User login
-
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]


Children with cystic fibrosis and their caregivers face sleep difficulties
, according to results from a new study.
Children aged 6-12 years had more sleep issues compared with preschoolers or teenagers, researchers also found, and the quality of sleep among caregivers was seen strongly linked to that of their children with CF.
For research published in the Journal of Cystic Fibrosis, Kelly C. Byars, PsyD, and colleagues at Cincinnati Children’s Medical Center and the University of Cincinnati surveyed parents of 91 medically stable patients with cystic fibrosis aged 18 and younger at a single CF treatment center between 2016 and 2017.
Fifty-four percent of the children in the study were female, the mean age was 9 years, and 90% of the caregivers were mothers. In addition to the sleep questionnaires, the researchers looked at the children’s available lung function data from around the time of the survey. Forced expiratory volume in one second (FEV1) measures showed the vast majority had no obstructive lung disease (73% of the cohort) or only mild symptoms (18%) at the time their caregivers were surveyed.
Overall, some 40% of caregivers said they had concerns about their own sleep, while 29% said they were concerned for their children’s sleep. Parents reported night waking, daytime sleepiness, and difficulty falling asleep as their main problems, and difficulty falling asleep as the top issue for their children, along with daytime sleepiness, night waking, and mouth breathing.
Sleep issues were most pronounced for children aged 6-12 and their caregivers, a group for which 44% of caregivers said they were concerned for their children’s sleep and 55% for their own sleep. For this same group only 8% of parents reported their children having nocturnal cough, and just 5% reported gastrointestinal problems at night.
Overall, the caregivers in the study reported inadequate sleep, with more than half saying they got less than 7 hours per night. Similarly, more than half of the school-age and adolescent patients with CF were getting less than the nightly minimum recommended by the American Academy of Sleep Medicine.
The researchers noted “large effects for parent and child associations for insomnia symptoms that may be amenable to treatment,” especially trouble returning to sleep and daytime sleepiness.
The study “is the first to examine parent reported sleep disturbances and sleep duration in both parents and their children with CF spanning a broad age range and including patients who were medically stable and predominantly free of lung dysfunction,” Dr. Byars and colleagues wrote in their analysis, adding that sleep health should be integrated into care protocols for CF patients and their families, and families of children with other chronic illnesses.
In a comment on Dr. Byars and colleagues’ study, Hovig Artinian, MD, a pediatric pulmonary and sleep medicine specialist at Helen DeVos Children’s Hospital in Grand Rapids, Mich., said the findings “highlight for all of us that we must regularly assess and address sleep disturbances in our children with CF specifically, but also in all children with chronic conditions.”
Children with CF “carry a heavy burden,” Dr. Artinian said, “balancing living their lives with daily interruptions to their typical day to complete multiple treatments. As a result, sleep can be impacted even when there are no other clinical or objective signs of illness, so that was not an entirely surprising finding.” Difficulties with sleep onset and maintenance can be prevalent in the absence of changes in children’s daytime behavior or any other psychological signs, Dr. Artinian said, noting that in his practice he routinely asks families whether children snore (something recommended by the American Academy of Pediatrics for all well-child checks) and whether they have any other concerns about their sleep.
“Even if the answer is ‘no’ the first time, the act of asking plants a seed in their minds to keep an eye open and to know they can discuss it with us at a future visit if concerns come up,” Dr. Artinian said.
Dr. Byars and colleagues noted several limitations to their study including its cross-sectional, single-center design, potential participant selection bias, reliance on parent reports of child sleep, and use of a novel, nonvalidated survey instrument.
The researchers received funding from the Boomer Esiason Foundation for their study and disclosed no financial conflicts of interest. Dr. Artinian had no relevant disclosures.
SOURCE: Byars K et al. J Cyst Fibros. 2020 May. doi: 10.1016/j.jcf.2020.04.003.
, according to results from a new study.
Children aged 6-12 years had more sleep issues compared with preschoolers or teenagers, researchers also found, and the quality of sleep among caregivers was seen strongly linked to that of their children with CF.
For research published in the Journal of Cystic Fibrosis, Kelly C. Byars, PsyD, and colleagues at Cincinnati Children’s Medical Center and the University of Cincinnati surveyed parents of 91 medically stable patients with cystic fibrosis aged 18 and younger at a single CF treatment center between 2016 and 2017.
Fifty-four percent of the children in the study were female, the mean age was 9 years, and 90% of the caregivers were mothers. In addition to the sleep questionnaires, the researchers looked at the children’s available lung function data from around the time of the survey. Forced expiratory volume in one second (FEV1) measures showed the vast majority had no obstructive lung disease (73% of the cohort) or only mild symptoms (18%) at the time their caregivers were surveyed.
Overall, some 40% of caregivers said they had concerns about their own sleep, while 29% said they were concerned for their children’s sleep. Parents reported night waking, daytime sleepiness, and difficulty falling asleep as their main problems, and difficulty falling asleep as the top issue for their children, along with daytime sleepiness, night waking, and mouth breathing.
Sleep issues were most pronounced for children aged 6-12 and their caregivers, a group for which 44% of caregivers said they were concerned for their children’s sleep and 55% for their own sleep. For this same group only 8% of parents reported their children having nocturnal cough, and just 5% reported gastrointestinal problems at night.
Overall, the caregivers in the study reported inadequate sleep, with more than half saying they got less than 7 hours per night. Similarly, more than half of the school-age and adolescent patients with CF were getting less than the nightly minimum recommended by the American Academy of Sleep Medicine.
The researchers noted “large effects for parent and child associations for insomnia symptoms that may be amenable to treatment,” especially trouble returning to sleep and daytime sleepiness.
The study “is the first to examine parent reported sleep disturbances and sleep duration in both parents and their children with CF spanning a broad age range and including patients who were medically stable and predominantly free of lung dysfunction,” Dr. Byars and colleagues wrote in their analysis, adding that sleep health should be integrated into care protocols for CF patients and their families, and families of children with other chronic illnesses.
In a comment on Dr. Byars and colleagues’ study, Hovig Artinian, MD, a pediatric pulmonary and sleep medicine specialist at Helen DeVos Children’s Hospital in Grand Rapids, Mich., said the findings “highlight for all of us that we must regularly assess and address sleep disturbances in our children with CF specifically, but also in all children with chronic conditions.”
Children with CF “carry a heavy burden,” Dr. Artinian said, “balancing living their lives with daily interruptions to their typical day to complete multiple treatments. As a result, sleep can be impacted even when there are no other clinical or objective signs of illness, so that was not an entirely surprising finding.” Difficulties with sleep onset and maintenance can be prevalent in the absence of changes in children’s daytime behavior or any other psychological signs, Dr. Artinian said, noting that in his practice he routinely asks families whether children snore (something recommended by the American Academy of Pediatrics for all well-child checks) and whether they have any other concerns about their sleep.
“Even if the answer is ‘no’ the first time, the act of asking plants a seed in their minds to keep an eye open and to know they can discuss it with us at a future visit if concerns come up,” Dr. Artinian said.
Dr. Byars and colleagues noted several limitations to their study including its cross-sectional, single-center design, potential participant selection bias, reliance on parent reports of child sleep, and use of a novel, nonvalidated survey instrument.
The researchers received funding from the Boomer Esiason Foundation for their study and disclosed no financial conflicts of interest. Dr. Artinian had no relevant disclosures.
SOURCE: Byars K et al. J Cyst Fibros. 2020 May. doi: 10.1016/j.jcf.2020.04.003.
, according to results from a new study.
Children aged 6-12 years had more sleep issues compared with preschoolers or teenagers, researchers also found, and the quality of sleep among caregivers was seen strongly linked to that of their children with CF.
For research published in the Journal of Cystic Fibrosis, Kelly C. Byars, PsyD, and colleagues at Cincinnati Children’s Medical Center and the University of Cincinnati surveyed parents of 91 medically stable patients with cystic fibrosis aged 18 and younger at a single CF treatment center between 2016 and 2017.
Fifty-four percent of the children in the study were female, the mean age was 9 years, and 90% of the caregivers were mothers. In addition to the sleep questionnaires, the researchers looked at the children’s available lung function data from around the time of the survey. Forced expiratory volume in one second (FEV1) measures showed the vast majority had no obstructive lung disease (73% of the cohort) or only mild symptoms (18%) at the time their caregivers were surveyed.
Overall, some 40% of caregivers said they had concerns about their own sleep, while 29% said they were concerned for their children’s sleep. Parents reported night waking, daytime sleepiness, and difficulty falling asleep as their main problems, and difficulty falling asleep as the top issue for their children, along with daytime sleepiness, night waking, and mouth breathing.
Sleep issues were most pronounced for children aged 6-12 and their caregivers, a group for which 44% of caregivers said they were concerned for their children’s sleep and 55% for their own sleep. For this same group only 8% of parents reported their children having nocturnal cough, and just 5% reported gastrointestinal problems at night.
Overall, the caregivers in the study reported inadequate sleep, with more than half saying they got less than 7 hours per night. Similarly, more than half of the school-age and adolescent patients with CF were getting less than the nightly minimum recommended by the American Academy of Sleep Medicine.
The researchers noted “large effects for parent and child associations for insomnia symptoms that may be amenable to treatment,” especially trouble returning to sleep and daytime sleepiness.
The study “is the first to examine parent reported sleep disturbances and sleep duration in both parents and their children with CF spanning a broad age range and including patients who were medically stable and predominantly free of lung dysfunction,” Dr. Byars and colleagues wrote in their analysis, adding that sleep health should be integrated into care protocols for CF patients and their families, and families of children with other chronic illnesses.
In a comment on Dr. Byars and colleagues’ study, Hovig Artinian, MD, a pediatric pulmonary and sleep medicine specialist at Helen DeVos Children’s Hospital in Grand Rapids, Mich., said the findings “highlight for all of us that we must regularly assess and address sleep disturbances in our children with CF specifically, but also in all children with chronic conditions.”
Children with CF “carry a heavy burden,” Dr. Artinian said, “balancing living their lives with daily interruptions to their typical day to complete multiple treatments. As a result, sleep can be impacted even when there are no other clinical or objective signs of illness, so that was not an entirely surprising finding.” Difficulties with sleep onset and maintenance can be prevalent in the absence of changes in children’s daytime behavior or any other psychological signs, Dr. Artinian said, noting that in his practice he routinely asks families whether children snore (something recommended by the American Academy of Pediatrics for all well-child checks) and whether they have any other concerns about their sleep.
“Even if the answer is ‘no’ the first time, the act of asking plants a seed in their minds to keep an eye open and to know they can discuss it with us at a future visit if concerns come up,” Dr. Artinian said.
Dr. Byars and colleagues noted several limitations to their study including its cross-sectional, single-center design, potential participant selection bias, reliance on parent reports of child sleep, and use of a novel, nonvalidated survey instrument.
The researchers received funding from the Boomer Esiason Foundation for their study and disclosed no financial conflicts of interest. Dr. Artinian had no relevant disclosures.
SOURCE: Byars K et al. J Cyst Fibros. 2020 May. doi: 10.1016/j.jcf.2020.04.003.
FROM THE JOURNAL OF CYSTIC FIBROSIS
Clinicians urged to use CURE ID to report COVID-19 cases
in conjunction with ongoing clinical trial efforts.
“By utilizing the CURE ID platform now for COVID-19 case collection – in conjunction with data gathered from other registries, EHR systems, and clinical trials – data collected during an outbreak can be improved and coordinated,” Heather A. Stone, MPH, said during a June 9 webinar sponsored by the Food and Drug Administration. “This may allow us to find possible treatments to help ease this pandemic, and prepare us better to fight the next one.”
During the hour-long webinar, Ms. Stone, a health science policy analyst in the office of medical policy at the FDA’s Center for Drug Evaluation and Research, demonstrated CURE ID, an Internet-based data repository first developed in 2013 as a collaboration between the FDA and the National Center for Advancing Translational Sciences, a part of the National Institutes of Health (NCATS/NIH). It provides licensed clinicians worldwide with an opportunity to report novel uses of existing drugs for patients with difficult-to-treat infectious diseases, including COVID-19, through a website, a smartphone, or other mobile device. The app can be downloaded for free at http://cure.ncats.io. It can also be downloaded from the Apple app store or the Google Play store by searching “CURE ID.”
According to Ms. Stone, the platform’s three main goals are to enhance the understanding of new uses of approved medical products, to facilitate clinical trials and drug development, and to serve as a resource for physicians to share information where no FDA-approved product (which has been proven to be safe and effective) exists for the new use. CURE ID enables users to report their own cases as well as read cases of neglected infectious diseases with no sufficient approved therapies from other clinicians around the world. “It also enables clinicians to engage directly with communities of disease experts around the world, breaking down geographic and specialty silos,” Ms. Stone said. “It also enables them to access information on approved therapies for each disease and as well on active clinical trials.”
To date, CURE-ID contains information on 325 infectious diseases, including 1,580 case reports and 18,907 clinical trials. Initial pilot priority diseases include COVID-19, mycetoma, atypical mycobacteria, drug-resistant gonorrhea, rare and resistant fungal infections, as well as multidrug resistant gram-negative bacteria.
As of June 9, COVID-19-related data on the platform includes 151 case reports that have been extracted from the published literature or entered by clinician users, 80 discussion posts, and links to 694 clinical trials, 303 journal articles, 212 news articles, and 34 events. A total of 65 repurposed drugs have been identified as potential treatments for the virus, including 15 drugs with 10 or more cases.
“This facilitates clinicians reporting their real-world experiences treating COVID-19 patients, when patients are unable to be enrolled in a clinical trial,” Ms. Stone said. “It includes an updated case report form tailored to COVID-19 and data fields that have been harmonized with other real-world data and clinical trial platforms.” She pointed out that voluntary submission of cases to CURE ID is not a substitute for filing information with regulatory and public health authorities, where required. The platform also enables data to be entered and adverse events to be automatically shared with the FDA’s MedWatch Adverse Reporting System.
Ms. Stone concluded the webinar by announcing the formation of a new private-public partnership between the Critical Path Institute and the FDA and NCATS/NIH known as the CURE Drug Repurposing Collaboratory. The effort will begin with a pilot project focused on furthering drug development for COVID-19 through use of the CURE ID platform. “The Collaboratory will demonstrate how data shared from clinicians in real-time can be used to inform ongoing and future clinical trials, and potentially drug labeling,” Ms. Stone said. She reported having no financial disclosures.
in conjunction with ongoing clinical trial efforts.
“By utilizing the CURE ID platform now for COVID-19 case collection – in conjunction with data gathered from other registries, EHR systems, and clinical trials – data collected during an outbreak can be improved and coordinated,” Heather A. Stone, MPH, said during a June 9 webinar sponsored by the Food and Drug Administration. “This may allow us to find possible treatments to help ease this pandemic, and prepare us better to fight the next one.”
During the hour-long webinar, Ms. Stone, a health science policy analyst in the office of medical policy at the FDA’s Center for Drug Evaluation and Research, demonstrated CURE ID, an Internet-based data repository first developed in 2013 as a collaboration between the FDA and the National Center for Advancing Translational Sciences, a part of the National Institutes of Health (NCATS/NIH). It provides licensed clinicians worldwide with an opportunity to report novel uses of existing drugs for patients with difficult-to-treat infectious diseases, including COVID-19, through a website, a smartphone, or other mobile device. The app can be downloaded for free at http://cure.ncats.io. It can also be downloaded from the Apple app store or the Google Play store by searching “CURE ID.”
According to Ms. Stone, the platform’s three main goals are to enhance the understanding of new uses of approved medical products, to facilitate clinical trials and drug development, and to serve as a resource for physicians to share information where no FDA-approved product (which has been proven to be safe and effective) exists for the new use. CURE ID enables users to report their own cases as well as read cases of neglected infectious diseases with no sufficient approved therapies from other clinicians around the world. “It also enables clinicians to engage directly with communities of disease experts around the world, breaking down geographic and specialty silos,” Ms. Stone said. “It also enables them to access information on approved therapies for each disease and as well on active clinical trials.”
To date, CURE-ID contains information on 325 infectious diseases, including 1,580 case reports and 18,907 clinical trials. Initial pilot priority diseases include COVID-19, mycetoma, atypical mycobacteria, drug-resistant gonorrhea, rare and resistant fungal infections, as well as multidrug resistant gram-negative bacteria.
As of June 9, COVID-19-related data on the platform includes 151 case reports that have been extracted from the published literature or entered by clinician users, 80 discussion posts, and links to 694 clinical trials, 303 journal articles, 212 news articles, and 34 events. A total of 65 repurposed drugs have been identified as potential treatments for the virus, including 15 drugs with 10 or more cases.
“This facilitates clinicians reporting their real-world experiences treating COVID-19 patients, when patients are unable to be enrolled in a clinical trial,” Ms. Stone said. “It includes an updated case report form tailored to COVID-19 and data fields that have been harmonized with other real-world data and clinical trial platforms.” She pointed out that voluntary submission of cases to CURE ID is not a substitute for filing information with regulatory and public health authorities, where required. The platform also enables data to be entered and adverse events to be automatically shared with the FDA’s MedWatch Adverse Reporting System.
Ms. Stone concluded the webinar by announcing the formation of a new private-public partnership between the Critical Path Institute and the FDA and NCATS/NIH known as the CURE Drug Repurposing Collaboratory. The effort will begin with a pilot project focused on furthering drug development for COVID-19 through use of the CURE ID platform. “The Collaboratory will demonstrate how data shared from clinicians in real-time can be used to inform ongoing and future clinical trials, and potentially drug labeling,” Ms. Stone said. She reported having no financial disclosures.
in conjunction with ongoing clinical trial efforts.
“By utilizing the CURE ID platform now for COVID-19 case collection – in conjunction with data gathered from other registries, EHR systems, and clinical trials – data collected during an outbreak can be improved and coordinated,” Heather A. Stone, MPH, said during a June 9 webinar sponsored by the Food and Drug Administration. “This may allow us to find possible treatments to help ease this pandemic, and prepare us better to fight the next one.”
During the hour-long webinar, Ms. Stone, a health science policy analyst in the office of medical policy at the FDA’s Center for Drug Evaluation and Research, demonstrated CURE ID, an Internet-based data repository first developed in 2013 as a collaboration between the FDA and the National Center for Advancing Translational Sciences, a part of the National Institutes of Health (NCATS/NIH). It provides licensed clinicians worldwide with an opportunity to report novel uses of existing drugs for patients with difficult-to-treat infectious diseases, including COVID-19, through a website, a smartphone, or other mobile device. The app can be downloaded for free at http://cure.ncats.io. It can also be downloaded from the Apple app store or the Google Play store by searching “CURE ID.”
According to Ms. Stone, the platform’s three main goals are to enhance the understanding of new uses of approved medical products, to facilitate clinical trials and drug development, and to serve as a resource for physicians to share information where no FDA-approved product (which has been proven to be safe and effective) exists for the new use. CURE ID enables users to report their own cases as well as read cases of neglected infectious diseases with no sufficient approved therapies from other clinicians around the world. “It also enables clinicians to engage directly with communities of disease experts around the world, breaking down geographic and specialty silos,” Ms. Stone said. “It also enables them to access information on approved therapies for each disease and as well on active clinical trials.”
To date, CURE-ID contains information on 325 infectious diseases, including 1,580 case reports and 18,907 clinical trials. Initial pilot priority diseases include COVID-19, mycetoma, atypical mycobacteria, drug-resistant gonorrhea, rare and resistant fungal infections, as well as multidrug resistant gram-negative bacteria.
As of June 9, COVID-19-related data on the platform includes 151 case reports that have been extracted from the published literature or entered by clinician users, 80 discussion posts, and links to 694 clinical trials, 303 journal articles, 212 news articles, and 34 events. A total of 65 repurposed drugs have been identified as potential treatments for the virus, including 15 drugs with 10 or more cases.
“This facilitates clinicians reporting their real-world experiences treating COVID-19 patients, when patients are unable to be enrolled in a clinical trial,” Ms. Stone said. “It includes an updated case report form tailored to COVID-19 and data fields that have been harmonized with other real-world data and clinical trial platforms.” She pointed out that voluntary submission of cases to CURE ID is not a substitute for filing information with regulatory and public health authorities, where required. The platform also enables data to be entered and adverse events to be automatically shared with the FDA’s MedWatch Adverse Reporting System.
Ms. Stone concluded the webinar by announcing the formation of a new private-public partnership between the Critical Path Institute and the FDA and NCATS/NIH known as the CURE Drug Repurposing Collaboratory. The effort will begin with a pilot project focused on furthering drug development for COVID-19 through use of the CURE ID platform. “The Collaboratory will demonstrate how data shared from clinicians in real-time can be used to inform ongoing and future clinical trials, and potentially drug labeling,” Ms. Stone said. She reported having no financial disclosures.
EULAR’s COVID-19 recommendations offer no surprises
As might be expected, the “EULAR [European League Against Rheumatism] provisional recommendations for the management of rheumatic and musculoskeletal diseases [RMDs] in the context of SARS-CoV-2” concur with much of the guidance already released on how best to manage patients during the current pandemic.
Highlights of the five overarching principles are that, contrary to earlier expectations, “there is no indication that patients with RMDs have an additional, or have a higher, risk of contracting the virus, or that they fare a worse course” than the general population, said the task force convener Robert Landewé, MD, PhD, professor of rheumatology at the University of Amsterdam.
“The second pertinent highlight is that, when it comes to managerial discussions, whether or not to stop or to start treatment for RMDs, rheumatologists should definitely be involved,” Dr. Landewé said during a live session at the annual European Congress of Rheumatology, held online this year due to COVID-19. “In practice, something that happens very often is that immunosuppressive drugs are stopped by medical specialists involved in the care of COVID but without any expertise in treating patients with rheumatic diseases. We should try to avoid that situation.”
The third highlight, something many rheumatologists may already be well aware of, is that rheumatology drugs are being used to treat COVID-19 patients without RMDs and a shortage of disease-modifying antirheumatic drugs (DMARDs) agents is a real possibility. As such, the fifth overarching highlight states that the availability of both synthetic and biologic DMARDs is “a delicate societal responsibility” and that “the off-label use of DMARDs in COVID-19 outside the context of clinical trials should be discouraged.”
The EULAR recommendation are now published online in Annals of the Rheumatic Diseases and they are “what you could call an unprecedented set of recommendations,” Dr. Landewé said. “We have never done this before,” he added, referring to the speed and way in which they had to be put together, remotely, and with little scientific evidence currently available. “Three months ago we hadn’t even heard about the virus.”
From the first patient being identified in the Hubei province of China in November 2019, to the first U.S. patient in the state of Washington on Jan. 20, 2020, and to the first European patient identified a little over 10 days later, the COVID-19 pandemic has taken the world by storm. It was only declared a pandemic on March 11, 2020, however, and Dr. Landewé noted that the response to the pandemic had been very variable – some countries locking down their borders early, while others took their time to make an appropriate response, if at all.
The rheumatology community was particularly concerned, Dr. Landewé said, because people with autoimmune diseases who were taking immunosuppressant drugs might be at higher risk for becoming infected with SARS-CoV-2, and may be at higher risk than others for a worse disease course. Thankfully, that seems not to be the case according to data that are emerging from new registries that have been set up, including EULAR’s own COVID-19 registry.
There are 13 recommendations that cover 4 themes: general measures and prevention of SARS-CoV-2 infection; the management of RMD patients during the pandemic; the management of RMD patients who have COVID-19; and the prevention of other pulmonary infections in RMD patients.
Highlighting the first three general recommendations, Dr. Landewé said: “Follow the regular guidelines in your country; if a patient with RMD does not have symptoms of COVID-19, simply continue RMD treatments,” albeit with a couple of exceptions.
The next four recommendation highlights are to avoid visits to the hospital or to the office; use remote monitoring via the telephone, for example; and if visits cannot be avoided, then take appropriate precautions. Finally, if you suspect a patient has COVID-19, do a test.
If patients test positive, then the next four recommendations cover what to do, such as continuing use of RMD treatments, but in the case of glucocorticoids this should be the lowest possible dose necessary. There is no consensus on what to do in cases of mild symptoms; the recommendation is to “decide on a case-by-case basis,” said Dr. Landewé. If a patient’s symptoms worsen, then “seek expert advice immediately and follow local treatment recommendations. The rheumatologist is not the expert to treat COVID-19,” he added. That responsibility lies with the pulmonologist, infectious disease specialist, or maybe the intensive care specialist, depending on local situations.
On the whole, the EULAR recommendations are pretty similar to those already released by the American College of Rheumatology, said Ted Mikuls, MD, of the University of Nebraska Medical Center, Omaha. The ACR recommendations are “slightly more prescriptive”, he suggested, with 25 final guidance statements. For example, general statements focused not only on the use of glucocorticoids, but also other medicines, such as antihypertensives.
“There’s really not a [lot of], I would say, major differences in the two efforts and that’s ... somewhat reassuring that we’re approaching the unknown from very different parts of the world, and driving in a very similar place,” commented Dr. Mikuls, who is a member of the ACR COVID-19 recommendations task force.
“I think one of the very important similarities that I would highlight is that, in the absence of known exposure, in the absence of COVID-19 infection, our panel felt very strongly about the importance of continuing rheumatic disease treatments,” Dr. Mikuls observed. The ACR guidelines also touch upon societal perspectives, including “some statements that were made very specific to lupus, and the use of antimalarials, given supply chain issues that we have encountered.”
Dr. Mikuls also said that the American recommendations emphasized that “you really have to manage active inflammatory rheumatic disease. Even in the context of the COVID-19 pandemic, given what we saw as the potential risk of unchecked inflammation and unchecked rheumatic disease.”
One notable difference, however, is that the European recommendations advise on immunizations and pneumonia prophylaxis, saying that all patients without COVID-19 symptoms should make sure they are up to date with any recommended vaccinations, “with a particular focus on pneumococcal and influenza vaccinations,” Dr. Landewé said.
Another difference is that the ACR recommendations are a living document and could potentially be updated monthly if the evidence arrives to allow that. In that sense, the American guidance is more agile, with EULAR expecting to update its recommendations every 3 months.
“The current evidence is extremely sparse and fragmented,” Dr. Landewé said. “We, as a task force are essentially flying blindly. We also have to cover many jurisdictions within Europe, with many conflicting opinions. So the last word to say is that updates are truly necessary, but we have to wait a while.”
SOURCE: Landewé RB et al. Ann Rheum Dis. 2020 Jun 5. doi: 10.1136/annrheumdis-2020-217877.
As might be expected, the “EULAR [European League Against Rheumatism] provisional recommendations for the management of rheumatic and musculoskeletal diseases [RMDs] in the context of SARS-CoV-2” concur with much of the guidance already released on how best to manage patients during the current pandemic.
Highlights of the five overarching principles are that, contrary to earlier expectations, “there is no indication that patients with RMDs have an additional, or have a higher, risk of contracting the virus, or that they fare a worse course” than the general population, said the task force convener Robert Landewé, MD, PhD, professor of rheumatology at the University of Amsterdam.
“The second pertinent highlight is that, when it comes to managerial discussions, whether or not to stop or to start treatment for RMDs, rheumatologists should definitely be involved,” Dr. Landewé said during a live session at the annual European Congress of Rheumatology, held online this year due to COVID-19. “In practice, something that happens very often is that immunosuppressive drugs are stopped by medical specialists involved in the care of COVID but without any expertise in treating patients with rheumatic diseases. We should try to avoid that situation.”
The third highlight, something many rheumatologists may already be well aware of, is that rheumatology drugs are being used to treat COVID-19 patients without RMDs and a shortage of disease-modifying antirheumatic drugs (DMARDs) agents is a real possibility. As such, the fifth overarching highlight states that the availability of both synthetic and biologic DMARDs is “a delicate societal responsibility” and that “the off-label use of DMARDs in COVID-19 outside the context of clinical trials should be discouraged.”
The EULAR recommendation are now published online in Annals of the Rheumatic Diseases and they are “what you could call an unprecedented set of recommendations,” Dr. Landewé said. “We have never done this before,” he added, referring to the speed and way in which they had to be put together, remotely, and with little scientific evidence currently available. “Three months ago we hadn’t even heard about the virus.”
From the first patient being identified in the Hubei province of China in November 2019, to the first U.S. patient in the state of Washington on Jan. 20, 2020, and to the first European patient identified a little over 10 days later, the COVID-19 pandemic has taken the world by storm. It was only declared a pandemic on March 11, 2020, however, and Dr. Landewé noted that the response to the pandemic had been very variable – some countries locking down their borders early, while others took their time to make an appropriate response, if at all.
The rheumatology community was particularly concerned, Dr. Landewé said, because people with autoimmune diseases who were taking immunosuppressant drugs might be at higher risk for becoming infected with SARS-CoV-2, and may be at higher risk than others for a worse disease course. Thankfully, that seems not to be the case according to data that are emerging from new registries that have been set up, including EULAR’s own COVID-19 registry.
There are 13 recommendations that cover 4 themes: general measures and prevention of SARS-CoV-2 infection; the management of RMD patients during the pandemic; the management of RMD patients who have COVID-19; and the prevention of other pulmonary infections in RMD patients.
Highlighting the first three general recommendations, Dr. Landewé said: “Follow the regular guidelines in your country; if a patient with RMD does not have symptoms of COVID-19, simply continue RMD treatments,” albeit with a couple of exceptions.
The next four recommendation highlights are to avoid visits to the hospital or to the office; use remote monitoring via the telephone, for example; and if visits cannot be avoided, then take appropriate precautions. Finally, if you suspect a patient has COVID-19, do a test.
If patients test positive, then the next four recommendations cover what to do, such as continuing use of RMD treatments, but in the case of glucocorticoids this should be the lowest possible dose necessary. There is no consensus on what to do in cases of mild symptoms; the recommendation is to “decide on a case-by-case basis,” said Dr. Landewé. If a patient’s symptoms worsen, then “seek expert advice immediately and follow local treatment recommendations. The rheumatologist is not the expert to treat COVID-19,” he added. That responsibility lies with the pulmonologist, infectious disease specialist, or maybe the intensive care specialist, depending on local situations.
On the whole, the EULAR recommendations are pretty similar to those already released by the American College of Rheumatology, said Ted Mikuls, MD, of the University of Nebraska Medical Center, Omaha. The ACR recommendations are “slightly more prescriptive”, he suggested, with 25 final guidance statements. For example, general statements focused not only on the use of glucocorticoids, but also other medicines, such as antihypertensives.
“There’s really not a [lot of], I would say, major differences in the two efforts and that’s ... somewhat reassuring that we’re approaching the unknown from very different parts of the world, and driving in a very similar place,” commented Dr. Mikuls, who is a member of the ACR COVID-19 recommendations task force.
“I think one of the very important similarities that I would highlight is that, in the absence of known exposure, in the absence of COVID-19 infection, our panel felt very strongly about the importance of continuing rheumatic disease treatments,” Dr. Mikuls observed. The ACR guidelines also touch upon societal perspectives, including “some statements that were made very specific to lupus, and the use of antimalarials, given supply chain issues that we have encountered.”
Dr. Mikuls also said that the American recommendations emphasized that “you really have to manage active inflammatory rheumatic disease. Even in the context of the COVID-19 pandemic, given what we saw as the potential risk of unchecked inflammation and unchecked rheumatic disease.”
One notable difference, however, is that the European recommendations advise on immunizations and pneumonia prophylaxis, saying that all patients without COVID-19 symptoms should make sure they are up to date with any recommended vaccinations, “with a particular focus on pneumococcal and influenza vaccinations,” Dr. Landewé said.
Another difference is that the ACR recommendations are a living document and could potentially be updated monthly if the evidence arrives to allow that. In that sense, the American guidance is more agile, with EULAR expecting to update its recommendations every 3 months.
“The current evidence is extremely sparse and fragmented,” Dr. Landewé said. “We, as a task force are essentially flying blindly. We also have to cover many jurisdictions within Europe, with many conflicting opinions. So the last word to say is that updates are truly necessary, but we have to wait a while.”
SOURCE: Landewé RB et al. Ann Rheum Dis. 2020 Jun 5. doi: 10.1136/annrheumdis-2020-217877.
As might be expected, the “EULAR [European League Against Rheumatism] provisional recommendations for the management of rheumatic and musculoskeletal diseases [RMDs] in the context of SARS-CoV-2” concur with much of the guidance already released on how best to manage patients during the current pandemic.
Highlights of the five overarching principles are that, contrary to earlier expectations, “there is no indication that patients with RMDs have an additional, or have a higher, risk of contracting the virus, or that they fare a worse course” than the general population, said the task force convener Robert Landewé, MD, PhD, professor of rheumatology at the University of Amsterdam.
“The second pertinent highlight is that, when it comes to managerial discussions, whether or not to stop or to start treatment for RMDs, rheumatologists should definitely be involved,” Dr. Landewé said during a live session at the annual European Congress of Rheumatology, held online this year due to COVID-19. “In practice, something that happens very often is that immunosuppressive drugs are stopped by medical specialists involved in the care of COVID but without any expertise in treating patients with rheumatic diseases. We should try to avoid that situation.”
The third highlight, something many rheumatologists may already be well aware of, is that rheumatology drugs are being used to treat COVID-19 patients without RMDs and a shortage of disease-modifying antirheumatic drugs (DMARDs) agents is a real possibility. As such, the fifth overarching highlight states that the availability of both synthetic and biologic DMARDs is “a delicate societal responsibility” and that “the off-label use of DMARDs in COVID-19 outside the context of clinical trials should be discouraged.”
The EULAR recommendation are now published online in Annals of the Rheumatic Diseases and they are “what you could call an unprecedented set of recommendations,” Dr. Landewé said. “We have never done this before,” he added, referring to the speed and way in which they had to be put together, remotely, and with little scientific evidence currently available. “Three months ago we hadn’t even heard about the virus.”
From the first patient being identified in the Hubei province of China in November 2019, to the first U.S. patient in the state of Washington on Jan. 20, 2020, and to the first European patient identified a little over 10 days later, the COVID-19 pandemic has taken the world by storm. It was only declared a pandemic on March 11, 2020, however, and Dr. Landewé noted that the response to the pandemic had been very variable – some countries locking down their borders early, while others took their time to make an appropriate response, if at all.
The rheumatology community was particularly concerned, Dr. Landewé said, because people with autoimmune diseases who were taking immunosuppressant drugs might be at higher risk for becoming infected with SARS-CoV-2, and may be at higher risk than others for a worse disease course. Thankfully, that seems not to be the case according to data that are emerging from new registries that have been set up, including EULAR’s own COVID-19 registry.
There are 13 recommendations that cover 4 themes: general measures and prevention of SARS-CoV-2 infection; the management of RMD patients during the pandemic; the management of RMD patients who have COVID-19; and the prevention of other pulmonary infections in RMD patients.
Highlighting the first three general recommendations, Dr. Landewé said: “Follow the regular guidelines in your country; if a patient with RMD does not have symptoms of COVID-19, simply continue RMD treatments,” albeit with a couple of exceptions.
The next four recommendation highlights are to avoid visits to the hospital or to the office; use remote monitoring via the telephone, for example; and if visits cannot be avoided, then take appropriate precautions. Finally, if you suspect a patient has COVID-19, do a test.
If patients test positive, then the next four recommendations cover what to do, such as continuing use of RMD treatments, but in the case of glucocorticoids this should be the lowest possible dose necessary. There is no consensus on what to do in cases of mild symptoms; the recommendation is to “decide on a case-by-case basis,” said Dr. Landewé. If a patient’s symptoms worsen, then “seek expert advice immediately and follow local treatment recommendations. The rheumatologist is not the expert to treat COVID-19,” he added. That responsibility lies with the pulmonologist, infectious disease specialist, or maybe the intensive care specialist, depending on local situations.
On the whole, the EULAR recommendations are pretty similar to those already released by the American College of Rheumatology, said Ted Mikuls, MD, of the University of Nebraska Medical Center, Omaha. The ACR recommendations are “slightly more prescriptive”, he suggested, with 25 final guidance statements. For example, general statements focused not only on the use of glucocorticoids, but also other medicines, such as antihypertensives.
“There’s really not a [lot of], I would say, major differences in the two efforts and that’s ... somewhat reassuring that we’re approaching the unknown from very different parts of the world, and driving in a very similar place,” commented Dr. Mikuls, who is a member of the ACR COVID-19 recommendations task force.
“I think one of the very important similarities that I would highlight is that, in the absence of known exposure, in the absence of COVID-19 infection, our panel felt very strongly about the importance of continuing rheumatic disease treatments,” Dr. Mikuls observed. The ACR guidelines also touch upon societal perspectives, including “some statements that were made very specific to lupus, and the use of antimalarials, given supply chain issues that we have encountered.”
Dr. Mikuls also said that the American recommendations emphasized that “you really have to manage active inflammatory rheumatic disease. Even in the context of the COVID-19 pandemic, given what we saw as the potential risk of unchecked inflammation and unchecked rheumatic disease.”
One notable difference, however, is that the European recommendations advise on immunizations and pneumonia prophylaxis, saying that all patients without COVID-19 symptoms should make sure they are up to date with any recommended vaccinations, “with a particular focus on pneumococcal and influenza vaccinations,” Dr. Landewé said.
Another difference is that the ACR recommendations are a living document and could potentially be updated monthly if the evidence arrives to allow that. In that sense, the American guidance is more agile, with EULAR expecting to update its recommendations every 3 months.
“The current evidence is extremely sparse and fragmented,” Dr. Landewé said. “We, as a task force are essentially flying blindly. We also have to cover many jurisdictions within Europe, with many conflicting opinions. So the last word to say is that updates are truly necessary, but we have to wait a while.”
SOURCE: Landewé RB et al. Ann Rheum Dis. 2020 Jun 5. doi: 10.1136/annrheumdis-2020-217877.
FROM THE EULAR 2020 E-CONGRESS
TNF inhibitors cut odds of VTE in RA patients
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Baloxavir effective, well tolerated for influenza treatment in children
according to Jeffrey Baker, MD, of Clinical Research Prime, Idaho Falls, and associates.
In the double-blind, randomized, controlled MiniSTONE-2 phase 3 trial, the investigators randomized 112 children aged 1-12 years to baloxavir and 57 to oseltamivir. The predominant influenza A subtype was H3N2 for both groups, followed by H1N1pdm09. Demographics and baseline characteristics were similar between treatment groups, the investigators wrote in the Pediatric Infectious Disease Journal.
The time to alleviation of signs and symptoms was a median 138 hours for those receiving baloxavir and 150 hours for those receiving oseltamivir, a nonsignificant difference. Duration of fever and of all symptoms also were similar between groups, as was the time to return to normal health and activity.
A total of 122 adverse events were reported in 84 children, with 95% of adverse events being resolved or resolving by the end of the study. The incidence of adverse events was 46% in those receiving baloxavir and 53% in those receiving oseltamivir, a nonsignificant difference, with the most common adverse event in both groups being gastrointestinal disorders. No deaths, serious adverse events, or hospitalizations were reported, but two patients receiving oseltamivir discontinued because of adverse events.
The study was funded by F. Hoffmann-La Roche. Dr. Baker and a coauthor received funding through their institutions for the conduct of the study; several coauthors reported being employed by and owning stocks in F. Hoffmann–La Roche. One coauthor reported receiving consultancy fees from F. Hoffmann–La Roche and grants from Shionogi.
SOURCE: Baker J et al. Pediatr Infect Dis J. 2020 Jun 5. doi: 10.1097/INF.0000000000002747.
according to Jeffrey Baker, MD, of Clinical Research Prime, Idaho Falls, and associates.
In the double-blind, randomized, controlled MiniSTONE-2 phase 3 trial, the investigators randomized 112 children aged 1-12 years to baloxavir and 57 to oseltamivir. The predominant influenza A subtype was H3N2 for both groups, followed by H1N1pdm09. Demographics and baseline characteristics were similar between treatment groups, the investigators wrote in the Pediatric Infectious Disease Journal.
The time to alleviation of signs and symptoms was a median 138 hours for those receiving baloxavir and 150 hours for those receiving oseltamivir, a nonsignificant difference. Duration of fever and of all symptoms also were similar between groups, as was the time to return to normal health and activity.
A total of 122 adverse events were reported in 84 children, with 95% of adverse events being resolved or resolving by the end of the study. The incidence of adverse events was 46% in those receiving baloxavir and 53% in those receiving oseltamivir, a nonsignificant difference, with the most common adverse event in both groups being gastrointestinal disorders. No deaths, serious adverse events, or hospitalizations were reported, but two patients receiving oseltamivir discontinued because of adverse events.
The study was funded by F. Hoffmann-La Roche. Dr. Baker and a coauthor received funding through their institutions for the conduct of the study; several coauthors reported being employed by and owning stocks in F. Hoffmann–La Roche. One coauthor reported receiving consultancy fees from F. Hoffmann–La Roche and grants from Shionogi.
SOURCE: Baker J et al. Pediatr Infect Dis J. 2020 Jun 5. doi: 10.1097/INF.0000000000002747.
according to Jeffrey Baker, MD, of Clinical Research Prime, Idaho Falls, and associates.
In the double-blind, randomized, controlled MiniSTONE-2 phase 3 trial, the investigators randomized 112 children aged 1-12 years to baloxavir and 57 to oseltamivir. The predominant influenza A subtype was H3N2 for both groups, followed by H1N1pdm09. Demographics and baseline characteristics were similar between treatment groups, the investigators wrote in the Pediatric Infectious Disease Journal.
The time to alleviation of signs and symptoms was a median 138 hours for those receiving baloxavir and 150 hours for those receiving oseltamivir, a nonsignificant difference. Duration of fever and of all symptoms also were similar between groups, as was the time to return to normal health and activity.
A total of 122 adverse events were reported in 84 children, with 95% of adverse events being resolved or resolving by the end of the study. The incidence of adverse events was 46% in those receiving baloxavir and 53% in those receiving oseltamivir, a nonsignificant difference, with the most common adverse event in both groups being gastrointestinal disorders. No deaths, serious adverse events, or hospitalizations were reported, but two patients receiving oseltamivir discontinued because of adverse events.
The study was funded by F. Hoffmann-La Roche. Dr. Baker and a coauthor received funding through their institutions for the conduct of the study; several coauthors reported being employed by and owning stocks in F. Hoffmann–La Roche. One coauthor reported receiving consultancy fees from F. Hoffmann–La Roche and grants from Shionogi.
SOURCE: Baker J et al. Pediatr Infect Dis J. 2020 Jun 5. doi: 10.1097/INF.0000000000002747.
FROM THE PEDIATRIC INFECTIOUS DISEASE JOURNAL
VICTORIA results deepen mystery of vericiguat in low-EF heart failure
Although clinical outcomes improved for patients with high-risk heart failure (HF) who received vericiguat (Merck/Bayer) on top of standard therapy in a major randomized trial, a subgroup study failed to show any corresponding gains in ventricular function.
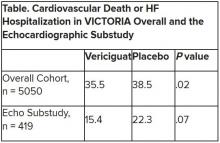
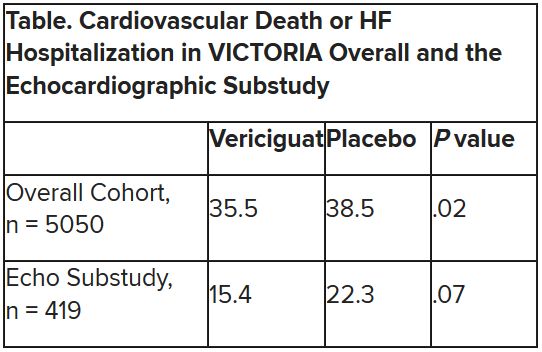
The discordant results from the 5,050-patient VICTORIA trial and its echocardiographic substudy highlight something of a mystery as to the mechanism of the investigational oral soluble guanylate cyclase stimulator’s clinical effects. In the overall trial, they included a drop in risk of cardiovascular (CV) death or first HF hospitalization, the primary endpoint.
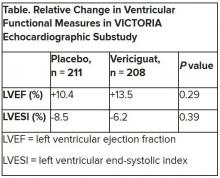
In the echo substudy, which assessed patients with evaluable echocardiograms at both baseline and 8 months, vericiguat, compared with placebo, had no significant effect on two measures of left ventricular (LV) function. Patients in the prospectively conducted substudy made up less than 10% of the total trial population.
Both LV ejection fraction (LVEF) and LV end-systolic volume index (LVESVI) significantly improved in the vericiguat and control groups, but vericiguat “had no additional significant effect,” said Burkert Pieske, MD, of Charité University Medicine Berlin.
Still, he said, there was “evidence of a lower risk of events, evidence of a clinical benefit,” for those who received vericiguat, although it fell slightly short of significance in the substudy cohort of fewer than 500 patients.
Dr. Pieske reported the VICTORIA echo substudy results June 5 in a Late-Breaking Science Session during HFA Discoveries, the online backup for the Heart Failure Association of the European Society of Cardiology annual scientific meeting.
The traditional live HFA meeting had been scheduled for Barcelona but was canceled this year as a result of the COVID-19 pandemic.
Pointing to the significant echo improvements in both treatment groups, invited discussant Rudolf A. de Boer, MD, PhD, University of Groningen (the Netherlands), said the substudy shows that HF in high-risk patients “is associated with a transient deterioration of LV function and geometry, which can to a certain extent be reversed over time.”
That the effect apparently wasn’t influenced by vericiguat “may be explained by the fact that, in randomized controlled trials, patients – including those on placebo – tend to be treated very well.” In clinical practice, he said, “less complete reverse remodeling may be expected.”
Dr. de Boer also pointed to likely survivor bias in the study, in that only patients who survived to at least 8 months were included. That meant, among other things, that they were likely at lower overall risk than the total VICTORIA population, leaving less room for any treatment effect.
“Further, likely because of the play of chance in this substudy, the LV volumes were smaller in the vericiguat group at baseline, creating less of an opportunity for vericiguat to make a difference,” he said. “It could be speculated that, with larger volumes, the window of opportunity for vericiguat would have been wider.”
But “most strikingly,” the lack of vericiguat effect on echo parameters contrasts with the clinical benefits associated with the drug in the main trial, and possibly in the echo substudy, Dr. de Boer said, “creating a dissociation between the surrogate echo parameters and the clinical hard endpoints. And it could be imagined that the rather crude echo measures presented here, LVEF and LV volume, miss a more subtle effect of vericiguat.”
For example, it’s possible that the drug’s clinical effect in heart failure does not depend on any improvements in ventricular function, Dr. de Boer said, adding that vericiguat “may potentially also have important effects on pulmonary and peripheral vasculature,” so he recommended future studies look for any changes in arterial and right ventricular function from the drug.
VICTORIA enrolled only patients with HF and reduced ejection fraction who had previously experienced a decompensation event, usually only within the last 3 months, as it turned out. Those assigned to vericiguat on top of standard drug and device therapies showed a modest 10% decline in adjusted relative risk (P = .019) for the trial’s primary endpoint, CV death or first HF hospitalization.
But when the results were unveiled at a meeting, trialists and observers were more enthused about the drug’s effect in absolute terms, which by one measure was 4.2 fewer events on vericiguat per 100 patient-years. That translated to a number to treat of 24 to prevent one event, said to be impressive, given that the study’s patients were so high risk.
The echo substudy included 419 prospectively selected patients, 208 on vericiguat and 211 assigned to placebo, who had evaluable echocardiograms at both baseline and 8 months, as assessed at the VICTORIA echo core lab. They averaged 64.5 years in age with a mean baseline LVEF of 29%; about 27% were women.
Their clinical outcomes paralleled the overall study, with lower event rates overall and a difference between treatment groups that fell short of significance.
Neither of the study’s primary endpoints, the two echo parameters, responded differently to vericiguat, compared with placebo.
The overall VICTORIA trial “showed a modest but useful benefit in the combined endpoint of hospitalizations and mortality, but all due to fewer hospitalizations,” Andrew J. Coats, MD, DSc, MBA, told this news organization.
“The echo substudy was smaller, and many drugs that reduce hospitalization do not do it through effects on LV function,” said Dr. Coats of the University of Warwick, Coventry, England, who wasn’t a part of VICTORIA. “Other mechanisms may be via improved peripheral vascular or renal effects.”
VICTORIA and the echocardiographic substudy were supported by Merck Sharp & Dohme and Bayer AG. Dr. Pieske disclosed serving on a speakers bureau, advisory board, or committee for Bayer Healthcare, Merck, Novartis, AstraZeneca, Stealth, Servier, Daiichi-Sankyo, Biotronic, Abbott Vascular, and Bristol-Myers Squibb. Dr. de Boer disclosed receiving speaker fees from Abbott, AstraZeneca, Novartis, and Roche. Dr. Coats disclosed receiving personal fees from Actimed, AstraZeneca, Faraday, WL Gore, Menarini, Novartis, Nutricia, Respicardia, Servier, Stealth Peptides, Verona, and Vifor.
A version of this article originally appeared on Medscape.com.
Although clinical outcomes improved for patients with high-risk heart failure (HF) who received vericiguat (Merck/Bayer) on top of standard therapy in a major randomized trial, a subgroup study failed to show any corresponding gains in ventricular function.
The discordant results from the 5,050-patient VICTORIA trial and its echocardiographic substudy highlight something of a mystery as to the mechanism of the investigational oral soluble guanylate cyclase stimulator’s clinical effects. In the overall trial, they included a drop in risk of cardiovascular (CV) death or first HF hospitalization, the primary endpoint.
In the echo substudy, which assessed patients with evaluable echocardiograms at both baseline and 8 months, vericiguat, compared with placebo, had no significant effect on two measures of left ventricular (LV) function. Patients in the prospectively conducted substudy made up less than 10% of the total trial population.
Both LV ejection fraction (LVEF) and LV end-systolic volume index (LVESVI) significantly improved in the vericiguat and control groups, but vericiguat “had no additional significant effect,” said Burkert Pieske, MD, of Charité University Medicine Berlin.
Still, he said, there was “evidence of a lower risk of events, evidence of a clinical benefit,” for those who received vericiguat, although it fell slightly short of significance in the substudy cohort of fewer than 500 patients.
Dr. Pieske reported the VICTORIA echo substudy results June 5 in a Late-Breaking Science Session during HFA Discoveries, the online backup for the Heart Failure Association of the European Society of Cardiology annual scientific meeting.
The traditional live HFA meeting had been scheduled for Barcelona but was canceled this year as a result of the COVID-19 pandemic.
Pointing to the significant echo improvements in both treatment groups, invited discussant Rudolf A. de Boer, MD, PhD, University of Groningen (the Netherlands), said the substudy shows that HF in high-risk patients “is associated with a transient deterioration of LV function and geometry, which can to a certain extent be reversed over time.”
That the effect apparently wasn’t influenced by vericiguat “may be explained by the fact that, in randomized controlled trials, patients – including those on placebo – tend to be treated very well.” In clinical practice, he said, “less complete reverse remodeling may be expected.”
Dr. de Boer also pointed to likely survivor bias in the study, in that only patients who survived to at least 8 months were included. That meant, among other things, that they were likely at lower overall risk than the total VICTORIA population, leaving less room for any treatment effect.
“Further, likely because of the play of chance in this substudy, the LV volumes were smaller in the vericiguat group at baseline, creating less of an opportunity for vericiguat to make a difference,” he said. “It could be speculated that, with larger volumes, the window of opportunity for vericiguat would have been wider.”
But “most strikingly,” the lack of vericiguat effect on echo parameters contrasts with the clinical benefits associated with the drug in the main trial, and possibly in the echo substudy, Dr. de Boer said, “creating a dissociation between the surrogate echo parameters and the clinical hard endpoints. And it could be imagined that the rather crude echo measures presented here, LVEF and LV volume, miss a more subtle effect of vericiguat.”
For example, it’s possible that the drug’s clinical effect in heart failure does not depend on any improvements in ventricular function, Dr. de Boer said, adding that vericiguat “may potentially also have important effects on pulmonary and peripheral vasculature,” so he recommended future studies look for any changes in arterial and right ventricular function from the drug.
VICTORIA enrolled only patients with HF and reduced ejection fraction who had previously experienced a decompensation event, usually only within the last 3 months, as it turned out. Those assigned to vericiguat on top of standard drug and device therapies showed a modest 10% decline in adjusted relative risk (P = .019) for the trial’s primary endpoint, CV death or first HF hospitalization.
But when the results were unveiled at a meeting, trialists and observers were more enthused about the drug’s effect in absolute terms, which by one measure was 4.2 fewer events on vericiguat per 100 patient-years. That translated to a number to treat of 24 to prevent one event, said to be impressive, given that the study’s patients were so high risk.
The echo substudy included 419 prospectively selected patients, 208 on vericiguat and 211 assigned to placebo, who had evaluable echocardiograms at both baseline and 8 months, as assessed at the VICTORIA echo core lab. They averaged 64.5 years in age with a mean baseline LVEF of 29%; about 27% were women.
Their clinical outcomes paralleled the overall study, with lower event rates overall and a difference between treatment groups that fell short of significance.
Neither of the study’s primary endpoints, the two echo parameters, responded differently to vericiguat, compared with placebo.
The overall VICTORIA trial “showed a modest but useful benefit in the combined endpoint of hospitalizations and mortality, but all due to fewer hospitalizations,” Andrew J. Coats, MD, DSc, MBA, told this news organization.
“The echo substudy was smaller, and many drugs that reduce hospitalization do not do it through effects on LV function,” said Dr. Coats of the University of Warwick, Coventry, England, who wasn’t a part of VICTORIA. “Other mechanisms may be via improved peripheral vascular or renal effects.”
VICTORIA and the echocardiographic substudy were supported by Merck Sharp & Dohme and Bayer AG. Dr. Pieske disclosed serving on a speakers bureau, advisory board, or committee for Bayer Healthcare, Merck, Novartis, AstraZeneca, Stealth, Servier, Daiichi-Sankyo, Biotronic, Abbott Vascular, and Bristol-Myers Squibb. Dr. de Boer disclosed receiving speaker fees from Abbott, AstraZeneca, Novartis, and Roche. Dr. Coats disclosed receiving personal fees from Actimed, AstraZeneca, Faraday, WL Gore, Menarini, Novartis, Nutricia, Respicardia, Servier, Stealth Peptides, Verona, and Vifor.
A version of this article originally appeared on Medscape.com.
Although clinical outcomes improved for patients with high-risk heart failure (HF) who received vericiguat (Merck/Bayer) on top of standard therapy in a major randomized trial, a subgroup study failed to show any corresponding gains in ventricular function.
The discordant results from the 5,050-patient VICTORIA trial and its echocardiographic substudy highlight something of a mystery as to the mechanism of the investigational oral soluble guanylate cyclase stimulator’s clinical effects. In the overall trial, they included a drop in risk of cardiovascular (CV) death or first HF hospitalization, the primary endpoint.
In the echo substudy, which assessed patients with evaluable echocardiograms at both baseline and 8 months, vericiguat, compared with placebo, had no significant effect on two measures of left ventricular (LV) function. Patients in the prospectively conducted substudy made up less than 10% of the total trial population.
Both LV ejection fraction (LVEF) and LV end-systolic volume index (LVESVI) significantly improved in the vericiguat and control groups, but vericiguat “had no additional significant effect,” said Burkert Pieske, MD, of Charité University Medicine Berlin.
Still, he said, there was “evidence of a lower risk of events, evidence of a clinical benefit,” for those who received vericiguat, although it fell slightly short of significance in the substudy cohort of fewer than 500 patients.
Dr. Pieske reported the VICTORIA echo substudy results June 5 in a Late-Breaking Science Session during HFA Discoveries, the online backup for the Heart Failure Association of the European Society of Cardiology annual scientific meeting.
The traditional live HFA meeting had been scheduled for Barcelona but was canceled this year as a result of the COVID-19 pandemic.
Pointing to the significant echo improvements in both treatment groups, invited discussant Rudolf A. de Boer, MD, PhD, University of Groningen (the Netherlands), said the substudy shows that HF in high-risk patients “is associated with a transient deterioration of LV function and geometry, which can to a certain extent be reversed over time.”
That the effect apparently wasn’t influenced by vericiguat “may be explained by the fact that, in randomized controlled trials, patients – including those on placebo – tend to be treated very well.” In clinical practice, he said, “less complete reverse remodeling may be expected.”
Dr. de Boer also pointed to likely survivor bias in the study, in that only patients who survived to at least 8 months were included. That meant, among other things, that they were likely at lower overall risk than the total VICTORIA population, leaving less room for any treatment effect.
“Further, likely because of the play of chance in this substudy, the LV volumes were smaller in the vericiguat group at baseline, creating less of an opportunity for vericiguat to make a difference,” he said. “It could be speculated that, with larger volumes, the window of opportunity for vericiguat would have been wider.”
But “most strikingly,” the lack of vericiguat effect on echo parameters contrasts with the clinical benefits associated with the drug in the main trial, and possibly in the echo substudy, Dr. de Boer said, “creating a dissociation between the surrogate echo parameters and the clinical hard endpoints. And it could be imagined that the rather crude echo measures presented here, LVEF and LV volume, miss a more subtle effect of vericiguat.”
For example, it’s possible that the drug’s clinical effect in heart failure does not depend on any improvements in ventricular function, Dr. de Boer said, adding that vericiguat “may potentially also have important effects on pulmonary and peripheral vasculature,” so he recommended future studies look for any changes in arterial and right ventricular function from the drug.
VICTORIA enrolled only patients with HF and reduced ejection fraction who had previously experienced a decompensation event, usually only within the last 3 months, as it turned out. Those assigned to vericiguat on top of standard drug and device therapies showed a modest 10% decline in adjusted relative risk (P = .019) for the trial’s primary endpoint, CV death or first HF hospitalization.
But when the results were unveiled at a meeting, trialists and observers were more enthused about the drug’s effect in absolute terms, which by one measure was 4.2 fewer events on vericiguat per 100 patient-years. That translated to a number to treat of 24 to prevent one event, said to be impressive, given that the study’s patients were so high risk.
The echo substudy included 419 prospectively selected patients, 208 on vericiguat and 211 assigned to placebo, who had evaluable echocardiograms at both baseline and 8 months, as assessed at the VICTORIA echo core lab. They averaged 64.5 years in age with a mean baseline LVEF of 29%; about 27% were women.
Their clinical outcomes paralleled the overall study, with lower event rates overall and a difference between treatment groups that fell short of significance.
Neither of the study’s primary endpoints, the two echo parameters, responded differently to vericiguat, compared with placebo.
The overall VICTORIA trial “showed a modest but useful benefit in the combined endpoint of hospitalizations and mortality, but all due to fewer hospitalizations,” Andrew J. Coats, MD, DSc, MBA, told this news organization.
“The echo substudy was smaller, and many drugs that reduce hospitalization do not do it through effects on LV function,” said Dr. Coats of the University of Warwick, Coventry, England, who wasn’t a part of VICTORIA. “Other mechanisms may be via improved peripheral vascular or renal effects.”
VICTORIA and the echocardiographic substudy were supported by Merck Sharp & Dohme and Bayer AG. Dr. Pieske disclosed serving on a speakers bureau, advisory board, or committee for Bayer Healthcare, Merck, Novartis, AstraZeneca, Stealth, Servier, Daiichi-Sankyo, Biotronic, Abbott Vascular, and Bristol-Myers Squibb. Dr. de Boer disclosed receiving speaker fees from Abbott, AstraZeneca, Novartis, and Roche. Dr. Coats disclosed receiving personal fees from Actimed, AstraZeneca, Faraday, WL Gore, Menarini, Novartis, Nutricia, Respicardia, Servier, Stealth Peptides, Verona, and Vifor.
A version of this article originally appeared on Medscape.com.
FROM ESC HEART FAILURE 2020
COVID-19: Where doctors can get help for emotional distress
Nisha Mehta, MD, said her phone has been ringing with calls from tearful and shaken physicians who are distressed and unsettled about their work and home situation and don’t know what to do.
What’s more, many frontline physicians are living apart from family to protect them from infection. “So many physicians have called me crying. ... They can’t even come home and get a hug,” Dr. Mehta said. “What I’m hearing from a lot of people who are in New York and New Jersey is not just that they go to work all day and it’s this exhausting process throughout the entire day, not only physically but also emotionally.”
Physician burnout has held a steady spotlight since long before the COVID-19 crisis began, Dr. Mehta said. “The reason for that is multifold, but in part, it’s hard for physicians to find an appropriate way to be able to process a lot of the emotions related to their work,” she said. “A lot of that brews below the surface, but COVID-19 has really brought many of these issues above that surface.”
Frustrated that governments weren’t doing enough to support health care workers during the pandemic, Dr. Mehta, a radiologist in Charlotte, N.C., decided there needed to be change. On April 4, Dr. Mehta and two physician colleagues submitted to Congress the COVID-19 Pandemic Physician Protection Act, which ensures, among other provisions, mental health coverage for health care workers. An accompanying petition on change.org had received nearly 300,000 signatures as of May 29.
Don’t suffer in silence
A career in medicine comes with immense stress in the best of times, she notes, and managing a pandemic in an already strained system has taken those challenges to newer heights. “We need better support structures at baseline for physician mental health,” said Dr. Mehta.
“That’s something we’ve always been lacking because it’s been against the culture of medicine for so long to say, ‘I’m having a hard time.’ ”
If you’re hurting, the first thing to recognize is that you are not alone in facing these challenges. This is true with respect not only to medical care but also to all of the family, financial, and business concerns physicians are currently facing. “Having all of those things hanging over your head is a lot. We’ve got to find ways to help each other out,” Dr. Mehta said.
Where to find support
Fortunately, the medical community has created several pathways to help its own.
The following list represents a cross-section of opportunities for caregivers to receive care for themselves.
Crisis hotlines
- Physician Support Line. This free and confidential hotline was launched on March 30 by Mona Masood, DO, a Philadelphia-area psychiatrist and moderator of a Facebook forum called the COVID-19 Physicians Group. The PSL is run by more than 600 volunteer psychiatrists who take calls from U.S. physicians 7 days a week from 8:00 a.m. to 1:00 a.m., with no appointment necessary. The toll-free number is 888-409-0141.
- For the Frontlines. This 24/7 help line provides free crisis counseling for frontline workers. They can text FRONTLINE to 741741 in the United States (support is also available for residents of Canada, Ireland, and the United Kingdom).
Resources from professional groups
- Action Collaborative on Clinician Well-Being and Resilience. Created by the National Academy of Medicine in 2017, the Action Collaborative comprises more than 60 organizations committed to reversing trends in clinician burnout. In response to the pandemic, the group has compiled a list of strategies and resources to support the health and well-being of clinicians who are providing healthcare during the COVID-19 outbreak.
- American Medical Association. The AMA has created a resource center dedicated to providing care for caregivers during the COVID-19 pandemic. The website includes specific guidance for managing mental health during the pandemic.
- American College of Physicians. The professional society of internal medicine physicians has created a comprehensive guide for physicians specific to COVID-19, with a section dedicated to clinician well-being that includes information about hotlines, counseling services, grief support, and more.
- American Hospital Association. The AHA’s website now includes regularly updated resources for healthcare clinicians and staff, as well as a special section dedicated to protecting and enabling healthcare workers in the midst of the pandemic.
Virtual psychological counseling
Not unlike the way telemedicine has allowed some physicians to keep seeing their patients, many modalities enable participation in therapy through video, chat, phone call, or any combination thereof. Look for a service that is convenient, flexible, and HIPAA compliant.
Traditional in-office mental health therapy has quickly moved to telemedicine. Many if not most insurers that cover counseling visits are paying for telepsychiatry or telecounseling. If you don’t know of an appropriate therapist, check the American Psychiatric Association or its state chapters; the American Psychological Association; or look for a licensed mental health counselor.
Because financial constraints are a potential barrier to therapy, Project Parachute, in cooperation with Eleos Health, has organized a cadre of therapists willing to provide pro bono online therapy for health care workers. The amount of free therapy provided to qualified frontline workers is up to the individual therapists. Discuss these parameters with your therapists up front.
Similar services are offered from companies such as Talkspace and BetterHelp on a subscription basis. These services are typically less expensive than in-person sessions. Ask about discounts for healthcare workers. Talkspace, for example, announced in March, “Effective immediately, healthcare workers across the country can get access to a free month of our...online therapy that includes unlimited text, video, and audio messaging with a licensed therapist.”
Online support groups and social media
For more on-demand peer support, look for groups such as the COR Sharing Circle for Healthcare Workers on Facebook. The site’s search engine can point users to plenty of other groups, many of which are closed (meaning posts are visible to members only).
Dr. Mehta hosts her own Facebook group called Physician Community. “I would like to think (and genuinely feel) that we’ve been doing a great job of supporting each other there with daily threads on challenges, treatments, pick-me-ups, vent posts, advocacy, and more,” she said.
For anyone in need, PeerRxMed is a free, peer-to-peer program for physicians and other health care workers that is designed to provide support, connection, encouragement, resources, and skill-building to optimize well-being.
For those craving spiritual comfort during this crisis, a number of churches have begun offering that experience virtually, too. First Unitarian Church of Worcester, Massachusetts, for example, offers weekly services via YouTube. Similar online programming is being offered from all sorts of organizations across denominations.
Apps
For DIY or on-the-spot coping support, apps can help physicians get through the day. Apps and websites that offer guided meditations and other relaxation tools include Headspace, Calm, and Insight Timer. Before downloading, look for special discounts and promotions for healthcare workers.
Additionally, COVID Coach is a free, secure app designed by the U.S. Department of Veterans Affairs that includes tools to help you cope with stress and stay well, safe, healthy, and connected. It also offers advice on navigating parenting, care giving, and working from home while social distancing, quarantined, or sheltering in place.
For practicing daily gratitude, Delightful Journal is a free app that offers journaling prompts, themes, reminders, and unlimited private space to record one’s thoughts.
Adopt a ritual
Although self-care for physicians is more crucial now than ever, it can look different for every individual. Along the same lines as keeping a journal, wellness experts often recommend beginning a “gratitude practice” to help provide solace and perspective.
Tweak and personalize these activities to suit your own needs, but be sure to use them even when you’re feeling well, said Mohana Karlekar, MD, medical director of palliative care and assistant professor at Vanderbilt University Medical Center, Nashville, Tenn.
One exercise she recommends is known as Three Good Things. “Every day, at the end of the day, think about three good things that have happened,” she explained. “You can always find the joys. And the joys don’t have to be enormous. There is joy – there is hope – in everything,” Dr. Karlekar said.
A version of this article originally appeared on Medscape.com.
Nisha Mehta, MD, said her phone has been ringing with calls from tearful and shaken physicians who are distressed and unsettled about their work and home situation and don’t know what to do.
What’s more, many frontline physicians are living apart from family to protect them from infection. “So many physicians have called me crying. ... They can’t even come home and get a hug,” Dr. Mehta said. “What I’m hearing from a lot of people who are in New York and New Jersey is not just that they go to work all day and it’s this exhausting process throughout the entire day, not only physically but also emotionally.”
Physician burnout has held a steady spotlight since long before the COVID-19 crisis began, Dr. Mehta said. “The reason for that is multifold, but in part, it’s hard for physicians to find an appropriate way to be able to process a lot of the emotions related to their work,” she said. “A lot of that brews below the surface, but COVID-19 has really brought many of these issues above that surface.”
Frustrated that governments weren’t doing enough to support health care workers during the pandemic, Dr. Mehta, a radiologist in Charlotte, N.C., decided there needed to be change. On April 4, Dr. Mehta and two physician colleagues submitted to Congress the COVID-19 Pandemic Physician Protection Act, which ensures, among other provisions, mental health coverage for health care workers. An accompanying petition on change.org had received nearly 300,000 signatures as of May 29.
Don’t suffer in silence
A career in medicine comes with immense stress in the best of times, she notes, and managing a pandemic in an already strained system has taken those challenges to newer heights. “We need better support structures at baseline for physician mental health,” said Dr. Mehta.
“That’s something we’ve always been lacking because it’s been against the culture of medicine for so long to say, ‘I’m having a hard time.’ ”
If you’re hurting, the first thing to recognize is that you are not alone in facing these challenges. This is true with respect not only to medical care but also to all of the family, financial, and business concerns physicians are currently facing. “Having all of those things hanging over your head is a lot. We’ve got to find ways to help each other out,” Dr. Mehta said.
Where to find support
Fortunately, the medical community has created several pathways to help its own.
The following list represents a cross-section of opportunities for caregivers to receive care for themselves.
Crisis hotlines
- Physician Support Line. This free and confidential hotline was launched on March 30 by Mona Masood, DO, a Philadelphia-area psychiatrist and moderator of a Facebook forum called the COVID-19 Physicians Group. The PSL is run by more than 600 volunteer psychiatrists who take calls from U.S. physicians 7 days a week from 8:00 a.m. to 1:00 a.m., with no appointment necessary. The toll-free number is 888-409-0141.
- For the Frontlines. This 24/7 help line provides free crisis counseling for frontline workers. They can text FRONTLINE to 741741 in the United States (support is also available for residents of Canada, Ireland, and the United Kingdom).
Resources from professional groups
- Action Collaborative on Clinician Well-Being and Resilience. Created by the National Academy of Medicine in 2017, the Action Collaborative comprises more than 60 organizations committed to reversing trends in clinician burnout. In response to the pandemic, the group has compiled a list of strategies and resources to support the health and well-being of clinicians who are providing healthcare during the COVID-19 outbreak.
- American Medical Association. The AMA has created a resource center dedicated to providing care for caregivers during the COVID-19 pandemic. The website includes specific guidance for managing mental health during the pandemic.
- American College of Physicians. The professional society of internal medicine physicians has created a comprehensive guide for physicians specific to COVID-19, with a section dedicated to clinician well-being that includes information about hotlines, counseling services, grief support, and more.
- American Hospital Association. The AHA’s website now includes regularly updated resources for healthcare clinicians and staff, as well as a special section dedicated to protecting and enabling healthcare workers in the midst of the pandemic.
Virtual psychological counseling
Not unlike the way telemedicine has allowed some physicians to keep seeing their patients, many modalities enable participation in therapy through video, chat, phone call, or any combination thereof. Look for a service that is convenient, flexible, and HIPAA compliant.
Traditional in-office mental health therapy has quickly moved to telemedicine. Many if not most insurers that cover counseling visits are paying for telepsychiatry or telecounseling. If you don’t know of an appropriate therapist, check the American Psychiatric Association or its state chapters; the American Psychological Association; or look for a licensed mental health counselor.
Because financial constraints are a potential barrier to therapy, Project Parachute, in cooperation with Eleos Health, has organized a cadre of therapists willing to provide pro bono online therapy for health care workers. The amount of free therapy provided to qualified frontline workers is up to the individual therapists. Discuss these parameters with your therapists up front.
Similar services are offered from companies such as Talkspace and BetterHelp on a subscription basis. These services are typically less expensive than in-person sessions. Ask about discounts for healthcare workers. Talkspace, for example, announced in March, “Effective immediately, healthcare workers across the country can get access to a free month of our...online therapy that includes unlimited text, video, and audio messaging with a licensed therapist.”
Online support groups and social media
For more on-demand peer support, look for groups such as the COR Sharing Circle for Healthcare Workers on Facebook. The site’s search engine can point users to plenty of other groups, many of which are closed (meaning posts are visible to members only).
Dr. Mehta hosts her own Facebook group called Physician Community. “I would like to think (and genuinely feel) that we’ve been doing a great job of supporting each other there with daily threads on challenges, treatments, pick-me-ups, vent posts, advocacy, and more,” she said.
For anyone in need, PeerRxMed is a free, peer-to-peer program for physicians and other health care workers that is designed to provide support, connection, encouragement, resources, and skill-building to optimize well-being.
For those craving spiritual comfort during this crisis, a number of churches have begun offering that experience virtually, too. First Unitarian Church of Worcester, Massachusetts, for example, offers weekly services via YouTube. Similar online programming is being offered from all sorts of organizations across denominations.
Apps
For DIY or on-the-spot coping support, apps can help physicians get through the day. Apps and websites that offer guided meditations and other relaxation tools include Headspace, Calm, and Insight Timer. Before downloading, look for special discounts and promotions for healthcare workers.
Additionally, COVID Coach is a free, secure app designed by the U.S. Department of Veterans Affairs that includes tools to help you cope with stress and stay well, safe, healthy, and connected. It also offers advice on navigating parenting, care giving, and working from home while social distancing, quarantined, or sheltering in place.
For practicing daily gratitude, Delightful Journal is a free app that offers journaling prompts, themes, reminders, and unlimited private space to record one’s thoughts.
Adopt a ritual
Although self-care for physicians is more crucial now than ever, it can look different for every individual. Along the same lines as keeping a journal, wellness experts often recommend beginning a “gratitude practice” to help provide solace and perspective.
Tweak and personalize these activities to suit your own needs, but be sure to use them even when you’re feeling well, said Mohana Karlekar, MD, medical director of palliative care and assistant professor at Vanderbilt University Medical Center, Nashville, Tenn.
One exercise she recommends is known as Three Good Things. “Every day, at the end of the day, think about three good things that have happened,” she explained. “You can always find the joys. And the joys don’t have to be enormous. There is joy – there is hope – in everything,” Dr. Karlekar said.
A version of this article originally appeared on Medscape.com.
Nisha Mehta, MD, said her phone has been ringing with calls from tearful and shaken physicians who are distressed and unsettled about their work and home situation and don’t know what to do.
What’s more, many frontline physicians are living apart from family to protect them from infection. “So many physicians have called me crying. ... They can’t even come home and get a hug,” Dr. Mehta said. “What I’m hearing from a lot of people who are in New York and New Jersey is not just that they go to work all day and it’s this exhausting process throughout the entire day, not only physically but also emotionally.”
Physician burnout has held a steady spotlight since long before the COVID-19 crisis began, Dr. Mehta said. “The reason for that is multifold, but in part, it’s hard for physicians to find an appropriate way to be able to process a lot of the emotions related to their work,” she said. “A lot of that brews below the surface, but COVID-19 has really brought many of these issues above that surface.”
Frustrated that governments weren’t doing enough to support health care workers during the pandemic, Dr. Mehta, a radiologist in Charlotte, N.C., decided there needed to be change. On April 4, Dr. Mehta and two physician colleagues submitted to Congress the COVID-19 Pandemic Physician Protection Act, which ensures, among other provisions, mental health coverage for health care workers. An accompanying petition on change.org had received nearly 300,000 signatures as of May 29.
Don’t suffer in silence
A career in medicine comes with immense stress in the best of times, she notes, and managing a pandemic in an already strained system has taken those challenges to newer heights. “We need better support structures at baseline for physician mental health,” said Dr. Mehta.
“That’s something we’ve always been lacking because it’s been against the culture of medicine for so long to say, ‘I’m having a hard time.’ ”
If you’re hurting, the first thing to recognize is that you are not alone in facing these challenges. This is true with respect not only to medical care but also to all of the family, financial, and business concerns physicians are currently facing. “Having all of those things hanging over your head is a lot. We’ve got to find ways to help each other out,” Dr. Mehta said.
Where to find support
Fortunately, the medical community has created several pathways to help its own.
The following list represents a cross-section of opportunities for caregivers to receive care for themselves.
Crisis hotlines
- Physician Support Line. This free and confidential hotline was launched on March 30 by Mona Masood, DO, a Philadelphia-area psychiatrist and moderator of a Facebook forum called the COVID-19 Physicians Group. The PSL is run by more than 600 volunteer psychiatrists who take calls from U.S. physicians 7 days a week from 8:00 a.m. to 1:00 a.m., with no appointment necessary. The toll-free number is 888-409-0141.
- For the Frontlines. This 24/7 help line provides free crisis counseling for frontline workers. They can text FRONTLINE to 741741 in the United States (support is also available for residents of Canada, Ireland, and the United Kingdom).
Resources from professional groups
- Action Collaborative on Clinician Well-Being and Resilience. Created by the National Academy of Medicine in 2017, the Action Collaborative comprises more than 60 organizations committed to reversing trends in clinician burnout. In response to the pandemic, the group has compiled a list of strategies and resources to support the health and well-being of clinicians who are providing healthcare during the COVID-19 outbreak.
- American Medical Association. The AMA has created a resource center dedicated to providing care for caregivers during the COVID-19 pandemic. The website includes specific guidance for managing mental health during the pandemic.
- American College of Physicians. The professional society of internal medicine physicians has created a comprehensive guide for physicians specific to COVID-19, with a section dedicated to clinician well-being that includes information about hotlines, counseling services, grief support, and more.
- American Hospital Association. The AHA’s website now includes regularly updated resources for healthcare clinicians and staff, as well as a special section dedicated to protecting and enabling healthcare workers in the midst of the pandemic.
Virtual psychological counseling
Not unlike the way telemedicine has allowed some physicians to keep seeing their patients, many modalities enable participation in therapy through video, chat, phone call, or any combination thereof. Look for a service that is convenient, flexible, and HIPAA compliant.
Traditional in-office mental health therapy has quickly moved to telemedicine. Many if not most insurers that cover counseling visits are paying for telepsychiatry or telecounseling. If you don’t know of an appropriate therapist, check the American Psychiatric Association or its state chapters; the American Psychological Association; or look for a licensed mental health counselor.
Because financial constraints are a potential barrier to therapy, Project Parachute, in cooperation with Eleos Health, has organized a cadre of therapists willing to provide pro bono online therapy for health care workers. The amount of free therapy provided to qualified frontline workers is up to the individual therapists. Discuss these parameters with your therapists up front.
Similar services are offered from companies such as Talkspace and BetterHelp on a subscription basis. These services are typically less expensive than in-person sessions. Ask about discounts for healthcare workers. Talkspace, for example, announced in March, “Effective immediately, healthcare workers across the country can get access to a free month of our...online therapy that includes unlimited text, video, and audio messaging with a licensed therapist.”
Online support groups and social media
For more on-demand peer support, look for groups such as the COR Sharing Circle for Healthcare Workers on Facebook. The site’s search engine can point users to plenty of other groups, many of which are closed (meaning posts are visible to members only).
Dr. Mehta hosts her own Facebook group called Physician Community. “I would like to think (and genuinely feel) that we’ve been doing a great job of supporting each other there with daily threads on challenges, treatments, pick-me-ups, vent posts, advocacy, and more,” she said.
For anyone in need, PeerRxMed is a free, peer-to-peer program for physicians and other health care workers that is designed to provide support, connection, encouragement, resources, and skill-building to optimize well-being.
For those craving spiritual comfort during this crisis, a number of churches have begun offering that experience virtually, too. First Unitarian Church of Worcester, Massachusetts, for example, offers weekly services via YouTube. Similar online programming is being offered from all sorts of organizations across denominations.
Apps
For DIY or on-the-spot coping support, apps can help physicians get through the day. Apps and websites that offer guided meditations and other relaxation tools include Headspace, Calm, and Insight Timer. Before downloading, look for special discounts and promotions for healthcare workers.
Additionally, COVID Coach is a free, secure app designed by the U.S. Department of Veterans Affairs that includes tools to help you cope with stress and stay well, safe, healthy, and connected. It also offers advice on navigating parenting, care giving, and working from home while social distancing, quarantined, or sheltering in place.
For practicing daily gratitude, Delightful Journal is a free app that offers journaling prompts, themes, reminders, and unlimited private space to record one’s thoughts.
Adopt a ritual
Although self-care for physicians is more crucial now than ever, it can look different for every individual. Along the same lines as keeping a journal, wellness experts often recommend beginning a “gratitude practice” to help provide solace and perspective.
Tweak and personalize these activities to suit your own needs, but be sure to use them even when you’re feeling well, said Mohana Karlekar, MD, medical director of palliative care and assistant professor at Vanderbilt University Medical Center, Nashville, Tenn.
One exercise she recommends is known as Three Good Things. “Every day, at the end of the day, think about three good things that have happened,” she explained. “You can always find the joys. And the joys don’t have to be enormous. There is joy – there is hope – in everything,” Dr. Karlekar said.
A version of this article originally appeared on Medscape.com.
Money worries during COVID-19? Six tips to keep your finances afloat
Even before Atlanta had an official shelter-in-place order, patients at the private plastic surgery practice of Nicholas Jones, MD, began canceling and rescheduled planned procedures.
After a few weeks, Dr. Jones, aged 40 years, stopped seeing patients entirely, but as a self-employed independent contractor, that means he’d lost most of his income. Dr. Jones still makes some money via a wound care job at a local nursing home, but he’s concerned that job may also be eliminated.
“I’m not hurting yet,” he said. “But I’m preparing for the worst possible scenario.”
In preparation, he and his fiancé have cut back on extraneous expenses like Uber Eats, magazine subscriptions, and streaming music services. Even though he has a 6-month emergency fund, Jones has reached out to utility companies, mortgage lenders, and student loan servicers to find out about any programs they offer to people who’ve suffered financially from the coronavirus crisis.
He’s also considered traveling to one of the COVID-19 epicenters – he has family in New Orleans and Chicago – to work in a hospital there. Jones has trauma experience and is double-boarded in general and plastic surgery.
“I could provide relief to those in need and also float through this troubled time with some financial relief,” he said.
Whereas much of the world’s attention has been on physicians who are on the front line and working around the clock in hospitals to help COVID-19 patients, thousands of other physicians are experiencing the opposite phenomenon – a slowdown or even stoppage of work (and income) altogether.
Even among those practices that remain open, the number of patients has declined as people avoid going to the office unless they absolutely have to.
At the same time, doctors in two-income households may have a spouse experiencing a job loss or income decline. Nearly 10 million Americans applied for unemployment benefits in the last 2 weeks of March, the largest number on record.
Still, while there’s uncertainty around how long the coronavirus crisis will last, experts agree that at some point America will return to a “new normal” and business operations will begin to reopen. For physicians experiencing a reduction in income who, like Jones, have an emergency fund with a few months’ worth of expenses, now’s the time to tap into it. (Or if you still have income, now’s the time to focus on growing that emergency fund to give yourself an even bigger safety net.)
If you’re among the more than half of Americans with less than 6 months of expenses saved for a rainy day, here’s how to stay afloat in the near term:
Cut back on expenses
Some household spending has naturally tapered off for many families because social distancing restrictions reduce spending on eating out, travel, and other leisure activities. But this is also an opportunity to look for other ways to reduce spending. Look through your credit card bills to see whether there are recurring payments you can cut, such as a payment to a gym that’s temporarily closed or a monthly subscription box that you don’t need.
Some gyms are not allowing membership termination right now, but it pays to ask. If a service you’re not using won’t facilitate the cancellation, call your credit card company to dispute and stop the charges, and report them to the Better Business Bureau.
You should also stop contributing to nonemergency savings accounts such as your retirement fund or your children’s college funds.
“A lot of people are hesitant to stop their automatic savings if they’ve been maxing out their 401(k) contribution or 529 accounts,” says Andrew Musbach, a certified financial planner and cofounder of MD Wealth Management in Chelsea, Mich. “But if you’re thinking long term, the reality is that missing a couple of months won’t make or break a plan. Cutting back on the amount you’re saving in the short term will increase your cash flow and is a good way to make ends meet.”
Take advantage of regulatory changes
Although many physicians won’t qualify for direct payments via the Coronavirus Aid Relief and Economic Security (CARES) Act (the $1,200 payments to individuals start phasing out once income hits $75,000 and disappear entirely for those making more than $99,000), there are other provisions in the stimulus bill that may help physicians. The bill, for example, boosts state unemployment payments by $600 per week for the next 4 months, meaning qualified workers could receive an average of nearly $1,000 per week, depending on their state, and there are new provisions providing unemployment payments to self-employed and contract workers.
The CARES Act also includes a break for federal student loan holders. Under that rule, you can skip your payments through September without incurring additional interest. Physicians in the loan forgiveness program will still get credit for payments skipped during this program.
Separately, the IRS has extended the tax deadline from April 15 to July 15, which means not only do you not have to file your taxes until then, you also don’t have to pay any taxes you owe until mid-July. The deadline for first quarter estimated tax payments has also moved to July 15. (If you’re expecting a refund, however, you should file ASAP, since the IRS will typically issue those within a few weeks of receiving your returns.)
Tap your home equity – if you’re planning to stay put
If you have good credit and still have some income, you might consider refinancing your home mortgage or opening a home equity line of credit. Interest rates have fallen recently amid economic turbulence, so if you haven’t refinanced recently you may be able to shave your monthly payment. If you need cash, a cash-out refinance, home equity line of credit, or a reverse mortgage (available if you’re over age 62) are among the lowest-cost ways to borrow.
“With interest rates so low, there can be a lot of benefit to refinancing and leveraging your house, especially if you’re planning to stay there,” says Jamie Hopkins, a director at the Carson Group. “The challenge is if you’re planning to move in the next few years. There’s a real risk that the housing market could go down in the next couple of years, and if you’re planning to sell, there’s a risk that you might not get back what you borrowed.”
Communicate early with your bank or landlord
If you don’t have the income to refinance, and you think you’re going to run into trouble making your housing payment, you should let your bank or landlord know as soon as possible. The CARES Act allows homeowners with federally backed mortgages to obtain a 180-day postponement of mortgage payments because of COVID-19 financial hardship, with the potential to extend for another 180 days. It also bans eviction by landlords with federal mortgages for 120 days.
Even if you don’t have a federally backed mortgage, you should still get in touch with your lender. Many mortgage servicers have their own forbearance programs for borrowers who can prove a temporary financial hardship. (Some banks are also waiving fees on early withdrawals on CDs and giving cardholders a reprieve on credit card payments.) Commercial landlords are also working with struggling tenants, so you may also be able to get some relief on your office lease as well.
“All of the lenders are setting up helplines for people affected,” says Amy Guerich, a partner with Stepp & Rothwell, a Kansas City–based financial planning firm. “The best thing you can do is contact them right away if you think that you’re going to have a problem vs. just letting the bills go.”
Consider retirement account withdrawals
Standard personal finance advice holds that you should exhaust all other options before pulling money out of your retirement account because of the high penalties for early withdrawals and because money removed from retirement accounts is no longer compounding over time.
Still, the CARES act has provisions making it less financially onerous to pull money from your retirement accounts. Under the new law, you can take a distribution of up to $100,000 from your IRA or 401(k) without having to pay the 10% early withdrawal penalty. You’ll owe ordinary income taxes on the withdrawal, but you have 3 years to pay them or to return the money to your retirement account.
“That’s a great relief provision, especially for higher-income physicians who might have a higher 401(k) balance,” said Jamie Hopkins.
Be smart about credit cards
Although using credit cards that you can’t pay off every month is typically an expensive way to access money, getting a new card with a low or zero percent introductory rate is a short-term strategy to consider when you’ve exhausted other options. If you have good credit, you may be able to qualify for a credit card with a 0% introductory interest rate on new transactions. Pay close attention to the fine print, including the cap on the balance you can carry without interest and whether you’ll be required to make minimum payments.
The average 0% credit card offer is for 11 months, but there are some cards that can extend the offer for up to a year-and-a-half. If you choose to use this strategy, you’ll need a plan to pay off the entire balance before the introductory period ends. If there’s a balance remaining once the rate resets, you may end up owing deferred interest on it.
The financial ramifications of the coronavirus can feel overwhelming, but it’s important not to panic. While it remains unclear how long the current crisis will last, making some smart money moves to preserve your cash in the meantime can help you stay afloat.
A version of this article originally appeared on Medscape.com.
Even before Atlanta had an official shelter-in-place order, patients at the private plastic surgery practice of Nicholas Jones, MD, began canceling and rescheduled planned procedures.
After a few weeks, Dr. Jones, aged 40 years, stopped seeing patients entirely, but as a self-employed independent contractor, that means he’d lost most of his income. Dr. Jones still makes some money via a wound care job at a local nursing home, but he’s concerned that job may also be eliminated.
“I’m not hurting yet,” he said. “But I’m preparing for the worst possible scenario.”
In preparation, he and his fiancé have cut back on extraneous expenses like Uber Eats, magazine subscriptions, and streaming music services. Even though he has a 6-month emergency fund, Jones has reached out to utility companies, mortgage lenders, and student loan servicers to find out about any programs they offer to people who’ve suffered financially from the coronavirus crisis.
He’s also considered traveling to one of the COVID-19 epicenters – he has family in New Orleans and Chicago – to work in a hospital there. Jones has trauma experience and is double-boarded in general and plastic surgery.
“I could provide relief to those in need and also float through this troubled time with some financial relief,” he said.
Whereas much of the world’s attention has been on physicians who are on the front line and working around the clock in hospitals to help COVID-19 patients, thousands of other physicians are experiencing the opposite phenomenon – a slowdown or even stoppage of work (and income) altogether.
Even among those practices that remain open, the number of patients has declined as people avoid going to the office unless they absolutely have to.
At the same time, doctors in two-income households may have a spouse experiencing a job loss or income decline. Nearly 10 million Americans applied for unemployment benefits in the last 2 weeks of March, the largest number on record.
Still, while there’s uncertainty around how long the coronavirus crisis will last, experts agree that at some point America will return to a “new normal” and business operations will begin to reopen. For physicians experiencing a reduction in income who, like Jones, have an emergency fund with a few months’ worth of expenses, now’s the time to tap into it. (Or if you still have income, now’s the time to focus on growing that emergency fund to give yourself an even bigger safety net.)
If you’re among the more than half of Americans with less than 6 months of expenses saved for a rainy day, here’s how to stay afloat in the near term:
Cut back on expenses
Some household spending has naturally tapered off for many families because social distancing restrictions reduce spending on eating out, travel, and other leisure activities. But this is also an opportunity to look for other ways to reduce spending. Look through your credit card bills to see whether there are recurring payments you can cut, such as a payment to a gym that’s temporarily closed or a monthly subscription box that you don’t need.
Some gyms are not allowing membership termination right now, but it pays to ask. If a service you’re not using won’t facilitate the cancellation, call your credit card company to dispute and stop the charges, and report them to the Better Business Bureau.
You should also stop contributing to nonemergency savings accounts such as your retirement fund or your children’s college funds.
“A lot of people are hesitant to stop their automatic savings if they’ve been maxing out their 401(k) contribution or 529 accounts,” says Andrew Musbach, a certified financial planner and cofounder of MD Wealth Management in Chelsea, Mich. “But if you’re thinking long term, the reality is that missing a couple of months won’t make or break a plan. Cutting back on the amount you’re saving in the short term will increase your cash flow and is a good way to make ends meet.”
Take advantage of regulatory changes
Although many physicians won’t qualify for direct payments via the Coronavirus Aid Relief and Economic Security (CARES) Act (the $1,200 payments to individuals start phasing out once income hits $75,000 and disappear entirely for those making more than $99,000), there are other provisions in the stimulus bill that may help physicians. The bill, for example, boosts state unemployment payments by $600 per week for the next 4 months, meaning qualified workers could receive an average of nearly $1,000 per week, depending on their state, and there are new provisions providing unemployment payments to self-employed and contract workers.
The CARES Act also includes a break for federal student loan holders. Under that rule, you can skip your payments through September without incurring additional interest. Physicians in the loan forgiveness program will still get credit for payments skipped during this program.
Separately, the IRS has extended the tax deadline from April 15 to July 15, which means not only do you not have to file your taxes until then, you also don’t have to pay any taxes you owe until mid-July. The deadline for first quarter estimated tax payments has also moved to July 15. (If you’re expecting a refund, however, you should file ASAP, since the IRS will typically issue those within a few weeks of receiving your returns.)
Tap your home equity – if you’re planning to stay put
If you have good credit and still have some income, you might consider refinancing your home mortgage or opening a home equity line of credit. Interest rates have fallen recently amid economic turbulence, so if you haven’t refinanced recently you may be able to shave your monthly payment. If you need cash, a cash-out refinance, home equity line of credit, or a reverse mortgage (available if you’re over age 62) are among the lowest-cost ways to borrow.
“With interest rates so low, there can be a lot of benefit to refinancing and leveraging your house, especially if you’re planning to stay there,” says Jamie Hopkins, a director at the Carson Group. “The challenge is if you’re planning to move in the next few years. There’s a real risk that the housing market could go down in the next couple of years, and if you’re planning to sell, there’s a risk that you might not get back what you borrowed.”
Communicate early with your bank or landlord
If you don’t have the income to refinance, and you think you’re going to run into trouble making your housing payment, you should let your bank or landlord know as soon as possible. The CARES Act allows homeowners with federally backed mortgages to obtain a 180-day postponement of mortgage payments because of COVID-19 financial hardship, with the potential to extend for another 180 days. It also bans eviction by landlords with federal mortgages for 120 days.
Even if you don’t have a federally backed mortgage, you should still get in touch with your lender. Many mortgage servicers have their own forbearance programs for borrowers who can prove a temporary financial hardship. (Some banks are also waiving fees on early withdrawals on CDs and giving cardholders a reprieve on credit card payments.) Commercial landlords are also working with struggling tenants, so you may also be able to get some relief on your office lease as well.
“All of the lenders are setting up helplines for people affected,” says Amy Guerich, a partner with Stepp & Rothwell, a Kansas City–based financial planning firm. “The best thing you can do is contact them right away if you think that you’re going to have a problem vs. just letting the bills go.”
Consider retirement account withdrawals
Standard personal finance advice holds that you should exhaust all other options before pulling money out of your retirement account because of the high penalties for early withdrawals and because money removed from retirement accounts is no longer compounding over time.
Still, the CARES act has provisions making it less financially onerous to pull money from your retirement accounts. Under the new law, you can take a distribution of up to $100,000 from your IRA or 401(k) without having to pay the 10% early withdrawal penalty. You’ll owe ordinary income taxes on the withdrawal, but you have 3 years to pay them or to return the money to your retirement account.
“That’s a great relief provision, especially for higher-income physicians who might have a higher 401(k) balance,” said Jamie Hopkins.
Be smart about credit cards
Although using credit cards that you can’t pay off every month is typically an expensive way to access money, getting a new card with a low or zero percent introductory rate is a short-term strategy to consider when you’ve exhausted other options. If you have good credit, you may be able to qualify for a credit card with a 0% introductory interest rate on new transactions. Pay close attention to the fine print, including the cap on the balance you can carry without interest and whether you’ll be required to make minimum payments.
The average 0% credit card offer is for 11 months, but there are some cards that can extend the offer for up to a year-and-a-half. If you choose to use this strategy, you’ll need a plan to pay off the entire balance before the introductory period ends. If there’s a balance remaining once the rate resets, you may end up owing deferred interest on it.
The financial ramifications of the coronavirus can feel overwhelming, but it’s important not to panic. While it remains unclear how long the current crisis will last, making some smart money moves to preserve your cash in the meantime can help you stay afloat.
A version of this article originally appeared on Medscape.com.
Even before Atlanta had an official shelter-in-place order, patients at the private plastic surgery practice of Nicholas Jones, MD, began canceling and rescheduled planned procedures.
After a few weeks, Dr. Jones, aged 40 years, stopped seeing patients entirely, but as a self-employed independent contractor, that means he’d lost most of his income. Dr. Jones still makes some money via a wound care job at a local nursing home, but he’s concerned that job may also be eliminated.
“I’m not hurting yet,” he said. “But I’m preparing for the worst possible scenario.”
In preparation, he and his fiancé have cut back on extraneous expenses like Uber Eats, magazine subscriptions, and streaming music services. Even though he has a 6-month emergency fund, Jones has reached out to utility companies, mortgage lenders, and student loan servicers to find out about any programs they offer to people who’ve suffered financially from the coronavirus crisis.
He’s also considered traveling to one of the COVID-19 epicenters – he has family in New Orleans and Chicago – to work in a hospital there. Jones has trauma experience and is double-boarded in general and plastic surgery.
“I could provide relief to those in need and also float through this troubled time with some financial relief,” he said.
Whereas much of the world’s attention has been on physicians who are on the front line and working around the clock in hospitals to help COVID-19 patients, thousands of other physicians are experiencing the opposite phenomenon – a slowdown or even stoppage of work (and income) altogether.
Even among those practices that remain open, the number of patients has declined as people avoid going to the office unless they absolutely have to.
At the same time, doctors in two-income households may have a spouse experiencing a job loss or income decline. Nearly 10 million Americans applied for unemployment benefits in the last 2 weeks of March, the largest number on record.
Still, while there’s uncertainty around how long the coronavirus crisis will last, experts agree that at some point America will return to a “new normal” and business operations will begin to reopen. For physicians experiencing a reduction in income who, like Jones, have an emergency fund with a few months’ worth of expenses, now’s the time to tap into it. (Or if you still have income, now’s the time to focus on growing that emergency fund to give yourself an even bigger safety net.)
If you’re among the more than half of Americans with less than 6 months of expenses saved for a rainy day, here’s how to stay afloat in the near term:
Cut back on expenses
Some household spending has naturally tapered off for many families because social distancing restrictions reduce spending on eating out, travel, and other leisure activities. But this is also an opportunity to look for other ways to reduce spending. Look through your credit card bills to see whether there are recurring payments you can cut, such as a payment to a gym that’s temporarily closed or a monthly subscription box that you don’t need.
Some gyms are not allowing membership termination right now, but it pays to ask. If a service you’re not using won’t facilitate the cancellation, call your credit card company to dispute and stop the charges, and report them to the Better Business Bureau.
You should also stop contributing to nonemergency savings accounts such as your retirement fund or your children’s college funds.
“A lot of people are hesitant to stop their automatic savings if they’ve been maxing out their 401(k) contribution or 529 accounts,” says Andrew Musbach, a certified financial planner and cofounder of MD Wealth Management in Chelsea, Mich. “But if you’re thinking long term, the reality is that missing a couple of months won’t make or break a plan. Cutting back on the amount you’re saving in the short term will increase your cash flow and is a good way to make ends meet.”
Take advantage of regulatory changes
Although many physicians won’t qualify for direct payments via the Coronavirus Aid Relief and Economic Security (CARES) Act (the $1,200 payments to individuals start phasing out once income hits $75,000 and disappear entirely for those making more than $99,000), there are other provisions in the stimulus bill that may help physicians. The bill, for example, boosts state unemployment payments by $600 per week for the next 4 months, meaning qualified workers could receive an average of nearly $1,000 per week, depending on their state, and there are new provisions providing unemployment payments to self-employed and contract workers.
The CARES Act also includes a break for federal student loan holders. Under that rule, you can skip your payments through September without incurring additional interest. Physicians in the loan forgiveness program will still get credit for payments skipped during this program.
Separately, the IRS has extended the tax deadline from April 15 to July 15, which means not only do you not have to file your taxes until then, you also don’t have to pay any taxes you owe until mid-July. The deadline for first quarter estimated tax payments has also moved to July 15. (If you’re expecting a refund, however, you should file ASAP, since the IRS will typically issue those within a few weeks of receiving your returns.)
Tap your home equity – if you’re planning to stay put
If you have good credit and still have some income, you might consider refinancing your home mortgage or opening a home equity line of credit. Interest rates have fallen recently amid economic turbulence, so if you haven’t refinanced recently you may be able to shave your monthly payment. If you need cash, a cash-out refinance, home equity line of credit, or a reverse mortgage (available if you’re over age 62) are among the lowest-cost ways to borrow.
“With interest rates so low, there can be a lot of benefit to refinancing and leveraging your house, especially if you’re planning to stay there,” says Jamie Hopkins, a director at the Carson Group. “The challenge is if you’re planning to move in the next few years. There’s a real risk that the housing market could go down in the next couple of years, and if you’re planning to sell, there’s a risk that you might not get back what you borrowed.”
Communicate early with your bank or landlord
If you don’t have the income to refinance, and you think you’re going to run into trouble making your housing payment, you should let your bank or landlord know as soon as possible. The CARES Act allows homeowners with federally backed mortgages to obtain a 180-day postponement of mortgage payments because of COVID-19 financial hardship, with the potential to extend for another 180 days. It also bans eviction by landlords with federal mortgages for 120 days.
Even if you don’t have a federally backed mortgage, you should still get in touch with your lender. Many mortgage servicers have their own forbearance programs for borrowers who can prove a temporary financial hardship. (Some banks are also waiving fees on early withdrawals on CDs and giving cardholders a reprieve on credit card payments.) Commercial landlords are also working with struggling tenants, so you may also be able to get some relief on your office lease as well.
“All of the lenders are setting up helplines for people affected,” says Amy Guerich, a partner with Stepp & Rothwell, a Kansas City–based financial planning firm. “The best thing you can do is contact them right away if you think that you’re going to have a problem vs. just letting the bills go.”
Consider retirement account withdrawals
Standard personal finance advice holds that you should exhaust all other options before pulling money out of your retirement account because of the high penalties for early withdrawals and because money removed from retirement accounts is no longer compounding over time.
Still, the CARES act has provisions making it less financially onerous to pull money from your retirement accounts. Under the new law, you can take a distribution of up to $100,000 from your IRA or 401(k) without having to pay the 10% early withdrawal penalty. You’ll owe ordinary income taxes on the withdrawal, but you have 3 years to pay them or to return the money to your retirement account.
“That’s a great relief provision, especially for higher-income physicians who might have a higher 401(k) balance,” said Jamie Hopkins.
Be smart about credit cards
Although using credit cards that you can’t pay off every month is typically an expensive way to access money, getting a new card with a low or zero percent introductory rate is a short-term strategy to consider when you’ve exhausted other options. If you have good credit, you may be able to qualify for a credit card with a 0% introductory interest rate on new transactions. Pay close attention to the fine print, including the cap on the balance you can carry without interest and whether you’ll be required to make minimum payments.
The average 0% credit card offer is for 11 months, but there are some cards that can extend the offer for up to a year-and-a-half. If you choose to use this strategy, you’ll need a plan to pay off the entire balance before the introductory period ends. If there’s a balance remaining once the rate resets, you may end up owing deferred interest on it.
The financial ramifications of the coronavirus can feel overwhelming, but it’s important not to panic. While it remains unclear how long the current crisis will last, making some smart money moves to preserve your cash in the meantime can help you stay afloat.
A version of this article originally appeared on Medscape.com.
Daily Recap: How to stay afloat financially during COVID-19, more bad news on e-cigs
Here are the stories our MDedge editors across specialties think you need to know about today:
Tips to keep your finances healthy during COVID-19
If you’re among the more than half of Americans with less than 6 months of expenses saved for a rainy day, here are some tips on how to stay afloat in the near term. Cut back on expenses: Look through your credit card bills to see whether there are recurring payments you can cut, such as a payment to a gym that’s temporarily closed or a monthly subscription box that you don’t need. Tap your home equity: If you have good credit and still have some income, you might consider refinancing your home mortgage or opening a home equity line of credit. Consider retirement account withdrawals: the CARES Act has provisions making it less financially onerous to pull money from your retirement accounts. Under the new law, you can take a distribution of up to $100,000 from your IRA or 401(k) without having to pay the 10% early withdrawal penalty. Read more.
Nursing homes overhaul infection control
The toll that COVID-19 has taken on nursing homes and their postacute and long-term care residents has a multilayered backstory involving underresourced organizational structures, inherent susceptibilities, minimally trained infection prevention staff, variable abilities to isolate and quarantine large numbers of patients and residents, and a lack of governmental support. “Nursing homes have been trying their best to combat this pandemic using the best infection control procedures they have, but blindfolded and with their hands tied behind their backs,” said Joseph G. Ouslander, MD, professor of geriatric medicine at Florida Atlantic University, Boca Raton. Experts in both long-term care and infectious disease said in interviews that, through the rest of the pandemic and beyond, nursing homes need the following: “Infection preventionists” to lead improvements in emergency preparedness and infection prevention and control, well-qualified and engaged medical directors, a survey/inspection process that focuses on education, and more resources and attention to structural reform. Read more.
WHO backtracks on asymptomatic SARS-CoV-2 transmission
Maria Van Kerkhove, PhD, WHO’s COVID-19 technical lead and an infectious disease epidemiologist, caused a stir on June 8 when she said that countries are reporting that many of their asymptomatic cases develop into cases of mild disease. For patients with truly asymptomatic disease, countries are “not finding secondary transmission onward. It’s very rare,” she said. But on June 9 – following a day of criticism – Dr. Van Kerkhove sought to clarify her comments on asymptomatic transmission during a live social media Q&A. She noted that while “the majority of transmission that we know about” is through individuals with symptoms, “there are a subset of people who don’t develop symptoms, and to truly understand how many people don’t have symptoms – we don’t actually have that answer yet.” Physicians and public health experts slammed the initial comments, saying that they created confusion. Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, weighed in on the controversial WHO comments, telling Good Morning America on June 10 that Dr. Van Kerkhove’s initial statement that asymptomatic SARS-CoV-2 transmission is a rare event is “not correct.” Read more.
E-cigs linked to smoking relapse
The use of electronic nicotine delivery systems is associated with increased risk of cigarette smoking relapse among former smokers, results from a large longitudinal cohort study demonstrated. The findings come from a survey of adult former smokers who participated in the Population Assessment of Tobacco and Health (PATH) Study (2013-2018). Adjusted hazard ratio (AHR) analysis revealed that the use of electronic nicotine delivery systems was associated with significant risk of cigarette smoking relapse among recent former smokers (AHR 1.63) and among long-term former smokers (AHR 3.79). The use of other tobacco products was similarly associated with a significant risk for cigarette smoking relapse among recent former smokers (AHR 1.97) and among long-term former smokers (AHR 3.82). “For the many clinicians treating former smokers who have successfully quit all nicotine products, the implications are that use of [electronic nicotine delivery systems] should be discouraged, just as use of all other tobacco products is discouraged,” researchers led by Colm D. Everard, PhD, reported in a study published in JAMA Network Open. Read more.
Formula feeding leads to early weaning
Breastfed infants who receive formula in the hospital are more than twofold more likely to wean during the first year, compared with infants who are exclusively breastfed, according to research published online in Pediatrics. The finding is based on an analysis of data from over 8,000 infants in the Minnesota Special Supplemental Nutrition Program for Women, Infants, and Children (WIC). “Our study strengthens the evidence that formula supplementation of breastfed infants negatively affects breastfeeding duration,” said Marcia Burton McCoy, MPH, of the Minnesota Department of Health’s WIC, and Pamela Heggie, MD, of the University of Minnesota in Minneapolis. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Tips to keep your finances healthy during COVID-19
If you’re among the more than half of Americans with less than 6 months of expenses saved for a rainy day, here are some tips on how to stay afloat in the near term. Cut back on expenses: Look through your credit card bills to see whether there are recurring payments you can cut, such as a payment to a gym that’s temporarily closed or a monthly subscription box that you don’t need. Tap your home equity: If you have good credit and still have some income, you might consider refinancing your home mortgage or opening a home equity line of credit. Consider retirement account withdrawals: the CARES Act has provisions making it less financially onerous to pull money from your retirement accounts. Under the new law, you can take a distribution of up to $100,000 from your IRA or 401(k) without having to pay the 10% early withdrawal penalty. Read more.
Nursing homes overhaul infection control
The toll that COVID-19 has taken on nursing homes and their postacute and long-term care residents has a multilayered backstory involving underresourced organizational structures, inherent susceptibilities, minimally trained infection prevention staff, variable abilities to isolate and quarantine large numbers of patients and residents, and a lack of governmental support. “Nursing homes have been trying their best to combat this pandemic using the best infection control procedures they have, but blindfolded and with their hands tied behind their backs,” said Joseph G. Ouslander, MD, professor of geriatric medicine at Florida Atlantic University, Boca Raton. Experts in both long-term care and infectious disease said in interviews that, through the rest of the pandemic and beyond, nursing homes need the following: “Infection preventionists” to lead improvements in emergency preparedness and infection prevention and control, well-qualified and engaged medical directors, a survey/inspection process that focuses on education, and more resources and attention to structural reform. Read more.
WHO backtracks on asymptomatic SARS-CoV-2 transmission
Maria Van Kerkhove, PhD, WHO’s COVID-19 technical lead and an infectious disease epidemiologist, caused a stir on June 8 when she said that countries are reporting that many of their asymptomatic cases develop into cases of mild disease. For patients with truly asymptomatic disease, countries are “not finding secondary transmission onward. It’s very rare,” she said. But on June 9 – following a day of criticism – Dr. Van Kerkhove sought to clarify her comments on asymptomatic transmission during a live social media Q&A. She noted that while “the majority of transmission that we know about” is through individuals with symptoms, “there are a subset of people who don’t develop symptoms, and to truly understand how many people don’t have symptoms – we don’t actually have that answer yet.” Physicians and public health experts slammed the initial comments, saying that they created confusion. Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, weighed in on the controversial WHO comments, telling Good Morning America on June 10 that Dr. Van Kerkhove’s initial statement that asymptomatic SARS-CoV-2 transmission is a rare event is “not correct.” Read more.
E-cigs linked to smoking relapse
The use of electronic nicotine delivery systems is associated with increased risk of cigarette smoking relapse among former smokers, results from a large longitudinal cohort study demonstrated. The findings come from a survey of adult former smokers who participated in the Population Assessment of Tobacco and Health (PATH) Study (2013-2018). Adjusted hazard ratio (AHR) analysis revealed that the use of electronic nicotine delivery systems was associated with significant risk of cigarette smoking relapse among recent former smokers (AHR 1.63) and among long-term former smokers (AHR 3.79). The use of other tobacco products was similarly associated with a significant risk for cigarette smoking relapse among recent former smokers (AHR 1.97) and among long-term former smokers (AHR 3.82). “For the many clinicians treating former smokers who have successfully quit all nicotine products, the implications are that use of [electronic nicotine delivery systems] should be discouraged, just as use of all other tobacco products is discouraged,” researchers led by Colm D. Everard, PhD, reported in a study published in JAMA Network Open. Read more.
Formula feeding leads to early weaning
Breastfed infants who receive formula in the hospital are more than twofold more likely to wean during the first year, compared with infants who are exclusively breastfed, according to research published online in Pediatrics. The finding is based on an analysis of data from over 8,000 infants in the Minnesota Special Supplemental Nutrition Program for Women, Infants, and Children (WIC). “Our study strengthens the evidence that formula supplementation of breastfed infants negatively affects breastfeeding duration,” said Marcia Burton McCoy, MPH, of the Minnesota Department of Health’s WIC, and Pamela Heggie, MD, of the University of Minnesota in Minneapolis. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Tips to keep your finances healthy during COVID-19
If you’re among the more than half of Americans with less than 6 months of expenses saved for a rainy day, here are some tips on how to stay afloat in the near term. Cut back on expenses: Look through your credit card bills to see whether there are recurring payments you can cut, such as a payment to a gym that’s temporarily closed or a monthly subscription box that you don’t need. Tap your home equity: If you have good credit and still have some income, you might consider refinancing your home mortgage or opening a home equity line of credit. Consider retirement account withdrawals: the CARES Act has provisions making it less financially onerous to pull money from your retirement accounts. Under the new law, you can take a distribution of up to $100,000 from your IRA or 401(k) without having to pay the 10% early withdrawal penalty. Read more.
Nursing homes overhaul infection control
The toll that COVID-19 has taken on nursing homes and their postacute and long-term care residents has a multilayered backstory involving underresourced organizational structures, inherent susceptibilities, minimally trained infection prevention staff, variable abilities to isolate and quarantine large numbers of patients and residents, and a lack of governmental support. “Nursing homes have been trying their best to combat this pandemic using the best infection control procedures they have, but blindfolded and with their hands tied behind their backs,” said Joseph G. Ouslander, MD, professor of geriatric medicine at Florida Atlantic University, Boca Raton. Experts in both long-term care and infectious disease said in interviews that, through the rest of the pandemic and beyond, nursing homes need the following: “Infection preventionists” to lead improvements in emergency preparedness and infection prevention and control, well-qualified and engaged medical directors, a survey/inspection process that focuses on education, and more resources and attention to structural reform. Read more.
WHO backtracks on asymptomatic SARS-CoV-2 transmission
Maria Van Kerkhove, PhD, WHO’s COVID-19 technical lead and an infectious disease epidemiologist, caused a stir on June 8 when she said that countries are reporting that many of their asymptomatic cases develop into cases of mild disease. For patients with truly asymptomatic disease, countries are “not finding secondary transmission onward. It’s very rare,” she said. But on June 9 – following a day of criticism – Dr. Van Kerkhove sought to clarify her comments on asymptomatic transmission during a live social media Q&A. She noted that while “the majority of transmission that we know about” is through individuals with symptoms, “there are a subset of people who don’t develop symptoms, and to truly understand how many people don’t have symptoms – we don’t actually have that answer yet.” Physicians and public health experts slammed the initial comments, saying that they created confusion. Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, weighed in on the controversial WHO comments, telling Good Morning America on June 10 that Dr. Van Kerkhove’s initial statement that asymptomatic SARS-CoV-2 transmission is a rare event is “not correct.” Read more.
E-cigs linked to smoking relapse
The use of electronic nicotine delivery systems is associated with increased risk of cigarette smoking relapse among former smokers, results from a large longitudinal cohort study demonstrated. The findings come from a survey of adult former smokers who participated in the Population Assessment of Tobacco and Health (PATH) Study (2013-2018). Adjusted hazard ratio (AHR) analysis revealed that the use of electronic nicotine delivery systems was associated with significant risk of cigarette smoking relapse among recent former smokers (AHR 1.63) and among long-term former smokers (AHR 3.79). The use of other tobacco products was similarly associated with a significant risk for cigarette smoking relapse among recent former smokers (AHR 1.97) and among long-term former smokers (AHR 3.82). “For the many clinicians treating former smokers who have successfully quit all nicotine products, the implications are that use of [electronic nicotine delivery systems] should be discouraged, just as use of all other tobacco products is discouraged,” researchers led by Colm D. Everard, PhD, reported in a study published in JAMA Network Open. Read more.
Formula feeding leads to early weaning
Breastfed infants who receive formula in the hospital are more than twofold more likely to wean during the first year, compared with infants who are exclusively breastfed, according to research published online in Pediatrics. The finding is based on an analysis of data from over 8,000 infants in the Minnesota Special Supplemental Nutrition Program for Women, Infants, and Children (WIC). “Our study strengthens the evidence that formula supplementation of breastfed infants negatively affects breastfeeding duration,” said Marcia Burton McCoy, MPH, of the Minnesota Department of Health’s WIC, and Pamela Heggie, MD, of the University of Minnesota in Minneapolis. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
No OS benefit with gefitinib vs. chemo for EGFR+ NSCLC
The median OS was 75.5 months in patients randomized to adjuvant gefitinib and 62.8 months in patients randomized to vinorelbine plus cisplatin.
Yi-Long Wu, MD, of Guangdong Lung Cancer Institute in Guangzhou, China, reported these results as part of the American Society of Clinical Oncology virtual scientific program.
Prior results from this trial had shown a disease-free survival (DFS) benefit with gefitinib, but this did not translate to an OS benefit at the final analysis, Dr. Wu said.
He noted, however, that the median OS of 75.5 months in the gefitinib arm “was one of the best in resected EGFR-mutant non–small cell lung cancer, compared with historical data.”
The findings also suggest a possible benefit with at least 18 months of gefitinib and show that adjuvant EGFR tyrosine kinase inhibitors (TKIs) should be considered the optimal therapy to improve DFS and achieve potentially better OS in this setting, Dr. Wu said.
Study details and DFS
The ADJUVANT trial (NCT01405079) randomized 222 patients, aged 18-75 years, with EGFR-mutant, stage II-IIIA (N1-N2) NSCLC who had undergone complete resection. Patients were enrolled at 27 sites between September 2011 and April 2014.
The patients were randomized 1:1 to receive 250 mg of gefitinib once daily for 24 months, or 25 mg/m2 of vinorelbine on days 1 and 8 plus 75 mg/m2 of cisplatin on day 1 every 3 weeks for 4 cycles.
The intent-to-treat (ITT) population included 111 patients in each arm. The per-protocol population included 106 patients in the gefitinib arm and 87 patients in the chemotherapy arm.
Primary results from this trial showed a significant improvement in DFS with gefitinib (Lancet Oncol. 2018 Jan;19[1]:139-48). That improvement was maintained in the final analysis.
The median DFS was 30.8 months in the gefitinib arm and 19.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The hazard ratio (HR) was 0.56 (P = .001) in the ITT population and 0.51 (P < .001) in the per-protocol population.
In the ITT population, the 5-year DFS rates were 22.6% in the gefitinib arm and 23.2% in the chemotherapy arm. In the per-protocol population, the 5-year DFS rates were 22.6% and 22.8%, respectively.
OS results
The median OS was 75.5 months in the gefitinib arm and 62.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The HR was 0.92 in both the ITT (P = .674) and per-protocol populations (P = .686).
In the ITT population, the 5-year OS rates were 53.2% in the gefitinib arm and 51.2% in the chemotherapy arm. In the per-protocol population, the 5-year OS rates were 53.2% and 50.7%, respectively.
Subgroup analyses by age, gender, lymph node status, and EGFR mutation showed trends toward improved OS with gefitinib, but the differences were not statistically significant.
The researchers conducted a post hoc analysis to assess the effect of subsequent treatment on patient outcomes. The analysis showed that patients who received gefitinib with subsequent EGFR-TKIs had the best responses and OS.
The median OS was not reached among patients who received gefitinib and subsequent EGFR-TKIs, whereas the median OS ranged from 15.6 months to 62.8 months in other groups. The shortest OS was observed in patients who received adjuvant chemotherapy without subsequent therapy.
The duration of gefitinib treatment also appeared to affect OS. The median OS was 35.7 months in patients who received gefitinib for less than 18 months, and the median OS was not reached in patients who received gefitinib for 18 months or longer (HR, 0.38; P < .001).
Implications and potential next steps
Despite the lack of OS improvement with gefitinib, “all of the patients on this study did much, much better than historical non–small cell lung cancer not specified by the EGFR mutation, with 70 months median survival compared to 35 months median survival for N2-positive disease,” said invited discussant Christopher G. Azzoli, MD, director of thoracic oncology at Lifespan Cancer Institute at Brown University in Providence, R.I.
“But you can’t avoid noticing how the curves come back together in terms of disease-free survival when your effective treatment is limited to 24 months,” he added.
An apparent risk of late brain recurrence in the gefitinib arm is also a concern, Dr. Azzoli said. “So ... longer duration of treatment with a drug that has better control of CNS [central nervous system] disease, such as osimertinib, may improve both DFS and OS,” he added.
Only about 50% of patients in the chemotherapy arm received a TKI at recurrence. The post hoc analysis showing that TKI recipients had the best outcomes raises the question of whether “the survival benefit could be conferred by delivering a superior drug merely at recurrence, or is there benefit to earlier delivery of an effective drug,” Dr. Azzoli said.
Given the high cost of continuous therapy, biomarker refinement could help improve treatment decision-making, he said, noting that “early testing of blood DNA to detect cancer in the body as minimal residual disease is showing promise,” and that many phase 3 studies of EGFR-TKIs are ongoing.
The current trial was sponsored by the Guangdong Association of Clinical Trials. Dr. Wu disclosed relationships with AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb/China, Lilly, MSD Oncology, Pfizer, and Roche. Dr. Azzoli reported having no disclosures.
SOURCE: Wu Y et al. ASCO 2020, Abstract 9005.
The median OS was 75.5 months in patients randomized to adjuvant gefitinib and 62.8 months in patients randomized to vinorelbine plus cisplatin.
Yi-Long Wu, MD, of Guangdong Lung Cancer Institute in Guangzhou, China, reported these results as part of the American Society of Clinical Oncology virtual scientific program.
Prior results from this trial had shown a disease-free survival (DFS) benefit with gefitinib, but this did not translate to an OS benefit at the final analysis, Dr. Wu said.
He noted, however, that the median OS of 75.5 months in the gefitinib arm “was one of the best in resected EGFR-mutant non–small cell lung cancer, compared with historical data.”
The findings also suggest a possible benefit with at least 18 months of gefitinib and show that adjuvant EGFR tyrosine kinase inhibitors (TKIs) should be considered the optimal therapy to improve DFS and achieve potentially better OS in this setting, Dr. Wu said.
Study details and DFS
The ADJUVANT trial (NCT01405079) randomized 222 patients, aged 18-75 years, with EGFR-mutant, stage II-IIIA (N1-N2) NSCLC who had undergone complete resection. Patients were enrolled at 27 sites between September 2011 and April 2014.
The patients were randomized 1:1 to receive 250 mg of gefitinib once daily for 24 months, or 25 mg/m2 of vinorelbine on days 1 and 8 plus 75 mg/m2 of cisplatin on day 1 every 3 weeks for 4 cycles.
The intent-to-treat (ITT) population included 111 patients in each arm. The per-protocol population included 106 patients in the gefitinib arm and 87 patients in the chemotherapy arm.
Primary results from this trial showed a significant improvement in DFS with gefitinib (Lancet Oncol. 2018 Jan;19[1]:139-48). That improvement was maintained in the final analysis.
The median DFS was 30.8 months in the gefitinib arm and 19.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The hazard ratio (HR) was 0.56 (P = .001) in the ITT population and 0.51 (P < .001) in the per-protocol population.
In the ITT population, the 5-year DFS rates were 22.6% in the gefitinib arm and 23.2% in the chemotherapy arm. In the per-protocol population, the 5-year DFS rates were 22.6% and 22.8%, respectively.
OS results
The median OS was 75.5 months in the gefitinib arm and 62.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The HR was 0.92 in both the ITT (P = .674) and per-protocol populations (P = .686).
In the ITT population, the 5-year OS rates were 53.2% in the gefitinib arm and 51.2% in the chemotherapy arm. In the per-protocol population, the 5-year OS rates were 53.2% and 50.7%, respectively.
Subgroup analyses by age, gender, lymph node status, and EGFR mutation showed trends toward improved OS with gefitinib, but the differences were not statistically significant.
The researchers conducted a post hoc analysis to assess the effect of subsequent treatment on patient outcomes. The analysis showed that patients who received gefitinib with subsequent EGFR-TKIs had the best responses and OS.
The median OS was not reached among patients who received gefitinib and subsequent EGFR-TKIs, whereas the median OS ranged from 15.6 months to 62.8 months in other groups. The shortest OS was observed in patients who received adjuvant chemotherapy without subsequent therapy.
The duration of gefitinib treatment also appeared to affect OS. The median OS was 35.7 months in patients who received gefitinib for less than 18 months, and the median OS was not reached in patients who received gefitinib for 18 months or longer (HR, 0.38; P < .001).
Implications and potential next steps
Despite the lack of OS improvement with gefitinib, “all of the patients on this study did much, much better than historical non–small cell lung cancer not specified by the EGFR mutation, with 70 months median survival compared to 35 months median survival for N2-positive disease,” said invited discussant Christopher G. Azzoli, MD, director of thoracic oncology at Lifespan Cancer Institute at Brown University in Providence, R.I.
“But you can’t avoid noticing how the curves come back together in terms of disease-free survival when your effective treatment is limited to 24 months,” he added.
An apparent risk of late brain recurrence in the gefitinib arm is also a concern, Dr. Azzoli said. “So ... longer duration of treatment with a drug that has better control of CNS [central nervous system] disease, such as osimertinib, may improve both DFS and OS,” he added.
Only about 50% of patients in the chemotherapy arm received a TKI at recurrence. The post hoc analysis showing that TKI recipients had the best outcomes raises the question of whether “the survival benefit could be conferred by delivering a superior drug merely at recurrence, or is there benefit to earlier delivery of an effective drug,” Dr. Azzoli said.
Given the high cost of continuous therapy, biomarker refinement could help improve treatment decision-making, he said, noting that “early testing of blood DNA to detect cancer in the body as minimal residual disease is showing promise,” and that many phase 3 studies of EGFR-TKIs are ongoing.
The current trial was sponsored by the Guangdong Association of Clinical Trials. Dr. Wu disclosed relationships with AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb/China, Lilly, MSD Oncology, Pfizer, and Roche. Dr. Azzoli reported having no disclosures.
SOURCE: Wu Y et al. ASCO 2020, Abstract 9005.
The median OS was 75.5 months in patients randomized to adjuvant gefitinib and 62.8 months in patients randomized to vinorelbine plus cisplatin.
Yi-Long Wu, MD, of Guangdong Lung Cancer Institute in Guangzhou, China, reported these results as part of the American Society of Clinical Oncology virtual scientific program.
Prior results from this trial had shown a disease-free survival (DFS) benefit with gefitinib, but this did not translate to an OS benefit at the final analysis, Dr. Wu said.
He noted, however, that the median OS of 75.5 months in the gefitinib arm “was one of the best in resected EGFR-mutant non–small cell lung cancer, compared with historical data.”
The findings also suggest a possible benefit with at least 18 months of gefitinib and show that adjuvant EGFR tyrosine kinase inhibitors (TKIs) should be considered the optimal therapy to improve DFS and achieve potentially better OS in this setting, Dr. Wu said.
Study details and DFS
The ADJUVANT trial (NCT01405079) randomized 222 patients, aged 18-75 years, with EGFR-mutant, stage II-IIIA (N1-N2) NSCLC who had undergone complete resection. Patients were enrolled at 27 sites between September 2011 and April 2014.
The patients were randomized 1:1 to receive 250 mg of gefitinib once daily for 24 months, or 25 mg/m2 of vinorelbine on days 1 and 8 plus 75 mg/m2 of cisplatin on day 1 every 3 weeks for 4 cycles.
The intent-to-treat (ITT) population included 111 patients in each arm. The per-protocol population included 106 patients in the gefitinib arm and 87 patients in the chemotherapy arm.
Primary results from this trial showed a significant improvement in DFS with gefitinib (Lancet Oncol. 2018 Jan;19[1]:139-48). That improvement was maintained in the final analysis.
The median DFS was 30.8 months in the gefitinib arm and 19.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The hazard ratio (HR) was 0.56 (P = .001) in the ITT population and 0.51 (P < .001) in the per-protocol population.
In the ITT population, the 5-year DFS rates were 22.6% in the gefitinib arm and 23.2% in the chemotherapy arm. In the per-protocol population, the 5-year DFS rates were 22.6% and 22.8%, respectively.
OS results
The median OS was 75.5 months in the gefitinib arm and 62.8 months in the chemotherapy arm for both the ITT and per-protocol populations. The HR was 0.92 in both the ITT (P = .674) and per-protocol populations (P = .686).
In the ITT population, the 5-year OS rates were 53.2% in the gefitinib arm and 51.2% in the chemotherapy arm. In the per-protocol population, the 5-year OS rates were 53.2% and 50.7%, respectively.
Subgroup analyses by age, gender, lymph node status, and EGFR mutation showed trends toward improved OS with gefitinib, but the differences were not statistically significant.
The researchers conducted a post hoc analysis to assess the effect of subsequent treatment on patient outcomes. The analysis showed that patients who received gefitinib with subsequent EGFR-TKIs had the best responses and OS.
The median OS was not reached among patients who received gefitinib and subsequent EGFR-TKIs, whereas the median OS ranged from 15.6 months to 62.8 months in other groups. The shortest OS was observed in patients who received adjuvant chemotherapy without subsequent therapy.
The duration of gefitinib treatment also appeared to affect OS. The median OS was 35.7 months in patients who received gefitinib for less than 18 months, and the median OS was not reached in patients who received gefitinib for 18 months or longer (HR, 0.38; P < .001).
Implications and potential next steps
Despite the lack of OS improvement with gefitinib, “all of the patients on this study did much, much better than historical non–small cell lung cancer not specified by the EGFR mutation, with 70 months median survival compared to 35 months median survival for N2-positive disease,” said invited discussant Christopher G. Azzoli, MD, director of thoracic oncology at Lifespan Cancer Institute at Brown University in Providence, R.I.
“But you can’t avoid noticing how the curves come back together in terms of disease-free survival when your effective treatment is limited to 24 months,” he added.
An apparent risk of late brain recurrence in the gefitinib arm is also a concern, Dr. Azzoli said. “So ... longer duration of treatment with a drug that has better control of CNS [central nervous system] disease, such as osimertinib, may improve both DFS and OS,” he added.
Only about 50% of patients in the chemotherapy arm received a TKI at recurrence. The post hoc analysis showing that TKI recipients had the best outcomes raises the question of whether “the survival benefit could be conferred by delivering a superior drug merely at recurrence, or is there benefit to earlier delivery of an effective drug,” Dr. Azzoli said.
Given the high cost of continuous therapy, biomarker refinement could help improve treatment decision-making, he said, noting that “early testing of blood DNA to detect cancer in the body as minimal residual disease is showing promise,” and that many phase 3 studies of EGFR-TKIs are ongoing.
The current trial was sponsored by the Guangdong Association of Clinical Trials. Dr. Wu disclosed relationships with AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb/China, Lilly, MSD Oncology, Pfizer, and Roche. Dr. Azzoli reported having no disclosures.
SOURCE: Wu Y et al. ASCO 2020, Abstract 9005.
FROM ASCO 2020