User login
Official Newspaper of the American College of Surgeons
FDA approves Sapien 3 transcatheter valve for bioprosthetic valve failure
The Food and Drug Administration announced June 5 the approval of an expanded indication for the Sapien 3 Transcatheter Heart Valve (THV) for patients with symptomatic heart disease caused by failure of a previously placed bioprosthetic aortic or mitral valve who have a risk of death or severe complications from repeat surgery.
This is the first FDA approval for the expanded use of the Sapien 3 THV as a valve-in-valve treatment. Such procedures provide an alternative to repeat surgery.
“For the first time, a regulatory agency is approving a transcatheter heart valve as a valve-in-valve treatment when bioprosthetic mitral or aortic valves fail in patients who are at high or greater risk of complications from repeat surgery,” Bram Zuckerman, MD, director of the division of cardiovascular devices at the FDA’s Center for Devices and Radiological Health, said in a press release. “This new approval offers U.S. patients with failing surgical bioprosthetic aortic or mitral valves a less-invasive treatment option.”
Originally, the FDA approved the Sapien 3 THV for transcatheter aortic valve replacement (TAVR) as an alternative to surgical aortic valve replacement for patients with native aortic stenosis whose risk for death or severe complications from surgery is high or greater. Then in 2016, the FDA expanded the TAVR indication for Sapien 3 THV to include patients who are at intermediate surgical risk for death or complications.
Read the full press release on the FDA’s website.
The Food and Drug Administration announced June 5 the approval of an expanded indication for the Sapien 3 Transcatheter Heart Valve (THV) for patients with symptomatic heart disease caused by failure of a previously placed bioprosthetic aortic or mitral valve who have a risk of death or severe complications from repeat surgery.
This is the first FDA approval for the expanded use of the Sapien 3 THV as a valve-in-valve treatment. Such procedures provide an alternative to repeat surgery.
“For the first time, a regulatory agency is approving a transcatheter heart valve as a valve-in-valve treatment when bioprosthetic mitral or aortic valves fail in patients who are at high or greater risk of complications from repeat surgery,” Bram Zuckerman, MD, director of the division of cardiovascular devices at the FDA’s Center for Devices and Radiological Health, said in a press release. “This new approval offers U.S. patients with failing surgical bioprosthetic aortic or mitral valves a less-invasive treatment option.”
Originally, the FDA approved the Sapien 3 THV for transcatheter aortic valve replacement (TAVR) as an alternative to surgical aortic valve replacement for patients with native aortic stenosis whose risk for death or severe complications from surgery is high or greater. Then in 2016, the FDA expanded the TAVR indication for Sapien 3 THV to include patients who are at intermediate surgical risk for death or complications.
Read the full press release on the FDA’s website.
The Food and Drug Administration announced June 5 the approval of an expanded indication for the Sapien 3 Transcatheter Heart Valve (THV) for patients with symptomatic heart disease caused by failure of a previously placed bioprosthetic aortic or mitral valve who have a risk of death or severe complications from repeat surgery.
This is the first FDA approval for the expanded use of the Sapien 3 THV as a valve-in-valve treatment. Such procedures provide an alternative to repeat surgery.
“For the first time, a regulatory agency is approving a transcatheter heart valve as a valve-in-valve treatment when bioprosthetic mitral or aortic valves fail in patients who are at high or greater risk of complications from repeat surgery,” Bram Zuckerman, MD, director of the division of cardiovascular devices at the FDA’s Center for Devices and Radiological Health, said in a press release. “This new approval offers U.S. patients with failing surgical bioprosthetic aortic or mitral valves a less-invasive treatment option.”
Originally, the FDA approved the Sapien 3 THV for transcatheter aortic valve replacement (TAVR) as an alternative to surgical aortic valve replacement for patients with native aortic stenosis whose risk for death or severe complications from surgery is high or greater. Then in 2016, the FDA expanded the TAVR indication for Sapien 3 THV to include patients who are at intermediate surgical risk for death or complications.
Read the full press release on the FDA’s website.
Medicare Advantage enrollment up again in 2017
Enrollment in Medicare Advantage plans increased for the 13th consecutive year in 2017 and now represents one-third of all Medicare beneficiaries, according to the Kaiser Family Foundation.
Currently, 19 million beneficiaries are enrolled in Medicare Advantage, more than triple the number who were enrolled when the program hit its low point of 5.3 million (13% of all beneficiaries) in 2003 and 2004 and 71% higher since the Affordable Care Act was passed in 2010, Kaiser said in a recent report.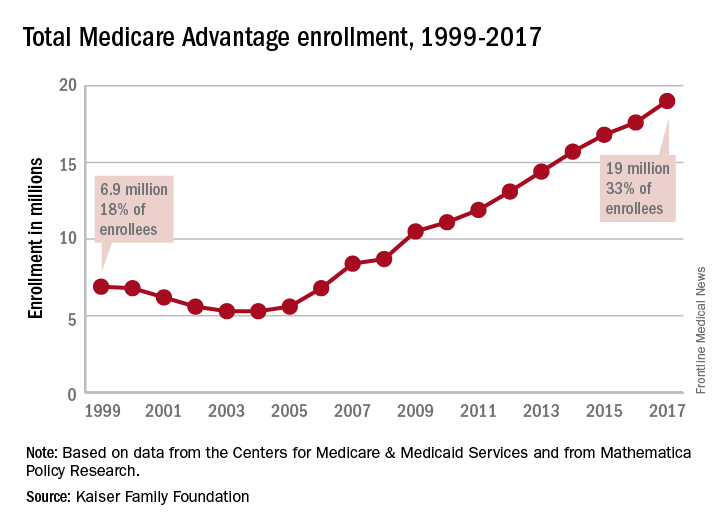
As growth continues, however, so does concentration among the private firms administering the plans. In 2017, UnitedHealthcare and Humana together account for 41% of enrollment in Medicare Advantage, “and, in 17 states, one company has more than half of all Medicare Advantage enrollment – an indicator that these markets may not be very competitive,” they noted.
Enrollment in Medicare Advantage plans increased for the 13th consecutive year in 2017 and now represents one-third of all Medicare beneficiaries, according to the Kaiser Family Foundation.
Currently, 19 million beneficiaries are enrolled in Medicare Advantage, more than triple the number who were enrolled when the program hit its low point of 5.3 million (13% of all beneficiaries) in 2003 and 2004 and 71% higher since the Affordable Care Act was passed in 2010, Kaiser said in a recent report.
As growth continues, however, so does concentration among the private firms administering the plans. In 2017, UnitedHealthcare and Humana together account for 41% of enrollment in Medicare Advantage, “and, in 17 states, one company has more than half of all Medicare Advantage enrollment – an indicator that these markets may not be very competitive,” they noted.
Enrollment in Medicare Advantage plans increased for the 13th consecutive year in 2017 and now represents one-third of all Medicare beneficiaries, according to the Kaiser Family Foundation.
Currently, 19 million beneficiaries are enrolled in Medicare Advantage, more than triple the number who were enrolled when the program hit its low point of 5.3 million (13% of all beneficiaries) in 2003 and 2004 and 71% higher since the Affordable Care Act was passed in 2010, Kaiser said in a recent report.
As growth continues, however, so does concentration among the private firms administering the plans. In 2017, UnitedHealthcare and Humana together account for 41% of enrollment in Medicare Advantage, “and, in 17 states, one company has more than half of all Medicare Advantage enrollment – an indicator that these markets may not be very competitive,” they noted.
BIO-RESORT: A mandate to prescreen PCI patients for silent diabetes
PARIS – Undetected diabetes and prediabetes are pervasive in patients undergoing percutaneous coronary intervention, and they’re associated with a sharply increased risk of major adverse cardiovascular events, according to the results of the potentially practice-changing BIO-RESORT Silent Diabetes Study, Clemens von Birgelen, MD, PhD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Our data support screening PCI all-comers for silent diabetes, which may help identify patients with an increased event risk and improve their therapy,” said Dr. von Birgelen, professor of cardiology at the Thoraxcentrum of Twente, a high-volume center for cardiac interventions in Enschede, the Netherlands.
A substantial one-third of subjects turned out to have abnormal glucose tolerance according to World Health Organization criteria and an International Expert Committee Report (Diabetes Care. 2009 Jul;32[7]:1327-34). In a multivariate analysis, their 1-year rate of the primary study endpoint – target vessel failure, a composite of cardiac death, target vessel-related MI, or target vessel revascularization – was an adjusted 2.2 times greater than in the 788 normoglycemic patients.
Moreover, among the 7% of study participants who met diagnostic criteria for silent diabetes, the risk of target vessel failure was more than 4.4 times greater than in the normoglycemic group.
“To a very great extent, periprocedural MI is the driving force behind this difference that we saw. From a biological point of view, I think that the vulnerability of the vessel in the diabetic or prediabetic patient features more brittle plaque with a higher risk of cholesterol embolization, and with more plaque mass that can be pushed to the side so that side branch vessels can become occluded, leading to periprocedural MI,” he observed.
Glucose metabolism was assessed in all participants by two methods using the conventional cutoffs: a 2-hour oral glucose tolerance test (OGTT), and the combination of fasting plasma glucose and hemoglobin A1c. By OGTT, 7% of patients had silent, previously unrecognized diabetes and another 13% had prediabetes. Using the combination of fasting plasma glucose and HbA1c, a total of 25% of subjects had silent diabetes or prediabetes. Fully 33% of participants had abnormal glucose metabolism by one yardstick or the other.
“What we have seen is there is a group of patients that are missed with either. With the OGTT you don’t see all the diabetics, and with HbA1c and fasting blood glucose you also miss some patients,” said Dr. von Birgelen.
The 1-year cumulative incidence of target vessel failure was 13.2% in patients with silent diabetes as identified by the OGTT and 12.1% in those detected by the alternative method, compared with rates of 2.8% and 3.1%, respectively, in normoglycemic PCI patients. The event rate was 6.1% in patients with prediabetes by OGTT and similar at 5.5% in those found to be prediabetic based on fasting blood glucose and HbA1c, versus rates of 2.8% and 3.1%, respectively, in normoglycemic patients.
“The findings of this study suggest that post-PCI event risk associated with hyperglycemia is a continuum without a clear threshold effect, extending well beyond the threshold that currently defines diabetes,” Dr. von Birgelen said.
Once again, it’s worth emphasizing that the elevated target vessel failure rates seen in patients with abnormal glucose metabolism were due mostly to increased rates of acute MI within the first 24 hours after PCI. The target vessel–related MI rate was 10.3% in patients with silent diabetes, compared with just 1.8% in normoglycemic controls.
Asked what the take-home message for clinicians is from this study, he noted that the Netherlands has a relatively low prevalence of diabetes, and a highly developed primary care medicine system.
“We have a very good one-to-one relationship between the patient and the GP. So if we find 7% silent diabetes and up to one-third of patients with undetected abnormal glucose tolerance in a country with a relatively low prevalence of diabetes, you may expect that in other countries with a higher prevalence and perhaps a less developed primary care system the rate may be much, much higher,” Dr. von Birgelen cautioned.
The implications for the daily clinical practice of interventional cardiology are clear, he continued: “We’ve seen in several trials that the new stents are doing a fantastic job. So if we want to further improve the outcomes in our patients we have to do something else. We should look for subgroups of our PCI patients who have a particularly high risk. And we all realize that diabetics are such a problem, but I think we have shown that the prediabetic patients are also important. So we should identify and pretreat these patients, perhaps with aggressive lipid-lowering therapy during the weeks before a scheduled elective PCI.”
“There are data showing that with aggressive lipid-lowering you might reduce the risk of periprocedural MI,” the cardiologist noted.
As a practical matter, screening via fasting blood glucose and HbA1c is probably the way to go in clinical practice, according to Dr. von Birgelen.
“In this study, we performed the OGTT because it is still considered by many the gold standard. But there is increasing evidence favoring HbA1c data and fasting blood glucose,” he said.
Other possible pre-PCI interventions worthy of consideration in patients found to have previously unsuspected abnormal glucose tolerance might include medical therapy aimed at normalizing glucose metabolism, as well as perhaps resorting to the most potent forms of dual-antiplatelet therapy in patients with stable angina who have impaired glucose tolerance. However, these are possibilities that should be tested in randomized controlled trials before widespread adoption, he added.
The BIO-RESORT Silent Diabetes Study, which will continue for 5 years of post-PCI follow-up, is a prespecified substudy of the previously reported BIO-RESORT trial, which addressed another issue entirely. It was a three-arm, patient-blinded clinical trial comparing 1-year safety and efficacy outcomes in nearly 3,500 PCI patients randomized to PCI with very thin strut biodegradable polymer everolimus- or sirolimus-eluting stents or a durable polymer zotarolimus-eluting stent. Outcomes proved noninferior across the three treatment groups (Lancet. 2016 Nov 26;388[10060]:2607-17).
Dr. von Birgelen observed that the silent diabetes study broke new ground. Prior studies of PCI outcomes in patients with unrecognized diabetes were limited to recipients of plain old balloon angioplasty, bare metal, or first-generation drug-eluting stents. And studies of PCI in patients with unrecognized prediabetes are virtually nonexistent.
As the principal investigator for both the parent BIO-RESORT trial and the silent diabetes substudy, Dr. von Birgelen received research grants from Biotronik, Boston Scientific, and Medtronic, the cosponsors. He applauded the three companies for funding the silent diabetes substudy in the interest of science even though it had no commercial relevance to their stent businesses.
PARIS – Undetected diabetes and prediabetes are pervasive in patients undergoing percutaneous coronary intervention, and they’re associated with a sharply increased risk of major adverse cardiovascular events, according to the results of the potentially practice-changing BIO-RESORT Silent Diabetes Study, Clemens von Birgelen, MD, PhD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Our data support screening PCI all-comers for silent diabetes, which may help identify patients with an increased event risk and improve their therapy,” said Dr. von Birgelen, professor of cardiology at the Thoraxcentrum of Twente, a high-volume center for cardiac interventions in Enschede, the Netherlands.
A substantial one-third of subjects turned out to have abnormal glucose tolerance according to World Health Organization criteria and an International Expert Committee Report (Diabetes Care. 2009 Jul;32[7]:1327-34). In a multivariate analysis, their 1-year rate of the primary study endpoint – target vessel failure, a composite of cardiac death, target vessel-related MI, or target vessel revascularization – was an adjusted 2.2 times greater than in the 788 normoglycemic patients.
Moreover, among the 7% of study participants who met diagnostic criteria for silent diabetes, the risk of target vessel failure was more than 4.4 times greater than in the normoglycemic group.
“To a very great extent, periprocedural MI is the driving force behind this difference that we saw. From a biological point of view, I think that the vulnerability of the vessel in the diabetic or prediabetic patient features more brittle plaque with a higher risk of cholesterol embolization, and with more plaque mass that can be pushed to the side so that side branch vessels can become occluded, leading to periprocedural MI,” he observed.
Glucose metabolism was assessed in all participants by two methods using the conventional cutoffs: a 2-hour oral glucose tolerance test (OGTT), and the combination of fasting plasma glucose and hemoglobin A1c. By OGTT, 7% of patients had silent, previously unrecognized diabetes and another 13% had prediabetes. Using the combination of fasting plasma glucose and HbA1c, a total of 25% of subjects had silent diabetes or prediabetes. Fully 33% of participants had abnormal glucose metabolism by one yardstick or the other.
“What we have seen is there is a group of patients that are missed with either. With the OGTT you don’t see all the diabetics, and with HbA1c and fasting blood glucose you also miss some patients,” said Dr. von Birgelen.
The 1-year cumulative incidence of target vessel failure was 13.2% in patients with silent diabetes as identified by the OGTT and 12.1% in those detected by the alternative method, compared with rates of 2.8% and 3.1%, respectively, in normoglycemic PCI patients. The event rate was 6.1% in patients with prediabetes by OGTT and similar at 5.5% in those found to be prediabetic based on fasting blood glucose and HbA1c, versus rates of 2.8% and 3.1%, respectively, in normoglycemic patients.
“The findings of this study suggest that post-PCI event risk associated with hyperglycemia is a continuum without a clear threshold effect, extending well beyond the threshold that currently defines diabetes,” Dr. von Birgelen said.
Once again, it’s worth emphasizing that the elevated target vessel failure rates seen in patients with abnormal glucose metabolism were due mostly to increased rates of acute MI within the first 24 hours after PCI. The target vessel–related MI rate was 10.3% in patients with silent diabetes, compared with just 1.8% in normoglycemic controls.
Asked what the take-home message for clinicians is from this study, he noted that the Netherlands has a relatively low prevalence of diabetes, and a highly developed primary care medicine system.
“We have a very good one-to-one relationship between the patient and the GP. So if we find 7% silent diabetes and up to one-third of patients with undetected abnormal glucose tolerance in a country with a relatively low prevalence of diabetes, you may expect that in other countries with a higher prevalence and perhaps a less developed primary care system the rate may be much, much higher,” Dr. von Birgelen cautioned.
The implications for the daily clinical practice of interventional cardiology are clear, he continued: “We’ve seen in several trials that the new stents are doing a fantastic job. So if we want to further improve the outcomes in our patients we have to do something else. We should look for subgroups of our PCI patients who have a particularly high risk. And we all realize that diabetics are such a problem, but I think we have shown that the prediabetic patients are also important. So we should identify and pretreat these patients, perhaps with aggressive lipid-lowering therapy during the weeks before a scheduled elective PCI.”
“There are data showing that with aggressive lipid-lowering you might reduce the risk of periprocedural MI,” the cardiologist noted.
As a practical matter, screening via fasting blood glucose and HbA1c is probably the way to go in clinical practice, according to Dr. von Birgelen.
“In this study, we performed the OGTT because it is still considered by many the gold standard. But there is increasing evidence favoring HbA1c data and fasting blood glucose,” he said.
Other possible pre-PCI interventions worthy of consideration in patients found to have previously unsuspected abnormal glucose tolerance might include medical therapy aimed at normalizing glucose metabolism, as well as perhaps resorting to the most potent forms of dual-antiplatelet therapy in patients with stable angina who have impaired glucose tolerance. However, these are possibilities that should be tested in randomized controlled trials before widespread adoption, he added.
The BIO-RESORT Silent Diabetes Study, which will continue for 5 years of post-PCI follow-up, is a prespecified substudy of the previously reported BIO-RESORT trial, which addressed another issue entirely. It was a three-arm, patient-blinded clinical trial comparing 1-year safety and efficacy outcomes in nearly 3,500 PCI patients randomized to PCI with very thin strut biodegradable polymer everolimus- or sirolimus-eluting stents or a durable polymer zotarolimus-eluting stent. Outcomes proved noninferior across the three treatment groups (Lancet. 2016 Nov 26;388[10060]:2607-17).
Dr. von Birgelen observed that the silent diabetes study broke new ground. Prior studies of PCI outcomes in patients with unrecognized diabetes were limited to recipients of plain old balloon angioplasty, bare metal, or first-generation drug-eluting stents. And studies of PCI in patients with unrecognized prediabetes are virtually nonexistent.
As the principal investigator for both the parent BIO-RESORT trial and the silent diabetes substudy, Dr. von Birgelen received research grants from Biotronik, Boston Scientific, and Medtronic, the cosponsors. He applauded the three companies for funding the silent diabetes substudy in the interest of science even though it had no commercial relevance to their stent businesses.
PARIS – Undetected diabetes and prediabetes are pervasive in patients undergoing percutaneous coronary intervention, and they’re associated with a sharply increased risk of major adverse cardiovascular events, according to the results of the potentially practice-changing BIO-RESORT Silent Diabetes Study, Clemens von Birgelen, MD, PhD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Our data support screening PCI all-comers for silent diabetes, which may help identify patients with an increased event risk and improve their therapy,” said Dr. von Birgelen, professor of cardiology at the Thoraxcentrum of Twente, a high-volume center for cardiac interventions in Enschede, the Netherlands.
A substantial one-third of subjects turned out to have abnormal glucose tolerance according to World Health Organization criteria and an International Expert Committee Report (Diabetes Care. 2009 Jul;32[7]:1327-34). In a multivariate analysis, their 1-year rate of the primary study endpoint – target vessel failure, a composite of cardiac death, target vessel-related MI, or target vessel revascularization – was an adjusted 2.2 times greater than in the 788 normoglycemic patients.
Moreover, among the 7% of study participants who met diagnostic criteria for silent diabetes, the risk of target vessel failure was more than 4.4 times greater than in the normoglycemic group.
“To a very great extent, periprocedural MI is the driving force behind this difference that we saw. From a biological point of view, I think that the vulnerability of the vessel in the diabetic or prediabetic patient features more brittle plaque with a higher risk of cholesterol embolization, and with more plaque mass that can be pushed to the side so that side branch vessels can become occluded, leading to periprocedural MI,” he observed.
Glucose metabolism was assessed in all participants by two methods using the conventional cutoffs: a 2-hour oral glucose tolerance test (OGTT), and the combination of fasting plasma glucose and hemoglobin A1c. By OGTT, 7% of patients had silent, previously unrecognized diabetes and another 13% had prediabetes. Using the combination of fasting plasma glucose and HbA1c, a total of 25% of subjects had silent diabetes or prediabetes. Fully 33% of participants had abnormal glucose metabolism by one yardstick or the other.
“What we have seen is there is a group of patients that are missed with either. With the OGTT you don’t see all the diabetics, and with HbA1c and fasting blood glucose you also miss some patients,” said Dr. von Birgelen.
The 1-year cumulative incidence of target vessel failure was 13.2% in patients with silent diabetes as identified by the OGTT and 12.1% in those detected by the alternative method, compared with rates of 2.8% and 3.1%, respectively, in normoglycemic PCI patients. The event rate was 6.1% in patients with prediabetes by OGTT and similar at 5.5% in those found to be prediabetic based on fasting blood glucose and HbA1c, versus rates of 2.8% and 3.1%, respectively, in normoglycemic patients.
“The findings of this study suggest that post-PCI event risk associated with hyperglycemia is a continuum without a clear threshold effect, extending well beyond the threshold that currently defines diabetes,” Dr. von Birgelen said.
Once again, it’s worth emphasizing that the elevated target vessel failure rates seen in patients with abnormal glucose metabolism were due mostly to increased rates of acute MI within the first 24 hours after PCI. The target vessel–related MI rate was 10.3% in patients with silent diabetes, compared with just 1.8% in normoglycemic controls.
Asked what the take-home message for clinicians is from this study, he noted that the Netherlands has a relatively low prevalence of diabetes, and a highly developed primary care medicine system.
“We have a very good one-to-one relationship between the patient and the GP. So if we find 7% silent diabetes and up to one-third of patients with undetected abnormal glucose tolerance in a country with a relatively low prevalence of diabetes, you may expect that in other countries with a higher prevalence and perhaps a less developed primary care system the rate may be much, much higher,” Dr. von Birgelen cautioned.
The implications for the daily clinical practice of interventional cardiology are clear, he continued: “We’ve seen in several trials that the new stents are doing a fantastic job. So if we want to further improve the outcomes in our patients we have to do something else. We should look for subgroups of our PCI patients who have a particularly high risk. And we all realize that diabetics are such a problem, but I think we have shown that the prediabetic patients are also important. So we should identify and pretreat these patients, perhaps with aggressive lipid-lowering therapy during the weeks before a scheduled elective PCI.”
“There are data showing that with aggressive lipid-lowering you might reduce the risk of periprocedural MI,” the cardiologist noted.
As a practical matter, screening via fasting blood glucose and HbA1c is probably the way to go in clinical practice, according to Dr. von Birgelen.
“In this study, we performed the OGTT because it is still considered by many the gold standard. But there is increasing evidence favoring HbA1c data and fasting blood glucose,” he said.
Other possible pre-PCI interventions worthy of consideration in patients found to have previously unsuspected abnormal glucose tolerance might include medical therapy aimed at normalizing glucose metabolism, as well as perhaps resorting to the most potent forms of dual-antiplatelet therapy in patients with stable angina who have impaired glucose tolerance. However, these are possibilities that should be tested in randomized controlled trials before widespread adoption, he added.
The BIO-RESORT Silent Diabetes Study, which will continue for 5 years of post-PCI follow-up, is a prespecified substudy of the previously reported BIO-RESORT trial, which addressed another issue entirely. It was a three-arm, patient-blinded clinical trial comparing 1-year safety and efficacy outcomes in nearly 3,500 PCI patients randomized to PCI with very thin strut biodegradable polymer everolimus- or sirolimus-eluting stents or a durable polymer zotarolimus-eluting stent. Outcomes proved noninferior across the three treatment groups (Lancet. 2016 Nov 26;388[10060]:2607-17).
Dr. von Birgelen observed that the silent diabetes study broke new ground. Prior studies of PCI outcomes in patients with unrecognized diabetes were limited to recipients of plain old balloon angioplasty, bare metal, or first-generation drug-eluting stents. And studies of PCI in patients with unrecognized prediabetes are virtually nonexistent.
As the principal investigator for both the parent BIO-RESORT trial and the silent diabetes substudy, Dr. von Birgelen received research grants from Biotronik, Boston Scientific, and Medtronic, the cosponsors. He applauded the three companies for funding the silent diabetes substudy in the interest of science even though it had no commercial relevance to their stent businesses.
AT EUROPCR
Key clinical point:
Major finding: One-third of patients undergoing PCI have unsuspected silent diabetes or prediabetes, placing them at increased risk for major adverse cardiac events.
Data source: This prospective observational study included 988 patients not known to have diabetes who underwent screening for abnormal glucose tolerance 6 weeks after PCI with stenting.
Disclosures: The study was cosponsored by Biotronik, Boston Scientific, and Medtronic.
Public favors Obamacare over Trumpcare
The American Health Care Act, the House Republicans’ replacement for the Affordable Care Act, is currently viewed less favorably than its predecessor, according to a new poll by the Kaiser Family Foundation.
In the survey, 40% of respondents said that they had a “very unfavorable” opinion of the AHCA, compared with 29% for the ACA. The “very favorable” opinions also favored the ACA: 29% to 12%, according to a Kaiser report released May 31.
The Kaiser Health Tracking Poll involved 1,205 adults and was conducted May 16-22, 2017.
The American Health Care Act, the House Republicans’ replacement for the Affordable Care Act, is currently viewed less favorably than its predecessor, according to a new poll by the Kaiser Family Foundation.
In the survey, 40% of respondents said that they had a “very unfavorable” opinion of the AHCA, compared with 29% for the ACA. The “very favorable” opinions also favored the ACA: 29% to 12%, according to a Kaiser report released May 31.
The Kaiser Health Tracking Poll involved 1,205 adults and was conducted May 16-22, 2017.
The American Health Care Act, the House Republicans’ replacement for the Affordable Care Act, is currently viewed less favorably than its predecessor, according to a new poll by the Kaiser Family Foundation.
In the survey, 40% of respondents said that they had a “very unfavorable” opinion of the AHCA, compared with 29% for the ACA. The “very favorable” opinions also favored the ACA: 29% to 12%, according to a Kaiser report released May 31.
The Kaiser Health Tracking Poll involved 1,205 adults and was conducted May 16-22, 2017.
Distance from transplant center predicted mortality in chronic liver disease
Living more than 150 miles from a liver transplant center was associated with a higher risk of mortality among patients with chronic liver failure, regardless of etiology, transplantation status, or whether patients had decompensated cirrhosis or hepatocellular carcinoma, according to a first-in-kind, population-based study reported in the June issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2017.02.023).
The findings underscore the need for accessible, specialized liver care irrespective of whether patients with chronic liver failure (CLF) are destined for transplantation, David S. Goldberg, MD, of the University of Pennsylvania, Philadelphia, wrote with his associates. The associations “do not provide cause and effect,” but underscore the need to consider “the broader impact of transplant-related policies that could decrease transplant volumes and threaten closures of smaller liver transplant centers that serve geographically isolated populations in the Southeast and Midwest,” they added.
A total of 879 (5.2%) patients lived more than 150 miles from the nearest liver transplant center, the analysis showed. Even after controlling for etiology of liver disease, this subgroup was at significantly greater risk of mortality (hazard ratio, 1.2; 95% confidence interval, 1.1-1.3; P less than .001) and of dying without undergoing transplantation (HR, 1.2; 95% CI, 1.1-1.3; P = .003) than were patients who were less geographically isolated. Distance from a transplant center also predicted overall and transplant-free mortality when modeled as a continuous variable, with hazard ratios of 1.02 (P = .02) and 1.03 (P = .04), respectively. “Although patients living more than 150 miles from a liver transplant center had fewer outpatient gastroenterologist visits, this covariate did not affect the final models,” the investigators reported. Rural locality did not predict mortality after controlling for distance from a transplant center, and neither did living in a low-income zip code, they added.
Data from the Centers for Disease Control and Prevention indicate that age-adjusted rates of death from liver disease are lowest in New York, where the entire population lives within 150 miles of a liver transplant center, the researchers noted. “By contrast, New Mexico and Wyoming have the highest age-adjusted death rates, and more than 95% of those states’ populations live more than 150 miles from a [transplant] center,” they emphasized. “The management of most patients with CLF is not centered on transplantation, but rather the spectrum of care for decompensated cirrhosis and hepatocellular carcinoma. Thus, maintaining access to specialized liver care is important for patients with CLF.”
Dr. Goldberg received support from the National Institutes of Health. The investigators had no conflicts.
Living more than 150 miles from a liver transplant center was associated with a higher risk of mortality among patients with chronic liver failure, regardless of etiology, transplantation status, or whether patients had decompensated cirrhosis or hepatocellular carcinoma, according to a first-in-kind, population-based study reported in the June issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2017.02.023).
The findings underscore the need for accessible, specialized liver care irrespective of whether patients with chronic liver failure (CLF) are destined for transplantation, David S. Goldberg, MD, of the University of Pennsylvania, Philadelphia, wrote with his associates. The associations “do not provide cause and effect,” but underscore the need to consider “the broader impact of transplant-related policies that could decrease transplant volumes and threaten closures of smaller liver transplant centers that serve geographically isolated populations in the Southeast and Midwest,” they added.
A total of 879 (5.2%) patients lived more than 150 miles from the nearest liver transplant center, the analysis showed. Even after controlling for etiology of liver disease, this subgroup was at significantly greater risk of mortality (hazard ratio, 1.2; 95% confidence interval, 1.1-1.3; P less than .001) and of dying without undergoing transplantation (HR, 1.2; 95% CI, 1.1-1.3; P = .003) than were patients who were less geographically isolated. Distance from a transplant center also predicted overall and transplant-free mortality when modeled as a continuous variable, with hazard ratios of 1.02 (P = .02) and 1.03 (P = .04), respectively. “Although patients living more than 150 miles from a liver transplant center had fewer outpatient gastroenterologist visits, this covariate did not affect the final models,” the investigators reported. Rural locality did not predict mortality after controlling for distance from a transplant center, and neither did living in a low-income zip code, they added.
Data from the Centers for Disease Control and Prevention indicate that age-adjusted rates of death from liver disease are lowest in New York, where the entire population lives within 150 miles of a liver transplant center, the researchers noted. “By contrast, New Mexico and Wyoming have the highest age-adjusted death rates, and more than 95% of those states’ populations live more than 150 miles from a [transplant] center,” they emphasized. “The management of most patients with CLF is not centered on transplantation, but rather the spectrum of care for decompensated cirrhosis and hepatocellular carcinoma. Thus, maintaining access to specialized liver care is important for patients with CLF.”
Dr. Goldberg received support from the National Institutes of Health. The investigators had no conflicts.
Living more than 150 miles from a liver transplant center was associated with a higher risk of mortality among patients with chronic liver failure, regardless of etiology, transplantation status, or whether patients had decompensated cirrhosis or hepatocellular carcinoma, according to a first-in-kind, population-based study reported in the June issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2017.02.023).
The findings underscore the need for accessible, specialized liver care irrespective of whether patients with chronic liver failure (CLF) are destined for transplantation, David S. Goldberg, MD, of the University of Pennsylvania, Philadelphia, wrote with his associates. The associations “do not provide cause and effect,” but underscore the need to consider “the broader impact of transplant-related policies that could decrease transplant volumes and threaten closures of smaller liver transplant centers that serve geographically isolated populations in the Southeast and Midwest,” they added.
A total of 879 (5.2%) patients lived more than 150 miles from the nearest liver transplant center, the analysis showed. Even after controlling for etiology of liver disease, this subgroup was at significantly greater risk of mortality (hazard ratio, 1.2; 95% confidence interval, 1.1-1.3; P less than .001) and of dying without undergoing transplantation (HR, 1.2; 95% CI, 1.1-1.3; P = .003) than were patients who were less geographically isolated. Distance from a transplant center also predicted overall and transplant-free mortality when modeled as a continuous variable, with hazard ratios of 1.02 (P = .02) and 1.03 (P = .04), respectively. “Although patients living more than 150 miles from a liver transplant center had fewer outpatient gastroenterologist visits, this covariate did not affect the final models,” the investigators reported. Rural locality did not predict mortality after controlling for distance from a transplant center, and neither did living in a low-income zip code, they added.
Data from the Centers for Disease Control and Prevention indicate that age-adjusted rates of death from liver disease are lowest in New York, where the entire population lives within 150 miles of a liver transplant center, the researchers noted. “By contrast, New Mexico and Wyoming have the highest age-adjusted death rates, and more than 95% of those states’ populations live more than 150 miles from a [transplant] center,” they emphasized. “The management of most patients with CLF is not centered on transplantation, but rather the spectrum of care for decompensated cirrhosis and hepatocellular carcinoma. Thus, maintaining access to specialized liver care is important for patients with CLF.”
Dr. Goldberg received support from the National Institutes of Health. The investigators had no conflicts.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Geographic isolation from a liver transplant center independently predicted mortality among patients with chronic liver failure.
Major finding: In adjusted analyses, patients who lived more than 150 miles from a liver transplant center were at significantly greater risk of mortality (HR, 1.2; 95% CI, 1.1-1.3; P less than .001) and of dying without undergoing transplantation (HR, 1.2; 95% CI, 1.1-1.3; P = .003) than were patients who were less geographically isolated.
Data source: A retrospective cohort study of 16,824 patients with chronic liver failure who were included in the Healthcare Integrated Research Database between 2006 and 2014.
Disclosures: Dr. Goldberg received support from the National Institutes of Health. The investigators had no conflicts.
Large-scale ERAS program reduces postoperative LOS, complications
Enhanced Recovery After Surgery (ERAS), a program implemented by Kaiser Permanente Northern California – a multihospital integrated health system – significantly reduced length of stay and complication rates, according to a report published in JAMA Surgery.
Beginning in 2014, when the ERAS program was implemented in 20 Kaiser hospitals, progress was made on the goal of improving inpatient safety, as well as improvements in-hospital mortality, rates of early ambulation, patient nutrition, and reduced opioid use, said Vincent X. Liu, MD, of the division of research, Kaiser Permanente Oakland, and his associates. Those outcomes were studied in the context of a similar group of patients in other, non-ERAS hospitals to determine the degree of change in each area.
ERAS aimed to reduce opioid use by encouraging multimodal analgesia, which included pre- and postoperative IV acetaminophen and NSAIDs, perioperative IV lidocaine, or peripheral nerve blocks. It encouraged ambulation within 12 hours of surgery completion and a daily goal of walking at least 21 feet during the first 3 postoperative days.
The program enhanced patient nutrition by reducing prolonged preoperative fasting, providing a high-carbohydrate beverage 2-4 hours before surgery, and allowing solids 8-12 hours before surgery. It also provided food within 12 hours of completing surgery. ERAS also encouraged patient engagement in care by use of educational materials and a calendar that detailed what the care process would entail. For clinicians, ERAS provided new electronic tools such as electronic medical record order sets to facilitate standardized practice.
In the first phase of their study, Dr. Liu and his associates assessed changes over time in patient safety outcomes among 3,768 patients undergoing elective colorectal resection and 5,002 undergoing emergency hip fracture repair.
Hospital length of stay decreased significantly after implementation of ERAS, from 5.1 to 4.2 days in the colorectal resection group and from 3.6 to 3.2 days in the hip fracture group. Complication rates decreased from 18.1% to 14.7% and from 30.8% to 24.9%, respectively. Early ambulation rates increased substantially, from 22.3% to 56.5% and from 2.8% to 21.2%, respectively.
The rate of improved nutrition rose from 13.0% to 39.2% in the colorectal resection group and from 45.6% to 57.1% in the hip repair group. And the total dose of morphine equivalents dropped from 52.4 to 30.6 and from 38.9 to 27.0, respectively (JAMA Surg. 2017 May 10. doi: 10.1001/jamasurg.2017.1032).
In the second phase of the study, the investigators compared these changes against the outcomes of two comparator groups who underwent similar surgeries (5,556 resection comparators and 1,523 hip repair comparators) during the same time frame but in hospitals that did not implement the ERAS program.
In this analysis, LOS was significantly shorter and complication rates were significantly lower for both procedures at the hospitals where the intervention was implemented, compared with the other hospitals. In-hospital mortality, opioid use, early ambulation, and discharge to home rather than a rehabilitation facility also favored the intervention groups.
“This study demonstrates the effectiveness of a systems-level approach to ERAS program implementation, even across widely divergent target populations,” Dr. Liu and his associates said.
The Gordon and Betty Moore Foundation, the Permanente Medical Group, the Kaiser Foundation Health Plan, and the National Institutes of Health funded the study. Dr. Liu and his associates reported having no relevant financial disclosures.
Findings from Liu et al. have clinical, research, and policy relevance. First, they went beyond select surgical procedures from single hospitals. The investigators have robustly taken implementation science to the next level, thus showing that thoughtfully planned quality-improvement endeavors that are integrated with robust research evaluation measures can positively affect our surgical patients. In a similar vein, these results underscore the value proposition of research conducted in large health care systems that goes beyond the limitation of traditional stand-alone hospitals, such as small sample size and referral and practice biases. [In addition,] this investigation raises many and exciting future research opportunities to an eager audience of stakeholders. What are the cost implications of such efforts? How can we better leverage electronic health records with smart tools to better implement and measure the effects of the ERAS program and other quality and safety initiatives? [We also] need to be mindful of its unintended consequences on vulnerable populations and financially strained hospitals.
Mohammed Bayasi, MD, FACS, and Waddah Al-Refaie, MD, FACS, are with the department of surgery, MedStar Georgetown University Hospital, Washington. Their comments are from an editorial (JAMA Surg. 2017 May 10. doi: 10.1001/jamasurg.2017.1051). They had no disclosures.
Findings from Liu et al. have clinical, research, and policy relevance. First, they went beyond select surgical procedures from single hospitals. The investigators have robustly taken implementation science to the next level, thus showing that thoughtfully planned quality-improvement endeavors that are integrated with robust research evaluation measures can positively affect our surgical patients. In a similar vein, these results underscore the value proposition of research conducted in large health care systems that goes beyond the limitation of traditional stand-alone hospitals, such as small sample size and referral and practice biases. [In addition,] this investigation raises many and exciting future research opportunities to an eager audience of stakeholders. What are the cost implications of such efforts? How can we better leverage electronic health records with smart tools to better implement and measure the effects of the ERAS program and other quality and safety initiatives? [We also] need to be mindful of its unintended consequences on vulnerable populations and financially strained hospitals.
Mohammed Bayasi, MD, FACS, and Waddah Al-Refaie, MD, FACS, are with the department of surgery, MedStar Georgetown University Hospital, Washington. Their comments are from an editorial (JAMA Surg. 2017 May 10. doi: 10.1001/jamasurg.2017.1051). They had no disclosures.
Findings from Liu et al. have clinical, research, and policy relevance. First, they went beyond select surgical procedures from single hospitals. The investigators have robustly taken implementation science to the next level, thus showing that thoughtfully planned quality-improvement endeavors that are integrated with robust research evaluation measures can positively affect our surgical patients. In a similar vein, these results underscore the value proposition of research conducted in large health care systems that goes beyond the limitation of traditional stand-alone hospitals, such as small sample size and referral and practice biases. [In addition,] this investigation raises many and exciting future research opportunities to an eager audience of stakeholders. What are the cost implications of such efforts? How can we better leverage electronic health records with smart tools to better implement and measure the effects of the ERAS program and other quality and safety initiatives? [We also] need to be mindful of its unintended consequences on vulnerable populations and financially strained hospitals.
Mohammed Bayasi, MD, FACS, and Waddah Al-Refaie, MD, FACS, are with the department of surgery, MedStar Georgetown University Hospital, Washington. Their comments are from an editorial (JAMA Surg. 2017 May 10. doi: 10.1001/jamasurg.2017.1051). They had no disclosures.
Enhanced Recovery After Surgery (ERAS), a program implemented by Kaiser Permanente Northern California – a multihospital integrated health system – significantly reduced length of stay and complication rates, according to a report published in JAMA Surgery.
Beginning in 2014, when the ERAS program was implemented in 20 Kaiser hospitals, progress was made on the goal of improving inpatient safety, as well as improvements in-hospital mortality, rates of early ambulation, patient nutrition, and reduced opioid use, said Vincent X. Liu, MD, of the division of research, Kaiser Permanente Oakland, and his associates. Those outcomes were studied in the context of a similar group of patients in other, non-ERAS hospitals to determine the degree of change in each area.
ERAS aimed to reduce opioid use by encouraging multimodal analgesia, which included pre- and postoperative IV acetaminophen and NSAIDs, perioperative IV lidocaine, or peripheral nerve blocks. It encouraged ambulation within 12 hours of surgery completion and a daily goal of walking at least 21 feet during the first 3 postoperative days.
The program enhanced patient nutrition by reducing prolonged preoperative fasting, providing a high-carbohydrate beverage 2-4 hours before surgery, and allowing solids 8-12 hours before surgery. It also provided food within 12 hours of completing surgery. ERAS also encouraged patient engagement in care by use of educational materials and a calendar that detailed what the care process would entail. For clinicians, ERAS provided new electronic tools such as electronic medical record order sets to facilitate standardized practice.
In the first phase of their study, Dr. Liu and his associates assessed changes over time in patient safety outcomes among 3,768 patients undergoing elective colorectal resection and 5,002 undergoing emergency hip fracture repair.
Hospital length of stay decreased significantly after implementation of ERAS, from 5.1 to 4.2 days in the colorectal resection group and from 3.6 to 3.2 days in the hip fracture group. Complication rates decreased from 18.1% to 14.7% and from 30.8% to 24.9%, respectively. Early ambulation rates increased substantially, from 22.3% to 56.5% and from 2.8% to 21.2%, respectively.
The rate of improved nutrition rose from 13.0% to 39.2% in the colorectal resection group and from 45.6% to 57.1% in the hip repair group. And the total dose of morphine equivalents dropped from 52.4 to 30.6 and from 38.9 to 27.0, respectively (JAMA Surg. 2017 May 10. doi: 10.1001/jamasurg.2017.1032).
In the second phase of the study, the investigators compared these changes against the outcomes of two comparator groups who underwent similar surgeries (5,556 resection comparators and 1,523 hip repair comparators) during the same time frame but in hospitals that did not implement the ERAS program.
In this analysis, LOS was significantly shorter and complication rates were significantly lower for both procedures at the hospitals where the intervention was implemented, compared with the other hospitals. In-hospital mortality, opioid use, early ambulation, and discharge to home rather than a rehabilitation facility also favored the intervention groups.
“This study demonstrates the effectiveness of a systems-level approach to ERAS program implementation, even across widely divergent target populations,” Dr. Liu and his associates said.
The Gordon and Betty Moore Foundation, the Permanente Medical Group, the Kaiser Foundation Health Plan, and the National Institutes of Health funded the study. Dr. Liu and his associates reported having no relevant financial disclosures.
Enhanced Recovery After Surgery (ERAS), a program implemented by Kaiser Permanente Northern California – a multihospital integrated health system – significantly reduced length of stay and complication rates, according to a report published in JAMA Surgery.
Beginning in 2014, when the ERAS program was implemented in 20 Kaiser hospitals, progress was made on the goal of improving inpatient safety, as well as improvements in-hospital mortality, rates of early ambulation, patient nutrition, and reduced opioid use, said Vincent X. Liu, MD, of the division of research, Kaiser Permanente Oakland, and his associates. Those outcomes were studied in the context of a similar group of patients in other, non-ERAS hospitals to determine the degree of change in each area.
ERAS aimed to reduce opioid use by encouraging multimodal analgesia, which included pre- and postoperative IV acetaminophen and NSAIDs, perioperative IV lidocaine, or peripheral nerve blocks. It encouraged ambulation within 12 hours of surgery completion and a daily goal of walking at least 21 feet during the first 3 postoperative days.
The program enhanced patient nutrition by reducing prolonged preoperative fasting, providing a high-carbohydrate beverage 2-4 hours before surgery, and allowing solids 8-12 hours before surgery. It also provided food within 12 hours of completing surgery. ERAS also encouraged patient engagement in care by use of educational materials and a calendar that detailed what the care process would entail. For clinicians, ERAS provided new electronic tools such as electronic medical record order sets to facilitate standardized practice.
In the first phase of their study, Dr. Liu and his associates assessed changes over time in patient safety outcomes among 3,768 patients undergoing elective colorectal resection and 5,002 undergoing emergency hip fracture repair.
Hospital length of stay decreased significantly after implementation of ERAS, from 5.1 to 4.2 days in the colorectal resection group and from 3.6 to 3.2 days in the hip fracture group. Complication rates decreased from 18.1% to 14.7% and from 30.8% to 24.9%, respectively. Early ambulation rates increased substantially, from 22.3% to 56.5% and from 2.8% to 21.2%, respectively.
The rate of improved nutrition rose from 13.0% to 39.2% in the colorectal resection group and from 45.6% to 57.1% in the hip repair group. And the total dose of morphine equivalents dropped from 52.4 to 30.6 and from 38.9 to 27.0, respectively (JAMA Surg. 2017 May 10. doi: 10.1001/jamasurg.2017.1032).
In the second phase of the study, the investigators compared these changes against the outcomes of two comparator groups who underwent similar surgeries (5,556 resection comparators and 1,523 hip repair comparators) during the same time frame but in hospitals that did not implement the ERAS program.
In this analysis, LOS was significantly shorter and complication rates were significantly lower for both procedures at the hospitals where the intervention was implemented, compared with the other hospitals. In-hospital mortality, opioid use, early ambulation, and discharge to home rather than a rehabilitation facility also favored the intervention groups.
“This study demonstrates the effectiveness of a systems-level approach to ERAS program implementation, even across widely divergent target populations,” Dr. Liu and his associates said.
The Gordon and Betty Moore Foundation, the Permanente Medical Group, the Kaiser Foundation Health Plan, and the National Institutes of Health funded the study. Dr. Liu and his associates reported having no relevant financial disclosures.
FROM JAMA SURGERY
Key clinical point: An Enhanced Recovery After Surgery program aimed at improving inpatient safety significantly reduced length of stay and complication rates at 20 California hospitals.
Major finding: After the ERAS program was implemented, hospital LOS decreased from 5.1 to 4.2 days in the colorectal resection group and from 3.6 to 3.2 days in the hip fracture group, and complication rates decreased from 18.1% to 14.7% and from 30.8% to 24.9%, respectively.
Data source: A “pre-post” comparison study of patients’ safety outcomes after implementation of an ERAS program, which involved 15,849 surgical patients at 20 hospitals.
Disclosures: The Gordon and Betty Moore Foundation, the Permanente Medical Group, the Kaiser Foundation Health Plan, and the National Institutes of Health funded the study. Dr. Liu and his associates reported having no relevant financial disclosures.
Gastric bands hit with high reoperation rates, rising costs
About one in five laparoscopic gastric band surgeries result in device-related reoperations and reoperations account for almost half of all Medicare expenditures for gastric band surgery, a large retrospective study has found.
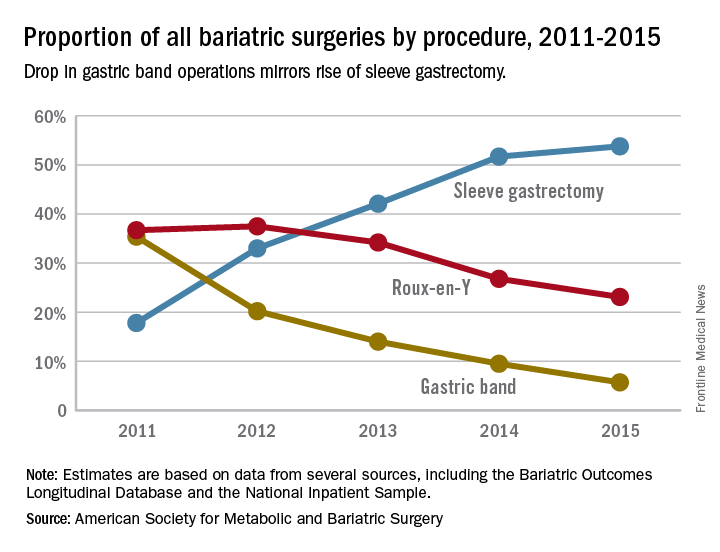
The laparoscopic adjustable gastric band for treatment of morbid obesity, approved in 2001 by the Food and Drug Administration, was once a common choice for bariatric patients. Although its use has declined from in recent years, the American Society for Metabolic and Bariatric Surgery estimated that 11,000 bands were placed in 2015 and many others remain in place (ASMBS, Estimate of bariatric surgery numbers, 2011-2015, https://goo.gl/f8iByl). Many of these gastric bands will need to be removed, replaced, or revised in a series of procedures over the coming years.
Of the 24,042 gastric band patients in this study group, 4,636 (18.5%) underwent reoperation, defined as band removal, band replacement, or revision to a different bariatric procedure, but not including band size adjustment. Patients who had reoperations were more likely to be women, to be white, and to have slightly lower rates of hypertension and diabetes. But they were also more likely to have received a psychiatric, anemia, or electrolyte disorder diagnosis at the time of their index operations.
Among the 4,636 patients who had reoperations, 17,539 such procedures were performed, an average of 3.8 procedures per patient, in addition to the index operation, over an average follow-up of 4.5 years. The most common reoperation was for band removal (41.8%). Other reasons included conversion to laparoscopic Roux-en-Y gastric bypass (13.1%) or laparoscopic sleeve gastrectomy (5.3%).
The study also looked at the regional differences, reflecting the comparative success of some programs in managing laparoscopic gastric band placement. Reoperation rates across the referral hospitals ranged from 5% to 95.5%, The study found a nearly a threefold variation in reoperation rates across geographic regions. The bottom quartile of hospital referral regions had an average reoperation rate of 13.3% (0.3 standard deviation) and the top quartile had an average reoperation rate of 39.1% (0.21 SD). Top-quartile regions were concentrated in the West, but were otherwise distributed throughout the country.
Most reoperations were elective admissions (79.9%), while 10% were classified as urgent and another 10.1% as emergency. So although previous studies have documented complications such as band slippage and gastric erosion, the preponderance of elective admissions suggests patient and clinician preferences, or weight loss failure, rather than emergency situations, may be the driving force in the reoperation trend.
The investigators concluded that patients should be fully informed about the likelihood of reoperation with the gastric band. In addition, the wide range of reoperation rates across regions and institutions suggests that more training or better patient selection may be needed to improve outcomes. However, they suggested that “taken together, these findings indicate that the gastric band is associated with high reoperation rates and considerable costs to the payers, which raises concerns about its safety, effectiveness, and value.” They added that “payers should reconsider their coverage of the gastric band device.”
Coauthor Justin B. Dimick, MD, disclosed a financial interest in ArborMetrix. The other coauthors reported having no financial disclosures. The Robert Wood Johnson Foundation, U.S. Department of Veterans Affairs, National Institute on Aging, and National Institute of Diabetes and Digestive and Kidney Diseases provided funding.
Dr. Ibrahim and his colleagues have suggested that payers reconsider covering the adjustable laparoscopic gastric band. I disagree and feel that this device still has a role, albeit limited in the modern bariatric surgical program. Many patients do well for a long period. A committed surgeon and program, and the ideal patient with a similar level of commitment, are needed to achieve these best outcomes. Now that patients and surgeons are better informed of the drawbacks to the device, use has decreased without external regulations or policies to drive this change. No single bariatric procedure is appropriate for all patients. Patients need options, and we need better data to help guide their decisions. Do not throw the baby out with the bathwater.
Jon C. Gould, MD, FACS, is with the Medical College of Wisconsin, Milwaukee. Dr. Gould made these comments in an editorial (JAMA Surg. 2017 May 17; doi: 10.1001/jamasurg.2017.1082) that accompanied the study. He has no disclosures.
Dr. Ibrahim and his colleagues have suggested that payers reconsider covering the adjustable laparoscopic gastric band. I disagree and feel that this device still has a role, albeit limited in the modern bariatric surgical program. Many patients do well for a long period. A committed surgeon and program, and the ideal patient with a similar level of commitment, are needed to achieve these best outcomes. Now that patients and surgeons are better informed of the drawbacks to the device, use has decreased without external regulations or policies to drive this change. No single bariatric procedure is appropriate for all patients. Patients need options, and we need better data to help guide their decisions. Do not throw the baby out with the bathwater.
Jon C. Gould, MD, FACS, is with the Medical College of Wisconsin, Milwaukee. Dr. Gould made these comments in an editorial (JAMA Surg. 2017 May 17; doi: 10.1001/jamasurg.2017.1082) that accompanied the study. He has no disclosures.
Dr. Ibrahim and his colleagues have suggested that payers reconsider covering the adjustable laparoscopic gastric band. I disagree and feel that this device still has a role, albeit limited in the modern bariatric surgical program. Many patients do well for a long period. A committed surgeon and program, and the ideal patient with a similar level of commitment, are needed to achieve these best outcomes. Now that patients and surgeons are better informed of the drawbacks to the device, use has decreased without external regulations or policies to drive this change. No single bariatric procedure is appropriate for all patients. Patients need options, and we need better data to help guide their decisions. Do not throw the baby out with the bathwater.
Jon C. Gould, MD, FACS, is with the Medical College of Wisconsin, Milwaukee. Dr. Gould made these comments in an editorial (JAMA Surg. 2017 May 17; doi: 10.1001/jamasurg.2017.1082) that accompanied the study. He has no disclosures.
About one in five laparoscopic gastric band surgeries result in device-related reoperations and reoperations account for almost half of all Medicare expenditures for gastric band surgery, a large retrospective study has found.
The laparoscopic adjustable gastric band for treatment of morbid obesity, approved in 2001 by the Food and Drug Administration, was once a common choice for bariatric patients. Although its use has declined from in recent years, the American Society for Metabolic and Bariatric Surgery estimated that 11,000 bands were placed in 2015 and many others remain in place (ASMBS, Estimate of bariatric surgery numbers, 2011-2015, https://goo.gl/f8iByl). Many of these gastric bands will need to be removed, replaced, or revised in a series of procedures over the coming years.
Of the 24,042 gastric band patients in this study group, 4,636 (18.5%) underwent reoperation, defined as band removal, band replacement, or revision to a different bariatric procedure, but not including band size adjustment. Patients who had reoperations were more likely to be women, to be white, and to have slightly lower rates of hypertension and diabetes. But they were also more likely to have received a psychiatric, anemia, or electrolyte disorder diagnosis at the time of their index operations.
Among the 4,636 patients who had reoperations, 17,539 such procedures were performed, an average of 3.8 procedures per patient, in addition to the index operation, over an average follow-up of 4.5 years. The most common reoperation was for band removal (41.8%). Other reasons included conversion to laparoscopic Roux-en-Y gastric bypass (13.1%) or laparoscopic sleeve gastrectomy (5.3%).

The study also looked at the regional differences, reflecting the comparative success of some programs in managing laparoscopic gastric band placement. Reoperation rates across the referral hospitals ranged from 5% to 95.5%, The study found a nearly a threefold variation in reoperation rates across geographic regions. The bottom quartile of hospital referral regions had an average reoperation rate of 13.3% (0.3 standard deviation) and the top quartile had an average reoperation rate of 39.1% (0.21 SD). Top-quartile regions were concentrated in the West, but were otherwise distributed throughout the country.
Most reoperations were elective admissions (79.9%), while 10% were classified as urgent and another 10.1% as emergency. So although previous studies have documented complications such as band slippage and gastric erosion, the preponderance of elective admissions suggests patient and clinician preferences, or weight loss failure, rather than emergency situations, may be the driving force in the reoperation trend.
The investigators concluded that patients should be fully informed about the likelihood of reoperation with the gastric band. In addition, the wide range of reoperation rates across regions and institutions suggests that more training or better patient selection may be needed to improve outcomes. However, they suggested that “taken together, these findings indicate that the gastric band is associated with high reoperation rates and considerable costs to the payers, which raises concerns about its safety, effectiveness, and value.” They added that “payers should reconsider their coverage of the gastric band device.”
Coauthor Justin B. Dimick, MD, disclosed a financial interest in ArborMetrix. The other coauthors reported having no financial disclosures. The Robert Wood Johnson Foundation, U.S. Department of Veterans Affairs, National Institute on Aging, and National Institute of Diabetes and Digestive and Kidney Diseases provided funding.
About one in five laparoscopic gastric band surgeries result in device-related reoperations and reoperations account for almost half of all Medicare expenditures for gastric band surgery, a large retrospective study has found.
The laparoscopic adjustable gastric band for treatment of morbid obesity, approved in 2001 by the Food and Drug Administration, was once a common choice for bariatric patients. Although its use has declined from in recent years, the American Society for Metabolic and Bariatric Surgery estimated that 11,000 bands were placed in 2015 and many others remain in place (ASMBS, Estimate of bariatric surgery numbers, 2011-2015, https://goo.gl/f8iByl). Many of these gastric bands will need to be removed, replaced, or revised in a series of procedures over the coming years.
Of the 24,042 gastric band patients in this study group, 4,636 (18.5%) underwent reoperation, defined as band removal, band replacement, or revision to a different bariatric procedure, but not including band size adjustment. Patients who had reoperations were more likely to be women, to be white, and to have slightly lower rates of hypertension and diabetes. But they were also more likely to have received a psychiatric, anemia, or electrolyte disorder diagnosis at the time of their index operations.
Among the 4,636 patients who had reoperations, 17,539 such procedures were performed, an average of 3.8 procedures per patient, in addition to the index operation, over an average follow-up of 4.5 years. The most common reoperation was for band removal (41.8%). Other reasons included conversion to laparoscopic Roux-en-Y gastric bypass (13.1%) or laparoscopic sleeve gastrectomy (5.3%).

The study also looked at the regional differences, reflecting the comparative success of some programs in managing laparoscopic gastric band placement. Reoperation rates across the referral hospitals ranged from 5% to 95.5%, The study found a nearly a threefold variation in reoperation rates across geographic regions. The bottom quartile of hospital referral regions had an average reoperation rate of 13.3% (0.3 standard deviation) and the top quartile had an average reoperation rate of 39.1% (0.21 SD). Top-quartile regions were concentrated in the West, but were otherwise distributed throughout the country.
Most reoperations were elective admissions (79.9%), while 10% were classified as urgent and another 10.1% as emergency. So although previous studies have documented complications such as band slippage and gastric erosion, the preponderance of elective admissions suggests patient and clinician preferences, or weight loss failure, rather than emergency situations, may be the driving force in the reoperation trend.
The investigators concluded that patients should be fully informed about the likelihood of reoperation with the gastric band. In addition, the wide range of reoperation rates across regions and institutions suggests that more training or better patient selection may be needed to improve outcomes. However, they suggested that “taken together, these findings indicate that the gastric band is associated with high reoperation rates and considerable costs to the payers, which raises concerns about its safety, effectiveness, and value.” They added that “payers should reconsider their coverage of the gastric band device.”
Coauthor Justin B. Dimick, MD, disclosed a financial interest in ArborMetrix. The other coauthors reported having no financial disclosures. The Robert Wood Johnson Foundation, U.S. Department of Veterans Affairs, National Institute on Aging, and National Institute of Diabetes and Digestive and Kidney Diseases provided funding.
FROM JAMA SURGERY
Key clinical point: Reoperations after gastric band placement are common and raise concerns about the safety, effectiveness, and value of the device.
Major finding: During the study period, reoperations accounted for 47.6% of Medicare payments for laparoscopic gastric band procedures.
Data source: Medicare Provider Analysis and Review file of 25,042 beneficiaries who had gastric band procedures between 2006 and 2013.
Disclosures: Coauthor Justin B. Dimick, MD, disclosed a financial interest in ArborMetrix. The other coauthors reported having no financial disclosures. The Robert Wood Johnson Foundation, U.S. Department of Veterans Affairs, National Institute on Aging, and National Institute of Diabetes and Digestive and Kidney Diseases provided funding.
Health reform action shifts to Senate, exacerbated by poor AHCA CBO score
With a highly anticipated impact analysis from the nonpartisan Congressional Budget Office, the much maligned American Health Care Act seems relegated to a Capitol Hill recycling bin and repeal/replace focus has moved to the Senate.
Sen. Orrin Hatch (R-Utah), chairman of the Senate Finance Committee, has reached out to physicians’ organizations to learn their priorities.
The organization also called for the reduction of regulatory burdens and increased cost transparency.
Additional recommendations include
• Support for advanceable, refundable tax credits to help individuals purchase private health care coverage, with the current tax credit structure in the ACA maintained.
• Maintenance of the ACA cost-sharing reductions for 2017 and 2018.
• Enhanced tax credits for young adults aged 19-30 years and modest funding of health savings accounts to help counter high deductibles.
The AMA also supports maintenance of the current Medicaid expansion while offering states even more flexibility for innovation.
Similarly, the American Osteopathic Association noted that any policy changes “should be to expand or at least maintain access to comprehensive, affordable coverage and care. With that said, we are also concerned about the growing potential for market instability and the deleterious effect that a collapse would have on patients’ access to insurance coverage and health care services,” according to a May 23 letter from Boyd Buser, DO, president of AOA.
The organization called for continued coverage of preventive care services with no copayment and expressed concern that “the changes being considered for the Medicaid program as part of health reform discussions will significantly impact coverage for these Americans.”
The House-passed American Health Care Act (H.R. 1628) would repeal the ACA’s Medicaid expansion provisions and implement a per-capita funding scheme for state Medicaid programs. “The AOA is concerned that this will leave states with insufficient funds to provide care to the most vulnerable in our society,” Dr. Buser wrote.
AOA recommended a number of consumer protections, including adequate funding of an invisible risk-sharing program that is maintained by an organization outside of the government; efforts to improve health literacy, and other cost-containment recommendations, particularly the maintenance of the cost-sharing reduction payments.
Even if the Senate were able to incorporate these recommendations, its leaders face an uphill battle to get enough support for passage.
“If, on the Republican side, they can come to an agreement on the bill, I think they have a chance of passing it,” Julius Hobson, senior policy analyst at the Washington law firm Polsinelli, said in an interview. “The GOP is not taking up the House-passed AHCA but is proceeding with writing its own reform legislation.
However, he pointed out that there is very little margin for error because the party only holds a two-seat majority and moderates Republican senators have expressed concerns about cuts to Medicaid.
If Senate Republicans do pass a bill that appeals to its more moderate members, that legislation is likely to face stiff opposition in the House.
“Given what we just saw the House go through, I don’t know” if a more moderate bill could pass in that chamber, he said, adding that, if “they don’t do this by the end of the calendar year, you can forget it.”
The potential death blow to the AHCA came in the form of a financial impact “score” from the Congressional Budget Office (CBO).
The government budget watchdog estimated the AHCA would reduce the budget deficit by $119 billion from 2017 to 2026, with the largest savings coming from “reductions in outlays for Medicaid and from the replacement of the Affordable Care Act’s subsidies for nongroup health insurance with new tax credits for nongroup health insurance. Those savings would be partially offset by other changes in coverage provisions – spending for a new Patient and State Stability Fund, designed to reduce premiums, and a reduction in revenues from repealing penalties on employers who do not offer insurance and on people who do not purchase insurance.”
The CBO score also predicted that an additional 23 million patients would be uninsured by 2026 – for a total of 51 million – compared with a total of 28 million uninsured under the ACA.
CBO analysts predicted that nongroup health insurance markets would stabilize before 2020 under the AHCA, but instability could increased if states seek waivers to modify the essential health benefits and community rating requirements.
Although premiums overall would decrease under the AHCA, certain subpopulations could see substantial increases under state waivers.
Additionally, the waivers could also impact the provision of care.
Analysis of the impact of the waiving of essential health benefits is the part doctors should be paying most attention to, Laura Wooster, AOA senior vice president of public policy, said in an interview.
“They would start seeing patients who potentially would have insurance plans that only cover certain benefits,” she said. That would “prevent them from providing certain services or care for their patients under the patients’ coverage. Even a more immediate administrative aspect of it that things would become more complicated if there isn’t a uniform standard, then it is going to be even more complicated for patients to navigate what’s covered, what’s not, and, if the physician takes the insurance, which services are they able to provide.”
She added that, if states waive any or all of the essential health benefits, those benefits would no longer be subject to lifetime and annual caps on spending.
Mr. Hobson suggested that the biggest part of the CBO score that doctors should be worried about is the spike in uninsured that could result from any reform effort.
“The concern for physicians will be that they are more likely to see more patients who don’t have insurance depending on the specialty,” Mr. Hobson said. “Primary care will certainly see a lot of that. Physicians in emergency departments will see more of them because, as you well know, when people don’t have insurance, that’s where they show up when they are at their sickest.”
He added that a “patient can come in, a physician can diagnose the problem, but, if the patient doesn’t have the insurance, he or she may not have the ability to spend the money to do what they need to do, which means they will be back.”
With a highly anticipated impact analysis from the nonpartisan Congressional Budget Office, the much maligned American Health Care Act seems relegated to a Capitol Hill recycling bin and repeal/replace focus has moved to the Senate.
Sen. Orrin Hatch (R-Utah), chairman of the Senate Finance Committee, has reached out to physicians’ organizations to learn their priorities.
The organization also called for the reduction of regulatory burdens and increased cost transparency.
Additional recommendations include
• Support for advanceable, refundable tax credits to help individuals purchase private health care coverage, with the current tax credit structure in the ACA maintained.
• Maintenance of the ACA cost-sharing reductions for 2017 and 2018.
• Enhanced tax credits for young adults aged 19-30 years and modest funding of health savings accounts to help counter high deductibles.
The AMA also supports maintenance of the current Medicaid expansion while offering states even more flexibility for innovation.
Similarly, the American Osteopathic Association noted that any policy changes “should be to expand or at least maintain access to comprehensive, affordable coverage and care. With that said, we are also concerned about the growing potential for market instability and the deleterious effect that a collapse would have on patients’ access to insurance coverage and health care services,” according to a May 23 letter from Boyd Buser, DO, president of AOA.
The organization called for continued coverage of preventive care services with no copayment and expressed concern that “the changes being considered for the Medicaid program as part of health reform discussions will significantly impact coverage for these Americans.”
The House-passed American Health Care Act (H.R. 1628) would repeal the ACA’s Medicaid expansion provisions and implement a per-capita funding scheme for state Medicaid programs. “The AOA is concerned that this will leave states with insufficient funds to provide care to the most vulnerable in our society,” Dr. Buser wrote.
AOA recommended a number of consumer protections, including adequate funding of an invisible risk-sharing program that is maintained by an organization outside of the government; efforts to improve health literacy, and other cost-containment recommendations, particularly the maintenance of the cost-sharing reduction payments.
Even if the Senate were able to incorporate these recommendations, its leaders face an uphill battle to get enough support for passage.
“If, on the Republican side, they can come to an agreement on the bill, I think they have a chance of passing it,” Julius Hobson, senior policy analyst at the Washington law firm Polsinelli, said in an interview. “The GOP is not taking up the House-passed AHCA but is proceeding with writing its own reform legislation.
However, he pointed out that there is very little margin for error because the party only holds a two-seat majority and moderates Republican senators have expressed concerns about cuts to Medicaid.
If Senate Republicans do pass a bill that appeals to its more moderate members, that legislation is likely to face stiff opposition in the House.
“Given what we just saw the House go through, I don’t know” if a more moderate bill could pass in that chamber, he said, adding that, if “they don’t do this by the end of the calendar year, you can forget it.”
The potential death blow to the AHCA came in the form of a financial impact “score” from the Congressional Budget Office (CBO).
The government budget watchdog estimated the AHCA would reduce the budget deficit by $119 billion from 2017 to 2026, with the largest savings coming from “reductions in outlays for Medicaid and from the replacement of the Affordable Care Act’s subsidies for nongroup health insurance with new tax credits for nongroup health insurance. Those savings would be partially offset by other changes in coverage provisions – spending for a new Patient and State Stability Fund, designed to reduce premiums, and a reduction in revenues from repealing penalties on employers who do not offer insurance and on people who do not purchase insurance.”
The CBO score also predicted that an additional 23 million patients would be uninsured by 2026 – for a total of 51 million – compared with a total of 28 million uninsured under the ACA.
CBO analysts predicted that nongroup health insurance markets would stabilize before 2020 under the AHCA, but instability could increased if states seek waivers to modify the essential health benefits and community rating requirements.
Although premiums overall would decrease under the AHCA, certain subpopulations could see substantial increases under state waivers.
Additionally, the waivers could also impact the provision of care.
Analysis of the impact of the waiving of essential health benefits is the part doctors should be paying most attention to, Laura Wooster, AOA senior vice president of public policy, said in an interview.
“They would start seeing patients who potentially would have insurance plans that only cover certain benefits,” she said. That would “prevent them from providing certain services or care for their patients under the patients’ coverage. Even a more immediate administrative aspect of it that things would become more complicated if there isn’t a uniform standard, then it is going to be even more complicated for patients to navigate what’s covered, what’s not, and, if the physician takes the insurance, which services are they able to provide.”
She added that, if states waive any or all of the essential health benefits, those benefits would no longer be subject to lifetime and annual caps on spending.
Mr. Hobson suggested that the biggest part of the CBO score that doctors should be worried about is the spike in uninsured that could result from any reform effort.
“The concern for physicians will be that they are more likely to see more patients who don’t have insurance depending on the specialty,” Mr. Hobson said. “Primary care will certainly see a lot of that. Physicians in emergency departments will see more of them because, as you well know, when people don’t have insurance, that’s where they show up when they are at their sickest.”
He added that a “patient can come in, a physician can diagnose the problem, but, if the patient doesn’t have the insurance, he or she may not have the ability to spend the money to do what they need to do, which means they will be back.”
With a highly anticipated impact analysis from the nonpartisan Congressional Budget Office, the much maligned American Health Care Act seems relegated to a Capitol Hill recycling bin and repeal/replace focus has moved to the Senate.
Sen. Orrin Hatch (R-Utah), chairman of the Senate Finance Committee, has reached out to physicians’ organizations to learn their priorities.
The organization also called for the reduction of regulatory burdens and increased cost transparency.
Additional recommendations include
• Support for advanceable, refundable tax credits to help individuals purchase private health care coverage, with the current tax credit structure in the ACA maintained.
• Maintenance of the ACA cost-sharing reductions for 2017 and 2018.
• Enhanced tax credits for young adults aged 19-30 years and modest funding of health savings accounts to help counter high deductibles.
The AMA also supports maintenance of the current Medicaid expansion while offering states even more flexibility for innovation.
Similarly, the American Osteopathic Association noted that any policy changes “should be to expand or at least maintain access to comprehensive, affordable coverage and care. With that said, we are also concerned about the growing potential for market instability and the deleterious effect that a collapse would have on patients’ access to insurance coverage and health care services,” according to a May 23 letter from Boyd Buser, DO, president of AOA.
The organization called for continued coverage of preventive care services with no copayment and expressed concern that “the changes being considered for the Medicaid program as part of health reform discussions will significantly impact coverage for these Americans.”
The House-passed American Health Care Act (H.R. 1628) would repeal the ACA’s Medicaid expansion provisions and implement a per-capita funding scheme for state Medicaid programs. “The AOA is concerned that this will leave states with insufficient funds to provide care to the most vulnerable in our society,” Dr. Buser wrote.
AOA recommended a number of consumer protections, including adequate funding of an invisible risk-sharing program that is maintained by an organization outside of the government; efforts to improve health literacy, and other cost-containment recommendations, particularly the maintenance of the cost-sharing reduction payments.
Even if the Senate were able to incorporate these recommendations, its leaders face an uphill battle to get enough support for passage.
“If, on the Republican side, they can come to an agreement on the bill, I think they have a chance of passing it,” Julius Hobson, senior policy analyst at the Washington law firm Polsinelli, said in an interview. “The GOP is not taking up the House-passed AHCA but is proceeding with writing its own reform legislation.
However, he pointed out that there is very little margin for error because the party only holds a two-seat majority and moderates Republican senators have expressed concerns about cuts to Medicaid.
If Senate Republicans do pass a bill that appeals to its more moderate members, that legislation is likely to face stiff opposition in the House.
“Given what we just saw the House go through, I don’t know” if a more moderate bill could pass in that chamber, he said, adding that, if “they don’t do this by the end of the calendar year, you can forget it.”
The potential death blow to the AHCA came in the form of a financial impact “score” from the Congressional Budget Office (CBO).
The government budget watchdog estimated the AHCA would reduce the budget deficit by $119 billion from 2017 to 2026, with the largest savings coming from “reductions in outlays for Medicaid and from the replacement of the Affordable Care Act’s subsidies for nongroup health insurance with new tax credits for nongroup health insurance. Those savings would be partially offset by other changes in coverage provisions – spending for a new Patient and State Stability Fund, designed to reduce premiums, and a reduction in revenues from repealing penalties on employers who do not offer insurance and on people who do not purchase insurance.”
The CBO score also predicted that an additional 23 million patients would be uninsured by 2026 – for a total of 51 million – compared with a total of 28 million uninsured under the ACA.
CBO analysts predicted that nongroup health insurance markets would stabilize before 2020 under the AHCA, but instability could increased if states seek waivers to modify the essential health benefits and community rating requirements.
Although premiums overall would decrease under the AHCA, certain subpopulations could see substantial increases under state waivers.
Additionally, the waivers could also impact the provision of care.
Analysis of the impact of the waiving of essential health benefits is the part doctors should be paying most attention to, Laura Wooster, AOA senior vice president of public policy, said in an interview.
“They would start seeing patients who potentially would have insurance plans that only cover certain benefits,” she said. That would “prevent them from providing certain services or care for their patients under the patients’ coverage. Even a more immediate administrative aspect of it that things would become more complicated if there isn’t a uniform standard, then it is going to be even more complicated for patients to navigate what’s covered, what’s not, and, if the physician takes the insurance, which services are they able to provide.”
She added that, if states waive any or all of the essential health benefits, those benefits would no longer be subject to lifetime and annual caps on spending.
Mr. Hobson suggested that the biggest part of the CBO score that doctors should be worried about is the spike in uninsured that could result from any reform effort.
“The concern for physicians will be that they are more likely to see more patients who don’t have insurance depending on the specialty,” Mr. Hobson said. “Primary care will certainly see a lot of that. Physicians in emergency departments will see more of them because, as you well know, when people don’t have insurance, that’s where they show up when they are at their sickest.”
He added that a “patient can come in, a physician can diagnose the problem, but, if the patient doesn’t have the insurance, he or she may not have the ability to spend the money to do what they need to do, which means they will be back.”
President’s budget: Malpractice reform but deep health care cuts
President Trump’s proposed budget for fiscal 2018 could mean crushing blows for some of the nation’s largest health care programs, if it makes it through Congress.
The proposed budget “clearly reflects the priorities of the Trump administration, which is to cut taxes, to increase defense spending and spending on boarder security, and also to cut domestic spending,” Timothy S. Jost, a health law professor at Washington and Lee University in Lexington, Va., said in an interview*.
The proposed CHIP reduction would eliminate the increased federal match provided by the Affordable Care Act and cap eligibility for federal CHIP funding at 250% of the federal poverty level, according to a summary of the proposal by the Kaiser Family Foundation. States would be able to transfer children in families with incomes below 133% of the poverty line who were moved from CHIP to Medicaid under the ACA back to CHIP.
If the cuts are approved, physicians face the challenge of caring for more uninsured and underinsured patients, said John D. Abramson, MD, a family physician and health policy lecturer at Harvard Medical School in Boston. He notes that the budget’s proposed Medicaid cuts come on top of Medicaid reductions included in the AHCA.
Meanwhile, the Food and Drug Administration stands to lose $850 million for 2018 through the budget, although medical product user fees would increase, resulting in a program increase of about $450 million. The President proposes to cut CDC funds by $1.3 billion and withdraw $252 million in funding from the Substance Abuse and Mental Health Services Administration. The cuts and savings are further outlined in a U.S. Department of Health & Human Services budget summary.
The cuts to mental health care funding would no doubt affect mental health care providers and the patients they seek to treat, Mr. Jost said. “Given the seriousness of the opioid crisis, it doesn’t seem like a great time to be cutting funding for substance abuse treatment.”
Medical research may also suffer under the proposed budget. The President intends to cut funding for the National Institutes of Health by $5.8 billion, while eliminating the Agency for Health Care Research and Quality as a stand alone agency and folding it into the NIH with less funding.
The American Association for the Advancement of Science expressed concern that the research cuts could devastate the country’s science and technology enterprise and weaken the nation’s economic growth.
“Slashing funding of critically important federal agencies threatens our nation’s ability to advance cures for disease, develop new energy technologies, improve public health, train the next generation of scientists and engineers and grow the American economy,” Rush Holt, association CEO said in a statement.
Other proposals in the budget could be positive for health care, experts say.
IPAB “really hasn’t had any impact on health care over the past 7 years,” Dr. Ayanian said. ““That’s potentially one area of compromise as senators and representatives discuss ways in which the Affordable Care Act can be reformed.”
Medical malpractice reforms are also included in the proposed budget. The proposal includes a $250,000 cap on noneconomic damages, a 3-year statute of limitations, and an established safe harbor for clinicians following evidence-based clinical practice guidelines. The changes are estimated to save the federal government $55 billion over 10 years.
“That’s an area of frequent concern for practicing physicians,” Dr. Ayanian said. “To the extent that reforms can be developed that are acceptable to patients and physicians that create the right incentives for improving health care quality and avoiding medical errors, that could be a positive. It’ll depend very much on the reform proposals that are developed. I think there is potential for bipartisan agreement in this area.”
Ms. Carpenter stressed that the President’s budget is only the first step in a lengthy budget process that will include congressional hearings and debate before legislators draft budget resolutions. “I expect Congress to debate some of the cuts to domestic spending, including things like NIH. There seems to be a good bit of alignment in terms of repeal and replace of the [ACA] and potentially some of the Medicaid reforms.”
“Trump’s budget breaks his promise not to touch Social Security, which at its core is lifeline insurance for Americans who can no longer work, not just in retirement, but also due to a disability,” Sen. Wyden said in a statement. “It also slashes Medicaid by over $600 billion beyond the damage done by Trumpcare, further threatening pediatric care, the nursing home benefit, special education programs in schools, and other health care like substance misuse treatment.”
House Energy and Commerce Committee Chairman Greg Walden (R-Ore.) commended the budget proposal as one that demonstrates President Trump’s commitment to balancing the budget and responsibly prioritizing taxpayer dollars.
“The initiatives modernizing our energy infrastructure and promoting our nation’s energy abundance would undoubtedly make positive impacts on our constituents’ lives,” he said in a statement. “The president’s proposals show the difficult choices facing the country as we work to reduce the deficit, protect our security, and grow jobs.”
Additional proposals in the President’s budget include:
• Nearly $70 billion in cuts for Social Security disability benefits over the next 10 years.
• A funding increase for the Veterans Administration, including $29 billion over the next decade for a program that enables veterans to seek outside health services from private doctors.
• A ban on funds for clinics and medical centers that provide abortions, such as Planned Parenthood.
• A $70 million increase for Medicare and Medicaid fraud prevention efforts in 2018.
• A $114 million funding increase for the Center for Medicare and Medicaid Innovation.
[email protected]
On Twitter @legal_med
*This story was updated May 26, 2017
President Trump’s proposed budget for fiscal 2018 could mean crushing blows for some of the nation’s largest health care programs, if it makes it through Congress.
The proposed budget “clearly reflects the priorities of the Trump administration, which is to cut taxes, to increase defense spending and spending on boarder security, and also to cut domestic spending,” Timothy S. Jost, a health law professor at Washington and Lee University in Lexington, Va., said in an interview*.
The proposed CHIP reduction would eliminate the increased federal match provided by the Affordable Care Act and cap eligibility for federal CHIP funding at 250% of the federal poverty level, according to a summary of the proposal by the Kaiser Family Foundation. States would be able to transfer children in families with incomes below 133% of the poverty line who were moved from CHIP to Medicaid under the ACA back to CHIP.
If the cuts are approved, physicians face the challenge of caring for more uninsured and underinsured patients, said John D. Abramson, MD, a family physician and health policy lecturer at Harvard Medical School in Boston. He notes that the budget’s proposed Medicaid cuts come on top of Medicaid reductions included in the AHCA.
Meanwhile, the Food and Drug Administration stands to lose $850 million for 2018 through the budget, although medical product user fees would increase, resulting in a program increase of about $450 million. The President proposes to cut CDC funds by $1.3 billion and withdraw $252 million in funding from the Substance Abuse and Mental Health Services Administration. The cuts and savings are further outlined in a U.S. Department of Health & Human Services budget summary.
The cuts to mental health care funding would no doubt affect mental health care providers and the patients they seek to treat, Mr. Jost said. “Given the seriousness of the opioid crisis, it doesn’t seem like a great time to be cutting funding for substance abuse treatment.”
Medical research may also suffer under the proposed budget. The President intends to cut funding for the National Institutes of Health by $5.8 billion, while eliminating the Agency for Health Care Research and Quality as a stand alone agency and folding it into the NIH with less funding.
The American Association for the Advancement of Science expressed concern that the research cuts could devastate the country’s science and technology enterprise and weaken the nation’s economic growth.
“Slashing funding of critically important federal agencies threatens our nation’s ability to advance cures for disease, develop new energy technologies, improve public health, train the next generation of scientists and engineers and grow the American economy,” Rush Holt, association CEO said in a statement.
Other proposals in the budget could be positive for health care, experts say.
IPAB “really hasn’t had any impact on health care over the past 7 years,” Dr. Ayanian said. ““That’s potentially one area of compromise as senators and representatives discuss ways in which the Affordable Care Act can be reformed.”
Medical malpractice reforms are also included in the proposed budget. The proposal includes a $250,000 cap on noneconomic damages, a 3-year statute of limitations, and an established safe harbor for clinicians following evidence-based clinical practice guidelines. The changes are estimated to save the federal government $55 billion over 10 years.
“That’s an area of frequent concern for practicing physicians,” Dr. Ayanian said. “To the extent that reforms can be developed that are acceptable to patients and physicians that create the right incentives for improving health care quality and avoiding medical errors, that could be a positive. It’ll depend very much on the reform proposals that are developed. I think there is potential for bipartisan agreement in this area.”
Ms. Carpenter stressed that the President’s budget is only the first step in a lengthy budget process that will include congressional hearings and debate before legislators draft budget resolutions. “I expect Congress to debate some of the cuts to domestic spending, including things like NIH. There seems to be a good bit of alignment in terms of repeal and replace of the [ACA] and potentially some of the Medicaid reforms.”
“Trump’s budget breaks his promise not to touch Social Security, which at its core is lifeline insurance for Americans who can no longer work, not just in retirement, but also due to a disability,” Sen. Wyden said in a statement. “It also slashes Medicaid by over $600 billion beyond the damage done by Trumpcare, further threatening pediatric care, the nursing home benefit, special education programs in schools, and other health care like substance misuse treatment.”
House Energy and Commerce Committee Chairman Greg Walden (R-Ore.) commended the budget proposal as one that demonstrates President Trump’s commitment to balancing the budget and responsibly prioritizing taxpayer dollars.
“The initiatives modernizing our energy infrastructure and promoting our nation’s energy abundance would undoubtedly make positive impacts on our constituents’ lives,” he said in a statement. “The president’s proposals show the difficult choices facing the country as we work to reduce the deficit, protect our security, and grow jobs.”
Additional proposals in the President’s budget include:
• Nearly $70 billion in cuts for Social Security disability benefits over the next 10 years.
• A funding increase for the Veterans Administration, including $29 billion over the next decade for a program that enables veterans to seek outside health services from private doctors.
• A ban on funds for clinics and medical centers that provide abortions, such as Planned Parenthood.
• A $70 million increase for Medicare and Medicaid fraud prevention efforts in 2018.
• A $114 million funding increase for the Center for Medicare and Medicaid Innovation.
[email protected]
On Twitter @legal_med
*This story was updated May 26, 2017
President Trump’s proposed budget for fiscal 2018 could mean crushing blows for some of the nation’s largest health care programs, if it makes it through Congress.
The proposed budget “clearly reflects the priorities of the Trump administration, which is to cut taxes, to increase defense spending and spending on boarder security, and also to cut domestic spending,” Timothy S. Jost, a health law professor at Washington and Lee University in Lexington, Va., said in an interview*.
The proposed CHIP reduction would eliminate the increased federal match provided by the Affordable Care Act and cap eligibility for federal CHIP funding at 250% of the federal poverty level, according to a summary of the proposal by the Kaiser Family Foundation. States would be able to transfer children in families with incomes below 133% of the poverty line who were moved from CHIP to Medicaid under the ACA back to CHIP.
If the cuts are approved, physicians face the challenge of caring for more uninsured and underinsured patients, said John D. Abramson, MD, a family physician and health policy lecturer at Harvard Medical School in Boston. He notes that the budget’s proposed Medicaid cuts come on top of Medicaid reductions included in the AHCA.
Meanwhile, the Food and Drug Administration stands to lose $850 million for 2018 through the budget, although medical product user fees would increase, resulting in a program increase of about $450 million. The President proposes to cut CDC funds by $1.3 billion and withdraw $252 million in funding from the Substance Abuse and Mental Health Services Administration. The cuts and savings are further outlined in a U.S. Department of Health & Human Services budget summary.
The cuts to mental health care funding would no doubt affect mental health care providers and the patients they seek to treat, Mr. Jost said. “Given the seriousness of the opioid crisis, it doesn’t seem like a great time to be cutting funding for substance abuse treatment.”
Medical research may also suffer under the proposed budget. The President intends to cut funding for the National Institutes of Health by $5.8 billion, while eliminating the Agency for Health Care Research and Quality as a stand alone agency and folding it into the NIH with less funding.
The American Association for the Advancement of Science expressed concern that the research cuts could devastate the country’s science and technology enterprise and weaken the nation’s economic growth.
“Slashing funding of critically important federal agencies threatens our nation’s ability to advance cures for disease, develop new energy technologies, improve public health, train the next generation of scientists and engineers and grow the American economy,” Rush Holt, association CEO said in a statement.
Other proposals in the budget could be positive for health care, experts say.
IPAB “really hasn’t had any impact on health care over the past 7 years,” Dr. Ayanian said. ““That’s potentially one area of compromise as senators and representatives discuss ways in which the Affordable Care Act can be reformed.”
Medical malpractice reforms are also included in the proposed budget. The proposal includes a $250,000 cap on noneconomic damages, a 3-year statute of limitations, and an established safe harbor for clinicians following evidence-based clinical practice guidelines. The changes are estimated to save the federal government $55 billion over 10 years.
“That’s an area of frequent concern for practicing physicians,” Dr. Ayanian said. “To the extent that reforms can be developed that are acceptable to patients and physicians that create the right incentives for improving health care quality and avoiding medical errors, that could be a positive. It’ll depend very much on the reform proposals that are developed. I think there is potential for bipartisan agreement in this area.”
Ms. Carpenter stressed that the President’s budget is only the first step in a lengthy budget process that will include congressional hearings and debate before legislators draft budget resolutions. “I expect Congress to debate some of the cuts to domestic spending, including things like NIH. There seems to be a good bit of alignment in terms of repeal and replace of the [ACA] and potentially some of the Medicaid reforms.”
“Trump’s budget breaks his promise not to touch Social Security, which at its core is lifeline insurance for Americans who can no longer work, not just in retirement, but also due to a disability,” Sen. Wyden said in a statement. “It also slashes Medicaid by over $600 billion beyond the damage done by Trumpcare, further threatening pediatric care, the nursing home benefit, special education programs in schools, and other health care like substance misuse treatment.”
House Energy and Commerce Committee Chairman Greg Walden (R-Ore.) commended the budget proposal as one that demonstrates President Trump’s commitment to balancing the budget and responsibly prioritizing taxpayer dollars.
“The initiatives modernizing our energy infrastructure and promoting our nation’s energy abundance would undoubtedly make positive impacts on our constituents’ lives,” he said in a statement. “The president’s proposals show the difficult choices facing the country as we work to reduce the deficit, protect our security, and grow jobs.”
Additional proposals in the President’s budget include:
• Nearly $70 billion in cuts for Social Security disability benefits over the next 10 years.
• A funding increase for the Veterans Administration, including $29 billion over the next decade for a program that enables veterans to seek outside health services from private doctors.
• A ban on funds for clinics and medical centers that provide abortions, such as Planned Parenthood.
• A $70 million increase for Medicare and Medicaid fraud prevention efforts in 2018.
• A $114 million funding increase for the Center for Medicare and Medicaid Innovation.
[email protected]
On Twitter @legal_med
*This story was updated May 26, 2017
New DES hailed for smallest coronary vessels
Paris – The first multicenter, prospective trial of a drug-eluting stent designed specifically to treat lesions in coronary vessels less than 2.25 mm in diameter showed excellent outcomes, with a 1-year target lesion failure rate of 5% for the Resolute Onyx 2.0 mm diameter zotarolimus-eluting stent.
This result in the pivotal trial easily surpassed the prespecified performance goal of a 19% target lesion failure rate, Matthew J. Price, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Hemodynamically significant lesions in such small vessels are “not uncommon, particularly in diabetic patients,” Dr. Price said in an interview. Indeed, 47% of patients in the clinical trial had diabetes.
At present, the only ways to treat coronary disease in arteries having a reference vessel diameter less than 2.25 mm are off-label placement of an oversized stent, with its attendant risk of complications; standard balloon angioplasty, which entails a particularly high restenosis rate in this setting; or medical management, the cardiologist noted.
He presented a multicenter, prospective, open-label, single-arm trial of 101 patients with documented ischemia-producing obstructions in coronary arteries having a reference vessel diameter less than 2.25 mm, a lesion length less than 27 mm, and evidence of ischemia attributable to the lesion, typically via fractional flow reserve. The mean diameter by quantitative coronary angiography was 1.91 mm.
The primary endpoint was the rate of target lesion failure at 12 months, a composite comprising cardiac death, target vessel MI, or clinically driven target lesion revascularization. This endpoint occurred in 5% of patients. There was a 3% target vessel MI rate and a 2% target lesion revascularization rate. There were no cardiac deaths.
“Importantly, the stent thrombosis rate in these patients with extremely small vessels was zero,” the cardiologist emphasized.
The mean angiographic in-stent late lumen loss at 13 months was 0.26 mm, which Dr. Price characterized as “quite good.” The in-segment binary angiographic restenosis rate was 20%.
“That’s slightly higher than you would expect to see in vessels with larger reference diameters. I think that’s because of the lack of headroom. You have a very small vessel, and, even with a very small stent, even a small amount of late loss will give you a larger percent diameter restenosis over time,” he explained.
The 19% target lesion failure rate selected as a performance goal in the trial was set somewhat arbitrarily. It wasn’t possible to randomize patients to a comparator arm because there are no approved stents for vessels less than 2.25 mm in diameter. The 19% figure was arrived at in discussion with the Food and Drug Administration on the basis of similarity to the performance goal used in clinical trials to gain approval of 2.25-mm, drug-eluting stents. Because the Onyx 2.0-mm-diameter trial was developed in collaboration with the FDA and the stent aced its primary endpoint and showed excellent clinical outcomes, Dr. Price anticipates the device will readily gain regulatory approval. In April 2017, the FDA approved the Resolute Onyx in sizes of 2.25- to 5.0-mm diameter.
The study met with an enthusiastic reception.
“That was terrific. It’s clearly an incredibly important unmet clinical need,” commented session cochair David R. Holmes Jr., MD, of the Mayo Clinic in Rochester, Minn.
Assuming the stent is approved, how should interventionalists put it into practice? he asked.
Dr. Price replied that, first, it’s important to step back and ask if percutaneous coronary intervention of a particular lesion in a very small coronary artery is clinically indicated. The stent itself is readily manipulatable. It is a thin-strut device constructed of a single strand of a cobalt alloy with enhanced radiopacity.
Investigators in the trial used the standard approach to dual antiplatelet therapy – at least 6 months, with 12 months preferable.
The 20% in-segment binary restenosis rate at 13 months provides a clear message for interventionalists, he continued. “What this tells me is that, while this is a very good stent, we can’t forget to treat the patient aggressively with medical therapy to stop the progression of prediabetes, diabetes, and small vessel disease in addition to treating obstructive lesions with a small stent.”
Asked if the lack of headroom in these extra-small arteries warrants liberal use of intraprocedural imaging to make sure the stent is perfectly apposed, Dr. Price replied that he doesn’t think so. He noted that intravascular ultrasound and optical coherence tomography were seldom used in the trial, yet the results were reassuringly excellent.
The study results were published simultaneously with Dr. Price’s presentation (JACC Cardiovasc Interv. 2017 May 17. doi: 10.1016/j.jcin.2017.05.004). The trial was sponsored by Medtronic. Dr. Price reported serving as a consultant and paid speaker on behalf of that company, as well as AstraZeneca, Boston Scientific, St. Jude Medical, and The Medicines Company.
Paris – The first multicenter, prospective trial of a drug-eluting stent designed specifically to treat lesions in coronary vessels less than 2.25 mm in diameter showed excellent outcomes, with a 1-year target lesion failure rate of 5% for the Resolute Onyx 2.0 mm diameter zotarolimus-eluting stent.
This result in the pivotal trial easily surpassed the prespecified performance goal of a 19% target lesion failure rate, Matthew J. Price, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Hemodynamically significant lesions in such small vessels are “not uncommon, particularly in diabetic patients,” Dr. Price said in an interview. Indeed, 47% of patients in the clinical trial had diabetes.
At present, the only ways to treat coronary disease in arteries having a reference vessel diameter less than 2.25 mm are off-label placement of an oversized stent, with its attendant risk of complications; standard balloon angioplasty, which entails a particularly high restenosis rate in this setting; or medical management, the cardiologist noted.
He presented a multicenter, prospective, open-label, single-arm trial of 101 patients with documented ischemia-producing obstructions in coronary arteries having a reference vessel diameter less than 2.25 mm, a lesion length less than 27 mm, and evidence of ischemia attributable to the lesion, typically via fractional flow reserve. The mean diameter by quantitative coronary angiography was 1.91 mm.
The primary endpoint was the rate of target lesion failure at 12 months, a composite comprising cardiac death, target vessel MI, or clinically driven target lesion revascularization. This endpoint occurred in 5% of patients. There was a 3% target vessel MI rate and a 2% target lesion revascularization rate. There were no cardiac deaths.
“Importantly, the stent thrombosis rate in these patients with extremely small vessels was zero,” the cardiologist emphasized.
The mean angiographic in-stent late lumen loss at 13 months was 0.26 mm, which Dr. Price characterized as “quite good.” The in-segment binary angiographic restenosis rate was 20%.
“That’s slightly higher than you would expect to see in vessels with larger reference diameters. I think that’s because of the lack of headroom. You have a very small vessel, and, even with a very small stent, even a small amount of late loss will give you a larger percent diameter restenosis over time,” he explained.
The 19% target lesion failure rate selected as a performance goal in the trial was set somewhat arbitrarily. It wasn’t possible to randomize patients to a comparator arm because there are no approved stents for vessels less than 2.25 mm in diameter. The 19% figure was arrived at in discussion with the Food and Drug Administration on the basis of similarity to the performance goal used in clinical trials to gain approval of 2.25-mm, drug-eluting stents. Because the Onyx 2.0-mm-diameter trial was developed in collaboration with the FDA and the stent aced its primary endpoint and showed excellent clinical outcomes, Dr. Price anticipates the device will readily gain regulatory approval. In April 2017, the FDA approved the Resolute Onyx in sizes of 2.25- to 5.0-mm diameter.
The study met with an enthusiastic reception.
“That was terrific. It’s clearly an incredibly important unmet clinical need,” commented session cochair David R. Holmes Jr., MD, of the Mayo Clinic in Rochester, Minn.
Assuming the stent is approved, how should interventionalists put it into practice? he asked.
Dr. Price replied that, first, it’s important to step back and ask if percutaneous coronary intervention of a particular lesion in a very small coronary artery is clinically indicated. The stent itself is readily manipulatable. It is a thin-strut device constructed of a single strand of a cobalt alloy with enhanced radiopacity.
Investigators in the trial used the standard approach to dual antiplatelet therapy – at least 6 months, with 12 months preferable.
The 20% in-segment binary restenosis rate at 13 months provides a clear message for interventionalists, he continued. “What this tells me is that, while this is a very good stent, we can’t forget to treat the patient aggressively with medical therapy to stop the progression of prediabetes, diabetes, and small vessel disease in addition to treating obstructive lesions with a small stent.”
Asked if the lack of headroom in these extra-small arteries warrants liberal use of intraprocedural imaging to make sure the stent is perfectly apposed, Dr. Price replied that he doesn’t think so. He noted that intravascular ultrasound and optical coherence tomography were seldom used in the trial, yet the results were reassuringly excellent.
The study results were published simultaneously with Dr. Price’s presentation (JACC Cardiovasc Interv. 2017 May 17. doi: 10.1016/j.jcin.2017.05.004). The trial was sponsored by Medtronic. Dr. Price reported serving as a consultant and paid speaker on behalf of that company, as well as AstraZeneca, Boston Scientific, St. Jude Medical, and The Medicines Company.
Paris – The first multicenter, prospective trial of a drug-eluting stent designed specifically to treat lesions in coronary vessels less than 2.25 mm in diameter showed excellent outcomes, with a 1-year target lesion failure rate of 5% for the Resolute Onyx 2.0 mm diameter zotarolimus-eluting stent.
This result in the pivotal trial easily surpassed the prespecified performance goal of a 19% target lesion failure rate, Matthew J. Price, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Hemodynamically significant lesions in such small vessels are “not uncommon, particularly in diabetic patients,” Dr. Price said in an interview. Indeed, 47% of patients in the clinical trial had diabetes.
At present, the only ways to treat coronary disease in arteries having a reference vessel diameter less than 2.25 mm are off-label placement of an oversized stent, with its attendant risk of complications; standard balloon angioplasty, which entails a particularly high restenosis rate in this setting; or medical management, the cardiologist noted.
He presented a multicenter, prospective, open-label, single-arm trial of 101 patients with documented ischemia-producing obstructions in coronary arteries having a reference vessel diameter less than 2.25 mm, a lesion length less than 27 mm, and evidence of ischemia attributable to the lesion, typically via fractional flow reserve. The mean diameter by quantitative coronary angiography was 1.91 mm.
The primary endpoint was the rate of target lesion failure at 12 months, a composite comprising cardiac death, target vessel MI, or clinically driven target lesion revascularization. This endpoint occurred in 5% of patients. There was a 3% target vessel MI rate and a 2% target lesion revascularization rate. There were no cardiac deaths.
“Importantly, the stent thrombosis rate in these patients with extremely small vessels was zero,” the cardiologist emphasized.
The mean angiographic in-stent late lumen loss at 13 months was 0.26 mm, which Dr. Price characterized as “quite good.” The in-segment binary angiographic restenosis rate was 20%.
“That’s slightly higher than you would expect to see in vessels with larger reference diameters. I think that’s because of the lack of headroom. You have a very small vessel, and, even with a very small stent, even a small amount of late loss will give you a larger percent diameter restenosis over time,” he explained.
The 19% target lesion failure rate selected as a performance goal in the trial was set somewhat arbitrarily. It wasn’t possible to randomize patients to a comparator arm because there are no approved stents for vessels less than 2.25 mm in diameter. The 19% figure was arrived at in discussion with the Food and Drug Administration on the basis of similarity to the performance goal used in clinical trials to gain approval of 2.25-mm, drug-eluting stents. Because the Onyx 2.0-mm-diameter trial was developed in collaboration with the FDA and the stent aced its primary endpoint and showed excellent clinical outcomes, Dr. Price anticipates the device will readily gain regulatory approval. In April 2017, the FDA approved the Resolute Onyx in sizes of 2.25- to 5.0-mm diameter.
The study met with an enthusiastic reception.
“That was terrific. It’s clearly an incredibly important unmet clinical need,” commented session cochair David R. Holmes Jr., MD, of the Mayo Clinic in Rochester, Minn.
Assuming the stent is approved, how should interventionalists put it into practice? he asked.
Dr. Price replied that, first, it’s important to step back and ask if percutaneous coronary intervention of a particular lesion in a very small coronary artery is clinically indicated. The stent itself is readily manipulatable. It is a thin-strut device constructed of a single strand of a cobalt alloy with enhanced radiopacity.
Investigators in the trial used the standard approach to dual antiplatelet therapy – at least 6 months, with 12 months preferable.
The 20% in-segment binary restenosis rate at 13 months provides a clear message for interventionalists, he continued. “What this tells me is that, while this is a very good stent, we can’t forget to treat the patient aggressively with medical therapy to stop the progression of prediabetes, diabetes, and small vessel disease in addition to treating obstructive lesions with a small stent.”
Asked if the lack of headroom in these extra-small arteries warrants liberal use of intraprocedural imaging to make sure the stent is perfectly apposed, Dr. Price replied that he doesn’t think so. He noted that intravascular ultrasound and optical coherence tomography were seldom used in the trial, yet the results were reassuringly excellent.
The study results were published simultaneously with Dr. Price’s presentation (JACC Cardiovasc Interv. 2017 May 17. doi: 10.1016/j.jcin.2017.05.004). The trial was sponsored by Medtronic. Dr. Price reported serving as a consultant and paid speaker on behalf of that company, as well as AstraZeneca, Boston Scientific, St. Jude Medical, and The Medicines Company.
AT EUROPCR
Key clinical point:
Major finding: At 12 months’ follow-up, the key outcomes were a 3% rate of target vessel MI, a 2% rate of clinically driven target lesion revascularization, no stent thrombosis, and no cardiac deaths.
Data source: A prospective, multicenter, open-label trial in 101 patients who underwent percutaneous coronary intervention for coronary lesions with a reference vessel diameter of less than 2.25 mm.
Disclosures: The trial was sponsored by Medtronic. Dr. Price reported serving as a consultant to and paid speaker on behalf of that company as well as AstraZeneca, Boston Scientific, St. Jude Medical, and The Medicines Company.