User login
COPD adds complexity to shared decision making for LDCT lung cancer screening
research suggests.
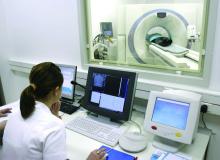
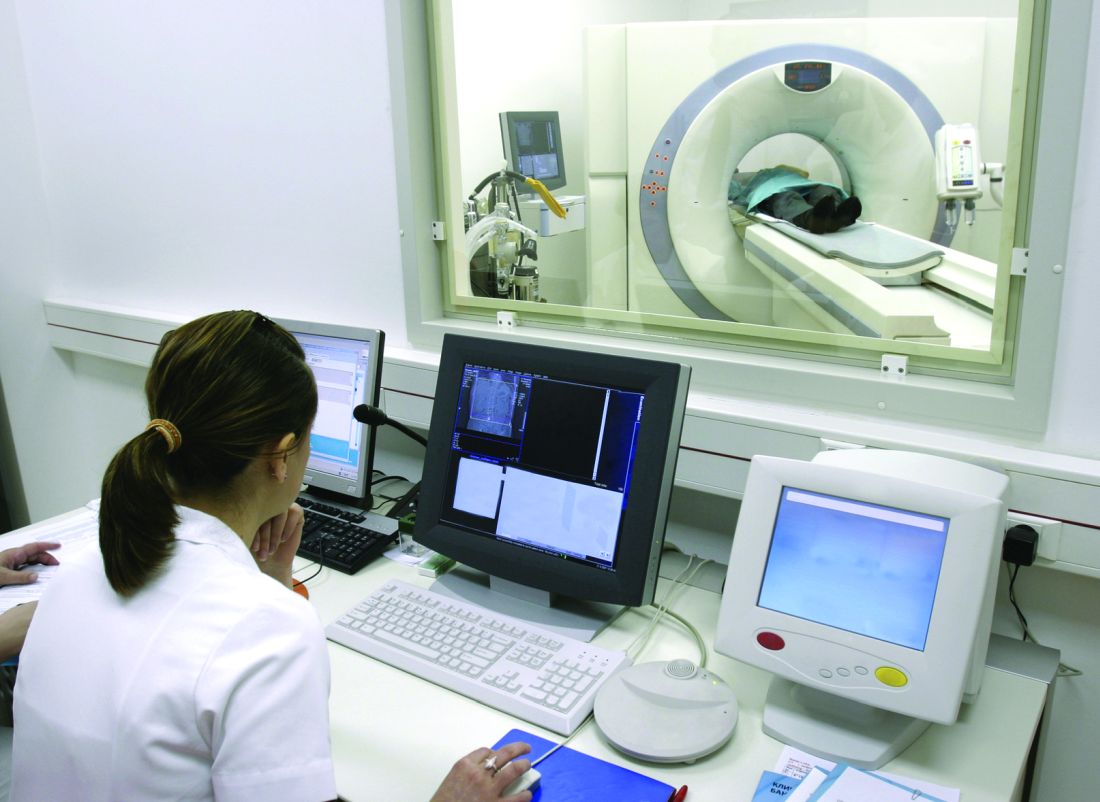
Jonathan M. Iaccarino, MD, of the pulmonary center at the Boston University, and coauthors reported the results of a secondary analysis of patient-level outcomes from 75,138 low-dose CT (LDCT) scans in 26,453 participants in the National Lung Screening Trial (Chest 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016).
Currently, LDCT screening is recommended annually for high-risk smokers aged 55-80 years. The National Lung Screening Trial showed that this screening achieved a 20% relative reduction in lung cancer mortality and 6.7% relative reduction in overall mortality in this group. The guidelines stress the importance of shared decision making, with discussion of the risks and benefits of screening.
Dr. Iaccarino and colleagues point out that decision aids for shared decision making need to include important baseline characteristics, such as the presence of COPD, as these can complicate the risk and benefit analysis.
In this study, they found that 14.2% of LDCT scans performed led to a subsequent diagnostic study and 1.5% resulted in an invasive procedure. In addition, 0.3% of scans resulted in a procedure-related complication, and in 89 cases (0.1%), this procedure-related complication was serious.
At the patient level, nearly one-third (30.5%) received a diagnostic study, 4.2% underwent an invasive procedure – 41% of whom ultimately were found not to have lung cancer – 0.9% had a procedure-related complication, and 0.3% had a serious procedure related complication. Furthermore, among those who experienced a serious complication, 12.5% were found not to have lung cancer.
“Our study analyzes cumulative outcomes at the level of the individual patient over the three years of LDCT screening during the NLST, showing higher rates of diagnostic procedures, invasive procedures, complications and serious complications than apparent when data is presented at the level of the individual test,” the authors wrote.
The 4,632 participants with COPD were significantly more likely to undergo diagnostic studies (36.2%), have an invasive procedure (6%), experience a procedure-related complication (1.5%) and experience a serious procedure-related complication (0.6%) than were participants without COPD. However, they also had a significantly higher incidence of lung cancer diagnosis than did participants without COPD (6.1% vs. 3.6%).
“While most decision aids note the risks of screening may be increased in those with COPD, our study helps quantify these increased risks as well as the increased likelihood of a lung cancer diagnosis, a critical advance given that providing personalized (rather than generic) information results in more accurate risk perception and more informed choices among individuals considering screening,” the authors wrote. “With the significant change in the balance of benefits and risks of screening in patients with COPD, it is critical to adjust the shared decision-making discussions accordingly.”
They also noted that other comorbidities, such as heart disease, vascular disease, and other lung diseases, would likely affect the balance of risk and benefit of LDCT screening, and that there was a need for further exploration of screening in these patients.
Noting the study’s limitations, the authors pointed that their analysis focused on outcomes that were not the primary outcomes of the National Lung Screening trial, and that they relied on self-reported COPD diagnoses.
The study was supported by the American Society of Clinical Oncology, the Charles A. King Trust, and Edith Nourse Rogers Memorial Veterans Hospital. No conflicts of interest were declared.
SOURCE: Iaccarino JM et al. CHEST 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016.
research suggests.
Jonathan M. Iaccarino, MD, of the pulmonary center at the Boston University, and coauthors reported the results of a secondary analysis of patient-level outcomes from 75,138 low-dose CT (LDCT) scans in 26,453 participants in the National Lung Screening Trial (Chest 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016).
Currently, LDCT screening is recommended annually for high-risk smokers aged 55-80 years. The National Lung Screening Trial showed that this screening achieved a 20% relative reduction in lung cancer mortality and 6.7% relative reduction in overall mortality in this group. The guidelines stress the importance of shared decision making, with discussion of the risks and benefits of screening.
Dr. Iaccarino and colleagues point out that decision aids for shared decision making need to include important baseline characteristics, such as the presence of COPD, as these can complicate the risk and benefit analysis.
In this study, they found that 14.2% of LDCT scans performed led to a subsequent diagnostic study and 1.5% resulted in an invasive procedure. In addition, 0.3% of scans resulted in a procedure-related complication, and in 89 cases (0.1%), this procedure-related complication was serious.
At the patient level, nearly one-third (30.5%) received a diagnostic study, 4.2% underwent an invasive procedure – 41% of whom ultimately were found not to have lung cancer – 0.9% had a procedure-related complication, and 0.3% had a serious procedure related complication. Furthermore, among those who experienced a serious complication, 12.5% were found not to have lung cancer.
“Our study analyzes cumulative outcomes at the level of the individual patient over the three years of LDCT screening during the NLST, showing higher rates of diagnostic procedures, invasive procedures, complications and serious complications than apparent when data is presented at the level of the individual test,” the authors wrote.
The 4,632 participants with COPD were significantly more likely to undergo diagnostic studies (36.2%), have an invasive procedure (6%), experience a procedure-related complication (1.5%) and experience a serious procedure-related complication (0.6%) than were participants without COPD. However, they also had a significantly higher incidence of lung cancer diagnosis than did participants without COPD (6.1% vs. 3.6%).
“While most decision aids note the risks of screening may be increased in those with COPD, our study helps quantify these increased risks as well as the increased likelihood of a lung cancer diagnosis, a critical advance given that providing personalized (rather than generic) information results in more accurate risk perception and more informed choices among individuals considering screening,” the authors wrote. “With the significant change in the balance of benefits and risks of screening in patients with COPD, it is critical to adjust the shared decision-making discussions accordingly.”
They also noted that other comorbidities, such as heart disease, vascular disease, and other lung diseases, would likely affect the balance of risk and benefit of LDCT screening, and that there was a need for further exploration of screening in these patients.
Noting the study’s limitations, the authors pointed that their analysis focused on outcomes that were not the primary outcomes of the National Lung Screening trial, and that they relied on self-reported COPD diagnoses.
The study was supported by the American Society of Clinical Oncology, the Charles A. King Trust, and Edith Nourse Rogers Memorial Veterans Hospital. No conflicts of interest were declared.
SOURCE: Iaccarino JM et al. CHEST 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016.
research suggests.
Jonathan M. Iaccarino, MD, of the pulmonary center at the Boston University, and coauthors reported the results of a secondary analysis of patient-level outcomes from 75,138 low-dose CT (LDCT) scans in 26,453 participants in the National Lung Screening Trial (Chest 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016).
Currently, LDCT screening is recommended annually for high-risk smokers aged 55-80 years. The National Lung Screening Trial showed that this screening achieved a 20% relative reduction in lung cancer mortality and 6.7% relative reduction in overall mortality in this group. The guidelines stress the importance of shared decision making, with discussion of the risks and benefits of screening.
Dr. Iaccarino and colleagues point out that decision aids for shared decision making need to include important baseline characteristics, such as the presence of COPD, as these can complicate the risk and benefit analysis.
In this study, they found that 14.2% of LDCT scans performed led to a subsequent diagnostic study and 1.5% resulted in an invasive procedure. In addition, 0.3% of scans resulted in a procedure-related complication, and in 89 cases (0.1%), this procedure-related complication was serious.
At the patient level, nearly one-third (30.5%) received a diagnostic study, 4.2% underwent an invasive procedure – 41% of whom ultimately were found not to have lung cancer – 0.9% had a procedure-related complication, and 0.3% had a serious procedure related complication. Furthermore, among those who experienced a serious complication, 12.5% were found not to have lung cancer.
“Our study analyzes cumulative outcomes at the level of the individual patient over the three years of LDCT screening during the NLST, showing higher rates of diagnostic procedures, invasive procedures, complications and serious complications than apparent when data is presented at the level of the individual test,” the authors wrote.
The 4,632 participants with COPD were significantly more likely to undergo diagnostic studies (36.2%), have an invasive procedure (6%), experience a procedure-related complication (1.5%) and experience a serious procedure-related complication (0.6%) than were participants without COPD. However, they also had a significantly higher incidence of lung cancer diagnosis than did participants without COPD (6.1% vs. 3.6%).
“While most decision aids note the risks of screening may be increased in those with COPD, our study helps quantify these increased risks as well as the increased likelihood of a lung cancer diagnosis, a critical advance given that providing personalized (rather than generic) information results in more accurate risk perception and more informed choices among individuals considering screening,” the authors wrote. “With the significant change in the balance of benefits and risks of screening in patients with COPD, it is critical to adjust the shared decision-making discussions accordingly.”
They also noted that other comorbidities, such as heart disease, vascular disease, and other lung diseases, would likely affect the balance of risk and benefit of LDCT screening, and that there was a need for further exploration of screening in these patients.
Noting the study’s limitations, the authors pointed that their analysis focused on outcomes that were not the primary outcomes of the National Lung Screening trial, and that they relied on self-reported COPD diagnoses.
The study was supported by the American Society of Clinical Oncology, the Charles A. King Trust, and Edith Nourse Rogers Memorial Veterans Hospital. No conflicts of interest were declared.
SOURCE: Iaccarino JM et al. CHEST 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016.
FROM CHEST
Cost a factor in breast cancer treatment decisions
Treatment costs are a significant factor in women’s decision making around breast cancer surgery, investigators reported.
With the health care costs of breast cancer estimated to reach $20 billion by 2020 in the United States, many of those costs are being shifted onto patients themselves, wrote Rachel A. Greenup, MD, from Duke University, Durham, N.C., and coauthors in the Journal of Oncology Practice.
“This financial hardship is now recognized as a major adverse effect of cancer care and has been associated with reduced quality of life, nonadherence, and an increased risk of early mortality,” they wrote.
Researchers surveyed 607 women with a history of breast cancer to examine the impact that cost had on their decisions about surgery and what financial harm they had experienced after breast cancer surgery.
Overall, 43% of women said they considered costs when making decisions about breast cancer treatment, 28% said cost influenced their decision making around breast cancer surgery, and 14% said costs were extremely important in that decision.
Women in the lowest income bracket – earning at or below $45,000 per year – identified cost as the most influential factor in their decision about breast cancer surgery, above loss of sensation, breast preservation or appearance, the need for long-term surveillance, or avoiding radiation.
However, more than three-quarters of women said they never discussed costs with their medical team.
Bilateral mastectomy, with and without reconstruction, was associated with higher patient-reported out-of-pocket costs, higher debt, higher rates of cancer-induced financial hardship, and higher rates of altered or reduced employment, compared with breast-conserving surgery.
More than one-third of participants reported significant to catastrophic financial burden because of their breast cancer care.
Even in the highest income brackets, two-thirds of women were financially unprepared for the cost of treatment, and 26% said their treatment costs were higher than expected.
The authors commented that “cost transparency” was uncommon between oncologically equivalent surgical treatments, “thus, patients with breast cancer may unknowingly be guiding therapeutic decisions that increase the risk of financial harm.”
“To date, patient out-of-pocket costs and subsequent risk of financial harm have not been routinely incorporated into shared decisions for breast cancer surgery, a process that has otherwise highly revered patient values,” they wrote.
The investigators suggested that revealing the greater risk for financial burden associated with treatments like bilateral mastectomy could help inform surgical treatment decisions.
The study was supported by the National Institutes of Health and the Duke Cancer Institute. Six authors reported honoraria, research funding, prior employment, and other support from the pharmaceutical sector.
SOURCE: Greenup RA et al. J Oncol Pract. 2019 Jul 29. doi: 10.1200/JOP.18.00796.
Treatment costs are a significant factor in women’s decision making around breast cancer surgery, investigators reported.
With the health care costs of breast cancer estimated to reach $20 billion by 2020 in the United States, many of those costs are being shifted onto patients themselves, wrote Rachel A. Greenup, MD, from Duke University, Durham, N.C., and coauthors in the Journal of Oncology Practice.
“This financial hardship is now recognized as a major adverse effect of cancer care and has been associated with reduced quality of life, nonadherence, and an increased risk of early mortality,” they wrote.
Researchers surveyed 607 women with a history of breast cancer to examine the impact that cost had on their decisions about surgery and what financial harm they had experienced after breast cancer surgery.
Overall, 43% of women said they considered costs when making decisions about breast cancer treatment, 28% said cost influenced their decision making around breast cancer surgery, and 14% said costs were extremely important in that decision.
Women in the lowest income bracket – earning at or below $45,000 per year – identified cost as the most influential factor in their decision about breast cancer surgery, above loss of sensation, breast preservation or appearance, the need for long-term surveillance, or avoiding radiation.
However, more than three-quarters of women said they never discussed costs with their medical team.
Bilateral mastectomy, with and without reconstruction, was associated with higher patient-reported out-of-pocket costs, higher debt, higher rates of cancer-induced financial hardship, and higher rates of altered or reduced employment, compared with breast-conserving surgery.
More than one-third of participants reported significant to catastrophic financial burden because of their breast cancer care.
Even in the highest income brackets, two-thirds of women were financially unprepared for the cost of treatment, and 26% said their treatment costs were higher than expected.
The authors commented that “cost transparency” was uncommon between oncologically equivalent surgical treatments, “thus, patients with breast cancer may unknowingly be guiding therapeutic decisions that increase the risk of financial harm.”
“To date, patient out-of-pocket costs and subsequent risk of financial harm have not been routinely incorporated into shared decisions for breast cancer surgery, a process that has otherwise highly revered patient values,” they wrote.
The investigators suggested that revealing the greater risk for financial burden associated with treatments like bilateral mastectomy could help inform surgical treatment decisions.
The study was supported by the National Institutes of Health and the Duke Cancer Institute. Six authors reported honoraria, research funding, prior employment, and other support from the pharmaceutical sector.
SOURCE: Greenup RA et al. J Oncol Pract. 2019 Jul 29. doi: 10.1200/JOP.18.00796.
Treatment costs are a significant factor in women’s decision making around breast cancer surgery, investigators reported.
With the health care costs of breast cancer estimated to reach $20 billion by 2020 in the United States, many of those costs are being shifted onto patients themselves, wrote Rachel A. Greenup, MD, from Duke University, Durham, N.C., and coauthors in the Journal of Oncology Practice.
“This financial hardship is now recognized as a major adverse effect of cancer care and has been associated with reduced quality of life, nonadherence, and an increased risk of early mortality,” they wrote.
Researchers surveyed 607 women with a history of breast cancer to examine the impact that cost had on their decisions about surgery and what financial harm they had experienced after breast cancer surgery.
Overall, 43% of women said they considered costs when making decisions about breast cancer treatment, 28% said cost influenced their decision making around breast cancer surgery, and 14% said costs were extremely important in that decision.
Women in the lowest income bracket – earning at or below $45,000 per year – identified cost as the most influential factor in their decision about breast cancer surgery, above loss of sensation, breast preservation or appearance, the need for long-term surveillance, or avoiding radiation.
However, more than three-quarters of women said they never discussed costs with their medical team.
Bilateral mastectomy, with and without reconstruction, was associated with higher patient-reported out-of-pocket costs, higher debt, higher rates of cancer-induced financial hardship, and higher rates of altered or reduced employment, compared with breast-conserving surgery.
More than one-third of participants reported significant to catastrophic financial burden because of their breast cancer care.
Even in the highest income brackets, two-thirds of women were financially unprepared for the cost of treatment, and 26% said their treatment costs were higher than expected.
The authors commented that “cost transparency” was uncommon between oncologically equivalent surgical treatments, “thus, patients with breast cancer may unknowingly be guiding therapeutic decisions that increase the risk of financial harm.”
“To date, patient out-of-pocket costs and subsequent risk of financial harm have not been routinely incorporated into shared decisions for breast cancer surgery, a process that has otherwise highly revered patient values,” they wrote.
The investigators suggested that revealing the greater risk for financial burden associated with treatments like bilateral mastectomy could help inform surgical treatment decisions.
The study was supported by the National Institutes of Health and the Duke Cancer Institute. Six authors reported honoraria, research funding, prior employment, and other support from the pharmaceutical sector.
SOURCE: Greenup RA et al. J Oncol Pract. 2019 Jul 29. doi: 10.1200/JOP.18.00796.
FROM THE JOURNAL OF ONCOLOGY PRACTICE
Brain imaging could predict response to CBT in depression
Researchers have used functional MRI to identify key differences between individuals with depression who respond to cognitive-behavioral therapy and those who don’t.
In a paper published in Science Advances, Filippo Queirazza, MD, PhD, and coauthors reported the outcomes of a study of 37 individuals with untreated depression who took part in an online, self-guided cognitive-behavioral therapy (CBT) program.
The participants – 18 of whom were women – attended an appointment before and 2 months after completing the therapy program, at which they were clinically evaluated by a psychiatrist, and underwent fMRI scanning. Only 26 subjects completed the program and attended the posttreatment appointment, wrote Dr. Queirazza of the Institute of Neuroscience and Psychology at the University of Glasgow, Scotland.
To look for indication of treatment response in fMRI scanning, researchers got participants to do a series of reverse-learning tasks while they were being scanned. That activity involved learning which of two stimuli gave the highest payoff rate.
“The choice of a relevant generative model in MDD is dictated by a wealth of behavioral and neural findings, suggesting that learning from positive (reward) and negative (punishment) feedback [also known as reinforcement learning] is substantially impaired in depressed subjects,” the authors wrote.
The imaging suggested that individuals who responded showed more pretreatment neural activity in areas of the brain that deal with the acquisition and processing of feedback information, and that the level of activity was in proportion to the magnitude of their later treatment response.
They also found that activity in right striatum was best at discriminating responders from nonresponders. Based on their findings, they were able to predict responders as well as did pretreatment BDI-II scores.
“Our finding that neural activity in the right striatum is positively correlated with CBT response is consistent with a previous report that, in a group of adolescents with depression, greater pretreatment striatal responses to both anticipation and presentation of positive feedback during a monetary reward task were linked to posttreatment reduction in depression severity, particularly of anxiety symptoms,” they wrote.
The study was supported by several entities, including the Chief Scientist Office and the Dr Mortimer and Theresa Sackler Foundation. No competing interests were declared.
SOURCE: Queirazza F et al. Sci Adv. 2019 Jul 31. doi: 10.1126/sciadv.aav4962.
Researchers have used functional MRI to identify key differences between individuals with depression who respond to cognitive-behavioral therapy and those who don’t.
In a paper published in Science Advances, Filippo Queirazza, MD, PhD, and coauthors reported the outcomes of a study of 37 individuals with untreated depression who took part in an online, self-guided cognitive-behavioral therapy (CBT) program.
The participants – 18 of whom were women – attended an appointment before and 2 months after completing the therapy program, at which they were clinically evaluated by a psychiatrist, and underwent fMRI scanning. Only 26 subjects completed the program and attended the posttreatment appointment, wrote Dr. Queirazza of the Institute of Neuroscience and Psychology at the University of Glasgow, Scotland.
To look for indication of treatment response in fMRI scanning, researchers got participants to do a series of reverse-learning tasks while they were being scanned. That activity involved learning which of two stimuli gave the highest payoff rate.
“The choice of a relevant generative model in MDD is dictated by a wealth of behavioral and neural findings, suggesting that learning from positive (reward) and negative (punishment) feedback [also known as reinforcement learning] is substantially impaired in depressed subjects,” the authors wrote.
The imaging suggested that individuals who responded showed more pretreatment neural activity in areas of the brain that deal with the acquisition and processing of feedback information, and that the level of activity was in proportion to the magnitude of their later treatment response.
They also found that activity in right striatum was best at discriminating responders from nonresponders. Based on their findings, they were able to predict responders as well as did pretreatment BDI-II scores.
“Our finding that neural activity in the right striatum is positively correlated with CBT response is consistent with a previous report that, in a group of adolescents with depression, greater pretreatment striatal responses to both anticipation and presentation of positive feedback during a monetary reward task were linked to posttreatment reduction in depression severity, particularly of anxiety symptoms,” they wrote.
The study was supported by several entities, including the Chief Scientist Office and the Dr Mortimer and Theresa Sackler Foundation. No competing interests were declared.
SOURCE: Queirazza F et al. Sci Adv. 2019 Jul 31. doi: 10.1126/sciadv.aav4962.
Researchers have used functional MRI to identify key differences between individuals with depression who respond to cognitive-behavioral therapy and those who don’t.
In a paper published in Science Advances, Filippo Queirazza, MD, PhD, and coauthors reported the outcomes of a study of 37 individuals with untreated depression who took part in an online, self-guided cognitive-behavioral therapy (CBT) program.
The participants – 18 of whom were women – attended an appointment before and 2 months after completing the therapy program, at which they were clinically evaluated by a psychiatrist, and underwent fMRI scanning. Only 26 subjects completed the program and attended the posttreatment appointment, wrote Dr. Queirazza of the Institute of Neuroscience and Psychology at the University of Glasgow, Scotland.
To look for indication of treatment response in fMRI scanning, researchers got participants to do a series of reverse-learning tasks while they were being scanned. That activity involved learning which of two stimuli gave the highest payoff rate.
“The choice of a relevant generative model in MDD is dictated by a wealth of behavioral and neural findings, suggesting that learning from positive (reward) and negative (punishment) feedback [also known as reinforcement learning] is substantially impaired in depressed subjects,” the authors wrote.
The imaging suggested that individuals who responded showed more pretreatment neural activity in areas of the brain that deal with the acquisition and processing of feedback information, and that the level of activity was in proportion to the magnitude of their later treatment response.
They also found that activity in right striatum was best at discriminating responders from nonresponders. Based on their findings, they were able to predict responders as well as did pretreatment BDI-II scores.
“Our finding that neural activity in the right striatum is positively correlated with CBT response is consistent with a previous report that, in a group of adolescents with depression, greater pretreatment striatal responses to both anticipation and presentation of positive feedback during a monetary reward task were linked to posttreatment reduction in depression severity, particularly of anxiety symptoms,” they wrote.
The study was supported by several entities, including the Chief Scientist Office and the Dr Mortimer and Theresa Sackler Foundation. No competing interests were declared.
SOURCE: Queirazza F et al. Sci Adv. 2019 Jul 31. doi: 10.1126/sciadv.aav4962.
FROM SCIENCE ADVANCES
Placental bacteria not linked to adverse pregnancy outcomes
such as preeclampsia or spontaneous preterm birth, a study has found.
Dr. Marcus C. de Goffau, of Wellcome Sanger Institute, Cambridge, England, and coauthors wrote that placental dysfunction is linked to a number of common adverse pregnancy outcomes, but in many cases the cause of that dysfunction is unknown.
“Several studies have used sequencing-based methods for bacterial detection (metagenomics and 16S rRNA gene amplicon sequencing), and have concluded that the placenta is physiologically colonized by a diverse population of bacteria (the ‘placental microbiome’) and that the nature of this colonization may differ between healthy and complicated pregnancies,” they wrote.
In a paper published in the July 31 edition of Nature (doi: 10.1038/s41586-019-1451-5), researchers reported the outcomes of a study involving two cohorts. The first included 80 babies delivered by prelabor caesarean section, of whom 20 were small for gestational age, 20 were delivered to mothers with preeclampsia, and 40 were matched controls. The second cohort comprised 100 patients with preeclampsia, 100 small-for-gestational-age infants, 100 preterm births, and 198 matched controls, with two controls having been used twice.
The bacterial content of the placentas was analyzed via deep metagenomic sequencing of total DNA and 16S rRNA gene amplicon sequencing in cohort 1, and 16S rRNA gene amplicon from two different kits for cohort 2’s samples. In the first cohort, samples were also spiked with Salmonella bongori as a positive control.
For cohort 1, researchers were able to detect the S. bongori in all samples, but all other bacterial signals showed clear links to different batches, which the authors said showed they were the result of contamination. This was further supported by the discovery that the Escherichia coli signal seen in a number of batches was all from the same bacterial strain.
In the second cohort, researchers found Bradyrhizobium in nearly all samples, and Burkholderia – which has been thought to play a role in preterm birth – in some samples.
However in the case of the Burkholderia, significant variation between different runs of tests was found, and both Bradyrhizobium and Burkholderia were also found in negative controls.
Researchers also saw a high prevalence of four “ecologically unexpected” bacterial groups but were able to show that these were likely contaminants from a reagent used to wash the placental samples.
The study found that vaginal organisms such as lactobacilli and vaginosis-associated bacteria were more abundant in the second cohort, where babies were delivered by a mix of vaginal, intrapartum and prelabor caesarean section, compared with the first cohort, who were all prelabor caesarean section deliveries.
The only organism that met all the criteria for a genuine placenta-associated bacterial signal was Streptococcus agalactiae, but the authors found no association between it and pregnancy outcomes.
However, they did sound a warning that the perinatal transmission of S. agalactiae from the mother’s genital tract to the neonate could be a cause of fatal sepsis in the infant.
“Further studies will be required to determine the association between the presence of the organism in the placenta and fetal or neonatal disease,” they wrote.
The researchers also found significant associations between the delivery-associated vaginal bacteria Lactobacillus iners and preeclampsia, and between Streptococcus anginosus and the Ureaplasma genus – both of which are associated with vaginosis – and preterm birth.
“We conclude that bacterial placental infection is not a major cause of placentally related complications of human pregnancy and that the human placenta does not have a resident microbiome,” the authors wrote. “Although we see no evidence of a placental microbiome, the frequency of detection of vaginal bacteria in the placenta increased after intrapartum [cesarean] section, suggesting ascending or haematogenous spread.”
The Medical Research Council and the National Institute for Health Research Cambridge Biomedical Research Centre supported the study. Five authors declared grants and support from private industry outside the submitted work.
SOURCE: de Goffau M et al. Nature. 2019 Jul 31. doi: 10.1038/s41586-019-1451-5.
such as preeclampsia or spontaneous preterm birth, a study has found.
Dr. Marcus C. de Goffau, of Wellcome Sanger Institute, Cambridge, England, and coauthors wrote that placental dysfunction is linked to a number of common adverse pregnancy outcomes, but in many cases the cause of that dysfunction is unknown.
“Several studies have used sequencing-based methods for bacterial detection (metagenomics and 16S rRNA gene amplicon sequencing), and have concluded that the placenta is physiologically colonized by a diverse population of bacteria (the ‘placental microbiome’) and that the nature of this colonization may differ between healthy and complicated pregnancies,” they wrote.
In a paper published in the July 31 edition of Nature (doi: 10.1038/s41586-019-1451-5), researchers reported the outcomes of a study involving two cohorts. The first included 80 babies delivered by prelabor caesarean section, of whom 20 were small for gestational age, 20 were delivered to mothers with preeclampsia, and 40 were matched controls. The second cohort comprised 100 patients with preeclampsia, 100 small-for-gestational-age infants, 100 preterm births, and 198 matched controls, with two controls having been used twice.
The bacterial content of the placentas was analyzed via deep metagenomic sequencing of total DNA and 16S rRNA gene amplicon sequencing in cohort 1, and 16S rRNA gene amplicon from two different kits for cohort 2’s samples. In the first cohort, samples were also spiked with Salmonella bongori as a positive control.
For cohort 1, researchers were able to detect the S. bongori in all samples, but all other bacterial signals showed clear links to different batches, which the authors said showed they were the result of contamination. This was further supported by the discovery that the Escherichia coli signal seen in a number of batches was all from the same bacterial strain.
In the second cohort, researchers found Bradyrhizobium in nearly all samples, and Burkholderia – which has been thought to play a role in preterm birth – in some samples.
However in the case of the Burkholderia, significant variation between different runs of tests was found, and both Bradyrhizobium and Burkholderia were also found in negative controls.
Researchers also saw a high prevalence of four “ecologically unexpected” bacterial groups but were able to show that these were likely contaminants from a reagent used to wash the placental samples.
The study found that vaginal organisms such as lactobacilli and vaginosis-associated bacteria were more abundant in the second cohort, where babies were delivered by a mix of vaginal, intrapartum and prelabor caesarean section, compared with the first cohort, who were all prelabor caesarean section deliveries.
The only organism that met all the criteria for a genuine placenta-associated bacterial signal was Streptococcus agalactiae, but the authors found no association between it and pregnancy outcomes.
However, they did sound a warning that the perinatal transmission of S. agalactiae from the mother’s genital tract to the neonate could be a cause of fatal sepsis in the infant.
“Further studies will be required to determine the association between the presence of the organism in the placenta and fetal or neonatal disease,” they wrote.
The researchers also found significant associations between the delivery-associated vaginal bacteria Lactobacillus iners and preeclampsia, and between Streptococcus anginosus and the Ureaplasma genus – both of which are associated with vaginosis – and preterm birth.
“We conclude that bacterial placental infection is not a major cause of placentally related complications of human pregnancy and that the human placenta does not have a resident microbiome,” the authors wrote. “Although we see no evidence of a placental microbiome, the frequency of detection of vaginal bacteria in the placenta increased after intrapartum [cesarean] section, suggesting ascending or haematogenous spread.”
The Medical Research Council and the National Institute for Health Research Cambridge Biomedical Research Centre supported the study. Five authors declared grants and support from private industry outside the submitted work.
SOURCE: de Goffau M et al. Nature. 2019 Jul 31. doi: 10.1038/s41586-019-1451-5.
such as preeclampsia or spontaneous preterm birth, a study has found.
Dr. Marcus C. de Goffau, of Wellcome Sanger Institute, Cambridge, England, and coauthors wrote that placental dysfunction is linked to a number of common adverse pregnancy outcomes, but in many cases the cause of that dysfunction is unknown.
“Several studies have used sequencing-based methods for bacterial detection (metagenomics and 16S rRNA gene amplicon sequencing), and have concluded that the placenta is physiologically colonized by a diverse population of bacteria (the ‘placental microbiome’) and that the nature of this colonization may differ between healthy and complicated pregnancies,” they wrote.
In a paper published in the July 31 edition of Nature (doi: 10.1038/s41586-019-1451-5), researchers reported the outcomes of a study involving two cohorts. The first included 80 babies delivered by prelabor caesarean section, of whom 20 were small for gestational age, 20 were delivered to mothers with preeclampsia, and 40 were matched controls. The second cohort comprised 100 patients with preeclampsia, 100 small-for-gestational-age infants, 100 preterm births, and 198 matched controls, with two controls having been used twice.
The bacterial content of the placentas was analyzed via deep metagenomic sequencing of total DNA and 16S rRNA gene amplicon sequencing in cohort 1, and 16S rRNA gene amplicon from two different kits for cohort 2’s samples. In the first cohort, samples were also spiked with Salmonella bongori as a positive control.
For cohort 1, researchers were able to detect the S. bongori in all samples, but all other bacterial signals showed clear links to different batches, which the authors said showed they were the result of contamination. This was further supported by the discovery that the Escherichia coli signal seen in a number of batches was all from the same bacterial strain.
In the second cohort, researchers found Bradyrhizobium in nearly all samples, and Burkholderia – which has been thought to play a role in preterm birth – in some samples.
However in the case of the Burkholderia, significant variation between different runs of tests was found, and both Bradyrhizobium and Burkholderia were also found in negative controls.
Researchers also saw a high prevalence of four “ecologically unexpected” bacterial groups but were able to show that these were likely contaminants from a reagent used to wash the placental samples.
The study found that vaginal organisms such as lactobacilli and vaginosis-associated bacteria were more abundant in the second cohort, where babies were delivered by a mix of vaginal, intrapartum and prelabor caesarean section, compared with the first cohort, who were all prelabor caesarean section deliveries.
The only organism that met all the criteria for a genuine placenta-associated bacterial signal was Streptococcus agalactiae, but the authors found no association between it and pregnancy outcomes.
However, they did sound a warning that the perinatal transmission of S. agalactiae from the mother’s genital tract to the neonate could be a cause of fatal sepsis in the infant.
“Further studies will be required to determine the association between the presence of the organism in the placenta and fetal or neonatal disease,” they wrote.
The researchers also found significant associations between the delivery-associated vaginal bacteria Lactobacillus iners and preeclampsia, and between Streptococcus anginosus and the Ureaplasma genus – both of which are associated with vaginosis – and preterm birth.
“We conclude that bacterial placental infection is not a major cause of placentally related complications of human pregnancy and that the human placenta does not have a resident microbiome,” the authors wrote. “Although we see no evidence of a placental microbiome, the frequency of detection of vaginal bacteria in the placenta increased after intrapartum [cesarean] section, suggesting ascending or haematogenous spread.”
The Medical Research Council and the National Institute for Health Research Cambridge Biomedical Research Centre supported the study. Five authors declared grants and support from private industry outside the submitted work.
SOURCE: de Goffau M et al. Nature. 2019 Jul 31. doi: 10.1038/s41586-019-1451-5.
FROM NATURE
Key clinical point: Researchers found no evidence of a placental biome or a link between placental bacterial contamination and adverse pregnancy outcomes.
Major finding: No bacterial contamination of the placenta was associated with adverse pregnancy outcomes such as preeclampsia.
Study details: Cohort study of more than 500 placental samples.
Disclosures: The Medical Research Council and the National Institute for Health Research Cambridge Biomedical Research Centre supported the study. Five authors declared grants and support from private industry outside the submitted work.
Source: de Goffau M et al. Nature. 2019 Jul 31. doi: 10.1038/s41586-019-1451-5.
Researchers combine genetic and clinical factors in new VTE risk score
MELBOURNE – A venous thromboembolism risk score that combines clinical risk factors, such as lymphoma type and stage, along with genetic variables, could offer a better way to predict venous thromboembolism in patients with lymphoma, according to new findings presented at the International Society on Thrombosis and Haemostasis congress.
Cristina Pascual, MD, of the Hospital Universitario Gregorio Marañon in Madrid presented data from a development and validation study of a clinical-genetic risk model for thrombosis in lymphoma in 208 patients with lymphoma, 31 of whom experienced a venous thromboembolic event.
While the relationship between cancer and increased thrombosis risk is well recognized, lymphoma patients are at particularly high risk, with an estimated thrombosis incidence of 5%-10%, Dr. Pascual said.
Currently, the Khorana score is the most validated risk score for thrombosis in patients with solid tumors, using factors such as tumor site, platelet and leukocyte count, hemoglobin levels, and body mass index. However, Dr. Pascual pointed out that just 10% of the validation cohort for the Khorana score were lymphoma patients, and it had previously been found to be not as useful for that population.
More recently, researchers had developed the ThroLy score for predicting thromboembolic events specifically in patients with lymphoma, incorporating clinical variables such as mediastinal involvement and extranodal localization.
Another group took a different approach by incorporating genetic risk factors for thrombosis to create Thrombo inCode-Oncology (TiC-Onco) for solid tumors. This assessment included four genetic variants known to increase the risk of thromboembolic events in cancer patients, as well as the clinical risk factors of body mass index, family history of thrombosis, primary tumor site, and tumor stage.
Dr. Pascual and colleagues developed a unique risk factor model that combined both the ThroLy and TiC-Onco elements.
In 208 patients with lymphoma who were not receiving anticoagulant treatment, researchers identified five clinical factors that were most predictive of venous thrombosis: a history of thrombosis, immobilization for more than 3 days, lymphoma type, Ann Arbor score for lymphoma stage, and mediastinal extension.
They combined these clinical risk factors with the genetic risk factors from the TiC-Onco score to develop the TiC-Onco–associated lymphoma score (TiC-Lympho).
When validated in the same group of patients, the TiC-Lympho score had a sensitivity of 93.55%, a specificity of 54.49%, positive predictive value of 26.36%, and negative predictive value of 97.94%.
The researchers also compared TiC-Lympho’s performance with that of the ThroLy and TiC-Onco models, and found it performed better on sensitivity and negative predictive value. The area under the curve for TiC-Lympho (0.783) was significantly higher than that seen with the other two risk models.
Session chair Kate Burbury, MBBS, of the Peter MacCallum Cancer Centre in Melbourne, raised the question of how the score – and particularly the genetic risk factor assessment – might be applied in the real-world clinical setting.
In an interview, Dr. Pascual said the findings represented preliminary data only, so the model was not ready to be applied to clinical practice yet. She also stressed that this was based on retrospective data, and needed to be further validated in other cohorts of lymphoma patients.
No conflicts of interest were reported.
SOURCE: Pascual C et al. 2019 ISTH Congress, Abstract OC 41.3.
MELBOURNE – A venous thromboembolism risk score that combines clinical risk factors, such as lymphoma type and stage, along with genetic variables, could offer a better way to predict venous thromboembolism in patients with lymphoma, according to new findings presented at the International Society on Thrombosis and Haemostasis congress.
Cristina Pascual, MD, of the Hospital Universitario Gregorio Marañon in Madrid presented data from a development and validation study of a clinical-genetic risk model for thrombosis in lymphoma in 208 patients with lymphoma, 31 of whom experienced a venous thromboembolic event.
While the relationship between cancer and increased thrombosis risk is well recognized, lymphoma patients are at particularly high risk, with an estimated thrombosis incidence of 5%-10%, Dr. Pascual said.
Currently, the Khorana score is the most validated risk score for thrombosis in patients with solid tumors, using factors such as tumor site, platelet and leukocyte count, hemoglobin levels, and body mass index. However, Dr. Pascual pointed out that just 10% of the validation cohort for the Khorana score were lymphoma patients, and it had previously been found to be not as useful for that population.
More recently, researchers had developed the ThroLy score for predicting thromboembolic events specifically in patients with lymphoma, incorporating clinical variables such as mediastinal involvement and extranodal localization.
Another group took a different approach by incorporating genetic risk factors for thrombosis to create Thrombo inCode-Oncology (TiC-Onco) for solid tumors. This assessment included four genetic variants known to increase the risk of thromboembolic events in cancer patients, as well as the clinical risk factors of body mass index, family history of thrombosis, primary tumor site, and tumor stage.
Dr. Pascual and colleagues developed a unique risk factor model that combined both the ThroLy and TiC-Onco elements.
In 208 patients with lymphoma who were not receiving anticoagulant treatment, researchers identified five clinical factors that were most predictive of venous thrombosis: a history of thrombosis, immobilization for more than 3 days, lymphoma type, Ann Arbor score for lymphoma stage, and mediastinal extension.
They combined these clinical risk factors with the genetic risk factors from the TiC-Onco score to develop the TiC-Onco–associated lymphoma score (TiC-Lympho).
When validated in the same group of patients, the TiC-Lympho score had a sensitivity of 93.55%, a specificity of 54.49%, positive predictive value of 26.36%, and negative predictive value of 97.94%.
The researchers also compared TiC-Lympho’s performance with that of the ThroLy and TiC-Onco models, and found it performed better on sensitivity and negative predictive value. The area under the curve for TiC-Lympho (0.783) was significantly higher than that seen with the other two risk models.
Session chair Kate Burbury, MBBS, of the Peter MacCallum Cancer Centre in Melbourne, raised the question of how the score – and particularly the genetic risk factor assessment – might be applied in the real-world clinical setting.
In an interview, Dr. Pascual said the findings represented preliminary data only, so the model was not ready to be applied to clinical practice yet. She also stressed that this was based on retrospective data, and needed to be further validated in other cohorts of lymphoma patients.
No conflicts of interest were reported.
SOURCE: Pascual C et al. 2019 ISTH Congress, Abstract OC 41.3.
MELBOURNE – A venous thromboembolism risk score that combines clinical risk factors, such as lymphoma type and stage, along with genetic variables, could offer a better way to predict venous thromboembolism in patients with lymphoma, according to new findings presented at the International Society on Thrombosis and Haemostasis congress.
Cristina Pascual, MD, of the Hospital Universitario Gregorio Marañon in Madrid presented data from a development and validation study of a clinical-genetic risk model for thrombosis in lymphoma in 208 patients with lymphoma, 31 of whom experienced a venous thromboembolic event.
While the relationship between cancer and increased thrombosis risk is well recognized, lymphoma patients are at particularly high risk, with an estimated thrombosis incidence of 5%-10%, Dr. Pascual said.
Currently, the Khorana score is the most validated risk score for thrombosis in patients with solid tumors, using factors such as tumor site, platelet and leukocyte count, hemoglobin levels, and body mass index. However, Dr. Pascual pointed out that just 10% of the validation cohort for the Khorana score were lymphoma patients, and it had previously been found to be not as useful for that population.
More recently, researchers had developed the ThroLy score for predicting thromboembolic events specifically in patients with lymphoma, incorporating clinical variables such as mediastinal involvement and extranodal localization.
Another group took a different approach by incorporating genetic risk factors for thrombosis to create Thrombo inCode-Oncology (TiC-Onco) for solid tumors. This assessment included four genetic variants known to increase the risk of thromboembolic events in cancer patients, as well as the clinical risk factors of body mass index, family history of thrombosis, primary tumor site, and tumor stage.
Dr. Pascual and colleagues developed a unique risk factor model that combined both the ThroLy and TiC-Onco elements.
In 208 patients with lymphoma who were not receiving anticoagulant treatment, researchers identified five clinical factors that were most predictive of venous thrombosis: a history of thrombosis, immobilization for more than 3 days, lymphoma type, Ann Arbor score for lymphoma stage, and mediastinal extension.
They combined these clinical risk factors with the genetic risk factors from the TiC-Onco score to develop the TiC-Onco–associated lymphoma score (TiC-Lympho).
When validated in the same group of patients, the TiC-Lympho score had a sensitivity of 93.55%, a specificity of 54.49%, positive predictive value of 26.36%, and negative predictive value of 97.94%.
The researchers also compared TiC-Lympho’s performance with that of the ThroLy and TiC-Onco models, and found it performed better on sensitivity and negative predictive value. The area under the curve for TiC-Lympho (0.783) was significantly higher than that seen with the other two risk models.
Session chair Kate Burbury, MBBS, of the Peter MacCallum Cancer Centre in Melbourne, raised the question of how the score – and particularly the genetic risk factor assessment – might be applied in the real-world clinical setting.
In an interview, Dr. Pascual said the findings represented preliminary data only, so the model was not ready to be applied to clinical practice yet. She also stressed that this was based on retrospective data, and needed to be further validated in other cohorts of lymphoma patients.
No conflicts of interest were reported.
SOURCE: Pascual C et al. 2019 ISTH Congress, Abstract OC 41.3.
REPORTING FROM 2019 ISTH CONGRESS
Concizumab looks feasible in hemophilia A and B treatment
MELBOURNE – A once-daily subcutaneous treatment that inhibits the tissue factor 4 pathway inhibitor has shown significant reductions in bleeding rates in patients with hemophilia A and B, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Jan Astermark, MD, PhD, of the Centre for Thrombosis and Haemostasis at Lund University in Sweden, presented data from two phase 2, dose-escalation trials of the monoclonal antibody concizumab.
The explorer 5 trial involved 36 adults with severe hemophilia A without inhibitors who were started on 0.15 mg/kg of concizumab for 24 weeks. If they experienced three or more bleeds during that time, they were escalated to 0.20 mg/kg, and then to 0.25mg/kg if they experienced an additional three bleeds. The initial 24-week treatment period was then extended by more than 52 weeks.
In the explorer 4 trial, 16 adults with hemophilia A and 10 adults with hemophilia B – all with inhibitors – were initially randomized 2:1 to either 24 weeks of 0.15mg/kg of concizumab, including a loading dose, or placebo, with similar dose escalation in response to breakthrough bleeds. After 24 weeks, the study continued with a 52-week extension, during which all patients were treated with concizumab.
Both studies saw reductions in bleeding rates associated with concizumab treatment.
In patients with hemophilia A and B with inhibitors, the mean annualized bleeding rate for all bleeds declined from 20.4 to 4.5 bleeds, spontaneous bleeds declined from 18.5 to 2.5, and joint bleeds declined from 15 to 3.2. All three of the reductions were statistically significant.
Almost all patients achieved a concizumab concentration of 100 ng/mL, which was the expected level based on data from the phase 1 trial. Some patients showed anticoncizumab antibodies, but these were transient and did not appear to have any effect on clinical outcomes, according to Dr. Astermark.
Most patients also reached a normal level of thrombin generation, although Dr. Astermark noted that there were some patients with hemophilia B with inhibitors who produced a lower amount of thrombin than normal.
Despite the increase in thrombin generation, there were no thromboembolic events, and no significant safety concerns emerged during the study, he reported.
“Importantly and interestingly, all patients completing the main phase went into the extension phase of this trial, indicating that it was something they think was a contribution to their treatment,” Dr. Astermark said.
Earlier in 2019, concizumab was granted breakthrough designation by the Food and Drug Administration for the treatment of patients with hemophilia B and inhibitors, allowing it to receive an accelerated review by the agency.
“What the FDA based their decision on was the B patients with inhibitors, because this is truly a group where we do not have so many options,” Dr. Astermark said in an interview. He also noted that the subcutaneous treatment, delivered via a pen-like device, was much more convenient for patients who, until now, had required repeated intravenous infusions.
Two phase 3 trials are now scheduled, in which patients will receive a higher loading dose than what was used in the phase 2 trials.
Novo Nordisk sponsored both studies. Dr. Astermark reported consultancies and research funding unrelated to the study.
SOURCE: Astermark J et al. 2019 ISTH Congress, Abstract LB 01.1.
MELBOURNE – A once-daily subcutaneous treatment that inhibits the tissue factor 4 pathway inhibitor has shown significant reductions in bleeding rates in patients with hemophilia A and B, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Jan Astermark, MD, PhD, of the Centre for Thrombosis and Haemostasis at Lund University in Sweden, presented data from two phase 2, dose-escalation trials of the monoclonal antibody concizumab.
The explorer 5 trial involved 36 adults with severe hemophilia A without inhibitors who were started on 0.15 mg/kg of concizumab for 24 weeks. If they experienced three or more bleeds during that time, they were escalated to 0.20 mg/kg, and then to 0.25mg/kg if they experienced an additional three bleeds. The initial 24-week treatment period was then extended by more than 52 weeks.
In the explorer 4 trial, 16 adults with hemophilia A and 10 adults with hemophilia B – all with inhibitors – were initially randomized 2:1 to either 24 weeks of 0.15mg/kg of concizumab, including a loading dose, or placebo, with similar dose escalation in response to breakthrough bleeds. After 24 weeks, the study continued with a 52-week extension, during which all patients were treated with concizumab.
Both studies saw reductions in bleeding rates associated with concizumab treatment.
In patients with hemophilia A and B with inhibitors, the mean annualized bleeding rate for all bleeds declined from 20.4 to 4.5 bleeds, spontaneous bleeds declined from 18.5 to 2.5, and joint bleeds declined from 15 to 3.2. All three of the reductions were statistically significant.
Almost all patients achieved a concizumab concentration of 100 ng/mL, which was the expected level based on data from the phase 1 trial. Some patients showed anticoncizumab antibodies, but these were transient and did not appear to have any effect on clinical outcomes, according to Dr. Astermark.
Most patients also reached a normal level of thrombin generation, although Dr. Astermark noted that there were some patients with hemophilia B with inhibitors who produced a lower amount of thrombin than normal.
Despite the increase in thrombin generation, there were no thromboembolic events, and no significant safety concerns emerged during the study, he reported.
“Importantly and interestingly, all patients completing the main phase went into the extension phase of this trial, indicating that it was something they think was a contribution to their treatment,” Dr. Astermark said.
Earlier in 2019, concizumab was granted breakthrough designation by the Food and Drug Administration for the treatment of patients with hemophilia B and inhibitors, allowing it to receive an accelerated review by the agency.
“What the FDA based their decision on was the B patients with inhibitors, because this is truly a group where we do not have so many options,” Dr. Astermark said in an interview. He also noted that the subcutaneous treatment, delivered via a pen-like device, was much more convenient for patients who, until now, had required repeated intravenous infusions.
Two phase 3 trials are now scheduled, in which patients will receive a higher loading dose than what was used in the phase 2 trials.
Novo Nordisk sponsored both studies. Dr. Astermark reported consultancies and research funding unrelated to the study.
SOURCE: Astermark J et al. 2019 ISTH Congress, Abstract LB 01.1.
MELBOURNE – A once-daily subcutaneous treatment that inhibits the tissue factor 4 pathway inhibitor has shown significant reductions in bleeding rates in patients with hemophilia A and B, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Jan Astermark, MD, PhD, of the Centre for Thrombosis and Haemostasis at Lund University in Sweden, presented data from two phase 2, dose-escalation trials of the monoclonal antibody concizumab.
The explorer 5 trial involved 36 adults with severe hemophilia A without inhibitors who were started on 0.15 mg/kg of concizumab for 24 weeks. If they experienced three or more bleeds during that time, they were escalated to 0.20 mg/kg, and then to 0.25mg/kg if they experienced an additional three bleeds. The initial 24-week treatment period was then extended by more than 52 weeks.
In the explorer 4 trial, 16 adults with hemophilia A and 10 adults with hemophilia B – all with inhibitors – were initially randomized 2:1 to either 24 weeks of 0.15mg/kg of concizumab, including a loading dose, or placebo, with similar dose escalation in response to breakthrough bleeds. After 24 weeks, the study continued with a 52-week extension, during which all patients were treated with concizumab.
Both studies saw reductions in bleeding rates associated with concizumab treatment.
In patients with hemophilia A and B with inhibitors, the mean annualized bleeding rate for all bleeds declined from 20.4 to 4.5 bleeds, spontaneous bleeds declined from 18.5 to 2.5, and joint bleeds declined from 15 to 3.2. All three of the reductions were statistically significant.
Almost all patients achieved a concizumab concentration of 100 ng/mL, which was the expected level based on data from the phase 1 trial. Some patients showed anticoncizumab antibodies, but these were transient and did not appear to have any effect on clinical outcomes, according to Dr. Astermark.
Most patients also reached a normal level of thrombin generation, although Dr. Astermark noted that there were some patients with hemophilia B with inhibitors who produced a lower amount of thrombin than normal.
Despite the increase in thrombin generation, there were no thromboembolic events, and no significant safety concerns emerged during the study, he reported.
“Importantly and interestingly, all patients completing the main phase went into the extension phase of this trial, indicating that it was something they think was a contribution to their treatment,” Dr. Astermark said.
Earlier in 2019, concizumab was granted breakthrough designation by the Food and Drug Administration for the treatment of patients with hemophilia B and inhibitors, allowing it to receive an accelerated review by the agency.
“What the FDA based their decision on was the B patients with inhibitors, because this is truly a group where we do not have so many options,” Dr. Astermark said in an interview. He also noted that the subcutaneous treatment, delivered via a pen-like device, was much more convenient for patients who, until now, had required repeated intravenous infusions.
Two phase 3 trials are now scheduled, in which patients will receive a higher loading dose than what was used in the phase 2 trials.
Novo Nordisk sponsored both studies. Dr. Astermark reported consultancies and research funding unrelated to the study.
SOURCE: Astermark J et al. 2019 ISTH Congress, Abstract LB 01.1.
REPORTING FROM 2019 ISTH CONGRESS
Acid-suppressing drug use associated with increased antiallergy drug use
Gastric acid–suppressing medications such as proton pump inhibitors are associated with a significant increase in subsequent antiallergy medication use, particularly in older individuals.
In a population-based study of health insurance data from 8.2 million people, Austrian researchers looked for prescriptions of gastric acid inhibitors, antiallergy drugs, or other commonly prescribed (lipid-modifying and antihypertensive) drugs as controls from 2009-2013.
According to results published in Nature Communications, gastric acid–suppressing drugs were associated with an overall 96% higher rate of subsequent prescriptions for antiallergy medications than among the general population not taking gastric acid–suppressing drugs (P less than .001). Among individuals aged 60 years or older, the rate of allergy medication prescriptions after acid-suppressing treatment was more than five times higher than that in the general population.
The rate of antiallergy medication use after acid-suppressing medication prescription was threefold higher than the rate seen after lipid-modifying or antihypertensive drug prescription.
“Our findings confirm an epidemiological association between gastric acid suppression and development of allergic symptoms, in line with previous mechanistic animal trials and human observational studies,” wrote Galateja Jordakieva, PhD, of the department of physical medicine, rehabilitation, and occupational medicine at the Medical University of Vienna, and coauthors.
All groups of acid-inhibiting medications were associated with a higher rate of subsequent antiallergy medication prescriptions. The only exception was prostaglandin E2 medications, but the authors said that here the numbers were too low to draw conclusions.
The hazard rate for antiallergy medications also increased with increasing numbers of daily doses of acid-suppressing medication, the study showed. The hazard rate for the lowest quartile – up to 20 daily doses per year – was a significant 28% higher than that of the general population, while the third quartile (68-213 daily dose per year) was associated with a 2.67-fold higher hazard. A similar increase was seen for the fourth quartile of acid suppression–medication dosing.
The authors established that just six daily doses of acid-suppressing drugs in a year were associated with significantly earlier prescriptions of antiallergy medication.
Stephen Evans, professor of pharmacoepidemiology at the London School of Hygiene & Tropical Medicine, emphasized in an associated commentary that the findings reflect association, not causation. “There are many possible explanations for the observed association ... In fact, the data make other explanations more than likely to be the cause of allergies.”
Coprescriptions of aspirin or nonsteroidal anti-inflammatory drugs (NSAIDs) are common among PPI users and “are among the drugs that are very well known to increase the risk of an allergic reaction.” The data show that antiallergy medicines are prescribed at a relatively similar rate at all ages, while PPI prescribing increases in the elderly.
“If the rate of prescription of PPIs shows a steep rise with age and they are a significant cause of allergies, then the anti-allergy medicines ... should also show a steep rise with age. The fact that they don’t, either means they show no relation or that any relation has a minimal effect on allergies,” Dr. Evans said. “Nearly all drugs can have very rare allergic reactions, including PPIs, but this paper does not help to show what the true rate is of these very rare reactions, or whether they are caused by PPIs alone. The design and analysis methods in the paper are likely to exaggerate their apparent occurrence.”
The study was supported by Burgenländische Gebietskrankenkasse and the Austrian Science Fund. No conflicts of interest were declared.
SOURCE: Jordakieva G et al. Nat Commun. 2019 Jul 30. doi: 10.1038/s41467-019-10914-6.
Gastric acid–suppressing medications such as proton pump inhibitors are associated with a significant increase in subsequent antiallergy medication use, particularly in older individuals.
In a population-based study of health insurance data from 8.2 million people, Austrian researchers looked for prescriptions of gastric acid inhibitors, antiallergy drugs, or other commonly prescribed (lipid-modifying and antihypertensive) drugs as controls from 2009-2013.
According to results published in Nature Communications, gastric acid–suppressing drugs were associated with an overall 96% higher rate of subsequent prescriptions for antiallergy medications than among the general population not taking gastric acid–suppressing drugs (P less than .001). Among individuals aged 60 years or older, the rate of allergy medication prescriptions after acid-suppressing treatment was more than five times higher than that in the general population.
The rate of antiallergy medication use after acid-suppressing medication prescription was threefold higher than the rate seen after lipid-modifying or antihypertensive drug prescription.
“Our findings confirm an epidemiological association between gastric acid suppression and development of allergic symptoms, in line with previous mechanistic animal trials and human observational studies,” wrote Galateja Jordakieva, PhD, of the department of physical medicine, rehabilitation, and occupational medicine at the Medical University of Vienna, and coauthors.
All groups of acid-inhibiting medications were associated with a higher rate of subsequent antiallergy medication prescriptions. The only exception was prostaglandin E2 medications, but the authors said that here the numbers were too low to draw conclusions.
The hazard rate for antiallergy medications also increased with increasing numbers of daily doses of acid-suppressing medication, the study showed. The hazard rate for the lowest quartile – up to 20 daily doses per year – was a significant 28% higher than that of the general population, while the third quartile (68-213 daily dose per year) was associated with a 2.67-fold higher hazard. A similar increase was seen for the fourth quartile of acid suppression–medication dosing.
The authors established that just six daily doses of acid-suppressing drugs in a year were associated with significantly earlier prescriptions of antiallergy medication.
Stephen Evans, professor of pharmacoepidemiology at the London School of Hygiene & Tropical Medicine, emphasized in an associated commentary that the findings reflect association, not causation. “There are many possible explanations for the observed association ... In fact, the data make other explanations more than likely to be the cause of allergies.”
Coprescriptions of aspirin or nonsteroidal anti-inflammatory drugs (NSAIDs) are common among PPI users and “are among the drugs that are very well known to increase the risk of an allergic reaction.” The data show that antiallergy medicines are prescribed at a relatively similar rate at all ages, while PPI prescribing increases in the elderly.
“If the rate of prescription of PPIs shows a steep rise with age and they are a significant cause of allergies, then the anti-allergy medicines ... should also show a steep rise with age. The fact that they don’t, either means they show no relation or that any relation has a minimal effect on allergies,” Dr. Evans said. “Nearly all drugs can have very rare allergic reactions, including PPIs, but this paper does not help to show what the true rate is of these very rare reactions, or whether they are caused by PPIs alone. The design and analysis methods in the paper are likely to exaggerate their apparent occurrence.”
The study was supported by Burgenländische Gebietskrankenkasse and the Austrian Science Fund. No conflicts of interest were declared.
SOURCE: Jordakieva G et al. Nat Commun. 2019 Jul 30. doi: 10.1038/s41467-019-10914-6.
Gastric acid–suppressing medications such as proton pump inhibitors are associated with a significant increase in subsequent antiallergy medication use, particularly in older individuals.
In a population-based study of health insurance data from 8.2 million people, Austrian researchers looked for prescriptions of gastric acid inhibitors, antiallergy drugs, or other commonly prescribed (lipid-modifying and antihypertensive) drugs as controls from 2009-2013.
According to results published in Nature Communications, gastric acid–suppressing drugs were associated with an overall 96% higher rate of subsequent prescriptions for antiallergy medications than among the general population not taking gastric acid–suppressing drugs (P less than .001). Among individuals aged 60 years or older, the rate of allergy medication prescriptions after acid-suppressing treatment was more than five times higher than that in the general population.
The rate of antiallergy medication use after acid-suppressing medication prescription was threefold higher than the rate seen after lipid-modifying or antihypertensive drug prescription.
“Our findings confirm an epidemiological association between gastric acid suppression and development of allergic symptoms, in line with previous mechanistic animal trials and human observational studies,” wrote Galateja Jordakieva, PhD, of the department of physical medicine, rehabilitation, and occupational medicine at the Medical University of Vienna, and coauthors.
All groups of acid-inhibiting medications were associated with a higher rate of subsequent antiallergy medication prescriptions. The only exception was prostaglandin E2 medications, but the authors said that here the numbers were too low to draw conclusions.
The hazard rate for antiallergy medications also increased with increasing numbers of daily doses of acid-suppressing medication, the study showed. The hazard rate for the lowest quartile – up to 20 daily doses per year – was a significant 28% higher than that of the general population, while the third quartile (68-213 daily dose per year) was associated with a 2.67-fold higher hazard. A similar increase was seen for the fourth quartile of acid suppression–medication dosing.
The authors established that just six daily doses of acid-suppressing drugs in a year were associated with significantly earlier prescriptions of antiallergy medication.
Stephen Evans, professor of pharmacoepidemiology at the London School of Hygiene & Tropical Medicine, emphasized in an associated commentary that the findings reflect association, not causation. “There are many possible explanations for the observed association ... In fact, the data make other explanations more than likely to be the cause of allergies.”
Coprescriptions of aspirin or nonsteroidal anti-inflammatory drugs (NSAIDs) are common among PPI users and “are among the drugs that are very well known to increase the risk of an allergic reaction.” The data show that antiallergy medicines are prescribed at a relatively similar rate at all ages, while PPI prescribing increases in the elderly.
“If the rate of prescription of PPIs shows a steep rise with age and they are a significant cause of allergies, then the anti-allergy medicines ... should also show a steep rise with age. The fact that they don’t, either means they show no relation or that any relation has a minimal effect on allergies,” Dr. Evans said. “Nearly all drugs can have very rare allergic reactions, including PPIs, but this paper does not help to show what the true rate is of these very rare reactions, or whether they are caused by PPIs alone. The design and analysis methods in the paper are likely to exaggerate their apparent occurrence.”
The study was supported by Burgenländische Gebietskrankenkasse and the Austrian Science Fund. No conflicts of interest were declared.
SOURCE: Jordakieva G et al. Nat Commun. 2019 Jul 30. doi: 10.1038/s41467-019-10914-6.
FROM NATURE COMMUNICATIONS
No increase in UTI risk with SGLT-2 inhibitors
Sodium-glucose cotransporter 2 inhibitors do not appear to increase the risk of urinary tract infections, compared with other antidiabetic medications, new research has found.
In a paper published in Annals of Internal Medicine, researchers reported the outcomes of a population-based, propensity-matched cohort study in which the analyzed data from 235,730 individuals newly prescribed sodium-glucose cotransporter 2 (SGLT-2) inhibitors, dipeptidyl peptidase–4 (DPP-4) inhibitors, and glucagonlike peptide–1 receptor (GLP-1) agonists.
In the first cohort, comparing SGLT-2 inhibitors with DDP-4 inhibitors, there were no significant differences between the two groups in the incidence rate of severe urinary tract infections (UTIs) (adjusted hazard ratio, 0.98; 95% confidence interval, 0.68-1.41).
In the second cohort, which compared patients taking GLP-1 receptor agonists with those taking SGLT-2 inhibitors, researchers saw a slightly lower incidence of severe UTIs among individuals on SGLT-2 inhibitors (HR, 0.72; 95% CI, 0.53-0.99; P = .04).
The analysis also failed to find any evidence that SGLT-2 inhibitors were associated with an increase in the risk of hospitalization or outpatient treatment for a UTI.
Chintan V. Dave, PharmD – now at Rutgers University, New Brunswick, N.J. – and colleagues from Brigham and Women’s Hospital and Harvard Medical School, Boston, wrote that the findings have significant clinical implications.
“Patients who may be good candidates to receive SGLT-2 inhibitors for diabetes control but who have a history of recurrent UTIs may be precluded from being prescribed these agents; because UTIs are highly prevalent in patients with diabetes, this could exclude a substantial number of patients from receiving an entire class of medications that has been shown to decrease risk for major cardiovascular events and death,” they wrote.
The researchers stressed that “other factors beyond risk for UTI events should be considered in decisions about whether to prescribe SGLT-2 therapy for patients with diabetes.”
The authors did note that their study was subject to the usual limitations of observational studies, such as susceptibility to confounding, and was also limited to people with health insurance.
An accompanying editorial by Kristian B. Filion, PhD, and Oriana H. Yu, MD, of McGill University, Montreal, commented that the data provided reassuring, real-world evidence on the potential safety issue of UTI risk with SGLT-2 inhibitors.
However, they also stressed that the study excluded individuals at high risk or with a history of UTIs, and that these were subgroups who required further investigation.
The study was supported by the Harvard Medical School and the National Institute on Aging. Three authors reported support from private industry outside the submitted work.
SOURCE: Dave CV et al. Ann Intern Med. 2019 Jul 29. doi: 10.7326/M18-3136.
Sodium-glucose cotransporter 2 inhibitors do not appear to increase the risk of urinary tract infections, compared with other antidiabetic medications, new research has found.
In a paper published in Annals of Internal Medicine, researchers reported the outcomes of a population-based, propensity-matched cohort study in which the analyzed data from 235,730 individuals newly prescribed sodium-glucose cotransporter 2 (SGLT-2) inhibitors, dipeptidyl peptidase–4 (DPP-4) inhibitors, and glucagonlike peptide–1 receptor (GLP-1) agonists.
In the first cohort, comparing SGLT-2 inhibitors with DDP-4 inhibitors, there were no significant differences between the two groups in the incidence rate of severe urinary tract infections (UTIs) (adjusted hazard ratio, 0.98; 95% confidence interval, 0.68-1.41).
In the second cohort, which compared patients taking GLP-1 receptor agonists with those taking SGLT-2 inhibitors, researchers saw a slightly lower incidence of severe UTIs among individuals on SGLT-2 inhibitors (HR, 0.72; 95% CI, 0.53-0.99; P = .04).
The analysis also failed to find any evidence that SGLT-2 inhibitors were associated with an increase in the risk of hospitalization or outpatient treatment for a UTI.
Chintan V. Dave, PharmD – now at Rutgers University, New Brunswick, N.J. – and colleagues from Brigham and Women’s Hospital and Harvard Medical School, Boston, wrote that the findings have significant clinical implications.
“Patients who may be good candidates to receive SGLT-2 inhibitors for diabetes control but who have a history of recurrent UTIs may be precluded from being prescribed these agents; because UTIs are highly prevalent in patients with diabetes, this could exclude a substantial number of patients from receiving an entire class of medications that has been shown to decrease risk for major cardiovascular events and death,” they wrote.
The researchers stressed that “other factors beyond risk for UTI events should be considered in decisions about whether to prescribe SGLT-2 therapy for patients with diabetes.”
The authors did note that their study was subject to the usual limitations of observational studies, such as susceptibility to confounding, and was also limited to people with health insurance.
An accompanying editorial by Kristian B. Filion, PhD, and Oriana H. Yu, MD, of McGill University, Montreal, commented that the data provided reassuring, real-world evidence on the potential safety issue of UTI risk with SGLT-2 inhibitors.
However, they also stressed that the study excluded individuals at high risk or with a history of UTIs, and that these were subgroups who required further investigation.
The study was supported by the Harvard Medical School and the National Institute on Aging. Three authors reported support from private industry outside the submitted work.
SOURCE: Dave CV et al. Ann Intern Med. 2019 Jul 29. doi: 10.7326/M18-3136.
Sodium-glucose cotransporter 2 inhibitors do not appear to increase the risk of urinary tract infections, compared with other antidiabetic medications, new research has found.
In a paper published in Annals of Internal Medicine, researchers reported the outcomes of a population-based, propensity-matched cohort study in which the analyzed data from 235,730 individuals newly prescribed sodium-glucose cotransporter 2 (SGLT-2) inhibitors, dipeptidyl peptidase–4 (DPP-4) inhibitors, and glucagonlike peptide–1 receptor (GLP-1) agonists.
In the first cohort, comparing SGLT-2 inhibitors with DDP-4 inhibitors, there were no significant differences between the two groups in the incidence rate of severe urinary tract infections (UTIs) (adjusted hazard ratio, 0.98; 95% confidence interval, 0.68-1.41).
In the second cohort, which compared patients taking GLP-1 receptor agonists with those taking SGLT-2 inhibitors, researchers saw a slightly lower incidence of severe UTIs among individuals on SGLT-2 inhibitors (HR, 0.72; 95% CI, 0.53-0.99; P = .04).
The analysis also failed to find any evidence that SGLT-2 inhibitors were associated with an increase in the risk of hospitalization or outpatient treatment for a UTI.
Chintan V. Dave, PharmD – now at Rutgers University, New Brunswick, N.J. – and colleagues from Brigham and Women’s Hospital and Harvard Medical School, Boston, wrote that the findings have significant clinical implications.
“Patients who may be good candidates to receive SGLT-2 inhibitors for diabetes control but who have a history of recurrent UTIs may be precluded from being prescribed these agents; because UTIs are highly prevalent in patients with diabetes, this could exclude a substantial number of patients from receiving an entire class of medications that has been shown to decrease risk for major cardiovascular events and death,” they wrote.
The researchers stressed that “other factors beyond risk for UTI events should be considered in decisions about whether to prescribe SGLT-2 therapy for patients with diabetes.”
The authors did note that their study was subject to the usual limitations of observational studies, such as susceptibility to confounding, and was also limited to people with health insurance.
An accompanying editorial by Kristian B. Filion, PhD, and Oriana H. Yu, MD, of McGill University, Montreal, commented that the data provided reassuring, real-world evidence on the potential safety issue of UTI risk with SGLT-2 inhibitors.
However, they also stressed that the study excluded individuals at high risk or with a history of UTIs, and that these were subgroups who required further investigation.
The study was supported by the Harvard Medical School and the National Institute on Aging. Three authors reported support from private industry outside the submitted work.
SOURCE: Dave CV et al. Ann Intern Med. 2019 Jul 29. doi: 10.7326/M18-3136.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Sodium-glucose cotransporter 2 inhibitors are not associated with a greater risk of urinary tract infections, compared with dipeptidyl peptidase-4 inhibitors or glucagonlike peptide–1 receptor agonists.
Major finding: The incidence of severe urinary tract infections is similar among patients taking sodium-glucose cotransporter 2 inhibitors, dipeptidyl peptidase–4, inhibitors or glucagonlike peptide–1 receptor agonists.
Study details: A population-based, propensity-matched cohort study in 235,730 individuals with type 2 diabetes.
Disclosures: The study was supported by the Harvard Medical School and the National Institute on Aging. Three authors reported support from private industry outside the submitted work.
Source: Dave CV et al. Ann Intern Med. 2019 Jul 29. doi: 10.7326/M18-3136.
RNA interference drug fitusiran looks effective in both hemophilia A and B
MELBOURNE – An investigational RNA interference therapeutic that suppresses the production of antithrombin has shown significant reductions in bleeding rates with no major safety events, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Fitusiran is a once-monthly, fixed-dose subcutaneous therapy that uses RNA interference to silence the gene for the endogenous anticoagulant antithrombin.
“The therapeutic hypothesis is based on the fact that hemophilia A and B are essentially thrombin-deficiency disorders, so if we lack factor VIII or factor IX, we can’t generate enough thrombin and we can’t produce a significant and substantial blood clot,” John Pasi, MBChB, PhD, of the Royal London Haemophilia Centre, Barts Health NHS Trust. “If we, however, administer fitusiran, which will suppress antithrombin production, we can rebalance coagulation, generate more thrombin and form a much more substantial clot.”
Dr. Pasi presented results of an interim analysis of safety and efficacy data from an open-label, phase 2 extension study in 34 individuals with hemophilia A or B, with or without inhibitors, who were treated either with 50-mg or 80-mg doses of fitusiran for a median of at least 2 years.
Researchers saw significant declines in annualized bleeding rates in patients with hemophilia A and B, with and without inhibitors. Among those without inhibitors, the median annualized bleeding rate declined from 2.00 in patients already on hemophilia prophylaxis and 12.00 in those using on-demand treatment to 1.08 overall. In patients with inhibitors, the median annualized bleeding rate dropped from 42.00 to 1.04.
The treatment was also associated with substantial reductions in antithrombin production and increases in thrombin generation.
One patient in the phase 1 study experienced a fatal cerebral venous sinus thrombosis, which subsequently led to introduction of a bleed management protocol.
“Following that last case, we revised and reviewed the bleed management guidelines in view of the fact that there might potentially be an interaction between the amount of replacement therapy and thrombin generation,” Dr. Pasi said. Since introduction of that protocol, there have been no related thrombotic events.
The majority of adverse events reported were mild and deemed not related to the study drug, Dr. Pasi said. These included headache, injection site erythema, and arthralgia. A total of 14 subjects – all of whom were positive for hepatitis C at baseline – experienced rises in ALT levels but these were asymptomatic and resolved spontaneously.
One patient with chronic active hepatitis C infection also showed significant ALT/AST elevation which led to discontinuation of treatment.
In an interview, Dr. Pasi said one of the biggest advantages of fitusiran was that it could be used in patients with hemophilia A and B. “You’ve got patients with hemophilia B who’ve got no options at the moment. That would be an obvious specific group that would gain from this.”
Another advantage was fitusiran’s stability and dosing, he said, pointing out that the treatment was fixed dosing and stable at room temperature. Fitusiran is now undergoing phase 3 trials.
The study was funded by Sanofi Genzyme and Alnylam Pharmaceuticals, and six authors were employees of Sanofi Genzyme. Dr. Pasi reported financial relationships with pharmaceutical companies, including Alnylam.
SOURCE: Pasi J et al. 2019 ISTH Congress, Abstract OC 11.3.
MELBOURNE – An investigational RNA interference therapeutic that suppresses the production of antithrombin has shown significant reductions in bleeding rates with no major safety events, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Fitusiran is a once-monthly, fixed-dose subcutaneous therapy that uses RNA interference to silence the gene for the endogenous anticoagulant antithrombin.
“The therapeutic hypothesis is based on the fact that hemophilia A and B are essentially thrombin-deficiency disorders, so if we lack factor VIII or factor IX, we can’t generate enough thrombin and we can’t produce a significant and substantial blood clot,” John Pasi, MBChB, PhD, of the Royal London Haemophilia Centre, Barts Health NHS Trust. “If we, however, administer fitusiran, which will suppress antithrombin production, we can rebalance coagulation, generate more thrombin and form a much more substantial clot.”
Dr. Pasi presented results of an interim analysis of safety and efficacy data from an open-label, phase 2 extension study in 34 individuals with hemophilia A or B, with or without inhibitors, who were treated either with 50-mg or 80-mg doses of fitusiran for a median of at least 2 years.
Researchers saw significant declines in annualized bleeding rates in patients with hemophilia A and B, with and without inhibitors. Among those without inhibitors, the median annualized bleeding rate declined from 2.00 in patients already on hemophilia prophylaxis and 12.00 in those using on-demand treatment to 1.08 overall. In patients with inhibitors, the median annualized bleeding rate dropped from 42.00 to 1.04.
The treatment was also associated with substantial reductions in antithrombin production and increases in thrombin generation.
One patient in the phase 1 study experienced a fatal cerebral venous sinus thrombosis, which subsequently led to introduction of a bleed management protocol.
“Following that last case, we revised and reviewed the bleed management guidelines in view of the fact that there might potentially be an interaction between the amount of replacement therapy and thrombin generation,” Dr. Pasi said. Since introduction of that protocol, there have been no related thrombotic events.
The majority of adverse events reported were mild and deemed not related to the study drug, Dr. Pasi said. These included headache, injection site erythema, and arthralgia. A total of 14 subjects – all of whom were positive for hepatitis C at baseline – experienced rises in ALT levels but these were asymptomatic and resolved spontaneously.
One patient with chronic active hepatitis C infection also showed significant ALT/AST elevation which led to discontinuation of treatment.
In an interview, Dr. Pasi said one of the biggest advantages of fitusiran was that it could be used in patients with hemophilia A and B. “You’ve got patients with hemophilia B who’ve got no options at the moment. That would be an obvious specific group that would gain from this.”
Another advantage was fitusiran’s stability and dosing, he said, pointing out that the treatment was fixed dosing and stable at room temperature. Fitusiran is now undergoing phase 3 trials.
The study was funded by Sanofi Genzyme and Alnylam Pharmaceuticals, and six authors were employees of Sanofi Genzyme. Dr. Pasi reported financial relationships with pharmaceutical companies, including Alnylam.
SOURCE: Pasi J et al. 2019 ISTH Congress, Abstract OC 11.3.
MELBOURNE – An investigational RNA interference therapeutic that suppresses the production of antithrombin has shown significant reductions in bleeding rates with no major safety events, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Fitusiran is a once-monthly, fixed-dose subcutaneous therapy that uses RNA interference to silence the gene for the endogenous anticoagulant antithrombin.
“The therapeutic hypothesis is based on the fact that hemophilia A and B are essentially thrombin-deficiency disorders, so if we lack factor VIII or factor IX, we can’t generate enough thrombin and we can’t produce a significant and substantial blood clot,” John Pasi, MBChB, PhD, of the Royal London Haemophilia Centre, Barts Health NHS Trust. “If we, however, administer fitusiran, which will suppress antithrombin production, we can rebalance coagulation, generate more thrombin and form a much more substantial clot.”
Dr. Pasi presented results of an interim analysis of safety and efficacy data from an open-label, phase 2 extension study in 34 individuals with hemophilia A or B, with or without inhibitors, who were treated either with 50-mg or 80-mg doses of fitusiran for a median of at least 2 years.
Researchers saw significant declines in annualized bleeding rates in patients with hemophilia A and B, with and without inhibitors. Among those without inhibitors, the median annualized bleeding rate declined from 2.00 in patients already on hemophilia prophylaxis and 12.00 in those using on-demand treatment to 1.08 overall. In patients with inhibitors, the median annualized bleeding rate dropped from 42.00 to 1.04.
The treatment was also associated with substantial reductions in antithrombin production and increases in thrombin generation.
One patient in the phase 1 study experienced a fatal cerebral venous sinus thrombosis, which subsequently led to introduction of a bleed management protocol.
“Following that last case, we revised and reviewed the bleed management guidelines in view of the fact that there might potentially be an interaction between the amount of replacement therapy and thrombin generation,” Dr. Pasi said. Since introduction of that protocol, there have been no related thrombotic events.
The majority of adverse events reported were mild and deemed not related to the study drug, Dr. Pasi said. These included headache, injection site erythema, and arthralgia. A total of 14 subjects – all of whom were positive for hepatitis C at baseline – experienced rises in ALT levels but these were asymptomatic and resolved spontaneously.
One patient with chronic active hepatitis C infection also showed significant ALT/AST elevation which led to discontinuation of treatment.
In an interview, Dr. Pasi said one of the biggest advantages of fitusiran was that it could be used in patients with hemophilia A and B. “You’ve got patients with hemophilia B who’ve got no options at the moment. That would be an obvious specific group that would gain from this.”
Another advantage was fitusiran’s stability and dosing, he said, pointing out that the treatment was fixed dosing and stable at room temperature. Fitusiran is now undergoing phase 3 trials.
The study was funded by Sanofi Genzyme and Alnylam Pharmaceuticals, and six authors were employees of Sanofi Genzyme. Dr. Pasi reported financial relationships with pharmaceutical companies, including Alnylam.
SOURCE: Pasi J et al. 2019 ISTH Congress, Abstract OC 11.3.
REPORTING FROM 2019 ISTH CONGRESS
Early phase trial shows durable responses to gene therapy for hemophilia A
MELBOURNE – A gene therapy treatment for hemophilia A has shown sustained reductions in bleeding rates 3 years after treatment, with no major safety issues, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Valoctocogene roxaparvovec is an investigational gene therapy that involves using an adenovirus-associated virus to deliver the gene for clotting factor VIII.
John Pasi, MBChB, PhD, of the Royal London Haemophilia Centre, Barts Health NHS Trust, presented the 3-year efficacy and safety results from the phase 1/2 trial of the therapy, involving 15 men with hemophilia A without inhibitors who received a single intravenous dose – either 4 x 1013 vector genomes (vg) per kg or 6 x 1013 vg/kg – of the therapy.
Participants’ mean annualized bleeding rate at baseline ranged from 6.5 among men who had been receiving prophylactic therapy to 25 among those who had been historically been treated on demand.
The treatment was associated with a substantial, significant reduction in mean annualized bleed rates; a 96% reduction in the 6 x 1013 vg/kg group by year 3, and 92% reduction in the 4 x 1013 vg/kg group by year 2.
By year 3, 86% of patients in the higher dose group had not experienced a bleed in the prior 12 months, all patients were off prophylaxis, and all had experienced resolution of target joints.
Mean factor VIII usage also decreased significantly, with a 96% reduction by year 3 in the higher dose cohort, and a 97% reduction by year 2 in the lower dose cohort.
The study also showed significant improvements in quality of life across all domains, Dr. Pasi reported.
There were no significant safety concerns raised during the study. Several patients experienced mild to moderate, transient rises in alanine aminotransferase levels at around 8-16 weeks after treatment, but there was no significant impact on liver function or on corticosteroid use. Two patients reported mild infusion reactions, which resolved within 48 hours with altering treatment.
The researchers also examined durability of factor VIII activity levels following the gene therapy, which was monitored using chromogenic assays. This revealed that after the initial increase following therapy, the factor VIII levels plateaued between years 2 and 3.
“We’ve got what we feel is really good clinical evidence of a persistent effect and we think this is dramatic,” Dr. Pasi said. A phase 3 trial is now underway.
A commenter from the audience, who remarked that the data were incredible and would make a huge difference for patients, asked about whether this represented a possible cure for the disease.
It’s premature to talk about a cure, Dr. Pasi said.
“It’s like watching paint dry; it’s going to take years before we know where we are,” he said in an interview.
However, this could represent massive and transformational change in the management of hemophilia A, he added.
On the question of whether this approach might also work in patients with inhibitors, Dr. Pasi said there were animal data suggesting that gene therapy could work in individuals with inhibitors, but the focus for the moment was on patients without inhibitors.
“But for patients that previously had a history of inhibitors and are now tolerant, that’s quite a significant group of patients that we were going to have to think about how we deal with that in due course,” he said.
The study was sponsored by manufacturer BioMarin Pharmaceutical. Dr. Pasi reported financial relationships with the study sponsor and other companies.
SOURCE: Pasi KJ et al. 2019 ISTH Congress, Abstract LB 01.2.
MELBOURNE – A gene therapy treatment for hemophilia A has shown sustained reductions in bleeding rates 3 years after treatment, with no major safety issues, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Valoctocogene roxaparvovec is an investigational gene therapy that involves using an adenovirus-associated virus to deliver the gene for clotting factor VIII.
John Pasi, MBChB, PhD, of the Royal London Haemophilia Centre, Barts Health NHS Trust, presented the 3-year efficacy and safety results from the phase 1/2 trial of the therapy, involving 15 men with hemophilia A without inhibitors who received a single intravenous dose – either 4 x 1013 vector genomes (vg) per kg or 6 x 1013 vg/kg – of the therapy.
Participants’ mean annualized bleeding rate at baseline ranged from 6.5 among men who had been receiving prophylactic therapy to 25 among those who had been historically been treated on demand.
The treatment was associated with a substantial, significant reduction in mean annualized bleed rates; a 96% reduction in the 6 x 1013 vg/kg group by year 3, and 92% reduction in the 4 x 1013 vg/kg group by year 2.
By year 3, 86% of patients in the higher dose group had not experienced a bleed in the prior 12 months, all patients were off prophylaxis, and all had experienced resolution of target joints.
Mean factor VIII usage also decreased significantly, with a 96% reduction by year 3 in the higher dose cohort, and a 97% reduction by year 2 in the lower dose cohort.
The study also showed significant improvements in quality of life across all domains, Dr. Pasi reported.
There were no significant safety concerns raised during the study. Several patients experienced mild to moderate, transient rises in alanine aminotransferase levels at around 8-16 weeks after treatment, but there was no significant impact on liver function or on corticosteroid use. Two patients reported mild infusion reactions, which resolved within 48 hours with altering treatment.
The researchers also examined durability of factor VIII activity levels following the gene therapy, which was monitored using chromogenic assays. This revealed that after the initial increase following therapy, the factor VIII levels plateaued between years 2 and 3.
“We’ve got what we feel is really good clinical evidence of a persistent effect and we think this is dramatic,” Dr. Pasi said. A phase 3 trial is now underway.
A commenter from the audience, who remarked that the data were incredible and would make a huge difference for patients, asked about whether this represented a possible cure for the disease.
It’s premature to talk about a cure, Dr. Pasi said.
“It’s like watching paint dry; it’s going to take years before we know where we are,” he said in an interview.
However, this could represent massive and transformational change in the management of hemophilia A, he added.
On the question of whether this approach might also work in patients with inhibitors, Dr. Pasi said there were animal data suggesting that gene therapy could work in individuals with inhibitors, but the focus for the moment was on patients without inhibitors.
“But for patients that previously had a history of inhibitors and are now tolerant, that’s quite a significant group of patients that we were going to have to think about how we deal with that in due course,” he said.
The study was sponsored by manufacturer BioMarin Pharmaceutical. Dr. Pasi reported financial relationships with the study sponsor and other companies.
SOURCE: Pasi KJ et al. 2019 ISTH Congress, Abstract LB 01.2.
MELBOURNE – A gene therapy treatment for hemophilia A has shown sustained reductions in bleeding rates 3 years after treatment, with no major safety issues, according to findings presented at the International Society on Thrombosis and Haemostasis congress.
Valoctocogene roxaparvovec is an investigational gene therapy that involves using an adenovirus-associated virus to deliver the gene for clotting factor VIII.
John Pasi, MBChB, PhD, of the Royal London Haemophilia Centre, Barts Health NHS Trust, presented the 3-year efficacy and safety results from the phase 1/2 trial of the therapy, involving 15 men with hemophilia A without inhibitors who received a single intravenous dose – either 4 x 1013 vector genomes (vg) per kg or 6 x 1013 vg/kg – of the therapy.
Participants’ mean annualized bleeding rate at baseline ranged from 6.5 among men who had been receiving prophylactic therapy to 25 among those who had been historically been treated on demand.
The treatment was associated with a substantial, significant reduction in mean annualized bleed rates; a 96% reduction in the 6 x 1013 vg/kg group by year 3, and 92% reduction in the 4 x 1013 vg/kg group by year 2.
By year 3, 86% of patients in the higher dose group had not experienced a bleed in the prior 12 months, all patients were off prophylaxis, and all had experienced resolution of target joints.
Mean factor VIII usage also decreased significantly, with a 96% reduction by year 3 in the higher dose cohort, and a 97% reduction by year 2 in the lower dose cohort.
The study also showed significant improvements in quality of life across all domains, Dr. Pasi reported.
There were no significant safety concerns raised during the study. Several patients experienced mild to moderate, transient rises in alanine aminotransferase levels at around 8-16 weeks after treatment, but there was no significant impact on liver function or on corticosteroid use. Two patients reported mild infusion reactions, which resolved within 48 hours with altering treatment.
The researchers also examined durability of factor VIII activity levels following the gene therapy, which was monitored using chromogenic assays. This revealed that after the initial increase following therapy, the factor VIII levels plateaued between years 2 and 3.
“We’ve got what we feel is really good clinical evidence of a persistent effect and we think this is dramatic,” Dr. Pasi said. A phase 3 trial is now underway.
A commenter from the audience, who remarked that the data were incredible and would make a huge difference for patients, asked about whether this represented a possible cure for the disease.
It’s premature to talk about a cure, Dr. Pasi said.
“It’s like watching paint dry; it’s going to take years before we know where we are,” he said in an interview.
However, this could represent massive and transformational change in the management of hemophilia A, he added.
On the question of whether this approach might also work in patients with inhibitors, Dr. Pasi said there were animal data suggesting that gene therapy could work in individuals with inhibitors, but the focus for the moment was on patients without inhibitors.
“But for patients that previously had a history of inhibitors and are now tolerant, that’s quite a significant group of patients that we were going to have to think about how we deal with that in due course,” he said.
The study was sponsored by manufacturer BioMarin Pharmaceutical. Dr. Pasi reported financial relationships with the study sponsor and other companies.
SOURCE: Pasi KJ et al. 2019 ISTH Congress, Abstract LB 01.2.
REPORTING FROM 2019 ISTH CONGRESS