User login
First exposure to general anesthesia not a risk factor for atopic dermatitis in the pediatric population
Key clinical point: The first exposure of pediatric individuals to general anesthesia (GA) is not associated with an increased or decreased risk of developing atopic dermatitis (AD).
Major finding: Multivariate analysis revealed that individuals who were exposed vs not exposed to GA did not have an increased or decreased risk of developing AD (adjusted hazard ratio 1.03; P = .701).
Study details: This retrospective cohort study included pediatric individuals aged ≤18 years who were (n = 7,681) or were not (n = 38,405; control individuals) exposed to GA.
Disclosures: This study was funded by a 2020 Amorepacific (South Korea) grant. The authors declared no conflicts of interest.
Source: Kim DC et al. No association between first exposure to general anaesthesia and atopic dermatitis in the paediatric population. Acta Derm Venereol. 2022;102:adv00813 (Nov 14). Doi: 10.2340/actadv.v102.2738
Key clinical point: The first exposure of pediatric individuals to general anesthesia (GA) is not associated with an increased or decreased risk of developing atopic dermatitis (AD).
Major finding: Multivariate analysis revealed that individuals who were exposed vs not exposed to GA did not have an increased or decreased risk of developing AD (adjusted hazard ratio 1.03; P = .701).
Study details: This retrospective cohort study included pediatric individuals aged ≤18 years who were (n = 7,681) or were not (n = 38,405; control individuals) exposed to GA.
Disclosures: This study was funded by a 2020 Amorepacific (South Korea) grant. The authors declared no conflicts of interest.
Source: Kim DC et al. No association between first exposure to general anaesthesia and atopic dermatitis in the paediatric population. Acta Derm Venereol. 2022;102:adv00813 (Nov 14). Doi: 10.2340/actadv.v102.2738
Key clinical point: The first exposure of pediatric individuals to general anesthesia (GA) is not associated with an increased or decreased risk of developing atopic dermatitis (AD).
Major finding: Multivariate analysis revealed that individuals who were exposed vs not exposed to GA did not have an increased or decreased risk of developing AD (adjusted hazard ratio 1.03; P = .701).
Study details: This retrospective cohort study included pediatric individuals aged ≤18 years who were (n = 7,681) or were not (n = 38,405; control individuals) exposed to GA.
Disclosures: This study was funded by a 2020 Amorepacific (South Korea) grant. The authors declared no conflicts of interest.
Source: Kim DC et al. No association between first exposure to general anaesthesia and atopic dermatitis in the paediatric population. Acta Derm Venereol. 2022;102:adv00813 (Nov 14). Doi: 10.2340/actadv.v102.2738
High treatment flexibility with baricitinib in moderate-to-severe atopic dermatitis
Key clinical point: Downtitrated baricitinib treatment was efficacious in most patients with moderate-to-severe atopic dermatitis (AD) through 16 weeks; clinically relevant efficacy was observed in most patients who were readministered the original dose due to downtitration or treatment withdrawal-related efficacy loss.
Major finding: In the 4-mg and 2-mg cohorts, 61% and 71% of patients maintained a validated Investigator’s Global Assessment for AD (vIGA-AD) score of 0/1/2 at downtitration week 16 and 80%/85%/88% and 90%/56%/86% of patients who switched to original dose in the continuous dosing/downtitration/treatment withdrawal group re-achieved vIGA-AD 0/1/2, respectively.
Study details: This BREEZE-AD3 trial substudy included 526 patients with moderate-to-severe AD treated with 4/2 mg baricitinib at trial entry who achieved vIGA-AD 0/1/2 at week 52, with each cohort being re-assigned to continuous dosing, downtitration, or treatment withdrawal.
Disclosures: This study was sponsored by Eli Lilly and Company under license from Incyte Corporation. Some authors reported ties with various sources, including Eli Lilly and Incyte. Three authors declared being current or former employees and shareholders of Eli Lilly.
Source: Reich K et al. Efficacy of downtitration or treatment withdrawal compared to continuous dosing after successful treatment with baricitinib in patients with moderate-to-severe atopic dermatitis in a randomised substudy from the long-term extension study, BREEZE-AD3. Br J Dermatol. 2022 (Nov 17). Doi: 10.1093/bjd/ljac057
Key clinical point: Downtitrated baricitinib treatment was efficacious in most patients with moderate-to-severe atopic dermatitis (AD) through 16 weeks; clinically relevant efficacy was observed in most patients who were readministered the original dose due to downtitration or treatment withdrawal-related efficacy loss.
Major finding: In the 4-mg and 2-mg cohorts, 61% and 71% of patients maintained a validated Investigator’s Global Assessment for AD (vIGA-AD) score of 0/1/2 at downtitration week 16 and 80%/85%/88% and 90%/56%/86% of patients who switched to original dose in the continuous dosing/downtitration/treatment withdrawal group re-achieved vIGA-AD 0/1/2, respectively.
Study details: This BREEZE-AD3 trial substudy included 526 patients with moderate-to-severe AD treated with 4/2 mg baricitinib at trial entry who achieved vIGA-AD 0/1/2 at week 52, with each cohort being re-assigned to continuous dosing, downtitration, or treatment withdrawal.
Disclosures: This study was sponsored by Eli Lilly and Company under license from Incyte Corporation. Some authors reported ties with various sources, including Eli Lilly and Incyte. Three authors declared being current or former employees and shareholders of Eli Lilly.
Source: Reich K et al. Efficacy of downtitration or treatment withdrawal compared to continuous dosing after successful treatment with baricitinib in patients with moderate-to-severe atopic dermatitis in a randomised substudy from the long-term extension study, BREEZE-AD3. Br J Dermatol. 2022 (Nov 17). Doi: 10.1093/bjd/ljac057
Key clinical point: Downtitrated baricitinib treatment was efficacious in most patients with moderate-to-severe atopic dermatitis (AD) through 16 weeks; clinically relevant efficacy was observed in most patients who were readministered the original dose due to downtitration or treatment withdrawal-related efficacy loss.
Major finding: In the 4-mg and 2-mg cohorts, 61% and 71% of patients maintained a validated Investigator’s Global Assessment for AD (vIGA-AD) score of 0/1/2 at downtitration week 16 and 80%/85%/88% and 90%/56%/86% of patients who switched to original dose in the continuous dosing/downtitration/treatment withdrawal group re-achieved vIGA-AD 0/1/2, respectively.
Study details: This BREEZE-AD3 trial substudy included 526 patients with moderate-to-severe AD treated with 4/2 mg baricitinib at trial entry who achieved vIGA-AD 0/1/2 at week 52, with each cohort being re-assigned to continuous dosing, downtitration, or treatment withdrawal.
Disclosures: This study was sponsored by Eli Lilly and Company under license from Incyte Corporation. Some authors reported ties with various sources, including Eli Lilly and Incyte. Three authors declared being current or former employees and shareholders of Eli Lilly.
Source: Reich K et al. Efficacy of downtitration or treatment withdrawal compared to continuous dosing after successful treatment with baricitinib in patients with moderate-to-severe atopic dermatitis in a randomised substudy from the long-term extension study, BREEZE-AD3. Br J Dermatol. 2022 (Nov 17). Doi: 10.1093/bjd/ljac057
Dupilumab is clinically effective and safe for treating pediatric atopic dermatitis in daily practice
Key clinical point: Dupilumab decreases disease severity, disease-associated symptoms, and severity-associated serum biomarker levels in pediatric patients with atopic dermatitis (AD) in daily practice.
Major finding: At week 28, 49.2% of patients achieved Eczema Area and Severity Index-75; 84.7%, 45.3%, and 77.4% achieved a ≥4-point improvement in the Patient-Oriented Eczema Measure, Numeric Rating Scale (NRS)-pruritus, and NRS-pain scores, respectively; 36.1% scored clear or almost clear on the Investigator’s Global Assessment. The levels of severity-associated markers soluble IL-2-receptor alpha (P < .05), periostin (P < .05), thymus- and activation-regulated chemokine (P < .005), and pulmonary- and activation-regulated chemokine (P < .005) were significantly reduced at week 4. Conjunctivitis (16.4%) and headache (6.6%) were the most common side effects.
Study details: This multicenter prospective observational study included 61 patients (children ≥6 to <12 years; adolescents ≥12 to <18 years) from the BioDay registry with moderate-to-severe AD who received dupilumab for 28 weeks.
Disclosures: The BioDay registry is sponsored by Sanofi/Regeneron and others. Some authors reported ties with various sources, including Sanofi.
Source: Kamphuis E, Boesjes CM et al. Dupilumab in daily practice for the treatment of pediatric atopic dermatitis: 28-week clinical and biomarker results from the BioDay registry. Pediatr Allergy Immunol. 2022;13(12):e13887 (Dec 5). Doi: 10.1111/pai.13887
Key clinical point: Dupilumab decreases disease severity, disease-associated symptoms, and severity-associated serum biomarker levels in pediatric patients with atopic dermatitis (AD) in daily practice.
Major finding: At week 28, 49.2% of patients achieved Eczema Area and Severity Index-75; 84.7%, 45.3%, and 77.4% achieved a ≥4-point improvement in the Patient-Oriented Eczema Measure, Numeric Rating Scale (NRS)-pruritus, and NRS-pain scores, respectively; 36.1% scored clear or almost clear on the Investigator’s Global Assessment. The levels of severity-associated markers soluble IL-2-receptor alpha (P < .05), periostin (P < .05), thymus- and activation-regulated chemokine (P < .005), and pulmonary- and activation-regulated chemokine (P < .005) were significantly reduced at week 4. Conjunctivitis (16.4%) and headache (6.6%) were the most common side effects.
Study details: This multicenter prospective observational study included 61 patients (children ≥6 to <12 years; adolescents ≥12 to <18 years) from the BioDay registry with moderate-to-severe AD who received dupilumab for 28 weeks.
Disclosures: The BioDay registry is sponsored by Sanofi/Regeneron and others. Some authors reported ties with various sources, including Sanofi.
Source: Kamphuis E, Boesjes CM et al. Dupilumab in daily practice for the treatment of pediatric atopic dermatitis: 28-week clinical and biomarker results from the BioDay registry. Pediatr Allergy Immunol. 2022;13(12):e13887 (Dec 5). Doi: 10.1111/pai.13887
Key clinical point: Dupilumab decreases disease severity, disease-associated symptoms, and severity-associated serum biomarker levels in pediatric patients with atopic dermatitis (AD) in daily practice.
Major finding: At week 28, 49.2% of patients achieved Eczema Area and Severity Index-75; 84.7%, 45.3%, and 77.4% achieved a ≥4-point improvement in the Patient-Oriented Eczema Measure, Numeric Rating Scale (NRS)-pruritus, and NRS-pain scores, respectively; 36.1% scored clear or almost clear on the Investigator’s Global Assessment. The levels of severity-associated markers soluble IL-2-receptor alpha (P < .05), periostin (P < .05), thymus- and activation-regulated chemokine (P < .005), and pulmonary- and activation-regulated chemokine (P < .005) were significantly reduced at week 4. Conjunctivitis (16.4%) and headache (6.6%) were the most common side effects.
Study details: This multicenter prospective observational study included 61 patients (children ≥6 to <12 years; adolescents ≥12 to <18 years) from the BioDay registry with moderate-to-severe AD who received dupilumab for 28 weeks.
Disclosures: The BioDay registry is sponsored by Sanofi/Regeneron and others. Some authors reported ties with various sources, including Sanofi.
Source: Kamphuis E, Boesjes CM et al. Dupilumab in daily practice for the treatment of pediatric atopic dermatitis: 28-week clinical and biomarker results from the BioDay registry. Pediatr Allergy Immunol. 2022;13(12):e13887 (Dec 5). Doi: 10.1111/pai.13887
Early emollient application: An effective strategy for atopic dermatitis prevention in infants
Key clinical point: Early application of emollients can effectively prevent atopic dermatitis (AD) in infants, with emollient emulsion seeming an optimal treatment option in infancy compared with creams or mixed emollients.
Major finding: The incidence of AD was significantly lower in high-risk infants receiving early emollients vs standard care (risk ratio 0.64; 95% CI 0.47-0.88), with surface under the cumulative ranking curve analysis revealing emollient emulsion (82.6%) as the optimal treatment for AD prevention in infants, followed by mixed emollient (77.4%) and emollient cream (21.9%).
Study details: This was a systematic review and network meta-analysis of 11 randomized controlled trials including 3483 infants without AD who received either prophylactic emollients (cream, emulsion, or mixed types) or standard care.
Disclosures: This study was supported by the Key Research and Development Project of Xinjiang Uygur Autonomous Region, China. The authors declared no conflicts of interest.
Source: Liang J, Hu F et al. Systematic review and network meta‐analysis of different types of emollient for the prevention of atopic dermatitis in infants. J Eur Acad Dermatol Venereol. 2022 (Nov 23). Doi: 10.1111/jdv.18688
Key clinical point: Early application of emollients can effectively prevent atopic dermatitis (AD) in infants, with emollient emulsion seeming an optimal treatment option in infancy compared with creams or mixed emollients.
Major finding: The incidence of AD was significantly lower in high-risk infants receiving early emollients vs standard care (risk ratio 0.64; 95% CI 0.47-0.88), with surface under the cumulative ranking curve analysis revealing emollient emulsion (82.6%) as the optimal treatment for AD prevention in infants, followed by mixed emollient (77.4%) and emollient cream (21.9%).
Study details: This was a systematic review and network meta-analysis of 11 randomized controlled trials including 3483 infants without AD who received either prophylactic emollients (cream, emulsion, or mixed types) or standard care.
Disclosures: This study was supported by the Key Research and Development Project of Xinjiang Uygur Autonomous Region, China. The authors declared no conflicts of interest.
Source: Liang J, Hu F et al. Systematic review and network meta‐analysis of different types of emollient for the prevention of atopic dermatitis in infants. J Eur Acad Dermatol Venereol. 2022 (Nov 23). Doi: 10.1111/jdv.18688
Key clinical point: Early application of emollients can effectively prevent atopic dermatitis (AD) in infants, with emollient emulsion seeming an optimal treatment option in infancy compared with creams or mixed emollients.
Major finding: The incidence of AD was significantly lower in high-risk infants receiving early emollients vs standard care (risk ratio 0.64; 95% CI 0.47-0.88), with surface under the cumulative ranking curve analysis revealing emollient emulsion (82.6%) as the optimal treatment for AD prevention in infants, followed by mixed emollient (77.4%) and emollient cream (21.9%).
Study details: This was a systematic review and network meta-analysis of 11 randomized controlled trials including 3483 infants without AD who received either prophylactic emollients (cream, emulsion, or mixed types) or standard care.
Disclosures: This study was supported by the Key Research and Development Project of Xinjiang Uygur Autonomous Region, China. The authors declared no conflicts of interest.
Source: Liang J, Hu F et al. Systematic review and network meta‐analysis of different types of emollient for the prevention of atopic dermatitis in infants. J Eur Acad Dermatol Venereol. 2022 (Nov 23). Doi: 10.1111/jdv.18688
Phase 3 studies confirm long-term disease control with ruxolitinib cream in atopic dermatitis
Key clinical point: Ruxolitinib cream demonstrated effective disease control and was well tolerated in patients with atopic dermatitis (AD) during 44 weeks of as-needed treatment.
Major finding: At week 52, 74.1%-77.8% of patients had an Investigator’s Global Assessment score of 0/1, with the mean affected body surface area being 1.4%-1.8%. Treatment-related adverse events were reported in 8.7%/7.4% of patients on 0.75%/1.5% ruxolitinib and in 2.0%/6.1% of those who switched from vehicle to 0.75%/1.5% ruxolitinib, respectively.
Study details: This study analyzed pooled data from two phase 3 studies, TRuE-AD1 and TRuE-AD2, including 1249 patients aged ≥12 years with AD who were randomly assigned to receive 0.75% or 1.5% ruxolitinib cream or vehicle for 8 weeks; thereafter, the vehicle group patients were re-assigned to receive either strength ruxolitinib cream for 44 weeks.
Disclosures: This study was funded by Incyte Corporation, U.S. Some authors reported ties with various sources, including Incyte. Four authors declared being current or former employees and shareholders of Incyte.
Source: Papp K et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: Results from two phase 3 studies. J Am Acad Dermatol. 2022 (Nov 25). Doi: 10.1016/j.jaad.2022.09.060
Key clinical point: Ruxolitinib cream demonstrated effective disease control and was well tolerated in patients with atopic dermatitis (AD) during 44 weeks of as-needed treatment.
Major finding: At week 52, 74.1%-77.8% of patients had an Investigator’s Global Assessment score of 0/1, with the mean affected body surface area being 1.4%-1.8%. Treatment-related adverse events were reported in 8.7%/7.4% of patients on 0.75%/1.5% ruxolitinib and in 2.0%/6.1% of those who switched from vehicle to 0.75%/1.5% ruxolitinib, respectively.
Study details: This study analyzed pooled data from two phase 3 studies, TRuE-AD1 and TRuE-AD2, including 1249 patients aged ≥12 years with AD who were randomly assigned to receive 0.75% or 1.5% ruxolitinib cream or vehicle for 8 weeks; thereafter, the vehicle group patients were re-assigned to receive either strength ruxolitinib cream for 44 weeks.
Disclosures: This study was funded by Incyte Corporation, U.S. Some authors reported ties with various sources, including Incyte. Four authors declared being current or former employees and shareholders of Incyte.
Source: Papp K et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: Results from two phase 3 studies. J Am Acad Dermatol. 2022 (Nov 25). Doi: 10.1016/j.jaad.2022.09.060
Key clinical point: Ruxolitinib cream demonstrated effective disease control and was well tolerated in patients with atopic dermatitis (AD) during 44 weeks of as-needed treatment.
Major finding: At week 52, 74.1%-77.8% of patients had an Investigator’s Global Assessment score of 0/1, with the mean affected body surface area being 1.4%-1.8%. Treatment-related adverse events were reported in 8.7%/7.4% of patients on 0.75%/1.5% ruxolitinib and in 2.0%/6.1% of those who switched from vehicle to 0.75%/1.5% ruxolitinib, respectively.
Study details: This study analyzed pooled data from two phase 3 studies, TRuE-AD1 and TRuE-AD2, including 1249 patients aged ≥12 years with AD who were randomly assigned to receive 0.75% or 1.5% ruxolitinib cream or vehicle for 8 weeks; thereafter, the vehicle group patients were re-assigned to receive either strength ruxolitinib cream for 44 weeks.
Disclosures: This study was funded by Incyte Corporation, U.S. Some authors reported ties with various sources, including Incyte. Four authors declared being current or former employees and shareholders of Incyte.
Source: Papp K et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: Results from two phase 3 studies. J Am Acad Dermatol. 2022 (Nov 25). Doi: 10.1016/j.jaad.2022.09.060
Tralokinumab improves microbial dysbiosis in lesional skin in AD
Key clinical point: Specific targeting of interleukin-13 alone with tralokinumab improved microbial diversity and reduced Staphylococcus aureus abundance in the lesional skin in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: The 16-week tralokinumab treatment reduced S. aureus abundance by 20.7-fold (P < .0001), whereas placebo led to a non-statistically significant reduction. Tralokinumab also led to a significant increase in microbial diversity as early as week 8 (P < .001) and also at week 16 (P < .05).
Study details: The data come from the phase 3 ECZTRA 1 trial including 802 patients with moderate-to-severe AD who were randomly assigned to receive 300 mg subcutaneous tralokinumab or placebo.
Disclosures: This study and the ECZTRA 1 trial were funded by LEO Pharma A/S. Some authors declared serving as speakers, consultants, investigators, scientific advisors, or clinical study investigators or receiving institutional research grants from various sources, including LEO Pharma.
Source: Beck LA et al. Tralokinumab treatment improves the skin microbiota by increasing the microbial diversity in adults with moderate-to-severe atopic dermatitis: Analysis of microbial diversity in ECZTRA 1, a randomized controlled trial. J Am Acad Dermatol. 2022 (Dec 2). Doi: 10.1016/j.jaad.2022.11.047
Key clinical point: Specific targeting of interleukin-13 alone with tralokinumab improved microbial diversity and reduced Staphylococcus aureus abundance in the lesional skin in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: The 16-week tralokinumab treatment reduced S. aureus abundance by 20.7-fold (P < .0001), whereas placebo led to a non-statistically significant reduction. Tralokinumab also led to a significant increase in microbial diversity as early as week 8 (P < .001) and also at week 16 (P < .05).
Study details: The data come from the phase 3 ECZTRA 1 trial including 802 patients with moderate-to-severe AD who were randomly assigned to receive 300 mg subcutaneous tralokinumab or placebo.
Disclosures: This study and the ECZTRA 1 trial were funded by LEO Pharma A/S. Some authors declared serving as speakers, consultants, investigators, scientific advisors, or clinical study investigators or receiving institutional research grants from various sources, including LEO Pharma.
Source: Beck LA et al. Tralokinumab treatment improves the skin microbiota by increasing the microbial diversity in adults with moderate-to-severe atopic dermatitis: Analysis of microbial diversity in ECZTRA 1, a randomized controlled trial. J Am Acad Dermatol. 2022 (Dec 2). Doi: 10.1016/j.jaad.2022.11.047
Key clinical point: Specific targeting of interleukin-13 alone with tralokinumab improved microbial diversity and reduced Staphylococcus aureus abundance in the lesional skin in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: The 16-week tralokinumab treatment reduced S. aureus abundance by 20.7-fold (P < .0001), whereas placebo led to a non-statistically significant reduction. Tralokinumab also led to a significant increase in microbial diversity as early as week 8 (P < .001) and also at week 16 (P < .05).
Study details: The data come from the phase 3 ECZTRA 1 trial including 802 patients with moderate-to-severe AD who were randomly assigned to receive 300 mg subcutaneous tralokinumab or placebo.
Disclosures: This study and the ECZTRA 1 trial were funded by LEO Pharma A/S. Some authors declared serving as speakers, consultants, investigators, scientific advisors, or clinical study investigators or receiving institutional research grants from various sources, including LEO Pharma.
Source: Beck LA et al. Tralokinumab treatment improves the skin microbiota by increasing the microbial diversity in adults with moderate-to-severe atopic dermatitis: Analysis of microbial diversity in ECZTRA 1, a randomized controlled trial. J Am Acad Dermatol. 2022 (Dec 2). Doi: 10.1016/j.jaad.2022.11.047
Tralokinumab improves microbial dysbiosis in lesional skin in AD
Key clinical point: Specific targeting of interleukin-13 alone with tralokinumab improved microbial diversity and reduced Staphylococcus aureus abundance in the lesional skin in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: The 16-week tralokinumab treatment reduced S. aureus abundance by 20.7-fold (P < .0001), whereas placebo led to a non-statistically significant reduction. Tralokinumab also led to a significant increase in microbial diversity as early as week 8 (P < .001) and also at week 16 (P < .05).
Study details: The data come from the phase 3 ECZTRA 1 trial including 802 patients with moderate-to-severe AD who were randomly assigned to receive 300 mg subcutaneous tralokinumab or placebo.
Disclosures: This study and the ECZTRA 1 trial were funded by LEO Pharma A/S. Some authors declared serving as speakers, consultants, investigators, scientific advisors, or clinical study investigators or receiving institutional research grants from various sources, including LEO Pharma.
Source: Beck LA et al. Tralokinumab treatment improves the skin microbiota by increasing the microbial diversity in adults with moderate-to-severe atopic dermatitis: Analysis of microbial diversity in ECZTRA 1, a randomized controlled trial. J Am Acad Dermatol. 2022 (Dec 2). Doi: 10.1016/j.jaad.2022.11.047
Key clinical point: Specific targeting of interleukin-13 alone with tralokinumab improved microbial diversity and reduced Staphylococcus aureus abundance in the lesional skin in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: The 16-week tralokinumab treatment reduced S. aureus abundance by 20.7-fold (P < .0001), whereas placebo led to a non-statistically significant reduction. Tralokinumab also led to a significant increase in microbial diversity as early as week 8 (P < .001) and also at week 16 (P < .05).
Study details: The data come from the phase 3 ECZTRA 1 trial including 802 patients with moderate-to-severe AD who were randomly assigned to receive 300 mg subcutaneous tralokinumab or placebo.
Disclosures: This study and the ECZTRA 1 trial were funded by LEO Pharma A/S. Some authors declared serving as speakers, consultants, investigators, scientific advisors, or clinical study investigators or receiving institutional research grants from various sources, including LEO Pharma.
Source: Beck LA et al. Tralokinumab treatment improves the skin microbiota by increasing the microbial diversity in adults with moderate-to-severe atopic dermatitis: Analysis of microbial diversity in ECZTRA 1, a randomized controlled trial. J Am Acad Dermatol. 2022 (Dec 2). Doi: 10.1016/j.jaad.2022.11.047
Key clinical point: Specific targeting of interleukin-13 alone with tralokinumab improved microbial diversity and reduced Staphylococcus aureus abundance in the lesional skin in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: The 16-week tralokinumab treatment reduced S. aureus abundance by 20.7-fold (P < .0001), whereas placebo led to a non-statistically significant reduction. Tralokinumab also led to a significant increase in microbial diversity as early as week 8 (P < .001) and also at week 16 (P < .05).
Study details: The data come from the phase 3 ECZTRA 1 trial including 802 patients with moderate-to-severe AD who were randomly assigned to receive 300 mg subcutaneous tralokinumab or placebo.
Disclosures: This study and the ECZTRA 1 trial were funded by LEO Pharma A/S. Some authors declared serving as speakers, consultants, investigators, scientific advisors, or clinical study investigators or receiving institutional research grants from various sources, including LEO Pharma.
Source: Beck LA et al. Tralokinumab treatment improves the skin microbiota by increasing the microbial diversity in adults with moderate-to-severe atopic dermatitis: Analysis of microbial diversity in ECZTRA 1, a randomized controlled trial. J Am Acad Dermatol. 2022 (Dec 2). Doi: 10.1016/j.jaad.2022.11.047
Meta-analysis supports safe use of topical calcineurin inhibitors in AD
Key clinical point: An exposure to topical calcineurin inhibitors did not increase the risk for cancer in patients with atopic dermatitis (AD), with findings being similar among infants, children, and adults.
Major finding: Compared with no exposure, topical calcineurin inhibitor exposure was not associated with an increased risk for cancer (odds ratio [OR] 1.03; 95% credible interval [CrI] 0.94-1.11), with neither pimecrolimus (OR 1.05; 95% CrI 0.94-1.15) nor tacrolimus (OR 0.99; 95% CrI 0.89-1.09) use revealing any association with increased cancer risk, across all age groups.
Study details: This was a systematic review and meta-analysis of 110 unique studies (52 randomized controlled trials and 69 nonrandomized studies) including 3.4 million patients with AD followed-up for a mean of 11 months.
Disclosures: This study was funded by the American Academy of Allergy, Asthma, and Immunology and the American College of Allergy, Asthma, and Immunology. L Schneider declared receiving consulting fees and payments to her institutions from, serving on data safety monitoring and advisory boards for, and holding stock or stock options in various sources.
Source: Devasenapathy N et al for the AAAAI/ACAAI Joint Task Force on Practice Parameters for Atopic Dermatitis Guideline Development Group. Cancer risk with topical calcineurin inhibitors, pimecrolimus and tacrolimus, for atopic dermatitis: A systematic review and meta-analysis. Lancet Child Adolesc Health. 2022 (Nov 9). Doi: 10.1016/S2352-4642(22)00283-8
Key clinical point: An exposure to topical calcineurin inhibitors did not increase the risk for cancer in patients with atopic dermatitis (AD), with findings being similar among infants, children, and adults.
Major finding: Compared with no exposure, topical calcineurin inhibitor exposure was not associated with an increased risk for cancer (odds ratio [OR] 1.03; 95% credible interval [CrI] 0.94-1.11), with neither pimecrolimus (OR 1.05; 95% CrI 0.94-1.15) nor tacrolimus (OR 0.99; 95% CrI 0.89-1.09) use revealing any association with increased cancer risk, across all age groups.
Study details: This was a systematic review and meta-analysis of 110 unique studies (52 randomized controlled trials and 69 nonrandomized studies) including 3.4 million patients with AD followed-up for a mean of 11 months.
Disclosures: This study was funded by the American Academy of Allergy, Asthma, and Immunology and the American College of Allergy, Asthma, and Immunology. L Schneider declared receiving consulting fees and payments to her institutions from, serving on data safety monitoring and advisory boards for, and holding stock or stock options in various sources.
Source: Devasenapathy N et al for the AAAAI/ACAAI Joint Task Force on Practice Parameters for Atopic Dermatitis Guideline Development Group. Cancer risk with topical calcineurin inhibitors, pimecrolimus and tacrolimus, for atopic dermatitis: A systematic review and meta-analysis. Lancet Child Adolesc Health. 2022 (Nov 9). Doi: 10.1016/S2352-4642(22)00283-8
Key clinical point: An exposure to topical calcineurin inhibitors did not increase the risk for cancer in patients with atopic dermatitis (AD), with findings being similar among infants, children, and adults.
Major finding: Compared with no exposure, topical calcineurin inhibitor exposure was not associated with an increased risk for cancer (odds ratio [OR] 1.03; 95% credible interval [CrI] 0.94-1.11), with neither pimecrolimus (OR 1.05; 95% CrI 0.94-1.15) nor tacrolimus (OR 0.99; 95% CrI 0.89-1.09) use revealing any association with increased cancer risk, across all age groups.
Study details: This was a systematic review and meta-analysis of 110 unique studies (52 randomized controlled trials and 69 nonrandomized studies) including 3.4 million patients with AD followed-up for a mean of 11 months.
Disclosures: This study was funded by the American Academy of Allergy, Asthma, and Immunology and the American College of Allergy, Asthma, and Immunology. L Schneider declared receiving consulting fees and payments to her institutions from, serving on data safety monitoring and advisory boards for, and holding stock or stock options in various sources.
Source: Devasenapathy N et al for the AAAAI/ACAAI Joint Task Force on Practice Parameters for Atopic Dermatitis Guideline Development Group. Cancer risk with topical calcineurin inhibitors, pimecrolimus and tacrolimus, for atopic dermatitis: A systematic review and meta-analysis. Lancet Child Adolesc Health. 2022 (Nov 9). Doi: 10.1016/S2352-4642(22)00283-8
Rocatinlimab shows promise in moderate-to-severe atopic dermatitis
Key clinical point: Treatment with rocatinlimab, a novel monoclonal antibody, significantly improved disease severity at all dosing regimens in patients with moderate-to-severe atopic dermatitis (AD) inadequately controlled with topical therapy, which was maintained in most patients even after treatment discontinuation.
Major finding: The least-squares mean percent reductions in the Eczema Area and Severity Index at week 16 were significantly greater with 150 mg rocatinlimab every 4 weeks vs placebo (−48.3% vs −15.0%; P = .0003), with all other active rocatinlimab dose regimens vs placebo showing improvement (all P < .05) and most patients showing sustained response during off-drug follow-up.
Study details: The data come from a multicenter phase 2b study including 274 patients with moderate-to-severe AD and inadequate response or intolerance to topical medications and who were randomly assigned to receive rocatinlimab or placebo.
Disclosures: This study was funded by Kyowa Kirin. Some authors reported ties with various sources, including Kyowa Kirin. E Esfandiari declared being an employee of Kyowa Kirin.
Source: Guttman-Yassky E et al. An anti-OX40 antibody to treat moderate-to-severe atopic dermatitis: A multicentre, double-blind, placebo-controlled phase 2b study. Lancet. 2022 (Dec 9). Doi: 10.1016/S0140-6736(22)02037-2
Key clinical point: Treatment with rocatinlimab, a novel monoclonal antibody, significantly improved disease severity at all dosing regimens in patients with moderate-to-severe atopic dermatitis (AD) inadequately controlled with topical therapy, which was maintained in most patients even after treatment discontinuation.
Major finding: The least-squares mean percent reductions in the Eczema Area and Severity Index at week 16 were significantly greater with 150 mg rocatinlimab every 4 weeks vs placebo (−48.3% vs −15.0%; P = .0003), with all other active rocatinlimab dose regimens vs placebo showing improvement (all P < .05) and most patients showing sustained response during off-drug follow-up.
Study details: The data come from a multicenter phase 2b study including 274 patients with moderate-to-severe AD and inadequate response or intolerance to topical medications and who were randomly assigned to receive rocatinlimab or placebo.
Disclosures: This study was funded by Kyowa Kirin. Some authors reported ties with various sources, including Kyowa Kirin. E Esfandiari declared being an employee of Kyowa Kirin.
Source: Guttman-Yassky E et al. An anti-OX40 antibody to treat moderate-to-severe atopic dermatitis: A multicentre, double-blind, placebo-controlled phase 2b study. Lancet. 2022 (Dec 9). Doi: 10.1016/S0140-6736(22)02037-2
Key clinical point: Treatment with rocatinlimab, a novel monoclonal antibody, significantly improved disease severity at all dosing regimens in patients with moderate-to-severe atopic dermatitis (AD) inadequately controlled with topical therapy, which was maintained in most patients even after treatment discontinuation.
Major finding: The least-squares mean percent reductions in the Eczema Area and Severity Index at week 16 were significantly greater with 150 mg rocatinlimab every 4 weeks vs placebo (−48.3% vs −15.0%; P = .0003), with all other active rocatinlimab dose regimens vs placebo showing improvement (all P < .05) and most patients showing sustained response during off-drug follow-up.
Study details: The data come from a multicenter phase 2b study including 274 patients with moderate-to-severe AD and inadequate response or intolerance to topical medications and who were randomly assigned to receive rocatinlimab or placebo.
Disclosures: This study was funded by Kyowa Kirin. Some authors reported ties with various sources, including Kyowa Kirin. E Esfandiari declared being an employee of Kyowa Kirin.
Source: Guttman-Yassky E et al. An anti-OX40 antibody to treat moderate-to-severe atopic dermatitis: A multicentre, double-blind, placebo-controlled phase 2b study. Lancet. 2022 (Dec 9). Doi: 10.1016/S0140-6736(22)02037-2
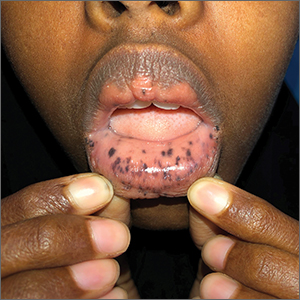
Macules and abdominal pain
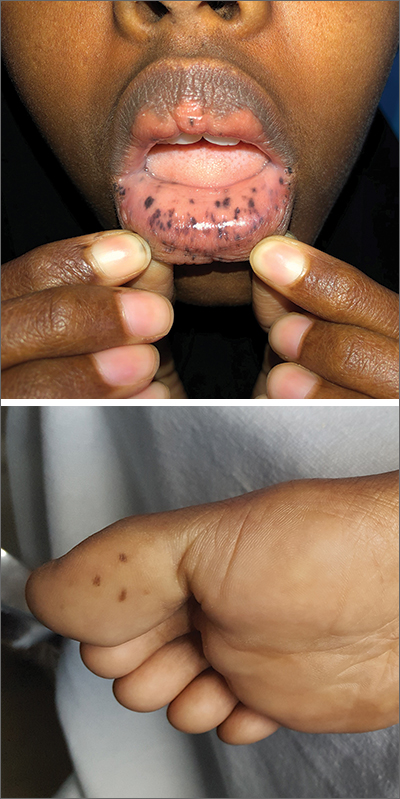
This patient was given a diagnosis of Peutz-Jeghers syndrome (PJS) based on the characteristic pigmented mucocutaneous macules and numerous polyps in her stomach and small bowel. PJS is an autosomal dominant syndrome characterized by mucocutaneous pigmentation, polyposis of the GI tract, and increased cancer risk. The prevalence is approximately 1 in 100,000.1 Genetic testing for the STK11 gene mutation, which is found in 70% of familial cases and 30% to 67% of sporadic cases, is not required for diagnosis.1
The bluish brown to black spots of PJS often are apparent at birth or in early infancy. They are most common on the lips, buccal mucosa, perioral region, palms, and soles.
The polyps may cause bleeding, anemia, and abdominal pain due to intussusception, obstruction, or infarction.2 Polyps usually are benign, but patients are at increased risk of GI and non-GI malignancies such as breast, pancreas, lung, and reproductive tract cancers.1
PJS can be differentiated from other causes of hyperpigmentation by clinical presentation and/or genetic testing. The diagnosis of PJS is made using the following criteria: (1) two or more histologically confirmed PJS polyps, (2) any number of PJS polyps and a family history of PJS, (3) characteristic mucocutaneous pigmentation and a family history of PJS, or (4) any number of PJS polyps and characteristic mucocutaneous pigmentation.2
It’s recommended that polyps be removed when technically feasible.3 Pigmented macules do not require treatment. Macules on the lips may disappear with time, while those on the buccal mucosa persist. The lip lesions can be lightened with chemical peels or laser.
This patient underwent laparotomy, which revealed a grossly dilated and gangrenous small bowel segment. Intussusception was not present and was thought to have spontaneously reduced. Resection and anastomosis of the affected small bowel was performed. The patient’s postoperative course was uneventful, and her diarrhea and abdominal pain resolved. A colonoscopy was normal, and the health care team explained that she would require follow-up and surveillance of her condition due to the high risk of future cancers.
This case was adapted from: Warsame MO, McMichael JR. Chronic abdominal pain and diarrhea. J Fam Pract. 2020;69:365,366,368.
Photos courtesy of Mohamed Omar Warsame, MBBS, and Josette R. McMichael, MD
1. Kopacova M, Tacheci I, Rejchrt S, et al. Peutz-Jeghers syndrome: diagnostic and therapeutic approach. World J Gastroenterol. 2009;15:5397-5408.
2. Beggs AD, Latchford AR, Vasen HF, et al. Peutz-Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010;59:975-986.
3. van Lier MG, Mathus-Vliegen EM, Wagner A, et al. High cumulative risk of intussusception in patients with Peutz-Jeghers syndrome: time to update surveillance guidelines? Am J Gastroenterol. 2011;106:940-945.

This patient was given a diagnosis of Peutz-Jeghers syndrome (PJS) based on the characteristic pigmented mucocutaneous macules and numerous polyps in her stomach and small bowel. PJS is an autosomal dominant syndrome characterized by mucocutaneous pigmentation, polyposis of the GI tract, and increased cancer risk. The prevalence is approximately 1 in 100,000.1 Genetic testing for the STK11 gene mutation, which is found in 70% of familial cases and 30% to 67% of sporadic cases, is not required for diagnosis.1
The bluish brown to black spots of PJS often are apparent at birth or in early infancy. They are most common on the lips, buccal mucosa, perioral region, palms, and soles.
The polyps may cause bleeding, anemia, and abdominal pain due to intussusception, obstruction, or infarction.2 Polyps usually are benign, but patients are at increased risk of GI and non-GI malignancies such as breast, pancreas, lung, and reproductive tract cancers.1
PJS can be differentiated from other causes of hyperpigmentation by clinical presentation and/or genetic testing. The diagnosis of PJS is made using the following criteria: (1) two or more histologically confirmed PJS polyps, (2) any number of PJS polyps and a family history of PJS, (3) characteristic mucocutaneous pigmentation and a family history of PJS, or (4) any number of PJS polyps and characteristic mucocutaneous pigmentation.2
It’s recommended that polyps be removed when technically feasible.3 Pigmented macules do not require treatment. Macules on the lips may disappear with time, while those on the buccal mucosa persist. The lip lesions can be lightened with chemical peels or laser.
This patient underwent laparotomy, which revealed a grossly dilated and gangrenous small bowel segment. Intussusception was not present and was thought to have spontaneously reduced. Resection and anastomosis of the affected small bowel was performed. The patient’s postoperative course was uneventful, and her diarrhea and abdominal pain resolved. A colonoscopy was normal, and the health care team explained that she would require follow-up and surveillance of her condition due to the high risk of future cancers.
This case was adapted from: Warsame MO, McMichael JR. Chronic abdominal pain and diarrhea. J Fam Pract. 2020;69:365,366,368.
Photos courtesy of Mohamed Omar Warsame, MBBS, and Josette R. McMichael, MD

This patient was given a diagnosis of Peutz-Jeghers syndrome (PJS) based on the characteristic pigmented mucocutaneous macules and numerous polyps in her stomach and small bowel. PJS is an autosomal dominant syndrome characterized by mucocutaneous pigmentation, polyposis of the GI tract, and increased cancer risk. The prevalence is approximately 1 in 100,000.1 Genetic testing for the STK11 gene mutation, which is found in 70% of familial cases and 30% to 67% of sporadic cases, is not required for diagnosis.1
The bluish brown to black spots of PJS often are apparent at birth or in early infancy. They are most common on the lips, buccal mucosa, perioral region, palms, and soles.
The polyps may cause bleeding, anemia, and abdominal pain due to intussusception, obstruction, or infarction.2 Polyps usually are benign, but patients are at increased risk of GI and non-GI malignancies such as breast, pancreas, lung, and reproductive tract cancers.1
PJS can be differentiated from other causes of hyperpigmentation by clinical presentation and/or genetic testing. The diagnosis of PJS is made using the following criteria: (1) two or more histologically confirmed PJS polyps, (2) any number of PJS polyps and a family history of PJS, (3) characteristic mucocutaneous pigmentation and a family history of PJS, or (4) any number of PJS polyps and characteristic mucocutaneous pigmentation.2
It’s recommended that polyps be removed when technically feasible.3 Pigmented macules do not require treatment. Macules on the lips may disappear with time, while those on the buccal mucosa persist. The lip lesions can be lightened with chemical peels or laser.
This patient underwent laparotomy, which revealed a grossly dilated and gangrenous small bowel segment. Intussusception was not present and was thought to have spontaneously reduced. Resection and anastomosis of the affected small bowel was performed. The patient’s postoperative course was uneventful, and her diarrhea and abdominal pain resolved. A colonoscopy was normal, and the health care team explained that she would require follow-up and surveillance of her condition due to the high risk of future cancers.
This case was adapted from: Warsame MO, McMichael JR. Chronic abdominal pain and diarrhea. J Fam Pract. 2020;69:365,366,368.
Photos courtesy of Mohamed Omar Warsame, MBBS, and Josette R. McMichael, MD
1. Kopacova M, Tacheci I, Rejchrt S, et al. Peutz-Jeghers syndrome: diagnostic and therapeutic approach. World J Gastroenterol. 2009;15:5397-5408.
2. Beggs AD, Latchford AR, Vasen HF, et al. Peutz-Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010;59:975-986.
3. van Lier MG, Mathus-Vliegen EM, Wagner A, et al. High cumulative risk of intussusception in patients with Peutz-Jeghers syndrome: time to update surveillance guidelines? Am J Gastroenterol. 2011;106:940-945.
1. Kopacova M, Tacheci I, Rejchrt S, et al. Peutz-Jeghers syndrome: diagnostic and therapeutic approach. World J Gastroenterol. 2009;15:5397-5408.
2. Beggs AD, Latchford AR, Vasen HF, et al. Peutz-Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010;59:975-986.
3. van Lier MG, Mathus-Vliegen EM, Wagner A, et al. High cumulative risk of intussusception in patients with Peutz-Jeghers syndrome: time to update surveillance guidelines? Am J Gastroenterol. 2011;106:940-945.