User login
Brain Tumors Glow 'Like Lava' With New Surgical Probe
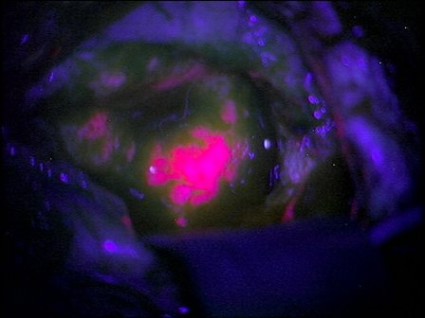
Neurosurgeons can now follow a glowing road map that points the way to cancerous brain tissue, leading thereby to a more effective surgical excision.
Researchers at the Norris Cotton Cancer Center and the Thayer School of Engineering at Dartmouth College, Hanover, N.H., have developed a probe that uses protoporphyrin IX fluorescence, oxygen saturation, hemoglobin concentration, and cell morphology to differentiate cancerous tissue from normal.
The probe identified 94% of glioma tissue in a small pilot study (J. Biomed. Opt. 2012 May 4 [doi:10.1117/1.JBO.17.5.056008]).
Research in Germany 15 years ago suggested that such a tool would identify only highly metabolic primary tumors. But augmenting the fluorescence technique with a computer algorithm that added the other cellular features gave surgeons a "jaw-dropping" view of low-grade tumors.
"The tumor glowed like lava," said Keith Paulsen, Ph.D., a professor of biomedical engineering at the school of engineering and a member of the cancer imaging and radiobiology research program at Norris Cotton Cancer Center.
The team will next evaluate their technique on lung cancers, with investigations of other tumor types to follow.
Neurosurgeons can now follow a glowing road map that points the way to cancerous brain tissue, leading thereby to a more effective surgical excision.
Researchers at the Norris Cotton Cancer Center and the Thayer School of Engineering at Dartmouth College, Hanover, N.H., have developed a probe that uses protoporphyrin IX fluorescence, oxygen saturation, hemoglobin concentration, and cell morphology to differentiate cancerous tissue from normal.
The probe identified 94% of glioma tissue in a small pilot study (J. Biomed. Opt. 2012 May 4 [doi:10.1117/1.JBO.17.5.056008]).
Research in Germany 15 years ago suggested that such a tool would identify only highly metabolic primary tumors. But augmenting the fluorescence technique with a computer algorithm that added the other cellular features gave surgeons a "jaw-dropping" view of low-grade tumors.
"The tumor glowed like lava," said Keith Paulsen, Ph.D., a professor of biomedical engineering at the school of engineering and a member of the cancer imaging and radiobiology research program at Norris Cotton Cancer Center.
The team will next evaluate their technique on lung cancers, with investigations of other tumor types to follow.
Neurosurgeons can now follow a glowing road map that points the way to cancerous brain tissue, leading thereby to a more effective surgical excision.
Researchers at the Norris Cotton Cancer Center and the Thayer School of Engineering at Dartmouth College, Hanover, N.H., have developed a probe that uses protoporphyrin IX fluorescence, oxygen saturation, hemoglobin concentration, and cell morphology to differentiate cancerous tissue from normal.
The probe identified 94% of glioma tissue in a small pilot study (J. Biomed. Opt. 2012 May 4 [doi:10.1117/1.JBO.17.5.056008]).
Research in Germany 15 years ago suggested that such a tool would identify only highly metabolic primary tumors. But augmenting the fluorescence technique with a computer algorithm that added the other cellular features gave surgeons a "jaw-dropping" view of low-grade tumors.
"The tumor glowed like lava," said Keith Paulsen, Ph.D., a professor of biomedical engineering at the school of engineering and a member of the cancer imaging and radiobiology research program at Norris Cotton Cancer Center.
The team will next evaluate their technique on lung cancers, with investigations of other tumor types to follow.
FROM THE JOURNAL OF BIOMEDICAL OPTICS
Experimental Drug Improves Cognition, Memory in Alzheimer's
VANCOUVER, B.C. – An investigational drug has shown promise in improving memory and cognition in patients with mild to moderate Alzheimer’s disease in a 6-month, randomized, placebo-controlled trial that was presented at the Alzheimer’s Association International Conference 2012.
The fact that the patients were still continuing to improve on statistically significant measurements of those aspects of the disease provided a hint, at least, that the drug EVP-6124, an alpha-7 nicotinic acetylcholine receptor partial agonist, could change the course of Alzheimer’s.
But it’s too soon to place any long-term bets, Dr. Dana Hilt said in an interview.
"This is being investigated as a procognitive and symptomatic drug," said Dr. Hilt, senior vice president of clinical development and chief medical officer of EnVivo Pharmaceuticals, which funded the trial. "It certainly appears to improve cognition in a clinically beneficial way at 6 months. But if the curves continue to diverge, is it possible that something more fundamental could be going on? Could this be maintained at 12 months? That’s what we need to know."
Dr. Paul S. Aisen, director of the Alzheimer's Disease Cooperative Study and a professor of neurosciences at the University of California, San Diego, said that EVP-6124’s phase II data looked very good.
"Reviewing the results, I would consider this to be a solidly positive trial," he said in an interview. "While we cannot rely on a relatively small phase II trial to accurately indicate the size of a treatment effect, the consistency of the findings here are encouraging. If efficacy is confirmed in additional trials, this may prove to be an important therapeutic option."
The study group comprised 409 patients who were randomized to one of three drug doses (0.3 mg, 1 mg, or 2 mg daily), or to placebo.
Half of the group were on stable doses of donepezil (Aricept) or rivastigmine (Exelon) and half were not taking any acetylcholinesterase inhibitor (AChEI), at least at the time of the trial, Dr. Hilt said. "Some of them had never taken an AChEI, and some had taken them in the past but were now off, for a number of reasons," including insurance coverage and lack of efficacy.
The subjects’ mean MMSE (Mini-Mental State Exam) at baseline score was 20, but the range was wide, from 12 to 29; the mean score on the CDR (Clinical Dementia Rating) scale was 6.
The primary end points were changes in the ADAS Cog-13 (Alzheimer’s Disease Assessment Scale Cognitive-13) and the CDR sum of boxes scores. Additional prespecified end points were the ADAS Cog-11 and composite measures of cognition and memory.
In each finding, Dr. Hilt noted effect size as well as statistical significance. Effect size is important in clinical trials because it suggests clinical efficacy better than statistical findings, he said in an interview.
"The effect size of approved drugs is somewhere around 0.15-0.28. In terms of clinical change, an effect size of 0.2 is something a trained clinician could detect. And effect size of 0.4 is something the next-door neighbor could detect."
During the first month of the study, the lower-dose groups and the placebo groups all improved their ADAS Cog-13 scores, a finding consistent with a placebo effect often seen in such studies. Interestingly, the 2-mg group remained at baseline for the first 4 months of the trial; after 4 months, cognition scores increased rapidly and outstripped all of the other groups. By the end of the study, the 2-mg group had a mean improvement of 1.5 points over baseline, whereas the placebo group had dropped below baseline, giving a total group separation of 2.2 points (P = .0189; effect size 0.39). This effect size is higher than the 0.28 estimated for high doses of donepezil, rivastigmine, or memantine (Namenda), Dr. Hilt said.
At the end of the treatment period, the curves were still diverging, suggesting that longer treatment might confer more benefit.
On the CDR sum of boxes, the 2-mg dose also performed significantly better than placebo. Again, there was the early upward trend for all groups, including placebo. But after 4 months, the placebo group and the active groups of 0.3 mg and 1 mg began losing those gains. The 2-mg group maintained its gain (P = .0253; effect size 0.31).
Findings on the secondary end points were similarly encouraging, although all were not statistically significant, Dr. Hilt said.
On the ADAS Cog-11, those taking the 2-mg dose faired significantly better than did those in any of the other groups. By the end of the study period, the lower-dose groups and placebo group had declined below baseline to the same degree. The 2-mg group had improved significantly and was still on an upward trend (P = .0151; effect size 0.34).
The ADCS-ADL (Alzheimer's Disease Cooperative Study Activities of Daily Living) scale showed the pattern of early improvement observed with other scales. The 2-mg group maintained its gains, but the between-group difference was not significant at the end of the study (P = .0925; effect size 0.20). Nor was there a significant 24-month difference in the MMSE (P = 0.0955; effect size 0.21).
A composite measure of cognition that included word recall on the ADAS-Cog, word recognition, word association, and orientation significantly favored the 2-mg group (P = .0037; effect size 0.42), as did a composite measure of memory (P = 0.0088; effect size 0.37).
Besides being richly distributed in the hippocampus and frontal cortex, alpha-7 nicotinic acetylcholine receptors are found in many other tissues, including the gut and skeletal muscles. A compound that attaches nonselectively could be loaded with adverse effects, including cardiovascular changes and unpleasant GI reactions. These adverse effects have proved difficult to overcome, and in fact are one reason that a number of nicotinic acetylcholine receptor studies have been discontinued.
"A number of companies tried to develop these, but at high dose levels that had indiscriminate stimulation of the receptors," Dr. Hilt said. "When you give a neurotransmitter to affect receptors in the brain, you have to live with the consequences of stimulating that receptor everywhere. The way we have dealt with this is to use a drug with a very long half-life, in very low concentrations."
The long half-life allows the drug to build up slowly, and keeps levels at a steady concentration without extreme peaks or nadirs. "It turns out this receptor is very finicky. Too high a level overstimulates it and too low a level doesn’t stimulate it at all. I’m convinced this is the major trouble with some of these other drugs."
Adverse events in the trial were mild gastrointestinal issues, including nausea and constipation. No patients of the active-drug study groups withdrew because of an adverse effect. The rate of adverse events in the 2-mg group also varied by baseline medication status, with more among those already taking an AChEI at baseline than among those not on the drugs (64% vs. 42%). Similar differences in adverse events rates occurred in the other treatment groups, although the rates were lower than in the 2-mg group. These differences reflect the expected problems with AChEI drugs, with the additional increase due to EVP-6124, Dr. Hilt said.
All told, there were 23 serious adverse events in the study: 4 in the placebo group, and 19 in the active groups. The investigators deemed that only two were treatment related; two additional events were considered possibly related to treatment.
Dr. Hilt said that EnVivo plans to go forward with a phase III trial early next year.
Dr. Aisen serves as a consultant to Eisai, Pfizer, Novartis, and other companies. He also receives research support from Pfizer and others.
VANCOUVER, B.C. – An investigational drug has shown promise in improving memory and cognition in patients with mild to moderate Alzheimer’s disease in a 6-month, randomized, placebo-controlled trial that was presented at the Alzheimer’s Association International Conference 2012.
The fact that the patients were still continuing to improve on statistically significant measurements of those aspects of the disease provided a hint, at least, that the drug EVP-6124, an alpha-7 nicotinic acetylcholine receptor partial agonist, could change the course of Alzheimer’s.
But it’s too soon to place any long-term bets, Dr. Dana Hilt said in an interview.
"This is being investigated as a procognitive and symptomatic drug," said Dr. Hilt, senior vice president of clinical development and chief medical officer of EnVivo Pharmaceuticals, which funded the trial. "It certainly appears to improve cognition in a clinically beneficial way at 6 months. But if the curves continue to diverge, is it possible that something more fundamental could be going on? Could this be maintained at 12 months? That’s what we need to know."
Dr. Paul S. Aisen, director of the Alzheimer's Disease Cooperative Study and a professor of neurosciences at the University of California, San Diego, said that EVP-6124’s phase II data looked very good.
"Reviewing the results, I would consider this to be a solidly positive trial," he said in an interview. "While we cannot rely on a relatively small phase II trial to accurately indicate the size of a treatment effect, the consistency of the findings here are encouraging. If efficacy is confirmed in additional trials, this may prove to be an important therapeutic option."
The study group comprised 409 patients who were randomized to one of three drug doses (0.3 mg, 1 mg, or 2 mg daily), or to placebo.
Half of the group were on stable doses of donepezil (Aricept) or rivastigmine (Exelon) and half were not taking any acetylcholinesterase inhibitor (AChEI), at least at the time of the trial, Dr. Hilt said. "Some of them had never taken an AChEI, and some had taken them in the past but were now off, for a number of reasons," including insurance coverage and lack of efficacy.
The subjects’ mean MMSE (Mini-Mental State Exam) at baseline score was 20, but the range was wide, from 12 to 29; the mean score on the CDR (Clinical Dementia Rating) scale was 6.
The primary end points were changes in the ADAS Cog-13 (Alzheimer’s Disease Assessment Scale Cognitive-13) and the CDR sum of boxes scores. Additional prespecified end points were the ADAS Cog-11 and composite measures of cognition and memory.
In each finding, Dr. Hilt noted effect size as well as statistical significance. Effect size is important in clinical trials because it suggests clinical efficacy better than statistical findings, he said in an interview.
"The effect size of approved drugs is somewhere around 0.15-0.28. In terms of clinical change, an effect size of 0.2 is something a trained clinician could detect. And effect size of 0.4 is something the next-door neighbor could detect."
During the first month of the study, the lower-dose groups and the placebo groups all improved their ADAS Cog-13 scores, a finding consistent with a placebo effect often seen in such studies. Interestingly, the 2-mg group remained at baseline for the first 4 months of the trial; after 4 months, cognition scores increased rapidly and outstripped all of the other groups. By the end of the study, the 2-mg group had a mean improvement of 1.5 points over baseline, whereas the placebo group had dropped below baseline, giving a total group separation of 2.2 points (P = .0189; effect size 0.39). This effect size is higher than the 0.28 estimated for high doses of donepezil, rivastigmine, or memantine (Namenda), Dr. Hilt said.
At the end of the treatment period, the curves were still diverging, suggesting that longer treatment might confer more benefit.
On the CDR sum of boxes, the 2-mg dose also performed significantly better than placebo. Again, there was the early upward trend for all groups, including placebo. But after 4 months, the placebo group and the active groups of 0.3 mg and 1 mg began losing those gains. The 2-mg group maintained its gain (P = .0253; effect size 0.31).
Findings on the secondary end points were similarly encouraging, although all were not statistically significant, Dr. Hilt said.
On the ADAS Cog-11, those taking the 2-mg dose faired significantly better than did those in any of the other groups. By the end of the study period, the lower-dose groups and placebo group had declined below baseline to the same degree. The 2-mg group had improved significantly and was still on an upward trend (P = .0151; effect size 0.34).
The ADCS-ADL (Alzheimer's Disease Cooperative Study Activities of Daily Living) scale showed the pattern of early improvement observed with other scales. The 2-mg group maintained its gains, but the between-group difference was not significant at the end of the study (P = .0925; effect size 0.20). Nor was there a significant 24-month difference in the MMSE (P = 0.0955; effect size 0.21).
A composite measure of cognition that included word recall on the ADAS-Cog, word recognition, word association, and orientation significantly favored the 2-mg group (P = .0037; effect size 0.42), as did a composite measure of memory (P = 0.0088; effect size 0.37).
Besides being richly distributed in the hippocampus and frontal cortex, alpha-7 nicotinic acetylcholine receptors are found in many other tissues, including the gut and skeletal muscles. A compound that attaches nonselectively could be loaded with adverse effects, including cardiovascular changes and unpleasant GI reactions. These adverse effects have proved difficult to overcome, and in fact are one reason that a number of nicotinic acetylcholine receptor studies have been discontinued.
"A number of companies tried to develop these, but at high dose levels that had indiscriminate stimulation of the receptors," Dr. Hilt said. "When you give a neurotransmitter to affect receptors in the brain, you have to live with the consequences of stimulating that receptor everywhere. The way we have dealt with this is to use a drug with a very long half-life, in very low concentrations."
The long half-life allows the drug to build up slowly, and keeps levels at a steady concentration without extreme peaks or nadirs. "It turns out this receptor is very finicky. Too high a level overstimulates it and too low a level doesn’t stimulate it at all. I’m convinced this is the major trouble with some of these other drugs."
Adverse events in the trial were mild gastrointestinal issues, including nausea and constipation. No patients of the active-drug study groups withdrew because of an adverse effect. The rate of adverse events in the 2-mg group also varied by baseline medication status, with more among those already taking an AChEI at baseline than among those not on the drugs (64% vs. 42%). Similar differences in adverse events rates occurred in the other treatment groups, although the rates were lower than in the 2-mg group. These differences reflect the expected problems with AChEI drugs, with the additional increase due to EVP-6124, Dr. Hilt said.
All told, there were 23 serious adverse events in the study: 4 in the placebo group, and 19 in the active groups. The investigators deemed that only two were treatment related; two additional events were considered possibly related to treatment.
Dr. Hilt said that EnVivo plans to go forward with a phase III trial early next year.
Dr. Aisen serves as a consultant to Eisai, Pfizer, Novartis, and other companies. He also receives research support from Pfizer and others.
VANCOUVER, B.C. – An investigational drug has shown promise in improving memory and cognition in patients with mild to moderate Alzheimer’s disease in a 6-month, randomized, placebo-controlled trial that was presented at the Alzheimer’s Association International Conference 2012.
The fact that the patients were still continuing to improve on statistically significant measurements of those aspects of the disease provided a hint, at least, that the drug EVP-6124, an alpha-7 nicotinic acetylcholine receptor partial agonist, could change the course of Alzheimer’s.
But it’s too soon to place any long-term bets, Dr. Dana Hilt said in an interview.
"This is being investigated as a procognitive and symptomatic drug," said Dr. Hilt, senior vice president of clinical development and chief medical officer of EnVivo Pharmaceuticals, which funded the trial. "It certainly appears to improve cognition in a clinically beneficial way at 6 months. But if the curves continue to diverge, is it possible that something more fundamental could be going on? Could this be maintained at 12 months? That’s what we need to know."
Dr. Paul S. Aisen, director of the Alzheimer's Disease Cooperative Study and a professor of neurosciences at the University of California, San Diego, said that EVP-6124’s phase II data looked very good.
"Reviewing the results, I would consider this to be a solidly positive trial," he said in an interview. "While we cannot rely on a relatively small phase II trial to accurately indicate the size of a treatment effect, the consistency of the findings here are encouraging. If efficacy is confirmed in additional trials, this may prove to be an important therapeutic option."
The study group comprised 409 patients who were randomized to one of three drug doses (0.3 mg, 1 mg, or 2 mg daily), or to placebo.
Half of the group were on stable doses of donepezil (Aricept) or rivastigmine (Exelon) and half were not taking any acetylcholinesterase inhibitor (AChEI), at least at the time of the trial, Dr. Hilt said. "Some of them had never taken an AChEI, and some had taken them in the past but were now off, for a number of reasons," including insurance coverage and lack of efficacy.
The subjects’ mean MMSE (Mini-Mental State Exam) at baseline score was 20, but the range was wide, from 12 to 29; the mean score on the CDR (Clinical Dementia Rating) scale was 6.
The primary end points were changes in the ADAS Cog-13 (Alzheimer’s Disease Assessment Scale Cognitive-13) and the CDR sum of boxes scores. Additional prespecified end points were the ADAS Cog-11 and composite measures of cognition and memory.
In each finding, Dr. Hilt noted effect size as well as statistical significance. Effect size is important in clinical trials because it suggests clinical efficacy better than statistical findings, he said in an interview.
"The effect size of approved drugs is somewhere around 0.15-0.28. In terms of clinical change, an effect size of 0.2 is something a trained clinician could detect. And effect size of 0.4 is something the next-door neighbor could detect."
During the first month of the study, the lower-dose groups and the placebo groups all improved their ADAS Cog-13 scores, a finding consistent with a placebo effect often seen in such studies. Interestingly, the 2-mg group remained at baseline for the first 4 months of the trial; after 4 months, cognition scores increased rapidly and outstripped all of the other groups. By the end of the study, the 2-mg group had a mean improvement of 1.5 points over baseline, whereas the placebo group had dropped below baseline, giving a total group separation of 2.2 points (P = .0189; effect size 0.39). This effect size is higher than the 0.28 estimated for high doses of donepezil, rivastigmine, or memantine (Namenda), Dr. Hilt said.
At the end of the treatment period, the curves were still diverging, suggesting that longer treatment might confer more benefit.
On the CDR sum of boxes, the 2-mg dose also performed significantly better than placebo. Again, there was the early upward trend for all groups, including placebo. But after 4 months, the placebo group and the active groups of 0.3 mg and 1 mg began losing those gains. The 2-mg group maintained its gain (P = .0253; effect size 0.31).
Findings on the secondary end points were similarly encouraging, although all were not statistically significant, Dr. Hilt said.
On the ADAS Cog-11, those taking the 2-mg dose faired significantly better than did those in any of the other groups. By the end of the study period, the lower-dose groups and placebo group had declined below baseline to the same degree. The 2-mg group had improved significantly and was still on an upward trend (P = .0151; effect size 0.34).
The ADCS-ADL (Alzheimer's Disease Cooperative Study Activities of Daily Living) scale showed the pattern of early improvement observed with other scales. The 2-mg group maintained its gains, but the between-group difference was not significant at the end of the study (P = .0925; effect size 0.20). Nor was there a significant 24-month difference in the MMSE (P = 0.0955; effect size 0.21).
A composite measure of cognition that included word recall on the ADAS-Cog, word recognition, word association, and orientation significantly favored the 2-mg group (P = .0037; effect size 0.42), as did a composite measure of memory (P = 0.0088; effect size 0.37).
Besides being richly distributed in the hippocampus and frontal cortex, alpha-7 nicotinic acetylcholine receptors are found in many other tissues, including the gut and skeletal muscles. A compound that attaches nonselectively could be loaded with adverse effects, including cardiovascular changes and unpleasant GI reactions. These adverse effects have proved difficult to overcome, and in fact are one reason that a number of nicotinic acetylcholine receptor studies have been discontinued.
"A number of companies tried to develop these, but at high dose levels that had indiscriminate stimulation of the receptors," Dr. Hilt said. "When you give a neurotransmitter to affect receptors in the brain, you have to live with the consequences of stimulating that receptor everywhere. The way we have dealt with this is to use a drug with a very long half-life, in very low concentrations."
The long half-life allows the drug to build up slowly, and keeps levels at a steady concentration without extreme peaks or nadirs. "It turns out this receptor is very finicky. Too high a level overstimulates it and too low a level doesn’t stimulate it at all. I’m convinced this is the major trouble with some of these other drugs."
Adverse events in the trial were mild gastrointestinal issues, including nausea and constipation. No patients of the active-drug study groups withdrew because of an adverse effect. The rate of adverse events in the 2-mg group also varied by baseline medication status, with more among those already taking an AChEI at baseline than among those not on the drugs (64% vs. 42%). Similar differences in adverse events rates occurred in the other treatment groups, although the rates were lower than in the 2-mg group. These differences reflect the expected problems with AChEI drugs, with the additional increase due to EVP-6124, Dr. Hilt said.
All told, there were 23 serious adverse events in the study: 4 in the placebo group, and 19 in the active groups. The investigators deemed that only two were treatment related; two additional events were considered possibly related to treatment.
Dr. Hilt said that EnVivo plans to go forward with a phase III trial early next year.
Dr. Aisen serves as a consultant to Eisai, Pfizer, Novartis, and other companies. He also receives research support from Pfizer and others.
AT THE ALZHEIMER'S ASSOCIATION INTERNATIONAL CONFERENCE 2012
Major Finding: An investigational alpha-7 partial agonist significantly improved cognition and memory in patients with mild to moderate Alzheimer’s disease in a 6-month, phase IIB study.
Data Source: The randomized, placebo-controlled study tested EVP-6142 in doses of 0.3 mg, 1 mg, and 2 mg in 409 patients.
Disclosures: EnVivo Pharmaceuticals, the company developing the drug, funded the study. Dr. Dana Hilt is a full-time employee of EnVivo. Dr. Aisen serves as a consultant to Eisai, Pfizer, Novartis, and other companies. He also receives research support from Pfizer and others.
Blood Test May Predict Alzheimer's in Cognitively Impaired
VANCOUVER, B.C. – An investigational gene-based blood test identified prodromal Alzheimer’s disease with an accuracy of 81%.
The test is based on a panel of 25 genes active in biological processes associated with Alzheimer’s, Dr. Magnus Sjögren said at the Alzheimer’s Association International Conference 2012.
The genes have functions that include regulating the processing of amyloid and tau, and are involved as well in mitochondrial transport and function, neuronal and brain function, inflammatory processes, and oxidative stress, said Dr. Sjögren, chief medical officer of DiaGenic, the Norwegian company that is developing the test.
Under development in Oslo, the test could be submitted to the U.S. Food and Drug Administration within 1 year. After submission, hopes are high for a swift approval, he said.
"If we can validate the test in a set of U.S. samples, we believe it could be approved in less than 2 years," Dr. Sjögren said in an interview. "We hope to be able to study it in samples from the Alzheimer’s Disease Neuroimaging Initiative [ADNI]."
A proof-of-concept study based on a 20-gene version had a 74% accuracy rate in identifying cases of mild cognitive impairment (MCI) that converted to Alzheimer’s dementia over a 2-year period. The study, conducted in 129 subjects, was presented at the 2011 Conference of Clinical Trials on Alzheimer's Disease (Poster P10).
Two versions of the gene panel have now been validated in two separate cohorts.
The first group consisted of 100 subjects: 50 with stable MCI, 25 with confirmed Alzheimer’s, and 25 age-matched healthy controls. They were followed for 2 years. In this group with 2 years of follow-up, the 20-gene panel successfully predicted prodromal Alzheimer’s with 82% accuracy, 80% sensitivity, and 84% specificity.
It was retested in another unique group of 75 subjects with MCI that converted to Alzheimer’s. In this 2-year study, the test successfully predicted conversion 70% of the time, with 69% sensitivity and 71% specificity.
Since then, the company has added five additional genes to the testing panel. When the new test was applied to this same group of 75 subjects, the overall accuracy rose to 81%, with a sensitivity of 77% and a specificity of 85%.
The test hasn’t been validated against any other diagnostic methods, including brain imaging or biomarkers, Dr. Sjögren said.
The original test to help in making a diagnosis of Alzheimer’s, which is approved in Europe, costs about $500, Dr. Sjögren said. He didn’t comment on the possible cost of a test that could identify cases of MCI/prodromal Alzheimer’s.
In Europe, he added, the Alzheimer’s test is used in physicians’ offices and provides results in a few days. Doctors and patients can then decide together which confirmatory tests to pursue.
The MCI conversion blood test, if approved, could be similarly used. "I believe it would be more economical than cerebrospinal fluid collection and certainly less invasive," he said.
VANCOUVER, B.C. – An investigational gene-based blood test identified prodromal Alzheimer’s disease with an accuracy of 81%.
The test is based on a panel of 25 genes active in biological processes associated with Alzheimer’s, Dr. Magnus Sjögren said at the Alzheimer’s Association International Conference 2012.
The genes have functions that include regulating the processing of amyloid and tau, and are involved as well in mitochondrial transport and function, neuronal and brain function, inflammatory processes, and oxidative stress, said Dr. Sjögren, chief medical officer of DiaGenic, the Norwegian company that is developing the test.
Under development in Oslo, the test could be submitted to the U.S. Food and Drug Administration within 1 year. After submission, hopes are high for a swift approval, he said.
"If we can validate the test in a set of U.S. samples, we believe it could be approved in less than 2 years," Dr. Sjögren said in an interview. "We hope to be able to study it in samples from the Alzheimer’s Disease Neuroimaging Initiative [ADNI]."
A proof-of-concept study based on a 20-gene version had a 74% accuracy rate in identifying cases of mild cognitive impairment (MCI) that converted to Alzheimer’s dementia over a 2-year period. The study, conducted in 129 subjects, was presented at the 2011 Conference of Clinical Trials on Alzheimer's Disease (Poster P10).
Two versions of the gene panel have now been validated in two separate cohorts.
The first group consisted of 100 subjects: 50 with stable MCI, 25 with confirmed Alzheimer’s, and 25 age-matched healthy controls. They were followed for 2 years. In this group with 2 years of follow-up, the 20-gene panel successfully predicted prodromal Alzheimer’s with 82% accuracy, 80% sensitivity, and 84% specificity.
It was retested in another unique group of 75 subjects with MCI that converted to Alzheimer’s. In this 2-year study, the test successfully predicted conversion 70% of the time, with 69% sensitivity and 71% specificity.
Since then, the company has added five additional genes to the testing panel. When the new test was applied to this same group of 75 subjects, the overall accuracy rose to 81%, with a sensitivity of 77% and a specificity of 85%.
The test hasn’t been validated against any other diagnostic methods, including brain imaging or biomarkers, Dr. Sjögren said.
The original test to help in making a diagnosis of Alzheimer’s, which is approved in Europe, costs about $500, Dr. Sjögren said. He didn’t comment on the possible cost of a test that could identify cases of MCI/prodromal Alzheimer’s.
In Europe, he added, the Alzheimer’s test is used in physicians’ offices and provides results in a few days. Doctors and patients can then decide together which confirmatory tests to pursue.
The MCI conversion blood test, if approved, could be similarly used. "I believe it would be more economical than cerebrospinal fluid collection and certainly less invasive," he said.
VANCOUVER, B.C. – An investigational gene-based blood test identified prodromal Alzheimer’s disease with an accuracy of 81%.
The test is based on a panel of 25 genes active in biological processes associated with Alzheimer’s, Dr. Magnus Sjögren said at the Alzheimer’s Association International Conference 2012.
The genes have functions that include regulating the processing of amyloid and tau, and are involved as well in mitochondrial transport and function, neuronal and brain function, inflammatory processes, and oxidative stress, said Dr. Sjögren, chief medical officer of DiaGenic, the Norwegian company that is developing the test.
Under development in Oslo, the test could be submitted to the U.S. Food and Drug Administration within 1 year. After submission, hopes are high for a swift approval, he said.
"If we can validate the test in a set of U.S. samples, we believe it could be approved in less than 2 years," Dr. Sjögren said in an interview. "We hope to be able to study it in samples from the Alzheimer’s Disease Neuroimaging Initiative [ADNI]."
A proof-of-concept study based on a 20-gene version had a 74% accuracy rate in identifying cases of mild cognitive impairment (MCI) that converted to Alzheimer’s dementia over a 2-year period. The study, conducted in 129 subjects, was presented at the 2011 Conference of Clinical Trials on Alzheimer's Disease (Poster P10).
Two versions of the gene panel have now been validated in two separate cohorts.
The first group consisted of 100 subjects: 50 with stable MCI, 25 with confirmed Alzheimer’s, and 25 age-matched healthy controls. They were followed for 2 years. In this group with 2 years of follow-up, the 20-gene panel successfully predicted prodromal Alzheimer’s with 82% accuracy, 80% sensitivity, and 84% specificity.
It was retested in another unique group of 75 subjects with MCI that converted to Alzheimer’s. In this 2-year study, the test successfully predicted conversion 70% of the time, with 69% sensitivity and 71% specificity.
Since then, the company has added five additional genes to the testing panel. When the new test was applied to this same group of 75 subjects, the overall accuracy rose to 81%, with a sensitivity of 77% and a specificity of 85%.
The test hasn’t been validated against any other diagnostic methods, including brain imaging or biomarkers, Dr. Sjögren said.
The original test to help in making a diagnosis of Alzheimer’s, which is approved in Europe, costs about $500, Dr. Sjögren said. He didn’t comment on the possible cost of a test that could identify cases of MCI/prodromal Alzheimer’s.
In Europe, he added, the Alzheimer’s test is used in physicians’ offices and provides results in a few days. Doctors and patients can then decide together which confirmatory tests to pursue.
The MCI conversion blood test, if approved, could be similarly used. "I believe it would be more economical than cerebrospinal fluid collection and certainly less invasive," he said.
AT THE ALZHEIMER'S ASSOCIATION INTERNATIONAL CONFERENCE 2012
Major Finding: An investigational blood test can predict with 81% accuracy which patients with mild cognitive impairment will convert to Alzheimer’s dementia within 2 years.
Data Source: The test has been validated in three 2-year, prospective studies on more than 200 subjects.
Disclosures: Dr. Sjögren is the chief medical officer of DiaGenic, the Norwegian company that is developing the test.
Sleep Problems May Increase Risk for Dementia
VANCOUVER, B.C. – Disordered sleep in mid- to late life is associated with an increased risk for future cognitive impairment and may alter the dynamics of the Alzheimer’s disease–associated protein amyloid-beta, according to several studies.
It’s important that physicians recognize disordered sleep as a modifiable risk factor, Dr. Kristine Yaffe said in an interview.
"Please attend to sleep hygiene in your elderly patients," said Dr. Yaffe, director of the memory disorders clinic at the San Francisco VA Medical Center. "Some people think that 50% of elderly people have some kind of sleep complaint. They are common and they are treatable."
Dr. Yaffe and her colleagues studied 1,309 elderly women who completed several days of sleep observation as part of a 15-year longitudinal study. They measured the motor activity of the women (mean age, 82 years) during sleep via wrist actigraphy. All of the participants had several neuropsychological evaluations and cognitive measurements during the study. A subset of 298 patients also underwent polysomnography.
After 5 years, women with sleep-disordered breathing were twice as likely to develop mild cognitive impairment or dementia as were those who slept normally. There was a similar risk level for women who had delayed sleep acrophase – difficulty falling asleep before the early morning hours and trouble waking up before late morning or early afternoon.
Women with greater nighttime wakefulness were more than twice as likely to show impaired global cognitive functioning, and twice as likely to have delayed verbal recall.
"We already know that sleep deprivation and abnormal sleep patterns are associated with falls and increased morbidity and mortality," said Dr. Yaffe, who also is a professor in the departments of psychiatry, neurology, epidemiology, and biostatistics at the University of California, San Francisco. "This is the first study showing that disordered sleep is a risk factor for later cognitive problems.
"It’s important for clinicians to check for sleep problems and excessive daytime sleepiness as a possibly treatable cause of later cognitive problems," she said at the Alzheimer’s Disease International Conference 2012. "Sleep habits are as important in prevention of dementia as diabetes and obesity."
Elizabeth Devore, Sc.D., of Brigham and Women’s Hospital, Boston, found similar results in her analysis of sleep data extracted from the longitudinal Nurses’ Health Study. The sleep study included information on 15,263 women, who were aged 70 or older at the time of their first cognitive evaluation. These subjects were followed for 6 years with cognitive testing every other year.
The women reported their sleep duration and quality at ages 40-65 and at ages 54-79. A normal night’s sleep was considered to be 7 hours.
When comparing sleep duration to later cognitive status, Dr. Devore found that:
• Those who slept 5 hours per day or less had lower average cognition than those who slept 7 hours per day.
• Those who slept 9 hours per day or more also had lower average cognition than those who slept 7 hours per day.
• Too little or too much sleep was cognitively equivalent to 2 years of aging.
When the researchers evaluated the effects of change in sleep duration from mid- to later life, they observed that women whose sleep changed by 2 hours per day or more had worse cognitive function than those with no change in sleep duration, independent of their initial sleep duration.
"Either too little or too much sleep, and sleep duration changes over time, might contribute to cognitive decline in older adults," she said. "This is up-and-coming research and has great implications for public health. We need to do more research in this area, especially looking at sleep duration over the course of life. It’s simple, and down the road it could lead to the development of sleep and circadian rhythm strategies to target therapy."
Dr. Devore also found that shortened or extended sleep patterns were also entwined with abnormalities in the processing of amyloid-beta (Abeta) peptides, which are believed to cause neuronal dysfunction in Alzheimer’s disease. Subjects with these extremes of sleep duration showed a skewed proportion of Abeta-40 and Abeta-42 peptides in cerebrospinal fluid, suggesting that more amyloid could be accruing in brain plaques.
Dr. Yafei Huang and her associates at Washington University, St. Louis, also found that the circadian pattern of Abeta secretion is altered in Alzheimer’s disease. Her study involved three groups: 12 Alzheimer’s patients with a mean age of 72 years, 8 age-matched controls, and 10 young, healthy controls with a mean age of 36 years.
Each subject underwent hourly sampling of cerebrospinal fluid (CSF) and plasma for 36 hours during waking and sleep times. Dr. Huang plotted the circadian secretion patterns of Abeta-40 and Abeta-42, and the amyloid precursor protein (APP).
While there were no significant time-linked associations of Abeta in plasma, there were differences in the CSF.
Overall, both Abeta-40 and -42 in CSF showed a linear increase during waking time, and dips during sleep. The age-matched controls and healthy young subjects – who were presumably amyloid negative – had a steady increase in APP, Abeta-40, and Abeta-42 during waking times. But this finding was absent in subjects with Alzheimer’s.
"In this group, the Abeta-40 increased much less, and the change in Abeta-42 was almost absent," Dr. Huang said.
This suggests that amyloid processing is impaired in patients with the disease. Different enzymes split the APP molecule into the benign Abeta-40, which is secreted into the CSF, and the toxic Abeta-42, which forms brain plaques. A low CSF Abeta-42 level indicates abnormal APP cleavage, leading to retained Abeta-42.
Some studies have suggested that hypoxia alters the enzymatic cleavage of APP, allowing more Abeta-42 production. This finding could have implications in people who experience years of sleep apnea, said Dr. Constantine Lykestos, who moderated the press briefing where the studies were presented.
"There is emerging evidence that hypoxia is associated with amyloidosis, but it’s too new a theory to really know," said Dr. Lykestos, director of the memory and Alzheimer’s treatment center at Johns Hopkins University, Baltimore. "The jury is still out on this relationship."
Dr. Yaffe, Dr. Huang, and Dr. Devore reported no relevant financial disclosures.
VANCOUVER, B.C. – Disordered sleep in mid- to late life is associated with an increased risk for future cognitive impairment and may alter the dynamics of the Alzheimer’s disease–associated protein amyloid-beta, according to several studies.
It’s important that physicians recognize disordered sleep as a modifiable risk factor, Dr. Kristine Yaffe said in an interview.
"Please attend to sleep hygiene in your elderly patients," said Dr. Yaffe, director of the memory disorders clinic at the San Francisco VA Medical Center. "Some people think that 50% of elderly people have some kind of sleep complaint. They are common and they are treatable."
Dr. Yaffe and her colleagues studied 1,309 elderly women who completed several days of sleep observation as part of a 15-year longitudinal study. They measured the motor activity of the women (mean age, 82 years) during sleep via wrist actigraphy. All of the participants had several neuropsychological evaluations and cognitive measurements during the study. A subset of 298 patients also underwent polysomnography.
After 5 years, women with sleep-disordered breathing were twice as likely to develop mild cognitive impairment or dementia as were those who slept normally. There was a similar risk level for women who had delayed sleep acrophase – difficulty falling asleep before the early morning hours and trouble waking up before late morning or early afternoon.
Women with greater nighttime wakefulness were more than twice as likely to show impaired global cognitive functioning, and twice as likely to have delayed verbal recall.
"We already know that sleep deprivation and abnormal sleep patterns are associated with falls and increased morbidity and mortality," said Dr. Yaffe, who also is a professor in the departments of psychiatry, neurology, epidemiology, and biostatistics at the University of California, San Francisco. "This is the first study showing that disordered sleep is a risk factor for later cognitive problems.
"It’s important for clinicians to check for sleep problems and excessive daytime sleepiness as a possibly treatable cause of later cognitive problems," she said at the Alzheimer’s Disease International Conference 2012. "Sleep habits are as important in prevention of dementia as diabetes and obesity."
Elizabeth Devore, Sc.D., of Brigham and Women’s Hospital, Boston, found similar results in her analysis of sleep data extracted from the longitudinal Nurses’ Health Study. The sleep study included information on 15,263 women, who were aged 70 or older at the time of their first cognitive evaluation. These subjects were followed for 6 years with cognitive testing every other year.
The women reported their sleep duration and quality at ages 40-65 and at ages 54-79. A normal night’s sleep was considered to be 7 hours.
When comparing sleep duration to later cognitive status, Dr. Devore found that:
• Those who slept 5 hours per day or less had lower average cognition than those who slept 7 hours per day.
• Those who slept 9 hours per day or more also had lower average cognition than those who slept 7 hours per day.
• Too little or too much sleep was cognitively equivalent to 2 years of aging.
When the researchers evaluated the effects of change in sleep duration from mid- to later life, they observed that women whose sleep changed by 2 hours per day or more had worse cognitive function than those with no change in sleep duration, independent of their initial sleep duration.
"Either too little or too much sleep, and sleep duration changes over time, might contribute to cognitive decline in older adults," she said. "This is up-and-coming research and has great implications for public health. We need to do more research in this area, especially looking at sleep duration over the course of life. It’s simple, and down the road it could lead to the development of sleep and circadian rhythm strategies to target therapy."
Dr. Devore also found that shortened or extended sleep patterns were also entwined with abnormalities in the processing of amyloid-beta (Abeta) peptides, which are believed to cause neuronal dysfunction in Alzheimer’s disease. Subjects with these extremes of sleep duration showed a skewed proportion of Abeta-40 and Abeta-42 peptides in cerebrospinal fluid, suggesting that more amyloid could be accruing in brain plaques.
Dr. Yafei Huang and her associates at Washington University, St. Louis, also found that the circadian pattern of Abeta secretion is altered in Alzheimer’s disease. Her study involved three groups: 12 Alzheimer’s patients with a mean age of 72 years, 8 age-matched controls, and 10 young, healthy controls with a mean age of 36 years.
Each subject underwent hourly sampling of cerebrospinal fluid (CSF) and plasma for 36 hours during waking and sleep times. Dr. Huang plotted the circadian secretion patterns of Abeta-40 and Abeta-42, and the amyloid precursor protein (APP).
While there were no significant time-linked associations of Abeta in plasma, there were differences in the CSF.
Overall, both Abeta-40 and -42 in CSF showed a linear increase during waking time, and dips during sleep. The age-matched controls and healthy young subjects – who were presumably amyloid negative – had a steady increase in APP, Abeta-40, and Abeta-42 during waking times. But this finding was absent in subjects with Alzheimer’s.
"In this group, the Abeta-40 increased much less, and the change in Abeta-42 was almost absent," Dr. Huang said.
This suggests that amyloid processing is impaired in patients with the disease. Different enzymes split the APP molecule into the benign Abeta-40, which is secreted into the CSF, and the toxic Abeta-42, which forms brain plaques. A low CSF Abeta-42 level indicates abnormal APP cleavage, leading to retained Abeta-42.
Some studies have suggested that hypoxia alters the enzymatic cleavage of APP, allowing more Abeta-42 production. This finding could have implications in people who experience years of sleep apnea, said Dr. Constantine Lykestos, who moderated the press briefing where the studies were presented.
"There is emerging evidence that hypoxia is associated with amyloidosis, but it’s too new a theory to really know," said Dr. Lykestos, director of the memory and Alzheimer’s treatment center at Johns Hopkins University, Baltimore. "The jury is still out on this relationship."
Dr. Yaffe, Dr. Huang, and Dr. Devore reported no relevant financial disclosures.
VANCOUVER, B.C. – Disordered sleep in mid- to late life is associated with an increased risk for future cognitive impairment and may alter the dynamics of the Alzheimer’s disease–associated protein amyloid-beta, according to several studies.
It’s important that physicians recognize disordered sleep as a modifiable risk factor, Dr. Kristine Yaffe said in an interview.
"Please attend to sleep hygiene in your elderly patients," said Dr. Yaffe, director of the memory disorders clinic at the San Francisco VA Medical Center. "Some people think that 50% of elderly people have some kind of sleep complaint. They are common and they are treatable."
Dr. Yaffe and her colleagues studied 1,309 elderly women who completed several days of sleep observation as part of a 15-year longitudinal study. They measured the motor activity of the women (mean age, 82 years) during sleep via wrist actigraphy. All of the participants had several neuropsychological evaluations and cognitive measurements during the study. A subset of 298 patients also underwent polysomnography.
After 5 years, women with sleep-disordered breathing were twice as likely to develop mild cognitive impairment or dementia as were those who slept normally. There was a similar risk level for women who had delayed sleep acrophase – difficulty falling asleep before the early morning hours and trouble waking up before late morning or early afternoon.
Women with greater nighttime wakefulness were more than twice as likely to show impaired global cognitive functioning, and twice as likely to have delayed verbal recall.
"We already know that sleep deprivation and abnormal sleep patterns are associated with falls and increased morbidity and mortality," said Dr. Yaffe, who also is a professor in the departments of psychiatry, neurology, epidemiology, and biostatistics at the University of California, San Francisco. "This is the first study showing that disordered sleep is a risk factor for later cognitive problems.
"It’s important for clinicians to check for sleep problems and excessive daytime sleepiness as a possibly treatable cause of later cognitive problems," she said at the Alzheimer’s Disease International Conference 2012. "Sleep habits are as important in prevention of dementia as diabetes and obesity."
Elizabeth Devore, Sc.D., of Brigham and Women’s Hospital, Boston, found similar results in her analysis of sleep data extracted from the longitudinal Nurses’ Health Study. The sleep study included information on 15,263 women, who were aged 70 or older at the time of their first cognitive evaluation. These subjects were followed for 6 years with cognitive testing every other year.
The women reported their sleep duration and quality at ages 40-65 and at ages 54-79. A normal night’s sleep was considered to be 7 hours.
When comparing sleep duration to later cognitive status, Dr. Devore found that:
• Those who slept 5 hours per day or less had lower average cognition than those who slept 7 hours per day.
• Those who slept 9 hours per day or more also had lower average cognition than those who slept 7 hours per day.
• Too little or too much sleep was cognitively equivalent to 2 years of aging.
When the researchers evaluated the effects of change in sleep duration from mid- to later life, they observed that women whose sleep changed by 2 hours per day or more had worse cognitive function than those with no change in sleep duration, independent of their initial sleep duration.
"Either too little or too much sleep, and sleep duration changes over time, might contribute to cognitive decline in older adults," she said. "This is up-and-coming research and has great implications for public health. We need to do more research in this area, especially looking at sleep duration over the course of life. It’s simple, and down the road it could lead to the development of sleep and circadian rhythm strategies to target therapy."
Dr. Devore also found that shortened or extended sleep patterns were also entwined with abnormalities in the processing of amyloid-beta (Abeta) peptides, which are believed to cause neuronal dysfunction in Alzheimer’s disease. Subjects with these extremes of sleep duration showed a skewed proportion of Abeta-40 and Abeta-42 peptides in cerebrospinal fluid, suggesting that more amyloid could be accruing in brain plaques.
Dr. Yafei Huang and her associates at Washington University, St. Louis, also found that the circadian pattern of Abeta secretion is altered in Alzheimer’s disease. Her study involved three groups: 12 Alzheimer’s patients with a mean age of 72 years, 8 age-matched controls, and 10 young, healthy controls with a mean age of 36 years.
Each subject underwent hourly sampling of cerebrospinal fluid (CSF) and plasma for 36 hours during waking and sleep times. Dr. Huang plotted the circadian secretion patterns of Abeta-40 and Abeta-42, and the amyloid precursor protein (APP).
While there were no significant time-linked associations of Abeta in plasma, there were differences in the CSF.
Overall, both Abeta-40 and -42 in CSF showed a linear increase during waking time, and dips during sleep. The age-matched controls and healthy young subjects – who were presumably amyloid negative – had a steady increase in APP, Abeta-40, and Abeta-42 during waking times. But this finding was absent in subjects with Alzheimer’s.
"In this group, the Abeta-40 increased much less, and the change in Abeta-42 was almost absent," Dr. Huang said.
This suggests that amyloid processing is impaired in patients with the disease. Different enzymes split the APP molecule into the benign Abeta-40, which is secreted into the CSF, and the toxic Abeta-42, which forms brain plaques. A low CSF Abeta-42 level indicates abnormal APP cleavage, leading to retained Abeta-42.
Some studies have suggested that hypoxia alters the enzymatic cleavage of APP, allowing more Abeta-42 production. This finding could have implications in people who experience years of sleep apnea, said Dr. Constantine Lykestos, who moderated the press briefing where the studies were presented.
"There is emerging evidence that hypoxia is associated with amyloidosis, but it’s too new a theory to really know," said Dr. Lykestos, director of the memory and Alzheimer’s treatment center at Johns Hopkins University, Baltimore. "The jury is still out on this relationship."
Dr. Yaffe, Dr. Huang, and Dr. Devore reported no relevant financial disclosures.
AT THE ALZHEIMER'S ASSOCIATION INTERNATIONAL CONFERENCE 2012
Virtual Program Has Real-Life Cognitive Impact
VANCOUVER, B.C. – An online community-wide healthy aging program has increased cognitive screening in physicians’ offices by 50% in 18 months, targeting the population so well that half of participants show cognitive impairment when clinically evaluated.
Since the Orange County (Calif.) Vital Aging Program debuted in September 2010, it has linked more than 6,000 residents to physicians for cognitive assessments – an average of 400 evaluations per month, Dr. William Shankle said at the Alzheimer’s Association International Conference 2012.
“Many people with cognitive impairment or early dementia are not seeing a physician, and for various reasons,” said Dr. Shankle, director of the memory and cognitive disorders unit at Hoag Neurosciences Institute in Newport Beach, Calif. “Most don’t think cognitive impairment is treatable, and most associate memory loss with Alzheimer’s disease.”
That association provokes a deep fear, putting many in a state of denial and dampening their desire to reach out to physicians. The ability to self-screen online with a survey based on clinically validated questions can ease that fear – about half of those who take the survey discover that their memory problems are simply the result of normal aging. For the other half, the self-assessment suggests that a formal evaluation can get them the help they need to deal proactively with a cognitive problem, Dr. Shankle said.
If a screen comes back positive, residents can search the Vital Aging website for a physician close to them who has the special training to help. The program also reaches out to the greater community of those without memory concerns. Visitors can explore educational links, sign up for a monthly newsletter, and find out about public lectures focusing on brain health and lifestyle changes that can optimize it.
In fact, a preliminary assessment shows that people who take the memory evaluation, attend a lecture, and then follow through with the suggested changes perform better on memory tests at a follow-up assessment 1 year later.
“We’ve seen that 75% of people who attend these lectures do go back to their doctors for a 1-year follow-up visit, which is great,” Dr. Shankle said. “Participants are very willing to educate themselves, make these positive lifestyle changes, and manage the risk factors that can influence their cognitive health.” For physicians, the program provides a portal to specially focused continuing medical education, he said. “These CME activities are differentiated to dig down to the individual physician’s level of knowledge and start there, or to provide more advanced training to those who already have knowledge of the issues.”
Community programs like these are one way to help manage the money pit that is dementia care. “Dementia costs a lot of money. On average, Medicare pays three times more per year to care for a dementia patient than a patient who doesn’t have it.
This creates an urgent need to address cognitive health care in a community – or this will bankrupt medical care. We have a system that is broken. This is one way to begin to fix it,” he said. Dr. Shankle disclosed that he is an employee of and holds stock in Medical Care Corp., which supplied some of the technology used in the Orange County Vital Aging Project.
VANCOUVER, B.C. – An online community-wide healthy aging program has increased cognitive screening in physicians’ offices by 50% in 18 months, targeting the population so well that half of participants show cognitive impairment when clinically evaluated.
Since the Orange County (Calif.) Vital Aging Program debuted in September 2010, it has linked more than 6,000 residents to physicians for cognitive assessments – an average of 400 evaluations per month, Dr. William Shankle said at the Alzheimer’s Association International Conference 2012.
“Many people with cognitive impairment or early dementia are not seeing a physician, and for various reasons,” said Dr. Shankle, director of the memory and cognitive disorders unit at Hoag Neurosciences Institute in Newport Beach, Calif. “Most don’t think cognitive impairment is treatable, and most associate memory loss with Alzheimer’s disease.”
That association provokes a deep fear, putting many in a state of denial and dampening their desire to reach out to physicians. The ability to self-screen online with a survey based on clinically validated questions can ease that fear – about half of those who take the survey discover that their memory problems are simply the result of normal aging. For the other half, the self-assessment suggests that a formal evaluation can get them the help they need to deal proactively with a cognitive problem, Dr. Shankle said.
If a screen comes back positive, residents can search the Vital Aging website for a physician close to them who has the special training to help. The program also reaches out to the greater community of those without memory concerns. Visitors can explore educational links, sign up for a monthly newsletter, and find out about public lectures focusing on brain health and lifestyle changes that can optimize it.
In fact, a preliminary assessment shows that people who take the memory evaluation, attend a lecture, and then follow through with the suggested changes perform better on memory tests at a follow-up assessment 1 year later.
“We’ve seen that 75% of people who attend these lectures do go back to their doctors for a 1-year follow-up visit, which is great,” Dr. Shankle said. “Participants are very willing to educate themselves, make these positive lifestyle changes, and manage the risk factors that can influence their cognitive health.” For physicians, the program provides a portal to specially focused continuing medical education, he said. “These CME activities are differentiated to dig down to the individual physician’s level of knowledge and start there, or to provide more advanced training to those who already have knowledge of the issues.”
Community programs like these are one way to help manage the money pit that is dementia care. “Dementia costs a lot of money. On average, Medicare pays three times more per year to care for a dementia patient than a patient who doesn’t have it.
This creates an urgent need to address cognitive health care in a community – or this will bankrupt medical care. We have a system that is broken. This is one way to begin to fix it,” he said. Dr. Shankle disclosed that he is an employee of and holds stock in Medical Care Corp., which supplied some of the technology used in the Orange County Vital Aging Project.
VANCOUVER, B.C. – An online community-wide healthy aging program has increased cognitive screening in physicians’ offices by 50% in 18 months, targeting the population so well that half of participants show cognitive impairment when clinically evaluated.
Since the Orange County (Calif.) Vital Aging Program debuted in September 2010, it has linked more than 6,000 residents to physicians for cognitive assessments – an average of 400 evaluations per month, Dr. William Shankle said at the Alzheimer’s Association International Conference 2012.
“Many people with cognitive impairment or early dementia are not seeing a physician, and for various reasons,” said Dr. Shankle, director of the memory and cognitive disorders unit at Hoag Neurosciences Institute in Newport Beach, Calif. “Most don’t think cognitive impairment is treatable, and most associate memory loss with Alzheimer’s disease.”
That association provokes a deep fear, putting many in a state of denial and dampening their desire to reach out to physicians. The ability to self-screen online with a survey based on clinically validated questions can ease that fear – about half of those who take the survey discover that their memory problems are simply the result of normal aging. For the other half, the self-assessment suggests that a formal evaluation can get them the help they need to deal proactively with a cognitive problem, Dr. Shankle said.
If a screen comes back positive, residents can search the Vital Aging website for a physician close to them who has the special training to help. The program also reaches out to the greater community of those without memory concerns. Visitors can explore educational links, sign up for a monthly newsletter, and find out about public lectures focusing on brain health and lifestyle changes that can optimize it.
In fact, a preliminary assessment shows that people who take the memory evaluation, attend a lecture, and then follow through with the suggested changes perform better on memory tests at a follow-up assessment 1 year later.
“We’ve seen that 75% of people who attend these lectures do go back to their doctors for a 1-year follow-up visit, which is great,” Dr. Shankle said. “Participants are very willing to educate themselves, make these positive lifestyle changes, and manage the risk factors that can influence their cognitive health.” For physicians, the program provides a portal to specially focused continuing medical education, he said. “These CME activities are differentiated to dig down to the individual physician’s level of knowledge and start there, or to provide more advanced training to those who already have knowledge of the issues.”
Community programs like these are one way to help manage the money pit that is dementia care. “Dementia costs a lot of money. On average, Medicare pays three times more per year to care for a dementia patient than a patient who doesn’t have it.
This creates an urgent need to address cognitive health care in a community – or this will bankrupt medical care. We have a system that is broken. This is one way to begin to fix it,” he said. Dr. Shankle disclosed that he is an employee of and holds stock in Medical Care Corp., which supplied some of the technology used in the Orange County Vital Aging Project.
AT THE ALZHEIMER’S ASSOCIATION INTERNATIONAL CONFERENCE 2012
Major Finding: An online cognitive assessment program has increased formal memory evaluations by 50% in 18 months, linking more than 6,000 residents to physicians who specialize in dementia care.
Data Source: The Orange County Vital Aging Project is an online tool that educates community residents and physicians on memory, cognition, and dementia.
Disclosures: Dr. Shankle disclosed that he is an employee of and holds stock in Medical Care Corp., which supplied some of the technology used in the Orange County Vital Aging Project.
Studies Clarify the Mental Benefits of Exercise in Aging
VANCOUVER, B.C. – Physical activity improves not only the heart and lungs, but the brain as well, providing a triad of benefits: improved cognition, better physiologic functioning, and increased brain volume and hints of revved-up neuronal growth.
Studies presented July 15 at the Alzheimer’s Association International Conference 2012 agreed: Getting active is essential for preserving cognitive health.
Exactly how exercise boosts brain function is still a matter of debate, Teresa Liu-Ambrose, Ph.D., said in an interview.
"In addition to getting the physical benefits of exercise, people are also getting social interaction and mental stimulation," said Dr. Liu-Ambrose, director of the Aging, Mobility, and Cognitive Neuroscience Lab at the University of British Columbia, Vancouver. Group activities, like exercise, can also help unwind the tangle of diminished executive function and social isolation, a damaging pair.
"When executive function begins to deteriorate, people know that they are losing some of their ability to interact, and so they withdraw and become isolated," she said. The lack of social interaction contributes to the downward slide of cognition.
Dr. Liu-Ambrose was the primary investigator of a randomized controlled trial that examined the benefits of three different exercise regimens, each conducted twice a week for 6 months: resistance training; aerobic training; and balance and tone training, which served as the comparator group.
The subjects were 86 women aged 70-80 years. They all still lived at home, but had probable mild cognitive impairment. The group is an important one because the natural history of Alzheimer’s disease suggests that about half of those who develop mild cognitive impairment will go on to develop Alzheimer’s dementia.
All of the programs were progressive, she said. Those in the weight program started with the largest weights they could handle with good form, which were increased as soon as the women exceeded the required number of repetitions. The aerobic group increased their walking speed based on heart rate and perceived exertion. The control group loaded their exercises as well, increasing repetitions and stretching as the trial progressed.
After 6 months, the strength training group had improved by 17% over baseline on measures of executive function, which was significantly better than either of the other groups. These women also experienced significant gains in associative memory.
Dr. Liu-Ambrose also saw physiologic correlates to the cognitive findings. Functional MRI showed improved blood flow in three brain regions involved with encoding and memorization of nonverbal associations: the right lingual gyrus, the occipital fusiform gyrus, and the right frontal pole.
The aerobic training group improved in balance, mobility, and cardiovascular capacity, but did not have the same kind of cognitive or brain physiologic gains, she added. The balance and tone group maintained their cognitive ability without any additional decline, but didn’t experience any additional benefit either.
"I want to stress that we were seeing a real improvement with resistance training – not just maintenance like we did in the control group. We’re talking about women who already have some level of cognitive impairment. The most positive way of looking at this is that exercise could be a magic bullet. There is evidence now that exercise may not only be preventive – but a potential treatment."
In a separate study, Kirk Erickson, Ph.D., presented evidence of the molecular underpinning of exercise-induced brain volume changes: Physical activity, he said, stimulates the release of molecules that promote neuronal growth.
Dr. Erickson, a cognitive neuroscience researcher at the University of Pittsburgh, described the results of a walking program that involved 120 cognitively healthy, elderly adults.
The participants had been sedentary for at least the previous 6 months. They were randomized either to a 1-year program of moderately intense walking or to a 1-year stretching and toning group. At baseline and at 1 year, Dr. Erickson and his colleagues measured brain volume and levels of brain-derived neurotrophic factor (BDNF).
BDNF is a protein that affects neurons in several ways: It supports healthy neuronal function, encourages stem cells to differentiate into neurons, and stimulates dendritic expansion. BDNF is particularly active in regions associated with memory and executive functioning.
At the study’s end, Dr. Erickson and his associates found that those in the walking program had about a 2% increase in the size of the anterior hippocampus, compared with the stretching and toning group. Increases in BDNF positively correlated with the volume change. He also saw increases in the volume of the prefrontal cortex, which correlated with increased cardiorespiratory fitness.
Just how to translate these controlled experimental situations into effective, real-life practice isn’t completely clear, Dr. Liu-Ambrose said, especially when patients already have difficulty with planning and prioritization.
Her colleague, Nader Fallah, Ph.D., also of the University of British Columbia, used his expertise in biostatistics to explore that problem. Using a similar cohort of women, he found that those with higher baseline cognitive function reaped the greatest benefit from resistance training.
Dr. Fallah conducted a secondary analysis of a 12-month, randomized, controlled trial of 155 women aged 65-75 years (Neurobiol. Aging 2012;33:1690-8). The women were assigned to resistance training or balance and tone training.
This new analysis found that women with lower baseline functioning not only experienced less improvement from exercise, but also responded equally to both resistance training and balance and tone training.
"To our knowledge, this is the first study to demonstrate that an individual’s baseline self-regulatory capacity impacts the amount of cognitive benefit the person will reap from targeted exercise training," Dr. Fallah said in a written statement.
Dr. Liu-Ambrose said the study sheds valuable light on the concept of personalizing exercise prescriptions.
"I think this brings us another dimension to consider," Dr. Liu-Ambrose said. "When we prescribe exercise, most of the time we focus on physical ability: ‘Can the knees take it, what is the cardiovascular state?’ But people who have better executive function at baseline can execute this prescription better. They can plan their time, prioritize their activities, and follow through with the commitment."
Dr. Fallah’s findings suggest that "cognitive priming" might improve outcomes in prescribed exercise. Computerized brain training games are one thing to consider. They’ve been shown to improve memory and executive function for both cognitively healthy and impaired older adults. Just a small improvement in executive function and memory could be enough to get a patient invested in an exercise program.
"But for those with more pronounced dementia, it’s really about engaging the caregiver," Dr. Liu-Ambrose concluded. "We need to give them the tools they need to implement this as part of the daily lives of their loved ones."
None of the researchers reported having any financial conflicts of interest.
VANCOUVER, B.C. – Physical activity improves not only the heart and lungs, but the brain as well, providing a triad of benefits: improved cognition, better physiologic functioning, and increased brain volume and hints of revved-up neuronal growth.
Studies presented July 15 at the Alzheimer’s Association International Conference 2012 agreed: Getting active is essential for preserving cognitive health.
Exactly how exercise boosts brain function is still a matter of debate, Teresa Liu-Ambrose, Ph.D., said in an interview.
"In addition to getting the physical benefits of exercise, people are also getting social interaction and mental stimulation," said Dr. Liu-Ambrose, director of the Aging, Mobility, and Cognitive Neuroscience Lab at the University of British Columbia, Vancouver. Group activities, like exercise, can also help unwind the tangle of diminished executive function and social isolation, a damaging pair.
"When executive function begins to deteriorate, people know that they are losing some of their ability to interact, and so they withdraw and become isolated," she said. The lack of social interaction contributes to the downward slide of cognition.
Dr. Liu-Ambrose was the primary investigator of a randomized controlled trial that examined the benefits of three different exercise regimens, each conducted twice a week for 6 months: resistance training; aerobic training; and balance and tone training, which served as the comparator group.
The subjects were 86 women aged 70-80 years. They all still lived at home, but had probable mild cognitive impairment. The group is an important one because the natural history of Alzheimer’s disease suggests that about half of those who develop mild cognitive impairment will go on to develop Alzheimer’s dementia.
All of the programs were progressive, she said. Those in the weight program started with the largest weights they could handle with good form, which were increased as soon as the women exceeded the required number of repetitions. The aerobic group increased their walking speed based on heart rate and perceived exertion. The control group loaded their exercises as well, increasing repetitions and stretching as the trial progressed.
After 6 months, the strength training group had improved by 17% over baseline on measures of executive function, which was significantly better than either of the other groups. These women also experienced significant gains in associative memory.
Dr. Liu-Ambrose also saw physiologic correlates to the cognitive findings. Functional MRI showed improved blood flow in three brain regions involved with encoding and memorization of nonverbal associations: the right lingual gyrus, the occipital fusiform gyrus, and the right frontal pole.
The aerobic training group improved in balance, mobility, and cardiovascular capacity, but did not have the same kind of cognitive or brain physiologic gains, she added. The balance and tone group maintained their cognitive ability without any additional decline, but didn’t experience any additional benefit either.
"I want to stress that we were seeing a real improvement with resistance training – not just maintenance like we did in the control group. We’re talking about women who already have some level of cognitive impairment. The most positive way of looking at this is that exercise could be a magic bullet. There is evidence now that exercise may not only be preventive – but a potential treatment."
In a separate study, Kirk Erickson, Ph.D., presented evidence of the molecular underpinning of exercise-induced brain volume changes: Physical activity, he said, stimulates the release of molecules that promote neuronal growth.
Dr. Erickson, a cognitive neuroscience researcher at the University of Pittsburgh, described the results of a walking program that involved 120 cognitively healthy, elderly adults.
The participants had been sedentary for at least the previous 6 months. They were randomized either to a 1-year program of moderately intense walking or to a 1-year stretching and toning group. At baseline and at 1 year, Dr. Erickson and his colleagues measured brain volume and levels of brain-derived neurotrophic factor (BDNF).
BDNF is a protein that affects neurons in several ways: It supports healthy neuronal function, encourages stem cells to differentiate into neurons, and stimulates dendritic expansion. BDNF is particularly active in regions associated with memory and executive functioning.
At the study’s end, Dr. Erickson and his associates found that those in the walking program had about a 2% increase in the size of the anterior hippocampus, compared with the stretching and toning group. Increases in BDNF positively correlated with the volume change. He also saw increases in the volume of the prefrontal cortex, which correlated with increased cardiorespiratory fitness.
Just how to translate these controlled experimental situations into effective, real-life practice isn’t completely clear, Dr. Liu-Ambrose said, especially when patients already have difficulty with planning and prioritization.
Her colleague, Nader Fallah, Ph.D., also of the University of British Columbia, used his expertise in biostatistics to explore that problem. Using a similar cohort of women, he found that those with higher baseline cognitive function reaped the greatest benefit from resistance training.
Dr. Fallah conducted a secondary analysis of a 12-month, randomized, controlled trial of 155 women aged 65-75 years (Neurobiol. Aging 2012;33:1690-8). The women were assigned to resistance training or balance and tone training.
This new analysis found that women with lower baseline functioning not only experienced less improvement from exercise, but also responded equally to both resistance training and balance and tone training.
"To our knowledge, this is the first study to demonstrate that an individual’s baseline self-regulatory capacity impacts the amount of cognitive benefit the person will reap from targeted exercise training," Dr. Fallah said in a written statement.
Dr. Liu-Ambrose said the study sheds valuable light on the concept of personalizing exercise prescriptions.
"I think this brings us another dimension to consider," Dr. Liu-Ambrose said. "When we prescribe exercise, most of the time we focus on physical ability: ‘Can the knees take it, what is the cardiovascular state?’ But people who have better executive function at baseline can execute this prescription better. They can plan their time, prioritize their activities, and follow through with the commitment."
Dr. Fallah’s findings suggest that "cognitive priming" might improve outcomes in prescribed exercise. Computerized brain training games are one thing to consider. They’ve been shown to improve memory and executive function for both cognitively healthy and impaired older adults. Just a small improvement in executive function and memory could be enough to get a patient invested in an exercise program.
"But for those with more pronounced dementia, it’s really about engaging the caregiver," Dr. Liu-Ambrose concluded. "We need to give them the tools they need to implement this as part of the daily lives of their loved ones."
None of the researchers reported having any financial conflicts of interest.
VANCOUVER, B.C. – Physical activity improves not only the heart and lungs, but the brain as well, providing a triad of benefits: improved cognition, better physiologic functioning, and increased brain volume and hints of revved-up neuronal growth.
Studies presented July 15 at the Alzheimer’s Association International Conference 2012 agreed: Getting active is essential for preserving cognitive health.
Exactly how exercise boosts brain function is still a matter of debate, Teresa Liu-Ambrose, Ph.D., said in an interview.
"In addition to getting the physical benefits of exercise, people are also getting social interaction and mental stimulation," said Dr. Liu-Ambrose, director of the Aging, Mobility, and Cognitive Neuroscience Lab at the University of British Columbia, Vancouver. Group activities, like exercise, can also help unwind the tangle of diminished executive function and social isolation, a damaging pair.
"When executive function begins to deteriorate, people know that they are losing some of their ability to interact, and so they withdraw and become isolated," she said. The lack of social interaction contributes to the downward slide of cognition.
Dr. Liu-Ambrose was the primary investigator of a randomized controlled trial that examined the benefits of three different exercise regimens, each conducted twice a week for 6 months: resistance training; aerobic training; and balance and tone training, which served as the comparator group.
The subjects were 86 women aged 70-80 years. They all still lived at home, but had probable mild cognitive impairment. The group is an important one because the natural history of Alzheimer’s disease suggests that about half of those who develop mild cognitive impairment will go on to develop Alzheimer’s dementia.
All of the programs were progressive, she said. Those in the weight program started with the largest weights they could handle with good form, which were increased as soon as the women exceeded the required number of repetitions. The aerobic group increased their walking speed based on heart rate and perceived exertion. The control group loaded their exercises as well, increasing repetitions and stretching as the trial progressed.
After 6 months, the strength training group had improved by 17% over baseline on measures of executive function, which was significantly better than either of the other groups. These women also experienced significant gains in associative memory.
Dr. Liu-Ambrose also saw physiologic correlates to the cognitive findings. Functional MRI showed improved blood flow in three brain regions involved with encoding and memorization of nonverbal associations: the right lingual gyrus, the occipital fusiform gyrus, and the right frontal pole.
The aerobic training group improved in balance, mobility, and cardiovascular capacity, but did not have the same kind of cognitive or brain physiologic gains, she added. The balance and tone group maintained their cognitive ability without any additional decline, but didn’t experience any additional benefit either.
"I want to stress that we were seeing a real improvement with resistance training – not just maintenance like we did in the control group. We’re talking about women who already have some level of cognitive impairment. The most positive way of looking at this is that exercise could be a magic bullet. There is evidence now that exercise may not only be preventive – but a potential treatment."
In a separate study, Kirk Erickson, Ph.D., presented evidence of the molecular underpinning of exercise-induced brain volume changes: Physical activity, he said, stimulates the release of molecules that promote neuronal growth.
Dr. Erickson, a cognitive neuroscience researcher at the University of Pittsburgh, described the results of a walking program that involved 120 cognitively healthy, elderly adults.
The participants had been sedentary for at least the previous 6 months. They were randomized either to a 1-year program of moderately intense walking or to a 1-year stretching and toning group. At baseline and at 1 year, Dr. Erickson and his colleagues measured brain volume and levels of brain-derived neurotrophic factor (BDNF).
BDNF is a protein that affects neurons in several ways: It supports healthy neuronal function, encourages stem cells to differentiate into neurons, and stimulates dendritic expansion. BDNF is particularly active in regions associated with memory and executive functioning.
At the study’s end, Dr. Erickson and his associates found that those in the walking program had about a 2% increase in the size of the anterior hippocampus, compared with the stretching and toning group. Increases in BDNF positively correlated with the volume change. He also saw increases in the volume of the prefrontal cortex, which correlated with increased cardiorespiratory fitness.
Just how to translate these controlled experimental situations into effective, real-life practice isn’t completely clear, Dr. Liu-Ambrose said, especially when patients already have difficulty with planning and prioritization.
Her colleague, Nader Fallah, Ph.D., also of the University of British Columbia, used his expertise in biostatistics to explore that problem. Using a similar cohort of women, he found that those with higher baseline cognitive function reaped the greatest benefit from resistance training.
Dr. Fallah conducted a secondary analysis of a 12-month, randomized, controlled trial of 155 women aged 65-75 years (Neurobiol. Aging 2012;33:1690-8). The women were assigned to resistance training or balance and tone training.
This new analysis found that women with lower baseline functioning not only experienced less improvement from exercise, but also responded equally to both resistance training and balance and tone training.
"To our knowledge, this is the first study to demonstrate that an individual’s baseline self-regulatory capacity impacts the amount of cognitive benefit the person will reap from targeted exercise training," Dr. Fallah said in a written statement.
Dr. Liu-Ambrose said the study sheds valuable light on the concept of personalizing exercise prescriptions.
"I think this brings us another dimension to consider," Dr. Liu-Ambrose said. "When we prescribe exercise, most of the time we focus on physical ability: ‘Can the knees take it, what is the cardiovascular state?’ But people who have better executive function at baseline can execute this prescription better. They can plan their time, prioritize their activities, and follow through with the commitment."
Dr. Fallah’s findings suggest that "cognitive priming" might improve outcomes in prescribed exercise. Computerized brain training games are one thing to consider. They’ve been shown to improve memory and executive function for both cognitively healthy and impaired older adults. Just a small improvement in executive function and memory could be enough to get a patient invested in an exercise program.
"But for those with more pronounced dementia, it’s really about engaging the caregiver," Dr. Liu-Ambrose concluded. "We need to give them the tools they need to implement this as part of the daily lives of their loved ones."
None of the researchers reported having any financial conflicts of interest.
AT THE ALZHEIMER'S ASSOCIATION INTERNATIONAL CONFERENCE 2012
New Act Outlaws 'Bath Salt' Compounds
Designer stimulants are among the latest compounds to make the country’s Schedule 1 list of illegal drugs.
On July 9, President Obama signed into law the Synthetic Drug Abuse Prevention Act of 2012. The law adds 31 compounds to schedule I, including 9 that fall into the cathinone-based "bath salts" category and 20 found in synthetic marijuana, commonly called spice or K2.
Sen. Amy Klobuchar (D-Minn.), who sponsored one of three bills contained in the law, applauded the president’s action.
"In Minnesota and across the country, we are seeing more and more tragedies where synthetic drugs are taking lives and tearing apart families," she said in a statement. "[The president’s] action means that this critical legislation to give law enforcement the tools they need to crack down on synthetic drugs is finally the law of the land."
The legislation bolsters existing laws in 44 states, which have previously outlawed specific compounds related to cathinone. However, underground chemists constantly alter the molecular makeup of the compounds, creating versions that are not covered in these laws and are therefore technically legal. The new act bans all drugs in the class now known as substituted cathinone enhancements.
The drugs are both powerful and dangerous, according to the American Association of Poison Control Centers. In 2010, poison control centers around the country began getting calls about reactions – some extreme – to unknown substances.
That year, "poison centers nationwide responded to about 3,200 calls related to synthetic marijuana and bath salts. In 2011, that number jumped to more than 13,000 calls. Sixty percent of the cases involved patients 25 and younger," Dr. Rick Dart, the association’s president, said in a statement.
Tachycardia, agitation, hallucinations, extreme paranoia, delusions, and elevations in blood pressure are common symptoms of exposure. Up to 40% of users can experience severe seizures. Psychosis occurs in 14%-40%. While in a psychotic state, some users exhibit dangerously bizarre behaviors.
Sensationalistic reports of cannibalism and users ripping off their clothing in fits of paranoia have hit national news recently, but the relationship to bath salts is often unclear, according to a report by Dr. Jason Jerry.
"There are simply no data regarding the prevalence of psychotic symptoms in the larger group of all synthetic stimulant users," he said.
Patients who arrive at the emergency department with such severe symptoms are often incorrectly given psychiatric diagnoses, said Dr. Jerry of the Cleveland Clinic. Now, reactions to the compounds should be among the first items in a differential diagnosis for these patients.
"Most patients ... will present with anxiety, agitation, and psychosis," he wrote (Cleve. Clin. J. Med. 2012;79:258-64). "Rather than assume that they are psychiatric patients, one should consider the possibility of [bath salts intoxication] and pay close attention to the possible medical sequelae associated with SLID [synthetic legal intoxicating drug] use, such as elevated blood pressure, tachycardia, and seizure."
Because the drugs have only recently been seen in the United States, toxicology screens are also relatively new. Several companies now offer such screens; almost all of them require that urine samples be sent for outside lab processing.
Most use mass spectroscopy to identity cathinone-related compounds. An enzyme-linked immunosorbent assay (ELISA) test has recently become available for in-house use.
A new mass spectroscopy test by Ameritox became available in July; the test can identify three cathinone compounds and five other chemicals often found in bath salts. But drug testing is just one step in getting a grip on the rising problem of synthetic stimulant use, Dr. Harry Leider, chief medical office of Ameritox, said in an interview.
"The solution to this problem is not simple. It involves multiple forces coming together."
Law enforcement and the medical community need to be on the "fast track, to determine if the drugs are in a community," he said.
"People need to be educated about the idea that something isn’t safe just because it’s legal. Doctors need to become more knowledgeable about the drugs and their effects. And we need support from payers so everyone can keep pace with the chemists, who keep turning out new compounds to keep ahead of the laws."
Designer stimulants are among the latest compounds to make the country’s Schedule 1 list of illegal drugs.
On July 9, President Obama signed into law the Synthetic Drug Abuse Prevention Act of 2012. The law adds 31 compounds to schedule I, including 9 that fall into the cathinone-based "bath salts" category and 20 found in synthetic marijuana, commonly called spice or K2.
Sen. Amy Klobuchar (D-Minn.), who sponsored one of three bills contained in the law, applauded the president’s action.
"In Minnesota and across the country, we are seeing more and more tragedies where synthetic drugs are taking lives and tearing apart families," she said in a statement. "[The president’s] action means that this critical legislation to give law enforcement the tools they need to crack down on synthetic drugs is finally the law of the land."
The legislation bolsters existing laws in 44 states, which have previously outlawed specific compounds related to cathinone. However, underground chemists constantly alter the molecular makeup of the compounds, creating versions that are not covered in these laws and are therefore technically legal. The new act bans all drugs in the class now known as substituted cathinone enhancements.
The drugs are both powerful and dangerous, according to the American Association of Poison Control Centers. In 2010, poison control centers around the country began getting calls about reactions – some extreme – to unknown substances.
That year, "poison centers nationwide responded to about 3,200 calls related to synthetic marijuana and bath salts. In 2011, that number jumped to more than 13,000 calls. Sixty percent of the cases involved patients 25 and younger," Dr. Rick Dart, the association’s president, said in a statement.
Tachycardia, agitation, hallucinations, extreme paranoia, delusions, and elevations in blood pressure are common symptoms of exposure. Up to 40% of users can experience severe seizures. Psychosis occurs in 14%-40%. While in a psychotic state, some users exhibit dangerously bizarre behaviors.
Sensationalistic reports of cannibalism and users ripping off their clothing in fits of paranoia have hit national news recently, but the relationship to bath salts is often unclear, according to a report by Dr. Jason Jerry.
"There are simply no data regarding the prevalence of psychotic symptoms in the larger group of all synthetic stimulant users," he said.
Patients who arrive at the emergency department with such severe symptoms are often incorrectly given psychiatric diagnoses, said Dr. Jerry of the Cleveland Clinic. Now, reactions to the compounds should be among the first items in a differential diagnosis for these patients.
"Most patients ... will present with anxiety, agitation, and psychosis," he wrote (Cleve. Clin. J. Med. 2012;79:258-64). "Rather than assume that they are psychiatric patients, one should consider the possibility of [bath salts intoxication] and pay close attention to the possible medical sequelae associated with SLID [synthetic legal intoxicating drug] use, such as elevated blood pressure, tachycardia, and seizure."
Because the drugs have only recently been seen in the United States, toxicology screens are also relatively new. Several companies now offer such screens; almost all of them require that urine samples be sent for outside lab processing.
Most use mass spectroscopy to identity cathinone-related compounds. An enzyme-linked immunosorbent assay (ELISA) test has recently become available for in-house use.
A new mass spectroscopy test by Ameritox became available in July; the test can identify three cathinone compounds and five other chemicals often found in bath salts. But drug testing is just one step in getting a grip on the rising problem of synthetic stimulant use, Dr. Harry Leider, chief medical office of Ameritox, said in an interview.
"The solution to this problem is not simple. It involves multiple forces coming together."
Law enforcement and the medical community need to be on the "fast track, to determine if the drugs are in a community," he said.
"People need to be educated about the idea that something isn’t safe just because it’s legal. Doctors need to become more knowledgeable about the drugs and their effects. And we need support from payers so everyone can keep pace with the chemists, who keep turning out new compounds to keep ahead of the laws."
Designer stimulants are among the latest compounds to make the country’s Schedule 1 list of illegal drugs.
On July 9, President Obama signed into law the Synthetic Drug Abuse Prevention Act of 2012. The law adds 31 compounds to schedule I, including 9 that fall into the cathinone-based "bath salts" category and 20 found in synthetic marijuana, commonly called spice or K2.
Sen. Amy Klobuchar (D-Minn.), who sponsored one of three bills contained in the law, applauded the president’s action.
"In Minnesota and across the country, we are seeing more and more tragedies where synthetic drugs are taking lives and tearing apart families," she said in a statement. "[The president’s] action means that this critical legislation to give law enforcement the tools they need to crack down on synthetic drugs is finally the law of the land."
The legislation bolsters existing laws in 44 states, which have previously outlawed specific compounds related to cathinone. However, underground chemists constantly alter the molecular makeup of the compounds, creating versions that are not covered in these laws and are therefore technically legal. The new act bans all drugs in the class now known as substituted cathinone enhancements.
The drugs are both powerful and dangerous, according to the American Association of Poison Control Centers. In 2010, poison control centers around the country began getting calls about reactions – some extreme – to unknown substances.
That year, "poison centers nationwide responded to about 3,200 calls related to synthetic marijuana and bath salts. In 2011, that number jumped to more than 13,000 calls. Sixty percent of the cases involved patients 25 and younger," Dr. Rick Dart, the association’s president, said in a statement.
Tachycardia, agitation, hallucinations, extreme paranoia, delusions, and elevations in blood pressure are common symptoms of exposure. Up to 40% of users can experience severe seizures. Psychosis occurs in 14%-40%. While in a psychotic state, some users exhibit dangerously bizarre behaviors.
Sensationalistic reports of cannibalism and users ripping off their clothing in fits of paranoia have hit national news recently, but the relationship to bath salts is often unclear, according to a report by Dr. Jason Jerry.
"There are simply no data regarding the prevalence of psychotic symptoms in the larger group of all synthetic stimulant users," he said.
Patients who arrive at the emergency department with such severe symptoms are often incorrectly given psychiatric diagnoses, said Dr. Jerry of the Cleveland Clinic. Now, reactions to the compounds should be among the first items in a differential diagnosis for these patients.
"Most patients ... will present with anxiety, agitation, and psychosis," he wrote (Cleve. Clin. J. Med. 2012;79:258-64). "Rather than assume that they are psychiatric patients, one should consider the possibility of [bath salts intoxication] and pay close attention to the possible medical sequelae associated with SLID [synthetic legal intoxicating drug] use, such as elevated blood pressure, tachycardia, and seizure."
Because the drugs have only recently been seen in the United States, toxicology screens are also relatively new. Several companies now offer such screens; almost all of them require that urine samples be sent for outside lab processing.
Most use mass spectroscopy to identity cathinone-related compounds. An enzyme-linked immunosorbent assay (ELISA) test has recently become available for in-house use.
A new mass spectroscopy test by Ameritox became available in July; the test can identify three cathinone compounds and five other chemicals often found in bath salts. But drug testing is just one step in getting a grip on the rising problem of synthetic stimulant use, Dr. Harry Leider, chief medical office of Ameritox, said in an interview.
"The solution to this problem is not simple. It involves multiple forces coming together."
Law enforcement and the medical community need to be on the "fast track, to determine if the drugs are in a community," he said.
"People need to be educated about the idea that something isn’t safe just because it’s legal. Doctors need to become more knowledgeable about the drugs and their effects. And we need support from payers so everyone can keep pace with the chemists, who keep turning out new compounds to keep ahead of the laws."
Studies Bolster Alzheimer's Amyloid Hypothesis
Two new studies appear to lend strong support for the theory that an overabundance of the amyloid-beta protein is the root cause of Alzheimer’s disease.
A genetic study of 1,800 residents of Iceland discovered a rare mutation that apparently protects against the disease in those who carry it, and even staves off the disease in those genetically programmed to develop it – probably by blocking the formation of amyloid-beta.
A second study, published July 11 in the New England Journal of Medicine, found that a processed fragment of amyloid-beta, called amyloid-beta-42 (Abeta42), begins to accumulate in the brain 25 years before symptoms appear in people with a mutation in one of three genes that is known to cause early-onset Alzheimer’s disease, suggesting that amyloid deposition initiates the cascade of physiologic and cognitive changes that culminate in full-blown AD.
"Our data suggest that amyloid deposition will develop and be detectable in all persons with a mutation while they are still asymptomatic," wrote Dr. Randall J. Bateman, professor of neurology at Washington University, St. Louis, and his colleagues. "These findings suggest that targeting amyloid-beta earlier in the course of the disease may provide better clinical outcomes than the treatment of mild to moderate dementia after substantial neuronal and synaptic loss has occurred" (N. Engl. J. Med. 2012 July 11 [doi: 10.1056/NEJMoa1202753]).
Dr. Kari Stefansson, chief executive officer of deCODE Genetics in Reykjavik, led the Icelandic study. He and his colleagues sought to discover any unusual genetic risk factors for AD in a group of about 1,800 subjects. The researchers focused on variants of the amyloid precursor protein gene (APP), because different enzymes can either split this protein into a harmless fragment or change it into Abeta42 – the form that aggregates into AD brain plaques.
In the initial group, Dr. Stefansson and his colleagues looked for every APP variant that occurred in more than one person, and then imputed these into a database of 72,000 Icelanders who had already been genotyped. A computer program estimated how many times each allele might occur in almost 300,000 relatives of these subjects (Nature 2012 July 11 [doi: 10.1038/nature11283]).
The researchers found a mutation (A673T) in APP that was significantly more common in those without AD than in those with it. The variant was rare, occurring in 0.14% of those with Alzheimer’s, 0.45% of overall population-based controls, 0.62% of controls aged 85 years or older, and 0.79% of controls older than 85 who were still cognitively intact. Nevertheless, the authors concluded, the presence of the variant exerted a protective effect against the disease.
This was even more apparent in risk analyses. Compared with the group of people with AD, the gene was four times more likely to appear in the general population, five times more likely to occur in those 85 years or older, and nearly eight times more common among a group of cognitively healthy subjects who were 85 or older.
The allele also appeared to protect against expected age-related cognitive decline in elderly people who did not have AD, the authors noted. In a comparison of 41 allele carriers and 3,700 noncarriers, all of whom were 80-100 years old, those with the protective allele had significantly better cognitive ability. The relationship remained strong even after removal of patients with AD from the noncarrier group.
"Removing known Alzheimer’s disease cases [from this analysis] suggests that the protective effect of A673T extends beyond the boundaries of the Alzheimer’s disease phenotype," the investigators wrote.
The mutation’s benefit probably arises from its influence over APP fragmentation, the researchers said. Three enzymes – alpha-, beta-, and gamma-secretase – can cleave the APP protein. If alpha-secretase splits the protein, no further fragmentation occurs. When gamma-secretase splits APP, it sets the stage for beta-secretase (BACE1) to finish the job, producing Abeta42, the form most likely to aggregate into brain plaques.
In vitro experiments indicated that the allele blocked the effect of BACE1, reducing the production of Abeta42 by up to 50%. This suggests that a well-tolerated BACE1 inhibitor might have a place in any future Alzheimer’s treatment regimen.
The New England Journal of Medicine study examined Abeta42 levels and cognitive function in 128 subjects enrolled in the Dominantly Inherited Alzheimer Network (DIAN) study. The 6-year, cross-sectional, longitudinal study is examining members of families who carry a mutation in one of the three genes that affect amyloid processing, including APP. All subjects have at least one parent with a mutation known to cause early-onset AD.
The study group’s mean age at enrollment was 39 years. About half of the carriers are symptomatic, compared with just 2% of the noncarriers. Parents of these subjects developed AD at a mean age of 46 years.
Because the likelihood of developing the disease is so high in carriers, their cognitive and brain health can serve as a natural history of the development of AD. By assuming that each person will develop symptoms at or about the same age as did the affected parent, researchers can plot changes that might occur in the years – or even decades – before the disease manifests.
For this analysis, Dr. Bateman and his colleagues examined cerebrospinal fluid (CSF), brain imaging, and cognitive data from 128 participants who had been tracked from the 2009 baseline enrollment through 2010.
There were significant differences between the carriers and noncarriers in every measure investigated, with signs developing up to 25 years before the expected onset of disease.
In cognitive measures, carriers scored significantly lower on the Mini-Mental State Exam starting at 5 years before expected symptom onset, and in delayed recall at 10 years. Noncarriers’ scores remained normal anywhere from 30 years before to 20 years after expected onset. Carriers showed increased atrophy of the bilateral hippocampi, occurring about 15 years before expected onset.
But the differences seen in amyloid deposition and CSF biomarkers most strongly support the amyloid hypotheses. There was no detectable Abeta42 accumulation in the brains of any of the noncarriers. Those who carried the mutation had significant amyloid deposition in the precuneus up to 15 years before the expected onset of AD symptoms. The amount of amyloid increased as subjects approached that time.
Similarly, CSF levels of Abeta42 in carriers decreased as the time of symptom onset approached, indicating that more of the protein was aggregating in the brain. The change was apparent by 10 years before symptom onset. This was accompanied by an increase in CSF tau, a protein that helps stabilize neurons. Its presence in CSF indicates neuronal dysfunction.
Based on these findings, the authors were able to construct a time line of changes that precede the expected time of symptom onset in people with these genetic variants.
CSF biomarkers are the first to change; 25 years before symptoms appear, Abeta42 begins to accumulate in the brain. By at least 15 years before symptoms, imaging will show Abeta42 plaques, and neurons begin to destabilize, increasing CSF tau. Cerebral hypometabolism appears about 10 years before the expected onset of symptoms.
"After these biologic changes, cognitive impairment can be detected, which culminates in clinical impairment and eventually dementia," the investigators wrote. "These findings suggest that the diagnosis of clinical dementia is made late in the course of the biologic cascade of autosomal dominant Alzheimer’s disease."
The results support the hypothesis that inherited AD and the more common sporadic AD may share a common pathophysiological cascade. If this is true, they noted, then the new ability to detect amyloid deposition with brain imaging agents could be much more useful than originally envisioned.
"If autosomal dominant Alzheimer’s disease is similar to late-onset Alzheimer’s disease, this finding suggests that Alzheimer’s dementia will eventually develop in persons with positive scans for amyloid deposition," Dr. Bateman and his associates wrote.
The DIAN study is being funded by the National Institute on Aging. Dr. Bateman and his colleagues disclosed numerous relationships with drug companies conducting Alzheimer’s therapy research. The Icelandic study was sponsored by deCODE Genetics, which employs some of the authors of the study.
A New Era for Alzheimer’s?
When asked to comment on the study results, Dr. Richard J. Caselli, professor of neurology at the Mayo Clinic in Scottsdale, Ariz., said, "Dr. Stefansson and his colleagues show convincingly that individuals with the A673T mutation have a greatly reduced rate of Alzheimer’s development as well as – to a somewhat less-robust degree – slowed cognitive aging even in the absence of Alzheimer’s disease.
"The investigators show the mechanism to be reduced production of amyloidogenic peptides by beta-secretase 1 (BACE1) activity, compared with the wild-type version of the gene. In contrast, the pathogenic A673V variant dramatically increases the amyloidogenic BACE1 products. The authors have essentially discovered nature’s cure for AD, and we can only hope that its pharmacological replication will soon follow," said Dr. Caselli, who is also the clinical core director of the Arizona Alzheimer’s Disease Center.
"The separate report by Dr. Bateman and his colleagues from the Dominantly Inherited Alzheimer Network is another remarkable work," Dr. Caselli continued, "providing unprecedented insight into the sequence of events in the same cohort with regard to biomarkers, imaging, and cognition in advance of symptoms, and using all the tools now in our arsenal: brain imaging, cerebrospinal fluid biomarkers, brain metabolism, and cognitive measures.
"More than 2 decades before any noticeable symptoms, pathophysiological changes are at work in patients who are destined to develop AD. This information may now provide us objective, identifiable outcomes with which to assess new therapies," said Dr. Caselli.
"Based upon the findings of Dr. Stefansson and his colleagues, it’s easy to imagine how a new BACE1 inhibitor could be applied to a preclinical Alzheimer’s population, with outcomes measured by the targets identified by Dr. Bateman and his coauthors – perhaps catapulting us into a new age of prevention therapy for AD," he noted.
Two new studies appear to lend strong support for the theory that an overabundance of the amyloid-beta protein is the root cause of Alzheimer’s disease.
A genetic study of 1,800 residents of Iceland discovered a rare mutation that apparently protects against the disease in those who carry it, and even staves off the disease in those genetically programmed to develop it – probably by blocking the formation of amyloid-beta.
A second study, published July 11 in the New England Journal of Medicine, found that a processed fragment of amyloid-beta, called amyloid-beta-42 (Abeta42), begins to accumulate in the brain 25 years before symptoms appear in people with a mutation in one of three genes that is known to cause early-onset Alzheimer’s disease, suggesting that amyloid deposition initiates the cascade of physiologic and cognitive changes that culminate in full-blown AD.
"Our data suggest that amyloid deposition will develop and be detectable in all persons with a mutation while they are still asymptomatic," wrote Dr. Randall J. Bateman, professor of neurology at Washington University, St. Louis, and his colleagues. "These findings suggest that targeting amyloid-beta earlier in the course of the disease may provide better clinical outcomes than the treatment of mild to moderate dementia after substantial neuronal and synaptic loss has occurred" (N. Engl. J. Med. 2012 July 11 [doi: 10.1056/NEJMoa1202753]).
Dr. Kari Stefansson, chief executive officer of deCODE Genetics in Reykjavik, led the Icelandic study. He and his colleagues sought to discover any unusual genetic risk factors for AD in a group of about 1,800 subjects. The researchers focused on variants of the amyloid precursor protein gene (APP), because different enzymes can either split this protein into a harmless fragment or change it into Abeta42 – the form that aggregates into AD brain plaques.
In the initial group, Dr. Stefansson and his colleagues looked for every APP variant that occurred in more than one person, and then imputed these into a database of 72,000 Icelanders who had already been genotyped. A computer program estimated how many times each allele might occur in almost 300,000 relatives of these subjects (Nature 2012 July 11 [doi: 10.1038/nature11283]).
The researchers found a mutation (A673T) in APP that was significantly more common in those without AD than in those with it. The variant was rare, occurring in 0.14% of those with Alzheimer’s, 0.45% of overall population-based controls, 0.62% of controls aged 85 years or older, and 0.79% of controls older than 85 who were still cognitively intact. Nevertheless, the authors concluded, the presence of the variant exerted a protective effect against the disease.
This was even more apparent in risk analyses. Compared with the group of people with AD, the gene was four times more likely to appear in the general population, five times more likely to occur in those 85 years or older, and nearly eight times more common among a group of cognitively healthy subjects who were 85 or older.
The allele also appeared to protect against expected age-related cognitive decline in elderly people who did not have AD, the authors noted. In a comparison of 41 allele carriers and 3,700 noncarriers, all of whom were 80-100 years old, those with the protective allele had significantly better cognitive ability. The relationship remained strong even after removal of patients with AD from the noncarrier group.
"Removing known Alzheimer’s disease cases [from this analysis] suggests that the protective effect of A673T extends beyond the boundaries of the Alzheimer’s disease phenotype," the investigators wrote.
The mutation’s benefit probably arises from its influence over APP fragmentation, the researchers said. Three enzymes – alpha-, beta-, and gamma-secretase – can cleave the APP protein. If alpha-secretase splits the protein, no further fragmentation occurs. When gamma-secretase splits APP, it sets the stage for beta-secretase (BACE1) to finish the job, producing Abeta42, the form most likely to aggregate into brain plaques.
In vitro experiments indicated that the allele blocked the effect of BACE1, reducing the production of Abeta42 by up to 50%. This suggests that a well-tolerated BACE1 inhibitor might have a place in any future Alzheimer’s treatment regimen.
The New England Journal of Medicine study examined Abeta42 levels and cognitive function in 128 subjects enrolled in the Dominantly Inherited Alzheimer Network (DIAN) study. The 6-year, cross-sectional, longitudinal study is examining members of families who carry a mutation in one of the three genes that affect amyloid processing, including APP. All subjects have at least one parent with a mutation known to cause early-onset AD.
The study group’s mean age at enrollment was 39 years. About half of the carriers are symptomatic, compared with just 2% of the noncarriers. Parents of these subjects developed AD at a mean age of 46 years.
Because the likelihood of developing the disease is so high in carriers, their cognitive and brain health can serve as a natural history of the development of AD. By assuming that each person will develop symptoms at or about the same age as did the affected parent, researchers can plot changes that might occur in the years – or even decades – before the disease manifests.
For this analysis, Dr. Bateman and his colleagues examined cerebrospinal fluid (CSF), brain imaging, and cognitive data from 128 participants who had been tracked from the 2009 baseline enrollment through 2010.
There were significant differences between the carriers and noncarriers in every measure investigated, with signs developing up to 25 years before the expected onset of disease.
In cognitive measures, carriers scored significantly lower on the Mini-Mental State Exam starting at 5 years before expected symptom onset, and in delayed recall at 10 years. Noncarriers’ scores remained normal anywhere from 30 years before to 20 years after expected onset. Carriers showed increased atrophy of the bilateral hippocampi, occurring about 15 years before expected onset.
But the differences seen in amyloid deposition and CSF biomarkers most strongly support the amyloid hypotheses. There was no detectable Abeta42 accumulation in the brains of any of the noncarriers. Those who carried the mutation had significant amyloid deposition in the precuneus up to 15 years before the expected onset of AD symptoms. The amount of amyloid increased as subjects approached that time.
Similarly, CSF levels of Abeta42 in carriers decreased as the time of symptom onset approached, indicating that more of the protein was aggregating in the brain. The change was apparent by 10 years before symptom onset. This was accompanied by an increase in CSF tau, a protein that helps stabilize neurons. Its presence in CSF indicates neuronal dysfunction.
Based on these findings, the authors were able to construct a time line of changes that precede the expected time of symptom onset in people with these genetic variants.
CSF biomarkers are the first to change; 25 years before symptoms appear, Abeta42 begins to accumulate in the brain. By at least 15 years before symptoms, imaging will show Abeta42 plaques, and neurons begin to destabilize, increasing CSF tau. Cerebral hypometabolism appears about 10 years before the expected onset of symptoms.
"After these biologic changes, cognitive impairment can be detected, which culminates in clinical impairment and eventually dementia," the investigators wrote. "These findings suggest that the diagnosis of clinical dementia is made late in the course of the biologic cascade of autosomal dominant Alzheimer’s disease."
The results support the hypothesis that inherited AD and the more common sporadic AD may share a common pathophysiological cascade. If this is true, they noted, then the new ability to detect amyloid deposition with brain imaging agents could be much more useful than originally envisioned.
"If autosomal dominant Alzheimer’s disease is similar to late-onset Alzheimer’s disease, this finding suggests that Alzheimer’s dementia will eventually develop in persons with positive scans for amyloid deposition," Dr. Bateman and his associates wrote.
The DIAN study is being funded by the National Institute on Aging. Dr. Bateman and his colleagues disclosed numerous relationships with drug companies conducting Alzheimer’s therapy research. The Icelandic study was sponsored by deCODE Genetics, which employs some of the authors of the study.
A New Era for Alzheimer’s?
When asked to comment on the study results, Dr. Richard J. Caselli, professor of neurology at the Mayo Clinic in Scottsdale, Ariz., said, "Dr. Stefansson and his colleagues show convincingly that individuals with the A673T mutation have a greatly reduced rate of Alzheimer’s development as well as – to a somewhat less-robust degree – slowed cognitive aging even in the absence of Alzheimer’s disease.
"The investigators show the mechanism to be reduced production of amyloidogenic peptides by beta-secretase 1 (BACE1) activity, compared with the wild-type version of the gene. In contrast, the pathogenic A673V variant dramatically increases the amyloidogenic BACE1 products. The authors have essentially discovered nature’s cure for AD, and we can only hope that its pharmacological replication will soon follow," said Dr. Caselli, who is also the clinical core director of the Arizona Alzheimer’s Disease Center.
"The separate report by Dr. Bateman and his colleagues from the Dominantly Inherited Alzheimer Network is another remarkable work," Dr. Caselli continued, "providing unprecedented insight into the sequence of events in the same cohort with regard to biomarkers, imaging, and cognition in advance of symptoms, and using all the tools now in our arsenal: brain imaging, cerebrospinal fluid biomarkers, brain metabolism, and cognitive measures.
"More than 2 decades before any noticeable symptoms, pathophysiological changes are at work in patients who are destined to develop AD. This information may now provide us objective, identifiable outcomes with which to assess new therapies," said Dr. Caselli.
"Based upon the findings of Dr. Stefansson and his colleagues, it’s easy to imagine how a new BACE1 inhibitor could be applied to a preclinical Alzheimer’s population, with outcomes measured by the targets identified by Dr. Bateman and his coauthors – perhaps catapulting us into a new age of prevention therapy for AD," he noted.
Two new studies appear to lend strong support for the theory that an overabundance of the amyloid-beta protein is the root cause of Alzheimer’s disease.
A genetic study of 1,800 residents of Iceland discovered a rare mutation that apparently protects against the disease in those who carry it, and even staves off the disease in those genetically programmed to develop it – probably by blocking the formation of amyloid-beta.
A second study, published July 11 in the New England Journal of Medicine, found that a processed fragment of amyloid-beta, called amyloid-beta-42 (Abeta42), begins to accumulate in the brain 25 years before symptoms appear in people with a mutation in one of three genes that is known to cause early-onset Alzheimer’s disease, suggesting that amyloid deposition initiates the cascade of physiologic and cognitive changes that culminate in full-blown AD.
"Our data suggest that amyloid deposition will develop and be detectable in all persons with a mutation while they are still asymptomatic," wrote Dr. Randall J. Bateman, professor of neurology at Washington University, St. Louis, and his colleagues. "These findings suggest that targeting amyloid-beta earlier in the course of the disease may provide better clinical outcomes than the treatment of mild to moderate dementia after substantial neuronal and synaptic loss has occurred" (N. Engl. J. Med. 2012 July 11 [doi: 10.1056/NEJMoa1202753]).
Dr. Kari Stefansson, chief executive officer of deCODE Genetics in Reykjavik, led the Icelandic study. He and his colleagues sought to discover any unusual genetic risk factors for AD in a group of about 1,800 subjects. The researchers focused on variants of the amyloid precursor protein gene (APP), because different enzymes can either split this protein into a harmless fragment or change it into Abeta42 – the form that aggregates into AD brain plaques.
In the initial group, Dr. Stefansson and his colleagues looked for every APP variant that occurred in more than one person, and then imputed these into a database of 72,000 Icelanders who had already been genotyped. A computer program estimated how many times each allele might occur in almost 300,000 relatives of these subjects (Nature 2012 July 11 [doi: 10.1038/nature11283]).
The researchers found a mutation (A673T) in APP that was significantly more common in those without AD than in those with it. The variant was rare, occurring in 0.14% of those with Alzheimer’s, 0.45% of overall population-based controls, 0.62% of controls aged 85 years or older, and 0.79% of controls older than 85 who were still cognitively intact. Nevertheless, the authors concluded, the presence of the variant exerted a protective effect against the disease.
This was even more apparent in risk analyses. Compared with the group of people with AD, the gene was four times more likely to appear in the general population, five times more likely to occur in those 85 years or older, and nearly eight times more common among a group of cognitively healthy subjects who were 85 or older.
The allele also appeared to protect against expected age-related cognitive decline in elderly people who did not have AD, the authors noted. In a comparison of 41 allele carriers and 3,700 noncarriers, all of whom were 80-100 years old, those with the protective allele had significantly better cognitive ability. The relationship remained strong even after removal of patients with AD from the noncarrier group.
"Removing known Alzheimer’s disease cases [from this analysis] suggests that the protective effect of A673T extends beyond the boundaries of the Alzheimer’s disease phenotype," the investigators wrote.
The mutation’s benefit probably arises from its influence over APP fragmentation, the researchers said. Three enzymes – alpha-, beta-, and gamma-secretase – can cleave the APP protein. If alpha-secretase splits the protein, no further fragmentation occurs. When gamma-secretase splits APP, it sets the stage for beta-secretase (BACE1) to finish the job, producing Abeta42, the form most likely to aggregate into brain plaques.
In vitro experiments indicated that the allele blocked the effect of BACE1, reducing the production of Abeta42 by up to 50%. This suggests that a well-tolerated BACE1 inhibitor might have a place in any future Alzheimer’s treatment regimen.
The New England Journal of Medicine study examined Abeta42 levels and cognitive function in 128 subjects enrolled in the Dominantly Inherited Alzheimer Network (DIAN) study. The 6-year, cross-sectional, longitudinal study is examining members of families who carry a mutation in one of the three genes that affect amyloid processing, including APP. All subjects have at least one parent with a mutation known to cause early-onset AD.
The study group’s mean age at enrollment was 39 years. About half of the carriers are symptomatic, compared with just 2% of the noncarriers. Parents of these subjects developed AD at a mean age of 46 years.
Because the likelihood of developing the disease is so high in carriers, their cognitive and brain health can serve as a natural history of the development of AD. By assuming that each person will develop symptoms at or about the same age as did the affected parent, researchers can plot changes that might occur in the years – or even decades – before the disease manifests.
For this analysis, Dr. Bateman and his colleagues examined cerebrospinal fluid (CSF), brain imaging, and cognitive data from 128 participants who had been tracked from the 2009 baseline enrollment through 2010.
There were significant differences between the carriers and noncarriers in every measure investigated, with signs developing up to 25 years before the expected onset of disease.
In cognitive measures, carriers scored significantly lower on the Mini-Mental State Exam starting at 5 years before expected symptom onset, and in delayed recall at 10 years. Noncarriers’ scores remained normal anywhere from 30 years before to 20 years after expected onset. Carriers showed increased atrophy of the bilateral hippocampi, occurring about 15 years before expected onset.
But the differences seen in amyloid deposition and CSF biomarkers most strongly support the amyloid hypotheses. There was no detectable Abeta42 accumulation in the brains of any of the noncarriers. Those who carried the mutation had significant amyloid deposition in the precuneus up to 15 years before the expected onset of AD symptoms. The amount of amyloid increased as subjects approached that time.
Similarly, CSF levels of Abeta42 in carriers decreased as the time of symptom onset approached, indicating that more of the protein was aggregating in the brain. The change was apparent by 10 years before symptom onset. This was accompanied by an increase in CSF tau, a protein that helps stabilize neurons. Its presence in CSF indicates neuronal dysfunction.
Based on these findings, the authors were able to construct a time line of changes that precede the expected time of symptom onset in people with these genetic variants.
CSF biomarkers are the first to change; 25 years before symptoms appear, Abeta42 begins to accumulate in the brain. By at least 15 years before symptoms, imaging will show Abeta42 plaques, and neurons begin to destabilize, increasing CSF tau. Cerebral hypometabolism appears about 10 years before the expected onset of symptoms.
"After these biologic changes, cognitive impairment can be detected, which culminates in clinical impairment and eventually dementia," the investigators wrote. "These findings suggest that the diagnosis of clinical dementia is made late in the course of the biologic cascade of autosomal dominant Alzheimer’s disease."
The results support the hypothesis that inherited AD and the more common sporadic AD may share a common pathophysiological cascade. If this is true, they noted, then the new ability to detect amyloid deposition with brain imaging agents could be much more useful than originally envisioned.
"If autosomal dominant Alzheimer’s disease is similar to late-onset Alzheimer’s disease, this finding suggests that Alzheimer’s dementia will eventually develop in persons with positive scans for amyloid deposition," Dr. Bateman and his associates wrote.
The DIAN study is being funded by the National Institute on Aging. Dr. Bateman and his colleagues disclosed numerous relationships with drug companies conducting Alzheimer’s therapy research. The Icelandic study was sponsored by deCODE Genetics, which employs some of the authors of the study.
A New Era for Alzheimer’s?
When asked to comment on the study results, Dr. Richard J. Caselli, professor of neurology at the Mayo Clinic in Scottsdale, Ariz., said, "Dr. Stefansson and his colleagues show convincingly that individuals with the A673T mutation have a greatly reduced rate of Alzheimer’s development as well as – to a somewhat less-robust degree – slowed cognitive aging even in the absence of Alzheimer’s disease.
"The investigators show the mechanism to be reduced production of amyloidogenic peptides by beta-secretase 1 (BACE1) activity, compared with the wild-type version of the gene. In contrast, the pathogenic A673V variant dramatically increases the amyloidogenic BACE1 products. The authors have essentially discovered nature’s cure for AD, and we can only hope that its pharmacological replication will soon follow," said Dr. Caselli, who is also the clinical core director of the Arizona Alzheimer’s Disease Center.
"The separate report by Dr. Bateman and his colleagues from the Dominantly Inherited Alzheimer Network is another remarkable work," Dr. Caselli continued, "providing unprecedented insight into the sequence of events in the same cohort with regard to biomarkers, imaging, and cognition in advance of symptoms, and using all the tools now in our arsenal: brain imaging, cerebrospinal fluid biomarkers, brain metabolism, and cognitive measures.
"More than 2 decades before any noticeable symptoms, pathophysiological changes are at work in patients who are destined to develop AD. This information may now provide us objective, identifiable outcomes with which to assess new therapies," said Dr. Caselli.
"Based upon the findings of Dr. Stefansson and his colleagues, it’s easy to imagine how a new BACE1 inhibitor could be applied to a preclinical Alzheimer’s population, with outcomes measured by the targets identified by Dr. Bateman and his coauthors – perhaps catapulting us into a new age of prevention therapy for AD," he noted.
FROM NATURE AND THE NEW ENGLAND JOURNAL OF MEDICINE
QoL Steadies 15 Years After Prostate Cancer Treatments
ATLANTA – At 15 years after treatment, disease-related quality of life measures appear similar in men with prostate cancer, regardless of whether they had radical prostatectomy or external radiation beam therapy.
The significant differences in sexual, urinary, and bowel function that were apparent in the first 5 years, largely disappeared as time advanced, Dr. Matthew Resnick reported, based on a new subanalysis of men in the Prostate Cancer Outcomes Study.
The convergence of outcomes may not have so much to do with treatment type as it does advancing age, he said at the annual meeting of the American Urologic Association. This is especially apparent in erectile dysfunction, according to Dr. Resnick, a urologic surgeon at Vanderbilt University Medical Center, Nashville, Tenn.
"Age really is the most significant determinant of decline in erectile function over time," he said. "About 50% of men older than 70 have erectile dysfunction."
The Prostate Cancer Outcomes Study (PCOS) enrolled patients from six Surveillance, Epidemiology, and End Results (SEER) tumor registries to examine long-term functional outcomes in men who were diagnosed with prostate cancer between 1994 and 1995.
This subanalysis included 1,655 men aged 55-74 years, who underwent either external beam radiation or radical prostatectomy during that period. They filled out quality of life surveys at baseline, and at 6 months and 1, 2, 5, and 15 years after their diagnosis. Since younger, healthier men may be more likely to have surgery, the authors used a propensity score analysis in an attempt to control for age. Most of the group (70%) underwent radical prostatectomy; 30% had radiation therapy.
Overall, men who had surgery were significantly more likely to experience urinary and sexual dysfunction, while the radiation group was significantly more likely to experience bowel dysfunction.
The differences were most obvious early in the follow-up period. At 2 and 5 years after treatment, men in the surgery group were significantly more likely to report erectile dysfunction than those were those treated with radiation. (odds ratio, 3.46 and 1.96 respectively). By 15 years, this difference had disappeared, however.
Men who had surgery were also significantly more likely to report having no sexual activity at 2 years (OR, 1.83), but at 5 and 15 years, there were no significant differences between the groups.
The surgical group was more likely to report urinary dysfunction compared with the radiation-treated cohort. Having no urinary control was significantly more likely at 2 and 5 years (OR, 6.2 and 5.1 respectively), but not at 15 years. Urine leakage was also significantly more likely in the surgery group at 2 and 5 years (OR, 13.0 and 5.5, respectively), but not at 15 years.
The need for pads to stay dry was significantly more likely at both 2 and 5 years for the surgery group (OR, 26 and 12, respectively). The surgery group was still significantly more likely to need pads at 15 years, although the difference was somewhat attenuated (OR, 4.5).
Not surprisingly, Dr. Resnick said, men in the surgery group were also significantly more likely to report being bothered by their urinary problems at 2 and 5 years, (OR, 6 and 7, respectively), but not at 15 years.
Finally, patients who underwent radiation therapy were more likely to be bothered by bowel dysfunction throughout the follow-up period; by 15 years after treatment, bowel issues were 3.4 times more common among this group.
While more studies on long-term functional outcomes are needed, Dr. Resnick said this information can be helpful in pretreatment decision-making – a time when patients need to weigh the risks of impaired function associated with each treatment paradigm.
"In terms of evaluating the patient up-front, we can use this information to elicit their preferences [for treatment]. That is an important piece of the plan."
The study was funded by the National Cancer Institute. Dr. Resnick had no financial disclosures.
*Headline was revised 07/12/12.
ATLANTA – At 15 years after treatment, disease-related quality of life measures appear similar in men with prostate cancer, regardless of whether they had radical prostatectomy or external radiation beam therapy.
The significant differences in sexual, urinary, and bowel function that were apparent in the first 5 years, largely disappeared as time advanced, Dr. Matthew Resnick reported, based on a new subanalysis of men in the Prostate Cancer Outcomes Study.
The convergence of outcomes may not have so much to do with treatment type as it does advancing age, he said at the annual meeting of the American Urologic Association. This is especially apparent in erectile dysfunction, according to Dr. Resnick, a urologic surgeon at Vanderbilt University Medical Center, Nashville, Tenn.
"Age really is the most significant determinant of decline in erectile function over time," he said. "About 50% of men older than 70 have erectile dysfunction."
The Prostate Cancer Outcomes Study (PCOS) enrolled patients from six Surveillance, Epidemiology, and End Results (SEER) tumor registries to examine long-term functional outcomes in men who were diagnosed with prostate cancer between 1994 and 1995.
This subanalysis included 1,655 men aged 55-74 years, who underwent either external beam radiation or radical prostatectomy during that period. They filled out quality of life surveys at baseline, and at 6 months and 1, 2, 5, and 15 years after their diagnosis. Since younger, healthier men may be more likely to have surgery, the authors used a propensity score analysis in an attempt to control for age. Most of the group (70%) underwent radical prostatectomy; 30% had radiation therapy.
Overall, men who had surgery were significantly more likely to experience urinary and sexual dysfunction, while the radiation group was significantly more likely to experience bowel dysfunction.
The differences were most obvious early in the follow-up period. At 2 and 5 years after treatment, men in the surgery group were significantly more likely to report erectile dysfunction than those were those treated with radiation. (odds ratio, 3.46 and 1.96 respectively). By 15 years, this difference had disappeared, however.
Men who had surgery were also significantly more likely to report having no sexual activity at 2 years (OR, 1.83), but at 5 and 15 years, there were no significant differences between the groups.
The surgical group was more likely to report urinary dysfunction compared with the radiation-treated cohort. Having no urinary control was significantly more likely at 2 and 5 years (OR, 6.2 and 5.1 respectively), but not at 15 years. Urine leakage was also significantly more likely in the surgery group at 2 and 5 years (OR, 13.0 and 5.5, respectively), but not at 15 years.
The need for pads to stay dry was significantly more likely at both 2 and 5 years for the surgery group (OR, 26 and 12, respectively). The surgery group was still significantly more likely to need pads at 15 years, although the difference was somewhat attenuated (OR, 4.5).
Not surprisingly, Dr. Resnick said, men in the surgery group were also significantly more likely to report being bothered by their urinary problems at 2 and 5 years, (OR, 6 and 7, respectively), but not at 15 years.
Finally, patients who underwent radiation therapy were more likely to be bothered by bowel dysfunction throughout the follow-up period; by 15 years after treatment, bowel issues were 3.4 times more common among this group.
While more studies on long-term functional outcomes are needed, Dr. Resnick said this information can be helpful in pretreatment decision-making – a time when patients need to weigh the risks of impaired function associated with each treatment paradigm.
"In terms of evaluating the patient up-front, we can use this information to elicit their preferences [for treatment]. That is an important piece of the plan."
The study was funded by the National Cancer Institute. Dr. Resnick had no financial disclosures.
*Headline was revised 07/12/12.
ATLANTA – At 15 years after treatment, disease-related quality of life measures appear similar in men with prostate cancer, regardless of whether they had radical prostatectomy or external radiation beam therapy.
The significant differences in sexual, urinary, and bowel function that were apparent in the first 5 years, largely disappeared as time advanced, Dr. Matthew Resnick reported, based on a new subanalysis of men in the Prostate Cancer Outcomes Study.
The convergence of outcomes may not have so much to do with treatment type as it does advancing age, he said at the annual meeting of the American Urologic Association. This is especially apparent in erectile dysfunction, according to Dr. Resnick, a urologic surgeon at Vanderbilt University Medical Center, Nashville, Tenn.
"Age really is the most significant determinant of decline in erectile function over time," he said. "About 50% of men older than 70 have erectile dysfunction."
The Prostate Cancer Outcomes Study (PCOS) enrolled patients from six Surveillance, Epidemiology, and End Results (SEER) tumor registries to examine long-term functional outcomes in men who were diagnosed with prostate cancer between 1994 and 1995.
This subanalysis included 1,655 men aged 55-74 years, who underwent either external beam radiation or radical prostatectomy during that period. They filled out quality of life surveys at baseline, and at 6 months and 1, 2, 5, and 15 years after their diagnosis. Since younger, healthier men may be more likely to have surgery, the authors used a propensity score analysis in an attempt to control for age. Most of the group (70%) underwent radical prostatectomy; 30% had radiation therapy.
Overall, men who had surgery were significantly more likely to experience urinary and sexual dysfunction, while the radiation group was significantly more likely to experience bowel dysfunction.
The differences were most obvious early in the follow-up period. At 2 and 5 years after treatment, men in the surgery group were significantly more likely to report erectile dysfunction than those were those treated with radiation. (odds ratio, 3.46 and 1.96 respectively). By 15 years, this difference had disappeared, however.
Men who had surgery were also significantly more likely to report having no sexual activity at 2 years (OR, 1.83), but at 5 and 15 years, there were no significant differences between the groups.
The surgical group was more likely to report urinary dysfunction compared with the radiation-treated cohort. Having no urinary control was significantly more likely at 2 and 5 years (OR, 6.2 and 5.1 respectively), but not at 15 years. Urine leakage was also significantly more likely in the surgery group at 2 and 5 years (OR, 13.0 and 5.5, respectively), but not at 15 years.
The need for pads to stay dry was significantly more likely at both 2 and 5 years for the surgery group (OR, 26 and 12, respectively). The surgery group was still significantly more likely to need pads at 15 years, although the difference was somewhat attenuated (OR, 4.5).
Not surprisingly, Dr. Resnick said, men in the surgery group were also significantly more likely to report being bothered by their urinary problems at 2 and 5 years, (OR, 6 and 7, respectively), but not at 15 years.
Finally, patients who underwent radiation therapy were more likely to be bothered by bowel dysfunction throughout the follow-up period; by 15 years after treatment, bowel issues were 3.4 times more common among this group.
While more studies on long-term functional outcomes are needed, Dr. Resnick said this information can be helpful in pretreatment decision-making – a time when patients need to weigh the risks of impaired function associated with each treatment paradigm.
"In terms of evaluating the patient up-front, we can use this information to elicit their preferences [for treatment]. That is an important piece of the plan."
The study was funded by the National Cancer Institute. Dr. Resnick had no financial disclosures.
*Headline was revised 07/12/12.
AT THE ANNUAL MEETING OF THE AMERICAN UROLOGIC ASSOCIATION
Major Finding: Men who underwent surgery were more likely to need pads to stay dry 15 years after treatment (odds ratio, 4.5), while those treated with radiation had more bowel complaints (OR 3.4). Other differences in quality of life were no longer significant.
Data Source: The Prostate Cancer Outcomes Study is a prospective longitudinal analysis of men with clinically localized disease.
Disclosures: The National Cancer Institute funded the study. Dr. Resnick had no financial disclosures.
Genome Sequencing Set to Hunt for Alzheimer's Clues
The largest-ever genome sequencing project is the newest weapon in the Alzheimer’s battle.
This new project will examine the genetic codes of more than 800 people, generating more than 165 terabytes – 165,000 gigabytes – of new information about Alzheimer’s disease and its genetic roots.
"This is the biggest single whole-genome scan that has ever been done in any disease, and in one of the best-characterized research cohorts ever seen," said William Thies, Ph.D. "We are bound to discover new things."
Improvements in both technology and cost make the project feasible for the first time. The genome sequencing will take about 16 weeks from start to finish, said Dr. Thies, chief medical and scientific officer of the Alzheimer’s Association. The project’s price tag is a modest $2 million ($1 million each from the Alzheimer’s Association and the nonprofit Brin Wojcicki Foundation).
The project is an offshoot of the Alzheimer’s Disease Neuroimaging Initiative (ADNI), Dr. Thies said in an interview. ADNI is a global effort to improve ways of identifying the best patients for clinical trials, using brain imaging and other biomarkers. The North American section of ADNI has a pool of 800 patients who have been followed since 2004.
"This will point us to new pathways in the disease process."
The cohort "has been characterized to an extent that is almost unheard of in genetic studies," Dr. Thies said.
Upon enrollment, subjects are stratified as having normal cognition, mild cognitive impairment, or Alzheimer’s disease. Close follow-up allows researchers to examine the natural history of Alzheimer’s progression and to identifying cognitive and physiological changes as they occur, as well as risk factors that predispose to disease.
Three genetic markers for familial (or early-onset) Alzheimer’s have been discovered so far: mutations in the genes for presenilin-1 and -2 and the amyloid precursor protein. The e4 allele of the gene for apolipoprotein E is known to have the greatest impact on risk for sporadic (or late-onset) Alzheimer’s.
Genomic sequencing of the ADNI cohort adds a new reach to its capabilities.
"I’m absolutely certain we will learn much more about how to diagnose and treat the disease as we look at this new genomic information," Dr. Thies said.
Like all of ADNI’s data, the genomic information will be publicly available to researchers all over the world. Public data sharing in research is a relatively new concept, and one that greatly increases the chances of success, he said. "More papers have been published outside of ADNI than by researchers in the group. That is really impressive. Sharing is clearly the road to progress."
How any new genetic information might be used is still an unknown, Dr. Thies said, but it’s likely to advance every aspect of Alzheimer’s care, from risk assessment to diagnosis to treatment.
"These kinds of data offer the possibility of being able to test people early in life and establish their risk of developing Alzheimer’s. Having that information is going to very rapidly put us into the world of risk stratification, so that we can optimally manage any interventions we develop."
Genetic information will also inform the search for preventive therapies, he added.
"This will point us to new pathways in the disease process. Once those are identified, we can try to find early interventions that might help reduce the risk of getting Alzheimer’s."
Some early work on the genetics of disease pathways will be featured at the upcoming Alzheimer’s Association International Conference in Vancouver, B.C.
"We are seeing risk genes beginning to clump around a number of areas, including those that have to do with amyloid, inflammation, vascular areas, and lipid metabolism," Dr. Thies said. "These genes may end up showing just a modest effect on risk, but could still open the door to early intervention. If you can identify and treat someone for 20 years, a small but consistent effect may be enough to prevent Alzheimer’s from developing."
Dr. Robert Green of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, will lead the project.
ADNI is funded by a number of bodies, including the National Institute on Aging and the National Institute of Biomedical Imaging and Bioengineering, pharmaceutical companies, and nonprofit groups.
The largest-ever genome sequencing project is the newest weapon in the Alzheimer’s battle.
This new project will examine the genetic codes of more than 800 people, generating more than 165 terabytes – 165,000 gigabytes – of new information about Alzheimer’s disease and its genetic roots.
"This is the biggest single whole-genome scan that has ever been done in any disease, and in one of the best-characterized research cohorts ever seen," said William Thies, Ph.D. "We are bound to discover new things."
Improvements in both technology and cost make the project feasible for the first time. The genome sequencing will take about 16 weeks from start to finish, said Dr. Thies, chief medical and scientific officer of the Alzheimer’s Association. The project’s price tag is a modest $2 million ($1 million each from the Alzheimer’s Association and the nonprofit Brin Wojcicki Foundation).
The project is an offshoot of the Alzheimer’s Disease Neuroimaging Initiative (ADNI), Dr. Thies said in an interview. ADNI is a global effort to improve ways of identifying the best patients for clinical trials, using brain imaging and other biomarkers. The North American section of ADNI has a pool of 800 patients who have been followed since 2004.
"This will point us to new pathways in the disease process."
The cohort "has been characterized to an extent that is almost unheard of in genetic studies," Dr. Thies said.
Upon enrollment, subjects are stratified as having normal cognition, mild cognitive impairment, or Alzheimer’s disease. Close follow-up allows researchers to examine the natural history of Alzheimer’s progression and to identifying cognitive and physiological changes as they occur, as well as risk factors that predispose to disease.
Three genetic markers for familial (or early-onset) Alzheimer’s have been discovered so far: mutations in the genes for presenilin-1 and -2 and the amyloid precursor protein. The e4 allele of the gene for apolipoprotein E is known to have the greatest impact on risk for sporadic (or late-onset) Alzheimer’s.
Genomic sequencing of the ADNI cohort adds a new reach to its capabilities.
"I’m absolutely certain we will learn much more about how to diagnose and treat the disease as we look at this new genomic information," Dr. Thies said.
Like all of ADNI’s data, the genomic information will be publicly available to researchers all over the world. Public data sharing in research is a relatively new concept, and one that greatly increases the chances of success, he said. "More papers have been published outside of ADNI than by researchers in the group. That is really impressive. Sharing is clearly the road to progress."
How any new genetic information might be used is still an unknown, Dr. Thies said, but it’s likely to advance every aspect of Alzheimer’s care, from risk assessment to diagnosis to treatment.
"These kinds of data offer the possibility of being able to test people early in life and establish their risk of developing Alzheimer’s. Having that information is going to very rapidly put us into the world of risk stratification, so that we can optimally manage any interventions we develop."
Genetic information will also inform the search for preventive therapies, he added.
"This will point us to new pathways in the disease process. Once those are identified, we can try to find early interventions that might help reduce the risk of getting Alzheimer’s."
Some early work on the genetics of disease pathways will be featured at the upcoming Alzheimer’s Association International Conference in Vancouver, B.C.
"We are seeing risk genes beginning to clump around a number of areas, including those that have to do with amyloid, inflammation, vascular areas, and lipid metabolism," Dr. Thies said. "These genes may end up showing just a modest effect on risk, but could still open the door to early intervention. If you can identify and treat someone for 20 years, a small but consistent effect may be enough to prevent Alzheimer’s from developing."
Dr. Robert Green of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, will lead the project.
ADNI is funded by a number of bodies, including the National Institute on Aging and the National Institute of Biomedical Imaging and Bioengineering, pharmaceutical companies, and nonprofit groups.
The largest-ever genome sequencing project is the newest weapon in the Alzheimer’s battle.
This new project will examine the genetic codes of more than 800 people, generating more than 165 terabytes – 165,000 gigabytes – of new information about Alzheimer’s disease and its genetic roots.
"This is the biggest single whole-genome scan that has ever been done in any disease, and in one of the best-characterized research cohorts ever seen," said William Thies, Ph.D. "We are bound to discover new things."
Improvements in both technology and cost make the project feasible for the first time. The genome sequencing will take about 16 weeks from start to finish, said Dr. Thies, chief medical and scientific officer of the Alzheimer’s Association. The project’s price tag is a modest $2 million ($1 million each from the Alzheimer’s Association and the nonprofit Brin Wojcicki Foundation).
The project is an offshoot of the Alzheimer’s Disease Neuroimaging Initiative (ADNI), Dr. Thies said in an interview. ADNI is a global effort to improve ways of identifying the best patients for clinical trials, using brain imaging and other biomarkers. The North American section of ADNI has a pool of 800 patients who have been followed since 2004.
"This will point us to new pathways in the disease process."
The cohort "has been characterized to an extent that is almost unheard of in genetic studies," Dr. Thies said.
Upon enrollment, subjects are stratified as having normal cognition, mild cognitive impairment, or Alzheimer’s disease. Close follow-up allows researchers to examine the natural history of Alzheimer’s progression and to identifying cognitive and physiological changes as they occur, as well as risk factors that predispose to disease.
Three genetic markers for familial (or early-onset) Alzheimer’s have been discovered so far: mutations in the genes for presenilin-1 and -2 and the amyloid precursor protein. The e4 allele of the gene for apolipoprotein E is known to have the greatest impact on risk for sporadic (or late-onset) Alzheimer’s.
Genomic sequencing of the ADNI cohort adds a new reach to its capabilities.
"I’m absolutely certain we will learn much more about how to diagnose and treat the disease as we look at this new genomic information," Dr. Thies said.
Like all of ADNI’s data, the genomic information will be publicly available to researchers all over the world. Public data sharing in research is a relatively new concept, and one that greatly increases the chances of success, he said. "More papers have been published outside of ADNI than by researchers in the group. That is really impressive. Sharing is clearly the road to progress."
How any new genetic information might be used is still an unknown, Dr. Thies said, but it’s likely to advance every aspect of Alzheimer’s care, from risk assessment to diagnosis to treatment.
"These kinds of data offer the possibility of being able to test people early in life and establish their risk of developing Alzheimer’s. Having that information is going to very rapidly put us into the world of risk stratification, so that we can optimally manage any interventions we develop."
Genetic information will also inform the search for preventive therapies, he added.
"This will point us to new pathways in the disease process. Once those are identified, we can try to find early interventions that might help reduce the risk of getting Alzheimer’s."
Some early work on the genetics of disease pathways will be featured at the upcoming Alzheimer’s Association International Conference in Vancouver, B.C.
"We are seeing risk genes beginning to clump around a number of areas, including those that have to do with amyloid, inflammation, vascular areas, and lipid metabolism," Dr. Thies said. "These genes may end up showing just a modest effect on risk, but could still open the door to early intervention. If you can identify and treat someone for 20 years, a small but consistent effect may be enough to prevent Alzheimer’s from developing."
Dr. Robert Green of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, will lead the project.
ADNI is funded by a number of bodies, including the National Institute on Aging and the National Institute of Biomedical Imaging and Bioengineering, pharmaceutical companies, and nonprofit groups.