User login
Doctors reconstruct noses, create vaginas using engineered human tissue
Researchers have successfully engineered human tissue from autologous tissue for the reconstruction of nasal alar lobules in one first-in-human trial while investigators in separate study were able to engineer and implant vaginas in young women with congenital aplagia.
The results move human tissue engineering closer to one day becoming mainstream health care, according to an editorial accompanying the two studies published in the Lancet.
"Progression from first-in-human experiences in only a few patients ... to a full integration into health systems involves many steps, and is often an uphill struggle," according to Dr. Martin A. Birchall, professor of laryngology at University College London, and Alexander M. Seifalian, Ph.D., professor of nanotechnology and regenerative medicine at the same institution, in an editorial on both studies (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60533-X]). However, because the human tissue engineering field is not unique in its need for larger trials and long-term follow-up to show efficacy in larger patient cohorts, further progress might be facilitated by the fact that "many countries now have large translational income streams, engaged biotech companies, and streamlined regulatory processes that might lower these barriers."
The two studies achieve three important milestones in the field of human tissue engineering, according to Dr. Birchall and Dr. Seifalian. They provide evidence that when biological scaffolds with or without cells are placed in the body, native tissue will grow in its place rather than scar tissue. And this can be achieved in pediatric populations.
The third – and possibly most important – milestone is that implanting large-scale engineered tissue is possible, since in situ angiogenesis was found to be adequate in the vaginal vaults constructed and implanted in the second study, wrote Dr. Birchall and Dr. Seifalian. The findings help to "edge tissue engineering towards the mainstream of organ and tissue replacement needs," they said.
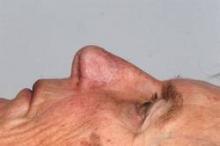
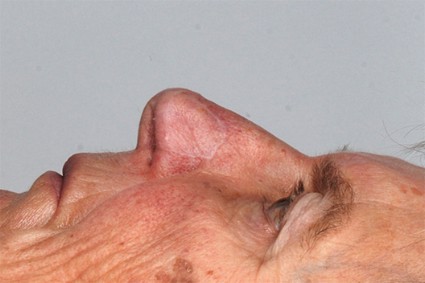
For the nasal reconstruction study, researchers used nasal septum cartilage cells from each of the five study participants to grow and shape cartilage grafts, which were then implanted in the respective patients whose alar lobule had been badly damaged by treatment for nonmelanoma skin cancer, according to Dr. Ilario Fulco and his associates in the departments of surgery and regenerative medicine at the University of Basel in Switzerland (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60544-4]).
Growth factor was used to expand the cells over the course of 2 weeks before they were seeded onto collagen membranes and cultured for another 2 weeks, until they grew into cartilage that was 40 times larger than the original biopsy. The cartilage was then shaped to fit the defect and was implanted.
At 1-year follow-up, all five reconstruction recipients reported they were satisfied with their ability to breathe, were happy with the look of their nose, and had not experienced any relevant adverse events.
"The engineered cartilage had clinical results comparable to the gold standard cartilage graft surgery," said Dr. Fulco. "This new technique could help the body accept the new tissue more easily, and improve the stability and functionality of the nostril."
Currently, for this kind of reconstruction, grafts are made using cartilage excised from the nasal septum, ear, or rib, which is painful and requires additional surgery, often fraught with complications.
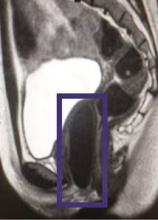
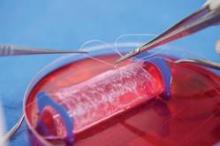
The second study dealt with four female patients, who had congenital vaginal aplagia. The investigators fashioned vaginal vaults from smooth muscle and vaginal epithelial cells taken from the patients’ own vulvar tissues and grown on vagina-shaped biodegradable scaffolds. The engineered vaults then were transplanted into each of the young women, who were aged 13-18 years at the time of surgery, according to Dr. Atlántida M. Raya-Rivera, a professor of bioengineering at the Metropolitan Autonomous University, Mexico City, and her associates.
At 8 years’ follow-up, all the vaginas were determined to be structurally and functionally normal. At that time, all four women reported that they were sexually active; were satisfied with their desire, arousal, lubrication, and orgasm; and did not experience pain during intercourse.
"Yearly tissue biopsy samples show that the reconstructed tissue is histologically and functionally similar to normal vaginal tissue," noted Dr. Raya-Rivera. The results mean that this method is a "viable" alternative to current vaginal reconstructive techniques because it requires only a small tissue sample and can help avoid the complications inherent when using nonvaginal tissue such as from the large intestine or skin; the complications include infections or grafts that shrink, she noted (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60542-0]).
"With the syndrome these women have, both the uterus and vagina can be missing or malformed. In this study, two of the patients were born with uteri, and there is potential for them to have children," Dr. Raya-Rivera said in an interview.
In both studies, the authors wrote respectively that further "fundamental studies" and "clinical experience" are warranted. Dr. Birchall and Dr. Seifalian concluded that the two studies show that "disruptive innovation might be nigh."
Funding for the study led by Dr. Fulco was provided by the department of surgery, University Hospital Basel, and Krebsliga bedier Basel; funding for the study led by Dr. Raya-Rivera was provided by Wake Forest University and Hospital Infantil de Mexico Federico Gomez. None of the researchers associated with the studies or the commentary reported having any relevant disclosures.
On Twitter @whitneymcknight
Researchers have successfully engineered human tissue from autologous tissue for the reconstruction of nasal alar lobules in one first-in-human trial while investigators in separate study were able to engineer and implant vaginas in young women with congenital aplagia.
The results move human tissue engineering closer to one day becoming mainstream health care, according to an editorial accompanying the two studies published in the Lancet.
"Progression from first-in-human experiences in only a few patients ... to a full integration into health systems involves many steps, and is often an uphill struggle," according to Dr. Martin A. Birchall, professor of laryngology at University College London, and Alexander M. Seifalian, Ph.D., professor of nanotechnology and regenerative medicine at the same institution, in an editorial on both studies (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60533-X]). However, because the human tissue engineering field is not unique in its need for larger trials and long-term follow-up to show efficacy in larger patient cohorts, further progress might be facilitated by the fact that "many countries now have large translational income streams, engaged biotech companies, and streamlined regulatory processes that might lower these barriers."
The two studies achieve three important milestones in the field of human tissue engineering, according to Dr. Birchall and Dr. Seifalian. They provide evidence that when biological scaffolds with or without cells are placed in the body, native tissue will grow in its place rather than scar tissue. And this can be achieved in pediatric populations.
The third – and possibly most important – milestone is that implanting large-scale engineered tissue is possible, since in situ angiogenesis was found to be adequate in the vaginal vaults constructed and implanted in the second study, wrote Dr. Birchall and Dr. Seifalian. The findings help to "edge tissue engineering towards the mainstream of organ and tissue replacement needs," they said.
For the nasal reconstruction study, researchers used nasal septum cartilage cells from each of the five study participants to grow and shape cartilage grafts, which were then implanted in the respective patients whose alar lobule had been badly damaged by treatment for nonmelanoma skin cancer, according to Dr. Ilario Fulco and his associates in the departments of surgery and regenerative medicine at the University of Basel in Switzerland (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60544-4]).
Growth factor was used to expand the cells over the course of 2 weeks before they were seeded onto collagen membranes and cultured for another 2 weeks, until they grew into cartilage that was 40 times larger than the original biopsy. The cartilage was then shaped to fit the defect and was implanted.
At 1-year follow-up, all five reconstruction recipients reported they were satisfied with their ability to breathe, were happy with the look of their nose, and had not experienced any relevant adverse events.
"The engineered cartilage had clinical results comparable to the gold standard cartilage graft surgery," said Dr. Fulco. "This new technique could help the body accept the new tissue more easily, and improve the stability and functionality of the nostril."
Currently, for this kind of reconstruction, grafts are made using cartilage excised from the nasal septum, ear, or rib, which is painful and requires additional surgery, often fraught with complications.
The second study dealt with four female patients, who had congenital vaginal aplagia. The investigators fashioned vaginal vaults from smooth muscle and vaginal epithelial cells taken from the patients’ own vulvar tissues and grown on vagina-shaped biodegradable scaffolds. The engineered vaults then were transplanted into each of the young women, who were aged 13-18 years at the time of surgery, according to Dr. Atlántida M. Raya-Rivera, a professor of bioengineering at the Metropolitan Autonomous University, Mexico City, and her associates.
At 8 years’ follow-up, all the vaginas were determined to be structurally and functionally normal. At that time, all four women reported that they were sexually active; were satisfied with their desire, arousal, lubrication, and orgasm; and did not experience pain during intercourse.
"Yearly tissue biopsy samples show that the reconstructed tissue is histologically and functionally similar to normal vaginal tissue," noted Dr. Raya-Rivera. The results mean that this method is a "viable" alternative to current vaginal reconstructive techniques because it requires only a small tissue sample and can help avoid the complications inherent when using nonvaginal tissue such as from the large intestine or skin; the complications include infections or grafts that shrink, she noted (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60542-0]).
"With the syndrome these women have, both the uterus and vagina can be missing or malformed. In this study, two of the patients were born with uteri, and there is potential for them to have children," Dr. Raya-Rivera said in an interview.
In both studies, the authors wrote respectively that further "fundamental studies" and "clinical experience" are warranted. Dr. Birchall and Dr. Seifalian concluded that the two studies show that "disruptive innovation might be nigh."
Funding for the study led by Dr. Fulco was provided by the department of surgery, University Hospital Basel, and Krebsliga bedier Basel; funding for the study led by Dr. Raya-Rivera was provided by Wake Forest University and Hospital Infantil de Mexico Federico Gomez. None of the researchers associated with the studies or the commentary reported having any relevant disclosures.
On Twitter @whitneymcknight
Researchers have successfully engineered human tissue from autologous tissue for the reconstruction of nasal alar lobules in one first-in-human trial while investigators in separate study were able to engineer and implant vaginas in young women with congenital aplagia.
The results move human tissue engineering closer to one day becoming mainstream health care, according to an editorial accompanying the two studies published in the Lancet.
"Progression from first-in-human experiences in only a few patients ... to a full integration into health systems involves many steps, and is often an uphill struggle," according to Dr. Martin A. Birchall, professor of laryngology at University College London, and Alexander M. Seifalian, Ph.D., professor of nanotechnology and regenerative medicine at the same institution, in an editorial on both studies (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60533-X]). However, because the human tissue engineering field is not unique in its need for larger trials and long-term follow-up to show efficacy in larger patient cohorts, further progress might be facilitated by the fact that "many countries now have large translational income streams, engaged biotech companies, and streamlined regulatory processes that might lower these barriers."
The two studies achieve three important milestones in the field of human tissue engineering, according to Dr. Birchall and Dr. Seifalian. They provide evidence that when biological scaffolds with or without cells are placed in the body, native tissue will grow in its place rather than scar tissue. And this can be achieved in pediatric populations.
The third – and possibly most important – milestone is that implanting large-scale engineered tissue is possible, since in situ angiogenesis was found to be adequate in the vaginal vaults constructed and implanted in the second study, wrote Dr. Birchall and Dr. Seifalian. The findings help to "edge tissue engineering towards the mainstream of organ and tissue replacement needs," they said.
For the nasal reconstruction study, researchers used nasal septum cartilage cells from each of the five study participants to grow and shape cartilage grafts, which were then implanted in the respective patients whose alar lobule had been badly damaged by treatment for nonmelanoma skin cancer, according to Dr. Ilario Fulco and his associates in the departments of surgery and regenerative medicine at the University of Basel in Switzerland (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60544-4]).
Growth factor was used to expand the cells over the course of 2 weeks before they were seeded onto collagen membranes and cultured for another 2 weeks, until they grew into cartilage that was 40 times larger than the original biopsy. The cartilage was then shaped to fit the defect and was implanted.
At 1-year follow-up, all five reconstruction recipients reported they were satisfied with their ability to breathe, were happy with the look of their nose, and had not experienced any relevant adverse events.
"The engineered cartilage had clinical results comparable to the gold standard cartilage graft surgery," said Dr. Fulco. "This new technique could help the body accept the new tissue more easily, and improve the stability and functionality of the nostril."
Currently, for this kind of reconstruction, grafts are made using cartilage excised from the nasal septum, ear, or rib, which is painful and requires additional surgery, often fraught with complications.
The second study dealt with four female patients, who had congenital vaginal aplagia. The investigators fashioned vaginal vaults from smooth muscle and vaginal epithelial cells taken from the patients’ own vulvar tissues and grown on vagina-shaped biodegradable scaffolds. The engineered vaults then were transplanted into each of the young women, who were aged 13-18 years at the time of surgery, according to Dr. Atlántida M. Raya-Rivera, a professor of bioengineering at the Metropolitan Autonomous University, Mexico City, and her associates.
At 8 years’ follow-up, all the vaginas were determined to be structurally and functionally normal. At that time, all four women reported that they were sexually active; were satisfied with their desire, arousal, lubrication, and orgasm; and did not experience pain during intercourse.
"Yearly tissue biopsy samples show that the reconstructed tissue is histologically and functionally similar to normal vaginal tissue," noted Dr. Raya-Rivera. The results mean that this method is a "viable" alternative to current vaginal reconstructive techniques because it requires only a small tissue sample and can help avoid the complications inherent when using nonvaginal tissue such as from the large intestine or skin; the complications include infections or grafts that shrink, she noted (Lancet 2014 April 11 [doi: 10.1016/S0140-6736(14)60542-0]).
"With the syndrome these women have, both the uterus and vagina can be missing or malformed. In this study, two of the patients were born with uteri, and there is potential for them to have children," Dr. Raya-Rivera said in an interview.
In both studies, the authors wrote respectively that further "fundamental studies" and "clinical experience" are warranted. Dr. Birchall and Dr. Seifalian concluded that the two studies show that "disruptive innovation might be nigh."
Funding for the study led by Dr. Fulco was provided by the department of surgery, University Hospital Basel, and Krebsliga bedier Basel; funding for the study led by Dr. Raya-Rivera was provided by Wake Forest University and Hospital Infantil de Mexico Federico Gomez. None of the researchers associated with the studies or the commentary reported having any relevant disclosures.
On Twitter @whitneymcknight
FROM THE LANCET
Major finding: Nasal septum cartilage cells and vaginal epithelial cells were used to grow human tissue for implantation.
Data source: An observational first-in-human trial of nasal reconstruction post skin cancer in five patients, and a pilot cohort study of vaginal implantation in four patients who had congenital vaginal aplagia.
Disclosures: Funding for the study led by Dr. Fulco was provided by the department of surgery, University Hospital Basel, and Krebsliga bedier Basel; funding for the study led by Dr. Raya-Rivera was provided by Wake Forest University and Hospital Infantil de Mexico Federico Gomez. None of the researchers associated with the studies or the commentary reported having any relevant disclosures.
Adjunctive ingenol mebutate gel for actinic keratosis caused few side effects, increased compliance
Ingenol mebutate gel for the treatment of actinic keratosis is an effective adjunct therapy to cryosurgery, and the absence of attendant serious local reactions can increase compliance, based on data from more than 100 patients.
"By the time you do see a reaction, because the treatment time is only 2 or 3 days, the patient is done applying it, so the compliance is increased," Dr. Miriam S. Bettencourt, a community-based dermatologist in Las Vegas.
Dr. Bettencourt conducted her chart review of 135 patients "out of curiosity," she said in an interview. She found that at follow-ups of 1-4 months, nearly 100% of patients who received ingenol mebutate gel as an adjunctive therapy with cryosurgery for AK on the face had clearance rates of at least 75%. In addition, clearance of at least 75% was seen in 89% of patients treated on the scalp and 84% of patients treated on the forearms and/or hands. Most of the patients were male (88%), ranging in age from 39 to 85 years, with a median age of 70 years.
"One of the questions I had was, in a clinical practice, whether [the gel] mimics the clinical trial results that showed only a mild to moderate reaction," said Dr. Bettencourt. Common side affects of topical AK treatments include erythema, flaking, scaling, and crusting. In more extreme cases, vesicular pustulation is possible.
"I found that patients really didn’t complain of pain," said Dr. Bettencourt. "Most local skin reactions were resolved within a week." She prescribed moisturizers for patients whose reactions were moderate.
The findings were published in the March issue of the Journal of Drugs in Dermatology (J. Drugs Dermatol. 2014:13:269-73).
The results negate the common tenet that the more severe the reaction to the topical, the deeper the healing of the AK, said Dr. Bettencourt. "The degree of reaction does not correlate with the level of improvement. We used to think so, but that’s not true." The results show that the reaction can be "tolerable" and short lived despite dramatic results, she said.
The gel was approved by the Food and Drug Administration for use in the United States in early 2012 under the name Picato (LEO Pharmaceuticals). Unlike other topicals that must be applied over the course of weeks, the gel is applied for 2 or 3 days, with side effects lasting much of that time.
In addition to increased compliance, Dr. Bettencourt said she also measured whether her off-label "creativity" such as prescribing higher concentrations for the scalp and instructing patients to spread the gel over larger areas, yielded worthwhile results.
"I found that, especially for the face, you can spread the medication without jeopardizing the efficacy," said Dr. Bettencourt. However, when the gel is applied to the scalp, "You lose a little bit of the efficacy, because that’s a lot to cover using the prescribed three tubes, but you still get good results."
In addition, Dr. Bettencourt said that the gel offers cosmetic advantages over other treatments such as the topical imiquimod or photodynamic therapy because there is no hypo- or hyperpigmentation. And, because there is no photosensitivity associated with the gel, Dr. Bettencourt said she can offer it year-round.
She still prescribes other topicals, however, because not all patients’ insurance plans cover the gel. Medicare, does cover the gel, she noted, but only if the patient has a supplemental insurance policy.
While the study was unsponsored, and Dr. Bettencourt had no financial conflicts to disclose, she noted that she has shared her data with LEO Pharmaceuticals, the gel’s manufacturer. "They were thrilled, and helped me to put the results together," she said.
On Twitter @whitneymcknight
Ingenol mebutate gel for the treatment of actinic keratosis is an effective adjunct therapy to cryosurgery, and the absence of attendant serious local reactions can increase compliance, based on data from more than 100 patients.
"By the time you do see a reaction, because the treatment time is only 2 or 3 days, the patient is done applying it, so the compliance is increased," Dr. Miriam S. Bettencourt, a community-based dermatologist in Las Vegas.
Dr. Bettencourt conducted her chart review of 135 patients "out of curiosity," she said in an interview. She found that at follow-ups of 1-4 months, nearly 100% of patients who received ingenol mebutate gel as an adjunctive therapy with cryosurgery for AK on the face had clearance rates of at least 75%. In addition, clearance of at least 75% was seen in 89% of patients treated on the scalp and 84% of patients treated on the forearms and/or hands. Most of the patients were male (88%), ranging in age from 39 to 85 years, with a median age of 70 years.
"One of the questions I had was, in a clinical practice, whether [the gel] mimics the clinical trial results that showed only a mild to moderate reaction," said Dr. Bettencourt. Common side affects of topical AK treatments include erythema, flaking, scaling, and crusting. In more extreme cases, vesicular pustulation is possible.
"I found that patients really didn’t complain of pain," said Dr. Bettencourt. "Most local skin reactions were resolved within a week." She prescribed moisturizers for patients whose reactions were moderate.
The findings were published in the March issue of the Journal of Drugs in Dermatology (J. Drugs Dermatol. 2014:13:269-73).
The results negate the common tenet that the more severe the reaction to the topical, the deeper the healing of the AK, said Dr. Bettencourt. "The degree of reaction does not correlate with the level of improvement. We used to think so, but that’s not true." The results show that the reaction can be "tolerable" and short lived despite dramatic results, she said.
The gel was approved by the Food and Drug Administration for use in the United States in early 2012 under the name Picato (LEO Pharmaceuticals). Unlike other topicals that must be applied over the course of weeks, the gel is applied for 2 or 3 days, with side effects lasting much of that time.
In addition to increased compliance, Dr. Bettencourt said she also measured whether her off-label "creativity" such as prescribing higher concentrations for the scalp and instructing patients to spread the gel over larger areas, yielded worthwhile results.
"I found that, especially for the face, you can spread the medication without jeopardizing the efficacy," said Dr. Bettencourt. However, when the gel is applied to the scalp, "You lose a little bit of the efficacy, because that’s a lot to cover using the prescribed three tubes, but you still get good results."
In addition, Dr. Bettencourt said that the gel offers cosmetic advantages over other treatments such as the topical imiquimod or photodynamic therapy because there is no hypo- or hyperpigmentation. And, because there is no photosensitivity associated with the gel, Dr. Bettencourt said she can offer it year-round.
She still prescribes other topicals, however, because not all patients’ insurance plans cover the gel. Medicare, does cover the gel, she noted, but only if the patient has a supplemental insurance policy.
While the study was unsponsored, and Dr. Bettencourt had no financial conflicts to disclose, she noted that she has shared her data with LEO Pharmaceuticals, the gel’s manufacturer. "They were thrilled, and helped me to put the results together," she said.
On Twitter @whitneymcknight
Ingenol mebutate gel for the treatment of actinic keratosis is an effective adjunct therapy to cryosurgery, and the absence of attendant serious local reactions can increase compliance, based on data from more than 100 patients.
"By the time you do see a reaction, because the treatment time is only 2 or 3 days, the patient is done applying it, so the compliance is increased," Dr. Miriam S. Bettencourt, a community-based dermatologist in Las Vegas.
Dr. Bettencourt conducted her chart review of 135 patients "out of curiosity," she said in an interview. She found that at follow-ups of 1-4 months, nearly 100% of patients who received ingenol mebutate gel as an adjunctive therapy with cryosurgery for AK on the face had clearance rates of at least 75%. In addition, clearance of at least 75% was seen in 89% of patients treated on the scalp and 84% of patients treated on the forearms and/or hands. Most of the patients were male (88%), ranging in age from 39 to 85 years, with a median age of 70 years.
"One of the questions I had was, in a clinical practice, whether [the gel] mimics the clinical trial results that showed only a mild to moderate reaction," said Dr. Bettencourt. Common side affects of topical AK treatments include erythema, flaking, scaling, and crusting. In more extreme cases, vesicular pustulation is possible.
"I found that patients really didn’t complain of pain," said Dr. Bettencourt. "Most local skin reactions were resolved within a week." She prescribed moisturizers for patients whose reactions were moderate.
The findings were published in the March issue of the Journal of Drugs in Dermatology (J. Drugs Dermatol. 2014:13:269-73).
The results negate the common tenet that the more severe the reaction to the topical, the deeper the healing of the AK, said Dr. Bettencourt. "The degree of reaction does not correlate with the level of improvement. We used to think so, but that’s not true." The results show that the reaction can be "tolerable" and short lived despite dramatic results, she said.
The gel was approved by the Food and Drug Administration for use in the United States in early 2012 under the name Picato (LEO Pharmaceuticals). Unlike other topicals that must be applied over the course of weeks, the gel is applied for 2 or 3 days, with side effects lasting much of that time.
In addition to increased compliance, Dr. Bettencourt said she also measured whether her off-label "creativity" such as prescribing higher concentrations for the scalp and instructing patients to spread the gel over larger areas, yielded worthwhile results.
"I found that, especially for the face, you can spread the medication without jeopardizing the efficacy," said Dr. Bettencourt. However, when the gel is applied to the scalp, "You lose a little bit of the efficacy, because that’s a lot to cover using the prescribed three tubes, but you still get good results."
In addition, Dr. Bettencourt said that the gel offers cosmetic advantages over other treatments such as the topical imiquimod or photodynamic therapy because there is no hypo- or hyperpigmentation. And, because there is no photosensitivity associated with the gel, Dr. Bettencourt said she can offer it year-round.
She still prescribes other topicals, however, because not all patients’ insurance plans cover the gel. Medicare, does cover the gel, she noted, but only if the patient has a supplemental insurance policy.
While the study was unsponsored, and Dr. Bettencourt had no financial conflicts to disclose, she noted that she has shared her data with LEO Pharmaceuticals, the gel’s manufacturer. "They were thrilled, and helped me to put the results together," she said.
On Twitter @whitneymcknight
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
Major finding: Nearly 100% of patients achieved at least 75% or greater clearance of AK on the face; more than 80% of patients achieved at least 75% or greater AK resolution on the scalp, forearm, and/or hand.
Data source: Retrospective chart review of 135 patients, median age 70 years, in a community practice.
Disclosures: Dr. Bettencourt had no conflicts of interest to disclose.
Getting Payers to Cover Obesity Treatment
WASHINGTON – Payers will cover obesity treatment, people will suffer less from cardiometabolic disease, and the economy will be less burdened by the costs of untreated complications and lost productivity related to obesity. That’s the vision of Dr. Jeffrey I. Mechanick and his colleagues at the American Association of Clinical Endocrinologists and the American College of Endocrinology, who’ve jointly released a consensus statement on obesity treatment.
In a video interview, AACE president Dr. Mechanick, discusses why payers haven’t covered obesity treatment as a matter of course in the past, but why they may going forward, and how a growing impatience with a fractured approach to obesity care spurred the AACE and ACE to create a cross-disciplinary conference that also included members of the nonmedical professions involved in obesity care.
"By limiting the conference just to the biomedical model, we wouldn’t have access to this type of information," Dr. Mechanick also of the Icahn School of Medicine at Mount Sinai, New York, said in a press conference at the meeting. "We learned that different stakeholders require different levels of evidence."
The value of measuring body mass index is a controversial topic in obesity treatment. Dr. Mechanick explains why BMI will still be part of the developing ACCE/ACE standards of care and how it will be incorporated with other factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Payers will cover obesity treatment, people will suffer less from cardiometabolic disease, and the economy will be less burdened by the costs of untreated complications and lost productivity related to obesity. That’s the vision of Dr. Jeffrey I. Mechanick and his colleagues at the American Association of Clinical Endocrinologists and the American College of Endocrinology, who’ve jointly released a consensus statement on obesity treatment.
In a video interview, AACE president Dr. Mechanick, discusses why payers haven’t covered obesity treatment as a matter of course in the past, but why they may going forward, and how a growing impatience with a fractured approach to obesity care spurred the AACE and ACE to create a cross-disciplinary conference that also included members of the nonmedical professions involved in obesity care.
"By limiting the conference just to the biomedical model, we wouldn’t have access to this type of information," Dr. Mechanick also of the Icahn School of Medicine at Mount Sinai, New York, said in a press conference at the meeting. "We learned that different stakeholders require different levels of evidence."
The value of measuring body mass index is a controversial topic in obesity treatment. Dr. Mechanick explains why BMI will still be part of the developing ACCE/ACE standards of care and how it will be incorporated with other factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Payers will cover obesity treatment, people will suffer less from cardiometabolic disease, and the economy will be less burdened by the costs of untreated complications and lost productivity related to obesity. That’s the vision of Dr. Jeffrey I. Mechanick and his colleagues at the American Association of Clinical Endocrinologists and the American College of Endocrinology, who’ve jointly released a consensus statement on obesity treatment.
In a video interview, AACE president Dr. Mechanick, discusses why payers haven’t covered obesity treatment as a matter of course in the past, but why they may going forward, and how a growing impatience with a fractured approach to obesity care spurred the AACE and ACE to create a cross-disciplinary conference that also included members of the nonmedical professions involved in obesity care.
"By limiting the conference just to the biomedical model, we wouldn’t have access to this type of information," Dr. Mechanick also of the Icahn School of Medicine at Mount Sinai, New York, said in a press conference at the meeting. "We learned that different stakeholders require different levels of evidence."
The value of measuring body mass index is a controversial topic in obesity treatment. Dr. Mechanick explains why BMI will still be part of the developing ACCE/ACE standards of care and how it will be incorporated with other factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: Ketamine, Botox, and SAMe: What will be approved for depression?
CHICAGO – Research continues into whether the anesthetic ketamine will deliver on what some see as its potential to be a notably effective treatment for refractory depression, but other medical treatments are also under review.
Dr. James Jefferson scanned the depression treatment horizon in a presentation at Psychiatry Update 2014, sponsored by Current Psychiatry and the American Academy of Clinical Psychiatrists. In a video interview, Dr. Jefferson of the University of Wisconsin, Madison, forecasts what he thinks will – or won’t – enter the armamentarium. Might that include Botox? And how promising is the nutritional supplement SAMe?
Dr. Jefferson shares his thoughts on these and other contenders, and he considers when atypical antipsychotics are appropriate for treating depression. He also delivers stern words to clinicians who do not read the package inserts in the medications they prescribe.
Current Psychiatry and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
CHICAGO – Research continues into whether the anesthetic ketamine will deliver on what some see as its potential to be a notably effective treatment for refractory depression, but other medical treatments are also under review.
Dr. James Jefferson scanned the depression treatment horizon in a presentation at Psychiatry Update 2014, sponsored by Current Psychiatry and the American Academy of Clinical Psychiatrists. In a video interview, Dr. Jefferson of the University of Wisconsin, Madison, forecasts what he thinks will – or won’t – enter the armamentarium. Might that include Botox? And how promising is the nutritional supplement SAMe?
Dr. Jefferson shares his thoughts on these and other contenders, and he considers when atypical antipsychotics are appropriate for treating depression. He also delivers stern words to clinicians who do not read the package inserts in the medications they prescribe.
Current Psychiatry and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
CHICAGO – Research continues into whether the anesthetic ketamine will deliver on what some see as its potential to be a notably effective treatment for refractory depression, but other medical treatments are also under review.
Dr. James Jefferson scanned the depression treatment horizon in a presentation at Psychiatry Update 2014, sponsored by Current Psychiatry and the American Academy of Clinical Psychiatrists. In a video interview, Dr. Jefferson of the University of Wisconsin, Madison, forecasts what he thinks will – or won’t – enter the armamentarium. Might that include Botox? And how promising is the nutritional supplement SAMe?
Dr. Jefferson shares his thoughts on these and other contenders, and he considers when atypical antipsychotics are appropriate for treating depression. He also delivers stern words to clinicians who do not read the package inserts in the medications they prescribe.
Current Psychiatry and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
EXPERT ANALYSIS FROM PSYCHIATRY UPDATE 2014
AUDIO: Despite low efficacy in OCD, adjunct antipsychotics still prescribed
CHICAGO – At best, only a third of adults treated with selective serotonin reuptake inhibitors for obsessive-compulsive disorder respond to adjunctive therapy with the second-generation antipsychotic risperidone.
That’s according to Dr. H. Blair Simpson, professor of psychiatry at Columbia University and the director of the Center for Obsessive-Compulsive and Related Disorders and the Anxiety and Related Disorders Clinic at the New York State Psychiatric Institute/Columbia University.
"And if they do, typically you will know within about 4 weeks," said Dr. Simpson at the annual conference of the Anxiety and Depression Association of America.
So, why do so many patients with OCD – even pediatric ones for whom there are even fewer efficacy data – receive the second-generation antipsychotic (SGA) instead of cognitive-behavioral therapy, which does have more efficacy data? And for patients who do respond, what is the best way to titrate them off the SGA?
In an interview, Dr. Simpson and her colleague Dr. Moira Rynn, director of the Child and Adolescent Psychiatric Evaluation Service at the New York State Psychiatric Institute/Columbia University, explore the answers to these questions, as well as discuss whether two standards of mental health care are developing by default – and if so, why, as academic centers offer a wider range of treatments for refractory cases of OCD than do community-based clinicians.
CHICAGO – At best, only a third of adults treated with selective serotonin reuptake inhibitors for obsessive-compulsive disorder respond to adjunctive therapy with the second-generation antipsychotic risperidone.
That’s according to Dr. H. Blair Simpson, professor of psychiatry at Columbia University and the director of the Center for Obsessive-Compulsive and Related Disorders and the Anxiety and Related Disorders Clinic at the New York State Psychiatric Institute/Columbia University.
"And if they do, typically you will know within about 4 weeks," said Dr. Simpson at the annual conference of the Anxiety and Depression Association of America.
So, why do so many patients with OCD – even pediatric ones for whom there are even fewer efficacy data – receive the second-generation antipsychotic (SGA) instead of cognitive-behavioral therapy, which does have more efficacy data? And for patients who do respond, what is the best way to titrate them off the SGA?
In an interview, Dr. Simpson and her colleague Dr. Moira Rynn, director of the Child and Adolescent Psychiatric Evaluation Service at the New York State Psychiatric Institute/Columbia University, explore the answers to these questions, as well as discuss whether two standards of mental health care are developing by default – and if so, why, as academic centers offer a wider range of treatments for refractory cases of OCD than do community-based clinicians.
CHICAGO – At best, only a third of adults treated with selective serotonin reuptake inhibitors for obsessive-compulsive disorder respond to adjunctive therapy with the second-generation antipsychotic risperidone.
That’s according to Dr. H. Blair Simpson, professor of psychiatry at Columbia University and the director of the Center for Obsessive-Compulsive and Related Disorders and the Anxiety and Related Disorders Clinic at the New York State Psychiatric Institute/Columbia University.
"And if they do, typically you will know within about 4 weeks," said Dr. Simpson at the annual conference of the Anxiety and Depression Association of America.
So, why do so many patients with OCD – even pediatric ones for whom there are even fewer efficacy data – receive the second-generation antipsychotic (SGA) instead of cognitive-behavioral therapy, which does have more efficacy data? And for patients who do respond, what is the best way to titrate them off the SGA?
In an interview, Dr. Simpson and her colleague Dr. Moira Rynn, director of the Child and Adolescent Psychiatric Evaluation Service at the New York State Psychiatric Institute/Columbia University, explore the answers to these questions, as well as discuss whether two standards of mental health care are developing by default – and if so, why, as academic centers offer a wider range of treatments for refractory cases of OCD than do community-based clinicians.
EXPERT ANALYSIS FROM THE AADA ANNUAL CONFERENCE
Olokizumab showed promise for RA patients nonresponsive to antitumor necrosis factor therapies
Olokizumab, a humanized monoclonal antibody specific for the interleukin-6 cytokine, showed promise for patients with rheumatoid arthritis who had an inadequate response to antitumor necrosis factor therapies in a 12-week, phase II trial.
Olokizumab showed comparable efficacy to tocilizumab in this patient population and at doses in line with this class of drug. Treatment-related adverse events and laboratory profiles in the trial also were consistent with the use of an IL-6 targeted therapy in patients with moderate to severe rheumatoid arthritis.
The randomized, double-blind, placebo- and active-controlled multicenter trial of 221 patients measured changes in 28-joint and C-reactive protein Disease Activity Score (DAS28-CRP) at week 12 as its primary endpoint. Secondary endpoints included the American College of Rheumatology (ACR) 20, ACR50, and ACR70 response rates at week 12. The patients were randomly assigned to one of nine treatment arms, receiving either an intravenous subcutaneous injection of the trial drug or placebo every 2 weeks or every 4 weeks in dosages of 60 mg, 120 mg, or 240 mg, or an 8 mg/kg infusion of tocilizumab (Actemra) every 4 weeks. Patients who were not randomized to tocilizumab also received a placebo infusion every 4 weeks (Ann. Rheum. Dis. 2014 March 18 [doi:10.1136/annrheumdis-2013-204760]).
First author Dr. Mark C. Genovese of Stanford (Calif.) University and his associates found that at week 12, olokizumab produced greater reductions in baseline DAS28-CRP scores than did placebo (P less than .001), regardless of the dose tested. ACR20 and ACR50 responses were numerically higher in the test group than in placebo, but the trial was not powered to detect between-group differences in ACR endpoints. The investigators recorded ACR20 responses in 32.5%-60.7% of olokizumab-treated patients and in 17.1%-29.9% of placebo-treated patients. ACR50 responses occurred in 11.5%-33.2% of patients who received olokizumab, compared with 1.3%-4.9% of placebo patients. Few patients in any treatment arm were ACR70 responders.
Regardless of dose, olokizumab demonstrated comparable efficacy to tocilizumab across multiple endpoints. Most adverse events, such as upper respiratory infection or gastrointestinal disorders, were mild to moderate, and did not vary significantly between the two drug treatment groups.
Funding for the study was provided by UCB Pharma. Dr. Genovese disclosed he receives funding from UCB beyond the scope of this study. The other researchers involved in this study disclosed an extensive list of industry relationships.
Olokizumab, a humanized monoclonal antibody specific for the interleukin-6 cytokine, showed promise for patients with rheumatoid arthritis who had an inadequate response to antitumor necrosis factor therapies in a 12-week, phase II trial.
Olokizumab showed comparable efficacy to tocilizumab in this patient population and at doses in line with this class of drug. Treatment-related adverse events and laboratory profiles in the trial also were consistent with the use of an IL-6 targeted therapy in patients with moderate to severe rheumatoid arthritis.
The randomized, double-blind, placebo- and active-controlled multicenter trial of 221 patients measured changes in 28-joint and C-reactive protein Disease Activity Score (DAS28-CRP) at week 12 as its primary endpoint. Secondary endpoints included the American College of Rheumatology (ACR) 20, ACR50, and ACR70 response rates at week 12. The patients were randomly assigned to one of nine treatment arms, receiving either an intravenous subcutaneous injection of the trial drug or placebo every 2 weeks or every 4 weeks in dosages of 60 mg, 120 mg, or 240 mg, or an 8 mg/kg infusion of tocilizumab (Actemra) every 4 weeks. Patients who were not randomized to tocilizumab also received a placebo infusion every 4 weeks (Ann. Rheum. Dis. 2014 March 18 [doi:10.1136/annrheumdis-2013-204760]).
First author Dr. Mark C. Genovese of Stanford (Calif.) University and his associates found that at week 12, olokizumab produced greater reductions in baseline DAS28-CRP scores than did placebo (P less than .001), regardless of the dose tested. ACR20 and ACR50 responses were numerically higher in the test group than in placebo, but the trial was not powered to detect between-group differences in ACR endpoints. The investigators recorded ACR20 responses in 32.5%-60.7% of olokizumab-treated patients and in 17.1%-29.9% of placebo-treated patients. ACR50 responses occurred in 11.5%-33.2% of patients who received olokizumab, compared with 1.3%-4.9% of placebo patients. Few patients in any treatment arm were ACR70 responders.
Regardless of dose, olokizumab demonstrated comparable efficacy to tocilizumab across multiple endpoints. Most adverse events, such as upper respiratory infection or gastrointestinal disorders, were mild to moderate, and did not vary significantly between the two drug treatment groups.
Funding for the study was provided by UCB Pharma. Dr. Genovese disclosed he receives funding from UCB beyond the scope of this study. The other researchers involved in this study disclosed an extensive list of industry relationships.
Olokizumab, a humanized monoclonal antibody specific for the interleukin-6 cytokine, showed promise for patients with rheumatoid arthritis who had an inadequate response to antitumor necrosis factor therapies in a 12-week, phase II trial.
Olokizumab showed comparable efficacy to tocilizumab in this patient population and at doses in line with this class of drug. Treatment-related adverse events and laboratory profiles in the trial also were consistent with the use of an IL-6 targeted therapy in patients with moderate to severe rheumatoid arthritis.
The randomized, double-blind, placebo- and active-controlled multicenter trial of 221 patients measured changes in 28-joint and C-reactive protein Disease Activity Score (DAS28-CRP) at week 12 as its primary endpoint. Secondary endpoints included the American College of Rheumatology (ACR) 20, ACR50, and ACR70 response rates at week 12. The patients were randomly assigned to one of nine treatment arms, receiving either an intravenous subcutaneous injection of the trial drug or placebo every 2 weeks or every 4 weeks in dosages of 60 mg, 120 mg, or 240 mg, or an 8 mg/kg infusion of tocilizumab (Actemra) every 4 weeks. Patients who were not randomized to tocilizumab also received a placebo infusion every 4 weeks (Ann. Rheum. Dis. 2014 March 18 [doi:10.1136/annrheumdis-2013-204760]).
First author Dr. Mark C. Genovese of Stanford (Calif.) University and his associates found that at week 12, olokizumab produced greater reductions in baseline DAS28-CRP scores than did placebo (P less than .001), regardless of the dose tested. ACR20 and ACR50 responses were numerically higher in the test group than in placebo, but the trial was not powered to detect between-group differences in ACR endpoints. The investigators recorded ACR20 responses in 32.5%-60.7% of olokizumab-treated patients and in 17.1%-29.9% of placebo-treated patients. ACR50 responses occurred in 11.5%-33.2% of patients who received olokizumab, compared with 1.3%-4.9% of placebo patients. Few patients in any treatment arm were ACR70 responders.
Regardless of dose, olokizumab demonstrated comparable efficacy to tocilizumab across multiple endpoints. Most adverse events, such as upper respiratory infection or gastrointestinal disorders, were mild to moderate, and did not vary significantly between the two drug treatment groups.
Funding for the study was provided by UCB Pharma. Dr. Genovese disclosed he receives funding from UCB beyond the scope of this study. The other researchers involved in this study disclosed an extensive list of industry relationships.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major finding: Olokizumab at all dose levels produced a greater reduction in DAS28-CRP than did placebo (P less than .001).
Data source: A phase II, randomized, double-blind, placebo- and active-controlled multicenter trial of 221 patients with moderate to severe RA who previously did not respond to antitumor necrosis factor therapies.
Disclosures: Funding for the study was provided by UCB Pharma. Dr. Genovese disclosed he receives funding from UCB beyond the scope of this study. The other researchers involved in this study disclosed an extensive list of industry relationships.
Evidence needed on obesity definition, treatment, AACE declares
WASHINGTON – Obesity requires a medical definition that goes beyond gauging a person’s body mass index if cost-effective care is to be delivered in an integrated fashion, according to a consensus statement issued by the American Association of Clinical Endocrinologists and the American College of Endocrinology.
"The definition of obesity as a disease is not perfect," Dr. W. Timothy Garvey, who chaired the AACE/ACE Obesity Consensus Conference, said in a media briefing. "We rely upon an [anthropometric] measure of body mass index, which is a measure of height versus weight, and there was consensus that this was ... divorced from the impact of weight gain on the health of the individual. This imprecision in our diagnosis of obesity was constraining us."
In 2013, the American Medical Association officially recognized obesity as a disease. Better codification of what actually constitutes "obesity, the disease," will allow a more integrated and effective approach to treating it, said Dr. Garvey, professor of medicine and chair of the department of nutrition sciences at the University of Alabama at Birmingham. To do so, the AACE/ACE held an intensive, 2-day session that largely featured spontaneous discussions between panelists and audience members representing four specific obesity "pillars": biomedical, government and regulation, health industry and economics, and research and education sectors.
A constant theme across the sectors was the need for a definition of obesity that accounts for cultural differences, ethnicity, and the presence or absence of cardiometabolic markers of disease in persons who are overweight or obese.
The conference’s multidisciplined approach informed the consensus statement that obesity is a chronic disease that should be treated with the established AACE/ACE obesity algorithm and met with lifestyle interventions. The consensus statement also addressed our current "obesogenic" environment, which many participants said was created in part by the abundance of nonnutritious foods.
In an interview, Dr. Susan Kansagra, deputy commissioner of the New York City Department of Health and Mental Hygiene, said that by working with local vendors and their suppliers, among other actions, her agency is focused on increasing access to more nutritious foods in neighborhoods across the city as a way to shape the food environment. "It’s not people who’ve changed over the past 30 years; it’s the environment," Dr. Kansagra said at the conference.
Also addressed by the consensus statement was the need for preventive care, particularly at the pediatric level, and more cohesive public awareness campaigns that could affect how private payers develop their reimbursement strategies. Audience member Dr. Robert Silverman, medical director of CIGNA Healthcare, said that payers would respond to the need for obesity care, but that what currently is missing is "a tie between the evidence and the complications [of obesity]."
"We learned that different stakeholders require different levels of evidence," AACE President Jeffrey I. Mechanick said in the media briefing. "So, we’re going to be able come up with a more efficient way to make recommendations about research so that private insurance carriers, the Centers for Medicare & Medicaid Services, or regulatory agencies have the type of data they require to facilitate the action [they need]."
These differences were brought to light during the conference as various audience members representing the Centers for Disease Control and Prevention, the Food and Drug Administration, the CMS, the National Institutes of Health, and others involved in research and policy making, addressed the panel to either explain or defend why their agency operates as it does.
In the case of the CMS, a statutory organization, it can apply coverage only according to what the agency is mandated to do, said Dr. Elizabeth Koller of the CMS. The level of evidence the agency looks for, she said, includes "hard endpoints of clinical relevance, like reductions in sleep apnea and degenerative joint disease." The CMS is also concerned about the lack of long-term data on interventions, the durability of interventions, and which characteristics are common in people who relapse in their disease, said Dr. Koller, who addressed the group as an audience member.
"Hearing from the CMS was incredibly helpful. We learned so much," said Dr. Mechanick, director of metabolic support at the Mt. Sinai School of Medicine in New York, in an interview.
Dr. Mechanick also said this was the first of three meetings, the next to be held in about a year, where the ultimate goal would be to use the evidence base they will have created to develop recommendations for all involved in delivering obesity care.
The talk was "polite," Dr. John Morton, chief of bariatric surgery at Stanford (Calif.) University and president of the American Society of Bariatric and Metabolic Surgery, said in an interview, but he said he thinks there is bias against people with obesity. "We wouldn’t be having this discussion if it were about cancer," he said in the interview. "Sometimes we think the consequences of obesity are the result of a personal decision, and that may skew people in a direction where they don’t necessarily want to provide help."
Regardless, Dr. Garvey said at the briefing, "the ‘old world’ thinking that obesity is a lifestyle choice has failed us."
Dr. Mechanick is a consultant for Abbott Nutrition. Dr. Garvey has multiple industry relationships, including with Merck, Vivus, and Eisai.
WASHINGTON – Obesity requires a medical definition that goes beyond gauging a person’s body mass index if cost-effective care is to be delivered in an integrated fashion, according to a consensus statement issued by the American Association of Clinical Endocrinologists and the American College of Endocrinology.
"The definition of obesity as a disease is not perfect," Dr. W. Timothy Garvey, who chaired the AACE/ACE Obesity Consensus Conference, said in a media briefing. "We rely upon an [anthropometric] measure of body mass index, which is a measure of height versus weight, and there was consensus that this was ... divorced from the impact of weight gain on the health of the individual. This imprecision in our diagnosis of obesity was constraining us."
In 2013, the American Medical Association officially recognized obesity as a disease. Better codification of what actually constitutes "obesity, the disease," will allow a more integrated and effective approach to treating it, said Dr. Garvey, professor of medicine and chair of the department of nutrition sciences at the University of Alabama at Birmingham. To do so, the AACE/ACE held an intensive, 2-day session that largely featured spontaneous discussions between panelists and audience members representing four specific obesity "pillars": biomedical, government and regulation, health industry and economics, and research and education sectors.
A constant theme across the sectors was the need for a definition of obesity that accounts for cultural differences, ethnicity, and the presence or absence of cardiometabolic markers of disease in persons who are overweight or obese.
The conference’s multidisciplined approach informed the consensus statement that obesity is a chronic disease that should be treated with the established AACE/ACE obesity algorithm and met with lifestyle interventions. The consensus statement also addressed our current "obesogenic" environment, which many participants said was created in part by the abundance of nonnutritious foods.
In an interview, Dr. Susan Kansagra, deputy commissioner of the New York City Department of Health and Mental Hygiene, said that by working with local vendors and their suppliers, among other actions, her agency is focused on increasing access to more nutritious foods in neighborhoods across the city as a way to shape the food environment. "It’s not people who’ve changed over the past 30 years; it’s the environment," Dr. Kansagra said at the conference.
Also addressed by the consensus statement was the need for preventive care, particularly at the pediatric level, and more cohesive public awareness campaigns that could affect how private payers develop their reimbursement strategies. Audience member Dr. Robert Silverman, medical director of CIGNA Healthcare, said that payers would respond to the need for obesity care, but that what currently is missing is "a tie between the evidence and the complications [of obesity]."
"We learned that different stakeholders require different levels of evidence," AACE President Jeffrey I. Mechanick said in the media briefing. "So, we’re going to be able come up with a more efficient way to make recommendations about research so that private insurance carriers, the Centers for Medicare & Medicaid Services, or regulatory agencies have the type of data they require to facilitate the action [they need]."
These differences were brought to light during the conference as various audience members representing the Centers for Disease Control and Prevention, the Food and Drug Administration, the CMS, the National Institutes of Health, and others involved in research and policy making, addressed the panel to either explain or defend why their agency operates as it does.
In the case of the CMS, a statutory organization, it can apply coverage only according to what the agency is mandated to do, said Dr. Elizabeth Koller of the CMS. The level of evidence the agency looks for, she said, includes "hard endpoints of clinical relevance, like reductions in sleep apnea and degenerative joint disease." The CMS is also concerned about the lack of long-term data on interventions, the durability of interventions, and which characteristics are common in people who relapse in their disease, said Dr. Koller, who addressed the group as an audience member.
"Hearing from the CMS was incredibly helpful. We learned so much," said Dr. Mechanick, director of metabolic support at the Mt. Sinai School of Medicine in New York, in an interview.
Dr. Mechanick also said this was the first of three meetings, the next to be held in about a year, where the ultimate goal would be to use the evidence base they will have created to develop recommendations for all involved in delivering obesity care.
The talk was "polite," Dr. John Morton, chief of bariatric surgery at Stanford (Calif.) University and president of the American Society of Bariatric and Metabolic Surgery, said in an interview, but he said he thinks there is bias against people with obesity. "We wouldn’t be having this discussion if it were about cancer," he said in the interview. "Sometimes we think the consequences of obesity are the result of a personal decision, and that may skew people in a direction where they don’t necessarily want to provide help."
Regardless, Dr. Garvey said at the briefing, "the ‘old world’ thinking that obesity is a lifestyle choice has failed us."
Dr. Mechanick is a consultant for Abbott Nutrition. Dr. Garvey has multiple industry relationships, including with Merck, Vivus, and Eisai.
WASHINGTON – Obesity requires a medical definition that goes beyond gauging a person’s body mass index if cost-effective care is to be delivered in an integrated fashion, according to a consensus statement issued by the American Association of Clinical Endocrinologists and the American College of Endocrinology.
"The definition of obesity as a disease is not perfect," Dr. W. Timothy Garvey, who chaired the AACE/ACE Obesity Consensus Conference, said in a media briefing. "We rely upon an [anthropometric] measure of body mass index, which is a measure of height versus weight, and there was consensus that this was ... divorced from the impact of weight gain on the health of the individual. This imprecision in our diagnosis of obesity was constraining us."
In 2013, the American Medical Association officially recognized obesity as a disease. Better codification of what actually constitutes "obesity, the disease," will allow a more integrated and effective approach to treating it, said Dr. Garvey, professor of medicine and chair of the department of nutrition sciences at the University of Alabama at Birmingham. To do so, the AACE/ACE held an intensive, 2-day session that largely featured spontaneous discussions between panelists and audience members representing four specific obesity "pillars": biomedical, government and regulation, health industry and economics, and research and education sectors.
A constant theme across the sectors was the need for a definition of obesity that accounts for cultural differences, ethnicity, and the presence or absence of cardiometabolic markers of disease in persons who are overweight or obese.
The conference’s multidisciplined approach informed the consensus statement that obesity is a chronic disease that should be treated with the established AACE/ACE obesity algorithm and met with lifestyle interventions. The consensus statement also addressed our current "obesogenic" environment, which many participants said was created in part by the abundance of nonnutritious foods.
In an interview, Dr. Susan Kansagra, deputy commissioner of the New York City Department of Health and Mental Hygiene, said that by working with local vendors and their suppliers, among other actions, her agency is focused on increasing access to more nutritious foods in neighborhoods across the city as a way to shape the food environment. "It’s not people who’ve changed over the past 30 years; it’s the environment," Dr. Kansagra said at the conference.
Also addressed by the consensus statement was the need for preventive care, particularly at the pediatric level, and more cohesive public awareness campaigns that could affect how private payers develop their reimbursement strategies. Audience member Dr. Robert Silverman, medical director of CIGNA Healthcare, said that payers would respond to the need for obesity care, but that what currently is missing is "a tie between the evidence and the complications [of obesity]."
"We learned that different stakeholders require different levels of evidence," AACE President Jeffrey I. Mechanick said in the media briefing. "So, we’re going to be able come up with a more efficient way to make recommendations about research so that private insurance carriers, the Centers for Medicare & Medicaid Services, or regulatory agencies have the type of data they require to facilitate the action [they need]."
These differences were brought to light during the conference as various audience members representing the Centers for Disease Control and Prevention, the Food and Drug Administration, the CMS, the National Institutes of Health, and others involved in research and policy making, addressed the panel to either explain or defend why their agency operates as it does.
In the case of the CMS, a statutory organization, it can apply coverage only according to what the agency is mandated to do, said Dr. Elizabeth Koller of the CMS. The level of evidence the agency looks for, she said, includes "hard endpoints of clinical relevance, like reductions in sleep apnea and degenerative joint disease." The CMS is also concerned about the lack of long-term data on interventions, the durability of interventions, and which characteristics are common in people who relapse in their disease, said Dr. Koller, who addressed the group as an audience member.
"Hearing from the CMS was incredibly helpful. We learned so much," said Dr. Mechanick, director of metabolic support at the Mt. Sinai School of Medicine in New York, in an interview.
Dr. Mechanick also said this was the first of three meetings, the next to be held in about a year, where the ultimate goal would be to use the evidence base they will have created to develop recommendations for all involved in delivering obesity care.
The talk was "polite," Dr. John Morton, chief of bariatric surgery at Stanford (Calif.) University and president of the American Society of Bariatric and Metabolic Surgery, said in an interview, but he said he thinks there is bias against people with obesity. "We wouldn’t be having this discussion if it were about cancer," he said in the interview. "Sometimes we think the consequences of obesity are the result of a personal decision, and that may skew people in a direction where they don’t necessarily want to provide help."
Regardless, Dr. Garvey said at the briefing, "the ‘old world’ thinking that obesity is a lifestyle choice has failed us."
Dr. Mechanick is a consultant for Abbott Nutrition. Dr. Garvey has multiple industry relationships, including with Merck, Vivus, and Eisai.
AT THE AACE/ACE CONSENSUS CONFERENCE ON OBESITY
VIDEO: Getting payers to cover obesity treatment
WASHINGTON – Payers will cover obesity treatment, people will suffer less from cardiometabolic disease, and the economy will be less burdened by the costs of untreated complications and lost productivity related to obesity. That’s the vision of Dr. Jeffrey I. Mechanick and his colleagues at the American Association of Clinical Endocrinologists and the American College of Endocrinology, who’ve jointly released a consensus statement on obesity treatment.
In a video interview, AACE president Dr. Mechanick, discusses why payers haven’t covered obesity treatment as a matter of course in the past, but why they may going forward, and how a growing impatience with a fractured approach to obesity care spurred the AACE and ACE to create a cross-disciplinary conference that also included members of the nonmedical professions involved in obesity care.
"By limiting the conference just to the biomedical model, we wouldn’t have access to this type of information," Dr. Mechanick also of the Icahn School of Medicine at Mount Sinai, New York, said in a press conference at the meeting. "We learned that different stakeholders require different levels of evidence."
The value of measuring body mass index is a controversial topic in obesity treatment. Dr. Mechanick explains why BMI will still be part of the developing ACCE/ACE standards of care and how it will be incorporated with other factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Payers will cover obesity treatment, people will suffer less from cardiometabolic disease, and the economy will be less burdened by the costs of untreated complications and lost productivity related to obesity. That’s the vision of Dr. Jeffrey I. Mechanick and his colleagues at the American Association of Clinical Endocrinologists and the American College of Endocrinology, who’ve jointly released a consensus statement on obesity treatment.
In a video interview, AACE president Dr. Mechanick, discusses why payers haven’t covered obesity treatment as a matter of course in the past, but why they may going forward, and how a growing impatience with a fractured approach to obesity care spurred the AACE and ACE to create a cross-disciplinary conference that also included members of the nonmedical professions involved in obesity care.
"By limiting the conference just to the biomedical model, we wouldn’t have access to this type of information," Dr. Mechanick also of the Icahn School of Medicine at Mount Sinai, New York, said in a press conference at the meeting. "We learned that different stakeholders require different levels of evidence."
The value of measuring body mass index is a controversial topic in obesity treatment. Dr. Mechanick explains why BMI will still be part of the developing ACCE/ACE standards of care and how it will be incorporated with other factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Payers will cover obesity treatment, people will suffer less from cardiometabolic disease, and the economy will be less burdened by the costs of untreated complications and lost productivity related to obesity. That’s the vision of Dr. Jeffrey I. Mechanick and his colleagues at the American Association of Clinical Endocrinologists and the American College of Endocrinology, who’ve jointly released a consensus statement on obesity treatment.
In a video interview, AACE president Dr. Mechanick, discusses why payers haven’t covered obesity treatment as a matter of course in the past, but why they may going forward, and how a growing impatience with a fractured approach to obesity care spurred the AACE and ACE to create a cross-disciplinary conference that also included members of the nonmedical professions involved in obesity care.
"By limiting the conference just to the biomedical model, we wouldn’t have access to this type of information," Dr. Mechanick also of the Icahn School of Medicine at Mount Sinai, New York, said in a press conference at the meeting. "We learned that different stakeholders require different levels of evidence."
The value of measuring body mass index is a controversial topic in obesity treatment. Dr. Mechanick explains why BMI will still be part of the developing ACCE/ACE standards of care and how it will be incorporated with other factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE AACE/ACE CONSENSUS CONFERENCE ON OBESITY
Is Bariatric Surgery the Right Treatment Model for Obesity?
WASHINGTON – Bariatric surgery has the greatest amount of data demonstrating its efficacy in reducing the comorbidities of obesity, as well as providing a long-term solution to the disease itself, according to Dr. John Morton, chief of bariatric surgery at Stanford (Calif.) University. Is it the model for obesity care that the nation needs to address the rising epidemic? If so, what stands in the way of its implementation? Dr. Morton offers a range of opinions in this audio interview.
WASHINGTON – Bariatric surgery has the greatest amount of data demonstrating its efficacy in reducing the comorbidities of obesity, as well as providing a long-term solution to the disease itself, according to Dr. John Morton, chief of bariatric surgery at Stanford (Calif.) University. Is it the model for obesity care that the nation needs to address the rising epidemic? If so, what stands in the way of its implementation? Dr. Morton offers a range of opinions in this audio interview.
WASHINGTON – Bariatric surgery has the greatest amount of data demonstrating its efficacy in reducing the comorbidities of obesity, as well as providing a long-term solution to the disease itself, according to Dr. John Morton, chief of bariatric surgery at Stanford (Calif.) University. Is it the model for obesity care that the nation needs to address the rising epidemic? If so, what stands in the way of its implementation? Dr. Morton offers a range of opinions in this audio interview.
EXPERT ANALYSIS FROM THE AACE/ACE CONSENSUS CONFERENCE ON OBESITY
Choice vs. regulation: the role of public policy in obesity management
WASHINGTON – It’s not people who’ve changed over the last 3 decades, but the environment around them that has brought on the obesity crisis, according to the deputy commissioner of the New York City Department of Health and Mental Hygiene, Dr. Susan M. Kansagra.
"The environment promotes people to consume more calories," Dr. Kansagra said in an audio interview during the American Association of Clinical Endocrinologists and American College of Endocrinology’s joint consensus conference on obesity. In this interview about the multifactorial nature of the disease, Dr. Kansagra discusses what role she sees as the role of public officials in shaping that environment.
WASHINGTON – It’s not people who’ve changed over the last 3 decades, but the environment around them that has brought on the obesity crisis, according to the deputy commissioner of the New York City Department of Health and Mental Hygiene, Dr. Susan M. Kansagra.
"The environment promotes people to consume more calories," Dr. Kansagra said in an audio interview during the American Association of Clinical Endocrinologists and American College of Endocrinology’s joint consensus conference on obesity. In this interview about the multifactorial nature of the disease, Dr. Kansagra discusses what role she sees as the role of public officials in shaping that environment.
WASHINGTON – It’s not people who’ve changed over the last 3 decades, but the environment around them that has brought on the obesity crisis, according to the deputy commissioner of the New York City Department of Health and Mental Hygiene, Dr. Susan M. Kansagra.
"The environment promotes people to consume more calories," Dr. Kansagra said in an audio interview during the American Association of Clinical Endocrinologists and American College of Endocrinology’s joint consensus conference on obesity. In this interview about the multifactorial nature of the disease, Dr. Kansagra discusses what role she sees as the role of public officials in shaping that environment.
EXPERT ANALYSIS FROM THE AACE/ACE CONSENSUS CONFERENCE ON OBESITY