User login
Hospital Dermatology: Review of Research in 2024-2025
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
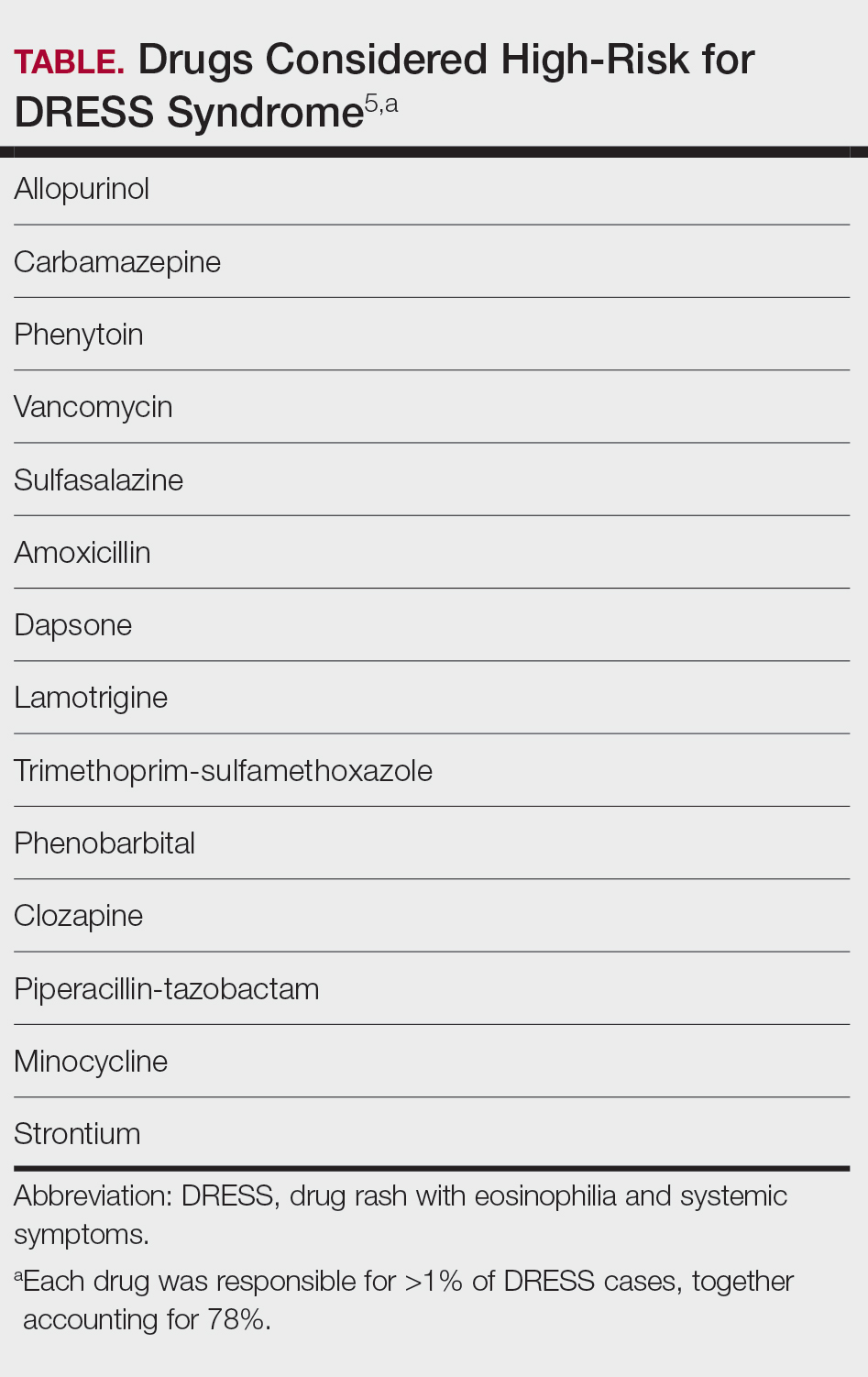
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6
A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6

A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6

A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
Hospital Dermatology: Review of Research in 2024-2025
Hospital Dermatology: Review of Research in 2024-2025
Practice Points
- In suspected drug reaction with eosinophilia and systemic symptoms, discontinue the offending drug; test for human herpesvirus 6, Epstein-Barr virus, and cytomegalovirus when available; and treat moderate cases with low-dose corticosteroids. Reserve interleukin 5 inhibitors for refractory disease.
- For Stevens-Johnson syndrome and toxic epidermal necrolysis (TEN), apply Niigata diagnostic criteria and clinical risk score for TEN, refer patients with 10% or more body surface area detachment to higher-level or burn care, and consider targeted therapies for refractory cases.
- When assessing infectious rashes, consider measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Review the patient’s vaccination and travel history, isolate suspected measles cases, and confirm atypical tinea with culture or DNA testing.
- To reduce unnecessary admissions and optimize care for hidradenitis suppurativa, avoid misdiagnosing flares as sepsis, implement multidisciplinary protocols, consider selective intravenous antibiotics, and support expanded inpatient dermatology coverage.