C-reactive protein, and elevated blood pressure.
Meanwhile, people with obesity – likely in part owing to negative weight-biased experiences in health care – are reluctant to discuss weight with their health care providers and are less likely to seek care at all for any conditions. When care is sought, people with obesity are more likely to receive substandard treatment, including receiving fewer preventive health screenings, decreased health education, and decreased time spent in appointments.
Remember that obesity is not a conscious choice
A fact that is conveniently forgotten by those who are most prone to fat shaming is that obesity, like every chronic noncommunicable disease, isn’t a choice that is consciously made by patients.
And yes, though there are lifestyle means that might affect weight, there are lifestyle means that might affect all chronic diseases – yet obesity is the only one we seem to moralize about. It’s also worth noting that other chronic diseases’ lifestyle levers tend not to be governed by thousands of genes and dozens of hormones; those trying to “lifestyle” their way out of obesity are swimming against strong physiologic currents that influence our most seminally important survival drive: eating.
But forgetting about physiologic currents, there is also staggering privilege associated with intentional perpetual behavior change around food and fitness in the name of health.
Whereas medicine and the world are right and quick to embrace the fights against racism, sexism, and homophobia, the push to confront weight bias is far rarer, despite the fact that it’s been shown to be rampant among health care professionals.
Protecting the rights of people with obesity
Perhaps though, times are changing. Movements are popping up to protect the rights of people with obesity while combating hate.
Of note, Brazil seems to have embraced a campaign to fight gordofobia — the Portuguese term used to describe weight-based discrimination. For instance, laws are being passed to ensure appropriate seating is supplied in schools for children with obesity, an annual day was formalized to promote the rights of people with obesity, preferential seating is provided on subways for people with obesity, and fines have been levied against at least one comedian for making fat jokes on the grounds of the state’s duty to protect minorities.
We need to take this fight to medicine. Given the incredibly depressing prevalence of weight bias among trainees, medical schools and residency programs should ensure countering weight bias is not only part of the curriculum but that it’s explicitly examined. National medical licensing examinations should include weight bias as well.
Though we’re closer than ever before to widely effective treatment options for obesity, it’s likely to still be decades before pharmaceutical options to treat obesity are as effective, accepted, and encouraged as medications to treat hypertension, dyslipidemia, diabetes, and more are today.
If you’re curious about your own implicit weight biases, consider taking Harvard’s Implicit Association Test for Weight. You might also want to take a few moments and review the Strategies to Overcome and Prevent Obesity Alliances’ Weight Can’t Wait guide for advice on the management of obesity in primary care.
Treat patients with obesity the same as you would those with any chronic condition.
Also, consider your physical office space. Do you have chairs suitable for patients with obesity (wide base and with arms to help patients rise)? A scale that measures up to high weights that’s in a private location? Appropriately sized blood pressure cuffs?
If not,
Examples include the family doctor who hadn’t checked my patient’s blood pressure in over a decade because he couldn’t be bothered buying an appropriately sized blood pressure cuff. Or the fertility doctor who told one of my patients that perhaps her weight reflected God’s will that she does not have children.
Finally, if reading this article about treating people with obesity the same as you would patients with other chronic, noncommunicable, lifestyle responsive diseases made you angry, there’s a great chance that you’re part of the problem.
Dr. Freedhoff, is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of The Diet Fix: Why Diets Fail and How to Make Yours Work. He has disclosed the following: He served as a director, officer, partner, employee, adviser, consultant, or trustee for Bariatric Medical Institute and Constant Health; has received research grant from Novo Nordisk, and has publicly shared opinions via Weighty Matters and social media. A version of this article first appeared on Medscape.com.
How old is too old to work as a doctor?
Air traffic controllers face mandatory retirement at age 56, with exceptions up to 61. Commercial airline pilots must bow out at 65; same for foreign service employees. Physicians, however, have no age limit, regardless of specialty.
As the profession rapidly ages – some 30% of the physician workforce is currently a senior, according to the American Medical Association – the topic of whether or not there should be a standard measure or age for retirement is front and center. The AMA’s Council on Medical Education formed a workgroup to look into the issue in 2015 and 2018, and in 2021, delegates adopted a set of guidelines for screening and assessing physicians, but stopped short of a mandate.
Mark Katlic, MD, chair of surgery at Lifebridge Health System, Baltimore, has devoted a decade to studying this topic. “I’m a bit of an outlier looking into this,” he says. “The public is unaware and seemingly unconcerned about the issue. Even among the medical profession, there’s been a series of fits and starts to develop a cohesive approach.”
One of the reasons guidelines – mandatory or otherwise – have been tough to come by is that aging brings with it a huge degree of variability. “If you look at a group of 80-year-olds, there will be much more variability than within a group of 40-year-olds,” Dr. Katlic pointed out.
Indeed, some 80-year-olds can easily continue to teach college courses, keep up in 10K running races, or perform delicate surgeries. Yet others in their peer group might struggle to properly button a shirt, walk a flight of stairs, or remember yesterday’s meals. Functional age is not the same as chronological age.
Frank Stockdale, MD, PhD, an 86-year-old practicing oncologist at Stanford (Calif.) University Health, counts himself in the camp opposed to age-based assessments. “It’s age discrimination,” he says. “Physicians receive assessments throughout their careers as part of the accreditation process – there’s no need to change that as doctors reach a certain age.”
Dr. Stockdale suggests that in many cases, malpractice suits are filed against mid-career doctors, not those of advanced age. “If you’re using the argument that there is an accumulation of deficits with age, the fact is that those deficits begin well before your 70s,” he said. “It’s better to have a uniform screening policy and begin at a much younger age.”
At Stanford, in fact, there was a former assessment policy that included cognitive testing, but physicians were successful in seeing that portion of testing eliminated. “It is a physical examination, by a physician of choice, certifying that for the privileges requested there is no physical or mental reason the candidate cannot safely perform them,” Dr. Stockdale explained.
In some cases, medical staffs have filed lawsuits to fight age-related testing. In New Haven, Conn., for instance, the U.S. Equal Employment Opportunity Commission (EEOC) filed a suit in 2020 on behalf of the Yale New Haven Hospital staff, alleging a discriminatory “late career practitioner policy.”
A similar case in Minnesota reached a settlement in 2021, providing monetary relief to staff impacted by out-of-pocket costs for the assessment, in addition to requiring that the hospital in question report to the EEOC any complaints related to age discrimination.
James Ellison, MD, MPH, chair in Memory Care and Geriatrics with ChristianaCare in Wilmington, Del., points out that aging can bring benefits for practicing physicians. “Age is very individualized and there are good and bad consequences,” he said. “Experience can build knowledge and confidence and expertise, and it does improve diagnostic accuracy.”
On the flip side, however, age-related brain changes include loss of volume and lower levels of some neurotransmitters, resulting in cognitive changes. “Functional changes occur too,” Dr. Ellison said.
“Just as some aging athletes may lose a degree of speed, strength, and flexibility, and some aging scientists may lose a part of their former cognitive speed, flexibility, and mental strength, aging health care providers can lose some of the physical coordination, strength, and visual acuity necessary to perform demanding surgical operations. They can also lose some of the processing speed, working memory, and executive function that allows them to excel in cognitive professional tasks.”
An estimated 5.8 million Americans age 65 and older have Alzheimer’s dementia, according to the Alzheimer’s Association.
Picking an arbitrary age for mandatory retirement isn’t the right approach for physicians, said Dr. Katlic. Rather, he said, the answer is to establish late-practitioner screening programs. “Very few hospitals have them, however,” he pointed out. “We do [at Lifebridge Health], and so do a few dozen others, but that’s out of hundreds.”
Instead, what typically plays out is that hospital staff might begin to notice a decline in a colleague. Things like a disheveled appearance or lack of hygiene, or trouble with memory, such as getting lost en route back to his or her office. Even dangerous behaviors such as nodding off during a procedure are not unheard of.
There are many examples of physician decline that fly under the radar. “Unfortunately, it’s unusual for cognitively impaired health care providers to recognize and report their own difficulties,” said Dr. Ellison. “Although peers are expected to report cognitively impaired colleagues, they often fail to do so. In some other countries, age-based assessment is an accepted policy. In the U.S., this is not a uniform policy.”
Sometimes physicians can remain on the job in spite of decline thanks to certain “props,” according to Dr. Ellison. “Good procedures, efficient supports, and various workarounds compensate,” he said, “but often are not sufficient to maintain high-quality practice.”
Most often, these situations play out slowly, until the problem becomes glaringly obvious and potentially dangerous, and someone in a position of power must step in.
“Often, it’s hearsay from a nurse or another staff member, and then a hospital president or chief of staff must make a career-affecting decision for the doctor in question,” said Dr. Katlic.
Because there is little self- or colleague policing – and barring official or binding guidelines on the aging physician issue – both Dr. Katlic and Dr. Ellison are proponents of late-career screening.
How screening can help
As it stands, Dr. Katlic maintains that the profession isn’t doing enough to ensure public safety. “We have peer review and recertification processes, but when you get down to it, we don’t police ourselves well,” he said. “All physicians are assessed throughout their careers as part of the hospital accreditation process, which is fair and adequate.”
Dr. Katlic said that there are three main benchmarks that physicians should be able to meet at an agreed upon age: a physical exam, a neurocognitive screening, and an eye exam. “At some reasonable age, I personally believe these exams should take place,” he said. “We can allow doctors to pick their own practitioners for the eye and physical exams, but the neurocognitive exam should be completed by a PhD neuropsychologist.”
At Lifebridge, for instance, these screenings begin at age 75 and take place every 2 years, during the recredentialing process. It applies to all specialties, not just surgeons. “Surgery is a little different in that it requires fine motor skills in addition to the others we test, but you want any physician to be cognitively intact,” Dr. Katlic pointed out. “All doctors need the ability to make decisions quickly, often under noisy, distracting conditions.”
Dr. Ellison supports applying the screenings to all specialties. “Let’s not forget that all physicians must be alert to the many ways in which their patients reveal what needs attention, evaluation, and treatment,” he said. “Some health care tasks could be performed without visual input; for example, perhaps psychotherapy could be provided competently by a clinician who lacks visual acuity. Auditory input might not be necessary for reading x-rays – but the information a health care provider gets from their eyes and ears is important, not just for surgeons.”
University of California San Diego has established what it calls its Physician Assessment and Clinical Education (PACE) program. One of the nation’s oldest and largest such programs, the hospital founded PACE in 1996. Most physicians taking part arrive as a requirement of disciplinary action from the state medical board, but a small percentage self-refers.
PACE involves two phases. The first is a 2-day set of tests and measures core competency knowledge. Phase 2 is more comprehensive and lasts 5 days. Here, within their specialty, physicians participate in the activities of the corresponding residency program. Faculty evaluates the physician, and a multidisciplinary team meets to review all the findings of the combined phases.
Depending on the results, doctors may face remediation steps that range from programs to address performance deficiencies to residency-level clinical experiences. According to a paper on the program published by the institution, “most physicians referred to the PACE program are found to have mild to moderate performance dyscompetence.”
In the case of the 2021 guidelines adopted by AMA delegates, there are nine principles for assessment. They should be evidence-based, ethical, relevant, accountable, fair and equitable, transparent, supportive, and nonburdensome, and should afford physicians due process protections.
Looking ahead
Even Dr. Katlic worries about the possibility of Congress intervening to establish federal-level, mandatory retirement age. “This just doesn’t make sense for our profession given the great variability we see,” he said. “My biggest hope is that more individual hospitals will institute these screenings.”
As the physician population ages – and the influx of new doctors shrinks – the slope becomes even more slippery. The AMA is predicting a physician shortage of nearly 40,000 by the year 2034. This strengthens arguments to keep existing physicians practicing for as long as possible and might make institutions less likely to screen.
It’s all a delicate balancing act and a continuing work in progress, said Dr. Ellison. “Ultimately, I believe we need to find a way to understand and address the possible implications for public safety, while at the same time protecting the privacy and dignity of our valued older physicians and other health care providers.”
A version of this article first appeared on Medscape.com.
Air traffic controllers face mandatory retirement at age 56, with exceptions up to 61. Commercial airline pilots must bow out at 65; same for foreign service employees. Physicians, however, have no age limit, regardless of specialty.
As the profession rapidly ages – some 30% of the physician workforce is currently a senior, according to the American Medical Association – the topic of whether or not there should be a standard measure or age for retirement is front and center. The AMA’s Council on Medical Education formed a workgroup to look into the issue in 2015 and 2018, and in 2021, delegates adopted a set of guidelines for screening and assessing physicians, but stopped short of a mandate.
Mark Katlic, MD, chair of surgery at Lifebridge Health System, Baltimore, has devoted a decade to studying this topic. “I’m a bit of an outlier looking into this,” he says. “The public is unaware and seemingly unconcerned about the issue. Even among the medical profession, there’s been a series of fits and starts to develop a cohesive approach.”
One of the reasons guidelines – mandatory or otherwise – have been tough to come by is that aging brings with it a huge degree of variability. “If you look at a group of 80-year-olds, there will be much more variability than within a group of 40-year-olds,” Dr. Katlic pointed out.
Indeed, some 80-year-olds can easily continue to teach college courses, keep up in 10K running races, or perform delicate surgeries. Yet others in their peer group might struggle to properly button a shirt, walk a flight of stairs, or remember yesterday’s meals. Functional age is not the same as chronological age.
Frank Stockdale, MD, PhD, an 86-year-old practicing oncologist at Stanford (Calif.) University Health, counts himself in the camp opposed to age-based assessments. “It’s age discrimination,” he says. “Physicians receive assessments throughout their careers as part of the accreditation process – there’s no need to change that as doctors reach a certain age.”
Dr. Stockdale suggests that in many cases, malpractice suits are filed against mid-career doctors, not those of advanced age. “If you’re using the argument that there is an accumulation of deficits with age, the fact is that those deficits begin well before your 70s,” he said. “It’s better to have a uniform screening policy and begin at a much younger age.”
At Stanford, in fact, there was a former assessment policy that included cognitive testing, but physicians were successful in seeing that portion of testing eliminated. “It is a physical examination, by a physician of choice, certifying that for the privileges requested there is no physical or mental reason the candidate cannot safely perform them,” Dr. Stockdale explained.
In some cases, medical staffs have filed lawsuits to fight age-related testing. In New Haven, Conn., for instance, the U.S. Equal Employment Opportunity Commission (EEOC) filed a suit in 2020 on behalf of the Yale New Haven Hospital staff, alleging a discriminatory “late career practitioner policy.”
A similar case in Minnesota reached a settlement in 2021, providing monetary relief to staff impacted by out-of-pocket costs for the assessment, in addition to requiring that the hospital in question report to the EEOC any complaints related to age discrimination.
James Ellison, MD, MPH, chair in Memory Care and Geriatrics with ChristianaCare in Wilmington, Del., points out that aging can bring benefits for practicing physicians. “Age is very individualized and there are good and bad consequences,” he said. “Experience can build knowledge and confidence and expertise, and it does improve diagnostic accuracy.”
On the flip side, however, age-related brain changes include loss of volume and lower levels of some neurotransmitters, resulting in cognitive changes. “Functional changes occur too,” Dr. Ellison said.
“Just as some aging athletes may lose a degree of speed, strength, and flexibility, and some aging scientists may lose a part of their former cognitive speed, flexibility, and mental strength, aging health care providers can lose some of the physical coordination, strength, and visual acuity necessary to perform demanding surgical operations. They can also lose some of the processing speed, working memory, and executive function that allows them to excel in cognitive professional tasks.”
An estimated 5.8 million Americans age 65 and older have Alzheimer’s dementia, according to the Alzheimer’s Association.
Picking an arbitrary age for mandatory retirement isn’t the right approach for physicians, said Dr. Katlic. Rather, he said, the answer is to establish late-practitioner screening programs. “Very few hospitals have them, however,” he pointed out. “We do [at Lifebridge Health], and so do a few dozen others, but that’s out of hundreds.”
Instead, what typically plays out is that hospital staff might begin to notice a decline in a colleague. Things like a disheveled appearance or lack of hygiene, or trouble with memory, such as getting lost en route back to his or her office. Even dangerous behaviors such as nodding off during a procedure are not unheard of.
There are many examples of physician decline that fly under the radar. “Unfortunately, it’s unusual for cognitively impaired health care providers to recognize and report their own difficulties,” said Dr. Ellison. “Although peers are expected to report cognitively impaired colleagues, they often fail to do so. In some other countries, age-based assessment is an accepted policy. In the U.S., this is not a uniform policy.”
Sometimes physicians can remain on the job in spite of decline thanks to certain “props,” according to Dr. Ellison. “Good procedures, efficient supports, and various workarounds compensate,” he said, “but often are not sufficient to maintain high-quality practice.”
Most often, these situations play out slowly, until the problem becomes glaringly obvious and potentially dangerous, and someone in a position of power must step in.
“Often, it’s hearsay from a nurse or another staff member, and then a hospital president or chief of staff must make a career-affecting decision for the doctor in question,” said Dr. Katlic.
Because there is little self- or colleague policing – and barring official or binding guidelines on the aging physician issue – both Dr. Katlic and Dr. Ellison are proponents of late-career screening.
How screening can help
As it stands, Dr. Katlic maintains that the profession isn’t doing enough to ensure public safety. “We have peer review and recertification processes, but when you get down to it, we don’t police ourselves well,” he said. “All physicians are assessed throughout their careers as part of the hospital accreditation process, which is fair and adequate.”
Dr. Katlic said that there are three main benchmarks that physicians should be able to meet at an agreed upon age: a physical exam, a neurocognitive screening, and an eye exam. “At some reasonable age, I personally believe these exams should take place,” he said. “We can allow doctors to pick their own practitioners for the eye and physical exams, but the neurocognitive exam should be completed by a PhD neuropsychologist.”
At Lifebridge, for instance, these screenings begin at age 75 and take place every 2 years, during the recredentialing process. It applies to all specialties, not just surgeons. “Surgery is a little different in that it requires fine motor skills in addition to the others we test, but you want any physician to be cognitively intact,” Dr. Katlic pointed out. “All doctors need the ability to make decisions quickly, often under noisy, distracting conditions.”
Dr. Ellison supports applying the screenings to all specialties. “Let’s not forget that all physicians must be alert to the many ways in which their patients reveal what needs attention, evaluation, and treatment,” he said. “Some health care tasks could be performed without visual input; for example, perhaps psychotherapy could be provided competently by a clinician who lacks visual acuity. Auditory input might not be necessary for reading x-rays – but the information a health care provider gets from their eyes and ears is important, not just for surgeons.”
University of California San Diego has established what it calls its Physician Assessment and Clinical Education (PACE) program. One of the nation’s oldest and largest such programs, the hospital founded PACE in 1996. Most physicians taking part arrive as a requirement of disciplinary action from the state medical board, but a small percentage self-refers.
PACE involves two phases. The first is a 2-day set of tests and measures core competency knowledge. Phase 2 is more comprehensive and lasts 5 days. Here, within their specialty, physicians participate in the activities of the corresponding residency program. Faculty evaluates the physician, and a multidisciplinary team meets to review all the findings of the combined phases.
Depending on the results, doctors may face remediation steps that range from programs to address performance deficiencies to residency-level clinical experiences. According to a paper on the program published by the institution, “most physicians referred to the PACE program are found to have mild to moderate performance dyscompetence.”
In the case of the 2021 guidelines adopted by AMA delegates, there are nine principles for assessment. They should be evidence-based, ethical, relevant, accountable, fair and equitable, transparent, supportive, and nonburdensome, and should afford physicians due process protections.
Looking ahead
Even Dr. Katlic worries about the possibility of Congress intervening to establish federal-level, mandatory retirement age. “This just doesn’t make sense for our profession given the great variability we see,” he said. “My biggest hope is that more individual hospitals will institute these screenings.”
As the physician population ages – and the influx of new doctors shrinks – the slope becomes even more slippery. The AMA is predicting a physician shortage of nearly 40,000 by the year 2034. This strengthens arguments to keep existing physicians practicing for as long as possible and might make institutions less likely to screen.
It’s all a delicate balancing act and a continuing work in progress, said Dr. Ellison. “Ultimately, I believe we need to find a way to understand and address the possible implications for public safety, while at the same time protecting the privacy and dignity of our valued older physicians and other health care providers.”
A version of this article first appeared on Medscape.com.
Air traffic controllers face mandatory retirement at age 56, with exceptions up to 61. Commercial airline pilots must bow out at 65; same for foreign service employees. Physicians, however, have no age limit, regardless of specialty.
As the profession rapidly ages – some 30% of the physician workforce is currently a senior, according to the American Medical Association – the topic of whether or not there should be a standard measure or age for retirement is front and center. The AMA’s Council on Medical Education formed a workgroup to look into the issue in 2015 and 2018, and in 2021, delegates adopted a set of guidelines for screening and assessing physicians, but stopped short of a mandate.
Mark Katlic, MD, chair of surgery at Lifebridge Health System, Baltimore, has devoted a decade to studying this topic. “I’m a bit of an outlier looking into this,” he says. “The public is unaware and seemingly unconcerned about the issue. Even among the medical profession, there’s been a series of fits and starts to develop a cohesive approach.”
One of the reasons guidelines – mandatory or otherwise – have been tough to come by is that aging brings with it a huge degree of variability. “If you look at a group of 80-year-olds, there will be much more variability than within a group of 40-year-olds,” Dr. Katlic pointed out.
Indeed, some 80-year-olds can easily continue to teach college courses, keep up in 10K running races, or perform delicate surgeries. Yet others in their peer group might struggle to properly button a shirt, walk a flight of stairs, or remember yesterday’s meals. Functional age is not the same as chronological age.
Frank Stockdale, MD, PhD, an 86-year-old practicing oncologist at Stanford (Calif.) University Health, counts himself in the camp opposed to age-based assessments. “It’s age discrimination,” he says. “Physicians receive assessments throughout their careers as part of the accreditation process – there’s no need to change that as doctors reach a certain age.”
Dr. Stockdale suggests that in many cases, malpractice suits are filed against mid-career doctors, not those of advanced age. “If you’re using the argument that there is an accumulation of deficits with age, the fact is that those deficits begin well before your 70s,” he said. “It’s better to have a uniform screening policy and begin at a much younger age.”
At Stanford, in fact, there was a former assessment policy that included cognitive testing, but physicians were successful in seeing that portion of testing eliminated. “It is a physical examination, by a physician of choice, certifying that for the privileges requested there is no physical or mental reason the candidate cannot safely perform them,” Dr. Stockdale explained.
In some cases, medical staffs have filed lawsuits to fight age-related testing. In New Haven, Conn., for instance, the U.S. Equal Employment Opportunity Commission (EEOC) filed a suit in 2020 on behalf of the Yale New Haven Hospital staff, alleging a discriminatory “late career practitioner policy.”
A similar case in Minnesota reached a settlement in 2021, providing monetary relief to staff impacted by out-of-pocket costs for the assessment, in addition to requiring that the hospital in question report to the EEOC any complaints related to age discrimination.
James Ellison, MD, MPH, chair in Memory Care and Geriatrics with ChristianaCare in Wilmington, Del., points out that aging can bring benefits for practicing physicians. “Age is very individualized and there are good and bad consequences,” he said. “Experience can build knowledge and confidence and expertise, and it does improve diagnostic accuracy.”
On the flip side, however, age-related brain changes include loss of volume and lower levels of some neurotransmitters, resulting in cognitive changes. “Functional changes occur too,” Dr. Ellison said.
“Just as some aging athletes may lose a degree of speed, strength, and flexibility, and some aging scientists may lose a part of their former cognitive speed, flexibility, and mental strength, aging health care providers can lose some of the physical coordination, strength, and visual acuity necessary to perform demanding surgical operations. They can also lose some of the processing speed, working memory, and executive function that allows them to excel in cognitive professional tasks.”
An estimated 5.8 million Americans age 65 and older have Alzheimer’s dementia, according to the Alzheimer’s Association.
Picking an arbitrary age for mandatory retirement isn’t the right approach for physicians, said Dr. Katlic. Rather, he said, the answer is to establish late-practitioner screening programs. “Very few hospitals have them, however,” he pointed out. “We do [at Lifebridge Health], and so do a few dozen others, but that’s out of hundreds.”
Instead, what typically plays out is that hospital staff might begin to notice a decline in a colleague. Things like a disheveled appearance or lack of hygiene, or trouble with memory, such as getting lost en route back to his or her office. Even dangerous behaviors such as nodding off during a procedure are not unheard of.
There are many examples of physician decline that fly under the radar. “Unfortunately, it’s unusual for cognitively impaired health care providers to recognize and report their own difficulties,” said Dr. Ellison. “Although peers are expected to report cognitively impaired colleagues, they often fail to do so. In some other countries, age-based assessment is an accepted policy. In the U.S., this is not a uniform policy.”
Sometimes physicians can remain on the job in spite of decline thanks to certain “props,” according to Dr. Ellison. “Good procedures, efficient supports, and various workarounds compensate,” he said, “but often are not sufficient to maintain high-quality practice.”
Most often, these situations play out slowly, until the problem becomes glaringly obvious and potentially dangerous, and someone in a position of power must step in.
“Often, it’s hearsay from a nurse or another staff member, and then a hospital president or chief of staff must make a career-affecting decision for the doctor in question,” said Dr. Katlic.
Because there is little self- or colleague policing – and barring official or binding guidelines on the aging physician issue – both Dr. Katlic and Dr. Ellison are proponents of late-career screening.
How screening can help
As it stands, Dr. Katlic maintains that the profession isn’t doing enough to ensure public safety. “We have peer review and recertification processes, but when you get down to it, we don’t police ourselves well,” he said. “All physicians are assessed throughout their careers as part of the hospital accreditation process, which is fair and adequate.”
Dr. Katlic said that there are three main benchmarks that physicians should be able to meet at an agreed upon age: a physical exam, a neurocognitive screening, and an eye exam. “At some reasonable age, I personally believe these exams should take place,” he said. “We can allow doctors to pick their own practitioners for the eye and physical exams, but the neurocognitive exam should be completed by a PhD neuropsychologist.”
At Lifebridge, for instance, these screenings begin at age 75 and take place every 2 years, during the recredentialing process. It applies to all specialties, not just surgeons. “Surgery is a little different in that it requires fine motor skills in addition to the others we test, but you want any physician to be cognitively intact,” Dr. Katlic pointed out. “All doctors need the ability to make decisions quickly, often under noisy, distracting conditions.”
Dr. Ellison supports applying the screenings to all specialties. “Let’s not forget that all physicians must be alert to the many ways in which their patients reveal what needs attention, evaluation, and treatment,” he said. “Some health care tasks could be performed without visual input; for example, perhaps psychotherapy could be provided competently by a clinician who lacks visual acuity. Auditory input might not be necessary for reading x-rays – but the information a health care provider gets from their eyes and ears is important, not just for surgeons.”
University of California San Diego has established what it calls its Physician Assessment and Clinical Education (PACE) program. One of the nation’s oldest and largest such programs, the hospital founded PACE in 1996. Most physicians taking part arrive as a requirement of disciplinary action from the state medical board, but a small percentage self-refers.
PACE involves two phases. The first is a 2-day set of tests and measures core competency knowledge. Phase 2 is more comprehensive and lasts 5 days. Here, within their specialty, physicians participate in the activities of the corresponding residency program. Faculty evaluates the physician, and a multidisciplinary team meets to review all the findings of the combined phases.
Depending on the results, doctors may face remediation steps that range from programs to address performance deficiencies to residency-level clinical experiences. According to a paper on the program published by the institution, “most physicians referred to the PACE program are found to have mild to moderate performance dyscompetence.”
In the case of the 2021 guidelines adopted by AMA delegates, there are nine principles for assessment. They should be evidence-based, ethical, relevant, accountable, fair and equitable, transparent, supportive, and nonburdensome, and should afford physicians due process protections.
Looking ahead
Even Dr. Katlic worries about the possibility of Congress intervening to establish federal-level, mandatory retirement age. “This just doesn’t make sense for our profession given the great variability we see,” he said. “My biggest hope is that more individual hospitals will institute these screenings.”
As the physician population ages – and the influx of new doctors shrinks – the slope becomes even more slippery. The AMA is predicting a physician shortage of nearly 40,000 by the year 2034. This strengthens arguments to keep existing physicians practicing for as long as possible and might make institutions less likely to screen.
It’s all a delicate balancing act and a continuing work in progress, said Dr. Ellison. “Ultimately, I believe we need to find a way to understand and address the possible implications for public safety, while at the same time protecting the privacy and dignity of our valued older physicians and other health care providers.”
A version of this article first appeared on Medscape.com.
30 years of fake nursing ends with 7-year prison sentence
A Canadian woman who officials allege faked being a registered nurse for some 30 years in Canada and the United States is scheduled to appear in court next month after being sentenced to 7 years in prison.
She was previously sentenced April 22 in an Ontario court after she pled guilty in January to seven offenses, including impersonation, assault with a weapon, and assault, according to CBC Radio-Canada.
Ms. Cleroux, who uses several aliases, had a long history of deception in three provinces in Canada, as well as in Colorado and Florida. The sentencing in Ontario stemmed from incidents at a medical and dental clinic in Ottawa last year, which included administration of medications to patients through needle injections, Ottawa Police reported in a press statement obtained by this news organization.
Authorities charged Ms. Cleroux in September with assault with a weapon and criminal negligence causing bodily harm, along with “personation to gain advantage,” obtaining by false pretense, and using a forged document, this news organization reported.
Ms. Cleroux has been in custody since her arrest by Ottawa Police in August.
The Vancouver Police Department (VPD) charged Ms. Cleroux last year with fraud of over $5,000 and personation with intent. VPD investigated claims that an employee at BC Women’s Hospital fraudulently identified herself as a nurse while working there between June 2020 and June 2021, according to a VPD press release.
Nursing colleges in British Columbia and Ontario issued warnings that she had used aliases and purported to be a registered nurse to gain employment. The aliases included Melanie Thompson, Melanie Smith, and Melanie Cleroux.
Ms. Cleroux was believed to be a student in a nursing school in Colorado, but she only completed 2 years of a 4-year nursing course and was never certified as a nurse, according to CBC. Her criminal record dates back 30 years and includes 67 adult convictions and other convictions in her youth, CBC reported.
A version of this article first appeared on Medscape.com.
A Canadian woman who officials allege faked being a registered nurse for some 30 years in Canada and the United States is scheduled to appear in court next month after being sentenced to 7 years in prison.
She was previously sentenced April 22 in an Ontario court after she pled guilty in January to seven offenses, including impersonation, assault with a weapon, and assault, according to CBC Radio-Canada.
Ms. Cleroux, who uses several aliases, had a long history of deception in three provinces in Canada, as well as in Colorado and Florida. The sentencing in Ontario stemmed from incidents at a medical and dental clinic in Ottawa last year, which included administration of medications to patients through needle injections, Ottawa Police reported in a press statement obtained by this news organization.
Authorities charged Ms. Cleroux in September with assault with a weapon and criminal negligence causing bodily harm, along with “personation to gain advantage,” obtaining by false pretense, and using a forged document, this news organization reported.
Ms. Cleroux has been in custody since her arrest by Ottawa Police in August.
The Vancouver Police Department (VPD) charged Ms. Cleroux last year with fraud of over $5,000 and personation with intent. VPD investigated claims that an employee at BC Women’s Hospital fraudulently identified herself as a nurse while working there between June 2020 and June 2021, according to a VPD press release.
Nursing colleges in British Columbia and Ontario issued warnings that she had used aliases and purported to be a registered nurse to gain employment. The aliases included Melanie Thompson, Melanie Smith, and Melanie Cleroux.
Ms. Cleroux was believed to be a student in a nursing school in Colorado, but she only completed 2 years of a 4-year nursing course and was never certified as a nurse, according to CBC. Her criminal record dates back 30 years and includes 67 adult convictions and other convictions in her youth, CBC reported.
A version of this article first appeared on Medscape.com.
A Canadian woman who officials allege faked being a registered nurse for some 30 years in Canada and the United States is scheduled to appear in court next month after being sentenced to 7 years in prison.
She was previously sentenced April 22 in an Ontario court after she pled guilty in January to seven offenses, including impersonation, assault with a weapon, and assault, according to CBC Radio-Canada.
Ms. Cleroux, who uses several aliases, had a long history of deception in three provinces in Canada, as well as in Colorado and Florida. The sentencing in Ontario stemmed from incidents at a medical and dental clinic in Ottawa last year, which included administration of medications to patients through needle injections, Ottawa Police reported in a press statement obtained by this news organization.
Authorities charged Ms. Cleroux in September with assault with a weapon and criminal negligence causing bodily harm, along with “personation to gain advantage,” obtaining by false pretense, and using a forged document, this news organization reported.
Ms. Cleroux has been in custody since her arrest by Ottawa Police in August.
The Vancouver Police Department (VPD) charged Ms. Cleroux last year with fraud of over $5,000 and personation with intent. VPD investigated claims that an employee at BC Women’s Hospital fraudulently identified herself as a nurse while working there between June 2020 and June 2021, according to a VPD press release.
Nursing colleges in British Columbia and Ontario issued warnings that she had used aliases and purported to be a registered nurse to gain employment. The aliases included Melanie Thompson, Melanie Smith, and Melanie Cleroux.
Ms. Cleroux was believed to be a student in a nursing school in Colorado, but she only completed 2 years of a 4-year nursing course and was never certified as a nurse, according to CBC. Her criminal record dates back 30 years and includes 67 adult convictions and other convictions in her youth, CBC reported.
A version of this article first appeared on Medscape.com.
Nap length linked to cognitive changes
No wonder we feel worse after naps
Some of us have hectic schedules that may make a nap feel more necessary. It’s common knowledge that naps shouldn’t be too long – maybe 20 minutes or so – but if you frequently take 3-hour naps and wake up thinking you’re late for school even though you’re 47 and have your PhD, this LOTME is for you.
Studies have shown that there is a link between napping during the day and Alzheimer’s/cognitive decline, but now we’ve got a double whammy for you: Longer and more frequent napping is linked to worse cognition after a year, and in turn, those with cognitive decline and Alzheimer’s are known to nap longer and more frequently during the day.
“We now know that the pathology related to cognitive decline can cause other changes in function,” he said. “It’s really a multisystem disorder, also including difficulty sleeping, changes in movement, changes in body composition, depression symptoms, behavioral changes, etc.,” coauthor Aron Buchman, MD, said in a statement from Rush University Medical Center.
The investigators monitored 1,400 patients over the course of 14 years with wrist bracelets that recorded when a person was not active during the day and considered that a nap.
At the beginning of the study, 75% of the study subjects had no cognitive impairment, 19.5% had some cognitive impairment, and approximately 4% had Alzheimer’s. Napping during the day only increased about 11 minutes a year for those with no signs of cognitive impairment, but those who showed significantly more signs of cognitive decline doubled their nap time and those actually diagnosed with Alzheimer’s tripled theirs.
The investigators did not imply that napping causes Alzheimer’s, but they noted that people who are older and nap more than an hour a day are 40% more likely to be at risk. It is something to consider and monitor.
Sometimes, after all, a nap seems like the best idea ever, but more often than not we wake up feeling 10 times worse. Our bodies may be giving us a heads up.
Pokemon Go away depression
The summer of 2016 was a great time if you happened to be a fan of Pokemon. Which is quite a lot of people. For almost 20 years millions have enjoyed the games and animated series, but Pokemon Go brought the thrill of catching Pokemon to life in a whole new way. For the first time, you could go out into the world and pretend you were a real Pokemon trainer, and everywhere you went, there would be others like you.
The ability to chase after Pikachu and Charizard in real life (well, augmented reality, but close enough) seemed to bring people a lot of joy, but seemed is never good enough for science. Can’t have anecdotes, we need data! So researchers at the London School of Economics and Political Science conducted a study into how Pokemon Go affected local Internet search rates of depression as the game was released slowly around the world.
Through analyzing Google Trend data of words like “depression,” “anxiety,” and “stress,” the researchers found that the release of Pokemon Go was significantly associated with a noticeable, though short-term, drop in depression-related Internet searches. Location-based augmented reality games may alleviate symptoms of mild depression, the researchers said, as they encourage physical activity, face-to-face socialization, and exposure to nature, though they added that simply going outside is likely not enough to combat clinical cases of severe depression.
Still, augmented reality games represent a viable target for public health investment, since they’re easy to use and inexpensive to make. That said, we’re not sure we want the FDA or CDC making a new Pokemon Go game. They’d probably end up filling the streets with Mr. Mime. And no one would leave their house for that.
And now a word from our sponsor
How many times has this happened to you? You need to repair a jet engine, inspect a nuclear reactor cooling system, AND perform bowel surgery, but you can’t carry around all the heavy, old-fashioned tools needed for those jobs.
Well, we’ve got one tool that can do it all! And that tool is a snake. No, it’s a robot.
It’s both! It’s the COntinuum roBot for Remote Applications. COBRA is the robot that looks like a snake! A snake that’s 5 meters long but only as thick as a pencil (about 9 mm in diameter). A robot with “extraordinary manoeuvrability and responsiveness due to … a compliant-joint structure and multiple continuous sections that enable it to bend at around 90 degrees,” according to the team at the University of Nottingham (England) that developed it.
COBRA comes equipped with a stereovision camera and a miniature cutting tool to perform complex industrial repair, but other devices can be interchanged for possible medical use.
COBRA and its joystick-like controller were designed to be easy to use. Dr. Oladejo Olaleye, the ear, nose, and throat and robotic surgeon at University Hospitals of Leicester who is directing its surgical development, was able to use COBRA on a dummy after just 5 minutes of training. He called it “the future of diagnostic endoscopy and therapeutic surgery.”
Don’t be the last aircraft engineer/nuclear technician/surgeon on your block to have this ultraslender, ultramaneuverable reptilian repair robot. Get your COBRA now! Operators are standing by.
Disclaimer: Robot is still under development and not yet on sale.
Rule, (worm) Britannia!
As long as there have been people, there have been parasitic worms living in their guts. Helminth infection is a continuing and largely ignored crisis in poor, tropical nations, though worm-based diseases have been basically eliminated from wealthier countries.
This wasn’t always the case, however, as a study published in PLOS Neglected Tropical Diseases (now there’s a specific topic) has found. The researchers detail the glorious history of helminth infestation in the United Kingdom from the Victorian era all the way back to prehistory, scouring hundreds of skeletons found in 17 sites across the country for eggs, which can remain intact for thousands of years.
The researchers found that two eras in particular had very high rates of infection. Unsurprisingly, the late medieval era was one of them, but the other is less obvious. The Romans were famous for their hygiene, their baths, and their plumbing, but maybe they also should be famous for the abundance of worms in their bellies. That doesn’t make sense at first: Shouldn’t good hygiene lower infection? The benefits of a good sewer system, however, are lessened when the waste containing said infectious organisms is used to fertilize crops. Recycling is generally a good thing, but less so when you’re recycling parasitic worms.
Curiously, of the three sites from the industrial age, only the one in London had high levels of worm infestation. Considering how dirty and cramped 19th-century British cities were, one might expect disease to run rampant (tuberculosis certainly did), but the sites in Oxford and Birmingham were almost devoid of worms. The researchers theorized that this was because of access to clean well water. Or maybe worms just have a thing for London. [Editor’s note: It’s probably not that.]
No wonder we feel worse after naps
Some of us have hectic schedules that may make a nap feel more necessary. It’s common knowledge that naps shouldn’t be too long – maybe 20 minutes or so – but if you frequently take 3-hour naps and wake up thinking you’re late for school even though you’re 47 and have your PhD, this LOTME is for you.
Studies have shown that there is a link between napping during the day and Alzheimer’s/cognitive decline, but now we’ve got a double whammy for you: Longer and more frequent napping is linked to worse cognition after a year, and in turn, those with cognitive decline and Alzheimer’s are known to nap longer and more frequently during the day.
“We now know that the pathology related to cognitive decline can cause other changes in function,” he said. “It’s really a multisystem disorder, also including difficulty sleeping, changes in movement, changes in body composition, depression symptoms, behavioral changes, etc.,” coauthor Aron Buchman, MD, said in a statement from Rush University Medical Center.
The investigators monitored 1,400 patients over the course of 14 years with wrist bracelets that recorded when a person was not active during the day and considered that a nap.
At the beginning of the study, 75% of the study subjects had no cognitive impairment, 19.5% had some cognitive impairment, and approximately 4% had Alzheimer’s. Napping during the day only increased about 11 minutes a year for those with no signs of cognitive impairment, but those who showed significantly more signs of cognitive decline doubled their nap time and those actually diagnosed with Alzheimer’s tripled theirs.
The investigators did not imply that napping causes Alzheimer’s, but they noted that people who are older and nap more than an hour a day are 40% more likely to be at risk. It is something to consider and monitor.
Sometimes, after all, a nap seems like the best idea ever, but more often than not we wake up feeling 10 times worse. Our bodies may be giving us a heads up.
Pokemon Go away depression
The summer of 2016 was a great time if you happened to be a fan of Pokemon. Which is quite a lot of people. For almost 20 years millions have enjoyed the games and animated series, but Pokemon Go brought the thrill of catching Pokemon to life in a whole new way. For the first time, you could go out into the world and pretend you were a real Pokemon trainer, and everywhere you went, there would be others like you.
The ability to chase after Pikachu and Charizard in real life (well, augmented reality, but close enough) seemed to bring people a lot of joy, but seemed is never good enough for science. Can’t have anecdotes, we need data! So researchers at the London School of Economics and Political Science conducted a study into how Pokemon Go affected local Internet search rates of depression as the game was released slowly around the world.
Through analyzing Google Trend data of words like “depression,” “anxiety,” and “stress,” the researchers found that the release of Pokemon Go was significantly associated with a noticeable, though short-term, drop in depression-related Internet searches. Location-based augmented reality games may alleviate symptoms of mild depression, the researchers said, as they encourage physical activity, face-to-face socialization, and exposure to nature, though they added that simply going outside is likely not enough to combat clinical cases of severe depression.
Still, augmented reality games represent a viable target for public health investment, since they’re easy to use and inexpensive to make. That said, we’re not sure we want the FDA or CDC making a new Pokemon Go game. They’d probably end up filling the streets with Mr. Mime. And no one would leave their house for that.
And now a word from our sponsor
How many times has this happened to you? You need to repair a jet engine, inspect a nuclear reactor cooling system, AND perform bowel surgery, but you can’t carry around all the heavy, old-fashioned tools needed for those jobs.
Well, we’ve got one tool that can do it all! And that tool is a snake. No, it’s a robot.
It’s both! It’s the COntinuum roBot for Remote Applications. COBRA is the robot that looks like a snake! A snake that’s 5 meters long but only as thick as a pencil (about 9 mm in diameter). A robot with “extraordinary manoeuvrability and responsiveness due to … a compliant-joint structure and multiple continuous sections that enable it to bend at around 90 degrees,” according to the team at the University of Nottingham (England) that developed it.
COBRA comes equipped with a stereovision camera and a miniature cutting tool to perform complex industrial repair, but other devices can be interchanged for possible medical use.
COBRA and its joystick-like controller were designed to be easy to use. Dr. Oladejo Olaleye, the ear, nose, and throat and robotic surgeon at University Hospitals of Leicester who is directing its surgical development, was able to use COBRA on a dummy after just 5 minutes of training. He called it “the future of diagnostic endoscopy and therapeutic surgery.”
Don’t be the last aircraft engineer/nuclear technician/surgeon on your block to have this ultraslender, ultramaneuverable reptilian repair robot. Get your COBRA now! Operators are standing by.
Disclaimer: Robot is still under development and not yet on sale.
Rule, (worm) Britannia!
As long as there have been people, there have been parasitic worms living in their guts. Helminth infection is a continuing and largely ignored crisis in poor, tropical nations, though worm-based diseases have been basically eliminated from wealthier countries.
This wasn’t always the case, however, as a study published in PLOS Neglected Tropical Diseases (now there’s a specific topic) has found. The researchers detail the glorious history of helminth infestation in the United Kingdom from the Victorian era all the way back to prehistory, scouring hundreds of skeletons found in 17 sites across the country for eggs, which can remain intact for thousands of years.
The researchers found that two eras in particular had very high rates of infection. Unsurprisingly, the late medieval era was one of them, but the other is less obvious. The Romans were famous for their hygiene, their baths, and their plumbing, but maybe they also should be famous for the abundance of worms in their bellies. That doesn’t make sense at first: Shouldn’t good hygiene lower infection? The benefits of a good sewer system, however, are lessened when the waste containing said infectious organisms is used to fertilize crops. Recycling is generally a good thing, but less so when you’re recycling parasitic worms.
Curiously, of the three sites from the industrial age, only the one in London had high levels of worm infestation. Considering how dirty and cramped 19th-century British cities were, one might expect disease to run rampant (tuberculosis certainly did), but the sites in Oxford and Birmingham were almost devoid of worms. The researchers theorized that this was because of access to clean well water. Or maybe worms just have a thing for London. [Editor’s note: It’s probably not that.]
No wonder we feel worse after naps
Some of us have hectic schedules that may make a nap feel more necessary. It’s common knowledge that naps shouldn’t be too long – maybe 20 minutes or so – but if you frequently take 3-hour naps and wake up thinking you’re late for school even though you’re 47 and have your PhD, this LOTME is for you.
Studies have shown that there is a link between napping during the day and Alzheimer’s/cognitive decline, but now we’ve got a double whammy for you: Longer and more frequent napping is linked to worse cognition after a year, and in turn, those with cognitive decline and Alzheimer’s are known to nap longer and more frequently during the day.
“We now know that the pathology related to cognitive decline can cause other changes in function,” he said. “It’s really a multisystem disorder, also including difficulty sleeping, changes in movement, changes in body composition, depression symptoms, behavioral changes, etc.,” coauthor Aron Buchman, MD, said in a statement from Rush University Medical Center.
The investigators monitored 1,400 patients over the course of 14 years with wrist bracelets that recorded when a person was not active during the day and considered that a nap.
At the beginning of the study, 75% of the study subjects had no cognitive impairment, 19.5% had some cognitive impairment, and approximately 4% had Alzheimer’s. Napping during the day only increased about 11 minutes a year for those with no signs of cognitive impairment, but those who showed significantly more signs of cognitive decline doubled their nap time and those actually diagnosed with Alzheimer’s tripled theirs.
The investigators did not imply that napping causes Alzheimer’s, but they noted that people who are older and nap more than an hour a day are 40% more likely to be at risk. It is something to consider and monitor.
Sometimes, after all, a nap seems like the best idea ever, but more often than not we wake up feeling 10 times worse. Our bodies may be giving us a heads up.
Pokemon Go away depression
The summer of 2016 was a great time if you happened to be a fan of Pokemon. Which is quite a lot of people. For almost 20 years millions have enjoyed the games and animated series, but Pokemon Go brought the thrill of catching Pokemon to life in a whole new way. For the first time, you could go out into the world and pretend you were a real Pokemon trainer, and everywhere you went, there would be others like you.
The ability to chase after Pikachu and Charizard in real life (well, augmented reality, but close enough) seemed to bring people a lot of joy, but seemed is never good enough for science. Can’t have anecdotes, we need data! So researchers at the London School of Economics and Political Science conducted a study into how Pokemon Go affected local Internet search rates of depression as the game was released slowly around the world.
Through analyzing Google Trend data of words like “depression,” “anxiety,” and “stress,” the researchers found that the release of Pokemon Go was significantly associated with a noticeable, though short-term, drop in depression-related Internet searches. Location-based augmented reality games may alleviate symptoms of mild depression, the researchers said, as they encourage physical activity, face-to-face socialization, and exposure to nature, though they added that simply going outside is likely not enough to combat clinical cases of severe depression.
Still, augmented reality games represent a viable target for public health investment, since they’re easy to use and inexpensive to make. That said, we’re not sure we want the FDA or CDC making a new Pokemon Go game. They’d probably end up filling the streets with Mr. Mime. And no one would leave their house for that.
And now a word from our sponsor
How many times has this happened to you? You need to repair a jet engine, inspect a nuclear reactor cooling system, AND perform bowel surgery, but you can’t carry around all the heavy, old-fashioned tools needed for those jobs.
Well, we’ve got one tool that can do it all! And that tool is a snake. No, it’s a robot.
It’s both! It’s the COntinuum roBot for Remote Applications. COBRA is the robot that looks like a snake! A snake that’s 5 meters long but only as thick as a pencil (about 9 mm in diameter). A robot with “extraordinary manoeuvrability and responsiveness due to … a compliant-joint structure and multiple continuous sections that enable it to bend at around 90 degrees,” according to the team at the University of Nottingham (England) that developed it.
COBRA comes equipped with a stereovision camera and a miniature cutting tool to perform complex industrial repair, but other devices can be interchanged for possible medical use.
COBRA and its joystick-like controller were designed to be easy to use. Dr. Oladejo Olaleye, the ear, nose, and throat and robotic surgeon at University Hospitals of Leicester who is directing its surgical development, was able to use COBRA on a dummy after just 5 minutes of training. He called it “the future of diagnostic endoscopy and therapeutic surgery.”
Don’t be the last aircraft engineer/nuclear technician/surgeon on your block to have this ultraslender, ultramaneuverable reptilian repair robot. Get your COBRA now! Operators are standing by.
Disclaimer: Robot is still under development and not yet on sale.
Rule, (worm) Britannia!
As long as there have been people, there have been parasitic worms living in their guts. Helminth infection is a continuing and largely ignored crisis in poor, tropical nations, though worm-based diseases have been basically eliminated from wealthier countries.
This wasn’t always the case, however, as a study published in PLOS Neglected Tropical Diseases (now there’s a specific topic) has found. The researchers detail the glorious history of helminth infestation in the United Kingdom from the Victorian era all the way back to prehistory, scouring hundreds of skeletons found in 17 sites across the country for eggs, which can remain intact for thousands of years.
The researchers found that two eras in particular had very high rates of infection. Unsurprisingly, the late medieval era was one of them, but the other is less obvious. The Romans were famous for their hygiene, their baths, and their plumbing, but maybe they also should be famous for the abundance of worms in their bellies. That doesn’t make sense at first: Shouldn’t good hygiene lower infection? The benefits of a good sewer system, however, are lessened when the waste containing said infectious organisms is used to fertilize crops. Recycling is generally a good thing, but less so when you’re recycling parasitic worms.
Curiously, of the three sites from the industrial age, only the one in London had high levels of worm infestation. Considering how dirty and cramped 19th-century British cities were, one might expect disease to run rampant (tuberculosis certainly did), but the sites in Oxford and Birmingham were almost devoid of worms. The researchers theorized that this was because of access to clean well water. Or maybe worms just have a thing for London. [Editor’s note: It’s probably not that.]
Will you have cardiac arrest? New tech may predict if and when
Deaths from COVID-19 may have caught more attention lately, but heart disease remains the leading cause of death in the United States.
More than 300,000 Americans will die this year of sudden cardiac arrest (also called sudden cardiac death, or SCD), when the heart abruptly stops working.
These events happen suddenly and often without warning, making them nearly impossible to predict. But that may be changing, thanks to 3D imaging and artificial intelligence (AI) technology under study at Johns Hopkins University, Baltimore.
There, researchers are working to create more accurate and personalized models of the heart – and not just any heart, your heart, if you have heart disease.
“Right now, a clinician can only say whether a patient is at risk or not at risk for sudden death,” says Dan Popescu, PhD, a Johns Hopkins research scientist and first author of a new study on AI’s ability to predict sudden cardiac arrest. “With this new technology, you can have much more nuanced predictions of probability of an event over time.”
Put another way: With AI, clinicians may be able not only to predict if someone is at risk for sudden cardiac arrest, but also when it is most likely to happen. They can do this using a much clearer and more personalized look at the electrical “wiring” of your heart.
Your heart, the conductor
Your heart isn’t just a metronome responsible for keeping a steady stream of blood pumping to tissues with every beat. It’s also a conductor through which vital energy flows.
To make the heart beat, electrical impulses flow from the top to the bottom of the organ. Healthy heart cells relay this electricity seamlessly. But in a heart damaged by inflammation or a past heart attack, scar tissue will block the energy flow.
When an electrical impulse encounters a scarred area, the signal can become erratic, disrupting the set top-to-bottom path and causing irregular heartbeats (arrhythmias), which increase someone’s danger of sudden cardiac death.
Seeing the heart in 3D
Today’s tests offer some insights into the heart’s makeup. For example, MRI scans can reveal damaged areas. PET scans can show inflammation. And EKGs can record the heart’s electrical signals from beat to beat.
But all these technologies offer only a snapshot, showing heart health at a moment in time. They can’t predict the future. That’s why scientists at Johns Hopkins are going further to develop 3D digital replicas of a person’s heart, known as computational heart models.
Computational models are computer-simulated replicas that combine mathematics, physics, and computer science. These models have been around for a long time and are used in many fields, ranging from manufacturing to economics.
In heart medicine, these models are populated with digital “cells,” which imitate living cells and can be programmed with different electrical properties, depending on whether they are healthy or diseased.
“Currently available imaging and testing (MRIs, PETs, EKGs) give some representation of the scarring, but you cannot translate that to what is going to happen over time,” says Natalia Trayanova, PhD, of the Johns Hopkins department of biomedical engineering.
“With computational heart models, we create a dynamic digital image of the heart. We can then give the digital image an electrical stimulus and assess how the heart is able to respond. Then you can better predict what is going to happen.”
The computerized 3D models also mean better, more accurate treatment for heart conditions.
For example, a common treatment for a type of arrhythmia known as atrial fibrillation is ablation, or burning some heart tissue. Ablation stops the erratic electrical impulses causing the arrhythmia, but it can also damage otherwise healthy heart cells.
A personalized computational heart model could allow doctors to see more accurately what areas should and shouldn’t be treated for a specific patient.
Using deep learning AI to predict health outcomes
Dr. Trayanova’s colleague Dr. Popescu is applying deep learning and AI to do more with computerized heart models to predict the future.
In a recent paper in Nature Cardiovascular Research, the research team showed their algorithm assessed the health of 269 patients and was able to predict the chance of sudden cardiac arrest up to 10 years in advance.
“This is really the first time ever, as far as we know, where deep learning technology has been proven to analyze scarring of the heart in a successful way,” Dr. Popescu says.
Dr. Popescu and Dr. Trayanova say the AI algorithm gathers information from the 3D computational heart models with patient data like MRIs, ethnicity, age, lifestyle, and other clinical information. Analyzing all these data can produce accurate and consistent estimates about how long patients might live if they are at risk for sudden death.
“You can’t afford to be wrong. If you are wrong, you can actually impact a patient’s quality of life dramatically,” Dr. Popescu says. “Having clinicians use this technology in the decision-making process will provide confidence in a better diagnosis and prognosis.”
While the current study was specifically about patients with a particular type of heart disease, Dr. Popescu says his algorithm can also be trained to assess other health conditions.
So when might you see this being used outside of a research study? Dr. Trayanova predicts 3D imaging of heart models could be available in 2 years, but first the technique must be tested in more clinical trials – some of which are happening right now.
Adding AI to the heart models will require more studies and Food and Drug Administration approval, so the timeline is less clear. But perhaps the biggest hurdle is that after approval the technologies would need to be adopted and used by clinicians and caregivers.
“The much harder question to answer is, ‘When will doctors be perfectly comfortable with AI tools?’ And I don’t know the answer,” Dr. Popescu says. “How to use AI as an aid in the decision-making process is something that’s not currently taught.”
A version of this article first appeared on WebMD.com.
Deaths from COVID-19 may have caught more attention lately, but heart disease remains the leading cause of death in the United States.
More than 300,000 Americans will die this year of sudden cardiac arrest (also called sudden cardiac death, or SCD), when the heart abruptly stops working.
These events happen suddenly and often without warning, making them nearly impossible to predict. But that may be changing, thanks to 3D imaging and artificial intelligence (AI) technology under study at Johns Hopkins University, Baltimore.
There, researchers are working to create more accurate and personalized models of the heart – and not just any heart, your heart, if you have heart disease.
“Right now, a clinician can only say whether a patient is at risk or not at risk for sudden death,” says Dan Popescu, PhD, a Johns Hopkins research scientist and first author of a new study on AI’s ability to predict sudden cardiac arrest. “With this new technology, you can have much more nuanced predictions of probability of an event over time.”
Put another way: With AI, clinicians may be able not only to predict if someone is at risk for sudden cardiac arrest, but also when it is most likely to happen. They can do this using a much clearer and more personalized look at the electrical “wiring” of your heart.
Your heart, the conductor
Your heart isn’t just a metronome responsible for keeping a steady stream of blood pumping to tissues with every beat. It’s also a conductor through which vital energy flows.
To make the heart beat, electrical impulses flow from the top to the bottom of the organ. Healthy heart cells relay this electricity seamlessly. But in a heart damaged by inflammation or a past heart attack, scar tissue will block the energy flow.
When an electrical impulse encounters a scarred area, the signal can become erratic, disrupting the set top-to-bottom path and causing irregular heartbeats (arrhythmias), which increase someone’s danger of sudden cardiac death.
Seeing the heart in 3D
Today’s tests offer some insights into the heart’s makeup. For example, MRI scans can reveal damaged areas. PET scans can show inflammation. And EKGs can record the heart’s electrical signals from beat to beat.
But all these technologies offer only a snapshot, showing heart health at a moment in time. They can’t predict the future. That’s why scientists at Johns Hopkins are going further to develop 3D digital replicas of a person’s heart, known as computational heart models.
Computational models are computer-simulated replicas that combine mathematics, physics, and computer science. These models have been around for a long time and are used in many fields, ranging from manufacturing to economics.
In heart medicine, these models are populated with digital “cells,” which imitate living cells and can be programmed with different electrical properties, depending on whether they are healthy or diseased.
“Currently available imaging and testing (MRIs, PETs, EKGs) give some representation of the scarring, but you cannot translate that to what is going to happen over time,” says Natalia Trayanova, PhD, of the Johns Hopkins department of biomedical engineering.
“With computational heart models, we create a dynamic digital image of the heart. We can then give the digital image an electrical stimulus and assess how the heart is able to respond. Then you can better predict what is going to happen.”
The computerized 3D models also mean better, more accurate treatment for heart conditions.
For example, a common treatment for a type of arrhythmia known as atrial fibrillation is ablation, or burning some heart tissue. Ablation stops the erratic electrical impulses causing the arrhythmia, but it can also damage otherwise healthy heart cells.
A personalized computational heart model could allow doctors to see more accurately what areas should and shouldn’t be treated for a specific patient.
Using deep learning AI to predict health outcomes
Dr. Trayanova’s colleague Dr. Popescu is applying deep learning and AI to do more with computerized heart models to predict the future.
In a recent paper in Nature Cardiovascular Research, the research team showed their algorithm assessed the health of 269 patients and was able to predict the chance of sudden cardiac arrest up to 10 years in advance.
“This is really the first time ever, as far as we know, where deep learning technology has been proven to analyze scarring of the heart in a successful way,” Dr. Popescu says.
Dr. Popescu and Dr. Trayanova say the AI algorithm gathers information from the 3D computational heart models with patient data like MRIs, ethnicity, age, lifestyle, and other clinical information. Analyzing all these data can produce accurate and consistent estimates about how long patients might live if they are at risk for sudden death.
“You can’t afford to be wrong. If you are wrong, you can actually impact a patient’s quality of life dramatically,” Dr. Popescu says. “Having clinicians use this technology in the decision-making process will provide confidence in a better diagnosis and prognosis.”
While the current study was specifically about patients with a particular type of heart disease, Dr. Popescu says his algorithm can also be trained to assess other health conditions.
So when might you see this being used outside of a research study? Dr. Trayanova predicts 3D imaging of heart models could be available in 2 years, but first the technique must be tested in more clinical trials – some of which are happening right now.
Adding AI to the heart models will require more studies and Food and Drug Administration approval, so the timeline is less clear. But perhaps the biggest hurdle is that after approval the technologies would need to be adopted and used by clinicians and caregivers.
“The much harder question to answer is, ‘When will doctors be perfectly comfortable with AI tools?’ And I don’t know the answer,” Dr. Popescu says. “How to use AI as an aid in the decision-making process is something that’s not currently taught.”
A version of this article first appeared on WebMD.com.
Deaths from COVID-19 may have caught more attention lately, but heart disease remains the leading cause of death in the United States.
More than 300,000 Americans will die this year of sudden cardiac arrest (also called sudden cardiac death, or SCD), when the heart abruptly stops working.
These events happen suddenly and often without warning, making them nearly impossible to predict. But that may be changing, thanks to 3D imaging and artificial intelligence (AI) technology under study at Johns Hopkins University, Baltimore.
There, researchers are working to create more accurate and personalized models of the heart – and not just any heart, your heart, if you have heart disease.
“Right now, a clinician can only say whether a patient is at risk or not at risk for sudden death,” says Dan Popescu, PhD, a Johns Hopkins research scientist and first author of a new study on AI’s ability to predict sudden cardiac arrest. “With this new technology, you can have much more nuanced predictions of probability of an event over time.”
Put another way: With AI, clinicians may be able not only to predict if someone is at risk for sudden cardiac arrest, but also when it is most likely to happen. They can do this using a much clearer and more personalized look at the electrical “wiring” of your heart.
Your heart, the conductor
Your heart isn’t just a metronome responsible for keeping a steady stream of blood pumping to tissues with every beat. It’s also a conductor through which vital energy flows.
To make the heart beat, electrical impulses flow from the top to the bottom of the organ. Healthy heart cells relay this electricity seamlessly. But in a heart damaged by inflammation or a past heart attack, scar tissue will block the energy flow.
When an electrical impulse encounters a scarred area, the signal can become erratic, disrupting the set top-to-bottom path and causing irregular heartbeats (arrhythmias), which increase someone’s danger of sudden cardiac death.
Seeing the heart in 3D
Today’s tests offer some insights into the heart’s makeup. For example, MRI scans can reveal damaged areas. PET scans can show inflammation. And EKGs can record the heart’s electrical signals from beat to beat.
But all these technologies offer only a snapshot, showing heart health at a moment in time. They can’t predict the future. That’s why scientists at Johns Hopkins are going further to develop 3D digital replicas of a person’s heart, known as computational heart models.
Computational models are computer-simulated replicas that combine mathematics, physics, and computer science. These models have been around for a long time and are used in many fields, ranging from manufacturing to economics.
In heart medicine, these models are populated with digital “cells,” which imitate living cells and can be programmed with different electrical properties, depending on whether they are healthy or diseased.
“Currently available imaging and testing (MRIs, PETs, EKGs) give some representation of the scarring, but you cannot translate that to what is going to happen over time,” says Natalia Trayanova, PhD, of the Johns Hopkins department of biomedical engineering.
“With computational heart models, we create a dynamic digital image of the heart. We can then give the digital image an electrical stimulus and assess how the heart is able to respond. Then you can better predict what is going to happen.”
The computerized 3D models also mean better, more accurate treatment for heart conditions.
For example, a common treatment for a type of arrhythmia known as atrial fibrillation is ablation, or burning some heart tissue. Ablation stops the erratic electrical impulses causing the arrhythmia, but it can also damage otherwise healthy heart cells.
A personalized computational heart model could allow doctors to see more accurately what areas should and shouldn’t be treated for a specific patient.
Using deep learning AI to predict health outcomes
Dr. Trayanova’s colleague Dr. Popescu is applying deep learning and AI to do more with computerized heart models to predict the future.
In a recent paper in Nature Cardiovascular Research, the research team showed their algorithm assessed the health of 269 patients and was able to predict the chance of sudden cardiac arrest up to 10 years in advance.
“This is really the first time ever, as far as we know, where deep learning technology has been proven to analyze scarring of the heart in a successful way,” Dr. Popescu says.
Dr. Popescu and Dr. Trayanova say the AI algorithm gathers information from the 3D computational heart models with patient data like MRIs, ethnicity, age, lifestyle, and other clinical information. Analyzing all these data can produce accurate and consistent estimates about how long patients might live if they are at risk for sudden death.
“You can’t afford to be wrong. If you are wrong, you can actually impact a patient’s quality of life dramatically,” Dr. Popescu says. “Having clinicians use this technology in the decision-making process will provide confidence in a better diagnosis and prognosis.”
While the current study was specifically about patients with a particular type of heart disease, Dr. Popescu says his algorithm can also be trained to assess other health conditions.
So when might you see this being used outside of a research study? Dr. Trayanova predicts 3D imaging of heart models could be available in 2 years, but first the technique must be tested in more clinical trials – some of which are happening right now.
Adding AI to the heart models will require more studies and Food and Drug Administration approval, so the timeline is less clear. But perhaps the biggest hurdle is that after approval the technologies would need to be adopted and used by clinicians and caregivers.
“The much harder question to answer is, ‘When will doctors be perfectly comfortable with AI tools?’ And I don’t know the answer,” Dr. Popescu says. “How to use AI as an aid in the decision-making process is something that’s not currently taught.”
A version of this article first appeared on WebMD.com.
Pfizer recalls more quinapril because of potential carcinogen
, the company announced.
The Accupril recall comes one month after Pfizer recalled six lots of Accuretic (Quinapril HCI/hydrochlorathiazide) tablets for the same problem.
Accupril is indicated for the treatment of hypertension and management of heart failure when added to conventional therapy, including diuretics and/or digitalis.
To date, Pfizer is not aware of any reports of adverse events related to the Accupril recall, and the company believes the benefit/risk profile remains positive based on currently available data.
“Although long-term ingestion of N-nitroso-quinapril may be associated with a potential increased cancer risk in humans, there is no immediate risk to patients taking this medication,” the company said April 22 in a news release.
Patients currently taking the recalled products are asked to consult with their doctor about alternative treatment options.
The recalled Accupril tablets were sold in 90-count bottles distributed nationwide to wholesalers and distributors in the United States and Puerto Rico from December 2019 to April 2022.
National drug codes (NDC), lot numbers, and expiration dates are listed in the company announcement posted on the Food and Drug Administration’s website.
Patients who are taking this product should consult with their health care provider or pharmacy to determine if they have the affected product. Those with the affected tablets should contact claims management firm Sedgwick by phone at 888-345-0481 Monday through Friday from 8 AM to 5 PM ET for instructions on how to return the product and obtain reimbursement.
Healthcare providers with questions regarding the recall can contact Pfizer by telephone at 800-438-1985, option 3, Monday through Friday from 8 AM to 9 PM ET.
Adverse reactions or quality problems related to this recall should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
, the company announced.
The Accupril recall comes one month after Pfizer recalled six lots of Accuretic (Quinapril HCI/hydrochlorathiazide) tablets for the same problem.
Accupril is indicated for the treatment of hypertension and management of heart failure when added to conventional therapy, including diuretics and/or digitalis.
To date, Pfizer is not aware of any reports of adverse events related to the Accupril recall, and the company believes the benefit/risk profile remains positive based on currently available data.
“Although long-term ingestion of N-nitroso-quinapril may be associated with a potential increased cancer risk in humans, there is no immediate risk to patients taking this medication,” the company said April 22 in a news release.
Patients currently taking the recalled products are asked to consult with their doctor about alternative treatment options.
The recalled Accupril tablets were sold in 90-count bottles distributed nationwide to wholesalers and distributors in the United States and Puerto Rico from December 2019 to April 2022.
National drug codes (NDC), lot numbers, and expiration dates are listed in the company announcement posted on the Food and Drug Administration’s website.
Patients who are taking this product should consult with their health care provider or pharmacy to determine if they have the affected product. Those with the affected tablets should contact claims management firm Sedgwick by phone at 888-345-0481 Monday through Friday from 8 AM to 5 PM ET for instructions on how to return the product and obtain reimbursement.
Healthcare providers with questions regarding the recall can contact Pfizer by telephone at 800-438-1985, option 3, Monday through Friday from 8 AM to 9 PM ET.
Adverse reactions or quality problems related to this recall should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
, the company announced.
The Accupril recall comes one month after Pfizer recalled six lots of Accuretic (Quinapril HCI/hydrochlorathiazide) tablets for the same problem.
Accupril is indicated for the treatment of hypertension and management of heart failure when added to conventional therapy, including diuretics and/or digitalis.
To date, Pfizer is not aware of any reports of adverse events related to the Accupril recall, and the company believes the benefit/risk profile remains positive based on currently available data.
“Although long-term ingestion of N-nitroso-quinapril may be associated with a potential increased cancer risk in humans, there is no immediate risk to patients taking this medication,” the company said April 22 in a news release.
Patients currently taking the recalled products are asked to consult with their doctor about alternative treatment options.
The recalled Accupril tablets were sold in 90-count bottles distributed nationwide to wholesalers and distributors in the United States and Puerto Rico from December 2019 to April 2022.
National drug codes (NDC), lot numbers, and expiration dates are listed in the company announcement posted on the Food and Drug Administration’s website.
Patients who are taking this product should consult with their health care provider or pharmacy to determine if they have the affected product. Those with the affected tablets should contact claims management firm Sedgwick by phone at 888-345-0481 Monday through Friday from 8 AM to 5 PM ET for instructions on how to return the product and obtain reimbursement.
Healthcare providers with questions regarding the recall can contact Pfizer by telephone at 800-438-1985, option 3, Monday through Friday from 8 AM to 9 PM ET.
Adverse reactions or quality problems related to this recall should be reported to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.