User login
Asenapine transdermal system for schizophrenia
The asenapine transdermal system is available in 3 patch sizes: 20, 30, and 40 cm2, which deliver 3.8, 5.7, and 7.6 mg/24 hours of asenapine, respectively.3 Based on the average exposure (area under the plasma concentration curve [AUC]) of asenapine, 3.8 mg/24 hours corresponds to 5 mg twice daily of sublingual asenapine, and 7.6 mg/24 hours corresponds to 10 mg twice daily of sublingual asenapine.3 The “in-between” dose strength of 5.7 mg/24 hours would correspond to exposure to a total of 15 mg/d of sublingual asenapine. The recommended starting dose for asenapine transdermal system is 3.8 mg/24 hours. The dosage may be increased to 5.7 mg/24 hours or 7.6 mg/24 hours, as needed, after 1 week. The safety of doses above 7.6 mg/24 hours has not been evaluated in clinical studies. Asenapine transdermal system is applied once daily and should be worn for 24 hours only, with only 1 patch at any time. Application sites include the upper arm, upper back, abdomen, and hip. A different application site of clean, dry, intact skin should be selected each time a new patch is applied. Although showering is permitted, the use of asenapine transdermal system during swimming or taking a bath has not been evaluated. Of note, prolonged application of heat over an asenapine transdermal system increases plasma concentrations of asenapine, and thus application of external heat sources (eg, heating pads) over the patch should be avoided.
How it works
Product labeling notes that asenapine is an atypical antipsychotic, and that its efficacy in schizophrenia could be mediated through a combination of antagonist activity at dopamine D2 and serotonin 5-HT2A receptors.3 The pharmacodynamic profile of asenapine is complex5 and receptor-binding assays performed using cloned human serotonin, norepinephrine, dopamine, histamine, and muscarinic receptors demonstrated picomolar affinity (extremely high) for 5-HT2C and 5-HT2A receptors, subnanomolar affinity (very high) for 5-HT7, 5-HT2B, 5-HT6, and D3 receptors, and nanomolar affinity (high) for D2 receptors, as well as histamine H1, D4, a1-adrenergic, a2-adrenergic, D1, 5-HT5, 5-HT1A, 5-HT1B, and histamine H2 receptors. Activity of asenapine is that of antagonism at these receptors. Asenapine has no appreciable affinity for muscarinic cholinergic receptors.
The asenapine receptor-binding “fingerprint” differs from that of other antipsychotics. Some of these receptor affinities are of special interest in terms of potential efficacy for pro-cognitive effects and amelioration of abnormal mood.5,9 In terms of tolerability, a relative absence of affinity to muscarinic receptors would predict a low risk for anticholinergic adverse effects, but antagonism at histamine H1 and at a1-adrenergic receptors, either alone or in combination, may cause sedation, and blockade of H1 receptors would also predict weight gain.9 Antagonism of a1-adrenergic receptors can be associated with orthostatic hypotension and neurally mediated reflex bradycardia.9
Clinical pharmacokinetics
Three open-label, randomized, phase 1 studies were conducted to assess the relative bioavailability of asenapine transdermal system vs sublingual asenapine.10 These included single- and multiple-dose studies and clinical trials that examined the effects of different application sites and ethnic groups, and the effect of external heat on medication absorption. Studies were conducted in healthy individuals, except for the multiple-dose study, which was performed in adults with schizophrenia. The AUC for asenapine transdermal system was within the range of that of equivalent doses of sublingual asenapine, but peak exposure (maximum concentration) was significantly lower. As already noted, the AUC of the asenapine patch for 3.8 mg/24 hours and 7.6 mg/24 hours corresponds to sublingual asenapine 5 mg and 10 mg twice daily, respectively. Maximum asenapine concentrations are typically reached between 12 and 24 hours, with sustained concentrations during the 24-hour wear time.3 On average, approximately 60% of the available asenapine is released from the transdermal system over 24 hours. Steady-state plasma concentrations for asenapine transdermal system were achieved approximately 72 hours after the first application and, in contrast to sublingual asenapine, the peak-trough fluctuations were small (peak-to-trough ratio is 1.5 for asenapine transdermal system compared with >3 for sublingual asenapine). Dose-proportionality at steady state was evident for asenapine transdermal system. This is in contrast to sublingual asenapine, where exposure increases 1.7-fold with a 2-fold increase in dose.4,5 Following patch removal, the apparent elimination half-life is approximately 30 hours.3 The pharmacokinetics of the patch did not vary with regards to the application site (upper arm, upper back, abdomen, or hip area), and the pharmacokinetic profile was similar across the ethnic groups that participated in the study. Direct exposure to external heat did increase both the rate and extent of absorption, so external heat sources should be avoided.3
Efficacy
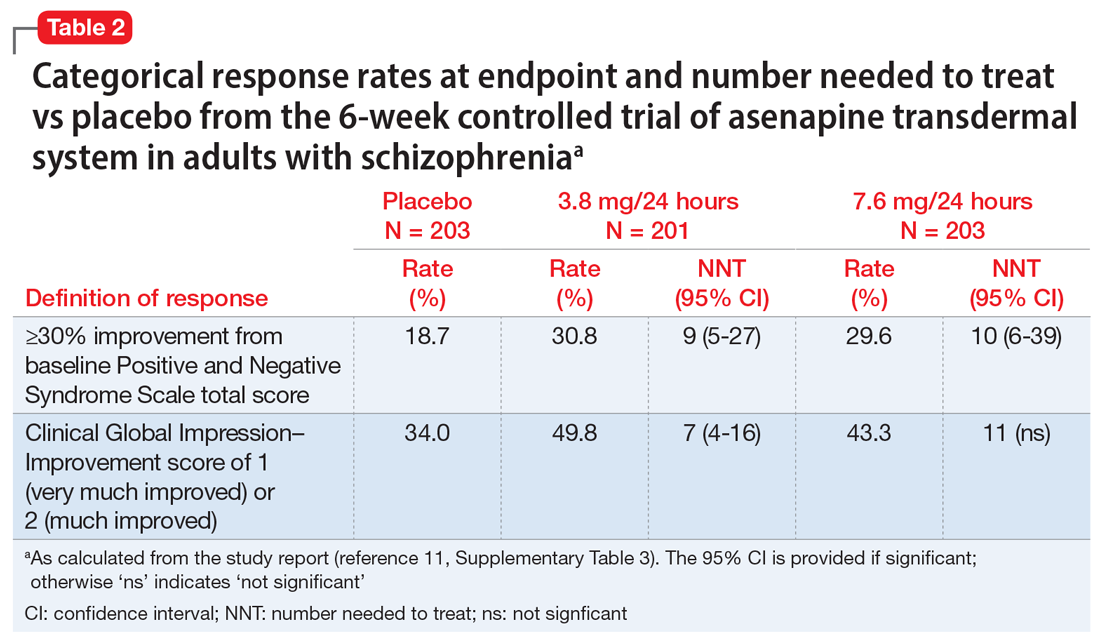
The efficacy profile for asenapine transdermal system would be expected to mirror that for sublingual asenapine.6,7 In addition to data supporting the use of asenapine as administered sublingually, a phase 3 study specifically assessed efficacy and safety of asenapine transdermal system in adults with schizophrenia.11,12 This study was conducted in the United States and 4 other countries at a total of 59 study sites, and 616 patients with acutely exacerbated schizophrenia were enrolled. After a 3- to 14-day screening/single-blind run-in washout period, participants entered a 6-week inpatient double-blind period. Randomization was 1:1:1 to asenapine transdermal system 3.8 mg/24 hours, 7.6 mg/24 hours, or a placebo patch. Each of the patch doses demonstrated significant improvement vs placebo at Week 6 for the primary (change in Positive and Negative Syndrome Scale [PANSS] total score) and key secondary (change in Clinical Global Impression-Severity of Illness) endpoints. Response at endpoint, as defined by a ≥30% improvement from baseline PANSS total score, or by a Clinical Global Impression–Improvement score of 1 (very much improved) or 2 (much improved), was also assessed. For either definition of response, both doses of asenapine transdermal system were superior to placebo, with number needed to treat (NNT) (Box) values <10 for the 3.8 mg/24 hours dose (Table 2). These effect sizes are similar to what is known about sublingual asenapine as determined in a meta-analysis performed by the manufacturer and using individual patient data.13
Box
Clinical trials produce a mountain of data that can be difficult to interpret and apply to clinical practice. When reading about studies, you may wonder:
- How large is the effect being measured?
- Is it clinically important?
- Are we reviewing a result that may be statistically significant but irrelevant for day-today patient care?
Number needed to treat (NNT) and number needed to harm (NNH)—two tools of evidence-based medicine—can help answer these questions. NNT helps us gauge effect size or clinical significance. It is different from knowing if a clinical trial result is statistically significant. NNT allows us to place a number on how often we can expect to encounter a difference between two interventions. If we see a therapeutic difference once every 100 patients (NNT of 100), the difference between the treatments is not of great concern under most circumstances. But if a difference in outcome is seen once in every 7 patients being treated with an intervention vs another (NNT of 7), the result will likely influence dayto-day practice.
How to calculate NNT (or NNH):
What is the NNT for an outcome for drug A vs drug B?
fA = frequency of outcome for drug A
fB = frequency of outcome for drug B
NNT = 1/[ fA - fB]
By convention, we round up the NNT to the next higher whole number.
For example, let’s say drugs A and B are used to treat depression, and they result in 6-week response rates of 55% and 75%, respectively. The NNT to encounter a difference between drug B and drug A in terms of responders at 6 weeks can be calculated as follows:
- Difference in response rates: .75 -.55 = .20
- NNT: 1/.20 = 5
A rule of thumb: NNT values for a medication vs placebo <10 usually denote a medication we use on a regular basis to treat patients.
a Adapted from Citrome L. Dissecting clinical trials with ‘number needed to treat.’ Current Psychiatry. 2007;6(3):66-71. Citrome L. Can you interpret confidence intervals? It’s not that difficult. Current Psychiatry. 2007;6(8):77-82. Additional information can be found in Citrome L, Ketter TA. When does a difference make a difference? Interpretation of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Int J Clin Pract. 2013;67(5):407-411 (free to access at onlinelibrary.wiley.com/doi/full/10.1111/ijcp.12142)
Overall tolerability and safety
The systemic safety and tolerability profile for asenapine transdermal system would be expected to be similar to that for sublingual asenapine, unless there are adverse events that are related to high peak plasma concentrations or large differences between peak and trough plasma concentrations.6 Nonsystemic local application site adverse events would, of course, differ between sublingual vs transdermal administration.
Continue to: Use of asenapine transdermal system...
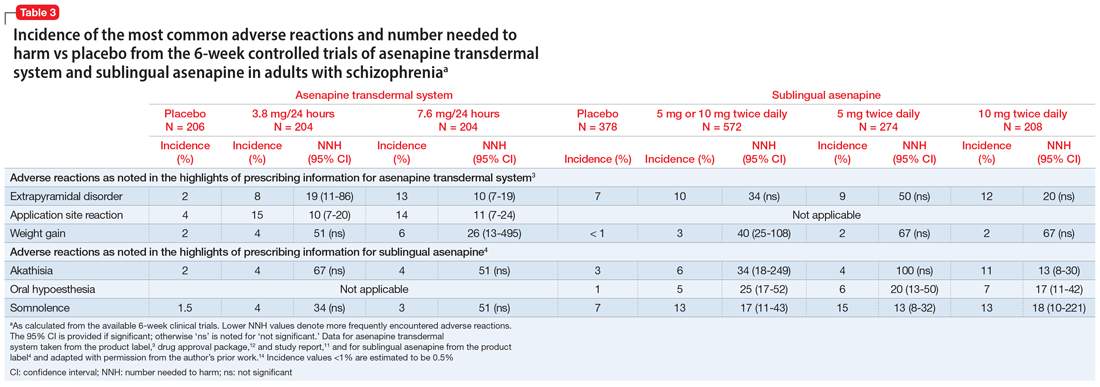
Use of asenapine transdermal system avoids the dysgeusia and oral hypoesthesia that can be observed with sublingual asenapine4,6; however, dermal effects need to be considered (see Dermal safety). The most commonly observed adverse reactions (incidence ≥5% and at least twice that for placebo) for asenapine transdermal system are extrapyramidal disorder, application site reaction, and weight gain.3 For sublingual asenapine for adults with schizophrenia, the list includes akathisia, oral hypoesthesia, and somnolence.4 These adverse events can be further described using the metric of number needed to harm (NNH) as shown in Table 3.3,4,11,12,14 Of note, extrapyramidal disorder and weight gain appear to be dose-related for asenapine transdermal system. Akathisia appears to be dose-related for sublingual asenapine but not for asenapine transdermal system. Somnolence appears to be associated with sublingual asenapine but not necessarily with asenapine transdermal system.
For sublingual asenapine, the additional indications (bipolar I disorder as acute monotherapy treatment of manic or mixed episodes in adults and pediatric patients age 10 to 17, adjunctive treatment to lithium or valproate in adults, and maintenance monotherapy treatment in adults) have varying commonly encountered adverse reactions.4 Both transdermal asenapine system and sublingual asenapine are contraindicated in patients with severe hepatic impairment (Child-Pugh C) and those with known hypersensitivity to asenapine or to any components in the formulation. Both formulations carry similar warnings in their prescribing information regarding increased mortality in older patients with dementia-related psychosis, cerebrovascular adverse reactions in older patients with dementia-related psychosis, neuroleptic malignant syndrome, tardive dyskinesia, metabolic changes, orthostatic hypotension, leukopenia (and neutropenia and agranulocytosis), QT prolongation, seizures, and potential for cognitive and motor impairment.
Adverse events leading to discontinuation of study treatment in the asenapine transdermal system pivotal trial occurred in 4.9%, 7.8%, and 6.8% of participants in the 3.8 mg/24 hour, 7.6 mg/24 hour, and placebo groups, respectively.11
Dermal safety
In the pivotal efficacy study,11 the incidence of adverse events at patch application sites was higher in the active groups vs placebo (Table 33,4,11,12,14). The most frequently reported patch application site reactions were erythema and pruritus, occurring in approximately 10% and 4% in the active treatment arms vs 1.5% and 1.9% for placebo, respectively. With the exception of 1 adverse event of severe application site erythema during Week 2 (participant received 7.6 mg/24 hour, erythema resolved without intervention, and the patient continued the study), all other patch application site events were mild or moderate in severity. Rates of discontinuation due to application site reactions or skin disorders were ≤0.5% across all groups. In the pharmacokinetic studies,10 no patches were removed because of unacceptable irritation.
Why Rx?
Asenapine transdermal system is the first antipsychotic “patch” FDA-approved for the treatment of adults with schizophrenia. Asenapine has been available since 2009 as a sublingual formulation administered twice daily. The pharmacokinetic profile of the once-daily transdermal system demonstrates dose-proportional kinetics and sustained delivery of asenapine with a low peak-to-trough plasma level ratio. Three dosage strengths (3.8, 5.7, and 7.6 mg/24 hours) are available, corresponding to blood levels attained with sublingual asenapine exposures of 10, 15, and 20 mg/d, respectively. Application sites are rotated daily and include the upper arms, upper back, abdomen, or hip. Dysgeusia and hypoesthesia of the tongue are avoided with the use of the patch, and there are no food or drink restrictions. Attention will be needed in case of dermal reactions, similar to that observed with other medication patches.
Bottom Line
The asenapine transdermal drug delivery system appears to be efficacious and reasonably well tolerated. The treatment of schizophrenia is complex and requires individualized choices in order to optimize outcomes. A patch may be the preferred formulation for selected patients, and caregivers will have the ability to visually check if the medication is being used.
Related Resource
- Hisamitsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
Drug Brand Names
Asenapine sublingual • Saphris
Asenapine transdermal system • Secuado
Lithium • Eskalith, Lithobid
Valproate • Depakote
1. Noven. US FDA approves SECUADO® (asenapine) transdermal system, the first-and-only transdermal patch for the treatment of adults with schizophrenia. October 15, 2019. Accessed January 15, 2021. https://www.noven.com/wp-content/uploads/2020/04/PR101519.pdf
2. US Food and Drug Administration. Center for Drug Evaluation and Research. Approval Package for: APPLICATION NUMBER: 212268Orig1s000. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000Approv.pdf
3. Hisam itsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
4. Allergan USA, Inc. SAPHRIS® (asenapine) sublingual tablets prescribing information. February 2017. Accessed January 15, 2021. https://media.allergan.com/actavis/actavis/media/allergan-pdf-documents/product-prescribing/Final_labeling_text_SAPHRIS-clean-02-2017.pdf
5. Citrome L. Asenapine review, part I: chemistry, receptor affinity profile, pharmacokinetics and metabolism. Expert Opin Drug Metab Toxicol. 2014;10(6):893-903.
6. Citrome L. Asenapine review, part II: clinical efficacy, safety and tolerability. Expert Opin Drug Saf. 2014;13(6):803-830.
7. Citrome L. Chapter 31: Asenapine. In: Schatzberg AF, Nemeroff CB, eds. The American Psychiatric Association Publishing Textbook of Psychopharmacology, 5th ed. American Psychiatric Association Publishing; 2017:797-808.
8. Citrome L, Zeni CM, Correll CU. Patches: established and emerging transdermal treatments in psychiatry. J Clin Psychiatry. 2019;80(4):18nr12554. doi: 10.4088/JCP.18nr12554
9. Shayegan DK, Stahl SM. Atypical antipsychotics: matching receptor profile to individual patient’s clinical profile. CNS Spectr. 2004;9(10 suppl 11):6-14.
10. Castelli M, Suzuki K, Komaroff M, et al. Pharmacokinetic profile of asenapine transdermal system HP-3070: The first antipsychotic patch in the US. Poster presented virtually at the American Society for Clinical Psychopharmacology (ASCP) 2020 Annual Meeting, May 29-30, 2020. https://www.psychiatrist.com/ascpcorner/Documents/ascp2020/3_ASCP%20Poster%20Abstracts%202020-JCP.pdf
11. Citrome L, Walling DP, Zeni CM, et al. Efficacy and safety of HP-3070, an asenapine transdermal system, in patients with schizophrenia: a phase 3, randomized, placebo-controlled study. J Clin Psychiatry. 2020;82(1):20m13602. doi: 10.4088/JCP.20m13602
12. US Food and Drug Administration. Drug Approval Package: SECAUDO. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000TOC.cfm
13. Szegedi A, Verweij P, van Duijnhoven W, et al. Meta-analyses of the efficacy of asenapine for acute schizophrenia: comparisons with placebo and other antipsychotics. J Clin Psychiatry. 2012;73(12):1533-1540.
14. Citrome L. Asenapine for schizophrenia and bipolar disorder: a review of the efficacy and safety profile for this newly approved sublingually absorbed second-generation antipsychotic. Int J Clin Pract. 2009;63(12):1762-1784.
The asenapine transdermal system is available in 3 patch sizes: 20, 30, and 40 cm2, which deliver 3.8, 5.7, and 7.6 mg/24 hours of asenapine, respectively.3 Based on the average exposure (area under the plasma concentration curve [AUC]) of asenapine, 3.8 mg/24 hours corresponds to 5 mg twice daily of sublingual asenapine, and 7.6 mg/24 hours corresponds to 10 mg twice daily of sublingual asenapine.3 The “in-between” dose strength of 5.7 mg/24 hours would correspond to exposure to a total of 15 mg/d of sublingual asenapine. The recommended starting dose for asenapine transdermal system is 3.8 mg/24 hours. The dosage may be increased to 5.7 mg/24 hours or 7.6 mg/24 hours, as needed, after 1 week. The safety of doses above 7.6 mg/24 hours has not been evaluated in clinical studies. Asenapine transdermal system is applied once daily and should be worn for 24 hours only, with only 1 patch at any time. Application sites include the upper arm, upper back, abdomen, and hip. A different application site of clean, dry, intact skin should be selected each time a new patch is applied. Although showering is permitted, the use of asenapine transdermal system during swimming or taking a bath has not been evaluated. Of note, prolonged application of heat over an asenapine transdermal system increases plasma concentrations of asenapine, and thus application of external heat sources (eg, heating pads) over the patch should be avoided.
How it works
Product labeling notes that asenapine is an atypical antipsychotic, and that its efficacy in schizophrenia could be mediated through a combination of antagonist activity at dopamine D2 and serotonin 5-HT2A receptors.3 The pharmacodynamic profile of asenapine is complex5 and receptor-binding assays performed using cloned human serotonin, norepinephrine, dopamine, histamine, and muscarinic receptors demonstrated picomolar affinity (extremely high) for 5-HT2C and 5-HT2A receptors, subnanomolar affinity (very high) for 5-HT7, 5-HT2B, 5-HT6, and D3 receptors, and nanomolar affinity (high) for D2 receptors, as well as histamine H1, D4, a1-adrenergic, a2-adrenergic, D1, 5-HT5, 5-HT1A, 5-HT1B, and histamine H2 receptors. Activity of asenapine is that of antagonism at these receptors. Asenapine has no appreciable affinity for muscarinic cholinergic receptors.
The asenapine receptor-binding “fingerprint” differs from that of other antipsychotics. Some of these receptor affinities are of special interest in terms of potential efficacy for pro-cognitive effects and amelioration of abnormal mood.5,9 In terms of tolerability, a relative absence of affinity to muscarinic receptors would predict a low risk for anticholinergic adverse effects, but antagonism at histamine H1 and at a1-adrenergic receptors, either alone or in combination, may cause sedation, and blockade of H1 receptors would also predict weight gain.9 Antagonism of a1-adrenergic receptors can be associated with orthostatic hypotension and neurally mediated reflex bradycardia.9
Clinical pharmacokinetics
Three open-label, randomized, phase 1 studies were conducted to assess the relative bioavailability of asenapine transdermal system vs sublingual asenapine.10 These included single- and multiple-dose studies and clinical trials that examined the effects of different application sites and ethnic groups, and the effect of external heat on medication absorption. Studies were conducted in healthy individuals, except for the multiple-dose study, which was performed in adults with schizophrenia. The AUC for asenapine transdermal system was within the range of that of equivalent doses of sublingual asenapine, but peak exposure (maximum concentration) was significantly lower. As already noted, the AUC of the asenapine patch for 3.8 mg/24 hours and 7.6 mg/24 hours corresponds to sublingual asenapine 5 mg and 10 mg twice daily, respectively. Maximum asenapine concentrations are typically reached between 12 and 24 hours, with sustained concentrations during the 24-hour wear time.3 On average, approximately 60% of the available asenapine is released from the transdermal system over 24 hours. Steady-state plasma concentrations for asenapine transdermal system were achieved approximately 72 hours after the first application and, in contrast to sublingual asenapine, the peak-trough fluctuations were small (peak-to-trough ratio is 1.5 for asenapine transdermal system compared with >3 for sublingual asenapine). Dose-proportionality at steady state was evident for asenapine transdermal system. This is in contrast to sublingual asenapine, where exposure increases 1.7-fold with a 2-fold increase in dose.4,5 Following patch removal, the apparent elimination half-life is approximately 30 hours.3 The pharmacokinetics of the patch did not vary with regards to the application site (upper arm, upper back, abdomen, or hip area), and the pharmacokinetic profile was similar across the ethnic groups that participated in the study. Direct exposure to external heat did increase both the rate and extent of absorption, so external heat sources should be avoided.3
Efficacy
The efficacy profile for asenapine transdermal system would be expected to mirror that for sublingual asenapine.6,7 In addition to data supporting the use of asenapine as administered sublingually, a phase 3 study specifically assessed efficacy and safety of asenapine transdermal system in adults with schizophrenia.11,12 This study was conducted in the United States and 4 other countries at a total of 59 study sites, and 616 patients with acutely exacerbated schizophrenia were enrolled. After a 3- to 14-day screening/single-blind run-in washout period, participants entered a 6-week inpatient double-blind period. Randomization was 1:1:1 to asenapine transdermal system 3.8 mg/24 hours, 7.6 mg/24 hours, or a placebo patch. Each of the patch doses demonstrated significant improvement vs placebo at Week 6 for the primary (change in Positive and Negative Syndrome Scale [PANSS] total score) and key secondary (change in Clinical Global Impression-Severity of Illness) endpoints. Response at endpoint, as defined by a ≥30% improvement from baseline PANSS total score, or by a Clinical Global Impression–Improvement score of 1 (very much improved) or 2 (much improved), was also assessed. For either definition of response, both doses of asenapine transdermal system were superior to placebo, with number needed to treat (NNT) (Box) values <10 for the 3.8 mg/24 hours dose (Table 2). These effect sizes are similar to what is known about sublingual asenapine as determined in a meta-analysis performed by the manufacturer and using individual patient data.13
Box
Clinical trials produce a mountain of data that can be difficult to interpret and apply to clinical practice. When reading about studies, you may wonder:
- How large is the effect being measured?
- Is it clinically important?
- Are we reviewing a result that may be statistically significant but irrelevant for day-today patient care?
Number needed to treat (NNT) and number needed to harm (NNH)—two tools of evidence-based medicine—can help answer these questions. NNT helps us gauge effect size or clinical significance. It is different from knowing if a clinical trial result is statistically significant. NNT allows us to place a number on how often we can expect to encounter a difference between two interventions. If we see a therapeutic difference once every 100 patients (NNT of 100), the difference between the treatments is not of great concern under most circumstances. But if a difference in outcome is seen once in every 7 patients being treated with an intervention vs another (NNT of 7), the result will likely influence dayto-day practice.
How to calculate NNT (or NNH):
What is the NNT for an outcome for drug A vs drug B?
fA = frequency of outcome for drug A
fB = frequency of outcome for drug B
NNT = 1/[ fA - fB]
By convention, we round up the NNT to the next higher whole number.
For example, let’s say drugs A and B are used to treat depression, and they result in 6-week response rates of 55% and 75%, respectively. The NNT to encounter a difference between drug B and drug A in terms of responders at 6 weeks can be calculated as follows:
- Difference in response rates: .75 -.55 = .20
- NNT: 1/.20 = 5
A rule of thumb: NNT values for a medication vs placebo <10 usually denote a medication we use on a regular basis to treat patients.
a Adapted from Citrome L. Dissecting clinical trials with ‘number needed to treat.’ Current Psychiatry. 2007;6(3):66-71. Citrome L. Can you interpret confidence intervals? It’s not that difficult. Current Psychiatry. 2007;6(8):77-82. Additional information can be found in Citrome L, Ketter TA. When does a difference make a difference? Interpretation of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Int J Clin Pract. 2013;67(5):407-411 (free to access at onlinelibrary.wiley.com/doi/full/10.1111/ijcp.12142)
Overall tolerability and safety
The systemic safety and tolerability profile for asenapine transdermal system would be expected to be similar to that for sublingual asenapine, unless there are adverse events that are related to high peak plasma concentrations or large differences between peak and trough plasma concentrations.6 Nonsystemic local application site adverse events would, of course, differ between sublingual vs transdermal administration.

Continue to: Use of asenapine transdermal system...
Use of asenapine transdermal system avoids the dysgeusia and oral hypoesthesia that can be observed with sublingual asenapine4,6; however, dermal effects need to be considered (see Dermal safety). The most commonly observed adverse reactions (incidence ≥5% and at least twice that for placebo) for asenapine transdermal system are extrapyramidal disorder, application site reaction, and weight gain.3 For sublingual asenapine for adults with schizophrenia, the list includes akathisia, oral hypoesthesia, and somnolence.4 These adverse events can be further described using the metric of number needed to harm (NNH) as shown in Table 3.3,4,11,12,14 Of note, extrapyramidal disorder and weight gain appear to be dose-related for asenapine transdermal system. Akathisia appears to be dose-related for sublingual asenapine but not for asenapine transdermal system. Somnolence appears to be associated with sublingual asenapine but not necessarily with asenapine transdermal system.

For sublingual asenapine, the additional indications (bipolar I disorder as acute monotherapy treatment of manic or mixed episodes in adults and pediatric patients age 10 to 17, adjunctive treatment to lithium or valproate in adults, and maintenance monotherapy treatment in adults) have varying commonly encountered adverse reactions.4 Both transdermal asenapine system and sublingual asenapine are contraindicated in patients with severe hepatic impairment (Child-Pugh C) and those with known hypersensitivity to asenapine or to any components in the formulation. Both formulations carry similar warnings in their prescribing information regarding increased mortality in older patients with dementia-related psychosis, cerebrovascular adverse reactions in older patients with dementia-related psychosis, neuroleptic malignant syndrome, tardive dyskinesia, metabolic changes, orthostatic hypotension, leukopenia (and neutropenia and agranulocytosis), QT prolongation, seizures, and potential for cognitive and motor impairment.
Adverse events leading to discontinuation of study treatment in the asenapine transdermal system pivotal trial occurred in 4.9%, 7.8%, and 6.8% of participants in the 3.8 mg/24 hour, 7.6 mg/24 hour, and placebo groups, respectively.11
Dermal safety
In the pivotal efficacy study,11 the incidence of adverse events at patch application sites was higher in the active groups vs placebo (Table 33,4,11,12,14). The most frequently reported patch application site reactions were erythema and pruritus, occurring in approximately 10% and 4% in the active treatment arms vs 1.5% and 1.9% for placebo, respectively. With the exception of 1 adverse event of severe application site erythema during Week 2 (participant received 7.6 mg/24 hour, erythema resolved without intervention, and the patient continued the study), all other patch application site events were mild or moderate in severity. Rates of discontinuation due to application site reactions or skin disorders were ≤0.5% across all groups. In the pharmacokinetic studies,10 no patches were removed because of unacceptable irritation.
Why Rx?
Asenapine transdermal system is the first antipsychotic “patch” FDA-approved for the treatment of adults with schizophrenia. Asenapine has been available since 2009 as a sublingual formulation administered twice daily. The pharmacokinetic profile of the once-daily transdermal system demonstrates dose-proportional kinetics and sustained delivery of asenapine with a low peak-to-trough plasma level ratio. Three dosage strengths (3.8, 5.7, and 7.6 mg/24 hours) are available, corresponding to blood levels attained with sublingual asenapine exposures of 10, 15, and 20 mg/d, respectively. Application sites are rotated daily and include the upper arms, upper back, abdomen, or hip. Dysgeusia and hypoesthesia of the tongue are avoided with the use of the patch, and there are no food or drink restrictions. Attention will be needed in case of dermal reactions, similar to that observed with other medication patches.
Bottom Line
The asenapine transdermal drug delivery system appears to be efficacious and reasonably well tolerated. The treatment of schizophrenia is complex and requires individualized choices in order to optimize outcomes. A patch may be the preferred formulation for selected patients, and caregivers will have the ability to visually check if the medication is being used.
Related Resource
- Hisamitsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
Drug Brand Names
Asenapine sublingual • Saphris
Asenapine transdermal system • Secuado
Lithium • Eskalith, Lithobid
Valproate • Depakote
The asenapine transdermal system is available in 3 patch sizes: 20, 30, and 40 cm2, which deliver 3.8, 5.7, and 7.6 mg/24 hours of asenapine, respectively.3 Based on the average exposure (area under the plasma concentration curve [AUC]) of asenapine, 3.8 mg/24 hours corresponds to 5 mg twice daily of sublingual asenapine, and 7.6 mg/24 hours corresponds to 10 mg twice daily of sublingual asenapine.3 The “in-between” dose strength of 5.7 mg/24 hours would correspond to exposure to a total of 15 mg/d of sublingual asenapine. The recommended starting dose for asenapine transdermal system is 3.8 mg/24 hours. The dosage may be increased to 5.7 mg/24 hours or 7.6 mg/24 hours, as needed, after 1 week. The safety of doses above 7.6 mg/24 hours has not been evaluated in clinical studies. Asenapine transdermal system is applied once daily and should be worn for 24 hours only, with only 1 patch at any time. Application sites include the upper arm, upper back, abdomen, and hip. A different application site of clean, dry, intact skin should be selected each time a new patch is applied. Although showering is permitted, the use of asenapine transdermal system during swimming or taking a bath has not been evaluated. Of note, prolonged application of heat over an asenapine transdermal system increases plasma concentrations of asenapine, and thus application of external heat sources (eg, heating pads) over the patch should be avoided.
How it works
Product labeling notes that asenapine is an atypical antipsychotic, and that its efficacy in schizophrenia could be mediated through a combination of antagonist activity at dopamine D2 and serotonin 5-HT2A receptors.3 The pharmacodynamic profile of asenapine is complex5 and receptor-binding assays performed using cloned human serotonin, norepinephrine, dopamine, histamine, and muscarinic receptors demonstrated picomolar affinity (extremely high) for 5-HT2C and 5-HT2A receptors, subnanomolar affinity (very high) for 5-HT7, 5-HT2B, 5-HT6, and D3 receptors, and nanomolar affinity (high) for D2 receptors, as well as histamine H1, D4, a1-adrenergic, a2-adrenergic, D1, 5-HT5, 5-HT1A, 5-HT1B, and histamine H2 receptors. Activity of asenapine is that of antagonism at these receptors. Asenapine has no appreciable affinity for muscarinic cholinergic receptors.
The asenapine receptor-binding “fingerprint” differs from that of other antipsychotics. Some of these receptor affinities are of special interest in terms of potential efficacy for pro-cognitive effects and amelioration of abnormal mood.5,9 In terms of tolerability, a relative absence of affinity to muscarinic receptors would predict a low risk for anticholinergic adverse effects, but antagonism at histamine H1 and at a1-adrenergic receptors, either alone or in combination, may cause sedation, and blockade of H1 receptors would also predict weight gain.9 Antagonism of a1-adrenergic receptors can be associated with orthostatic hypotension and neurally mediated reflex bradycardia.9
Clinical pharmacokinetics
Three open-label, randomized, phase 1 studies were conducted to assess the relative bioavailability of asenapine transdermal system vs sublingual asenapine.10 These included single- and multiple-dose studies and clinical trials that examined the effects of different application sites and ethnic groups, and the effect of external heat on medication absorption. Studies were conducted in healthy individuals, except for the multiple-dose study, which was performed in adults with schizophrenia. The AUC for asenapine transdermal system was within the range of that of equivalent doses of sublingual asenapine, but peak exposure (maximum concentration) was significantly lower. As already noted, the AUC of the asenapine patch for 3.8 mg/24 hours and 7.6 mg/24 hours corresponds to sublingual asenapine 5 mg and 10 mg twice daily, respectively. Maximum asenapine concentrations are typically reached between 12 and 24 hours, with sustained concentrations during the 24-hour wear time.3 On average, approximately 60% of the available asenapine is released from the transdermal system over 24 hours. Steady-state plasma concentrations for asenapine transdermal system were achieved approximately 72 hours after the first application and, in contrast to sublingual asenapine, the peak-trough fluctuations were small (peak-to-trough ratio is 1.5 for asenapine transdermal system compared with >3 for sublingual asenapine). Dose-proportionality at steady state was evident for asenapine transdermal system. This is in contrast to sublingual asenapine, where exposure increases 1.7-fold with a 2-fold increase in dose.4,5 Following patch removal, the apparent elimination half-life is approximately 30 hours.3 The pharmacokinetics of the patch did not vary with regards to the application site (upper arm, upper back, abdomen, or hip area), and the pharmacokinetic profile was similar across the ethnic groups that participated in the study. Direct exposure to external heat did increase both the rate and extent of absorption, so external heat sources should be avoided.3
Efficacy
The efficacy profile for asenapine transdermal system would be expected to mirror that for sublingual asenapine.6,7 In addition to data supporting the use of asenapine as administered sublingually, a phase 3 study specifically assessed efficacy and safety of asenapine transdermal system in adults with schizophrenia.11,12 This study was conducted in the United States and 4 other countries at a total of 59 study sites, and 616 patients with acutely exacerbated schizophrenia were enrolled. After a 3- to 14-day screening/single-blind run-in washout period, participants entered a 6-week inpatient double-blind period. Randomization was 1:1:1 to asenapine transdermal system 3.8 mg/24 hours, 7.6 mg/24 hours, or a placebo patch. Each of the patch doses demonstrated significant improvement vs placebo at Week 6 for the primary (change in Positive and Negative Syndrome Scale [PANSS] total score) and key secondary (change in Clinical Global Impression-Severity of Illness) endpoints. Response at endpoint, as defined by a ≥30% improvement from baseline PANSS total score, or by a Clinical Global Impression–Improvement score of 1 (very much improved) or 2 (much improved), was also assessed. For either definition of response, both doses of asenapine transdermal system were superior to placebo, with number needed to treat (NNT) (Box) values <10 for the 3.8 mg/24 hours dose (Table 2). These effect sizes are similar to what is known about sublingual asenapine as determined in a meta-analysis performed by the manufacturer and using individual patient data.13
Box
Clinical trials produce a mountain of data that can be difficult to interpret and apply to clinical practice. When reading about studies, you may wonder:
- How large is the effect being measured?
- Is it clinically important?
- Are we reviewing a result that may be statistically significant but irrelevant for day-today patient care?
Number needed to treat (NNT) and number needed to harm (NNH)—two tools of evidence-based medicine—can help answer these questions. NNT helps us gauge effect size or clinical significance. It is different from knowing if a clinical trial result is statistically significant. NNT allows us to place a number on how often we can expect to encounter a difference between two interventions. If we see a therapeutic difference once every 100 patients (NNT of 100), the difference between the treatments is not of great concern under most circumstances. But if a difference in outcome is seen once in every 7 patients being treated with an intervention vs another (NNT of 7), the result will likely influence dayto-day practice.
How to calculate NNT (or NNH):
What is the NNT for an outcome for drug A vs drug B?
fA = frequency of outcome for drug A
fB = frequency of outcome for drug B
NNT = 1/[ fA - fB]
By convention, we round up the NNT to the next higher whole number.
For example, let’s say drugs A and B are used to treat depression, and they result in 6-week response rates of 55% and 75%, respectively. The NNT to encounter a difference between drug B and drug A in terms of responders at 6 weeks can be calculated as follows:
- Difference in response rates: .75 -.55 = .20
- NNT: 1/.20 = 5
A rule of thumb: NNT values for a medication vs placebo <10 usually denote a medication we use on a regular basis to treat patients.
a Adapted from Citrome L. Dissecting clinical trials with ‘number needed to treat.’ Current Psychiatry. 2007;6(3):66-71. Citrome L. Can you interpret confidence intervals? It’s not that difficult. Current Psychiatry. 2007;6(8):77-82. Additional information can be found in Citrome L, Ketter TA. When does a difference make a difference? Interpretation of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Int J Clin Pract. 2013;67(5):407-411 (free to access at onlinelibrary.wiley.com/doi/full/10.1111/ijcp.12142)
Overall tolerability and safety
The systemic safety and tolerability profile for asenapine transdermal system would be expected to be similar to that for sublingual asenapine, unless there are adverse events that are related to high peak plasma concentrations or large differences between peak and trough plasma concentrations.6 Nonsystemic local application site adverse events would, of course, differ between sublingual vs transdermal administration.

Continue to: Use of asenapine transdermal system...
Use of asenapine transdermal system avoids the dysgeusia and oral hypoesthesia that can be observed with sublingual asenapine4,6; however, dermal effects need to be considered (see Dermal safety). The most commonly observed adverse reactions (incidence ≥5% and at least twice that for placebo) for asenapine transdermal system are extrapyramidal disorder, application site reaction, and weight gain.3 For sublingual asenapine for adults with schizophrenia, the list includes akathisia, oral hypoesthesia, and somnolence.4 These adverse events can be further described using the metric of number needed to harm (NNH) as shown in Table 3.3,4,11,12,14 Of note, extrapyramidal disorder and weight gain appear to be dose-related for asenapine transdermal system. Akathisia appears to be dose-related for sublingual asenapine but not for asenapine transdermal system. Somnolence appears to be associated with sublingual asenapine but not necessarily with asenapine transdermal system.

For sublingual asenapine, the additional indications (bipolar I disorder as acute monotherapy treatment of manic or mixed episodes in adults and pediatric patients age 10 to 17, adjunctive treatment to lithium or valproate in adults, and maintenance monotherapy treatment in adults) have varying commonly encountered adverse reactions.4 Both transdermal asenapine system and sublingual asenapine are contraindicated in patients with severe hepatic impairment (Child-Pugh C) and those with known hypersensitivity to asenapine or to any components in the formulation. Both formulations carry similar warnings in their prescribing information regarding increased mortality in older patients with dementia-related psychosis, cerebrovascular adverse reactions in older patients with dementia-related psychosis, neuroleptic malignant syndrome, tardive dyskinesia, metabolic changes, orthostatic hypotension, leukopenia (and neutropenia and agranulocytosis), QT prolongation, seizures, and potential for cognitive and motor impairment.
Adverse events leading to discontinuation of study treatment in the asenapine transdermal system pivotal trial occurred in 4.9%, 7.8%, and 6.8% of participants in the 3.8 mg/24 hour, 7.6 mg/24 hour, and placebo groups, respectively.11
Dermal safety
In the pivotal efficacy study,11 the incidence of adverse events at patch application sites was higher in the active groups vs placebo (Table 33,4,11,12,14). The most frequently reported patch application site reactions were erythema and pruritus, occurring in approximately 10% and 4% in the active treatment arms vs 1.5% and 1.9% for placebo, respectively. With the exception of 1 adverse event of severe application site erythema during Week 2 (participant received 7.6 mg/24 hour, erythema resolved without intervention, and the patient continued the study), all other patch application site events were mild or moderate in severity. Rates of discontinuation due to application site reactions or skin disorders were ≤0.5% across all groups. In the pharmacokinetic studies,10 no patches were removed because of unacceptable irritation.
Why Rx?
Asenapine transdermal system is the first antipsychotic “patch” FDA-approved for the treatment of adults with schizophrenia. Asenapine has been available since 2009 as a sublingual formulation administered twice daily. The pharmacokinetic profile of the once-daily transdermal system demonstrates dose-proportional kinetics and sustained delivery of asenapine with a low peak-to-trough plasma level ratio. Three dosage strengths (3.8, 5.7, and 7.6 mg/24 hours) are available, corresponding to blood levels attained with sublingual asenapine exposures of 10, 15, and 20 mg/d, respectively. Application sites are rotated daily and include the upper arms, upper back, abdomen, or hip. Dysgeusia and hypoesthesia of the tongue are avoided with the use of the patch, and there are no food or drink restrictions. Attention will be needed in case of dermal reactions, similar to that observed with other medication patches.
Bottom Line
The asenapine transdermal drug delivery system appears to be efficacious and reasonably well tolerated. The treatment of schizophrenia is complex and requires individualized choices in order to optimize outcomes. A patch may be the preferred formulation for selected patients, and caregivers will have the ability to visually check if the medication is being used.
Related Resource
- Hisamitsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
Drug Brand Names
Asenapine sublingual • Saphris
Asenapine transdermal system • Secuado
Lithium • Eskalith, Lithobid
Valproate • Depakote
1. Noven. US FDA approves SECUADO® (asenapine) transdermal system, the first-and-only transdermal patch for the treatment of adults with schizophrenia. October 15, 2019. Accessed January 15, 2021. https://www.noven.com/wp-content/uploads/2020/04/PR101519.pdf
2. US Food and Drug Administration. Center for Drug Evaluation and Research. Approval Package for: APPLICATION NUMBER: 212268Orig1s000. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000Approv.pdf
3. Hisam itsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
4. Allergan USA, Inc. SAPHRIS® (asenapine) sublingual tablets prescribing information. February 2017. Accessed January 15, 2021. https://media.allergan.com/actavis/actavis/media/allergan-pdf-documents/product-prescribing/Final_labeling_text_SAPHRIS-clean-02-2017.pdf
5. Citrome L. Asenapine review, part I: chemistry, receptor affinity profile, pharmacokinetics and metabolism. Expert Opin Drug Metab Toxicol. 2014;10(6):893-903.
6. Citrome L. Asenapine review, part II: clinical efficacy, safety and tolerability. Expert Opin Drug Saf. 2014;13(6):803-830.
7. Citrome L. Chapter 31: Asenapine. In: Schatzberg AF, Nemeroff CB, eds. The American Psychiatric Association Publishing Textbook of Psychopharmacology, 5th ed. American Psychiatric Association Publishing; 2017:797-808.
8. Citrome L, Zeni CM, Correll CU. Patches: established and emerging transdermal treatments in psychiatry. J Clin Psychiatry. 2019;80(4):18nr12554. doi: 10.4088/JCP.18nr12554
9. Shayegan DK, Stahl SM. Atypical antipsychotics: matching receptor profile to individual patient’s clinical profile. CNS Spectr. 2004;9(10 suppl 11):6-14.
10. Castelli M, Suzuki K, Komaroff M, et al. Pharmacokinetic profile of asenapine transdermal system HP-3070: The first antipsychotic patch in the US. Poster presented virtually at the American Society for Clinical Psychopharmacology (ASCP) 2020 Annual Meeting, May 29-30, 2020. https://www.psychiatrist.com/ascpcorner/Documents/ascp2020/3_ASCP%20Poster%20Abstracts%202020-JCP.pdf
11. Citrome L, Walling DP, Zeni CM, et al. Efficacy and safety of HP-3070, an asenapine transdermal system, in patients with schizophrenia: a phase 3, randomized, placebo-controlled study. J Clin Psychiatry. 2020;82(1):20m13602. doi: 10.4088/JCP.20m13602
12. US Food and Drug Administration. Drug Approval Package: SECAUDO. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000TOC.cfm
13. Szegedi A, Verweij P, van Duijnhoven W, et al. Meta-analyses of the efficacy of asenapine for acute schizophrenia: comparisons with placebo and other antipsychotics. J Clin Psychiatry. 2012;73(12):1533-1540.
14. Citrome L. Asenapine for schizophrenia and bipolar disorder: a review of the efficacy and safety profile for this newly approved sublingually absorbed second-generation antipsychotic. Int J Clin Pract. 2009;63(12):1762-1784.
1. Noven. US FDA approves SECUADO® (asenapine) transdermal system, the first-and-only transdermal patch for the treatment of adults with schizophrenia. October 15, 2019. Accessed January 15, 2021. https://www.noven.com/wp-content/uploads/2020/04/PR101519.pdf
2. US Food and Drug Administration. Center for Drug Evaluation and Research. Approval Package for: APPLICATION NUMBER: 212268Orig1s000. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000Approv.pdf
3. Hisam itsu Pharmaceutical Co., Inc. SECUADO® (asenapine) transdermal system prescribing information. October 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212268s000lbl.pdf
4. Allergan USA, Inc. SAPHRIS® (asenapine) sublingual tablets prescribing information. February 2017. Accessed January 15, 2021. https://media.allergan.com/actavis/actavis/media/allergan-pdf-documents/product-prescribing/Final_labeling_text_SAPHRIS-clean-02-2017.pdf
5. Citrome L. Asenapine review, part I: chemistry, receptor affinity profile, pharmacokinetics and metabolism. Expert Opin Drug Metab Toxicol. 2014;10(6):893-903.
6. Citrome L. Asenapine review, part II: clinical efficacy, safety and tolerability. Expert Opin Drug Saf. 2014;13(6):803-830.
7. Citrome L. Chapter 31: Asenapine. In: Schatzberg AF, Nemeroff CB, eds. The American Psychiatric Association Publishing Textbook of Psychopharmacology, 5th ed. American Psychiatric Association Publishing; 2017:797-808.
8. Citrome L, Zeni CM, Correll CU. Patches: established and emerging transdermal treatments in psychiatry. J Clin Psychiatry. 2019;80(4):18nr12554. doi: 10.4088/JCP.18nr12554
9. Shayegan DK, Stahl SM. Atypical antipsychotics: matching receptor profile to individual patient’s clinical profile. CNS Spectr. 2004;9(10 suppl 11):6-14.
10. Castelli M, Suzuki K, Komaroff M, et al. Pharmacokinetic profile of asenapine transdermal system HP-3070: The first antipsychotic patch in the US. Poster presented virtually at the American Society for Clinical Psychopharmacology (ASCP) 2020 Annual Meeting, May 29-30, 2020. https://www.psychiatrist.com/ascpcorner/Documents/ascp2020/3_ASCP%20Poster%20Abstracts%202020-JCP.pdf
11. Citrome L, Walling DP, Zeni CM, et al. Efficacy and safety of HP-3070, an asenapine transdermal system, in patients with schizophrenia: a phase 3, randomized, placebo-controlled study. J Clin Psychiatry. 2020;82(1):20m13602. doi: 10.4088/JCP.20m13602
12. US Food and Drug Administration. Drug Approval Package: SECAUDO. October 11, 2019. Accessed January 15, 2021. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212268Orig1s000TOC.cfm
13. Szegedi A, Verweij P, van Duijnhoven W, et al. Meta-analyses of the efficacy of asenapine for acute schizophrenia: comparisons with placebo and other antipsychotics. J Clin Psychiatry. 2012;73(12):1533-1540.
14. Citrome L. Asenapine for schizophrenia and bipolar disorder: a review of the efficacy and safety profile for this newly approved sublingually absorbed second-generation antipsychotic. Int J Clin Pract. 2009;63(12):1762-1784.
Elaborate hallucinations, but is it a psychotic disorder?
CASE Visual, auditory, and tactile hallucinations
Mr. B, age 93, is brought to the emergency department by his son after experiencing hallucinations where he reportedly saw and heard individuals in his home. In frustration, Mr. B wielded a knife because he “wanted them to go away.”
Mr. B and his son report that the hallucinations had begun 2 years ago, without prior trauma, medication changes, changes in social situation, or other apparent precipitating events. The hallucinations “come and go,” without preceding symptoms, but have recurring content involving a friendly man named “Harry,” people coming out of the television, 2 children playing, and water covering the floor. Mr. B acknowledges these are hallucinations and had not felt threatened by them until recently, when he wielded the knife. He often tries to talk to them, but they do not reply.
Mr. B also reports intermittent auditory hallucinations including voices at home (non-command) and papers rustling. He also describes tactile hallucinations, where he says he can feel Harry and others prodding him, knocking things out of his hands, or splashing him with water.
Mr. B is admitted to the hospital because he is a danger to himself and others. While on the inpatient unit, Mr. B is pleasant with staff, and eats and sleeps normally; however, he continues to have hallucinations of Harry. Mr. B reports seeing Harry in the hall, and says that Harry pulls out Mr. B’s earpiece and steals his fork. Mr. B also reports hearing a sound “like a bee buzzing.” Mr. B is started on risperidone, 1 mg nightly, for a presumed psychotic disorder.
HISTORY Independent and in good health
Mr. B lives alone and is independent in his activities of daily living. He spends his days at home, often visited by his children, who bring him groceries and other necessities.
Mr. B takes no medications, and has no history of psychiatric treatment; psychotic, manic, or depressive episodes; posttraumatic stress disorder; obsessive-compulsive disorder; or recent emotional stress. His medical history includes chronic progressive hearing loss, which is managed with hearing aids; macular degeneration; and prior bilateral cataract surgeries.
EVALUATION Mental status exam and objective findings
During his evaluation, Mr. B appears well-nourished, and wears glasses and hearing aids. During the interview, he is euthymic with appropriately reactive affect. He is talkative but redirectable, with a goal-directed thought process. Mr. B does not appear to be internally preoccupied. His hearing is impaired, and he often requires questions to be repeated loudly. He is oriented to person, place, and time. There are no signs of delusions, paranoia, thought blocking, thought broadcasting/insertion, or referential thinking. He denies depressed mood, anhedonia, fatigue, sleep changes, or manic symptoms. He denies the occurrence of auditory or visual hallucinations during the evaluation.
Continue to: A neurologic exam shows...
A neurologic exam shows impaired hearing bilaterally and impaired visual acuity. Even with glasses, both eyes have acuity only to finger counting. All other cranial nerves are normal, and Mr. B’s strength, sensation, and cerebellar function are all intact, without rigidity, numbness, or tingling. His gait is steady without a walker, with symmetric arm swing and slight dragging of his feet. His vitals are stable, with normal orthostatic pressures.
Other objective data include a score of 24/30 on the Mini-Mental State Examination, notable for deficits in visuospatial orientation, attention, and calculation, with language and copying limited by poor vision. Mr. B scores 16/22 on the Montreal Cognitive Assessment (MoCA)-Blind (adapted version of MoCA), which is equivalent to a 22/30 on the MoCA, indicating some mild cognitive impairment; however, this modified test is still limited by his poor hearing. His serum and urine laboratory workup show no liver, kidney, metabolic, or electrolyte abnormalities, no sign of infection, negative urine drug screen, and normal B12 and thyroid-stimulating hormone levels. He undergoes a brain MRI, which shows chronic microvascular ischemic change, without mass lesions, infarction, or other pathology.
[polldaddy:10729178]
The authors’ observations
Given Mr. B’s presentation, we ruled out a primary psychotic disorder. He had no psychiatric history, with organized thought, a reactive affect, and no delusions, paranoia, or other psychotic symptoms, all pointing against psychosis. His brain MRI showed no malignancy or other lesions. He had no substance use history to suggest intoxication/withdrawal. His intact attention and orientation did not suggest delirium, and his serum and urine studies were all negative. Although his blaming Harry for knocking things out of his hands could suggest confabulation, Mr. B had no other signs of Korsakoff syndrome, such as ataxia, general confusion, or malnourishment.
We also considered early dementia. There was suspicion for Lewy body dementia given Mr. B’s prominent fluctuating visual hallucinations; however, he displayed no other signs of the disorder, such as parkinsonism, dysautonomia, or sensitivity to the antipsychotic (risperidone 1 mg nightly) started on admission. The presence of 1 core feature of Lewy body dementia—visual hallucinations—indicated a possible, but not probable, diagnosis. Additionally, Mr. B did not have the characteristic features of other types of dementia, such as the stepwise progression of vascular dementia, the behavioral disinhibition of frontotemporal dementia, or the insidious forgetfulness, confusion, language problems, or paranoia that may appear in Alzheimer’s disease. Remarkably, he had a relatively normal brain MRI for his age, given chronic microvascular ischemic changes, and cognitive testing that indicated only mild impairment further pointed against a dementia process.
Charles Bonnet syndrome
Based on Mr. B’s severe vision loss and history of ocular surgeries, we diagnosed him with CBS, described as visual hallucinations in the presence of impaired vision. Charles Bonnet syndrome has been observed in several disorders that affect vision, most commonly macular degeneration, diabetic retinopathy, and glaucoma, with an estimated prevalence of 11% to 39% in older patients with ocular disease.1,2 Visual hallucinations in CBS occur due to ocular disease, likely resulting from changes in afferent sensory input to visual cortical regions of the brain. Table 13 outlines the features of visual hallucinations in patients with CBS. The subsequent disinhibition and spontaneous firing of the visual association cortices leads to the “release hallucinations” of the syndrome.4 The disorder is thought to be significantly underdiagnosed—in a survey of patients with CBS, only 15% had reported their visual hallucinations to a physician.5

Continue to: Mr. B's symptoms...
Mr. B’s symptoms are atypical for CBS, but they fit the diagnosis when considering the entire clinical picture. While hallucinations in CBS are more often simple shapes, complex hallucinations including people and scenes have been noted in several instances.6
Similar to Mr. B’s case, patients with CBS can have recurring figures in their hallucinations, and the images may even move across the visual field.1 Patients with CBS also frequently recognize that their hallucinations are not real, and may or may not be distressed by them.4 Patients with CBS often have hallucinations multiple times daily, lasting from a few seconds to many minutes,7 consistent with Mr. B’s temporary symptoms.
Although auditory and tactile hallucinations are typically not included in CBS, they can also be explained by Mr. B’s significant sensory impairment. Severe hearing impairment in geriatric adults has been associated with auditory hallucinations8; in 1 survey, half of these hallucinations consisted of voices.9 In contrast, tactile hallucinations are not described in sensory deprivation literature. However, in the context of Mr. B’s severe comorbid hearing and vision loss, we propose that these hallucinations reflect his interpretation of sensory events around him, and their integration into his extensive hallucination framework. In other words, Harry poking him and causing him to drop things may be Mr. B’s way of rationalizing events that he has trouble perceiving entirely, or his mild forgetfulness. Mr. B’s social isolation is another factor that may worsen his sensory deprivation and contribute to his extensive hallucinations.10 Additionally, his mild cognitive deficits on testing with chronic microvascular changes on the MRI may suggest a mild vascular-related dementia process, which could also exacerbate his hallucinations. While classic CBS occurs without cognitive impairment, dementia can often co-occur with CBS.11
TREATMENT No significant improvement with medications
During his inpatient stay, Mr. B is treated with risperidone, 1 mg nightly, and is also started on donepezil, 5 mg/d, to treat a possible comorbid dementia. However, he continues to hallucinate without significant improvement.
[polldaddy:10729181]
The authors’ observations
There is no definitive treatment for CBS, and while the hallucinations may spontaneously resolve, per case reports, this typically occurs only as visual loss progresses to total blindness.12 However, many patients can have the hallucinations remit after the underlying ocular etiology is corrected, such as through ocular surgery.13 Other optical interventions, such as special glasses or contact lenses, may help maximize remaining vision.8 In patients without this option, such as Mr. B, there are limited data on beneficial medications for CBS.
Continue to: Evidence for treatment of CBS...
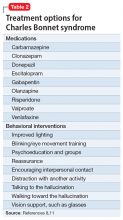
Evidence for treatment of CBS with antipsychotic medications is mixed. Some case studies have found them to be ineffective, while others have found agents such as olanzapine or risperidone to be partially helpful in reducing symptoms.14 There are also data from case reports that may support the use of cholinesterase inhibitors such as donepezil, antiepileptics (carbamazepine, valproate, gabapentin, and clonazepam), and certain antidepressants (escitalopram, venlafaxine) (Table 28,11).3
Addressing loneliness and social isolation
With minimal definitive evidence for pharmacologic management, the most important intervention for treating CBS may be changing the patient’s sensory environment. Specifically, loneliness and social isolation are major exacerbating factors of CBS, and many clinicians advocate for the consistent presence of a sympathetic professional. Reassurance that hallucinations are from ocular disease rather than a primary mental disorder may be extremely relieving for patients.11 A psychoeducation or support group may also be beneficial, not only for giving patients more social contact, but also for teaching them coping skills or strategies to reduce hallucinations, such as distraction, turning on more lights, or even certain eye/blinking movements.11 Table 28,11 (page 49) outlines behavioral interventions for CBS.
Regardless of etiology, Mr. B’s hallucinations significantly affected his quality of life. During his inpatient stay, he was treated with
OUTCOME Home care and family involvement
After discussion with Mr. B and his family about the risks and benefits of medication, the risperidone and donepezil are discontinued. Ultimately, it is determined that Mr. B requires a higher level of home care, both for his safety and to improve his social contact. Mr. B returns home with a combination of a professional home health aide and increased family involvement.
Bottom Line
When evaluating visual hallucinations in older adults, Charles Bonnet syndrome (CBS) should be considered. Sensory deprivation and social isolation are significant risk factors for CBS. While evidence is inconclusive for medical treatment, reassurance and behavioral interventions can often improve symptoms.
Continue to: Related Resources
Related Resources
- Charles Bonnet Syndrome Foundation. http://www.charlesbonnetsyndrome.org
- Schultz G, Melzack R. The Charles Bonnet syndrome: ‘phantom visual images’. Perception. 1991;20:809-825.
- Menon GJ, Rahman I, Menon SJ, et al. Complex visual hallucinations in the visually impaired: the Charles Bonnet syndrome. Surv Ophthalmol. 2003;48(1):58-72.
Drug Brand Names
Carbamazepine • Tegretol
Clonazepam • Klonopin
Donepezil • Aricept
Escitalopram • Lexapro
Gabapentin • Neurontin
Olanzapine • Zyprexa
Risperidone • Risperdal
Valproate • Depakote
Venlafaxine • Effexor
1. Menon GJ, Rahman I, Menon SJ, et al. Complex visual hallucinations in the visually impaired: the Charles Bonnet syndrome. Surv Ophthalmol. 2003;48(1):58-72.
2. Cox TM, Ffytche DH. Negative outcome Charles Bonnet syndrome. Br J Ophthalmol. 2014;98(9):1236-1239.
3. Pelak VS. Visual release hallucinations (Charles Bonnet syndrome). UpToDate. Updated February 5, 2019. Accessed September 17, 2020. https://www.uptodate.com/contents/visual-release-hallucinations-charles-bonnet-syndrome
4. Burke W. The neural basis of Charles Bonnet hallucinations: a hypothesis. J Neurol Neurosurg Psychiatry. 2002;73(5):535-541.
5. Scott IU, Schein OD, Feuer WJ, et al. Visual hallucinations in patients with retinal disease. Am J Ophthalmol. 2001;131(5):590-598.
6. Lepore FE. Spontaneous visual phenomena with visual loss: 104 patients with lesions of retinal and neural afferent pathways. Neurology. 1990;40(3 Pt 1):444-447.
7. Nesher R, Nesher G, Epstein E, et al. Charles Bonnet syndrome in glaucoma patients with low vision. J Glaucoma. 2001;10(5):396-400.
8. Pang L. Hallucinations experienced by visually impaired: Charles Bonnet syndrome. Optom Vis Sci. 2016;93(12):1466-1478.
9. Linszen M, Van Zanten G, Teunisse R, et al. Auditory hallucinations in adults with hearing impairment: a large prevalence study. Psychological Medicine. 2019;49(1):132-139.
10. Teunisse RJ, Cruysberg JR, Hoefnagels WH, et al. Social and psychological characteristics of elderly visually handicapped patients with the Charles Bonnet syndrome. Compr Psychiatry. 1999;40(4):315-319.
11. Eperjesi F, Akbarali A. Rehabilitation in Charles Bonnet syndrome: a review of treatment options. Clin Exp Optom. 2004;87(3):149-152.
12. Fernandez A, Lichtshein G, Vieweg WVR. The Charles Bonnet syndrome: a review. J Nen Ment Dis. 1997;185(3):195-200.
13. Rosenbaum F, Harati Y, Rolak L, et al. Visual hallucinations in sane people: Charles Bonnet syndrome. J Am Geriatr Soc. 1987;35(1):66-68.
14. Coletti Moja M, Milano E, Gasverde S, et al. Olanzapine therapy in hallucinatory visions related to Bonnet syndrome. Neurol Sci. 2005;26(3):168-170.
CASE Visual, auditory, and tactile hallucinations
Mr. B, age 93, is brought to the emergency department by his son after experiencing hallucinations where he reportedly saw and heard individuals in his home. In frustration, Mr. B wielded a knife because he “wanted them to go away.”
Mr. B and his son report that the hallucinations had begun 2 years ago, without prior trauma, medication changes, changes in social situation, or other apparent precipitating events. The hallucinations “come and go,” without preceding symptoms, but have recurring content involving a friendly man named “Harry,” people coming out of the television, 2 children playing, and water covering the floor. Mr. B acknowledges these are hallucinations and had not felt threatened by them until recently, when he wielded the knife. He often tries to talk to them, but they do not reply.
Mr. B also reports intermittent auditory hallucinations including voices at home (non-command) and papers rustling. He also describes tactile hallucinations, where he says he can feel Harry and others prodding him, knocking things out of his hands, or splashing him with water.
Mr. B is admitted to the hospital because he is a danger to himself and others. While on the inpatient unit, Mr. B is pleasant with staff, and eats and sleeps normally; however, he continues to have hallucinations of Harry. Mr. B reports seeing Harry in the hall, and says that Harry pulls out Mr. B’s earpiece and steals his fork. Mr. B also reports hearing a sound “like a bee buzzing.” Mr. B is started on risperidone, 1 mg nightly, for a presumed psychotic disorder.
HISTORY Independent and in good health
Mr. B lives alone and is independent in his activities of daily living. He spends his days at home, often visited by his children, who bring him groceries and other necessities.
Mr. B takes no medications, and has no history of psychiatric treatment; psychotic, manic, or depressive episodes; posttraumatic stress disorder; obsessive-compulsive disorder; or recent emotional stress. His medical history includes chronic progressive hearing loss, which is managed with hearing aids; macular degeneration; and prior bilateral cataract surgeries.
EVALUATION Mental status exam and objective findings
During his evaluation, Mr. B appears well-nourished, and wears glasses and hearing aids. During the interview, he is euthymic with appropriately reactive affect. He is talkative but redirectable, with a goal-directed thought process. Mr. B does not appear to be internally preoccupied. His hearing is impaired, and he often requires questions to be repeated loudly. He is oriented to person, place, and time. There are no signs of delusions, paranoia, thought blocking, thought broadcasting/insertion, or referential thinking. He denies depressed mood, anhedonia, fatigue, sleep changes, or manic symptoms. He denies the occurrence of auditory or visual hallucinations during the evaluation.
Continue to: A neurologic exam shows...
A neurologic exam shows impaired hearing bilaterally and impaired visual acuity. Even with glasses, both eyes have acuity only to finger counting. All other cranial nerves are normal, and Mr. B’s strength, sensation, and cerebellar function are all intact, without rigidity, numbness, or tingling. His gait is steady without a walker, with symmetric arm swing and slight dragging of his feet. His vitals are stable, with normal orthostatic pressures.
Other objective data include a score of 24/30 on the Mini-Mental State Examination, notable for deficits in visuospatial orientation, attention, and calculation, with language and copying limited by poor vision. Mr. B scores 16/22 on the Montreal Cognitive Assessment (MoCA)-Blind (adapted version of MoCA), which is equivalent to a 22/30 on the MoCA, indicating some mild cognitive impairment; however, this modified test is still limited by his poor hearing. His serum and urine laboratory workup show no liver, kidney, metabolic, or electrolyte abnormalities, no sign of infection, negative urine drug screen, and normal B12 and thyroid-stimulating hormone levels. He undergoes a brain MRI, which shows chronic microvascular ischemic change, without mass lesions, infarction, or other pathology.
[polldaddy:10729178]
The authors’ observations
Given Mr. B’s presentation, we ruled out a primary psychotic disorder. He had no psychiatric history, with organized thought, a reactive affect, and no delusions, paranoia, or other psychotic symptoms, all pointing against psychosis. His brain MRI showed no malignancy or other lesions. He had no substance use history to suggest intoxication/withdrawal. His intact attention and orientation did not suggest delirium, and his serum and urine studies were all negative. Although his blaming Harry for knocking things out of his hands could suggest confabulation, Mr. B had no other signs of Korsakoff syndrome, such as ataxia, general confusion, or malnourishment.
We also considered early dementia. There was suspicion for Lewy body dementia given Mr. B’s prominent fluctuating visual hallucinations; however, he displayed no other signs of the disorder, such as parkinsonism, dysautonomia, or sensitivity to the antipsychotic (risperidone 1 mg nightly) started on admission. The presence of 1 core feature of Lewy body dementia—visual hallucinations—indicated a possible, but not probable, diagnosis. Additionally, Mr. B did not have the characteristic features of other types of dementia, such as the stepwise progression of vascular dementia, the behavioral disinhibition of frontotemporal dementia, or the insidious forgetfulness, confusion, language problems, or paranoia that may appear in Alzheimer’s disease. Remarkably, he had a relatively normal brain MRI for his age, given chronic microvascular ischemic changes, and cognitive testing that indicated only mild impairment further pointed against a dementia process.
Charles Bonnet syndrome
Based on Mr. B’s severe vision loss and history of ocular surgeries, we diagnosed him with CBS, described as visual hallucinations in the presence of impaired vision. Charles Bonnet syndrome has been observed in several disorders that affect vision, most commonly macular degeneration, diabetic retinopathy, and glaucoma, with an estimated prevalence of 11% to 39% in older patients with ocular disease.1,2 Visual hallucinations in CBS occur due to ocular disease, likely resulting from changes in afferent sensory input to visual cortical regions of the brain. Table 13 outlines the features of visual hallucinations in patients with CBS. The subsequent disinhibition and spontaneous firing of the visual association cortices leads to the “release hallucinations” of the syndrome.4 The disorder is thought to be significantly underdiagnosed—in a survey of patients with CBS, only 15% had reported their visual hallucinations to a physician.5

Continue to: Mr. B's symptoms...
Mr. B’s symptoms are atypical for CBS, but they fit the diagnosis when considering the entire clinical picture. While hallucinations in CBS are more often simple shapes, complex hallucinations including people and scenes have been noted in several instances.6
Similar to Mr. B’s case, patients with CBS can have recurring figures in their hallucinations, and the images may even move across the visual field.1 Patients with CBS also frequently recognize that their hallucinations are not real, and may or may not be distressed by them.4 Patients with CBS often have hallucinations multiple times daily, lasting from a few seconds to many minutes,7 consistent with Mr. B’s temporary symptoms.
Although auditory and tactile hallucinations are typically not included in CBS, they can also be explained by Mr. B’s significant sensory impairment. Severe hearing impairment in geriatric adults has been associated with auditory hallucinations8; in 1 survey, half of these hallucinations consisted of voices.9 In contrast, tactile hallucinations are not described in sensory deprivation literature. However, in the context of Mr. B’s severe comorbid hearing and vision loss, we propose that these hallucinations reflect his interpretation of sensory events around him, and their integration into his extensive hallucination framework. In other words, Harry poking him and causing him to drop things may be Mr. B’s way of rationalizing events that he has trouble perceiving entirely, or his mild forgetfulness. Mr. B’s social isolation is another factor that may worsen his sensory deprivation and contribute to his extensive hallucinations.10 Additionally, his mild cognitive deficits on testing with chronic microvascular changes on the MRI may suggest a mild vascular-related dementia process, which could also exacerbate his hallucinations. While classic CBS occurs without cognitive impairment, dementia can often co-occur with CBS.11
TREATMENT No significant improvement with medications
During his inpatient stay, Mr. B is treated with risperidone, 1 mg nightly, and is also started on donepezil, 5 mg/d, to treat a possible comorbid dementia. However, he continues to hallucinate without significant improvement.
[polldaddy:10729181]
The authors’ observations
There is no definitive treatment for CBS, and while the hallucinations may spontaneously resolve, per case reports, this typically occurs only as visual loss progresses to total blindness.12 However, many patients can have the hallucinations remit after the underlying ocular etiology is corrected, such as through ocular surgery.13 Other optical interventions, such as special glasses or contact lenses, may help maximize remaining vision.8 In patients without this option, such as Mr. B, there are limited data on beneficial medications for CBS.
Continue to: Evidence for treatment of CBS...
Evidence for treatment of CBS with antipsychotic medications is mixed. Some case studies have found them to be ineffective, while others have found agents such as olanzapine or risperidone to be partially helpful in reducing symptoms.14 There are also data from case reports that may support the use of cholinesterase inhibitors such as donepezil, antiepileptics (carbamazepine, valproate, gabapentin, and clonazepam), and certain antidepressants (escitalopram, venlafaxine) (Table 28,11).3
Addressing loneliness and social isolation
With minimal definitive evidence for pharmacologic management, the most important intervention for treating CBS may be changing the patient’s sensory environment. Specifically, loneliness and social isolation are major exacerbating factors of CBS, and many clinicians advocate for the consistent presence of a sympathetic professional. Reassurance that hallucinations are from ocular disease rather than a primary mental disorder may be extremely relieving for patients.11 A psychoeducation or support group may also be beneficial, not only for giving patients more social contact, but also for teaching them coping skills or strategies to reduce hallucinations, such as distraction, turning on more lights, or even certain eye/blinking movements.11 Table 28,11 (page 49) outlines behavioral interventions for CBS.
Regardless of etiology, Mr. B’s hallucinations significantly affected his quality of life. During his inpatient stay, he was treated with
OUTCOME Home care and family involvement
After discussion with Mr. B and his family about the risks and benefits of medication, the risperidone and donepezil are discontinued. Ultimately, it is determined that Mr. B requires a higher level of home care, both for his safety and to improve his social contact. Mr. B returns home with a combination of a professional home health aide and increased family involvement.
Bottom Line
When evaluating visual hallucinations in older adults, Charles Bonnet syndrome (CBS) should be considered. Sensory deprivation and social isolation are significant risk factors for CBS. While evidence is inconclusive for medical treatment, reassurance and behavioral interventions can often improve symptoms.
Continue to: Related Resources
Related Resources
- Charles Bonnet Syndrome Foundation. http://www.charlesbonnetsyndrome.org
- Schultz G, Melzack R. The Charles Bonnet syndrome: ‘phantom visual images’. Perception. 1991;20:809-825.
- Menon GJ, Rahman I, Menon SJ, et al. Complex visual hallucinations in the visually impaired: the Charles Bonnet syndrome. Surv Ophthalmol. 2003;48(1):58-72.
Drug Brand Names
Carbamazepine • Tegretol
Clonazepam • Klonopin
Donepezil • Aricept
Escitalopram • Lexapro
Gabapentin • Neurontin
Olanzapine • Zyprexa
Risperidone • Risperdal
Valproate • Depakote
Venlafaxine • Effexor
CASE Visual, auditory, and tactile hallucinations
Mr. B, age 93, is brought to the emergency department by his son after experiencing hallucinations where he reportedly saw and heard individuals in his home. In frustration, Mr. B wielded a knife because he “wanted them to go away.”
Mr. B and his son report that the hallucinations had begun 2 years ago, without prior trauma, medication changes, changes in social situation, or other apparent precipitating events. The hallucinations “come and go,” without preceding symptoms, but have recurring content involving a friendly man named “Harry,” people coming out of the television, 2 children playing, and water covering the floor. Mr. B acknowledges these are hallucinations and had not felt threatened by them until recently, when he wielded the knife. He often tries to talk to them, but they do not reply.
Mr. B also reports intermittent auditory hallucinations including voices at home (non-command) and papers rustling. He also describes tactile hallucinations, where he says he can feel Harry and others prodding him, knocking things out of his hands, or splashing him with water.
Mr. B is admitted to the hospital because he is a danger to himself and others. While on the inpatient unit, Mr. B is pleasant with staff, and eats and sleeps normally; however, he continues to have hallucinations of Harry. Mr. B reports seeing Harry in the hall, and says that Harry pulls out Mr. B’s earpiece and steals his fork. Mr. B also reports hearing a sound “like a bee buzzing.” Mr. B is started on risperidone, 1 mg nightly, for a presumed psychotic disorder.
HISTORY Independent and in good health
Mr. B lives alone and is independent in his activities of daily living. He spends his days at home, often visited by his children, who bring him groceries and other necessities.
Mr. B takes no medications, and has no history of psychiatric treatment; psychotic, manic, or depressive episodes; posttraumatic stress disorder; obsessive-compulsive disorder; or recent emotional stress. His medical history includes chronic progressive hearing loss, which is managed with hearing aids; macular degeneration; and prior bilateral cataract surgeries.
EVALUATION Mental status exam and objective findings
During his evaluation, Mr. B appears well-nourished, and wears glasses and hearing aids. During the interview, he is euthymic with appropriately reactive affect. He is talkative but redirectable, with a goal-directed thought process. Mr. B does not appear to be internally preoccupied. His hearing is impaired, and he often requires questions to be repeated loudly. He is oriented to person, place, and time. There are no signs of delusions, paranoia, thought blocking, thought broadcasting/insertion, or referential thinking. He denies depressed mood, anhedonia, fatigue, sleep changes, or manic symptoms. He denies the occurrence of auditory or visual hallucinations during the evaluation.
Continue to: A neurologic exam shows...
A neurologic exam shows impaired hearing bilaterally and impaired visual acuity. Even with glasses, both eyes have acuity only to finger counting. All other cranial nerves are normal, and Mr. B’s strength, sensation, and cerebellar function are all intact, without rigidity, numbness, or tingling. His gait is steady without a walker, with symmetric arm swing and slight dragging of his feet. His vitals are stable, with normal orthostatic pressures.
Other objective data include a score of 24/30 on the Mini-Mental State Examination, notable for deficits in visuospatial orientation, attention, and calculation, with language and copying limited by poor vision. Mr. B scores 16/22 on the Montreal Cognitive Assessment (MoCA)-Blind (adapted version of MoCA), which is equivalent to a 22/30 on the MoCA, indicating some mild cognitive impairment; however, this modified test is still limited by his poor hearing. His serum and urine laboratory workup show no liver, kidney, metabolic, or electrolyte abnormalities, no sign of infection, negative urine drug screen, and normal B12 and thyroid-stimulating hormone levels. He undergoes a brain MRI, which shows chronic microvascular ischemic change, without mass lesions, infarction, or other pathology.
[polldaddy:10729178]
The authors’ observations
Given Mr. B’s presentation, we ruled out a primary psychotic disorder. He had no psychiatric history, with organized thought, a reactive affect, and no delusions, paranoia, or other psychotic symptoms, all pointing against psychosis. His brain MRI showed no malignancy or other lesions. He had no substance use history to suggest intoxication/withdrawal. His intact attention and orientation did not suggest delirium, and his serum and urine studies were all negative. Although his blaming Harry for knocking things out of his hands could suggest confabulation, Mr. B had no other signs of Korsakoff syndrome, such as ataxia, general confusion, or malnourishment.
We also considered early dementia. There was suspicion for Lewy body dementia given Mr. B’s prominent fluctuating visual hallucinations; however, he displayed no other signs of the disorder, such as parkinsonism, dysautonomia, or sensitivity to the antipsychotic (risperidone 1 mg nightly) started on admission. The presence of 1 core feature of Lewy body dementia—visual hallucinations—indicated a possible, but not probable, diagnosis. Additionally, Mr. B did not have the characteristic features of other types of dementia, such as the stepwise progression of vascular dementia, the behavioral disinhibition of frontotemporal dementia, or the insidious forgetfulness, confusion, language problems, or paranoia that may appear in Alzheimer’s disease. Remarkably, he had a relatively normal brain MRI for his age, given chronic microvascular ischemic changes, and cognitive testing that indicated only mild impairment further pointed against a dementia process.
Charles Bonnet syndrome
Based on Mr. B’s severe vision loss and history of ocular surgeries, we diagnosed him with CBS, described as visual hallucinations in the presence of impaired vision. Charles Bonnet syndrome has been observed in several disorders that affect vision, most commonly macular degeneration, diabetic retinopathy, and glaucoma, with an estimated prevalence of 11% to 39% in older patients with ocular disease.1,2 Visual hallucinations in CBS occur due to ocular disease, likely resulting from changes in afferent sensory input to visual cortical regions of the brain. Table 13 outlines the features of visual hallucinations in patients with CBS. The subsequent disinhibition and spontaneous firing of the visual association cortices leads to the “release hallucinations” of the syndrome.4 The disorder is thought to be significantly underdiagnosed—in a survey of patients with CBS, only 15% had reported their visual hallucinations to a physician.5

Continue to: Mr. B's symptoms...
Mr. B’s symptoms are atypical for CBS, but they fit the diagnosis when considering the entire clinical picture. While hallucinations in CBS are more often simple shapes, complex hallucinations including people and scenes have been noted in several instances.6
Similar to Mr. B’s case, patients with CBS can have recurring figures in their hallucinations, and the images may even move across the visual field.1 Patients with CBS also frequently recognize that their hallucinations are not real, and may or may not be distressed by them.4 Patients with CBS often have hallucinations multiple times daily, lasting from a few seconds to many minutes,7 consistent with Mr. B’s temporary symptoms.
Although auditory and tactile hallucinations are typically not included in CBS, they can also be explained by Mr. B’s significant sensory impairment. Severe hearing impairment in geriatric adults has been associated with auditory hallucinations8; in 1 survey, half of these hallucinations consisted of voices.9 In contrast, tactile hallucinations are not described in sensory deprivation literature. However, in the context of Mr. B’s severe comorbid hearing and vision loss, we propose that these hallucinations reflect his interpretation of sensory events around him, and their integration into his extensive hallucination framework. In other words, Harry poking him and causing him to drop things may be Mr. B’s way of rationalizing events that he has trouble perceiving entirely, or his mild forgetfulness. Mr. B’s social isolation is another factor that may worsen his sensory deprivation and contribute to his extensive hallucinations.10 Additionally, his mild cognitive deficits on testing with chronic microvascular changes on the MRI may suggest a mild vascular-related dementia process, which could also exacerbate his hallucinations. While classic CBS occurs without cognitive impairment, dementia can often co-occur with CBS.11
TREATMENT No significant improvement with medications
During his inpatient stay, Mr. B is treated with risperidone, 1 mg nightly, and is also started on donepezil, 5 mg/d, to treat a possible comorbid dementia. However, he continues to hallucinate without significant improvement.
[polldaddy:10729181]
The authors’ observations
There is no definitive treatment for CBS, and while the hallucinations may spontaneously resolve, per case reports, this typically occurs only as visual loss progresses to total blindness.12 However, many patients can have the hallucinations remit after the underlying ocular etiology is corrected, such as through ocular surgery.13 Other optical interventions, such as special glasses or contact lenses, may help maximize remaining vision.8 In patients without this option, such as Mr. B, there are limited data on beneficial medications for CBS.
Continue to: Evidence for treatment of CBS...
Evidence for treatment of CBS with antipsychotic medications is mixed. Some case studies have found them to be ineffective, while others have found agents such as olanzapine or risperidone to be partially helpful in reducing symptoms.14 There are also data from case reports that may support the use of cholinesterase inhibitors such as donepezil, antiepileptics (carbamazepine, valproate, gabapentin, and clonazepam), and certain antidepressants (escitalopram, venlafaxine) (Table 28,11).3
Addressing loneliness and social isolation
With minimal definitive evidence for pharmacologic management, the most important intervention for treating CBS may be changing the patient’s sensory environment. Specifically, loneliness and social isolation are major exacerbating factors of CBS, and many clinicians advocate for the consistent presence of a sympathetic professional. Reassurance that hallucinations are from ocular disease rather than a primary mental disorder may be extremely relieving for patients.11 A psychoeducation or support group may also be beneficial, not only for giving patients more social contact, but also for teaching them coping skills or strategies to reduce hallucinations, such as distraction, turning on more lights, or even certain eye/blinking movements.11 Table 28,11 (page 49) outlines behavioral interventions for CBS.
Regardless of etiology, Mr. B’s hallucinations significantly affected his quality of life. During his inpatient stay, he was treated with
OUTCOME Home care and family involvement
After discussion with Mr. B and his family about the risks and benefits of medication, the risperidone and donepezil are discontinued. Ultimately, it is determined that Mr. B requires a higher level of home care, both for his safety and to improve his social contact. Mr. B returns home with a combination of a professional home health aide and increased family involvement.
Bottom Line
When evaluating visual hallucinations in older adults, Charles Bonnet syndrome (CBS) should be considered. Sensory deprivation and social isolation are significant risk factors for CBS. While evidence is inconclusive for medical treatment, reassurance and behavioral interventions can often improve symptoms.
Continue to: Related Resources
Related Resources
- Charles Bonnet Syndrome Foundation. http://www.charlesbonnetsyndrome.org
- Schultz G, Melzack R. The Charles Bonnet syndrome: ‘phantom visual images’. Perception. 1991;20:809-825.
- Menon GJ, Rahman I, Menon SJ, et al. Complex visual hallucinations in the visually impaired: the Charles Bonnet syndrome. Surv Ophthalmol. 2003;48(1):58-72.
Drug Brand Names
Carbamazepine • Tegretol
Clonazepam • Klonopin
Donepezil • Aricept
Escitalopram • Lexapro
Gabapentin • Neurontin
Olanzapine • Zyprexa
Risperidone • Risperdal
Valproate • Depakote
Venlafaxine • Effexor
1. Menon GJ, Rahman I, Menon SJ, et al. Complex visual hallucinations in the visually impaired: the Charles Bonnet syndrome. Surv Ophthalmol. 2003;48(1):58-72.
2. Cox TM, Ffytche DH. Negative outcome Charles Bonnet syndrome. Br J Ophthalmol. 2014;98(9):1236-1239.
3. Pelak VS. Visual release hallucinations (Charles Bonnet syndrome). UpToDate. Updated February 5, 2019. Accessed September 17, 2020. https://www.uptodate.com/contents/visual-release-hallucinations-charles-bonnet-syndrome
4. Burke W. The neural basis of Charles Bonnet hallucinations: a hypothesis. J Neurol Neurosurg Psychiatry. 2002;73(5):535-541.
5. Scott IU, Schein OD, Feuer WJ, et al. Visual hallucinations in patients with retinal disease. Am J Ophthalmol. 2001;131(5):590-598.
6. Lepore FE. Spontaneous visual phenomena with visual loss: 104 patients with lesions of retinal and neural afferent pathways. Neurology. 1990;40(3 Pt 1):444-447.
7. Nesher R, Nesher G, Epstein E, et al. Charles Bonnet syndrome in glaucoma patients with low vision. J Glaucoma. 2001;10(5):396-400.
8. Pang L. Hallucinations experienced by visually impaired: Charles Bonnet syndrome. Optom Vis Sci. 2016;93(12):1466-1478.
9. Linszen M, Van Zanten G, Teunisse R, et al. Auditory hallucinations in adults with hearing impairment: a large prevalence study. Psychological Medicine. 2019;49(1):132-139.
10. Teunisse RJ, Cruysberg JR, Hoefnagels WH, et al. Social and psychological characteristics of elderly visually handicapped patients with the Charles Bonnet syndrome. Compr Psychiatry. 1999;40(4):315-319.
11. Eperjesi F, Akbarali A. Rehabilitation in Charles Bonnet syndrome: a review of treatment options. Clin Exp Optom. 2004;87(3):149-152.
12. Fernandez A, Lichtshein G, Vieweg WVR. The Charles Bonnet syndrome: a review. J Nen Ment Dis. 1997;185(3):195-200.
13. Rosenbaum F, Harati Y, Rolak L, et al. Visual hallucinations in sane people: Charles Bonnet syndrome. J Am Geriatr Soc. 1987;35(1):66-68.
14. Coletti Moja M, Milano E, Gasverde S, et al. Olanzapine therapy in hallucinatory visions related to Bonnet syndrome. Neurol Sci. 2005;26(3):168-170.
1. Menon GJ, Rahman I, Menon SJ, et al. Complex visual hallucinations in the visually impaired: the Charles Bonnet syndrome. Surv Ophthalmol. 2003;48(1):58-72.
2. Cox TM, Ffytche DH. Negative outcome Charles Bonnet syndrome. Br J Ophthalmol. 2014;98(9):1236-1239.
3. Pelak VS. Visual release hallucinations (Charles Bonnet syndrome). UpToDate. Updated February 5, 2019. Accessed September 17, 2020. https://www.uptodate.com/contents/visual-release-hallucinations-charles-bonnet-syndrome
4. Burke W. The neural basis of Charles Bonnet hallucinations: a hypothesis. J Neurol Neurosurg Psychiatry. 2002;73(5):535-541.
5. Scott IU, Schein OD, Feuer WJ, et al. Visual hallucinations in patients with retinal disease. Am J Ophthalmol. 2001;131(5):590-598.
6. Lepore FE. Spontaneous visual phenomena with visual loss: 104 patients with lesions of retinal and neural afferent pathways. Neurology. 1990;40(3 Pt 1):444-447.
7. Nesher R, Nesher G, Epstein E, et al. Charles Bonnet syndrome in glaucoma patients with low vision. J Glaucoma. 2001;10(5):396-400.
8. Pang L. Hallucinations experienced by visually impaired: Charles Bonnet syndrome. Optom Vis Sci. 2016;93(12):1466-1478.
9. Linszen M, Van Zanten G, Teunisse R, et al. Auditory hallucinations in adults with hearing impairment: a large prevalence study. Psychological Medicine. 2019;49(1):132-139.
10. Teunisse RJ, Cruysberg JR, Hoefnagels WH, et al. Social and psychological characteristics of elderly visually handicapped patients with the Charles Bonnet syndrome. Compr Psychiatry. 1999;40(4):315-319.
11. Eperjesi F, Akbarali A. Rehabilitation in Charles Bonnet syndrome: a review of treatment options. Clin Exp Optom. 2004;87(3):149-152.
12. Fernandez A, Lichtshein G, Vieweg WVR. The Charles Bonnet syndrome: a review. J Nen Ment Dis. 1997;185(3):195-200.
13. Rosenbaum F, Harati Y, Rolak L, et al. Visual hallucinations in sane people: Charles Bonnet syndrome. J Am Geriatr Soc. 1987;35(1):66-68.
14. Coletti Moja M, Milano E, Gasverde S, et al. Olanzapine therapy in hallucinatory visions related to Bonnet syndrome. Neurol Sci. 2005;26(3):168-170.
APA apologizes for past support of racism in psychiatry
The American Psychiatric Association has issued a formal apology for its past support of structural racism in psychiatry.
The apology, issued Jan. 18, coincided with the federal holiday honoring the life and work of civil rights activist Dr. Martin Luther King Jr.
“We apologize for our role in perpetrating structural racism in this country, and we hope to begin to make amends for APA’s and psychiatry’s history of actions, intentional and not, that hurt Black, indigenous, and people of color,” APA President Jeffrey Geller, MD, MPH, said in a statement.
The apology was written and issued by the APA Board of Trustees. It acknowledges practices and events in psychiatry that contributed to racial inequality, and expresses the organization’s commitment to developing antiracist policies that promote equity in mental health for all.
“This apology is one important step we needed to take to move forward to a more equitable future. The board is issuing this document on Martin Luther King Jr. Day, because we hope that it honors his life’s work of reconciliation and equality. We do not take that legacy or his call to action lightly and will continue our important work,” said Dr. Geller.
One involved the Eastern State Hospital in Williamsburg, Va., the nation’s first psychiatric care facility, founded in 1773.
Eastern State, which for a time in the 1800s was called the Eastern Lunatic Asylum, was not segregated when founded. However, 70 years later, when the 13 founders of what is now the APA met to discuss improvements in mental health care delivery, the treatment system they created and the organization they founded aligned with that era’s racist social and political policies. In this system, Black patients received psychiatric care separately from White patients, the APA said.
The APA also acknowledged failing to act in Black Americans’ best interest at critical points in the United States’ sociopolitical evolution throughout the 19th and 20th centuries.
“This inactivity was notably evident while white supremacists lynched Black people during the Reconstruction Era as well as when Jim Crow segregation was in effect, which led to ‘separate but equal’ standards of care starting in 1896,” the APA said.
Later, the APA failed to declare support for Brown v. Board of Education of Topeka in 1954, along with further major civil rights legislation designed to improve social and psychological conditions for Black people, the organization admitted.
Throughout the decades that followed, psychiatric misdiagnosis among Black, indigenous, and people of color populations were also common, the APA acknowledged.
For example, late 20th century psychiatrists commonly attributed their minority patients’ frustrations to schizophrenia, while categorizing similar behaviors as “neuroticism” in White patients.
The APA pointed to one study which found that APA members diagnosed more Black than White patients with schizophrenia, even when both had otherwise identical clinical presentations.
“This reveals the basis for embedded discrimination within psychiatry that has contributed to reduced quality of care” for Black, indigenous, and people of color, and “perpetuation of dangerous stereotypes,” the APA said.
Saul Levin, MD, the APA’s medical director and CEO, said the Board of Trustees has taken “an important step in issuing this apology. The APA administration is committed to working toward inclusion, health equity, and fairness that everyone deserves.”
The APA Board of Trustees began drafting the apology late last year after it concluded that events and persistent inequities in health care and psychiatry had highlighted an organizational need for action.
The APA’s Presidential Task Force on Structural Racism is continuing with efforts to educate and engage members on the issue and implement changes within the organization.
A version of this article first appeared on Medscape.com.
The American Psychiatric Association has issued a formal apology for its past support of structural racism in psychiatry.
The apology, issued Jan. 18, coincided with the federal holiday honoring the life and work of civil rights activist Dr. Martin Luther King Jr.
“We apologize for our role in perpetrating structural racism in this country, and we hope to begin to make amends for APA’s and psychiatry’s history of actions, intentional and not, that hurt Black, indigenous, and people of color,” APA President Jeffrey Geller, MD, MPH, said in a statement.
The apology was written and issued by the APA Board of Trustees. It acknowledges practices and events in psychiatry that contributed to racial inequality, and expresses the organization’s commitment to developing antiracist policies that promote equity in mental health for all.
“This apology is one important step we needed to take to move forward to a more equitable future. The board is issuing this document on Martin Luther King Jr. Day, because we hope that it honors his life’s work of reconciliation and equality. We do not take that legacy or his call to action lightly and will continue our important work,” said Dr. Geller.
One involved the Eastern State Hospital in Williamsburg, Va., the nation’s first psychiatric care facility, founded in 1773.
Eastern State, which for a time in the 1800s was called the Eastern Lunatic Asylum, was not segregated when founded. However, 70 years later, when the 13 founders of what is now the APA met to discuss improvements in mental health care delivery, the treatment system they created and the organization they founded aligned with that era’s racist social and political policies. In this system, Black patients received psychiatric care separately from White patients, the APA said.
The APA also acknowledged failing to act in Black Americans’ best interest at critical points in the United States’ sociopolitical evolution throughout the 19th and 20th centuries.
“This inactivity was notably evident while white supremacists lynched Black people during the Reconstruction Era as well as when Jim Crow segregation was in effect, which led to ‘separate but equal’ standards of care starting in 1896,” the APA said.
Later, the APA failed to declare support for Brown v. Board of Education of Topeka in 1954, along with further major civil rights legislation designed to improve social and psychological conditions for Black people, the organization admitted.
Throughout the decades that followed, psychiatric misdiagnosis among Black, indigenous, and people of color populations were also common, the APA acknowledged.
For example, late 20th century psychiatrists commonly attributed their minority patients’ frustrations to schizophrenia, while categorizing similar behaviors as “neuroticism” in White patients.
The APA pointed to one study which found that APA members diagnosed more Black than White patients with schizophrenia, even when both had otherwise identical clinical presentations.
“This reveals the basis for embedded discrimination within psychiatry that has contributed to reduced quality of care” for Black, indigenous, and people of color, and “perpetuation of dangerous stereotypes,” the APA said.
Saul Levin, MD, the APA’s medical director and CEO, said the Board of Trustees has taken “an important step in issuing this apology. The APA administration is committed to working toward inclusion, health equity, and fairness that everyone deserves.”
The APA Board of Trustees began drafting the apology late last year after it concluded that events and persistent inequities in health care and psychiatry had highlighted an organizational need for action.
The APA’s Presidential Task Force on Structural Racism is continuing with efforts to educate and engage members on the issue and implement changes within the organization.
A version of this article first appeared on Medscape.com.
The American Psychiatric Association has issued a formal apology for its past support of structural racism in psychiatry.
The apology, issued Jan. 18, coincided with the federal holiday honoring the life and work of civil rights activist Dr. Martin Luther King Jr.
“We apologize for our role in perpetrating structural racism in this country, and we hope to begin to make amends for APA’s and psychiatry’s history of actions, intentional and not, that hurt Black, indigenous, and people of color,” APA President Jeffrey Geller, MD, MPH, said in a statement.
The apology was written and issued by the APA Board of Trustees. It acknowledges practices and events in psychiatry that contributed to racial inequality, and expresses the organization’s commitment to developing antiracist policies that promote equity in mental health for all.
“This apology is one important step we needed to take to move forward to a more equitable future. The board is issuing this document on Martin Luther King Jr. Day, because we hope that it honors his life’s work of reconciliation and equality. We do not take that legacy or his call to action lightly and will continue our important work,” said Dr. Geller.
One involved the Eastern State Hospital in Williamsburg, Va., the nation’s first psychiatric care facility, founded in 1773.
Eastern State, which for a time in the 1800s was called the Eastern Lunatic Asylum, was not segregated when founded. However, 70 years later, when the 13 founders of what is now the APA met to discuss improvements in mental health care delivery, the treatment system they created and the organization they founded aligned with that era’s racist social and political policies. In this system, Black patients received psychiatric care separately from White patients, the APA said.
The APA also acknowledged failing to act in Black Americans’ best interest at critical points in the United States’ sociopolitical evolution throughout the 19th and 20th centuries.
“This inactivity was notably evident while white supremacists lynched Black people during the Reconstruction Era as well as when Jim Crow segregation was in effect, which led to ‘separate but equal’ standards of care starting in 1896,” the APA said.
Later, the APA failed to declare support for Brown v. Board of Education of Topeka in 1954, along with further major civil rights legislation designed to improve social and psychological conditions for Black people, the organization admitted.
Throughout the decades that followed, psychiatric misdiagnosis among Black, indigenous, and people of color populations were also common, the APA acknowledged.
For example, late 20th century psychiatrists commonly attributed their minority patients’ frustrations to schizophrenia, while categorizing similar behaviors as “neuroticism” in White patients.
The APA pointed to one study which found that APA members diagnosed more Black than White patients with schizophrenia, even when both had otherwise identical clinical presentations.
“This reveals the basis for embedded discrimination within psychiatry that has contributed to reduced quality of care” for Black, indigenous, and people of color, and “perpetuation of dangerous stereotypes,” the APA said.
Saul Levin, MD, the APA’s medical director and CEO, said the Board of Trustees has taken “an important step in issuing this apology. The APA administration is committed to working toward inclusion, health equity, and fairness that everyone deserves.”
The APA Board of Trustees began drafting the apology late last year after it concluded that events and persistent inequities in health care and psychiatry had highlighted an organizational need for action.
The APA’s Presidential Task Force on Structural Racism is continuing with efforts to educate and engage members on the issue and implement changes within the organization.
A version of this article first appeared on Medscape.com.
Machine learning flags key risk factors for suicide attempts
A history of suicidal behaviors or ideation, functional impairment related to mental health disorders, and socioeconomic disadvantage are the three most important risk factors predicting subsequent suicide attempts, new research suggests.
Investigators applied a machine-learning model to data on over 34,500 adults drawn from a large national survey database. After analyzing more than 2,500 survey questions, key areas were identified that yielded the most accurate predictions of who might be at risk for later suicide attempt.
These predictors included experiencing previous suicidal behaviors and ideation or functional impairment because of emotional problems, being at a younger age, having a lower educational achievement, and experiencing a recent financial crisis.
“Our machine learning model confirmed well-known risk factors of suicide attempt, including previous suicidal behavior and depression; and we also identified functional impairment, such as doing activities less carefully or accomplishing less because of emotional problems, as a new important risk,” lead author Angel Garcia de la Garza, PhD candidate in the department of biostatistics, Columbia University, New York, said in an interview.
“We hope our results provide a novel avenue for future suicide risk assessment,” Mr. Garcia de la Garza said.
The findings were published online Jan. 6 in JAMA Psychiatry.
‘Rich’ dataset
Previous research using machine learning approaches to study nonfatal suicide attempt prediction has focused on high-risk patients in clinical treatment. However, more than one-third of individuals making nonfatal suicide attempts do not receive mental health treatment, Mr. Garcia de la Garza noted.
To gain further insight into predictors of suicide risk in nonclinical populations, the researchers turned to the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), a longitudinal survey of noninstitutionalized U.S. adults.
“We wanted to extend our understanding of suicide attempt risk factors beyond high-risk clinical populations to the general adult population; and the richness of the NESARC dataset provides a unique opportunity to do so,” Mr. Garcia de la Garza said.
The NESARC surveys were conducted in two waves: Wave 1 (2001-2002) and wave 2 (2004-2005), in which participants self-reported nonfatal suicide attempts in the preceding 3 years since wave 1.
Assessment of wave 1 participants was based on the Alcohol Use Disorder and Associated Disabilities Interview Schedule DSM-IV.
“This survey’s extensive assessment instrument contained a detailed evaluation of substance use, psychiatric disorders, and symptoms not routinely available in electronic health records,” Mr. Garcia de la Garza noted.
The wave 1 survey contained 2,805 separate questions. From participants’ responses, the investigators derived 180 variables for three categories: past-year, prior-to-past-year, and lifetime mental disorders.
They then identified 2,978 factors associated with suicide attempts and used a statistical method called balanced random forest to classify suicide attempts at wave 2. Each variable was accorded an “importance score” using identified wave 1 features.
The outcome variable of attempted suicide at any point during the 3 years prior to the wave 2 interview was defined by combining responses to three wave 2 questions:
- In your entire life, did you ever attempt suicide?
- If yes, how old were you the first time?
- If the most recent event occurred within the last 3 years, how old were you during the most recent time?
Suicide risk severity was classified into four groups (low, medium, high, and very high) on the basis of the top-performing risk factors.
A statistical model combining survey design and nonresponse weights enabled estimates to be representative of the U.S. population, based on the 2000 census.
Out-of-fold model prediction assessed performance of the model, using area under receiver operator curve (AUC), sensitivity, and specificity.
Daily functioning
Of all participants, 70.2% (n = 34,653; almost 60% women) completed wave 2 interviews. The weighted mean ages at waves 1 and 2 were 45.1 and 48.2 years, respectively.
Of wave 2 respondents, 0.6% (n = 222) attempted suicide during the preceding 3 years.
Half of those who attempted suicide within the first year were classified as “very high risk,” while 33.2% of those who attempted suicide between the first and second year and 33.3% of those who attempted suicide between the second and third year were classified as “very high risk.”
Among participants who attempted suicide between the third year and follow-up, 16.48% were classified as “very high risk.”
The model accurately captured classification of participants, even across demographic characteristics, such as age, sex, race, and income.
Younger individuals (aged 18-36 years) were at higher risk, compared with older individuals. In addition, women were at higher risk than were men, White participants were at higher risk than were non-White participants, and individuals with lower income were at greater risk than were those with higher income.
The model found that 1.8% of the U.S. population had a 10% or greater risk of a suicide attempt.
The most important risk factors identified were the three questions about previous suicidal ideation or behavior; three items from the 12-Item Short Form Health Survey (feeling downhearted, doing activities less carefully, or accomplishing less because of emotional problems); younger age; lower educational achievement; and recent financial crisis.
“The clinical assessment of suicide risk typically focuses on acute suicidal symptoms, together with depression, anxiety, substance misuse, and recent stressful events,” coinvestigator Mark Olfson, MD, PhD, professor of epidemiology, Columbia University Irving Medical Center, New York, said in an interview.
Dr. Olfson said.
Extra vigilance
Commenting on the study in an interview, April C. Foreman, PhD, an executive board member of the American Association of Suicidology, noted that some of the findings were not surprising.
“When discharging a patient from inpatient care, or seeing them in primary care, bring up mental health concerns proactively and ask whether they have ever attempted suicide or harmed themselves – even a long time ago – just as you ask about a family history of heart disease or cancer, or other health issues,” said Dr. Foreman, chief medical officer of the Kevin and Margaret Hines Foundation.
She noted that half of people who die by suicide have a primary care visit within the preceding month.
“Primary care is a great place to get a suicide history and follow the patient with extra vigilance, just as you would with any other risk factors,” Dr. Foreman said.
The study was funded by the National Institute on Alcohol Abuse and Alcoholism and its Intramural Program. The study authors and Dr. Foreman have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A history of suicidal behaviors or ideation, functional impairment related to mental health disorders, and socioeconomic disadvantage are the three most important risk factors predicting subsequent suicide attempts, new research suggests.
Investigators applied a machine-learning model to data on over 34,500 adults drawn from a large national survey database. After analyzing more than 2,500 survey questions, key areas were identified that yielded the most accurate predictions of who might be at risk for later suicide attempt.
These predictors included experiencing previous suicidal behaviors and ideation or functional impairment because of emotional problems, being at a younger age, having a lower educational achievement, and experiencing a recent financial crisis.
“Our machine learning model confirmed well-known risk factors of suicide attempt, including previous suicidal behavior and depression; and we also identified functional impairment, such as doing activities less carefully or accomplishing less because of emotional problems, as a new important risk,” lead author Angel Garcia de la Garza, PhD candidate in the department of biostatistics, Columbia University, New York, said in an interview.
“We hope our results provide a novel avenue for future suicide risk assessment,” Mr. Garcia de la Garza said.
The findings were published online Jan. 6 in JAMA Psychiatry.
‘Rich’ dataset
Previous research using machine learning approaches to study nonfatal suicide attempt prediction has focused on high-risk patients in clinical treatment. However, more than one-third of individuals making nonfatal suicide attempts do not receive mental health treatment, Mr. Garcia de la Garza noted.
To gain further insight into predictors of suicide risk in nonclinical populations, the researchers turned to the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), a longitudinal survey of noninstitutionalized U.S. adults.
“We wanted to extend our understanding of suicide attempt risk factors beyond high-risk clinical populations to the general adult population; and the richness of the NESARC dataset provides a unique opportunity to do so,” Mr. Garcia de la Garza said.
The NESARC surveys were conducted in two waves: Wave 1 (2001-2002) and wave 2 (2004-2005), in which participants self-reported nonfatal suicide attempts in the preceding 3 years since wave 1.
Assessment of wave 1 participants was based on the Alcohol Use Disorder and Associated Disabilities Interview Schedule DSM-IV.
“This survey’s extensive assessment instrument contained a detailed evaluation of substance use, psychiatric disorders, and symptoms not routinely available in electronic health records,” Mr. Garcia de la Garza noted.
The wave 1 survey contained 2,805 separate questions. From participants’ responses, the investigators derived 180 variables for three categories: past-year, prior-to-past-year, and lifetime mental disorders.
They then identified 2,978 factors associated with suicide attempts and used a statistical method called balanced random forest to classify suicide attempts at wave 2. Each variable was accorded an “importance score” using identified wave 1 features.
The outcome variable of attempted suicide at any point during the 3 years prior to the wave 2 interview was defined by combining responses to three wave 2 questions:
- In your entire life, did you ever attempt suicide?
- If yes, how old were you the first time?
- If the most recent event occurred within the last 3 years, how old were you during the most recent time?
Suicide risk severity was classified into four groups (low, medium, high, and very high) on the basis of the top-performing risk factors.
A statistical model combining survey design and nonresponse weights enabled estimates to be representative of the U.S. population, based on the 2000 census.
Out-of-fold model prediction assessed performance of the model, using area under receiver operator curve (AUC), sensitivity, and specificity.
Daily functioning
Of all participants, 70.2% (n = 34,653; almost 60% women) completed wave 2 interviews. The weighted mean ages at waves 1 and 2 were 45.1 and 48.2 years, respectively.
Of wave 2 respondents, 0.6% (n = 222) attempted suicide during the preceding 3 years.
Half of those who attempted suicide within the first year were classified as “very high risk,” while 33.2% of those who attempted suicide between the first and second year and 33.3% of those who attempted suicide between the second and third year were classified as “very high risk.”
Among participants who attempted suicide between the third year and follow-up, 16.48% were classified as “very high risk.”
The model accurately captured classification of participants, even across demographic characteristics, such as age, sex, race, and income.
Younger individuals (aged 18-36 years) were at higher risk, compared with older individuals. In addition, women were at higher risk than were men, White participants were at higher risk than were non-White participants, and individuals with lower income were at greater risk than were those with higher income.
The model found that 1.8% of the U.S. population had a 10% or greater risk of a suicide attempt.
The most important risk factors identified were the three questions about previous suicidal ideation or behavior; three items from the 12-Item Short Form Health Survey (feeling downhearted, doing activities less carefully, or accomplishing less because of emotional problems); younger age; lower educational achievement; and recent financial crisis.
“The clinical assessment of suicide risk typically focuses on acute suicidal symptoms, together with depression, anxiety, substance misuse, and recent stressful events,” coinvestigator Mark Olfson, MD, PhD, professor of epidemiology, Columbia University Irving Medical Center, New York, said in an interview.
Dr. Olfson said.
Extra vigilance
Commenting on the study in an interview, April C. Foreman, PhD, an executive board member of the American Association of Suicidology, noted that some of the findings were not surprising.
“When discharging a patient from inpatient care, or seeing them in primary care, bring up mental health concerns proactively and ask whether they have ever attempted suicide or harmed themselves – even a long time ago – just as you ask about a family history of heart disease or cancer, or other health issues,” said Dr. Foreman, chief medical officer of the Kevin and Margaret Hines Foundation.
She noted that half of people who die by suicide have a primary care visit within the preceding month.
“Primary care is a great place to get a suicide history and follow the patient with extra vigilance, just as you would with any other risk factors,” Dr. Foreman said.
The study was funded by the National Institute on Alcohol Abuse and Alcoholism and its Intramural Program. The study authors and Dr. Foreman have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A history of suicidal behaviors or ideation, functional impairment related to mental health disorders, and socioeconomic disadvantage are the three most important risk factors predicting subsequent suicide attempts, new research suggests.
Investigators applied a machine-learning model to data on over 34,500 adults drawn from a large national survey database. After analyzing more than 2,500 survey questions, key areas were identified that yielded the most accurate predictions of who might be at risk for later suicide attempt.
These predictors included experiencing previous suicidal behaviors and ideation or functional impairment because of emotional problems, being at a younger age, having a lower educational achievement, and experiencing a recent financial crisis.
“Our machine learning model confirmed well-known risk factors of suicide attempt, including previous suicidal behavior and depression; and we also identified functional impairment, such as doing activities less carefully or accomplishing less because of emotional problems, as a new important risk,” lead author Angel Garcia de la Garza, PhD candidate in the department of biostatistics, Columbia University, New York, said in an interview.
“We hope our results provide a novel avenue for future suicide risk assessment,” Mr. Garcia de la Garza said.
The findings were published online Jan. 6 in JAMA Psychiatry.
‘Rich’ dataset
Previous research using machine learning approaches to study nonfatal suicide attempt prediction has focused on high-risk patients in clinical treatment. However, more than one-third of individuals making nonfatal suicide attempts do not receive mental health treatment, Mr. Garcia de la Garza noted.
To gain further insight into predictors of suicide risk in nonclinical populations, the researchers turned to the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), a longitudinal survey of noninstitutionalized U.S. adults.
“We wanted to extend our understanding of suicide attempt risk factors beyond high-risk clinical populations to the general adult population; and the richness of the NESARC dataset provides a unique opportunity to do so,” Mr. Garcia de la Garza said.
The NESARC surveys were conducted in two waves: Wave 1 (2001-2002) and wave 2 (2004-2005), in which participants self-reported nonfatal suicide attempts in the preceding 3 years since wave 1.
Assessment of wave 1 participants was based on the Alcohol Use Disorder and Associated Disabilities Interview Schedule DSM-IV.
“This survey’s extensive assessment instrument contained a detailed evaluation of substance use, psychiatric disorders, and symptoms not routinely available in electronic health records,” Mr. Garcia de la Garza noted.
The wave 1 survey contained 2,805 separate questions. From participants’ responses, the investigators derived 180 variables for three categories: past-year, prior-to-past-year, and lifetime mental disorders.
They then identified 2,978 factors associated with suicide attempts and used a statistical method called balanced random forest to classify suicide attempts at wave 2. Each variable was accorded an “importance score” using identified wave 1 features.
The outcome variable of attempted suicide at any point during the 3 years prior to the wave 2 interview was defined by combining responses to three wave 2 questions:
- In your entire life, did you ever attempt suicide?
- If yes, how old were you the first time?
- If the most recent event occurred within the last 3 years, how old were you during the most recent time?
Suicide risk severity was classified into four groups (low, medium, high, and very high) on the basis of the top-performing risk factors.
A statistical model combining survey design and nonresponse weights enabled estimates to be representative of the U.S. population, based on the 2000 census.
Out-of-fold model prediction assessed performance of the model, using area under receiver operator curve (AUC), sensitivity, and specificity.
Daily functioning
Of all participants, 70.2% (n = 34,653; almost 60% women) completed wave 2 interviews. The weighted mean ages at waves 1 and 2 were 45.1 and 48.2 years, respectively.
Of wave 2 respondents, 0.6% (n = 222) attempted suicide during the preceding 3 years.
Half of those who attempted suicide within the first year were classified as “very high risk,” while 33.2% of those who attempted suicide between the first and second year and 33.3% of those who attempted suicide between the second and third year were classified as “very high risk.”
Among participants who attempted suicide between the third year and follow-up, 16.48% were classified as “very high risk.”
The model accurately captured classification of participants, even across demographic characteristics, such as age, sex, race, and income.
Younger individuals (aged 18-36 years) were at higher risk, compared with older individuals. In addition, women were at higher risk than were men, White participants were at higher risk than were non-White participants, and individuals with lower income were at greater risk than were those with higher income.
The model found that 1.8% of the U.S. population had a 10% or greater risk of a suicide attempt.
The most important risk factors identified were the three questions about previous suicidal ideation or behavior; three items from the 12-Item Short Form Health Survey (feeling downhearted, doing activities less carefully, or accomplishing less because of emotional problems); younger age; lower educational achievement; and recent financial crisis.
“The clinical assessment of suicide risk typically focuses on acute suicidal symptoms, together with depression, anxiety, substance misuse, and recent stressful events,” coinvestigator Mark Olfson, MD, PhD, professor of epidemiology, Columbia University Irving Medical Center, New York, said in an interview.
Dr. Olfson said.
Extra vigilance
Commenting on the study in an interview, April C. Foreman, PhD, an executive board member of the American Association of Suicidology, noted that some of the findings were not surprising.
“When discharging a patient from inpatient care, or seeing them in primary care, bring up mental health concerns proactively and ask whether they have ever attempted suicide or harmed themselves – even a long time ago – just as you ask about a family history of heart disease or cancer, or other health issues,” said Dr. Foreman, chief medical officer of the Kevin and Margaret Hines Foundation.
She noted that half of people who die by suicide have a primary care visit within the preceding month.
“Primary care is a great place to get a suicide history and follow the patient with extra vigilance, just as you would with any other risk factors,” Dr. Foreman said.
The study was funded by the National Institute on Alcohol Abuse and Alcoholism and its Intramural Program. The study authors and Dr. Foreman have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Constipation: A potentially serious adverse effect of clozapine that’s often overlooked
Clozapine is the most effective second-generation antipsychotic for the treatment of refractory schizophrenia. It can reduce delusions and hallucinations in patients who are unresponsive to other antipsychotic medications. Further, clozapine is the only agent known to reduce suicidal urges.1
Unfortunately, clozapine is associated with numerous adverse effects, most notably agranulocytosis, a rare but potentially fatal adverse effect that occurs in approximately 1% to 2% of patients during the first year of treatment.2 Other adverse effects associated with clozapine are weight gain, sedation, orthostatic hypotension, sialorrhea, constipation, hyperglycemia, hyperlipidemia, myocarditis, and seizures. Among these adverse effects, constipation, which can progress to life-threatening gastrointestinal (GI) hypomotility and ileus, is often overlooked. Up to 60% of patients who are administered clozapine experience constipation.3 A recent review found that potentially life-threatening clozapine-induced ileus occurred in approximately 3 per 1,000 patients, and 28 deaths have been documented.4
In this case report, I describe a patient who received clozapine and experienced constipation that led to an intestinal obstruction. I discuss the importance of prompt diagnosis and treatment approaches to prevent severe constipation in patients who are prescribed clozapine.
CASE REPORT
Mr. L, age 24, has schizophrenia, depression, mild intellectual disability, and congenital human immunodeficiency virus (HIV). He has had multiple unsuccessful antipsychotic trials but is compliant with highly active antiretroviral therapy for HIV. After experiencing worsening aggressive behavior for a third time, Mr. L was involuntarily committed to our Crises Response Center.
Mr. L was admitted to the acute inpatient psychiatry unit. He reported having auditory hallucinations, which included whispering sounds with intermittent music, mostly at night. He also reported decreased sleep, poor appetite, and low energy, but denied feelings of depression or mania.
During the mental status examination, Mr. L was calm and cooperative, but easily distracted. He said he smoked cigarettes but denied any current alcohol or illicit drug use. Mr. L’s urine drug screen was negative.
External medication records showed Mr. L had been prescribed haloperidol, risperidone, chlorpromazine, olanzapine, aripiprazole, quetiapine, bupropion, sodium valproate, and topiramate, for the treatment of schizophrenia, with no significant improvement.
Continue to: On hospital Day 3, Mr. L...
On hospital Day 3, Mr. L was started on clozapine, 12.5 mg at bedtime, and titrated to 300 mg by Day 15. The clozapine was titrated slowly; initially the dose was doubled every 2 days up to 100 mg every night at bedtime, then it was increased by 50 mg every 2 to 3 days up to 300 mg every night at bedtime. A baseline complete blood count with differential confirmed that his absolute neutrophil count (ANC) was >1,500 µL, which is above the reference range. Mr. L was closely monitored for agranulocytosis and had weekly blood work for ANC. Additionally, his information was updated regularly on the Clozapine Risk Evaluation and Mitigation Strategy website.
After Mr. L began the clozapine regimen, he had reduced mood lability, paranoia, and delusions; significantly improved auditory and visual hallucinations; and reduced distress. His sleep was improved, and he appeared pleasant with clear sensorium. During this period, Mr. L developed sialorrhea and was administered glycopyrrolate and prescribed diphenhydramine, as needed for sleep. Although he had been prescribed oral benztropine for extrapyramidal side effects prophylaxis, this medication was never administered to him during his stay in the hospital. He became stable on this regimen, and the treatment team started working on his discharge.
On hospital Day 20, Mr. L complained about abdominal pain. At first, the pain was localized to right upper quadrant; later, he had diffuse abdominal pain with distension. He reported that he had no bowel movement for 1 day. The treatment team instructed him to take nothing by mouth, and all antipsychotic and anticholinergic medications were held. Given Mr. L’s HIV status, the treatment team ordered liver function tests (LFTs) and an abdominal x-ray. Mr. L’s LFT results were normal and the x-ray findings were inconclusive. However, a CT scan of the abdomen showed an obstruction due to a 3.5-cm stoolball in the proximal transverse colon with fecal impaction. Mr. L was started on a saline enema, which resulted in him having 2 to 3 episodes of watery diarrhea, and his abdominal pain resolved.
Although Mr. L reported feeling better and started eating again, there were concerns about his watery bowel movement, so a repeat abdominal x-ray was ordered. The x-ray confirmed that Mr. L had a persistent bowel obstruction. Mr. L’s abdominal pain returned. At this time, the pain was diffuse and severe, and Mr. L was vomiting. Mr. L was started on a bisacodyl suppository immediately, and then twice daily as needed. Subsequently, Mr. L had a solid bowel movement and relief of all GI symptoms. Mr. L was administered docusate sodium twice daily. Repeated x-rays of the abdomen confirmed the obstructive changes of the small bowel had resolved.
Why constipation may be overlooked
Although constipation is a common adverse effect of many psychotropic medications, when it emerges during clozapine therapy, it can lead to ileus, which can be fatal. Mr. L’s case highlighted that clozapine use can cause intestinal obstruction, a condition that can deteriorate within a few hours to life-threatening ileus. The extent of fecal impaction can be masked by spurious diarrhea, as illustrated in Mr. L’s case.5 Clozapine has anti-serotonergic properties (5HT-2A antagonist) that may result in reduced intestinal nociception pain. This discrepancy between physical symptoms and the severity of illness may cause delays in diagnosis.4 As soon as the treatment team determined Mr. L was constipated, all medications with anticholinergic effects were held. Patients also may have difficulty reporting intestinal pain due to psychotic symptoms such as paranoia or thought disorder.6
Take steps to prevent constipation
To prevent constipation in patients receiving clozapine, minimize the use of systemic anticholinergic agents because of the adverse effects of this interaction. For example, in Mr. L’s case, he received both clozapine and glycopyrrolate. In addition, all patients who are prescribed clozapine should receive docusate sodium to prevent constipation. However, because docusate sodium alone is usually not sufficient, consider adding another agent. Osmotic laxatives, such as polyethylene glycol 3350, are suitable additional agents. If this combination does not work, then consider senna glycoside or bisacodyl, which will increase intestinal motility and help with the flow of water into the bowel, thereby improving constipation. Bulk agents should be avoided because they can make constipation worse, especially if the patient is not drinking enough water, which is often the case with patients who have psychosis.7
Ask patients about GI symptoms
Clinicians need to observe and monitor patients who receive clozapine for signs of constipation, including the frequency and difficulty of defecation during treatment.4 It is important to ask patients about bowel function. Before starting treatment with clozapine, discuss the risks of clozapine-induced intestinal obstruction with patients and caregivers, and encourage them to report any GI symptoms. Also, provide dietary advice and recommend the as-needed use of laxatives.
1. Patchan KM, Richardson C, Vyas G, et al. The risk of suicide after clozapine discontinuation: cause for concern. Ann Clinical Psychiatry. 2015;27(4):253-256.
2. Alvir JM, Lieberman JA, Safferman AZ, et al. Clozapine-induced agranulocytosis. Incidence and risk factors in the United States. N Engl J Med. 1993;329(3):162-167.
3. Hayes G, Gibler B. Clozapine-induced constipation. Am J Psychiatry. 1995;152(2):298.
4. Palmer SE, McLean RM, Ellis PM, et al. Life-threatening clozapine-induced gastrointestinal hypomotility: an analysis of 102 cases. J Clin Psychiatry. 2008;69(5):759-768.
5. Drew L, Herdson P. Clozapine and constipation: a serious issue. Aust N Z J Psychiatry. 1997; 31(1):149-150.
6. Bickerstaff LK, Harris SC, Leggett RS, et al. Pain insensitivity in schizophrenic patients: a surgical dilemma. Arch Surg. 1988;123(1):49-51.
7. Psychopharmacology Institute. How to manage adverse effects of clozapine – Part 1. Updated June 3, 2020. Accessed December 7, 2020. https://psychopharmacologyinstitute.com/publication/how-to-manage-adverse-effects-of-clozapine-part-1-2476
Clozapine is the most effective second-generation antipsychotic for the treatment of refractory schizophrenia. It can reduce delusions and hallucinations in patients who are unresponsive to other antipsychotic medications. Further, clozapine is the only agent known to reduce suicidal urges.1
Unfortunately, clozapine is associated with numerous adverse effects, most notably agranulocytosis, a rare but potentially fatal adverse effect that occurs in approximately 1% to 2% of patients during the first year of treatment.2 Other adverse effects associated with clozapine are weight gain, sedation, orthostatic hypotension, sialorrhea, constipation, hyperglycemia, hyperlipidemia, myocarditis, and seizures. Among these adverse effects, constipation, which can progress to life-threatening gastrointestinal (GI) hypomotility and ileus, is often overlooked. Up to 60% of patients who are administered clozapine experience constipation.3 A recent review found that potentially life-threatening clozapine-induced ileus occurred in approximately 3 per 1,000 patients, and 28 deaths have been documented.4
In this case report, I describe a patient who received clozapine and experienced constipation that led to an intestinal obstruction. I discuss the importance of prompt diagnosis and treatment approaches to prevent severe constipation in patients who are prescribed clozapine.
CASE REPORT
Mr. L, age 24, has schizophrenia, depression, mild intellectual disability, and congenital human immunodeficiency virus (HIV). He has had multiple unsuccessful antipsychotic trials but is compliant with highly active antiretroviral therapy for HIV. After experiencing worsening aggressive behavior for a third time, Mr. L was involuntarily committed to our Crises Response Center.
Mr. L was admitted to the acute inpatient psychiatry unit. He reported having auditory hallucinations, which included whispering sounds with intermittent music, mostly at night. He also reported decreased sleep, poor appetite, and low energy, but denied feelings of depression or mania.
During the mental status examination, Mr. L was calm and cooperative, but easily distracted. He said he smoked cigarettes but denied any current alcohol or illicit drug use. Mr. L’s urine drug screen was negative.
External medication records showed Mr. L had been prescribed haloperidol, risperidone, chlorpromazine, olanzapine, aripiprazole, quetiapine, bupropion, sodium valproate, and topiramate, for the treatment of schizophrenia, with no significant improvement.
Continue to: On hospital Day 3, Mr. L...
On hospital Day 3, Mr. L was started on clozapine, 12.5 mg at bedtime, and titrated to 300 mg by Day 15. The clozapine was titrated slowly; initially the dose was doubled every 2 days up to 100 mg every night at bedtime, then it was increased by 50 mg every 2 to 3 days up to 300 mg every night at bedtime. A baseline complete blood count with differential confirmed that his absolute neutrophil count (ANC) was >1,500 µL, which is above the reference range. Mr. L was closely monitored for agranulocytosis and had weekly blood work for ANC. Additionally, his information was updated regularly on the Clozapine Risk Evaluation and Mitigation Strategy website.
After Mr. L began the clozapine regimen, he had reduced mood lability, paranoia, and delusions; significantly improved auditory and visual hallucinations; and reduced distress. His sleep was improved, and he appeared pleasant with clear sensorium. During this period, Mr. L developed sialorrhea and was administered glycopyrrolate and prescribed diphenhydramine, as needed for sleep. Although he had been prescribed oral benztropine for extrapyramidal side effects prophylaxis, this medication was never administered to him during his stay in the hospital. He became stable on this regimen, and the treatment team started working on his discharge.
On hospital Day 20, Mr. L complained about abdominal pain. At first, the pain was localized to right upper quadrant; later, he had diffuse abdominal pain with distension. He reported that he had no bowel movement for 1 day. The treatment team instructed him to take nothing by mouth, and all antipsychotic and anticholinergic medications were held. Given Mr. L’s HIV status, the treatment team ordered liver function tests (LFTs) and an abdominal x-ray. Mr. L’s LFT results were normal and the x-ray findings were inconclusive. However, a CT scan of the abdomen showed an obstruction due to a 3.5-cm stoolball in the proximal transverse colon with fecal impaction. Mr. L was started on a saline enema, which resulted in him having 2 to 3 episodes of watery diarrhea, and his abdominal pain resolved.
Although Mr. L reported feeling better and started eating again, there were concerns about his watery bowel movement, so a repeat abdominal x-ray was ordered. The x-ray confirmed that Mr. L had a persistent bowel obstruction. Mr. L’s abdominal pain returned. At this time, the pain was diffuse and severe, and Mr. L was vomiting. Mr. L was started on a bisacodyl suppository immediately, and then twice daily as needed. Subsequently, Mr. L had a solid bowel movement and relief of all GI symptoms. Mr. L was administered docusate sodium twice daily. Repeated x-rays of the abdomen confirmed the obstructive changes of the small bowel had resolved.
Why constipation may be overlooked
Although constipation is a common adverse effect of many psychotropic medications, when it emerges during clozapine therapy, it can lead to ileus, which can be fatal. Mr. L’s case highlighted that clozapine use can cause intestinal obstruction, a condition that can deteriorate within a few hours to life-threatening ileus. The extent of fecal impaction can be masked by spurious diarrhea, as illustrated in Mr. L’s case.5 Clozapine has anti-serotonergic properties (5HT-2A antagonist) that may result in reduced intestinal nociception pain. This discrepancy between physical symptoms and the severity of illness may cause delays in diagnosis.4 As soon as the treatment team determined Mr. L was constipated, all medications with anticholinergic effects were held. Patients also may have difficulty reporting intestinal pain due to psychotic symptoms such as paranoia or thought disorder.6
Take steps to prevent constipation
To prevent constipation in patients receiving clozapine, minimize the use of systemic anticholinergic agents because of the adverse effects of this interaction. For example, in Mr. L’s case, he received both clozapine and glycopyrrolate. In addition, all patients who are prescribed clozapine should receive docusate sodium to prevent constipation. However, because docusate sodium alone is usually not sufficient, consider adding another agent. Osmotic laxatives, such as polyethylene glycol 3350, are suitable additional agents. If this combination does not work, then consider senna glycoside or bisacodyl, which will increase intestinal motility and help with the flow of water into the bowel, thereby improving constipation. Bulk agents should be avoided because they can make constipation worse, especially if the patient is not drinking enough water, which is often the case with patients who have psychosis.7
Ask patients about GI symptoms
Clinicians need to observe and monitor patients who receive clozapine for signs of constipation, including the frequency and difficulty of defecation during treatment.4 It is important to ask patients about bowel function. Before starting treatment with clozapine, discuss the risks of clozapine-induced intestinal obstruction with patients and caregivers, and encourage them to report any GI symptoms. Also, provide dietary advice and recommend the as-needed use of laxatives.
Clozapine is the most effective second-generation antipsychotic for the treatment of refractory schizophrenia. It can reduce delusions and hallucinations in patients who are unresponsive to other antipsychotic medications. Further, clozapine is the only agent known to reduce suicidal urges.1
Unfortunately, clozapine is associated with numerous adverse effects, most notably agranulocytosis, a rare but potentially fatal adverse effect that occurs in approximately 1% to 2% of patients during the first year of treatment.2 Other adverse effects associated with clozapine are weight gain, sedation, orthostatic hypotension, sialorrhea, constipation, hyperglycemia, hyperlipidemia, myocarditis, and seizures. Among these adverse effects, constipation, which can progress to life-threatening gastrointestinal (GI) hypomotility and ileus, is often overlooked. Up to 60% of patients who are administered clozapine experience constipation.3 A recent review found that potentially life-threatening clozapine-induced ileus occurred in approximately 3 per 1,000 patients, and 28 deaths have been documented.4
In this case report, I describe a patient who received clozapine and experienced constipation that led to an intestinal obstruction. I discuss the importance of prompt diagnosis and treatment approaches to prevent severe constipation in patients who are prescribed clozapine.
CASE REPORT
Mr. L, age 24, has schizophrenia, depression, mild intellectual disability, and congenital human immunodeficiency virus (HIV). He has had multiple unsuccessful antipsychotic trials but is compliant with highly active antiretroviral therapy for HIV. After experiencing worsening aggressive behavior for a third time, Mr. L was involuntarily committed to our Crises Response Center.
Mr. L was admitted to the acute inpatient psychiatry unit. He reported having auditory hallucinations, which included whispering sounds with intermittent music, mostly at night. He also reported decreased sleep, poor appetite, and low energy, but denied feelings of depression or mania.
During the mental status examination, Mr. L was calm and cooperative, but easily distracted. He said he smoked cigarettes but denied any current alcohol or illicit drug use. Mr. L’s urine drug screen was negative.
External medication records showed Mr. L had been prescribed haloperidol, risperidone, chlorpromazine, olanzapine, aripiprazole, quetiapine, bupropion, sodium valproate, and topiramate, for the treatment of schizophrenia, with no significant improvement.
Continue to: On hospital Day 3, Mr. L...
On hospital Day 3, Mr. L was started on clozapine, 12.5 mg at bedtime, and titrated to 300 mg by Day 15. The clozapine was titrated slowly; initially the dose was doubled every 2 days up to 100 mg every night at bedtime, then it was increased by 50 mg every 2 to 3 days up to 300 mg every night at bedtime. A baseline complete blood count with differential confirmed that his absolute neutrophil count (ANC) was >1,500 µL, which is above the reference range. Mr. L was closely monitored for agranulocytosis and had weekly blood work for ANC. Additionally, his information was updated regularly on the Clozapine Risk Evaluation and Mitigation Strategy website.
After Mr. L began the clozapine regimen, he had reduced mood lability, paranoia, and delusions; significantly improved auditory and visual hallucinations; and reduced distress. His sleep was improved, and he appeared pleasant with clear sensorium. During this period, Mr. L developed sialorrhea and was administered glycopyrrolate and prescribed diphenhydramine, as needed for sleep. Although he had been prescribed oral benztropine for extrapyramidal side effects prophylaxis, this medication was never administered to him during his stay in the hospital. He became stable on this regimen, and the treatment team started working on his discharge.
On hospital Day 20, Mr. L complained about abdominal pain. At first, the pain was localized to right upper quadrant; later, he had diffuse abdominal pain with distension. He reported that he had no bowel movement for 1 day. The treatment team instructed him to take nothing by mouth, and all antipsychotic and anticholinergic medications were held. Given Mr. L’s HIV status, the treatment team ordered liver function tests (LFTs) and an abdominal x-ray. Mr. L’s LFT results were normal and the x-ray findings were inconclusive. However, a CT scan of the abdomen showed an obstruction due to a 3.5-cm stoolball in the proximal transverse colon with fecal impaction. Mr. L was started on a saline enema, which resulted in him having 2 to 3 episodes of watery diarrhea, and his abdominal pain resolved.
Although Mr. L reported feeling better and started eating again, there were concerns about his watery bowel movement, so a repeat abdominal x-ray was ordered. The x-ray confirmed that Mr. L had a persistent bowel obstruction. Mr. L’s abdominal pain returned. At this time, the pain was diffuse and severe, and Mr. L was vomiting. Mr. L was started on a bisacodyl suppository immediately, and then twice daily as needed. Subsequently, Mr. L had a solid bowel movement and relief of all GI symptoms. Mr. L was administered docusate sodium twice daily. Repeated x-rays of the abdomen confirmed the obstructive changes of the small bowel had resolved.
Why constipation may be overlooked
Although constipation is a common adverse effect of many psychotropic medications, when it emerges during clozapine therapy, it can lead to ileus, which can be fatal. Mr. L’s case highlighted that clozapine use can cause intestinal obstruction, a condition that can deteriorate within a few hours to life-threatening ileus. The extent of fecal impaction can be masked by spurious diarrhea, as illustrated in Mr. L’s case.5 Clozapine has anti-serotonergic properties (5HT-2A antagonist) that may result in reduced intestinal nociception pain. This discrepancy between physical symptoms and the severity of illness may cause delays in diagnosis.4 As soon as the treatment team determined Mr. L was constipated, all medications with anticholinergic effects were held. Patients also may have difficulty reporting intestinal pain due to psychotic symptoms such as paranoia or thought disorder.6
Take steps to prevent constipation
To prevent constipation in patients receiving clozapine, minimize the use of systemic anticholinergic agents because of the adverse effects of this interaction. For example, in Mr. L’s case, he received both clozapine and glycopyrrolate. In addition, all patients who are prescribed clozapine should receive docusate sodium to prevent constipation. However, because docusate sodium alone is usually not sufficient, consider adding another agent. Osmotic laxatives, such as polyethylene glycol 3350, are suitable additional agents. If this combination does not work, then consider senna glycoside or bisacodyl, which will increase intestinal motility and help with the flow of water into the bowel, thereby improving constipation. Bulk agents should be avoided because they can make constipation worse, especially if the patient is not drinking enough water, which is often the case with patients who have psychosis.7
Ask patients about GI symptoms
Clinicians need to observe and monitor patients who receive clozapine for signs of constipation, including the frequency and difficulty of defecation during treatment.4 It is important to ask patients about bowel function. Before starting treatment with clozapine, discuss the risks of clozapine-induced intestinal obstruction with patients and caregivers, and encourage them to report any GI symptoms. Also, provide dietary advice and recommend the as-needed use of laxatives.
1. Patchan KM, Richardson C, Vyas G, et al. The risk of suicide after clozapine discontinuation: cause for concern. Ann Clinical Psychiatry. 2015;27(4):253-256.
2. Alvir JM, Lieberman JA, Safferman AZ, et al. Clozapine-induced agranulocytosis. Incidence and risk factors in the United States. N Engl J Med. 1993;329(3):162-167.
3. Hayes G, Gibler B. Clozapine-induced constipation. Am J Psychiatry. 1995;152(2):298.
4. Palmer SE, McLean RM, Ellis PM, et al. Life-threatening clozapine-induced gastrointestinal hypomotility: an analysis of 102 cases. J Clin Psychiatry. 2008;69(5):759-768.
5. Drew L, Herdson P. Clozapine and constipation: a serious issue. Aust N Z J Psychiatry. 1997; 31(1):149-150.
6. Bickerstaff LK, Harris SC, Leggett RS, et al. Pain insensitivity in schizophrenic patients: a surgical dilemma. Arch Surg. 1988;123(1):49-51.
7. Psychopharmacology Institute. How to manage adverse effects of clozapine – Part 1. Updated June 3, 2020. Accessed December 7, 2020. https://psychopharmacologyinstitute.com/publication/how-to-manage-adverse-effects-of-clozapine-part-1-2476
1. Patchan KM, Richardson C, Vyas G, et al. The risk of suicide after clozapine discontinuation: cause for concern. Ann Clinical Psychiatry. 2015;27(4):253-256.
2. Alvir JM, Lieberman JA, Safferman AZ, et al. Clozapine-induced agranulocytosis. Incidence and risk factors in the United States. N Engl J Med. 1993;329(3):162-167.
3. Hayes G, Gibler B. Clozapine-induced constipation. Am J Psychiatry. 1995;152(2):298.
4. Palmer SE, McLean RM, Ellis PM, et al. Life-threatening clozapine-induced gastrointestinal hypomotility: an analysis of 102 cases. J Clin Psychiatry. 2008;69(5):759-768.
5. Drew L, Herdson P. Clozapine and constipation: a serious issue. Aust N Z J Psychiatry. 1997; 31(1):149-150.
6. Bickerstaff LK, Harris SC, Leggett RS, et al. Pain insensitivity in schizophrenic patients: a surgical dilemma. Arch Surg. 1988;123(1):49-51.
7. Psychopharmacology Institute. How to manage adverse effects of clozapine – Part 1. Updated June 3, 2020. Accessed December 7, 2020. https://psychopharmacologyinstitute.com/publication/how-to-manage-adverse-effects-of-clozapine-part-1-2476
Give psych patients the COVID vaccination now, experts say
With COVID-19 vaccinations now underway, mental health experts around the world continue to push for patients with serious mental illness (SMI) to be considered a high-priority group for the vaccine.
Research shows that patients with SMI are at increased risk of being infected with SARS-CoV-2 and have higher rates of hospitalization and poor outcomes, Nicola Warren, MBBS, University of Queensland, Brisbane, Australia, and coauthors write in a viewpoint published online Dec. 15 in JAMA Psychiatry
Factors behind the worse outcomes in individuals with SMI include concomitant medications, poorer premorbid general health, physical comorbidity, reduced access to medical care, and environmental and lifestyle factors such as lower socioeconomic status, overcrowding, smoking, and obesity.
“In light of these vulnerabilities, it is important that people with SMI are a priority group to receive a vaccination,” Dr. Warren and colleagues say.
Yet there are challenges at the individual and public health level in getting people with SMI vaccinated against COVID-19, they point out.
Challenges at the individual level include getting people with SMI to recognize the importance of the vaccine and combating negative beliefs about safety and misconceptions that the vaccine itself can make them sick with COVID-19.
Mental health professionals are “uniquely skilled” to deliver vaccine education, “being able to adapt for those with communication difficulties and balance factors influencing decision-making,” Dr. Warren and colleagues write.
, like getting to a vaccination clinic.
Research has shown that running vaccination clinics parallel to mental health services can boost vaccination rates by 25%, the authors note. Therefore, one solution may be to embed vaccination clinics within mental health services, Dr. Warren and colleagues suggest.
Join the chorus
Plans and policies to ensure rapid delivery of the COVID-19 vaccine are “vital,” they conclude. “Mental health clinicians have a key role in advocating for priority access to a COVID-19 vaccination for those with SMI, as well as facilitating its uptake,” they add.
Dr. Warren and her colleagues join a chorus of other mental health care providers who have sounded the alarm on the risks of COVID-19 for patients with SMI and the need to get them vaccinated early.
In a perspective article published last month in World Psychiatry, Marc De Hert, MD, PhD, professor of psychiatry at KU Leuven (Belgium), and coauthors called for individuals with SMI to have priority status for any COVID-19 vaccine, as reported by this news organization.
Dr. De Hert and colleagues noted that there is an ethical duty to prioritize vaccination for people with SMI given their increased risk of worse outcomes following COVID-19 infection and the structural barriers faced by people with SMI in accessing a vaccine.
Joining the chorus, Benjamin Druss, MD, MPH, from Emory University, Atlanta, Georgia, warned in a JAMA Psychiatry viewpoint in April that the COVID-19 pandemic represents a looming crisis for patients with SMI and the health care systems that serve them.
“Careful planning and execution at multiple levels will be essential for minimizing the adverse outcomes of this pandemic for this vulnerable population,” Dr. Druss wrote.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
With COVID-19 vaccinations now underway, mental health experts around the world continue to push for patients with serious mental illness (SMI) to be considered a high-priority group for the vaccine.
Research shows that patients with SMI are at increased risk of being infected with SARS-CoV-2 and have higher rates of hospitalization and poor outcomes, Nicola Warren, MBBS, University of Queensland, Brisbane, Australia, and coauthors write in a viewpoint published online Dec. 15 in JAMA Psychiatry
Factors behind the worse outcomes in individuals with SMI include concomitant medications, poorer premorbid general health, physical comorbidity, reduced access to medical care, and environmental and lifestyle factors such as lower socioeconomic status, overcrowding, smoking, and obesity.
“In light of these vulnerabilities, it is important that people with SMI are a priority group to receive a vaccination,” Dr. Warren and colleagues say.
Yet there are challenges at the individual and public health level in getting people with SMI vaccinated against COVID-19, they point out.
Challenges at the individual level include getting people with SMI to recognize the importance of the vaccine and combating negative beliefs about safety and misconceptions that the vaccine itself can make them sick with COVID-19.
Mental health professionals are “uniquely skilled” to deliver vaccine education, “being able to adapt for those with communication difficulties and balance factors influencing decision-making,” Dr. Warren and colleagues write.
, like getting to a vaccination clinic.
Research has shown that running vaccination clinics parallel to mental health services can boost vaccination rates by 25%, the authors note. Therefore, one solution may be to embed vaccination clinics within mental health services, Dr. Warren and colleagues suggest.
Join the chorus
Plans and policies to ensure rapid delivery of the COVID-19 vaccine are “vital,” they conclude. “Mental health clinicians have a key role in advocating for priority access to a COVID-19 vaccination for those with SMI, as well as facilitating its uptake,” they add.
Dr. Warren and her colleagues join a chorus of other mental health care providers who have sounded the alarm on the risks of COVID-19 for patients with SMI and the need to get them vaccinated early.
In a perspective article published last month in World Psychiatry, Marc De Hert, MD, PhD, professor of psychiatry at KU Leuven (Belgium), and coauthors called for individuals with SMI to have priority status for any COVID-19 vaccine, as reported by this news organization.
Dr. De Hert and colleagues noted that there is an ethical duty to prioritize vaccination for people with SMI given their increased risk of worse outcomes following COVID-19 infection and the structural barriers faced by people with SMI in accessing a vaccine.
Joining the chorus, Benjamin Druss, MD, MPH, from Emory University, Atlanta, Georgia, warned in a JAMA Psychiatry viewpoint in April that the COVID-19 pandemic represents a looming crisis for patients with SMI and the health care systems that serve them.
“Careful planning and execution at multiple levels will be essential for minimizing the adverse outcomes of this pandemic for this vulnerable population,” Dr. Druss wrote.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
With COVID-19 vaccinations now underway, mental health experts around the world continue to push for patients with serious mental illness (SMI) to be considered a high-priority group for the vaccine.
Research shows that patients with SMI are at increased risk of being infected with SARS-CoV-2 and have higher rates of hospitalization and poor outcomes, Nicola Warren, MBBS, University of Queensland, Brisbane, Australia, and coauthors write in a viewpoint published online Dec. 15 in JAMA Psychiatry
Factors behind the worse outcomes in individuals with SMI include concomitant medications, poorer premorbid general health, physical comorbidity, reduced access to medical care, and environmental and lifestyle factors such as lower socioeconomic status, overcrowding, smoking, and obesity.
“In light of these vulnerabilities, it is important that people with SMI are a priority group to receive a vaccination,” Dr. Warren and colleagues say.
Yet there are challenges at the individual and public health level in getting people with SMI vaccinated against COVID-19, they point out.
Challenges at the individual level include getting people with SMI to recognize the importance of the vaccine and combating negative beliefs about safety and misconceptions that the vaccine itself can make them sick with COVID-19.
Mental health professionals are “uniquely skilled” to deliver vaccine education, “being able to adapt for those with communication difficulties and balance factors influencing decision-making,” Dr. Warren and colleagues write.
, like getting to a vaccination clinic.
Research has shown that running vaccination clinics parallel to mental health services can boost vaccination rates by 25%, the authors note. Therefore, one solution may be to embed vaccination clinics within mental health services, Dr. Warren and colleagues suggest.
Join the chorus
Plans and policies to ensure rapid delivery of the COVID-19 vaccine are “vital,” they conclude. “Mental health clinicians have a key role in advocating for priority access to a COVID-19 vaccination for those with SMI, as well as facilitating its uptake,” they add.
Dr. Warren and her colleagues join a chorus of other mental health care providers who have sounded the alarm on the risks of COVID-19 for patients with SMI and the need to get them vaccinated early.
In a perspective article published last month in World Psychiatry, Marc De Hert, MD, PhD, professor of psychiatry at KU Leuven (Belgium), and coauthors called for individuals with SMI to have priority status for any COVID-19 vaccine, as reported by this news organization.
Dr. De Hert and colleagues noted that there is an ethical duty to prioritize vaccination for people with SMI given their increased risk of worse outcomes following COVID-19 infection and the structural barriers faced by people with SMI in accessing a vaccine.
Joining the chorus, Benjamin Druss, MD, MPH, from Emory University, Atlanta, Georgia, warned in a JAMA Psychiatry viewpoint in April that the COVID-19 pandemic represents a looming crisis for patients with SMI and the health care systems that serve them.
“Careful planning and execution at multiple levels will be essential for minimizing the adverse outcomes of this pandemic for this vulnerable population,” Dr. Druss wrote.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19: Hand sanitizer poisonings soar, psych patients at high risk
Cases of poisoning – intentional and unintentional – from ingestion of alcohol-based hand sanitizer have soared during the COVID-19 pandemic.
In the United Kingdom alone, alcohol-based hand sanitizer poisonings reported to the National Poisons Information Service jumped 157% – from 155 between January 1 and September 16, 2019, to 398 between Jan. 1 and Sept. 14, 2020, new research shows.
More needs to be done to protect those at risk of unintentional and intentional swallowing of alcohol-based hand sanitizer, including children, people with dementia/confusion, and those with mental health issues, according to Georgia Richards, DPhil student, Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences, University of Oxford (England).
“If providers are supplying alcohol-based hand sanitizers in the community to reduce the spread of SARS-CoV-2, Ms. Richards said in an interview.
The study was published online Dec. 1 in BMJ Evidence-Based Medicine.
European, U.S. poisoning rates soar
In the paper Ms. Richards described two deaths that occurred in hospitals in England.
In one case, a 30-year-old woman, detained in a psychiatric unit who received the antidepressant venlafaxine was found dead in her hospital bed with a container of hand-sanitizing gel beside her.
“The gel was readily accessible to patients on the ward from a communal dispenser, and patients were allowed to fill cups or other containers with it to keep in their rooms,” Ms. Richards reported.
A postmortem analysis found a high level of alcohol in her blood (214 mg of alcohol in 100 mL of blood). The medical cause of death was listed as “ingestion of alcohol and venlafaxine.” The coroner concluded that the combination of these substances suppressed the patient’s breathing, leading to her death.
The other case involved a 76-year-old man who unintentionally swallowed an unknown quantity of alcohol-based hand-sanitizing foam attached to the foot of his hospital bed.
The patient had a history of agitation and depression and was treated with antidepressants. He had become increasingly confused over the preceding 9 months, possibly because of vascular dementia.
His blood ethanol concentration was 463 mg/dL (100 mmol/L) initially and 354 mg/dL (77mmol/L) 10 hours later. He was admitted to the ICU, where he received lorazepam and haloperidol and treated with ventilation, with a plan to allow the alcohol to be naturally metabolized.
The patient developed complications and died 6 days later. The primary causes of death were bronchopneumonia and acute alcohol toxicity, secondary to acute delirium and coronary artery disease.
Since COVID-19 started, alcohol-based hand sanitizers are among the most sought-after commodities around the world. The volume of these products – now found in homes, hospitals, schools, workplaces, and elsewhere – “may be a cause for concern,” Ms. Richards wrote.
Yet, warnings about the toxicity and lethality of intentional or unintentional ingestion of these products have not been widely disseminated, she noted.
To reduce the risk of harm, Ms. Richards suggested educating the public and health care professionals, improving warning labels on products, and increasing the awareness and reporting of such exposures to public health authorities.
“While governments and public health authorities have successfully heightened our awareness of, and need for, better hand hygiene during the COVID-19 outbreak, they must also make the public aware of the potential harms and encourage the reporting of such harms to poisons information centers,” she noted.
Increases in alcohol-based hand sanitizer poisoning during the pandemic have also been reported in the United States.
The American Association of Poison Control Centers reports that data from the National Poison Data System show 32,892 hand sanitizer exposure cases reported to the 55 U.S. poison control centers from Jan. 1 to Nov. 15, 2020 – an increase of 73%, compared with the same time period during the previous year.
An increase in self-harm
Weighing in on this issue, Robert Bassett, DO, associate medical director of the Poison Control Center at Children’s Hospital of Philadelphia, said in an interview that “cleaning agents and disinfectants have been around for eons and their potential for toxicity hasn’t changed.
“Now with COVID, and this hypervigilance when it comes to cleanliness, there is increased access and the exposure risk has gone up,” he said.
“One of the sad casualties of an overstressed health care system and a globally depressed environment is worsening behavioral health emergencies and, as part of that, the risk of self-harm goes up,” Dr. Bassett added.
“The consensus is that there has been an exacerbation of behavioral health emergencies and behavioral health needs since COVID started and hand sanitizers are readily accessible to someone who may be looking to self-harm,” he said.
This research had no specific funding. Ms. Richards is the editorial registrar of BMJ Evidence Based Medicine and is developing a website to track preventable deaths. Dr. Bassett disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Cases of poisoning – intentional and unintentional – from ingestion of alcohol-based hand sanitizer have soared during the COVID-19 pandemic.
In the United Kingdom alone, alcohol-based hand sanitizer poisonings reported to the National Poisons Information Service jumped 157% – from 155 between January 1 and September 16, 2019, to 398 between Jan. 1 and Sept. 14, 2020, new research shows.
More needs to be done to protect those at risk of unintentional and intentional swallowing of alcohol-based hand sanitizer, including children, people with dementia/confusion, and those with mental health issues, according to Georgia Richards, DPhil student, Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences, University of Oxford (England).
“If providers are supplying alcohol-based hand sanitizers in the community to reduce the spread of SARS-CoV-2, Ms. Richards said in an interview.
The study was published online Dec. 1 in BMJ Evidence-Based Medicine.
European, U.S. poisoning rates soar
In the paper Ms. Richards described two deaths that occurred in hospitals in England.
In one case, a 30-year-old woman, detained in a psychiatric unit who received the antidepressant venlafaxine was found dead in her hospital bed with a container of hand-sanitizing gel beside her.
“The gel was readily accessible to patients on the ward from a communal dispenser, and patients were allowed to fill cups or other containers with it to keep in their rooms,” Ms. Richards reported.
A postmortem analysis found a high level of alcohol in her blood (214 mg of alcohol in 100 mL of blood). The medical cause of death was listed as “ingestion of alcohol and venlafaxine.” The coroner concluded that the combination of these substances suppressed the patient’s breathing, leading to her death.
The other case involved a 76-year-old man who unintentionally swallowed an unknown quantity of alcohol-based hand-sanitizing foam attached to the foot of his hospital bed.
The patient had a history of agitation and depression and was treated with antidepressants. He had become increasingly confused over the preceding 9 months, possibly because of vascular dementia.
His blood ethanol concentration was 463 mg/dL (100 mmol/L) initially and 354 mg/dL (77mmol/L) 10 hours later. He was admitted to the ICU, where he received lorazepam and haloperidol and treated with ventilation, with a plan to allow the alcohol to be naturally metabolized.
The patient developed complications and died 6 days later. The primary causes of death were bronchopneumonia and acute alcohol toxicity, secondary to acute delirium and coronary artery disease.
Since COVID-19 started, alcohol-based hand sanitizers are among the most sought-after commodities around the world. The volume of these products – now found in homes, hospitals, schools, workplaces, and elsewhere – “may be a cause for concern,” Ms. Richards wrote.
Yet, warnings about the toxicity and lethality of intentional or unintentional ingestion of these products have not been widely disseminated, she noted.
To reduce the risk of harm, Ms. Richards suggested educating the public and health care professionals, improving warning labels on products, and increasing the awareness and reporting of such exposures to public health authorities.
“While governments and public health authorities have successfully heightened our awareness of, and need for, better hand hygiene during the COVID-19 outbreak, they must also make the public aware of the potential harms and encourage the reporting of such harms to poisons information centers,” she noted.
Increases in alcohol-based hand sanitizer poisoning during the pandemic have also been reported in the United States.
The American Association of Poison Control Centers reports that data from the National Poison Data System show 32,892 hand sanitizer exposure cases reported to the 55 U.S. poison control centers from Jan. 1 to Nov. 15, 2020 – an increase of 73%, compared with the same time period during the previous year.
An increase in self-harm
Weighing in on this issue, Robert Bassett, DO, associate medical director of the Poison Control Center at Children’s Hospital of Philadelphia, said in an interview that “cleaning agents and disinfectants have been around for eons and their potential for toxicity hasn’t changed.
“Now with COVID, and this hypervigilance when it comes to cleanliness, there is increased access and the exposure risk has gone up,” he said.
“One of the sad casualties of an overstressed health care system and a globally depressed environment is worsening behavioral health emergencies and, as part of that, the risk of self-harm goes up,” Dr. Bassett added.
“The consensus is that there has been an exacerbation of behavioral health emergencies and behavioral health needs since COVID started and hand sanitizers are readily accessible to someone who may be looking to self-harm,” he said.
This research had no specific funding. Ms. Richards is the editorial registrar of BMJ Evidence Based Medicine and is developing a website to track preventable deaths. Dr. Bassett disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Cases of poisoning – intentional and unintentional – from ingestion of alcohol-based hand sanitizer have soared during the COVID-19 pandemic.
In the United Kingdom alone, alcohol-based hand sanitizer poisonings reported to the National Poisons Information Service jumped 157% – from 155 between January 1 and September 16, 2019, to 398 between Jan. 1 and Sept. 14, 2020, new research shows.
More needs to be done to protect those at risk of unintentional and intentional swallowing of alcohol-based hand sanitizer, including children, people with dementia/confusion, and those with mental health issues, according to Georgia Richards, DPhil student, Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences, University of Oxford (England).
“If providers are supplying alcohol-based hand sanitizers in the community to reduce the spread of SARS-CoV-2, Ms. Richards said in an interview.
The study was published online Dec. 1 in BMJ Evidence-Based Medicine.
European, U.S. poisoning rates soar
In the paper Ms. Richards described two deaths that occurred in hospitals in England.
In one case, a 30-year-old woman, detained in a psychiatric unit who received the antidepressant venlafaxine was found dead in her hospital bed with a container of hand-sanitizing gel beside her.
“The gel was readily accessible to patients on the ward from a communal dispenser, and patients were allowed to fill cups or other containers with it to keep in their rooms,” Ms. Richards reported.
A postmortem analysis found a high level of alcohol in her blood (214 mg of alcohol in 100 mL of blood). The medical cause of death was listed as “ingestion of alcohol and venlafaxine.” The coroner concluded that the combination of these substances suppressed the patient’s breathing, leading to her death.
The other case involved a 76-year-old man who unintentionally swallowed an unknown quantity of alcohol-based hand-sanitizing foam attached to the foot of his hospital bed.
The patient had a history of agitation and depression and was treated with antidepressants. He had become increasingly confused over the preceding 9 months, possibly because of vascular dementia.
His blood ethanol concentration was 463 mg/dL (100 mmol/L) initially and 354 mg/dL (77mmol/L) 10 hours later. He was admitted to the ICU, where he received lorazepam and haloperidol and treated with ventilation, with a plan to allow the alcohol to be naturally metabolized.
The patient developed complications and died 6 days later. The primary causes of death were bronchopneumonia and acute alcohol toxicity, secondary to acute delirium and coronary artery disease.
Since COVID-19 started, alcohol-based hand sanitizers are among the most sought-after commodities around the world. The volume of these products – now found in homes, hospitals, schools, workplaces, and elsewhere – “may be a cause for concern,” Ms. Richards wrote.
Yet, warnings about the toxicity and lethality of intentional or unintentional ingestion of these products have not been widely disseminated, she noted.
To reduce the risk of harm, Ms. Richards suggested educating the public and health care professionals, improving warning labels on products, and increasing the awareness and reporting of such exposures to public health authorities.
“While governments and public health authorities have successfully heightened our awareness of, and need for, better hand hygiene during the COVID-19 outbreak, they must also make the public aware of the potential harms and encourage the reporting of such harms to poisons information centers,” she noted.
Increases in alcohol-based hand sanitizer poisoning during the pandemic have also been reported in the United States.
The American Association of Poison Control Centers reports that data from the National Poison Data System show 32,892 hand sanitizer exposure cases reported to the 55 U.S. poison control centers from Jan. 1 to Nov. 15, 2020 – an increase of 73%, compared with the same time period during the previous year.
An increase in self-harm
Weighing in on this issue, Robert Bassett, DO, associate medical director of the Poison Control Center at Children’s Hospital of Philadelphia, said in an interview that “cleaning agents and disinfectants have been around for eons and their potential for toxicity hasn’t changed.
“Now with COVID, and this hypervigilance when it comes to cleanliness, there is increased access and the exposure risk has gone up,” he said.
“One of the sad casualties of an overstressed health care system and a globally depressed environment is worsening behavioral health emergencies and, as part of that, the risk of self-harm goes up,” Dr. Bassett added.
“The consensus is that there has been an exacerbation of behavioral health emergencies and behavioral health needs since COVID started and hand sanitizers are readily accessible to someone who may be looking to self-harm,” he said.
This research had no specific funding. Ms. Richards is the editorial registrar of BMJ Evidence Based Medicine and is developing a website to track preventable deaths. Dr. Bassett disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Managing metabolic syndrome in patients with schizophrenia
Mr. N, age 55, has a long, documented history of schizophrenia. His overall baseline functioning has been poor because he is socially isolated, does not work, and lives in subsidized housing paid for by the county where he lives. His psychosocial circumstances have limited his ability to afford or otherwise obtain nutritious food or participate in any type of regular exercise program. He has been maintained on olanzapine, 20 mg nightly, for the past 5 years. During the past year, his functioning and overall quality of life have declined even further after he was diagnosed with hypertension. Mr. N’s in-office blood pressure was 160/95 mm Hg (normal range: systolic blood pressure, 90 to 120 mm Hg, and diastolic blood pressure, 60 to 80 mm Hg). He says his primary care physician informed him that he is pre-diabetic after his hemoglobin A1c came back at 6.0 mg/dL (normal range <5.7 mg/dL) and his body mass index was 32 kg/m2 (normal range 18.5 to 24.9 kg/m2). Currently, Mr. N’s psychiatric symptoms are stable, but his functional decline is now largely driven by metabolic parameters. Along with lifestyle changes and nonpharmacologic interventions, what else should you consider to help him?
In addition to positive, negative, and cognitive symptoms, schizophrenia is accompanied by disturbances in metabolism,1 inflammatory markers,2 and sleep/wake cycles.3 Current treatment strategies focus on addressing symptoms and functioning, but the metabolic and inflammatory targets that account for significant morbidity and mortality remain largely unaddressed.

Some patients with schizophrenia meet the criteria for metabolic syndrome, a cluster of conditions—including obesity, insulin resistance, dyslipidemia, and hypertension—that increase the risk of cardiovascular disease and type 2 diabetes mellitus (Table 14). Metabolic syndrome and its related consequences are a major barrier to the successful treatment of patients with schizophrenia, and lead to increased mortality. Druss et al5 found that individuals with significant mental illness died on average 8.2 years earlier than age-matched controls. The most common cause of death was cardiovascular disease (Table 25).
“Off-label” prescribing has been used in an attempt to delay or treat emerging metabolic syndrome in individuals with schizophrenia. Unfortunately, comprehensive strategies with a uniform application in clinical settings remain elusive. In this article, we review 3 off-label agents—metformin, topiramate, and melatonin—that may be used to address weight gain and metabolic syndrome in patients with schizophrenia.
Metformin
Metformin is an oral medication used to treat type 2 diabetes. It works by decreasing glucose absorption, suppressing gluconeogenesis in the liver, and increasing insulin sensitivity in peripheral tissues. It was FDA-approved for use in the United States in 1994. In addition to improving glucose homeostasis, metformin has also been associated with decreased body mass index (BMI), triglycerides, and low-density lipoprotein (LDL) cholesterol, and increased high-density lipoprotein (HDL) cholesterol in individuals at risk for diabetes.6
Recent consensus guidelines suggest that metformin has sufficient evidence to support its clinical use for preventing or treating antipsychotic-induced weight gain.7 A meta-analysis that included >40 randomized clinical trials (RCTs) found that metformin8-11:
- reduces antipsychotic-induced weight gain (approximately 3 kg, up to 5 kg in patients with first-episode psychosis)
- reduces fasting glucose levels, hemoglobin A1c, fasting insulin levels, and insulin resistance
- leads to a more favorable lipid profile (reduced triglycerides, LDL, and total cholesterol, and increased HDL).
Not surprisingly, metformin’s effects are augmented when used in conjunction with lifestyle interventions (diet and exercise), leading to further weight reductions of 1.5 kg and BMI reductions of 1.08 kg/m2 when compared with metformin alone.11 The mechanism underlying metformin’s attenuation of antipsychotic-induced weight gain is not fully understood, but preclinical studies suggest that it may prevent olanzapine-induced brown adipose tissue loss,12,13 alter Wnt signaling (an assortment of signal transduction pathways important for glucose homeostasis and metabolism),13 and influence the gut microbiome.14
Continue to: Metformin is generally...
Metformin is generally well tolerated. Common adverse effects include diarrhea, nausea, and abdominal pain, which are generally transient and can be ameliorated by using the extended-release formulation and lower starting doses.15 The frequency of medication discontinuation was minimal and similar in patients receiving metformin vs placebo.8,16 Despite these positive findings, most studies of metformin have had a follow-up of ≤24 weeks, and its long-term effects on antipsychotic-induced weight gain and metabolic parameters remain unknown.
When prescribing metformin for a patient with schizophrenia, consider a starting dose of 500 mg twice daily.
Topiramate
Topiramate is FDA-approved for treating generalized tonic-clonic and complex partial seizures17 and for migraine prophylaxis. More recently, it has been used off-label for weight loss in both psychiatric and non-psychiatric patients. Topiramate’s proposed mechanism for weight loss is by decreasing plasma leptin levels and increasing plasma adiponectin. A recent literature review of 8 RCTS that included 336 patients who received second-generation antipsychotics (SGAs) and adjunctive placebo or topiramate (100 to 300 mg/d) found that patients who received topiramate lost a statistically significant 2.83 kg vs placebo.18 Several case studies confirm similar findings, showing that patients with schizophrenia lost 2 to 5 kg when started on topiramate along with an SGA.19 Importantly, weight loss has been observed both in patients started on topiramate prophylactically along with an SGA, and those who had been receiving SGAs for an extended period of time before starting topiramate.
Tolerability has been a concern in patients receiving topiramate. Frequent complaints include cognitive dulling, sedation, and coldness or tingling of the extremities. In a meta-analysis of topiramate, metformin, and other medications used to induce weight loss in patients receiving SGAs, Zhuo et al20 found that topiramate was reported intolerable more frequently than other agents, although the difference was not statistically significant.
When prescribing topiramate for a patient with schizophrenia, consider a starting dose of 25 mg at bedtime.
Continue to: Melatonin
Melatonin
Melatonin is a naturally occurring hormone that is available over-the-counter and is frequently used to treat insomnia. Melatonin appears to have few adverse effects, is not habit-forming, and is inexpensive. It is a hormone produced primarily by the pineal gland, although it is also produced by many other cell types, including the skin, gut, bone marrow, thymus, and retina.21,22 Melatonin is a highly conserved essential hormone23 that acts via both G protein-coupled membrane bound receptors and nuclear receptors.23-25 Its ability to function both intra- and extracellularly implies it has an essential role in maintaining homeostatic mechanisms. Melatonin’s putative mechanism of action may derive from its effects on circadian rhythms, which in turn affect systolic blood pressure, glycemic control, and oxidative stress. In rodents, pinealectomy led to the rapid development of hypertension and metabolic syndrome. Daily administration of melatonin26 in these animals restored metabolism by decreasing abdominal fat and plasma leptin levels. These studies suggest that melatonin plays a central role in metabolism.
A recent study of patients with first-episode psychosis (n = 48) examined the effects of melatonin (3 mg/d) as an add-on treatment to olanzapine vs placebo.27 Compared with those in the placebo group, participants in the melatonin group experienced a statistically significant decrease in body weight, BMI, waist circumference, and triglyceride levels.27 In another study, the melatonin receptor agonist ramelteon was used in conjunction with SGAs.28 Augmentation with ramelteon led to significantly lower rises in total cholesterol levels compared with placebo.28
When recommending melatonin for a patient with schizophrenia, suggest that he/she begin by taking a starting dose of 3 mg nightly.
Weighing the options
Which medication to prescribe for a patient such as Mr. N would depend on the patient’s specific complaint/health target.
Weight gain or diabetes. If the patient’s primary concerns are avoiding weight gain or the development of diabetes, metformin is an excellent starting point.
Continue to: Migraines or desire to lose weight
Migraines or desire to lose weight. If the patient reports frequent migraines or a history of migraines, or if he/she is interested in weight loss, a trial of topiramate may be appropriate.
Sleep difficulties. If sleep is the patient’s primary concern, then adding melatonin might be a good first choice.
At this point, the available data points to metformin as the most efficacious medication in ameliorating some of the metabolic adverse effects associated with the long-term use of SGAs.8-11 Comprehensive treatment of patients with schizophrenia should include addressing underlying metabolic issues not only to improve health outcomes and reduce morbidity and mortality, but also to improve psychosocial functioning and quality of life.
Bottom Line
Preventing or treating metabolic syndrome is an important consideration in all patients with schizophrenia. Metformin, topiramate, and melatonin show some promise in helping ameliorate metabolic syndrome and its associated morbidity and mortality, and also may help improve patients’ functioning and quality of life.
Related Resources
- Mitchell AJ, Vancampfort D, Sweers K, et al. Prevalence of metabolic syndrome and metabolic abnormalities in schizophrenia and related disorders--a systematic review and meta-analysis. Schizophr Bull. 2013;39(2):306-318.
- Majeed MH, Khalil HA. Cardiovascular adverse effects of psychotropics: what to look for. Current Psychiatry. 2018; 17(7):54-55
- Wake, LA, Balon R. Should psychiatrists prescribe nonpsychotropic medications? Current Psychiatry. 2019; 18(11):52-56.
Drug Brand Names
Metformin • Glucophage
Olanzapine • Zyprexa
Ramelteon • Rozerem
Topiramate • Topamax
1. Bushe C, Holt R. Prevalence of diabetes and impaired glucose tolerance in patients with schizophrenia. Br J Psychiatry Suppl. 2004;184(suppl 47):S67-S71.
2. Harvey PD. Inflammation in schizophrenia: what it means and how to treat it. Am J Geriatr Psychiatry. 2017;25(1):62-63.
3. Chouinard S, Poulin J, Stip E. Sleep in untreated patients with schizophrenia: a meta-analysis. Schizophr Bull. 2004;30(4):957-967.
4. Huang PL. A comprehensive definition for metabolic syndrome. Dis Model Mech. 2009;2(5-6):231-237.
5. Druss BG, Zhao L, Von Esenwein S, et al. Understanding excess mortality in persons with mental illness: 17-year follow up of a nationally representative US survey. Med Care. 2011;49(6):599-604.
6. Salpeter SR, Buckley NS, Kahn JA, et al. Meta-analysis: metformin treatment in persons at risk for diabetes mellitus. Am J Med. 2008;121(2):149-157.
7. Faulkner G, Duncan M. Metformin to reduce weight gain and metabolic disturbance in schizophrenia. Evid Based Ment Health. 2015;18(3):89.
8. Jarskog LF, Hamer RM, Catellier DJ, et al. Metformin for weight loss and metabolic control in overweight outpatients with schizophrenia and schizoaffective disorder. Am J Psychiatry. 2013;170(9):1032-1040.
9. Mizuno Y, Suzuki T, Nakagawa A, et al. Pharmacological strategies to counteract antipsychotic-induced weight gain and metabolic adverse effects in schizophrenia: a systematic review and meta-analysis. Schizophr Bull. 2014;40(6):1385-1403.
10. Siskind DJ, Leung J, Russell AW, et al. Metformin for clozapine associated obesity: a systematic review and meta-analysis. PLoS One. 2016;11(6):e0156208. doi: 10.1371/journal.pone.0156208.
11. Wu T, Horowitz M, Rayner CK. New insights into the anti-diabetic actions of metformin: from the liver to the gut. Expert Rev Gastroenterol Hepatol. 2017;11(2):157-166.
12. Hu Y, Young AJ, Ehli EA, et al. Metformin and berberine prevent olanzapine-induced weight gain in rats. PLoS One. 2014;9(3):e93310. doi: 10.1371/journal.pone.0093310.
13. Li R, Ou J, Li L, et al. The Wnt signaling pathway effector TCF7L2 mediates olanzapine-induced weight gain and insulin resistance. Front Pharmacol. 2018;9:379.
14. Luo C, Wang X, Huang H, et al. Effect of metformin on antipsychotic-induced metabolic dysfunction: the potential role of gut-brain axis. Front Pharmacol. 2019;10:371.
15. Flory JH, Keating SJ, Siscovick D, et al. Identifying prevalence and risk factors for metformin non-persistence: a retrospective cohort study using an electronic health record. BMJ Open. 2018;8(7):e021505. doi: 10.1136/bmjopen-2018-021505.
16. Wang M, Tong JH, Zhu G, et al. Metformin for treatment of antipsychotic-induced weight gain: a randomized, placebo-controlled study. Schizophr Res. 2012;138(1):54-57.
17. Maryanoff BE. Phenotypic assessment and the discovery of topiramate. ACS Med Chem Lett. 2016;7(7):662-665.
18. Mahmood S, Booker I, Huang J, et al. Effect of topiramate on weight gain in patients receiving atypical antipsychotic agents. J Clin Psychopharmacol. 2013;33(1):90-94.
19. Lin YH, Liu CY, Hsiao MC. Management of atypical antipsychotic-induced weight gain in schizophrenic patients with topiramate. Psychiatry Clin Neurosci. 2005;59(5):613-615.
20. Zhuo C, Xu Y, Liu S, et al. Topiramate and metformin are effective add-on treatments in controlling antipsychotic-induced weight gain: a systematic review and network meta-analysis. Front Pharmacol. 2018;9:1393.
21. Nduhirabandi F, du Toit EF, Lochner A. Melatonin and the metabolic syndrome: a tool for effective therapy in obesity-associated abnormalities? Acta Physiol (Oxf). 2012;205(2):209-223.
22. Srinivasan V, Ohta Y, Espino J, et al. Metabolic syndrome, its pathophysiology and the role of melatonin. Recent Pat Endocr Metab Immune Drug Discov. 2013;7(1):11-25.
23. Hardeland R, Pandi-Perumal SR, Cardinali DP. Melatonin. Int J Biochem Cell Biol. 2006;38(3):313-316.
24. Hardeland R, Cardinali DP, Srinivasan V, et al. Melatonin--a pleiotropic, orchestrating regulator molecule. Prog Neurobiol. 2011;93(3):350-384.
25. Wiesenberg I, Missbach M, Carlberg C. The potential role of the transcription factor RZR/ROR as a mediator of nuclear melatonin signaling. Restor Neurol Neurosci. 1998;12(2-3):143-150.
26. Nava M, Quiroz Y, Vaziri N, et al. Melatonin reduces renal interstitial inflammation and improves hypertension in spontaneously hypertensive rats. Am J Physiol Renal Physiol. 2003;284(3):F447-F454.
27. Modabbernia A, Heidari P, Soleimani R, et al. Melatonin for prevention of metabolic side-effects of olanzapine in patients with first-episode schizophrenia: randomized double-blind placebo-controlled study. J Psychiatr Res. 2014;53:133-140.
28. Borba CP, Fan X, Copeland PM, et al. Placebo-controlled pilot study of ramelteon for adiposity and lipids in patients with schizophrenia. J Clin Psychopharmacol. 2011;31(5):653-658.
Mr. N, age 55, has a long, documented history of schizophrenia. His overall baseline functioning has been poor because he is socially isolated, does not work, and lives in subsidized housing paid for by the county where he lives. His psychosocial circumstances have limited his ability to afford or otherwise obtain nutritious food or participate in any type of regular exercise program. He has been maintained on olanzapine, 20 mg nightly, for the past 5 years. During the past year, his functioning and overall quality of life have declined even further after he was diagnosed with hypertension. Mr. N’s in-office blood pressure was 160/95 mm Hg (normal range: systolic blood pressure, 90 to 120 mm Hg, and diastolic blood pressure, 60 to 80 mm Hg). He says his primary care physician informed him that he is pre-diabetic after his hemoglobin A1c came back at 6.0 mg/dL (normal range <5.7 mg/dL) and his body mass index was 32 kg/m2 (normal range 18.5 to 24.9 kg/m2). Currently, Mr. N’s psychiatric symptoms are stable, but his functional decline is now largely driven by metabolic parameters. Along with lifestyle changes and nonpharmacologic interventions, what else should you consider to help him?
In addition to positive, negative, and cognitive symptoms, schizophrenia is accompanied by disturbances in metabolism,1 inflammatory markers,2 and sleep/wake cycles.3 Current treatment strategies focus on addressing symptoms and functioning, but the metabolic and inflammatory targets that account for significant morbidity and mortality remain largely unaddressed.

Some patients with schizophrenia meet the criteria for metabolic syndrome, a cluster of conditions—including obesity, insulin resistance, dyslipidemia, and hypertension—that increase the risk of cardiovascular disease and type 2 diabetes mellitus (Table 14). Metabolic syndrome and its related consequences are a major barrier to the successful treatment of patients with schizophrenia, and lead to increased mortality. Druss et al5 found that individuals with significant mental illness died on average 8.2 years earlier than age-matched controls. The most common cause of death was cardiovascular disease (Table 25).
“Off-label” prescribing has been used in an attempt to delay or treat emerging metabolic syndrome in individuals with schizophrenia. Unfortunately, comprehensive strategies with a uniform application in clinical settings remain elusive. In this article, we review 3 off-label agents—metformin, topiramate, and melatonin—that may be used to address weight gain and metabolic syndrome in patients with schizophrenia.
Metformin
Metformin is an oral medication used to treat type 2 diabetes. It works by decreasing glucose absorption, suppressing gluconeogenesis in the liver, and increasing insulin sensitivity in peripheral tissues. It was FDA-approved for use in the United States in 1994. In addition to improving glucose homeostasis, metformin has also been associated with decreased body mass index (BMI), triglycerides, and low-density lipoprotein (LDL) cholesterol, and increased high-density lipoprotein (HDL) cholesterol in individuals at risk for diabetes.6
Recent consensus guidelines suggest that metformin has sufficient evidence to support its clinical use for preventing or treating antipsychotic-induced weight gain.7 A meta-analysis that included >40 randomized clinical trials (RCTs) found that metformin8-11:
- reduces antipsychotic-induced weight gain (approximately 3 kg, up to 5 kg in patients with first-episode psychosis)
- reduces fasting glucose levels, hemoglobin A1c, fasting insulin levels, and insulin resistance
- leads to a more favorable lipid profile (reduced triglycerides, LDL, and total cholesterol, and increased HDL).
Not surprisingly, metformin’s effects are augmented when used in conjunction with lifestyle interventions (diet and exercise), leading to further weight reductions of 1.5 kg and BMI reductions of 1.08 kg/m2 when compared with metformin alone.11 The mechanism underlying metformin’s attenuation of antipsychotic-induced weight gain is not fully understood, but preclinical studies suggest that it may prevent olanzapine-induced brown adipose tissue loss,12,13 alter Wnt signaling (an assortment of signal transduction pathways important for glucose homeostasis and metabolism),13 and influence the gut microbiome.14
Continue to: Metformin is generally...
Metformin is generally well tolerated. Common adverse effects include diarrhea, nausea, and abdominal pain, which are generally transient and can be ameliorated by using the extended-release formulation and lower starting doses.15 The frequency of medication discontinuation was minimal and similar in patients receiving metformin vs placebo.8,16 Despite these positive findings, most studies of metformin have had a follow-up of ≤24 weeks, and its long-term effects on antipsychotic-induced weight gain and metabolic parameters remain unknown.
When prescribing metformin for a patient with schizophrenia, consider a starting dose of 500 mg twice daily.
Topiramate
Topiramate is FDA-approved for treating generalized tonic-clonic and complex partial seizures17 and for migraine prophylaxis. More recently, it has been used off-label for weight loss in both psychiatric and non-psychiatric patients. Topiramate’s proposed mechanism for weight loss is by decreasing plasma leptin levels and increasing plasma adiponectin. A recent literature review of 8 RCTS that included 336 patients who received second-generation antipsychotics (SGAs) and adjunctive placebo or topiramate (100 to 300 mg/d) found that patients who received topiramate lost a statistically significant 2.83 kg vs placebo.18 Several case studies confirm similar findings, showing that patients with schizophrenia lost 2 to 5 kg when started on topiramate along with an SGA.19 Importantly, weight loss has been observed both in patients started on topiramate prophylactically along with an SGA, and those who had been receiving SGAs for an extended period of time before starting topiramate.
Tolerability has been a concern in patients receiving topiramate. Frequent complaints include cognitive dulling, sedation, and coldness or tingling of the extremities. In a meta-analysis of topiramate, metformin, and other medications used to induce weight loss in patients receiving SGAs, Zhuo et al20 found that topiramate was reported intolerable more frequently than other agents, although the difference was not statistically significant.
When prescribing topiramate for a patient with schizophrenia, consider a starting dose of 25 mg at bedtime.
Continue to: Melatonin
Melatonin
Melatonin is a naturally occurring hormone that is available over-the-counter and is frequently used to treat insomnia. Melatonin appears to have few adverse effects, is not habit-forming, and is inexpensive. It is a hormone produced primarily by the pineal gland, although it is also produced by many other cell types, including the skin, gut, bone marrow, thymus, and retina.21,22 Melatonin is a highly conserved essential hormone23 that acts via both G protein-coupled membrane bound receptors and nuclear receptors.23-25 Its ability to function both intra- and extracellularly implies it has an essential role in maintaining homeostatic mechanisms. Melatonin’s putative mechanism of action may derive from its effects on circadian rhythms, which in turn affect systolic blood pressure, glycemic control, and oxidative stress. In rodents, pinealectomy led to the rapid development of hypertension and metabolic syndrome. Daily administration of melatonin26 in these animals restored metabolism by decreasing abdominal fat and plasma leptin levels. These studies suggest that melatonin plays a central role in metabolism.
A recent study of patients with first-episode psychosis (n = 48) examined the effects of melatonin (3 mg/d) as an add-on treatment to olanzapine vs placebo.27 Compared with those in the placebo group, participants in the melatonin group experienced a statistically significant decrease in body weight, BMI, waist circumference, and triglyceride levels.27 In another study, the melatonin receptor agonist ramelteon was used in conjunction with SGAs.28 Augmentation with ramelteon led to significantly lower rises in total cholesterol levels compared with placebo.28
When recommending melatonin for a patient with schizophrenia, suggest that he/she begin by taking a starting dose of 3 mg nightly.
Weighing the options
Which medication to prescribe for a patient such as Mr. N would depend on the patient’s specific complaint/health target.
Weight gain or diabetes. If the patient’s primary concerns are avoiding weight gain or the development of diabetes, metformin is an excellent starting point.
Continue to: Migraines or desire to lose weight
Migraines or desire to lose weight. If the patient reports frequent migraines or a history of migraines, or if he/she is interested in weight loss, a trial of topiramate may be appropriate.
Sleep difficulties. If sleep is the patient’s primary concern, then adding melatonin might be a good first choice.
At this point, the available data points to metformin as the most efficacious medication in ameliorating some of the metabolic adverse effects associated with the long-term use of SGAs.8-11 Comprehensive treatment of patients with schizophrenia should include addressing underlying metabolic issues not only to improve health outcomes and reduce morbidity and mortality, but also to improve psychosocial functioning and quality of life.
Bottom Line
Preventing or treating metabolic syndrome is an important consideration in all patients with schizophrenia. Metformin, topiramate, and melatonin show some promise in helping ameliorate metabolic syndrome and its associated morbidity and mortality, and also may help improve patients’ functioning and quality of life.
Related Resources
- Mitchell AJ, Vancampfort D, Sweers K, et al. Prevalence of metabolic syndrome and metabolic abnormalities in schizophrenia and related disorders--a systematic review and meta-analysis. Schizophr Bull. 2013;39(2):306-318.
- Majeed MH, Khalil HA. Cardiovascular adverse effects of psychotropics: what to look for. Current Psychiatry. 2018; 17(7):54-55
- Wake, LA, Balon R. Should psychiatrists prescribe nonpsychotropic medications? Current Psychiatry. 2019; 18(11):52-56.
Drug Brand Names
Metformin • Glucophage
Olanzapine • Zyprexa
Ramelteon • Rozerem
Topiramate • Topamax
Mr. N, age 55, has a long, documented history of schizophrenia. His overall baseline functioning has been poor because he is socially isolated, does not work, and lives in subsidized housing paid for by the county where he lives. His psychosocial circumstances have limited his ability to afford or otherwise obtain nutritious food or participate in any type of regular exercise program. He has been maintained on olanzapine, 20 mg nightly, for the past 5 years. During the past year, his functioning and overall quality of life have declined even further after he was diagnosed with hypertension. Mr. N’s in-office blood pressure was 160/95 mm Hg (normal range: systolic blood pressure, 90 to 120 mm Hg, and diastolic blood pressure, 60 to 80 mm Hg). He says his primary care physician informed him that he is pre-diabetic after his hemoglobin A1c came back at 6.0 mg/dL (normal range <5.7 mg/dL) and his body mass index was 32 kg/m2 (normal range 18.5 to 24.9 kg/m2). Currently, Mr. N’s psychiatric symptoms are stable, but his functional decline is now largely driven by metabolic parameters. Along with lifestyle changes and nonpharmacologic interventions, what else should you consider to help him?
In addition to positive, negative, and cognitive symptoms, schizophrenia is accompanied by disturbances in metabolism,1 inflammatory markers,2 and sleep/wake cycles.3 Current treatment strategies focus on addressing symptoms and functioning, but the metabolic and inflammatory targets that account for significant morbidity and mortality remain largely unaddressed.

Some patients with schizophrenia meet the criteria for metabolic syndrome, a cluster of conditions—including obesity, insulin resistance, dyslipidemia, and hypertension—that increase the risk of cardiovascular disease and type 2 diabetes mellitus (Table 14). Metabolic syndrome and its related consequences are a major barrier to the successful treatment of patients with schizophrenia, and lead to increased mortality. Druss et al5 found that individuals with significant mental illness died on average 8.2 years earlier than age-matched controls. The most common cause of death was cardiovascular disease (Table 25).
“Off-label” prescribing has been used in an attempt to delay or treat emerging metabolic syndrome in individuals with schizophrenia. Unfortunately, comprehensive strategies with a uniform application in clinical settings remain elusive. In this article, we review 3 off-label agents—metformin, topiramate, and melatonin—that may be used to address weight gain and metabolic syndrome in patients with schizophrenia.
Metformin
Metformin is an oral medication used to treat type 2 diabetes. It works by decreasing glucose absorption, suppressing gluconeogenesis in the liver, and increasing insulin sensitivity in peripheral tissues. It was FDA-approved for use in the United States in 1994. In addition to improving glucose homeostasis, metformin has also been associated with decreased body mass index (BMI), triglycerides, and low-density lipoprotein (LDL) cholesterol, and increased high-density lipoprotein (HDL) cholesterol in individuals at risk for diabetes.6
Recent consensus guidelines suggest that metformin has sufficient evidence to support its clinical use for preventing or treating antipsychotic-induced weight gain.7 A meta-analysis that included >40 randomized clinical trials (RCTs) found that metformin8-11:
- reduces antipsychotic-induced weight gain (approximately 3 kg, up to 5 kg in patients with first-episode psychosis)
- reduces fasting glucose levels, hemoglobin A1c, fasting insulin levels, and insulin resistance
- leads to a more favorable lipid profile (reduced triglycerides, LDL, and total cholesterol, and increased HDL).
Not surprisingly, metformin’s effects are augmented when used in conjunction with lifestyle interventions (diet and exercise), leading to further weight reductions of 1.5 kg and BMI reductions of 1.08 kg/m2 when compared with metformin alone.11 The mechanism underlying metformin’s attenuation of antipsychotic-induced weight gain is not fully understood, but preclinical studies suggest that it may prevent olanzapine-induced brown adipose tissue loss,12,13 alter Wnt signaling (an assortment of signal transduction pathways important for glucose homeostasis and metabolism),13 and influence the gut microbiome.14
Continue to: Metformin is generally...
Metformin is generally well tolerated. Common adverse effects include diarrhea, nausea, and abdominal pain, which are generally transient and can be ameliorated by using the extended-release formulation and lower starting doses.15 The frequency of medication discontinuation was minimal and similar in patients receiving metformin vs placebo.8,16 Despite these positive findings, most studies of metformin have had a follow-up of ≤24 weeks, and its long-term effects on antipsychotic-induced weight gain and metabolic parameters remain unknown.
When prescribing metformin for a patient with schizophrenia, consider a starting dose of 500 mg twice daily.
Topiramate
Topiramate is FDA-approved for treating generalized tonic-clonic and complex partial seizures17 and for migraine prophylaxis. More recently, it has been used off-label for weight loss in both psychiatric and non-psychiatric patients. Topiramate’s proposed mechanism for weight loss is by decreasing plasma leptin levels and increasing plasma adiponectin. A recent literature review of 8 RCTS that included 336 patients who received second-generation antipsychotics (SGAs) and adjunctive placebo or topiramate (100 to 300 mg/d) found that patients who received topiramate lost a statistically significant 2.83 kg vs placebo.18 Several case studies confirm similar findings, showing that patients with schizophrenia lost 2 to 5 kg when started on topiramate along with an SGA.19 Importantly, weight loss has been observed both in patients started on topiramate prophylactically along with an SGA, and those who had been receiving SGAs for an extended period of time before starting topiramate.
Tolerability has been a concern in patients receiving topiramate. Frequent complaints include cognitive dulling, sedation, and coldness or tingling of the extremities. In a meta-analysis of topiramate, metformin, and other medications used to induce weight loss in patients receiving SGAs, Zhuo et al20 found that topiramate was reported intolerable more frequently than other agents, although the difference was not statistically significant.
When prescribing topiramate for a patient with schizophrenia, consider a starting dose of 25 mg at bedtime.
Continue to: Melatonin
Melatonin
Melatonin is a naturally occurring hormone that is available over-the-counter and is frequently used to treat insomnia. Melatonin appears to have few adverse effects, is not habit-forming, and is inexpensive. It is a hormone produced primarily by the pineal gland, although it is also produced by many other cell types, including the skin, gut, bone marrow, thymus, and retina.21,22 Melatonin is a highly conserved essential hormone23 that acts via both G protein-coupled membrane bound receptors and nuclear receptors.23-25 Its ability to function both intra- and extracellularly implies it has an essential role in maintaining homeostatic mechanisms. Melatonin’s putative mechanism of action may derive from its effects on circadian rhythms, which in turn affect systolic blood pressure, glycemic control, and oxidative stress. In rodents, pinealectomy led to the rapid development of hypertension and metabolic syndrome. Daily administration of melatonin26 in these animals restored metabolism by decreasing abdominal fat and plasma leptin levels. These studies suggest that melatonin plays a central role in metabolism.
A recent study of patients with first-episode psychosis (n = 48) examined the effects of melatonin (3 mg/d) as an add-on treatment to olanzapine vs placebo.27 Compared with those in the placebo group, participants in the melatonin group experienced a statistically significant decrease in body weight, BMI, waist circumference, and triglyceride levels.27 In another study, the melatonin receptor agonist ramelteon was used in conjunction with SGAs.28 Augmentation with ramelteon led to significantly lower rises in total cholesterol levels compared with placebo.28
When recommending melatonin for a patient with schizophrenia, suggest that he/she begin by taking a starting dose of 3 mg nightly.
Weighing the options
Which medication to prescribe for a patient such as Mr. N would depend on the patient’s specific complaint/health target.
Weight gain or diabetes. If the patient’s primary concerns are avoiding weight gain or the development of diabetes, metformin is an excellent starting point.
Continue to: Migraines or desire to lose weight
Migraines or desire to lose weight. If the patient reports frequent migraines or a history of migraines, or if he/she is interested in weight loss, a trial of topiramate may be appropriate.
Sleep difficulties. If sleep is the patient’s primary concern, then adding melatonin might be a good first choice.
At this point, the available data points to metformin as the most efficacious medication in ameliorating some of the metabolic adverse effects associated with the long-term use of SGAs.8-11 Comprehensive treatment of patients with schizophrenia should include addressing underlying metabolic issues not only to improve health outcomes and reduce morbidity and mortality, but also to improve psychosocial functioning and quality of life.
Bottom Line
Preventing or treating metabolic syndrome is an important consideration in all patients with schizophrenia. Metformin, topiramate, and melatonin show some promise in helping ameliorate metabolic syndrome and its associated morbidity and mortality, and also may help improve patients’ functioning and quality of life.
Related Resources
- Mitchell AJ, Vancampfort D, Sweers K, et al. Prevalence of metabolic syndrome and metabolic abnormalities in schizophrenia and related disorders--a systematic review and meta-analysis. Schizophr Bull. 2013;39(2):306-318.
- Majeed MH, Khalil HA. Cardiovascular adverse effects of psychotropics: what to look for. Current Psychiatry. 2018; 17(7):54-55
- Wake, LA, Balon R. Should psychiatrists prescribe nonpsychotropic medications? Current Psychiatry. 2019; 18(11):52-56.
Drug Brand Names
Metformin • Glucophage
Olanzapine • Zyprexa
Ramelteon • Rozerem
Topiramate • Topamax
1. Bushe C, Holt R. Prevalence of diabetes and impaired glucose tolerance in patients with schizophrenia. Br J Psychiatry Suppl. 2004;184(suppl 47):S67-S71.
2. Harvey PD. Inflammation in schizophrenia: what it means and how to treat it. Am J Geriatr Psychiatry. 2017;25(1):62-63.
3. Chouinard S, Poulin J, Stip E. Sleep in untreated patients with schizophrenia: a meta-analysis. Schizophr Bull. 2004;30(4):957-967.
4. Huang PL. A comprehensive definition for metabolic syndrome. Dis Model Mech. 2009;2(5-6):231-237.
5. Druss BG, Zhao L, Von Esenwein S, et al. Understanding excess mortality in persons with mental illness: 17-year follow up of a nationally representative US survey. Med Care. 2011;49(6):599-604.
6. Salpeter SR, Buckley NS, Kahn JA, et al. Meta-analysis: metformin treatment in persons at risk for diabetes mellitus. Am J Med. 2008;121(2):149-157.
7. Faulkner G, Duncan M. Metformin to reduce weight gain and metabolic disturbance in schizophrenia. Evid Based Ment Health. 2015;18(3):89.
8. Jarskog LF, Hamer RM, Catellier DJ, et al. Metformin for weight loss and metabolic control in overweight outpatients with schizophrenia and schizoaffective disorder. Am J Psychiatry. 2013;170(9):1032-1040.
9. Mizuno Y, Suzuki T, Nakagawa A, et al. Pharmacological strategies to counteract antipsychotic-induced weight gain and metabolic adverse effects in schizophrenia: a systematic review and meta-analysis. Schizophr Bull. 2014;40(6):1385-1403.
10. Siskind DJ, Leung J, Russell AW, et al. Metformin for clozapine associated obesity: a systematic review and meta-analysis. PLoS One. 2016;11(6):e0156208. doi: 10.1371/journal.pone.0156208.
11. Wu T, Horowitz M, Rayner CK. New insights into the anti-diabetic actions of metformin: from the liver to the gut. Expert Rev Gastroenterol Hepatol. 2017;11(2):157-166.
12. Hu Y, Young AJ, Ehli EA, et al. Metformin and berberine prevent olanzapine-induced weight gain in rats. PLoS One. 2014;9(3):e93310. doi: 10.1371/journal.pone.0093310.
13. Li R, Ou J, Li L, et al. The Wnt signaling pathway effector TCF7L2 mediates olanzapine-induced weight gain and insulin resistance. Front Pharmacol. 2018;9:379.
14. Luo C, Wang X, Huang H, et al. Effect of metformin on antipsychotic-induced metabolic dysfunction: the potential role of gut-brain axis. Front Pharmacol. 2019;10:371.
15. Flory JH, Keating SJ, Siscovick D, et al. Identifying prevalence and risk factors for metformin non-persistence: a retrospective cohort study using an electronic health record. BMJ Open. 2018;8(7):e021505. doi: 10.1136/bmjopen-2018-021505.
16. Wang M, Tong JH, Zhu G, et al. Metformin for treatment of antipsychotic-induced weight gain: a randomized, placebo-controlled study. Schizophr Res. 2012;138(1):54-57.
17. Maryanoff BE. Phenotypic assessment and the discovery of topiramate. ACS Med Chem Lett. 2016;7(7):662-665.
18. Mahmood S, Booker I, Huang J, et al. Effect of topiramate on weight gain in patients receiving atypical antipsychotic agents. J Clin Psychopharmacol. 2013;33(1):90-94.
19. Lin YH, Liu CY, Hsiao MC. Management of atypical antipsychotic-induced weight gain in schizophrenic patients with topiramate. Psychiatry Clin Neurosci. 2005;59(5):613-615.
20. Zhuo C, Xu Y, Liu S, et al. Topiramate and metformin are effective add-on treatments in controlling antipsychotic-induced weight gain: a systematic review and network meta-analysis. Front Pharmacol. 2018;9:1393.
21. Nduhirabandi F, du Toit EF, Lochner A. Melatonin and the metabolic syndrome: a tool for effective therapy in obesity-associated abnormalities? Acta Physiol (Oxf). 2012;205(2):209-223.
22. Srinivasan V, Ohta Y, Espino J, et al. Metabolic syndrome, its pathophysiology and the role of melatonin. Recent Pat Endocr Metab Immune Drug Discov. 2013;7(1):11-25.
23. Hardeland R, Pandi-Perumal SR, Cardinali DP. Melatonin. Int J Biochem Cell Biol. 2006;38(3):313-316.
24. Hardeland R, Cardinali DP, Srinivasan V, et al. Melatonin--a pleiotropic, orchestrating regulator molecule. Prog Neurobiol. 2011;93(3):350-384.
25. Wiesenberg I, Missbach M, Carlberg C. The potential role of the transcription factor RZR/ROR as a mediator of nuclear melatonin signaling. Restor Neurol Neurosci. 1998;12(2-3):143-150.
26. Nava M, Quiroz Y, Vaziri N, et al. Melatonin reduces renal interstitial inflammation and improves hypertension in spontaneously hypertensive rats. Am J Physiol Renal Physiol. 2003;284(3):F447-F454.
27. Modabbernia A, Heidari P, Soleimani R, et al. Melatonin for prevention of metabolic side-effects of olanzapine in patients with first-episode schizophrenia: randomized double-blind placebo-controlled study. J Psychiatr Res. 2014;53:133-140.
28. Borba CP, Fan X, Copeland PM, et al. Placebo-controlled pilot study of ramelteon for adiposity and lipids in patients with schizophrenia. J Clin Psychopharmacol. 2011;31(5):653-658.
1. Bushe C, Holt R. Prevalence of diabetes and impaired glucose tolerance in patients with schizophrenia. Br J Psychiatry Suppl. 2004;184(suppl 47):S67-S71.
2. Harvey PD. Inflammation in schizophrenia: what it means and how to treat it. Am J Geriatr Psychiatry. 2017;25(1):62-63.
3. Chouinard S, Poulin J, Stip E. Sleep in untreated patients with schizophrenia: a meta-analysis. Schizophr Bull. 2004;30(4):957-967.
4. Huang PL. A comprehensive definition for metabolic syndrome. Dis Model Mech. 2009;2(5-6):231-237.
5. Druss BG, Zhao L, Von Esenwein S, et al. Understanding excess mortality in persons with mental illness: 17-year follow up of a nationally representative US survey. Med Care. 2011;49(6):599-604.
6. Salpeter SR, Buckley NS, Kahn JA, et al. Meta-analysis: metformin treatment in persons at risk for diabetes mellitus. Am J Med. 2008;121(2):149-157.
7. Faulkner G, Duncan M. Metformin to reduce weight gain and metabolic disturbance in schizophrenia. Evid Based Ment Health. 2015;18(3):89.
8. Jarskog LF, Hamer RM, Catellier DJ, et al. Metformin for weight loss and metabolic control in overweight outpatients with schizophrenia and schizoaffective disorder. Am J Psychiatry. 2013;170(9):1032-1040.
9. Mizuno Y, Suzuki T, Nakagawa A, et al. Pharmacological strategies to counteract antipsychotic-induced weight gain and metabolic adverse effects in schizophrenia: a systematic review and meta-analysis. Schizophr Bull. 2014;40(6):1385-1403.
10. Siskind DJ, Leung J, Russell AW, et al. Metformin for clozapine associated obesity: a systematic review and meta-analysis. PLoS One. 2016;11(6):e0156208. doi: 10.1371/journal.pone.0156208.
11. Wu T, Horowitz M, Rayner CK. New insights into the anti-diabetic actions of metformin: from the liver to the gut. Expert Rev Gastroenterol Hepatol. 2017;11(2):157-166.
12. Hu Y, Young AJ, Ehli EA, et al. Metformin and berberine prevent olanzapine-induced weight gain in rats. PLoS One. 2014;9(3):e93310. doi: 10.1371/journal.pone.0093310.
13. Li R, Ou J, Li L, et al. The Wnt signaling pathway effector TCF7L2 mediates olanzapine-induced weight gain and insulin resistance. Front Pharmacol. 2018;9:379.
14. Luo C, Wang X, Huang H, et al. Effect of metformin on antipsychotic-induced metabolic dysfunction: the potential role of gut-brain axis. Front Pharmacol. 2019;10:371.
15. Flory JH, Keating SJ, Siscovick D, et al. Identifying prevalence and risk factors for metformin non-persistence: a retrospective cohort study using an electronic health record. BMJ Open. 2018;8(7):e021505. doi: 10.1136/bmjopen-2018-021505.
16. Wang M, Tong JH, Zhu G, et al. Metformin for treatment of antipsychotic-induced weight gain: a randomized, placebo-controlled study. Schizophr Res. 2012;138(1):54-57.
17. Maryanoff BE. Phenotypic assessment and the discovery of topiramate. ACS Med Chem Lett. 2016;7(7):662-665.
18. Mahmood S, Booker I, Huang J, et al. Effect of topiramate on weight gain in patients receiving atypical antipsychotic agents. J Clin Psychopharmacol. 2013;33(1):90-94.
19. Lin YH, Liu CY, Hsiao MC. Management of atypical antipsychotic-induced weight gain in schizophrenic patients with topiramate. Psychiatry Clin Neurosci. 2005;59(5):613-615.
20. Zhuo C, Xu Y, Liu S, et al. Topiramate and metformin are effective add-on treatments in controlling antipsychotic-induced weight gain: a systematic review and network meta-analysis. Front Pharmacol. 2018;9:1393.
21. Nduhirabandi F, du Toit EF, Lochner A. Melatonin and the metabolic syndrome: a tool for effective therapy in obesity-associated abnormalities? Acta Physiol (Oxf). 2012;205(2):209-223.
22. Srinivasan V, Ohta Y, Espino J, et al. Metabolic syndrome, its pathophysiology and the role of melatonin. Recent Pat Endocr Metab Immune Drug Discov. 2013;7(1):11-25.
23. Hardeland R, Pandi-Perumal SR, Cardinali DP. Melatonin. Int J Biochem Cell Biol. 2006;38(3):313-316.
24. Hardeland R, Cardinali DP, Srinivasan V, et al. Melatonin--a pleiotropic, orchestrating regulator molecule. Prog Neurobiol. 2011;93(3):350-384.
25. Wiesenberg I, Missbach M, Carlberg C. The potential role of the transcription factor RZR/ROR as a mediator of nuclear melatonin signaling. Restor Neurol Neurosci. 1998;12(2-3):143-150.
26. Nava M, Quiroz Y, Vaziri N, et al. Melatonin reduces renal interstitial inflammation and improves hypertension in spontaneously hypertensive rats. Am J Physiol Renal Physiol. 2003;284(3):F447-F454.
27. Modabbernia A, Heidari P, Soleimani R, et al. Melatonin for prevention of metabolic side-effects of olanzapine in patients with first-episode schizophrenia: randomized double-blind placebo-controlled study. J Psychiatr Res. 2014;53:133-140.
28. Borba CP, Fan X, Copeland PM, et al. Placebo-controlled pilot study of ramelteon for adiposity and lipids in patients with schizophrenia. J Clin Psychopharmacol. 2011;31(5):653-658.
Food preservative for early psychosis: Final word?
Adjunctive use of sodium benzoate (BZ), a common food preservative that has previously shown promise in the treatment of chronic refractory psychosis, appears to be ineffective in the early stages of the disorder, new research suggests.
Results of a randomized controlled trial show the agent was no more effective than placebo in reducing early psychosis symptoms, although it was safe and well tolerated.
“Both groups of patients improved over the 12 weeks of the study, [suggesting] that most people with early psychosis will get well with antipsychotic medication and psychosocial interventions and adding sodium benzoate to their treatment does not add any additional benefits,” the study’s lead author, James Scott, MBBS, PhD, head of mental health research, QIMR Berghofer Medical Research Institute, Herston, Australia, told Medscape Medical News.
The paper was published online November 10 in JAMA Network Open.
Positive outcomes in chronic disease
Despite treatment with antipsychotics, many patients with psychosis experience persistent impairment, the investigators note.
Most antipsychotics are dopaminergic in action, but it is now recognized that the pathophysiology underlying psychosis extends beyond dopaminergic dysregulation with hypofunction of the N-methyl-D-aspartate (NMDA) receptors also implicated but not addressed by standard antipsychotics, they add.
NMDA receptors consist of two main subunits – the glutamate and glycine-binding sites. D-amino acids (DAAs) are agonists of the glycine subunit and have shown promise as adjunctive therapies for the treatment of schizophrenia, the investigators note.
DAAs are subject to oxidation by the flavoenzyme D-amino acid oxidase (DAAO). The oxidation limits their bioavailability and can cause nephrotoxic side effects. The food preservative BZ, which is not related to the benzodiazepine class of medications, inhibits DAAO and therefore may make DAAs safer and more effective.
Scott noted that two previous trials of BZ – a 2013 study and a 2017 investigation – in chronic, treatment-refractory schizophrenia have “reported excellent outcomes with significant improvement in clinical symptoms.”
“We saw that sodium benzoate was a safe and well-tolerated agent, and we thought it was important to conduct a trial of this medication in people in the early stages of psychotic illness,” he said.
To investigate, the researchers randomly assigned 100 individuals who were experiencing early psychosis, which was defined as illness onset within the last 2 years, to receive either 500 mg of BZ twice daily or placebo for 12 weeks.
Participants (mean [SD] age 21.4 [4.1] years, 73% male) were required to be taking antipsychotic medications for at least 1 continuous month during the previous 2 years and to be free of comorbid physical illnesses requiring additional treatment or hospitalization.
Most participants (84%) had schizophrenia and the remainder had affective psychoses. Most participants (88%) lived independently.
The BZ and the placebo groups were similar with respect to baseline characteristics, except that the mean waist circumference was higher in the placebo group than in the BZ group.
The majority of patients were being treated with antipsychotics alone (83%), followed by antipsychotics in combination with mood stabilizers (13%) and a small number were taking mood stabilizers alone (4%). The most commonly used antipsychotics were olanzapine and aripiprazole.
Not recommended
Psychosis was confirmed using the Positive and Negative Syndrome Scale (PANSS), and the inclusion criteria was a baseline score of ≥ 55. Secondary outcomes were scores on the Hamilton Depression Rating Scale, the clinician-rated Global Assessment of Function, and the Assessment of Quality of Life Scale.
The researchers also measured concentrations of the amino acids oxidized by DAAO (D-alanine and L-alanine, D-serine and L-serine).
Although both groups experienced a reduction in total PANSS scores during the study, there were no significant differences in PANSS total score between the BZ and the placebo groups at 12 weeks (endpoint least-square mean difference [SE] −1.2 [2.4] t = −0.49, P = .63).
There were also no significant differences between the groups on all PANSS subscales as well as any of the secondary clinical measures (P < .007).
A total of 122 adverse events (AEs) overall were reported by 66 participants, but rates of AEs were comparable between the BZ and placebo groups at 55% vs. 46% respectively. There were 11 serious AEs reported by 10 participants. Only one of these was related to the study drug.
There were no statistically significant changes in amino acid concentrations between the two groups.
The authors note several limitations of the study, including the possibility that protective agents may need longer times than 12 weeks – possibly as long as 6 to 12 months – to show efficacy. Moreover, the dose of BZ needed to produce a response remains “uncertain.”
“ and should further investigate whether benzoate acts by altering amino acid levels or by reducing oxidative stress in people with schizophrenia,” the authors suggest.
Scott added that further research in patients with treatment-refractory schizophrenia is needed to determine whether BZ “has a role in this patient population.”
The authors conclude that at present, “the routine use of this agent as an adjunctive treatment for early psychosis is not recommended.”
No surprise?
Commenting on the study for Medscape Medical News, Kenji Hashimoto, PhD, Division of Clinical Neuroscience, Chiba University Center for Forensic Mental Health, Chiba, Japan, said that, although previous studies of BZ showed benefit in stable patients with chronic schizophrenia, “it is unlikely that [sodium] benzoate may have beneficial effects in the acute phase of psychosis.”
Hashimoto, who was not involved with the study, noted that BZ is a “weak DAAO inhibitor and that DAAO expression in the frontal cortex of human beings is very low.”
This project was supported by a John Cade Fellowship from the National Health and Medical Research Council and support from the Queensland Centre for Mental Health Research, which receives funding from the Queensland Health Department. Scott is supported by an NHMRC Practitioner Fellowship. The other authors’ disclosures are listed on the original article. Hashimoto has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Adjunctive use of sodium benzoate (BZ), a common food preservative that has previously shown promise in the treatment of chronic refractory psychosis, appears to be ineffective in the early stages of the disorder, new research suggests.
Results of a randomized controlled trial show the agent was no more effective than placebo in reducing early psychosis symptoms, although it was safe and well tolerated.
“Both groups of patients improved over the 12 weeks of the study, [suggesting] that most people with early psychosis will get well with antipsychotic medication and psychosocial interventions and adding sodium benzoate to their treatment does not add any additional benefits,” the study’s lead author, James Scott, MBBS, PhD, head of mental health research, QIMR Berghofer Medical Research Institute, Herston, Australia, told Medscape Medical News.
The paper was published online November 10 in JAMA Network Open.
Positive outcomes in chronic disease
Despite treatment with antipsychotics, many patients with psychosis experience persistent impairment, the investigators note.
Most antipsychotics are dopaminergic in action, but it is now recognized that the pathophysiology underlying psychosis extends beyond dopaminergic dysregulation with hypofunction of the N-methyl-D-aspartate (NMDA) receptors also implicated but not addressed by standard antipsychotics, they add.
NMDA receptors consist of two main subunits – the glutamate and glycine-binding sites. D-amino acids (DAAs) are agonists of the glycine subunit and have shown promise as adjunctive therapies for the treatment of schizophrenia, the investigators note.
DAAs are subject to oxidation by the flavoenzyme D-amino acid oxidase (DAAO). The oxidation limits their bioavailability and can cause nephrotoxic side effects. The food preservative BZ, which is not related to the benzodiazepine class of medications, inhibits DAAO and therefore may make DAAs safer and more effective.
Scott noted that two previous trials of BZ – a 2013 study and a 2017 investigation – in chronic, treatment-refractory schizophrenia have “reported excellent outcomes with significant improvement in clinical symptoms.”
“We saw that sodium benzoate was a safe and well-tolerated agent, and we thought it was important to conduct a trial of this medication in people in the early stages of psychotic illness,” he said.
To investigate, the researchers randomly assigned 100 individuals who were experiencing early psychosis, which was defined as illness onset within the last 2 years, to receive either 500 mg of BZ twice daily or placebo for 12 weeks.
Participants (mean [SD] age 21.4 [4.1] years, 73% male) were required to be taking antipsychotic medications for at least 1 continuous month during the previous 2 years and to be free of comorbid physical illnesses requiring additional treatment or hospitalization.
Most participants (84%) had schizophrenia and the remainder had affective psychoses. Most participants (88%) lived independently.
The BZ and the placebo groups were similar with respect to baseline characteristics, except that the mean waist circumference was higher in the placebo group than in the BZ group.
The majority of patients were being treated with antipsychotics alone (83%), followed by antipsychotics in combination with mood stabilizers (13%) and a small number were taking mood stabilizers alone (4%). The most commonly used antipsychotics were olanzapine and aripiprazole.
Not recommended
Psychosis was confirmed using the Positive and Negative Syndrome Scale (PANSS), and the inclusion criteria was a baseline score of ≥ 55. Secondary outcomes were scores on the Hamilton Depression Rating Scale, the clinician-rated Global Assessment of Function, and the Assessment of Quality of Life Scale.
The researchers also measured concentrations of the amino acids oxidized by DAAO (D-alanine and L-alanine, D-serine and L-serine).
Although both groups experienced a reduction in total PANSS scores during the study, there were no significant differences in PANSS total score between the BZ and the placebo groups at 12 weeks (endpoint least-square mean difference [SE] −1.2 [2.4] t = −0.49, P = .63).
There were also no significant differences between the groups on all PANSS subscales as well as any of the secondary clinical measures (P < .007).
A total of 122 adverse events (AEs) overall were reported by 66 participants, but rates of AEs were comparable between the BZ and placebo groups at 55% vs. 46% respectively. There were 11 serious AEs reported by 10 participants. Only one of these was related to the study drug.
There were no statistically significant changes in amino acid concentrations between the two groups.
The authors note several limitations of the study, including the possibility that protective agents may need longer times than 12 weeks – possibly as long as 6 to 12 months – to show efficacy. Moreover, the dose of BZ needed to produce a response remains “uncertain.”
“ and should further investigate whether benzoate acts by altering amino acid levels or by reducing oxidative stress in people with schizophrenia,” the authors suggest.
Scott added that further research in patients with treatment-refractory schizophrenia is needed to determine whether BZ “has a role in this patient population.”
The authors conclude that at present, “the routine use of this agent as an adjunctive treatment for early psychosis is not recommended.”
No surprise?
Commenting on the study for Medscape Medical News, Kenji Hashimoto, PhD, Division of Clinical Neuroscience, Chiba University Center for Forensic Mental Health, Chiba, Japan, said that, although previous studies of BZ showed benefit in stable patients with chronic schizophrenia, “it is unlikely that [sodium] benzoate may have beneficial effects in the acute phase of psychosis.”
Hashimoto, who was not involved with the study, noted that BZ is a “weak DAAO inhibitor and that DAAO expression in the frontal cortex of human beings is very low.”
This project was supported by a John Cade Fellowship from the National Health and Medical Research Council and support from the Queensland Centre for Mental Health Research, which receives funding from the Queensland Health Department. Scott is supported by an NHMRC Practitioner Fellowship. The other authors’ disclosures are listed on the original article. Hashimoto has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Adjunctive use of sodium benzoate (BZ), a common food preservative that has previously shown promise in the treatment of chronic refractory psychosis, appears to be ineffective in the early stages of the disorder, new research suggests.
Results of a randomized controlled trial show the agent was no more effective than placebo in reducing early psychosis symptoms, although it was safe and well tolerated.
“Both groups of patients improved over the 12 weeks of the study, [suggesting] that most people with early psychosis will get well with antipsychotic medication and psychosocial interventions and adding sodium benzoate to their treatment does not add any additional benefits,” the study’s lead author, James Scott, MBBS, PhD, head of mental health research, QIMR Berghofer Medical Research Institute, Herston, Australia, told Medscape Medical News.
The paper was published online November 10 in JAMA Network Open.
Positive outcomes in chronic disease
Despite treatment with antipsychotics, many patients with psychosis experience persistent impairment, the investigators note.
Most antipsychotics are dopaminergic in action, but it is now recognized that the pathophysiology underlying psychosis extends beyond dopaminergic dysregulation with hypofunction of the N-methyl-D-aspartate (NMDA) receptors also implicated but not addressed by standard antipsychotics, they add.
NMDA receptors consist of two main subunits – the glutamate and glycine-binding sites. D-amino acids (DAAs) are agonists of the glycine subunit and have shown promise as adjunctive therapies for the treatment of schizophrenia, the investigators note.
DAAs are subject to oxidation by the flavoenzyme D-amino acid oxidase (DAAO). The oxidation limits their bioavailability and can cause nephrotoxic side effects. The food preservative BZ, which is not related to the benzodiazepine class of medications, inhibits DAAO and therefore may make DAAs safer and more effective.
Scott noted that two previous trials of BZ – a 2013 study and a 2017 investigation – in chronic, treatment-refractory schizophrenia have “reported excellent outcomes with significant improvement in clinical symptoms.”
“We saw that sodium benzoate was a safe and well-tolerated agent, and we thought it was important to conduct a trial of this medication in people in the early stages of psychotic illness,” he said.
To investigate, the researchers randomly assigned 100 individuals who were experiencing early psychosis, which was defined as illness onset within the last 2 years, to receive either 500 mg of BZ twice daily or placebo for 12 weeks.
Participants (mean [SD] age 21.4 [4.1] years, 73% male) were required to be taking antipsychotic medications for at least 1 continuous month during the previous 2 years and to be free of comorbid physical illnesses requiring additional treatment or hospitalization.
Most participants (84%) had schizophrenia and the remainder had affective psychoses. Most participants (88%) lived independently.
The BZ and the placebo groups were similar with respect to baseline characteristics, except that the mean waist circumference was higher in the placebo group than in the BZ group.
The majority of patients were being treated with antipsychotics alone (83%), followed by antipsychotics in combination with mood stabilizers (13%) and a small number were taking mood stabilizers alone (4%). The most commonly used antipsychotics were olanzapine and aripiprazole.
Not recommended
Psychosis was confirmed using the Positive and Negative Syndrome Scale (PANSS), and the inclusion criteria was a baseline score of ≥ 55. Secondary outcomes were scores on the Hamilton Depression Rating Scale, the clinician-rated Global Assessment of Function, and the Assessment of Quality of Life Scale.
The researchers also measured concentrations of the amino acids oxidized by DAAO (D-alanine and L-alanine, D-serine and L-serine).
Although both groups experienced a reduction in total PANSS scores during the study, there were no significant differences in PANSS total score between the BZ and the placebo groups at 12 weeks (endpoint least-square mean difference [SE] −1.2 [2.4] t = −0.49, P = .63).
There were also no significant differences between the groups on all PANSS subscales as well as any of the secondary clinical measures (P < .007).
A total of 122 adverse events (AEs) overall were reported by 66 participants, but rates of AEs were comparable between the BZ and placebo groups at 55% vs. 46% respectively. There were 11 serious AEs reported by 10 participants. Only one of these was related to the study drug.
There were no statistically significant changes in amino acid concentrations between the two groups.
The authors note several limitations of the study, including the possibility that protective agents may need longer times than 12 weeks – possibly as long as 6 to 12 months – to show efficacy. Moreover, the dose of BZ needed to produce a response remains “uncertain.”
“ and should further investigate whether benzoate acts by altering amino acid levels or by reducing oxidative stress in people with schizophrenia,” the authors suggest.
Scott added that further research in patients with treatment-refractory schizophrenia is needed to determine whether BZ “has a role in this patient population.”
The authors conclude that at present, “the routine use of this agent as an adjunctive treatment for early psychosis is not recommended.”
No surprise?
Commenting on the study for Medscape Medical News, Kenji Hashimoto, PhD, Division of Clinical Neuroscience, Chiba University Center for Forensic Mental Health, Chiba, Japan, said that, although previous studies of BZ showed benefit in stable patients with chronic schizophrenia, “it is unlikely that [sodium] benzoate may have beneficial effects in the acute phase of psychosis.”
Hashimoto, who was not involved with the study, noted that BZ is a “weak DAAO inhibitor and that DAAO expression in the frontal cortex of human beings is very low.”
This project was supported by a John Cade Fellowship from the National Health and Medical Research Council and support from the Queensland Centre for Mental Health Research, which receives funding from the Queensland Health Department. Scott is supported by an NHMRC Practitioner Fellowship. The other authors’ disclosures are listed on the original article. Hashimoto has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Inattention to heightened CV risk common theme in clozapine deaths teaser
Death while on clozapine for schizophrenia is often associated with substandard treatment of cardiometabolic risk factors, Sharon Taub, MD, reported at the virtual congress of the European College of Neuropsychopharmacology.
“Inadequate treatment for metabolic syndrome was found to be a mortality predictor while on clozapine therapy. Patients who died were less likely to receive appropriate treatment for hyperlipidemia and type 2 diabetes, despite having been diagnosed with those conditions,” she said in presenting the results of her retrospective cohort study.
“Better preventive care, with special attention to those conditions, might prevent morbidity and improve life expectancy in this population,” concluded Dr. Taub of Geha Mental Health Center in Petah Tikva, Israel, and Tel Aviv University.
She reported on all 1,817 patients on clozapine for schizophrenia included in a large Israeli health care electronic medical records database, of whom 112, or 6.2%, died during 2 years of follow-up. Mortality while on the atypical antipsychotic was associated with a higher prevalence of hyperlipidemia, type 2 diabetes, hypertension, known ischemic heart disease, and a history of acute MI, compared with survivors.
Similarly, only 16.3% of those known to have type 2 diabetes who died while on clozapine were on hypoglycemic agents, compared with 67.1% of diabetic survivors. The between-group difference in the use of antihypertensive drug therapy for patients diagnosed with hypertension – 28.6% in nonsurvivors on clozapine, 40.1% in survivors – did not achieve statistical significance.
In a multivariate analysis adjusted for age, sex, and socioeconomic status, schizophrenia patients with type 2 diabetes who weren’t on hypoglycemic medication were at 695% increased risk of mortality, compared with those who were. Similarly, hyperlipidemic patients on clozapine who weren’t on a statin had a 579% increase in mortality risk.
Patients who died while on clozapine had no increased risk of use of medical services while living in the community.
This evidence of a pattern of inadequate care with regard to management of cardiometabolic risk factors in patients on clozapine is disturbing for several reasons. For one, clozapine is known to be associated with increased risk of serious side effects, including development or worsening of metabolic syndrome. Also, clozapine is an important drug in psychiatric practice: “Clozapine is the only antipsychotic indicated for refractory schizophrenia. It is highly effective in treatment-resistant disease, present in 25%-30% of individuals with schizophrenia,” Dr. Taub noted. “Clozapine is underused, mostly because of severe side effects. Its administration is often postponed.”
She reported having no financial conflicts regarding her study, which was voted by conference attendees one of the top presentations at ECNP 2020.
Death while on clozapine for schizophrenia is often associated with substandard treatment of cardiometabolic risk factors, Sharon Taub, MD, reported at the virtual congress of the European College of Neuropsychopharmacology.
“Inadequate treatment for metabolic syndrome was found to be a mortality predictor while on clozapine therapy. Patients who died were less likely to receive appropriate treatment for hyperlipidemia and type 2 diabetes, despite having been diagnosed with those conditions,” she said in presenting the results of her retrospective cohort study.
“Better preventive care, with special attention to those conditions, might prevent morbidity and improve life expectancy in this population,” concluded Dr. Taub of Geha Mental Health Center in Petah Tikva, Israel, and Tel Aviv University.
She reported on all 1,817 patients on clozapine for schizophrenia included in a large Israeli health care electronic medical records database, of whom 112, or 6.2%, died during 2 years of follow-up. Mortality while on the atypical antipsychotic was associated with a higher prevalence of hyperlipidemia, type 2 diabetes, hypertension, known ischemic heart disease, and a history of acute MI, compared with survivors.
Similarly, only 16.3% of those known to have type 2 diabetes who died while on clozapine were on hypoglycemic agents, compared with 67.1% of diabetic survivors. The between-group difference in the use of antihypertensive drug therapy for patients diagnosed with hypertension – 28.6% in nonsurvivors on clozapine, 40.1% in survivors – did not achieve statistical significance.
In a multivariate analysis adjusted for age, sex, and socioeconomic status, schizophrenia patients with type 2 diabetes who weren’t on hypoglycemic medication were at 695% increased risk of mortality, compared with those who were. Similarly, hyperlipidemic patients on clozapine who weren’t on a statin had a 579% increase in mortality risk.
Patients who died while on clozapine had no increased risk of use of medical services while living in the community.
This evidence of a pattern of inadequate care with regard to management of cardiometabolic risk factors in patients on clozapine is disturbing for several reasons. For one, clozapine is known to be associated with increased risk of serious side effects, including development or worsening of metabolic syndrome. Also, clozapine is an important drug in psychiatric practice: “Clozapine is the only antipsychotic indicated for refractory schizophrenia. It is highly effective in treatment-resistant disease, present in 25%-30% of individuals with schizophrenia,” Dr. Taub noted. “Clozapine is underused, mostly because of severe side effects. Its administration is often postponed.”
She reported having no financial conflicts regarding her study, which was voted by conference attendees one of the top presentations at ECNP 2020.
Death while on clozapine for schizophrenia is often associated with substandard treatment of cardiometabolic risk factors, Sharon Taub, MD, reported at the virtual congress of the European College of Neuropsychopharmacology.
“Inadequate treatment for metabolic syndrome was found to be a mortality predictor while on clozapine therapy. Patients who died were less likely to receive appropriate treatment for hyperlipidemia and type 2 diabetes, despite having been diagnosed with those conditions,” she said in presenting the results of her retrospective cohort study.
“Better preventive care, with special attention to those conditions, might prevent morbidity and improve life expectancy in this population,” concluded Dr. Taub of Geha Mental Health Center in Petah Tikva, Israel, and Tel Aviv University.
She reported on all 1,817 patients on clozapine for schizophrenia included in a large Israeli health care electronic medical records database, of whom 112, or 6.2%, died during 2 years of follow-up. Mortality while on the atypical antipsychotic was associated with a higher prevalence of hyperlipidemia, type 2 diabetes, hypertension, known ischemic heart disease, and a history of acute MI, compared with survivors.
Similarly, only 16.3% of those known to have type 2 diabetes who died while on clozapine were on hypoglycemic agents, compared with 67.1% of diabetic survivors. The between-group difference in the use of antihypertensive drug therapy for patients diagnosed with hypertension – 28.6% in nonsurvivors on clozapine, 40.1% in survivors – did not achieve statistical significance.
In a multivariate analysis adjusted for age, sex, and socioeconomic status, schizophrenia patients with type 2 diabetes who weren’t on hypoglycemic medication were at 695% increased risk of mortality, compared with those who were. Similarly, hyperlipidemic patients on clozapine who weren’t on a statin had a 579% increase in mortality risk.
Patients who died while on clozapine had no increased risk of use of medical services while living in the community.
This evidence of a pattern of inadequate care with regard to management of cardiometabolic risk factors in patients on clozapine is disturbing for several reasons. For one, clozapine is known to be associated with increased risk of serious side effects, including development or worsening of metabolic syndrome. Also, clozapine is an important drug in psychiatric practice: “Clozapine is the only antipsychotic indicated for refractory schizophrenia. It is highly effective in treatment-resistant disease, present in 25%-30% of individuals with schizophrenia,” Dr. Taub noted. “Clozapine is underused, mostly because of severe side effects. Its administration is often postponed.”
She reported having no financial conflicts regarding her study, which was voted by conference attendees one of the top presentations at ECNP 2020.
FROM ECNP 2020