User login
-
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]


Antibiotic-resistant bacteria emerging in community settings
A new study from the Centers for Disease Control and Prevention found that
Traditionally, CRE has been thought of as a nosocomial infection, acquired in a hospital or other health care facility (nursing home, long-term acute care hospital, dialysis center, etc.). This is the first population-level study to show otherwise, with fully 10% of the CRE isolates found to be community acquired.
CREs are a group of multidrug-resistant bacteria considered an urgent health threat by the CDC because they can rapidly spread between patients, especially those who are most seriously ill and vulnerable, and because they are so difficult to treat. These patients often require treatment with toxic antibiotics, such as colistin, and carry a high mortality rate – up to 50% in some studies.
Overall, 30% of CREs carry a carbapenemase – an enzyme that can make them resistant to carbapenem antibiotics. The genes for this are readily transferable between bacteria and help account for their spread in hospitals.
But in this study, published in the American Journal of Infection Control, of the 12 isolates that underwent whole-genome sequencing, 42% of the CA-CRE isolates carried the carbapenemase gene. Lead author Sandra Bulens, MPH, a health scientist in the CDC’s division of health care quality promotion, said in an interview, “The findings highlight the potential for CP-CRE to move from health care settings into the community. The fact that 5 of the 12 isolates harbored a carbapenemase gene introduces new challenges for controlling spread of CP-CRE.”
CDC researchers analyzed data from eight U.S. metropolitan areas between 2012 and 2015 as part of the CDC’s Emerging Infections Program (EIP) health care–associated infections – community interface activity, which conducts surveillance for CRE and other drug-resistant gram-negative bacteria. Cases of CA-CRE were compared with HCA-CRE, with 1499 cases in 1,194 case-patients being analyzed. Though Klebsiella pneumoniae was the most common isolate, there were some differences between metropolitan areas.
The incidence of CRE cases per 100,000 population was 2.96 (95% confidence interval, 2.81-3.11) overall and 0.29 (95% CI, 0.25-0.25) for CA-CRE. Most CA-CRE cases were in White persons (73%) and women (84%). Urine cultures were the source of 98% of all CA-CRE cases, compared with 86% of HCA-CRE cases (P < .001). Though small numbers, the numbers of patients with CA-CRE without apparent underlying medical condition (n = 51; 37%) was greater when compared with patients with HCA-CRE (n = 36; 3%; P < .001).
Asked for independent comment, Lance Price, PhD, of George Washington University and the founding director of GW’s Antibiotic Resistance Action Center, Washington, said, “what’s striking about these data is that: ‘Who is the front line, at least in the United States for CRE?’ It’s women, older women. ... At some point, we have to frame drug resistance as a women’s health issue.”
Dr. Price noted that the 10% of patients with CA-CRE acquired it in the community. “I would argue that probably none of them had any idea, because there’s this silent community epidemic,” he said. “It’s asymptomatic carriage and transmission in the community. Somebody can be this walking reservoir of these really dangerous bacteria and have no idea.”
This is an increasingly serious problem for women, Dr. Price said, because, “with a community-acquired bladder infection, you’re going to call your doctor or go to an urgent care, and they’re not going to test you. They’re going to guess what you have, and they’re going to prescribe an antibiotic, and that antibiotic is going to fail. So then your bladder infection continues, and then you wait a few more days, and you start to get flank pain and kidney infection. ... If you start getting a fever, they might admit you. They are going to start treating you immediately, and they might miss it because you’ve got this organism that’s resistant to all the best antibiotics. ... The gateway to the blood is the UTI.”
Because of such empiric treatment and increasing resistance, the risk for treatment failure is quite high, especially for older women. Ms. Bulens, however, said that, “[although] 10% of CRE were in persons without health care risk factors, the proportion of all UTIs in this population that are CRE is going to be very, very small.”
This study involved cultures from 2012 to 2015. Before the pandemic, from 2012 to 2017, U.S. deaths from antibiotic resistance fell by 18% overall and by 30% in hospitals.
But in the first year of the COVID-19 pandemic, there was a 15% increase in infections and deaths from antibiotic-resistant (AMR), hospital-acquired bacteria. In 2020, 29,400 patients died from AMR infections. There was a 78% increase in carbapenem-resistant Acinetobacter baumannii health care–associated infections, a 35% increase in carbapenem-resistant Enterobacterales, and 32% increases in both multidrug-resistant Pseudomonas aeruginosa and extended-spectrum beta-lactamase–producing Enterobacterales. Aside from gram-negative bacteria, methicillin-resistant Staphylococcus aureus rose 13%, and Candida auris rose 60%. But owing to limited surveillance, recent sound figures are lacking.
Ms. Bulens and Dr. Price reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study from the Centers for Disease Control and Prevention found that
Traditionally, CRE has been thought of as a nosocomial infection, acquired in a hospital or other health care facility (nursing home, long-term acute care hospital, dialysis center, etc.). This is the first population-level study to show otherwise, with fully 10% of the CRE isolates found to be community acquired.
CREs are a group of multidrug-resistant bacteria considered an urgent health threat by the CDC because they can rapidly spread between patients, especially those who are most seriously ill and vulnerable, and because they are so difficult to treat. These patients often require treatment with toxic antibiotics, such as colistin, and carry a high mortality rate – up to 50% in some studies.
Overall, 30% of CREs carry a carbapenemase – an enzyme that can make them resistant to carbapenem antibiotics. The genes for this are readily transferable between bacteria and help account for their spread in hospitals.
But in this study, published in the American Journal of Infection Control, of the 12 isolates that underwent whole-genome sequencing, 42% of the CA-CRE isolates carried the carbapenemase gene. Lead author Sandra Bulens, MPH, a health scientist in the CDC’s division of health care quality promotion, said in an interview, “The findings highlight the potential for CP-CRE to move from health care settings into the community. The fact that 5 of the 12 isolates harbored a carbapenemase gene introduces new challenges for controlling spread of CP-CRE.”
CDC researchers analyzed data from eight U.S. metropolitan areas between 2012 and 2015 as part of the CDC’s Emerging Infections Program (EIP) health care–associated infections – community interface activity, which conducts surveillance for CRE and other drug-resistant gram-negative bacteria. Cases of CA-CRE were compared with HCA-CRE, with 1499 cases in 1,194 case-patients being analyzed. Though Klebsiella pneumoniae was the most common isolate, there were some differences between metropolitan areas.
The incidence of CRE cases per 100,000 population was 2.96 (95% confidence interval, 2.81-3.11) overall and 0.29 (95% CI, 0.25-0.25) for CA-CRE. Most CA-CRE cases were in White persons (73%) and women (84%). Urine cultures were the source of 98% of all CA-CRE cases, compared with 86% of HCA-CRE cases (P < .001). Though small numbers, the numbers of patients with CA-CRE without apparent underlying medical condition (n = 51; 37%) was greater when compared with patients with HCA-CRE (n = 36; 3%; P < .001).
Asked for independent comment, Lance Price, PhD, of George Washington University and the founding director of GW’s Antibiotic Resistance Action Center, Washington, said, “what’s striking about these data is that: ‘Who is the front line, at least in the United States for CRE?’ It’s women, older women. ... At some point, we have to frame drug resistance as a women’s health issue.”
Dr. Price noted that the 10% of patients with CA-CRE acquired it in the community. “I would argue that probably none of them had any idea, because there’s this silent community epidemic,” he said. “It’s asymptomatic carriage and transmission in the community. Somebody can be this walking reservoir of these really dangerous bacteria and have no idea.”
This is an increasingly serious problem for women, Dr. Price said, because, “with a community-acquired bladder infection, you’re going to call your doctor or go to an urgent care, and they’re not going to test you. They’re going to guess what you have, and they’re going to prescribe an antibiotic, and that antibiotic is going to fail. So then your bladder infection continues, and then you wait a few more days, and you start to get flank pain and kidney infection. ... If you start getting a fever, they might admit you. They are going to start treating you immediately, and they might miss it because you’ve got this organism that’s resistant to all the best antibiotics. ... The gateway to the blood is the UTI.”
Because of such empiric treatment and increasing resistance, the risk for treatment failure is quite high, especially for older women. Ms. Bulens, however, said that, “[although] 10% of CRE were in persons without health care risk factors, the proportion of all UTIs in this population that are CRE is going to be very, very small.”
This study involved cultures from 2012 to 2015. Before the pandemic, from 2012 to 2017, U.S. deaths from antibiotic resistance fell by 18% overall and by 30% in hospitals.
But in the first year of the COVID-19 pandemic, there was a 15% increase in infections and deaths from antibiotic-resistant (AMR), hospital-acquired bacteria. In 2020, 29,400 patients died from AMR infections. There was a 78% increase in carbapenem-resistant Acinetobacter baumannii health care–associated infections, a 35% increase in carbapenem-resistant Enterobacterales, and 32% increases in both multidrug-resistant Pseudomonas aeruginosa and extended-spectrum beta-lactamase–producing Enterobacterales. Aside from gram-negative bacteria, methicillin-resistant Staphylococcus aureus rose 13%, and Candida auris rose 60%. But owing to limited surveillance, recent sound figures are lacking.
Ms. Bulens and Dr. Price reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study from the Centers for Disease Control and Prevention found that
Traditionally, CRE has been thought of as a nosocomial infection, acquired in a hospital or other health care facility (nursing home, long-term acute care hospital, dialysis center, etc.). This is the first population-level study to show otherwise, with fully 10% of the CRE isolates found to be community acquired.
CREs are a group of multidrug-resistant bacteria considered an urgent health threat by the CDC because they can rapidly spread between patients, especially those who are most seriously ill and vulnerable, and because they are so difficult to treat. These patients often require treatment with toxic antibiotics, such as colistin, and carry a high mortality rate – up to 50% in some studies.
Overall, 30% of CREs carry a carbapenemase – an enzyme that can make them resistant to carbapenem antibiotics. The genes for this are readily transferable between bacteria and help account for their spread in hospitals.
But in this study, published in the American Journal of Infection Control, of the 12 isolates that underwent whole-genome sequencing, 42% of the CA-CRE isolates carried the carbapenemase gene. Lead author Sandra Bulens, MPH, a health scientist in the CDC’s division of health care quality promotion, said in an interview, “The findings highlight the potential for CP-CRE to move from health care settings into the community. The fact that 5 of the 12 isolates harbored a carbapenemase gene introduces new challenges for controlling spread of CP-CRE.”
CDC researchers analyzed data from eight U.S. metropolitan areas between 2012 and 2015 as part of the CDC’s Emerging Infections Program (EIP) health care–associated infections – community interface activity, which conducts surveillance for CRE and other drug-resistant gram-negative bacteria. Cases of CA-CRE were compared with HCA-CRE, with 1499 cases in 1,194 case-patients being analyzed. Though Klebsiella pneumoniae was the most common isolate, there were some differences between metropolitan areas.
The incidence of CRE cases per 100,000 population was 2.96 (95% confidence interval, 2.81-3.11) overall and 0.29 (95% CI, 0.25-0.25) for CA-CRE. Most CA-CRE cases were in White persons (73%) and women (84%). Urine cultures were the source of 98% of all CA-CRE cases, compared with 86% of HCA-CRE cases (P < .001). Though small numbers, the numbers of patients with CA-CRE without apparent underlying medical condition (n = 51; 37%) was greater when compared with patients with HCA-CRE (n = 36; 3%; P < .001).
Asked for independent comment, Lance Price, PhD, of George Washington University and the founding director of GW’s Antibiotic Resistance Action Center, Washington, said, “what’s striking about these data is that: ‘Who is the front line, at least in the United States for CRE?’ It’s women, older women. ... At some point, we have to frame drug resistance as a women’s health issue.”
Dr. Price noted that the 10% of patients with CA-CRE acquired it in the community. “I would argue that probably none of them had any idea, because there’s this silent community epidemic,” he said. “It’s asymptomatic carriage and transmission in the community. Somebody can be this walking reservoir of these really dangerous bacteria and have no idea.”
This is an increasingly serious problem for women, Dr. Price said, because, “with a community-acquired bladder infection, you’re going to call your doctor or go to an urgent care, and they’re not going to test you. They’re going to guess what you have, and they’re going to prescribe an antibiotic, and that antibiotic is going to fail. So then your bladder infection continues, and then you wait a few more days, and you start to get flank pain and kidney infection. ... If you start getting a fever, they might admit you. They are going to start treating you immediately, and they might miss it because you’ve got this organism that’s resistant to all the best antibiotics. ... The gateway to the blood is the UTI.”
Because of such empiric treatment and increasing resistance, the risk for treatment failure is quite high, especially for older women. Ms. Bulens, however, said that, “[although] 10% of CRE were in persons without health care risk factors, the proportion of all UTIs in this population that are CRE is going to be very, very small.”
This study involved cultures from 2012 to 2015. Before the pandemic, from 2012 to 2017, U.S. deaths from antibiotic resistance fell by 18% overall and by 30% in hospitals.
But in the first year of the COVID-19 pandemic, there was a 15% increase in infections and deaths from antibiotic-resistant (AMR), hospital-acquired bacteria. In 2020, 29,400 patients died from AMR infections. There was a 78% increase in carbapenem-resistant Acinetobacter baumannii health care–associated infections, a 35% increase in carbapenem-resistant Enterobacterales, and 32% increases in both multidrug-resistant Pseudomonas aeruginosa and extended-spectrum beta-lactamase–producing Enterobacterales. Aside from gram-negative bacteria, methicillin-resistant Staphylococcus aureus rose 13%, and Candida auris rose 60%. But owing to limited surveillance, recent sound figures are lacking.
Ms. Bulens and Dr. Price reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF INFECTION CONTROL
Thoracic Oncology & Chest Procedures Network
Ultrasound and Chest Imaging Section
Advanced critical care echocardiography: A noninvasive tool for hemodynamic assessment in critically ill patients
Hemodynamic assessments in critically ill patients are important to guide accurate management; however, traditional invasive methods of measuring cardiac output have significant limitations, including risks of infection and bleeding. ACCE can provide a multitude of hemodynamic measurements from cardiac output (CO) to right ventricular systolic pressure (RVSP) and left atrial pressure (LAP). Combinations of left ventricular function parameters, along with estimation of filling pressures, can help distinguish between types of shock. Schmidt and colleagues (Sci Rep. 2022;12[1]:7187) demonstrated that measurement of these indices in the majority of patients helped elucidate the cause for hemodynamic compromise. They found presence of a cardiac index (CI) < 2.5/min.m2 was associated with a doubling of ICU mortality as compared with predictions based on severity of illness scores in otherwise hemodynamically stable patients. Hollenberg and colleagues (Am J Cardiol. 2021;153:135-39) demonstrated the feasibility of a simpler stratification using the left ventricular ejection fraction (LVEF) and CI in coronavirus disease 2019 patients with shock, where low CI despite having a preserved LVEF was associated with worse outcomes.
Quick, reliable data are an intensivist’s friend. Utilizing ACCE at the bedside adds another tool in our arsenal to provide real-time hemodynamic data that can be used to manage patients in the ICU. ACCE also allows repeated measurements to determine changes based on therapeutic interventions initiated.
In recognition of the importance of ACCE as a tool for intensivists, the National Board of Echocardiography (NBE) now offers a pathway toward board certification with the Examination of Special Competence in Critical Care Echocardiography (CCEeXAM). CHEST continues to offer cutting-edge courses in ACCE, as well as a board review course for learners interested in sitting for the CCEeXAM.
Amik Sodhi, MD, FCCP
Gul Zaidi, MD, FCCP
Members-at-Large
Ultrasound and Chest Imaging Section
Advanced critical care echocardiography: A noninvasive tool for hemodynamic assessment in critically ill patients
Hemodynamic assessments in critically ill patients are important to guide accurate management; however, traditional invasive methods of measuring cardiac output have significant limitations, including risks of infection and bleeding. ACCE can provide a multitude of hemodynamic measurements from cardiac output (CO) to right ventricular systolic pressure (RVSP) and left atrial pressure (LAP). Combinations of left ventricular function parameters, along with estimation of filling pressures, can help distinguish between types of shock. Schmidt and colleagues (Sci Rep. 2022;12[1]:7187) demonstrated that measurement of these indices in the majority of patients helped elucidate the cause for hemodynamic compromise. They found presence of a cardiac index (CI) < 2.5/min.m2 was associated with a doubling of ICU mortality as compared with predictions based on severity of illness scores in otherwise hemodynamically stable patients. Hollenberg and colleagues (Am J Cardiol. 2021;153:135-39) demonstrated the feasibility of a simpler stratification using the left ventricular ejection fraction (LVEF) and CI in coronavirus disease 2019 patients with shock, where low CI despite having a preserved LVEF was associated with worse outcomes.
Quick, reliable data are an intensivist’s friend. Utilizing ACCE at the bedside adds another tool in our arsenal to provide real-time hemodynamic data that can be used to manage patients in the ICU. ACCE also allows repeated measurements to determine changes based on therapeutic interventions initiated.
In recognition of the importance of ACCE as a tool for intensivists, the National Board of Echocardiography (NBE) now offers a pathway toward board certification with the Examination of Special Competence in Critical Care Echocardiography (CCEeXAM). CHEST continues to offer cutting-edge courses in ACCE, as well as a board review course for learners interested in sitting for the CCEeXAM.
Amik Sodhi, MD, FCCP
Gul Zaidi, MD, FCCP
Members-at-Large
Ultrasound and Chest Imaging Section
Advanced critical care echocardiography: A noninvasive tool for hemodynamic assessment in critically ill patients
Hemodynamic assessments in critically ill patients are important to guide accurate management; however, traditional invasive methods of measuring cardiac output have significant limitations, including risks of infection and bleeding. ACCE can provide a multitude of hemodynamic measurements from cardiac output (CO) to right ventricular systolic pressure (RVSP) and left atrial pressure (LAP). Combinations of left ventricular function parameters, along with estimation of filling pressures, can help distinguish between types of shock. Schmidt and colleagues (Sci Rep. 2022;12[1]:7187) demonstrated that measurement of these indices in the majority of patients helped elucidate the cause for hemodynamic compromise. They found presence of a cardiac index (CI) < 2.5/min.m2 was associated with a doubling of ICU mortality as compared with predictions based on severity of illness scores in otherwise hemodynamically stable patients. Hollenberg and colleagues (Am J Cardiol. 2021;153:135-39) demonstrated the feasibility of a simpler stratification using the left ventricular ejection fraction (LVEF) and CI in coronavirus disease 2019 patients with shock, where low CI despite having a preserved LVEF was associated with worse outcomes.
Quick, reliable data are an intensivist’s friend. Utilizing ACCE at the bedside adds another tool in our arsenal to provide real-time hemodynamic data that can be used to manage patients in the ICU. ACCE also allows repeated measurements to determine changes based on therapeutic interventions initiated.
In recognition of the importance of ACCE as a tool for intensivists, the National Board of Echocardiography (NBE) now offers a pathway toward board certification with the Examination of Special Competence in Critical Care Echocardiography (CCEeXAM). CHEST continues to offer cutting-edge courses in ACCE, as well as a board review course for learners interested in sitting for the CCEeXAM.
Amik Sodhi, MD, FCCP
Gul Zaidi, MD, FCCP
Members-at-Large
Thoracic Oncology & Chest Procedures Network
Interventional Procedures Section
Mind the gap: Improving adherence to lung cancer screening follow-up
The gap in adherence rates between a disciplined clinical trial and the heterogenous patchwork of U.S. health care is hardly unusual, but as lung cancer remains the number one cancer killer both worldwide and in the United States, one such disparity bears closer scrutiny.
In 2011, the National Lung Screening Trial (NLST) demonstrated a 20% reduction in lung cancer mortality with the implementation of low dose CT scan screening with 95% adherence to CT scan follow-up within 15 months of initial screening imaging (Aberle, et al. N Engl J Med. 2011;365[5]:395-409). Unfortunately, estimates of real-world adherence to lung cancer screening (LCS) follow-up fall to 51% even within an extended 18-month window (Hirsch, et al. Ann Am Thorac Soc. 2019;16[10]:1329-32).
Recent studies compared adherence to LCS follow-up between centralized and decentralized screening programs. Centralized programs used dedicated program coordinators and a tracking system, while decentralized programs relied on primary care providers.(Sakoda, et al. JAMA Network Open. 2021;4[4]:e218559). A subsequent study demonstrated adherence of 70% vs 41% among patients in centralized vs decentralized programs, respectively (Smith, et al. Chest. 2022;161[3]:818-25).
This gap is even more pronounced in majority-Black populations. Kunitomo and colleagues showed 33% lower odds of adherence to LCS follow-up compared with White patients (Kunitomo, et al. Chest. 2022;161[1]:266-75). Another study in a diverse, majority-Black patient population showed only 31% adherence to LCS follow-up at 1 year (Erkmen, et al. Cancer Causes Control. 2021;32[3]:291-8).
How could we close this gap? Centralized LCS programs show promise of increasing adherence to LCS follow-up. Heightened awareness of and targeted investment to mitigate racial inequities in LCS is imperative.
Jose De Cardenas MD
John Howe, MD
Members-at-Large
Interventional Procedures Section
Mind the gap: Improving adherence to lung cancer screening follow-up
The gap in adherence rates between a disciplined clinical trial and the heterogenous patchwork of U.S. health care is hardly unusual, but as lung cancer remains the number one cancer killer both worldwide and in the United States, one such disparity bears closer scrutiny.
In 2011, the National Lung Screening Trial (NLST) demonstrated a 20% reduction in lung cancer mortality with the implementation of low dose CT scan screening with 95% adherence to CT scan follow-up within 15 months of initial screening imaging (Aberle, et al. N Engl J Med. 2011;365[5]:395-409). Unfortunately, estimates of real-world adherence to lung cancer screening (LCS) follow-up fall to 51% even within an extended 18-month window (Hirsch, et al. Ann Am Thorac Soc. 2019;16[10]:1329-32).
Recent studies compared adherence to LCS follow-up between centralized and decentralized screening programs. Centralized programs used dedicated program coordinators and a tracking system, while decentralized programs relied on primary care providers.(Sakoda, et al. JAMA Network Open. 2021;4[4]:e218559). A subsequent study demonstrated adherence of 70% vs 41% among patients in centralized vs decentralized programs, respectively (Smith, et al. Chest. 2022;161[3]:818-25).
This gap is even more pronounced in majority-Black populations. Kunitomo and colleagues showed 33% lower odds of adherence to LCS follow-up compared with White patients (Kunitomo, et al. Chest. 2022;161[1]:266-75). Another study in a diverse, majority-Black patient population showed only 31% adherence to LCS follow-up at 1 year (Erkmen, et al. Cancer Causes Control. 2021;32[3]:291-8).
How could we close this gap? Centralized LCS programs show promise of increasing adherence to LCS follow-up. Heightened awareness of and targeted investment to mitigate racial inequities in LCS is imperative.
Jose De Cardenas MD
John Howe, MD
Members-at-Large
Interventional Procedures Section
Mind the gap: Improving adherence to lung cancer screening follow-up
The gap in adherence rates between a disciplined clinical trial and the heterogenous patchwork of U.S. health care is hardly unusual, but as lung cancer remains the number one cancer killer both worldwide and in the United States, one such disparity bears closer scrutiny.
In 2011, the National Lung Screening Trial (NLST) demonstrated a 20% reduction in lung cancer mortality with the implementation of low dose CT scan screening with 95% adherence to CT scan follow-up within 15 months of initial screening imaging (Aberle, et al. N Engl J Med. 2011;365[5]:395-409). Unfortunately, estimates of real-world adherence to lung cancer screening (LCS) follow-up fall to 51% even within an extended 18-month window (Hirsch, et al. Ann Am Thorac Soc. 2019;16[10]:1329-32).
Recent studies compared adherence to LCS follow-up between centralized and decentralized screening programs. Centralized programs used dedicated program coordinators and a tracking system, while decentralized programs relied on primary care providers.(Sakoda, et al. JAMA Network Open. 2021;4[4]:e218559). A subsequent study demonstrated adherence of 70% vs 41% among patients in centralized vs decentralized programs, respectively (Smith, et al. Chest. 2022;161[3]:818-25).
This gap is even more pronounced in majority-Black populations. Kunitomo and colleagues showed 33% lower odds of adherence to LCS follow-up compared with White patients (Kunitomo, et al. Chest. 2022;161[1]:266-75). Another study in a diverse, majority-Black patient population showed only 31% adherence to LCS follow-up at 1 year (Erkmen, et al. Cancer Causes Control. 2021;32[3]:291-8).
How could we close this gap? Centralized LCS programs show promise of increasing adherence to LCS follow-up. Heightened awareness of and targeted investment to mitigate racial inequities in LCS is imperative.
Jose De Cardenas MD
John Howe, MD
Members-at-Large
One in eight COVID patients likely to develop long COVID: Large study
a large study published in The Lancet indicates.
The researchers determined that percentage by comparing long-term symptoms in people infected by SARS-CoV-2 with similar symptoms in uninfected people over the same time period.
Among the group of infected study participants in the Netherlands, 21.4% had at least one new or severely increased symptom 3-5 months after infection compared with before infection. When that group of 21.4% was compared with 8.7% of uninfected people in the same study, the researchers were able to calculate a prevalence 12.7% with long COVID.
“This finding shows that post–COVID-19 condition is an urgent problem with a mounting human toll,” the study authors wrote.
The research design was novel, two editorialists said in an accompanying commentary.
Christopher Brightling, PhD, and Rachael Evans, MBChB, PhD, of the Institute for Lung Health, University of Leicester (England), noted: “This is a major advance on prior long COVID prevalence estimates as it includes a matched uninfected group and accounts for symptoms before COVID-19 infection.”
Symptoms that persist
The Lancet study found that 3-5 months after COVID (compared with before COVID) and compared with the non-COVID comparison group, the symptoms that persist were chest pain, breathing difficulties, pain when breathing, muscle pain, loss of taste and/or smell, tingling extremities, lump in throat, feeling hot and cold alternately, heavy limbs, and tiredness.
The authors noted that symptoms such as brain fog were found to be relevant to long COVID after the data collection period for this paper and were not included in this research.
Researcher Aranka V. Ballering, MSc, PhD candidate, said in an interview that the researchers found fever is a symptom that is clearly present during the acute phase of the disease and it peaks the day of the COVID-19 diagnosis, but also wears off.
Loss of taste and smell, however, rapidly increases in severity when COVID-19 is diagnosed, but also persists and is still present 3-5 months after COVID.
Ms. Ballering, with the department of psychiatry at the University of Groningen (the Netherlands), said she was surprised by the sex difference made evident in their research: “Women showed more severe persistent symptoms than men.”
Closer to a clearer definition
The authors said their findings also pinpoint symptoms that bring us closer to a better definition of long COVID, which has many different definitions globally.
“These symptoms have the highest discriminative ability to distinguish between post–COVID-19 condition and non–COVID-19–related symptoms,” they wrote.
Researchers collected data by asking participants in the northern Netherlands, who were part of the population-based Lifelines COVID-19 study, to regularly complete digital questionnaires on 23 symptoms commonly associated with long COVID. The questionnaire was sent out 24 times to the same people between March 2020 and August 2021. At that time, people had the Alpha or earlier variants.
Participants were considered COVID-19 positive if they had either a positive test or a doctor’s diagnosis of COVID-19.
Of 76,422 study participants, the 5.5% (4,231) who had COVID were matched to 8,462 controls. Researchers accounted for sex, age, and time of completing questionnaires.
Effect of hospitalization, vaccination unclear
Ms. Ballering said it’s unclear from this data whether vaccination or whether a person was hospitalized would change the prevalence of persistent symptoms.
Because of the period when the data were collected, “the vast majority of our study population was not fully vaccinated,” she said.
However, she pointed to recent research that shows that immunization against COVID is only partially effective against persistent somatic symptoms after COVID.
Also, only 5% of men and 2.5% of women in the study were hospitalized as a result of COVID-19, so the findings can’t easily be generalized to hospitalized patients.
The Lifelines study was an add-on study to the multidisciplinary, prospective, population-based, observational Dutch Lifelines cohort study examining 167,729 people in the Netherlands. Almost all were White, a limitation of the study, and 58% were female. Average age was 54.
The editorialists also noted additional limitations of the study were that this research “did not fully consider the impact on mental health” and was conducted in one region in the Netherlands.
Janko Nikolich-Žugich, MD, PhD, director of the Aegis Consortium for Pandemic-Free Future and head of the immunobiology department at University of Arizona, Tucson, said in an interview that he agreed with the editorialists that a primary benefit of this study is that it corrected for symptoms people had before COVID, something other studies have not been able to do.
However, he cautioned about generalizing the results for the United States and other countries because of the lack of diversity in the study population with regard to education level, socioeconomic factors, and race. He pointed out that access issues are also different in the Netherlands, which has universal health care.
He said brain fog as a symptom of long COVID is of high interest and will be important to include in future studies that are able to extend the study period.
The work was funded by ZonMw; the Dutch Ministry of Health, Welfare, and Sport; Dutch Ministry of Economic Affairs; University Medical Center Groningen, University of Groningen; and the provinces of Drenthe, Friesland, and Groningen. The study authors and Dr. Nikolich-Žugich have reported no relevant financial relationships. Dr. Brightling has received consultancy and or grants paid to his institution from GlaxoSmithKline, AstraZeneca, Boehringer Ingelheim, Novartis, Chiesi, Genentech, Roche, Sanofi, Regeneron, Mologic, and 4DPharma for asthma and chronic obstructive pulmonary disease research. Dr. Evans has received consultancy fees from AstraZeneca on the topic of long COVID and from GlaxoSmithKline on digital health, and speaker’s fees from Boehringer Ingelheim on long COVID.
A version of this article first appeared on Medscape.com.
a large study published in The Lancet indicates.
The researchers determined that percentage by comparing long-term symptoms in people infected by SARS-CoV-2 with similar symptoms in uninfected people over the same time period.
Among the group of infected study participants in the Netherlands, 21.4% had at least one new or severely increased symptom 3-5 months after infection compared with before infection. When that group of 21.4% was compared with 8.7% of uninfected people in the same study, the researchers were able to calculate a prevalence 12.7% with long COVID.
“This finding shows that post–COVID-19 condition is an urgent problem with a mounting human toll,” the study authors wrote.
The research design was novel, two editorialists said in an accompanying commentary.
Christopher Brightling, PhD, and Rachael Evans, MBChB, PhD, of the Institute for Lung Health, University of Leicester (England), noted: “This is a major advance on prior long COVID prevalence estimates as it includes a matched uninfected group and accounts for symptoms before COVID-19 infection.”
Symptoms that persist
The Lancet study found that 3-5 months after COVID (compared with before COVID) and compared with the non-COVID comparison group, the symptoms that persist were chest pain, breathing difficulties, pain when breathing, muscle pain, loss of taste and/or smell, tingling extremities, lump in throat, feeling hot and cold alternately, heavy limbs, and tiredness.
The authors noted that symptoms such as brain fog were found to be relevant to long COVID after the data collection period for this paper and were not included in this research.
Researcher Aranka V. Ballering, MSc, PhD candidate, said in an interview that the researchers found fever is a symptom that is clearly present during the acute phase of the disease and it peaks the day of the COVID-19 diagnosis, but also wears off.
Loss of taste and smell, however, rapidly increases in severity when COVID-19 is diagnosed, but also persists and is still present 3-5 months after COVID.
Ms. Ballering, with the department of psychiatry at the University of Groningen (the Netherlands), said she was surprised by the sex difference made evident in their research: “Women showed more severe persistent symptoms than men.”
Closer to a clearer definition
The authors said their findings also pinpoint symptoms that bring us closer to a better definition of long COVID, which has many different definitions globally.
“These symptoms have the highest discriminative ability to distinguish between post–COVID-19 condition and non–COVID-19–related symptoms,” they wrote.
Researchers collected data by asking participants in the northern Netherlands, who were part of the population-based Lifelines COVID-19 study, to regularly complete digital questionnaires on 23 symptoms commonly associated with long COVID. The questionnaire was sent out 24 times to the same people between March 2020 and August 2021. At that time, people had the Alpha or earlier variants.
Participants were considered COVID-19 positive if they had either a positive test or a doctor’s diagnosis of COVID-19.
Of 76,422 study participants, the 5.5% (4,231) who had COVID were matched to 8,462 controls. Researchers accounted for sex, age, and time of completing questionnaires.
Effect of hospitalization, vaccination unclear
Ms. Ballering said it’s unclear from this data whether vaccination or whether a person was hospitalized would change the prevalence of persistent symptoms.
Because of the period when the data were collected, “the vast majority of our study population was not fully vaccinated,” she said.
However, she pointed to recent research that shows that immunization against COVID is only partially effective against persistent somatic symptoms after COVID.
Also, only 5% of men and 2.5% of women in the study were hospitalized as a result of COVID-19, so the findings can’t easily be generalized to hospitalized patients.
The Lifelines study was an add-on study to the multidisciplinary, prospective, population-based, observational Dutch Lifelines cohort study examining 167,729 people in the Netherlands. Almost all were White, a limitation of the study, and 58% were female. Average age was 54.
The editorialists also noted additional limitations of the study were that this research “did not fully consider the impact on mental health” and was conducted in one region in the Netherlands.
Janko Nikolich-Žugich, MD, PhD, director of the Aegis Consortium for Pandemic-Free Future and head of the immunobiology department at University of Arizona, Tucson, said in an interview that he agreed with the editorialists that a primary benefit of this study is that it corrected for symptoms people had before COVID, something other studies have not been able to do.
However, he cautioned about generalizing the results for the United States and other countries because of the lack of diversity in the study population with regard to education level, socioeconomic factors, and race. He pointed out that access issues are also different in the Netherlands, which has universal health care.
He said brain fog as a symptom of long COVID is of high interest and will be important to include in future studies that are able to extend the study period.
The work was funded by ZonMw; the Dutch Ministry of Health, Welfare, and Sport; Dutch Ministry of Economic Affairs; University Medical Center Groningen, University of Groningen; and the provinces of Drenthe, Friesland, and Groningen. The study authors and Dr. Nikolich-Žugich have reported no relevant financial relationships. Dr. Brightling has received consultancy and or grants paid to his institution from GlaxoSmithKline, AstraZeneca, Boehringer Ingelheim, Novartis, Chiesi, Genentech, Roche, Sanofi, Regeneron, Mologic, and 4DPharma for asthma and chronic obstructive pulmonary disease research. Dr. Evans has received consultancy fees from AstraZeneca on the topic of long COVID and from GlaxoSmithKline on digital health, and speaker’s fees from Boehringer Ingelheim on long COVID.
A version of this article first appeared on Medscape.com.
a large study published in The Lancet indicates.
The researchers determined that percentage by comparing long-term symptoms in people infected by SARS-CoV-2 with similar symptoms in uninfected people over the same time period.
Among the group of infected study participants in the Netherlands, 21.4% had at least one new or severely increased symptom 3-5 months after infection compared with before infection. When that group of 21.4% was compared with 8.7% of uninfected people in the same study, the researchers were able to calculate a prevalence 12.7% with long COVID.
“This finding shows that post–COVID-19 condition is an urgent problem with a mounting human toll,” the study authors wrote.
The research design was novel, two editorialists said in an accompanying commentary.
Christopher Brightling, PhD, and Rachael Evans, MBChB, PhD, of the Institute for Lung Health, University of Leicester (England), noted: “This is a major advance on prior long COVID prevalence estimates as it includes a matched uninfected group and accounts for symptoms before COVID-19 infection.”
Symptoms that persist
The Lancet study found that 3-5 months after COVID (compared with before COVID) and compared with the non-COVID comparison group, the symptoms that persist were chest pain, breathing difficulties, pain when breathing, muscle pain, loss of taste and/or smell, tingling extremities, lump in throat, feeling hot and cold alternately, heavy limbs, and tiredness.
The authors noted that symptoms such as brain fog were found to be relevant to long COVID after the data collection period for this paper and were not included in this research.
Researcher Aranka V. Ballering, MSc, PhD candidate, said in an interview that the researchers found fever is a symptom that is clearly present during the acute phase of the disease and it peaks the day of the COVID-19 diagnosis, but also wears off.
Loss of taste and smell, however, rapidly increases in severity when COVID-19 is diagnosed, but also persists and is still present 3-5 months after COVID.
Ms. Ballering, with the department of psychiatry at the University of Groningen (the Netherlands), said she was surprised by the sex difference made evident in their research: “Women showed more severe persistent symptoms than men.”
Closer to a clearer definition
The authors said their findings also pinpoint symptoms that bring us closer to a better definition of long COVID, which has many different definitions globally.
“These symptoms have the highest discriminative ability to distinguish between post–COVID-19 condition and non–COVID-19–related symptoms,” they wrote.
Researchers collected data by asking participants in the northern Netherlands, who were part of the population-based Lifelines COVID-19 study, to regularly complete digital questionnaires on 23 symptoms commonly associated with long COVID. The questionnaire was sent out 24 times to the same people between March 2020 and August 2021. At that time, people had the Alpha or earlier variants.
Participants were considered COVID-19 positive if they had either a positive test or a doctor’s diagnosis of COVID-19.
Of 76,422 study participants, the 5.5% (4,231) who had COVID were matched to 8,462 controls. Researchers accounted for sex, age, and time of completing questionnaires.
Effect of hospitalization, vaccination unclear
Ms. Ballering said it’s unclear from this data whether vaccination or whether a person was hospitalized would change the prevalence of persistent symptoms.
Because of the period when the data were collected, “the vast majority of our study population was not fully vaccinated,” she said.
However, she pointed to recent research that shows that immunization against COVID is only partially effective against persistent somatic symptoms after COVID.
Also, only 5% of men and 2.5% of women in the study were hospitalized as a result of COVID-19, so the findings can’t easily be generalized to hospitalized patients.
The Lifelines study was an add-on study to the multidisciplinary, prospective, population-based, observational Dutch Lifelines cohort study examining 167,729 people in the Netherlands. Almost all were White, a limitation of the study, and 58% were female. Average age was 54.
The editorialists also noted additional limitations of the study were that this research “did not fully consider the impact on mental health” and was conducted in one region in the Netherlands.
Janko Nikolich-Žugich, MD, PhD, director of the Aegis Consortium for Pandemic-Free Future and head of the immunobiology department at University of Arizona, Tucson, said in an interview that he agreed with the editorialists that a primary benefit of this study is that it corrected for symptoms people had before COVID, something other studies have not been able to do.
However, he cautioned about generalizing the results for the United States and other countries because of the lack of diversity in the study population with regard to education level, socioeconomic factors, and race. He pointed out that access issues are also different in the Netherlands, which has universal health care.
He said brain fog as a symptom of long COVID is of high interest and will be important to include in future studies that are able to extend the study period.
The work was funded by ZonMw; the Dutch Ministry of Health, Welfare, and Sport; Dutch Ministry of Economic Affairs; University Medical Center Groningen, University of Groningen; and the provinces of Drenthe, Friesland, and Groningen. The study authors and Dr. Nikolich-Žugich have reported no relevant financial relationships. Dr. Brightling has received consultancy and or grants paid to his institution from GlaxoSmithKline, AstraZeneca, Boehringer Ingelheim, Novartis, Chiesi, Genentech, Roche, Sanofi, Regeneron, Mologic, and 4DPharma for asthma and chronic obstructive pulmonary disease research. Dr. Evans has received consultancy fees from AstraZeneca on the topic of long COVID and from GlaxoSmithKline on digital health, and speaker’s fees from Boehringer Ingelheim on long COVID.
A version of this article first appeared on Medscape.com.
FROM THE LANCET
Sleep Medicine Network
Home-based Mechanical Ventilation and Neuromuscular Disease Section
Navigating the latest device supply chain challenge: Mechanical airway clearance
(distal airways). Cough augmentation techniques provide lung volume recruitment on the insufflation phase, in addition to mobilization of secretions with augmentation of the peak expiratory flow rate to >160 L/min on the exhalation phase.
A mechanical insufflation-exsufflation (MI-E) device (T70 Cough Assist - Phillips) is now on indefinite backorder. This creates a dangerous situation for our patients requiring cough augmentation for survival. Alternative options that provide both MI-E and high frequency oscillation include two systems (Synclara Cough System – Hill-rom and the Biwaze Cough System-ABM Respiratory Care).
The Synclara can only be obtained in a direct-to-patient model, contracting with individual respiratory therapists, outside of the standard durable medical equipment model. The final MI-E model option is the VOCSYN multifunctional ventilator (ventilator, cough assist, nebulizer, oxygen concentrator, suction). This multifunction ventilator has had variable acceptance with HCPCS code E0467. If the VOCSYN is chosen, the patient cannot have been issued any component devices or have reached the 36-month cap for oxygen equipment (CR 10854 special payment rule, 42 CFR414.222).
As the supply of devices is exhausted, we will need to shift to evidence-based manual options. Manual cough augmentation can be done effectively with a bag-valve mask, using breath stacking to achieve maximal lung insufflation, optimizing the length tension relationship of elastic recoil on exhalation to increase peak cough flow (PCF).
This can be done alone but is more effective when combined with manually assisted cough (Bach JR. Chest. 1993;104[5]:1553-62). These interventions require training of the caregivers, using resources such as those found at www.canventottawa.ca.
With continued supply chain instability, manual airway clearance techniques should be considered in patients with less advanced cough impairment (PCF 160-270 L/min), to save the remaining devices for those with PCF of <160 L/min.
Jeanette Brown, MD, PhD
Karin Provost, DO, PhD
Members-at-Large
Home-based Mechanical Ventilation and Neuromuscular Disease Section
Navigating the latest device supply chain challenge: Mechanical airway clearance
(distal airways). Cough augmentation techniques provide lung volume recruitment on the insufflation phase, in addition to mobilization of secretions with augmentation of the peak expiratory flow rate to >160 L/min on the exhalation phase.
A mechanical insufflation-exsufflation (MI-E) device (T70 Cough Assist - Phillips) is now on indefinite backorder. This creates a dangerous situation for our patients requiring cough augmentation for survival. Alternative options that provide both MI-E and high frequency oscillation include two systems (Synclara Cough System – Hill-rom and the Biwaze Cough System-ABM Respiratory Care).
The Synclara can only be obtained in a direct-to-patient model, contracting with individual respiratory therapists, outside of the standard durable medical equipment model. The final MI-E model option is the VOCSYN multifunctional ventilator (ventilator, cough assist, nebulizer, oxygen concentrator, suction). This multifunction ventilator has had variable acceptance with HCPCS code E0467. If the VOCSYN is chosen, the patient cannot have been issued any component devices or have reached the 36-month cap for oxygen equipment (CR 10854 special payment rule, 42 CFR414.222).
As the supply of devices is exhausted, we will need to shift to evidence-based manual options. Manual cough augmentation can be done effectively with a bag-valve mask, using breath stacking to achieve maximal lung insufflation, optimizing the length tension relationship of elastic recoil on exhalation to increase peak cough flow (PCF).
This can be done alone but is more effective when combined with manually assisted cough (Bach JR. Chest. 1993;104[5]:1553-62). These interventions require training of the caregivers, using resources such as those found at www.canventottawa.ca.
With continued supply chain instability, manual airway clearance techniques should be considered in patients with less advanced cough impairment (PCF 160-270 L/min), to save the remaining devices for those with PCF of <160 L/min.
Jeanette Brown, MD, PhD
Karin Provost, DO, PhD
Members-at-Large
Home-based Mechanical Ventilation and Neuromuscular Disease Section
Navigating the latest device supply chain challenge: Mechanical airway clearance
(distal airways). Cough augmentation techniques provide lung volume recruitment on the insufflation phase, in addition to mobilization of secretions with augmentation of the peak expiratory flow rate to >160 L/min on the exhalation phase.
A mechanical insufflation-exsufflation (MI-E) device (T70 Cough Assist - Phillips) is now on indefinite backorder. This creates a dangerous situation for our patients requiring cough augmentation for survival. Alternative options that provide both MI-E and high frequency oscillation include two systems (Synclara Cough System – Hill-rom and the Biwaze Cough System-ABM Respiratory Care).
The Synclara can only be obtained in a direct-to-patient model, contracting with individual respiratory therapists, outside of the standard durable medical equipment model. The final MI-E model option is the VOCSYN multifunctional ventilator (ventilator, cough assist, nebulizer, oxygen concentrator, suction). This multifunction ventilator has had variable acceptance with HCPCS code E0467. If the VOCSYN is chosen, the patient cannot have been issued any component devices or have reached the 36-month cap for oxygen equipment (CR 10854 special payment rule, 42 CFR414.222).
As the supply of devices is exhausted, we will need to shift to evidence-based manual options. Manual cough augmentation can be done effectively with a bag-valve mask, using breath stacking to achieve maximal lung insufflation, optimizing the length tension relationship of elastic recoil on exhalation to increase peak cough flow (PCF).
This can be done alone but is more effective when combined with manually assisted cough (Bach JR. Chest. 1993;104[5]:1553-62). These interventions require training of the caregivers, using resources such as those found at www.canventottawa.ca.
With continued supply chain instability, manual airway clearance techniques should be considered in patients with less advanced cough impairment (PCF 160-270 L/min), to save the remaining devices for those with PCF of <160 L/min.
Jeanette Brown, MD, PhD
Karin Provost, DO, PhD
Members-at-Large
Airways Disorders Network
Asthma and COPD Section
Go TEAM! Shared decision-making tool for patient-clinician collaboration in severe asthma
Shared decision-making is associated with improved medication adherence in adults (Wilson, et al. Am J Respir Crit Care Med. 2010;181[6]:566-77) and quality of life and asthma control in children (Taylor, et al. J Asthma. 2018;55[6]:675-83). The Global Initiative for Asthma committee recommends a patient-clinician partnership. Activated and engaged patients play a major role in their asthma management (https://ginasthma.org/gina-reports). Shared decision-making discussions should include potential benefits and harms of the therapeutic options, patient’s values and lifestyle preferences, and addressing concerns.
The CHEST Foundation, the Allergy and Asthma Network, and the American College of Allergy, Asthma, and Immunology developed an online shared decision- making tool for severe asthma (https://asthma.chestnet.org/sdm-tool).
This tool utilizes patient’s values, specifics about triggers, asthma control, medication side effects, and lifestyle preferences to identify personalized management options. The tool provides information about recommended therapeutic options in simple terms, including potential benefits, possible side effects, expected treatment frequency and duration, and financial aid information. The treatment options currently explained in this tool include anti-immunoglobulin E, anti-interleukin-5, anti-interleukin-4/13, bronchial thermoplasty, long-acting muscarinic antagonist, macrolides, oral corticosteroids, and standard of care.
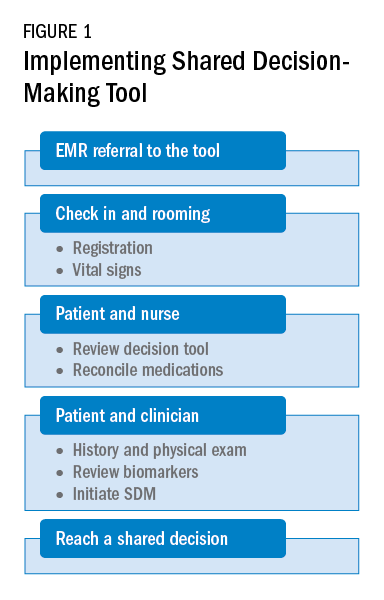
As a team, the patient and the health care professional can use this tool during office visits to help guide management. Figure 1 shows a suggested workflow to utilize the tool in clinical practice.
Potential barriers include excess time and increased human resources. Barrier mitigation may include reviewing the tool and reconciling the medications before the clinician enters the room. With these interventions, many clinician encounters may be completed in 10 to 15 minutes.
Farrukh Abbas, MBBS
Fellow-in-Training
Asthma and COPD Section
Go TEAM! Shared decision-making tool for patient-clinician collaboration in severe asthma
Shared decision-making is associated with improved medication adherence in adults (Wilson, et al. Am J Respir Crit Care Med. 2010;181[6]:566-77) and quality of life and asthma control in children (Taylor, et al. J Asthma. 2018;55[6]:675-83). The Global Initiative for Asthma committee recommends a patient-clinician partnership. Activated and engaged patients play a major role in their asthma management (https://ginasthma.org/gina-reports). Shared decision-making discussions should include potential benefits and harms of the therapeutic options, patient’s values and lifestyle preferences, and addressing concerns.
The CHEST Foundation, the Allergy and Asthma Network, and the American College of Allergy, Asthma, and Immunology developed an online shared decision- making tool for severe asthma (https://asthma.chestnet.org/sdm-tool).
This tool utilizes patient’s values, specifics about triggers, asthma control, medication side effects, and lifestyle preferences to identify personalized management options. The tool provides information about recommended therapeutic options in simple terms, including potential benefits, possible side effects, expected treatment frequency and duration, and financial aid information. The treatment options currently explained in this tool include anti-immunoglobulin E, anti-interleukin-5, anti-interleukin-4/13, bronchial thermoplasty, long-acting muscarinic antagonist, macrolides, oral corticosteroids, and standard of care.
As a team, the patient and the health care professional can use this tool during office visits to help guide management. Figure 1 shows a suggested workflow to utilize the tool in clinical practice.
Potential barriers include excess time and increased human resources. Barrier mitigation may include reviewing the tool and reconciling the medications before the clinician enters the room. With these interventions, many clinician encounters may be completed in 10 to 15 minutes.

Farrukh Abbas, MBBS
Fellow-in-Training
Asthma and COPD Section
Go TEAM! Shared decision-making tool for patient-clinician collaboration in severe asthma
Shared decision-making is associated with improved medication adherence in adults (Wilson, et al. Am J Respir Crit Care Med. 2010;181[6]:566-77) and quality of life and asthma control in children (Taylor, et al. J Asthma. 2018;55[6]:675-83). The Global Initiative for Asthma committee recommends a patient-clinician partnership. Activated and engaged patients play a major role in their asthma management (https://ginasthma.org/gina-reports). Shared decision-making discussions should include potential benefits and harms of the therapeutic options, patient’s values and lifestyle preferences, and addressing concerns.
The CHEST Foundation, the Allergy and Asthma Network, and the American College of Allergy, Asthma, and Immunology developed an online shared decision- making tool for severe asthma (https://asthma.chestnet.org/sdm-tool).
This tool utilizes patient’s values, specifics about triggers, asthma control, medication side effects, and lifestyle preferences to identify personalized management options. The tool provides information about recommended therapeutic options in simple terms, including potential benefits, possible side effects, expected treatment frequency and duration, and financial aid information. The treatment options currently explained in this tool include anti-immunoglobulin E, anti-interleukin-5, anti-interleukin-4/13, bronchial thermoplasty, long-acting muscarinic antagonist, macrolides, oral corticosteroids, and standard of care.
As a team, the patient and the health care professional can use this tool during office visits to help guide management. Figure 1 shows a suggested workflow to utilize the tool in clinical practice.
Potential barriers include excess time and increased human resources. Barrier mitigation may include reviewing the tool and reconciling the medications before the clinician enters the room. With these interventions, many clinician encounters may be completed in 10 to 15 minutes.

Farrukh Abbas, MBBS
Fellow-in-Training
Long COVID doubles risk of some serious outcomes in children, teens
Researchers from the Centers for Disease Control and Prevention report that
Heart inflammation; a blood clot in the lung; or a blood clot in the lower leg, thigh, or pelvis were the most common bad outcomes in a new study. Even though the risk was higher for these and some other serious events, the overall numbers were small.
“Many of these conditions were rare or uncommon among children in this analysis, but even a small increase in these conditions is notable,” a CDC new release stated.
The investigators said their findings stress the importance of COVID-19 vaccination in Americans under the age of 18.
The study was published online in the CDC’s Morbidity and Mortality Weekly Report.
Less is known about long COVID in children
Lyudmyla Kompaniyets, PhD, and colleagues noted that most research on long COVID to date has been done in adults, so little information is available about the risks to Americans ages 17 and younger.
To learn more, they compared post–COVID-19 symptoms and conditions between 781,419 children and teenagers with confirmed COVID-19 to another 2,344,257 without COVID-19. They looked at medical claims and laboratory data for these children and teenagers from March 1, 2020, through Jan. 31, 2022, to see who got any of 15 specific outcomes linked to long COVID-19.
Long COVID was defined as a condition where symptoms that last for or begin at least 4 weeks after a COVID-19 diagnosis.
Compared to children with no history of a COVID-19 diagnosis, the long COVID-19 group was 101% more likely to have an acute pulmonary embolism, 99% more likely to have myocarditis or cardiomyopathy, 87% more likely to have a venous thromboembolic event, 32% more likely to have acute and unspecified renal failure, and 23% more likely to have type 1 diabetes.
“This report points to the fact that the risks of COVID infection itself, both in terms of the acute effects, MIS-C [multisystem inflammatory syndrome in children], as well as the long-term effects, are real, are concerning, and are potentially very serious,” said Stuart Berger, MD, chair of the American Academy of Pediatrics Section on Cardiology and Cardiac Surgery.
“The message that we should take away from this is that we should be very keen on all the methods of prevention for COVID, especially the vaccine,” said Dr. Berger, chief of cardiology in the department of pediatrics at Northwestern University in Chicago.
A ‘wake-up call’
The study findings are “sobering” and are “a reminder of the seriousness of COVID infection,” says Gregory Poland, MD, an infectious disease expert at the Mayo Clinic in Rochester, Minn.
“When you look in particular at the more serious complications from COVID in this young age group, those are life-altering complications that will have consequences and ramifications throughout their lives,” he said.
“I would take this as a serious wake-up call to parents [at a time when] the immunization rates in younger children are so pitifully low,” Dr. Poland said.
Still early days
The study is suggestive but not definitive, said Peter Katona, MD, professor of medicine and infectious diseases expert at the UCLA Fielding School of Public Health.
It’s still too early to draw conclusions about long COVID, including in children, because many questions remain, he said: Should long COVID be defined as symptoms at 1 month or 3 months after infection? How do you define brain fog?
Dr. Katona and colleagues are studying long COVID intervention among students at UCLA to answer some of these questions, including the incidence and effect of early intervention.
The study had “at least seven limitations,” the researchers noted. Among them was the use of medical claims data that noted long COVID outcomes but not how severe they were; some people in the no COVID group might have had the illness but not been diagnosed; and the researchers did not adjust for vaccination status.
Dr. Poland noted that the study was done during surges in COVID variants including Delta and Omicron. In other words, any long COVID effects linked to more recent variants such as BA.5 or BA.2.75 are unknown.
A version of this article first appeared on WebMD.com.
Researchers from the Centers for Disease Control and Prevention report that
Heart inflammation; a blood clot in the lung; or a blood clot in the lower leg, thigh, or pelvis were the most common bad outcomes in a new study. Even though the risk was higher for these and some other serious events, the overall numbers were small.
“Many of these conditions were rare or uncommon among children in this analysis, but even a small increase in these conditions is notable,” a CDC new release stated.
The investigators said their findings stress the importance of COVID-19 vaccination in Americans under the age of 18.
The study was published online in the CDC’s Morbidity and Mortality Weekly Report.
Less is known about long COVID in children
Lyudmyla Kompaniyets, PhD, and colleagues noted that most research on long COVID to date has been done in adults, so little information is available about the risks to Americans ages 17 and younger.
To learn more, they compared post–COVID-19 symptoms and conditions between 781,419 children and teenagers with confirmed COVID-19 to another 2,344,257 without COVID-19. They looked at medical claims and laboratory data for these children and teenagers from March 1, 2020, through Jan. 31, 2022, to see who got any of 15 specific outcomes linked to long COVID-19.
Long COVID was defined as a condition where symptoms that last for or begin at least 4 weeks after a COVID-19 diagnosis.
Compared to children with no history of a COVID-19 diagnosis, the long COVID-19 group was 101% more likely to have an acute pulmonary embolism, 99% more likely to have myocarditis or cardiomyopathy, 87% more likely to have a venous thromboembolic event, 32% more likely to have acute and unspecified renal failure, and 23% more likely to have type 1 diabetes.
“This report points to the fact that the risks of COVID infection itself, both in terms of the acute effects, MIS-C [multisystem inflammatory syndrome in children], as well as the long-term effects, are real, are concerning, and are potentially very serious,” said Stuart Berger, MD, chair of the American Academy of Pediatrics Section on Cardiology and Cardiac Surgery.
“The message that we should take away from this is that we should be very keen on all the methods of prevention for COVID, especially the vaccine,” said Dr. Berger, chief of cardiology in the department of pediatrics at Northwestern University in Chicago.
A ‘wake-up call’
The study findings are “sobering” and are “a reminder of the seriousness of COVID infection,” says Gregory Poland, MD, an infectious disease expert at the Mayo Clinic in Rochester, Minn.
“When you look in particular at the more serious complications from COVID in this young age group, those are life-altering complications that will have consequences and ramifications throughout their lives,” he said.
“I would take this as a serious wake-up call to parents [at a time when] the immunization rates in younger children are so pitifully low,” Dr. Poland said.
Still early days
The study is suggestive but not definitive, said Peter Katona, MD, professor of medicine and infectious diseases expert at the UCLA Fielding School of Public Health.
It’s still too early to draw conclusions about long COVID, including in children, because many questions remain, he said: Should long COVID be defined as symptoms at 1 month or 3 months after infection? How do you define brain fog?
Dr. Katona and colleagues are studying long COVID intervention among students at UCLA to answer some of these questions, including the incidence and effect of early intervention.
The study had “at least seven limitations,” the researchers noted. Among them was the use of medical claims data that noted long COVID outcomes but not how severe they were; some people in the no COVID group might have had the illness but not been diagnosed; and the researchers did not adjust for vaccination status.
Dr. Poland noted that the study was done during surges in COVID variants including Delta and Omicron. In other words, any long COVID effects linked to more recent variants such as BA.5 or BA.2.75 are unknown.
A version of this article first appeared on WebMD.com.
Researchers from the Centers for Disease Control and Prevention report that
Heart inflammation; a blood clot in the lung; or a blood clot in the lower leg, thigh, or pelvis were the most common bad outcomes in a new study. Even though the risk was higher for these and some other serious events, the overall numbers were small.
“Many of these conditions were rare or uncommon among children in this analysis, but even a small increase in these conditions is notable,” a CDC new release stated.
The investigators said their findings stress the importance of COVID-19 vaccination in Americans under the age of 18.
The study was published online in the CDC’s Morbidity and Mortality Weekly Report.
Less is known about long COVID in children
Lyudmyla Kompaniyets, PhD, and colleagues noted that most research on long COVID to date has been done in adults, so little information is available about the risks to Americans ages 17 and younger.
To learn more, they compared post–COVID-19 symptoms and conditions between 781,419 children and teenagers with confirmed COVID-19 to another 2,344,257 without COVID-19. They looked at medical claims and laboratory data for these children and teenagers from March 1, 2020, through Jan. 31, 2022, to see who got any of 15 specific outcomes linked to long COVID-19.
Long COVID was defined as a condition where symptoms that last for or begin at least 4 weeks after a COVID-19 diagnosis.
Compared to children with no history of a COVID-19 diagnosis, the long COVID-19 group was 101% more likely to have an acute pulmonary embolism, 99% more likely to have myocarditis or cardiomyopathy, 87% more likely to have a venous thromboembolic event, 32% more likely to have acute and unspecified renal failure, and 23% more likely to have type 1 diabetes.
“This report points to the fact that the risks of COVID infection itself, both in terms of the acute effects, MIS-C [multisystem inflammatory syndrome in children], as well as the long-term effects, are real, are concerning, and are potentially very serious,” said Stuart Berger, MD, chair of the American Academy of Pediatrics Section on Cardiology and Cardiac Surgery.
“The message that we should take away from this is that we should be very keen on all the methods of prevention for COVID, especially the vaccine,” said Dr. Berger, chief of cardiology in the department of pediatrics at Northwestern University in Chicago.
A ‘wake-up call’
The study findings are “sobering” and are “a reminder of the seriousness of COVID infection,” says Gregory Poland, MD, an infectious disease expert at the Mayo Clinic in Rochester, Minn.
“When you look in particular at the more serious complications from COVID in this young age group, those are life-altering complications that will have consequences and ramifications throughout their lives,” he said.
“I would take this as a serious wake-up call to parents [at a time when] the immunization rates in younger children are so pitifully low,” Dr. Poland said.
Still early days
The study is suggestive but not definitive, said Peter Katona, MD, professor of medicine and infectious diseases expert at the UCLA Fielding School of Public Health.
It’s still too early to draw conclusions about long COVID, including in children, because many questions remain, he said: Should long COVID be defined as symptoms at 1 month or 3 months after infection? How do you define brain fog?
Dr. Katona and colleagues are studying long COVID intervention among students at UCLA to answer some of these questions, including the incidence and effect of early intervention.
The study had “at least seven limitations,” the researchers noted. Among them was the use of medical claims data that noted long COVID outcomes but not how severe they were; some people in the no COVID group might have had the illness but not been diagnosed; and the researchers did not adjust for vaccination status.
Dr. Poland noted that the study was done during surges in COVID variants including Delta and Omicron. In other words, any long COVID effects linked to more recent variants such as BA.5 or BA.2.75 are unknown.
A version of this article first appeared on WebMD.com.
FROM THE MMWR
Addressing racial bias in pulse oximetry
Pulse oximetry is a vital monitoring tool in the ICU and in pulmonary medicine. Regrettably, re-emerging data show that pulse oximeters do not accurately measure blood oxygen levels in Black patients, presumably due to their skin tone. (i.e., low arterial oxygen saturation despite a seemingly normal pulse oximetry reading). While inaccuracy of pulse oximeter measurements in patients with darker skin has been recognized for decades, recent studies have highlighted this as an ongoing problem with potentially severe consequences for Black patients and other patients of color.
One recent study found that Black patients had almost three times the likelihood of occult hypoxemia compared with White patients (Sjoding, MW, et al. N Engl J Med. 2020;383[25]:2477-8).
Subsequent studies have confirmed this to be a widespread problem across various clinical settings in hundreds of hospitals (Wong AI, et al. JAMA Netw Open. 2021;4[11]:e2131674; Valbuena VS, et al. Chest. 2022;161[4]:971-8). A recent retrospective cohort study of patients with COVID-19 found that occult hypoxemia in Black and Hispanic patients was associated with delayed eligibility for potentially lifesaving COVID-19 therapies (Fawzy AF, et al. JAMA Intern Med. 2022; published online May 31, 2022).
Now that numerous studies have demonstrated the inaccuracy of pulse oximetry with the potential to cause harm to historically marginalized racial and ethnic groups, must we abandon the use of pulse oximetry? We would argue that pulse oximeters remain valuable tools, but for now, we must adapt our practice until better devices are widely adopted.
First, it is crucial that health professionals are aware that pulse oximeters may underestimate the true extent of hypoxemia for all patients, but particularly for patients with darker skin. Acknowledging this device flaw is essential to avoid harm to our patients.
Second, clinicians must have heightened skepticism for seemingly normal pulse oximetry values when caring for symptomatic patients at risk of occult hypoxemia.
Until better pulse oximeters are widely available, clinicians must consider workarounds aimed at ensuring timely identification of hypoxemia in Black patients and other patients of color.
These patients may need invasive monitoring of arterial oxygenation, including arterial blood gas checks or an arterial catheter. However, invasive monitoring comes at the cost of discomfort to patients and potential complications, such as vessel or nerve damage.
Invasive monitoring of patients at risk for occult hypoxemia is not an equitable or acceptable long-term solution for this problem. As advocates for patients, clinicians and professional organizations should lobby regulatory bodies to ensure pulse oximeters are accurate for all patients.
We must also call on government leaders to move this process forward. For example, in response to efforts by the United Kingdom’s Intensive Care Society, the Health Secretary of the UK, Sajid Javid, has called for a review of pulse oximeters as part of a larger review assessing structural issues in health care that lead to worse outcomes in racial and ethnic minorities (BBC News. https://www.bbc.com/news/uk-59363544. Published online Nov. 21, 2021).
Device companies are largely for-profit corporations with obligations to their shareholders. It seems that existing incentives are insufficient to motivate investment in less biased technology and real-world evaluations of their devices.
We previously called for buyers of pulse oximeters to change the incentives of device companies – that is, for “hospitals to commit to only purchasing pulse oximeters that have been shown to work equally well in patients of colour.” (Hidalgo DC, et al. Lancet Respir Med. 2021;9[4]:E37). And, indeed, we worry that hospitals are putting themselves at medicolegal risk by not raising their purchasing standards. Since it is now widely known that pulse oximeters are inaccurate in certain patients, could there be liability for hospitals that continue to use devices we know to be disproportionately inaccurate by race?
Device companies must commit to fixing racial bias in pulse oximeters. Change is feasible, and pulse oximeters can be redesigned to be accurate and reliable among all patients using existing technology that is decades-old.
In the 1960s and 1970s, Hewlett Packard worked with NASA to noninvasively measure oxygen saturation in astronauts (Moran-Thomas, M. Wired. Published online June 4, 2021. https://www.wired.com/story/pulse-oximeters-equity). The device was designed to work for all skin tones and could be calibrated based on an individual’s skin tone. However, Hewlett Packard moved away from medical devices in the 1980s, shelving their design while other companies took over the oximeter market.
Lastly, as new devices are designed, they must be proven to work for all patients. Testing should be conducted in real-world clinical settings using metrics aligned with clinical care, since we know testing in artificial environments may not generalize to critically ill patients. Testing standards historically used by the FDA, such as only requiring device testing in a small number of non-White individuals, may miss clinically relevant hypoxemia. Non-inferiority studies are particularly susceptible to poor design or under-powering, and rigorous standards are needed from unbiased sources.
While potential solutions are currently being evaluated, the fact remains that the inaccuracy of pulse oximeters has been known for decades without any meaningful action taken to correct the problem.
As Valeria Valbuena, author of a study demonstrating inaccuracy of pulse oximetry in patients about to undergo ECMO, points out, “Using White patients as the standard in biomedical design has led to both differential care and innovation inertia for optimizing the way devices and algorithms work for patients of racial and ethnic minoritized groups” (Valbuena VS. JAMA Intern Med. 2022. doi: 10.1001/jamainternmed.2022.1903).
We know that hypoxemia is dangerous for our patients and that this is only one example of the long-standing systemic racism leading to harm in historically marginalized racial and ethnic groups. It is unacceptable that the devices we rely on to care for our patients are disproportionately inaccurate in non-White patients.
We hope that with increased awareness of this problem, meaningful action will be taken by device companies to ensure pulse oximeters work accurately for all patients.
From the Division of Pulmonary and Critical Care, Department of Medicine and the Center for Bioethics and Social Sciences in Medicine, University of Michigan Medical School (Drs. Harlan and Valley), and the Institute for Healthcare Policy and Innovation (Dr. Valley), University of Michigan, Ann Arbor, MI; and the Division of Pulmonary Sciences and Critical Care Medicine, University of Colorado School of Medicine, Aurora, CO (Dr. Colon Hidalgo).
Pulse oximetry is a vital monitoring tool in the ICU and in pulmonary medicine. Regrettably, re-emerging data show that pulse oximeters do not accurately measure blood oxygen levels in Black patients, presumably due to their skin tone. (i.e., low arterial oxygen saturation despite a seemingly normal pulse oximetry reading). While inaccuracy of pulse oximeter measurements in patients with darker skin has been recognized for decades, recent studies have highlighted this as an ongoing problem with potentially severe consequences for Black patients and other patients of color.
One recent study found that Black patients had almost three times the likelihood of occult hypoxemia compared with White patients (Sjoding, MW, et al. N Engl J Med. 2020;383[25]:2477-8).
Subsequent studies have confirmed this to be a widespread problem across various clinical settings in hundreds of hospitals (Wong AI, et al. JAMA Netw Open. 2021;4[11]:e2131674; Valbuena VS, et al. Chest. 2022;161[4]:971-8). A recent retrospective cohort study of patients with COVID-19 found that occult hypoxemia in Black and Hispanic patients was associated with delayed eligibility for potentially lifesaving COVID-19 therapies (Fawzy AF, et al. JAMA Intern Med. 2022; published online May 31, 2022).
Now that numerous studies have demonstrated the inaccuracy of pulse oximetry with the potential to cause harm to historically marginalized racial and ethnic groups, must we abandon the use of pulse oximetry? We would argue that pulse oximeters remain valuable tools, but for now, we must adapt our practice until better devices are widely adopted.
First, it is crucial that health professionals are aware that pulse oximeters may underestimate the true extent of hypoxemia for all patients, but particularly for patients with darker skin. Acknowledging this device flaw is essential to avoid harm to our patients.
Second, clinicians must have heightened skepticism for seemingly normal pulse oximetry values when caring for symptomatic patients at risk of occult hypoxemia.
Until better pulse oximeters are widely available, clinicians must consider workarounds aimed at ensuring timely identification of hypoxemia in Black patients and other patients of color.
These patients may need invasive monitoring of arterial oxygenation, including arterial blood gas checks or an arterial catheter. However, invasive monitoring comes at the cost of discomfort to patients and potential complications, such as vessel or nerve damage.
Invasive monitoring of patients at risk for occult hypoxemia is not an equitable or acceptable long-term solution for this problem. As advocates for patients, clinicians and professional organizations should lobby regulatory bodies to ensure pulse oximeters are accurate for all patients.
We must also call on government leaders to move this process forward. For example, in response to efforts by the United Kingdom’s Intensive Care Society, the Health Secretary of the UK, Sajid Javid, has called for a review of pulse oximeters as part of a larger review assessing structural issues in health care that lead to worse outcomes in racial and ethnic minorities (BBC News. https://www.bbc.com/news/uk-59363544. Published online Nov. 21, 2021).
Device companies are largely for-profit corporations with obligations to their shareholders. It seems that existing incentives are insufficient to motivate investment in less biased technology and real-world evaluations of their devices.
We previously called for buyers of pulse oximeters to change the incentives of device companies – that is, for “hospitals to commit to only purchasing pulse oximeters that have been shown to work equally well in patients of colour.” (Hidalgo DC, et al. Lancet Respir Med. 2021;9[4]:E37). And, indeed, we worry that hospitals are putting themselves at medicolegal risk by not raising their purchasing standards. Since it is now widely known that pulse oximeters are inaccurate in certain patients, could there be liability for hospitals that continue to use devices we know to be disproportionately inaccurate by race?
Device companies must commit to fixing racial bias in pulse oximeters. Change is feasible, and pulse oximeters can be redesigned to be accurate and reliable among all patients using existing technology that is decades-old.
In the 1960s and 1970s, Hewlett Packard worked with NASA to noninvasively measure oxygen saturation in astronauts (Moran-Thomas, M. Wired. Published online June 4, 2021. https://www.wired.com/story/pulse-oximeters-equity). The device was designed to work for all skin tones and could be calibrated based on an individual’s skin tone. However, Hewlett Packard moved away from medical devices in the 1980s, shelving their design while other companies took over the oximeter market.
Lastly, as new devices are designed, they must be proven to work for all patients. Testing should be conducted in real-world clinical settings using metrics aligned with clinical care, since we know testing in artificial environments may not generalize to critically ill patients. Testing standards historically used by the FDA, such as only requiring device testing in a small number of non-White individuals, may miss clinically relevant hypoxemia. Non-inferiority studies are particularly susceptible to poor design or under-powering, and rigorous standards are needed from unbiased sources.
While potential solutions are currently being evaluated, the fact remains that the inaccuracy of pulse oximeters has been known for decades without any meaningful action taken to correct the problem.
As Valeria Valbuena, author of a study demonstrating inaccuracy of pulse oximetry in patients about to undergo ECMO, points out, “Using White patients as the standard in biomedical design has led to both differential care and innovation inertia for optimizing the way devices and algorithms work for patients of racial and ethnic minoritized groups” (Valbuena VS. JAMA Intern Med. 2022. doi: 10.1001/jamainternmed.2022.1903).
We know that hypoxemia is dangerous for our patients and that this is only one example of the long-standing systemic racism leading to harm in historically marginalized racial and ethnic groups. It is unacceptable that the devices we rely on to care for our patients are disproportionately inaccurate in non-White patients.
We hope that with increased awareness of this problem, meaningful action will be taken by device companies to ensure pulse oximeters work accurately for all patients.
From the Division of Pulmonary and Critical Care, Department of Medicine and the Center for Bioethics and Social Sciences in Medicine, University of Michigan Medical School (Drs. Harlan and Valley), and the Institute for Healthcare Policy and Innovation (Dr. Valley), University of Michigan, Ann Arbor, MI; and the Division of Pulmonary Sciences and Critical Care Medicine, University of Colorado School of Medicine, Aurora, CO (Dr. Colon Hidalgo).
Pulse oximetry is a vital monitoring tool in the ICU and in pulmonary medicine. Regrettably, re-emerging data show that pulse oximeters do not accurately measure blood oxygen levels in Black patients, presumably due to their skin tone. (i.e., low arterial oxygen saturation despite a seemingly normal pulse oximetry reading). While inaccuracy of pulse oximeter measurements in patients with darker skin has been recognized for decades, recent studies have highlighted this as an ongoing problem with potentially severe consequences for Black patients and other patients of color.
One recent study found that Black patients had almost three times the likelihood of occult hypoxemia compared with White patients (Sjoding, MW, et al. N Engl J Med. 2020;383[25]:2477-8).
Subsequent studies have confirmed this to be a widespread problem across various clinical settings in hundreds of hospitals (Wong AI, et al. JAMA Netw Open. 2021;4[11]:e2131674; Valbuena VS, et al. Chest. 2022;161[4]:971-8). A recent retrospective cohort study of patients with COVID-19 found that occult hypoxemia in Black and Hispanic patients was associated with delayed eligibility for potentially lifesaving COVID-19 therapies (Fawzy AF, et al. JAMA Intern Med. 2022; published online May 31, 2022).
Now that numerous studies have demonstrated the inaccuracy of pulse oximetry with the potential to cause harm to historically marginalized racial and ethnic groups, must we abandon the use of pulse oximetry? We would argue that pulse oximeters remain valuable tools, but for now, we must adapt our practice until better devices are widely adopted.
First, it is crucial that health professionals are aware that pulse oximeters may underestimate the true extent of hypoxemia for all patients, but particularly for patients with darker skin. Acknowledging this device flaw is essential to avoid harm to our patients.
Second, clinicians must have heightened skepticism for seemingly normal pulse oximetry values when caring for symptomatic patients at risk of occult hypoxemia.
Until better pulse oximeters are widely available, clinicians must consider workarounds aimed at ensuring timely identification of hypoxemia in Black patients and other patients of color.
These patients may need invasive monitoring of arterial oxygenation, including arterial blood gas checks or an arterial catheter. However, invasive monitoring comes at the cost of discomfort to patients and potential complications, such as vessel or nerve damage.
Invasive monitoring of patients at risk for occult hypoxemia is not an equitable or acceptable long-term solution for this problem. As advocates for patients, clinicians and professional organizations should lobby regulatory bodies to ensure pulse oximeters are accurate for all patients.
We must also call on government leaders to move this process forward. For example, in response to efforts by the United Kingdom’s Intensive Care Society, the Health Secretary of the UK, Sajid Javid, has called for a review of pulse oximeters as part of a larger review assessing structural issues in health care that lead to worse outcomes in racial and ethnic minorities (BBC News. https://www.bbc.com/news/uk-59363544. Published online Nov. 21, 2021).
Device companies are largely for-profit corporations with obligations to their shareholders. It seems that existing incentives are insufficient to motivate investment in less biased technology and real-world evaluations of their devices.
We previously called for buyers of pulse oximeters to change the incentives of device companies – that is, for “hospitals to commit to only purchasing pulse oximeters that have been shown to work equally well in patients of colour.” (Hidalgo DC, et al. Lancet Respir Med. 2021;9[4]:E37). And, indeed, we worry that hospitals are putting themselves at medicolegal risk by not raising their purchasing standards. Since it is now widely known that pulse oximeters are inaccurate in certain patients, could there be liability for hospitals that continue to use devices we know to be disproportionately inaccurate by race?
Device companies must commit to fixing racial bias in pulse oximeters. Change is feasible, and pulse oximeters can be redesigned to be accurate and reliable among all patients using existing technology that is decades-old.
In the 1960s and 1970s, Hewlett Packard worked with NASA to noninvasively measure oxygen saturation in astronauts (Moran-Thomas, M. Wired. Published online June 4, 2021. https://www.wired.com/story/pulse-oximeters-equity). The device was designed to work for all skin tones and could be calibrated based on an individual’s skin tone. However, Hewlett Packard moved away from medical devices in the 1980s, shelving their design while other companies took over the oximeter market.
Lastly, as new devices are designed, they must be proven to work for all patients. Testing should be conducted in real-world clinical settings using metrics aligned with clinical care, since we know testing in artificial environments may not generalize to critically ill patients. Testing standards historically used by the FDA, such as only requiring device testing in a small number of non-White individuals, may miss clinically relevant hypoxemia. Non-inferiority studies are particularly susceptible to poor design or under-powering, and rigorous standards are needed from unbiased sources.
While potential solutions are currently being evaluated, the fact remains that the inaccuracy of pulse oximeters has been known for decades without any meaningful action taken to correct the problem.
As Valeria Valbuena, author of a study demonstrating inaccuracy of pulse oximetry in patients about to undergo ECMO, points out, “Using White patients as the standard in biomedical design has led to both differential care and innovation inertia for optimizing the way devices and algorithms work for patients of racial and ethnic minoritized groups” (Valbuena VS. JAMA Intern Med. 2022. doi: 10.1001/jamainternmed.2022.1903).
We know that hypoxemia is dangerous for our patients and that this is only one example of the long-standing systemic racism leading to harm in historically marginalized racial and ethnic groups. It is unacceptable that the devices we rely on to care for our patients are disproportionately inaccurate in non-White patients.
We hope that with increased awareness of this problem, meaningful action will be taken by device companies to ensure pulse oximeters work accurately for all patients.
From the Division of Pulmonary and Critical Care, Department of Medicine and the Center for Bioethics and Social Sciences in Medicine, University of Michigan Medical School (Drs. Harlan and Valley), and the Institute for Healthcare Policy and Innovation (Dr. Valley), University of Michigan, Ann Arbor, MI; and the Division of Pulmonary Sciences and Critical Care Medicine, University of Colorado School of Medicine, Aurora, CO (Dr. Colon Hidalgo).
Registry launched to reach more patients with PF
The Pulmonary Fibrosis Foundation (PFF) has launched a new initiative in which they hope to capture a far more diverse representation of patients with pulmonary fibrosis (PF) than the current registry allows them to do, a press release from the PFF indicated.
“The existing registry we have – the PFF Patient Registry – is limited to our care centers, which are primarily academic clinical institutions and we have only a few thousand patients within that registry,” Junelle Speller, MBA, vice president of the PFF Registry, told this news organization.
“We wanted to go beyond these care centers and capture patients in community centers, and in rural settings to provide a more complete understanding of patients with this disease and, of course, have a larger sample size,” she added.
“ across all parts of the United States and, most importantly, accelerate the research on PF toward improving earlier diagnosis, treatment, and outcomes for these patients,” Ms. Speller said.
Passive versus active
The PFF Community Registry differs in its structure and purpose from the PFF Patient Registry, as Ms. Speller explained. First, the PFF Patient Registry, established in 2016, is “passive” in its nature in that whatever information is entered into a patient’s electronic medical record or clinical chart on a routine office visit is abstracted and captured in the registry. By contrast, the PFF Community Registry is asking for self-reported data from patients, “so it’s more of an ‘active’ registry and will give us a chance to have a bidirectional connection with participants, provide email updates and newsletters, and give patients an opportunity to participate in future studies within the registry as well as in clinical trials,” she noted.
The two registries still overlap in that both capture demographic data on patients’ medical and family histories as well as any medications patients may be taking, but the Community Registry will also capture information with respect to education, employment, patient-reported outcomes, and quality of life metrics. “It will also let us know how patients feel about continued education on the disease itself and patient participation in support groups,” Ms. Speller observed.
The Community Registry will also collect information from lung transplant recipients who have had PF or any other form of interstitial lung disease (ILD) as well as information from caregivers and family members affected by the patient’s disease. As Ms. Speller noted, both PF and other forms of ILD (of which there are more than 200 types) are all characterized by inflammation or scarring in the lung. “Patients are often misdiagnosed, and it can take months, even years, to identify the disease,” Ms. Speller said.
From there, it can be a very long and difficult road ahead, with no cure in sight, although several antifibrotic drugs do help slow disease progression. Typically, onset is around the age of 60 and symptoms include chronic dry cough, fatigue, shortness of breath, weakness, discomfort in the chest, and sometimes unexplained weight loss. Some patients do have a history of smoking, but not all, Ms. Speller noted. So far, registry data suggest PF largely occurs in White patients.
“We’re very excited about the Community Registry, particularly about reaching into communities that we haven’t been able to reach with our existing registry,” Ms. Speller noted. “The rural population in particular is often underserved, so we are really looking forward to capturing data from these patients as well as those from community centers within smaller and larger cities,” she observed.
“A powerful aspect of the Community Registry is that we can use the information gained from it to understand the experience of individuals living with PF, and how it impacts their lives and those of their families and caregivers,” Kevin Flaherty, MD, steering committee chair, PFF Registry, said in a statement.
“Researchers can also look at the data to better understand fibrotic lung diseases and learn about effective approaches to improve patient care,” he added.
Patients who wish to join the PFF Community Registry can sign up at pffregistry.org. To learn more about PF and its risk factors, readers are invited to visit www.AboutPF.org. More than 250,000 patients in the United States are living with either PF or other types of ILD.
Ms. Speller and Dr. Flaherty disclosed no financial conflicts of interest. The PFF Registry is supported by its founding partner, Genentech, Visionary Partner, United Therapeutics, and its sustaining partner, Boehringer Ingelheim, as well as many donors.
A version of this article first appeared on Medscape.com.
This article was updated 8/8/22.
The Pulmonary Fibrosis Foundation (PFF) has launched a new initiative in which they hope to capture a far more diverse representation of patients with pulmonary fibrosis (PF) than the current registry allows them to do, a press release from the PFF indicated.
“The existing registry we have – the PFF Patient Registry – is limited to our care centers, which are primarily academic clinical institutions and we have only a few thousand patients within that registry,” Junelle Speller, MBA, vice president of the PFF Registry, told this news organization.
“We wanted to go beyond these care centers and capture patients in community centers, and in rural settings to provide a more complete understanding of patients with this disease and, of course, have a larger sample size,” she added.
“ across all parts of the United States and, most importantly, accelerate the research on PF toward improving earlier diagnosis, treatment, and outcomes for these patients,” Ms. Speller said.
Passive versus active
The PFF Community Registry differs in its structure and purpose from the PFF Patient Registry, as Ms. Speller explained. First, the PFF Patient Registry, established in 2016, is “passive” in its nature in that whatever information is entered into a patient’s electronic medical record or clinical chart on a routine office visit is abstracted and captured in the registry. By contrast, the PFF Community Registry is asking for self-reported data from patients, “so it’s more of an ‘active’ registry and will give us a chance to have a bidirectional connection with participants, provide email updates and newsletters, and give patients an opportunity to participate in future studies within the registry as well as in clinical trials,” she noted.
The two registries still overlap in that both capture demographic data on patients’ medical and family histories as well as any medications patients may be taking, but the Community Registry will also capture information with respect to education, employment, patient-reported outcomes, and quality of life metrics. “It will also let us know how patients feel about continued education on the disease itself and patient participation in support groups,” Ms. Speller observed.
The Community Registry will also collect information from lung transplant recipients who have had PF or any other form of interstitial lung disease (ILD) as well as information from caregivers and family members affected by the patient’s disease. As Ms. Speller noted, both PF and other forms of ILD (of which there are more than 200 types) are all characterized by inflammation or scarring in the lung. “Patients are often misdiagnosed, and it can take months, even years, to identify the disease,” Ms. Speller said.
From there, it can be a very long and difficult road ahead, with no cure in sight, although several antifibrotic drugs do help slow disease progression. Typically, onset is around the age of 60 and symptoms include chronic dry cough, fatigue, shortness of breath, weakness, discomfort in the chest, and sometimes unexplained weight loss. Some patients do have a history of smoking, but not all, Ms. Speller noted. So far, registry data suggest PF largely occurs in White patients.
“We’re very excited about the Community Registry, particularly about reaching into communities that we haven’t been able to reach with our existing registry,” Ms. Speller noted. “The rural population in particular is often underserved, so we are really looking forward to capturing data from these patients as well as those from community centers within smaller and larger cities,” she observed.
“A powerful aspect of the Community Registry is that we can use the information gained from it to understand the experience of individuals living with PF, and how it impacts their lives and those of their families and caregivers,” Kevin Flaherty, MD, steering committee chair, PFF Registry, said in a statement.
“Researchers can also look at the data to better understand fibrotic lung diseases and learn about effective approaches to improve patient care,” he added.
Patients who wish to join the PFF Community Registry can sign up at pffregistry.org. To learn more about PF and its risk factors, readers are invited to visit www.AboutPF.org. More than 250,000 patients in the United States are living with either PF or other types of ILD.
Ms. Speller and Dr. Flaherty disclosed no financial conflicts of interest. The PFF Registry is supported by its founding partner, Genentech, Visionary Partner, United Therapeutics, and its sustaining partner, Boehringer Ingelheim, as well as many donors.
A version of this article first appeared on Medscape.com.
This article was updated 8/8/22.
The Pulmonary Fibrosis Foundation (PFF) has launched a new initiative in which they hope to capture a far more diverse representation of patients with pulmonary fibrosis (PF) than the current registry allows them to do, a press release from the PFF indicated.
“The existing registry we have – the PFF Patient Registry – is limited to our care centers, which are primarily academic clinical institutions and we have only a few thousand patients within that registry,” Junelle Speller, MBA, vice president of the PFF Registry, told this news organization.
“We wanted to go beyond these care centers and capture patients in community centers, and in rural settings to provide a more complete understanding of patients with this disease and, of course, have a larger sample size,” she added.
“ across all parts of the United States and, most importantly, accelerate the research on PF toward improving earlier diagnosis, treatment, and outcomes for these patients,” Ms. Speller said.
Passive versus active
The PFF Community Registry differs in its structure and purpose from the PFF Patient Registry, as Ms. Speller explained. First, the PFF Patient Registry, established in 2016, is “passive” in its nature in that whatever information is entered into a patient’s electronic medical record or clinical chart on a routine office visit is abstracted and captured in the registry. By contrast, the PFF Community Registry is asking for self-reported data from patients, “so it’s more of an ‘active’ registry and will give us a chance to have a bidirectional connection with participants, provide email updates and newsletters, and give patients an opportunity to participate in future studies within the registry as well as in clinical trials,” she noted.
The two registries still overlap in that both capture demographic data on patients’ medical and family histories as well as any medications patients may be taking, but the Community Registry will also capture information with respect to education, employment, patient-reported outcomes, and quality of life metrics. “It will also let us know how patients feel about continued education on the disease itself and patient participation in support groups,” Ms. Speller observed.
The Community Registry will also collect information from lung transplant recipients who have had PF or any other form of interstitial lung disease (ILD) as well as information from caregivers and family members affected by the patient’s disease. As Ms. Speller noted, both PF and other forms of ILD (of which there are more than 200 types) are all characterized by inflammation or scarring in the lung. “Patients are often misdiagnosed, and it can take months, even years, to identify the disease,” Ms. Speller said.
From there, it can be a very long and difficult road ahead, with no cure in sight, although several antifibrotic drugs do help slow disease progression. Typically, onset is around the age of 60 and symptoms include chronic dry cough, fatigue, shortness of breath, weakness, discomfort in the chest, and sometimes unexplained weight loss. Some patients do have a history of smoking, but not all, Ms. Speller noted. So far, registry data suggest PF largely occurs in White patients.
“We’re very excited about the Community Registry, particularly about reaching into communities that we haven’t been able to reach with our existing registry,” Ms. Speller noted. “The rural population in particular is often underserved, so we are really looking forward to capturing data from these patients as well as those from community centers within smaller and larger cities,” she observed.
“A powerful aspect of the Community Registry is that we can use the information gained from it to understand the experience of individuals living with PF, and how it impacts their lives and those of their families and caregivers,” Kevin Flaherty, MD, steering committee chair, PFF Registry, said in a statement.
“Researchers can also look at the data to better understand fibrotic lung diseases and learn about effective approaches to improve patient care,” he added.
Patients who wish to join the PFF Community Registry can sign up at pffregistry.org. To learn more about PF and its risk factors, readers are invited to visit www.AboutPF.org. More than 250,000 patients in the United States are living with either PF or other types of ILD.
Ms. Speller and Dr. Flaherty disclosed no financial conflicts of interest. The PFF Registry is supported by its founding partner, Genentech, Visionary Partner, United Therapeutics, and its sustaining partner, Boehringer Ingelheim, as well as many donors.
A version of this article first appeared on Medscape.com.
This article was updated 8/8/22.
Robotic bronchoscopy 2022
Over the last several years, hundreds of millions of dollars have been spent on robotic bronchoscopy systems in the United States. The release of robotic scopes was made to great fanfare, translating into the market being infiltrated with these systems. With base costs in the hundreds of thousands of dollars, robotic bronchoscope systems are easily the most expensive singular capital investment in the bronchoscopy suite. I frequently get asked questions from those who have not yet made that purchase: “Should I buy a robot?” “How could I justify a new robot purchase to my hospital?” “Is the hype real?” These are complex questions to answer. Before one can answer, I think it’s best to look back on the last 2 decades of bronchoscopy for peripheral lung nodules to get a better understanding of the value proposition robotic bronchoscopes may offer.
Guided bronchoscopy for lung nodules has significantly evolved over the past 2 decades, shifting diagnostic procedures from interventional radiologist to the pulmonologist. Some of these advances were based in redesigns of the bronchoscope (ultrathin bronchoscopy) or application of technology to the bronchoscope (radial EBUS, virtual bronchoscopy); but, these were not broadly applicable to the pulmonology community at large. It was not until the development of electromagnetic navigational bronchoscopy (ENB) that widespread adoption of bronchoscopy for lung nodules occurred. By and large, ENB fueled a rapid expansion of nodule bronchoscopy, mainly due to its ease of use and novel approach. Initial studies of ENB had impressive results; however, studies were criticized for having small numbers, inadequate follow-up, spurious definitions of yield, and that they were being done at highly specialized centers. The NAVIGATE trial was launched to address these criticisms among “real world” conditions. Sponsored by Medtronic, it studied ENB (superDimension platform, v6.0 or higher) across 29 academic and medical centers in the United States, enrolling over 1,000 patients (Folch EE, et al. J Thorac Oncol. 2019;14[3]:445-58. Epub 2018 Nov 23), and reported a diagnostic yield of 73%.
This led to a drive to improve upon yield, resulting in development of new technologies specifically designed to address some of the factors thought associated with diminished yield, and, out of this, robotic bronchoscopy was born. These factors included CT scan-body registration divergence, deflection of the extended working channel (EWC) by rigid biopsy tools, and inability to accurately “aim” the EWC-biopsy tool at the nodule; these were especially problematic in nodules not associated with airways. Robotic scopes were specifically designed to reach into the peripheral lung airways similar to an EWC, but with better structural integrity and steerability. This tip integrity would resist tool-related displacement, and steerability would allow for improved targeting of nodules during the biopsy.
There are two robots approved by the FDA at the time of this writing (Auris Monarch, Intuitive Ion), with a third awaiting FDA clearance (Noah Galaxy). In general, though the engineering of the robotic scopes to improve structural tip integrity are similar, the approach to navigation and targeting vary significantly. The Monarch platform uses electromagnetic guidance, similar to other traditional ENB platforms. The Ion platform does not use ENB; instead, it uses fiberoptic shape sensing technology, which analyzes the shape and orientation of the scope to provide location information. There are potential advantages to shape sensing, the most notable being the absence of electromagnetics; this allows for use of fluoroscopy during the procedure, which otherwise would have interfered with ENB-based navigation. There are other subtle differences between the two robots. The Monarch uses a scope-in-scope design, with a robotic scope contained within a robotic sheath; the Ion uses a single robotic scope. The Ion scope diameter is 3.5 mm, whereas the Monarch diameter is 4.4 mm; this may be a potential advantage when having to navigate through smaller airways.
So, which robot is better suited to reach peripheral nodules more consistently and accurately? I get asked this question a lot, since I have both platforms at my institution. But, answering with my own opinion based on my institution’s anecdotal experience would be irresponsible. I’m more of a “what does the data show?” person. Luckily, we do have clinical trials in both robot technologies. It should be noted here that there will likely never be a head-to-head randomized trial, so evaluating published studies with each platform is going to be the best method we have for comparison going forward, albeit an imperfect one. It should also be noted that many of the early robotic bronchoscopy trials have to be looked at with caution, as yield definitions tended not to be conservative and/or the follow-up of non-malignant was not robust. With that in mind, let’s review representative high-quality studies for each platform.
The best study to date using the Ion platform came out of Memorial Sloan Kettering Cancer Center (Kalchiem-Dekel O, et al. Chest. 2022;161[2];572-82). This single-site study reported on 159 nodule biopsies, with the primary outcome being diagnostic yield. The patients had 1 year of follow-up, and the definition of yield was conservative. The average lesion size was 18 mm, and nodule locations and characteristics were representative of real-world conditions. Overall diagnostic yield was 81.7%; however, it dropped to under 70% for nodules under 20 mm in size.
The largest study to date using the Monarch platform was also a single center study, this from the University of Chicago (Agrawal, et al. Ann Thorac Surg. 2022 Jan 17;S0003-4975(22)00042-X. Online ahead of print). This study included 124 nodules with at least 12 months of follow-up; diagnostic yield definition was conservative. Median nodule size was 20.5 mm, with distribution and characteristics representative of real-world conditions. Overall accuracy was 77%, and, similar to the Ion study, dropped to under 70% when nodule size was smaller than 20 mm.
Overall, both robot studies seemed to show a modest improvement in diagnostic yield when compared with ENB, and their outcomes were overall similar. It is important to remember that these were studies of each center’s first experiences with early versions of each technology; over time, the technology will continue to improve, as will operator skill and experience, and with that, perhaps improvements in yield will be seen, as well.
Interestingly, both studies evaluated target localization using radial EBUS (rEBUS), which also allowed for airway-nodule relationships to be reported. In Kalchiem-Dekel’s study, 85% of cases used rEBUS to determine localization, and, of these, 91.2% of cases showed accurate localization. In Agrawal’s study, rEBUS was used in all cases with a reported localization of 94%. In both, yield did not seem to be affected by airway-nodule relationships, perhaps explained by more robust tip control of the robotic scope. However, localization did not equate to yield in all cases, which brings up a very important question: Can the yield of robotic bronchoscopy be further improved with better real-time on-board imaging, such as CBCT scanning or C-arm based tomography? Currently, there is a study using 3D technology (Cios 3D Mobile Spin) in conjunction with the Ion platform to evaluate this.
So, let’s circle back to where we started. I think if you look at the totality of the data, it is clear that the robotic platforms currently offer a modest improvement in diagnostic yield over traditional ENB, with individual performances that are somewhat equivalent despite differences in design and operation. But does this improvement in yield justify the cost? Individual hospitals will have to make that decision. The capital cost and per-use price of the scope is significant, which has to be balanced against each center’s current performance with non-robotic bronchoscopy.
To date, there have been over 25,000 robotic procedures performed in the United States, so enthusiasm across diverse centers is being maintained. Whether this enthusiasm is driven by yield or novelty, or both, I’m not sure. With other nonrobotic platforms having reached, or soon to reach, the market, this is a good time to be in the business of bronchoscopy.
Dr. Cicenia is in the Section of Bronchoscopy at Cleveland Clinic’s Respiratory Institute, Cleveland, Ohio.
Over the last several years, hundreds of millions of dollars have been spent on robotic bronchoscopy systems in the United States. The release of robotic scopes was made to great fanfare, translating into the market being infiltrated with these systems. With base costs in the hundreds of thousands of dollars, robotic bronchoscope systems are easily the most expensive singular capital investment in the bronchoscopy suite. I frequently get asked questions from those who have not yet made that purchase: “Should I buy a robot?” “How could I justify a new robot purchase to my hospital?” “Is the hype real?” These are complex questions to answer. Before one can answer, I think it’s best to look back on the last 2 decades of bronchoscopy for peripheral lung nodules to get a better understanding of the value proposition robotic bronchoscopes may offer.
Guided bronchoscopy for lung nodules has significantly evolved over the past 2 decades, shifting diagnostic procedures from interventional radiologist to the pulmonologist. Some of these advances were based in redesigns of the bronchoscope (ultrathin bronchoscopy) or application of technology to the bronchoscope (radial EBUS, virtual bronchoscopy); but, these were not broadly applicable to the pulmonology community at large. It was not until the development of electromagnetic navigational bronchoscopy (ENB) that widespread adoption of bronchoscopy for lung nodules occurred. By and large, ENB fueled a rapid expansion of nodule bronchoscopy, mainly due to its ease of use and novel approach. Initial studies of ENB had impressive results; however, studies were criticized for having small numbers, inadequate follow-up, spurious definitions of yield, and that they were being done at highly specialized centers. The NAVIGATE trial was launched to address these criticisms among “real world” conditions. Sponsored by Medtronic, it studied ENB (superDimension platform, v6.0 or higher) across 29 academic and medical centers in the United States, enrolling over 1,000 patients (Folch EE, et al. J Thorac Oncol. 2019;14[3]:445-58. Epub 2018 Nov 23), and reported a diagnostic yield of 73%.
This led to a drive to improve upon yield, resulting in development of new technologies specifically designed to address some of the factors thought associated with diminished yield, and, out of this, robotic bronchoscopy was born. These factors included CT scan-body registration divergence, deflection of the extended working channel (EWC) by rigid biopsy tools, and inability to accurately “aim” the EWC-biopsy tool at the nodule; these were especially problematic in nodules not associated with airways. Robotic scopes were specifically designed to reach into the peripheral lung airways similar to an EWC, but with better structural integrity and steerability. This tip integrity would resist tool-related displacement, and steerability would allow for improved targeting of nodules during the biopsy.
There are two robots approved by the FDA at the time of this writing (Auris Monarch, Intuitive Ion), with a third awaiting FDA clearance (Noah Galaxy). In general, though the engineering of the robotic scopes to improve structural tip integrity are similar, the approach to navigation and targeting vary significantly. The Monarch platform uses electromagnetic guidance, similar to other traditional ENB platforms. The Ion platform does not use ENB; instead, it uses fiberoptic shape sensing technology, which analyzes the shape and orientation of the scope to provide location information. There are potential advantages to shape sensing, the most notable being the absence of electromagnetics; this allows for use of fluoroscopy during the procedure, which otherwise would have interfered with ENB-based navigation. There are other subtle differences between the two robots. The Monarch uses a scope-in-scope design, with a robotic scope contained within a robotic sheath; the Ion uses a single robotic scope. The Ion scope diameter is 3.5 mm, whereas the Monarch diameter is 4.4 mm; this may be a potential advantage when having to navigate through smaller airways.
So, which robot is better suited to reach peripheral nodules more consistently and accurately? I get asked this question a lot, since I have both platforms at my institution. But, answering with my own opinion based on my institution’s anecdotal experience would be irresponsible. I’m more of a “what does the data show?” person. Luckily, we do have clinical trials in both robot technologies. It should be noted here that there will likely never be a head-to-head randomized trial, so evaluating published studies with each platform is going to be the best method we have for comparison going forward, albeit an imperfect one. It should also be noted that many of the early robotic bronchoscopy trials have to be looked at with caution, as yield definitions tended not to be conservative and/or the follow-up of non-malignant was not robust. With that in mind, let’s review representative high-quality studies for each platform.
The best study to date using the Ion platform came out of Memorial Sloan Kettering Cancer Center (Kalchiem-Dekel O, et al. Chest. 2022;161[2];572-82). This single-site study reported on 159 nodule biopsies, with the primary outcome being diagnostic yield. The patients had 1 year of follow-up, and the definition of yield was conservative. The average lesion size was 18 mm, and nodule locations and characteristics were representative of real-world conditions. Overall diagnostic yield was 81.7%; however, it dropped to under 70% for nodules under 20 mm in size.
The largest study to date using the Monarch platform was also a single center study, this from the University of Chicago (Agrawal, et al. Ann Thorac Surg. 2022 Jan 17;S0003-4975(22)00042-X. Online ahead of print). This study included 124 nodules with at least 12 months of follow-up; diagnostic yield definition was conservative. Median nodule size was 20.5 mm, with distribution and characteristics representative of real-world conditions. Overall accuracy was 77%, and, similar to the Ion study, dropped to under 70% when nodule size was smaller than 20 mm.
Overall, both robot studies seemed to show a modest improvement in diagnostic yield when compared with ENB, and their outcomes were overall similar. It is important to remember that these were studies of each center’s first experiences with early versions of each technology; over time, the technology will continue to improve, as will operator skill and experience, and with that, perhaps improvements in yield will be seen, as well.
Interestingly, both studies evaluated target localization using radial EBUS (rEBUS), which also allowed for airway-nodule relationships to be reported. In Kalchiem-Dekel’s study, 85% of cases used rEBUS to determine localization, and, of these, 91.2% of cases showed accurate localization. In Agrawal’s study, rEBUS was used in all cases with a reported localization of 94%. In both, yield did not seem to be affected by airway-nodule relationships, perhaps explained by more robust tip control of the robotic scope. However, localization did not equate to yield in all cases, which brings up a very important question: Can the yield of robotic bronchoscopy be further improved with better real-time on-board imaging, such as CBCT scanning or C-arm based tomography? Currently, there is a study using 3D technology (Cios 3D Mobile Spin) in conjunction with the Ion platform to evaluate this.
So, let’s circle back to where we started. I think if you look at the totality of the data, it is clear that the robotic platforms currently offer a modest improvement in diagnostic yield over traditional ENB, with individual performances that are somewhat equivalent despite differences in design and operation. But does this improvement in yield justify the cost? Individual hospitals will have to make that decision. The capital cost and per-use price of the scope is significant, which has to be balanced against each center’s current performance with non-robotic bronchoscopy.
To date, there have been over 25,000 robotic procedures performed in the United States, so enthusiasm across diverse centers is being maintained. Whether this enthusiasm is driven by yield or novelty, or both, I’m not sure. With other nonrobotic platforms having reached, or soon to reach, the market, this is a good time to be in the business of bronchoscopy.
Dr. Cicenia is in the Section of Bronchoscopy at Cleveland Clinic’s Respiratory Institute, Cleveland, Ohio.
Over the last several years, hundreds of millions of dollars have been spent on robotic bronchoscopy systems in the United States. The release of robotic scopes was made to great fanfare, translating into the market being infiltrated with these systems. With base costs in the hundreds of thousands of dollars, robotic bronchoscope systems are easily the most expensive singular capital investment in the bronchoscopy suite. I frequently get asked questions from those who have not yet made that purchase: “Should I buy a robot?” “How could I justify a new robot purchase to my hospital?” “Is the hype real?” These are complex questions to answer. Before one can answer, I think it’s best to look back on the last 2 decades of bronchoscopy for peripheral lung nodules to get a better understanding of the value proposition robotic bronchoscopes may offer.
Guided bronchoscopy for lung nodules has significantly evolved over the past 2 decades, shifting diagnostic procedures from interventional radiologist to the pulmonologist. Some of these advances were based in redesigns of the bronchoscope (ultrathin bronchoscopy) or application of technology to the bronchoscope (radial EBUS, virtual bronchoscopy); but, these were not broadly applicable to the pulmonology community at large. It was not until the development of electromagnetic navigational bronchoscopy (ENB) that widespread adoption of bronchoscopy for lung nodules occurred. By and large, ENB fueled a rapid expansion of nodule bronchoscopy, mainly due to its ease of use and novel approach. Initial studies of ENB had impressive results; however, studies were criticized for having small numbers, inadequate follow-up, spurious definitions of yield, and that they were being done at highly specialized centers. The NAVIGATE trial was launched to address these criticisms among “real world” conditions. Sponsored by Medtronic, it studied ENB (superDimension platform, v6.0 or higher) across 29 academic and medical centers in the United States, enrolling over 1,000 patients (Folch EE, et al. J Thorac Oncol. 2019;14[3]:445-58. Epub 2018 Nov 23), and reported a diagnostic yield of 73%.
This led to a drive to improve upon yield, resulting in development of new technologies specifically designed to address some of the factors thought associated with diminished yield, and, out of this, robotic bronchoscopy was born. These factors included CT scan-body registration divergence, deflection of the extended working channel (EWC) by rigid biopsy tools, and inability to accurately “aim” the EWC-biopsy tool at the nodule; these were especially problematic in nodules not associated with airways. Robotic scopes were specifically designed to reach into the peripheral lung airways similar to an EWC, but with better structural integrity and steerability. This tip integrity would resist tool-related displacement, and steerability would allow for improved targeting of nodules during the biopsy.
There are two robots approved by the FDA at the time of this writing (Auris Monarch, Intuitive Ion), with a third awaiting FDA clearance (Noah Galaxy). In general, though the engineering of the robotic scopes to improve structural tip integrity are similar, the approach to navigation and targeting vary significantly. The Monarch platform uses electromagnetic guidance, similar to other traditional ENB platforms. The Ion platform does not use ENB; instead, it uses fiberoptic shape sensing technology, which analyzes the shape and orientation of the scope to provide location information. There are potential advantages to shape sensing, the most notable being the absence of electromagnetics; this allows for use of fluoroscopy during the procedure, which otherwise would have interfered with ENB-based navigation. There are other subtle differences between the two robots. The Monarch uses a scope-in-scope design, with a robotic scope contained within a robotic sheath; the Ion uses a single robotic scope. The Ion scope diameter is 3.5 mm, whereas the Monarch diameter is 4.4 mm; this may be a potential advantage when having to navigate through smaller airways.
So, which robot is better suited to reach peripheral nodules more consistently and accurately? I get asked this question a lot, since I have both platforms at my institution. But, answering with my own opinion based on my institution’s anecdotal experience would be irresponsible. I’m more of a “what does the data show?” person. Luckily, we do have clinical trials in both robot technologies. It should be noted here that there will likely never be a head-to-head randomized trial, so evaluating published studies with each platform is going to be the best method we have for comparison going forward, albeit an imperfect one. It should also be noted that many of the early robotic bronchoscopy trials have to be looked at with caution, as yield definitions tended not to be conservative and/or the follow-up of non-malignant was not robust. With that in mind, let’s review representative high-quality studies for each platform.
The best study to date using the Ion platform came out of Memorial Sloan Kettering Cancer Center (Kalchiem-Dekel O, et al. Chest. 2022;161[2];572-82). This single-site study reported on 159 nodule biopsies, with the primary outcome being diagnostic yield. The patients had 1 year of follow-up, and the definition of yield was conservative. The average lesion size was 18 mm, and nodule locations and characteristics were representative of real-world conditions. Overall diagnostic yield was 81.7%; however, it dropped to under 70% for nodules under 20 mm in size.
The largest study to date using the Monarch platform was also a single center study, this from the University of Chicago (Agrawal, et al. Ann Thorac Surg. 2022 Jan 17;S0003-4975(22)00042-X. Online ahead of print). This study included 124 nodules with at least 12 months of follow-up; diagnostic yield definition was conservative. Median nodule size was 20.5 mm, with distribution and characteristics representative of real-world conditions. Overall accuracy was 77%, and, similar to the Ion study, dropped to under 70% when nodule size was smaller than 20 mm.
Overall, both robot studies seemed to show a modest improvement in diagnostic yield when compared with ENB, and their outcomes were overall similar. It is important to remember that these were studies of each center’s first experiences with early versions of each technology; over time, the technology will continue to improve, as will operator skill and experience, and with that, perhaps improvements in yield will be seen, as well.
Interestingly, both studies evaluated target localization using radial EBUS (rEBUS), which also allowed for airway-nodule relationships to be reported. In Kalchiem-Dekel’s study, 85% of cases used rEBUS to determine localization, and, of these, 91.2% of cases showed accurate localization. In Agrawal’s study, rEBUS was used in all cases with a reported localization of 94%. In both, yield did not seem to be affected by airway-nodule relationships, perhaps explained by more robust tip control of the robotic scope. However, localization did not equate to yield in all cases, which brings up a very important question: Can the yield of robotic bronchoscopy be further improved with better real-time on-board imaging, such as CBCT scanning or C-arm based tomography? Currently, there is a study using 3D technology (Cios 3D Mobile Spin) in conjunction with the Ion platform to evaluate this.
So, let’s circle back to where we started. I think if you look at the totality of the data, it is clear that the robotic platforms currently offer a modest improvement in diagnostic yield over traditional ENB, with individual performances that are somewhat equivalent despite differences in design and operation. But does this improvement in yield justify the cost? Individual hospitals will have to make that decision. The capital cost and per-use price of the scope is significant, which has to be balanced against each center’s current performance with non-robotic bronchoscopy.
To date, there have been over 25,000 robotic procedures performed in the United States, so enthusiasm across diverse centers is being maintained. Whether this enthusiasm is driven by yield or novelty, or both, I’m not sure. With other nonrobotic platforms having reached, or soon to reach, the market, this is a good time to be in the business of bronchoscopy.
Dr. Cicenia is in the Section of Bronchoscopy at Cleveland Clinic’s Respiratory Institute, Cleveland, Ohio.












