User login
Secukinumab shows promise in hidradenitis suppurativa
PARIS – David Rosmarin, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“It was especially notable that secukinumab was effective in five of the six patients who had previously failed anti-TNF [tumor necrosis factor] therapy,” said Dr. Rosmarin, a dermatologist at Tufts University, Boston.
At present, the sole medication approved for treatment of hidradenitis suppurativa (HS) is the TNF inhibitor adalimumab (Humira). The rationale for investigating secukinumab (Cosentyx), a biologic that blocks the interleukin-17A receptor and is approved for treatment of psoriasis, psoriatic arthritis, and ankylosing spondylitis, lies in the observation that HS lesions exhibit an increased ratio of Th17- to T-regulatory cells, compared with normal skin. Lesional skin also features elevated IL-17 levels. These abnormalities can be reversed by anti-TNF therapy, he explained.
Dr. Rosmarin presented a 28-week, open-label study in which 18 patients with moderate to severe HS received an induction regimen consisting of 300 mg secukinumab given subcutaneously once weekly for 5 weeks and were then randomized to the same dose of the biologic given either every 2 or 4 weeks until week 24.
The primary endpoint was achievement of a Hidradenitis Suppurativa Clinical Response (HiSCR) at 24 weeks. This requires no increase in the number of draining fistulae or abscesses, compared with baseline, plus at least a 50% reduction in total inflammatory nodules. Secondary endpoints were the mean change from baseline in the Sartorius Scale as well as on the Dermatology Life Quality Index (DLQI).
Participants were an average of 33 years and had a disease duration of 12 years; 14 patients were Hurley Stage II, the rest Stage III. Their mean baseline DLQI was 12.
“Patients with hidradenitis suppurativa have a horrible quality of life,” Dr. Rosmarin commented. “When we compare it to patients who have atopic dermatitis, psoriasis, or chronic idiopathic urticaria, oftentimes patients with hidradenitis suppurativa suffer the worst and have the most quality of life impairment.”
Of the 18 patients, 14 – 7 of 9 in each group – achieved HiSCR. This included five of the six patients who were previous nonresponders to adalimumab or another anti-TNF biologic.
“The other thing we noticed was the rapidity of response, which is important to a lot of our patients. It took an average of 7 weeks to achieve HiSCR, and eight patients achieved HiSCR during the induction phase of treatment,” the dermatologist said.
Mean scores on the Sartorius Scale dropped by 28%. Similarly, scores on the DLQI improved by a mean of 3.6 points, or 26%. Nine patients experienced a reduction of 5 points or more on the DLQI. “This happened largely in the first 1-2 months of therapy,” Dr. Rosmarin continued.
Secukinumab was well tolerated. There were no treatment discontinuations because of adverse events. Four patients, all in the biweekly dosing arm, developed Candida infections, all easily cured using topical ketoconazole.
The next step will be to conduct a large, placebo-controlled, randomized trial to firmly establish the efficacy of secukinumab for HS. Also, the optimal dosing of the biologic for induction and long-term maintenance therapy have yet to be determined. Over the long term, it will be important to see whether marked improvement in HS is accompanied by a reduction in the elevated cardiovascular risk associated with this inflammatory disease, he added.
In 2019, a trial will get underway to compare two doses of secukinumab for patients with HS. Based on a search of clinical trials at ClinicalTrials.gov, a wide range of monoclonal antibody therapies are being investigated for the treatment of HS.
The results of this preliminary study of secukinumab emphasize the importance of the Th17 pathway in HS and open the door to alternative strategies targeting this pathway. Dr. Rosmarin noted that he and his coinvestigators have collected a case series of positive responses to guselkumab (Tremfya), which targets the IL-23 p19 subunit, which also lies along the Th17 pathway.
The secukinumab study was sponsored by Novartis. Dr. Rosmarin reported serving as a consultant to or on speakers’ bureaus for that company and more than half a dozen other pharmaceutical companies.
PARIS – David Rosmarin, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“It was especially notable that secukinumab was effective in five of the six patients who had previously failed anti-TNF [tumor necrosis factor] therapy,” said Dr. Rosmarin, a dermatologist at Tufts University, Boston.
At present, the sole medication approved for treatment of hidradenitis suppurativa (HS) is the TNF inhibitor adalimumab (Humira). The rationale for investigating secukinumab (Cosentyx), a biologic that blocks the interleukin-17A receptor and is approved for treatment of psoriasis, psoriatic arthritis, and ankylosing spondylitis, lies in the observation that HS lesions exhibit an increased ratio of Th17- to T-regulatory cells, compared with normal skin. Lesional skin also features elevated IL-17 levels. These abnormalities can be reversed by anti-TNF therapy, he explained.
Dr. Rosmarin presented a 28-week, open-label study in which 18 patients with moderate to severe HS received an induction regimen consisting of 300 mg secukinumab given subcutaneously once weekly for 5 weeks and were then randomized to the same dose of the biologic given either every 2 or 4 weeks until week 24.
The primary endpoint was achievement of a Hidradenitis Suppurativa Clinical Response (HiSCR) at 24 weeks. This requires no increase in the number of draining fistulae or abscesses, compared with baseline, plus at least a 50% reduction in total inflammatory nodules. Secondary endpoints were the mean change from baseline in the Sartorius Scale as well as on the Dermatology Life Quality Index (DLQI).
Participants were an average of 33 years and had a disease duration of 12 years; 14 patients were Hurley Stage II, the rest Stage III. Their mean baseline DLQI was 12.
“Patients with hidradenitis suppurativa have a horrible quality of life,” Dr. Rosmarin commented. “When we compare it to patients who have atopic dermatitis, psoriasis, or chronic idiopathic urticaria, oftentimes patients with hidradenitis suppurativa suffer the worst and have the most quality of life impairment.”
Of the 18 patients, 14 – 7 of 9 in each group – achieved HiSCR. This included five of the six patients who were previous nonresponders to adalimumab or another anti-TNF biologic.
“The other thing we noticed was the rapidity of response, which is important to a lot of our patients. It took an average of 7 weeks to achieve HiSCR, and eight patients achieved HiSCR during the induction phase of treatment,” the dermatologist said.
Mean scores on the Sartorius Scale dropped by 28%. Similarly, scores on the DLQI improved by a mean of 3.6 points, or 26%. Nine patients experienced a reduction of 5 points or more on the DLQI. “This happened largely in the first 1-2 months of therapy,” Dr. Rosmarin continued.
Secukinumab was well tolerated. There were no treatment discontinuations because of adverse events. Four patients, all in the biweekly dosing arm, developed Candida infections, all easily cured using topical ketoconazole.
The next step will be to conduct a large, placebo-controlled, randomized trial to firmly establish the efficacy of secukinumab for HS. Also, the optimal dosing of the biologic for induction and long-term maintenance therapy have yet to be determined. Over the long term, it will be important to see whether marked improvement in HS is accompanied by a reduction in the elevated cardiovascular risk associated with this inflammatory disease, he added.
In 2019, a trial will get underway to compare two doses of secukinumab for patients with HS. Based on a search of clinical trials at ClinicalTrials.gov, a wide range of monoclonal antibody therapies are being investigated for the treatment of HS.
The results of this preliminary study of secukinumab emphasize the importance of the Th17 pathway in HS and open the door to alternative strategies targeting this pathway. Dr. Rosmarin noted that he and his coinvestigators have collected a case series of positive responses to guselkumab (Tremfya), which targets the IL-23 p19 subunit, which also lies along the Th17 pathway.
The secukinumab study was sponsored by Novartis. Dr. Rosmarin reported serving as a consultant to or on speakers’ bureaus for that company and more than half a dozen other pharmaceutical companies.
PARIS – David Rosmarin, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“It was especially notable that secukinumab was effective in five of the six patients who had previously failed anti-TNF [tumor necrosis factor] therapy,” said Dr. Rosmarin, a dermatologist at Tufts University, Boston.
At present, the sole medication approved for treatment of hidradenitis suppurativa (HS) is the TNF inhibitor adalimumab (Humira). The rationale for investigating secukinumab (Cosentyx), a biologic that blocks the interleukin-17A receptor and is approved for treatment of psoriasis, psoriatic arthritis, and ankylosing spondylitis, lies in the observation that HS lesions exhibit an increased ratio of Th17- to T-regulatory cells, compared with normal skin. Lesional skin also features elevated IL-17 levels. These abnormalities can be reversed by anti-TNF therapy, he explained.
Dr. Rosmarin presented a 28-week, open-label study in which 18 patients with moderate to severe HS received an induction regimen consisting of 300 mg secukinumab given subcutaneously once weekly for 5 weeks and were then randomized to the same dose of the biologic given either every 2 or 4 weeks until week 24.
The primary endpoint was achievement of a Hidradenitis Suppurativa Clinical Response (HiSCR) at 24 weeks. This requires no increase in the number of draining fistulae or abscesses, compared with baseline, plus at least a 50% reduction in total inflammatory nodules. Secondary endpoints were the mean change from baseline in the Sartorius Scale as well as on the Dermatology Life Quality Index (DLQI).
Participants were an average of 33 years and had a disease duration of 12 years; 14 patients were Hurley Stage II, the rest Stage III. Their mean baseline DLQI was 12.
“Patients with hidradenitis suppurativa have a horrible quality of life,” Dr. Rosmarin commented. “When we compare it to patients who have atopic dermatitis, psoriasis, or chronic idiopathic urticaria, oftentimes patients with hidradenitis suppurativa suffer the worst and have the most quality of life impairment.”
Of the 18 patients, 14 – 7 of 9 in each group – achieved HiSCR. This included five of the six patients who were previous nonresponders to adalimumab or another anti-TNF biologic.
“The other thing we noticed was the rapidity of response, which is important to a lot of our patients. It took an average of 7 weeks to achieve HiSCR, and eight patients achieved HiSCR during the induction phase of treatment,” the dermatologist said.
Mean scores on the Sartorius Scale dropped by 28%. Similarly, scores on the DLQI improved by a mean of 3.6 points, or 26%. Nine patients experienced a reduction of 5 points or more on the DLQI. “This happened largely in the first 1-2 months of therapy,” Dr. Rosmarin continued.
Secukinumab was well tolerated. There were no treatment discontinuations because of adverse events. Four patients, all in the biweekly dosing arm, developed Candida infections, all easily cured using topical ketoconazole.
The next step will be to conduct a large, placebo-controlled, randomized trial to firmly establish the efficacy of secukinumab for HS. Also, the optimal dosing of the biologic for induction and long-term maintenance therapy have yet to be determined. Over the long term, it will be important to see whether marked improvement in HS is accompanied by a reduction in the elevated cardiovascular risk associated with this inflammatory disease, he added.
In 2019, a trial will get underway to compare two doses of secukinumab for patients with HS. Based on a search of clinical trials at ClinicalTrials.gov, a wide range of monoclonal antibody therapies are being investigated for the treatment of HS.
The results of this preliminary study of secukinumab emphasize the importance of the Th17 pathway in HS and open the door to alternative strategies targeting this pathway. Dr. Rosmarin noted that he and his coinvestigators have collected a case series of positive responses to guselkumab (Tremfya), which targets the IL-23 p19 subunit, which also lies along the Th17 pathway.
The secukinumab study was sponsored by Novartis. Dr. Rosmarin reported serving as a consultant to or on speakers’ bureaus for that company and more than half a dozen other pharmaceutical companies.
REPORTING FROM THE EADV CONGRESS
Key clinical point: Secukinumab shows considerable promise for treatment of hidradenitis suppurativa.
Major finding: Hidradenitis suppurativa improved markedly in response to secukinumab in 14 of 18 patients.
Study details: This prospective, open-label, 28-week study included 18 patients with hidradenitis suppurativa who were randomized to one of two secukinumab dosing regimens.
Disclosures: The study was sponsored by Novartis. The presenter reported serving as a consultant to or on speakers’ bureaus for that company and more than half a dozen other pharmaceutical companies.
Sandoz halts pursuit of U.S. approval for rituximab biosimilar
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.
Experience – not evidence – is best guide in keloid treatment
LAS VEGAS – The first written mention of keloids in history came about 4,500 years ago. But there is still so much more to be learned about keloids, according to dermatologist Hilary E. Baldwin, MD.
Yes, there is more information about who gets keloids and where they appear on the body. “What we do not know is everything else,” Dr. Baldwin said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We don’t even know which treatments work. Not a single treatment has any evidence basis behind it. It’s all fly-by-night, seat-of-your-pants, based on your knowledge and that of those who treat keloids frequently.”
. Cases show an unknown factor serves as a contributing factor, she said.
There’s no gender disparity in keloids, she said, and they’re more likely to occur after puberty than before. Family history isn’t helpful.
Also, while darker skin produces keloids more easily and consistently than other skin types, these keloids aren’t the hardest to treat, said Dr. Baldwin, medical director of the Acne Treatment and Research Center in Morristown, N.J. “From my personal experience – but not from data or papers – keloids are the most difficult to treat and recur the most when you see them in a Caucasian patient.”
Patients want the keloids to disappear forever along with any evidence that they ever existed. “Virtually every patient says ‘I want this cut off, I want it gone,’” Dr. Baldwin said. “And they want it gone yesterday. Few understand this is a long process. I tell them we’ll become extraordinarily good friends over the next 6-12 months while we get rid of this thing.” Moreover, it’s not possible to eliminate keloids without leaving a sign behind. “Most want it eradicated with normal skin,” but the skin beneath a keloid “will never look normal,” she observed.
The following are some tips for treating patients with keloids that she provided during her presentation:
- Surgery is usually not an option, and it may be necessary to convince patients about other treatment options. “Most don’t want corticosteroid injections, but that is how we treat them and keep them from coming back,” Dr. Baldwin said.
- Surgery may be appropriate in certain cases, such as keloids that are shaped like mushrooms and are attached to the skin via a stalk. Patient compliance with adjunctive therapy (injected corticosteroids) is crucial to prevent recurrence after surgery. Be aware that some patients – such as those who are afraid of needles – may refuse to return. Earlobes, a common site of keloids because of earrings, are a special case in surgery. Patients with earlobe keloids often fare well after surgery, Dr. Baldwin said, and recurrence is less common than in other parts of the body, especially when corticosteroids are added. It helps that patients are often more compliant with adjunctive therapy, she added, because the keloids are highly visible, and they want to wear earrings again.
- Ask patients what bothers them the most about the keloids. Some may want to eliminate burning, itching, or stinging, and Dr. Baldwin attempts to treat those issues. Patients may want keloids to be softer, flatter, or lighter, she said.
- Modification of the keloid’s appearance may be enough for some patients. For example, one of her patients objected to extensive keloids around his breasts because they gave him the appearance of cleavage. Another believed – accurately – that his keloid looked like male genitalia. Dr. Baldwin treated it with the keloid equivalent of castration (surgically removing its “testicles”) and circumcision of sorts (a flattening of its “glans penis” with corticosteroids). “It didn’t look pretty,” she said, “but it no longer looked offensive to him.”
- Many patients present with keloids on the chest, back, and deltoids, “which have to have been from acne, but you can’t actually see the pimples,” Dr. Baldwin said. In these cases, she may turn to isotretinoin. This is not for treatment of keloids, but is a preventive strategy to stop further lesions from appearing and causing more keloids, she explained.
- In addition to corticosteroids, other postsurgical options include freezing, interferon, pressure dressings, dextran hydrogel scaffolding, and radiation therapy.
Moving forward, researchers “are looking at biologics, TNF-alpha inhibitors, and tyrosine kinase inhibitors in hopes that we can find something that will stop the incessant forward movement of some of our keloid patients,” Dr. Baldwin noted.
Dr. Baldwin reported no relevant disclosures.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – The first written mention of keloids in history came about 4,500 years ago. But there is still so much more to be learned about keloids, according to dermatologist Hilary E. Baldwin, MD.
Yes, there is more information about who gets keloids and where they appear on the body. “What we do not know is everything else,” Dr. Baldwin said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We don’t even know which treatments work. Not a single treatment has any evidence basis behind it. It’s all fly-by-night, seat-of-your-pants, based on your knowledge and that of those who treat keloids frequently.”
. Cases show an unknown factor serves as a contributing factor, she said.
There’s no gender disparity in keloids, she said, and they’re more likely to occur after puberty than before. Family history isn’t helpful.
Also, while darker skin produces keloids more easily and consistently than other skin types, these keloids aren’t the hardest to treat, said Dr. Baldwin, medical director of the Acne Treatment and Research Center in Morristown, N.J. “From my personal experience – but not from data or papers – keloids are the most difficult to treat and recur the most when you see them in a Caucasian patient.”
Patients want the keloids to disappear forever along with any evidence that they ever existed. “Virtually every patient says ‘I want this cut off, I want it gone,’” Dr. Baldwin said. “And they want it gone yesterday. Few understand this is a long process. I tell them we’ll become extraordinarily good friends over the next 6-12 months while we get rid of this thing.” Moreover, it’s not possible to eliminate keloids without leaving a sign behind. “Most want it eradicated with normal skin,” but the skin beneath a keloid “will never look normal,” she observed.
The following are some tips for treating patients with keloids that she provided during her presentation:
- Surgery is usually not an option, and it may be necessary to convince patients about other treatment options. “Most don’t want corticosteroid injections, but that is how we treat them and keep them from coming back,” Dr. Baldwin said.
- Surgery may be appropriate in certain cases, such as keloids that are shaped like mushrooms and are attached to the skin via a stalk. Patient compliance with adjunctive therapy (injected corticosteroids) is crucial to prevent recurrence after surgery. Be aware that some patients – such as those who are afraid of needles – may refuse to return. Earlobes, a common site of keloids because of earrings, are a special case in surgery. Patients with earlobe keloids often fare well after surgery, Dr. Baldwin said, and recurrence is less common than in other parts of the body, especially when corticosteroids are added. It helps that patients are often more compliant with adjunctive therapy, she added, because the keloids are highly visible, and they want to wear earrings again.
- Ask patients what bothers them the most about the keloids. Some may want to eliminate burning, itching, or stinging, and Dr. Baldwin attempts to treat those issues. Patients may want keloids to be softer, flatter, or lighter, she said.
- Modification of the keloid’s appearance may be enough for some patients. For example, one of her patients objected to extensive keloids around his breasts because they gave him the appearance of cleavage. Another believed – accurately – that his keloid looked like male genitalia. Dr. Baldwin treated it with the keloid equivalent of castration (surgically removing its “testicles”) and circumcision of sorts (a flattening of its “glans penis” with corticosteroids). “It didn’t look pretty,” she said, “but it no longer looked offensive to him.”
- Many patients present with keloids on the chest, back, and deltoids, “which have to have been from acne, but you can’t actually see the pimples,” Dr. Baldwin said. In these cases, she may turn to isotretinoin. This is not for treatment of keloids, but is a preventive strategy to stop further lesions from appearing and causing more keloids, she explained.
- In addition to corticosteroids, other postsurgical options include freezing, interferon, pressure dressings, dextran hydrogel scaffolding, and radiation therapy.
Moving forward, researchers “are looking at biologics, TNF-alpha inhibitors, and tyrosine kinase inhibitors in hopes that we can find something that will stop the incessant forward movement of some of our keloid patients,” Dr. Baldwin noted.
Dr. Baldwin reported no relevant disclosures.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – The first written mention of keloids in history came about 4,500 years ago. But there is still so much more to be learned about keloids, according to dermatologist Hilary E. Baldwin, MD.
Yes, there is more information about who gets keloids and where they appear on the body. “What we do not know is everything else,” Dr. Baldwin said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. “We don’t even know which treatments work. Not a single treatment has any evidence basis behind it. It’s all fly-by-night, seat-of-your-pants, based on your knowledge and that of those who treat keloids frequently.”
. Cases show an unknown factor serves as a contributing factor, she said.
There’s no gender disparity in keloids, she said, and they’re more likely to occur after puberty than before. Family history isn’t helpful.
Also, while darker skin produces keloids more easily and consistently than other skin types, these keloids aren’t the hardest to treat, said Dr. Baldwin, medical director of the Acne Treatment and Research Center in Morristown, N.J. “From my personal experience – but not from data or papers – keloids are the most difficult to treat and recur the most when you see them in a Caucasian patient.”
Patients want the keloids to disappear forever along with any evidence that they ever existed. “Virtually every patient says ‘I want this cut off, I want it gone,’” Dr. Baldwin said. “And they want it gone yesterday. Few understand this is a long process. I tell them we’ll become extraordinarily good friends over the next 6-12 months while we get rid of this thing.” Moreover, it’s not possible to eliminate keloids without leaving a sign behind. “Most want it eradicated with normal skin,” but the skin beneath a keloid “will never look normal,” she observed.
The following are some tips for treating patients with keloids that she provided during her presentation:
- Surgery is usually not an option, and it may be necessary to convince patients about other treatment options. “Most don’t want corticosteroid injections, but that is how we treat them and keep them from coming back,” Dr. Baldwin said.
- Surgery may be appropriate in certain cases, such as keloids that are shaped like mushrooms and are attached to the skin via a stalk. Patient compliance with adjunctive therapy (injected corticosteroids) is crucial to prevent recurrence after surgery. Be aware that some patients – such as those who are afraid of needles – may refuse to return. Earlobes, a common site of keloids because of earrings, are a special case in surgery. Patients with earlobe keloids often fare well after surgery, Dr. Baldwin said, and recurrence is less common than in other parts of the body, especially when corticosteroids are added. It helps that patients are often more compliant with adjunctive therapy, she added, because the keloids are highly visible, and they want to wear earrings again.
- Ask patients what bothers them the most about the keloids. Some may want to eliminate burning, itching, or stinging, and Dr. Baldwin attempts to treat those issues. Patients may want keloids to be softer, flatter, or lighter, she said.
- Modification of the keloid’s appearance may be enough for some patients. For example, one of her patients objected to extensive keloids around his breasts because they gave him the appearance of cleavage. Another believed – accurately – that his keloid looked like male genitalia. Dr. Baldwin treated it with the keloid equivalent of castration (surgically removing its “testicles”) and circumcision of sorts (a flattening of its “glans penis” with corticosteroids). “It didn’t look pretty,” she said, “but it no longer looked offensive to him.”
- Many patients present with keloids on the chest, back, and deltoids, “which have to have been from acne, but you can’t actually see the pimples,” Dr. Baldwin said. In these cases, she may turn to isotretinoin. This is not for treatment of keloids, but is a preventive strategy to stop further lesions from appearing and causing more keloids, she explained.
- In addition to corticosteroids, other postsurgical options include freezing, interferon, pressure dressings, dextran hydrogel scaffolding, and radiation therapy.
Moving forward, researchers “are looking at biologics, TNF-alpha inhibitors, and tyrosine kinase inhibitors in hopes that we can find something that will stop the incessant forward movement of some of our keloid patients,” Dr. Baldwin noted.
Dr. Baldwin reported no relevant disclosures.
SDEF and this news organization are owned by the same parent company.
AT SDEF LAS VEGAS DERMATOLOGY SEMINAR
Investigational agent VT-1161 looks promising for onychomycosis
LAS VEGAS – An investigational oral therapy for onychomycosis could be on the horizon as a new treatment option.
VT-1161 is a cytochrome P51 (CYP51) inhibitor with potent in vitro activity against several species of tinea and yeast, David M. Pariser, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. It is highly selective for fungal CYP51 over human cytochrome P enzymes.
Dr. Pariser served as an . Patients enrolled were aged 18-70 years, with a mean age of 49. Most were men (80%) and white (85%), as is typical in trials for onychomycosis drugs, noted Dr. Pariser, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Distal subungual onychomycosis was evident in 25%-75% of the nail, and patients needed 2 mm of clear nail to be included in the trial. Disease was confirmed by positive KOH staining and culture.
Patients were randomized into four active treatment groups; a fifth received placebo. All active treatment groups were started on 14 days of a daily loading dose of VT-1161, with one arm getting 300 mg per day and one getting 600 mg per day.
After the loading dose, two groups were treated for 12 weeks with VT-1161 (weekly doses of 300 mg and 600 mg); two other groups had treatment extended out to 24 weeks.
Few patients, roughly 20% in each treatment arm, had achieved the primary endpoint, a complete cure (0% nail involvement and negative KOH stain and culture) at the end of active treatment, Dr. Pariser said.
“I personally think that all of these onychomycosis studies should be carried out for longer than a year, up to 2 years, even if you were able to get rid of all the fungus, because that’s how long it’s going to take for the nail to grow out, especially in older patients with slower nail growth,” Dr. Pariser said.
At 48 weeks, approximately 40% of patients in each active treatment arm achieved complete cure, Dr. Pariser noted. “The results indicated that there was not a lot of difference in outcomes based on the dose the patient received.”
About 60%-70% of treated patients sustained mycologic cure of onychomycosis at 60 weeks.
No serious drug-related adverse events were seen in the study, and no patients dropped out because of lab abnormalities, including liver function tests. Other adverse events were rare and occurred equally in the treatment and placebo arms and consisted of dermatitis, headache, and cough. Nausea, cough, and dysgeusia each occurred in 2% of patients and could have been related to the study drug, Dr. Pariser said.
Looking at both mycologic and complete cure rates of available onychomycosis treatments, the results from the RENOVATE trial are comparable to approved systemic therapies, and superior to topicals, he said.
Dr. Pariser disclosed serving as an investigator for pharmaceutical manufactures Viamet (maker of VT-1161), Valeant, and Ancor/Pharmaderm.
SDEF and this news organization are owned by the same parent company.
[email protected]
LAS VEGAS – An investigational oral therapy for onychomycosis could be on the horizon as a new treatment option.
VT-1161 is a cytochrome P51 (CYP51) inhibitor with potent in vitro activity against several species of tinea and yeast, David M. Pariser, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. It is highly selective for fungal CYP51 over human cytochrome P enzymes.
Dr. Pariser served as an . Patients enrolled were aged 18-70 years, with a mean age of 49. Most were men (80%) and white (85%), as is typical in trials for onychomycosis drugs, noted Dr. Pariser, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Distal subungual onychomycosis was evident in 25%-75% of the nail, and patients needed 2 mm of clear nail to be included in the trial. Disease was confirmed by positive KOH staining and culture.
Patients were randomized into four active treatment groups; a fifth received placebo. All active treatment groups were started on 14 days of a daily loading dose of VT-1161, with one arm getting 300 mg per day and one getting 600 mg per day.
After the loading dose, two groups were treated for 12 weeks with VT-1161 (weekly doses of 300 mg and 600 mg); two other groups had treatment extended out to 24 weeks.
Few patients, roughly 20% in each treatment arm, had achieved the primary endpoint, a complete cure (0% nail involvement and negative KOH stain and culture) at the end of active treatment, Dr. Pariser said.
“I personally think that all of these onychomycosis studies should be carried out for longer than a year, up to 2 years, even if you were able to get rid of all the fungus, because that’s how long it’s going to take for the nail to grow out, especially in older patients with slower nail growth,” Dr. Pariser said.
At 48 weeks, approximately 40% of patients in each active treatment arm achieved complete cure, Dr. Pariser noted. “The results indicated that there was not a lot of difference in outcomes based on the dose the patient received.”
About 60%-70% of treated patients sustained mycologic cure of onychomycosis at 60 weeks.
No serious drug-related adverse events were seen in the study, and no patients dropped out because of lab abnormalities, including liver function tests. Other adverse events were rare and occurred equally in the treatment and placebo arms and consisted of dermatitis, headache, and cough. Nausea, cough, and dysgeusia each occurred in 2% of patients and could have been related to the study drug, Dr. Pariser said.
Looking at both mycologic and complete cure rates of available onychomycosis treatments, the results from the RENOVATE trial are comparable to approved systemic therapies, and superior to topicals, he said.
Dr. Pariser disclosed serving as an investigator for pharmaceutical manufactures Viamet (maker of VT-1161), Valeant, and Ancor/Pharmaderm.
SDEF and this news organization are owned by the same parent company.
[email protected]
LAS VEGAS – An investigational oral therapy for onychomycosis could be on the horizon as a new treatment option.
VT-1161 is a cytochrome P51 (CYP51) inhibitor with potent in vitro activity against several species of tinea and yeast, David M. Pariser, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. It is highly selective for fungal CYP51 over human cytochrome P enzymes.
Dr. Pariser served as an . Patients enrolled were aged 18-70 years, with a mean age of 49. Most were men (80%) and white (85%), as is typical in trials for onychomycosis drugs, noted Dr. Pariser, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Distal subungual onychomycosis was evident in 25%-75% of the nail, and patients needed 2 mm of clear nail to be included in the trial. Disease was confirmed by positive KOH staining and culture.
Patients were randomized into four active treatment groups; a fifth received placebo. All active treatment groups were started on 14 days of a daily loading dose of VT-1161, with one arm getting 300 mg per day and one getting 600 mg per day.
After the loading dose, two groups were treated for 12 weeks with VT-1161 (weekly doses of 300 mg and 600 mg); two other groups had treatment extended out to 24 weeks.
Few patients, roughly 20% in each treatment arm, had achieved the primary endpoint, a complete cure (0% nail involvement and negative KOH stain and culture) at the end of active treatment, Dr. Pariser said.
“I personally think that all of these onychomycosis studies should be carried out for longer than a year, up to 2 years, even if you were able to get rid of all the fungus, because that’s how long it’s going to take for the nail to grow out, especially in older patients with slower nail growth,” Dr. Pariser said.
At 48 weeks, approximately 40% of patients in each active treatment arm achieved complete cure, Dr. Pariser noted. “The results indicated that there was not a lot of difference in outcomes based on the dose the patient received.”
About 60%-70% of treated patients sustained mycologic cure of onychomycosis at 60 weeks.
No serious drug-related adverse events were seen in the study, and no patients dropped out because of lab abnormalities, including liver function tests. Other adverse events were rare and occurred equally in the treatment and placebo arms and consisted of dermatitis, headache, and cough. Nausea, cough, and dysgeusia each occurred in 2% of patients and could have been related to the study drug, Dr. Pariser said.
Looking at both mycologic and complete cure rates of available onychomycosis treatments, the results from the RENOVATE trial are comparable to approved systemic therapies, and superior to topicals, he said.
Dr. Pariser disclosed serving as an investigator for pharmaceutical manufactures Viamet (maker of VT-1161), Valeant, and Ancor/Pharmaderm.
SDEF and this news organization are owned by the same parent company.
[email protected]
REPORTING FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Delusional infestation: not so rare
PARIS – Ever wonder, when encountering an occasional patient afflicted with delusional infestation, just how common this mental disorder is?
John J. Kohorst, MD, and his coinvestigators at the Mayo Clinic in Rochester, Minn., have the evidence-based answer.
The age- and sex-adjusted point prevalence of delusional infestation among Olmsted County, Minn., residents on the final day of 2010 was 27.3 cases per 100,000 person-years, he reported at the annual congress of the European Academy of Dermatology and Venereology.
“This is the than previously suspected,” according to the dermatologist.
He and his coinvestigators retrospectively analyzed data from the Rochester Epidemiology Project. They identified 22 female and 13 male county residents with a firm diagnosis of delusional infestation, also known as delusional parasitosis. This disorder is marked by a patient’s fixed false belief that they are infested with insects, worms, or other pathogens.
The prevalence was similar in men and women. The most striking study finding was how heavily age-dependent delusional infestation was. Before age 40, the prevalence was a mere 1.2 cases per 100,000 person-years. Among 40- to 59-year-old Olmsted County residents, it was 35/100,000, jumping to 64.5/100,000 in the 60- to 79-year-old age bracket, then doubling to 130.1 cases per 100,000 person-years in individuals aged 80 or older.
Dr. Kohorst reported having no financial conflicts regarding his study, conducted free of commercial support.
PARIS – Ever wonder, when encountering an occasional patient afflicted with delusional infestation, just how common this mental disorder is?
John J. Kohorst, MD, and his coinvestigators at the Mayo Clinic in Rochester, Minn., have the evidence-based answer.
The age- and sex-adjusted point prevalence of delusional infestation among Olmsted County, Minn., residents on the final day of 2010 was 27.3 cases per 100,000 person-years, he reported at the annual congress of the European Academy of Dermatology and Venereology.
“This is the than previously suspected,” according to the dermatologist.
He and his coinvestigators retrospectively analyzed data from the Rochester Epidemiology Project. They identified 22 female and 13 male county residents with a firm diagnosis of delusional infestation, also known as delusional parasitosis. This disorder is marked by a patient’s fixed false belief that they are infested with insects, worms, or other pathogens.
The prevalence was similar in men and women. The most striking study finding was how heavily age-dependent delusional infestation was. Before age 40, the prevalence was a mere 1.2 cases per 100,000 person-years. Among 40- to 59-year-old Olmsted County residents, it was 35/100,000, jumping to 64.5/100,000 in the 60- to 79-year-old age bracket, then doubling to 130.1 cases per 100,000 person-years in individuals aged 80 or older.
Dr. Kohorst reported having no financial conflicts regarding his study, conducted free of commercial support.
PARIS – Ever wonder, when encountering an occasional patient afflicted with delusional infestation, just how common this mental disorder is?
John J. Kohorst, MD, and his coinvestigators at the Mayo Clinic in Rochester, Minn., have the evidence-based answer.
The age- and sex-adjusted point prevalence of delusional infestation among Olmsted County, Minn., residents on the final day of 2010 was 27.3 cases per 100,000 person-years, he reported at the annual congress of the European Academy of Dermatology and Venereology.
“This is the than previously suspected,” according to the dermatologist.
He and his coinvestigators retrospectively analyzed data from the Rochester Epidemiology Project. They identified 22 female and 13 male county residents with a firm diagnosis of delusional infestation, also known as delusional parasitosis. This disorder is marked by a patient’s fixed false belief that they are infested with insects, worms, or other pathogens.
The prevalence was similar in men and women. The most striking study finding was how heavily age-dependent delusional infestation was. Before age 40, the prevalence was a mere 1.2 cases per 100,000 person-years. Among 40- to 59-year-old Olmsted County residents, it was 35/100,000, jumping to 64.5/100,000 in the 60- to 79-year-old age bracket, then doubling to 130.1 cases per 100,000 person-years in individuals aged 80 or older.
Dr. Kohorst reported having no financial conflicts regarding his study, conducted free of commercial support.
REPORTING FROM THE EADV CONGRESS
Key clinical point: Delusional infestation may be more common than previously suspected, particularly among older age groups.
Major finding: The age- and sex-adjusted point prevalence of delusional infestation among residents of one county in southeastern Minnesota is 27.3 cases per 100,000 person-years.
Study details: This was a retrospective analysis of data from the Rochester (Minn.) Epidemiology Project.
Disclosures: The presenter reported having no financial conflicts regarding his study, conducted free of commercial support.
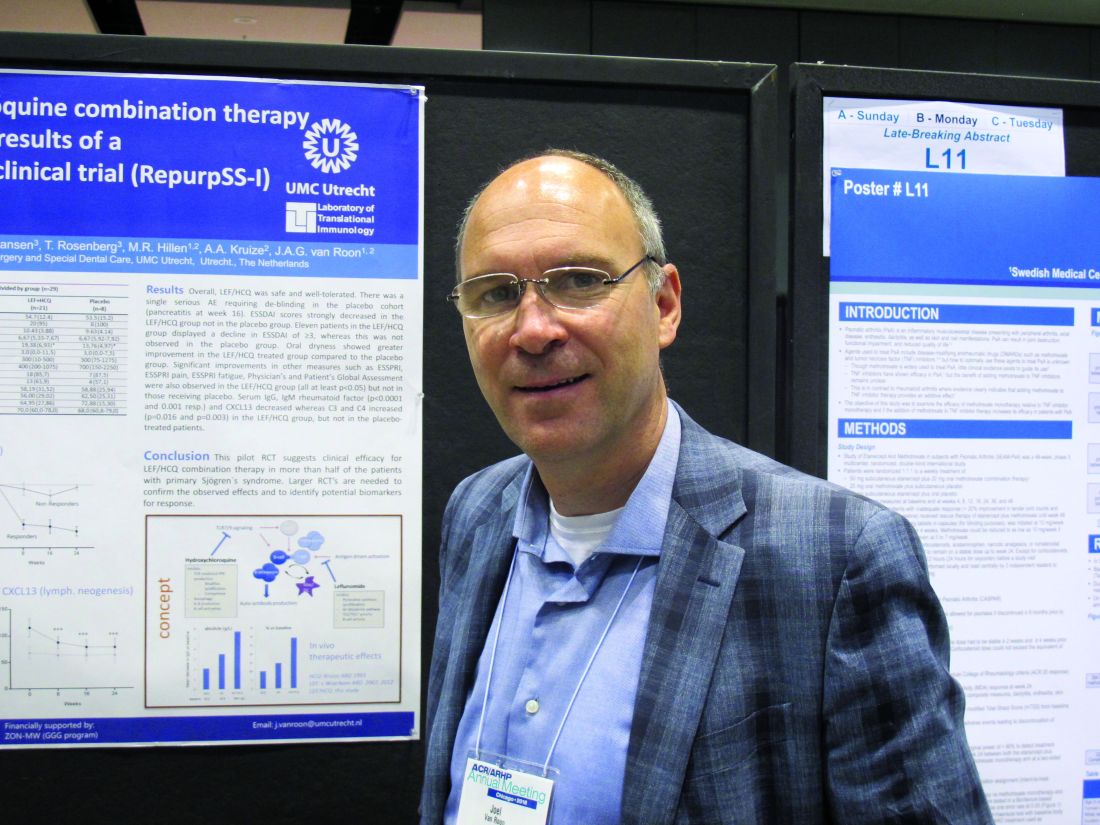
Leflunomide-hydroxychloroquine combo shows promise in primary Sjögren’s pilot study
CHICAGO – Combination therapy with leflunomide and hydroxychloroquine met all goals for efficacy, safety, and tolerability among patients with primary Sjögren’s syndrome in a randomized, placebo-controlled pilot study, lending support to evidence suggesting the two drugs have additive benefits.
The combined treatment was associated with a statistically significant decrease in the EULAR Sjögren’s syndrome disease activity index (ESSDAI) over 24 weeks – the primary endpoint of the study – in 21 patients in the treatment group. The ESSDAI score on combination treatment dropped from about 10 at baseline to about 6 at 24 weeks, compared with no change from a baseline of about 10 in eight patients in the placebo group. An ESSDAI decrease of 3 or more points occurred in 11 patients in the combination therapy group, compared with none in the placebo group, Joel A.G. van Roon, PhD, a researcher in the Laboratory of Translational Immunology at the University Medical Center Utrecht, the Netherlands, reported in a late-breaking poster at the annual meeting of the American College of Rheumatology.
Both leflunomide and hydroxychloroquine have been shown to inhibit B-cell hyperactivity, but the clinical benefits have been modest and not statistically significant. Since the two agents have complementary inhibitory properties on different immune cells – including B and T cells and plasmacytoid dendritic cells, and based on in vitro findings of additive benefits with respect to inhibition of T- and B-cell activation and CXCL13 production, Dr. van Roon and his colleagues conducted this double-blind, single-center, proof-of-concept pilot study (REPURpSS-1) to assess the efficacy, safety, and tolerability of combined treatment in primary Sjögren’s syndrome.
In all, 29 patients with clinically active disease, defined by ESSDAI of 5 or greater, were randomized 2:1 to receive either 20 mg of leflunomide daily plus 400 mg of hydroxychloroquine daily or placebo/placebo for 24 weeks.
Secondary endpoints such as oral dryness also improved significantly in the treatment group versus the placebo group. Stimulated whole saliva flow increased from about 800 mcL/5 min to about 1,400 mcL/5 min and decreased from about 1,250 to about 1,000 mcL/5 min in the groups, respectively. Median EULAR Sjögren’s syndrome patient reported index (ESSPRI), ESSPRI pain, and ESSPRI fatigue scores, as well as Physician’s and Patient’s Global Assessment scores each improved significantly in the treatment group (at least P less than .05 in all cases) but not in the placebo groups, said Dr. van Roon.
Additionally, serum IgG, IgM rheumatoid factor, and chemokine CXCL13 – a marker for lymphoid neogenesis – decreased significantly, and complement components 3 and 4 (C3 and C4) increased significantly by 24 weeks in the treatment group, but not in the placebo group. B-cell hyperactivity as measured by serum IgG decreased from about 20 g/L to about 14 g/L versus no change from about 15 g/L at baseline in the placebo group, he noted.
“Overall, combination leflunomide and hydroxychloroquine was safe and well tolerated, but larger randomized, controlled trials are needed to confirm the observed effects and to identify potential biomarkers for response,” he concluded.
This study was supported by ZonMw (the Netherlands Organization for Health Research and Development). Dr. van Roon reported having no relevant disclosures.
SOURCE: van Roon JAG et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract L10.
CHICAGO – Combination therapy with leflunomide and hydroxychloroquine met all goals for efficacy, safety, and tolerability among patients with primary Sjögren’s syndrome in a randomized, placebo-controlled pilot study, lending support to evidence suggesting the two drugs have additive benefits.
The combined treatment was associated with a statistically significant decrease in the EULAR Sjögren’s syndrome disease activity index (ESSDAI) over 24 weeks – the primary endpoint of the study – in 21 patients in the treatment group. The ESSDAI score on combination treatment dropped from about 10 at baseline to about 6 at 24 weeks, compared with no change from a baseline of about 10 in eight patients in the placebo group. An ESSDAI decrease of 3 or more points occurred in 11 patients in the combination therapy group, compared with none in the placebo group, Joel A.G. van Roon, PhD, a researcher in the Laboratory of Translational Immunology at the University Medical Center Utrecht, the Netherlands, reported in a late-breaking poster at the annual meeting of the American College of Rheumatology.
Both leflunomide and hydroxychloroquine have been shown to inhibit B-cell hyperactivity, but the clinical benefits have been modest and not statistically significant. Since the two agents have complementary inhibitory properties on different immune cells – including B and T cells and plasmacytoid dendritic cells, and based on in vitro findings of additive benefits with respect to inhibition of T- and B-cell activation and CXCL13 production, Dr. van Roon and his colleagues conducted this double-blind, single-center, proof-of-concept pilot study (REPURpSS-1) to assess the efficacy, safety, and tolerability of combined treatment in primary Sjögren’s syndrome.
In all, 29 patients with clinically active disease, defined by ESSDAI of 5 or greater, were randomized 2:1 to receive either 20 mg of leflunomide daily plus 400 mg of hydroxychloroquine daily or placebo/placebo for 24 weeks.
Secondary endpoints such as oral dryness also improved significantly in the treatment group versus the placebo group. Stimulated whole saliva flow increased from about 800 mcL/5 min to about 1,400 mcL/5 min and decreased from about 1,250 to about 1,000 mcL/5 min in the groups, respectively. Median EULAR Sjögren’s syndrome patient reported index (ESSPRI), ESSPRI pain, and ESSPRI fatigue scores, as well as Physician’s and Patient’s Global Assessment scores each improved significantly in the treatment group (at least P less than .05 in all cases) but not in the placebo groups, said Dr. van Roon.
Additionally, serum IgG, IgM rheumatoid factor, and chemokine CXCL13 – a marker for lymphoid neogenesis – decreased significantly, and complement components 3 and 4 (C3 and C4) increased significantly by 24 weeks in the treatment group, but not in the placebo group. B-cell hyperactivity as measured by serum IgG decreased from about 20 g/L to about 14 g/L versus no change from about 15 g/L at baseline in the placebo group, he noted.
“Overall, combination leflunomide and hydroxychloroquine was safe and well tolerated, but larger randomized, controlled trials are needed to confirm the observed effects and to identify potential biomarkers for response,” he concluded.
This study was supported by ZonMw (the Netherlands Organization for Health Research and Development). Dr. van Roon reported having no relevant disclosures.
SOURCE: van Roon JAG et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract L10.
CHICAGO – Combination therapy with leflunomide and hydroxychloroquine met all goals for efficacy, safety, and tolerability among patients with primary Sjögren’s syndrome in a randomized, placebo-controlled pilot study, lending support to evidence suggesting the two drugs have additive benefits.
The combined treatment was associated with a statistically significant decrease in the EULAR Sjögren’s syndrome disease activity index (ESSDAI) over 24 weeks – the primary endpoint of the study – in 21 patients in the treatment group. The ESSDAI score on combination treatment dropped from about 10 at baseline to about 6 at 24 weeks, compared with no change from a baseline of about 10 in eight patients in the placebo group. An ESSDAI decrease of 3 or more points occurred in 11 patients in the combination therapy group, compared with none in the placebo group, Joel A.G. van Roon, PhD, a researcher in the Laboratory of Translational Immunology at the University Medical Center Utrecht, the Netherlands, reported in a late-breaking poster at the annual meeting of the American College of Rheumatology.
Both leflunomide and hydroxychloroquine have been shown to inhibit B-cell hyperactivity, but the clinical benefits have been modest and not statistically significant. Since the two agents have complementary inhibitory properties on different immune cells – including B and T cells and plasmacytoid dendritic cells, and based on in vitro findings of additive benefits with respect to inhibition of T- and B-cell activation and CXCL13 production, Dr. van Roon and his colleagues conducted this double-blind, single-center, proof-of-concept pilot study (REPURpSS-1) to assess the efficacy, safety, and tolerability of combined treatment in primary Sjögren’s syndrome.
In all, 29 patients with clinically active disease, defined by ESSDAI of 5 or greater, were randomized 2:1 to receive either 20 mg of leflunomide daily plus 400 mg of hydroxychloroquine daily or placebo/placebo for 24 weeks.
Secondary endpoints such as oral dryness also improved significantly in the treatment group versus the placebo group. Stimulated whole saliva flow increased from about 800 mcL/5 min to about 1,400 mcL/5 min and decreased from about 1,250 to about 1,000 mcL/5 min in the groups, respectively. Median EULAR Sjögren’s syndrome patient reported index (ESSPRI), ESSPRI pain, and ESSPRI fatigue scores, as well as Physician’s and Patient’s Global Assessment scores each improved significantly in the treatment group (at least P less than .05 in all cases) but not in the placebo groups, said Dr. van Roon.
Additionally, serum IgG, IgM rheumatoid factor, and chemokine CXCL13 – a marker for lymphoid neogenesis – decreased significantly, and complement components 3 and 4 (C3 and C4) increased significantly by 24 weeks in the treatment group, but not in the placebo group. B-cell hyperactivity as measured by serum IgG decreased from about 20 g/L to about 14 g/L versus no change from about 15 g/L at baseline in the placebo group, he noted.
“Overall, combination leflunomide and hydroxychloroquine was safe and well tolerated, but larger randomized, controlled trials are needed to confirm the observed effects and to identify potential biomarkers for response,” he concluded.
This study was supported by ZonMw (the Netherlands Organization for Health Research and Development). Dr. van Roon reported having no relevant disclosures.
SOURCE: van Roon JAG et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract L10.
REPORTING FROM THE ACR ANNUAL MEETING
Key clinical point: but larger randomized, controlled trials are needed to confirm the observed effects.
Major finding: Combined treatment was associated with a decline in EULAR Sjögren’s syndrome disease activity index score from about 10 at baseline to about 6 at 24 weeks.
Study details: A randomized, placebo-controlled pilot study of 29 patients.
Disclosures: This study was supported by ZonMw (the Netherlands Organization for Health Research and Development). Dr. van Roon reported having no relevant disclosures.
Source: van Roon JAG et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract L10.
Serlopitant quells prurigo nodularis in phase 2 study
PARIS – The in a 127-patient, randomized, placebo-controlled trial, Sonja Stander, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
She used her speaker’s platform not only to present key outcomes of the phase 2 study, but also to explain some of the underappreciated aspects of prurigo nodularis and how the results shed new light on the pathogenesis of the disease.
“There is a knowledge gap regarding this disease,” observed Dr. Stander, professor of dermatology at the University of Munster (Germany).
Prurigo nodularis is a chronic skin condition characterized by numerous intensely pruritic, symmetrically distributed, erosive nodular lesions. As Dr. Stander and her fellow members of the EADV Task Force Pruritus recently reported, prurigo nodularis should be considered as one of the clinical manifestations of chronic prurigo, hallmarks of which include neuronal sensitization to itch and development of an itch-scratch cycle (J Eur Acad Dermatol Venereol. 2018 Jul;32[7]:1059-65). At present, there is no approved treatment for prurigo nodularis in the United States or Europe.
Despite its name, prurigo nodularis is not just about the itch. Additional symptoms were frequently reported by participants in the serlopitant study. Indeed, while 97% of participants reported experiencing devilish pruritus, 52% reported a burning sensation on involved skin, 41% said the lesions were painful, and 36% indicated they were plagued by a stinging sensation. All of these disturbing sensations were significantly reduced with 8 weeks of serlopitant in the trial.
The clinical implications of these baseline findings are clear: “We have to ask not only about itch, but also about pain and burning and stinging, which are really an issue with these patients,” the dermatologist said.
Dr. Stander noted that prurigo nodularis has a large negative impact on quality of life as reflected in study participants’ mean baseline Dermatology Life Quality Index score of 61. And it’s a disease that sticks around: At enrollment, 38% of subjects had a 1- to 5-year history of prurigo nodularis, 20% had had the disease for 5-10 years, and fully 29% had had prurigo nodularis for longer than 10 years.
“This is not a chronic relapsing disease. These patients had prurigo nodularis all the time,” Dr. Stander said.
Consistent with the findings of other studies, half of patients with prurigo nodularis had an atopic predisposition as defined by an Erlangen Atopy Questionnaire score of 10 or higher.
Patients in the phase 2 study were randomized to either a 15-mg loading dose of oral serlopitant followed by 5 mg/day for 8 weeks or to placebo. All four of the most common manifestations of prurigo nodularis – itching, burning, pain, and stinging – were reduced to a significantly greater extent in the serlopitant group than with placebo. For example, the proportion of serlopitant-treated patients who rated their itching as none or mild went from zero at baseline to 54% at week 8, as compared with 26% among placebo-treated controls. After 8 weeks, 46% of serlopitant-treated patients rated their itching as moderate, severe, or very severe, as did 71% of controls.
Similarly, the proportion of patients in the serlopitant group who reported no or only a mild burning sensation climbed from 41% at baseline to 73% after 8 weeks. In the control group, the proportion improved from 36% to 47% over the course of 8 weeks. At week 8, 27% of patients on serlopitant characterized themselves as experiencing a moderate, severe, or very severe burning sensation; the rate was twice as high in controls.
Although the stinging sensation associated with prurigo nodularis was significantly reduced by serlopitant, the effect was less robust than with the other common sensory expressions of the disease. The proportion of patients with no or mild stinging increased by an absolute 23% after 8 weeks on serlopitant and by 6% with placebo. At week 8, 22% of patients treated with the neurokinin-1 receptor antagonist complained of a moderate to very severe stinging sensation, as did 40% of controls.
As a neurokinin-1 receptor antagonist, serlopitant inhibits substance P. The drug’s efficacy suggests that substance P plays a role in inducing the itch and other sensory symptoms of prurigo nodularis, according to Dr. Stander.
Based upon the favorable phase 2 results, multiple phase 3 clinical trials of serlopitant for prurigo nodularis are underway in the United States and elsewhere, including a year-long safety study.
Serlopitant is being developed by Menlo Therapeutics. Dr. Stander reported receiving research funding from and serving as a consultant to the company.
PARIS – The in a 127-patient, randomized, placebo-controlled trial, Sonja Stander, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
She used her speaker’s platform not only to present key outcomes of the phase 2 study, but also to explain some of the underappreciated aspects of prurigo nodularis and how the results shed new light on the pathogenesis of the disease.
“There is a knowledge gap regarding this disease,” observed Dr. Stander, professor of dermatology at the University of Munster (Germany).
Prurigo nodularis is a chronic skin condition characterized by numerous intensely pruritic, symmetrically distributed, erosive nodular lesions. As Dr. Stander and her fellow members of the EADV Task Force Pruritus recently reported, prurigo nodularis should be considered as one of the clinical manifestations of chronic prurigo, hallmarks of which include neuronal sensitization to itch and development of an itch-scratch cycle (J Eur Acad Dermatol Venereol. 2018 Jul;32[7]:1059-65). At present, there is no approved treatment for prurigo nodularis in the United States or Europe.
Despite its name, prurigo nodularis is not just about the itch. Additional symptoms were frequently reported by participants in the serlopitant study. Indeed, while 97% of participants reported experiencing devilish pruritus, 52% reported a burning sensation on involved skin, 41% said the lesions were painful, and 36% indicated they were plagued by a stinging sensation. All of these disturbing sensations were significantly reduced with 8 weeks of serlopitant in the trial.
The clinical implications of these baseline findings are clear: “We have to ask not only about itch, but also about pain and burning and stinging, which are really an issue with these patients,” the dermatologist said.
Dr. Stander noted that prurigo nodularis has a large negative impact on quality of life as reflected in study participants’ mean baseline Dermatology Life Quality Index score of 61. And it’s a disease that sticks around: At enrollment, 38% of subjects had a 1- to 5-year history of prurigo nodularis, 20% had had the disease for 5-10 years, and fully 29% had had prurigo nodularis for longer than 10 years.
“This is not a chronic relapsing disease. These patients had prurigo nodularis all the time,” Dr. Stander said.
Consistent with the findings of other studies, half of patients with prurigo nodularis had an atopic predisposition as defined by an Erlangen Atopy Questionnaire score of 10 or higher.
Patients in the phase 2 study were randomized to either a 15-mg loading dose of oral serlopitant followed by 5 mg/day for 8 weeks or to placebo. All four of the most common manifestations of prurigo nodularis – itching, burning, pain, and stinging – were reduced to a significantly greater extent in the serlopitant group than with placebo. For example, the proportion of serlopitant-treated patients who rated their itching as none or mild went from zero at baseline to 54% at week 8, as compared with 26% among placebo-treated controls. After 8 weeks, 46% of serlopitant-treated patients rated their itching as moderate, severe, or very severe, as did 71% of controls.
Similarly, the proportion of patients in the serlopitant group who reported no or only a mild burning sensation climbed from 41% at baseline to 73% after 8 weeks. In the control group, the proportion improved from 36% to 47% over the course of 8 weeks. At week 8, 27% of patients on serlopitant characterized themselves as experiencing a moderate, severe, or very severe burning sensation; the rate was twice as high in controls.
Although the stinging sensation associated with prurigo nodularis was significantly reduced by serlopitant, the effect was less robust than with the other common sensory expressions of the disease. The proportion of patients with no or mild stinging increased by an absolute 23% after 8 weeks on serlopitant and by 6% with placebo. At week 8, 22% of patients treated with the neurokinin-1 receptor antagonist complained of a moderate to very severe stinging sensation, as did 40% of controls.
As a neurokinin-1 receptor antagonist, serlopitant inhibits substance P. The drug’s efficacy suggests that substance P plays a role in inducing the itch and other sensory symptoms of prurigo nodularis, according to Dr. Stander.
Based upon the favorable phase 2 results, multiple phase 3 clinical trials of serlopitant for prurigo nodularis are underway in the United States and elsewhere, including a year-long safety study.
Serlopitant is being developed by Menlo Therapeutics. Dr. Stander reported receiving research funding from and serving as a consultant to the company.
PARIS – The in a 127-patient, randomized, placebo-controlled trial, Sonja Stander, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
She used her speaker’s platform not only to present key outcomes of the phase 2 study, but also to explain some of the underappreciated aspects of prurigo nodularis and how the results shed new light on the pathogenesis of the disease.
“There is a knowledge gap regarding this disease,” observed Dr. Stander, professor of dermatology at the University of Munster (Germany).
Prurigo nodularis is a chronic skin condition characterized by numerous intensely pruritic, symmetrically distributed, erosive nodular lesions. As Dr. Stander and her fellow members of the EADV Task Force Pruritus recently reported, prurigo nodularis should be considered as one of the clinical manifestations of chronic prurigo, hallmarks of which include neuronal sensitization to itch and development of an itch-scratch cycle (J Eur Acad Dermatol Venereol. 2018 Jul;32[7]:1059-65). At present, there is no approved treatment for prurigo nodularis in the United States or Europe.
Despite its name, prurigo nodularis is not just about the itch. Additional symptoms were frequently reported by participants in the serlopitant study. Indeed, while 97% of participants reported experiencing devilish pruritus, 52% reported a burning sensation on involved skin, 41% said the lesions were painful, and 36% indicated they were plagued by a stinging sensation. All of these disturbing sensations were significantly reduced with 8 weeks of serlopitant in the trial.
The clinical implications of these baseline findings are clear: “We have to ask not only about itch, but also about pain and burning and stinging, which are really an issue with these patients,” the dermatologist said.
Dr. Stander noted that prurigo nodularis has a large negative impact on quality of life as reflected in study participants’ mean baseline Dermatology Life Quality Index score of 61. And it’s a disease that sticks around: At enrollment, 38% of subjects had a 1- to 5-year history of prurigo nodularis, 20% had had the disease for 5-10 years, and fully 29% had had prurigo nodularis for longer than 10 years.
“This is not a chronic relapsing disease. These patients had prurigo nodularis all the time,” Dr. Stander said.
Consistent with the findings of other studies, half of patients with prurigo nodularis had an atopic predisposition as defined by an Erlangen Atopy Questionnaire score of 10 or higher.
Patients in the phase 2 study were randomized to either a 15-mg loading dose of oral serlopitant followed by 5 mg/day for 8 weeks or to placebo. All four of the most common manifestations of prurigo nodularis – itching, burning, pain, and stinging – were reduced to a significantly greater extent in the serlopitant group than with placebo. For example, the proportion of serlopitant-treated patients who rated their itching as none or mild went from zero at baseline to 54% at week 8, as compared with 26% among placebo-treated controls. After 8 weeks, 46% of serlopitant-treated patients rated their itching as moderate, severe, or very severe, as did 71% of controls.
Similarly, the proportion of patients in the serlopitant group who reported no or only a mild burning sensation climbed from 41% at baseline to 73% after 8 weeks. In the control group, the proportion improved from 36% to 47% over the course of 8 weeks. At week 8, 27% of patients on serlopitant characterized themselves as experiencing a moderate, severe, or very severe burning sensation; the rate was twice as high in controls.
Although the stinging sensation associated with prurigo nodularis was significantly reduced by serlopitant, the effect was less robust than with the other common sensory expressions of the disease. The proportion of patients with no or mild stinging increased by an absolute 23% after 8 weeks on serlopitant and by 6% with placebo. At week 8, 22% of patients treated with the neurokinin-1 receptor antagonist complained of a moderate to very severe stinging sensation, as did 40% of controls.
As a neurokinin-1 receptor antagonist, serlopitant inhibits substance P. The drug’s efficacy suggests that substance P plays a role in inducing the itch and other sensory symptoms of prurigo nodularis, according to Dr. Stander.
Based upon the favorable phase 2 results, multiple phase 3 clinical trials of serlopitant for prurigo nodularis are underway in the United States and elsewhere, including a year-long safety study.
Serlopitant is being developed by Menlo Therapeutics. Dr. Stander reported receiving research funding from and serving as a consultant to the company.
REPORTING FROM THE EADV CONGRESS
Key clinical point: Serlopitant decreases the disruptive itch, burning, pain, and stinging of prurigo nodularis.
Major finding: The proportion of prurigo nodularis patients who rated their itching as none or mild went from zero at baseline to 54% after 8 weeks on oral serlopitant.
Study details: This 8-week, randomized, double-blind phase 2 study included 127 patients with prurigo nodularis who were randomized to oral serlopitant or placebo.
Disclosures: Serlopitant is being developed by Menlo Therapeutics. The presenter reported receiving research funding from and serving as a consultant to the company.
SLE low-disease definition receives prospective validation
CHICAGO – The long path toward a validated definition of low disease activity in patients with systemic lupus erythematosus may be nearing an end as a definition first proposed more than 5 years ago received validation with data from more than 1,700 patients in a prospective, multicenter study.
The next step is to test this definition in treat-to-target intervention studies, and to apply the definition in other clinical trials as well as in routine clinical practice, Vera Golder, MD, said at the annual meeting of the American College of Rheumatology.
The tested definition has five elements that a patient needs to achieve to be considered in a lupus low disease activity state (LLDAS):
• A systemic lupus erythematosus (SLE) disease activity index (SLEDAI) score of 4 or less with no major organ involvement.
• No new disease activity.
• A physician’s global assessment of the patient of 1 or less on a 0-3 scale.
• Maintenance on a prednisolone dosage of 7.5 mg/day or less.
• Maintenance on a standard immunosuppressive regimen.
The Asia Pacific Lupus Collaboration (APLC) first proposed this definition of LLDAS in 2013 (Ann Rheum Dis. 2013 June;72[Suppl 3]:THU0298), and was the organization behind the latest test of its validity. The APLC based its LLDAS definition on recommendations made by an international working party (Ann Rheum Dis. 2014 June;73[6]:958-67).
The APLC prospectively collected data from 1,735 SLE patients at 13 centers in eight countries during May 2013–December 2016, with a median follow-up of 2.2 years. During that time, 78% of the patients achieved the LLDAS at least once. Two-thirds of the patients had at least one sustained period of LLDAS of at least 3 months, and overall the enrolled patients spent 69% of the time in the LLDAS, reported Dr. Golder, a rheumatologist at Monash University in Melbourne.
The validation analysis she described focused on examining the correlation between the amount of time that patients spent in the defined LLDAS and their subsequent clinical outcomes. The analysis showed that when patients were in the LLDAS their rate of subsequent flare or damage accrual was substantially reduced.
Patients in the LLDAS for at least half the time had a 51% reduced rate of subsequent flare and a 47% reduced rate of subsequent damage accrual, she reported. Patients with a LLDAS of 3-6 months had a 57% reduced rate of damage accrual. As time spent in continuous LLDAS continued to increase the rate of subsequent damage accrual continued to drop until the duration reached more than 9 months, at which point the rate of subsequent damage fell to nearly 90% lower than that of patients without this amount of sustained LLDAS. Patients with LLDAS sustained for more than 9 months and as long as 12 months had an 86% reduction in subsequent damage accrual. Periods of sustained LLDAS that extended longer than a year continued to maintain a nearly 90% reduced rate of damage accrual, Dr. Golder said.
The Asia Pacific Lupus Collaboration has received grants from AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Janssen, and UCB. Dr. Golder had no disclosures.
SOURCE: Golder V et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2786.
Having a validated, formal definition of low disease activity in patients with systemic lupus erythematosus will be very helpful to clinicians and patients with this disease. The lack of such a widely accepted definition of a treatment target until now has been a significant issue that had made it more challenging to advise and treat patients.
The concept of a low disease state has been much easier to define in other rheumatic diseases, but lupus has posed a major challenge because of its very heterogeneous presentation. This heterogeneity has led to the creation of several measures of disease activity as well as multiple serologic parameters that also help define disease activity. It’s unrealistic to expect most lupus patients to be in a low disease activity state all the time.
Validation of a reasonable definition of low activity is a great, pragmatic step forward for our field that will help clinicians better care for their patients with systemic lupus erythematosus.
Lisa R. Sammaritano, MD , is a rheumatologist at the Hospital for Special Surgery and Cornell University in New York. She had no disclosures. She made these comments in an interview.
Having a validated, formal definition of low disease activity in patients with systemic lupus erythematosus will be very helpful to clinicians and patients with this disease. The lack of such a widely accepted definition of a treatment target until now has been a significant issue that had made it more challenging to advise and treat patients.
The concept of a low disease state has been much easier to define in other rheumatic diseases, but lupus has posed a major challenge because of its very heterogeneous presentation. This heterogeneity has led to the creation of several measures of disease activity as well as multiple serologic parameters that also help define disease activity. It’s unrealistic to expect most lupus patients to be in a low disease activity state all the time.
Validation of a reasonable definition of low activity is a great, pragmatic step forward for our field that will help clinicians better care for their patients with systemic lupus erythematosus.
Lisa R. Sammaritano, MD , is a rheumatologist at the Hospital for Special Surgery and Cornell University in New York. She had no disclosures. She made these comments in an interview.
Having a validated, formal definition of low disease activity in patients with systemic lupus erythematosus will be very helpful to clinicians and patients with this disease. The lack of such a widely accepted definition of a treatment target until now has been a significant issue that had made it more challenging to advise and treat patients.
The concept of a low disease state has been much easier to define in other rheumatic diseases, but lupus has posed a major challenge because of its very heterogeneous presentation. This heterogeneity has led to the creation of several measures of disease activity as well as multiple serologic parameters that also help define disease activity. It’s unrealistic to expect most lupus patients to be in a low disease activity state all the time.
Validation of a reasonable definition of low activity is a great, pragmatic step forward for our field that will help clinicians better care for their patients with systemic lupus erythematosus.
Lisa R. Sammaritano, MD , is a rheumatologist at the Hospital for Special Surgery and Cornell University in New York. She had no disclosures. She made these comments in an interview.
CHICAGO – The long path toward a validated definition of low disease activity in patients with systemic lupus erythematosus may be nearing an end as a definition first proposed more than 5 years ago received validation with data from more than 1,700 patients in a prospective, multicenter study.
The next step is to test this definition in treat-to-target intervention studies, and to apply the definition in other clinical trials as well as in routine clinical practice, Vera Golder, MD, said at the annual meeting of the American College of Rheumatology.
The tested definition has five elements that a patient needs to achieve to be considered in a lupus low disease activity state (LLDAS):
• A systemic lupus erythematosus (SLE) disease activity index (SLEDAI) score of 4 or less with no major organ involvement.
• No new disease activity.
• A physician’s global assessment of the patient of 1 or less on a 0-3 scale.
• Maintenance on a prednisolone dosage of 7.5 mg/day or less.
• Maintenance on a standard immunosuppressive regimen.
The Asia Pacific Lupus Collaboration (APLC) first proposed this definition of LLDAS in 2013 (Ann Rheum Dis. 2013 June;72[Suppl 3]:THU0298), and was the organization behind the latest test of its validity. The APLC based its LLDAS definition on recommendations made by an international working party (Ann Rheum Dis. 2014 June;73[6]:958-67).
The APLC prospectively collected data from 1,735 SLE patients at 13 centers in eight countries during May 2013–December 2016, with a median follow-up of 2.2 years. During that time, 78% of the patients achieved the LLDAS at least once. Two-thirds of the patients had at least one sustained period of LLDAS of at least 3 months, and overall the enrolled patients spent 69% of the time in the LLDAS, reported Dr. Golder, a rheumatologist at Monash University in Melbourne.
The validation analysis she described focused on examining the correlation between the amount of time that patients spent in the defined LLDAS and their subsequent clinical outcomes. The analysis showed that when patients were in the LLDAS their rate of subsequent flare or damage accrual was substantially reduced.
Patients in the LLDAS for at least half the time had a 51% reduced rate of subsequent flare and a 47% reduced rate of subsequent damage accrual, she reported. Patients with a LLDAS of 3-6 months had a 57% reduced rate of damage accrual. As time spent in continuous LLDAS continued to increase the rate of subsequent damage accrual continued to drop until the duration reached more than 9 months, at which point the rate of subsequent damage fell to nearly 90% lower than that of patients without this amount of sustained LLDAS. Patients with LLDAS sustained for more than 9 months and as long as 12 months had an 86% reduction in subsequent damage accrual. Periods of sustained LLDAS that extended longer than a year continued to maintain a nearly 90% reduced rate of damage accrual, Dr. Golder said.
The Asia Pacific Lupus Collaboration has received grants from AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Janssen, and UCB. Dr. Golder had no disclosures.
SOURCE: Golder V et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2786.
CHICAGO – The long path toward a validated definition of low disease activity in patients with systemic lupus erythematosus may be nearing an end as a definition first proposed more than 5 years ago received validation with data from more than 1,700 patients in a prospective, multicenter study.
The next step is to test this definition in treat-to-target intervention studies, and to apply the definition in other clinical trials as well as in routine clinical practice, Vera Golder, MD, said at the annual meeting of the American College of Rheumatology.
The tested definition has five elements that a patient needs to achieve to be considered in a lupus low disease activity state (LLDAS):
• A systemic lupus erythematosus (SLE) disease activity index (SLEDAI) score of 4 or less with no major organ involvement.
• No new disease activity.
• A physician’s global assessment of the patient of 1 or less on a 0-3 scale.
• Maintenance on a prednisolone dosage of 7.5 mg/day or less.
• Maintenance on a standard immunosuppressive regimen.
The Asia Pacific Lupus Collaboration (APLC) first proposed this definition of LLDAS in 2013 (Ann Rheum Dis. 2013 June;72[Suppl 3]:THU0298), and was the organization behind the latest test of its validity. The APLC based its LLDAS definition on recommendations made by an international working party (Ann Rheum Dis. 2014 June;73[6]:958-67).
The APLC prospectively collected data from 1,735 SLE patients at 13 centers in eight countries during May 2013–December 2016, with a median follow-up of 2.2 years. During that time, 78% of the patients achieved the LLDAS at least once. Two-thirds of the patients had at least one sustained period of LLDAS of at least 3 months, and overall the enrolled patients spent 69% of the time in the LLDAS, reported Dr. Golder, a rheumatologist at Monash University in Melbourne.
The validation analysis she described focused on examining the correlation between the amount of time that patients spent in the defined LLDAS and their subsequent clinical outcomes. The analysis showed that when patients were in the LLDAS their rate of subsequent flare or damage accrual was substantially reduced.
Patients in the LLDAS for at least half the time had a 51% reduced rate of subsequent flare and a 47% reduced rate of subsequent damage accrual, she reported. Patients with a LLDAS of 3-6 months had a 57% reduced rate of damage accrual. As time spent in continuous LLDAS continued to increase the rate of subsequent damage accrual continued to drop until the duration reached more than 9 months, at which point the rate of subsequent damage fell to nearly 90% lower than that of patients without this amount of sustained LLDAS. Patients with LLDAS sustained for more than 9 months and as long as 12 months had an 86% reduction in subsequent damage accrual. Periods of sustained LLDAS that extended longer than a year continued to maintain a nearly 90% reduced rate of damage accrual, Dr. Golder said.
The Asia Pacific Lupus Collaboration has received grants from AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Janssen, and UCB. Dr. Golder had no disclosures.
SOURCE: Golder V et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2786.
REPORTING FROM THE ACR ANNUAL MEETING
Key clinical point: A proposed definition of low disease activity in systemic lupus erythematosus received prospective validation.
Major finding: Patients meeting the definition for 9-12 months had an 86% reduced rate of subsequent damage accrual.
Study details: A prospective, multicenter study of 1,735 patients.
Disclosures: The Asia Pacific Lupus Collaboration has received grants from AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Janssen, and UCB. Dr. Golder had no disclosures.
Source: Golder V et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2786.
November 2018
Desmoplastic trichilemmoma
that presents as a solitary, skin colored lesion on the midface. Lesions may appear smooth or verrucous. Lesions may occur alongside trichoepitheliomas. They may also occur on genital skin and resemble condyloma acuminata.
Histopathology reveals downward lobular growth of the epidermis. Keratinocytes are clear secondary to periodic acid-Schiff (PAS)–positive glycogen in the cells. In desmoplastic trichilemmoma, small clusters of cells are arranged in an infiltrative pattern that resembles invasive carcinoma. Often, the desmoplastic areas are surrounded by benign-appearing trichilemmomas, which helps to make the diagnosis. Desmoplastic trichilemmomas can also occur within nevus sebaceous. As trichilemmoma is a benign growth; no treatment is needed. However, if further removal is desired, electrodesiccation, cryotherapy, shave removal, or excision are treatment options. Rarely seen, the malignant counterpart to trichilemmomas is a trichilemmal carcinoma, which requires surgical excision or Mohs.
The appearance of multiple trichilemmomas is a marker for Cowden syndrome. Cowden syndrome is a rare autosomal dominant disorder in which there is a mutation in a tumor-suppressor gene called PTEN. Patients may have oral mucosal papillomas, sclerotic fibromas, acral keratotic papules, and are at risk for the development of adenocarcinoma of the breast, gastrointestinal tract, and thyroid.
Trichoepithelioma is a benign neoplasm derived from follicular germ cells that presents as a skin-colored papule on the midface, especially the nose. Multiple trichoepitheliomas are a marker for Brooke-Spiegler syndrome. A desmoplastic trichoepithelioma is a variant that has stromal sclerosis on pathology. It is a benign lesion, although may be difficult to differentiate from sclerosing basal cell or microcystic adnexal carcinoma.
Angiofibroma, or fibrous papule, is a commonly seen, benign, skin-colored papule also often occurring on the nose. They can be treated for cosmetic purposes. Multiple lesions are associated with tuberous sclerosis.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit a case for possible publication, send an email to [email protected].
Desmoplastic trichilemmoma
that presents as a solitary, skin colored lesion on the midface. Lesions may appear smooth or verrucous. Lesions may occur alongside trichoepitheliomas. They may also occur on genital skin and resemble condyloma acuminata.
Histopathology reveals downward lobular growth of the epidermis. Keratinocytes are clear secondary to periodic acid-Schiff (PAS)–positive glycogen in the cells. In desmoplastic trichilemmoma, small clusters of cells are arranged in an infiltrative pattern that resembles invasive carcinoma. Often, the desmoplastic areas are surrounded by benign-appearing trichilemmomas, which helps to make the diagnosis. Desmoplastic trichilemmomas can also occur within nevus sebaceous. As trichilemmoma is a benign growth; no treatment is needed. However, if further removal is desired, electrodesiccation, cryotherapy, shave removal, or excision are treatment options. Rarely seen, the malignant counterpart to trichilemmomas is a trichilemmal carcinoma, which requires surgical excision or Mohs.
The appearance of multiple trichilemmomas is a marker for Cowden syndrome. Cowden syndrome is a rare autosomal dominant disorder in which there is a mutation in a tumor-suppressor gene called PTEN. Patients may have oral mucosal papillomas, sclerotic fibromas, acral keratotic papules, and are at risk for the development of adenocarcinoma of the breast, gastrointestinal tract, and thyroid.
Trichoepithelioma is a benign neoplasm derived from follicular germ cells that presents as a skin-colored papule on the midface, especially the nose. Multiple trichoepitheliomas are a marker for Brooke-Spiegler syndrome. A desmoplastic trichoepithelioma is a variant that has stromal sclerosis on pathology. It is a benign lesion, although may be difficult to differentiate from sclerosing basal cell or microcystic adnexal carcinoma.
Angiofibroma, or fibrous papule, is a commonly seen, benign, skin-colored papule also often occurring on the nose. They can be treated for cosmetic purposes. Multiple lesions are associated with tuberous sclerosis.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit a case for possible publication, send an email to [email protected].
Desmoplastic trichilemmoma
that presents as a solitary, skin colored lesion on the midface. Lesions may appear smooth or verrucous. Lesions may occur alongside trichoepitheliomas. They may also occur on genital skin and resemble condyloma acuminata.
Histopathology reveals downward lobular growth of the epidermis. Keratinocytes are clear secondary to periodic acid-Schiff (PAS)–positive glycogen in the cells. In desmoplastic trichilemmoma, small clusters of cells are arranged in an infiltrative pattern that resembles invasive carcinoma. Often, the desmoplastic areas are surrounded by benign-appearing trichilemmomas, which helps to make the diagnosis. Desmoplastic trichilemmomas can also occur within nevus sebaceous. As trichilemmoma is a benign growth; no treatment is needed. However, if further removal is desired, electrodesiccation, cryotherapy, shave removal, or excision are treatment options. Rarely seen, the malignant counterpart to trichilemmomas is a trichilemmal carcinoma, which requires surgical excision or Mohs.
The appearance of multiple trichilemmomas is a marker for Cowden syndrome. Cowden syndrome is a rare autosomal dominant disorder in which there is a mutation in a tumor-suppressor gene called PTEN. Patients may have oral mucosal papillomas, sclerotic fibromas, acral keratotic papules, and are at risk for the development of adenocarcinoma of the breast, gastrointestinal tract, and thyroid.
Trichoepithelioma is a benign neoplasm derived from follicular germ cells that presents as a skin-colored papule on the midface, especially the nose. Multiple trichoepitheliomas are a marker for Brooke-Spiegler syndrome. A desmoplastic trichoepithelioma is a variant that has stromal sclerosis on pathology. It is a benign lesion, although may be difficult to differentiate from sclerosing basal cell or microcystic adnexal carcinoma.
Angiofibroma, or fibrous papule, is a commonly seen, benign, skin-colored papule also often occurring on the nose. They can be treated for cosmetic purposes. Multiple lesions are associated with tuberous sclerosis.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at edermatologynews.com. To submit a case for possible publication, send an email to [email protected].
Review finds some therapies show promise in managing prurigo nodularis
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Recent success in the use of (PN) suggests that safer and more effective systemic therapies that provide relief and management of this debilitating chronic skin condition are possible, reported Azam A. Qureshi, BA, of the department of dermatology, George Washington University, Washington, and his coauthors.
Their report was published in the Journal of the American Academy of Dermatology.
They conducted a systematic review of 35 clinical studies of different therapies for PN, published between Jan. 1, 1990, and March 22, 2018, using PubMed and Scopus databases. Their goal was twofold: to provide a summary of current evidence-based therapies and their corresponding level of evidence ratings, and to help researchers “identify gaps in PN treatment development and study.”
From 706 studies, the authors selected 35 clinical studies of treatment strategies for PN, for which the pathogenesis is virtually unknown, they noted. The studies included 15 prospective cohort studies, 11 retrospective reviews, 8 randomized controlled trials (with 10-127 patients), and 1 case series. Studies that failed to report treatment outcomes and those with fewer than five patients were excluded from the review.
Treatment modalities included topical agents, phototherapy and photochemotherapy, thalidomide, systemic immunomodulatory drugs, antiepileptics and antidepressants, and emerging treatment approaches. Many of the treatments evaluated in the review were found to offer limited promise for clinical application as a result of low efficacy or frequent side effects. The authors attributed the overall lack of success with treatments to the “heterogeneous nature of the etiology of chronic pruritus.”
Thalidomide was found to have limited use given its poor safety profile; lenalidomide achieved better outcomes, but treatment in one case was stopped because of possible drug-induced neuropathy or myopathy, they wrote. The systemic immunomodulatory agents methotrexate and cyclosporine exhibited successful treatment but also were found to have poor safety profiles. Greater promise was seen with antiepileptics and antidepressants, which were associated with fewer side effects. “Antidepressants, such as mirtazapine and amitriptyline, and antiepileptics, such as gabapentin and pregabalin, can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” senior author Adam Friedman, MD, professor of dermatology at George Washington University, said in an interview.
Among the most positive outcomes achieved were those in which promising newer treatments, including targeting IL-31 signaling and opioid receptor modulation, were used. In particular, the authors cited nemolizumab, an IL-31 receptor A antagonist, which is currently in a phase 2 trial of PN. Beneficial effects of extended release nalbuphine, an opioid k-receptor agonist and mu-receptor antagonist, were found in an unpublished multicenter, double-blind randomized controlled trial, with no serious adverse effects attributed to treatment.
Among the emerging treatments are the neurokinin-1 receptor antagonists, which include serlopitant, which showed beneficial effects compared with placebo in an 8-week randomized controlled study, they wrote. The neurokinin-1 receptor “is a target of substance P, a mediator of itch and a probable pathogenic agent in PN,” they wrote. “Binding by these agents likely disrupts substance P signaling, thereby halting PN pathogenesis,” they added.
“Antidepressants such as mirtazapine and amitriptyline and antiepileptics such as gabapentin and pregabalin can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” Dr. Friedman said in the interview.
Mr. Qureshi had no relevant financial disclosures. Dr. Friedman has received honoraria for participation in advisory boards for Menlo Therapeutics, and serves as an investigator for Kiniksa Pharmaceuticals. A third author received honoraria for participation in advisory boards for Menlo, and several other companies, and received grant/research support as an investigator from Menlo, Kiniksa, and several other companies.
SOURCE: Qureshi AA et al. J Am Acad Dermatol. 2018. doi: 10.1016/j.jaad.2018.09.020.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Recent success in the use of (PN) suggests that safer and more effective systemic therapies that provide relief and management of this debilitating chronic skin condition are possible, reported Azam A. Qureshi, BA, of the department of dermatology, George Washington University, Washington, and his coauthors.
Their report was published in the Journal of the American Academy of Dermatology.
They conducted a systematic review of 35 clinical studies of different therapies for PN, published between Jan. 1, 1990, and March 22, 2018, using PubMed and Scopus databases. Their goal was twofold: to provide a summary of current evidence-based therapies and their corresponding level of evidence ratings, and to help researchers “identify gaps in PN treatment development and study.”
From 706 studies, the authors selected 35 clinical studies of treatment strategies for PN, for which the pathogenesis is virtually unknown, they noted. The studies included 15 prospective cohort studies, 11 retrospective reviews, 8 randomized controlled trials (with 10-127 patients), and 1 case series. Studies that failed to report treatment outcomes and those with fewer than five patients were excluded from the review.
Treatment modalities included topical agents, phototherapy and photochemotherapy, thalidomide, systemic immunomodulatory drugs, antiepileptics and antidepressants, and emerging treatment approaches. Many of the treatments evaluated in the review were found to offer limited promise for clinical application as a result of low efficacy or frequent side effects. The authors attributed the overall lack of success with treatments to the “heterogeneous nature of the etiology of chronic pruritus.”
Thalidomide was found to have limited use given its poor safety profile; lenalidomide achieved better outcomes, but treatment in one case was stopped because of possible drug-induced neuropathy or myopathy, they wrote. The systemic immunomodulatory agents methotrexate and cyclosporine exhibited successful treatment but also were found to have poor safety profiles. Greater promise was seen with antiepileptics and antidepressants, which were associated with fewer side effects. “Antidepressants, such as mirtazapine and amitriptyline, and antiepileptics, such as gabapentin and pregabalin, can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” senior author Adam Friedman, MD, professor of dermatology at George Washington University, said in an interview.
Among the most positive outcomes achieved were those in which promising newer treatments, including targeting IL-31 signaling and opioid receptor modulation, were used. In particular, the authors cited nemolizumab, an IL-31 receptor A antagonist, which is currently in a phase 2 trial of PN. Beneficial effects of extended release nalbuphine, an opioid k-receptor agonist and mu-receptor antagonist, were found in an unpublished multicenter, double-blind randomized controlled trial, with no serious adverse effects attributed to treatment.
Among the emerging treatments are the neurokinin-1 receptor antagonists, which include serlopitant, which showed beneficial effects compared with placebo in an 8-week randomized controlled study, they wrote. The neurokinin-1 receptor “is a target of substance P, a mediator of itch and a probable pathogenic agent in PN,” they wrote. “Binding by these agents likely disrupts substance P signaling, thereby halting PN pathogenesis,” they added.
“Antidepressants such as mirtazapine and amitriptyline and antiepileptics such as gabapentin and pregabalin can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” Dr. Friedman said in the interview.
Mr. Qureshi had no relevant financial disclosures. Dr. Friedman has received honoraria for participation in advisory boards for Menlo Therapeutics, and serves as an investigator for Kiniksa Pharmaceuticals. A third author received honoraria for participation in advisory boards for Menlo, and several other companies, and received grant/research support as an investigator from Menlo, Kiniksa, and several other companies.
SOURCE: Qureshi AA et al. J Am Acad Dermatol. 2018. doi: 10.1016/j.jaad.2018.09.020.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Recent success in the use of (PN) suggests that safer and more effective systemic therapies that provide relief and management of this debilitating chronic skin condition are possible, reported Azam A. Qureshi, BA, of the department of dermatology, George Washington University, Washington, and his coauthors.
Their report was published in the Journal of the American Academy of Dermatology.
They conducted a systematic review of 35 clinical studies of different therapies for PN, published between Jan. 1, 1990, and March 22, 2018, using PubMed and Scopus databases. Their goal was twofold: to provide a summary of current evidence-based therapies and their corresponding level of evidence ratings, and to help researchers “identify gaps in PN treatment development and study.”
From 706 studies, the authors selected 35 clinical studies of treatment strategies for PN, for which the pathogenesis is virtually unknown, they noted. The studies included 15 prospective cohort studies, 11 retrospective reviews, 8 randomized controlled trials (with 10-127 patients), and 1 case series. Studies that failed to report treatment outcomes and those with fewer than five patients were excluded from the review.
Treatment modalities included topical agents, phototherapy and photochemotherapy, thalidomide, systemic immunomodulatory drugs, antiepileptics and antidepressants, and emerging treatment approaches. Many of the treatments evaluated in the review were found to offer limited promise for clinical application as a result of low efficacy or frequent side effects. The authors attributed the overall lack of success with treatments to the “heterogeneous nature of the etiology of chronic pruritus.”
Thalidomide was found to have limited use given its poor safety profile; lenalidomide achieved better outcomes, but treatment in one case was stopped because of possible drug-induced neuropathy or myopathy, they wrote. The systemic immunomodulatory agents methotrexate and cyclosporine exhibited successful treatment but also were found to have poor safety profiles. Greater promise was seen with antiepileptics and antidepressants, which were associated with fewer side effects. “Antidepressants, such as mirtazapine and amitriptyline, and antiepileptics, such as gabapentin and pregabalin, can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” senior author Adam Friedman, MD, professor of dermatology at George Washington University, said in an interview.
Among the most positive outcomes achieved were those in which promising newer treatments, including targeting IL-31 signaling and opioid receptor modulation, were used. In particular, the authors cited nemolizumab, an IL-31 receptor A antagonist, which is currently in a phase 2 trial of PN. Beneficial effects of extended release nalbuphine, an opioid k-receptor agonist and mu-receptor antagonist, were found in an unpublished multicenter, double-blind randomized controlled trial, with no serious adverse effects attributed to treatment.
Among the emerging treatments are the neurokinin-1 receptor antagonists, which include serlopitant, which showed beneficial effects compared with placebo in an 8-week randomized controlled study, they wrote. The neurokinin-1 receptor “is a target of substance P, a mediator of itch and a probable pathogenic agent in PN,” they wrote. “Binding by these agents likely disrupts substance P signaling, thereby halting PN pathogenesis,” they added.
“Antidepressants such as mirtazapine and amitriptyline and antiepileptics such as gabapentin and pregabalin can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” Dr. Friedman said in the interview.
Mr. Qureshi had no relevant financial disclosures. Dr. Friedman has received honoraria for participation in advisory boards for Menlo Therapeutics, and serves as an investigator for Kiniksa Pharmaceuticals. A third author received honoraria for participation in advisory boards for Menlo, and several other companies, and received grant/research support as an investigator from Menlo, Kiniksa, and several other companies.
SOURCE: Qureshi AA et al. J Am Acad Dermatol. 2018. doi: 10.1016/j.jaad.2018.09.020.
Key clinical point: Additional research is needed to develop more individualized treatment strategies, but data for some treatments are promising.
Major finding: Treatments identified as having beneficial effects in the review included nemolizumab, an IL-31 receptor A antagonist; nalbuphine, an opioid k-receptor agonist and mu-receptor antagonist, and serlopitant, a neurokinin-1 receptor antagonist.
Study details: A systematic review of 35 studies evaluating treatments for PN, published between Jan. 1, 1990, and March 22, 2018.
Disclosures: Mr. Qureshi had no relevant financial disclosures. Dr. Friedman has received honoraria for participation in advisory boards for Menlo Therapeutics, and serves as an investigator for Kiniksa Pharmaceuticals. A third author received honoraria for participation in advisory boards for Menlo, and several other companies, and received grant/research support as an investigator from Menlo, Kiniksa, and several other companies.
Source: Qureshi AA et al. J Am Acad Dermatol. 2018. doi: 10.1016/j.jaad.2018.09.020.