User login
AI in Mammography: Inside the Tangible Benefits Ready Now
In this Practical AI column, we’ve explored everything from large language models to the nuances of trial matching, but one of the most immediate and impactful applications of AI is unfolding right now in breast imaging. For oncologists, this isn’t an abstract future — with new screening guidelines, dense-breast mandates, and a shrinking radiology workforce, it’s the imaging reports and patient questions landing in your clinic today.
Here is what oncologists need to know, and how to put it to work for their patients.
Why AI in Mammography Matters
More than 200 million women undergo breast cancer screening each year. In the US alone, 10% of the 40 million women screened annually require additional diagnostic imaging, and 4%–5% of these women are eventually diagnosed with breast cancer.
Two major shifts are redefining breast cancer screening in the US: The US Preventive Services Task Force (USPSTF) now recommends biennial screening from age 40 to 74 years, and notifying patients of breast density is a federal requirement as of September 10, 2024. That means more mammograms, more patient questions, and more downstream oncology decisions. Patients will increasingly ask about “dense” breast results and what to do next. Add a national radiologist shortage into the mix, and the pressure on timely callbacks, biopsies, and treatment planning will only grow.
Can AI Help Without Compromising Care?
The short answer is yes. With AI, we may be able to transform these rate-limiting steps into opportunities for earlier detection, decentralized screening, and smarter triage and save hundreds of thousands of women from an unnecessary diagnostic procedure, if implemented deliberately.
Don’t Confuse Today’s AI With Yesterday’s CAD
Think of older computer-aided detection (CAD) like a 1990s chemotherapy drug: It sometimes helped, but it came with significant toxicity and rarely delivered consistent survival benefits. Today’s deep-learning AI is closer to targeted therapy — trained on millions of “trial participants” (mammograms), more precise, and applied in specific contexts where it adds value. If you once dismissed CAD as noise, it’s time to revisit what AI can now offer.
The role of AI is broader than drawing boxes. It provides second readings, worklist triage, risk prediction, density assessment, and decision support. FDA has cleared several AI tools for both 2D and digital breast tomosynthesis (DBT), which include iCAD ProFound (DBT), ScreenPoint Transpara (2D/DBT), and Lunit INSIGHT DBT.
Some of the strongest evidence for AI in mammography is as a second reader during screening. Large trials show that AI plus one radiologist can match reading from two radiologists, cutting workload by about 40%. For example, the MASAI randomized trial showed that AI-supported screening achieved similar cancer detection but cut human screen-reading workload about 44% vs standard double reading (39,996 vs 40,024 participants). The primary interval cancer outcomes are maturing, but the safety analysis is reassuring.
Reducing second reads and arbitration time are important for clinicians because it frees capacity for callbacks and diagnostic workups. This will be especially key given that screening now starts at age 40. That will mean about 21 to 22 million more women are newly eligible, translating to about 10 to 11 million additional mammograms each year under biennial screening.
Another important area where AI can make its mark in mammography is triage and time to diagnosis. The results from a randomized implementation study showed that AI-prioritized worklists accelerated time to additional imaging and biopsy diagnosis without harming efficiency for others — exactly the kind of outcome patients feel.
Multiple studies have demonstrated improved diagnostic performance and shorter reading times when AI supports DBT interpretation, which is important because DBT can otherwise be time intensive.
We are also seeing rapid advancement in risk-based screening, moving beyond a single dense vs not dense approach. Deep-learning risk models, such as Mirai, predict 1- to 5-year breast cancer risk directly from the mammogram, and these tools are now being assessed prospectively to guide supplemental MRI. Cost-effectiveness modeling supports risk-stratified intervals vs one-size-fits-all schedules.
Finally, automated density tools, such as Transpara Density and Volpara, offer objective, reproducible volumetric measures that map to the Breast Imaging-Reporting and Data System, which is useful for Mammography Quality Standards Act-required reporting and as inputs to risk calculators.
While early evidence suggests AI may help surface future or interval cancers earlier, including more invasive tumors, the definitive impacts on interval cancer rates and mortality require longitudinal follow-up, which is now in progress.
Pitfalls to Watch For
Bias is real. Studies show false-positive differences by race, age, and density. AI can even infer racial identity from images, potentially amplifying disparities. Performance can also shift by vendor, demographics, and prevalence.
A Radiology study of 4855 DBT exams showed that an algorithm produced more false-positive case scores in Black patients and older patients (aged 71-80 years) patients and in women with extremely dense breasts. This can happen because AI can infer proxies for race directly from images, even when humans cannot, and this can propagate disparities if not addressed. External validations and reviews emphasize that performance can shift with device manufacturer, demographics, and prevalence, which is why all tools need to undergo local validation and calibration.
Here’s a pragmatic adoption checklist before going live with an AI tool.
- Confirm FDA clearance: Verify the name and version of the algorithm, imaging modes (2D vs DBT), and operating points. Confirm 510(k) numbers.
- Local validation: Test on your patient mix and vendor stack (Hologic, GE, Siemens, Fuji). Compare this to your baseline recall rate, positive predictive value of recall (PPV1), cancer detection rate, and reading time. Commit to recalibration if drift occurs.
- Equity plan: Monitor false-positive and negative false-rates by age, race/ethnicity, and density; document corrective actions if disparities emerge. (This isn’t optional.)
- Workflow clarity: Is AI a second reader, an additional reader, or a triage tool? Who arbitrates discordance? What’s the escalation path for high-risk or interval cancer-like patterns?
- Regulatory strategy: Confirm whether the vendor has (or will file) a Predetermined Change Control Plan so models can be updated safely without repeated submissions. Also confirm how you’ll be notified about performance-relevant changes.
- Data governance: Audit logs of AI outputs, retention, protected health information handling, and the patient communication policy for AI-assisted reads.
After going live, set up a quarterly dashboard. It should include cancer detection rate per 1000 patients, recall rate, PPV1, interval cancer rate (as it matures), reading time, and turnaround time to diagnostic imaging or biopsy — all stratified by age, race/ethnicity, and density.
Here, I dissect what this discussion means through the lens of Moravec’s paradox (machines excel at what clinicians find hard, and vice versa) and offer a possible playbook for putting these tools to work.
What to Tell Patients
When speaking with patients, emphasize that a radiologist still reads their mammogram. AI helps with consistency and efficiency; it doesn’t replace human oversight. Patients with dense breasts should still expect a standard notice; discussion of individualized risk factors, such as family history, genetics, and prior biopsies; and consideration of supplemental imaging if risk warrants. But it’s also important to tell these patients that while dense breasts are common, they do not automatically mean high cancer risk.
As for screening schedules, remind patients that screening is at least biennial from 40 to 74 years of age per the USPSTF guidelines; however, specialty groups may recommend starting on an annual schedule at 40.
What You Can Implement Now
There are multiple practical use cases you can introduce now. One is to use AI as a second reader or an additional reader safety net to preserve detection while reducing human workload. This helps your breast center absorb screening expansion to age 40 without diluting quality. Another is to turn on AI triage to shorten the time to callback and biopsy for the few who need it most — patients notice and appreciate faster answers. You can also begin adopting automated density plus risk models to move beyond “dense/not dense.” For selected patients, AI-informed risk can justify MRI or tailored intervals.
Here’s a quick cheat sheet (for your next leadership or tumor-board meeting).
Do:
- Use AI as a second or additional reader or triage tool, not as a black box.
- Track cancer detection rate, recall, PPV1, interval cancers, and reading time, stratified by age, race, and breast density.
- Pair automated density with AI risk to personalize screening and supplemental imaging.
- Enroll patients in future clinical trials, such as PRISM, the first large-scale randomized controlled trial of AI for screening mammography. This US-based, $16 million, seven-site study is funded by the Patient-Centered Outcomes Research Institute.
Don’t:
- Assume “AI = CAD.” The 2015 CAD story is over; modern deep learning systems are different and require different oversight.
- Go live without a local validation and equity plan or without clarity on software updates.
- Forget to remind patients that screening starts at age 40, and dense breast notifications are now universal. Use the visit to discuss risk, supplemental imaging, and why a human still directs their care.
The Bottom Line
AI won’t replace radiologists or read mammograms for us — just as PET scans didn’t replace oncologists and stethoscopes didn’t make cardiologists obsolete. What it will do is catch what the tired human eye might miss, shave days off anxious waiting, and turn breast density into data instead of doubt. For oncologists, that means staging sooner, enrolling smarter, and spending more time talking with patients instead of chasing callbacks.
In short, AI may not take the picture, but it helps us frame the story, making it sharper, faster, and with fewer blind spots. By pairing this powerful technology with rigorous, equity-focused local validation and transparent governance under the FDA’s emerging Predetermined Change Control Plan framework, we can realize the tangible benefits of practical AI for our patients without widening disparities.
Now, during Breast Cancer Awareness Month, how about we add on AI to that pink ribbon — how cool would that be?
Thoughts? Drop me a line at [email protected]. Let’s keep the conversation — and pink ribbons — going.
Arturo Loaiza-Bonilla, MD, MSEd, is the co-founder and chief medical AI officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr Loaiza-Bonilla serves as Systemwide Chief of Hematology and Oncology at St. Luke’s University Health Network, where he maintains a connection to patient care by attending to patients 2 days a week.
A version of this article first appeared on Medscape.com.
In this Practical AI column, we’ve explored everything from large language models to the nuances of trial matching, but one of the most immediate and impactful applications of AI is unfolding right now in breast imaging. For oncologists, this isn’t an abstract future — with new screening guidelines, dense-breast mandates, and a shrinking radiology workforce, it’s the imaging reports and patient questions landing in your clinic today.
Here is what oncologists need to know, and how to put it to work for their patients.
Why AI in Mammography Matters
More than 200 million women undergo breast cancer screening each year. In the US alone, 10% of the 40 million women screened annually require additional diagnostic imaging, and 4%–5% of these women are eventually diagnosed with breast cancer.
Two major shifts are redefining breast cancer screening in the US: The US Preventive Services Task Force (USPSTF) now recommends biennial screening from age 40 to 74 years, and notifying patients of breast density is a federal requirement as of September 10, 2024. That means more mammograms, more patient questions, and more downstream oncology decisions. Patients will increasingly ask about “dense” breast results and what to do next. Add a national radiologist shortage into the mix, and the pressure on timely callbacks, biopsies, and treatment planning will only grow.
Can AI Help Without Compromising Care?
The short answer is yes. With AI, we may be able to transform these rate-limiting steps into opportunities for earlier detection, decentralized screening, and smarter triage and save hundreds of thousands of women from an unnecessary diagnostic procedure, if implemented deliberately.
Don’t Confuse Today’s AI With Yesterday’s CAD
Think of older computer-aided detection (CAD) like a 1990s chemotherapy drug: It sometimes helped, but it came with significant toxicity and rarely delivered consistent survival benefits. Today’s deep-learning AI is closer to targeted therapy — trained on millions of “trial participants” (mammograms), more precise, and applied in specific contexts where it adds value. If you once dismissed CAD as noise, it’s time to revisit what AI can now offer.
The role of AI is broader than drawing boxes. It provides second readings, worklist triage, risk prediction, density assessment, and decision support. FDA has cleared several AI tools for both 2D and digital breast tomosynthesis (DBT), which include iCAD ProFound (DBT), ScreenPoint Transpara (2D/DBT), and Lunit INSIGHT DBT.
Some of the strongest evidence for AI in mammography is as a second reader during screening. Large trials show that AI plus one radiologist can match reading from two radiologists, cutting workload by about 40%. For example, the MASAI randomized trial showed that AI-supported screening achieved similar cancer detection but cut human screen-reading workload about 44% vs standard double reading (39,996 vs 40,024 participants). The primary interval cancer outcomes are maturing, but the safety analysis is reassuring.
Reducing second reads and arbitration time are important for clinicians because it frees capacity for callbacks and diagnostic workups. This will be especially key given that screening now starts at age 40. That will mean about 21 to 22 million more women are newly eligible, translating to about 10 to 11 million additional mammograms each year under biennial screening.
Another important area where AI can make its mark in mammography is triage and time to diagnosis. The results from a randomized implementation study showed that AI-prioritized worklists accelerated time to additional imaging and biopsy diagnosis without harming efficiency for others — exactly the kind of outcome patients feel.
Multiple studies have demonstrated improved diagnostic performance and shorter reading times when AI supports DBT interpretation, which is important because DBT can otherwise be time intensive.
We are also seeing rapid advancement in risk-based screening, moving beyond a single dense vs not dense approach. Deep-learning risk models, such as Mirai, predict 1- to 5-year breast cancer risk directly from the mammogram, and these tools are now being assessed prospectively to guide supplemental MRI. Cost-effectiveness modeling supports risk-stratified intervals vs one-size-fits-all schedules.
Finally, automated density tools, such as Transpara Density and Volpara, offer objective, reproducible volumetric measures that map to the Breast Imaging-Reporting and Data System, which is useful for Mammography Quality Standards Act-required reporting and as inputs to risk calculators.
While early evidence suggests AI may help surface future or interval cancers earlier, including more invasive tumors, the definitive impacts on interval cancer rates and mortality require longitudinal follow-up, which is now in progress.
Pitfalls to Watch For
Bias is real. Studies show false-positive differences by race, age, and density. AI can even infer racial identity from images, potentially amplifying disparities. Performance can also shift by vendor, demographics, and prevalence.
A Radiology study of 4855 DBT exams showed that an algorithm produced more false-positive case scores in Black patients and older patients (aged 71-80 years) patients and in women with extremely dense breasts. This can happen because AI can infer proxies for race directly from images, even when humans cannot, and this can propagate disparities if not addressed. External validations and reviews emphasize that performance can shift with device manufacturer, demographics, and prevalence, which is why all tools need to undergo local validation and calibration.
Here’s a pragmatic adoption checklist before going live with an AI tool.
- Confirm FDA clearance: Verify the name and version of the algorithm, imaging modes (2D vs DBT), and operating points. Confirm 510(k) numbers.
- Local validation: Test on your patient mix and vendor stack (Hologic, GE, Siemens, Fuji). Compare this to your baseline recall rate, positive predictive value of recall (PPV1), cancer detection rate, and reading time. Commit to recalibration if drift occurs.
- Equity plan: Monitor false-positive and negative false-rates by age, race/ethnicity, and density; document corrective actions if disparities emerge. (This isn’t optional.)
- Workflow clarity: Is AI a second reader, an additional reader, or a triage tool? Who arbitrates discordance? What’s the escalation path for high-risk or interval cancer-like patterns?
- Regulatory strategy: Confirm whether the vendor has (or will file) a Predetermined Change Control Plan so models can be updated safely without repeated submissions. Also confirm how you’ll be notified about performance-relevant changes.
- Data governance: Audit logs of AI outputs, retention, protected health information handling, and the patient communication policy for AI-assisted reads.
After going live, set up a quarterly dashboard. It should include cancer detection rate per 1000 patients, recall rate, PPV1, interval cancer rate (as it matures), reading time, and turnaround time to diagnostic imaging or biopsy — all stratified by age, race/ethnicity, and density.
Here, I dissect what this discussion means through the lens of Moravec’s paradox (machines excel at what clinicians find hard, and vice versa) and offer a possible playbook for putting these tools to work.
What to Tell Patients
When speaking with patients, emphasize that a radiologist still reads their mammogram. AI helps with consistency and efficiency; it doesn’t replace human oversight. Patients with dense breasts should still expect a standard notice; discussion of individualized risk factors, such as family history, genetics, and prior biopsies; and consideration of supplemental imaging if risk warrants. But it’s also important to tell these patients that while dense breasts are common, they do not automatically mean high cancer risk.
As for screening schedules, remind patients that screening is at least biennial from 40 to 74 years of age per the USPSTF guidelines; however, specialty groups may recommend starting on an annual schedule at 40.
What You Can Implement Now
There are multiple practical use cases you can introduce now. One is to use AI as a second reader or an additional reader safety net to preserve detection while reducing human workload. This helps your breast center absorb screening expansion to age 40 without diluting quality. Another is to turn on AI triage to shorten the time to callback and biopsy for the few who need it most — patients notice and appreciate faster answers. You can also begin adopting automated density plus risk models to move beyond “dense/not dense.” For selected patients, AI-informed risk can justify MRI or tailored intervals.
Here’s a quick cheat sheet (for your next leadership or tumor-board meeting).
Do:
- Use AI as a second or additional reader or triage tool, not as a black box.
- Track cancer detection rate, recall, PPV1, interval cancers, and reading time, stratified by age, race, and breast density.
- Pair automated density with AI risk to personalize screening and supplemental imaging.
- Enroll patients in future clinical trials, such as PRISM, the first large-scale randomized controlled trial of AI for screening mammography. This US-based, $16 million, seven-site study is funded by the Patient-Centered Outcomes Research Institute.
Don’t:
- Assume “AI = CAD.” The 2015 CAD story is over; modern deep learning systems are different and require different oversight.
- Go live without a local validation and equity plan or without clarity on software updates.
- Forget to remind patients that screening starts at age 40, and dense breast notifications are now universal. Use the visit to discuss risk, supplemental imaging, and why a human still directs their care.
The Bottom Line
AI won’t replace radiologists or read mammograms for us — just as PET scans didn’t replace oncologists and stethoscopes didn’t make cardiologists obsolete. What it will do is catch what the tired human eye might miss, shave days off anxious waiting, and turn breast density into data instead of doubt. For oncologists, that means staging sooner, enrolling smarter, and spending more time talking with patients instead of chasing callbacks.
In short, AI may not take the picture, but it helps us frame the story, making it sharper, faster, and with fewer blind spots. By pairing this powerful technology with rigorous, equity-focused local validation and transparent governance under the FDA’s emerging Predetermined Change Control Plan framework, we can realize the tangible benefits of practical AI for our patients without widening disparities.
Now, during Breast Cancer Awareness Month, how about we add on AI to that pink ribbon — how cool would that be?
Thoughts? Drop me a line at [email protected]. Let’s keep the conversation — and pink ribbons — going.
Arturo Loaiza-Bonilla, MD, MSEd, is the co-founder and chief medical AI officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr Loaiza-Bonilla serves as Systemwide Chief of Hematology and Oncology at St. Luke’s University Health Network, where he maintains a connection to patient care by attending to patients 2 days a week.
A version of this article first appeared on Medscape.com.
In this Practical AI column, we’ve explored everything from large language models to the nuances of trial matching, but one of the most immediate and impactful applications of AI is unfolding right now in breast imaging. For oncologists, this isn’t an abstract future — with new screening guidelines, dense-breast mandates, and a shrinking radiology workforce, it’s the imaging reports and patient questions landing in your clinic today.
Here is what oncologists need to know, and how to put it to work for their patients.
Why AI in Mammography Matters
More than 200 million women undergo breast cancer screening each year. In the US alone, 10% of the 40 million women screened annually require additional diagnostic imaging, and 4%–5% of these women are eventually diagnosed with breast cancer.
Two major shifts are redefining breast cancer screening in the US: The US Preventive Services Task Force (USPSTF) now recommends biennial screening from age 40 to 74 years, and notifying patients of breast density is a federal requirement as of September 10, 2024. That means more mammograms, more patient questions, and more downstream oncology decisions. Patients will increasingly ask about “dense” breast results and what to do next. Add a national radiologist shortage into the mix, and the pressure on timely callbacks, biopsies, and treatment planning will only grow.
Can AI Help Without Compromising Care?
The short answer is yes. With AI, we may be able to transform these rate-limiting steps into opportunities for earlier detection, decentralized screening, and smarter triage and save hundreds of thousands of women from an unnecessary diagnostic procedure, if implemented deliberately.
Don’t Confuse Today’s AI With Yesterday’s CAD
Think of older computer-aided detection (CAD) like a 1990s chemotherapy drug: It sometimes helped, but it came with significant toxicity and rarely delivered consistent survival benefits. Today’s deep-learning AI is closer to targeted therapy — trained on millions of “trial participants” (mammograms), more precise, and applied in specific contexts where it adds value. If you once dismissed CAD as noise, it’s time to revisit what AI can now offer.
The role of AI is broader than drawing boxes. It provides second readings, worklist triage, risk prediction, density assessment, and decision support. FDA has cleared several AI tools for both 2D and digital breast tomosynthesis (DBT), which include iCAD ProFound (DBT), ScreenPoint Transpara (2D/DBT), and Lunit INSIGHT DBT.
Some of the strongest evidence for AI in mammography is as a second reader during screening. Large trials show that AI plus one radiologist can match reading from two radiologists, cutting workload by about 40%. For example, the MASAI randomized trial showed that AI-supported screening achieved similar cancer detection but cut human screen-reading workload about 44% vs standard double reading (39,996 vs 40,024 participants). The primary interval cancer outcomes are maturing, but the safety analysis is reassuring.
Reducing second reads and arbitration time are important for clinicians because it frees capacity for callbacks and diagnostic workups. This will be especially key given that screening now starts at age 40. That will mean about 21 to 22 million more women are newly eligible, translating to about 10 to 11 million additional mammograms each year under biennial screening.
Another important area where AI can make its mark in mammography is triage and time to diagnosis. The results from a randomized implementation study showed that AI-prioritized worklists accelerated time to additional imaging and biopsy diagnosis without harming efficiency for others — exactly the kind of outcome patients feel.
Multiple studies have demonstrated improved diagnostic performance and shorter reading times when AI supports DBT interpretation, which is important because DBT can otherwise be time intensive.
We are also seeing rapid advancement in risk-based screening, moving beyond a single dense vs not dense approach. Deep-learning risk models, such as Mirai, predict 1- to 5-year breast cancer risk directly from the mammogram, and these tools are now being assessed prospectively to guide supplemental MRI. Cost-effectiveness modeling supports risk-stratified intervals vs one-size-fits-all schedules.
Finally, automated density tools, such as Transpara Density and Volpara, offer objective, reproducible volumetric measures that map to the Breast Imaging-Reporting and Data System, which is useful for Mammography Quality Standards Act-required reporting and as inputs to risk calculators.
While early evidence suggests AI may help surface future or interval cancers earlier, including more invasive tumors, the definitive impacts on interval cancer rates and mortality require longitudinal follow-up, which is now in progress.
Pitfalls to Watch For
Bias is real. Studies show false-positive differences by race, age, and density. AI can even infer racial identity from images, potentially amplifying disparities. Performance can also shift by vendor, demographics, and prevalence.
A Radiology study of 4855 DBT exams showed that an algorithm produced more false-positive case scores in Black patients and older patients (aged 71-80 years) patients and in women with extremely dense breasts. This can happen because AI can infer proxies for race directly from images, even when humans cannot, and this can propagate disparities if not addressed. External validations and reviews emphasize that performance can shift with device manufacturer, demographics, and prevalence, which is why all tools need to undergo local validation and calibration.
Here’s a pragmatic adoption checklist before going live with an AI tool.
- Confirm FDA clearance: Verify the name and version of the algorithm, imaging modes (2D vs DBT), and operating points. Confirm 510(k) numbers.
- Local validation: Test on your patient mix and vendor stack (Hologic, GE, Siemens, Fuji). Compare this to your baseline recall rate, positive predictive value of recall (PPV1), cancer detection rate, and reading time. Commit to recalibration if drift occurs.
- Equity plan: Monitor false-positive and negative false-rates by age, race/ethnicity, and density; document corrective actions if disparities emerge. (This isn’t optional.)
- Workflow clarity: Is AI a second reader, an additional reader, or a triage tool? Who arbitrates discordance? What’s the escalation path for high-risk or interval cancer-like patterns?
- Regulatory strategy: Confirm whether the vendor has (or will file) a Predetermined Change Control Plan so models can be updated safely without repeated submissions. Also confirm how you’ll be notified about performance-relevant changes.
- Data governance: Audit logs of AI outputs, retention, protected health information handling, and the patient communication policy for AI-assisted reads.
After going live, set up a quarterly dashboard. It should include cancer detection rate per 1000 patients, recall rate, PPV1, interval cancer rate (as it matures), reading time, and turnaround time to diagnostic imaging or biopsy — all stratified by age, race/ethnicity, and density.
Here, I dissect what this discussion means through the lens of Moravec’s paradox (machines excel at what clinicians find hard, and vice versa) and offer a possible playbook for putting these tools to work.
What to Tell Patients
When speaking with patients, emphasize that a radiologist still reads their mammogram. AI helps with consistency and efficiency; it doesn’t replace human oversight. Patients with dense breasts should still expect a standard notice; discussion of individualized risk factors, such as family history, genetics, and prior biopsies; and consideration of supplemental imaging if risk warrants. But it’s also important to tell these patients that while dense breasts are common, they do not automatically mean high cancer risk.
As for screening schedules, remind patients that screening is at least biennial from 40 to 74 years of age per the USPSTF guidelines; however, specialty groups may recommend starting on an annual schedule at 40.
What You Can Implement Now
There are multiple practical use cases you can introduce now. One is to use AI as a second reader or an additional reader safety net to preserve detection while reducing human workload. This helps your breast center absorb screening expansion to age 40 without diluting quality. Another is to turn on AI triage to shorten the time to callback and biopsy for the few who need it most — patients notice and appreciate faster answers. You can also begin adopting automated density plus risk models to move beyond “dense/not dense.” For selected patients, AI-informed risk can justify MRI or tailored intervals.
Here’s a quick cheat sheet (for your next leadership or tumor-board meeting).
Do:
- Use AI as a second or additional reader or triage tool, not as a black box.
- Track cancer detection rate, recall, PPV1, interval cancers, and reading time, stratified by age, race, and breast density.
- Pair automated density with AI risk to personalize screening and supplemental imaging.
- Enroll patients in future clinical trials, such as PRISM, the first large-scale randomized controlled trial of AI for screening mammography. This US-based, $16 million, seven-site study is funded by the Patient-Centered Outcomes Research Institute.
Don’t:
- Assume “AI = CAD.” The 2015 CAD story is over; modern deep learning systems are different and require different oversight.
- Go live without a local validation and equity plan or without clarity on software updates.
- Forget to remind patients that screening starts at age 40, and dense breast notifications are now universal. Use the visit to discuss risk, supplemental imaging, and why a human still directs their care.
The Bottom Line
AI won’t replace radiologists or read mammograms for us — just as PET scans didn’t replace oncologists and stethoscopes didn’t make cardiologists obsolete. What it will do is catch what the tired human eye might miss, shave days off anxious waiting, and turn breast density into data instead of doubt. For oncologists, that means staging sooner, enrolling smarter, and spending more time talking with patients instead of chasing callbacks.
In short, AI may not take the picture, but it helps us frame the story, making it sharper, faster, and with fewer blind spots. By pairing this powerful technology with rigorous, equity-focused local validation and transparent governance under the FDA’s emerging Predetermined Change Control Plan framework, we can realize the tangible benefits of practical AI for our patients without widening disparities.
Now, during Breast Cancer Awareness Month, how about we add on AI to that pink ribbon — how cool would that be?
Thoughts? Drop me a line at [email protected]. Let’s keep the conversation — and pink ribbons — going.
Arturo Loaiza-Bonilla, MD, MSEd, is the co-founder and chief medical AI officer at Massive Bio, a company connecting patients to clinical trials using artificial intelligence. His research and professional interests focus on precision medicine, clinical trial design, digital health, entrepreneurship, and patient advocacy. Dr Loaiza-Bonilla serves as Systemwide Chief of Hematology and Oncology at St. Luke’s University Health Network, where he maintains a connection to patient care by attending to patients 2 days a week.
A version of this article first appeared on Medscape.com.
Unique Presentation of Postpartum Hypereosinophilic Syndrome With Atypical Features and Therapeutic Challenges
Unique Presentation of Postpartum Hypereosinophilic Syndrome With Atypical Features and Therapeutic Challenges
Hypereosinophilic syndrome (HES) is defined by marked, persistent absolute eosinophil count (AEC) > 1500 cells/μL on ≥ 2 peripheral smears separated by ≥ 1 month with evidence of accompanied end-organ damage, in the absence of other causes of eosinophilia such as malignancy, atopy, or parasitic infections.1-5 Hypereosinophilic infiltration can impact almost every organ system; however, the most profound complications in patients with HES are related to leukemias and cardiac manifestations of the disease.3,4 Although rare, the associated morbidity and mortality of HES are considerable, making prompt recognition and treatment essential. Management involves targeted therapy based on pathologic classification of HES and on decreasing associated inflammation, fibrosis, and end-organ damage.3,5-7
The patient in this case report met the diagnostic criteria for HES. However, this patient had several clinical and laboratory features that made it difficult to characterize a specific HES variant. Moreover, she had additional immunomodulating factors in the setting of pregnancy. This is the first documented case of HES of undetermined etiology diagnosed postpartum and managed in the setting of a new pregnancy.2,8
CASE PRESENTATION
A 32-year-old female active-duty military service member with allergic rhinitis and a history of childhood eczema was referred to allergy/immunology for evaluation of a new, progressive pruritic rash. Symptoms started 3 months after the birth of her first child, with a new diffuse erythematous skin rash sparing her palms, soles, and mucosal surfaces. Given her history of atopy, the rash was initially treated as severe atopic dermatitis with appropriate topical medications. The rash gradually worsened, with the development of intermittent facial swelling, night sweats, dyspnea, recurrent epigastric abdominal pain, and nausea with vomiting, resulting in decreased oral intake and weight loss.
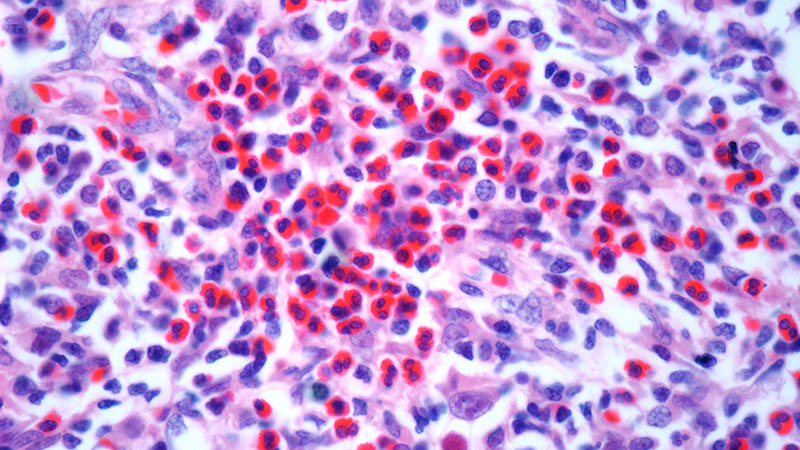
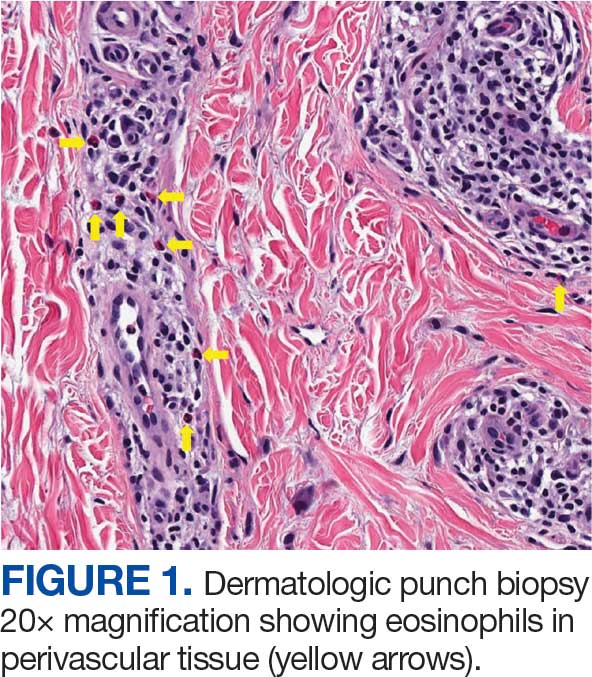
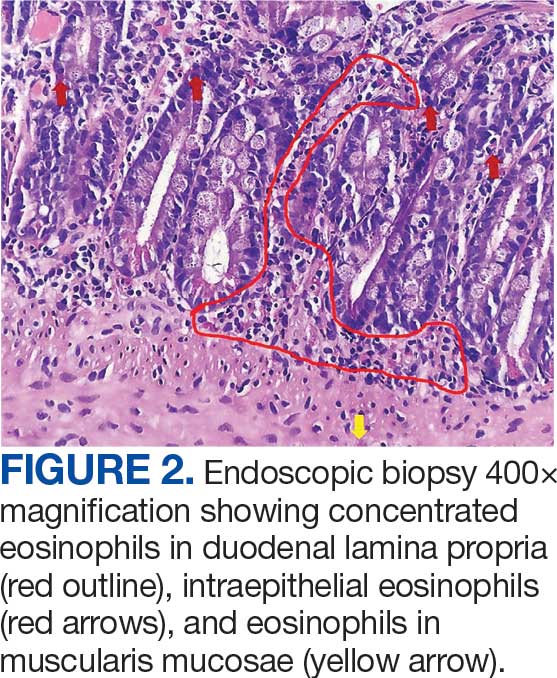
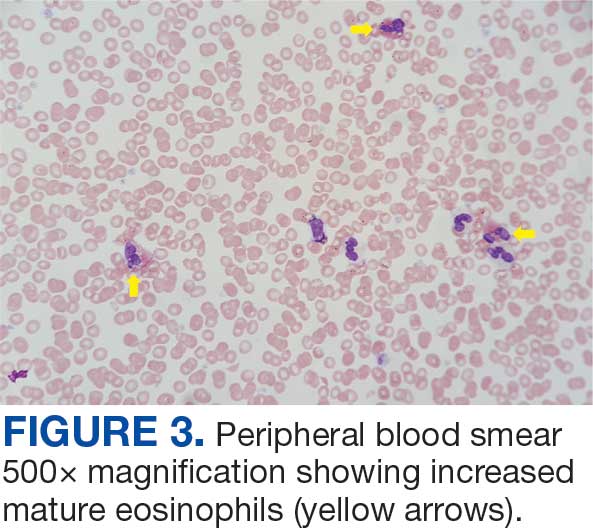
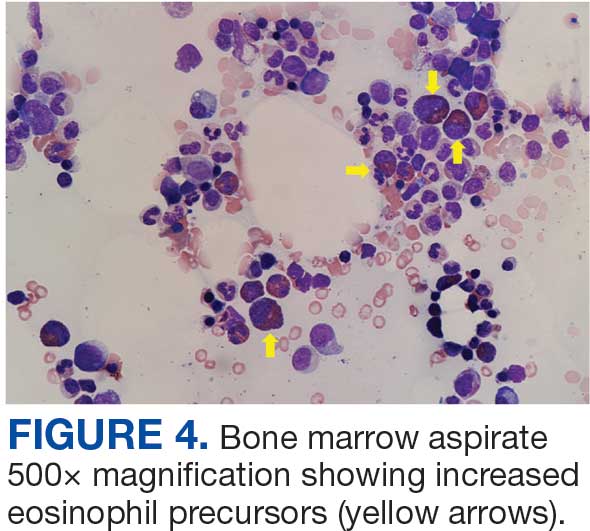
The patient was hospitalized and received an expedited multidisciplinary evaluation by dermatology, hematology/oncology, and gastroenterology. Her AEC of 4787 cells/μL peaked on admission and was markedly elevated from the 1070 cells/μL reported in the third trimester of her pregnancy. She was found to have mature eosinophilia on skin biopsy (Figure 1), endoscopic duodenal biopsy (Figure 2), peripheral blood smear (Figure 3), and bone marrow biopsy (Figure 4).
Radiographic imaging of the chest, abdomen, and pelvis revealed hepatomegaly without detectable neoplasm. There was no clinical evidence of cardiac involvement, and evaluation with electrocardiography and echocardiography did not indicate myocarditis. Extensive laboratory testing revealed no genetic mutations indicative of familial, myeloproliferative, or lymphocytic variants of HES.
The patient received topical emollients, omeprazole 40 mg daily, and ondansetron 8 mg 3 times daily as needed for symptom management, and was started on oral prednisone 40 mg daily with improvement in dyspnea, night sweats, and gastrointestinal complaints. During the patient's 6-day hospitalization and treatment, her AECs gradually decreased to 2110 cells/μL, and decreased to 1600 cells/μL over the course of a month, remaining in the hypereosinophilic range. The patient was discovered to be pregnant while symptoms were improving, resulting in stepwise discontinuation of oral steroids, but she reported continued improvement in symptoms.
DISCUSSION
Peripheral eosinophilia has a broad differential diagnoses, including HES, parasitic infections, atopic hypersensitivity diseases, eosinophilic lung diseases, eosinophilic gastrointestinal diseases, vasculitides such as eosinophilic granulomatosis with polyangiitis, genetic syndromes predisposing to eosinophilia, episodic angioedema with eosinophilia, and chronic metabolic disease with adrenal insufficiency.1-5 HES, although rare, is a disease process with potentially devastating associated morbidity and mortality if not promptly recognized and treated. HES is further delineated by hypereosinophilia with associated eosinophil-mediated organ damage or dysfunction.3-5
Clinical manifestations of HES can differ greatly depending on the HES variant and degree of organ involvement at the time of diagnosis and throughout the disease course. Patients with HES, as well as those with asymptomatic eosinophilia or hypereosinophilia, should be closely monitored for disease progression. In addition to trending peripheral AECs, clinicians should screen for symptoms of organ involvement and perform targeted evaluation of the suspected organs to promptly identify early signs of organ involvement and initiate treatment.1-4 Recommendations regarding screening intervals vary widely from monthly to annually, depending on a patient’s specific clinical picture.
HES has been subdivided into clinically relevant variants, including myeloproliferative (M-HES), T lymphocytic (L-HES), organ-restricted (or overlap) HES, familial HES, idiopathic HES, and specific syndromes with associated hypereosinophilia.3-5,9 Patients with M-HES have elevated circulating leukocyte precursors and clinical manifestations, including but not limited to hepatosplenomegaly, anemia, and thrombocytopenia. The most commonly associated genetic mutations include the FIP1L1-PDGFR-α fusion, BCR-ABL1, PDGFRA/B, JAK2, KIT, and FGFR1.3-6 L-HES usually has predominant skin and soft tissue involvement secondary to immunoglobulin E-mediated actions with clonal expansion of T cells (most commonly CD3-4+ or CD3+CD4-CD8-).3,5,6 Familial HES, a rare variant, follows an autosomal dominant inheritance pattern and is usually present at birth. It involves chromosome 5, which contains genes coding for cytokines that drive eosinophilic proliferation, including interleukin (IL)-3, IL-5, and granulocyte-macrophage colony-stimulating factor.5,9 Hypereosinophilia in the setting of end-organ damage restricted to a single organ is considered organ-restricted HES. There can be significant hepatic and gastrointestinal dysfunction, with or without malabsorption.
HES can also manifest with hematologic malignancy, restrictive obliterative cardiomyopathies, renal injury manifested by hematuria and electrolyte derangements, and neurologic complications including hemiparesis, dysarthria, and even coma.6 Endothelial damage due to eosinophil-driven inflammation can result in thrombus formation and increased risk of thromboembolic events in various organs.3 Idiopathic HES, otherwise known as HES of unknown etiology or significance, is a diagnosis of exclusion and constitutes a cohort of patients who do not fit into the other delineated categories.3-5 These patients often have multisystem involvement, making classification and treatment a challenge.5
The patient described in this case met the diagnostic criteria for HES, but her complicated clinical and laboratory features were challenging to characterize into a specific variant of HES. Organ-restricted HES was ruled out due to skin, marrow, and duodenal infiltration. She also had the potential for lung involvement based on her clinical symptoms, however no biopsy was obtained. Laboratory testing revealed no deletions or mutations indicative of familial, myeloproliferative, or lymphocytic variants. Her multisystem involvement without an underlying associated syndrome suggests idiopathic HES or HES of undetermined significance.1-5
Most patients with HES are diagnosed between the ages of 20 and 50 years.10 While HES has its peak incidence in the fourth decade of life, acute onset of new symptoms 3 months postpartum makes this an unusual presentation. In this unique case, it is important to highlight the role of the physiologic changes of pregnancy in inflammatory mediation. The physiologic changes that occur in pregnancy to ensure fetal tolerance can have profound implications for leukocyte count, AEC, and subsequent inflammatory responses. The phenomenon of inflammatory amelioration during pregnancy is well-documented, but there has only been 1 known published case report discussing decreasing HES symptoms during pregnancy with prepregnancy and postpartum hypereosinophilia.8 It is suggested that this amelioration is secondary to cortisol and progesterone shifts that occur in pregnancy. Physiologic increases in adrenocorticotropic hormone in pregnancy leads to subsequent secretion of endogenous steroids by the adrenal cortex. In turn, pregnancy can lead to leukocytosis and eosinopenia.8 Overall, pregnancy can have beneficial immunomodulating properties in the spectrum of hypereosinophilic syndromes. Even so, this patient with HES diagnosed postpartum remains at risk for the sequelae of hypereosinophilia, regardless of potential for AEC reduction during pregnancy. Therefore, treatment considerations need to be made with the safety of the maternal-fetal dyad as a priority.
Treatment
The treatment of symptomatic HES without acute life-threatening features or associated malignancy is generally determined by clinical variant.2-4 There is insufficient data to support initiation of treatment solely based on persistently elevated AEC. Patients with peripheral eosinophilia and hypereosinophilia should be monitored periodically with appropriate subspecialist evaluation for occult end-organ involvement, and targeted therapies should be deferred until an HES diagnosis.1-4 First-line therapy in most HES variants is systemic glucocorticoids.2,3,7 Since the disease course for this patient did not precisely match an HES variant, it was challenging to ascertain the optimal personalized treatment regimen. The approach to therapy was further complicated by newly identified pregnancy necessitating cessation of systemic glucocorticoids. In addition to glucocorticoids, hydroxyurea and interferon-α are among treatments historically used for HES, with tyrosine kinase inhibitors and monoclonal antibodies targeting IL-5 becoming more common.1-4 Although this patient may ultimately benefit from an IL-5 targeting biologic medication such as mepolizumab, safety in pregnancy is not well-studied and may require close clinical monitoring with treatment deferred until after delivery if possible.3,7,8,11
Military service members with frequent geographic relocation have additional barriers to timely diagnosis with often-limited access to subspecialty care depending on the duty station. While the patient was able to receive care at a large military medical center with many subspecialists, prompt recognition and timely referral to specialists would be even more critical at a smaller treatment facility. Depending on the severity and variant of HES, patients may warrant evaluation and treatment by hematology/oncology, cardiology, pulmonology, and immunology. Although HES can present in young children and older adults, this condition is most often diagnosed during the third and fourth decades of life, putting clinicians on the front line of hypereosinophilia identification and evaluation.10 Military physicians have the additional duty to not only think ahead in their diverse clinical settings to ensure proper care for patients, but also maintain a broad differential inclusive of more rare disease processes such as HES.
CONCLUSIONS
This case emphasizes how uncontrolled or untreated HES can lead to significant end-organ damage involving multiple systems and high morbidity. Prompt recognition of hypereosinophilia with potential HES can help expedite coordination of multidisciplinary care across multiple specialties to minimize delays in diagnosis and treatment. Doing so may minimize associated morbidity and mortality, especially in individuals located at more remote duty stations or deployed to austere environments.
- Cogan E, Roufosse F. Clinical management of the hypereosinophilic syndromes. Expert Rev Hematol. 2012;5:275-290. doi: 10.1586/ehm.12.14
- Klion A. Hypereosinophilic syndrome: approach to treatment in the era of precision medicine. Hematology Am Soc Hematol Educ Program. 2018;2018:326-331. doi:10.1182/asheducation-2018.1.326
- Shomali W, Gotlib J. World health organization-defined eosinophilic disorders: 2022 update on diagnosis, risk stratification, and management. Am J Hematol. 2022;97:129-148. doi:10.1002/ajh.26352
- Helbig G, Klion AD. Hypereosinophilic syndromes - an enigmatic group of disorders with an intriguing clinical spectrum and challenging treatment. Blood Rev. 2021;49:100809. doi:10.1016/j.blre.2021.100809
- Valent P, Klion AD, Horny HP, et al. Contemporary consensus proposal on criteria and classification of eosinophilic disorders and related syndromes. J Allergy Clin Immunol. 2012;130:607-612.e9. doi:10.1016/j.jaci.2012.02.019
- Roufosse FE, Goldman M, Cogan E. Hypereosinophilic syndromes. Orphanet J Rare Dis. 2007;2:37. doi:10.1186/1750-1172-2-37
- Pitlick MM, Li JT, Pongdee T. Current and emerging biologic therapies targeting eosinophilic disorders. World Allergy Organ J. 2022;15:100676. doi:10.1016/j.waojou.2022.10067
- Ault P, Cortes J, Lynn A, Keating M, Verstovsek S. Pregnancy in a patient with hypereosinophilic syndrome. Leuk Res. 2009;33:186-187. doi:10.1016/j.leukres.2008.05.013
- Rioux JD, Stone VA, Daly MJ, et al. Familial eosinophilia maps to the cytokine gene cluster on human chromosomal region 5q31-q33. Am J Hum Genet. 1998;63:1086-1094. doi:10.1086/302053
- Williams KW, Ware J, Abiodun A, et al. Hypereosinophilia in children and adults: a retrospective comparison. J Allergy Clin Immunol Pract. 2016;4:941-947.e1. doi:10.1016/j.jaip.2016.03.020
- Pane F, Lefevre G, Kwon N, et al. Characterization of disease flares and impact of mepolizumab in patients with hypereosinophilic syndrome. Front Immunol. 2022;13:935996. doi:10.3389/fimmu.2022.935996
Hypereosinophilic syndrome (HES) is defined by marked, persistent absolute eosinophil count (AEC) > 1500 cells/μL on ≥ 2 peripheral smears separated by ≥ 1 month with evidence of accompanied end-organ damage, in the absence of other causes of eosinophilia such as malignancy, atopy, or parasitic infections.1-5 Hypereosinophilic infiltration can impact almost every organ system; however, the most profound complications in patients with HES are related to leukemias and cardiac manifestations of the disease.3,4 Although rare, the associated morbidity and mortality of HES are considerable, making prompt recognition and treatment essential. Management involves targeted therapy based on pathologic classification of HES and on decreasing associated inflammation, fibrosis, and end-organ damage.3,5-7
The patient in this case report met the diagnostic criteria for HES. However, this patient had several clinical and laboratory features that made it difficult to characterize a specific HES variant. Moreover, she had additional immunomodulating factors in the setting of pregnancy. This is the first documented case of HES of undetermined etiology diagnosed postpartum and managed in the setting of a new pregnancy.2,8
CASE PRESENTATION
A 32-year-old female active-duty military service member with allergic rhinitis and a history of childhood eczema was referred to allergy/immunology for evaluation of a new, progressive pruritic rash. Symptoms started 3 months after the birth of her first child, with a new diffuse erythematous skin rash sparing her palms, soles, and mucosal surfaces. Given her history of atopy, the rash was initially treated as severe atopic dermatitis with appropriate topical medications. The rash gradually worsened, with the development of intermittent facial swelling, night sweats, dyspnea, recurrent epigastric abdominal pain, and nausea with vomiting, resulting in decreased oral intake and weight loss.
The patient was hospitalized and received an expedited multidisciplinary evaluation by dermatology, hematology/oncology, and gastroenterology. Her AEC of 4787 cells/μL peaked on admission and was markedly elevated from the 1070 cells/μL reported in the third trimester of her pregnancy. She was found to have mature eosinophilia on skin biopsy (Figure 1), endoscopic duodenal biopsy (Figure 2), peripheral blood smear (Figure 3), and bone marrow biopsy (Figure 4).




Radiographic imaging of the chest, abdomen, and pelvis revealed hepatomegaly without detectable neoplasm. There was no clinical evidence of cardiac involvement, and evaluation with electrocardiography and echocardiography did not indicate myocarditis. Extensive laboratory testing revealed no genetic mutations indicative of familial, myeloproliferative, or lymphocytic variants of HES.
The patient received topical emollients, omeprazole 40 mg daily, and ondansetron 8 mg 3 times daily as needed for symptom management, and was started on oral prednisone 40 mg daily with improvement in dyspnea, night sweats, and gastrointestinal complaints. During the patient's 6-day hospitalization and treatment, her AECs gradually decreased to 2110 cells/μL, and decreased to 1600 cells/μL over the course of a month, remaining in the hypereosinophilic range. The patient was discovered to be pregnant while symptoms were improving, resulting in stepwise discontinuation of oral steroids, but she reported continued improvement in symptoms.
DISCUSSION
Peripheral eosinophilia has a broad differential diagnoses, including HES, parasitic infections, atopic hypersensitivity diseases, eosinophilic lung diseases, eosinophilic gastrointestinal diseases, vasculitides such as eosinophilic granulomatosis with polyangiitis, genetic syndromes predisposing to eosinophilia, episodic angioedema with eosinophilia, and chronic metabolic disease with adrenal insufficiency.1-5 HES, although rare, is a disease process with potentially devastating associated morbidity and mortality if not promptly recognized and treated. HES is further delineated by hypereosinophilia with associated eosinophil-mediated organ damage or dysfunction.3-5
Clinical manifestations of HES can differ greatly depending on the HES variant and degree of organ involvement at the time of diagnosis and throughout the disease course. Patients with HES, as well as those with asymptomatic eosinophilia or hypereosinophilia, should be closely monitored for disease progression. In addition to trending peripheral AECs, clinicians should screen for symptoms of organ involvement and perform targeted evaluation of the suspected organs to promptly identify early signs of organ involvement and initiate treatment.1-4 Recommendations regarding screening intervals vary widely from monthly to annually, depending on a patient’s specific clinical picture.
HES has been subdivided into clinically relevant variants, including myeloproliferative (M-HES), T lymphocytic (L-HES), organ-restricted (or overlap) HES, familial HES, idiopathic HES, and specific syndromes with associated hypereosinophilia.3-5,9 Patients with M-HES have elevated circulating leukocyte precursors and clinical manifestations, including but not limited to hepatosplenomegaly, anemia, and thrombocytopenia. The most commonly associated genetic mutations include the FIP1L1-PDGFR-α fusion, BCR-ABL1, PDGFRA/B, JAK2, KIT, and FGFR1.3-6 L-HES usually has predominant skin and soft tissue involvement secondary to immunoglobulin E-mediated actions with clonal expansion of T cells (most commonly CD3-4+ or CD3+CD4-CD8-).3,5,6 Familial HES, a rare variant, follows an autosomal dominant inheritance pattern and is usually present at birth. It involves chromosome 5, which contains genes coding for cytokines that drive eosinophilic proliferation, including interleukin (IL)-3, IL-5, and granulocyte-macrophage colony-stimulating factor.5,9 Hypereosinophilia in the setting of end-organ damage restricted to a single organ is considered organ-restricted HES. There can be significant hepatic and gastrointestinal dysfunction, with or without malabsorption.
HES can also manifest with hematologic malignancy, restrictive obliterative cardiomyopathies, renal injury manifested by hematuria and electrolyte derangements, and neurologic complications including hemiparesis, dysarthria, and even coma.6 Endothelial damage due to eosinophil-driven inflammation can result in thrombus formation and increased risk of thromboembolic events in various organs.3 Idiopathic HES, otherwise known as HES of unknown etiology or significance, is a diagnosis of exclusion and constitutes a cohort of patients who do not fit into the other delineated categories.3-5 These patients often have multisystem involvement, making classification and treatment a challenge.5
The patient described in this case met the diagnostic criteria for HES, but her complicated clinical and laboratory features were challenging to characterize into a specific variant of HES. Organ-restricted HES was ruled out due to skin, marrow, and duodenal infiltration. She also had the potential for lung involvement based on her clinical symptoms, however no biopsy was obtained. Laboratory testing revealed no deletions or mutations indicative of familial, myeloproliferative, or lymphocytic variants. Her multisystem involvement without an underlying associated syndrome suggests idiopathic HES or HES of undetermined significance.1-5
Most patients with HES are diagnosed between the ages of 20 and 50 years.10 While HES has its peak incidence in the fourth decade of life, acute onset of new symptoms 3 months postpartum makes this an unusual presentation. In this unique case, it is important to highlight the role of the physiologic changes of pregnancy in inflammatory mediation. The physiologic changes that occur in pregnancy to ensure fetal tolerance can have profound implications for leukocyte count, AEC, and subsequent inflammatory responses. The phenomenon of inflammatory amelioration during pregnancy is well-documented, but there has only been 1 known published case report discussing decreasing HES symptoms during pregnancy with prepregnancy and postpartum hypereosinophilia.8 It is suggested that this amelioration is secondary to cortisol and progesterone shifts that occur in pregnancy. Physiologic increases in adrenocorticotropic hormone in pregnancy leads to subsequent secretion of endogenous steroids by the adrenal cortex. In turn, pregnancy can lead to leukocytosis and eosinopenia.8 Overall, pregnancy can have beneficial immunomodulating properties in the spectrum of hypereosinophilic syndromes. Even so, this patient with HES diagnosed postpartum remains at risk for the sequelae of hypereosinophilia, regardless of potential for AEC reduction during pregnancy. Therefore, treatment considerations need to be made with the safety of the maternal-fetal dyad as a priority.
Treatment
The treatment of symptomatic HES without acute life-threatening features or associated malignancy is generally determined by clinical variant.2-4 There is insufficient data to support initiation of treatment solely based on persistently elevated AEC. Patients with peripheral eosinophilia and hypereosinophilia should be monitored periodically with appropriate subspecialist evaluation for occult end-organ involvement, and targeted therapies should be deferred until an HES diagnosis.1-4 First-line therapy in most HES variants is systemic glucocorticoids.2,3,7 Since the disease course for this patient did not precisely match an HES variant, it was challenging to ascertain the optimal personalized treatment regimen. The approach to therapy was further complicated by newly identified pregnancy necessitating cessation of systemic glucocorticoids. In addition to glucocorticoids, hydroxyurea and interferon-α are among treatments historically used for HES, with tyrosine kinase inhibitors and monoclonal antibodies targeting IL-5 becoming more common.1-4 Although this patient may ultimately benefit from an IL-5 targeting biologic medication such as mepolizumab, safety in pregnancy is not well-studied and may require close clinical monitoring with treatment deferred until after delivery if possible.3,7,8,11
Military service members with frequent geographic relocation have additional barriers to timely diagnosis with often-limited access to subspecialty care depending on the duty station. While the patient was able to receive care at a large military medical center with many subspecialists, prompt recognition and timely referral to specialists would be even more critical at a smaller treatment facility. Depending on the severity and variant of HES, patients may warrant evaluation and treatment by hematology/oncology, cardiology, pulmonology, and immunology. Although HES can present in young children and older adults, this condition is most often diagnosed during the third and fourth decades of life, putting clinicians on the front line of hypereosinophilia identification and evaluation.10 Military physicians have the additional duty to not only think ahead in their diverse clinical settings to ensure proper care for patients, but also maintain a broad differential inclusive of more rare disease processes such as HES.
CONCLUSIONS
This case emphasizes how uncontrolled or untreated HES can lead to significant end-organ damage involving multiple systems and high morbidity. Prompt recognition of hypereosinophilia with potential HES can help expedite coordination of multidisciplinary care across multiple specialties to minimize delays in diagnosis and treatment. Doing so may minimize associated morbidity and mortality, especially in individuals located at more remote duty stations or deployed to austere environments.
Hypereosinophilic syndrome (HES) is defined by marked, persistent absolute eosinophil count (AEC) > 1500 cells/μL on ≥ 2 peripheral smears separated by ≥ 1 month with evidence of accompanied end-organ damage, in the absence of other causes of eosinophilia such as malignancy, atopy, or parasitic infections.1-5 Hypereosinophilic infiltration can impact almost every organ system; however, the most profound complications in patients with HES are related to leukemias and cardiac manifestations of the disease.3,4 Although rare, the associated morbidity and mortality of HES are considerable, making prompt recognition and treatment essential. Management involves targeted therapy based on pathologic classification of HES and on decreasing associated inflammation, fibrosis, and end-organ damage.3,5-7
The patient in this case report met the diagnostic criteria for HES. However, this patient had several clinical and laboratory features that made it difficult to characterize a specific HES variant. Moreover, she had additional immunomodulating factors in the setting of pregnancy. This is the first documented case of HES of undetermined etiology diagnosed postpartum and managed in the setting of a new pregnancy.2,8
CASE PRESENTATION
A 32-year-old female active-duty military service member with allergic rhinitis and a history of childhood eczema was referred to allergy/immunology for evaluation of a new, progressive pruritic rash. Symptoms started 3 months after the birth of her first child, with a new diffuse erythematous skin rash sparing her palms, soles, and mucosal surfaces. Given her history of atopy, the rash was initially treated as severe atopic dermatitis with appropriate topical medications. The rash gradually worsened, with the development of intermittent facial swelling, night sweats, dyspnea, recurrent epigastric abdominal pain, and nausea with vomiting, resulting in decreased oral intake and weight loss.
The patient was hospitalized and received an expedited multidisciplinary evaluation by dermatology, hematology/oncology, and gastroenterology. Her AEC of 4787 cells/μL peaked on admission and was markedly elevated from the 1070 cells/μL reported in the third trimester of her pregnancy. She was found to have mature eosinophilia on skin biopsy (Figure 1), endoscopic duodenal biopsy (Figure 2), peripheral blood smear (Figure 3), and bone marrow biopsy (Figure 4).




Radiographic imaging of the chest, abdomen, and pelvis revealed hepatomegaly without detectable neoplasm. There was no clinical evidence of cardiac involvement, and evaluation with electrocardiography and echocardiography did not indicate myocarditis. Extensive laboratory testing revealed no genetic mutations indicative of familial, myeloproliferative, or lymphocytic variants of HES.
The patient received topical emollients, omeprazole 40 mg daily, and ondansetron 8 mg 3 times daily as needed for symptom management, and was started on oral prednisone 40 mg daily with improvement in dyspnea, night sweats, and gastrointestinal complaints. During the patient's 6-day hospitalization and treatment, her AECs gradually decreased to 2110 cells/μL, and decreased to 1600 cells/μL over the course of a month, remaining in the hypereosinophilic range. The patient was discovered to be pregnant while symptoms were improving, resulting in stepwise discontinuation of oral steroids, but she reported continued improvement in symptoms.
DISCUSSION
Peripheral eosinophilia has a broad differential diagnoses, including HES, parasitic infections, atopic hypersensitivity diseases, eosinophilic lung diseases, eosinophilic gastrointestinal diseases, vasculitides such as eosinophilic granulomatosis with polyangiitis, genetic syndromes predisposing to eosinophilia, episodic angioedema with eosinophilia, and chronic metabolic disease with adrenal insufficiency.1-5 HES, although rare, is a disease process with potentially devastating associated morbidity and mortality if not promptly recognized and treated. HES is further delineated by hypereosinophilia with associated eosinophil-mediated organ damage or dysfunction.3-5
Clinical manifestations of HES can differ greatly depending on the HES variant and degree of organ involvement at the time of diagnosis and throughout the disease course. Patients with HES, as well as those with asymptomatic eosinophilia or hypereosinophilia, should be closely monitored for disease progression. In addition to trending peripheral AECs, clinicians should screen for symptoms of organ involvement and perform targeted evaluation of the suspected organs to promptly identify early signs of organ involvement and initiate treatment.1-4 Recommendations regarding screening intervals vary widely from monthly to annually, depending on a patient’s specific clinical picture.
HES has been subdivided into clinically relevant variants, including myeloproliferative (M-HES), T lymphocytic (L-HES), organ-restricted (or overlap) HES, familial HES, idiopathic HES, and specific syndromes with associated hypereosinophilia.3-5,9 Patients with M-HES have elevated circulating leukocyte precursors and clinical manifestations, including but not limited to hepatosplenomegaly, anemia, and thrombocytopenia. The most commonly associated genetic mutations include the FIP1L1-PDGFR-α fusion, BCR-ABL1, PDGFRA/B, JAK2, KIT, and FGFR1.3-6 L-HES usually has predominant skin and soft tissue involvement secondary to immunoglobulin E-mediated actions with clonal expansion of T cells (most commonly CD3-4+ or CD3+CD4-CD8-).3,5,6 Familial HES, a rare variant, follows an autosomal dominant inheritance pattern and is usually present at birth. It involves chromosome 5, which contains genes coding for cytokines that drive eosinophilic proliferation, including interleukin (IL)-3, IL-5, and granulocyte-macrophage colony-stimulating factor.5,9 Hypereosinophilia in the setting of end-organ damage restricted to a single organ is considered organ-restricted HES. There can be significant hepatic and gastrointestinal dysfunction, with or without malabsorption.
HES can also manifest with hematologic malignancy, restrictive obliterative cardiomyopathies, renal injury manifested by hematuria and electrolyte derangements, and neurologic complications including hemiparesis, dysarthria, and even coma.6 Endothelial damage due to eosinophil-driven inflammation can result in thrombus formation and increased risk of thromboembolic events in various organs.3 Idiopathic HES, otherwise known as HES of unknown etiology or significance, is a diagnosis of exclusion and constitutes a cohort of patients who do not fit into the other delineated categories.3-5 These patients often have multisystem involvement, making classification and treatment a challenge.5
The patient described in this case met the diagnostic criteria for HES, but her complicated clinical and laboratory features were challenging to characterize into a specific variant of HES. Organ-restricted HES was ruled out due to skin, marrow, and duodenal infiltration. She also had the potential for lung involvement based on her clinical symptoms, however no biopsy was obtained. Laboratory testing revealed no deletions or mutations indicative of familial, myeloproliferative, or lymphocytic variants. Her multisystem involvement without an underlying associated syndrome suggests idiopathic HES or HES of undetermined significance.1-5
Most patients with HES are diagnosed between the ages of 20 and 50 years.10 While HES has its peak incidence in the fourth decade of life, acute onset of new symptoms 3 months postpartum makes this an unusual presentation. In this unique case, it is important to highlight the role of the physiologic changes of pregnancy in inflammatory mediation. The physiologic changes that occur in pregnancy to ensure fetal tolerance can have profound implications for leukocyte count, AEC, and subsequent inflammatory responses. The phenomenon of inflammatory amelioration during pregnancy is well-documented, but there has only been 1 known published case report discussing decreasing HES symptoms during pregnancy with prepregnancy and postpartum hypereosinophilia.8 It is suggested that this amelioration is secondary to cortisol and progesterone shifts that occur in pregnancy. Physiologic increases in adrenocorticotropic hormone in pregnancy leads to subsequent secretion of endogenous steroids by the adrenal cortex. In turn, pregnancy can lead to leukocytosis and eosinopenia.8 Overall, pregnancy can have beneficial immunomodulating properties in the spectrum of hypereosinophilic syndromes. Even so, this patient with HES diagnosed postpartum remains at risk for the sequelae of hypereosinophilia, regardless of potential for AEC reduction during pregnancy. Therefore, treatment considerations need to be made with the safety of the maternal-fetal dyad as a priority.
Treatment
The treatment of symptomatic HES without acute life-threatening features or associated malignancy is generally determined by clinical variant.2-4 There is insufficient data to support initiation of treatment solely based on persistently elevated AEC. Patients with peripheral eosinophilia and hypereosinophilia should be monitored periodically with appropriate subspecialist evaluation for occult end-organ involvement, and targeted therapies should be deferred until an HES diagnosis.1-4 First-line therapy in most HES variants is systemic glucocorticoids.2,3,7 Since the disease course for this patient did not precisely match an HES variant, it was challenging to ascertain the optimal personalized treatment regimen. The approach to therapy was further complicated by newly identified pregnancy necessitating cessation of systemic glucocorticoids. In addition to glucocorticoids, hydroxyurea and interferon-α are among treatments historically used for HES, with tyrosine kinase inhibitors and monoclonal antibodies targeting IL-5 becoming more common.1-4 Although this patient may ultimately benefit from an IL-5 targeting biologic medication such as mepolizumab, safety in pregnancy is not well-studied and may require close clinical monitoring with treatment deferred until after delivery if possible.3,7,8,11
Military service members with frequent geographic relocation have additional barriers to timely diagnosis with often-limited access to subspecialty care depending on the duty station. While the patient was able to receive care at a large military medical center with many subspecialists, prompt recognition and timely referral to specialists would be even more critical at a smaller treatment facility. Depending on the severity and variant of HES, patients may warrant evaluation and treatment by hematology/oncology, cardiology, pulmonology, and immunology. Although HES can present in young children and older adults, this condition is most often diagnosed during the third and fourth decades of life, putting clinicians on the front line of hypereosinophilia identification and evaluation.10 Military physicians have the additional duty to not only think ahead in their diverse clinical settings to ensure proper care for patients, but also maintain a broad differential inclusive of more rare disease processes such as HES.
CONCLUSIONS
This case emphasizes how uncontrolled or untreated HES can lead to significant end-organ damage involving multiple systems and high morbidity. Prompt recognition of hypereosinophilia with potential HES can help expedite coordination of multidisciplinary care across multiple specialties to minimize delays in diagnosis and treatment. Doing so may minimize associated morbidity and mortality, especially in individuals located at more remote duty stations or deployed to austere environments.
- Cogan E, Roufosse F. Clinical management of the hypereosinophilic syndromes. Expert Rev Hematol. 2012;5:275-290. doi: 10.1586/ehm.12.14
- Klion A. Hypereosinophilic syndrome: approach to treatment in the era of precision medicine. Hematology Am Soc Hematol Educ Program. 2018;2018:326-331. doi:10.1182/asheducation-2018.1.326
- Shomali W, Gotlib J. World health organization-defined eosinophilic disorders: 2022 update on diagnosis, risk stratification, and management. Am J Hematol. 2022;97:129-148. doi:10.1002/ajh.26352
- Helbig G, Klion AD. Hypereosinophilic syndromes - an enigmatic group of disorders with an intriguing clinical spectrum and challenging treatment. Blood Rev. 2021;49:100809. doi:10.1016/j.blre.2021.100809
- Valent P, Klion AD, Horny HP, et al. Contemporary consensus proposal on criteria and classification of eosinophilic disorders and related syndromes. J Allergy Clin Immunol. 2012;130:607-612.e9. doi:10.1016/j.jaci.2012.02.019
- Roufosse FE, Goldman M, Cogan E. Hypereosinophilic syndromes. Orphanet J Rare Dis. 2007;2:37. doi:10.1186/1750-1172-2-37
- Pitlick MM, Li JT, Pongdee T. Current and emerging biologic therapies targeting eosinophilic disorders. World Allergy Organ J. 2022;15:100676. doi:10.1016/j.waojou.2022.10067
- Ault P, Cortes J, Lynn A, Keating M, Verstovsek S. Pregnancy in a patient with hypereosinophilic syndrome. Leuk Res. 2009;33:186-187. doi:10.1016/j.leukres.2008.05.013
- Rioux JD, Stone VA, Daly MJ, et al. Familial eosinophilia maps to the cytokine gene cluster on human chromosomal region 5q31-q33. Am J Hum Genet. 1998;63:1086-1094. doi:10.1086/302053
- Williams KW, Ware J, Abiodun A, et al. Hypereosinophilia in children and adults: a retrospective comparison. J Allergy Clin Immunol Pract. 2016;4:941-947.e1. doi:10.1016/j.jaip.2016.03.020
- Pane F, Lefevre G, Kwon N, et al. Characterization of disease flares and impact of mepolizumab in patients with hypereosinophilic syndrome. Front Immunol. 2022;13:935996. doi:10.3389/fimmu.2022.935996
- Cogan E, Roufosse F. Clinical management of the hypereosinophilic syndromes. Expert Rev Hematol. 2012;5:275-290. doi: 10.1586/ehm.12.14
- Klion A. Hypereosinophilic syndrome: approach to treatment in the era of precision medicine. Hematology Am Soc Hematol Educ Program. 2018;2018:326-331. doi:10.1182/asheducation-2018.1.326
- Shomali W, Gotlib J. World health organization-defined eosinophilic disorders: 2022 update on diagnosis, risk stratification, and management. Am J Hematol. 2022;97:129-148. doi:10.1002/ajh.26352
- Helbig G, Klion AD. Hypereosinophilic syndromes - an enigmatic group of disorders with an intriguing clinical spectrum and challenging treatment. Blood Rev. 2021;49:100809. doi:10.1016/j.blre.2021.100809
- Valent P, Klion AD, Horny HP, et al. Contemporary consensus proposal on criteria and classification of eosinophilic disorders and related syndromes. J Allergy Clin Immunol. 2012;130:607-612.e9. doi:10.1016/j.jaci.2012.02.019
- Roufosse FE, Goldman M, Cogan E. Hypereosinophilic syndromes. Orphanet J Rare Dis. 2007;2:37. doi:10.1186/1750-1172-2-37
- Pitlick MM, Li JT, Pongdee T. Current and emerging biologic therapies targeting eosinophilic disorders. World Allergy Organ J. 2022;15:100676. doi:10.1016/j.waojou.2022.10067
- Ault P, Cortes J, Lynn A, Keating M, Verstovsek S. Pregnancy in a patient with hypereosinophilic syndrome. Leuk Res. 2009;33:186-187. doi:10.1016/j.leukres.2008.05.013
- Rioux JD, Stone VA, Daly MJ, et al. Familial eosinophilia maps to the cytokine gene cluster on human chromosomal region 5q31-q33. Am J Hum Genet. 1998;63:1086-1094. doi:10.1086/302053
- Williams KW, Ware J, Abiodun A, et al. Hypereosinophilia in children and adults: a retrospective comparison. J Allergy Clin Immunol Pract. 2016;4:941-947.e1. doi:10.1016/j.jaip.2016.03.020
- Pane F, Lefevre G, Kwon N, et al. Characterization of disease flares and impact of mepolizumab in patients with hypereosinophilic syndrome. Front Immunol. 2022;13:935996. doi:10.3389/fimmu.2022.935996
Unique Presentation of Postpartum Hypereosinophilic Syndrome With Atypical Features and Therapeutic Challenges
Unique Presentation of Postpartum Hypereosinophilic Syndrome With Atypical Features and Therapeutic Challenges
Evaluation of Subcutaneous Contraception for Patient Self-Administration at North Florida/South Georgia Veterans Health System
Evaluation of Subcutaneous Contraception for Patient Self-Administration at North Florida/South Georgia Veterans Health System
Medroxyprogesterone acetate is an injectable medication indicated for contraception and management of endometriosis-associated pain in females of reproductive age.1 Medroxyprogesterone inhibits gonadotropin secretion, which prevents follicular maturation and ovulation. This leads to endometrial thinning and a contraceptive effect. Adverse drug reactions (ADRs), such as weight gain, menstrual bleeding irregularities, and bone loss appear to be dose- and time-related. Two formulations of medroxyprogesterone acetate are available: 150 mg depot medroxyprogesterone acetate intramuscular (DMPA-IM) and 104 mg DMPA subcutaneous (DMPA-SC).2 Originally, medroxyprogesterone acetate injections required administration by a health care worker. While the current labeling for DMPA-SC still indicates a requirement for administration by a health care worker, data show that the medication can be safe and effective when self-administered.3
Self-Administered Contraception
The 2019 World Health Organization (WHO) guideline on self-care interventions recommends making self-administered injectable contraception available to individuals of reproductive age.3 The WHO recommendation is based on evidence from the Depo Self-Administration Study, which included 401 patients randomized 1:1 to receive self-administered or clinic-administered DMPA-SC. This study concluded that self-administration improved continuation of contraception.4
The North Florida/South Georgia Veterans Health System (NFSGVHS) is the largest US Department of Veterans Affairs (VA) health care system, serving > 22,000 female veterans. All primary care practitioners (PCP) have been trained in women’s health (WH).
The WH patient-aligned care team (PACT) clinical pharmacy practitioner (CPP) proposed using DMPA-SC for outpatient self-administration to increase access, improve patient satisfaction, and reduce burden on patients and nurses for administration appointments. The Pharmacy and Therapeutics Committee (P&T), WH Medical Director, and Chief of Gynecology approved the proposal. DMPA-SC was added to the ordering menu with order sets. The order set included instructions that outlined the 12-week dosing interval, instructions to contact the prescriber if the injection was > 2 weeks overdue (aligning with dosing recommendations for administration every 12 to 14 weeks), and an optional order for a home pregnancy test if necessary. These instructions were designed to ensure proper self-administration of the medication and timely follow-up care.
The gynecology and PACT health care practitioners (HCPs), including physicians, pharmacists, nurses, and medical assistants, received DMPA-SC education, which consisted of a review of medication, ADRs, contraindications, and administration. An NFSGVHS procedure was developed to ensure patients received self-administration education. DMPA-SC prescriptions were mailed to patients with scheduled nursing appointments. The patient would then bring DMPA-SC to the nursing appointment where they received administration instruction and completed the first injection under nurse supervision to ensure appropriate technique. Patients were offered supplementary educational documents and a calendar to keep track of injection days. The patients were responsible for ordering refills and administering subsequent injections at home. Once all stakeholders received education and order sets were in place, prescribers and nurses could begin offering the option for initiation of self-administered DMPA-SC to patients. All conversions or new prescriptions were initiated by prescribers as a part of usual care.
Medication Use Evaluation
A medication use evaluation was conducted about 1 year after the rollout to assess use, adherence, and impact of DMPA-SC for patient-self administration as a new contraceptive option for NFSGVHS patients.
A retrospective chart review was conducted for patients dispensed DMPA-SC from June 1, 2022, to July 1, 2023. Baseline body mass index (BMI), recorded prior to initiation of DMPA-SC, was compared with the most recent BMI on record at the completion of the study to evaluate weight change. Nursing visit attendance for the first injection was also assessed. Adherence was evaluated by reviewing the date of the initial DMPA-SC prescription, the date of the patient's first nursing visit, and subsequent refill patterns. A 2-week margin of error was established to account for the flexibility within the recommended dosing interval and delays in postal service delivery.
Forty patients were initiated on DMPA-SC for patient self-administration. The mean age of patients was 37.2 years. All 40 patients were female. Twenty-two patients (55%) identified as Black, 17 (43%) as White, and 1 (3%) as Asian. The majority (90%) of patients were non-Hispanic. The mean baseline BMI was 30 and BMI after DMPA-SC initiation was 30.4.
Twenty-eight (70%) patients had a nursing appointment, adhering to the NFSGVHS protocol. Five patients (13%) discontinued use and switched to DMPA-IM administered by an HCP and 4 (10%) discontinued use following an ADR (hives, mood changes, bruising, and menometrorrhagia). Of the 31 patients who continued therapy, 25 (81%) were refilling appropriately (Table).

Six patients with unidentified reasons for nonadherence were contacted to determine if there were unmet contraceptive needs. This subgroup included patients with an active prescription for DMPA-SC that did not meet refill expectations. Nonadherence was mostly due to forgetfulness, however 1 patient was unable to refill her DMPA-SC in a timely manner due to an outside hospital admission and another was unreachable. These conversations were documented in the electronic health record (EHR) and all patients requesting follow-up, reinitiation of therapy, or alternative regimens, the appropriate parties were notified to coordinate care.
Discussion
The uptake in DMPA-SC prescribing suggests prescribers and patients have embraced self-administration as an option for contraception. Most patients were appropriately scheduled for nursing appointments to reinforce education and ensure appropriate self-injection technique, as outlined in the NFSGVHS procedure.
The need to improve adherence to NFSGVHS procedure was identified because not all patients had scheduled nursing appointments. This is concerning because some patients may have started self-injecting DMPA-SC without proper education, which could lead to improper injection technique and diminished effectiveness. Nursing appointments ensure appropriate self-injection techniques and reinforce the importance of refilling every 12 weeks for proper effectiveness. Nonadherence to contraceptive therapy may result in unintended pregnancy, although no pregnancies were reported by patients in this study. Pharmacist involvement in DMPA-SC initiation and follow-up monitoring may help ensure adherence to local procedure for initiation and improve patient adherence.
There is limited evidence comparing weight gain related to DMPA-SC vs DMPA-IM. However, in a small, 2-year, randomized study, weight changes were considered comparable for both cohorts with a mean increase of 3.5 kg in the DMPA-IM group vs 3.4 kg in the DMPA-SC group.5 While our analysis did not formally evaluate weight changes, BMI data were collected to evaluate for evidence of weight change. The duration of therapy varied per patient and may not have been long enough to see comparable weight changes.
Strengths of this project include the use of the PACT multidisciplinary approach in primary care including physicians, pharmacists, and nurses. The NFSGVHS EHR is comprehensive, and data including appointments and pharmacy refill information was readily available for collection and evaluation. Limitations included inconsistent documentation in the patient’s EHR which made collection of some data difficult.
Cost Estimates
NFSGVHS had 231 patients prescribed DMPA-IM at the time of DMPA-SC rollout and 40 patients initiated DMPA-SC therapy in the first year. There are possible cost savings associated with the use of DMPA-SC compared to DMPA-IM. Although DMPA-IM costs about $120 annually and DMPA-SC costs about $252 annually, this does not account for indirect costs such as supplies, overhead cost, nursing visits, and patient travel.6 Additionally, allowing patients to self-administer the DMPA-SC injection at home provides nurses time to care for other patients.
Moving forward, the PACT and gynecology teams will receive instruction on the importance of adhering to NFSGVHS procedures to ensure new patients prescribed DMPA-SC receive education and present for nursing appointments to ensure appropriate self-injection.
DMPA has historically been administered in the clinic setting by an HCP; therefore, the prescriber was available to assess adherence to therapy based on patient’s attendance to scheduled clinic appointments. Some prescribers may feel apprehensive about shifting the onus of medication adherence to the patient when prescribing DMPA-SC. However, this model is comparable to any other prescription form of birth control, such as combined hormonal contraceptive pills, where the prescriber expects the patient to take the medication as prescribed and refill their prescriptions in a timely manner to avoid gaps in therapy. The findings of this project suggest the majority of patients who were prescribed self-administered DMPA-SC for contraception were adherent to therapy. The utility of self-administration of DMPA-SC for other labeled or off-label indications was not evaluated; however, it is possible that patients who are motivated to self-administer the medication (regardless of indication) would also demonstrate similar adherence rates.
Conclusions
The majority of patients who started DMPA-SC tolerated the medication well and continued to refill therapy within the recommended time period. Patient self-administration of DMPA-SC can enhance access by removing barriers to administration, increase patient autonomy and contraceptive continuation rates. Overall, the increase in DMPA-SC prescriptions suggests that patients and HCPs support the option for DMPA-SC self-administration at NFSGVHS.
- Depo-SubQ Provera. Package insert. Pharmacia & Upjohn Co; 2019.
- Kaunitz AM. Depot medroxyprogesterone acetate. UpToDate. Updated June 12, 2025. Accessed July 11, 2025. https://www.uptodate.com/contents/depot-medroxyprogesterone-acetate-dmpa-formulations-patient-selection-and-drug-administration
- World Health Organization. WHO guideline on self-care interventions for health and well-being, 2022 revision. World Health Organization. 2022. Accessed July 17, 2025. https://iris.who.int/bitstream/handle/10665/357828/9789240052192-eng.pdf
- Kohn JE, Simons HR, Della Badia L, et al. Increased 1-year continuation of DMPA among women randomized to self-administration: results from a randomized controlled trial at Planned Parenthood. Contraception. 2018;97(3):198-204. doi:10.1016/j.contraception.2017.11.009
- Kaunitz AM, Darney PD, Ross D, Wolter KD, Speroff L. Subcutaneous DMPA vs. intramuscular DMPA: a 2-year randomized study of contraceptive efficacy and bone mineral density. Contraception. 2009;80(1):7-17. doi:10.1016/j.contraception.2009.02.005
- UpToDate, Lexidrug. Medroxyprogesterone acetate. Accessed July 16, 2025. https://online.lexi.com
Medroxyprogesterone acetate is an injectable medication indicated for contraception and management of endometriosis-associated pain in females of reproductive age.1 Medroxyprogesterone inhibits gonadotropin secretion, which prevents follicular maturation and ovulation. This leads to endometrial thinning and a contraceptive effect. Adverse drug reactions (ADRs), such as weight gain, menstrual bleeding irregularities, and bone loss appear to be dose- and time-related. Two formulations of medroxyprogesterone acetate are available: 150 mg depot medroxyprogesterone acetate intramuscular (DMPA-IM) and 104 mg DMPA subcutaneous (DMPA-SC).2 Originally, medroxyprogesterone acetate injections required administration by a health care worker. While the current labeling for DMPA-SC still indicates a requirement for administration by a health care worker, data show that the medication can be safe and effective when self-administered.3
Self-Administered Contraception
The 2019 World Health Organization (WHO) guideline on self-care interventions recommends making self-administered injectable contraception available to individuals of reproductive age.3 The WHO recommendation is based on evidence from the Depo Self-Administration Study, which included 401 patients randomized 1:1 to receive self-administered or clinic-administered DMPA-SC. This study concluded that self-administration improved continuation of contraception.4
The North Florida/South Georgia Veterans Health System (NFSGVHS) is the largest US Department of Veterans Affairs (VA) health care system, serving > 22,000 female veterans. All primary care practitioners (PCP) have been trained in women’s health (WH).
The WH patient-aligned care team (PACT) clinical pharmacy practitioner (CPP) proposed using DMPA-SC for outpatient self-administration to increase access, improve patient satisfaction, and reduce burden on patients and nurses for administration appointments. The Pharmacy and Therapeutics Committee (P&T), WH Medical Director, and Chief of Gynecology approved the proposal. DMPA-SC was added to the ordering menu with order sets. The order set included instructions that outlined the 12-week dosing interval, instructions to contact the prescriber if the injection was > 2 weeks overdue (aligning with dosing recommendations for administration every 12 to 14 weeks), and an optional order for a home pregnancy test if necessary. These instructions were designed to ensure proper self-administration of the medication and timely follow-up care.
The gynecology and PACT health care practitioners (HCPs), including physicians, pharmacists, nurses, and medical assistants, received DMPA-SC education, which consisted of a review of medication, ADRs, contraindications, and administration. An NFSGVHS procedure was developed to ensure patients received self-administration education. DMPA-SC prescriptions were mailed to patients with scheduled nursing appointments. The patient would then bring DMPA-SC to the nursing appointment where they received administration instruction and completed the first injection under nurse supervision to ensure appropriate technique. Patients were offered supplementary educational documents and a calendar to keep track of injection days. The patients were responsible for ordering refills and administering subsequent injections at home. Once all stakeholders received education and order sets were in place, prescribers and nurses could begin offering the option for initiation of self-administered DMPA-SC to patients. All conversions or new prescriptions were initiated by prescribers as a part of usual care.
Medication Use Evaluation
A medication use evaluation was conducted about 1 year after the rollout to assess use, adherence, and impact of DMPA-SC for patient-self administration as a new contraceptive option for NFSGVHS patients.
A retrospective chart review was conducted for patients dispensed DMPA-SC from June 1, 2022, to July 1, 2023. Baseline body mass index (BMI), recorded prior to initiation of DMPA-SC, was compared with the most recent BMI on record at the completion of the study to evaluate weight change. Nursing visit attendance for the first injection was also assessed. Adherence was evaluated by reviewing the date of the initial DMPA-SC prescription, the date of the patient's first nursing visit, and subsequent refill patterns. A 2-week margin of error was established to account for the flexibility within the recommended dosing interval and delays in postal service delivery.
Forty patients were initiated on DMPA-SC for patient self-administration. The mean age of patients was 37.2 years. All 40 patients were female. Twenty-two patients (55%) identified as Black, 17 (43%) as White, and 1 (3%) as Asian. The majority (90%) of patients were non-Hispanic. The mean baseline BMI was 30 and BMI after DMPA-SC initiation was 30.4.
Twenty-eight (70%) patients had a nursing appointment, adhering to the NFSGVHS protocol. Five patients (13%) discontinued use and switched to DMPA-IM administered by an HCP and 4 (10%) discontinued use following an ADR (hives, mood changes, bruising, and menometrorrhagia). Of the 31 patients who continued therapy, 25 (81%) were refilling appropriately (Table).

Six patients with unidentified reasons for nonadherence were contacted to determine if there were unmet contraceptive needs. This subgroup included patients with an active prescription for DMPA-SC that did not meet refill expectations. Nonadherence was mostly due to forgetfulness, however 1 patient was unable to refill her DMPA-SC in a timely manner due to an outside hospital admission and another was unreachable. These conversations were documented in the electronic health record (EHR) and all patients requesting follow-up, reinitiation of therapy, or alternative regimens, the appropriate parties were notified to coordinate care.
Discussion
The uptake in DMPA-SC prescribing suggests prescribers and patients have embraced self-administration as an option for contraception. Most patients were appropriately scheduled for nursing appointments to reinforce education and ensure appropriate self-injection technique, as outlined in the NFSGVHS procedure.
The need to improve adherence to NFSGVHS procedure was identified because not all patients had scheduled nursing appointments. This is concerning because some patients may have started self-injecting DMPA-SC without proper education, which could lead to improper injection technique and diminished effectiveness. Nursing appointments ensure appropriate self-injection techniques and reinforce the importance of refilling every 12 weeks for proper effectiveness. Nonadherence to contraceptive therapy may result in unintended pregnancy, although no pregnancies were reported by patients in this study. Pharmacist involvement in DMPA-SC initiation and follow-up monitoring may help ensure adherence to local procedure for initiation and improve patient adherence.
There is limited evidence comparing weight gain related to DMPA-SC vs DMPA-IM. However, in a small, 2-year, randomized study, weight changes were considered comparable for both cohorts with a mean increase of 3.5 kg in the DMPA-IM group vs 3.4 kg in the DMPA-SC group.5 While our analysis did not formally evaluate weight changes, BMI data were collected to evaluate for evidence of weight change. The duration of therapy varied per patient and may not have been long enough to see comparable weight changes.
Strengths of this project include the use of the PACT multidisciplinary approach in primary care including physicians, pharmacists, and nurses. The NFSGVHS EHR is comprehensive, and data including appointments and pharmacy refill information was readily available for collection and evaluation. Limitations included inconsistent documentation in the patient’s EHR which made collection of some data difficult.
Cost Estimates
NFSGVHS had 231 patients prescribed DMPA-IM at the time of DMPA-SC rollout and 40 patients initiated DMPA-SC therapy in the first year. There are possible cost savings associated with the use of DMPA-SC compared to DMPA-IM. Although DMPA-IM costs about $120 annually and DMPA-SC costs about $252 annually, this does not account for indirect costs such as supplies, overhead cost, nursing visits, and patient travel.6 Additionally, allowing patients to self-administer the DMPA-SC injection at home provides nurses time to care for other patients.
Moving forward, the PACT and gynecology teams will receive instruction on the importance of adhering to NFSGVHS procedures to ensure new patients prescribed DMPA-SC receive education and present for nursing appointments to ensure appropriate self-injection.
DMPA has historically been administered in the clinic setting by an HCP; therefore, the prescriber was available to assess adherence to therapy based on patient’s attendance to scheduled clinic appointments. Some prescribers may feel apprehensive about shifting the onus of medication adherence to the patient when prescribing DMPA-SC. However, this model is comparable to any other prescription form of birth control, such as combined hormonal contraceptive pills, where the prescriber expects the patient to take the medication as prescribed and refill their prescriptions in a timely manner to avoid gaps in therapy. The findings of this project suggest the majority of patients who were prescribed self-administered DMPA-SC for contraception were adherent to therapy. The utility of self-administration of DMPA-SC for other labeled or off-label indications was not evaluated; however, it is possible that patients who are motivated to self-administer the medication (regardless of indication) would also demonstrate similar adherence rates.
Conclusions
The majority of patients who started DMPA-SC tolerated the medication well and continued to refill therapy within the recommended time period. Patient self-administration of DMPA-SC can enhance access by removing barriers to administration, increase patient autonomy and contraceptive continuation rates. Overall, the increase in DMPA-SC prescriptions suggests that patients and HCPs support the option for DMPA-SC self-administration at NFSGVHS.
Medroxyprogesterone acetate is an injectable medication indicated for contraception and management of endometriosis-associated pain in females of reproductive age.1 Medroxyprogesterone inhibits gonadotropin secretion, which prevents follicular maturation and ovulation. This leads to endometrial thinning and a contraceptive effect. Adverse drug reactions (ADRs), such as weight gain, menstrual bleeding irregularities, and bone loss appear to be dose- and time-related. Two formulations of medroxyprogesterone acetate are available: 150 mg depot medroxyprogesterone acetate intramuscular (DMPA-IM) and 104 mg DMPA subcutaneous (DMPA-SC).2 Originally, medroxyprogesterone acetate injections required administration by a health care worker. While the current labeling for DMPA-SC still indicates a requirement for administration by a health care worker, data show that the medication can be safe and effective when self-administered.3
Self-Administered Contraception
The 2019 World Health Organization (WHO) guideline on self-care interventions recommends making self-administered injectable contraception available to individuals of reproductive age.3 The WHO recommendation is based on evidence from the Depo Self-Administration Study, which included 401 patients randomized 1:1 to receive self-administered or clinic-administered DMPA-SC. This study concluded that self-administration improved continuation of contraception.4
The North Florida/South Georgia Veterans Health System (NFSGVHS) is the largest US Department of Veterans Affairs (VA) health care system, serving > 22,000 female veterans. All primary care practitioners (PCP) have been trained in women’s health (WH).
The WH patient-aligned care team (PACT) clinical pharmacy practitioner (CPP) proposed using DMPA-SC for outpatient self-administration to increase access, improve patient satisfaction, and reduce burden on patients and nurses for administration appointments. The Pharmacy and Therapeutics Committee (P&T), WH Medical Director, and Chief of Gynecology approved the proposal. DMPA-SC was added to the ordering menu with order sets. The order set included instructions that outlined the 12-week dosing interval, instructions to contact the prescriber if the injection was > 2 weeks overdue (aligning with dosing recommendations for administration every 12 to 14 weeks), and an optional order for a home pregnancy test if necessary. These instructions were designed to ensure proper self-administration of the medication and timely follow-up care.
The gynecology and PACT health care practitioners (HCPs), including physicians, pharmacists, nurses, and medical assistants, received DMPA-SC education, which consisted of a review of medication, ADRs, contraindications, and administration. An NFSGVHS procedure was developed to ensure patients received self-administration education. DMPA-SC prescriptions were mailed to patients with scheduled nursing appointments. The patient would then bring DMPA-SC to the nursing appointment where they received administration instruction and completed the first injection under nurse supervision to ensure appropriate technique. Patients were offered supplementary educational documents and a calendar to keep track of injection days. The patients were responsible for ordering refills and administering subsequent injections at home. Once all stakeholders received education and order sets were in place, prescribers and nurses could begin offering the option for initiation of self-administered DMPA-SC to patients. All conversions or new prescriptions were initiated by prescribers as a part of usual care.
Medication Use Evaluation
A medication use evaluation was conducted about 1 year after the rollout to assess use, adherence, and impact of DMPA-SC for patient-self administration as a new contraceptive option for NFSGVHS patients.
A retrospective chart review was conducted for patients dispensed DMPA-SC from June 1, 2022, to July 1, 2023. Baseline body mass index (BMI), recorded prior to initiation of DMPA-SC, was compared with the most recent BMI on record at the completion of the study to evaluate weight change. Nursing visit attendance for the first injection was also assessed. Adherence was evaluated by reviewing the date of the initial DMPA-SC prescription, the date of the patient's first nursing visit, and subsequent refill patterns. A 2-week margin of error was established to account for the flexibility within the recommended dosing interval and delays in postal service delivery.
Forty patients were initiated on DMPA-SC for patient self-administration. The mean age of patients was 37.2 years. All 40 patients were female. Twenty-two patients (55%) identified as Black, 17 (43%) as White, and 1 (3%) as Asian. The majority (90%) of patients were non-Hispanic. The mean baseline BMI was 30 and BMI after DMPA-SC initiation was 30.4.
Twenty-eight (70%) patients had a nursing appointment, adhering to the NFSGVHS protocol. Five patients (13%) discontinued use and switched to DMPA-IM administered by an HCP and 4 (10%) discontinued use following an ADR (hives, mood changes, bruising, and menometrorrhagia). Of the 31 patients who continued therapy, 25 (81%) were refilling appropriately (Table).

Six patients with unidentified reasons for nonadherence were contacted to determine if there were unmet contraceptive needs. This subgroup included patients with an active prescription for DMPA-SC that did not meet refill expectations. Nonadherence was mostly due to forgetfulness, however 1 patient was unable to refill her DMPA-SC in a timely manner due to an outside hospital admission and another was unreachable. These conversations were documented in the electronic health record (EHR) and all patients requesting follow-up, reinitiation of therapy, or alternative regimens, the appropriate parties were notified to coordinate care.
Discussion
The uptake in DMPA-SC prescribing suggests prescribers and patients have embraced self-administration as an option for contraception. Most patients were appropriately scheduled for nursing appointments to reinforce education and ensure appropriate self-injection technique, as outlined in the NFSGVHS procedure.
The need to improve adherence to NFSGVHS procedure was identified because not all patients had scheduled nursing appointments. This is concerning because some patients may have started self-injecting DMPA-SC without proper education, which could lead to improper injection technique and diminished effectiveness. Nursing appointments ensure appropriate self-injection techniques and reinforce the importance of refilling every 12 weeks for proper effectiveness. Nonadherence to contraceptive therapy may result in unintended pregnancy, although no pregnancies were reported by patients in this study. Pharmacist involvement in DMPA-SC initiation and follow-up monitoring may help ensure adherence to local procedure for initiation and improve patient adherence.
There is limited evidence comparing weight gain related to DMPA-SC vs DMPA-IM. However, in a small, 2-year, randomized study, weight changes were considered comparable for both cohorts with a mean increase of 3.5 kg in the DMPA-IM group vs 3.4 kg in the DMPA-SC group.5 While our analysis did not formally evaluate weight changes, BMI data were collected to evaluate for evidence of weight change. The duration of therapy varied per patient and may not have been long enough to see comparable weight changes.
Strengths of this project include the use of the PACT multidisciplinary approach in primary care including physicians, pharmacists, and nurses. The NFSGVHS EHR is comprehensive, and data including appointments and pharmacy refill information was readily available for collection and evaluation. Limitations included inconsistent documentation in the patient’s EHR which made collection of some data difficult.
Cost Estimates
NFSGVHS had 231 patients prescribed DMPA-IM at the time of DMPA-SC rollout and 40 patients initiated DMPA-SC therapy in the first year. There are possible cost savings associated with the use of DMPA-SC compared to DMPA-IM. Although DMPA-IM costs about $120 annually and DMPA-SC costs about $252 annually, this does not account for indirect costs such as supplies, overhead cost, nursing visits, and patient travel.6 Additionally, allowing patients to self-administer the DMPA-SC injection at home provides nurses time to care for other patients.
Moving forward, the PACT and gynecology teams will receive instruction on the importance of adhering to NFSGVHS procedures to ensure new patients prescribed DMPA-SC receive education and present for nursing appointments to ensure appropriate self-injection.
DMPA has historically been administered in the clinic setting by an HCP; therefore, the prescriber was available to assess adherence to therapy based on patient’s attendance to scheduled clinic appointments. Some prescribers may feel apprehensive about shifting the onus of medication adherence to the patient when prescribing DMPA-SC. However, this model is comparable to any other prescription form of birth control, such as combined hormonal contraceptive pills, where the prescriber expects the patient to take the medication as prescribed and refill their prescriptions in a timely manner to avoid gaps in therapy. The findings of this project suggest the majority of patients who were prescribed self-administered DMPA-SC for contraception were adherent to therapy. The utility of self-administration of DMPA-SC for other labeled or off-label indications was not evaluated; however, it is possible that patients who are motivated to self-administer the medication (regardless of indication) would also demonstrate similar adherence rates.
Conclusions
The majority of patients who started DMPA-SC tolerated the medication well and continued to refill therapy within the recommended time period. Patient self-administration of DMPA-SC can enhance access by removing barriers to administration, increase patient autonomy and contraceptive continuation rates. Overall, the increase in DMPA-SC prescriptions suggests that patients and HCPs support the option for DMPA-SC self-administration at NFSGVHS.
- Depo-SubQ Provera. Package insert. Pharmacia & Upjohn Co; 2019.
- Kaunitz AM. Depot medroxyprogesterone acetate. UpToDate. Updated June 12, 2025. Accessed July 11, 2025. https://www.uptodate.com/contents/depot-medroxyprogesterone-acetate-dmpa-formulations-patient-selection-and-drug-administration
- World Health Organization. WHO guideline on self-care interventions for health and well-being, 2022 revision. World Health Organization. 2022. Accessed July 17, 2025. https://iris.who.int/bitstream/handle/10665/357828/9789240052192-eng.pdf
- Kohn JE, Simons HR, Della Badia L, et al. Increased 1-year continuation of DMPA among women randomized to self-administration: results from a randomized controlled trial at Planned Parenthood. Contraception. 2018;97(3):198-204. doi:10.1016/j.contraception.2017.11.009
- Kaunitz AM, Darney PD, Ross D, Wolter KD, Speroff L. Subcutaneous DMPA vs. intramuscular DMPA: a 2-year randomized study of contraceptive efficacy and bone mineral density. Contraception. 2009;80(1):7-17. doi:10.1016/j.contraception.2009.02.005
- UpToDate, Lexidrug. Medroxyprogesterone acetate. Accessed July 16, 2025. https://online.lexi.com
- Depo-SubQ Provera. Package insert. Pharmacia & Upjohn Co; 2019.
- Kaunitz AM. Depot medroxyprogesterone acetate. UpToDate. Updated June 12, 2025. Accessed July 11, 2025. https://www.uptodate.com/contents/depot-medroxyprogesterone-acetate-dmpa-formulations-patient-selection-and-drug-administration
- World Health Organization. WHO guideline on self-care interventions for health and well-being, 2022 revision. World Health Organization. 2022. Accessed July 17, 2025. https://iris.who.int/bitstream/handle/10665/357828/9789240052192-eng.pdf
- Kohn JE, Simons HR, Della Badia L, et al. Increased 1-year continuation of DMPA among women randomized to self-administration: results from a randomized controlled trial at Planned Parenthood. Contraception. 2018;97(3):198-204. doi:10.1016/j.contraception.2017.11.009
- Kaunitz AM, Darney PD, Ross D, Wolter KD, Speroff L. Subcutaneous DMPA vs. intramuscular DMPA: a 2-year randomized study of contraceptive efficacy and bone mineral density. Contraception. 2009;80(1):7-17. doi:10.1016/j.contraception.2009.02.005
- UpToDate, Lexidrug. Medroxyprogesterone acetate. Accessed July 16, 2025. https://online.lexi.com
Evaluation of Subcutaneous Contraception for Patient Self-Administration at North Florida/South Georgia Veterans Health System
Evaluation of Subcutaneous Contraception for Patient Self-Administration at North Florida/South Georgia Veterans Health System
Data Trends 2025: Women's Health
Data Trends 2025: Women's Health
Click here to view more from Federal Health Care Data Trends 2025.
- Women Veterans Health Care: Facts and Statistics. US Department of Veterans Affairs. Published 2022. Accessed May 23, 2025. https://www.womenshealth.va.gov/materials-and-resources/facts-and-statistics.asp
- Sourcebook: Women Veterans in the Veterans Health Administration. Volume 5: Longitudinal Trends in Sociodemographics and Utilization, Including Type, Modality, and Source of Care. US Department of Veterans Affairs; 2024. Accessed May 23, 2025. https://www.womenshealth.va.gov/WOMENSHEALTH/docs/VHA-Source-book-V5-FINAL.pdf
- Goldstein KM, et al. JAMA Netw Open. 2025;8(4):e256372. doi:10.1001/ jamanetworkopen.2025.6372
- Sheahan KL, et al. J Gen Intern Med. 2022;37(Suppl 3):791-798. doi:10.1007/s11606-022-07585-3
- Adams RE, et al. BMC Womens Health. 2021;21(1):1-10. doi:10.1186/s12905-021-01183-z
- Haskell SG, et al. J Womens Health (Larchmt). 2010;19(2):267-271. doi:10.1089/jwh.2008.1262
- VHA Directive 1330.01(1): Health Care Services for Women Veterans. US Department of Veterans Affairs; February 15, 2023. Accessed May 23, 2025. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=10576
- VHA Directive 1115(1): Military Sexual Trauma (MST) Program. US Department of Veterans Affairs; May 8, 2018. Accessed May 23, 2025. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=6432
- Marshall V, et al. Womens Health Issues. 2021;31(2):150-157. doi:10.1016/j.whi.2020.10.005
- Washington DL, et al. J Gen Intern Med. 2011;26(suppl 2):655-661. doi:10.1007/s11606-011-1772-z
- Hadlandsmyth K, et al. Eur J Pain. 2024;28(8):1311-1319. doi:10.1002/ejp.2258
- Military Sexual Trauma Fact Sheet–VA Mental Health. US Department of Veterans Affairs. May 1, 2021. Accessed March 21, 2025. https://www.mentalhealth.va.gov/docs/mst_general_factsheet.pdf
- National Center for Veterans Analysis and Statistics. Population Tables: the nation, age/sex. US Department of Veterans Affairs website. Accessed March 21, 2025. https://www.va.gov/vetdata/Veteran_Population.asp
- Serving Her Country: Exploring the Characteristics of Women Veterans. US Department of Veterans Affairs. Accessed March 21, 2025. https://www.data.va.gov/stories/s/Women-Veterans-in-2023/wci3-yrsv/
- Gasperi M, et al. JAMA Netw Open. 2024;7(3):e242299. doi:10.1001/jamanetworkopen.2024.2299
- US Department of Veterans Affairs. Study of Barriers for Women Veterans to VA Health Care: Final Report. February 2024. Accessed March 21, 2025. https://www.womenshealth.va.gov/materials-and-resources/publications-and-reports.asp
- Iverson KM, et al. J Gen Intern Med. 2019;34(11):2435-2442. doi:10.1007/s11606-019-05240-y
- Spinelli S, et al. J Gen Intern Med. 2022;37(suppl 3):837-841. doi:10.1007/s11606-022-07577-3
- Carlson K, et al. In: StatPearls. StatPearls Publishing; 2025. Updated January 22,2025. Accessed March 21, 2025. https://www.ncbi.nlm.nih.gov/books/NBK519070/
Monteith LL, et al. J Interpers Violence. 2023;38(11-12):7578-7601. doi:10.1177/08862605221145725
Click here to view more from Federal Health Care Data Trends 2025.
Click here to view more from Federal Health Care Data Trends 2025.
- Women Veterans Health Care: Facts and Statistics. US Department of Veterans Affairs. Published 2022. Accessed May 23, 2025. https://www.womenshealth.va.gov/materials-and-resources/facts-and-statistics.asp
- Sourcebook: Women Veterans in the Veterans Health Administration. Volume 5: Longitudinal Trends in Sociodemographics and Utilization, Including Type, Modality, and Source of Care. US Department of Veterans Affairs; 2024. Accessed May 23, 2025. https://www.womenshealth.va.gov/WOMENSHEALTH/docs/VHA-Source-book-V5-FINAL.pdf
- Goldstein KM, et al. JAMA Netw Open. 2025;8(4):e256372. doi:10.1001/ jamanetworkopen.2025.6372
- Sheahan KL, et al. J Gen Intern Med. 2022;37(Suppl 3):791-798. doi:10.1007/s11606-022-07585-3
- Adams RE, et al. BMC Womens Health. 2021;21(1):1-10. doi:10.1186/s12905-021-01183-z
- Haskell SG, et al. J Womens Health (Larchmt). 2010;19(2):267-271. doi:10.1089/jwh.2008.1262
- VHA Directive 1330.01(1): Health Care Services for Women Veterans. US Department of Veterans Affairs; February 15, 2023. Accessed May 23, 2025. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=10576
- VHA Directive 1115(1): Military Sexual Trauma (MST) Program. US Department of Veterans Affairs; May 8, 2018. Accessed May 23, 2025. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=6432
- Marshall V, et al. Womens Health Issues. 2021;31(2):150-157. doi:10.1016/j.whi.2020.10.005
- Washington DL, et al. J Gen Intern Med. 2011;26(suppl 2):655-661. doi:10.1007/s11606-011-1772-z
- Hadlandsmyth K, et al. Eur J Pain. 2024;28(8):1311-1319. doi:10.1002/ejp.2258
- Military Sexual Trauma Fact Sheet–VA Mental Health. US Department of Veterans Affairs. May 1, 2021. Accessed March 21, 2025. https://www.mentalhealth.va.gov/docs/mst_general_factsheet.pdf
- National Center for Veterans Analysis and Statistics. Population Tables: the nation, age/sex. US Department of Veterans Affairs website. Accessed March 21, 2025. https://www.va.gov/vetdata/Veteran_Population.asp
- Serving Her Country: Exploring the Characteristics of Women Veterans. US Department of Veterans Affairs. Accessed March 21, 2025. https://www.data.va.gov/stories/s/Women-Veterans-in-2023/wci3-yrsv/
- Gasperi M, et al. JAMA Netw Open. 2024;7(3):e242299. doi:10.1001/jamanetworkopen.2024.2299
- US Department of Veterans Affairs. Study of Barriers for Women Veterans to VA Health Care: Final Report. February 2024. Accessed March 21, 2025. https://www.womenshealth.va.gov/materials-and-resources/publications-and-reports.asp
- Iverson KM, et al. J Gen Intern Med. 2019;34(11):2435-2442. doi:10.1007/s11606-019-05240-y
- Spinelli S, et al. J Gen Intern Med. 2022;37(suppl 3):837-841. doi:10.1007/s11606-022-07577-3
- Carlson K, et al. In: StatPearls. StatPearls Publishing; 2025. Updated January 22,2025. Accessed March 21, 2025. https://www.ncbi.nlm.nih.gov/books/NBK519070/
Monteith LL, et al. J Interpers Violence. 2023;38(11-12):7578-7601. doi:10.1177/08862605221145725
- Women Veterans Health Care: Facts and Statistics. US Department of Veterans Affairs. Published 2022. Accessed May 23, 2025. https://www.womenshealth.va.gov/materials-and-resources/facts-and-statistics.asp
- Sourcebook: Women Veterans in the Veterans Health Administration. Volume 5: Longitudinal Trends in Sociodemographics and Utilization, Including Type, Modality, and Source of Care. US Department of Veterans Affairs; 2024. Accessed May 23, 2025. https://www.womenshealth.va.gov/WOMENSHEALTH/docs/VHA-Source-book-V5-FINAL.pdf
- Goldstein KM, et al. JAMA Netw Open. 2025;8(4):e256372. doi:10.1001/ jamanetworkopen.2025.6372
- Sheahan KL, et al. J Gen Intern Med. 2022;37(Suppl 3):791-798. doi:10.1007/s11606-022-07585-3
- Adams RE, et al. BMC Womens Health. 2021;21(1):1-10. doi:10.1186/s12905-021-01183-z
- Haskell SG, et al. J Womens Health (Larchmt). 2010;19(2):267-271. doi:10.1089/jwh.2008.1262
- VHA Directive 1330.01(1): Health Care Services for Women Veterans. US Department of Veterans Affairs; February 15, 2023. Accessed May 23, 2025. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=10576
- VHA Directive 1115(1): Military Sexual Trauma (MST) Program. US Department of Veterans Affairs; May 8, 2018. Accessed May 23, 2025. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=6432
- Marshall V, et al. Womens Health Issues. 2021;31(2):150-157. doi:10.1016/j.whi.2020.10.005
- Washington DL, et al. J Gen Intern Med. 2011;26(suppl 2):655-661. doi:10.1007/s11606-011-1772-z
- Hadlandsmyth K, et al. Eur J Pain. 2024;28(8):1311-1319. doi:10.1002/ejp.2258
- Military Sexual Trauma Fact Sheet–VA Mental Health. US Department of Veterans Affairs. May 1, 2021. Accessed March 21, 2025. https://www.mentalhealth.va.gov/docs/mst_general_factsheet.pdf
- National Center for Veterans Analysis and Statistics. Population Tables: the nation, age/sex. US Department of Veterans Affairs website. Accessed March 21, 2025. https://www.va.gov/vetdata/Veteran_Population.asp
- Serving Her Country: Exploring the Characteristics of Women Veterans. US Department of Veterans Affairs. Accessed March 21, 2025. https://www.data.va.gov/stories/s/Women-Veterans-in-2023/wci3-yrsv/
- Gasperi M, et al. JAMA Netw Open. 2024;7(3):e242299. doi:10.1001/jamanetworkopen.2024.2299
- US Department of Veterans Affairs. Study of Barriers for Women Veterans to VA Health Care: Final Report. February 2024. Accessed March 21, 2025. https://www.womenshealth.va.gov/materials-and-resources/publications-and-reports.asp
- Iverson KM, et al. J Gen Intern Med. 2019;34(11):2435-2442. doi:10.1007/s11606-019-05240-y
- Spinelli S, et al. J Gen Intern Med. 2022;37(suppl 3):837-841. doi:10.1007/s11606-022-07577-3
- Carlson K, et al. In: StatPearls. StatPearls Publishing; 2025. Updated January 22,2025. Accessed March 21, 2025. https://www.ncbi.nlm.nih.gov/books/NBK519070/
Monteith LL, et al. J Interpers Violence. 2023;38(11-12):7578-7601. doi:10.1177/08862605221145725
Data Trends 2025: Women's Health
Data Trends 2025: Women's Health
Veterans and Nonveterans Show Similar Mammogram Rates
TOPLINE: A national survey of 8996 females reveals comparable mammography screening rates between those who identify as veterans (57.9%) and nonveterans (55.2%).
METHODOLOGY:
Researchers analyzed data from the 2019 National Health Interview Survey, a cross-sectional national survey tracking health information.
Female respondents aged 40 to 74 years without history of breast cancer were included in the analysis.
Analysis evaluated the association between screening and veteran status through logistic regression, adjusting for potential confounders.
Survey procedures accounted for complex sampling design to obtain valid estimates for the civilian, noninstitutionalized US population.
TAKEAWAY:
Analysis included 8996 female survey respondents, including 169 veterans (1.9%) and 320 (3.2%) reported having military health coverage.
Mammography screening rates within the last year were comparable between veterans (57.9%) and nonveterans (55.2%).
Veteran status showed no significant association with differences in mammography screening percentages (P = .96).
Among insured participants, military health insurance demonstrated no significant association with mammography screening percentages (P = .13).
The authors suggest that radiology practices should design proactive outreach strategies to address the needs of the growing number of female veterans who may face increased breast cancer risk due to military environmental exposures.
IN PRACTICE: “Although the results from our study demonstrate comparable mammography screening percentages, veterans may face additional risk factors for breast cancer due to occupational,” the authors argue.
SOURCE: This summary is based on a preprint published online in the Journal of the American College of Radiology: Milton A, Miles R, Gettle LM, Van Geertruyden P, Narayan AK. Utilization of Mammography Screening in Female Veterans: Cross-Sectional Survey Results from the National Health Interview Survey. J Am Coll Radiol. Published online April 24, 2025. doi:10.1016/j.jacr.2025.04.017
LIMITATIONS: The study relied on self-reported adherence data, which could overestimate screening percentages. Data collection occurred prior to updated United States Preventive Services Task Force guidelines recommending routine mammography screening for women starting at age 40 years every 2 years. The relatively small number of female veteran respondents limited the precision of population estimates. Additionally, the data were collected before the COVID-19 pandemic, which has been associated with reduced mammographic screening, particularly in medically underserved populations.
DISCLOSURES: Anand Narayan disclosed receiving financial support from Susan G. Komen Breast Cancer Foundation and National Academy of Medicine. The study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The remaining authors reported no potential conflicts of interest. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: A national survey of 8996 females reveals comparable mammography screening rates between those who identify as veterans (57.9%) and nonveterans (55.2%).
METHODOLOGY:
Researchers analyzed data from the 2019 National Health Interview Survey, a cross-sectional national survey tracking health information.
Female respondents aged 40 to 74 years without history of breast cancer were included in the analysis.
Analysis evaluated the association between screening and veteran status through logistic regression, adjusting for potential confounders.
Survey procedures accounted for complex sampling design to obtain valid estimates for the civilian, noninstitutionalized US population.
TAKEAWAY:
Analysis included 8996 female survey respondents, including 169 veterans (1.9%) and 320 (3.2%) reported having military health coverage.
Mammography screening rates within the last year were comparable between veterans (57.9%) and nonveterans (55.2%).
Veteran status showed no significant association with differences in mammography screening percentages (P = .96).
Among insured participants, military health insurance demonstrated no significant association with mammography screening percentages (P = .13).
The authors suggest that radiology practices should design proactive outreach strategies to address the needs of the growing number of female veterans who may face increased breast cancer risk due to military environmental exposures.
IN PRACTICE: “Although the results from our study demonstrate comparable mammography screening percentages, veterans may face additional risk factors for breast cancer due to occupational,” the authors argue.
SOURCE: This summary is based on a preprint published online in the Journal of the American College of Radiology: Milton A, Miles R, Gettle LM, Van Geertruyden P, Narayan AK. Utilization of Mammography Screening in Female Veterans: Cross-Sectional Survey Results from the National Health Interview Survey. J Am Coll Radiol. Published online April 24, 2025. doi:10.1016/j.jacr.2025.04.017
LIMITATIONS: The study relied on self-reported adherence data, which could overestimate screening percentages. Data collection occurred prior to updated United States Preventive Services Task Force guidelines recommending routine mammography screening for women starting at age 40 years every 2 years. The relatively small number of female veteran respondents limited the precision of population estimates. Additionally, the data were collected before the COVID-19 pandemic, which has been associated with reduced mammographic screening, particularly in medically underserved populations.
DISCLOSURES: Anand Narayan disclosed receiving financial support from Susan G. Komen Breast Cancer Foundation and National Academy of Medicine. The study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The remaining authors reported no potential conflicts of interest. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: A national survey of 8996 females reveals comparable mammography screening rates between those who identify as veterans (57.9%) and nonveterans (55.2%).
METHODOLOGY:
Researchers analyzed data from the 2019 National Health Interview Survey, a cross-sectional national survey tracking health information.
Female respondents aged 40 to 74 years without history of breast cancer were included in the analysis.
Analysis evaluated the association between screening and veteran status through logistic regression, adjusting for potential confounders.
Survey procedures accounted for complex sampling design to obtain valid estimates for the civilian, noninstitutionalized US population.
TAKEAWAY:
Analysis included 8996 female survey respondents, including 169 veterans (1.9%) and 320 (3.2%) reported having military health coverage.
Mammography screening rates within the last year were comparable between veterans (57.9%) and nonveterans (55.2%).
Veteran status showed no significant association with differences in mammography screening percentages (P = .96).
Among insured participants, military health insurance demonstrated no significant association with mammography screening percentages (P = .13).
The authors suggest that radiology practices should design proactive outreach strategies to address the needs of the growing number of female veterans who may face increased breast cancer risk due to military environmental exposures.
IN PRACTICE: “Although the results from our study demonstrate comparable mammography screening percentages, veterans may face additional risk factors for breast cancer due to occupational,” the authors argue.
SOURCE: This summary is based on a preprint published online in the Journal of the American College of Radiology: Milton A, Miles R, Gettle LM, Van Geertruyden P, Narayan AK. Utilization of Mammography Screening in Female Veterans: Cross-Sectional Survey Results from the National Health Interview Survey. J Am Coll Radiol. Published online April 24, 2025. doi:10.1016/j.jacr.2025.04.017
LIMITATIONS: The study relied on self-reported adherence data, which could overestimate screening percentages. Data collection occurred prior to updated United States Preventive Services Task Force guidelines recommending routine mammography screening for women starting at age 40 years every 2 years. The relatively small number of female veteran respondents limited the precision of population estimates. Additionally, the data were collected before the COVID-19 pandemic, which has been associated with reduced mammographic screening, particularly in medically underserved populations.
DISCLOSURES: Anand Narayan disclosed receiving financial support from Susan G. Komen Breast Cancer Foundation and National Academy of Medicine. The study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The remaining authors reported no potential conflicts of interest. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Sexual Assaults in Military Down, Benefits Claims Up
The number of sexual assaults in the US military dropped for the first time in a decade, according an annual report from the Pentagon, while benefits claims for assault survivors are on the rise.
Records show that 29,000 active-duty members reported being sexually assaulted in 2023, or 7000 fewer than in 2021. A confidential survey also found the number of service members who experienced some type of unwanted sexual contact dropped nearly 20%, leaving the Pentagon “cautiously optimistic“ its investments in preventing sexual assault and building a healthy climate are having an impact.
Despite these investments, issues persist. An Army anesthesiologist recently pleaded guilty to 41 charges of sexual misconduct involving 21 victims at Madigan Army Medical Center at Joint Base Lewis-McChord in Washington. The alleged incidents occurred between 2019 and 2022 and involved the doctor unnecessarily focusing on the genital area of patients during what he described as routine examinations. Maj. Michael Stockin faces nearly 14 years in prison, should the judge accept the plea agreement Stockin and his attorneys made with government prosecutors.
Additionally, a report from the Watson Institute of International and Public Affairs at Brown University indicated that 24% of active-duty women and 1.9% of active-duty men experienced sexual assault from 2001 to 2021.During post-9/11 wars, “the prioritization of force readiness above all else allowed the problem of sexual assault to fester, papering over internal violence and gender inequalities within military institutions,” the report said. There was also a slight uptick in reports of military sexual assaults in 2020, when troops were largely on lockdown as a result of the COVID-19 pandemic.
Efforts to address sexual assault in the military have increased in the past 10 years to the tune of 10 Department of Defense Inspector General engagements, 60 Government Accountability Office recommendations, > 200 government panel and task force recommendations, > 150 Congressional provisions, and > 50 Secretary of Defense initiatives. Additionally, the 2022 National Defense Authorization gave authority in sexual assault cases to independent prosecutors rather than commanders. Other reforms have included incorporating trauma-informed practices in the claims process.
Meanwhile, the US Department of Veterans Affairs (VA) has also been attempting to convince more sexual assault survivors to file claims for benefits. Assistant Deputy Under Secretary for Field Operations Kenesha Britton said in December that the VA has held 3500 events in the past 14 months focused on benefits for victims of military sexual assault and harassment. It appears to be working, as the VA received 57,400 claims for military sexual trauma in fiscal year 2024 (an 18% increase from 2023), and approved > 63% of them, compared to 40% more than a decade ago. Prior to Oct. 1, VA staffers processed > 11,000 cases in a single day twice. Since that date, they have processed that amount on 9 separate occasions.
“We recognize the remarkable courage it takes for survivors of military sexual trauma to seek the benefits and support they’ve earned,” Britton said. “Our mission is driven by a commitment to ensure survivors are met with care, dignity and sensitivity throughout the claims process.”
The increase in trust is a byproduct of the outreach campaigns, VA Under Secretary for Benefits Josh Jacobs said: “[M]ore veterans are coming in to apply for benefits and I think that has to do with building trust because we are actively trying to reach veterans telling them we want to connect them with their earned benefits.”
The number of sexual assaults in the US military dropped for the first time in a decade, according an annual report from the Pentagon, while benefits claims for assault survivors are on the rise.
Records show that 29,000 active-duty members reported being sexually assaulted in 2023, or 7000 fewer than in 2021. A confidential survey also found the number of service members who experienced some type of unwanted sexual contact dropped nearly 20%, leaving the Pentagon “cautiously optimistic“ its investments in preventing sexual assault and building a healthy climate are having an impact.
Despite these investments, issues persist. An Army anesthesiologist recently pleaded guilty to 41 charges of sexual misconduct involving 21 victims at Madigan Army Medical Center at Joint Base Lewis-McChord in Washington. The alleged incidents occurred between 2019 and 2022 and involved the doctor unnecessarily focusing on the genital area of patients during what he described as routine examinations. Maj. Michael Stockin faces nearly 14 years in prison, should the judge accept the plea agreement Stockin and his attorneys made with government prosecutors.
Additionally, a report from the Watson Institute of International and Public Affairs at Brown University indicated that 24% of active-duty women and 1.9% of active-duty men experienced sexual assault from 2001 to 2021.During post-9/11 wars, “the prioritization of force readiness above all else allowed the problem of sexual assault to fester, papering over internal violence and gender inequalities within military institutions,” the report said. There was also a slight uptick in reports of military sexual assaults in 2020, when troops were largely on lockdown as a result of the COVID-19 pandemic.
Efforts to address sexual assault in the military have increased in the past 10 years to the tune of 10 Department of Defense Inspector General engagements, 60 Government Accountability Office recommendations, > 200 government panel and task force recommendations, > 150 Congressional provisions, and > 50 Secretary of Defense initiatives. Additionally, the 2022 National Defense Authorization gave authority in sexual assault cases to independent prosecutors rather than commanders. Other reforms have included incorporating trauma-informed practices in the claims process.
Meanwhile, the US Department of Veterans Affairs (VA) has also been attempting to convince more sexual assault survivors to file claims for benefits. Assistant Deputy Under Secretary for Field Operations Kenesha Britton said in December that the VA has held 3500 events in the past 14 months focused on benefits for victims of military sexual assault and harassment. It appears to be working, as the VA received 57,400 claims for military sexual trauma in fiscal year 2024 (an 18% increase from 2023), and approved > 63% of them, compared to 40% more than a decade ago. Prior to Oct. 1, VA staffers processed > 11,000 cases in a single day twice. Since that date, they have processed that amount on 9 separate occasions.
“We recognize the remarkable courage it takes for survivors of military sexual trauma to seek the benefits and support they’ve earned,” Britton said. “Our mission is driven by a commitment to ensure survivors are met with care, dignity and sensitivity throughout the claims process.”
The increase in trust is a byproduct of the outreach campaigns, VA Under Secretary for Benefits Josh Jacobs said: “[M]ore veterans are coming in to apply for benefits and I think that has to do with building trust because we are actively trying to reach veterans telling them we want to connect them with their earned benefits.”
The number of sexual assaults in the US military dropped for the first time in a decade, according an annual report from the Pentagon, while benefits claims for assault survivors are on the rise.
Records show that 29,000 active-duty members reported being sexually assaulted in 2023, or 7000 fewer than in 2021. A confidential survey also found the number of service members who experienced some type of unwanted sexual contact dropped nearly 20%, leaving the Pentagon “cautiously optimistic“ its investments in preventing sexual assault and building a healthy climate are having an impact.
Despite these investments, issues persist. An Army anesthesiologist recently pleaded guilty to 41 charges of sexual misconduct involving 21 victims at Madigan Army Medical Center at Joint Base Lewis-McChord in Washington. The alleged incidents occurred between 2019 and 2022 and involved the doctor unnecessarily focusing on the genital area of patients during what he described as routine examinations. Maj. Michael Stockin faces nearly 14 years in prison, should the judge accept the plea agreement Stockin and his attorneys made with government prosecutors.
Additionally, a report from the Watson Institute of International and Public Affairs at Brown University indicated that 24% of active-duty women and 1.9% of active-duty men experienced sexual assault from 2001 to 2021.During post-9/11 wars, “the prioritization of force readiness above all else allowed the problem of sexual assault to fester, papering over internal violence and gender inequalities within military institutions,” the report said. There was also a slight uptick in reports of military sexual assaults in 2020, when troops were largely on lockdown as a result of the COVID-19 pandemic.
Efforts to address sexual assault in the military have increased in the past 10 years to the tune of 10 Department of Defense Inspector General engagements, 60 Government Accountability Office recommendations, > 200 government panel and task force recommendations, > 150 Congressional provisions, and > 50 Secretary of Defense initiatives. Additionally, the 2022 National Defense Authorization gave authority in sexual assault cases to independent prosecutors rather than commanders. Other reforms have included incorporating trauma-informed practices in the claims process.
Meanwhile, the US Department of Veterans Affairs (VA) has also been attempting to convince more sexual assault survivors to file claims for benefits. Assistant Deputy Under Secretary for Field Operations Kenesha Britton said in December that the VA has held 3500 events in the past 14 months focused on benefits for victims of military sexual assault and harassment. It appears to be working, as the VA received 57,400 claims for military sexual trauma in fiscal year 2024 (an 18% increase from 2023), and approved > 63% of them, compared to 40% more than a decade ago. Prior to Oct. 1, VA staffers processed > 11,000 cases in a single day twice. Since that date, they have processed that amount on 9 separate occasions.
“We recognize the remarkable courage it takes for survivors of military sexual trauma to seek the benefits and support they’ve earned,” Britton said. “Our mission is driven by a commitment to ensure survivors are met with care, dignity and sensitivity throughout the claims process.”
The increase in trust is a byproduct of the outreach campaigns, VA Under Secretary for Benefits Josh Jacobs said: “[M]ore veterans are coming in to apply for benefits and I think that has to do with building trust because we are actively trying to reach veterans telling them we want to connect them with their earned benefits.”
High Radon Levels Linked to Gestational Diabetes
New data link higher county-level radon exposure to gestational diabetes (GD) in women who haven’t previously given birth, emphasizing the need to consider environmental risks in maternal and fetal healthcare.
Yijia Zhang, PhD, with the Department of Obstetrics and Gynecology, Vagelos College of Physicians and Surgeons at Columbia University Irving Medical Center in New York, and colleagues found in a study of 9107 nulliparous pregnant women that those living in US counties with higher radon levels (2 picocuries [pCi]/L) had higher odds of developing GD than those in counties with lower (< 1 pCi/L) radon levels (odds ratio [OR], 1.37; 95% CI, 1.02-1.84.) The researchers used three radon categories, and the middle level was 1 to < 2 pCi/L.
Findings were published online on January 10 in JAMA Network Open. The researchers used data from The Nulliparous Pregnancy Outcomes Study: Monitoring Mothers-to-Be (nuMoM2b), a multicenter, prospective cohort study that examines factors associated with pregnancy-related outcomes.
“To our knowledge, this is the first study to examine the association between radon exposure and the risk of GD,” the authors wrote.
GD Affects 10% of Pregnancies
GD affects about 10% of pregnancies every year in the United States, according to the Centers for Disease Control and Prevention, and can affect women and offspring long term as it raises mothers’ risk of type 2 diabetes and cardiovascular disease and raises the risk for childhood obesity. Radon exposure’s link with lung cancer risk has been well established, but its link to other health risks is uncertain, the authors note.
The authors said their findings are hypothesis-generating and said, “It is vital to conduct studies that incorporate individual-level indoor radon exposure data,” to get closer to understanding the underlying mechanisms.
Individual-Level Exposure Measures Needed
They note that the average radon level in a county might not reflect an individual’s exposure and individual-level residential factors involved with radon exposure, such as household mitigation, and whether a dwelling has a basement, for instance, “are crucial for enhancing the precision of exposure assessment.”
In an invited commentary, Alberto Ruano-Ravina, PhD, and Lucía Martín-Gisbert, MSc, both with the Department of Preventive Medicine and Public Health at the University of Santiago de Compostela in Galicia, Spain, also urged that individual-level studies be conducted to further investigate radon’s link to health risks, noting that “[r]adon is possibly the most prevalent indoor carcinogen to which human beings are exposed.”
“There is no reason for not having these studies once we have some evidence of an association from ecological studies,” they wrote. They point out that reliable radon assessments are easy and inexpensive.
“The potential association of radon exposure with gestational diabetes or any other disease should be better analyzed using exclusively radon-prone areas. An observance of a dose-response effect may be indicative of a causal relationship, and it could be easily evidenced in radon-prone areas should such a relationship exist,” the commenters wrote.
Such areas have low, medium, high, and extremely high concentration levels, the commenters wrote. Zhang’s team, they point out, had to use only three exposure levels because the number of residents in high-exposure areas (exceeding 3 pCi/L) was too small.
“It is time now to move forward and really understand the full implications of radon exposure for health,” they concluded.
One coauthor reported serving on the board of directors for Merck for Mothers and as a board member for March for Moms outside the submitted work. One coauthor reported grants from the National Heart, Lung, and Blood Institute and the National Institutes of Health (NIH) during the conduct of the study. Four coauthors reported grants from the NIH during the conduct of the study. One coauthor reported grants from the NIH during the conduct of the study and being a cofounder of Naima Health and receiving personal fees from Organon outside the submitted work. Both commenters reported no relevant financial disclosures.
A version of this article appeared on Medscape.com.
New data link higher county-level radon exposure to gestational diabetes (GD) in women who haven’t previously given birth, emphasizing the need to consider environmental risks in maternal and fetal healthcare.
Yijia Zhang, PhD, with the Department of Obstetrics and Gynecology, Vagelos College of Physicians and Surgeons at Columbia University Irving Medical Center in New York, and colleagues found in a study of 9107 nulliparous pregnant women that those living in US counties with higher radon levels (2 picocuries [pCi]/L) had higher odds of developing GD than those in counties with lower (< 1 pCi/L) radon levels (odds ratio [OR], 1.37; 95% CI, 1.02-1.84.) The researchers used three radon categories, and the middle level was 1 to < 2 pCi/L.
Findings were published online on January 10 in JAMA Network Open. The researchers used data from The Nulliparous Pregnancy Outcomes Study: Monitoring Mothers-to-Be (nuMoM2b), a multicenter, prospective cohort study that examines factors associated with pregnancy-related outcomes.
“To our knowledge, this is the first study to examine the association between radon exposure and the risk of GD,” the authors wrote.
GD Affects 10% of Pregnancies
GD affects about 10% of pregnancies every year in the United States, according to the Centers for Disease Control and Prevention, and can affect women and offspring long term as it raises mothers’ risk of type 2 diabetes and cardiovascular disease and raises the risk for childhood obesity. Radon exposure’s link with lung cancer risk has been well established, but its link to other health risks is uncertain, the authors note.
The authors said their findings are hypothesis-generating and said, “It is vital to conduct studies that incorporate individual-level indoor radon exposure data,” to get closer to understanding the underlying mechanisms.
Individual-Level Exposure Measures Needed
They note that the average radon level in a county might not reflect an individual’s exposure and individual-level residential factors involved with radon exposure, such as household mitigation, and whether a dwelling has a basement, for instance, “are crucial for enhancing the precision of exposure assessment.”
In an invited commentary, Alberto Ruano-Ravina, PhD, and Lucía Martín-Gisbert, MSc, both with the Department of Preventive Medicine and Public Health at the University of Santiago de Compostela in Galicia, Spain, also urged that individual-level studies be conducted to further investigate radon’s link to health risks, noting that “[r]adon is possibly the most prevalent indoor carcinogen to which human beings are exposed.”
“There is no reason for not having these studies once we have some evidence of an association from ecological studies,” they wrote. They point out that reliable radon assessments are easy and inexpensive.
“The potential association of radon exposure with gestational diabetes or any other disease should be better analyzed using exclusively radon-prone areas. An observance of a dose-response effect may be indicative of a causal relationship, and it could be easily evidenced in radon-prone areas should such a relationship exist,” the commenters wrote.
Such areas have low, medium, high, and extremely high concentration levels, the commenters wrote. Zhang’s team, they point out, had to use only three exposure levels because the number of residents in high-exposure areas (exceeding 3 pCi/L) was too small.
“It is time now to move forward and really understand the full implications of radon exposure for health,” they concluded.
One coauthor reported serving on the board of directors for Merck for Mothers and as a board member for March for Moms outside the submitted work. One coauthor reported grants from the National Heart, Lung, and Blood Institute and the National Institutes of Health (NIH) during the conduct of the study. Four coauthors reported grants from the NIH during the conduct of the study. One coauthor reported grants from the NIH during the conduct of the study and being a cofounder of Naima Health and receiving personal fees from Organon outside the submitted work. Both commenters reported no relevant financial disclosures.
A version of this article appeared on Medscape.com.
New data link higher county-level radon exposure to gestational diabetes (GD) in women who haven’t previously given birth, emphasizing the need to consider environmental risks in maternal and fetal healthcare.
Yijia Zhang, PhD, with the Department of Obstetrics and Gynecology, Vagelos College of Physicians and Surgeons at Columbia University Irving Medical Center in New York, and colleagues found in a study of 9107 nulliparous pregnant women that those living in US counties with higher radon levels (2 picocuries [pCi]/L) had higher odds of developing GD than those in counties with lower (< 1 pCi/L) radon levels (odds ratio [OR], 1.37; 95% CI, 1.02-1.84.) The researchers used three radon categories, and the middle level was 1 to < 2 pCi/L.
Findings were published online on January 10 in JAMA Network Open. The researchers used data from The Nulliparous Pregnancy Outcomes Study: Monitoring Mothers-to-Be (nuMoM2b), a multicenter, prospective cohort study that examines factors associated with pregnancy-related outcomes.
“To our knowledge, this is the first study to examine the association between radon exposure and the risk of GD,” the authors wrote.
GD Affects 10% of Pregnancies
GD affects about 10% of pregnancies every year in the United States, according to the Centers for Disease Control and Prevention, and can affect women and offspring long term as it raises mothers’ risk of type 2 diabetes and cardiovascular disease and raises the risk for childhood obesity. Radon exposure’s link with lung cancer risk has been well established, but its link to other health risks is uncertain, the authors note.
The authors said their findings are hypothesis-generating and said, “It is vital to conduct studies that incorporate individual-level indoor radon exposure data,” to get closer to understanding the underlying mechanisms.
Individual-Level Exposure Measures Needed
They note that the average radon level in a county might not reflect an individual’s exposure and individual-level residential factors involved with radon exposure, such as household mitigation, and whether a dwelling has a basement, for instance, “are crucial for enhancing the precision of exposure assessment.”
In an invited commentary, Alberto Ruano-Ravina, PhD, and Lucía Martín-Gisbert, MSc, both with the Department of Preventive Medicine and Public Health at the University of Santiago de Compostela in Galicia, Spain, also urged that individual-level studies be conducted to further investigate radon’s link to health risks, noting that “[r]adon is possibly the most prevalent indoor carcinogen to which human beings are exposed.”
“There is no reason for not having these studies once we have some evidence of an association from ecological studies,” they wrote. They point out that reliable radon assessments are easy and inexpensive.
“The potential association of radon exposure with gestational diabetes or any other disease should be better analyzed using exclusively radon-prone areas. An observance of a dose-response effect may be indicative of a causal relationship, and it could be easily evidenced in radon-prone areas should such a relationship exist,” the commenters wrote.
Such areas have low, medium, high, and extremely high concentration levels, the commenters wrote. Zhang’s team, they point out, had to use only three exposure levels because the number of residents in high-exposure areas (exceeding 3 pCi/L) was too small.
“It is time now to move forward and really understand the full implications of radon exposure for health,” they concluded.
One coauthor reported serving on the board of directors for Merck for Mothers and as a board member for March for Moms outside the submitted work. One coauthor reported grants from the National Heart, Lung, and Blood Institute and the National Institutes of Health (NIH) during the conduct of the study. Four coauthors reported grants from the NIH during the conduct of the study. One coauthor reported grants from the NIH during the conduct of the study and being a cofounder of Naima Health and receiving personal fees from Organon outside the submitted work. Both commenters reported no relevant financial disclosures.
A version of this article appeared on Medscape.com.
Parenting in Later Life: How Old Is Too Old?
This transcript has been edited for clarity.
I want to talk about something that’s extremely controversial, but something that needs public discussion, in my view, as sometimes it doesn’t get the attention it deserves. That is: Are you ever too old to become a parent?
In my experience, this topic comes up when women — often, single women — decide that they haven’t had a child and they consider pursuing fertility services using in vitro fertilization, donor sperm, a younger woman’s egg, or an egg they’ve preserved, and they say they’d like to have a child.
I don’t have any huge objection to a younger woman with good health and energy trying to pursue parenting, but we’ve seen women try to do this in their 60s. It does seem to me, biologically, that is a high risk for anyone to undertake a pregnancy at that age. I think there’s agreement from obstetricians that they’re high risk.
I think it’s dangerous, if you’re going to be the single parent at that age, that you may wind up entering a nursing home by the time your child enters, say, high school. In thinking about parenting, sure, we want to think about our own values and what we want, and normally, people don’t tell us what to do. I’m not calling for any legislation here. I’m calling for an ethical discussion about the rights and wrongs of parenting at older age.
In response to the case I made against single women over age 60 trying to have children, it’s often brought up to me that men do it. Recently, there was a story about Al Pacino, who had a kid — I think he’s now 84, so he must have had the child at 83.
In an interview with Newsweek, he said he had this child with his ex, who was 30, a woman named Noor Alfallah. He also said he doesn’t see the child very much. He communicates mainly with that child as a co-parent through digital texting and internet contact. He said he uses video basically as a parent.
Why that is, I’m not sure. Did he have a falling out with his ex and has he been excluded? Is he in poor health such that he can’t really do parenting anymore?
I cite his case, and there are many other celebrities that we’ve heard about over the years who’ve had kids in their 80s, such as the former talk show host Larry King and, I believe, Clint Eastwood. There are cases that hit the news all the time about older men.
I think the same question should apply ethically. Again, I’m not saying we’re going to ban it or outlaw it, but it’s something we have to discuss and think through. I think doctors involved in helping a very old parent should raise the questions so that people can at least discuss them.
If you’re going to have a kid at 84, it means you’re not going to be around in any competent way by the time the kid hits high school. I’m not sure that’s in the child’s best interest. Certainly, there is the case that a younger woman could adequately raise the kid, but if something happens to her, you’re not going to be around in that age category to parent at all.
It’s also the case that older parents, if you’re using your sperm, may have the same issues as women, whose eggs age in their late 30s into their 40s; you’re more likely to transmit a genetic disease. We don’t talk about it often, but it is a fact that someone who’s thinking about parenting either naturally or using infertility techniques really should be responsible and think about it.
Bottom line: Am I going to say we should let Congress or a state legislature step in and say, you’re going to go to jail if you have a kid at age X? No. Ethics is there for a reason; it’s trying to make sure that you don’t do things that harm or hurt the interests of a kid.
If two older people have a child and they’re not likely to be there for a crucial period — say, the teenage years — and they haven’t made provisions for the care of the child, if both die, that’s a problem.
Am I doing this because I’m just going to do what I want to do, or am I going to really look out for the best interests of any child I might create?
This is food for thought about the question of when anyone is too old to parent. I know that’s partly determined by partner, resources, and many other variables, but I don’t believe that we should ignore the discussion of the ethics of the decision just out of respect for the idea that we’re not going to legislate.
Dr. Caplan is with the Division of Medical Ethics at New York University’s Grossman School of Medicine. He has disclosed relevant financial relationships with Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position) and Medscape.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I want to talk about something that’s extremely controversial, but something that needs public discussion, in my view, as sometimes it doesn’t get the attention it deserves. That is: Are you ever too old to become a parent?
In my experience, this topic comes up when women — often, single women — decide that they haven’t had a child and they consider pursuing fertility services using in vitro fertilization, donor sperm, a younger woman’s egg, or an egg they’ve preserved, and they say they’d like to have a child.
I don’t have any huge objection to a younger woman with good health and energy trying to pursue parenting, but we’ve seen women try to do this in their 60s. It does seem to me, biologically, that is a high risk for anyone to undertake a pregnancy at that age. I think there’s agreement from obstetricians that they’re high risk.
I think it’s dangerous, if you’re going to be the single parent at that age, that you may wind up entering a nursing home by the time your child enters, say, high school. In thinking about parenting, sure, we want to think about our own values and what we want, and normally, people don’t tell us what to do. I’m not calling for any legislation here. I’m calling for an ethical discussion about the rights and wrongs of parenting at older age.
In response to the case I made against single women over age 60 trying to have children, it’s often brought up to me that men do it. Recently, there was a story about Al Pacino, who had a kid — I think he’s now 84, so he must have had the child at 83.
In an interview with Newsweek, he said he had this child with his ex, who was 30, a woman named Noor Alfallah. He also said he doesn’t see the child very much. He communicates mainly with that child as a co-parent through digital texting and internet contact. He said he uses video basically as a parent.
Why that is, I’m not sure. Did he have a falling out with his ex and has he been excluded? Is he in poor health such that he can’t really do parenting anymore?
I cite his case, and there are many other celebrities that we’ve heard about over the years who’ve had kids in their 80s, such as the former talk show host Larry King and, I believe, Clint Eastwood. There are cases that hit the news all the time about older men.
I think the same question should apply ethically. Again, I’m not saying we’re going to ban it or outlaw it, but it’s something we have to discuss and think through. I think doctors involved in helping a very old parent should raise the questions so that people can at least discuss them.
If you’re going to have a kid at 84, it means you’re not going to be around in any competent way by the time the kid hits high school. I’m not sure that’s in the child’s best interest. Certainly, there is the case that a younger woman could adequately raise the kid, but if something happens to her, you’re not going to be around in that age category to parent at all.
It’s also the case that older parents, if you’re using your sperm, may have the same issues as women, whose eggs age in their late 30s into their 40s; you’re more likely to transmit a genetic disease. We don’t talk about it often, but it is a fact that someone who’s thinking about parenting either naturally or using infertility techniques really should be responsible and think about it.
Bottom line: Am I going to say we should let Congress or a state legislature step in and say, you’re going to go to jail if you have a kid at age X? No. Ethics is there for a reason; it’s trying to make sure that you don’t do things that harm or hurt the interests of a kid.
If two older people have a child and they’re not likely to be there for a crucial period — say, the teenage years — and they haven’t made provisions for the care of the child, if both die, that’s a problem.
Am I doing this because I’m just going to do what I want to do, or am I going to really look out for the best interests of any child I might create?
This is food for thought about the question of when anyone is too old to parent. I know that’s partly determined by partner, resources, and many other variables, but I don’t believe that we should ignore the discussion of the ethics of the decision just out of respect for the idea that we’re not going to legislate.
Dr. Caplan is with the Division of Medical Ethics at New York University’s Grossman School of Medicine. He has disclosed relevant financial relationships with Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position) and Medscape.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I want to talk about something that’s extremely controversial, but something that needs public discussion, in my view, as sometimes it doesn’t get the attention it deserves. That is: Are you ever too old to become a parent?
In my experience, this topic comes up when women — often, single women — decide that they haven’t had a child and they consider pursuing fertility services using in vitro fertilization, donor sperm, a younger woman’s egg, or an egg they’ve preserved, and they say they’d like to have a child.
I don’t have any huge objection to a younger woman with good health and energy trying to pursue parenting, but we’ve seen women try to do this in their 60s. It does seem to me, biologically, that is a high risk for anyone to undertake a pregnancy at that age. I think there’s agreement from obstetricians that they’re high risk.
I think it’s dangerous, if you’re going to be the single parent at that age, that you may wind up entering a nursing home by the time your child enters, say, high school. In thinking about parenting, sure, we want to think about our own values and what we want, and normally, people don’t tell us what to do. I’m not calling for any legislation here. I’m calling for an ethical discussion about the rights and wrongs of parenting at older age.
In response to the case I made against single women over age 60 trying to have children, it’s often brought up to me that men do it. Recently, there was a story about Al Pacino, who had a kid — I think he’s now 84, so he must have had the child at 83.
In an interview with Newsweek, he said he had this child with his ex, who was 30, a woman named Noor Alfallah. He also said he doesn’t see the child very much. He communicates mainly with that child as a co-parent through digital texting and internet contact. He said he uses video basically as a parent.
Why that is, I’m not sure. Did he have a falling out with his ex and has he been excluded? Is he in poor health such that he can’t really do parenting anymore?
I cite his case, and there are many other celebrities that we’ve heard about over the years who’ve had kids in their 80s, such as the former talk show host Larry King and, I believe, Clint Eastwood. There are cases that hit the news all the time about older men.
I think the same question should apply ethically. Again, I’m not saying we’re going to ban it or outlaw it, but it’s something we have to discuss and think through. I think doctors involved in helping a very old parent should raise the questions so that people can at least discuss them.
If you’re going to have a kid at 84, it means you’re not going to be around in any competent way by the time the kid hits high school. I’m not sure that’s in the child’s best interest. Certainly, there is the case that a younger woman could adequately raise the kid, but if something happens to her, you’re not going to be around in that age category to parent at all.
It’s also the case that older parents, if you’re using your sperm, may have the same issues as women, whose eggs age in their late 30s into their 40s; you’re more likely to transmit a genetic disease. We don’t talk about it often, but it is a fact that someone who’s thinking about parenting either naturally or using infertility techniques really should be responsible and think about it.
Bottom line: Am I going to say we should let Congress or a state legislature step in and say, you’re going to go to jail if you have a kid at age X? No. Ethics is there for a reason; it’s trying to make sure that you don’t do things that harm or hurt the interests of a kid.
If two older people have a child and they’re not likely to be there for a crucial period — say, the teenage years — and they haven’t made provisions for the care of the child, if both die, that’s a problem.
Am I doing this because I’m just going to do what I want to do, or am I going to really look out for the best interests of any child I might create?
This is food for thought about the question of when anyone is too old to parent. I know that’s partly determined by partner, resources, and many other variables, but I don’t believe that we should ignore the discussion of the ethics of the decision just out of respect for the idea that we’re not going to legislate.
Dr. Caplan is with the Division of Medical Ethics at New York University’s Grossman School of Medicine. He has disclosed relevant financial relationships with Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position) and Medscape.
A version of this article appeared on Medscape.com.
Do Antibiotics Before Conception Affect Fertility?
Is there a connection between antibiotics taken before conception and adverse outcomes, such as reduced fertility, miscarriages, and congenital malformations?
A meta-analysis published in the journal eClinicalMedicine suggests a potential link between antibiotics taken before conception and negative outcomes, such as reduced fertility, miscarriages, and congenital malformations. However, a German expert in reproductive toxicology warned against drawing false conclusions.
“It would be fatal if women who want to have children refused necessary antibiotic treatment because they are afraid of infertility, miscarriages, and malformations,” said Wolfgang Paulus, MD, from the Reproductive Toxicology Advisory Center at the University Women’s Hospital in Ulm, Germany. In an interview, the expert criticized not only the authors’ conclusions but also the selection of studies included in the meta-analysis.
Confusion Over Use and Exposure
The meta-analysis, conducted by Bekalu Kassie Alemu, PhD, and colleagues from the Department of Obstetrics and Gynecology at The Chinese University of Hong Kong included 15 studies involving over 1.2 million women to examine how preconception antibiotic use affects fertility and pregnancy outcomes. In most studies (n = 11) that were included in the meta-analysis, fertility was examined as an endpoint, primarily in infertile women. One study involved Danish pharmacy employees who handled antibiotics at work.
“Not only was the therapeutic use of antibiotics not examined in this study, but the biological plausibility is completely lacking in this context,” Paulus noted.
The possible effects of preconception antibiotics on miscarriages were investigated in four studies, while two studies focused on congenital malformations as an endpoint.
Mixed Findings on Infertility
Regarding infertility, the authors reported abnormalities in macrolides and sulfonamides. Women who had received macrolide antibiotics, such as azithromycin, before conception showed a 35% reduction in fertility rates.
However, Paulus questioned whether this was solely because of macrolides. “Macrolide antibiotics are typically used for chlamydia, and chlamydia infection is a significant factor in women with unmet fertility desires,” he explained. Often, the chlamydia has already caused damage, such as inflammatory processes in the fallopian tubes, contributing to infertility that cannot be resolved by administering antibiotics.
The meta-analysis also showed that women who received sulfonamide before conception had a 2.35-fold increased likelihood of infertility. However, this association is not always one-sided. The results for tetracyclines were heterogeneous; while chlortetracycline appeared to increase the risk for infertility, exposure to oxytetracycline appeared to decrease it.
Treatment with oxytetracycline and beta-lactam antibiotics (except penicillin G) was associated with a 64% lower likelihood of infertility. The authors also found that fluoroquinolone antibiotics were associated with a 13% lower likelihood of infertility.
Miscarriage and Malformation Risks
Alemu and colleagues found a significant association between the use of antibiotics before conception and adverse pregnancy outcomes, showing a 34% increased risk for miscarriages and an 85% higher risk for congenital malformations with the use of trimethoprim during preconception. These findings highlight the need for caution regarding antibiotic use in women who are planning to conceive.
“Most antibiotics have half-lives of only a few hours. Therefore, antibiotics administered before conception can hardly have a direct effect on embryonic development,” Paulus noted. He pointed out that extensive data exist on most antibiotic classes included in this meta-analysis regarding childhood anomalies when used during the sensitive phase of organ development. These data do not indicate an increased risk for malformation. Therefore, the increased risk for malformations due to exposure before conception seems less plausible.
Alemu and colleagues assumed that antibiotics might negatively affect female reproductive health by disrupting the gut microbiome. The reasons for the reduced risk for infertility associated with beta-lactams and fluoroquinolones require further investigation. They reach a significant conclusion: “Preconception antibiotics exposure in females increases the risk of infertility, miscarriage, and congenital anomalies.” However, differences exist between the antibiotic classes. While the risk for infertility, spontaneous miscarriages, and congenital malformations increases with the use of macrolide antibiotics, sulfonamides, and trimethoprim, it decreases with the use of beta-lactams and fluoroquinolone antibiotics.
Expert Disagreement
“It is conceivable that the use of antibiotics damages the physiological environment, such as in the vaginal area. This may allow unwanted microbes to establish themselves, leading to more adverse outcomes such as infertility and miscarriages,” Paulus acknowledged.
Disruption of the microbiome due to antibiotic therapy could also result in a deficiency in relevant vitamins and trace elements (eg, folic acid), which could contribute to organogenesis disorders. Therefore, it may be beneficial to stabilize the gut and vaginal flora using probiotics after antibiotic treatment.
However, Paulus disagrees with the study conclusions. First, the studies included in the meta-analysis, which were largely observational, did not allow for the direct effect of antibiotics on the examined outcomes. Second, “quinolone antibiotics are highlighted as positive here, as if they were less problematic for patients trying to have children.”
Quinolone antibiotics are generally “frowned upon,” regardless of whether the patient wants to have children, as they can cause damage to the tendons, muscles, joints, and nervous system. They are currently used only as reserve medications.
“Quinolone antibiotics should not be administered during pregnancy, as they have already caused problems in animal studies, and they should not be used before pregnancy because of their side-effect profile,” Paulus stressed.
Serious Consequences
Paulus clarified:
In these cases, antibiotic treatment is appropriate, and there should be no fear of adverse effects on fertility or pregnancy outcomes. “If antibiotics are not given and the infection worsens, the patient will be even less likely to conceive successfully.”
This story was translated from Medscape’s German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Is there a connection between antibiotics taken before conception and adverse outcomes, such as reduced fertility, miscarriages, and congenital malformations?
A meta-analysis published in the journal eClinicalMedicine suggests a potential link between antibiotics taken before conception and negative outcomes, such as reduced fertility, miscarriages, and congenital malformations. However, a German expert in reproductive toxicology warned against drawing false conclusions.
“It would be fatal if women who want to have children refused necessary antibiotic treatment because they are afraid of infertility, miscarriages, and malformations,” said Wolfgang Paulus, MD, from the Reproductive Toxicology Advisory Center at the University Women’s Hospital in Ulm, Germany. In an interview, the expert criticized not only the authors’ conclusions but also the selection of studies included in the meta-analysis.
Confusion Over Use and Exposure
The meta-analysis, conducted by Bekalu Kassie Alemu, PhD, and colleagues from the Department of Obstetrics and Gynecology at The Chinese University of Hong Kong included 15 studies involving over 1.2 million women to examine how preconception antibiotic use affects fertility and pregnancy outcomes. In most studies (n = 11) that were included in the meta-analysis, fertility was examined as an endpoint, primarily in infertile women. One study involved Danish pharmacy employees who handled antibiotics at work.
“Not only was the therapeutic use of antibiotics not examined in this study, but the biological plausibility is completely lacking in this context,” Paulus noted.
The possible effects of preconception antibiotics on miscarriages were investigated in four studies, while two studies focused on congenital malformations as an endpoint.
Mixed Findings on Infertility
Regarding infertility, the authors reported abnormalities in macrolides and sulfonamides. Women who had received macrolide antibiotics, such as azithromycin, before conception showed a 35% reduction in fertility rates.
However, Paulus questioned whether this was solely because of macrolides. “Macrolide antibiotics are typically used for chlamydia, and chlamydia infection is a significant factor in women with unmet fertility desires,” he explained. Often, the chlamydia has already caused damage, such as inflammatory processes in the fallopian tubes, contributing to infertility that cannot be resolved by administering antibiotics.
The meta-analysis also showed that women who received sulfonamide before conception had a 2.35-fold increased likelihood of infertility. However, this association is not always one-sided. The results for tetracyclines were heterogeneous; while chlortetracycline appeared to increase the risk for infertility, exposure to oxytetracycline appeared to decrease it.
Treatment with oxytetracycline and beta-lactam antibiotics (except penicillin G) was associated with a 64% lower likelihood of infertility. The authors also found that fluoroquinolone antibiotics were associated with a 13% lower likelihood of infertility.
Miscarriage and Malformation Risks
Alemu and colleagues found a significant association between the use of antibiotics before conception and adverse pregnancy outcomes, showing a 34% increased risk for miscarriages and an 85% higher risk for congenital malformations with the use of trimethoprim during preconception. These findings highlight the need for caution regarding antibiotic use in women who are planning to conceive.
“Most antibiotics have half-lives of only a few hours. Therefore, antibiotics administered before conception can hardly have a direct effect on embryonic development,” Paulus noted. He pointed out that extensive data exist on most antibiotic classes included in this meta-analysis regarding childhood anomalies when used during the sensitive phase of organ development. These data do not indicate an increased risk for malformation. Therefore, the increased risk for malformations due to exposure before conception seems less plausible.
Alemu and colleagues assumed that antibiotics might negatively affect female reproductive health by disrupting the gut microbiome. The reasons for the reduced risk for infertility associated with beta-lactams and fluoroquinolones require further investigation. They reach a significant conclusion: “Preconception antibiotics exposure in females increases the risk of infertility, miscarriage, and congenital anomalies.” However, differences exist between the antibiotic classes. While the risk for infertility, spontaneous miscarriages, and congenital malformations increases with the use of macrolide antibiotics, sulfonamides, and trimethoprim, it decreases with the use of beta-lactams and fluoroquinolone antibiotics.
Expert Disagreement
“It is conceivable that the use of antibiotics damages the physiological environment, such as in the vaginal area. This may allow unwanted microbes to establish themselves, leading to more adverse outcomes such as infertility and miscarriages,” Paulus acknowledged.
Disruption of the microbiome due to antibiotic therapy could also result in a deficiency in relevant vitamins and trace elements (eg, folic acid), which could contribute to organogenesis disorders. Therefore, it may be beneficial to stabilize the gut and vaginal flora using probiotics after antibiotic treatment.
However, Paulus disagrees with the study conclusions. First, the studies included in the meta-analysis, which were largely observational, did not allow for the direct effect of antibiotics on the examined outcomes. Second, “quinolone antibiotics are highlighted as positive here, as if they were less problematic for patients trying to have children.”
Quinolone antibiotics are generally “frowned upon,” regardless of whether the patient wants to have children, as they can cause damage to the tendons, muscles, joints, and nervous system. They are currently used only as reserve medications.
“Quinolone antibiotics should not be administered during pregnancy, as they have already caused problems in animal studies, and they should not be used before pregnancy because of their side-effect profile,” Paulus stressed.
Serious Consequences
Paulus clarified:
In these cases, antibiotic treatment is appropriate, and there should be no fear of adverse effects on fertility or pregnancy outcomes. “If antibiotics are not given and the infection worsens, the patient will be even less likely to conceive successfully.”
This story was translated from Medscape’s German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Is there a connection between antibiotics taken before conception and adverse outcomes, such as reduced fertility, miscarriages, and congenital malformations?
A meta-analysis published in the journal eClinicalMedicine suggests a potential link between antibiotics taken before conception and negative outcomes, such as reduced fertility, miscarriages, and congenital malformations. However, a German expert in reproductive toxicology warned against drawing false conclusions.
“It would be fatal if women who want to have children refused necessary antibiotic treatment because they are afraid of infertility, miscarriages, and malformations,” said Wolfgang Paulus, MD, from the Reproductive Toxicology Advisory Center at the University Women’s Hospital in Ulm, Germany. In an interview, the expert criticized not only the authors’ conclusions but also the selection of studies included in the meta-analysis.
Confusion Over Use and Exposure
The meta-analysis, conducted by Bekalu Kassie Alemu, PhD, and colleagues from the Department of Obstetrics and Gynecology at The Chinese University of Hong Kong included 15 studies involving over 1.2 million women to examine how preconception antibiotic use affects fertility and pregnancy outcomes. In most studies (n = 11) that were included in the meta-analysis, fertility was examined as an endpoint, primarily in infertile women. One study involved Danish pharmacy employees who handled antibiotics at work.
“Not only was the therapeutic use of antibiotics not examined in this study, but the biological plausibility is completely lacking in this context,” Paulus noted.
The possible effects of preconception antibiotics on miscarriages were investigated in four studies, while two studies focused on congenital malformations as an endpoint.
Mixed Findings on Infertility
Regarding infertility, the authors reported abnormalities in macrolides and sulfonamides. Women who had received macrolide antibiotics, such as azithromycin, before conception showed a 35% reduction in fertility rates.
However, Paulus questioned whether this was solely because of macrolides. “Macrolide antibiotics are typically used for chlamydia, and chlamydia infection is a significant factor in women with unmet fertility desires,” he explained. Often, the chlamydia has already caused damage, such as inflammatory processes in the fallopian tubes, contributing to infertility that cannot be resolved by administering antibiotics.
The meta-analysis also showed that women who received sulfonamide before conception had a 2.35-fold increased likelihood of infertility. However, this association is not always one-sided. The results for tetracyclines were heterogeneous; while chlortetracycline appeared to increase the risk for infertility, exposure to oxytetracycline appeared to decrease it.
Treatment with oxytetracycline and beta-lactam antibiotics (except penicillin G) was associated with a 64% lower likelihood of infertility. The authors also found that fluoroquinolone antibiotics were associated with a 13% lower likelihood of infertility.
Miscarriage and Malformation Risks
Alemu and colleagues found a significant association between the use of antibiotics before conception and adverse pregnancy outcomes, showing a 34% increased risk for miscarriages and an 85% higher risk for congenital malformations with the use of trimethoprim during preconception. These findings highlight the need for caution regarding antibiotic use in women who are planning to conceive.
“Most antibiotics have half-lives of only a few hours. Therefore, antibiotics administered before conception can hardly have a direct effect on embryonic development,” Paulus noted. He pointed out that extensive data exist on most antibiotic classes included in this meta-analysis regarding childhood anomalies when used during the sensitive phase of organ development. These data do not indicate an increased risk for malformation. Therefore, the increased risk for malformations due to exposure before conception seems less plausible.
Alemu and colleagues assumed that antibiotics might negatively affect female reproductive health by disrupting the gut microbiome. The reasons for the reduced risk for infertility associated with beta-lactams and fluoroquinolones require further investigation. They reach a significant conclusion: “Preconception antibiotics exposure in females increases the risk of infertility, miscarriage, and congenital anomalies.” However, differences exist between the antibiotic classes. While the risk for infertility, spontaneous miscarriages, and congenital malformations increases with the use of macrolide antibiotics, sulfonamides, and trimethoprim, it decreases with the use of beta-lactams and fluoroquinolone antibiotics.
Expert Disagreement
“It is conceivable that the use of antibiotics damages the physiological environment, such as in the vaginal area. This may allow unwanted microbes to establish themselves, leading to more adverse outcomes such as infertility and miscarriages,” Paulus acknowledged.
Disruption of the microbiome due to antibiotic therapy could also result in a deficiency in relevant vitamins and trace elements (eg, folic acid), which could contribute to organogenesis disorders. Therefore, it may be beneficial to stabilize the gut and vaginal flora using probiotics after antibiotic treatment.
However, Paulus disagrees with the study conclusions. First, the studies included in the meta-analysis, which were largely observational, did not allow for the direct effect of antibiotics on the examined outcomes. Second, “quinolone antibiotics are highlighted as positive here, as if they were less problematic for patients trying to have children.”
Quinolone antibiotics are generally “frowned upon,” regardless of whether the patient wants to have children, as they can cause damage to the tendons, muscles, joints, and nervous system. They are currently used only as reserve medications.
“Quinolone antibiotics should not be administered during pregnancy, as they have already caused problems in animal studies, and they should not be used before pregnancy because of their side-effect profile,” Paulus stressed.
Serious Consequences
Paulus clarified:
In these cases, antibiotic treatment is appropriate, and there should be no fear of adverse effects on fertility or pregnancy outcomes. “If antibiotics are not given and the infection worsens, the patient will be even less likely to conceive successfully.”
This story was translated from Medscape’s German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
FROM ECLINICALMEDICINE
CDK 4/6 Blocker Prolongs Survival in HER2+ Metastatic Breast Cancer
according to the results of the phase 3 PATINA study.
This regimen “may represent a new standard of care” for these patients, said principal investigator and presenter Otto Metzger, MD, a medical breast oncologist at the Dana-Farber Cancer Institute in Boston, Massachusetts, who presented the findings at the San Antonio Breast Cancer Symposium (SABCS) 2024.
The open-label PATINA trial, which was conducted in Europe, Australia, New Zealand, and the United States, included a total of 518 patients. Patients received first-line treatment of six to eight cycles of induction chemotherapy plus anti-HER2 therapy. Researchers then randomized patients to either palbociclib plus anti-HER2 and endocrine therapy (n = 261) or to anti-HER2 and endocrine therapy alone (n = 257).
Patients did not progress on induction therapy, which likely would have signaled early resistance to anti-HER2 treatment. For anti-HER2 therapy, 97.3% received a combination of trastuzumab and pertuzumab. For endocrine therapy, 90.9% received an aromatase inhibitor.
Metzger and colleagues found that median progression-free survival was 1.3 years longer in patients receiving palbociclib — 3.7 years in the palbociclib arm vs 2.4 years in the control group (hazard ratio [HR], 0.74; P = .0074).
Although overall survival outcomes are immature, 5-year survival rates were slightly better in the palbociclib arm — 74.3% with palbociclib vs 69.8% without it — but the difference was not statistically significant.
Grade 3 neutropenia was the most frequent adverse event in the palbociclib arm (63.2% vs 2%). Grades 2 and 3 fatigue, stomatitis, and diarrhea were also more common with palbociclib. Grade 4 adverse events occurred in 12.3% of those receiving palbociclib and 8.9% of those who did not. There were no treatment-related deaths.
“We’re very impressed with the results,” said Metzger.
On the basis of previous studies, it’s believed that CDK 4/6 inhibition counteracts the development of resistance to anti-HER2 and endocrine therapies, which likely explains the benefit found in the trial.
But even without CDK 4/6 inhibition, the progression-free survival of 2.4 years in the control arm “far exceed[ed] our expectations,” Metzger reported. This may have occurred because the control arm received endocrine therapy, something previous trials of anti-HER2 therapy have avoided because of tolerability and other concerns.
These findings, however, support “the common use of endocrine therapy,” Metzger said.
‘Incredible’ Results
The progression-free survival as well as overall survival results in the trial are “incredible,” said study discussant Sara Hurvitz, MD, a medical breast oncologist at the Fred Hutch Cancer Center in Seattle, Washington. This is “historic and very important data.”
Hurvitz even suggested the results might mean that patients who fit the PATINA criteria can avoid the toxicity of upfront trastuzumab deruxtecan and use the PATINA regimen instead, potentially preserving their quality of life for longer.
Another study discussant, Virginia Kaklamani, MD, a medical breast oncologist at the University of Texas MD Anderson Cancer Center, San Antonio, had a similar thought.
In PATINA, “we’re talking about patients being on a treatment that’s well tolerated, where patients continue to work and continue with their lives despite being on treatment for metastatic breast cancer for 4 years, which is remarkable,” Kaklamani said.
Many of us have dabbled with giving CDK 4/6 inhibitors in triple-positive breast cancer, but “now we have more definitive data,” she said. The approach can help “maintain the quality of life of our patients for a longer period of time” and delay the use of chemotherapy in the second line, she added.
Metzger said Pfizer, the maker of palbociclib, plans to file for a HER2-positive indication with the Food and Drug Administration based on the trial results.
For now, the CDK 4/6 blocker is only indicated in combination with endocrine therapy for HR-positive, HER2-negative metastatic disease.
In response to a question about using the PATINA regimen in patients who don’t get chemotherapy induction, Metzger noted that, “while the study didn’t test this directly, I would argue that this data is quite compelling” for using palbociclib plus anti-HER2 and endocrine therapy, even without chemotherapy induction.
The work was funded by palbociclib maker Pfizer. Metzger had no disclosures. Hurvitz has numerous industry ties, including being a researcher and advisor to Pfizer. Kaklamani also has numerous industry ties, including reporting personal/consulting fees from Pfizer Canada.
A version of this article first appeared on Medscape.com.
according to the results of the phase 3 PATINA study.
This regimen “may represent a new standard of care” for these patients, said principal investigator and presenter Otto Metzger, MD, a medical breast oncologist at the Dana-Farber Cancer Institute in Boston, Massachusetts, who presented the findings at the San Antonio Breast Cancer Symposium (SABCS) 2024.
The open-label PATINA trial, which was conducted in Europe, Australia, New Zealand, and the United States, included a total of 518 patients. Patients received first-line treatment of six to eight cycles of induction chemotherapy plus anti-HER2 therapy. Researchers then randomized patients to either palbociclib plus anti-HER2 and endocrine therapy (n = 261) or to anti-HER2 and endocrine therapy alone (n = 257).
Patients did not progress on induction therapy, which likely would have signaled early resistance to anti-HER2 treatment. For anti-HER2 therapy, 97.3% received a combination of trastuzumab and pertuzumab. For endocrine therapy, 90.9% received an aromatase inhibitor.
Metzger and colleagues found that median progression-free survival was 1.3 years longer in patients receiving palbociclib — 3.7 years in the palbociclib arm vs 2.4 years in the control group (hazard ratio [HR], 0.74; P = .0074).
Although overall survival outcomes are immature, 5-year survival rates were slightly better in the palbociclib arm — 74.3% with palbociclib vs 69.8% without it — but the difference was not statistically significant.
Grade 3 neutropenia was the most frequent adverse event in the palbociclib arm (63.2% vs 2%). Grades 2 and 3 fatigue, stomatitis, and diarrhea were also more common with palbociclib. Grade 4 adverse events occurred in 12.3% of those receiving palbociclib and 8.9% of those who did not. There were no treatment-related deaths.
“We’re very impressed with the results,” said Metzger.
On the basis of previous studies, it’s believed that CDK 4/6 inhibition counteracts the development of resistance to anti-HER2 and endocrine therapies, which likely explains the benefit found in the trial.
But even without CDK 4/6 inhibition, the progression-free survival of 2.4 years in the control arm “far exceed[ed] our expectations,” Metzger reported. This may have occurred because the control arm received endocrine therapy, something previous trials of anti-HER2 therapy have avoided because of tolerability and other concerns.
These findings, however, support “the common use of endocrine therapy,” Metzger said.
‘Incredible’ Results
The progression-free survival as well as overall survival results in the trial are “incredible,” said study discussant Sara Hurvitz, MD, a medical breast oncologist at the Fred Hutch Cancer Center in Seattle, Washington. This is “historic and very important data.”
Hurvitz even suggested the results might mean that patients who fit the PATINA criteria can avoid the toxicity of upfront trastuzumab deruxtecan and use the PATINA regimen instead, potentially preserving their quality of life for longer.
Another study discussant, Virginia Kaklamani, MD, a medical breast oncologist at the University of Texas MD Anderson Cancer Center, San Antonio, had a similar thought.
In PATINA, “we’re talking about patients being on a treatment that’s well tolerated, where patients continue to work and continue with their lives despite being on treatment for metastatic breast cancer for 4 years, which is remarkable,” Kaklamani said.
Many of us have dabbled with giving CDK 4/6 inhibitors in triple-positive breast cancer, but “now we have more definitive data,” she said. The approach can help “maintain the quality of life of our patients for a longer period of time” and delay the use of chemotherapy in the second line, she added.
Metzger said Pfizer, the maker of palbociclib, plans to file for a HER2-positive indication with the Food and Drug Administration based on the trial results.
For now, the CDK 4/6 blocker is only indicated in combination with endocrine therapy for HR-positive, HER2-negative metastatic disease.
In response to a question about using the PATINA regimen in patients who don’t get chemotherapy induction, Metzger noted that, “while the study didn’t test this directly, I would argue that this data is quite compelling” for using palbociclib plus anti-HER2 and endocrine therapy, even without chemotherapy induction.
The work was funded by palbociclib maker Pfizer. Metzger had no disclosures. Hurvitz has numerous industry ties, including being a researcher and advisor to Pfizer. Kaklamani also has numerous industry ties, including reporting personal/consulting fees from Pfizer Canada.
A version of this article first appeared on Medscape.com.
according to the results of the phase 3 PATINA study.
This regimen “may represent a new standard of care” for these patients, said principal investigator and presenter Otto Metzger, MD, a medical breast oncologist at the Dana-Farber Cancer Institute in Boston, Massachusetts, who presented the findings at the San Antonio Breast Cancer Symposium (SABCS) 2024.
The open-label PATINA trial, which was conducted in Europe, Australia, New Zealand, and the United States, included a total of 518 patients. Patients received first-line treatment of six to eight cycles of induction chemotherapy plus anti-HER2 therapy. Researchers then randomized patients to either palbociclib plus anti-HER2 and endocrine therapy (n = 261) or to anti-HER2 and endocrine therapy alone (n = 257).
Patients did not progress on induction therapy, which likely would have signaled early resistance to anti-HER2 treatment. For anti-HER2 therapy, 97.3% received a combination of trastuzumab and pertuzumab. For endocrine therapy, 90.9% received an aromatase inhibitor.
Metzger and colleagues found that median progression-free survival was 1.3 years longer in patients receiving palbociclib — 3.7 years in the palbociclib arm vs 2.4 years in the control group (hazard ratio [HR], 0.74; P = .0074).
Although overall survival outcomes are immature, 5-year survival rates were slightly better in the palbociclib arm — 74.3% with palbociclib vs 69.8% without it — but the difference was not statistically significant.
Grade 3 neutropenia was the most frequent adverse event in the palbociclib arm (63.2% vs 2%). Grades 2 and 3 fatigue, stomatitis, and diarrhea were also more common with palbociclib. Grade 4 adverse events occurred in 12.3% of those receiving palbociclib and 8.9% of those who did not. There were no treatment-related deaths.
“We’re very impressed with the results,” said Metzger.
On the basis of previous studies, it’s believed that CDK 4/6 inhibition counteracts the development of resistance to anti-HER2 and endocrine therapies, which likely explains the benefit found in the trial.
But even without CDK 4/6 inhibition, the progression-free survival of 2.4 years in the control arm “far exceed[ed] our expectations,” Metzger reported. This may have occurred because the control arm received endocrine therapy, something previous trials of anti-HER2 therapy have avoided because of tolerability and other concerns.
These findings, however, support “the common use of endocrine therapy,” Metzger said.
‘Incredible’ Results
The progression-free survival as well as overall survival results in the trial are “incredible,” said study discussant Sara Hurvitz, MD, a medical breast oncologist at the Fred Hutch Cancer Center in Seattle, Washington. This is “historic and very important data.”
Hurvitz even suggested the results might mean that patients who fit the PATINA criteria can avoid the toxicity of upfront trastuzumab deruxtecan and use the PATINA regimen instead, potentially preserving their quality of life for longer.
Another study discussant, Virginia Kaklamani, MD, a medical breast oncologist at the University of Texas MD Anderson Cancer Center, San Antonio, had a similar thought.
In PATINA, “we’re talking about patients being on a treatment that’s well tolerated, where patients continue to work and continue with their lives despite being on treatment for metastatic breast cancer for 4 years, which is remarkable,” Kaklamani said.
Many of us have dabbled with giving CDK 4/6 inhibitors in triple-positive breast cancer, but “now we have more definitive data,” she said. The approach can help “maintain the quality of life of our patients for a longer period of time” and delay the use of chemotherapy in the second line, she added.
Metzger said Pfizer, the maker of palbociclib, plans to file for a HER2-positive indication with the Food and Drug Administration based on the trial results.
For now, the CDK 4/6 blocker is only indicated in combination with endocrine therapy for HR-positive, HER2-negative metastatic disease.
In response to a question about using the PATINA regimen in patients who don’t get chemotherapy induction, Metzger noted that, “while the study didn’t test this directly, I would argue that this data is quite compelling” for using palbociclib plus anti-HER2 and endocrine therapy, even without chemotherapy induction.
The work was funded by palbociclib maker Pfizer. Metzger had no disclosures. Hurvitz has numerous industry ties, including being a researcher and advisor to Pfizer. Kaklamani also has numerous industry ties, including reporting personal/consulting fees from Pfizer Canada.
A version of this article first appeared on Medscape.com.
FROM SABCS 2024