User login
Bioprinted organs – a made-to-order future
SAN FRANCISCO – Gastroenterology might be the proving ground of one of medicine’s boldest new ventures – the creation of fully functional human organs built with a 3-D printer.
Gut is a perfect beginning project, Dr. John Geibel said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology. It has a very simple shape – just a long hollow tube. Epithelial cells grow and turn over very quickly, suggesting that a length of artificial intestine could be grown relatively quickly. And although intestine is composed of a number of distinct layers, a 3-D bioprinter would have no trouble laying down concentric circles of each one to recreate their natural morphology.
“It will take time. It will take planning. But this is going to happen,” said Dr. Geibel of Yale University, New Haven, Conn.
To create a length of intestine, the print heads of a bioprinter would be loaded with cells from all of the gut layers – the serosa, the different muscle strata, the mucosa. Each would be laid down in its respective anatomic ring, supported all around by a hydrogel. The print sequence would be repeated over and over until the required length of intestine was created. From then, Dr. Geibel said, it would be only a matter of days before the cells knit themselves together so well that the gel could be dissolved and the new tissue ready for transplant.
Liver would likely be the next organ up for printing, with the ultimate goal of creating fully transplantable organs. The need is enormous, and can’t be overstated. Patients who need a new liver wait an average of 4 years before they receive one. Even then, there is no guarantee that the selected organ will function as it should. Rejection is always a concern. Immunosuppression is a lifelong challenge.
The liver is much more complicated than a length of gut. It is cellularly complex and highly vascularized. But liver-printing is already a reality. Bioprinted “3-D liver-in-a-dish,” created by San Diego–based Organovo, has function, if not form. The cells work together; they grow, divide, and secrete bile acids. However, they exist as a formless, nonvascular blob.
As it stands (or rather, lies) now, bioprinted liver is a perfect preclinical model – perfectly replicating how the liver would respond to drugs without any of the messy adverse events that hurt patients. But it needs some backbone, or more accurately, some matrix, in order to morph again and grow into a complete organ. A liver-shaped collagen matrix could provide the necessary frame for cells to grow in and around; tunnels through it would form pathways for a similarly engineered vasculature.
The logical leap between dish and transplant is likely to be some sort of nonformed “piggyback liver,” which could be used as a bridge to keep liver failure patients alive until a traditional donor organ is found, Dr. Geibel said.
The Holy Grail, of course, would be a transplantable organ, created from the patient’s own cells – a made-to-order liver that’s 100% compatible with the recipient, who is actually also the donor.
“Think of the difference in quality of life these could make for patients who need transplants. The possibility of living without the fear of organ rejection, without the need for lifelong immunosuppression. Without the fear of having no alternative if the organ didn’t function well, or failed. If it did, we could simply grow another and try again.”
SAN FRANCISCO – Gastroenterology might be the proving ground of one of medicine’s boldest new ventures – the creation of fully functional human organs built with a 3-D printer.
Gut is a perfect beginning project, Dr. John Geibel said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology. It has a very simple shape – just a long hollow tube. Epithelial cells grow and turn over very quickly, suggesting that a length of artificial intestine could be grown relatively quickly. And although intestine is composed of a number of distinct layers, a 3-D bioprinter would have no trouble laying down concentric circles of each one to recreate their natural morphology.
“It will take time. It will take planning. But this is going to happen,” said Dr. Geibel of Yale University, New Haven, Conn.
To create a length of intestine, the print heads of a bioprinter would be loaded with cells from all of the gut layers – the serosa, the different muscle strata, the mucosa. Each would be laid down in its respective anatomic ring, supported all around by a hydrogel. The print sequence would be repeated over and over until the required length of intestine was created. From then, Dr. Geibel said, it would be only a matter of days before the cells knit themselves together so well that the gel could be dissolved and the new tissue ready for transplant.
Liver would likely be the next organ up for printing, with the ultimate goal of creating fully transplantable organs. The need is enormous, and can’t be overstated. Patients who need a new liver wait an average of 4 years before they receive one. Even then, there is no guarantee that the selected organ will function as it should. Rejection is always a concern. Immunosuppression is a lifelong challenge.
The liver is much more complicated than a length of gut. It is cellularly complex and highly vascularized. But liver-printing is already a reality. Bioprinted “3-D liver-in-a-dish,” created by San Diego–based Organovo, has function, if not form. The cells work together; they grow, divide, and secrete bile acids. However, they exist as a formless, nonvascular blob.
As it stands (or rather, lies) now, bioprinted liver is a perfect preclinical model – perfectly replicating how the liver would respond to drugs without any of the messy adverse events that hurt patients. But it needs some backbone, or more accurately, some matrix, in order to morph again and grow into a complete organ. A liver-shaped collagen matrix could provide the necessary frame for cells to grow in and around; tunnels through it would form pathways for a similarly engineered vasculature.
The logical leap between dish and transplant is likely to be some sort of nonformed “piggyback liver,” which could be used as a bridge to keep liver failure patients alive until a traditional donor organ is found, Dr. Geibel said.
The Holy Grail, of course, would be a transplantable organ, created from the patient’s own cells – a made-to-order liver that’s 100% compatible with the recipient, who is actually also the donor.
“Think of the difference in quality of life these could make for patients who need transplants. The possibility of living without the fear of organ rejection, without the need for lifelong immunosuppression. Without the fear of having no alternative if the organ didn’t function well, or failed. If it did, we could simply grow another and try again.”
SAN FRANCISCO – Gastroenterology might be the proving ground of one of medicine’s boldest new ventures – the creation of fully functional human organs built with a 3-D printer.
Gut is a perfect beginning project, Dr. John Geibel said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology. It has a very simple shape – just a long hollow tube. Epithelial cells grow and turn over very quickly, suggesting that a length of artificial intestine could be grown relatively quickly. And although intestine is composed of a number of distinct layers, a 3-D bioprinter would have no trouble laying down concentric circles of each one to recreate their natural morphology.
“It will take time. It will take planning. But this is going to happen,” said Dr. Geibel of Yale University, New Haven, Conn.
To create a length of intestine, the print heads of a bioprinter would be loaded with cells from all of the gut layers – the serosa, the different muscle strata, the mucosa. Each would be laid down in its respective anatomic ring, supported all around by a hydrogel. The print sequence would be repeated over and over until the required length of intestine was created. From then, Dr. Geibel said, it would be only a matter of days before the cells knit themselves together so well that the gel could be dissolved and the new tissue ready for transplant.
Liver would likely be the next organ up for printing, with the ultimate goal of creating fully transplantable organs. The need is enormous, and can’t be overstated. Patients who need a new liver wait an average of 4 years before they receive one. Even then, there is no guarantee that the selected organ will function as it should. Rejection is always a concern. Immunosuppression is a lifelong challenge.
The liver is much more complicated than a length of gut. It is cellularly complex and highly vascularized. But liver-printing is already a reality. Bioprinted “3-D liver-in-a-dish,” created by San Diego–based Organovo, has function, if not form. The cells work together; they grow, divide, and secrete bile acids. However, they exist as a formless, nonvascular blob.
As it stands (or rather, lies) now, bioprinted liver is a perfect preclinical model – perfectly replicating how the liver would respond to drugs without any of the messy adverse events that hurt patients. But it needs some backbone, or more accurately, some matrix, in order to morph again and grow into a complete organ. A liver-shaped collagen matrix could provide the necessary frame for cells to grow in and around; tunnels through it would form pathways for a similarly engineered vasculature.
The logical leap between dish and transplant is likely to be some sort of nonformed “piggyback liver,” which could be used as a bridge to keep liver failure patients alive until a traditional donor organ is found, Dr. Geibel said.
The Holy Grail, of course, would be a transplantable organ, created from the patient’s own cells – a made-to-order liver that’s 100% compatible with the recipient, who is actually also the donor.
“Think of the difference in quality of life these could make for patients who need transplants. The possibility of living without the fear of organ rejection, without the need for lifelong immunosuppression. Without the fear of having no alternative if the organ didn’t function well, or failed. If it did, we could simply grow another and try again.”
AT THE 2015 AGA TECH SUMMIT
2014 – For GI tech interventions, it was a very good year
SAN FRANCISCO– The past 18 months may well have been a watershed moment for medical technology in the gastroenterology field, with bullish venture investment in GI med/biotech, Dr. Jay Pasricha said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Both small and large companies did well, raising millions in private capital, said Dr. Pasricha of Johns Hopkins University, Baltimore. Several devices moved forward with Food and Drug Administration approval as well.
“I think we have crossed the Rubicon,” he said. “It’s really exciting after years of watching this momentum build. I think GI technology is really going to take off finally, and there is the promise of much more to come.”
The SmartPill food chain perfectly illustrates Dr. Pasricha’s optimistic view. In 2013, Covidien purchased Israel-based Given Imaging for $860 million, acquiring all seven of the company’s product lines, including PillCam, a minimally invasive, nonsedation, optical endoscopy technology that is swallowed to visualize the small bowel, esophagus, and colon; the ManoScan high-resolution manometry technology; Bravo capsule-based pH monitoring; and SmartPill motility monitoring systems.
It didn’t take long for an even bigger fish to join the frenzy. Just this January, Medtronic purchased Covidien for about $50 billion – the largest purchase in medical device history. “The question now, however, is ‘How will it impact us in the GI field in general?’” Dr. Pasricha said.
He outlined three possible scenarios.
“The first, which I fervently hope will happen, is that we finally realize a validation of this space” of GI technologic interventions. “This really does look like the event that will put this field on the map.”
He expressed some hope that the Medtronic purchase will alert big-name surgical device companies to the possibilities inherent in minimally invasive GI interventions. “I hope it will attract key surgical players to the field ,,, shake things up and lead to more research and development, and a rapid advance of technology.”
It took an event like this to set the field’s stars in motion. But dawn hasn’t broken yet, he cautioned.
“It’s also possible that Medtronic will look at the field and decide it really doesn’t like everything in GI, and that it will focus on just a couple of fields, like neuromodulation, at the cost of others.”
The third scenario is the gloomiest, Dr. Pasricha said – although he said it’s probably the least likely.
“It is possible that GI will just turn out to be a speck of dust in Medtronic’s big picture. We’ll just have to wait and see what happens in the next couple of years.”
Overall, 2014 was series of good-news stories for GI med/biotech. The year saw an overall 61% increase in venture capital investments in the arena – nearly $50 billion among 4,356 deals. Nearly $9 billion of this went directly to life science investments, in a 29% jump over 2013. Almost $3 billion was directed to medical device investments – up 27%. Devices in both early- and late-stage development felt the financial love (according to a recent PricewaterhouseCoopers report).
Less invasive GERD interventions fared particularly well last year. A landmark paper demonstrated that Endogastric Solutions’ (San Mateo, Calif.) transoral incisionless fundoplication significantly outperformed medical therapy with proton pump inhibition. EndoGastric Solutions is currently working with the AGA Center for GI Innovation and Technology to collect real-world data evaluating safety, efficacy, and comparative outcomes for this procedure compared with laparoscopic surgery, through the STAR Registry.
EndoStim (Amsterdam) is running a hot race as well – ready to launch a pivotal trial sometime this year. Its implantable device stimulates the lower esophageal sphincter to control reflux. The device is not yet approved for reimbursement in any country. But on March 11, the German Institute for the Hospital Remuneration System granted it a status 1 level, which entitles hospitals to negotiate with payers for at least partial coverage.
All of this activity bodes well for the future, Dr. Pasricha said.
“We have seen GERD procedures come and go now for 15 years. Now the data are really starting to support endoscopic treatment here. I think we are past the inflection point and will see this approach really take over the field.”
The American Medical Association is keeping abreast of all the progress. On March 10, the AMA created a CPT code for transoral incisionless fundoplication. “This will really legitimize this entire field. In 5 years, these techniques may become serious contenders for first-line therapy instead of chronic PPI use. And third-generation therapies will likely emerge to tackle the more difficult cases that are now excluded, like patients with a large hiatal hernia. There’s no question but that transoral is going to be the preferred way of treating GERD.”
Endoscopy wasn’t the only subspecialty to bask in this new sunny clime. In January, the FDA approved St. Paul–based EnteroMedics’ Maestro system – a neuromodulatory approach to weight loss for obese adults. Maestro is an implanted vagus nerve stimulator that modulates neural signaling, controlling gastric emptying. It also seems to affect hunger signaling, Dr. Pasricha said.
Despite Maestro’s unique method of action – and the fact that it’s the first new weight-loss device approved since 2007 – EnteroMedics’ bottom line hasn’t surged, yet. But it may be too early to look for the impact and in general the outlook for nonsurgical approaches to obesity remains bullish.
Dr. Pasricha reminded the audience about some very basic core principles for successful innovation. “To succeed, you have to have two of three key things. You have to fill an unmet need. The intervention should be simple. And it should be effective.”
Since most GERD patients do fine on medical therapy, those interventions won’t succeed based on need alone. But endoscopic GERD treatments are simple and quite effective. Obesity, on the other hand, presents an enormous unmet need. But the current interventions – including Maestro – are far from simple. And efficacy hasn’t been completely confirmed yet, Dr. Pasricha said.
A couple of other obesity devices are in the works. Apollo Endosurgery (Austin) is focusing on the ORBERA single intragastric balloon. ReShape Medical (San Clemente, Calif.) reported positive data from its pivotal phase III trial of its own intragastric balloon. The device consists of two linked balloons, which the company maintains fit the stomach contours better than does a single balloon.
Over 6 months, patients treated with the dual balloon experienced a mean total body weight loss of 16% (about 17 kg). It seemed to impart some lasting benefit, as those who had continued nutritional counseling after removal maintained about 94% of their weight loss over 7 months of follow-up.
On the basis of these data, ReShape was able to secure close to $7 million in private funding to continue work on the device.
“The great potential for balloon therapy is to make it easier for patients to enter the area of bariatric surgery,” Dr. Pasricha said. “They are not naturally drawn into surgery – they usually have to be pushed into it. But with balloon treatment, they are able to enter, and the threshold for more interventions is then much lower.”
SAN FRANCISCO– The past 18 months may well have been a watershed moment for medical technology in the gastroenterology field, with bullish venture investment in GI med/biotech, Dr. Jay Pasricha said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Both small and large companies did well, raising millions in private capital, said Dr. Pasricha of Johns Hopkins University, Baltimore. Several devices moved forward with Food and Drug Administration approval as well.
“I think we have crossed the Rubicon,” he said. “It’s really exciting after years of watching this momentum build. I think GI technology is really going to take off finally, and there is the promise of much more to come.”
The SmartPill food chain perfectly illustrates Dr. Pasricha’s optimistic view. In 2013, Covidien purchased Israel-based Given Imaging for $860 million, acquiring all seven of the company’s product lines, including PillCam, a minimally invasive, nonsedation, optical endoscopy technology that is swallowed to visualize the small bowel, esophagus, and colon; the ManoScan high-resolution manometry technology; Bravo capsule-based pH monitoring; and SmartPill motility monitoring systems.
It didn’t take long for an even bigger fish to join the frenzy. Just this January, Medtronic purchased Covidien for about $50 billion – the largest purchase in medical device history. “The question now, however, is ‘How will it impact us in the GI field in general?’” Dr. Pasricha said.
He outlined three possible scenarios.
“The first, which I fervently hope will happen, is that we finally realize a validation of this space” of GI technologic interventions. “This really does look like the event that will put this field on the map.”
He expressed some hope that the Medtronic purchase will alert big-name surgical device companies to the possibilities inherent in minimally invasive GI interventions. “I hope it will attract key surgical players to the field ,,, shake things up and lead to more research and development, and a rapid advance of technology.”
It took an event like this to set the field’s stars in motion. But dawn hasn’t broken yet, he cautioned.
“It’s also possible that Medtronic will look at the field and decide it really doesn’t like everything in GI, and that it will focus on just a couple of fields, like neuromodulation, at the cost of others.”
The third scenario is the gloomiest, Dr. Pasricha said – although he said it’s probably the least likely.
“It is possible that GI will just turn out to be a speck of dust in Medtronic’s big picture. We’ll just have to wait and see what happens in the next couple of years.”
Overall, 2014 was series of good-news stories for GI med/biotech. The year saw an overall 61% increase in venture capital investments in the arena – nearly $50 billion among 4,356 deals. Nearly $9 billion of this went directly to life science investments, in a 29% jump over 2013. Almost $3 billion was directed to medical device investments – up 27%. Devices in both early- and late-stage development felt the financial love (according to a recent PricewaterhouseCoopers report).
Less invasive GERD interventions fared particularly well last year. A landmark paper demonstrated that Endogastric Solutions’ (San Mateo, Calif.) transoral incisionless fundoplication significantly outperformed medical therapy with proton pump inhibition. EndoGastric Solutions is currently working with the AGA Center for GI Innovation and Technology to collect real-world data evaluating safety, efficacy, and comparative outcomes for this procedure compared with laparoscopic surgery, through the STAR Registry.
EndoStim (Amsterdam) is running a hot race as well – ready to launch a pivotal trial sometime this year. Its implantable device stimulates the lower esophageal sphincter to control reflux. The device is not yet approved for reimbursement in any country. But on March 11, the German Institute for the Hospital Remuneration System granted it a status 1 level, which entitles hospitals to negotiate with payers for at least partial coverage.
All of this activity bodes well for the future, Dr. Pasricha said.
“We have seen GERD procedures come and go now for 15 years. Now the data are really starting to support endoscopic treatment here. I think we are past the inflection point and will see this approach really take over the field.”
The American Medical Association is keeping abreast of all the progress. On March 10, the AMA created a CPT code for transoral incisionless fundoplication. “This will really legitimize this entire field. In 5 years, these techniques may become serious contenders for first-line therapy instead of chronic PPI use. And third-generation therapies will likely emerge to tackle the more difficult cases that are now excluded, like patients with a large hiatal hernia. There’s no question but that transoral is going to be the preferred way of treating GERD.”
Endoscopy wasn’t the only subspecialty to bask in this new sunny clime. In January, the FDA approved St. Paul–based EnteroMedics’ Maestro system – a neuromodulatory approach to weight loss for obese adults. Maestro is an implanted vagus nerve stimulator that modulates neural signaling, controlling gastric emptying. It also seems to affect hunger signaling, Dr. Pasricha said.
Despite Maestro’s unique method of action – and the fact that it’s the first new weight-loss device approved since 2007 – EnteroMedics’ bottom line hasn’t surged, yet. But it may be too early to look for the impact and in general the outlook for nonsurgical approaches to obesity remains bullish.
Dr. Pasricha reminded the audience about some very basic core principles for successful innovation. “To succeed, you have to have two of three key things. You have to fill an unmet need. The intervention should be simple. And it should be effective.”
Since most GERD patients do fine on medical therapy, those interventions won’t succeed based on need alone. But endoscopic GERD treatments are simple and quite effective. Obesity, on the other hand, presents an enormous unmet need. But the current interventions – including Maestro – are far from simple. And efficacy hasn’t been completely confirmed yet, Dr. Pasricha said.
A couple of other obesity devices are in the works. Apollo Endosurgery (Austin) is focusing on the ORBERA single intragastric balloon. ReShape Medical (San Clemente, Calif.) reported positive data from its pivotal phase III trial of its own intragastric balloon. The device consists of two linked balloons, which the company maintains fit the stomach contours better than does a single balloon.
Over 6 months, patients treated with the dual balloon experienced a mean total body weight loss of 16% (about 17 kg). It seemed to impart some lasting benefit, as those who had continued nutritional counseling after removal maintained about 94% of their weight loss over 7 months of follow-up.
On the basis of these data, ReShape was able to secure close to $7 million in private funding to continue work on the device.
“The great potential for balloon therapy is to make it easier for patients to enter the area of bariatric surgery,” Dr. Pasricha said. “They are not naturally drawn into surgery – they usually have to be pushed into it. But with balloon treatment, they are able to enter, and the threshold for more interventions is then much lower.”
SAN FRANCISCO– The past 18 months may well have been a watershed moment for medical technology in the gastroenterology field, with bullish venture investment in GI med/biotech, Dr. Jay Pasricha said at the 2015 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Both small and large companies did well, raising millions in private capital, said Dr. Pasricha of Johns Hopkins University, Baltimore. Several devices moved forward with Food and Drug Administration approval as well.
“I think we have crossed the Rubicon,” he said. “It’s really exciting after years of watching this momentum build. I think GI technology is really going to take off finally, and there is the promise of much more to come.”
The SmartPill food chain perfectly illustrates Dr. Pasricha’s optimistic view. In 2013, Covidien purchased Israel-based Given Imaging for $860 million, acquiring all seven of the company’s product lines, including PillCam, a minimally invasive, nonsedation, optical endoscopy technology that is swallowed to visualize the small bowel, esophagus, and colon; the ManoScan high-resolution manometry technology; Bravo capsule-based pH monitoring; and SmartPill motility monitoring systems.
It didn’t take long for an even bigger fish to join the frenzy. Just this January, Medtronic purchased Covidien for about $50 billion – the largest purchase in medical device history. “The question now, however, is ‘How will it impact us in the GI field in general?’” Dr. Pasricha said.
He outlined three possible scenarios.
“The first, which I fervently hope will happen, is that we finally realize a validation of this space” of GI technologic interventions. “This really does look like the event that will put this field on the map.”
He expressed some hope that the Medtronic purchase will alert big-name surgical device companies to the possibilities inherent in minimally invasive GI interventions. “I hope it will attract key surgical players to the field ,,, shake things up and lead to more research and development, and a rapid advance of technology.”
It took an event like this to set the field’s stars in motion. But dawn hasn’t broken yet, he cautioned.
“It’s also possible that Medtronic will look at the field and decide it really doesn’t like everything in GI, and that it will focus on just a couple of fields, like neuromodulation, at the cost of others.”
The third scenario is the gloomiest, Dr. Pasricha said – although he said it’s probably the least likely.
“It is possible that GI will just turn out to be a speck of dust in Medtronic’s big picture. We’ll just have to wait and see what happens in the next couple of years.”
Overall, 2014 was series of good-news stories for GI med/biotech. The year saw an overall 61% increase in venture capital investments in the arena – nearly $50 billion among 4,356 deals. Nearly $9 billion of this went directly to life science investments, in a 29% jump over 2013. Almost $3 billion was directed to medical device investments – up 27%. Devices in both early- and late-stage development felt the financial love (according to a recent PricewaterhouseCoopers report).
Less invasive GERD interventions fared particularly well last year. A landmark paper demonstrated that Endogastric Solutions’ (San Mateo, Calif.) transoral incisionless fundoplication significantly outperformed medical therapy with proton pump inhibition. EndoGastric Solutions is currently working with the AGA Center for GI Innovation and Technology to collect real-world data evaluating safety, efficacy, and comparative outcomes for this procedure compared with laparoscopic surgery, through the STAR Registry.
EndoStim (Amsterdam) is running a hot race as well – ready to launch a pivotal trial sometime this year. Its implantable device stimulates the lower esophageal sphincter to control reflux. The device is not yet approved for reimbursement in any country. But on March 11, the German Institute for the Hospital Remuneration System granted it a status 1 level, which entitles hospitals to negotiate with payers for at least partial coverage.
All of this activity bodes well for the future, Dr. Pasricha said.
“We have seen GERD procedures come and go now for 15 years. Now the data are really starting to support endoscopic treatment here. I think we are past the inflection point and will see this approach really take over the field.”
The American Medical Association is keeping abreast of all the progress. On March 10, the AMA created a CPT code for transoral incisionless fundoplication. “This will really legitimize this entire field. In 5 years, these techniques may become serious contenders for first-line therapy instead of chronic PPI use. And third-generation therapies will likely emerge to tackle the more difficult cases that are now excluded, like patients with a large hiatal hernia. There’s no question but that transoral is going to be the preferred way of treating GERD.”
Endoscopy wasn’t the only subspecialty to bask in this new sunny clime. In January, the FDA approved St. Paul–based EnteroMedics’ Maestro system – a neuromodulatory approach to weight loss for obese adults. Maestro is an implanted vagus nerve stimulator that modulates neural signaling, controlling gastric emptying. It also seems to affect hunger signaling, Dr. Pasricha said.
Despite Maestro’s unique method of action – and the fact that it’s the first new weight-loss device approved since 2007 – EnteroMedics’ bottom line hasn’t surged, yet. But it may be too early to look for the impact and in general the outlook for nonsurgical approaches to obesity remains bullish.
Dr. Pasricha reminded the audience about some very basic core principles for successful innovation. “To succeed, you have to have two of three key things. You have to fill an unmet need. The intervention should be simple. And it should be effective.”
Since most GERD patients do fine on medical therapy, those interventions won’t succeed based on need alone. But endoscopic GERD treatments are simple and quite effective. Obesity, on the other hand, presents an enormous unmet need. But the current interventions – including Maestro – are far from simple. And efficacy hasn’t been completely confirmed yet, Dr. Pasricha said.
A couple of other obesity devices are in the works. Apollo Endosurgery (Austin) is focusing on the ORBERA single intragastric balloon. ReShape Medical (San Clemente, Calif.) reported positive data from its pivotal phase III trial of its own intragastric balloon. The device consists of two linked balloons, which the company maintains fit the stomach contours better than does a single balloon.
Over 6 months, patients treated with the dual balloon experienced a mean total body weight loss of 16% (about 17 kg). It seemed to impart some lasting benefit, as those who had continued nutritional counseling after removal maintained about 94% of their weight loss over 7 months of follow-up.
On the basis of these data, ReShape was able to secure close to $7 million in private funding to continue work on the device.
“The great potential for balloon therapy is to make it easier for patients to enter the area of bariatric surgery,” Dr. Pasricha said. “They are not naturally drawn into surgery – they usually have to be pushed into it. But with balloon treatment, they are able to enter, and the threshold for more interventions is then much lower.”
Microbiome may drive the course of diabetes, obesity
Humans can host up to 6 pounds of microbes in our gut alone. We comprise about 10 trillion human cells – they comprise 100 trillion bacterial cells. Their genetic material outstrips us even more impressively – we are the product of 20,000 human genes. But inside our gut, we carry up to 2 million microbial genes.
Emerging research suggests that these complex communities – which interact fluidly not only with their human host, but also with each other – play important roles in health and disease. They appear to confer both protection from and risk for many chronic illnesses, from asthma and allergies to obesity and diabetes.
And what we don’t know about them dwarfs what we do know, according to Dr. Robert Ratner, the chief medical officer of the American Diabetes Association.
“We’re literally at the forefront of an unknown universe,” Dr. Ratner said at the annual advanced postgraduate course held by the ADA. “Research into the microbiome and how it interacts with human health is one of our most intriguing investigations.”
The microbiome is as vastly individual as every person who carries it. The differences are myriad, in the amount and variety of species, and in the sheer numbers of microbes that make up each community. Each region of the gut, from mouth to rectum, hosts a completely different population.
Two main phyla populate the gut: Bacteroidetes, which are involved in protein and carbohydrate breakdown, and Firmicutes, which promote the absorption of fat. The ratios of these communities, however, has changed over the last 30 years, diverging along a path that mirrors global spikes in obesity, diabetes, and allergic and inflammatory disorders.
“We have seen dramatic increases worldwide in these disorders,” Dr. Ratner said. “We’ve also seen decreased diversity of the microbiome, with a progressive change in density from Bacteroidetes to Firmicutes. These are associations – not causations – but I think the time has come to ask ‘What are we doing to change these bacteria?’ ”
The rampant use of antibiotics is the first place suspicion falls – and it’s no wonder, since the epidemiologic changes Dr. Ratner described began to appear shortly after World War II. Antibiotics could exert their flora-changing effects a couple of ways, he noted.
They directly alter the composition of communities in the person who consumes the drug, even if just in the short-term. There is someevidence that early-life antibiotics, though changing the microbiome only temporarily, can change fat metabolism for the entire lifespan (Cell 2014;158:705-21).
The associations between an altered gut microbiome and long-term health is unproven. But a picture does seem to emerge when viewed in light of the exponential increases in obesity and its attendant rise in diabetes.
The Type 1 Diabetes Prediction and Prevention Project (DIPP) is an effort to predict and search for means to delay or prevent type 1 diabetes. Launched in 1994, it’s following a cohort of children who had genetic risk factors for type 1 diabetes. Data have consistently shown that those who develop the disease have significantly lower diversity in their gut flora, Dr. Ratner said. Certain species of Bacteroides increased the risk of autoimmunity by up to 20 times.
Again, he said, this is association, not causation. But some very new evidence suggests that these are functional, not just observational, links. “It does now appear that our microbes are actually controlling our metabolism,” he said. “Some of these species liberate lipopolysaccharides, which function as endotoxins. These cross the mucosal barrier in the intestine, enter the interstitial space, and set up an inflammation that impacts the liver and adipocytes, potentially decreasing insulin sensitivity.”
Unpublished data from the laboratory of Dr. Martin J. Blaser at New York University show for the first time that a specific bacterium can cause diabetes, and removing it cures the disease. The bacterium in question, Ralstonia, is a gram-negative pathogen that contaminates drinking water. Mice engineered as a model of prediabetes gained much more weight when they consumed live Ralstonia than when they got a heat-inactivated version. They also developed insulin resistance and hyperglycemia in the presence of the live version. But when the same mice were given a Ralstonia antibody, they lost weight and their glycemic profile normalized.
“This is the first direct evidence we have of causality,” Dr. Ratner said.
Strong evidence of causation is also emerging in the surgical realm. Roux-en-Y gastric bypass seems to change the microbiome in a way that facilitates weight loss, beyond caloric intake.Randy Seeley, Ph.D., of the University of Michigan, Ann Arbor, has been studying how weight-loss surgery affects the microbiome. He theorizes that the physical manipulation of the gut induces what he calls “enteroplasticity” – a fluid adaptation of both the gut’s structure and its bacterial communities to the altered physical and chemical environment. “There’s more going on during postsurgical weight loss than just food restriction and malabsorption,” Dr. Seeley said in an interview. “It’s logical to think that when you do this kind of surgery, the microbes in the intestine will change.”
Roseburia intestinalis is a Firmicute that typically increases after bariatric surgery. It’s also found to be deficient in people who have diabetes. Dr. Seeley coauthored an article showing that Roseburia and other beneficial firmicutes increased significantly in mice that underwent vertical sleeve gastrectomy (Nature 2014;509:183-8). These mice lost weight after surgery, as would be expected, but then showed a preference for the high-protein and carbohydrate-rich foods that Firmicutes need. Their microbiome also showed decreases in the concentration of fat-loving Bacteroides species.
But interestingly, weight loss and microbiome improvement happened only in mice that had a normal bile acid–signaling system. Immediately after surgery, mice engineered to lack bile acid receptors did eat less and lose weight. But a week later, their appetites came back full force, and they actually seemed driven to eat fat. They quickly returned to their presurgical weight. Their microbiome didn’t show the same improvement as their cousins with normal signaling pathways, suggesting that a complex interaction between bacteria and the gut’s physical alteration may be driving weight loss.
“Is weight-loss surgery, then, fixing this ‘broken’ component of the microbiome?” Dr. Seeley asked. “Is Roseburia the causal agent of improvement? Or is it a marker of improvement in an entire community? I would say it’s probably the entire community changing, and changing its interaction with its host organism.”
Dr. Seeley disclosed that he has received financial support from Johnson & Johnson, Novo Nordisk, and Eisai.
On Twitter @alz_gal
Humans can host up to 6 pounds of microbes in our gut alone. We comprise about 10 trillion human cells – they comprise 100 trillion bacterial cells. Their genetic material outstrips us even more impressively – we are the product of 20,000 human genes. But inside our gut, we carry up to 2 million microbial genes.
Emerging research suggests that these complex communities – which interact fluidly not only with their human host, but also with each other – play important roles in health and disease. They appear to confer both protection from and risk for many chronic illnesses, from asthma and allergies to obesity and diabetes.
And what we don’t know about them dwarfs what we do know, according to Dr. Robert Ratner, the chief medical officer of the American Diabetes Association.
“We’re literally at the forefront of an unknown universe,” Dr. Ratner said at the annual advanced postgraduate course held by the ADA. “Research into the microbiome and how it interacts with human health is one of our most intriguing investigations.”
The microbiome is as vastly individual as every person who carries it. The differences are myriad, in the amount and variety of species, and in the sheer numbers of microbes that make up each community. Each region of the gut, from mouth to rectum, hosts a completely different population.
Two main phyla populate the gut: Bacteroidetes, which are involved in protein and carbohydrate breakdown, and Firmicutes, which promote the absorption of fat. The ratios of these communities, however, has changed over the last 30 years, diverging along a path that mirrors global spikes in obesity, diabetes, and allergic and inflammatory disorders.
“We have seen dramatic increases worldwide in these disorders,” Dr. Ratner said. “We’ve also seen decreased diversity of the microbiome, with a progressive change in density from Bacteroidetes to Firmicutes. These are associations – not causations – but I think the time has come to ask ‘What are we doing to change these bacteria?’ ”
The rampant use of antibiotics is the first place suspicion falls – and it’s no wonder, since the epidemiologic changes Dr. Ratner described began to appear shortly after World War II. Antibiotics could exert their flora-changing effects a couple of ways, he noted.
They directly alter the composition of communities in the person who consumes the drug, even if just in the short-term. There is someevidence that early-life antibiotics, though changing the microbiome only temporarily, can change fat metabolism for the entire lifespan (Cell 2014;158:705-21).
The associations between an altered gut microbiome and long-term health is unproven. But a picture does seem to emerge when viewed in light of the exponential increases in obesity and its attendant rise in diabetes.
The Type 1 Diabetes Prediction and Prevention Project (DIPP) is an effort to predict and search for means to delay or prevent type 1 diabetes. Launched in 1994, it’s following a cohort of children who had genetic risk factors for type 1 diabetes. Data have consistently shown that those who develop the disease have significantly lower diversity in their gut flora, Dr. Ratner said. Certain species of Bacteroides increased the risk of autoimmunity by up to 20 times.
Again, he said, this is association, not causation. But some very new evidence suggests that these are functional, not just observational, links. “It does now appear that our microbes are actually controlling our metabolism,” he said. “Some of these species liberate lipopolysaccharides, which function as endotoxins. These cross the mucosal barrier in the intestine, enter the interstitial space, and set up an inflammation that impacts the liver and adipocytes, potentially decreasing insulin sensitivity.”
Unpublished data from the laboratory of Dr. Martin J. Blaser at New York University show for the first time that a specific bacterium can cause diabetes, and removing it cures the disease. The bacterium in question, Ralstonia, is a gram-negative pathogen that contaminates drinking water. Mice engineered as a model of prediabetes gained much more weight when they consumed live Ralstonia than when they got a heat-inactivated version. They also developed insulin resistance and hyperglycemia in the presence of the live version. But when the same mice were given a Ralstonia antibody, they lost weight and their glycemic profile normalized.
“This is the first direct evidence we have of causality,” Dr. Ratner said.
Strong evidence of causation is also emerging in the surgical realm. Roux-en-Y gastric bypass seems to change the microbiome in a way that facilitates weight loss, beyond caloric intake.Randy Seeley, Ph.D., of the University of Michigan, Ann Arbor, has been studying how weight-loss surgery affects the microbiome. He theorizes that the physical manipulation of the gut induces what he calls “enteroplasticity” – a fluid adaptation of both the gut’s structure and its bacterial communities to the altered physical and chemical environment. “There’s more going on during postsurgical weight loss than just food restriction and malabsorption,” Dr. Seeley said in an interview. “It’s logical to think that when you do this kind of surgery, the microbes in the intestine will change.”
Roseburia intestinalis is a Firmicute that typically increases after bariatric surgery. It’s also found to be deficient in people who have diabetes. Dr. Seeley coauthored an article showing that Roseburia and other beneficial firmicutes increased significantly in mice that underwent vertical sleeve gastrectomy (Nature 2014;509:183-8). These mice lost weight after surgery, as would be expected, but then showed a preference for the high-protein and carbohydrate-rich foods that Firmicutes need. Their microbiome also showed decreases in the concentration of fat-loving Bacteroides species.
But interestingly, weight loss and microbiome improvement happened only in mice that had a normal bile acid–signaling system. Immediately after surgery, mice engineered to lack bile acid receptors did eat less and lose weight. But a week later, their appetites came back full force, and they actually seemed driven to eat fat. They quickly returned to their presurgical weight. Their microbiome didn’t show the same improvement as their cousins with normal signaling pathways, suggesting that a complex interaction between bacteria and the gut’s physical alteration may be driving weight loss.
“Is weight-loss surgery, then, fixing this ‘broken’ component of the microbiome?” Dr. Seeley asked. “Is Roseburia the causal agent of improvement? Or is it a marker of improvement in an entire community? I would say it’s probably the entire community changing, and changing its interaction with its host organism.”
Dr. Seeley disclosed that he has received financial support from Johnson & Johnson, Novo Nordisk, and Eisai.
On Twitter @alz_gal
Humans can host up to 6 pounds of microbes in our gut alone. We comprise about 10 trillion human cells – they comprise 100 trillion bacterial cells. Their genetic material outstrips us even more impressively – we are the product of 20,000 human genes. But inside our gut, we carry up to 2 million microbial genes.
Emerging research suggests that these complex communities – which interact fluidly not only with their human host, but also with each other – play important roles in health and disease. They appear to confer both protection from and risk for many chronic illnesses, from asthma and allergies to obesity and diabetes.
And what we don’t know about them dwarfs what we do know, according to Dr. Robert Ratner, the chief medical officer of the American Diabetes Association.
“We’re literally at the forefront of an unknown universe,” Dr. Ratner said at the annual advanced postgraduate course held by the ADA. “Research into the microbiome and how it interacts with human health is one of our most intriguing investigations.”
The microbiome is as vastly individual as every person who carries it. The differences are myriad, in the amount and variety of species, and in the sheer numbers of microbes that make up each community. Each region of the gut, from mouth to rectum, hosts a completely different population.
Two main phyla populate the gut: Bacteroidetes, which are involved in protein and carbohydrate breakdown, and Firmicutes, which promote the absorption of fat. The ratios of these communities, however, has changed over the last 30 years, diverging along a path that mirrors global spikes in obesity, diabetes, and allergic and inflammatory disorders.
“We have seen dramatic increases worldwide in these disorders,” Dr. Ratner said. “We’ve also seen decreased diversity of the microbiome, with a progressive change in density from Bacteroidetes to Firmicutes. These are associations – not causations – but I think the time has come to ask ‘What are we doing to change these bacteria?’ ”
The rampant use of antibiotics is the first place suspicion falls – and it’s no wonder, since the epidemiologic changes Dr. Ratner described began to appear shortly after World War II. Antibiotics could exert their flora-changing effects a couple of ways, he noted.
They directly alter the composition of communities in the person who consumes the drug, even if just in the short-term. There is someevidence that early-life antibiotics, though changing the microbiome only temporarily, can change fat metabolism for the entire lifespan (Cell 2014;158:705-21).
The associations between an altered gut microbiome and long-term health is unproven. But a picture does seem to emerge when viewed in light of the exponential increases in obesity and its attendant rise in diabetes.
The Type 1 Diabetes Prediction and Prevention Project (DIPP) is an effort to predict and search for means to delay or prevent type 1 diabetes. Launched in 1994, it’s following a cohort of children who had genetic risk factors for type 1 diabetes. Data have consistently shown that those who develop the disease have significantly lower diversity in their gut flora, Dr. Ratner said. Certain species of Bacteroides increased the risk of autoimmunity by up to 20 times.
Again, he said, this is association, not causation. But some very new evidence suggests that these are functional, not just observational, links. “It does now appear that our microbes are actually controlling our metabolism,” he said. “Some of these species liberate lipopolysaccharides, which function as endotoxins. These cross the mucosal barrier in the intestine, enter the interstitial space, and set up an inflammation that impacts the liver and adipocytes, potentially decreasing insulin sensitivity.”
Unpublished data from the laboratory of Dr. Martin J. Blaser at New York University show for the first time that a specific bacterium can cause diabetes, and removing it cures the disease. The bacterium in question, Ralstonia, is a gram-negative pathogen that contaminates drinking water. Mice engineered as a model of prediabetes gained much more weight when they consumed live Ralstonia than when they got a heat-inactivated version. They also developed insulin resistance and hyperglycemia in the presence of the live version. But when the same mice were given a Ralstonia antibody, they lost weight and their glycemic profile normalized.
“This is the first direct evidence we have of causality,” Dr. Ratner said.
Strong evidence of causation is also emerging in the surgical realm. Roux-en-Y gastric bypass seems to change the microbiome in a way that facilitates weight loss, beyond caloric intake.Randy Seeley, Ph.D., of the University of Michigan, Ann Arbor, has been studying how weight-loss surgery affects the microbiome. He theorizes that the physical manipulation of the gut induces what he calls “enteroplasticity” – a fluid adaptation of both the gut’s structure and its bacterial communities to the altered physical and chemical environment. “There’s more going on during postsurgical weight loss than just food restriction and malabsorption,” Dr. Seeley said in an interview. “It’s logical to think that when you do this kind of surgery, the microbes in the intestine will change.”
Roseburia intestinalis is a Firmicute that typically increases after bariatric surgery. It’s also found to be deficient in people who have diabetes. Dr. Seeley coauthored an article showing that Roseburia and other beneficial firmicutes increased significantly in mice that underwent vertical sleeve gastrectomy (Nature 2014;509:183-8). These mice lost weight after surgery, as would be expected, but then showed a preference for the high-protein and carbohydrate-rich foods that Firmicutes need. Their microbiome also showed decreases in the concentration of fat-loving Bacteroides species.
But interestingly, weight loss and microbiome improvement happened only in mice that had a normal bile acid–signaling system. Immediately after surgery, mice engineered to lack bile acid receptors did eat less and lose weight. But a week later, their appetites came back full force, and they actually seemed driven to eat fat. They quickly returned to their presurgical weight. Their microbiome didn’t show the same improvement as their cousins with normal signaling pathways, suggesting that a complex interaction between bacteria and the gut’s physical alteration may be driving weight loss.
“Is weight-loss surgery, then, fixing this ‘broken’ component of the microbiome?” Dr. Seeley asked. “Is Roseburia the causal agent of improvement? Or is it a marker of improvement in an entire community? I would say it’s probably the entire community changing, and changing its interaction with its host organism.”
Dr. Seeley disclosed that he has received financial support from Johnson & Johnson, Novo Nordisk, and Eisai.
On Twitter @alz_gal
EXPERT ANALYSIS FROM THE ADA ADVANCED POSTGRADUATE COURSE
Device closure of PFO doesn’t drop combined major adverse event risk
NASHVILLE, TENN. – Device closure of a patent foramen ovale didn’t reduce the composite risk of death, stroke, or transient ischemic attack when compared against medical therapy alone in a pooled analysis of three trials.
Device intervention, did, however, reduce the individual risk of recurrent stroke, although the absolute reduction seemed modest, Dr. David M. Kent said at the International Stroke Conference, sponsored by the American Heart Association.
Overall device closure conferred about a 30% annual risk reduction of recurrent stroke upon those with patent foramen ovale (PFO) who had experienced a prior cryptogenic stroke. About 30 patients would have to be treated to prevent one stroke over 5 years, said Dr. Kent of Tufts University, Boston.
“It’s not a huge benefit – it’s not like thrombectomy. But it’s comparable to the benefit of high-dose statins seen in the SPARCL [Stroke Prevention by Aggressive Reduction in Cholesterol Levels] trial.”
Dr. Kent and his colleagues’ analysis used three trials: the CLOSURE trial, which used the STARflex device, and two trials that tested the Amplatzer device, the PC TRIAL and RESPECT.
Altogether, the pooled analysis comprised about 2,300 patients. Of these, 440 were lost to 5-year follow-up; their outcomes were imputed into an intent-to-treat analysis based on last-known clinical status.
Dr. Kent and his colleagues also conducted a subanalysis of only the two Amplatzer device trials, which comprised 1,400 patients.
The primary outcome for each analysis was a composite of recurrent stroke, transient ischemic attack (TIA), and early death. The secondary outcome was recurrent stroke alone.
The patients were a mean of 45 years old. About a third had dyslipidemia, and a third had hypertension. The PFO was considered large in 60%.
During the 5-year follow-up period, there were 58 strokes and 54 TIAs. Four patients died during the trial – two in the device arms and two in the medical therapy arms.
When all three trials were analyzed together, PFO closure was not significantly better than medical therapy in the composite endpoint (1.5% vs. 2.3%). The difference in recurrent stroke rate alone was significant (0.7% vs. 1.3%), although Dr. Kent did note that the event rate was very low in both groups.
In the two Amplatzer trials, the device also did not significantly reduce the risk of the composite endpoint. It did confer significant benefit on the stroke-only outcome (hazard ratio, 0.41; 0.4% vs. 1.1%). Again, Dr. Kent said, the event rate was very low for both intervention groups.
There was no significantly increased risk of bleeding with PFO closure in the analysis of all three trials or in the two Amplatzer device trials alone, although both did find a significantly increased risk of atrial fibrillation. In the three-trial analysis, rates of atrial fibrillation were 1.5% vs. 0.48%; in the Amplatzer-only analysis, rates were 0.87% vs. 0.47%. The investigators weren’t able to identify any factors that might predispose to a safety event.
The analyses were sponsored by the National Institutes of Health. Dr. Kent had no financial disclosures, although several coauthors had ties with multiple pharmaceutical companies, or were investigators on the original trials, which were sponsored by the device manufacturers.
On Twitter @alz_gal
NASHVILLE, TENN. – Device closure of a patent foramen ovale didn’t reduce the composite risk of death, stroke, or transient ischemic attack when compared against medical therapy alone in a pooled analysis of three trials.
Device intervention, did, however, reduce the individual risk of recurrent stroke, although the absolute reduction seemed modest, Dr. David M. Kent said at the International Stroke Conference, sponsored by the American Heart Association.
Overall device closure conferred about a 30% annual risk reduction of recurrent stroke upon those with patent foramen ovale (PFO) who had experienced a prior cryptogenic stroke. About 30 patients would have to be treated to prevent one stroke over 5 years, said Dr. Kent of Tufts University, Boston.
“It’s not a huge benefit – it’s not like thrombectomy. But it’s comparable to the benefit of high-dose statins seen in the SPARCL [Stroke Prevention by Aggressive Reduction in Cholesterol Levels] trial.”
Dr. Kent and his colleagues’ analysis used three trials: the CLOSURE trial, which used the STARflex device, and two trials that tested the Amplatzer device, the PC TRIAL and RESPECT.
Altogether, the pooled analysis comprised about 2,300 patients. Of these, 440 were lost to 5-year follow-up; their outcomes were imputed into an intent-to-treat analysis based on last-known clinical status.
Dr. Kent and his colleagues also conducted a subanalysis of only the two Amplatzer device trials, which comprised 1,400 patients.
The primary outcome for each analysis was a composite of recurrent stroke, transient ischemic attack (TIA), and early death. The secondary outcome was recurrent stroke alone.
The patients were a mean of 45 years old. About a third had dyslipidemia, and a third had hypertension. The PFO was considered large in 60%.
During the 5-year follow-up period, there were 58 strokes and 54 TIAs. Four patients died during the trial – two in the device arms and two in the medical therapy arms.
When all three trials were analyzed together, PFO closure was not significantly better than medical therapy in the composite endpoint (1.5% vs. 2.3%). The difference in recurrent stroke rate alone was significant (0.7% vs. 1.3%), although Dr. Kent did note that the event rate was very low in both groups.
In the two Amplatzer trials, the device also did not significantly reduce the risk of the composite endpoint. It did confer significant benefit on the stroke-only outcome (hazard ratio, 0.41; 0.4% vs. 1.1%). Again, Dr. Kent said, the event rate was very low for both intervention groups.
There was no significantly increased risk of bleeding with PFO closure in the analysis of all three trials or in the two Amplatzer device trials alone, although both did find a significantly increased risk of atrial fibrillation. In the three-trial analysis, rates of atrial fibrillation were 1.5% vs. 0.48%; in the Amplatzer-only analysis, rates were 0.87% vs. 0.47%. The investigators weren’t able to identify any factors that might predispose to a safety event.
The analyses were sponsored by the National Institutes of Health. Dr. Kent had no financial disclosures, although several coauthors had ties with multiple pharmaceutical companies, or were investigators on the original trials, which were sponsored by the device manufacturers.
On Twitter @alz_gal
NASHVILLE, TENN. – Device closure of a patent foramen ovale didn’t reduce the composite risk of death, stroke, or transient ischemic attack when compared against medical therapy alone in a pooled analysis of three trials.
Device intervention, did, however, reduce the individual risk of recurrent stroke, although the absolute reduction seemed modest, Dr. David M. Kent said at the International Stroke Conference, sponsored by the American Heart Association.
Overall device closure conferred about a 30% annual risk reduction of recurrent stroke upon those with patent foramen ovale (PFO) who had experienced a prior cryptogenic stroke. About 30 patients would have to be treated to prevent one stroke over 5 years, said Dr. Kent of Tufts University, Boston.
“It’s not a huge benefit – it’s not like thrombectomy. But it’s comparable to the benefit of high-dose statins seen in the SPARCL [Stroke Prevention by Aggressive Reduction in Cholesterol Levels] trial.”
Dr. Kent and his colleagues’ analysis used three trials: the CLOSURE trial, which used the STARflex device, and two trials that tested the Amplatzer device, the PC TRIAL and RESPECT.
Altogether, the pooled analysis comprised about 2,300 patients. Of these, 440 were lost to 5-year follow-up; their outcomes were imputed into an intent-to-treat analysis based on last-known clinical status.
Dr. Kent and his colleagues also conducted a subanalysis of only the two Amplatzer device trials, which comprised 1,400 patients.
The primary outcome for each analysis was a composite of recurrent stroke, transient ischemic attack (TIA), and early death. The secondary outcome was recurrent stroke alone.
The patients were a mean of 45 years old. About a third had dyslipidemia, and a third had hypertension. The PFO was considered large in 60%.
During the 5-year follow-up period, there were 58 strokes and 54 TIAs. Four patients died during the trial – two in the device arms and two in the medical therapy arms.
When all three trials were analyzed together, PFO closure was not significantly better than medical therapy in the composite endpoint (1.5% vs. 2.3%). The difference in recurrent stroke rate alone was significant (0.7% vs. 1.3%), although Dr. Kent did note that the event rate was very low in both groups.
In the two Amplatzer trials, the device also did not significantly reduce the risk of the composite endpoint. It did confer significant benefit on the stroke-only outcome (hazard ratio, 0.41; 0.4% vs. 1.1%). Again, Dr. Kent said, the event rate was very low for both intervention groups.
There was no significantly increased risk of bleeding with PFO closure in the analysis of all three trials or in the two Amplatzer device trials alone, although both did find a significantly increased risk of atrial fibrillation. In the three-trial analysis, rates of atrial fibrillation were 1.5% vs. 0.48%; in the Amplatzer-only analysis, rates were 0.87% vs. 0.47%. The investigators weren’t able to identify any factors that might predispose to a safety event.
The analyses were sponsored by the National Institutes of Health. Dr. Kent had no financial disclosures, although several coauthors had ties with multiple pharmaceutical companies, or were investigators on the original trials, which were sponsored by the device manufacturers.
On Twitter @alz_gal
AT THE INTERNATIONAL STROKE CONFERENCE
Key clinical point: Device closure of a patent foramen ovale seems to reduce the risk of recurrent stroke – but not a composite of death, stroke, or TIA – better than medical therapy.
Major finding: Device closure of PFO conferred about a 30% decline in the risk of recurrent stroke over a 5-year follow-up.
Data source: The analysis pooled results for 2,300 patients who participated in three trials.
Disclosures: The analyses were sponsored by the National Institutes of Health. Dr. Kent had no financial disclosures, although several coauthors had ties with multiple pharmaceutical companies, or were investigators on the original trials, which were sponsored by the device manufacturers.
Long-acting basal insulins aim to reduce hypoglycemia
NEW YORK – Three new insulins hold the potential for decreasing the risk of severe hypoglycemia episodes in patients with diabetes, Dr. Elizabeth R. Seaquist said at the annual advanced postgraduate course held by the American Diabetes Association.
None of the products are yet available in the the United States, although one of the insulins was approved in February by the Food and Drug Administration and is expected to be on the market later this year. The new insulin glargine (Toujeo; Sanofi), a once-daily long-acting basal insulin, received its FDA approval on Feb. 25.
Toujeo has a flatter glycemic profile and a longer duration of action than does its predecessor insulin glargine, Lantus, said Dr. Seaquist, professor of medicine and the Pennock Family Chair in Diabetes Research at the University of Minnesota, Minneapolis.
The approval of Toujeo was based on results from the EDITION clinical trial series, which included more than 3,500 adults with type 1 or type 2 diabetes. In those studies, once-daily Toujeo was compared with once-daily insulin Lantus in open-label, randomized, active-control, parallel, treat-to-target studies of up to 26 weeks of duration with 6 months’ safety extension, according to a Sanofi press release.
Insulin degludec, a once-daily, long-acting basal insulin analogue, is available in the European Union as Tresiba (Novo Nordisk) at strengths of 100 U/mL and 200 U/mL. The FDA denied its approval in 2013, after a review committee expressed concern about a potential increased risk of major cardiovascular events. The FDA has requested that additional cardiovascular data from a dedicated cardiovascular outcomes trial be provided before considering approval.
Basal insulin peglispro (Lilly) has successfully completed all its phase III studies and is on track for FDA submission this year. The primary efficacy endpoint of noninferior HbA1c, compared with insulin glargine was met in both the IMAGINE-1 and IMAGINE-3 trials, and significantly more patients taking basal insulin peglispro achieved an HbA1c of less than 7%. Further, patients taking basal insulin peglispro experienced weight loss and patients taking insulin glargine experienced weight gain, according to Lilly press releases.
Both trials – in which patients were taking mealtime and basal insulin – also found a significantly lower rate of nocturnal hypoglycemia with basal insulin peglispro. Because of a higher rate of daytime hypoglycemic events, however, there was a statistically significant increase in the rate of total hypoglycemia for patients taking basal insulin peglispro.
In the open-label IMAGINE-1 trial, patients taking basal insulin peglispro reported a statistically significant higher rate of severe hypoglycemic events. However, in the larger, blinded IMAGINE-3 trial the rate of severe hypoglycemic events for treatment with basal insulin peglispro was numerically lower, but the difference was not statistically significant.
The fear of hypoglycemia can prompt patients to be nonadherent with their insulin regimen, Dr. Seaquist said. “And we all know that many patients don’t reach their glycemic goal, so they need additional medications that then put them at an increased risk of hypoglycemia.”
According to one review article, an intensively treated individual with type 1 diabetes can experience up to 10 episodes of symptomatic hypoglycemia per week and severe temporarily disabling hypoglycemia at least once a year. Hypoglycemia is also relatively common in type 2 diabetes, with prevalence rates of 70%-80% in clinical trials using insulin to achieve good metabolic control.
Dr. Seaquist disclosed that she is a consultant for Sanofi Aventis, the maker of Toujeo.
NEW YORK – Three new insulins hold the potential for decreasing the risk of severe hypoglycemia episodes in patients with diabetes, Dr. Elizabeth R. Seaquist said at the annual advanced postgraduate course held by the American Diabetes Association.
None of the products are yet available in the the United States, although one of the insulins was approved in February by the Food and Drug Administration and is expected to be on the market later this year. The new insulin glargine (Toujeo; Sanofi), a once-daily long-acting basal insulin, received its FDA approval on Feb. 25.
Toujeo has a flatter glycemic profile and a longer duration of action than does its predecessor insulin glargine, Lantus, said Dr. Seaquist, professor of medicine and the Pennock Family Chair in Diabetes Research at the University of Minnesota, Minneapolis.
The approval of Toujeo was based on results from the EDITION clinical trial series, which included more than 3,500 adults with type 1 or type 2 diabetes. In those studies, once-daily Toujeo was compared with once-daily insulin Lantus in open-label, randomized, active-control, parallel, treat-to-target studies of up to 26 weeks of duration with 6 months’ safety extension, according to a Sanofi press release.
Insulin degludec, a once-daily, long-acting basal insulin analogue, is available in the European Union as Tresiba (Novo Nordisk) at strengths of 100 U/mL and 200 U/mL. The FDA denied its approval in 2013, after a review committee expressed concern about a potential increased risk of major cardiovascular events. The FDA has requested that additional cardiovascular data from a dedicated cardiovascular outcomes trial be provided before considering approval.
Basal insulin peglispro (Lilly) has successfully completed all its phase III studies and is on track for FDA submission this year. The primary efficacy endpoint of noninferior HbA1c, compared with insulin glargine was met in both the IMAGINE-1 and IMAGINE-3 trials, and significantly more patients taking basal insulin peglispro achieved an HbA1c of less than 7%. Further, patients taking basal insulin peglispro experienced weight loss and patients taking insulin glargine experienced weight gain, according to Lilly press releases.
Both trials – in which patients were taking mealtime and basal insulin – also found a significantly lower rate of nocturnal hypoglycemia with basal insulin peglispro. Because of a higher rate of daytime hypoglycemic events, however, there was a statistically significant increase in the rate of total hypoglycemia for patients taking basal insulin peglispro.
In the open-label IMAGINE-1 trial, patients taking basal insulin peglispro reported a statistically significant higher rate of severe hypoglycemic events. However, in the larger, blinded IMAGINE-3 trial the rate of severe hypoglycemic events for treatment with basal insulin peglispro was numerically lower, but the difference was not statistically significant.
The fear of hypoglycemia can prompt patients to be nonadherent with their insulin regimen, Dr. Seaquist said. “And we all know that many patients don’t reach their glycemic goal, so they need additional medications that then put them at an increased risk of hypoglycemia.”
According to one review article, an intensively treated individual with type 1 diabetes can experience up to 10 episodes of symptomatic hypoglycemia per week and severe temporarily disabling hypoglycemia at least once a year. Hypoglycemia is also relatively common in type 2 diabetes, with prevalence rates of 70%-80% in clinical trials using insulin to achieve good metabolic control.
Dr. Seaquist disclosed that she is a consultant for Sanofi Aventis, the maker of Toujeo.
NEW YORK – Three new insulins hold the potential for decreasing the risk of severe hypoglycemia episodes in patients with diabetes, Dr. Elizabeth R. Seaquist said at the annual advanced postgraduate course held by the American Diabetes Association.
None of the products are yet available in the the United States, although one of the insulins was approved in February by the Food and Drug Administration and is expected to be on the market later this year. The new insulin glargine (Toujeo; Sanofi), a once-daily long-acting basal insulin, received its FDA approval on Feb. 25.
Toujeo has a flatter glycemic profile and a longer duration of action than does its predecessor insulin glargine, Lantus, said Dr. Seaquist, professor of medicine and the Pennock Family Chair in Diabetes Research at the University of Minnesota, Minneapolis.
The approval of Toujeo was based on results from the EDITION clinical trial series, which included more than 3,500 adults with type 1 or type 2 diabetes. In those studies, once-daily Toujeo was compared with once-daily insulin Lantus in open-label, randomized, active-control, parallel, treat-to-target studies of up to 26 weeks of duration with 6 months’ safety extension, according to a Sanofi press release.
Insulin degludec, a once-daily, long-acting basal insulin analogue, is available in the European Union as Tresiba (Novo Nordisk) at strengths of 100 U/mL and 200 U/mL. The FDA denied its approval in 2013, after a review committee expressed concern about a potential increased risk of major cardiovascular events. The FDA has requested that additional cardiovascular data from a dedicated cardiovascular outcomes trial be provided before considering approval.
Basal insulin peglispro (Lilly) has successfully completed all its phase III studies and is on track for FDA submission this year. The primary efficacy endpoint of noninferior HbA1c, compared with insulin glargine was met in both the IMAGINE-1 and IMAGINE-3 trials, and significantly more patients taking basal insulin peglispro achieved an HbA1c of less than 7%. Further, patients taking basal insulin peglispro experienced weight loss and patients taking insulin glargine experienced weight gain, according to Lilly press releases.
Both trials – in which patients were taking mealtime and basal insulin – also found a significantly lower rate of nocturnal hypoglycemia with basal insulin peglispro. Because of a higher rate of daytime hypoglycemic events, however, there was a statistically significant increase in the rate of total hypoglycemia for patients taking basal insulin peglispro.
In the open-label IMAGINE-1 trial, patients taking basal insulin peglispro reported a statistically significant higher rate of severe hypoglycemic events. However, in the larger, blinded IMAGINE-3 trial the rate of severe hypoglycemic events for treatment with basal insulin peglispro was numerically lower, but the difference was not statistically significant.
The fear of hypoglycemia can prompt patients to be nonadherent with their insulin regimen, Dr. Seaquist said. “And we all know that many patients don’t reach their glycemic goal, so they need additional medications that then put them at an increased risk of hypoglycemia.”
According to one review article, an intensively treated individual with type 1 diabetes can experience up to 10 episodes of symptomatic hypoglycemia per week and severe temporarily disabling hypoglycemia at least once a year. Hypoglycemia is also relatively common in type 2 diabetes, with prevalence rates of 70%-80% in clinical trials using insulin to achieve good metabolic control.
Dr. Seaquist disclosed that she is a consultant for Sanofi Aventis, the maker of Toujeo.
EXPERT ANALYSIS FROM THE ADA ADVANCED POSTGRADUATE COURSE
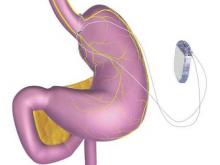
Vagal stimulation may help upper limb stroke recovery
NASHVILLE, TENN. – Patients with upper limbs affected by ischemic stroke who paired traditional rehabilitation exercises with pulsed vagus nerve stimulation boosted functional scores significantly higher than did those who performed exercises alone in a small, randomized pilot trial.
Taken together with the low rate of adverse events associated with device implantation, the study suggests that coupling the interventions is feasible and likely to be beneficial, Dr. Jesse Dawson of the University of Glasgow, Scotland, said at the International Stroke Conference, sponsored by the American Heart Association.
The vagus nerve stimulator (VNS) is typically used to suppress epileptiform discharges and circumvent seizures. The usual stimulation pattern is continuous cycles of 30 seconds on and 5 minutes off. In his randomized, controlled trial, Dr. Dawson set the device to deliver 0.5-second pulses that coincided with each repetition of a rehabilitative movement. When simulated, the nerve releases two proneuroplastic neurotransmitters, acetylcholine and norepinephrine, which then disperse over the cerebral cortex.
“Our theory was that if we timed these releases at specific periods during rehabilitation therapy, we might be able to drive neuroplasticity toward those specific tasks,” Dr. Dawson said at the conference. The technique has proved effective in both aged rats and rat stroke models, he added.
The trial comprised 20 patients who had experienced an ischemic stroke about 2 years prior. Each was left with residual dysfunction in an upper extremity; seven had a paretic limb. The mean Action Research Arm Test (ARAT) score was 33, and the mean upper extremity Fugl-Meyer score was 43, indicating moderate impairment.
Ten patients underwent VNS implantation. Nine completed the trial. One withdrew after 2 weeks because of a transient vocal cord palsy. This later resolved spontaneously.
Other adverse events related to the VNS were also transient. They included taste disturbance, chest pain, mild dysphagia, and nausea after a therapy session.
The 6-week intervention consisted of 18 sessions, each lasting 2 hours. In each, the rehabilitative movement was repeated 300-400 times.
In a per-protocol analysis, there was no significant difference in the upper extremity Fugl-Meyer score at the study’s end. However, when the patient who had withdrawn was excluded from the analysis, the results did become statistically significant. Patients in the dual-therapy group gained almost 10 points, compared with a 3-point gain in the exercise-only group. The ARAT scores were not different at study’s end.
In light of the positive initial results, a sham-controlled, randomized trial is in the works. The trial will randomize 20-25 patients to either the VNS-paired exercise or exercise-only interventions. All participants will receive the VNS device, but only the paired intervention group will receive actual stimuli.
Patricia Smith, Ph.D., the Doris E. Porter Professor in Physical Therapy at the University of Texas Southwestern, Dallas, is the lead investigator.
MicroTransponder, which makes the VNS unit, is sponsoring both the studies. Neither Dr. Dawson nor Dr. Smith have any financial ties to the company.
On Twitter @alz_gal
NASHVILLE, TENN. – Patients with upper limbs affected by ischemic stroke who paired traditional rehabilitation exercises with pulsed vagus nerve stimulation boosted functional scores significantly higher than did those who performed exercises alone in a small, randomized pilot trial.
Taken together with the low rate of adverse events associated with device implantation, the study suggests that coupling the interventions is feasible and likely to be beneficial, Dr. Jesse Dawson of the University of Glasgow, Scotland, said at the International Stroke Conference, sponsored by the American Heart Association.
The vagus nerve stimulator (VNS) is typically used to suppress epileptiform discharges and circumvent seizures. The usual stimulation pattern is continuous cycles of 30 seconds on and 5 minutes off. In his randomized, controlled trial, Dr. Dawson set the device to deliver 0.5-second pulses that coincided with each repetition of a rehabilitative movement. When simulated, the nerve releases two proneuroplastic neurotransmitters, acetylcholine and norepinephrine, which then disperse over the cerebral cortex.
“Our theory was that if we timed these releases at specific periods during rehabilitation therapy, we might be able to drive neuroplasticity toward those specific tasks,” Dr. Dawson said at the conference. The technique has proved effective in both aged rats and rat stroke models, he added.
The trial comprised 20 patients who had experienced an ischemic stroke about 2 years prior. Each was left with residual dysfunction in an upper extremity; seven had a paretic limb. The mean Action Research Arm Test (ARAT) score was 33, and the mean upper extremity Fugl-Meyer score was 43, indicating moderate impairment.
Ten patients underwent VNS implantation. Nine completed the trial. One withdrew after 2 weeks because of a transient vocal cord palsy. This later resolved spontaneously.
Other adverse events related to the VNS were also transient. They included taste disturbance, chest pain, mild dysphagia, and nausea after a therapy session.
The 6-week intervention consisted of 18 sessions, each lasting 2 hours. In each, the rehabilitative movement was repeated 300-400 times.
In a per-protocol analysis, there was no significant difference in the upper extremity Fugl-Meyer score at the study’s end. However, when the patient who had withdrawn was excluded from the analysis, the results did become statistically significant. Patients in the dual-therapy group gained almost 10 points, compared with a 3-point gain in the exercise-only group. The ARAT scores were not different at study’s end.
In light of the positive initial results, a sham-controlled, randomized trial is in the works. The trial will randomize 20-25 patients to either the VNS-paired exercise or exercise-only interventions. All participants will receive the VNS device, but only the paired intervention group will receive actual stimuli.
Patricia Smith, Ph.D., the Doris E. Porter Professor in Physical Therapy at the University of Texas Southwestern, Dallas, is the lead investigator.
MicroTransponder, which makes the VNS unit, is sponsoring both the studies. Neither Dr. Dawson nor Dr. Smith have any financial ties to the company.
On Twitter @alz_gal
NASHVILLE, TENN. – Patients with upper limbs affected by ischemic stroke who paired traditional rehabilitation exercises with pulsed vagus nerve stimulation boosted functional scores significantly higher than did those who performed exercises alone in a small, randomized pilot trial.
Taken together with the low rate of adverse events associated with device implantation, the study suggests that coupling the interventions is feasible and likely to be beneficial, Dr. Jesse Dawson of the University of Glasgow, Scotland, said at the International Stroke Conference, sponsored by the American Heart Association.
The vagus nerve stimulator (VNS) is typically used to suppress epileptiform discharges and circumvent seizures. The usual stimulation pattern is continuous cycles of 30 seconds on and 5 minutes off. In his randomized, controlled trial, Dr. Dawson set the device to deliver 0.5-second pulses that coincided with each repetition of a rehabilitative movement. When simulated, the nerve releases two proneuroplastic neurotransmitters, acetylcholine and norepinephrine, which then disperse over the cerebral cortex.
“Our theory was that if we timed these releases at specific periods during rehabilitation therapy, we might be able to drive neuroplasticity toward those specific tasks,” Dr. Dawson said at the conference. The technique has proved effective in both aged rats and rat stroke models, he added.
The trial comprised 20 patients who had experienced an ischemic stroke about 2 years prior. Each was left with residual dysfunction in an upper extremity; seven had a paretic limb. The mean Action Research Arm Test (ARAT) score was 33, and the mean upper extremity Fugl-Meyer score was 43, indicating moderate impairment.
Ten patients underwent VNS implantation. Nine completed the trial. One withdrew after 2 weeks because of a transient vocal cord palsy. This later resolved spontaneously.
Other adverse events related to the VNS were also transient. They included taste disturbance, chest pain, mild dysphagia, and nausea after a therapy session.
The 6-week intervention consisted of 18 sessions, each lasting 2 hours. In each, the rehabilitative movement was repeated 300-400 times.
In a per-protocol analysis, there was no significant difference in the upper extremity Fugl-Meyer score at the study’s end. However, when the patient who had withdrawn was excluded from the analysis, the results did become statistically significant. Patients in the dual-therapy group gained almost 10 points, compared with a 3-point gain in the exercise-only group. The ARAT scores were not different at study’s end.
In light of the positive initial results, a sham-controlled, randomized trial is in the works. The trial will randomize 20-25 patients to either the VNS-paired exercise or exercise-only interventions. All participants will receive the VNS device, but only the paired intervention group will receive actual stimuli.
Patricia Smith, Ph.D., the Doris E. Porter Professor in Physical Therapy at the University of Texas Southwestern, Dallas, is the lead investigator.
MicroTransponder, which makes the VNS unit, is sponsoring both the studies. Neither Dr. Dawson nor Dr. Smith have any financial ties to the company.
On Twitter @alz_gal
AT THE INTERNATIONAL STROKE CONFERENCE
Key clinical point: Vagus nerve stimulation paired with physical therapy may improve functional recovery in upper limbs after stroke.
Major finding: The paired intervention boosted the upper extremity Fugl-Meyer score by 10 points in the intervention group and 3 points in the control group.
Data source: A randomized trial of 20 patients about 2 years post stroke who had residual dysfunction in an upper extremity.
Disclosures: MicroTransponder, which makes the VNS unit, is sponsoring both the studies. Dr. Dawson has no financial ties to the company.
For hospitalized patients, nutrition and medications affect blood glucose management
Balancing blood sugar in any hospitalized patient can be a tightrope walk, but it is especially challenging when patients can’t eat or are taking steroids.
These patients need a proactive approach that prevents both hyper- and hypoglycemia, both of which have long been tied to poorer outcomes in critically ill patients, Dr. Mary Korytkowski said at the annual advanced postgraduate course held by the American Diabetes Association.
All hospitalized patients should have a blood glucose test on admission. This baseline measure will determine in-hospital glycemic management, which can be fairly straightforward as long as patients are eating regularly. Things get tricky if patients can’t take any food orally and need enteral or parenteral nutrition. Glucocorticoids can also wreak havoc on blood sugars, even in nondiabetic patients.
“There is no question that [blood sugar extremes] impact outcomes, whether or not the patient has [a history of] diabetes,” said Dr. Korytkowski of the University of Pittsburgh. “The question is, what do we do about it?”
The NICE-SUGAR study determined that low-end blood sugar targets may actually be dangerous for hospitalized patients. Since its publication in 2012, glycemic goals have been increased. For non–critically ill patients, a pre-meal blood glucose level should be somewhere between 100-140 mg/dL. For critically ill patients, the goal is more generous at 110-180 mg/dL. Management should aim to sustain those levels, with as few out-of-goal fluctuations as possible.
Patients who have diabetes and are NPO (nothing by mouth) should have their scheduled nutritional insulin held, and their glucose levels maintained with basal and correction insulin, Dr. Korytkowski said. The dose of insulin glargine or insulin detemir will have to be decreased by up to 40% to stay within target; the dose of NPH insulin will probably have to be decreased by 30%-50%.
Those who are converted to NPO after getting their full insulin dose should then be put on a 5% or 10% dextrose solution, which should continue as long as they are getting insulin. “We have this written into our standing orders now,” Dr. Korytkowski said. Of course, routine blood glucose monitoring is essential here.
All patients who receive enteral or parenteral nutrition need a baseline blood glucose level and point-of-care testing regardless of their diabetes history. For those without diabetes, testing can be discontinued if blood glucose stays below 140 mg/dL for 48 hours without insulin.
Those with diabetes should get basal insulin twice a day in addition to their normal regimen, plus correction insulin doses as needed. If this regimen stabilizes glucose at 140-180 mg/dL, it can continue. If levels are above 180 mg/dL on at least two measurements within 24 hours, then adding 25%-50% to the correction insulin dose, along with the scheduled insulin, is indicated.
Steroid diabetes is not uncommon among patients who have been getting glucocorticoids for an extended time, such as those with chronic inflammatory disorders. Steroids oppose insulin action and stimulate gluconeogenesis, leading to an increase in hepatic glucose output. NPH insulin is indicated for these patients. A suggested schedule is 0.1 unit of NPH insulin for every 10 mg prednisone. For example, 0.4 U/kg per day for 40 mg prednisone; 0.3 U/kg per day for 30 mg, and so on.
Dr. Korytkowski disclosed that she is a consultant for Sanofi-Aventis.
On Twitter @alz_gal
Balancing blood sugar in any hospitalized patient can be a tightrope walk, but it is especially challenging when patients can’t eat or are taking steroids.
These patients need a proactive approach that prevents both hyper- and hypoglycemia, both of which have long been tied to poorer outcomes in critically ill patients, Dr. Mary Korytkowski said at the annual advanced postgraduate course held by the American Diabetes Association.
All hospitalized patients should have a blood glucose test on admission. This baseline measure will determine in-hospital glycemic management, which can be fairly straightforward as long as patients are eating regularly. Things get tricky if patients can’t take any food orally and need enteral or parenteral nutrition. Glucocorticoids can also wreak havoc on blood sugars, even in nondiabetic patients.
“There is no question that [blood sugar extremes] impact outcomes, whether or not the patient has [a history of] diabetes,” said Dr. Korytkowski of the University of Pittsburgh. “The question is, what do we do about it?”
The NICE-SUGAR study determined that low-end blood sugar targets may actually be dangerous for hospitalized patients. Since its publication in 2012, glycemic goals have been increased. For non–critically ill patients, a pre-meal blood glucose level should be somewhere between 100-140 mg/dL. For critically ill patients, the goal is more generous at 110-180 mg/dL. Management should aim to sustain those levels, with as few out-of-goal fluctuations as possible.
Patients who have diabetes and are NPO (nothing by mouth) should have their scheduled nutritional insulin held, and their glucose levels maintained with basal and correction insulin, Dr. Korytkowski said. The dose of insulin glargine or insulin detemir will have to be decreased by up to 40% to stay within target; the dose of NPH insulin will probably have to be decreased by 30%-50%.
Those who are converted to NPO after getting their full insulin dose should then be put on a 5% or 10% dextrose solution, which should continue as long as they are getting insulin. “We have this written into our standing orders now,” Dr. Korytkowski said. Of course, routine blood glucose monitoring is essential here.
All patients who receive enteral or parenteral nutrition need a baseline blood glucose level and point-of-care testing regardless of their diabetes history. For those without diabetes, testing can be discontinued if blood glucose stays below 140 mg/dL for 48 hours without insulin.
Those with diabetes should get basal insulin twice a day in addition to their normal regimen, plus correction insulin doses as needed. If this regimen stabilizes glucose at 140-180 mg/dL, it can continue. If levels are above 180 mg/dL on at least two measurements within 24 hours, then adding 25%-50% to the correction insulin dose, along with the scheduled insulin, is indicated.
Steroid diabetes is not uncommon among patients who have been getting glucocorticoids for an extended time, such as those with chronic inflammatory disorders. Steroids oppose insulin action and stimulate gluconeogenesis, leading to an increase in hepatic glucose output. NPH insulin is indicated for these patients. A suggested schedule is 0.1 unit of NPH insulin for every 10 mg prednisone. For example, 0.4 U/kg per day for 40 mg prednisone; 0.3 U/kg per day for 30 mg, and so on.
Dr. Korytkowski disclosed that she is a consultant for Sanofi-Aventis.
On Twitter @alz_gal
Balancing blood sugar in any hospitalized patient can be a tightrope walk, but it is especially challenging when patients can’t eat or are taking steroids.
These patients need a proactive approach that prevents both hyper- and hypoglycemia, both of which have long been tied to poorer outcomes in critically ill patients, Dr. Mary Korytkowski said at the annual advanced postgraduate course held by the American Diabetes Association.
All hospitalized patients should have a blood glucose test on admission. This baseline measure will determine in-hospital glycemic management, which can be fairly straightforward as long as patients are eating regularly. Things get tricky if patients can’t take any food orally and need enteral or parenteral nutrition. Glucocorticoids can also wreak havoc on blood sugars, even in nondiabetic patients.
“There is no question that [blood sugar extremes] impact outcomes, whether or not the patient has [a history of] diabetes,” said Dr. Korytkowski of the University of Pittsburgh. “The question is, what do we do about it?”
The NICE-SUGAR study determined that low-end blood sugar targets may actually be dangerous for hospitalized patients. Since its publication in 2012, glycemic goals have been increased. For non–critically ill patients, a pre-meal blood glucose level should be somewhere between 100-140 mg/dL. For critically ill patients, the goal is more generous at 110-180 mg/dL. Management should aim to sustain those levels, with as few out-of-goal fluctuations as possible.
Patients who have diabetes and are NPO (nothing by mouth) should have their scheduled nutritional insulin held, and their glucose levels maintained with basal and correction insulin, Dr. Korytkowski said. The dose of insulin glargine or insulin detemir will have to be decreased by up to 40% to stay within target; the dose of NPH insulin will probably have to be decreased by 30%-50%.
Those who are converted to NPO after getting their full insulin dose should then be put on a 5% or 10% dextrose solution, which should continue as long as they are getting insulin. “We have this written into our standing orders now,” Dr. Korytkowski said. Of course, routine blood glucose monitoring is essential here.
All patients who receive enteral or parenteral nutrition need a baseline blood glucose level and point-of-care testing regardless of their diabetes history. For those without diabetes, testing can be discontinued if blood glucose stays below 140 mg/dL for 48 hours without insulin.
Those with diabetes should get basal insulin twice a day in addition to their normal regimen, plus correction insulin doses as needed. If this regimen stabilizes glucose at 140-180 mg/dL, it can continue. If levels are above 180 mg/dL on at least two measurements within 24 hours, then adding 25%-50% to the correction insulin dose, along with the scheduled insulin, is indicated.
Steroid diabetes is not uncommon among patients who have been getting glucocorticoids for an extended time, such as those with chronic inflammatory disorders. Steroids oppose insulin action and stimulate gluconeogenesis, leading to an increase in hepatic glucose output. NPH insulin is indicated for these patients. A suggested schedule is 0.1 unit of NPH insulin for every 10 mg prednisone. For example, 0.4 U/kg per day for 40 mg prednisone; 0.3 U/kg per day for 30 mg, and so on.
Dr. Korytkowski disclosed that she is a consultant for Sanofi-Aventis.
On Twitter @alz_gal
EXPERT ANALYSIS AT THE ADA ADVANCED POSTGRADUATE COURSE
Recent data suggest little pancreatic risk with incretin medications
NEW YORK–Incretin-based diabetes medications may not be as harmful to the pancreas as previously feared.
Emerging clinical data, combined with concerns about the accuracy of polyclonal GLP-1 receptor antibody staining, suggest that there may be little to no risk for either treatment-related pancreatitis or pancreatic cancer in patients with diabetes, Dr. Vanita R. Aroda said at the annual advanced postgraduate course held by the American Diabetes Association.
“No study has definitely concluded that there is a causal link, and but we should still keep the risk as something to at least be aware of,” said Dr. Aroda of the MedStar Research Institute, Hyattsville, Md. “Until we have more information, patients who have pancreatic risk factors probably shouldn’t receive the drugs.”
A number of confounders cast doubt on past studies showing a pathologic association between incretins and pancreatic adverse events. Although there are suggestive clinical data, preclinical data based on GLP-1 receptor staining is unreliable, because the polyclonal antibodies generally used don’t consistently stain GLP-1 transfected cells.
“In nontransfected cells, you would expect to see no uptake, but with two of the antibodies, you see quite a bit of uptake in nontransfected as well as transfected cells.”
A third polyclonal antibody has difficulty showing uptake in either type of cell. Only one, the monoclonal 3F52 antibody released in 2013, seems to reliably differentiate GLP1 receptors in these models.
In Dr. Aroda’s opinion, the more reliable results from the monoclonal antibody cast much doubt on all previous preclinical studies that used the polyclonal antibodies.
Nonrandomized data based on case reporting also add to the uncertainty, she said. Data in the Food and Drug Administration’s adverse events reporting system have shown a five- to six-time increased risk of incretin-associated pancreatitis and pancreatic cancer, but “even the FDA has said that spontaneous reports cannot be used to determine the true incidence rates of drug adverse events.” Issues of underreporting, lack of established background rates, and inadequate documentation of clinical details (comorbidities, concomitant medications, and medication errors) cloud the picture. Clarity will remain uncertain as long as decisions about safety are made based on such data.
Similar problems plague studies of discharge data, reflected in the fact that many of them have come to conflicting determinations. The most recent data, however, come from a meta-analysis comprising more than 1.3 million patients among nine insurance claims databases in North America and the European Union. The analysis found no increased risk of acute pancreatitis and any incretin-based therapy (Diabetes Obes. Metab. 2015;17:32-41).
Another recent meta-analysis showed that the adjusted risk of acute pancreatitis was not increased among Danes who had been treated with an incretin drug (Diabetes Care 2015 [doi:10.2337/dc13-2983])
A 2013 study of brain-dead organ donors did find a 40% increased pancreatic mass in diabetes patients treated with incretin therapy, compared to those who didn’t get it and to control patients. Treated patients also over-expressed both exocrine cell proliferation and pancreatic intraepithelial neoplasia (Diabetes 2013;62:2595-604).
“This study has been analyzed and reanalyzed,” Dr. Aroda said. “But it’s really comparing apples to oranges. Five patients with diabetes weren’t on any medication; four were on insulin and four on metformin only; and seven were on two or more agents. Age ranged from 40 to 58 years, and we know age is a big risk factor here.”
Dr. Aroda also examined individual drugs in her own extensive patient database. She found no differences in pancreatitis or cancer associated with liraglutide, exenatide, lixisenatide, or sitagliptin.
The European Medicines Agency is conducting a large evaluation of cardiovascular, cerebrovascular, renal and pancreatic events among diabetes patients taking noninsulin glucose-lowering agents.(SAFEGUARD). It will include combined data covering more than 240 million patient/years.
The National Institutes of Health is also looking at the issue with a consortium study of chronic pancreatitis and pancreatic cancer in diabetes patients.
These projects should result in a considerable amount of reliable data on the issue, Dr. Aroda said.
In the meantime, neither the American Diabetes Association, the Endocrine Society or any European agencies have changed their treatment guidelines with regard to incretin-based therapies.
Dr. Aroda had no financial disclosures.
On Twitter @alz_gal
NEW YORK–Incretin-based diabetes medications may not be as harmful to the pancreas as previously feared.
Emerging clinical data, combined with concerns about the accuracy of polyclonal GLP-1 receptor antibody staining, suggest that there may be little to no risk for either treatment-related pancreatitis or pancreatic cancer in patients with diabetes, Dr. Vanita R. Aroda said at the annual advanced postgraduate course held by the American Diabetes Association.
“No study has definitely concluded that there is a causal link, and but we should still keep the risk as something to at least be aware of,” said Dr. Aroda of the MedStar Research Institute, Hyattsville, Md. “Until we have more information, patients who have pancreatic risk factors probably shouldn’t receive the drugs.”
A number of confounders cast doubt on past studies showing a pathologic association between incretins and pancreatic adverse events. Although there are suggestive clinical data, preclinical data based on GLP-1 receptor staining is unreliable, because the polyclonal antibodies generally used don’t consistently stain GLP-1 transfected cells.
“In nontransfected cells, you would expect to see no uptake, but with two of the antibodies, you see quite a bit of uptake in nontransfected as well as transfected cells.”
A third polyclonal antibody has difficulty showing uptake in either type of cell. Only one, the monoclonal 3F52 antibody released in 2013, seems to reliably differentiate GLP1 receptors in these models.
In Dr. Aroda’s opinion, the more reliable results from the monoclonal antibody cast much doubt on all previous preclinical studies that used the polyclonal antibodies.
Nonrandomized data based on case reporting also add to the uncertainty, she said. Data in the Food and Drug Administration’s adverse events reporting system have shown a five- to six-time increased risk of incretin-associated pancreatitis and pancreatic cancer, but “even the FDA has said that spontaneous reports cannot be used to determine the true incidence rates of drug adverse events.” Issues of underreporting, lack of established background rates, and inadequate documentation of clinical details (comorbidities, concomitant medications, and medication errors) cloud the picture. Clarity will remain uncertain as long as decisions about safety are made based on such data.
Similar problems plague studies of discharge data, reflected in the fact that many of them have come to conflicting determinations. The most recent data, however, come from a meta-analysis comprising more than 1.3 million patients among nine insurance claims databases in North America and the European Union. The analysis found no increased risk of acute pancreatitis and any incretin-based therapy (Diabetes Obes. Metab. 2015;17:32-41).
Another recent meta-analysis showed that the adjusted risk of acute pancreatitis was not increased among Danes who had been treated with an incretin drug (Diabetes Care 2015 [doi:10.2337/dc13-2983])
A 2013 study of brain-dead organ donors did find a 40% increased pancreatic mass in diabetes patients treated with incretin therapy, compared to those who didn’t get it and to control patients. Treated patients also over-expressed both exocrine cell proliferation and pancreatic intraepithelial neoplasia (Diabetes 2013;62:2595-604).
“This study has been analyzed and reanalyzed,” Dr. Aroda said. “But it’s really comparing apples to oranges. Five patients with diabetes weren’t on any medication; four were on insulin and four on metformin only; and seven were on two or more agents. Age ranged from 40 to 58 years, and we know age is a big risk factor here.”
Dr. Aroda also examined individual drugs in her own extensive patient database. She found no differences in pancreatitis or cancer associated with liraglutide, exenatide, lixisenatide, or sitagliptin.
The European Medicines Agency is conducting a large evaluation of cardiovascular, cerebrovascular, renal and pancreatic events among diabetes patients taking noninsulin glucose-lowering agents.(SAFEGUARD). It will include combined data covering more than 240 million patient/years.
The National Institutes of Health is also looking at the issue with a consortium study of chronic pancreatitis and pancreatic cancer in diabetes patients.
These projects should result in a considerable amount of reliable data on the issue, Dr. Aroda said.
In the meantime, neither the American Diabetes Association, the Endocrine Society or any European agencies have changed their treatment guidelines with regard to incretin-based therapies.
Dr. Aroda had no financial disclosures.
On Twitter @alz_gal
NEW YORK–Incretin-based diabetes medications may not be as harmful to the pancreas as previously feared.
Emerging clinical data, combined with concerns about the accuracy of polyclonal GLP-1 receptor antibody staining, suggest that there may be little to no risk for either treatment-related pancreatitis or pancreatic cancer in patients with diabetes, Dr. Vanita R. Aroda said at the annual advanced postgraduate course held by the American Diabetes Association.
“No study has definitely concluded that there is a causal link, and but we should still keep the risk as something to at least be aware of,” said Dr. Aroda of the MedStar Research Institute, Hyattsville, Md. “Until we have more information, patients who have pancreatic risk factors probably shouldn’t receive the drugs.”
A number of confounders cast doubt on past studies showing a pathologic association between incretins and pancreatic adverse events. Although there are suggestive clinical data, preclinical data based on GLP-1 receptor staining is unreliable, because the polyclonal antibodies generally used don’t consistently stain GLP-1 transfected cells.
“In nontransfected cells, you would expect to see no uptake, but with two of the antibodies, you see quite a bit of uptake in nontransfected as well as transfected cells.”
A third polyclonal antibody has difficulty showing uptake in either type of cell. Only one, the monoclonal 3F52 antibody released in 2013, seems to reliably differentiate GLP1 receptors in these models.
In Dr. Aroda’s opinion, the more reliable results from the monoclonal antibody cast much doubt on all previous preclinical studies that used the polyclonal antibodies.
Nonrandomized data based on case reporting also add to the uncertainty, she said. Data in the Food and Drug Administration’s adverse events reporting system have shown a five- to six-time increased risk of incretin-associated pancreatitis and pancreatic cancer, but “even the FDA has said that spontaneous reports cannot be used to determine the true incidence rates of drug adverse events.” Issues of underreporting, lack of established background rates, and inadequate documentation of clinical details (comorbidities, concomitant medications, and medication errors) cloud the picture. Clarity will remain uncertain as long as decisions about safety are made based on such data.
Similar problems plague studies of discharge data, reflected in the fact that many of them have come to conflicting determinations. The most recent data, however, come from a meta-analysis comprising more than 1.3 million patients among nine insurance claims databases in North America and the European Union. The analysis found no increased risk of acute pancreatitis and any incretin-based therapy (Diabetes Obes. Metab. 2015;17:32-41).
Another recent meta-analysis showed that the adjusted risk of acute pancreatitis was not increased among Danes who had been treated with an incretin drug (Diabetes Care 2015 [doi:10.2337/dc13-2983])
A 2013 study of brain-dead organ donors did find a 40% increased pancreatic mass in diabetes patients treated with incretin therapy, compared to those who didn’t get it and to control patients. Treated patients also over-expressed both exocrine cell proliferation and pancreatic intraepithelial neoplasia (Diabetes 2013;62:2595-604).
“This study has been analyzed and reanalyzed,” Dr. Aroda said. “But it’s really comparing apples to oranges. Five patients with diabetes weren’t on any medication; four were on insulin and four on metformin only; and seven were on two or more agents. Age ranged from 40 to 58 years, and we know age is a big risk factor here.”
Dr. Aroda also examined individual drugs in her own extensive patient database. She found no differences in pancreatitis or cancer associated with liraglutide, exenatide, lixisenatide, or sitagliptin.
The European Medicines Agency is conducting a large evaluation of cardiovascular, cerebrovascular, renal and pancreatic events among diabetes patients taking noninsulin glucose-lowering agents.(SAFEGUARD). It will include combined data covering more than 240 million patient/years.
The National Institutes of Health is also looking at the issue with a consortium study of chronic pancreatitis and pancreatic cancer in diabetes patients.
These projects should result in a considerable amount of reliable data on the issue, Dr. Aroda said.
In the meantime, neither the American Diabetes Association, the Endocrine Society or any European agencies have changed their treatment guidelines with regard to incretin-based therapies.
Dr. Aroda had no financial disclosures.
On Twitter @alz_gal
FROM THE ADA POSTGRADUATE COURSE
Consortium opens Alzheimer’s data to global research community
A public-private Alzheimer’s disease research consortium has christened its open-access research gateway, hoping to speed drug development by sharing data.
The Accelerating Medicines Partnership-Alzheimer’s Disease Knowledge Portal (AMP-AD) provides access to data from four large translational research projects that are investigating novel therapeutic targets in genomic, epigenomic, proteomic, and metabolic pathways. The projects are unique in that each represents a collaboration of federal, academic, and industry investigators. The data portal allows unlimited, embargo-free sharing of data with the global research community; all data must be posted within 3 months of acquisition by the teams.
The first wave of data from these studies is now available to registered users.
The gateway is part of the AMP-AD Target Discovery and Preclinical Validation Project. Led by the National Institute on Aging, the project’s goal is to shorten the time between drug target discovery and new drug development. Each of the four studies is leveraging data from more than 2,000 brain samples included in several existing datasets.
Freely sharing such information will increase the transparency, reproducibility, and translatability of basic research discoveries, program director Suzana Petanceska, Ph.D., said in a press statement.
“The era of big data and open science can be a game-changer in our ability to choose therapeutic targets for Alzheimer’s that may lead to effective therapies tailored to diverse patients,” Dr. Petanceska said. “Simply stated, we can work more effectively together than separately.”
The four research studies are:
• The discovery and preclinical validation of novel targets associated with AD-associated molecular processes.
• Description of AD pathologic processes using a biologic systems approach.
• Discovery of potential proteomic drug targets.
• Identifying potentially druggable targets in innate immune-signaling pathways.
The AMP-AD project is part of the larger National Institutes of Health–led Accelerating Medicines Partnership, which is bringing the same public-private research effort to bear on rheumatoid arthritis, lupus, and diabetes.
On Twitter @alz_gal
A public-private Alzheimer’s disease research consortium has christened its open-access research gateway, hoping to speed drug development by sharing data.
The Accelerating Medicines Partnership-Alzheimer’s Disease Knowledge Portal (AMP-AD) provides access to data from four large translational research projects that are investigating novel therapeutic targets in genomic, epigenomic, proteomic, and metabolic pathways. The projects are unique in that each represents a collaboration of federal, academic, and industry investigators. The data portal allows unlimited, embargo-free sharing of data with the global research community; all data must be posted within 3 months of acquisition by the teams.
The first wave of data from these studies is now available to registered users.
The gateway is part of the AMP-AD Target Discovery and Preclinical Validation Project. Led by the National Institute on Aging, the project’s goal is to shorten the time between drug target discovery and new drug development. Each of the four studies is leveraging data from more than 2,000 brain samples included in several existing datasets.
Freely sharing such information will increase the transparency, reproducibility, and translatability of basic research discoveries, program director Suzana Petanceska, Ph.D., said in a press statement.
“The era of big data and open science can be a game-changer in our ability to choose therapeutic targets for Alzheimer’s that may lead to effective therapies tailored to diverse patients,” Dr. Petanceska said. “Simply stated, we can work more effectively together than separately.”
The four research studies are:
• The discovery and preclinical validation of novel targets associated with AD-associated molecular processes.
• Description of AD pathologic processes using a biologic systems approach.
• Discovery of potential proteomic drug targets.
• Identifying potentially druggable targets in innate immune-signaling pathways.
The AMP-AD project is part of the larger National Institutes of Health–led Accelerating Medicines Partnership, which is bringing the same public-private research effort to bear on rheumatoid arthritis, lupus, and diabetes.
On Twitter @alz_gal
A public-private Alzheimer’s disease research consortium has christened its open-access research gateway, hoping to speed drug development by sharing data.
The Accelerating Medicines Partnership-Alzheimer’s Disease Knowledge Portal (AMP-AD) provides access to data from four large translational research projects that are investigating novel therapeutic targets in genomic, epigenomic, proteomic, and metabolic pathways. The projects are unique in that each represents a collaboration of federal, academic, and industry investigators. The data portal allows unlimited, embargo-free sharing of data with the global research community; all data must be posted within 3 months of acquisition by the teams.
The first wave of data from these studies is now available to registered users.
The gateway is part of the AMP-AD Target Discovery and Preclinical Validation Project. Led by the National Institute on Aging, the project’s goal is to shorten the time between drug target discovery and new drug development. Each of the four studies is leveraging data from more than 2,000 brain samples included in several existing datasets.
Freely sharing such information will increase the transparency, reproducibility, and translatability of basic research discoveries, program director Suzana Petanceska, Ph.D., said in a press statement.
“The era of big data and open science can be a game-changer in our ability to choose therapeutic targets for Alzheimer’s that may lead to effective therapies tailored to diverse patients,” Dr. Petanceska said. “Simply stated, we can work more effectively together than separately.”
The four research studies are:
• The discovery and preclinical validation of novel targets associated with AD-associated molecular processes.
• Description of AD pathologic processes using a biologic systems approach.
• Discovery of potential proteomic drug targets.
• Identifying potentially druggable targets in innate immune-signaling pathways.
The AMP-AD project is part of the larger National Institutes of Health–led Accelerating Medicines Partnership, which is bringing the same public-private research effort to bear on rheumatoid arthritis, lupus, and diabetes.
On Twitter @alz_gal
FDA: Limit Testosterone Use to Men With Specific Medical Conditions
Testosterone therapy is not appropriate for men with age-related low testosterone because it may be associated with an increased risk of cardiovascular events, the Food and Drug Administration has determined.
The agency will now require labeling changes to all prescription testosterone products, clarifying their appropriate use, and warning about a possible increased risk of heart attack and stroke in any patient taking the hormone. Testosterone is approved only for men with specific medical conditions, but is widely used off label for men with age-related low testosterone.
“Health care professionals should make patients aware of this possible risk when deciding whether to start or continue a patient on testosterone therapy,” an FDA statement said. “We are also requiring manufacturers of approved testosterone products to conduct a well-designed clinical trial to more clearly address the question of whether an increased risk of heart attack or stroke exists among users of these products. We are encouraging these manufacturers to work together on a clinical trial, but they are allowed to work separately if they so choose.”
The statement arose from a September 2014 recommendation by the FDA’s Bone, Reproductive, and Urologic Drugs Advisory Committee and Drug Safety and Risk Management Advisory Committee. The groups reviewed studies of aging men using testosterone – some of which reported an increased risk of heart attack, stroke, or death associated with testosterone treatment – and voted 20-1 that the current indication, as worded in the labeling for all testosterone products, should be tightened to make it clear that testosterone therapy is not indicated for men with age-related reductions in testosterone.
Any clinician who prescribes testosterone to a patient who later experiences a cardiovascular event should report that to FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
Testosterone therapy is not appropriate for men with age-related low testosterone because it may be associated with an increased risk of cardiovascular events, the Food and Drug Administration has determined.
The agency will now require labeling changes to all prescription testosterone products, clarifying their appropriate use, and warning about a possible increased risk of heart attack and stroke in any patient taking the hormone. Testosterone is approved only for men with specific medical conditions, but is widely used off label for men with age-related low testosterone.
“Health care professionals should make patients aware of this possible risk when deciding whether to start or continue a patient on testosterone therapy,” an FDA statement said. “We are also requiring manufacturers of approved testosterone products to conduct a well-designed clinical trial to more clearly address the question of whether an increased risk of heart attack or stroke exists among users of these products. We are encouraging these manufacturers to work together on a clinical trial, but they are allowed to work separately if they so choose.”
The statement arose from a September 2014 recommendation by the FDA’s Bone, Reproductive, and Urologic Drugs Advisory Committee and Drug Safety and Risk Management Advisory Committee. The groups reviewed studies of aging men using testosterone – some of which reported an increased risk of heart attack, stroke, or death associated with testosterone treatment – and voted 20-1 that the current indication, as worded in the labeling for all testosterone products, should be tightened to make it clear that testosterone therapy is not indicated for men with age-related reductions in testosterone.
Any clinician who prescribes testosterone to a patient who later experiences a cardiovascular event should report that to FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
Testosterone therapy is not appropriate for men with age-related low testosterone because it may be associated with an increased risk of cardiovascular events, the Food and Drug Administration has determined.
The agency will now require labeling changes to all prescription testosterone products, clarifying their appropriate use, and warning about a possible increased risk of heart attack and stroke in any patient taking the hormone. Testosterone is approved only for men with specific medical conditions, but is widely used off label for men with age-related low testosterone.
“Health care professionals should make patients aware of this possible risk when deciding whether to start or continue a patient on testosterone therapy,” an FDA statement said. “We are also requiring manufacturers of approved testosterone products to conduct a well-designed clinical trial to more clearly address the question of whether an increased risk of heart attack or stroke exists among users of these products. We are encouraging these manufacturers to work together on a clinical trial, but they are allowed to work separately if they so choose.”
The statement arose from a September 2014 recommendation by the FDA’s Bone, Reproductive, and Urologic Drugs Advisory Committee and Drug Safety and Risk Management Advisory Committee. The groups reviewed studies of aging men using testosterone – some of which reported an increased risk of heart attack, stroke, or death associated with testosterone treatment – and voted 20-1 that the current indication, as worded in the labeling for all testosterone products, should be tightened to make it clear that testosterone therapy is not indicated for men with age-related reductions in testosterone.
Any clinician who prescribes testosterone to a patient who later experiences a cardiovascular event should report that to FDA’s MedWatch Safety Information and Adverse Event Reporting Program.