User login
Rachel Whitmer, PhD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Rachel Whitmer, PhD
VIDEO: Biogels boost targeted delivery of growth factor in severe wounds
DANA POINT, CALIF.– Materials science meets medicine. That’s the new frontier for many specialties, thanks to advances in the understanding of native tissue dynamics and how bioengineered materials will perform in vivo.
Sarah Heilshorn, Ph.D., of the department of materials science and engineering and the department of bioengineering at Stanford (Calif.) University, was one of several presenters at this year’s Summit in Aesthetic Medicine, with a focus on the use of synthetic materials for reconstructive surgery and treatment for acute and traumatic tissue injury.
In this video, Dr. Heilshorn explains how materials science and bioengineering are promoting angiogenesis to help patients who have suffered spinal injuries, chronic wounds, and conditions involving severe tissue injury at the meeting held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications. She describes how revascularization, bone regeneration, and tissue growth are all possible through the use of highly specific and targeted biogels injected into the body to improve delivery of growth factors.
Dr. Heilshorn said she had no relevant financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
DANA POINT, CALIF.– Materials science meets medicine. That’s the new frontier for many specialties, thanks to advances in the understanding of native tissue dynamics and how bioengineered materials will perform in vivo.
Sarah Heilshorn, Ph.D., of the department of materials science and engineering and the department of bioengineering at Stanford (Calif.) University, was one of several presenters at this year’s Summit in Aesthetic Medicine, with a focus on the use of synthetic materials for reconstructive surgery and treatment for acute and traumatic tissue injury.
In this video, Dr. Heilshorn explains how materials science and bioengineering are promoting angiogenesis to help patients who have suffered spinal injuries, chronic wounds, and conditions involving severe tissue injury at the meeting held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications. She describes how revascularization, bone regeneration, and tissue growth are all possible through the use of highly specific and targeted biogels injected into the body to improve delivery of growth factors.
Dr. Heilshorn said she had no relevant financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
DANA POINT, CALIF.– Materials science meets medicine. That’s the new frontier for many specialties, thanks to advances in the understanding of native tissue dynamics and how bioengineered materials will perform in vivo.
Sarah Heilshorn, Ph.D., of the department of materials science and engineering and the department of bioengineering at Stanford (Calif.) University, was one of several presenters at this year’s Summit in Aesthetic Medicine, with a focus on the use of synthetic materials for reconstructive surgery and treatment for acute and traumatic tissue injury.
In this video, Dr. Heilshorn explains how materials science and bioengineering are promoting angiogenesis to help patients who have suffered spinal injuries, chronic wounds, and conditions involving severe tissue injury at the meeting held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications. She describes how revascularization, bone regeneration, and tissue growth are all possible through the use of highly specific and targeted biogels injected into the body to improve delivery of growth factors.
Dr. Heilshorn said she had no relevant financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM THE SUMMIT IN AESTHETIC MEDICINE 2014
VIDEO: Late-life hypertension may reduce dementia risk
COPENHAGEN – When it comes to treatment of hypertension, perhaps not all age groups should be treated equally, according to findings from a study of very elderly patients.
The 90+ Study, which analyzed data from 625 dementia-free participants, showed that developing high blood pressure in the 80s and 90s was associated with a reduced risk of developing dementia, researchers reported at the Alzheimer’s Association’s International Conference.
They also found that as the participants’ blood pressure level increased, their risk of dementia decreased. Results remained the same after researchers adjusted for hypertension medication and history of stroke or heart disease.
The findings are important because people who are in their 80s and 90s are among the fastest growing segments of the U.S. population, said Maria M. Corrada, Sc.D., an epidemiologist at the University of California, Irvine.
In a video interview, Dr. Corrada explains the research findings and the implications for clinical practice.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
COPENHAGEN – When it comes to treatment of hypertension, perhaps not all age groups should be treated equally, according to findings from a study of very elderly patients.
The 90+ Study, which analyzed data from 625 dementia-free participants, showed that developing high blood pressure in the 80s and 90s was associated with a reduced risk of developing dementia, researchers reported at the Alzheimer’s Association’s International Conference.
They also found that as the participants’ blood pressure level increased, their risk of dementia decreased. Results remained the same after researchers adjusted for hypertension medication and history of stroke or heart disease.
The findings are important because people who are in their 80s and 90s are among the fastest growing segments of the U.S. population, said Maria M. Corrada, Sc.D., an epidemiologist at the University of California, Irvine.
In a video interview, Dr. Corrada explains the research findings and the implications for clinical practice.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
COPENHAGEN – When it comes to treatment of hypertension, perhaps not all age groups should be treated equally, according to findings from a study of very elderly patients.
The 90+ Study, which analyzed data from 625 dementia-free participants, showed that developing high blood pressure in the 80s and 90s was associated with a reduced risk of developing dementia, researchers reported at the Alzheimer’s Association’s International Conference.
They also found that as the participants’ blood pressure level increased, their risk of dementia decreased. Results remained the same after researchers adjusted for hypertension medication and history of stroke or heart disease.
The findings are important because people who are in their 80s and 90s are among the fastest growing segments of the U.S. population, said Maria M. Corrada, Sc.D., an epidemiologist at the University of California, Irvine.
In a video interview, Dr. Corrada explains the research findings and the implications for clinical practice.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
AT AAIC 2014
VIDEO: Lifestyle improvements prevented cognitive decline in at-risk elderly
COPENHAGEN – When older adults who were at risk of Alzheimer’s disease adopted more healthful lifestyles, including diet and exercise, their cognitive performance improved, according to findings from a 2-year randomized, controlled trial.
The study is yet more proof that "it’s never too late to adopt a healthful lifestyle," said Dr. Ralph A. Nixon, chair of the Alzheimer’s Association Medical and Scientific Advisory Council.
The multimodal intervention study, called the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER), added nutritional guidance, physical exercise, cognitive training, and vascular risk factor management to the treatment of half of 1,260 participants who were between ages 60 and 77 years. The other half of participants received regular health advice.
To their surprise, researchers achieved their primary outcome – improvement in cognitive performance – within 2 years. In fact, the benefit was significant in all cognitive domains, including memory, executive function, and psychomotor speed, compared with the control group.
Only 11% of the participants dropped out. There were no serious adverse events.
This is the first long-term study to assess the impact of simultaneous improvement of several lifestyle factors on elderly cognition, the investigators said. The researchers are now conducting cost analysis, and they plan to follow the patients for 7 years to assess the longer-term effect of the interventions.
The findings demonstrate that multidomain intervention is feasible and can reduce the risk of cognitive impairment among the at-risk elderly population, said the study’s lead investigator, Dr. Miia Kivipelto. She hopes that this proof-of-concept study becomes a model for future programs in this group.
In a video interview, Dr. Kivipelto, a professor at the Karolinska Institute in Stockholm, discusses the study’s findings and its implications for clinical practice.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
COPENHAGEN – When older adults who were at risk of Alzheimer’s disease adopted more healthful lifestyles, including diet and exercise, their cognitive performance improved, according to findings from a 2-year randomized, controlled trial.
The study is yet more proof that "it’s never too late to adopt a healthful lifestyle," said Dr. Ralph A. Nixon, chair of the Alzheimer’s Association Medical and Scientific Advisory Council.
The multimodal intervention study, called the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER), added nutritional guidance, physical exercise, cognitive training, and vascular risk factor management to the treatment of half of 1,260 participants who were between ages 60 and 77 years. The other half of participants received regular health advice.
To their surprise, researchers achieved their primary outcome – improvement in cognitive performance – within 2 years. In fact, the benefit was significant in all cognitive domains, including memory, executive function, and psychomotor speed, compared with the control group.
Only 11% of the participants dropped out. There were no serious adverse events.
This is the first long-term study to assess the impact of simultaneous improvement of several lifestyle factors on elderly cognition, the investigators said. The researchers are now conducting cost analysis, and they plan to follow the patients for 7 years to assess the longer-term effect of the interventions.
The findings demonstrate that multidomain intervention is feasible and can reduce the risk of cognitive impairment among the at-risk elderly population, said the study’s lead investigator, Dr. Miia Kivipelto. She hopes that this proof-of-concept study becomes a model for future programs in this group.
In a video interview, Dr. Kivipelto, a professor at the Karolinska Institute in Stockholm, discusses the study’s findings and its implications for clinical practice.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
COPENHAGEN – When older adults who were at risk of Alzheimer’s disease adopted more healthful lifestyles, including diet and exercise, their cognitive performance improved, according to findings from a 2-year randomized, controlled trial.
The study is yet more proof that "it’s never too late to adopt a healthful lifestyle," said Dr. Ralph A. Nixon, chair of the Alzheimer’s Association Medical and Scientific Advisory Council.
The multimodal intervention study, called the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER), added nutritional guidance, physical exercise, cognitive training, and vascular risk factor management to the treatment of half of 1,260 participants who were between ages 60 and 77 years. The other half of participants received regular health advice.
To their surprise, researchers achieved their primary outcome – improvement in cognitive performance – within 2 years. In fact, the benefit was significant in all cognitive domains, including memory, executive function, and psychomotor speed, compared with the control group.
Only 11% of the participants dropped out. There were no serious adverse events.
This is the first long-term study to assess the impact of simultaneous improvement of several lifestyle factors on elderly cognition, the investigators said. The researchers are now conducting cost analysis, and they plan to follow the patients for 7 years to assess the longer-term effect of the interventions.
The findings demonstrate that multidomain intervention is feasible and can reduce the risk of cognitive impairment among the at-risk elderly population, said the study’s lead investigator, Dr. Miia Kivipelto. She hopes that this proof-of-concept study becomes a model for future programs in this group.
In a video interview, Dr. Kivipelto, a professor at the Karolinska Institute in Stockholm, discusses the study’s findings and its implications for clinical practice.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
AT AAIC 2014
VIDEO: Dementia risk spikes in older veterans with sleep disorders, PTSD
COPENHAGEN – Older veterans who had sleep disturbances were at a 30% greater risk of developing dementia, according to a retrospective analysis of 200,000 medical records presented at the Alzheimer’s Association International Conference 2014.
And having posttraumatic stress disorder (PTSD) in addition to sleep disturbances put veterans at an 80% greater risk.
"As veterans are turning 65 and older, it’s important for us to understand who in that population is at an increased risk of developing dementia, so when we have that therapy or lifestyle intervention, we can intervene at that point," said Heather M. Snyder, Ph.D., director of medical and scientific operations at the Alzheimer’s Association. Dr. Snyder was not involved in the study.
For the study, researchers studied the records of veterans 55 years and older for 8 years. They found that almost 11% of the veterans with sleep disturbance developed dementia, compared with 9% of those without sleep disturbance, almost a 30% risk increase. The results were similar for veterans who had sleep apnea and nonapnea insomnia.
Meanwhile, researchers found no significant interaction between sleep disturbance and traumatic brain injury or PTSD, with regard to increased risk of dementia. However, veterans who had both PTSD and sleep disturbance had an 80% increased risk of developing dementia.
In a video interview, Dr. Kristine Yaffe, professor of psychiatry and neurology at the University of California, San Francisco, explains the study’s findings, shares practice pearls, and discusses the implications for younger veterans.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
COPENHAGEN – Older veterans who had sleep disturbances were at a 30% greater risk of developing dementia, according to a retrospective analysis of 200,000 medical records presented at the Alzheimer’s Association International Conference 2014.
And having posttraumatic stress disorder (PTSD) in addition to sleep disturbances put veterans at an 80% greater risk.
"As veterans are turning 65 and older, it’s important for us to understand who in that population is at an increased risk of developing dementia, so when we have that therapy or lifestyle intervention, we can intervene at that point," said Heather M. Snyder, Ph.D., director of medical and scientific operations at the Alzheimer’s Association. Dr. Snyder was not involved in the study.
For the study, researchers studied the records of veterans 55 years and older for 8 years. They found that almost 11% of the veterans with sleep disturbance developed dementia, compared with 9% of those without sleep disturbance, almost a 30% risk increase. The results were similar for veterans who had sleep apnea and nonapnea insomnia.
Meanwhile, researchers found no significant interaction between sleep disturbance and traumatic brain injury or PTSD, with regard to increased risk of dementia. However, veterans who had both PTSD and sleep disturbance had an 80% increased risk of developing dementia.
In a video interview, Dr. Kristine Yaffe, professor of psychiatry and neurology at the University of California, San Francisco, explains the study’s findings, shares practice pearls, and discusses the implications for younger veterans.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
COPENHAGEN – Older veterans who had sleep disturbances were at a 30% greater risk of developing dementia, according to a retrospective analysis of 200,000 medical records presented at the Alzheimer’s Association International Conference 2014.
And having posttraumatic stress disorder (PTSD) in addition to sleep disturbances put veterans at an 80% greater risk.
"As veterans are turning 65 and older, it’s important for us to understand who in that population is at an increased risk of developing dementia, so when we have that therapy or lifestyle intervention, we can intervene at that point," said Heather M. Snyder, Ph.D., director of medical and scientific operations at the Alzheimer’s Association. Dr. Snyder was not involved in the study.
For the study, researchers studied the records of veterans 55 years and older for 8 years. They found that almost 11% of the veterans with sleep disturbance developed dementia, compared with 9% of those without sleep disturbance, almost a 30% risk increase. The results were similar for veterans who had sleep apnea and nonapnea insomnia.
Meanwhile, researchers found no significant interaction between sleep disturbance and traumatic brain injury or PTSD, with regard to increased risk of dementia. However, veterans who had both PTSD and sleep disturbance had an 80% increased risk of developing dementia.
In a video interview, Dr. Kristine Yaffe, professor of psychiatry and neurology at the University of California, San Francisco, explains the study’s findings, shares practice pearls, and discusses the implications for younger veterans.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @naseemmiller
AT AAIC 2014
VIDEO: In patients with dementia, ask about guns at home
COPENHAGEN – A new study revealing the presence of firearms in the homes of more than 12% of community-dwelling dementia patients in the Midwest highlights a difficult situation for physicians treating such patients.
"Doctors have no legal right here to remove weapons from the home," noted study investigator Jason Hsieh, a fourth-year medical student at Cleveland Clinic Lerner College of Medicine. "The only thing we can do is try to identify if one exists, and encourage family members and caregivers to consider removing it or at least store it locked and unloaded in an inaccessible location."
In a video interview at the Alzheimer’s Association International Conference 2014, Mr. Hsieh discussed the study’s findings and the strategies physicians might use to keep their community-dwelling patients with dementia safe.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @alz_gal
COPENHAGEN – A new study revealing the presence of firearms in the homes of more than 12% of community-dwelling dementia patients in the Midwest highlights a difficult situation for physicians treating such patients.
"Doctors have no legal right here to remove weapons from the home," noted study investigator Jason Hsieh, a fourth-year medical student at Cleveland Clinic Lerner College of Medicine. "The only thing we can do is try to identify if one exists, and encourage family members and caregivers to consider removing it or at least store it locked and unloaded in an inaccessible location."
In a video interview at the Alzheimer’s Association International Conference 2014, Mr. Hsieh discussed the study’s findings and the strategies physicians might use to keep their community-dwelling patients with dementia safe.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @alz_gal
COPENHAGEN – A new study revealing the presence of firearms in the homes of more than 12% of community-dwelling dementia patients in the Midwest highlights a difficult situation for physicians treating such patients.
"Doctors have no legal right here to remove weapons from the home," noted study investigator Jason Hsieh, a fourth-year medical student at Cleveland Clinic Lerner College of Medicine. "The only thing we can do is try to identify if one exists, and encourage family members and caregivers to consider removing it or at least store it locked and unloaded in an inaccessible location."
In a video interview at the Alzheimer’s Association International Conference 2014, Mr. Hsieh discussed the study’s findings and the strategies physicians might use to keep their community-dwelling patients with dementia safe.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @alz_gal
AT AAIC 2014
VIDEO: Lasers plus angiogenesis inhibitors equal potential for treating port wine stains
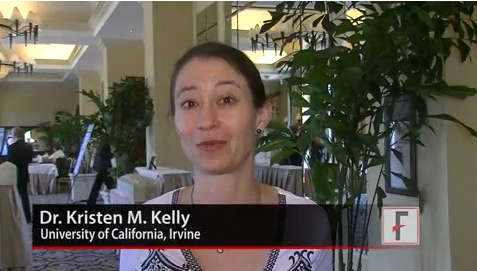
DANA POINT, CALIF. – Although lasers are standard in the treatment of port wine stains, they don’t give full clearance or prevent recurrence, according to Dr. Kristen M. Kelly of the University of California, Irvine.
Using imaging, Dr. Kelly and her team observed that lasers provided patients with "very nice" destruction of blood vessels, but in as little as a week, revascularization would begin. The investigators sought a way to interfere with angiogenesis in order to extend clearance of the port wine stains.
In this video, recorded during the Summit in Aesthetic Medicine 2014, Dr. Kelly says that she doesn’t think "we have the optimal treatment yet." However, a proof of concept study she and others conducted that combined laser treatment with Food and Drug Administration–approved agents, such as imiquimod and rapamycin, showed "a slight improvement over lasers alone."
Dr. Kelly added that the current research is only a "first step," and that other agents will prove equally or more effective as antiangiogenesis gains recognition as a viable treatment for a range of cutaneous conditions.
The Summit in Aesthetic Medicine is held by the Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Kelly said she had no relevant financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
DANA POINT, CALIF. – Although lasers are standard in the treatment of port wine stains, they don’t give full clearance or prevent recurrence, according to Dr. Kristen M. Kelly of the University of California, Irvine.
Using imaging, Dr. Kelly and her team observed that lasers provided patients with "very nice" destruction of blood vessels, but in as little as a week, revascularization would begin. The investigators sought a way to interfere with angiogenesis in order to extend clearance of the port wine stains.
In this video, recorded during the Summit in Aesthetic Medicine 2014, Dr. Kelly says that she doesn’t think "we have the optimal treatment yet." However, a proof of concept study she and others conducted that combined laser treatment with Food and Drug Administration–approved agents, such as imiquimod and rapamycin, showed "a slight improvement over lasers alone."
Dr. Kelly added that the current research is only a "first step," and that other agents will prove equally or more effective as antiangiogenesis gains recognition as a viable treatment for a range of cutaneous conditions.
The Summit in Aesthetic Medicine is held by the Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Kelly said she had no relevant financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
DANA POINT, CALIF. – Although lasers are standard in the treatment of port wine stains, they don’t give full clearance or prevent recurrence, according to Dr. Kristen M. Kelly of the University of California, Irvine.
Using imaging, Dr. Kelly and her team observed that lasers provided patients with "very nice" destruction of blood vessels, but in as little as a week, revascularization would begin. The investigators sought a way to interfere with angiogenesis in order to extend clearance of the port wine stains.
In this video, recorded during the Summit in Aesthetic Medicine 2014, Dr. Kelly says that she doesn’t think "we have the optimal treatment yet." However, a proof of concept study she and others conducted that combined laser treatment with Food and Drug Administration–approved agents, such as imiquimod and rapamycin, showed "a slight improvement over lasers alone."
Dr. Kelly added that the current research is only a "first step," and that other agents will prove equally or more effective as antiangiogenesis gains recognition as a viable treatment for a range of cutaneous conditions.
The Summit in Aesthetic Medicine is held by the Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Kelly said she had no relevant financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
EXPERT ANALYSIS FROM SUMMIT IN AESTHETIC MEDICINE
VIDEO: Consider cognitive function in elderly before surgery
COPENHAGEN – Almost half of patients with mild cognitive impairment progressed to dementia within 1 year of undergoing either arthroplasty or coronary angiography, according to a small Australian study.
Baseline cognitive impairment appeared to be the main driver of progression, increasing the risk more than sevenfold – significantly more than age or heart attack, Lisbeth Evered, Ph.D., said at the annual Alzheimer’s Association International Conference.
"After 12 months, 42% met the criteria for dementia," said Dr. Evered, a researcher at the University of Melbourne. "The expected annual progression from mild cognitive impairment [MCI] to dementia would be about 10%-12% per year."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Her study included 67 patients with a mean age of 70 years. All had MCI at baseline, with a mean score of 23 on the Mini-Mental State Exam (MMSE). They underwent either arthroplasty (26 patients) or coronary angiography (41 patients).
A year after surgery, about 34% of the arthroplasty patients and 46% of the angiography patients had progressed to dementia. Baseline cognitive impairment was the only factor significantly associated with the change.
Postsurgical cognitive decline, both transient and long lasting, is a well-documented phenomenon, with studies going back to the late 1800s. Although the causative link isn’t entirely clear, anesthetics have long been implicated, said Dr. Evered. In animal models, some anesthesia drugs do seem to precipitate an Alzheimer’s-like amyloidosis and tau hyperphosphorylation.
More recent animal data suggest that inflammation might be a powerful influence.
"When a patient has surgery with a general anesthetic, they experience peripheral inflammation," Dr. Evered explained. "In a healthy normal brain, there’s plenty of cognitive reserve, and although there might be some cognitive decline afterward, the person won’t really notice and will certainly recover."
In a vulnerable brain, however, the inflammation may be amplified and may cause significant collateral damage that accelerates cognitive decline. "The problem is, we don’t know why they are vulnerable or what we might do about it," she noted.
The best approach now is to routinely assess cognition before surgery and monitor it afterward, Dr. Evered explained. "Then, the perioperative period is not something occurring in isolation," she noted. "We will be better able to identify those at risk and implement strategies to improve their outcomes."
In a video interview, Dr. Evered and Dr. Brendan Silbert of St. Vincent’s Hospital, Melbourne, discuss the study and its implications.
Dr. Evered had no financial disclosures
On Twitter @alz_gal
COPENHAGEN – Almost half of patients with mild cognitive impairment progressed to dementia within 1 year of undergoing either arthroplasty or coronary angiography, according to a small Australian study.
Baseline cognitive impairment appeared to be the main driver of progression, increasing the risk more than sevenfold – significantly more than age or heart attack, Lisbeth Evered, Ph.D., said at the annual Alzheimer’s Association International Conference.
"After 12 months, 42% met the criteria for dementia," said Dr. Evered, a researcher at the University of Melbourne. "The expected annual progression from mild cognitive impairment [MCI] to dementia would be about 10%-12% per year."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Her study included 67 patients with a mean age of 70 years. All had MCI at baseline, with a mean score of 23 on the Mini-Mental State Exam (MMSE). They underwent either arthroplasty (26 patients) or coronary angiography (41 patients).
A year after surgery, about 34% of the arthroplasty patients and 46% of the angiography patients had progressed to dementia. Baseline cognitive impairment was the only factor significantly associated with the change.
Postsurgical cognitive decline, both transient and long lasting, is a well-documented phenomenon, with studies going back to the late 1800s. Although the causative link isn’t entirely clear, anesthetics have long been implicated, said Dr. Evered. In animal models, some anesthesia drugs do seem to precipitate an Alzheimer’s-like amyloidosis and tau hyperphosphorylation.
More recent animal data suggest that inflammation might be a powerful influence.
"When a patient has surgery with a general anesthetic, they experience peripheral inflammation," Dr. Evered explained. "In a healthy normal brain, there’s plenty of cognitive reserve, and although there might be some cognitive decline afterward, the person won’t really notice and will certainly recover."
In a vulnerable brain, however, the inflammation may be amplified and may cause significant collateral damage that accelerates cognitive decline. "The problem is, we don’t know why they are vulnerable or what we might do about it," she noted.
The best approach now is to routinely assess cognition before surgery and monitor it afterward, Dr. Evered explained. "Then, the perioperative period is not something occurring in isolation," she noted. "We will be better able to identify those at risk and implement strategies to improve their outcomes."
In a video interview, Dr. Evered and Dr. Brendan Silbert of St. Vincent’s Hospital, Melbourne, discuss the study and its implications.
Dr. Evered had no financial disclosures
On Twitter @alz_gal
COPENHAGEN – Almost half of patients with mild cognitive impairment progressed to dementia within 1 year of undergoing either arthroplasty or coronary angiography, according to a small Australian study.
Baseline cognitive impairment appeared to be the main driver of progression, increasing the risk more than sevenfold – significantly more than age or heart attack, Lisbeth Evered, Ph.D., said at the annual Alzheimer’s Association International Conference.
"After 12 months, 42% met the criteria for dementia," said Dr. Evered, a researcher at the University of Melbourne. "The expected annual progression from mild cognitive impairment [MCI] to dementia would be about 10%-12% per year."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Her study included 67 patients with a mean age of 70 years. All had MCI at baseline, with a mean score of 23 on the Mini-Mental State Exam (MMSE). They underwent either arthroplasty (26 patients) or coronary angiography (41 patients).
A year after surgery, about 34% of the arthroplasty patients and 46% of the angiography patients had progressed to dementia. Baseline cognitive impairment was the only factor significantly associated with the change.
Postsurgical cognitive decline, both transient and long lasting, is a well-documented phenomenon, with studies going back to the late 1800s. Although the causative link isn’t entirely clear, anesthetics have long been implicated, said Dr. Evered. In animal models, some anesthesia drugs do seem to precipitate an Alzheimer’s-like amyloidosis and tau hyperphosphorylation.
More recent animal data suggest that inflammation might be a powerful influence.
"When a patient has surgery with a general anesthetic, they experience peripheral inflammation," Dr. Evered explained. "In a healthy normal brain, there’s plenty of cognitive reserve, and although there might be some cognitive decline afterward, the person won’t really notice and will certainly recover."
In a vulnerable brain, however, the inflammation may be amplified and may cause significant collateral damage that accelerates cognitive decline. "The problem is, we don’t know why they are vulnerable or what we might do about it," she noted.
The best approach now is to routinely assess cognition before surgery and monitor it afterward, Dr. Evered explained. "Then, the perioperative period is not something occurring in isolation," she noted. "We will be better able to identify those at risk and implement strategies to improve their outcomes."
In a video interview, Dr. Evered and Dr. Brendan Silbert of St. Vincent’s Hospital, Melbourne, discuss the study and its implications.
Dr. Evered had no financial disclosures
On Twitter @alz_gal
AT AAIC 2014
VIDEO: Investigative eye amyloid test shines a light on Alzheimer’s pathology
COPENHAGEN – When it comes to beta-amyloid plaques, the eyes may be more than a poetic window to the soul – they also may be mirrors of pathology developing in the brain.
It turns out that the eye, as a virtual extension of the brain itself, accumulates the same amyloid pathology that damages the brain when Alzheimer’s disease strikes. Visualizing these plaques in the retina and in the lens may eventually become a low-cost, noninvasive screening tool for early disease, according to research presented at the Alzheimer’s Association International Conference 2014.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
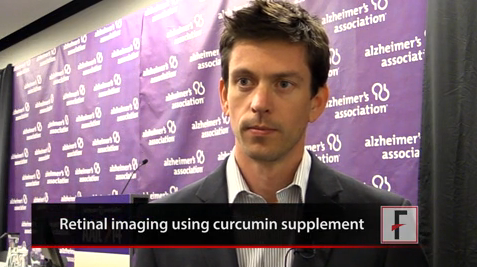
"Our first 45 subjects showed a strong correlation between retinal amyloid and brain plaques," said Shaun Frost of the Commonwealth Scientific and Industrial Research Organisation (CSIRO) in Australia. "In fact, we had 100% sensitivity and no false positives, something that’s critical for an Alzheimer’s test, because we don’t want to leave anyone behind when it comes to early signs."
Curcumin, an extract of the spice turmeric, is the fluorescent agent in the test. Mr. Frost said investigators were first drawn to it because some studies suggest a lower rate of Alzheimer’s in India, where turmeric is a common seasoning.
Paul Hartung, president of Cognoptix in Acton, Mass., said his company focuses on lens amyloid, which shows a correlation to brain plaques. Cognoptix is accumulating data on a device that identifies plaques using dynamic light scattering and a fluorescent molecule delivered in an ophthalmic ointment.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In the phase II study he presented, the test differentiated 20 people with Alzheimer’s from 20 healthy controls with an 85% sensitivity and 95% specificity. It also correlated significantly with brain plaques seen on PET amyloid imaging.
A phase III trial is in the works, after which Cognoptix intends to seek approval from the Food and Drug Administration. Mr. Hartung hopes the device could be marketed by 2016.
The lens amyloid trial is a collaboration between CSIRO; Edith Cowan University in Mt. Lawley, Australia; the McCusker Alzheimer’s Research Foundation; California-based NeuroVision Imaging; and the Australian Imaging, Biomarker & Lifestyle Flagship Study of Ageing. Mr. Frost had no financial disclosures. Mr. Hartung is a full-time employee of Cognotpix, which is developing the test for commercial use.
On Twitter @alz_gal
COPENHAGEN – When it comes to beta-amyloid plaques, the eyes may be more than a poetic window to the soul – they also may be mirrors of pathology developing in the brain.
It turns out that the eye, as a virtual extension of the brain itself, accumulates the same amyloid pathology that damages the brain when Alzheimer’s disease strikes. Visualizing these plaques in the retina and in the lens may eventually become a low-cost, noninvasive screening tool for early disease, according to research presented at the Alzheimer’s Association International Conference 2014.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
"Our first 45 subjects showed a strong correlation between retinal amyloid and brain plaques," said Shaun Frost of the Commonwealth Scientific and Industrial Research Organisation (CSIRO) in Australia. "In fact, we had 100% sensitivity and no false positives, something that’s critical for an Alzheimer’s test, because we don’t want to leave anyone behind when it comes to early signs."
Curcumin, an extract of the spice turmeric, is the fluorescent agent in the test. Mr. Frost said investigators were first drawn to it because some studies suggest a lower rate of Alzheimer’s in India, where turmeric is a common seasoning.
Paul Hartung, president of Cognoptix in Acton, Mass., said his company focuses on lens amyloid, which shows a correlation to brain plaques. Cognoptix is accumulating data on a device that identifies plaques using dynamic light scattering and a fluorescent molecule delivered in an ophthalmic ointment.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In the phase II study he presented, the test differentiated 20 people with Alzheimer’s from 20 healthy controls with an 85% sensitivity and 95% specificity. It also correlated significantly with brain plaques seen on PET amyloid imaging.
A phase III trial is in the works, after which Cognoptix intends to seek approval from the Food and Drug Administration. Mr. Hartung hopes the device could be marketed by 2016.
The lens amyloid trial is a collaboration between CSIRO; Edith Cowan University in Mt. Lawley, Australia; the McCusker Alzheimer’s Research Foundation; California-based NeuroVision Imaging; and the Australian Imaging, Biomarker & Lifestyle Flagship Study of Ageing. Mr. Frost had no financial disclosures. Mr. Hartung is a full-time employee of Cognotpix, which is developing the test for commercial use.
On Twitter @alz_gal
COPENHAGEN – When it comes to beta-amyloid plaques, the eyes may be more than a poetic window to the soul – they also may be mirrors of pathology developing in the brain.
It turns out that the eye, as a virtual extension of the brain itself, accumulates the same amyloid pathology that damages the brain when Alzheimer’s disease strikes. Visualizing these plaques in the retina and in the lens may eventually become a low-cost, noninvasive screening tool for early disease, according to research presented at the Alzheimer’s Association International Conference 2014.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
"Our first 45 subjects showed a strong correlation between retinal amyloid and brain plaques," said Shaun Frost of the Commonwealth Scientific and Industrial Research Organisation (CSIRO) in Australia. "In fact, we had 100% sensitivity and no false positives, something that’s critical for an Alzheimer’s test, because we don’t want to leave anyone behind when it comes to early signs."
Curcumin, an extract of the spice turmeric, is the fluorescent agent in the test. Mr. Frost said investigators were first drawn to it because some studies suggest a lower rate of Alzheimer’s in India, where turmeric is a common seasoning.
Paul Hartung, president of Cognoptix in Acton, Mass., said his company focuses on lens amyloid, which shows a correlation to brain plaques. Cognoptix is accumulating data on a device that identifies plaques using dynamic light scattering and a fluorescent molecule delivered in an ophthalmic ointment.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In the phase II study he presented, the test differentiated 20 people with Alzheimer’s from 20 healthy controls with an 85% sensitivity and 95% specificity. It also correlated significantly with brain plaques seen on PET amyloid imaging.
A phase III trial is in the works, after which Cognoptix intends to seek approval from the Food and Drug Administration. Mr. Hartung hopes the device could be marketed by 2016.
The lens amyloid trial is a collaboration between CSIRO; Edith Cowan University in Mt. Lawley, Australia; the McCusker Alzheimer’s Research Foundation; California-based NeuroVision Imaging; and the Australian Imaging, Biomarker & Lifestyle Flagship Study of Ageing. Mr. Frost had no financial disclosures. Mr. Hartung is a full-time employee of Cognotpix, which is developing the test for commercial use.
On Twitter @alz_gal
AT AAIC 2014