User login
Clinical Psychiatry News is the online destination and multimedia properties of Clinica Psychiatry News, the independent news publication for psychiatrists. Since 1971, Clinical Psychiatry News has been the leading source of news and commentary about clinical developments in psychiatry as well as health care policy and regulations that affect the physician's practice.
Dear Drupal User: You're seeing this because you're logged in to Drupal, and not redirected to MDedge.com/psychiatry.
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
ketamine
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
suicide
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-cpn')]
div[contains(@class, 'pane-pub-home-cpn')]
div[contains(@class, 'pane-pub-topic-cpn')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Seeking help for burnout may be a gamble for doctors
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.
Pandemic tied to significant drop in residents’ PTSD rates
TOPLINE
First-year medical residents training during COVID-19 were significantly less likely to have posttraumatic stress disorder and workplace trauma, compared with their counterparts who trained before the pandemic, and reported fewer work hours, higher workload satisfaction, and fewer medical errors, new research shows.
METHODOLOGY
- Studies have reported a high prevalence of PTSD symptoms among residents during the pandemic, but it’s unclear if this prevalence differs from prepandemic levels.
- Using the Intern Health Study, a longitudinal cohort study of 1st-year residents, researchers investigated differences in PTSD symptoms among those training before the pandemic (2018-2019) and during its first wave (March to June, 2020).
- The study included 1,957 first-year residents (48.2% female; mean age, 27.6 years) who completed a baseline survey 2 months before their residency start, and then quarterly surveys during their intern year, with the fourth quarterly survey including a screen for PTSD.
- Researchers assessed differences in nonresidency factors and residency-related factors before and during the pandemic and examined exposure to workplace trauma.
TAKEAWAY
- (7.1% vs. 10.7%; odds ratio, 0.64; 95% confidence interval, 0.46-0.88; P = .01).
- They were also less likely to have workplace trauma exposure (50.9% vs. 56.6%; OR, 0.80; 95% CI, 0.66-0.95; P = .01).
- Residents training during the pandemic compared to prepandemic reported significantly lower weekly duty hours (score mean difference –3.1 hours; 95% CI, –4.1 to −2.0 hours), lower mean reports of medical errors (MD, −0.04; 95% CI, –0.06 to –0.01), and higher workload satisfaction (MD, 0.2; 95% CI, 0.2-0.3).
- However, after accounting for these residency-related factors, training during the pandemic was no longer associated with lower odds of presenting PTSD symptoms.
IN PRACTICE
While the findings show residents training during the first pandemic wave were less likely to have PTSD, future studies should further follow these residents’ PTSD symptoms and investigate whether interventions targeting residency-related factors could reduce their PTSD risk moving forward, the investigators note.
SOURCE
The study was carried out by Michelle K. Ptak, BA, department of psychology, University of Michigan, Ann Arbor, and colleagues. It was published online Aug. 22 in JAMA Network Open.
LIMITATIONS
The study used self-reports and included only the first pandemic wave, 1st-year residents, and prepandemic data for a single academic year. Survey participation decreased during the pandemic, and it’s possible there were unmeasured factors associated with PTSD risk.
DISCLOSURES
The study was supported by the National Institute of Mental Health and the National Institutes of Health. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE
First-year medical residents training during COVID-19 were significantly less likely to have posttraumatic stress disorder and workplace trauma, compared with their counterparts who trained before the pandemic, and reported fewer work hours, higher workload satisfaction, and fewer medical errors, new research shows.
METHODOLOGY
- Studies have reported a high prevalence of PTSD symptoms among residents during the pandemic, but it’s unclear if this prevalence differs from prepandemic levels.
- Using the Intern Health Study, a longitudinal cohort study of 1st-year residents, researchers investigated differences in PTSD symptoms among those training before the pandemic (2018-2019) and during its first wave (March to June, 2020).
- The study included 1,957 first-year residents (48.2% female; mean age, 27.6 years) who completed a baseline survey 2 months before their residency start, and then quarterly surveys during their intern year, with the fourth quarterly survey including a screen for PTSD.
- Researchers assessed differences in nonresidency factors and residency-related factors before and during the pandemic and examined exposure to workplace trauma.
TAKEAWAY
- (7.1% vs. 10.7%; odds ratio, 0.64; 95% confidence interval, 0.46-0.88; P = .01).
- They were also less likely to have workplace trauma exposure (50.9% vs. 56.6%; OR, 0.80; 95% CI, 0.66-0.95; P = .01).
- Residents training during the pandemic compared to prepandemic reported significantly lower weekly duty hours (score mean difference –3.1 hours; 95% CI, –4.1 to −2.0 hours), lower mean reports of medical errors (MD, −0.04; 95% CI, –0.06 to –0.01), and higher workload satisfaction (MD, 0.2; 95% CI, 0.2-0.3).
- However, after accounting for these residency-related factors, training during the pandemic was no longer associated with lower odds of presenting PTSD symptoms.
IN PRACTICE
While the findings show residents training during the first pandemic wave were less likely to have PTSD, future studies should further follow these residents’ PTSD symptoms and investigate whether interventions targeting residency-related factors could reduce their PTSD risk moving forward, the investigators note.
SOURCE
The study was carried out by Michelle K. Ptak, BA, department of psychology, University of Michigan, Ann Arbor, and colleagues. It was published online Aug. 22 in JAMA Network Open.
LIMITATIONS
The study used self-reports and included only the first pandemic wave, 1st-year residents, and prepandemic data for a single academic year. Survey participation decreased during the pandemic, and it’s possible there were unmeasured factors associated with PTSD risk.
DISCLOSURES
The study was supported by the National Institute of Mental Health and the National Institutes of Health. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE
First-year medical residents training during COVID-19 were significantly less likely to have posttraumatic stress disorder and workplace trauma, compared with their counterparts who trained before the pandemic, and reported fewer work hours, higher workload satisfaction, and fewer medical errors, new research shows.
METHODOLOGY
- Studies have reported a high prevalence of PTSD symptoms among residents during the pandemic, but it’s unclear if this prevalence differs from prepandemic levels.
- Using the Intern Health Study, a longitudinal cohort study of 1st-year residents, researchers investigated differences in PTSD symptoms among those training before the pandemic (2018-2019) and during its first wave (March to June, 2020).
- The study included 1,957 first-year residents (48.2% female; mean age, 27.6 years) who completed a baseline survey 2 months before their residency start, and then quarterly surveys during their intern year, with the fourth quarterly survey including a screen for PTSD.
- Researchers assessed differences in nonresidency factors and residency-related factors before and during the pandemic and examined exposure to workplace trauma.
TAKEAWAY
- (7.1% vs. 10.7%; odds ratio, 0.64; 95% confidence interval, 0.46-0.88; P = .01).
- They were also less likely to have workplace trauma exposure (50.9% vs. 56.6%; OR, 0.80; 95% CI, 0.66-0.95; P = .01).
- Residents training during the pandemic compared to prepandemic reported significantly lower weekly duty hours (score mean difference –3.1 hours; 95% CI, –4.1 to −2.0 hours), lower mean reports of medical errors (MD, −0.04; 95% CI, –0.06 to –0.01), and higher workload satisfaction (MD, 0.2; 95% CI, 0.2-0.3).
- However, after accounting for these residency-related factors, training during the pandemic was no longer associated with lower odds of presenting PTSD symptoms.
IN PRACTICE
While the findings show residents training during the first pandemic wave were less likely to have PTSD, future studies should further follow these residents’ PTSD symptoms and investigate whether interventions targeting residency-related factors could reduce their PTSD risk moving forward, the investigators note.
SOURCE
The study was carried out by Michelle K. Ptak, BA, department of psychology, University of Michigan, Ann Arbor, and colleagues. It was published online Aug. 22 in JAMA Network Open.
LIMITATIONS
The study used self-reports and included only the first pandemic wave, 1st-year residents, and prepandemic data for a single academic year. Survey participation decreased during the pandemic, and it’s possible there were unmeasured factors associated with PTSD risk.
DISCLOSURES
The study was supported by the National Institute of Mental Health and the National Institutes of Health. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Nurses maintain more stigma toward pregnant women with OUD
Opioid use disorder among pregnant women continues to rise, and untreated opioid use is associated with complications including preterm delivery, placental abruption, and stillbirth, wrote Alexis Braverman, MD, of the University of Illinois, Chicago, and colleagues. However, many perinatal women who seek care and medications for opioid use disorder (OUD) report stigma that limits their ability to reduce these risks.
In a study published in the American Journal on Addictions , the researchers conducted an anonymous survey of 132 health care workers at six outpatient locations and a main hospital of an urban medical center. The survey was designed to assess attitudes toward pregnant women who were using opioids. The 119 complete responses in the final analysis included 40 nurses and 79 clinicians across ob.gyn., family medicine, and pediatrics. A total of 19 respondents were waivered to prescribe outpatient buprenorphine for OUD.
Nurses were significantly less likely than clinicians to agree that OUD is a chronic illness, to feel sympathy for women who use opioids during pregnancy, and to see pregnancy as an opportunity for behavior change (P = .000, P = .003, and P = .001, respectively).
Overall, family medicine providers and clinicians with 11-20 years of practice experience were significantly more sympathetic to pregnant women who used opioids, compared with providers from other departments and with fewer years of practice (P = .025 and P = .039, respectively).
Providers in pediatrics departments were significantly more likely than those from other departments to agree strongly with feeling anger at pregnant women who use opioids (P = .009), and that these women should not be allowed to parent (P = .013). However, providers in pediatrics were significantly more comfortable than those in other departments with discussing the involvement of social services in patient care (P = .020) and with counseling patients on neonatal opioid withdrawal syndrome, known as NOWS (P = .027).
“We hypothesize that nurses who perform more acute, inpatient work rather than outpatient work may not be exposed as frequently to a patient’s personal progress on their journey with OUD,” and therefore might not be exposed to the rewarding experiences and progress made by patients, the researchers wrote in their discussion.
However, the overall low level of comfort in discussing NOWS and social service involvement across provider groups (one-quarter for pediatrics, one-fifth for ob.gyn, and one-sixth for family medicine) highlights the need for further training in this area, they said.
The findings were limited by several factors, including the potential for responder bias; however, the results identify a need for greater training in stigma reduction and in counseling families on issues related to OUD, the researchers said. More studies are needed to examine attitude changes after the implementation of stigma reduction strategies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Opioid use disorder among pregnant women continues to rise, and untreated opioid use is associated with complications including preterm delivery, placental abruption, and stillbirth, wrote Alexis Braverman, MD, of the University of Illinois, Chicago, and colleagues. However, many perinatal women who seek care and medications for opioid use disorder (OUD) report stigma that limits their ability to reduce these risks.
In a study published in the American Journal on Addictions , the researchers conducted an anonymous survey of 132 health care workers at six outpatient locations and a main hospital of an urban medical center. The survey was designed to assess attitudes toward pregnant women who were using opioids. The 119 complete responses in the final analysis included 40 nurses and 79 clinicians across ob.gyn., family medicine, and pediatrics. A total of 19 respondents were waivered to prescribe outpatient buprenorphine for OUD.
Nurses were significantly less likely than clinicians to agree that OUD is a chronic illness, to feel sympathy for women who use opioids during pregnancy, and to see pregnancy as an opportunity for behavior change (P = .000, P = .003, and P = .001, respectively).
Overall, family medicine providers and clinicians with 11-20 years of practice experience were significantly more sympathetic to pregnant women who used opioids, compared with providers from other departments and with fewer years of practice (P = .025 and P = .039, respectively).
Providers in pediatrics departments were significantly more likely than those from other departments to agree strongly with feeling anger at pregnant women who use opioids (P = .009), and that these women should not be allowed to parent (P = .013). However, providers in pediatrics were significantly more comfortable than those in other departments with discussing the involvement of social services in patient care (P = .020) and with counseling patients on neonatal opioid withdrawal syndrome, known as NOWS (P = .027).
“We hypothesize that nurses who perform more acute, inpatient work rather than outpatient work may not be exposed as frequently to a patient’s personal progress on their journey with OUD,” and therefore might not be exposed to the rewarding experiences and progress made by patients, the researchers wrote in their discussion.
However, the overall low level of comfort in discussing NOWS and social service involvement across provider groups (one-quarter for pediatrics, one-fifth for ob.gyn, and one-sixth for family medicine) highlights the need for further training in this area, they said.
The findings were limited by several factors, including the potential for responder bias; however, the results identify a need for greater training in stigma reduction and in counseling families on issues related to OUD, the researchers said. More studies are needed to examine attitude changes after the implementation of stigma reduction strategies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Opioid use disorder among pregnant women continues to rise, and untreated opioid use is associated with complications including preterm delivery, placental abruption, and stillbirth, wrote Alexis Braverman, MD, of the University of Illinois, Chicago, and colleagues. However, many perinatal women who seek care and medications for opioid use disorder (OUD) report stigma that limits their ability to reduce these risks.
In a study published in the American Journal on Addictions , the researchers conducted an anonymous survey of 132 health care workers at six outpatient locations and a main hospital of an urban medical center. The survey was designed to assess attitudes toward pregnant women who were using opioids. The 119 complete responses in the final analysis included 40 nurses and 79 clinicians across ob.gyn., family medicine, and pediatrics. A total of 19 respondents were waivered to prescribe outpatient buprenorphine for OUD.
Nurses were significantly less likely than clinicians to agree that OUD is a chronic illness, to feel sympathy for women who use opioids during pregnancy, and to see pregnancy as an opportunity for behavior change (P = .000, P = .003, and P = .001, respectively).
Overall, family medicine providers and clinicians with 11-20 years of practice experience were significantly more sympathetic to pregnant women who used opioids, compared with providers from other departments and with fewer years of practice (P = .025 and P = .039, respectively).
Providers in pediatrics departments were significantly more likely than those from other departments to agree strongly with feeling anger at pregnant women who use opioids (P = .009), and that these women should not be allowed to parent (P = .013). However, providers in pediatrics were significantly more comfortable than those in other departments with discussing the involvement of social services in patient care (P = .020) and with counseling patients on neonatal opioid withdrawal syndrome, known as NOWS (P = .027).
“We hypothesize that nurses who perform more acute, inpatient work rather than outpatient work may not be exposed as frequently to a patient’s personal progress on their journey with OUD,” and therefore might not be exposed to the rewarding experiences and progress made by patients, the researchers wrote in their discussion.
However, the overall low level of comfort in discussing NOWS and social service involvement across provider groups (one-quarter for pediatrics, one-fifth for ob.gyn, and one-sixth for family medicine) highlights the need for further training in this area, they said.
The findings were limited by several factors, including the potential for responder bias; however, the results identify a need for greater training in stigma reduction and in counseling families on issues related to OUD, the researchers said. More studies are needed to examine attitude changes after the implementation of stigma reduction strategies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM THE AMERICAN JOURNAL ON ADDICTIONS
Barbie has an anxiety disorder
And it’s a great time to be a therapist
The Barbie movie is generating a lot of feelings, ranging from praise to vitriol. However one feels about the movie, let’s all pause and reflect for a moment on the fact that the number-one grossing film of 2023 is about our childhood doll trying to treat her anxiety disorder.
“Life imitates art more than art imitates life.” So said Oscar Wilde in 1889.
When my adult daughter, a childhood Barbie enthusiast, asked me to see the film, we put on pink and went. Twice. Little did I know that it would stir up so many thoughts and feelings. The one I want to share is how blessed I feel at this moment in time to be a mental health care provider! No longer is mental health something to be whispered about at the water cooler; instead, even Barbie is suffering. And with all the controversy in the press about the movie, no one seems at all surprised by this storyline.
I was raised by two child psychiatrists and have been practicing as an adult psychiatrist since 1991. The start of the pandemic was the most difficult time of my career, as almost every patient was struggling simultaneously, as was I. Three long years later, we are gradually emerging from our shared trauma. How ironic, now with the opportunity to go back to work, I have elected to maintain the majority of my practice online from home. It seems that most patients and providers prefer this mode of treatment, with a full 90 percent of practitioners saying they are using a hybrid model.
As mental health professionals, we know that anywhere from 3% to 49% of those experiencing trauma will develop posttraumatic stress disorder (PTSD), and we have been trained to treat them.
But what happens when an entire global population is exposed simultaneously to trauma? Historians and social scientists refer to such events by many different names, such as: Singularity, Black Swan Event, and Tipping Point. These events are incredibly rare, and afterwards everything is different. These global traumas always lead to massive change.
I think we are at that tipping point. This is the singularity. This is our Black Swan Event. Within a 3-year span, we have experienced the following:
- A global traumatic event (COVID-19).
- A sudden and seemingly permanent shift from office to remote video meetings mostly from home.
- Upending of traditional fundamentals of the stock market as the game literally stopped in January 2021.
- Rapid and widespread availability of Artificial Intelligence.
- The first generation to be fully raised on the Internet and social media (Gen Z) is now entering the workforce.
- Ongoing war in Ukraine.
That’s already an overwhelming list, and I could go on, but let’s get back to Barbie’s anxiety disorder.
The awareness about and acceptance of mental health issues has never been higher. The access to treatment never greater. There are now more online therapy options than ever. Treatment options have dramatically expanded in recent years, from Transcranial Magnetic Stimulation (TMS) to ketamine centers and psychedelics, as well as more mainstream options such as dialectical behavior therapy (DBT), cognitive behavioral therapy (CBT), selective serotonin reuptake inhibitors (SSRIs), and so many more.
What is particularly unique about this moment is the direct access to care. Self-help books abound with many making it to the New York Times bestseller list. YouTube is loaded with fantastic content on overcoming many mental health issues, although one should be careful with selecting reliable sources. Apps like HeadSpace and Calm are being downloaded by millions of people around the globe. Investors provided a record-breaking $1.5 billion to mental health startups in 2020 alone.
For most practitioners, our phones have been ringing off the hook since 2020. Applications to psychology, psychiatric residency, social work, and counseling degree programs are on the rise, with workforce shortages expected to continue for decades. Psychological expertise has been embraced by businesses especially for DEI (diversity, equity, and inclusion). Mental health experts are the most asked-for experts through media request services. Elite athletes are talking openly about bringing us on their teams.
In this unique moment, when everything seems set to transform into something else, it is time for mental health professionals to exert some agency and influence over where mental health will go from here. I think the next frontier for mental health specialists is to figure out how to speak collectively and help guide society.
Neil Howe, in his sweeping book “The Fourth Turning is Here,” says we have another 10 years in this “Millennial Crisis” phase. He calls this our “winter,” and it remains to be seen how we will emerge from our current challenges. I think we can make a difference.
If the Barbie movie is indeed a canary in the coal mine, I see positive trends ahead as we move past some of the societal and structural issues facing us, and work together to create a more open and egalitarian society. We must find creative solutions that will solve truly massive problems threatening our well-being and perhaps even our existence.
I am so grateful to be able to continue to practice and share my thoughts with you here from my home office, and I hope you can take a break and see this movie, which is not only entertaining but also thought- and emotion-provoking.
Dr. Ritvo has almost 30 years’ experience in psychiatry and is currently practicing telemedicine. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018). She has no conflicts of interest.
And it’s a great time to be a therapist
And it’s a great time to be a therapist
The Barbie movie is generating a lot of feelings, ranging from praise to vitriol. However one feels about the movie, let’s all pause and reflect for a moment on the fact that the number-one grossing film of 2023 is about our childhood doll trying to treat her anxiety disorder.
“Life imitates art more than art imitates life.” So said Oscar Wilde in 1889.
When my adult daughter, a childhood Barbie enthusiast, asked me to see the film, we put on pink and went. Twice. Little did I know that it would stir up so many thoughts and feelings. The one I want to share is how blessed I feel at this moment in time to be a mental health care provider! No longer is mental health something to be whispered about at the water cooler; instead, even Barbie is suffering. And with all the controversy in the press about the movie, no one seems at all surprised by this storyline.
I was raised by two child psychiatrists and have been practicing as an adult psychiatrist since 1991. The start of the pandemic was the most difficult time of my career, as almost every patient was struggling simultaneously, as was I. Three long years later, we are gradually emerging from our shared trauma. How ironic, now with the opportunity to go back to work, I have elected to maintain the majority of my practice online from home. It seems that most patients and providers prefer this mode of treatment, with a full 90 percent of practitioners saying they are using a hybrid model.
As mental health professionals, we know that anywhere from 3% to 49% of those experiencing trauma will develop posttraumatic stress disorder (PTSD), and we have been trained to treat them.
But what happens when an entire global population is exposed simultaneously to trauma? Historians and social scientists refer to such events by many different names, such as: Singularity, Black Swan Event, and Tipping Point. These events are incredibly rare, and afterwards everything is different. These global traumas always lead to massive change.
I think we are at that tipping point. This is the singularity. This is our Black Swan Event. Within a 3-year span, we have experienced the following:
- A global traumatic event (COVID-19).
- A sudden and seemingly permanent shift from office to remote video meetings mostly from home.
- Upending of traditional fundamentals of the stock market as the game literally stopped in January 2021.
- Rapid and widespread availability of Artificial Intelligence.
- The first generation to be fully raised on the Internet and social media (Gen Z) is now entering the workforce.
- Ongoing war in Ukraine.
That’s already an overwhelming list, and I could go on, but let’s get back to Barbie’s anxiety disorder.
The awareness about and acceptance of mental health issues has never been higher. The access to treatment never greater. There are now more online therapy options than ever. Treatment options have dramatically expanded in recent years, from Transcranial Magnetic Stimulation (TMS) to ketamine centers and psychedelics, as well as more mainstream options such as dialectical behavior therapy (DBT), cognitive behavioral therapy (CBT), selective serotonin reuptake inhibitors (SSRIs), and so many more.
What is particularly unique about this moment is the direct access to care. Self-help books abound with many making it to the New York Times bestseller list. YouTube is loaded with fantastic content on overcoming many mental health issues, although one should be careful with selecting reliable sources. Apps like HeadSpace and Calm are being downloaded by millions of people around the globe. Investors provided a record-breaking $1.5 billion to mental health startups in 2020 alone.
For most practitioners, our phones have been ringing off the hook since 2020. Applications to psychology, psychiatric residency, social work, and counseling degree programs are on the rise, with workforce shortages expected to continue for decades. Psychological expertise has been embraced by businesses especially for DEI (diversity, equity, and inclusion). Mental health experts are the most asked-for experts through media request services. Elite athletes are talking openly about bringing us on their teams.
In this unique moment, when everything seems set to transform into something else, it is time for mental health professionals to exert some agency and influence over where mental health will go from here. I think the next frontier for mental health specialists is to figure out how to speak collectively and help guide society.
Neil Howe, in his sweeping book “The Fourth Turning is Here,” says we have another 10 years in this “Millennial Crisis” phase. He calls this our “winter,” and it remains to be seen how we will emerge from our current challenges. I think we can make a difference.
If the Barbie movie is indeed a canary in the coal mine, I see positive trends ahead as we move past some of the societal and structural issues facing us, and work together to create a more open and egalitarian society. We must find creative solutions that will solve truly massive problems threatening our well-being and perhaps even our existence.
I am so grateful to be able to continue to practice and share my thoughts with you here from my home office, and I hope you can take a break and see this movie, which is not only entertaining but also thought- and emotion-provoking.
Dr. Ritvo has almost 30 years’ experience in psychiatry and is currently practicing telemedicine. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018). She has no conflicts of interest.
The Barbie movie is generating a lot of feelings, ranging from praise to vitriol. However one feels about the movie, let’s all pause and reflect for a moment on the fact that the number-one grossing film of 2023 is about our childhood doll trying to treat her anxiety disorder.
“Life imitates art more than art imitates life.” So said Oscar Wilde in 1889.
When my adult daughter, a childhood Barbie enthusiast, asked me to see the film, we put on pink and went. Twice. Little did I know that it would stir up so many thoughts and feelings. The one I want to share is how blessed I feel at this moment in time to be a mental health care provider! No longer is mental health something to be whispered about at the water cooler; instead, even Barbie is suffering. And with all the controversy in the press about the movie, no one seems at all surprised by this storyline.
I was raised by two child psychiatrists and have been practicing as an adult psychiatrist since 1991. The start of the pandemic was the most difficult time of my career, as almost every patient was struggling simultaneously, as was I. Three long years later, we are gradually emerging from our shared trauma. How ironic, now with the opportunity to go back to work, I have elected to maintain the majority of my practice online from home. It seems that most patients and providers prefer this mode of treatment, with a full 90 percent of practitioners saying they are using a hybrid model.
As mental health professionals, we know that anywhere from 3% to 49% of those experiencing trauma will develop posttraumatic stress disorder (PTSD), and we have been trained to treat them.
But what happens when an entire global population is exposed simultaneously to trauma? Historians and social scientists refer to such events by many different names, such as: Singularity, Black Swan Event, and Tipping Point. These events are incredibly rare, and afterwards everything is different. These global traumas always lead to massive change.
I think we are at that tipping point. This is the singularity. This is our Black Swan Event. Within a 3-year span, we have experienced the following:
- A global traumatic event (COVID-19).
- A sudden and seemingly permanent shift from office to remote video meetings mostly from home.
- Upending of traditional fundamentals of the stock market as the game literally stopped in January 2021.
- Rapid and widespread availability of Artificial Intelligence.
- The first generation to be fully raised on the Internet and social media (Gen Z) is now entering the workforce.
- Ongoing war in Ukraine.
That’s already an overwhelming list, and I could go on, but let’s get back to Barbie’s anxiety disorder.
The awareness about and acceptance of mental health issues has never been higher. The access to treatment never greater. There are now more online therapy options than ever. Treatment options have dramatically expanded in recent years, from Transcranial Magnetic Stimulation (TMS) to ketamine centers and psychedelics, as well as more mainstream options such as dialectical behavior therapy (DBT), cognitive behavioral therapy (CBT), selective serotonin reuptake inhibitors (SSRIs), and so many more.
What is particularly unique about this moment is the direct access to care. Self-help books abound with many making it to the New York Times bestseller list. YouTube is loaded with fantastic content on overcoming many mental health issues, although one should be careful with selecting reliable sources. Apps like HeadSpace and Calm are being downloaded by millions of people around the globe. Investors provided a record-breaking $1.5 billion to mental health startups in 2020 alone.
For most practitioners, our phones have been ringing off the hook since 2020. Applications to psychology, psychiatric residency, social work, and counseling degree programs are on the rise, with workforce shortages expected to continue for decades. Psychological expertise has been embraced by businesses especially for DEI (diversity, equity, and inclusion). Mental health experts are the most asked-for experts through media request services. Elite athletes are talking openly about bringing us on their teams.
In this unique moment, when everything seems set to transform into something else, it is time for mental health professionals to exert some agency and influence over where mental health will go from here. I think the next frontier for mental health specialists is to figure out how to speak collectively and help guide society.
Neil Howe, in his sweeping book “The Fourth Turning is Here,” says we have another 10 years in this “Millennial Crisis” phase. He calls this our “winter,” and it remains to be seen how we will emerge from our current challenges. I think we can make a difference.
If the Barbie movie is indeed a canary in the coal mine, I see positive trends ahead as we move past some of the societal and structural issues facing us, and work together to create a more open and egalitarian society. We must find creative solutions that will solve truly massive problems threatening our well-being and perhaps even our existence.
I am so grateful to be able to continue to practice and share my thoughts with you here from my home office, and I hope you can take a break and see this movie, which is not only entertaining but also thought- and emotion-provoking.
Dr. Ritvo has almost 30 years’ experience in psychiatry and is currently practicing telemedicine. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018). She has no conflicts of interest.
The cult of the suicide risk assessment
Suicide is not a trivial matter – it upends families, robs partners of a loved one, prevents children from having a parent, and can destroy a parent’s most cherished being. It is not surprising that societies have repeatedly made it a goal to study and reduce suicide within their populations.
The suicide rate in the United States is trending upward, from about 10 per 100,000 in 2000 to about 15 per 100,000 in more recent reports. The increasing suicide rates have been accompanied by increasing distress among many strata of society. From a public health level, analysts are not just witnessing increasing suicide rates, but a shocking rise in all “deaths of despair,”1 among which suicide can be considered the ultimate example.
On an individual level, many know someone who has died of suicide or suffered from a serious suicide attempt. From the public health level to the individual level, advocacy has called for various interventions in the field of psychiatry to remedy this tragic problem.
Psychiatrists have been firsthand witnesses to this increasing demand for suicide interventions. When in residency, the norm was to perform a suicide risk assessment at the time of admission to the hospital and again at the time of discharge. As the years passed, the new normal within psychiatric hospitals has shifted to asking about suicidality on a daily basis.
In what seems to us like an escalating arms race, the emerging standard of care at many facilities is now not only for daily suicide risk assessments by each psychiatrist, but also to require nurses to ask about suicidality during every 8-hour shift – in addition to documented inquiries about suicidality by other allied staff on the psychiatric unit. As a result, it is not uncommon for a patient hospitalized at an academic center to receive more than half a dozen suicide risk assessments in a day (first by the medical student, at least once – often more than once – by the resident, again by the attending psychiatrist, then the social worker and three nurses in 24 hours).
One of the concerns about such an approach is the lack of logic inherent to many risk assessment tools and symptom scales. Many of us are familiar with the Patient Health Questionnaire (PHQ-9) to assess depression.2 The PHQ-9 asks to consider “over the last 2 weeks, how often have you ...” in relation to nine symptoms associated with depression. It has always defied reason to perform a PHQ-9 every day and expect the answers to change from “nearly every day” to “not at all,” considering only 1 day has passed since the last time the patient has answered the questions. Yet daily, or near daily, PHQ-9 scores are a frequently used tool of tracking symptom improvement in response to treatments, such as electroconvulsive therapy, performed multiple times a week.
One can argue that the patient’s perspective on how symptomatic he or she has been over the past 2 weeks may change rapidly with alleviation of a depressed mood. However, the PHQ-9 is both reported to be, and often regarded as, an objective score. If one wishes to utilize it as such, the defense of its use should not be that it is a subjective report with just as much utility as “Rate your depression on a scale of 0-27.”
Similarly, many suicide scales were intended to assess thoughts of suicide in the past month3 or have been re-tooled to address this particular concern by asking “since the last contact.”4 It is baffling to see a chart with many dozens of suicide risk assessments with at times widely differing answers, yet all measuring thoughts of suicide in the past month. Is one to expect the answer to “How many times have you had these thoughts [of suicide ideation]? (1) Less than once a week (2) Once a week ...” to change between 8 a.m. and noon? Furthermore, for the purpose of assessing acute risk of suicidality in the immediate future, to only consider symptoms since the last contact – or past 2 weeks, past month, etc. – is of unclear significance.
Provider liability
Another concern is the liability placed on providers. A common problem encountered in the inpatient setting is insurance companies refusing to reimburse a hospital stay for depressed patients denying suicidality.
Any provider in the position of caring for such a patient must ask: What is the likelihood of someone providing a false negative – a false denial of suicidality? Is the likelihood of a suicidal person denying suicidality different if asked 5 or 10 or more times in a day? There are innumerable instances where a patient at a very high risk of self-harm has denied suicidality, been discharged from the hospital, and suffered terrible consequences. Ethically, the psychiatrist aware of this risk is no more at ease discharging these patients, whether it is one suicide risk scale or a dozen that suggests a patient is at low risk.
Alternatively, it may feel untenable from a medicolegal perspective for a psychiatrist to discharge a patient denying suicidality when the chart includes over a dozen previously documented elevated suicide risk assessments in the past 72 hours. By placing the job of suicide risk assessment in the hands of providers of varying levels of training and responsibility, a situation is created in which the seasoned psychiatrist who would otherwise be comfortable discharging a patient feels unable to do so because every other note-writer in the record – from the triage nurse to the medical assistant to the sitter in the emergency department – has recorded the patient as high risk for suicide. When put in such a position, the thought often occurs that systems of care, rather than individual providers, are protected most by ever escalating requirements for suicide risk documentation. To make a clinical decision contrary to the body of suicide risk documentation puts the provider at risk of being scapegoated by the system of care, which can point to its illogical and ineffective, though profusely documented, suicide prevention protocols.
Limitations of risk assessments
Considering the ongoing rise in the use of suicide risk assessments, one would expect that the evidence for their efficacy was robust and well established. Yet a thorough review of suicide risk assessments funded by the MacArthur Foundation, which examined decades of research, came to disheartening conclusions: “predictive ability has not improved over the past 50 years”; “no risk factor category or subcategory is substantially stronger than any other”; and “predicting solely according to base rates may be comparable to prediction with current risk factors.”5
Those findings were consistent with the conclusions of many other studies, which have summarized the utility of suicide risk assessments as follows: “occurrence of suicide is too low to identify those individuals who are likely to die by suicide”;6 “suicide prediction models produce accurate overall classification models, but their accuracy of predicting a future event is near zero”;7 “risk stratification is too inaccurate to be clinically useful and might even be harmful”;8 “suicide risk prediction [lacks] any items or information that to a useful degree permit the identification of persons who will complete suicide”;9 “existing suicide prediction tools have little current clinical value”;10 “our current preoccupation with risk assessment has ... created a mythology with no evidence to support it.”11 And that’s to cite just a few.
Sadly, we have known about the limitations of suicide risk assessments for many decades. In 1983 a large VA prospective study, which aimed to identify veterans who will die by suicide, examined 4,800 patients with a wide range of instruments and measures.12 This study concluded that “discriminant analysis was clearly inadequate in correctly classifying the subjects. For an event as rare as suicide, our predictive tools and guides are simply not equal to the task.” The authors described the feelings of many in stating “courts and public opinion expect physicians to be able to pick out the particular persons who will later commit suicide. Although we may reconstruct causal chains and motives, we do not possess the tools to predict suicides.”
Yet, even several decades prior, in 1954, Dr. Albert Rosen performed an elegant statistical analysis and predicted that, considering the low base rate of suicide, suicide risk assessments are “of no practical value, for it would be impossible to treat the prodigious number of false positives.”13 It seems that we continue to be unable to accept Dr. Rosen’s premonition despite decades of confirmatory evidence.
“Quantity over quality”
Regardless of those sobering reports,
One can reasonably argue that the periodic performance of a suicide risk assessment may have clinical utility in reminding us of modifiable risk factors such as intoxication, social isolation, and access to lethal means. One can also reasonably argue that these risk assessments may provide useful education to patients and their families on epidemiological risk factors such as gender, age, and marital status. But our pursuit of serial suicide risk assessments throughout the day is encouraging providers to focus on a particular risk factor that changes from moment to moment and has particularly low validity, that being self-reported suicidality.
Reported suicidality is one of the few risk factors that can change from shift to shift. But 80% of people who die by suicide had not previously expressed suicidality, and 98.3% of people who have endorsed suicidality do not die by suicide.14 While the former statistic may improve with increased assessment, the later will likely worsen.
Suicide is not a trivial matter. We admire those that study it and advocate for better interventions. We have compassion for those who have suffered the loss of a loved one to suicide. Our patients have died as a result of the human limitations surrounding suicide prevention. Recognizing the weight of suicide and making an effort to avoid minimizing its immense consequences drive our desire to be honest with ourselves, our patients and their families, and society. That includes the unfortunate truth regarding the current state of the evidence and our ability to enact change.
It is our concern that the rising fascination with repeated suicide risk assessment is misguided in its current form and serves the purpose of appeasing administrators more than reflecting a scientific understanding of the literature. More sadly, we are concerned that this “quantity-over-quality” approach is yet another barrier to practicing what may be one of the few interventions with any hope of meaningfully impacting a patient’s risk of suicide in the clinical setting – spending time connecting with our patients.
Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. Dr. Compton is a member of the psychiatry faculty at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research. Dr. Badre and Dr. Compton have no conflicts of interest.
References
1. Joint Economic Committee. (2019). Long Term Trends in Deaths of Despair. SCP Report 4-19.
2. Kroenke K and Spitzer RL. The PHQ-9: A new depression diagnostic and severity measure. Psychiatr Ann. 2013;32(9):509-15. doi: 10.3928/0048-5713-20020901-06.
3. Columbia-Suicide Severity Rating Scale (C-SSRS) Full Lifetime/Recent.
4. Columbia-Suicide Severity Rating Scale (C-SSRS) Full Since Last Contact.
5. Franklin JC et al. Risk factors for suicidal thoughts and behaviors: A meta-analysis of 50 years of research. Psychol Bull. 2017 Feb;143(2):187-232. doi: 10.1037/bul0000084.
6. Beautrais AL. Further suicidal behavior among medically serious suicide attempters. Suicide Life Threat Behav. 2004 Spring;34(1):1-11. doi: 10.1521/suli.34.1.1.27772.
7. Belsher BE. Prediction models for suicide attempts and deaths: A systematic review and simulation. JAMA Psychiatry. 2019 Jun 1;76(6):642-651. doi: 10.1001/jamapsychiatry.2019.0174.
8. Carter G et al. Royal Australian and New Zealand College of Psychiatrists clinical practice guideline for the management of deliberate self-harm. Aust N Z J Psychiatry. 2016 Oct;50(10):939-1000. doi: 10.1177/0004867416661039.
9. Fosse R et al. Predictors of suicide in the patient population admitted to a locked-door psychiatric acute ward. PLoS One. 2017 Mar 16;12(3):e0173958. doi: 10.1371/journal.pone.0173958.
10. Kessler RC et al. Suicide prediction models: A critical review of recent research with recommendations for the way forward. Mol Psychiatry. 2020 Jan;25(1):168-79. doi: 10.1038/s41380-019-0531-0.
11. Mulder R. Problems with suicide risk assessment. Aust N Z J Psychiatry. 2011 Aug;45(8):605-7. doi: 10.3109/00048674.2011.594786.
12. Pokorny AD. Prediction of suicide in psychiatric patients: Report of a prospective study. Arch Gen Psychiatry. 1983 Mar;40(3):249-57. doi: 10.1001/archpsyc.1983.01790030019002.
13. Rosen A. Detection of suicidal patients: An example of some limitations in the prediction of infrequent events. J Consult Psychol. 1954 Dec;18(6):397-403. doi: 10.1037/h0058579.
14. McHugh CM et al. (2019). Association between suicidal ideation and suicide: Meta-analyses of odds ratios, sensitivity, specificity and positive predictive value. BJPsych Open. 2019 Mar;5(2):e18. doi: 10.1192/bjo.2018.88.
Suicide is not a trivial matter – it upends families, robs partners of a loved one, prevents children from having a parent, and can destroy a parent’s most cherished being. It is not surprising that societies have repeatedly made it a goal to study and reduce suicide within their populations.
The suicide rate in the United States is trending upward, from about 10 per 100,000 in 2000 to about 15 per 100,000 in more recent reports. The increasing suicide rates have been accompanied by increasing distress among many strata of society. From a public health level, analysts are not just witnessing increasing suicide rates, but a shocking rise in all “deaths of despair,”1 among which suicide can be considered the ultimate example.
On an individual level, many know someone who has died of suicide or suffered from a serious suicide attempt. From the public health level to the individual level, advocacy has called for various interventions in the field of psychiatry to remedy this tragic problem.
Psychiatrists have been firsthand witnesses to this increasing demand for suicide interventions. When in residency, the norm was to perform a suicide risk assessment at the time of admission to the hospital and again at the time of discharge. As the years passed, the new normal within psychiatric hospitals has shifted to asking about suicidality on a daily basis.
In what seems to us like an escalating arms race, the emerging standard of care at many facilities is now not only for daily suicide risk assessments by each psychiatrist, but also to require nurses to ask about suicidality during every 8-hour shift – in addition to documented inquiries about suicidality by other allied staff on the psychiatric unit. As a result, it is not uncommon for a patient hospitalized at an academic center to receive more than half a dozen suicide risk assessments in a day (first by the medical student, at least once – often more than once – by the resident, again by the attending psychiatrist, then the social worker and three nurses in 24 hours).
One of the concerns about such an approach is the lack of logic inherent to many risk assessment tools and symptom scales. Many of us are familiar with the Patient Health Questionnaire (PHQ-9) to assess depression.2 The PHQ-9 asks to consider “over the last 2 weeks, how often have you ...” in relation to nine symptoms associated with depression. It has always defied reason to perform a PHQ-9 every day and expect the answers to change from “nearly every day” to “not at all,” considering only 1 day has passed since the last time the patient has answered the questions. Yet daily, or near daily, PHQ-9 scores are a frequently used tool of tracking symptom improvement in response to treatments, such as electroconvulsive therapy, performed multiple times a week.
One can argue that the patient’s perspective on how symptomatic he or she has been over the past 2 weeks may change rapidly with alleviation of a depressed mood. However, the PHQ-9 is both reported to be, and often regarded as, an objective score. If one wishes to utilize it as such, the defense of its use should not be that it is a subjective report with just as much utility as “Rate your depression on a scale of 0-27.”
Similarly, many suicide scales were intended to assess thoughts of suicide in the past month3 or have been re-tooled to address this particular concern by asking “since the last contact.”4 It is baffling to see a chart with many dozens of suicide risk assessments with at times widely differing answers, yet all measuring thoughts of suicide in the past month. Is one to expect the answer to “How many times have you had these thoughts [of suicide ideation]? (1) Less than once a week (2) Once a week ...” to change between 8 a.m. and noon? Furthermore, for the purpose of assessing acute risk of suicidality in the immediate future, to only consider symptoms since the last contact – or past 2 weeks, past month, etc. – is of unclear significance.
Provider liability
Another concern is the liability placed on providers. A common problem encountered in the inpatient setting is insurance companies refusing to reimburse a hospital stay for depressed patients denying suicidality.
Any provider in the position of caring for such a patient must ask: What is the likelihood of someone providing a false negative – a false denial of suicidality? Is the likelihood of a suicidal person denying suicidality different if asked 5 or 10 or more times in a day? There are innumerable instances where a patient at a very high risk of self-harm has denied suicidality, been discharged from the hospital, and suffered terrible consequences. Ethically, the psychiatrist aware of this risk is no more at ease discharging these patients, whether it is one suicide risk scale or a dozen that suggests a patient is at low risk.
Alternatively, it may feel untenable from a medicolegal perspective for a psychiatrist to discharge a patient denying suicidality when the chart includes over a dozen previously documented elevated suicide risk assessments in the past 72 hours. By placing the job of suicide risk assessment in the hands of providers of varying levels of training and responsibility, a situation is created in which the seasoned psychiatrist who would otherwise be comfortable discharging a patient feels unable to do so because every other note-writer in the record – from the triage nurse to the medical assistant to the sitter in the emergency department – has recorded the patient as high risk for suicide. When put in such a position, the thought often occurs that systems of care, rather than individual providers, are protected most by ever escalating requirements for suicide risk documentation. To make a clinical decision contrary to the body of suicide risk documentation puts the provider at risk of being scapegoated by the system of care, which can point to its illogical and ineffective, though profusely documented, suicide prevention protocols.
Limitations of risk assessments
Considering the ongoing rise in the use of suicide risk assessments, one would expect that the evidence for their efficacy was robust and well established. Yet a thorough review of suicide risk assessments funded by the MacArthur Foundation, which examined decades of research, came to disheartening conclusions: “predictive ability has not improved over the past 50 years”; “no risk factor category or subcategory is substantially stronger than any other”; and “predicting solely according to base rates may be comparable to prediction with current risk factors.”5
Those findings were consistent with the conclusions of many other studies, which have summarized the utility of suicide risk assessments as follows: “occurrence of suicide is too low to identify those individuals who are likely to die by suicide”;6 “suicide prediction models produce accurate overall classification models, but their accuracy of predicting a future event is near zero”;7 “risk stratification is too inaccurate to be clinically useful and might even be harmful”;8 “suicide risk prediction [lacks] any items or information that to a useful degree permit the identification of persons who will complete suicide”;9 “existing suicide prediction tools have little current clinical value”;10 “our current preoccupation with risk assessment has ... created a mythology with no evidence to support it.”11 And that’s to cite just a few.
Sadly, we have known about the limitations of suicide risk assessments for many decades. In 1983 a large VA prospective study, which aimed to identify veterans who will die by suicide, examined 4,800 patients with a wide range of instruments and measures.12 This study concluded that “discriminant analysis was clearly inadequate in correctly classifying the subjects. For an event as rare as suicide, our predictive tools and guides are simply not equal to the task.” The authors described the feelings of many in stating “courts and public opinion expect physicians to be able to pick out the particular persons who will later commit suicide. Although we may reconstruct causal chains and motives, we do not possess the tools to predict suicides.”
Yet, even several decades prior, in 1954, Dr. Albert Rosen performed an elegant statistical analysis and predicted that, considering the low base rate of suicide, suicide risk assessments are “of no practical value, for it would be impossible to treat the prodigious number of false positives.”13 It seems that we continue to be unable to accept Dr. Rosen’s premonition despite decades of confirmatory evidence.
“Quantity over quality”
Regardless of those sobering reports,
One can reasonably argue that the periodic performance of a suicide risk assessment may have clinical utility in reminding us of modifiable risk factors such as intoxication, social isolation, and access to lethal means. One can also reasonably argue that these risk assessments may provide useful education to patients and their families on epidemiological risk factors such as gender, age, and marital status. But our pursuit of serial suicide risk assessments throughout the day is encouraging providers to focus on a particular risk factor that changes from moment to moment and has particularly low validity, that being self-reported suicidality.
Reported suicidality is one of the few risk factors that can change from shift to shift. But 80% of people who die by suicide had not previously expressed suicidality, and 98.3% of people who have endorsed suicidality do not die by suicide.14 While the former statistic may improve with increased assessment, the later will likely worsen.
Suicide is not a trivial matter. We admire those that study it and advocate for better interventions. We have compassion for those who have suffered the loss of a loved one to suicide. Our patients have died as a result of the human limitations surrounding suicide prevention. Recognizing the weight of suicide and making an effort to avoid minimizing its immense consequences drive our desire to be honest with ourselves, our patients and their families, and society. That includes the unfortunate truth regarding the current state of the evidence and our ability to enact change.
It is our concern that the rising fascination with repeated suicide risk assessment is misguided in its current form and serves the purpose of appeasing administrators more than reflecting a scientific understanding of the literature. More sadly, we are concerned that this “quantity-over-quality” approach is yet another barrier to practicing what may be one of the few interventions with any hope of meaningfully impacting a patient’s risk of suicide in the clinical setting – spending time connecting with our patients.
Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. Dr. Compton is a member of the psychiatry faculty at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research. Dr. Badre and Dr. Compton have no conflicts of interest.
References
1. Joint Economic Committee. (2019). Long Term Trends in Deaths of Despair. SCP Report 4-19.
2. Kroenke K and Spitzer RL. The PHQ-9: A new depression diagnostic and severity measure. Psychiatr Ann. 2013;32(9):509-15. doi: 10.3928/0048-5713-20020901-06.
3. Columbia-Suicide Severity Rating Scale (C-SSRS) Full Lifetime/Recent.
4. Columbia-Suicide Severity Rating Scale (C-SSRS) Full Since Last Contact.
5. Franklin JC et al. Risk factors for suicidal thoughts and behaviors: A meta-analysis of 50 years of research. Psychol Bull. 2017 Feb;143(2):187-232. doi: 10.1037/bul0000084.
6. Beautrais AL. Further suicidal behavior among medically serious suicide attempters. Suicide Life Threat Behav. 2004 Spring;34(1):1-11. doi: 10.1521/suli.34.1.1.27772.
7. Belsher BE. Prediction models for suicide attempts and deaths: A systematic review and simulation. JAMA Psychiatry. 2019 Jun 1;76(6):642-651. doi: 10.1001/jamapsychiatry.2019.0174.
8. Carter G et al. Royal Australian and New Zealand College of Psychiatrists clinical practice guideline for the management of deliberate self-harm. Aust N Z J Psychiatry. 2016 Oct;50(10):939-1000. doi: 10.1177/0004867416661039.
9. Fosse R et al. Predictors of suicide in the patient population admitted to a locked-door psychiatric acute ward. PLoS One. 2017 Mar 16;12(3):e0173958. doi: 10.1371/journal.pone.0173958.
10. Kessler RC et al. Suicide prediction models: A critical review of recent research with recommendations for the way forward. Mol Psychiatry. 2020 Jan;25(1):168-79. doi: 10.1038/s41380-019-0531-0.
11. Mulder R. Problems with suicide risk assessment. Aust N Z J Psychiatry. 2011 Aug;45(8):605-7. doi: 10.3109/00048674.2011.594786.
12. Pokorny AD. Prediction of suicide in psychiatric patients: Report of a prospective study. Arch Gen Psychiatry. 1983 Mar;40(3):249-57. doi: 10.1001/archpsyc.1983.01790030019002.
13. Rosen A. Detection of suicidal patients: An example of some limitations in the prediction of infrequent events. J Consult Psychol. 1954 Dec;18(6):397-403. doi: 10.1037/h0058579.
14. McHugh CM et al. (2019). Association between suicidal ideation and suicide: Meta-analyses of odds ratios, sensitivity, specificity and positive predictive value. BJPsych Open. 2019 Mar;5(2):e18. doi: 10.1192/bjo.2018.88.
Suicide is not a trivial matter – it upends families, robs partners of a loved one, prevents children from having a parent, and can destroy a parent’s most cherished being. It is not surprising that societies have repeatedly made it a goal to study and reduce suicide within their populations.
The suicide rate in the United States is trending upward, from about 10 per 100,000 in 2000 to about 15 per 100,000 in more recent reports. The increasing suicide rates have been accompanied by increasing distress among many strata of society. From a public health level, analysts are not just witnessing increasing suicide rates, but a shocking rise in all “deaths of despair,”1 among which suicide can be considered the ultimate example.
On an individual level, many know someone who has died of suicide or suffered from a serious suicide attempt. From the public health level to the individual level, advocacy has called for various interventions in the field of psychiatry to remedy this tragic problem.
Psychiatrists have been firsthand witnesses to this increasing demand for suicide interventions. When in residency, the norm was to perform a suicide risk assessment at the time of admission to the hospital and again at the time of discharge. As the years passed, the new normal within psychiatric hospitals has shifted to asking about suicidality on a daily basis.
In what seems to us like an escalating arms race, the emerging standard of care at many facilities is now not only for daily suicide risk assessments by each psychiatrist, but also to require nurses to ask about suicidality during every 8-hour shift – in addition to documented inquiries about suicidality by other allied staff on the psychiatric unit. As a result, it is not uncommon for a patient hospitalized at an academic center to receive more than half a dozen suicide risk assessments in a day (first by the medical student, at least once – often more than once – by the resident, again by the attending psychiatrist, then the social worker and three nurses in 24 hours).
One of the concerns about such an approach is the lack of logic inherent to many risk assessment tools and symptom scales. Many of us are familiar with the Patient Health Questionnaire (PHQ-9) to assess depression.2 The PHQ-9 asks to consider “over the last 2 weeks, how often have you ...” in relation to nine symptoms associated with depression. It has always defied reason to perform a PHQ-9 every day and expect the answers to change from “nearly every day” to “not at all,” considering only 1 day has passed since the last time the patient has answered the questions. Yet daily, or near daily, PHQ-9 scores are a frequently used tool of tracking symptom improvement in response to treatments, such as electroconvulsive therapy, performed multiple times a week.
One can argue that the patient’s perspective on how symptomatic he or she has been over the past 2 weeks may change rapidly with alleviation of a depressed mood. However, the PHQ-9 is both reported to be, and often regarded as, an objective score. If one wishes to utilize it as such, the defense of its use should not be that it is a subjective report with just as much utility as “Rate your depression on a scale of 0-27.”
Similarly, many suicide scales were intended to assess thoughts of suicide in the past month3 or have been re-tooled to address this particular concern by asking “since the last contact.”4 It is baffling to see a chart with many dozens of suicide risk assessments with at times widely differing answers, yet all measuring thoughts of suicide in the past month. Is one to expect the answer to “How many times have you had these thoughts [of suicide ideation]? (1) Less than once a week (2) Once a week ...” to change between 8 a.m. and noon? Furthermore, for the purpose of assessing acute risk of suicidality in the immediate future, to only consider symptoms since the last contact – or past 2 weeks, past month, etc. – is of unclear significance.
Provider liability
Another concern is the liability placed on providers. A common problem encountered in the inpatient setting is insurance companies refusing to reimburse a hospital stay for depressed patients denying suicidality.
Any provider in the position of caring for such a patient must ask: What is the likelihood of someone providing a false negative – a false denial of suicidality? Is the likelihood of a suicidal person denying suicidality different if asked 5 or 10 or more times in a day? There are innumerable instances where a patient at a very high risk of self-harm has denied suicidality, been discharged from the hospital, and suffered terrible consequences. Ethically, the psychiatrist aware of this risk is no more at ease discharging these patients, whether it is one suicide risk scale or a dozen that suggests a patient is at low risk.
Alternatively, it may feel untenable from a medicolegal perspective for a psychiatrist to discharge a patient denying suicidality when the chart includes over a dozen previously documented elevated suicide risk assessments in the past 72 hours. By placing the job of suicide risk assessment in the hands of providers of varying levels of training and responsibility, a situation is created in which the seasoned psychiatrist who would otherwise be comfortable discharging a patient feels unable to do so because every other note-writer in the record – from the triage nurse to the medical assistant to the sitter in the emergency department – has recorded the patient as high risk for suicide. When put in such a position, the thought often occurs that systems of care, rather than individual providers, are protected most by ever escalating requirements for suicide risk documentation. To make a clinical decision contrary to the body of suicide risk documentation puts the provider at risk of being scapegoated by the system of care, which can point to its illogical and ineffective, though profusely documented, suicide prevention protocols.
Limitations of risk assessments
Considering the ongoing rise in the use of suicide risk assessments, one would expect that the evidence for their efficacy was robust and well established. Yet a thorough review of suicide risk assessments funded by the MacArthur Foundation, which examined decades of research, came to disheartening conclusions: “predictive ability has not improved over the past 50 years”; “no risk factor category or subcategory is substantially stronger than any other”; and “predicting solely according to base rates may be comparable to prediction with current risk factors.”5
Those findings were consistent with the conclusions of many other studies, which have summarized the utility of suicide risk assessments as follows: “occurrence of suicide is too low to identify those individuals who are likely to die by suicide”;6 “suicide prediction models produce accurate overall classification models, but their accuracy of predicting a future event is near zero”;7 “risk stratification is too inaccurate to be clinically useful and might even be harmful”;8 “suicide risk prediction [lacks] any items or information that to a useful degree permit the identification of persons who will complete suicide”;9 “existing suicide prediction tools have little current clinical value”;10 “our current preoccupation with risk assessment has ... created a mythology with no evidence to support it.”11 And that’s to cite just a few.
Sadly, we have known about the limitations of suicide risk assessments for many decades. In 1983 a large VA prospective study, which aimed to identify veterans who will die by suicide, examined 4,800 patients with a wide range of instruments and measures.12 This study concluded that “discriminant analysis was clearly inadequate in correctly classifying the subjects. For an event as rare as suicide, our predictive tools and guides are simply not equal to the task.” The authors described the feelings of many in stating “courts and public opinion expect physicians to be able to pick out the particular persons who will later commit suicide. Although we may reconstruct causal chains and motives, we do not possess the tools to predict suicides.”
Yet, even several decades prior, in 1954, Dr. Albert Rosen performed an elegant statistical analysis and predicted that, considering the low base rate of suicide, suicide risk assessments are “of no practical value, for it would be impossible to treat the prodigious number of false positives.”13 It seems that we continue to be unable to accept Dr. Rosen’s premonition despite decades of confirmatory evidence.
“Quantity over quality”
Regardless of those sobering reports,
One can reasonably argue that the periodic performance of a suicide risk assessment may have clinical utility in reminding us of modifiable risk factors such as intoxication, social isolation, and access to lethal means. One can also reasonably argue that these risk assessments may provide useful education to patients and their families on epidemiological risk factors such as gender, age, and marital status. But our pursuit of serial suicide risk assessments throughout the day is encouraging providers to focus on a particular risk factor that changes from moment to moment and has particularly low validity, that being self-reported suicidality.
Reported suicidality is one of the few risk factors that can change from shift to shift. But 80% of people who die by suicide had not previously expressed suicidality, and 98.3% of people who have endorsed suicidality do not die by suicide.14 While the former statistic may improve with increased assessment, the later will likely worsen.
Suicide is not a trivial matter. We admire those that study it and advocate for better interventions. We have compassion for those who have suffered the loss of a loved one to suicide. Our patients have died as a result of the human limitations surrounding suicide prevention. Recognizing the weight of suicide and making an effort to avoid minimizing its immense consequences drive our desire to be honest with ourselves, our patients and their families, and society. That includes the unfortunate truth regarding the current state of the evidence and our ability to enact change.
It is our concern that the rising fascination with repeated suicide risk assessment is misguided in its current form and serves the purpose of appeasing administrators more than reflecting a scientific understanding of the literature. More sadly, we are concerned that this “quantity-over-quality” approach is yet another barrier to practicing what may be one of the few interventions with any hope of meaningfully impacting a patient’s risk of suicide in the clinical setting – spending time connecting with our patients.
Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. Dr. Compton is a member of the psychiatry faculty at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research. Dr. Badre and Dr. Compton have no conflicts of interest.
References
1. Joint Economic Committee. (2019). Long Term Trends in Deaths of Despair. SCP Report 4-19.
2. Kroenke K and Spitzer RL. The PHQ-9: A new depression diagnostic and severity measure. Psychiatr Ann. 2013;32(9):509-15. doi: 10.3928/0048-5713-20020901-06.
3. Columbia-Suicide Severity Rating Scale (C-SSRS) Full Lifetime/Recent.
4. Columbia-Suicide Severity Rating Scale (C-SSRS) Full Since Last Contact.
5. Franklin JC et al. Risk factors for suicidal thoughts and behaviors: A meta-analysis of 50 years of research. Psychol Bull. 2017 Feb;143(2):187-232. doi: 10.1037/bul0000084.
6. Beautrais AL. Further suicidal behavior among medically serious suicide attempters. Suicide Life Threat Behav. 2004 Spring;34(1):1-11. doi: 10.1521/suli.34.1.1.27772.
7. Belsher BE. Prediction models for suicide attempts and deaths: A systematic review and simulation. JAMA Psychiatry. 2019 Jun 1;76(6):642-651. doi: 10.1001/jamapsychiatry.2019.0174.
8. Carter G et al. Royal Australian and New Zealand College of Psychiatrists clinical practice guideline for the management of deliberate self-harm. Aust N Z J Psychiatry. 2016 Oct;50(10):939-1000. doi: 10.1177/0004867416661039.
9. Fosse R et al. Predictors of suicide in the patient population admitted to a locked-door psychiatric acute ward. PLoS One. 2017 Mar 16;12(3):e0173958. doi: 10.1371/journal.pone.0173958.
10. Kessler RC et al. Suicide prediction models: A critical review of recent research with recommendations for the way forward. Mol Psychiatry. 2020 Jan;25(1):168-79. doi: 10.1038/s41380-019-0531-0.
11. Mulder R. Problems with suicide risk assessment. Aust N Z J Psychiatry. 2011 Aug;45(8):605-7. doi: 10.3109/00048674.2011.594786.
12. Pokorny AD. Prediction of suicide in psychiatric patients: Report of a prospective study. Arch Gen Psychiatry. 1983 Mar;40(3):249-57. doi: 10.1001/archpsyc.1983.01790030019002.
13. Rosen A. Detection of suicidal patients: An example of some limitations in the prediction of infrequent events. J Consult Psychol. 1954 Dec;18(6):397-403. doi: 10.1037/h0058579.
14. McHugh CM et al. (2019). Association between suicidal ideation and suicide: Meta-analyses of odds ratios, sensitivity, specificity and positive predictive value. BJPsych Open. 2019 Mar;5(2):e18. doi: 10.1192/bjo.2018.88.
Can a decrease in dopamine lead to binge eating?
In medical school, we were repeatedly advised that there is both a science and an art to the practice of medicine. In these days of doc-in-a-box online consultations for obesity, it’s tempting to think that there’s a one-size-fits-all purely scientific approach for these new weight loss medications. Yet, for every nine patients who lose weight seemingly effortlessly on this class of medication, there is always one whose body stubbornly refuses to submit.
Adam is a 58-year-old man who came to me recently because he was having difficulty losing weight. Over the past 20 years, he’d been steadily gaining weight and now, technically has morbid obesity (a term which should arguably be obsolete). His weight gain is complicated by high blood pressure, high cholesterol, and obstructive sleep apnea. His sleep apnea has caused such profound exhaustion that he no longer has the energy to work out. He also has significant ADHD, which has been left untreated because of his ability to white-knuckle it through his many daily meetings and calls. A married father of three, he is a successful portfolio manager at a high-yield bond fund.
Adam tends to eat minimally during the day, thereby baffling his colleagues with the stark contrast between his minimal caloric intake and his large belly. However, when he returns from work late at night (kids safely tucked into bed), the floodgates open. He reports polishing off pints of ice cream, scarfing down bags of cookies, inhaling trays of brownies. No carbohydrate is off limits to him once he steps off the Metro North train and crosses the threshold from work to home.
Does Adam simply lack the desire or common-sense willpower to make the necessary changes in his lifestyle or is there something more complicated at play?
I would argue that Adam’s ADHD triggered a binge-eating disorder (BED) that festered unchecked over the past 20 years. Patients with BED typically eat massive quantities of food over short periods of time – often when they’re not even hungry. Adam admitted that he would generally continue to eat well after feeling stuffed to the brim.
The answer probably lies with dopamine, a neurotransmitter produced in the reward centers of the brain that regulates how people experience pleasure and control impulses. We believe that people with ADHD have low levels of dopamine (it’s actually a bit more complicated, but this is the general idea). These low levels of dopamine lead people to self-medicate with sugars, salt, and fats to increase dopamine levels.
Lisdexamfetamine (Vyvanse) is a Food and Drug Administration–approved treatment option for both ADHD and binge eating. It raises the levels of dopamine (as well as norepinephrine) in the brain’s reward center. Often, the strong urge to binge subsides rapidly once ADHD is properly treated.
Rather than starting Adam on a semaglutide or similar agent, I opted to start him on lisdexamfetamine. When I spoke to him 1 week later, he confided that the world suddenly shifted into focus, and he was able to plan his meals throughout the day and resist the urge to binge late at night.
I may eventually add a semaglutide-like medication if his weight loss plateaus, but for now, I will focus on raising his dopamine levels to tackle the underlying cause of his weight gain.
Dr. Messer is a clinical assistant professor at the Icahn School of Medicine at Mount Sinai, New York. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
In medical school, we were repeatedly advised that there is both a science and an art to the practice of medicine. In these days of doc-in-a-box online consultations for obesity, it’s tempting to think that there’s a one-size-fits-all purely scientific approach for these new weight loss medications. Yet, for every nine patients who lose weight seemingly effortlessly on this class of medication, there is always one whose body stubbornly refuses to submit.
Adam is a 58-year-old man who came to me recently because he was having difficulty losing weight. Over the past 20 years, he’d been steadily gaining weight and now, technically has morbid obesity (a term which should arguably be obsolete). His weight gain is complicated by high blood pressure, high cholesterol, and obstructive sleep apnea. His sleep apnea has caused such profound exhaustion that he no longer has the energy to work out. He also has significant ADHD, which has been left untreated because of his ability to white-knuckle it through his many daily meetings and calls. A married father of three, he is a successful portfolio manager at a high-yield bond fund.
Adam tends to eat minimally during the day, thereby baffling his colleagues with the stark contrast between his minimal caloric intake and his large belly. However, when he returns from work late at night (kids safely tucked into bed), the floodgates open. He reports polishing off pints of ice cream, scarfing down bags of cookies, inhaling trays of brownies. No carbohydrate is off limits to him once he steps off the Metro North train and crosses the threshold from work to home.
Does Adam simply lack the desire or common-sense willpower to make the necessary changes in his lifestyle or is there something more complicated at play?
I would argue that Adam’s ADHD triggered a binge-eating disorder (BED) that festered unchecked over the past 20 years. Patients with BED typically eat massive quantities of food over short periods of time – often when they’re not even hungry. Adam admitted that he would generally continue to eat well after feeling stuffed to the brim.
The answer probably lies with dopamine, a neurotransmitter produced in the reward centers of the brain that regulates how people experience pleasure and control impulses. We believe that people with ADHD have low levels of dopamine (it’s actually a bit more complicated, but this is the general idea). These low levels of dopamine lead people to self-medicate with sugars, salt, and fats to increase dopamine levels.
Lisdexamfetamine (Vyvanse) is a Food and Drug Administration–approved treatment option for both ADHD and binge eating. It raises the levels of dopamine (as well as norepinephrine) in the brain’s reward center. Often, the strong urge to binge subsides rapidly once ADHD is properly treated.
Rather than starting Adam on a semaglutide or similar agent, I opted to start him on lisdexamfetamine. When I spoke to him 1 week later, he confided that the world suddenly shifted into focus, and he was able to plan his meals throughout the day and resist the urge to binge late at night.
I may eventually add a semaglutide-like medication if his weight loss plateaus, but for now, I will focus on raising his dopamine levels to tackle the underlying cause of his weight gain.
Dr. Messer is a clinical assistant professor at the Icahn School of Medicine at Mount Sinai, New York. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
In medical school, we were repeatedly advised that there is both a science and an art to the practice of medicine. In these days of doc-in-a-box online consultations for obesity, it’s tempting to think that there’s a one-size-fits-all purely scientific approach for these new weight loss medications. Yet, for every nine patients who lose weight seemingly effortlessly on this class of medication, there is always one whose body stubbornly refuses to submit.
Adam is a 58-year-old man who came to me recently because he was having difficulty losing weight. Over the past 20 years, he’d been steadily gaining weight and now, technically has morbid obesity (a term which should arguably be obsolete). His weight gain is complicated by high blood pressure, high cholesterol, and obstructive sleep apnea. His sleep apnea has caused such profound exhaustion that he no longer has the energy to work out. He also has significant ADHD, which has been left untreated because of his ability to white-knuckle it through his many daily meetings and calls. A married father of three, he is a successful portfolio manager at a high-yield bond fund.
Adam tends to eat minimally during the day, thereby baffling his colleagues with the stark contrast between his minimal caloric intake and his large belly. However, when he returns from work late at night (kids safely tucked into bed), the floodgates open. He reports polishing off pints of ice cream, scarfing down bags of cookies, inhaling trays of brownies. No carbohydrate is off limits to him once he steps off the Metro North train and crosses the threshold from work to home.
Does Adam simply lack the desire or common-sense willpower to make the necessary changes in his lifestyle or is there something more complicated at play?
I would argue that Adam’s ADHD triggered a binge-eating disorder (BED) that festered unchecked over the past 20 years. Patients with BED typically eat massive quantities of food over short periods of time – often when they’re not even hungry. Adam admitted that he would generally continue to eat well after feeling stuffed to the brim.
The answer probably lies with dopamine, a neurotransmitter produced in the reward centers of the brain that regulates how people experience pleasure and control impulses. We believe that people with ADHD have low levels of dopamine (it’s actually a bit more complicated, but this is the general idea). These low levels of dopamine lead people to self-medicate with sugars, salt, and fats to increase dopamine levels.
Lisdexamfetamine (Vyvanse) is a Food and Drug Administration–approved treatment option for both ADHD and binge eating. It raises the levels of dopamine (as well as norepinephrine) in the brain’s reward center. Often, the strong urge to binge subsides rapidly once ADHD is properly treated.
Rather than starting Adam on a semaglutide or similar agent, I opted to start him on lisdexamfetamine. When I spoke to him 1 week later, he confided that the world suddenly shifted into focus, and he was able to plan his meals throughout the day and resist the urge to binge late at night.
I may eventually add a semaglutide-like medication if his weight loss plateaus, but for now, I will focus on raising his dopamine levels to tackle the underlying cause of his weight gain.
Dr. Messer is a clinical assistant professor at the Icahn School of Medicine at Mount Sinai, New York. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Psilocybin reduces symptoms, disability in major depression
The randomized, phase 2 trial was conducted at 11 sites across the United States and is the latest to demonstrate the psychedelic drug’s potential as a treatment for depression.
The project was funded by Usona Institute, a nonprofit medical research organization based in Madison, Wisc. The institute issued a press statement, but researchers did not comment further on the findings.
“As the largest and most rigorous study conducted across a wide spectrum of individuals with major depressive disorder, the results show promise for all people struggling with this condition,” lead author Charles Raison, MD, director of clinical and translational research at Usona, said in the statement.
The 34 coauthors on the study are affiliated with public universities, research centers, and private companies. Eight of the investigators are identified as employees of Usona Institute.
Declining further comment, an institute spokesperson told this news organization that, “Usona has chosen the approach of no interviews, and this applies for all coauthors.”
The findings were published online in JAMA.
Largest study to date
Usona’s investigational psilocybin drug has been granted a breakthrough designation by the Food and Drug Administration, a process designed to speed drug development and review.
Previous smaller studies have suggested a rapid antidepressant response with psilocybin, but they have been small, unblinded, and have had short duration of follow-up, they write. This randomized, double-blind, phase 2 clinical trial is the largest study of psilocybin for depression to date, the researchers note.
It included 104 adults aged 21-65 years with MDD who had a current depressive episode of at least 60 days and a Montgomery-Åsberg Depression Rating Scale (MADRS) total score of 28 or more at baseline.
Participants had to be free of psychedelic drugs for at least 5 years, have had no active suicidal ideation or suicidal behavior in the prior 12 months, no personal or first-degree family history of psychosis or mania, and no history of moderate/severe alcohol or drug use disorder.
Before the study, participants had a 7- to 35-day screening period for psychiatric medication tapering, underwent baseline assessments, and received 6-8 hours of preparation with two facilitators who would be with them during dosing.
Dosing occurred within 7 days of baseline assessments. During the 6- to 8-hour session, participants received either a single 25-mg oral dose of psilocybin or 100-mg dose of niacin. One participant randomly assigned to receive psilocybin received the incorrect treatment, resulting in 50 participants receiving psilocybin and 54 receiving niacin.
Participants returned the next day, the next week, and then every 2 weeks for assessments, for a follow-up of 6 weeks.
Psychosocial support
Participants who received psilocybin reported significantly greater improvements in MDD symptoms, compared with those who received niacin. MADRS scores – a scale from 0 to 60 where higher scores indicate more severe depression – showed greater reductions with treatment vs. placebo at 8 days (mean difference, −12.0; 95% confidence interval, −16.6 to −7.4; P < .001), and at day 43 (mean difference, −12.3; 95% CI, −17.5 to −7.2; P < .001).
More participants receiving psilocybin had sustained depressive symptom response (42% vs. 11%; P = .002) and more improvement in the Sheehan Disability Scale score, which measures functional disability, 43 days after treatment (P < .001).
The effects persisted through the end of the study, although the differences between groups were no longer significant by week 6.
“This is another exciting piece of evidence that adds to the current literature regarding the potential efficacy of psilocybin for the treatment of mental health conditions, particularly depression,” said Greg Fonzo, MD, codirector of the Center for Psychedelic Research and Therapy at the University of Texas at Austin, who commented on the findings.
Significantly more people in the psilocybin group reported at least one treatment-related adverse event (AE, 82% vs. 44%), although most were mild to moderate. Headache and nausea were the most common side effects and most resolved within 1 day of dosing.
While those numbers are high, Dr. Fonzo said they’re not out of line with AEs reported in other studies.
“Particularly with the types of adverse events reported here, like headache and nausea, those are things you would typically expect to see in this treatment,” said Dr. Fonzo, who was not part of the research.
“But it is high, and it underscores that this is not a treatment without certain risks, even though it was good that they were primarily mild in severity,” he added.
A ‘stepping stone’ to FDA approval?
The use of tools to measure disability in addition to symptoms of depression severity is a strength of the study, Dr. Fonzo added. The use of an active comparator and the 6-week follow-up also offer something new over previous studies.
Despite the longer follow up, questions remain about the durability of response, something only a longer study could answer, Dr. Fonzo said. The small and homogeneous sample-size are also a concern. Nearly 90% of participants were White, and more than half had an income of $75,000 a year or higher.
“It’s another stepping stone in the process to FDA approval, but the next step in that process would be much larger phase 3 trials that would have much larger samples, a longer follow-up, and hopefully have a more inclusive swath of the population,” Dr. Fonzo said.
But perhaps one of the most significant limitations is the use of niacin as an active comparator, said Caleb Alexander, MD, codirector of the Center for Drug Safety and Effectiveness at Johns Hopkins University in Baltimore.
The use of an agent that doesn’t produce effects similar to those expected from a psychedelic introduced the potential for functional unblinding, Dr. Alexander said. Investigators did not ask participants to guess whether they received psilocybin or niacin, so the quality of the blinding was not assessed in the study.
“We’d like to see the use of [an] active comparator that might have a chance of obscuring to people as to whether they’ve been randomized to the treatment arm or control arm,” said Dr. Alexander, who wasn’t involved in the study. “Why not use a benzodiazepine or another drug that produces a transient euphoria that would better obscure whether or not people were receiving the psilocybin?”
The authors of an accompanying editorial shared these concerns, also noting that the study included “a significant number of patients who did not respond to therapy.”
“It is important to analyze and understand adverse outcomes in psychedelic trials and conduct longitudinal studies to determine how sustained the effects will be and what may initiate a recrudescence of symptoms,” write Rachel Yehuda, PhD, and Amy Lehrner, PhD, both of the Peters VA Medical Center and Icahn School of Medicine at Mount Sinai, New York.
“Future studies will help identify who is most likely to benefit from psychedelics, whether booster or repeated treatment is safe and beneficial, and what the optimal dose and therapeutic frameworks are.”
A long-term follow-up of the current trial was terminated last year because of low enrollment. The spokesperson with Usona Institute did not respond to questions about that study, and the institute’s statement only added that preparations are underway to launch another study that “will provide additional safety and efficacy data to support submission of a new drug application to the FDA.”
Usona published its manufacturing process that it used to synthesize psilocybin in an open-access journal and signed a statement on “open science and open praxis” with psilocybin and similar substances, which appears on their website. That statement was signed by 31 research and service organizations around the world and nearly 150 scientists, scholars, and practitioners.
The study was funded by Usona Institute. Dr. Raison reported receiving personal fees from Usona Institute and grants to Usona Institute from Dr. Bronner’s All-One, Fournier Family Foundation, Good Ventures, Steven and Alexandra Cohen Foundation, Tiny Blue Dot Foundation, Turnbull Family Foundation, and William A. Linton during the conduct of the study; and personal fees from Novartis, Sage/Biogen, Emory Healthcare, and Vail Health outside the submitted work. Dr. Fonzo and Dr. Alexander report no relevant financial relationships. Dr. Yehuda reports receiving nonfinancial support from the Multidisciplinary Association for Psychedelic Studies Public Benefit (MAPS PBC) and grants from COMPASS Pathways. Dr. Lehrner is an investigator on trials sponsored by MAPS PBC and COMPASS Pathways.
A version of this article first appeared on Medscape.com.
The randomized, phase 2 trial was conducted at 11 sites across the United States and is the latest to demonstrate the psychedelic drug’s potential as a treatment for depression.
The project was funded by Usona Institute, a nonprofit medical research organization based in Madison, Wisc. The institute issued a press statement, but researchers did not comment further on the findings.
“As the largest and most rigorous study conducted across a wide spectrum of individuals with major depressive disorder, the results show promise for all people struggling with this condition,” lead author Charles Raison, MD, director of clinical and translational research at Usona, said in the statement.
The 34 coauthors on the study are affiliated with public universities, research centers, and private companies. Eight of the investigators are identified as employees of Usona Institute.
Declining further comment, an institute spokesperson told this news organization that, “Usona has chosen the approach of no interviews, and this applies for all coauthors.”
The findings were published online in JAMA.
Largest study to date
Usona’s investigational psilocybin drug has been granted a breakthrough designation by the Food and Drug Administration, a process designed to speed drug development and review.
Previous smaller studies have suggested a rapid antidepressant response with psilocybin, but they have been small, unblinded, and have had short duration of follow-up, they write. This randomized, double-blind, phase 2 clinical trial is the largest study of psilocybin for depression to date, the researchers note.
It included 104 adults aged 21-65 years with MDD who had a current depressive episode of at least 60 days and a Montgomery-Åsberg Depression Rating Scale (MADRS) total score of 28 or more at baseline.
Participants had to be free of psychedelic drugs for at least 5 years, have had no active suicidal ideation or suicidal behavior in the prior 12 months, no personal or first-degree family history of psychosis or mania, and no history of moderate/severe alcohol or drug use disorder.
Before the study, participants had a 7- to 35-day screening period for psychiatric medication tapering, underwent baseline assessments, and received 6-8 hours of preparation with two facilitators who would be with them during dosing.
Dosing occurred within 7 days of baseline assessments. During the 6- to 8-hour session, participants received either a single 25-mg oral dose of psilocybin or 100-mg dose of niacin. One participant randomly assigned to receive psilocybin received the incorrect treatment, resulting in 50 participants receiving psilocybin and 54 receiving niacin.
Participants returned the next day, the next week, and then every 2 weeks for assessments, for a follow-up of 6 weeks.
Psychosocial support
Participants who received psilocybin reported significantly greater improvements in MDD symptoms, compared with those who received niacin. MADRS scores – a scale from 0 to 60 where higher scores indicate more severe depression – showed greater reductions with treatment vs. placebo at 8 days (mean difference, −12.0; 95% confidence interval, −16.6 to −7.4; P < .001), and at day 43 (mean difference, −12.3; 95% CI, −17.5 to −7.2; P < .001).
More participants receiving psilocybin had sustained depressive symptom response (42% vs. 11%; P = .002) and more improvement in the Sheehan Disability Scale score, which measures functional disability, 43 days after treatment (P < .001).
The effects persisted through the end of the study, although the differences between groups were no longer significant by week 6.
“This is another exciting piece of evidence that adds to the current literature regarding the potential efficacy of psilocybin for the treatment of mental health conditions, particularly depression,” said Greg Fonzo, MD, codirector of the Center for Psychedelic Research and Therapy at the University of Texas at Austin, who commented on the findings.
Significantly more people in the psilocybin group reported at least one treatment-related adverse event (AE, 82% vs. 44%), although most were mild to moderate. Headache and nausea were the most common side effects and most resolved within 1 day of dosing.
While those numbers are high, Dr. Fonzo said they’re not out of line with AEs reported in other studies.
“Particularly with the types of adverse events reported here, like headache and nausea, those are things you would typically expect to see in this treatment,” said Dr. Fonzo, who was not part of the research.
“But it is high, and it underscores that this is not a treatment without certain risks, even though it was good that they were primarily mild in severity,” he added.
A ‘stepping stone’ to FDA approval?
The use of tools to measure disability in addition to symptoms of depression severity is a strength of the study, Dr. Fonzo added. The use of an active comparator and the 6-week follow-up also offer something new over previous studies.
Despite the longer follow up, questions remain about the durability of response, something only a longer study could answer, Dr. Fonzo said. The small and homogeneous sample-size are also a concern. Nearly 90% of participants were White, and more than half had an income of $75,000 a year or higher.
“It’s another stepping stone in the process to FDA approval, but the next step in that process would be much larger phase 3 trials that would have much larger samples, a longer follow-up, and hopefully have a more inclusive swath of the population,” Dr. Fonzo said.
But perhaps one of the most significant limitations is the use of niacin as an active comparator, said Caleb Alexander, MD, codirector of the Center for Drug Safety and Effectiveness at Johns Hopkins University in Baltimore.
The use of an agent that doesn’t produce effects similar to those expected from a psychedelic introduced the potential for functional unblinding, Dr. Alexander said. Investigators did not ask participants to guess whether they received psilocybin or niacin, so the quality of the blinding was not assessed in the study.
“We’d like to see the use of [an] active comparator that might have a chance of obscuring to people as to whether they’ve been randomized to the treatment arm or control arm,” said Dr. Alexander, who wasn’t involved in the study. “Why not use a benzodiazepine or another drug that produces a transient euphoria that would better obscure whether or not people were receiving the psilocybin?”
The authors of an accompanying editorial shared these concerns, also noting that the study included “a significant number of patients who did not respond to therapy.”
“It is important to analyze and understand adverse outcomes in psychedelic trials and conduct longitudinal studies to determine how sustained the effects will be and what may initiate a recrudescence of symptoms,” write Rachel Yehuda, PhD, and Amy Lehrner, PhD, both of the Peters VA Medical Center and Icahn School of Medicine at Mount Sinai, New York.
“Future studies will help identify who is most likely to benefit from psychedelics, whether booster or repeated treatment is safe and beneficial, and what the optimal dose and therapeutic frameworks are.”
A long-term follow-up of the current trial was terminated last year because of low enrollment. The spokesperson with Usona Institute did not respond to questions about that study, and the institute’s statement only added that preparations are underway to launch another study that “will provide additional safety and efficacy data to support submission of a new drug application to the FDA.”
Usona published its manufacturing process that it used to synthesize psilocybin in an open-access journal and signed a statement on “open science and open praxis” with psilocybin and similar substances, which appears on their website. That statement was signed by 31 research and service organizations around the world and nearly 150 scientists, scholars, and practitioners.
The study was funded by Usona Institute. Dr. Raison reported receiving personal fees from Usona Institute and grants to Usona Institute from Dr. Bronner’s All-One, Fournier Family Foundation, Good Ventures, Steven and Alexandra Cohen Foundation, Tiny Blue Dot Foundation, Turnbull Family Foundation, and William A. Linton during the conduct of the study; and personal fees from Novartis, Sage/Biogen, Emory Healthcare, and Vail Health outside the submitted work. Dr. Fonzo and Dr. Alexander report no relevant financial relationships. Dr. Yehuda reports receiving nonfinancial support from the Multidisciplinary Association for Psychedelic Studies Public Benefit (MAPS PBC) and grants from COMPASS Pathways. Dr. Lehrner is an investigator on trials sponsored by MAPS PBC and COMPASS Pathways.
A version of this article first appeared on Medscape.com.
The randomized, phase 2 trial was conducted at 11 sites across the United States and is the latest to demonstrate the psychedelic drug’s potential as a treatment for depression.
The project was funded by Usona Institute, a nonprofit medical research organization based in Madison, Wisc. The institute issued a press statement, but researchers did not comment further on the findings.
“As the largest and most rigorous study conducted across a wide spectrum of individuals with major depressive disorder, the results show promise for all people struggling with this condition,” lead author Charles Raison, MD, director of clinical and translational research at Usona, said in the statement.
The 34 coauthors on the study are affiliated with public universities, research centers, and private companies. Eight of the investigators are identified as employees of Usona Institute.
Declining further comment, an institute spokesperson told this news organization that, “Usona has chosen the approach of no interviews, and this applies for all coauthors.”
The findings were published online in JAMA.
Largest study to date
Usona’s investigational psilocybin drug has been granted a breakthrough designation by the Food and Drug Administration, a process designed to speed drug development and review.
Previous smaller studies have suggested a rapid antidepressant response with psilocybin, but they have been small, unblinded, and have had short duration of follow-up, they write. This randomized, double-blind, phase 2 clinical trial is the largest study of psilocybin for depression to date, the researchers note.
It included 104 adults aged 21-65 years with MDD who had a current depressive episode of at least 60 days and a Montgomery-Åsberg Depression Rating Scale (MADRS) total score of 28 or more at baseline.
Participants had to be free of psychedelic drugs for at least 5 years, have had no active suicidal ideation or suicidal behavior in the prior 12 months, no personal or first-degree family history of psychosis or mania, and no history of moderate/severe alcohol or drug use disorder.
Before the study, participants had a 7- to 35-day screening period for psychiatric medication tapering, underwent baseline assessments, and received 6-8 hours of preparation with two facilitators who would be with them during dosing.
Dosing occurred within 7 days of baseline assessments. During the 6- to 8-hour session, participants received either a single 25-mg oral dose of psilocybin or 100-mg dose of niacin. One participant randomly assigned to receive psilocybin received the incorrect treatment, resulting in 50 participants receiving psilocybin and 54 receiving niacin.
Participants returned the next day, the next week, and then every 2 weeks for assessments, for a follow-up of 6 weeks.
Psychosocial support
Participants who received psilocybin reported significantly greater improvements in MDD symptoms, compared with those who received niacin. MADRS scores – a scale from 0 to 60 where higher scores indicate more severe depression – showed greater reductions with treatment vs. placebo at 8 days (mean difference, −12.0; 95% confidence interval, −16.6 to −7.4; P < .001), and at day 43 (mean difference, −12.3; 95% CI, −17.5 to −7.2; P < .001).
More participants receiving psilocybin had sustained depressive symptom response (42% vs. 11%; P = .002) and more improvement in the Sheehan Disability Scale score, which measures functional disability, 43 days after treatment (P < .001).
The effects persisted through the end of the study, although the differences between groups were no longer significant by week 6.
“This is another exciting piece of evidence that adds to the current literature regarding the potential efficacy of psilocybin for the treatment of mental health conditions, particularly depression,” said Greg Fonzo, MD, codirector of the Center for Psychedelic Research and Therapy at the University of Texas at Austin, who commented on the findings.
Significantly more people in the psilocybin group reported at least one treatment-related adverse event (AE, 82% vs. 44%), although most were mild to moderate. Headache and nausea were the most common side effects and most resolved within 1 day of dosing.
While those numbers are high, Dr. Fonzo said they’re not out of line with AEs reported in other studies.
“Particularly with the types of adverse events reported here, like headache and nausea, those are things you would typically expect to see in this treatment,” said Dr. Fonzo, who was not part of the research.
“But it is high, and it underscores that this is not a treatment without certain risks, even though it was good that they were primarily mild in severity,” he added.
A ‘stepping stone’ to FDA approval?
The use of tools to measure disability in addition to symptoms of depression severity is a strength of the study, Dr. Fonzo added. The use of an active comparator and the 6-week follow-up also offer something new over previous studies.
Despite the longer follow up, questions remain about the durability of response, something only a longer study could answer, Dr. Fonzo said. The small and homogeneous sample-size are also a concern. Nearly 90% of participants were White, and more than half had an income of $75,000 a year or higher.
“It’s another stepping stone in the process to FDA approval, but the next step in that process would be much larger phase 3 trials that would have much larger samples, a longer follow-up, and hopefully have a more inclusive swath of the population,” Dr. Fonzo said.
But perhaps one of the most significant limitations is the use of niacin as an active comparator, said Caleb Alexander, MD, codirector of the Center for Drug Safety and Effectiveness at Johns Hopkins University in Baltimore.
The use of an agent that doesn’t produce effects similar to those expected from a psychedelic introduced the potential for functional unblinding, Dr. Alexander said. Investigators did not ask participants to guess whether they received psilocybin or niacin, so the quality of the blinding was not assessed in the study.
“We’d like to see the use of [an] active comparator that might have a chance of obscuring to people as to whether they’ve been randomized to the treatment arm or control arm,” said Dr. Alexander, who wasn’t involved in the study. “Why not use a benzodiazepine or another drug that produces a transient euphoria that would better obscure whether or not people were receiving the psilocybin?”
The authors of an accompanying editorial shared these concerns, also noting that the study included “a significant number of patients who did not respond to therapy.”
“It is important to analyze and understand adverse outcomes in psychedelic trials and conduct longitudinal studies to determine how sustained the effects will be and what may initiate a recrudescence of symptoms,” write Rachel Yehuda, PhD, and Amy Lehrner, PhD, both of the Peters VA Medical Center and Icahn School of Medicine at Mount Sinai, New York.
“Future studies will help identify who is most likely to benefit from psychedelics, whether booster or repeated treatment is safe and beneficial, and what the optimal dose and therapeutic frameworks are.”
A long-term follow-up of the current trial was terminated last year because of low enrollment. The spokesperson with Usona Institute did not respond to questions about that study, and the institute’s statement only added that preparations are underway to launch another study that “will provide additional safety and efficacy data to support submission of a new drug application to the FDA.”
Usona published its manufacturing process that it used to synthesize psilocybin in an open-access journal and signed a statement on “open science and open praxis” with psilocybin and similar substances, which appears on their website. That statement was signed by 31 research and service organizations around the world and nearly 150 scientists, scholars, and practitioners.
The study was funded by Usona Institute. Dr. Raison reported receiving personal fees from Usona Institute and grants to Usona Institute from Dr. Bronner’s All-One, Fournier Family Foundation, Good Ventures, Steven and Alexandra Cohen Foundation, Tiny Blue Dot Foundation, Turnbull Family Foundation, and William A. Linton during the conduct of the study; and personal fees from Novartis, Sage/Biogen, Emory Healthcare, and Vail Health outside the submitted work. Dr. Fonzo and Dr. Alexander report no relevant financial relationships. Dr. Yehuda reports receiving nonfinancial support from the Multidisciplinary Association for Psychedelic Studies Public Benefit (MAPS PBC) and grants from COMPASS Pathways. Dr. Lehrner is an investigator on trials sponsored by MAPS PBC and COMPASS Pathways.
A version of this article first appeared on Medscape.com.
FROM JAMA
Suicidal behavior tied to increased all-cause mortality in MDD
Investigators studied close to 143,000 patients, encompassing more than 150,000 MDD episodes. Episodes of depression with suicidal behavior (MDD-SB) were compared to MDD episodes without suicidal behavior (MDD-non-SB).
Suicidal behavior was associated with a 2.6-fold higher rate of all-cause mortality, as well as considerably higher health care resource utilization (HCRU) and work loss, compared with matched controls.
Patients with depression who had attempted suicide were younger and more commonly suffering from other psychiatric comorbidities, such as anxiety and addiction. Important risk factors for suicidal acts within a year after the onset of a depressive episode were previous suicide attempts, substance use disorder, anxiety, and sleeping disorders.
“The findings tell us that the care provided for this particular group needs to be developed,” lead author Johan Lundberg, MD, PhD, adjunct professor in psychiatry and senior physician in psychiatry, Karolinska Institute, Stockholm, told this news organization.
“The take-home message is that, when treating patients with increased risk of suicidal behavior, one should offer treatments with this in mind,” said Dr. Lundberg, also the head of the section of mood disorders, Northern Stockholm Psychiatry Clinic. “One possible option is lithium augmentation.”
The study was published online in JAMA Psychiatry.
Identifying subgroups
Depression is associated with increased all-cause mortality, the authors write. Suicidal behavior and previous suicide attempts are known to increase the risk of suicide-associated mortality, with up to 13% of patients with nonfatal suicide attempts dying of suicide at a later time.
Previous studies investigating the association between suicidal behavior and mortality have been limited by nonrandom sampling due to “nonuniversal access to health care and/or exclusion of primary care data,” they state.
For this reason, it’s not established to what extent these estimates actually represent patients with MDD as a whole, or to what extent suicidal behavior is a risk factor for all-cause mortality.
“We think there is a need to identify subgroups within the very large group of individuals with MDD in order to improve treatment outcomes,” Dr. Lundberg said.
To do so, the researchers turned to data from the Stockholm MDD Cohort (SMC), which comprises all patients diagnosed with MDD in any health care setting in the regions of Stockholm from 2010 to 2018. They identified 5 years of recorded MDD episodes (n = 158,169) in patients aged 18 years and older (n = 145,577). A single patient could contribute more than one episode.
At index, MDD-SB patients (n = 2,219; mean age, 41 years) were matched with MDD-non-SB patients (9,574; mean age, 41 years) based on age, sex, year of MDD diagnosis, and socioeconomic status. In total, 2,219 episodes (63.2% in women, 36.8% in men) were compared to 11,109 episodes (63.4% in women, 36.6% in men), respectively.
Enhanced monitoring, optimized treatment
The median time from the start of the episode until the first suicidal behavior was 165 days.
The all-cause mortality rate in the MDD-SB and MDD-non-SB groups was 2.5 per 100 person-years vs. 1 per 100 person-years, respectively (based on 466 deaths), corresponding to a hazard ratio of 2.62 (95% confidence interval, 2.15-3.20).
Patients in the MDD-SB group were younger, were more frequently diagnosed while in specialized care, and had sustained more work loss than their counterparts in the MDD-non-SB group. They also showed a gradual increase in the prevalence of comorbid conditions from about 12 months before index, with this increase being “most pronounced” for anxiety, stress, substance use, and personality disorders.
MDD-SB episodes were associated with higher HCRU and more work loss, compared with MDD-non-SB episodes.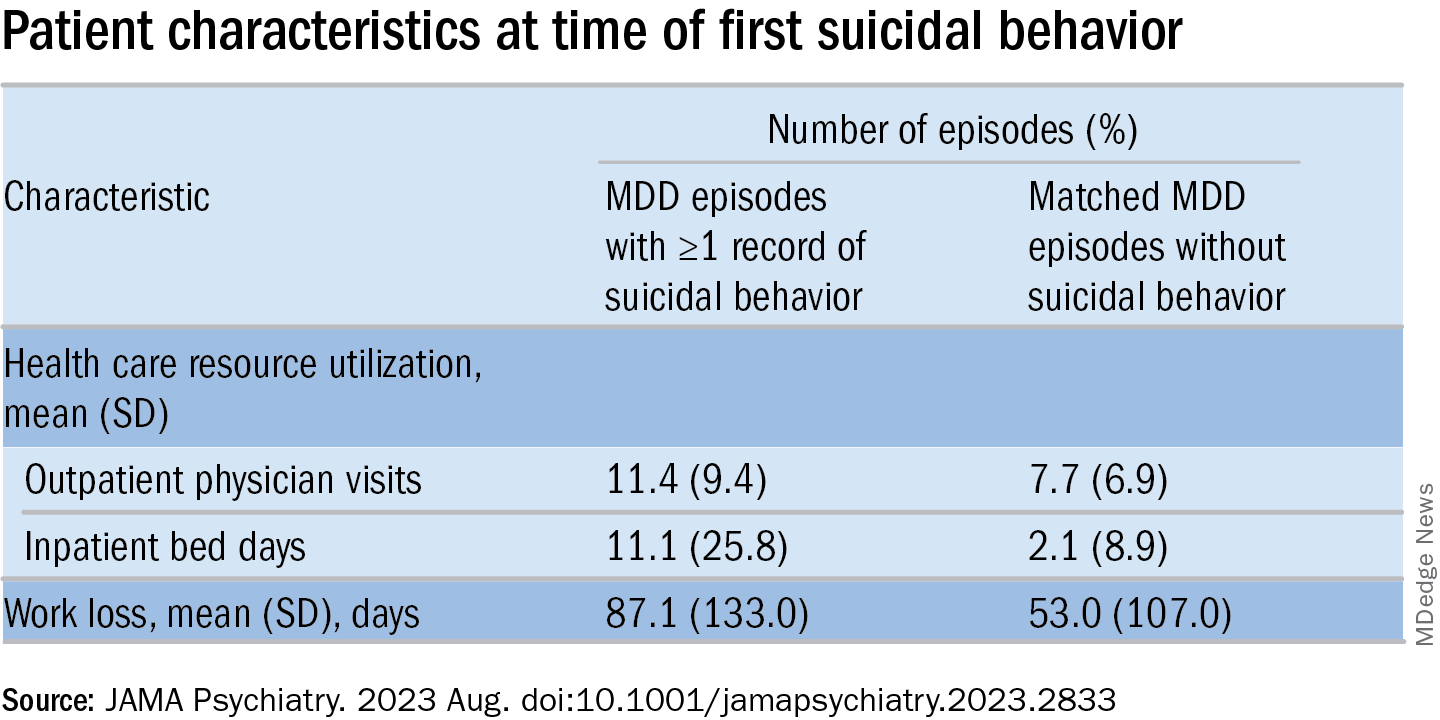
The researchers calculated a risk score for factors associated with suicidal behavior within 1 year after the start of an MDD episode (outcome). The two most important risk factors for suicidal behavior were a history of suicidal behavior together with age, which had a “U-shaped association” with the outcome, they write, with individuals younger than age 20 and older than age 70 having the highest risks.
The final risk score included additional factors that increased the risk of the outcome (in descending order): history of substance use, history of sleep disorders, health care level in which MDD was diagnosed, history of antidepressant use, and history of anxiety disorders.
These results “indicate that patients at risk for suicidal behavior can be identified at an early stage to allow for enhanced monitoring and optimized treatment with the goal of preventing suicidal behavior and reducing mortality,” the authors state.
The specific causes of death weren’t analyzed in this particular paper, Dr. Lundberg noted. A previous study conducted by the same group found the risk of death was doubled in MDD patients, compared with controls.
“We don’t speculate about which causes other than suicide might explain the difference” and account for the increased mortality risk, he said. “This should be studied in future projects.”
Complicated family of destructive behaviors
In a comment, Russell Copelan, MD, a former emergency department psychiatrist at the University of Colorado Affiliated Hospital and currently an expert consultant to the American Association of Suicidology, said a take-home message of the study is that suicide is “a complex and complicated family of destructive behaviors.”
The findings “should not suggest a wait-and-see clinical approach,” warned Dr. Copelan, who wasn’t involved with the study.
Underrecognized or misdiagnosed anxiety, agitation, and insomnia may be “barriers to remission and treatment response,” he noted.
Dr. Copelan, who is also the founder and CEO of eMed Logic, which offers assessment tools for suicide and violence, encouraged clinicians “not to minimize the proportion of patients who experience anxiety, agitation, and insomnia in response to what some may consider a personal misfortune, such as interpersonal, employment, or financial crisis.”
A version of this article first appeared on Medscape.com.
Investigators studied close to 143,000 patients, encompassing more than 150,000 MDD episodes. Episodes of depression with suicidal behavior (MDD-SB) were compared to MDD episodes without suicidal behavior (MDD-non-SB).
Suicidal behavior was associated with a 2.6-fold higher rate of all-cause mortality, as well as considerably higher health care resource utilization (HCRU) and work loss, compared with matched controls.
Patients with depression who had attempted suicide were younger and more commonly suffering from other psychiatric comorbidities, such as anxiety and addiction. Important risk factors for suicidal acts within a year after the onset of a depressive episode were previous suicide attempts, substance use disorder, anxiety, and sleeping disorders.
“The findings tell us that the care provided for this particular group needs to be developed,” lead author Johan Lundberg, MD, PhD, adjunct professor in psychiatry and senior physician in psychiatry, Karolinska Institute, Stockholm, told this news organization.
“The take-home message is that, when treating patients with increased risk of suicidal behavior, one should offer treatments with this in mind,” said Dr. Lundberg, also the head of the section of mood disorders, Northern Stockholm Psychiatry Clinic. “One possible option is lithium augmentation.”
The study was published online in JAMA Psychiatry.
Identifying subgroups
Depression is associated with increased all-cause mortality, the authors write. Suicidal behavior and previous suicide attempts are known to increase the risk of suicide-associated mortality, with up to 13% of patients with nonfatal suicide attempts dying of suicide at a later time.
Previous studies investigating the association between suicidal behavior and mortality have been limited by nonrandom sampling due to “nonuniversal access to health care and/or exclusion of primary care data,” they state.
For this reason, it’s not established to what extent these estimates actually represent patients with MDD as a whole, or to what extent suicidal behavior is a risk factor for all-cause mortality.
“We think there is a need to identify subgroups within the very large group of individuals with MDD in order to improve treatment outcomes,” Dr. Lundberg said.
To do so, the researchers turned to data from the Stockholm MDD Cohort (SMC), which comprises all patients diagnosed with MDD in any health care setting in the regions of Stockholm from 2010 to 2018. They identified 5 years of recorded MDD episodes (n = 158,169) in patients aged 18 years and older (n = 145,577). A single patient could contribute more than one episode.
At index, MDD-SB patients (n = 2,219; mean age, 41 years) were matched with MDD-non-SB patients (9,574; mean age, 41 years) based on age, sex, year of MDD diagnosis, and socioeconomic status. In total, 2,219 episodes (63.2% in women, 36.8% in men) were compared to 11,109 episodes (63.4% in women, 36.6% in men), respectively.
Enhanced monitoring, optimized treatment
The median time from the start of the episode until the first suicidal behavior was 165 days.
The all-cause mortality rate in the MDD-SB and MDD-non-SB groups was 2.5 per 100 person-years vs. 1 per 100 person-years, respectively (based on 466 deaths), corresponding to a hazard ratio of 2.62 (95% confidence interval, 2.15-3.20).
Patients in the MDD-SB group were younger, were more frequently diagnosed while in specialized care, and had sustained more work loss than their counterparts in the MDD-non-SB group. They also showed a gradual increase in the prevalence of comorbid conditions from about 12 months before index, with this increase being “most pronounced” for anxiety, stress, substance use, and personality disorders.
MDD-SB episodes were associated with higher HCRU and more work loss, compared with MDD-non-SB episodes.
The researchers calculated a risk score for factors associated with suicidal behavior within 1 year after the start of an MDD episode (outcome). The two most important risk factors for suicidal behavior were a history of suicidal behavior together with age, which had a “U-shaped association” with the outcome, they write, with individuals younger than age 20 and older than age 70 having the highest risks.
The final risk score included additional factors that increased the risk of the outcome (in descending order): history of substance use, history of sleep disorders, health care level in which MDD was diagnosed, history of antidepressant use, and history of anxiety disorders.
These results “indicate that patients at risk for suicidal behavior can be identified at an early stage to allow for enhanced monitoring and optimized treatment with the goal of preventing suicidal behavior and reducing mortality,” the authors state.
The specific causes of death weren’t analyzed in this particular paper, Dr. Lundberg noted. A previous study conducted by the same group found the risk of death was doubled in MDD patients, compared with controls.
“We don’t speculate about which causes other than suicide might explain the difference” and account for the increased mortality risk, he said. “This should be studied in future projects.”
Complicated family of destructive behaviors
In a comment, Russell Copelan, MD, a former emergency department psychiatrist at the University of Colorado Affiliated Hospital and currently an expert consultant to the American Association of Suicidology, said a take-home message of the study is that suicide is “a complex and complicated family of destructive behaviors.”
The findings “should not suggest a wait-and-see clinical approach,” warned Dr. Copelan, who wasn’t involved with the study.
Underrecognized or misdiagnosed anxiety, agitation, and insomnia may be “barriers to remission and treatment response,” he noted.
Dr. Copelan, who is also the founder and CEO of eMed Logic, which offers assessment tools for suicide and violence, encouraged clinicians “not to minimize the proportion of patients who experience anxiety, agitation, and insomnia in response to what some may consider a personal misfortune, such as interpersonal, employment, or financial crisis.”
A version of this article first appeared on Medscape.com.
Investigators studied close to 143,000 patients, encompassing more than 150,000 MDD episodes. Episodes of depression with suicidal behavior (MDD-SB) were compared to MDD episodes without suicidal behavior (MDD-non-SB).
Suicidal behavior was associated with a 2.6-fold higher rate of all-cause mortality, as well as considerably higher health care resource utilization (HCRU) and work loss, compared with matched controls.
Patients with depression who had attempted suicide were younger and more commonly suffering from other psychiatric comorbidities, such as anxiety and addiction. Important risk factors for suicidal acts within a year after the onset of a depressive episode were previous suicide attempts, substance use disorder, anxiety, and sleeping disorders.
“The findings tell us that the care provided for this particular group needs to be developed,” lead author Johan Lundberg, MD, PhD, adjunct professor in psychiatry and senior physician in psychiatry, Karolinska Institute, Stockholm, told this news organization.
“The take-home message is that, when treating patients with increased risk of suicidal behavior, one should offer treatments with this in mind,” said Dr. Lundberg, also the head of the section of mood disorders, Northern Stockholm Psychiatry Clinic. “One possible option is lithium augmentation.”
The study was published online in JAMA Psychiatry.
Identifying subgroups
Depression is associated with increased all-cause mortality, the authors write. Suicidal behavior and previous suicide attempts are known to increase the risk of suicide-associated mortality, with up to 13% of patients with nonfatal suicide attempts dying of suicide at a later time.
Previous studies investigating the association between suicidal behavior and mortality have been limited by nonrandom sampling due to “nonuniversal access to health care and/or exclusion of primary care data,” they state.
For this reason, it’s not established to what extent these estimates actually represent patients with MDD as a whole, or to what extent suicidal behavior is a risk factor for all-cause mortality.
“We think there is a need to identify subgroups within the very large group of individuals with MDD in order to improve treatment outcomes,” Dr. Lundberg said.
To do so, the researchers turned to data from the Stockholm MDD Cohort (SMC), which comprises all patients diagnosed with MDD in any health care setting in the regions of Stockholm from 2010 to 2018. They identified 5 years of recorded MDD episodes (n = 158,169) in patients aged 18 years and older (n = 145,577). A single patient could contribute more than one episode.
At index, MDD-SB patients (n = 2,219; mean age, 41 years) were matched with MDD-non-SB patients (9,574; mean age, 41 years) based on age, sex, year of MDD diagnosis, and socioeconomic status. In total, 2,219 episodes (63.2% in women, 36.8% in men) were compared to 11,109 episodes (63.4% in women, 36.6% in men), respectively.
Enhanced monitoring, optimized treatment
The median time from the start of the episode until the first suicidal behavior was 165 days.
The all-cause mortality rate in the MDD-SB and MDD-non-SB groups was 2.5 per 100 person-years vs. 1 per 100 person-years, respectively (based on 466 deaths), corresponding to a hazard ratio of 2.62 (95% confidence interval, 2.15-3.20).
Patients in the MDD-SB group were younger, were more frequently diagnosed while in specialized care, and had sustained more work loss than their counterparts in the MDD-non-SB group. They also showed a gradual increase in the prevalence of comorbid conditions from about 12 months before index, with this increase being “most pronounced” for anxiety, stress, substance use, and personality disorders.
MDD-SB episodes were associated with higher HCRU and more work loss, compared with MDD-non-SB episodes.
The researchers calculated a risk score for factors associated with suicidal behavior within 1 year after the start of an MDD episode (outcome). The two most important risk factors for suicidal behavior were a history of suicidal behavior together with age, which had a “U-shaped association” with the outcome, they write, with individuals younger than age 20 and older than age 70 having the highest risks.
The final risk score included additional factors that increased the risk of the outcome (in descending order): history of substance use, history of sleep disorders, health care level in which MDD was diagnosed, history of antidepressant use, and history of anxiety disorders.
These results “indicate that patients at risk for suicidal behavior can be identified at an early stage to allow for enhanced monitoring and optimized treatment with the goal of preventing suicidal behavior and reducing mortality,” the authors state.
The specific causes of death weren’t analyzed in this particular paper, Dr. Lundberg noted. A previous study conducted by the same group found the risk of death was doubled in MDD patients, compared with controls.
“We don’t speculate about which causes other than suicide might explain the difference” and account for the increased mortality risk, he said. “This should be studied in future projects.”
Complicated family of destructive behaviors
In a comment, Russell Copelan, MD, a former emergency department psychiatrist at the University of Colorado Affiliated Hospital and currently an expert consultant to the American Association of Suicidology, said a take-home message of the study is that suicide is “a complex and complicated family of destructive behaviors.”
The findings “should not suggest a wait-and-see clinical approach,” warned Dr. Copelan, who wasn’t involved with the study.
Underrecognized or misdiagnosed anxiety, agitation, and insomnia may be “barriers to remission and treatment response,” he noted.
Dr. Copelan, who is also the founder and CEO of eMed Logic, which offers assessment tools for suicide and violence, encouraged clinicians “not to minimize the proportion of patients who experience anxiety, agitation, and insomnia in response to what some may consider a personal misfortune, such as interpersonal, employment, or financial crisis.”
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Screen bipolar patients for eating disorders
Previous research of bipolar disorder (BD) shows a high rate of comorbidities with other psychiatric disorders, including eating disorders (EDs), Valentin Flaudias, PhD, of Nantes (France) University and colleagues wrote.
“There is growing evidence that, compared with individuals with BD alone, individuals with both BD and EDs have a more severe clinical profile, including increased mood instability, alcohol use disorders, anxiety disorders, more depressive episodes, more rapid cycling, increased suicidality, and poorer response to medication,” but studies of BD type-specific ED prevalence have been inconsistent, they said.
In a study published in the Journal of Affective Disorders, the researchers reviewed data from 2,929 outpatients who underwent assessments for BD at 1 of 12 psychiatric centers in France. Of these, 1,505 met criteria for type I and 1,424 met criteria for type II. The post hoc analysis included identification of lifetime prevalence of ED. Diagnosis was based on the DSM-4-TR and the researchers considered three ED types: anorexia nervosa (AN), bulimia nervosa (BN), and binge-eating disorder (BED). Subtypes of BD were type I and type II. DSM not otherwise specified diagnoses for BD and EDs were excluded. The mean age of the participants was 40.5 years, and 61% were women.
A total of 479 individuals met criteria for comorbid EDs (16.4%). ED prevalence was significantly higher in BD type II patients than in BD type I patients (20.6 % vs. 12.4 %, P < .001). The overall breakdown according to ED subtype was 30% for AN, 13% for BN, and 56% for BED. The researchers found no significant differences in patients with AN, BN, or BED according to BD subtype.
In a multivariate analysis, BD patients with ED were more likely than those without ED to be women (77% vs. 55%), especially those with AN (95% vs. 82%).
BD patients with ED also tended to be younger than those without ED (37 years vs. 41 years) and reported more frequent suicide attempts (50% vs. 35%). Younger age and more frequent suicide attempts were further significant among BD patients with AN, compared with those with BED, but BD patients with BED reported higher levels of childhood trauma.
BD patients with ED also reported higher levels of depressive symptoms than those without ED, although history of psychosis was less frequent among BD patients with AN and BED compared with BD patients without EDs.
Overall, “after controlling for other variables, the independent factors differentiating BD patients with versus without ED were primarily younger age, female gender, abnormal BMI, increased affective lability and higher comorbidity with anxiety disorders,” the researchers wrote. In addition, presence of EDs except for AN was associated with decreased current functioning.
The findings were limited by several factors including the cross-sectional design, lack of a control group of non-BD individuals, and the consideration of ED over a lifetime, and small number of BN cases, the researchers noted.
However, the results suggest a high prevalence of ED in BD patients and highlight the need to screen BD patients for ED and provide integrated care. More research is needed to explore the evolution of the two conditions as comorbidities and to examine subtypes and of both conditions and their interactions, they concluded.
The study was supported by the FondaMental Foundation, French National Institute for Health and Medical Research, Public Hospitals of Paris, and the French National Research Agency’s Investment for the Future program. The researchers had no financial conflicts to disclose.
Previous research of bipolar disorder (BD) shows a high rate of comorbidities with other psychiatric disorders, including eating disorders (EDs), Valentin Flaudias, PhD, of Nantes (France) University and colleagues wrote.
“There is growing evidence that, compared with individuals with BD alone, individuals with both BD and EDs have a more severe clinical profile, including increased mood instability, alcohol use disorders, anxiety disorders, more depressive episodes, more rapid cycling, increased suicidality, and poorer response to medication,” but studies of BD type-specific ED prevalence have been inconsistent, they said.
In a study published in the Journal of Affective Disorders, the researchers reviewed data from 2,929 outpatients who underwent assessments for BD at 1 of 12 psychiatric centers in France. Of these, 1,505 met criteria for type I and 1,424 met criteria for type II. The post hoc analysis included identification of lifetime prevalence of ED. Diagnosis was based on the DSM-4-TR and the researchers considered three ED types: anorexia nervosa (AN), bulimia nervosa (BN), and binge-eating disorder (BED). Subtypes of BD were type I and type II. DSM not otherwise specified diagnoses for BD and EDs were excluded. The mean age of the participants was 40.5 years, and 61% were women.
A total of 479 individuals met criteria for comorbid EDs (16.4%). ED prevalence was significantly higher in BD type II patients than in BD type I patients (20.6 % vs. 12.4 %, P < .001). The overall breakdown according to ED subtype was 30% for AN, 13% for BN, and 56% for BED. The researchers found no significant differences in patients with AN, BN, or BED according to BD subtype.
In a multivariate analysis, BD patients with ED were more likely than those without ED to be women (77% vs. 55%), especially those with AN (95% vs. 82%).
BD patients with ED also tended to be younger than those without ED (37 years vs. 41 years) and reported more frequent suicide attempts (50% vs. 35%). Younger age and more frequent suicide attempts were further significant among BD patients with AN, compared with those with BED, but BD patients with BED reported higher levels of childhood trauma.
BD patients with ED also reported higher levels of depressive symptoms than those without ED, although history of psychosis was less frequent among BD patients with AN and BED compared with BD patients without EDs.
Overall, “after controlling for other variables, the independent factors differentiating BD patients with versus without ED were primarily younger age, female gender, abnormal BMI, increased affective lability and higher comorbidity with anxiety disorders,” the researchers wrote. In addition, presence of EDs except for AN was associated with decreased current functioning.
The findings were limited by several factors including the cross-sectional design, lack of a control group of non-BD individuals, and the consideration of ED over a lifetime, and small number of BN cases, the researchers noted.
However, the results suggest a high prevalence of ED in BD patients and highlight the need to screen BD patients for ED and provide integrated care. More research is needed to explore the evolution of the two conditions as comorbidities and to examine subtypes and of both conditions and their interactions, they concluded.
The study was supported by the FondaMental Foundation, French National Institute for Health and Medical Research, Public Hospitals of Paris, and the French National Research Agency’s Investment for the Future program. The researchers had no financial conflicts to disclose.
Previous research of bipolar disorder (BD) shows a high rate of comorbidities with other psychiatric disorders, including eating disorders (EDs), Valentin Flaudias, PhD, of Nantes (France) University and colleagues wrote.
“There is growing evidence that, compared with individuals with BD alone, individuals with both BD and EDs have a more severe clinical profile, including increased mood instability, alcohol use disorders, anxiety disorders, more depressive episodes, more rapid cycling, increased suicidality, and poorer response to medication,” but studies of BD type-specific ED prevalence have been inconsistent, they said.
In a study published in the Journal of Affective Disorders, the researchers reviewed data from 2,929 outpatients who underwent assessments for BD at 1 of 12 psychiatric centers in France. Of these, 1,505 met criteria for type I and 1,424 met criteria for type II. The post hoc analysis included identification of lifetime prevalence of ED. Diagnosis was based on the DSM-4-TR and the researchers considered three ED types: anorexia nervosa (AN), bulimia nervosa (BN), and binge-eating disorder (BED). Subtypes of BD were type I and type II. DSM not otherwise specified diagnoses for BD and EDs were excluded. The mean age of the participants was 40.5 years, and 61% were women.
A total of 479 individuals met criteria for comorbid EDs (16.4%). ED prevalence was significantly higher in BD type II patients than in BD type I patients (20.6 % vs. 12.4 %, P < .001). The overall breakdown according to ED subtype was 30% for AN, 13% for BN, and 56% for BED. The researchers found no significant differences in patients with AN, BN, or BED according to BD subtype.
In a multivariate analysis, BD patients with ED were more likely than those without ED to be women (77% vs. 55%), especially those with AN (95% vs. 82%).
BD patients with ED also tended to be younger than those without ED (37 years vs. 41 years) and reported more frequent suicide attempts (50% vs. 35%). Younger age and more frequent suicide attempts were further significant among BD patients with AN, compared with those with BED, but BD patients with BED reported higher levels of childhood trauma.
BD patients with ED also reported higher levels of depressive symptoms than those without ED, although history of psychosis was less frequent among BD patients with AN and BED compared with BD patients without EDs.
Overall, “after controlling for other variables, the independent factors differentiating BD patients with versus without ED were primarily younger age, female gender, abnormal BMI, increased affective lability and higher comorbidity with anxiety disorders,” the researchers wrote. In addition, presence of EDs except for AN was associated with decreased current functioning.
The findings were limited by several factors including the cross-sectional design, lack of a control group of non-BD individuals, and the consideration of ED over a lifetime, and small number of BN cases, the researchers noted.
However, the results suggest a high prevalence of ED in BD patients and highlight the need to screen BD patients for ED and provide integrated care. More research is needed to explore the evolution of the two conditions as comorbidities and to examine subtypes and of both conditions and their interactions, they concluded.
The study was supported by the FondaMental Foundation, French National Institute for Health and Medical Research, Public Hospitals of Paris, and the French National Research Agency’s Investment for the Future program. The researchers had no financial conflicts to disclose.
FROM THE JOURNAL OF AFFECTIVE DISORDERS
Your patient bequeathed money to you: Can you accept it?
Michael Victoroff, MD, described the phone call he received from an attorney asking a thorny ethics question involving a patient’s gift to another physician. Dr. Victoroff, a past member of the ethics committee of the American Academy of Family Physicians, had definite thoughts about it.
“The attorney was representing the daughters of an elderly gentleman who had moved from the East Coast to Colorado to be closer to them,” said Dr. Victoroff, who teaches bioethics in the MBA program at the University of Denver and also practices at the University of Colorado School of Medicine.
“The father visited his new primary care physician frequently because he had multiple health issues.”
The patient was happy with the doctor’s medical care and over time that they developed a friendship. Dr. Victoroff emphasized that no sexual or romantic impropriety ever took place between the patient and his physician.
“But the social relationship went beyond the ordinary doctor-patient boundaries. The patient ultimately named the doctor as his health care proxy in the event that he became unable to make decisions regarding his care. He also mentioned he was going to leave her $100,000 in his will,” says Dr. Victoroff.
The physician did accept the role of proxy, “which raises a whole host of ethical issues,” says Dr. Victoroff. As it happened, she was never called upon to exercise that decision-making authority, since the patient died suddenly and was mentally competent at the time.
for her to accept such a substantial bequest from a patient, and they hired an attorney to contest the will.
No law against it
Dennis Hursh, attorney and managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, noted in an interview that, “the problem isn’t legal per se. Rather, the problem is an ethical one.”
Legally speaking, there’s no prohibition against receiving a bequest or other form of gift from a patient. “People are free to dispose of their estates in whatever way they see fit, and no law technically precludes a physician from accepting a bequest,” says Dr. Victoroff. “But this presupposes there is nothing improper going on, such as extortion, deception, coercion, or exercising undue influence.”
The issue of bequeathing money to their physician gained attention in a recent case that took place in Australia. Peter Alexakis, MD, received a whopping bequest of $24 million from a patient. The elderly patient had changed his will to name Dr. Alexakis as the sole beneficiary – after Dr. Alexakis had visited him at home 92 times during the preceding months. The original heirs filed a lawsuit in Australia’s Supreme Court against Dr. Alexakis, contesting the will.
The lawsuit was unsuccessful in court, but Dr. Alexakis was found guilty of malpractice by Australia’s Health Care Complaints Commission after being reported to the HCCC by the palliative care physicians who were treating the patient. They alleged that Dr. Alexakis had interfered with their care of the patient. The more serious allegation was that the doctor had engaged in a deliberate strategy to exploit the relationship for financial gain.
Dr. Alexakis was chastised by the HCCC for engaging in “obtuse” and “suspicious” behavior and for “blurring the boundaries of the doctor-patient relationship.”
There are three domains – legal, ethical, and practical – when it comes to accepting bequests or any gifts from patients, says Dr. Victoroff.
“[In] the legal domain, for example, if you receive a bequest from anyone, patient or otherwise, you have to know your local laws about estates and taxes and so forth and obey them,” he said.
Attorney Hursh pointed out that the Australian doctor wasn’t found guilty of wrongdoing in a court of law but rather of unethical conduct by the Australian medical licensing entity.
Patients giving gifts is often a part of a physician’s life
When Ian Schorr, MD, first started out in practice, he was surprised that patients began bringing him gifts of food to express gratitude for his care.
“I thought it was unethical to accept their gifts, so I turned them down and wouldn’t accept so much as a cookie,” Dr. Schorr, a now-retired ophthalmologist, told this news organization. “But that changed because my office staff told me that some patients were feeling disappointed and insulted. I realized that some people want to express appreciation in ways that go beyond a monetary payment.”
The next time he received a gift from a patient, he “accepted it gracefully.” And he wrote a thank you note, which he continued to do any time he received a gift from a patient.
Kenneth Prager, MD, professor of clinical medicine, director of clinical ethics and chairman of the Medical Ethics Committee at Columbia University Medical Center, New York, says, “I have literally received hundreds of gifts, the vast majority being tokens of patients’ appreciation,” he said. “I’ll get boxes of chocolate or cakes, or sometimes articles of clothing.”
Occasionally, Dr. Prager receives a “somewhat larger gift” – for example, two tickets to a baseball game. “To reject these gifts would be a slap in the face to the patient,” he says, but “where it gets more ethically cloudy is when a gift is very substantial.”
Dr. Prager has never been offered a “substantial” gift or bequest personally. “But a patient whose brother I cared for has indicated that she has left instructions in her will to endow an associate chair of ethics in my honor, and I didn’t decline that,” he said.
The AMA Code of Ethics confirms that accepting gifts offered “as an expression of gratitude or a reflection of the patient’s cultural tradition” can “enhance the patient-physician relationship.” But sometimes gifts “may signal psychological needs that require the physician’s attention.” Accepting such gifts is “likely to damage the patient-physician relationship.”
Potential damage to the therapeutic relationship applies to all physicians but especially for psychiatrists and mental health professionals. “There are more stringent ethical requirements when it comes to these disciplines, where gift-giving gets into the territory of transference or may have particular psychological meaning, and accepting the gift may muddy the therapeutic waters,” Dr. Victoroff said.
Impact on the patient’s family and on other patients
The AMA statement encourages physicians to be “sensitive to the gift’s value, relative to the patient’s or physician’s means.” Physicians should decline gifts that are “disproportionately or inappropriately large, or when the physician would be uncomfortable to have colleagues know the gift had been accepted.”
They should also decline a bequest from a patient if they have reason to believe that to accept it “would present an emotional or financial hardship to the patient’s family.”
“If Bill Gates were leaving $100,000 to his doctor, I imagine Melinda would be just fine,” Mr. Hursh said. “But under ordinary circumstances, if the patient’s family might feel the impact of the bequest, it would be unethical to accept it and could be grounds for revocation of the doctor’s license.”
The AMA statement also warns physicians that by offering a gift, some patients may be seeking to “secure or influence care or to secure preferential treatment,” which can “undermine physicians’ obligation to provide services fairly to all patients.”
For this reason, bequests are “sticky,” said Laurel Lyckholm, MD, professor of hematology and oncology at West Virginia University School of Medicine. In the case of institutions where patients or community members donate money, “we know whose names are on the plaques that hang on the hospital walls, so it’s a delicate balance. What if there’s only one bed or one ventilator? Will the wife of the donor get preferential treatment?”
Follow institutional policy
A “very small gift, such as a fruitcake, is fine,” says Dr. Lyckholm, author of an essay on accepting gifts from patients. She said there’s a dollar amount ($15) that her institution mandates, above which a gift – even food – is considered too expensive to accept. “I was a nurse before I became a physician, and people always tried to give us gifts because we were so close to the minute-by-minute care of the patients,” she said. “We were not allowed to accept money or anything lavish.”
But in the case of small gifts, “the risk-benefit analysis is that there’s much more risk not to take it and to hurt the patient’s feelings.”
Gifts above $15 are given to charity. “I explain to patients that I’m not allowed to take such a large gift, but I’d love to give it to the hospital’s Rosenbaum Family House that provides patients and their relatives with lodging, or to the homeless shelter in Morgantown.”
Dr. Lyckholm, who serves on the ethics committee at J.W. Ruby Memorial Hospital, once was offered expensive tickets and said to the patient, “This is so incredibly thoughtful and kind, but I can’t accept them. I would like to give the tickets to a charity that can auction them off.”
She advises physicians to find out their institution’s policies. Many institutions have policies about what gifts their staff – whether physicians, nurses, or other health care professionals – can accept.
Passing the ‘smell test’
Accepting a large gift from a patient could potentially make it look like you might have exercised undue influence.
“That concern brings us to the third domain, which is very practical and all about appearances and perceptions,” Dr. Victoroff said.
He noted that there is “an inherent power differential between a physician and a patient. The very nature of the relationship can create a risk of ‘undue influence’ on the doctor’s part, even if it’s not apparent to the doctor.” For this reason, it’s necessary to be utterly transparent about how the bequest came about.
He suggests that if a patient informs you that he or she would like to leave money to you, it might be wise to suggest a meeting with the patient’s family, thus establishing some transparency.
It may not be possible to meet with the patient’s family for logistical reasons or because the patient would prefer not to involve their family in their estate planning. But in any case, it’s advisable to document any conversation in the patient’s chart, Dr. Victoroff advised.
“You should make a contemporaneous note that the patient initiated the suggestion and that you counseled them about the implications, no differently than you would with an interaction of a clinical nature,” he suggests. That way, if money has been left to you and is disputed, there’s a clear record that you didn’t solicit it or use any undue influence to bring it about.
He also recommended getting advice from a trusted colleague or a member of your institution’s ethics committee. “Taking time to get a second opinion about an ethical question is a safeguard, like having a chaperone in the room during an examination.”
Ultimately, “there is no human relationship without potential conflicts of interest. Our job is to manage those as best as we can, and sunlight is the best antidote to bad appearances,” Dr. Victoroff said.
A version of this article appeared on Medscape.com.
Michael Victoroff, MD, described the phone call he received from an attorney asking a thorny ethics question involving a patient’s gift to another physician. Dr. Victoroff, a past member of the ethics committee of the American Academy of Family Physicians, had definite thoughts about it.
“The attorney was representing the daughters of an elderly gentleman who had moved from the East Coast to Colorado to be closer to them,” said Dr. Victoroff, who teaches bioethics in the MBA program at the University of Denver and also practices at the University of Colorado School of Medicine.
“The father visited his new primary care physician frequently because he had multiple health issues.”
The patient was happy with the doctor’s medical care and over time that they developed a friendship. Dr. Victoroff emphasized that no sexual or romantic impropriety ever took place between the patient and his physician.
“But the social relationship went beyond the ordinary doctor-patient boundaries. The patient ultimately named the doctor as his health care proxy in the event that he became unable to make decisions regarding his care. He also mentioned he was going to leave her $100,000 in his will,” says Dr. Victoroff.
The physician did accept the role of proxy, “which raises a whole host of ethical issues,” says Dr. Victoroff. As it happened, she was never called upon to exercise that decision-making authority, since the patient died suddenly and was mentally competent at the time.
for her to accept such a substantial bequest from a patient, and they hired an attorney to contest the will.
No law against it
Dennis Hursh, attorney and managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, noted in an interview that, “the problem isn’t legal per se. Rather, the problem is an ethical one.”
Legally speaking, there’s no prohibition against receiving a bequest or other form of gift from a patient. “People are free to dispose of their estates in whatever way they see fit, and no law technically precludes a physician from accepting a bequest,” says Dr. Victoroff. “But this presupposes there is nothing improper going on, such as extortion, deception, coercion, or exercising undue influence.”
The issue of bequeathing money to their physician gained attention in a recent case that took place in Australia. Peter Alexakis, MD, received a whopping bequest of $24 million from a patient. The elderly patient had changed his will to name Dr. Alexakis as the sole beneficiary – after Dr. Alexakis had visited him at home 92 times during the preceding months. The original heirs filed a lawsuit in Australia’s Supreme Court against Dr. Alexakis, contesting the will.
The lawsuit was unsuccessful in court, but Dr. Alexakis was found guilty of malpractice by Australia’s Health Care Complaints Commission after being reported to the HCCC by the palliative care physicians who were treating the patient. They alleged that Dr. Alexakis had interfered with their care of the patient. The more serious allegation was that the doctor had engaged in a deliberate strategy to exploit the relationship for financial gain.
Dr. Alexakis was chastised by the HCCC for engaging in “obtuse” and “suspicious” behavior and for “blurring the boundaries of the doctor-patient relationship.”
There are three domains – legal, ethical, and practical – when it comes to accepting bequests or any gifts from patients, says Dr. Victoroff.
“[In] the legal domain, for example, if you receive a bequest from anyone, patient or otherwise, you have to know your local laws about estates and taxes and so forth and obey them,” he said.
Attorney Hursh pointed out that the Australian doctor wasn’t found guilty of wrongdoing in a court of law but rather of unethical conduct by the Australian medical licensing entity.
Patients giving gifts is often a part of a physician’s life
When Ian Schorr, MD, first started out in practice, he was surprised that patients began bringing him gifts of food to express gratitude for his care.
“I thought it was unethical to accept their gifts, so I turned them down and wouldn’t accept so much as a cookie,” Dr. Schorr, a now-retired ophthalmologist, told this news organization. “But that changed because my office staff told me that some patients were feeling disappointed and insulted. I realized that some people want to express appreciation in ways that go beyond a monetary payment.”
The next time he received a gift from a patient, he “accepted it gracefully.” And he wrote a thank you note, which he continued to do any time he received a gift from a patient.
Kenneth Prager, MD, professor of clinical medicine, director of clinical ethics and chairman of the Medical Ethics Committee at Columbia University Medical Center, New York, says, “I have literally received hundreds of gifts, the vast majority being tokens of patients’ appreciation,” he said. “I’ll get boxes of chocolate or cakes, or sometimes articles of clothing.”
Occasionally, Dr. Prager receives a “somewhat larger gift” – for example, two tickets to a baseball game. “To reject these gifts would be a slap in the face to the patient,” he says, but “where it gets more ethically cloudy is when a gift is very substantial.”
Dr. Prager has never been offered a “substantial” gift or bequest personally. “But a patient whose brother I cared for has indicated that she has left instructions in her will to endow an associate chair of ethics in my honor, and I didn’t decline that,” he said.
The AMA Code of Ethics confirms that accepting gifts offered “as an expression of gratitude or a reflection of the patient’s cultural tradition” can “enhance the patient-physician relationship.” But sometimes gifts “may signal psychological needs that require the physician’s attention.” Accepting such gifts is “likely to damage the patient-physician relationship.”
Potential damage to the therapeutic relationship applies to all physicians but especially for psychiatrists and mental health professionals. “There are more stringent ethical requirements when it comes to these disciplines, where gift-giving gets into the territory of transference or may have particular psychological meaning, and accepting the gift may muddy the therapeutic waters,” Dr. Victoroff said.
Impact on the patient’s family and on other patients
The AMA statement encourages physicians to be “sensitive to the gift’s value, relative to the patient’s or physician’s means.” Physicians should decline gifts that are “disproportionately or inappropriately large, or when the physician would be uncomfortable to have colleagues know the gift had been accepted.”
They should also decline a bequest from a patient if they have reason to believe that to accept it “would present an emotional or financial hardship to the patient’s family.”
“If Bill Gates were leaving $100,000 to his doctor, I imagine Melinda would be just fine,” Mr. Hursh said. “But under ordinary circumstances, if the patient’s family might feel the impact of the bequest, it would be unethical to accept it and could be grounds for revocation of the doctor’s license.”
The AMA statement also warns physicians that by offering a gift, some patients may be seeking to “secure or influence care or to secure preferential treatment,” which can “undermine physicians’ obligation to provide services fairly to all patients.”
For this reason, bequests are “sticky,” said Laurel Lyckholm, MD, professor of hematology and oncology at West Virginia University School of Medicine. In the case of institutions where patients or community members donate money, “we know whose names are on the plaques that hang on the hospital walls, so it’s a delicate balance. What if there’s only one bed or one ventilator? Will the wife of the donor get preferential treatment?”
Follow institutional policy
A “very small gift, such as a fruitcake, is fine,” says Dr. Lyckholm, author of an essay on accepting gifts from patients. She said there’s a dollar amount ($15) that her institution mandates, above which a gift – even food – is considered too expensive to accept. “I was a nurse before I became a physician, and people always tried to give us gifts because we were so close to the minute-by-minute care of the patients,” she said. “We were not allowed to accept money or anything lavish.”
But in the case of small gifts, “the risk-benefit analysis is that there’s much more risk not to take it and to hurt the patient’s feelings.”
Gifts above $15 are given to charity. “I explain to patients that I’m not allowed to take such a large gift, but I’d love to give it to the hospital’s Rosenbaum Family House that provides patients and their relatives with lodging, or to the homeless shelter in Morgantown.”
Dr. Lyckholm, who serves on the ethics committee at J.W. Ruby Memorial Hospital, once was offered expensive tickets and said to the patient, “This is so incredibly thoughtful and kind, but I can’t accept them. I would like to give the tickets to a charity that can auction them off.”
She advises physicians to find out their institution’s policies. Many institutions have policies about what gifts their staff – whether physicians, nurses, or other health care professionals – can accept.
Passing the ‘smell test’
Accepting a large gift from a patient could potentially make it look like you might have exercised undue influence.
“That concern brings us to the third domain, which is very practical and all about appearances and perceptions,” Dr. Victoroff said.
He noted that there is “an inherent power differential between a physician and a patient. The very nature of the relationship can create a risk of ‘undue influence’ on the doctor’s part, even if it’s not apparent to the doctor.” For this reason, it’s necessary to be utterly transparent about how the bequest came about.
He suggests that if a patient informs you that he or she would like to leave money to you, it might be wise to suggest a meeting with the patient’s family, thus establishing some transparency.
It may not be possible to meet with the patient’s family for logistical reasons or because the patient would prefer not to involve their family in their estate planning. But in any case, it’s advisable to document any conversation in the patient’s chart, Dr. Victoroff advised.
“You should make a contemporaneous note that the patient initiated the suggestion and that you counseled them about the implications, no differently than you would with an interaction of a clinical nature,” he suggests. That way, if money has been left to you and is disputed, there’s a clear record that you didn’t solicit it or use any undue influence to bring it about.
He also recommended getting advice from a trusted colleague or a member of your institution’s ethics committee. “Taking time to get a second opinion about an ethical question is a safeguard, like having a chaperone in the room during an examination.”
Ultimately, “there is no human relationship without potential conflicts of interest. Our job is to manage those as best as we can, and sunlight is the best antidote to bad appearances,” Dr. Victoroff said.
A version of this article appeared on Medscape.com.
Michael Victoroff, MD, described the phone call he received from an attorney asking a thorny ethics question involving a patient’s gift to another physician. Dr. Victoroff, a past member of the ethics committee of the American Academy of Family Physicians, had definite thoughts about it.
“The attorney was representing the daughters of an elderly gentleman who had moved from the East Coast to Colorado to be closer to them,” said Dr. Victoroff, who teaches bioethics in the MBA program at the University of Denver and also practices at the University of Colorado School of Medicine.
“The father visited his new primary care physician frequently because he had multiple health issues.”
The patient was happy with the doctor’s medical care and over time that they developed a friendship. Dr. Victoroff emphasized that no sexual or romantic impropriety ever took place between the patient and his physician.
“But the social relationship went beyond the ordinary doctor-patient boundaries. The patient ultimately named the doctor as his health care proxy in the event that he became unable to make decisions regarding his care. He also mentioned he was going to leave her $100,000 in his will,” says Dr. Victoroff.
The physician did accept the role of proxy, “which raises a whole host of ethical issues,” says Dr. Victoroff. As it happened, she was never called upon to exercise that decision-making authority, since the patient died suddenly and was mentally competent at the time.
for her to accept such a substantial bequest from a patient, and they hired an attorney to contest the will.
No law against it
Dennis Hursh, attorney and managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, noted in an interview that, “the problem isn’t legal per se. Rather, the problem is an ethical one.”
Legally speaking, there’s no prohibition against receiving a bequest or other form of gift from a patient. “People are free to dispose of their estates in whatever way they see fit, and no law technically precludes a physician from accepting a bequest,” says Dr. Victoroff. “But this presupposes there is nothing improper going on, such as extortion, deception, coercion, or exercising undue influence.”
The issue of bequeathing money to their physician gained attention in a recent case that took place in Australia. Peter Alexakis, MD, received a whopping bequest of $24 million from a patient. The elderly patient had changed his will to name Dr. Alexakis as the sole beneficiary – after Dr. Alexakis had visited him at home 92 times during the preceding months. The original heirs filed a lawsuit in Australia’s Supreme Court against Dr. Alexakis, contesting the will.
The lawsuit was unsuccessful in court, but Dr. Alexakis was found guilty of malpractice by Australia’s Health Care Complaints Commission after being reported to the HCCC by the palliative care physicians who were treating the patient. They alleged that Dr. Alexakis had interfered with their care of the patient. The more serious allegation was that the doctor had engaged in a deliberate strategy to exploit the relationship for financial gain.
Dr. Alexakis was chastised by the HCCC for engaging in “obtuse” and “suspicious” behavior and for “blurring the boundaries of the doctor-patient relationship.”
There are three domains – legal, ethical, and practical – when it comes to accepting bequests or any gifts from patients, says Dr. Victoroff.
“[In] the legal domain, for example, if you receive a bequest from anyone, patient or otherwise, you have to know your local laws about estates and taxes and so forth and obey them,” he said.
Attorney Hursh pointed out that the Australian doctor wasn’t found guilty of wrongdoing in a court of law but rather of unethical conduct by the Australian medical licensing entity.
Patients giving gifts is often a part of a physician’s life
When Ian Schorr, MD, first started out in practice, he was surprised that patients began bringing him gifts of food to express gratitude for his care.
“I thought it was unethical to accept their gifts, so I turned them down and wouldn’t accept so much as a cookie,” Dr. Schorr, a now-retired ophthalmologist, told this news organization. “But that changed because my office staff told me that some patients were feeling disappointed and insulted. I realized that some people want to express appreciation in ways that go beyond a monetary payment.”
The next time he received a gift from a patient, he “accepted it gracefully.” And he wrote a thank you note, which he continued to do any time he received a gift from a patient.
Kenneth Prager, MD, professor of clinical medicine, director of clinical ethics and chairman of the Medical Ethics Committee at Columbia University Medical Center, New York, says, “I have literally received hundreds of gifts, the vast majority being tokens of patients’ appreciation,” he said. “I’ll get boxes of chocolate or cakes, or sometimes articles of clothing.”
Occasionally, Dr. Prager receives a “somewhat larger gift” – for example, two tickets to a baseball game. “To reject these gifts would be a slap in the face to the patient,” he says, but “where it gets more ethically cloudy is when a gift is very substantial.”
Dr. Prager has never been offered a “substantial” gift or bequest personally. “But a patient whose brother I cared for has indicated that she has left instructions in her will to endow an associate chair of ethics in my honor, and I didn’t decline that,” he said.
The AMA Code of Ethics confirms that accepting gifts offered “as an expression of gratitude or a reflection of the patient’s cultural tradition” can “enhance the patient-physician relationship.” But sometimes gifts “may signal psychological needs that require the physician’s attention.” Accepting such gifts is “likely to damage the patient-physician relationship.”
Potential damage to the therapeutic relationship applies to all physicians but especially for psychiatrists and mental health professionals. “There are more stringent ethical requirements when it comes to these disciplines, where gift-giving gets into the territory of transference or may have particular psychological meaning, and accepting the gift may muddy the therapeutic waters,” Dr. Victoroff said.
Impact on the patient’s family and on other patients
The AMA statement encourages physicians to be “sensitive to the gift’s value, relative to the patient’s or physician’s means.” Physicians should decline gifts that are “disproportionately or inappropriately large, or when the physician would be uncomfortable to have colleagues know the gift had been accepted.”
They should also decline a bequest from a patient if they have reason to believe that to accept it “would present an emotional or financial hardship to the patient’s family.”
“If Bill Gates were leaving $100,000 to his doctor, I imagine Melinda would be just fine,” Mr. Hursh said. “But under ordinary circumstances, if the patient’s family might feel the impact of the bequest, it would be unethical to accept it and could be grounds for revocation of the doctor’s license.”
The AMA statement also warns physicians that by offering a gift, some patients may be seeking to “secure or influence care or to secure preferential treatment,” which can “undermine physicians’ obligation to provide services fairly to all patients.”
For this reason, bequests are “sticky,” said Laurel Lyckholm, MD, professor of hematology and oncology at West Virginia University School of Medicine. In the case of institutions where patients or community members donate money, “we know whose names are on the plaques that hang on the hospital walls, so it’s a delicate balance. What if there’s only one bed or one ventilator? Will the wife of the donor get preferential treatment?”
Follow institutional policy
A “very small gift, such as a fruitcake, is fine,” says Dr. Lyckholm, author of an essay on accepting gifts from patients. She said there’s a dollar amount ($15) that her institution mandates, above which a gift – even food – is considered too expensive to accept. “I was a nurse before I became a physician, and people always tried to give us gifts because we were so close to the minute-by-minute care of the patients,” she said. “We were not allowed to accept money or anything lavish.”
But in the case of small gifts, “the risk-benefit analysis is that there’s much more risk not to take it and to hurt the patient’s feelings.”
Gifts above $15 are given to charity. “I explain to patients that I’m not allowed to take such a large gift, but I’d love to give it to the hospital’s Rosenbaum Family House that provides patients and their relatives with lodging, or to the homeless shelter in Morgantown.”
Dr. Lyckholm, who serves on the ethics committee at J.W. Ruby Memorial Hospital, once was offered expensive tickets and said to the patient, “This is so incredibly thoughtful and kind, but I can’t accept them. I would like to give the tickets to a charity that can auction them off.”
She advises physicians to find out their institution’s policies. Many institutions have policies about what gifts their staff – whether physicians, nurses, or other health care professionals – can accept.
Passing the ‘smell test’
Accepting a large gift from a patient could potentially make it look like you might have exercised undue influence.
“That concern brings us to the third domain, which is very practical and all about appearances and perceptions,” Dr. Victoroff said.
He noted that there is “an inherent power differential between a physician and a patient. The very nature of the relationship can create a risk of ‘undue influence’ on the doctor’s part, even if it’s not apparent to the doctor.” For this reason, it’s necessary to be utterly transparent about how the bequest came about.
He suggests that if a patient informs you that he or she would like to leave money to you, it might be wise to suggest a meeting with the patient’s family, thus establishing some transparency.
It may not be possible to meet with the patient’s family for logistical reasons or because the patient would prefer not to involve their family in their estate planning. But in any case, it’s advisable to document any conversation in the patient’s chart, Dr. Victoroff advised.
“You should make a contemporaneous note that the patient initiated the suggestion and that you counseled them about the implications, no differently than you would with an interaction of a clinical nature,” he suggests. That way, if money has been left to you and is disputed, there’s a clear record that you didn’t solicit it or use any undue influence to bring it about.
He also recommended getting advice from a trusted colleague or a member of your institution’s ethics committee. “Taking time to get a second opinion about an ethical question is a safeguard, like having a chaperone in the room during an examination.”
Ultimately, “there is no human relationship without potential conflicts of interest. Our job is to manage those as best as we can, and sunlight is the best antidote to bad appearances,” Dr. Victoroff said.
A version of this article appeared on Medscape.com.












