User login
Surgical Treatment of Nonmelanoma Skin Cancer in Older Adult Veterans
Skin cancer is the most diagnosed cancer in the United States. Nonmelanoma skin cancers (NMSC), which include basal cell carcinoma and squamous cell carcinoma, are usually cured with removal.1 The incidence of NMSC increases with age and is commonly found in nursing homes and geriatric units. These cancers are not usually metastatic or fatal but can cause local destruction and disfigurement if neglected.2 The current standard of care is to treat diagnosed NMSC; however, the dermatology and geriatric care literature have questioned the logic of treating asymptomatic skin cancers that will not affect a patient’s life expectancy.2-4
Forty-seven percent of the current living veteran population is aged ≥ 65 years.5 Older adult patients are frequently referred to the US Department of Veterans Affairs (VA) surgical service for the treatment of NMSC. The veteran population includes a higher percentage of individuals at an elevated risk of skin cancers (older, White, and male) compared with the general population.6 World War II veterans deployed in regions closer to the equator have been found to have an elevated risk of melanoma and nonmelanoma skin carcinomas.7 A retrospective study of Vietnam veterans exposed to Agent Orange (2,3,7,8-tetrachlorodibenzodioxin) found a significantly higher risk of invasive NMSC in Fitzpatrick skin types I-IV compared with an age-matched subset of the general population.8 Younger veterans who were deployed in Afghanistan and Iraq for Operation Enduring Freedom/Operation Iraqi Freedom worked at more equatorial latitudes than the rest of the US population and may be at increased risk of NMSC. Inadequate sunscreen access, immediate safety concerns, outdoor recreational activities, harsh weather, and insufficient emphasis on sun protection have created a multifactorial challenge for the military population. Riemenschneider and colleagues recommended targeted screening for at-risk veteran patients and prioritizing annual skin cancer screenings during medical mission physical examinations for active military.7
The plastic surgery service regularly receives consults from dermatology, general surgery, and primary care to remove skin cancers on the face, scalp, hands, and forearms. Skin cancer treatment can create serious hardships for older adult patients and their families with multiple appointments for the consult, procedure, and follow-up. Patients are often told to hold their anticoagulant medications when the surgery will be performed on a highly vascular region, such as the scalp or face. This can create wide swings in their laboratory test values and result in life-threatening complications from either bleeding or clotting. The appropriateness of offering surgery to patients with serious comorbidities and a limited life expectancy has been questioned.2-4 The purpose of this study was to measure the morbidity and unrelated 5-year mortality for patients with skin cancer referred to the plastic surgery service to help patients and families make a more informed treatment decision, particularly when the patients are aged > 80 years and have significant life-threatening comorbidities.
Methods
The University of Florida and Malcom Randall VA Medical Center Institutional review board in Gainesville, approved a retrospective review of all consults completed by the plastic surgery service for the treatment of NMSC performed from July 1, 2011 to June 30, 2015. Data collected included age and common life-limiting comorbidities at the time of referral. Morbidities were found on the electronic health record, including coronary artery disease (CAD), congestive heart failure (CHF), cerebral vascular disease (CVD), peripheral vascular disease, dementia, chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), tobacco use, diabetes mellitus (DM), liver disease, alcohol use, and obstructive sleep apnea.
Treatment, complications, and 5-year mortality were recorded. A χ2 analysis with P value < .05 was used to determine statistical significance between individual risk factors and 5-year mortality. The relative risk of 5-year mortality was calculated by combining advanced age (aged > 80 years) with the individual comorbidities.
Results
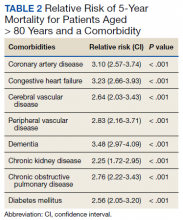
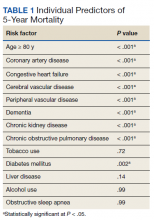
Over 4 years, 800 consults for NMSC were completed by the plastic surgery service. Treatment decisions included 210 excisions (with or without reconstruction) in the operating room, 402 excisions (with or without reconstruction) under local anesthesia in clinic, 55 Mohs surgical dermatology referrals, 21 other service or hospital referrals, and 112 patient who were observed, declined intervention, or died prior to intervention. Five-year mortality was 28.6%. No patients died of NMSC. The median age at consult submission for patients deceased 5 years later was 78 years. Complication rate was 5% and included wound infection, dehiscence, bleeding, or graft loss. Two patients, both deceased within 5 years, had unplanned admissions due to bleeding from either a skin graft donor site or recipient bleeding. Aged ≥ 80 years, CAD, CHF, CVD, peripheral vascular disease, dementia, CKD, COPD, and DM were all found individually to be statistically significant predictors of 5-year mortality (Table 1). Combining aged ≥ 80 years plus CAD, CHF, or dementia all increased the 5-year mortality by a relative risk of > 3 (Table 2).
Discussion
The standard of care is to treat NMSC. Most NMSCs are treated surgically without consideration of patient age or life expectancy.2,4,9,10 A prospective cohort study involving a university-based private practice and a VA medical center in San Francisco found a 22.6% overall 5-year mortality and a 43.3% mortality in the group defined as limited life expectancy (LLE) based on age (≥ 85 years) and medical comorbidities. None died due to the NMSC. Leading cause of death was cardiac, cerebrovascular, and respiratory disease, lung and prostate cancer, and Alzheimer disease. The authors suggested the LLE group may be exposed to wound complications without benefiting from the treatment.4
Another study of 440 patients receiving excision for biopsy-proven facial NMSC at the Roudebush VA Medical Center in Indianapolis, Indiana, found no residual carcinoma in 35.3% of excisions, and in patients aged > 90 years, more than half of the excisions had no residual carcinoma. More than half of the patients aged > 90 years died within 1 year, not as a result of the NMSC. The authors argued for watchful waiting in select patients to maximize comfort and outcomes.10
NMSCs are often asymptomatic and not immediately life threatening. Although NMSCs tend to have a favorable prognosis, studies have found that NMSC may be a marker for other poor health outcomes. A significant increased risk for all-cause mortality was found for patients with a history of SCC, which may be attributed to immune status.11 The aging veteran population has more complex health care needs to be considered when developing surgical treatment plans. These medical problems may limit their life expectancy much sooner than the skin cancer will become symptomatic. We found that individuals aged ≥ 80 years who had CAD, CHF, or dementia had a relative risk of 3 or higher for 5-year mortality. The leading cause of death in the United States in years 2011 to 2015 was heart disease. Alzheimer disease was the sixth leading cause of death in those same years.12-14
Skin cancer excisions do not typically require general anesthesia, deep sedation, or large fluid shifts; however, studies have found that when frail patients undergo low-risk procedures, they tend to have a higher mortality rate than their healthier counterparts.15 Frailty is a concept that identifies patients who are at increased risk of dying in 6 to 60 months due to a decline in their physical reserve. Frail patients have increased rates of perioperative mortality and complications. Various tools have been used to assess the components of physical performance, speed, mobility, nutrition status, mental health, and cognition.16 Frailty screening has been initiated in several VA hospitals, including our own in Gainesville, Florida, with the goal of decreasing postoperative morbidity and mortality in older adult patients.17 The patients are given a 1-page screening assessment that asks about their living situation, medical conditions, nutrition status, cognition, and activities of daily living. The results can trigger the clinician to rethink the surgical plan and mobilize more resources to optimize the patient’s health. This study period precedes the initiative at our institution.
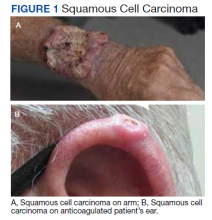
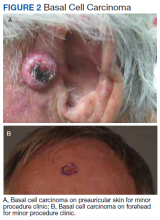
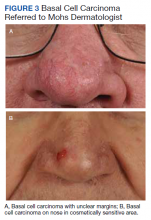
The plastic surgery service’s routine practice is to excise skin cancers in the operating room if sedation or general anesthesia will be needed (Figure 1A), for optimal control of bleeding (Figure 1B) in a patient who cannot safely stop blood thinners, or for excision of a highly vascularized area such as the scalp. Surgery is offered in an office-based setting if the area can be closed primarily, left open to close secondarily, or closed with a small skin graft under local anesthesia only (Figure 2). We prefer treating frail patients in the minor procedure clinic, when possible, to avoid the risks of sedation and the additional preoperative visits and transportation requirements. NMSC with unclear margins (Figure 3A) or in cosmetically sensitive areas where tissue needs to be preserved (Figure 3B) are referred to the Mohs dermatologist. The skin cancers in this study were most frequently found on the face, scalp, hands, and forearms based on referral patterns.
Other treatment options for NMSC include curettage and electrodessication, cryotherapy, and radiation; however, ours is a surgical service and patients are typically referred to us by primary care or dermatology when those are not reasonable or desirable options.18 Published complication rates of patients having skin cancer surgery without age restriction have a rate of 3% to 6%, which is consistent with our study of 5%.19-21 Two bleeding complications that needed to be admitted did not require more than a bedside procedure and neither required transfusions. One patient had been instructed to continue taking coumadin during the perioperative office-based procedure due to a recent carotid stent placement in the setting of a rapidly growing basal cell on an easily accessible location.
The most noted comorbidity in patients with wound complications was found to be DM; however, this was not found to be a statistically significant risk factor for wound complications (P = .10). We do not have a set rule for advising for or against NMSC surgery. We do counsel frail patients and their families that not all cancer is immediately life threatening and will work with them to do whatever makes the most sense to achieve their goals, occasionally accepting positive margins in order to debulk a symptomatic growth. The objective of this paper is to contribute to the discussion of performing invasive procedures on older adult veterans with life-limiting comorbidities. Patients and their families will have different thresholds for what they feel needs intervention, especially if other medical problems are consuming much of their time. We also have the community care referral option for patients whose treatment decisions are being dictated by travel hardships.
Strengths and Limitations
A strength of this study is that the data were obtained from a closed system. Patients tend to stay long-term within the VA and their health record is accessible throughout the country as long as they are seen at a VA facility. Complications, therefore, return to the treating service or primary care, who would route the patient back to the surgeon.
One limitation of the study is that this is a retrospective review from 2011. The authors are limited to data that are recorded in the patient record. Multiple health care professionals saw the patients and notes lack consistency in detail. Size of the lesions were not consistently recorded and did not get logged into our database for that reason.
Conclusions
Treatment of NMSC in older adult patients has a low morbidity but needs to be balanced against a patient and family’s goals when the patient presents with life-limiting comorbidities. An elevated 5-year mortality in patients aged > 80 years with serious unrelated medical conditions is intuitive, but this study may help put treatment plans into perspective for families and health care professionals who want to provide an indicated service while maximizing patient quality of life.
Acknowledgments
This manuscript is the result of work supported with resources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
1. American Cancer Society. Cancer Facts & Figures 2021. Accessed May 26, 2022. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2021/cancer-facts-and-figures-2021.pdf
2. Albert A, Knoll MA, Conti JA, Zbar RIS. Non-melanoma skin cancers in the older patient. Curr Oncol Rep. 2019;21(9):79. Published 2019 Jul 29. doi:10.1007/s11912-019-0828-9
3. Linos E, Chren MM, Stijacic Cenzer I, Covinsky KE. Skin cancer in U.S. elderly adults: does life expectancy play a role in treatment decisions? J Am Geriatr Soc. 2016;64(8):1610-1615. doi:10.1111/jgs.14202
4. Linos E, Parvataneni R, Stuart SE, Boscardin WJ, Landefeld CS, Chren MM. Treatment of nonfatal conditions at the end of life: nonmelanoma skin cancer. JAMA Intern Med. 2013;173(11):1006-1012. doi:10.1001/jamainternmed.2013.639
5. O’Malley KA, Vinson L, Kaiser AP, Sager Z, Hinrichs K. Mental health and aging veterans: how the Veterans Health Administration meets the needs of aging veterans. Public Policy Aging Rep. 2020;30(1):19-23. doi:10.1093/ppar/prz027
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Profile of veterans: 2017. Accessed May 26, 2022. https://www.va.gov/vetdata/docs/SpecialReports/Profile_of_Veterans_2017.pdf 7. Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78(6):1185-1192. doi:10.1016/j.jaad.2017.11.062
8. Clemens MW, Kochuba AL, Carter ME, Han K, Liu J, Evans K. Association between Agent Orange exposure and nonmelanotic invasive skin cancer: a pilot study. Plast Reconstr Surg. 2014;133(2):432-437. doi:10.1097/01.prs.0000436859.40151.cf
9. Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80(2):303-317. doi:10.1016/j.jaad.2018.03.060
10. Chauhan R, Munger BN, Chu MW, et al. Age at diagnosis as a relative contraindication for intervention in facial nonmelanoma skin cancer. JAMA Surg. 2018;153(4):390-392. doi:10.1001/jamasurg.2017.5073
11. Barton V, Armeson K, Hampras S, et al. Nonmelanoma skin cancer and risk of all-cause and cancer-related mortality: a systematic review. Arch Dermatol Res. 2017;309(4):243-251. doi:10.1007/s00403-017-1724-5
12. Kochanek KD, Murphy SL, Xu JQ, Arias E. Mortality in the United States, 2013. NCHS Data Brief 178. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db178.htm
13. Xu JQ, Kochanek KD, Murphy SL, Arias E. Mortality in the United States, 2012. NCHS Data Brief 168. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db168.htm
14. Xu JQ, Murphy SL, Kochanek KD, Arias E. Mortality in the United States, 2015. NCHS Data Brief 267. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db267.htm
15. Varley PR , Borrebach JD, Arya S, et al. Clinical utility of the risk analysis index as a prospective frailty screening tool within a multi-practice, multi-hospital integrated healthcare system. Ann Surg. 2021;274(6):e1230-e1237. doi:10.1097/SLA.0000000000003808
16. Hall DE, Arya S , Schmid KK, et al. Development and initial validation of the risk analysis index for measuring frailty in surgical populations. JAMA Surg. 2017;152(2):175-182. doi:10.1001/jamasurg.2016.4202
17. US Department of Veterans Affairs, Health Services Research & Development. Improving healthcare for aging veterans. Updated August 30, 2017. Accessed May 26, 2022. https://www.hsrd.research.va.gov/news/feature/aging0917.cfm
18. Leus AJG, Frie M, Haisma MS, et al. Treatment of keratinocyte carcinoma in elderly patients – a review of the current literature. J Eur Acad Dermatol Venereol. 2020;34(9):1932-1943. doi:10.1111/jdv.16268
19. Amici JM, Rogues AM, Lasheras A, et al. A prospective study of the incidence of complications associated with dermatological surgery. Br J Dermatol. 2005;153(5):967-971. doi:10.1111/j.1365-2133.2005.06861.x
20. Arguello-Guerra L, Vargas-Chandomid E, Díaz-González JM, Méndez-Flores S, Ruelas-Villavicencio A, Domínguez-Cherit J. Incidence of complications in dermatological surgery of melanoma and non-melanoma skin cancer in patients with multiple comorbidity and/or antiplatelet-anticoagulants. Five-year experience in our hospital. Cir Cir. 2019;86(1):15-23. doi:10.24875/CIRUE.M18000003
21. Keith DJ, de Berker DA, Bray AP, Cheung ST, Brain A, Mohd Mustapa MF. British Association of Dermatologists’ national audit on nonmelanoma skin cancer excision, 2014. Clin Exp Dermatol. 2017;42(1):46-53. doi:10.1111/ced.12990
Skin cancer is the most diagnosed cancer in the United States. Nonmelanoma skin cancers (NMSC), which include basal cell carcinoma and squamous cell carcinoma, are usually cured with removal.1 The incidence of NMSC increases with age and is commonly found in nursing homes and geriatric units. These cancers are not usually metastatic or fatal but can cause local destruction and disfigurement if neglected.2 The current standard of care is to treat diagnosed NMSC; however, the dermatology and geriatric care literature have questioned the logic of treating asymptomatic skin cancers that will not affect a patient’s life expectancy.2-4
Forty-seven percent of the current living veteran population is aged ≥ 65 years.5 Older adult patients are frequently referred to the US Department of Veterans Affairs (VA) surgical service for the treatment of NMSC. The veteran population includes a higher percentage of individuals at an elevated risk of skin cancers (older, White, and male) compared with the general population.6 World War II veterans deployed in regions closer to the equator have been found to have an elevated risk of melanoma and nonmelanoma skin carcinomas.7 A retrospective study of Vietnam veterans exposed to Agent Orange (2,3,7,8-tetrachlorodibenzodioxin) found a significantly higher risk of invasive NMSC in Fitzpatrick skin types I-IV compared with an age-matched subset of the general population.8 Younger veterans who were deployed in Afghanistan and Iraq for Operation Enduring Freedom/Operation Iraqi Freedom worked at more equatorial latitudes than the rest of the US population and may be at increased risk of NMSC. Inadequate sunscreen access, immediate safety concerns, outdoor recreational activities, harsh weather, and insufficient emphasis on sun protection have created a multifactorial challenge for the military population. Riemenschneider and colleagues recommended targeted screening for at-risk veteran patients and prioritizing annual skin cancer screenings during medical mission physical examinations for active military.7
The plastic surgery service regularly receives consults from dermatology, general surgery, and primary care to remove skin cancers on the face, scalp, hands, and forearms. Skin cancer treatment can create serious hardships for older adult patients and their families with multiple appointments for the consult, procedure, and follow-up. Patients are often told to hold their anticoagulant medications when the surgery will be performed on a highly vascular region, such as the scalp or face. This can create wide swings in their laboratory test values and result in life-threatening complications from either bleeding or clotting. The appropriateness of offering surgery to patients with serious comorbidities and a limited life expectancy has been questioned.2-4 The purpose of this study was to measure the morbidity and unrelated 5-year mortality for patients with skin cancer referred to the plastic surgery service to help patients and families make a more informed treatment decision, particularly when the patients are aged > 80 years and have significant life-threatening comorbidities.
Methods
The University of Florida and Malcom Randall VA Medical Center Institutional review board in Gainesville, approved a retrospective review of all consults completed by the plastic surgery service for the treatment of NMSC performed from July 1, 2011 to June 30, 2015. Data collected included age and common life-limiting comorbidities at the time of referral. Morbidities were found on the electronic health record, including coronary artery disease (CAD), congestive heart failure (CHF), cerebral vascular disease (CVD), peripheral vascular disease, dementia, chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), tobacco use, diabetes mellitus (DM), liver disease, alcohol use, and obstructive sleep apnea.
Treatment, complications, and 5-year mortality were recorded. A χ2 analysis with P value < .05 was used to determine statistical significance between individual risk factors and 5-year mortality. The relative risk of 5-year mortality was calculated by combining advanced age (aged > 80 years) with the individual comorbidities.
Results
Over 4 years, 800 consults for NMSC were completed by the plastic surgery service. Treatment decisions included 210 excisions (with or without reconstruction) in the operating room, 402 excisions (with or without reconstruction) under local anesthesia in clinic, 55 Mohs surgical dermatology referrals, 21 other service or hospital referrals, and 112 patient who were observed, declined intervention, or died prior to intervention. Five-year mortality was 28.6%. No patients died of NMSC. The median age at consult submission for patients deceased 5 years later was 78 years. Complication rate was 5% and included wound infection, dehiscence, bleeding, or graft loss. Two patients, both deceased within 5 years, had unplanned admissions due to bleeding from either a skin graft donor site or recipient bleeding. Aged ≥ 80 years, CAD, CHF, CVD, peripheral vascular disease, dementia, CKD, COPD, and DM were all found individually to be statistically significant predictors of 5-year mortality (Table 1). Combining aged ≥ 80 years plus CAD, CHF, or dementia all increased the 5-year mortality by a relative risk of > 3 (Table 2).
Discussion
The standard of care is to treat NMSC. Most NMSCs are treated surgically without consideration of patient age or life expectancy.2,4,9,10 A prospective cohort study involving a university-based private practice and a VA medical center in San Francisco found a 22.6% overall 5-year mortality and a 43.3% mortality in the group defined as limited life expectancy (LLE) based on age (≥ 85 years) and medical comorbidities. None died due to the NMSC. Leading cause of death was cardiac, cerebrovascular, and respiratory disease, lung and prostate cancer, and Alzheimer disease. The authors suggested the LLE group may be exposed to wound complications without benefiting from the treatment.4
Another study of 440 patients receiving excision for biopsy-proven facial NMSC at the Roudebush VA Medical Center in Indianapolis, Indiana, found no residual carcinoma in 35.3% of excisions, and in patients aged > 90 years, more than half of the excisions had no residual carcinoma. More than half of the patients aged > 90 years died within 1 year, not as a result of the NMSC. The authors argued for watchful waiting in select patients to maximize comfort and outcomes.10
NMSCs are often asymptomatic and not immediately life threatening. Although NMSCs tend to have a favorable prognosis, studies have found that NMSC may be a marker for other poor health outcomes. A significant increased risk for all-cause mortality was found for patients with a history of SCC, which may be attributed to immune status.11 The aging veteran population has more complex health care needs to be considered when developing surgical treatment plans. These medical problems may limit their life expectancy much sooner than the skin cancer will become symptomatic. We found that individuals aged ≥ 80 years who had CAD, CHF, or dementia had a relative risk of 3 or higher for 5-year mortality. The leading cause of death in the United States in years 2011 to 2015 was heart disease. Alzheimer disease was the sixth leading cause of death in those same years.12-14
Skin cancer excisions do not typically require general anesthesia, deep sedation, or large fluid shifts; however, studies have found that when frail patients undergo low-risk procedures, they tend to have a higher mortality rate than their healthier counterparts.15 Frailty is a concept that identifies patients who are at increased risk of dying in 6 to 60 months due to a decline in their physical reserve. Frail patients have increased rates of perioperative mortality and complications. Various tools have been used to assess the components of physical performance, speed, mobility, nutrition status, mental health, and cognition.16 Frailty screening has been initiated in several VA hospitals, including our own in Gainesville, Florida, with the goal of decreasing postoperative morbidity and mortality in older adult patients.17 The patients are given a 1-page screening assessment that asks about their living situation, medical conditions, nutrition status, cognition, and activities of daily living. The results can trigger the clinician to rethink the surgical plan and mobilize more resources to optimize the patient’s health. This study period precedes the initiative at our institution.
The plastic surgery service’s routine practice is to excise skin cancers in the operating room if sedation or general anesthesia will be needed (Figure 1A), for optimal control of bleeding (Figure 1B) in a patient who cannot safely stop blood thinners, or for excision of a highly vascularized area such as the scalp. Surgery is offered in an office-based setting if the area can be closed primarily, left open to close secondarily, or closed with a small skin graft under local anesthesia only (Figure 2). We prefer treating frail patients in the minor procedure clinic, when possible, to avoid the risks of sedation and the additional preoperative visits and transportation requirements. NMSC with unclear margins (Figure 3A) or in cosmetically sensitive areas where tissue needs to be preserved (Figure 3B) are referred to the Mohs dermatologist. The skin cancers in this study were most frequently found on the face, scalp, hands, and forearms based on referral patterns.
Other treatment options for NMSC include curettage and electrodessication, cryotherapy, and radiation; however, ours is a surgical service and patients are typically referred to us by primary care or dermatology when those are not reasonable or desirable options.18 Published complication rates of patients having skin cancer surgery without age restriction have a rate of 3% to 6%, which is consistent with our study of 5%.19-21 Two bleeding complications that needed to be admitted did not require more than a bedside procedure and neither required transfusions. One patient had been instructed to continue taking coumadin during the perioperative office-based procedure due to a recent carotid stent placement in the setting of a rapidly growing basal cell on an easily accessible location.
The most noted comorbidity in patients with wound complications was found to be DM; however, this was not found to be a statistically significant risk factor for wound complications (P = .10). We do not have a set rule for advising for or against NMSC surgery. We do counsel frail patients and their families that not all cancer is immediately life threatening and will work with them to do whatever makes the most sense to achieve their goals, occasionally accepting positive margins in order to debulk a symptomatic growth. The objective of this paper is to contribute to the discussion of performing invasive procedures on older adult veterans with life-limiting comorbidities. Patients and their families will have different thresholds for what they feel needs intervention, especially if other medical problems are consuming much of their time. We also have the community care referral option for patients whose treatment decisions are being dictated by travel hardships.
Strengths and Limitations
A strength of this study is that the data were obtained from a closed system. Patients tend to stay long-term within the VA and their health record is accessible throughout the country as long as they are seen at a VA facility. Complications, therefore, return to the treating service or primary care, who would route the patient back to the surgeon.
One limitation of the study is that this is a retrospective review from 2011. The authors are limited to data that are recorded in the patient record. Multiple health care professionals saw the patients and notes lack consistency in detail. Size of the lesions were not consistently recorded and did not get logged into our database for that reason.
Conclusions
Treatment of NMSC in older adult patients has a low morbidity but needs to be balanced against a patient and family’s goals when the patient presents with life-limiting comorbidities. An elevated 5-year mortality in patients aged > 80 years with serious unrelated medical conditions is intuitive, but this study may help put treatment plans into perspective for families and health care professionals who want to provide an indicated service while maximizing patient quality of life.
Acknowledgments
This manuscript is the result of work supported with resources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
Skin cancer is the most diagnosed cancer in the United States. Nonmelanoma skin cancers (NMSC), which include basal cell carcinoma and squamous cell carcinoma, are usually cured with removal.1 The incidence of NMSC increases with age and is commonly found in nursing homes and geriatric units. These cancers are not usually metastatic or fatal but can cause local destruction and disfigurement if neglected.2 The current standard of care is to treat diagnosed NMSC; however, the dermatology and geriatric care literature have questioned the logic of treating asymptomatic skin cancers that will not affect a patient’s life expectancy.2-4
Forty-seven percent of the current living veteran population is aged ≥ 65 years.5 Older adult patients are frequently referred to the US Department of Veterans Affairs (VA) surgical service for the treatment of NMSC. The veteran population includes a higher percentage of individuals at an elevated risk of skin cancers (older, White, and male) compared with the general population.6 World War II veterans deployed in regions closer to the equator have been found to have an elevated risk of melanoma and nonmelanoma skin carcinomas.7 A retrospective study of Vietnam veterans exposed to Agent Orange (2,3,7,8-tetrachlorodibenzodioxin) found a significantly higher risk of invasive NMSC in Fitzpatrick skin types I-IV compared with an age-matched subset of the general population.8 Younger veterans who were deployed in Afghanistan and Iraq for Operation Enduring Freedom/Operation Iraqi Freedom worked at more equatorial latitudes than the rest of the US population and may be at increased risk of NMSC. Inadequate sunscreen access, immediate safety concerns, outdoor recreational activities, harsh weather, and insufficient emphasis on sun protection have created a multifactorial challenge for the military population. Riemenschneider and colleagues recommended targeted screening for at-risk veteran patients and prioritizing annual skin cancer screenings during medical mission physical examinations for active military.7
The plastic surgery service regularly receives consults from dermatology, general surgery, and primary care to remove skin cancers on the face, scalp, hands, and forearms. Skin cancer treatment can create serious hardships for older adult patients and their families with multiple appointments for the consult, procedure, and follow-up. Patients are often told to hold their anticoagulant medications when the surgery will be performed on a highly vascular region, such as the scalp or face. This can create wide swings in their laboratory test values and result in life-threatening complications from either bleeding or clotting. The appropriateness of offering surgery to patients with serious comorbidities and a limited life expectancy has been questioned.2-4 The purpose of this study was to measure the morbidity and unrelated 5-year mortality for patients with skin cancer referred to the plastic surgery service to help patients and families make a more informed treatment decision, particularly when the patients are aged > 80 years and have significant life-threatening comorbidities.
Methods
The University of Florida and Malcom Randall VA Medical Center Institutional review board in Gainesville, approved a retrospective review of all consults completed by the plastic surgery service for the treatment of NMSC performed from July 1, 2011 to June 30, 2015. Data collected included age and common life-limiting comorbidities at the time of referral. Morbidities were found on the electronic health record, including coronary artery disease (CAD), congestive heart failure (CHF), cerebral vascular disease (CVD), peripheral vascular disease, dementia, chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), tobacco use, diabetes mellitus (DM), liver disease, alcohol use, and obstructive sleep apnea.
Treatment, complications, and 5-year mortality were recorded. A χ2 analysis with P value < .05 was used to determine statistical significance between individual risk factors and 5-year mortality. The relative risk of 5-year mortality was calculated by combining advanced age (aged > 80 years) with the individual comorbidities.
Results
Over 4 years, 800 consults for NMSC were completed by the plastic surgery service. Treatment decisions included 210 excisions (with or without reconstruction) in the operating room, 402 excisions (with or without reconstruction) under local anesthesia in clinic, 55 Mohs surgical dermatology referrals, 21 other service or hospital referrals, and 112 patient who were observed, declined intervention, or died prior to intervention. Five-year mortality was 28.6%. No patients died of NMSC. The median age at consult submission for patients deceased 5 years later was 78 years. Complication rate was 5% and included wound infection, dehiscence, bleeding, or graft loss. Two patients, both deceased within 5 years, had unplanned admissions due to bleeding from either a skin graft donor site or recipient bleeding. Aged ≥ 80 years, CAD, CHF, CVD, peripheral vascular disease, dementia, CKD, COPD, and DM were all found individually to be statistically significant predictors of 5-year mortality (Table 1). Combining aged ≥ 80 years plus CAD, CHF, or dementia all increased the 5-year mortality by a relative risk of > 3 (Table 2).
Discussion
The standard of care is to treat NMSC. Most NMSCs are treated surgically without consideration of patient age or life expectancy.2,4,9,10 A prospective cohort study involving a university-based private practice and a VA medical center in San Francisco found a 22.6% overall 5-year mortality and a 43.3% mortality in the group defined as limited life expectancy (LLE) based on age (≥ 85 years) and medical comorbidities. None died due to the NMSC. Leading cause of death was cardiac, cerebrovascular, and respiratory disease, lung and prostate cancer, and Alzheimer disease. The authors suggested the LLE group may be exposed to wound complications without benefiting from the treatment.4
Another study of 440 patients receiving excision for biopsy-proven facial NMSC at the Roudebush VA Medical Center in Indianapolis, Indiana, found no residual carcinoma in 35.3% of excisions, and in patients aged > 90 years, more than half of the excisions had no residual carcinoma. More than half of the patients aged > 90 years died within 1 year, not as a result of the NMSC. The authors argued for watchful waiting in select patients to maximize comfort and outcomes.10
NMSCs are often asymptomatic and not immediately life threatening. Although NMSCs tend to have a favorable prognosis, studies have found that NMSC may be a marker for other poor health outcomes. A significant increased risk for all-cause mortality was found for patients with a history of SCC, which may be attributed to immune status.11 The aging veteran population has more complex health care needs to be considered when developing surgical treatment plans. These medical problems may limit their life expectancy much sooner than the skin cancer will become symptomatic. We found that individuals aged ≥ 80 years who had CAD, CHF, or dementia had a relative risk of 3 or higher for 5-year mortality. The leading cause of death in the United States in years 2011 to 2015 was heart disease. Alzheimer disease was the sixth leading cause of death in those same years.12-14
Skin cancer excisions do not typically require general anesthesia, deep sedation, or large fluid shifts; however, studies have found that when frail patients undergo low-risk procedures, they tend to have a higher mortality rate than their healthier counterparts.15 Frailty is a concept that identifies patients who are at increased risk of dying in 6 to 60 months due to a decline in their physical reserve. Frail patients have increased rates of perioperative mortality and complications. Various tools have been used to assess the components of physical performance, speed, mobility, nutrition status, mental health, and cognition.16 Frailty screening has been initiated in several VA hospitals, including our own in Gainesville, Florida, with the goal of decreasing postoperative morbidity and mortality in older adult patients.17 The patients are given a 1-page screening assessment that asks about their living situation, medical conditions, nutrition status, cognition, and activities of daily living. The results can trigger the clinician to rethink the surgical plan and mobilize more resources to optimize the patient’s health. This study period precedes the initiative at our institution.
The plastic surgery service’s routine practice is to excise skin cancers in the operating room if sedation or general anesthesia will be needed (Figure 1A), for optimal control of bleeding (Figure 1B) in a patient who cannot safely stop blood thinners, or for excision of a highly vascularized area such as the scalp. Surgery is offered in an office-based setting if the area can be closed primarily, left open to close secondarily, or closed with a small skin graft under local anesthesia only (Figure 2). We prefer treating frail patients in the minor procedure clinic, when possible, to avoid the risks of sedation and the additional preoperative visits and transportation requirements. NMSC with unclear margins (Figure 3A) or in cosmetically sensitive areas where tissue needs to be preserved (Figure 3B) are referred to the Mohs dermatologist. The skin cancers in this study were most frequently found on the face, scalp, hands, and forearms based on referral patterns.
Other treatment options for NMSC include curettage and electrodessication, cryotherapy, and radiation; however, ours is a surgical service and patients are typically referred to us by primary care or dermatology when those are not reasonable or desirable options.18 Published complication rates of patients having skin cancer surgery without age restriction have a rate of 3% to 6%, which is consistent with our study of 5%.19-21 Two bleeding complications that needed to be admitted did not require more than a bedside procedure and neither required transfusions. One patient had been instructed to continue taking coumadin during the perioperative office-based procedure due to a recent carotid stent placement in the setting of a rapidly growing basal cell on an easily accessible location.
The most noted comorbidity in patients with wound complications was found to be DM; however, this was not found to be a statistically significant risk factor for wound complications (P = .10). We do not have a set rule for advising for or against NMSC surgery. We do counsel frail patients and their families that not all cancer is immediately life threatening and will work with them to do whatever makes the most sense to achieve their goals, occasionally accepting positive margins in order to debulk a symptomatic growth. The objective of this paper is to contribute to the discussion of performing invasive procedures on older adult veterans with life-limiting comorbidities. Patients and their families will have different thresholds for what they feel needs intervention, especially if other medical problems are consuming much of their time. We also have the community care referral option for patients whose treatment decisions are being dictated by travel hardships.
Strengths and Limitations
A strength of this study is that the data were obtained from a closed system. Patients tend to stay long-term within the VA and their health record is accessible throughout the country as long as they are seen at a VA facility. Complications, therefore, return to the treating service or primary care, who would route the patient back to the surgeon.
One limitation of the study is that this is a retrospective review from 2011. The authors are limited to data that are recorded in the patient record. Multiple health care professionals saw the patients and notes lack consistency in detail. Size of the lesions were not consistently recorded and did not get logged into our database for that reason.
Conclusions
Treatment of NMSC in older adult patients has a low morbidity but needs to be balanced against a patient and family’s goals when the patient presents with life-limiting comorbidities. An elevated 5-year mortality in patients aged > 80 years with serious unrelated medical conditions is intuitive, but this study may help put treatment plans into perspective for families and health care professionals who want to provide an indicated service while maximizing patient quality of life.
Acknowledgments
This manuscript is the result of work supported with resources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
1. American Cancer Society. Cancer Facts & Figures 2021. Accessed May 26, 2022. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2021/cancer-facts-and-figures-2021.pdf
2. Albert A, Knoll MA, Conti JA, Zbar RIS. Non-melanoma skin cancers in the older patient. Curr Oncol Rep. 2019;21(9):79. Published 2019 Jul 29. doi:10.1007/s11912-019-0828-9
3. Linos E, Chren MM, Stijacic Cenzer I, Covinsky KE. Skin cancer in U.S. elderly adults: does life expectancy play a role in treatment decisions? J Am Geriatr Soc. 2016;64(8):1610-1615. doi:10.1111/jgs.14202
4. Linos E, Parvataneni R, Stuart SE, Boscardin WJ, Landefeld CS, Chren MM. Treatment of nonfatal conditions at the end of life: nonmelanoma skin cancer. JAMA Intern Med. 2013;173(11):1006-1012. doi:10.1001/jamainternmed.2013.639
5. O’Malley KA, Vinson L, Kaiser AP, Sager Z, Hinrichs K. Mental health and aging veterans: how the Veterans Health Administration meets the needs of aging veterans. Public Policy Aging Rep. 2020;30(1):19-23. doi:10.1093/ppar/prz027
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Profile of veterans: 2017. Accessed May 26, 2022. https://www.va.gov/vetdata/docs/SpecialReports/Profile_of_Veterans_2017.pdf 7. Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78(6):1185-1192. doi:10.1016/j.jaad.2017.11.062
8. Clemens MW, Kochuba AL, Carter ME, Han K, Liu J, Evans K. Association between Agent Orange exposure and nonmelanotic invasive skin cancer: a pilot study. Plast Reconstr Surg. 2014;133(2):432-437. doi:10.1097/01.prs.0000436859.40151.cf
9. Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80(2):303-317. doi:10.1016/j.jaad.2018.03.060
10. Chauhan R, Munger BN, Chu MW, et al. Age at diagnosis as a relative contraindication for intervention in facial nonmelanoma skin cancer. JAMA Surg. 2018;153(4):390-392. doi:10.1001/jamasurg.2017.5073
11. Barton V, Armeson K, Hampras S, et al. Nonmelanoma skin cancer and risk of all-cause and cancer-related mortality: a systematic review. Arch Dermatol Res. 2017;309(4):243-251. doi:10.1007/s00403-017-1724-5
12. Kochanek KD, Murphy SL, Xu JQ, Arias E. Mortality in the United States, 2013. NCHS Data Brief 178. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db178.htm
13. Xu JQ, Kochanek KD, Murphy SL, Arias E. Mortality in the United States, 2012. NCHS Data Brief 168. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db168.htm
14. Xu JQ, Murphy SL, Kochanek KD, Arias E. Mortality in the United States, 2015. NCHS Data Brief 267. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db267.htm
15. Varley PR , Borrebach JD, Arya S, et al. Clinical utility of the risk analysis index as a prospective frailty screening tool within a multi-practice, multi-hospital integrated healthcare system. Ann Surg. 2021;274(6):e1230-e1237. doi:10.1097/SLA.0000000000003808
16. Hall DE, Arya S , Schmid KK, et al. Development and initial validation of the risk analysis index for measuring frailty in surgical populations. JAMA Surg. 2017;152(2):175-182. doi:10.1001/jamasurg.2016.4202
17. US Department of Veterans Affairs, Health Services Research & Development. Improving healthcare for aging veterans. Updated August 30, 2017. Accessed May 26, 2022. https://www.hsrd.research.va.gov/news/feature/aging0917.cfm
18. Leus AJG, Frie M, Haisma MS, et al. Treatment of keratinocyte carcinoma in elderly patients – a review of the current literature. J Eur Acad Dermatol Venereol. 2020;34(9):1932-1943. doi:10.1111/jdv.16268
19. Amici JM, Rogues AM, Lasheras A, et al. A prospective study of the incidence of complications associated with dermatological surgery. Br J Dermatol. 2005;153(5):967-971. doi:10.1111/j.1365-2133.2005.06861.x
20. Arguello-Guerra L, Vargas-Chandomid E, Díaz-González JM, Méndez-Flores S, Ruelas-Villavicencio A, Domínguez-Cherit J. Incidence of complications in dermatological surgery of melanoma and non-melanoma skin cancer in patients with multiple comorbidity and/or antiplatelet-anticoagulants. Five-year experience in our hospital. Cir Cir. 2019;86(1):15-23. doi:10.24875/CIRUE.M18000003
21. Keith DJ, de Berker DA, Bray AP, Cheung ST, Brain A, Mohd Mustapa MF. British Association of Dermatologists’ national audit on nonmelanoma skin cancer excision, 2014. Clin Exp Dermatol. 2017;42(1):46-53. doi:10.1111/ced.12990
1. American Cancer Society. Cancer Facts & Figures 2021. Accessed May 26, 2022. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2021/cancer-facts-and-figures-2021.pdf
2. Albert A, Knoll MA, Conti JA, Zbar RIS. Non-melanoma skin cancers in the older patient. Curr Oncol Rep. 2019;21(9):79. Published 2019 Jul 29. doi:10.1007/s11912-019-0828-9
3. Linos E, Chren MM, Stijacic Cenzer I, Covinsky KE. Skin cancer in U.S. elderly adults: does life expectancy play a role in treatment decisions? J Am Geriatr Soc. 2016;64(8):1610-1615. doi:10.1111/jgs.14202
4. Linos E, Parvataneni R, Stuart SE, Boscardin WJ, Landefeld CS, Chren MM. Treatment of nonfatal conditions at the end of life: nonmelanoma skin cancer. JAMA Intern Med. 2013;173(11):1006-1012. doi:10.1001/jamainternmed.2013.639
5. O’Malley KA, Vinson L, Kaiser AP, Sager Z, Hinrichs K. Mental health and aging veterans: how the Veterans Health Administration meets the needs of aging veterans. Public Policy Aging Rep. 2020;30(1):19-23. doi:10.1093/ppar/prz027
6. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Profile of veterans: 2017. Accessed May 26, 2022. https://www.va.gov/vetdata/docs/SpecialReports/Profile_of_Veterans_2017.pdf 7. Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78(6):1185-1192. doi:10.1016/j.jaad.2017.11.062
8. Clemens MW, Kochuba AL, Carter ME, Han K, Liu J, Evans K. Association between Agent Orange exposure and nonmelanotic invasive skin cancer: a pilot study. Plast Reconstr Surg. 2014;133(2):432-437. doi:10.1097/01.prs.0000436859.40151.cf
9. Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80(2):303-317. doi:10.1016/j.jaad.2018.03.060
10. Chauhan R, Munger BN, Chu MW, et al. Age at diagnosis as a relative contraindication for intervention in facial nonmelanoma skin cancer. JAMA Surg. 2018;153(4):390-392. doi:10.1001/jamasurg.2017.5073
11. Barton V, Armeson K, Hampras S, et al. Nonmelanoma skin cancer and risk of all-cause and cancer-related mortality: a systematic review. Arch Dermatol Res. 2017;309(4):243-251. doi:10.1007/s00403-017-1724-5
12. Kochanek KD, Murphy SL, Xu JQ, Arias E. Mortality in the United States, 2013. NCHS Data Brief 178. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db178.htm
13. Xu JQ, Kochanek KD, Murphy SL, Arias E. Mortality in the United States, 2012. NCHS Data Brief 168. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db168.htm
14. Xu JQ, Murphy SL, Kochanek KD, Arias E. Mortality in the United States, 2015. NCHS Data Brief 267. Accessed May 26, 2022. https://www.cdc.gov/nchs/products/databriefs/db267.htm
15. Varley PR , Borrebach JD, Arya S, et al. Clinical utility of the risk analysis index as a prospective frailty screening tool within a multi-practice, multi-hospital integrated healthcare system. Ann Surg. 2021;274(6):e1230-e1237. doi:10.1097/SLA.0000000000003808
16. Hall DE, Arya S , Schmid KK, et al. Development and initial validation of the risk analysis index for measuring frailty in surgical populations. JAMA Surg. 2017;152(2):175-182. doi:10.1001/jamasurg.2016.4202
17. US Department of Veterans Affairs, Health Services Research & Development. Improving healthcare for aging veterans. Updated August 30, 2017. Accessed May 26, 2022. https://www.hsrd.research.va.gov/news/feature/aging0917.cfm
18. Leus AJG, Frie M, Haisma MS, et al. Treatment of keratinocyte carcinoma in elderly patients – a review of the current literature. J Eur Acad Dermatol Venereol. 2020;34(9):1932-1943. doi:10.1111/jdv.16268
19. Amici JM, Rogues AM, Lasheras A, et al. A prospective study of the incidence of complications associated with dermatological surgery. Br J Dermatol. 2005;153(5):967-971. doi:10.1111/j.1365-2133.2005.06861.x
20. Arguello-Guerra L, Vargas-Chandomid E, Díaz-González JM, Méndez-Flores S, Ruelas-Villavicencio A, Domínguez-Cherit J. Incidence of complications in dermatological surgery of melanoma and non-melanoma skin cancer in patients with multiple comorbidity and/or antiplatelet-anticoagulants. Five-year experience in our hospital. Cir Cir. 2019;86(1):15-23. doi:10.24875/CIRUE.M18000003
21. Keith DJ, de Berker DA, Bray AP, Cheung ST, Brain A, Mohd Mustapa MF. British Association of Dermatologists’ national audit on nonmelanoma skin cancer excision, 2014. Clin Exp Dermatol. 2017;42(1):46-53. doi:10.1111/ced.12990
SARS-CoV-2: A Novel Precipitant of Ischemic Priapism
Priapism is a disorder that occurs when the penis maintains a prolonged erection in the absence of appropriate stimulation. The disorder is typically divided into subgroups based on arterial flow: low flow (ischemic) and high flow (nonischemic). Ischemic priapism is the most common form and results from venous congestion due to obstructed outflow and inability of cavernous smooth muscle to contract, resulting in compartment syndrome, tissue hypoxia, hypercapnia, and acidosis.1 Conditions that result in hypercoagulable states and hyperviscosity are associated with ischemic priapism. COVID-19 is well known to cause an acute respiratory illness and systemic inflammatory response and has been increasingly associated with coagulopathy. Studies have shown that 20% to 55% of patients admitted to the hospital for COVID-19 show objective laboratory evidence of a hypercoagulable state.2
To date, there are 6 reported cases of priapism occurring in the setting of COVID-19 with all cases demonstrating the ischemic subtype. The onset of priapism from the beginning of infectious symptoms ranged from 2 days to more than a month. Five of the cases occurred in patients with critical COVID-19 and 1 in the setting of mild disease.3-8 Two critically ill patients did not receive treatment for their ischemic priapism as they were transitioned to expectant management and/or comfort measures.Most were treated with cavernosal blood aspiration and intracavernosal injections of phenylephrine or ethylephrine. Some patients were managed with prophylactic doses of anticoagulation after the identification of priapism; others were transitioned to therapeutic doses. Two patients were followed postdischarge; one patient reported normal nighttime erections with sexual desire 2 weeks postdischarge, and another patient, who underwent a bilateral T-shunt procedure after unsuccessful phenylephrine injections, reported complete erectile dysfunction at 3 months postdischarge.4,7 There was a potentially confounding variable in 2 cases in which propofol infusions were used for sedation management in the setting of mechanical ventilation.6,8 Propofol has been linked to priapism through its blockade of sympathetic activation resulting in persistent relaxation of cavernosal smooth muscle.9 We present a unique case of COVID-19–associated ischemic priapism as our patient had moderate rather than critical COVID-19.
Case Presentation
A 67-year-old male patient presented to the emergency department for a painful erection of 34-hour duration. The patient had been exposed to COVID-19 roughly 2 months prior. Since the exposure, he had experienced headache, nonproductive cough, sore throat, and decreased appetite with weight loss. His medical history included hypertension, thoracic aortic aneurysm, B-cell type chronic lymphocytic leukemia (CLL), and obstructive sleep apnea. Daily outpatient medications included atenolol 100 mg, hydrochlorothiazide 25 mg, and omeprazole 20 mg. The patient stopped tobacco use about 30 years previously. He reported no alcohol consumption or illicit drug use and had no previous episodes of prolonged erection.
The patient was afebrile, hemodynamically stable, and had an oxygen saturation of 92% on room air. Physical examination revealed clear breath sounds and an erect circumcised penis without any lesions, discoloration, or skin necrosis. Laboratory data were remarkable for the following values: 125,660 cells/μL white blood cells (WBCs), 13.82 × 103/ μL neutrophils, 110.58 × 103/μL lymphocytes, 1.26 × 103/μL monocytes, no blasts, 9.4 gm/dL hemoglobin, 100.3 fl mean corpuscular volume, 417,000 cells/μL platelets, 23,671 ng/mL D-dimer, 29.6 seconds activated partial thromboplastin time (aPTT), 16.3 seconds prothrombin time, 743 mg/dL fibrinogen, 474 U/L lactate dehydrogenase, and 202.1 mg/dL haptoglobin. A nasopharyngeal reverse transcription polymerase chain reaction test resulted positive for the SARS-CoV-2 virus, and subsequent chest X-ray revealed bilateral, hazy opacities predominantly in a peripheral distribution. Computed tomography (CT) angiogram of the chest did not reveal pulmonary emboli, pneumothorax, effusions, or lobar consolidation. However, it displayed bilateral ground-glass opacities with interstitial consolidation worst in the upper lobes. Corporal aspiration and blood gas analysis revealed a pH of 7.05, P
Differential Diagnosis
The first consideration in the differential diagnosis of priapism is to differentiate between ischemic and nonischemic. Based on the abnormal blood gas results above, this case clearly falls within the ischemic spectrum. Ischemic priapism secondary to CLL-induced hyperleukocytosis was considered. It has been noted that up to 20% of priapism cases in adults are related to hematologic disorders.10 While it is not uncommon to see hyperleukocytosis (total WBC count > 100 × 109/L) in CLL, leukostasis is rare with most reports demonstrating WBC counts > 1000 × 109/L.11 Hematology, vascular surgery, and urology services were consulted and agreed that ischemic priapism was due to microthrombi or pelvic vein thrombosis secondary to COVID-19–associated coagulopathy (CAC) was the most likely etiology.
Treatment
After corporal aspiration, intracorporal phenylephrine was administered. Diluted phenylephrine (100 ug/mL) was injected every 5 to 10 minutes while intermittently aspirating and irrigating multiple sites along the lateral length of the penile shaft. This initial procedure reduced the erection from 100% to 30% rigidity, with repeat blood gas analysis revealing minimal improvement. CT of the abdomen and pelvis with IV contrast revealed no evidence of pelvic thrombi. A second round of phenylephrine injections were administered, resulting in detumescence. The patient was treated with 2 to 3 L/min of oxygen supplementation via nasal cannula, a 5-day course of remdesivir and low-intensity heparin drip. Following the initial low-intensity heparin drip, the patient transitioned to therapeutic enoxaparin and subsequently was discharged on apixaban for a 3-month course. Since discharge, the patient followed up with hematology. He tolerated and completed the anticoagulation regimen without any recurrences of priapism or residual deficits.
Discussion
Recent studies have overwhelmingly analyzed the incidence and presentation of thrombotic complications in critically ill patients with COVID-19. CAC has been postulated to result from endotheliopathy along with immune cell activation and propagation of coagulation. While COVID-19 has been noted to create lung injury through binding angiotensin-converting enzyme 2 receptors expressed on alveolar pneumocytes, it increasingly has been found to affect endothelial cells throughout the body. Recent postmortem analyses have demonstrated direct viral infection of endothelial cells with consequent diffuse endothelial inflammation, as evidenced by viral inclusions, sequestered immune cells, and endothelial apoptosis.12,13 Manifestations of this endotheliopathy have been delineated through various studies.
An early retrospective study in Wuhan, China, illustrated that 36% of the first 99 patients hospitalized with COVID-19 demonstrated an elevated D-dimer, 6% an elevated aPTT, and 5% an elevated prothrombin time.14 Another retrospective study conducted in Wuhan found a 25% incidence of venous thromboembolic complications in critically ill patients with severe COVID-19.15 In the Netherlands, a study reported the incidence of arterial and venous thrombotic complications to be 31% in 184 critically ill patients with COVID-19, with 81% of these cases involving pulmonary emboli.16
To our knowledge, our patient is the seventh reported case of ischemic priapism occurring in the setting of a COVID-19 infection, and the first to have occurred in its moderate form. Ischemic priapism is often a consequence of penile venous outflow obstruction and resultant stasis of hypoxic blood.7 The prothrombotic state induced by CAC has been proposed to cause the obstruction of small emissary veins in the subtunical space and in turn lead to venous stasis, which propagates the formation of ischemic priapism.8 Furthermore, 4 of the previously reported cases shared laboratory data on their patients, and all demonstrated elevated D-dimer and fibrinogen levels, which strengthens this hypothesis.3,5,7,8 CLL presents a potential confounding variable in this case; however, as we have reviewed earlier, the risk of leukostasis at WBC counts < 1000 × 109/L is very low.11 It is also probable that the patient had some level of immune dysregulation secondary to CLL, leading to his prolonged course and slow clearance of the virus.
Conclusions
Although only a handful of CAC cases leading to ischemic priapism have been reported, the true incidence may be much higher. While our case highlights the importance of considering COVID-19 infection in the differential diagnosis of ischemic priapism, more research is needed to understand incidence and definitively establish a causative relationship.
1. Pryor J, Akkus E, Alter G, et al. Priapism. J Sex Med. 2004;1(1):116-120. doi:10.1111/j.1743-6109.2004.10117.x
2. Lee SG, Fralick M, Sholzberg M. Coagulopathy associated with COVID-19. CMAJ. 2020;192(21):E583. doi:10.1503/cmaj.200685
3. Lam G, McCarthy R, Haider R. A peculiar case of priapism: the hypercoagulable state in patients with severe COVID-19 infection. Eur J Case Rep Intern Med. 2020;7(8):001779. doi:10.12890/2020_001779
4. Addar A, Al Fraidi O, Nazer A, Althonayan N, Ghazwani Y. Priapism for 10 days in a patient with SARS-CoV-2 pneumonia: a case report. J Surg Case Rep. 2021;2021(4):rjab020. doi:10.1093/jscr/rjab020
5. Lamamri M, Chebbi A, Mamane J, et al. Priapism in a patient with coronavirus disease 2019 (COVID-19). Am J Emerg Med. 2021;39:251.e5-251.e7. doi:10.1016/j.ajem.2020.06.027
6. Silverman ML, VanDerVeer SJ, Donnelly TJ. Priapism in COVID-19: a thromboembolic complication. Am J Emerg Med. 2021;45:686.e5-686.e6. doi:10.1016/j.ajem.2020.12.072
7. Giuliano AFM, Vulpi M, Passerini F, et al. SARS-CoV-2 infection as a determining factor to the precipitation of ischemic priapism in a young patient with asymptomatic COVID-19. Case Rep Urol. 2021;2021:9936891. doi:10.1155/2021/9936891
8. Carreno BD, Perez CP, Vasquez D, Oyola JA, Suarez O, Bedoya C. Veno-occlusive priapism in COVID-19 disease. Urol Int. 2021;105(9-10):916-919. doi:10.1159/000514421
9. Senthilkumaran S, Shah S, Ganapathysubramanian, Balamurgan N, Thirumalaikolundusubramanian P. Propofol and priapism. Indian J Pharmacol. 2010;42(4):238-239. doi:10.4103/0253-7613.68430
10. Qu M, Lu X, Wang L, Liu Z, Sun Y, Gao X. Priapism secondary to chronic myeloid leukemia treated by a surgical cavernosa-corpus spongiosum shunt: case report. Asian J Urol. 2019;6(4):373-376. doi:10.1016/j.ajur.2018.12.004
11. Singh N, Singh Lubana S, Dabrowski L, Sidhu G. Leukostasis in chronic lymphocytic leukemia. Am J Case Rep. 2020;21:e924798. doi:10.12659/AJCR.924798
12. Varga Z, Flammer AJ, Steiger P, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417-1418. doi:10.1016/S0140-6736(20)30937-5
13. Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135(23):2033-2040. doi:10.1182/blood.2020006000
14. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507-513. doi:10.1016/S0140-6736(20)30211-7
15. Cui S, Chen S, Li X, Liu S, Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost. 2020;18(6):1421-1424. doi:10.1111/jth.14830
16. Klok FA, Kruip M, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145-147. doi:10.1016/j.thromres.2020.04.013
Priapism is a disorder that occurs when the penis maintains a prolonged erection in the absence of appropriate stimulation. The disorder is typically divided into subgroups based on arterial flow: low flow (ischemic) and high flow (nonischemic). Ischemic priapism is the most common form and results from venous congestion due to obstructed outflow and inability of cavernous smooth muscle to contract, resulting in compartment syndrome, tissue hypoxia, hypercapnia, and acidosis.1 Conditions that result in hypercoagulable states and hyperviscosity are associated with ischemic priapism. COVID-19 is well known to cause an acute respiratory illness and systemic inflammatory response and has been increasingly associated with coagulopathy. Studies have shown that 20% to 55% of patients admitted to the hospital for COVID-19 show objective laboratory evidence of a hypercoagulable state.2
To date, there are 6 reported cases of priapism occurring in the setting of COVID-19 with all cases demonstrating the ischemic subtype. The onset of priapism from the beginning of infectious symptoms ranged from 2 days to more than a month. Five of the cases occurred in patients with critical COVID-19 and 1 in the setting of mild disease.3-8 Two critically ill patients did not receive treatment for their ischemic priapism as they were transitioned to expectant management and/or comfort measures.Most were treated with cavernosal blood aspiration and intracavernosal injections of phenylephrine or ethylephrine. Some patients were managed with prophylactic doses of anticoagulation after the identification of priapism; others were transitioned to therapeutic doses. Two patients were followed postdischarge; one patient reported normal nighttime erections with sexual desire 2 weeks postdischarge, and another patient, who underwent a bilateral T-shunt procedure after unsuccessful phenylephrine injections, reported complete erectile dysfunction at 3 months postdischarge.4,7 There was a potentially confounding variable in 2 cases in which propofol infusions were used for sedation management in the setting of mechanical ventilation.6,8 Propofol has been linked to priapism through its blockade of sympathetic activation resulting in persistent relaxation of cavernosal smooth muscle.9 We present a unique case of COVID-19–associated ischemic priapism as our patient had moderate rather than critical COVID-19.
Case Presentation
A 67-year-old male patient presented to the emergency department for a painful erection of 34-hour duration. The patient had been exposed to COVID-19 roughly 2 months prior. Since the exposure, he had experienced headache, nonproductive cough, sore throat, and decreased appetite with weight loss. His medical history included hypertension, thoracic aortic aneurysm, B-cell type chronic lymphocytic leukemia (CLL), and obstructive sleep apnea. Daily outpatient medications included atenolol 100 mg, hydrochlorothiazide 25 mg, and omeprazole 20 mg. The patient stopped tobacco use about 30 years previously. He reported no alcohol consumption or illicit drug use and had no previous episodes of prolonged erection.
The patient was afebrile, hemodynamically stable, and had an oxygen saturation of 92% on room air. Physical examination revealed clear breath sounds and an erect circumcised penis without any lesions, discoloration, or skin necrosis. Laboratory data were remarkable for the following values: 125,660 cells/μL white blood cells (WBCs), 13.82 × 103/ μL neutrophils, 110.58 × 103/μL lymphocytes, 1.26 × 103/μL monocytes, no blasts, 9.4 gm/dL hemoglobin, 100.3 fl mean corpuscular volume, 417,000 cells/μL platelets, 23,671 ng/mL D-dimer, 29.6 seconds activated partial thromboplastin time (aPTT), 16.3 seconds prothrombin time, 743 mg/dL fibrinogen, 474 U/L lactate dehydrogenase, and 202.1 mg/dL haptoglobin. A nasopharyngeal reverse transcription polymerase chain reaction test resulted positive for the SARS-CoV-2 virus, and subsequent chest X-ray revealed bilateral, hazy opacities predominantly in a peripheral distribution. Computed tomography (CT) angiogram of the chest did not reveal pulmonary emboli, pneumothorax, effusions, or lobar consolidation. However, it displayed bilateral ground-glass opacities with interstitial consolidation worst in the upper lobes. Corporal aspiration and blood gas analysis revealed a pH of 7.05, P
Differential Diagnosis
The first consideration in the differential diagnosis of priapism is to differentiate between ischemic and nonischemic. Based on the abnormal blood gas results above, this case clearly falls within the ischemic spectrum. Ischemic priapism secondary to CLL-induced hyperleukocytosis was considered. It has been noted that up to 20% of priapism cases in adults are related to hematologic disorders.10 While it is not uncommon to see hyperleukocytosis (total WBC count > 100 × 109/L) in CLL, leukostasis is rare with most reports demonstrating WBC counts > 1000 × 109/L.11 Hematology, vascular surgery, and urology services were consulted and agreed that ischemic priapism was due to microthrombi or pelvic vein thrombosis secondary to COVID-19–associated coagulopathy (CAC) was the most likely etiology.
Treatment
After corporal aspiration, intracorporal phenylephrine was administered. Diluted phenylephrine (100 ug/mL) was injected every 5 to 10 minutes while intermittently aspirating and irrigating multiple sites along the lateral length of the penile shaft. This initial procedure reduced the erection from 100% to 30% rigidity, with repeat blood gas analysis revealing minimal improvement. CT of the abdomen and pelvis with IV contrast revealed no evidence of pelvic thrombi. A second round of phenylephrine injections were administered, resulting in detumescence. The patient was treated with 2 to 3 L/min of oxygen supplementation via nasal cannula, a 5-day course of remdesivir and low-intensity heparin drip. Following the initial low-intensity heparin drip, the patient transitioned to therapeutic enoxaparin and subsequently was discharged on apixaban for a 3-month course. Since discharge, the patient followed up with hematology. He tolerated and completed the anticoagulation regimen without any recurrences of priapism or residual deficits.
Discussion
Recent studies have overwhelmingly analyzed the incidence and presentation of thrombotic complications in critically ill patients with COVID-19. CAC has been postulated to result from endotheliopathy along with immune cell activation and propagation of coagulation. While COVID-19 has been noted to create lung injury through binding angiotensin-converting enzyme 2 receptors expressed on alveolar pneumocytes, it increasingly has been found to affect endothelial cells throughout the body. Recent postmortem analyses have demonstrated direct viral infection of endothelial cells with consequent diffuse endothelial inflammation, as evidenced by viral inclusions, sequestered immune cells, and endothelial apoptosis.12,13 Manifestations of this endotheliopathy have been delineated through various studies.
An early retrospective study in Wuhan, China, illustrated that 36% of the first 99 patients hospitalized with COVID-19 demonstrated an elevated D-dimer, 6% an elevated aPTT, and 5% an elevated prothrombin time.14 Another retrospective study conducted in Wuhan found a 25% incidence of venous thromboembolic complications in critically ill patients with severe COVID-19.15 In the Netherlands, a study reported the incidence of arterial and venous thrombotic complications to be 31% in 184 critically ill patients with COVID-19, with 81% of these cases involving pulmonary emboli.16
To our knowledge, our patient is the seventh reported case of ischemic priapism occurring in the setting of a COVID-19 infection, and the first to have occurred in its moderate form. Ischemic priapism is often a consequence of penile venous outflow obstruction and resultant stasis of hypoxic blood.7 The prothrombotic state induced by CAC has been proposed to cause the obstruction of small emissary veins in the subtunical space and in turn lead to venous stasis, which propagates the formation of ischemic priapism.8 Furthermore, 4 of the previously reported cases shared laboratory data on their patients, and all demonstrated elevated D-dimer and fibrinogen levels, which strengthens this hypothesis.3,5,7,8 CLL presents a potential confounding variable in this case; however, as we have reviewed earlier, the risk of leukostasis at WBC counts < 1000 × 109/L is very low.11 It is also probable that the patient had some level of immune dysregulation secondary to CLL, leading to his prolonged course and slow clearance of the virus.
Conclusions
Although only a handful of CAC cases leading to ischemic priapism have been reported, the true incidence may be much higher. While our case highlights the importance of considering COVID-19 infection in the differential diagnosis of ischemic priapism, more research is needed to understand incidence and definitively establish a causative relationship.
Priapism is a disorder that occurs when the penis maintains a prolonged erection in the absence of appropriate stimulation. The disorder is typically divided into subgroups based on arterial flow: low flow (ischemic) and high flow (nonischemic). Ischemic priapism is the most common form and results from venous congestion due to obstructed outflow and inability of cavernous smooth muscle to contract, resulting in compartment syndrome, tissue hypoxia, hypercapnia, and acidosis.1 Conditions that result in hypercoagulable states and hyperviscosity are associated with ischemic priapism. COVID-19 is well known to cause an acute respiratory illness and systemic inflammatory response and has been increasingly associated with coagulopathy. Studies have shown that 20% to 55% of patients admitted to the hospital for COVID-19 show objective laboratory evidence of a hypercoagulable state.2
To date, there are 6 reported cases of priapism occurring in the setting of COVID-19 with all cases demonstrating the ischemic subtype. The onset of priapism from the beginning of infectious symptoms ranged from 2 days to more than a month. Five of the cases occurred in patients with critical COVID-19 and 1 in the setting of mild disease.3-8 Two critically ill patients did not receive treatment for their ischemic priapism as they were transitioned to expectant management and/or comfort measures.Most were treated with cavernosal blood aspiration and intracavernosal injections of phenylephrine or ethylephrine. Some patients were managed with prophylactic doses of anticoagulation after the identification of priapism; others were transitioned to therapeutic doses. Two patients were followed postdischarge; one patient reported normal nighttime erections with sexual desire 2 weeks postdischarge, and another patient, who underwent a bilateral T-shunt procedure after unsuccessful phenylephrine injections, reported complete erectile dysfunction at 3 months postdischarge.4,7 There was a potentially confounding variable in 2 cases in which propofol infusions were used for sedation management in the setting of mechanical ventilation.6,8 Propofol has been linked to priapism through its blockade of sympathetic activation resulting in persistent relaxation of cavernosal smooth muscle.9 We present a unique case of COVID-19–associated ischemic priapism as our patient had moderate rather than critical COVID-19.
Case Presentation
A 67-year-old male patient presented to the emergency department for a painful erection of 34-hour duration. The patient had been exposed to COVID-19 roughly 2 months prior. Since the exposure, he had experienced headache, nonproductive cough, sore throat, and decreased appetite with weight loss. His medical history included hypertension, thoracic aortic aneurysm, B-cell type chronic lymphocytic leukemia (CLL), and obstructive sleep apnea. Daily outpatient medications included atenolol 100 mg, hydrochlorothiazide 25 mg, and omeprazole 20 mg. The patient stopped tobacco use about 30 years previously. He reported no alcohol consumption or illicit drug use and had no previous episodes of prolonged erection.
The patient was afebrile, hemodynamically stable, and had an oxygen saturation of 92% on room air. Physical examination revealed clear breath sounds and an erect circumcised penis without any lesions, discoloration, or skin necrosis. Laboratory data were remarkable for the following values: 125,660 cells/μL white blood cells (WBCs), 13.82 × 103/ μL neutrophils, 110.58 × 103/μL lymphocytes, 1.26 × 103/μL monocytes, no blasts, 9.4 gm/dL hemoglobin, 100.3 fl mean corpuscular volume, 417,000 cells/μL platelets, 23,671 ng/mL D-dimer, 29.6 seconds activated partial thromboplastin time (aPTT), 16.3 seconds prothrombin time, 743 mg/dL fibrinogen, 474 U/L lactate dehydrogenase, and 202.1 mg/dL haptoglobin. A nasopharyngeal reverse transcription polymerase chain reaction test resulted positive for the SARS-CoV-2 virus, and subsequent chest X-ray revealed bilateral, hazy opacities predominantly in a peripheral distribution. Computed tomography (CT) angiogram of the chest did not reveal pulmonary emboli, pneumothorax, effusions, or lobar consolidation. However, it displayed bilateral ground-glass opacities with interstitial consolidation worst in the upper lobes. Corporal aspiration and blood gas analysis revealed a pH of 7.05, P
Differential Diagnosis
The first consideration in the differential diagnosis of priapism is to differentiate between ischemic and nonischemic. Based on the abnormal blood gas results above, this case clearly falls within the ischemic spectrum. Ischemic priapism secondary to CLL-induced hyperleukocytosis was considered. It has been noted that up to 20% of priapism cases in adults are related to hematologic disorders.10 While it is not uncommon to see hyperleukocytosis (total WBC count > 100 × 109/L) in CLL, leukostasis is rare with most reports demonstrating WBC counts > 1000 × 109/L.11 Hematology, vascular surgery, and urology services were consulted and agreed that ischemic priapism was due to microthrombi or pelvic vein thrombosis secondary to COVID-19–associated coagulopathy (CAC) was the most likely etiology.
Treatment
After corporal aspiration, intracorporal phenylephrine was administered. Diluted phenylephrine (100 ug/mL) was injected every 5 to 10 minutes while intermittently aspirating and irrigating multiple sites along the lateral length of the penile shaft. This initial procedure reduced the erection from 100% to 30% rigidity, with repeat blood gas analysis revealing minimal improvement. CT of the abdomen and pelvis with IV contrast revealed no evidence of pelvic thrombi. A second round of phenylephrine injections were administered, resulting in detumescence. The patient was treated with 2 to 3 L/min of oxygen supplementation via nasal cannula, a 5-day course of remdesivir and low-intensity heparin drip. Following the initial low-intensity heparin drip, the patient transitioned to therapeutic enoxaparin and subsequently was discharged on apixaban for a 3-month course. Since discharge, the patient followed up with hematology. He tolerated and completed the anticoagulation regimen without any recurrences of priapism or residual deficits.
Discussion
Recent studies have overwhelmingly analyzed the incidence and presentation of thrombotic complications in critically ill patients with COVID-19. CAC has been postulated to result from endotheliopathy along with immune cell activation and propagation of coagulation. While COVID-19 has been noted to create lung injury through binding angiotensin-converting enzyme 2 receptors expressed on alveolar pneumocytes, it increasingly has been found to affect endothelial cells throughout the body. Recent postmortem analyses have demonstrated direct viral infection of endothelial cells with consequent diffuse endothelial inflammation, as evidenced by viral inclusions, sequestered immune cells, and endothelial apoptosis.12,13 Manifestations of this endotheliopathy have been delineated through various studies.
An early retrospective study in Wuhan, China, illustrated that 36% of the first 99 patients hospitalized with COVID-19 demonstrated an elevated D-dimer, 6% an elevated aPTT, and 5% an elevated prothrombin time.14 Another retrospective study conducted in Wuhan found a 25% incidence of venous thromboembolic complications in critically ill patients with severe COVID-19.15 In the Netherlands, a study reported the incidence of arterial and venous thrombotic complications to be 31% in 184 critically ill patients with COVID-19, with 81% of these cases involving pulmonary emboli.16
To our knowledge, our patient is the seventh reported case of ischemic priapism occurring in the setting of a COVID-19 infection, and the first to have occurred in its moderate form. Ischemic priapism is often a consequence of penile venous outflow obstruction and resultant stasis of hypoxic blood.7 The prothrombotic state induced by CAC has been proposed to cause the obstruction of small emissary veins in the subtunical space and in turn lead to venous stasis, which propagates the formation of ischemic priapism.8 Furthermore, 4 of the previously reported cases shared laboratory data on their patients, and all demonstrated elevated D-dimer and fibrinogen levels, which strengthens this hypothesis.3,5,7,8 CLL presents a potential confounding variable in this case; however, as we have reviewed earlier, the risk of leukostasis at WBC counts < 1000 × 109/L is very low.11 It is also probable that the patient had some level of immune dysregulation secondary to CLL, leading to his prolonged course and slow clearance of the virus.
Conclusions
Although only a handful of CAC cases leading to ischemic priapism have been reported, the true incidence may be much higher. While our case highlights the importance of considering COVID-19 infection in the differential diagnosis of ischemic priapism, more research is needed to understand incidence and definitively establish a causative relationship.
1. Pryor J, Akkus E, Alter G, et al. Priapism. J Sex Med. 2004;1(1):116-120. doi:10.1111/j.1743-6109.2004.10117.x
2. Lee SG, Fralick M, Sholzberg M. Coagulopathy associated with COVID-19. CMAJ. 2020;192(21):E583. doi:10.1503/cmaj.200685
3. Lam G, McCarthy R, Haider R. A peculiar case of priapism: the hypercoagulable state in patients with severe COVID-19 infection. Eur J Case Rep Intern Med. 2020;7(8):001779. doi:10.12890/2020_001779
4. Addar A, Al Fraidi O, Nazer A, Althonayan N, Ghazwani Y. Priapism for 10 days in a patient with SARS-CoV-2 pneumonia: a case report. J Surg Case Rep. 2021;2021(4):rjab020. doi:10.1093/jscr/rjab020
5. Lamamri M, Chebbi A, Mamane J, et al. Priapism in a patient with coronavirus disease 2019 (COVID-19). Am J Emerg Med. 2021;39:251.e5-251.e7. doi:10.1016/j.ajem.2020.06.027
6. Silverman ML, VanDerVeer SJ, Donnelly TJ. Priapism in COVID-19: a thromboembolic complication. Am J Emerg Med. 2021;45:686.e5-686.e6. doi:10.1016/j.ajem.2020.12.072
7. Giuliano AFM, Vulpi M, Passerini F, et al. SARS-CoV-2 infection as a determining factor to the precipitation of ischemic priapism in a young patient with asymptomatic COVID-19. Case Rep Urol. 2021;2021:9936891. doi:10.1155/2021/9936891
8. Carreno BD, Perez CP, Vasquez D, Oyola JA, Suarez O, Bedoya C. Veno-occlusive priapism in COVID-19 disease. Urol Int. 2021;105(9-10):916-919. doi:10.1159/000514421
9. Senthilkumaran S, Shah S, Ganapathysubramanian, Balamurgan N, Thirumalaikolundusubramanian P. Propofol and priapism. Indian J Pharmacol. 2010;42(4):238-239. doi:10.4103/0253-7613.68430
10. Qu M, Lu X, Wang L, Liu Z, Sun Y, Gao X. Priapism secondary to chronic myeloid leukemia treated by a surgical cavernosa-corpus spongiosum shunt: case report. Asian J Urol. 2019;6(4):373-376. doi:10.1016/j.ajur.2018.12.004
11. Singh N, Singh Lubana S, Dabrowski L, Sidhu G. Leukostasis in chronic lymphocytic leukemia. Am J Case Rep. 2020;21:e924798. doi:10.12659/AJCR.924798
12. Varga Z, Flammer AJ, Steiger P, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417-1418. doi:10.1016/S0140-6736(20)30937-5
13. Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135(23):2033-2040. doi:10.1182/blood.2020006000
14. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507-513. doi:10.1016/S0140-6736(20)30211-7
15. Cui S, Chen S, Li X, Liu S, Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost. 2020;18(6):1421-1424. doi:10.1111/jth.14830
16. Klok FA, Kruip M, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145-147. doi:10.1016/j.thromres.2020.04.013
1. Pryor J, Akkus E, Alter G, et al. Priapism. J Sex Med. 2004;1(1):116-120. doi:10.1111/j.1743-6109.2004.10117.x
2. Lee SG, Fralick M, Sholzberg M. Coagulopathy associated with COVID-19. CMAJ. 2020;192(21):E583. doi:10.1503/cmaj.200685
3. Lam G, McCarthy R, Haider R. A peculiar case of priapism: the hypercoagulable state in patients with severe COVID-19 infection. Eur J Case Rep Intern Med. 2020;7(8):001779. doi:10.12890/2020_001779
4. Addar A, Al Fraidi O, Nazer A, Althonayan N, Ghazwani Y. Priapism for 10 days in a patient with SARS-CoV-2 pneumonia: a case report. J Surg Case Rep. 2021;2021(4):rjab020. doi:10.1093/jscr/rjab020
5. Lamamri M, Chebbi A, Mamane J, et al. Priapism in a patient with coronavirus disease 2019 (COVID-19). Am J Emerg Med. 2021;39:251.e5-251.e7. doi:10.1016/j.ajem.2020.06.027
6. Silverman ML, VanDerVeer SJ, Donnelly TJ. Priapism in COVID-19: a thromboembolic complication. Am J Emerg Med. 2021;45:686.e5-686.e6. doi:10.1016/j.ajem.2020.12.072
7. Giuliano AFM, Vulpi M, Passerini F, et al. SARS-CoV-2 infection as a determining factor to the precipitation of ischemic priapism in a young patient with asymptomatic COVID-19. Case Rep Urol. 2021;2021:9936891. doi:10.1155/2021/9936891
8. Carreno BD, Perez CP, Vasquez D, Oyola JA, Suarez O, Bedoya C. Veno-occlusive priapism in COVID-19 disease. Urol Int. 2021;105(9-10):916-919. doi:10.1159/000514421
9. Senthilkumaran S, Shah S, Ganapathysubramanian, Balamurgan N, Thirumalaikolundusubramanian P. Propofol and priapism. Indian J Pharmacol. 2010;42(4):238-239. doi:10.4103/0253-7613.68430
10. Qu M, Lu X, Wang L, Liu Z, Sun Y, Gao X. Priapism secondary to chronic myeloid leukemia treated by a surgical cavernosa-corpus spongiosum shunt: case report. Asian J Urol. 2019;6(4):373-376. doi:10.1016/j.ajur.2018.12.004
11. Singh N, Singh Lubana S, Dabrowski L, Sidhu G. Leukostasis in chronic lymphocytic leukemia. Am J Case Rep. 2020;21:e924798. doi:10.12659/AJCR.924798
12. Varga Z, Flammer AJ, Steiger P, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417-1418. doi:10.1016/S0140-6736(20)30937-5
13. Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135(23):2033-2040. doi:10.1182/blood.2020006000
14. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507-513. doi:10.1016/S0140-6736(20)30211-7
15. Cui S, Chen S, Li X, Liu S, Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost. 2020;18(6):1421-1424. doi:10.1111/jth.14830
16. Klok FA, Kruip M, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145-147. doi:10.1016/j.thromres.2020.04.013
Nodular Sclerosing Hodgkin Lymphoma With Paraneoplastic Cerebellar Degeneration
Paraneoplastic syndrome is a rare disorder involving manifestations of immune dysregulation triggered by malignancy. The immune system develops antibodies to the malignancy, which can cause cross reactivation with various tissues in the body, resulting in an autoimmune response. Paraneoplastic cerebellar degeneration (PCD) is a rare condition caused by immune-mediated damage to the Purkinje cells of the cerebellar tract. Symptoms may include gait instability, double vision, decreased fine motor skills, and ataxia, with progression to brainstem-associated symptoms, such as nystagmus, dysarthria, and dysphagia. Early detection and treatment of the underlying malignancy is critical to halt the progression of autoimmune-mediated destruction. We present a case of a young adult female patient with PCD caused by Purkinje cell cytoplasmic–Tr (PCA-Tr) antibody with Hodgkin lymphoma.
Case Presentation
A 20-year-old previously healthy active-duty female patient presented to the emergency department with acute worsening of chronic intermittent, recurrent episodes of lightheadedness and vertigo. Symptoms persisted for 9 months until acutely worsening over the 2 weeks prior to presentation. She reported left eye double vision but did not report seeing spots, photophobia, tinnitus, or headache. She felt off-balance, leaning on nearby objects to remain standing. Symptoms primarily occurred during ambulation; however, occasionally they happened at rest. Episodes lasted up to several minutes and occurred up to 15 times a day. The patient reported no fever, night sweats, unexplained weight loss, muscle aches, weakness, numbness or tingling, loss of bowel or bladder function, or rash. She had no recent illnesses, changes to medications, or recent travel. Oral intake to include food and water was adequate and unchanged. The patient had a remote history of mild concussions without loss of consciousness while playing sports 4 years previously. She reported no recent trauma. Nine months before, she received treatment for benign paroxysmal positional vertigo (BPPV) with the Epley maneuver with full resolution of symptoms lasting several days. She reported no prescription or over-the-counter medications, herbal remedies, or supplements. She reported no other medical or surgical history and no pertinent social or family history.
Physical examination revealed a nontoxic-appearing female patient with intermittent conversational dysarthria, saccadic pursuits, horizontal nystagmus with lateral gaze, and vertical nystagmus with vertical gaze. The patient exhibited dysdiadochokinesia, or impaired ability to perform rapid alternating hand movements with repetition. Finger-to-nose testing was impaired and heel-to-shin motion remained intact. A Romberg test was positive, and the patient had tandem gait instability. Strength testing, sensation, reflexes, and cranial nerves were otherwise intact. Initial laboratory testing was unremarkable except for mild normocytic anemia. Her infectious workup, including testing for venereal disease, HIV, COVID-19, and Coccidioidies was negative. Heavy metals analysis and urine drug screen were negative. Ophthalmology was consulted and workup revealed small amplitude downbeat nystagmus in primary gaze, sustained gaze evoked lateral beating jerk nystagmus with rebound nystagmus R>L gaze, but there was no evidence of afferent package defect and optic nerve function remained intact. Magnetic resonance imaging of the brain demonstrated cerebellar vermis hypoplasia with prominence of the superior cerebellar folia. Due to concerns for autoimmune encephalitis, a lumbar puncture was performed. Antibody testing revealed PCA-Tr antibodies, which is commonly associated with Hodgkin lymphoma, prompting further evaluation for malignancy.
Computed tomography (CT) of the chest with contrast demonstrated multiple mediastinal masses with a conglomeration of lymph nodes along the right paratracheal region. Further evaluation was performed with a positron emission tomography (PET)–CT, revealing a large conglomeration of hypermetabolic pretracheal, mediastinal, and right supraclavicular lymph that were suggestive of lymphoma. Mediastinoscopy with excisional lymph node biopsy was performed with immunohistochemical staining confirming diagnosis of a nodular sclerosing variant of Hodgkin lymphoma. The patient was treated with IV immunoglobulin at 0.4g/kg daily for 5 days. A central venous catheter was placed into the patient’s right internal jugular vein and a chemotherapy regimen of doxorubicin 46 mg, vinblastine 11 mg, bleomycin 19 units, and dacarbazine 700 mg was initiated. The patient’s symptoms improved with resolution of dysarthria; however, her visual impairment and gait instability persisted. Repeat PET-CT imaging 2 months later revealed interval improvement with decreased intensity and extent of the hypermetabolic lymph nodes and no new hypermetabolic foci.
Discussion
PCA-Tr antibodies affect the delta/notchlike epidermal growth factor–related receptor, expressed on the dendrites of cerebellar Purkinje cells.1 These fibers are the only output neurons of the cerebellar cortex and are critical to the coordination of motor movements, accounting for the ataxia experienced by patients with this subtype of PCD.2 The link between Hodgkin lymphoma and PCA-Tr antibodies has been established; however, most reports involve men with a median age of 61 years with lymphoma-associated symptoms (such as lymphadenopathy) or systemic symptoms (fever, night sweats, or weight loss) preceding neurologic manifestations in 80% of cases.3
Our patient was a young, previously healthy adult female who initially presented with vertigo, a common concern with frequently benign origins. Although there was temporary resolution of symptoms after Epley maneuvers, symptoms recurred and progressed over several months to include brainstem manifestations of nystagmus, diplopia, and dysarthria. Previous reports indicate that after remission of the Hodgkin lymphoma, PCA-Tr antibodies disappear and symptoms can improve or resolve.4,5 Treatment has just begun for our patient and although there has been initial clinical improvement, given the chronicity of symptoms, it is unclear if complete resolution will be achieved.
Conclusions
PCD can result in debilitating neurologic dysfunction and may be associated with malignancy such as Hodgkin lymphoma. This case offers unique insight due to the patient’s demographics and presentation, which involved brainstem pathology typically associated with late-onset disease and preceded by constitutional symptoms. Clinical suspicion of this rare disorder should be considered in all ages, especially if symptoms are progressive or neurologic manifestations arise, as early detection and treatment of the underlying malignancy are paramount to the prevention of significant disability.
1. de Graaff E, Maat P, Hulsenboom E, et al. Identification of delta/notch-like epidermal growth factor-related receptor as the Tr antigen in paraneoplastic cerebellar degeneration. Ann Neurol. 2012;71(6):815-824. doi:10.1002/ana.23550
2. MacKenzie-Graham A, Tiwari-Woodruff SK, Sharma G, et al. Purkinje cell loss in experimental autoimmune encephalomyelitis. Neuroimage. 2009;48(4):637-651. doi:10.1016/j.neuroimage.2009.06.073
3. Bernal F, Shams’ili S, Rojas I, et al. Anti-Tr antibodies as markers of paraneoplastic cerebellar degeneration and Hodgkin’s disease. Neurology. 2003;60(2):230-234. doi:10.1212/01.wnl.0000041495.87539.98
4. Graus F, Ariño H, Dalmau J. Paraneoplastic neurological syndromes in Hodgkin and non-Hodgkin lymphomas. Blood. 2014;123(21):3230-3238. doi:10.1182/blood-2014-03-537506
5. Aly R, Emmady PD. Paraneoplastic cerebellar degeneration. Updated May 8, 2022. Accessed March 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK560638
Paraneoplastic syndrome is a rare disorder involving manifestations of immune dysregulation triggered by malignancy. The immune system develops antibodies to the malignancy, which can cause cross reactivation with various tissues in the body, resulting in an autoimmune response. Paraneoplastic cerebellar degeneration (PCD) is a rare condition caused by immune-mediated damage to the Purkinje cells of the cerebellar tract. Symptoms may include gait instability, double vision, decreased fine motor skills, and ataxia, with progression to brainstem-associated symptoms, such as nystagmus, dysarthria, and dysphagia. Early detection and treatment of the underlying malignancy is critical to halt the progression of autoimmune-mediated destruction. We present a case of a young adult female patient with PCD caused by Purkinje cell cytoplasmic–Tr (PCA-Tr) antibody with Hodgkin lymphoma.
Case Presentation
A 20-year-old previously healthy active-duty female patient presented to the emergency department with acute worsening of chronic intermittent, recurrent episodes of lightheadedness and vertigo. Symptoms persisted for 9 months until acutely worsening over the 2 weeks prior to presentation. She reported left eye double vision but did not report seeing spots, photophobia, tinnitus, or headache. She felt off-balance, leaning on nearby objects to remain standing. Symptoms primarily occurred during ambulation; however, occasionally they happened at rest. Episodes lasted up to several minutes and occurred up to 15 times a day. The patient reported no fever, night sweats, unexplained weight loss, muscle aches, weakness, numbness or tingling, loss of bowel or bladder function, or rash. She had no recent illnesses, changes to medications, or recent travel. Oral intake to include food and water was adequate and unchanged. The patient had a remote history of mild concussions without loss of consciousness while playing sports 4 years previously. She reported no recent trauma. Nine months before, she received treatment for benign paroxysmal positional vertigo (BPPV) with the Epley maneuver with full resolution of symptoms lasting several days. She reported no prescription or over-the-counter medications, herbal remedies, or supplements. She reported no other medical or surgical history and no pertinent social or family history.
Physical examination revealed a nontoxic-appearing female patient with intermittent conversational dysarthria, saccadic pursuits, horizontal nystagmus with lateral gaze, and vertical nystagmus with vertical gaze. The patient exhibited dysdiadochokinesia, or impaired ability to perform rapid alternating hand movements with repetition. Finger-to-nose testing was impaired and heel-to-shin motion remained intact. A Romberg test was positive, and the patient had tandem gait instability. Strength testing, sensation, reflexes, and cranial nerves were otherwise intact. Initial laboratory testing was unremarkable except for mild normocytic anemia. Her infectious workup, including testing for venereal disease, HIV, COVID-19, and Coccidioidies was negative. Heavy metals analysis and urine drug screen were negative. Ophthalmology was consulted and workup revealed small amplitude downbeat nystagmus in primary gaze, sustained gaze evoked lateral beating jerk nystagmus with rebound nystagmus R>L gaze, but there was no evidence of afferent package defect and optic nerve function remained intact. Magnetic resonance imaging of the brain demonstrated cerebellar vermis hypoplasia with prominence of the superior cerebellar folia. Due to concerns for autoimmune encephalitis, a lumbar puncture was performed. Antibody testing revealed PCA-Tr antibodies, which is commonly associated with Hodgkin lymphoma, prompting further evaluation for malignancy.
Computed tomography (CT) of the chest with contrast demonstrated multiple mediastinal masses with a conglomeration of lymph nodes along the right paratracheal region. Further evaluation was performed with a positron emission tomography (PET)–CT, revealing a large conglomeration of hypermetabolic pretracheal, mediastinal, and right supraclavicular lymph that were suggestive of lymphoma. Mediastinoscopy with excisional lymph node biopsy was performed with immunohistochemical staining confirming diagnosis of a nodular sclerosing variant of Hodgkin lymphoma. The patient was treated with IV immunoglobulin at 0.4g/kg daily for 5 days. A central venous catheter was placed into the patient’s right internal jugular vein and a chemotherapy regimen of doxorubicin 46 mg, vinblastine 11 mg, bleomycin 19 units, and dacarbazine 700 mg was initiated. The patient’s symptoms improved with resolution of dysarthria; however, her visual impairment and gait instability persisted. Repeat PET-CT imaging 2 months later revealed interval improvement with decreased intensity and extent of the hypermetabolic lymph nodes and no new hypermetabolic foci.
Discussion
PCA-Tr antibodies affect the delta/notchlike epidermal growth factor–related receptor, expressed on the dendrites of cerebellar Purkinje cells.1 These fibers are the only output neurons of the cerebellar cortex and are critical to the coordination of motor movements, accounting for the ataxia experienced by patients with this subtype of PCD.2 The link between Hodgkin lymphoma and PCA-Tr antibodies has been established; however, most reports involve men with a median age of 61 years with lymphoma-associated symptoms (such as lymphadenopathy) or systemic symptoms (fever, night sweats, or weight loss) preceding neurologic manifestations in 80% of cases.3
Our patient was a young, previously healthy adult female who initially presented with vertigo, a common concern with frequently benign origins. Although there was temporary resolution of symptoms after Epley maneuvers, symptoms recurred and progressed over several months to include brainstem manifestations of nystagmus, diplopia, and dysarthria. Previous reports indicate that after remission of the Hodgkin lymphoma, PCA-Tr antibodies disappear and symptoms can improve or resolve.4,5 Treatment has just begun for our patient and although there has been initial clinical improvement, given the chronicity of symptoms, it is unclear if complete resolution will be achieved.
Conclusions
PCD can result in debilitating neurologic dysfunction and may be associated with malignancy such as Hodgkin lymphoma. This case offers unique insight due to the patient’s demographics and presentation, which involved brainstem pathology typically associated with late-onset disease and preceded by constitutional symptoms. Clinical suspicion of this rare disorder should be considered in all ages, especially if symptoms are progressive or neurologic manifestations arise, as early detection and treatment of the underlying malignancy are paramount to the prevention of significant disability.
Paraneoplastic syndrome is a rare disorder involving manifestations of immune dysregulation triggered by malignancy. The immune system develops antibodies to the malignancy, which can cause cross reactivation with various tissues in the body, resulting in an autoimmune response. Paraneoplastic cerebellar degeneration (PCD) is a rare condition caused by immune-mediated damage to the Purkinje cells of the cerebellar tract. Symptoms may include gait instability, double vision, decreased fine motor skills, and ataxia, with progression to brainstem-associated symptoms, such as nystagmus, dysarthria, and dysphagia. Early detection and treatment of the underlying malignancy is critical to halt the progression of autoimmune-mediated destruction. We present a case of a young adult female patient with PCD caused by Purkinje cell cytoplasmic–Tr (PCA-Tr) antibody with Hodgkin lymphoma.
Case Presentation
A 20-year-old previously healthy active-duty female patient presented to the emergency department with acute worsening of chronic intermittent, recurrent episodes of lightheadedness and vertigo. Symptoms persisted for 9 months until acutely worsening over the 2 weeks prior to presentation. She reported left eye double vision but did not report seeing spots, photophobia, tinnitus, or headache. She felt off-balance, leaning on nearby objects to remain standing. Symptoms primarily occurred during ambulation; however, occasionally they happened at rest. Episodes lasted up to several minutes and occurred up to 15 times a day. The patient reported no fever, night sweats, unexplained weight loss, muscle aches, weakness, numbness or tingling, loss of bowel or bladder function, or rash. She had no recent illnesses, changes to medications, or recent travel. Oral intake to include food and water was adequate and unchanged. The patient had a remote history of mild concussions without loss of consciousness while playing sports 4 years previously. She reported no recent trauma. Nine months before, she received treatment for benign paroxysmal positional vertigo (BPPV) with the Epley maneuver with full resolution of symptoms lasting several days. She reported no prescription or over-the-counter medications, herbal remedies, or supplements. She reported no other medical or surgical history and no pertinent social or family history.
Physical examination revealed a nontoxic-appearing female patient with intermittent conversational dysarthria, saccadic pursuits, horizontal nystagmus with lateral gaze, and vertical nystagmus with vertical gaze. The patient exhibited dysdiadochokinesia, or impaired ability to perform rapid alternating hand movements with repetition. Finger-to-nose testing was impaired and heel-to-shin motion remained intact. A Romberg test was positive, and the patient had tandem gait instability. Strength testing, sensation, reflexes, and cranial nerves were otherwise intact. Initial laboratory testing was unremarkable except for mild normocytic anemia. Her infectious workup, including testing for venereal disease, HIV, COVID-19, and Coccidioidies was negative. Heavy metals analysis and urine drug screen were negative. Ophthalmology was consulted and workup revealed small amplitude downbeat nystagmus in primary gaze, sustained gaze evoked lateral beating jerk nystagmus with rebound nystagmus R>L gaze, but there was no evidence of afferent package defect and optic nerve function remained intact. Magnetic resonance imaging of the brain demonstrated cerebellar vermis hypoplasia with prominence of the superior cerebellar folia. Due to concerns for autoimmune encephalitis, a lumbar puncture was performed. Antibody testing revealed PCA-Tr antibodies, which is commonly associated with Hodgkin lymphoma, prompting further evaluation for malignancy.
Computed tomography (CT) of the chest with contrast demonstrated multiple mediastinal masses with a conglomeration of lymph nodes along the right paratracheal region. Further evaluation was performed with a positron emission tomography (PET)–CT, revealing a large conglomeration of hypermetabolic pretracheal, mediastinal, and right supraclavicular lymph that were suggestive of lymphoma. Mediastinoscopy with excisional lymph node biopsy was performed with immunohistochemical staining confirming diagnosis of a nodular sclerosing variant of Hodgkin lymphoma. The patient was treated with IV immunoglobulin at 0.4g/kg daily for 5 days. A central venous catheter was placed into the patient’s right internal jugular vein and a chemotherapy regimen of doxorubicin 46 mg, vinblastine 11 mg, bleomycin 19 units, and dacarbazine 700 mg was initiated. The patient’s symptoms improved with resolution of dysarthria; however, her visual impairment and gait instability persisted. Repeat PET-CT imaging 2 months later revealed interval improvement with decreased intensity and extent of the hypermetabolic lymph nodes and no new hypermetabolic foci.
Discussion
PCA-Tr antibodies affect the delta/notchlike epidermal growth factor–related receptor, expressed on the dendrites of cerebellar Purkinje cells.1 These fibers are the only output neurons of the cerebellar cortex and are critical to the coordination of motor movements, accounting for the ataxia experienced by patients with this subtype of PCD.2 The link between Hodgkin lymphoma and PCA-Tr antibodies has been established; however, most reports involve men with a median age of 61 years with lymphoma-associated symptoms (such as lymphadenopathy) or systemic symptoms (fever, night sweats, or weight loss) preceding neurologic manifestations in 80% of cases.3
Our patient was a young, previously healthy adult female who initially presented with vertigo, a common concern with frequently benign origins. Although there was temporary resolution of symptoms after Epley maneuvers, symptoms recurred and progressed over several months to include brainstem manifestations of nystagmus, diplopia, and dysarthria. Previous reports indicate that after remission of the Hodgkin lymphoma, PCA-Tr antibodies disappear and symptoms can improve or resolve.4,5 Treatment has just begun for our patient and although there has been initial clinical improvement, given the chronicity of symptoms, it is unclear if complete resolution will be achieved.
Conclusions
PCD can result in debilitating neurologic dysfunction and may be associated with malignancy such as Hodgkin lymphoma. This case offers unique insight due to the patient’s demographics and presentation, which involved brainstem pathology typically associated with late-onset disease and preceded by constitutional symptoms. Clinical suspicion of this rare disorder should be considered in all ages, especially if symptoms are progressive or neurologic manifestations arise, as early detection and treatment of the underlying malignancy are paramount to the prevention of significant disability.
1. de Graaff E, Maat P, Hulsenboom E, et al. Identification of delta/notch-like epidermal growth factor-related receptor as the Tr antigen in paraneoplastic cerebellar degeneration. Ann Neurol. 2012;71(6):815-824. doi:10.1002/ana.23550
2. MacKenzie-Graham A, Tiwari-Woodruff SK, Sharma G, et al. Purkinje cell loss in experimental autoimmune encephalomyelitis. Neuroimage. 2009;48(4):637-651. doi:10.1016/j.neuroimage.2009.06.073
3. Bernal F, Shams’ili S, Rojas I, et al. Anti-Tr antibodies as markers of paraneoplastic cerebellar degeneration and Hodgkin’s disease. Neurology. 2003;60(2):230-234. doi:10.1212/01.wnl.0000041495.87539.98
4. Graus F, Ariño H, Dalmau J. Paraneoplastic neurological syndromes in Hodgkin and non-Hodgkin lymphomas. Blood. 2014;123(21):3230-3238. doi:10.1182/blood-2014-03-537506
5. Aly R, Emmady PD. Paraneoplastic cerebellar degeneration. Updated May 8, 2022. Accessed March 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK560638
1. de Graaff E, Maat P, Hulsenboom E, et al. Identification of delta/notch-like epidermal growth factor-related receptor as the Tr antigen in paraneoplastic cerebellar degeneration. Ann Neurol. 2012;71(6):815-824. doi:10.1002/ana.23550
2. MacKenzie-Graham A, Tiwari-Woodruff SK, Sharma G, et al. Purkinje cell loss in experimental autoimmune encephalomyelitis. Neuroimage. 2009;48(4):637-651. doi:10.1016/j.neuroimage.2009.06.073
3. Bernal F, Shams’ili S, Rojas I, et al. Anti-Tr antibodies as markers of paraneoplastic cerebellar degeneration and Hodgkin’s disease. Neurology. 2003;60(2):230-234. doi:10.1212/01.wnl.0000041495.87539.98
4. Graus F, Ariño H, Dalmau J. Paraneoplastic neurological syndromes in Hodgkin and non-Hodgkin lymphomas. Blood. 2014;123(21):3230-3238. doi:10.1182/blood-2014-03-537506
5. Aly R, Emmady PD. Paraneoplastic cerebellar degeneration. Updated May 8, 2022. Accessed March 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK560638
Hidradenitis Suppurativa: Workup
Hidradenitis Suppurativa Treatment
Chest pain and difficulty swallowing
The posteroanterior (PA) and lateral view CXRs, as well as the CT scan, revealed the presence of retrosternal air and confirmed the patient’s diagnosis of pneumomediastinum.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by the Hamman sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
Pneumomediastinum is generally a self-limiting condition. Thus, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home. If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation.3 If the result of this test is abnormal, a follow-up study with barium is recommended.3
The patient underwent both swallow studies, and they were negative. This patient received subsequent serial CXRs that showed improvement in the pneumomediastinum. Once the patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged (after a 3-day hospitalization) with close follow-up. One week later, her pain had almost entirely resolved.
This case was adapted from: Gawrys B, Shaha D. Pleuritic chest pain and globus pharyngeus. J Fam Pract. 2015;64:305-307.
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Elsevier Health Sciences; 2005:2039-2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057. doi: 10.1148/radiographics.20.4.g00jl13104
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334. doi: 10.1016/j.rmed.2008.03.023

The posteroanterior (PA) and lateral view CXRs, as well as the CT scan, revealed the presence of retrosternal air and confirmed the patient’s diagnosis of pneumomediastinum.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by the Hamman sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
Pneumomediastinum is generally a self-limiting condition. Thus, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home. If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation.3 If the result of this test is abnormal, a follow-up study with barium is recommended.3
The patient underwent both swallow studies, and they were negative. This patient received subsequent serial CXRs that showed improvement in the pneumomediastinum. Once the patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged (after a 3-day hospitalization) with close follow-up. One week later, her pain had almost entirely resolved.
This case was adapted from: Gawrys B, Shaha D. Pleuritic chest pain and globus pharyngeus. J Fam Pract. 2015;64:305-307.

The posteroanterior (PA) and lateral view CXRs, as well as the CT scan, revealed the presence of retrosternal air and confirmed the patient’s diagnosis of pneumomediastinum.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by the Hamman sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
Pneumomediastinum is generally a self-limiting condition. Thus, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home. If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation.3 If the result of this test is abnormal, a follow-up study with barium is recommended.3
The patient underwent both swallow studies, and they were negative. This patient received subsequent serial CXRs that showed improvement in the pneumomediastinum. Once the patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged (after a 3-day hospitalization) with close follow-up. One week later, her pain had almost entirely resolved.
This case was adapted from: Gawrys B, Shaha D. Pleuritic chest pain and globus pharyngeus. J Fam Pract. 2015;64:305-307.
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Elsevier Health Sciences; 2005:2039-2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057. doi: 10.1148/radiographics.20.4.g00jl13104
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334. doi: 10.1016/j.rmed.2008.03.023
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Elsevier Health Sciences; 2005:2039-2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057. doi: 10.1148/radiographics.20.4.g00jl13104
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334. doi: 10.1016/j.rmed.2008.03.023
2022 Update on menopause
This year’s Menopause Update focuses on 2 menopause-related issues relevant to ObGyns and our menopausal patients:
- choosing the safest regimens, particularly with respect to risk of breast cancer, when prescribing hormone therapy (HT) to menopausal women
- reviewing the risks and benefits of premenopausal bilateral salpingo-oophorectomy and the pros and cons of replacement HT in surgically menopausal patients.
We hope that you find this updated information useful as you care for menopausal women.
Revisiting menopausal HT and the risk of breast cancer: What we know now
Abenhaim HA, Suissa S, Azoulay L, et al. Menopausal hormone therapy formulation and breast cancer risk. Obstet Gynecol. 2022;139:1103-1110. doi: 10.1097/AOG.0000000000004723.
Reevaluation of the Women’s Health Initiative randomized controlled trials (WHI RCTs), long-term (median follow-up more than 20 years) cumulative follow-up data, and results from additional studies have suggested that estrogen therapy (ET) alone in menopausal women with prior hysterectomy does not increase the risk of breast cancer. By contrast, estrogen with progestin (synthetic progestogens that include medroxyprogesterone acetate [MPA] and norethindrone acetate) slightly increases the risk of breast cancer. In the past 10 years, several publications have shed light on whether the type of progestogen affects the risk of breast cancer and can help provide evidence-based information to guide clinicians.
Breast cancer risk with combined HT and synthetic progestin
In the first part of the WHI RCT, women were randomly assigned to receive either conjugated equine estrogen (CEE) plus synthetic progestin (MPA) or a placebo. Combined estrogen-progestin therapy (EPT) was associated with a modestly elevated risk of breast cancer.1 In the second part of the WHI trial, CEE only (estrogen alone, ET) was compared with placebo among women with prior hysterectomy, with no effect found on breast cancer incidence.2
Most older observational studies published in 2003 to 2005 found that neither CEE nor estradiol appeared to increase the risk of breast cancer when used alone.3-5 However, estrogen use in combination with synthetic progestins (MPA, norethindrone, levonorgestrel, and norgestrel) has been associated with an increased risk of breast cancer,4,6 while the elevated risk of breast cancer with micronized progesterone has been less substantial.7,8
Continue to: Newer data suggest the type of progestogen used affects risk...
Newer data suggest the type of progestogen used affects risk
In a report published in the June 2022 issue of Obstetrics and Gynecology, Abenhaim and colleagues used a nested population-based case-control study of administrative data available in the UK Clinical Practice Research Datalink and provider prescriptions to evaluate the additive effect on the risk of breast cancer of the type of progestogen (micronized progesterone or synthetic progestins) when combined with estradiol for the treatment of menopausal symptoms.9 A cohort of 561,379 women was included in the case-control study (10:1 ratio), 43,183 in the case group (patients diagnosed with invasive breast cancer), and 431,830 in the matched control group.
Overall, in the stratified analysis, a small but significant increase in the risk of breast cancer was found in ever users of menopausal HT (odds ratio [OR], 1.12; 95% confidence interval [CI], 1.09–1.15). Neither estradiol (OR, 1.04; 95% CI, 1.00–1.09) nor CEE (OR, 1.01; 95% CI, 0.96–1.06) was associated with an elevated risk of being diagnosed with invasive breast cancer. Of note, no elevated risk of breast cancer was associated with combination estrogen-progesterone therapy. However, the risk of breast cancer for women who had used synthetic progestins, mostly MPA, was significantly elevated (OR, 1.28; 95% CI, 1.22-1.35). Notably, this modestly elevated odds ratio with the use of estrogen-progestin HT is almost identical to that observed with CEE/ MPA in the WHI.1 Similar findings were found in women aged 50 to 60 years.
The adjusted analyses from the large WHI RCTs provide additional support: the synthetic progestin MPA combined with CEE showed a higher risk of breast cancer than CEE alone in women with prior hysterectomy.10
In the long-term follow-up of the WHI RCTs, after a median of 20.3 years postrandomization, prior randomization to CEE alone for postmenopausal women with prior hysterectomy was associated with a significantly lowered risk of breast cancer incidence and mortality.11 By contrast, prior randomization to CEE plus MPA (EPT) for women with an intact uterus was associated with a small but significantly increased incidence of breast cancer but no significant difference in breast cancer mortality.
In the French E3N EPIC population-based prospective cohort study, Fournier and colleagues4,5 found that women who received estrogen combined with synthetic progestins (mostly MPA) had a higher risk of breast cancer, with an age-adjusted relative risk of 1.4 (95% CI, 1.2–1.7), a finding not seen in women who received estrogen combined with micronized progesterone, similar to findings by Cordina-Duverger and colleagues and Simin and colleagues.12,13 In the E3N study, only 948 women were identified with breast cancer; 268 of these had used synthetic progestins.4,5
Both the Abenhaim cohort9 and the longterm outcomes of WHI RCT trial data11 found a significant contributing effect of MPA (synthetic progestin) in the risk of breast cancer. Progestogens are not thought to exert a class effect. Although it is clear that progestogens (progesterone or progestins) prevent estrogeninduced endometrial neoplasia when dosed adequately, different types of progestogens have a differential risk of breast epithelium proliferation and carcinogenic potential.14 A systematic review by Stute and colleagues found that micronized progesterone did not appear to alter mammographic breast density assessments or breast biopsy results.15
Progesterone capsules, available in generic form in 100-mg and 200-mg doses, are formulated with peanut oil, and they should be taken at bedtime as progesterone can induce drowsiness.
When combined with standard-dose estrogen, including oral estradiol 1.0 mg, transdermal estradiol 0.05 mg, or oral conjugated equine estrogen 0.625 mg, the appropriate dose of progesterone is 100 mg if used continuously or 200 mg if used as cyclic therapy. With higher doses of estrogen, progesterone 200 mg should be taken continuously.
An oral formulation that combines estradiol 1 mg and progesterone 100 mg does not contain peanut oil and, accordingly, can be used safely by those with peanut allergies. This combination product is marketed under the name Bijuva (TherapeuticsMD, Boca Raton, Florida).1
Reference
1. Lobo RA, Archer DF, Kagan R, et al. A 17β-estradiol-progesterone oral capsule for vasomotor symptoms in postmenopausal women: a randomized controlled trial. Obstet Gynecol. 2018;132:161-170. doi: 10.1097/AOG.0000000000002645. Erratum in: Obstet Gynecol. 2018;132:786.
Race considerations
The study by Abenhaim and colleagues was unable to address the issues of race or ethnicity.9 However, in the racially diverse WHI trial of women with prior hysterectomy, estrogen-alone use significantly reduced breast cancer incidence in all participants.10,16 Post hoc analysis of the 1,616 Black women with prior hysterectomy in the WHI RCT showed a significantly decreased breast cancer incidence with use of estrogen alone (hazard ratio [HR], 0.47; 95% CI, 0.26–0.82).1 When race was evaluated in the long-term cumulative follow-up of the WHI trial, estrogen-alone use significantly reduced breast cancer incidence in Black women, with no adverse effect on coronary heart disease, global index, or all-cause mortality, and with fewer cases of venous thromboembolism.17 The global index findings were favorable for Black women in their 50s and those with vasomotor symptoms.
Continue to: Impact of HT in women with an elevated risk of breast cancer...
Impact of HT in women with an elevated risk of breast cancer
Abenhaim and colleagues could not evaluate the effect of HT in women with a baseline elevated risk of breast cancer.9 For these women, HT may be recommended after premature surgical menopause due to increased risks for coronary heart disease, osteoporosis, genitourinary syndrome of menopause, and cognitive changes when estrogen is not taken postsurgery through to at least the average age of menopause, considered age 51.18,19
Marchetti and colleagues reviewed 3 clinical trials that assessed breast cancer events in 1,100 BRCA gene mutation carriers with intact breasts who underwent risk-reducing salpingo-oophorectomy (RRSO) who used or did not use HT.20 For BRCA1 and BRCA2 mutation carriers who received HT after RRSO, no elevated risk of breast cancer risk was seen (HR, 0.98; 95% CI, 0.63–1.52). There was a nonsignificant reduction in breast cancer risk for the estrogen-alone users compared with EPT HT (OR, 0.53; 95% CI, 0.25–1.15). Thus, short-term use of HT, estrogen alone or EPT, does not appear to elevate the risk of breast cancer after RRSO in these high-risk women.
Individualizing HT for menopausal symptoms
The data presented provide reassuring evidence that longer-term use of ET does not appear to increase breast cancer risk, regardless of the type of estrogen (CEE or estradiol).4,5,9,11 For women with a uterus, micronized progesterone has less (if any) effect on breast cancer risk. By contrast, the use of synthetic progestins (such as MPA), when combined with estrogen, has been associated with a small but real increased breast cancer risk.
The most evident benefit of HT is in treating vasomotor symptoms and preventing bone loss for those at elevated risk in healthy women without contraindications who initiate systemic HT when younger than age 60 or within 10 years of menopause onset. Benefit and risk ratio depends on age and time from menopause onset when HT is initiated. Hormone therapy safety varies depending on type, dose, duration, route of administration, timing of initiation, and whether, and type, of progestogen is used. Transdermal estradiol, particularly when dosed at 0.05 mg or less, has been shown to have less thrombotic and stroke risk than oral estrogen.21
Individualizing treatment includes using the best available evidence to maximize benefits and minimize risks, with periodic reevaluation of benefits and risks of continuing or discontinuing HT or changing to lower doses. ObGyns who follow best practices in prescribing systemic HT can now help menopausal patients with bothersome symptoms take advantage of systemic HT’s benefits while providing reassurance regarding menopausal HT’s safety.18 Transdermal therapy is a safer option for women at elevated baseline risk of venous thrombosis (for example, obese women) and older patients. Likewise, given its safety with respect to risk of breast cancer, the use of micronized progesterone over synthetic progestins should be considered when prescribing EPT to women with an intact uterus.
We can replace fear of HT with evidence-based discussions.22 For women with prior hysterectomy who have menopausal symptoms that impact their quality of life, ET at menopause does not appear to increase the risk of breast cancer. For women with an intact uterus who are considering use of estrogen and progestogen, extended-duration use of combination HT with synthetic progestins slightly elevates the risk of breast cancer, while the use of micronized progesterone does not appear to elevate breast cancer risk. Likewise, transdermal estrogen does not appear to elevate thrombosis risk.
Continue to: Benefits of avoiding BSO in women at average risk of ovarian cancer...
Benefits of avoiding BSO in women at average risk of ovarian cancer
Erickson Z, Rocca WA, Smith CY, et al. Time trends in unilateral and bilateral oophorectomy in a geographically defined American population. Obstet Gynecol. 2022;139:724-734. doi: 10.1097/ AOG.0000000000004728.
In 2005, gynecologist William Parker, MD, and colleagues used modeling methodology to assess the long-term risks and benefits of performing bilateral salpingo-oophorectomy (BSO) at the time of hysterectomy for benign disease in women at average risk for ovarian cancer.23 They concluded that practicing ovarian conservation until age 65 increased women’s long-term survival. Among their findings were that women with BSO before age 55 had an 8.6% excess overall mortality by age 80, while those with oophorectomy before age 59 had 3.9% excess mortality. They noted a sustained, but decreasing, mortality benefit until the age of 75 and stated that at no age did their model suggest higher mortality in women who chose ovarian conservation. Parker and colleagues concluded that ovarian conservation until at least age 65 benefited long-term survival for women at average risk for ovarian cancer when undergoing hysterectomy for benign disease.23
Certain risks decreased, others increased
A second report in 2009 by Parker and colleagues from the large prospective Nurses’ Health Study found that, while BSO at the time of hysterectomy for benign disease was associated with a decreased risk of breast and ovarian cancer, BSO was associated with an increased risk of all-cause mortality, fatal and nonfatal coronary heart disease, and lung cancer.24 Similar to the findings of the 2005 report, the authors noted that in no analysis or age group was BSO associated with increased survival. They also noted that compared with those who underwent BSO before age 50 and used ET, women with no history of ET use had an approximately 2-fold elevated risk of new onset coronary heart disease (HR, 1.98; 95% CI, 1.18–3.32).24
In 2007, Walter Rocca, MD, a Mayo Clinic neurologist with a particular interest in the epidemiology of dementia, and colleagues at the Mayo Clinic published results of a study that assessed a cohort of women who had undergone unilateral oophorectomy or BSO prior to the onset of menopause.25 The risk of cognitive impairment or dementia was higher in these women compared with women who had intact ovaries (HR, 1.46; 95% CI, 1.13-1.90). Of note, this elevated risk was confined to those who underwent oophorectomy before 49 years of age and were not prescribed estrogen until age 50 or older.25
In a subsequent publication, Rocca and colleagues pointed out that BSO prior to menopause not only is associated with higher rates of all-cause mortality and cognitive impairment but also with coronary heart disease, parkinsonism, osteoporosis, and other chronic conditions associated with aging, including metabolic, mental health, and arthritic disorders.26
Oophorectomy trends tracked
Given these and other reports27 that highlighted the health risks of premenopausal BSO in women at average risk for ovarian cancer, Rocca and colleagues recently assessed trends in the occurrence of unilateral oophorectomy or BSO versus ovarian conservation among all women residing in the Minnesota county (Olmsted) in which Mayo Clinic is located, and who underwent gynecologic surgery between 1950 and 2018.28
The investigators limited their analysis to women who had undergone unilateral oophorectomy or BSO between ages 18 and 49 years (these women are assumed to have been premenopausal). The authors considered as indications for oophorectomy primary or metastatic ovarian cancer, risk-reducing BSO for women at elevated risk for ovarian cancer (for example, strong family history or known BRCA gene mutation), adnexal mass, endometriosis, torsion, and other benign gynecologic conditions that included pelvic pain, abscess, oophoritis, or ectopic pregnancy. When more than 1 indication for ovarian surgery was present, the authors used the most clinically important indication. Unilateral oophorectomy or BSO was considered not indicated if the surgery was performed during another primary procedure (usually hysterectomy) without indication, or if the surgeon referred to the ovarian surgery as elective.
Results. Among 5,154 women who had oophorectomies between 1950 and 2018, the proportion of these women who underwent unilateral oophorectomy and BSO was 40.6% and 59.4%, respectively.
For most years between 1950 and 1979, the incidence of unilateral oophorectomy was higher than BSO. However, from 1980 to 2004, the incidence of BSO increased more than 2-fold while the incidence of unilateral surgery declined. After 2005, however, both types of ovarian surgery declined. During the years 2005–2018, a marked decline in BSO occurred, with the reduced incidence in premenopausal BSO most notable among women undergoing hysterectomy or those without an indication for oophorectomy.
Historically, ObGyns were taught that the benefits of removing normal ovaries (to prevent ovarian cancer) in average-risk women at the time of hysterectomy outweighed the risks. We agree with the authors’ speculation that beginning with Parker’s 2005 publication,23 ObGyns have become more conservative in performing unindicated BSO in women at average risk for ovarian cancer, now recognizing that the harms of this procedure often outweigh any benefits.28
Women with BRCA1 and BRCA2 gene mutations are at elevated risk for ovarian, tubal, and breast malignancies. In this population, risk-reducing BSO dramatically lowers future risk of ovarian and tubal cancer.
Data addressing the effect of RRSO in BRCA1 and BRCA2 gene mutation carriers continue to be evaluated, with differences between the 2 mutations, but they suggest that the surgery reduces not only ovarian cancer and tubal cancer but also possibly breast cancer.29
Many of our patients are fearful regarding the possibility that they could be diagnosed with breast or ovarian cancer, and in their minds, fears regarding these 2 potentially deadly diseases outweigh concerns about more common causes of death in women, including cardiovascular disease. Accordingly, counseling women at average risk for ovarian cancer who are planning hysterectomy for benign indications can be challenging. In recent years, ObGyns have increasingly been performing opportunistic bilateral salpingectomy (OS) in women at average risk of ovarian cancer at the time of hysterectomy for benign disease. It is important to note that the studies we refer to in this Update addressed BSO, not OS. We hope that the findings we have reviewed here assist clinicians in helping women to understand the risks and benefits associated with premenopausal BSO and the need to discuss the pros and cons of HT for these women before surgery.
Continue to: Trends show decline in ET use in surgically menopausal women...
Trends show decline in ET use in surgically menopausal women
Suzuki Y, Huang Y, Melamed A, et al. Use of estrogen therapy after surgical menopause in women who are premenopausal. Obstet Gynecol. 2022;139:756-763. doi: 10.1097/AOG.0000000000004762.
In addition to highlighting the risks associated with premenopausal BSO in women at average risk for ovarian cancer, the reports referred to above also underscore that the use of replacement menopausal HT in premenopausal women who undergo BSO prevents morbidity and mortality that otherwise accompanies surgical menopause. In addition, the North American Menopause Society (NAMS) recommends replacement menopausal HT in the setting of induced early menopause when no contraindications are present.18
To assess the prevalence of HT use in surgically menopausal women, investigators at Columbia University College of Physicians and Surgeons used a national database that captures health insurance claims for some 280 million US patients, focusing on women aged 18 to 50 years who underwent BSO from 2008 to 2019.30 The great majority of women in this database have private insurance. Although the authors used the term estrogen therapy in their article, this term refers to systemic estrogen alone or with progestogen, as well as vaginal ET (personal communication with Jason Wright, MD, a coauthor of the study, May 19, 2022). In this Update section, we use the term HT to include use of any systemic HT or vaginal estrogen.
Prevalence of HT use changed over time period and patient age range
Among almost 61,980 evaluable women who had undergone BSO (median age, 45 years; 75.1% with concomitant hysterectomy; median follow-up time, 27 months), with no history of gynecologic or breast cancer, HT was used within 3 years of BSO by 64.5%. The highest percentage of women in this cohort who used HT peaked in 2008 (69.5%), declining to 58.2% by 2016. The median duration of HT use was 5.3 months. The prevalence of HT use 3 years after BSO declined with age, from 79.1% in women aged 18–29 to 60.0% in women aged 45–50.30
This report, published in the June 2022 issue of Obstetrics and Gynecology, makes several sobering observations: Many surgically menopausal women aged 50 years and younger are not prescribed HT, the proportion of such women receiving a prescription for HT is declining over time, and the duration of HT use following BSO is short. ●
As ObGyn physicians, we can play an important role by educating healthy women with induced menopause who are younger than the average age of spontaneous menopause, and who have no contraindications, that the benefits of HT far outweigh risks. Many of these women will benefit from longer-term HT, using doses substantially higher than are used in women who undergo spontaneous menopause.31,32 After reaching the age of menopause, healthy women without contraindications may continue to benefit from HT into their 50s or beyond if they have vasomotor symptoms, bone loss, or other indications for treatment.18,19
- Chlebowski RT, Hendrix SL, Langer RD, et al; WHI Investigators. Influence of estrogen plus progestin on breast cancer and mammography in healthy postmenopausal women: the Women’s Health Initiative randomized trial. JAMA. 2003;289:3243-3253. doi: 10.1001/jama.289.24.3243.
- Anderson GL, Limacher M, Assaf AR, et al; Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized controlled trial. JAMA. 2004;291:1701-1712. doi: 10.1001/jama.291.14.1701.
- Opatrny L, Dell’Aniello S, Assouline S, et al. Hormone replacement therapy use and variations in the risk of breast cancer. BJOG. 2008;115:169-175. doi: 10.1111/j.14710528.2007.01520.x.
- Fournier A, Berrino F, Riboli E, et al. Breast cancer risk in relation to different types of hormone replacement therapy in the E3N-EPIC cohort. Int J Cancer. 2005;114:448-454. doi: 10.1002/ijc.20710.
- Fournier A, Berrino F, Clavel-Chapelon F. Unequal risks for breast cancer associated with different hormone replacement therapies: results from the E3N cohort study. Breast Cancer Res Treat. 2008;107:103-111. doi: 10.1007/s10549-007-9523-x.
- Beral V; Million Women Study Collaborators. Breast cancer and hormone-replacement therapy in the million women study. Lancet. 2003;362:419–27. doi: 10.1016/s01406736(03)14065-2.
- Yang Z, Hu Y, Zhang J, et al. Estradiol therapy and breast cancer risk in perimenopausal and postmenopausal women: a systematic review and meta-analysis. Gynecol Endocrinol. 2017;33:87-92. doi: 10.1080/09513590.2016.1248932.
- Asi N, Mohammed K, Haydour Q, et al. Progesterone vs synthetic progestins and the risk of breast cancer: a systematic review and meta-analysis. Syst Rev. 2016;5:121. doi: 10.1186/ s13643-016-0294-5.
- Abenhaim HA, Suissa S, Azoulay L, et al. Menopausal hormone therapy formulation and breast cancer risk. Obstet Gynecol. 2022;139:1103-1110. doi: 10.1097/AOG.0000000000004723.
- Chlebowski RT, Rohan TE, Manson JE, et al. Breast cancer after use of estrogen plus progestin and estrogen alone: analyses of data from 2 Women’s Health Initiative randomized clinical trials. JAMA Oncol. 2015;1:296-305. doi: 10.1001/ jamaoncol.2015.0494.
- Chlebowski RT, Anderson GL, Aragaki A, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
- Cordina-Duverger E, Truong T, Anger A, et al. Risk of breast cancer by type of menopausal hormone therapy: a case-control study among postmenopausal women in France. PLoS One. 2013;8:e78016. doi: 10.1371/journal.pone.0078016.
- Simin J, Tamimi R, Lagergren J, et al. Menopausal hormone therapy and cancer risk: an overestimated risk? Eur J Cancer. 2017;84:60–8. doi: 10.1016/j.ejca. 2017.07.012.
- Stanczyk FZ, Hapgood JP, Winer S, et al. Progestogens used in postmenopausal hormone therapy: differences in their pharmacological properties, intracellular actions, and clinical effects. Endocr Rev. 2013;34:171-208. doi: 10.1210/er.20121008.
- Stute P, Wildt L, Neulen J. The impact of micronized progesterone on breast cancer risk: a systematic review. Climacteric. 2018;21:111-122. doi: 10.1080/13697137.2017.1421925.
- Anderson GL, Chlebowski RT, Aragaki A, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the Women’s Health Initiative randomised placebo-controlled trial. Lancet Oncol. 2012;13:476-486. doi: 10.1016/S1470-2045(12)70075-X.
- Chlebowski RT, Barrington W, Aragaki AK, et al. Estrogen alone and health outcomes in black women by African ancestry: a secondary analyses of a randomized controlled trial. Menopause. 2017;24:133-141. doi: 10.1097/ GME.0000000000000733.
- The NAMS 2017 Hormone Therapy Position Statement Advisory Panel. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause. 2017;24:728-753. doi: 10.1097/GME.0000000000000921.
- Pinkerton JV. Hormone therapy for postmenopausal women. N Engl J Med. 2020;382(5):446-455. doi: 10.1056/ NEJMcp1714787.
- Marchetti C, De Felice F, Boccia S, et al. Hormone replacement therapy after prophylactic risk-reducing salpingooophorectomy and breast cancer risk in BRCA1 and BRCA2 mutation carriers: a meta-analysis. Crit Rev Oncol Hematol. 2018;132:111-115. doi: 10.1016/j.critrevonc.2018.09.018.
- Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of venous thromboembolism: nested case-control studies using the QResearch and CPRD databases. BMJ. 2019;364:k4810. doi: 10.1136/bmj.k4810.
- Pinkerton JV. Hormone therapy: key points from NAMS 2017 Position Statement. Clin Obstet Gynecol. 2018;61:447453. doi: 10.1097/GRF.0000000000000383.
- Parker WH, Broder MS, Liu Z, et al. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226. doi: 10.1097/01. AOG.0000167394.38215.56.
- Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:10271037. doi: 10.1097/AOG.0b013e3181a11c64.
- Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:10741083. doi: 10.1212/01.wnl.0000276984.19542.e6.
- Rocca WA, Gazzuola Rocca L, Smith CY, et al Loss of ovarian hormones and accelerated somatic and mental aging. Physiology (Bethesda). 2018;33:374-383. doi: 10.1152/ physiol.00024.2018.
- Mytton J, Evison F, Chilton PJ, et al. Removal of all ovarian tissue versus conserving ovarian tissue at time of hysterectomy in premenopausal patients with benign disease: study using routine data and data linkage. BMJ. 2017;356:j372. doi: 10.1136/bmj.j372.
- Erickson Z, Rocca WA, Smith CY, et al. Time trends in unilateral and bilateral oophorectomy in a geographically defined American population. Obstet Gynecol. 2022;139:724-734. doi: 10.1097/AOG.0000000000004728.
- Choi YH, Terry MB, Daly MB, et al. Association of risk-reducing salpingo-oophorectomy with breast cancer risk in women with BRCA1 and BRCA2 pathogenic variants. JAMA Oncol. 2021;7:585-592. doi: 10.1001/jamaoncol.2020 .7995.
- Suzuki Y, Huang Y, Melamed A, et al. Use of estrogen therapy after surgical menopause in women who are premenopausal. Obstet Gynecol. 2022;139:756-763. doi: 10.1097/ AOG.0000000000004762.
- Faubion S, Kaunitz AM, Kapoor E. HT for women who have had BSO before the age of natural menopause: discerning the nuances. OBG Manag. 2022;34(2):20-27, 45. doi: 10.12788/ obgm.0174.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430. doi: 10.1001/ jama.2021.3305.
This year’s Menopause Update focuses on 2 menopause-related issues relevant to ObGyns and our menopausal patients:
- choosing the safest regimens, particularly with respect to risk of breast cancer, when prescribing hormone therapy (HT) to menopausal women
- reviewing the risks and benefits of premenopausal bilateral salpingo-oophorectomy and the pros and cons of replacement HT in surgically menopausal patients.
We hope that you find this updated information useful as you care for menopausal women.
Revisiting menopausal HT and the risk of breast cancer: What we know now
Abenhaim HA, Suissa S, Azoulay L, et al. Menopausal hormone therapy formulation and breast cancer risk. Obstet Gynecol. 2022;139:1103-1110. doi: 10.1097/AOG.0000000000004723.
Reevaluation of the Women’s Health Initiative randomized controlled trials (WHI RCTs), long-term (median follow-up more than 20 years) cumulative follow-up data, and results from additional studies have suggested that estrogen therapy (ET) alone in menopausal women with prior hysterectomy does not increase the risk of breast cancer. By contrast, estrogen with progestin (synthetic progestogens that include medroxyprogesterone acetate [MPA] and norethindrone acetate) slightly increases the risk of breast cancer. In the past 10 years, several publications have shed light on whether the type of progestogen affects the risk of breast cancer and can help provide evidence-based information to guide clinicians.
Breast cancer risk with combined HT and synthetic progestin
In the first part of the WHI RCT, women were randomly assigned to receive either conjugated equine estrogen (CEE) plus synthetic progestin (MPA) or a placebo. Combined estrogen-progestin therapy (EPT) was associated with a modestly elevated risk of breast cancer.1 In the second part of the WHI trial, CEE only (estrogen alone, ET) was compared with placebo among women with prior hysterectomy, with no effect found on breast cancer incidence.2
Most older observational studies published in 2003 to 2005 found that neither CEE nor estradiol appeared to increase the risk of breast cancer when used alone.3-5 However, estrogen use in combination with synthetic progestins (MPA, norethindrone, levonorgestrel, and norgestrel) has been associated with an increased risk of breast cancer,4,6 while the elevated risk of breast cancer with micronized progesterone has been less substantial.7,8
Continue to: Newer data suggest the type of progestogen used affects risk...
Newer data suggest the type of progestogen used affects risk
In a report published in the June 2022 issue of Obstetrics and Gynecology, Abenhaim and colleagues used a nested population-based case-control study of administrative data available in the UK Clinical Practice Research Datalink and provider prescriptions to evaluate the additive effect on the risk of breast cancer of the type of progestogen (micronized progesterone or synthetic progestins) when combined with estradiol for the treatment of menopausal symptoms.9 A cohort of 561,379 women was included in the case-control study (10:1 ratio), 43,183 in the case group (patients diagnosed with invasive breast cancer), and 431,830 in the matched control group.
Overall, in the stratified analysis, a small but significant increase in the risk of breast cancer was found in ever users of menopausal HT (odds ratio [OR], 1.12; 95% confidence interval [CI], 1.09–1.15). Neither estradiol (OR, 1.04; 95% CI, 1.00–1.09) nor CEE (OR, 1.01; 95% CI, 0.96–1.06) was associated with an elevated risk of being diagnosed with invasive breast cancer. Of note, no elevated risk of breast cancer was associated with combination estrogen-progesterone therapy. However, the risk of breast cancer for women who had used synthetic progestins, mostly MPA, was significantly elevated (OR, 1.28; 95% CI, 1.22-1.35). Notably, this modestly elevated odds ratio with the use of estrogen-progestin HT is almost identical to that observed with CEE/ MPA in the WHI.1 Similar findings were found in women aged 50 to 60 years.
The adjusted analyses from the large WHI RCTs provide additional support: the synthetic progestin MPA combined with CEE showed a higher risk of breast cancer than CEE alone in women with prior hysterectomy.10
In the long-term follow-up of the WHI RCTs, after a median of 20.3 years postrandomization, prior randomization to CEE alone for postmenopausal women with prior hysterectomy was associated with a significantly lowered risk of breast cancer incidence and mortality.11 By contrast, prior randomization to CEE plus MPA (EPT) for women with an intact uterus was associated with a small but significantly increased incidence of breast cancer but no significant difference in breast cancer mortality.
In the French E3N EPIC population-based prospective cohort study, Fournier and colleagues4,5 found that women who received estrogen combined with synthetic progestins (mostly MPA) had a higher risk of breast cancer, with an age-adjusted relative risk of 1.4 (95% CI, 1.2–1.7), a finding not seen in women who received estrogen combined with micronized progesterone, similar to findings by Cordina-Duverger and colleagues and Simin and colleagues.12,13 In the E3N study, only 948 women were identified with breast cancer; 268 of these had used synthetic progestins.4,5
Both the Abenhaim cohort9 and the longterm outcomes of WHI RCT trial data11 found a significant contributing effect of MPA (synthetic progestin) in the risk of breast cancer. Progestogens are not thought to exert a class effect. Although it is clear that progestogens (progesterone or progestins) prevent estrogeninduced endometrial neoplasia when dosed adequately, different types of progestogens have a differential risk of breast epithelium proliferation and carcinogenic potential.14 A systematic review by Stute and colleagues found that micronized progesterone did not appear to alter mammographic breast density assessments or breast biopsy results.15
Progesterone capsules, available in generic form in 100-mg and 200-mg doses, are formulated with peanut oil, and they should be taken at bedtime as progesterone can induce drowsiness.
When combined with standard-dose estrogen, including oral estradiol 1.0 mg, transdermal estradiol 0.05 mg, or oral conjugated equine estrogen 0.625 mg, the appropriate dose of progesterone is 100 mg if used continuously or 200 mg if used as cyclic therapy. With higher doses of estrogen, progesterone 200 mg should be taken continuously.
An oral formulation that combines estradiol 1 mg and progesterone 100 mg does not contain peanut oil and, accordingly, can be used safely by those with peanut allergies. This combination product is marketed under the name Bijuva (TherapeuticsMD, Boca Raton, Florida).1
Reference
1. Lobo RA, Archer DF, Kagan R, et al. A 17β-estradiol-progesterone oral capsule for vasomotor symptoms in postmenopausal women: a randomized controlled trial. Obstet Gynecol. 2018;132:161-170. doi: 10.1097/AOG.0000000000002645. Erratum in: Obstet Gynecol. 2018;132:786.
Race considerations
The study by Abenhaim and colleagues was unable to address the issues of race or ethnicity.9 However, in the racially diverse WHI trial of women with prior hysterectomy, estrogen-alone use significantly reduced breast cancer incidence in all participants.10,16 Post hoc analysis of the 1,616 Black women with prior hysterectomy in the WHI RCT showed a significantly decreased breast cancer incidence with use of estrogen alone (hazard ratio [HR], 0.47; 95% CI, 0.26–0.82).1 When race was evaluated in the long-term cumulative follow-up of the WHI trial, estrogen-alone use significantly reduced breast cancer incidence in Black women, with no adverse effect on coronary heart disease, global index, or all-cause mortality, and with fewer cases of venous thromboembolism.17 The global index findings were favorable for Black women in their 50s and those with vasomotor symptoms.
Continue to: Impact of HT in women with an elevated risk of breast cancer...
Impact of HT in women with an elevated risk of breast cancer
Abenhaim and colleagues could not evaluate the effect of HT in women with a baseline elevated risk of breast cancer.9 For these women, HT may be recommended after premature surgical menopause due to increased risks for coronary heart disease, osteoporosis, genitourinary syndrome of menopause, and cognitive changes when estrogen is not taken postsurgery through to at least the average age of menopause, considered age 51.18,19
Marchetti and colleagues reviewed 3 clinical trials that assessed breast cancer events in 1,100 BRCA gene mutation carriers with intact breasts who underwent risk-reducing salpingo-oophorectomy (RRSO) who used or did not use HT.20 For BRCA1 and BRCA2 mutation carriers who received HT after RRSO, no elevated risk of breast cancer risk was seen (HR, 0.98; 95% CI, 0.63–1.52). There was a nonsignificant reduction in breast cancer risk for the estrogen-alone users compared with EPT HT (OR, 0.53; 95% CI, 0.25–1.15). Thus, short-term use of HT, estrogen alone or EPT, does not appear to elevate the risk of breast cancer after RRSO in these high-risk women.
Individualizing HT for menopausal symptoms
The data presented provide reassuring evidence that longer-term use of ET does not appear to increase breast cancer risk, regardless of the type of estrogen (CEE or estradiol).4,5,9,11 For women with a uterus, micronized progesterone has less (if any) effect on breast cancer risk. By contrast, the use of synthetic progestins (such as MPA), when combined with estrogen, has been associated with a small but real increased breast cancer risk.
The most evident benefit of HT is in treating vasomotor symptoms and preventing bone loss for those at elevated risk in healthy women without contraindications who initiate systemic HT when younger than age 60 or within 10 years of menopause onset. Benefit and risk ratio depends on age and time from menopause onset when HT is initiated. Hormone therapy safety varies depending on type, dose, duration, route of administration, timing of initiation, and whether, and type, of progestogen is used. Transdermal estradiol, particularly when dosed at 0.05 mg or less, has been shown to have less thrombotic and stroke risk than oral estrogen.21
Individualizing treatment includes using the best available evidence to maximize benefits and minimize risks, with periodic reevaluation of benefits and risks of continuing or discontinuing HT or changing to lower doses. ObGyns who follow best practices in prescribing systemic HT can now help menopausal patients with bothersome symptoms take advantage of systemic HT’s benefits while providing reassurance regarding menopausal HT’s safety.18 Transdermal therapy is a safer option for women at elevated baseline risk of venous thrombosis (for example, obese women) and older patients. Likewise, given its safety with respect to risk of breast cancer, the use of micronized progesterone over synthetic progestins should be considered when prescribing EPT to women with an intact uterus.
We can replace fear of HT with evidence-based discussions.22 For women with prior hysterectomy who have menopausal symptoms that impact their quality of life, ET at menopause does not appear to increase the risk of breast cancer. For women with an intact uterus who are considering use of estrogen and progestogen, extended-duration use of combination HT with synthetic progestins slightly elevates the risk of breast cancer, while the use of micronized progesterone does not appear to elevate breast cancer risk. Likewise, transdermal estrogen does not appear to elevate thrombosis risk.
Continue to: Benefits of avoiding BSO in women at average risk of ovarian cancer...
Benefits of avoiding BSO in women at average risk of ovarian cancer
Erickson Z, Rocca WA, Smith CY, et al. Time trends in unilateral and bilateral oophorectomy in a geographically defined American population. Obstet Gynecol. 2022;139:724-734. doi: 10.1097/ AOG.0000000000004728.
In 2005, gynecologist William Parker, MD, and colleagues used modeling methodology to assess the long-term risks and benefits of performing bilateral salpingo-oophorectomy (BSO) at the time of hysterectomy for benign disease in women at average risk for ovarian cancer.23 They concluded that practicing ovarian conservation until age 65 increased women’s long-term survival. Among their findings were that women with BSO before age 55 had an 8.6% excess overall mortality by age 80, while those with oophorectomy before age 59 had 3.9% excess mortality. They noted a sustained, but decreasing, mortality benefit until the age of 75 and stated that at no age did their model suggest higher mortality in women who chose ovarian conservation. Parker and colleagues concluded that ovarian conservation until at least age 65 benefited long-term survival for women at average risk for ovarian cancer when undergoing hysterectomy for benign disease.23
Certain risks decreased, others increased
A second report in 2009 by Parker and colleagues from the large prospective Nurses’ Health Study found that, while BSO at the time of hysterectomy for benign disease was associated with a decreased risk of breast and ovarian cancer, BSO was associated with an increased risk of all-cause mortality, fatal and nonfatal coronary heart disease, and lung cancer.24 Similar to the findings of the 2005 report, the authors noted that in no analysis or age group was BSO associated with increased survival. They also noted that compared with those who underwent BSO before age 50 and used ET, women with no history of ET use had an approximately 2-fold elevated risk of new onset coronary heart disease (HR, 1.98; 95% CI, 1.18–3.32).24
In 2007, Walter Rocca, MD, a Mayo Clinic neurologist with a particular interest in the epidemiology of dementia, and colleagues at the Mayo Clinic published results of a study that assessed a cohort of women who had undergone unilateral oophorectomy or BSO prior to the onset of menopause.25 The risk of cognitive impairment or dementia was higher in these women compared with women who had intact ovaries (HR, 1.46; 95% CI, 1.13-1.90). Of note, this elevated risk was confined to those who underwent oophorectomy before 49 years of age and were not prescribed estrogen until age 50 or older.25
In a subsequent publication, Rocca and colleagues pointed out that BSO prior to menopause not only is associated with higher rates of all-cause mortality and cognitive impairment but also with coronary heart disease, parkinsonism, osteoporosis, and other chronic conditions associated with aging, including metabolic, mental health, and arthritic disorders.26
Oophorectomy trends tracked
Given these and other reports27 that highlighted the health risks of premenopausal BSO in women at average risk for ovarian cancer, Rocca and colleagues recently assessed trends in the occurrence of unilateral oophorectomy or BSO versus ovarian conservation among all women residing in the Minnesota county (Olmsted) in which Mayo Clinic is located, and who underwent gynecologic surgery between 1950 and 2018.28
The investigators limited their analysis to women who had undergone unilateral oophorectomy or BSO between ages 18 and 49 years (these women are assumed to have been premenopausal). The authors considered as indications for oophorectomy primary or metastatic ovarian cancer, risk-reducing BSO for women at elevated risk for ovarian cancer (for example, strong family history or known BRCA gene mutation), adnexal mass, endometriosis, torsion, and other benign gynecologic conditions that included pelvic pain, abscess, oophoritis, or ectopic pregnancy. When more than 1 indication for ovarian surgery was present, the authors used the most clinically important indication. Unilateral oophorectomy or BSO was considered not indicated if the surgery was performed during another primary procedure (usually hysterectomy) without indication, or if the surgeon referred to the ovarian surgery as elective.
Results. Among 5,154 women who had oophorectomies between 1950 and 2018, the proportion of these women who underwent unilateral oophorectomy and BSO was 40.6% and 59.4%, respectively.
For most years between 1950 and 1979, the incidence of unilateral oophorectomy was higher than BSO. However, from 1980 to 2004, the incidence of BSO increased more than 2-fold while the incidence of unilateral surgery declined. After 2005, however, both types of ovarian surgery declined. During the years 2005–2018, a marked decline in BSO occurred, with the reduced incidence in premenopausal BSO most notable among women undergoing hysterectomy or those without an indication for oophorectomy.
Historically, ObGyns were taught that the benefits of removing normal ovaries (to prevent ovarian cancer) in average-risk women at the time of hysterectomy outweighed the risks. We agree with the authors’ speculation that beginning with Parker’s 2005 publication,23 ObGyns have become more conservative in performing unindicated BSO in women at average risk for ovarian cancer, now recognizing that the harms of this procedure often outweigh any benefits.28
Women with BRCA1 and BRCA2 gene mutations are at elevated risk for ovarian, tubal, and breast malignancies. In this population, risk-reducing BSO dramatically lowers future risk of ovarian and tubal cancer.
Data addressing the effect of RRSO in BRCA1 and BRCA2 gene mutation carriers continue to be evaluated, with differences between the 2 mutations, but they suggest that the surgery reduces not only ovarian cancer and tubal cancer but also possibly breast cancer.29
Many of our patients are fearful regarding the possibility that they could be diagnosed with breast or ovarian cancer, and in their minds, fears regarding these 2 potentially deadly diseases outweigh concerns about more common causes of death in women, including cardiovascular disease. Accordingly, counseling women at average risk for ovarian cancer who are planning hysterectomy for benign indications can be challenging. In recent years, ObGyns have increasingly been performing opportunistic bilateral salpingectomy (OS) in women at average risk of ovarian cancer at the time of hysterectomy for benign disease. It is important to note that the studies we refer to in this Update addressed BSO, not OS. We hope that the findings we have reviewed here assist clinicians in helping women to understand the risks and benefits associated with premenopausal BSO and the need to discuss the pros and cons of HT for these women before surgery.
Continue to: Trends show decline in ET use in surgically menopausal women...
Trends show decline in ET use in surgically menopausal women
Suzuki Y, Huang Y, Melamed A, et al. Use of estrogen therapy after surgical menopause in women who are premenopausal. Obstet Gynecol. 2022;139:756-763. doi: 10.1097/AOG.0000000000004762.
In addition to highlighting the risks associated with premenopausal BSO in women at average risk for ovarian cancer, the reports referred to above also underscore that the use of replacement menopausal HT in premenopausal women who undergo BSO prevents morbidity and mortality that otherwise accompanies surgical menopause. In addition, the North American Menopause Society (NAMS) recommends replacement menopausal HT in the setting of induced early menopause when no contraindications are present.18
To assess the prevalence of HT use in surgically menopausal women, investigators at Columbia University College of Physicians and Surgeons used a national database that captures health insurance claims for some 280 million US patients, focusing on women aged 18 to 50 years who underwent BSO from 2008 to 2019.30 The great majority of women in this database have private insurance. Although the authors used the term estrogen therapy in their article, this term refers to systemic estrogen alone or with progestogen, as well as vaginal ET (personal communication with Jason Wright, MD, a coauthor of the study, May 19, 2022). In this Update section, we use the term HT to include use of any systemic HT or vaginal estrogen.
Prevalence of HT use changed over time period and patient age range
Among almost 61,980 evaluable women who had undergone BSO (median age, 45 years; 75.1% with concomitant hysterectomy; median follow-up time, 27 months), with no history of gynecologic or breast cancer, HT was used within 3 years of BSO by 64.5%. The highest percentage of women in this cohort who used HT peaked in 2008 (69.5%), declining to 58.2% by 2016. The median duration of HT use was 5.3 months. The prevalence of HT use 3 years after BSO declined with age, from 79.1% in women aged 18–29 to 60.0% in women aged 45–50.30
This report, published in the June 2022 issue of Obstetrics and Gynecology, makes several sobering observations: Many surgically menopausal women aged 50 years and younger are not prescribed HT, the proportion of such women receiving a prescription for HT is declining over time, and the duration of HT use following BSO is short. ●
As ObGyn physicians, we can play an important role by educating healthy women with induced menopause who are younger than the average age of spontaneous menopause, and who have no contraindications, that the benefits of HT far outweigh risks. Many of these women will benefit from longer-term HT, using doses substantially higher than are used in women who undergo spontaneous menopause.31,32 After reaching the age of menopause, healthy women without contraindications may continue to benefit from HT into their 50s or beyond if they have vasomotor symptoms, bone loss, or other indications for treatment.18,19
This year’s Menopause Update focuses on 2 menopause-related issues relevant to ObGyns and our menopausal patients:
- choosing the safest regimens, particularly with respect to risk of breast cancer, when prescribing hormone therapy (HT) to menopausal women
- reviewing the risks and benefits of premenopausal bilateral salpingo-oophorectomy and the pros and cons of replacement HT in surgically menopausal patients.
We hope that you find this updated information useful as you care for menopausal women.
Revisiting menopausal HT and the risk of breast cancer: What we know now
Abenhaim HA, Suissa S, Azoulay L, et al. Menopausal hormone therapy formulation and breast cancer risk. Obstet Gynecol. 2022;139:1103-1110. doi: 10.1097/AOG.0000000000004723.
Reevaluation of the Women’s Health Initiative randomized controlled trials (WHI RCTs), long-term (median follow-up more than 20 years) cumulative follow-up data, and results from additional studies have suggested that estrogen therapy (ET) alone in menopausal women with prior hysterectomy does not increase the risk of breast cancer. By contrast, estrogen with progestin (synthetic progestogens that include medroxyprogesterone acetate [MPA] and norethindrone acetate) slightly increases the risk of breast cancer. In the past 10 years, several publications have shed light on whether the type of progestogen affects the risk of breast cancer and can help provide evidence-based information to guide clinicians.
Breast cancer risk with combined HT and synthetic progestin
In the first part of the WHI RCT, women were randomly assigned to receive either conjugated equine estrogen (CEE) plus synthetic progestin (MPA) or a placebo. Combined estrogen-progestin therapy (EPT) was associated with a modestly elevated risk of breast cancer.1 In the second part of the WHI trial, CEE only (estrogen alone, ET) was compared with placebo among women with prior hysterectomy, with no effect found on breast cancer incidence.2
Most older observational studies published in 2003 to 2005 found that neither CEE nor estradiol appeared to increase the risk of breast cancer when used alone.3-5 However, estrogen use in combination with synthetic progestins (MPA, norethindrone, levonorgestrel, and norgestrel) has been associated with an increased risk of breast cancer,4,6 while the elevated risk of breast cancer with micronized progesterone has been less substantial.7,8
Continue to: Newer data suggest the type of progestogen used affects risk...
Newer data suggest the type of progestogen used affects risk
In a report published in the June 2022 issue of Obstetrics and Gynecology, Abenhaim and colleagues used a nested population-based case-control study of administrative data available in the UK Clinical Practice Research Datalink and provider prescriptions to evaluate the additive effect on the risk of breast cancer of the type of progestogen (micronized progesterone or synthetic progestins) when combined with estradiol for the treatment of menopausal symptoms.9 A cohort of 561,379 women was included in the case-control study (10:1 ratio), 43,183 in the case group (patients diagnosed with invasive breast cancer), and 431,830 in the matched control group.
Overall, in the stratified analysis, a small but significant increase in the risk of breast cancer was found in ever users of menopausal HT (odds ratio [OR], 1.12; 95% confidence interval [CI], 1.09–1.15). Neither estradiol (OR, 1.04; 95% CI, 1.00–1.09) nor CEE (OR, 1.01; 95% CI, 0.96–1.06) was associated with an elevated risk of being diagnosed with invasive breast cancer. Of note, no elevated risk of breast cancer was associated with combination estrogen-progesterone therapy. However, the risk of breast cancer for women who had used synthetic progestins, mostly MPA, was significantly elevated (OR, 1.28; 95% CI, 1.22-1.35). Notably, this modestly elevated odds ratio with the use of estrogen-progestin HT is almost identical to that observed with CEE/ MPA in the WHI.1 Similar findings were found in women aged 50 to 60 years.
The adjusted analyses from the large WHI RCTs provide additional support: the synthetic progestin MPA combined with CEE showed a higher risk of breast cancer than CEE alone in women with prior hysterectomy.10
In the long-term follow-up of the WHI RCTs, after a median of 20.3 years postrandomization, prior randomization to CEE alone for postmenopausal women with prior hysterectomy was associated with a significantly lowered risk of breast cancer incidence and mortality.11 By contrast, prior randomization to CEE plus MPA (EPT) for women with an intact uterus was associated with a small but significantly increased incidence of breast cancer but no significant difference in breast cancer mortality.
In the French E3N EPIC population-based prospective cohort study, Fournier and colleagues4,5 found that women who received estrogen combined with synthetic progestins (mostly MPA) had a higher risk of breast cancer, with an age-adjusted relative risk of 1.4 (95% CI, 1.2–1.7), a finding not seen in women who received estrogen combined with micronized progesterone, similar to findings by Cordina-Duverger and colleagues and Simin and colleagues.12,13 In the E3N study, only 948 women were identified with breast cancer; 268 of these had used synthetic progestins.4,5
Both the Abenhaim cohort9 and the longterm outcomes of WHI RCT trial data11 found a significant contributing effect of MPA (synthetic progestin) in the risk of breast cancer. Progestogens are not thought to exert a class effect. Although it is clear that progestogens (progesterone or progestins) prevent estrogeninduced endometrial neoplasia when dosed adequately, different types of progestogens have a differential risk of breast epithelium proliferation and carcinogenic potential.14 A systematic review by Stute and colleagues found that micronized progesterone did not appear to alter mammographic breast density assessments or breast biopsy results.15
Progesterone capsules, available in generic form in 100-mg and 200-mg doses, are formulated with peanut oil, and they should be taken at bedtime as progesterone can induce drowsiness.
When combined with standard-dose estrogen, including oral estradiol 1.0 mg, transdermal estradiol 0.05 mg, or oral conjugated equine estrogen 0.625 mg, the appropriate dose of progesterone is 100 mg if used continuously or 200 mg if used as cyclic therapy. With higher doses of estrogen, progesterone 200 mg should be taken continuously.
An oral formulation that combines estradiol 1 mg and progesterone 100 mg does not contain peanut oil and, accordingly, can be used safely by those with peanut allergies. This combination product is marketed under the name Bijuva (TherapeuticsMD, Boca Raton, Florida).1
Reference
1. Lobo RA, Archer DF, Kagan R, et al. A 17β-estradiol-progesterone oral capsule for vasomotor symptoms in postmenopausal women: a randomized controlled trial. Obstet Gynecol. 2018;132:161-170. doi: 10.1097/AOG.0000000000002645. Erratum in: Obstet Gynecol. 2018;132:786.
Race considerations
The study by Abenhaim and colleagues was unable to address the issues of race or ethnicity.9 However, in the racially diverse WHI trial of women with prior hysterectomy, estrogen-alone use significantly reduced breast cancer incidence in all participants.10,16 Post hoc analysis of the 1,616 Black women with prior hysterectomy in the WHI RCT showed a significantly decreased breast cancer incidence with use of estrogen alone (hazard ratio [HR], 0.47; 95% CI, 0.26–0.82).1 When race was evaluated in the long-term cumulative follow-up of the WHI trial, estrogen-alone use significantly reduced breast cancer incidence in Black women, with no adverse effect on coronary heart disease, global index, or all-cause mortality, and with fewer cases of venous thromboembolism.17 The global index findings were favorable for Black women in their 50s and those with vasomotor symptoms.
Continue to: Impact of HT in women with an elevated risk of breast cancer...
Impact of HT in women with an elevated risk of breast cancer
Abenhaim and colleagues could not evaluate the effect of HT in women with a baseline elevated risk of breast cancer.9 For these women, HT may be recommended after premature surgical menopause due to increased risks for coronary heart disease, osteoporosis, genitourinary syndrome of menopause, and cognitive changes when estrogen is not taken postsurgery through to at least the average age of menopause, considered age 51.18,19
Marchetti and colleagues reviewed 3 clinical trials that assessed breast cancer events in 1,100 BRCA gene mutation carriers with intact breasts who underwent risk-reducing salpingo-oophorectomy (RRSO) who used or did not use HT.20 For BRCA1 and BRCA2 mutation carriers who received HT after RRSO, no elevated risk of breast cancer risk was seen (HR, 0.98; 95% CI, 0.63–1.52). There was a nonsignificant reduction in breast cancer risk for the estrogen-alone users compared with EPT HT (OR, 0.53; 95% CI, 0.25–1.15). Thus, short-term use of HT, estrogen alone or EPT, does not appear to elevate the risk of breast cancer after RRSO in these high-risk women.
Individualizing HT for menopausal symptoms
The data presented provide reassuring evidence that longer-term use of ET does not appear to increase breast cancer risk, regardless of the type of estrogen (CEE or estradiol).4,5,9,11 For women with a uterus, micronized progesterone has less (if any) effect on breast cancer risk. By contrast, the use of synthetic progestins (such as MPA), when combined with estrogen, has been associated with a small but real increased breast cancer risk.
The most evident benefit of HT is in treating vasomotor symptoms and preventing bone loss for those at elevated risk in healthy women without contraindications who initiate systemic HT when younger than age 60 or within 10 years of menopause onset. Benefit and risk ratio depends on age and time from menopause onset when HT is initiated. Hormone therapy safety varies depending on type, dose, duration, route of administration, timing of initiation, and whether, and type, of progestogen is used. Transdermal estradiol, particularly when dosed at 0.05 mg or less, has been shown to have less thrombotic and stroke risk than oral estrogen.21
Individualizing treatment includes using the best available evidence to maximize benefits and minimize risks, with periodic reevaluation of benefits and risks of continuing or discontinuing HT or changing to lower doses. ObGyns who follow best practices in prescribing systemic HT can now help menopausal patients with bothersome symptoms take advantage of systemic HT’s benefits while providing reassurance regarding menopausal HT’s safety.18 Transdermal therapy is a safer option for women at elevated baseline risk of venous thrombosis (for example, obese women) and older patients. Likewise, given its safety with respect to risk of breast cancer, the use of micronized progesterone over synthetic progestins should be considered when prescribing EPT to women with an intact uterus.
We can replace fear of HT with evidence-based discussions.22 For women with prior hysterectomy who have menopausal symptoms that impact their quality of life, ET at menopause does not appear to increase the risk of breast cancer. For women with an intact uterus who are considering use of estrogen and progestogen, extended-duration use of combination HT with synthetic progestins slightly elevates the risk of breast cancer, while the use of micronized progesterone does not appear to elevate breast cancer risk. Likewise, transdermal estrogen does not appear to elevate thrombosis risk.
Continue to: Benefits of avoiding BSO in women at average risk of ovarian cancer...
Benefits of avoiding BSO in women at average risk of ovarian cancer
Erickson Z, Rocca WA, Smith CY, et al. Time trends in unilateral and bilateral oophorectomy in a geographically defined American population. Obstet Gynecol. 2022;139:724-734. doi: 10.1097/ AOG.0000000000004728.
In 2005, gynecologist William Parker, MD, and colleagues used modeling methodology to assess the long-term risks and benefits of performing bilateral salpingo-oophorectomy (BSO) at the time of hysterectomy for benign disease in women at average risk for ovarian cancer.23 They concluded that practicing ovarian conservation until age 65 increased women’s long-term survival. Among their findings were that women with BSO before age 55 had an 8.6% excess overall mortality by age 80, while those with oophorectomy before age 59 had 3.9% excess mortality. They noted a sustained, but decreasing, mortality benefit until the age of 75 and stated that at no age did their model suggest higher mortality in women who chose ovarian conservation. Parker and colleagues concluded that ovarian conservation until at least age 65 benefited long-term survival for women at average risk for ovarian cancer when undergoing hysterectomy for benign disease.23
Certain risks decreased, others increased
A second report in 2009 by Parker and colleagues from the large prospective Nurses’ Health Study found that, while BSO at the time of hysterectomy for benign disease was associated with a decreased risk of breast and ovarian cancer, BSO was associated with an increased risk of all-cause mortality, fatal and nonfatal coronary heart disease, and lung cancer.24 Similar to the findings of the 2005 report, the authors noted that in no analysis or age group was BSO associated with increased survival. They also noted that compared with those who underwent BSO before age 50 and used ET, women with no history of ET use had an approximately 2-fold elevated risk of new onset coronary heart disease (HR, 1.98; 95% CI, 1.18–3.32).24
In 2007, Walter Rocca, MD, a Mayo Clinic neurologist with a particular interest in the epidemiology of dementia, and colleagues at the Mayo Clinic published results of a study that assessed a cohort of women who had undergone unilateral oophorectomy or BSO prior to the onset of menopause.25 The risk of cognitive impairment or dementia was higher in these women compared with women who had intact ovaries (HR, 1.46; 95% CI, 1.13-1.90). Of note, this elevated risk was confined to those who underwent oophorectomy before 49 years of age and were not prescribed estrogen until age 50 or older.25
In a subsequent publication, Rocca and colleagues pointed out that BSO prior to menopause not only is associated with higher rates of all-cause mortality and cognitive impairment but also with coronary heart disease, parkinsonism, osteoporosis, and other chronic conditions associated with aging, including metabolic, mental health, and arthritic disorders.26
Oophorectomy trends tracked
Given these and other reports27 that highlighted the health risks of premenopausal BSO in women at average risk for ovarian cancer, Rocca and colleagues recently assessed trends in the occurrence of unilateral oophorectomy or BSO versus ovarian conservation among all women residing in the Minnesota county (Olmsted) in which Mayo Clinic is located, and who underwent gynecologic surgery between 1950 and 2018.28
The investigators limited their analysis to women who had undergone unilateral oophorectomy or BSO between ages 18 and 49 years (these women are assumed to have been premenopausal). The authors considered as indications for oophorectomy primary or metastatic ovarian cancer, risk-reducing BSO for women at elevated risk for ovarian cancer (for example, strong family history or known BRCA gene mutation), adnexal mass, endometriosis, torsion, and other benign gynecologic conditions that included pelvic pain, abscess, oophoritis, or ectopic pregnancy. When more than 1 indication for ovarian surgery was present, the authors used the most clinically important indication. Unilateral oophorectomy or BSO was considered not indicated if the surgery was performed during another primary procedure (usually hysterectomy) without indication, or if the surgeon referred to the ovarian surgery as elective.
Results. Among 5,154 women who had oophorectomies between 1950 and 2018, the proportion of these women who underwent unilateral oophorectomy and BSO was 40.6% and 59.4%, respectively.
For most years between 1950 and 1979, the incidence of unilateral oophorectomy was higher than BSO. However, from 1980 to 2004, the incidence of BSO increased more than 2-fold while the incidence of unilateral surgery declined. After 2005, however, both types of ovarian surgery declined. During the years 2005–2018, a marked decline in BSO occurred, with the reduced incidence in premenopausal BSO most notable among women undergoing hysterectomy or those without an indication for oophorectomy.
Historically, ObGyns were taught that the benefits of removing normal ovaries (to prevent ovarian cancer) in average-risk women at the time of hysterectomy outweighed the risks. We agree with the authors’ speculation that beginning with Parker’s 2005 publication,23 ObGyns have become more conservative in performing unindicated BSO in women at average risk for ovarian cancer, now recognizing that the harms of this procedure often outweigh any benefits.28
Women with BRCA1 and BRCA2 gene mutations are at elevated risk for ovarian, tubal, and breast malignancies. In this population, risk-reducing BSO dramatically lowers future risk of ovarian and tubal cancer.
Data addressing the effect of RRSO in BRCA1 and BRCA2 gene mutation carriers continue to be evaluated, with differences between the 2 mutations, but they suggest that the surgery reduces not only ovarian cancer and tubal cancer but also possibly breast cancer.29
Many of our patients are fearful regarding the possibility that they could be diagnosed with breast or ovarian cancer, and in their minds, fears regarding these 2 potentially deadly diseases outweigh concerns about more common causes of death in women, including cardiovascular disease. Accordingly, counseling women at average risk for ovarian cancer who are planning hysterectomy for benign indications can be challenging. In recent years, ObGyns have increasingly been performing opportunistic bilateral salpingectomy (OS) in women at average risk of ovarian cancer at the time of hysterectomy for benign disease. It is important to note that the studies we refer to in this Update addressed BSO, not OS. We hope that the findings we have reviewed here assist clinicians in helping women to understand the risks and benefits associated with premenopausal BSO and the need to discuss the pros and cons of HT for these women before surgery.
Continue to: Trends show decline in ET use in surgically menopausal women...
Trends show decline in ET use in surgically menopausal women
Suzuki Y, Huang Y, Melamed A, et al. Use of estrogen therapy after surgical menopause in women who are premenopausal. Obstet Gynecol. 2022;139:756-763. doi: 10.1097/AOG.0000000000004762.
In addition to highlighting the risks associated with premenopausal BSO in women at average risk for ovarian cancer, the reports referred to above also underscore that the use of replacement menopausal HT in premenopausal women who undergo BSO prevents morbidity and mortality that otherwise accompanies surgical menopause. In addition, the North American Menopause Society (NAMS) recommends replacement menopausal HT in the setting of induced early menopause when no contraindications are present.18
To assess the prevalence of HT use in surgically menopausal women, investigators at Columbia University College of Physicians and Surgeons used a national database that captures health insurance claims for some 280 million US patients, focusing on women aged 18 to 50 years who underwent BSO from 2008 to 2019.30 The great majority of women in this database have private insurance. Although the authors used the term estrogen therapy in their article, this term refers to systemic estrogen alone or with progestogen, as well as vaginal ET (personal communication with Jason Wright, MD, a coauthor of the study, May 19, 2022). In this Update section, we use the term HT to include use of any systemic HT or vaginal estrogen.
Prevalence of HT use changed over time period and patient age range
Among almost 61,980 evaluable women who had undergone BSO (median age, 45 years; 75.1% with concomitant hysterectomy; median follow-up time, 27 months), with no history of gynecologic or breast cancer, HT was used within 3 years of BSO by 64.5%. The highest percentage of women in this cohort who used HT peaked in 2008 (69.5%), declining to 58.2% by 2016. The median duration of HT use was 5.3 months. The prevalence of HT use 3 years after BSO declined with age, from 79.1% in women aged 18–29 to 60.0% in women aged 45–50.30
This report, published in the June 2022 issue of Obstetrics and Gynecology, makes several sobering observations: Many surgically menopausal women aged 50 years and younger are not prescribed HT, the proportion of such women receiving a prescription for HT is declining over time, and the duration of HT use following BSO is short. ●
As ObGyn physicians, we can play an important role by educating healthy women with induced menopause who are younger than the average age of spontaneous menopause, and who have no contraindications, that the benefits of HT far outweigh risks. Many of these women will benefit from longer-term HT, using doses substantially higher than are used in women who undergo spontaneous menopause.31,32 After reaching the age of menopause, healthy women without contraindications may continue to benefit from HT into their 50s or beyond if they have vasomotor symptoms, bone loss, or other indications for treatment.18,19
- Chlebowski RT, Hendrix SL, Langer RD, et al; WHI Investigators. Influence of estrogen plus progestin on breast cancer and mammography in healthy postmenopausal women: the Women’s Health Initiative randomized trial. JAMA. 2003;289:3243-3253. doi: 10.1001/jama.289.24.3243.
- Anderson GL, Limacher M, Assaf AR, et al; Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized controlled trial. JAMA. 2004;291:1701-1712. doi: 10.1001/jama.291.14.1701.
- Opatrny L, Dell’Aniello S, Assouline S, et al. Hormone replacement therapy use and variations in the risk of breast cancer. BJOG. 2008;115:169-175. doi: 10.1111/j.14710528.2007.01520.x.
- Fournier A, Berrino F, Riboli E, et al. Breast cancer risk in relation to different types of hormone replacement therapy in the E3N-EPIC cohort. Int J Cancer. 2005;114:448-454. doi: 10.1002/ijc.20710.
- Fournier A, Berrino F, Clavel-Chapelon F. Unequal risks for breast cancer associated with different hormone replacement therapies: results from the E3N cohort study. Breast Cancer Res Treat. 2008;107:103-111. doi: 10.1007/s10549-007-9523-x.
- Beral V; Million Women Study Collaborators. Breast cancer and hormone-replacement therapy in the million women study. Lancet. 2003;362:419–27. doi: 10.1016/s01406736(03)14065-2.
- Yang Z, Hu Y, Zhang J, et al. Estradiol therapy and breast cancer risk in perimenopausal and postmenopausal women: a systematic review and meta-analysis. Gynecol Endocrinol. 2017;33:87-92. doi: 10.1080/09513590.2016.1248932.
- Asi N, Mohammed K, Haydour Q, et al. Progesterone vs synthetic progestins and the risk of breast cancer: a systematic review and meta-analysis. Syst Rev. 2016;5:121. doi: 10.1186/ s13643-016-0294-5.
- Abenhaim HA, Suissa S, Azoulay L, et al. Menopausal hormone therapy formulation and breast cancer risk. Obstet Gynecol. 2022;139:1103-1110. doi: 10.1097/AOG.0000000000004723.
- Chlebowski RT, Rohan TE, Manson JE, et al. Breast cancer after use of estrogen plus progestin and estrogen alone: analyses of data from 2 Women’s Health Initiative randomized clinical trials. JAMA Oncol. 2015;1:296-305. doi: 10.1001/ jamaoncol.2015.0494.
- Chlebowski RT, Anderson GL, Aragaki A, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
- Cordina-Duverger E, Truong T, Anger A, et al. Risk of breast cancer by type of menopausal hormone therapy: a case-control study among postmenopausal women in France. PLoS One. 2013;8:e78016. doi: 10.1371/journal.pone.0078016.
- Simin J, Tamimi R, Lagergren J, et al. Menopausal hormone therapy and cancer risk: an overestimated risk? Eur J Cancer. 2017;84:60–8. doi: 10.1016/j.ejca. 2017.07.012.
- Stanczyk FZ, Hapgood JP, Winer S, et al. Progestogens used in postmenopausal hormone therapy: differences in their pharmacological properties, intracellular actions, and clinical effects. Endocr Rev. 2013;34:171-208. doi: 10.1210/er.20121008.
- Stute P, Wildt L, Neulen J. The impact of micronized progesterone on breast cancer risk: a systematic review. Climacteric. 2018;21:111-122. doi: 10.1080/13697137.2017.1421925.
- Anderson GL, Chlebowski RT, Aragaki A, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the Women’s Health Initiative randomised placebo-controlled trial. Lancet Oncol. 2012;13:476-486. doi: 10.1016/S1470-2045(12)70075-X.
- Chlebowski RT, Barrington W, Aragaki AK, et al. Estrogen alone and health outcomes in black women by African ancestry: a secondary analyses of a randomized controlled trial. Menopause. 2017;24:133-141. doi: 10.1097/ GME.0000000000000733.
- The NAMS 2017 Hormone Therapy Position Statement Advisory Panel. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause. 2017;24:728-753. doi: 10.1097/GME.0000000000000921.
- Pinkerton JV. Hormone therapy for postmenopausal women. N Engl J Med. 2020;382(5):446-455. doi: 10.1056/ NEJMcp1714787.
- Marchetti C, De Felice F, Boccia S, et al. Hormone replacement therapy after prophylactic risk-reducing salpingooophorectomy and breast cancer risk in BRCA1 and BRCA2 mutation carriers: a meta-analysis. Crit Rev Oncol Hematol. 2018;132:111-115. doi: 10.1016/j.critrevonc.2018.09.018.
- Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of venous thromboembolism: nested case-control studies using the QResearch and CPRD databases. BMJ. 2019;364:k4810. doi: 10.1136/bmj.k4810.
- Pinkerton JV. Hormone therapy: key points from NAMS 2017 Position Statement. Clin Obstet Gynecol. 2018;61:447453. doi: 10.1097/GRF.0000000000000383.
- Parker WH, Broder MS, Liu Z, et al. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226. doi: 10.1097/01. AOG.0000167394.38215.56.
- Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:10271037. doi: 10.1097/AOG.0b013e3181a11c64.
- Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:10741083. doi: 10.1212/01.wnl.0000276984.19542.e6.
- Rocca WA, Gazzuola Rocca L, Smith CY, et al Loss of ovarian hormones and accelerated somatic and mental aging. Physiology (Bethesda). 2018;33:374-383. doi: 10.1152/ physiol.00024.2018.
- Mytton J, Evison F, Chilton PJ, et al. Removal of all ovarian tissue versus conserving ovarian tissue at time of hysterectomy in premenopausal patients with benign disease: study using routine data and data linkage. BMJ. 2017;356:j372. doi: 10.1136/bmj.j372.
- Erickson Z, Rocca WA, Smith CY, et al. Time trends in unilateral and bilateral oophorectomy in a geographically defined American population. Obstet Gynecol. 2022;139:724-734. doi: 10.1097/AOG.0000000000004728.
- Choi YH, Terry MB, Daly MB, et al. Association of risk-reducing salpingo-oophorectomy with breast cancer risk in women with BRCA1 and BRCA2 pathogenic variants. JAMA Oncol. 2021;7:585-592. doi: 10.1001/jamaoncol.2020 .7995.
- Suzuki Y, Huang Y, Melamed A, et al. Use of estrogen therapy after surgical menopause in women who are premenopausal. Obstet Gynecol. 2022;139:756-763. doi: 10.1097/ AOG.0000000000004762.
- Faubion S, Kaunitz AM, Kapoor E. HT for women who have had BSO before the age of natural menopause: discerning the nuances. OBG Manag. 2022;34(2):20-27, 45. doi: 10.12788/ obgm.0174.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430. doi: 10.1001/ jama.2021.3305.
- Chlebowski RT, Hendrix SL, Langer RD, et al; WHI Investigators. Influence of estrogen plus progestin on breast cancer and mammography in healthy postmenopausal women: the Women’s Health Initiative randomized trial. JAMA. 2003;289:3243-3253. doi: 10.1001/jama.289.24.3243.
- Anderson GL, Limacher M, Assaf AR, et al; Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized controlled trial. JAMA. 2004;291:1701-1712. doi: 10.1001/jama.291.14.1701.
- Opatrny L, Dell’Aniello S, Assouline S, et al. Hormone replacement therapy use and variations in the risk of breast cancer. BJOG. 2008;115:169-175. doi: 10.1111/j.14710528.2007.01520.x.
- Fournier A, Berrino F, Riboli E, et al. Breast cancer risk in relation to different types of hormone replacement therapy in the E3N-EPIC cohort. Int J Cancer. 2005;114:448-454. doi: 10.1002/ijc.20710.
- Fournier A, Berrino F, Clavel-Chapelon F. Unequal risks for breast cancer associated with different hormone replacement therapies: results from the E3N cohort study. Breast Cancer Res Treat. 2008;107:103-111. doi: 10.1007/s10549-007-9523-x.
- Beral V; Million Women Study Collaborators. Breast cancer and hormone-replacement therapy in the million women study. Lancet. 2003;362:419–27. doi: 10.1016/s01406736(03)14065-2.
- Yang Z, Hu Y, Zhang J, et al. Estradiol therapy and breast cancer risk in perimenopausal and postmenopausal women: a systematic review and meta-analysis. Gynecol Endocrinol. 2017;33:87-92. doi: 10.1080/09513590.2016.1248932.
- Asi N, Mohammed K, Haydour Q, et al. Progesterone vs synthetic progestins and the risk of breast cancer: a systematic review and meta-analysis. Syst Rev. 2016;5:121. doi: 10.1186/ s13643-016-0294-5.
- Abenhaim HA, Suissa S, Azoulay L, et al. Menopausal hormone therapy formulation and breast cancer risk. Obstet Gynecol. 2022;139:1103-1110. doi: 10.1097/AOG.0000000000004723.
- Chlebowski RT, Rohan TE, Manson JE, et al. Breast cancer after use of estrogen plus progestin and estrogen alone: analyses of data from 2 Women’s Health Initiative randomized clinical trials. JAMA Oncol. 2015;1:296-305. doi: 10.1001/ jamaoncol.2015.0494.
- Chlebowski RT, Anderson GL, Aragaki A, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
- Cordina-Duverger E, Truong T, Anger A, et al. Risk of breast cancer by type of menopausal hormone therapy: a case-control study among postmenopausal women in France. PLoS One. 2013;8:e78016. doi: 10.1371/journal.pone.0078016.
- Simin J, Tamimi R, Lagergren J, et al. Menopausal hormone therapy and cancer risk: an overestimated risk? Eur J Cancer. 2017;84:60–8. doi: 10.1016/j.ejca. 2017.07.012.
- Stanczyk FZ, Hapgood JP, Winer S, et al. Progestogens used in postmenopausal hormone therapy: differences in their pharmacological properties, intracellular actions, and clinical effects. Endocr Rev. 2013;34:171-208. doi: 10.1210/er.20121008.
- Stute P, Wildt L, Neulen J. The impact of micronized progesterone on breast cancer risk: a systematic review. Climacteric. 2018;21:111-122. doi: 10.1080/13697137.2017.1421925.
- Anderson GL, Chlebowski RT, Aragaki A, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the Women’s Health Initiative randomised placebo-controlled trial. Lancet Oncol. 2012;13:476-486. doi: 10.1016/S1470-2045(12)70075-X.
- Chlebowski RT, Barrington W, Aragaki AK, et al. Estrogen alone and health outcomes in black women by African ancestry: a secondary analyses of a randomized controlled trial. Menopause. 2017;24:133-141. doi: 10.1097/ GME.0000000000000733.
- The NAMS 2017 Hormone Therapy Position Statement Advisory Panel. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause. 2017;24:728-753. doi: 10.1097/GME.0000000000000921.
- Pinkerton JV. Hormone therapy for postmenopausal women. N Engl J Med. 2020;382(5):446-455. doi: 10.1056/ NEJMcp1714787.
- Marchetti C, De Felice F, Boccia S, et al. Hormone replacement therapy after prophylactic risk-reducing salpingooophorectomy and breast cancer risk in BRCA1 and BRCA2 mutation carriers: a meta-analysis. Crit Rev Oncol Hematol. 2018;132:111-115. doi: 10.1016/j.critrevonc.2018.09.018.
- Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of venous thromboembolism: nested case-control studies using the QResearch and CPRD databases. BMJ. 2019;364:k4810. doi: 10.1136/bmj.k4810.
- Pinkerton JV. Hormone therapy: key points from NAMS 2017 Position Statement. Clin Obstet Gynecol. 2018;61:447453. doi: 10.1097/GRF.0000000000000383.
- Parker WH, Broder MS, Liu Z, et al. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226. doi: 10.1097/01. AOG.0000167394.38215.56.
- Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:10271037. doi: 10.1097/AOG.0b013e3181a11c64.
- Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:10741083. doi: 10.1212/01.wnl.0000276984.19542.e6.
- Rocca WA, Gazzuola Rocca L, Smith CY, et al Loss of ovarian hormones and accelerated somatic and mental aging. Physiology (Bethesda). 2018;33:374-383. doi: 10.1152/ physiol.00024.2018.
- Mytton J, Evison F, Chilton PJ, et al. Removal of all ovarian tissue versus conserving ovarian tissue at time of hysterectomy in premenopausal patients with benign disease: study using routine data and data linkage. BMJ. 2017;356:j372. doi: 10.1136/bmj.j372.
- Erickson Z, Rocca WA, Smith CY, et al. Time trends in unilateral and bilateral oophorectomy in a geographically defined American population. Obstet Gynecol. 2022;139:724-734. doi: 10.1097/AOG.0000000000004728.
- Choi YH, Terry MB, Daly MB, et al. Association of risk-reducing salpingo-oophorectomy with breast cancer risk in women with BRCA1 and BRCA2 pathogenic variants. JAMA Oncol. 2021;7:585-592. doi: 10.1001/jamaoncol.2020 .7995.
- Suzuki Y, Huang Y, Melamed A, et al. Use of estrogen therapy after surgical menopause in women who are premenopausal. Obstet Gynecol. 2022;139:756-763. doi: 10.1097/ AOG.0000000000004762.
- Faubion S, Kaunitz AM, Kapoor E. HT for women who have had BSO before the age of natural menopause: discerning the nuances. OBG Manag. 2022;34(2):20-27, 45. doi: 10.12788/ obgm.0174.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430. doi: 10.1001/ jama.2021.3305.
Should treatment be initiated for mild chronic hypertension in pregnancy to improve outcomes?
Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792. doi: 10.1056/NEJMoa2201295.
Expert Commentary
In the nonpregnant population, medical management of hypertension >140/90 mm Hg is standard practice. By contrast, much higher blood pressures (BPs; up to 160/110 mm Hg) traditionally have been tolerated in pregnant patients with chronic hypertension without initiating treatment, and existing medications are often discontinued during pregnancy. Concern for impaired fetal growth as well as lack of data on improved outcomes have led to different recommendations for the management of mild chronic hypertension in pregnancy. However, chronic hypertension affects a substantial number of pregnant patients and is known to be a risk factor for severe short-term pregnancy and long-term health complications. With preliminary data suggesting that BPs >140/90 mm Hg prior to 20 weeks’ gestation are associated with an increase in adverse outcomes, Tita and colleagues sought to determine the effects of decreasing BP in pregnant patients with mild chronic hypertension.
Details about the study
This is an investigator-initiated, multicenter, pragmatic, open-label, randomized control trial of 2,408 patients with mild chronic hypertension. The active treatment group was treated with antihypertensive medication (including titration of existing medication), targeting a BP of <140/90 mm Hg. The control group only received medication for severe hypertension (≥160 mm Hg systolic or ≥105 mm Hg diastolic). The primary outcome of the study was a composite of preeclampsia with severe features, medically indicated preterm delivery prior to 35 weeks’ gestation (not spontaneous labor or rupture of membranes), placental abruption, and fetal or neonatal death. Birthweight that was less than the 10th percentile was used as a safety outcome. The hypothesis was that treatment would decrease the rate of adverse pregnancy and fetal/neonatal outcomes.
The patient population of singleton pregnancies at a gestational age of less than 23 weeks included 56% with known chronic hypertension on medications, 22% with known chronic hypertension without medications, and 22% with newly diagnosed (during pregnancy) chronic hypertension. The treatment group primarily received labetalol (61.7%) or nifedipine (35.6%); the maximum dose of a single agent was used as tolerated prior to adding a second agent. The control group only received an antihypertensive medication for severe hypertension.
Treatment of chronic hypertension demonstrated a decreased risk of the composite adverse outcome with an adjusted risk ratio of 0.82 (95% confidence interval [CI], 0.74 to 0.92; P<.001) and a number needed to treat (NNT) of 14.7. When analyzed separately, a similar risk reduction was noted for both preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation. There was no statistical difference between the groups for birth weight <10th percentile and <5th percentile (adjusted risk ratio, 1.04 [0.82–1.31] vs 0.89 [0.62–1.26], respectively).
Planned subgroup analysis by type of chronic hypertension, race/ethnic group, diabetes status, gestational age at baseline, and body mass index (BMI) demonstrated a similar treatment effect to the overall composite primary outcome, with the exception of patients with newly diagn
Study strengths and weaknesses
The study strengths cited are a large sample size, multiple study sites, an independent data and safety monitoring board with close oversight, and centralized blinded confirmation of outcomes. Another strength is that the patient population of the study was similar to the overall population of pregnant patients in the United States with chronic hypertension in terms of age, race, and ethnicity.
The weaknesses of the study include the open-label design and the high ratio of screened to enrolled patients. Both of these issues appear related to the study design (ethics and logistics of a blinded treatment and gestational age cutoff) and the physiology of pregnancy (expected decrease in BP in the second trimester rendering patients ineligible due to lower BP). The study was also not powered to assess treatment effect in all of the subgroups, and further evaluation of patients with newly diagnosed chronic hypertension and BMI ≥ 40 kg/m2 is needed. ●
Pregnant patients with chronic hypertension should continue or initiate antihypertensive medication to target a BP goal of <140/90 mm Hg. This substantial practice change is supported by the significant decrease demonstrated in this study in adverse outcomes such as preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation without an increase in small-for-gestational-age newborns.
Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792. doi: 10.1056/NEJMoa2201295.
Expert Commentary
In the nonpregnant population, medical management of hypertension >140/90 mm Hg is standard practice. By contrast, much higher blood pressures (BPs; up to 160/110 mm Hg) traditionally have been tolerated in pregnant patients with chronic hypertension without initiating treatment, and existing medications are often discontinued during pregnancy. Concern for impaired fetal growth as well as lack of data on improved outcomes have led to different recommendations for the management of mild chronic hypertension in pregnancy. However, chronic hypertension affects a substantial number of pregnant patients and is known to be a risk factor for severe short-term pregnancy and long-term health complications. With preliminary data suggesting that BPs >140/90 mm Hg prior to 20 weeks’ gestation are associated with an increase in adverse outcomes, Tita and colleagues sought to determine the effects of decreasing BP in pregnant patients with mild chronic hypertension.
Details about the study
This is an investigator-initiated, multicenter, pragmatic, open-label, randomized control trial of 2,408 patients with mild chronic hypertension. The active treatment group was treated with antihypertensive medication (including titration of existing medication), targeting a BP of <140/90 mm Hg. The control group only received medication for severe hypertension (≥160 mm Hg systolic or ≥105 mm Hg diastolic). The primary outcome of the study was a composite of preeclampsia with severe features, medically indicated preterm delivery prior to 35 weeks’ gestation (not spontaneous labor or rupture of membranes), placental abruption, and fetal or neonatal death. Birthweight that was less than the 10th percentile was used as a safety outcome. The hypothesis was that treatment would decrease the rate of adverse pregnancy and fetal/neonatal outcomes.
The patient population of singleton pregnancies at a gestational age of less than 23 weeks included 56% with known chronic hypertension on medications, 22% with known chronic hypertension without medications, and 22% with newly diagnosed (during pregnancy) chronic hypertension. The treatment group primarily received labetalol (61.7%) or nifedipine (35.6%); the maximum dose of a single agent was used as tolerated prior to adding a second agent. The control group only received an antihypertensive medication for severe hypertension.
Treatment of chronic hypertension demonstrated a decreased risk of the composite adverse outcome with an adjusted risk ratio of 0.82 (95% confidence interval [CI], 0.74 to 0.92; P<.001) and a number needed to treat (NNT) of 14.7. When analyzed separately, a similar risk reduction was noted for both preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation. There was no statistical difference between the groups for birth weight <10th percentile and <5th percentile (adjusted risk ratio, 1.04 [0.82–1.31] vs 0.89 [0.62–1.26], respectively).
Planned subgroup analysis by type of chronic hypertension, race/ethnic group, diabetes status, gestational age at baseline, and body mass index (BMI) demonstrated a similar treatment effect to the overall composite primary outcome, with the exception of patients with newly diagn
Study strengths and weaknesses
The study strengths cited are a large sample size, multiple study sites, an independent data and safety monitoring board with close oversight, and centralized blinded confirmation of outcomes. Another strength is that the patient population of the study was similar to the overall population of pregnant patients in the United States with chronic hypertension in terms of age, race, and ethnicity.
The weaknesses of the study include the open-label design and the high ratio of screened to enrolled patients. Both of these issues appear related to the study design (ethics and logistics of a blinded treatment and gestational age cutoff) and the physiology of pregnancy (expected decrease in BP in the second trimester rendering patients ineligible due to lower BP). The study was also not powered to assess treatment effect in all of the subgroups, and further evaluation of patients with newly diagnosed chronic hypertension and BMI ≥ 40 kg/m2 is needed. ●
Pregnant patients with chronic hypertension should continue or initiate antihypertensive medication to target a BP goal of <140/90 mm Hg. This substantial practice change is supported by the significant decrease demonstrated in this study in adverse outcomes such as preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation without an increase in small-for-gestational-age newborns.
Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792. doi: 10.1056/NEJMoa2201295.
Expert Commentary
In the nonpregnant population, medical management of hypertension >140/90 mm Hg is standard practice. By contrast, much higher blood pressures (BPs; up to 160/110 mm Hg) traditionally have been tolerated in pregnant patients with chronic hypertension without initiating treatment, and existing medications are often discontinued during pregnancy. Concern for impaired fetal growth as well as lack of data on improved outcomes have led to different recommendations for the management of mild chronic hypertension in pregnancy. However, chronic hypertension affects a substantial number of pregnant patients and is known to be a risk factor for severe short-term pregnancy and long-term health complications. With preliminary data suggesting that BPs >140/90 mm Hg prior to 20 weeks’ gestation are associated with an increase in adverse outcomes, Tita and colleagues sought to determine the effects of decreasing BP in pregnant patients with mild chronic hypertension.
Details about the study
This is an investigator-initiated, multicenter, pragmatic, open-label, randomized control trial of 2,408 patients with mild chronic hypertension. The active treatment group was treated with antihypertensive medication (including titration of existing medication), targeting a BP of <140/90 mm Hg. The control group only received medication for severe hypertension (≥160 mm Hg systolic or ≥105 mm Hg diastolic). The primary outcome of the study was a composite of preeclampsia with severe features, medically indicated preterm delivery prior to 35 weeks’ gestation (not spontaneous labor or rupture of membranes), placental abruption, and fetal or neonatal death. Birthweight that was less than the 10th percentile was used as a safety outcome. The hypothesis was that treatment would decrease the rate of adverse pregnancy and fetal/neonatal outcomes.
The patient population of singleton pregnancies at a gestational age of less than 23 weeks included 56% with known chronic hypertension on medications, 22% with known chronic hypertension without medications, and 22% with newly diagnosed (during pregnancy) chronic hypertension. The treatment group primarily received labetalol (61.7%) or nifedipine (35.6%); the maximum dose of a single agent was used as tolerated prior to adding a second agent. The control group only received an antihypertensive medication for severe hypertension.
Treatment of chronic hypertension demonstrated a decreased risk of the composite adverse outcome with an adjusted risk ratio of 0.82 (95% confidence interval [CI], 0.74 to 0.92; P<.001) and a number needed to treat (NNT) of 14.7. When analyzed separately, a similar risk reduction was noted for both preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation. There was no statistical difference between the groups for birth weight <10th percentile and <5th percentile (adjusted risk ratio, 1.04 [0.82–1.31] vs 0.89 [0.62–1.26], respectively).
Planned subgroup analysis by type of chronic hypertension, race/ethnic group, diabetes status, gestational age at baseline, and body mass index (BMI) demonstrated a similar treatment effect to the overall composite primary outcome, with the exception of patients with newly diagn
Study strengths and weaknesses
The study strengths cited are a large sample size, multiple study sites, an independent data and safety monitoring board with close oversight, and centralized blinded confirmation of outcomes. Another strength is that the patient population of the study was similar to the overall population of pregnant patients in the United States with chronic hypertension in terms of age, race, and ethnicity.
The weaknesses of the study include the open-label design and the high ratio of screened to enrolled patients. Both of these issues appear related to the study design (ethics and logistics of a blinded treatment and gestational age cutoff) and the physiology of pregnancy (expected decrease in BP in the second trimester rendering patients ineligible due to lower BP). The study was also not powered to assess treatment effect in all of the subgroups, and further evaluation of patients with newly diagnosed chronic hypertension and BMI ≥ 40 kg/m2 is needed. ●
Pregnant patients with chronic hypertension should continue or initiate antihypertensive medication to target a BP goal of <140/90 mm Hg. This substantial practice change is supported by the significant decrease demonstrated in this study in adverse outcomes such as preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation without an increase in small-for-gestational-age newborns.
Best practices for evaluating pelvic pain in patients with Essure tubal occlusion devices
The evaluation and management of chronic pelvic pain in patients with a history of Essure device (Bayer HealthCare Pharmaceuticals Inc, Whippany, New Jersey) insertion have posed many challenges for both clinicians and patients. The availability of high-quality, evidence-based clinical guidance has been limited. We have reviewed the currently available published data, and here provide an overview of takeaways, as well as share our perspective and approach on evaluating and managing chronic pelvic pain in this unique patient population.
The device
The Essure microinsert is a hysteroscopically placed device that facilitates permanent sterilization by occluding the bilateral proximal fallopian tubes. The microinsert has an inner and outer nitinol coil that attaches the device to the proximal fallopian tube to ensure retention. The inner coil releases polyethylene terephthalate fibers that cause tubal fiber proliferation to occlude the lumen of the fallopian tube and achieve sterilization.
The device was first approved by the US Food and Drug Administration (FDA) in 2002. In subsequent years, the device was well received and widely used, with approximately 750,000 women worldwide undergoing Essure placement.1,2 Shortly after approval, many adverse events (AEs), including pelvic pain and abnormal uterine bleeding, were reported, resulting in a public meeting of the FDA Obstetrics and Gynecology Devices Panel in September 2015. A postmarket surveillance study on the device ensued to assess complication rates including unplanned pregnancy, pelvic pain, and surgery for removal. In February 2016, the FDA issued a black box warning and a patient decision checklist.3,4 In December 2018, Bayer stopped selling and distributing Essure in the United States.5 A 4-year follow-up surveillance study on Essure was submitted to the FDA in March 2020.
Adverse outcomes
Common AEs related to the Essure device include heavy uterine bleeding, pelvic pain, and other quality-of-life symptoms such as fatigue and weight gain.6-8 The main safety endpoints for the mandated FDA postmarket 522 surveillance studies were chronic lower abdominal and pelvic pain; abnormal uterine bleeding; hypersensitivity; allergic reaction, as well as autoimmune disorders incorporating inflammatory markers and human leukocyte antigen; and gynecologic surgery for device removal.9 Postmarket surveillence has shown that most AEs are related to placement complications or pelvic pain after Essure insertion. However, there have been several reports of autoimmune diseases categorized as serious AEs, such as new-onset systemic lupus erythematosus, rheumatoid arthritis, and worsening ulcerative colitis, after Essure insertion.5
Evaluation of symptoms
Prevalence of pelvic pain following device placement
We conducted a PubMed and MEDLINE search from January 2000 to May 2020, which identified 43 studies citing AEs related to device placement, including pelvic or abdominal pain, abnormal uterine bleeding, hypersensitivity, and autoimmune disorders. A particularly debilitating and frequently cited AE was new-onset pelvic pain or worsening of preexisting pelvic pain. Perforation of the uterus or fallopian tube, resulting in displacement of the device into the peritoneal cavity, or fragmentation of the microinsert was reported as a serious AE that occurred after device placement. However, due to the complexity of chronic pelvic pain pathogenesis, the effect of the insert on patients with existing chronic pelvic pain remains unknown.
Authors of a large retrospective study found that approximately 2.7% of 1,430 patients developed new-onset or worsening pelvic pain after device placement. New-onset pelvic pain in 1% of patients was thought to be secondary to device placement, without a coexisting pathology or diagnosis.10
In a retrospective study by Clark and colleagues, 22 of 50 women (44%) with pelvic pain after microinsert placement were found to have at least one other cause of pelvic pain. The most common alternative diagnoses were endometriosis, adenomyosis, salpingitis, and adhesive disease. Nine of the 50 patients (18%) were found to have endometriosis upon surgical removal of the microinsert.7
Another case series examined outcomes in 29 patients undergoing laparoscopic device removal due to new-onset pelvic pain. Intraoperative findings included endometriosis in 5 patients (17.2%) and pelvic adhesions in 3 (10.3%).2 Chronic pelvic pain secondary to endometriosis may be exacerbated with Essure insertion due to discontinuation of hormonal birth control after device placement,7 and this diagnosis along with adenomyosis should be strongly considered in patients whose pelvic pain began when hormonal contraception was discontinued after placement of the device.
Continue to: Risk factors...
Risk factors
Authors of a retrospective cohort study found that patients with prior diagnosis of a chronic pain syndrome, low back pain, headaches, or fibromyalgia were 5 to 6 times more likely to report acute and chronic pain after hysteroscopic sterilization with Essure.11 Since chronic pain is often thought to be driven by a hyperalgesic state of the central nervous system, as previously shown in patients with conditions such as vulvodynia, interstitial cystitis, and fibromyalgia,12 a hyperalgesic state can potentially explain why some patients are more susceptible to developing worsening pain.
Van Limburg and colleagues conducted a retrospective cohort study with prospective follow-up on 284 women who underwent Essure sterilization. Among these patients, 48% reported negative AEs; risk factors included young age at placement, increasing gravidity, and no prior abdominal surgery.13
Onset of pain
The timing and onset of pelvic pain vary widely, suggesting there is no particular time frame for this AE after device placement.2,6,14-18 A case series by Arjona and colleagues analyzed the incidence of chronic pelvic pain in 4,274 patients after Essure sterilization. Seven patients (0.16%) reported chronic pelvic pain that necessitated device removal. In 6 of the women, the pelvic pain began within 1 week of device placement. In 3 of the 6 cases, the surgeon reported the removal procedures as “difficult.” In all 6 cases, the level of pelvic pain increased with time and was not alleviated with standard analgesic medications.6
In another case series of 26 patients, the authors evaluated patients undergoing laparoscopic removal of Essure secondary to pelvic pain and reported that the time range for symptom presentation was immediate to 85 months. Thirteen of 26 patients (50%) reported pain onset within less than 1 month of device placement, 5 of 26 patients (19.2%) reported pain between 1 and 12 months after device placement, and 8 of 26 patients (30.8%) reported pain onset more than 12 months after microinsert placement.2 In this study, 17.2% of operative reports indicated difficulty with device placement. It is unclear whether difficulty with placement was associated with development of subsequent abdominal or pelvic pain; however, the relevance of initial insertion difficulty diminished with longer follow-up.
Workup and evaluation
We found 5 studies that provided some framework for evaluating a patient with new-onset or worsening pelvic pain after microinsert placement. Overall, correct placement and functionality of the device should be confirmed by either hysterosalpingogram (HSG) or transvaginal ultrasonography (TVUS). The gold standard to determine tubal occlusion is the HSG. However, TVUS may be a dependable alternative, and either test can accurately demonstrate Essure location.19 Patients often prefer TVUS over HSG due to the low cost, minimal discomfort, and short examination time.1 TVUS is a noninvasive and reasonable test to start the initial assessment. The Essure devices are highly echogenic on pelvic ultrasound and easily identifiable by the proximity of the device to the uterotubal junction and its relationship with the surrounding soft tissue. If the device perforates the peritoneal cavity, then the echogenic bowel can impede adequate visualization of the Essure microinsert. If the Essure insert is not visualized on TVUS, an HSG will not only confirm placement but also test insert functionality. After confirming correct placement of the device, the provider can proceed with standard workup for chronic pelvic pain.
If one or more of the devices are malpositioned, the devices are generally presumed to be the etiology of the new pain. Multiple case reports demonstrate pain due to Essure misconfiguration or perforation with subsequent resolution of symptoms after device removal.18,20,21 A case study by Alcantara and colleagues described a patient with chronic pelvic pain and an Essure coil that was curved in an elliptical shape, not adhering to the anatomic course of the fallopian tube. The patient reported pain resolution after laparoscopic removal of the device.20 Another case report by Mahmoud et al described a subserosal malpositioned device that caused acute pelvic pain 4 months after sterilization. The patient reported resolution of pain after the microinsert was removed via laparoscopy.21 These reports highlight the importance of considering malpositioned devices as the etiology of new pelvic pain after Essure placement.

Continue to: Device removal and patient outcomes...
Device removal and patient outcomes
Removal
Several studies that we evaluated included a discussion on the methods for Essure removal. which are divided into 2 general categories: hysteroscopy and laparoscopy.
Hysteroscopic removal is generally used when the device was placed less than 12 weeks prior to removal.7,19 After 12 weeks, removal is more difficult due to fibrosis within the fallopian tubes. A risk with hysteroscopic removal is failure to remove all fibers, which allows inflammation and fibrosis to continue.7 This risk is mitigated via laparoscopic hysterectomy or mini-cornuectomy with bilateral salpingectomy, where the devices can be removed en bloc and without excessive traction.
Laparoscopic Essure removal procedures described in the literature include salpingostomy and traction on the device, salpingectomy, and salpingectomy with mini-cornuectomy. The incision and traction method is typically performed via a 2- to 3-cm incision on the antimesial edge of the fallopian tube along with a circumferential incision to surround the interstitial tubal area. The implant is carefully extracted from the fallopian tube and cornua, and a salpingectomy is then performed.22 The implant is removed prior to the salpingectomy to ensure that the Essure device is removed in its entirety prior to performing a salpingectomy.
A prospective observational study evaluated laparoscopic removal of Essure devices in 80 women with or without cornual excision. Results suggest that the incision and traction method poses more technical difficulties than the cornuectomy approach.23 Surgeons reported significant difficulty controlling the tensile pressure with traction, whereas use of the cornuectomy approach eliminated this risk and decreased the risk of fragmentation and incomplete removal.23,24
Charavil and colleagues demonstrated in a prospective observational study that a vaginal hysterectomy with bilateral salpingectomy is a feasible approach to Essure removal. Twenty-six vaginal hysterectomies with bilateral salpingectomy and Essure removal were performed without conversion to laparoscopy or laparotomy. The surgeons performed an en bloc removal of each hemiuterus along with the ipsilateral tube, which ensured complete removal of the Essure device. Each case was confirmed with an x-ray of the surgical specimen.25
If device fragmentation occurs, there are different methods recommended for locating fragments. A case report of bilateral uterine perforation after uncomplicated Essure placement used a preoperative computed tomography (CT) scan to locate the Essure fragments, but no intraoperative imaging was performed to confirm complete fragment removal.26 The patient continued reporting chronic pelvic pain and ultimately underwent exploratory laparotomy with intraoperative fluoroscopy. Using fluoroscopy, investigators identified omental fragments that were missed on preoperative CT imaging. Fluoroscopy is not commonly used intraoperatively, but it may have added benefit for localizing retained fragments.
A retrospective cohort study reviewed the use of intraoperative x-ray of the removed specimen to confirm complete Essure removal.27 If an x-ray of the removed specimen showed incomplete removal, an intraoperative pelvic x-ray was performed to locate missing fragments. X-ray of the removed devices confirmed complete removal in 63 of 72 patients (87.5%). Six of 9 women with an unsatisfactory specimen x-ray had no residual fragments identified during pelvic x-ray, and the device removal was deemed adequate. The remaining 3 women had radiologic evidence of incomplete device removal and required additional dissection for complete removal. Overall, use of x-ray or fluoroscopy is a relatively safe and accessible way to ensure complete removal of the Essure device and is worth consideration, especially when retained device fragments are suspected.
Symptom resolution
We reviewed 5 studies that examined pain outcomes after removal of the Essure devices. Casey et al found that 23 of 26 patients (88.5%) reported significant pain relief at the postoperative visit, while 3 of 26 (11.5%) reported persistent pelvic pain.2 Two of 3 case series examined other outcomes in addition to postoperative pelvic pain, including sexual function and activities of daily living.7,14 In the first case series by Brito and colleagues, 8 of 11 patients (72.7%) reported an improvement in pelvic pain, ability to perform daily activities, sexual life, and overall quality of life after Essure removal. For the remaining 3 patients with persistent pelvic pain after surgical removal of the device, 2 patients reported worsening pain symptoms and dyspareunia.14 In this study, 5 of 11 patients reported a history of chronic pelvic pain at baseline. In a retrospective case series by Clark et al, 28 of 32 women (87.5%) reported some improvement in all domains, with 24 of 32 patients (75%) reporting almost total or complete improvement in quality of life, sexual life, pelvic pain, and scores related to activities of daily living. Pain and quality-of-life scores were similar for women who underwent uterine-preserving surgery and for those who underwent hysterectomy. Ten of 32 women (31.3%) reported persistent or worsening symptoms after the Essure removal surgery. In these patients, the authors recommended consideration of other autoimmune and hypersensitivity etiologies.7
In a retrospective cohort study by Kamencic et al from 2002 to 2013 of 1,430 patients who underwent Essure placement with postplacement imaging, 62 patients (4.3%) required a second surgery after Essure placement due to pelvic pain.10 This study also found that 4 of 62 patients (0.3%) had no other obvious cause for the pelvic pain. All 4 of these women had complete resolution of their pain with removal of the Essure microinsert device. A prospective observational study by Chene e
Summary
Although Essure products were withdrawn from the market in the United States in 2018, many patients still experience significant AEs associated with the device. The goal of the perspectives and data presented here is to assist clinicians in addressing and managing the pain experienced by patients after device insertion. ●
- Connor VF. Essure: a review six years later. J Minim Invasive Gynecol. 2009;16:282-290. doi:10.1016/j.jmig.2009.02.009.
- Casey J, Aguirre F, Yunker A. Outcomes of laparoscopic removal of the Essure sterilization device for pelvic pain: a case series. Contraception. 2016;94:190-192. doi:10.1016/j.contraception.2016.03.017.
- Jackson I. Essure device removed entirely from market, with 99% of unused birth control implants retrieved: FDA. AboutLawsuits.com. January 13, 2020. https://www.aboutlawsuits.com/Essure-removal-update-166509. Accessed June 7, 2022.
- US Food and Drug Administration. Labeling for permanent hysteroscopically-placed tubal implants intended for sterilization. October 31, 2016. https://www.fda.gov/media/96315/download. Accessed June 7, 2022.
- US Food and Drug Administration. FDA activities related to Essure. March 14, 2022. https://www.fda.gov/medical-devices/essure-permanent-birth-control/fda-activities-related-essure. Accessed June 8, 2022.
- Arjona Berral JE, Rodríguez Jiménez B, Velasco Sánchez E, et al. Essure and chronic pelvic pain: a population-based cohort. J Obstet Gynaecol. 2014;34:712-713. doi:10.3109/01443615.2014.92075.
- Clark NV, Rademaker D, Mushinski AA, et al. Essure removal for the treatment of device-attributed symptoms: an expanded case series and follow-up survey. J Minim Invasive Gynecol. 2017;24:971-976. doi:10.1016/j.jmig.2017.05.015.
- Sills ES, Rickers NS, Li X. Surgical management after hysteroscopic sterilization: minimally invasive approach incorporating intraoperative fluoroscopy for symptomatic patients with >2 Essure devices. Surg Technol Int. 2018;32:156-161.
- Administration USF and D. 522 Postmarket Surveillance Studies. Center for Devices and Radiological Health; 2020.
- Kamencic H, Thiel L, Karreman E, et al. Does Essure cause significant de novo pain? A retrospective review of indications for second surgeries after Essure placement. J Minim Invasive Gynecol. 2016;23:1158-1162. doi:10.1016/j.jmig.2016.08.823.
- Yunker AC, Ritch JM, Robinson EF, et al. Incidence and risk factors for chronic pelvic pain after hysteroscopic sterilization. J Minim Invasive Gynecol. 2015;22:390-994. doi:10.1016/j.jmig.2014.06.007.
- Phillips K, Clauw DJ. Central pain mechanisms in chronic pain states--maybe it is all in their head. Best Pract Res Clin Rheumatol. 2011;25:141-154. doi:10.1016/j.berh.2011.02.005.
- van Limburg Stirum EVJ, Clark NV, Lindsey A, et al. Factors associated with negative patient experiences with Essure sterilization. JSLS. 2020;24(1):e2019.00065. doi:10.4293/JSLS.2019.00065.
- Brito LG, Cohen SL, Goggins ER, et al. Essure surgical removal and subsequent symptom resolution: case series and follow-up survey. J Minim Invasive Gynecol. 2015;22:910-913. doi:10.1016/j.jmig.2015.03.018.
- Maassen LW, van Gastel DM, Haveman I, et al. Removal of Essure sterilization devices: a retrospective cohort study in the Netherlands. J Minim Invasive Gynecol. 2019;26:1056-1062. doi:10.1016/j.jmig.2018.10.009.
- Sills ES, Palermo GD. Surgical excision of Essure devices with ESHRE class IIb uterine malformation: sequential hysteroscopic-laparoscopic approach to the septate uterus. Facts Views Vis Obgyn. 2016;8:49-52.
- Ricci G, Restaino S, Di Lorenzo G, et al. Risk of Essure microinsert abdominal migration: case report and review of literature. Ther Clin Risk Manag. 2014;10:963-968. doi:10.2147/TCRM.S65634.
- Borley J, Shabajee N, Tan TL. A kink is not always a perforation: assessing Essure hysteroscopic sterilization placement. Fertil Steril. 2011;95:2429.e15-7. doi:10.1016/j.fertnstert.2011.02.006.
- Djeffal H, Blouet M, Pizzoferato AC, et al. Imaging findings in Essure-related complications: a pictorial review.7Br J Radiol. 2018;91(1090):20170686. doi:10.1259/bjr.20170686.
- Lora Alcantara I, Rezai S, Kirby C, et al. Essure surgical removal and subsequent resolution of chronic pelvic pain: a case report and review of the literature. Case Rep Obstet Gynecol. 2016;2016:6961202. doi:10.1155/2016/6961202.
- Mahmoud MS, Fridman D, Merhi ZO. Subserosal misplacement of Essure device manifested by late-onset acute pelvic pain. Fertil Steril. 2009;92:2038.e1-3. doi:10.1016/j.fertnstert.2009.07.1677.
- Tissot M, Petry S, Lecointre L, et al. Two surgical techniques for Essure device ablation: the hysteroscopic way and the laparoscopic way by salpingectomy with tubal interstitial resection. J Minim Invasive Gynecol. 2019;26(4):603. doi:10.1016/j.jmig.2018.07.017.
- Chene G, Cerruto E, Moret S, et al. Quality of life after laparoscopic removal of Essure sterilization devices. Eur J Obstet Gynecol Reprod Biol X. 2019;3:100054. doi:10.1016/j.eurox.2019.100054.
- Thiel L, Rattray D, Thiel J. Laparoscopic cornuectomy as a technique for removal of Essure microinserts. J Minim Invasive Gynecol. 2017;24(1):10. doi:10.1016/j.jmig.2016.07.004.
- Charavil A, Agostini A, Rambeaud C, et al. Vaginal hysterectomy with salpingectomy for Essure insert removal. J Minim Invasive Gynecol. 2019;2:695-701. doi:10.1016/j.jmig.2018.07.019.
- Howard DL, Christenson PJ, Strickland JL. Use of intraoperative fluoroscopy during laparotomy to identify fragments of retained Essure microinserts: case report. J Minim Invasive Gynecol. 2012;19:667-670. doi:10.1016/j.jmig.2012.04.007.
- Miquel L, Crochet P, Francini S, et al. Laparoscopic Essure device removal by en bloc salpingectomy-cornuectomy with intraoperative x-ray checking: a retrospective cohort study. J Minim Invasive Gynecol. 2020;27:697-703. doi:10.1016/j. jmig.2019.06.006.
The evaluation and management of chronic pelvic pain in patients with a history of Essure device (Bayer HealthCare Pharmaceuticals Inc, Whippany, New Jersey) insertion have posed many challenges for both clinicians and patients. The availability of high-quality, evidence-based clinical guidance has been limited. We have reviewed the currently available published data, and here provide an overview of takeaways, as well as share our perspective and approach on evaluating and managing chronic pelvic pain in this unique patient population.
The device
The Essure microinsert is a hysteroscopically placed device that facilitates permanent sterilization by occluding the bilateral proximal fallopian tubes. The microinsert has an inner and outer nitinol coil that attaches the device to the proximal fallopian tube to ensure retention. The inner coil releases polyethylene terephthalate fibers that cause tubal fiber proliferation to occlude the lumen of the fallopian tube and achieve sterilization.
The device was first approved by the US Food and Drug Administration (FDA) in 2002. In subsequent years, the device was well received and widely used, with approximately 750,000 women worldwide undergoing Essure placement.1,2 Shortly after approval, many adverse events (AEs), including pelvic pain and abnormal uterine bleeding, were reported, resulting in a public meeting of the FDA Obstetrics and Gynecology Devices Panel in September 2015. A postmarket surveillance study on the device ensued to assess complication rates including unplanned pregnancy, pelvic pain, and surgery for removal. In February 2016, the FDA issued a black box warning and a patient decision checklist.3,4 In December 2018, Bayer stopped selling and distributing Essure in the United States.5 A 4-year follow-up surveillance study on Essure was submitted to the FDA in March 2020.
Adverse outcomes
Common AEs related to the Essure device include heavy uterine bleeding, pelvic pain, and other quality-of-life symptoms such as fatigue and weight gain.6-8 The main safety endpoints for the mandated FDA postmarket 522 surveillance studies were chronic lower abdominal and pelvic pain; abnormal uterine bleeding; hypersensitivity; allergic reaction, as well as autoimmune disorders incorporating inflammatory markers and human leukocyte antigen; and gynecologic surgery for device removal.9 Postmarket surveillence has shown that most AEs are related to placement complications or pelvic pain after Essure insertion. However, there have been several reports of autoimmune diseases categorized as serious AEs, such as new-onset systemic lupus erythematosus, rheumatoid arthritis, and worsening ulcerative colitis, after Essure insertion.5
Evaluation of symptoms
Prevalence of pelvic pain following device placement
We conducted a PubMed and MEDLINE search from January 2000 to May 2020, which identified 43 studies citing AEs related to device placement, including pelvic or abdominal pain, abnormal uterine bleeding, hypersensitivity, and autoimmune disorders. A particularly debilitating and frequently cited AE was new-onset pelvic pain or worsening of preexisting pelvic pain. Perforation of the uterus or fallopian tube, resulting in displacement of the device into the peritoneal cavity, or fragmentation of the microinsert was reported as a serious AE that occurred after device placement. However, due to the complexity of chronic pelvic pain pathogenesis, the effect of the insert on patients with existing chronic pelvic pain remains unknown.
Authors of a large retrospective study found that approximately 2.7% of 1,430 patients developed new-onset or worsening pelvic pain after device placement. New-onset pelvic pain in 1% of patients was thought to be secondary to device placement, without a coexisting pathology or diagnosis.10
In a retrospective study by Clark and colleagues, 22 of 50 women (44%) with pelvic pain after microinsert placement were found to have at least one other cause of pelvic pain. The most common alternative diagnoses were endometriosis, adenomyosis, salpingitis, and adhesive disease. Nine of the 50 patients (18%) were found to have endometriosis upon surgical removal of the microinsert.7
Another case series examined outcomes in 29 patients undergoing laparoscopic device removal due to new-onset pelvic pain. Intraoperative findings included endometriosis in 5 patients (17.2%) and pelvic adhesions in 3 (10.3%).2 Chronic pelvic pain secondary to endometriosis may be exacerbated with Essure insertion due to discontinuation of hormonal birth control after device placement,7 and this diagnosis along with adenomyosis should be strongly considered in patients whose pelvic pain began when hormonal contraception was discontinued after placement of the device.
Continue to: Risk factors...
Risk factors
Authors of a retrospective cohort study found that patients with prior diagnosis of a chronic pain syndrome, low back pain, headaches, or fibromyalgia were 5 to 6 times more likely to report acute and chronic pain after hysteroscopic sterilization with Essure.11 Since chronic pain is often thought to be driven by a hyperalgesic state of the central nervous system, as previously shown in patients with conditions such as vulvodynia, interstitial cystitis, and fibromyalgia,12 a hyperalgesic state can potentially explain why some patients are more susceptible to developing worsening pain.
Van Limburg and colleagues conducted a retrospective cohort study with prospective follow-up on 284 women who underwent Essure sterilization. Among these patients, 48% reported negative AEs; risk factors included young age at placement, increasing gravidity, and no prior abdominal surgery.13
Onset of pain
The timing and onset of pelvic pain vary widely, suggesting there is no particular time frame for this AE after device placement.2,6,14-18 A case series by Arjona and colleagues analyzed the incidence of chronic pelvic pain in 4,274 patients after Essure sterilization. Seven patients (0.16%) reported chronic pelvic pain that necessitated device removal. In 6 of the women, the pelvic pain began within 1 week of device placement. In 3 of the 6 cases, the surgeon reported the removal procedures as “difficult.” In all 6 cases, the level of pelvic pain increased with time and was not alleviated with standard analgesic medications.6
In another case series of 26 patients, the authors evaluated patients undergoing laparoscopic removal of Essure secondary to pelvic pain and reported that the time range for symptom presentation was immediate to 85 months. Thirteen of 26 patients (50%) reported pain onset within less than 1 month of device placement, 5 of 26 patients (19.2%) reported pain between 1 and 12 months after device placement, and 8 of 26 patients (30.8%) reported pain onset more than 12 months after microinsert placement.2 In this study, 17.2% of operative reports indicated difficulty with device placement. It is unclear whether difficulty with placement was associated with development of subsequent abdominal or pelvic pain; however, the relevance of initial insertion difficulty diminished with longer follow-up.
Workup and evaluation
We found 5 studies that provided some framework for evaluating a patient with new-onset or worsening pelvic pain after microinsert placement. Overall, correct placement and functionality of the device should be confirmed by either hysterosalpingogram (HSG) or transvaginal ultrasonography (TVUS). The gold standard to determine tubal occlusion is the HSG. However, TVUS may be a dependable alternative, and either test can accurately demonstrate Essure location.19 Patients often prefer TVUS over HSG due to the low cost, minimal discomfort, and short examination time.1 TVUS is a noninvasive and reasonable test to start the initial assessment. The Essure devices are highly echogenic on pelvic ultrasound and easily identifiable by the proximity of the device to the uterotubal junction and its relationship with the surrounding soft tissue. If the device perforates the peritoneal cavity, then the echogenic bowel can impede adequate visualization of the Essure microinsert. If the Essure insert is not visualized on TVUS, an HSG will not only confirm placement but also test insert functionality. After confirming correct placement of the device, the provider can proceed with standard workup for chronic pelvic pain.
If one or more of the devices are malpositioned, the devices are generally presumed to be the etiology of the new pain. Multiple case reports demonstrate pain due to Essure misconfiguration or perforation with subsequent resolution of symptoms after device removal.18,20,21 A case study by Alcantara and colleagues described a patient with chronic pelvic pain and an Essure coil that was curved in an elliptical shape, not adhering to the anatomic course of the fallopian tube. The patient reported pain resolution after laparoscopic removal of the device.20 Another case report by Mahmoud et al described a subserosal malpositioned device that caused acute pelvic pain 4 months after sterilization. The patient reported resolution of pain after the microinsert was removed via laparoscopy.21 These reports highlight the importance of considering malpositioned devices as the etiology of new pelvic pain after Essure placement.

Continue to: Device removal and patient outcomes...
Device removal and patient outcomes
Removal
Several studies that we evaluated included a discussion on the methods for Essure removal. which are divided into 2 general categories: hysteroscopy and laparoscopy.
Hysteroscopic removal is generally used when the device was placed less than 12 weeks prior to removal.7,19 After 12 weeks, removal is more difficult due to fibrosis within the fallopian tubes. A risk with hysteroscopic removal is failure to remove all fibers, which allows inflammation and fibrosis to continue.7 This risk is mitigated via laparoscopic hysterectomy or mini-cornuectomy with bilateral salpingectomy, where the devices can be removed en bloc and without excessive traction.
Laparoscopic Essure removal procedures described in the literature include salpingostomy and traction on the device, salpingectomy, and salpingectomy with mini-cornuectomy. The incision and traction method is typically performed via a 2- to 3-cm incision on the antimesial edge of the fallopian tube along with a circumferential incision to surround the interstitial tubal area. The implant is carefully extracted from the fallopian tube and cornua, and a salpingectomy is then performed.22 The implant is removed prior to the salpingectomy to ensure that the Essure device is removed in its entirety prior to performing a salpingectomy.
A prospective observational study evaluated laparoscopic removal of Essure devices in 80 women with or without cornual excision. Results suggest that the incision and traction method poses more technical difficulties than the cornuectomy approach.23 Surgeons reported significant difficulty controlling the tensile pressure with traction, whereas use of the cornuectomy approach eliminated this risk and decreased the risk of fragmentation and incomplete removal.23,24
Charavil and colleagues demonstrated in a prospective observational study that a vaginal hysterectomy with bilateral salpingectomy is a feasible approach to Essure removal. Twenty-six vaginal hysterectomies with bilateral salpingectomy and Essure removal were performed without conversion to laparoscopy or laparotomy. The surgeons performed an en bloc removal of each hemiuterus along with the ipsilateral tube, which ensured complete removal of the Essure device. Each case was confirmed with an x-ray of the surgical specimen.25
If device fragmentation occurs, there are different methods recommended for locating fragments. A case report of bilateral uterine perforation after uncomplicated Essure placement used a preoperative computed tomography (CT) scan to locate the Essure fragments, but no intraoperative imaging was performed to confirm complete fragment removal.26 The patient continued reporting chronic pelvic pain and ultimately underwent exploratory laparotomy with intraoperative fluoroscopy. Using fluoroscopy, investigators identified omental fragments that were missed on preoperative CT imaging. Fluoroscopy is not commonly used intraoperatively, but it may have added benefit for localizing retained fragments.
A retrospective cohort study reviewed the use of intraoperative x-ray of the removed specimen to confirm complete Essure removal.27 If an x-ray of the removed specimen showed incomplete removal, an intraoperative pelvic x-ray was performed to locate missing fragments. X-ray of the removed devices confirmed complete removal in 63 of 72 patients (87.5%). Six of 9 women with an unsatisfactory specimen x-ray had no residual fragments identified during pelvic x-ray, and the device removal was deemed adequate. The remaining 3 women had radiologic evidence of incomplete device removal and required additional dissection for complete removal. Overall, use of x-ray or fluoroscopy is a relatively safe and accessible way to ensure complete removal of the Essure device and is worth consideration, especially when retained device fragments are suspected.

Symptom resolution
We reviewed 5 studies that examined pain outcomes after removal of the Essure devices. Casey et al found that 23 of 26 patients (88.5%) reported significant pain relief at the postoperative visit, while 3 of 26 (11.5%) reported persistent pelvic pain.2 Two of 3 case series examined other outcomes in addition to postoperative pelvic pain, including sexual function and activities of daily living.7,14 In the first case series by Brito and colleagues, 8 of 11 patients (72.7%) reported an improvement in pelvic pain, ability to perform daily activities, sexual life, and overall quality of life after Essure removal. For the remaining 3 patients with persistent pelvic pain after surgical removal of the device, 2 patients reported worsening pain symptoms and dyspareunia.14 In this study, 5 of 11 patients reported a history of chronic pelvic pain at baseline. In a retrospective case series by Clark et al, 28 of 32 women (87.5%) reported some improvement in all domains, with 24 of 32 patients (75%) reporting almost total or complete improvement in quality of life, sexual life, pelvic pain, and scores related to activities of daily living. Pain and quality-of-life scores were similar for women who underwent uterine-preserving surgery and for those who underwent hysterectomy. Ten of 32 women (31.3%) reported persistent or worsening symptoms after the Essure removal surgery. In these patients, the authors recommended consideration of other autoimmune and hypersensitivity etiologies.7
In a retrospective cohort study by Kamencic et al from 2002 to 2013 of 1,430 patients who underwent Essure placement with postplacement imaging, 62 patients (4.3%) required a second surgery after Essure placement due to pelvic pain.10 This study also found that 4 of 62 patients (0.3%) had no other obvious cause for the pelvic pain. All 4 of these women had complete resolution of their pain with removal of the Essure microinsert device. A prospective observational study by Chene e
Summary
Although Essure products were withdrawn from the market in the United States in 2018, many patients still experience significant AEs associated with the device. The goal of the perspectives and data presented here is to assist clinicians in addressing and managing the pain experienced by patients after device insertion. ●
The evaluation and management of chronic pelvic pain in patients with a history of Essure device (Bayer HealthCare Pharmaceuticals Inc, Whippany, New Jersey) insertion have posed many challenges for both clinicians and patients. The availability of high-quality, evidence-based clinical guidance has been limited. We have reviewed the currently available published data, and here provide an overview of takeaways, as well as share our perspective and approach on evaluating and managing chronic pelvic pain in this unique patient population.
The device
The Essure microinsert is a hysteroscopically placed device that facilitates permanent sterilization by occluding the bilateral proximal fallopian tubes. The microinsert has an inner and outer nitinol coil that attaches the device to the proximal fallopian tube to ensure retention. The inner coil releases polyethylene terephthalate fibers that cause tubal fiber proliferation to occlude the lumen of the fallopian tube and achieve sterilization.
The device was first approved by the US Food and Drug Administration (FDA) in 2002. In subsequent years, the device was well received and widely used, with approximately 750,000 women worldwide undergoing Essure placement.1,2 Shortly after approval, many adverse events (AEs), including pelvic pain and abnormal uterine bleeding, were reported, resulting in a public meeting of the FDA Obstetrics and Gynecology Devices Panel in September 2015. A postmarket surveillance study on the device ensued to assess complication rates including unplanned pregnancy, pelvic pain, and surgery for removal. In February 2016, the FDA issued a black box warning and a patient decision checklist.3,4 In December 2018, Bayer stopped selling and distributing Essure in the United States.5 A 4-year follow-up surveillance study on Essure was submitted to the FDA in March 2020.
Adverse outcomes
Common AEs related to the Essure device include heavy uterine bleeding, pelvic pain, and other quality-of-life symptoms such as fatigue and weight gain.6-8 The main safety endpoints for the mandated FDA postmarket 522 surveillance studies were chronic lower abdominal and pelvic pain; abnormal uterine bleeding; hypersensitivity; allergic reaction, as well as autoimmune disorders incorporating inflammatory markers and human leukocyte antigen; and gynecologic surgery for device removal.9 Postmarket surveillence has shown that most AEs are related to placement complications or pelvic pain after Essure insertion. However, there have been several reports of autoimmune diseases categorized as serious AEs, such as new-onset systemic lupus erythematosus, rheumatoid arthritis, and worsening ulcerative colitis, after Essure insertion.5
Evaluation of symptoms
Prevalence of pelvic pain following device placement
We conducted a PubMed and MEDLINE search from January 2000 to May 2020, which identified 43 studies citing AEs related to device placement, including pelvic or abdominal pain, abnormal uterine bleeding, hypersensitivity, and autoimmune disorders. A particularly debilitating and frequently cited AE was new-onset pelvic pain or worsening of preexisting pelvic pain. Perforation of the uterus or fallopian tube, resulting in displacement of the device into the peritoneal cavity, or fragmentation of the microinsert was reported as a serious AE that occurred after device placement. However, due to the complexity of chronic pelvic pain pathogenesis, the effect of the insert on patients with existing chronic pelvic pain remains unknown.
Authors of a large retrospective study found that approximately 2.7% of 1,430 patients developed new-onset or worsening pelvic pain after device placement. New-onset pelvic pain in 1% of patients was thought to be secondary to device placement, without a coexisting pathology or diagnosis.10
In a retrospective study by Clark and colleagues, 22 of 50 women (44%) with pelvic pain after microinsert placement were found to have at least one other cause of pelvic pain. The most common alternative diagnoses were endometriosis, adenomyosis, salpingitis, and adhesive disease. Nine of the 50 patients (18%) were found to have endometriosis upon surgical removal of the microinsert.7
Another case series examined outcomes in 29 patients undergoing laparoscopic device removal due to new-onset pelvic pain. Intraoperative findings included endometriosis in 5 patients (17.2%) and pelvic adhesions in 3 (10.3%).2 Chronic pelvic pain secondary to endometriosis may be exacerbated with Essure insertion due to discontinuation of hormonal birth control after device placement,7 and this diagnosis along with adenomyosis should be strongly considered in patients whose pelvic pain began when hormonal contraception was discontinued after placement of the device.
Continue to: Risk factors...
Risk factors
Authors of a retrospective cohort study found that patients with prior diagnosis of a chronic pain syndrome, low back pain, headaches, or fibromyalgia were 5 to 6 times more likely to report acute and chronic pain after hysteroscopic sterilization with Essure.11 Since chronic pain is often thought to be driven by a hyperalgesic state of the central nervous system, as previously shown in patients with conditions such as vulvodynia, interstitial cystitis, and fibromyalgia,12 a hyperalgesic state can potentially explain why some patients are more susceptible to developing worsening pain.
Van Limburg and colleagues conducted a retrospective cohort study with prospective follow-up on 284 women who underwent Essure sterilization. Among these patients, 48% reported negative AEs; risk factors included young age at placement, increasing gravidity, and no prior abdominal surgery.13
Onset of pain
The timing and onset of pelvic pain vary widely, suggesting there is no particular time frame for this AE after device placement.2,6,14-18 A case series by Arjona and colleagues analyzed the incidence of chronic pelvic pain in 4,274 patients after Essure sterilization. Seven patients (0.16%) reported chronic pelvic pain that necessitated device removal. In 6 of the women, the pelvic pain began within 1 week of device placement. In 3 of the 6 cases, the surgeon reported the removal procedures as “difficult.” In all 6 cases, the level of pelvic pain increased with time and was not alleviated with standard analgesic medications.6
In another case series of 26 patients, the authors evaluated patients undergoing laparoscopic removal of Essure secondary to pelvic pain and reported that the time range for symptom presentation was immediate to 85 months. Thirteen of 26 patients (50%) reported pain onset within less than 1 month of device placement, 5 of 26 patients (19.2%) reported pain between 1 and 12 months after device placement, and 8 of 26 patients (30.8%) reported pain onset more than 12 months after microinsert placement.2 In this study, 17.2% of operative reports indicated difficulty with device placement. It is unclear whether difficulty with placement was associated with development of subsequent abdominal or pelvic pain; however, the relevance of initial insertion difficulty diminished with longer follow-up.
Workup and evaluation
We found 5 studies that provided some framework for evaluating a patient with new-onset or worsening pelvic pain after microinsert placement. Overall, correct placement and functionality of the device should be confirmed by either hysterosalpingogram (HSG) or transvaginal ultrasonography (TVUS). The gold standard to determine tubal occlusion is the HSG. However, TVUS may be a dependable alternative, and either test can accurately demonstrate Essure location.19 Patients often prefer TVUS over HSG due to the low cost, minimal discomfort, and short examination time.1 TVUS is a noninvasive and reasonable test to start the initial assessment. The Essure devices are highly echogenic on pelvic ultrasound and easily identifiable by the proximity of the device to the uterotubal junction and its relationship with the surrounding soft tissue. If the device perforates the peritoneal cavity, then the echogenic bowel can impede adequate visualization of the Essure microinsert. If the Essure insert is not visualized on TVUS, an HSG will not only confirm placement but also test insert functionality. After confirming correct placement of the device, the provider can proceed with standard workup for chronic pelvic pain.
If one or more of the devices are malpositioned, the devices are generally presumed to be the etiology of the new pain. Multiple case reports demonstrate pain due to Essure misconfiguration or perforation with subsequent resolution of symptoms after device removal.18,20,21 A case study by Alcantara and colleagues described a patient with chronic pelvic pain and an Essure coil that was curved in an elliptical shape, not adhering to the anatomic course of the fallopian tube. The patient reported pain resolution after laparoscopic removal of the device.20 Another case report by Mahmoud et al described a subserosal malpositioned device that caused acute pelvic pain 4 months after sterilization. The patient reported resolution of pain after the microinsert was removed via laparoscopy.21 These reports highlight the importance of considering malpositioned devices as the etiology of new pelvic pain after Essure placement.

Continue to: Device removal and patient outcomes...
Device removal and patient outcomes
Removal
Several studies that we evaluated included a discussion on the methods for Essure removal. which are divided into 2 general categories: hysteroscopy and laparoscopy.
Hysteroscopic removal is generally used when the device was placed less than 12 weeks prior to removal.7,19 After 12 weeks, removal is more difficult due to fibrosis within the fallopian tubes. A risk with hysteroscopic removal is failure to remove all fibers, which allows inflammation and fibrosis to continue.7 This risk is mitigated via laparoscopic hysterectomy or mini-cornuectomy with bilateral salpingectomy, where the devices can be removed en bloc and without excessive traction.
Laparoscopic Essure removal procedures described in the literature include salpingostomy and traction on the device, salpingectomy, and salpingectomy with mini-cornuectomy. The incision and traction method is typically performed via a 2- to 3-cm incision on the antimesial edge of the fallopian tube along with a circumferential incision to surround the interstitial tubal area. The implant is carefully extracted from the fallopian tube and cornua, and a salpingectomy is then performed.22 The implant is removed prior to the salpingectomy to ensure that the Essure device is removed in its entirety prior to performing a salpingectomy.
A prospective observational study evaluated laparoscopic removal of Essure devices in 80 women with or without cornual excision. Results suggest that the incision and traction method poses more technical difficulties than the cornuectomy approach.23 Surgeons reported significant difficulty controlling the tensile pressure with traction, whereas use of the cornuectomy approach eliminated this risk and decreased the risk of fragmentation and incomplete removal.23,24
Charavil and colleagues demonstrated in a prospective observational study that a vaginal hysterectomy with bilateral salpingectomy is a feasible approach to Essure removal. Twenty-six vaginal hysterectomies with bilateral salpingectomy and Essure removal were performed without conversion to laparoscopy or laparotomy. The surgeons performed an en bloc removal of each hemiuterus along with the ipsilateral tube, which ensured complete removal of the Essure device. Each case was confirmed with an x-ray of the surgical specimen.25
If device fragmentation occurs, there are different methods recommended for locating fragments. A case report of bilateral uterine perforation after uncomplicated Essure placement used a preoperative computed tomography (CT) scan to locate the Essure fragments, but no intraoperative imaging was performed to confirm complete fragment removal.26 The patient continued reporting chronic pelvic pain and ultimately underwent exploratory laparotomy with intraoperative fluoroscopy. Using fluoroscopy, investigators identified omental fragments that were missed on preoperative CT imaging. Fluoroscopy is not commonly used intraoperatively, but it may have added benefit for localizing retained fragments.
A retrospective cohort study reviewed the use of intraoperative x-ray of the removed specimen to confirm complete Essure removal.27 If an x-ray of the removed specimen showed incomplete removal, an intraoperative pelvic x-ray was performed to locate missing fragments. X-ray of the removed devices confirmed complete removal in 63 of 72 patients (87.5%). Six of 9 women with an unsatisfactory specimen x-ray had no residual fragments identified during pelvic x-ray, and the device removal was deemed adequate. The remaining 3 women had radiologic evidence of incomplete device removal and required additional dissection for complete removal. Overall, use of x-ray or fluoroscopy is a relatively safe and accessible way to ensure complete removal of the Essure device and is worth consideration, especially when retained device fragments are suspected.

Symptom resolution
We reviewed 5 studies that examined pain outcomes after removal of the Essure devices. Casey et al found that 23 of 26 patients (88.5%) reported significant pain relief at the postoperative visit, while 3 of 26 (11.5%) reported persistent pelvic pain.2 Two of 3 case series examined other outcomes in addition to postoperative pelvic pain, including sexual function and activities of daily living.7,14 In the first case series by Brito and colleagues, 8 of 11 patients (72.7%) reported an improvement in pelvic pain, ability to perform daily activities, sexual life, and overall quality of life after Essure removal. For the remaining 3 patients with persistent pelvic pain after surgical removal of the device, 2 patients reported worsening pain symptoms and dyspareunia.14 In this study, 5 of 11 patients reported a history of chronic pelvic pain at baseline. In a retrospective case series by Clark et al, 28 of 32 women (87.5%) reported some improvement in all domains, with 24 of 32 patients (75%) reporting almost total or complete improvement in quality of life, sexual life, pelvic pain, and scores related to activities of daily living. Pain and quality-of-life scores were similar for women who underwent uterine-preserving surgery and for those who underwent hysterectomy. Ten of 32 women (31.3%) reported persistent or worsening symptoms after the Essure removal surgery. In these patients, the authors recommended consideration of other autoimmune and hypersensitivity etiologies.7
In a retrospective cohort study by Kamencic et al from 2002 to 2013 of 1,430 patients who underwent Essure placement with postplacement imaging, 62 patients (4.3%) required a second surgery after Essure placement due to pelvic pain.10 This study also found that 4 of 62 patients (0.3%) had no other obvious cause for the pelvic pain. All 4 of these women had complete resolution of their pain with removal of the Essure microinsert device. A prospective observational study by Chene e
Summary
Although Essure products were withdrawn from the market in the United States in 2018, many patients still experience significant AEs associated with the device. The goal of the perspectives and data presented here is to assist clinicians in addressing and managing the pain experienced by patients after device insertion. ●
- Connor VF. Essure: a review six years later. J Minim Invasive Gynecol. 2009;16:282-290. doi:10.1016/j.jmig.2009.02.009.
- Casey J, Aguirre F, Yunker A. Outcomes of laparoscopic removal of the Essure sterilization device for pelvic pain: a case series. Contraception. 2016;94:190-192. doi:10.1016/j.contraception.2016.03.017.
- Jackson I. Essure device removed entirely from market, with 99% of unused birth control implants retrieved: FDA. AboutLawsuits.com. January 13, 2020. https://www.aboutlawsuits.com/Essure-removal-update-166509. Accessed June 7, 2022.
- US Food and Drug Administration. Labeling for permanent hysteroscopically-placed tubal implants intended for sterilization. October 31, 2016. https://www.fda.gov/media/96315/download. Accessed June 7, 2022.
- US Food and Drug Administration. FDA activities related to Essure. March 14, 2022. https://www.fda.gov/medical-devices/essure-permanent-birth-control/fda-activities-related-essure. Accessed June 8, 2022.
- Arjona Berral JE, Rodríguez Jiménez B, Velasco Sánchez E, et al. Essure and chronic pelvic pain: a population-based cohort. J Obstet Gynaecol. 2014;34:712-713. doi:10.3109/01443615.2014.92075.
- Clark NV, Rademaker D, Mushinski AA, et al. Essure removal for the treatment of device-attributed symptoms: an expanded case series and follow-up survey. J Minim Invasive Gynecol. 2017;24:971-976. doi:10.1016/j.jmig.2017.05.015.
- Sills ES, Rickers NS, Li X. Surgical management after hysteroscopic sterilization: minimally invasive approach incorporating intraoperative fluoroscopy for symptomatic patients with >2 Essure devices. Surg Technol Int. 2018;32:156-161.
- Administration USF and D. 522 Postmarket Surveillance Studies. Center for Devices and Radiological Health; 2020.
- Kamencic H, Thiel L, Karreman E, et al. Does Essure cause significant de novo pain? A retrospective review of indications for second surgeries after Essure placement. J Minim Invasive Gynecol. 2016;23:1158-1162. doi:10.1016/j.jmig.2016.08.823.
- Yunker AC, Ritch JM, Robinson EF, et al. Incidence and risk factors for chronic pelvic pain after hysteroscopic sterilization. J Minim Invasive Gynecol. 2015;22:390-994. doi:10.1016/j.jmig.2014.06.007.
- Phillips K, Clauw DJ. Central pain mechanisms in chronic pain states--maybe it is all in their head. Best Pract Res Clin Rheumatol. 2011;25:141-154. doi:10.1016/j.berh.2011.02.005.
- van Limburg Stirum EVJ, Clark NV, Lindsey A, et al. Factors associated with negative patient experiences with Essure sterilization. JSLS. 2020;24(1):e2019.00065. doi:10.4293/JSLS.2019.00065.
- Brito LG, Cohen SL, Goggins ER, et al. Essure surgical removal and subsequent symptom resolution: case series and follow-up survey. J Minim Invasive Gynecol. 2015;22:910-913. doi:10.1016/j.jmig.2015.03.018.
- Maassen LW, van Gastel DM, Haveman I, et al. Removal of Essure sterilization devices: a retrospective cohort study in the Netherlands. J Minim Invasive Gynecol. 2019;26:1056-1062. doi:10.1016/j.jmig.2018.10.009.
- Sills ES, Palermo GD. Surgical excision of Essure devices with ESHRE class IIb uterine malformation: sequential hysteroscopic-laparoscopic approach to the septate uterus. Facts Views Vis Obgyn. 2016;8:49-52.
- Ricci G, Restaino S, Di Lorenzo G, et al. Risk of Essure microinsert abdominal migration: case report and review of literature. Ther Clin Risk Manag. 2014;10:963-968. doi:10.2147/TCRM.S65634.
- Borley J, Shabajee N, Tan TL. A kink is not always a perforation: assessing Essure hysteroscopic sterilization placement. Fertil Steril. 2011;95:2429.e15-7. doi:10.1016/j.fertnstert.2011.02.006.
- Djeffal H, Blouet M, Pizzoferato AC, et al. Imaging findings in Essure-related complications: a pictorial review.7Br J Radiol. 2018;91(1090):20170686. doi:10.1259/bjr.20170686.
- Lora Alcantara I, Rezai S, Kirby C, et al. Essure surgical removal and subsequent resolution of chronic pelvic pain: a case report and review of the literature. Case Rep Obstet Gynecol. 2016;2016:6961202. doi:10.1155/2016/6961202.
- Mahmoud MS, Fridman D, Merhi ZO. Subserosal misplacement of Essure device manifested by late-onset acute pelvic pain. Fertil Steril. 2009;92:2038.e1-3. doi:10.1016/j.fertnstert.2009.07.1677.
- Tissot M, Petry S, Lecointre L, et al. Two surgical techniques for Essure device ablation: the hysteroscopic way and the laparoscopic way by salpingectomy with tubal interstitial resection. J Minim Invasive Gynecol. 2019;26(4):603. doi:10.1016/j.jmig.2018.07.017.
- Chene G, Cerruto E, Moret S, et al. Quality of life after laparoscopic removal of Essure sterilization devices. Eur J Obstet Gynecol Reprod Biol X. 2019;3:100054. doi:10.1016/j.eurox.2019.100054.
- Thiel L, Rattray D, Thiel J. Laparoscopic cornuectomy as a technique for removal of Essure microinserts. J Minim Invasive Gynecol. 2017;24(1):10. doi:10.1016/j.jmig.2016.07.004.
- Charavil A, Agostini A, Rambeaud C, et al. Vaginal hysterectomy with salpingectomy for Essure insert removal. J Minim Invasive Gynecol. 2019;2:695-701. doi:10.1016/j.jmig.2018.07.019.
- Howard DL, Christenson PJ, Strickland JL. Use of intraoperative fluoroscopy during laparotomy to identify fragments of retained Essure microinserts: case report. J Minim Invasive Gynecol. 2012;19:667-670. doi:10.1016/j.jmig.2012.04.007.
- Miquel L, Crochet P, Francini S, et al. Laparoscopic Essure device removal by en bloc salpingectomy-cornuectomy with intraoperative x-ray checking: a retrospective cohort study. J Minim Invasive Gynecol. 2020;27:697-703. doi:10.1016/j. jmig.2019.06.006.
- Connor VF. Essure: a review six years later. J Minim Invasive Gynecol. 2009;16:282-290. doi:10.1016/j.jmig.2009.02.009.
- Casey J, Aguirre F, Yunker A. Outcomes of laparoscopic removal of the Essure sterilization device for pelvic pain: a case series. Contraception. 2016;94:190-192. doi:10.1016/j.contraception.2016.03.017.
- Jackson I. Essure device removed entirely from market, with 99% of unused birth control implants retrieved: FDA. AboutLawsuits.com. January 13, 2020. https://www.aboutlawsuits.com/Essure-removal-update-166509. Accessed June 7, 2022.
- US Food and Drug Administration. Labeling for permanent hysteroscopically-placed tubal implants intended for sterilization. October 31, 2016. https://www.fda.gov/media/96315/download. Accessed June 7, 2022.
- US Food and Drug Administration. FDA activities related to Essure. March 14, 2022. https://www.fda.gov/medical-devices/essure-permanent-birth-control/fda-activities-related-essure. Accessed June 8, 2022.
- Arjona Berral JE, Rodríguez Jiménez B, Velasco Sánchez E, et al. Essure and chronic pelvic pain: a population-based cohort. J Obstet Gynaecol. 2014;34:712-713. doi:10.3109/01443615.2014.92075.
- Clark NV, Rademaker D, Mushinski AA, et al. Essure removal for the treatment of device-attributed symptoms: an expanded case series and follow-up survey. J Minim Invasive Gynecol. 2017;24:971-976. doi:10.1016/j.jmig.2017.05.015.
- Sills ES, Rickers NS, Li X. Surgical management after hysteroscopic sterilization: minimally invasive approach incorporating intraoperative fluoroscopy for symptomatic patients with >2 Essure devices. Surg Technol Int. 2018;32:156-161.
- Administration USF and D. 522 Postmarket Surveillance Studies. Center for Devices and Radiological Health; 2020.
- Kamencic H, Thiel L, Karreman E, et al. Does Essure cause significant de novo pain? A retrospective review of indications for second surgeries after Essure placement. J Minim Invasive Gynecol. 2016;23:1158-1162. doi:10.1016/j.jmig.2016.08.823.
- Yunker AC, Ritch JM, Robinson EF, et al. Incidence and risk factors for chronic pelvic pain after hysteroscopic sterilization. J Minim Invasive Gynecol. 2015;22:390-994. doi:10.1016/j.jmig.2014.06.007.
- Phillips K, Clauw DJ. Central pain mechanisms in chronic pain states--maybe it is all in their head. Best Pract Res Clin Rheumatol. 2011;25:141-154. doi:10.1016/j.berh.2011.02.005.
- van Limburg Stirum EVJ, Clark NV, Lindsey A, et al. Factors associated with negative patient experiences with Essure sterilization. JSLS. 2020;24(1):e2019.00065. doi:10.4293/JSLS.2019.00065.
- Brito LG, Cohen SL, Goggins ER, et al. Essure surgical removal and subsequent symptom resolution: case series and follow-up survey. J Minim Invasive Gynecol. 2015;22:910-913. doi:10.1016/j.jmig.2015.03.018.
- Maassen LW, van Gastel DM, Haveman I, et al. Removal of Essure sterilization devices: a retrospective cohort study in the Netherlands. J Minim Invasive Gynecol. 2019;26:1056-1062. doi:10.1016/j.jmig.2018.10.009.
- Sills ES, Palermo GD. Surgical excision of Essure devices with ESHRE class IIb uterine malformation: sequential hysteroscopic-laparoscopic approach to the septate uterus. Facts Views Vis Obgyn. 2016;8:49-52.
- Ricci G, Restaino S, Di Lorenzo G, et al. Risk of Essure microinsert abdominal migration: case report and review of literature. Ther Clin Risk Manag. 2014;10:963-968. doi:10.2147/TCRM.S65634.
- Borley J, Shabajee N, Tan TL. A kink is not always a perforation: assessing Essure hysteroscopic sterilization placement. Fertil Steril. 2011;95:2429.e15-7. doi:10.1016/j.fertnstert.2011.02.006.
- Djeffal H, Blouet M, Pizzoferato AC, et al. Imaging findings in Essure-related complications: a pictorial review.7Br J Radiol. 2018;91(1090):20170686. doi:10.1259/bjr.20170686.
- Lora Alcantara I, Rezai S, Kirby C, et al. Essure surgical removal and subsequent resolution of chronic pelvic pain: a case report and review of the literature. Case Rep Obstet Gynecol. 2016;2016:6961202. doi:10.1155/2016/6961202.
- Mahmoud MS, Fridman D, Merhi ZO. Subserosal misplacement of Essure device manifested by late-onset acute pelvic pain. Fertil Steril. 2009;92:2038.e1-3. doi:10.1016/j.fertnstert.2009.07.1677.
- Tissot M, Petry S, Lecointre L, et al. Two surgical techniques for Essure device ablation: the hysteroscopic way and the laparoscopic way by salpingectomy with tubal interstitial resection. J Minim Invasive Gynecol. 2019;26(4):603. doi:10.1016/j.jmig.2018.07.017.
- Chene G, Cerruto E, Moret S, et al. Quality of life after laparoscopic removal of Essure sterilization devices. Eur J Obstet Gynecol Reprod Biol X. 2019;3:100054. doi:10.1016/j.eurox.2019.100054.
- Thiel L, Rattray D, Thiel J. Laparoscopic cornuectomy as a technique for removal of Essure microinserts. J Minim Invasive Gynecol. 2017;24(1):10. doi:10.1016/j.jmig.2016.07.004.
- Charavil A, Agostini A, Rambeaud C, et al. Vaginal hysterectomy with salpingectomy for Essure insert removal. J Minim Invasive Gynecol. 2019;2:695-701. doi:10.1016/j.jmig.2018.07.019.
- Howard DL, Christenson PJ, Strickland JL. Use of intraoperative fluoroscopy during laparotomy to identify fragments of retained Essure microinserts: case report. J Minim Invasive Gynecol. 2012;19:667-670. doi:10.1016/j.jmig.2012.04.007.
- Miquel L, Crochet P, Francini S, et al. Laparoscopic Essure device removal by en bloc salpingectomy-cornuectomy with intraoperative x-ray checking: a retrospective cohort study. J Minim Invasive Gynecol. 2020;27:697-703. doi:10.1016/j. jmig.2019.06.006.
Misoprostol: Clinical pharmacology in obstetrics and gynecology
Oxytocin and prostaglandins are critically important regulators of uterine contraction. Obstetrician-gynecologists commonly prescribe oxytocin and prostaglandin agonists (misoprostol, dinoprostone) to stimulate uterine contraction for the induction of labor, prevention and treatment of postpartum hemorrhage, and treatment of miscarriage and fetal demise. The focus of this editorial is the clinical pharmacology of misoprostol.
Misoprostol is approved by the US Food and Drug Administration (FDA) for the prevention and treatment of nonsteroidal anti-inflammatory drug–induced gastric ulcers and for patients at high risk for gastric ulcers, including those with a history of gastric ulcers. The approved misoprostol route and dose for this indication is oral administration of 200 µg four times daily with food.1 Recent food intake and antacid use reduces the absorption of orally administered misoprostol. There are no FDA-approved indications for the use of misoprostol as a single agent in obstetrics and gynecology. The FDA has approved the combination of mifepristone and misoprostol for medication abortion in the first trimester. In contrast to misoprostol, PGE2 (dinoprostone) is approved by the FDA as a vaginal insert containing 10 mg of dinoprostone for the initiation and/or continuation of cervical ripening in patients at or near term in whom there is a medical or obstetric indication for induction of labor (Cervidil; Ferring Pharmaceuticals Inc, Parsippany, New Jersey).2
Pharmacology of misoprostol
Misoprostol is a prostaglandin E1 (PGE1) agonist analogue. Prostaglandin E1 (alprostadil) is rapidly metabolized, has a half-life in the range of minutes and is not orally active, requiring administration by intravenous infusion or injection. It is indicated to maintain a patent ductus arteriosus in newborns with ductal-dependent circulation and to treat erectile dysfunction.3 In contrast to PGE1, misoprostol has a methyl ester group at carbon-1 (C-1) that increases potency and duration of action. Misoprostol also has no hydroxyl group at C-15, replacing that moiety with the addition of both a methyl- and hydroxyl- group at C-16 (FIGURE). These molecular changes improve oral activity and increase duration of action.4 Pure misoprostol is a viscous oil. It is formulated into tables by dispersing the oil on hydroxypropyl methyl cellulose before compounding into tablets. Unlike naturally occurring prostaglandins (PGE1), misoprostol tablets are stabile at room temperature for years.4
Following absorption, the methyl ester at C-1 is enzymatically cleaved, yielding misoprostol acid, the active drug.4 Misoprostol binds to the E prostanoid receptor 3 (EP-3).5 Activation of myometrial EP-3 receptor induces an increase in intracellular phosphoinositol turnover and calcium mobilization, resulting in an increase in intracellular-free calcium, triggering actin-myosin contractility.6 The increase in free calcium is propagated cell-to-cell through gap junctions that link the myometrial cells to facilitate the generation of a coordinated contraction.
Misoprostol: Various routes of administration are not equal
Misoprostol can be given by an oral, buccal, vaginal, or rectal route of administration. To study the effect of the route of administration on uterine tone and contractility, investigators randomly assigned patients at 8 to 11 weeks’ gestation to receive misoprostol 400 µg as a single dose by the oral or vaginal route. Uterine tone and contractility were measured using an intrauterine pressure transducer. Compared to vaginal administration, oral administration of misprostol was associated with rapid attainment of peak plasma level at 30 minutes, followed by a decline in concentration by 60 minutes. This rapid onset and rapid offset of plasma concentration was paralleled by the onset of uterine tone within 8 minutes, but surprisingly no sustained uterine contractions.7 By contrast, following vaginal administration of misoprostol, serum levels rose slowly and peaked in 1 to 2 hours. Uterine tone increased within 21 minutes, and sustained uterine contractions were recorded for 4 hours.7 The rapid rise and fall in plasma misoprostol following oral administration and the more sustained plasma misoprostol concentration over 4 hours has been previously reported.8 In a second study involving patients 8 to 11 weeks’ gestation, the effect of a single dose of misoprostol 400 µg by an oral or vaginal route on uterine contractility was compared using an intrauterine pressure transducer.9 Confirming previous results, the time from misoprostol administration to increased uterine tone was more rapid with oral than with vaginal administration (8 min vs 19 min). Over the course of 4 hours, uterine contraction activity was greater with vaginal than with oral administration (454 vs 166 Montevideo units).9
Both studies reported that oral administration of misoprostol resulted in more rapid onset and offset of action than vaginal administration. Oral administration of a single dose of misoprostol 400 µg did not result in sustained uterine contractions in most patients in the first trimester. Vaginal administration produced a slower onset of increased uterine tone but sustained uterine contractions over 4 hours. Compared with vaginal administration of misoprostol, the rapid onset and offset of action of oral misoprostol may reduce the rate of tachysystole and changes in fetal heart rate observed with vaginal administration.10
An important finding is that buccal and vaginal administration of misoprostol have similar effects on uterine tone in the first trimester.11 To study the effect of buccal and vaginal administration of misoprostol on uterine tone, patients 6 to 13 weeks’ gestation were randomly allocated to receive a single dose of misoprostol 400 µg by a buccal or vaginal route.11 Uterine activity over 5 hours following administration was assessed using an intrauterine pressure transducer. Uterine tone 20 to 30 minutes after buccal or vaginal administration of misoprostol (400 µg) was 27 and 28 mm Hg, respectively. Peak uterine tone, as measured by an intrauterine pressure transducer, for buccal and vaginal administration of misoprostol was 49 mm Hg and 54 mm Hg, respectively. Total Alexandria units (AU) over 5 hours following buccal or vaginal administration was 6,537 AU and 6,090 AU, respectively.11
An AU is calculated as the average amplitude of the contractions (mm Hg) multiplied by the average duration of the contractions (min) multiplied by average frequency of contraction over 10 minutes.12 By contrast, a Montevideo unit does not include an assessment of contraction duration and is calculated as average amplitude of contractions (mm Hg) multiplied by frequency of uterine contractions over 10 minutes.12
In contrast to buccal or vaginal administration, rectal administration of misoprostol resulted in much lower peak uterine tone and contractility as measured by a pressure transducer. Uterine tone 20 to 30 minutes after vaginal and rectal administration of misoprostol (400 µg) was 28 and 19 mm Hg, respectively.11 Peak uterine tone, as measured by an intrauterine pressure transducer, for vaginal and rectal administration of misoprostol was 54 and 31 mm Hg, respectively. AUs over 5 hours following vaginal and rectal administration was 6,090 AU and 2,768 AU, respectively.11 Compared with buccal and vaginal administration of misoprostol, rectal administration produced less sustained uterine contractions in the first trimester of pregnancy. To achieve maximal sustained uterine contractions, buccal and vaginal routes of administration are superior to oral and rectal administration.
Continue to: Misoprostol and cervical ripening...
Misoprostol and cervical ripening
Misoprostol is commonly used to soften and ripen the cervix. Some of the cervical ripening effects of misoprostol are likely due to increased uterine tone. In addition, misoprostol may have a direct effect on the collagen structure of the cervix. To study the effect of misoprostol on the cervix, pregnant patients in the first trimester were randomly assigned to receive misoprostol 200 µg by vaginal self-administration, isosorbide mononitrate (IMN) 40 mg by vaginal self-administration or no treatment the evening prior to pregnancy termination.13 The following day, before uterine evacuation, a cervical biopsy was obtained for electron microscopy studies and immunohistochemistry to assess the presence of enzymes involved in collagen degradation, including matrix metalloproteinase 1 (MMP-1) and matrix metalloproteinase 9 (MMP-9). Electron microscopy demonstrated that pretreatment with misoprostol resulted in a pronounced splitting and disorganization of collagen fibers.13 Compared with misoprostol treatment, IMN produced less splitting and disorganization of collagen fibers, and in the no treatment group, no marked changes in the collagen framework were observed.
Compared with no treatment, misoprostol and IMN pretreatment were associated with marked increases in MMP-1 and MMP-9 as assessed by immunohistochemistry. Misoprostol pretreatment also resulted in a significant increase in interleukin-8 concentration compared with IMN pretreatment and no treatment (8.8 vs 2.7 vs 2.4 pg/mg tissue), respectively.13 Other investigators have also reported that misoprostol increased cervical leukocyte influx and collagen disrupting enzymes MMP-8 and MMP-9.14,15
An open-label clinical trial compared the efficacy of misoprostol versus Foley catheter for labor induction at term in 1,859 patients ≥ 37 weeks’ gestation with a Bishop score <6.16 Patients were randomly allocated to misoprostol (50 µg orally every 4 hours up to 3 times in 24 hours) versus placement of a 16 F or 18 F Foley catheter introduced through the cervix, filled with 30 mL of sodium chloride or water. The investigators reported that oral misoprostol and Foley catheter cervical ripening had similar safety and effectiveness for cervical ripening as a prelude to induction of labor, including no statistically significant differences in 5-minute Apgar score <7, umbilical cord artery pH ≤ 7.05, postpartum hemorrhage, or cesarean birth rate.16
Bottom line
Misoprostol and oxytocin are commonly prescribed in obstetric practice for cervical ripening and induction of labor, respectively. The dose and route of administration of misoprostol influences the effect on the uterus. For cervical ripening, where rapid onset and offset may help to reduce the risk of uterine tachysystole and worrisome fetal heart rate changes, low-dose (50 µg) oral administration of misoprostol may be a preferred dose and route. For the treatment of miscarriage and fetal demise, to stimulate sustained uterine contractions over many hours, buccal and vaginal administration of misoprostol are preferred. Rectal administration is generally inferior to buccal and vaginal administration for stimulating sustained uterine contractions and its uses should be limited. ●
Common side effects of misoprostol are abdominal cramping, diarrhea, nausea, vomiting, headache, and fever. Elevated temperature following misoprostol administration is a concerning side effect that may require further investigation to rule out an infection, especially if the elevated temperature persists for > 4 hours. The preoptic area of the anterior hypothalamus (POAH) plays a major role in thermoregulation. When an infection causes an increase in endogenous pyrogens, including interleukin-1β, interleukin-6 and tumor necrosis factor, prostaglandins are generated in the region of the POAH, increasing the thermoregulatory set point, triggering cutaneous vasoconstriction and shivering and non-shivering thermogenesis.1 Misoprostol, especially at doses >400 µg commonly causes both patient-reported chills and temperature elevation >38° C.
In a study comparing misoprostol and oxytocin for the management of the third stage of labor, 597 patients were randomly allocated to receive oxytocin 10 units by intramuscular injection or misoprostol 400 µg or 600 µg by the oral route.2 Patient-reported shivering occurred in 13%, 19%, and 28% of patients receiving oxytocin, misoprostol 400 µg and misoprostol 800 µg, respectively. A recorded temperature >38° C occurred within 1 hour of medication administration in approximately 3%, 2%, and 7.5% of patients receiving oxytocin, misoprostol 400 µg, and misoprostol 800 µg, respectively. In another study, 453 patients scheduled for a cesarean birth were randomly allocated to receive 1 of 3 doses of rectal misoprostol 200 μg, 400 μg, or 600 μg before incision. Fever was detected in 2.6%, 9.9%, and 5.1% of the patients receiving misoprostol 200 μg, 400 μg, or 600 μg, respectively.3
References
1. Aronoff DM, Neilson EG. Antipyretics: mechanisms of action and clinical use in fever suppression. Am J Med. 2001;111:304-315. doi: 10.1016/s0002-9343(01)00834-8.
2. Lumbiganon P, Hofmeyr J, Gumezoglu AM, et al. Misoprostol dose-related shivering and pyrexia in the third stage of labor. WHO Collaborative Trial of Misoprostol in the Management of the Third Stage of Labor. Br J Obstet Gynaecol. 1999;106:304-308. doi: 10.1111/j.1471-0528.1999.tb08266.x.
3. Sweed M, El-Said M, Abou-Gamrah AA, et al. Comparison between 200, 400 and 600 microgram rectal misoprostol before cesarean section: a randomized clinical trial. J Obstet Gynaecol Res. 2019;45:585-591. doi: 10.1111 /jog.13883.
- Cytotec [package insert]. Chicago, IL: GD Searle & Co. https://www.accessdata.fda.gov/drugsatfda_docs/label/2002/19268slr037.pdf. Accessed June 20, 2022.
- Cervidil [package insert]. St Louis, MO: Forrest Pharmaceuticals Inc.; May 2006. Accessed June 20, 2022.
- Caverject [package insert]. New York, NY: Pfizer Inc.; March 2014. Accessed June 20, 2022.
- Collins PW. Misoprostol: discovery, development and clinical applications. Med Res Rev. 1990;10:149-172. doi: 10.1002/med.2610100202.
- Audit M, White KI, Breton B, et al. Crystal structure of misoprostol bound to the labor inducer prostaglandin E2 receptor. Nat Chem Biol. 2019;15:11-17. doi: 10.1038/s41589-018-0160-y.
- Pallliser KH, Hirst JJ, Ooi G, et al. Prostaglandin E and F receptor expression and myometrial sensitivity in labor onset in the sheep. Biol Reprod. 2005;72:937-943. doi: 10.1095/biolreprod.104.035311.
- Gemzell-Danilesson K, Marions L, Rodriguez A, et al. Comparison between oral and vaginal administration of misoprostol on uterine contractility. Obstet Gynecol. 1999;93:275-280. doi: 10.1016/s0029-7844(98)00436-0.
- Zieman M, Fong SK, Benowitz NL, et al. Absorption kinetics of misoprostol with oral or vaginal administration. Obstet Gynecol. 1997;90:88-92. doi: 10.1016/S0029-7844(97)00111-7.
- Aronsson A, Bygdeman M, Gemzell-Danielsson K. Effects of misoprostol on uterine contractility following different routes of administration. Hum Reprod. 2004;19:81-84. doi: 10.1093/humrep/deh005.
- Young DC, Delaney T, Armson BA, et al. Oral misoprostol, low dose vaginal misoprostol and vaginal dinoprostone for labor induction: randomized controlled trial. PLOS One. 2020;15:e0227245. doi: 10.1371/journal.pone.0227245.
- Meckstroth KR, Whitaker AK, Bertisch S, et al. Misoprostol administered by epithelial routes. Drug absorption and uterine response. Obstet Gynecol. 2006;108:582-590. doi: 10.1097/01.AOG.0000230398.32794.9d.
- el-Sahwi S, Gaafar AA, Toppozada HK. A new unit for evaluation of uterine activity. Am J Obstet Gynecol. 1967;98:900-903. doi: 10.1016/0002-9378(67)90074-9.
- Vukas N, Ekerhovd E, Abrahamsson G, et al. Cervical priming in the first trimester: morphological and biochemical effects of misoprostol and isosorbide mononitrate. Acta Obstet Gyecol. 2009;88:43-51. doi: 10.1080/00016340802585440.
- Aronsson A, Ulfgren AK, Stabi B, et al. The effect of orally and vaginally administered misoprostol on inflammatory mediators and cervical ripening during early pregnancy. Contraception. 2005;72:33-39. doi: 10.1016/j.contraception.2005.02.012.
- Denison FC, Riley SC, Elliott CL, et al. The effect of mifepristone administration on leukocyte populations, matrix metalloproteinases and inflammatory mediators in the first trimester cervix. Mol Hum Reprod. 2000;6:541-548. doi: 10.1093/molehr/6.6.541.
- ten Eikelder MLG, Rengerink KO, Jozwiak M, et al. Induction of labour at term with oral misoprostol versus a Foley catheter (PROBAAT-II): a multicentre randomised controlled non-inferiority trial. Lancet. 2016;387:1619-1628. doi: 10.1016 /S0140-6736(16)00084-2.
Oxytocin and prostaglandins are critically important regulators of uterine contraction. Obstetrician-gynecologists commonly prescribe oxytocin and prostaglandin agonists (misoprostol, dinoprostone) to stimulate uterine contraction for the induction of labor, prevention and treatment of postpartum hemorrhage, and treatment of miscarriage and fetal demise. The focus of this editorial is the clinical pharmacology of misoprostol.
Misoprostol is approved by the US Food and Drug Administration (FDA) for the prevention and treatment of nonsteroidal anti-inflammatory drug–induced gastric ulcers and for patients at high risk for gastric ulcers, including those with a history of gastric ulcers. The approved misoprostol route and dose for this indication is oral administration of 200 µg four times daily with food.1 Recent food intake and antacid use reduces the absorption of orally administered misoprostol. There are no FDA-approved indications for the use of misoprostol as a single agent in obstetrics and gynecology. The FDA has approved the combination of mifepristone and misoprostol for medication abortion in the first trimester. In contrast to misoprostol, PGE2 (dinoprostone) is approved by the FDA as a vaginal insert containing 10 mg of dinoprostone for the initiation and/or continuation of cervical ripening in patients at or near term in whom there is a medical or obstetric indication for induction of labor (Cervidil; Ferring Pharmaceuticals Inc, Parsippany, New Jersey).2
Pharmacology of misoprostol
Misoprostol is a prostaglandin E1 (PGE1) agonist analogue. Prostaglandin E1 (alprostadil) is rapidly metabolized, has a half-life in the range of minutes and is not orally active, requiring administration by intravenous infusion or injection. It is indicated to maintain a patent ductus arteriosus in newborns with ductal-dependent circulation and to treat erectile dysfunction.3 In contrast to PGE1, misoprostol has a methyl ester group at carbon-1 (C-1) that increases potency and duration of action. Misoprostol also has no hydroxyl group at C-15, replacing that moiety with the addition of both a methyl- and hydroxyl- group at C-16 (FIGURE). These molecular changes improve oral activity and increase duration of action.4 Pure misoprostol is a viscous oil. It is formulated into tables by dispersing the oil on hydroxypropyl methyl cellulose before compounding into tablets. Unlike naturally occurring prostaglandins (PGE1), misoprostol tablets are stabile at room temperature for years.4
Following absorption, the methyl ester at C-1 is enzymatically cleaved, yielding misoprostol acid, the active drug.4 Misoprostol binds to the E prostanoid receptor 3 (EP-3).5 Activation of myometrial EP-3 receptor induces an increase in intracellular phosphoinositol turnover and calcium mobilization, resulting in an increase in intracellular-free calcium, triggering actin-myosin contractility.6 The increase in free calcium is propagated cell-to-cell through gap junctions that link the myometrial cells to facilitate the generation of a coordinated contraction.

Misoprostol: Various routes of administration are not equal
Misoprostol can be given by an oral, buccal, vaginal, or rectal route of administration. To study the effect of the route of administration on uterine tone and contractility, investigators randomly assigned patients at 8 to 11 weeks’ gestation to receive misoprostol 400 µg as a single dose by the oral or vaginal route. Uterine tone and contractility were measured using an intrauterine pressure transducer. Compared to vaginal administration, oral administration of misprostol was associated with rapid attainment of peak plasma level at 30 minutes, followed by a decline in concentration by 60 minutes. This rapid onset and rapid offset of plasma concentration was paralleled by the onset of uterine tone within 8 minutes, but surprisingly no sustained uterine contractions.7 By contrast, following vaginal administration of misoprostol, serum levels rose slowly and peaked in 1 to 2 hours. Uterine tone increased within 21 minutes, and sustained uterine contractions were recorded for 4 hours.7 The rapid rise and fall in plasma misoprostol following oral administration and the more sustained plasma misoprostol concentration over 4 hours has been previously reported.8 In a second study involving patients 8 to 11 weeks’ gestation, the effect of a single dose of misoprostol 400 µg by an oral or vaginal route on uterine contractility was compared using an intrauterine pressure transducer.9 Confirming previous results, the time from misoprostol administration to increased uterine tone was more rapid with oral than with vaginal administration (8 min vs 19 min). Over the course of 4 hours, uterine contraction activity was greater with vaginal than with oral administration (454 vs 166 Montevideo units).9
Both studies reported that oral administration of misoprostol resulted in more rapid onset and offset of action than vaginal administration. Oral administration of a single dose of misoprostol 400 µg did not result in sustained uterine contractions in most patients in the first trimester. Vaginal administration produced a slower onset of increased uterine tone but sustained uterine contractions over 4 hours. Compared with vaginal administration of misoprostol, the rapid onset and offset of action of oral misoprostol may reduce the rate of tachysystole and changes in fetal heart rate observed with vaginal administration.10
An important finding is that buccal and vaginal administration of misoprostol have similar effects on uterine tone in the first trimester.11 To study the effect of buccal and vaginal administration of misoprostol on uterine tone, patients 6 to 13 weeks’ gestation were randomly allocated to receive a single dose of misoprostol 400 µg by a buccal or vaginal route.11 Uterine activity over 5 hours following administration was assessed using an intrauterine pressure transducer. Uterine tone 20 to 30 minutes after buccal or vaginal administration of misoprostol (400 µg) was 27 and 28 mm Hg, respectively. Peak uterine tone, as measured by an intrauterine pressure transducer, for buccal and vaginal administration of misoprostol was 49 mm Hg and 54 mm Hg, respectively. Total Alexandria units (AU) over 5 hours following buccal or vaginal administration was 6,537 AU and 6,090 AU, respectively.11
An AU is calculated as the average amplitude of the contractions (mm Hg) multiplied by the average duration of the contractions (min) multiplied by average frequency of contraction over 10 minutes.12 By contrast, a Montevideo unit does not include an assessment of contraction duration and is calculated as average amplitude of contractions (mm Hg) multiplied by frequency of uterine contractions over 10 minutes.12
In contrast to buccal or vaginal administration, rectal administration of misoprostol resulted in much lower peak uterine tone and contractility as measured by a pressure transducer. Uterine tone 20 to 30 minutes after vaginal and rectal administration of misoprostol (400 µg) was 28 and 19 mm Hg, respectively.11 Peak uterine tone, as measured by an intrauterine pressure transducer, for vaginal and rectal administration of misoprostol was 54 and 31 mm Hg, respectively. AUs over 5 hours following vaginal and rectal administration was 6,090 AU and 2,768 AU, respectively.11 Compared with buccal and vaginal administration of misoprostol, rectal administration produced less sustained uterine contractions in the first trimester of pregnancy. To achieve maximal sustained uterine contractions, buccal and vaginal routes of administration are superior to oral and rectal administration.
Continue to: Misoprostol and cervical ripening...
Misoprostol and cervical ripening
Misoprostol is commonly used to soften and ripen the cervix. Some of the cervical ripening effects of misoprostol are likely due to increased uterine tone. In addition, misoprostol may have a direct effect on the collagen structure of the cervix. To study the effect of misoprostol on the cervix, pregnant patients in the first trimester were randomly assigned to receive misoprostol 200 µg by vaginal self-administration, isosorbide mononitrate (IMN) 40 mg by vaginal self-administration or no treatment the evening prior to pregnancy termination.13 The following day, before uterine evacuation, a cervical biopsy was obtained for electron microscopy studies and immunohistochemistry to assess the presence of enzymes involved in collagen degradation, including matrix metalloproteinase 1 (MMP-1) and matrix metalloproteinase 9 (MMP-9). Electron microscopy demonstrated that pretreatment with misoprostol resulted in a pronounced splitting and disorganization of collagen fibers.13 Compared with misoprostol treatment, IMN produced less splitting and disorganization of collagen fibers, and in the no treatment group, no marked changes in the collagen framework were observed.
Compared with no treatment, misoprostol and IMN pretreatment were associated with marked increases in MMP-1 and MMP-9 as assessed by immunohistochemistry. Misoprostol pretreatment also resulted in a significant increase in interleukin-8 concentration compared with IMN pretreatment and no treatment (8.8 vs 2.7 vs 2.4 pg/mg tissue), respectively.13 Other investigators have also reported that misoprostol increased cervical leukocyte influx and collagen disrupting enzymes MMP-8 and MMP-9.14,15
An open-label clinical trial compared the efficacy of misoprostol versus Foley catheter for labor induction at term in 1,859 patients ≥ 37 weeks’ gestation with a Bishop score <6.16 Patients were randomly allocated to misoprostol (50 µg orally every 4 hours up to 3 times in 24 hours) versus placement of a 16 F or 18 F Foley catheter introduced through the cervix, filled with 30 mL of sodium chloride or water. The investigators reported that oral misoprostol and Foley catheter cervical ripening had similar safety and effectiveness for cervical ripening as a prelude to induction of labor, including no statistically significant differences in 5-minute Apgar score <7, umbilical cord artery pH ≤ 7.05, postpartum hemorrhage, or cesarean birth rate.16
Bottom line
Misoprostol and oxytocin are commonly prescribed in obstetric practice for cervical ripening and induction of labor, respectively. The dose and route of administration of misoprostol influences the effect on the uterus. For cervical ripening, where rapid onset and offset may help to reduce the risk of uterine tachysystole and worrisome fetal heart rate changes, low-dose (50 µg) oral administration of misoprostol may be a preferred dose and route. For the treatment of miscarriage and fetal demise, to stimulate sustained uterine contractions over many hours, buccal and vaginal administration of misoprostol are preferred. Rectal administration is generally inferior to buccal and vaginal administration for stimulating sustained uterine contractions and its uses should be limited. ●
Common side effects of misoprostol are abdominal cramping, diarrhea, nausea, vomiting, headache, and fever. Elevated temperature following misoprostol administration is a concerning side effect that may require further investigation to rule out an infection, especially if the elevated temperature persists for > 4 hours. The preoptic area of the anterior hypothalamus (POAH) plays a major role in thermoregulation. When an infection causes an increase in endogenous pyrogens, including interleukin-1β, interleukin-6 and tumor necrosis factor, prostaglandins are generated in the region of the POAH, increasing the thermoregulatory set point, triggering cutaneous vasoconstriction and shivering and non-shivering thermogenesis.1 Misoprostol, especially at doses >400 µg commonly causes both patient-reported chills and temperature elevation >38° C.
In a study comparing misoprostol and oxytocin for the management of the third stage of labor, 597 patients were randomly allocated to receive oxytocin 10 units by intramuscular injection or misoprostol 400 µg or 600 µg by the oral route.2 Patient-reported shivering occurred in 13%, 19%, and 28% of patients receiving oxytocin, misoprostol 400 µg and misoprostol 800 µg, respectively. A recorded temperature >38° C occurred within 1 hour of medication administration in approximately 3%, 2%, and 7.5% of patients receiving oxytocin, misoprostol 400 µg, and misoprostol 800 µg, respectively. In another study, 453 patients scheduled for a cesarean birth were randomly allocated to receive 1 of 3 doses of rectal misoprostol 200 μg, 400 μg, or 600 μg before incision. Fever was detected in 2.6%, 9.9%, and 5.1% of the patients receiving misoprostol 200 μg, 400 μg, or 600 μg, respectively.3
References
1. Aronoff DM, Neilson EG. Antipyretics: mechanisms of action and clinical use in fever suppression. Am J Med. 2001;111:304-315. doi: 10.1016/s0002-9343(01)00834-8.
2. Lumbiganon P, Hofmeyr J, Gumezoglu AM, et al. Misoprostol dose-related shivering and pyrexia in the third stage of labor. WHO Collaborative Trial of Misoprostol in the Management of the Third Stage of Labor. Br J Obstet Gynaecol. 1999;106:304-308. doi: 10.1111/j.1471-0528.1999.tb08266.x.
3. Sweed M, El-Said M, Abou-Gamrah AA, et al. Comparison between 200, 400 and 600 microgram rectal misoprostol before cesarean section: a randomized clinical trial. J Obstet Gynaecol Res. 2019;45:585-591. doi: 10.1111 /jog.13883.
Oxytocin and prostaglandins are critically important regulators of uterine contraction. Obstetrician-gynecologists commonly prescribe oxytocin and prostaglandin agonists (misoprostol, dinoprostone) to stimulate uterine contraction for the induction of labor, prevention and treatment of postpartum hemorrhage, and treatment of miscarriage and fetal demise. The focus of this editorial is the clinical pharmacology of misoprostol.
Misoprostol is approved by the US Food and Drug Administration (FDA) for the prevention and treatment of nonsteroidal anti-inflammatory drug–induced gastric ulcers and for patients at high risk for gastric ulcers, including those with a history of gastric ulcers. The approved misoprostol route and dose for this indication is oral administration of 200 µg four times daily with food.1 Recent food intake and antacid use reduces the absorption of orally administered misoprostol. There are no FDA-approved indications for the use of misoprostol as a single agent in obstetrics and gynecology. The FDA has approved the combination of mifepristone and misoprostol for medication abortion in the first trimester. In contrast to misoprostol, PGE2 (dinoprostone) is approved by the FDA as a vaginal insert containing 10 mg of dinoprostone for the initiation and/or continuation of cervical ripening in patients at or near term in whom there is a medical or obstetric indication for induction of labor (Cervidil; Ferring Pharmaceuticals Inc, Parsippany, New Jersey).2
Pharmacology of misoprostol
Misoprostol is a prostaglandin E1 (PGE1) agonist analogue. Prostaglandin E1 (alprostadil) is rapidly metabolized, has a half-life in the range of minutes and is not orally active, requiring administration by intravenous infusion or injection. It is indicated to maintain a patent ductus arteriosus in newborns with ductal-dependent circulation and to treat erectile dysfunction.3 In contrast to PGE1, misoprostol has a methyl ester group at carbon-1 (C-1) that increases potency and duration of action. Misoprostol also has no hydroxyl group at C-15, replacing that moiety with the addition of both a methyl- and hydroxyl- group at C-16 (FIGURE). These molecular changes improve oral activity and increase duration of action.4 Pure misoprostol is a viscous oil. It is formulated into tables by dispersing the oil on hydroxypropyl methyl cellulose before compounding into tablets. Unlike naturally occurring prostaglandins (PGE1), misoprostol tablets are stabile at room temperature for years.4
Following absorption, the methyl ester at C-1 is enzymatically cleaved, yielding misoprostol acid, the active drug.4 Misoprostol binds to the E prostanoid receptor 3 (EP-3).5 Activation of myometrial EP-3 receptor induces an increase in intracellular phosphoinositol turnover and calcium mobilization, resulting in an increase in intracellular-free calcium, triggering actin-myosin contractility.6 The increase in free calcium is propagated cell-to-cell through gap junctions that link the myometrial cells to facilitate the generation of a coordinated contraction.

Misoprostol: Various routes of administration are not equal
Misoprostol can be given by an oral, buccal, vaginal, or rectal route of administration. To study the effect of the route of administration on uterine tone and contractility, investigators randomly assigned patients at 8 to 11 weeks’ gestation to receive misoprostol 400 µg as a single dose by the oral or vaginal route. Uterine tone and contractility were measured using an intrauterine pressure transducer. Compared to vaginal administration, oral administration of misprostol was associated with rapid attainment of peak plasma level at 30 minutes, followed by a decline in concentration by 60 minutes. This rapid onset and rapid offset of plasma concentration was paralleled by the onset of uterine tone within 8 minutes, but surprisingly no sustained uterine contractions.7 By contrast, following vaginal administration of misoprostol, serum levels rose slowly and peaked in 1 to 2 hours. Uterine tone increased within 21 minutes, and sustained uterine contractions were recorded for 4 hours.7 The rapid rise and fall in plasma misoprostol following oral administration and the more sustained plasma misoprostol concentration over 4 hours has been previously reported.8 In a second study involving patients 8 to 11 weeks’ gestation, the effect of a single dose of misoprostol 400 µg by an oral or vaginal route on uterine contractility was compared using an intrauterine pressure transducer.9 Confirming previous results, the time from misoprostol administration to increased uterine tone was more rapid with oral than with vaginal administration (8 min vs 19 min). Over the course of 4 hours, uterine contraction activity was greater with vaginal than with oral administration (454 vs 166 Montevideo units).9
Both studies reported that oral administration of misoprostol resulted in more rapid onset and offset of action than vaginal administration. Oral administration of a single dose of misoprostol 400 µg did not result in sustained uterine contractions in most patients in the first trimester. Vaginal administration produced a slower onset of increased uterine tone but sustained uterine contractions over 4 hours. Compared with vaginal administration of misoprostol, the rapid onset and offset of action of oral misoprostol may reduce the rate of tachysystole and changes in fetal heart rate observed with vaginal administration.10
An important finding is that buccal and vaginal administration of misoprostol have similar effects on uterine tone in the first trimester.11 To study the effect of buccal and vaginal administration of misoprostol on uterine tone, patients 6 to 13 weeks’ gestation were randomly allocated to receive a single dose of misoprostol 400 µg by a buccal or vaginal route.11 Uterine activity over 5 hours following administration was assessed using an intrauterine pressure transducer. Uterine tone 20 to 30 minutes after buccal or vaginal administration of misoprostol (400 µg) was 27 and 28 mm Hg, respectively. Peak uterine tone, as measured by an intrauterine pressure transducer, for buccal and vaginal administration of misoprostol was 49 mm Hg and 54 mm Hg, respectively. Total Alexandria units (AU) over 5 hours following buccal or vaginal administration was 6,537 AU and 6,090 AU, respectively.11
An AU is calculated as the average amplitude of the contractions (mm Hg) multiplied by the average duration of the contractions (min) multiplied by average frequency of contraction over 10 minutes.12 By contrast, a Montevideo unit does not include an assessment of contraction duration and is calculated as average amplitude of contractions (mm Hg) multiplied by frequency of uterine contractions over 10 minutes.12
In contrast to buccal or vaginal administration, rectal administration of misoprostol resulted in much lower peak uterine tone and contractility as measured by a pressure transducer. Uterine tone 20 to 30 minutes after vaginal and rectal administration of misoprostol (400 µg) was 28 and 19 mm Hg, respectively.11 Peak uterine tone, as measured by an intrauterine pressure transducer, for vaginal and rectal administration of misoprostol was 54 and 31 mm Hg, respectively. AUs over 5 hours following vaginal and rectal administration was 6,090 AU and 2,768 AU, respectively.11 Compared with buccal and vaginal administration of misoprostol, rectal administration produced less sustained uterine contractions in the first trimester of pregnancy. To achieve maximal sustained uterine contractions, buccal and vaginal routes of administration are superior to oral and rectal administration.
Continue to: Misoprostol and cervical ripening...
Misoprostol and cervical ripening
Misoprostol is commonly used to soften and ripen the cervix. Some of the cervical ripening effects of misoprostol are likely due to increased uterine tone. In addition, misoprostol may have a direct effect on the collagen structure of the cervix. To study the effect of misoprostol on the cervix, pregnant patients in the first trimester were randomly assigned to receive misoprostol 200 µg by vaginal self-administration, isosorbide mononitrate (IMN) 40 mg by vaginal self-administration or no treatment the evening prior to pregnancy termination.13 The following day, before uterine evacuation, a cervical biopsy was obtained for electron microscopy studies and immunohistochemistry to assess the presence of enzymes involved in collagen degradation, including matrix metalloproteinase 1 (MMP-1) and matrix metalloproteinase 9 (MMP-9). Electron microscopy demonstrated that pretreatment with misoprostol resulted in a pronounced splitting and disorganization of collagen fibers.13 Compared with misoprostol treatment, IMN produced less splitting and disorganization of collagen fibers, and in the no treatment group, no marked changes in the collagen framework were observed.
Compared with no treatment, misoprostol and IMN pretreatment were associated with marked increases in MMP-1 and MMP-9 as assessed by immunohistochemistry. Misoprostol pretreatment also resulted in a significant increase in interleukin-8 concentration compared with IMN pretreatment and no treatment (8.8 vs 2.7 vs 2.4 pg/mg tissue), respectively.13 Other investigators have also reported that misoprostol increased cervical leukocyte influx and collagen disrupting enzymes MMP-8 and MMP-9.14,15
An open-label clinical trial compared the efficacy of misoprostol versus Foley catheter for labor induction at term in 1,859 patients ≥ 37 weeks’ gestation with a Bishop score <6.16 Patients were randomly allocated to misoprostol (50 µg orally every 4 hours up to 3 times in 24 hours) versus placement of a 16 F or 18 F Foley catheter introduced through the cervix, filled with 30 mL of sodium chloride or water. The investigators reported that oral misoprostol and Foley catheter cervical ripening had similar safety and effectiveness for cervical ripening as a prelude to induction of labor, including no statistically significant differences in 5-minute Apgar score <7, umbilical cord artery pH ≤ 7.05, postpartum hemorrhage, or cesarean birth rate.16
Bottom line
Misoprostol and oxytocin are commonly prescribed in obstetric practice for cervical ripening and induction of labor, respectively. The dose and route of administration of misoprostol influences the effect on the uterus. For cervical ripening, where rapid onset and offset may help to reduce the risk of uterine tachysystole and worrisome fetal heart rate changes, low-dose (50 µg) oral administration of misoprostol may be a preferred dose and route. For the treatment of miscarriage and fetal demise, to stimulate sustained uterine contractions over many hours, buccal and vaginal administration of misoprostol are preferred. Rectal administration is generally inferior to buccal and vaginal administration for stimulating sustained uterine contractions and its uses should be limited. ●
Common side effects of misoprostol are abdominal cramping, diarrhea, nausea, vomiting, headache, and fever. Elevated temperature following misoprostol administration is a concerning side effect that may require further investigation to rule out an infection, especially if the elevated temperature persists for > 4 hours. The preoptic area of the anterior hypothalamus (POAH) plays a major role in thermoregulation. When an infection causes an increase in endogenous pyrogens, including interleukin-1β, interleukin-6 and tumor necrosis factor, prostaglandins are generated in the region of the POAH, increasing the thermoregulatory set point, triggering cutaneous vasoconstriction and shivering and non-shivering thermogenesis.1 Misoprostol, especially at doses >400 µg commonly causes both patient-reported chills and temperature elevation >38° C.
In a study comparing misoprostol and oxytocin for the management of the third stage of labor, 597 patients were randomly allocated to receive oxytocin 10 units by intramuscular injection or misoprostol 400 µg or 600 µg by the oral route.2 Patient-reported shivering occurred in 13%, 19%, and 28% of patients receiving oxytocin, misoprostol 400 µg and misoprostol 800 µg, respectively. A recorded temperature >38° C occurred within 1 hour of medication administration in approximately 3%, 2%, and 7.5% of patients receiving oxytocin, misoprostol 400 µg, and misoprostol 800 µg, respectively. In another study, 453 patients scheduled for a cesarean birth were randomly allocated to receive 1 of 3 doses of rectal misoprostol 200 μg, 400 μg, or 600 μg before incision. Fever was detected in 2.6%, 9.9%, and 5.1% of the patients receiving misoprostol 200 μg, 400 μg, or 600 μg, respectively.3
References
1. Aronoff DM, Neilson EG. Antipyretics: mechanisms of action and clinical use in fever suppression. Am J Med. 2001;111:304-315. doi: 10.1016/s0002-9343(01)00834-8.
2. Lumbiganon P, Hofmeyr J, Gumezoglu AM, et al. Misoprostol dose-related shivering and pyrexia in the third stage of labor. WHO Collaborative Trial of Misoprostol in the Management of the Third Stage of Labor. Br J Obstet Gynaecol. 1999;106:304-308. doi: 10.1111/j.1471-0528.1999.tb08266.x.
3. Sweed M, El-Said M, Abou-Gamrah AA, et al. Comparison between 200, 400 and 600 microgram rectal misoprostol before cesarean section: a randomized clinical trial. J Obstet Gynaecol Res. 2019;45:585-591. doi: 10.1111 /jog.13883.
- Cytotec [package insert]. Chicago, IL: GD Searle & Co. https://www.accessdata.fda.gov/drugsatfda_docs/label/2002/19268slr037.pdf. Accessed June 20, 2022.
- Cervidil [package insert]. St Louis, MO: Forrest Pharmaceuticals Inc.; May 2006. Accessed June 20, 2022.
- Caverject [package insert]. New York, NY: Pfizer Inc.; March 2014. Accessed June 20, 2022.
- Collins PW. Misoprostol: discovery, development and clinical applications. Med Res Rev. 1990;10:149-172. doi: 10.1002/med.2610100202.
- Audit M, White KI, Breton B, et al. Crystal structure of misoprostol bound to the labor inducer prostaglandin E2 receptor. Nat Chem Biol. 2019;15:11-17. doi: 10.1038/s41589-018-0160-y.
- Pallliser KH, Hirst JJ, Ooi G, et al. Prostaglandin E and F receptor expression and myometrial sensitivity in labor onset in the sheep. Biol Reprod. 2005;72:937-943. doi: 10.1095/biolreprod.104.035311.
- Gemzell-Danilesson K, Marions L, Rodriguez A, et al. Comparison between oral and vaginal administration of misoprostol on uterine contractility. Obstet Gynecol. 1999;93:275-280. doi: 10.1016/s0029-7844(98)00436-0.
- Zieman M, Fong SK, Benowitz NL, et al. Absorption kinetics of misoprostol with oral or vaginal administration. Obstet Gynecol. 1997;90:88-92. doi: 10.1016/S0029-7844(97)00111-7.
- Aronsson A, Bygdeman M, Gemzell-Danielsson K. Effects of misoprostol on uterine contractility following different routes of administration. Hum Reprod. 2004;19:81-84. doi: 10.1093/humrep/deh005.
- Young DC, Delaney T, Armson BA, et al. Oral misoprostol, low dose vaginal misoprostol and vaginal dinoprostone for labor induction: randomized controlled trial. PLOS One. 2020;15:e0227245. doi: 10.1371/journal.pone.0227245.
- Meckstroth KR, Whitaker AK, Bertisch S, et al. Misoprostol administered by epithelial routes. Drug absorption and uterine response. Obstet Gynecol. 2006;108:582-590. doi: 10.1097/01.AOG.0000230398.32794.9d.
- el-Sahwi S, Gaafar AA, Toppozada HK. A new unit for evaluation of uterine activity. Am J Obstet Gynecol. 1967;98:900-903. doi: 10.1016/0002-9378(67)90074-9.
- Vukas N, Ekerhovd E, Abrahamsson G, et al. Cervical priming in the first trimester: morphological and biochemical effects of misoprostol and isosorbide mononitrate. Acta Obstet Gyecol. 2009;88:43-51. doi: 10.1080/00016340802585440.
- Aronsson A, Ulfgren AK, Stabi B, et al. The effect of orally and vaginally administered misoprostol on inflammatory mediators and cervical ripening during early pregnancy. Contraception. 2005;72:33-39. doi: 10.1016/j.contraception.2005.02.012.
- Denison FC, Riley SC, Elliott CL, et al. The effect of mifepristone administration on leukocyte populations, matrix metalloproteinases and inflammatory mediators in the first trimester cervix. Mol Hum Reprod. 2000;6:541-548. doi: 10.1093/molehr/6.6.541.
- ten Eikelder MLG, Rengerink KO, Jozwiak M, et al. Induction of labour at term with oral misoprostol versus a Foley catheter (PROBAAT-II): a multicentre randomised controlled non-inferiority trial. Lancet. 2016;387:1619-1628. doi: 10.1016 /S0140-6736(16)00084-2.
- Cytotec [package insert]. Chicago, IL: GD Searle & Co. https://www.accessdata.fda.gov/drugsatfda_docs/label/2002/19268slr037.pdf. Accessed June 20, 2022.
- Cervidil [package insert]. St Louis, MO: Forrest Pharmaceuticals Inc.; May 2006. Accessed June 20, 2022.
- Caverject [package insert]. New York, NY: Pfizer Inc.; March 2014. Accessed June 20, 2022.
- Collins PW. Misoprostol: discovery, development and clinical applications. Med Res Rev. 1990;10:149-172. doi: 10.1002/med.2610100202.
- Audit M, White KI, Breton B, et al. Crystal structure of misoprostol bound to the labor inducer prostaglandin E2 receptor. Nat Chem Biol. 2019;15:11-17. doi: 10.1038/s41589-018-0160-y.
- Pallliser KH, Hirst JJ, Ooi G, et al. Prostaglandin E and F receptor expression and myometrial sensitivity in labor onset in the sheep. Biol Reprod. 2005;72:937-943. doi: 10.1095/biolreprod.104.035311.
- Gemzell-Danilesson K, Marions L, Rodriguez A, et al. Comparison between oral and vaginal administration of misoprostol on uterine contractility. Obstet Gynecol. 1999;93:275-280. doi: 10.1016/s0029-7844(98)00436-0.
- Zieman M, Fong SK, Benowitz NL, et al. Absorption kinetics of misoprostol with oral or vaginal administration. Obstet Gynecol. 1997;90:88-92. doi: 10.1016/S0029-7844(97)00111-7.
- Aronsson A, Bygdeman M, Gemzell-Danielsson K. Effects of misoprostol on uterine contractility following different routes of administration. Hum Reprod. 2004;19:81-84. doi: 10.1093/humrep/deh005.
- Young DC, Delaney T, Armson BA, et al. Oral misoprostol, low dose vaginal misoprostol and vaginal dinoprostone for labor induction: randomized controlled trial. PLOS One. 2020;15:e0227245. doi: 10.1371/journal.pone.0227245.
- Meckstroth KR, Whitaker AK, Bertisch S, et al. Misoprostol administered by epithelial routes. Drug absorption and uterine response. Obstet Gynecol. 2006;108:582-590. doi: 10.1097/01.AOG.0000230398.32794.9d.
- el-Sahwi S, Gaafar AA, Toppozada HK. A new unit for evaluation of uterine activity. Am J Obstet Gynecol. 1967;98:900-903. doi: 10.1016/0002-9378(67)90074-9.
- Vukas N, Ekerhovd E, Abrahamsson G, et al. Cervical priming in the first trimester: morphological and biochemical effects of misoprostol and isosorbide mononitrate. Acta Obstet Gyecol. 2009;88:43-51. doi: 10.1080/00016340802585440.
- Aronsson A, Ulfgren AK, Stabi B, et al. The effect of orally and vaginally administered misoprostol on inflammatory mediators and cervical ripening during early pregnancy. Contraception. 2005;72:33-39. doi: 10.1016/j.contraception.2005.02.012.
- Denison FC, Riley SC, Elliott CL, et al. The effect of mifepristone administration on leukocyte populations, matrix metalloproteinases and inflammatory mediators in the first trimester cervix. Mol Hum Reprod. 2000;6:541-548. doi: 10.1093/molehr/6.6.541.
- ten Eikelder MLG, Rengerink KO, Jozwiak M, et al. Induction of labour at term with oral misoprostol versus a Foley catheter (PROBAAT-II): a multicentre randomised controlled non-inferiority trial. Lancet. 2016;387:1619-1628. doi: 10.1016 /S0140-6736(16)00084-2.