User login
FDA okays new indication for esketamine nasal spray
The Food and Drug Administration has approved the supplemental new drug application for esketamine nasal spray (Spravato, Janssen Pharmaceuticals) to treat depressive symptoms in adults with major depressive disorder (MDD) and acute suicidal ideation or behavior.
The FDA approved esketamine nasal spray for treatment-resistant depression in March 2019, as reported by Medscape Medical News.
– which evaluated the efficacy and safety of the nasal spray in addition to a comprehensive standard of care in adults with MDD who had active suicidal ideation with intent.
The standard of care included initial hospitalization, a newly initiated or optimized oral antidepressant, and twice-weekly treatment visits for 4 weeks. During that time, patients received esketamine nasal spray 84 mg or placebo nasal spray.
Results from the trials showed that the active treatment significantly reduced depressive symptoms within 24 hours, with some patients starting to respond as early as 4 hours after the first dose.
“Traditional oral antidepressants need weeks or more to take effect, so the availability of a medicine that can begin providing relief within a day is potentially life changing,” Theresa Nguyen, chief program officer at Mental Health America, said in a company news release.
“The clinical trials supporting this new indication provide compelling evidence that esketamine may offer clinicians a new way to provide support to patients quickly in the midst of an urgent depressive episode and help set them on the path to remission,” Gerard Sanacora, MD, PhD, director of the Yale Depression Research Program, New Haven, Conn., and esketamine clinical trial investigator, said in the same release.
A full course of treatment for MDD with acute suicidal ideation or behavior is twice weekly for 4 weeks, “after which evidence of therapeutic benefit should be evaluated to determine need for continued treatment,” the company said.
Because of the risk for serious adverse events, including sedation and dissociation, and the potential for abuse or misuse, esketamine nasal spray is only available through a restricted distribution system – the Spravato Risk Evaluation and Mitigation Strategy (REMS).
The patient self-administers esketamine nasal spray only in REMS-certified health care settings. Patients are not permitted to take the drug home.
Full prescribing information is available online.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has approved the supplemental new drug application for esketamine nasal spray (Spravato, Janssen Pharmaceuticals) to treat depressive symptoms in adults with major depressive disorder (MDD) and acute suicidal ideation or behavior.
The FDA approved esketamine nasal spray for treatment-resistant depression in March 2019, as reported by Medscape Medical News.
– which evaluated the efficacy and safety of the nasal spray in addition to a comprehensive standard of care in adults with MDD who had active suicidal ideation with intent.
The standard of care included initial hospitalization, a newly initiated or optimized oral antidepressant, and twice-weekly treatment visits for 4 weeks. During that time, patients received esketamine nasal spray 84 mg or placebo nasal spray.
Results from the trials showed that the active treatment significantly reduced depressive symptoms within 24 hours, with some patients starting to respond as early as 4 hours after the first dose.
“Traditional oral antidepressants need weeks or more to take effect, so the availability of a medicine that can begin providing relief within a day is potentially life changing,” Theresa Nguyen, chief program officer at Mental Health America, said in a company news release.
“The clinical trials supporting this new indication provide compelling evidence that esketamine may offer clinicians a new way to provide support to patients quickly in the midst of an urgent depressive episode and help set them on the path to remission,” Gerard Sanacora, MD, PhD, director of the Yale Depression Research Program, New Haven, Conn., and esketamine clinical trial investigator, said in the same release.
A full course of treatment for MDD with acute suicidal ideation or behavior is twice weekly for 4 weeks, “after which evidence of therapeutic benefit should be evaluated to determine need for continued treatment,” the company said.
Because of the risk for serious adverse events, including sedation and dissociation, and the potential for abuse or misuse, esketamine nasal spray is only available through a restricted distribution system – the Spravato Risk Evaluation and Mitigation Strategy (REMS).
The patient self-administers esketamine nasal spray only in REMS-certified health care settings. Patients are not permitted to take the drug home.
Full prescribing information is available online.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has approved the supplemental new drug application for esketamine nasal spray (Spravato, Janssen Pharmaceuticals) to treat depressive symptoms in adults with major depressive disorder (MDD) and acute suicidal ideation or behavior.
The FDA approved esketamine nasal spray for treatment-resistant depression in March 2019, as reported by Medscape Medical News.
– which evaluated the efficacy and safety of the nasal spray in addition to a comprehensive standard of care in adults with MDD who had active suicidal ideation with intent.
The standard of care included initial hospitalization, a newly initiated or optimized oral antidepressant, and twice-weekly treatment visits for 4 weeks. During that time, patients received esketamine nasal spray 84 mg or placebo nasal spray.
Results from the trials showed that the active treatment significantly reduced depressive symptoms within 24 hours, with some patients starting to respond as early as 4 hours after the first dose.
“Traditional oral antidepressants need weeks or more to take effect, so the availability of a medicine that can begin providing relief within a day is potentially life changing,” Theresa Nguyen, chief program officer at Mental Health America, said in a company news release.
“The clinical trials supporting this new indication provide compelling evidence that esketamine may offer clinicians a new way to provide support to patients quickly in the midst of an urgent depressive episode and help set them on the path to remission,” Gerard Sanacora, MD, PhD, director of the Yale Depression Research Program, New Haven, Conn., and esketamine clinical trial investigator, said in the same release.
A full course of treatment for MDD with acute suicidal ideation or behavior is twice weekly for 4 weeks, “after which evidence of therapeutic benefit should be evaluated to determine need for continued treatment,” the company said.
Because of the risk for serious adverse events, including sedation and dissociation, and the potential for abuse or misuse, esketamine nasal spray is only available through a restricted distribution system – the Spravato Risk Evaluation and Mitigation Strategy (REMS).
The patient self-administers esketamine nasal spray only in REMS-certified health care settings. Patients are not permitted to take the drug home.
Full prescribing information is available online.
A version of this article originally appeared on Medscape.com.
Global study to track COVID-19’s impact on the brain
At its annual meeting, the Alzheimer’s Association announced the launch of a global study to examine the impact of COVID-19 on the brain, as well as policy recommendations to better address the COVID-19 crisis in long-term care facilities. The study will be led by researchers at the Alzheimer’s Association and the University of Texas Health, San Antonio, with participation from more than 30 countries and technical guidance from the World Health Organization.
The target sample size is 20,000-40,000 total participants.
Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, announced the study’s launch during a COVID-19–focused panel discussion at the virtual annual meeting of the Alzheimer’s Association International Conference 2020.
“To build a strong foundation for this research, we will align with existing studies, such as the Framingham Heart Study, and clinicians from around the world on how the data are going to be collected, obtained, and shared. We are going to have cross-study collaborations to understand the impact of the virus on the brain directly,” said Dr. Carrillo. “We will have some very good data to present next year at AAIC.”
‘Frightening’ headlines
As previously reported, mounting evidence suggests that SARS-CoV-2 invades the central nervous system, causing a wide range of neurologic and neuropsychiatric complications, including stroke, psychosis, altered mental state, and dementia-like syndrome. It’s likely that “dementia does not increase the risk for COVID-19, just like dementia does not increase risk for the flu. But increased age, being in a long-term care setting, and common health conditions that often accompany dementia may increase the risk,” Dr. Carrillo said.
Panel member Beth Kallmyer, MSW, vice president of care and support at the Alzheimer’s Association, spoke about the ongoing challenges long-term care facilities are facing during the pandemic. “You’ve all seen the headlines, and they’re frightening, frankly,” she said. An estimated 59,000 residents and employees of long-term care have died as a result of COVID-19, which is 42% of all U.S. deaths.
The long-term care community is being impacted at “significantly greater rates than the rest of society and yet we don’t have things in place to protect them. We also know that individuals living with dementia make up a large percentage of those that are living in long-term care,” Ms. Kallmyer said.
She noted that infection control is always a challenge in long-term care settings, but infection control during a pandemic “takes it to a whole other level.” Quarantining is hard for anyone, “but when you layer dementia on top of that we have a real challenge.” One long-term care provider told Ms. Kallmyer that “we might be saving them from COVID, but we’re losing them to social isolation and cognitive decline.”
New recommendations
Ms. Kallmyer outlined new policy recommendations from the Alzheimer’s Association to address the COVID-19 crisis in long-term and community-based care settings. They include:
- Testing every resident, employee, and visitor each time they leave and come back, so residents would not need to be confined to their own rooms
- Having a single portal that is easy and efficient for reporting cases
- Developing “surge activation” protocols to respond to hot spots, including the possibility of “strike teams” that go in and help during an outbreak
- Making sure all long-term care providers have full access to all needed personal protective equipment (PPE)
“Five months in and long-term care providers still don’t have adequate PPE. This is unacceptable,” said Ms. Kallmyer. “We have to be able to provide them with PPE.”
Panel member Gregory A. Jicha, MD, PhD, Sanders-Brown Center on Aging, University of Kentucky, Lexington, spoke about the critical need to continue Alzheimer’s disease research during the pandemic, noting that the number of promising targets for Alzheimer’s disease and related dementias has “never been higher or more comprehensive.”
Measures to ensure safety of researchers and participants include screening for symptoms (50% effective), social distancing (93% effective), minimizing exposure time (50% effective), limiting staff to 50% (50% effective), cloth/paper masks (80% effective), and testing (99.25% effective), Dr. Jicha noted.
With no safety measures in place, the risk of getting COVID-19 from a research visit is 1 in 20; when all these safety measures are combined, the risk is 1 in over 1.5 million, so “we can essentially eradicate or minimize the risks for COVID to less that of a lightning strike,” he said.
Dr. Carrillo, Ms. Kallmyer, and Dr. Jicha disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
At its annual meeting, the Alzheimer’s Association announced the launch of a global study to examine the impact of COVID-19 on the brain, as well as policy recommendations to better address the COVID-19 crisis in long-term care facilities. The study will be led by researchers at the Alzheimer’s Association and the University of Texas Health, San Antonio, with participation from more than 30 countries and technical guidance from the World Health Organization.
The target sample size is 20,000-40,000 total participants.
Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, announced the study’s launch during a COVID-19–focused panel discussion at the virtual annual meeting of the Alzheimer’s Association International Conference 2020.
“To build a strong foundation for this research, we will align with existing studies, such as the Framingham Heart Study, and clinicians from around the world on how the data are going to be collected, obtained, and shared. We are going to have cross-study collaborations to understand the impact of the virus on the brain directly,” said Dr. Carrillo. “We will have some very good data to present next year at AAIC.”
‘Frightening’ headlines
As previously reported, mounting evidence suggests that SARS-CoV-2 invades the central nervous system, causing a wide range of neurologic and neuropsychiatric complications, including stroke, psychosis, altered mental state, and dementia-like syndrome. It’s likely that “dementia does not increase the risk for COVID-19, just like dementia does not increase risk for the flu. But increased age, being in a long-term care setting, and common health conditions that often accompany dementia may increase the risk,” Dr. Carrillo said.
Panel member Beth Kallmyer, MSW, vice president of care and support at the Alzheimer’s Association, spoke about the ongoing challenges long-term care facilities are facing during the pandemic. “You’ve all seen the headlines, and they’re frightening, frankly,” she said. An estimated 59,000 residents and employees of long-term care have died as a result of COVID-19, which is 42% of all U.S. deaths.
The long-term care community is being impacted at “significantly greater rates than the rest of society and yet we don’t have things in place to protect them. We also know that individuals living with dementia make up a large percentage of those that are living in long-term care,” Ms. Kallmyer said.
She noted that infection control is always a challenge in long-term care settings, but infection control during a pandemic “takes it to a whole other level.” Quarantining is hard for anyone, “but when you layer dementia on top of that we have a real challenge.” One long-term care provider told Ms. Kallmyer that “we might be saving them from COVID, but we’re losing them to social isolation and cognitive decline.”
New recommendations
Ms. Kallmyer outlined new policy recommendations from the Alzheimer’s Association to address the COVID-19 crisis in long-term and community-based care settings. They include:
- Testing every resident, employee, and visitor each time they leave and come back, so residents would not need to be confined to their own rooms
- Having a single portal that is easy and efficient for reporting cases
- Developing “surge activation” protocols to respond to hot spots, including the possibility of “strike teams” that go in and help during an outbreak
- Making sure all long-term care providers have full access to all needed personal protective equipment (PPE)
“Five months in and long-term care providers still don’t have adequate PPE. This is unacceptable,” said Ms. Kallmyer. “We have to be able to provide them with PPE.”
Panel member Gregory A. Jicha, MD, PhD, Sanders-Brown Center on Aging, University of Kentucky, Lexington, spoke about the critical need to continue Alzheimer’s disease research during the pandemic, noting that the number of promising targets for Alzheimer’s disease and related dementias has “never been higher or more comprehensive.”
Measures to ensure safety of researchers and participants include screening for symptoms (50% effective), social distancing (93% effective), minimizing exposure time (50% effective), limiting staff to 50% (50% effective), cloth/paper masks (80% effective), and testing (99.25% effective), Dr. Jicha noted.
With no safety measures in place, the risk of getting COVID-19 from a research visit is 1 in 20; when all these safety measures are combined, the risk is 1 in over 1.5 million, so “we can essentially eradicate or minimize the risks for COVID to less that of a lightning strike,” he said.
Dr. Carrillo, Ms. Kallmyer, and Dr. Jicha disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
At its annual meeting, the Alzheimer’s Association announced the launch of a global study to examine the impact of COVID-19 on the brain, as well as policy recommendations to better address the COVID-19 crisis in long-term care facilities. The study will be led by researchers at the Alzheimer’s Association and the University of Texas Health, San Antonio, with participation from more than 30 countries and technical guidance from the World Health Organization.
The target sample size is 20,000-40,000 total participants.
Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, announced the study’s launch during a COVID-19–focused panel discussion at the virtual annual meeting of the Alzheimer’s Association International Conference 2020.
“To build a strong foundation for this research, we will align with existing studies, such as the Framingham Heart Study, and clinicians from around the world on how the data are going to be collected, obtained, and shared. We are going to have cross-study collaborations to understand the impact of the virus on the brain directly,” said Dr. Carrillo. “We will have some very good data to present next year at AAIC.”
‘Frightening’ headlines
As previously reported, mounting evidence suggests that SARS-CoV-2 invades the central nervous system, causing a wide range of neurologic and neuropsychiatric complications, including stroke, psychosis, altered mental state, and dementia-like syndrome. It’s likely that “dementia does not increase the risk for COVID-19, just like dementia does not increase risk for the flu. But increased age, being in a long-term care setting, and common health conditions that often accompany dementia may increase the risk,” Dr. Carrillo said.
Panel member Beth Kallmyer, MSW, vice president of care and support at the Alzheimer’s Association, spoke about the ongoing challenges long-term care facilities are facing during the pandemic. “You’ve all seen the headlines, and they’re frightening, frankly,” she said. An estimated 59,000 residents and employees of long-term care have died as a result of COVID-19, which is 42% of all U.S. deaths.
The long-term care community is being impacted at “significantly greater rates than the rest of society and yet we don’t have things in place to protect them. We also know that individuals living with dementia make up a large percentage of those that are living in long-term care,” Ms. Kallmyer said.
She noted that infection control is always a challenge in long-term care settings, but infection control during a pandemic “takes it to a whole other level.” Quarantining is hard for anyone, “but when you layer dementia on top of that we have a real challenge.” One long-term care provider told Ms. Kallmyer that “we might be saving them from COVID, but we’re losing them to social isolation and cognitive decline.”
New recommendations
Ms. Kallmyer outlined new policy recommendations from the Alzheimer’s Association to address the COVID-19 crisis in long-term and community-based care settings. They include:
- Testing every resident, employee, and visitor each time they leave and come back, so residents would not need to be confined to their own rooms
- Having a single portal that is easy and efficient for reporting cases
- Developing “surge activation” protocols to respond to hot spots, including the possibility of “strike teams” that go in and help during an outbreak
- Making sure all long-term care providers have full access to all needed personal protective equipment (PPE)
“Five months in and long-term care providers still don’t have adequate PPE. This is unacceptable,” said Ms. Kallmyer. “We have to be able to provide them with PPE.”
Panel member Gregory A. Jicha, MD, PhD, Sanders-Brown Center on Aging, University of Kentucky, Lexington, spoke about the critical need to continue Alzheimer’s disease research during the pandemic, noting that the number of promising targets for Alzheimer’s disease and related dementias has “never been higher or more comprehensive.”
Measures to ensure safety of researchers and participants include screening for symptoms (50% effective), social distancing (93% effective), minimizing exposure time (50% effective), limiting staff to 50% (50% effective), cloth/paper masks (80% effective), and testing (99.25% effective), Dr. Jicha noted.
With no safety measures in place, the risk of getting COVID-19 from a research visit is 1 in 20; when all these safety measures are combined, the risk is 1 in over 1.5 million, so “we can essentially eradicate or minimize the risks for COVID to less that of a lightning strike,” he said.
Dr. Carrillo, Ms. Kallmyer, and Dr. Jicha disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AAIC 2020
Infection ups mortality risk in patients with dementia
Infection increases mortality risk among patients with dementia, new research suggests. A large, registry-based cohort study showed that
“This is the first study to our knowledge to show that increased mortality is observed across all infection types in people with dementia and that increased mortality is seen both short and long term,” said coinvestigator Janet Janbek, a PhD student at the Danish Dementia Research Center, Rigshospitalet, University of Copenhagen.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Large Danish cohort
The investigators analyzed data from Danish national health registries for nearly 1.5 million individuals aged 65 years and older who had visited the hospital with an infection. There were 575,260 deaths during more than 12.7 million person-years of follow-up.
Patients with dementia who also had a hospital visit for infection died at a 6.5 times higher rate than participants without dementia or an infection. Those with either dementia alone or infection-related contacts alone had a threefold increased rate of death.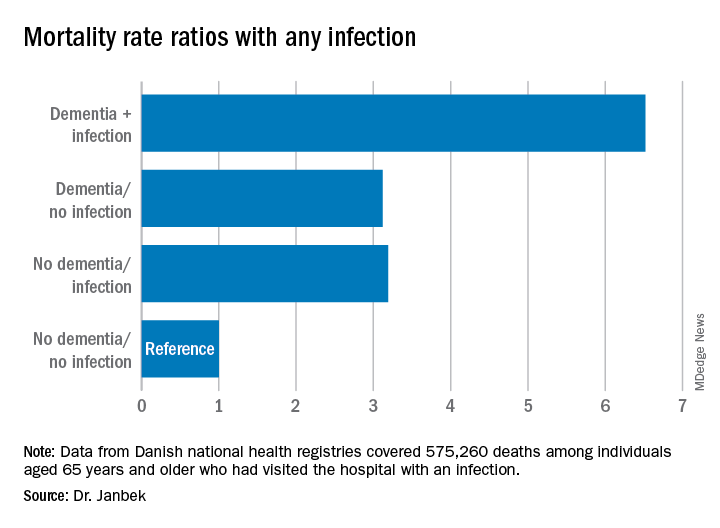
The mortality rate was highest within the first 30 days following the hospital visit for infection. However, the rate remained elevated for 10 years after the initial infection-related hospital visit.
Mortality rates from all infections, including major infections, such as sepsis, down to minor ear infections were elevated in patients with dementia, compared with people who did not have dementia or an infection-related hospital visit.
Ms. Janbek said there are several possible explanations for the association of infection and increased mortality risk in those with dementia. “After a hospital contact with a severe infection, people with dementia may become more reliant on external care, become more frail, and have declined functional levels, which might explain the observed association.”
It might also be that patients with dementia have more severe infections than those without dementia at the time of hospital contact, possibly because of delayed diagnosis, which could explain the higher mortality rates, said Ms. Janbek.
“It is also plausible that infections play a role in worsening dementia and subsequently lead to increased mortality,” she noted.
“Clinicians and health care personnel need to pay closer attention to infections of all types in people with dementia, and steps toward better clinical management and improved posthospital care need to be explored and undertaken. We need to identify possible preventive measures and targeted interventions in people with dementia and infections,” Ms. Janbek said.
‘Interesting observation’
Commenting on the study, Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, said it presents “an interesting observation.” However, “we can’t make any direct assumptions from this research per se about infections and dementia and whether they are causative in any way,” noted Dr. Edelmayer, who was not involved with the study.
Instead, the study highlighted the importance of “taking care of our overall health and making sure that individuals that might be vulnerable to infection, like those who are already living with dementia, are getting the best care possible,” she said.
Ms. Janbek and Dr. Edelmayer have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Infection increases mortality risk among patients with dementia, new research suggests. A large, registry-based cohort study showed that
“This is the first study to our knowledge to show that increased mortality is observed across all infection types in people with dementia and that increased mortality is seen both short and long term,” said coinvestigator Janet Janbek, a PhD student at the Danish Dementia Research Center, Rigshospitalet, University of Copenhagen.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Large Danish cohort
The investigators analyzed data from Danish national health registries for nearly 1.5 million individuals aged 65 years and older who had visited the hospital with an infection. There were 575,260 deaths during more than 12.7 million person-years of follow-up.
Patients with dementia who also had a hospital visit for infection died at a 6.5 times higher rate than participants without dementia or an infection. Those with either dementia alone or infection-related contacts alone had a threefold increased rate of death.
The mortality rate was highest within the first 30 days following the hospital visit for infection. However, the rate remained elevated for 10 years after the initial infection-related hospital visit.
Mortality rates from all infections, including major infections, such as sepsis, down to minor ear infections were elevated in patients with dementia, compared with people who did not have dementia or an infection-related hospital visit.
Ms. Janbek said there are several possible explanations for the association of infection and increased mortality risk in those with dementia. “After a hospital contact with a severe infection, people with dementia may become more reliant on external care, become more frail, and have declined functional levels, which might explain the observed association.”
It might also be that patients with dementia have more severe infections than those without dementia at the time of hospital contact, possibly because of delayed diagnosis, which could explain the higher mortality rates, said Ms. Janbek.
“It is also plausible that infections play a role in worsening dementia and subsequently lead to increased mortality,” she noted.
“Clinicians and health care personnel need to pay closer attention to infections of all types in people with dementia, and steps toward better clinical management and improved posthospital care need to be explored and undertaken. We need to identify possible preventive measures and targeted interventions in people with dementia and infections,” Ms. Janbek said.
‘Interesting observation’
Commenting on the study, Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, said it presents “an interesting observation.” However, “we can’t make any direct assumptions from this research per se about infections and dementia and whether they are causative in any way,” noted Dr. Edelmayer, who was not involved with the study.
Instead, the study highlighted the importance of “taking care of our overall health and making sure that individuals that might be vulnerable to infection, like those who are already living with dementia, are getting the best care possible,” she said.
Ms. Janbek and Dr. Edelmayer have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Infection increases mortality risk among patients with dementia, new research suggests. A large, registry-based cohort study showed that
“This is the first study to our knowledge to show that increased mortality is observed across all infection types in people with dementia and that increased mortality is seen both short and long term,” said coinvestigator Janet Janbek, a PhD student at the Danish Dementia Research Center, Rigshospitalet, University of Copenhagen.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Large Danish cohort
The investigators analyzed data from Danish national health registries for nearly 1.5 million individuals aged 65 years and older who had visited the hospital with an infection. There were 575,260 deaths during more than 12.7 million person-years of follow-up.
Patients with dementia who also had a hospital visit for infection died at a 6.5 times higher rate than participants without dementia or an infection. Those with either dementia alone or infection-related contacts alone had a threefold increased rate of death.
The mortality rate was highest within the first 30 days following the hospital visit for infection. However, the rate remained elevated for 10 years after the initial infection-related hospital visit.
Mortality rates from all infections, including major infections, such as sepsis, down to minor ear infections were elevated in patients with dementia, compared with people who did not have dementia or an infection-related hospital visit.
Ms. Janbek said there are several possible explanations for the association of infection and increased mortality risk in those with dementia. “After a hospital contact with a severe infection, people with dementia may become more reliant on external care, become more frail, and have declined functional levels, which might explain the observed association.”
It might also be that patients with dementia have more severe infections than those without dementia at the time of hospital contact, possibly because of delayed diagnosis, which could explain the higher mortality rates, said Ms. Janbek.
“It is also plausible that infections play a role in worsening dementia and subsequently lead to increased mortality,” she noted.
“Clinicians and health care personnel need to pay closer attention to infections of all types in people with dementia, and steps toward better clinical management and improved posthospital care need to be explored and undertaken. We need to identify possible preventive measures and targeted interventions in people with dementia and infections,” Ms. Janbek said.
‘Interesting observation’
Commenting on the study, Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, said it presents “an interesting observation.” However, “we can’t make any direct assumptions from this research per se about infections and dementia and whether they are causative in any way,” noted Dr. Edelmayer, who was not involved with the study.
Instead, the study highlighted the importance of “taking care of our overall health and making sure that individuals that might be vulnerable to infection, like those who are already living with dementia, are getting the best care possible,” she said.
Ms. Janbek and Dr. Edelmayer have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AAIC 2020
P-tau217 differentiates Alzheimer’s disease from other neurodegenerative conditions
new research suggests.
Results from a large multinational study showed that the level of P-tau217 in blood collected during life was an accurate predictor of tau brain changes seen in brain tissue after death. In addition, increasing blood P-tau217 levels can be detected in some individuals up to 20 years before the average age of onset of the early cognitive decline that signals Alzheimer’s disease, researchers reported.
“While there is still more work to be done, this biomarker has the potential to have a transformational impact on research, treatment, prevention, and therapy development, and in the clinical setting,” said senior author Eric M. Reiman, MD, executive director of Banner Alzheimer’s Institute in Phoenix.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference and simultaneously published online July 28 in JAMA.
Three cohorts
The international team of researchers evaluated the P-tau217 blood test in 1,402 adults from three cohorts. The first cohort was made up of 81 individuals in the Arizona (Banner Sun Health Research Institute) Brain Donation program and included clinical, blood, and neuropathologic data. The second cohort included 699 individuals in the Swedish BioFINDER-2 study and provided clinical, brain imaging, cerebrospinal fluid (CSF), and blood data. The third cohort was made up of 522 participants from the Columbian autosomal-dominant Alzheimer’s disease kindred, including 365 PSEN1 E280A mutation carriers and 257 mutation noncarriers.
In the Arizona cohort, plasma P-tau217 discriminated neuropathologically defined Alzheimer’s disease from non-Alzheimer’s disease (area under the curve, 0.89; 95% CI, 0.81-0.97) with significantly higher accuracy than plasma P-tau181 and neurofilament light chain (NfL) (AUC range, 0.50-0.72; P < .05).
In the Swedish BioFINDER-2 cohort, the discriminative accuracy of plasma P-tau217 for clinical Alzheimer’s disease dementia versus other neurodegenerative diseases was 96% (AUC, 0.96; 95% CI, 0.93-0.98).
This was significantly higher than plasma P-tau181, plasma NfL, and MRI measures (AUC range, 0.50-0.81; P < .001), but was not significantly different than CSF P-tau217, CSF P-tau181, and tau-PET (AUC range, 0.90-0.99; P > .15).
In the Colombian cohort, plasma P-tau217 levels were significantly greater among PSEN1 mutation carriers than noncarriers starting at around age 25 years, which is 20 years prior to the estimated onset of mild cognitive impairment among mutation carriers.
Additionally, plasma P-tau217 levels correlated with cerebral tau tangles, and discriminated abnormal versus normal tau-PET scans with significantly higher accuracy than plasma P-tau181, plasma NfL, CSF P-tau181, CSF Abeta42:Abeta40 ratio, and MRI measures.
The blood test “opens the possibility of early diagnosis of Alzheimer’s disease before the dementia stage, which is very important for clinical trials evaluating novel therapies that might stop or slow down the disease process,” presenting author Oskar Hansson, MD, PhD, of Lund (Sweden) University, said in a statement.
Further research is now needed to optimize the P-tau217 blood test, validate the findings in unselected and diverse populations, and determine its potential role in the clinic, the investigators noted.
Potential game changer?
Commenting on the study, Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation, noted his enthusiasm for the test. “This tau blood test will be a real game changer, advancing clinical care and research,” said Dr. Fillit, who was not involved in the research.
“This is a real breakthrough: a simple and accessible blood test that can diagnose Alzheimer’s disease better than the more costly and invasive methods currently available like PET scans and cerebrospinal fluid biomarkers,” he said.
The P-tau217 blood test “is like the equivalent of the cholesterol test for heart disease, but for Alzheimer’s disease,” Dr. Fillit added.
As previously reported, another study presented at AAIC 2020 compared P-tau217 with P-tau181 to determine which could best identify individuals with Alzheimer’s disease. Results showed that, although the two biomarkers were similar overall, P-tau217 had a slight edge in terms of accuracy.
The study by Reiman et al. was funded by the Swedish Research Council, the Knut and Alice Wallenberg Foundation, and the Swedish Alzheimer Foundation. Dr. Hansson reported receiving grants from Roche, Biogen, and Pfizer, and receiving nonfinancial support from GE Healthcare, AVID Radiopharmaceuticals, and Euroimmun. Dr. Reiman has received grants from Roche/Roche Diagnostics and received personal fees from Alkahest, Alzheon, Aural Analytics, Denali, Green Valley, MagQ, Takeda/Zinfandel, and United Neuroscience. He is also a cofounder of AlzPath, which aims to further develop P-tau217 and fluid biomarkers; holds a patent owned by Banner Health for a strategy to use biomarkers to accelerate evaluation of Alzheimer prevention therapies; and is a principal investigator of prevention trials that include research agreements with Genentech/Roche and Novartis/Amgen, PET studies that include research agreements with Avid/Lilly, and several National Institute of Health–supported research studies. Dr. Fillit reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research suggests.
Results from a large multinational study showed that the level of P-tau217 in blood collected during life was an accurate predictor of tau brain changes seen in brain tissue after death. In addition, increasing blood P-tau217 levels can be detected in some individuals up to 20 years before the average age of onset of the early cognitive decline that signals Alzheimer’s disease, researchers reported.
“While there is still more work to be done, this biomarker has the potential to have a transformational impact on research, treatment, prevention, and therapy development, and in the clinical setting,” said senior author Eric M. Reiman, MD, executive director of Banner Alzheimer’s Institute in Phoenix.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference and simultaneously published online July 28 in JAMA.
Three cohorts
The international team of researchers evaluated the P-tau217 blood test in 1,402 adults from three cohorts. The first cohort was made up of 81 individuals in the Arizona (Banner Sun Health Research Institute) Brain Donation program and included clinical, blood, and neuropathologic data. The second cohort included 699 individuals in the Swedish BioFINDER-2 study and provided clinical, brain imaging, cerebrospinal fluid (CSF), and blood data. The third cohort was made up of 522 participants from the Columbian autosomal-dominant Alzheimer’s disease kindred, including 365 PSEN1 E280A mutation carriers and 257 mutation noncarriers.
In the Arizona cohort, plasma P-tau217 discriminated neuropathologically defined Alzheimer’s disease from non-Alzheimer’s disease (area under the curve, 0.89; 95% CI, 0.81-0.97) with significantly higher accuracy than plasma P-tau181 and neurofilament light chain (NfL) (AUC range, 0.50-0.72; P < .05).
In the Swedish BioFINDER-2 cohort, the discriminative accuracy of plasma P-tau217 for clinical Alzheimer’s disease dementia versus other neurodegenerative diseases was 96% (AUC, 0.96; 95% CI, 0.93-0.98).
This was significantly higher than plasma P-tau181, plasma NfL, and MRI measures (AUC range, 0.50-0.81; P < .001), but was not significantly different than CSF P-tau217, CSF P-tau181, and tau-PET (AUC range, 0.90-0.99; P > .15).
In the Colombian cohort, plasma P-tau217 levels were significantly greater among PSEN1 mutation carriers than noncarriers starting at around age 25 years, which is 20 years prior to the estimated onset of mild cognitive impairment among mutation carriers.
Additionally, plasma P-tau217 levels correlated with cerebral tau tangles, and discriminated abnormal versus normal tau-PET scans with significantly higher accuracy than plasma P-tau181, plasma NfL, CSF P-tau181, CSF Abeta42:Abeta40 ratio, and MRI measures.
The blood test “opens the possibility of early diagnosis of Alzheimer’s disease before the dementia stage, which is very important for clinical trials evaluating novel therapies that might stop or slow down the disease process,” presenting author Oskar Hansson, MD, PhD, of Lund (Sweden) University, said in a statement.
Further research is now needed to optimize the P-tau217 blood test, validate the findings in unselected and diverse populations, and determine its potential role in the clinic, the investigators noted.
Potential game changer?
Commenting on the study, Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation, noted his enthusiasm for the test. “This tau blood test will be a real game changer, advancing clinical care and research,” said Dr. Fillit, who was not involved in the research.
“This is a real breakthrough: a simple and accessible blood test that can diagnose Alzheimer’s disease better than the more costly and invasive methods currently available like PET scans and cerebrospinal fluid biomarkers,” he said.
The P-tau217 blood test “is like the equivalent of the cholesterol test for heart disease, but for Alzheimer’s disease,” Dr. Fillit added.
As previously reported, another study presented at AAIC 2020 compared P-tau217 with P-tau181 to determine which could best identify individuals with Alzheimer’s disease. Results showed that, although the two biomarkers were similar overall, P-tau217 had a slight edge in terms of accuracy.
The study by Reiman et al. was funded by the Swedish Research Council, the Knut and Alice Wallenberg Foundation, and the Swedish Alzheimer Foundation. Dr. Hansson reported receiving grants from Roche, Biogen, and Pfizer, and receiving nonfinancial support from GE Healthcare, AVID Radiopharmaceuticals, and Euroimmun. Dr. Reiman has received grants from Roche/Roche Diagnostics and received personal fees from Alkahest, Alzheon, Aural Analytics, Denali, Green Valley, MagQ, Takeda/Zinfandel, and United Neuroscience. He is also a cofounder of AlzPath, which aims to further develop P-tau217 and fluid biomarkers; holds a patent owned by Banner Health for a strategy to use biomarkers to accelerate evaluation of Alzheimer prevention therapies; and is a principal investigator of prevention trials that include research agreements with Genentech/Roche and Novartis/Amgen, PET studies that include research agreements with Avid/Lilly, and several National Institute of Health–supported research studies. Dr. Fillit reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research suggests.
Results from a large multinational study showed that the level of P-tau217 in blood collected during life was an accurate predictor of tau brain changes seen in brain tissue after death. In addition, increasing blood P-tau217 levels can be detected in some individuals up to 20 years before the average age of onset of the early cognitive decline that signals Alzheimer’s disease, researchers reported.
“While there is still more work to be done, this biomarker has the potential to have a transformational impact on research, treatment, prevention, and therapy development, and in the clinical setting,” said senior author Eric M. Reiman, MD, executive director of Banner Alzheimer’s Institute in Phoenix.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference and simultaneously published online July 28 in JAMA.
Three cohorts
The international team of researchers evaluated the P-tau217 blood test in 1,402 adults from three cohorts. The first cohort was made up of 81 individuals in the Arizona (Banner Sun Health Research Institute) Brain Donation program and included clinical, blood, and neuropathologic data. The second cohort included 699 individuals in the Swedish BioFINDER-2 study and provided clinical, brain imaging, cerebrospinal fluid (CSF), and blood data. The third cohort was made up of 522 participants from the Columbian autosomal-dominant Alzheimer’s disease kindred, including 365 PSEN1 E280A mutation carriers and 257 mutation noncarriers.
In the Arizona cohort, plasma P-tau217 discriminated neuropathologically defined Alzheimer’s disease from non-Alzheimer’s disease (area under the curve, 0.89; 95% CI, 0.81-0.97) with significantly higher accuracy than plasma P-tau181 and neurofilament light chain (NfL) (AUC range, 0.50-0.72; P < .05).
In the Swedish BioFINDER-2 cohort, the discriminative accuracy of plasma P-tau217 for clinical Alzheimer’s disease dementia versus other neurodegenerative diseases was 96% (AUC, 0.96; 95% CI, 0.93-0.98).
This was significantly higher than plasma P-tau181, plasma NfL, and MRI measures (AUC range, 0.50-0.81; P < .001), but was not significantly different than CSF P-tau217, CSF P-tau181, and tau-PET (AUC range, 0.90-0.99; P > .15).
In the Colombian cohort, plasma P-tau217 levels were significantly greater among PSEN1 mutation carriers than noncarriers starting at around age 25 years, which is 20 years prior to the estimated onset of mild cognitive impairment among mutation carriers.
Additionally, plasma P-tau217 levels correlated with cerebral tau tangles, and discriminated abnormal versus normal tau-PET scans with significantly higher accuracy than plasma P-tau181, plasma NfL, CSF P-tau181, CSF Abeta42:Abeta40 ratio, and MRI measures.
The blood test “opens the possibility of early diagnosis of Alzheimer’s disease before the dementia stage, which is very important for clinical trials evaluating novel therapies that might stop or slow down the disease process,” presenting author Oskar Hansson, MD, PhD, of Lund (Sweden) University, said in a statement.
Further research is now needed to optimize the P-tau217 blood test, validate the findings in unselected and diverse populations, and determine its potential role in the clinic, the investigators noted.
Potential game changer?
Commenting on the study, Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation, noted his enthusiasm for the test. “This tau blood test will be a real game changer, advancing clinical care and research,” said Dr. Fillit, who was not involved in the research.
“This is a real breakthrough: a simple and accessible blood test that can diagnose Alzheimer’s disease better than the more costly and invasive methods currently available like PET scans and cerebrospinal fluid biomarkers,” he said.
The P-tau217 blood test “is like the equivalent of the cholesterol test for heart disease, but for Alzheimer’s disease,” Dr. Fillit added.
As previously reported, another study presented at AAIC 2020 compared P-tau217 with P-tau181 to determine which could best identify individuals with Alzheimer’s disease. Results showed that, although the two biomarkers were similar overall, P-tau217 had a slight edge in terms of accuracy.
The study by Reiman et al. was funded by the Swedish Research Council, the Knut and Alice Wallenberg Foundation, and the Swedish Alzheimer Foundation. Dr. Hansson reported receiving grants from Roche, Biogen, and Pfizer, and receiving nonfinancial support from GE Healthcare, AVID Radiopharmaceuticals, and Euroimmun. Dr. Reiman has received grants from Roche/Roche Diagnostics and received personal fees from Alkahest, Alzheon, Aural Analytics, Denali, Green Valley, MagQ, Takeda/Zinfandel, and United Neuroscience. He is also a cofounder of AlzPath, which aims to further develop P-tau217 and fluid biomarkers; holds a patent owned by Banner Health for a strategy to use biomarkers to accelerate evaluation of Alzheimer prevention therapies; and is a principal investigator of prevention trials that include research agreements with Genentech/Roche and Novartis/Amgen, PET studies that include research agreements with Avid/Lilly, and several National Institute of Health–supported research studies. Dr. Fillit reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AAIC 2020
Flu and pneumonia vaccination tied to lower dementia risk
In a cohort study of more than 9,000 older adults, receiving a single influenza vaccination was associated with a 17% lower prevalence of Alzheimer’s disease compared with not receiving the vaccine. In addition, for those who were vaccinated more than once over the years, there was an additional 13% reduction in Alzheimer’s disease incidence.
In another study, which included more than 5,000 older participants, being vaccinated against pneumonia between the ages of 65 and 75 reduced the risk of developing Alzheimer’s disease by 30%.
The subject of vaccines “is obviously very topical with the COVID-19 pandemic,” said Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association. “While these are very preliminary data, these studies do suggest that with vaccination against both respiratory illnesses, there is the potential to lower risk for developing cognitive decline and dementia,” said Dr. Edelmayer, who was not involved in the research.
The findings of both studies were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Lower Alzheimer’s disease prevalence
The influenza vaccine study was presented by Albert Amran, a fourth-year medical student at McGovern Medical School at the University of Texas Health Science Center at Houston. The researchers used electronic health record data to create a propensity-matched cohort of 9,066 vaccinated and unvaccinated adults ages 60 and older.
Influenza vaccination, increased frequency of administration, and younger age at time of vaccination were all associated with reduced incidence of Alzheimer’s disease, Mr. Amran reported.
Being vaccinated for influenza was significantly linked to a lower prevalence of Alzheimer’s disease (odds ratio [OR], 0.83; P < .0001) in comparison with not being vaccinated. Receiving more than one vaccination over the years was associated with an additional reduction in AD incidence (OR, 0.87; P = .0342). The protection appeared to be strongest for those who received their first vaccination at a younger age, for example, at age 60 versus 70.
Mr. Amran and research colleagues have two theories as to why influenza vaccination may protect the brain.
One is that vaccination may aid the immune system as people age. “As people get older, their immune systems become less able to control infection. We’ve seen this with the ongoing pandemic, with older people at much higher risk for dying. Giving people the vaccine once a year may help keep the immune system in shape,” Mr. Amran said.
Another theory is that the prevention of influenza itself may be relevant. “Flu infections can be extremely deadly in older patients. Maybe the results of our study will give another reason for people to get vaccinated,” Mr. Amran said.
Pneumonia vaccine
The other study was presented by Svetlana Ukraintseva, PhD, of Duke University, Durham, N.C.
Dr. Ukraintseva and colleagues investigated associations between pneumococcal vaccine, with and without an accompanying influenza vaccine, and the risk for Alzheimer’s disease among 5,146 participants in the Cardiovascular Health Study. Covariates included sex, race, birth cohort, education, smoking, and a known genetic risk factor for Alzheimer’s disease: the rs2075650 G allele in the TOMM40 gene.
In a logistic model with all covariates, vaccination against pneumonia between ages 65 and 75 was significantly associated with reduced risk of developing AD (OR, 0.70; P < .04). The largest reduction in Alzheimer’s disease risk (OR, 0.62; P < .04) was among those vaccinated against pneumonia who were noncarriers of the rs2075650 G allele.
Total number of vaccinations against pneumonia and influenza between ages 65 and 75 was also associated with a lower risk for Alzheimer’s disease (OR, 0.88; P < .01). However, the effect was not evident for the influenza vaccination alone.
“The fact that very different pathogens – viral, bacterial, fungal – have been linked to Alzheimer’s disease indicates a possibility that compromised host immunity may play a role in Alzheimer’s disease through increasing overall brain’s vulnerability to various microbes,” said Dr. Ukraintseva.
The current findings support further investigation of pneumococcal vaccine as a “reasonable candidate for repurposing in personalized AD prevention,” she noted. “These results also support the important role of boosting overall immune robustness/resilience in preventing Alzheimer’s disease,” Dr. Ukraintseva added.
Her group is currently working on confirming the findings in another population.
Brain protective?
“Neither study can prove that the benefit is directly related to the vaccine itself, but what they can indicate is that potentially, vaccines are a way to protect your health and brain,” Dr. Edelmayer said.
In a statement, Maria Carrillo, PhD, chief science officer for the Alzheimer’s Association, noted that more research is needed.
The new data call “for further studies in large, diverse clinical trials to inform whether vaccinations as a public health strategy decrease our risk for developing dementia as we age,” Dr. Carillo said.
Funding for the influenza vaccine study was provided by the Christopher Sarofim Family Professorship in Biomedical Informatics and Bioengineering, a UT STARs Award, the Cancer Prevention and Research Institute of Texas, and the National Institutes of Health. Funding for the pneumonia study was provided by the National Institute on Aging. Dr. Amran, Dr. Ukraintseva, Dr. Edelmayer, and Dr. Carrillo have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In a cohort study of more than 9,000 older adults, receiving a single influenza vaccination was associated with a 17% lower prevalence of Alzheimer’s disease compared with not receiving the vaccine. In addition, for those who were vaccinated more than once over the years, there was an additional 13% reduction in Alzheimer’s disease incidence.
In another study, which included more than 5,000 older participants, being vaccinated against pneumonia between the ages of 65 and 75 reduced the risk of developing Alzheimer’s disease by 30%.
The subject of vaccines “is obviously very topical with the COVID-19 pandemic,” said Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association. “While these are very preliminary data, these studies do suggest that with vaccination against both respiratory illnesses, there is the potential to lower risk for developing cognitive decline and dementia,” said Dr. Edelmayer, who was not involved in the research.
The findings of both studies were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Lower Alzheimer’s disease prevalence
The influenza vaccine study was presented by Albert Amran, a fourth-year medical student at McGovern Medical School at the University of Texas Health Science Center at Houston. The researchers used electronic health record data to create a propensity-matched cohort of 9,066 vaccinated and unvaccinated adults ages 60 and older.
Influenza vaccination, increased frequency of administration, and younger age at time of vaccination were all associated with reduced incidence of Alzheimer’s disease, Mr. Amran reported.
Being vaccinated for influenza was significantly linked to a lower prevalence of Alzheimer’s disease (odds ratio [OR], 0.83; P < .0001) in comparison with not being vaccinated. Receiving more than one vaccination over the years was associated with an additional reduction in AD incidence (OR, 0.87; P = .0342). The protection appeared to be strongest for those who received their first vaccination at a younger age, for example, at age 60 versus 70.
Mr. Amran and research colleagues have two theories as to why influenza vaccination may protect the brain.
One is that vaccination may aid the immune system as people age. “As people get older, their immune systems become less able to control infection. We’ve seen this with the ongoing pandemic, with older people at much higher risk for dying. Giving people the vaccine once a year may help keep the immune system in shape,” Mr. Amran said.
Another theory is that the prevention of influenza itself may be relevant. “Flu infections can be extremely deadly in older patients. Maybe the results of our study will give another reason for people to get vaccinated,” Mr. Amran said.
Pneumonia vaccine
The other study was presented by Svetlana Ukraintseva, PhD, of Duke University, Durham, N.C.
Dr. Ukraintseva and colleagues investigated associations between pneumococcal vaccine, with and without an accompanying influenza vaccine, and the risk for Alzheimer’s disease among 5,146 participants in the Cardiovascular Health Study. Covariates included sex, race, birth cohort, education, smoking, and a known genetic risk factor for Alzheimer’s disease: the rs2075650 G allele in the TOMM40 gene.
In a logistic model with all covariates, vaccination against pneumonia between ages 65 and 75 was significantly associated with reduced risk of developing AD (OR, 0.70; P < .04). The largest reduction in Alzheimer’s disease risk (OR, 0.62; P < .04) was among those vaccinated against pneumonia who were noncarriers of the rs2075650 G allele.
Total number of vaccinations against pneumonia and influenza between ages 65 and 75 was also associated with a lower risk for Alzheimer’s disease (OR, 0.88; P < .01). However, the effect was not evident for the influenza vaccination alone.
“The fact that very different pathogens – viral, bacterial, fungal – have been linked to Alzheimer’s disease indicates a possibility that compromised host immunity may play a role in Alzheimer’s disease through increasing overall brain’s vulnerability to various microbes,” said Dr. Ukraintseva.
The current findings support further investigation of pneumococcal vaccine as a “reasonable candidate for repurposing in personalized AD prevention,” she noted. “These results also support the important role of boosting overall immune robustness/resilience in preventing Alzheimer’s disease,” Dr. Ukraintseva added.
Her group is currently working on confirming the findings in another population.
Brain protective?
“Neither study can prove that the benefit is directly related to the vaccine itself, but what they can indicate is that potentially, vaccines are a way to protect your health and brain,” Dr. Edelmayer said.
In a statement, Maria Carrillo, PhD, chief science officer for the Alzheimer’s Association, noted that more research is needed.
The new data call “for further studies in large, diverse clinical trials to inform whether vaccinations as a public health strategy decrease our risk for developing dementia as we age,” Dr. Carillo said.
Funding for the influenza vaccine study was provided by the Christopher Sarofim Family Professorship in Biomedical Informatics and Bioengineering, a UT STARs Award, the Cancer Prevention and Research Institute of Texas, and the National Institutes of Health. Funding for the pneumonia study was provided by the National Institute on Aging. Dr. Amran, Dr. Ukraintseva, Dr. Edelmayer, and Dr. Carrillo have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In a cohort study of more than 9,000 older adults, receiving a single influenza vaccination was associated with a 17% lower prevalence of Alzheimer’s disease compared with not receiving the vaccine. In addition, for those who were vaccinated more than once over the years, there was an additional 13% reduction in Alzheimer’s disease incidence.
In another study, which included more than 5,000 older participants, being vaccinated against pneumonia between the ages of 65 and 75 reduced the risk of developing Alzheimer’s disease by 30%.
The subject of vaccines “is obviously very topical with the COVID-19 pandemic,” said Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association. “While these are very preliminary data, these studies do suggest that with vaccination against both respiratory illnesses, there is the potential to lower risk for developing cognitive decline and dementia,” said Dr. Edelmayer, who was not involved in the research.
The findings of both studies were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Lower Alzheimer’s disease prevalence
The influenza vaccine study was presented by Albert Amran, a fourth-year medical student at McGovern Medical School at the University of Texas Health Science Center at Houston. The researchers used electronic health record data to create a propensity-matched cohort of 9,066 vaccinated and unvaccinated adults ages 60 and older.
Influenza vaccination, increased frequency of administration, and younger age at time of vaccination were all associated with reduced incidence of Alzheimer’s disease, Mr. Amran reported.
Being vaccinated for influenza was significantly linked to a lower prevalence of Alzheimer’s disease (odds ratio [OR], 0.83; P < .0001) in comparison with not being vaccinated. Receiving more than one vaccination over the years was associated with an additional reduction in AD incidence (OR, 0.87; P = .0342). The protection appeared to be strongest for those who received their first vaccination at a younger age, for example, at age 60 versus 70.
Mr. Amran and research colleagues have two theories as to why influenza vaccination may protect the brain.
One is that vaccination may aid the immune system as people age. “As people get older, their immune systems become less able to control infection. We’ve seen this with the ongoing pandemic, with older people at much higher risk for dying. Giving people the vaccine once a year may help keep the immune system in shape,” Mr. Amran said.
Another theory is that the prevention of influenza itself may be relevant. “Flu infections can be extremely deadly in older patients. Maybe the results of our study will give another reason for people to get vaccinated,” Mr. Amran said.
Pneumonia vaccine
The other study was presented by Svetlana Ukraintseva, PhD, of Duke University, Durham, N.C.
Dr. Ukraintseva and colleagues investigated associations between pneumococcal vaccine, with and without an accompanying influenza vaccine, and the risk for Alzheimer’s disease among 5,146 participants in the Cardiovascular Health Study. Covariates included sex, race, birth cohort, education, smoking, and a known genetic risk factor for Alzheimer’s disease: the rs2075650 G allele in the TOMM40 gene.
In a logistic model with all covariates, vaccination against pneumonia between ages 65 and 75 was significantly associated with reduced risk of developing AD (OR, 0.70; P < .04). The largest reduction in Alzheimer’s disease risk (OR, 0.62; P < .04) was among those vaccinated against pneumonia who were noncarriers of the rs2075650 G allele.
Total number of vaccinations against pneumonia and influenza between ages 65 and 75 was also associated with a lower risk for Alzheimer’s disease (OR, 0.88; P < .01). However, the effect was not evident for the influenza vaccination alone.
“The fact that very different pathogens – viral, bacterial, fungal – have been linked to Alzheimer’s disease indicates a possibility that compromised host immunity may play a role in Alzheimer’s disease through increasing overall brain’s vulnerability to various microbes,” said Dr. Ukraintseva.
The current findings support further investigation of pneumococcal vaccine as a “reasonable candidate for repurposing in personalized AD prevention,” she noted. “These results also support the important role of boosting overall immune robustness/resilience in preventing Alzheimer’s disease,” Dr. Ukraintseva added.
Her group is currently working on confirming the findings in another population.
Brain protective?
“Neither study can prove that the benefit is directly related to the vaccine itself, but what they can indicate is that potentially, vaccines are a way to protect your health and brain,” Dr. Edelmayer said.
In a statement, Maria Carrillo, PhD, chief science officer for the Alzheimer’s Association, noted that more research is needed.
The new data call “for further studies in large, diverse clinical trials to inform whether vaccinations as a public health strategy decrease our risk for developing dementia as we age,” Dr. Carillo said.
Funding for the influenza vaccine study was provided by the Christopher Sarofim Family Professorship in Biomedical Informatics and Bioengineering, a UT STARs Award, the Cancer Prevention and Research Institute of Texas, and the National Institutes of Health. Funding for the pneumonia study was provided by the National Institute on Aging. Dr. Amran, Dr. Ukraintseva, Dr. Edelmayer, and Dr. Carrillo have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AAIC 2020
US News releases latest top hospitals list, adds COVID heroes
This year’s rankings include special recognition of the “herculean efforts” by the nation’s healthcare professionals in fighting COVID-19, often at great personal risk.
“The US News Hospital Heroes series is a cornerstone of this year’s rankings package, profiling more than 65 health care heroes from across the country, along with commentary from top executives at hospitals who faced the pandemic head on,” a news release from the magazine explains.
“The pandemic has altered, perhaps permanently, how patients get care and from whom they get it. Amid the disruption, we are steadfastly committed to providing the public with authoritative data for comparing hospital quality,” Ben Harder, managing editor and chief of health analysis at US News, said in the release.
“No hospital’s clinical team came through this unprecedented health crisis unscathed. Our Hospital Heroes series is a tribute to recognizing individuals at urban and rural hospitals in communities across the country who have gone above and beyond during this unparalleled time in history,” said Harder.
Mayo Clinic Still Number One
Following Mayo Clinic, Cleveland Clinic in Ohio takes the number two spot this year (up from number four last year) in the magazine’s annual honor roll, which highlights hospitals that deliver “exceptional treatment across multiple areas of care.”
Johns Hopkins Hospital in Baltimore, Maryland, holds the number three spot, while New York-Presbyterian Hospital–Columbia and Cornell in New York City and UCLA Medical Center, Los Angeles, tie for the number four spot.
Massachusetts General Hospital in Boston, which held the number two spot last year, has fallen to number six. Rounding out the top 10, in order, are Cedars-Sinai Medical Center, Los Angeles; UCSF Medical Center, San Francisco; NYU Langone Hospitals, New York City; Northwestern Memorial Hospital, Chicago, Illinois.
2020–2021 Best Hospitals Honor Roll
1. Mayo Clinic, Rochester, Minnesota
2. Cleveland Clinic, Ohio
3. Johns Hopkins Hospital, Baltimore, Maryland
4. (tie) New York–Presbyterian Hospital–Columbia and Cornell, New York City
4. (tie) UCLA Medical Center, Los Angeles
6. Massachusetts General Hospital, Boston
7. Cedars-Sinai Medical Center, San Francisco
8. UCSF Medical Center, San Francisco
9. NYU Langone Hospitals, New York, New York City
10. Northwestern Memorial Hospital, Chicago
11. University of Michigan Hospitals–Michigan Medicine, Ann Arbor
12. Brigham and Women’s Hospital, Boston
13. Stanford Health Care–Stanford Hospital, Palo Alto, California
14. Mount Sinai Hospital, New York City
15. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
16. Mayo Clinic–Phoenix
17. Rush University Medical Center, Chicago
18. (tie) Barnes-Jewish Hospital, Saint Louis
18. (tie) Keck Hospital of USC, Los Angeles
20. Houston Methodist Hospital, Texas
In the 2020–2021 Best Hospitals: Specialty Rankings, University of Texas MD Anderson Cancer Center continues to hold the number one spot in cancer, the Hospital for Special Surgery is number one in orthopedics, and the Cleveland Clinic is number one in cardiology and heart surgery.
For this year’s rankings, US News developed a new cardiac rating that measures the quality of hospitals› transcatheter aortic valve replacement, which is rapidly being adopted as a minimally invasive alternative to aortic valve surgery.
Top Five for Cancer
1. University of Texas MD Anderson Cancer Center, Houston
2. Memorial Sloan Kettering Cancer Center, New York City
3. Mayo Clinic, Rochester, Minnesota
4. Johns Hopkins Hospital, Baltimore, Maryland
5. Cleveland Clinic, Ohio
Top Five for Cardiology and Heart Surgery
1. Cleveland Clinic, Ohio
2. Mayo Clinic, Rochester, Minnesota
3. Cedars-Sinai Medical Center, Los Angeles
4. New York–Presbyterian Hospital–Columbia and Cornell, NYC
5. Massachusetts General Hospital, Boston
Top Five for Orthopedics
1. Hospital for Special Surgery, New York City
2. Mayo Clinic, Rochester, Minnesota
3. Cedars-Sinai Medical Center, Los Angeles
4. NYU Langone Orthopedic Hospital, New York City
5. Rush University Medical Center, Chicago
For the 2020–2021 rankings and ratings, US News compared more than 4500 medical centers across the country in 16 specialties and 10 procedures and conditions. Of these, 563 were recognized as Best Regional Hospitals on the basis of their strong performance in multiple areas of care. The top 20 hospitals, which deliver exceptional treatment across many areas of care, were also named to the honor roll.
The magazine notes that data for the 2020–2021 Best Hospitals rankings and ratings come from a period predating the COVID-19 pandemic and were not affected by the pandemic’s impact on hospitals. The methodologies are based largely on objective measures, such as risk-adjusted survival and discharge-to-home rates, volume, and quality of nursing, among other care-related indicators.
The full report on hospital ranking is available online.
This article first appeared on Medscape.com.
This year’s rankings include special recognition of the “herculean efforts” by the nation’s healthcare professionals in fighting COVID-19, often at great personal risk.
“The US News Hospital Heroes series is a cornerstone of this year’s rankings package, profiling more than 65 health care heroes from across the country, along with commentary from top executives at hospitals who faced the pandemic head on,” a news release from the magazine explains.
“The pandemic has altered, perhaps permanently, how patients get care and from whom they get it. Amid the disruption, we are steadfastly committed to providing the public with authoritative data for comparing hospital quality,” Ben Harder, managing editor and chief of health analysis at US News, said in the release.
“No hospital’s clinical team came through this unprecedented health crisis unscathed. Our Hospital Heroes series is a tribute to recognizing individuals at urban and rural hospitals in communities across the country who have gone above and beyond during this unparalleled time in history,” said Harder.
Mayo Clinic Still Number One
Following Mayo Clinic, Cleveland Clinic in Ohio takes the number two spot this year (up from number four last year) in the magazine’s annual honor roll, which highlights hospitals that deliver “exceptional treatment across multiple areas of care.”
Johns Hopkins Hospital in Baltimore, Maryland, holds the number three spot, while New York-Presbyterian Hospital–Columbia and Cornell in New York City and UCLA Medical Center, Los Angeles, tie for the number four spot.
Massachusetts General Hospital in Boston, which held the number two spot last year, has fallen to number six. Rounding out the top 10, in order, are Cedars-Sinai Medical Center, Los Angeles; UCSF Medical Center, San Francisco; NYU Langone Hospitals, New York City; Northwestern Memorial Hospital, Chicago, Illinois.
2020–2021 Best Hospitals Honor Roll
1. Mayo Clinic, Rochester, Minnesota
2. Cleveland Clinic, Ohio
3. Johns Hopkins Hospital, Baltimore, Maryland
4. (tie) New York–Presbyterian Hospital–Columbia and Cornell, New York City
4. (tie) UCLA Medical Center, Los Angeles
6. Massachusetts General Hospital, Boston
7. Cedars-Sinai Medical Center, San Francisco
8. UCSF Medical Center, San Francisco
9. NYU Langone Hospitals, New York, New York City
10. Northwestern Memorial Hospital, Chicago
11. University of Michigan Hospitals–Michigan Medicine, Ann Arbor
12. Brigham and Women’s Hospital, Boston
13. Stanford Health Care–Stanford Hospital, Palo Alto, California
14. Mount Sinai Hospital, New York City
15. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
16. Mayo Clinic–Phoenix
17. Rush University Medical Center, Chicago
18. (tie) Barnes-Jewish Hospital, Saint Louis
18. (tie) Keck Hospital of USC, Los Angeles
20. Houston Methodist Hospital, Texas
In the 2020–2021 Best Hospitals: Specialty Rankings, University of Texas MD Anderson Cancer Center continues to hold the number one spot in cancer, the Hospital for Special Surgery is number one in orthopedics, and the Cleveland Clinic is number one in cardiology and heart surgery.
For this year’s rankings, US News developed a new cardiac rating that measures the quality of hospitals› transcatheter aortic valve replacement, which is rapidly being adopted as a minimally invasive alternative to aortic valve surgery.
Top Five for Cancer
1. University of Texas MD Anderson Cancer Center, Houston
2. Memorial Sloan Kettering Cancer Center, New York City
3. Mayo Clinic, Rochester, Minnesota
4. Johns Hopkins Hospital, Baltimore, Maryland
5. Cleveland Clinic, Ohio
Top Five for Cardiology and Heart Surgery
1. Cleveland Clinic, Ohio
2. Mayo Clinic, Rochester, Minnesota
3. Cedars-Sinai Medical Center, Los Angeles
4. New York–Presbyterian Hospital–Columbia and Cornell, NYC
5. Massachusetts General Hospital, Boston
Top Five for Orthopedics
1. Hospital for Special Surgery, New York City
2. Mayo Clinic, Rochester, Minnesota
3. Cedars-Sinai Medical Center, Los Angeles
4. NYU Langone Orthopedic Hospital, New York City
5. Rush University Medical Center, Chicago
For the 2020–2021 rankings and ratings, US News compared more than 4500 medical centers across the country in 16 specialties and 10 procedures and conditions. Of these, 563 were recognized as Best Regional Hospitals on the basis of their strong performance in multiple areas of care. The top 20 hospitals, which deliver exceptional treatment across many areas of care, were also named to the honor roll.
The magazine notes that data for the 2020–2021 Best Hospitals rankings and ratings come from a period predating the COVID-19 pandemic and were not affected by the pandemic’s impact on hospitals. The methodologies are based largely on objective measures, such as risk-adjusted survival and discharge-to-home rates, volume, and quality of nursing, among other care-related indicators.
The full report on hospital ranking is available online.
This article first appeared on Medscape.com.
This year’s rankings include special recognition of the “herculean efforts” by the nation’s healthcare professionals in fighting COVID-19, often at great personal risk.
“The US News Hospital Heroes series is a cornerstone of this year’s rankings package, profiling more than 65 health care heroes from across the country, along with commentary from top executives at hospitals who faced the pandemic head on,” a news release from the magazine explains.
“The pandemic has altered, perhaps permanently, how patients get care and from whom they get it. Amid the disruption, we are steadfastly committed to providing the public with authoritative data for comparing hospital quality,” Ben Harder, managing editor and chief of health analysis at US News, said in the release.
“No hospital’s clinical team came through this unprecedented health crisis unscathed. Our Hospital Heroes series is a tribute to recognizing individuals at urban and rural hospitals in communities across the country who have gone above and beyond during this unparalleled time in history,” said Harder.
Mayo Clinic Still Number One
Following Mayo Clinic, Cleveland Clinic in Ohio takes the number two spot this year (up from number four last year) in the magazine’s annual honor roll, which highlights hospitals that deliver “exceptional treatment across multiple areas of care.”
Johns Hopkins Hospital in Baltimore, Maryland, holds the number three spot, while New York-Presbyterian Hospital–Columbia and Cornell in New York City and UCLA Medical Center, Los Angeles, tie for the number four spot.
Massachusetts General Hospital in Boston, which held the number two spot last year, has fallen to number six. Rounding out the top 10, in order, are Cedars-Sinai Medical Center, Los Angeles; UCSF Medical Center, San Francisco; NYU Langone Hospitals, New York City; Northwestern Memorial Hospital, Chicago, Illinois.
2020–2021 Best Hospitals Honor Roll
1. Mayo Clinic, Rochester, Minnesota
2. Cleveland Clinic, Ohio
3. Johns Hopkins Hospital, Baltimore, Maryland
4. (tie) New York–Presbyterian Hospital–Columbia and Cornell, New York City
4. (tie) UCLA Medical Center, Los Angeles
6. Massachusetts General Hospital, Boston
7. Cedars-Sinai Medical Center, San Francisco
8. UCSF Medical Center, San Francisco
9. NYU Langone Hospitals, New York, New York City
10. Northwestern Memorial Hospital, Chicago
11. University of Michigan Hospitals–Michigan Medicine, Ann Arbor
12. Brigham and Women’s Hospital, Boston
13. Stanford Health Care–Stanford Hospital, Palo Alto, California
14. Mount Sinai Hospital, New York City
15. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
16. Mayo Clinic–Phoenix
17. Rush University Medical Center, Chicago
18. (tie) Barnes-Jewish Hospital, Saint Louis
18. (tie) Keck Hospital of USC, Los Angeles
20. Houston Methodist Hospital, Texas
In the 2020–2021 Best Hospitals: Specialty Rankings, University of Texas MD Anderson Cancer Center continues to hold the number one spot in cancer, the Hospital for Special Surgery is number one in orthopedics, and the Cleveland Clinic is number one in cardiology and heart surgery.
For this year’s rankings, US News developed a new cardiac rating that measures the quality of hospitals› transcatheter aortic valve replacement, which is rapidly being adopted as a minimally invasive alternative to aortic valve surgery.
Top Five for Cancer
1. University of Texas MD Anderson Cancer Center, Houston
2. Memorial Sloan Kettering Cancer Center, New York City
3. Mayo Clinic, Rochester, Minnesota
4. Johns Hopkins Hospital, Baltimore, Maryland
5. Cleveland Clinic, Ohio
Top Five for Cardiology and Heart Surgery
1. Cleveland Clinic, Ohio
2. Mayo Clinic, Rochester, Minnesota
3. Cedars-Sinai Medical Center, Los Angeles
4. New York–Presbyterian Hospital–Columbia and Cornell, NYC
5. Massachusetts General Hospital, Boston
Top Five for Orthopedics
1. Hospital for Special Surgery, New York City
2. Mayo Clinic, Rochester, Minnesota
3. Cedars-Sinai Medical Center, Los Angeles
4. NYU Langone Orthopedic Hospital, New York City
5. Rush University Medical Center, Chicago
For the 2020–2021 rankings and ratings, US News compared more than 4500 medical centers across the country in 16 specialties and 10 procedures and conditions. Of these, 563 were recognized as Best Regional Hospitals on the basis of their strong performance in multiple areas of care. The top 20 hospitals, which deliver exceptional treatment across many areas of care, were also named to the honor roll.
The magazine notes that data for the 2020–2021 Best Hospitals rankings and ratings come from a period predating the COVID-19 pandemic and were not affected by the pandemic’s impact on hospitals. The methodologies are based largely on objective measures, such as risk-adjusted survival and discharge-to-home rates, volume, and quality of nursing, among other care-related indicators.
The full report on hospital ranking is available online.
This article first appeared on Medscape.com.
Levothyroxine: No LV benefit in subclinical hypothyroidism with MI
For patients with acute myocardial infarction (MI) and mild subclinical hypothyroidism (SCH), treatment with levothyroxine does not improve left ventricular function, according to results of the Thyroid in Acute Myocardial Infarction (ThyrAMI-2) trial.
“SCH is common, affecting approximately 10% of the adult population, and has been associated with worse outcomes in patients with cardiovascular disease in observational studies,” Salman Razvi, MD, Translational and Clinical Research Institute, Newcastle University, Newcastle upon Tyne, England, said in an interview.
This study shows that levothyroxine treatment for patients with SCH and acute MI is “unlikely to be of benefit,” he said.
“This study says that treating the thyroid failure does not help nor harm such patients,” Terry F. Davies, MD, director, division of endocrinology, diabetes, and bone diseases, Mount Sinai Beth Israel Medical Center, New York, said in an interview. He was not involved in the study, which was published online July 21 in JAMA.
Participants included 95 adults (mean age, 63.5 years; 72 men) with persistent mild SCH who presented with acute MI at six hospitals in the United Kingdom. Most (69%) had ST-segment elevation MI.
Inclusion criteria were age older than 18 years and serum thyrotropin level >4.0 mU/L with a normal free thyroxine level on two occasions 7-10 days apart and with one thyrotropin value <10 mU/L.
Forty-six participants were randomly allocated to receive levothyroxine starting at 25 mcg titrated to aim for serum thyrotropin levels between 0.4 and 2.5 mU/L and 49 to matching placebo capsules taken once daily for 52 weeks.
The primary outcome was left ventricular ejection fraction (LVEF) at 52 weeks, assessed via MRI, with adjustment for age, sex, acute MI type, affected coronary artery territory, and baseline LVEF.
Secondary outcomes were LV volume, infarct size, adverse events, and patient-reported outcome measures of health status, health-related quality of life, and depression.
The median daily dose of levothyroxine at the end of the study was 50 mcg. Adherence to study medication was 94% during the course of the study.
At week 52, mean LVEF improved from 51.3% at baseline to 53.8% in the levothyroxine group and from 54.0% to 56.1% in the placebo group.
The difference was not significant between groups, with an adjusted between-group difference of 0.76% (95% confidence interval, –0.93% to 2.46%; P = .37).
There were also no significant differences in any of the secondary outcomes. There were 15 (33.3%) cardiovascular adverse events in the levothyroxine group and 18 (36.7%) in the placebo group.
Recent clinical practice guidelines have highlighted a lack of high-quality data to make recommendations regarding the management of mild SCH, particularly for patients with cardiovascular disease, Dr. Razvi and colleagues noted in their article.
“On the basis of these findings, screening for and subsequent treatment of subclinical hypothyroidism in patients with acute myocardial infarction to preserve LV function is not justified,” they concluded.
Important caveats
The investigators noted several important caveats and limitations. The trial recruited patients with mild SCH because this group constitutes the majority of patients with SCH and for whom there is the “greatest uncertainty” regarding treatment efficacy. It’s not known whether targeting treatment for individuals with more severe disease may be beneficial.
The therapeutic benefit of levothyroxine may have been blunted, owing to the delay between coronary occlusion and the start of levothyroxine (median delay, 17 days). It’s unclear whether earlier treatment or treatment for a longer period may be beneficial.
But Dr. Davies noted that “treatment is usually avoided in the emergency situation,” and therefore he doesn’t think the treatment delay is a limitation; rather, “it would appear prudent,” he said in the interview.
“The real issues with an otherwise very careful study is the small size of the population despite the statistical assessment that this was all that was needed and, secondly, the small dose of thyroxine used,” Dr. Davies said.
The authors agree that the low dose of levothyroxine is a limitation. The median dose at the end of the study – 50 mcg daily – is “lower than that used in other trials that have demonstrated a benefit of treatment on endothelial function and lipid profiles,” they pointed out.
Dr. Davies noted that thyroid tests are “usually routine” for patients with MI. “Mild subclinical thyroid failure has been associated with worse cardiac outcomes, [but] treating such patients with thyroid hormone is very controversial since thyroid hormone can induce arrhythmias,” he said.
The study was supported in part by the National Institute for Health Research (NIHR) at the University of Leeds. Dr. Razvi received grants from the NIHR and nonfinancial support from Amdipharm Pharmaceuticals UK during the conduct of the study and personal fees from Merck and Abbott Pharmaceuticals outside the submitted work. Dr. Davies has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
For patients with acute myocardial infarction (MI) and mild subclinical hypothyroidism (SCH), treatment with levothyroxine does not improve left ventricular function, according to results of the Thyroid in Acute Myocardial Infarction (ThyrAMI-2) trial.
“SCH is common, affecting approximately 10% of the adult population, and has been associated with worse outcomes in patients with cardiovascular disease in observational studies,” Salman Razvi, MD, Translational and Clinical Research Institute, Newcastle University, Newcastle upon Tyne, England, said in an interview.
This study shows that levothyroxine treatment for patients with SCH and acute MI is “unlikely to be of benefit,” he said.
“This study says that treating the thyroid failure does not help nor harm such patients,” Terry F. Davies, MD, director, division of endocrinology, diabetes, and bone diseases, Mount Sinai Beth Israel Medical Center, New York, said in an interview. He was not involved in the study, which was published online July 21 in JAMA.
Participants included 95 adults (mean age, 63.5 years; 72 men) with persistent mild SCH who presented with acute MI at six hospitals in the United Kingdom. Most (69%) had ST-segment elevation MI.
Inclusion criteria were age older than 18 years and serum thyrotropin level >4.0 mU/L with a normal free thyroxine level on two occasions 7-10 days apart and with one thyrotropin value <10 mU/L.
Forty-six participants were randomly allocated to receive levothyroxine starting at 25 mcg titrated to aim for serum thyrotropin levels between 0.4 and 2.5 mU/L and 49 to matching placebo capsules taken once daily for 52 weeks.
The primary outcome was left ventricular ejection fraction (LVEF) at 52 weeks, assessed via MRI, with adjustment for age, sex, acute MI type, affected coronary artery territory, and baseline LVEF.
Secondary outcomes were LV volume, infarct size, adverse events, and patient-reported outcome measures of health status, health-related quality of life, and depression.
The median daily dose of levothyroxine at the end of the study was 50 mcg. Adherence to study medication was 94% during the course of the study.
At week 52, mean LVEF improved from 51.3% at baseline to 53.8% in the levothyroxine group and from 54.0% to 56.1% in the placebo group.
The difference was not significant between groups, with an adjusted between-group difference of 0.76% (95% confidence interval, –0.93% to 2.46%; P = .37).
There were also no significant differences in any of the secondary outcomes. There were 15 (33.3%) cardiovascular adverse events in the levothyroxine group and 18 (36.7%) in the placebo group.
Recent clinical practice guidelines have highlighted a lack of high-quality data to make recommendations regarding the management of mild SCH, particularly for patients with cardiovascular disease, Dr. Razvi and colleagues noted in their article.
“On the basis of these findings, screening for and subsequent treatment of subclinical hypothyroidism in patients with acute myocardial infarction to preserve LV function is not justified,” they concluded.
Important caveats
The investigators noted several important caveats and limitations. The trial recruited patients with mild SCH because this group constitutes the majority of patients with SCH and for whom there is the “greatest uncertainty” regarding treatment efficacy. It’s not known whether targeting treatment for individuals with more severe disease may be beneficial.
The therapeutic benefit of levothyroxine may have been blunted, owing to the delay between coronary occlusion and the start of levothyroxine (median delay, 17 days). It’s unclear whether earlier treatment or treatment for a longer period may be beneficial.
But Dr. Davies noted that “treatment is usually avoided in the emergency situation,” and therefore he doesn’t think the treatment delay is a limitation; rather, “it would appear prudent,” he said in the interview.
“The real issues with an otherwise very careful study is the small size of the population despite the statistical assessment that this was all that was needed and, secondly, the small dose of thyroxine used,” Dr. Davies said.
The authors agree that the low dose of levothyroxine is a limitation. The median dose at the end of the study – 50 mcg daily – is “lower than that used in other trials that have demonstrated a benefit of treatment on endothelial function and lipid profiles,” they pointed out.
Dr. Davies noted that thyroid tests are “usually routine” for patients with MI. “Mild subclinical thyroid failure has been associated with worse cardiac outcomes, [but] treating such patients with thyroid hormone is very controversial since thyroid hormone can induce arrhythmias,” he said.
The study was supported in part by the National Institute for Health Research (NIHR) at the University of Leeds. Dr. Razvi received grants from the NIHR and nonfinancial support from Amdipharm Pharmaceuticals UK during the conduct of the study and personal fees from Merck and Abbott Pharmaceuticals outside the submitted work. Dr. Davies has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
For patients with acute myocardial infarction (MI) and mild subclinical hypothyroidism (SCH), treatment with levothyroxine does not improve left ventricular function, according to results of the Thyroid in Acute Myocardial Infarction (ThyrAMI-2) trial.
“SCH is common, affecting approximately 10% of the adult population, and has been associated with worse outcomes in patients with cardiovascular disease in observational studies,” Salman Razvi, MD, Translational and Clinical Research Institute, Newcastle University, Newcastle upon Tyne, England, said in an interview.
This study shows that levothyroxine treatment for patients with SCH and acute MI is “unlikely to be of benefit,” he said.
“This study says that treating the thyroid failure does not help nor harm such patients,” Terry F. Davies, MD, director, division of endocrinology, diabetes, and bone diseases, Mount Sinai Beth Israel Medical Center, New York, said in an interview. He was not involved in the study, which was published online July 21 in JAMA.
Participants included 95 adults (mean age, 63.5 years; 72 men) with persistent mild SCH who presented with acute MI at six hospitals in the United Kingdom. Most (69%) had ST-segment elevation MI.
Inclusion criteria were age older than 18 years and serum thyrotropin level >4.0 mU/L with a normal free thyroxine level on two occasions 7-10 days apart and with one thyrotropin value <10 mU/L.
Forty-six participants were randomly allocated to receive levothyroxine starting at 25 mcg titrated to aim for serum thyrotropin levels between 0.4 and 2.5 mU/L and 49 to matching placebo capsules taken once daily for 52 weeks.
The primary outcome was left ventricular ejection fraction (LVEF) at 52 weeks, assessed via MRI, with adjustment for age, sex, acute MI type, affected coronary artery territory, and baseline LVEF.
Secondary outcomes were LV volume, infarct size, adverse events, and patient-reported outcome measures of health status, health-related quality of life, and depression.
The median daily dose of levothyroxine at the end of the study was 50 mcg. Adherence to study medication was 94% during the course of the study.
At week 52, mean LVEF improved from 51.3% at baseline to 53.8% in the levothyroxine group and from 54.0% to 56.1% in the placebo group.
The difference was not significant between groups, with an adjusted between-group difference of 0.76% (95% confidence interval, –0.93% to 2.46%; P = .37).
There were also no significant differences in any of the secondary outcomes. There were 15 (33.3%) cardiovascular adverse events in the levothyroxine group and 18 (36.7%) in the placebo group.
Recent clinical practice guidelines have highlighted a lack of high-quality data to make recommendations regarding the management of mild SCH, particularly for patients with cardiovascular disease, Dr. Razvi and colleagues noted in their article.
“On the basis of these findings, screening for and subsequent treatment of subclinical hypothyroidism in patients with acute myocardial infarction to preserve LV function is not justified,” they concluded.
Important caveats
The investigators noted several important caveats and limitations. The trial recruited patients with mild SCH because this group constitutes the majority of patients with SCH and for whom there is the “greatest uncertainty” regarding treatment efficacy. It’s not known whether targeting treatment for individuals with more severe disease may be beneficial.
The therapeutic benefit of levothyroxine may have been blunted, owing to the delay between coronary occlusion and the start of levothyroxine (median delay, 17 days). It’s unclear whether earlier treatment or treatment for a longer period may be beneficial.
But Dr. Davies noted that “treatment is usually avoided in the emergency situation,” and therefore he doesn’t think the treatment delay is a limitation; rather, “it would appear prudent,” he said in the interview.
“The real issues with an otherwise very careful study is the small size of the population despite the statistical assessment that this was all that was needed and, secondly, the small dose of thyroxine used,” Dr. Davies said.
The authors agree that the low dose of levothyroxine is a limitation. The median dose at the end of the study – 50 mcg daily – is “lower than that used in other trials that have demonstrated a benefit of treatment on endothelial function and lipid profiles,” they pointed out.
Dr. Davies noted that thyroid tests are “usually routine” for patients with MI. “Mild subclinical thyroid failure has been associated with worse cardiac outcomes, [but] treating such patients with thyroid hormone is very controversial since thyroid hormone can induce arrhythmias,” he said.
The study was supported in part by the National Institute for Health Research (NIHR) at the University of Leeds. Dr. Razvi received grants from the NIHR and nonfinancial support from Amdipharm Pharmaceuticals UK during the conduct of the study and personal fees from Merck and Abbott Pharmaceuticals outside the submitted work. Dr. Davies has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
More than half of psychiatrists have net worth under $1 million
Most psychiatrists live at or below their means, and more than half have a net worth of less than $1 million, according to the Medscape Psychiatrist Debt and Net Worth Report 2020.
An important caveat, however, is that the data for this year’s report were collected as part of the Medscape Physician Compensation Report 2020, prior to Feb. 11, 2020 – and before COVID-19 was declared a pandemic.
Although it will be some time before medical practices become accustomed to a new version of normal,
Among the lowest earners
According to the Medscape Psychiatrist Compensation Report 2020, psychiatrists are among the lowest earners of all physicians, earning $268,000 on average this year. However, that’s up somewhat from last year’s earnings of $260,000.
Among psychiatrists, 57% report a net worth (total assets minus total liabilities) of less than $1 million; 38% have a net worth between $1 million and $5 million, and 5% top $5 million in net worth.
Among specialists, orthopedists are most likely (at 19%) to top the $5 million level, followed by plastic surgeons and gastroenterologists (both at 16%), according to the overall Medscape Physician Debt and Net Worth Report 2020. The report is based on more than 17,000 physicians and spans 30 specialties.
By gender, more female than male psychiatrists have a net worth of less than $1 million (61% vs. 53%). The percentages are closer between female and male psychiatrists who have a net worth between $1 million and $5 million, at 36% and 41%, respectively. In addition, more male than female psychiatrists have a net worth above $5 million (6% vs. 3%).
As expected, the older the physician, the more money they have as earnings increase and early-career debt decreases. This is the case for psychiatrists, whose net worth over $1 million increased with age.
However, psychiatrists are among the most likely of all physicians to have a net worth of less than $500,000 (at 37%), which is on par with public health and preventive medicine physicians (also at 37%) and just below family medicine physicians (at 46%), pediatricians (44%), neurologists (41%), and internal medicine physicians (40%).
For nearly two-thirds of psychiatrists (63%), mortgage payments on their primary residence are the major expense; 39% of psychiatrists have a mortgage of $300,000 or less, and 12% have a mortgage topping $500,000; 31% have no mortgage at all. Six in 10 psychiatrists live in a house that is 3,000 sq ft or smaller.
Mortgage aside, other top ongoing expenses for psychiatrists are car payments (38%), school loan payments (28%), credit card debt (26%), and medical expenses for self or loved one (20%). At 28%, psychiatrists are toward the top of the list of all physicians when it comes to those still paying off loans for education.
Spending vs. saving
The average American has four credit cards, according to the credit reporting agency Experian. More than half of psychiatrists said they have four or fewer credit cards (35% have three or four credit cards, 20% have one or two cards, and 1% have none). A quarter of psychiatrists (25%) have five or six credit cards, and 19% have seven or more at their disposal.
Only a small percentage of psychiatrists (7%) say they live above their means; 53% live at their means, and 39% live below their means.
Most psychiatrists (62%) contribute $1,000 or more to a tax-deferred retirement or college savings account each month; 13% do not do this on a regular basis. Nearly two-thirds (65%) of psychiatrists contribute to a taxable savings account, a tool many use when tax-deferred contributions have reached their limit.
Nearly half of psychiatrists (47%) rely on a mental budget for personal expenses; 16% rely on a written budget or use software or an app for budgeting; and 37% don’t have a budget for personal expenses.
Three-quarters of psychiatrists did not experience a financial loss in 2019. Of those who did have a loss, the main causes were bad investments (6%) and practice-related problems (6%). Other factors were job loss (self or spouse/partner), real estate loss (4%), legal fees (3%), and divorce (3%).
Among psychiatrists who have joint finances with a spouse or partner, 51% pool their income to pay household expenses. For 14%, the person who earns more pays more of the bills and/or expenses. Only a small percentage divide bills and expenses equally, regardless of how much each makes (5%).
About 43% of psychiatrists currently work with a financial planner or have done so in the past, 40% never did, and 17% met with a financial planner but did not pursue working with that person.
A version of this article originally appeared on Medscape.com.
Most psychiatrists live at or below their means, and more than half have a net worth of less than $1 million, according to the Medscape Psychiatrist Debt and Net Worth Report 2020.
An important caveat, however, is that the data for this year’s report were collected as part of the Medscape Physician Compensation Report 2020, prior to Feb. 11, 2020 – and before COVID-19 was declared a pandemic.
Although it will be some time before medical practices become accustomed to a new version of normal,
Among the lowest earners
According to the Medscape Psychiatrist Compensation Report 2020, psychiatrists are among the lowest earners of all physicians, earning $268,000 on average this year. However, that’s up somewhat from last year’s earnings of $260,000.
Among psychiatrists, 57% report a net worth (total assets minus total liabilities) of less than $1 million; 38% have a net worth between $1 million and $5 million, and 5% top $5 million in net worth.
Among specialists, orthopedists are most likely (at 19%) to top the $5 million level, followed by plastic surgeons and gastroenterologists (both at 16%), according to the overall Medscape Physician Debt and Net Worth Report 2020. The report is based on more than 17,000 physicians and spans 30 specialties.
By gender, more female than male psychiatrists have a net worth of less than $1 million (61% vs. 53%). The percentages are closer between female and male psychiatrists who have a net worth between $1 million and $5 million, at 36% and 41%, respectively. In addition, more male than female psychiatrists have a net worth above $5 million (6% vs. 3%).
As expected, the older the physician, the more money they have as earnings increase and early-career debt decreases. This is the case for psychiatrists, whose net worth over $1 million increased with age.
However, psychiatrists are among the most likely of all physicians to have a net worth of less than $500,000 (at 37%), which is on par with public health and preventive medicine physicians (also at 37%) and just below family medicine physicians (at 46%), pediatricians (44%), neurologists (41%), and internal medicine physicians (40%).
For nearly two-thirds of psychiatrists (63%), mortgage payments on their primary residence are the major expense; 39% of psychiatrists have a mortgage of $300,000 or less, and 12% have a mortgage topping $500,000; 31% have no mortgage at all. Six in 10 psychiatrists live in a house that is 3,000 sq ft or smaller.
Mortgage aside, other top ongoing expenses for psychiatrists are car payments (38%), school loan payments (28%), credit card debt (26%), and medical expenses for self or loved one (20%). At 28%, psychiatrists are toward the top of the list of all physicians when it comes to those still paying off loans for education.
Spending vs. saving
The average American has four credit cards, according to the credit reporting agency Experian. More than half of psychiatrists said they have four or fewer credit cards (35% have three or four credit cards, 20% have one or two cards, and 1% have none). A quarter of psychiatrists (25%) have five or six credit cards, and 19% have seven or more at their disposal.
Only a small percentage of psychiatrists (7%) say they live above their means; 53% live at their means, and 39% live below their means.
Most psychiatrists (62%) contribute $1,000 or more to a tax-deferred retirement or college savings account each month; 13% do not do this on a regular basis. Nearly two-thirds (65%) of psychiatrists contribute to a taxable savings account, a tool many use when tax-deferred contributions have reached their limit.
Nearly half of psychiatrists (47%) rely on a mental budget for personal expenses; 16% rely on a written budget or use software or an app for budgeting; and 37% don’t have a budget for personal expenses.
Three-quarters of psychiatrists did not experience a financial loss in 2019. Of those who did have a loss, the main causes were bad investments (6%) and practice-related problems (6%). Other factors were job loss (self or spouse/partner), real estate loss (4%), legal fees (3%), and divorce (3%).
Among psychiatrists who have joint finances with a spouse or partner, 51% pool their income to pay household expenses. For 14%, the person who earns more pays more of the bills and/or expenses. Only a small percentage divide bills and expenses equally, regardless of how much each makes (5%).
About 43% of psychiatrists currently work with a financial planner or have done so in the past, 40% never did, and 17% met with a financial planner but did not pursue working with that person.
A version of this article originally appeared on Medscape.com.
Most psychiatrists live at or below their means, and more than half have a net worth of less than $1 million, according to the Medscape Psychiatrist Debt and Net Worth Report 2020.
An important caveat, however, is that the data for this year’s report were collected as part of the Medscape Physician Compensation Report 2020, prior to Feb. 11, 2020 – and before COVID-19 was declared a pandemic.
Although it will be some time before medical practices become accustomed to a new version of normal,
Among the lowest earners
According to the Medscape Psychiatrist Compensation Report 2020, psychiatrists are among the lowest earners of all physicians, earning $268,000 on average this year. However, that’s up somewhat from last year’s earnings of $260,000.
Among psychiatrists, 57% report a net worth (total assets minus total liabilities) of less than $1 million; 38% have a net worth between $1 million and $5 million, and 5% top $5 million in net worth.
Among specialists, orthopedists are most likely (at 19%) to top the $5 million level, followed by plastic surgeons and gastroenterologists (both at 16%), according to the overall Medscape Physician Debt and Net Worth Report 2020. The report is based on more than 17,000 physicians and spans 30 specialties.
By gender, more female than male psychiatrists have a net worth of less than $1 million (61% vs. 53%). The percentages are closer between female and male psychiatrists who have a net worth between $1 million and $5 million, at 36% and 41%, respectively. In addition, more male than female psychiatrists have a net worth above $5 million (6% vs. 3%).
As expected, the older the physician, the more money they have as earnings increase and early-career debt decreases. This is the case for psychiatrists, whose net worth over $1 million increased with age.
However, psychiatrists are among the most likely of all physicians to have a net worth of less than $500,000 (at 37%), which is on par with public health and preventive medicine physicians (also at 37%) and just below family medicine physicians (at 46%), pediatricians (44%), neurologists (41%), and internal medicine physicians (40%).
For nearly two-thirds of psychiatrists (63%), mortgage payments on their primary residence are the major expense; 39% of psychiatrists have a mortgage of $300,000 or less, and 12% have a mortgage topping $500,000; 31% have no mortgage at all. Six in 10 psychiatrists live in a house that is 3,000 sq ft or smaller.
Mortgage aside, other top ongoing expenses for psychiatrists are car payments (38%), school loan payments (28%), credit card debt (26%), and medical expenses for self or loved one (20%). At 28%, psychiatrists are toward the top of the list of all physicians when it comes to those still paying off loans for education.
Spending vs. saving
The average American has four credit cards, according to the credit reporting agency Experian. More than half of psychiatrists said they have four or fewer credit cards (35% have three or four credit cards, 20% have one or two cards, and 1% have none). A quarter of psychiatrists (25%) have five or six credit cards, and 19% have seven or more at their disposal.
Only a small percentage of psychiatrists (7%) say they live above their means; 53% live at their means, and 39% live below their means.
Most psychiatrists (62%) contribute $1,000 or more to a tax-deferred retirement or college savings account each month; 13% do not do this on a regular basis. Nearly two-thirds (65%) of psychiatrists contribute to a taxable savings account, a tool many use when tax-deferred contributions have reached their limit.
Nearly half of psychiatrists (47%) rely on a mental budget for personal expenses; 16% rely on a written budget or use software or an app for budgeting; and 37% don’t have a budget for personal expenses.
Three-quarters of psychiatrists did not experience a financial loss in 2019. Of those who did have a loss, the main causes were bad investments (6%) and practice-related problems (6%). Other factors were job loss (self or spouse/partner), real estate loss (4%), legal fees (3%), and divorce (3%).
Among psychiatrists who have joint finances with a spouse or partner, 51% pool their income to pay household expenses. For 14%, the person who earns more pays more of the bills and/or expenses. Only a small percentage divide bills and expenses equally, regardless of how much each makes (5%).
About 43% of psychiatrists currently work with a financial planner or have done so in the past, 40% never did, and 17% met with a financial planner but did not pursue working with that person.
A version of this article originally appeared on Medscape.com.
Most neurologists live within their means, are savers
An important caveat, however, is that data for this year’s report were collected prior to Feb. 11, 2020, as part of the Medscape Physician Compensation Report 2020. The financial picture has changed for many physicians since then because of COVID-19’s impact on medical practices.
While it will be some time before medical practices become accustomed to a new version of normal, the report data provide a picture of the debt load and net worth of neurologists.
Below the middle earners
According to the Medscape Neurologist Compensation Report 2020, neurologists are below the middle earners of all physicians, earning $280,000 on average this year. That’s up from $267,000 last year. More than half of neurologists (58%) report a net worth (total assets minus total liabilities) of less than $1 million (52% men, 70% women), while 36% have a net worth between $1 and $5 million (40% men, 29% women), and 6% top $5 million in net worth (8% men, 1% women).
Among specialists, orthopedists are most likely to top the $5 million level (at 19%), followed by plastic surgeons and gastroenterologists (both at 16%), according to the Medscape Physician Debt and Net Worth Report 2020.
Conversely, about two in five neurologists (41%) have a net worth under $500,000, just below family medicine physicians (46%) and pediatricians (44%).
For roughly two thirds of neurologists (64%), their major expense is mortgage payment on their primary residence. Around one third of neurologists have a mortgage of $300,000 or less, 28% have no mortgage or a mortgage that is paid off, and 17% have a mortgage topping $500,000. Six in 10 neurologists live in a house that is 3000 square feet or smaller.
Mortgage aside, other top ongoing expenses for neurologists are car payments (35%), credit card debt (28%), school loans (25%), and childcare (19%). At 25%, neurologists are in the middle of the list when it comes to physicians still paying off loans for education.
Spender or saver?
The average American has four credit cards, according to the credit reporting agency Experian. Among neurologists, more than half said they have four or fewer credit cards, including 37% with three to four cards, 16% with one to two cards, and 1% with no cards. A little more than a quarter of neurologists (28%) have five to six credit cards and 18% have more than seven at their disposal.
Only a small percentage of neurologists (7%) say they live above their means; 57% live at their means and 35% live below their means.
More than half (59%) of neurologists contribute $1,000 or more to a tax-deferred retirement or college savings account each month, while 12% do not do this on a regular basis. About two thirds of neurologists make contributions to a taxable savings account, a tool many use when tax-deferred contributions have reached their limit.
Nearly half of neurologists (48%) rely on a “mental budget” for personal expenses, while 18% rely on a written budget or use software or an app for budgeting; 34% do not have a budget for personal expenses.
Nearly three quarters of neurologists did not experience a financial loss in 2019. Of those that did, the main causes were bad investments (10%) and practice-related issues (9%), followed by legal fees (5%), real estate loss (4%), and divorce (1%).
Among neurologists with joint finances with a spouse or partner, 62% pool their income to pay household expenses. For 12%, the person who earns more pays more of the bills and/or expenses. Only a small percentage divide bills and expenses equally (4%).
Forty-three percent of neurologists said they currently work with a financial planner or had in the past, 38% never did, and 19% met with a financial planner but did not pursue working with that person.
This article first appeared on Medscape.com.
An important caveat, however, is that data for this year’s report were collected prior to Feb. 11, 2020, as part of the Medscape Physician Compensation Report 2020. The financial picture has changed for many physicians since then because of COVID-19’s impact on medical practices.
While it will be some time before medical practices become accustomed to a new version of normal, the report data provide a picture of the debt load and net worth of neurologists.
Below the middle earners
According to the Medscape Neurologist Compensation Report 2020, neurologists are below the middle earners of all physicians, earning $280,000 on average this year. That’s up from $267,000 last year. More than half of neurologists (58%) report a net worth (total assets minus total liabilities) of less than $1 million (52% men, 70% women), while 36% have a net worth between $1 and $5 million (40% men, 29% women), and 6% top $5 million in net worth (8% men, 1% women).

Among specialists, orthopedists are most likely to top the $5 million level (at 19%), followed by plastic surgeons and gastroenterologists (both at 16%), according to the Medscape Physician Debt and Net Worth Report 2020.
Conversely, about two in five neurologists (41%) have a net worth under $500,000, just below family medicine physicians (46%) and pediatricians (44%).
For roughly two thirds of neurologists (64%), their major expense is mortgage payment on their primary residence. Around one third of neurologists have a mortgage of $300,000 or less, 28% have no mortgage or a mortgage that is paid off, and 17% have a mortgage topping $500,000. Six in 10 neurologists live in a house that is 3000 square feet or smaller.
Mortgage aside, other top ongoing expenses for neurologists are car payments (35%), credit card debt (28%), school loans (25%), and childcare (19%). At 25%, neurologists are in the middle of the list when it comes to physicians still paying off loans for education.
Spender or saver?
The average American has four credit cards, according to the credit reporting agency Experian. Among neurologists, more than half said they have four or fewer credit cards, including 37% with three to four cards, 16% with one to two cards, and 1% with no cards. A little more than a quarter of neurologists (28%) have five to six credit cards and 18% have more than seven at their disposal.
Only a small percentage of neurologists (7%) say they live above their means; 57% live at their means and 35% live below their means.
More than half (59%) of neurologists contribute $1,000 or more to a tax-deferred retirement or college savings account each month, while 12% do not do this on a regular basis. About two thirds of neurologists make contributions to a taxable savings account, a tool many use when tax-deferred contributions have reached their limit.
Nearly half of neurologists (48%) rely on a “mental budget” for personal expenses, while 18% rely on a written budget or use software or an app for budgeting; 34% do not have a budget for personal expenses.
Nearly three quarters of neurologists did not experience a financial loss in 2019. Of those that did, the main causes were bad investments (10%) and practice-related issues (9%), followed by legal fees (5%), real estate loss (4%), and divorce (1%).
Among neurologists with joint finances with a spouse or partner, 62% pool their income to pay household expenses. For 12%, the person who earns more pays more of the bills and/or expenses. Only a small percentage divide bills and expenses equally (4%).
Forty-three percent of neurologists said they currently work with a financial planner or had in the past, 38% never did, and 19% met with a financial planner but did not pursue working with that person.
This article first appeared on Medscape.com.
An important caveat, however, is that data for this year’s report were collected prior to Feb. 11, 2020, as part of the Medscape Physician Compensation Report 2020. The financial picture has changed for many physicians since then because of COVID-19’s impact on medical practices.
While it will be some time before medical practices become accustomed to a new version of normal, the report data provide a picture of the debt load and net worth of neurologists.
Below the middle earners
According to the Medscape Neurologist Compensation Report 2020, neurologists are below the middle earners of all physicians, earning $280,000 on average this year. That’s up from $267,000 last year. More than half of neurologists (58%) report a net worth (total assets minus total liabilities) of less than $1 million (52% men, 70% women), while 36% have a net worth between $1 and $5 million (40% men, 29% women), and 6% top $5 million in net worth (8% men, 1% women).

Among specialists, orthopedists are most likely to top the $5 million level (at 19%), followed by plastic surgeons and gastroenterologists (both at 16%), according to the Medscape Physician Debt and Net Worth Report 2020.
Conversely, about two in five neurologists (41%) have a net worth under $500,000, just below family medicine physicians (46%) and pediatricians (44%).
For roughly two thirds of neurologists (64%), their major expense is mortgage payment on their primary residence. Around one third of neurologists have a mortgage of $300,000 or less, 28% have no mortgage or a mortgage that is paid off, and 17% have a mortgage topping $500,000. Six in 10 neurologists live in a house that is 3000 square feet or smaller.
Mortgage aside, other top ongoing expenses for neurologists are car payments (35%), credit card debt (28%), school loans (25%), and childcare (19%). At 25%, neurologists are in the middle of the list when it comes to physicians still paying off loans for education.
Spender or saver?
The average American has four credit cards, according to the credit reporting agency Experian. Among neurologists, more than half said they have four or fewer credit cards, including 37% with three to four cards, 16% with one to two cards, and 1% with no cards. A little more than a quarter of neurologists (28%) have five to six credit cards and 18% have more than seven at their disposal.
Only a small percentage of neurologists (7%) say they live above their means; 57% live at their means and 35% live below their means.
More than half (59%) of neurologists contribute $1,000 or more to a tax-deferred retirement or college savings account each month, while 12% do not do this on a regular basis. About two thirds of neurologists make contributions to a taxable savings account, a tool many use when tax-deferred contributions have reached their limit.
Nearly half of neurologists (48%) rely on a “mental budget” for personal expenses, while 18% rely on a written budget or use software or an app for budgeting; 34% do not have a budget for personal expenses.
Nearly three quarters of neurologists did not experience a financial loss in 2019. Of those that did, the main causes were bad investments (10%) and practice-related issues (9%), followed by legal fees (5%), real estate loss (4%), and divorce (1%).
Among neurologists with joint finances with a spouse or partner, 62% pool their income to pay household expenses. For 12%, the person who earns more pays more of the bills and/or expenses. Only a small percentage divide bills and expenses equally (4%).
Forty-three percent of neurologists said they currently work with a financial planner or had in the past, 38% never did, and 19% met with a financial planner but did not pursue working with that person.
This article first appeared on Medscape.com.
FDA approves low-sodium treatment option for narcolepsy
Xywav is a novel oxybate product with a unique composition of cations, resulting in 92% less sodium than sodium oxybate (Xyrem, Jazz Pharmaceuticals) at the recommended dosage range of 6 to 9 grams, the company said in a news release.
The FDA approved the drug based on a phase 3 trial involving 201 patients who had narcolepsy with cataplexy.
As reported by Medscape Medical News from the World Sleep 2019 meeting, Xywav demonstrated highly statistically significant differences (P < .0001) in weekly number of cataplexy attacks (primary efficacy endpoint) and Epworth Sleepiness Scale scores (key secondary outcome) vs placebo.
“Based on the efficacy demonstrated in the clinical program, the approval of Xywav is important for people living with cataplexy or EDS associated with narcolepsy,” lead investigator Richard K. Bogan, MD, said in the company’s news release.
He noted that the average American consumes too much sodium. “Excess sodium intake has been linked with increases in blood pressure, hypertension, stroke, and other cardiovascular disease,” said Dr. Bogan, associate clinical professor at the University of South Carolina School of Medicine, Columbia.
“Xywav makes it possible for patients to have a lower-sodium oxybate treatment option. This may help patients taking sodium oxybate better align with daily sodium intake recommendations, including those by the American Heart Association,” he added.
The overall safety profile of Xywav is in line with sodium oxybate, the company said. The most common adverse reactions in adults, occurring in at least 5% of participants, were headache, nausea, dizziness, decreased appetite, parasomnia, diarrhea, hyperhidrosis (excessive sweating), anxiety, and vomiting.
Xywav has a boxed warning as a CNS depressant and for its potential for abuse and misuse. As a result, the drug is only available through a Risk Evaluation and Mitigation Strategy (REMS) program.
The US Drug Enforcement Agency has designated Xywav as a schedule III drug, meaning it has a moderate to low potential for physical and psychological dependence.
The company plans to launch Xywav by the end of the year. Full prescribing information and a medication guide are available online.
This article first appeared on Medscape.com.
Xywav is a novel oxybate product with a unique composition of cations, resulting in 92% less sodium than sodium oxybate (Xyrem, Jazz Pharmaceuticals) at the recommended dosage range of 6 to 9 grams, the company said in a news release.
The FDA approved the drug based on a phase 3 trial involving 201 patients who had narcolepsy with cataplexy.
As reported by Medscape Medical News from the World Sleep 2019 meeting, Xywav demonstrated highly statistically significant differences (P < .0001) in weekly number of cataplexy attacks (primary efficacy endpoint) and Epworth Sleepiness Scale scores (key secondary outcome) vs placebo.
“Based on the efficacy demonstrated in the clinical program, the approval of Xywav is important for people living with cataplexy or EDS associated with narcolepsy,” lead investigator Richard K. Bogan, MD, said in the company’s news release.
He noted that the average American consumes too much sodium. “Excess sodium intake has been linked with increases in blood pressure, hypertension, stroke, and other cardiovascular disease,” said Dr. Bogan, associate clinical professor at the University of South Carolina School of Medicine, Columbia.
“Xywav makes it possible for patients to have a lower-sodium oxybate treatment option. This may help patients taking sodium oxybate better align with daily sodium intake recommendations, including those by the American Heart Association,” he added.
The overall safety profile of Xywav is in line with sodium oxybate, the company said. The most common adverse reactions in adults, occurring in at least 5% of participants, were headache, nausea, dizziness, decreased appetite, parasomnia, diarrhea, hyperhidrosis (excessive sweating), anxiety, and vomiting.
Xywav has a boxed warning as a CNS depressant and for its potential for abuse and misuse. As a result, the drug is only available through a Risk Evaluation and Mitigation Strategy (REMS) program.
The US Drug Enforcement Agency has designated Xywav as a schedule III drug, meaning it has a moderate to low potential for physical and psychological dependence.
The company plans to launch Xywav by the end of the year. Full prescribing information and a medication guide are available online.
This article first appeared on Medscape.com.
Xywav is a novel oxybate product with a unique composition of cations, resulting in 92% less sodium than sodium oxybate (Xyrem, Jazz Pharmaceuticals) at the recommended dosage range of 6 to 9 grams, the company said in a news release.
The FDA approved the drug based on a phase 3 trial involving 201 patients who had narcolepsy with cataplexy.
As reported by Medscape Medical News from the World Sleep 2019 meeting, Xywav demonstrated highly statistically significant differences (P < .0001) in weekly number of cataplexy attacks (primary efficacy endpoint) and Epworth Sleepiness Scale scores (key secondary outcome) vs placebo.
“Based on the efficacy demonstrated in the clinical program, the approval of Xywav is important for people living with cataplexy or EDS associated with narcolepsy,” lead investigator Richard K. Bogan, MD, said in the company’s news release.
He noted that the average American consumes too much sodium. “Excess sodium intake has been linked with increases in blood pressure, hypertension, stroke, and other cardiovascular disease,” said Dr. Bogan, associate clinical professor at the University of South Carolina School of Medicine, Columbia.
“Xywav makes it possible for patients to have a lower-sodium oxybate treatment option. This may help patients taking sodium oxybate better align with daily sodium intake recommendations, including those by the American Heart Association,” he added.
The overall safety profile of Xywav is in line with sodium oxybate, the company said. The most common adverse reactions in adults, occurring in at least 5% of participants, were headache, nausea, dizziness, decreased appetite, parasomnia, diarrhea, hyperhidrosis (excessive sweating), anxiety, and vomiting.
Xywav has a boxed warning as a CNS depressant and for its potential for abuse and misuse. As a result, the drug is only available through a Risk Evaluation and Mitigation Strategy (REMS) program.
The US Drug Enforcement Agency has designated Xywav as a schedule III drug, meaning it has a moderate to low potential for physical and psychological dependence.
The company plans to launch Xywav by the end of the year. Full prescribing information and a medication guide are available online.
This article first appeared on Medscape.com.
