User login
BMI and reproduction – weighing the evidence
Arguably, no topic during an infertility consultation generates more of an emotional reaction than discussing body mass index (BMI), particularly when it is high. Patients have become increasingly sensitive to weight discussions with their physicians because of concerns about body shaming. Among patients with an elevated BMI, criticism on social media of health care professionals’ counseling and a preemptive presentation of “Don’t Weigh Me” cards have become popular responses. Despite the medical evidence on impaired reproduction with an abnormal BMI, patients are choosing to forgo the topic. Research has demonstrated “extensive evidence [of] strong weight bias” in a wide range of health staff.1 A “viral” TikTok study revealed that medical “gaslighting” founded in weight stigma and bias is harmful, as reported on KevinMD.com.2 This month, we review the effect of abnormal BMI, both high and low, on reproduction and pregnancy.
A method to assess relative weight was first described in 1832 as its ratio in kilograms divided by the square of the height in meters, or the Quetelet Index. The search for a functional assessment of relative body weight began after World War II when reports by actuaries noted the increased mortality of overweight policyholders. The relationship between weight and cardiovascular disease was further revealed in epidemiologic studies. The Quetelet Index became the BMI in 1972.3
Weight measurement is a mainstay in the assessment of a patient’s vital signs along with blood pressure, pulse rate, respiration rate, and temperature. Weight is vital to the calculation of medication dosage – for instance, administration of conscious sedative drugs, methotrexate, and gonadotropins. Some state boards of medicine, such as Florida, have a limitation on patient BMI at office-based surgery centers (40 kg/m2).
Obesity is a disease
As reported by the World Health Organization in 2022, the disease of obesity is an epidemic afflicting more than 1 billion people worldwide, or 1 in 8 individuals globally.4 The health implications of an elevated BMI include increased mortality, diabetes, heart disease, and stroke, physical limitations to activities of daily living, and complications affecting reproduction.
Female obesity is related to poorer outcomes in natural and assisted conception, including an increased risk of miscarriage. Compared with normal-weight women, those with obesity are three times more likely to have ovulatory dysfunction,5 infertility,6 a lower chance for conception,7 higher rate of miscarriage, and low birth weight.8,9During pregnancy, women with obesity have three to four times higher rates of gestational diabetes and preeclampsia,10 as well as likelihood of delivering preterm,11 having a fetus with macrosomia and birth defects, and a 1.3- to 2.1-times higher risk of stillbirth.12
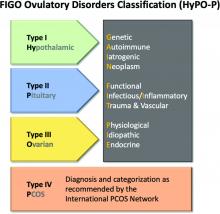
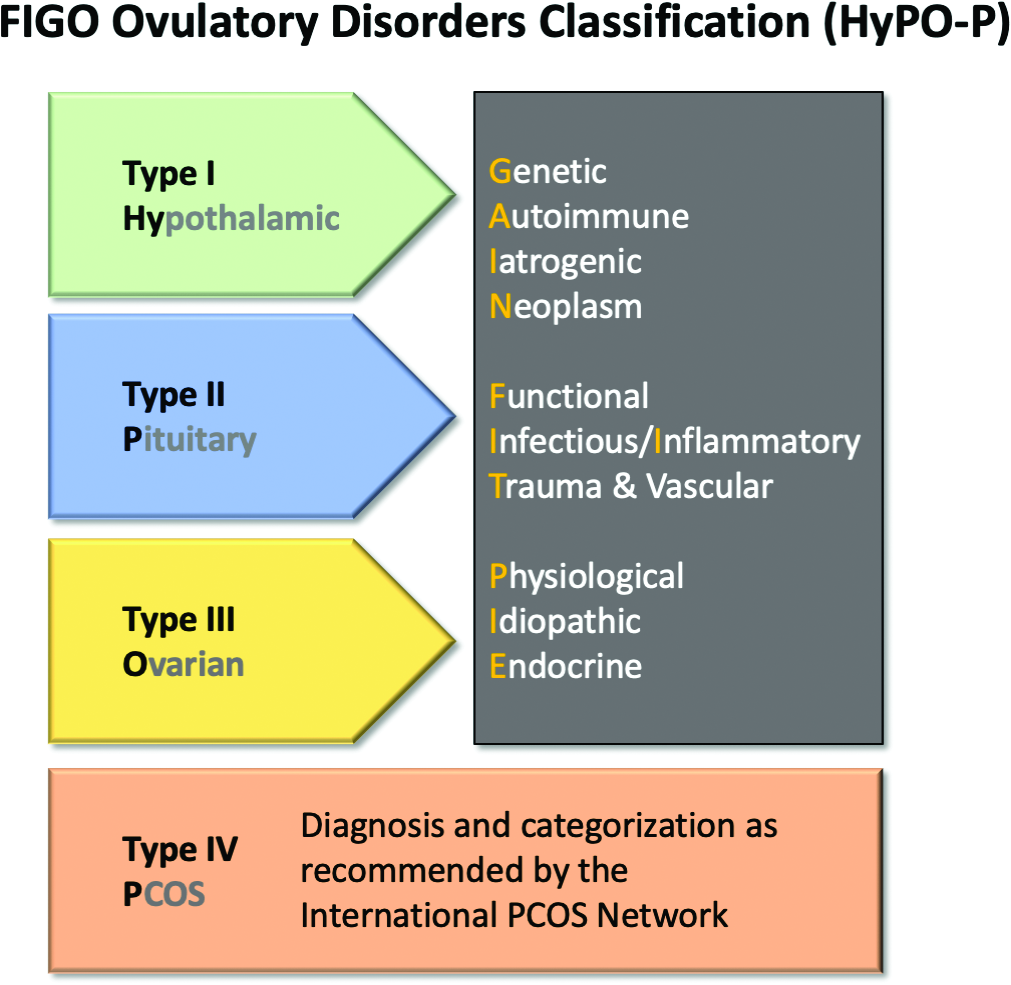
Obesity is present in 40%-80% of women with polycystic ovary syndrome,13 the most common cause of ovulatory dysfunction from dysregulation of the hypothalamic-pituitary-ovarian axis. While PCOS is associated with reproductive and metabolic consequences, even in regularly ovulating women, increasing obesity appears to be associated with decreasing spontaneous pregnancy rates and increased time to pregnancy.14
Obesity and IVF
Women with obesity have reduced success with assisted reproductive technology, an increased number of canceled cycles, and poorer quality oocytes retrieved. A prospective cohort study of nearly 2,000 women reported that every 5 kg of body weight increase (from the patient’s baseline weight at age 18) was associated with a 5% increase in the mean duration of time required for conception (95% confidence interval, 3%-7%).15 Given that approximately 90% of these women had regular menstrual cycles, ovulatory dysfunction was not the suspected pathophysiology.
A meta-analysis of 21 cohort studies reported a lower likelihood of live birth following in vitro fertilization for women with obesity, compared with normal-weight women (risk ratio, 0.85; 95% CI, 0.82-0.87).16 A further subgroup analysis that evaluated only women with PCOS showed a reduction in the live birth rate following IVF for individuals with obesity, compared with normal-weight individuals (RR, 0.78; 95% CI, 0.74-0.82).
In a retrospective study of almost 500,000 fresh autologous IVF cycles, women with obesity had a 6% reduction in pregnancy rates and a 13% reduction in live birth rates, compared with normal-weight women. Both high and low BMI were associated with an increased risk of low birth weight and preterm delivery.17 The live birth rates per transfer for normal-weight and higher-weight women were 38% and 33%, respectively.
Contrarily, a randomized controlled trial showed that an intensive weight-reduction program resulted in a large weight loss but did not substantially affect live birth rates in women with obesity scheduled for IVF.18
Low BMI
A noteworthy cause of low BMI is functional hypothalamic amenorrhea (FHA), a disorder with low energy availability either from decreased caloric intake and/or excessive energy expenditure associated with eating disorders, excessive exercise, and stress. Consequently, a reduced GnRH drive results in a decreased pulse frequency and amplitude leading to low levels of follicle-stimulating hormone and luteinizing hormone, resulting in anovulation. Correction of lifestyle behaviors related to FHA can restore menstrual cycles. After normal weight is achieved, it appears unlikely that fertility is affected.19 In 47% of adolescent patients with anorexia, menses spontaneously returned within the first 12 months after admission, with an improved prognosis in secondary over primary amenorrhea.20,21 Interestingly, mildly and significantly underweight infertile women have pregnancy and live birth rates similar to normal-weight patients after IVF treatment.22
Pregnancy is complicated in underweight women, resulting in an increased risk of anemia, fetal growth retardation, and low birth weight, as well as preterm birth.21
Take-home message
The extremes of BMI both impair natural reproduction. Elevated BMI reduces success with IVF but rapid weight loss prior to IVF does not improve outcomes. A normal BMI is the goal for optimal reproductive and pregnancy health.
Dr. Trolice is director of the IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Talumaa B et al. Obesity Rev. 2022;23:e13494.
2. https://bit.ly/3rHCivE.
3. Eknoyan G. Nephrol Dial Transplant. 2008;23:47-51.
4. Wells JCK. Dis Models Mech. 2012;5:595-607.
5. Brewer CJ and Balen AH. Reproduction. 2010;140:347-64.
6. Silvestris E et al. Reprod Biol Endocrinol. 2018;16:22.
7. Wise LA et al. Hum Reprod. 2010;25:253-64.
8. Bellver J. Curr Opin Obstet Gynecol. 2022;34:114-21.
9. Dickey RP et al. Am J Obstet Gynecol. 2013;209:349.e1.
10. Alwash SM et al. Obes Res Clin Pract. 2021;15:425-30.
11. Cnattingius S et al. JAMA. 2013;309:2362-70.
12. Aune D et al. JAMA. 2014;311:1536-46.
13. Sam S. Obes Manag. 2007;3:69-73.
14. van der Steeg JW et al. Hum Reprod. 2008;23:324-8.
15. Gaskins AJ et al. Obstet Gynecol. 2015;126:850-8.
16. Sermondade N et al. Hum Reprod Update. 2019;25:439-519.
17. Kawwass JF et al. Fertil Steril. 2016;106[7]:1742-50.
18. Einarsson S et al. Hum Reprod. 2017;32:1621-30.
19. Chaer R et al. Diseases. 2020;8:46.
20. Dempfle A et al. Psychiatry. 2013;13:308.
21. Verma A and Shrimali L. J Clin Diagn Res. 2012;6:1531-3.
22. Romanski PA et al. Reprod Biomed Online. 2020;42:366-74.
Arguably, no topic during an infertility consultation generates more of an emotional reaction than discussing body mass index (BMI), particularly when it is high. Patients have become increasingly sensitive to weight discussions with their physicians because of concerns about body shaming. Among patients with an elevated BMI, criticism on social media of health care professionals’ counseling and a preemptive presentation of “Don’t Weigh Me” cards have become popular responses. Despite the medical evidence on impaired reproduction with an abnormal BMI, patients are choosing to forgo the topic. Research has demonstrated “extensive evidence [of] strong weight bias” in a wide range of health staff.1 A “viral” TikTok study revealed that medical “gaslighting” founded in weight stigma and bias is harmful, as reported on KevinMD.com.2 This month, we review the effect of abnormal BMI, both high and low, on reproduction and pregnancy.
A method to assess relative weight was first described in 1832 as its ratio in kilograms divided by the square of the height in meters, or the Quetelet Index. The search for a functional assessment of relative body weight began after World War II when reports by actuaries noted the increased mortality of overweight policyholders. The relationship between weight and cardiovascular disease was further revealed in epidemiologic studies. The Quetelet Index became the BMI in 1972.3
Weight measurement is a mainstay in the assessment of a patient’s vital signs along with blood pressure, pulse rate, respiration rate, and temperature. Weight is vital to the calculation of medication dosage – for instance, administration of conscious sedative drugs, methotrexate, and gonadotropins. Some state boards of medicine, such as Florida, have a limitation on patient BMI at office-based surgery centers (40 kg/m2).
Obesity is a disease
As reported by the World Health Organization in 2022, the disease of obesity is an epidemic afflicting more than 1 billion people worldwide, or 1 in 8 individuals globally.4 The health implications of an elevated BMI include increased mortality, diabetes, heart disease, and stroke, physical limitations to activities of daily living, and complications affecting reproduction.
Female obesity is related to poorer outcomes in natural and assisted conception, including an increased risk of miscarriage. Compared with normal-weight women, those with obesity are three times more likely to have ovulatory dysfunction,5 infertility,6 a lower chance for conception,7 higher rate of miscarriage, and low birth weight.8,9During pregnancy, women with obesity have three to four times higher rates of gestational diabetes and preeclampsia,10 as well as likelihood of delivering preterm,11 having a fetus with macrosomia and birth defects, and a 1.3- to 2.1-times higher risk of stillbirth.12
Obesity is present in 40%-80% of women with polycystic ovary syndrome,13 the most common cause of ovulatory dysfunction from dysregulation of the hypothalamic-pituitary-ovarian axis. While PCOS is associated with reproductive and metabolic consequences, even in regularly ovulating women, increasing obesity appears to be associated with decreasing spontaneous pregnancy rates and increased time to pregnancy.14
Obesity and IVF
Women with obesity have reduced success with assisted reproductive technology, an increased number of canceled cycles, and poorer quality oocytes retrieved. A prospective cohort study of nearly 2,000 women reported that every 5 kg of body weight increase (from the patient’s baseline weight at age 18) was associated with a 5% increase in the mean duration of time required for conception (95% confidence interval, 3%-7%).15 Given that approximately 90% of these women had regular menstrual cycles, ovulatory dysfunction was not the suspected pathophysiology.
A meta-analysis of 21 cohort studies reported a lower likelihood of live birth following in vitro fertilization for women with obesity, compared with normal-weight women (risk ratio, 0.85; 95% CI, 0.82-0.87).16 A further subgroup analysis that evaluated only women with PCOS showed a reduction in the live birth rate following IVF for individuals with obesity, compared with normal-weight individuals (RR, 0.78; 95% CI, 0.74-0.82).
In a retrospective study of almost 500,000 fresh autologous IVF cycles, women with obesity had a 6% reduction in pregnancy rates and a 13% reduction in live birth rates, compared with normal-weight women. Both high and low BMI were associated with an increased risk of low birth weight and preterm delivery.17 The live birth rates per transfer for normal-weight and higher-weight women were 38% and 33%, respectively.
Contrarily, a randomized controlled trial showed that an intensive weight-reduction program resulted in a large weight loss but did not substantially affect live birth rates in women with obesity scheduled for IVF.18
Low BMI
A noteworthy cause of low BMI is functional hypothalamic amenorrhea (FHA), a disorder with low energy availability either from decreased caloric intake and/or excessive energy expenditure associated with eating disorders, excessive exercise, and stress. Consequently, a reduced GnRH drive results in a decreased pulse frequency and amplitude leading to low levels of follicle-stimulating hormone and luteinizing hormone, resulting in anovulation. Correction of lifestyle behaviors related to FHA can restore menstrual cycles. After normal weight is achieved, it appears unlikely that fertility is affected.19 In 47% of adolescent patients with anorexia, menses spontaneously returned within the first 12 months after admission, with an improved prognosis in secondary over primary amenorrhea.20,21 Interestingly, mildly and significantly underweight infertile women have pregnancy and live birth rates similar to normal-weight patients after IVF treatment.22
Pregnancy is complicated in underweight women, resulting in an increased risk of anemia, fetal growth retardation, and low birth weight, as well as preterm birth.21
Take-home message
The extremes of BMI both impair natural reproduction. Elevated BMI reduces success with IVF but rapid weight loss prior to IVF does not improve outcomes. A normal BMI is the goal for optimal reproductive and pregnancy health.
Dr. Trolice is director of the IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Talumaa B et al. Obesity Rev. 2022;23:e13494.
2. https://bit.ly/3rHCivE.
3. Eknoyan G. Nephrol Dial Transplant. 2008;23:47-51.
4. Wells JCK. Dis Models Mech. 2012;5:595-607.
5. Brewer CJ and Balen AH. Reproduction. 2010;140:347-64.
6. Silvestris E et al. Reprod Biol Endocrinol. 2018;16:22.
7. Wise LA et al. Hum Reprod. 2010;25:253-64.
8. Bellver J. Curr Opin Obstet Gynecol. 2022;34:114-21.
9. Dickey RP et al. Am J Obstet Gynecol. 2013;209:349.e1.
10. Alwash SM et al. Obes Res Clin Pract. 2021;15:425-30.
11. Cnattingius S et al. JAMA. 2013;309:2362-70.
12. Aune D et al. JAMA. 2014;311:1536-46.
13. Sam S. Obes Manag. 2007;3:69-73.
14. van der Steeg JW et al. Hum Reprod. 2008;23:324-8.
15. Gaskins AJ et al. Obstet Gynecol. 2015;126:850-8.
16. Sermondade N et al. Hum Reprod Update. 2019;25:439-519.
17. Kawwass JF et al. Fertil Steril. 2016;106[7]:1742-50.
18. Einarsson S et al. Hum Reprod. 2017;32:1621-30.
19. Chaer R et al. Diseases. 2020;8:46.
20. Dempfle A et al. Psychiatry. 2013;13:308.
21. Verma A and Shrimali L. J Clin Diagn Res. 2012;6:1531-3.
22. Romanski PA et al. Reprod Biomed Online. 2020;42:366-74.
Arguably, no topic during an infertility consultation generates more of an emotional reaction than discussing body mass index (BMI), particularly when it is high. Patients have become increasingly sensitive to weight discussions with their physicians because of concerns about body shaming. Among patients with an elevated BMI, criticism on social media of health care professionals’ counseling and a preemptive presentation of “Don’t Weigh Me” cards have become popular responses. Despite the medical evidence on impaired reproduction with an abnormal BMI, patients are choosing to forgo the topic. Research has demonstrated “extensive evidence [of] strong weight bias” in a wide range of health staff.1 A “viral” TikTok study revealed that medical “gaslighting” founded in weight stigma and bias is harmful, as reported on KevinMD.com.2 This month, we review the effect of abnormal BMI, both high and low, on reproduction and pregnancy.
A method to assess relative weight was first described in 1832 as its ratio in kilograms divided by the square of the height in meters, or the Quetelet Index. The search for a functional assessment of relative body weight began after World War II when reports by actuaries noted the increased mortality of overweight policyholders. The relationship between weight and cardiovascular disease was further revealed in epidemiologic studies. The Quetelet Index became the BMI in 1972.3
Weight measurement is a mainstay in the assessment of a patient’s vital signs along with blood pressure, pulse rate, respiration rate, and temperature. Weight is vital to the calculation of medication dosage – for instance, administration of conscious sedative drugs, methotrexate, and gonadotropins. Some state boards of medicine, such as Florida, have a limitation on patient BMI at office-based surgery centers (40 kg/m2).
Obesity is a disease
As reported by the World Health Organization in 2022, the disease of obesity is an epidemic afflicting more than 1 billion people worldwide, or 1 in 8 individuals globally.4 The health implications of an elevated BMI include increased mortality, diabetes, heart disease, and stroke, physical limitations to activities of daily living, and complications affecting reproduction.
Female obesity is related to poorer outcomes in natural and assisted conception, including an increased risk of miscarriage. Compared with normal-weight women, those with obesity are three times more likely to have ovulatory dysfunction,5 infertility,6 a lower chance for conception,7 higher rate of miscarriage, and low birth weight.8,9During pregnancy, women with obesity have three to four times higher rates of gestational diabetes and preeclampsia,10 as well as likelihood of delivering preterm,11 having a fetus with macrosomia and birth defects, and a 1.3- to 2.1-times higher risk of stillbirth.12
Obesity is present in 40%-80% of women with polycystic ovary syndrome,13 the most common cause of ovulatory dysfunction from dysregulation of the hypothalamic-pituitary-ovarian axis. While PCOS is associated with reproductive and metabolic consequences, even in regularly ovulating women, increasing obesity appears to be associated with decreasing spontaneous pregnancy rates and increased time to pregnancy.14
Obesity and IVF
Women with obesity have reduced success with assisted reproductive technology, an increased number of canceled cycles, and poorer quality oocytes retrieved. A prospective cohort study of nearly 2,000 women reported that every 5 kg of body weight increase (from the patient’s baseline weight at age 18) was associated with a 5% increase in the mean duration of time required for conception (95% confidence interval, 3%-7%).15 Given that approximately 90% of these women had regular menstrual cycles, ovulatory dysfunction was not the suspected pathophysiology.
A meta-analysis of 21 cohort studies reported a lower likelihood of live birth following in vitro fertilization for women with obesity, compared with normal-weight women (risk ratio, 0.85; 95% CI, 0.82-0.87).16 A further subgroup analysis that evaluated only women with PCOS showed a reduction in the live birth rate following IVF for individuals with obesity, compared with normal-weight individuals (RR, 0.78; 95% CI, 0.74-0.82).
In a retrospective study of almost 500,000 fresh autologous IVF cycles, women with obesity had a 6% reduction in pregnancy rates and a 13% reduction in live birth rates, compared with normal-weight women. Both high and low BMI were associated with an increased risk of low birth weight and preterm delivery.17 The live birth rates per transfer for normal-weight and higher-weight women were 38% and 33%, respectively.
Contrarily, a randomized controlled trial showed that an intensive weight-reduction program resulted in a large weight loss but did not substantially affect live birth rates in women with obesity scheduled for IVF.18
Low BMI
A noteworthy cause of low BMI is functional hypothalamic amenorrhea (FHA), a disorder with low energy availability either from decreased caloric intake and/or excessive energy expenditure associated with eating disorders, excessive exercise, and stress. Consequently, a reduced GnRH drive results in a decreased pulse frequency and amplitude leading to low levels of follicle-stimulating hormone and luteinizing hormone, resulting in anovulation. Correction of lifestyle behaviors related to FHA can restore menstrual cycles. After normal weight is achieved, it appears unlikely that fertility is affected.19 In 47% of adolescent patients with anorexia, menses spontaneously returned within the first 12 months after admission, with an improved prognosis in secondary over primary amenorrhea.20,21 Interestingly, mildly and significantly underweight infertile women have pregnancy and live birth rates similar to normal-weight patients after IVF treatment.22
Pregnancy is complicated in underweight women, resulting in an increased risk of anemia, fetal growth retardation, and low birth weight, as well as preterm birth.21
Take-home message
The extremes of BMI both impair natural reproduction. Elevated BMI reduces success with IVF but rapid weight loss prior to IVF does not improve outcomes. A normal BMI is the goal for optimal reproductive and pregnancy health.
Dr. Trolice is director of the IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Talumaa B et al. Obesity Rev. 2022;23:e13494.
2. https://bit.ly/3rHCivE.
3. Eknoyan G. Nephrol Dial Transplant. 2008;23:47-51.
4. Wells JCK. Dis Models Mech. 2012;5:595-607.
5. Brewer CJ and Balen AH. Reproduction. 2010;140:347-64.
6. Silvestris E et al. Reprod Biol Endocrinol. 2018;16:22.
7. Wise LA et al. Hum Reprod. 2010;25:253-64.
8. Bellver J. Curr Opin Obstet Gynecol. 2022;34:114-21.
9. Dickey RP et al. Am J Obstet Gynecol. 2013;209:349.e1.
10. Alwash SM et al. Obes Res Clin Pract. 2021;15:425-30.
11. Cnattingius S et al. JAMA. 2013;309:2362-70.
12. Aune D et al. JAMA. 2014;311:1536-46.
13. Sam S. Obes Manag. 2007;3:69-73.
14. van der Steeg JW et al. Hum Reprod. 2008;23:324-8.
15. Gaskins AJ et al. Obstet Gynecol. 2015;126:850-8.
16. Sermondade N et al. Hum Reprod Update. 2019;25:439-519.
17. Kawwass JF et al. Fertil Steril. 2016;106[7]:1742-50.
18. Einarsson S et al. Hum Reprod. 2017;32:1621-30.
19. Chaer R et al. Diseases. 2020;8:46.
20. Dempfle A et al. Psychiatry. 2013;13:308.
21. Verma A and Shrimali L. J Clin Diagn Res. 2012;6:1531-3.
22. Romanski PA et al. Reprod Biomed Online. 2020;42:366-74.
Fertility physicians say they lack access to miscarriage drugs
In a survey taken before the Supreme Court’s Dobbs ruling regarding abortion rights, two-thirds of assisted reproduction technology (ART) physicians who don’t offer mifepristone/misoprostol to patients with early pregnancy loss (EPL) reported that they lack access to the drugs.
The numbers are likely higher now. In the wake of the court ruling, some physicians in states with new abortion restrictions fear they won’t be able to properly treat women with miscarriages. Access to mifepristone, a component of medication abortions along with misoprostol, is at the center of their concerns.
“These restrictions that were put in place to restrict abortion care have far-reaching implications regarding miscarriages and early pregnancy loss and the assisted reproduction community is not immune,” obstetrics and gynecology specialist Zachary Anderson, MD, a resident physician at the University of Southern California, Los Angeles, said in an interview. He presented the findings at the American Society for Reproductive Medicine’s 2022 meeting.
Early pregnancy loss – defined as a miscarriage within 12 weeks and 6 days of conception – is common in all pregnancies and affects an estimated 15% of those who rely on in vitro fertilization (IVF). In women who conceive through intrauterine insemination or IVF, “an abnormal karyotype embryo/fetus is the cause of miscarriage in more than two-thirds of cases,” Mark P. Trolice, MD, director of the IVF Center and professor of obstetrics and gynecology at the University of Central Florida, Orlando, said in an interview. “The options of management are observation – with no ability to determine when passage of the products of conception will occur – vs. mifepristone/misoprostol or suction D&C.”
Dr. Trolice added that “most woman select the medical treatment protocol, which is 200 mg mifepristone orally followed by 800 mcg misoprostol vaginally 24 hours later. If no signs of heavy bleeding occur after 3 hours following misoprostol, the patient should repeat the dose of 800 micrograms vaginally.”
According to the Reuters news service, some abortion bans target mifepristone. In October 2022, the American College of Obstetricians and Gynecologists asked the Food and Drug Administration to approve mifepristone for use in miscarriage management; such use is now off label, although it is approved to end early pregnancies in conjunction with misoprostol.
For the new study, researchers sent anonymous surveys to 826 members of the Society of Reproductive Endocrinology and Infertility and received 101 responses (12% response rate, 51% women, 86% non-Hispanic White, average age 52, 52% urban, and 51% in private practice).
More than two-thirds (70%) said they diagnosed early pregnancy loss at least once a week; 47% prefer treatment with misoprostol alone, 18% surgery in an operating room, 15% expectant management (monitoring a miscarriage as it occurs without medical intervention), 10% surgery in the office, and 3% mifepristone-misoprostol.
Of those who don’t offer mifepristone-misoprostol, 68% said they lack access, and 26% said they lack familiarity with the treatment.
Study coauthor Brian T. Nguyen, MD, MSc, assistant professor of obstetrics and gynecology at USC, said in an interview that mifepristone, a highly effective drug, is treated differently from other medications “for no good reason.”
Dr. Anderson, who led the study, urged colleagues to get the appropriate certification to prescribe mifepristone. “Providers overestimate how difficult it is to become certified to prescribe it,” he said.
Dr. Trolice, who is familiar with the study findings, said the response rate is low, and the results might be biased because those with preconceived opinions may be more likely to respond.
However, he said, “The results are not surprising in that medication is more commonly preferred (nearly 50%) given the devastation of a miscarriage and the desire to expedite resolution. Approximately one-third prefer surgical management, which would allow for genetic testing of the embryo/fetus to potentially determine a cause of the pregnancy loss.”
As for the medications used to treat early pregnancy loss, many ART physicians “treat pregnancy loss with misoprostol both pre- and post Dobbs,” he said. “The difficulty in obtaining mifepristone remains.”
The study authors and Dr. Trolice report no disclosures.
In a survey taken before the Supreme Court’s Dobbs ruling regarding abortion rights, two-thirds of assisted reproduction technology (ART) physicians who don’t offer mifepristone/misoprostol to patients with early pregnancy loss (EPL) reported that they lack access to the drugs.
The numbers are likely higher now. In the wake of the court ruling, some physicians in states with new abortion restrictions fear they won’t be able to properly treat women with miscarriages. Access to mifepristone, a component of medication abortions along with misoprostol, is at the center of their concerns.
“These restrictions that were put in place to restrict abortion care have far-reaching implications regarding miscarriages and early pregnancy loss and the assisted reproduction community is not immune,” obstetrics and gynecology specialist Zachary Anderson, MD, a resident physician at the University of Southern California, Los Angeles, said in an interview. He presented the findings at the American Society for Reproductive Medicine’s 2022 meeting.
Early pregnancy loss – defined as a miscarriage within 12 weeks and 6 days of conception – is common in all pregnancies and affects an estimated 15% of those who rely on in vitro fertilization (IVF). In women who conceive through intrauterine insemination or IVF, “an abnormal karyotype embryo/fetus is the cause of miscarriage in more than two-thirds of cases,” Mark P. Trolice, MD, director of the IVF Center and professor of obstetrics and gynecology at the University of Central Florida, Orlando, said in an interview. “The options of management are observation – with no ability to determine when passage of the products of conception will occur – vs. mifepristone/misoprostol or suction D&C.”
Dr. Trolice added that “most woman select the medical treatment protocol, which is 200 mg mifepristone orally followed by 800 mcg misoprostol vaginally 24 hours later. If no signs of heavy bleeding occur after 3 hours following misoprostol, the patient should repeat the dose of 800 micrograms vaginally.”
According to the Reuters news service, some abortion bans target mifepristone. In October 2022, the American College of Obstetricians and Gynecologists asked the Food and Drug Administration to approve mifepristone for use in miscarriage management; such use is now off label, although it is approved to end early pregnancies in conjunction with misoprostol.
For the new study, researchers sent anonymous surveys to 826 members of the Society of Reproductive Endocrinology and Infertility and received 101 responses (12% response rate, 51% women, 86% non-Hispanic White, average age 52, 52% urban, and 51% in private practice).
More than two-thirds (70%) said they diagnosed early pregnancy loss at least once a week; 47% prefer treatment with misoprostol alone, 18% surgery in an operating room, 15% expectant management (monitoring a miscarriage as it occurs without medical intervention), 10% surgery in the office, and 3% mifepristone-misoprostol.
Of those who don’t offer mifepristone-misoprostol, 68% said they lack access, and 26% said they lack familiarity with the treatment.
Study coauthor Brian T. Nguyen, MD, MSc, assistant professor of obstetrics and gynecology at USC, said in an interview that mifepristone, a highly effective drug, is treated differently from other medications “for no good reason.”
Dr. Anderson, who led the study, urged colleagues to get the appropriate certification to prescribe mifepristone. “Providers overestimate how difficult it is to become certified to prescribe it,” he said.
Dr. Trolice, who is familiar with the study findings, said the response rate is low, and the results might be biased because those with preconceived opinions may be more likely to respond.
However, he said, “The results are not surprising in that medication is more commonly preferred (nearly 50%) given the devastation of a miscarriage and the desire to expedite resolution. Approximately one-third prefer surgical management, which would allow for genetic testing of the embryo/fetus to potentially determine a cause of the pregnancy loss.”
As for the medications used to treat early pregnancy loss, many ART physicians “treat pregnancy loss with misoprostol both pre- and post Dobbs,” he said. “The difficulty in obtaining mifepristone remains.”
The study authors and Dr. Trolice report no disclosures.
In a survey taken before the Supreme Court’s Dobbs ruling regarding abortion rights, two-thirds of assisted reproduction technology (ART) physicians who don’t offer mifepristone/misoprostol to patients with early pregnancy loss (EPL) reported that they lack access to the drugs.
The numbers are likely higher now. In the wake of the court ruling, some physicians in states with new abortion restrictions fear they won’t be able to properly treat women with miscarriages. Access to mifepristone, a component of medication abortions along with misoprostol, is at the center of their concerns.
“These restrictions that were put in place to restrict abortion care have far-reaching implications regarding miscarriages and early pregnancy loss and the assisted reproduction community is not immune,” obstetrics and gynecology specialist Zachary Anderson, MD, a resident physician at the University of Southern California, Los Angeles, said in an interview. He presented the findings at the American Society for Reproductive Medicine’s 2022 meeting.
Early pregnancy loss – defined as a miscarriage within 12 weeks and 6 days of conception – is common in all pregnancies and affects an estimated 15% of those who rely on in vitro fertilization (IVF). In women who conceive through intrauterine insemination or IVF, “an abnormal karyotype embryo/fetus is the cause of miscarriage in more than two-thirds of cases,” Mark P. Trolice, MD, director of the IVF Center and professor of obstetrics and gynecology at the University of Central Florida, Orlando, said in an interview. “The options of management are observation – with no ability to determine when passage of the products of conception will occur – vs. mifepristone/misoprostol or suction D&C.”
Dr. Trolice added that “most woman select the medical treatment protocol, which is 200 mg mifepristone orally followed by 800 mcg misoprostol vaginally 24 hours later. If no signs of heavy bleeding occur after 3 hours following misoprostol, the patient should repeat the dose of 800 micrograms vaginally.”
According to the Reuters news service, some abortion bans target mifepristone. In October 2022, the American College of Obstetricians and Gynecologists asked the Food and Drug Administration to approve mifepristone for use in miscarriage management; such use is now off label, although it is approved to end early pregnancies in conjunction with misoprostol.
For the new study, researchers sent anonymous surveys to 826 members of the Society of Reproductive Endocrinology and Infertility and received 101 responses (12% response rate, 51% women, 86% non-Hispanic White, average age 52, 52% urban, and 51% in private practice).
More than two-thirds (70%) said they diagnosed early pregnancy loss at least once a week; 47% prefer treatment with misoprostol alone, 18% surgery in an operating room, 15% expectant management (monitoring a miscarriage as it occurs without medical intervention), 10% surgery in the office, and 3% mifepristone-misoprostol.
Of those who don’t offer mifepristone-misoprostol, 68% said they lack access, and 26% said they lack familiarity with the treatment.
Study coauthor Brian T. Nguyen, MD, MSc, assistant professor of obstetrics and gynecology at USC, said in an interview that mifepristone, a highly effective drug, is treated differently from other medications “for no good reason.”
Dr. Anderson, who led the study, urged colleagues to get the appropriate certification to prescribe mifepristone. “Providers overestimate how difficult it is to become certified to prescribe it,” he said.
Dr. Trolice, who is familiar with the study findings, said the response rate is low, and the results might be biased because those with preconceived opinions may be more likely to respond.
However, he said, “The results are not surprising in that medication is more commonly preferred (nearly 50%) given the devastation of a miscarriage and the desire to expedite resolution. Approximately one-third prefer surgical management, which would allow for genetic testing of the embryo/fetus to potentially determine a cause of the pregnancy loss.”
As for the medications used to treat early pregnancy loss, many ART physicians “treat pregnancy loss with misoprostol both pre- and post Dobbs,” he said. “The difficulty in obtaining mifepristone remains.”
The study authors and Dr. Trolice report no disclosures.
FROM ASRM 2022
Early estrogen loss increases cardiovascular risk in women
The relationship between estrogen levels and heart health makes it particularly important for clinicians to be aware of those patients who might be at risk for cardiovascular disease despite not having other traditional risk factors, according to a presentation Oct. 12 at the North American Menopause Society annual meeting in Atlanta.
”Endogenous estrogens are protective for cardiovascular disease in premenopausal women,” Chrisandra L. Shufelt, MD, chair of the division of general internal medicine and associate director of the Women’s Health Research Center at Mayo Clinic in Jacksonville, Fla., told attendees. Yet, “a substantial population of young women are dying prematurely from cardiovascular disease,” with rates of cardiovascular death increasing in women aged 35-44 even as rates have decreased in postmenopausal women and in men. One potential reason may be premature estrogen loss.
Dr. Shufelt reminded attendees of four major causes of premature estrogen loss: Natural premature menopause, surgical menopause, chemotherapy-induced menopause, and premature ovarian insufficiency. But she would go on to discuss a less widely recognized condition, functional hypothalamic amenorrhea, that also may be contributing to increased cardiovascular risk.
First, Dr. Shufelt reviewed the evidence supporting the relationship between estrogen and cardiovascular health, starting with the Framingham study’s findings that cardiovascular disease is approximately two to four times more common in postmenopausal women than in premenopausal women, depending on the age range.
“Menopause at an early age, particularly under the age of 40, matters,” Dr. Shufelt said. “So we should be discussing this with our patients.”
Surgical menopause makes a difference to cardiovascular health as well, she said. In women under age 35, for example, the risk of a nonfatal heart attack in those with a bilateral oophorectomy was 7.7 times greater than in women who retained both ovaries and their uterus, and 1.5 times greater in women who had a hysterectomy without bilateral oophorectomy.
In a 2019 study, surgical premature menopause was associated with an 87% increased risk of heart disease even after researchers accounted for age, cardiovascular risk factors, and some forms of hormone therapy. The increased risk from natural premature menopause, on the other hand, was lower – a 36% increased risk of heart disease – compared with those producing endogenous hormones. Although randomized controlled trials are unavailable and unlikely to be done, the Nurses’ Health Study and the Danish Nurses Cohort Study, both observational studies, found that heart disease risk was diminished in those taking hormone therapy after surgical premature menopause.
Recommendations for premature or early menopause, from a wide range of different medical societies including NAMS, are that women without contraindications be given estrogen-based hormone therapy until the average age of natural menopause. Though not included in the same guidance, research has also shown that estrogen after oophorectomy does not increase the risk of breast cancer in women with a BRCA1 mutation, Dr. Shufelt said. Hormone therapy for premature or early menopause should adequately replace the levels women have lost and that means younger menopausal women often need higher doses than what older women receive, such as 2 mg/day of oral estradiol rather than the standard doses of 0.5 or 1 mg/day.
Functional hypothalamic amenorrhea and cardiovascular risk
Dr. Shufelt then discussed functional hypothalamic amenorrhea (hypogonadotropic hypogonadism), a common type of secondary amenorrhea that affects at least 1.4 million U.S. women. Diagnosis includes lack of a period for at least 3 months in someone who previously menstruated plus lab values below 50 pg/mL for estradiol, below 10 mIU/L for follicle stimulating hormone, and below 10 mIU/L for luteinizing hormone. Causes of this reversible form of infertility can include stress, overexercising, undereating, or some combination of these, plus an underlying genetic predisposition.
“After ruling out polycystic ovary syndrome, prolactinoma, and thyroid dysfunction, clinicians need to consider the diagnosis of hypothalamic amenorrhea,” Dr. Shufelt said. This condition goes beyond low estrogen levels: Women have elevated cortisol, low thyroid levels, low leptin levels, and increased ghrelin.
”This is not going away,” Dr. Shufelt said, sharing data on stress levels among U.S. adults, particularly Gen Z and millennial adults, noting that the ongoing “national mental health crisis” may be contributing to functional hypothalamic amenorrhea.
A 2020 substudy from the Nurses’ Health Study II found an increased risk of premature death in those who didn’t have a period or always had irregular periods starting as early as 14-17 years old. The increased risk of premature death rose with age in those with irregular or absent cycles – a 37% higher risk in 18- to 22-year-olds and a 39% increased risk in 29- to 46-year-olds.
But clinicians aren’t adequately identifying the “phenotype of the hypothalamic women,” Dr. Shufelt said, despite research showing overlap between hypothalamic amenorrhea and a higher risk of cardiovascular disease. Hypothalamic amenorrhea is so understudied that the last original research on the topic was in 2008, Dr. Shufelt said in an interview. ”No research except mine has been done to evaluate heart health in these young women,” she said.
Dr. Shufelt described a study she led involving 30 women with functional hypothalamic amenorrhea, 29 women with normal menstrual cycles, and 30 women who were recently menopausal and not on hormone therapy. The women with hypothalamic amenorrhea had average stress levels but their depression scores were higher than those of the other two groups.
The results showed that women with hypothalamic amenorrhea had lower estradiol and leptin levels and higher testosterone levels compared with the control group, and they had higher cortisol levels than those of both groups. Despite having similar body mass indexes as the control and menopausal groups, women with hypothalamic amenorrhea had lower blood pressure than that of the other two groups, yet they had higher cholesterol levels than those of the control group. EndoPAT© (Itamar Medical) testing showed that they had poor vascular function.
“In fact, one-third of the women [with hypothalamic amenorrhea] entered the trial with a diagnosis of what would be considered endothelial dysfunction,” Dr. Shufelt said. “Our results demonstrated significantly higher circulating levels of serum proinflammatory cytokines in the women with hypothalamic amenorrhea compared to eumenorrheic controls.”
Dr. Shufelt’s team then tested whether giving estradiol to the women with hypothalamic amenorrhea for 12 weeks would improve their vascular health, but they saw no significant differences between the women who received estrogen and those who received placebo.
“Endothelial function is partly mediated by estrogen, and it was expected that giving back estrogen would ‘fix’ the endothelium, but that is not what happened,” Nanette Santoro, MD, professor and chair of obstetrics and gynecology at the University of Colorado at Denver, Aurora, said in interview. “The mechanisms that maintain vascular function in women are not limited to hormones,” said Dr. Santoro, who was not involved in Dr. Shufelt’s study but attended her lecture. “We need to think beyond the simple model of estrogen-good, no-estrogen-bad.”
Dr. Santoro noted how easy it is to overlook the women who may have cardiovascular risk because of hypothalamic amenorrhea.
“Because many women with functional hypothalamic amenorrhea are super athletic and do not have the typical features of people with cardiometabolic disease – such as glucose intolerance, obesity, abnormal cholesterol or triglycerides, or high blood pressure – clinicians tend to think of them as healthy and to think that simply giving back hormones will fix the problems with bone density and vascular function, but that is not enough,” Dr. Santoro said. “The cognitive-behavioral therapy model for treatment of women with functional hypothalamic amenorrhea addresses the stress-related factors that drive the disorder, and this needs to be considered the standard of care for treatment.”
Stephanie S. Faubion, MD, professor of medicine and director of Mayo Clinic’s Center for Women’s Health in Jacksonville, Fla., who was not involved in Dr. Shufelt’s presentation, also emphasized the importance of recognizing functional hypothalamic amenorrhea.
“This is an underrecognized entity to begin with, and the fact that these women appear to be at increased risk for vascular dysfunction and potentially increased risk for cardiovascular disease down the road makes it even more important for clinicians to identify them and provide interventions early on,” Dr. Faubion said in an interview. “These women need to be identified and the etiology of the amenorrhea addressed, whether it relates to overexercising, being underweight, or experiencing significant stressors that have led to the loss of menstrual cycles.”
Dr. Shufelt’s research was funded by the National Institutes of Health. She had no disclosures. Dr. Santoro is a member of the scientific advisory board for Astellas, Menogenix, Amazon Ember, and Que Oncology, and she consults for Ansh Labs. Dr. Faubion had no disclosures.
The relationship between estrogen levels and heart health makes it particularly important for clinicians to be aware of those patients who might be at risk for cardiovascular disease despite not having other traditional risk factors, according to a presentation Oct. 12 at the North American Menopause Society annual meeting in Atlanta.
”Endogenous estrogens are protective for cardiovascular disease in premenopausal women,” Chrisandra L. Shufelt, MD, chair of the division of general internal medicine and associate director of the Women’s Health Research Center at Mayo Clinic in Jacksonville, Fla., told attendees. Yet, “a substantial population of young women are dying prematurely from cardiovascular disease,” with rates of cardiovascular death increasing in women aged 35-44 even as rates have decreased in postmenopausal women and in men. One potential reason may be premature estrogen loss.
Dr. Shufelt reminded attendees of four major causes of premature estrogen loss: Natural premature menopause, surgical menopause, chemotherapy-induced menopause, and premature ovarian insufficiency. But she would go on to discuss a less widely recognized condition, functional hypothalamic amenorrhea, that also may be contributing to increased cardiovascular risk.
First, Dr. Shufelt reviewed the evidence supporting the relationship between estrogen and cardiovascular health, starting with the Framingham study’s findings that cardiovascular disease is approximately two to four times more common in postmenopausal women than in premenopausal women, depending on the age range.
“Menopause at an early age, particularly under the age of 40, matters,” Dr. Shufelt said. “So we should be discussing this with our patients.”
Surgical menopause makes a difference to cardiovascular health as well, she said. In women under age 35, for example, the risk of a nonfatal heart attack in those with a bilateral oophorectomy was 7.7 times greater than in women who retained both ovaries and their uterus, and 1.5 times greater in women who had a hysterectomy without bilateral oophorectomy.
In a 2019 study, surgical premature menopause was associated with an 87% increased risk of heart disease even after researchers accounted for age, cardiovascular risk factors, and some forms of hormone therapy. The increased risk from natural premature menopause, on the other hand, was lower – a 36% increased risk of heart disease – compared with those producing endogenous hormones. Although randomized controlled trials are unavailable and unlikely to be done, the Nurses’ Health Study and the Danish Nurses Cohort Study, both observational studies, found that heart disease risk was diminished in those taking hormone therapy after surgical premature menopause.
Recommendations for premature or early menopause, from a wide range of different medical societies including NAMS, are that women without contraindications be given estrogen-based hormone therapy until the average age of natural menopause. Though not included in the same guidance, research has also shown that estrogen after oophorectomy does not increase the risk of breast cancer in women with a BRCA1 mutation, Dr. Shufelt said. Hormone therapy for premature or early menopause should adequately replace the levels women have lost and that means younger menopausal women often need higher doses than what older women receive, such as 2 mg/day of oral estradiol rather than the standard doses of 0.5 or 1 mg/day.
Functional hypothalamic amenorrhea and cardiovascular risk
Dr. Shufelt then discussed functional hypothalamic amenorrhea (hypogonadotropic hypogonadism), a common type of secondary amenorrhea that affects at least 1.4 million U.S. women. Diagnosis includes lack of a period for at least 3 months in someone who previously menstruated plus lab values below 50 pg/mL for estradiol, below 10 mIU/L for follicle stimulating hormone, and below 10 mIU/L for luteinizing hormone. Causes of this reversible form of infertility can include stress, overexercising, undereating, or some combination of these, plus an underlying genetic predisposition.
“After ruling out polycystic ovary syndrome, prolactinoma, and thyroid dysfunction, clinicians need to consider the diagnosis of hypothalamic amenorrhea,” Dr. Shufelt said. This condition goes beyond low estrogen levels: Women have elevated cortisol, low thyroid levels, low leptin levels, and increased ghrelin.
”This is not going away,” Dr. Shufelt said, sharing data on stress levels among U.S. adults, particularly Gen Z and millennial adults, noting that the ongoing “national mental health crisis” may be contributing to functional hypothalamic amenorrhea.
A 2020 substudy from the Nurses’ Health Study II found an increased risk of premature death in those who didn’t have a period or always had irregular periods starting as early as 14-17 years old. The increased risk of premature death rose with age in those with irregular or absent cycles – a 37% higher risk in 18- to 22-year-olds and a 39% increased risk in 29- to 46-year-olds.
But clinicians aren’t adequately identifying the “phenotype of the hypothalamic women,” Dr. Shufelt said, despite research showing overlap between hypothalamic amenorrhea and a higher risk of cardiovascular disease. Hypothalamic amenorrhea is so understudied that the last original research on the topic was in 2008, Dr. Shufelt said in an interview. ”No research except mine has been done to evaluate heart health in these young women,” she said.
Dr. Shufelt described a study she led involving 30 women with functional hypothalamic amenorrhea, 29 women with normal menstrual cycles, and 30 women who were recently menopausal and not on hormone therapy. The women with hypothalamic amenorrhea had average stress levels but their depression scores were higher than those of the other two groups.
The results showed that women with hypothalamic amenorrhea had lower estradiol and leptin levels and higher testosterone levels compared with the control group, and they had higher cortisol levels than those of both groups. Despite having similar body mass indexes as the control and menopausal groups, women with hypothalamic amenorrhea had lower blood pressure than that of the other two groups, yet they had higher cholesterol levels than those of the control group. EndoPAT© (Itamar Medical) testing showed that they had poor vascular function.
“In fact, one-third of the women [with hypothalamic amenorrhea] entered the trial with a diagnosis of what would be considered endothelial dysfunction,” Dr. Shufelt said. “Our results demonstrated significantly higher circulating levels of serum proinflammatory cytokines in the women with hypothalamic amenorrhea compared to eumenorrheic controls.”
Dr. Shufelt’s team then tested whether giving estradiol to the women with hypothalamic amenorrhea for 12 weeks would improve their vascular health, but they saw no significant differences between the women who received estrogen and those who received placebo.
“Endothelial function is partly mediated by estrogen, and it was expected that giving back estrogen would ‘fix’ the endothelium, but that is not what happened,” Nanette Santoro, MD, professor and chair of obstetrics and gynecology at the University of Colorado at Denver, Aurora, said in interview. “The mechanisms that maintain vascular function in women are not limited to hormones,” said Dr. Santoro, who was not involved in Dr. Shufelt’s study but attended her lecture. “We need to think beyond the simple model of estrogen-good, no-estrogen-bad.”
Dr. Santoro noted how easy it is to overlook the women who may have cardiovascular risk because of hypothalamic amenorrhea.
“Because many women with functional hypothalamic amenorrhea are super athletic and do not have the typical features of people with cardiometabolic disease – such as glucose intolerance, obesity, abnormal cholesterol or triglycerides, or high blood pressure – clinicians tend to think of them as healthy and to think that simply giving back hormones will fix the problems with bone density and vascular function, but that is not enough,” Dr. Santoro said. “The cognitive-behavioral therapy model for treatment of women with functional hypothalamic amenorrhea addresses the stress-related factors that drive the disorder, and this needs to be considered the standard of care for treatment.”
Stephanie S. Faubion, MD, professor of medicine and director of Mayo Clinic’s Center for Women’s Health in Jacksonville, Fla., who was not involved in Dr. Shufelt’s presentation, also emphasized the importance of recognizing functional hypothalamic amenorrhea.
“This is an underrecognized entity to begin with, and the fact that these women appear to be at increased risk for vascular dysfunction and potentially increased risk for cardiovascular disease down the road makes it even more important for clinicians to identify them and provide interventions early on,” Dr. Faubion said in an interview. “These women need to be identified and the etiology of the amenorrhea addressed, whether it relates to overexercising, being underweight, or experiencing significant stressors that have led to the loss of menstrual cycles.”
Dr. Shufelt’s research was funded by the National Institutes of Health. She had no disclosures. Dr. Santoro is a member of the scientific advisory board for Astellas, Menogenix, Amazon Ember, and Que Oncology, and she consults for Ansh Labs. Dr. Faubion had no disclosures.
The relationship between estrogen levels and heart health makes it particularly important for clinicians to be aware of those patients who might be at risk for cardiovascular disease despite not having other traditional risk factors, according to a presentation Oct. 12 at the North American Menopause Society annual meeting in Atlanta.
”Endogenous estrogens are protective for cardiovascular disease in premenopausal women,” Chrisandra L. Shufelt, MD, chair of the division of general internal medicine and associate director of the Women’s Health Research Center at Mayo Clinic in Jacksonville, Fla., told attendees. Yet, “a substantial population of young women are dying prematurely from cardiovascular disease,” with rates of cardiovascular death increasing in women aged 35-44 even as rates have decreased in postmenopausal women and in men. One potential reason may be premature estrogen loss.
Dr. Shufelt reminded attendees of four major causes of premature estrogen loss: Natural premature menopause, surgical menopause, chemotherapy-induced menopause, and premature ovarian insufficiency. But she would go on to discuss a less widely recognized condition, functional hypothalamic amenorrhea, that also may be contributing to increased cardiovascular risk.
First, Dr. Shufelt reviewed the evidence supporting the relationship between estrogen and cardiovascular health, starting with the Framingham study’s findings that cardiovascular disease is approximately two to four times more common in postmenopausal women than in premenopausal women, depending on the age range.
“Menopause at an early age, particularly under the age of 40, matters,” Dr. Shufelt said. “So we should be discussing this with our patients.”
Surgical menopause makes a difference to cardiovascular health as well, she said. In women under age 35, for example, the risk of a nonfatal heart attack in those with a bilateral oophorectomy was 7.7 times greater than in women who retained both ovaries and their uterus, and 1.5 times greater in women who had a hysterectomy without bilateral oophorectomy.
In a 2019 study, surgical premature menopause was associated with an 87% increased risk of heart disease even after researchers accounted for age, cardiovascular risk factors, and some forms of hormone therapy. The increased risk from natural premature menopause, on the other hand, was lower – a 36% increased risk of heart disease – compared with those producing endogenous hormones. Although randomized controlled trials are unavailable and unlikely to be done, the Nurses’ Health Study and the Danish Nurses Cohort Study, both observational studies, found that heart disease risk was diminished in those taking hormone therapy after surgical premature menopause.
Recommendations for premature or early menopause, from a wide range of different medical societies including NAMS, are that women without contraindications be given estrogen-based hormone therapy until the average age of natural menopause. Though not included in the same guidance, research has also shown that estrogen after oophorectomy does not increase the risk of breast cancer in women with a BRCA1 mutation, Dr. Shufelt said. Hormone therapy for premature or early menopause should adequately replace the levels women have lost and that means younger menopausal women often need higher doses than what older women receive, such as 2 mg/day of oral estradiol rather than the standard doses of 0.5 or 1 mg/day.
Functional hypothalamic amenorrhea and cardiovascular risk
Dr. Shufelt then discussed functional hypothalamic amenorrhea (hypogonadotropic hypogonadism), a common type of secondary amenorrhea that affects at least 1.4 million U.S. women. Diagnosis includes lack of a period for at least 3 months in someone who previously menstruated plus lab values below 50 pg/mL for estradiol, below 10 mIU/L for follicle stimulating hormone, and below 10 mIU/L for luteinizing hormone. Causes of this reversible form of infertility can include stress, overexercising, undereating, or some combination of these, plus an underlying genetic predisposition.
“After ruling out polycystic ovary syndrome, prolactinoma, and thyroid dysfunction, clinicians need to consider the diagnosis of hypothalamic amenorrhea,” Dr. Shufelt said. This condition goes beyond low estrogen levels: Women have elevated cortisol, low thyroid levels, low leptin levels, and increased ghrelin.
”This is not going away,” Dr. Shufelt said, sharing data on stress levels among U.S. adults, particularly Gen Z and millennial adults, noting that the ongoing “national mental health crisis” may be contributing to functional hypothalamic amenorrhea.
A 2020 substudy from the Nurses’ Health Study II found an increased risk of premature death in those who didn’t have a period or always had irregular periods starting as early as 14-17 years old. The increased risk of premature death rose with age in those with irregular or absent cycles – a 37% higher risk in 18- to 22-year-olds and a 39% increased risk in 29- to 46-year-olds.
But clinicians aren’t adequately identifying the “phenotype of the hypothalamic women,” Dr. Shufelt said, despite research showing overlap between hypothalamic amenorrhea and a higher risk of cardiovascular disease. Hypothalamic amenorrhea is so understudied that the last original research on the topic was in 2008, Dr. Shufelt said in an interview. ”No research except mine has been done to evaluate heart health in these young women,” she said.
Dr. Shufelt described a study she led involving 30 women with functional hypothalamic amenorrhea, 29 women with normal menstrual cycles, and 30 women who were recently menopausal and not on hormone therapy. The women with hypothalamic amenorrhea had average stress levels but their depression scores were higher than those of the other two groups.
The results showed that women with hypothalamic amenorrhea had lower estradiol and leptin levels and higher testosterone levels compared with the control group, and they had higher cortisol levels than those of both groups. Despite having similar body mass indexes as the control and menopausal groups, women with hypothalamic amenorrhea had lower blood pressure than that of the other two groups, yet they had higher cholesterol levels than those of the control group. EndoPAT© (Itamar Medical) testing showed that they had poor vascular function.
“In fact, one-third of the women [with hypothalamic amenorrhea] entered the trial with a diagnosis of what would be considered endothelial dysfunction,” Dr. Shufelt said. “Our results demonstrated significantly higher circulating levels of serum proinflammatory cytokines in the women with hypothalamic amenorrhea compared to eumenorrheic controls.”
Dr. Shufelt’s team then tested whether giving estradiol to the women with hypothalamic amenorrhea for 12 weeks would improve their vascular health, but they saw no significant differences between the women who received estrogen and those who received placebo.
“Endothelial function is partly mediated by estrogen, and it was expected that giving back estrogen would ‘fix’ the endothelium, but that is not what happened,” Nanette Santoro, MD, professor and chair of obstetrics and gynecology at the University of Colorado at Denver, Aurora, said in interview. “The mechanisms that maintain vascular function in women are not limited to hormones,” said Dr. Santoro, who was not involved in Dr. Shufelt’s study but attended her lecture. “We need to think beyond the simple model of estrogen-good, no-estrogen-bad.”
Dr. Santoro noted how easy it is to overlook the women who may have cardiovascular risk because of hypothalamic amenorrhea.
“Because many women with functional hypothalamic amenorrhea are super athletic and do not have the typical features of people with cardiometabolic disease – such as glucose intolerance, obesity, abnormal cholesterol or triglycerides, or high blood pressure – clinicians tend to think of them as healthy and to think that simply giving back hormones will fix the problems with bone density and vascular function, but that is not enough,” Dr. Santoro said. “The cognitive-behavioral therapy model for treatment of women with functional hypothalamic amenorrhea addresses the stress-related factors that drive the disorder, and this needs to be considered the standard of care for treatment.”
Stephanie S. Faubion, MD, professor of medicine and director of Mayo Clinic’s Center for Women’s Health in Jacksonville, Fla., who was not involved in Dr. Shufelt’s presentation, also emphasized the importance of recognizing functional hypothalamic amenorrhea.
“This is an underrecognized entity to begin with, and the fact that these women appear to be at increased risk for vascular dysfunction and potentially increased risk for cardiovascular disease down the road makes it even more important for clinicians to identify them and provide interventions early on,” Dr. Faubion said in an interview. “These women need to be identified and the etiology of the amenorrhea addressed, whether it relates to overexercising, being underweight, or experiencing significant stressors that have led to the loss of menstrual cycles.”
Dr. Shufelt’s research was funded by the National Institutes of Health. She had no disclosures. Dr. Santoro is a member of the scientific advisory board for Astellas, Menogenix, Amazon Ember, and Que Oncology, and she consults for Ansh Labs. Dr. Faubion had no disclosures.
FROM NAMS 2022
It’s about location: PCOS symptoms differ depending where you live
Geographic location within the United States appears to have an impact on the specific symptoms of polycystic ovary syndrome (PCOS) that any one particular woman will develop, according to a new prospective cohort study.
Women in California were more likely to exhibit high levels of testosterone (hyperandrogenism), while women in Alabama with PCOS had more metabolic dysfunction and hirsutism.
And although the women in Alabama were younger and had a higher body mass index (BMI), even after adjusting for these factors, the clinical differences were still present between the geographic locations, the authors said.
“This study suggests there are regional differences in hormonal and metabolic parameters in women with PCOS in California and Alabama, highlighting the impact of differing genetic and environmental modulators on PCOS development,” Katherine VanHise, MD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues wrote in their article, published online in the Journal of Clinical Endocrinology and Metabolism.
Genetic and environmental factors play a role
Prior research has looked at variations in symptoms of PCOS across countries and identified differences in hirsutism and its prevalence, which is greater in Middle Eastern, Mediterranean, and Indian women, noted senior author Margareta D. Pisarska, MD.
And women of some other backgrounds “are at increased risk of developing metabolic syndrome and insulin resistance, including South Asian, African, and Hispanic women, so they are at a greater risk trajectory of developing manifestations later on in life that can ultimately lead to adverse outcomes in overall health,” Dr. Pisarska, director of the division of reproductive endocrinology and infertility in obstetrics and gynecology at Cedars-Sinai, told this news organization.
“We do see regional differences in the diagnosis of PCOS [in the United States] as well as the manifestations of PCOS including high andrenoemia, hirsutism, and metabolic parameters ... and we need to better understand it because, at least in the entire population, weight was not the entire factor contributing to these differences,” she explained.
“So there are definitely environmental factors and possibly genetic factors that we need to take into consideration as we try to study these women and try to help them decrease their risk of metabolic syndrome later in life,” she noted.
Differences not attributable to race either
PCOS is a common endocrine disorder affecting women and female adolescents worldwide. Diagnosis usually requires at least two of the following to be present: ovulatory dysfunction, hyperandrogenism, and/or polycystic ovarian morphology.
Because of the prior work that had identified differences in symptoms among women with PCOS in different countries, the investigators set out to determine if women of the same race would have distinct hormonal and metabolic traits of PCOS in two geographical locations in the United States, suggesting geo-epidemiologic contributors of the disease
They evaluated 889 women at the University of Alabama at Birmingham and 721 at Cedars-Sinai Medical Center. Participants in Birmingham were a mean age of 28 years, had a mean BMI of 33.1 kg/m2, a mean waist-to-hip ratio of 0.8, and a mean hirsute rate of 84.6%. Participants in California were a mean age of 29.5 years, had an average BMI of 30.1 kg/m2, a mean waist-to-hip ratio of 0.9, and a mean hirsute rate of 72.8%.
The study team gathered data on menstrual cycle history, metabolic and hormonal parameters, and demographic data for each participant. They assessed hirsutism based on modified Ferriman-Gallwey scores of four or more. Patients were classified as having hyperandrogenemia if they had elevated androgen values greater than the 95th percentile of all values or androgen values that exceeded laboratory reference ranges.
The findings showed that Alabama women with PCOS had elevated homeostatic model assessment for insulin resistance scores (adjusted beta coefficient, 3.6; P < .001) and were more likely to be hirsute (adjusted odds ratio, 1.8; P < .001) after adjustment for BMI and age than those in California.
In contrast, women with PCOS in California were more likely to have elevated free testosterone and total testosterone values than women in Alabama (both P < .001). These findings persisted after adjusting for age and BMI.
When stratified by White race, these findings were similar. Notably, BMI and waist-to-hip ratio did not vary between regions in Black women with PCOS, although variations in metabolic dysfunction and androgen profiles persisted.
“This study supports regional differences in hormonal and metabolic parameters in women with PCOS in the United States, highlighting the impact of the environment on PCOS phenotype. Individuals of the same race in different geographical locations of the United States may have differing genetic predispositions for developing diseases such as PCOS,” the researchers said.
“Ongoing research is needed to identify modifiable environmental risk factors for PCOS that may be race and ethnic specific to bring precision medicine to the management of PCOS,” they conclude.
This work was supported in part by grants from the National Institutes of Health and an endowment of the Helping Hand of Los Angeles. Dr. VanHise reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Geographic location within the United States appears to have an impact on the specific symptoms of polycystic ovary syndrome (PCOS) that any one particular woman will develop, according to a new prospective cohort study.
Women in California were more likely to exhibit high levels of testosterone (hyperandrogenism), while women in Alabama with PCOS had more metabolic dysfunction and hirsutism.
And although the women in Alabama were younger and had a higher body mass index (BMI), even after adjusting for these factors, the clinical differences were still present between the geographic locations, the authors said.
“This study suggests there are regional differences in hormonal and metabolic parameters in women with PCOS in California and Alabama, highlighting the impact of differing genetic and environmental modulators on PCOS development,” Katherine VanHise, MD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues wrote in their article, published online in the Journal of Clinical Endocrinology and Metabolism.
Genetic and environmental factors play a role
Prior research has looked at variations in symptoms of PCOS across countries and identified differences in hirsutism and its prevalence, which is greater in Middle Eastern, Mediterranean, and Indian women, noted senior author Margareta D. Pisarska, MD.
And women of some other backgrounds “are at increased risk of developing metabolic syndrome and insulin resistance, including South Asian, African, and Hispanic women, so they are at a greater risk trajectory of developing manifestations later on in life that can ultimately lead to adverse outcomes in overall health,” Dr. Pisarska, director of the division of reproductive endocrinology and infertility in obstetrics and gynecology at Cedars-Sinai, told this news organization.
“We do see regional differences in the diagnosis of PCOS [in the United States] as well as the manifestations of PCOS including high andrenoemia, hirsutism, and metabolic parameters ... and we need to better understand it because, at least in the entire population, weight was not the entire factor contributing to these differences,” she explained.
“So there are definitely environmental factors and possibly genetic factors that we need to take into consideration as we try to study these women and try to help them decrease their risk of metabolic syndrome later in life,” she noted.
Differences not attributable to race either
PCOS is a common endocrine disorder affecting women and female adolescents worldwide. Diagnosis usually requires at least two of the following to be present: ovulatory dysfunction, hyperandrogenism, and/or polycystic ovarian morphology.
Because of the prior work that had identified differences in symptoms among women with PCOS in different countries, the investigators set out to determine if women of the same race would have distinct hormonal and metabolic traits of PCOS in two geographical locations in the United States, suggesting geo-epidemiologic contributors of the disease
They evaluated 889 women at the University of Alabama at Birmingham and 721 at Cedars-Sinai Medical Center. Participants in Birmingham were a mean age of 28 years, had a mean BMI of 33.1 kg/m2, a mean waist-to-hip ratio of 0.8, and a mean hirsute rate of 84.6%. Participants in California were a mean age of 29.5 years, had an average BMI of 30.1 kg/m2, a mean waist-to-hip ratio of 0.9, and a mean hirsute rate of 72.8%.
The study team gathered data on menstrual cycle history, metabolic and hormonal parameters, and demographic data for each participant. They assessed hirsutism based on modified Ferriman-Gallwey scores of four or more. Patients were classified as having hyperandrogenemia if they had elevated androgen values greater than the 95th percentile of all values or androgen values that exceeded laboratory reference ranges.
The findings showed that Alabama women with PCOS had elevated homeostatic model assessment for insulin resistance scores (adjusted beta coefficient, 3.6; P < .001) and were more likely to be hirsute (adjusted odds ratio, 1.8; P < .001) after adjustment for BMI and age than those in California.
In contrast, women with PCOS in California were more likely to have elevated free testosterone and total testosterone values than women in Alabama (both P < .001). These findings persisted after adjusting for age and BMI.
When stratified by White race, these findings were similar. Notably, BMI and waist-to-hip ratio did not vary between regions in Black women with PCOS, although variations in metabolic dysfunction and androgen profiles persisted.
“This study supports regional differences in hormonal and metabolic parameters in women with PCOS in the United States, highlighting the impact of the environment on PCOS phenotype. Individuals of the same race in different geographical locations of the United States may have differing genetic predispositions for developing diseases such as PCOS,” the researchers said.
“Ongoing research is needed to identify modifiable environmental risk factors for PCOS that may be race and ethnic specific to bring precision medicine to the management of PCOS,” they conclude.
This work was supported in part by grants from the National Institutes of Health and an endowment of the Helping Hand of Los Angeles. Dr. VanHise reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Geographic location within the United States appears to have an impact on the specific symptoms of polycystic ovary syndrome (PCOS) that any one particular woman will develop, according to a new prospective cohort study.
Women in California were more likely to exhibit high levels of testosterone (hyperandrogenism), while women in Alabama with PCOS had more metabolic dysfunction and hirsutism.
And although the women in Alabama were younger and had a higher body mass index (BMI), even after adjusting for these factors, the clinical differences were still present between the geographic locations, the authors said.
“This study suggests there are regional differences in hormonal and metabolic parameters in women with PCOS in California and Alabama, highlighting the impact of differing genetic and environmental modulators on PCOS development,” Katherine VanHise, MD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues wrote in their article, published online in the Journal of Clinical Endocrinology and Metabolism.
Genetic and environmental factors play a role
Prior research has looked at variations in symptoms of PCOS across countries and identified differences in hirsutism and its prevalence, which is greater in Middle Eastern, Mediterranean, and Indian women, noted senior author Margareta D. Pisarska, MD.
And women of some other backgrounds “are at increased risk of developing metabolic syndrome and insulin resistance, including South Asian, African, and Hispanic women, so they are at a greater risk trajectory of developing manifestations later on in life that can ultimately lead to adverse outcomes in overall health,” Dr. Pisarska, director of the division of reproductive endocrinology and infertility in obstetrics and gynecology at Cedars-Sinai, told this news organization.
“We do see regional differences in the diagnosis of PCOS [in the United States] as well as the manifestations of PCOS including high andrenoemia, hirsutism, and metabolic parameters ... and we need to better understand it because, at least in the entire population, weight was not the entire factor contributing to these differences,” she explained.
“So there are definitely environmental factors and possibly genetic factors that we need to take into consideration as we try to study these women and try to help them decrease their risk of metabolic syndrome later in life,” she noted.
Differences not attributable to race either
PCOS is a common endocrine disorder affecting women and female adolescents worldwide. Diagnosis usually requires at least two of the following to be present: ovulatory dysfunction, hyperandrogenism, and/or polycystic ovarian morphology.
Because of the prior work that had identified differences in symptoms among women with PCOS in different countries, the investigators set out to determine if women of the same race would have distinct hormonal and metabolic traits of PCOS in two geographical locations in the United States, suggesting geo-epidemiologic contributors of the disease
They evaluated 889 women at the University of Alabama at Birmingham and 721 at Cedars-Sinai Medical Center. Participants in Birmingham were a mean age of 28 years, had a mean BMI of 33.1 kg/m2, a mean waist-to-hip ratio of 0.8, and a mean hirsute rate of 84.6%. Participants in California were a mean age of 29.5 years, had an average BMI of 30.1 kg/m2, a mean waist-to-hip ratio of 0.9, and a mean hirsute rate of 72.8%.
The study team gathered data on menstrual cycle history, metabolic and hormonal parameters, and demographic data for each participant. They assessed hirsutism based on modified Ferriman-Gallwey scores of four or more. Patients were classified as having hyperandrogenemia if they had elevated androgen values greater than the 95th percentile of all values or androgen values that exceeded laboratory reference ranges.
The findings showed that Alabama women with PCOS had elevated homeostatic model assessment for insulin resistance scores (adjusted beta coefficient, 3.6; P < .001) and were more likely to be hirsute (adjusted odds ratio, 1.8; P < .001) after adjustment for BMI and age than those in California.
In contrast, women with PCOS in California were more likely to have elevated free testosterone and total testosterone values than women in Alabama (both P < .001). These findings persisted after adjusting for age and BMI.
When stratified by White race, these findings were similar. Notably, BMI and waist-to-hip ratio did not vary between regions in Black women with PCOS, although variations in metabolic dysfunction and androgen profiles persisted.
“This study supports regional differences in hormonal and metabolic parameters in women with PCOS in the United States, highlighting the impact of the environment on PCOS phenotype. Individuals of the same race in different geographical locations of the United States may have differing genetic predispositions for developing diseases such as PCOS,” the researchers said.
“Ongoing research is needed to identify modifiable environmental risk factors for PCOS that may be race and ethnic specific to bring precision medicine to the management of PCOS,” they conclude.
This work was supported in part by grants from the National Institutes of Health and an endowment of the Helping Hand of Los Angeles. Dr. VanHise reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY AND METABOLISM
Children born from frozen embryos may have increased cancer risk
, a large registry study suggests.
The results, however, “should be interpreted cautiously,” the authors noted, given the low number of cancer cases reported among children born using FET.
Still, the findings do “raise concerns considering the increasing use of FET, in particular freeze-all strategies without clear medical indications,” the authors concluded.
The study was published online in PLOS Medicine.
The number of children born after FET has increased globally and even exceeds the number of those born after fresh embryo transfer in many countries. In the United States, for instance, the FET rate has doubled since 2015; FETs constituted almost 80% of all embryo transfers using assisted reproductive technology (ART) without a donor in 2019.
Despite the benefits associated with FET, which include improved embryo survival and higher live birth rates, some previous research has hinted at a higher risk of childhood cancer in this population.
In the current study, researchers from the University of Gothenburg, Sweden, wanted to better understand the risk of childhood cancer following FET. The investigators analyzed data from 171,774 children born via ART, including 22,630 born after FET, as well as roughly 7.7 million children born after spontaneous conception in Denmark, Finland, Norway, and Sweden.
After a mean follow-up of about 10 years, the incidence rate of cancer diagnosed before age 18 years was 16.7 per 100,000 person-years for children born after spontaneous conception (16,184 cases) and 19.3 per 100,000 person-years for children born after ART (329 cases).
The researchers found no increased risk of cancer before age 18 years in the group of children conceived via ART compared with those conceived spontaneously.
However, children born after FET had a significantly higher risk of cancer compared with children born after fresh embryo transfer (adjusted hazard ratio [aHR], 1.59) and spontaneous conception (aHR, 1.65). Specifically with regard to ART, the incidence rate for those born after FET was 30.1 per 100,000 person-years – 48 total cases – compared with 18.8 per 1000,000 person-years after fresh embryo transfer.
Adjustment for macrosomia, birth weight, or major birth defects influenced the association only marginally.
For specific cancer types, children born after FET had more than a twofold higher risk for leukemia in comparison with those born after fresh embryo transfer (aHR, 2.25) and spontaneous conception (aHR, 2.22).
Still, the authors said these results should be interpreted “cautiously,” given the small number of children diagnosed with cancer after FET. The researchers also acknowledged that they do not know why children born after FET would face a higher risk of cancer.
These findings, however, do align with those from a 2019 Dutch population-based study. In the Dutch study, which included more than 24,000 ART-conceived children and more than 23,000 naturally conceived children, the risk of cancer after ART was not higher overall, but it was greater when only those conceived after FET were considered (aHR 1.80); this increased risk, however, was not statistically significant.
“Since the use of FET is substantially increasing, it is important to tease out whether the increased cancer risk is a true risk increase due to the ART procedures using FET, or due to chance or confounding by other factors,” authors of the 2019 Dutch study, Mandy Spaan, PhD, and Flora E. van Leeuwen, PhD, said in an interview.
“But, as childhood cancer is (fortunately) a rare disease, it is very difficult to study this research question among ART children due to limited numbers,” said Dr. Spaan and Dr. van Leeuwen, who are with the Netherlands Cancer Institute.
Given this, the two experts call for additional large population-based cohort studies to investigate the risk of cancer after ART, especially FET, and for a subsequent analysis that pools these data. They hope this strategy “will lead to reliable estimates” and provide information on the risks of FET in comparison with approaches that involve fresh embryos.
The current study had no commercial funding. The study authors as well as Dr. Spaan and Dr. van Leeuwen have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a large registry study suggests.
The results, however, “should be interpreted cautiously,” the authors noted, given the low number of cancer cases reported among children born using FET.
Still, the findings do “raise concerns considering the increasing use of FET, in particular freeze-all strategies without clear medical indications,” the authors concluded.
The study was published online in PLOS Medicine.
The number of children born after FET has increased globally and even exceeds the number of those born after fresh embryo transfer in many countries. In the United States, for instance, the FET rate has doubled since 2015; FETs constituted almost 80% of all embryo transfers using assisted reproductive technology (ART) without a donor in 2019.
Despite the benefits associated with FET, which include improved embryo survival and higher live birth rates, some previous research has hinted at a higher risk of childhood cancer in this population.
In the current study, researchers from the University of Gothenburg, Sweden, wanted to better understand the risk of childhood cancer following FET. The investigators analyzed data from 171,774 children born via ART, including 22,630 born after FET, as well as roughly 7.7 million children born after spontaneous conception in Denmark, Finland, Norway, and Sweden.
After a mean follow-up of about 10 years, the incidence rate of cancer diagnosed before age 18 years was 16.7 per 100,000 person-years for children born after spontaneous conception (16,184 cases) and 19.3 per 100,000 person-years for children born after ART (329 cases).
The researchers found no increased risk of cancer before age 18 years in the group of children conceived via ART compared with those conceived spontaneously.
However, children born after FET had a significantly higher risk of cancer compared with children born after fresh embryo transfer (adjusted hazard ratio [aHR], 1.59) and spontaneous conception (aHR, 1.65). Specifically with regard to ART, the incidence rate for those born after FET was 30.1 per 100,000 person-years – 48 total cases – compared with 18.8 per 1000,000 person-years after fresh embryo transfer.
Adjustment for macrosomia, birth weight, or major birth defects influenced the association only marginally.
For specific cancer types, children born after FET had more than a twofold higher risk for leukemia in comparison with those born after fresh embryo transfer (aHR, 2.25) and spontaneous conception (aHR, 2.22).
Still, the authors said these results should be interpreted “cautiously,” given the small number of children diagnosed with cancer after FET. The researchers also acknowledged that they do not know why children born after FET would face a higher risk of cancer.
These findings, however, do align with those from a 2019 Dutch population-based study. In the Dutch study, which included more than 24,000 ART-conceived children and more than 23,000 naturally conceived children, the risk of cancer after ART was not higher overall, but it was greater when only those conceived after FET were considered (aHR 1.80); this increased risk, however, was not statistically significant.
“Since the use of FET is substantially increasing, it is important to tease out whether the increased cancer risk is a true risk increase due to the ART procedures using FET, or due to chance or confounding by other factors,” authors of the 2019 Dutch study, Mandy Spaan, PhD, and Flora E. van Leeuwen, PhD, said in an interview.
“But, as childhood cancer is (fortunately) a rare disease, it is very difficult to study this research question among ART children due to limited numbers,” said Dr. Spaan and Dr. van Leeuwen, who are with the Netherlands Cancer Institute.
Given this, the two experts call for additional large population-based cohort studies to investigate the risk of cancer after ART, especially FET, and for a subsequent analysis that pools these data. They hope this strategy “will lead to reliable estimates” and provide information on the risks of FET in comparison with approaches that involve fresh embryos.
The current study had no commercial funding. The study authors as well as Dr. Spaan and Dr. van Leeuwen have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a large registry study suggests.
The results, however, “should be interpreted cautiously,” the authors noted, given the low number of cancer cases reported among children born using FET.
Still, the findings do “raise concerns considering the increasing use of FET, in particular freeze-all strategies without clear medical indications,” the authors concluded.
The study was published online in PLOS Medicine.
The number of children born after FET has increased globally and even exceeds the number of those born after fresh embryo transfer in many countries. In the United States, for instance, the FET rate has doubled since 2015; FETs constituted almost 80% of all embryo transfers using assisted reproductive technology (ART) without a donor in 2019.
Despite the benefits associated with FET, which include improved embryo survival and higher live birth rates, some previous research has hinted at a higher risk of childhood cancer in this population.
In the current study, researchers from the University of Gothenburg, Sweden, wanted to better understand the risk of childhood cancer following FET. The investigators analyzed data from 171,774 children born via ART, including 22,630 born after FET, as well as roughly 7.7 million children born after spontaneous conception in Denmark, Finland, Norway, and Sweden.
After a mean follow-up of about 10 years, the incidence rate of cancer diagnosed before age 18 years was 16.7 per 100,000 person-years for children born after spontaneous conception (16,184 cases) and 19.3 per 100,000 person-years for children born after ART (329 cases).
The researchers found no increased risk of cancer before age 18 years in the group of children conceived via ART compared with those conceived spontaneously.
However, children born after FET had a significantly higher risk of cancer compared with children born after fresh embryo transfer (adjusted hazard ratio [aHR], 1.59) and spontaneous conception (aHR, 1.65). Specifically with regard to ART, the incidence rate for those born after FET was 30.1 per 100,000 person-years – 48 total cases – compared with 18.8 per 1000,000 person-years after fresh embryo transfer.
Adjustment for macrosomia, birth weight, or major birth defects influenced the association only marginally.
For specific cancer types, children born after FET had more than a twofold higher risk for leukemia in comparison with those born after fresh embryo transfer (aHR, 2.25) and spontaneous conception (aHR, 2.22).
Still, the authors said these results should be interpreted “cautiously,” given the small number of children diagnosed with cancer after FET. The researchers also acknowledged that they do not know why children born after FET would face a higher risk of cancer.
These findings, however, do align with those from a 2019 Dutch population-based study. In the Dutch study, which included more than 24,000 ART-conceived children and more than 23,000 naturally conceived children, the risk of cancer after ART was not higher overall, but it was greater when only those conceived after FET were considered (aHR 1.80); this increased risk, however, was not statistically significant.
“Since the use of FET is substantially increasing, it is important to tease out whether the increased cancer risk is a true risk increase due to the ART procedures using FET, or due to chance or confounding by other factors,” authors of the 2019 Dutch study, Mandy Spaan, PhD, and Flora E. van Leeuwen, PhD, said in an interview.
“But, as childhood cancer is (fortunately) a rare disease, it is very difficult to study this research question among ART children due to limited numbers,” said Dr. Spaan and Dr. van Leeuwen, who are with the Netherlands Cancer Institute.
Given this, the two experts call for additional large population-based cohort studies to investigate the risk of cancer after ART, especially FET, and for a subsequent analysis that pools these data. They hope this strategy “will lead to reliable estimates” and provide information on the risks of FET in comparison with approaches that involve fresh embryos.
The current study had no commercial funding. The study authors as well as Dr. Spaan and Dr. van Leeuwen have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PLOS MEDICINE
Natural fertility: When less can be more
As reproductive specialists, part of our obligation is to improve a woman’s or couple’s ability to conceive in the most cost-effective manner, ideally through natural attempts at conception. While assisted reproductive technologies (ART) have provided impressive pregnancy rates across many diagnoses, including unexplained infertility, this advanced procedure comes with a significant financial cost to those without insurance and an emotional burden from the lack of a guaranteed outcome. Infertility procedures have minimal associated but potentially significant risks, most importantly multiple gestations. Contrary to popular belief, ovulation induction with intrauterine insemination (IUI) treatment has a greater risk of high-order multiple gestation when compared with IVF, given the inability of the former to control the number of embryos that may enter and implant in the endometrial cavity and the increased use of single embryo transfers with the latter. The specialist should evaluate the woman or couple for the basic issues of ovulation, tubal, and sperm function, as well as for lifestyle and environmental factors that can impede reproduction. As a result, “one size fits all” should not apply to patients, specifically those with infertility. This month’s column will present the detrimental effect of environmental and lifestyle factors on the goal of enhancing fertility through natural cycles of urine luteinizing-hormone timed intercourse.
Nutrition
Often overlooked in the infertility evaluation, an optimal diet improves fertility for both partners. Processed meat has been associated with reduced sperm quality. In ART, red meat has been associated with decreased embryo blastocyst formation. Lower trans fatty acids and higher omega-3s may improve fecundity. Considered one of the best overall diets, the Mediterranean diet consists of plant-based foods, such as whole grains, vegetables, legumes, fruits, nuts, seeds, herbs, and spices. Olive oil is the main source of added fat whereas fish, seafood, dairy, and poultry should be eaten in moderation. Fatty fish, such as mackerel, herring, sardines, albacore tuna, and salmon, are rich in omega-3 fatty acids, which have been shown to improve fecundity and IVF success, and have a positive association with blastocyst embryo development.1-3
Stress
The emotional effect of an infertility diagnosis has been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities.4 Whether stress causes or is a result of infertility has been a longstanding debate.5 Nevertheless, stress is the number-one reason patients discontinue fertility treatment.6 As fertility specialists, we must be cognizant of the devastation endured by infertility patients and maintain an open dialogue, as well as provide resources for coping strategies and counseling.
One popular method of improving mental health and fertility has been acupuncture. Initial enthusiasm originated from one of the first studies to explore the use of acupuncture during IVF. This was a prospective randomized study that showed treated patients had an approximately 100% improvement in clinical pregnancy rate. Unfortunately, there was no appropriate control group, just untreated controls.7 A subsequent study by the same investigator added a placebo acupuncture control group and did not show a statistically significant increase in pregnancy rates.8 Finally, a meta-analysis and reanalysis did not demonstrate any improvement in pregnancy outcome, whereas three of the studies analyzed suggested a possible reduction in pregnancies; placebo acupuncture was shown to have a higher success rate.9-11 While acupuncture is relatively safe, there appears to be only a placebo effect that may be helpful.
The effect of stress on reproduction has been addressed in one of my previous columns.
Alcohol and caffeine
The damaging effects of alcohol on the fetus during pregnancy are legion – abnormal facial features, microcephaly, low birth weight, hyperactive behavior, vision or hearing deficits, speech and language delays, and intellectual disability. Less known is the amount of alcohol that may have an effect during preconception. One of the first reports on the effect of alcohol on IVF concluded: a 13% decrease in the number of eggs aspirated; a 2.86 times increase in risk of not achieving pregnancy; and a 2.21 times increase in risk of miscarriage. For men, one additional drink per day increased the risk of not achieving a live birth from 2.28 to 8.32 times.12 Subsequent studies demonstrate a 16% reduction in IVF pregnancies in women who have at least four drinks per week; when the couple drank at least four drinks per week, the pregnancy rate decreased by 21%.13
However, a study from Denmark did not demonstrate a negative effect of low to moderate pretreatment amounts of alcohol and caffeine on IVF outcomes.14 Nevertheless, there is evidence that reducing or abstaining from alcohol intake may improve IVF outcomes.15 While there have been reports of higher miscarriage rates from caffeine,16,17 not all reports support a negative association.18
Smoking
The use of tobacco has been estimated to contribute to 13% of female infertility in a dose-response manner, including secondhand smoke. During ART, smoking reduces ovarian response to gonadotropins and decreases IVF success by up to 50%. Discontinuing smoking for 6 months beforehand appears to restore normal outcomes.19-20
The American Society for Reproductive Medicine Practice Committee on smoking provides the following invaluable information to share with patients on the harmful reproductive effects of smoking:21
- Early menopause by accelerating the loss of eggs.
- Higher rates of miscarriage and ectopic pregnancy.
- A decrease in sperm function.
- Possible genetic damage to eggs and sperm.
- Reduced sperm in son from maternal smoking.
Weight and exercise
Compared with normal-weight women, those with obesity are three times more likely to have ovulatory dysfunction;22 a lower chance for conception;23 and infertility.24 Obese women have higher rates of miscarriage and recurrent miscarriage, reduced success with ART, an increased number of canceled cycles, and poorer quality oocytes retrieved. During pregnancy, obese women have three to four times higher rates of gestational diabetes and preeclampsia,25 as well as likelihood of having a fetus with macrosomia and birth defects, and a 1.3-2.1 times higher risk of stillbirth.26
Regarding physical activity, the rate of pregnancies (39.0% vs. 16.0%, P = .002) and live births (24.4% vs. 7.4% (P = .004) were higher with regular exercise vs. being sedentary. Obese women who exercised regularly had a live birth rate over threefold higher compared with those who were not active.27 Moderation should be employed given that women who exercise to exhaustion have 2.3 times the odds of fertility problems.28 In men, obesity has been shown to increase estrogens and reduce spermatogenesis. Exercise has improved semen parameters and testosterone. Paternal physical and sedentary activities were not related to clinical pregnancy or live birth rates following infertility treatment.29 As in women, men experience negative effects from high-intensity exercise, including bicycling, which can result in decreased semen parameters, follicle-stimulating hormone, LH, and testosterone levels.30
In couples desiring a more natural approach to infertility, fertility specialists can address environmental and lifestyle factors that may improve reproduction. When natural attempts at conception are not applicable or successful, IUI and ART are appropriate treatment options after considering estimated success rates as well as the physical, emotional, and financial investment of patients.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Wise LA et al. Am J Epidemiol. 2018;187:60-74.
2. Chui Y-H. Hum Reprod. 2018;33:156-65.
3. Ferreira Braga DPA et al. Reprod Biomed Online. 2015;31:30-8.
4. Domar AD et al. J Psychosom Obstet Gynaecol. 1993;14[suppl]:45-52.
5. Trolice MP. J Assist Reprod Genet. 2021 Apr;38[4]:873-5.
6. Gameiro S et al. Hum Reprod Update. 2012;18[6]:652-69.
7. Paulus WE et al. Fertil Steril. 2002;77:721-4.
8. Paulus WE et al. Hum Reprod. 2003;18:S18(abstr).
9. Wing SSE et al. Hum Reprod. 2009;24:341-8.
10. Hong Zheng C et al. Fertil Steril. 2012;97:599-611.
11. Meldrum DR et al. Fertil Steril. 2013;99:1821-4.
12. Klonoff-Cohen H et al. Fertil Steril. 2003;79:330-9.
13. Rossi BV et al. Obstet Gynecol. 2011;117:136-42.
14. Abadia L et al. Hum Reprod. 2017;32:1846-54.
15. Gormack AA et al. Hum Reprod. 2015;30:1617.
16. James JE. BMJ Evid Based Med. 2021;26:114-15.
17. Gaskins AJ et al. Eur J Nutr. 2018 Feb;57:107-17.
18. Machtinger R et al. Fertil Steril. 2017;108:1026-33.
19. Hughes EG et al. Fertil Steril. 1994;62:807.
20. de Ziegler D et al. Fertil Steril. 2013;100:927-8.
21. Practice Committee of the American Society for Reproductive Medicine. Fertil Steril. 2018;110:611-8.
22. Brewer CJ, Balen AH. Reproduction. 2010;140:347-64.
23. Wise LA et al. Hum Reprod. 2010;25:253-64.
24. Silvestris S et al. Reprod Biol Endocrinol. 2018;16[1]:22.
25. Alwash SM et al. Obes Res Clin Pract. 2021;15:425-30.
26. Aune D et al. JAMA. 2014;311:1536-46.
27. Palomba S et al. Reprod Biomed Online. 2014;29:72-9.
28. Gudmundsdottir SL et al. Hum Reprod. 2009;24[12]:3196-204.
29. Gaskins AJ et al. Hum Reprod. 2014;29:2575-82.
30. Wise LA et al. Fertil Steril. 2011;95:1025-30.
As reproductive specialists, part of our obligation is to improve a woman’s or couple’s ability to conceive in the most cost-effective manner, ideally through natural attempts at conception. While assisted reproductive technologies (ART) have provided impressive pregnancy rates across many diagnoses, including unexplained infertility, this advanced procedure comes with a significant financial cost to those without insurance and an emotional burden from the lack of a guaranteed outcome. Infertility procedures have minimal associated but potentially significant risks, most importantly multiple gestations. Contrary to popular belief, ovulation induction with intrauterine insemination (IUI) treatment has a greater risk of high-order multiple gestation when compared with IVF, given the inability of the former to control the number of embryos that may enter and implant in the endometrial cavity and the increased use of single embryo transfers with the latter. The specialist should evaluate the woman or couple for the basic issues of ovulation, tubal, and sperm function, as well as for lifestyle and environmental factors that can impede reproduction. As a result, “one size fits all” should not apply to patients, specifically those with infertility. This month’s column will present the detrimental effect of environmental and lifestyle factors on the goal of enhancing fertility through natural cycles of urine luteinizing-hormone timed intercourse.
Nutrition
Often overlooked in the infertility evaluation, an optimal diet improves fertility for both partners. Processed meat has been associated with reduced sperm quality. In ART, red meat has been associated with decreased embryo blastocyst formation. Lower trans fatty acids and higher omega-3s may improve fecundity. Considered one of the best overall diets, the Mediterranean diet consists of plant-based foods, such as whole grains, vegetables, legumes, fruits, nuts, seeds, herbs, and spices. Olive oil is the main source of added fat whereas fish, seafood, dairy, and poultry should be eaten in moderation. Fatty fish, such as mackerel, herring, sardines, albacore tuna, and salmon, are rich in omega-3 fatty acids, which have been shown to improve fecundity and IVF success, and have a positive association with blastocyst embryo development.1-3
Stress
The emotional effect of an infertility diagnosis has been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities.4 Whether stress causes or is a result of infertility has been a longstanding debate.5 Nevertheless, stress is the number-one reason patients discontinue fertility treatment.6 As fertility specialists, we must be cognizant of the devastation endured by infertility patients and maintain an open dialogue, as well as provide resources for coping strategies and counseling.
One popular method of improving mental health and fertility has been acupuncture. Initial enthusiasm originated from one of the first studies to explore the use of acupuncture during IVF. This was a prospective randomized study that showed treated patients had an approximately 100% improvement in clinical pregnancy rate. Unfortunately, there was no appropriate control group, just untreated controls.7 A subsequent study by the same investigator added a placebo acupuncture control group and did not show a statistically significant increase in pregnancy rates.8 Finally, a meta-analysis and reanalysis did not demonstrate any improvement in pregnancy outcome, whereas three of the studies analyzed suggested a possible reduction in pregnancies; placebo acupuncture was shown to have a higher success rate.9-11 While acupuncture is relatively safe, there appears to be only a placebo effect that may be helpful.
The effect of stress on reproduction has been addressed in one of my previous columns.
Alcohol and caffeine
The damaging effects of alcohol on the fetus during pregnancy are legion – abnormal facial features, microcephaly, low birth weight, hyperactive behavior, vision or hearing deficits, speech and language delays, and intellectual disability. Less known is the amount of alcohol that may have an effect during preconception. One of the first reports on the effect of alcohol on IVF concluded: a 13% decrease in the number of eggs aspirated; a 2.86 times increase in risk of not achieving pregnancy; and a 2.21 times increase in risk of miscarriage. For men, one additional drink per day increased the risk of not achieving a live birth from 2.28 to 8.32 times.12 Subsequent studies demonstrate a 16% reduction in IVF pregnancies in women who have at least four drinks per week; when the couple drank at least four drinks per week, the pregnancy rate decreased by 21%.13
However, a study from Denmark did not demonstrate a negative effect of low to moderate pretreatment amounts of alcohol and caffeine on IVF outcomes.14 Nevertheless, there is evidence that reducing or abstaining from alcohol intake may improve IVF outcomes.15 While there have been reports of higher miscarriage rates from caffeine,16,17 not all reports support a negative association.18
Smoking
The use of tobacco has been estimated to contribute to 13% of female infertility in a dose-response manner, including secondhand smoke. During ART, smoking reduces ovarian response to gonadotropins and decreases IVF success by up to 50%. Discontinuing smoking for 6 months beforehand appears to restore normal outcomes.19-20
The American Society for Reproductive Medicine Practice Committee on smoking provides the following invaluable information to share with patients on the harmful reproductive effects of smoking:21
- Early menopause by accelerating the loss of eggs.
- Higher rates of miscarriage and ectopic pregnancy.
- A decrease in sperm function.
- Possible genetic damage to eggs and sperm.
- Reduced sperm in son from maternal smoking.
Weight and exercise
Compared with normal-weight women, those with obesity are three times more likely to have ovulatory dysfunction;22 a lower chance for conception;23 and infertility.24 Obese women have higher rates of miscarriage and recurrent miscarriage, reduced success with ART, an increased number of canceled cycles, and poorer quality oocytes retrieved. During pregnancy, obese women have three to four times higher rates of gestational diabetes and preeclampsia,25 as well as likelihood of having a fetus with macrosomia and birth defects, and a 1.3-2.1 times higher risk of stillbirth.26
Regarding physical activity, the rate of pregnancies (39.0% vs. 16.0%, P = .002) and live births (24.4% vs. 7.4% (P = .004) were higher with regular exercise vs. being sedentary. Obese women who exercised regularly had a live birth rate over threefold higher compared with those who were not active.27 Moderation should be employed given that women who exercise to exhaustion have 2.3 times the odds of fertility problems.28 In men, obesity has been shown to increase estrogens and reduce spermatogenesis. Exercise has improved semen parameters and testosterone. Paternal physical and sedentary activities were not related to clinical pregnancy or live birth rates following infertility treatment.29 As in women, men experience negative effects from high-intensity exercise, including bicycling, which can result in decreased semen parameters, follicle-stimulating hormone, LH, and testosterone levels.30
In couples desiring a more natural approach to infertility, fertility specialists can address environmental and lifestyle factors that may improve reproduction. When natural attempts at conception are not applicable or successful, IUI and ART are appropriate treatment options after considering estimated success rates as well as the physical, emotional, and financial investment of patients.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Wise LA et al. Am J Epidemiol. 2018;187:60-74.
2. Chui Y-H. Hum Reprod. 2018;33:156-65.
3. Ferreira Braga DPA et al. Reprod Biomed Online. 2015;31:30-8.
4. Domar AD et al. J Psychosom Obstet Gynaecol. 1993;14[suppl]:45-52.
5. Trolice MP. J Assist Reprod Genet. 2021 Apr;38[4]:873-5.
6. Gameiro S et al. Hum Reprod Update. 2012;18[6]:652-69.
7. Paulus WE et al. Fertil Steril. 2002;77:721-4.
8. Paulus WE et al. Hum Reprod. 2003;18:S18(abstr).
9. Wing SSE et al. Hum Reprod. 2009;24:341-8.
10. Hong Zheng C et al. Fertil Steril. 2012;97:599-611.
11. Meldrum DR et al. Fertil Steril. 2013;99:1821-4.
12. Klonoff-Cohen H et al. Fertil Steril. 2003;79:330-9.
13. Rossi BV et al. Obstet Gynecol. 2011;117:136-42.
14. Abadia L et al. Hum Reprod. 2017;32:1846-54.
15. Gormack AA et al. Hum Reprod. 2015;30:1617.
16. James JE. BMJ Evid Based Med. 2021;26:114-15.
17. Gaskins AJ et al. Eur J Nutr. 2018 Feb;57:107-17.
18. Machtinger R et al. Fertil Steril. 2017;108:1026-33.
19. Hughes EG et al. Fertil Steril. 1994;62:807.
20. de Ziegler D et al. Fertil Steril. 2013;100:927-8.
21. Practice Committee of the American Society for Reproductive Medicine. Fertil Steril. 2018;110:611-8.
22. Brewer CJ, Balen AH. Reproduction. 2010;140:347-64.
23. Wise LA et al. Hum Reprod. 2010;25:253-64.
24. Silvestris S et al. Reprod Biol Endocrinol. 2018;16[1]:22.
25. Alwash SM et al. Obes Res Clin Pract. 2021;15:425-30.
26. Aune D et al. JAMA. 2014;311:1536-46.
27. Palomba S et al. Reprod Biomed Online. 2014;29:72-9.
28. Gudmundsdottir SL et al. Hum Reprod. 2009;24[12]:3196-204.
29. Gaskins AJ et al. Hum Reprod. 2014;29:2575-82.
30. Wise LA et al. Fertil Steril. 2011;95:1025-30.
As reproductive specialists, part of our obligation is to improve a woman’s or couple’s ability to conceive in the most cost-effective manner, ideally through natural attempts at conception. While assisted reproductive technologies (ART) have provided impressive pregnancy rates across many diagnoses, including unexplained infertility, this advanced procedure comes with a significant financial cost to those without insurance and an emotional burden from the lack of a guaranteed outcome. Infertility procedures have minimal associated but potentially significant risks, most importantly multiple gestations. Contrary to popular belief, ovulation induction with intrauterine insemination (IUI) treatment has a greater risk of high-order multiple gestation when compared with IVF, given the inability of the former to control the number of embryos that may enter and implant in the endometrial cavity and the increased use of single embryo transfers with the latter. The specialist should evaluate the woman or couple for the basic issues of ovulation, tubal, and sperm function, as well as for lifestyle and environmental factors that can impede reproduction. As a result, “one size fits all” should not apply to patients, specifically those with infertility. This month’s column will present the detrimental effect of environmental and lifestyle factors on the goal of enhancing fertility through natural cycles of urine luteinizing-hormone timed intercourse.
Nutrition
Often overlooked in the infertility evaluation, an optimal diet improves fertility for both partners. Processed meat has been associated with reduced sperm quality. In ART, red meat has been associated with decreased embryo blastocyst formation. Lower trans fatty acids and higher omega-3s may improve fecundity. Considered one of the best overall diets, the Mediterranean diet consists of plant-based foods, such as whole grains, vegetables, legumes, fruits, nuts, seeds, herbs, and spices. Olive oil is the main source of added fat whereas fish, seafood, dairy, and poultry should be eaten in moderation. Fatty fish, such as mackerel, herring, sardines, albacore tuna, and salmon, are rich in omega-3 fatty acids, which have been shown to improve fecundity and IVF success, and have a positive association with blastocyst embryo development.1-3
Stress
The emotional effect of an infertility diagnosis has been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities.4 Whether stress causes or is a result of infertility has been a longstanding debate.5 Nevertheless, stress is the number-one reason patients discontinue fertility treatment.6 As fertility specialists, we must be cognizant of the devastation endured by infertility patients and maintain an open dialogue, as well as provide resources for coping strategies and counseling.
One popular method of improving mental health and fertility has been acupuncture. Initial enthusiasm originated from one of the first studies to explore the use of acupuncture during IVF. This was a prospective randomized study that showed treated patients had an approximately 100% improvement in clinical pregnancy rate. Unfortunately, there was no appropriate control group, just untreated controls.7 A subsequent study by the same investigator added a placebo acupuncture control group and did not show a statistically significant increase in pregnancy rates.8 Finally, a meta-analysis and reanalysis did not demonstrate any improvement in pregnancy outcome, whereas three of the studies analyzed suggested a possible reduction in pregnancies; placebo acupuncture was shown to have a higher success rate.9-11 While acupuncture is relatively safe, there appears to be only a placebo effect that may be helpful.
The effect of stress on reproduction has been addressed in one of my previous columns.
Alcohol and caffeine
The damaging effects of alcohol on the fetus during pregnancy are legion – abnormal facial features, microcephaly, low birth weight, hyperactive behavior, vision or hearing deficits, speech and language delays, and intellectual disability. Less known is the amount of alcohol that may have an effect during preconception. One of the first reports on the effect of alcohol on IVF concluded: a 13% decrease in the number of eggs aspirated; a 2.86 times increase in risk of not achieving pregnancy; and a 2.21 times increase in risk of miscarriage. For men, one additional drink per day increased the risk of not achieving a live birth from 2.28 to 8.32 times.12 Subsequent studies demonstrate a 16% reduction in IVF pregnancies in women who have at least four drinks per week; when the couple drank at least four drinks per week, the pregnancy rate decreased by 21%.13
However, a study from Denmark did not demonstrate a negative effect of low to moderate pretreatment amounts of alcohol and caffeine on IVF outcomes.14 Nevertheless, there is evidence that reducing or abstaining from alcohol intake may improve IVF outcomes.15 While there have been reports of higher miscarriage rates from caffeine,16,17 not all reports support a negative association.18
Smoking
The use of tobacco has been estimated to contribute to 13% of female infertility in a dose-response manner, including secondhand smoke. During ART, smoking reduces ovarian response to gonadotropins and decreases IVF success by up to 50%. Discontinuing smoking for 6 months beforehand appears to restore normal outcomes.19-20
The American Society for Reproductive Medicine Practice Committee on smoking provides the following invaluable information to share with patients on the harmful reproductive effects of smoking:21
- Early menopause by accelerating the loss of eggs.
- Higher rates of miscarriage and ectopic pregnancy.
- A decrease in sperm function.
- Possible genetic damage to eggs and sperm.
- Reduced sperm in son from maternal smoking.
Weight and exercise
Compared with normal-weight women, those with obesity are three times more likely to have ovulatory dysfunction;22 a lower chance for conception;23 and infertility.24 Obese women have higher rates of miscarriage and recurrent miscarriage, reduced success with ART, an increased number of canceled cycles, and poorer quality oocytes retrieved. During pregnancy, obese women have three to four times higher rates of gestational diabetes and preeclampsia,25 as well as likelihood of having a fetus with macrosomia and birth defects, and a 1.3-2.1 times higher risk of stillbirth.26
Regarding physical activity, the rate of pregnancies (39.0% vs. 16.0%, P = .002) and live births (24.4% vs. 7.4% (P = .004) were higher with regular exercise vs. being sedentary. Obese women who exercised regularly had a live birth rate over threefold higher compared with those who were not active.27 Moderation should be employed given that women who exercise to exhaustion have 2.3 times the odds of fertility problems.28 In men, obesity has been shown to increase estrogens and reduce spermatogenesis. Exercise has improved semen parameters and testosterone. Paternal physical and sedentary activities were not related to clinical pregnancy or live birth rates following infertility treatment.29 As in women, men experience negative effects from high-intensity exercise, including bicycling, which can result in decreased semen parameters, follicle-stimulating hormone, LH, and testosterone levels.30
In couples desiring a more natural approach to infertility, fertility specialists can address environmental and lifestyle factors that may improve reproduction. When natural attempts at conception are not applicable or successful, IUI and ART are appropriate treatment options after considering estimated success rates as well as the physical, emotional, and financial investment of patients.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Wise LA et al. Am J Epidemiol. 2018;187:60-74.
2. Chui Y-H. Hum Reprod. 2018;33:156-65.
3. Ferreira Braga DPA et al. Reprod Biomed Online. 2015;31:30-8.
4. Domar AD et al. J Psychosom Obstet Gynaecol. 1993;14[suppl]:45-52.
5. Trolice MP. J Assist Reprod Genet. 2021 Apr;38[4]:873-5.
6. Gameiro S et al. Hum Reprod Update. 2012;18[6]:652-69.
7. Paulus WE et al. Fertil Steril. 2002;77:721-4.
8. Paulus WE et al. Hum Reprod. 2003;18:S18(abstr).
9. Wing SSE et al. Hum Reprod. 2009;24:341-8.
10. Hong Zheng C et al. Fertil Steril. 2012;97:599-611.
11. Meldrum DR et al. Fertil Steril. 2013;99:1821-4.
12. Klonoff-Cohen H et al. Fertil Steril. 2003;79:330-9.
13. Rossi BV et al. Obstet Gynecol. 2011;117:136-42.
14. Abadia L et al. Hum Reprod. 2017;32:1846-54.
15. Gormack AA et al. Hum Reprod. 2015;30:1617.
16. James JE. BMJ Evid Based Med. 2021;26:114-15.
17. Gaskins AJ et al. Eur J Nutr. 2018 Feb;57:107-17.
18. Machtinger R et al. Fertil Steril. 2017;108:1026-33.
19. Hughes EG et al. Fertil Steril. 1994;62:807.
20. de Ziegler D et al. Fertil Steril. 2013;100:927-8.
21. Practice Committee of the American Society for Reproductive Medicine. Fertil Steril. 2018;110:611-8.
22. Brewer CJ, Balen AH. Reproduction. 2010;140:347-64.
23. Wise LA et al. Hum Reprod. 2010;25:253-64.
24. Silvestris S et al. Reprod Biol Endocrinol. 2018;16[1]:22.
25. Alwash SM et al. Obes Res Clin Pract. 2021;15:425-30.
26. Aune D et al. JAMA. 2014;311:1536-46.
27. Palomba S et al. Reprod Biomed Online. 2014;29:72-9.
28. Gudmundsdottir SL et al. Hum Reprod. 2009;24[12]:3196-204.
29. Gaskins AJ et al. Hum Reprod. 2014;29:2575-82.
30. Wise LA et al. Fertil Steril. 2011;95:1025-30.
New ovulatory disorder classifications from FIGO replace 50-year-old system
The first major revision in the systematic description of ovulatory disorders in nearly 50 years has been proposed by a consensus of experts organized by the International Federation of Gynecology and Obstetrics.
“The FIGO HyPO-P system for the classification of ovulatory disorders is submitted for consideration as a worldwide standard,” according to the writing committee, who published their methodology and their proposed applications in the International Journal of Gynecology and Obstetrics.
The classification system was created to replace the much-modified World Health Organization system first described in 1973. Since that time, many modifications have been proposed to accommodate advances in imaging and new information about underlying pathologies, but there has been no subsequent authoritative reference with these modifications or any other newer organizing system.
The new consensus was developed under the aegis of FIGO, but the development group consisted of representatives from national organizations and the major subspecialty societies. Recognized experts in ovulatory disorders and representatives from lay advocacy organizations also participated.
The HyPO-P system is based largely on anatomy. The acronym refers to ovulatory disorders related to the hypothalamus (type I), the pituitary (type II), and the ovary (type III).
Polycystic ovary syndrome (PCOS), one of the most common ovulatory disorders, was given a separate category (type IV) because of its complexity as well as the fact that PCOS is a heterogeneous systemic disorder with manifestations not limited to an impact on ovarian function.
As the first level of classification, three of the four primary categories (I-III) focus attention on the dominant anatomic source of the change in ovulatory function. The original WHO classification system identified as many as seven major groups, but they were based primarily on assays for gonadotropins and estradiol.
The new system “provides a different structure for determining the diagnosis. Blood tests are not a necessary first step,” explained Malcolm G. Munro, MD, clinical professor, department of obstetrics and gynecology, University of California, Los Angeles. Dr. Munro was the first author of the publication.
The classification system “is not as focused on the specific steps for investigation of ovulatory dysfunction as much as it explains how to structure an investigation of the girl or woman with an ovulatory disorder and then how to characterize the underlying cause,” Dr. Munro said in an interview. “It is designed to allow everyone, whether clinicians, researchers, or patients, to speak the same language.”
New system employs four categories
The four primary categories provide just the first level of classification. The next step is encapsulated in the GAIN-FIT-PIE acronym, which frames the presumed or documented categories of etiologies for the primary categories. GAIN stands for genetic, autoimmune, iatrogenic, or neoplasm etiologies. FIT stands for functional, infectious/inflammatory, or trauma and vascular etiologies. PIE stands for physiological, idiopathic, and endocrine etiologies.
By this methodology, a patient with irregular menses, galactorrhea, and elevated prolactin and an MRI showing a pituitary tumor would be identified a type 2-N, signifying pituitary (type 2) involvement with a neoplasm (N).
A third level of classification permits specific diagnostic entities to be named, allowing the patient in the example above to receive a diagnosis of a prolactin-secreting adenoma.
Not all etiologies can be identified with current diagnostic studies, even assuming clinicians have access to the resources, such as advanced imaging, that will increase diagnostic yield. As a result, the authors acknowledged that the classification system will be “aspirational” in at least some patients, but the structure of this system is expected to lead to greater precision in understanding the causes and defining features of ovulatory disorders, which, in turn, might facilitate new research initiatives.
In the published report, diagnostic protocols based on symptoms were described as being “beyond the spectrum” of this initial description. Rather, Dr. Munro explained that the most important contribution of this new classification system are standardization and communication. The system will be amenable for educating trainees and patients, for communicating between clinicians, and as a framework for research where investigators focus on more homogeneous populations of patients.
“There are many causes of ovulatory disorders that are not related to ovarian function. This is one message. Another is that ovulatory disorders are not binary. They occur on a spectrum. These range from transient instances of delayed or failed ovulation to chronic anovulation,” he said.
The new system is “ a welcome update,” according to Mark P. Trolice, MD, director of the IVF Center and professor of obstetrics and gynecology at the University of Central Florida, both in Orlando.
Dr. Trolice pointed to the clinical value of placing PCOS in a separate category. He noted that it affects 8%-13% of women, making it the most common single cause of ovulatory dysfunction.
“Another area that required clarification from prior WHO classifications was hyperprolactinemia, which is now placed in the type II category,” Dr. Trolice said in an interview.
Better terminology can help address a complex set of disorders with multiple causes and variable manifestations.
“In the evaluation of ovulation dysfunction, it is important to remember that regular menstrual intervals do not ensure ovulation,” Dr. Trolice pointed out. Even though a serum progesterone level of higher than 3 ng/mL is one of the simplest laboratory markers for ovulation, this level, he noted, “can vary through the luteal phase and even throughout the day.”
The proposed classification system, while providing a framework for describing ovulatory disorders, is designed to be adaptable, permitting advances in the understanding of the causes of ovulatory dysfunction, in the diagnosis of the causes, and in the treatments to be incorporated.
“No system should be considered permanent,” according to Dr. Munro and his coauthors. “Review and careful modification and revision should be carried out regularly.”
Dr. Munro reports financial relationships with AbbVie, American Regent, Daiichi Sankyo, Hologic, Myovant, and Pharmacosmos. Dr. Trolice reports no potential conflicts of interest.
The first major revision in the systematic description of ovulatory disorders in nearly 50 years has been proposed by a consensus of experts organized by the International Federation of Gynecology and Obstetrics.
“The FIGO HyPO-P system for the classification of ovulatory disorders is submitted for consideration as a worldwide standard,” according to the writing committee, who published their methodology and their proposed applications in the International Journal of Gynecology and Obstetrics.
The classification system was created to replace the much-modified World Health Organization system first described in 1973. Since that time, many modifications have been proposed to accommodate advances in imaging and new information about underlying pathologies, but there has been no subsequent authoritative reference with these modifications or any other newer organizing system.
The new consensus was developed under the aegis of FIGO, but the development group consisted of representatives from national organizations and the major subspecialty societies. Recognized experts in ovulatory disorders and representatives from lay advocacy organizations also participated.
The HyPO-P system is based largely on anatomy. The acronym refers to ovulatory disorders related to the hypothalamus (type I), the pituitary (type II), and the ovary (type III).
Polycystic ovary syndrome (PCOS), one of the most common ovulatory disorders, was given a separate category (type IV) because of its complexity as well as the fact that PCOS is a heterogeneous systemic disorder with manifestations not limited to an impact on ovarian function.
As the first level of classification, three of the four primary categories (I-III) focus attention on the dominant anatomic source of the change in ovulatory function. The original WHO classification system identified as many as seven major groups, but they were based primarily on assays for gonadotropins and estradiol.
The new system “provides a different structure for determining the diagnosis. Blood tests are not a necessary first step,” explained Malcolm G. Munro, MD, clinical professor, department of obstetrics and gynecology, University of California, Los Angeles. Dr. Munro was the first author of the publication.
The classification system “is not as focused on the specific steps for investigation of ovulatory dysfunction as much as it explains how to structure an investigation of the girl or woman with an ovulatory disorder and then how to characterize the underlying cause,” Dr. Munro said in an interview. “It is designed to allow everyone, whether clinicians, researchers, or patients, to speak the same language.”
New system employs four categories
The four primary categories provide just the first level of classification. The next step is encapsulated in the GAIN-FIT-PIE acronym, which frames the presumed or documented categories of etiologies for the primary categories. GAIN stands for genetic, autoimmune, iatrogenic, or neoplasm etiologies. FIT stands for functional, infectious/inflammatory, or trauma and vascular etiologies. PIE stands for physiological, idiopathic, and endocrine etiologies.
By this methodology, a patient with irregular menses, galactorrhea, and elevated prolactin and an MRI showing a pituitary tumor would be identified a type 2-N, signifying pituitary (type 2) involvement with a neoplasm (N).
A third level of classification permits specific diagnostic entities to be named, allowing the patient in the example above to receive a diagnosis of a prolactin-secreting adenoma.
Not all etiologies can be identified with current diagnostic studies, even assuming clinicians have access to the resources, such as advanced imaging, that will increase diagnostic yield. As a result, the authors acknowledged that the classification system will be “aspirational” in at least some patients, but the structure of this system is expected to lead to greater precision in understanding the causes and defining features of ovulatory disorders, which, in turn, might facilitate new research initiatives.
In the published report, diagnostic protocols based on symptoms were described as being “beyond the spectrum” of this initial description. Rather, Dr. Munro explained that the most important contribution of this new classification system are standardization and communication. The system will be amenable for educating trainees and patients, for communicating between clinicians, and as a framework for research where investigators focus on more homogeneous populations of patients.
“There are many causes of ovulatory disorders that are not related to ovarian function. This is one message. Another is that ovulatory disorders are not binary. They occur on a spectrum. These range from transient instances of delayed or failed ovulation to chronic anovulation,” he said.
The new system is “ a welcome update,” according to Mark P. Trolice, MD, director of the IVF Center and professor of obstetrics and gynecology at the University of Central Florida, both in Orlando.
Dr. Trolice pointed to the clinical value of placing PCOS in a separate category. He noted that it affects 8%-13% of women, making it the most common single cause of ovulatory dysfunction.
“Another area that required clarification from prior WHO classifications was hyperprolactinemia, which is now placed in the type II category,” Dr. Trolice said in an interview.
Better terminology can help address a complex set of disorders with multiple causes and variable manifestations.
“In the evaluation of ovulation dysfunction, it is important to remember that regular menstrual intervals do not ensure ovulation,” Dr. Trolice pointed out. Even though a serum progesterone level of higher than 3 ng/mL is one of the simplest laboratory markers for ovulation, this level, he noted, “can vary through the luteal phase and even throughout the day.”
The proposed classification system, while providing a framework for describing ovulatory disorders, is designed to be adaptable, permitting advances in the understanding of the causes of ovulatory dysfunction, in the diagnosis of the causes, and in the treatments to be incorporated.
“No system should be considered permanent,” according to Dr. Munro and his coauthors. “Review and careful modification and revision should be carried out regularly.”
Dr. Munro reports financial relationships with AbbVie, American Regent, Daiichi Sankyo, Hologic, Myovant, and Pharmacosmos. Dr. Trolice reports no potential conflicts of interest.
The first major revision in the systematic description of ovulatory disorders in nearly 50 years has been proposed by a consensus of experts organized by the International Federation of Gynecology and Obstetrics.
“The FIGO HyPO-P system for the classification of ovulatory disorders is submitted for consideration as a worldwide standard,” according to the writing committee, who published their methodology and their proposed applications in the International Journal of Gynecology and Obstetrics.
The classification system was created to replace the much-modified World Health Organization system first described in 1973. Since that time, many modifications have been proposed to accommodate advances in imaging and new information about underlying pathologies, but there has been no subsequent authoritative reference with these modifications or any other newer organizing system.
The new consensus was developed under the aegis of FIGO, but the development group consisted of representatives from national organizations and the major subspecialty societies. Recognized experts in ovulatory disorders and representatives from lay advocacy organizations also participated.
The HyPO-P system is based largely on anatomy. The acronym refers to ovulatory disorders related to the hypothalamus (type I), the pituitary (type II), and the ovary (type III).
Polycystic ovary syndrome (PCOS), one of the most common ovulatory disorders, was given a separate category (type IV) because of its complexity as well as the fact that PCOS is a heterogeneous systemic disorder with manifestations not limited to an impact on ovarian function.
As the first level of classification, three of the four primary categories (I-III) focus attention on the dominant anatomic source of the change in ovulatory function. The original WHO classification system identified as many as seven major groups, but they were based primarily on assays for gonadotropins and estradiol.
The new system “provides a different structure for determining the diagnosis. Blood tests are not a necessary first step,” explained Malcolm G. Munro, MD, clinical professor, department of obstetrics and gynecology, University of California, Los Angeles. Dr. Munro was the first author of the publication.
The classification system “is not as focused on the specific steps for investigation of ovulatory dysfunction as much as it explains how to structure an investigation of the girl or woman with an ovulatory disorder and then how to characterize the underlying cause,” Dr. Munro said in an interview. “It is designed to allow everyone, whether clinicians, researchers, or patients, to speak the same language.”
New system employs four categories
The four primary categories provide just the first level of classification. The next step is encapsulated in the GAIN-FIT-PIE acronym, which frames the presumed or documented categories of etiologies for the primary categories. GAIN stands for genetic, autoimmune, iatrogenic, or neoplasm etiologies. FIT stands for functional, infectious/inflammatory, or trauma and vascular etiologies. PIE stands for physiological, idiopathic, and endocrine etiologies.
By this methodology, a patient with irregular menses, galactorrhea, and elevated prolactin and an MRI showing a pituitary tumor would be identified a type 2-N, signifying pituitary (type 2) involvement with a neoplasm (N).
A third level of classification permits specific diagnostic entities to be named, allowing the patient in the example above to receive a diagnosis of a prolactin-secreting adenoma.
Not all etiologies can be identified with current diagnostic studies, even assuming clinicians have access to the resources, such as advanced imaging, that will increase diagnostic yield. As a result, the authors acknowledged that the classification system will be “aspirational” in at least some patients, but the structure of this system is expected to lead to greater precision in understanding the causes and defining features of ovulatory disorders, which, in turn, might facilitate new research initiatives.
In the published report, diagnostic protocols based on symptoms were described as being “beyond the spectrum” of this initial description. Rather, Dr. Munro explained that the most important contribution of this new classification system are standardization and communication. The system will be amenable for educating trainees and patients, for communicating between clinicians, and as a framework for research where investigators focus on more homogeneous populations of patients.
“There are many causes of ovulatory disorders that are not related to ovarian function. This is one message. Another is that ovulatory disorders are not binary. They occur on a spectrum. These range from transient instances of delayed or failed ovulation to chronic anovulation,” he said.
The new system is “ a welcome update,” according to Mark P. Trolice, MD, director of the IVF Center and professor of obstetrics and gynecology at the University of Central Florida, both in Orlando.
Dr. Trolice pointed to the clinical value of placing PCOS in a separate category. He noted that it affects 8%-13% of women, making it the most common single cause of ovulatory dysfunction.
“Another area that required clarification from prior WHO classifications was hyperprolactinemia, which is now placed in the type II category,” Dr. Trolice said in an interview.
Better terminology can help address a complex set of disorders with multiple causes and variable manifestations.
“In the evaluation of ovulation dysfunction, it is important to remember that regular menstrual intervals do not ensure ovulation,” Dr. Trolice pointed out. Even though a serum progesterone level of higher than 3 ng/mL is one of the simplest laboratory markers for ovulation, this level, he noted, “can vary through the luteal phase and even throughout the day.”
The proposed classification system, while providing a framework for describing ovulatory disorders, is designed to be adaptable, permitting advances in the understanding of the causes of ovulatory dysfunction, in the diagnosis of the causes, and in the treatments to be incorporated.
“No system should be considered permanent,” according to Dr. Munro and his coauthors. “Review and careful modification and revision should be carried out regularly.”
Dr. Munro reports financial relationships with AbbVie, American Regent, Daiichi Sankyo, Hologic, Myovant, and Pharmacosmos. Dr. Trolice reports no potential conflicts of interest.
FROM INTERNATIONAL JOURNAL OF GYNECOLOGY AND OBSTETRICS
State of the science in PCOS: Emerging neuroendocrine involvement driving research
Polycystic ovary syndrome (PCOS) affects an estimated 8%-13% of women, and yet “it has been quite a black box for many years,” as Margo Hudson, MD, an assistant professor of endocrinology, diabetes, and hypertension at Harvard Medical School, Boston, puts it. That black box encompasses not only uncertainty about the etiology and pathophysiology of the condition but even what constitutes a diagnosis.
Even the international guidelines on PCOS management endorsed by the American Society for Reproductive Medicine – a document developed over 15 months with the input of 37 medical organizations covering 71 countries – notes that PCOS diagnosis is “controversial and assessment and management are inconsistent.” The result, the guidelines note, is that “the needs of women with PCOS are not being adequately met.”
One of the earliest diagnostic criteria, defined in 1990 by the National Institutes of Health, required only hyperandrogenism and irregular menstruation. Then the 2003 Rotterdam Criteria added presence of polycystic ovaries on ultrasound as a third criterion. Then the Androgen Excess Society determined that PCOS required presence of hyperandrogenism with either polycystic ovaries or oligo/amenorrhea anovulation. Yet the Endocrine Society notes that excess androgen levels are seen in 60%-80% of those with PCOS, suggesting it’s not an essential requirement for diagnosis, leaving most to diagnose it in people who have two of the three key criteria. The only real agreement on diagnosis is the need to eliminate other potential diagnoses first, making PCOS always a diagnosis of exclusion.
Further, though PCOS is known as the leading cause of infertility in women, it is more than a reproductive condition, with metabolic and psychological features as well. Then there is the range of comorbidities, none of which occur in all patients with PCOS but all of which occur in a majority and which are themselves interrelated. Insulin resistance is a common feature, occurring in 50%-70% of people with PCOS. Accordingly, metabolic syndrome occurs in at least a third of people with PCOS and type 2 diabetes prevalence is higher in those with PCOS as well.
Obesity occurs in an estimated 80% of women with PCOS in the United States, though it affects only about 50% of women with PCOS outside the United States, and those with PCOS have an increased risk of hypertension. Mood disorders, particularly anxiety and depression but also, to a lesser extent, bipolar disorder and obsessive-compulsive disorder, are more likely in people with PCOS. And given that these comorbidities are all cardiovascular risk factors, it’s unsurprising that recent studies are finding those with PCOS to be at greater risk for cardiometabolic disease and major cardiovascular events.
“The reality is that PCOS is a heterogenous entity. It’s not one thing – it’s a syndrome,” Lubna Pal, MBBS, a professor of ob.gyn. and director of the PCOS Program at Yale University, New Haven, Conn., said in an interview. A whole host of factors are likely playing a role in the causes of PCOS, and those factors interact differently within different people. “We’re looking at things like lipid metabolism, fetal origins, the gut microbiome, genetics, epigenetics, and then dietary and environmental factors,” Nichole Tyson, MD, division chief of pediatric and adolescent gynecology and a clinical associate professor at Stanford (Calif.) Medicine Children’s Health, said in an interview. And most studies have identified associations that may or may not be causal. Take, for example, endocrine disruptors. BPA levels have been shown to be higher in women with PCOS than women without, but that correlation may or may not be related to the etiology of the condition.
The hypothalamic-pituitary-gonadal axis
In trying to understand the pathophysiology of the condition, much of the latest research has zeroed in on potential mechanisms in the hypothalamic-pituitary-gonadal axis. “A consistent feature of PCOS is disordered gonadotropin secretion with elevated mean LH [luteinizing hormone], low or low normal FSH [follicle-stimulating hormone], and a persistently rapid frequency of GnRH [gonadotropin-releasing hormone] pulse secretion,” wrote authors of a scientific statement on aspects of PCOS.
“I think the balance is heading more to central neurologic control of the reproductive system and that disturbances there impact the GnRH cells in the hypothalamus, which then go on to give us the findings that we can measure peripherally with the LH-FSH ratio,” Dr. Hudson said in an interview.
The increased LH levels are thought to be a major driver of increased androgen levels. Current thinking suggests that the primary driver of increased LH is GnRH pulsatility, supported not only by human studies but by animal models as well. This leads to the question of what drives GnRH dysregulation. One hypothesis posits that GABA neurons play a role here, given findings that GABA levels in cerebrospinal fluid were higher in women with PCOS than those with normal ovulation.
But the culprit garnering the most attention is kisspeptin, a protein encoded by the KISS1 gene that stimulates GnRH neurons and has been linked to regulation of LH and FSH secretion. Kisspeptin, along with neurokinin B and dynorphin, is part of the triumvirate that comprises KNDy neurons, also recently implicated in menopausal vasomotor symptoms. Multiple systematic reviewsand meta-analyses have found a correlation between higher kisspeptin levels in the blood and higher circulating LH levels, regardless of body mass index. While kisspeptin is expressed in several tissues, including liver, pancreas, gonad, and adipose, it’s neural kisspeptin signaling that appears most likely to play a role in activating GnRH hormones and disrupting normal function of the hypothalamic-pituitary-gonadal axis.
But as noted, in at least one systematic review of kisspeptin and PCOS, “findings from animal studies suggest that kisspeptin levels are not increased in all subtypes of PCOS.” And another review found “altered” levels of kisspeptin levels in non-PCOS patients who had obesity, potentially raising questions about any associations between kisspeptin and obesity or insulin resistance.
Remaining chicken-and-egg questions
A hallmark of PCOS has long been, and continues to be, the string of chicken-or-egg questions that plague understanding of it. One of these is how depression and anxiety fit into the etiology of PCOS. Exploring the role of specific neurons that may overstimulate GnRH pulsatility may hold clues to a common underlying mechanism for the involvement of depression and anxiety in patients with PCOS, Dr. Hudson speculated. While previous assumptions often attributed depression and anxiety in PCOS to the symptoms – such as thin scalp hair and increased facial hair, excess weight, acne, and irregular periods – Dr. Hudson pointed out that women can address many of these symptoms with laser hair removal, weight loss, acne treatment, and similar interventions, yet they still have a lot of underlying mental health issues.
It’s also unclear whether metabolic factors so common with PCOS, particularly insulin resistance and obesity, are a result of the condition or are contributors to it. Is insulin resistance contributing to dysregulation in the neurons that interferes with normal functioning of the hypothalamic-pituitary-adrenal axis? Is abnormal functioning along this axis contributing to insulin resistance? Or neither? Or both? Or does it depend? The authors of one paper wrote that “insulin may play both direct and indirect roles in the pathogenesis of androgen excess in PCOS,” since insulin can “stimulate ovarian androgen production” and “enhance ovarian growth and follicular cyst formation in rats.”
Dr. Pal noted that “obesity itself can evolve into a PCOS-like picture,” raising questions about whether obesity or insulin resistance might be part of the causal pathway to PCOS, or whether either can trigger its development in those genetically predisposed.
“Obesity does appear to exacerbate many aspects of the PCOS phenotype, particularly those risk factors related to metabolic syndrome,” wrote the authors of a scientific statement on aspects of PCOS, but they add that “it is currently debated whether obesity per se can cause PCOS.” While massive weight loss in those with PCOS and obesity has improved multiple reproductive and metabolic issues, it hasn’t resolved all of them, they write.
Dr. Hudson said she expects there’s “some degree of appetite dysregulation and metabolic dysregulation” that contributes, but then there are other women who don’t have much of an appetite or overeat and still struggle with their weight. Evidence has also found insulin resistance in women of normal weight with PCOS. “There may be some kind of metabolic dysregulation that they have at some level, and others are clearly bothered by overeating,” Dr. Hudson said.
Similarly, it’s not clear whether the recent discovery of increased cardiovascular risks in people with PCOS is a result of the comorbidities so common with PCOS, such as obesity, or whether an underlying mechanism links the cardiovascular risk and the dysregulation of hormones. Dr. Pal would argue that, again, it’s probably both, depending on the patient.
Then there is the key feature of hyperandrogenemia. “An outstanding debate is whether the elevated androgens in PCOS women are merely a downstream endocrine response to hyperactive GnRH and LH secretion driving the ovary, or do the elevated androgens themselves act in the brain (or pituitary) during development and/or adulthood to sculpt and maintain the hypersecretion of GnRH and LH?” wrote Eulalia A. Coutinho, PhD, and Alexander S. Kauffman, PhD, in a 2019 review of the brain’s role in PCOS.
These problems may be bidirectional or part of various feedback loops. Sleep apnea is more common in people with PCOS, Dr. Tyson noted, but sleep apnea is also linked to cardiovascular, metabolic, and depression risks, and depression can play a role in obesity, which increases the risk of obstructive sleep apnea. “So you’re in this vicious cycle,” Dr. Tyson said. That’s why she also believes it’s important to change the dialogue and perspective on PCOS, to reduce the stigma attached to it, and work with patients to empower them in treating its symptoms and reducing their risk of comorbidities.
Recent and upcoming changes in treatment
Current treatment of PCOS already changes according to the symptoms posing the greatest problems at each stage of a person’s life, Dr. Hudson said. Younger women tend to be more bothered about the cosmetic effects of PCOS, including hair growth patterns and acne, but as they grow out of adolescence and into their 20s and 30s, infertility becomes a bigger concern for many. Then, as they start approaching menopause, metabolic and cardiovascular issues take the lead, with more of a focus on lipids, diabetes risk, and heart health.
In some ways, management of PCOS hasn’t changed much in the past several decades, except in an increased awareness of the metabolic and cardiovascular risks, which has led to more frequent screening to catch potential conditions earlier in life. What has changed, however, is improvements in the treatments used for symptoms, such as expanded bariatric surgery options and GLP-1 agonists for treating obesity. Other examples include better options for menstrual management, such as new progesterone IUDs, and optimized fertility treatments, Dr. Tyson said.
“I think with more of these large-scale studies about the pathophysiology of PCOS and how it may look in different people and the different outcomes, we may be able to tailor our treatments even further,” Dr. Tyson said. She emphasized the importance of identifying the condition early, particularly in adolescents, even if it’s identifying young people at risk for the condition rather than actually having it yet.
Early identification “gives us this chance to do a lot of preventative care and motivate older teens to have a great lifestyle, work on their diet and exercise, and manage cardiovascular” risk factors, Dr. Tyson said.
“What we do know and recognize is that there’s so many spokes to this PCOS wheel that there really should be a multidisciplinary approach to care,” Dr. Tyson said. “When I think about who would be the real doctors for patients with PCOS, these would be gynecologists, endocrinologists, dermatologists, nutritionists, psychologists, sleep specialists, and primary care at a minimum.”
Dr. Pal worries that the label of PCOS leaves it in the laps of ob.gyns. whereas, “if it was called something else, everybody would be involved in being vigilant and managing those patients.” She frequently reiterated that the label of PCOS is less important than ensuring clinicians treat the symptoms that most bother the patient.
And even if kisspeptin does play a causal role in PCOS for some patients, it’s only a subset of individuals with PCOS who would benefit from therapies developed to target it. Given the complexity of the syndrome and its many manifestations, a “galaxy of pathways” are involved in different potential subtypes of the condition. “You can’t treat PCOS as one entity,” Dr. Pal said.
Still, Dr. Hudson is optimistic that the research into potential neuroendocrine contributions to PCOS will yield therapies that go beyond just managing symptoms.
“There aren’t a lot of treatments available yet, but there may be some on the horizon,” Dr. Hudson said. “We’re still in this very primitive stage in terms of therapeutics, where we’re only addressing specific symptoms, and we haven’t been able to really address the underlying cause because we haven’t understood it as well and because we don’t have therapies that can target it,” Dr. Hudson said. “But once there are therapies developed that will target some of these central mechanisms, I think it will change completely the approach to treating PCOS for patients.”
This story was updated on Sept. 6, 2022.
Polycystic ovary syndrome (PCOS) affects an estimated 8%-13% of women, and yet “it has been quite a black box for many years,” as Margo Hudson, MD, an assistant professor of endocrinology, diabetes, and hypertension at Harvard Medical School, Boston, puts it. That black box encompasses not only uncertainty about the etiology and pathophysiology of the condition but even what constitutes a diagnosis.
Even the international guidelines on PCOS management endorsed by the American Society for Reproductive Medicine – a document developed over 15 months with the input of 37 medical organizations covering 71 countries – notes that PCOS diagnosis is “controversial and assessment and management are inconsistent.” The result, the guidelines note, is that “the needs of women with PCOS are not being adequately met.”
One of the earliest diagnostic criteria, defined in 1990 by the National Institutes of Health, required only hyperandrogenism and irregular menstruation. Then the 2003 Rotterdam Criteria added presence of polycystic ovaries on ultrasound as a third criterion. Then the Androgen Excess Society determined that PCOS required presence of hyperandrogenism with either polycystic ovaries or oligo/amenorrhea anovulation. Yet the Endocrine Society notes that excess androgen levels are seen in 60%-80% of those with PCOS, suggesting it’s not an essential requirement for diagnosis, leaving most to diagnose it in people who have two of the three key criteria. The only real agreement on diagnosis is the need to eliminate other potential diagnoses first, making PCOS always a diagnosis of exclusion.
Further, though PCOS is known as the leading cause of infertility in women, it is more than a reproductive condition, with metabolic and psychological features as well. Then there is the range of comorbidities, none of which occur in all patients with PCOS but all of which occur in a majority and which are themselves interrelated. Insulin resistance is a common feature, occurring in 50%-70% of people with PCOS. Accordingly, metabolic syndrome occurs in at least a third of people with PCOS and type 2 diabetes prevalence is higher in those with PCOS as well.
Obesity occurs in an estimated 80% of women with PCOS in the United States, though it affects only about 50% of women with PCOS outside the United States, and those with PCOS have an increased risk of hypertension. Mood disorders, particularly anxiety and depression but also, to a lesser extent, bipolar disorder and obsessive-compulsive disorder, are more likely in people with PCOS. And given that these comorbidities are all cardiovascular risk factors, it’s unsurprising that recent studies are finding those with PCOS to be at greater risk for cardiometabolic disease and major cardiovascular events.
“The reality is that PCOS is a heterogenous entity. It’s not one thing – it’s a syndrome,” Lubna Pal, MBBS, a professor of ob.gyn. and director of the PCOS Program at Yale University, New Haven, Conn., said in an interview. A whole host of factors are likely playing a role in the causes of PCOS, and those factors interact differently within different people. “We’re looking at things like lipid metabolism, fetal origins, the gut microbiome, genetics, epigenetics, and then dietary and environmental factors,” Nichole Tyson, MD, division chief of pediatric and adolescent gynecology and a clinical associate professor at Stanford (Calif.) Medicine Children’s Health, said in an interview. And most studies have identified associations that may or may not be causal. Take, for example, endocrine disruptors. BPA levels have been shown to be higher in women with PCOS than women without, but that correlation may or may not be related to the etiology of the condition.
The hypothalamic-pituitary-gonadal axis
In trying to understand the pathophysiology of the condition, much of the latest research has zeroed in on potential mechanisms in the hypothalamic-pituitary-gonadal axis. “A consistent feature of PCOS is disordered gonadotropin secretion with elevated mean LH [luteinizing hormone], low or low normal FSH [follicle-stimulating hormone], and a persistently rapid frequency of GnRH [gonadotropin-releasing hormone] pulse secretion,” wrote authors of a scientific statement on aspects of PCOS.
“I think the balance is heading more to central neurologic control of the reproductive system and that disturbances there impact the GnRH cells in the hypothalamus, which then go on to give us the findings that we can measure peripherally with the LH-FSH ratio,” Dr. Hudson said in an interview.
The increased LH levels are thought to be a major driver of increased androgen levels. Current thinking suggests that the primary driver of increased LH is GnRH pulsatility, supported not only by human studies but by animal models as well. This leads to the question of what drives GnRH dysregulation. One hypothesis posits that GABA neurons play a role here, given findings that GABA levels in cerebrospinal fluid were higher in women with PCOS than those with normal ovulation.
But the culprit garnering the most attention is kisspeptin, a protein encoded by the KISS1 gene that stimulates GnRH neurons and has been linked to regulation of LH and FSH secretion. Kisspeptin, along with neurokinin B and dynorphin, is part of the triumvirate that comprises KNDy neurons, also recently implicated in menopausal vasomotor symptoms. Multiple systematic reviewsand meta-analyses have found a correlation between higher kisspeptin levels in the blood and higher circulating LH levels, regardless of body mass index. While kisspeptin is expressed in several tissues, including liver, pancreas, gonad, and adipose, it’s neural kisspeptin signaling that appears most likely to play a role in activating GnRH hormones and disrupting normal function of the hypothalamic-pituitary-gonadal axis.
But as noted, in at least one systematic review of kisspeptin and PCOS, “findings from animal studies suggest that kisspeptin levels are not increased in all subtypes of PCOS.” And another review found “altered” levels of kisspeptin levels in non-PCOS patients who had obesity, potentially raising questions about any associations between kisspeptin and obesity or insulin resistance.
Remaining chicken-and-egg questions
A hallmark of PCOS has long been, and continues to be, the string of chicken-or-egg questions that plague understanding of it. One of these is how depression and anxiety fit into the etiology of PCOS. Exploring the role of specific neurons that may overstimulate GnRH pulsatility may hold clues to a common underlying mechanism for the involvement of depression and anxiety in patients with PCOS, Dr. Hudson speculated. While previous assumptions often attributed depression and anxiety in PCOS to the symptoms – such as thin scalp hair and increased facial hair, excess weight, acne, and irregular periods – Dr. Hudson pointed out that women can address many of these symptoms with laser hair removal, weight loss, acne treatment, and similar interventions, yet they still have a lot of underlying mental health issues.
It’s also unclear whether metabolic factors so common with PCOS, particularly insulin resistance and obesity, are a result of the condition or are contributors to it. Is insulin resistance contributing to dysregulation in the neurons that interferes with normal functioning of the hypothalamic-pituitary-adrenal axis? Is abnormal functioning along this axis contributing to insulin resistance? Or neither? Or both? Or does it depend? The authors of one paper wrote that “insulin may play both direct and indirect roles in the pathogenesis of androgen excess in PCOS,” since insulin can “stimulate ovarian androgen production” and “enhance ovarian growth and follicular cyst formation in rats.”
Dr. Pal noted that “obesity itself can evolve into a PCOS-like picture,” raising questions about whether obesity or insulin resistance might be part of the causal pathway to PCOS, or whether either can trigger its development in those genetically predisposed.
“Obesity does appear to exacerbate many aspects of the PCOS phenotype, particularly those risk factors related to metabolic syndrome,” wrote the authors of a scientific statement on aspects of PCOS, but they add that “it is currently debated whether obesity per se can cause PCOS.” While massive weight loss in those with PCOS and obesity has improved multiple reproductive and metabolic issues, it hasn’t resolved all of them, they write.
Dr. Hudson said she expects there’s “some degree of appetite dysregulation and metabolic dysregulation” that contributes, but then there are other women who don’t have much of an appetite or overeat and still struggle with their weight. Evidence has also found insulin resistance in women of normal weight with PCOS. “There may be some kind of metabolic dysregulation that they have at some level, and others are clearly bothered by overeating,” Dr. Hudson said.
Similarly, it’s not clear whether the recent discovery of increased cardiovascular risks in people with PCOS is a result of the comorbidities so common with PCOS, such as obesity, or whether an underlying mechanism links the cardiovascular risk and the dysregulation of hormones. Dr. Pal would argue that, again, it’s probably both, depending on the patient.
Then there is the key feature of hyperandrogenemia. “An outstanding debate is whether the elevated androgens in PCOS women are merely a downstream endocrine response to hyperactive GnRH and LH secretion driving the ovary, or do the elevated androgens themselves act in the brain (or pituitary) during development and/or adulthood to sculpt and maintain the hypersecretion of GnRH and LH?” wrote Eulalia A. Coutinho, PhD, and Alexander S. Kauffman, PhD, in a 2019 review of the brain’s role in PCOS.
These problems may be bidirectional or part of various feedback loops. Sleep apnea is more common in people with PCOS, Dr. Tyson noted, but sleep apnea is also linked to cardiovascular, metabolic, and depression risks, and depression can play a role in obesity, which increases the risk of obstructive sleep apnea. “So you’re in this vicious cycle,” Dr. Tyson said. That’s why she also believes it’s important to change the dialogue and perspective on PCOS, to reduce the stigma attached to it, and work with patients to empower them in treating its symptoms and reducing their risk of comorbidities.
Recent and upcoming changes in treatment
Current treatment of PCOS already changes according to the symptoms posing the greatest problems at each stage of a person’s life, Dr. Hudson said. Younger women tend to be more bothered about the cosmetic effects of PCOS, including hair growth patterns and acne, but as they grow out of adolescence and into their 20s and 30s, infertility becomes a bigger concern for many. Then, as they start approaching menopause, metabolic and cardiovascular issues take the lead, with more of a focus on lipids, diabetes risk, and heart health.
In some ways, management of PCOS hasn’t changed much in the past several decades, except in an increased awareness of the metabolic and cardiovascular risks, which has led to more frequent screening to catch potential conditions earlier in life. What has changed, however, is improvements in the treatments used for symptoms, such as expanded bariatric surgery options and GLP-1 agonists for treating obesity. Other examples include better options for menstrual management, such as new progesterone IUDs, and optimized fertility treatments, Dr. Tyson said.
“I think with more of these large-scale studies about the pathophysiology of PCOS and how it may look in different people and the different outcomes, we may be able to tailor our treatments even further,” Dr. Tyson said. She emphasized the importance of identifying the condition early, particularly in adolescents, even if it’s identifying young people at risk for the condition rather than actually having it yet.
Early identification “gives us this chance to do a lot of preventative care and motivate older teens to have a great lifestyle, work on their diet and exercise, and manage cardiovascular” risk factors, Dr. Tyson said.
“What we do know and recognize is that there’s so many spokes to this PCOS wheel that there really should be a multidisciplinary approach to care,” Dr. Tyson said. “When I think about who would be the real doctors for patients with PCOS, these would be gynecologists, endocrinologists, dermatologists, nutritionists, psychologists, sleep specialists, and primary care at a minimum.”
Dr. Pal worries that the label of PCOS leaves it in the laps of ob.gyns. whereas, “if it was called something else, everybody would be involved in being vigilant and managing those patients.” She frequently reiterated that the label of PCOS is less important than ensuring clinicians treat the symptoms that most bother the patient.
And even if kisspeptin does play a causal role in PCOS for some patients, it’s only a subset of individuals with PCOS who would benefit from therapies developed to target it. Given the complexity of the syndrome and its many manifestations, a “galaxy of pathways” are involved in different potential subtypes of the condition. “You can’t treat PCOS as one entity,” Dr. Pal said.
Still, Dr. Hudson is optimistic that the research into potential neuroendocrine contributions to PCOS will yield therapies that go beyond just managing symptoms.
“There aren’t a lot of treatments available yet, but there may be some on the horizon,” Dr. Hudson said. “We’re still in this very primitive stage in terms of therapeutics, where we’re only addressing specific symptoms, and we haven’t been able to really address the underlying cause because we haven’t understood it as well and because we don’t have therapies that can target it,” Dr. Hudson said. “But once there are therapies developed that will target some of these central mechanisms, I think it will change completely the approach to treating PCOS for patients.”
This story was updated on Sept. 6, 2022.
Polycystic ovary syndrome (PCOS) affects an estimated 8%-13% of women, and yet “it has been quite a black box for many years,” as Margo Hudson, MD, an assistant professor of endocrinology, diabetes, and hypertension at Harvard Medical School, Boston, puts it. That black box encompasses not only uncertainty about the etiology and pathophysiology of the condition but even what constitutes a diagnosis.
Even the international guidelines on PCOS management endorsed by the American Society for Reproductive Medicine – a document developed over 15 months with the input of 37 medical organizations covering 71 countries – notes that PCOS diagnosis is “controversial and assessment and management are inconsistent.” The result, the guidelines note, is that “the needs of women with PCOS are not being adequately met.”
One of the earliest diagnostic criteria, defined in 1990 by the National Institutes of Health, required only hyperandrogenism and irregular menstruation. Then the 2003 Rotterdam Criteria added presence of polycystic ovaries on ultrasound as a third criterion. Then the Androgen Excess Society determined that PCOS required presence of hyperandrogenism with either polycystic ovaries or oligo/amenorrhea anovulation. Yet the Endocrine Society notes that excess androgen levels are seen in 60%-80% of those with PCOS, suggesting it’s not an essential requirement for diagnosis, leaving most to diagnose it in people who have two of the three key criteria. The only real agreement on diagnosis is the need to eliminate other potential diagnoses first, making PCOS always a diagnosis of exclusion.
Further, though PCOS is known as the leading cause of infertility in women, it is more than a reproductive condition, with metabolic and psychological features as well. Then there is the range of comorbidities, none of which occur in all patients with PCOS but all of which occur in a majority and which are themselves interrelated. Insulin resistance is a common feature, occurring in 50%-70% of people with PCOS. Accordingly, metabolic syndrome occurs in at least a third of people with PCOS and type 2 diabetes prevalence is higher in those with PCOS as well.
Obesity occurs in an estimated 80% of women with PCOS in the United States, though it affects only about 50% of women with PCOS outside the United States, and those with PCOS have an increased risk of hypertension. Mood disorders, particularly anxiety and depression but also, to a lesser extent, bipolar disorder and obsessive-compulsive disorder, are more likely in people with PCOS. And given that these comorbidities are all cardiovascular risk factors, it’s unsurprising that recent studies are finding those with PCOS to be at greater risk for cardiometabolic disease and major cardiovascular events.
“The reality is that PCOS is a heterogenous entity. It’s not one thing – it’s a syndrome,” Lubna Pal, MBBS, a professor of ob.gyn. and director of the PCOS Program at Yale University, New Haven, Conn., said in an interview. A whole host of factors are likely playing a role in the causes of PCOS, and those factors interact differently within different people. “We’re looking at things like lipid metabolism, fetal origins, the gut microbiome, genetics, epigenetics, and then dietary and environmental factors,” Nichole Tyson, MD, division chief of pediatric and adolescent gynecology and a clinical associate professor at Stanford (Calif.) Medicine Children’s Health, said in an interview. And most studies have identified associations that may or may not be causal. Take, for example, endocrine disruptors. BPA levels have been shown to be higher in women with PCOS than women without, but that correlation may or may not be related to the etiology of the condition.
The hypothalamic-pituitary-gonadal axis
In trying to understand the pathophysiology of the condition, much of the latest research has zeroed in on potential mechanisms in the hypothalamic-pituitary-gonadal axis. “A consistent feature of PCOS is disordered gonadotropin secretion with elevated mean LH [luteinizing hormone], low or low normal FSH [follicle-stimulating hormone], and a persistently rapid frequency of GnRH [gonadotropin-releasing hormone] pulse secretion,” wrote authors of a scientific statement on aspects of PCOS.
“I think the balance is heading more to central neurologic control of the reproductive system and that disturbances there impact the GnRH cells in the hypothalamus, which then go on to give us the findings that we can measure peripherally with the LH-FSH ratio,” Dr. Hudson said in an interview.
The increased LH levels are thought to be a major driver of increased androgen levels. Current thinking suggests that the primary driver of increased LH is GnRH pulsatility, supported not only by human studies but by animal models as well. This leads to the question of what drives GnRH dysregulation. One hypothesis posits that GABA neurons play a role here, given findings that GABA levels in cerebrospinal fluid were higher in women with PCOS than those with normal ovulation.
But the culprit garnering the most attention is kisspeptin, a protein encoded by the KISS1 gene that stimulates GnRH neurons and has been linked to regulation of LH and FSH secretion. Kisspeptin, along with neurokinin B and dynorphin, is part of the triumvirate that comprises KNDy neurons, also recently implicated in menopausal vasomotor symptoms. Multiple systematic reviewsand meta-analyses have found a correlation between higher kisspeptin levels in the blood and higher circulating LH levels, regardless of body mass index. While kisspeptin is expressed in several tissues, including liver, pancreas, gonad, and adipose, it’s neural kisspeptin signaling that appears most likely to play a role in activating GnRH hormones and disrupting normal function of the hypothalamic-pituitary-gonadal axis.
But as noted, in at least one systematic review of kisspeptin and PCOS, “findings from animal studies suggest that kisspeptin levels are not increased in all subtypes of PCOS.” And another review found “altered” levels of kisspeptin levels in non-PCOS patients who had obesity, potentially raising questions about any associations between kisspeptin and obesity or insulin resistance.
Remaining chicken-and-egg questions
A hallmark of PCOS has long been, and continues to be, the string of chicken-or-egg questions that plague understanding of it. One of these is how depression and anxiety fit into the etiology of PCOS. Exploring the role of specific neurons that may overstimulate GnRH pulsatility may hold clues to a common underlying mechanism for the involvement of depression and anxiety in patients with PCOS, Dr. Hudson speculated. While previous assumptions often attributed depression and anxiety in PCOS to the symptoms – such as thin scalp hair and increased facial hair, excess weight, acne, and irregular periods – Dr. Hudson pointed out that women can address many of these symptoms with laser hair removal, weight loss, acne treatment, and similar interventions, yet they still have a lot of underlying mental health issues.
It’s also unclear whether metabolic factors so common with PCOS, particularly insulin resistance and obesity, are a result of the condition or are contributors to it. Is insulin resistance contributing to dysregulation in the neurons that interferes with normal functioning of the hypothalamic-pituitary-adrenal axis? Is abnormal functioning along this axis contributing to insulin resistance? Or neither? Or both? Or does it depend? The authors of one paper wrote that “insulin may play both direct and indirect roles in the pathogenesis of androgen excess in PCOS,” since insulin can “stimulate ovarian androgen production” and “enhance ovarian growth and follicular cyst formation in rats.”
Dr. Pal noted that “obesity itself can evolve into a PCOS-like picture,” raising questions about whether obesity or insulin resistance might be part of the causal pathway to PCOS, or whether either can trigger its development in those genetically predisposed.
“Obesity does appear to exacerbate many aspects of the PCOS phenotype, particularly those risk factors related to metabolic syndrome,” wrote the authors of a scientific statement on aspects of PCOS, but they add that “it is currently debated whether obesity per se can cause PCOS.” While massive weight loss in those with PCOS and obesity has improved multiple reproductive and metabolic issues, it hasn’t resolved all of them, they write.
Dr. Hudson said she expects there’s “some degree of appetite dysregulation and metabolic dysregulation” that contributes, but then there are other women who don’t have much of an appetite or overeat and still struggle with their weight. Evidence has also found insulin resistance in women of normal weight with PCOS. “There may be some kind of metabolic dysregulation that they have at some level, and others are clearly bothered by overeating,” Dr. Hudson said.
Similarly, it’s not clear whether the recent discovery of increased cardiovascular risks in people with PCOS is a result of the comorbidities so common with PCOS, such as obesity, or whether an underlying mechanism links the cardiovascular risk and the dysregulation of hormones. Dr. Pal would argue that, again, it’s probably both, depending on the patient.
Then there is the key feature of hyperandrogenemia. “An outstanding debate is whether the elevated androgens in PCOS women are merely a downstream endocrine response to hyperactive GnRH and LH secretion driving the ovary, or do the elevated androgens themselves act in the brain (or pituitary) during development and/or adulthood to sculpt and maintain the hypersecretion of GnRH and LH?” wrote Eulalia A. Coutinho, PhD, and Alexander S. Kauffman, PhD, in a 2019 review of the brain’s role in PCOS.
These problems may be bidirectional or part of various feedback loops. Sleep apnea is more common in people with PCOS, Dr. Tyson noted, but sleep apnea is also linked to cardiovascular, metabolic, and depression risks, and depression can play a role in obesity, which increases the risk of obstructive sleep apnea. “So you’re in this vicious cycle,” Dr. Tyson said. That’s why she also believes it’s important to change the dialogue and perspective on PCOS, to reduce the stigma attached to it, and work with patients to empower them in treating its symptoms and reducing their risk of comorbidities.
Recent and upcoming changes in treatment
Current treatment of PCOS already changes according to the symptoms posing the greatest problems at each stage of a person’s life, Dr. Hudson said. Younger women tend to be more bothered about the cosmetic effects of PCOS, including hair growth patterns and acne, but as they grow out of adolescence and into their 20s and 30s, infertility becomes a bigger concern for many. Then, as they start approaching menopause, metabolic and cardiovascular issues take the lead, with more of a focus on lipids, diabetes risk, and heart health.
In some ways, management of PCOS hasn’t changed much in the past several decades, except in an increased awareness of the metabolic and cardiovascular risks, which has led to more frequent screening to catch potential conditions earlier in life. What has changed, however, is improvements in the treatments used for symptoms, such as expanded bariatric surgery options and GLP-1 agonists for treating obesity. Other examples include better options for menstrual management, such as new progesterone IUDs, and optimized fertility treatments, Dr. Tyson said.
“I think with more of these large-scale studies about the pathophysiology of PCOS and how it may look in different people and the different outcomes, we may be able to tailor our treatments even further,” Dr. Tyson said. She emphasized the importance of identifying the condition early, particularly in adolescents, even if it’s identifying young people at risk for the condition rather than actually having it yet.
Early identification “gives us this chance to do a lot of preventative care and motivate older teens to have a great lifestyle, work on their diet and exercise, and manage cardiovascular” risk factors, Dr. Tyson said.
“What we do know and recognize is that there’s so many spokes to this PCOS wheel that there really should be a multidisciplinary approach to care,” Dr. Tyson said. “When I think about who would be the real doctors for patients with PCOS, these would be gynecologists, endocrinologists, dermatologists, nutritionists, psychologists, sleep specialists, and primary care at a minimum.”
Dr. Pal worries that the label of PCOS leaves it in the laps of ob.gyns. whereas, “if it was called something else, everybody would be involved in being vigilant and managing those patients.” She frequently reiterated that the label of PCOS is less important than ensuring clinicians treat the symptoms that most bother the patient.
And even if kisspeptin does play a causal role in PCOS for some patients, it’s only a subset of individuals with PCOS who would benefit from therapies developed to target it. Given the complexity of the syndrome and its many manifestations, a “galaxy of pathways” are involved in different potential subtypes of the condition. “You can’t treat PCOS as one entity,” Dr. Pal said.
Still, Dr. Hudson is optimistic that the research into potential neuroendocrine contributions to PCOS will yield therapies that go beyond just managing symptoms.
“There aren’t a lot of treatments available yet, but there may be some on the horizon,” Dr. Hudson said. “We’re still in this very primitive stage in terms of therapeutics, where we’re only addressing specific symptoms, and we haven’t been able to really address the underlying cause because we haven’t understood it as well and because we don’t have therapies that can target it,” Dr. Hudson said. “But once there are therapies developed that will target some of these central mechanisms, I think it will change completely the approach to treating PCOS for patients.”
This story was updated on Sept. 6, 2022.
Hyperthyroidism rebound in pregnancy boosts adverse outcomes
Discontinuing antithyroid drugs during early pregnancy is linked to a possible rebound of hyperthyroidism and a high risk of adverse pregnancy outcomes, new research shows.
“Our study provides preliminary evidence that the risk of rebound increases in women with subnormal thyroid-stimulating hormone (TSH) and/or positive thyrotropin receptor antibody (TRAb) who stop antithyroid drugs in early pregnancy,” first author Xin Hou told this news organization.
“When discussing the pros and cons of antithyroid drug withdrawal early in pregnancy [clinicians] should consider the level of TSH and TRAb in early pregnancy,” said Hou, of the department of endocrinology and metabolism, Institute of Endocrinology, The First Affiliated Hospital of China Medical University, Shenyang.
Suvi Turunen, MD, of the University of Oulu (Finland), who has also conducted research on the issue, said the study adds important insights.
“I find this study very interesting,” Dr. Turunen said in an interview. “It is well known that medical treatment of hyperthyroidism outweighs the potential harms of antithyroid treatment.”
The new findings add to the evidence, she added. “I think that withdrawal of antithyroid drugs should be carefully considered, especially with autoantibody-positive patients,” Dr. Turunen said.
Hyperthyroidism a risk in pregnancy – with or without treatment
The potential risks of hyperthyroidism in pregnancy are well established and can range from preeclampsia to premature birth or miscarriage.
However, antithyroid drugs, including methimazole and propylthiouracil, carry their own risks. In crossing the placental barrier, the drugs can increase the risk of birth defects, particularly during 6-10 weeks of gestation, yet their discontinuation is linked to as much as a 50%-60% risk of relapse, the authors explain.
Because of the risks, the American Thyroid Association recommends that “women with a stable euthyroid state on 5-10 mg methimazole per day achieved within a few months, and a falling TRAb level, are likely candidates to withdraw from antithyroid drug therapy in early pregnancy,” the authors noted.
However, as the recommendations for women who are already pregnant are largely based on evidence from nonpregnant patients, Hou and colleagues sought to evaluate withdrawal among women who were pregnant.
For the study, published in Thyroid, they enrolled 63 women who were pregnant and part of an outpatient service of the department of endocrinology and metabolism at The First Affiliated Hospital of China Medical University, between September 2014 and March 2017, who had well-controlled hyperthyroidism in early pregnancy and discontinued the drugs.
The women were an average age of 27 years, and 28 were multigravida. Twenty-two had a history of miscarriage.
A follow-up of the patients until the end of their pregnancy showed that, overall, 20 (31.7%) had a rebound of hyperthyroidism during their pregnancy after withdrawing from the drugs.
Key factors associated with the highest risk of a rebound after discontinuation included having subnormal TSH levels (TSH < 0.35 mIU/L; odds ratio, 5.12; P = .03) or having positive TRAb (TRAb > 1.75 IU/L; OR, 3.79; P = .02) at the time of medication withdrawal, compared with those with either normal TSH levels or negative TRAb.
The combination of both subnormal TSH and positive TRAb at the time of antithyroid medication withdrawal further boosted the risk of hyperthyroidism rebound (83.3%, 5 of 6), compared with those who had both normal TSH and negative TRAb (13%, 3 of 23; OR, 33.33; P = .003).
Adverse pregnancy outcomes increased
Importantly, among the 20 patients who had a rebound, 11 (55%) had adverse pregnancy outcomes, including miscarriage, premature birth, induced labor, gestational hypertension, and gestational diabetes, compared with only 4 (9.3%) of the 43 who had no rebound (OR, 11.92; P = .0002).
Neonatal abnormalities were also higher among those experiencing a rebound (20% vs. 4.7%), however, the authors noted that “larger prospective studies are required to conclude whether antithyroid drug withdrawal affects fetal outcome.”
In the rebound group, the mean duration of antithyroid medication use was 24.7 months versus 35.1 months in the nonrebound group, however, the difference was not statistically significant (P = .07). And 40% of the rebound group had a history of miscarriage versus 32.6% in the non-rebound group, but was also not significantly different (P = .56).
The authors noted that half of those in the rebound group developed hyperthyroidism more than 4 weeks after their withdrawal from antithyroid medications, “which seemed to have circumvented the most sensitive period of teratogenesis between 6 and 10 weeks of pregnancy.”
Hou added that restarting antithyroid medication did not increase the risk of adverse outcomes for offspring.
“A low dose of antithyroid medications may be a good choice for women with subnormal TSH and/or positive TRAb in early pregnancy,” Hou concluded. “Because of the small size of our study, a larger prospective study is needed to overcome the potential selection bias and to verify the conclusions.”
Findings consistent with Finnish study
In her own recent study, which included 2,144 women in Finland who experienced hyperthyroidism during pregnancy, Dr. Turunen and colleagues found that having hyperthyroidism, with or without antithyroid drug treatment, was associated with an increased odds of pregnancy and/or prenatal complications, compared with those without thyroid disease.
“In our study, we observed an increased risk of adverse pregnancy outcomes also in mothers with previous diagnosis and/or treatment of hyperthyroidism, not only with overt hyperthyroidism treated with antithyroid drugs,” she told this news organization.
“I think that especially those patients with positive antibodies [TRAbs] are at risk even if they are euthyroid,” she noted. “Withdrawal of antithyroid drugs in these patients is a risk.”
“Probably continuing antithyroid treatment with low dose is a better option,” she said.
The authors and Dr. Turunen reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Discontinuing antithyroid drugs during early pregnancy is linked to a possible rebound of hyperthyroidism and a high risk of adverse pregnancy outcomes, new research shows.
“Our study provides preliminary evidence that the risk of rebound increases in women with subnormal thyroid-stimulating hormone (TSH) and/or positive thyrotropin receptor antibody (TRAb) who stop antithyroid drugs in early pregnancy,” first author Xin Hou told this news organization.
“When discussing the pros and cons of antithyroid drug withdrawal early in pregnancy [clinicians] should consider the level of TSH and TRAb in early pregnancy,” said Hou, of the department of endocrinology and metabolism, Institute of Endocrinology, The First Affiliated Hospital of China Medical University, Shenyang.
Suvi Turunen, MD, of the University of Oulu (Finland), who has also conducted research on the issue, said the study adds important insights.
“I find this study very interesting,” Dr. Turunen said in an interview. “It is well known that medical treatment of hyperthyroidism outweighs the potential harms of antithyroid treatment.”
The new findings add to the evidence, she added. “I think that withdrawal of antithyroid drugs should be carefully considered, especially with autoantibody-positive patients,” Dr. Turunen said.
Hyperthyroidism a risk in pregnancy – with or without treatment
The potential risks of hyperthyroidism in pregnancy are well established and can range from preeclampsia to premature birth or miscarriage.
However, antithyroid drugs, including methimazole and propylthiouracil, carry their own risks. In crossing the placental barrier, the drugs can increase the risk of birth defects, particularly during 6-10 weeks of gestation, yet their discontinuation is linked to as much as a 50%-60% risk of relapse, the authors explain.
Because of the risks, the American Thyroid Association recommends that “women with a stable euthyroid state on 5-10 mg methimazole per day achieved within a few months, and a falling TRAb level, are likely candidates to withdraw from antithyroid drug therapy in early pregnancy,” the authors noted.
However, as the recommendations for women who are already pregnant are largely based on evidence from nonpregnant patients, Hou and colleagues sought to evaluate withdrawal among women who were pregnant.
For the study, published in Thyroid, they enrolled 63 women who were pregnant and part of an outpatient service of the department of endocrinology and metabolism at The First Affiliated Hospital of China Medical University, between September 2014 and March 2017, who had well-controlled hyperthyroidism in early pregnancy and discontinued the drugs.
The women were an average age of 27 years, and 28 were multigravida. Twenty-two had a history of miscarriage.
A follow-up of the patients until the end of their pregnancy showed that, overall, 20 (31.7%) had a rebound of hyperthyroidism during their pregnancy after withdrawing from the drugs.
Key factors associated with the highest risk of a rebound after discontinuation included having subnormal TSH levels (TSH < 0.35 mIU/L; odds ratio, 5.12; P = .03) or having positive TRAb (TRAb > 1.75 IU/L; OR, 3.79; P = .02) at the time of medication withdrawal, compared with those with either normal TSH levels or negative TRAb.
The combination of both subnormal TSH and positive TRAb at the time of antithyroid medication withdrawal further boosted the risk of hyperthyroidism rebound (83.3%, 5 of 6), compared with those who had both normal TSH and negative TRAb (13%, 3 of 23; OR, 33.33; P = .003).
Adverse pregnancy outcomes increased
Importantly, among the 20 patients who had a rebound, 11 (55%) had adverse pregnancy outcomes, including miscarriage, premature birth, induced labor, gestational hypertension, and gestational diabetes, compared with only 4 (9.3%) of the 43 who had no rebound (OR, 11.92; P = .0002).
Neonatal abnormalities were also higher among those experiencing a rebound (20% vs. 4.7%), however, the authors noted that “larger prospective studies are required to conclude whether antithyroid drug withdrawal affects fetal outcome.”
In the rebound group, the mean duration of antithyroid medication use was 24.7 months versus 35.1 months in the nonrebound group, however, the difference was not statistically significant (P = .07). And 40% of the rebound group had a history of miscarriage versus 32.6% in the non-rebound group, but was also not significantly different (P = .56).
The authors noted that half of those in the rebound group developed hyperthyroidism more than 4 weeks after their withdrawal from antithyroid medications, “which seemed to have circumvented the most sensitive period of teratogenesis between 6 and 10 weeks of pregnancy.”
Hou added that restarting antithyroid medication did not increase the risk of adverse outcomes for offspring.
“A low dose of antithyroid medications may be a good choice for women with subnormal TSH and/or positive TRAb in early pregnancy,” Hou concluded. “Because of the small size of our study, a larger prospective study is needed to overcome the potential selection bias and to verify the conclusions.”
Findings consistent with Finnish study
In her own recent study, which included 2,144 women in Finland who experienced hyperthyroidism during pregnancy, Dr. Turunen and colleagues found that having hyperthyroidism, with or without antithyroid drug treatment, was associated with an increased odds of pregnancy and/or prenatal complications, compared with those without thyroid disease.
“In our study, we observed an increased risk of adverse pregnancy outcomes also in mothers with previous diagnosis and/or treatment of hyperthyroidism, not only with overt hyperthyroidism treated with antithyroid drugs,” she told this news organization.
“I think that especially those patients with positive antibodies [TRAbs] are at risk even if they are euthyroid,” she noted. “Withdrawal of antithyroid drugs in these patients is a risk.”
“Probably continuing antithyroid treatment with low dose is a better option,” she said.
The authors and Dr. Turunen reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Discontinuing antithyroid drugs during early pregnancy is linked to a possible rebound of hyperthyroidism and a high risk of adverse pregnancy outcomes, new research shows.
“Our study provides preliminary evidence that the risk of rebound increases in women with subnormal thyroid-stimulating hormone (TSH) and/or positive thyrotropin receptor antibody (TRAb) who stop antithyroid drugs in early pregnancy,” first author Xin Hou told this news organization.
“When discussing the pros and cons of antithyroid drug withdrawal early in pregnancy [clinicians] should consider the level of TSH and TRAb in early pregnancy,” said Hou, of the department of endocrinology and metabolism, Institute of Endocrinology, The First Affiliated Hospital of China Medical University, Shenyang.
Suvi Turunen, MD, of the University of Oulu (Finland), who has also conducted research on the issue, said the study adds important insights.
“I find this study very interesting,” Dr. Turunen said in an interview. “It is well known that medical treatment of hyperthyroidism outweighs the potential harms of antithyroid treatment.”
The new findings add to the evidence, she added. “I think that withdrawal of antithyroid drugs should be carefully considered, especially with autoantibody-positive patients,” Dr. Turunen said.
Hyperthyroidism a risk in pregnancy – with or without treatment
The potential risks of hyperthyroidism in pregnancy are well established and can range from preeclampsia to premature birth or miscarriage.
However, antithyroid drugs, including methimazole and propylthiouracil, carry their own risks. In crossing the placental barrier, the drugs can increase the risk of birth defects, particularly during 6-10 weeks of gestation, yet their discontinuation is linked to as much as a 50%-60% risk of relapse, the authors explain.
Because of the risks, the American Thyroid Association recommends that “women with a stable euthyroid state on 5-10 mg methimazole per day achieved within a few months, and a falling TRAb level, are likely candidates to withdraw from antithyroid drug therapy in early pregnancy,” the authors noted.
However, as the recommendations for women who are already pregnant are largely based on evidence from nonpregnant patients, Hou and colleagues sought to evaluate withdrawal among women who were pregnant.
For the study, published in Thyroid, they enrolled 63 women who were pregnant and part of an outpatient service of the department of endocrinology and metabolism at The First Affiliated Hospital of China Medical University, between September 2014 and March 2017, who had well-controlled hyperthyroidism in early pregnancy and discontinued the drugs.
The women were an average age of 27 years, and 28 were multigravida. Twenty-two had a history of miscarriage.
A follow-up of the patients until the end of their pregnancy showed that, overall, 20 (31.7%) had a rebound of hyperthyroidism during their pregnancy after withdrawing from the drugs.
Key factors associated with the highest risk of a rebound after discontinuation included having subnormal TSH levels (TSH < 0.35 mIU/L; odds ratio, 5.12; P = .03) or having positive TRAb (TRAb > 1.75 IU/L; OR, 3.79; P = .02) at the time of medication withdrawal, compared with those with either normal TSH levels or negative TRAb.
The combination of both subnormal TSH and positive TRAb at the time of antithyroid medication withdrawal further boosted the risk of hyperthyroidism rebound (83.3%, 5 of 6), compared with those who had both normal TSH and negative TRAb (13%, 3 of 23; OR, 33.33; P = .003).
Adverse pregnancy outcomes increased
Importantly, among the 20 patients who had a rebound, 11 (55%) had adverse pregnancy outcomes, including miscarriage, premature birth, induced labor, gestational hypertension, and gestational diabetes, compared with only 4 (9.3%) of the 43 who had no rebound (OR, 11.92; P = .0002).
Neonatal abnormalities were also higher among those experiencing a rebound (20% vs. 4.7%), however, the authors noted that “larger prospective studies are required to conclude whether antithyroid drug withdrawal affects fetal outcome.”
In the rebound group, the mean duration of antithyroid medication use was 24.7 months versus 35.1 months in the nonrebound group, however, the difference was not statistically significant (P = .07). And 40% of the rebound group had a history of miscarriage versus 32.6% in the non-rebound group, but was also not significantly different (P = .56).
The authors noted that half of those in the rebound group developed hyperthyroidism more than 4 weeks after their withdrawal from antithyroid medications, “which seemed to have circumvented the most sensitive period of teratogenesis between 6 and 10 weeks of pregnancy.”
Hou added that restarting antithyroid medication did not increase the risk of adverse outcomes for offspring.
“A low dose of antithyroid medications may be a good choice for women with subnormal TSH and/or positive TRAb in early pregnancy,” Hou concluded. “Because of the small size of our study, a larger prospective study is needed to overcome the potential selection bias and to verify the conclusions.”
Findings consistent with Finnish study
In her own recent study, which included 2,144 women in Finland who experienced hyperthyroidism during pregnancy, Dr. Turunen and colleagues found that having hyperthyroidism, with or without antithyroid drug treatment, was associated with an increased odds of pregnancy and/or prenatal complications, compared with those without thyroid disease.
“In our study, we observed an increased risk of adverse pregnancy outcomes also in mothers with previous diagnosis and/or treatment of hyperthyroidism, not only with overt hyperthyroidism treated with antithyroid drugs,” she told this news organization.
“I think that especially those patients with positive antibodies [TRAbs] are at risk even if they are euthyroid,” she noted. “Withdrawal of antithyroid drugs in these patients is a risk.”
“Probably continuing antithyroid treatment with low dose is a better option,” she said.
The authors and Dr. Turunen reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THYROID
Endometriosis and infertility – Combining a chronic physical and emotional pain
Pain is classified as chronic when it lasts or recurs for more than 3-6 months (“Classification of chronic pain” 2nd ed. Seattle: IASP Press, 1994). This universally accepted definition does not distinguish between physical and emotional pain. Categorically, pain is pain. Two prevalent chronic gynecologic diseases are closely related medically and emotionally. Forty percent to 50% of women with endometriosis have infertility; 30%-50% of women with infertility are found to have coexisting endometriosis. The approach to both is, typically, symptomatic treatment. In this month’s column, I examine the relationship between these ailments and how we can advise women on management.
Endometriosis is simply defined as the displacement of normal endometrial glands and stroma from their natural anatomical location to elsewhere in the body. With the recent identification of the disease in the spleen, endometriosis has been found in every organ system. Endometriosis is identified in 6%-10% of the general female population. The prevalence ranges from 2% to 11% among asymptomatic women and from 5% to 21% in women hospitalized for pelvic pain (Best Pract Res Clin Obstet Gynaecol. 2018;51:1-15). Compared with fertile women, infertile women are six to eight times more likely to have endometriosis (Fertil Steril. 2012;98:591-8).
Retrograde menstruation is the presumed theory for the origins of endometriosis, that is, the reflux of menstrual debris containing active endometrial cells through the fallopian tubes into the peritoneal cavity (Am J Obstet Gynecol. 1927;14:422-69). Because of the varied etiologies of the most common symptoms of endometriosis, dysmenorrhea, dyspareunia, dyschezia, and infertility, women visit, on average, seven physicians before being diagnosed (Fertil Steril. 2011;96:366). The delay in promptly identifying endometriosis is further impaired by the lack of specific biomarkers, awareness, and inadequate evaluation (N Engl J Med. 2020;382:1244-56).
The 2008 U.S. health care costs for endometriosis were approximately $4,000 per affected woman, analogous to the costs for other chronic conditions such as type 2 diabetes, Crohn’s disease, and rheumatoid arthritis (Hum Reprod. 2012;27:1292-9). The management of symptoms further increases the financial burden because of the effect of the disease on physical, mental, sexual, and social well-being, as well as productivity (Health Qual Life Outcomes. 2019;17:123).
We have known the paradoxical relationship between the stage of endometriosis and symptoms: Women with low-stage disease may present with severe pain and/or infertility but those with advanced-stage disease may be asymptomatic. Endometriotic cells and tissue elicit a localized immune and inflammatory response with the production of cytokines, chemokines, and prostaglandins. Given the usual intra-abdominal location and the small size of implants, endometriosis requires a surgical diagnosis, ideally with histopathology for confirmation. However, imaging – transvaginal ultrasound or MRI – has more than 90% sensitivity and specificity for identifying endometriomas (Cochrane Database Syst Rev. 2016;2[2]:CD009591).
The effect of endometriosis on fertility, particularly in women with minimal to mild stages, is not clear, and many studies have been retrospective. Tubal factor infertility can be a result of endometriosis. Per the 2020 Cochrane Database Systemic Reviews (2020 Oct;2020[10]:CD011031), “Compared to diagnostic laparoscopy only, it is uncertain whether laparoscopic surgery reduces overall pain associated with minimal to severe endometriosis; no data were reported on live birth. There is moderate-quality evidence that laparoscopic surgery increases viable intrauterine pregnancy rates confirmed by ultrasound compared to diagnostic laparoscopy only.” In women undergoing IVF, more advanced stages of endometriosis have reduced pregnancy outcomes as shown in recent meta-analyses (Obstet Gynecol. 2015;125:79-88).
The revised ASRM (rASRM) surgical staging classification of endometriosis has been widely used to describe the degree, although it poorly correlates with fertility potential (Fertil Steril. 2012;98:591-8). Women diagnosed with endometriosis may benefit from the Endometriosis Fertility Index (EFI), published in 2010 as a useful scoring system to predict postoperative non-IVF pregnancy rates (both by natural means and intrauterine insemination) based on patient characteristics, rASRM staging and “least function” score of the adnexa (Fertil Steril. 2010;94:1609-15).
Compared with diagnostic laparoscopy only, it is uncertain whether laparoscopic surgery reduces overall pain associated with minimal to severe endometriosis. “Further research is needed considering the management of different subtypes of endometriosis and comparing laparoscopic interventions with lifestyle and medical interventions (Cochrane Database Syst Rev. 2020 Oct;2020[10]:CD011031).”
The treatment of endometriosis is directly related to the desire for and timing of fertility since therapy is often contraceptive, as opposed to surgery. Because endometriosis is exacerbated by estradiol, the mainstay of medical therapy is initially combined hormonal or progestin-only contraception as a means of reducing pelvic pain by reducing estradiol production and action, respectively. GnRH-agonist suppression of follicle stimulation hormone and luteinizing hormone remains the standard for inactivating endogenous estradiol. In 2018, the U.S. Food and Drug Administration approved elagolix for the treatment of pain associated with endometriosis – the first pill specifically approved for endometriosis pain relief. An off-label approach for women is letrozole, the aromatase inhibitor, to reduce circulating estradiol levels. Unfortunately, estradiol suppression cannot be used solely long term without add-back therapy, because of the risk of bone loss and vasomotor symptoms.
Excision of endometriomas adversely affects ovarian follicular reserve (as indicated by lower levels of anti-müllerian hormone and reduced ovarian antral follicle counts on ultrasound). For women who want to preserve their fertility, the potential benefits of surgery should be weighed against these negative effects. Surgical treatment of endometriosis in women without other identifiable infertility factors may improve rates of spontaneous pregnancy. In women with moderate to severe endometriosis, intrauterine insemination with ovarian stimulation may be of value, particularly with preceding GnRH-agonist therapy (J Endometr Pelvic Pain Disord. 2018;10[3]:158-73).
Despite the reduction in IVF outcomes in women with moderate to severe endometriosis, it remains unclear whether surgery improves the likelihood of pregnancy with IVF as does the concurrent use of prolonged GnRH agonist during IVF stimulation. (Fertil Steril. 2012;98:591-8).
Summary
- Medical therapy alone does not appear to improve fertility in endometriosis.
- Surgical treatment of endometriosis improves natural fertility, particularly in lower-stage endometriosis.
- EFI is a useful tool to predict postoperative natural fertility and assess the need for IVF.
- Despite advanced endometriosis reducing IVF outcomes, surgery or medical pretreatment to increase IVF success remains unproven.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
Pain is classified as chronic when it lasts or recurs for more than 3-6 months (“Classification of chronic pain” 2nd ed. Seattle: IASP Press, 1994). This universally accepted definition does not distinguish between physical and emotional pain. Categorically, pain is pain. Two prevalent chronic gynecologic diseases are closely related medically and emotionally. Forty percent to 50% of women with endometriosis have infertility; 30%-50% of women with infertility are found to have coexisting endometriosis. The approach to both is, typically, symptomatic treatment. In this month’s column, I examine the relationship between these ailments and how we can advise women on management.
Endometriosis is simply defined as the displacement of normal endometrial glands and stroma from their natural anatomical location to elsewhere in the body. With the recent identification of the disease in the spleen, endometriosis has been found in every organ system. Endometriosis is identified in 6%-10% of the general female population. The prevalence ranges from 2% to 11% among asymptomatic women and from 5% to 21% in women hospitalized for pelvic pain (Best Pract Res Clin Obstet Gynaecol. 2018;51:1-15). Compared with fertile women, infertile women are six to eight times more likely to have endometriosis (Fertil Steril. 2012;98:591-8).
Retrograde menstruation is the presumed theory for the origins of endometriosis, that is, the reflux of menstrual debris containing active endometrial cells through the fallopian tubes into the peritoneal cavity (Am J Obstet Gynecol. 1927;14:422-69). Because of the varied etiologies of the most common symptoms of endometriosis, dysmenorrhea, dyspareunia, dyschezia, and infertility, women visit, on average, seven physicians before being diagnosed (Fertil Steril. 2011;96:366). The delay in promptly identifying endometriosis is further impaired by the lack of specific biomarkers, awareness, and inadequate evaluation (N Engl J Med. 2020;382:1244-56).
The 2008 U.S. health care costs for endometriosis were approximately $4,000 per affected woman, analogous to the costs for other chronic conditions such as type 2 diabetes, Crohn’s disease, and rheumatoid arthritis (Hum Reprod. 2012;27:1292-9). The management of symptoms further increases the financial burden because of the effect of the disease on physical, mental, sexual, and social well-being, as well as productivity (Health Qual Life Outcomes. 2019;17:123).
We have known the paradoxical relationship between the stage of endometriosis and symptoms: Women with low-stage disease may present with severe pain and/or infertility but those with advanced-stage disease may be asymptomatic. Endometriotic cells and tissue elicit a localized immune and inflammatory response with the production of cytokines, chemokines, and prostaglandins. Given the usual intra-abdominal location and the small size of implants, endometriosis requires a surgical diagnosis, ideally with histopathology for confirmation. However, imaging – transvaginal ultrasound or MRI – has more than 90% sensitivity and specificity for identifying endometriomas (Cochrane Database Syst Rev. 2016;2[2]:CD009591).
The effect of endometriosis on fertility, particularly in women with minimal to mild stages, is not clear, and many studies have been retrospective. Tubal factor infertility can be a result of endometriosis. Per the 2020 Cochrane Database Systemic Reviews (2020 Oct;2020[10]:CD011031), “Compared to diagnostic laparoscopy only, it is uncertain whether laparoscopic surgery reduces overall pain associated with minimal to severe endometriosis; no data were reported on live birth. There is moderate-quality evidence that laparoscopic surgery increases viable intrauterine pregnancy rates confirmed by ultrasound compared to diagnostic laparoscopy only.” In women undergoing IVF, more advanced stages of endometriosis have reduced pregnancy outcomes as shown in recent meta-analyses (Obstet Gynecol. 2015;125:79-88).
The revised ASRM (rASRM) surgical staging classification of endometriosis has been widely used to describe the degree, although it poorly correlates with fertility potential (Fertil Steril. 2012;98:591-8). Women diagnosed with endometriosis may benefit from the Endometriosis Fertility Index (EFI), published in 2010 as a useful scoring system to predict postoperative non-IVF pregnancy rates (both by natural means and intrauterine insemination) based on patient characteristics, rASRM staging and “least function” score of the adnexa (Fertil Steril. 2010;94:1609-15).
Compared with diagnostic laparoscopy only, it is uncertain whether laparoscopic surgery reduces overall pain associated with minimal to severe endometriosis. “Further research is needed considering the management of different subtypes of endometriosis and comparing laparoscopic interventions with lifestyle and medical interventions (Cochrane Database Syst Rev. 2020 Oct;2020[10]:CD011031).”
The treatment of endometriosis is directly related to the desire for and timing of fertility since therapy is often contraceptive, as opposed to surgery. Because endometriosis is exacerbated by estradiol, the mainstay of medical therapy is initially combined hormonal or progestin-only contraception as a means of reducing pelvic pain by reducing estradiol production and action, respectively. GnRH-agonist suppression of follicle stimulation hormone and luteinizing hormone remains the standard for inactivating endogenous estradiol. In 2018, the U.S. Food and Drug Administration approved elagolix for the treatment of pain associated with endometriosis – the first pill specifically approved for endometriosis pain relief. An off-label approach for women is letrozole, the aromatase inhibitor, to reduce circulating estradiol levels. Unfortunately, estradiol suppression cannot be used solely long term without add-back therapy, because of the risk of bone loss and vasomotor symptoms.
Excision of endometriomas adversely affects ovarian follicular reserve (as indicated by lower levels of anti-müllerian hormone and reduced ovarian antral follicle counts on ultrasound). For women who want to preserve their fertility, the potential benefits of surgery should be weighed against these negative effects. Surgical treatment of endometriosis in women without other identifiable infertility factors may improve rates of spontaneous pregnancy. In women with moderate to severe endometriosis, intrauterine insemination with ovarian stimulation may be of value, particularly with preceding GnRH-agonist therapy (J Endometr Pelvic Pain Disord. 2018;10[3]:158-73).
Despite the reduction in IVF outcomes in women with moderate to severe endometriosis, it remains unclear whether surgery improves the likelihood of pregnancy with IVF as does the concurrent use of prolonged GnRH agonist during IVF stimulation. (Fertil Steril. 2012;98:591-8).
Summary
- Medical therapy alone does not appear to improve fertility in endometriosis.
- Surgical treatment of endometriosis improves natural fertility, particularly in lower-stage endometriosis.
- EFI is a useful tool to predict postoperative natural fertility and assess the need for IVF.
- Despite advanced endometriosis reducing IVF outcomes, surgery or medical pretreatment to increase IVF success remains unproven.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
Pain is classified as chronic when it lasts or recurs for more than 3-6 months (“Classification of chronic pain” 2nd ed. Seattle: IASP Press, 1994). This universally accepted definition does not distinguish between physical and emotional pain. Categorically, pain is pain. Two prevalent chronic gynecologic diseases are closely related medically and emotionally. Forty percent to 50% of women with endometriosis have infertility; 30%-50% of women with infertility are found to have coexisting endometriosis. The approach to both is, typically, symptomatic treatment. In this month’s column, I examine the relationship between these ailments and how we can advise women on management.
Endometriosis is simply defined as the displacement of normal endometrial glands and stroma from their natural anatomical location to elsewhere in the body. With the recent identification of the disease in the spleen, endometriosis has been found in every organ system. Endometriosis is identified in 6%-10% of the general female population. The prevalence ranges from 2% to 11% among asymptomatic women and from 5% to 21% in women hospitalized for pelvic pain (Best Pract Res Clin Obstet Gynaecol. 2018;51:1-15). Compared with fertile women, infertile women are six to eight times more likely to have endometriosis (Fertil Steril. 2012;98:591-8).
Retrograde menstruation is the presumed theory for the origins of endometriosis, that is, the reflux of menstrual debris containing active endometrial cells through the fallopian tubes into the peritoneal cavity (Am J Obstet Gynecol. 1927;14:422-69). Because of the varied etiologies of the most common symptoms of endometriosis, dysmenorrhea, dyspareunia, dyschezia, and infertility, women visit, on average, seven physicians before being diagnosed (Fertil Steril. 2011;96:366). The delay in promptly identifying endometriosis is further impaired by the lack of specific biomarkers, awareness, and inadequate evaluation (N Engl J Med. 2020;382:1244-56).
The 2008 U.S. health care costs for endometriosis were approximately $4,000 per affected woman, analogous to the costs for other chronic conditions such as type 2 diabetes, Crohn’s disease, and rheumatoid arthritis (Hum Reprod. 2012;27:1292-9). The management of symptoms further increases the financial burden because of the effect of the disease on physical, mental, sexual, and social well-being, as well as productivity (Health Qual Life Outcomes. 2019;17:123).
We have known the paradoxical relationship between the stage of endometriosis and symptoms: Women with low-stage disease may present with severe pain and/or infertility but those with advanced-stage disease may be asymptomatic. Endometriotic cells and tissue elicit a localized immune and inflammatory response with the production of cytokines, chemokines, and prostaglandins. Given the usual intra-abdominal location and the small size of implants, endometriosis requires a surgical diagnosis, ideally with histopathology for confirmation. However, imaging – transvaginal ultrasound or MRI – has more than 90% sensitivity and specificity for identifying endometriomas (Cochrane Database Syst Rev. 2016;2[2]:CD009591).
The effect of endometriosis on fertility, particularly in women with minimal to mild stages, is not clear, and many studies have been retrospective. Tubal factor infertility can be a result of endometriosis. Per the 2020 Cochrane Database Systemic Reviews (2020 Oct;2020[10]:CD011031), “Compared to diagnostic laparoscopy only, it is uncertain whether laparoscopic surgery reduces overall pain associated with minimal to severe endometriosis; no data were reported on live birth. There is moderate-quality evidence that laparoscopic surgery increases viable intrauterine pregnancy rates confirmed by ultrasound compared to diagnostic laparoscopy only.” In women undergoing IVF, more advanced stages of endometriosis have reduced pregnancy outcomes as shown in recent meta-analyses (Obstet Gynecol. 2015;125:79-88).
The revised ASRM (rASRM) surgical staging classification of endometriosis has been widely used to describe the degree, although it poorly correlates with fertility potential (Fertil Steril. 2012;98:591-8). Women diagnosed with endometriosis may benefit from the Endometriosis Fertility Index (EFI), published in 2010 as a useful scoring system to predict postoperative non-IVF pregnancy rates (both by natural means and intrauterine insemination) based on patient characteristics, rASRM staging and “least function” score of the adnexa (Fertil Steril. 2010;94:1609-15).
Compared with diagnostic laparoscopy only, it is uncertain whether laparoscopic surgery reduces overall pain associated with minimal to severe endometriosis. “Further research is needed considering the management of different subtypes of endometriosis and comparing laparoscopic interventions with lifestyle and medical interventions (Cochrane Database Syst Rev. 2020 Oct;2020[10]:CD011031).”
The treatment of endometriosis is directly related to the desire for and timing of fertility since therapy is often contraceptive, as opposed to surgery. Because endometriosis is exacerbated by estradiol, the mainstay of medical therapy is initially combined hormonal or progestin-only contraception as a means of reducing pelvic pain by reducing estradiol production and action, respectively. GnRH-agonist suppression of follicle stimulation hormone and luteinizing hormone remains the standard for inactivating endogenous estradiol. In 2018, the U.S. Food and Drug Administration approved elagolix for the treatment of pain associated with endometriosis – the first pill specifically approved for endometriosis pain relief. An off-label approach for women is letrozole, the aromatase inhibitor, to reduce circulating estradiol levels. Unfortunately, estradiol suppression cannot be used solely long term without add-back therapy, because of the risk of bone loss and vasomotor symptoms.
Excision of endometriomas adversely affects ovarian follicular reserve (as indicated by lower levels of anti-müllerian hormone and reduced ovarian antral follicle counts on ultrasound). For women who want to preserve their fertility, the potential benefits of surgery should be weighed against these negative effects. Surgical treatment of endometriosis in women without other identifiable infertility factors may improve rates of spontaneous pregnancy. In women with moderate to severe endometriosis, intrauterine insemination with ovarian stimulation may be of value, particularly with preceding GnRH-agonist therapy (J Endometr Pelvic Pain Disord. 2018;10[3]:158-73).
Despite the reduction in IVF outcomes in women with moderate to severe endometriosis, it remains unclear whether surgery improves the likelihood of pregnancy with IVF as does the concurrent use of prolonged GnRH agonist during IVF stimulation. (Fertil Steril. 2012;98:591-8).
Summary
- Medical therapy alone does not appear to improve fertility in endometriosis.
- Surgical treatment of endometriosis improves natural fertility, particularly in lower-stage endometriosis.
- EFI is a useful tool to predict postoperative natural fertility and assess the need for IVF.
- Despite advanced endometriosis reducing IVF outcomes, surgery or medical pretreatment to increase IVF success remains unproven.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.