User login
Caring for Noncritically Ill Coronavirus Patients
The early days of the coronavirus disease 2019 (COVID-19) pandemic were fraught with uncertainty as hospitalists struggled to develop standards of care for noncritically ill patients. Although data were available from intensive care units (ICUs) in Asia and Europe, it was unclear whether these findings applied to the acute but noncritically ill patients who would ultimately make up most coronavirus admissions. Which therapeutics could benefit these patients? Who needs continuous cardiopulmonary monitoring? And perhaps most importantly, which patients are at risk for clinical deterioration?
In this issue, Nemer et al begin to answer these questions using a retrospective analysis of 350 noncritically ill COVID-19 patients admitted to non-ICU care at Cleveland Clinic hospitals in Ohio and Florida between March 13 and May 1, 2020.1 The primary outcome was a composite of three endpoints: increased respiratory support (high-flow nasal cannula, noninvasive positive pressure ventilation, or intubation), ICU transfer, or death. The primary outcome occurred in 18% of all patients and the risk was greatest among patients with high admission levels of C-reactive protein (CRP). This analysis found that while clinically significant arrhythmias occurred in 14% of patients, 90% of those were in patients with either known cardiac disease or an elevated admission troponin T level and in only one case (<1%) necessitated transition to a higher level of care. Overall mortality for COVID-19 patients initially admitted to non-ICU settings was 3%.
While several tests have been proposed as clinically relevant to coronavirus disease, those recommendations are based on studies performed on critically ill patients outside of the US and have focused on survival, not clinical deterioration.2,3 In their cohort of noncritically ill patients in the US, Nemer et al found that not only is CRP associated with clinical worsening, but that increasing levels of CRP are associated with increasing risk of deterioration. Perhaps even more interesting was the finding that no patient with a normal CRP suffered the composite outcome, including death. The authors did not report levels of other laboratory tests that have been associated with severe coronavirus disease, such as platelets, fibrin degradation products, or prolonged prothrombin time/activated partial thromboplastin time. As many clinicians will note, CRP’s lack of specificity may be its Achilles heel, potentially lowering its prognostic value. Still, given its wide availability, low cost, and rapid turnaround, CRP could serve as a screening tool to risk stratify admitted coronavirus patients, while also providing reassurance when it is normal.
The results of this study could also impact use of hospital resources. The findings regarding the low risk of arrhythmias provide support for limiting the use of continuous cardiac monitoring in noncritically ill patients without previous histories of cardiac disease or elevated admission troponin levels. Patients with normal admission CRP levels could potentially be monitored safely with intermittent pulse oximetry. Continuous pulse oximetry and cardiac monitoring are already overused in many hospitals, and in the case of coronavirus the implications are even more significant given the importance of minimizing unnecessary healthcare worker exposures.
The vast majority (79% to 90%) of patients hospitalized for coronavirus will be cared for in non–ICU settings,4,5 yet most research has thus far focused on ICU patients. Nemer et al provide much-needed information on how to care for the noncritically ill coronavirus patients whom hospitalists are most likely to treat. As a resurgence of infections is expected this winter, this work has the potential to help physicians identify not only those who have the highest probability of deteriorating, but also those who may not. In a world of limited resources, knowing which patient is unlikely to deteriorate may be just as important as recognizing which one is.
1. Nemer D, Wilner BR, Burkle A, et al. Clinical characteristics and outcomes of non-ICU hospitalization for COVID-19 in a nonepicenter, centrally monitored healthcare system. J Hosp Med. 2021;16:7-14. https://doi.org/10.12788/jhm.3510
2. Lippi G, Pleban M, Henry B. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin Chim Acta. 2020;506:145-148. https://doi.org/10.1016/j.cca.2020.03.022
3. Klok FA, Kruip MJHA, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145-147. https://doi.org/10.1016/j.thromres.2020.04.013
4. Giannakeas V, Bhatia D, Warkentin M, et al. Estimating the maximum capacity of COVID-19 cases manageable per day given a health care system’s constrained resources. Ann Intern Med. April 16, 2020. https://doi.org/10.7326/M20-1169
5. Tsai T, Jacobson B, Jha A. American hospital capacity and projected need for COVID-19 patient care. Health Affairs blog. March 17, 2020. Accessed October 12, 2020. https://www.healthaffairs.org/do/10.1377/hblog20200317.457910/full/
The early days of the coronavirus disease 2019 (COVID-19) pandemic were fraught with uncertainty as hospitalists struggled to develop standards of care for noncritically ill patients. Although data were available from intensive care units (ICUs) in Asia and Europe, it was unclear whether these findings applied to the acute but noncritically ill patients who would ultimately make up most coronavirus admissions. Which therapeutics could benefit these patients? Who needs continuous cardiopulmonary monitoring? And perhaps most importantly, which patients are at risk for clinical deterioration?
In this issue, Nemer et al begin to answer these questions using a retrospective analysis of 350 noncritically ill COVID-19 patients admitted to non-ICU care at Cleveland Clinic hospitals in Ohio and Florida between March 13 and May 1, 2020.1 The primary outcome was a composite of three endpoints: increased respiratory support (high-flow nasal cannula, noninvasive positive pressure ventilation, or intubation), ICU transfer, or death. The primary outcome occurred in 18% of all patients and the risk was greatest among patients with high admission levels of C-reactive protein (CRP). This analysis found that while clinically significant arrhythmias occurred in 14% of patients, 90% of those were in patients with either known cardiac disease or an elevated admission troponin T level and in only one case (<1%) necessitated transition to a higher level of care. Overall mortality for COVID-19 patients initially admitted to non-ICU settings was 3%.
While several tests have been proposed as clinically relevant to coronavirus disease, those recommendations are based on studies performed on critically ill patients outside of the US and have focused on survival, not clinical deterioration.2,3 In their cohort of noncritically ill patients in the US, Nemer et al found that not only is CRP associated with clinical worsening, but that increasing levels of CRP are associated with increasing risk of deterioration. Perhaps even more interesting was the finding that no patient with a normal CRP suffered the composite outcome, including death. The authors did not report levels of other laboratory tests that have been associated with severe coronavirus disease, such as platelets, fibrin degradation products, or prolonged prothrombin time/activated partial thromboplastin time. As many clinicians will note, CRP’s lack of specificity may be its Achilles heel, potentially lowering its prognostic value. Still, given its wide availability, low cost, and rapid turnaround, CRP could serve as a screening tool to risk stratify admitted coronavirus patients, while also providing reassurance when it is normal.
The results of this study could also impact use of hospital resources. The findings regarding the low risk of arrhythmias provide support for limiting the use of continuous cardiac monitoring in noncritically ill patients without previous histories of cardiac disease or elevated admission troponin levels. Patients with normal admission CRP levels could potentially be monitored safely with intermittent pulse oximetry. Continuous pulse oximetry and cardiac monitoring are already overused in many hospitals, and in the case of coronavirus the implications are even more significant given the importance of minimizing unnecessary healthcare worker exposures.
The vast majority (79% to 90%) of patients hospitalized for coronavirus will be cared for in non–ICU settings,4,5 yet most research has thus far focused on ICU patients. Nemer et al provide much-needed information on how to care for the noncritically ill coronavirus patients whom hospitalists are most likely to treat. As a resurgence of infections is expected this winter, this work has the potential to help physicians identify not only those who have the highest probability of deteriorating, but also those who may not. In a world of limited resources, knowing which patient is unlikely to deteriorate may be just as important as recognizing which one is.
The early days of the coronavirus disease 2019 (COVID-19) pandemic were fraught with uncertainty as hospitalists struggled to develop standards of care for noncritically ill patients. Although data were available from intensive care units (ICUs) in Asia and Europe, it was unclear whether these findings applied to the acute but noncritically ill patients who would ultimately make up most coronavirus admissions. Which therapeutics could benefit these patients? Who needs continuous cardiopulmonary monitoring? And perhaps most importantly, which patients are at risk for clinical deterioration?
In this issue, Nemer et al begin to answer these questions using a retrospective analysis of 350 noncritically ill COVID-19 patients admitted to non-ICU care at Cleveland Clinic hospitals in Ohio and Florida between March 13 and May 1, 2020.1 The primary outcome was a composite of three endpoints: increased respiratory support (high-flow nasal cannula, noninvasive positive pressure ventilation, or intubation), ICU transfer, or death. The primary outcome occurred in 18% of all patients and the risk was greatest among patients with high admission levels of C-reactive protein (CRP). This analysis found that while clinically significant arrhythmias occurred in 14% of patients, 90% of those were in patients with either known cardiac disease or an elevated admission troponin T level and in only one case (<1%) necessitated transition to a higher level of care. Overall mortality for COVID-19 patients initially admitted to non-ICU settings was 3%.
While several tests have been proposed as clinically relevant to coronavirus disease, those recommendations are based on studies performed on critically ill patients outside of the US and have focused on survival, not clinical deterioration.2,3 In their cohort of noncritically ill patients in the US, Nemer et al found that not only is CRP associated with clinical worsening, but that increasing levels of CRP are associated with increasing risk of deterioration. Perhaps even more interesting was the finding that no patient with a normal CRP suffered the composite outcome, including death. The authors did not report levels of other laboratory tests that have been associated with severe coronavirus disease, such as platelets, fibrin degradation products, or prolonged prothrombin time/activated partial thromboplastin time. As many clinicians will note, CRP’s lack of specificity may be its Achilles heel, potentially lowering its prognostic value. Still, given its wide availability, low cost, and rapid turnaround, CRP could serve as a screening tool to risk stratify admitted coronavirus patients, while also providing reassurance when it is normal.
The results of this study could also impact use of hospital resources. The findings regarding the low risk of arrhythmias provide support for limiting the use of continuous cardiac monitoring in noncritically ill patients without previous histories of cardiac disease or elevated admission troponin levels. Patients with normal admission CRP levels could potentially be monitored safely with intermittent pulse oximetry. Continuous pulse oximetry and cardiac monitoring are already overused in many hospitals, and in the case of coronavirus the implications are even more significant given the importance of minimizing unnecessary healthcare worker exposures.
The vast majority (79% to 90%) of patients hospitalized for coronavirus will be cared for in non–ICU settings,4,5 yet most research has thus far focused on ICU patients. Nemer et al provide much-needed information on how to care for the noncritically ill coronavirus patients whom hospitalists are most likely to treat. As a resurgence of infections is expected this winter, this work has the potential to help physicians identify not only those who have the highest probability of deteriorating, but also those who may not. In a world of limited resources, knowing which patient is unlikely to deteriorate may be just as important as recognizing which one is.
1. Nemer D, Wilner BR, Burkle A, et al. Clinical characteristics and outcomes of non-ICU hospitalization for COVID-19 in a nonepicenter, centrally monitored healthcare system. J Hosp Med. 2021;16:7-14. https://doi.org/10.12788/jhm.3510
2. Lippi G, Pleban M, Henry B. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin Chim Acta. 2020;506:145-148. https://doi.org/10.1016/j.cca.2020.03.022
3. Klok FA, Kruip MJHA, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145-147. https://doi.org/10.1016/j.thromres.2020.04.013
4. Giannakeas V, Bhatia D, Warkentin M, et al. Estimating the maximum capacity of COVID-19 cases manageable per day given a health care system’s constrained resources. Ann Intern Med. April 16, 2020. https://doi.org/10.7326/M20-1169
5. Tsai T, Jacobson B, Jha A. American hospital capacity and projected need for COVID-19 patient care. Health Affairs blog. March 17, 2020. Accessed October 12, 2020. https://www.healthaffairs.org/do/10.1377/hblog20200317.457910/full/
1. Nemer D, Wilner BR, Burkle A, et al. Clinical characteristics and outcomes of non-ICU hospitalization for COVID-19 in a nonepicenter, centrally monitored healthcare system. J Hosp Med. 2021;16:7-14. https://doi.org/10.12788/jhm.3510
2. Lippi G, Pleban M, Henry B. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin Chim Acta. 2020;506:145-148. https://doi.org/10.1016/j.cca.2020.03.022
3. Klok FA, Kruip MJHA, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145-147. https://doi.org/10.1016/j.thromres.2020.04.013
4. Giannakeas V, Bhatia D, Warkentin M, et al. Estimating the maximum capacity of COVID-19 cases manageable per day given a health care system’s constrained resources. Ann Intern Med. April 16, 2020. https://doi.org/10.7326/M20-1169
5. Tsai T, Jacobson B, Jha A. American hospital capacity and projected need for COVID-19 patient care. Health Affairs blog. March 17, 2020. Accessed October 12, 2020. https://www.healthaffairs.org/do/10.1377/hblog20200317.457910/full/
© 2021 Society of Hospital Medicine
Telephone: 608-262-2434 Email: [email protected]; Twitter: @kaiksow.
Leadership & Professional Development: From Seed to Fruit—How to Get Your Academic Project Across the Finish Line
“Our goals can only be reached through the vehicle of a plan. There is no other route to success.”
—Pablo Picasso
Whether it be a research manuscript, quality improvement (QI) initiative, or educational curriculum, busy clinicians often struggle getting projects past the idea stage. Barriers to completion, such as a busy clinical schedule or lack of experience and mentorship, are well known. Importantly, these projects serve as “academic currency” used for promotion and advancement and also create generalizable knowledge, which can help others improve clinical practice or operational processes. Those who are successful in completing their academic project frequently follow a well-structured path. Consider the following principles to get your idea across the finish line:
Find a blueprint. Among most academic projects, whether a research paper, QI project or new curriculum, an underlying formula is commonly applied. Before starting, do your background research. Is there a paper or method that resembles your desired approach? Is there a question or concept that caught your eye? Using a blueprint from existing evidence allows you to identify important structures, phrases, and terms to inform your manuscript. Once you have identified the blueprint, define your project and approach.
Find a mentor. While career mentorship is important for professional growth, you first need a project mentor. Being a project mentor is a smaller ask for a more senior colleague than being a career mentor, and it’s a great way to test-drive a potential long-term working relationship. Moreover, the successful completion of one project can potentially lead to further opportunities, and perhaps even a long-term career mentor.
Take initiative. In business, there is a common adage: “Never bring a problem to your boss without a proposed solution in hand.”1 In academics, consider: “Never show up with an idea without bringing a proposal.” By bringing a defined proposal to the conversation, your inquiry is more likely to get a response because (a) it is not a blind-ask and (b) it creates a foundation to build on. This is analogous to an early learner presenting their assessment and plan in the clinical setting; you don’t stop at the diagnosis (your idea) without having a plan for how you want to manage it.
Get an accountability partner. Publicly committing to a goal increases the probability of accomplishing your task by 65%, while having an accountability partner increases that by 95%.2 An accountability partner serves as a coach to help you accomplish a task. This individual can be a colleague, spouse, or friend and is typically not a part of the project. By leveraging peer pressure, you increase the odds of successfully completing your project.
Carve out dedicated time. The entrepreneur and author Jim Rohn once said, “Discipline is the bridge between goals and accomplishments.”3 To complete a project, you have to make the time to do the work. While many believe that successful writers sit and write for hours on end, many famous writers only wrote for a few hours at a time—but they did so consistently.4 Create your routine by setting aside consistent, defined time to work on your project. To extract the most value, select a time of the day in which you work best (eg, early morning). Then, set a timer for 30 minutes and write—or work.
Because you are a busy clinician with constant demands on your time, having the skillset to reliably turn an idea into “academic currency” is a necessity. Having a plan and following these principles will help you earn that academic coin.
1. Gallo A. The right way to bring a problem to your boss. Harvard Business Review. December 5, 2014. Accessed April 11, 2020. https://hbr.org/2014/12/the-right-way-to-bring-a-problem-to-your-boss
2. Hardy B. Accountability partners are great. But “success” partners will change your life. May 14, 2019. Accessed April 11, 2020. Medium. https://medium.com/@benjaminhardy/accountability-partners-are-great-but-...
3. Rohn J. 10 unforgettable quotes by Jim Rohn. Accessed June 20, 2020. https://www.success.com/10-unforgettable-quotes-by-jim-rohn/
4. Clear J. Atomic Habits: An Easy & Proven Way to Build Good Habits & Break Bad Ones. Avery; 2018. https://jamesclear.com/atomic-habits
“Our goals can only be reached through the vehicle of a plan. There is no other route to success.”
—Pablo Picasso
Whether it be a research manuscript, quality improvement (QI) initiative, or educational curriculum, busy clinicians often struggle getting projects past the idea stage. Barriers to completion, such as a busy clinical schedule or lack of experience and mentorship, are well known. Importantly, these projects serve as “academic currency” used for promotion and advancement and also create generalizable knowledge, which can help others improve clinical practice or operational processes. Those who are successful in completing their academic project frequently follow a well-structured path. Consider the following principles to get your idea across the finish line:
Find a blueprint. Among most academic projects, whether a research paper, QI project or new curriculum, an underlying formula is commonly applied. Before starting, do your background research. Is there a paper or method that resembles your desired approach? Is there a question or concept that caught your eye? Using a blueprint from existing evidence allows you to identify important structures, phrases, and terms to inform your manuscript. Once you have identified the blueprint, define your project and approach.
Find a mentor. While career mentorship is important for professional growth, you first need a project mentor. Being a project mentor is a smaller ask for a more senior colleague than being a career mentor, and it’s a great way to test-drive a potential long-term working relationship. Moreover, the successful completion of one project can potentially lead to further opportunities, and perhaps even a long-term career mentor.
Take initiative. In business, there is a common adage: “Never bring a problem to your boss without a proposed solution in hand.”1 In academics, consider: “Never show up with an idea without bringing a proposal.” By bringing a defined proposal to the conversation, your inquiry is more likely to get a response because (a) it is not a blind-ask and (b) it creates a foundation to build on. This is analogous to an early learner presenting their assessment and plan in the clinical setting; you don’t stop at the diagnosis (your idea) without having a plan for how you want to manage it.
Get an accountability partner. Publicly committing to a goal increases the probability of accomplishing your task by 65%, while having an accountability partner increases that by 95%.2 An accountability partner serves as a coach to help you accomplish a task. This individual can be a colleague, spouse, or friend and is typically not a part of the project. By leveraging peer pressure, you increase the odds of successfully completing your project.
Carve out dedicated time. The entrepreneur and author Jim Rohn once said, “Discipline is the bridge between goals and accomplishments.”3 To complete a project, you have to make the time to do the work. While many believe that successful writers sit and write for hours on end, many famous writers only wrote for a few hours at a time—but they did so consistently.4 Create your routine by setting aside consistent, defined time to work on your project. To extract the most value, select a time of the day in which you work best (eg, early morning). Then, set a timer for 30 minutes and write—or work.
Because you are a busy clinician with constant demands on your time, having the skillset to reliably turn an idea into “academic currency” is a necessity. Having a plan and following these principles will help you earn that academic coin.
“Our goals can only be reached through the vehicle of a plan. There is no other route to success.”
—Pablo Picasso
Whether it be a research manuscript, quality improvement (QI) initiative, or educational curriculum, busy clinicians often struggle getting projects past the idea stage. Barriers to completion, such as a busy clinical schedule or lack of experience and mentorship, are well known. Importantly, these projects serve as “academic currency” used for promotion and advancement and also create generalizable knowledge, which can help others improve clinical practice or operational processes. Those who are successful in completing their academic project frequently follow a well-structured path. Consider the following principles to get your idea across the finish line:
Find a blueprint. Among most academic projects, whether a research paper, QI project or new curriculum, an underlying formula is commonly applied. Before starting, do your background research. Is there a paper or method that resembles your desired approach? Is there a question or concept that caught your eye? Using a blueprint from existing evidence allows you to identify important structures, phrases, and terms to inform your manuscript. Once you have identified the blueprint, define your project and approach.
Find a mentor. While career mentorship is important for professional growth, you first need a project mentor. Being a project mentor is a smaller ask for a more senior colleague than being a career mentor, and it’s a great way to test-drive a potential long-term working relationship. Moreover, the successful completion of one project can potentially lead to further opportunities, and perhaps even a long-term career mentor.
Take initiative. In business, there is a common adage: “Never bring a problem to your boss without a proposed solution in hand.”1 In academics, consider: “Never show up with an idea without bringing a proposal.” By bringing a defined proposal to the conversation, your inquiry is more likely to get a response because (a) it is not a blind-ask and (b) it creates a foundation to build on. This is analogous to an early learner presenting their assessment and plan in the clinical setting; you don’t stop at the diagnosis (your idea) without having a plan for how you want to manage it.
Get an accountability partner. Publicly committing to a goal increases the probability of accomplishing your task by 65%, while having an accountability partner increases that by 95%.2 An accountability partner serves as a coach to help you accomplish a task. This individual can be a colleague, spouse, or friend and is typically not a part of the project. By leveraging peer pressure, you increase the odds of successfully completing your project.
Carve out dedicated time. The entrepreneur and author Jim Rohn once said, “Discipline is the bridge between goals and accomplishments.”3 To complete a project, you have to make the time to do the work. While many believe that successful writers sit and write for hours on end, many famous writers only wrote for a few hours at a time—but they did so consistently.4 Create your routine by setting aside consistent, defined time to work on your project. To extract the most value, select a time of the day in which you work best (eg, early morning). Then, set a timer for 30 minutes and write—or work.
Because you are a busy clinician with constant demands on your time, having the skillset to reliably turn an idea into “academic currency” is a necessity. Having a plan and following these principles will help you earn that academic coin.
1. Gallo A. The right way to bring a problem to your boss. Harvard Business Review. December 5, 2014. Accessed April 11, 2020. https://hbr.org/2014/12/the-right-way-to-bring-a-problem-to-your-boss
2. Hardy B. Accountability partners are great. But “success” partners will change your life. May 14, 2019. Accessed April 11, 2020. Medium. https://medium.com/@benjaminhardy/accountability-partners-are-great-but-...
3. Rohn J. 10 unforgettable quotes by Jim Rohn. Accessed June 20, 2020. https://www.success.com/10-unforgettable-quotes-by-jim-rohn/
4. Clear J. Atomic Habits: An Easy & Proven Way to Build Good Habits & Break Bad Ones. Avery; 2018. https://jamesclear.com/atomic-habits
1. Gallo A. The right way to bring a problem to your boss. Harvard Business Review. December 5, 2014. Accessed April 11, 2020. https://hbr.org/2014/12/the-right-way-to-bring-a-problem-to-your-boss
2. Hardy B. Accountability partners are great. But “success” partners will change your life. May 14, 2019. Accessed April 11, 2020. Medium. https://medium.com/@benjaminhardy/accountability-partners-are-great-but-...
3. Rohn J. 10 unforgettable quotes by Jim Rohn. Accessed June 20, 2020. https://www.success.com/10-unforgettable-quotes-by-jim-rohn/
4. Clear J. Atomic Habits: An Easy & Proven Way to Build Good Habits & Break Bad Ones. Avery; 2018. https://jamesclear.com/atomic-habits
© 2021 Society of Hospital Medicine
Email: [email protected]; Telephone: 415-221-4810 x22084; Twitter: @sharminzi.
The Light at the End of the Tunnel: Reflections on 2020 and Hopes for 2021
We enter the new year still in the midst of the coronavirus disease 2019 (COVID-19) pandemic and remain humbled by its impact. It is remarkable how much, and how little, has changed. Hospitalists in the early days of the COVID-19 pandemic were struggling. We were caring for patients who were suffering and dying from a new and mysterious disease. There weren’t enough tests (or, if there were tests, there weren’t swabs).1 We were using protocols for managing respiratory failure that, we would learn later, may not have been the best for improving outcomes. Rumors of unproven therapies came from everywhere: our patients, our colleagues, and even the highest realms of the federal government. We also knew very little about how best to protect ourselves. In many cases, we did not have enough personal protective equipment (PPE). There were no face shields, or “zoom rounds,” or even awareness that we probably shouldn’t sit in the tiny conference room (maskless) discussing patients with the large team of doctors, nurses, respiratory therapists, and social workers.
Perhaps worst of all, we were haunted. We were alarmed by the large numbers of young patients who were ill, and our elderly patients, many of whom we knew and had cared for many times, had suddenly just stopped showing up.2 In our free moments, we worried about them; maybe they were afraid to come to the hospital, maybe they were home sick with COVID-19, or maybe they had died alone. And children, initially thought to be spared the most serious consequences of COVID-19, started coming to the hospital with a rare but severe new COVID-19-associated complication, termed multisystem inflammatory syndrome in children (MIS-C). We had to learn to manage yet another manifestation of COVID-19, largely through trial and error.
And, of course, clinical care was only one of our many responsibilities. We were also busy hunting for ventilators, setting up makeshift medical wards and intensive care units, revamping medical education, and scouring the literature for any information to help guide patient care. We worried about getting sick ourselves and bringing the disease home to our families. Our impatience grew as day after day there was no (and still is no) coordinated federal response.
A glimmer of hope slowly emerged. Our colleagues designed and rapidly evaluated respiratory protocols and provided early evidence about the strategies (eg, proning) that were associated with improved outcomes.3 Researchers began to generate knowledge and move us beyond rumors regarding potential therapies. We cheered as our administrators concocted unusual strategies to remedy the PPE and testing shortages.4
At the Journal of Hospital Medicine, we were faced with another challenge: How would we describe the chaos and the challenges of being a physician during the COVID-19 era? How would we document the way our colleagues were rising to the challenge and identifying opportunities to rethink hospital care in the United States?
In April, we began to receive a deluge of personal essays from frontline physicians about their experiences with COVID-19. Generally, medical journals publish and disseminate original, high-impact research. Personal essays rarely fit this model. Given the unprecedented circumstances, however, we decided these essays could help chronicle an important moment in medical history. In our May 2020 issue, we published only these essays. We continue to publish them online almost daily.
Some of the essays described how the healthcare system—previously thought to be hyperspecialized, profit-driven, and resistant to change—pivoted within days, as hospitalist physicians trained other physicians to “unspecialize” and pediatricians began to care for adults in an otherwise overwhelmed hospital system.5,6 Another essay focused on the need to trust that medical students who had graduated early would be able to function as physicians.7 And yet another essay expressed concern about the widespread use of unproven therapies in hospitalized patients. “Even in times of global pandemic, we need to consider potential harms and adverse consequences of novel treatments,’’ the physicians wrote. “Sometimes inaction is preferable to action.”8
Several essays reflected on the impact of the pandemic on healthcare disparities, suggesting that the pandemic had made (the well-known but often ignored) differences in health outcomes between White patients and racial minorities more obvious. Still another essay reflected on the intersection between structural racism, poor access to care, and interpersonal racism, describing the grief caused by losses of Black lives to both police violence and COVID-19.9
There also were personal stories of hardship and survival. One hospitalist physician with asthma described coughing as ``the new leprosy.”10 She wrote, “This is a particularly unpropitious time in history to be a Chinese-American doctor who can’t stop coughing.”
There were drawbacks to our decision to focus on personal essays. Although it was clear even before the pandemic, COVID-19 has highlighted that a path for quick dissemination of original peer-reviewed research is needed. If existing medical journals do not fill that role, websites that publish and disseminate non–peer-reviewed work (aka, “preprints”) will become the preferred method for distribution of high-impact, timely original research.11 The journal’s pivot to reviewing and publishing personal essays may have kept us from improving our approach to rapid peer review and dissemination. In those early days, however, there was no peer-reviewed work to publish, but there was an intense desire (from our members and physicians generally) for information and stories from the front lines. In a way, the essays we published were early “case reports,” that hypothesized about how we might rethink healthcare delivery in pandemic conditions.
Furthermore, our decision to solicit and publish personal essays addressing shortcomings of the federal response and consequences of the pandemic meant that the Journal of Hospital Medicine became part of the pandemic’s political discourse. As editors, we have historically kept the journal away from political arguments or endorsements. In this case, however, we decided that even if some of the opinions were political, they were an appropriate response to the widespread anti-science rhetoric endorsed by the current administration. The resultant erosion of trust in public health has undoubtedly contributed to persistence of the pandemic.12 A stance against masks, for example, rejects the recommendations of nearly all scientists in favor of (a selfish and problematic idea of) “self-determination.” Those who proclaim that such a mandate infringes on their personal freedom reject evidence-based recommendations of scientists and disregard public health strategies meant to protect everyone.
As we reflect on the past year, our most important lesson may be that our previous emphasis on publishing high-impact original research likely missed important personal and professional insights, insights that could have changed practice, improved patient experience, and reduced physician burnout. Anecdotes are not scientific evidence, but we have discovered their incredible power to help us learn, empathize, commiserate, and survive. Hospitals learned that they must adapt in the moment, a notion that runs counter to the notoriously slow pace of change in paradigms of healthcare. Hospitalists learned to “find their battle buddies” to ward off isolation and to cherish their teams in the face of overwhelming trauma, an approach requiring empathy, humility, and compassion.13 We won’t soon forget that, when things were most dire, it was stories—not data—that gave us hope. We look forward to 2021 with great optimism. New vaccines and new federal leaders who value and respect science give us hope that the end of the pandemic is in sight. We are indebted to all frontline workers who have transformed care delivery and remained courageous in the face of great personal risk. As a journal, we will continue, as one scientist noted, to use our “platform for advocacy, unabashedly.”14
1. Shuren J, Stenzel T. Covid-19 molecular diagnostic testing - lessons learned. N Engl J Med. 2020;383:e97. https://doi.org/10.1056/NEJMp2023830
2. Rosenbaum L. The untold toll - the pandemic’s effects on patients without Covid-19. N Engl J Med. 2020;382:2368-2371. https://doi.org/10.1056/NEJMms2009984
3. Westafer LM, Elia T, Medarametla V, Lagu T. A transdisciplinary COVID-19 early respiratory intervention protocol: an implementation story. J Hosp Med. 2020;15:372-374. https://doi.org/10.12788/jhm.3456
4. Lagu T, Artenstein AW, Werner RM. Fool me twice: the role for hospitals and health systems in fixing the broken PPE supply chain. J Hosp Med. 2020;15:570-571. https://doi.org/10.12788/jhm.3489
5. Cram P, Anderson ML, Shaughnessy EE. All hands on deck: learning to “un-specialize” in the COVID-19 pandemic. J Hosp Med. 2020;15:314-315. https://doi.org/10.12788/jhm.3426
6. Biala D, Siegel EJ, Silver L, Schindel B, Smith KM. Deployed: pediatric residents caring for adults during COVID-19’s first wave in New York City. J Hosp Med. 2020; Published ahead of print. https://doi.org/10.12788/jhm.3527
7. Kinnear B, Kelleher M, Olson AP, Sall D, Schumacher DJ. Developing trust with early medical school graduates during the COVID-19 pandemic. J Hosp Med. 2020;15:367-369. https://doi.org/10.12788/jhm.3463
8. Canfield GS, Schultz JS, Windham S, et al. Empiric therapies for covid-19: destined to fail by ignoring the lessons of history. J Hosp Med. 2020;15:434-436. https://doi.org/10.12788/jhm.3469
9. Manning KD. When grief and crises intersect: perspectives of a Black physician in the time of two pandemics. J Hosp Med. 2020;15:566-567. https://doi.org/10.12788/jhm.3481
10. Chang T. Do I have coronavirus? J Hosp Med. 2020;15:277-278. https://doi.org/10.12788/jhm.3430
11. Guterman EL, Braunstein LZ. Preprints during the COVID-19 pandemic: public health emergencies and medical literature. J Hosp Med. 2020;15:634-636. https://doi.org/10.12788/jhm.3491
12. Udow-Phillips M, Lantz PM. Trust in public health is essential amid the COVID-19 pandemic. J Hosp Med. 2020;15:431-433. https://doi.org/10.12788/jhm.3474
13. Hertling M. Ten tips for a crisis: lessons from a soldier. J Hosp Med. 2020;15:275-276. https://doi.org/10.12788/jhm.3424
14. O’Glasser A [@aoglasser]. #JHMChat I also need to readily admit that part of the reason I’m a loyal, enthusiastic @JHospMedicine reader is because [Tweet]. November 16, 2020. Accessed November 28, 2020. https://twitter.com/aoglasser/status/1328529564595720192
We enter the new year still in the midst of the coronavirus disease 2019 (COVID-19) pandemic and remain humbled by its impact. It is remarkable how much, and how little, has changed. Hospitalists in the early days of the COVID-19 pandemic were struggling. We were caring for patients who were suffering and dying from a new and mysterious disease. There weren’t enough tests (or, if there were tests, there weren’t swabs).1 We were using protocols for managing respiratory failure that, we would learn later, may not have been the best for improving outcomes. Rumors of unproven therapies came from everywhere: our patients, our colleagues, and even the highest realms of the federal government. We also knew very little about how best to protect ourselves. In many cases, we did not have enough personal protective equipment (PPE). There were no face shields, or “zoom rounds,” or even awareness that we probably shouldn’t sit in the tiny conference room (maskless) discussing patients with the large team of doctors, nurses, respiratory therapists, and social workers.
Perhaps worst of all, we were haunted. We were alarmed by the large numbers of young patients who were ill, and our elderly patients, many of whom we knew and had cared for many times, had suddenly just stopped showing up.2 In our free moments, we worried about them; maybe they were afraid to come to the hospital, maybe they were home sick with COVID-19, or maybe they had died alone. And children, initially thought to be spared the most serious consequences of COVID-19, started coming to the hospital with a rare but severe new COVID-19-associated complication, termed multisystem inflammatory syndrome in children (MIS-C). We had to learn to manage yet another manifestation of COVID-19, largely through trial and error.
And, of course, clinical care was only one of our many responsibilities. We were also busy hunting for ventilators, setting up makeshift medical wards and intensive care units, revamping medical education, and scouring the literature for any information to help guide patient care. We worried about getting sick ourselves and bringing the disease home to our families. Our impatience grew as day after day there was no (and still is no) coordinated federal response.
A glimmer of hope slowly emerged. Our colleagues designed and rapidly evaluated respiratory protocols and provided early evidence about the strategies (eg, proning) that were associated with improved outcomes.3 Researchers began to generate knowledge and move us beyond rumors regarding potential therapies. We cheered as our administrators concocted unusual strategies to remedy the PPE and testing shortages.4
At the Journal of Hospital Medicine, we were faced with another challenge: How would we describe the chaos and the challenges of being a physician during the COVID-19 era? How would we document the way our colleagues were rising to the challenge and identifying opportunities to rethink hospital care in the United States?
In April, we began to receive a deluge of personal essays from frontline physicians about their experiences with COVID-19. Generally, medical journals publish and disseminate original, high-impact research. Personal essays rarely fit this model. Given the unprecedented circumstances, however, we decided these essays could help chronicle an important moment in medical history. In our May 2020 issue, we published only these essays. We continue to publish them online almost daily.
Some of the essays described how the healthcare system—previously thought to be hyperspecialized, profit-driven, and resistant to change—pivoted within days, as hospitalist physicians trained other physicians to “unspecialize” and pediatricians began to care for adults in an otherwise overwhelmed hospital system.5,6 Another essay focused on the need to trust that medical students who had graduated early would be able to function as physicians.7 And yet another essay expressed concern about the widespread use of unproven therapies in hospitalized patients. “Even in times of global pandemic, we need to consider potential harms and adverse consequences of novel treatments,’’ the physicians wrote. “Sometimes inaction is preferable to action.”8
Several essays reflected on the impact of the pandemic on healthcare disparities, suggesting that the pandemic had made (the well-known but often ignored) differences in health outcomes between White patients and racial minorities more obvious. Still another essay reflected on the intersection between structural racism, poor access to care, and interpersonal racism, describing the grief caused by losses of Black lives to both police violence and COVID-19.9
There also were personal stories of hardship and survival. One hospitalist physician with asthma described coughing as ``the new leprosy.”10 She wrote, “This is a particularly unpropitious time in history to be a Chinese-American doctor who can’t stop coughing.”
There were drawbacks to our decision to focus on personal essays. Although it was clear even before the pandemic, COVID-19 has highlighted that a path for quick dissemination of original peer-reviewed research is needed. If existing medical journals do not fill that role, websites that publish and disseminate non–peer-reviewed work (aka, “preprints”) will become the preferred method for distribution of high-impact, timely original research.11 The journal’s pivot to reviewing and publishing personal essays may have kept us from improving our approach to rapid peer review and dissemination. In those early days, however, there was no peer-reviewed work to publish, but there was an intense desire (from our members and physicians generally) for information and stories from the front lines. In a way, the essays we published were early “case reports,” that hypothesized about how we might rethink healthcare delivery in pandemic conditions.
Furthermore, our decision to solicit and publish personal essays addressing shortcomings of the federal response and consequences of the pandemic meant that the Journal of Hospital Medicine became part of the pandemic’s political discourse. As editors, we have historically kept the journal away from political arguments or endorsements. In this case, however, we decided that even if some of the opinions were political, they were an appropriate response to the widespread anti-science rhetoric endorsed by the current administration. The resultant erosion of trust in public health has undoubtedly contributed to persistence of the pandemic.12 A stance against masks, for example, rejects the recommendations of nearly all scientists in favor of (a selfish and problematic idea of) “self-determination.” Those who proclaim that such a mandate infringes on their personal freedom reject evidence-based recommendations of scientists and disregard public health strategies meant to protect everyone.
As we reflect on the past year, our most important lesson may be that our previous emphasis on publishing high-impact original research likely missed important personal and professional insights, insights that could have changed practice, improved patient experience, and reduced physician burnout. Anecdotes are not scientific evidence, but we have discovered their incredible power to help us learn, empathize, commiserate, and survive. Hospitals learned that they must adapt in the moment, a notion that runs counter to the notoriously slow pace of change in paradigms of healthcare. Hospitalists learned to “find their battle buddies” to ward off isolation and to cherish their teams in the face of overwhelming trauma, an approach requiring empathy, humility, and compassion.13 We won’t soon forget that, when things were most dire, it was stories—not data—that gave us hope. We look forward to 2021 with great optimism. New vaccines and new federal leaders who value and respect science give us hope that the end of the pandemic is in sight. We are indebted to all frontline workers who have transformed care delivery and remained courageous in the face of great personal risk. As a journal, we will continue, as one scientist noted, to use our “platform for advocacy, unabashedly.”14
We enter the new year still in the midst of the coronavirus disease 2019 (COVID-19) pandemic and remain humbled by its impact. It is remarkable how much, and how little, has changed. Hospitalists in the early days of the COVID-19 pandemic were struggling. We were caring for patients who were suffering and dying from a new and mysterious disease. There weren’t enough tests (or, if there were tests, there weren’t swabs).1 We were using protocols for managing respiratory failure that, we would learn later, may not have been the best for improving outcomes. Rumors of unproven therapies came from everywhere: our patients, our colleagues, and even the highest realms of the federal government. We also knew very little about how best to protect ourselves. In many cases, we did not have enough personal protective equipment (PPE). There were no face shields, or “zoom rounds,” or even awareness that we probably shouldn’t sit in the tiny conference room (maskless) discussing patients with the large team of doctors, nurses, respiratory therapists, and social workers.
Perhaps worst of all, we were haunted. We were alarmed by the large numbers of young patients who were ill, and our elderly patients, many of whom we knew and had cared for many times, had suddenly just stopped showing up.2 In our free moments, we worried about them; maybe they were afraid to come to the hospital, maybe they were home sick with COVID-19, or maybe they had died alone. And children, initially thought to be spared the most serious consequences of COVID-19, started coming to the hospital with a rare but severe new COVID-19-associated complication, termed multisystem inflammatory syndrome in children (MIS-C). We had to learn to manage yet another manifestation of COVID-19, largely through trial and error.
And, of course, clinical care was only one of our many responsibilities. We were also busy hunting for ventilators, setting up makeshift medical wards and intensive care units, revamping medical education, and scouring the literature for any information to help guide patient care. We worried about getting sick ourselves and bringing the disease home to our families. Our impatience grew as day after day there was no (and still is no) coordinated federal response.
A glimmer of hope slowly emerged. Our colleagues designed and rapidly evaluated respiratory protocols and provided early evidence about the strategies (eg, proning) that were associated with improved outcomes.3 Researchers began to generate knowledge and move us beyond rumors regarding potential therapies. We cheered as our administrators concocted unusual strategies to remedy the PPE and testing shortages.4
At the Journal of Hospital Medicine, we were faced with another challenge: How would we describe the chaos and the challenges of being a physician during the COVID-19 era? How would we document the way our colleagues were rising to the challenge and identifying opportunities to rethink hospital care in the United States?
In April, we began to receive a deluge of personal essays from frontline physicians about their experiences with COVID-19. Generally, medical journals publish and disseminate original, high-impact research. Personal essays rarely fit this model. Given the unprecedented circumstances, however, we decided these essays could help chronicle an important moment in medical history. In our May 2020 issue, we published only these essays. We continue to publish them online almost daily.
Some of the essays described how the healthcare system—previously thought to be hyperspecialized, profit-driven, and resistant to change—pivoted within days, as hospitalist physicians trained other physicians to “unspecialize” and pediatricians began to care for adults in an otherwise overwhelmed hospital system.5,6 Another essay focused on the need to trust that medical students who had graduated early would be able to function as physicians.7 And yet another essay expressed concern about the widespread use of unproven therapies in hospitalized patients. “Even in times of global pandemic, we need to consider potential harms and adverse consequences of novel treatments,’’ the physicians wrote. “Sometimes inaction is preferable to action.”8
Several essays reflected on the impact of the pandemic on healthcare disparities, suggesting that the pandemic had made (the well-known but often ignored) differences in health outcomes between White patients and racial minorities more obvious. Still another essay reflected on the intersection between structural racism, poor access to care, and interpersonal racism, describing the grief caused by losses of Black lives to both police violence and COVID-19.9
There also were personal stories of hardship and survival. One hospitalist physician with asthma described coughing as ``the new leprosy.”10 She wrote, “This is a particularly unpropitious time in history to be a Chinese-American doctor who can’t stop coughing.”
There were drawbacks to our decision to focus on personal essays. Although it was clear even before the pandemic, COVID-19 has highlighted that a path for quick dissemination of original peer-reviewed research is needed. If existing medical journals do not fill that role, websites that publish and disseminate non–peer-reviewed work (aka, “preprints”) will become the preferred method for distribution of high-impact, timely original research.11 The journal’s pivot to reviewing and publishing personal essays may have kept us from improving our approach to rapid peer review and dissemination. In those early days, however, there was no peer-reviewed work to publish, but there was an intense desire (from our members and physicians generally) for information and stories from the front lines. In a way, the essays we published were early “case reports,” that hypothesized about how we might rethink healthcare delivery in pandemic conditions.
Furthermore, our decision to solicit and publish personal essays addressing shortcomings of the federal response and consequences of the pandemic meant that the Journal of Hospital Medicine became part of the pandemic’s political discourse. As editors, we have historically kept the journal away from political arguments or endorsements. In this case, however, we decided that even if some of the opinions were political, they were an appropriate response to the widespread anti-science rhetoric endorsed by the current administration. The resultant erosion of trust in public health has undoubtedly contributed to persistence of the pandemic.12 A stance against masks, for example, rejects the recommendations of nearly all scientists in favor of (a selfish and problematic idea of) “self-determination.” Those who proclaim that such a mandate infringes on their personal freedom reject evidence-based recommendations of scientists and disregard public health strategies meant to protect everyone.
As we reflect on the past year, our most important lesson may be that our previous emphasis on publishing high-impact original research likely missed important personal and professional insights, insights that could have changed practice, improved patient experience, and reduced physician burnout. Anecdotes are not scientific evidence, but we have discovered their incredible power to help us learn, empathize, commiserate, and survive. Hospitals learned that they must adapt in the moment, a notion that runs counter to the notoriously slow pace of change in paradigms of healthcare. Hospitalists learned to “find their battle buddies” to ward off isolation and to cherish their teams in the face of overwhelming trauma, an approach requiring empathy, humility, and compassion.13 We won’t soon forget that, when things were most dire, it was stories—not data—that gave us hope. We look forward to 2021 with great optimism. New vaccines and new federal leaders who value and respect science give us hope that the end of the pandemic is in sight. We are indebted to all frontline workers who have transformed care delivery and remained courageous in the face of great personal risk. As a journal, we will continue, as one scientist noted, to use our “platform for advocacy, unabashedly.”14
1. Shuren J, Stenzel T. Covid-19 molecular diagnostic testing - lessons learned. N Engl J Med. 2020;383:e97. https://doi.org/10.1056/NEJMp2023830
2. Rosenbaum L. The untold toll - the pandemic’s effects on patients without Covid-19. N Engl J Med. 2020;382:2368-2371. https://doi.org/10.1056/NEJMms2009984
3. Westafer LM, Elia T, Medarametla V, Lagu T. A transdisciplinary COVID-19 early respiratory intervention protocol: an implementation story. J Hosp Med. 2020;15:372-374. https://doi.org/10.12788/jhm.3456
4. Lagu T, Artenstein AW, Werner RM. Fool me twice: the role for hospitals and health systems in fixing the broken PPE supply chain. J Hosp Med. 2020;15:570-571. https://doi.org/10.12788/jhm.3489
5. Cram P, Anderson ML, Shaughnessy EE. All hands on deck: learning to “un-specialize” in the COVID-19 pandemic. J Hosp Med. 2020;15:314-315. https://doi.org/10.12788/jhm.3426
6. Biala D, Siegel EJ, Silver L, Schindel B, Smith KM. Deployed: pediatric residents caring for adults during COVID-19’s first wave in New York City. J Hosp Med. 2020; Published ahead of print. https://doi.org/10.12788/jhm.3527
7. Kinnear B, Kelleher M, Olson AP, Sall D, Schumacher DJ. Developing trust with early medical school graduates during the COVID-19 pandemic. J Hosp Med. 2020;15:367-369. https://doi.org/10.12788/jhm.3463
8. Canfield GS, Schultz JS, Windham S, et al. Empiric therapies for covid-19: destined to fail by ignoring the lessons of history. J Hosp Med. 2020;15:434-436. https://doi.org/10.12788/jhm.3469
9. Manning KD. When grief and crises intersect: perspectives of a Black physician in the time of two pandemics. J Hosp Med. 2020;15:566-567. https://doi.org/10.12788/jhm.3481
10. Chang T. Do I have coronavirus? J Hosp Med. 2020;15:277-278. https://doi.org/10.12788/jhm.3430
11. Guterman EL, Braunstein LZ. Preprints during the COVID-19 pandemic: public health emergencies and medical literature. J Hosp Med. 2020;15:634-636. https://doi.org/10.12788/jhm.3491
12. Udow-Phillips M, Lantz PM. Trust in public health is essential amid the COVID-19 pandemic. J Hosp Med. 2020;15:431-433. https://doi.org/10.12788/jhm.3474
13. Hertling M. Ten tips for a crisis: lessons from a soldier. J Hosp Med. 2020;15:275-276. https://doi.org/10.12788/jhm.3424
14. O’Glasser A [@aoglasser]. #JHMChat I also need to readily admit that part of the reason I’m a loyal, enthusiastic @JHospMedicine reader is because [Tweet]. November 16, 2020. Accessed November 28, 2020. https://twitter.com/aoglasser/status/1328529564595720192
1. Shuren J, Stenzel T. Covid-19 molecular diagnostic testing - lessons learned. N Engl J Med. 2020;383:e97. https://doi.org/10.1056/NEJMp2023830
2. Rosenbaum L. The untold toll - the pandemic’s effects on patients without Covid-19. N Engl J Med. 2020;382:2368-2371. https://doi.org/10.1056/NEJMms2009984
3. Westafer LM, Elia T, Medarametla V, Lagu T. A transdisciplinary COVID-19 early respiratory intervention protocol: an implementation story. J Hosp Med. 2020;15:372-374. https://doi.org/10.12788/jhm.3456
4. Lagu T, Artenstein AW, Werner RM. Fool me twice: the role for hospitals and health systems in fixing the broken PPE supply chain. J Hosp Med. 2020;15:570-571. https://doi.org/10.12788/jhm.3489
5. Cram P, Anderson ML, Shaughnessy EE. All hands on deck: learning to “un-specialize” in the COVID-19 pandemic. J Hosp Med. 2020;15:314-315. https://doi.org/10.12788/jhm.3426
6. Biala D, Siegel EJ, Silver L, Schindel B, Smith KM. Deployed: pediatric residents caring for adults during COVID-19’s first wave in New York City. J Hosp Med. 2020; Published ahead of print. https://doi.org/10.12788/jhm.3527
7. Kinnear B, Kelleher M, Olson AP, Sall D, Schumacher DJ. Developing trust with early medical school graduates during the COVID-19 pandemic. J Hosp Med. 2020;15:367-369. https://doi.org/10.12788/jhm.3463
8. Canfield GS, Schultz JS, Windham S, et al. Empiric therapies for covid-19: destined to fail by ignoring the lessons of history. J Hosp Med. 2020;15:434-436. https://doi.org/10.12788/jhm.3469
9. Manning KD. When grief and crises intersect: perspectives of a Black physician in the time of two pandemics. J Hosp Med. 2020;15:566-567. https://doi.org/10.12788/jhm.3481
10. Chang T. Do I have coronavirus? J Hosp Med. 2020;15:277-278. https://doi.org/10.12788/jhm.3430
11. Guterman EL, Braunstein LZ. Preprints during the COVID-19 pandemic: public health emergencies and medical literature. J Hosp Med. 2020;15:634-636. https://doi.org/10.12788/jhm.3491
12. Udow-Phillips M, Lantz PM. Trust in public health is essential amid the COVID-19 pandemic. J Hosp Med. 2020;15:431-433. https://doi.org/10.12788/jhm.3474
13. Hertling M. Ten tips for a crisis: lessons from a soldier. J Hosp Med. 2020;15:275-276. https://doi.org/10.12788/jhm.3424
14. O’Glasser A [@aoglasser]. #JHMChat I also need to readily admit that part of the reason I’m a loyal, enthusiastic @JHospMedicine reader is because [Tweet]. November 16, 2020. Accessed November 28, 2020. https://twitter.com/aoglasser/status/1328529564595720192
© 2021 Society of Hospital Medicine
Email: [email protected]; Telephone: 513-636-6222; Twitter: @SamirShahMD.
Defining a New Normal While Awaiting the Pandemic’s Next Wave
Hospitalists have played a central role in the massive response to the coronavirus disease 2019 (COVID-19) pandemic by creating innovative staffing models, rapidly learning about the disease and teaching others, and working closely with hospital executive leadership to create surge capacity.1 Some hospitals and regions have weathered an initial storm and are now experiencing a slower influx of COVID-19 patients, while others are now seeing a surge, which is expected to persist for the foreseeable future—the marathon has begun.2 We have entered a new COVID-19 reality: disrupted care models, harsh financial consequences,3 and uncertainty about which adaptations should be preserved and for how long. Common operational challenges will define the new normal. In this Perspective, we share strategies to address these challenges, focusing on three emerging themes: realigning staffing to patient volumes, safely managing space limitations, and navigating the financial ramifications of COVID-19 for hospital medicine groups.
BALANCING STAFFING AND PATIENT VOLUME
Hospital medicine groups face uncertainty about future patient volumes and their characteristics. It is unclear when, how, or even whether hospital medicine groups should return to “normal” pre-COVID staffing models. The following principles can guide staffing decisions.
First, maintain nonhospitalist backup pools and define triggers to activate these providers. Despite the impulse to return to prior staffing models, this recovery period provides an opportunity for leaders to create transparent activation protocols and provide additional training to enable seamless backup. In preparation for a surge, our hospital medicine group quickly assembled an emergency staffing pool composed of advanced practice providers, primary care providers, medicine subspecialists, and surgeons who were prepared to temporarily assume unfamiliar roles. Thankfully, we were able to manage our COVID-19 patients without much emergency hospitalist staffing, but for other hospitals with larger community outbreaks, the emergency backup workforce proved invaluable.
Second, use appropriate safeguards and delegate certain aspects of COVID-related care to other healthcare team members. As staff are deployed and redeployed, consider how interprofessional team members can be reintegrated into evaluation and triage protocols. For example, registered nurses can determine appropriate isolation precautions for patients with COVID and patients under investigation.
Third, consider hospital-specific specialty care patterns when planning for COVID-19 redeployment to ensure access to equally critical, nonelective services. For example, Level 1 trauma centers may expect seasonal increases in trauma patient volumes, so consider staffing trauma teams (including surgeons, anesthesiologists, and operating room staff) for their usual roles to prevent critical coverage gaps. Concurrently, hospital medicine consulting and comanagement teams must also be available to support the trauma service. These staffing needs affect who will be available for redeployment for future COVID-related care.
MANAGING THE PHYSICAL LIMITATIONS OF SPACE
As the number of COVID cases increased, numerous hospitals created geographic “hot zones” with defined cold (uncontaminated), warm (transitional), and hot (contaminated) areas by either partitioning off a section of an acute care medical ward or repurposing an entire ward as a COVID-19 unit, and similar zones were made in intensive care units. Hot zones required significant early investments to change infrastructure, including equipping rooms for negative pressurization with HEPA filtration towers and training staff on safety protocols for entering these spaces, performing necessary patient care, and exiting. Ultimately, these investments proved worthwhile and allowed for decreased personal protective equipment (PPE) use, as well as improved efficiency and staff safety. However, as hospitals ramp up non-COVID care, deciding how to best reconfigure or downsize these hot zones has become challenging.
With time to regroup, the newly experienced end users of hot zones—hospitalists, other staff who worked in these spaces, and patients—must be included in discussions with engineers, architects, and administrators regarding future construction. Hot zone plans should specifically address how physical separation of COVID and non-COVID patients will be maintained while providing safe and efficient care. With elective surgeries increasing and non-COVID patients returning to hospitals, leaders must consider the psychological effects that seeing hospital staff doffing PPE and crossing an invisible barrier to a ‘‘cold” area of the floor has on patients and their families. It is important to maintain hot zones in areas that can dynamically flex to accommodate waves of the current and future pandemics, especially because hospitals may be asked to care for patients from overwhelmed distant sites even if the pandemic is locally controlled. We are experimenting with modifications to hospital traffic patterns including “no pass through” zones, one-way hallways, and separate entries and exits to clinical floors for COVID and non-COVID patients. With vigilant adherence to infection prevention guidelines and PPE use, we have not seen hospital-acquired infections with this model of care.
Modifying space and flow patterns also enables clustered care for COVID patients, which allows for the temporary use of modular teams.4 This tactic may be especially useful during surge periods, during which PPE conservation is paramount and isolating cohorts of providers provides an extra layer of safety. In the longer run, however, isolating providers from their peers risks worsening morale and increasing burnout.
NAVIGATING THE FINANCIAL CHALLENGES
The path forward must ensure safety but also allow for a financially sustainable balance of COVID and non-COVID care. To prepare for surges, health systems canceled elective surgeries and other services that generate essential revenue. At both private and public hospitals, systemwide measures have been taken to mitigate these financial losses. These measures have included salary, retirement, and continuing medical education benefit reductions for physicians and senior leadership; limits to physician hiring and recruitment; leaner operations with systemwide expense reductions; and mandatory and voluntary staff furloughs. The frontline hospital staff, including physicians, nurses, technologists, and food and environmental service workers, who have made great sacrifices during this pandemic, may also now be facing significant personal financial consequences.
The following recommendations are offered from the perspective that crisis creates opportunity for hospital medicine leaders grappling with budget shortfalls.
First, maximize budget transparency by explicitly defining the principles and priorities that govern budget decisions, which allows hospitalist group members to understand how the organization determines budget cuts. For example, stating that a key priority is to minimize staff layoffs makes consequent salary reductions more understandable.
Second, solicit hospital medicine group members’ input on these shared challenges and invite their help in identifying and prioritizing potential cost-saving or cost-cutting measures.
Third, highlight hospitalists’ nonfiscal contributions, especially in terms of crisis leadership, to continue engagement with executive leaders.5 This may include a dialogue about the disproportionate influence of work relative value unit production on salary and about how to create compensation systems that can also recognize crisis readiness as an important feature of sustainability and quality care. The next pandemic surge may be weeks or months away, and hospitalists will again need to be leaders in the response.
Fourth, use this crisis to foster fiscal innovation and accelerate participation in value improvement work, such as redesigning pay-for-performance metrics. Financially strapped institutions will value hospitalists who are good financial stewards. For example, leverage hospitalist expertise in progression of care to facilitate timely disposition of COVID patients, thereby minimizing costly extended hospitalizations.
Lastly, hospital medicine groups must match staffing to patient volume to the extent possible. Approximately two-thirds of hospitalist groups entered this crisis already understaffed and partially reliant on moonlighters,6 which allowed some variation of labor expenses to match lower patient volume. During the recovery phase, hospital volumes may either be significantly below or above baseline; many patients are understandably avoiding hospitals due to fear of COVID. However, delayed care may create a different kind of peak demand for services. For hospitalists, uncertainty about expected clinical roles, COVID vs non-COVID patient mix, and patient volume can be stressful. We recommend sustained, frequent communication about census trends and how shifts will be covered to ensure adequate, long-term staffing. Maintaining trust and morale will be equally, if not more, important in the next phase.
CONCLUSION
As we settle into the marathon, hospital medicine leadership must balance competing priorities with increasing finesse. Our hospital medicine group has benefited from continually discussing operational challenges and refining our strategies as we plan for what is ahead. We have highlighted three mission-critical themes and recommend that hospital and hospital medicine group leaders remain mindful of these challenges and potential strategies. Each of our four academic hospitals has considered similar trade-offs and will proceed along slightly different trajectories to meet unique needs. Looking to the future, we anticipate additional challenges requiring greater ongoing attention alongside those already identified. These include mitigating provider burnout, optimizing resident and student education, and maintaining scholarly work as COVID unpredictably waxes and wanes. By accumulating confidence and wisdom about post-COVID hospital medicine group functions, we hope to provide hospitalists with the energy to keep the pace in the next phase of the marathon.
- Garg M, Wray CM. Hospital medicine management in the time of COVID-19: preparing for a sprint and a marathon. J Hosp Med . 2020;15(5):305-307. https://doi.org/10.12788/jhm.3427
- COVIDView - A weekly Surveillance Summary of U.S. COVID-19 Activity. US Centers for Disease Control and Prevention. July 9, 2020. Accessed July 13, 2020. https://www.cdc.gov/coronavirus/2019-ncov/covid-data/pdf/covidview-07-10-2020.pdf
- Khullar D, Bond AM, Schpero WL. COVID-19 and the financial health of US hospitals. JAMA. Published online May 4, 2020. https://doi.org/10.1001/jama.2020.6269
- Wang CJ, Bair H, Yeh CC. How to prevent and manage hospital-based infections during coronavirus outbreaks: five lessons from Taiwan. J Hosp Med . 2020;15(6):370-371. https://doi.org/10.12788/jhm.3452
- White AA, McIlraith T, Chivu AM, et al. Collaboration, not calculation: a qualitative study of how hospital executives value hospital medicine groups. J Hosp Med. 2019;14(11):662-667. https://doi.org/10.12788/jhm.3249
- 2018 State of Hospital Medicine: 2018 Report Based on 2017 Data . Society of Hospital Medicine; 2018. Accessed July 27, 2020. https://sohm.hospitalmedicine.org/
Hospitalists have played a central role in the massive response to the coronavirus disease 2019 (COVID-19) pandemic by creating innovative staffing models, rapidly learning about the disease and teaching others, and working closely with hospital executive leadership to create surge capacity.1 Some hospitals and regions have weathered an initial storm and are now experiencing a slower influx of COVID-19 patients, while others are now seeing a surge, which is expected to persist for the foreseeable future—the marathon has begun.2 We have entered a new COVID-19 reality: disrupted care models, harsh financial consequences,3 and uncertainty about which adaptations should be preserved and for how long. Common operational challenges will define the new normal. In this Perspective, we share strategies to address these challenges, focusing on three emerging themes: realigning staffing to patient volumes, safely managing space limitations, and navigating the financial ramifications of COVID-19 for hospital medicine groups.
BALANCING STAFFING AND PATIENT VOLUME
Hospital medicine groups face uncertainty about future patient volumes and their characteristics. It is unclear when, how, or even whether hospital medicine groups should return to “normal” pre-COVID staffing models. The following principles can guide staffing decisions.
First, maintain nonhospitalist backup pools and define triggers to activate these providers. Despite the impulse to return to prior staffing models, this recovery period provides an opportunity for leaders to create transparent activation protocols and provide additional training to enable seamless backup. In preparation for a surge, our hospital medicine group quickly assembled an emergency staffing pool composed of advanced practice providers, primary care providers, medicine subspecialists, and surgeons who were prepared to temporarily assume unfamiliar roles. Thankfully, we were able to manage our COVID-19 patients without much emergency hospitalist staffing, but for other hospitals with larger community outbreaks, the emergency backup workforce proved invaluable.
Second, use appropriate safeguards and delegate certain aspects of COVID-related care to other healthcare team members. As staff are deployed and redeployed, consider how interprofessional team members can be reintegrated into evaluation and triage protocols. For example, registered nurses can determine appropriate isolation precautions for patients with COVID and patients under investigation.
Third, consider hospital-specific specialty care patterns when planning for COVID-19 redeployment to ensure access to equally critical, nonelective services. For example, Level 1 trauma centers may expect seasonal increases in trauma patient volumes, so consider staffing trauma teams (including surgeons, anesthesiologists, and operating room staff) for their usual roles to prevent critical coverage gaps. Concurrently, hospital medicine consulting and comanagement teams must also be available to support the trauma service. These staffing needs affect who will be available for redeployment for future COVID-related care.
MANAGING THE PHYSICAL LIMITATIONS OF SPACE
As the number of COVID cases increased, numerous hospitals created geographic “hot zones” with defined cold (uncontaminated), warm (transitional), and hot (contaminated) areas by either partitioning off a section of an acute care medical ward or repurposing an entire ward as a COVID-19 unit, and similar zones were made in intensive care units. Hot zones required significant early investments to change infrastructure, including equipping rooms for negative pressurization with HEPA filtration towers and training staff on safety protocols for entering these spaces, performing necessary patient care, and exiting. Ultimately, these investments proved worthwhile and allowed for decreased personal protective equipment (PPE) use, as well as improved efficiency and staff safety. However, as hospitals ramp up non-COVID care, deciding how to best reconfigure or downsize these hot zones has become challenging.
With time to regroup, the newly experienced end users of hot zones—hospitalists, other staff who worked in these spaces, and patients—must be included in discussions with engineers, architects, and administrators regarding future construction. Hot zone plans should specifically address how physical separation of COVID and non-COVID patients will be maintained while providing safe and efficient care. With elective surgeries increasing and non-COVID patients returning to hospitals, leaders must consider the psychological effects that seeing hospital staff doffing PPE and crossing an invisible barrier to a ‘‘cold” area of the floor has on patients and their families. It is important to maintain hot zones in areas that can dynamically flex to accommodate waves of the current and future pandemics, especially because hospitals may be asked to care for patients from overwhelmed distant sites even if the pandemic is locally controlled. We are experimenting with modifications to hospital traffic patterns including “no pass through” zones, one-way hallways, and separate entries and exits to clinical floors for COVID and non-COVID patients. With vigilant adherence to infection prevention guidelines and PPE use, we have not seen hospital-acquired infections with this model of care.
Modifying space and flow patterns also enables clustered care for COVID patients, which allows for the temporary use of modular teams.4 This tactic may be especially useful during surge periods, during which PPE conservation is paramount and isolating cohorts of providers provides an extra layer of safety. In the longer run, however, isolating providers from their peers risks worsening morale and increasing burnout.
NAVIGATING THE FINANCIAL CHALLENGES
The path forward must ensure safety but also allow for a financially sustainable balance of COVID and non-COVID care. To prepare for surges, health systems canceled elective surgeries and other services that generate essential revenue. At both private and public hospitals, systemwide measures have been taken to mitigate these financial losses. These measures have included salary, retirement, and continuing medical education benefit reductions for physicians and senior leadership; limits to physician hiring and recruitment; leaner operations with systemwide expense reductions; and mandatory and voluntary staff furloughs. The frontline hospital staff, including physicians, nurses, technologists, and food and environmental service workers, who have made great sacrifices during this pandemic, may also now be facing significant personal financial consequences.
The following recommendations are offered from the perspective that crisis creates opportunity for hospital medicine leaders grappling with budget shortfalls.
First, maximize budget transparency by explicitly defining the principles and priorities that govern budget decisions, which allows hospitalist group members to understand how the organization determines budget cuts. For example, stating that a key priority is to minimize staff layoffs makes consequent salary reductions more understandable.
Second, solicit hospital medicine group members’ input on these shared challenges and invite their help in identifying and prioritizing potential cost-saving or cost-cutting measures.
Third, highlight hospitalists’ nonfiscal contributions, especially in terms of crisis leadership, to continue engagement with executive leaders.5 This may include a dialogue about the disproportionate influence of work relative value unit production on salary and about how to create compensation systems that can also recognize crisis readiness as an important feature of sustainability and quality care. The next pandemic surge may be weeks or months away, and hospitalists will again need to be leaders in the response.
Fourth, use this crisis to foster fiscal innovation and accelerate participation in value improvement work, such as redesigning pay-for-performance metrics. Financially strapped institutions will value hospitalists who are good financial stewards. For example, leverage hospitalist expertise in progression of care to facilitate timely disposition of COVID patients, thereby minimizing costly extended hospitalizations.
Lastly, hospital medicine groups must match staffing to patient volume to the extent possible. Approximately two-thirds of hospitalist groups entered this crisis already understaffed and partially reliant on moonlighters,6 which allowed some variation of labor expenses to match lower patient volume. During the recovery phase, hospital volumes may either be significantly below or above baseline; many patients are understandably avoiding hospitals due to fear of COVID. However, delayed care may create a different kind of peak demand for services. For hospitalists, uncertainty about expected clinical roles, COVID vs non-COVID patient mix, and patient volume can be stressful. We recommend sustained, frequent communication about census trends and how shifts will be covered to ensure adequate, long-term staffing. Maintaining trust and morale will be equally, if not more, important in the next phase.
CONCLUSION
As we settle into the marathon, hospital medicine leadership must balance competing priorities with increasing finesse. Our hospital medicine group has benefited from continually discussing operational challenges and refining our strategies as we plan for what is ahead. We have highlighted three mission-critical themes and recommend that hospital and hospital medicine group leaders remain mindful of these challenges and potential strategies. Each of our four academic hospitals has considered similar trade-offs and will proceed along slightly different trajectories to meet unique needs. Looking to the future, we anticipate additional challenges requiring greater ongoing attention alongside those already identified. These include mitigating provider burnout, optimizing resident and student education, and maintaining scholarly work as COVID unpredictably waxes and wanes. By accumulating confidence and wisdom about post-COVID hospital medicine group functions, we hope to provide hospitalists with the energy to keep the pace in the next phase of the marathon.
Hospitalists have played a central role in the massive response to the coronavirus disease 2019 (COVID-19) pandemic by creating innovative staffing models, rapidly learning about the disease and teaching others, and working closely with hospital executive leadership to create surge capacity.1 Some hospitals and regions have weathered an initial storm and are now experiencing a slower influx of COVID-19 patients, while others are now seeing a surge, which is expected to persist for the foreseeable future—the marathon has begun.2 We have entered a new COVID-19 reality: disrupted care models, harsh financial consequences,3 and uncertainty about which adaptations should be preserved and for how long. Common operational challenges will define the new normal. In this Perspective, we share strategies to address these challenges, focusing on three emerging themes: realigning staffing to patient volumes, safely managing space limitations, and navigating the financial ramifications of COVID-19 for hospital medicine groups.
BALANCING STAFFING AND PATIENT VOLUME
Hospital medicine groups face uncertainty about future patient volumes and their characteristics. It is unclear when, how, or even whether hospital medicine groups should return to “normal” pre-COVID staffing models. The following principles can guide staffing decisions.
First, maintain nonhospitalist backup pools and define triggers to activate these providers. Despite the impulse to return to prior staffing models, this recovery period provides an opportunity for leaders to create transparent activation protocols and provide additional training to enable seamless backup. In preparation for a surge, our hospital medicine group quickly assembled an emergency staffing pool composed of advanced practice providers, primary care providers, medicine subspecialists, and surgeons who were prepared to temporarily assume unfamiliar roles. Thankfully, we were able to manage our COVID-19 patients without much emergency hospitalist staffing, but for other hospitals with larger community outbreaks, the emergency backup workforce proved invaluable.
Second, use appropriate safeguards and delegate certain aspects of COVID-related care to other healthcare team members. As staff are deployed and redeployed, consider how interprofessional team members can be reintegrated into evaluation and triage protocols. For example, registered nurses can determine appropriate isolation precautions for patients with COVID and patients under investigation.
Third, consider hospital-specific specialty care patterns when planning for COVID-19 redeployment to ensure access to equally critical, nonelective services. For example, Level 1 trauma centers may expect seasonal increases in trauma patient volumes, so consider staffing trauma teams (including surgeons, anesthesiologists, and operating room staff) for their usual roles to prevent critical coverage gaps. Concurrently, hospital medicine consulting and comanagement teams must also be available to support the trauma service. These staffing needs affect who will be available for redeployment for future COVID-related care.
MANAGING THE PHYSICAL LIMITATIONS OF SPACE
As the number of COVID cases increased, numerous hospitals created geographic “hot zones” with defined cold (uncontaminated), warm (transitional), and hot (contaminated) areas by either partitioning off a section of an acute care medical ward or repurposing an entire ward as a COVID-19 unit, and similar zones were made in intensive care units. Hot zones required significant early investments to change infrastructure, including equipping rooms for negative pressurization with HEPA filtration towers and training staff on safety protocols for entering these spaces, performing necessary patient care, and exiting. Ultimately, these investments proved worthwhile and allowed for decreased personal protective equipment (PPE) use, as well as improved efficiency and staff safety. However, as hospitals ramp up non-COVID care, deciding how to best reconfigure or downsize these hot zones has become challenging.
With time to regroup, the newly experienced end users of hot zones—hospitalists, other staff who worked in these spaces, and patients—must be included in discussions with engineers, architects, and administrators regarding future construction. Hot zone plans should specifically address how physical separation of COVID and non-COVID patients will be maintained while providing safe and efficient care. With elective surgeries increasing and non-COVID patients returning to hospitals, leaders must consider the psychological effects that seeing hospital staff doffing PPE and crossing an invisible barrier to a ‘‘cold” area of the floor has on patients and their families. It is important to maintain hot zones in areas that can dynamically flex to accommodate waves of the current and future pandemics, especially because hospitals may be asked to care for patients from overwhelmed distant sites even if the pandemic is locally controlled. We are experimenting with modifications to hospital traffic patterns including “no pass through” zones, one-way hallways, and separate entries and exits to clinical floors for COVID and non-COVID patients. With vigilant adherence to infection prevention guidelines and PPE use, we have not seen hospital-acquired infections with this model of care.
Modifying space and flow patterns also enables clustered care for COVID patients, which allows for the temporary use of modular teams.4 This tactic may be especially useful during surge periods, during which PPE conservation is paramount and isolating cohorts of providers provides an extra layer of safety. In the longer run, however, isolating providers from their peers risks worsening morale and increasing burnout.
NAVIGATING THE FINANCIAL CHALLENGES
The path forward must ensure safety but also allow for a financially sustainable balance of COVID and non-COVID care. To prepare for surges, health systems canceled elective surgeries and other services that generate essential revenue. At both private and public hospitals, systemwide measures have been taken to mitigate these financial losses. These measures have included salary, retirement, and continuing medical education benefit reductions for physicians and senior leadership; limits to physician hiring and recruitment; leaner operations with systemwide expense reductions; and mandatory and voluntary staff furloughs. The frontline hospital staff, including physicians, nurses, technologists, and food and environmental service workers, who have made great sacrifices during this pandemic, may also now be facing significant personal financial consequences.
The following recommendations are offered from the perspective that crisis creates opportunity for hospital medicine leaders grappling with budget shortfalls.
First, maximize budget transparency by explicitly defining the principles and priorities that govern budget decisions, which allows hospitalist group members to understand how the organization determines budget cuts. For example, stating that a key priority is to minimize staff layoffs makes consequent salary reductions more understandable.
Second, solicit hospital medicine group members’ input on these shared challenges and invite their help in identifying and prioritizing potential cost-saving or cost-cutting measures.
Third, highlight hospitalists’ nonfiscal contributions, especially in terms of crisis leadership, to continue engagement with executive leaders.5 This may include a dialogue about the disproportionate influence of work relative value unit production on salary and about how to create compensation systems that can also recognize crisis readiness as an important feature of sustainability and quality care. The next pandemic surge may be weeks or months away, and hospitalists will again need to be leaders in the response.
Fourth, use this crisis to foster fiscal innovation and accelerate participation in value improvement work, such as redesigning pay-for-performance metrics. Financially strapped institutions will value hospitalists who are good financial stewards. For example, leverage hospitalist expertise in progression of care to facilitate timely disposition of COVID patients, thereby minimizing costly extended hospitalizations.
Lastly, hospital medicine groups must match staffing to patient volume to the extent possible. Approximately two-thirds of hospitalist groups entered this crisis already understaffed and partially reliant on moonlighters,6 which allowed some variation of labor expenses to match lower patient volume. During the recovery phase, hospital volumes may either be significantly below or above baseline; many patients are understandably avoiding hospitals due to fear of COVID. However, delayed care may create a different kind of peak demand for services. For hospitalists, uncertainty about expected clinical roles, COVID vs non-COVID patient mix, and patient volume can be stressful. We recommend sustained, frequent communication about census trends and how shifts will be covered to ensure adequate, long-term staffing. Maintaining trust and morale will be equally, if not more, important in the next phase.
CONCLUSION
As we settle into the marathon, hospital medicine leadership must balance competing priorities with increasing finesse. Our hospital medicine group has benefited from continually discussing operational challenges and refining our strategies as we plan for what is ahead. We have highlighted three mission-critical themes and recommend that hospital and hospital medicine group leaders remain mindful of these challenges and potential strategies. Each of our four academic hospitals has considered similar trade-offs and will proceed along slightly different trajectories to meet unique needs. Looking to the future, we anticipate additional challenges requiring greater ongoing attention alongside those already identified. These include mitigating provider burnout, optimizing resident and student education, and maintaining scholarly work as COVID unpredictably waxes and wanes. By accumulating confidence and wisdom about post-COVID hospital medicine group functions, we hope to provide hospitalists with the energy to keep the pace in the next phase of the marathon.
- Garg M, Wray CM. Hospital medicine management in the time of COVID-19: preparing for a sprint and a marathon. J Hosp Med . 2020;15(5):305-307. https://doi.org/10.12788/jhm.3427
- COVIDView - A weekly Surveillance Summary of U.S. COVID-19 Activity. US Centers for Disease Control and Prevention. July 9, 2020. Accessed July 13, 2020. https://www.cdc.gov/coronavirus/2019-ncov/covid-data/pdf/covidview-07-10-2020.pdf
- Khullar D, Bond AM, Schpero WL. COVID-19 and the financial health of US hospitals. JAMA. Published online May 4, 2020. https://doi.org/10.1001/jama.2020.6269
- Wang CJ, Bair H, Yeh CC. How to prevent and manage hospital-based infections during coronavirus outbreaks: five lessons from Taiwan. J Hosp Med . 2020;15(6):370-371. https://doi.org/10.12788/jhm.3452
- White AA, McIlraith T, Chivu AM, et al. Collaboration, not calculation: a qualitative study of how hospital executives value hospital medicine groups. J Hosp Med. 2019;14(11):662-667. https://doi.org/10.12788/jhm.3249
- 2018 State of Hospital Medicine: 2018 Report Based on 2017 Data . Society of Hospital Medicine; 2018. Accessed July 27, 2020. https://sohm.hospitalmedicine.org/
- Garg M, Wray CM. Hospital medicine management in the time of COVID-19: preparing for a sprint and a marathon. J Hosp Med . 2020;15(5):305-307. https://doi.org/10.12788/jhm.3427
- COVIDView - A weekly Surveillance Summary of U.S. COVID-19 Activity. US Centers for Disease Control and Prevention. July 9, 2020. Accessed July 13, 2020. https://www.cdc.gov/coronavirus/2019-ncov/covid-data/pdf/covidview-07-10-2020.pdf
- Khullar D, Bond AM, Schpero WL. COVID-19 and the financial health of US hospitals. JAMA. Published online May 4, 2020. https://doi.org/10.1001/jama.2020.6269
- Wang CJ, Bair H, Yeh CC. How to prevent and manage hospital-based infections during coronavirus outbreaks: five lessons from Taiwan. J Hosp Med . 2020;15(6):370-371. https://doi.org/10.12788/jhm.3452
- White AA, McIlraith T, Chivu AM, et al. Collaboration, not calculation: a qualitative study of how hospital executives value hospital medicine groups. J Hosp Med. 2019;14(11):662-667. https://doi.org/10.12788/jhm.3249
- 2018 State of Hospital Medicine: 2018 Report Based on 2017 Data . Society of Hospital Medicine; 2018. Accessed July 27, 2020. https://sohm.hospitalmedicine.org/
© 2021 Society of Hospital Medicine
Email: [email protected].
The Need for Standardized Metrics to Drive Decision-making During the COVID-19 Pandemic
The rapid onset of the novel coronavirus disease 2019 (COVID-19) pandemic forced the US healthcare system to scramble to prepare for a health crisis with many unknowns. Early on, it was unclear exactly how the virus was transmitted, how many people would fall ill or how ill they would get, what treatments would be most efficacious, and what resources were needed to care for patients.1 Given the short window the healthcare system had to prepare, many initial and important decisions were made quickly and often at a local level, with limited coordination and standardization across localities and organizations. These decisions included what services could be offered, how best to allocate potentially scarce resources (such as personal protective equipment and ventilators), and how much surge capacity to build.2,3 In short, many of the early decisions about the pandemic were understandably varied, and the lack of standardized metrics to help guide decision-making did not help the situation.
CHALLENGES WITH MANAGING THE PANDEMIC WITHOUT STANDARDIZED METRICS
Unfortunately, as the COVID-19 pandemic continues, there has been insufficient movement toward standardizing definitions for many key measures needed to manage the public health response. Even small differences in definitions can have important implications for decision-making.4 For example, public health officials have recommended communities achieve a positivity rate of 5% or lower for 14 straight days before easing virus-related restrictions.5 In Maryland, two different entities are calculating positivity rates for the state using different methodologies and producing different results, which can have significant public health and economic implications for the state. Johns Hopkins University’s Resource Center calculates the positivity rate by comparing the number of people who tested positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) to all people who were tested. This method consistently produces a positivity rate for Maryland above the 5% threshold. In contrast, the state of Maryland calculates the positivity rate by comparing the number of positive tests for SARS-CoV-2 to the number of tests conducted, even if the same person had multiple tests (unless the tests are performed the same day at the same location). This method consistently produces a positivity rate for Maryland below the 5% threshold.6
THE POLITICIZATION OF THE DATA
The lack of standardized definitions leads not only to debate and confusion over what steps to take next, but also opens the door to politicization of pandemic data. This is readily apparent when considering mortality due to COVID-19. For example, different states use different definitions for COVID-19 mortality. Alabama defines COVID-19 mortality by only including patients who tested positive for the SARS-CoV-2 virus and the cause of death was attributed to COVID-19. In contrast, Colorado’s COVID-19 mortality definition includes those patients who are believed to have died of COVID-19, but does not require confirmation of SARS-CoV-2 infection by a positive test.7 Further compounding the challenge, some politicians reference the COVID-19 mortality rate as a comparison of those who died from COVID-19 with those who were sick with COVID-19, reflecting the success rate of treating patients with COVID-19, an area in which the United States has done relatively well compared with other countries. This definition of the mortality rate suits a narrative of successful pandemic management.8 However, many public health officials suggest the COVID-19 mortality rate should be defined by comparing the number of deaths from COVID-19 as a percentage of the population, which reflects the percentage of the population dying from the disease. In this regard, the United States has not done as well relative to other countries.9 These different definitions highlight how the United States lacks a standardized way to compare its performance across states and with other countries, even on a straightforward measure like mortality.
CURRENT METRICS THAT NEED STANDARDIZATION
The lack of clarity on, and politicization of, pandemic data demonstrate the need to take stock of what metrics require standardization to help public health officials and health system leaders manage the pandemic response moving forward. The Table provides examples of currently used metrics that would benefit from better standardization to inform decision-making across a broad range of settings, including public health, hospitals, physician clinics, and nursing homes. For example, a commonly referenced metric during the pandemic has been a moving average of the incidence rate of positive COVID-19 cases in a defined geographic area (eg, a state).10,11 This data point is helpful to healthcare delivery organizations for understanding the change in COVID-19 cases in their cities and states, which can inform planning on whether or not to continue elective surgeries or how many beds need to be kept in reserve status for a potential surge of hospitalizations. But there has not been a consensus around whether the reporting of COVID-19 positive tests should reflect the day the test was performed or the day the test results were available. The day the test results were available can be influenced by lengthy or uneven turnaround times for the results (eg, backlogs in labs) and can paint a false picture of trends with the virus.
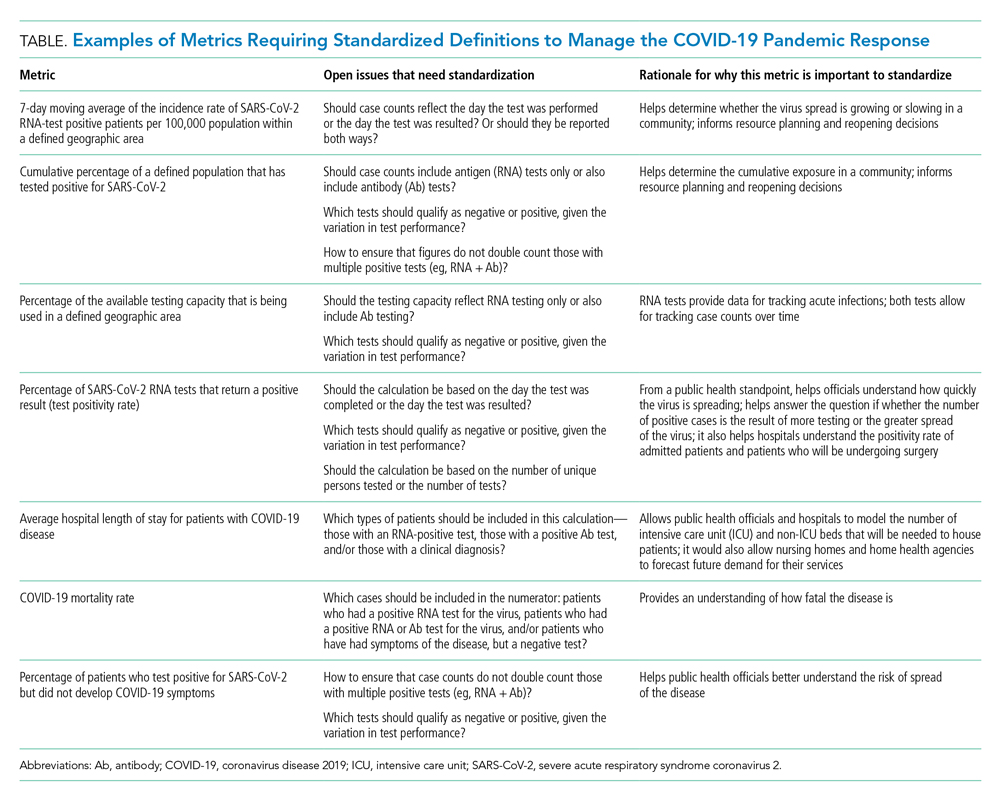
As another example, knowing the percentage of the population that has tested positive for COVID-19 can help inform both resource planning and reopening decisions. But there has been variation in whether counts of positive COVID-19 tests should only include antigen tests, or antibody tests as well. This exact question played out when the Centers for Disease Control and Prevention (CDC) made decisions that differed from those of many states about whether to include antibody tests in their publicly announced COVID-19 testing numbers,12 perhaps undermining public confidence in the reported data.
MOVING FORWARD WITH STANDARDIZING DEFINITIONS
To capture currently unstandardized metrics with broad applicability, the United States should form a consensus task force to identify and define metrics and, over time, refine them based on current science and public health priorities. The task force would require a mix of individuals with various skill sets, such as expertise in infectious diseases and epidemiology, healthcare operations, statistics, performance measurement, and public health. The US Department of Health and Human Services is likely the appropriate sponsor, with representation from the National Institutes of Health, the CDC, and the Agency for Healthcare Research and Quality, in partnership with national provider and public health group representatives.
Once standardized definitions for metrics have been agreed upon, the metric definitions will need to be made readily available to the public and healthcare organizations. Standardization will permit collection of electronic health records for quick calculation and review, with an output of dashboards for reporting. It would also prevent every public health and healthcare delivery organization from having to define its own metrics, freeing them up to focus on planning. Several metrics already have standard definitions, and those metrics have proven useful for decision-making. For example, there is agreement that the turnaround time for a SARS-CoV-2 test is measured by the difference in time between when the test was performed and when the test results were available. This standard definition allows for performance comparisons across different laboratories within the same service area and comparisons across different regions of the country. Once the metrics are standardized, public health leaders and healthcare organizations can use variation in performance and outcomes to identify leading indicators for planning.
CONCLUSION
Amid the COVID-19 pandemic, the US healthcare system finds itself in a state of managing uncertainty for a prolonged period of time. The unprecedented nature of this crisis means that best practices will not always be clear. Providing access to clearly defined, standardized metrics will be essential to public health officials and healthcare organization leaders’ ability to manage through this pandemic. The risk of not moving in this direction means forcing leaders to make decisions without the best information available. Good data will be essential to guiding the US healthcare system through this extraordinary crisis.
- Weston S, Frieman MB. COVID-19: knowns, unknowns, and questions. mSphere. 2020;5(2):e00203-20. https://doi.org/10.1128/mSphere.00203-20
- Griffin KM, Karas MG, Ivascu NS, Lief L. Hospital preparedness for COVID-19: a practical guide from a critical care perspective. Am J Respir Crit Care Med. 2020;201(11):1337-1344. https://doi.org/10.1164/rccm.202004-1037CP
- De Georgeo MR, De Georgeo JM, Egan TM, et al. Containing SARS-CoV-2 in hospitals facing finite PPE, limited testing, and physical space variability: navigating resource constrained enhanced traffic control bundling. J Microbiol Immunol. 2020;S1684-1182(20)30166-3. https://doi.org/10.1016/j.jmii.2020.07.009
- Fischhoff B. Making decisions in a COVID-19 world. JAMA. 2020;324(2):139-140. https://doi.org/10.1001/jama.2020.10178
- Collins K. Is your state doing enough coronavirus testing? New York Times. October 14, 2020. Updated October 29, 2020. Accessed October 14, 2020. https://www.nytimes.com/interactive/2020/us/coronavirus-testing.html
- Ruiz N. Why is Maryland’s coronavirus positivity rate always lower than what Johns Hopkins says it is — and does it matter? Baltimore Sun. September 10, 2020. Accessed October 14, 2020. https://www.baltimoresun.com/coronavirus/bs-md-maryland-coronavirus-positivity-rate-hopkins-20200817-zoepxdjlxbazdm6kabrjehbemq-story.html
- Brown E, Reinhard B, Thebault R. Which deaths count toward the covid-19 death toll? It depends on the state. Washington Post. April 16, 2020. Accessed July 23, 2020. https://www.washingtonpost.com/investigations/which-deaths-count-toward-the-covid-19-death-toll-it-depends-on-the-state/2020/04/16/bca84ae0-7991-11ea-a130-df573469f094_story.html
- Carlisle M. Here’s what Trump got wrong about America’s COVID-19 death rate. Time. August 4, 2020. Accessed October 14, 2020. https://time.com/5875411/trump-covid-19-death-rate-interview/
- Mortality analyses. Johns Hopkins University & Medicine Coronavirus Resource Center. October 14, 2020. Updated October 29, 2020. Accessed October 14, 2020. https://coronavirus.jhu.edu/data/mortality
- COVID-19 daily case incidence rate maps. Kentucky Cabinet for Health and Family Services. Accessed October 14, 2020. https://chfs.ky.gov/Pages/cv19maps.aspx
- COVID-19 trajectory animations. Pennsylvania Department of Health. Accessed October 14, 2020. https://www.health.pa.gov/topics/disease/coronavirus/Pages/Data-Animations.aspx
- Stolberg SG, Kaplan S, Mervosh S. CDC test counting error leaves epidemiologists ‘really baffled.’ New York Times. May 22, 2020. Updated June 3, 2020. Accessed July 23, 2020. https://www.nytimes.com/2020/05/22/us/politics/coronavirus-tests-cdc.html
The rapid onset of the novel coronavirus disease 2019 (COVID-19) pandemic forced the US healthcare system to scramble to prepare for a health crisis with many unknowns. Early on, it was unclear exactly how the virus was transmitted, how many people would fall ill or how ill they would get, what treatments would be most efficacious, and what resources were needed to care for patients.1 Given the short window the healthcare system had to prepare, many initial and important decisions were made quickly and often at a local level, with limited coordination and standardization across localities and organizations. These decisions included what services could be offered, how best to allocate potentially scarce resources (such as personal protective equipment and ventilators), and how much surge capacity to build.2,3 In short, many of the early decisions about the pandemic were understandably varied, and the lack of standardized metrics to help guide decision-making did not help the situation.
CHALLENGES WITH MANAGING THE PANDEMIC WITHOUT STANDARDIZED METRICS
Unfortunately, as the COVID-19 pandemic continues, there has been insufficient movement toward standardizing definitions for many key measures needed to manage the public health response. Even small differences in definitions can have important implications for decision-making.4 For example, public health officials have recommended communities achieve a positivity rate of 5% or lower for 14 straight days before easing virus-related restrictions.5 In Maryland, two different entities are calculating positivity rates for the state using different methodologies and producing different results, which can have significant public health and economic implications for the state. Johns Hopkins University’s Resource Center calculates the positivity rate by comparing the number of people who tested positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) to all people who were tested. This method consistently produces a positivity rate for Maryland above the 5% threshold. In contrast, the state of Maryland calculates the positivity rate by comparing the number of positive tests for SARS-CoV-2 to the number of tests conducted, even if the same person had multiple tests (unless the tests are performed the same day at the same location). This method consistently produces a positivity rate for Maryland below the 5% threshold.6
THE POLITICIZATION OF THE DATA
The lack of standardized definitions leads not only to debate and confusion over what steps to take next, but also opens the door to politicization of pandemic data. This is readily apparent when considering mortality due to COVID-19. For example, different states use different definitions for COVID-19 mortality. Alabama defines COVID-19 mortality by only including patients who tested positive for the SARS-CoV-2 virus and the cause of death was attributed to COVID-19. In contrast, Colorado’s COVID-19 mortality definition includes those patients who are believed to have died of COVID-19, but does not require confirmation of SARS-CoV-2 infection by a positive test.7 Further compounding the challenge, some politicians reference the COVID-19 mortality rate as a comparison of those who died from COVID-19 with those who were sick with COVID-19, reflecting the success rate of treating patients with COVID-19, an area in which the United States has done relatively well compared with other countries. This definition of the mortality rate suits a narrative of successful pandemic management.8 However, many public health officials suggest the COVID-19 mortality rate should be defined by comparing the number of deaths from COVID-19 as a percentage of the population, which reflects the percentage of the population dying from the disease. In this regard, the United States has not done as well relative to other countries.9 These different definitions highlight how the United States lacks a standardized way to compare its performance across states and with other countries, even on a straightforward measure like mortality.
CURRENT METRICS THAT NEED STANDARDIZATION
The lack of clarity on, and politicization of, pandemic data demonstrate the need to take stock of what metrics require standardization to help public health officials and health system leaders manage the pandemic response moving forward. The Table provides examples of currently used metrics that would benefit from better standardization to inform decision-making across a broad range of settings, including public health, hospitals, physician clinics, and nursing homes. For example, a commonly referenced metric during the pandemic has been a moving average of the incidence rate of positive COVID-19 cases in a defined geographic area (eg, a state).10,11 This data point is helpful to healthcare delivery organizations for understanding the change in COVID-19 cases in their cities and states, which can inform planning on whether or not to continue elective surgeries or how many beds need to be kept in reserve status for a potential surge of hospitalizations. But there has not been a consensus around whether the reporting of COVID-19 positive tests should reflect the day the test was performed or the day the test results were available. The day the test results were available can be influenced by lengthy or uneven turnaround times for the results (eg, backlogs in labs) and can paint a false picture of trends with the virus.

As another example, knowing the percentage of the population that has tested positive for COVID-19 can help inform both resource planning and reopening decisions. But there has been variation in whether counts of positive COVID-19 tests should only include antigen tests, or antibody tests as well. This exact question played out when the Centers for Disease Control and Prevention (CDC) made decisions that differed from those of many states about whether to include antibody tests in their publicly announced COVID-19 testing numbers,12 perhaps undermining public confidence in the reported data.
MOVING FORWARD WITH STANDARDIZING DEFINITIONS
To capture currently unstandardized metrics with broad applicability, the United States should form a consensus task force to identify and define metrics and, over time, refine them based on current science and public health priorities. The task force would require a mix of individuals with various skill sets, such as expertise in infectious diseases and epidemiology, healthcare operations, statistics, performance measurement, and public health. The US Department of Health and Human Services is likely the appropriate sponsor, with representation from the National Institutes of Health, the CDC, and the Agency for Healthcare Research and Quality, in partnership with national provider and public health group representatives.
Once standardized definitions for metrics have been agreed upon, the metric definitions will need to be made readily available to the public and healthcare organizations. Standardization will permit collection of electronic health records for quick calculation and review, with an output of dashboards for reporting. It would also prevent every public health and healthcare delivery organization from having to define its own metrics, freeing them up to focus on planning. Several metrics already have standard definitions, and those metrics have proven useful for decision-making. For example, there is agreement that the turnaround time for a SARS-CoV-2 test is measured by the difference in time between when the test was performed and when the test results were available. This standard definition allows for performance comparisons across different laboratories within the same service area and comparisons across different regions of the country. Once the metrics are standardized, public health leaders and healthcare organizations can use variation in performance and outcomes to identify leading indicators for planning.
CONCLUSION
Amid the COVID-19 pandemic, the US healthcare system finds itself in a state of managing uncertainty for a prolonged period of time. The unprecedented nature of this crisis means that best practices will not always be clear. Providing access to clearly defined, standardized metrics will be essential to public health officials and healthcare organization leaders’ ability to manage through this pandemic. The risk of not moving in this direction means forcing leaders to make decisions without the best information available. Good data will be essential to guiding the US healthcare system through this extraordinary crisis.
The rapid onset of the novel coronavirus disease 2019 (COVID-19) pandemic forced the US healthcare system to scramble to prepare for a health crisis with many unknowns. Early on, it was unclear exactly how the virus was transmitted, how many people would fall ill or how ill they would get, what treatments would be most efficacious, and what resources were needed to care for patients.1 Given the short window the healthcare system had to prepare, many initial and important decisions were made quickly and often at a local level, with limited coordination and standardization across localities and organizations. These decisions included what services could be offered, how best to allocate potentially scarce resources (such as personal protective equipment and ventilators), and how much surge capacity to build.2,3 In short, many of the early decisions about the pandemic were understandably varied, and the lack of standardized metrics to help guide decision-making did not help the situation.
CHALLENGES WITH MANAGING THE PANDEMIC WITHOUT STANDARDIZED METRICS
Unfortunately, as the COVID-19 pandemic continues, there has been insufficient movement toward standardizing definitions for many key measures needed to manage the public health response. Even small differences in definitions can have important implications for decision-making.4 For example, public health officials have recommended communities achieve a positivity rate of 5% or lower for 14 straight days before easing virus-related restrictions.5 In Maryland, two different entities are calculating positivity rates for the state using different methodologies and producing different results, which can have significant public health and economic implications for the state. Johns Hopkins University’s Resource Center calculates the positivity rate by comparing the number of people who tested positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) to all people who were tested. This method consistently produces a positivity rate for Maryland above the 5% threshold. In contrast, the state of Maryland calculates the positivity rate by comparing the number of positive tests for SARS-CoV-2 to the number of tests conducted, even if the same person had multiple tests (unless the tests are performed the same day at the same location). This method consistently produces a positivity rate for Maryland below the 5% threshold.6
THE POLITICIZATION OF THE DATA
The lack of standardized definitions leads not only to debate and confusion over what steps to take next, but also opens the door to politicization of pandemic data. This is readily apparent when considering mortality due to COVID-19. For example, different states use different definitions for COVID-19 mortality. Alabama defines COVID-19 mortality by only including patients who tested positive for the SARS-CoV-2 virus and the cause of death was attributed to COVID-19. In contrast, Colorado’s COVID-19 mortality definition includes those patients who are believed to have died of COVID-19, but does not require confirmation of SARS-CoV-2 infection by a positive test.7 Further compounding the challenge, some politicians reference the COVID-19 mortality rate as a comparison of those who died from COVID-19 with those who were sick with COVID-19, reflecting the success rate of treating patients with COVID-19, an area in which the United States has done relatively well compared with other countries. This definition of the mortality rate suits a narrative of successful pandemic management.8 However, many public health officials suggest the COVID-19 mortality rate should be defined by comparing the number of deaths from COVID-19 as a percentage of the population, which reflects the percentage of the population dying from the disease. In this regard, the United States has not done as well relative to other countries.9 These different definitions highlight how the United States lacks a standardized way to compare its performance across states and with other countries, even on a straightforward measure like mortality.
CURRENT METRICS THAT NEED STANDARDIZATION
The lack of clarity on, and politicization of, pandemic data demonstrate the need to take stock of what metrics require standardization to help public health officials and health system leaders manage the pandemic response moving forward. The Table provides examples of currently used metrics that would benefit from better standardization to inform decision-making across a broad range of settings, including public health, hospitals, physician clinics, and nursing homes. For example, a commonly referenced metric during the pandemic has been a moving average of the incidence rate of positive COVID-19 cases in a defined geographic area (eg, a state).10,11 This data point is helpful to healthcare delivery organizations for understanding the change in COVID-19 cases in their cities and states, which can inform planning on whether or not to continue elective surgeries or how many beds need to be kept in reserve status for a potential surge of hospitalizations. But there has not been a consensus around whether the reporting of COVID-19 positive tests should reflect the day the test was performed or the day the test results were available. The day the test results were available can be influenced by lengthy or uneven turnaround times for the results (eg, backlogs in labs) and can paint a false picture of trends with the virus.

As another example, knowing the percentage of the population that has tested positive for COVID-19 can help inform both resource planning and reopening decisions. But there has been variation in whether counts of positive COVID-19 tests should only include antigen tests, or antibody tests as well. This exact question played out when the Centers for Disease Control and Prevention (CDC) made decisions that differed from those of many states about whether to include antibody tests in their publicly announced COVID-19 testing numbers,12 perhaps undermining public confidence in the reported data.
MOVING FORWARD WITH STANDARDIZING DEFINITIONS
To capture currently unstandardized metrics with broad applicability, the United States should form a consensus task force to identify and define metrics and, over time, refine them based on current science and public health priorities. The task force would require a mix of individuals with various skill sets, such as expertise in infectious diseases and epidemiology, healthcare operations, statistics, performance measurement, and public health. The US Department of Health and Human Services is likely the appropriate sponsor, with representation from the National Institutes of Health, the CDC, and the Agency for Healthcare Research and Quality, in partnership with national provider and public health group representatives.
Once standardized definitions for metrics have been agreed upon, the metric definitions will need to be made readily available to the public and healthcare organizations. Standardization will permit collection of electronic health records for quick calculation and review, with an output of dashboards for reporting. It would also prevent every public health and healthcare delivery organization from having to define its own metrics, freeing them up to focus on planning. Several metrics already have standard definitions, and those metrics have proven useful for decision-making. For example, there is agreement that the turnaround time for a SARS-CoV-2 test is measured by the difference in time between when the test was performed and when the test results were available. This standard definition allows for performance comparisons across different laboratories within the same service area and comparisons across different regions of the country. Once the metrics are standardized, public health leaders and healthcare organizations can use variation in performance and outcomes to identify leading indicators for planning.
CONCLUSION
Amid the COVID-19 pandemic, the US healthcare system finds itself in a state of managing uncertainty for a prolonged period of time. The unprecedented nature of this crisis means that best practices will not always be clear. Providing access to clearly defined, standardized metrics will be essential to public health officials and healthcare organization leaders’ ability to manage through this pandemic. The risk of not moving in this direction means forcing leaders to make decisions without the best information available. Good data will be essential to guiding the US healthcare system through this extraordinary crisis.
- Weston S, Frieman MB. COVID-19: knowns, unknowns, and questions. mSphere. 2020;5(2):e00203-20. https://doi.org/10.1128/mSphere.00203-20
- Griffin KM, Karas MG, Ivascu NS, Lief L. Hospital preparedness for COVID-19: a practical guide from a critical care perspective. Am J Respir Crit Care Med. 2020;201(11):1337-1344. https://doi.org/10.1164/rccm.202004-1037CP
- De Georgeo MR, De Georgeo JM, Egan TM, et al. Containing SARS-CoV-2 in hospitals facing finite PPE, limited testing, and physical space variability: navigating resource constrained enhanced traffic control bundling. J Microbiol Immunol. 2020;S1684-1182(20)30166-3. https://doi.org/10.1016/j.jmii.2020.07.009
- Fischhoff B. Making decisions in a COVID-19 world. JAMA. 2020;324(2):139-140. https://doi.org/10.1001/jama.2020.10178
- Collins K. Is your state doing enough coronavirus testing? New York Times. October 14, 2020. Updated October 29, 2020. Accessed October 14, 2020. https://www.nytimes.com/interactive/2020/us/coronavirus-testing.html
- Ruiz N. Why is Maryland’s coronavirus positivity rate always lower than what Johns Hopkins says it is — and does it matter? Baltimore Sun. September 10, 2020. Accessed October 14, 2020. https://www.baltimoresun.com/coronavirus/bs-md-maryland-coronavirus-positivity-rate-hopkins-20200817-zoepxdjlxbazdm6kabrjehbemq-story.html
- Brown E, Reinhard B, Thebault R. Which deaths count toward the covid-19 death toll? It depends on the state. Washington Post. April 16, 2020. Accessed July 23, 2020. https://www.washingtonpost.com/investigations/which-deaths-count-toward-the-covid-19-death-toll-it-depends-on-the-state/2020/04/16/bca84ae0-7991-11ea-a130-df573469f094_story.html
- Carlisle M. Here’s what Trump got wrong about America’s COVID-19 death rate. Time. August 4, 2020. Accessed October 14, 2020. https://time.com/5875411/trump-covid-19-death-rate-interview/
- Mortality analyses. Johns Hopkins University & Medicine Coronavirus Resource Center. October 14, 2020. Updated October 29, 2020. Accessed October 14, 2020. https://coronavirus.jhu.edu/data/mortality
- COVID-19 daily case incidence rate maps. Kentucky Cabinet for Health and Family Services. Accessed October 14, 2020. https://chfs.ky.gov/Pages/cv19maps.aspx
- COVID-19 trajectory animations. Pennsylvania Department of Health. Accessed October 14, 2020. https://www.health.pa.gov/topics/disease/coronavirus/Pages/Data-Animations.aspx
- Stolberg SG, Kaplan S, Mervosh S. CDC test counting error leaves epidemiologists ‘really baffled.’ New York Times. May 22, 2020. Updated June 3, 2020. Accessed July 23, 2020. https://www.nytimes.com/2020/05/22/us/politics/coronavirus-tests-cdc.html
- Weston S, Frieman MB. COVID-19: knowns, unknowns, and questions. mSphere. 2020;5(2):e00203-20. https://doi.org/10.1128/mSphere.00203-20
- Griffin KM, Karas MG, Ivascu NS, Lief L. Hospital preparedness for COVID-19: a practical guide from a critical care perspective. Am J Respir Crit Care Med. 2020;201(11):1337-1344. https://doi.org/10.1164/rccm.202004-1037CP
- De Georgeo MR, De Georgeo JM, Egan TM, et al. Containing SARS-CoV-2 in hospitals facing finite PPE, limited testing, and physical space variability: navigating resource constrained enhanced traffic control bundling. J Microbiol Immunol. 2020;S1684-1182(20)30166-3. https://doi.org/10.1016/j.jmii.2020.07.009
- Fischhoff B. Making decisions in a COVID-19 world. JAMA. 2020;324(2):139-140. https://doi.org/10.1001/jama.2020.10178
- Collins K. Is your state doing enough coronavirus testing? New York Times. October 14, 2020. Updated October 29, 2020. Accessed October 14, 2020. https://www.nytimes.com/interactive/2020/us/coronavirus-testing.html
- Ruiz N. Why is Maryland’s coronavirus positivity rate always lower than what Johns Hopkins says it is — and does it matter? Baltimore Sun. September 10, 2020. Accessed October 14, 2020. https://www.baltimoresun.com/coronavirus/bs-md-maryland-coronavirus-positivity-rate-hopkins-20200817-zoepxdjlxbazdm6kabrjehbemq-story.html
- Brown E, Reinhard B, Thebault R. Which deaths count toward the covid-19 death toll? It depends on the state. Washington Post. April 16, 2020. Accessed July 23, 2020. https://www.washingtonpost.com/investigations/which-deaths-count-toward-the-covid-19-death-toll-it-depends-on-the-state/2020/04/16/bca84ae0-7991-11ea-a130-df573469f094_story.html
- Carlisle M. Here’s what Trump got wrong about America’s COVID-19 death rate. Time. August 4, 2020. Accessed October 14, 2020. https://time.com/5875411/trump-covid-19-death-rate-interview/
- Mortality analyses. Johns Hopkins University & Medicine Coronavirus Resource Center. October 14, 2020. Updated October 29, 2020. Accessed October 14, 2020. https://coronavirus.jhu.edu/data/mortality
- COVID-19 daily case incidence rate maps. Kentucky Cabinet for Health and Family Services. Accessed October 14, 2020. https://chfs.ky.gov/Pages/cv19maps.aspx
- COVID-19 trajectory animations. Pennsylvania Department of Health. Accessed October 14, 2020. https://www.health.pa.gov/topics/disease/coronavirus/Pages/Data-Animations.aspx
- Stolberg SG, Kaplan S, Mervosh S. CDC test counting error leaves epidemiologists ‘really baffled.’ New York Times. May 22, 2020. Updated June 3, 2020. Accessed July 23, 2020. https://www.nytimes.com/2020/05/22/us/politics/coronavirus-tests-cdc.html
© 2021 Society of Hospital Medicine
Email: [email protected]; Telephone: 832-816-5618; Twitter: @JMatthewAustin.
Opportunities for Improving Population Health in the Post–COVID-19 Era
The novel coronavirus disease of 2019 (COVID-19), caused by the SARS-CoV-2 pathogen, has resulted in a health crisis unlike any other experienced in the past century, with millions of people infected and over one million people dying from COVID-19 worldwide. The pandemic has disproportionately impacted historically marginalized groups, resulting in higher rates of infection, hospitalization, and death in racial/ethnic minority populations, including Black, Hispanic/Latinx, and Native American populations, compared with the White population.1 Statistics suggest that it is not just socioeconomic differences but also structural racism that has played a role in worse health outcomes in minority populations. However, the health inequities uncovered by the pandemic represent an opportunity—a “plastic hour” in which improvements at the population level may be uniquely possible.2 As healthcare providers, we must take advantage of this moment and work toward improving healthcare and increasing health equity in the post–COVID-19 era. We highlight three strategies to guide us toward achieving this goal: (1) prioritizing health system equity and government improvements to population health, (2) fostering community resilience, and (3) promoting equity in economic sustainability.
HEALTH SYSTEM AND GOVERNMENT IMPROVEMENTS TO POPULATION HEALTH
The COVID-19 pandemic has revealed deep-seated structural and medical vulnerabilities in the US healthcare system, with distressing racial/ethnic differences in COVID-19 infection continuing to emerge.3 Despite variation in the availability and quality of these data, disparities observed in COVID-19 have tracked closely with historical inequities in access to healthcare and discrimination within the healthcare system.4 Any approach to addressing these inequities must appreciate the intersection between social and medical vulnerabilities.
It is notable that healthcare systems serving the most vulnerable populations have borne the brunt of the economic toll of COVID-19. Hospitals in socioeconomically challenged areas lost millions of dollars due to the postponement of elective procedures and reallocation of most resources to COVID-related hospital admissions. Many community-based practices, already stretched in caring for medically and socially complex patients, had to shut their doors. These losses have left patients without the support of their network of healthcare and community service organizations—at the same time that many of them have also lost support for food and housing, employer-based health insurance, and in-person schooling and childcare.
The current circumstances due to the COVID-19 pandemic, therefore, require us to reconsider many aspects of both healthcare and the social safety net, including the reliance on financial penalties as a strategy to improve health quality, which ultimately has a disproportionate impact on communities of color.5 The present situation may also allow for the federal, state, and local governments, as well as health systems and payers, to make targeted investments in healthcare, public health, and community programs. For example, an increased healthcare system investment on preventive and primary care will be essential to reducing the chronic risk factors that underlie COVID-19 infection and death. Efforts by payers to reduce economic incentives for unnecessary elective procedures, while simultaneously providing incentives to increase the focus on preventive care, would further stimulate this effort. Although there is controversy over the inclusion of social risk in financial and value-based health system payment models, novel approaches to this problem (eg, consideration of improvement over achievement of static targets) may provide an opportunity for struggling health systems to invest in new strategies for underserved populations. Additionally, investing in a care system that allows racial, language, and cultural concordance between clinicians and patients would both promote a diverse workforce and improve quality of care. Health system equity will also depend upon bold policy advances such as expansion of Medicaid to all states, separation of health insurance from employment, and targeted government and health system investments around social risk (eg, food and housing insecurity). These programs will help vulnerable communities close the gap on disparities in health outcomes that have been so persistent.
Some of these specific concerns were addressed by the Coronavirus Aid, Relief, and Economic Security (CARES) Act that was implemented by the US Congress to address the broad needs of Americans during the acute crisis.6 The CARES Act provided supplementary funding to community health centers and healthcare systems caring for the uninsured. Cash assistance was provided to most US taxpayers along with financial support to those experiencing unemployment through July 31, 2020, measures that have yet to be extended. In addition to the CARES Act, policymakers proposed establishing a COVID-19 Racial and Ethnic Disparities Task Force Act to drive equitable recommendations and provide oversight to the nation’s response to COVID-19.7
While these measures were critical to the immediate pandemic response, future US congressional relief plans are needed to ensure equity remains a tenet of state and federal policy post COVID-19, particularly with respect to social determinants of health. Additional recommendations for federal relief include rent assistance for low-income families, eviction stoppages, and increased funding for short-term food insecurity. With respect to long-term goals, this is the time to address broader injustices, such as lack of affordable housing, lack of a sensible national strategy around food security, and a lack of equitable educational and justice systems. This moment also offers an opportunity to consider the best way to address the impact of centuries of structural racism. If we place equity at the center of policy implementation, we will certainly see downstream health consequences—ones that would begin to address the health disparities present long before the current pandemic.
FOSTERING COMMUNITY RESILIENCE
While national, state, and local responses to COVID-19 are required to bolster population health when we emerge from the COVID-19 crisis, a focus on community resilience is also needed. Community resilience, or the ability to prevent, withstand, and mitigate the stress of a disaster like COVID-19, requires integration of emergency preparedness practices into community disaster programs, with ongoing efforts to mitigate disparities in chronic disease management. A framework for community resilience includes (1) engaging with communities in planning, response, and post–COVID-19 recovery, (2) ensuring communities have access to high quality, culturally concordant health and social services, and (3) developing robust community networks to mobilize individuals, community services, and public health infrastructure in times of emergency.8
After seeing the devastating effects of Hurricane Katrina in 2005, researchers, public health officials, and community leaders founded the Los Angeles County Community Disaster Resilience (LACCDR) project. Through this collaborative effort, the LACCDR established partnerships across 16 communities to foster community resilience during health emergencies against the backdrop of daily chronic stressors such as violence, segregation, poverty, and homelessness.8 A model such as this to improve health systems and public health integration post-COVID will support health provisions and help build trust in communities wherein there is a high distrust of the healthcare system. Engaging with community partners early to ensure that its members have access to basic needs (eg, food, water, shelter), public health needs (eg, timely information, personal protective equipment such as face coverings and cleaning supplies), and affordable testing and vaccination will help prevent disparities that could affect the most vulnerable in future phases of the COVID-19 crisis.
PROMOTING EQUITY AS A SUSTAINABLE ECONOMIC STRATEGY
Over 40 million Americans were seeking unemployment benefits at the peak of the economic repercussions of the COVID-19 pandemic. Unfortunately, low-income, rural, and minority communities disproportionately experienced this economic shock. Given the relationship between wealth and health, successfully achieving equity post-COVID-19 will require deeper financial investments in underserved communities.9 Healthcare organizations, which represent 18% of the United States gross domestic product and employ nearly 9% of all working individuals, are uniquely positioned to have a direct influence on this strategy.
One equity-based strategy is for healthcare institutions to pursue an anchor mission. Anchor missions have increased a health system’s investment in social services, including providing housing and food resources.10 Additionally, hospitals such as Brigham and Women’s, Boston Children’s Hospital, and Bon Secours Health System, are working with a diverse group of entrepreneurs to create jobs and build wealth in underserved communities by employing local and minority-owned businesses to support critical supply chain purchasing decisions regarding food, maintenance, and construction projects.11 These local and inclusive hiring and procurement measures can be bolstered by continued place-based investments by all health system leaders in vulnerable communities.
CONCLUSION
Since the first enslaved Africans were brought to America over 400 years ago, racial and ethnic minorities have experienced struggle and triumph, sadness and joy. The bonds of a long legacy of discrimination are so deep that we must be intentional in our pursuit of equity—during and beyond the COVID-19 pandemic. Placing equity at the center of healthcare system practice and policy implementation, fostering community resilience and emergency preparedness, and prioritizing equity in economic strategic planning are key steps toward addressing the population-level inequities exposed by the COVID-19 pandemic. As the once touted “great equalizer” rages on, we must remember that we are all jointly affected by the distress caused by the novel coronavirus and we also must be more aware than ever of our interconnectedness. We can use this time of pandemic to fight more than ever to ensure that all populations can enjoy just and optimal health.
Acknowledgments
The authors would like to thank Dr Denise Polit for her review of this manuscript.
- Williams DR, Cooper LA. COVID–19 and health equity–a new kind of “herd immunity”. JAMA. 2020;323(24):2478-2480. https://doi.org/10.1001/jama.2020.8051
- Packer G. America’s plastic hour is upon us. The Atlantic. October 2020. Accessed September 28, 2020. https://www.theatlantic.com/magazine/archive/2020/10/make-america-again/615478/
- Gross CP, Essien UR, Pasha S, Gross JR, Wang SY, Nunez-Smith M. Racial and ethnic disparities in population-level Covid-19 mortality. J Gen Intern Med. 2020;35(10):3097-3099. https://doi.org/10.1007/s11606-020-06081-w
- Institute of Medicine (US) Committee on Understanding and Eliminating Racial and Ethnic Disparities in Health Care, Smedley BD, Stith AY, Nelson AR, eds. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. National Academies Press (US); 2003. https://doi.org/10.17226/12875
- Zuckerman RB, Joynt Maddox KE, Sheingold SH, Chen LM, Epstein AM. Effect of a hospital-wide measure on the readmissions reduction program. N Engl J Med. 2017;377(16):1551-1558. https://doi.org/10.1056/nejmsa1701791
- Cochrane E. House passes relief for small businesses and aid for hospitals and testing. New York Times. April 23, 2020. Accessed May 21, 2020. https://www.nytimes.com/2020/04/23/us/politics/house-passes-relief-for-small-businesses-and-aid-for-hospitals-and-testing.html
- Harris announces legislation to establish task force to combat racial and ethnic disparities in COVID-19. News release. Kamala D. Harris US Senator for California; April 30, 2020. Accessed May 21, 2020. https://www.harris.senate.gov/news/press-releases/harris-announces-legislation-to-establish-task-force-to-combat-racial-and-ethnic-disparities-in-covid-19
- Chandra A, Williams M, Plough A, et al. Getting actionable about community resilience: the Los Angeles County Community Disaster Resilience project. Am J Public Health. 2013;103(7):1181-1189. https://doi.org/10.2105/ajph.2013.301270
- Rawshani A, Svensson AM, Zethelius B, Eliasson B, Rosengren A, Gudbjörnsdottir S. Association between socioeconomic status and mortality, cardiovascular disease, and cancer in patients with type 2 diabetes. JAMA Intern Med. 2016;176(8):1146-1154. https://doi.org/10.1001/jamainternmed.2016.2940
- Horwitz LI, Chang C, Arcilla HN, Knickman JR. Quantifying health systems’ investment in social determinants of health, by sector, 2017-19. Health Aff (Millwood). 2020;39(2):192-198. https://doi.org/10.1377/hlthaff.2019.01246
- Nanos J. Diverse, locally owned food start-ups make the menus at Harvard, UMass, and BC. Boston Globe. January 24, 2020. Accessed September 28, 2020. https://www.bostonglobe.com/business/2020/01/24/diverse-locally-owned-food-start-ups-make-menus-harvard-umass-and/WwJFew6KVgXu1NyIK1BNqI/story.html
The novel coronavirus disease of 2019 (COVID-19), caused by the SARS-CoV-2 pathogen, has resulted in a health crisis unlike any other experienced in the past century, with millions of people infected and over one million people dying from COVID-19 worldwide. The pandemic has disproportionately impacted historically marginalized groups, resulting in higher rates of infection, hospitalization, and death in racial/ethnic minority populations, including Black, Hispanic/Latinx, and Native American populations, compared with the White population.1 Statistics suggest that it is not just socioeconomic differences but also structural racism that has played a role in worse health outcomes in minority populations. However, the health inequities uncovered by the pandemic represent an opportunity—a “plastic hour” in which improvements at the population level may be uniquely possible.2 As healthcare providers, we must take advantage of this moment and work toward improving healthcare and increasing health equity in the post–COVID-19 era. We highlight three strategies to guide us toward achieving this goal: (1) prioritizing health system equity and government improvements to population health, (2) fostering community resilience, and (3) promoting equity in economic sustainability.
HEALTH SYSTEM AND GOVERNMENT IMPROVEMENTS TO POPULATION HEALTH
The COVID-19 pandemic has revealed deep-seated structural and medical vulnerabilities in the US healthcare system, with distressing racial/ethnic differences in COVID-19 infection continuing to emerge.3 Despite variation in the availability and quality of these data, disparities observed in COVID-19 have tracked closely with historical inequities in access to healthcare and discrimination within the healthcare system.4 Any approach to addressing these inequities must appreciate the intersection between social and medical vulnerabilities.
It is notable that healthcare systems serving the most vulnerable populations have borne the brunt of the economic toll of COVID-19. Hospitals in socioeconomically challenged areas lost millions of dollars due to the postponement of elective procedures and reallocation of most resources to COVID-related hospital admissions. Many community-based practices, already stretched in caring for medically and socially complex patients, had to shut their doors. These losses have left patients without the support of their network of healthcare and community service organizations—at the same time that many of them have also lost support for food and housing, employer-based health insurance, and in-person schooling and childcare.
The current circumstances due to the COVID-19 pandemic, therefore, require us to reconsider many aspects of both healthcare and the social safety net, including the reliance on financial penalties as a strategy to improve health quality, which ultimately has a disproportionate impact on communities of color.5 The present situation may also allow for the federal, state, and local governments, as well as health systems and payers, to make targeted investments in healthcare, public health, and community programs. For example, an increased healthcare system investment on preventive and primary care will be essential to reducing the chronic risk factors that underlie COVID-19 infection and death. Efforts by payers to reduce economic incentives for unnecessary elective procedures, while simultaneously providing incentives to increase the focus on preventive care, would further stimulate this effort. Although there is controversy over the inclusion of social risk in financial and value-based health system payment models, novel approaches to this problem (eg, consideration of improvement over achievement of static targets) may provide an opportunity for struggling health systems to invest in new strategies for underserved populations. Additionally, investing in a care system that allows racial, language, and cultural concordance between clinicians and patients would both promote a diverse workforce and improve quality of care. Health system equity will also depend upon bold policy advances such as expansion of Medicaid to all states, separation of health insurance from employment, and targeted government and health system investments around social risk (eg, food and housing insecurity). These programs will help vulnerable communities close the gap on disparities in health outcomes that have been so persistent.
Some of these specific concerns were addressed by the Coronavirus Aid, Relief, and Economic Security (CARES) Act that was implemented by the US Congress to address the broad needs of Americans during the acute crisis.6 The CARES Act provided supplementary funding to community health centers and healthcare systems caring for the uninsured. Cash assistance was provided to most US taxpayers along with financial support to those experiencing unemployment through July 31, 2020, measures that have yet to be extended. In addition to the CARES Act, policymakers proposed establishing a COVID-19 Racial and Ethnic Disparities Task Force Act to drive equitable recommendations and provide oversight to the nation’s response to COVID-19.7
While these measures were critical to the immediate pandemic response, future US congressional relief plans are needed to ensure equity remains a tenet of state and federal policy post COVID-19, particularly with respect to social determinants of health. Additional recommendations for federal relief include rent assistance for low-income families, eviction stoppages, and increased funding for short-term food insecurity. With respect to long-term goals, this is the time to address broader injustices, such as lack of affordable housing, lack of a sensible national strategy around food security, and a lack of equitable educational and justice systems. This moment also offers an opportunity to consider the best way to address the impact of centuries of structural racism. If we place equity at the center of policy implementation, we will certainly see downstream health consequences—ones that would begin to address the health disparities present long before the current pandemic.
FOSTERING COMMUNITY RESILIENCE
While national, state, and local responses to COVID-19 are required to bolster population health when we emerge from the COVID-19 crisis, a focus on community resilience is also needed. Community resilience, or the ability to prevent, withstand, and mitigate the stress of a disaster like COVID-19, requires integration of emergency preparedness practices into community disaster programs, with ongoing efforts to mitigate disparities in chronic disease management. A framework for community resilience includes (1) engaging with communities in planning, response, and post–COVID-19 recovery, (2) ensuring communities have access to high quality, culturally concordant health and social services, and (3) developing robust community networks to mobilize individuals, community services, and public health infrastructure in times of emergency.8
After seeing the devastating effects of Hurricane Katrina in 2005, researchers, public health officials, and community leaders founded the Los Angeles County Community Disaster Resilience (LACCDR) project. Through this collaborative effort, the LACCDR established partnerships across 16 communities to foster community resilience during health emergencies against the backdrop of daily chronic stressors such as violence, segregation, poverty, and homelessness.8 A model such as this to improve health systems and public health integration post-COVID will support health provisions and help build trust in communities wherein there is a high distrust of the healthcare system. Engaging with community partners early to ensure that its members have access to basic needs (eg, food, water, shelter), public health needs (eg, timely information, personal protective equipment such as face coverings and cleaning supplies), and affordable testing and vaccination will help prevent disparities that could affect the most vulnerable in future phases of the COVID-19 crisis.
PROMOTING EQUITY AS A SUSTAINABLE ECONOMIC STRATEGY
Over 40 million Americans were seeking unemployment benefits at the peak of the economic repercussions of the COVID-19 pandemic. Unfortunately, low-income, rural, and minority communities disproportionately experienced this economic shock. Given the relationship between wealth and health, successfully achieving equity post-COVID-19 will require deeper financial investments in underserved communities.9 Healthcare organizations, which represent 18% of the United States gross domestic product and employ nearly 9% of all working individuals, are uniquely positioned to have a direct influence on this strategy.
One equity-based strategy is for healthcare institutions to pursue an anchor mission. Anchor missions have increased a health system’s investment in social services, including providing housing and food resources.10 Additionally, hospitals such as Brigham and Women’s, Boston Children’s Hospital, and Bon Secours Health System, are working with a diverse group of entrepreneurs to create jobs and build wealth in underserved communities by employing local and minority-owned businesses to support critical supply chain purchasing decisions regarding food, maintenance, and construction projects.11 These local and inclusive hiring and procurement measures can be bolstered by continued place-based investments by all health system leaders in vulnerable communities.
CONCLUSION
Since the first enslaved Africans were brought to America over 400 years ago, racial and ethnic minorities have experienced struggle and triumph, sadness and joy. The bonds of a long legacy of discrimination are so deep that we must be intentional in our pursuit of equity—during and beyond the COVID-19 pandemic. Placing equity at the center of healthcare system practice and policy implementation, fostering community resilience and emergency preparedness, and prioritizing equity in economic strategic planning are key steps toward addressing the population-level inequities exposed by the COVID-19 pandemic. As the once touted “great equalizer” rages on, we must remember that we are all jointly affected by the distress caused by the novel coronavirus and we also must be more aware than ever of our interconnectedness. We can use this time of pandemic to fight more than ever to ensure that all populations can enjoy just and optimal health.
Acknowledgments
The authors would like to thank Dr Denise Polit for her review of this manuscript.
The novel coronavirus disease of 2019 (COVID-19), caused by the SARS-CoV-2 pathogen, has resulted in a health crisis unlike any other experienced in the past century, with millions of people infected and over one million people dying from COVID-19 worldwide. The pandemic has disproportionately impacted historically marginalized groups, resulting in higher rates of infection, hospitalization, and death in racial/ethnic minority populations, including Black, Hispanic/Latinx, and Native American populations, compared with the White population.1 Statistics suggest that it is not just socioeconomic differences but also structural racism that has played a role in worse health outcomes in minority populations. However, the health inequities uncovered by the pandemic represent an opportunity—a “plastic hour” in which improvements at the population level may be uniquely possible.2 As healthcare providers, we must take advantage of this moment and work toward improving healthcare and increasing health equity in the post–COVID-19 era. We highlight three strategies to guide us toward achieving this goal: (1) prioritizing health system equity and government improvements to population health, (2) fostering community resilience, and (3) promoting equity in economic sustainability.
HEALTH SYSTEM AND GOVERNMENT IMPROVEMENTS TO POPULATION HEALTH
The COVID-19 pandemic has revealed deep-seated structural and medical vulnerabilities in the US healthcare system, with distressing racial/ethnic differences in COVID-19 infection continuing to emerge.3 Despite variation in the availability and quality of these data, disparities observed in COVID-19 have tracked closely with historical inequities in access to healthcare and discrimination within the healthcare system.4 Any approach to addressing these inequities must appreciate the intersection between social and medical vulnerabilities.
It is notable that healthcare systems serving the most vulnerable populations have borne the brunt of the economic toll of COVID-19. Hospitals in socioeconomically challenged areas lost millions of dollars due to the postponement of elective procedures and reallocation of most resources to COVID-related hospital admissions. Many community-based practices, already stretched in caring for medically and socially complex patients, had to shut their doors. These losses have left patients without the support of their network of healthcare and community service organizations—at the same time that many of them have also lost support for food and housing, employer-based health insurance, and in-person schooling and childcare.
The current circumstances due to the COVID-19 pandemic, therefore, require us to reconsider many aspects of both healthcare and the social safety net, including the reliance on financial penalties as a strategy to improve health quality, which ultimately has a disproportionate impact on communities of color.5 The present situation may also allow for the federal, state, and local governments, as well as health systems and payers, to make targeted investments in healthcare, public health, and community programs. For example, an increased healthcare system investment on preventive and primary care will be essential to reducing the chronic risk factors that underlie COVID-19 infection and death. Efforts by payers to reduce economic incentives for unnecessary elective procedures, while simultaneously providing incentives to increase the focus on preventive care, would further stimulate this effort. Although there is controversy over the inclusion of social risk in financial and value-based health system payment models, novel approaches to this problem (eg, consideration of improvement over achievement of static targets) may provide an opportunity for struggling health systems to invest in new strategies for underserved populations. Additionally, investing in a care system that allows racial, language, and cultural concordance between clinicians and patients would both promote a diverse workforce and improve quality of care. Health system equity will also depend upon bold policy advances such as expansion of Medicaid to all states, separation of health insurance from employment, and targeted government and health system investments around social risk (eg, food and housing insecurity). These programs will help vulnerable communities close the gap on disparities in health outcomes that have been so persistent.
Some of these specific concerns were addressed by the Coronavirus Aid, Relief, and Economic Security (CARES) Act that was implemented by the US Congress to address the broad needs of Americans during the acute crisis.6 The CARES Act provided supplementary funding to community health centers and healthcare systems caring for the uninsured. Cash assistance was provided to most US taxpayers along with financial support to those experiencing unemployment through July 31, 2020, measures that have yet to be extended. In addition to the CARES Act, policymakers proposed establishing a COVID-19 Racial and Ethnic Disparities Task Force Act to drive equitable recommendations and provide oversight to the nation’s response to COVID-19.7
While these measures were critical to the immediate pandemic response, future US congressional relief plans are needed to ensure equity remains a tenet of state and federal policy post COVID-19, particularly with respect to social determinants of health. Additional recommendations for federal relief include rent assistance for low-income families, eviction stoppages, and increased funding for short-term food insecurity. With respect to long-term goals, this is the time to address broader injustices, such as lack of affordable housing, lack of a sensible national strategy around food security, and a lack of equitable educational and justice systems. This moment also offers an opportunity to consider the best way to address the impact of centuries of structural racism. If we place equity at the center of policy implementation, we will certainly see downstream health consequences—ones that would begin to address the health disparities present long before the current pandemic.
FOSTERING COMMUNITY RESILIENCE
While national, state, and local responses to COVID-19 are required to bolster population health when we emerge from the COVID-19 crisis, a focus on community resilience is also needed. Community resilience, or the ability to prevent, withstand, and mitigate the stress of a disaster like COVID-19, requires integration of emergency preparedness practices into community disaster programs, with ongoing efforts to mitigate disparities in chronic disease management. A framework for community resilience includes (1) engaging with communities in planning, response, and post–COVID-19 recovery, (2) ensuring communities have access to high quality, culturally concordant health and social services, and (3) developing robust community networks to mobilize individuals, community services, and public health infrastructure in times of emergency.8
After seeing the devastating effects of Hurricane Katrina in 2005, researchers, public health officials, and community leaders founded the Los Angeles County Community Disaster Resilience (LACCDR) project. Through this collaborative effort, the LACCDR established partnerships across 16 communities to foster community resilience during health emergencies against the backdrop of daily chronic stressors such as violence, segregation, poverty, and homelessness.8 A model such as this to improve health systems and public health integration post-COVID will support health provisions and help build trust in communities wherein there is a high distrust of the healthcare system. Engaging with community partners early to ensure that its members have access to basic needs (eg, food, water, shelter), public health needs (eg, timely information, personal protective equipment such as face coverings and cleaning supplies), and affordable testing and vaccination will help prevent disparities that could affect the most vulnerable in future phases of the COVID-19 crisis.
PROMOTING EQUITY AS A SUSTAINABLE ECONOMIC STRATEGY
Over 40 million Americans were seeking unemployment benefits at the peak of the economic repercussions of the COVID-19 pandemic. Unfortunately, low-income, rural, and minority communities disproportionately experienced this economic shock. Given the relationship between wealth and health, successfully achieving equity post-COVID-19 will require deeper financial investments in underserved communities.9 Healthcare organizations, which represent 18% of the United States gross domestic product and employ nearly 9% of all working individuals, are uniquely positioned to have a direct influence on this strategy.
One equity-based strategy is for healthcare institutions to pursue an anchor mission. Anchor missions have increased a health system’s investment in social services, including providing housing and food resources.10 Additionally, hospitals such as Brigham and Women’s, Boston Children’s Hospital, and Bon Secours Health System, are working with a diverse group of entrepreneurs to create jobs and build wealth in underserved communities by employing local and minority-owned businesses to support critical supply chain purchasing decisions regarding food, maintenance, and construction projects.11 These local and inclusive hiring and procurement measures can be bolstered by continued place-based investments by all health system leaders in vulnerable communities.
CONCLUSION
Since the first enslaved Africans were brought to America over 400 years ago, racial and ethnic minorities have experienced struggle and triumph, sadness and joy. The bonds of a long legacy of discrimination are so deep that we must be intentional in our pursuit of equity—during and beyond the COVID-19 pandemic. Placing equity at the center of healthcare system practice and policy implementation, fostering community resilience and emergency preparedness, and prioritizing equity in economic strategic planning are key steps toward addressing the population-level inequities exposed by the COVID-19 pandemic. As the once touted “great equalizer” rages on, we must remember that we are all jointly affected by the distress caused by the novel coronavirus and we also must be more aware than ever of our interconnectedness. We can use this time of pandemic to fight more than ever to ensure that all populations can enjoy just and optimal health.
Acknowledgments
The authors would like to thank Dr Denise Polit for her review of this manuscript.
- Williams DR, Cooper LA. COVID–19 and health equity–a new kind of “herd immunity”. JAMA. 2020;323(24):2478-2480. https://doi.org/10.1001/jama.2020.8051
- Packer G. America’s plastic hour is upon us. The Atlantic. October 2020. Accessed September 28, 2020. https://www.theatlantic.com/magazine/archive/2020/10/make-america-again/615478/
- Gross CP, Essien UR, Pasha S, Gross JR, Wang SY, Nunez-Smith M. Racial and ethnic disparities in population-level Covid-19 mortality. J Gen Intern Med. 2020;35(10):3097-3099. https://doi.org/10.1007/s11606-020-06081-w
- Institute of Medicine (US) Committee on Understanding and Eliminating Racial and Ethnic Disparities in Health Care, Smedley BD, Stith AY, Nelson AR, eds. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. National Academies Press (US); 2003. https://doi.org/10.17226/12875
- Zuckerman RB, Joynt Maddox KE, Sheingold SH, Chen LM, Epstein AM. Effect of a hospital-wide measure on the readmissions reduction program. N Engl J Med. 2017;377(16):1551-1558. https://doi.org/10.1056/nejmsa1701791
- Cochrane E. House passes relief for small businesses and aid for hospitals and testing. New York Times. April 23, 2020. Accessed May 21, 2020. https://www.nytimes.com/2020/04/23/us/politics/house-passes-relief-for-small-businesses-and-aid-for-hospitals-and-testing.html
- Harris announces legislation to establish task force to combat racial and ethnic disparities in COVID-19. News release. Kamala D. Harris US Senator for California; April 30, 2020. Accessed May 21, 2020. https://www.harris.senate.gov/news/press-releases/harris-announces-legislation-to-establish-task-force-to-combat-racial-and-ethnic-disparities-in-covid-19
- Chandra A, Williams M, Plough A, et al. Getting actionable about community resilience: the Los Angeles County Community Disaster Resilience project. Am J Public Health. 2013;103(7):1181-1189. https://doi.org/10.2105/ajph.2013.301270
- Rawshani A, Svensson AM, Zethelius B, Eliasson B, Rosengren A, Gudbjörnsdottir S. Association between socioeconomic status and mortality, cardiovascular disease, and cancer in patients with type 2 diabetes. JAMA Intern Med. 2016;176(8):1146-1154. https://doi.org/10.1001/jamainternmed.2016.2940
- Horwitz LI, Chang C, Arcilla HN, Knickman JR. Quantifying health systems’ investment in social determinants of health, by sector, 2017-19. Health Aff (Millwood). 2020;39(2):192-198. https://doi.org/10.1377/hlthaff.2019.01246
- Nanos J. Diverse, locally owned food start-ups make the menus at Harvard, UMass, and BC. Boston Globe. January 24, 2020. Accessed September 28, 2020. https://www.bostonglobe.com/business/2020/01/24/diverse-locally-owned-food-start-ups-make-menus-harvard-umass-and/WwJFew6KVgXu1NyIK1BNqI/story.html
- Williams DR, Cooper LA. COVID–19 and health equity–a new kind of “herd immunity”. JAMA. 2020;323(24):2478-2480. https://doi.org/10.1001/jama.2020.8051
- Packer G. America’s plastic hour is upon us. The Atlantic. October 2020. Accessed September 28, 2020. https://www.theatlantic.com/magazine/archive/2020/10/make-america-again/615478/
- Gross CP, Essien UR, Pasha S, Gross JR, Wang SY, Nunez-Smith M. Racial and ethnic disparities in population-level Covid-19 mortality. J Gen Intern Med. 2020;35(10):3097-3099. https://doi.org/10.1007/s11606-020-06081-w
- Institute of Medicine (US) Committee on Understanding and Eliminating Racial and Ethnic Disparities in Health Care, Smedley BD, Stith AY, Nelson AR, eds. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. National Academies Press (US); 2003. https://doi.org/10.17226/12875
- Zuckerman RB, Joynt Maddox KE, Sheingold SH, Chen LM, Epstein AM. Effect of a hospital-wide measure on the readmissions reduction program. N Engl J Med. 2017;377(16):1551-1558. https://doi.org/10.1056/nejmsa1701791
- Cochrane E. House passes relief for small businesses and aid for hospitals and testing. New York Times. April 23, 2020. Accessed May 21, 2020. https://www.nytimes.com/2020/04/23/us/politics/house-passes-relief-for-small-businesses-and-aid-for-hospitals-and-testing.html
- Harris announces legislation to establish task force to combat racial and ethnic disparities in COVID-19. News release. Kamala D. Harris US Senator for California; April 30, 2020. Accessed May 21, 2020. https://www.harris.senate.gov/news/press-releases/harris-announces-legislation-to-establish-task-force-to-combat-racial-and-ethnic-disparities-in-covid-19
- Chandra A, Williams M, Plough A, et al. Getting actionable about community resilience: the Los Angeles County Community Disaster Resilience project. Am J Public Health. 2013;103(7):1181-1189. https://doi.org/10.2105/ajph.2013.301270
- Rawshani A, Svensson AM, Zethelius B, Eliasson B, Rosengren A, Gudbjörnsdottir S. Association between socioeconomic status and mortality, cardiovascular disease, and cancer in patients with type 2 diabetes. JAMA Intern Med. 2016;176(8):1146-1154. https://doi.org/10.1001/jamainternmed.2016.2940
- Horwitz LI, Chang C, Arcilla HN, Knickman JR. Quantifying health systems’ investment in social determinants of health, by sector, 2017-19. Health Aff (Millwood). 2020;39(2):192-198. https://doi.org/10.1377/hlthaff.2019.01246
- Nanos J. Diverse, locally owned food start-ups make the menus at Harvard, UMass, and BC. Boston Globe. January 24, 2020. Accessed September 28, 2020. https://www.bostonglobe.com/business/2020/01/24/diverse-locally-owned-food-start-ups-make-menus-harvard-umass-and/WwJFew6KVgXu1NyIK1BNqI/story.html
© 2021 Society of Hospital Medicine
Email: [email protected].
Language Barriers, Equity, and COVID-19: The Impact of a Novel Spanish Language Care Group
Our knowledge of how natural catastrophes affect vulnerable populations should have helped us anticipate how coronavirus disease 2019 (COVID-19) would strike the United States. This disaster has followed the well-heeled path of its predecessors, predictably bending to the influence of social determinants of health,1 structural inequality, and limited access to healthcare. Communities of color were hit early, hit hard,2 and yet again, became our nation’s canary in the coal mine. Hospitals across the country have had a front seat to this novel coronavirus’ disproportionate effect across the diverse communities we serve. Several of the cities and neighborhoods adjacent to our hospital are home to the area’s highest density of limited English proficient (LEP), immigrant, Spanish-speaking individuals.3,4 Our neighbors in these areas are more likely to have lower socioeconomic status, live in crowded housing, work in service industries deemed to be essential, and depend on shared and mass transit to get to work.5,6 As became clear, many in these communities could not work from home, get groceries delivered, or adequately social distance; these were pandemic luxuries afforded to other, more affluent areas.7
THE COVID-19 SURGE
In the weeks between March 25, 2020, and April 13, 2020, the Massachusetts General Hospital in Boston entered a COVID-19 surge now familiar to hospitals across the world. Like our peer institutions, we made broad and creative structural changes to inpatient services to meet the surge and we followed the numbers with anticipation. Over that 2-week period, we indeed saw the COVID-19–positive inpatient population swell as we had feared. However, with each page from the Emergency Department a disturbing trend was borne out:
ADMIT: 53-year-old Spanish-speaker with tachypnea.
ADMIT: 57-year-old factory worker, Spanish-speaking, sick for 10 days, intubated in the ED.
ADMIT: 58-year-old bodega employee, Spanish-speaking, febrile and breathless.
It buzzed across the medical floors and intensive care units: “What is going on in our Spanish speaking neighborhoods?” In fact, our shared anecdotal view was soon confirmed by admission statistics. Over the interval that our total COVID-19 census alarmingly rose sevenfold, the LEP Spanish-speaking census traced a striking curve, increasing nearly 20 times, to constitute over 40% of all COVID-19 patients (Figure). These communities were bearing a disproportionate share of the local burden of the pandemic.
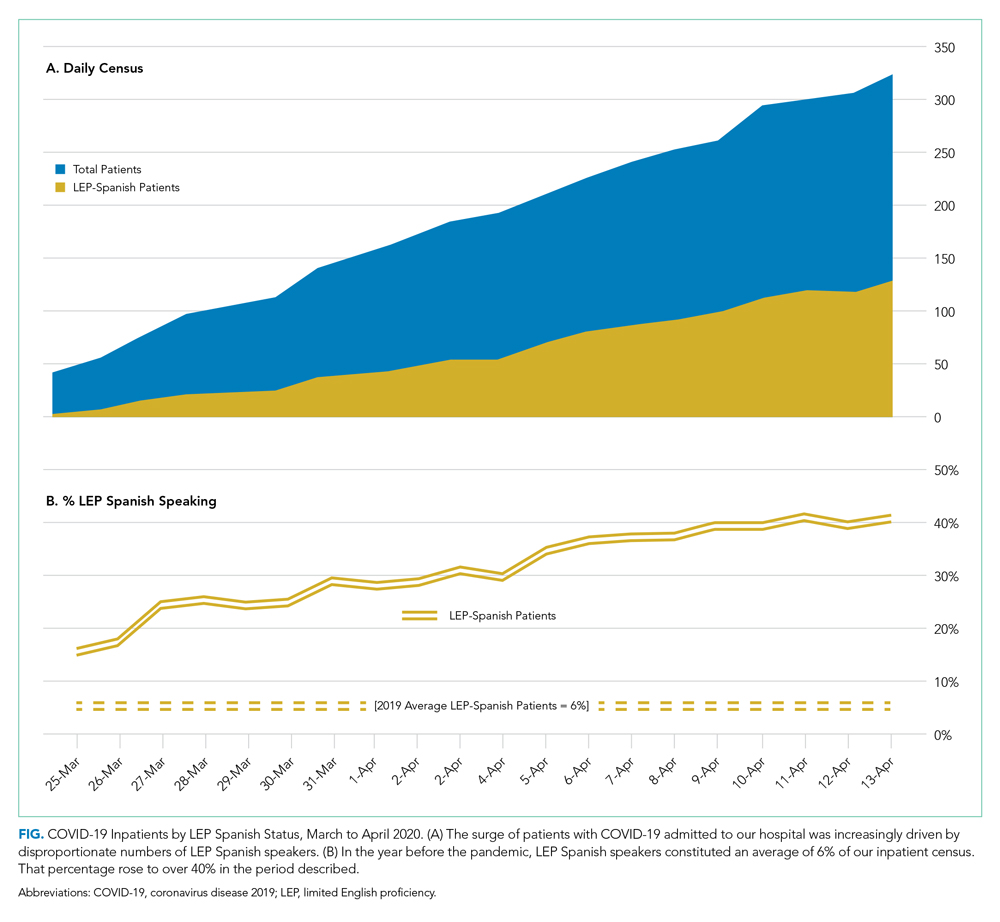
There is consensus in the health care community about the impact of LEP on quality of care, and how, if unaddressed, significant disparities emerge.8 In fact, there is a broadly accepted professional,9 ethical,10 and legal11,12 imperative for hospitals to address the language needs of LEP patients using interpreter services. However, clinicians often feel forced to rely on their own limited language skills to bridge the communication divide, especially in time-limited, critical situations.13 And regrettably, the highly problematic strategy of relying upon family members to aid with communication is still commonly used. The ideal approach, however, is to invest in developing care models that recognize language as an asset and leverage the skills of multilingual clinicians who care for patients in their own language, in a culturally and linguistically competent way.14 It is not surprising that, when clinicians and patients communicate in the same language, there is demonstrably improved adherence to treatment plans,15 increased patient insight into health conditions,16 and improved delivery of health education.17
FORMATION OF THE SPANISH LANGUAGE CARE GROUP
COVID-19 created unique challenges to our interpreter services. The overwhelming number of LEP Spanish-speaking patients made it difficult for our existing interpreter staff to provide in-person translation. Virtual interpreter services were always available; however, using telephone interpretation in full personal protective equipment with patients who were already isolated and dealing with a scary diagnosis did not feel adequate to the need. In response to what we were seeing, on April 13, 2020, the idea emerged from the Chief Equity and Inclusion Officer, a native Spanish speaker, to assemble a team of native Spanish-speaking doctors, deploying them to assist in the clinical care of those LEP Spanish-speaking patients admitted with COVID-19. Out of this idea grew a creative and novel care delivery model, fashioned to prioritize culturally and linguistically competent care. It was deployed a few days later as the Spanish Language Care Group (SLCG). The belief was that this group’s members were uniquely equipped to work directly with existing frontline teams on the floors, intensive care units and the emergency department. As doctors, they were able to act as extensions of those teams, independently carrying out patient-facing clinical tasks, in Spanish, on an ad hoc basis. They took on history taking, procedural consents, clinical updates, discharge instructions, serious illness conversations and family meetings. They comforted and educated the frightened, connected with families, and unearthed relevant patient history that would have otherwise gone unnoticed. In many cases the SLCG member was the main figure communicating with patients as their clinical status deteriorated, as they were intubated, as they faced their worst fears about COVID-19.
At the time the group was assembled, each SLCG physician was verified as Qualified Bilingual Staff, already clinically credentialled at the hospital, and ready to volunteer to meet the need on the medicine COVID surge services. They practiced in virtually every division and department, including Anesthesia, Cardiology, Dermatology, Emergency Medicine, Gastroenterology, General Medicine, Neurology, Pediatrics, Psychiatry, and Radiology. With the assistance of leadership in Hospital Medicine, this team was rapidly deployed to inpatient teams to assist with the clinical care of COVID-19 patients. In total, 51 physicians—representing 14 countries of origin—participated in the effort, and their titles ranged from intern to full professor. Fourteen of them were formally deployed in the COVID surge context with approval of their departmental and divisional leadership. With such a robust response and institutional support, the SLCG was able to provide 24-hour coverage in support of the Medicine teams. During the peak of this hospital’s COVID surge, seven SLCG members were deployed daily 7
For those patients in their most vulnerable moments, the impact of the SLCG’s work is hard to overestimate, and it has also been measured by overwhelmingly positive feedback from surge care teams: “The quality of care we provided today would have been impossible without [the SLCG]. I’m so grateful and was nearly moved to tears realizing how stunted our relationships with these patients have been due to language barrier.” Another team said that the SLCG doctor was able to “care for the patient in the same way I would have if I could speak Spanish” and “it is like day and night.”After the spring 2020 surge of COVID-19, procedural work resumed, so the SLCG doctors—many of whose usual clinical activity was suspended by the pandemic—returned to their proper perch on the organization chart. But as they reflect on their experience with the group, they report that it stirred a strong and very personal sense of purpose and vocation. Should a subsequent surge of COVID-19 occur, they are committed to building on the foundation that they have laid.
DEPLOYING A LANGUAGE CARE GROUP TEAM
For hospitals that may consider deploying a team such as the SLCG, we can offer a number of concrete actions and policy recommendations. First, in preparation for the COVID surge we identified hospital clinicians with multilingual skills through the deployment of a multilingual registry. Such a registry is critical to understanding which clinicians among existing staff have these skills and who can be approached to join the team. Second, the inpatient medicine surge leadership team at our hospital, immediately recognizing the importance of this effort, developed a staffing strategy to integrate the SLCG into the institutional surge response. The benefit that the team offers needs to be made clear to those at the highest levels of operations and planning. Third, a strong and well-established Center for Diversity and Inclusion, and its leadership, helped facilitate our group’s staffing and organization. For hospitals looking to embrace the strength that their diversity-oriented recruitment efforts have afforded them, we recommend creating a centralized space in which professional relationships can grow and deepen, diverse perspectives can be explored, and embedded cultural and language skills can be championed.
The US healthcare system has much to learn from this phase of the COVID-19 era. Our experience with the Spanish Language Care Group has highlighted the value of language-concordant care, the power of cultural and linguistic competency, and the resiliency that diversity brings to a hospital’s professional staff. Our urgent response to COVID-19 has unroofed a long-simmering challenge: the detriment to care that arises when language becomes an obstacle. We are bringing a new focus to this issue and learning to view it through an equity lens. This is lending new energy to an ongoing conversation about how this hospital thinks about diversity, equity, and healthcare access in these pandemic times and into the hoped-for beyond.
Acknowledgments
The authors wish to express their profound gratitude to the members of the Spanish Language Care Group who brought such humanity and professionalism to the care of our patients during a uniquely vulnerable time.
- Social Determinants of Health. World Health Organization. Accessed November 10, 2020. https://www.who.int/health-topics/social-determinants-of-health#tab=tab_1
- Buchanan L, Patel JK, Rosenthal BM, Singhvi A. A month of coronavirus in New York City: see the hardest-hit areas. New York Times. April 1, 2020. Accessed November 10, 2020. https://www.nytimes.com/interactive/2020/04/01/nyregion/nyc-coronavirus-cases-map.html
- QuickFacts: Chelsea city, Massachusetts. United States Census Bureau. Accessed November 10, 2020. https://www.census.gov/quickfacts/chelseacitymassachusetts
- Boston by the Numbers 2018. Research Division, Boston Planning & Development Agency. September 2018. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/3e8bfacf-27c1-4b55-adee-29c5d79f4a38
- Demographic Profile of Adult Limited English Speakers in Massachusetts. Research Division, Boston Planning & Development Agency. February 2019. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/dfe1117a-af16-4257-b0f5-1d95dbd575fe
- Boston in Context: Neighborhoods 2012-2016 American Community Survey. Research Division, Boston Planning & Development Agency. March 2018. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/55f2d86f-eccf-4f68-8d8d-c631fefb0161
- Canipe C. The social distancing of America. Reuters Graphics. April 2, 2020. Accessed November 10, 2020. https://graphics.reuters.com/HEALTH-CORONAVIRUS/USA/qmypmkmwpra/
- Betancourt J, Green AR, Carrillo JE, Park ER. Cultural competency and health care disparities: key perspectives and trends. Health Aff (Millwood). 2005;24(2):499-505. https://doi.org/10.1377/hlthaff.24.2.499
- Racial and Ethnic Disparities in Health Care, Updated 2010. American College of Physicians; 2010. Accessed November 10, 2020. https://www.acponline.org/system/files/documents/advocacy/current_policy_papers/assets/racial_disparities.pdf
- 1.1.3 Patient rights. In: Chapter 1: Opinions on Patient-Physician Relationships. Code of Medical Ethics. American Medical Association; 2016. https://www.ama-assn.org/sites/default/files/media-browser/code-of-medical-ethics-chapter-1.pdf
- Title VI of the Civil Rights Act of 1964, as amended, 42 USC §2000d et seq. July 2, 1964.
- Patient Protection and Affordable Care Act of 2010, Pub L No. 111-148, 124 Stat 119 (2010) §1557.
- Regenstein M, Andres E, Wynia MK. Appropriate use of non-English-language skills in clinical care. JAMA. 2013;309(2):145-146. https://doi.org/10.1001/jama.2012.116984
- Ngo-Metzger Q, Sorkin DH, Phillips RS, et al. Providing high-quality care for limited English proficient patients: the importance of language concordance and interpreter use. J Gen Intern Med. 2007;22(Suppl) 2:324-330.
- Manson A. Language concordance as a determinant of patient compliance and emergency room use in patients with asthma. Med Care. 1988;26(12):1119-1128. https://doi.org/10.1097/00005650-198812000-00003
- Seijo R, Gomez H, Garcia M, Shelton D. Acculturation, access to care, and use of preventive services by Hispanics: findings from HANES 1982-84. Am J Public Health. 1991;80(suppl):11-19
- Shapiro J, Saltzer EB. Cross-cultural aspects of physician-patient communications patterns. Urban Health. 1981;10(10):10-15.
Our knowledge of how natural catastrophes affect vulnerable populations should have helped us anticipate how coronavirus disease 2019 (COVID-19) would strike the United States. This disaster has followed the well-heeled path of its predecessors, predictably bending to the influence of social determinants of health,1 structural inequality, and limited access to healthcare. Communities of color were hit early, hit hard,2 and yet again, became our nation’s canary in the coal mine. Hospitals across the country have had a front seat to this novel coronavirus’ disproportionate effect across the diverse communities we serve. Several of the cities and neighborhoods adjacent to our hospital are home to the area’s highest density of limited English proficient (LEP), immigrant, Spanish-speaking individuals.3,4 Our neighbors in these areas are more likely to have lower socioeconomic status, live in crowded housing, work in service industries deemed to be essential, and depend on shared and mass transit to get to work.5,6 As became clear, many in these communities could not work from home, get groceries delivered, or adequately social distance; these were pandemic luxuries afforded to other, more affluent areas.7
THE COVID-19 SURGE
In the weeks between March 25, 2020, and April 13, 2020, the Massachusetts General Hospital in Boston entered a COVID-19 surge now familiar to hospitals across the world. Like our peer institutions, we made broad and creative structural changes to inpatient services to meet the surge and we followed the numbers with anticipation. Over that 2-week period, we indeed saw the COVID-19–positive inpatient population swell as we had feared. However, with each page from the Emergency Department a disturbing trend was borne out:
ADMIT: 53-year-old Spanish-speaker with tachypnea.
ADMIT: 57-year-old factory worker, Spanish-speaking, sick for 10 days, intubated in the ED.
ADMIT: 58-year-old bodega employee, Spanish-speaking, febrile and breathless.
It buzzed across the medical floors and intensive care units: “What is going on in our Spanish speaking neighborhoods?” In fact, our shared anecdotal view was soon confirmed by admission statistics. Over the interval that our total COVID-19 census alarmingly rose sevenfold, the LEP Spanish-speaking census traced a striking curve, increasing nearly 20 times, to constitute over 40% of all COVID-19 patients (Figure). These communities were bearing a disproportionate share of the local burden of the pandemic.

There is consensus in the health care community about the impact of LEP on quality of care, and how, if unaddressed, significant disparities emerge.8 In fact, there is a broadly accepted professional,9 ethical,10 and legal11,12 imperative for hospitals to address the language needs of LEP patients using interpreter services. However, clinicians often feel forced to rely on their own limited language skills to bridge the communication divide, especially in time-limited, critical situations.13 And regrettably, the highly problematic strategy of relying upon family members to aid with communication is still commonly used. The ideal approach, however, is to invest in developing care models that recognize language as an asset and leverage the skills of multilingual clinicians who care for patients in their own language, in a culturally and linguistically competent way.14 It is not surprising that, when clinicians and patients communicate in the same language, there is demonstrably improved adherence to treatment plans,15 increased patient insight into health conditions,16 and improved delivery of health education.17
FORMATION OF THE SPANISH LANGUAGE CARE GROUP
COVID-19 created unique challenges to our interpreter services. The overwhelming number of LEP Spanish-speaking patients made it difficult for our existing interpreter staff to provide in-person translation. Virtual interpreter services were always available; however, using telephone interpretation in full personal protective equipment with patients who were already isolated and dealing with a scary diagnosis did not feel adequate to the need. In response to what we were seeing, on April 13, 2020, the idea emerged from the Chief Equity and Inclusion Officer, a native Spanish speaker, to assemble a team of native Spanish-speaking doctors, deploying them to assist in the clinical care of those LEP Spanish-speaking patients admitted with COVID-19. Out of this idea grew a creative and novel care delivery model, fashioned to prioritize culturally and linguistically competent care. It was deployed a few days later as the Spanish Language Care Group (SLCG). The belief was that this group’s members were uniquely equipped to work directly with existing frontline teams on the floors, intensive care units and the emergency department. As doctors, they were able to act as extensions of those teams, independently carrying out patient-facing clinical tasks, in Spanish, on an ad hoc basis. They took on history taking, procedural consents, clinical updates, discharge instructions, serious illness conversations and family meetings. They comforted and educated the frightened, connected with families, and unearthed relevant patient history that would have otherwise gone unnoticed. In many cases the SLCG member was the main figure communicating with patients as their clinical status deteriorated, as they were intubated, as they faced their worst fears about COVID-19.
At the time the group was assembled, each SLCG physician was verified as Qualified Bilingual Staff, already clinically credentialled at the hospital, and ready to volunteer to meet the need on the medicine COVID surge services. They practiced in virtually every division and department, including Anesthesia, Cardiology, Dermatology, Emergency Medicine, Gastroenterology, General Medicine, Neurology, Pediatrics, Psychiatry, and Radiology. With the assistance of leadership in Hospital Medicine, this team was rapidly deployed to inpatient teams to assist with the clinical care of COVID-19 patients. In total, 51 physicians—representing 14 countries of origin—participated in the effort, and their titles ranged from intern to full professor. Fourteen of them were formally deployed in the COVID surge context with approval of their departmental and divisional leadership. With such a robust response and institutional support, the SLCG was able to provide 24-hour coverage in support of the Medicine teams. During the peak of this hospital’s COVID surge, seven SLCG members were deployed daily 7
For those patients in their most vulnerable moments, the impact of the SLCG’s work is hard to overestimate, and it has also been measured by overwhelmingly positive feedback from surge care teams: “The quality of care we provided today would have been impossible without [the SLCG]. I’m so grateful and was nearly moved to tears realizing how stunted our relationships with these patients have been due to language barrier.” Another team said that the SLCG doctor was able to “care for the patient in the same way I would have if I could speak Spanish” and “it is like day and night.”After the spring 2020 surge of COVID-19, procedural work resumed, so the SLCG doctors—many of whose usual clinical activity was suspended by the pandemic—returned to their proper perch on the organization chart. But as they reflect on their experience with the group, they report that it stirred a strong and very personal sense of purpose and vocation. Should a subsequent surge of COVID-19 occur, they are committed to building on the foundation that they have laid.
DEPLOYING A LANGUAGE CARE GROUP TEAM
For hospitals that may consider deploying a team such as the SLCG, we can offer a number of concrete actions and policy recommendations. First, in preparation for the COVID surge we identified hospital clinicians with multilingual skills through the deployment of a multilingual registry. Such a registry is critical to understanding which clinicians among existing staff have these skills and who can be approached to join the team. Second, the inpatient medicine surge leadership team at our hospital, immediately recognizing the importance of this effort, developed a staffing strategy to integrate the SLCG into the institutional surge response. The benefit that the team offers needs to be made clear to those at the highest levels of operations and planning. Third, a strong and well-established Center for Diversity and Inclusion, and its leadership, helped facilitate our group’s staffing and organization. For hospitals looking to embrace the strength that their diversity-oriented recruitment efforts have afforded them, we recommend creating a centralized space in which professional relationships can grow and deepen, diverse perspectives can be explored, and embedded cultural and language skills can be championed.
The US healthcare system has much to learn from this phase of the COVID-19 era. Our experience with the Spanish Language Care Group has highlighted the value of language-concordant care, the power of cultural and linguistic competency, and the resiliency that diversity brings to a hospital’s professional staff. Our urgent response to COVID-19 has unroofed a long-simmering challenge: the detriment to care that arises when language becomes an obstacle. We are bringing a new focus to this issue and learning to view it through an equity lens. This is lending new energy to an ongoing conversation about how this hospital thinks about diversity, equity, and healthcare access in these pandemic times and into the hoped-for beyond.
Acknowledgments
The authors wish to express their profound gratitude to the members of the Spanish Language Care Group who brought such humanity and professionalism to the care of our patients during a uniquely vulnerable time.
Our knowledge of how natural catastrophes affect vulnerable populations should have helped us anticipate how coronavirus disease 2019 (COVID-19) would strike the United States. This disaster has followed the well-heeled path of its predecessors, predictably bending to the influence of social determinants of health,1 structural inequality, and limited access to healthcare. Communities of color were hit early, hit hard,2 and yet again, became our nation’s canary in the coal mine. Hospitals across the country have had a front seat to this novel coronavirus’ disproportionate effect across the diverse communities we serve. Several of the cities and neighborhoods adjacent to our hospital are home to the area’s highest density of limited English proficient (LEP), immigrant, Spanish-speaking individuals.3,4 Our neighbors in these areas are more likely to have lower socioeconomic status, live in crowded housing, work in service industries deemed to be essential, and depend on shared and mass transit to get to work.5,6 As became clear, many in these communities could not work from home, get groceries delivered, or adequately social distance; these were pandemic luxuries afforded to other, more affluent areas.7
THE COVID-19 SURGE
In the weeks between March 25, 2020, and April 13, 2020, the Massachusetts General Hospital in Boston entered a COVID-19 surge now familiar to hospitals across the world. Like our peer institutions, we made broad and creative structural changes to inpatient services to meet the surge and we followed the numbers with anticipation. Over that 2-week period, we indeed saw the COVID-19–positive inpatient population swell as we had feared. However, with each page from the Emergency Department a disturbing trend was borne out:
ADMIT: 53-year-old Spanish-speaker with tachypnea.
ADMIT: 57-year-old factory worker, Spanish-speaking, sick for 10 days, intubated in the ED.
ADMIT: 58-year-old bodega employee, Spanish-speaking, febrile and breathless.
It buzzed across the medical floors and intensive care units: “What is going on in our Spanish speaking neighborhoods?” In fact, our shared anecdotal view was soon confirmed by admission statistics. Over the interval that our total COVID-19 census alarmingly rose sevenfold, the LEP Spanish-speaking census traced a striking curve, increasing nearly 20 times, to constitute over 40% of all COVID-19 patients (Figure). These communities were bearing a disproportionate share of the local burden of the pandemic.

There is consensus in the health care community about the impact of LEP on quality of care, and how, if unaddressed, significant disparities emerge.8 In fact, there is a broadly accepted professional,9 ethical,10 and legal11,12 imperative for hospitals to address the language needs of LEP patients using interpreter services. However, clinicians often feel forced to rely on their own limited language skills to bridge the communication divide, especially in time-limited, critical situations.13 And regrettably, the highly problematic strategy of relying upon family members to aid with communication is still commonly used. The ideal approach, however, is to invest in developing care models that recognize language as an asset and leverage the skills of multilingual clinicians who care for patients in their own language, in a culturally and linguistically competent way.14 It is not surprising that, when clinicians and patients communicate in the same language, there is demonstrably improved adherence to treatment plans,15 increased patient insight into health conditions,16 and improved delivery of health education.17
FORMATION OF THE SPANISH LANGUAGE CARE GROUP
COVID-19 created unique challenges to our interpreter services. The overwhelming number of LEP Spanish-speaking patients made it difficult for our existing interpreter staff to provide in-person translation. Virtual interpreter services were always available; however, using telephone interpretation in full personal protective equipment with patients who were already isolated and dealing with a scary diagnosis did not feel adequate to the need. In response to what we were seeing, on April 13, 2020, the idea emerged from the Chief Equity and Inclusion Officer, a native Spanish speaker, to assemble a team of native Spanish-speaking doctors, deploying them to assist in the clinical care of those LEP Spanish-speaking patients admitted with COVID-19. Out of this idea grew a creative and novel care delivery model, fashioned to prioritize culturally and linguistically competent care. It was deployed a few days later as the Spanish Language Care Group (SLCG). The belief was that this group’s members were uniquely equipped to work directly with existing frontline teams on the floors, intensive care units and the emergency department. As doctors, they were able to act as extensions of those teams, independently carrying out patient-facing clinical tasks, in Spanish, on an ad hoc basis. They took on history taking, procedural consents, clinical updates, discharge instructions, serious illness conversations and family meetings. They comforted and educated the frightened, connected with families, and unearthed relevant patient history that would have otherwise gone unnoticed. In many cases the SLCG member was the main figure communicating with patients as their clinical status deteriorated, as they were intubated, as they faced their worst fears about COVID-19.
At the time the group was assembled, each SLCG physician was verified as Qualified Bilingual Staff, already clinically credentialled at the hospital, and ready to volunteer to meet the need on the medicine COVID surge services. They practiced in virtually every division and department, including Anesthesia, Cardiology, Dermatology, Emergency Medicine, Gastroenterology, General Medicine, Neurology, Pediatrics, Psychiatry, and Radiology. With the assistance of leadership in Hospital Medicine, this team was rapidly deployed to inpatient teams to assist with the clinical care of COVID-19 patients. In total, 51 physicians—representing 14 countries of origin—participated in the effort, and their titles ranged from intern to full professor. Fourteen of them were formally deployed in the COVID surge context with approval of their departmental and divisional leadership. With such a robust response and institutional support, the SLCG was able to provide 24-hour coverage in support of the Medicine teams. During the peak of this hospital’s COVID surge, seven SLCG members were deployed daily 7
For those patients in their most vulnerable moments, the impact of the SLCG’s work is hard to overestimate, and it has also been measured by overwhelmingly positive feedback from surge care teams: “The quality of care we provided today would have been impossible without [the SLCG]. I’m so grateful and was nearly moved to tears realizing how stunted our relationships with these patients have been due to language barrier.” Another team said that the SLCG doctor was able to “care for the patient in the same way I would have if I could speak Spanish” and “it is like day and night.”After the spring 2020 surge of COVID-19, procedural work resumed, so the SLCG doctors—many of whose usual clinical activity was suspended by the pandemic—returned to their proper perch on the organization chart. But as they reflect on their experience with the group, they report that it stirred a strong and very personal sense of purpose and vocation. Should a subsequent surge of COVID-19 occur, they are committed to building on the foundation that they have laid.
DEPLOYING A LANGUAGE CARE GROUP TEAM
For hospitals that may consider deploying a team such as the SLCG, we can offer a number of concrete actions and policy recommendations. First, in preparation for the COVID surge we identified hospital clinicians with multilingual skills through the deployment of a multilingual registry. Such a registry is critical to understanding which clinicians among existing staff have these skills and who can be approached to join the team. Second, the inpatient medicine surge leadership team at our hospital, immediately recognizing the importance of this effort, developed a staffing strategy to integrate the SLCG into the institutional surge response. The benefit that the team offers needs to be made clear to those at the highest levels of operations and planning. Third, a strong and well-established Center for Diversity and Inclusion, and its leadership, helped facilitate our group’s staffing and organization. For hospitals looking to embrace the strength that their diversity-oriented recruitment efforts have afforded them, we recommend creating a centralized space in which professional relationships can grow and deepen, diverse perspectives can be explored, and embedded cultural and language skills can be championed.
The US healthcare system has much to learn from this phase of the COVID-19 era. Our experience with the Spanish Language Care Group has highlighted the value of language-concordant care, the power of cultural and linguistic competency, and the resiliency that diversity brings to a hospital’s professional staff. Our urgent response to COVID-19 has unroofed a long-simmering challenge: the detriment to care that arises when language becomes an obstacle. We are bringing a new focus to this issue and learning to view it through an equity lens. This is lending new energy to an ongoing conversation about how this hospital thinks about diversity, equity, and healthcare access in these pandemic times and into the hoped-for beyond.
Acknowledgments
The authors wish to express their profound gratitude to the members of the Spanish Language Care Group who brought such humanity and professionalism to the care of our patients during a uniquely vulnerable time.
- Social Determinants of Health. World Health Organization. Accessed November 10, 2020. https://www.who.int/health-topics/social-determinants-of-health#tab=tab_1
- Buchanan L, Patel JK, Rosenthal BM, Singhvi A. A month of coronavirus in New York City: see the hardest-hit areas. New York Times. April 1, 2020. Accessed November 10, 2020. https://www.nytimes.com/interactive/2020/04/01/nyregion/nyc-coronavirus-cases-map.html
- QuickFacts: Chelsea city, Massachusetts. United States Census Bureau. Accessed November 10, 2020. https://www.census.gov/quickfacts/chelseacitymassachusetts
- Boston by the Numbers 2018. Research Division, Boston Planning & Development Agency. September 2018. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/3e8bfacf-27c1-4b55-adee-29c5d79f4a38
- Demographic Profile of Adult Limited English Speakers in Massachusetts. Research Division, Boston Planning & Development Agency. February 2019. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/dfe1117a-af16-4257-b0f5-1d95dbd575fe
- Boston in Context: Neighborhoods 2012-2016 American Community Survey. Research Division, Boston Planning & Development Agency. March 2018. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/55f2d86f-eccf-4f68-8d8d-c631fefb0161
- Canipe C. The social distancing of America. Reuters Graphics. April 2, 2020. Accessed November 10, 2020. https://graphics.reuters.com/HEALTH-CORONAVIRUS/USA/qmypmkmwpra/
- Betancourt J, Green AR, Carrillo JE, Park ER. Cultural competency and health care disparities: key perspectives and trends. Health Aff (Millwood). 2005;24(2):499-505. https://doi.org/10.1377/hlthaff.24.2.499
- Racial and Ethnic Disparities in Health Care, Updated 2010. American College of Physicians; 2010. Accessed November 10, 2020. https://www.acponline.org/system/files/documents/advocacy/current_policy_papers/assets/racial_disparities.pdf
- 1.1.3 Patient rights. In: Chapter 1: Opinions on Patient-Physician Relationships. Code of Medical Ethics. American Medical Association; 2016. https://www.ama-assn.org/sites/default/files/media-browser/code-of-medical-ethics-chapter-1.pdf
- Title VI of the Civil Rights Act of 1964, as amended, 42 USC §2000d et seq. July 2, 1964.
- Patient Protection and Affordable Care Act of 2010, Pub L No. 111-148, 124 Stat 119 (2010) §1557.
- Regenstein M, Andres E, Wynia MK. Appropriate use of non-English-language skills in clinical care. JAMA. 2013;309(2):145-146. https://doi.org/10.1001/jama.2012.116984
- Ngo-Metzger Q, Sorkin DH, Phillips RS, et al. Providing high-quality care for limited English proficient patients: the importance of language concordance and interpreter use. J Gen Intern Med. 2007;22(Suppl) 2:324-330.
- Manson A. Language concordance as a determinant of patient compliance and emergency room use in patients with asthma. Med Care. 1988;26(12):1119-1128. https://doi.org/10.1097/00005650-198812000-00003
- Seijo R, Gomez H, Garcia M, Shelton D. Acculturation, access to care, and use of preventive services by Hispanics: findings from HANES 1982-84. Am J Public Health. 1991;80(suppl):11-19
- Shapiro J, Saltzer EB. Cross-cultural aspects of physician-patient communications patterns. Urban Health. 1981;10(10):10-15.
- Social Determinants of Health. World Health Organization. Accessed November 10, 2020. https://www.who.int/health-topics/social-determinants-of-health#tab=tab_1
- Buchanan L, Patel JK, Rosenthal BM, Singhvi A. A month of coronavirus in New York City: see the hardest-hit areas. New York Times. April 1, 2020. Accessed November 10, 2020. https://www.nytimes.com/interactive/2020/04/01/nyregion/nyc-coronavirus-cases-map.html
- QuickFacts: Chelsea city, Massachusetts. United States Census Bureau. Accessed November 10, 2020. https://www.census.gov/quickfacts/chelseacitymassachusetts
- Boston by the Numbers 2018. Research Division, Boston Planning & Development Agency. September 2018. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/3e8bfacf-27c1-4b55-adee-29c5d79f4a38
- Demographic Profile of Adult Limited English Speakers in Massachusetts. Research Division, Boston Planning & Development Agency. February 2019. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/dfe1117a-af16-4257-b0f5-1d95dbd575fe
- Boston in Context: Neighborhoods 2012-2016 American Community Survey. Research Division, Boston Planning & Development Agency. March 2018. Accessed November 10, 2020. http://www.bostonplans.org/getattachment/55f2d86f-eccf-4f68-8d8d-c631fefb0161
- Canipe C. The social distancing of America. Reuters Graphics. April 2, 2020. Accessed November 10, 2020. https://graphics.reuters.com/HEALTH-CORONAVIRUS/USA/qmypmkmwpra/
- Betancourt J, Green AR, Carrillo JE, Park ER. Cultural competency and health care disparities: key perspectives and trends. Health Aff (Millwood). 2005;24(2):499-505. https://doi.org/10.1377/hlthaff.24.2.499
- Racial and Ethnic Disparities in Health Care, Updated 2010. American College of Physicians; 2010. Accessed November 10, 2020. https://www.acponline.org/system/files/documents/advocacy/current_policy_papers/assets/racial_disparities.pdf
- 1.1.3 Patient rights. In: Chapter 1: Opinions on Patient-Physician Relationships. Code of Medical Ethics. American Medical Association; 2016. https://www.ama-assn.org/sites/default/files/media-browser/code-of-medical-ethics-chapter-1.pdf
- Title VI of the Civil Rights Act of 1964, as amended, 42 USC §2000d et seq. July 2, 1964.
- Patient Protection and Affordable Care Act of 2010, Pub L No. 111-148, 124 Stat 119 (2010) §1557.
- Regenstein M, Andres E, Wynia MK. Appropriate use of non-English-language skills in clinical care. JAMA. 2013;309(2):145-146. https://doi.org/10.1001/jama.2012.116984
- Ngo-Metzger Q, Sorkin DH, Phillips RS, et al. Providing high-quality care for limited English proficient patients: the importance of language concordance and interpreter use. J Gen Intern Med. 2007;22(Suppl) 2:324-330.
- Manson A. Language concordance as a determinant of patient compliance and emergency room use in patients with asthma. Med Care. 1988;26(12):1119-1128. https://doi.org/10.1097/00005650-198812000-00003
- Seijo R, Gomez H, Garcia M, Shelton D. Acculturation, access to care, and use of preventive services by Hispanics: findings from HANES 1982-84. Am J Public Health. 1991;80(suppl):11-19
- Shapiro J, Saltzer EB. Cross-cultural aspects of physician-patient communications patterns. Urban Health. 1981;10(10):10-15.
© 2020 Society of Hospital Medicine
Email: [email protected]; Telephone: 617-898-7722; Twitter: @StevenKnuesel
Racial Health Disparities, COVID-19, and a Way Forward for US Health Systems
The coronavirus disease 2019 (COVID-19) pandemic highlights long-standing inequities in health along racial/ethnic lines in the United States. Black, Hispanic, and Indigenous people have been disproportionately affected during the pandemic. For example, the age-adjusted mortality rate among Black people with COVID-19 is 3.4 times as high as that of White people.1
Structural racism shapes social forces, institutions, and ideologies that generate and reinforce racial inequities across different aspects of life. In this perspective, we discuss how, in the COVID-19 context, structural racism shapes access to and quality of care, as well as socioeconomic and health status. We offer guidance to health systems and healthcare providers on addressing health inequities.
HEALTHCARE QUALITY AND ACCESS
Disparities in access to and quality of care contribute to racial health disparities. At the onset of the COVID-19 pandemic in the United States, guidelines for COVID-19 testing were restrictive, only investigating those who had symptoms and had recently traveled to Wuhan, China, or had contact with someone who may have had the virus.2 News reports show disparities in access to testing, with testing sites favoring wealthier, Whiter communities, a feature of racial residential segregation.3 Residential segregation has also contributed to a concentration of closures among urban public hospitals, affecting access to care.4 In New York City (NYC) and Boston, early hotspots of the pandemic, Black and Hispanic patients and underinsured/uninsured patients were significantly less likely to access care from academic medical centers (AMCs) compared with White, privately insured patients.5 AMCs boast greater resources, and inequalities produced by this segregated system of care are often exacerbated by governmental allocation of resources. For instance, NYC’s public hospitals care for the city’s low-income residents (who are disproportionately insured by Medicaid), yet received far less federal aid from the Provider Relief Fund COVID-19 High Impact Payments, which favored larger, private hospitals in Manhattan. These public hospitals, however, face looming Medicaid cuts.6 Similarly, the federal government delayed the release of funds to health centers located on Native American reservations, adversely affecting the Indian Health Service’s preparedness to face the pandemic.7 In tandem with the effects of residential segregation, these data highlight the tiered nature of the US healthcare system, a structure that significantly impacts the quality of care patients receive along racial and socioeconomic lines. Furthermore, studies have documented racial disparities in the provision of advanced therapies: in the case of predicting algorithms that identify patients with complex illnesses, reliance on cost (thus, previous utilization data) rather than actual illness means that only 17.5% of Black patients receive additional help.8
SOCIOECONOMIC STATUS, OCCUPATIONAL AND RESIDENTIAL RISK
Healthcare alone does not explain the observed disparities. The disproportionately high risk of contracting the SARS-CoV-2 virus among Black, Hispanic and Indigenous people can be explained by factors that render physical distancing a luxury. First, in terms of occupational hazards, only 1 in 5 Black and 1 in 6 Hispanic workers can work remotely compared with 1 in 3 White workers. Additionally, Black and Hispanic workers are more likely to have jobs classified as critical in industries such as food retail, hospitality, and public transit. In NYC, Metropolitan Transportation Authority (MTA) employees reported using their own masks and home disinfectant at work, only to be reprimanded. By April 8, 2020, at least 41 MTA workers had died of COVID-19, and more than 6,000 were ill or self-quarantining, resulting in a transit crisis with increasingly long wait times and crowded subway platforms.9 Jason Hargrove, a Black bus driver in Detroit, shared a video underscoring the dangers of his work in which he says, “We’re out here as public workers, doing our job…but for you to get on the bus and stand on the bus, and cough several times without covering up your mouth . . . in the middle of a pandemic…some folks don’t care.” He died of COVID-19 complications 11 days after sharing his video.10 Such conditions likely also increased riders’ risk of contracting COVID-19. And while in aggregate, essential workers in healthcare receive more personal protective equipment (PPE) than those in other occupations, within NYC hospitals, the rationing of PPE was such that low-wage, nonmedical workers (79% of whom are Black or Hispanic) were given less PPE or none at all compared with nurses and physicians.11
Beyond occupational hazards, Black and Hispanic people are more likely to live in multigenerational homes, an identified risk factor of COVID-19 infection.12 Furthermore, Black and Hispanic people are overrepresented among homeless people as well as among those incarcerated. These social conditions, all products of structural racism, substantially and adversely affect the health status of Black, Hispanic, and Indigenous people, especially as it relates to comorbidities associated with higher COVID-19 mortality.
DISPARITIES IN HEALTH STATUS
Black people are disproportionately represented among COVID-19 patients requiring hospitalization, consistent with more severe disease or delayed presentation. For instance, among a cohort of 3,626 patients in a health system in Louisiana, 76.9% of COVID-19 patients hospitalized and 70.6% of those who died were Black, even though Black people comprise only 31% of this health system’s patient population.13 Conditions associated with COVID-19 mortality include heart failure, obesity, and chronic obstructive pulmonary disease. Black, Hispanic, and Indigenous people have higher rates of these chronic illnesses,14 increasing COVID-19 mortality risk. The increased prevalence of these illnesses is attributable to the aforementioned social conditions and environmental factors and to the additional stress associated with repeated exposure to discrimination.15
RECOMMENDATIONS
Although the disparities highlighted during the pandemic are staggering, this moment can serve as a portal to reimagine a more equitable healthcare system. Health systems and providers should (1) remain vigilant in addressing bias and its effects on patient care; (2) implement strategies to mitigate structural bias and use data to rapidly mitigate disparities in quality of care and transitions in care; and (3) address inequities, diversity, and inclusion across the entire healthcare workforce.
Addressing Provider Bias
At the patient care level, healthcare providers have a role in ensuring patients have positive experiences with the healthcare system; this is an opportunity to address medical distrust. Providers should recognize the burden of psychosocial stress and place-based risk that contributes to patients’ presentations and clinical courses. In patient encounters, this awareness should translate to action, acknowledging patients’ experiences and individuality and upholding their dignity. Under conditions of burnout, physicians’ biases are more likely to manifest in patient encounters,16 and although stress and burnout among providers are likely at an all-time high during the COVID-19 pandemic, patients of color must not suffer disproportionately.
Addressing Structural Bias in Care Provision
Health systems should establish checklist-based protocols in order to mitigate the impact of bias on patient care, such as on referrals for advanced therapies. Algorithms used to automate certain aspects of care should not be biased against Black, Hispanic, and Indigenous patients, as has been the case with algorithms that lead to Black patients receiving lower levels of care compared with White patients with similar clinical presentations.8 Health systems should therefore systematically collect racial and sociodemographic data and implement rapid-cycle evaluation of processes and outcomes to root out biases. In tracking their own performance in providing equitable care, health systems should create feedback systems that inform individual providers of their practices for improvement, and individual departments should hold frequent “morbidity and mortality” style reviews of practices and outcomes to continuously improve. Additionally, collaborations with and financial support of community-based organizations to ensure safe transitions of care and to contribute to addressing patients’ unmet social needs should become the norm. This is particularly relevant for COVID-19 survivors who may face long-term chronic physical and mental sequelae such as post–intensive care syndrome and require multidisciplinary care.17
Workforce Equity, Diversity, and Inclusion
Health systems should also examine and address the ways in which they contribute to racial health inequities beyond healthcare provision. Among healthcare organizations, hospitals employ the majority of low-wage healthcare workers, most of them Black or Hispanic women. Nearly half of Black and Hispanic female healthcare workers earn less than $15 hourly (cited as a living wage, which could help prevent a significant number of premature deaths),18 and a quarter are uninsured or on Medicaid. Raising the hourly minimum wage to at least $15 would reduce poverty among female healthcare workers by 27.1%.19 Mortality decreases as income increases, and the lowest-income healthcare workers have a nearly six-fold higher risk of death relative to their highest-earning counterparts, a gradient steeper compared with other fields.20 Health systems should guarantee occupational safety and adequate wages and benefits and provide employees with career-advancing opportunities that would facilitate upward mobility.
In addition to the aforementioned structural inequities embedded within the healthcare infrastructure, low-wage Black healthcare workers report experiencing interpersonal discrimination at work, such as being assigned more tasks compared with their White peers and having others higher up the hierarchy, such as supervisors, nurses, and physicians, assume they are incompetent. Workplace discrimination spans the organizational hierarchy. Black nurses and physicians report both interpersonal and organizational discrimination from patients and other healthcare workers and in terms of barriers to opportunities through hiring and credentialing processes.21 Black physicians are at greater risk of burnout and attrition, which is partly attributable to experiencing discrimination.22,23
To address these experiences, health systems should invest in creating a work climate that is inclusive and explicitly stands against racism and other forms of discrimination. The rise of the Black Lives Matter movement has contributed to improving people’s attitudes toward Black people over the past years,24 whereas implicit bias trainings, commonly employed to improve diversity and inclusion, may unwittingly further entrench the denial of the impact of racism (by attributing it to implicit rather than explicit attitudes)25 or heighten intergroup racial anxiety and reduce individuals’ intentions to engage in intergroup contact.26 Moreover, evidence shows interracial contact in medical school yields more positive explicit and implicit attitudes toward Black people among non–Black medical trainees, whereas bias trainings do not,27 and a positive racial climate in medical school yields a greater interest in serving underserved and minority populations among non–Black medical trainees.28 In other words, fostering a culture and structure that champions racial justice and diversifying the healthcare workforce would synergistically improve non–Black healthcare workers’ attitudes toward Black people while also improving the working conditions of Black healthcare workers and the experiences of Black patients. Healthcare is the fastest growing industry in the United States, and such initiatives would likely have a tremendous impact on moving the needle toward health equity.
CONCLUSION
The COVID-19 disparities were predictable. This pandemic may not end any time soon and certainly will not be the last we experience. Therefore, healthcare workers and health systems should recognize the societal barriers patients and workers face and implement strategies to eliminate biased practices in the provision of healthcare as well as through the compensation structure and workplace protection of healthcare workers, especially when the healthcare system experiences undue stress.
1. The color of coronavirus: COVID-19 deaths by race and ethnicity in the U.S. APM Research Lab. October 15, 2020. Accessed October 24, 2020. https://www.apmresearchlab.org/covid/deaths-by-race
2. Wang J, Huth L, Umlauf T. How the CDC’s restrictive testing guidelines hid the coronavirus epidemic. Wall Street Journal. March 22, 2020. Accessed June 20, 2020. https://www.wsj.com/articles/how-the-cdcs-restrictive-testing-guidelines-hid-the-coronavirus-epidemic-11584882001
3. McMinn S, Carlsen A, Jaspers B, Talbot R, Adeline S. In large Texas cities, access to coronavirus testing may depend on where you live. NPR. May 27, 2020. Accessed June 20, 2020. https://www.npr.org/sections/health-shots/2020/05/27/862215848/across-texas-black-and-hispanic-neighborhoods-have-fewer-coronavirus-testing-sit
4. Ko M, Needleman J, Derose KP, Laugesen MJ, Ponce NA. Residential segregation and the survival of U.S. urban public hospitals. Med Care Res Rev. 2014;71(3):243-260. https://doi.org/10.1177/1077558713515079
5. Tikkanen RS, Woolhandler S, Himmelstein DU, et al. Hospital payer and racial/ethnic mix at private academic medical centers in Boston and New York City. Int J Health Serv. 2017;47(3):460-476. https://doi.org/10.1177/0020731416689549
6. Eisenbberg A. New York’s safety-net hospitals were the front lines of the coronavirus. Now they’re facing ruin. May 16, 2020. Accessed October 24, 2020. Politico. https://www.politico.com/states/new-york/albany/story/2020/05/16/new-yorks-safety-net-hospitals-were-the-front-lines-of-the-coronavirus-now-theyre-facing-ruin-1284316
7. Cancryn A. Exclusive: emergency coronavirus funds for American Indian health stalled. Politico. March 20, 2020. Accessed June 20, 2020. https://www.politico.com/news/2020/03/20/coronavirus-american-indian-health-138724
8. Obermeyer Z, Powers B, Vogeli C, Mullainathan S. Dissecting racial bias in an algorithm used to manage the health of populations. Science. 2019;366(6464):447-453. https://doi.org/10.1126/science.aax2342
9. Goldbaum C. 41 transit workers dead: crisis takes staggering toll on subways. New York Times. April 8, 2020. Accessed June 20, 2020. https://www.nytimes.com/2020/04/08/nyregion/coronavirus-nyc-mta-subway.html
10. Levenson M. 11 days after fuming about a coughing passenger, a bus driver died from the coronavirus. New York Times. April 4, 2020. Accessed June 20, 2020. https://www.nytimes.com/2020/04/04/us/detroit-bus-driver-coronavirus.html
11. Hong N. 3 hospital workers gave out masks. Weeks later, they all were dead. New York Times. May 4, 2020. Accessed July 18, 2020. https://www.nytimes.com/2020/05/04/nyregion/coronavirus-ny-hospital-workers.html
12. Emeruwa UN, Ona S, Shaman JL, et al. Associations between built environment, neighborhood socioeconomic status, and SARS-CoV-2 infection among pregnant women in New York City. JAMA. 2020;324(4):390-392. https://doi.org/10.1001/jama.2020.11370
13. Price-Haywood EG, Burton J, Fort D, Seoane L. Hospitalization and mortality among black patients and white patients with Covid-19. N Engl J Med. 2020;382(26):2534-2543. https://doi.org/10.1056/nejmsa2011686
14. Williams DR, Mohammed SA, Leavell J, Collins C. Race, socioeconomic status, and health: complexities, ongoing challenges, and research opportunities. Ann NY Acad Sci. 2010;1186(1):69-101. https://doi.org/10.1111/j.1749-6632.2009.05339.x
15. Williams DR, Jackson PB. Social sources of racial disparities in health. Health Aff. 2005;24(2):325-334. https://doi.org/10.1377/hlthaff.24.2.325
16. Dyrbye L, Herrin J, West CP, et al. Association of racial bias with burnout among resident physicians. JAMA Netw Open. 2019;2(7):e197457. https://doi.org/10.1001/jamanetworkopen.2019.7457
17. Johnson SF, Nguemeni Tiako MJ, Flash MJE, Lamas DJ, Alba GA. Disparities in the recovery from critical illness due to COVID-19 [correspondence]. Lancet Psychiatry. 2020;7(8):e54-e55. https://doi.org/10.1016/S2215-0366(20)30292-3
18. Tsao TY, Konty KJ, Van Wye G, et al. Estimating potential reductions in premature mortality in New York City from raising the minimum wage to $15. Am J Public Health. 2016;106(6):1036-1041. https://doi.org/10.2105/AJPH.2016.303188
19. Himmelstein KEW, Venkataramani AS. Economic vulnerability among US female health care workers: potential impact of a $15-per-hour minimum wage. Am J Public Health. 2019;109(2):198-205. https://doi.org/10.2105/AJPH.2018.304801
20. Matta S, Chatterjee P, Venkataramani AS. The income-based mortality gradient among US health care workers: cohort study. J Gen Intern Med. Ahead of print. June 2020:1-3. https://doi.org/10.1007/s11606-020-05989-7
21. Wingfield AH, Chavez K. Getting in, getting hired, getting sideways looks: organizational hierarchy and perceptions of racial discrimination. Am Sociol Rev. 2020;85(1):31-57. https://doi.org/10.1177/0003122419894335
22. Nuñez-Smith M, Pilgrim N, Wynia M, et al. Race/ethnicity and workplace discrimination: results of a national survey of physicians. J Gen Intern Med. 2009;24(11):1198-1204. https://doi.org/10.1007/s11606-009-1103-9
23. Nuñez-Smith M, Pilgrim N, Wynia M, et al. Health care workplace discrimination and physician turnover. J Natl Med Assoc. 2009;101(12):1274-1282. https://doi.org/10.1016/S0027-9684(15)31139-1
24. Sawyer J, Gampa A. Implicit and explicit racial attitudes changed during Black Lives Matter. Pers Soc Psychol Bull. 2018;44(7):1039-1059. https://doi.org/10.1177/0146167218757454
25. Daumeyer NM, Onyeador IN, Brown X, Richeson JA. Consequences of attributing discrimination to implicit vs. explicit bias. J Exp Soc Psychol. 2019;84. https://doi.org/10.1016/j.jesp.2019.04.010
26. Perry SP, Dovidio JF, Murphy MC, van Ryn M. The joint effect of bias awareness and self-reported prejudice on intergroup anxiety and intentions for intergroup contact. Cult Divers Ethn Minor Psychol. 2015;21(1):89-96. https://doi.org/10.1037/a0037147
27. Onyeador IN, Wittlin NM, Burke SE, et al. The value of interracial contact for reducing anti-Black bias among non-Black physicians: a Cognitive Habits and Growth Evaluation (CHANGE) study report. Psychol Sci. 2020;31(1):18-30. https://doi.org/10.1177/0956797619879139
28. Phelan SM, Burke SE, Cunningham BA, et al. The effects of racism in medical education on students’ decisions to practice in underserved or minority communities. Acad Med. 2019;94(8):1178-1189. https://doi.org/10.1097/ACM.0000000000002719
The coronavirus disease 2019 (COVID-19) pandemic highlights long-standing inequities in health along racial/ethnic lines in the United States. Black, Hispanic, and Indigenous people have been disproportionately affected during the pandemic. For example, the age-adjusted mortality rate among Black people with COVID-19 is 3.4 times as high as that of White people.1
Structural racism shapes social forces, institutions, and ideologies that generate and reinforce racial inequities across different aspects of life. In this perspective, we discuss how, in the COVID-19 context, structural racism shapes access to and quality of care, as well as socioeconomic and health status. We offer guidance to health systems and healthcare providers on addressing health inequities.
HEALTHCARE QUALITY AND ACCESS
Disparities in access to and quality of care contribute to racial health disparities. At the onset of the COVID-19 pandemic in the United States, guidelines for COVID-19 testing were restrictive, only investigating those who had symptoms and had recently traveled to Wuhan, China, or had contact with someone who may have had the virus.2 News reports show disparities in access to testing, with testing sites favoring wealthier, Whiter communities, a feature of racial residential segregation.3 Residential segregation has also contributed to a concentration of closures among urban public hospitals, affecting access to care.4 In New York City (NYC) and Boston, early hotspots of the pandemic, Black and Hispanic patients and underinsured/uninsured patients were significantly less likely to access care from academic medical centers (AMCs) compared with White, privately insured patients.5 AMCs boast greater resources, and inequalities produced by this segregated system of care are often exacerbated by governmental allocation of resources. For instance, NYC’s public hospitals care for the city’s low-income residents (who are disproportionately insured by Medicaid), yet received far less federal aid from the Provider Relief Fund COVID-19 High Impact Payments, which favored larger, private hospitals in Manhattan. These public hospitals, however, face looming Medicaid cuts.6 Similarly, the federal government delayed the release of funds to health centers located on Native American reservations, adversely affecting the Indian Health Service’s preparedness to face the pandemic.7 In tandem with the effects of residential segregation, these data highlight the tiered nature of the US healthcare system, a structure that significantly impacts the quality of care patients receive along racial and socioeconomic lines. Furthermore, studies have documented racial disparities in the provision of advanced therapies: in the case of predicting algorithms that identify patients with complex illnesses, reliance on cost (thus, previous utilization data) rather than actual illness means that only 17.5% of Black patients receive additional help.8
SOCIOECONOMIC STATUS, OCCUPATIONAL AND RESIDENTIAL RISK
Healthcare alone does not explain the observed disparities. The disproportionately high risk of contracting the SARS-CoV-2 virus among Black, Hispanic and Indigenous people can be explained by factors that render physical distancing a luxury. First, in terms of occupational hazards, only 1 in 5 Black and 1 in 6 Hispanic workers can work remotely compared with 1 in 3 White workers. Additionally, Black and Hispanic workers are more likely to have jobs classified as critical in industries such as food retail, hospitality, and public transit. In NYC, Metropolitan Transportation Authority (MTA) employees reported using their own masks and home disinfectant at work, only to be reprimanded. By April 8, 2020, at least 41 MTA workers had died of COVID-19, and more than 6,000 were ill or self-quarantining, resulting in a transit crisis with increasingly long wait times and crowded subway platforms.9 Jason Hargrove, a Black bus driver in Detroit, shared a video underscoring the dangers of his work in which he says, “We’re out here as public workers, doing our job…but for you to get on the bus and stand on the bus, and cough several times without covering up your mouth . . . in the middle of a pandemic…some folks don’t care.” He died of COVID-19 complications 11 days after sharing his video.10 Such conditions likely also increased riders’ risk of contracting COVID-19. And while in aggregate, essential workers in healthcare receive more personal protective equipment (PPE) than those in other occupations, within NYC hospitals, the rationing of PPE was such that low-wage, nonmedical workers (79% of whom are Black or Hispanic) were given less PPE or none at all compared with nurses and physicians.11
Beyond occupational hazards, Black and Hispanic people are more likely to live in multigenerational homes, an identified risk factor of COVID-19 infection.12 Furthermore, Black and Hispanic people are overrepresented among homeless people as well as among those incarcerated. These social conditions, all products of structural racism, substantially and adversely affect the health status of Black, Hispanic, and Indigenous people, especially as it relates to comorbidities associated with higher COVID-19 mortality.
DISPARITIES IN HEALTH STATUS
Black people are disproportionately represented among COVID-19 patients requiring hospitalization, consistent with more severe disease or delayed presentation. For instance, among a cohort of 3,626 patients in a health system in Louisiana, 76.9% of COVID-19 patients hospitalized and 70.6% of those who died were Black, even though Black people comprise only 31% of this health system’s patient population.13 Conditions associated with COVID-19 mortality include heart failure, obesity, and chronic obstructive pulmonary disease. Black, Hispanic, and Indigenous people have higher rates of these chronic illnesses,14 increasing COVID-19 mortality risk. The increased prevalence of these illnesses is attributable to the aforementioned social conditions and environmental factors and to the additional stress associated with repeated exposure to discrimination.15
RECOMMENDATIONS
Although the disparities highlighted during the pandemic are staggering, this moment can serve as a portal to reimagine a more equitable healthcare system. Health systems and providers should (1) remain vigilant in addressing bias and its effects on patient care; (2) implement strategies to mitigate structural bias and use data to rapidly mitigate disparities in quality of care and transitions in care; and (3) address inequities, diversity, and inclusion across the entire healthcare workforce.
Addressing Provider Bias
At the patient care level, healthcare providers have a role in ensuring patients have positive experiences with the healthcare system; this is an opportunity to address medical distrust. Providers should recognize the burden of psychosocial stress and place-based risk that contributes to patients’ presentations and clinical courses. In patient encounters, this awareness should translate to action, acknowledging patients’ experiences and individuality and upholding their dignity. Under conditions of burnout, physicians’ biases are more likely to manifest in patient encounters,16 and although stress and burnout among providers are likely at an all-time high during the COVID-19 pandemic, patients of color must not suffer disproportionately.
Addressing Structural Bias in Care Provision
Health systems should establish checklist-based protocols in order to mitigate the impact of bias on patient care, such as on referrals for advanced therapies. Algorithms used to automate certain aspects of care should not be biased against Black, Hispanic, and Indigenous patients, as has been the case with algorithms that lead to Black patients receiving lower levels of care compared with White patients with similar clinical presentations.8 Health systems should therefore systematically collect racial and sociodemographic data and implement rapid-cycle evaluation of processes and outcomes to root out biases. In tracking their own performance in providing equitable care, health systems should create feedback systems that inform individual providers of their practices for improvement, and individual departments should hold frequent “morbidity and mortality” style reviews of practices and outcomes to continuously improve. Additionally, collaborations with and financial support of community-based organizations to ensure safe transitions of care and to contribute to addressing patients’ unmet social needs should become the norm. This is particularly relevant for COVID-19 survivors who may face long-term chronic physical and mental sequelae such as post–intensive care syndrome and require multidisciplinary care.17
Workforce Equity, Diversity, and Inclusion
Health systems should also examine and address the ways in which they contribute to racial health inequities beyond healthcare provision. Among healthcare organizations, hospitals employ the majority of low-wage healthcare workers, most of them Black or Hispanic women. Nearly half of Black and Hispanic female healthcare workers earn less than $15 hourly (cited as a living wage, which could help prevent a significant number of premature deaths),18 and a quarter are uninsured or on Medicaid. Raising the hourly minimum wage to at least $15 would reduce poverty among female healthcare workers by 27.1%.19 Mortality decreases as income increases, and the lowest-income healthcare workers have a nearly six-fold higher risk of death relative to their highest-earning counterparts, a gradient steeper compared with other fields.20 Health systems should guarantee occupational safety and adequate wages and benefits and provide employees with career-advancing opportunities that would facilitate upward mobility.
In addition to the aforementioned structural inequities embedded within the healthcare infrastructure, low-wage Black healthcare workers report experiencing interpersonal discrimination at work, such as being assigned more tasks compared with their White peers and having others higher up the hierarchy, such as supervisors, nurses, and physicians, assume they are incompetent. Workplace discrimination spans the organizational hierarchy. Black nurses and physicians report both interpersonal and organizational discrimination from patients and other healthcare workers and in terms of barriers to opportunities through hiring and credentialing processes.21 Black physicians are at greater risk of burnout and attrition, which is partly attributable to experiencing discrimination.22,23
To address these experiences, health systems should invest in creating a work climate that is inclusive and explicitly stands against racism and other forms of discrimination. The rise of the Black Lives Matter movement has contributed to improving people’s attitudes toward Black people over the past years,24 whereas implicit bias trainings, commonly employed to improve diversity and inclusion, may unwittingly further entrench the denial of the impact of racism (by attributing it to implicit rather than explicit attitudes)25 or heighten intergroup racial anxiety and reduce individuals’ intentions to engage in intergroup contact.26 Moreover, evidence shows interracial contact in medical school yields more positive explicit and implicit attitudes toward Black people among non–Black medical trainees, whereas bias trainings do not,27 and a positive racial climate in medical school yields a greater interest in serving underserved and minority populations among non–Black medical trainees.28 In other words, fostering a culture and structure that champions racial justice and diversifying the healthcare workforce would synergistically improve non–Black healthcare workers’ attitudes toward Black people while also improving the working conditions of Black healthcare workers and the experiences of Black patients. Healthcare is the fastest growing industry in the United States, and such initiatives would likely have a tremendous impact on moving the needle toward health equity.
CONCLUSION
The COVID-19 disparities were predictable. This pandemic may not end any time soon and certainly will not be the last we experience. Therefore, healthcare workers and health systems should recognize the societal barriers patients and workers face and implement strategies to eliminate biased practices in the provision of healthcare as well as through the compensation structure and workplace protection of healthcare workers, especially when the healthcare system experiences undue stress.
The coronavirus disease 2019 (COVID-19) pandemic highlights long-standing inequities in health along racial/ethnic lines in the United States. Black, Hispanic, and Indigenous people have been disproportionately affected during the pandemic. For example, the age-adjusted mortality rate among Black people with COVID-19 is 3.4 times as high as that of White people.1
Structural racism shapes social forces, institutions, and ideologies that generate and reinforce racial inequities across different aspects of life. In this perspective, we discuss how, in the COVID-19 context, structural racism shapes access to and quality of care, as well as socioeconomic and health status. We offer guidance to health systems and healthcare providers on addressing health inequities.
HEALTHCARE QUALITY AND ACCESS
Disparities in access to and quality of care contribute to racial health disparities. At the onset of the COVID-19 pandemic in the United States, guidelines for COVID-19 testing were restrictive, only investigating those who had symptoms and had recently traveled to Wuhan, China, or had contact with someone who may have had the virus.2 News reports show disparities in access to testing, with testing sites favoring wealthier, Whiter communities, a feature of racial residential segregation.3 Residential segregation has also contributed to a concentration of closures among urban public hospitals, affecting access to care.4 In New York City (NYC) and Boston, early hotspots of the pandemic, Black and Hispanic patients and underinsured/uninsured patients were significantly less likely to access care from academic medical centers (AMCs) compared with White, privately insured patients.5 AMCs boast greater resources, and inequalities produced by this segregated system of care are often exacerbated by governmental allocation of resources. For instance, NYC’s public hospitals care for the city’s low-income residents (who are disproportionately insured by Medicaid), yet received far less federal aid from the Provider Relief Fund COVID-19 High Impact Payments, which favored larger, private hospitals in Manhattan. These public hospitals, however, face looming Medicaid cuts.6 Similarly, the federal government delayed the release of funds to health centers located on Native American reservations, adversely affecting the Indian Health Service’s preparedness to face the pandemic.7 In tandem with the effects of residential segregation, these data highlight the tiered nature of the US healthcare system, a structure that significantly impacts the quality of care patients receive along racial and socioeconomic lines. Furthermore, studies have documented racial disparities in the provision of advanced therapies: in the case of predicting algorithms that identify patients with complex illnesses, reliance on cost (thus, previous utilization data) rather than actual illness means that only 17.5% of Black patients receive additional help.8
SOCIOECONOMIC STATUS, OCCUPATIONAL AND RESIDENTIAL RISK
Healthcare alone does not explain the observed disparities. The disproportionately high risk of contracting the SARS-CoV-2 virus among Black, Hispanic and Indigenous people can be explained by factors that render physical distancing a luxury. First, in terms of occupational hazards, only 1 in 5 Black and 1 in 6 Hispanic workers can work remotely compared with 1 in 3 White workers. Additionally, Black and Hispanic workers are more likely to have jobs classified as critical in industries such as food retail, hospitality, and public transit. In NYC, Metropolitan Transportation Authority (MTA) employees reported using their own masks and home disinfectant at work, only to be reprimanded. By April 8, 2020, at least 41 MTA workers had died of COVID-19, and more than 6,000 were ill or self-quarantining, resulting in a transit crisis with increasingly long wait times and crowded subway platforms.9 Jason Hargrove, a Black bus driver in Detroit, shared a video underscoring the dangers of his work in which he says, “We’re out here as public workers, doing our job…but for you to get on the bus and stand on the bus, and cough several times without covering up your mouth . . . in the middle of a pandemic…some folks don’t care.” He died of COVID-19 complications 11 days after sharing his video.10 Such conditions likely also increased riders’ risk of contracting COVID-19. And while in aggregate, essential workers in healthcare receive more personal protective equipment (PPE) than those in other occupations, within NYC hospitals, the rationing of PPE was such that low-wage, nonmedical workers (79% of whom are Black or Hispanic) were given less PPE or none at all compared with nurses and physicians.11
Beyond occupational hazards, Black and Hispanic people are more likely to live in multigenerational homes, an identified risk factor of COVID-19 infection.12 Furthermore, Black and Hispanic people are overrepresented among homeless people as well as among those incarcerated. These social conditions, all products of structural racism, substantially and adversely affect the health status of Black, Hispanic, and Indigenous people, especially as it relates to comorbidities associated with higher COVID-19 mortality.
DISPARITIES IN HEALTH STATUS
Black people are disproportionately represented among COVID-19 patients requiring hospitalization, consistent with more severe disease or delayed presentation. For instance, among a cohort of 3,626 patients in a health system in Louisiana, 76.9% of COVID-19 patients hospitalized and 70.6% of those who died were Black, even though Black people comprise only 31% of this health system’s patient population.13 Conditions associated with COVID-19 mortality include heart failure, obesity, and chronic obstructive pulmonary disease. Black, Hispanic, and Indigenous people have higher rates of these chronic illnesses,14 increasing COVID-19 mortality risk. The increased prevalence of these illnesses is attributable to the aforementioned social conditions and environmental factors and to the additional stress associated with repeated exposure to discrimination.15
RECOMMENDATIONS
Although the disparities highlighted during the pandemic are staggering, this moment can serve as a portal to reimagine a more equitable healthcare system. Health systems and providers should (1) remain vigilant in addressing bias and its effects on patient care; (2) implement strategies to mitigate structural bias and use data to rapidly mitigate disparities in quality of care and transitions in care; and (3) address inequities, diversity, and inclusion across the entire healthcare workforce.
Addressing Provider Bias
At the patient care level, healthcare providers have a role in ensuring patients have positive experiences with the healthcare system; this is an opportunity to address medical distrust. Providers should recognize the burden of psychosocial stress and place-based risk that contributes to patients’ presentations and clinical courses. In patient encounters, this awareness should translate to action, acknowledging patients’ experiences and individuality and upholding their dignity. Under conditions of burnout, physicians’ biases are more likely to manifest in patient encounters,16 and although stress and burnout among providers are likely at an all-time high during the COVID-19 pandemic, patients of color must not suffer disproportionately.
Addressing Structural Bias in Care Provision
Health systems should establish checklist-based protocols in order to mitigate the impact of bias on patient care, such as on referrals for advanced therapies. Algorithms used to automate certain aspects of care should not be biased against Black, Hispanic, and Indigenous patients, as has been the case with algorithms that lead to Black patients receiving lower levels of care compared with White patients with similar clinical presentations.8 Health systems should therefore systematically collect racial and sociodemographic data and implement rapid-cycle evaluation of processes and outcomes to root out biases. In tracking their own performance in providing equitable care, health systems should create feedback systems that inform individual providers of their practices for improvement, and individual departments should hold frequent “morbidity and mortality” style reviews of practices and outcomes to continuously improve. Additionally, collaborations with and financial support of community-based organizations to ensure safe transitions of care and to contribute to addressing patients’ unmet social needs should become the norm. This is particularly relevant for COVID-19 survivors who may face long-term chronic physical and mental sequelae such as post–intensive care syndrome and require multidisciplinary care.17
Workforce Equity, Diversity, and Inclusion
Health systems should also examine and address the ways in which they contribute to racial health inequities beyond healthcare provision. Among healthcare organizations, hospitals employ the majority of low-wage healthcare workers, most of them Black or Hispanic women. Nearly half of Black and Hispanic female healthcare workers earn less than $15 hourly (cited as a living wage, which could help prevent a significant number of premature deaths),18 and a quarter are uninsured or on Medicaid. Raising the hourly minimum wage to at least $15 would reduce poverty among female healthcare workers by 27.1%.19 Mortality decreases as income increases, and the lowest-income healthcare workers have a nearly six-fold higher risk of death relative to their highest-earning counterparts, a gradient steeper compared with other fields.20 Health systems should guarantee occupational safety and adequate wages and benefits and provide employees with career-advancing opportunities that would facilitate upward mobility.
In addition to the aforementioned structural inequities embedded within the healthcare infrastructure, low-wage Black healthcare workers report experiencing interpersonal discrimination at work, such as being assigned more tasks compared with their White peers and having others higher up the hierarchy, such as supervisors, nurses, and physicians, assume they are incompetent. Workplace discrimination spans the organizational hierarchy. Black nurses and physicians report both interpersonal and organizational discrimination from patients and other healthcare workers and in terms of barriers to opportunities through hiring and credentialing processes.21 Black physicians are at greater risk of burnout and attrition, which is partly attributable to experiencing discrimination.22,23
To address these experiences, health systems should invest in creating a work climate that is inclusive and explicitly stands against racism and other forms of discrimination. The rise of the Black Lives Matter movement has contributed to improving people’s attitudes toward Black people over the past years,24 whereas implicit bias trainings, commonly employed to improve diversity and inclusion, may unwittingly further entrench the denial of the impact of racism (by attributing it to implicit rather than explicit attitudes)25 or heighten intergroup racial anxiety and reduce individuals’ intentions to engage in intergroup contact.26 Moreover, evidence shows interracial contact in medical school yields more positive explicit and implicit attitudes toward Black people among non–Black medical trainees, whereas bias trainings do not,27 and a positive racial climate in medical school yields a greater interest in serving underserved and minority populations among non–Black medical trainees.28 In other words, fostering a culture and structure that champions racial justice and diversifying the healthcare workforce would synergistically improve non–Black healthcare workers’ attitudes toward Black people while also improving the working conditions of Black healthcare workers and the experiences of Black patients. Healthcare is the fastest growing industry in the United States, and such initiatives would likely have a tremendous impact on moving the needle toward health equity.
CONCLUSION
The COVID-19 disparities were predictable. This pandemic may not end any time soon and certainly will not be the last we experience. Therefore, healthcare workers and health systems should recognize the societal barriers patients and workers face and implement strategies to eliminate biased practices in the provision of healthcare as well as through the compensation structure and workplace protection of healthcare workers, especially when the healthcare system experiences undue stress.
1. The color of coronavirus: COVID-19 deaths by race and ethnicity in the U.S. APM Research Lab. October 15, 2020. Accessed October 24, 2020. https://www.apmresearchlab.org/covid/deaths-by-race
2. Wang J, Huth L, Umlauf T. How the CDC’s restrictive testing guidelines hid the coronavirus epidemic. Wall Street Journal. March 22, 2020. Accessed June 20, 2020. https://www.wsj.com/articles/how-the-cdcs-restrictive-testing-guidelines-hid-the-coronavirus-epidemic-11584882001
3. McMinn S, Carlsen A, Jaspers B, Talbot R, Adeline S. In large Texas cities, access to coronavirus testing may depend on where you live. NPR. May 27, 2020. Accessed June 20, 2020. https://www.npr.org/sections/health-shots/2020/05/27/862215848/across-texas-black-and-hispanic-neighborhoods-have-fewer-coronavirus-testing-sit
4. Ko M, Needleman J, Derose KP, Laugesen MJ, Ponce NA. Residential segregation and the survival of U.S. urban public hospitals. Med Care Res Rev. 2014;71(3):243-260. https://doi.org/10.1177/1077558713515079
5. Tikkanen RS, Woolhandler S, Himmelstein DU, et al. Hospital payer and racial/ethnic mix at private academic medical centers in Boston and New York City. Int J Health Serv. 2017;47(3):460-476. https://doi.org/10.1177/0020731416689549
6. Eisenbberg A. New York’s safety-net hospitals were the front lines of the coronavirus. Now they’re facing ruin. May 16, 2020. Accessed October 24, 2020. Politico. https://www.politico.com/states/new-york/albany/story/2020/05/16/new-yorks-safety-net-hospitals-were-the-front-lines-of-the-coronavirus-now-theyre-facing-ruin-1284316
7. Cancryn A. Exclusive: emergency coronavirus funds for American Indian health stalled. Politico. March 20, 2020. Accessed June 20, 2020. https://www.politico.com/news/2020/03/20/coronavirus-american-indian-health-138724
8. Obermeyer Z, Powers B, Vogeli C, Mullainathan S. Dissecting racial bias in an algorithm used to manage the health of populations. Science. 2019;366(6464):447-453. https://doi.org/10.1126/science.aax2342
9. Goldbaum C. 41 transit workers dead: crisis takes staggering toll on subways. New York Times. April 8, 2020. Accessed June 20, 2020. https://www.nytimes.com/2020/04/08/nyregion/coronavirus-nyc-mta-subway.html
10. Levenson M. 11 days after fuming about a coughing passenger, a bus driver died from the coronavirus. New York Times. April 4, 2020. Accessed June 20, 2020. https://www.nytimes.com/2020/04/04/us/detroit-bus-driver-coronavirus.html
11. Hong N. 3 hospital workers gave out masks. Weeks later, they all were dead. New York Times. May 4, 2020. Accessed July 18, 2020. https://www.nytimes.com/2020/05/04/nyregion/coronavirus-ny-hospital-workers.html
12. Emeruwa UN, Ona S, Shaman JL, et al. Associations between built environment, neighborhood socioeconomic status, and SARS-CoV-2 infection among pregnant women in New York City. JAMA. 2020;324(4):390-392. https://doi.org/10.1001/jama.2020.11370
13. Price-Haywood EG, Burton J, Fort D, Seoane L. Hospitalization and mortality among black patients and white patients with Covid-19. N Engl J Med. 2020;382(26):2534-2543. https://doi.org/10.1056/nejmsa2011686
14. Williams DR, Mohammed SA, Leavell J, Collins C. Race, socioeconomic status, and health: complexities, ongoing challenges, and research opportunities. Ann NY Acad Sci. 2010;1186(1):69-101. https://doi.org/10.1111/j.1749-6632.2009.05339.x
15. Williams DR, Jackson PB. Social sources of racial disparities in health. Health Aff. 2005;24(2):325-334. https://doi.org/10.1377/hlthaff.24.2.325
16. Dyrbye L, Herrin J, West CP, et al. Association of racial bias with burnout among resident physicians. JAMA Netw Open. 2019;2(7):e197457. https://doi.org/10.1001/jamanetworkopen.2019.7457
17. Johnson SF, Nguemeni Tiako MJ, Flash MJE, Lamas DJ, Alba GA. Disparities in the recovery from critical illness due to COVID-19 [correspondence]. Lancet Psychiatry. 2020;7(8):e54-e55. https://doi.org/10.1016/S2215-0366(20)30292-3
18. Tsao TY, Konty KJ, Van Wye G, et al. Estimating potential reductions in premature mortality in New York City from raising the minimum wage to $15. Am J Public Health. 2016;106(6):1036-1041. https://doi.org/10.2105/AJPH.2016.303188
19. Himmelstein KEW, Venkataramani AS. Economic vulnerability among US female health care workers: potential impact of a $15-per-hour minimum wage. Am J Public Health. 2019;109(2):198-205. https://doi.org/10.2105/AJPH.2018.304801
20. Matta S, Chatterjee P, Venkataramani AS. The income-based mortality gradient among US health care workers: cohort study. J Gen Intern Med. Ahead of print. June 2020:1-3. https://doi.org/10.1007/s11606-020-05989-7
21. Wingfield AH, Chavez K. Getting in, getting hired, getting sideways looks: organizational hierarchy and perceptions of racial discrimination. Am Sociol Rev. 2020;85(1):31-57. https://doi.org/10.1177/0003122419894335
22. Nuñez-Smith M, Pilgrim N, Wynia M, et al. Race/ethnicity and workplace discrimination: results of a national survey of physicians. J Gen Intern Med. 2009;24(11):1198-1204. https://doi.org/10.1007/s11606-009-1103-9
23. Nuñez-Smith M, Pilgrim N, Wynia M, et al. Health care workplace discrimination and physician turnover. J Natl Med Assoc. 2009;101(12):1274-1282. https://doi.org/10.1016/S0027-9684(15)31139-1
24. Sawyer J, Gampa A. Implicit and explicit racial attitudes changed during Black Lives Matter. Pers Soc Psychol Bull. 2018;44(7):1039-1059. https://doi.org/10.1177/0146167218757454
25. Daumeyer NM, Onyeador IN, Brown X, Richeson JA. Consequences of attributing discrimination to implicit vs. explicit bias. J Exp Soc Psychol. 2019;84. https://doi.org/10.1016/j.jesp.2019.04.010
26. Perry SP, Dovidio JF, Murphy MC, van Ryn M. The joint effect of bias awareness and self-reported prejudice on intergroup anxiety and intentions for intergroup contact. Cult Divers Ethn Minor Psychol. 2015;21(1):89-96. https://doi.org/10.1037/a0037147
27. Onyeador IN, Wittlin NM, Burke SE, et al. The value of interracial contact for reducing anti-Black bias among non-Black physicians: a Cognitive Habits and Growth Evaluation (CHANGE) study report. Psychol Sci. 2020;31(1):18-30. https://doi.org/10.1177/0956797619879139
28. Phelan SM, Burke SE, Cunningham BA, et al. The effects of racism in medical education on students’ decisions to practice in underserved or minority communities. Acad Med. 2019;94(8):1178-1189. https://doi.org/10.1097/ACM.0000000000002719
1. The color of coronavirus: COVID-19 deaths by race and ethnicity in the U.S. APM Research Lab. October 15, 2020. Accessed October 24, 2020. https://www.apmresearchlab.org/covid/deaths-by-race
2. Wang J, Huth L, Umlauf T. How the CDC’s restrictive testing guidelines hid the coronavirus epidemic. Wall Street Journal. March 22, 2020. Accessed June 20, 2020. https://www.wsj.com/articles/how-the-cdcs-restrictive-testing-guidelines-hid-the-coronavirus-epidemic-11584882001
3. McMinn S, Carlsen A, Jaspers B, Talbot R, Adeline S. In large Texas cities, access to coronavirus testing may depend on where you live. NPR. May 27, 2020. Accessed June 20, 2020. https://www.npr.org/sections/health-shots/2020/05/27/862215848/across-texas-black-and-hispanic-neighborhoods-have-fewer-coronavirus-testing-sit
4. Ko M, Needleman J, Derose KP, Laugesen MJ, Ponce NA. Residential segregation and the survival of U.S. urban public hospitals. Med Care Res Rev. 2014;71(3):243-260. https://doi.org/10.1177/1077558713515079
5. Tikkanen RS, Woolhandler S, Himmelstein DU, et al. Hospital payer and racial/ethnic mix at private academic medical centers in Boston and New York City. Int J Health Serv. 2017;47(3):460-476. https://doi.org/10.1177/0020731416689549
6. Eisenbberg A. New York’s safety-net hospitals were the front lines of the coronavirus. Now they’re facing ruin. May 16, 2020. Accessed October 24, 2020. Politico. https://www.politico.com/states/new-york/albany/story/2020/05/16/new-yorks-safety-net-hospitals-were-the-front-lines-of-the-coronavirus-now-theyre-facing-ruin-1284316
7. Cancryn A. Exclusive: emergency coronavirus funds for American Indian health stalled. Politico. March 20, 2020. Accessed June 20, 2020. https://www.politico.com/news/2020/03/20/coronavirus-american-indian-health-138724
8. Obermeyer Z, Powers B, Vogeli C, Mullainathan S. Dissecting racial bias in an algorithm used to manage the health of populations. Science. 2019;366(6464):447-453. https://doi.org/10.1126/science.aax2342
9. Goldbaum C. 41 transit workers dead: crisis takes staggering toll on subways. New York Times. April 8, 2020. Accessed June 20, 2020. https://www.nytimes.com/2020/04/08/nyregion/coronavirus-nyc-mta-subway.html
10. Levenson M. 11 days after fuming about a coughing passenger, a bus driver died from the coronavirus. New York Times. April 4, 2020. Accessed June 20, 2020. https://www.nytimes.com/2020/04/04/us/detroit-bus-driver-coronavirus.html
11. Hong N. 3 hospital workers gave out masks. Weeks later, they all were dead. New York Times. May 4, 2020. Accessed July 18, 2020. https://www.nytimes.com/2020/05/04/nyregion/coronavirus-ny-hospital-workers.html
12. Emeruwa UN, Ona S, Shaman JL, et al. Associations between built environment, neighborhood socioeconomic status, and SARS-CoV-2 infection among pregnant women in New York City. JAMA. 2020;324(4):390-392. https://doi.org/10.1001/jama.2020.11370
13. Price-Haywood EG, Burton J, Fort D, Seoane L. Hospitalization and mortality among black patients and white patients with Covid-19. N Engl J Med. 2020;382(26):2534-2543. https://doi.org/10.1056/nejmsa2011686
14. Williams DR, Mohammed SA, Leavell J, Collins C. Race, socioeconomic status, and health: complexities, ongoing challenges, and research opportunities. Ann NY Acad Sci. 2010;1186(1):69-101. https://doi.org/10.1111/j.1749-6632.2009.05339.x
15. Williams DR, Jackson PB. Social sources of racial disparities in health. Health Aff. 2005;24(2):325-334. https://doi.org/10.1377/hlthaff.24.2.325
16. Dyrbye L, Herrin J, West CP, et al. Association of racial bias with burnout among resident physicians. JAMA Netw Open. 2019;2(7):e197457. https://doi.org/10.1001/jamanetworkopen.2019.7457
17. Johnson SF, Nguemeni Tiako MJ, Flash MJE, Lamas DJ, Alba GA. Disparities in the recovery from critical illness due to COVID-19 [correspondence]. Lancet Psychiatry. 2020;7(8):e54-e55. https://doi.org/10.1016/S2215-0366(20)30292-3
18. Tsao TY, Konty KJ, Van Wye G, et al. Estimating potential reductions in premature mortality in New York City from raising the minimum wage to $15. Am J Public Health. 2016;106(6):1036-1041. https://doi.org/10.2105/AJPH.2016.303188
19. Himmelstein KEW, Venkataramani AS. Economic vulnerability among US female health care workers: potential impact of a $15-per-hour minimum wage. Am J Public Health. 2019;109(2):198-205. https://doi.org/10.2105/AJPH.2018.304801
20. Matta S, Chatterjee P, Venkataramani AS. The income-based mortality gradient among US health care workers: cohort study. J Gen Intern Med. Ahead of print. June 2020:1-3. https://doi.org/10.1007/s11606-020-05989-7
21. Wingfield AH, Chavez K. Getting in, getting hired, getting sideways looks: organizational hierarchy and perceptions of racial discrimination. Am Sociol Rev. 2020;85(1):31-57. https://doi.org/10.1177/0003122419894335
22. Nuñez-Smith M, Pilgrim N, Wynia M, et al. Race/ethnicity and workplace discrimination: results of a national survey of physicians. J Gen Intern Med. 2009;24(11):1198-1204. https://doi.org/10.1007/s11606-009-1103-9
23. Nuñez-Smith M, Pilgrim N, Wynia M, et al. Health care workplace discrimination and physician turnover. J Natl Med Assoc. 2009;101(12):1274-1282. https://doi.org/10.1016/S0027-9684(15)31139-1
24. Sawyer J, Gampa A. Implicit and explicit racial attitudes changed during Black Lives Matter. Pers Soc Psychol Bull. 2018;44(7):1039-1059. https://doi.org/10.1177/0146167218757454
25. Daumeyer NM, Onyeador IN, Brown X, Richeson JA. Consequences of attributing discrimination to implicit vs. explicit bias. J Exp Soc Psychol. 2019;84. https://doi.org/10.1016/j.jesp.2019.04.010
26. Perry SP, Dovidio JF, Murphy MC, van Ryn M. The joint effect of bias awareness and self-reported prejudice on intergroup anxiety and intentions for intergroup contact. Cult Divers Ethn Minor Psychol. 2015;21(1):89-96. https://doi.org/10.1037/a0037147
27. Onyeador IN, Wittlin NM, Burke SE, et al. The value of interracial contact for reducing anti-Black bias among non-Black physicians: a Cognitive Habits and Growth Evaluation (CHANGE) study report. Psychol Sci. 2020;31(1):18-30. https://doi.org/10.1177/0956797619879139
28. Phelan SM, Burke SE, Cunningham BA, et al. The effects of racism in medical education on students’ decisions to practice in underserved or minority communities. Acad Med. 2019;94(8):1178-1189. https://doi.org/10.1097/ACM.0000000000002719
© 2021 Society of Hospital Medicine
Things We Do for No Reason™: Universal Venous Thromboembolism Chemoprophylaxis in Low-Risk Hospitalized Medical Patients

Inspired by the ABIM Foundation’s Choosing Wisel y ® campaign, the “Things We Do for No Reason ™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A hospitalist admits a 68-year-old woman for community-acquired pneumonia with a past medical history of hypertension, gastroesophageal reflux disease, and osteoarthritis. Her hospitalist consults physical therapy to maximize mobility; continues her home medications including pantoprazole, hydrochlorothiazide, and acetaminophen; and initiates antimicrobial therapy with ceftriaxone and azithromycin. The hospital admission order set requires administration of subcutaneous unfractionated heparin for venous thromboembolism chemoprophylaxis.
WHY YOU MIGHT THINK UNIVERSAL CHEMOPROPHYLAXIS IS NECESSARY
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE), ranks among the leading preventable causes of morbidity and mortality in hospitalized patients.1 DVTs can rapidly progress to a PE, which account for 5% to 10% of in-hospital deaths.1 The negative sequelae of in-hospital VTE, including prolonged hospital stay, increased healthcare costs, and greater risks associated with pharmacologic treatment, add $9 to $18.2 billion in US healthcare expenditures each year.2 Various risk-assessment models (RAMs) identify medical patients at high risk for developing VTE based on the presence of risk factors including acute heart failure, prior history of VTE, and reduced mobility.3 Since hospitalization may itself increase the risk for VTE, medical patients often receive universal chemoprophylaxis with anticoagulants such as unfractionated heparin (UFH), low-molecular-weight heparin (LMWH), or fondaparinux.3 A meta-analysis of randomized controlled trials (RCTs) published by Wein et al supports the use of VTE chemoprophylaxis in high-risk patients.4 It showed statistically significant reductions in rates of PE in high-risk hospitalized medical patients with UFH (risk ratio [RR], 0.64; 95% CI, 0.50-0.82) or LMWH chemoprophylaxis (RR, 0.37; 95% CI, 0.21-0.64), compared with controls.
In recognition of the magnitude of the problem, national organizations have emphasized routine chemoprophylaxis for prevention of in-hospital VTE as a top-priority measure for patient safety.5,6 The Joint Commission includes chemoprophylaxis as a quality core metric and failure to adhere to such standards compromises hospital accreditation.5 Since 2008, the Centers for Medicare & Medicaid Services no longer reimburses hospitals for preventable VTE and requires institutions to document the rationale for omitting chemoprophylaxis if not commenced on hospital admission.6
WHY CHEMOPROPHYLAXIS FOR LOW-RISK MEDICAL PATIENTS IS UNNECESSARY
In order to understand why chemoprophylaxis fails to benefit low-risk medical patients, it is necessary to critically examine the benefits identified in trials of high-risk patients. Although RCTs and meta-analysis of chemoprophylaxis have consistently demonstrated a reduction in VTE, prevention of asymptomatic VTE identified on screening with ultrasound or venography accounts for more than 90% of the composite outcome in the three key trials.7-9 Hospitalists do not routinely screen for asymptomatic VTE, and incorporation of these events into composite VTE outcomes inflates the magnitude of benefit gained by chemoprophylaxis. Importantly, the standard of care does not include screening for asymptomatic DVTs, and studies have estimated that only 10% to 15% of asymptomatic DVTs progress to a symptomatic VTE.10
A meta-analysis of trials evaluating unselected general medical patients (ie, not those with specific high-risk conditions such as acute myocardial infarction) did not show a reduction in symptomatic VTE with chemoprophylaxis (odds ratio [OR], 0.59; 95% CI, 0.29-1.23).11 In the meta-analysis by Wein et al, which did include patients with specific high-risk conditions, chemoprophylaxis produced a small absolute risk reduction, resulting in a number needed to treat (NNT) of 345 to prevent one PE.4 This demonstrates that, even in high-risk patients, the magnitude of benefit is small. Population-level data also question the benefit of chemoprophylaxis. Flanders et al stratified 35 Michigan hospitals into high-, moderate-, and low-performance tertiles, with performance based on the rate of chemoprophylaxis use on admission for general medical patients at high-risk for VTE. The authors found no significant difference in the rate of VTE at 90 days among tertiles.12 These findings question the usefulness of universal chemoprophylaxis when applied in a real-world setting.
The high rates of VTE in the absence of chemoprophylaxis reported in historic trials may overestimate the contemporary risk. A 2019 multicenter, observational study examined the rate of hospital-acquired DVT for 1,170 low- and high-risk patients with acute medical illness admitted to the internal medicine ward.13 Of them, 250 (21%) underwent prophylaxis with parenteral anticoagulants (mean Padua Prediction Score, 4.5). The remaining 920 (79%) were not treated with prophylaxis (mean Padua Prediction Score, 2.5). All patients underwent ultrasound at admission and discharge. The average length of stay was 13 days, and just three patients (0.3%) experienced in-hospital DVT, two of whom were receiving chemoprophylaxis. Only one (0.09%) DVT was symptomatic.
It should be emphasized that any evidence favoring chemoprophylaxis comes from studies of patients at high-risk of VTE. No data show benefit for low-risk patients. Therefore, any risk of chemoprophylaxis likely outweighs the benefits in low-risk patients. Importantly, the risks are underappreciated. A 2014 meta-analysis reported an increased risk of major hemorrhage (OR, 1.81; 95% CI, 1.10-2.98; P = .02) in high-risk medically ill patients on chemoprophylaxis.14 This results in a number needed to harm for major bleeding of 336, a value similar to the NNT for benefit reported by Wein et al.4 Heparin-induced thrombocytopenia, a potentially limb- and life-threatening complication of UFH or LMWH exposure, has an overall incidence of 0.3% to 0.7% in hospitalized patients on chemoprophylaxis.3 Finally, the most commonly used chemoprophylaxis medications are administered subcutaneously, resulting in injection site pain. Unsurprisingly, hospitalized patients refuse chemoprophylaxis more frequently than any other medication.15
The negative implications of inappropriate chemoprophylaxis extend beyond direct harms to patients. Poor stratification and overuse results in unnecessary healthcare costs. One single-center retrospective review demonstrated that, after integration of chemoprophylaxis into hospital order sets, 76% of patients received unnecessary administration of chemoprophylaxis, resulting in an annualized expenditure of $77,652.16 This does not take into account costs associated with major bleeds.
Unfortunately, the pendulum has shifted from an era of underprescribing chemoprophylaxis to hospitalized medical patients to one of overprescribing. Data published in 2018 suggest that providers overuse chemoprophylaxis in low-risk medical patients at more than double the rate of underusing it in high-risk patients (57% vs 21%).17
Several national societies, including the often cited American College of Chest Physicians (ACCP) and American Society of Hematology (ASH), provide guidance on the use of VTE chemoprophylaxis in acutely ill medical inpatients.3,18 The ASH guidelines conditionally recommend VTE chemoprophylaxis rather than no chemoprophylaxis.18 However, the guidelines do not provide guidance on a risk-stratified approach and disclose that this recommendation is supported by a low certainty in the evidence of the net health benefit gained.18 Guidelines from ACCP lean towards individualized care and recommend against the use of VTE chemoprophylaxis for hospitalized acutely ill, low-risk medical patients.3
WHAT YOU SHOULD DO INSTEAD
Clinicians should risk stratify using validated RAMs when making a patient-centered treatment plan on admission. The table outlines the most common RAMs with evidence for use in acute medically ill hospitalized patients. Although RAMs have limitations (eg, lack of prospective validation and complexity), the ACCP guidelines advocate for their use.3
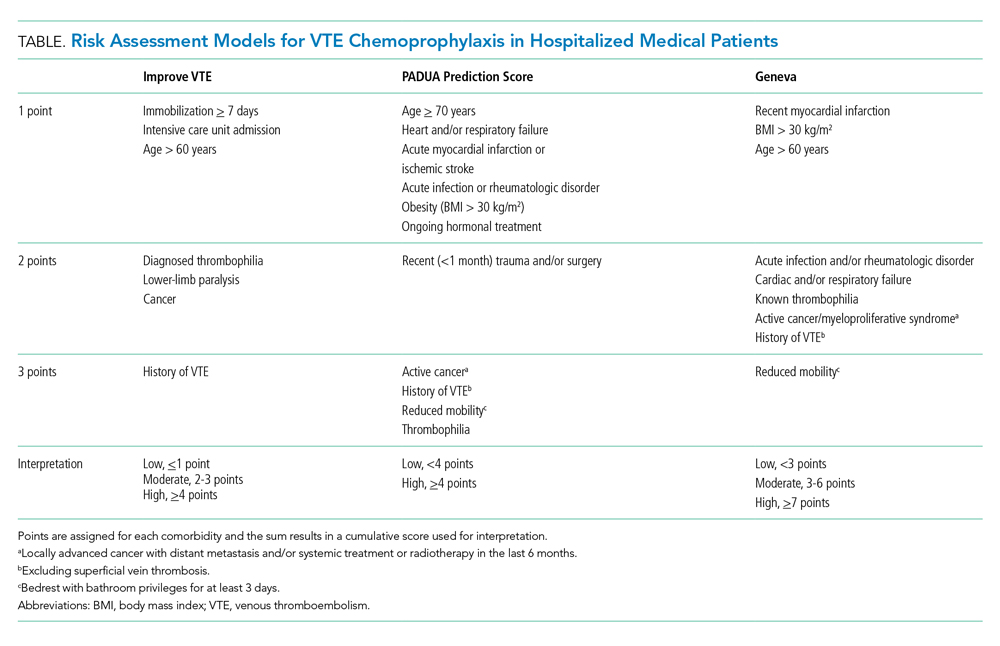
Given that immobility independently increases risk for VTE, early mobilization is a simple and cost-effective way to potentially prevent VTE in low-risk patients. In addition to this potential benefit, early mobilization shortens the length of hospital stay, improves functional status and rates of delirium in hospitalized elderly patients, and hastens postoperative recovery after major surgeries.19
RECOMMENDATIONS
- Incorporate a patient-centered, risk-stratified approach to identify low-risk patients. This can be done manually or with use of RAMS embedded in the electronic health record.
- Do not prescribe chemoprophylaxis to low-risk hospitalized medical patients.
- Emphasize the importance of early mobilization in hospitalized patients.
CONCLUSION
In regard to the case, the hospitalist should use a RAM developed for the nonsurgical, non–critically ill patient to determine her need for chemoprophylaxis. Based on the clinical data presented, the three RAMs available would classify the patient as low risk for developing an in-hospital VTE. She should not receive chemoprophylaxis given the lack of data demonstrating benefit in this population. To mitigate the potential risk of bleeding, heparin-induced thrombocytopenia, and painful injections, the hospitalist should discontinue heparin. The hospitalist should advocate for early mobilization and minimize the duration of hospital stay as appropriate.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing [email protected].
- Francis CW. Clinical practice. prophylaxis for thromboembolism in hospitalized medical patients. N Engl J Med. 2007;356(14):1438-1444. https://doi.org/10.1056/nejmcp067264
- Mahan CE, Borrego ME, Woersching AL, et al. Venous thromboembolism: annualised United States models for total, hospital-acquired and preventable costs utilising long-term attack rates. Thromb Haemost. 2012;108(2):291-302. https://doi.org/10.1160/th12-03-0162
- Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e195S-e226S. https://doi.org/10.1378/chest.11-2296
- Wein L, Wein S, Haas SJ, Shaw J, Krum H. Pharmacological venous thromboembolism prophylaxis in hospitalized medical patients: a meta-analysis of randomized controlled trials. Arch Intern Med. 2007;167(14):1476-1486. https://doi.org/10.1001/archinte.167.14.1476
- Performance Measurement. The Joint Commission. Updated October 26, 2020. Accessed November 8, 2019. http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/VTE.htm
- Venous Thromboembolism Prophylaxis. Centers for Medicare & Medicaid Services. Updated May 6, 2020. Accessed November 8, 2019. https://ecqi.healthit.gov/ecqm/eh/2019/cms108v7
- Cohen AT, Davidson BL, Gallus AS, et al. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial. BMJ. 2006;332(7537):325-329. https://doi.org/10.1136/bmj.38733.466748.7c
- Leizorovicz A, Cohen AT, Turpie AG, et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004;110(7):874-879. https://doi.org/10.1161/01.cir.0000138928.83266.24
- Samama MM, Cohen AT, Darmon JY, et. al. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. prophylaxis in medical patients with enoxaparin study group. N Engl J Med. 1999;341(11):793-800. https://doi.org/10.1056/nejm199909093411103
- Segers AE, Prins MH, Lensing AW, Buller HR. Is contrast venography a valid surrogate outcome measure in venous thromboembolism prevention studies? J Thromb Haemost. 2005;3(5):1099-1102. https://doi.org/10.1111/j.1538-7836.2005.01317.x
- Vardi M, Steinberg M, Haran M, Cohen S. Benefits versus risks of pharmacological prophylaxis to prevent symptomatic venous thromboembolism in unselected medical patients revisited. Meta-analysis of the medical literature. J Thromb Thrombolysis. 2012;34(1):11-19. https://doi.org/10.1007/s11239-012-0730-x
- Flanders SA, Greene MT, Grant P, et al. Hospital performance for pharmacologic venous thromboembolism prophylaxis and rate of venous thromboembolism: a cohort study. JAMA Intern Med. 2014;174(10):1577-1584. https://doi.org/10.1001/jamainternmed.2014.3384
- Loffredo L, Arienti V, Vidili G, et al. Low rate of intrahospital deep venous thrombosis in acutely ill medical patients: results from the AURELIO study. Mayo Clin Proc. 2019;94(1):37-43. https://doi.org/10.1016/j.mayocp.2018.07.020
- Alikhan R, Bedenis R, Cohen AT. Heparin for the prevention of venous thromboembolism in acutely ill medical patients (excluding stroke and myocardial infarction). Cochrane Database Syst Rev. 2014;2014(5):CD003747. https://doi.org/10.1002/14651858.cd003747.pub4
- Popoola VO, Lau BD, Tan E, et al. Nonadministration of medication doses for venous thromboembolism prophylaxis in a cohort of hospitalized patients. Am J Health Syst Pharm. 2018;75(6):392-397. https://doi.org/10.2146/ajhp161057
- Chaudhary R, Damluji A, Batukbhai B, et al. Venous Thromboembolism prophylaxis: inadequate and overprophylaxis when comparing perceived versus calculated risk. Mayo Clin Proc Innov Qual Outcomes. 2017;1(3):242-247. https://doi.org/10.1016/j.mayocpiqo.2017.10.003
- Grant PJ, Conlon A, Chopra V, Flanders SA. Use of venous thromboembolism prophylaxis in hospitalized patients. JAMA Intern Med. 2018;178(8):1122-1124. https://doi.org/10.1001/jamainternmed.2018.2022
- Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2(22):3198-3225. https://doi.org/10.1182/bloodadvances.2018022954
- Pashikanti L, Von Ah D. Impact of early mobilization protocol on the medical-surgical inpatient population: an integrated review of literature. Clin Nurse Spec. 2012;26(2):87-94. https://doi.org/10.1097/nur.0b013e31824590e6

Inspired by the ABIM Foundation’s Choosing Wisel y ® campaign, the “Things We Do for No Reason ™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A hospitalist admits a 68-year-old woman for community-acquired pneumonia with a past medical history of hypertension, gastroesophageal reflux disease, and osteoarthritis. Her hospitalist consults physical therapy to maximize mobility; continues her home medications including pantoprazole, hydrochlorothiazide, and acetaminophen; and initiates antimicrobial therapy with ceftriaxone and azithromycin. The hospital admission order set requires administration of subcutaneous unfractionated heparin for venous thromboembolism chemoprophylaxis.
WHY YOU MIGHT THINK UNIVERSAL CHEMOPROPHYLAXIS IS NECESSARY
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE), ranks among the leading preventable causes of morbidity and mortality in hospitalized patients.1 DVTs can rapidly progress to a PE, which account for 5% to 10% of in-hospital deaths.1 The negative sequelae of in-hospital VTE, including prolonged hospital stay, increased healthcare costs, and greater risks associated with pharmacologic treatment, add $9 to $18.2 billion in US healthcare expenditures each year.2 Various risk-assessment models (RAMs) identify medical patients at high risk for developing VTE based on the presence of risk factors including acute heart failure, prior history of VTE, and reduced mobility.3 Since hospitalization may itself increase the risk for VTE, medical patients often receive universal chemoprophylaxis with anticoagulants such as unfractionated heparin (UFH), low-molecular-weight heparin (LMWH), or fondaparinux.3 A meta-analysis of randomized controlled trials (RCTs) published by Wein et al supports the use of VTE chemoprophylaxis in high-risk patients.4 It showed statistically significant reductions in rates of PE in high-risk hospitalized medical patients with UFH (risk ratio [RR], 0.64; 95% CI, 0.50-0.82) or LMWH chemoprophylaxis (RR, 0.37; 95% CI, 0.21-0.64), compared with controls.
In recognition of the magnitude of the problem, national organizations have emphasized routine chemoprophylaxis for prevention of in-hospital VTE as a top-priority measure for patient safety.5,6 The Joint Commission includes chemoprophylaxis as a quality core metric and failure to adhere to such standards compromises hospital accreditation.5 Since 2008, the Centers for Medicare & Medicaid Services no longer reimburses hospitals for preventable VTE and requires institutions to document the rationale for omitting chemoprophylaxis if not commenced on hospital admission.6
WHY CHEMOPROPHYLAXIS FOR LOW-RISK MEDICAL PATIENTS IS UNNECESSARY
In order to understand why chemoprophylaxis fails to benefit low-risk medical patients, it is necessary to critically examine the benefits identified in trials of high-risk patients. Although RCTs and meta-analysis of chemoprophylaxis have consistently demonstrated a reduction in VTE, prevention of asymptomatic VTE identified on screening with ultrasound or venography accounts for more than 90% of the composite outcome in the three key trials.7-9 Hospitalists do not routinely screen for asymptomatic VTE, and incorporation of these events into composite VTE outcomes inflates the magnitude of benefit gained by chemoprophylaxis. Importantly, the standard of care does not include screening for asymptomatic DVTs, and studies have estimated that only 10% to 15% of asymptomatic DVTs progress to a symptomatic VTE.10
A meta-analysis of trials evaluating unselected general medical patients (ie, not those with specific high-risk conditions such as acute myocardial infarction) did not show a reduction in symptomatic VTE with chemoprophylaxis (odds ratio [OR], 0.59; 95% CI, 0.29-1.23).11 In the meta-analysis by Wein et al, which did include patients with specific high-risk conditions, chemoprophylaxis produced a small absolute risk reduction, resulting in a number needed to treat (NNT) of 345 to prevent one PE.4 This demonstrates that, even in high-risk patients, the magnitude of benefit is small. Population-level data also question the benefit of chemoprophylaxis. Flanders et al stratified 35 Michigan hospitals into high-, moderate-, and low-performance tertiles, with performance based on the rate of chemoprophylaxis use on admission for general medical patients at high-risk for VTE. The authors found no significant difference in the rate of VTE at 90 days among tertiles.12 These findings question the usefulness of universal chemoprophylaxis when applied in a real-world setting.
The high rates of VTE in the absence of chemoprophylaxis reported in historic trials may overestimate the contemporary risk. A 2019 multicenter, observational study examined the rate of hospital-acquired DVT for 1,170 low- and high-risk patients with acute medical illness admitted to the internal medicine ward.13 Of them, 250 (21%) underwent prophylaxis with parenteral anticoagulants (mean Padua Prediction Score, 4.5). The remaining 920 (79%) were not treated with prophylaxis (mean Padua Prediction Score, 2.5). All patients underwent ultrasound at admission and discharge. The average length of stay was 13 days, and just three patients (0.3%) experienced in-hospital DVT, two of whom were receiving chemoprophylaxis. Only one (0.09%) DVT was symptomatic.
It should be emphasized that any evidence favoring chemoprophylaxis comes from studies of patients at high-risk of VTE. No data show benefit for low-risk patients. Therefore, any risk of chemoprophylaxis likely outweighs the benefits in low-risk patients. Importantly, the risks are underappreciated. A 2014 meta-analysis reported an increased risk of major hemorrhage (OR, 1.81; 95% CI, 1.10-2.98; P = .02) in high-risk medically ill patients on chemoprophylaxis.14 This results in a number needed to harm for major bleeding of 336, a value similar to the NNT for benefit reported by Wein et al.4 Heparin-induced thrombocytopenia, a potentially limb- and life-threatening complication of UFH or LMWH exposure, has an overall incidence of 0.3% to 0.7% in hospitalized patients on chemoprophylaxis.3 Finally, the most commonly used chemoprophylaxis medications are administered subcutaneously, resulting in injection site pain. Unsurprisingly, hospitalized patients refuse chemoprophylaxis more frequently than any other medication.15
The negative implications of inappropriate chemoprophylaxis extend beyond direct harms to patients. Poor stratification and overuse results in unnecessary healthcare costs. One single-center retrospective review demonstrated that, after integration of chemoprophylaxis into hospital order sets, 76% of patients received unnecessary administration of chemoprophylaxis, resulting in an annualized expenditure of $77,652.16 This does not take into account costs associated with major bleeds.
Unfortunately, the pendulum has shifted from an era of underprescribing chemoprophylaxis to hospitalized medical patients to one of overprescribing. Data published in 2018 suggest that providers overuse chemoprophylaxis in low-risk medical patients at more than double the rate of underusing it in high-risk patients (57% vs 21%).17
Several national societies, including the often cited American College of Chest Physicians (ACCP) and American Society of Hematology (ASH), provide guidance on the use of VTE chemoprophylaxis in acutely ill medical inpatients.3,18 The ASH guidelines conditionally recommend VTE chemoprophylaxis rather than no chemoprophylaxis.18 However, the guidelines do not provide guidance on a risk-stratified approach and disclose that this recommendation is supported by a low certainty in the evidence of the net health benefit gained.18 Guidelines from ACCP lean towards individualized care and recommend against the use of VTE chemoprophylaxis for hospitalized acutely ill, low-risk medical patients.3
WHAT YOU SHOULD DO INSTEAD
Clinicians should risk stratify using validated RAMs when making a patient-centered treatment plan on admission. The table outlines the most common RAMs with evidence for use in acute medically ill hospitalized patients. Although RAMs have limitations (eg, lack of prospective validation and complexity), the ACCP guidelines advocate for their use.3

Given that immobility independently increases risk for VTE, early mobilization is a simple and cost-effective way to potentially prevent VTE in low-risk patients. In addition to this potential benefit, early mobilization shortens the length of hospital stay, improves functional status and rates of delirium in hospitalized elderly patients, and hastens postoperative recovery after major surgeries.19
RECOMMENDATIONS
- Incorporate a patient-centered, risk-stratified approach to identify low-risk patients. This can be done manually or with use of RAMS embedded in the electronic health record.
- Do not prescribe chemoprophylaxis to low-risk hospitalized medical patients.
- Emphasize the importance of early mobilization in hospitalized patients.
CONCLUSION
In regard to the case, the hospitalist should use a RAM developed for the nonsurgical, non–critically ill patient to determine her need for chemoprophylaxis. Based on the clinical data presented, the three RAMs available would classify the patient as low risk for developing an in-hospital VTE. She should not receive chemoprophylaxis given the lack of data demonstrating benefit in this population. To mitigate the potential risk of bleeding, heparin-induced thrombocytopenia, and painful injections, the hospitalist should discontinue heparin. The hospitalist should advocate for early mobilization and minimize the duration of hospital stay as appropriate.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing [email protected].

Inspired by the ABIM Foundation’s Choosing Wisel y ® campaign, the “Things We Do for No Reason ™” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent clear-cut conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A hospitalist admits a 68-year-old woman for community-acquired pneumonia with a past medical history of hypertension, gastroesophageal reflux disease, and osteoarthritis. Her hospitalist consults physical therapy to maximize mobility; continues her home medications including pantoprazole, hydrochlorothiazide, and acetaminophen; and initiates antimicrobial therapy with ceftriaxone and azithromycin. The hospital admission order set requires administration of subcutaneous unfractionated heparin for venous thromboembolism chemoprophylaxis.
WHY YOU MIGHT THINK UNIVERSAL CHEMOPROPHYLAXIS IS NECESSARY
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE), ranks among the leading preventable causes of morbidity and mortality in hospitalized patients.1 DVTs can rapidly progress to a PE, which account for 5% to 10% of in-hospital deaths.1 The negative sequelae of in-hospital VTE, including prolonged hospital stay, increased healthcare costs, and greater risks associated with pharmacologic treatment, add $9 to $18.2 billion in US healthcare expenditures each year.2 Various risk-assessment models (RAMs) identify medical patients at high risk for developing VTE based on the presence of risk factors including acute heart failure, prior history of VTE, and reduced mobility.3 Since hospitalization may itself increase the risk for VTE, medical patients often receive universal chemoprophylaxis with anticoagulants such as unfractionated heparin (UFH), low-molecular-weight heparin (LMWH), or fondaparinux.3 A meta-analysis of randomized controlled trials (RCTs) published by Wein et al supports the use of VTE chemoprophylaxis in high-risk patients.4 It showed statistically significant reductions in rates of PE in high-risk hospitalized medical patients with UFH (risk ratio [RR], 0.64; 95% CI, 0.50-0.82) or LMWH chemoprophylaxis (RR, 0.37; 95% CI, 0.21-0.64), compared with controls.
In recognition of the magnitude of the problem, national organizations have emphasized routine chemoprophylaxis for prevention of in-hospital VTE as a top-priority measure for patient safety.5,6 The Joint Commission includes chemoprophylaxis as a quality core metric and failure to adhere to such standards compromises hospital accreditation.5 Since 2008, the Centers for Medicare & Medicaid Services no longer reimburses hospitals for preventable VTE and requires institutions to document the rationale for omitting chemoprophylaxis if not commenced on hospital admission.6
WHY CHEMOPROPHYLAXIS FOR LOW-RISK MEDICAL PATIENTS IS UNNECESSARY
In order to understand why chemoprophylaxis fails to benefit low-risk medical patients, it is necessary to critically examine the benefits identified in trials of high-risk patients. Although RCTs and meta-analysis of chemoprophylaxis have consistently demonstrated a reduction in VTE, prevention of asymptomatic VTE identified on screening with ultrasound or venography accounts for more than 90% of the composite outcome in the three key trials.7-9 Hospitalists do not routinely screen for asymptomatic VTE, and incorporation of these events into composite VTE outcomes inflates the magnitude of benefit gained by chemoprophylaxis. Importantly, the standard of care does not include screening for asymptomatic DVTs, and studies have estimated that only 10% to 15% of asymptomatic DVTs progress to a symptomatic VTE.10
A meta-analysis of trials evaluating unselected general medical patients (ie, not those with specific high-risk conditions such as acute myocardial infarction) did not show a reduction in symptomatic VTE with chemoprophylaxis (odds ratio [OR], 0.59; 95% CI, 0.29-1.23).11 In the meta-analysis by Wein et al, which did include patients with specific high-risk conditions, chemoprophylaxis produced a small absolute risk reduction, resulting in a number needed to treat (NNT) of 345 to prevent one PE.4 This demonstrates that, even in high-risk patients, the magnitude of benefit is small. Population-level data also question the benefit of chemoprophylaxis. Flanders et al stratified 35 Michigan hospitals into high-, moderate-, and low-performance tertiles, with performance based on the rate of chemoprophylaxis use on admission for general medical patients at high-risk for VTE. The authors found no significant difference in the rate of VTE at 90 days among tertiles.12 These findings question the usefulness of universal chemoprophylaxis when applied in a real-world setting.
The high rates of VTE in the absence of chemoprophylaxis reported in historic trials may overestimate the contemporary risk. A 2019 multicenter, observational study examined the rate of hospital-acquired DVT for 1,170 low- and high-risk patients with acute medical illness admitted to the internal medicine ward.13 Of them, 250 (21%) underwent prophylaxis with parenteral anticoagulants (mean Padua Prediction Score, 4.5). The remaining 920 (79%) were not treated with prophylaxis (mean Padua Prediction Score, 2.5). All patients underwent ultrasound at admission and discharge. The average length of stay was 13 days, and just three patients (0.3%) experienced in-hospital DVT, two of whom were receiving chemoprophylaxis. Only one (0.09%) DVT was symptomatic.
It should be emphasized that any evidence favoring chemoprophylaxis comes from studies of patients at high-risk of VTE. No data show benefit for low-risk patients. Therefore, any risk of chemoprophylaxis likely outweighs the benefits in low-risk patients. Importantly, the risks are underappreciated. A 2014 meta-analysis reported an increased risk of major hemorrhage (OR, 1.81; 95% CI, 1.10-2.98; P = .02) in high-risk medically ill patients on chemoprophylaxis.14 This results in a number needed to harm for major bleeding of 336, a value similar to the NNT for benefit reported by Wein et al.4 Heparin-induced thrombocytopenia, a potentially limb- and life-threatening complication of UFH or LMWH exposure, has an overall incidence of 0.3% to 0.7% in hospitalized patients on chemoprophylaxis.3 Finally, the most commonly used chemoprophylaxis medications are administered subcutaneously, resulting in injection site pain. Unsurprisingly, hospitalized patients refuse chemoprophylaxis more frequently than any other medication.15
The negative implications of inappropriate chemoprophylaxis extend beyond direct harms to patients. Poor stratification and overuse results in unnecessary healthcare costs. One single-center retrospective review demonstrated that, after integration of chemoprophylaxis into hospital order sets, 76% of patients received unnecessary administration of chemoprophylaxis, resulting in an annualized expenditure of $77,652.16 This does not take into account costs associated with major bleeds.
Unfortunately, the pendulum has shifted from an era of underprescribing chemoprophylaxis to hospitalized medical patients to one of overprescribing. Data published in 2018 suggest that providers overuse chemoprophylaxis in low-risk medical patients at more than double the rate of underusing it in high-risk patients (57% vs 21%).17
Several national societies, including the often cited American College of Chest Physicians (ACCP) and American Society of Hematology (ASH), provide guidance on the use of VTE chemoprophylaxis in acutely ill medical inpatients.3,18 The ASH guidelines conditionally recommend VTE chemoprophylaxis rather than no chemoprophylaxis.18 However, the guidelines do not provide guidance on a risk-stratified approach and disclose that this recommendation is supported by a low certainty in the evidence of the net health benefit gained.18 Guidelines from ACCP lean towards individualized care and recommend against the use of VTE chemoprophylaxis for hospitalized acutely ill, low-risk medical patients.3
WHAT YOU SHOULD DO INSTEAD
Clinicians should risk stratify using validated RAMs when making a patient-centered treatment plan on admission. The table outlines the most common RAMs with evidence for use in acute medically ill hospitalized patients. Although RAMs have limitations (eg, lack of prospective validation and complexity), the ACCP guidelines advocate for their use.3

Given that immobility independently increases risk for VTE, early mobilization is a simple and cost-effective way to potentially prevent VTE in low-risk patients. In addition to this potential benefit, early mobilization shortens the length of hospital stay, improves functional status and rates of delirium in hospitalized elderly patients, and hastens postoperative recovery after major surgeries.19
RECOMMENDATIONS
- Incorporate a patient-centered, risk-stratified approach to identify low-risk patients. This can be done manually or with use of RAMS embedded in the electronic health record.
- Do not prescribe chemoprophylaxis to low-risk hospitalized medical patients.
- Emphasize the importance of early mobilization in hospitalized patients.
CONCLUSION
In regard to the case, the hospitalist should use a RAM developed for the nonsurgical, non–critically ill patient to determine her need for chemoprophylaxis. Based on the clinical data presented, the three RAMs available would classify the patient as low risk for developing an in-hospital VTE. She should not receive chemoprophylaxis given the lack of data demonstrating benefit in this population. To mitigate the potential risk of bleeding, heparin-induced thrombocytopenia, and painful injections, the hospitalist should discontinue heparin. The hospitalist should advocate for early mobilization and minimize the duration of hospital stay as appropriate.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason™”? Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason™” topics by emailing [email protected].
- Francis CW. Clinical practice. prophylaxis for thromboembolism in hospitalized medical patients. N Engl J Med. 2007;356(14):1438-1444. https://doi.org/10.1056/nejmcp067264
- Mahan CE, Borrego ME, Woersching AL, et al. Venous thromboembolism: annualised United States models for total, hospital-acquired and preventable costs utilising long-term attack rates. Thromb Haemost. 2012;108(2):291-302. https://doi.org/10.1160/th12-03-0162
- Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e195S-e226S. https://doi.org/10.1378/chest.11-2296
- Wein L, Wein S, Haas SJ, Shaw J, Krum H. Pharmacological venous thromboembolism prophylaxis in hospitalized medical patients: a meta-analysis of randomized controlled trials. Arch Intern Med. 2007;167(14):1476-1486. https://doi.org/10.1001/archinte.167.14.1476
- Performance Measurement. The Joint Commission. Updated October 26, 2020. Accessed November 8, 2019. http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/VTE.htm
- Venous Thromboembolism Prophylaxis. Centers for Medicare & Medicaid Services. Updated May 6, 2020. Accessed November 8, 2019. https://ecqi.healthit.gov/ecqm/eh/2019/cms108v7
- Cohen AT, Davidson BL, Gallus AS, et al. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial. BMJ. 2006;332(7537):325-329. https://doi.org/10.1136/bmj.38733.466748.7c
- Leizorovicz A, Cohen AT, Turpie AG, et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004;110(7):874-879. https://doi.org/10.1161/01.cir.0000138928.83266.24
- Samama MM, Cohen AT, Darmon JY, et. al. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. prophylaxis in medical patients with enoxaparin study group. N Engl J Med. 1999;341(11):793-800. https://doi.org/10.1056/nejm199909093411103
- Segers AE, Prins MH, Lensing AW, Buller HR. Is contrast venography a valid surrogate outcome measure in venous thromboembolism prevention studies? J Thromb Haemost. 2005;3(5):1099-1102. https://doi.org/10.1111/j.1538-7836.2005.01317.x
- Vardi M, Steinberg M, Haran M, Cohen S. Benefits versus risks of pharmacological prophylaxis to prevent symptomatic venous thromboembolism in unselected medical patients revisited. Meta-analysis of the medical literature. J Thromb Thrombolysis. 2012;34(1):11-19. https://doi.org/10.1007/s11239-012-0730-x
- Flanders SA, Greene MT, Grant P, et al. Hospital performance for pharmacologic venous thromboembolism prophylaxis and rate of venous thromboembolism: a cohort study. JAMA Intern Med. 2014;174(10):1577-1584. https://doi.org/10.1001/jamainternmed.2014.3384
- Loffredo L, Arienti V, Vidili G, et al. Low rate of intrahospital deep venous thrombosis in acutely ill medical patients: results from the AURELIO study. Mayo Clin Proc. 2019;94(1):37-43. https://doi.org/10.1016/j.mayocp.2018.07.020
- Alikhan R, Bedenis R, Cohen AT. Heparin for the prevention of venous thromboembolism in acutely ill medical patients (excluding stroke and myocardial infarction). Cochrane Database Syst Rev. 2014;2014(5):CD003747. https://doi.org/10.1002/14651858.cd003747.pub4
- Popoola VO, Lau BD, Tan E, et al. Nonadministration of medication doses for venous thromboembolism prophylaxis in a cohort of hospitalized patients. Am J Health Syst Pharm. 2018;75(6):392-397. https://doi.org/10.2146/ajhp161057
- Chaudhary R, Damluji A, Batukbhai B, et al. Venous Thromboembolism prophylaxis: inadequate and overprophylaxis when comparing perceived versus calculated risk. Mayo Clin Proc Innov Qual Outcomes. 2017;1(3):242-247. https://doi.org/10.1016/j.mayocpiqo.2017.10.003
- Grant PJ, Conlon A, Chopra V, Flanders SA. Use of venous thromboembolism prophylaxis in hospitalized patients. JAMA Intern Med. 2018;178(8):1122-1124. https://doi.org/10.1001/jamainternmed.2018.2022
- Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2(22):3198-3225. https://doi.org/10.1182/bloodadvances.2018022954
- Pashikanti L, Von Ah D. Impact of early mobilization protocol on the medical-surgical inpatient population: an integrated review of literature. Clin Nurse Spec. 2012;26(2):87-94. https://doi.org/10.1097/nur.0b013e31824590e6
- Francis CW. Clinical practice. prophylaxis for thromboembolism in hospitalized medical patients. N Engl J Med. 2007;356(14):1438-1444. https://doi.org/10.1056/nejmcp067264
- Mahan CE, Borrego ME, Woersching AL, et al. Venous thromboembolism: annualised United States models for total, hospital-acquired and preventable costs utilising long-term attack rates. Thromb Haemost. 2012;108(2):291-302. https://doi.org/10.1160/th12-03-0162
- Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e195S-e226S. https://doi.org/10.1378/chest.11-2296
- Wein L, Wein S, Haas SJ, Shaw J, Krum H. Pharmacological venous thromboembolism prophylaxis in hospitalized medical patients: a meta-analysis of randomized controlled trials. Arch Intern Med. 2007;167(14):1476-1486. https://doi.org/10.1001/archinte.167.14.1476
- Performance Measurement. The Joint Commission. Updated October 26, 2020. Accessed November 8, 2019. http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/VTE.htm
- Venous Thromboembolism Prophylaxis. Centers for Medicare & Medicaid Services. Updated May 6, 2020. Accessed November 8, 2019. https://ecqi.healthit.gov/ecqm/eh/2019/cms108v7
- Cohen AT, Davidson BL, Gallus AS, et al. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial. BMJ. 2006;332(7537):325-329. https://doi.org/10.1136/bmj.38733.466748.7c
- Leizorovicz A, Cohen AT, Turpie AG, et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004;110(7):874-879. https://doi.org/10.1161/01.cir.0000138928.83266.24
- Samama MM, Cohen AT, Darmon JY, et. al. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. prophylaxis in medical patients with enoxaparin study group. N Engl J Med. 1999;341(11):793-800. https://doi.org/10.1056/nejm199909093411103
- Segers AE, Prins MH, Lensing AW, Buller HR. Is contrast venography a valid surrogate outcome measure in venous thromboembolism prevention studies? J Thromb Haemost. 2005;3(5):1099-1102. https://doi.org/10.1111/j.1538-7836.2005.01317.x
- Vardi M, Steinberg M, Haran M, Cohen S. Benefits versus risks of pharmacological prophylaxis to prevent symptomatic venous thromboembolism in unselected medical patients revisited. Meta-analysis of the medical literature. J Thromb Thrombolysis. 2012;34(1):11-19. https://doi.org/10.1007/s11239-012-0730-x
- Flanders SA, Greene MT, Grant P, et al. Hospital performance for pharmacologic venous thromboembolism prophylaxis and rate of venous thromboembolism: a cohort study. JAMA Intern Med. 2014;174(10):1577-1584. https://doi.org/10.1001/jamainternmed.2014.3384
- Loffredo L, Arienti V, Vidili G, et al. Low rate of intrahospital deep venous thrombosis in acutely ill medical patients: results from the AURELIO study. Mayo Clin Proc. 2019;94(1):37-43. https://doi.org/10.1016/j.mayocp.2018.07.020
- Alikhan R, Bedenis R, Cohen AT. Heparin for the prevention of venous thromboembolism in acutely ill medical patients (excluding stroke and myocardial infarction). Cochrane Database Syst Rev. 2014;2014(5):CD003747. https://doi.org/10.1002/14651858.cd003747.pub4
- Popoola VO, Lau BD, Tan E, et al. Nonadministration of medication doses for venous thromboembolism prophylaxis in a cohort of hospitalized patients. Am J Health Syst Pharm. 2018;75(6):392-397. https://doi.org/10.2146/ajhp161057
- Chaudhary R, Damluji A, Batukbhai B, et al. Venous Thromboembolism prophylaxis: inadequate and overprophylaxis when comparing perceived versus calculated risk. Mayo Clin Proc Innov Qual Outcomes. 2017;1(3):242-247. https://doi.org/10.1016/j.mayocpiqo.2017.10.003
- Grant PJ, Conlon A, Chopra V, Flanders SA. Use of venous thromboembolism prophylaxis in hospitalized patients. JAMA Intern Med. 2018;178(8):1122-1124. https://doi.org/10.1001/jamainternmed.2018.2022
- Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2(22):3198-3225. https://doi.org/10.1182/bloodadvances.2018022954
- Pashikanti L, Von Ah D. Impact of early mobilization protocol on the medical-surgical inpatient population: an integrated review of literature. Clin Nurse Spec. 2012;26(2):87-94. https://doi.org/10.1097/nur.0b013e31824590e6
© 2020 Society of Hospital Medicine
Email: [email protected]; Telephone: 267-627-4207; Twitter @theABofPharmaC.
Gender Distribution in Pediatric Hospital Medicine Leadership
There is a growing appreciation of gender disparities in career advancement in medicine. By 2004, approximately 50% of medical school graduates were women, yet considerable differences persist between genders in compensation, faculty rank, and leadership positions.1-3 According to the Association of American Medical Colleges (AAMC), women account for only 25% of full professors, 18% of department chairs, and 18% of medical school deans.1 Women are also underrepresented in other areas of leadership such as division directors, professional society leadership, and hospital executives.4-6
Specialties that are predominantly women, including pediatrics, are not immune to gender disparities. Women represent 71% of pediatric residents1 and currently constitute two-thirds of active pediatricians in the United States.7 However, there is a disproportionately low number of women ascending the pediatric academic ladder, with only 35% of full professors2 and 28% of department chairs being women.1 Pediatrics also was noted to have the fifth-largest gender pay gap across 40 specialties.3 These disparities can contribute to burnout, poorer patient outcomes, and decreased advancement of women known as the “leaky pipeline.”1,8,9
There is some evidence that gender disparities may be improving among younger professionals with increasing percentages of women as leaders and decreasing pay gaps.10,11 These potential positive trends provide hope that fields in medicine early in their development may demonstrate fewer gender disparities. One of the youngest fields of medicine is pediatric hospital medicine (PHM), which officially became a recognized pediatric subspecialty in 2017.12 There is no literature to date describing gender disparities in PHM. We aimed to explore the gender distribution of university-based PHM program leadership and to compare this gender distribution with that seen in the broader field of PHM.
METHODS
This study was Institutional Review Board–approved as non–human subjects research through University of Chicago, Chicago, Illinois. From January to March 2020, the authors performed web-based searches for PHM division directors or program leaders in the United States. Because there is no single database of PHM programs in the United States, we used the AAMC list of Liaison Committee on Medical Education (LCME)–accredited US medical schools; medical schools in Puerto Rico were not included, nor were pending and provisional institutions. If an institution had multiple practice sites for its students, the primary site for third-year medical student clerkship rotations was included. If a medical school had multiple branches, each with its own primary inpatient pediatrics site, these sites were included. If there was no PHM division director, a program leader (lead hospitalist) was substituted and counted as long as the role was formally designated. This leadership role is herein referred to under the umbrella term of “division director.”
We searched medical school web pages, affiliated hospital web pages, and Google. All program leadership information (divisional and fellowship, if present) was confirmed through direct communication with the program, most commonly with division directors, and included name, gender, title, and presence of associate/assistant leader, gender, and title. Associate division directors were only included if it was a formal leadership position. Associate directors of research, quality, etc, were not included due to the limited number of formal positions noted on further review. Of note, the terms “associate” and “assistant” are referring to leadership positions and not academic ranks.
Fellowship leadership was included if affiliated with a US medical school in the primary list. Medical schools with multiple PHM fellowships were included as separate observations. The leadership was confirmed using the methods described above and cross-referenced through the PHM Fellowship Program website. PHM fellowship programs starting in 2020 were included if leadership was determined.
All leadership positions were verified by two authors, and all authors reviewed the master list to identify errors.
To determine the overall gender breakdown in the specialty, we used three estimates: 2019 American Board of Pediatrics (ABP) PHM Board Certification Exam applicants, the 2019 American Academy of Pediatrics Section on Hospital Medicine membership, and a random sample of all PHM faculty in 25% of the programs included in this study.4
Descriptive statistics using 95% confidence intervals for proportions were used. Differences between proportions were evaluated using a two-proportion z test with the null hypothesis that the two proportions are the same and significance set at P < .05.
RESULTS
Of the 150 AAMC LCME–accredited medical school departments of pediatrics evaluated, a total of 142 programs were included; eight programs were excluded due to not providing inpatient pediatric services.
Division Leadership
The proportion of women PHM division directors was 55% (95% CI, 47%-63%) in this sample of 146 leaders from 142 programs (4 programs had coleaders). In the 113 programs with standalone PHM divisions or sections, the proportion of women division directors was 56% (95% CI, 47%-64%). In the 29 hospitalist groups that were not standalone (ie, embedded in another division), the proportion of women leaders was similar at 52% (95% CI, 34%-69%). In 24 programs with 27 formally designated associate directors (1 program had 3 associate directors and 1 program had 2), 81% of associate directors were women (95% CI, 63%-92%).
Fellowship Leadership
A total of 51 PHM fellowship programs had 53 directors (2 had codirectors), and 66% of the fellowship directors were women (95% CI, 53%-77%). A total of 31 programs had 34 assistant directors (3 programs had 2 assistants), and 82% of the assistant fellowship directors were women (95% CI, 66%-92%).
Comparison With the Field at Large
The inaugural ABP PHM board certification exam in 2019 had 1,627 applicants with 70% women (95% CI, 68%-73%) (Suzanne Woods, MD, email communication, December 4, 2019). The American Academy of Pediatrics Section on Hospital Medicine, the largest PHM-specific organization, has 2,299 practicing physician members with 71% women (95% CI, 69%-73%) (Niccole Alexander, email communication, November 25, 2019). Our random sample of 25% of university-based PHM programs contained 1,063 faculty members with 72% women (95% CI, 69%-75%).
The Table provides P values for comparisons of the proportion of women in each of the above-described leadership roles compared to the most conservative estimate of women in the field from the estimates given above (ie, 70%). Compared with the field at large, women appear to be underrepresented as division directors (70% vs 55%; P < .001) but not as fellowship directors (70% vs 66%; P = .5). There is a higher proportion of women in all associate/assistant director roles, compared with the population (82% vs 70%; P = .04).
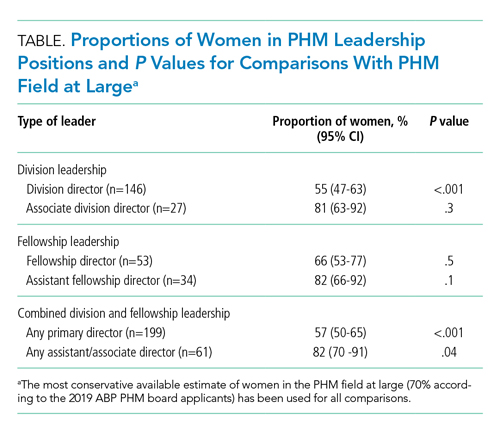
DISCUSSION
We found a significant difference between the proportion of women as PHM division directors (55%) when compared with the proportion of women physicians in PHM (70%), which suggests that women are underrepresented in clinical leadership at university-based pediatric hospitalist programs. Similar findings are described in other specialties, including notably adult hospital medicine.4 Burden et al found that only 16% of hospital medicine program leaders were women despite an equal number of women and men in the field. PHM has a much larger proportion of women, compared with that of hospital medicine, and yet women are still underrepresented as program leaders.
We found no disparities between the proportion of women as PHM fellowship directors and the field at large. These results are similar to those of other studies, which showed a higher number of women in educational leadership roles and lower representation in roles with influence over policy and allocation of resources.13,14 Although the proportion of women in educational roles itself is not a concern, there is evidence that these positions may be undervalued by some institutions, which provide these positions with lower salaries and fewer opportunities for career advancement.13,14
Interestingly, women are well-represented in associate/assistant director roles at both the division and fellowship leader level when comparing the distribution in those roles with that of the PHM field at large. This finding suggests that the pipeline of women is robust and potentially may indicate positive change. Alternatively, this finding may reflect a previously described phenomenon of the “sticky floor” in which women are “stuck” in these supportive roles and do not necessarily advance to higher-impact positions.15 We found a statistically significant higher proportion of women in the combined group of all associate/assistant directors compared with the overall population, which raises the concern that supportive leadership roles may represent “women’s work.”16 Future studies are needed to track whether these women truly advance or whether women are overrepresented in supportive leadership positions at the expense of primary leadership positions.
Adequate representation of women alone is not sufficient to achieve gender equity in medicine. We need to understand why there is a lower representation of women in leadership positions. Some barriers have already been described, including gender bias in promotions,17 higher demands outside of work,18 and lower pay,3 though none are specific to PHM. A further qualitative exploration of PHM leadership would help describe any barriers women in PHM specifically may be facing in their career trajectory. In addition, more information is needed to explore the experience of women with intersectional identities in PHM, especially since they may experience increased bias and discrimination.19
Limitations of this study include the lack of a centralized list of PHM programs and data on PHM workforce. Our three estimates for the proportion of women in PHM were similar at 70%-71%; however, these are only proxies for the true gender distribution of PHM physicians, which is unknown. PHM leadership targets of close to 70% women would be reflective of the field at large; however, institutional variation may exist, and ideally leadership should be diverse and reflective of its faculty members. Our study only describes university-based PHM programs and, therefore, is not necessarily generalizable to nonuniversity programs. Further studies are needed to evaluate any potential differences based on program type. In our study, gender was used in binary terms; however, we acknowledge that gender exists on a spectrum.
CONCLUSION
As a specialty early in development with a robust pipeline of women, PHM is in a unique position to lead the way in gender equity. However, women appear to be underrepresented as division directors at university-based PHM programs. Achieving proportional representation of women leaders is imperative for tapping into the full potential of the community and ensuring that the goals of the field are representative of the population.
Acknowledgment
Special thanks to Lucille Lester, MD, who asked the question that started this road to discovery.
1. Lautenberger DM, Dandar VM. State of Women in Academic Medicine 2018-2019 Exploring Pathways to Equity. AAMC; 2020. Accessed April 10, 2020. https://www.aamc.org/data-reports/data/2018-2019-state-women-academic-medicine-exploring-pathways-equity
2. Table 13: U.S. Medical School Faculty by Sex, Rank, and Department, 2017. AAMC; 2019. Accessed June 25, 2020. https://www.aamc.org/download/486102/data/17table13.pdf
3. 2019 Physician Compensation Report. Doximity; March 2019. Accessed April 11, 2020. https://s3.amazonaws.com/s3.doximity.com/press/doximity_third_annual_physician_compensation_report_round3.pdf
4. Burden M, Frank MG, Keniston A, et al. Gender disparities in leadership and scholarly productivity of academic hospitalists. J Hosp Med. 2015;10(8):481-485. https://doi.org/10.1002/jhm.2340
5. Silver J, Ghalib R, Poorman JA, et al. Analysis of gender equity in leadership of physician-focused medical specialty societies, 2008-2017. JAMA Intern Med. 2019:179(3):433-435. https://doi.org/10.1001/jamainternmed.2018.5303
6. Thomas R, Cooper M, Konar E, et al. Lean In: Women in the Workplace 2019. McKinsey & Company; 2019. Accessed July 1, 2020. https://wiw-report.s3.amazonaws.com/Women_in_the_Workplace_2019.pdf
7. Table 1.3: Number and Percentage of Active Physicians by Sex and Specialty, 2017. AAMC; 2017. Accessed April 12, 2020. https://www.aamc.org/data-reports/workforce/interactive-data/active-physicians-sex-and-specialty-2017
8. Taka F, Nomura K, Horie S, et al. Organizational climate with gender equity and burnout among university academics in Japan. Ind Health. 2016;54(6):480-487. https://doi.org/10.2486/indhealth.2016-0126
9. Tsugawa Y, Jena A, Figueroa J, Orav EJ, Blumenthal DM, Jha AK. Comparison of hospital mortality and readmission rates for medicare patients treated by male vs female physicians. JAMA Intern Med. 2017;177(2):206-213. https://doi.org/10.1001/jamainternmed.2016.7875
10. Bissing MA, Lange EMS, Davila WF, et al. Status of women in academic anesthesiology: a 10-year update. Anesth Analg. 2019;128(1):137-143. https://doi.org/10.1213/ane.0000000000003691
11. Graf N, Brown A, Patten E. The narrowing, but persistent, gender gap in pay. Pew Research Center; March 22, 2019. Accessed April 20, 2020. https://www.pewresearch.org/fact-tank/2019/03/22/gender-pay-gap-facts/
12. American Board of Medical Specialties Officially Recognizes Pediatric Hospital Medicine Subspecialty Certification. News release. American Board of Medical Specialties; November 9, 2016. Accessed June 25, 2020. https://www.abms.org/media/120095/abms-recognizes-pediatric-hospital-medicine-as-a-subspecialty.pdf
13. Hofler LG, Hacker MR, Dodge LE, Schutzberg R, Ricciotti HA. Comparison of women in department leadership in obstetrics and gynecology with other specialties. Obstet Gynecol. 2016;127(3):442-447. https://doi.org/10.1097/aog.0000000000001290
14. Weiss A, Lee KC, Tapia V, et al. Equity in surgical leadership for women: more work to do. Am J Surg. 2014;208:494-498. https://doi.org/10.1016/j.amjsurg.2013.11.005
15. Tesch BJ, Wood HM, Helwig AL, Nattinger AB. Promotion of women physicians in academic medicine. Glass ceiling or sticky floor? JAMA. 1995;273(13):1022-1025.
16. Pelley E, Carnes M. When a specialty becomes “women’s work”: trends in and implications of specialty gender segregation in medicine. Acad Med. 2020;95(10):1499-1506. https://doi.org/10.1097/acm.0000000000003555
17. Steinpreis RE, Anders KA, Ritzke D. The impact of gender on the review of the curricula vitae of job applicants and tenure candidates: a national empirical study. Sex Roles. 1999;41(7):509-528. https://doi.org/10.1023/A:1018839203698
18. Jolly S, Griffith KA, DeCastro R, Stewart A, Ubel P, Jagsi R. Gender differences in time spent on parenting and domestic responsibilities by high-achieving young physician-researchers. Ann Intern Med. 2014;160(5):344-353. https://doi.org/10.7326/m13-0974
19. Ginther DK, Kahn S, Schaffer WT. Gender, race/ethnicity, and National Institutes of Health R01 research awards: is there evidence of a double bind for women of color? Acad Med. 2016;91(8):1098-1107. https://doi.org/10.1097/acm.0000000000001278
There is a growing appreciation of gender disparities in career advancement in medicine. By 2004, approximately 50% of medical school graduates were women, yet considerable differences persist between genders in compensation, faculty rank, and leadership positions.1-3 According to the Association of American Medical Colleges (AAMC), women account for only 25% of full professors, 18% of department chairs, and 18% of medical school deans.1 Women are also underrepresented in other areas of leadership such as division directors, professional society leadership, and hospital executives.4-6
Specialties that are predominantly women, including pediatrics, are not immune to gender disparities. Women represent 71% of pediatric residents1 and currently constitute two-thirds of active pediatricians in the United States.7 However, there is a disproportionately low number of women ascending the pediatric academic ladder, with only 35% of full professors2 and 28% of department chairs being women.1 Pediatrics also was noted to have the fifth-largest gender pay gap across 40 specialties.3 These disparities can contribute to burnout, poorer patient outcomes, and decreased advancement of women known as the “leaky pipeline.”1,8,9
There is some evidence that gender disparities may be improving among younger professionals with increasing percentages of women as leaders and decreasing pay gaps.10,11 These potential positive trends provide hope that fields in medicine early in their development may demonstrate fewer gender disparities. One of the youngest fields of medicine is pediatric hospital medicine (PHM), which officially became a recognized pediatric subspecialty in 2017.12 There is no literature to date describing gender disparities in PHM. We aimed to explore the gender distribution of university-based PHM program leadership and to compare this gender distribution with that seen in the broader field of PHM.
METHODS
This study was Institutional Review Board–approved as non–human subjects research through University of Chicago, Chicago, Illinois. From January to March 2020, the authors performed web-based searches for PHM division directors or program leaders in the United States. Because there is no single database of PHM programs in the United States, we used the AAMC list of Liaison Committee on Medical Education (LCME)–accredited US medical schools; medical schools in Puerto Rico were not included, nor were pending and provisional institutions. If an institution had multiple practice sites for its students, the primary site for third-year medical student clerkship rotations was included. If a medical school had multiple branches, each with its own primary inpatient pediatrics site, these sites were included. If there was no PHM division director, a program leader (lead hospitalist) was substituted and counted as long as the role was formally designated. This leadership role is herein referred to under the umbrella term of “division director.”
We searched medical school web pages, affiliated hospital web pages, and Google. All program leadership information (divisional and fellowship, if present) was confirmed through direct communication with the program, most commonly with division directors, and included name, gender, title, and presence of associate/assistant leader, gender, and title. Associate division directors were only included if it was a formal leadership position. Associate directors of research, quality, etc, were not included due to the limited number of formal positions noted on further review. Of note, the terms “associate” and “assistant” are referring to leadership positions and not academic ranks.
Fellowship leadership was included if affiliated with a US medical school in the primary list. Medical schools with multiple PHM fellowships were included as separate observations. The leadership was confirmed using the methods described above and cross-referenced through the PHM Fellowship Program website. PHM fellowship programs starting in 2020 were included if leadership was determined.
All leadership positions were verified by two authors, and all authors reviewed the master list to identify errors.
To determine the overall gender breakdown in the specialty, we used three estimates: 2019 American Board of Pediatrics (ABP) PHM Board Certification Exam applicants, the 2019 American Academy of Pediatrics Section on Hospital Medicine membership, and a random sample of all PHM faculty in 25% of the programs included in this study.4
Descriptive statistics using 95% confidence intervals for proportions were used. Differences between proportions were evaluated using a two-proportion z test with the null hypothesis that the two proportions are the same and significance set at P < .05.
RESULTS
Of the 150 AAMC LCME–accredited medical school departments of pediatrics evaluated, a total of 142 programs were included; eight programs were excluded due to not providing inpatient pediatric services.
Division Leadership
The proportion of women PHM division directors was 55% (95% CI, 47%-63%) in this sample of 146 leaders from 142 programs (4 programs had coleaders). In the 113 programs with standalone PHM divisions or sections, the proportion of women division directors was 56% (95% CI, 47%-64%). In the 29 hospitalist groups that were not standalone (ie, embedded in another division), the proportion of women leaders was similar at 52% (95% CI, 34%-69%). In 24 programs with 27 formally designated associate directors (1 program had 3 associate directors and 1 program had 2), 81% of associate directors were women (95% CI, 63%-92%).
Fellowship Leadership
A total of 51 PHM fellowship programs had 53 directors (2 had codirectors), and 66% of the fellowship directors were women (95% CI, 53%-77%). A total of 31 programs had 34 assistant directors (3 programs had 2 assistants), and 82% of the assistant fellowship directors were women (95% CI, 66%-92%).
Comparison With the Field at Large
The inaugural ABP PHM board certification exam in 2019 had 1,627 applicants with 70% women (95% CI, 68%-73%) (Suzanne Woods, MD, email communication, December 4, 2019). The American Academy of Pediatrics Section on Hospital Medicine, the largest PHM-specific organization, has 2,299 practicing physician members with 71% women (95% CI, 69%-73%) (Niccole Alexander, email communication, November 25, 2019). Our random sample of 25% of university-based PHM programs contained 1,063 faculty members with 72% women (95% CI, 69%-75%).
The Table provides P values for comparisons of the proportion of women in each of the above-described leadership roles compared to the most conservative estimate of women in the field from the estimates given above (ie, 70%). Compared with the field at large, women appear to be underrepresented as division directors (70% vs 55%; P < .001) but not as fellowship directors (70% vs 66%; P = .5). There is a higher proportion of women in all associate/assistant director roles, compared with the population (82% vs 70%; P = .04).

DISCUSSION
We found a significant difference between the proportion of women as PHM division directors (55%) when compared with the proportion of women physicians in PHM (70%), which suggests that women are underrepresented in clinical leadership at university-based pediatric hospitalist programs. Similar findings are described in other specialties, including notably adult hospital medicine.4 Burden et al found that only 16% of hospital medicine program leaders were women despite an equal number of women and men in the field. PHM has a much larger proportion of women, compared with that of hospital medicine, and yet women are still underrepresented as program leaders.
We found no disparities between the proportion of women as PHM fellowship directors and the field at large. These results are similar to those of other studies, which showed a higher number of women in educational leadership roles and lower representation in roles with influence over policy and allocation of resources.13,14 Although the proportion of women in educational roles itself is not a concern, there is evidence that these positions may be undervalued by some institutions, which provide these positions with lower salaries and fewer opportunities for career advancement.13,14
Interestingly, women are well-represented in associate/assistant director roles at both the division and fellowship leader level when comparing the distribution in those roles with that of the PHM field at large. This finding suggests that the pipeline of women is robust and potentially may indicate positive change. Alternatively, this finding may reflect a previously described phenomenon of the “sticky floor” in which women are “stuck” in these supportive roles and do not necessarily advance to higher-impact positions.15 We found a statistically significant higher proportion of women in the combined group of all associate/assistant directors compared with the overall population, which raises the concern that supportive leadership roles may represent “women’s work.”16 Future studies are needed to track whether these women truly advance or whether women are overrepresented in supportive leadership positions at the expense of primary leadership positions.
Adequate representation of women alone is not sufficient to achieve gender equity in medicine. We need to understand why there is a lower representation of women in leadership positions. Some barriers have already been described, including gender bias in promotions,17 higher demands outside of work,18 and lower pay,3 though none are specific to PHM. A further qualitative exploration of PHM leadership would help describe any barriers women in PHM specifically may be facing in their career trajectory. In addition, more information is needed to explore the experience of women with intersectional identities in PHM, especially since they may experience increased bias and discrimination.19
Limitations of this study include the lack of a centralized list of PHM programs and data on PHM workforce. Our three estimates for the proportion of women in PHM were similar at 70%-71%; however, these are only proxies for the true gender distribution of PHM physicians, which is unknown. PHM leadership targets of close to 70% women would be reflective of the field at large; however, institutional variation may exist, and ideally leadership should be diverse and reflective of its faculty members. Our study only describes university-based PHM programs and, therefore, is not necessarily generalizable to nonuniversity programs. Further studies are needed to evaluate any potential differences based on program type. In our study, gender was used in binary terms; however, we acknowledge that gender exists on a spectrum.
CONCLUSION
As a specialty early in development with a robust pipeline of women, PHM is in a unique position to lead the way in gender equity. However, women appear to be underrepresented as division directors at university-based PHM programs. Achieving proportional representation of women leaders is imperative for tapping into the full potential of the community and ensuring that the goals of the field are representative of the population.
Acknowledgment
Special thanks to Lucille Lester, MD, who asked the question that started this road to discovery.
There is a growing appreciation of gender disparities in career advancement in medicine. By 2004, approximately 50% of medical school graduates were women, yet considerable differences persist between genders in compensation, faculty rank, and leadership positions.1-3 According to the Association of American Medical Colleges (AAMC), women account for only 25% of full professors, 18% of department chairs, and 18% of medical school deans.1 Women are also underrepresented in other areas of leadership such as division directors, professional society leadership, and hospital executives.4-6
Specialties that are predominantly women, including pediatrics, are not immune to gender disparities. Women represent 71% of pediatric residents1 and currently constitute two-thirds of active pediatricians in the United States.7 However, there is a disproportionately low number of women ascending the pediatric academic ladder, with only 35% of full professors2 and 28% of department chairs being women.1 Pediatrics also was noted to have the fifth-largest gender pay gap across 40 specialties.3 These disparities can contribute to burnout, poorer patient outcomes, and decreased advancement of women known as the “leaky pipeline.”1,8,9
There is some evidence that gender disparities may be improving among younger professionals with increasing percentages of women as leaders and decreasing pay gaps.10,11 These potential positive trends provide hope that fields in medicine early in their development may demonstrate fewer gender disparities. One of the youngest fields of medicine is pediatric hospital medicine (PHM), which officially became a recognized pediatric subspecialty in 2017.12 There is no literature to date describing gender disparities in PHM. We aimed to explore the gender distribution of university-based PHM program leadership and to compare this gender distribution with that seen in the broader field of PHM.
METHODS
This study was Institutional Review Board–approved as non–human subjects research through University of Chicago, Chicago, Illinois. From January to March 2020, the authors performed web-based searches for PHM division directors or program leaders in the United States. Because there is no single database of PHM programs in the United States, we used the AAMC list of Liaison Committee on Medical Education (LCME)–accredited US medical schools; medical schools in Puerto Rico were not included, nor were pending and provisional institutions. If an institution had multiple practice sites for its students, the primary site for third-year medical student clerkship rotations was included. If a medical school had multiple branches, each with its own primary inpatient pediatrics site, these sites were included. If there was no PHM division director, a program leader (lead hospitalist) was substituted and counted as long as the role was formally designated. This leadership role is herein referred to under the umbrella term of “division director.”
We searched medical school web pages, affiliated hospital web pages, and Google. All program leadership information (divisional and fellowship, if present) was confirmed through direct communication with the program, most commonly with division directors, and included name, gender, title, and presence of associate/assistant leader, gender, and title. Associate division directors were only included if it was a formal leadership position. Associate directors of research, quality, etc, were not included due to the limited number of formal positions noted on further review. Of note, the terms “associate” and “assistant” are referring to leadership positions and not academic ranks.
Fellowship leadership was included if affiliated with a US medical school in the primary list. Medical schools with multiple PHM fellowships were included as separate observations. The leadership was confirmed using the methods described above and cross-referenced through the PHM Fellowship Program website. PHM fellowship programs starting in 2020 were included if leadership was determined.
All leadership positions were verified by two authors, and all authors reviewed the master list to identify errors.
To determine the overall gender breakdown in the specialty, we used three estimates: 2019 American Board of Pediatrics (ABP) PHM Board Certification Exam applicants, the 2019 American Academy of Pediatrics Section on Hospital Medicine membership, and a random sample of all PHM faculty in 25% of the programs included in this study.4
Descriptive statistics using 95% confidence intervals for proportions were used. Differences between proportions were evaluated using a two-proportion z test with the null hypothesis that the two proportions are the same and significance set at P < .05.
RESULTS
Of the 150 AAMC LCME–accredited medical school departments of pediatrics evaluated, a total of 142 programs were included; eight programs were excluded due to not providing inpatient pediatric services.
Division Leadership
The proportion of women PHM division directors was 55% (95% CI, 47%-63%) in this sample of 146 leaders from 142 programs (4 programs had coleaders). In the 113 programs with standalone PHM divisions or sections, the proportion of women division directors was 56% (95% CI, 47%-64%). In the 29 hospitalist groups that were not standalone (ie, embedded in another division), the proportion of women leaders was similar at 52% (95% CI, 34%-69%). In 24 programs with 27 formally designated associate directors (1 program had 3 associate directors and 1 program had 2), 81% of associate directors were women (95% CI, 63%-92%).
Fellowship Leadership
A total of 51 PHM fellowship programs had 53 directors (2 had codirectors), and 66% of the fellowship directors were women (95% CI, 53%-77%). A total of 31 programs had 34 assistant directors (3 programs had 2 assistants), and 82% of the assistant fellowship directors were women (95% CI, 66%-92%).
Comparison With the Field at Large
The inaugural ABP PHM board certification exam in 2019 had 1,627 applicants with 70% women (95% CI, 68%-73%) (Suzanne Woods, MD, email communication, December 4, 2019). The American Academy of Pediatrics Section on Hospital Medicine, the largest PHM-specific organization, has 2,299 practicing physician members with 71% women (95% CI, 69%-73%) (Niccole Alexander, email communication, November 25, 2019). Our random sample of 25% of university-based PHM programs contained 1,063 faculty members with 72% women (95% CI, 69%-75%).
The Table provides P values for comparisons of the proportion of women in each of the above-described leadership roles compared to the most conservative estimate of women in the field from the estimates given above (ie, 70%). Compared with the field at large, women appear to be underrepresented as division directors (70% vs 55%; P < .001) but not as fellowship directors (70% vs 66%; P = .5). There is a higher proportion of women in all associate/assistant director roles, compared with the population (82% vs 70%; P = .04).

DISCUSSION
We found a significant difference between the proportion of women as PHM division directors (55%) when compared with the proportion of women physicians in PHM (70%), which suggests that women are underrepresented in clinical leadership at university-based pediatric hospitalist programs. Similar findings are described in other specialties, including notably adult hospital medicine.4 Burden et al found that only 16% of hospital medicine program leaders were women despite an equal number of women and men in the field. PHM has a much larger proportion of women, compared with that of hospital medicine, and yet women are still underrepresented as program leaders.
We found no disparities between the proportion of women as PHM fellowship directors and the field at large. These results are similar to those of other studies, which showed a higher number of women in educational leadership roles and lower representation in roles with influence over policy and allocation of resources.13,14 Although the proportion of women in educational roles itself is not a concern, there is evidence that these positions may be undervalued by some institutions, which provide these positions with lower salaries and fewer opportunities for career advancement.13,14
Interestingly, women are well-represented in associate/assistant director roles at both the division and fellowship leader level when comparing the distribution in those roles with that of the PHM field at large. This finding suggests that the pipeline of women is robust and potentially may indicate positive change. Alternatively, this finding may reflect a previously described phenomenon of the “sticky floor” in which women are “stuck” in these supportive roles and do not necessarily advance to higher-impact positions.15 We found a statistically significant higher proportion of women in the combined group of all associate/assistant directors compared with the overall population, which raises the concern that supportive leadership roles may represent “women’s work.”16 Future studies are needed to track whether these women truly advance or whether women are overrepresented in supportive leadership positions at the expense of primary leadership positions.
Adequate representation of women alone is not sufficient to achieve gender equity in medicine. We need to understand why there is a lower representation of women in leadership positions. Some barriers have already been described, including gender bias in promotions,17 higher demands outside of work,18 and lower pay,3 though none are specific to PHM. A further qualitative exploration of PHM leadership would help describe any barriers women in PHM specifically may be facing in their career trajectory. In addition, more information is needed to explore the experience of women with intersectional identities in PHM, especially since they may experience increased bias and discrimination.19
Limitations of this study include the lack of a centralized list of PHM programs and data on PHM workforce. Our three estimates for the proportion of women in PHM were similar at 70%-71%; however, these are only proxies for the true gender distribution of PHM physicians, which is unknown. PHM leadership targets of close to 70% women would be reflective of the field at large; however, institutional variation may exist, and ideally leadership should be diverse and reflective of its faculty members. Our study only describes university-based PHM programs and, therefore, is not necessarily generalizable to nonuniversity programs. Further studies are needed to evaluate any potential differences based on program type. In our study, gender was used in binary terms; however, we acknowledge that gender exists on a spectrum.
CONCLUSION
As a specialty early in development with a robust pipeline of women, PHM is in a unique position to lead the way in gender equity. However, women appear to be underrepresented as division directors at university-based PHM programs. Achieving proportional representation of women leaders is imperative for tapping into the full potential of the community and ensuring that the goals of the field are representative of the population.
Acknowledgment
Special thanks to Lucille Lester, MD, who asked the question that started this road to discovery.
1. Lautenberger DM, Dandar VM. State of Women in Academic Medicine 2018-2019 Exploring Pathways to Equity. AAMC; 2020. Accessed April 10, 2020. https://www.aamc.org/data-reports/data/2018-2019-state-women-academic-medicine-exploring-pathways-equity
2. Table 13: U.S. Medical School Faculty by Sex, Rank, and Department, 2017. AAMC; 2019. Accessed June 25, 2020. https://www.aamc.org/download/486102/data/17table13.pdf
3. 2019 Physician Compensation Report. Doximity; March 2019. Accessed April 11, 2020. https://s3.amazonaws.com/s3.doximity.com/press/doximity_third_annual_physician_compensation_report_round3.pdf
4. Burden M, Frank MG, Keniston A, et al. Gender disparities in leadership and scholarly productivity of academic hospitalists. J Hosp Med. 2015;10(8):481-485. https://doi.org/10.1002/jhm.2340
5. Silver J, Ghalib R, Poorman JA, et al. Analysis of gender equity in leadership of physician-focused medical specialty societies, 2008-2017. JAMA Intern Med. 2019:179(3):433-435. https://doi.org/10.1001/jamainternmed.2018.5303
6. Thomas R, Cooper M, Konar E, et al. Lean In: Women in the Workplace 2019. McKinsey & Company; 2019. Accessed July 1, 2020. https://wiw-report.s3.amazonaws.com/Women_in_the_Workplace_2019.pdf
7. Table 1.3: Number and Percentage of Active Physicians by Sex and Specialty, 2017. AAMC; 2017. Accessed April 12, 2020. https://www.aamc.org/data-reports/workforce/interactive-data/active-physicians-sex-and-specialty-2017
8. Taka F, Nomura K, Horie S, et al. Organizational climate with gender equity and burnout among university academics in Japan. Ind Health. 2016;54(6):480-487. https://doi.org/10.2486/indhealth.2016-0126
9. Tsugawa Y, Jena A, Figueroa J, Orav EJ, Blumenthal DM, Jha AK. Comparison of hospital mortality and readmission rates for medicare patients treated by male vs female physicians. JAMA Intern Med. 2017;177(2):206-213. https://doi.org/10.1001/jamainternmed.2016.7875
10. Bissing MA, Lange EMS, Davila WF, et al. Status of women in academic anesthesiology: a 10-year update. Anesth Analg. 2019;128(1):137-143. https://doi.org/10.1213/ane.0000000000003691
11. Graf N, Brown A, Patten E. The narrowing, but persistent, gender gap in pay. Pew Research Center; March 22, 2019. Accessed April 20, 2020. https://www.pewresearch.org/fact-tank/2019/03/22/gender-pay-gap-facts/
12. American Board of Medical Specialties Officially Recognizes Pediatric Hospital Medicine Subspecialty Certification. News release. American Board of Medical Specialties; November 9, 2016. Accessed June 25, 2020. https://www.abms.org/media/120095/abms-recognizes-pediatric-hospital-medicine-as-a-subspecialty.pdf
13. Hofler LG, Hacker MR, Dodge LE, Schutzberg R, Ricciotti HA. Comparison of women in department leadership in obstetrics and gynecology with other specialties. Obstet Gynecol. 2016;127(3):442-447. https://doi.org/10.1097/aog.0000000000001290
14. Weiss A, Lee KC, Tapia V, et al. Equity in surgical leadership for women: more work to do. Am J Surg. 2014;208:494-498. https://doi.org/10.1016/j.amjsurg.2013.11.005
15. Tesch BJ, Wood HM, Helwig AL, Nattinger AB. Promotion of women physicians in academic medicine. Glass ceiling or sticky floor? JAMA. 1995;273(13):1022-1025.
16. Pelley E, Carnes M. When a specialty becomes “women’s work”: trends in and implications of specialty gender segregation in medicine. Acad Med. 2020;95(10):1499-1506. https://doi.org/10.1097/acm.0000000000003555
17. Steinpreis RE, Anders KA, Ritzke D. The impact of gender on the review of the curricula vitae of job applicants and tenure candidates: a national empirical study. Sex Roles. 1999;41(7):509-528. https://doi.org/10.1023/A:1018839203698
18. Jolly S, Griffith KA, DeCastro R, Stewart A, Ubel P, Jagsi R. Gender differences in time spent on parenting and domestic responsibilities by high-achieving young physician-researchers. Ann Intern Med. 2014;160(5):344-353. https://doi.org/10.7326/m13-0974
19. Ginther DK, Kahn S, Schaffer WT. Gender, race/ethnicity, and National Institutes of Health R01 research awards: is there evidence of a double bind for women of color? Acad Med. 2016;91(8):1098-1107. https://doi.org/10.1097/acm.0000000000001278
1. Lautenberger DM, Dandar VM. State of Women in Academic Medicine 2018-2019 Exploring Pathways to Equity. AAMC; 2020. Accessed April 10, 2020. https://www.aamc.org/data-reports/data/2018-2019-state-women-academic-medicine-exploring-pathways-equity
2. Table 13: U.S. Medical School Faculty by Sex, Rank, and Department, 2017. AAMC; 2019. Accessed June 25, 2020. https://www.aamc.org/download/486102/data/17table13.pdf
3. 2019 Physician Compensation Report. Doximity; March 2019. Accessed April 11, 2020. https://s3.amazonaws.com/s3.doximity.com/press/doximity_third_annual_physician_compensation_report_round3.pdf
4. Burden M, Frank MG, Keniston A, et al. Gender disparities in leadership and scholarly productivity of academic hospitalists. J Hosp Med. 2015;10(8):481-485. https://doi.org/10.1002/jhm.2340
5. Silver J, Ghalib R, Poorman JA, et al. Analysis of gender equity in leadership of physician-focused medical specialty societies, 2008-2017. JAMA Intern Med. 2019:179(3):433-435. https://doi.org/10.1001/jamainternmed.2018.5303
6. Thomas R, Cooper M, Konar E, et al. Lean In: Women in the Workplace 2019. McKinsey & Company; 2019. Accessed July 1, 2020. https://wiw-report.s3.amazonaws.com/Women_in_the_Workplace_2019.pdf
7. Table 1.3: Number and Percentage of Active Physicians by Sex and Specialty, 2017. AAMC; 2017. Accessed April 12, 2020. https://www.aamc.org/data-reports/workforce/interactive-data/active-physicians-sex-and-specialty-2017
8. Taka F, Nomura K, Horie S, et al. Organizational climate with gender equity and burnout among university academics in Japan. Ind Health. 2016;54(6):480-487. https://doi.org/10.2486/indhealth.2016-0126
9. Tsugawa Y, Jena A, Figueroa J, Orav EJ, Blumenthal DM, Jha AK. Comparison of hospital mortality and readmission rates for medicare patients treated by male vs female physicians. JAMA Intern Med. 2017;177(2):206-213. https://doi.org/10.1001/jamainternmed.2016.7875
10. Bissing MA, Lange EMS, Davila WF, et al. Status of women in academic anesthesiology: a 10-year update. Anesth Analg. 2019;128(1):137-143. https://doi.org/10.1213/ane.0000000000003691
11. Graf N, Brown A, Patten E. The narrowing, but persistent, gender gap in pay. Pew Research Center; March 22, 2019. Accessed April 20, 2020. https://www.pewresearch.org/fact-tank/2019/03/22/gender-pay-gap-facts/
12. American Board of Medical Specialties Officially Recognizes Pediatric Hospital Medicine Subspecialty Certification. News release. American Board of Medical Specialties; November 9, 2016. Accessed June 25, 2020. https://www.abms.org/media/120095/abms-recognizes-pediatric-hospital-medicine-as-a-subspecialty.pdf
13. Hofler LG, Hacker MR, Dodge LE, Schutzberg R, Ricciotti HA. Comparison of women in department leadership in obstetrics and gynecology with other specialties. Obstet Gynecol. 2016;127(3):442-447. https://doi.org/10.1097/aog.0000000000001290
14. Weiss A, Lee KC, Tapia V, et al. Equity in surgical leadership for women: more work to do. Am J Surg. 2014;208:494-498. https://doi.org/10.1016/j.amjsurg.2013.11.005
15. Tesch BJ, Wood HM, Helwig AL, Nattinger AB. Promotion of women physicians in academic medicine. Glass ceiling or sticky floor? JAMA. 1995;273(13):1022-1025.
16. Pelley E, Carnes M. When a specialty becomes “women’s work”: trends in and implications of specialty gender segregation in medicine. Acad Med. 2020;95(10):1499-1506. https://doi.org/10.1097/acm.0000000000003555
17. Steinpreis RE, Anders KA, Ritzke D. The impact of gender on the review of the curricula vitae of job applicants and tenure candidates: a national empirical study. Sex Roles. 1999;41(7):509-528. https://doi.org/10.1023/A:1018839203698
18. Jolly S, Griffith KA, DeCastro R, Stewart A, Ubel P, Jagsi R. Gender differences in time spent on parenting and domestic responsibilities by high-achieving young physician-researchers. Ann Intern Med. 2014;160(5):344-353. https://doi.org/10.7326/m13-0974
19. Ginther DK, Kahn S, Schaffer WT. Gender, race/ethnicity, and National Institutes of Health R01 research awards: is there evidence of a double bind for women of color? Acad Med. 2016;91(8):1098-1107. https://doi.org/10.1097/acm.0000000000001278
© 2021 Society of Hospital Medicine