User login
Green light therapy: A stop sign for pain?
MILWAUKEE – Exposure to green light therapy may significantly reduce pain in patients with chronic pain conditions, including migraine and fibromyalgia, an expert reported at the scientific meeting of the American Pain Society.
“There’s a subset of patients in every clinic whose pain doesn’t respond to medical therapies,” said Mohab M. Ibrahim, MD, PhD. “I always wonder what could be done with these patients.”
Dr. Ibrahim, who directs the chronic pain clinic at the University of Arizona, Tucson, walked attendees through an experimental process that began with an observation and has led to human clinical trials of green light therapy.
“Despite being a pharmacologist, I’m really interested in nonpharmacologic methods to manage pain,” he said.
Dr. Ibrahim said the idea for green light therapy came to him when he was speaking with his brother, who experiences migraines. His brother said his headaches were alleviated with time outside, in his back yard, or in one of the many parks in the city where he lives.
Knowing that spending time in nature had salutary effects in general, Dr. Ibrahim, an anesthesiologist and pain management specialist, wondered whether exposure to the sort of light found in nature, with blue skies and the green of a tree canopy, could help control pain.
To begin with, Dr. Ibrahim said, “the question was, do different colors have different behavior aspects on animals?”
Dr. Ibrahim and his collaborators exposed rats to light of various wavelengths across the color spectrum, as well as white and infrared light. They found that the rats who were exposed to blue and green light had a significantly longer latency period before withdrawing their hands and feet from a painfully hot stimulus, showing an antinociceptive effect with these wavelengths similar to that seen with analgesic medication.
“At that point, I decided to pursue green light and to forego blue, because blue can change the circadian rhythm,” said Dr. Ibrahim, adding, “Most pain patients have sleep disturbances to begin with, so to compound that issue is probably not a good idea.”
Dr. Ibrahim and his colleagues wanted to determine whether the analgesic effect had to do with rats seeing the green light or just being exposed to the light. Accordingly, the researchers fitted some rats with tiny, specially manufactured, completely opaque contact lenses. As a control, the researchers applied completely clear contact lenses to another group of rats. “I can’t tell you how many times we got bit, but by the end we got pretty good at it,” said Dr. Ibrahim.
Only the rats with clear lenses had prolonged latency in paw withdrawal to a noxious stimulus with green light exposure; for the rats with the blackout contact lenses, the effect was gone, “suggesting that the visual system is essential in mediating this effect,” noted Dr. Ibrahim.
This series of experiments also showed durable effects of green light exposure. In addition, the analgesic effect of green light did not wane over time, and higher “doses” were not required to achieve the same effect (as is the case with opioids, for example) (Pain. 2017 Feb;158[2]:347-60).
Clues to the mechanism of action came when Dr. Ibrahim and his colleagues administered naloxone to green light-exposed rats. “Naloxone reversed the effects of the green light, suggesting that the endogenous opioid system plays a role in this,” he said, adding that enkephalins were increased two- to threefold in the green light-exposed rats’ spinal cords, and astrocyte activation was reduced as well.
Similar experiments using a rat model of neuropathic pain showed a reversal of pain symptoms with green light exposure, offering promise that green light therapy could be effective in alleviating chronic as well as acute pain.
Moving to humans, Dr. Ibrahim enrolled a small group of individuals from his pain clinic who had refractory migraine into a study that exposed them either to white light or to green light. “These are patients who have failed everything…They have come to me, but I have nothing else to offer them,” he said.
The study had a crossover design. Participants in the small study had baseline pain scores of about 8/10, with no significant drop in pain with white light exposure. However, when the white light patients were crossed over to green light exposure, pain scores dropped to about 3/10. “That’s a greater than 50% reduction in the intensity of their migraine.”
Similar effects were seen in patients with fibromyalgia: “It was exactly the same story…When patients with white light exposure were crossed over [to green light], they had significant reductions in pain,” said Dr. Ibrahim.
“Their opioid use also decreased,” said Dr. Ibrahim. Medication use dropped in green light-exposed patients with migraine and fibromyalgia from an aggregate of about 280 morphine milligram equivalents (MME) to about 150 MME by the end of the study. The small size of the pilot study meant that those differences were not statistically significant.
“A multimodal approach to manage chronic pain patients is probably the best approach that we have so far,” said Dr. Ibrahim. An ongoing clinical trial randomizes patients with chronic pain to white light or green light therapy for two hours daily for 10 weeks, tracking pain scores, medication, and quality of life measures.
Future directions, said Dr. Ibrahim, include a study of the efficacy of green light therapy for patients with interstitial cystitis; another study will investigate green light for postoperative pain control. Sleep may also be improved by green light exposure, and Dr. Ibrahim and his colleagues plan to study this as well.
Dr. Ibrahim reported that his research was supported by the National Institutes of Health. He reported that he had no relevant conflicts of interest.
MILWAUKEE – Exposure to green light therapy may significantly reduce pain in patients with chronic pain conditions, including migraine and fibromyalgia, an expert reported at the scientific meeting of the American Pain Society.
“There’s a subset of patients in every clinic whose pain doesn’t respond to medical therapies,” said Mohab M. Ibrahim, MD, PhD. “I always wonder what could be done with these patients.”
Dr. Ibrahim, who directs the chronic pain clinic at the University of Arizona, Tucson, walked attendees through an experimental process that began with an observation and has led to human clinical trials of green light therapy.
“Despite being a pharmacologist, I’m really interested in nonpharmacologic methods to manage pain,” he said.
Dr. Ibrahim said the idea for green light therapy came to him when he was speaking with his brother, who experiences migraines. His brother said his headaches were alleviated with time outside, in his back yard, or in one of the many parks in the city where he lives.
Knowing that spending time in nature had salutary effects in general, Dr. Ibrahim, an anesthesiologist and pain management specialist, wondered whether exposure to the sort of light found in nature, with blue skies and the green of a tree canopy, could help control pain.
To begin with, Dr. Ibrahim said, “the question was, do different colors have different behavior aspects on animals?”
Dr. Ibrahim and his collaborators exposed rats to light of various wavelengths across the color spectrum, as well as white and infrared light. They found that the rats who were exposed to blue and green light had a significantly longer latency period before withdrawing their hands and feet from a painfully hot stimulus, showing an antinociceptive effect with these wavelengths similar to that seen with analgesic medication.
“At that point, I decided to pursue green light and to forego blue, because blue can change the circadian rhythm,” said Dr. Ibrahim, adding, “Most pain patients have sleep disturbances to begin with, so to compound that issue is probably not a good idea.”
Dr. Ibrahim and his colleagues wanted to determine whether the analgesic effect had to do with rats seeing the green light or just being exposed to the light. Accordingly, the researchers fitted some rats with tiny, specially manufactured, completely opaque contact lenses. As a control, the researchers applied completely clear contact lenses to another group of rats. “I can’t tell you how many times we got bit, but by the end we got pretty good at it,” said Dr. Ibrahim.
Only the rats with clear lenses had prolonged latency in paw withdrawal to a noxious stimulus with green light exposure; for the rats with the blackout contact lenses, the effect was gone, “suggesting that the visual system is essential in mediating this effect,” noted Dr. Ibrahim.
This series of experiments also showed durable effects of green light exposure. In addition, the analgesic effect of green light did not wane over time, and higher “doses” were not required to achieve the same effect (as is the case with opioids, for example) (Pain. 2017 Feb;158[2]:347-60).
Clues to the mechanism of action came when Dr. Ibrahim and his colleagues administered naloxone to green light-exposed rats. “Naloxone reversed the effects of the green light, suggesting that the endogenous opioid system plays a role in this,” he said, adding that enkephalins were increased two- to threefold in the green light-exposed rats’ spinal cords, and astrocyte activation was reduced as well.
Similar experiments using a rat model of neuropathic pain showed a reversal of pain symptoms with green light exposure, offering promise that green light therapy could be effective in alleviating chronic as well as acute pain.
Moving to humans, Dr. Ibrahim enrolled a small group of individuals from his pain clinic who had refractory migraine into a study that exposed them either to white light or to green light. “These are patients who have failed everything…They have come to me, but I have nothing else to offer them,” he said.
The study had a crossover design. Participants in the small study had baseline pain scores of about 8/10, with no significant drop in pain with white light exposure. However, when the white light patients were crossed over to green light exposure, pain scores dropped to about 3/10. “That’s a greater than 50% reduction in the intensity of their migraine.”
Similar effects were seen in patients with fibromyalgia: “It was exactly the same story…When patients with white light exposure were crossed over [to green light], they had significant reductions in pain,” said Dr. Ibrahim.
“Their opioid use also decreased,” said Dr. Ibrahim. Medication use dropped in green light-exposed patients with migraine and fibromyalgia from an aggregate of about 280 morphine milligram equivalents (MME) to about 150 MME by the end of the study. The small size of the pilot study meant that those differences were not statistically significant.
“A multimodal approach to manage chronic pain patients is probably the best approach that we have so far,” said Dr. Ibrahim. An ongoing clinical trial randomizes patients with chronic pain to white light or green light therapy for two hours daily for 10 weeks, tracking pain scores, medication, and quality of life measures.
Future directions, said Dr. Ibrahim, include a study of the efficacy of green light therapy for patients with interstitial cystitis; another study will investigate green light for postoperative pain control. Sleep may also be improved by green light exposure, and Dr. Ibrahim and his colleagues plan to study this as well.
Dr. Ibrahim reported that his research was supported by the National Institutes of Health. He reported that he had no relevant conflicts of interest.
MILWAUKEE – Exposure to green light therapy may significantly reduce pain in patients with chronic pain conditions, including migraine and fibromyalgia, an expert reported at the scientific meeting of the American Pain Society.
“There’s a subset of patients in every clinic whose pain doesn’t respond to medical therapies,” said Mohab M. Ibrahim, MD, PhD. “I always wonder what could be done with these patients.”
Dr. Ibrahim, who directs the chronic pain clinic at the University of Arizona, Tucson, walked attendees through an experimental process that began with an observation and has led to human clinical trials of green light therapy.
“Despite being a pharmacologist, I’m really interested in nonpharmacologic methods to manage pain,” he said.
Dr. Ibrahim said the idea for green light therapy came to him when he was speaking with his brother, who experiences migraines. His brother said his headaches were alleviated with time outside, in his back yard, or in one of the many parks in the city where he lives.
Knowing that spending time in nature had salutary effects in general, Dr. Ibrahim, an anesthesiologist and pain management specialist, wondered whether exposure to the sort of light found in nature, with blue skies and the green of a tree canopy, could help control pain.
To begin with, Dr. Ibrahim said, “the question was, do different colors have different behavior aspects on animals?”
Dr. Ibrahim and his collaborators exposed rats to light of various wavelengths across the color spectrum, as well as white and infrared light. They found that the rats who were exposed to blue and green light had a significantly longer latency period before withdrawing their hands and feet from a painfully hot stimulus, showing an antinociceptive effect with these wavelengths similar to that seen with analgesic medication.
“At that point, I decided to pursue green light and to forego blue, because blue can change the circadian rhythm,” said Dr. Ibrahim, adding, “Most pain patients have sleep disturbances to begin with, so to compound that issue is probably not a good idea.”
Dr. Ibrahim and his colleagues wanted to determine whether the analgesic effect had to do with rats seeing the green light or just being exposed to the light. Accordingly, the researchers fitted some rats with tiny, specially manufactured, completely opaque contact lenses. As a control, the researchers applied completely clear contact lenses to another group of rats. “I can’t tell you how many times we got bit, but by the end we got pretty good at it,” said Dr. Ibrahim.
Only the rats with clear lenses had prolonged latency in paw withdrawal to a noxious stimulus with green light exposure; for the rats with the blackout contact lenses, the effect was gone, “suggesting that the visual system is essential in mediating this effect,” noted Dr. Ibrahim.
This series of experiments also showed durable effects of green light exposure. In addition, the analgesic effect of green light did not wane over time, and higher “doses” were not required to achieve the same effect (as is the case with opioids, for example) (Pain. 2017 Feb;158[2]:347-60).
Clues to the mechanism of action came when Dr. Ibrahim and his colleagues administered naloxone to green light-exposed rats. “Naloxone reversed the effects of the green light, suggesting that the endogenous opioid system plays a role in this,” he said, adding that enkephalins were increased two- to threefold in the green light-exposed rats’ spinal cords, and astrocyte activation was reduced as well.
Similar experiments using a rat model of neuropathic pain showed a reversal of pain symptoms with green light exposure, offering promise that green light therapy could be effective in alleviating chronic as well as acute pain.
Moving to humans, Dr. Ibrahim enrolled a small group of individuals from his pain clinic who had refractory migraine into a study that exposed them either to white light or to green light. “These are patients who have failed everything…They have come to me, but I have nothing else to offer them,” he said.
The study had a crossover design. Participants in the small study had baseline pain scores of about 8/10, with no significant drop in pain with white light exposure. However, when the white light patients were crossed over to green light exposure, pain scores dropped to about 3/10. “That’s a greater than 50% reduction in the intensity of their migraine.”
Similar effects were seen in patients with fibromyalgia: “It was exactly the same story…When patients with white light exposure were crossed over [to green light], they had significant reductions in pain,” said Dr. Ibrahim.
“Their opioid use also decreased,” said Dr. Ibrahim. Medication use dropped in green light-exposed patients with migraine and fibromyalgia from an aggregate of about 280 morphine milligram equivalents (MME) to about 150 MME by the end of the study. The small size of the pilot study meant that those differences were not statistically significant.
“A multimodal approach to manage chronic pain patients is probably the best approach that we have so far,” said Dr. Ibrahim. An ongoing clinical trial randomizes patients with chronic pain to white light or green light therapy for two hours daily for 10 weeks, tracking pain scores, medication, and quality of life measures.
Future directions, said Dr. Ibrahim, include a study of the efficacy of green light therapy for patients with interstitial cystitis; another study will investigate green light for postoperative pain control. Sleep may also be improved by green light exposure, and Dr. Ibrahim and his colleagues plan to study this as well.
Dr. Ibrahim reported that his research was supported by the National Institutes of Health. He reported that he had no relevant conflicts of interest.
REPORTING FROM APS 2019
In a tight vote, FDA panel backs mannitol for CF
A Food and Drug Administration Advisory Committee voted that the benefit-risk profile of an inhaled treatment for cystic fibrosis merits approval of the drug – dry powder mannitol (DPM).
Mannitol is a naturally occurring sugar alcohol that is used as a low-calorie sweetener; it is generally recognized as safe when taken enterically. Inhaled DPM, marketed as Aridol, is currently approved as a bronchoprovocation agent. For the current indication, DPM is given as 10x40-mg capsules twice daily.
In a 9-7 vote, the FDA’s Pulmonary-Allergy Drugs Advisory Committee (PADAC) decided that DPM’s modest potential to improve pulmonary function in adults with cystic fibrosis (CF) outweighed a potential signal for increased exacerbations seen in clinical trials.
Chiesi USA Inc. is seeking approval of DPM for the management of cystic fibrosis to improve pulmonary function in patients 18 years of age and older in conjunction with standard therapies. It plans to market DPM as Bronchitol.
Some committee members who voted against approval, including PADAC chair David H. Au, MD, worried that DPM’s ease of use might prompt patients and caregivers to substitute it for inhaled hypertonic saline, a medication that’s more burdensome to use but has a longer track record for efficacy and safety. While hypertonic saline requires cumbersome equipment and cleaning regimens and takes 20-30 minutes to administer, DPM is administered over about 5 minutes via a series of capsules inserted into a small inhaler device.
“I was very impressed by conversations that we heard from the community that this will be viewed as a substitute drug [for hypertonic saline],” said Dr. Au, professor of medicine at the University of Washington, Seattle. “Before we make that leap of faith ... we have to better understand how it has to be used.” He also acknowledged that making the call for DPM was “challenging.”
Other committee members were reassured by the fact that DPM is approved for adult use in 35 countries; it’s been in use since 2011 in Australia for adults and children.
Some members also noted an unmet need in CF therapies and placed confidence in those treating CF patients to find ways to use DPM safely and effectively. “I’m really counting on the cystic fibrosis clinicians who do this for a living to figure out where to use this in their armamentarium,” said John M. Kelso, MD, an allergist at Scripps Clinic, San Diego.
In 2012, the initial new drug application submitted by Pharmaxis, which then held marketing rights to DPM, resulted in a “no” vote for approval from PADAC, and eventual FDA denial of approval. The initial submission was supported by two phase 3 clinical trials, 301 and 302, that included pediatric patients. In the pediatric population, there was concern for increased hemoptysis with DPM, so the FDA advised the drug’s marketers to consider seeking approval for an adult population only in its reapplication. The current submission followed a new double-blind, randomized, placebo-controlled trial, study 303, that included adults with CF aged 18 or over.
All three studies had similar designs, tracking change from baseline in forced expiratory volume in one second (FEV1) from baseline to the end of the 26-week study period. In addition to this primary endpoint, secondary endpoints included other pulmonary function measures, as well as the number of protocol-defined pulmonary exacerbations (PDPEs). Participants also reported quality of life and symptom measures on the Cystic Fibrosis Questionnaire–Revised (CFQ-R).
In study 301, the dropout rate approached one in three participants with higher discontinuation in the intervention than the control arm, causing significant statistical problems in dealing with missing data. Thus, said the FDA’s Robert Lim, MD, though this study had positive results for FEV1, it was not “statistically robust.”
The second study, 302, did not meet its primary endpoint, and there was “no support from secondary endpoints” for efficacy, said Dr. Lim, a clinical team leader in the FDA’s Division of Pulmonary, Allergy, and Rheumatology Products.
The current submission was also supported by a new post hoc subgroup analysis of adults in studies 301 and 302. A total of 414 patients receiving DPM and 347 receiving placebo (DPM at a nontherapeutic level) were included in the integrated analysis of patients from all three studies. Studies 301 and 302 both had open-label extension arms, allowing more patients to be included in safety data.
The problems caused by the missing data from study 301 were addressed in the design of study 303 by encouraging patients who discontinued the study drug to continue data collection efforts for the study. Dropout rates were lower overall in study 303 and balanced between arms.
Over the 26-week duration of study 303, investigators saw a statistically significant improvement in FEV1 of about 50 mL, according to the FDA’s analysis. Post hoc analyses of studies 301 and 302 showed point estimate increases of approximately 80 mL, according to Dr. Lim.
In its presentations, Chiesi USA presented its integrated analysis of adult data from the three clinical trials. The analysis showed an increase in FEV1 from baseline of 73 mL for the DPM group, compared with an increase of 7 mL for the control group, using an intention-to-treat population (P less than .001). The committee heard evidence that in adults with CF, pulmonary function typically decreases by 1%-3% annually.
The PDPE rate was slightly higher in the DPM group than in the control group in studies 302 and 303, but the differences were not statistically significant. These findings have a backdrop of an overall low rate of PDPEs ranging from 0.221 to 0.995 per year, according to Chiesi presenter Scott Donaldson, MD, a pulmonologist who directs the adult cystic fibrosis center at the University of North Carolina at Chapel Hill.
When looking at the subgroup of United States study participants, the DPM integrated cohort included more patients with a history of prior pulmonary exacerbations. In the DPM group, 45% of U.S. participants had at least one exacerbation in the prior year, and 20% had two or more exacerbations, compared with 38% and 14%, respectively, in the control group. Chiesi argued that this imbalance was likely responsible for the increased exacerbation rate.
The sponsor and the FDA used different imputation methods to account for missing data from the earlier studies, complicating interpretation of the potential signal for increased exacerbations.
Quality of life data were similar between groups across the studies.
In the end, the view of the “yes” voters was encapsulated by James M. Tracy, DO, an allergist in private practice in Omaha, Neb. “This is not a drug for everybody; but absolutely, it’s a drug for somebody. Ultimately we have to make that decision – I do think that we study populations, but we really take care of people.”
The FDA usually follows the recommendations of its advisory panels.
A Food and Drug Administration Advisory Committee voted that the benefit-risk profile of an inhaled treatment for cystic fibrosis merits approval of the drug – dry powder mannitol (DPM).
Mannitol is a naturally occurring sugar alcohol that is used as a low-calorie sweetener; it is generally recognized as safe when taken enterically. Inhaled DPM, marketed as Aridol, is currently approved as a bronchoprovocation agent. For the current indication, DPM is given as 10x40-mg capsules twice daily.
In a 9-7 vote, the FDA’s Pulmonary-Allergy Drugs Advisory Committee (PADAC) decided that DPM’s modest potential to improve pulmonary function in adults with cystic fibrosis (CF) outweighed a potential signal for increased exacerbations seen in clinical trials.
Chiesi USA Inc. is seeking approval of DPM for the management of cystic fibrosis to improve pulmonary function in patients 18 years of age and older in conjunction with standard therapies. It plans to market DPM as Bronchitol.
Some committee members who voted against approval, including PADAC chair David H. Au, MD, worried that DPM’s ease of use might prompt patients and caregivers to substitute it for inhaled hypertonic saline, a medication that’s more burdensome to use but has a longer track record for efficacy and safety. While hypertonic saline requires cumbersome equipment and cleaning regimens and takes 20-30 minutes to administer, DPM is administered over about 5 minutes via a series of capsules inserted into a small inhaler device.
“I was very impressed by conversations that we heard from the community that this will be viewed as a substitute drug [for hypertonic saline],” said Dr. Au, professor of medicine at the University of Washington, Seattle. “Before we make that leap of faith ... we have to better understand how it has to be used.” He also acknowledged that making the call for DPM was “challenging.”
Other committee members were reassured by the fact that DPM is approved for adult use in 35 countries; it’s been in use since 2011 in Australia for adults and children.
Some members also noted an unmet need in CF therapies and placed confidence in those treating CF patients to find ways to use DPM safely and effectively. “I’m really counting on the cystic fibrosis clinicians who do this for a living to figure out where to use this in their armamentarium,” said John M. Kelso, MD, an allergist at Scripps Clinic, San Diego.
In 2012, the initial new drug application submitted by Pharmaxis, which then held marketing rights to DPM, resulted in a “no” vote for approval from PADAC, and eventual FDA denial of approval. The initial submission was supported by two phase 3 clinical trials, 301 and 302, that included pediatric patients. In the pediatric population, there was concern for increased hemoptysis with DPM, so the FDA advised the drug’s marketers to consider seeking approval for an adult population only in its reapplication. The current submission followed a new double-blind, randomized, placebo-controlled trial, study 303, that included adults with CF aged 18 or over.
All three studies had similar designs, tracking change from baseline in forced expiratory volume in one second (FEV1) from baseline to the end of the 26-week study period. In addition to this primary endpoint, secondary endpoints included other pulmonary function measures, as well as the number of protocol-defined pulmonary exacerbations (PDPEs). Participants also reported quality of life and symptom measures on the Cystic Fibrosis Questionnaire–Revised (CFQ-R).
In study 301, the dropout rate approached one in three participants with higher discontinuation in the intervention than the control arm, causing significant statistical problems in dealing with missing data. Thus, said the FDA’s Robert Lim, MD, though this study had positive results for FEV1, it was not “statistically robust.”
The second study, 302, did not meet its primary endpoint, and there was “no support from secondary endpoints” for efficacy, said Dr. Lim, a clinical team leader in the FDA’s Division of Pulmonary, Allergy, and Rheumatology Products.
The current submission was also supported by a new post hoc subgroup analysis of adults in studies 301 and 302. A total of 414 patients receiving DPM and 347 receiving placebo (DPM at a nontherapeutic level) were included in the integrated analysis of patients from all three studies. Studies 301 and 302 both had open-label extension arms, allowing more patients to be included in safety data.
The problems caused by the missing data from study 301 were addressed in the design of study 303 by encouraging patients who discontinued the study drug to continue data collection efforts for the study. Dropout rates were lower overall in study 303 and balanced between arms.
Over the 26-week duration of study 303, investigators saw a statistically significant improvement in FEV1 of about 50 mL, according to the FDA’s analysis. Post hoc analyses of studies 301 and 302 showed point estimate increases of approximately 80 mL, according to Dr. Lim.
In its presentations, Chiesi USA presented its integrated analysis of adult data from the three clinical trials. The analysis showed an increase in FEV1 from baseline of 73 mL for the DPM group, compared with an increase of 7 mL for the control group, using an intention-to-treat population (P less than .001). The committee heard evidence that in adults with CF, pulmonary function typically decreases by 1%-3% annually.
The PDPE rate was slightly higher in the DPM group than in the control group in studies 302 and 303, but the differences were not statistically significant. These findings have a backdrop of an overall low rate of PDPEs ranging from 0.221 to 0.995 per year, according to Chiesi presenter Scott Donaldson, MD, a pulmonologist who directs the adult cystic fibrosis center at the University of North Carolina at Chapel Hill.
When looking at the subgroup of United States study participants, the DPM integrated cohort included more patients with a history of prior pulmonary exacerbations. In the DPM group, 45% of U.S. participants had at least one exacerbation in the prior year, and 20% had two or more exacerbations, compared with 38% and 14%, respectively, in the control group. Chiesi argued that this imbalance was likely responsible for the increased exacerbation rate.
The sponsor and the FDA used different imputation methods to account for missing data from the earlier studies, complicating interpretation of the potential signal for increased exacerbations.
Quality of life data were similar between groups across the studies.
In the end, the view of the “yes” voters was encapsulated by James M. Tracy, DO, an allergist in private practice in Omaha, Neb. “This is not a drug for everybody; but absolutely, it’s a drug for somebody. Ultimately we have to make that decision – I do think that we study populations, but we really take care of people.”
The FDA usually follows the recommendations of its advisory panels.
A Food and Drug Administration Advisory Committee voted that the benefit-risk profile of an inhaled treatment for cystic fibrosis merits approval of the drug – dry powder mannitol (DPM).
Mannitol is a naturally occurring sugar alcohol that is used as a low-calorie sweetener; it is generally recognized as safe when taken enterically. Inhaled DPM, marketed as Aridol, is currently approved as a bronchoprovocation agent. For the current indication, DPM is given as 10x40-mg capsules twice daily.
In a 9-7 vote, the FDA’s Pulmonary-Allergy Drugs Advisory Committee (PADAC) decided that DPM’s modest potential to improve pulmonary function in adults with cystic fibrosis (CF) outweighed a potential signal for increased exacerbations seen in clinical trials.
Chiesi USA Inc. is seeking approval of DPM for the management of cystic fibrosis to improve pulmonary function in patients 18 years of age and older in conjunction with standard therapies. It plans to market DPM as Bronchitol.
Some committee members who voted against approval, including PADAC chair David H. Au, MD, worried that DPM’s ease of use might prompt patients and caregivers to substitute it for inhaled hypertonic saline, a medication that’s more burdensome to use but has a longer track record for efficacy and safety. While hypertonic saline requires cumbersome equipment and cleaning regimens and takes 20-30 minutes to administer, DPM is administered over about 5 minutes via a series of capsules inserted into a small inhaler device.
“I was very impressed by conversations that we heard from the community that this will be viewed as a substitute drug [for hypertonic saline],” said Dr. Au, professor of medicine at the University of Washington, Seattle. “Before we make that leap of faith ... we have to better understand how it has to be used.” He also acknowledged that making the call for DPM was “challenging.”
Other committee members were reassured by the fact that DPM is approved for adult use in 35 countries; it’s been in use since 2011 in Australia for adults and children.
Some members also noted an unmet need in CF therapies and placed confidence in those treating CF patients to find ways to use DPM safely and effectively. “I’m really counting on the cystic fibrosis clinicians who do this for a living to figure out where to use this in their armamentarium,” said John M. Kelso, MD, an allergist at Scripps Clinic, San Diego.
In 2012, the initial new drug application submitted by Pharmaxis, which then held marketing rights to DPM, resulted in a “no” vote for approval from PADAC, and eventual FDA denial of approval. The initial submission was supported by two phase 3 clinical trials, 301 and 302, that included pediatric patients. In the pediatric population, there was concern for increased hemoptysis with DPM, so the FDA advised the drug’s marketers to consider seeking approval for an adult population only in its reapplication. The current submission followed a new double-blind, randomized, placebo-controlled trial, study 303, that included adults with CF aged 18 or over.
All three studies had similar designs, tracking change from baseline in forced expiratory volume in one second (FEV1) from baseline to the end of the 26-week study period. In addition to this primary endpoint, secondary endpoints included other pulmonary function measures, as well as the number of protocol-defined pulmonary exacerbations (PDPEs). Participants also reported quality of life and symptom measures on the Cystic Fibrosis Questionnaire–Revised (CFQ-R).
In study 301, the dropout rate approached one in three participants with higher discontinuation in the intervention than the control arm, causing significant statistical problems in dealing with missing data. Thus, said the FDA’s Robert Lim, MD, though this study had positive results for FEV1, it was not “statistically robust.”
The second study, 302, did not meet its primary endpoint, and there was “no support from secondary endpoints” for efficacy, said Dr. Lim, a clinical team leader in the FDA’s Division of Pulmonary, Allergy, and Rheumatology Products.
The current submission was also supported by a new post hoc subgroup analysis of adults in studies 301 and 302. A total of 414 patients receiving DPM and 347 receiving placebo (DPM at a nontherapeutic level) were included in the integrated analysis of patients from all three studies. Studies 301 and 302 both had open-label extension arms, allowing more patients to be included in safety data.
The problems caused by the missing data from study 301 were addressed in the design of study 303 by encouraging patients who discontinued the study drug to continue data collection efforts for the study. Dropout rates were lower overall in study 303 and balanced between arms.
Over the 26-week duration of study 303, investigators saw a statistically significant improvement in FEV1 of about 50 mL, according to the FDA’s analysis. Post hoc analyses of studies 301 and 302 showed point estimate increases of approximately 80 mL, according to Dr. Lim.
In its presentations, Chiesi USA presented its integrated analysis of adult data from the three clinical trials. The analysis showed an increase in FEV1 from baseline of 73 mL for the DPM group, compared with an increase of 7 mL for the control group, using an intention-to-treat population (P less than .001). The committee heard evidence that in adults with CF, pulmonary function typically decreases by 1%-3% annually.
The PDPE rate was slightly higher in the DPM group than in the control group in studies 302 and 303, but the differences were not statistically significant. These findings have a backdrop of an overall low rate of PDPEs ranging from 0.221 to 0.995 per year, according to Chiesi presenter Scott Donaldson, MD, a pulmonologist who directs the adult cystic fibrosis center at the University of North Carolina at Chapel Hill.
When looking at the subgroup of United States study participants, the DPM integrated cohort included more patients with a history of prior pulmonary exacerbations. In the DPM group, 45% of U.S. participants had at least one exacerbation in the prior year, and 20% had two or more exacerbations, compared with 38% and 14%, respectively, in the control group. Chiesi argued that this imbalance was likely responsible for the increased exacerbation rate.
The sponsor and the FDA used different imputation methods to account for missing data from the earlier studies, complicating interpretation of the potential signal for increased exacerbations.
Quality of life data were similar between groups across the studies.
In the end, the view of the “yes” voters was encapsulated by James M. Tracy, DO, an allergist in private practice in Omaha, Neb. “This is not a drug for everybody; but absolutely, it’s a drug for somebody. Ultimately we have to make that decision – I do think that we study populations, but we really take care of people.”
The FDA usually follows the recommendations of its advisory panels.
FROM AN FDA ADVISORY COMMITTEE HEARING
Sleep, chronic pain, and OUD have a complex relationship
MILWAUKEE – Individuals with chronic pain frequently have disrupted sleep and also may be at risk for opioid use disorder. However, even with advanced monitoring, it’s not clear how sleep modulates pain and opioid cravings.
Sleep has an impact on positive and negative affect, but new research shows that the link between sleep and mood states that may contribute to opioid use disorder is not straightforward. At the scientific meeting of the American Pain Society, Patrick Finan, PhD, of Johns Hopkins University, Baltimore, discussed how sleep and mood affect cravings for opioids among those in treatment for opioid use disorder (OUD).
said Dr. Finan, who told attendees that one key question he and his colleagues were seeking to answer was whether those with OUD and chronic pain had more disturbed sleep than those with OUD alone. Also, the researchers wanted to know whether the ups and downs of sleep on a day-to-day basis were reflected in pain scores among those with OUD, as would be predicted by prevailing models.
Finally, two “proximal indicators” of relapse risk, affect and heroin craving, might be affected by both sleep and pain, and Dr. Finan and collaborators sought to explore that association.
The work was part of a larger study looking at the natural history of OUD and OUD with comorbid chronic pain. To participate in this parent study, adults with OUD had to be seeking treatment or currently enrolled in methadone or buprenorphine maintenance treatment, and without current major depressive disorder. Also, patients could not have a history of significant mental illness, cognitive impairment, or a medical condition that would interfere with study participation. A total of 56 patients participated, and 20 of these individuals also had chronic pain.
Those with OUD and chronic pain qualified if they had pain (not related to opioid withdrawal) averaging above 3 on a 0-10 pain rating scale over the past week; additional criteria included pain for at least the past 3 months, with 10 or more days per month of pain.
Pain ratings were captured via a smartphone app that prompted participants to enter a pain rating at three random times during each day. Each evening, patients also completed a sleep diary giving information about bedtime, sleep onset latency, waking after sleep onset, and wake time for the preceding day.
A self-applied ambulatory electroencephalogram applied to the forehead was used for up to 7 consecutive nights to capture sleep continuity estimates; the device has been validated against polysomnography data in other work. Participants were given incentives to use the device, and this “yielded strong adherence,” with an average of 5 nights of use per participant, Dr. Finan said.
Patients were an average age of about 49 years, and were 75% male. African American participants made up just over half of the cohort, and 43% were white. Participants were roughly evenly divided in the type of maintenance therapy they were taking. Overall, 39% of participants had a positive urine toxicology screen.
For patients with chronic pain, 45% of all momentary pain reports had a pain score over zero, with a mean of 32 days of pain. Looking at the data another way, 58% of all patient-days had at least one momentary report of pain greater than zero, said Dr. Finan. On average, participants recorded a pain score of 2.27.
Brief Pain Inventory scores at baseline showed a mean severity of 5, and a pain interference score of 5.07.
Participants with OUD and chronic pain did not differ across any EEG-recorded sleep measures, compared with those with OUD alone. However, subjective reports of sleep were actually better overall for those with chronic pain than the objective EEG reports. The EEG recordings captured an average of 9.11 minutes more of waking after sleep onset (P less than .001). Also, total sleep time was 10.37 minutes shorter as recorded by the EEG than by self-report (P less than .001). Overall sleep efficiency was also worse by 5.96 minutes according to the EEG, compared with self-report (P less than .001).
“Sleep is objectively poor but subjectively ‘normal’ and variable in opioid use disorder patients,” Dr. Finan said. In aggregate, however, neither diary-based subjective nor EEG-based objective sleep measures differed between those with and without chronic pain in the research cohort. This phenomenon of sleep efficiency being self-reported as higher than objective measures capture sleep has also been seen in those newly abstinent from cocaine, Dr. Finan said, adding that it’s possible individuals with substance use disorder who are new to treatment simply feel better than they have in some time along many dimensions, with sleep being one such domain.
Pain on a given day didn’t predict poor sleep on that night, except that sleep onset took slightly longer (P = .01), said Dr. Finan. He noted that “there was no substantive effect on other sleep continuity parameters.”
Looking at how negative affect mediated craving for heroin, Dr. Finan and colleagues found that negative affect–related craving was significantly greater for those with chronic pain (P less than .001). Unlike findings in patients without OUD, having disrupted sleep continuity was more associated with increased daily negative affect, rather than decreased positive affect. And this increased negative affect was associated with heroin cravings, said Dr. Finan. “In the past few years, we’ve seen quite a few studies that have found some abnormalities in the reward system in patients with chronic pain.” Whether poor sleep is a mediator of these abnormalities deserves further study.
The study was supported by the National Institutes of Health. Dr. Finan reported no outside sources of funding.
MILWAUKEE – Individuals with chronic pain frequently have disrupted sleep and also may be at risk for opioid use disorder. However, even with advanced monitoring, it’s not clear how sleep modulates pain and opioid cravings.
Sleep has an impact on positive and negative affect, but new research shows that the link between sleep and mood states that may contribute to opioid use disorder is not straightforward. At the scientific meeting of the American Pain Society, Patrick Finan, PhD, of Johns Hopkins University, Baltimore, discussed how sleep and mood affect cravings for opioids among those in treatment for opioid use disorder (OUD).
said Dr. Finan, who told attendees that one key question he and his colleagues were seeking to answer was whether those with OUD and chronic pain had more disturbed sleep than those with OUD alone. Also, the researchers wanted to know whether the ups and downs of sleep on a day-to-day basis were reflected in pain scores among those with OUD, as would be predicted by prevailing models.
Finally, two “proximal indicators” of relapse risk, affect and heroin craving, might be affected by both sleep and pain, and Dr. Finan and collaborators sought to explore that association.
The work was part of a larger study looking at the natural history of OUD and OUD with comorbid chronic pain. To participate in this parent study, adults with OUD had to be seeking treatment or currently enrolled in methadone or buprenorphine maintenance treatment, and without current major depressive disorder. Also, patients could not have a history of significant mental illness, cognitive impairment, or a medical condition that would interfere with study participation. A total of 56 patients participated, and 20 of these individuals also had chronic pain.
Those with OUD and chronic pain qualified if they had pain (not related to opioid withdrawal) averaging above 3 on a 0-10 pain rating scale over the past week; additional criteria included pain for at least the past 3 months, with 10 or more days per month of pain.
Pain ratings were captured via a smartphone app that prompted participants to enter a pain rating at three random times during each day. Each evening, patients also completed a sleep diary giving information about bedtime, sleep onset latency, waking after sleep onset, and wake time for the preceding day.
A self-applied ambulatory electroencephalogram applied to the forehead was used for up to 7 consecutive nights to capture sleep continuity estimates; the device has been validated against polysomnography data in other work. Participants were given incentives to use the device, and this “yielded strong adherence,” with an average of 5 nights of use per participant, Dr. Finan said.
Patients were an average age of about 49 years, and were 75% male. African American participants made up just over half of the cohort, and 43% were white. Participants were roughly evenly divided in the type of maintenance therapy they were taking. Overall, 39% of participants had a positive urine toxicology screen.
For patients with chronic pain, 45% of all momentary pain reports had a pain score over zero, with a mean of 32 days of pain. Looking at the data another way, 58% of all patient-days had at least one momentary report of pain greater than zero, said Dr. Finan. On average, participants recorded a pain score of 2.27.
Brief Pain Inventory scores at baseline showed a mean severity of 5, and a pain interference score of 5.07.
Participants with OUD and chronic pain did not differ across any EEG-recorded sleep measures, compared with those with OUD alone. However, subjective reports of sleep were actually better overall for those with chronic pain than the objective EEG reports. The EEG recordings captured an average of 9.11 minutes more of waking after sleep onset (P less than .001). Also, total sleep time was 10.37 minutes shorter as recorded by the EEG than by self-report (P less than .001). Overall sleep efficiency was also worse by 5.96 minutes according to the EEG, compared with self-report (P less than .001).
“Sleep is objectively poor but subjectively ‘normal’ and variable in opioid use disorder patients,” Dr. Finan said. In aggregate, however, neither diary-based subjective nor EEG-based objective sleep measures differed between those with and without chronic pain in the research cohort. This phenomenon of sleep efficiency being self-reported as higher than objective measures capture sleep has also been seen in those newly abstinent from cocaine, Dr. Finan said, adding that it’s possible individuals with substance use disorder who are new to treatment simply feel better than they have in some time along many dimensions, with sleep being one such domain.
Pain on a given day didn’t predict poor sleep on that night, except that sleep onset took slightly longer (P = .01), said Dr. Finan. He noted that “there was no substantive effect on other sleep continuity parameters.”
Looking at how negative affect mediated craving for heroin, Dr. Finan and colleagues found that negative affect–related craving was significantly greater for those with chronic pain (P less than .001). Unlike findings in patients without OUD, having disrupted sleep continuity was more associated with increased daily negative affect, rather than decreased positive affect. And this increased negative affect was associated with heroin cravings, said Dr. Finan. “In the past few years, we’ve seen quite a few studies that have found some abnormalities in the reward system in patients with chronic pain.” Whether poor sleep is a mediator of these abnormalities deserves further study.
The study was supported by the National Institutes of Health. Dr. Finan reported no outside sources of funding.
MILWAUKEE – Individuals with chronic pain frequently have disrupted sleep and also may be at risk for opioid use disorder. However, even with advanced monitoring, it’s not clear how sleep modulates pain and opioid cravings.
Sleep has an impact on positive and negative affect, but new research shows that the link between sleep and mood states that may contribute to opioid use disorder is not straightforward. At the scientific meeting of the American Pain Society, Patrick Finan, PhD, of Johns Hopkins University, Baltimore, discussed how sleep and mood affect cravings for opioids among those in treatment for opioid use disorder (OUD).
said Dr. Finan, who told attendees that one key question he and his colleagues were seeking to answer was whether those with OUD and chronic pain had more disturbed sleep than those with OUD alone. Also, the researchers wanted to know whether the ups and downs of sleep on a day-to-day basis were reflected in pain scores among those with OUD, as would be predicted by prevailing models.
Finally, two “proximal indicators” of relapse risk, affect and heroin craving, might be affected by both sleep and pain, and Dr. Finan and collaborators sought to explore that association.
The work was part of a larger study looking at the natural history of OUD and OUD with comorbid chronic pain. To participate in this parent study, adults with OUD had to be seeking treatment or currently enrolled in methadone or buprenorphine maintenance treatment, and without current major depressive disorder. Also, patients could not have a history of significant mental illness, cognitive impairment, or a medical condition that would interfere with study participation. A total of 56 patients participated, and 20 of these individuals also had chronic pain.
Those with OUD and chronic pain qualified if they had pain (not related to opioid withdrawal) averaging above 3 on a 0-10 pain rating scale over the past week; additional criteria included pain for at least the past 3 months, with 10 or more days per month of pain.
Pain ratings were captured via a smartphone app that prompted participants to enter a pain rating at three random times during each day. Each evening, patients also completed a sleep diary giving information about bedtime, sleep onset latency, waking after sleep onset, and wake time for the preceding day.
A self-applied ambulatory electroencephalogram applied to the forehead was used for up to 7 consecutive nights to capture sleep continuity estimates; the device has been validated against polysomnography data in other work. Participants were given incentives to use the device, and this “yielded strong adherence,” with an average of 5 nights of use per participant, Dr. Finan said.
Patients were an average age of about 49 years, and were 75% male. African American participants made up just over half of the cohort, and 43% were white. Participants were roughly evenly divided in the type of maintenance therapy they were taking. Overall, 39% of participants had a positive urine toxicology screen.
For patients with chronic pain, 45% of all momentary pain reports had a pain score over zero, with a mean of 32 days of pain. Looking at the data another way, 58% of all patient-days had at least one momentary report of pain greater than zero, said Dr. Finan. On average, participants recorded a pain score of 2.27.
Brief Pain Inventory scores at baseline showed a mean severity of 5, and a pain interference score of 5.07.
Participants with OUD and chronic pain did not differ across any EEG-recorded sleep measures, compared with those with OUD alone. However, subjective reports of sleep were actually better overall for those with chronic pain than the objective EEG reports. The EEG recordings captured an average of 9.11 minutes more of waking after sleep onset (P less than .001). Also, total sleep time was 10.37 minutes shorter as recorded by the EEG than by self-report (P less than .001). Overall sleep efficiency was also worse by 5.96 minutes according to the EEG, compared with self-report (P less than .001).
“Sleep is objectively poor but subjectively ‘normal’ and variable in opioid use disorder patients,” Dr. Finan said. In aggregate, however, neither diary-based subjective nor EEG-based objective sleep measures differed between those with and without chronic pain in the research cohort. This phenomenon of sleep efficiency being self-reported as higher than objective measures capture sleep has also been seen in those newly abstinent from cocaine, Dr. Finan said, adding that it’s possible individuals with substance use disorder who are new to treatment simply feel better than they have in some time along many dimensions, with sleep being one such domain.
Pain on a given day didn’t predict poor sleep on that night, except that sleep onset took slightly longer (P = .01), said Dr. Finan. He noted that “there was no substantive effect on other sleep continuity parameters.”
Looking at how negative affect mediated craving for heroin, Dr. Finan and colleagues found that negative affect–related craving was significantly greater for those with chronic pain (P less than .001). Unlike findings in patients without OUD, having disrupted sleep continuity was more associated with increased daily negative affect, rather than decreased positive affect. And this increased negative affect was associated with heroin cravings, said Dr. Finan. “In the past few years, we’ve seen quite a few studies that have found some abnormalities in the reward system in patients with chronic pain.” Whether poor sleep is a mediator of these abnormalities deserves further study.
The study was supported by the National Institutes of Health. Dr. Finan reported no outside sources of funding.
REPORTING FROM APS 2019
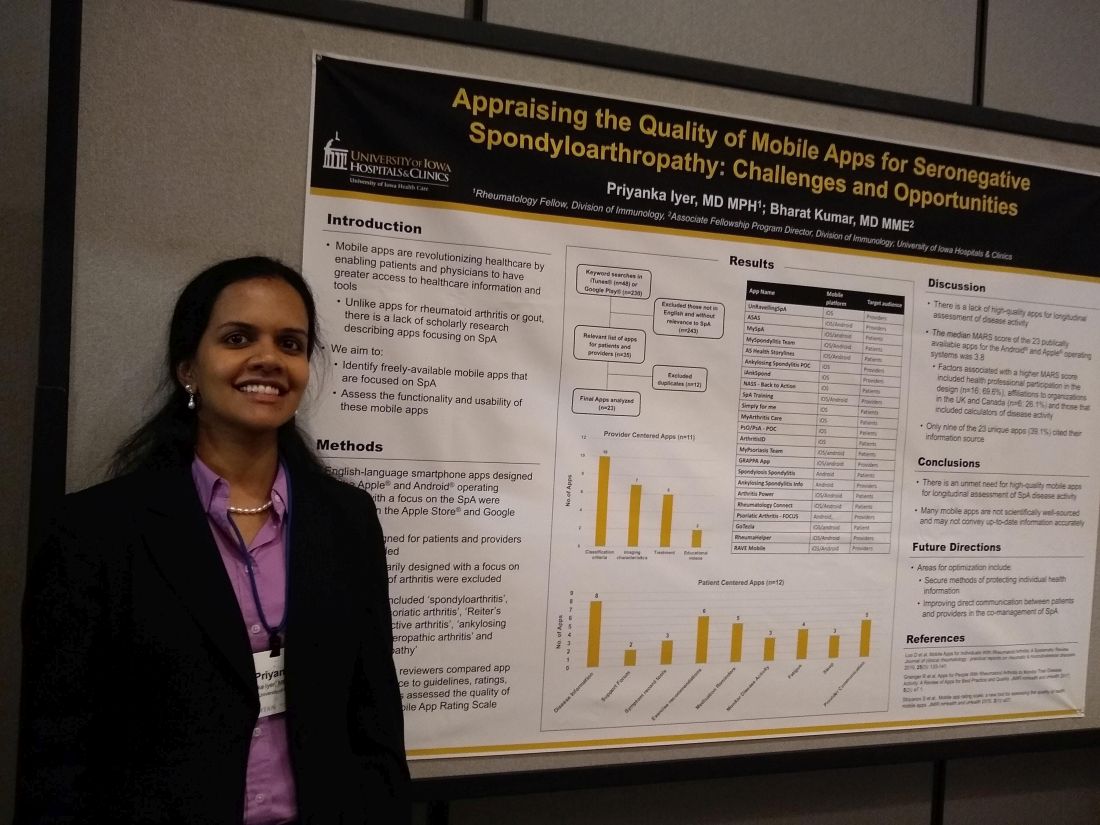
Mobile SpA apps abound, but there’s room for quality improvement
MADISON, WISC. – according to a recent review.
In assessing the 23 publicly available apps aimed at patients or providers, the median score on a common assessment of smartphone apps was just 3.8 on a 5-point scale, said Priyanka Iyer, MBBS, MPH.
Speaking in an interview at the annual meeting of the Spondyloarthritis Research and Treatment Network (SPARTAN), Dr. Iyer pointed out several ways that apps could be optimized. Foremost, she said, is providing secure ways to store and transmit protected health information. Also, apps still haven’t realized their potential to support true comanagement of spondyloarthritis (SpA) via secure, direct patient-provider communication.
“This is an area that we researched previously in rheumatoid arthritis and gout,” explained Dr. Iyer, a rheumatology fellow at the University of Iowa, Iowa City. “We found 23 apps that are available between the Android and iOS platforms; most of them are actually centered towards patients.” In their review, Dr. Iyer and coauthor, Bharat Kumar, MD, had excluded apps that primarily focused on other types of arthritis, using search terms that focused on SpA.
In looking at the 11 provider-centered apps and the 12 that were patient focused, Dr. Iyer and coauthor independently reviewed features of each app. Factors they considered included adherence to guidelines, amount of correct medical information provided, and specific features including capacity to store imaging and test results, and ability to host patient-provider communication.
Of the provider-centered apps, 10 contained appropriate classification criteria, and 7 also contained medical imaging characteristics of the target conditions. Six apps guided providers through treatment options, and two had educational videos.
Of the 12 patient-centered apps, 8 provided disease information, and 6 gave exercise recommendations. Five of the apps had prompts that reminded patients to take medication, and three had tools to help patients record and track symptoms. Similarly, three apps had features to help patients monitor disease activity. Two of the apps were primarily access points for a patient support forum.
Additionally, each app was evaluated by each reviewer using the Mobile App Rating Scale (MARS), said Dr. Iyer. “The overall rating was pretty low, at 3.8 [of a possible 5.0]. Factors that increased the MARS scores included affiliations to organizations in the United Kingdom and Canada; for patients who use these apps, their information is automatically transmitted to their providers, and they are able to also access imaging and most of their other health care information on the app.”
Another factor associated with a higher MARS score was design that included health professional participation, which was the case for 16 apps (69.6%). Apps that included calculators of disease activity were also more likely to achieve a higher MARS score, Dr. Iyer and coauthor wrote.
Notably, just 9 of 23 apps (39.1%) included citations referencing their source for medical information.
“I think future areas for improvement and for development of apps include securing individual health information to allow direct communication between patients and providers,” Dr. Iyer said. “I hope that some patients use these apps to learn, and to help their self-management improve.”
“There is an unmet need for high-quality mobile apps for longitudinal assessment of SpA disease activity,” Dr. Iyer and colleagues wrote in the poster accompanying the presentation. “Many mobile apps are not scientifically well sourced and may not convey up-to-date information accurately.”
The authors reported no conflicts of interest and no outside sources of funding.
SOURCE: Iyer P et al. SPARTAN 2019.
MADISON, WISC. – according to a recent review.
In assessing the 23 publicly available apps aimed at patients or providers, the median score on a common assessment of smartphone apps was just 3.8 on a 5-point scale, said Priyanka Iyer, MBBS, MPH.
Speaking in an interview at the annual meeting of the Spondyloarthritis Research and Treatment Network (SPARTAN), Dr. Iyer pointed out several ways that apps could be optimized. Foremost, she said, is providing secure ways to store and transmit protected health information. Also, apps still haven’t realized their potential to support true comanagement of spondyloarthritis (SpA) via secure, direct patient-provider communication.
“This is an area that we researched previously in rheumatoid arthritis and gout,” explained Dr. Iyer, a rheumatology fellow at the University of Iowa, Iowa City. “We found 23 apps that are available between the Android and iOS platforms; most of them are actually centered towards patients.” In their review, Dr. Iyer and coauthor, Bharat Kumar, MD, had excluded apps that primarily focused on other types of arthritis, using search terms that focused on SpA.
In looking at the 11 provider-centered apps and the 12 that were patient focused, Dr. Iyer and coauthor independently reviewed features of each app. Factors they considered included adherence to guidelines, amount of correct medical information provided, and specific features including capacity to store imaging and test results, and ability to host patient-provider communication.
Of the provider-centered apps, 10 contained appropriate classification criteria, and 7 also contained medical imaging characteristics of the target conditions. Six apps guided providers through treatment options, and two had educational videos.
Of the 12 patient-centered apps, 8 provided disease information, and 6 gave exercise recommendations. Five of the apps had prompts that reminded patients to take medication, and three had tools to help patients record and track symptoms. Similarly, three apps had features to help patients monitor disease activity. Two of the apps were primarily access points for a patient support forum.
Additionally, each app was evaluated by each reviewer using the Mobile App Rating Scale (MARS), said Dr. Iyer. “The overall rating was pretty low, at 3.8 [of a possible 5.0]. Factors that increased the MARS scores included affiliations to organizations in the United Kingdom and Canada; for patients who use these apps, their information is automatically transmitted to their providers, and they are able to also access imaging and most of their other health care information on the app.”
Another factor associated with a higher MARS score was design that included health professional participation, which was the case for 16 apps (69.6%). Apps that included calculators of disease activity were also more likely to achieve a higher MARS score, Dr. Iyer and coauthor wrote.
Notably, just 9 of 23 apps (39.1%) included citations referencing their source for medical information.
“I think future areas for improvement and for development of apps include securing individual health information to allow direct communication between patients and providers,” Dr. Iyer said. “I hope that some patients use these apps to learn, and to help their self-management improve.”
“There is an unmet need for high-quality mobile apps for longitudinal assessment of SpA disease activity,” Dr. Iyer and colleagues wrote in the poster accompanying the presentation. “Many mobile apps are not scientifically well sourced and may not convey up-to-date information accurately.”
The authors reported no conflicts of interest and no outside sources of funding.
SOURCE: Iyer P et al. SPARTAN 2019.
MADISON, WISC. – according to a recent review.
In assessing the 23 publicly available apps aimed at patients or providers, the median score on a common assessment of smartphone apps was just 3.8 on a 5-point scale, said Priyanka Iyer, MBBS, MPH.
Speaking in an interview at the annual meeting of the Spondyloarthritis Research and Treatment Network (SPARTAN), Dr. Iyer pointed out several ways that apps could be optimized. Foremost, she said, is providing secure ways to store and transmit protected health information. Also, apps still haven’t realized their potential to support true comanagement of spondyloarthritis (SpA) via secure, direct patient-provider communication.
“This is an area that we researched previously in rheumatoid arthritis and gout,” explained Dr. Iyer, a rheumatology fellow at the University of Iowa, Iowa City. “We found 23 apps that are available between the Android and iOS platforms; most of them are actually centered towards patients.” In their review, Dr. Iyer and coauthor, Bharat Kumar, MD, had excluded apps that primarily focused on other types of arthritis, using search terms that focused on SpA.
In looking at the 11 provider-centered apps and the 12 that were patient focused, Dr. Iyer and coauthor independently reviewed features of each app. Factors they considered included adherence to guidelines, amount of correct medical information provided, and specific features including capacity to store imaging and test results, and ability to host patient-provider communication.
Of the provider-centered apps, 10 contained appropriate classification criteria, and 7 also contained medical imaging characteristics of the target conditions. Six apps guided providers through treatment options, and two had educational videos.
Of the 12 patient-centered apps, 8 provided disease information, and 6 gave exercise recommendations. Five of the apps had prompts that reminded patients to take medication, and three had tools to help patients record and track symptoms. Similarly, three apps had features to help patients monitor disease activity. Two of the apps were primarily access points for a patient support forum.
Additionally, each app was evaluated by each reviewer using the Mobile App Rating Scale (MARS), said Dr. Iyer. “The overall rating was pretty low, at 3.8 [of a possible 5.0]. Factors that increased the MARS scores included affiliations to organizations in the United Kingdom and Canada; for patients who use these apps, their information is automatically transmitted to their providers, and they are able to also access imaging and most of their other health care information on the app.”
Another factor associated with a higher MARS score was design that included health professional participation, which was the case for 16 apps (69.6%). Apps that included calculators of disease activity were also more likely to achieve a higher MARS score, Dr. Iyer and coauthor wrote.
Notably, just 9 of 23 apps (39.1%) included citations referencing their source for medical information.
“I think future areas for improvement and for development of apps include securing individual health information to allow direct communication between patients and providers,” Dr. Iyer said. “I hope that some patients use these apps to learn, and to help their self-management improve.”
“There is an unmet need for high-quality mobile apps for longitudinal assessment of SpA disease activity,” Dr. Iyer and colleagues wrote in the poster accompanying the presentation. “Many mobile apps are not scientifically well sourced and may not convey up-to-date information accurately.”
The authors reported no conflicts of interest and no outside sources of funding.
SOURCE: Iyer P et al. SPARTAN 2019.
REPORTING FROM SPARTAN 2019
Outpatient program successfully tackles substance use and chronic pain
MILWAUKEE – An interdisciplinary intensive outpatient treatment program addressing chronic pain and substance use disorder effectively addressed both diagnoses in a military population.
Intensive outpatient programs (IOPs) frequently address these conditions within a biopsychosocial format, but it’s not common for IOPs to have this dual focus on chronic pain and substance use disorder (SUD), said Michael Stockin, MD, speaking in an interview at the scientific meeting of the American Pain Society.
Dr. Stockin said he and his collaborators recognized that, especially among a military population, the two conditions have considerable overlap, so it made sense to integrate behavioral treatment for both conditions in an intensive outpatient program. “Our hypothesis was that if you can use an intensive outpatient program to address substance use disorder, maybe you can actually add a chronic pain curriculum – like a functional restoration program to it.
“As a result of our study, we did find that there were significant differences in worst pain scores as a result of the program. In the people who took both the substance use disorder and chronic pain curriculum, we found significant reductions in total impairment, worst pain, and they also had less … substance use as well,” said Dr. Stockin.
In a quality improvement project, Dr. Stockin and collaborators compared short-term outcomes for patients who received IOP treatment addressing both chronic pain and SUD with those receiving SUD-only IOP.
For those participating in the joint IOP, scores indicating worst pain on the 0-10 numeric rating scale were reduced significantly, from 7.55 to 6.23 (P = .013). Scores on a functional measure of impairment, the Pain Outcomes Questionnaire Short Form (POQ-SF) also dropped significantly, from 84.92 to 63.50 (P = .034). The vitality domain of the POQ-SF also showed that patients had less impairment after participation in the joint IOP, with scores in that domain dropping from 20.17 to 17.25 (P = .024).
Looking at the total cohort, patient scores on the Brief Addiction Monitor (BAM) dropped significantly from baseline to the end of the intervention, indicating reduced substance use (P = .041). Mean scores for participants in the joint IOP were higher at baseline than for those in the SUD-only IOP (1.000 vs. 0.565). However, those participating in the joint IOP had lower mean postintervention BAM scores than the SUD-only cohort (0.071 vs. 0.174).
American veterans experience more severe pain and have a higher prevalence of chronic pain than nonveterans. Similarly, wrote Dr. Stockin, a chronic pain fellow in pain management at Walter Reed National Military Medical Center, Bethesda, Md., and colleagues in the poster presentation.
The project enrolled a total of 66 patients (10 female and 56 male). Of these, 18 participated in the joint SUD–chronic pain program, and 48 received usual treatment of the SUD-only IOP treatment. The mean overall age was 33.2 years, and 71.2% of participants were white.
Overall, 51 patients (77.3%) of participants had alcohol use disorder. Participants included active duty service members, veterans, and their dependents. Opioid and cannabis use disorders were experienced by a total of eight patients, and seven more patients had diagnoses of alcohol use disorder along with other substance use disorders.
All patients completed the BAM and received urine toxicology and alcohol breath testing at enrollment; drug and alcohol screening was completed at other points during the IOP treatment for both groups as well.
The joint IOP ran 3 full days a week, with a substance use curriculum in the morning and a pain management program in the afternoon; the SUD-only participants had three morning sessions weekly. Both interventions lasted 6 weeks, and Dr. Stockin said he and his colleagues would like to acquire longitudinal data to assess the durability of gains seen from the joint IOP.
The multidisciplinary team running the joint IOP was made up of an addiction/pain medicine physician, a clinical health psychologist, a physical therapist, social workers, and a nurse.
“This project is the first of its kind to find a significant reduction in pain burden while concurrently treating addiction and pain in an outpatient military health care setting,” Dr. Stockin and colleagues wrote in the poster accompanying the presentation.
“We had outcomes in both substance use and chronic pain that were positive, so it suggests that in the military health system, people may actually benefit from treating both chronic pain and substance use disorder concurrently. If you could harmonize those programs, you might be able to get good outcomes for soldiers and their families,” Dr. Stockin said.
Dr. Stockin reported no conflicts of interest. The project was funded by the Defense Health Agency.
MILWAUKEE – An interdisciplinary intensive outpatient treatment program addressing chronic pain and substance use disorder effectively addressed both diagnoses in a military population.
Intensive outpatient programs (IOPs) frequently address these conditions within a biopsychosocial format, but it’s not common for IOPs to have this dual focus on chronic pain and substance use disorder (SUD), said Michael Stockin, MD, speaking in an interview at the scientific meeting of the American Pain Society.
Dr. Stockin said he and his collaborators recognized that, especially among a military population, the two conditions have considerable overlap, so it made sense to integrate behavioral treatment for both conditions in an intensive outpatient program. “Our hypothesis was that if you can use an intensive outpatient program to address substance use disorder, maybe you can actually add a chronic pain curriculum – like a functional restoration program to it.
“As a result of our study, we did find that there were significant differences in worst pain scores as a result of the program. In the people who took both the substance use disorder and chronic pain curriculum, we found significant reductions in total impairment, worst pain, and they also had less … substance use as well,” said Dr. Stockin.
In a quality improvement project, Dr. Stockin and collaborators compared short-term outcomes for patients who received IOP treatment addressing both chronic pain and SUD with those receiving SUD-only IOP.
For those participating in the joint IOP, scores indicating worst pain on the 0-10 numeric rating scale were reduced significantly, from 7.55 to 6.23 (P = .013). Scores on a functional measure of impairment, the Pain Outcomes Questionnaire Short Form (POQ-SF) also dropped significantly, from 84.92 to 63.50 (P = .034). The vitality domain of the POQ-SF also showed that patients had less impairment after participation in the joint IOP, with scores in that domain dropping from 20.17 to 17.25 (P = .024).
Looking at the total cohort, patient scores on the Brief Addiction Monitor (BAM) dropped significantly from baseline to the end of the intervention, indicating reduced substance use (P = .041). Mean scores for participants in the joint IOP were higher at baseline than for those in the SUD-only IOP (1.000 vs. 0.565). However, those participating in the joint IOP had lower mean postintervention BAM scores than the SUD-only cohort (0.071 vs. 0.174).
American veterans experience more severe pain and have a higher prevalence of chronic pain than nonveterans. Similarly, wrote Dr. Stockin, a chronic pain fellow in pain management at Walter Reed National Military Medical Center, Bethesda, Md., and colleagues in the poster presentation.
The project enrolled a total of 66 patients (10 female and 56 male). Of these, 18 participated in the joint SUD–chronic pain program, and 48 received usual treatment of the SUD-only IOP treatment. The mean overall age was 33.2 years, and 71.2% of participants were white.
Overall, 51 patients (77.3%) of participants had alcohol use disorder. Participants included active duty service members, veterans, and their dependents. Opioid and cannabis use disorders were experienced by a total of eight patients, and seven more patients had diagnoses of alcohol use disorder along with other substance use disorders.
All patients completed the BAM and received urine toxicology and alcohol breath testing at enrollment; drug and alcohol screening was completed at other points during the IOP treatment for both groups as well.
The joint IOP ran 3 full days a week, with a substance use curriculum in the morning and a pain management program in the afternoon; the SUD-only participants had three morning sessions weekly. Both interventions lasted 6 weeks, and Dr. Stockin said he and his colleagues would like to acquire longitudinal data to assess the durability of gains seen from the joint IOP.
The multidisciplinary team running the joint IOP was made up of an addiction/pain medicine physician, a clinical health psychologist, a physical therapist, social workers, and a nurse.
“This project is the first of its kind to find a significant reduction in pain burden while concurrently treating addiction and pain in an outpatient military health care setting,” Dr. Stockin and colleagues wrote in the poster accompanying the presentation.
“We had outcomes in both substance use and chronic pain that were positive, so it suggests that in the military health system, people may actually benefit from treating both chronic pain and substance use disorder concurrently. If you could harmonize those programs, you might be able to get good outcomes for soldiers and their families,” Dr. Stockin said.
Dr. Stockin reported no conflicts of interest. The project was funded by the Defense Health Agency.
MILWAUKEE – An interdisciplinary intensive outpatient treatment program addressing chronic pain and substance use disorder effectively addressed both diagnoses in a military population.
Intensive outpatient programs (IOPs) frequently address these conditions within a biopsychosocial format, but it’s not common for IOPs to have this dual focus on chronic pain and substance use disorder (SUD), said Michael Stockin, MD, speaking in an interview at the scientific meeting of the American Pain Society.
Dr. Stockin said he and his collaborators recognized that, especially among a military population, the two conditions have considerable overlap, so it made sense to integrate behavioral treatment for both conditions in an intensive outpatient program. “Our hypothesis was that if you can use an intensive outpatient program to address substance use disorder, maybe you can actually add a chronic pain curriculum – like a functional restoration program to it.
“As a result of our study, we did find that there were significant differences in worst pain scores as a result of the program. In the people who took both the substance use disorder and chronic pain curriculum, we found significant reductions in total impairment, worst pain, and they also had less … substance use as well,” said Dr. Stockin.
In a quality improvement project, Dr. Stockin and collaborators compared short-term outcomes for patients who received IOP treatment addressing both chronic pain and SUD with those receiving SUD-only IOP.
For those participating in the joint IOP, scores indicating worst pain on the 0-10 numeric rating scale were reduced significantly, from 7.55 to 6.23 (P = .013). Scores on a functional measure of impairment, the Pain Outcomes Questionnaire Short Form (POQ-SF) also dropped significantly, from 84.92 to 63.50 (P = .034). The vitality domain of the POQ-SF also showed that patients had less impairment after participation in the joint IOP, with scores in that domain dropping from 20.17 to 17.25 (P = .024).
Looking at the total cohort, patient scores on the Brief Addiction Monitor (BAM) dropped significantly from baseline to the end of the intervention, indicating reduced substance use (P = .041). Mean scores for participants in the joint IOP were higher at baseline than for those in the SUD-only IOP (1.000 vs. 0.565). However, those participating in the joint IOP had lower mean postintervention BAM scores than the SUD-only cohort (0.071 vs. 0.174).
American veterans experience more severe pain and have a higher prevalence of chronic pain than nonveterans. Similarly, wrote Dr. Stockin, a chronic pain fellow in pain management at Walter Reed National Military Medical Center, Bethesda, Md., and colleagues in the poster presentation.
The project enrolled a total of 66 patients (10 female and 56 male). Of these, 18 participated in the joint SUD–chronic pain program, and 48 received usual treatment of the SUD-only IOP treatment. The mean overall age was 33.2 years, and 71.2% of participants were white.
Overall, 51 patients (77.3%) of participants had alcohol use disorder. Participants included active duty service members, veterans, and their dependents. Opioid and cannabis use disorders were experienced by a total of eight patients, and seven more patients had diagnoses of alcohol use disorder along with other substance use disorders.
All patients completed the BAM and received urine toxicology and alcohol breath testing at enrollment; drug and alcohol screening was completed at other points during the IOP treatment for both groups as well.
The joint IOP ran 3 full days a week, with a substance use curriculum in the morning and a pain management program in the afternoon; the SUD-only participants had three morning sessions weekly. Both interventions lasted 6 weeks, and Dr. Stockin said he and his colleagues would like to acquire longitudinal data to assess the durability of gains seen from the joint IOP.
The multidisciplinary team running the joint IOP was made up of an addiction/pain medicine physician, a clinical health psychologist, a physical therapist, social workers, and a nurse.
“This project is the first of its kind to find a significant reduction in pain burden while concurrently treating addiction and pain in an outpatient military health care setting,” Dr. Stockin and colleagues wrote in the poster accompanying the presentation.
“We had outcomes in both substance use and chronic pain that were positive, so it suggests that in the military health system, people may actually benefit from treating both chronic pain and substance use disorder concurrently. If you could harmonize those programs, you might be able to get good outcomes for soldiers and their families,” Dr. Stockin said.
Dr. Stockin reported no conflicts of interest. The project was funded by the Defense Health Agency.
REPORTING FROM APS 2019
Key clinical point: An intensive, 6-week joint substance use disorder and chronic pain intensive outpatient program significantly reduced both substance use and pain.
Major finding: Patients had less pain and reduced substance use after completing the program, compared with baseline (P = .013 and .041, respectively).
Study details: A quality improvement project including 66 patients at a military health facility.
Disclosures: The study was sponsored by the Defense Health Agency. Dr. Stockin reported no conflicts of interest.
In chronic pain, catastrophizing contributes to disrupted brain circuitry
MILWAUKEE – When a patient with acute pain tumbles into a chronic pain state, many factors are at play, according to the widely accepted biopsychosocial theory of pain. Emotional, cognitive, and environmental components all contribute to the persistent and recalcitrant symptoms chronic pain patients experience.
Now, modern neuroimaging techniques show how for some, pain signals hijack the brain’s regulatory networks, allowing rumination and catastrophizing to intrude on the exteroception that’s critical to how humans interact with one another and the world. Interrupting catastrophizing with nonpharmacologic techniques yields measurable improvements – and there’s promise that a single treatment session can make a lasting difference.
“Psychosocial phenotypes, such as catastrophizing, are part of a complex biopsychosocial web of contributors to chronic pain. ,” said Robert R. Edwards, PhD, a psychologist at Brigham and Women’s Hospital/Harvard Medical School (Boston) Pain Management Center. Dr. Edwards moderated a session focused on catastrophizing at the scientific meeting of the American Pain Society.
Through magnetic resonance imaging techniques that measure functional connectivity, researchers can now see how nodes in the brain form connected networks that are differentially activated.
For example, the brain’s salience network (SLN) responds to stimuli that merit attention, such as evoked or clinical pain, Vitaly Napadow, PhD, said during his presentation. Key nodes in the SLN include the anterior cingulate cortex, the anterior insula, and the anterior temporoparietal junction. One function of the salience network, he said, is to regulate switching between the default mode network (DMN) – an interoceptive network – and the central executive network, usually active in exteroceptive tasks.
“The default mode network has been found to play an important role in pain processing,” Dr. Napadow said. These brain regions are more active in self-referential cognition – thinking about oneself – than when performing external tasks, he said. Consistently, studies have found decreased DMN deactivation in patients with chronic pain; essentially, the constant low hum of pain-focused DMN activity never turns off in a chronic pain state.
For patients with chronic pain, high levels of catastrophizing mean greater impact on functional brain connectivity, said Dr. Napadow, director of the Center for Integrative Pain NeuroImaging at the Martino Center for Biomedical Imaging at Massachusetts General Hospital and Harvard Medical School, Boston.
Looking at patients with chronic low back pain, he and his research team looked for connections between the DMN and the insula, which has a central role in pain processing. This connectivity was increased only in patients with high catastrophizing scores, said Dr. Napadow, with increased DMN-insula connectivity associated with increased pain scores only for this subgroup (Pain. 2019 Mar 4. doi: 10.1097/j.pain.0000000000001541).
“The model that we’re moving toward is that chronic pain leads to a blurring in the canonical network” of brain connectivity, Dr. Napadow said. “The speculation here is that the DMN-SLN linkage could be a sort of neural substrate for a common perception that chronic pain patients have – that their pain becomes part of who they are. Their interoceptive state becomes linked to the pain they are feeling: They are their pain.”
Where to turn with this information, which has large clinical implications? “Catastrophizing is a consistent risk factor for poor pain treatment outcomes, especially when we’re talking about pharmacologic treatments,” Dr. Edwards said. Also, chronic pain patients with the highest catastrophizing scores have the most opioid-related side effects, he said.
“Cognitive-behavioral therapy is potentially the most effective at reducing this risk factor,” said Dr. Edwards, noting that long-term effects were seen at 6 and 12 months post treatment. “These are significant, moderate-sized effects; there is some evidence that effects are largest in those with the highest baseline pain catastrophizing scores.”
“CBT is considered the gold standard, mainly because it’s the best studied” among treatment modalities, psychologist Beth Darnall, PhD, pointed out in her presentation. There’s evidence that other nonpharmacologic interventions can reduce catastrophizing: Psychology-informed yoga practices, physical therapy, and certain medical devices, such as high-frequency transcutaneous electric nerve stimulation units, may all have efficacy against catastrophizing and the downward spiral of chronic pain.
Still, a randomized controlled trial of CBT for pain in patients with fibromyalgia showed that the benefit, measured as reduction in pain interference with daily functioning, was almost twice as high in the high-catastrophizing group, “suggesting the potential utility of this method for patients at greatest risk,” said Dr. Edwards.
“We see a specific pattern of alterations in chronic pain similar to that seen in anxiety disorder; this suggests that some individuals are primed for the experience of pain,” said Dr. Darnall, clinical professor of anesthesiology, perioperative medicine, and pain medicine at Stanford (Calif.) University. “We are not born with the understanding of how to modulate pain and the distress it causes us.”
When she talks to patients, Dr. Darnall said: “I describe pain as being our ‘harm alarm.’ ... I like to describe it to people that ‘you have a very protective nervous system.’ ”
Dr. Darnall and her colleagues reported success with a pilot study of a single 2.5-hour-long session that addressed pain catastrophizing. From a baseline score of 26.1 on the Pain Catastrophizing Scale to a score of 13.8 at week 4, the 57 participants saw a significant decrease in mean scores on the scale (d [effect size] = 1.15).
On the strength of these early findings, Dr. Darnall and her collaborators are embarking on a randomized controlled trial ; the 3-arm comparative effectiveness study will compare a single-session intervention against 8 weeks of CBT or education-only classes for individuals with catastrophizing and chronic pain. The trial is structured to test the hypothesis that the single-session intervention will be noninferior to the full 8 weeks of CBT, Dr. Darnall said.
Building on the importance of avoiding stigmatizing and pejorative terms when talking about pain and catastrophizing, Dr. Darnall said she’s moved away from using the term “catastrophizing” in patient interactions. The one-session intervention is called “Empowered Relief – Train Your Brain Away from Pain.”
There’s a practical promise to a single-session class: Dr. Darnall has taught up to 85 patients at once, she said, adding, “This is a low-cost and scalable intervention.”
Dr. Edwards and Dr. Napadow reported funding from the National Institutes of Health, and they reported no conflicts of interest. Dr. Darnall reported funding from the NIH and the Patient-Centered Outcomes Research Institute. She serves on the scientific advisory board of Axial Healthcare and has several commercial publications about pain.
MILWAUKEE – When a patient with acute pain tumbles into a chronic pain state, many factors are at play, according to the widely accepted biopsychosocial theory of pain. Emotional, cognitive, and environmental components all contribute to the persistent and recalcitrant symptoms chronic pain patients experience.
Now, modern neuroimaging techniques show how for some, pain signals hijack the brain’s regulatory networks, allowing rumination and catastrophizing to intrude on the exteroception that’s critical to how humans interact with one another and the world. Interrupting catastrophizing with nonpharmacologic techniques yields measurable improvements – and there’s promise that a single treatment session can make a lasting difference.
“Psychosocial phenotypes, such as catastrophizing, are part of a complex biopsychosocial web of contributors to chronic pain. ,” said Robert R. Edwards, PhD, a psychologist at Brigham and Women’s Hospital/Harvard Medical School (Boston) Pain Management Center. Dr. Edwards moderated a session focused on catastrophizing at the scientific meeting of the American Pain Society.
Through magnetic resonance imaging techniques that measure functional connectivity, researchers can now see how nodes in the brain form connected networks that are differentially activated.
For example, the brain’s salience network (SLN) responds to stimuli that merit attention, such as evoked or clinical pain, Vitaly Napadow, PhD, said during his presentation. Key nodes in the SLN include the anterior cingulate cortex, the anterior insula, and the anterior temporoparietal junction. One function of the salience network, he said, is to regulate switching between the default mode network (DMN) – an interoceptive network – and the central executive network, usually active in exteroceptive tasks.
“The default mode network has been found to play an important role in pain processing,” Dr. Napadow said. These brain regions are more active in self-referential cognition – thinking about oneself – than when performing external tasks, he said. Consistently, studies have found decreased DMN deactivation in patients with chronic pain; essentially, the constant low hum of pain-focused DMN activity never turns off in a chronic pain state.
For patients with chronic pain, high levels of catastrophizing mean greater impact on functional brain connectivity, said Dr. Napadow, director of the Center for Integrative Pain NeuroImaging at the Martino Center for Biomedical Imaging at Massachusetts General Hospital and Harvard Medical School, Boston.
Looking at patients with chronic low back pain, he and his research team looked for connections between the DMN and the insula, which has a central role in pain processing. This connectivity was increased only in patients with high catastrophizing scores, said Dr. Napadow, with increased DMN-insula connectivity associated with increased pain scores only for this subgroup (Pain. 2019 Mar 4. doi: 10.1097/j.pain.0000000000001541).
“The model that we’re moving toward is that chronic pain leads to a blurring in the canonical network” of brain connectivity, Dr. Napadow said. “The speculation here is that the DMN-SLN linkage could be a sort of neural substrate for a common perception that chronic pain patients have – that their pain becomes part of who they are. Their interoceptive state becomes linked to the pain they are feeling: They are their pain.”
Where to turn with this information, which has large clinical implications? “Catastrophizing is a consistent risk factor for poor pain treatment outcomes, especially when we’re talking about pharmacologic treatments,” Dr. Edwards said. Also, chronic pain patients with the highest catastrophizing scores have the most opioid-related side effects, he said.
“Cognitive-behavioral therapy is potentially the most effective at reducing this risk factor,” said Dr. Edwards, noting that long-term effects were seen at 6 and 12 months post treatment. “These are significant, moderate-sized effects; there is some evidence that effects are largest in those with the highest baseline pain catastrophizing scores.”
“CBT is considered the gold standard, mainly because it’s the best studied” among treatment modalities, psychologist Beth Darnall, PhD, pointed out in her presentation. There’s evidence that other nonpharmacologic interventions can reduce catastrophizing: Psychology-informed yoga practices, physical therapy, and certain medical devices, such as high-frequency transcutaneous electric nerve stimulation units, may all have efficacy against catastrophizing and the downward spiral of chronic pain.
Still, a randomized controlled trial of CBT for pain in patients with fibromyalgia showed that the benefit, measured as reduction in pain interference with daily functioning, was almost twice as high in the high-catastrophizing group, “suggesting the potential utility of this method for patients at greatest risk,” said Dr. Edwards.
“We see a specific pattern of alterations in chronic pain similar to that seen in anxiety disorder; this suggests that some individuals are primed for the experience of pain,” said Dr. Darnall, clinical professor of anesthesiology, perioperative medicine, and pain medicine at Stanford (Calif.) University. “We are not born with the understanding of how to modulate pain and the distress it causes us.”
When she talks to patients, Dr. Darnall said: “I describe pain as being our ‘harm alarm.’ ... I like to describe it to people that ‘you have a very protective nervous system.’ ”
Dr. Darnall and her colleagues reported success with a pilot study of a single 2.5-hour-long session that addressed pain catastrophizing. From a baseline score of 26.1 on the Pain Catastrophizing Scale to a score of 13.8 at week 4, the 57 participants saw a significant decrease in mean scores on the scale (d [effect size] = 1.15).
On the strength of these early findings, Dr. Darnall and her collaborators are embarking on a randomized controlled trial ; the 3-arm comparative effectiveness study will compare a single-session intervention against 8 weeks of CBT or education-only classes for individuals with catastrophizing and chronic pain. The trial is structured to test the hypothesis that the single-session intervention will be noninferior to the full 8 weeks of CBT, Dr. Darnall said.
Building on the importance of avoiding stigmatizing and pejorative terms when talking about pain and catastrophizing, Dr. Darnall said she’s moved away from using the term “catastrophizing” in patient interactions. The one-session intervention is called “Empowered Relief – Train Your Brain Away from Pain.”
There’s a practical promise to a single-session class: Dr. Darnall has taught up to 85 patients at once, she said, adding, “This is a low-cost and scalable intervention.”
Dr. Edwards and Dr. Napadow reported funding from the National Institutes of Health, and they reported no conflicts of interest. Dr. Darnall reported funding from the NIH and the Patient-Centered Outcomes Research Institute. She serves on the scientific advisory board of Axial Healthcare and has several commercial publications about pain.
MILWAUKEE – When a patient with acute pain tumbles into a chronic pain state, many factors are at play, according to the widely accepted biopsychosocial theory of pain. Emotional, cognitive, and environmental components all contribute to the persistent and recalcitrant symptoms chronic pain patients experience.
Now, modern neuroimaging techniques show how for some, pain signals hijack the brain’s regulatory networks, allowing rumination and catastrophizing to intrude on the exteroception that’s critical to how humans interact with one another and the world. Interrupting catastrophizing with nonpharmacologic techniques yields measurable improvements – and there’s promise that a single treatment session can make a lasting difference.
“Psychosocial phenotypes, such as catastrophizing, are part of a complex biopsychosocial web of contributors to chronic pain. ,” said Robert R. Edwards, PhD, a psychologist at Brigham and Women’s Hospital/Harvard Medical School (Boston) Pain Management Center. Dr. Edwards moderated a session focused on catastrophizing at the scientific meeting of the American Pain Society.
Through magnetic resonance imaging techniques that measure functional connectivity, researchers can now see how nodes in the brain form connected networks that are differentially activated.
For example, the brain’s salience network (SLN) responds to stimuli that merit attention, such as evoked or clinical pain, Vitaly Napadow, PhD, said during his presentation. Key nodes in the SLN include the anterior cingulate cortex, the anterior insula, and the anterior temporoparietal junction. One function of the salience network, he said, is to regulate switching between the default mode network (DMN) – an interoceptive network – and the central executive network, usually active in exteroceptive tasks.
“The default mode network has been found to play an important role in pain processing,” Dr. Napadow said. These brain regions are more active in self-referential cognition – thinking about oneself – than when performing external tasks, he said. Consistently, studies have found decreased DMN deactivation in patients with chronic pain; essentially, the constant low hum of pain-focused DMN activity never turns off in a chronic pain state.
For patients with chronic pain, high levels of catastrophizing mean greater impact on functional brain connectivity, said Dr. Napadow, director of the Center for Integrative Pain NeuroImaging at the Martino Center for Biomedical Imaging at Massachusetts General Hospital and Harvard Medical School, Boston.
Looking at patients with chronic low back pain, he and his research team looked for connections between the DMN and the insula, which has a central role in pain processing. This connectivity was increased only in patients with high catastrophizing scores, said Dr. Napadow, with increased DMN-insula connectivity associated with increased pain scores only for this subgroup (Pain. 2019 Mar 4. doi: 10.1097/j.pain.0000000000001541).
“The model that we’re moving toward is that chronic pain leads to a blurring in the canonical network” of brain connectivity, Dr. Napadow said. “The speculation here is that the DMN-SLN linkage could be a sort of neural substrate for a common perception that chronic pain patients have – that their pain becomes part of who they are. Their interoceptive state becomes linked to the pain they are feeling: They are their pain.”
Where to turn with this information, which has large clinical implications? “Catastrophizing is a consistent risk factor for poor pain treatment outcomes, especially when we’re talking about pharmacologic treatments,” Dr. Edwards said. Also, chronic pain patients with the highest catastrophizing scores have the most opioid-related side effects, he said.
“Cognitive-behavioral therapy is potentially the most effective at reducing this risk factor,” said Dr. Edwards, noting that long-term effects were seen at 6 and 12 months post treatment. “These are significant, moderate-sized effects; there is some evidence that effects are largest in those with the highest baseline pain catastrophizing scores.”
“CBT is considered the gold standard, mainly because it’s the best studied” among treatment modalities, psychologist Beth Darnall, PhD, pointed out in her presentation. There’s evidence that other nonpharmacologic interventions can reduce catastrophizing: Psychology-informed yoga practices, physical therapy, and certain medical devices, such as high-frequency transcutaneous electric nerve stimulation units, may all have efficacy against catastrophizing and the downward spiral of chronic pain.
Still, a randomized controlled trial of CBT for pain in patients with fibromyalgia showed that the benefit, measured as reduction in pain interference with daily functioning, was almost twice as high in the high-catastrophizing group, “suggesting the potential utility of this method for patients at greatest risk,” said Dr. Edwards.
“We see a specific pattern of alterations in chronic pain similar to that seen in anxiety disorder; this suggests that some individuals are primed for the experience of pain,” said Dr. Darnall, clinical professor of anesthesiology, perioperative medicine, and pain medicine at Stanford (Calif.) University. “We are not born with the understanding of how to modulate pain and the distress it causes us.”
When she talks to patients, Dr. Darnall said: “I describe pain as being our ‘harm alarm.’ ... I like to describe it to people that ‘you have a very protective nervous system.’ ”
Dr. Darnall and her colleagues reported success with a pilot study of a single 2.5-hour-long session that addressed pain catastrophizing. From a baseline score of 26.1 on the Pain Catastrophizing Scale to a score of 13.8 at week 4, the 57 participants saw a significant decrease in mean scores on the scale (d [effect size] = 1.15).
On the strength of these early findings, Dr. Darnall and her collaborators are embarking on a randomized controlled trial ; the 3-arm comparative effectiveness study will compare a single-session intervention against 8 weeks of CBT or education-only classes for individuals with catastrophizing and chronic pain. The trial is structured to test the hypothesis that the single-session intervention will be noninferior to the full 8 weeks of CBT, Dr. Darnall said.
Building on the importance of avoiding stigmatizing and pejorative terms when talking about pain and catastrophizing, Dr. Darnall said she’s moved away from using the term “catastrophizing” in patient interactions. The one-session intervention is called “Empowered Relief – Train Your Brain Away from Pain.”
There’s a practical promise to a single-session class: Dr. Darnall has taught up to 85 patients at once, she said, adding, “This is a low-cost and scalable intervention.”
Dr. Edwards and Dr. Napadow reported funding from the National Institutes of Health, and they reported no conflicts of interest. Dr. Darnall reported funding from the NIH and the Patient-Centered Outcomes Research Institute. She serves on the scientific advisory board of Axial Healthcare and has several commercial publications about pain.
REPORTING FROM APS 2019
In pain treatment, racial bias common among physician trainees
MILWAUKEE – More than 40% of white physician trainees demonstrated racial bias in medical decision making about treatment of low back pain, as did 31% of nonwhite trainees. However, just 6% of white residents and fellows, and 10% of the nonwhite residents and fellows, reported that patient race had factored into their treatment decisions in a virtual patient task.
The 444 medical residents and fellows who participated viewed video vignettes presenting 12 virtual patients who presented with low back pain, wrote Alexis Grant of Indiana University–Purdue University Indianapolis and her colleagues. In a poster presentation at the scientific meeting of the American Pain Society, Ms. Grant, a doctoral student in clinical psychology, and her collaborators explained that participants agreed to view a series of 12 videos of virtual patients.
The videos presented male and female virtual patients who were black or white and who had jobs associated with low or high socioeconomic status (SES). Information in text vignettes accompanying the videos included occupation, pain etiology, physical exam findings, and pain intensity by self-report.
After viewing the videos and reading the vignettes, participating clinicians were asked to use a 0-100 visual analog scale to report their likelihood of referring patients to a pain specialist or to physical therapy and of recommending opioid or nonopioid analgesia.
“Next, they rated the degree to which they considered different sources of patient information when making treatment decision,” Ms. Grant and her coauthors wrote. Statistical analysis “examined the extent to which providers demonstrated statistically reliable treatment differences across patient race and SES.” These findings were compared with how clinicians reported they used patient race and SES in decision making.
Demonstrated race-based decision making occurred for 41% of white and 31% of nonwhite clinicians. About two-thirds of providers (67.3%) were white, and of the remainder, 26.3% were Asian, 4.4% were classified as “other,” and 2.1% were black. The respondents were aged a mean 29.7 years, and were 42.3% female.
In addition, Ms. Grant and her coauthors estimated provider SES by asking about parental SES, dividing respondents into low (less than $38,000), medium ($38,000-$75,000), and high (greater than $75,000) SES categories.
and similar across levels of provider SES, at 41%, 43%, and 38% for low, medium, and high SES residents and fellows, respectively. However, the disconnect between reported and demonstrated bias that was seen with race was not seen with SES bias, with 43%-48% of providers in each SES group reporting that they had factored patient SES into their treatment decision making.
“These results suggest that providers have low awareness of making different pain treatment decisions” for black patients, compared with decision making for white patients, Ms. Grant and her colleagues wrote. “Decision-making awareness did not substantially differ across provider race or SES.” She and her collaborators called for more research into whether raising awareness about demonstrated racial bias in decision making can improve both racial and socioeconomic gaps in pain care.
The authors reported funding from the National Institutes of Health. They reported no conflicts of interest.
MILWAUKEE – More than 40% of white physician trainees demonstrated racial bias in medical decision making about treatment of low back pain, as did 31% of nonwhite trainees. However, just 6% of white residents and fellows, and 10% of the nonwhite residents and fellows, reported that patient race had factored into their treatment decisions in a virtual patient task.
The 444 medical residents and fellows who participated viewed video vignettes presenting 12 virtual patients who presented with low back pain, wrote Alexis Grant of Indiana University–Purdue University Indianapolis and her colleagues. In a poster presentation at the scientific meeting of the American Pain Society, Ms. Grant, a doctoral student in clinical psychology, and her collaborators explained that participants agreed to view a series of 12 videos of virtual patients.
The videos presented male and female virtual patients who were black or white and who had jobs associated with low or high socioeconomic status (SES). Information in text vignettes accompanying the videos included occupation, pain etiology, physical exam findings, and pain intensity by self-report.
After viewing the videos and reading the vignettes, participating clinicians were asked to use a 0-100 visual analog scale to report their likelihood of referring patients to a pain specialist or to physical therapy and of recommending opioid or nonopioid analgesia.
“Next, they rated the degree to which they considered different sources of patient information when making treatment decision,” Ms. Grant and her coauthors wrote. Statistical analysis “examined the extent to which providers demonstrated statistically reliable treatment differences across patient race and SES.” These findings were compared with how clinicians reported they used patient race and SES in decision making.
Demonstrated race-based decision making occurred for 41% of white and 31% of nonwhite clinicians. About two-thirds of providers (67.3%) were white, and of the remainder, 26.3% were Asian, 4.4% were classified as “other,” and 2.1% were black. The respondents were aged a mean 29.7 years, and were 42.3% female.
In addition, Ms. Grant and her coauthors estimated provider SES by asking about parental SES, dividing respondents into low (less than $38,000), medium ($38,000-$75,000), and high (greater than $75,000) SES categories.
and similar across levels of provider SES, at 41%, 43%, and 38% for low, medium, and high SES residents and fellows, respectively. However, the disconnect between reported and demonstrated bias that was seen with race was not seen with SES bias, with 43%-48% of providers in each SES group reporting that they had factored patient SES into their treatment decision making.
“These results suggest that providers have low awareness of making different pain treatment decisions” for black patients, compared with decision making for white patients, Ms. Grant and her colleagues wrote. “Decision-making awareness did not substantially differ across provider race or SES.” She and her collaborators called for more research into whether raising awareness about demonstrated racial bias in decision making can improve both racial and socioeconomic gaps in pain care.
The authors reported funding from the National Institutes of Health. They reported no conflicts of interest.
MILWAUKEE – More than 40% of white physician trainees demonstrated racial bias in medical decision making about treatment of low back pain, as did 31% of nonwhite trainees. However, just 6% of white residents and fellows, and 10% of the nonwhite residents and fellows, reported that patient race had factored into their treatment decisions in a virtual patient task.
The 444 medical residents and fellows who participated viewed video vignettes presenting 12 virtual patients who presented with low back pain, wrote Alexis Grant of Indiana University–Purdue University Indianapolis and her colleagues. In a poster presentation at the scientific meeting of the American Pain Society, Ms. Grant, a doctoral student in clinical psychology, and her collaborators explained that participants agreed to view a series of 12 videos of virtual patients.
The videos presented male and female virtual patients who were black or white and who had jobs associated with low or high socioeconomic status (SES). Information in text vignettes accompanying the videos included occupation, pain etiology, physical exam findings, and pain intensity by self-report.
After viewing the videos and reading the vignettes, participating clinicians were asked to use a 0-100 visual analog scale to report their likelihood of referring patients to a pain specialist or to physical therapy and of recommending opioid or nonopioid analgesia.
“Next, they rated the degree to which they considered different sources of patient information when making treatment decision,” Ms. Grant and her coauthors wrote. Statistical analysis “examined the extent to which providers demonstrated statistically reliable treatment differences across patient race and SES.” These findings were compared with how clinicians reported they used patient race and SES in decision making.
Demonstrated race-based decision making occurred for 41% of white and 31% of nonwhite clinicians. About two-thirds of providers (67.3%) were white, and of the remainder, 26.3% were Asian, 4.4% were classified as “other,” and 2.1% were black. The respondents were aged a mean 29.7 years, and were 42.3% female.
In addition, Ms. Grant and her coauthors estimated provider SES by asking about parental SES, dividing respondents into low (less than $38,000), medium ($38,000-$75,000), and high (greater than $75,000) SES categories.
and similar across levels of provider SES, at 41%, 43%, and 38% for low, medium, and high SES residents and fellows, respectively. However, the disconnect between reported and demonstrated bias that was seen with race was not seen with SES bias, with 43%-48% of providers in each SES group reporting that they had factored patient SES into their treatment decision making.
“These results suggest that providers have low awareness of making different pain treatment decisions” for black patients, compared with decision making for white patients, Ms. Grant and her colleagues wrote. “Decision-making awareness did not substantially differ across provider race or SES.” She and her collaborators called for more research into whether raising awareness about demonstrated racial bias in decision making can improve both racial and socioeconomic gaps in pain care.
The authors reported funding from the National Institutes of Health. They reported no conflicts of interest.
REPORTING FROM APS 2019
Research, clinical practice come together at transgender care symposium
NEW ORLEANS – A well-attended afternoon symposium on transgender medicine gave participants at the annual meeting of the Endocrine Society a solid grounding in transgender care, from prepubescence through adulthood. Here, Joshua Safer, MD, and Michael Irwig, MD, discuss highlights of the symposium, which brought together research, best practices, and clinical practice pearls.
In his presentation, Dr. Safer focused on The basics, he said, begin with presenting well-established, scientific principles supporting current standards of transgender care.
Dr. Safer, executive director of the Mount Sinai Center for Transgender Medicine and Surgery, New York, pointed out the critical role of gonadotropin-releasing hormone antagonists in delaying puberty for transgender girls. Blockade of puberty – and elevated testosterone – can forestall otherwise irreversible male secondary sex characteristics. These include laryngeal enlargement and bony changes of facial structure, for example.
Dr. Irwig, director of andrology at George Washington University, Washington, laid out the basics of transgender hormone therapy, including clinical pearls, such as avoiding ethinyl estradiol because of the heightened risk of venous thromboembolism.
Dr. Safer is a member of the editorial advisory board of Clinical Endocrinology News. He reported that he has received consulting fees from Endo Pharmaceuticals and that his spouse is an employee of Parexel. Dr. Irwig reported no relevant conflicts of interest or financial disclosures.
NEW ORLEANS – A well-attended afternoon symposium on transgender medicine gave participants at the annual meeting of the Endocrine Society a solid grounding in transgender care, from prepubescence through adulthood. Here, Joshua Safer, MD, and Michael Irwig, MD, discuss highlights of the symposium, which brought together research, best practices, and clinical practice pearls.
In his presentation, Dr. Safer focused on The basics, he said, begin with presenting well-established, scientific principles supporting current standards of transgender care.
Dr. Safer, executive director of the Mount Sinai Center for Transgender Medicine and Surgery, New York, pointed out the critical role of gonadotropin-releasing hormone antagonists in delaying puberty for transgender girls. Blockade of puberty – and elevated testosterone – can forestall otherwise irreversible male secondary sex characteristics. These include laryngeal enlargement and bony changes of facial structure, for example.
Dr. Irwig, director of andrology at George Washington University, Washington, laid out the basics of transgender hormone therapy, including clinical pearls, such as avoiding ethinyl estradiol because of the heightened risk of venous thromboembolism.
Dr. Safer is a member of the editorial advisory board of Clinical Endocrinology News. He reported that he has received consulting fees from Endo Pharmaceuticals and that his spouse is an employee of Parexel. Dr. Irwig reported no relevant conflicts of interest or financial disclosures.
NEW ORLEANS – A well-attended afternoon symposium on transgender medicine gave participants at the annual meeting of the Endocrine Society a solid grounding in transgender care, from prepubescence through adulthood. Here, Joshua Safer, MD, and Michael Irwig, MD, discuss highlights of the symposium, which brought together research, best practices, and clinical practice pearls.
In his presentation, Dr. Safer focused on The basics, he said, begin with presenting well-established, scientific principles supporting current standards of transgender care.
Dr. Safer, executive director of the Mount Sinai Center for Transgender Medicine and Surgery, New York, pointed out the critical role of gonadotropin-releasing hormone antagonists in delaying puberty for transgender girls. Blockade of puberty – and elevated testosterone – can forestall otherwise irreversible male secondary sex characteristics. These include laryngeal enlargement and bony changes of facial structure, for example.
Dr. Irwig, director of andrology at George Washington University, Washington, laid out the basics of transgender hormone therapy, including clinical pearls, such as avoiding ethinyl estradiol because of the heightened risk of venous thromboembolism.
Dr. Safer is a member of the editorial advisory board of Clinical Endocrinology News. He reported that he has received consulting fees from Endo Pharmaceuticals and that his spouse is an employee of Parexel. Dr. Irwig reported no relevant conflicts of interest or financial disclosures.
EXPERT ANALYSIS FROM ENDO 2019
Ibrexafungerp effective against C. auris in two early case reports
A novel antifungal successfully eradicated Candida auris in two critically ill patients with fungemia, according to data presented in a poster session at the European Congress of Clinical Microbiology & Infectious Diseases.
The case reports, drawn from the phase 3 CARES study of the oral formulation of ibrexafungerp, demonstrated complete response to the glucan synthase inhibitor, according to Deven Juneja, MD, and his coauthors of the Max Super Specialty Hospital, New Delhi.
The first patient was an Asian male, aged 58 years, who had a previous history of diabetes and experienced a protracted ICU stay after acute ischemic stroke. He developed septic shock after aspiration pneumonia, and also experienced a popliteal thrombosis and liver, spleen, and kidney infarcts.
The patient had received empiric antibiotics with the addition of fluconazole; the antifungal was later switched to micafungin after C. auris was identified from blood cultures. Despite clinical improvement on micafungin, blood cultures remained positive for C. auris, so ibrexafungerp was started and continued for 17 days. Blood cultures became negative by day 3 of ibrexafungerp and remained negative for the follow-up period. The patient later developed Klebsiella pneumonia and died.
The second patient, an Asian female, aged 64 years, presented with a lower respiratory tract infection accompanied by fever and hypotension. She had a previous history of diabetes, hypertension, and chronic kidney disease with maintenance hemodialysis. Her fever also persisted despite antibiotics, and C. auris was isolated from her blood cultures with the subsequent initiation of ibrexafungerp. Her blood cultures were still positive at day 3 of ibrexafungerp, but negative at day 9 and 21. She completed 22 days of ibrexafungerp therapy and was asymptomatic with no evidence of C. auris recurrence at a 6-week follow-up visit.
The male patient experienced 2 days of loose stools soon after initiating ibrexafungerp; the female patient had no adverse events.
“These cases provide initial evidence of efficacy and safety of ibrexafungerp in the treatment of candidemia caused by C. auris, including in patients who failed previous therapies,” wrote Dr. Juneja and his coauthors in the late-breaking poster.
Ibrexafungerp belongs to a novel class of glucan synthase inhibitors called triterpenoids. Scynexis funded the CARES study and also is evaluating it alone or in combination with other antifungals for treatment of vulvovaginal candidiasis, invasive pulmonary aspergillosis, and refractory invasive and/or severe fungal disease.
SOURCE: Juneja D et al. ECCMID 2019, Poster L0028.
A novel antifungal successfully eradicated Candida auris in two critically ill patients with fungemia, according to data presented in a poster session at the European Congress of Clinical Microbiology & Infectious Diseases.
The case reports, drawn from the phase 3 CARES study of the oral formulation of ibrexafungerp, demonstrated complete response to the glucan synthase inhibitor, according to Deven Juneja, MD, and his coauthors of the Max Super Specialty Hospital, New Delhi.
The first patient was an Asian male, aged 58 years, who had a previous history of diabetes and experienced a protracted ICU stay after acute ischemic stroke. He developed septic shock after aspiration pneumonia, and also experienced a popliteal thrombosis and liver, spleen, and kidney infarcts.
The patient had received empiric antibiotics with the addition of fluconazole; the antifungal was later switched to micafungin after C. auris was identified from blood cultures. Despite clinical improvement on micafungin, blood cultures remained positive for C. auris, so ibrexafungerp was started and continued for 17 days. Blood cultures became negative by day 3 of ibrexafungerp and remained negative for the follow-up period. The patient later developed Klebsiella pneumonia and died.
The second patient, an Asian female, aged 64 years, presented with a lower respiratory tract infection accompanied by fever and hypotension. She had a previous history of diabetes, hypertension, and chronic kidney disease with maintenance hemodialysis. Her fever also persisted despite antibiotics, and C. auris was isolated from her blood cultures with the subsequent initiation of ibrexafungerp. Her blood cultures were still positive at day 3 of ibrexafungerp, but negative at day 9 and 21. She completed 22 days of ibrexafungerp therapy and was asymptomatic with no evidence of C. auris recurrence at a 6-week follow-up visit.
The male patient experienced 2 days of loose stools soon after initiating ibrexafungerp; the female patient had no adverse events.
“These cases provide initial evidence of efficacy and safety of ibrexafungerp in the treatment of candidemia caused by C. auris, including in patients who failed previous therapies,” wrote Dr. Juneja and his coauthors in the late-breaking poster.
Ibrexafungerp belongs to a novel class of glucan synthase inhibitors called triterpenoids. Scynexis funded the CARES study and also is evaluating it alone or in combination with other antifungals for treatment of vulvovaginal candidiasis, invasive pulmonary aspergillosis, and refractory invasive and/or severe fungal disease.
SOURCE: Juneja D et al. ECCMID 2019, Poster L0028.
A novel antifungal successfully eradicated Candida auris in two critically ill patients with fungemia, according to data presented in a poster session at the European Congress of Clinical Microbiology & Infectious Diseases.
The case reports, drawn from the phase 3 CARES study of the oral formulation of ibrexafungerp, demonstrated complete response to the glucan synthase inhibitor, according to Deven Juneja, MD, and his coauthors of the Max Super Specialty Hospital, New Delhi.
The first patient was an Asian male, aged 58 years, who had a previous history of diabetes and experienced a protracted ICU stay after acute ischemic stroke. He developed septic shock after aspiration pneumonia, and also experienced a popliteal thrombosis and liver, spleen, and kidney infarcts.
The patient had received empiric antibiotics with the addition of fluconazole; the antifungal was later switched to micafungin after C. auris was identified from blood cultures. Despite clinical improvement on micafungin, blood cultures remained positive for C. auris, so ibrexafungerp was started and continued for 17 days. Blood cultures became negative by day 3 of ibrexafungerp and remained negative for the follow-up period. The patient later developed Klebsiella pneumonia and died.
The second patient, an Asian female, aged 64 years, presented with a lower respiratory tract infection accompanied by fever and hypotension. She had a previous history of diabetes, hypertension, and chronic kidney disease with maintenance hemodialysis. Her fever also persisted despite antibiotics, and C. auris was isolated from her blood cultures with the subsequent initiation of ibrexafungerp. Her blood cultures were still positive at day 3 of ibrexafungerp, but negative at day 9 and 21. She completed 22 days of ibrexafungerp therapy and was asymptomatic with no evidence of C. auris recurrence at a 6-week follow-up visit.
The male patient experienced 2 days of loose stools soon after initiating ibrexafungerp; the female patient had no adverse events.
“These cases provide initial evidence of efficacy and safety of ibrexafungerp in the treatment of candidemia caused by C. auris, including in patients who failed previous therapies,” wrote Dr. Juneja and his coauthors in the late-breaking poster.
Ibrexafungerp belongs to a novel class of glucan synthase inhibitors called triterpenoids. Scynexis funded the CARES study and also is evaluating it alone or in combination with other antifungals for treatment of vulvovaginal candidiasis, invasive pulmonary aspergillosis, and refractory invasive and/or severe fungal disease.
SOURCE: Juneja D et al. ECCMID 2019, Poster L0028.
FROM ECCMID 2019
Candida auris: Dangerous and here to stay
Critical care units and long-term care facilities are on alert for cases of Candida auris, a novel fungal infection that is both dangerous to vulnerable patients and difficult to eradicate. The increased profile of C. auris is not a welcome development but is no surprise to critical care physicians.
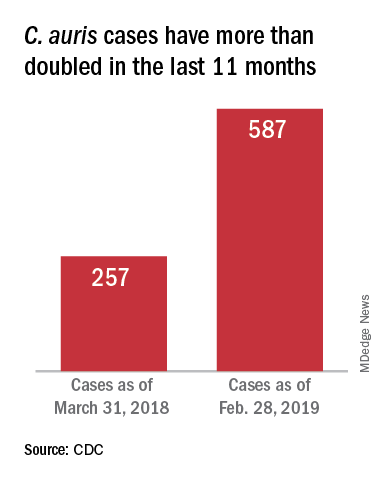
This pathogen was first identified in 2009 and has since been found in increasing numbers of patients all over the world. As expected, cases of C. auris are on the rise in the United States.
The Centers for Disease Control and Prevention stated “Candida auris is an emerging fungus that presents a serious global health threat.” This is an opportunistic pathogen that hits critically ill patients and those with compromised immunity.
On March 29, 2019, CDC reported that confirmed clinical cases of C. auris in the United States have more than doubled over the past year, from 257 cases in 2018 to 587 cases with an additional 1,056 colonized patients identified as of February 2019. “Most C. auris cases in the United States have been detected in the New York City area, New Jersey, and the Chicago area. Strains of C. auris in the United States have been linked to other parts of the world. U.S. C. auris cases are a result of inadvertent introduction into the United States from a patient who recently received health care in a country where C. auris has been reported or a result of local spread after such an introduction.”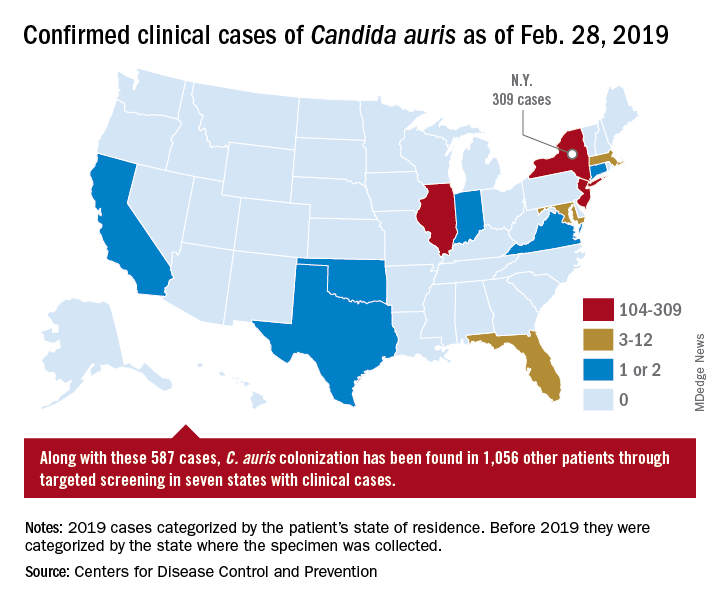
Case reports have found a mortality rate of up to 50% in patients with C. auris candidemia. The total number of cases is still small, but the trajectory is clear. The hunt is on in labs all over the world for optimal treatments and processes to handle outbreaks.
Jeniel Nett, MD, an infectious disease specialist, and a team of investigators at the University of Wisconsin, Madison, have focused their research on the characteristics of C. auris and its progression in patients and in medical facilities.
According to Dr. Nett, it’s not clear why this emerging threat has cropped up in multiple locations globally. “Candida auris was first recognized in 2009, in Japan, and relatively quickly we saw emergence of this species in relatively distant locations,” she said, adding that independent clades in these locations ruled out transmission as the source of the multiple outbreaks. Antifungal resistance is an epidemiologic area of concern and increased antifungal use may be a contributor, she said.

Once established, the organism is persistent: “It is found on mattresses, on bedsheets, IV poles, and a lot of reusable equipment,” said Dr. Nett in an interview. “It appears to persist in the environment for weeks – maybe longer.” In addition, “it seems to behave differently than a lot of the Candida species that we see; it readily colonizes the skin” to a much greater extent than does other Candida species, she said. “This allows it to be transmitted readily person to person, particularly in the hospitalized setting.” However, it can also colonize both the urinary and respiratory tracts, she said.
Which patients are susceptible to C. auris candidemia? “Many of these patients have undergone multiple procedures; they may have undergone mechanical ventilation as well as different surgical procedures,” said Dr. Nett. Affected patients often have received many rounds of antibiotic and antifungal treatment as well, she said, and may have an underlying illness like diabetes or malignancy.
Studies of C. auris outbreaks have begun to appear in the literature and give clinicians some perspective on the progression of an outbreak and potential strategies for containment. A prospective cohort study of a large outbreak of C. auris was conducted by Alba Ruiz-Gaitán, MD, and her colleagues at La Fe University and Polytechnic Hospital, Valencia, Spain (Expert Rev Anti Infect Ther. 2019 Apr;17[4]:295-305). The researchers followed 114 patients who were colonized with C. auris or had C. auris candidemia. The patients were compared with 114 case-matched controls within the hospital’s adult surgical and medical intensive care units over an 11-month period during the hospital’s protracted outbreak.
The investigators found a crude mortality rate of 58.5% at 30 days for patients with C. auris candidemia. All isolates in the study were completely resistant to fluconazole and had reduced susceptibility to voriconazole.
In critical care units at Hospital La Fe, investigators found C. auris on 25% of blood pressure cuffs, 10% of patient tables and keyboards, and 8% of infusion pumps.
Among the patients at Hospital La Fe, multivariable analysis revealed that those most likely to develop C. auris colonization or candidemia were individuals with polytrauma, cardiovascular disease, and cancer.
Patients receiving parenteral nutrition (odds ratio, 3.49), mechanical ventilation (OR, 2.43), and especially those having indwelling central venous catheters (OR, 13.48) were more likely to be colonized or have candidemia as well, according to Dr. Ruiz-Gaitán and her coauthors.
Once identified, how should C. auris be treated? “The majority of strains – upward of 90% – are resistant to fluconazole,” said Dr. Nett. “Moreover, 30%-50% of them are resistant to another antifungal, often amphotericin B. The isolates that we see in the United States are most often susceptible to an echinocandin, and echinocandins remain the choice for treatment of Candida auris pending susceptibility tests.”
However, in Valencia, “The susceptibility to echinocandins presented interesting features. These antifungals were not fungicidal against C. auris,” wrote Dr. Ruiz-Gaitán and her colleagues. They found that for caspofungin, “most isolates presented a clear paradoxical growth after 24 hours of incubation.” Additionally, fungal growth was inhibited at lower caspofungin concentrations, but rebounded at higher levels. Similar patterns were seen for anidulafungin and micafungin, they said.
These findings meant that Hospital La Fe patients received initial treatment with echinocandins, with the addition of liposomal amphotericin B or isavuconazole if candidemia persisted or clinical response was not seen, wrote the investigators.
Patient presentation is similar to other forms of candidiasis, said Dr. Nett. “Patients often have fever, chills, leukocytosis, and this persists despite antibacterial therapy… If Candida auris is suspected, the first course of action would be to place the patient in isolation, and laboratory staff should be alerted regarding the diagnosis.”
Most large clinical laboratories, she said, can now detect C. auris. Matrix-assisted laser desorption/ionization–time of flight is the identification technique of choice, provided that the databases are updated.
Smaller laboratories that use phenotypic tests may misidentify C. auris as another Candida species, or even as Saccharomyces cerevisiae – common beer yeast. Facilities without matrix-assisted laser desorption/ionization can find guidance for interpretation of phenotypic testing on the CDC website as well, said Dr. Nett.
After experiencing what they believe to be the largest C. auris outbreak at a single European hospital, Dr. Ruiz-Gaitán and her colleagues offered best-practice tips for treatment of patients with C. auris candidemia. These include removing mechanical devices as early as is safely practical; performing ophthalmologic examinations for endophthalmitis, a known C. auris complication; obtaining blood cultures every other day to track antimicrobial therapy to the point of sterilization; and searching for metastatic foci if blood cultures remain positive.
All instances of C. auris laboratory identification should be reported to the CDC at [email protected], and to local and state health agencies. The CDC recommends strict isolation and cleaning protocols, similar to those used for the spore-forming Clostridium difficile.
Dr. Nett reported funding support from the National Institutes of Health, the Burroughs Wellcome Fund, and the Doris Duke Charitable Foundation. She reported no conflicts of interest. Dr. Ruiz-Gaitán and her collaborators reported funding from Instituto de Salud Carlos III, Spain, and the Spanish Ministry of Science and University. They reported no conflicts of interest.
Critical care units and long-term care facilities are on alert for cases of Candida auris, a novel fungal infection that is both dangerous to vulnerable patients and difficult to eradicate. The increased profile of C. auris is not a welcome development but is no surprise to critical care physicians.

This pathogen was first identified in 2009 and has since been found in increasing numbers of patients all over the world. As expected, cases of C. auris are on the rise in the United States.
The Centers for Disease Control and Prevention stated “Candida auris is an emerging fungus that presents a serious global health threat.” This is an opportunistic pathogen that hits critically ill patients and those with compromised immunity.
On March 29, 2019, CDC reported that confirmed clinical cases of C. auris in the United States have more than doubled over the past year, from 257 cases in 2018 to 587 cases with an additional 1,056 colonized patients identified as of February 2019. “Most C. auris cases in the United States have been detected in the New York City area, New Jersey, and the Chicago area. Strains of C. auris in the United States have been linked to other parts of the world. U.S. C. auris cases are a result of inadvertent introduction into the United States from a patient who recently received health care in a country where C. auris has been reported or a result of local spread after such an introduction.”
Case reports have found a mortality rate of up to 50% in patients with C. auris candidemia. The total number of cases is still small, but the trajectory is clear. The hunt is on in labs all over the world for optimal treatments and processes to handle outbreaks.
Jeniel Nett, MD, an infectious disease specialist, and a team of investigators at the University of Wisconsin, Madison, have focused their research on the characteristics of C. auris and its progression in patients and in medical facilities.
According to Dr. Nett, it’s not clear why this emerging threat has cropped up in multiple locations globally. “Candida auris was first recognized in 2009, in Japan, and relatively quickly we saw emergence of this species in relatively distant locations,” she said, adding that independent clades in these locations ruled out transmission as the source of the multiple outbreaks. Antifungal resistance is an epidemiologic area of concern and increased antifungal use may be a contributor, she said.

Once established, the organism is persistent: “It is found on mattresses, on bedsheets, IV poles, and a lot of reusable equipment,” said Dr. Nett in an interview. “It appears to persist in the environment for weeks – maybe longer.” In addition, “it seems to behave differently than a lot of the Candida species that we see; it readily colonizes the skin” to a much greater extent than does other Candida species, she said. “This allows it to be transmitted readily person to person, particularly in the hospitalized setting.” However, it can also colonize both the urinary and respiratory tracts, she said.
Which patients are susceptible to C. auris candidemia? “Many of these patients have undergone multiple procedures; they may have undergone mechanical ventilation as well as different surgical procedures,” said Dr. Nett. Affected patients often have received many rounds of antibiotic and antifungal treatment as well, she said, and may have an underlying illness like diabetes or malignancy.
Studies of C. auris outbreaks have begun to appear in the literature and give clinicians some perspective on the progression of an outbreak and potential strategies for containment. A prospective cohort study of a large outbreak of C. auris was conducted by Alba Ruiz-Gaitán, MD, and her colleagues at La Fe University and Polytechnic Hospital, Valencia, Spain (Expert Rev Anti Infect Ther. 2019 Apr;17[4]:295-305). The researchers followed 114 patients who were colonized with C. auris or had C. auris candidemia. The patients were compared with 114 case-matched controls within the hospital’s adult surgical and medical intensive care units over an 11-month period during the hospital’s protracted outbreak.
The investigators found a crude mortality rate of 58.5% at 30 days for patients with C. auris candidemia. All isolates in the study were completely resistant to fluconazole and had reduced susceptibility to voriconazole.
In critical care units at Hospital La Fe, investigators found C. auris on 25% of blood pressure cuffs, 10% of patient tables and keyboards, and 8% of infusion pumps.
Among the patients at Hospital La Fe, multivariable analysis revealed that those most likely to develop C. auris colonization or candidemia were individuals with polytrauma, cardiovascular disease, and cancer.
Patients receiving parenteral nutrition (odds ratio, 3.49), mechanical ventilation (OR, 2.43), and especially those having indwelling central venous catheters (OR, 13.48) were more likely to be colonized or have candidemia as well, according to Dr. Ruiz-Gaitán and her coauthors.
Once identified, how should C. auris be treated? “The majority of strains – upward of 90% – are resistant to fluconazole,” said Dr. Nett. “Moreover, 30%-50% of them are resistant to another antifungal, often amphotericin B. The isolates that we see in the United States are most often susceptible to an echinocandin, and echinocandins remain the choice for treatment of Candida auris pending susceptibility tests.”
However, in Valencia, “The susceptibility to echinocandins presented interesting features. These antifungals were not fungicidal against C. auris,” wrote Dr. Ruiz-Gaitán and her colleagues. They found that for caspofungin, “most isolates presented a clear paradoxical growth after 24 hours of incubation.” Additionally, fungal growth was inhibited at lower caspofungin concentrations, but rebounded at higher levels. Similar patterns were seen for anidulafungin and micafungin, they said.
These findings meant that Hospital La Fe patients received initial treatment with echinocandins, with the addition of liposomal amphotericin B or isavuconazole if candidemia persisted or clinical response was not seen, wrote the investigators.
Patient presentation is similar to other forms of candidiasis, said Dr. Nett. “Patients often have fever, chills, leukocytosis, and this persists despite antibacterial therapy… If Candida auris is suspected, the first course of action would be to place the patient in isolation, and laboratory staff should be alerted regarding the diagnosis.”
Most large clinical laboratories, she said, can now detect C. auris. Matrix-assisted laser desorption/ionization–time of flight is the identification technique of choice, provided that the databases are updated.
Smaller laboratories that use phenotypic tests may misidentify C. auris as another Candida species, or even as Saccharomyces cerevisiae – common beer yeast. Facilities without matrix-assisted laser desorption/ionization can find guidance for interpretation of phenotypic testing on the CDC website as well, said Dr. Nett.
After experiencing what they believe to be the largest C. auris outbreak at a single European hospital, Dr. Ruiz-Gaitán and her colleagues offered best-practice tips for treatment of patients with C. auris candidemia. These include removing mechanical devices as early as is safely practical; performing ophthalmologic examinations for endophthalmitis, a known C. auris complication; obtaining blood cultures every other day to track antimicrobial therapy to the point of sterilization; and searching for metastatic foci if blood cultures remain positive.
All instances of C. auris laboratory identification should be reported to the CDC at [email protected], and to local and state health agencies. The CDC recommends strict isolation and cleaning protocols, similar to those used for the spore-forming Clostridium difficile.
Dr. Nett reported funding support from the National Institutes of Health, the Burroughs Wellcome Fund, and the Doris Duke Charitable Foundation. She reported no conflicts of interest. Dr. Ruiz-Gaitán and her collaborators reported funding from Instituto de Salud Carlos III, Spain, and the Spanish Ministry of Science and University. They reported no conflicts of interest.
Critical care units and long-term care facilities are on alert for cases of Candida auris, a novel fungal infection that is both dangerous to vulnerable patients and difficult to eradicate. The increased profile of C. auris is not a welcome development but is no surprise to critical care physicians.

This pathogen was first identified in 2009 and has since been found in increasing numbers of patients all over the world. As expected, cases of C. auris are on the rise in the United States.
The Centers for Disease Control and Prevention stated “Candida auris is an emerging fungus that presents a serious global health threat.” This is an opportunistic pathogen that hits critically ill patients and those with compromised immunity.
On March 29, 2019, CDC reported that confirmed clinical cases of C. auris in the United States have more than doubled over the past year, from 257 cases in 2018 to 587 cases with an additional 1,056 colonized patients identified as of February 2019. “Most C. auris cases in the United States have been detected in the New York City area, New Jersey, and the Chicago area. Strains of C. auris in the United States have been linked to other parts of the world. U.S. C. auris cases are a result of inadvertent introduction into the United States from a patient who recently received health care in a country where C. auris has been reported or a result of local spread after such an introduction.”
Case reports have found a mortality rate of up to 50% in patients with C. auris candidemia. The total number of cases is still small, but the trajectory is clear. The hunt is on in labs all over the world for optimal treatments and processes to handle outbreaks.
Jeniel Nett, MD, an infectious disease specialist, and a team of investigators at the University of Wisconsin, Madison, have focused their research on the characteristics of C. auris and its progression in patients and in medical facilities.
According to Dr. Nett, it’s not clear why this emerging threat has cropped up in multiple locations globally. “Candida auris was first recognized in 2009, in Japan, and relatively quickly we saw emergence of this species in relatively distant locations,” she said, adding that independent clades in these locations ruled out transmission as the source of the multiple outbreaks. Antifungal resistance is an epidemiologic area of concern and increased antifungal use may be a contributor, she said.

Once established, the organism is persistent: “It is found on mattresses, on bedsheets, IV poles, and a lot of reusable equipment,” said Dr. Nett in an interview. “It appears to persist in the environment for weeks – maybe longer.” In addition, “it seems to behave differently than a lot of the Candida species that we see; it readily colonizes the skin” to a much greater extent than does other Candida species, she said. “This allows it to be transmitted readily person to person, particularly in the hospitalized setting.” However, it can also colonize both the urinary and respiratory tracts, she said.
Which patients are susceptible to C. auris candidemia? “Many of these patients have undergone multiple procedures; they may have undergone mechanical ventilation as well as different surgical procedures,” said Dr. Nett. Affected patients often have received many rounds of antibiotic and antifungal treatment as well, she said, and may have an underlying illness like diabetes or malignancy.
Studies of C. auris outbreaks have begun to appear in the literature and give clinicians some perspective on the progression of an outbreak and potential strategies for containment. A prospective cohort study of a large outbreak of C. auris was conducted by Alba Ruiz-Gaitán, MD, and her colleagues at La Fe University and Polytechnic Hospital, Valencia, Spain (Expert Rev Anti Infect Ther. 2019 Apr;17[4]:295-305). The researchers followed 114 patients who were colonized with C. auris or had C. auris candidemia. The patients were compared with 114 case-matched controls within the hospital’s adult surgical and medical intensive care units over an 11-month period during the hospital’s protracted outbreak.
The investigators found a crude mortality rate of 58.5% at 30 days for patients with C. auris candidemia. All isolates in the study were completely resistant to fluconazole and had reduced susceptibility to voriconazole.
In critical care units at Hospital La Fe, investigators found C. auris on 25% of blood pressure cuffs, 10% of patient tables and keyboards, and 8% of infusion pumps.
Among the patients at Hospital La Fe, multivariable analysis revealed that those most likely to develop C. auris colonization or candidemia were individuals with polytrauma, cardiovascular disease, and cancer.
Patients receiving parenteral nutrition (odds ratio, 3.49), mechanical ventilation (OR, 2.43), and especially those having indwelling central venous catheters (OR, 13.48) were more likely to be colonized or have candidemia as well, according to Dr. Ruiz-Gaitán and her coauthors.
Once identified, how should C. auris be treated? “The majority of strains – upward of 90% – are resistant to fluconazole,” said Dr. Nett. “Moreover, 30%-50% of them are resistant to another antifungal, often amphotericin B. The isolates that we see in the United States are most often susceptible to an echinocandin, and echinocandins remain the choice for treatment of Candida auris pending susceptibility tests.”
However, in Valencia, “The susceptibility to echinocandins presented interesting features. These antifungals were not fungicidal against C. auris,” wrote Dr. Ruiz-Gaitán and her colleagues. They found that for caspofungin, “most isolates presented a clear paradoxical growth after 24 hours of incubation.” Additionally, fungal growth was inhibited at lower caspofungin concentrations, but rebounded at higher levels. Similar patterns were seen for anidulafungin and micafungin, they said.
These findings meant that Hospital La Fe patients received initial treatment with echinocandins, with the addition of liposomal amphotericin B or isavuconazole if candidemia persisted or clinical response was not seen, wrote the investigators.
Patient presentation is similar to other forms of candidiasis, said Dr. Nett. “Patients often have fever, chills, leukocytosis, and this persists despite antibacterial therapy… If Candida auris is suspected, the first course of action would be to place the patient in isolation, and laboratory staff should be alerted regarding the diagnosis.”
Most large clinical laboratories, she said, can now detect C. auris. Matrix-assisted laser desorption/ionization–time of flight is the identification technique of choice, provided that the databases are updated.
Smaller laboratories that use phenotypic tests may misidentify C. auris as another Candida species, or even as Saccharomyces cerevisiae – common beer yeast. Facilities without matrix-assisted laser desorption/ionization can find guidance for interpretation of phenotypic testing on the CDC website as well, said Dr. Nett.
After experiencing what they believe to be the largest C. auris outbreak at a single European hospital, Dr. Ruiz-Gaitán and her colleagues offered best-practice tips for treatment of patients with C. auris candidemia. These include removing mechanical devices as early as is safely practical; performing ophthalmologic examinations for endophthalmitis, a known C. auris complication; obtaining blood cultures every other day to track antimicrobial therapy to the point of sterilization; and searching for metastatic foci if blood cultures remain positive.
All instances of C. auris laboratory identification should be reported to the CDC at [email protected], and to local and state health agencies. The CDC recommends strict isolation and cleaning protocols, similar to those used for the spore-forming Clostridium difficile.
Dr. Nett reported funding support from the National Institutes of Health, the Burroughs Wellcome Fund, and the Doris Duke Charitable Foundation. She reported no conflicts of interest. Dr. Ruiz-Gaitán and her collaborators reported funding from Instituto de Salud Carlos III, Spain, and the Spanish Ministry of Science and University. They reported no conflicts of interest.