User login
EC approves dasatinib plus chemo for kids with Ph+ ALL
The European Commission has approved dasatinib (Sprycel) for use in combination with chemotherapy for the treatment of pediatric patients with newly diagnosed, Philadelphia chromosome–positive (Ph+) acute lymphoblastic leukemia (ALL).
Dasatinib will be available in tablet form and as a powder for oral suspension, Bristol-Myers Squib said in a press release.
The approval was based on an event-free survival rate of 65.5% (95% confidence interval, 57.7-73.7) and an overall survival rate of 91.5% (95% CI, 84.2-95.5) in a phase 2 trial that evaluated the addition of dasatinib to a chemotherapy regimen modeled on a Berlin-Frankfurt-Münster high-risk backbone in pediatric patients with newly diagnosed Ph+ ALL.
Patients treated in the study (n = 106) were all aged younger than 18 years and received dasatinib at a daily dose of 60 mg/m2 on a continuous dosing regimen for up to 24 months, in combination with chemotherapy. About 77 % of patients (n = 82) received tablets exclusively; 23% of patients (n = 24) received the powder for oral suspension at least once.
Hematologic adverse events included grade 3 or 4 febrile neutropenia (75.5%), sepsis (23.6%), and bacteremia (24.5%). Nonhematologic, noninfectious grade 3 or 4 adverse events attributed to dasatinib and reported in more than 10% of patients included elevated ALT (21.7%) and AST (10.4%). Additional grade 3 or 4 adverse events attributed to dasatinib were pleural effusion (3.8%), edema (2.8%), hemorrhage (5.7%), and cardiac failure (0.8%). No events of pulmonary hypertension or pulmonary arterial hypertension were reported, the company said in the press release.
Dasatinib is already approved by the European Commission to treat children with Ph+ chronic myeloid leukemia in the chronic phase, which includes newly diagnosed patients and those with resistance or intolerance to imatinib.
The European Commission has approved dasatinib (Sprycel) for use in combination with chemotherapy for the treatment of pediatric patients with newly diagnosed, Philadelphia chromosome–positive (Ph+) acute lymphoblastic leukemia (ALL).
Dasatinib will be available in tablet form and as a powder for oral suspension, Bristol-Myers Squib said in a press release.
The approval was based on an event-free survival rate of 65.5% (95% confidence interval, 57.7-73.7) and an overall survival rate of 91.5% (95% CI, 84.2-95.5) in a phase 2 trial that evaluated the addition of dasatinib to a chemotherapy regimen modeled on a Berlin-Frankfurt-Münster high-risk backbone in pediatric patients with newly diagnosed Ph+ ALL.
Patients treated in the study (n = 106) were all aged younger than 18 years and received dasatinib at a daily dose of 60 mg/m2 on a continuous dosing regimen for up to 24 months, in combination with chemotherapy. About 77 % of patients (n = 82) received tablets exclusively; 23% of patients (n = 24) received the powder for oral suspension at least once.
Hematologic adverse events included grade 3 or 4 febrile neutropenia (75.5%), sepsis (23.6%), and bacteremia (24.5%). Nonhematologic, noninfectious grade 3 or 4 adverse events attributed to dasatinib and reported in more than 10% of patients included elevated ALT (21.7%) and AST (10.4%). Additional grade 3 or 4 adverse events attributed to dasatinib were pleural effusion (3.8%), edema (2.8%), hemorrhage (5.7%), and cardiac failure (0.8%). No events of pulmonary hypertension or pulmonary arterial hypertension were reported, the company said in the press release.
Dasatinib is already approved by the European Commission to treat children with Ph+ chronic myeloid leukemia in the chronic phase, which includes newly diagnosed patients and those with resistance or intolerance to imatinib.
The European Commission has approved dasatinib (Sprycel) for use in combination with chemotherapy for the treatment of pediatric patients with newly diagnosed, Philadelphia chromosome–positive (Ph+) acute lymphoblastic leukemia (ALL).
Dasatinib will be available in tablet form and as a powder for oral suspension, Bristol-Myers Squib said in a press release.
The approval was based on an event-free survival rate of 65.5% (95% confidence interval, 57.7-73.7) and an overall survival rate of 91.5% (95% CI, 84.2-95.5) in a phase 2 trial that evaluated the addition of dasatinib to a chemotherapy regimen modeled on a Berlin-Frankfurt-Münster high-risk backbone in pediatric patients with newly diagnosed Ph+ ALL.
Patients treated in the study (n = 106) were all aged younger than 18 years and received dasatinib at a daily dose of 60 mg/m2 on a continuous dosing regimen for up to 24 months, in combination with chemotherapy. About 77 % of patients (n = 82) received tablets exclusively; 23% of patients (n = 24) received the powder for oral suspension at least once.
Hematologic adverse events included grade 3 or 4 febrile neutropenia (75.5%), sepsis (23.6%), and bacteremia (24.5%). Nonhematologic, noninfectious grade 3 or 4 adverse events attributed to dasatinib and reported in more than 10% of patients included elevated ALT (21.7%) and AST (10.4%). Additional grade 3 or 4 adverse events attributed to dasatinib were pleural effusion (3.8%), edema (2.8%), hemorrhage (5.7%), and cardiac failure (0.8%). No events of pulmonary hypertension or pulmonary arterial hypertension were reported, the company said in the press release.
Dasatinib is already approved by the European Commission to treat children with Ph+ chronic myeloid leukemia in the chronic phase, which includes newly diagnosed patients and those with resistance or intolerance to imatinib.
Eltrombopag ‘cannot be recommended’ during AML induction
The thrombopoietic agent eltrombopag (Promacta) did more harm than good when given to adults with acute myeloid leukemia (AML) during standard induction chemotherapy, results of a randomized phase 2 trial show.
Patients who were randomly assigned to receive standard induction chemotherapy with daunorubicin and cytarabine plus eltrombopag had a higher incidence of serious adverse events and death from hemorrhage within 30 days of the last eltrombopag dose, compared with patients who received chemotherapy and placebo, reported Noelle Frey, MD, from the University of Pennsylvania in Philadelphia, and colleagues.
“Overall survival was also numerically longer in the placebo group, compared with the eltrombopag group. It remains unclear why there were more deaths, particularly due to hemorrhage within 30 days after the last dose of treatment, in the eltrombopag group,” they wrote in the Lancet Haematology.
The investigators had expected better results, based on eltrombopag’s demonstrated efficacy against thrombocytopenia (a common feature of AML, exacerbated by chemotherapy), and because of evidence suggesting that the thrombopoietin-receptor agonist might also have antileukemic properties.
They set out to test the safety, tolerability, and efficacy of eltrombopag added to standard induction therapy in patients with treatment-naive AML of any subtype except M3 (acute promyelocytic leukemia) or M7 (acute megakaryocytic leukemia).
Patients received chemotherapy with daunorubicin in a bolus intravenous infusion at a dose of 90 mg/m2 on days 1-3 for patients 18-60 years of age, or 60 mg/m2 for patients older than 60 years, plus cytarabine continuous intravenous infusion at a dose of 100 mg/m2 on days 1-7. The 148 patients were randomized in groups of 74 each to receive either eltrombopag 200 mg (100 mg for patients of east Asian heritage) or placebo, once daily.
Eltrombopag was continued until platelet counts were 200 × 109/L or higher, remission, or 42 days after the start of induction chemotherapy.
Grade 3 or 4 adverse events occurring in 10% or more of patients – a primary endpoint – were febrile neutropenia, which occurred in 42% of patients receiving eltrombopag, compared with 39% receiving placebo, decreased white blood cell count in 11% vs. 7%, and hypophosphatemia in 4% and 13%, respectively,
Serious adverse events occurred in 34% of patients on eltrombopag, compared with 20% on placebo. Similarly, 53% of patients receiving eltrombopag died, compared with 41% of patients receiving the placebo.
Most of the deaths were attributable to AML, including 19 patients (26%) on eltrombopag and 10 (14%) on placebo. Eleven patients on eltrombopag and four on placebo died within 30 days of the last dose of study treatment.
Hemorrhage accounted for the deaths of five patients on eltrombopag and three on placebo, and sepsis accounted for the deaths of five and six patients, respectively.
Both the incidence of thromboembolic events and mean change in left ventricular ejection fraction were similar between the groups.
Median overall survival was 15.4 months in the eltrombopag group vs. 25.7 months in the placebo group, although this difference was not statistically significant, likely because of the sample size.
The investigators were at a loss to explain why the eltrombopag-treated patients had numerically worse outcomes.
“In the present study, eltrombopag did not improve the time to platelet recovery or the incidences of grade 3-4 thrombocytopenia, neutropenia, or anemia, compared with placebo. Furthermore, the study did not reveal any differences in investigator-assessed response to treatment. These findings were unexpected given outcomes from previous studies of eltrombopag monotherapy in patients with myelodysplastic syndromes or acute myeloid leukemia,” they wrote.
Although the reasons behind the findings are unclear, “the data from this trial do not support a favorable benefit-risk profile for eltrombopag in combination with induction chemotherapy in patients with acute myeloid leukemia,” the investigators wrote.
The study was funded by Novartis. Dr. Frey reported nonfinancial support from Novartis during the conduct of the study and consultancy fees from Novartis outside of the submitted work. Multiple coauthors reported similar relationships with Novartis and/or other companies.
SOURCE: Frey N et al. Lancet Haematol. 2019 Jan 28. doi: 10.1016/S2352-3026(18)30231-X.
The thrombopoietic agent eltrombopag (Promacta) did more harm than good when given to adults with acute myeloid leukemia (AML) during standard induction chemotherapy, results of a randomized phase 2 trial show.
Patients who were randomly assigned to receive standard induction chemotherapy with daunorubicin and cytarabine plus eltrombopag had a higher incidence of serious adverse events and death from hemorrhage within 30 days of the last eltrombopag dose, compared with patients who received chemotherapy and placebo, reported Noelle Frey, MD, from the University of Pennsylvania in Philadelphia, and colleagues.
“Overall survival was also numerically longer in the placebo group, compared with the eltrombopag group. It remains unclear why there were more deaths, particularly due to hemorrhage within 30 days after the last dose of treatment, in the eltrombopag group,” they wrote in the Lancet Haematology.
The investigators had expected better results, based on eltrombopag’s demonstrated efficacy against thrombocytopenia (a common feature of AML, exacerbated by chemotherapy), and because of evidence suggesting that the thrombopoietin-receptor agonist might also have antileukemic properties.
They set out to test the safety, tolerability, and efficacy of eltrombopag added to standard induction therapy in patients with treatment-naive AML of any subtype except M3 (acute promyelocytic leukemia) or M7 (acute megakaryocytic leukemia).
Patients received chemotherapy with daunorubicin in a bolus intravenous infusion at a dose of 90 mg/m2 on days 1-3 for patients 18-60 years of age, or 60 mg/m2 for patients older than 60 years, plus cytarabine continuous intravenous infusion at a dose of 100 mg/m2 on days 1-7. The 148 patients were randomized in groups of 74 each to receive either eltrombopag 200 mg (100 mg for patients of east Asian heritage) or placebo, once daily.
Eltrombopag was continued until platelet counts were 200 × 109/L or higher, remission, or 42 days after the start of induction chemotherapy.
Grade 3 or 4 adverse events occurring in 10% or more of patients – a primary endpoint – were febrile neutropenia, which occurred in 42% of patients receiving eltrombopag, compared with 39% receiving placebo, decreased white blood cell count in 11% vs. 7%, and hypophosphatemia in 4% and 13%, respectively,
Serious adverse events occurred in 34% of patients on eltrombopag, compared with 20% on placebo. Similarly, 53% of patients receiving eltrombopag died, compared with 41% of patients receiving the placebo.
Most of the deaths were attributable to AML, including 19 patients (26%) on eltrombopag and 10 (14%) on placebo. Eleven patients on eltrombopag and four on placebo died within 30 days of the last dose of study treatment.
Hemorrhage accounted for the deaths of five patients on eltrombopag and three on placebo, and sepsis accounted for the deaths of five and six patients, respectively.
Both the incidence of thromboembolic events and mean change in left ventricular ejection fraction were similar between the groups.
Median overall survival was 15.4 months in the eltrombopag group vs. 25.7 months in the placebo group, although this difference was not statistically significant, likely because of the sample size.
The investigators were at a loss to explain why the eltrombopag-treated patients had numerically worse outcomes.
“In the present study, eltrombopag did not improve the time to platelet recovery or the incidences of grade 3-4 thrombocytopenia, neutropenia, or anemia, compared with placebo. Furthermore, the study did not reveal any differences in investigator-assessed response to treatment. These findings were unexpected given outcomes from previous studies of eltrombopag monotherapy in patients with myelodysplastic syndromes or acute myeloid leukemia,” they wrote.
Although the reasons behind the findings are unclear, “the data from this trial do not support a favorable benefit-risk profile for eltrombopag in combination with induction chemotherapy in patients with acute myeloid leukemia,” the investigators wrote.
The study was funded by Novartis. Dr. Frey reported nonfinancial support from Novartis during the conduct of the study and consultancy fees from Novartis outside of the submitted work. Multiple coauthors reported similar relationships with Novartis and/or other companies.
SOURCE: Frey N et al. Lancet Haematol. 2019 Jan 28. doi: 10.1016/S2352-3026(18)30231-X.
The thrombopoietic agent eltrombopag (Promacta) did more harm than good when given to adults with acute myeloid leukemia (AML) during standard induction chemotherapy, results of a randomized phase 2 trial show.
Patients who were randomly assigned to receive standard induction chemotherapy with daunorubicin and cytarabine plus eltrombopag had a higher incidence of serious adverse events and death from hemorrhage within 30 days of the last eltrombopag dose, compared with patients who received chemotherapy and placebo, reported Noelle Frey, MD, from the University of Pennsylvania in Philadelphia, and colleagues.
“Overall survival was also numerically longer in the placebo group, compared with the eltrombopag group. It remains unclear why there were more deaths, particularly due to hemorrhage within 30 days after the last dose of treatment, in the eltrombopag group,” they wrote in the Lancet Haematology.
The investigators had expected better results, based on eltrombopag’s demonstrated efficacy against thrombocytopenia (a common feature of AML, exacerbated by chemotherapy), and because of evidence suggesting that the thrombopoietin-receptor agonist might also have antileukemic properties.
They set out to test the safety, tolerability, and efficacy of eltrombopag added to standard induction therapy in patients with treatment-naive AML of any subtype except M3 (acute promyelocytic leukemia) or M7 (acute megakaryocytic leukemia).
Patients received chemotherapy with daunorubicin in a bolus intravenous infusion at a dose of 90 mg/m2 on days 1-3 for patients 18-60 years of age, or 60 mg/m2 for patients older than 60 years, plus cytarabine continuous intravenous infusion at a dose of 100 mg/m2 on days 1-7. The 148 patients were randomized in groups of 74 each to receive either eltrombopag 200 mg (100 mg for patients of east Asian heritage) or placebo, once daily.
Eltrombopag was continued until platelet counts were 200 × 109/L or higher, remission, or 42 days after the start of induction chemotherapy.
Grade 3 or 4 adverse events occurring in 10% or more of patients – a primary endpoint – were febrile neutropenia, which occurred in 42% of patients receiving eltrombopag, compared with 39% receiving placebo, decreased white blood cell count in 11% vs. 7%, and hypophosphatemia in 4% and 13%, respectively,
Serious adverse events occurred in 34% of patients on eltrombopag, compared with 20% on placebo. Similarly, 53% of patients receiving eltrombopag died, compared with 41% of patients receiving the placebo.
Most of the deaths were attributable to AML, including 19 patients (26%) on eltrombopag and 10 (14%) on placebo. Eleven patients on eltrombopag and four on placebo died within 30 days of the last dose of study treatment.
Hemorrhage accounted for the deaths of five patients on eltrombopag and three on placebo, and sepsis accounted for the deaths of five and six patients, respectively.
Both the incidence of thromboembolic events and mean change in left ventricular ejection fraction were similar between the groups.
Median overall survival was 15.4 months in the eltrombopag group vs. 25.7 months in the placebo group, although this difference was not statistically significant, likely because of the sample size.
The investigators were at a loss to explain why the eltrombopag-treated patients had numerically worse outcomes.
“In the present study, eltrombopag did not improve the time to platelet recovery or the incidences of grade 3-4 thrombocytopenia, neutropenia, or anemia, compared with placebo. Furthermore, the study did not reveal any differences in investigator-assessed response to treatment. These findings were unexpected given outcomes from previous studies of eltrombopag monotherapy in patients with myelodysplastic syndromes or acute myeloid leukemia,” they wrote.
Although the reasons behind the findings are unclear, “the data from this trial do not support a favorable benefit-risk profile for eltrombopag in combination with induction chemotherapy in patients with acute myeloid leukemia,” the investigators wrote.
The study was funded by Novartis. Dr. Frey reported nonfinancial support from Novartis during the conduct of the study and consultancy fees from Novartis outside of the submitted work. Multiple coauthors reported similar relationships with Novartis and/or other companies.
SOURCE: Frey N et al. Lancet Haematol. 2019 Jan 28. doi: 10.1016/S2352-3026(18)30231-X.
FROM LANCET HAEMATOLOGY
Key clinical point:
Major finding: Overall survival was shorter for patients assigned to eltrombopag than placebo, at 15.4 months versus 25.7 months. The difference was not statistically significant.
Study details: Randomized phase 2 trial in 148 adults with treatment-naive acute myeloid leukemia.
Disclosures: The study was funded by Novartis. Dr. Frey reported nonfinancial support from Novartis during the conduct of the study and consultancy fees from Novartis outside of the submitted work. Multiple coauthors reported similar relationships with Novartis and other companies.
Source: Frey N et al. Lancet Haematol. 2019 Jan 28. doi: 10.1016/S2352-3026(18)30231-X.
PD-1 blockade plus CD19 CAR T boosts CAR T-cell persistence
SAN DIEGO – Checkpoint inhibition can be used safely and effectively with CD19-directed chimeric antigen receptor (CAR) T-cell therapy in children with relapsed B-cell acute lymphoblastic leukemia (ALL), and it may bolster CAR T-cell effects and persistence, suggest the findings in a series of 14 patients at the Children’s Hospital of Philadelphia.
Combined programmed death-1 (PD-1) blockade and CAR T-cell therapy appeared to have particular benefit in patients with early B-cell recovery and in those with bulky extramedullary disease, Shannon Maude, MD, PhD, reported during a press conference at the annual meeting of the American Society of Hematology.
The patients, aged 4-17 years with heavily pretreated relapsed B-ALL (13 patients) or B lymphoblastic lymphoma (1 patient), were treated with CD19-directed CAR T-cell therapy, including CTL019 in 4 patients and CTL119 in 10 patients, followed by pembrolizumab (in 13 patients) or nivolumab (in 1 patient).
Six patients received the combination therapy because of early B-cell recovery after initial CAR T-cell infusion, four patients had relapsed or refractory (R/R) bulky extramedullary disease, and four patients had failed to respond or relapsed after initial CAR T-cell therapy.
Three of the six with poor persistence of response reestablished B-cell aplasia (a reflection of CAR T-cell function) after reinfusion of the CAR T-cell product followed by infusion with PD-1 blockade, and they have “sustained CR [complete response] with B-cell aplasia, showing continued persistence of their CAR T cells,” said Dr. Maude, an attending physician in the Cancer Center at Children’s Hospital of Philadelphia.
Of the four patients with R/R bulky extramedullary disease, two patients had a partial response and two patients had CR, she said, explaining that it was hypothesized that the “PD-1 checkpoint pathway may be activated through the microenvironment in that extramedullary situation.”
However, all four patients who had partial or no response to initial CAR T-cell therapy progressed after PD-1 administration, she said, noting that “in one patient, this progression was marked by reduced CD19 expression, which was probably the mode of escape from CD19 CAR T cells.”
Prior studies have shown that patients who respond to CAR T-cell therapy have persistence of CD19 CAR T cells, whereas those with loss of CD19 CAR T cells within 6 months of infusion have a higher rate of relapse, Dr. Maude explained.
“Our hypothesis was that T cells, upon activation, may become exhausted through activation of immune checkpoint pathways, that one such pathway – PD-1 – may be involved in early loss of CD19 CAR T cells and therefore that the combination [of CD19 CAR T-cell therapy] with PD-1 checkpoint blockade may improve the function of the CAR T cells and their persistence,” she said.
The combined approach was well tolerated in this study, she said, noting that mild cytokine release syndrome symptoms and fever typical of CAR T-cell proliferative responses were observed in three patients within 2 days of starting pembrolizumab.
Other adverse effects associated with PD-1 inhibition, including acute pancreatitis, hypothyroidism, arthralgias, and urticaria, occurred in one patient each. There were four cases of grade 3-4 cytopenias that were deemed tolerable or reversible upon discontinuation.
“We show that PD-1 checkpoint inhibitors can be safely combined with CD19 CAR T-cell therapy and that this mechanism may be useful to improve CAR T-cell persistence,” Dr. Maude said.
These findings, which showed particular benefit in patients with poor persistence marked by early B-cell recovery and in those with R/R bulky extramedullary disease, should help inform future use of checkpoint inhibitors after CAR T-cell therapy, she added.
Dr. Maude reported financial ties to Novartis.
SOURCE: Li AM et al. ASH 2018, Abstract 556.
SAN DIEGO – Checkpoint inhibition can be used safely and effectively with CD19-directed chimeric antigen receptor (CAR) T-cell therapy in children with relapsed B-cell acute lymphoblastic leukemia (ALL), and it may bolster CAR T-cell effects and persistence, suggest the findings in a series of 14 patients at the Children’s Hospital of Philadelphia.
Combined programmed death-1 (PD-1) blockade and CAR T-cell therapy appeared to have particular benefit in patients with early B-cell recovery and in those with bulky extramedullary disease, Shannon Maude, MD, PhD, reported during a press conference at the annual meeting of the American Society of Hematology.
The patients, aged 4-17 years with heavily pretreated relapsed B-ALL (13 patients) or B lymphoblastic lymphoma (1 patient), were treated with CD19-directed CAR T-cell therapy, including CTL019 in 4 patients and CTL119 in 10 patients, followed by pembrolizumab (in 13 patients) or nivolumab (in 1 patient).
Six patients received the combination therapy because of early B-cell recovery after initial CAR T-cell infusion, four patients had relapsed or refractory (R/R) bulky extramedullary disease, and four patients had failed to respond or relapsed after initial CAR T-cell therapy.
Three of the six with poor persistence of response reestablished B-cell aplasia (a reflection of CAR T-cell function) after reinfusion of the CAR T-cell product followed by infusion with PD-1 blockade, and they have “sustained CR [complete response] with B-cell aplasia, showing continued persistence of their CAR T cells,” said Dr. Maude, an attending physician in the Cancer Center at Children’s Hospital of Philadelphia.
Of the four patients with R/R bulky extramedullary disease, two patients had a partial response and two patients had CR, she said, explaining that it was hypothesized that the “PD-1 checkpoint pathway may be activated through the microenvironment in that extramedullary situation.”
However, all four patients who had partial or no response to initial CAR T-cell therapy progressed after PD-1 administration, she said, noting that “in one patient, this progression was marked by reduced CD19 expression, which was probably the mode of escape from CD19 CAR T cells.”
Prior studies have shown that patients who respond to CAR T-cell therapy have persistence of CD19 CAR T cells, whereas those with loss of CD19 CAR T cells within 6 months of infusion have a higher rate of relapse, Dr. Maude explained.
“Our hypothesis was that T cells, upon activation, may become exhausted through activation of immune checkpoint pathways, that one such pathway – PD-1 – may be involved in early loss of CD19 CAR T cells and therefore that the combination [of CD19 CAR T-cell therapy] with PD-1 checkpoint blockade may improve the function of the CAR T cells and their persistence,” she said.
The combined approach was well tolerated in this study, she said, noting that mild cytokine release syndrome symptoms and fever typical of CAR T-cell proliferative responses were observed in three patients within 2 days of starting pembrolizumab.
Other adverse effects associated with PD-1 inhibition, including acute pancreatitis, hypothyroidism, arthralgias, and urticaria, occurred in one patient each. There were four cases of grade 3-4 cytopenias that were deemed tolerable or reversible upon discontinuation.
“We show that PD-1 checkpoint inhibitors can be safely combined with CD19 CAR T-cell therapy and that this mechanism may be useful to improve CAR T-cell persistence,” Dr. Maude said.
These findings, which showed particular benefit in patients with poor persistence marked by early B-cell recovery and in those with R/R bulky extramedullary disease, should help inform future use of checkpoint inhibitors after CAR T-cell therapy, she added.
Dr. Maude reported financial ties to Novartis.
SOURCE: Li AM et al. ASH 2018, Abstract 556.
SAN DIEGO – Checkpoint inhibition can be used safely and effectively with CD19-directed chimeric antigen receptor (CAR) T-cell therapy in children with relapsed B-cell acute lymphoblastic leukemia (ALL), and it may bolster CAR T-cell effects and persistence, suggest the findings in a series of 14 patients at the Children’s Hospital of Philadelphia.
Combined programmed death-1 (PD-1) blockade and CAR T-cell therapy appeared to have particular benefit in patients with early B-cell recovery and in those with bulky extramedullary disease, Shannon Maude, MD, PhD, reported during a press conference at the annual meeting of the American Society of Hematology.
The patients, aged 4-17 years with heavily pretreated relapsed B-ALL (13 patients) or B lymphoblastic lymphoma (1 patient), were treated with CD19-directed CAR T-cell therapy, including CTL019 in 4 patients and CTL119 in 10 patients, followed by pembrolizumab (in 13 patients) or nivolumab (in 1 patient).
Six patients received the combination therapy because of early B-cell recovery after initial CAR T-cell infusion, four patients had relapsed or refractory (R/R) bulky extramedullary disease, and four patients had failed to respond or relapsed after initial CAR T-cell therapy.
Three of the six with poor persistence of response reestablished B-cell aplasia (a reflection of CAR T-cell function) after reinfusion of the CAR T-cell product followed by infusion with PD-1 blockade, and they have “sustained CR [complete response] with B-cell aplasia, showing continued persistence of their CAR T cells,” said Dr. Maude, an attending physician in the Cancer Center at Children’s Hospital of Philadelphia.
Of the four patients with R/R bulky extramedullary disease, two patients had a partial response and two patients had CR, she said, explaining that it was hypothesized that the “PD-1 checkpoint pathway may be activated through the microenvironment in that extramedullary situation.”
However, all four patients who had partial or no response to initial CAR T-cell therapy progressed after PD-1 administration, she said, noting that “in one patient, this progression was marked by reduced CD19 expression, which was probably the mode of escape from CD19 CAR T cells.”
Prior studies have shown that patients who respond to CAR T-cell therapy have persistence of CD19 CAR T cells, whereas those with loss of CD19 CAR T cells within 6 months of infusion have a higher rate of relapse, Dr. Maude explained.
“Our hypothesis was that T cells, upon activation, may become exhausted through activation of immune checkpoint pathways, that one such pathway – PD-1 – may be involved in early loss of CD19 CAR T cells and therefore that the combination [of CD19 CAR T-cell therapy] with PD-1 checkpoint blockade may improve the function of the CAR T cells and their persistence,” she said.
The combined approach was well tolerated in this study, she said, noting that mild cytokine release syndrome symptoms and fever typical of CAR T-cell proliferative responses were observed in three patients within 2 days of starting pembrolizumab.
Other adverse effects associated with PD-1 inhibition, including acute pancreatitis, hypothyroidism, arthralgias, and urticaria, occurred in one patient each. There were four cases of grade 3-4 cytopenias that were deemed tolerable or reversible upon discontinuation.
“We show that PD-1 checkpoint inhibitors can be safely combined with CD19 CAR T-cell therapy and that this mechanism may be useful to improve CAR T-cell persistence,” Dr. Maude said.
These findings, which showed particular benefit in patients with poor persistence marked by early B-cell recovery and in those with R/R bulky extramedullary disease, should help inform future use of checkpoint inhibitors after CAR T-cell therapy, she added.
Dr. Maude reported financial ties to Novartis.
SOURCE: Li AM et al. ASH 2018, Abstract 556.
REPORTING FROM ASH 2018
Key clinical point: Major finding: Three of six patients with poor CAR T-cell persistence had a complete response after treatment with the combination therapy. Study details: A clinical study of 14 patients with relapsed B-ALL.
Disclosures: Dr. Maude reported financial relationships with Novartis.
Source: Li AM et al. ASH 2018, Abstract 556.
Chronic Myeloid Leukemia: Evaluation and Diagnosis
Chronic myeloid leukemia (CML) is a rare myeloproliferative neoplasm that is characterized by the presence of the Philadelphia (Ph) chromosome and uninhibited expansion of bone marrow stem cells. The Ph chromosome arises from a reciprocal translocation between the Abelson (ABL) region on chromosome 9 and the breakpoint cluster region (BCR) of chromosome 22 (t(9;22)(q34;q11.2), resulting in the BCR-ABL1 fusion gene.1BCR-ABL1 encodes an oncoprotein with constitutive tyrosine kinase activity that promotes growth and replication through downstream pathways, which is the driving factor in the pathogenesis of CML.1
Typical treatment for CML involves life-long use of oral BCR-ABL tyrosine kinase inhibitors (TKI). Currently, 5 TKIs have regulatory approval for treatment of this disease. With the introduction of imatinib in 2001 and the subsequent development of second- (dasatinib, nilotinib, bosutinib) and third-generation (ponatinib) TKIs, CML has become a chronic disease with a life-expectancy that is similar to that of the general population. This article reviews the diagnosis of CML and the parameters used for monitoring response to TKI therapy; the selection of initial TKI therapy is reviewed in a separate follow-up article.
Epidemiology
According to SEER data estimates, 8430 new cases of CML were diagnosed in the United States in 2018. CML is a disease of older adults, with a median age of 65 years at diagnosis, and there is a slight male predominance. Between 2011 and 2015, the number of new CML cases was 1.8 per 100,000 persons. The median overall survival (OS) in patients with newly diagnosed chronic-phase CML (CP-CML) has not been reached.2 Given the effective treatments available for managing CML, it is estimated that the prevalence of CML in the United States will plateau at 180,000 patients by 2050.3
Diagnosis
Case Presentation
A 53-year-old woman presents to her primary care physician with complaints of fatigue, early satiety, left upper quadrant abdominal pain, and an 8-lb unintentional weight loss over the prior month. Her past medical history is significant for uncontrolled diabetes, coronary artery disease requiring placement of 3 cardiac stents 2 years prior, and chronic obstructive pulmonary disease (COPD) related to a 30-pack-year history of smoking. On physicial exam her spleen is palpated 8 cm below the left costal margin. A complete blood count (CBC) with differential identifies a total white blood cell (WBC) count of 124,000/μL, with a left-shifted differential including 6% basophils, 3% eosinophils, and 3% blasts; hemoglobin is 12.4 g/dL and platelet count is 801 × 103/µL.
- How is the diagnosis of CML made?
Clinical Features
The diagnosis of CML is often suspected based on an incidental finding of leukocytosis and, in some cases, thrombocytosis. In many cases, this is an incidental finding on routine blood work, but approximately 50% of patients will present with constitutional symptoms associated with the disease. Characteristic features of the WBC differential include left-shifted maturation with neutrophilia and immature circulating myeloid cells. Basophilia and eosinophilia are often present as well. Splenomegaly is a common sign, present in 50% to 90% of patients at diagnosis. In those patients with symptoms related to CML at diagnosis, the most common presentation includes increasing fatigue, fevers, night sweats, early satiety, and weight loss. The diagnosis is confirmed by cytogenetic studies showing the Ph chromosome abnormality, t(9; 22)(q3.4;q1.1), and/or reverse transcriptase polymerase chain reaction (PCR) showing BCR-ABL1 transcripts.
- What further testing is needed when evaluating a patient for CML?
There are 3 distinct phases of CML: chronic phase (CP), accelerated phase (AP), and blast phase (BP). Bone marrow biopsy and aspiration at diagnosis are mandatory in order to determine the phase of the disease at diagnosis. This distinction is based on the percentage of blasts, promyelocytes, and basophils present as well as the platelet count and presence or absence of extramedullary disease.4 The vast majority of patients at diagnosis have CML that is in the chronic phase. The typical appearance in CP-CML is a hypercellular marrow with granulocytic and occasionally megakaryocytic hyperplasia. In many cases, basophilia and/or eosinophilia are noted as well. Dysplasia is not a typical finding in CML.5 Bone marrow fibrosis can be seen in up to one-third of patients at diagnosis, and may indicate a slightly worse prognosis.6 Although a diagnosis of CML can be made without a bone marrow biopsy, complete staging and prognostication are only possible with information gained from this test, including baseline karyotype and confirmation of CP versus a more advanced phase of CML.
The criteria for diagnosing AP-CML has not been agreed upon by various groups, but the modified MD Anderson Cancer Center (MDACC) criteria are used in the majority of clinical trials evaluating the efficacy of TKIs in preventing progression to advanced phases of CML. MDACC criteria define AP-CML as the presence of one of the following: 15% to 29% blasts in the peripheral blood or bone marrow, ≥ 30% peripheral blasts plus promyelocytes, ≥ 20% basophils in the blood or bone marrow, platelet count ≤ 100 × 103/μL unrelated to therapy, and clonal cytogenetic evolution in Ph-positive metaphases (Table).7
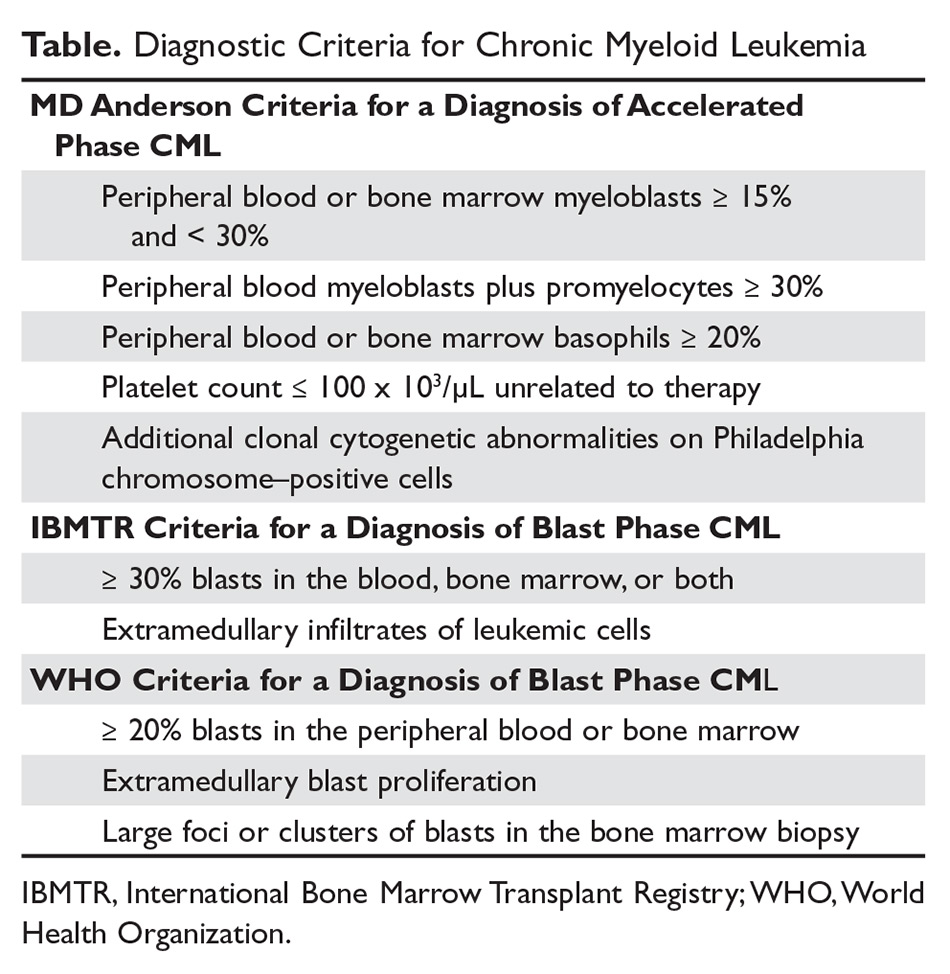
BP-CML is typically defined using the criteria developed by the International Bone Marrow Transplant Registry (IBMTR): ≥ 30% blasts in the peripheral blood and/or the bone marrow or the presence of extramedullary disease.8 Although not typically used in clinical trials, the revised World Health Organization (WHO) criteria for BP-CML include ≥ 20% blasts in the peripheral blood or bone marrow, extramedullary blast proliferation, and large foci or clusters of blasts in the bone marrow biopsy (Table).9 The defining feature of CML is the presence of the Ph chromosome abnormality. In a small subset of patients, additional chromosomal abnormalities (ACA) in the Ph-positive cells may be identified at diagnosis. Some reports indicate that the presence of “major route” ACA (trisomy 8, isochromosome 17q, a second Ph chromosome, or trisomy 19) at diagnosis may negatively impact prognosis, but other reports contradict these findings.10,11
The typical BCR breakpoint in CML is the major breakpoint cluster region (M-BCR), which results in a 210-kDa protein (p210). Alternate breakpoints that are less frequently identified are the minor BCR (mBCR or p190), which is more commonly found in Ph-positive acute lymphoblastic leukemia (ALL), and the micro BCR (µBCR or p230), which is much less common and is often characterized by chronic neutrophilia.12 Identifying which BCR-ABL1 transcript is present in each patient using qualitative PCR is crucial in order to ensure proper monitoring during treatment.
The most sensitive method for detecting BCR-ABL1 mRNA transcripts is the quantitative real-time PCR (RQ-PCR) assay, which is typically done on peripheral blood. RQ-PCR is capable of detecting a single CML cell in the presence of ≥ 100,000 normal cells. This test should be done during the initial diagnostic workup in order to confirm the presence of BCR-ABL1 transcripts, and it is used as a standard method for monitoring response to TKI therapy.13 The International Scale (IS) is a standardized approach to reporting RQ-PCR results that was developed to allow comparison of results across various laboratories and has become the gold standard for reporting BCR-ABL1 transcript values.14
Determining Risk Scores
Calculating a patient’s Sokal score or EURO risk score at diagnosis remains an important component of the diagnostic workup in CP-CML, as this information has prognostic and therapeutic implications (an online calculator is available through European LeukemiaNet [ELN]). The risk for disease progression to the accelerated or blast phases is higher in patients with intermediate- or high-risk scores compared to those with a low-risk score at diagnosis. The risk of progression in intermediate- or high-risk patients is lower when a second-generation TKI (dasatinib, nilotinib, or bosutinib) is used as frontline therapy compared to imatinib, and therefore, the National Comprehensive Cancer Network (NCCN) CML Panel recommends starting with a second-generation TKI in these patients.15-19
Monitoring Response to Therapy
Case Continued
Fluorescent in-situ hybridization using a peripheral blood sample to detect the BCR-ABL gene rearrangement is performed and is positive in 87% of cells. Bone marrow biopsy and aspiration show a 95% cellular bone marrow with granulocytic hyperplasia and 1% blasts. Cytogenetics are 46,XX,t(9;22)(q34;q11.2).20 RQ-PCR assay performed to measure BCR-ABL1 transcripts in the peripheral blood shows a value of 98% IS. The patient is ultimately given a diagnosis of CP-CML. Her Sokal risk score is 1.42, making her disease high risk.
How is response to TKI therapy measured and monitored?
After confirming a diagnosis of CML and selecting the most appropriate TKI for first-line therapy, the successful management of a CML patient relies on close monitoring and follow-up to ensure patients are meeting the desired treatment milestones. Responses in CML can be assessed based on hematologic parameters, cytogenetic results, and molecular responses. A complete hematologic response (CHR) implies complete normalization of peripheral blood counts (with the exception of TKI-induced cytopenias) and resolution of any palpable splenomegaly. The majority of patients will achieve a CHR within 4 to 6 weeks after initiating CML-directed therapy.21
Cytogenetic Response
Cytogenetic responses are defined by the decrease in the number of Ph chromosome–positive metaphases when assessed on bone marrow cytogenetics. A partial cytogenetic response (PCyR) is defined as having 1% to 35% Ph-positive metaphases, a major cytogenetic response (MCyR) as having 0% to 35% Ph-positive metaphases, and a CCyR implies that no Ph-positive metaphases are identified on bone marrow cytogenetics. An ideal response is the achievement of PCyR after 3 months on a TKI and a CCyR after 12 months on a TKI.22
Molecular Response
Once a patient has achieved a CCyR, monitoring their response to therapy can only be done using RQ-PCR to measure BCR-ABL1 transcripts in the peripheral blood. The NCCN and the ELN recommend monitoring RQ-PCR from the peripheral blood every 3 months in order to assess response to TKIs.19,23 As noted, the International Scale (IS) has become the gold standard reporting system for all BCR-ABL1 transcript levels in the majority of laboratories worldwide.14,24 Molecular responses are based on a log-reduction in BCR-ABL1 transcripts from a standardized baseline. Many molecular responses can be correlated with cytogenetic responses such that if reliable RQ-PCR testing is available, monitoring can be done using only peripheral blood RQ-PCR rather than repeat bone marrow biopsies. For example, an early molecular response (EMR) is defined as a RQ-PCR value of ≤ 10% IS, which is approximately equivalent to a PCyR.25 A value of 1% IS is approximately equivalent to CCyR. A major molecular response (MMR) is a ≥ 3-log reduction in BCR-ABL1 transcripts from baseline and is a value of ≤ 0.1% IS. Deeper levels of molecular response are best described by the log-reduction in BCR-ABL1 transcripts, with a 4-log reduction denoted as MR4.0, a 4.5-log reduction as MR4.5, and so forth. Complete molecular response (CMR) is defined by the level of sensitivity of the RQ-PCR assay being used.14
The definition of relapsed disease in CML is dependent on the type of response the patient had previously achieved. Relapse could be the loss of a hematologic or cytogenetic response, but fluctuations in BCR-ABL1 transcripts on routine RQ-PCR do not necessarily indicate relapsed CML. A 1-log increase in the level of BCR-ABL1 transcripts with a concurrent loss of MMR should prompt a bone marrow biopsy in order to assess for the loss of CCyR, and thus a cytogenetic relapse; however, this loss of MMR does not define relapse in and of itself. In the setting of relapsed disease, testing should be done to look for possible ABL kinase domain mutations, and alternate therapy should be selected.19
Multiple reports have identified the prognostic relevance of achieving an EMR at 3 and 6 months after starting TKI therapy. Marin and colleagues reported that in 282 imatinib-treated patients, there was a significant improvement in 8-year OS, progression-free survival, and cumulative incidence of CCyR and CMR in patients who had BCR-ABL1 transcripts < 9.84% IS after 3 months on treatment.25 This data highlights the importance of early molecular monitoring in order to ensure the best outcomes for patients with CP-CML.
The NCCN CML guidelines and ELN recommendations both agree that an ideal response after 3 months on a TKI is BCR-ABL1 transcripts < 10% IS, but treatment is not considered to be failing at this point if the patient marginally misses this milestone. After 6 months on treatment, an ideal response is considered BCR-ABL1 transcripts < 1%–10% IS. Ideally, patients will have BCR-ABL1 transcripts < 0.1%–1% IS by the time they complete 12 months of TKI therapy, suggesting that these patients have at least achieved a CCyR.19,23 Even after patients achieve these early milestones, frequent monitoring by RQ-PCR is required to ensure that they are maintaining their response to treatment. This will help to ensure patient compliance with treatment and will also help to identify a select subset of patients who could potentially be considered for an attempt at TKI cessation (not discussed in detail here) after a minimum of 3 years on therapy.19,26
Conclusion
Given the successful treatments available for patients with CML, it is crucial to identify patients with this disease, ensure they receive a complete, appropriate diagnostic workup including a bone marrow biopsy and aspiration with cytogenetic testing, and select the best therapy for each individual patient. Once on treatment, the importance of frequent monitoring cannot be overstated.
1. Faderl S, Talpaz M, Estrov Z, et al. The biology of chronic myeloid leukemia. N Engl J Med. 1999;341:164-172.
2. Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Leukemia - Chronic Myeloid Leukemia (CML). 2018.
3. Huang X, Cortes J, Kantarjian H. Estimations of the increasing prevalence and plateau prevalence of chronic myeloid leukemia in the era of tyrosine kinase inhibitor therapy. Cancer. 2012;118:3123-3127.
4. Savage DG, Szydlo RM, Chase A, et al. Bone marrow transplantation for chronic myeloid leukaemia: the effects of differing criteria for defining chronic phase on probabilities of survival and relapse. Br J Haematol. 1997;99:30-35.
5. Knox WF, Bhavnani M, Davson J, Geary CG. Histological classification of chronic granulocytic leukaemia. Clin Lab Haematol. 1984;6:171-175.
6. Kvasnicka HM, Thiele J, Schmitt-Graeff A, et al. Impact of bone marrow morphology on multivariate risk classification in chronic myelogenous leukemia. Acta Haematol. 2003;109:53-56.
7. Cortes JE, Talpaz M, O’Brien S, et al. Staging of chronic myeloid leukemia in the imatinib era: an evaluation of the World Health Organization proposal. Cancer. 2006;106:1306-1315.
8. Druker BJ. Chronic myeloid leukemia In: DeVita VT, Lawrence TS, Rosenburg SA, eds. DeVita, Hellman, and Rosenberg’s Cancer Principles & Practice of Oncology. 8th ed. Philadelphia, PA: Lippincott, Williams and Wilkins; 2007:2267-2304.
9. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391-2405.
10. Fabarius A, Leitner A, Hochhaus A, et al. Impact of additional cytogenetic aberrations at diagnosis on prognosis of CML: long-term observation of 1151 patients from the randomized CML Study IV. Blood. 2011;118:6760-6768.
11. Alhuraiji A, Kantarjian H, Boddu P, et al. Prognostic significance of additional chromosomal abnormalities at the time of diagnosis in patients with chronic myeloid leukemia treated with frontline tyrosine kinase inhibitors. Am J Hematol. 2018;93:84-90.
12. Melo JV. BCR-ABL gene variants. Baillieres Clin Haematol. 1997;10:203-222.
13. Kantarjian HM, Talpaz M, Cortes J, et al. Quantitative polymerase chain reaction monitoring of BCR-ABL during therapy with imatinib mesylate (STI571; gleevec) in chronic-phase chronic myelogenous leukemia. Clin Cancer Res. 2003;9:160-166.
14. Hughes T, Deininger M, Hochhaus A, et al. Monitoring CML patients responding to treatment with tyrosine kinase inhibitors: review and recommendations for harmonizing current methodology for detecting BCR-ABL transcripts and kinase domain mutations and for expressing results. Blood. 2006;108:28-37.
15. Hochhaus A, Larson RA, Guilhot F, et al. Long-term outcomes of imatinib treatment for chronic myeloid leukemia. N Engl J Med. 2017;376:917-927.
16. Cortes JE, Saglio G, Kantarjian HM, et al. Final 5-year study results of DASISION: the Dasatinib Versus Imatinib Study in Treatment-Naive Chronic Myeloid Leukemia Patients trial. J Clin Oncol. 2016;34:2333-3340.
17. Hochhaus A, Saglio G, Hughes TP, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30:1044-1054.
18. Cortes JE, Gambacorti-Passerini C, Deininger MW, et al. Bosutinib versus imatinib for newly diagnosed chronic myeloid leukemia: results from the randomized BFORE trial. J Clin Oncol. 2018;36:231-237.
19. Radich JP, Deininger M, Abboud CN, et al. Chronic Myeloid Leukemia, Version 1.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:1108-1135.
20. Druker BJ, Talpaz M, Resta DJ, et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med. 2001;344:1031-1037.
21. Faderl S, Talpaz M, Estrov Z, Kantarjian HM. Chronic myelogenous leukemia: biology and therapy. Ann Intern Med. 1999;131:207-219.
22. O’Brien SG, Guilhot F, Larson RA, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003;348:994-1004.
23. Baccarani M, Deininger MW, Rosti G, et al. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood. 2013;122:872-884.
24. Larripa I, Ruiz MS, Gutierrez M, Bianchini M. [Guidelines for molecular monitoring of BCR-ABL1 in chronic myeloid leukemia patients by RT-qPCR.] Medicina (B Aires). 2017;77:61-72.
25. Marin D, Ibrahim AR, Lucas C, et al. Assessment of BCR-ABL1 transcript levels at 3 months is the only requirement for predicting outcome for patients with chronic myeloid leukemia treated with tyrosine kinase inhibitors. J Clin Oncol. 2012;30:232-238.
26. Hughes TP, Ross DM. Moving treatment-free remission into mainstream clinical practice in CML. Blood. 2016;128:17-23.
Chronic myeloid leukemia (CML) is a rare myeloproliferative neoplasm that is characterized by the presence of the Philadelphia (Ph) chromosome and uninhibited expansion of bone marrow stem cells. The Ph chromosome arises from a reciprocal translocation between the Abelson (ABL) region on chromosome 9 and the breakpoint cluster region (BCR) of chromosome 22 (t(9;22)(q34;q11.2), resulting in the BCR-ABL1 fusion gene.1BCR-ABL1 encodes an oncoprotein with constitutive tyrosine kinase activity that promotes growth and replication through downstream pathways, which is the driving factor in the pathogenesis of CML.1
Typical treatment for CML involves life-long use of oral BCR-ABL tyrosine kinase inhibitors (TKI). Currently, 5 TKIs have regulatory approval for treatment of this disease. With the introduction of imatinib in 2001 and the subsequent development of second- (dasatinib, nilotinib, bosutinib) and third-generation (ponatinib) TKIs, CML has become a chronic disease with a life-expectancy that is similar to that of the general population. This article reviews the diagnosis of CML and the parameters used for monitoring response to TKI therapy; the selection of initial TKI therapy is reviewed in a separate follow-up article.
Epidemiology
According to SEER data estimates, 8430 new cases of CML were diagnosed in the United States in 2018. CML is a disease of older adults, with a median age of 65 years at diagnosis, and there is a slight male predominance. Between 2011 and 2015, the number of new CML cases was 1.8 per 100,000 persons. The median overall survival (OS) in patients with newly diagnosed chronic-phase CML (CP-CML) has not been reached.2 Given the effective treatments available for managing CML, it is estimated that the prevalence of CML in the United States will plateau at 180,000 patients by 2050.3
Diagnosis
Case Presentation
A 53-year-old woman presents to her primary care physician with complaints of fatigue, early satiety, left upper quadrant abdominal pain, and an 8-lb unintentional weight loss over the prior month. Her past medical history is significant for uncontrolled diabetes, coronary artery disease requiring placement of 3 cardiac stents 2 years prior, and chronic obstructive pulmonary disease (COPD) related to a 30-pack-year history of smoking. On physicial exam her spleen is palpated 8 cm below the left costal margin. A complete blood count (CBC) with differential identifies a total white blood cell (WBC) count of 124,000/μL, with a left-shifted differential including 6% basophils, 3% eosinophils, and 3% blasts; hemoglobin is 12.4 g/dL and platelet count is 801 × 103/µL.
- How is the diagnosis of CML made?
Clinical Features
The diagnosis of CML is often suspected based on an incidental finding of leukocytosis and, in some cases, thrombocytosis. In many cases, this is an incidental finding on routine blood work, but approximately 50% of patients will present with constitutional symptoms associated with the disease. Characteristic features of the WBC differential include left-shifted maturation with neutrophilia and immature circulating myeloid cells. Basophilia and eosinophilia are often present as well. Splenomegaly is a common sign, present in 50% to 90% of patients at diagnosis. In those patients with symptoms related to CML at diagnosis, the most common presentation includes increasing fatigue, fevers, night sweats, early satiety, and weight loss. The diagnosis is confirmed by cytogenetic studies showing the Ph chromosome abnormality, t(9; 22)(q3.4;q1.1), and/or reverse transcriptase polymerase chain reaction (PCR) showing BCR-ABL1 transcripts.
- What further testing is needed when evaluating a patient for CML?
There are 3 distinct phases of CML: chronic phase (CP), accelerated phase (AP), and blast phase (BP). Bone marrow biopsy and aspiration at diagnosis are mandatory in order to determine the phase of the disease at diagnosis. This distinction is based on the percentage of blasts, promyelocytes, and basophils present as well as the platelet count and presence or absence of extramedullary disease.4 The vast majority of patients at diagnosis have CML that is in the chronic phase. The typical appearance in CP-CML is a hypercellular marrow with granulocytic and occasionally megakaryocytic hyperplasia. In many cases, basophilia and/or eosinophilia are noted as well. Dysplasia is not a typical finding in CML.5 Bone marrow fibrosis can be seen in up to one-third of patients at diagnosis, and may indicate a slightly worse prognosis.6 Although a diagnosis of CML can be made without a bone marrow biopsy, complete staging and prognostication are only possible with information gained from this test, including baseline karyotype and confirmation of CP versus a more advanced phase of CML.
The criteria for diagnosing AP-CML has not been agreed upon by various groups, but the modified MD Anderson Cancer Center (MDACC) criteria are used in the majority of clinical trials evaluating the efficacy of TKIs in preventing progression to advanced phases of CML. MDACC criteria define AP-CML as the presence of one of the following: 15% to 29% blasts in the peripheral blood or bone marrow, ≥ 30% peripheral blasts plus promyelocytes, ≥ 20% basophils in the blood or bone marrow, platelet count ≤ 100 × 103/μL unrelated to therapy, and clonal cytogenetic evolution in Ph-positive metaphases (Table).7

BP-CML is typically defined using the criteria developed by the International Bone Marrow Transplant Registry (IBMTR): ≥ 30% blasts in the peripheral blood and/or the bone marrow or the presence of extramedullary disease.8 Although not typically used in clinical trials, the revised World Health Organization (WHO) criteria for BP-CML include ≥ 20% blasts in the peripheral blood or bone marrow, extramedullary blast proliferation, and large foci or clusters of blasts in the bone marrow biopsy (Table).9 The defining feature of CML is the presence of the Ph chromosome abnormality. In a small subset of patients, additional chromosomal abnormalities (ACA) in the Ph-positive cells may be identified at diagnosis. Some reports indicate that the presence of “major route” ACA (trisomy 8, isochromosome 17q, a second Ph chromosome, or trisomy 19) at diagnosis may negatively impact prognosis, but other reports contradict these findings.10,11
The typical BCR breakpoint in CML is the major breakpoint cluster region (M-BCR), which results in a 210-kDa protein (p210). Alternate breakpoints that are less frequently identified are the minor BCR (mBCR or p190), which is more commonly found in Ph-positive acute lymphoblastic leukemia (ALL), and the micro BCR (µBCR or p230), which is much less common and is often characterized by chronic neutrophilia.12 Identifying which BCR-ABL1 transcript is present in each patient using qualitative PCR is crucial in order to ensure proper monitoring during treatment.
The most sensitive method for detecting BCR-ABL1 mRNA transcripts is the quantitative real-time PCR (RQ-PCR) assay, which is typically done on peripheral blood. RQ-PCR is capable of detecting a single CML cell in the presence of ≥ 100,000 normal cells. This test should be done during the initial diagnostic workup in order to confirm the presence of BCR-ABL1 transcripts, and it is used as a standard method for monitoring response to TKI therapy.13 The International Scale (IS) is a standardized approach to reporting RQ-PCR results that was developed to allow comparison of results across various laboratories and has become the gold standard for reporting BCR-ABL1 transcript values.14
Determining Risk Scores
Calculating a patient’s Sokal score or EURO risk score at diagnosis remains an important component of the diagnostic workup in CP-CML, as this information has prognostic and therapeutic implications (an online calculator is available through European LeukemiaNet [ELN]). The risk for disease progression to the accelerated or blast phases is higher in patients with intermediate- or high-risk scores compared to those with a low-risk score at diagnosis. The risk of progression in intermediate- or high-risk patients is lower when a second-generation TKI (dasatinib, nilotinib, or bosutinib) is used as frontline therapy compared to imatinib, and therefore, the National Comprehensive Cancer Network (NCCN) CML Panel recommends starting with a second-generation TKI in these patients.15-19
Monitoring Response to Therapy
Case Continued
Fluorescent in-situ hybridization using a peripheral blood sample to detect the BCR-ABL gene rearrangement is performed and is positive in 87% of cells. Bone marrow biopsy and aspiration show a 95% cellular bone marrow with granulocytic hyperplasia and 1% blasts. Cytogenetics are 46,XX,t(9;22)(q34;q11.2).20 RQ-PCR assay performed to measure BCR-ABL1 transcripts in the peripheral blood shows a value of 98% IS. The patient is ultimately given a diagnosis of CP-CML. Her Sokal risk score is 1.42, making her disease high risk.
How is response to TKI therapy measured and monitored?
After confirming a diagnosis of CML and selecting the most appropriate TKI for first-line therapy, the successful management of a CML patient relies on close monitoring and follow-up to ensure patients are meeting the desired treatment milestones. Responses in CML can be assessed based on hematologic parameters, cytogenetic results, and molecular responses. A complete hematologic response (CHR) implies complete normalization of peripheral blood counts (with the exception of TKI-induced cytopenias) and resolution of any palpable splenomegaly. The majority of patients will achieve a CHR within 4 to 6 weeks after initiating CML-directed therapy.21
Cytogenetic Response
Cytogenetic responses are defined by the decrease in the number of Ph chromosome–positive metaphases when assessed on bone marrow cytogenetics. A partial cytogenetic response (PCyR) is defined as having 1% to 35% Ph-positive metaphases, a major cytogenetic response (MCyR) as having 0% to 35% Ph-positive metaphases, and a CCyR implies that no Ph-positive metaphases are identified on bone marrow cytogenetics. An ideal response is the achievement of PCyR after 3 months on a TKI and a CCyR after 12 months on a TKI.22
Molecular Response
Once a patient has achieved a CCyR, monitoring their response to therapy can only be done using RQ-PCR to measure BCR-ABL1 transcripts in the peripheral blood. The NCCN and the ELN recommend monitoring RQ-PCR from the peripheral blood every 3 months in order to assess response to TKIs.19,23 As noted, the International Scale (IS) has become the gold standard reporting system for all BCR-ABL1 transcript levels in the majority of laboratories worldwide.14,24 Molecular responses are based on a log-reduction in BCR-ABL1 transcripts from a standardized baseline. Many molecular responses can be correlated with cytogenetic responses such that if reliable RQ-PCR testing is available, monitoring can be done using only peripheral blood RQ-PCR rather than repeat bone marrow biopsies. For example, an early molecular response (EMR) is defined as a RQ-PCR value of ≤ 10% IS, which is approximately equivalent to a PCyR.25 A value of 1% IS is approximately equivalent to CCyR. A major molecular response (MMR) is a ≥ 3-log reduction in BCR-ABL1 transcripts from baseline and is a value of ≤ 0.1% IS. Deeper levels of molecular response are best described by the log-reduction in BCR-ABL1 transcripts, with a 4-log reduction denoted as MR4.0, a 4.5-log reduction as MR4.5, and so forth. Complete molecular response (CMR) is defined by the level of sensitivity of the RQ-PCR assay being used.14
The definition of relapsed disease in CML is dependent on the type of response the patient had previously achieved. Relapse could be the loss of a hematologic or cytogenetic response, but fluctuations in BCR-ABL1 transcripts on routine RQ-PCR do not necessarily indicate relapsed CML. A 1-log increase in the level of BCR-ABL1 transcripts with a concurrent loss of MMR should prompt a bone marrow biopsy in order to assess for the loss of CCyR, and thus a cytogenetic relapse; however, this loss of MMR does not define relapse in and of itself. In the setting of relapsed disease, testing should be done to look for possible ABL kinase domain mutations, and alternate therapy should be selected.19
Multiple reports have identified the prognostic relevance of achieving an EMR at 3 and 6 months after starting TKI therapy. Marin and colleagues reported that in 282 imatinib-treated patients, there was a significant improvement in 8-year OS, progression-free survival, and cumulative incidence of CCyR and CMR in patients who had BCR-ABL1 transcripts < 9.84% IS after 3 months on treatment.25 This data highlights the importance of early molecular monitoring in order to ensure the best outcomes for patients with CP-CML.
The NCCN CML guidelines and ELN recommendations both agree that an ideal response after 3 months on a TKI is BCR-ABL1 transcripts < 10% IS, but treatment is not considered to be failing at this point if the patient marginally misses this milestone. After 6 months on treatment, an ideal response is considered BCR-ABL1 transcripts < 1%–10% IS. Ideally, patients will have BCR-ABL1 transcripts < 0.1%–1% IS by the time they complete 12 months of TKI therapy, suggesting that these patients have at least achieved a CCyR.19,23 Even after patients achieve these early milestones, frequent monitoring by RQ-PCR is required to ensure that they are maintaining their response to treatment. This will help to ensure patient compliance with treatment and will also help to identify a select subset of patients who could potentially be considered for an attempt at TKI cessation (not discussed in detail here) after a minimum of 3 years on therapy.19,26
Conclusion
Given the successful treatments available for patients with CML, it is crucial to identify patients with this disease, ensure they receive a complete, appropriate diagnostic workup including a bone marrow biopsy and aspiration with cytogenetic testing, and select the best therapy for each individual patient. Once on treatment, the importance of frequent monitoring cannot be overstated.
Chronic myeloid leukemia (CML) is a rare myeloproliferative neoplasm that is characterized by the presence of the Philadelphia (Ph) chromosome and uninhibited expansion of bone marrow stem cells. The Ph chromosome arises from a reciprocal translocation between the Abelson (ABL) region on chromosome 9 and the breakpoint cluster region (BCR) of chromosome 22 (t(9;22)(q34;q11.2), resulting in the BCR-ABL1 fusion gene.1BCR-ABL1 encodes an oncoprotein with constitutive tyrosine kinase activity that promotes growth and replication through downstream pathways, which is the driving factor in the pathogenesis of CML.1
Typical treatment for CML involves life-long use of oral BCR-ABL tyrosine kinase inhibitors (TKI). Currently, 5 TKIs have regulatory approval for treatment of this disease. With the introduction of imatinib in 2001 and the subsequent development of second- (dasatinib, nilotinib, bosutinib) and third-generation (ponatinib) TKIs, CML has become a chronic disease with a life-expectancy that is similar to that of the general population. This article reviews the diagnosis of CML and the parameters used for monitoring response to TKI therapy; the selection of initial TKI therapy is reviewed in a separate follow-up article.
Epidemiology
According to SEER data estimates, 8430 new cases of CML were diagnosed in the United States in 2018. CML is a disease of older adults, with a median age of 65 years at diagnosis, and there is a slight male predominance. Between 2011 and 2015, the number of new CML cases was 1.8 per 100,000 persons. The median overall survival (OS) in patients with newly diagnosed chronic-phase CML (CP-CML) has not been reached.2 Given the effective treatments available for managing CML, it is estimated that the prevalence of CML in the United States will plateau at 180,000 patients by 2050.3
Diagnosis
Case Presentation
A 53-year-old woman presents to her primary care physician with complaints of fatigue, early satiety, left upper quadrant abdominal pain, and an 8-lb unintentional weight loss over the prior month. Her past medical history is significant for uncontrolled diabetes, coronary artery disease requiring placement of 3 cardiac stents 2 years prior, and chronic obstructive pulmonary disease (COPD) related to a 30-pack-year history of smoking. On physicial exam her spleen is palpated 8 cm below the left costal margin. A complete blood count (CBC) with differential identifies a total white blood cell (WBC) count of 124,000/μL, with a left-shifted differential including 6% basophils, 3% eosinophils, and 3% blasts; hemoglobin is 12.4 g/dL and platelet count is 801 × 103/µL.
- How is the diagnosis of CML made?
Clinical Features
The diagnosis of CML is often suspected based on an incidental finding of leukocytosis and, in some cases, thrombocytosis. In many cases, this is an incidental finding on routine blood work, but approximately 50% of patients will present with constitutional symptoms associated with the disease. Characteristic features of the WBC differential include left-shifted maturation with neutrophilia and immature circulating myeloid cells. Basophilia and eosinophilia are often present as well. Splenomegaly is a common sign, present in 50% to 90% of patients at diagnosis. In those patients with symptoms related to CML at diagnosis, the most common presentation includes increasing fatigue, fevers, night sweats, early satiety, and weight loss. The diagnosis is confirmed by cytogenetic studies showing the Ph chromosome abnormality, t(9; 22)(q3.4;q1.1), and/or reverse transcriptase polymerase chain reaction (PCR) showing BCR-ABL1 transcripts.
- What further testing is needed when evaluating a patient for CML?
There are 3 distinct phases of CML: chronic phase (CP), accelerated phase (AP), and blast phase (BP). Bone marrow biopsy and aspiration at diagnosis are mandatory in order to determine the phase of the disease at diagnosis. This distinction is based on the percentage of blasts, promyelocytes, and basophils present as well as the platelet count and presence or absence of extramedullary disease.4 The vast majority of patients at diagnosis have CML that is in the chronic phase. The typical appearance in CP-CML is a hypercellular marrow with granulocytic and occasionally megakaryocytic hyperplasia. In many cases, basophilia and/or eosinophilia are noted as well. Dysplasia is not a typical finding in CML.5 Bone marrow fibrosis can be seen in up to one-third of patients at diagnosis, and may indicate a slightly worse prognosis.6 Although a diagnosis of CML can be made without a bone marrow biopsy, complete staging and prognostication are only possible with information gained from this test, including baseline karyotype and confirmation of CP versus a more advanced phase of CML.
The criteria for diagnosing AP-CML has not been agreed upon by various groups, but the modified MD Anderson Cancer Center (MDACC) criteria are used in the majority of clinical trials evaluating the efficacy of TKIs in preventing progression to advanced phases of CML. MDACC criteria define AP-CML as the presence of one of the following: 15% to 29% blasts in the peripheral blood or bone marrow, ≥ 30% peripheral blasts plus promyelocytes, ≥ 20% basophils in the blood or bone marrow, platelet count ≤ 100 × 103/μL unrelated to therapy, and clonal cytogenetic evolution in Ph-positive metaphases (Table).7

BP-CML is typically defined using the criteria developed by the International Bone Marrow Transplant Registry (IBMTR): ≥ 30% blasts in the peripheral blood and/or the bone marrow or the presence of extramedullary disease.8 Although not typically used in clinical trials, the revised World Health Organization (WHO) criteria for BP-CML include ≥ 20% blasts in the peripheral blood or bone marrow, extramedullary blast proliferation, and large foci or clusters of blasts in the bone marrow biopsy (Table).9 The defining feature of CML is the presence of the Ph chromosome abnormality. In a small subset of patients, additional chromosomal abnormalities (ACA) in the Ph-positive cells may be identified at diagnosis. Some reports indicate that the presence of “major route” ACA (trisomy 8, isochromosome 17q, a second Ph chromosome, or trisomy 19) at diagnosis may negatively impact prognosis, but other reports contradict these findings.10,11
The typical BCR breakpoint in CML is the major breakpoint cluster region (M-BCR), which results in a 210-kDa protein (p210). Alternate breakpoints that are less frequently identified are the minor BCR (mBCR or p190), which is more commonly found in Ph-positive acute lymphoblastic leukemia (ALL), and the micro BCR (µBCR or p230), which is much less common and is often characterized by chronic neutrophilia.12 Identifying which BCR-ABL1 transcript is present in each patient using qualitative PCR is crucial in order to ensure proper monitoring during treatment.
The most sensitive method for detecting BCR-ABL1 mRNA transcripts is the quantitative real-time PCR (RQ-PCR) assay, which is typically done on peripheral blood. RQ-PCR is capable of detecting a single CML cell in the presence of ≥ 100,000 normal cells. This test should be done during the initial diagnostic workup in order to confirm the presence of BCR-ABL1 transcripts, and it is used as a standard method for monitoring response to TKI therapy.13 The International Scale (IS) is a standardized approach to reporting RQ-PCR results that was developed to allow comparison of results across various laboratories and has become the gold standard for reporting BCR-ABL1 transcript values.14
Determining Risk Scores
Calculating a patient’s Sokal score or EURO risk score at diagnosis remains an important component of the diagnostic workup in CP-CML, as this information has prognostic and therapeutic implications (an online calculator is available through European LeukemiaNet [ELN]). The risk for disease progression to the accelerated or blast phases is higher in patients with intermediate- or high-risk scores compared to those with a low-risk score at diagnosis. The risk of progression in intermediate- or high-risk patients is lower when a second-generation TKI (dasatinib, nilotinib, or bosutinib) is used as frontline therapy compared to imatinib, and therefore, the National Comprehensive Cancer Network (NCCN) CML Panel recommends starting with a second-generation TKI in these patients.15-19
Monitoring Response to Therapy
Case Continued
Fluorescent in-situ hybridization using a peripheral blood sample to detect the BCR-ABL gene rearrangement is performed and is positive in 87% of cells. Bone marrow biopsy and aspiration show a 95% cellular bone marrow with granulocytic hyperplasia and 1% blasts. Cytogenetics are 46,XX,t(9;22)(q34;q11.2).20 RQ-PCR assay performed to measure BCR-ABL1 transcripts in the peripheral blood shows a value of 98% IS. The patient is ultimately given a diagnosis of CP-CML. Her Sokal risk score is 1.42, making her disease high risk.
How is response to TKI therapy measured and monitored?
After confirming a diagnosis of CML and selecting the most appropriate TKI for first-line therapy, the successful management of a CML patient relies on close monitoring and follow-up to ensure patients are meeting the desired treatment milestones. Responses in CML can be assessed based on hematologic parameters, cytogenetic results, and molecular responses. A complete hematologic response (CHR) implies complete normalization of peripheral blood counts (with the exception of TKI-induced cytopenias) and resolution of any palpable splenomegaly. The majority of patients will achieve a CHR within 4 to 6 weeks after initiating CML-directed therapy.21
Cytogenetic Response
Cytogenetic responses are defined by the decrease in the number of Ph chromosome–positive metaphases when assessed on bone marrow cytogenetics. A partial cytogenetic response (PCyR) is defined as having 1% to 35% Ph-positive metaphases, a major cytogenetic response (MCyR) as having 0% to 35% Ph-positive metaphases, and a CCyR implies that no Ph-positive metaphases are identified on bone marrow cytogenetics. An ideal response is the achievement of PCyR after 3 months on a TKI and a CCyR after 12 months on a TKI.22
Molecular Response
Once a patient has achieved a CCyR, monitoring their response to therapy can only be done using RQ-PCR to measure BCR-ABL1 transcripts in the peripheral blood. The NCCN and the ELN recommend monitoring RQ-PCR from the peripheral blood every 3 months in order to assess response to TKIs.19,23 As noted, the International Scale (IS) has become the gold standard reporting system for all BCR-ABL1 transcript levels in the majority of laboratories worldwide.14,24 Molecular responses are based on a log-reduction in BCR-ABL1 transcripts from a standardized baseline. Many molecular responses can be correlated with cytogenetic responses such that if reliable RQ-PCR testing is available, monitoring can be done using only peripheral blood RQ-PCR rather than repeat bone marrow biopsies. For example, an early molecular response (EMR) is defined as a RQ-PCR value of ≤ 10% IS, which is approximately equivalent to a PCyR.25 A value of 1% IS is approximately equivalent to CCyR. A major molecular response (MMR) is a ≥ 3-log reduction in BCR-ABL1 transcripts from baseline and is a value of ≤ 0.1% IS. Deeper levels of molecular response are best described by the log-reduction in BCR-ABL1 transcripts, with a 4-log reduction denoted as MR4.0, a 4.5-log reduction as MR4.5, and so forth. Complete molecular response (CMR) is defined by the level of sensitivity of the RQ-PCR assay being used.14
The definition of relapsed disease in CML is dependent on the type of response the patient had previously achieved. Relapse could be the loss of a hematologic or cytogenetic response, but fluctuations in BCR-ABL1 transcripts on routine RQ-PCR do not necessarily indicate relapsed CML. A 1-log increase in the level of BCR-ABL1 transcripts with a concurrent loss of MMR should prompt a bone marrow biopsy in order to assess for the loss of CCyR, and thus a cytogenetic relapse; however, this loss of MMR does not define relapse in and of itself. In the setting of relapsed disease, testing should be done to look for possible ABL kinase domain mutations, and alternate therapy should be selected.19
Multiple reports have identified the prognostic relevance of achieving an EMR at 3 and 6 months after starting TKI therapy. Marin and colleagues reported that in 282 imatinib-treated patients, there was a significant improvement in 8-year OS, progression-free survival, and cumulative incidence of CCyR and CMR in patients who had BCR-ABL1 transcripts < 9.84% IS after 3 months on treatment.25 This data highlights the importance of early molecular monitoring in order to ensure the best outcomes for patients with CP-CML.
The NCCN CML guidelines and ELN recommendations both agree that an ideal response after 3 months on a TKI is BCR-ABL1 transcripts < 10% IS, but treatment is not considered to be failing at this point if the patient marginally misses this milestone. After 6 months on treatment, an ideal response is considered BCR-ABL1 transcripts < 1%–10% IS. Ideally, patients will have BCR-ABL1 transcripts < 0.1%–1% IS by the time they complete 12 months of TKI therapy, suggesting that these patients have at least achieved a CCyR.19,23 Even after patients achieve these early milestones, frequent monitoring by RQ-PCR is required to ensure that they are maintaining their response to treatment. This will help to ensure patient compliance with treatment and will also help to identify a select subset of patients who could potentially be considered for an attempt at TKI cessation (not discussed in detail here) after a minimum of 3 years on therapy.19,26
Conclusion
Given the successful treatments available for patients with CML, it is crucial to identify patients with this disease, ensure they receive a complete, appropriate diagnostic workup including a bone marrow biopsy and aspiration with cytogenetic testing, and select the best therapy for each individual patient. Once on treatment, the importance of frequent monitoring cannot be overstated.
1. Faderl S, Talpaz M, Estrov Z, et al. The biology of chronic myeloid leukemia. N Engl J Med. 1999;341:164-172.
2. Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Leukemia - Chronic Myeloid Leukemia (CML). 2018.
3. Huang X, Cortes J, Kantarjian H. Estimations of the increasing prevalence and plateau prevalence of chronic myeloid leukemia in the era of tyrosine kinase inhibitor therapy. Cancer. 2012;118:3123-3127.
4. Savage DG, Szydlo RM, Chase A, et al. Bone marrow transplantation for chronic myeloid leukaemia: the effects of differing criteria for defining chronic phase on probabilities of survival and relapse. Br J Haematol. 1997;99:30-35.
5. Knox WF, Bhavnani M, Davson J, Geary CG. Histological classification of chronic granulocytic leukaemia. Clin Lab Haematol. 1984;6:171-175.
6. Kvasnicka HM, Thiele J, Schmitt-Graeff A, et al. Impact of bone marrow morphology on multivariate risk classification in chronic myelogenous leukemia. Acta Haematol. 2003;109:53-56.
7. Cortes JE, Talpaz M, O’Brien S, et al. Staging of chronic myeloid leukemia in the imatinib era: an evaluation of the World Health Organization proposal. Cancer. 2006;106:1306-1315.
8. Druker BJ. Chronic myeloid leukemia In: DeVita VT, Lawrence TS, Rosenburg SA, eds. DeVita, Hellman, and Rosenberg’s Cancer Principles & Practice of Oncology. 8th ed. Philadelphia, PA: Lippincott, Williams and Wilkins; 2007:2267-2304.
9. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391-2405.
10. Fabarius A, Leitner A, Hochhaus A, et al. Impact of additional cytogenetic aberrations at diagnosis on prognosis of CML: long-term observation of 1151 patients from the randomized CML Study IV. Blood. 2011;118:6760-6768.
11. Alhuraiji A, Kantarjian H, Boddu P, et al. Prognostic significance of additional chromosomal abnormalities at the time of diagnosis in patients with chronic myeloid leukemia treated with frontline tyrosine kinase inhibitors. Am J Hematol. 2018;93:84-90.
12. Melo JV. BCR-ABL gene variants. Baillieres Clin Haematol. 1997;10:203-222.
13. Kantarjian HM, Talpaz M, Cortes J, et al. Quantitative polymerase chain reaction monitoring of BCR-ABL during therapy with imatinib mesylate (STI571; gleevec) in chronic-phase chronic myelogenous leukemia. Clin Cancer Res. 2003;9:160-166.
14. Hughes T, Deininger M, Hochhaus A, et al. Monitoring CML patients responding to treatment with tyrosine kinase inhibitors: review and recommendations for harmonizing current methodology for detecting BCR-ABL transcripts and kinase domain mutations and for expressing results. Blood. 2006;108:28-37.
15. Hochhaus A, Larson RA, Guilhot F, et al. Long-term outcomes of imatinib treatment for chronic myeloid leukemia. N Engl J Med. 2017;376:917-927.
16. Cortes JE, Saglio G, Kantarjian HM, et al. Final 5-year study results of DASISION: the Dasatinib Versus Imatinib Study in Treatment-Naive Chronic Myeloid Leukemia Patients trial. J Clin Oncol. 2016;34:2333-3340.
17. Hochhaus A, Saglio G, Hughes TP, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30:1044-1054.
18. Cortes JE, Gambacorti-Passerini C, Deininger MW, et al. Bosutinib versus imatinib for newly diagnosed chronic myeloid leukemia: results from the randomized BFORE trial. J Clin Oncol. 2018;36:231-237.
19. Radich JP, Deininger M, Abboud CN, et al. Chronic Myeloid Leukemia, Version 1.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:1108-1135.
20. Druker BJ, Talpaz M, Resta DJ, et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med. 2001;344:1031-1037.
21. Faderl S, Talpaz M, Estrov Z, Kantarjian HM. Chronic myelogenous leukemia: biology and therapy. Ann Intern Med. 1999;131:207-219.
22. O’Brien SG, Guilhot F, Larson RA, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003;348:994-1004.
23. Baccarani M, Deininger MW, Rosti G, et al. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood. 2013;122:872-884.
24. Larripa I, Ruiz MS, Gutierrez M, Bianchini M. [Guidelines for molecular monitoring of BCR-ABL1 in chronic myeloid leukemia patients by RT-qPCR.] Medicina (B Aires). 2017;77:61-72.
25. Marin D, Ibrahim AR, Lucas C, et al. Assessment of BCR-ABL1 transcript levels at 3 months is the only requirement for predicting outcome for patients with chronic myeloid leukemia treated with tyrosine kinase inhibitors. J Clin Oncol. 2012;30:232-238.
26. Hughes TP, Ross DM. Moving treatment-free remission into mainstream clinical practice in CML. Blood. 2016;128:17-23.
1. Faderl S, Talpaz M, Estrov Z, et al. The biology of chronic myeloid leukemia. N Engl J Med. 1999;341:164-172.
2. Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Leukemia - Chronic Myeloid Leukemia (CML). 2018.
3. Huang X, Cortes J, Kantarjian H. Estimations of the increasing prevalence and plateau prevalence of chronic myeloid leukemia in the era of tyrosine kinase inhibitor therapy. Cancer. 2012;118:3123-3127.
4. Savage DG, Szydlo RM, Chase A, et al. Bone marrow transplantation for chronic myeloid leukaemia: the effects of differing criteria for defining chronic phase on probabilities of survival and relapse. Br J Haematol. 1997;99:30-35.
5. Knox WF, Bhavnani M, Davson J, Geary CG. Histological classification of chronic granulocytic leukaemia. Clin Lab Haematol. 1984;6:171-175.
6. Kvasnicka HM, Thiele J, Schmitt-Graeff A, et al. Impact of bone marrow morphology on multivariate risk classification in chronic myelogenous leukemia. Acta Haematol. 2003;109:53-56.
7. Cortes JE, Talpaz M, O’Brien S, et al. Staging of chronic myeloid leukemia in the imatinib era: an evaluation of the World Health Organization proposal. Cancer. 2006;106:1306-1315.
8. Druker BJ. Chronic myeloid leukemia In: DeVita VT, Lawrence TS, Rosenburg SA, eds. DeVita, Hellman, and Rosenberg’s Cancer Principles & Practice of Oncology. 8th ed. Philadelphia, PA: Lippincott, Williams and Wilkins; 2007:2267-2304.
9. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391-2405.
10. Fabarius A, Leitner A, Hochhaus A, et al. Impact of additional cytogenetic aberrations at diagnosis on prognosis of CML: long-term observation of 1151 patients from the randomized CML Study IV. Blood. 2011;118:6760-6768.
11. Alhuraiji A, Kantarjian H, Boddu P, et al. Prognostic significance of additional chromosomal abnormalities at the time of diagnosis in patients with chronic myeloid leukemia treated with frontline tyrosine kinase inhibitors. Am J Hematol. 2018;93:84-90.
12. Melo JV. BCR-ABL gene variants. Baillieres Clin Haematol. 1997;10:203-222.
13. Kantarjian HM, Talpaz M, Cortes J, et al. Quantitative polymerase chain reaction monitoring of BCR-ABL during therapy with imatinib mesylate (STI571; gleevec) in chronic-phase chronic myelogenous leukemia. Clin Cancer Res. 2003;9:160-166.
14. Hughes T, Deininger M, Hochhaus A, et al. Monitoring CML patients responding to treatment with tyrosine kinase inhibitors: review and recommendations for harmonizing current methodology for detecting BCR-ABL transcripts and kinase domain mutations and for expressing results. Blood. 2006;108:28-37.
15. Hochhaus A, Larson RA, Guilhot F, et al. Long-term outcomes of imatinib treatment for chronic myeloid leukemia. N Engl J Med. 2017;376:917-927.
16. Cortes JE, Saglio G, Kantarjian HM, et al. Final 5-year study results of DASISION: the Dasatinib Versus Imatinib Study in Treatment-Naive Chronic Myeloid Leukemia Patients trial. J Clin Oncol. 2016;34:2333-3340.
17. Hochhaus A, Saglio G, Hughes TP, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30:1044-1054.
18. Cortes JE, Gambacorti-Passerini C, Deininger MW, et al. Bosutinib versus imatinib for newly diagnosed chronic myeloid leukemia: results from the randomized BFORE trial. J Clin Oncol. 2018;36:231-237.
19. Radich JP, Deininger M, Abboud CN, et al. Chronic Myeloid Leukemia, Version 1.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:1108-1135.
20. Druker BJ, Talpaz M, Resta DJ, et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med. 2001;344:1031-1037.
21. Faderl S, Talpaz M, Estrov Z, Kantarjian HM. Chronic myelogenous leukemia: biology and therapy. Ann Intern Med. 1999;131:207-219.
22. O’Brien SG, Guilhot F, Larson RA, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003;348:994-1004.
23. Baccarani M, Deininger MW, Rosti G, et al. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood. 2013;122:872-884.
24. Larripa I, Ruiz MS, Gutierrez M, Bianchini M. [Guidelines for molecular monitoring of BCR-ABL1 in chronic myeloid leukemia patients by RT-qPCR.] Medicina (B Aires). 2017;77:61-72.
25. Marin D, Ibrahim AR, Lucas C, et al. Assessment of BCR-ABL1 transcript levels at 3 months is the only requirement for predicting outcome for patients with chronic myeloid leukemia treated with tyrosine kinase inhibitors. J Clin Oncol. 2012;30:232-238.
26. Hughes TP, Ross DM. Moving treatment-free remission into mainstream clinical practice in CML. Blood. 2016;128:17-23.
Immunotherapy’s cardiac effects require early monitoring, management
WASHINGTON – Unquestionably, immunotherapy is revolutionizing the care of patients with various solid tumors and hematologic malignancies.
But it’s equally true that there’s no such thing as either a free lunch or a cancer therapy free of side effects, whether it’s increased risk for heart failure associated with anthracycline-based chemotherapy, or inflammatory conditions, arrhythmias, and thromboembolic events associated with immune checkpoint inhibitors, said R. Frank Cornell, MD, of Vanderbilt University Medical Center in Nashville, Tenn.
“Early awareness and intervention is critical for improved outcomes, and a multidisciplinary approach between oncology, cardiology, the clinic nurse, and other health care providers is critical in managing these patients with these complicated therapies,” he said at the American College of Cardiology’s Advancing the Cardiovascular Care of the Oncology Patient meeting.
Checkpoint inhibitors and the heart
Toxicities associated with immune checkpoint inhibitors such as the programmed death 1/ligand 1 (PD-1/PD-L1) inhibitors nivolumab (Opdivo) and pembrolizumab (Keytruda) and the cytotoxic T-lymphocyte antigen 4 antibody ipilimumab (Yervoy) tend to mimic autoimmune conditions, Dr. Cornell said.
Cardiovascular events associated with these agents, while uncommon, include myocarditis, pericarditis, arrhythmias, impaired ventricular function with heart failure, vasculitis, and venous thromboembolism, he said, citing an American Society of Clinical Oncology (ASCO) clinical practice guideline (J Clin Oncol 2018;36[17]:1714-68).
Dr. Cornell described the case of a 63-year-old woman with disseminated metastatic melanoma who presented to the emergency department 10 days after starting on combination therapy with ipilimumab and nivolumab. She had developed shortness of breath, pleuritic chest pain, and a mild cough for 1 or 2 days.
Her cardiac laboratory markers had been normal at baseline, but were markedly elevated on presentation, and electrocardiograms showed complete heart block and subsequent ventricular tachycardia.
The patient was started on high-dose prednisone, but she died in hospital, and an autopsy showed that the cause of death was infiltration into the myocardium of CD3-positive and CD8-positive T lymphocytes.
“So how do we manage this? This is a good opportunity, I think, for further cardiology and oncology collaboration to develop more robust guidelines for what we can do to best prevent this,” Dr. Cornell said.
Patients started on the ipilimumab/nivolumab combination should be tested weekly for cardiac troponin, creatine kinase (CK) and CK-muscle/brain (CK-MB) weekly for the first 3-4 weeks of therapy. Therapy should be stopped if troponin levels continue to rise, and the patient should be started on high-dose steroids, he said.
The role of other anti-inflammatory agents such as infliximab (Remicade and biosimilars) is unclear and needs further study, he added.
Dr. Cornell cited a 2018 letter to The Lancet by Javid J. Moslehi, MD, and colleagues from Vanderbilt describing an increase in reports of fatal myocarditis among patients treated with checkpoint inhibitors.
“We highlight the high mortality rate with severe immune checkpoint inhibitor–related myocarditis, which is more frequent with combination PD-1 and CTLA-4 blockade, but can also occur with monotherapy. Myocarditis was observed across immune checkpoint inhibitor regimens, although it remains too early to determine whether the incidence differs between use of anti-PD1 and anti-PD-L1 drugs. Furthermore, this condition occurs early on during therapy and across cancer types,” they wrote.
Most of the patients had no preexisting cardiovascular disease, and most were not taking medications for hypertension, cardiovascular disease, or diabetes.
CAR-T cells and cardiac disease
The primary cardiac complications associated with CAR-T cell therapy are related to the cytokine release syndrome (CRS), a condition marked by progressive elevation in inflammatory cytokines that in turn leads to marked elevations in C-reactive protein (CRP), interferon gamma, tumor necrosis factor al, and release of pro-inflammatory cytokines including interleukin (IL) 6, IL-10, IL-12, and IL-1 beta.
In rare instances, CRS can lead to disseminated intravascular coagulation (DIC), capillary leak syndrome, and a hemophagocytic lymphohistiocytosis-like (HLH) syndrome, Dr. Cornell said.
Package inserts for the two Food and Drug Administration–approved CAR-T cell products, axicabtagene ciloleucel (Yescarta) and tisagenlecleucel (Kymriah) show that each was associated in clinical trials with a high incidence of CRS.
Among patients treated with axicabtagene ciloleucel, 94% developed CRS, which was grade 3 or greater in severity in 13%. The median time to onset was 2 days, and the median duration was 7 days. Cardiovascular adverse events included grade 3 or greater tachycardia in 2%, arrhythmias in 7%, edema in 1%, dyspnea in 3%, pleural effusion in 2%, hypotension in 15%, hypertension in 6%, and thrombosis in 1%.
Among patients treated with tisagenlecleucel, 79% treated for B-cell acute lymphoblastic leukemia (B-ALL) and 74% treated for diffuse large B cell lymphoma (DLBCL) developed CRS, which was grade 3 or greater in 49% and 23% of patients, respectively. The median time to onset was 3 days, and the median duration of CRS was 8 days.
Cardiovascular adverse events of grade 3 or greater among these patients included tachycardia in 4%, fluid overload in 7%, edema in 1%, dyspnea in 12%, pulmonary edema in 4%, hypotension in 22%, and hypertension in 6%.
Risk factors for CRS include high pre-infusion tumor burden, active infections, and concurrent inflammatory processes, Dr. Cornell said.
Prevention of cardiovascular complications of CAR-T cell therapy requires management of CRS. Patients with grade 2 or greater CRS should receive the anti-IL-6 agent tocilizumab (Actemra) 8 mg/kg intravenously over 1 hour to a maximum dose of 800 mg. Tocilizumab infusions can be repeated every 8 hours as needed if the patient is not responsive to intravenous fluids or increasing supplement oxygen, but should be limited to a maximum of three doses over 24 hours, and a maximum total of four doses.
Patients with grade 3 CRS should also receive intravenous methylprednisolone 1 mg/kg twice daily or the equivalent amount of dexamethasone, with corticosteroids continued until the severity of CRS is grade 1 or less, then tapered over 3 days,
Patients with grade 4 CRS should also receive IV methylprednisolone 1,000 mg per day for 3 days, and if symptoms improve, continue management as per grade 3, Dr. Cornell said.
Dr. Cornell reported having nothing to disclose.
WASHINGTON – Unquestionably, immunotherapy is revolutionizing the care of patients with various solid tumors and hematologic malignancies.
But it’s equally true that there’s no such thing as either a free lunch or a cancer therapy free of side effects, whether it’s increased risk for heart failure associated with anthracycline-based chemotherapy, or inflammatory conditions, arrhythmias, and thromboembolic events associated with immune checkpoint inhibitors, said R. Frank Cornell, MD, of Vanderbilt University Medical Center in Nashville, Tenn.
“Early awareness and intervention is critical for improved outcomes, and a multidisciplinary approach between oncology, cardiology, the clinic nurse, and other health care providers is critical in managing these patients with these complicated therapies,” he said at the American College of Cardiology’s Advancing the Cardiovascular Care of the Oncology Patient meeting.
Checkpoint inhibitors and the heart
Toxicities associated with immune checkpoint inhibitors such as the programmed death 1/ligand 1 (PD-1/PD-L1) inhibitors nivolumab (Opdivo) and pembrolizumab (Keytruda) and the cytotoxic T-lymphocyte antigen 4 antibody ipilimumab (Yervoy) tend to mimic autoimmune conditions, Dr. Cornell said.
Cardiovascular events associated with these agents, while uncommon, include myocarditis, pericarditis, arrhythmias, impaired ventricular function with heart failure, vasculitis, and venous thromboembolism, he said, citing an American Society of Clinical Oncology (ASCO) clinical practice guideline (J Clin Oncol 2018;36[17]:1714-68).
Dr. Cornell described the case of a 63-year-old woman with disseminated metastatic melanoma who presented to the emergency department 10 days after starting on combination therapy with ipilimumab and nivolumab. She had developed shortness of breath, pleuritic chest pain, and a mild cough for 1 or 2 days.
Her cardiac laboratory markers had been normal at baseline, but were markedly elevated on presentation, and electrocardiograms showed complete heart block and subsequent ventricular tachycardia.
The patient was started on high-dose prednisone, but she died in hospital, and an autopsy showed that the cause of death was infiltration into the myocardium of CD3-positive and CD8-positive T lymphocytes.
“So how do we manage this? This is a good opportunity, I think, for further cardiology and oncology collaboration to develop more robust guidelines for what we can do to best prevent this,” Dr. Cornell said.
Patients started on the ipilimumab/nivolumab combination should be tested weekly for cardiac troponin, creatine kinase (CK) and CK-muscle/brain (CK-MB) weekly for the first 3-4 weeks of therapy. Therapy should be stopped if troponin levels continue to rise, and the patient should be started on high-dose steroids, he said.
The role of other anti-inflammatory agents such as infliximab (Remicade and biosimilars) is unclear and needs further study, he added.
Dr. Cornell cited a 2018 letter to The Lancet by Javid J. Moslehi, MD, and colleagues from Vanderbilt describing an increase in reports of fatal myocarditis among patients treated with checkpoint inhibitors.
“We highlight the high mortality rate with severe immune checkpoint inhibitor–related myocarditis, which is more frequent with combination PD-1 and CTLA-4 blockade, but can also occur with monotherapy. Myocarditis was observed across immune checkpoint inhibitor regimens, although it remains too early to determine whether the incidence differs between use of anti-PD1 and anti-PD-L1 drugs. Furthermore, this condition occurs early on during therapy and across cancer types,” they wrote.
Most of the patients had no preexisting cardiovascular disease, and most were not taking medications for hypertension, cardiovascular disease, or diabetes.
CAR-T cells and cardiac disease
The primary cardiac complications associated with CAR-T cell therapy are related to the cytokine release syndrome (CRS), a condition marked by progressive elevation in inflammatory cytokines that in turn leads to marked elevations in C-reactive protein (CRP), interferon gamma, tumor necrosis factor al, and release of pro-inflammatory cytokines including interleukin (IL) 6, IL-10, IL-12, and IL-1 beta.
In rare instances, CRS can lead to disseminated intravascular coagulation (DIC), capillary leak syndrome, and a hemophagocytic lymphohistiocytosis-like (HLH) syndrome, Dr. Cornell said.
Package inserts for the two Food and Drug Administration–approved CAR-T cell products, axicabtagene ciloleucel (Yescarta) and tisagenlecleucel (Kymriah) show that each was associated in clinical trials with a high incidence of CRS.
Among patients treated with axicabtagene ciloleucel, 94% developed CRS, which was grade 3 or greater in severity in 13%. The median time to onset was 2 days, and the median duration was 7 days. Cardiovascular adverse events included grade 3 or greater tachycardia in 2%, arrhythmias in 7%, edema in 1%, dyspnea in 3%, pleural effusion in 2%, hypotension in 15%, hypertension in 6%, and thrombosis in 1%.
Among patients treated with tisagenlecleucel, 79% treated for B-cell acute lymphoblastic leukemia (B-ALL) and 74% treated for diffuse large B cell lymphoma (DLBCL) developed CRS, which was grade 3 or greater in 49% and 23% of patients, respectively. The median time to onset was 3 days, and the median duration of CRS was 8 days.
Cardiovascular adverse events of grade 3 or greater among these patients included tachycardia in 4%, fluid overload in 7%, edema in 1%, dyspnea in 12%, pulmonary edema in 4%, hypotension in 22%, and hypertension in 6%.
Risk factors for CRS include high pre-infusion tumor burden, active infections, and concurrent inflammatory processes, Dr. Cornell said.
Prevention of cardiovascular complications of CAR-T cell therapy requires management of CRS. Patients with grade 2 or greater CRS should receive the anti-IL-6 agent tocilizumab (Actemra) 8 mg/kg intravenously over 1 hour to a maximum dose of 800 mg. Tocilizumab infusions can be repeated every 8 hours as needed if the patient is not responsive to intravenous fluids or increasing supplement oxygen, but should be limited to a maximum of three doses over 24 hours, and a maximum total of four doses.
Patients with grade 3 CRS should also receive intravenous methylprednisolone 1 mg/kg twice daily or the equivalent amount of dexamethasone, with corticosteroids continued until the severity of CRS is grade 1 or less, then tapered over 3 days,
Patients with grade 4 CRS should also receive IV methylprednisolone 1,000 mg per day for 3 days, and if symptoms improve, continue management as per grade 3, Dr. Cornell said.
Dr. Cornell reported having nothing to disclose.
WASHINGTON – Unquestionably, immunotherapy is revolutionizing the care of patients with various solid tumors and hematologic malignancies.
But it’s equally true that there’s no such thing as either a free lunch or a cancer therapy free of side effects, whether it’s increased risk for heart failure associated with anthracycline-based chemotherapy, or inflammatory conditions, arrhythmias, and thromboembolic events associated with immune checkpoint inhibitors, said R. Frank Cornell, MD, of Vanderbilt University Medical Center in Nashville, Tenn.
“Early awareness and intervention is critical for improved outcomes, and a multidisciplinary approach between oncology, cardiology, the clinic nurse, and other health care providers is critical in managing these patients with these complicated therapies,” he said at the American College of Cardiology’s Advancing the Cardiovascular Care of the Oncology Patient meeting.
Checkpoint inhibitors and the heart
Toxicities associated with immune checkpoint inhibitors such as the programmed death 1/ligand 1 (PD-1/PD-L1) inhibitors nivolumab (Opdivo) and pembrolizumab (Keytruda) and the cytotoxic T-lymphocyte antigen 4 antibody ipilimumab (Yervoy) tend to mimic autoimmune conditions, Dr. Cornell said.
Cardiovascular events associated with these agents, while uncommon, include myocarditis, pericarditis, arrhythmias, impaired ventricular function with heart failure, vasculitis, and venous thromboembolism, he said, citing an American Society of Clinical Oncology (ASCO) clinical practice guideline (J Clin Oncol 2018;36[17]:1714-68).
Dr. Cornell described the case of a 63-year-old woman with disseminated metastatic melanoma who presented to the emergency department 10 days after starting on combination therapy with ipilimumab and nivolumab. She had developed shortness of breath, pleuritic chest pain, and a mild cough for 1 or 2 days.
Her cardiac laboratory markers had been normal at baseline, but were markedly elevated on presentation, and electrocardiograms showed complete heart block and subsequent ventricular tachycardia.
The patient was started on high-dose prednisone, but she died in hospital, and an autopsy showed that the cause of death was infiltration into the myocardium of CD3-positive and CD8-positive T lymphocytes.
“So how do we manage this? This is a good opportunity, I think, for further cardiology and oncology collaboration to develop more robust guidelines for what we can do to best prevent this,” Dr. Cornell said.
Patients started on the ipilimumab/nivolumab combination should be tested weekly for cardiac troponin, creatine kinase (CK) and CK-muscle/brain (CK-MB) weekly for the first 3-4 weeks of therapy. Therapy should be stopped if troponin levels continue to rise, and the patient should be started on high-dose steroids, he said.
The role of other anti-inflammatory agents such as infliximab (Remicade and biosimilars) is unclear and needs further study, he added.
Dr. Cornell cited a 2018 letter to The Lancet by Javid J. Moslehi, MD, and colleagues from Vanderbilt describing an increase in reports of fatal myocarditis among patients treated with checkpoint inhibitors.
“We highlight the high mortality rate with severe immune checkpoint inhibitor–related myocarditis, which is more frequent with combination PD-1 and CTLA-4 blockade, but can also occur with monotherapy. Myocarditis was observed across immune checkpoint inhibitor regimens, although it remains too early to determine whether the incidence differs between use of anti-PD1 and anti-PD-L1 drugs. Furthermore, this condition occurs early on during therapy and across cancer types,” they wrote.
Most of the patients had no preexisting cardiovascular disease, and most were not taking medications for hypertension, cardiovascular disease, or diabetes.
CAR-T cells and cardiac disease
The primary cardiac complications associated with CAR-T cell therapy are related to the cytokine release syndrome (CRS), a condition marked by progressive elevation in inflammatory cytokines that in turn leads to marked elevations in C-reactive protein (CRP), interferon gamma, tumor necrosis factor al, and release of pro-inflammatory cytokines including interleukin (IL) 6, IL-10, IL-12, and IL-1 beta.
In rare instances, CRS can lead to disseminated intravascular coagulation (DIC), capillary leak syndrome, and a hemophagocytic lymphohistiocytosis-like (HLH) syndrome, Dr. Cornell said.
Package inserts for the two Food and Drug Administration–approved CAR-T cell products, axicabtagene ciloleucel (Yescarta) and tisagenlecleucel (Kymriah) show that each was associated in clinical trials with a high incidence of CRS.
Among patients treated with axicabtagene ciloleucel, 94% developed CRS, which was grade 3 or greater in severity in 13%. The median time to onset was 2 days, and the median duration was 7 days. Cardiovascular adverse events included grade 3 or greater tachycardia in 2%, arrhythmias in 7%, edema in 1%, dyspnea in 3%, pleural effusion in 2%, hypotension in 15%, hypertension in 6%, and thrombosis in 1%.
Among patients treated with tisagenlecleucel, 79% treated for B-cell acute lymphoblastic leukemia (B-ALL) and 74% treated for diffuse large B cell lymphoma (DLBCL) developed CRS, which was grade 3 or greater in 49% and 23% of patients, respectively. The median time to onset was 3 days, and the median duration of CRS was 8 days.
Cardiovascular adverse events of grade 3 or greater among these patients included tachycardia in 4%, fluid overload in 7%, edema in 1%, dyspnea in 12%, pulmonary edema in 4%, hypotension in 22%, and hypertension in 6%.
Risk factors for CRS include high pre-infusion tumor burden, active infections, and concurrent inflammatory processes, Dr. Cornell said.
Prevention of cardiovascular complications of CAR-T cell therapy requires management of CRS. Patients with grade 2 or greater CRS should receive the anti-IL-6 agent tocilizumab (Actemra) 8 mg/kg intravenously over 1 hour to a maximum dose of 800 mg. Tocilizumab infusions can be repeated every 8 hours as needed if the patient is not responsive to intravenous fluids or increasing supplement oxygen, but should be limited to a maximum of three doses over 24 hours, and a maximum total of four doses.
Patients with grade 3 CRS should also receive intravenous methylprednisolone 1 mg/kg twice daily or the equivalent amount of dexamethasone, with corticosteroids continued until the severity of CRS is grade 1 or less, then tapered over 3 days,
Patients with grade 4 CRS should also receive IV methylprednisolone 1,000 mg per day for 3 days, and if symptoms improve, continue management as per grade 3, Dr. Cornell said.
Dr. Cornell reported having nothing to disclose.
REPORTING FROM ACC CARDIO-ONCOLOGY
Key clinical point: Monitor for cardiac symptoms and treat or interrupt immunotherapy as needed.
Major finding: Immune checkpoint inhibitors and CAR T-cell therapies are associated with distinct cardiovascular adverse events.
Study details: Review of strategies for managing the cardiovascular consequences of cancer immunotherapies.
Disclosures: Dr. Cornell reported having nothing to disclose.
FDA approves ibrutinib plus obinutuzumab for CLL/SLL
The Food and Drug Administration has approved ibrutinib (Imbruvica) for use in combination with obinutuzumab to treat adults with previously untreated chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL).
This is the tenth FDA approval for ibrutinib, a Bruton tyrosine kinase inhibitor jointly developed and commercialized by Pharmacyclics, an AbbVie company, and Janssen Biotech.
The approval is supported by the phase 3 iLLUMINATE trial (NCT02264574).
Results from this study were recently presented at the annual meeting of the American Society of Hematology (Blood. 2018;132:691) and published in the Lancet Oncology (2019 Jan;20[1]:43-56).
The iLLUMINATE trial enrolled newly diagnosed CLL patients who were randomized to receive ibrutinib plus obinutuzumab (n = 113) or chlorambucil plus obinutuzumab (n = 116).
The median follow-up was 31.3 months. The overall response rate was 88% in the ibrutinib arm and 73% in the chlorambucil arm. The complete response rate, including complete response with incomplete marrow recovery, was 19% and 8%, respectively.
The median progression-free survival was not reached in the ibrutinib arm and was 19.0 months in the chlorambucil arm (hazard ratio, 0.23; 95% confidence interval, 0.15-0.37; P less than .0001). The estimated 30-month progression-free survival was 79% and 31%, respectively.
The most common grade 3/4 adverse events in both arms were neutropenia (36% in the ibrutinib arm and 46% in the chlorambucil arm) and thrombocytopenia (19% and 10%, respectively).
There were 10 deaths caused by adverse events in the ibrutinib arm and 3 in the chlorambucil arm. One death was considered possibly related to ibrutinib (sudden death), and another was considered possibly related to chlorambucil (neuroendocrine carcinoma of the skin).
The Food and Drug Administration has approved ibrutinib (Imbruvica) for use in combination with obinutuzumab to treat adults with previously untreated chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL).
This is the tenth FDA approval for ibrutinib, a Bruton tyrosine kinase inhibitor jointly developed and commercialized by Pharmacyclics, an AbbVie company, and Janssen Biotech.
The approval is supported by the phase 3 iLLUMINATE trial (NCT02264574).
Results from this study were recently presented at the annual meeting of the American Society of Hematology (Blood. 2018;132:691) and published in the Lancet Oncology (2019 Jan;20[1]:43-56).
The iLLUMINATE trial enrolled newly diagnosed CLL patients who were randomized to receive ibrutinib plus obinutuzumab (n = 113) or chlorambucil plus obinutuzumab (n = 116).
The median follow-up was 31.3 months. The overall response rate was 88% in the ibrutinib arm and 73% in the chlorambucil arm. The complete response rate, including complete response with incomplete marrow recovery, was 19% and 8%, respectively.
The median progression-free survival was not reached in the ibrutinib arm and was 19.0 months in the chlorambucil arm (hazard ratio, 0.23; 95% confidence interval, 0.15-0.37; P less than .0001). The estimated 30-month progression-free survival was 79% and 31%, respectively.
The most common grade 3/4 adverse events in both arms were neutropenia (36% in the ibrutinib arm and 46% in the chlorambucil arm) and thrombocytopenia (19% and 10%, respectively).
There were 10 deaths caused by adverse events in the ibrutinib arm and 3 in the chlorambucil arm. One death was considered possibly related to ibrutinib (sudden death), and another was considered possibly related to chlorambucil (neuroendocrine carcinoma of the skin).
The Food and Drug Administration has approved ibrutinib (Imbruvica) for use in combination with obinutuzumab to treat adults with previously untreated chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL).
This is the tenth FDA approval for ibrutinib, a Bruton tyrosine kinase inhibitor jointly developed and commercialized by Pharmacyclics, an AbbVie company, and Janssen Biotech.
The approval is supported by the phase 3 iLLUMINATE trial (NCT02264574).
Results from this study were recently presented at the annual meeting of the American Society of Hematology (Blood. 2018;132:691) and published in the Lancet Oncology (2019 Jan;20[1]:43-56).
The iLLUMINATE trial enrolled newly diagnosed CLL patients who were randomized to receive ibrutinib plus obinutuzumab (n = 113) or chlorambucil plus obinutuzumab (n = 116).
The median follow-up was 31.3 months. The overall response rate was 88% in the ibrutinib arm and 73% in the chlorambucil arm. The complete response rate, including complete response with incomplete marrow recovery, was 19% and 8%, respectively.
The median progression-free survival was not reached in the ibrutinib arm and was 19.0 months in the chlorambucil arm (hazard ratio, 0.23; 95% confidence interval, 0.15-0.37; P less than .0001). The estimated 30-month progression-free survival was 79% and 31%, respectively.
The most common grade 3/4 adverse events in both arms were neutropenia (36% in the ibrutinib arm and 46% in the chlorambucil arm) and thrombocytopenia (19% and 10%, respectively).
There were 10 deaths caused by adverse events in the ibrutinib arm and 3 in the chlorambucil arm. One death was considered possibly related to ibrutinib (sudden death), and another was considered possibly related to chlorambucil (neuroendocrine carcinoma of the skin).
EC approves blinatumomab for MRD-positive BCP-ALL
The European Commission (EC) has expanded the approved indication for blinatumomab (Blincyto).
The drug is now approved in Europe to treat adults with Philadelphia chromosome–negative (Ph–), CD19-positive B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in first or second complete remission with minimal residual disease (MRD) of at least 0.1%.
Blinatumomab is already approved in Europe to treat adults with Ph–, CD19-positive relapsed/refractory BCP-ALL and children aged 1 year or older who have relapsed/refractory Ph–, CD19-positive BCP-ALL and have received at least two prior therapies or relapsed after allogeneic hematopoietic stem cell transplant.
The drug was approved in the United States in March 2018 for the treatment of adults and children with BCP-ALL in first or second complete remission with MRD of at least 0.1%.
The EC’s decision to approve blinatumomab in MRD-positive patients was supported by the phase 2 BLAST trial (Blood. 2018;131[14]:1522-31).
The EC’s approval is also based on a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP).
That opinion, issued in November 2018, was a reversal of the opinion the committee issued in July 2018. At that time, the CHMP said the available data did not support approval for blinatumomab to treat MRD-positive BCP-ALL.
The CHMP acknowledged that blinatumomab produced MRD negativity in many patients in the BLAST trial but said there was no strong evidence that this led to improved survival. As a result, the CHMP said the benefits of blinatumomab do not outweigh its risks in MRD-positive BCP-ALL patients.
However, Amgen requested a reexamination of the CHMP’s opinion. During the reexamination, the CHMP reviewed all the data and consulted a group of experts.
The experts echoed the CHMP’s prior sentiment that there was no strong evidence of improved survival in MRD-positive patients treated with blinatumomab. However, they also said the data indicate a good response to blinatumomab, with around 78% of patients becoming negative for MRD after treatment.
Noting that MRD-positive patients have a high risk of relapse and few treatment options, the CHMP concluded that the benefits of blinatumomab outweigh its risks in this patient population.
The CHMP recommended expanding the approved indication for blinatumomab but also requested that Amgen provide data from ongoing studies of the drug in MRD-positive patients.
The European Commission (EC) has expanded the approved indication for blinatumomab (Blincyto).
The drug is now approved in Europe to treat adults with Philadelphia chromosome–negative (Ph–), CD19-positive B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in first or second complete remission with minimal residual disease (MRD) of at least 0.1%.
Blinatumomab is already approved in Europe to treat adults with Ph–, CD19-positive relapsed/refractory BCP-ALL and children aged 1 year or older who have relapsed/refractory Ph–, CD19-positive BCP-ALL and have received at least two prior therapies or relapsed after allogeneic hematopoietic stem cell transplant.
The drug was approved in the United States in March 2018 for the treatment of adults and children with BCP-ALL in first or second complete remission with MRD of at least 0.1%.
The EC’s decision to approve blinatumomab in MRD-positive patients was supported by the phase 2 BLAST trial (Blood. 2018;131[14]:1522-31).
The EC’s approval is also based on a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP).
That opinion, issued in November 2018, was a reversal of the opinion the committee issued in July 2018. At that time, the CHMP said the available data did not support approval for blinatumomab to treat MRD-positive BCP-ALL.
The CHMP acknowledged that blinatumomab produced MRD negativity in many patients in the BLAST trial but said there was no strong evidence that this led to improved survival. As a result, the CHMP said the benefits of blinatumomab do not outweigh its risks in MRD-positive BCP-ALL patients.
However, Amgen requested a reexamination of the CHMP’s opinion. During the reexamination, the CHMP reviewed all the data and consulted a group of experts.
The experts echoed the CHMP’s prior sentiment that there was no strong evidence of improved survival in MRD-positive patients treated with blinatumomab. However, they also said the data indicate a good response to blinatumomab, with around 78% of patients becoming negative for MRD after treatment.
Noting that MRD-positive patients have a high risk of relapse and few treatment options, the CHMP concluded that the benefits of blinatumomab outweigh its risks in this patient population.
The CHMP recommended expanding the approved indication for blinatumomab but also requested that Amgen provide data from ongoing studies of the drug in MRD-positive patients.
The European Commission (EC) has expanded the approved indication for blinatumomab (Blincyto).
The drug is now approved in Europe to treat adults with Philadelphia chromosome–negative (Ph–), CD19-positive B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in first or second complete remission with minimal residual disease (MRD) of at least 0.1%.
Blinatumomab is already approved in Europe to treat adults with Ph–, CD19-positive relapsed/refractory BCP-ALL and children aged 1 year or older who have relapsed/refractory Ph–, CD19-positive BCP-ALL and have received at least two prior therapies or relapsed after allogeneic hematopoietic stem cell transplant.
The drug was approved in the United States in March 2018 for the treatment of adults and children with BCP-ALL in first or second complete remission with MRD of at least 0.1%.
The EC’s decision to approve blinatumomab in MRD-positive patients was supported by the phase 2 BLAST trial (Blood. 2018;131[14]:1522-31).
The EC’s approval is also based on a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP).
That opinion, issued in November 2018, was a reversal of the opinion the committee issued in July 2018. At that time, the CHMP said the available data did not support approval for blinatumomab to treat MRD-positive BCP-ALL.
The CHMP acknowledged that blinatumomab produced MRD negativity in many patients in the BLAST trial but said there was no strong evidence that this led to improved survival. As a result, the CHMP said the benefits of blinatumomab do not outweigh its risks in MRD-positive BCP-ALL patients.
However, Amgen requested a reexamination of the CHMP’s opinion. During the reexamination, the CHMP reviewed all the data and consulted a group of experts.
The experts echoed the CHMP’s prior sentiment that there was no strong evidence of improved survival in MRD-positive patients treated with blinatumomab. However, they also said the data indicate a good response to blinatumomab, with around 78% of patients becoming negative for MRD after treatment.
Noting that MRD-positive patients have a high risk of relapse and few treatment options, the CHMP concluded that the benefits of blinatumomab outweigh its risks in this patient population.
The CHMP recommended expanding the approved indication for blinatumomab but also requested that Amgen provide data from ongoing studies of the drug in MRD-positive patients.
Uninterrupted ibrutinib with CAR T could improve CLL outcomes
SAN DIEGO – Ibrutinib treatment continued before, during, and after infusion of the CD19-specific chimeric antigen receptor (CAR) T-cell therapy JCAR014 in patients with relapsed or refractory chronic lymphocytic leukemia (CLL) appears to improve patient responses and decrease the risk of severe cytokine release syndrome.
The findings come from a comparison of sequential cohorts from a phase 1/2 study.
At 4 weeks after infusion, the approach was highly efficacious; overall response rates by 2008 International Workshop on CLL (IWCLL) criteria were 83% in 24 patients who received the uninterrupted ibrutinib regimen along with the JCAR014 therapy – a combination of CD4 and CD8 T cells – and 65% in 19 patients from a prior cohort who did not receive continuous ibrutinib, Jordan Gauthier, MD, reported at the annual meeting of the American Society of Hematology.
Concurrent ibrutinib was generally well tolerated, with 13 of 19 patients in the ibrutinib cohort receiving treatment as planned without discontinuation. The rates of grade 1 or higher cytokine release syndrome (CRS) were statistically similar in the ibrutinib and no-ibrutinib cohorts (74% and 92%, respectively). However, the rates of severe CRS (grade 3 or higher) were, strikingly, 0% and 25%, respectively, said Dr. Gauthier, a senior fellow in the Turtle Lab at Fred Hutchinson Cancer Center, Seattle.
Neurotoxicity occurred in 32% and 42% of patients in the groups; severe neurotoxicity occurred in 26% and 29%, respectively.
In the ibrutinib cohort, one patient with grade 2 CRS developed fatal presumed cardiac arrhythmia; in the no-ibrutinib cohort, one patient died from a CAR T cell–related toxicity.
Notably, a trend toward better expansion of CD8 CAR T cells and a significantly greater expansion of CD4 CAR T cells was observed in the ibrutinib cohort, he said.
The study was designed to assess JCAR014, and based on the initial cohort findings published in 2017, established a regimen of cyclophosphamide and fludarabine (Cy/Flu) lymphodepletion followed by JCAR014 infusion at 2 x 106 CAR T cells/kg. The study was not a randomized, head-to-head comparison but the groups were similar with respect to both patient and disease characteristics, Dr. Gauthier noted.
The outcomes in the first cohort were then compared retrospectively with those from the subsequent cohort of patients who received Cy/Flu with 2 x 106 CAR T cells/kg with concurrent ibrutinib administered at 420 mg per day from at least 2 weeks prior to leukapheresis until at least 3 months after JCAR014 infusion.
The rationale for uninterrupted ibrutinib in relapsed/refractory CLL patients receiving JCAR014 included potential prevention of tumor flare, mobilization of CLL cells into the blood from the lymph nodes, improvement of CAR T-cell function, and a decrease in CAR T-cell related toxicity, he said.
The concurrent administration of ibrutinib and JCAR014 was feasible for most patients. “[It] induced high response rates and deep responses early on at 4 weeks, and it was associated with higher in vivo expansion of CD4 CAR T cells and with lower rates of severe toxicity,” Dr. Gauthier said. “The next step is to hopefully validate these findings in a prospective phase 1/2 study.”
Dr. Gauthier reported having no financial disclosures.
SOURCE: Gauthier J et al. ASH 18, Abstract 299.
SAN DIEGO – Ibrutinib treatment continued before, during, and after infusion of the CD19-specific chimeric antigen receptor (CAR) T-cell therapy JCAR014 in patients with relapsed or refractory chronic lymphocytic leukemia (CLL) appears to improve patient responses and decrease the risk of severe cytokine release syndrome.
The findings come from a comparison of sequential cohorts from a phase 1/2 study.
At 4 weeks after infusion, the approach was highly efficacious; overall response rates by 2008 International Workshop on CLL (IWCLL) criteria were 83% in 24 patients who received the uninterrupted ibrutinib regimen along with the JCAR014 therapy – a combination of CD4 and CD8 T cells – and 65% in 19 patients from a prior cohort who did not receive continuous ibrutinib, Jordan Gauthier, MD, reported at the annual meeting of the American Society of Hematology.
Concurrent ibrutinib was generally well tolerated, with 13 of 19 patients in the ibrutinib cohort receiving treatment as planned without discontinuation. The rates of grade 1 or higher cytokine release syndrome (CRS) were statistically similar in the ibrutinib and no-ibrutinib cohorts (74% and 92%, respectively). However, the rates of severe CRS (grade 3 or higher) were, strikingly, 0% and 25%, respectively, said Dr. Gauthier, a senior fellow in the Turtle Lab at Fred Hutchinson Cancer Center, Seattle.
Neurotoxicity occurred in 32% and 42% of patients in the groups; severe neurotoxicity occurred in 26% and 29%, respectively.
In the ibrutinib cohort, one patient with grade 2 CRS developed fatal presumed cardiac arrhythmia; in the no-ibrutinib cohort, one patient died from a CAR T cell–related toxicity.
Notably, a trend toward better expansion of CD8 CAR T cells and a significantly greater expansion of CD4 CAR T cells was observed in the ibrutinib cohort, he said.
The study was designed to assess JCAR014, and based on the initial cohort findings published in 2017, established a regimen of cyclophosphamide and fludarabine (Cy/Flu) lymphodepletion followed by JCAR014 infusion at 2 x 106 CAR T cells/kg. The study was not a randomized, head-to-head comparison but the groups were similar with respect to both patient and disease characteristics, Dr. Gauthier noted.
The outcomes in the first cohort were then compared retrospectively with those from the subsequent cohort of patients who received Cy/Flu with 2 x 106 CAR T cells/kg with concurrent ibrutinib administered at 420 mg per day from at least 2 weeks prior to leukapheresis until at least 3 months after JCAR014 infusion.
The rationale for uninterrupted ibrutinib in relapsed/refractory CLL patients receiving JCAR014 included potential prevention of tumor flare, mobilization of CLL cells into the blood from the lymph nodes, improvement of CAR T-cell function, and a decrease in CAR T-cell related toxicity, he said.
The concurrent administration of ibrutinib and JCAR014 was feasible for most patients. “[It] induced high response rates and deep responses early on at 4 weeks, and it was associated with higher in vivo expansion of CD4 CAR T cells and with lower rates of severe toxicity,” Dr. Gauthier said. “The next step is to hopefully validate these findings in a prospective phase 1/2 study.”
Dr. Gauthier reported having no financial disclosures.
SOURCE: Gauthier J et al. ASH 18, Abstract 299.
SAN DIEGO – Ibrutinib treatment continued before, during, and after infusion of the CD19-specific chimeric antigen receptor (CAR) T-cell therapy JCAR014 in patients with relapsed or refractory chronic lymphocytic leukemia (CLL) appears to improve patient responses and decrease the risk of severe cytokine release syndrome.
The findings come from a comparison of sequential cohorts from a phase 1/2 study.
At 4 weeks after infusion, the approach was highly efficacious; overall response rates by 2008 International Workshop on CLL (IWCLL) criteria were 83% in 24 patients who received the uninterrupted ibrutinib regimen along with the JCAR014 therapy – a combination of CD4 and CD8 T cells – and 65% in 19 patients from a prior cohort who did not receive continuous ibrutinib, Jordan Gauthier, MD, reported at the annual meeting of the American Society of Hematology.
Concurrent ibrutinib was generally well tolerated, with 13 of 19 patients in the ibrutinib cohort receiving treatment as planned without discontinuation. The rates of grade 1 or higher cytokine release syndrome (CRS) were statistically similar in the ibrutinib and no-ibrutinib cohorts (74% and 92%, respectively). However, the rates of severe CRS (grade 3 or higher) were, strikingly, 0% and 25%, respectively, said Dr. Gauthier, a senior fellow in the Turtle Lab at Fred Hutchinson Cancer Center, Seattle.
Neurotoxicity occurred in 32% and 42% of patients in the groups; severe neurotoxicity occurred in 26% and 29%, respectively.
In the ibrutinib cohort, one patient with grade 2 CRS developed fatal presumed cardiac arrhythmia; in the no-ibrutinib cohort, one patient died from a CAR T cell–related toxicity.
Notably, a trend toward better expansion of CD8 CAR T cells and a significantly greater expansion of CD4 CAR T cells was observed in the ibrutinib cohort, he said.
The study was designed to assess JCAR014, and based on the initial cohort findings published in 2017, established a regimen of cyclophosphamide and fludarabine (Cy/Flu) lymphodepletion followed by JCAR014 infusion at 2 x 106 CAR T cells/kg. The study was not a randomized, head-to-head comparison but the groups were similar with respect to both patient and disease characteristics, Dr. Gauthier noted.
The outcomes in the first cohort were then compared retrospectively with those from the subsequent cohort of patients who received Cy/Flu with 2 x 106 CAR T cells/kg with concurrent ibrutinib administered at 420 mg per day from at least 2 weeks prior to leukapheresis until at least 3 months after JCAR014 infusion.
The rationale for uninterrupted ibrutinib in relapsed/refractory CLL patients receiving JCAR014 included potential prevention of tumor flare, mobilization of CLL cells into the blood from the lymph nodes, improvement of CAR T-cell function, and a decrease in CAR T-cell related toxicity, he said.
The concurrent administration of ibrutinib and JCAR014 was feasible for most patients. “[It] induced high response rates and deep responses early on at 4 weeks, and it was associated with higher in vivo expansion of CD4 CAR T cells and with lower rates of severe toxicity,” Dr. Gauthier said. “The next step is to hopefully validate these findings in a prospective phase 1/2 study.”
Dr. Gauthier reported having no financial disclosures.
SOURCE: Gauthier J et al. ASH 18, Abstract 299.
REPORTING FROM ASH 2018
Key clinical point:
Major finding: Severe cytokine release syndrome occurred in 0% versus 25% of patients in the ibrutinib and no-ibrutinib cohorts, respectively.
Study details: A retrospective comparison of 43 patients in two cohorts from a phase 1/2 study.
Disclosures: Dr. Gauthier reported having no financial disclosures.
Source: Gauthier J et al. ASH 2018, Abstract 299.
Aplastic Anemia: Current Treatment
Aplastic anemia is a rare hematologic disorder marked by pancytopenia and a hypocellular marrow. Aplastic anemia results from either inherited or acquired causes, and the treatment approach varies significantly between the 2 causes. This article reviews the treatment of inherited and acquired forms of aplastic anemia. The approach to evaluation and diagnosis of aplastic anemia is reviewed in a separate article.
Inherited Aplastic Anemia
First-line treatment options for patients with inherited marrow failure syndromes (IMFS) are androgen therapy and hematopoietic stem cell transplant (HSCT). When evaluating patients for HSCT, it is critical to identify the presence of an IMFS, as the risk and mortality associated with the conditioning regimen, stem cell source, graft-versus-host disease (GVHD), and secondary malignancies differ between patients with IMFS and those with acquired marrow failure syndromes or hematologic malignancies.
Potential sibling donors need to be screened for donor candidacy as well as for the inherited defect.1 Among patients with Fanconi anemia or a telomere biology disorder, the stem cell source must be considered, with bone marrow demonstrating lower rates of acute GVHD than a peripheral blood stem cell source.2-4 In IMFS patients, the donor cell type may affect the choice of conditioning regimen.5,6 Reduced-intensity conditioning in lieu of myeloablative conditioning without total body irradiation has proved feasible in patients with Fanconi anemia, and is associated with a reduced risk of secondary malignancies.5,6 Incorporation of fludarabine in the conditioning regimen of patients without a matched sibling donor is associated with superior engraftment and survival2,5,7 compared to cyclophosphamide conditioning, which was historically used in matched related donors.6,8 The addition of fludarabine appears to be especially beneficial in older patients, in whom its use is associated with lower rates of graft failure, likely due to increased immunosuppression at the time of engraftment.7,9 Fludarabine has also been incorporated into conditioning regimens for patients with a telomere biology disorder, but outcomes data is limited.5
For patients presenting with acute myeloid leukemia (AML) or a high-risk myelodysplastic syndrome (MDS) who are subsequently diagnosed with an IMFS, treatment can be more complex, as these patients are at high risk for toxicity from standard chemotherapy. Limited data suggests that induction therapy and transplantation are feasible in this group of patients, and this approach is associated with increased overall survival (OS) despite lower OS rates than those of IMFS patients who present prior to the development of MDS or AML.10,11 Further work is needed to determine the optimal induction regimen that balances the risks of treatment-related mortality and complications associated with conditioning regimens, risk of relapse, and risk of secondary malignancies, especially in the cohort of patients diagnosed at an older age.
Acquired Aplastic Anemia
Supportive Care
While the workup and treatment plan is being established, attention should be directed at supportive care for prevention of complications. The most common complications leading to death in patients with significant pancytopenia and neutropenia are opportunistic infections and hemorrhagic complications.12
Transfusion support is critical to avoid symptomatic anemia and hemorrhagic complications related to thrombocytopenia, which typically occur with platelet counts lower than 10,000 cells/µL. However, transfusion carries the risk of alloimmunization (which may persist for years following transfusion) and transfusion-related graft versus host disease (trGVHD), and thus use of transfusion should be minimized when possible.13,14 Transfusion support is often required to prevent complications associated with thrombocytopenia and anemia; all blood products given to patients with aplastic anemia should be irradiated and leukoreduced to reduce the risk of both alloimmunization and trGVHD. Guidelines from the British Society for Haematology recommend routine screening for Rh and Kell antibodies to reduce the risk of alloimmunization.15 Infectious complications remain a common cause of morbidity and mortality in patients with aplastic anemia who have prolonged neutropenia (defined as an absolute neutrophil count [ANC] < 500 cells/µL).16-19 Therefore, patients should receive broad-spectrum antibiotics with antipseudomonal coverage. In a study by Tichelli and colleagues evaluating the role of granulocyte-colony stimulating factor (G-CSF) in patients with SAA receiving immunosuppressive therapy, 55% of all patient deaths were secondary to infection.20 There was no OS benefit seen in patients who received G-CSF, though a significantly lower rate of infection was observed in the G-CSF arm compared to those not receiving G-CSF (56% versus 81%, P = 0.006).This difference was largely driven by a decrease in infectious episodes in patients with very severe aplastic anemia (VSAA) treated with G-CSF as compared to those who did not receive this therapy (22% versus 48%, P = 0.014).20
Angio-invasive pulmonary aspergillosis and Zygomycetes (eg, Rhizopus, Mucor species) remain major causes of mortality related to opportunistic mycotic infections in patients with aplastic anemia.18 The infectious risk is directly related to the duration and severity of neutropenia, with one study demonstrating a significant increase in risk in AML patients with neutropenia lasting longer than 3 weeks.21 Invasive fungal infections carry a high mortality in patients with severe neutropenia, though due to earlier recognition and empiric antifungal therapy with extended-spectrum azoles, overall mortality secondary to invasive fungal infections is declining.19,22
While neutropenia related to cytotoxic chemotherapy is commonly associated with gram-negative bacteria due to disruption of mucosal barriers, patients with aplastic anemia have an increased incidence of gram-positive bacteremia with staphylococcal species compared to other neutropenic populations.18,19 This appears to be changing with time. Valdez and colleagues demonstrated a decrease in prevalence of coagulase-negative staphylococcal infections, increased prevalence of gram-positive bacilli bacteremia, and no change in prevalence of gram-negative bacteremia in patients with aplastic anemia treated between 1989 and 2008.22 Gram-negative bacteremia caused by Stenotrophomonas maltophila, Escherichia coli, Klebsiella pneumoniae, Citrobacter, and Proteus has also been reported.19 Despite a lack of clinical trials investigating the role of antifungal and antibacterial prophylaxis for patients with aplastic anemia, most centers initiate antifungal prophylaxis in patients with severe aplastic anema (SAA) or VSAA with an anti-mold agent such as voriconazole or posaconazole (which has the additional benefit compared to voriconazole of covering Mucor species).17,23 This is especially true for patients who have received ATG or undergone HSCT. For antimicrobial prophylaxis, a fluoroquinolone antibiotic with a spectrum of activity against Pseudomonas should be considered for patients with an ANC < 500 cells/µL.17 Acyclovir or valacyclovir prophylaxis is recommended for varicella-zoster virus and herpes simplex virus. Cytomegalovirus reactivation is minimal in patients with aplastic anemia, unless multiple courses of ATG are used.
Iron overload is another complication the provider must be aware of in the setting of increased transfusions in aplastic anemia patients. Lee and colleagues demonstrated that iron chelation therapy using deferasirox is effective at reducing serum ferritin levels in patients with aplastic anemia (median ferritin level: 3254 ng/mL prior to therapy, 1854 ng/mL following), and is associated with no serious adverse events (most common adverse events included nausea, diarrhea, vomiting, and rash).24 Approximately 25% of patients in this trial demonstrated an increase in creatinine, with patients taking concomitant cyclosporine more affected than those on chelation therapy alone.24 For patients following HSCT or with improved hematopoiesis following immunosuppressive therapy, phlebotomy can be used to treat iron overload in lieu of chelation therapy.15
Approach to Therapy
The main treatment options for SAA and VSAA include allogeneic bone marrow transplant and immunosuppression. The deciding factors as to which treatment is best initially depends on the availability of HLA-matched related donors and age (Figure 1 and Figure 2). Survival is decreased in patients with SAA or VSAA who delay initiation of therapy, and therefore prompt referral for HLA typing and evaluation for bone marrow transplant is a very important first step in managing aplastic anemia.
Matched Sibling Donor Transplant
Current standards of care recommend HLA-matched sibling donor transplant for patients with SAA or VSAA who are younger than 50 years of age, with the caveat that integration of fludarabine and reduced cyclophosphamide dosing along with ATG shows the best overall outcomes. Locasciulli and colleagues examined outcomes in patients given either immunosuppressive therapy or sibling HSCT between 1991-1996 and 1997-2002, respectively, and found that sibling HSCT was associated with a superior 10-year OS compared to immunosuppressive therapy (73% versus 68%).25 Interestingly in this study, there was no OS improvement seen with immunosuppressive therapy alone (69% versus 73%) between the 2 time periods, despite increased OS in both sibling HSCT (74% and 80%) and matched unrelated donor HSCT (38% and 65%).25 Though total body irradiation has been used in the past, it is typically not included in current conditioning regimens for matched related donor transplants.26
Current conditioning regimens typically use a combination of cyclophosphamide and ATG27,28 with or without fludarabine. Fludarabine-based conditioning regimens have shown promise in patients undergoing sibling HSCT. Maury and colleagues evaluated the role of fludarabine in addition to low-dose cyclophosphamide and ATG compared to cyclophosphamide alone or in combination with ATG in patients over age 30 undergoing sibling HSCT.9 There was a nonsignificant improvement in 5-year OS in the fludarabine arm compared to controls (77% ± 8% versus 60% ± 3%, P = 0.14) in the pooled analysis, but when adjusted for age the fludarabine arm had a significantly lower relative risk (RR) of death (RR, 0.44; P = 0.04) compared to the control arm. Shin et al reported outcomes with fludarabine/cyclophosphamide/ATG, with excellent overall outcomes and no difference in patients older or younger than 40 years.29 In addition, Kim et al evaluated their experience with patients older than 40 years of age receiving matched related donors, finding comparable outcomes in those aged 41 to 50 years compared to younger patients. Outcomes did decline in those over the age of 50 years.30 Long-term data for matched related donor transplant for aplastic anemia shows excellent long-term outcomes, with minimal chronic GVHD and good performance status.31 Hence, these factors support the role of matched related donor transplant as the initial treatment in SAA and VSAA.
Regarding the role of transplant for patients who lack a matched related donor, a growing body of literature demonstrating identical outcomes between matched related and matched unrelated donor (MUD) transplants for pediatric patients32,33 supports recent recommendations for upfront unrelated donor transplantation for aplastic anemia.34,35
Immunosuppressive Therapy
For patients without an HLA-matched sibling donor or those who are older than 50 years of age, immunosuppressive therapy is the first-line therapy. ATG and cyclosporine A are the treatments of choice.36 The potential effectiveness of immunosuppressive therapy in treating aplastic anemia was initially observed in patients in whom autologous transplant failed but who still experienced hematopoietic reconstitution despite the failed graft; this observation led to the hypothesis that the conditioning regimen may have an effect on hematopoiesis.16,36,37
Anti-thymocyte globulin. Immunosuppressive therapy with ATG has been used for the treatment of aplastic anemia since the 1980s.38 Historically, rabbit ATG had been used, but a 2011 study of horse ATG demonstrated superior hematological response at 6 months compared to rabbit ATG (68% versus 37%).16 Superior survival was also seen with horse ATG compared to rabbit ATG (3-year OS: 96% versus 76%). Due to these results, horse ATG is preferred over rabbit ATG. ATG should be used in combination with cyclosporine A to optimize outcomes.
Cyclosporine A. Early studies also demonstrated the efficacy of cyclosporine A in the treatment of aplastic anemia, with response rates equivalent to that of ATG monotherapy.39 Recent publications still note the efficacy of cyclosporine A in the treatment of aplastic anemia. Its role as an affordable option for single-agent therapy in developing countries is intriguing.39
The combination of the ATG and cyclosporine A was proven superior to either agent alone in a study by Frickhofen et al.37 In this study patients were randomly assigned to a control arm that received ATG plus methylprednisolone or to an arm that received ATG plus cyclosporine A and methylprednisolone. At 6 months, 70% of patients in the cyclosporine A arm had a complete remission (CR) or partial remission compared to 46% in the control arm.40 Further work confirmed the long-term efficacy of this regimen, reporting a 7-year OS of 55%.41 Among a pediatric population, immunosuppressive therapy was associated with an 83% 10-year OS.42
It is recommended that patients remain on cyclosporine therapy for a minimum of 6 months, after which a gradual taper may be considered, although there is variation among practitioners, with some continuing immunosuppressive therapy for a minimum of 12 months due to a proportion of patients being cyclosporine dependent.42,43 A study found that within a population of patients who responded to immunosuppressive therapy, 18% became cyclosporine dependent.42 The median duration of cyclosporine A treatment at full dose was 12 months, with tapering completed over a median of 19 months after patients had been in a stable CR for a minimum of 3 months. Relapse occurred more often when patients were tapered quickly (decrease ≥ 0.8 mg/kg/month) compared to slowly (0.4-0.7 mg/kg/month) or very slowly (< 0.3 mg/kg/month).
Immunosuppressive therapy plus eltrombopag. Townsley and colleagues recently investigated incorporating the use of the thrombopoietin receptor agonist eltrombopag with immunosuppressive therapy as first-line therapy in aplastic anemia.44 When given at a dose of 150 mg daily in patients aged 12 years and older or 75 mg daily in patients younger than 12 years, in conjunction with cyclosporine A and ATG, patients demonstrated markedly improved hematological response compared to historical treatment with standard immunosuppressive therapy alone.44 In the patient cohort administered eltrombopag starting on day 1 and continuing for 6 months, the complete response rate was 58%. Eltrombopag led to improvement in all cell lines among all treatment subgroups, and OS (censored for patients who proceeded to transplant) was 99% at 2 years.45 Overall, toxicities associated with this therapy were low, with liver enzyme elevations most commonly observed.44 Recently, a phase 2 trial of immunosuppressive therapy with or without eltrombopag was reported. Of the 38 patients enrolled, overall response, complete response, and time to response were not statistically different.46 With this recent finding, the role of eltrombopag in addition to immunosuppressive therapy is not clearly defined, and further studies are warranted.
OS for patients who do not respond to immunosuppressive therapy is approximately 57% at 5 years, largely due to improved supportive measures among this patient population.4,22 Therefore, it is important to recognize those patients who have a low chance of response so that second-line therapy can be pursued to improve outcomes.
Matched Unrelated Donor Transplant
For patients with refractory disease following immunosuppressive therapy who lack a matched sibling donor, MUD HSCT is considered standard therapy given the marked improvement in overall outcomes with modulating conditioning regimens and high-resolution HLA typing. A European Society for Blood and Marrow Transplantation analysis comparing matched sibling HSCT to MUD HSCT noted significantly higher rates of acute grade II-IV and grade III-V GVHD (grade II-IV 13% versus 25%, grade III-IV 5% versus 10%) among patients undergoing MUD transplant.47 Chronic GVHD rates were 14% in the sibling group, as compared to 26% in the MUD group. Additional benefits seen in this analysis included improved survival when transplanted under age 20 years (84% versus 72%), when transplanted within 6 months of diagnosis (85% versus 72%), the use of ATG in the conditioning regimen (81% versus 73%), and when the donor and recipient were cytomegalovirus-negative compared to other combinations (82% versus 76%).47 Interestingly, this study demonstrated that OS was not significantly increased when using a sibling HSCT compared to a MUD HSCT, likely as a result of improved understanding of conditioning regimens, GVHD prophylaxis, and supportive care.
Additional studies of MUD HSCT have shown outcomes similar to those seen in sibling HSCT.4,43 A French study found a significant increase in survival in patients undergoing MUD HSCT compared to historical cohorts (2000-2005: OS 52%; 2006-2012: OS 74%).33 The majority of patients underwent conditioning with cyclophosphamide or a combination of busulfan and cyclophosphamide, with or without fludarabine; 81% of patients included underwent in vivo T-cell depletion, and a bone marrow donor source was utilized. OS was significantly lower in patients over age 30 years undergoing MUD HSCT (57%) compared to those under age 30 years (70%). Improved OS was also seen when patients underwent transplant within 1 year of diagnosis and when a 10/10 matched donor (compared to a 9/10 mismatched donor) was utilized.4 A 2015 study investigated the role of MUD HSCT as frontline therapy instead of immunosuppressive therapy in patients without a matched sibling donor.33 The 2-year OS was 96% in the MUD HSCT cohort compared to 91%, 94%, and 74% in historical cohorts of sibling HSCT, frontline immunosuppressive therapy, and second-line MUD HSCT following failed immunosuppressive therapy, respectively. Additionally, event-free survival in the MUD HSCT cohort (defined by the authors as death, lack of response, relapse, occurrence of clonal evolution/clinical paroxysmal nocturnal hemoglobinuria, malignancies developing over follow‐up, and transplant for patients receiving immunosuppressive therapy frontline) was similar compared to sibling HSCT and superior to frontline immunosuppressive therapy and second-line MUD HSCT. Furthermore, Samarasinghe et al highlighted the importance of in vivo T-cell depletion with either ATG or alemtuzumab (anti-CD52 monoclonal antibody) in the prevention of acute and chronic GVHD in both sibling HSCT and MUD HSCT.48
With continued improvement of less toxic and more immunomodulating conditioning regimens, utilization of bone marrow as a donor cell source, in vivo T-cell depletion, and use of GVHD and antimicrobial prophylaxis, more clinical evidence supports elevating MUD HSCT in the treatment plan for patients without a matched sibling donor.49 However, there is still a large population of patients without matched sibling or unrelated donor options. In an effort to expand the transplant pool and thus avoid clonal hematopoiesis, clinically significant paroxysmal nocturnal hemoglobinuria, and relapsed aplastic anemia, more work continues to recognize the expanding role of alternative donor transplants (cord blood and haploidentical) as another viable treatment strategy for aplastic anemia after immunosuppressive therapy failure.50
Summary
Aplastic anemia is a rare but potentially life-threatening disorder characterized by pancytopenia and a marked reduction in the hematopoietic stem cell compartment. Treatment should be instituted as soon as the dignosis of aplastic anemia is established. Treatment outcomes are excellent with modern supportive care and the current approach to allogeneic transplantation, and therefore referral to a bone marrow transplant program to evaluate for early transplantation is the new standard of care.
1. Peffault De Latour R, Le Rademacher J, Antin JH, et al. Allogeneic hematopoietic stem cell transplantation in Fanconi anemia: the European Group for Blood and Marrow Transplantation experience.” Blood. 2013;122:4279-4286.
2. Auerbach AD. Diagnosis of Fanconi anemia by diepoxybutane analysis. Curr Protoc Hum Genet. 2015;85:8.7.1-17.
3. Eapen M, et al. Effect of stem cell source on outcomes after unrelated donor transplantation in severe aplastic anemia. Blood. 2011;118:2618-2621.
4. Devillier R, Dalle JH, Kulasekararaj A, et al. Unrelated alternative donor transplantation for severe acquired aplastic anemia: a study from the French Society of Bone Marrow Transplantation and Cell Therapies and the Severe Aplastic Anemia Working Party of EBMT. Haematologica. 2016;101:884-890.
5. Peffault de Latour R, Peters C, Gibson B, et al. Recommendations on hematopoietic stem cell transplantation for inherited bone marrow failure syndromes.” Bone Marrow Transplant. 2015;50:1168-1172.
6. De Medeiros CR, Zanis-Neto J, Pasquini R. Bone marrow transplantation for patients with Fanconi anemia: reduced doses of cyclophosphamide without irradiation as conditioning. Bone Marrow Transplant. 1999;24:849-852.
7. Mohanan E, Panetta JC, Lakshmi KM, et al. Population pharmacokinetics of fludarabine in patients with aplastic anemia and Fanconi anemia undergoing allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2017;52:977-983.
8 Gluckman E, Auerbach AD, Horowitz MM, et al. Bone marrow transplantation for Fanconi anemia. Blood. 1995;86:2856-2862.
9. Maury S, Bacigalupo A, Anderlini P, et al. Improved outcome of patients older than 30 years receiving HLA-identical sibling hematopoietic stem cell transplantation for severe acquired aplastic anemia using fludarabine-based conditioning: a comparison with conventional conditioning regimen. Haematologica. 2009;94:1312-1315.
10. Talbot A, Peffault de Latour R, Raffoux E, et al. Sequential treatment for allogeneic hematopoietic stem cell transplantation in Fanconi anemia with acute myeloid leukemia. Haematologica. 2014;99:e199-200.
11. Ayas M, Saber W, Davies SM, et al. Allogeneic hematopoietic cell transplantation for fanconi anemia in patients with pretransplantation cytogenetic abnormalities, myelodysplastic syndrome, or acute leukemia. J Clin Oncol. 2013;31:1669-1676.
12. Vaht K, Göransson M, Carlson K, et al. Incidence and outcome of acquired aplastic anemia: real-world data from patients diagnosed in Sweden from 2000–2011. Haematologica. 2017;102:1683-1690.
13. Passweg JR, Marsh JC. Aplastic anemia: first-line treatment by immunosuppression and sibling marrow transplantation. Hematology Am Soc Hematol Educ Program. 2010;2010:36-42.
14. Laundy GJ, Bradley BA, Rees BM, et al. Incidence and specificity of HLA antibodies in multitransfused patients with acquired aplastic anemia. Transfusion. 2004;44:814-825.
15. Killick SB, Bown N, Cavenagh J, et al. Guidelines for the diagnosis and management of adult aplastic anaemia. Br J Haematol. 2016;172:187-207.
16. Scheinberg P, Nunez O, Weinstein B, et al. Horse versus rabbit antithymocyte globulin in acquired aplastic anemia. N Eng J Med. 2011;365:430-438.
17. Höchsmann B, Moicean A, Risitano A, et al. Supportive care in severe and very severe aplastic anemia. Bone Marrow Transplant. 2013;48:168-173.
18. Valdez JM, Scheinberg P, Young NS, Walsh TJ. Infections in patients with aplastic anemia. Sem Hematol. 2009;46:269-276.
19. Torres HA, Bodey GP, Rolston KV, et al. Infections in patients with aplastic anemia: experience at a tertiary care cancer center. Cancer. 2003;98:86-93.
20. Tichelli A, Schrezenmeier H, Socié G, et al. A randomized controlled study in patients with newly diagnosed severe aplastic anemia receiving antithymocyte globulin (ATG), cyclosporine, with or without G-CSF: a study of the SAA Working Party of the European Group for Blood and Marrow Transplantation. Blood. 2011;117:4434-4441.
21. Gerson SL, Talbot GH, Hurwitz S, et al. Prolonged granulocytopenia: the major risk factor for invasive pulmonary aspergillosis in patients with acute leukemia. Ann Intern Med. 1984;100:345-351.
22. Valdez JM, Scheinberg P, Nunez O, et al. Decreased infection-related mortality and improved survival in severe aplastic anemia in the past two decades. Clin Infect Dis. 2011;52:726-735.
23. Robenshtok E, Gafter-Gvili A, Goldberg E, et al. Antifungal prophylaxis in cancer patients after chemotherapy or hematopoietic stem-cell transplantation: systematic review and meta-analysis. J Clin Oncol. 2007;25:5471-5489.
24. Lee JW, Yoon SS, Shen ZX, et al. Iron chelation therapy with deferasirox in patients with aplastic anemia: a subgroup analysis of 116 patients from the EPIC trial. Blood. 2010;116:2448-2554.
25. Locasciulli A, Oneto R, Bacigalupo A, et al. Outcome of patients with acquired aplastic anemia given first line bone marrow transplantation or immunosuppressive treatment in the last decade: a report from the European Group for Blood and Marrow Transplantation. Haematologica. 2007;92:11-8.
26. Deeg HJ, Amylon MD, Harris RE, et al. Marrow transplants from unrelated donors for patients with aplastic anemia: minimum effective dose of total body irradiation. Biol Blood Marrow Transplant. 2001;7:208-215.
27. Kahl C, Leisenring W, Joachim Deeg H, et al. Cyclophosphamide and antithymocyte globulin as a conditioning regimen for allogeneic marrow transplantation in patients with aplastic anaemia: a long‐term follow‐up. Br J Haematol. 2005;130:747-751.
28. Socié G. Allogeneic BM transplantation for the treatment of aplastic anemia: current results and expanding donor possibilities. ASH Education Program Book. 2013;2013:82-86.
29. Shin SH, Jeon YW, Yoon JH, et al. Comparable outcomes between younger (<40 years) and older (>40 years) adult patients with severe aplastic anemia after HLA-matched sibling stem cell transplantation using fludarabine-based conditioning. Bone Marrow Transplant. 2016;51:1456-1463.
30. Kim H, Lee KH, Yoon SS, et al; Korean Society of Blood and Marrow Transplantation. Allogeneic hematopoietic stem cell transplant for adults over 40 years old with acquired aplastic anemia. Biol Blood Marrow Transplant. 2012;18:1500-1508.
31. Mortensen BK, Jacobsen N, Heilmann C, Sengelov H. Allogeneic hematopoietic cell transplantation for severe aplastic anemia: similar long-term overall survival after transplantation with related donors compared to unrelated donors. Bone Marrow Transplant. 2016;51:288-290.
32. Dufour C, Svahn J, Bacigalupo A. Front-line immunosuppressive treatment of acquired aplastic anemia. Bone Marrow Transplant. 2013;48:174-177.
33. Dufour C, Veys P, Carraro E, et al. Similar outcome of upfront-unrelated and matched sibling stem cell transplantation in idiopathic paediatric aplastic anaemia. A study on the behalf of the UK Paediatric BMT Working Party, Paediatric Diseases Working Party and Severe Aplastic Anaemia Working Party of the EBMT. Br. J Haematol. 2015;151:585-594.
34. Georges GE, Doney K, Storb R. Severe aplastic anemia: allogeneic bone marrow transplantation as first-line treatment. Blood Adv. 2018;2:2020-2028.
35. Yoshida N, Kojima S. Updated guidelines for the treatment of acquired aplastic anemia in children. Curr Oncol Rep. 2018;20:67.
36. Mathe G, Amiel JL, Schwarzenberg L, et al. Bone marrow graft in man after conditioning by antilymphocytic serum. Br Med J. 1970;2:131-136.
37. Frickhofen N, Kaltwasser JP, Schrezenmeier H, et al, German Aplastic Anemia Study Group. Treatment of aplastic anemia with antilymphocyte globulin and methylprednisolone with or without cyclosporine. N Engl J Med. 1991;324:1297-1304.
38. Speck B, Gratwohl A, Nissen C, et al. Treatment of severe aplastic anaemia with antilymphocyte globulin or bone-marrow transplantation. Br Med J. 1981;282:860-863.
39. Al-Ghazaly J, Al-Dubai W, Al-Jahafi AK, et al. Cyclosporine monotherapy for severe aplastic anemia: a developing country experience. Ann Saudi Med. 2005;25:375-379.
40. Scheinberg P, Young NS. How I treat acquired aplastic anemia. Blood. 2012;120:1185-1196.
41. Rosenfeld S, Follmann D, Nunez O, Young NS. Antithymocyte globulin and cyclosporine for severe aplastic anemia: association between hematologic response and long-term outcome. JAMA. 2003;289:1130-1135.
42. Saracco P, Quarello P, Iori AP, et al. Cyclosporin A response and dependence in children with acquired aplastic anaemia: a multicentre retrospective study with long‐term observation follow‐up. Br J Haematol. 2008;140:197-205.
43. Bacigalupo A. How I treat acquired aplastic anemia. Blood. 2017;129:1428-1436.
44. Townsley DM, Scheinberg P, Winkler T, et al. Eltrombopag added to standard immunosuppression for aplastic anemia. N Engl J Med. 2017;376:1540-1550.
45. Olnes MJ, Scheinberg P, Calvo KR, et al. Eltrombopag and improved hematopoiesis in refractory aplastic anemia. N Engl J Med. 2012;367:11-19.
46. Assi R, Garcia-Manero G, Ravandi F, et al. Addition of eltrombopag to immunosuppressive therapy in patients with newly diagnosed aplastic anemia. Cancer. 2018 Oct 11. doi: 10.1002/cncr.31658.
47. Bacigalupo A, Socié G, Hamladji RM, et al. Current outcome of HLA identical sibling vs. unrelated donor transplants in severe aplastic anemia: an EBMT analysis. Haematologica. 2015;100:696-702.
48. Samarasinghe S, Iacobelli S, Knol C, et al. Impact of different in vivo T cell depletion strategies on outcomes following hematopoietic stem cell transplantation for idiopathic aplastic anaemia: a study on behalf of the EBMT SAA Working Party. 2018Oct 17. doi: 10.1002/ajh.25314.
49. Clesham K, Dowse R, Samarasinghe S. Upfront matched unrelated donor transplantation in aplastic anemia. Hematol Oncol Clin North Am. 2018;32:619-628.
50. DeZern AE, Brodsky RA. Haploidentical donor bone marrow transplantation for severe aplastic anemia. Hematol Oncol Clin North Am. 2018;32:629-642.
Aplastic anemia is a rare hematologic disorder marked by pancytopenia and a hypocellular marrow. Aplastic anemia results from either inherited or acquired causes, and the treatment approach varies significantly between the 2 causes. This article reviews the treatment of inherited and acquired forms of aplastic anemia. The approach to evaluation and diagnosis of aplastic anemia is reviewed in a separate article.
Inherited Aplastic Anemia
First-line treatment options for patients with inherited marrow failure syndromes (IMFS) are androgen therapy and hematopoietic stem cell transplant (HSCT). When evaluating patients for HSCT, it is critical to identify the presence of an IMFS, as the risk and mortality associated with the conditioning regimen, stem cell source, graft-versus-host disease (GVHD), and secondary malignancies differ between patients with IMFS and those with acquired marrow failure syndromes or hematologic malignancies.
Potential sibling donors need to be screened for donor candidacy as well as for the inherited defect.1 Among patients with Fanconi anemia or a telomere biology disorder, the stem cell source must be considered, with bone marrow demonstrating lower rates of acute GVHD than a peripheral blood stem cell source.2-4 In IMFS patients, the donor cell type may affect the choice of conditioning regimen.5,6 Reduced-intensity conditioning in lieu of myeloablative conditioning without total body irradiation has proved feasible in patients with Fanconi anemia, and is associated with a reduced risk of secondary malignancies.5,6 Incorporation of fludarabine in the conditioning regimen of patients without a matched sibling donor is associated with superior engraftment and survival2,5,7 compared to cyclophosphamide conditioning, which was historically used in matched related donors.6,8 The addition of fludarabine appears to be especially beneficial in older patients, in whom its use is associated with lower rates of graft failure, likely due to increased immunosuppression at the time of engraftment.7,9 Fludarabine has also been incorporated into conditioning regimens for patients with a telomere biology disorder, but outcomes data is limited.5
For patients presenting with acute myeloid leukemia (AML) or a high-risk myelodysplastic syndrome (MDS) who are subsequently diagnosed with an IMFS, treatment can be more complex, as these patients are at high risk for toxicity from standard chemotherapy. Limited data suggests that induction therapy and transplantation are feasible in this group of patients, and this approach is associated with increased overall survival (OS) despite lower OS rates than those of IMFS patients who present prior to the development of MDS or AML.10,11 Further work is needed to determine the optimal induction regimen that balances the risks of treatment-related mortality and complications associated with conditioning regimens, risk of relapse, and risk of secondary malignancies, especially in the cohort of patients diagnosed at an older age.
Acquired Aplastic Anemia
Supportive Care
While the workup and treatment plan is being established, attention should be directed at supportive care for prevention of complications. The most common complications leading to death in patients with significant pancytopenia and neutropenia are opportunistic infections and hemorrhagic complications.12
Transfusion support is critical to avoid symptomatic anemia and hemorrhagic complications related to thrombocytopenia, which typically occur with platelet counts lower than 10,000 cells/µL. However, transfusion carries the risk of alloimmunization (which may persist for years following transfusion) and transfusion-related graft versus host disease (trGVHD), and thus use of transfusion should be minimized when possible.13,14 Transfusion support is often required to prevent complications associated with thrombocytopenia and anemia; all blood products given to patients with aplastic anemia should be irradiated and leukoreduced to reduce the risk of both alloimmunization and trGVHD. Guidelines from the British Society for Haematology recommend routine screening for Rh and Kell antibodies to reduce the risk of alloimmunization.15 Infectious complications remain a common cause of morbidity and mortality in patients with aplastic anemia who have prolonged neutropenia (defined as an absolute neutrophil count [ANC] < 500 cells/µL).16-19 Therefore, patients should receive broad-spectrum antibiotics with antipseudomonal coverage. In a study by Tichelli and colleagues evaluating the role of granulocyte-colony stimulating factor (G-CSF) in patients with SAA receiving immunosuppressive therapy, 55% of all patient deaths were secondary to infection.20 There was no OS benefit seen in patients who received G-CSF, though a significantly lower rate of infection was observed in the G-CSF arm compared to those not receiving G-CSF (56% versus 81%, P = 0.006).This difference was largely driven by a decrease in infectious episodes in patients with very severe aplastic anemia (VSAA) treated with G-CSF as compared to those who did not receive this therapy (22% versus 48%, P = 0.014).20
Angio-invasive pulmonary aspergillosis and Zygomycetes (eg, Rhizopus, Mucor species) remain major causes of mortality related to opportunistic mycotic infections in patients with aplastic anemia.18 The infectious risk is directly related to the duration and severity of neutropenia, with one study demonstrating a significant increase in risk in AML patients with neutropenia lasting longer than 3 weeks.21 Invasive fungal infections carry a high mortality in patients with severe neutropenia, though due to earlier recognition and empiric antifungal therapy with extended-spectrum azoles, overall mortality secondary to invasive fungal infections is declining.19,22
While neutropenia related to cytotoxic chemotherapy is commonly associated with gram-negative bacteria due to disruption of mucosal barriers, patients with aplastic anemia have an increased incidence of gram-positive bacteremia with staphylococcal species compared to other neutropenic populations.18,19 This appears to be changing with time. Valdez and colleagues demonstrated a decrease in prevalence of coagulase-negative staphylococcal infections, increased prevalence of gram-positive bacilli bacteremia, and no change in prevalence of gram-negative bacteremia in patients with aplastic anemia treated between 1989 and 2008.22 Gram-negative bacteremia caused by Stenotrophomonas maltophila, Escherichia coli, Klebsiella pneumoniae, Citrobacter, and Proteus has also been reported.19 Despite a lack of clinical trials investigating the role of antifungal and antibacterial prophylaxis for patients with aplastic anemia, most centers initiate antifungal prophylaxis in patients with severe aplastic anema (SAA) or VSAA with an anti-mold agent such as voriconazole or posaconazole (which has the additional benefit compared to voriconazole of covering Mucor species).17,23 This is especially true for patients who have received ATG or undergone HSCT. For antimicrobial prophylaxis, a fluoroquinolone antibiotic with a spectrum of activity against Pseudomonas should be considered for patients with an ANC < 500 cells/µL.17 Acyclovir or valacyclovir prophylaxis is recommended for varicella-zoster virus and herpes simplex virus. Cytomegalovirus reactivation is minimal in patients with aplastic anemia, unless multiple courses of ATG are used.
Iron overload is another complication the provider must be aware of in the setting of increased transfusions in aplastic anemia patients. Lee and colleagues demonstrated that iron chelation therapy using deferasirox is effective at reducing serum ferritin levels in patients with aplastic anemia (median ferritin level: 3254 ng/mL prior to therapy, 1854 ng/mL following), and is associated with no serious adverse events (most common adverse events included nausea, diarrhea, vomiting, and rash).24 Approximately 25% of patients in this trial demonstrated an increase in creatinine, with patients taking concomitant cyclosporine more affected than those on chelation therapy alone.24 For patients following HSCT or with improved hematopoiesis following immunosuppressive therapy, phlebotomy can be used to treat iron overload in lieu of chelation therapy.15
Approach to Therapy
The main treatment options for SAA and VSAA include allogeneic bone marrow transplant and immunosuppression. The deciding factors as to which treatment is best initially depends on the availability of HLA-matched related donors and age (Figure 1 and Figure 2). Survival is decreased in patients with SAA or VSAA who delay initiation of therapy, and therefore prompt referral for HLA typing and evaluation for bone marrow transplant is a very important first step in managing aplastic anemia.


Matched Sibling Donor Transplant
Current standards of care recommend HLA-matched sibling donor transplant for patients with SAA or VSAA who are younger than 50 years of age, with the caveat that integration of fludarabine and reduced cyclophosphamide dosing along with ATG shows the best overall outcomes. Locasciulli and colleagues examined outcomes in patients given either immunosuppressive therapy or sibling HSCT between 1991-1996 and 1997-2002, respectively, and found that sibling HSCT was associated with a superior 10-year OS compared to immunosuppressive therapy (73% versus 68%).25 Interestingly in this study, there was no OS improvement seen with immunosuppressive therapy alone (69% versus 73%) between the 2 time periods, despite increased OS in both sibling HSCT (74% and 80%) and matched unrelated donor HSCT (38% and 65%).25 Though total body irradiation has been used in the past, it is typically not included in current conditioning regimens for matched related donor transplants.26
Current conditioning regimens typically use a combination of cyclophosphamide and ATG27,28 with or without fludarabine. Fludarabine-based conditioning regimens have shown promise in patients undergoing sibling HSCT. Maury and colleagues evaluated the role of fludarabine in addition to low-dose cyclophosphamide and ATG compared to cyclophosphamide alone or in combination with ATG in patients over age 30 undergoing sibling HSCT.9 There was a nonsignificant improvement in 5-year OS in the fludarabine arm compared to controls (77% ± 8% versus 60% ± 3%, P = 0.14) in the pooled analysis, but when adjusted for age the fludarabine arm had a significantly lower relative risk (RR) of death (RR, 0.44; P = 0.04) compared to the control arm. Shin et al reported outcomes with fludarabine/cyclophosphamide/ATG, with excellent overall outcomes and no difference in patients older or younger than 40 years.29 In addition, Kim et al evaluated their experience with patients older than 40 years of age receiving matched related donors, finding comparable outcomes in those aged 41 to 50 years compared to younger patients. Outcomes did decline in those over the age of 50 years.30 Long-term data for matched related donor transplant for aplastic anemia shows excellent long-term outcomes, with minimal chronic GVHD and good performance status.31 Hence, these factors support the role of matched related donor transplant as the initial treatment in SAA and VSAA.
Regarding the role of transplant for patients who lack a matched related donor, a growing body of literature demonstrating identical outcomes between matched related and matched unrelated donor (MUD) transplants for pediatric patients32,33 supports recent recommendations for upfront unrelated donor transplantation for aplastic anemia.34,35
Immunosuppressive Therapy
For patients without an HLA-matched sibling donor or those who are older than 50 years of age, immunosuppressive therapy is the first-line therapy. ATG and cyclosporine A are the treatments of choice.36 The potential effectiveness of immunosuppressive therapy in treating aplastic anemia was initially observed in patients in whom autologous transplant failed but who still experienced hematopoietic reconstitution despite the failed graft; this observation led to the hypothesis that the conditioning regimen may have an effect on hematopoiesis.16,36,37
Anti-thymocyte globulin. Immunosuppressive therapy with ATG has been used for the treatment of aplastic anemia since the 1980s.38 Historically, rabbit ATG had been used, but a 2011 study of horse ATG demonstrated superior hematological response at 6 months compared to rabbit ATG (68% versus 37%).16 Superior survival was also seen with horse ATG compared to rabbit ATG (3-year OS: 96% versus 76%). Due to these results, horse ATG is preferred over rabbit ATG. ATG should be used in combination with cyclosporine A to optimize outcomes.
Cyclosporine A. Early studies also demonstrated the efficacy of cyclosporine A in the treatment of aplastic anemia, with response rates equivalent to that of ATG monotherapy.39 Recent publications still note the efficacy of cyclosporine A in the treatment of aplastic anemia. Its role as an affordable option for single-agent therapy in developing countries is intriguing.39
The combination of the ATG and cyclosporine A was proven superior to either agent alone in a study by Frickhofen et al.37 In this study patients were randomly assigned to a control arm that received ATG plus methylprednisolone or to an arm that received ATG plus cyclosporine A and methylprednisolone. At 6 months, 70% of patients in the cyclosporine A arm had a complete remission (CR) or partial remission compared to 46% in the control arm.40 Further work confirmed the long-term efficacy of this regimen, reporting a 7-year OS of 55%.41 Among a pediatric population, immunosuppressive therapy was associated with an 83% 10-year OS.42
It is recommended that patients remain on cyclosporine therapy for a minimum of 6 months, after which a gradual taper may be considered, although there is variation among practitioners, with some continuing immunosuppressive therapy for a minimum of 12 months due to a proportion of patients being cyclosporine dependent.42,43 A study found that within a population of patients who responded to immunosuppressive therapy, 18% became cyclosporine dependent.42 The median duration of cyclosporine A treatment at full dose was 12 months, with tapering completed over a median of 19 months after patients had been in a stable CR for a minimum of 3 months. Relapse occurred more often when patients were tapered quickly (decrease ≥ 0.8 mg/kg/month) compared to slowly (0.4-0.7 mg/kg/month) or very slowly (< 0.3 mg/kg/month).
Immunosuppressive therapy plus eltrombopag. Townsley and colleagues recently investigated incorporating the use of the thrombopoietin receptor agonist eltrombopag with immunosuppressive therapy as first-line therapy in aplastic anemia.44 When given at a dose of 150 mg daily in patients aged 12 years and older or 75 mg daily in patients younger than 12 years, in conjunction with cyclosporine A and ATG, patients demonstrated markedly improved hematological response compared to historical treatment with standard immunosuppressive therapy alone.44 In the patient cohort administered eltrombopag starting on day 1 and continuing for 6 months, the complete response rate was 58%. Eltrombopag led to improvement in all cell lines among all treatment subgroups, and OS (censored for patients who proceeded to transplant) was 99% at 2 years.45 Overall, toxicities associated with this therapy were low, with liver enzyme elevations most commonly observed.44 Recently, a phase 2 trial of immunosuppressive therapy with or without eltrombopag was reported. Of the 38 patients enrolled, overall response, complete response, and time to response were not statistically different.46 With this recent finding, the role of eltrombopag in addition to immunosuppressive therapy is not clearly defined, and further studies are warranted.
OS for patients who do not respond to immunosuppressive therapy is approximately 57% at 5 years, largely due to improved supportive measures among this patient population.4,22 Therefore, it is important to recognize those patients who have a low chance of response so that second-line therapy can be pursued to improve outcomes.
Matched Unrelated Donor Transplant
For patients with refractory disease following immunosuppressive therapy who lack a matched sibling donor, MUD HSCT is considered standard therapy given the marked improvement in overall outcomes with modulating conditioning regimens and high-resolution HLA typing. A European Society for Blood and Marrow Transplantation analysis comparing matched sibling HSCT to MUD HSCT noted significantly higher rates of acute grade II-IV and grade III-V GVHD (grade II-IV 13% versus 25%, grade III-IV 5% versus 10%) among patients undergoing MUD transplant.47 Chronic GVHD rates were 14% in the sibling group, as compared to 26% in the MUD group. Additional benefits seen in this analysis included improved survival when transplanted under age 20 years (84% versus 72%), when transplanted within 6 months of diagnosis (85% versus 72%), the use of ATG in the conditioning regimen (81% versus 73%), and when the donor and recipient were cytomegalovirus-negative compared to other combinations (82% versus 76%).47 Interestingly, this study demonstrated that OS was not significantly increased when using a sibling HSCT compared to a MUD HSCT, likely as a result of improved understanding of conditioning regimens, GVHD prophylaxis, and supportive care.
Additional studies of MUD HSCT have shown outcomes similar to those seen in sibling HSCT.4,43 A French study found a significant increase in survival in patients undergoing MUD HSCT compared to historical cohorts (2000-2005: OS 52%; 2006-2012: OS 74%).33 The majority of patients underwent conditioning with cyclophosphamide or a combination of busulfan and cyclophosphamide, with or without fludarabine; 81% of patients included underwent in vivo T-cell depletion, and a bone marrow donor source was utilized. OS was significantly lower in patients over age 30 years undergoing MUD HSCT (57%) compared to those under age 30 years (70%). Improved OS was also seen when patients underwent transplant within 1 year of diagnosis and when a 10/10 matched donor (compared to a 9/10 mismatched donor) was utilized.4 A 2015 study investigated the role of MUD HSCT as frontline therapy instead of immunosuppressive therapy in patients without a matched sibling donor.33 The 2-year OS was 96% in the MUD HSCT cohort compared to 91%, 94%, and 74% in historical cohorts of sibling HSCT, frontline immunosuppressive therapy, and second-line MUD HSCT following failed immunosuppressive therapy, respectively. Additionally, event-free survival in the MUD HSCT cohort (defined by the authors as death, lack of response, relapse, occurrence of clonal evolution/clinical paroxysmal nocturnal hemoglobinuria, malignancies developing over follow‐up, and transplant for patients receiving immunosuppressive therapy frontline) was similar compared to sibling HSCT and superior to frontline immunosuppressive therapy and second-line MUD HSCT. Furthermore, Samarasinghe et al highlighted the importance of in vivo T-cell depletion with either ATG or alemtuzumab (anti-CD52 monoclonal antibody) in the prevention of acute and chronic GVHD in both sibling HSCT and MUD HSCT.48
With continued improvement of less toxic and more immunomodulating conditioning regimens, utilization of bone marrow as a donor cell source, in vivo T-cell depletion, and use of GVHD and antimicrobial prophylaxis, more clinical evidence supports elevating MUD HSCT in the treatment plan for patients without a matched sibling donor.49 However, there is still a large population of patients without matched sibling or unrelated donor options. In an effort to expand the transplant pool and thus avoid clonal hematopoiesis, clinically significant paroxysmal nocturnal hemoglobinuria, and relapsed aplastic anemia, more work continues to recognize the expanding role of alternative donor transplants (cord blood and haploidentical) as another viable treatment strategy for aplastic anemia after immunosuppressive therapy failure.50
Summary
Aplastic anemia is a rare but potentially life-threatening disorder characterized by pancytopenia and a marked reduction in the hematopoietic stem cell compartment. Treatment should be instituted as soon as the dignosis of aplastic anemia is established. Treatment outcomes are excellent with modern supportive care and the current approach to allogeneic transplantation, and therefore referral to a bone marrow transplant program to evaluate for early transplantation is the new standard of care.
Aplastic anemia is a rare hematologic disorder marked by pancytopenia and a hypocellular marrow. Aplastic anemia results from either inherited or acquired causes, and the treatment approach varies significantly between the 2 causes. This article reviews the treatment of inherited and acquired forms of aplastic anemia. The approach to evaluation and diagnosis of aplastic anemia is reviewed in a separate article.
Inherited Aplastic Anemia
First-line treatment options for patients with inherited marrow failure syndromes (IMFS) are androgen therapy and hematopoietic stem cell transplant (HSCT). When evaluating patients for HSCT, it is critical to identify the presence of an IMFS, as the risk and mortality associated with the conditioning regimen, stem cell source, graft-versus-host disease (GVHD), and secondary malignancies differ between patients with IMFS and those with acquired marrow failure syndromes or hematologic malignancies.
Potential sibling donors need to be screened for donor candidacy as well as for the inherited defect.1 Among patients with Fanconi anemia or a telomere biology disorder, the stem cell source must be considered, with bone marrow demonstrating lower rates of acute GVHD than a peripheral blood stem cell source.2-4 In IMFS patients, the donor cell type may affect the choice of conditioning regimen.5,6 Reduced-intensity conditioning in lieu of myeloablative conditioning without total body irradiation has proved feasible in patients with Fanconi anemia, and is associated with a reduced risk of secondary malignancies.5,6 Incorporation of fludarabine in the conditioning regimen of patients without a matched sibling donor is associated with superior engraftment and survival2,5,7 compared to cyclophosphamide conditioning, which was historically used in matched related donors.6,8 The addition of fludarabine appears to be especially beneficial in older patients, in whom its use is associated with lower rates of graft failure, likely due to increased immunosuppression at the time of engraftment.7,9 Fludarabine has also been incorporated into conditioning regimens for patients with a telomere biology disorder, but outcomes data is limited.5
For patients presenting with acute myeloid leukemia (AML) or a high-risk myelodysplastic syndrome (MDS) who are subsequently diagnosed with an IMFS, treatment can be more complex, as these patients are at high risk for toxicity from standard chemotherapy. Limited data suggests that induction therapy and transplantation are feasible in this group of patients, and this approach is associated with increased overall survival (OS) despite lower OS rates than those of IMFS patients who present prior to the development of MDS or AML.10,11 Further work is needed to determine the optimal induction regimen that balances the risks of treatment-related mortality and complications associated with conditioning regimens, risk of relapse, and risk of secondary malignancies, especially in the cohort of patients diagnosed at an older age.
Acquired Aplastic Anemia
Supportive Care
While the workup and treatment plan is being established, attention should be directed at supportive care for prevention of complications. The most common complications leading to death in patients with significant pancytopenia and neutropenia are opportunistic infections and hemorrhagic complications.12
Transfusion support is critical to avoid symptomatic anemia and hemorrhagic complications related to thrombocytopenia, which typically occur with platelet counts lower than 10,000 cells/µL. However, transfusion carries the risk of alloimmunization (which may persist for years following transfusion) and transfusion-related graft versus host disease (trGVHD), and thus use of transfusion should be minimized when possible.13,14 Transfusion support is often required to prevent complications associated with thrombocytopenia and anemia; all blood products given to patients with aplastic anemia should be irradiated and leukoreduced to reduce the risk of both alloimmunization and trGVHD. Guidelines from the British Society for Haematology recommend routine screening for Rh and Kell antibodies to reduce the risk of alloimmunization.15 Infectious complications remain a common cause of morbidity and mortality in patients with aplastic anemia who have prolonged neutropenia (defined as an absolute neutrophil count [ANC] < 500 cells/µL).16-19 Therefore, patients should receive broad-spectrum antibiotics with antipseudomonal coverage. In a study by Tichelli and colleagues evaluating the role of granulocyte-colony stimulating factor (G-CSF) in patients with SAA receiving immunosuppressive therapy, 55% of all patient deaths were secondary to infection.20 There was no OS benefit seen in patients who received G-CSF, though a significantly lower rate of infection was observed in the G-CSF arm compared to those not receiving G-CSF (56% versus 81%, P = 0.006).This difference was largely driven by a decrease in infectious episodes in patients with very severe aplastic anemia (VSAA) treated with G-CSF as compared to those who did not receive this therapy (22% versus 48%, P = 0.014).20
Angio-invasive pulmonary aspergillosis and Zygomycetes (eg, Rhizopus, Mucor species) remain major causes of mortality related to opportunistic mycotic infections in patients with aplastic anemia.18 The infectious risk is directly related to the duration and severity of neutropenia, with one study demonstrating a significant increase in risk in AML patients with neutropenia lasting longer than 3 weeks.21 Invasive fungal infections carry a high mortality in patients with severe neutropenia, though due to earlier recognition and empiric antifungal therapy with extended-spectrum azoles, overall mortality secondary to invasive fungal infections is declining.19,22
While neutropenia related to cytotoxic chemotherapy is commonly associated with gram-negative bacteria due to disruption of mucosal barriers, patients with aplastic anemia have an increased incidence of gram-positive bacteremia with staphylococcal species compared to other neutropenic populations.18,19 This appears to be changing with time. Valdez and colleagues demonstrated a decrease in prevalence of coagulase-negative staphylococcal infections, increased prevalence of gram-positive bacilli bacteremia, and no change in prevalence of gram-negative bacteremia in patients with aplastic anemia treated between 1989 and 2008.22 Gram-negative bacteremia caused by Stenotrophomonas maltophila, Escherichia coli, Klebsiella pneumoniae, Citrobacter, and Proteus has also been reported.19 Despite a lack of clinical trials investigating the role of antifungal and antibacterial prophylaxis for patients with aplastic anemia, most centers initiate antifungal prophylaxis in patients with severe aplastic anema (SAA) or VSAA with an anti-mold agent such as voriconazole or posaconazole (which has the additional benefit compared to voriconazole of covering Mucor species).17,23 This is especially true for patients who have received ATG or undergone HSCT. For antimicrobial prophylaxis, a fluoroquinolone antibiotic with a spectrum of activity against Pseudomonas should be considered for patients with an ANC < 500 cells/µL.17 Acyclovir or valacyclovir prophylaxis is recommended for varicella-zoster virus and herpes simplex virus. Cytomegalovirus reactivation is minimal in patients with aplastic anemia, unless multiple courses of ATG are used.
Iron overload is another complication the provider must be aware of in the setting of increased transfusions in aplastic anemia patients. Lee and colleagues demonstrated that iron chelation therapy using deferasirox is effective at reducing serum ferritin levels in patients with aplastic anemia (median ferritin level: 3254 ng/mL prior to therapy, 1854 ng/mL following), and is associated with no serious adverse events (most common adverse events included nausea, diarrhea, vomiting, and rash).24 Approximately 25% of patients in this trial demonstrated an increase in creatinine, with patients taking concomitant cyclosporine more affected than those on chelation therapy alone.24 For patients following HSCT or with improved hematopoiesis following immunosuppressive therapy, phlebotomy can be used to treat iron overload in lieu of chelation therapy.15
Approach to Therapy
The main treatment options for SAA and VSAA include allogeneic bone marrow transplant and immunosuppression. The deciding factors as to which treatment is best initially depends on the availability of HLA-matched related donors and age (Figure 1 and Figure 2). Survival is decreased in patients with SAA or VSAA who delay initiation of therapy, and therefore prompt referral for HLA typing and evaluation for bone marrow transplant is a very important first step in managing aplastic anemia.


Matched Sibling Donor Transplant
Current standards of care recommend HLA-matched sibling donor transplant for patients with SAA or VSAA who are younger than 50 years of age, with the caveat that integration of fludarabine and reduced cyclophosphamide dosing along with ATG shows the best overall outcomes. Locasciulli and colleagues examined outcomes in patients given either immunosuppressive therapy or sibling HSCT between 1991-1996 and 1997-2002, respectively, and found that sibling HSCT was associated with a superior 10-year OS compared to immunosuppressive therapy (73% versus 68%).25 Interestingly in this study, there was no OS improvement seen with immunosuppressive therapy alone (69% versus 73%) between the 2 time periods, despite increased OS in both sibling HSCT (74% and 80%) and matched unrelated donor HSCT (38% and 65%).25 Though total body irradiation has been used in the past, it is typically not included in current conditioning regimens for matched related donor transplants.26
Current conditioning regimens typically use a combination of cyclophosphamide and ATG27,28 with or without fludarabine. Fludarabine-based conditioning regimens have shown promise in patients undergoing sibling HSCT. Maury and colleagues evaluated the role of fludarabine in addition to low-dose cyclophosphamide and ATG compared to cyclophosphamide alone or in combination with ATG in patients over age 30 undergoing sibling HSCT.9 There was a nonsignificant improvement in 5-year OS in the fludarabine arm compared to controls (77% ± 8% versus 60% ± 3%, P = 0.14) in the pooled analysis, but when adjusted for age the fludarabine arm had a significantly lower relative risk (RR) of death (RR, 0.44; P = 0.04) compared to the control arm. Shin et al reported outcomes with fludarabine/cyclophosphamide/ATG, with excellent overall outcomes and no difference in patients older or younger than 40 years.29 In addition, Kim et al evaluated their experience with patients older than 40 years of age receiving matched related donors, finding comparable outcomes in those aged 41 to 50 years compared to younger patients. Outcomes did decline in those over the age of 50 years.30 Long-term data for matched related donor transplant for aplastic anemia shows excellent long-term outcomes, with minimal chronic GVHD and good performance status.31 Hence, these factors support the role of matched related donor transplant as the initial treatment in SAA and VSAA.
Regarding the role of transplant for patients who lack a matched related donor, a growing body of literature demonstrating identical outcomes between matched related and matched unrelated donor (MUD) transplants for pediatric patients32,33 supports recent recommendations for upfront unrelated donor transplantation for aplastic anemia.34,35
Immunosuppressive Therapy
For patients without an HLA-matched sibling donor or those who are older than 50 years of age, immunosuppressive therapy is the first-line therapy. ATG and cyclosporine A are the treatments of choice.36 The potential effectiveness of immunosuppressive therapy in treating aplastic anemia was initially observed in patients in whom autologous transplant failed but who still experienced hematopoietic reconstitution despite the failed graft; this observation led to the hypothesis that the conditioning regimen may have an effect on hematopoiesis.16,36,37
Anti-thymocyte globulin. Immunosuppressive therapy with ATG has been used for the treatment of aplastic anemia since the 1980s.38 Historically, rabbit ATG had been used, but a 2011 study of horse ATG demonstrated superior hematological response at 6 months compared to rabbit ATG (68% versus 37%).16 Superior survival was also seen with horse ATG compared to rabbit ATG (3-year OS: 96% versus 76%). Due to these results, horse ATG is preferred over rabbit ATG. ATG should be used in combination with cyclosporine A to optimize outcomes.
Cyclosporine A. Early studies also demonstrated the efficacy of cyclosporine A in the treatment of aplastic anemia, with response rates equivalent to that of ATG monotherapy.39 Recent publications still note the efficacy of cyclosporine A in the treatment of aplastic anemia. Its role as an affordable option for single-agent therapy in developing countries is intriguing.39
The combination of the ATG and cyclosporine A was proven superior to either agent alone in a study by Frickhofen et al.37 In this study patients were randomly assigned to a control arm that received ATG plus methylprednisolone or to an arm that received ATG plus cyclosporine A and methylprednisolone. At 6 months, 70% of patients in the cyclosporine A arm had a complete remission (CR) or partial remission compared to 46% in the control arm.40 Further work confirmed the long-term efficacy of this regimen, reporting a 7-year OS of 55%.41 Among a pediatric population, immunosuppressive therapy was associated with an 83% 10-year OS.42
It is recommended that patients remain on cyclosporine therapy for a minimum of 6 months, after which a gradual taper may be considered, although there is variation among practitioners, with some continuing immunosuppressive therapy for a minimum of 12 months due to a proportion of patients being cyclosporine dependent.42,43 A study found that within a population of patients who responded to immunosuppressive therapy, 18% became cyclosporine dependent.42 The median duration of cyclosporine A treatment at full dose was 12 months, with tapering completed over a median of 19 months after patients had been in a stable CR for a minimum of 3 months. Relapse occurred more often when patients were tapered quickly (decrease ≥ 0.8 mg/kg/month) compared to slowly (0.4-0.7 mg/kg/month) or very slowly (< 0.3 mg/kg/month).
Immunosuppressive therapy plus eltrombopag. Townsley and colleagues recently investigated incorporating the use of the thrombopoietin receptor agonist eltrombopag with immunosuppressive therapy as first-line therapy in aplastic anemia.44 When given at a dose of 150 mg daily in patients aged 12 years and older or 75 mg daily in patients younger than 12 years, in conjunction with cyclosporine A and ATG, patients demonstrated markedly improved hematological response compared to historical treatment with standard immunosuppressive therapy alone.44 In the patient cohort administered eltrombopag starting on day 1 and continuing for 6 months, the complete response rate was 58%. Eltrombopag led to improvement in all cell lines among all treatment subgroups, and OS (censored for patients who proceeded to transplant) was 99% at 2 years.45 Overall, toxicities associated with this therapy were low, with liver enzyme elevations most commonly observed.44 Recently, a phase 2 trial of immunosuppressive therapy with or without eltrombopag was reported. Of the 38 patients enrolled, overall response, complete response, and time to response were not statistically different.46 With this recent finding, the role of eltrombopag in addition to immunosuppressive therapy is not clearly defined, and further studies are warranted.
OS for patients who do not respond to immunosuppressive therapy is approximately 57% at 5 years, largely due to improved supportive measures among this patient population.4,22 Therefore, it is important to recognize those patients who have a low chance of response so that second-line therapy can be pursued to improve outcomes.
Matched Unrelated Donor Transplant
For patients with refractory disease following immunosuppressive therapy who lack a matched sibling donor, MUD HSCT is considered standard therapy given the marked improvement in overall outcomes with modulating conditioning regimens and high-resolution HLA typing. A European Society for Blood and Marrow Transplantation analysis comparing matched sibling HSCT to MUD HSCT noted significantly higher rates of acute grade II-IV and grade III-V GVHD (grade II-IV 13% versus 25%, grade III-IV 5% versus 10%) among patients undergoing MUD transplant.47 Chronic GVHD rates were 14% in the sibling group, as compared to 26% in the MUD group. Additional benefits seen in this analysis included improved survival when transplanted under age 20 years (84% versus 72%), when transplanted within 6 months of diagnosis (85% versus 72%), the use of ATG in the conditioning regimen (81% versus 73%), and when the donor and recipient were cytomegalovirus-negative compared to other combinations (82% versus 76%).47 Interestingly, this study demonstrated that OS was not significantly increased when using a sibling HSCT compared to a MUD HSCT, likely as a result of improved understanding of conditioning regimens, GVHD prophylaxis, and supportive care.
Additional studies of MUD HSCT have shown outcomes similar to those seen in sibling HSCT.4,43 A French study found a significant increase in survival in patients undergoing MUD HSCT compared to historical cohorts (2000-2005: OS 52%; 2006-2012: OS 74%).33 The majority of patients underwent conditioning with cyclophosphamide or a combination of busulfan and cyclophosphamide, with or without fludarabine; 81% of patients included underwent in vivo T-cell depletion, and a bone marrow donor source was utilized. OS was significantly lower in patients over age 30 years undergoing MUD HSCT (57%) compared to those under age 30 years (70%). Improved OS was also seen when patients underwent transplant within 1 year of diagnosis and when a 10/10 matched donor (compared to a 9/10 mismatched donor) was utilized.4 A 2015 study investigated the role of MUD HSCT as frontline therapy instead of immunosuppressive therapy in patients without a matched sibling donor.33 The 2-year OS was 96% in the MUD HSCT cohort compared to 91%, 94%, and 74% in historical cohorts of sibling HSCT, frontline immunosuppressive therapy, and second-line MUD HSCT following failed immunosuppressive therapy, respectively. Additionally, event-free survival in the MUD HSCT cohort (defined by the authors as death, lack of response, relapse, occurrence of clonal evolution/clinical paroxysmal nocturnal hemoglobinuria, malignancies developing over follow‐up, and transplant for patients receiving immunosuppressive therapy frontline) was similar compared to sibling HSCT and superior to frontline immunosuppressive therapy and second-line MUD HSCT. Furthermore, Samarasinghe et al highlighted the importance of in vivo T-cell depletion with either ATG or alemtuzumab (anti-CD52 monoclonal antibody) in the prevention of acute and chronic GVHD in both sibling HSCT and MUD HSCT.48
With continued improvement of less toxic and more immunomodulating conditioning regimens, utilization of bone marrow as a donor cell source, in vivo T-cell depletion, and use of GVHD and antimicrobial prophylaxis, more clinical evidence supports elevating MUD HSCT in the treatment plan for patients without a matched sibling donor.49 However, there is still a large population of patients without matched sibling or unrelated donor options. In an effort to expand the transplant pool and thus avoid clonal hematopoiesis, clinically significant paroxysmal nocturnal hemoglobinuria, and relapsed aplastic anemia, more work continues to recognize the expanding role of alternative donor transplants (cord blood and haploidentical) as another viable treatment strategy for aplastic anemia after immunosuppressive therapy failure.50
Summary
Aplastic anemia is a rare but potentially life-threatening disorder characterized by pancytopenia and a marked reduction in the hematopoietic stem cell compartment. Treatment should be instituted as soon as the dignosis of aplastic anemia is established. Treatment outcomes are excellent with modern supportive care and the current approach to allogeneic transplantation, and therefore referral to a bone marrow transplant program to evaluate for early transplantation is the new standard of care.
1. Peffault De Latour R, Le Rademacher J, Antin JH, et al. Allogeneic hematopoietic stem cell transplantation in Fanconi anemia: the European Group for Blood and Marrow Transplantation experience.” Blood. 2013;122:4279-4286.
2. Auerbach AD. Diagnosis of Fanconi anemia by diepoxybutane analysis. Curr Protoc Hum Genet. 2015;85:8.7.1-17.
3. Eapen M, et al. Effect of stem cell source on outcomes after unrelated donor transplantation in severe aplastic anemia. Blood. 2011;118:2618-2621.
4. Devillier R, Dalle JH, Kulasekararaj A, et al. Unrelated alternative donor transplantation for severe acquired aplastic anemia: a study from the French Society of Bone Marrow Transplantation and Cell Therapies and the Severe Aplastic Anemia Working Party of EBMT. Haematologica. 2016;101:884-890.
5. Peffault de Latour R, Peters C, Gibson B, et al. Recommendations on hematopoietic stem cell transplantation for inherited bone marrow failure syndromes.” Bone Marrow Transplant. 2015;50:1168-1172.
6. De Medeiros CR, Zanis-Neto J, Pasquini R. Bone marrow transplantation for patients with Fanconi anemia: reduced doses of cyclophosphamide without irradiation as conditioning. Bone Marrow Transplant. 1999;24:849-852.
7. Mohanan E, Panetta JC, Lakshmi KM, et al. Population pharmacokinetics of fludarabine in patients with aplastic anemia and Fanconi anemia undergoing allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2017;52:977-983.
8 Gluckman E, Auerbach AD, Horowitz MM, et al. Bone marrow transplantation for Fanconi anemia. Blood. 1995;86:2856-2862.
9. Maury S, Bacigalupo A, Anderlini P, et al. Improved outcome of patients older than 30 years receiving HLA-identical sibling hematopoietic stem cell transplantation for severe acquired aplastic anemia using fludarabine-based conditioning: a comparison with conventional conditioning regimen. Haematologica. 2009;94:1312-1315.
10. Talbot A, Peffault de Latour R, Raffoux E, et al. Sequential treatment for allogeneic hematopoietic stem cell transplantation in Fanconi anemia with acute myeloid leukemia. Haematologica. 2014;99:e199-200.
11. Ayas M, Saber W, Davies SM, et al. Allogeneic hematopoietic cell transplantation for fanconi anemia in patients with pretransplantation cytogenetic abnormalities, myelodysplastic syndrome, or acute leukemia. J Clin Oncol. 2013;31:1669-1676.
12. Vaht K, Göransson M, Carlson K, et al. Incidence and outcome of acquired aplastic anemia: real-world data from patients diagnosed in Sweden from 2000–2011. Haematologica. 2017;102:1683-1690.
13. Passweg JR, Marsh JC. Aplastic anemia: first-line treatment by immunosuppression and sibling marrow transplantation. Hematology Am Soc Hematol Educ Program. 2010;2010:36-42.
14. Laundy GJ, Bradley BA, Rees BM, et al. Incidence and specificity of HLA antibodies in multitransfused patients with acquired aplastic anemia. Transfusion. 2004;44:814-825.
15. Killick SB, Bown N, Cavenagh J, et al. Guidelines for the diagnosis and management of adult aplastic anaemia. Br J Haematol. 2016;172:187-207.
16. Scheinberg P, Nunez O, Weinstein B, et al. Horse versus rabbit antithymocyte globulin in acquired aplastic anemia. N Eng J Med. 2011;365:430-438.
17. Höchsmann B, Moicean A, Risitano A, et al. Supportive care in severe and very severe aplastic anemia. Bone Marrow Transplant. 2013;48:168-173.
18. Valdez JM, Scheinberg P, Young NS, Walsh TJ. Infections in patients with aplastic anemia. Sem Hematol. 2009;46:269-276.
19. Torres HA, Bodey GP, Rolston KV, et al. Infections in patients with aplastic anemia: experience at a tertiary care cancer center. Cancer. 2003;98:86-93.
20. Tichelli A, Schrezenmeier H, Socié G, et al. A randomized controlled study in patients with newly diagnosed severe aplastic anemia receiving antithymocyte globulin (ATG), cyclosporine, with or without G-CSF: a study of the SAA Working Party of the European Group for Blood and Marrow Transplantation. Blood. 2011;117:4434-4441.
21. Gerson SL, Talbot GH, Hurwitz S, et al. Prolonged granulocytopenia: the major risk factor for invasive pulmonary aspergillosis in patients with acute leukemia. Ann Intern Med. 1984;100:345-351.
22. Valdez JM, Scheinberg P, Nunez O, et al. Decreased infection-related mortality and improved survival in severe aplastic anemia in the past two decades. Clin Infect Dis. 2011;52:726-735.
23. Robenshtok E, Gafter-Gvili A, Goldberg E, et al. Antifungal prophylaxis in cancer patients after chemotherapy or hematopoietic stem-cell transplantation: systematic review and meta-analysis. J Clin Oncol. 2007;25:5471-5489.
24. Lee JW, Yoon SS, Shen ZX, et al. Iron chelation therapy with deferasirox in patients with aplastic anemia: a subgroup analysis of 116 patients from the EPIC trial. Blood. 2010;116:2448-2554.
25. Locasciulli A, Oneto R, Bacigalupo A, et al. Outcome of patients with acquired aplastic anemia given first line bone marrow transplantation or immunosuppressive treatment in the last decade: a report from the European Group for Blood and Marrow Transplantation. Haematologica. 2007;92:11-8.
26. Deeg HJ, Amylon MD, Harris RE, et al. Marrow transplants from unrelated donors for patients with aplastic anemia: minimum effective dose of total body irradiation. Biol Blood Marrow Transplant. 2001;7:208-215.
27. Kahl C, Leisenring W, Joachim Deeg H, et al. Cyclophosphamide and antithymocyte globulin as a conditioning regimen for allogeneic marrow transplantation in patients with aplastic anaemia: a long‐term follow‐up. Br J Haematol. 2005;130:747-751.
28. Socié G. Allogeneic BM transplantation for the treatment of aplastic anemia: current results and expanding donor possibilities. ASH Education Program Book. 2013;2013:82-86.
29. Shin SH, Jeon YW, Yoon JH, et al. Comparable outcomes between younger (<40 years) and older (>40 years) adult patients with severe aplastic anemia after HLA-matched sibling stem cell transplantation using fludarabine-based conditioning. Bone Marrow Transplant. 2016;51:1456-1463.
30. Kim H, Lee KH, Yoon SS, et al; Korean Society of Blood and Marrow Transplantation. Allogeneic hematopoietic stem cell transplant for adults over 40 years old with acquired aplastic anemia. Biol Blood Marrow Transplant. 2012;18:1500-1508.
31. Mortensen BK, Jacobsen N, Heilmann C, Sengelov H. Allogeneic hematopoietic cell transplantation for severe aplastic anemia: similar long-term overall survival after transplantation with related donors compared to unrelated donors. Bone Marrow Transplant. 2016;51:288-290.
32. Dufour C, Svahn J, Bacigalupo A. Front-line immunosuppressive treatment of acquired aplastic anemia. Bone Marrow Transplant. 2013;48:174-177.
33. Dufour C, Veys P, Carraro E, et al. Similar outcome of upfront-unrelated and matched sibling stem cell transplantation in idiopathic paediatric aplastic anaemia. A study on the behalf of the UK Paediatric BMT Working Party, Paediatric Diseases Working Party and Severe Aplastic Anaemia Working Party of the EBMT. Br. J Haematol. 2015;151:585-594.
34. Georges GE, Doney K, Storb R. Severe aplastic anemia: allogeneic bone marrow transplantation as first-line treatment. Blood Adv. 2018;2:2020-2028.
35. Yoshida N, Kojima S. Updated guidelines for the treatment of acquired aplastic anemia in children. Curr Oncol Rep. 2018;20:67.
36. Mathe G, Amiel JL, Schwarzenberg L, et al. Bone marrow graft in man after conditioning by antilymphocytic serum. Br Med J. 1970;2:131-136.
37. Frickhofen N, Kaltwasser JP, Schrezenmeier H, et al, German Aplastic Anemia Study Group. Treatment of aplastic anemia with antilymphocyte globulin and methylprednisolone with or without cyclosporine. N Engl J Med. 1991;324:1297-1304.
38. Speck B, Gratwohl A, Nissen C, et al. Treatment of severe aplastic anaemia with antilymphocyte globulin or bone-marrow transplantation. Br Med J. 1981;282:860-863.
39. Al-Ghazaly J, Al-Dubai W, Al-Jahafi AK, et al. Cyclosporine monotherapy for severe aplastic anemia: a developing country experience. Ann Saudi Med. 2005;25:375-379.
40. Scheinberg P, Young NS. How I treat acquired aplastic anemia. Blood. 2012;120:1185-1196.
41. Rosenfeld S, Follmann D, Nunez O, Young NS. Antithymocyte globulin and cyclosporine for severe aplastic anemia: association between hematologic response and long-term outcome. JAMA. 2003;289:1130-1135.
42. Saracco P, Quarello P, Iori AP, et al. Cyclosporin A response and dependence in children with acquired aplastic anaemia: a multicentre retrospective study with long‐term observation follow‐up. Br J Haematol. 2008;140:197-205.
43. Bacigalupo A. How I treat acquired aplastic anemia. Blood. 2017;129:1428-1436.
44. Townsley DM, Scheinberg P, Winkler T, et al. Eltrombopag added to standard immunosuppression for aplastic anemia. N Engl J Med. 2017;376:1540-1550.
45. Olnes MJ, Scheinberg P, Calvo KR, et al. Eltrombopag and improved hematopoiesis in refractory aplastic anemia. N Engl J Med. 2012;367:11-19.
46. Assi R, Garcia-Manero G, Ravandi F, et al. Addition of eltrombopag to immunosuppressive therapy in patients with newly diagnosed aplastic anemia. Cancer. 2018 Oct 11. doi: 10.1002/cncr.31658.
47. Bacigalupo A, Socié G, Hamladji RM, et al. Current outcome of HLA identical sibling vs. unrelated donor transplants in severe aplastic anemia: an EBMT analysis. Haematologica. 2015;100:696-702.
48. Samarasinghe S, Iacobelli S, Knol C, et al. Impact of different in vivo T cell depletion strategies on outcomes following hematopoietic stem cell transplantation for idiopathic aplastic anaemia: a study on behalf of the EBMT SAA Working Party. 2018Oct 17. doi: 10.1002/ajh.25314.
49. Clesham K, Dowse R, Samarasinghe S. Upfront matched unrelated donor transplantation in aplastic anemia. Hematol Oncol Clin North Am. 2018;32:619-628.
50. DeZern AE, Brodsky RA. Haploidentical donor bone marrow transplantation for severe aplastic anemia. Hematol Oncol Clin North Am. 2018;32:629-642.
1. Peffault De Latour R, Le Rademacher J, Antin JH, et al. Allogeneic hematopoietic stem cell transplantation in Fanconi anemia: the European Group for Blood and Marrow Transplantation experience.” Blood. 2013;122:4279-4286.
2. Auerbach AD. Diagnosis of Fanconi anemia by diepoxybutane analysis. Curr Protoc Hum Genet. 2015;85:8.7.1-17.
3. Eapen M, et al. Effect of stem cell source on outcomes after unrelated donor transplantation in severe aplastic anemia. Blood. 2011;118:2618-2621.
4. Devillier R, Dalle JH, Kulasekararaj A, et al. Unrelated alternative donor transplantation for severe acquired aplastic anemia: a study from the French Society of Bone Marrow Transplantation and Cell Therapies and the Severe Aplastic Anemia Working Party of EBMT. Haematologica. 2016;101:884-890.
5. Peffault de Latour R, Peters C, Gibson B, et al. Recommendations on hematopoietic stem cell transplantation for inherited bone marrow failure syndromes.” Bone Marrow Transplant. 2015;50:1168-1172.
6. De Medeiros CR, Zanis-Neto J, Pasquini R. Bone marrow transplantation for patients with Fanconi anemia: reduced doses of cyclophosphamide without irradiation as conditioning. Bone Marrow Transplant. 1999;24:849-852.
7. Mohanan E, Panetta JC, Lakshmi KM, et al. Population pharmacokinetics of fludarabine in patients with aplastic anemia and Fanconi anemia undergoing allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2017;52:977-983.
8 Gluckman E, Auerbach AD, Horowitz MM, et al. Bone marrow transplantation for Fanconi anemia. Blood. 1995;86:2856-2862.
9. Maury S, Bacigalupo A, Anderlini P, et al. Improved outcome of patients older than 30 years receiving HLA-identical sibling hematopoietic stem cell transplantation for severe acquired aplastic anemia using fludarabine-based conditioning: a comparison with conventional conditioning regimen. Haematologica. 2009;94:1312-1315.
10. Talbot A, Peffault de Latour R, Raffoux E, et al. Sequential treatment for allogeneic hematopoietic stem cell transplantation in Fanconi anemia with acute myeloid leukemia. Haematologica. 2014;99:e199-200.
11. Ayas M, Saber W, Davies SM, et al. Allogeneic hematopoietic cell transplantation for fanconi anemia in patients with pretransplantation cytogenetic abnormalities, myelodysplastic syndrome, or acute leukemia. J Clin Oncol. 2013;31:1669-1676.
12. Vaht K, Göransson M, Carlson K, et al. Incidence and outcome of acquired aplastic anemia: real-world data from patients diagnosed in Sweden from 2000–2011. Haematologica. 2017;102:1683-1690.
13. Passweg JR, Marsh JC. Aplastic anemia: first-line treatment by immunosuppression and sibling marrow transplantation. Hematology Am Soc Hematol Educ Program. 2010;2010:36-42.
14. Laundy GJ, Bradley BA, Rees BM, et al. Incidence and specificity of HLA antibodies in multitransfused patients with acquired aplastic anemia. Transfusion. 2004;44:814-825.
15. Killick SB, Bown N, Cavenagh J, et al. Guidelines for the diagnosis and management of adult aplastic anaemia. Br J Haematol. 2016;172:187-207.
16. Scheinberg P, Nunez O, Weinstein B, et al. Horse versus rabbit antithymocyte globulin in acquired aplastic anemia. N Eng J Med. 2011;365:430-438.
17. Höchsmann B, Moicean A, Risitano A, et al. Supportive care in severe and very severe aplastic anemia. Bone Marrow Transplant. 2013;48:168-173.
18. Valdez JM, Scheinberg P, Young NS, Walsh TJ. Infections in patients with aplastic anemia. Sem Hematol. 2009;46:269-276.
19. Torres HA, Bodey GP, Rolston KV, et al. Infections in patients with aplastic anemia: experience at a tertiary care cancer center. Cancer. 2003;98:86-93.
20. Tichelli A, Schrezenmeier H, Socié G, et al. A randomized controlled study in patients with newly diagnosed severe aplastic anemia receiving antithymocyte globulin (ATG), cyclosporine, with or without G-CSF: a study of the SAA Working Party of the European Group for Blood and Marrow Transplantation. Blood. 2011;117:4434-4441.
21. Gerson SL, Talbot GH, Hurwitz S, et al. Prolonged granulocytopenia: the major risk factor for invasive pulmonary aspergillosis in patients with acute leukemia. Ann Intern Med. 1984;100:345-351.
22. Valdez JM, Scheinberg P, Nunez O, et al. Decreased infection-related mortality and improved survival in severe aplastic anemia in the past two decades. Clin Infect Dis. 2011;52:726-735.
23. Robenshtok E, Gafter-Gvili A, Goldberg E, et al. Antifungal prophylaxis in cancer patients after chemotherapy or hematopoietic stem-cell transplantation: systematic review and meta-analysis. J Clin Oncol. 2007;25:5471-5489.
24. Lee JW, Yoon SS, Shen ZX, et al. Iron chelation therapy with deferasirox in patients with aplastic anemia: a subgroup analysis of 116 patients from the EPIC trial. Blood. 2010;116:2448-2554.
25. Locasciulli A, Oneto R, Bacigalupo A, et al. Outcome of patients with acquired aplastic anemia given first line bone marrow transplantation or immunosuppressive treatment in the last decade: a report from the European Group for Blood and Marrow Transplantation. Haematologica. 2007;92:11-8.
26. Deeg HJ, Amylon MD, Harris RE, et al. Marrow transplants from unrelated donors for patients with aplastic anemia: minimum effective dose of total body irradiation. Biol Blood Marrow Transplant. 2001;7:208-215.
27. Kahl C, Leisenring W, Joachim Deeg H, et al. Cyclophosphamide and antithymocyte globulin as a conditioning regimen for allogeneic marrow transplantation in patients with aplastic anaemia: a long‐term follow‐up. Br J Haematol. 2005;130:747-751.
28. Socié G. Allogeneic BM transplantation for the treatment of aplastic anemia: current results and expanding donor possibilities. ASH Education Program Book. 2013;2013:82-86.
29. Shin SH, Jeon YW, Yoon JH, et al. Comparable outcomes between younger (<40 years) and older (>40 years) adult patients with severe aplastic anemia after HLA-matched sibling stem cell transplantation using fludarabine-based conditioning. Bone Marrow Transplant. 2016;51:1456-1463.
30. Kim H, Lee KH, Yoon SS, et al; Korean Society of Blood and Marrow Transplantation. Allogeneic hematopoietic stem cell transplant for adults over 40 years old with acquired aplastic anemia. Biol Blood Marrow Transplant. 2012;18:1500-1508.
31. Mortensen BK, Jacobsen N, Heilmann C, Sengelov H. Allogeneic hematopoietic cell transplantation for severe aplastic anemia: similar long-term overall survival after transplantation with related donors compared to unrelated donors. Bone Marrow Transplant. 2016;51:288-290.
32. Dufour C, Svahn J, Bacigalupo A. Front-line immunosuppressive treatment of acquired aplastic anemia. Bone Marrow Transplant. 2013;48:174-177.
33. Dufour C, Veys P, Carraro E, et al. Similar outcome of upfront-unrelated and matched sibling stem cell transplantation in idiopathic paediatric aplastic anaemia. A study on the behalf of the UK Paediatric BMT Working Party, Paediatric Diseases Working Party and Severe Aplastic Anaemia Working Party of the EBMT. Br. J Haematol. 2015;151:585-594.
34. Georges GE, Doney K, Storb R. Severe aplastic anemia: allogeneic bone marrow transplantation as first-line treatment. Blood Adv. 2018;2:2020-2028.
35. Yoshida N, Kojima S. Updated guidelines for the treatment of acquired aplastic anemia in children. Curr Oncol Rep. 2018;20:67.
36. Mathe G, Amiel JL, Schwarzenberg L, et al. Bone marrow graft in man after conditioning by antilymphocytic serum. Br Med J. 1970;2:131-136.
37. Frickhofen N, Kaltwasser JP, Schrezenmeier H, et al, German Aplastic Anemia Study Group. Treatment of aplastic anemia with antilymphocyte globulin and methylprednisolone with or without cyclosporine. N Engl J Med. 1991;324:1297-1304.
38. Speck B, Gratwohl A, Nissen C, et al. Treatment of severe aplastic anaemia with antilymphocyte globulin or bone-marrow transplantation. Br Med J. 1981;282:860-863.
39. Al-Ghazaly J, Al-Dubai W, Al-Jahafi AK, et al. Cyclosporine monotherapy for severe aplastic anemia: a developing country experience. Ann Saudi Med. 2005;25:375-379.
40. Scheinberg P, Young NS. How I treat acquired aplastic anemia. Blood. 2012;120:1185-1196.
41. Rosenfeld S, Follmann D, Nunez O, Young NS. Antithymocyte globulin and cyclosporine for severe aplastic anemia: association between hematologic response and long-term outcome. JAMA. 2003;289:1130-1135.
42. Saracco P, Quarello P, Iori AP, et al. Cyclosporin A response and dependence in children with acquired aplastic anaemia: a multicentre retrospective study with long‐term observation follow‐up. Br J Haematol. 2008;140:197-205.
43. Bacigalupo A. How I treat acquired aplastic anemia. Blood. 2017;129:1428-1436.
44. Townsley DM, Scheinberg P, Winkler T, et al. Eltrombopag added to standard immunosuppression for aplastic anemia. N Engl J Med. 2017;376:1540-1550.
45. Olnes MJ, Scheinberg P, Calvo KR, et al. Eltrombopag and improved hematopoiesis in refractory aplastic anemia. N Engl J Med. 2012;367:11-19.
46. Assi R, Garcia-Manero G, Ravandi F, et al. Addition of eltrombopag to immunosuppressive therapy in patients with newly diagnosed aplastic anemia. Cancer. 2018 Oct 11. doi: 10.1002/cncr.31658.
47. Bacigalupo A, Socié G, Hamladji RM, et al. Current outcome of HLA identical sibling vs. unrelated donor transplants in severe aplastic anemia: an EBMT analysis. Haematologica. 2015;100:696-702.
48. Samarasinghe S, Iacobelli S, Knol C, et al. Impact of different in vivo T cell depletion strategies on outcomes following hematopoietic stem cell transplantation for idiopathic aplastic anaemia: a study on behalf of the EBMT SAA Working Party. 2018Oct 17. doi: 10.1002/ajh.25314.
49. Clesham K, Dowse R, Samarasinghe S. Upfront matched unrelated donor transplantation in aplastic anemia. Hematol Oncol Clin North Am. 2018;32:619-628.
50. DeZern AE, Brodsky RA. Haploidentical donor bone marrow transplantation for severe aplastic anemia. Hematol Oncol Clin North Am. 2018;32:629-642.
AML, myeloma risk higher for breast cancer survivors
Breast cancer survivors should continue to be monitored for hematologic malignancies, especially acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), results of a population-based study from France suggest.
Among nearly 440,000 women with an incident breast cancer diagnosis, the incidence of AML was nearly three times higher and the incidence of MDS was five times higher than that of women in the general population. Women with breast cancer also were at higher risk for multiple myeloma (MM) and acute lymphoblastic leukemia/lymphocytic lymphoma (ALL/LL) compared with the background population, reported Marie Joelle Jabagi, PharmD, MPH, of the University of Paris Sud, France, and her colleagues.
“These findings serve to better inform practicing oncologists, and breast cancer survivors should be advised of the increased risk of developing certain hematologic malignant neoplasms after their first cancer diagnosis,” they wrote in JAMA Network Open.
Breast cancers are the malignant solid tumors most frequently associated with risk for myeloid neoplasms, but there is little information on the risk for secondary lymphoid malignancies among breast cancer patients, the investigators stated.
“In addition, real-life data on secondary hematologic malignant neoplasm incidence are scarce, especially in the recent period marked by major advances in breast cancer treatments,” they wrote.
To get better estimates of the incidence of myeloid and lymphoid neoplasms in this population, they conducted a retrospective review of information from the French National Health Data System on all French women from the ages of 20 to 85 years who had an incident breast cancer diagnosis from July 1, 2006, through Dec. 31, 2015.
In all, 439,704 women with a median age of 59 years were identified. They were followed until a diagnosis of a hematologic malignancy, death, or loss to follow-up, or until Dec. 31, 2016.
Data on the breast cancer patients were compared with those for all French women in the general population who were registered in the general national health insurance program from January 2007 through the end of 2016.
During a median follow-up of 5 years, there were 3,046 cases of hematologic neoplasms among the breast cancer patients, including 509 cases of AML, for a crude incidence rate (CIR) of 24.5 per 100,000 person-years (py); 832 cases of MDS for a CIR of 40.1/100,000 py; and 267 cases of myeloproliferative neoplasms (MPN), for a CIR of 12.8/100,000 py.
In addition, there were 420 cases of MM for a CIR of 20.3/100,000 py; 912 cases of Hodgkin or non-Hodgkin lymphoma (HL/NHL) for a CIR of 44.4/100,000 py, and 106 cases of ALL/LL for a CIR of 5.1/100,000 py.
Breast cancer survivors had significantly higher incidences, compared with the general population, of AML (standardized incidence ratio [SIR] 2.8, 95% confidence interval [CI], 2.5-3.2), MDS (SIR 5.0, CI, 4.4-5.7), MM (SIR 1.5, CI, 1.3-17), and ALL/LL (SIR 2.0, CI, 1.3-3.0). There was a trend toward significance for both MPN and HL/NHL, but the lower limit of the confidence intervals for these conditions either crossed or touched 1.
In a review of the literature, the authors found that “[s]everal studies linked AML and MDS to chemotherapeutic agents, radiation treatment, and supportive treatment with granulocyte colony-stimulating factor. These results are consistent with other available data showing a 2½-fold to 3½-fold increased risk of AML.”
They noted that their estimate of a five-fold increase in risk for MDS was higher than the 3.7-fold risk reported in a previous registry cohort analysis, suggesting that risk for MDS among breast cancer patients may be underestimated.
“The recent discovery of the gene signatures that guide treatment decisions in early-stage breast cancer might reduce the number of patients exposed to cytotoxic chemotherapy and its complications, including hematologic malignant neoplasm. Therefore, continuing to monitor hematologic malignant neoplasm trends is necessary, especially given that approaches to cancer treatment are rapidly evolving. Further research is also required to assess the modality of treatment for and the genetic predisposition to these secondary malignant neoplasms,” the authors concluded.
SOURCE: Jabagi MJ et al. JAMA Network Open. 2019 Jan 18. doi: 10.1001/jamanetworkopen.2018.7147.
Breast cancer survivors should continue to be monitored for hematologic malignancies, especially acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), results of a population-based study from France suggest.
Among nearly 440,000 women with an incident breast cancer diagnosis, the incidence of AML was nearly three times higher and the incidence of MDS was five times higher than that of women in the general population. Women with breast cancer also were at higher risk for multiple myeloma (MM) and acute lymphoblastic leukemia/lymphocytic lymphoma (ALL/LL) compared with the background population, reported Marie Joelle Jabagi, PharmD, MPH, of the University of Paris Sud, France, and her colleagues.
“These findings serve to better inform practicing oncologists, and breast cancer survivors should be advised of the increased risk of developing certain hematologic malignant neoplasms after their first cancer diagnosis,” they wrote in JAMA Network Open.
Breast cancers are the malignant solid tumors most frequently associated with risk for myeloid neoplasms, but there is little information on the risk for secondary lymphoid malignancies among breast cancer patients, the investigators stated.
“In addition, real-life data on secondary hematologic malignant neoplasm incidence are scarce, especially in the recent period marked by major advances in breast cancer treatments,” they wrote.
To get better estimates of the incidence of myeloid and lymphoid neoplasms in this population, they conducted a retrospective review of information from the French National Health Data System on all French women from the ages of 20 to 85 years who had an incident breast cancer diagnosis from July 1, 2006, through Dec. 31, 2015.
In all, 439,704 women with a median age of 59 years were identified. They were followed until a diagnosis of a hematologic malignancy, death, or loss to follow-up, or until Dec. 31, 2016.
Data on the breast cancer patients were compared with those for all French women in the general population who were registered in the general national health insurance program from January 2007 through the end of 2016.
During a median follow-up of 5 years, there were 3,046 cases of hematologic neoplasms among the breast cancer patients, including 509 cases of AML, for a crude incidence rate (CIR) of 24.5 per 100,000 person-years (py); 832 cases of MDS for a CIR of 40.1/100,000 py; and 267 cases of myeloproliferative neoplasms (MPN), for a CIR of 12.8/100,000 py.
In addition, there were 420 cases of MM for a CIR of 20.3/100,000 py; 912 cases of Hodgkin or non-Hodgkin lymphoma (HL/NHL) for a CIR of 44.4/100,000 py, and 106 cases of ALL/LL for a CIR of 5.1/100,000 py.
Breast cancer survivors had significantly higher incidences, compared with the general population, of AML (standardized incidence ratio [SIR] 2.8, 95% confidence interval [CI], 2.5-3.2), MDS (SIR 5.0, CI, 4.4-5.7), MM (SIR 1.5, CI, 1.3-17), and ALL/LL (SIR 2.0, CI, 1.3-3.0). There was a trend toward significance for both MPN and HL/NHL, but the lower limit of the confidence intervals for these conditions either crossed or touched 1.
In a review of the literature, the authors found that “[s]everal studies linked AML and MDS to chemotherapeutic agents, radiation treatment, and supportive treatment with granulocyte colony-stimulating factor. These results are consistent with other available data showing a 2½-fold to 3½-fold increased risk of AML.”
They noted that their estimate of a five-fold increase in risk for MDS was higher than the 3.7-fold risk reported in a previous registry cohort analysis, suggesting that risk for MDS among breast cancer patients may be underestimated.
“The recent discovery of the gene signatures that guide treatment decisions in early-stage breast cancer might reduce the number of patients exposed to cytotoxic chemotherapy and its complications, including hematologic malignant neoplasm. Therefore, continuing to monitor hematologic malignant neoplasm trends is necessary, especially given that approaches to cancer treatment are rapidly evolving. Further research is also required to assess the modality of treatment for and the genetic predisposition to these secondary malignant neoplasms,” the authors concluded.
SOURCE: Jabagi MJ et al. JAMA Network Open. 2019 Jan 18. doi: 10.1001/jamanetworkopen.2018.7147.
Breast cancer survivors should continue to be monitored for hematologic malignancies, especially acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), results of a population-based study from France suggest.
Among nearly 440,000 women with an incident breast cancer diagnosis, the incidence of AML was nearly three times higher and the incidence of MDS was five times higher than that of women in the general population. Women with breast cancer also were at higher risk for multiple myeloma (MM) and acute lymphoblastic leukemia/lymphocytic lymphoma (ALL/LL) compared with the background population, reported Marie Joelle Jabagi, PharmD, MPH, of the University of Paris Sud, France, and her colleagues.
“These findings serve to better inform practicing oncologists, and breast cancer survivors should be advised of the increased risk of developing certain hematologic malignant neoplasms after their first cancer diagnosis,” they wrote in JAMA Network Open.
Breast cancers are the malignant solid tumors most frequently associated with risk for myeloid neoplasms, but there is little information on the risk for secondary lymphoid malignancies among breast cancer patients, the investigators stated.
“In addition, real-life data on secondary hematologic malignant neoplasm incidence are scarce, especially in the recent period marked by major advances in breast cancer treatments,” they wrote.
To get better estimates of the incidence of myeloid and lymphoid neoplasms in this population, they conducted a retrospective review of information from the French National Health Data System on all French women from the ages of 20 to 85 years who had an incident breast cancer diagnosis from July 1, 2006, through Dec. 31, 2015.
In all, 439,704 women with a median age of 59 years were identified. They were followed until a diagnosis of a hematologic malignancy, death, or loss to follow-up, or until Dec. 31, 2016.
Data on the breast cancer patients were compared with those for all French women in the general population who were registered in the general national health insurance program from January 2007 through the end of 2016.
During a median follow-up of 5 years, there were 3,046 cases of hematologic neoplasms among the breast cancer patients, including 509 cases of AML, for a crude incidence rate (CIR) of 24.5 per 100,000 person-years (py); 832 cases of MDS for a CIR of 40.1/100,000 py; and 267 cases of myeloproliferative neoplasms (MPN), for a CIR of 12.8/100,000 py.
In addition, there were 420 cases of MM for a CIR of 20.3/100,000 py; 912 cases of Hodgkin or non-Hodgkin lymphoma (HL/NHL) for a CIR of 44.4/100,000 py, and 106 cases of ALL/LL for a CIR of 5.1/100,000 py.
Breast cancer survivors had significantly higher incidences, compared with the general population, of AML (standardized incidence ratio [SIR] 2.8, 95% confidence interval [CI], 2.5-3.2), MDS (SIR 5.0, CI, 4.4-5.7), MM (SIR 1.5, CI, 1.3-17), and ALL/LL (SIR 2.0, CI, 1.3-3.0). There was a trend toward significance for both MPN and HL/NHL, but the lower limit of the confidence intervals for these conditions either crossed or touched 1.
In a review of the literature, the authors found that “[s]everal studies linked AML and MDS to chemotherapeutic agents, radiation treatment, and supportive treatment with granulocyte colony-stimulating factor. These results are consistent with other available data showing a 2½-fold to 3½-fold increased risk of AML.”
They noted that their estimate of a five-fold increase in risk for MDS was higher than the 3.7-fold risk reported in a previous registry cohort analysis, suggesting that risk for MDS among breast cancer patients may be underestimated.
“The recent discovery of the gene signatures that guide treatment decisions in early-stage breast cancer might reduce the number of patients exposed to cytotoxic chemotherapy and its complications, including hematologic malignant neoplasm. Therefore, continuing to monitor hematologic malignant neoplasm trends is necessary, especially given that approaches to cancer treatment are rapidly evolving. Further research is also required to assess the modality of treatment for and the genetic predisposition to these secondary malignant neoplasms,” the authors concluded.
SOURCE: Jabagi MJ et al. JAMA Network Open. 2019 Jan 18. doi: 10.1001/jamanetworkopen.2018.7147.
FROM JAMA NETWORK OPEN
Key clinical point: Breast cancer survivors should be monitored for hematologic malignancies.
Major finding: The standardized incidence ratio for AML was 2.8 and the SIR for multiple myeloma was 5.0 among French breast cancer survivors compared with women in the general French population.
Study details: Retrospective analysis of data on 439,704 women aged 20-85 years with a breast cancer diagnosis.
Disclosures: The authors did not report a study funding source. Coauthor Anthony Goncalves, MD, reported nonfinancial support from Roche, Novartis, Pfizer, Celgene, MSD, Lilly, and Astra Zeneca outside of the submitted work. No other disclosures were reported.
Source: Jabagi MJ et al. JAMA Network Open. 2019 Jan 18. doi: 10.1001/jamanetworkopen.2018.7147.





